
IPCS INTERNATIONAL PROGRAMME ON CHEMICAL SAFETY
Health and Safety Guide No. 26
PYRROLIZIDINE ALKALOIDS
HEALTH AND SAFETY GUIDE
UNITED NATIONS ENVIRONMENT PROGRAMME
INTERNATIONAL LABOUR ORGANISATION
WORLD HEALTH ORGANIZATION
WORLD HEALTH ORGANIZATION, GENEVA 1989
This is a companion volume to Environmental Health Criteria 80:
Pyrrolizidine Alkaloids.
Published by the World Health Organization for the International
Programme on Chemical Safety (a collaborative programme of the United
Nations Environment Programme, the International Labour Organisation,
and the World Health Organization)
This report contains the collective views of an international group of
experts and does not necessarily represent the decisions or the stated
policy of the United Nations Environment Programme, the International
Labour Organisation, or the World Health Organization
ISBN 92 4 154347 8
ISSN 0259-7268
The World Health Organization welcomes requests for permission to
reproduce or translate its publications, in part or in full.
Applications and enquiries should be addressed to the Office of
Publications, World Health Organization, Geneva, Switzerland, which
will be glad to provide the latest information on any changes made to
the text, plans for new editions, and reprints and translations
already available.
(c) World Health Organization 1989
Publications of the World Health Organization enjoy copyright
protection in accordance with the provisions of Protocol 2 of the
Universal Copyright Convention. All rights reserved.
The designations employed and the presentation of the material in this
publication do not imply the expression of any opinion whatsoever on
the part of the Secretariat of the World Health Organization
concerning the legal status of any country, territory, city or area or
of its authorities, or concerning the delimitation of its frontiers or
boundaries.
The mention of specific companies or of certain manufacturers'
products does not imply that they are endorsed or recommended by the
World Health Organization in preference to others of a similar nature
that are not mentioned. Errors and omissions excepted, the names of
proprietary products are distinguished by initial capital letters.
CONTENTS
INTRODUCTION
1. PRODUCT IDENTITY AND USES
1.1. Identity
1.2. Physical and chemical properties
1.3. Uses
2. SUMMARY AND EVALUATION
2.1. Human exposure to pyrrolizidine alkaloids
2.2. Metabolism and excretion
2.3. Effects on experimental animals
2.4. Effects on human health
2.5. Effects on the environment
3. RISK EVALUATION
4. PREVENTIVE MEASURES AND MEDICAL ADVICE
4.1. Main hazards for man and the environment
4.2. Prevention of PA poisoning
4.3. Advice to physicians
4.4. Precautionary measures to protect the environment and to
prevent disease
4.5. First aid
4.6. Medical management
5. CURRENT REGULATIONS, GUIDELINES, AND STANDARDS
INTRODUCTION
The Environmental Health Criteria (EHC) documents produced by the
International Programme on Chemical Safety include an assessment of
the effects on the environment and on human health of exposure to a
chemical or combination of chemicals, or physical or biological
agents. They also provide guidelines for setting exposure limits.
The purpose of a Health and Safety Guide is to facilitate the
application of these guidelines in national chemical safety
programmes. The first three sections of a Health and Safety Guide
highlight the relevant technical information in the corresponding EHC.
Section 4 includes advice on preventive and protective measures.
The target readership includes occupational health services, those in
ministries, governmental agencies, industry, and trade unions who are
involved in the safe use of chemicals and the avoidance of
environmental health hazards, and those wanting more information on
this topic. An attempt has been made to use only terms that will be
familiar to the intended user. However, sections 1 and 2 inevitably
contain some technical terms. Further background information on the
subject of this Guide may be found in Environmental Health Criteria
80: Pyrrolizidine Alkaloids.
Revision of the information in this Guide will take place in due
course, and the eventual aim is to use standardized terminology.
Comments on any difficulties encountered in using the Guide would be
very helpful and should be addressed to:
The Manager
International Programme on Chemical Safety
Division of Environmental Health
World Health Organization
1211 Geneva 27
Switzerland
THE INFORMATION IN THIS GUIDE SHOULD BE CONSIDERED AS A STARTING POINT
TO A COMPREHENSIVE HEALTH AND SAFETY PROGRAMME
1. PRODUCT IDENTITY AND USES
1.1 Identity
The toxic pyrrolizidine alkaloids (PAs) are a large group of related
compounds (about 160 are known) that occur in plants, mainly in
species of Crotalaria (Leguminosae), Senecio and related genera
(Compositae), Heliotropium, Trichodesma, Symphytum, Echium, and
other genera of the Boraginaceae. The chemical structures of some
alkaloids that are important in relation to human disease are shown
below.
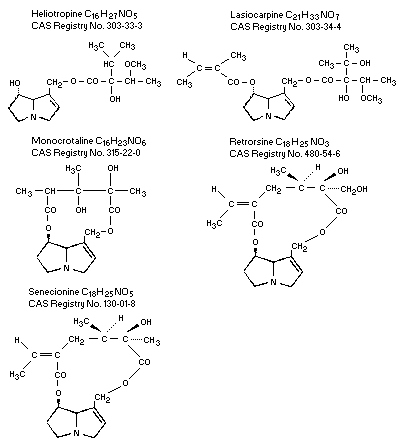 1.2 Physical and Chemical Properties
The pure alkaloids are mostly crystalline solids; some are gums or
amorphous solids. Some are only slightly soluble in water, but all
dissolve when neutralized with acid. They occur in the plants partly
as N-oxides, which are water soluble. The alkaloids are fairly
stable, but are subject to hydrolysis in alkaline solution and to
enzymatic decomposition. The latter occurs in some plant species
during wilting and drying. The stability of the alkaloids when the
plants are cooked is not known.
1.3 Uses
One alkaloid, monocrotaline, is marketed commercially as a fine
chemical for research purposes. Another, indicine N-oxide, is being
tested as an antitumour drug in human beings.
2. SUMMARY AND CONCLUSIONS
2.1 Human Exposure to Pyrrolizidine Alkaloids
The contamination of cereal grains with seeds of PA-containing plants
has caused epidemics of human poisoning in four countries. The plants
involved were species of Heliotropium, Trichodesma, Crotalaria, and
Senecio. Some of these plants thrive under arid conditions and their
growth may be favoured by drought. Another important form of exposure
is the use of PA-containing herbs as medicines, food, or beverages;
this has resulted in occasional cases of human poisoning. The most
important of such PA-containing plants are species of Heliotropium,
Crotalaria, Senecio, and Symphytum. Symphytum species (comfrey)
are also available in the form of ointments or as digestive aids in
the form of capsules.
Low-level exposure may occur in some countries through the presence of
PAs in foods, such as honey and milk, but no reports of human
poisoning caused through these media are available.
2.2 Metabolism and Excretion
PAs are readily absorbed from the digestive tract and cause harmful
effects only after undergoing activation in the liver to toxic
metabolites. Effects include a variety of changes leading to permanent
damage to genes and chromosomes, the ability of the cells to divide,
or to the development of cancer, and even cell death. The alkaloids
are quickly converted to harmless compounds and are largely cleared
from the body within a few hours of absorption, so that no traces of
the ingested PAs or their breakdown products are detectable in the
body tissues and fluids. There are no indications that the alkaloids
themselves accumulate in animal tissues, but their effects are
cumulative, even at low rates of intake. The proportion excreted in
urine varies according to the water solubility of the alkaloid.
Estimates of the total intake over a long period can only be
approximate.
2.3 Effects on Experimental Animals
The acute toxicity of PAs varies widely. The rat LD50s of most
alkaloids known to be significant for human health are in the range of
34-300 mg/kg, though some approach 1000 mg/kg. When ingested, the
toxicity of the N-oxide of an alkaloid is similar to that of the
parent alkaloid.
PAs are toxic for a wide variety of domestic, laboratory, and farm
animals, pigs and poultry being the most sensitive of farm animals
followed by horses and cattle, and sheep and goats, which are the most
resistant; sheep and goats are affected only after extended periods of
exposure.
The toxic effects of most PAs occur primarily and mainly in the liver.
Some PAs are particularly prone to cause damage in the lungs,
principally to the blood vessels, resulting in a rise in blood
pressure in the lungs, which leads to secondary effects on the
functioning of the right side of the heart. The kidneys and some other
organs are less commonly affected. The brain is the principal target
organ of one or two PAs.
The effects of PAs in animals may be acute or chronic, depending on
the dose level and the period of survival after exposure. In acute
poisoning, death occurs within about 7 days, due to severe liver
damage. Chronic liver damage may follow administration of a single
sublethal dose or of repeated low doses.
PAs produce aberrations in the chromosomes in plant and mammalian
cells and induce changes in genes that are perpetuated in subsequent
cell divisions in mutagenesis test systems. Several PAs have been
shown to be capable of producing cancer, chiefly in the liver in rats.
2.4 Effects on Human Health
Although all age groups are affected, children are particularly
vulnerable to the effects of PAs. The symptoms, which are generally
acute in onset, are characterized by upper abdominal discomfort that
develops rapidly and progresses to swelling of the abdomen resulting
in increased girth, sometimes accompanied by a reduction in the
quantity of urine excreted and swelling of the feet. The disease is
called veno-occlusive disease (VOD) because of the characteristic
obstruction of the small venous blood channels that carry blood from
the liver back to the heart. The disease often progresses rapidly and
mortality is high. There may be vomiting of blood in advanced stages
of the disease. While many patients recover, the disease may continue
for a long time in others resulting in a severely scarred liver - a
condition called cirrhosis. Some patients may have only vague symptoms
and the only sign of the disease may be persistent enlargement of the
liver.
The liver is usually the target organ but, in an epidemic caused by
contamination of the staple cereal with the seeds of Trichodesma,
the brain and the nervous system were mainly affected.
Chromosome aberrations have been reported in the blood cells of
children affected by VOD but, as yet, there is no evidence pointing to
an increased incidence of cancer of the liver or other organs or
congenital anomalies in the newborn offspring of patients exposed to
PAs.
2.5 Effects on the Environment
Plants containing PAs are likely to grow as weeds among staple food
crops and pastures, especially following drought, and consumption of
such crops can cause large scale outbreaks of toxic disease in both
man and farm animals. Little is known about the effects on wildlife
but, in one report, the death of deer was ascribed to their grazing on
toxic plants.
PAs are believed to be biodegradable so that water supplies are not
suspect.
3. RISK EVALUATION
No-observed-adverse-effect levels have not been established in
experimental animal studies with PAs. Estimates of intakes causing
toxic effects in human beings indicate that they are more sensitive
than rats and domestic animals. Rats dosed with lasiocarpine at a rate
equivalent to 0.2 mg/kg body weight per day (equivalent to about
2 mg/kg in the diet) developed tumours. Pigs fed monocrotaline at
1.8 mg/kg of feed (equivalent to about 0.08 mg/kg body weight per day)
developed chronic liver damage in several months. The lowest intake
rate causing VOD in a human being was estimated to be 0.015 mg/kg body
weight per day, and was the result of self-medication with a comfrey
preparation. In view of the established ability of some PAs to produce
cancer in rats, plant products containing them should not be eaten or
drunk.
4. PREVENTIVE MEASURES AND MEDICAL ADVICE
4.1 Main Hazards for Man and the Environment
Consumption of contaminated grain or the use of PA-containing plants
as herbal medicines, beverages, or food by man, or grazing on
contaminated pastures by animals, may cause acute or chronic disease.
The principal target organ in man and most animals is the liver,
resulting in collection of fluid in the abdomen and swelling of feet,
etc. In experimental animals, administration of PAs has been known to
produce cancer. Though there is no proof yet of cancer developing in
man, such a possibility cannot be entirely ruled out.
4.2 Prevention of PA Poisoning
Prevention of exposure is the only effective method of limiting
toxicity due to PAs. Even low doses over a period of time may present
a health risk and exposure should be avoided or minimized as far as
possible. Measures are required at several action points.
4.3 Advice to Physicians
Occurrence of symptoms of rapidly increasing swelling of the abdomen
accompanied by dragging discomfort in the right upper abdomen,
particularly if affecting more than one member of the family or
several members of a community over a limited period and geographical
area, should arouse suspicion. The diet of such persons should be
examined for possible contamination with the seeds of toxic plants and
the affected persons should be questioned regarding the possible
intake of herbal preparations. If any such history is positive, the
suspected toxic seeds/herbs should be analysed for the presence of PAs
using a simple field test, which can be carried out in a not too
highly specialized chemical laboratory.
Examination of the liver by needle biopsy in patients or at post-
mortem examination is most likely to provide the crucial clue.
Haemorrhagic centrilobular necrosis and occlusion of the hepatic vein
radicles in the liver are the hallmarks of the disease, if found in
patients with the symptoms described above. Needle biopsy of the liver
should be performed only in a properly equipped hospital with
facilities for blood transfusion.
4.4 Precautionary Measures to Protect the Environment and to Prevent
Disease
(a) Toxic PA-containing flora growing in the region, particularly
those that may contaminate cereal grain crops, and those that are used
locally as herbal foods, beverages, or medicines, whether grown
locally or imported, should be identified.
(b) Appropriate agrotechnical practices should be followed for the
prevention/control of the growth of PA-containing plants among cereal
food crops and pastures.
(c) Appropriate systems should be developed for the routine monitoring
of seed grain and harvested grain crops to detect the presence of
PA-containing plants and seeds.
(d) If the crop is found to be contaminated, immediate steps should be
taken to remove the toxic plants from the fields and to rid the
domestic and commercial cereal grain stores of the toxic seeds by
sieving or winnowing and destroying the toxic seeds/plants.
(e) The suspected contaminant and its toxic PAs should be identified
in specialized laboratories.
(f) Public awareness of the risks arising from the consumption of
grain contaminated by seeds, and the use of herbal preparations
containing PAs as food or medicines, should be created through the
media.
(g) The import and sale of seeds, herbs, and herbal preparations
containing PAs should be restricted.
(h) In the event of an outbreak, epidemiological investigations of the
affected population should be carried out, and plans made for long-
term follow-up.
(i) Cases of PA poisoning should be recorded and surveillance
organized by Poison Control Centres or other appropriate agencies of
the government.
4.5 First Aid
Immediate first aid measures are rarely possible for an acute exposure
but may be required in the management of emergencies, such as vomiting
of blood from bleeding blood vessels in the oesophagus.
No specific antidote is known for PA-induced toxic effects. The only
immediate valuable measure is to:
- identify the source of the PA and
- STOP any continued exposure.
4.6 Medical Management
No specific antidote or therapy for PA toxicity is known. Treatment is
supportive.
5. CURRENT REGULATIONS, GUIDELINES AND STANDARDS
COUNTRY RESTRICTED ITEM NATURE OF RESTRICTION
Australia Symphytum (comfrey) Scheduled as a poison,
(Any part of the dried effectively preventing sale
plant and its extracts or for medicinal purposes
preparations for human
internal use)
Austria Senecio numorensis Medicinal preparation must
Symphytum officinale be registered with the
Petasites officinalis Federal Ministry of Health
Tussilago farfara and Environmental Protection
Pulmonaria officinalis and sold only in pharmacies
(used as medicinal
preparations)
Canada Senecio jacobaea (ragwort) Proposed for inclusion in
Symphytum asperum Lepech the list of adulterants, thus
(prickly comfrey) prohibiting sale of such
Symphytum officinale L. compounds in or as food
root (common comfrey) (Section B.01.046 of the
Symphytum x uplandicum Canadian Food and Drug
Nym. (Russian comfrey) Regulations)
Echimidine or any of its Proposed for inclusion in
salts or any of the following the list of adulterants, thus
plant species or extracts prohibiting sale of these
or tinctures thereof: substances in or as drugs
(Section C.01.038 of the
(i) Symphytum asperum Canadian Food and Drug
(ii) Symphytum x uplandicum Regulations)
(iii) Any other plant species
containing echimidine
Indonesia Symphytum officinale L. Dispensaries and drug stores
or its parts either fresh not allowed to prepare, sell,
or dried or advertise it
USA Crotalaria spectabilis Compliance policy guidelines
Crotalaria sagittalis require that the presence of an
Crotalaria striata average of at least one whole
seed per pound of product
represents the criterion for
direct reference seizure to
the office of regulatory
affairs (ORA)HFC-25 and
for direct citation by the
district offices (OB/01/82)
COUNTRY RESTRICTED ITEM NATURE OF RESTRICTION
Lasiocarpine Any solid waste (except
domestic) containing
lasiocarpine must be listed
as hazardous waste (subject
to regulation and
notification requirements);
where lasiocarpine is a
principal organic hazardous
constituent in the EPA
permit, incineration must
achieve a destruction and
removal efficiency of 99.99%
USSR Stored grain: Contamination limit
Heliotropium lasiocarpum
-Seed 0.1%
Trichodesma incanum
-Seed 0%
-Seed grain Contamination with both
species is prohibited
(Note: It has been calculated that
0.1% tolerance for H. lasiocarpum
seeds in stored grain could result in
1.82 mg of PA per kg of stored wheat)
Note: Besides the above, a number of countries have regulatory proposals under
development and several others enforce administrative or compliance guidelines
based on general prohibitory provisions for the basic food and drug products;
studies are being carried out to further evaluate the risk.
1.2 Physical and Chemical Properties
The pure alkaloids are mostly crystalline solids; some are gums or
amorphous solids. Some are only slightly soluble in water, but all
dissolve when neutralized with acid. They occur in the plants partly
as N-oxides, which are water soluble. The alkaloids are fairly
stable, but are subject to hydrolysis in alkaline solution and to
enzymatic decomposition. The latter occurs in some plant species
during wilting and drying. The stability of the alkaloids when the
plants are cooked is not known.
1.3 Uses
One alkaloid, monocrotaline, is marketed commercially as a fine
chemical for research purposes. Another, indicine N-oxide, is being
tested as an antitumour drug in human beings.
2. SUMMARY AND CONCLUSIONS
2.1 Human Exposure to Pyrrolizidine Alkaloids
The contamination of cereal grains with seeds of PA-containing plants
has caused epidemics of human poisoning in four countries. The plants
involved were species of Heliotropium, Trichodesma, Crotalaria, and
Senecio. Some of these plants thrive under arid conditions and their
growth may be favoured by drought. Another important form of exposure
is the use of PA-containing herbs as medicines, food, or beverages;
this has resulted in occasional cases of human poisoning. The most
important of such PA-containing plants are species of Heliotropium,
Crotalaria, Senecio, and Symphytum. Symphytum species (comfrey)
are also available in the form of ointments or as digestive aids in
the form of capsules.
Low-level exposure may occur in some countries through the presence of
PAs in foods, such as honey and milk, but no reports of human
poisoning caused through these media are available.
2.2 Metabolism and Excretion
PAs are readily absorbed from the digestive tract and cause harmful
effects only after undergoing activation in the liver to toxic
metabolites. Effects include a variety of changes leading to permanent
damage to genes and chromosomes, the ability of the cells to divide,
or to the development of cancer, and even cell death. The alkaloids
are quickly converted to harmless compounds and are largely cleared
from the body within a few hours of absorption, so that no traces of
the ingested PAs or their breakdown products are detectable in the
body tissues and fluids. There are no indications that the alkaloids
themselves accumulate in animal tissues, but their effects are
cumulative, even at low rates of intake. The proportion excreted in
urine varies according to the water solubility of the alkaloid.
Estimates of the total intake over a long period can only be
approximate.
2.3 Effects on Experimental Animals
The acute toxicity of PAs varies widely. The rat LD50s of most
alkaloids known to be significant for human health are in the range of
34-300 mg/kg, though some approach 1000 mg/kg. When ingested, the
toxicity of the N-oxide of an alkaloid is similar to that of the
parent alkaloid.
PAs are toxic for a wide variety of domestic, laboratory, and farm
animals, pigs and poultry being the most sensitive of farm animals
followed by horses and cattle, and sheep and goats, which are the most
resistant; sheep and goats are affected only after extended periods of
exposure.
The toxic effects of most PAs occur primarily and mainly in the liver.
Some PAs are particularly prone to cause damage in the lungs,
principally to the blood vessels, resulting in a rise in blood
pressure in the lungs, which leads to secondary effects on the
functioning of the right side of the heart. The kidneys and some other
organs are less commonly affected. The brain is the principal target
organ of one or two PAs.
The effects of PAs in animals may be acute or chronic, depending on
the dose level and the period of survival after exposure. In acute
poisoning, death occurs within about 7 days, due to severe liver
damage. Chronic liver damage may follow administration of a single
sublethal dose or of repeated low doses.
PAs produce aberrations in the chromosomes in plant and mammalian
cells and induce changes in genes that are perpetuated in subsequent
cell divisions in mutagenesis test systems. Several PAs have been
shown to be capable of producing cancer, chiefly in the liver in rats.
2.4 Effects on Human Health
Although all age groups are affected, children are particularly
vulnerable to the effects of PAs. The symptoms, which are generally
acute in onset, are characterized by upper abdominal discomfort that
develops rapidly and progresses to swelling of the abdomen resulting
in increased girth, sometimes accompanied by a reduction in the
quantity of urine excreted and swelling of the feet. The disease is
called veno-occlusive disease (VOD) because of the characteristic
obstruction of the small venous blood channels that carry blood from
the liver back to the heart. The disease often progresses rapidly and
mortality is high. There may be vomiting of blood in advanced stages
of the disease. While many patients recover, the disease may continue
for a long time in others resulting in a severely scarred liver - a
condition called cirrhosis. Some patients may have only vague symptoms
and the only sign of the disease may be persistent enlargement of the
liver.
The liver is usually the target organ but, in an epidemic caused by
contamination of the staple cereal with the seeds of Trichodesma,
the brain and the nervous system were mainly affected.
Chromosome aberrations have been reported in the blood cells of
children affected by VOD but, as yet, there is no evidence pointing to
an increased incidence of cancer of the liver or other organs or
congenital anomalies in the newborn offspring of patients exposed to
PAs.
2.5 Effects on the Environment
Plants containing PAs are likely to grow as weeds among staple food
crops and pastures, especially following drought, and consumption of
such crops can cause large scale outbreaks of toxic disease in both
man and farm animals. Little is known about the effects on wildlife
but, in one report, the death of deer was ascribed to their grazing on
toxic plants.
PAs are believed to be biodegradable so that water supplies are not
suspect.
3. RISK EVALUATION
No-observed-adverse-effect levels have not been established in
experimental animal studies with PAs. Estimates of intakes causing
toxic effects in human beings indicate that they are more sensitive
than rats and domestic animals. Rats dosed with lasiocarpine at a rate
equivalent to 0.2 mg/kg body weight per day (equivalent to about
2 mg/kg in the diet) developed tumours. Pigs fed monocrotaline at
1.8 mg/kg of feed (equivalent to about 0.08 mg/kg body weight per day)
developed chronic liver damage in several months. The lowest intake
rate causing VOD in a human being was estimated to be 0.015 mg/kg body
weight per day, and was the result of self-medication with a comfrey
preparation. In view of the established ability of some PAs to produce
cancer in rats, plant products containing them should not be eaten or
drunk.
4. PREVENTIVE MEASURES AND MEDICAL ADVICE
4.1 Main Hazards for Man and the Environment
Consumption of contaminated grain or the use of PA-containing plants
as herbal medicines, beverages, or food by man, or grazing on
contaminated pastures by animals, may cause acute or chronic disease.
The principal target organ in man and most animals is the liver,
resulting in collection of fluid in the abdomen and swelling of feet,
etc. In experimental animals, administration of PAs has been known to
produce cancer. Though there is no proof yet of cancer developing in
man, such a possibility cannot be entirely ruled out.
4.2 Prevention of PA Poisoning
Prevention of exposure is the only effective method of limiting
toxicity due to PAs. Even low doses over a period of time may present
a health risk and exposure should be avoided or minimized as far as
possible. Measures are required at several action points.
4.3 Advice to Physicians
Occurrence of symptoms of rapidly increasing swelling of the abdomen
accompanied by dragging discomfort in the right upper abdomen,
particularly if affecting more than one member of the family or
several members of a community over a limited period and geographical
area, should arouse suspicion. The diet of such persons should be
examined for possible contamination with the seeds of toxic plants and
the affected persons should be questioned regarding the possible
intake of herbal preparations. If any such history is positive, the
suspected toxic seeds/herbs should be analysed for the presence of PAs
using a simple field test, which can be carried out in a not too
highly specialized chemical laboratory.
Examination of the liver by needle biopsy in patients or at post-
mortem examination is most likely to provide the crucial clue.
Haemorrhagic centrilobular necrosis and occlusion of the hepatic vein
radicles in the liver are the hallmarks of the disease, if found in
patients with the symptoms described above. Needle biopsy of the liver
should be performed only in a properly equipped hospital with
facilities for blood transfusion.
4.4 Precautionary Measures to Protect the Environment and to Prevent
Disease
(a) Toxic PA-containing flora growing in the region, particularly
those that may contaminate cereal grain crops, and those that are used
locally as herbal foods, beverages, or medicines, whether grown
locally or imported, should be identified.
(b) Appropriate agrotechnical practices should be followed for the
prevention/control of the growth of PA-containing plants among cereal
food crops and pastures.
(c) Appropriate systems should be developed for the routine monitoring
of seed grain and harvested grain crops to detect the presence of
PA-containing plants and seeds.
(d) If the crop is found to be contaminated, immediate steps should be
taken to remove the toxic plants from the fields and to rid the
domestic and commercial cereal grain stores of the toxic seeds by
sieving or winnowing and destroying the toxic seeds/plants.
(e) The suspected contaminant and its toxic PAs should be identified
in specialized laboratories.
(f) Public awareness of the risks arising from the consumption of
grain contaminated by seeds, and the use of herbal preparations
containing PAs as food or medicines, should be created through the
media.
(g) The import and sale of seeds, herbs, and herbal preparations
containing PAs should be restricted.
(h) In the event of an outbreak, epidemiological investigations of the
affected population should be carried out, and plans made for long-
term follow-up.
(i) Cases of PA poisoning should be recorded and surveillance
organized by Poison Control Centres or other appropriate agencies of
the government.
4.5 First Aid
Immediate first aid measures are rarely possible for an acute exposure
but may be required in the management of emergencies, such as vomiting
of blood from bleeding blood vessels in the oesophagus.
No specific antidote is known for PA-induced toxic effects. The only
immediate valuable measure is to:
- identify the source of the PA and
- STOP any continued exposure.
4.6 Medical Management
No specific antidote or therapy for PA toxicity is known. Treatment is
supportive.
5. CURRENT REGULATIONS, GUIDELINES AND STANDARDS
COUNTRY RESTRICTED ITEM NATURE OF RESTRICTION
Australia Symphytum (comfrey) Scheduled as a poison,
(Any part of the dried effectively preventing sale
plant and its extracts or for medicinal purposes
preparations for human
internal use)
Austria Senecio numorensis Medicinal preparation must
Symphytum officinale be registered with the
Petasites officinalis Federal Ministry of Health
Tussilago farfara and Environmental Protection
Pulmonaria officinalis and sold only in pharmacies
(used as medicinal
preparations)
Canada Senecio jacobaea (ragwort) Proposed for inclusion in
Symphytum asperum Lepech the list of adulterants, thus
(prickly comfrey) prohibiting sale of such
Symphytum officinale L. compounds in or as food
root (common comfrey) (Section B.01.046 of the
Symphytum x uplandicum Canadian Food and Drug
Nym. (Russian comfrey) Regulations)
Echimidine or any of its Proposed for inclusion in
salts or any of the following the list of adulterants, thus
plant species or extracts prohibiting sale of these
or tinctures thereof: substances in or as drugs
(Section C.01.038 of the
(i) Symphytum asperum Canadian Food and Drug
(ii) Symphytum x uplandicum Regulations)
(iii) Any other plant species
containing echimidine
Indonesia Symphytum officinale L. Dispensaries and drug stores
or its parts either fresh not allowed to prepare, sell,
or dried or advertise it
USA Crotalaria spectabilis Compliance policy guidelines
Crotalaria sagittalis require that the presence of an
Crotalaria striata average of at least one whole
seed per pound of product
represents the criterion for
direct reference seizure to
the office of regulatory
affairs (ORA)HFC-25 and
for direct citation by the
district offices (OB/01/82)
COUNTRY RESTRICTED ITEM NATURE OF RESTRICTION
Lasiocarpine Any solid waste (except
domestic) containing
lasiocarpine must be listed
as hazardous waste (subject
to regulation and
notification requirements);
where lasiocarpine is a
principal organic hazardous
constituent in the EPA
permit, incineration must
achieve a destruction and
removal efficiency of 99.99%
USSR Stored grain: Contamination limit
Heliotropium lasiocarpum
-Seed 0.1%
Trichodesma incanum
-Seed 0%
-Seed grain Contamination with both
species is prohibited
(Note: It has been calculated that
0.1% tolerance for H. lasiocarpum
seeds in stored grain could result in
1.82 mg of PA per kg of stored wheat)
Note: Besides the above, a number of countries have regulatory proposals under
development and several others enforce administrative or compliance guidelines
based on general prohibitory provisions for the basic food and drug products;
studies are being carried out to further evaluate the risk.
 1.2 Physical and Chemical Properties
The pure alkaloids are mostly crystalline solids; some are gums or
amorphous solids. Some are only slightly soluble in water, but all
dissolve when neutralized with acid. They occur in the plants partly
as N-oxides, which are water soluble. The alkaloids are fairly
stable, but are subject to hydrolysis in alkaline solution and to
enzymatic decomposition. The latter occurs in some plant species
during wilting and drying. The stability of the alkaloids when the
plants are cooked is not known.
1.3 Uses
One alkaloid, monocrotaline, is marketed commercially as a fine
chemical for research purposes. Another, indicine N-oxide, is being
tested as an antitumour drug in human beings.
2. SUMMARY AND CONCLUSIONS
2.1 Human Exposure to Pyrrolizidine Alkaloids
The contamination of cereal grains with seeds of PA-containing plants
has caused epidemics of human poisoning in four countries. The plants
involved were species of Heliotropium, Trichodesma, Crotalaria, and
Senecio. Some of these plants thrive under arid conditions and their
growth may be favoured by drought. Another important form of exposure
is the use of PA-containing herbs as medicines, food, or beverages;
this has resulted in occasional cases of human poisoning. The most
important of such PA-containing plants are species of Heliotropium,
Crotalaria, Senecio, and Symphytum. Symphytum species (comfrey)
are also available in the form of ointments or as digestive aids in
the form of capsules.
Low-level exposure may occur in some countries through the presence of
PAs in foods, such as honey and milk, but no reports of human
poisoning caused through these media are available.
2.2 Metabolism and Excretion
PAs are readily absorbed from the digestive tract and cause harmful
effects only after undergoing activation in the liver to toxic
metabolites. Effects include a variety of changes leading to permanent
damage to genes and chromosomes, the ability of the cells to divide,
or to the development of cancer, and even cell death. The alkaloids
are quickly converted to harmless compounds and are largely cleared
from the body within a few hours of absorption, so that no traces of
the ingested PAs or their breakdown products are detectable in the
body tissues and fluids. There are no indications that the alkaloids
themselves accumulate in animal tissues, but their effects are
cumulative, even at low rates of intake. The proportion excreted in
urine varies according to the water solubility of the alkaloid.
Estimates of the total intake over a long period can only be
approximate.
2.3 Effects on Experimental Animals
The acute toxicity of PAs varies widely. The rat LD50s of most
alkaloids known to be significant for human health are in the range of
34-300 mg/kg, though some approach 1000 mg/kg. When ingested, the
toxicity of the N-oxide of an alkaloid is similar to that of the
parent alkaloid.
PAs are toxic for a wide variety of domestic, laboratory, and farm
animals, pigs and poultry being the most sensitive of farm animals
followed by horses and cattle, and sheep and goats, which are the most
resistant; sheep and goats are affected only after extended periods of
exposure.
The toxic effects of most PAs occur primarily and mainly in the liver.
Some PAs are particularly prone to cause damage in the lungs,
principally to the blood vessels, resulting in a rise in blood
pressure in the lungs, which leads to secondary effects on the
functioning of the right side of the heart. The kidneys and some other
organs are less commonly affected. The brain is the principal target
organ of one or two PAs.
The effects of PAs in animals may be acute or chronic, depending on
the dose level and the period of survival after exposure. In acute
poisoning, death occurs within about 7 days, due to severe liver
damage. Chronic liver damage may follow administration of a single
sublethal dose or of repeated low doses.
PAs produce aberrations in the chromosomes in plant and mammalian
cells and induce changes in genes that are perpetuated in subsequent
cell divisions in mutagenesis test systems. Several PAs have been
shown to be capable of producing cancer, chiefly in the liver in rats.
2.4 Effects on Human Health
Although all age groups are affected, children are particularly
vulnerable to the effects of PAs. The symptoms, which are generally
acute in onset, are characterized by upper abdominal discomfort that
develops rapidly and progresses to swelling of the abdomen resulting
in increased girth, sometimes accompanied by a reduction in the
quantity of urine excreted and swelling of the feet. The disease is
called veno-occlusive disease (VOD) because of the characteristic
obstruction of the small venous blood channels that carry blood from
the liver back to the heart. The disease often progresses rapidly and
mortality is high. There may be vomiting of blood in advanced stages
of the disease. While many patients recover, the disease may continue
for a long time in others resulting in a severely scarred liver - a
condition called cirrhosis. Some patients may have only vague symptoms
and the only sign of the disease may be persistent enlargement of the
liver.
The liver is usually the target organ but, in an epidemic caused by
contamination of the staple cereal with the seeds of Trichodesma,
the brain and the nervous system were mainly affected.
Chromosome aberrations have been reported in the blood cells of
children affected by VOD but, as yet, there is no evidence pointing to
an increased incidence of cancer of the liver or other organs or
congenital anomalies in the newborn offspring of patients exposed to
PAs.
2.5 Effects on the Environment
Plants containing PAs are likely to grow as weeds among staple food
crops and pastures, especially following drought, and consumption of
such crops can cause large scale outbreaks of toxic disease in both
man and farm animals. Little is known about the effects on wildlife
but, in one report, the death of deer was ascribed to their grazing on
toxic plants.
PAs are believed to be biodegradable so that water supplies are not
suspect.
3. RISK EVALUATION
No-observed-adverse-effect levels have not been established in
experimental animal studies with PAs. Estimates of intakes causing
toxic effects in human beings indicate that they are more sensitive
than rats and domestic animals. Rats dosed with lasiocarpine at a rate
equivalent to 0.2 mg/kg body weight per day (equivalent to about
2 mg/kg in the diet) developed tumours. Pigs fed monocrotaline at
1.8 mg/kg of feed (equivalent to about 0.08 mg/kg body weight per day)
developed chronic liver damage in several months. The lowest intake
rate causing VOD in a human being was estimated to be 0.015 mg/kg body
weight per day, and was the result of self-medication with a comfrey
preparation. In view of the established ability of some PAs to produce
cancer in rats, plant products containing them should not be eaten or
drunk.
4. PREVENTIVE MEASURES AND MEDICAL ADVICE
4.1 Main Hazards for Man and the Environment
Consumption of contaminated grain or the use of PA-containing plants
as herbal medicines, beverages, or food by man, or grazing on
contaminated pastures by animals, may cause acute or chronic disease.
The principal target organ in man and most animals is the liver,
resulting in collection of fluid in the abdomen and swelling of feet,
etc. In experimental animals, administration of PAs has been known to
produce cancer. Though there is no proof yet of cancer developing in
man, such a possibility cannot be entirely ruled out.
4.2 Prevention of PA Poisoning
Prevention of exposure is the only effective method of limiting
toxicity due to PAs. Even low doses over a period of time may present
a health risk and exposure should be avoided or minimized as far as
possible. Measures are required at several action points.
4.3 Advice to Physicians
Occurrence of symptoms of rapidly increasing swelling of the abdomen
accompanied by dragging discomfort in the right upper abdomen,
particularly if affecting more than one member of the family or
several members of a community over a limited period and geographical
area, should arouse suspicion. The diet of such persons should be
examined for possible contamination with the seeds of toxic plants and
the affected persons should be questioned regarding the possible
intake of herbal preparations. If any such history is positive, the
suspected toxic seeds/herbs should be analysed for the presence of PAs
using a simple field test, which can be carried out in a not too
highly specialized chemical laboratory.
Examination of the liver by needle biopsy in patients or at post-
mortem examination is most likely to provide the crucial clue.
Haemorrhagic centrilobular necrosis and occlusion of the hepatic vein
radicles in the liver are the hallmarks of the disease, if found in
patients with the symptoms described above. Needle biopsy of the liver
should be performed only in a properly equipped hospital with
facilities for blood transfusion.
4.4 Precautionary Measures to Protect the Environment and to Prevent
Disease
(a) Toxic PA-containing flora growing in the region, particularly
those that may contaminate cereal grain crops, and those that are used
locally as herbal foods, beverages, or medicines, whether grown
locally or imported, should be identified.
(b) Appropriate agrotechnical practices should be followed for the
prevention/control of the growth of PA-containing plants among cereal
food crops and pastures.
(c) Appropriate systems should be developed for the routine monitoring
of seed grain and harvested grain crops to detect the presence of
PA-containing plants and seeds.
(d) If the crop is found to be contaminated, immediate steps should be
taken to remove the toxic plants from the fields and to rid the
domestic and commercial cereal grain stores of the toxic seeds by
sieving or winnowing and destroying the toxic seeds/plants.
(e) The suspected contaminant and its toxic PAs should be identified
in specialized laboratories.
(f) Public awareness of the risks arising from the consumption of
grain contaminated by seeds, and the use of herbal preparations
containing PAs as food or medicines, should be created through the
media.
(g) The import and sale of seeds, herbs, and herbal preparations
containing PAs should be restricted.
(h) In the event of an outbreak, epidemiological investigations of the
affected population should be carried out, and plans made for long-
term follow-up.
(i) Cases of PA poisoning should be recorded and surveillance
organized by Poison Control Centres or other appropriate agencies of
the government.
4.5 First Aid
Immediate first aid measures are rarely possible for an acute exposure
but may be required in the management of emergencies, such as vomiting
of blood from bleeding blood vessels in the oesophagus.
No specific antidote is known for PA-induced toxic effects. The only
immediate valuable measure is to:
- identify the source of the PA and
- STOP any continued exposure.
4.6 Medical Management
No specific antidote or therapy for PA toxicity is known. Treatment is
supportive.
5. CURRENT REGULATIONS, GUIDELINES AND STANDARDS
COUNTRY RESTRICTED ITEM NATURE OF RESTRICTION
Australia Symphytum (comfrey) Scheduled as a poison,
(Any part of the dried effectively preventing sale
plant and its extracts or for medicinal purposes
preparations for human
internal use)
Austria Senecio numorensis Medicinal preparation must
Symphytum officinale be registered with the
Petasites officinalis Federal Ministry of Health
Tussilago farfara and Environmental Protection
Pulmonaria officinalis and sold only in pharmacies
(used as medicinal
preparations)
Canada Senecio jacobaea (ragwort) Proposed for inclusion in
Symphytum asperum Lepech the list of adulterants, thus
(prickly comfrey) prohibiting sale of such
Symphytum officinale L. compounds in or as food
root (common comfrey) (Section B.01.046 of the
Symphytum x uplandicum Canadian Food and Drug
Nym. (Russian comfrey) Regulations)
Echimidine or any of its Proposed for inclusion in
salts or any of the following the list of adulterants, thus
plant species or extracts prohibiting sale of these
or tinctures thereof: substances in or as drugs
(Section C.01.038 of the
(i) Symphytum asperum Canadian Food and Drug
(ii) Symphytum x uplandicum Regulations)
(iii) Any other plant species
containing echimidine
Indonesia Symphytum officinale L. Dispensaries and drug stores
or its parts either fresh not allowed to prepare, sell,
or dried or advertise it
USA Crotalaria spectabilis Compliance policy guidelines
Crotalaria sagittalis require that the presence of an
Crotalaria striata average of at least one whole
seed per pound of product
represents the criterion for
direct reference seizure to
the office of regulatory
affairs (ORA)HFC-25 and
for direct citation by the
district offices (OB/01/82)
COUNTRY RESTRICTED ITEM NATURE OF RESTRICTION
Lasiocarpine Any solid waste (except
domestic) containing
lasiocarpine must be listed
as hazardous waste (subject
to regulation and
notification requirements);
where lasiocarpine is a
principal organic hazardous
constituent in the EPA
permit, incineration must
achieve a destruction and
removal efficiency of 99.99%
USSR Stored grain: Contamination limit
Heliotropium lasiocarpum
-Seed 0.1%
Trichodesma incanum
-Seed 0%
-Seed grain Contamination with both
species is prohibited
(Note: It has been calculated that
0.1% tolerance for H. lasiocarpum
seeds in stored grain could result in
1.82 mg of PA per kg of stored wheat)
Note: Besides the above, a number of countries have regulatory proposals under
development and several others enforce administrative or compliance guidelines
based on general prohibitory provisions for the basic food and drug products;
studies are being carried out to further evaluate the risk.
1.2 Physical and Chemical Properties
The pure alkaloids are mostly crystalline solids; some are gums or
amorphous solids. Some are only slightly soluble in water, but all
dissolve when neutralized with acid. They occur in the plants partly
as N-oxides, which are water soluble. The alkaloids are fairly
stable, but are subject to hydrolysis in alkaline solution and to
enzymatic decomposition. The latter occurs in some plant species
during wilting and drying. The stability of the alkaloids when the
plants are cooked is not known.
1.3 Uses
One alkaloid, monocrotaline, is marketed commercially as a fine
chemical for research purposes. Another, indicine N-oxide, is being
tested as an antitumour drug in human beings.
2. SUMMARY AND CONCLUSIONS
2.1 Human Exposure to Pyrrolizidine Alkaloids
The contamination of cereal grains with seeds of PA-containing plants
has caused epidemics of human poisoning in four countries. The plants
involved were species of Heliotropium, Trichodesma, Crotalaria, and
Senecio. Some of these plants thrive under arid conditions and their
growth may be favoured by drought. Another important form of exposure
is the use of PA-containing herbs as medicines, food, or beverages;
this has resulted in occasional cases of human poisoning. The most
important of such PA-containing plants are species of Heliotropium,
Crotalaria, Senecio, and Symphytum. Symphytum species (comfrey)
are also available in the form of ointments or as digestive aids in
the form of capsules.
Low-level exposure may occur in some countries through the presence of
PAs in foods, such as honey and milk, but no reports of human
poisoning caused through these media are available.
2.2 Metabolism and Excretion
PAs are readily absorbed from the digestive tract and cause harmful
effects only after undergoing activation in the liver to toxic
metabolites. Effects include a variety of changes leading to permanent
damage to genes and chromosomes, the ability of the cells to divide,
or to the development of cancer, and even cell death. The alkaloids
are quickly converted to harmless compounds and are largely cleared
from the body within a few hours of absorption, so that no traces of
the ingested PAs or their breakdown products are detectable in the
body tissues and fluids. There are no indications that the alkaloids
themselves accumulate in animal tissues, but their effects are
cumulative, even at low rates of intake. The proportion excreted in
urine varies according to the water solubility of the alkaloid.
Estimates of the total intake over a long period can only be
approximate.
2.3 Effects on Experimental Animals
The acute toxicity of PAs varies widely. The rat LD50s of most
alkaloids known to be significant for human health are in the range of
34-300 mg/kg, though some approach 1000 mg/kg. When ingested, the
toxicity of the N-oxide of an alkaloid is similar to that of the
parent alkaloid.
PAs are toxic for a wide variety of domestic, laboratory, and farm
animals, pigs and poultry being the most sensitive of farm animals
followed by horses and cattle, and sheep and goats, which are the most
resistant; sheep and goats are affected only after extended periods of
exposure.
The toxic effects of most PAs occur primarily and mainly in the liver.
Some PAs are particularly prone to cause damage in the lungs,
principally to the blood vessels, resulting in a rise in blood
pressure in the lungs, which leads to secondary effects on the
functioning of the right side of the heart. The kidneys and some other
organs are less commonly affected. The brain is the principal target
organ of one or two PAs.
The effects of PAs in animals may be acute or chronic, depending on
the dose level and the period of survival after exposure. In acute
poisoning, death occurs within about 7 days, due to severe liver
damage. Chronic liver damage may follow administration of a single
sublethal dose or of repeated low doses.
PAs produce aberrations in the chromosomes in plant and mammalian
cells and induce changes in genes that are perpetuated in subsequent
cell divisions in mutagenesis test systems. Several PAs have been
shown to be capable of producing cancer, chiefly in the liver in rats.
2.4 Effects on Human Health
Although all age groups are affected, children are particularly
vulnerable to the effects of PAs. The symptoms, which are generally
acute in onset, are characterized by upper abdominal discomfort that
develops rapidly and progresses to swelling of the abdomen resulting
in increased girth, sometimes accompanied by a reduction in the
quantity of urine excreted and swelling of the feet. The disease is
called veno-occlusive disease (VOD) because of the characteristic
obstruction of the small venous blood channels that carry blood from
the liver back to the heart. The disease often progresses rapidly and
mortality is high. There may be vomiting of blood in advanced stages
of the disease. While many patients recover, the disease may continue
for a long time in others resulting in a severely scarred liver - a
condition called cirrhosis. Some patients may have only vague symptoms
and the only sign of the disease may be persistent enlargement of the
liver.
The liver is usually the target organ but, in an epidemic caused by
contamination of the staple cereal with the seeds of Trichodesma,
the brain and the nervous system were mainly affected.
Chromosome aberrations have been reported in the blood cells of
children affected by VOD but, as yet, there is no evidence pointing to
an increased incidence of cancer of the liver or other organs or
congenital anomalies in the newborn offspring of patients exposed to
PAs.
2.5 Effects on the Environment
Plants containing PAs are likely to grow as weeds among staple food
crops and pastures, especially following drought, and consumption of
such crops can cause large scale outbreaks of toxic disease in both
man and farm animals. Little is known about the effects on wildlife
but, in one report, the death of deer was ascribed to their grazing on
toxic plants.
PAs are believed to be biodegradable so that water supplies are not
suspect.
3. RISK EVALUATION
No-observed-adverse-effect levels have not been established in
experimental animal studies with PAs. Estimates of intakes causing
toxic effects in human beings indicate that they are more sensitive
than rats and domestic animals. Rats dosed with lasiocarpine at a rate
equivalent to 0.2 mg/kg body weight per day (equivalent to about
2 mg/kg in the diet) developed tumours. Pigs fed monocrotaline at
1.8 mg/kg of feed (equivalent to about 0.08 mg/kg body weight per day)
developed chronic liver damage in several months. The lowest intake
rate causing VOD in a human being was estimated to be 0.015 mg/kg body
weight per day, and was the result of self-medication with a comfrey
preparation. In view of the established ability of some PAs to produce
cancer in rats, plant products containing them should not be eaten or
drunk.
4. PREVENTIVE MEASURES AND MEDICAL ADVICE
4.1 Main Hazards for Man and the Environment
Consumption of contaminated grain or the use of PA-containing plants
as herbal medicines, beverages, or food by man, or grazing on
contaminated pastures by animals, may cause acute or chronic disease.
The principal target organ in man and most animals is the liver,
resulting in collection of fluid in the abdomen and swelling of feet,
etc. In experimental animals, administration of PAs has been known to
produce cancer. Though there is no proof yet of cancer developing in
man, such a possibility cannot be entirely ruled out.
4.2 Prevention of PA Poisoning
Prevention of exposure is the only effective method of limiting
toxicity due to PAs. Even low doses over a period of time may present
a health risk and exposure should be avoided or minimized as far as
possible. Measures are required at several action points.
4.3 Advice to Physicians
Occurrence of symptoms of rapidly increasing swelling of the abdomen
accompanied by dragging discomfort in the right upper abdomen,
particularly if affecting more than one member of the family or
several members of a community over a limited period and geographical
area, should arouse suspicion. The diet of such persons should be
examined for possible contamination with the seeds of toxic plants and
the affected persons should be questioned regarding the possible
intake of herbal preparations. If any such history is positive, the
suspected toxic seeds/herbs should be analysed for the presence of PAs
using a simple field test, which can be carried out in a not too
highly specialized chemical laboratory.
Examination of the liver by needle biopsy in patients or at post-
mortem examination is most likely to provide the crucial clue.
Haemorrhagic centrilobular necrosis and occlusion of the hepatic vein
radicles in the liver are the hallmarks of the disease, if found in
patients with the symptoms described above. Needle biopsy of the liver
should be performed only in a properly equipped hospital with
facilities for blood transfusion.
4.4 Precautionary Measures to Protect the Environment and to Prevent
Disease
(a) Toxic PA-containing flora growing in the region, particularly
those that may contaminate cereal grain crops, and those that are used
locally as herbal foods, beverages, or medicines, whether grown
locally or imported, should be identified.
(b) Appropriate agrotechnical practices should be followed for the
prevention/control of the growth of PA-containing plants among cereal
food crops and pastures.
(c) Appropriate systems should be developed for the routine monitoring
of seed grain and harvested grain crops to detect the presence of
PA-containing plants and seeds.
(d) If the crop is found to be contaminated, immediate steps should be
taken to remove the toxic plants from the fields and to rid the
domestic and commercial cereal grain stores of the toxic seeds by
sieving or winnowing and destroying the toxic seeds/plants.
(e) The suspected contaminant and its toxic PAs should be identified
in specialized laboratories.
(f) Public awareness of the risks arising from the consumption of
grain contaminated by seeds, and the use of herbal preparations
containing PAs as food or medicines, should be created through the
media.
(g) The import and sale of seeds, herbs, and herbal preparations
containing PAs should be restricted.
(h) In the event of an outbreak, epidemiological investigations of the
affected population should be carried out, and plans made for long-
term follow-up.
(i) Cases of PA poisoning should be recorded and surveillance
organized by Poison Control Centres or other appropriate agencies of
the government.
4.5 First Aid
Immediate first aid measures are rarely possible for an acute exposure
but may be required in the management of emergencies, such as vomiting
of blood from bleeding blood vessels in the oesophagus.
No specific antidote is known for PA-induced toxic effects. The only
immediate valuable measure is to:
- identify the source of the PA and
- STOP any continued exposure.
4.6 Medical Management
No specific antidote or therapy for PA toxicity is known. Treatment is
supportive.
5. CURRENT REGULATIONS, GUIDELINES AND STANDARDS
COUNTRY RESTRICTED ITEM NATURE OF RESTRICTION
Australia Symphytum (comfrey) Scheduled as a poison,
(Any part of the dried effectively preventing sale
plant and its extracts or for medicinal purposes
preparations for human
internal use)
Austria Senecio numorensis Medicinal preparation must
Symphytum officinale be registered with the
Petasites officinalis Federal Ministry of Health
Tussilago farfara and Environmental Protection
Pulmonaria officinalis and sold only in pharmacies
(used as medicinal
preparations)
Canada Senecio jacobaea (ragwort) Proposed for inclusion in
Symphytum asperum Lepech the list of adulterants, thus
(prickly comfrey) prohibiting sale of such
Symphytum officinale L. compounds in or as food
root (common comfrey) (Section B.01.046 of the
Symphytum x uplandicum Canadian Food and Drug
Nym. (Russian comfrey) Regulations)
Echimidine or any of its Proposed for inclusion in
salts or any of the following the list of adulterants, thus
plant species or extracts prohibiting sale of these
or tinctures thereof: substances in or as drugs
(Section C.01.038 of the
(i) Symphytum asperum Canadian Food and Drug
(ii) Symphytum x uplandicum Regulations)
(iii) Any other plant species
containing echimidine
Indonesia Symphytum officinale L. Dispensaries and drug stores
or its parts either fresh not allowed to prepare, sell,
or dried or advertise it
USA Crotalaria spectabilis Compliance policy guidelines
Crotalaria sagittalis require that the presence of an
Crotalaria striata average of at least one whole
seed per pound of product
represents the criterion for
direct reference seizure to
the office of regulatory
affairs (ORA)HFC-25 and
for direct citation by the
district offices (OB/01/82)
COUNTRY RESTRICTED ITEM NATURE OF RESTRICTION
Lasiocarpine Any solid waste (except
domestic) containing
lasiocarpine must be listed
as hazardous waste (subject
to regulation and
notification requirements);
where lasiocarpine is a
principal organic hazardous
constituent in the EPA
permit, incineration must
achieve a destruction and
removal efficiency of 99.99%
USSR Stored grain: Contamination limit
Heliotropium lasiocarpum
-Seed 0.1%
Trichodesma incanum
-Seed 0%
-Seed grain Contamination with both
species is prohibited
(Note: It has been calculated that
0.1% tolerance for H. lasiocarpum
seeds in stored grain could result in
1.82 mg of PA per kg of stored wheat)
Note: Besides the above, a number of countries have regulatory proposals under
development and several others enforce administrative or compliance guidelines
based on general prohibitory provisions for the basic food and drug products;
studies are being carried out to further evaluate the risk.
