
IPCS INTERNATIONAL PROGRAMME ON CHEMICAL SAFETY
Health and Safety Guide No. 46
BARIUM
HEALTH AND SAFETY GUIDE
UNITED NATIONS ENVIRONMENT PROGRAMME
INTERNATIONAL LABOUR ORGANISATION
WORLD HEALTH ORGANIZATION
WORLD HEALTH ORGANIZATION, GENEVA 1991
This is a companion volume to Environmental Health Criteria 107:
Barium
Published by the World Health Organization for the International
Programme on Chemical Safety (a collaborative programme of the United
Nations Environment Programme, the International Labour Organisation,
and the World Health Organization)
This report contains the collective views of an international group of
experts and does not necessarily represent the decisions or the stated
policy of the United Nations Environment Programme, the International
Labour Organisation, or the World Health Organization
WHO Library Cataloguing in Publication Data
Barium : health and safety guide.
(Health and safety guide ; no. 46)
1. Barium - standards I. Series
ISBN 92 4 151046 3 (NLM Classification: QV 618)
ISSN 0259-7268
(c) World Health Organization 1991
Publications of the World Health Organization enjoy copyright
protection in accordance with the provisions of Protocol 2 of the
Universal Copyright Convention. For rights of reproduction or
translation of WHO publications, in part or in toto, application
should be made to the Office of Publications, World Health
Organization, Geneva, Switzerland. The World Health Organization
welcomes such applications.
The designations employed and the presentation of the material in this
publication do not imply the expression of any opinion whatsoever on
the part of the Secretariat of the World Health Organization
concerning the legal status of any country, territory, city or area or
of its authorities, or concerning the delimitation of its frontiers or
boundaries.
The mention of specific companies or of certain manufacturers'
products does not imply that they are endorsed or recommended by the
World Health Organization in preference to others of a similar nature
that are not mentioned. Errors and omissions excepted, the names of
proprietary products are distinguished by initial capital letters.
CONTENTS
INTRODUCTION
1. PRODUCT IDENTITY AND USES
1.1. Identity
1.2. Physical and chemical properties
1.3. Occurrence
1.4. Analytical methods
1.5. Production and uses
2. SUMMARY AND EVALUATION
2.1. Human exposure
2.2. Uptake, metabolism, and excretion
2.3. Effects on experimental animals
2.4. Effects on human beings
2.5. Effects on the environment
3. CONCLUSIONS AND RECOMMENDATIONS
4. HUMAN HEALTH HAZARDS, PREVENTION AND PROTECTION, EMERGENCY ACTION
4.1. Main human health hazards, prevention and protection,
first aid
4.1.1. Advice to physicians
4.1.2. Health surveillance advice
4.2. Explosion and fire hazards, storage, transport,
spillage, and disposal
4.2.1. Barium metal
4.2.1.1 Explosion hazards
4.2.1.2 Fire hazards
4.2.1.3 Prevention
4.2.1.4 Extinguishing agents
4.2.1.5 Storage
4.2.1.6 Transport
4.2.1.7 Spillage
4.2.1.8 Disposal
4.2.2. Barium compounds
4.2.2.1 Explosion and fire hazards
4.2.2.2 Prevention
4.2.2.3 Extinguishing agents
4.2.2.4 Storage
4.2.2.5 Transport
4.2.2.6 Spillage
4.2.2.7 Disposal
5. HAZARDS FOR THE ENVIRONMENT AND THEIR PREVENTION
6. SUMMARY OF CHEMICAL SAFETY INFORMATION
7. CURRENT REGULATIONS, GUIDELINES, AND STANDARDS
7.1. Previous evaluations by international bodies
7.2. Exposure limit values
7.3. Specific restrictions
7.4. Labelling, packaging, and transport
7.5. Other measures
7.6. Waste disposal
BIBLIOGRAPHY
INTRODUCTION
The Environmental Health Criteria (EHC) documents produced by the
International Programme on Chemical Safety include an assessment of
the effects on the environment and on human health of exposure to a
chemical or combination of chemicals, or physical or biological
agents. They also provide guidelines for setting exposure limits.
The purpose of a Health and Safety Guide is to facilitate the
application of these guidelines in national chemical safety
programmes. The first three sections of a Health and Safety Guide
highlight the relevant technical information in the corresponding EHC.
Section 4 includes advice on preventive and protective measures and
emergency action; health workers should be thoroughly familiar with
the medical information to ensure that they can act efficiently in an
emergency. Within the Guide is a Summary of Chemical Safety
Information which should be readily available, and should be clearly
explained, to all who could come into contact with the chemical. The
section on regulatory information has been extracted from the legal
file of the International Register of Potentially Toxic Chemicals
(IRPTC) and from other United Nations sources.
The target readership includes occupational health services, those in
ministries, governmental agencies, industry, and trade unions who are
involved in the safe use of chemicals and the avoidance of
environmental health hazards, and those wanting more information on
this topic. An attempt has been made to use only terms that will be
familiar to the intended user. However, sections 1 and 2 inevitably
contain some technical terms. A bibliography has been included for
readers who require further background information.
Revision of the information in this Guide will take place in due
course, and the eventual aim is to use standardized terminology.
Comments on any difficulties encountered in using the Guide would be
very helpful and should be addressed to:
The Manager
International Programme on Chemical Safety
Division of Environmental Health
World Health Organization
1211 Geneva 27
Switzerland
THE INFORMATION IN THIS GUIDE SHOULD BE CONSIDERED AS A STARTING POINT
TO A COMPREHENSIVE HEALTH AND SAFETY PROGRAMME
1. PRODUCT IDENTITY AND USES
1.1 Identity
Symbol: Ba
CAS registry number: 7440-39-3
1.2 Physical and Chemical Properties
Some physical and chemical properties of barium are given in the
Summary of Chemical Safety Information (section 6).
1.3 Occurrence
Barium is an alkaline earth metal that occurs in nature in a combined
form. It is present in rocks, minerals, soils, air, natural waters,
and fossil fuels. Some barium salts (e.g., acetate, nitrate, and
chloride) are quite soluble in water, whereas others (e.g., arsenate,
carbonate, oxalate, chromate, fluoride, sulfate, and phosphate) are
very poorly soluble. The water solubility of barium salts, except for
barium sulfate, increases with decreasing pH.
1.4 Analytical Methods
Sampling and handling procedures for barium are those used in general
analytical practice. Three atomic absorption spectrophotometry (AAS)
methods, recommended for the determination of barium, include:the
direct aspiration method, the furnace technique, and emission
spectrometry, using an inductively coupled plasma (ICP) source. Mass
spectrometry, X-ray fluorescence, and neutron activation analysis are
used for special applications.
1.5 Production and Uses
The major raw materials from which barium compounds are obtained are
barite (barium sulfate) and witherite (barium carbonate). The barite
and witherite are converted to metallic barium and other barium
compounds, including barium oxide, hydroxide, peroxide, and a variety
of salts.
Metallic barium is used in the manufacture of alloys. Barium
compounds are used: as loaders for paper, soap, rubber, and linoleum;
in the manufacture of valves, and in the production of lights and
green flares. They are also used: as pigments in the manufacture of
paints; in cement where concrete is exposed to salt-water; in the
radio industry to capture the last traces of gases in vacuum tubes; in
the ceramic and glass industries; as insecticides and rodenticides; as
extinguishers for radium, uranium, and plutonium; and as contrast
material in X-ray medical examination.
2. SUMMARY AND EVALUATION
2.1 Human Exposure
The general population is exposed to barium through air,
drinking-water, and food.
The concentration in air has been estimated to be <0.05 µg/m3
and, assuming an average ventilatory rate of 20 m3/day, a daily
inhalation intake of barium of approximately 1.0 µg can be derived.
Occupational exposure to barium has been found to range from 0.02 to
6.1 mg/m3.
Recent studies from the USA showed exposure levels in drinking-water
ranging from 1 to 20 µg/litre. On the basis of this range, and
assuming a daily consumption of 2 litres of drinking-water, the daily
intake of barium would be 2-40 µg. However, in certain regions of the
USA, barium concentrations of up to 10 000 µg/litre have been
reported. In the United Kingdom (Wales), the average daily intake of
barium from drinking-water has been estimated to be approximately
86 µg.
The major route of barium exposure is through the diet. On the basis
of data from the USA, the dietary intake of barium ranges from 300 to
1700 µg/day, with an average concentration ranging from
600-900 µg/day. In the United Kingdom (Wales), the average intake has
been estimated to be 1240 µg/day.
2.2 Uptake, Metabolism, and Excretion
In human beings, the absorption of barium from the gastrointestinal
tract largely depends on age and the solubility of the compound. Less
than 10% of an ingested quantity is believed to be absorbed in adults;
however, absorption may be significantly higher in children. Inhaled
barium compounds are absorbed through the lungs or directly from the
basal membrane. Poorly soluble compounds may accumulate in the lungs
and removal is slow. Absorbed barium enters the bloodstream and
various soft tissues, and is deposited in the bone. A barium level of
about 20 mg has been found in the average-sized person (70 kg), 93% of
which is localized in the bone.
The metabolism of barium is similar to that of calcium; however,
unlike calcium, barium has no known biological function.
Barium is eliminated in both the faeces and the urine, elimination
varying according to the route of administration and the solubility of
the compound. Barium, which had been absorbed and transported by the
plasma, was found to be almost entirely eliminated from the body
within 24 h.
2.3 Effects on Experimental Animals
Barium compounds, particularly soluble compounds, are toxic in
animals. Oral LD50s in the rat for barium chloride, fluoride, and
nitrate were reported to be 118, 250, and 335 mg/kg body weight,
respectively. Acute effects of barium ingestion include salivation,
nausea, diarrhoea, tachycardia, hypokalaemia, twitching, flaccid
paralysis of skeletal muscle, respiratory muscle paralysis, and
ventricular fibrillation. Respiratory muscle paralysis and
ventricular fibrillation may lead to death. Studies on dogs showed
that acute effects were due to the rapid onset of substantial
hypokalaemia (abnormally low potassium levels in the blood) and could
be prevented or reversed by potassium administration.
No clinical signs of toxicity or microscopic alterations were seen in
rats given tap water containing <250 mg barium/litre for periods of
up to 13 weeks.
Lethal concentrations following acute inhalation exposure have not
been reported. However, at concentrations higher than 5.2 mg
barium/m3, rats exhibited decreases in body weight, blood glucose,
and haemoglobin, and an increase in leukocytes.
In rabbits, topical administration of barium resulted in mild skin
irritation and severe eye irritation.
The results of one study on mice and rats did not reveal any evidence
of carcinogenicity for barium.
Both oral and inhalation exposure to barium carbonate induced adverse
reproductive effects in male and female rats.
There is limited evidence of the teratogenic potential of barium.
2.4 Effects on Human Beings
Barium doses as low as 0.2-0.5 g (3-7 mg/kg body weight), generally
resulting from the ingestion of barium chloride or carbonate, have
been found to lead to toxic effects in adult human beings. In
untreated cases, doses of 3-5 g (40-70 mg/kg body weight) were lethal.
Clinical features of barium poisoning include: acute gastroenteritis,
loss of deep reflexes with onset of muscular paralysis, and
progressive muscular paralysis. The muscular paralysis appears to be
related to severe hypokalaemia. In most reported cases, rapid and
uneventful recovery occurred after treatment with infused potassium
salts (carbonate or lactate) and/or oral administration of sodium
sulfate.
Adverse health effects have been observed in sensitive individuals
(e.g., diuresis patients) following exposure to barium as a medical
X-ray contrast medium.
In the workplace, inhaled barium has resulted in baritosis. A
significantly higher prevalence of hypertension was observed in
workers exposed to high levels of airborne barium than in unexposed
workers.
The results of early, limited, epidemiological studies, relating
exposure to low levels of barium with cardiovascular disease and
mortality, were inconsistent and inconclusive. In a later
epidemiological study, no conclusive evidence of barium-induced
effects on blood pressure was revealed. No effects on blood pressure
were identified in a short-term study in which volunteers consumed
increasing levels of barium up to 10 mg/litre in drinking-water.
There is no conclusive evidence that barium compounds induce
reproductive, teratogenic, or carcinogenic effects in human beings.
2.5 Effects on the Environment
Barium has been reported to inhibit growth and cellular processes in
microorganisms, and to affect the development of germinating spores.
No information was obtained on the adverse effects of barium on
terrestrial plants or wildlife. No toxic effects have been reported
in aquatic plants due to barium at the usual concentrations found in
water. Reported LC50 values for fish in fresh water range from
46 to 78 mg/litre. Reproduction and growth in Daphnia spp. were
impaired by barium concentrations of 5.8 mg/litre.
3. CONCLUSIONS AND RECOMMENDATIONS
Barium, at concentrations normally found in the environment, does not
pose any significant risk for the general population. However, for
specific subpopulations, and under conditions of high barium exposure,
the potential for adverse health effects should be taken into account.
Few data are available to evaluate the risk to the environment posed
by barium. On the basis of the available information on the toxic
effects of barium in Daphnia spp., it appears that high
concentrations may represent a risk to populations of some aquatic
organisms.
In order to adopt adequate preventive measures in the workplace, more
data on exposure levels should be obtained, including human
monitoring data.
4. HUMAN HEALTH HAZARDS, PREVENTION AND PROTECTION, EMERGENCY ACTION
4.1 Main Human Health Hazards, Prevention and Protection, First Aid
The three stages of poisoning from soluble barium compounds are acute
gastroenteritis, loss of reflexes with the onset of muscular
paralysis, and progressive muscular paralysis. When ingested, barium
compounds exert profound effects on all muscles, especially the heart.
In addition, exposure to metallic barium may result in severe burns,
through explosion and fire. The human health hazards associated with
certain types of exposure to metallic barium, together with preventive
and protective measures and first-aid recommendations, are listed in
the Summary of Chemical Safety Information (section 6).
4.1.1 Advice to physicians
Immediate care should consist of the administration of 30 g sodium
sulfate in 250 ml of water (a glass of water), repeated in one hour.
In the case of very severe symptoms, intravenous administration of
potassium bicarbonate or lactate, to overcome hypokalaemia, is
necessary, and should be carried out, under controlled medical
conditions, only by qualified medical personnel.
4.1.2 Health surveillance advice
Persons handling barium and its compounds should undergo periodic
medical examination. Special consideration should be given to the
skin, eyes, heart, and lungs, and serum potassium levels should be
measured at each examination.
4.2 Explosion and Fire Hazards, Storage, Transport, Spillage, and
Disposal
Barium metals and barium compounds have been described separately;
section 4.2.1 refers to barium as a metal, while section 4.2.2 refers
to barium compounds.
4.2.1 Barium metal
4.2.1.1 Explosion hazards
Metallic barium reacts exothermically on contact with water, releasing
flammable hydrogen gas, which may be ignited by the heat of the
reaction. Fine powders or dusts of barium can explode when heated.
Barium runoff to sewer water may create an explosion or fire.
4.2.1.2 Fire hazards
Barium in the form of finely ground powder or fumes is flammable or
explosive, when exposed to heat or flames.
4.2.1.3 Prevention
Keep barium away from water, heat, sparks, and open flames.
4.2.1.4 Extinguishing agents
Move barium containers from fire area, if possible. Do not use water,
carbon dioxide, or foam. In case of small fires, use dry chemical
powder, soda ash, or lime. In case of large fires, withdraw from area
and let fire burn.
4.2.1.5 Storage
Store barium under inert gas, petroleum, argon, or oxygen-free liquid.
Store away from the following: water, most acids, ammonia, halogens,
and oxidizers.
4.2.1.6 Transport
Package barium in sealed containers. Pack glass bottles together with
cushioning material in a wooden box; pack cans together in a wooden
box. Label metal drums with the warning "Dangerous When Wet". Limit
transportation of barium to cargo ships or passenger ships carrying
not more than 25 passengers; on ships, limit stowage to above the deck
or under the deck, preferably the latter. During transportation by
air (single or combination packagings), steel drums, aluminum drums,
plastic drums, or plastic jerricans are required as the inner shell,
and wooden, plywood, or fibreboard boxes are required as the outer
shell.
4.2.1.7 Spillage
In case of spillage, shut off ignition sources. Do not touch spilled
material. Stop leak, if possible to do so without risk. Water should
not come into contact with spilled material or the inside of the
container. For small dry spills, use a clean shovel to place material
into a clean, dry container and cover; move containers from area of
spillage. Cover dry powder spills with plastic sheet or tarp to
minimize spreading. Keep unnecessary persons away. Isolate area and
prevent anyone from entering.
4.2.1.8 Disposal
Material in the elemental state should be recovered for re-use or
recycling. Recovered metal should be stored under a layer of kerosene
and kept dry.
4.2.2 Barium compounds
4.2.2.1 Explosion and fire hazards
Specific soluble barium compounds, such as barium oxide, peroxide, and
nitrate, can cause an explosion or fire, when in contact with water,
carbon dioxide, or hydrogen sulfide.
4.2.2.2 Prevention
Handle with care, avoid inhalation and contact with skin. Do not eat,
drink, or smoke in the workplace.
4.2.2.3 Extinguishing agents
Move containers from the fire area, if possible. In case of small
fires, use dry chemical powder, soda, or lime. In case of large
fires, withdraw from the area and let the fire burn.
4.2.2.4 Storage
Store in closed labelled containers.
4.2.2.5 Transport
Package barium compounds in sealed containers. Pack glass bottles,
together with cushioning material, in a wooden box; pack cans together
in a wooden box. Label metal drums with the warning "Dangerous When
Wet". Limit transportation of barium compounds to cargo ships or
passenger ships carrying not more than 25 passengers; on ships, limit
stowage to above the deck or under the deck, preferably the latter.
During transportation by air (single or combination packagings), steel
drums, aluminum drums, plastic drums, or plastic jerricans are
required as the inner shell, and wooden, plywood, or fibreboard boxes,
as the outer shell.
4.2.2.6 Spillage
In case of spillage, shut off ignition sources. Do not touch spilled
material. Stop leak, if possible to do so without risk. Water should
not come into contact with the spilled material or the inside of the
container. For small dry spills, use a clean shovel to place in
clean, dry container and cover; move containers from area of spillage.
Take up liquid spills with sand or absorbent material and place in
containers for later disposal. Larger spills should be contained with
a barrier for later disposal. Cover dry powder spills with plastic
sheet or tarpaulin, to minimize spreading. Keep unnecessary persons
away. Isolate area, and prevent anyone from entering.
4.2.2.7 Disposal
Soluble barium compounds can be removed from water by the addition of
controlled amounts of sulfuric acid or acidified sulfate salts, to
form insoluble barium sulfate.
5. HAZARDS FOR THE ENVIRONMENT AND THEIR PREVENTION
As barium at a concentration of 5.8 mg/litre has been observed to
impair reproduction and growth in Daphnia spp., similar
concentrations may present a risk for other aquatic organisms.
Avoid contamination of soil, water, and the atmosphere by using proper
methods of processing, transport, and waste disposal.
6. SUMMARY OF CHEMICAL SAFETY INFORMATION
This summary should be easily available to all health workers
concerned with, and users of, barium. It should be displayed at, or
near, entrances to areas where there is potential exposure to barium,
and on processing equipment and containers. The summary should be
translated into the appropriate language(s). All persons potentially
exposed to the chemical should also have the instructions in the
summary clearly explained.
Space is available for insertion of the National Occupational
Exposure Limit, the address and telephone number of the National
Poison Control Centre, and local trade names.
Barium (Ba)
PHYSICAL PROPERTIES OTHER CHARACTERISTICS
Relative molecular mass 56 Extremely reactive with water, ammonia, halogens,
Appearance yellowish-white oxygen and most acids
solid
Melting point (°C) 725
Boiling point (°C) 1640
Solubility in water (20°C) reacts with
release of H2
Density (20°C) 3.51
Vapour pressure (20°C) 2
Electronegativity 1.02
Flame coloration test green
HAZARDS/SYMPTOMS PREVENTION AND PROTECTION FIRST AID
SKIN: Irritation Avoid exposure; wear protective Remove contaminated clothing; wash skin
clothing and gloves with plenty of water and soap
EYE: Irritation; redness: pain Wear safety goggles if there is a Flush eyes with plenty of water for 15
possibility of eye contact minutes
INHALATION (metallic powder): Avoid exposure by using ventilation, Fresh air, administer oxygen, if
Sore throat, coughing, shortness local exhaust, or breathing protection required
of breath, trembling, vomiting by a suitable respirator
INGESTION: Salivation, vomiting, Do not eat, drink, or smoke when Rinse mouth; induce vomiting in
abdominal pain; gastroenteritis; handling the material conscious patients; administer sodium
giddiness; stimulation of all muscle sulfate (30 g in 250 ml water); if
types; hypokalaemia; delayed kidney swallowed, begin gastric lavage (stomach
damage; vasoconstriction, irregular wash) followed by saline catharsis,
heartbeat; death may occur from seek medical attention immediately
cardiac or respiratory failure
SPILLAGE STORAGE FIRE AND EXPLOSION
Do not touch spilled material; Store under inert gas, petroleum, Finely ground particle or
do not get water on the spillage; argon, or oxygen-free liquid fumes are flammable or
for dry spills, use a clean shovel explosive
to place material in clean, dry
container
WASTE DISPOSAL
Recover elemental barium for re-use National Occupational Exposure Limit:
or recycling; store under a layer of
kerosene and keep it dry National Poison Control Centre:
7. CURRENT REGULATIONS, GUIDELINES, AND STANDARDS
The information given in this section has been extracted from the
International Register of Potentially Toxic Chemicals (IRPTC) legal
file. A full reference to the original national document from which
the information was extracted can be obtained from IRPTC. When no
effective date appears in the IRPTC legal file, the year of the
reference from which the data are taken is indicated by (r).
The reader should be aware that regulatory decisions about chemicals
taken in a certain country can only be fully understood in the
framework of the legislation of that country. Furthermore, the
regulations and guidelines of all countries are subject to change and
should always be verified with appropriate regulatory authorities
before application.
7.1 Previous Evaluations by International Bodies
The International Agency for Research on Cancer (IARC) evaluated the
carcinogenicity of barium chromate (VI) and concluded that there was
sufficient evidence for carcinogenicity in human beings (positive
carcinogen) and insufficient evidence in animals. However, the
carcinogenic properties of barium chromate are related to the presence
of chromium (VI) and not barium.
7.2 Exposure Limit Values
See table on pages 24-25.
7.3 Specific Restrictions
No specific restrictions were found.
7.4 Labelling, Packaging, and Transport
European Economic Community legislation requires labelling as a
dangerous substance using the symbol:
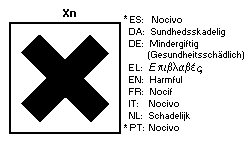 The label must read:
Harmful by inhalation - avoid contact with skin.
The European Economic Community legislation on labelling classifies
barium as a poisonous substance for the purpose of determining the
label on preparations containing this substance.
The United Kingdom legislation on regulations requires that all
general requirements concerning labelling, containers, storage, and
transport are laid down; this applies to all barium salts except
barium sulfate.
The label that both recommend is:
The label must read:
Harmful by inhalation - avoid contact with skin.
The European Economic Community legislation on labelling classifies
barium as a poisonous substance for the purpose of determining the
label on preparations containing this substance.
The United Kingdom legislation on regulations requires that all
general requirements concerning labelling, containers, storage, and
transport are laid down; this applies to all barium salts except
barium sulfate.
The label that both recommend is:
 EXPOSURE LIMIT VALUES
Medium Specification Country/ Exposure limit description Value Effective
organization date
AIR Occupational Argentina Maximum allowable concentration (MAC) 0.5 mg/m3 1979
- Time-weighted average (TWA)
Short-term exposure limit (STEL) 0.5 mg/m3 1979
Australia Threshold limit value (TLV) 0.5 mg/m3 1985(r)
- Time-weighted average (TWA)
Belgium Threshold limit value (TLV) 0.5 mg/m3 1980(r)
- Time-weighted average (TWA)
Canada Maximum allowable concentration (MAC) 0.5 mg/m3 1985(r)
- Time-weighted average (TWA)
Finland Maximum allowable concentration (MAC) 0.5 mg/m3 1985(r)
- Time-weighted average (TWA)
Germany, Maximum at work-site concentration (MAK) 0.5 mg/m3 1987(r)
Federal - 8 h time-weighted average
Republic of Short-term exposure limit (STEL) - 30 minutes 1.0 mg/m3 1987(r)
United Kingdom Recommended limits (RECL) 0.5 mg/m3 1987(r)
- Time-weighted average
USA (ACGIH) Threshold limit value (TLV) 0.5 mg/m3 1986(r)
- Time weighted average
USA (OSHA) Threshold limit value (TLV) 0.5 mg/m3 1985(r)
- Time weighted average
Medium Specification Country/ Exposure limit description Value Effective
organization date
WATER Drinking- Canada Maximum allowable concentration (MAC) 1.0 mg/litre 1987
- Guideline level for water quality
European Maximum allowable concentration (MAC) 0.1 mg/litre 1982
Economic - Guideline level requirements
Community
USA Reported quantity requirement (RQR) 1.0 mg/litre 1981(r)
- Bottled water
Maximum allowable concentration (MAC) 1.0 mg/litre 1986(r)
- Public water systems
Surface European Reported quantity requirement (RQR) 0.1 mg/litre 1977
Economic - intended for the abstraction of
Community drinking water; simple water treatment
methods
- for normal and intensive methods 1.0 mg/litre 1977
FOOD USA Maximum permissible concentration (MPC) 0.5 g/kg 1986 (r)
- for colours that may be used in food,
drugs, and/or cosmetics
TWA = Time-weighted average over one working day (usually 8 h).
7.5 Other Measures
United Kingdom legislation classifies barium as a poisonous substance
that is not to be sold except by a person lawfully conducting a retail
pharmacy business, and requires that proper records be kept of the
sales.
Under the Hazardous Products Act, Canadian legislation prohibits the
advertising, sale, or import of toys, equipment, or other products,
for use by children in learning or play, which have a decorative or
protective coating that contains 0.1% barium.
7.6 Waste Disposal
European Economic Community legislation on waste disposal requires
that Member States should take the necessary steps to limit the
introduction of barium and its compounds into ground water, by
subjecting to prior investigation and authorization all direct
discharges, and disposal or tipping that might lead to indirect
discharges, and all indirect discharges.
United Kingdom legislation classifies barium as a "special waste" that
requires completion of a consignment note for the disposal
authorities. This applies to controlled waste containing, or
consisting of, barium compounds.
United States legislation requires an owner or operator of a certain
type of hazardous waste storage, treatment, or disposal facility to
install a ground water monitoring system and to periodically report on
the concentrations of this substance. In addition, permits are
required for the discharge of any pollutant from any point source into
US waters. Quantitative data must be reported in the application.
The level applies to total barium. Waste regulations also require
that the analytical method(s) used in determining the presence of
barium in solid waste be specified.
BIBLIOGRAPHY
ACGIH (1986) Documentation of the threshold limit values and
biological exposure indices. 5th ed, Cincinnati, Ohio, American
Conference of Governmental Industrial Hygienists, p.47.
BRANSFORD, J., ed. (1989) Material Safety Data Sheet (MSDS).
CD-ROM. New York, Occupational Health Services, Inc.
BRETHERICK, L., ed. (1986) Hazards in the chemical laboratory.
London, London Royal Society of Chemistry.
IARC (1980) Some metals and metallic compounds. Lyon, International
Agency for Research on Cancer, p. 205 (IARC Monographs on the
Evaluation of the Carcinogenic Risk of Chemicals to Humans, Vol. 23).
IATA (1986) IATA Dangerous goods regulations. 27th ed., Montreal,
International Air Transport Association.
IMCO (1983) International maritime dangerous goods Code. Vol. III.
Amendment 21-83. London, Intergovernmental Maritime Consultative
Organization, p. 4147.
IRPTC (1989) Data profile: Barium. Geneva, (International Register
of Potentially Toxic Chemicals), United Nations Environment Programme.
ITI (1978) Toxic and hazardous industrial chemicals safety manual.
Tokyo, International Technical Information Institute, pp. 57-58.
LENGA, R. (1985) The Sigma-Aldrich library of chemical safety data.
Steinheim, Federal Republic of Germany, Sigma-Aldrich Chemical
Corporation, p. 143.
NATIONAL LIBRARY OF MEDICINE (1989) Hazardous Substance Data Bank
(HSDB). Online, Washington, DC, National Institutes of Health,
MEDLARS Management Section.
PROCTOR, N. & HUGHES, J. (1978) Chemical hazards of the workplace.
Philadelphia, Pennsylvania, J.B. Lippincott Company, pp. 116-117.
SITTIG, M. (1979) Hazardous and toxic effects of industrial
chemicals. Park Ridge, New Jersey, Noyes Data Corporation,
pp. 51-52.
US EPA (1988) Risk screening guide. Barium. Washington, DC, US
Environmental Protection Agency, Office of Toxic Substances
(Attachment SARA Section 313 ROADMAPS Information).
US EPA (1989) Integrated Risk Information System (IRIS). Reference
dose (RfD) for oral exposure for barium. Online, Cincinnati, Ohio,
Office of Health and Environmental Assessment, Environmental Criteria
and Assessment Office.
US NIOSH (1989) Registry of toxic effects of chemical substances
(RTECS). Online, Cincinnati, Ohio, National Institute of
Occupational Safety and Health, US Department of Health and Human
Services, Public Health Service, Centers for Disease Control.
WHO (1989) Environmental Health Criteria 107: Barium. Geneva, World
Health Organization.
EXPOSURE LIMIT VALUES
Medium Specification Country/ Exposure limit description Value Effective
organization date
AIR Occupational Argentina Maximum allowable concentration (MAC) 0.5 mg/m3 1979
- Time-weighted average (TWA)
Short-term exposure limit (STEL) 0.5 mg/m3 1979
Australia Threshold limit value (TLV) 0.5 mg/m3 1985(r)
- Time-weighted average (TWA)
Belgium Threshold limit value (TLV) 0.5 mg/m3 1980(r)
- Time-weighted average (TWA)
Canada Maximum allowable concentration (MAC) 0.5 mg/m3 1985(r)
- Time-weighted average (TWA)
Finland Maximum allowable concentration (MAC) 0.5 mg/m3 1985(r)
- Time-weighted average (TWA)
Germany, Maximum at work-site concentration (MAK) 0.5 mg/m3 1987(r)
Federal - 8 h time-weighted average
Republic of Short-term exposure limit (STEL) - 30 minutes 1.0 mg/m3 1987(r)
United Kingdom Recommended limits (RECL) 0.5 mg/m3 1987(r)
- Time-weighted average
USA (ACGIH) Threshold limit value (TLV) 0.5 mg/m3 1986(r)
- Time weighted average
USA (OSHA) Threshold limit value (TLV) 0.5 mg/m3 1985(r)
- Time weighted average
Medium Specification Country/ Exposure limit description Value Effective
organization date
WATER Drinking- Canada Maximum allowable concentration (MAC) 1.0 mg/litre 1987
- Guideline level for water quality
European Maximum allowable concentration (MAC) 0.1 mg/litre 1982
Economic - Guideline level requirements
Community
USA Reported quantity requirement (RQR) 1.0 mg/litre 1981(r)
- Bottled water
Maximum allowable concentration (MAC) 1.0 mg/litre 1986(r)
- Public water systems
Surface European Reported quantity requirement (RQR) 0.1 mg/litre 1977
Economic - intended for the abstraction of
Community drinking water; simple water treatment
methods
- for normal and intensive methods 1.0 mg/litre 1977
FOOD USA Maximum permissible concentration (MPC) 0.5 g/kg 1986 (r)
- for colours that may be used in food,
drugs, and/or cosmetics
TWA = Time-weighted average over one working day (usually 8 h).
7.5 Other Measures
United Kingdom legislation classifies barium as a poisonous substance
that is not to be sold except by a person lawfully conducting a retail
pharmacy business, and requires that proper records be kept of the
sales.
Under the Hazardous Products Act, Canadian legislation prohibits the
advertising, sale, or import of toys, equipment, or other products,
for use by children in learning or play, which have a decorative or
protective coating that contains 0.1% barium.
7.6 Waste Disposal
European Economic Community legislation on waste disposal requires
that Member States should take the necessary steps to limit the
introduction of barium and its compounds into ground water, by
subjecting to prior investigation and authorization all direct
discharges, and disposal or tipping that might lead to indirect
discharges, and all indirect discharges.
United Kingdom legislation classifies barium as a "special waste" that
requires completion of a consignment note for the disposal
authorities. This applies to controlled waste containing, or
consisting of, barium compounds.
United States legislation requires an owner or operator of a certain
type of hazardous waste storage, treatment, or disposal facility to
install a ground water monitoring system and to periodically report on
the concentrations of this substance. In addition, permits are
required for the discharge of any pollutant from any point source into
US waters. Quantitative data must be reported in the application.
The level applies to total barium. Waste regulations also require
that the analytical method(s) used in determining the presence of
barium in solid waste be specified.
BIBLIOGRAPHY
ACGIH (1986) Documentation of the threshold limit values and
biological exposure indices. 5th ed, Cincinnati, Ohio, American
Conference of Governmental Industrial Hygienists, p.47.
BRANSFORD, J., ed. (1989) Material Safety Data Sheet (MSDS).
CD-ROM. New York, Occupational Health Services, Inc.
BRETHERICK, L., ed. (1986) Hazards in the chemical laboratory.
London, London Royal Society of Chemistry.
IARC (1980) Some metals and metallic compounds. Lyon, International
Agency for Research on Cancer, p. 205 (IARC Monographs on the
Evaluation of the Carcinogenic Risk of Chemicals to Humans, Vol. 23).
IATA (1986) IATA Dangerous goods regulations. 27th ed., Montreal,
International Air Transport Association.
IMCO (1983) International maritime dangerous goods Code. Vol. III.
Amendment 21-83. London, Intergovernmental Maritime Consultative
Organization, p. 4147.
IRPTC (1989) Data profile: Barium. Geneva, (International Register
of Potentially Toxic Chemicals), United Nations Environment Programme.
ITI (1978) Toxic and hazardous industrial chemicals safety manual.
Tokyo, International Technical Information Institute, pp. 57-58.
LENGA, R. (1985) The Sigma-Aldrich library of chemical safety data.
Steinheim, Federal Republic of Germany, Sigma-Aldrich Chemical
Corporation, p. 143.
NATIONAL LIBRARY OF MEDICINE (1989) Hazardous Substance Data Bank
(HSDB). Online, Washington, DC, National Institutes of Health,
MEDLARS Management Section.
PROCTOR, N. & HUGHES, J. (1978) Chemical hazards of the workplace.
Philadelphia, Pennsylvania, J.B. Lippincott Company, pp. 116-117.
SITTIG, M. (1979) Hazardous and toxic effects of industrial
chemicals. Park Ridge, New Jersey, Noyes Data Corporation,
pp. 51-52.
US EPA (1988) Risk screening guide. Barium. Washington, DC, US
Environmental Protection Agency, Office of Toxic Substances
(Attachment SARA Section 313 ROADMAPS Information).
US EPA (1989) Integrated Risk Information System (IRIS). Reference
dose (RfD) for oral exposure for barium. Online, Cincinnati, Ohio,
Office of Health and Environmental Assessment, Environmental Criteria
and Assessment Office.
US NIOSH (1989) Registry of toxic effects of chemical substances
(RTECS). Online, Cincinnati, Ohio, National Institute of
Occupational Safety and Health, US Department of Health and Human
Services, Public Health Service, Centers for Disease Control.
WHO (1989) Environmental Health Criteria 107: Barium. Geneva, World
Health Organization.
 The label must read:
Harmful by inhalation - avoid contact with skin.
The European Economic Community legislation on labelling classifies
barium as a poisonous substance for the purpose of determining the
label on preparations containing this substance.
The United Kingdom legislation on regulations requires that all
general requirements concerning labelling, containers, storage, and
transport are laid down; this applies to all barium salts except
barium sulfate.
The label that both recommend is:
The label must read:
Harmful by inhalation - avoid contact with skin.
The European Economic Community legislation on labelling classifies
barium as a poisonous substance for the purpose of determining the
label on preparations containing this substance.
The United Kingdom legislation on regulations requires that all
general requirements concerning labelling, containers, storage, and
transport are laid down; this applies to all barium salts except
barium sulfate.
The label that both recommend is:
 EXPOSURE LIMIT VALUES
Medium Specification Country/ Exposure limit description Value Effective
organization date
AIR Occupational Argentina Maximum allowable concentration (MAC) 0.5 mg/m3 1979
- Time-weighted average (TWA)
Short-term exposure limit (STEL) 0.5 mg/m3 1979
Australia Threshold limit value (TLV) 0.5 mg/m3 1985(r)
- Time-weighted average (TWA)
Belgium Threshold limit value (TLV) 0.5 mg/m3 1980(r)
- Time-weighted average (TWA)
Canada Maximum allowable concentration (MAC) 0.5 mg/m3 1985(r)
- Time-weighted average (TWA)
Finland Maximum allowable concentration (MAC) 0.5 mg/m3 1985(r)
- Time-weighted average (TWA)
Germany, Maximum at work-site concentration (MAK) 0.5 mg/m3 1987(r)
Federal - 8 h time-weighted average
Republic of Short-term exposure limit (STEL) - 30 minutes 1.0 mg/m3 1987(r)
United Kingdom Recommended limits (RECL) 0.5 mg/m3 1987(r)
- Time-weighted average
USA (ACGIH) Threshold limit value (TLV) 0.5 mg/m3 1986(r)
- Time weighted average
USA (OSHA) Threshold limit value (TLV) 0.5 mg/m3 1985(r)
- Time weighted average
Medium Specification Country/ Exposure limit description Value Effective
organization date
WATER Drinking- Canada Maximum allowable concentration (MAC) 1.0 mg/litre 1987
- Guideline level for water quality
European Maximum allowable concentration (MAC) 0.1 mg/litre 1982
Economic - Guideline level requirements
Community
USA Reported quantity requirement (RQR) 1.0 mg/litre 1981(r)
- Bottled water
Maximum allowable concentration (MAC) 1.0 mg/litre 1986(r)
- Public water systems
Surface European Reported quantity requirement (RQR) 0.1 mg/litre 1977
Economic - intended for the abstraction of
Community drinking water; simple water treatment
methods
- for normal and intensive methods 1.0 mg/litre 1977
FOOD USA Maximum permissible concentration (MPC) 0.5 g/kg 1986 (r)
- for colours that may be used in food,
drugs, and/or cosmetics
TWA = Time-weighted average over one working day (usually 8 h).
7.5 Other Measures
United Kingdom legislation classifies barium as a poisonous substance
that is not to be sold except by a person lawfully conducting a retail
pharmacy business, and requires that proper records be kept of the
sales.
Under the Hazardous Products Act, Canadian legislation prohibits the
advertising, sale, or import of toys, equipment, or other products,
for use by children in learning or play, which have a decorative or
protective coating that contains 0.1% barium.
7.6 Waste Disposal
European Economic Community legislation on waste disposal requires
that Member States should take the necessary steps to limit the
introduction of barium and its compounds into ground water, by
subjecting to prior investigation and authorization all direct
discharges, and disposal or tipping that might lead to indirect
discharges, and all indirect discharges.
United Kingdom legislation classifies barium as a "special waste" that
requires completion of a consignment note for the disposal
authorities. This applies to controlled waste containing, or
consisting of, barium compounds.
United States legislation requires an owner or operator of a certain
type of hazardous waste storage, treatment, or disposal facility to
install a ground water monitoring system and to periodically report on
the concentrations of this substance. In addition, permits are
required for the discharge of any pollutant from any point source into
US waters. Quantitative data must be reported in the application.
The level applies to total barium. Waste regulations also require
that the analytical method(s) used in determining the presence of
barium in solid waste be specified.
BIBLIOGRAPHY
ACGIH (1986) Documentation of the threshold limit values and
biological exposure indices. 5th ed, Cincinnati, Ohio, American
Conference of Governmental Industrial Hygienists, p.47.
BRANSFORD, J., ed. (1989) Material Safety Data Sheet (MSDS).
CD-ROM. New York, Occupational Health Services, Inc.
BRETHERICK, L., ed. (1986) Hazards in the chemical laboratory.
London, London Royal Society of Chemistry.
IARC (1980) Some metals and metallic compounds. Lyon, International
Agency for Research on Cancer, p. 205 (IARC Monographs on the
Evaluation of the Carcinogenic Risk of Chemicals to Humans, Vol. 23).
IATA (1986) IATA Dangerous goods regulations. 27th ed., Montreal,
International Air Transport Association.
IMCO (1983) International maritime dangerous goods Code. Vol. III.
Amendment 21-83. London, Intergovernmental Maritime Consultative
Organization, p. 4147.
IRPTC (1989) Data profile: Barium. Geneva, (International Register
of Potentially Toxic Chemicals), United Nations Environment Programme.
ITI (1978) Toxic and hazardous industrial chemicals safety manual.
Tokyo, International Technical Information Institute, pp. 57-58.
LENGA, R. (1985) The Sigma-Aldrich library of chemical safety data.
Steinheim, Federal Republic of Germany, Sigma-Aldrich Chemical
Corporation, p. 143.
NATIONAL LIBRARY OF MEDICINE (1989) Hazardous Substance Data Bank
(HSDB). Online, Washington, DC, National Institutes of Health,
MEDLARS Management Section.
PROCTOR, N. & HUGHES, J. (1978) Chemical hazards of the workplace.
Philadelphia, Pennsylvania, J.B. Lippincott Company, pp. 116-117.
SITTIG, M. (1979) Hazardous and toxic effects of industrial
chemicals. Park Ridge, New Jersey, Noyes Data Corporation,
pp. 51-52.
US EPA (1988) Risk screening guide. Barium. Washington, DC, US
Environmental Protection Agency, Office of Toxic Substances
(Attachment SARA Section 313 ROADMAPS Information).
US EPA (1989) Integrated Risk Information System (IRIS). Reference
dose (RfD) for oral exposure for barium. Online, Cincinnati, Ohio,
Office of Health and Environmental Assessment, Environmental Criteria
and Assessment Office.
US NIOSH (1989) Registry of toxic effects of chemical substances
(RTECS). Online, Cincinnati, Ohio, National Institute of
Occupational Safety and Health, US Department of Health and Human
Services, Public Health Service, Centers for Disease Control.
WHO (1989) Environmental Health Criteria 107: Barium. Geneva, World
Health Organization.
EXPOSURE LIMIT VALUES
Medium Specification Country/ Exposure limit description Value Effective
organization date
AIR Occupational Argentina Maximum allowable concentration (MAC) 0.5 mg/m3 1979
- Time-weighted average (TWA)
Short-term exposure limit (STEL) 0.5 mg/m3 1979
Australia Threshold limit value (TLV) 0.5 mg/m3 1985(r)
- Time-weighted average (TWA)
Belgium Threshold limit value (TLV) 0.5 mg/m3 1980(r)
- Time-weighted average (TWA)
Canada Maximum allowable concentration (MAC) 0.5 mg/m3 1985(r)
- Time-weighted average (TWA)
Finland Maximum allowable concentration (MAC) 0.5 mg/m3 1985(r)
- Time-weighted average (TWA)
Germany, Maximum at work-site concentration (MAK) 0.5 mg/m3 1987(r)
Federal - 8 h time-weighted average
Republic of Short-term exposure limit (STEL) - 30 minutes 1.0 mg/m3 1987(r)
United Kingdom Recommended limits (RECL) 0.5 mg/m3 1987(r)
- Time-weighted average
USA (ACGIH) Threshold limit value (TLV) 0.5 mg/m3 1986(r)
- Time weighted average
USA (OSHA) Threshold limit value (TLV) 0.5 mg/m3 1985(r)
- Time weighted average
Medium Specification Country/ Exposure limit description Value Effective
organization date
WATER Drinking- Canada Maximum allowable concentration (MAC) 1.0 mg/litre 1987
- Guideline level for water quality
European Maximum allowable concentration (MAC) 0.1 mg/litre 1982
Economic - Guideline level requirements
Community
USA Reported quantity requirement (RQR) 1.0 mg/litre 1981(r)
- Bottled water
Maximum allowable concentration (MAC) 1.0 mg/litre 1986(r)
- Public water systems
Surface European Reported quantity requirement (RQR) 0.1 mg/litre 1977
Economic - intended for the abstraction of
Community drinking water; simple water treatment
methods
- for normal and intensive methods 1.0 mg/litre 1977
FOOD USA Maximum permissible concentration (MPC) 0.5 g/kg 1986 (r)
- for colours that may be used in food,
drugs, and/or cosmetics
TWA = Time-weighted average over one working day (usually 8 h).
7.5 Other Measures
United Kingdom legislation classifies barium as a poisonous substance
that is not to be sold except by a person lawfully conducting a retail
pharmacy business, and requires that proper records be kept of the
sales.
Under the Hazardous Products Act, Canadian legislation prohibits the
advertising, sale, or import of toys, equipment, or other products,
for use by children in learning or play, which have a decorative or
protective coating that contains 0.1% barium.
7.6 Waste Disposal
European Economic Community legislation on waste disposal requires
that Member States should take the necessary steps to limit the
introduction of barium and its compounds into ground water, by
subjecting to prior investigation and authorization all direct
discharges, and disposal or tipping that might lead to indirect
discharges, and all indirect discharges.
United Kingdom legislation classifies barium as a "special waste" that
requires completion of a consignment note for the disposal
authorities. This applies to controlled waste containing, or
consisting of, barium compounds.
United States legislation requires an owner or operator of a certain
type of hazardous waste storage, treatment, or disposal facility to
install a ground water monitoring system and to periodically report on
the concentrations of this substance. In addition, permits are
required for the discharge of any pollutant from any point source into
US waters. Quantitative data must be reported in the application.
The level applies to total barium. Waste regulations also require
that the analytical method(s) used in determining the presence of
barium in solid waste be specified.
BIBLIOGRAPHY
ACGIH (1986) Documentation of the threshold limit values and
biological exposure indices. 5th ed, Cincinnati, Ohio, American
Conference of Governmental Industrial Hygienists, p.47.
BRANSFORD, J., ed. (1989) Material Safety Data Sheet (MSDS).
CD-ROM. New York, Occupational Health Services, Inc.
BRETHERICK, L., ed. (1986) Hazards in the chemical laboratory.
London, London Royal Society of Chemistry.
IARC (1980) Some metals and metallic compounds. Lyon, International
Agency for Research on Cancer, p. 205 (IARC Monographs on the
Evaluation of the Carcinogenic Risk of Chemicals to Humans, Vol. 23).
IATA (1986) IATA Dangerous goods regulations. 27th ed., Montreal,
International Air Transport Association.
IMCO (1983) International maritime dangerous goods Code. Vol. III.
Amendment 21-83. London, Intergovernmental Maritime Consultative
Organization, p. 4147.
IRPTC (1989) Data profile: Barium. Geneva, (International Register
of Potentially Toxic Chemicals), United Nations Environment Programme.
ITI (1978) Toxic and hazardous industrial chemicals safety manual.
Tokyo, International Technical Information Institute, pp. 57-58.
LENGA, R. (1985) The Sigma-Aldrich library of chemical safety data.
Steinheim, Federal Republic of Germany, Sigma-Aldrich Chemical
Corporation, p. 143.
NATIONAL LIBRARY OF MEDICINE (1989) Hazardous Substance Data Bank
(HSDB). Online, Washington, DC, National Institutes of Health,
MEDLARS Management Section.
PROCTOR, N. & HUGHES, J. (1978) Chemical hazards of the workplace.
Philadelphia, Pennsylvania, J.B. Lippincott Company, pp. 116-117.
SITTIG, M. (1979) Hazardous and toxic effects of industrial
chemicals. Park Ridge, New Jersey, Noyes Data Corporation,
pp. 51-52.
US EPA (1988) Risk screening guide. Barium. Washington, DC, US
Environmental Protection Agency, Office of Toxic Substances
(Attachment SARA Section 313 ROADMAPS Information).
US EPA (1989) Integrated Risk Information System (IRIS). Reference
dose (RfD) for oral exposure for barium. Online, Cincinnati, Ohio,
Office of Health and Environmental Assessment, Environmental Criteria
and Assessment Office.
US NIOSH (1989) Registry of toxic effects of chemical substances
(RTECS). Online, Cincinnati, Ohio, National Institute of
Occupational Safety and Health, US Department of Health and Human
Services, Public Health Service, Centers for Disease Control.
WHO (1989) Environmental Health Criteria 107: Barium. Geneva, World
Health Organization.
