
IPCS INTERNATIONAL PROGRAMME ON CHEMICAL SAFETY
Health and Safety Guide No. 52
DIQUAT
HEALTH AND SAFETY GUIDE
UNITED NATIONS ENVIRONMENT PROGRAMME
INTERNATIONAL LABOUR ORGANISATION
WORLD HEALTH ORGANIZATION
WORLD HEALTH ORGANIZATION, GENEVA 1991
This is a companion volume to Environmental Health Criteria 39:
Paraquat and Diquat
Published by the World Health Organization for the International
Programme on Chemical Safety (a collaborative programme of the United
Nations Environment Programme, the International Labour Organisation,
and the World Health Organization)
This report contains the collective views of an international group of
experts and does not necessarily represent the decisions or the stated
policy of the United Nations Environment Programme, the International
Labour Organisation, or the World Health Organization
WHO Library Cataloguing in Publication Data
Diquat : health and safety guide.
(Health and safety guide ; no. 52)
1. Diquat - standards I. Series
ISBN 92 4 151052 8 (NLM Classification: WA 240)
ISSN 0259-7268
(c) World Health Organization 1991
Publications of the World Health Organization enjoy copyright
protection in accordance with the provisions of Protocol 2 of the
Universal Copyright Convention. For rights of reproduction or
translation of WHO publications, in part or in toto, application
should be made to the Office of Publications, World Health
Organization, Geneva, Switzerland. The World Health Organization
welcomes such applications.
The designations employed and the presentation of the material in this
publication do not imply the expression of any opinion whatsoever on
the part of the Secretariat of the World Health Organization
concerning the legal status of any country, territory, city or area or
of its authorities, or concerning the delimitation of its frontiers or
boundaries.
The mention of specific companies or of certain manufacturers'
products does not imply that they are endorsed or recommended by the
World Health Organization in preference to others of a similar nature
that are not mentioned. Errors and omissions excepted, the names of
proprietary products are distinguished by initial capital letters.
CONTENTS
INTRODUCTION
1. PRODUCT IDENTITY AND USES
1.1. Identity
1.2. Physical and chemical properties
1.3. Analytical methods
1.4. Uses
2. SUMMARY AND EVALUATION
2.1. Environmental distribution and transformation
2.2. Kinetics and metabolism
2.3. Effects of experimental animals
2.4. Effects on human beings
2.4.1. Occupational exposure
2.4.1. General population exposure and cases of poisoning
2.5. Effects on the environment
3. CONCLUSIONS AND RECOMMENDATIONS
3.1. Conclusions
3.1.1. General population exposure
3.1.2. Occupational exposure
3.1.3. Environment
3.2. Recommendations
3.2.1. Environment
3.2.2. Prevention and treatment
4. HUMAN HEALTH HAZARDS, PREVENTION AND PROTECTION, EMERGENCY ACTION
4.1. Main human health hazards, prevention and protection,
first aid
4.1.1. Advice to physicians
4.1.2. Health surveillance advice
4.1.3. Personal protection and hygienic measures
4.2. Explosion and fire hazards
4.3. Storage
4.4. Transport
4.4.1. Accident procedures
4.5. Spillage and disposal
4.5.1. Spillage
4.5.2. Disposal
5. HAZARDS FOR THE ENVIRONMENT AND THEIR PREVENTION
6. SUMMARY OF CHEMICAL SAFETY INFORMATION
7. CURRENT REGULATIONS, GUIDELINES, AND STANDARDS
7.1. Previous evaluations by international bodies
7.2. Exposure limit values
7.3. Specific restrictions
7.4. Labelling, packaging, and transport
7.5. Waste disposal
BIBLIOGRAPHY
INTRODUCTION
The Environmental Health Criteria (EHC) documents produced by the
International Programme on Chemical Safety include an assessment of
the effects on the environment and on human health of exposure to a
chemical or combination of chemicals, or physical or biological
agents. They also provide guidelines for setting exposure limits.
The purpose of a Health and Safety Guide is to facilitate the
application of these guidelines in national chemical safety
programmes. The first three sections of a Health and Safety Guide
highlight the relevant technical information in the corresponding EHC.
Section 4 includes advice on preventive and protective measures and
emergency action; health workers should be thoroughly familiar with
the medical information to ensure that they can act efficiently in an
emergency. Within the Guide is a Summary of Chemical Safety
Information which should be readily available, and should be clearly
explained, to all who could come into contact with the chemical. The
section on regulatory information has been extracted from the legal
file of the International Register of Potentially Toxic Chemicals
(IRPTC) and from other United Nations sources.
The target readership includes occupational health services, those in
ministries, governmental agencies, industry, and trade unions who are
involved in the safe use of chemicals and the avoidance of
environmental health hazards, and those wanting more information on
this topic. An attempt has been made to use only terms that will be
familiar to the intended user. However, sections 1 and 2 inevitably
contain some technical terms. A bibliography has been included for
readers who require further background information.
Revision of the information in this Guide will take place in due
course, and the eventual aim is to use standardized terminology.
Comments on any difficulties encountered in using the Guide would be
very helpful and should be addressed to:
The Manager
International Programme on Chemical Safety
Division of Environmental Health
World Health Organization
1211 Geneva 27
Switzerland
THE INFORMATION IN THIS GUIDE SHOULD BE CONSIDERED AS A STARTING POINT
TO A COMPREHENSIVE HEALTH AND SAFETY PROGRAMME
1. PRODUCT IDENTITY AND USES
1.1 Identity
Common name: Diquat
Molecular formula: C12H12N2Br2
1,1'-ethylene-2,2'-bipyridylium
dibromide
Chemical structure:
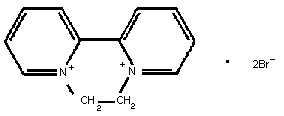 CAS chemical name: 6,7-dihydrodipyrido[1,2-a:2'1'-c]-
pyrazinediium (9CI)
Trade names (inter alia): Reglone; Aquacide; Pathclear
CAS registry number: 2764-72-9 (ion)
85-00-7 (dibromide)
4032-26-2 (dichloride)
RTECS registry number: JM5685000 (ion)
JM5690000 (dibromide)
JM5750000 (dichloride)
JM5755000 (diodide)
Relative molecular mass: 184.2 (ion)
1.2 Physical and Chemical Properties
The commonly available analytical standard is diquat dibromide
monohydrate, which is an odourless, pale yellow, crystalline powder.
Some of the physical properties of diquat dibromide are listed in the
Summary of Chemical Safety Information (section 6).
The herbicide is supplied mainly as an aqueous solution of the
dibromide.
It is slightly soluble in alcohol, and practically insoluble in
non-polar organic solvents. Diquat is non-explosive and non-flammable
in aqueous formulations.
Diquat is stable in neutral or acid solutions, but is hydrolysed by
alkali. It is inactivated by inert clay and by anionic surfactants.
1.3 Analytical Methods
Product analysis is by ultraviolet (UV) spectroscopy and residue
analysis, by colorimetry. Impurities can be determined by gas-liquid
chromatography (GLC).
1.4 Uses
Diquat is a non-selective contact herbicide that is used for: the
control of both broad-leaved weeds among crops, and submerged and
floating weeds in water bodies; the destruction of potato haulms; and
seed crop desiccation (rice, sunflower, etc.).
It is sold primarily as a 20% w/v solution of the dibromide salt, and
is usually formulated to contain wetters.
2. SUMMARY AND EVALUATION
2.1 Environmental Distribution and Transformation
Photochemical degradation takes place when diquat-treated plants are
exposed to normal daylight and continues after the plants have died.
The products formed are of lower toxicity than diquat. The rapidity
of photochemical degradation on plant and soil surfaces minimizes the
hazards of diquat for the environment.
(a) Soil
Diquat rapidly becomes tightly bound to clay particles in the soil,
and is then inert. Toxic breakdown products are not expected to occur
in the soil under normal conditions of agricultural use. Total diquat
residues in the soil, after repeated spraying, ranged from 0.2 to
3.9 mg/kg. In field studies, on the 15th day after a single
application of diquat, residues were less than 0.1 mg/kg. No adverse
effects were found on soil microorganisms, fungi, or invertebrates,
and no phytotoxic effects on crops have been reported from these bound
residues.
(b) Water
Following its use as an aquatic herbicide at normal application rates,
diquat residues in water have been found to decrease rapidly to
essentially undetectable levels within 7-14 days.
(c) Air
Diquat is not volatile. However, during spraying applications,
droplets of diquat solution may be present in the air, as an aerosol.
2.2 Kinetics and Metabolism
Diquat is poorly absorbed from the intestinal tract and skin. Diquat
monopyridone is the major metabolite of diquat in the body; of lesser
importance is diquat dipyridone. Both metabolites are considerably
less toxic than diquat itself. Less than 20% of an oral dose is
metabolized. Diquat is primarily metabolized in the gastrointestinal
tract.
Diquat accumulates in the kidneys, but not in the lungs. The kidneys
are the major route of excretion, but a considerable amount of diquat
can also be excreted in the bile, varying according to the animal
species.
2.3 Effects on Experimental Animals
Diquat is less toxic than paraquat and does not cause the specific
lung lesion associated with paraquat poisoning. The toxic effects of
diquat depend on its ability to undergo a single electron addition, to
form a radical. This reacts with molecular oxygen to reform diquat, at
the same time producing a superoxide anion. This oxygen radical may
directly or indirectly cause cell death.
The initial toxic effect of diquat in animals is gastrointestinal
damage, resulting in diarrhoea, with consequent dehydration. High
doses of diquat have produced toxic effects in the liver, kidney, and
the nervous and endocrine systems. High concentrations are also
irritating to the skin. Development of eye cataracts has been
reported in rats and dogs following long-term treatment with diquat.
This observation has not been reported in human beings. Diquat is
embryotoxic, but it has not been found to be teratogenic in rats and
mice. Carcinogenic effects were not induced in long-term studies on
rats given diquat at levels of up to 720 mg/kg diet. The results of
in vitro mutagenicity studies have been inconclusive, but have
generally suggested weak activity; the results of in vivo studies have
been negative. Thus, on the basis of animal studies, low-level
exposure to diquat is unlikely to induce toxic effects in man. The
no-observed-effect level in rats has been estimated to be 0.75 mg
diquat ion/kg body weight per day.
2.4 Effects on Human Beings
2.4.1 Occupational exposure
There may be inhalation, dermal, and, to some extent, oral
occupational exposure. Spray aerosols and dust particles settle in
the upper respiratory tract. Diquat aerosol concentrations ranged
from 0.06 to 0.56 mg/m3, according to the spraying method. At
distances of 200 and 400 m from the treated field, they decreased to
0.09 mg/m3 and less than 0.01 mg/m3, respectively. Inhalation
exposure to diquat was found to be very low in comparison with dermal
(0.17-1.82 mg/h) exposure, during application for aquatic weed
control. Skin irritation, epistaxis, nail damage, and delayed wound
healing have been reported. However, no data have been found in the
literature on severe or fatal cases of occupational intoxication,
acute ocular damage, or occupational contact dermatitis caused by
diquat.
2.4.2 General population exposure and cases of poisoning
Inhalation exposure of the general population to diquat may occur from
spray drift off treated fields, but this is thought to be
insignificant. There are no published data on total diquat intake
among the general population but, on the basis of residue data, this
is expected to be insignificant.
Few cases of diquat poisoning have been reported. Most have been due
to the intentional ingestion of concentrated formulations, but
accidental ingestion has occurred, for example, as a result of
decanting liquid concentrates into a beverage bottle. Diquat
poisoning involves severe ill-effects similar to those caused by
paraquat, with two notable differences: (a) diarrhoea is a prominent
feature, and (b) pulmonary fibrosis has not been described.
The acute lethal dose of diquat dibromide is considered to be 6-12 g
for human beings. Recovery from diquat poisoning depends on the cause
of ingestion, the dose absorbed, the renal damage, and prompt
initiation of therapy. No long-term adverse effects have been
reported in those who have survived acute diquat poisoning.
2.5 Effects on the Environment
The fact that diquat is used as a herbicide indicates that it is toxic
for aquatic and terrestrial vegetation.
Diquat undergoes rapid photochemical degradation in aqueous solution
and on surfaces. The major degradation products produced have been
identified and are of lower acute oral toxicity for rats than diquat
itself.
Diquat reaching the soil becomes rapidly and strongly adsorbed on clay
minerals in the soil. This process inactivates the herbicidal
activity of diquat. Strongly-bound diquat has no adverse effects on
soil microfauna or soil microbial processes.
Diquat residues disappear rapidly from water by adsorption on aquatic
weeds and by strong adsorption on bottom mud. The toxicity of diquat
for fish is low and it does not accumulate in them. Normal
applications of diquat for aquatic weed control are not harmful for
aquatic animal life. However, in applying diquat to water containing
heavy weed growth, care should be taken to treat only a part of the
weed growth, since oxygen consumed by subsequent weed decay may
decrease dissolved oxygen levels to an extent that may be dangerous
for fish. Treated water should not be used for overhead irrigation
until a period of 10 days has elapsed following treatment.
3. CONCLUSIONS AND RECOMMENDATIONS
3.1 Conclusions
3.1.1 General population exposure
Residue levels of diquat in food and drinking-water, resulting from
its normal use, are unlikely to present a health hazard for the
general population.
Diquat has caused some fatalities following suicidal ingestion.
Occasional accidental fatalities have followed ingestion of decanted
diquat. Ill effects similar to those caused by paraquat occur, but
the characteristic fibrosis of the lungs is not a feature.
3.1.2 Occupational exposure
With reasonable work practices, including safety precautions, hygiene
measures, and proper supervision, occupational exposure during the
manufacture, formulation, and application of diquat will not cause a
hazard. However, the undiluted concentrate must be handled with great
care, because contamination of the eyes and skin (with possible
consequent dermal absorption) can result from improper work practices.
3.1.3 Environment
Diquat in soil binds rapidly and tightly to clay particles, and
residual phytotoxicity from freely available diquat is unlikely.
Under normal conditions of use, the toxicity of diquat for aquatic
animal life is low, though resulting depletion of water oxygen, due to
weed decay, may pose a problem. Diquat does not seem to present an
environmental hazard.
3.2 Recommendations
3.2.1 General
Where practical and reasonable, the availability and use of the 20%
liquid product should be limited to bona fide agriculturalists,
horticulturalists, and professional users, who work with trained
personnel, properly maintained equipment, and adequate supervision.
Every effort should be made to prevent the practice of decanting, or
rebottling, of the product into containers that have not been properly
labelled.
3.2.2 Prevention and treatment
Attention should be drawn to the fact that persons with skin lesions
(either pre-existing or following contamination with diquat) should
not be permitted to take any part in spraying procedures, until the
skin condition has resolved.
It must be stressed that treatment of persons with diquat poisoning
should be instituted as early as possible. The likelihood of recovery
from a potentially fatal dose is greatest when therapy begins within
5-6 h of poisoning.
4. HUMAN HEALTH HAZARDS, PREVENTION AND PROTECTION, EMERGENCY ACTION
4.1 Main Human Health Hazards, Prevention and Protection, First Aid
Diquat is toxic and can be fatal, if swallowed. Contact with
undiluted liquid products can cause damage to the skin or eyes, and
the utmost care must be taken to avoid exposure during handling
operations and application in the field. Where inhalation exposure to
aerosols containing diquat is likely, proper respiratory protection
equipment should be used.
The human health hazards, preventive and protective measures, and
first-aid recommendations are listed in the Summary of Chemical Safety
Information (section 6).
4.1.1 Advice to physicians
The most important measures are the immediate neutralization of
ingested diquat by 15% Fuller's earth, bentonite, or activated
charcoal, and urgent removal of the poison by vomiting or, when
possible, gastric lavage. The urgency of these measures is such that,
where transfer to hospital may involve a delay of an hour or more,
this emergency treatment may have to be given by a paramedical person,
e.g., a nurse or a medical assistant. The delay should not be more
than 4-5 h. Furthermore, Fuller's earth should be given together with
a strong purgative, such as magnesium sulfate or mannitol.
Admission to a hospital (preferably a specialized intensive care unit)
either directly, or after emergency treatment elsewhere, is essential.
When a person has swallowed a lethal dose, the most important single
determinant of survival is the early commencement of treatment.
Depending on local facilities, patients who reach hospital after the
initial treatment will have further treatment aimed at neutralizing
diquat in the gastrointestinal tract (Fuller's earth, bentonite,
activated charcoal) or its excretion in the faeces (purgatives, 10%
mannitol, gut lavage). In addition, attempts to remove absorbed
diquat from the circulation (haemoperfusion, haemodialysis) or aid its
excretion by the kidney (forced diuresis) can be instituted.
Care must be exercised in the administration of most of these
treatments, as the following serious complications may occur:
perforation of the oesophagus during gastric intubation; serious blood
chemistry disturbance when severe diarrhoea is induced; fluid overload
during forced diuresis.
In centres where facilities for analytical procedures are available,
measurement of urinary or, ideally, plasma levels of diquat may give
guidelines for the required intensity of treatment, and likely
prognosis.
It should be noted that, as with the great majority of chemicals,
there is no specific antidote.
In cases of skin and eye contamination, irrigation with water
(preferably running-water) should be commenced urgently and must be
continued uninterrupted for at least 15 minutes (timed by the clock).
Eye cases should always be taken for medical treatment. In cases of
skin contamination by the concentrate, or extensive and/or prolonged
contamination by the diluted material (particularly where signs of
skin irritation are present), the patient must be assessed at hospital
for systemic poisoning.
4.1.2 Health surveillance advice
Pre-employment and annual medical examinations should be made
available to all persons who are regularly exposed to diquat at
potentially hazardous levels. Special attention should be paid to the
condition of the skin, kidney function, urinalysis, and liver function
tests.
4.1.3 Personal protection and hygienic measures
* Avoid all contact with the skin, eyes, nose, and mouth, when
handling concentrated diquat. Wear PVC, neoprene, or butyl
rubber gloves (preferably gauntlet form), neoprene apron, rubber
boots, and face-shield;
* Take off heavily contaminated clothing immediately, and wash
underlying skin;
* Wash clothes before re-use;
* When using diquat, do not eat, drink, or smoke;
* Wash concentrate from skin or eyes, immediately;
* Do not inhale spray;
* Wash hands and exposed skin before meals and after work;
* Keep away from food, drink, and animal feed;
* Diquat for spraying should be adequately diluted;
* It should not be used by people suffering from dermatitis, or by
people with wounds, notably on the hands, until these have
healed.
4.2 Explosion and Fire Hazards
Diquat products are generally not flammable. If involved in a fire,
control with dry powder or alcohol-resistant foam. Advise the fire
service that protective clothing and self-contained breathing
apparatus should be worn, to avoid skin contamination and the
breathing of toxic fumes. Confine the use of water spray to the
cooling of unaffected stock, thus avoiding the accumulation of
polluted run-off from the site.
4.3 Storage
Store technical material and formulations away from heat, under lock
and key, and out of reach of children, animals, and unauthorized
personnel. Store in an area designated for pesticide storage,
preferably without drains. Store away from foodstuffs and animal
feed.
4.4 Transport
Before dispatch, ensure that containers are sound and that labels are
securely fixed and undamaged. Comply with local transport
regulations. Do not load together with foodstuffs or animal feed.
4.4.1 Accident procedures
Avoid exposure by the use of appropriate protective clothing, gloves,
and goggles or masks. Keep spectators away from leaking or spilled
product, and prevent smoking and the use of naked flames in the
immediate vicinity.
Extinguish fires with dry powder, carbon dioxide, alcohol-resistant
foam, sand, or earth.
Prevent liquid from spreading to other cargo, vegetation, or waterways
by containing it with the most readily available barrier material,
e.g., earth or sand.
Absorb spilled liquid, and cover contaminated areas with earth, lime,
sand, or other absorbent material; place in a secure container for
subsequent safe disposal.
4.5 Spillage and Disposal
4.5.1 Spillage
Avoid exposure by using appropriate protective clothing and
face-shields.
Empty any product remaining in damaged or leaking containers into a
clean empty drum, and label.
Absorb spillage with lime, damp sawdust, sand, or earth and dispose of
safely (see below). If spillage is large, contain it by building a
barrier of earth or sandbags.
Decontaminate empty, damaged, or leaking containers with a 10% sodium
carbonate solution, added at the rate of at least 1 litre per 20-litre
drum. Puncture or crush containers to prevent re-use.
4.5.2 Disposal
Waste containing diquat should be burnt in a proper high temperature
incinerator with effluent scrubbing.
Where no incinerator is available, contaminated absorbents or surplus
products should be decomposed by hydrolysis at pH 12 or above. A 5%
sodium hydroxide (caustic soda) solution, or saturated (7-10%) sodium
carbonate (washing soda) solution, can be used. Before disposal of
the resultant waste, the material must be analysed to ensure that the
active ingredient has been degraded to a safe level.
Diquat is rapidly inactivated by clay soil. If the above mentioned
methods are not possible, it can be buried in an approved landfill.
Never pour untreated waste or surplus products into public sewers, or
where there is any danger of run-off or seepage into streams,
water-courses, open waterways, ditches, fields with drainage systems,
or the catchment areas of boreholes, wells, springs, or ponds.
5. HAZARDS FOR THE ENVIRONMENT AND THEIR PREVENTION
Diquat is toxic for aquatic and terrestrial vegetation.
Avoid spraying over bodies of water. Do not contaminate ponds,
waterways, or ditches with product or used containers.
When using diquat for aquatic weed control, follow instructions on the
label.
6. SUMMARY OF CHEMICAL SAFETY INFORMATION
This summary should be easily available to all health workers
concerned with, and users of, diquat. It should be displayed at, or
near, entrances to areas where there is potential exposure to diquat,
and on processing equipment and containers. The summary should be
translated into the appropriate language(s). All persons potentially
exposed to the chemical should also have the instructions in the
summary clearly explained.
Space is available for insertion of the National Occupational
Exposure Limit, the address and telephone number of the National
Poison Control Centre, and for local trade names.
DIQUAT
C12H12N2Br2, 1,1'-ethylene-2,2'-bipyridylium dibromide
CAS registry no. 85-00-7
RTECS registry no. JM5690000
PHYSICAL PROPERTIES OTHER CHARACTERISTICS
Physical state crystalline powder A non-selective contact herbicide and desiccant
Colour yellow for terrestrial and aquatic applications; slightly
Odour odourless soluble in alcohol and practically insoluble
Relative molecular mass 184.2 (ion) in non-polar organic solvents; stable in neutral
Specific gravity (20°C) 1.200 and acidic solutions, but hydrolysed by alkali;
Melting point 180°C inactivated by inert clay and by anionic surfactants
Boiling point approximately 300°C
with decomposition
Solubility in water (20°C) 700 g/litre
pH of liquid formulation 6.0-7.0
Vapour pressure not measurable
HAZARDS/SYMPTOMS PREVENTION AND PROTECTION FIRST AID
SKIN: Irritating to skin; Proper application technique, proper Remove contaminated clothing; wash skin with
concentrated material may cause skin protection, including impervious soap and water; launder clothes before re-use
blisters, re-use and affect nails clothing and gloves
EYES: Severe irritant Wear face-shield or goggles Flush immediately with clean water for at least
15 minutes; seek medical advice and observe for
delayed effects
INHALATION: Irritant to Avoid inhalation of fine dust Fresh air
respiratory system and mist; use proper
respiratory protection
INGESTION: Unlikely Do not eat, drink, or smoke
occupational hazard during work; wash hands
Accidental or deliberate ingestion Obtain medical attention immediately; transport
may cause vomiting, abdominal pain, to hospital urgently; induce vomiting;
and diarrhoea, followed by burns in do not delay
mouth and throat; kidney damage and
shock may occur, but no lung damage
SPILLAGE STORAGE FIRE AND EXPLOSION
Absorb spillage with lime, damp Store in locked, well-ventilated Non-flammable and non-explosive
sawdust, sand, or earth; sweep up, storeroom, away from foodstuffs
place in closed container, and and animal feed, children,
dispose of safely; avoid and unauthorized personnel
contamination of personnel,
ponds, and waterways
WASTE DISPOSAL
Burn in high-temperature National Occupational Exposure UN No. 2782, 3015, 3016
incinerator with effluent scrubbing; Limit:
alternatively, treat with 5%
caustic soda as a hydrolysing agent; National Poison Control Centre:
comply with local regulations
Local trade names:
7. CURRENT REGULATIONS, GUIDELINES, AND STANDARDS
The information given in this section has been extracted from the
International Register of Potentially Toxic Chemicals (IRPTC) legal
file. A full reference to the original national document from which
the information was extracted can be obtained from IRPTC. When no
effective date appears in the IRPTC legal file, the year of the
reference from which the data are taken is indicated by (r).
The reader should be aware that regulatory decisions about chemicals,
taken in a certain country, can only be fully understood in the
framework of the legislation of that country. The regulations and
guidelines of all countries are subject to change and should always be
verified with appropriate regulatory authorities before application.
7.1 Previous Evaluations by International Bodies
The Joint FAO/WHO Meeting on Pesticide Residues (JMPR) reviewed
residues and toxicity data on diquat in 1970, 1972, 1976, 1977, and
1978. In 1977, it estimated that the acceptable daily intake (ADI)
for man was 0-0.008 mg/kg body weight, expressed as diquat ion.
The JMPR has also recommended maximum residue levels (tolerances) for
diquat in food commodities of plant and animal origin.
A data sheet on diquat has been prepared by WHO/FAO (1979) in a series
of "Data sheets on chemical pesticides" (No. 44). Technical diquat is
qualified as moderately hazardous in normal use (WHO, 1990).
7.2 Exposure Limit Values
Some exposure limit values are given in the table on pages 26-27.
7.3 Specific Restrictions
In some countries, diquat is only registered for certain specified
applications, or for use under certain specified conditions, e.g., in
the Federal Republic of Germany, Sweden, the United Kingdom, the USA,
and the USSR. For example, in the United Kingdom and the USA, the use
of the 20% liquid is restricted to bona fide certified professionals.
In the Federal Republic of Germany, diquat may not be handled by
adolescents and pregnant or nursing women.
EXPOSURE LIMIT VALUES
Medium Specification Country/ Exposure limit descriptiona Value Effective
organization date
AIR Work-place Australia Threshold limit value (TLV)
- Time-weighted average (TWA) 0.5 mg/m3 1983
- Short-term exposure limit (STEL) 1 mg/m3
Belgium Tolerable limit value (TLV)
- Time-weighted average (TWA) 0.5 mg/m3 1988
- Short-term exposure level (STEL) 1 mg/m3
Bulgaria Maximum permissible concentration
- Time-weighted average (TWA) 0.5 mg/m3 1987
Finland Maximum permissible concentration
- Time-weighted average (TWA) 0.5 mg/m3 1988
- Short-term exposure limit (STEL) 1.5 mg/m3
Netherlands Maximum permissible limit
- Time-weighted average (TWA) 0.5 mg/m3 1986
Switzerland Maximum work-site concentration (MAK)
- Time-weighted average (TWA) 0.5 mg/m3 1987
United Kingdom Recommended limit (RECL)
- 8-h time-weighted average (TWA) 0.5 mg/m3 1987
- Short-term exposure level (STEL) 1 mg/m3
(10-min time-weighted average)
USA (OSHA) Permissible exposure limit (PEL)
- Time-weighted average (TWA) 0.5 mg/m3 1989
USA (ACGIH) Threshold limit value (TLV)
- Time-weighted average (TWA) 0.5 mg/m3 1987
Medium Specification Country/ Exposure limit descriptiona Value Effective
organization date
USSR Preliminary safety level (vapour
and aerosol)
- Ceiling value (CLV) 0.2 mg/m3 1978
FOOD Intake from FAO/WHO Acceptable daily intake (ADI) 0-0.008 mg/kg 1977
(diquat ion) body weight
FOOD General FAO/WHO Maximum residue limit (MRL) 0.01-5 mg/kg 1982
for specified products
(diquat ion)
European Maximum levels in fruit and
Community vegetables (expressed as diquat
cation)
- vegetables 0.1 mg/kg 1989
- other products 0.05 mg/kg 1989
a TWA=time-weighted average over one working day (usually 8h).
7.4 Labelling, Packaging, and Transport
The United Nations Committee of Experts on the Transportation of
Dangerous Goods classifies diquat (at liquid concentrations of
45-100%) in:
- Hazard Class 6.1: poisonous substance;
- Packing Group III: a substance presenting a relatively low
risk of poisoning in transport.
The label should be as follows:
CAS chemical name: 6,7-dihydrodipyrido[1,2-a:2'1'-c]-
pyrazinediium (9CI)
Trade names (inter alia): Reglone; Aquacide; Pathclear
CAS registry number: 2764-72-9 (ion)
85-00-7 (dibromide)
4032-26-2 (dichloride)
RTECS registry number: JM5685000 (ion)
JM5690000 (dibromide)
JM5750000 (dichloride)
JM5755000 (diodide)
Relative molecular mass: 184.2 (ion)
1.2 Physical and Chemical Properties
The commonly available analytical standard is diquat dibromide
monohydrate, which is an odourless, pale yellow, crystalline powder.
Some of the physical properties of diquat dibromide are listed in the
Summary of Chemical Safety Information (section 6).
The herbicide is supplied mainly as an aqueous solution of the
dibromide.
It is slightly soluble in alcohol, and practically insoluble in
non-polar organic solvents. Diquat is non-explosive and non-flammable
in aqueous formulations.
Diquat is stable in neutral or acid solutions, but is hydrolysed by
alkali. It is inactivated by inert clay and by anionic surfactants.
1.3 Analytical Methods
Product analysis is by ultraviolet (UV) spectroscopy and residue
analysis, by colorimetry. Impurities can be determined by gas-liquid
chromatography (GLC).
1.4 Uses
Diquat is a non-selective contact herbicide that is used for: the
control of both broad-leaved weeds among crops, and submerged and
floating weeds in water bodies; the destruction of potato haulms; and
seed crop desiccation (rice, sunflower, etc.).
It is sold primarily as a 20% w/v solution of the dibromide salt, and
is usually formulated to contain wetters.
2. SUMMARY AND EVALUATION
2.1 Environmental Distribution and Transformation
Photochemical degradation takes place when diquat-treated plants are
exposed to normal daylight and continues after the plants have died.
The products formed are of lower toxicity than diquat. The rapidity
of photochemical degradation on plant and soil surfaces minimizes the
hazards of diquat for the environment.
(a) Soil
Diquat rapidly becomes tightly bound to clay particles in the soil,
and is then inert. Toxic breakdown products are not expected to occur
in the soil under normal conditions of agricultural use. Total diquat
residues in the soil, after repeated spraying, ranged from 0.2 to
3.9 mg/kg. In field studies, on the 15th day after a single
application of diquat, residues were less than 0.1 mg/kg. No adverse
effects were found on soil microorganisms, fungi, or invertebrates,
and no phytotoxic effects on crops have been reported from these bound
residues.
(b) Water
Following its use as an aquatic herbicide at normal application rates,
diquat residues in water have been found to decrease rapidly to
essentially undetectable levels within 7-14 days.
(c) Air
Diquat is not volatile. However, during spraying applications,
droplets of diquat solution may be present in the air, as an aerosol.
2.2 Kinetics and Metabolism
Diquat is poorly absorbed from the intestinal tract and skin. Diquat
monopyridone is the major metabolite of diquat in the body; of lesser
importance is diquat dipyridone. Both metabolites are considerably
less toxic than diquat itself. Less than 20% of an oral dose is
metabolized. Diquat is primarily metabolized in the gastrointestinal
tract.
Diquat accumulates in the kidneys, but not in the lungs. The kidneys
are the major route of excretion, but a considerable amount of diquat
can also be excreted in the bile, varying according to the animal
species.
2.3 Effects on Experimental Animals
Diquat is less toxic than paraquat and does not cause the specific
lung lesion associated with paraquat poisoning. The toxic effects of
diquat depend on its ability to undergo a single electron addition, to
form a radical. This reacts with molecular oxygen to reform diquat, at
the same time producing a superoxide anion. This oxygen radical may
directly or indirectly cause cell death.
The initial toxic effect of diquat in animals is gastrointestinal
damage, resulting in diarrhoea, with consequent dehydration. High
doses of diquat have produced toxic effects in the liver, kidney, and
the nervous and endocrine systems. High concentrations are also
irritating to the skin. Development of eye cataracts has been
reported in rats and dogs following long-term treatment with diquat.
This observation has not been reported in human beings. Diquat is
embryotoxic, but it has not been found to be teratogenic in rats and
mice. Carcinogenic effects were not induced in long-term studies on
rats given diquat at levels of up to 720 mg/kg diet. The results of
in vitro mutagenicity studies have been inconclusive, but have
generally suggested weak activity; the results of in vivo studies have
been negative. Thus, on the basis of animal studies, low-level
exposure to diquat is unlikely to induce toxic effects in man. The
no-observed-effect level in rats has been estimated to be 0.75 mg
diquat ion/kg body weight per day.
2.4 Effects on Human Beings
2.4.1 Occupational exposure
There may be inhalation, dermal, and, to some extent, oral
occupational exposure. Spray aerosols and dust particles settle in
the upper respiratory tract. Diquat aerosol concentrations ranged
from 0.06 to 0.56 mg/m3, according to the spraying method. At
distances of 200 and 400 m from the treated field, they decreased to
0.09 mg/m3 and less than 0.01 mg/m3, respectively. Inhalation
exposure to diquat was found to be very low in comparison with dermal
(0.17-1.82 mg/h) exposure, during application for aquatic weed
control. Skin irritation, epistaxis, nail damage, and delayed wound
healing have been reported. However, no data have been found in the
literature on severe or fatal cases of occupational intoxication,
acute ocular damage, or occupational contact dermatitis caused by
diquat.
2.4.2 General population exposure and cases of poisoning
Inhalation exposure of the general population to diquat may occur from
spray drift off treated fields, but this is thought to be
insignificant. There are no published data on total diquat intake
among the general population but, on the basis of residue data, this
is expected to be insignificant.
Few cases of diquat poisoning have been reported. Most have been due
to the intentional ingestion of concentrated formulations, but
accidental ingestion has occurred, for example, as a result of
decanting liquid concentrates into a beverage bottle. Diquat
poisoning involves severe ill-effects similar to those caused by
paraquat, with two notable differences: (a) diarrhoea is a prominent
feature, and (b) pulmonary fibrosis has not been described.
The acute lethal dose of diquat dibromide is considered to be 6-12 g
for human beings. Recovery from diquat poisoning depends on the cause
of ingestion, the dose absorbed, the renal damage, and prompt
initiation of therapy. No long-term adverse effects have been
reported in those who have survived acute diquat poisoning.
2.5 Effects on the Environment
The fact that diquat is used as a herbicide indicates that it is toxic
for aquatic and terrestrial vegetation.
Diquat undergoes rapid photochemical degradation in aqueous solution
and on surfaces. The major degradation products produced have been
identified and are of lower acute oral toxicity for rats than diquat
itself.
Diquat reaching the soil becomes rapidly and strongly adsorbed on clay
minerals in the soil. This process inactivates the herbicidal
activity of diquat. Strongly-bound diquat has no adverse effects on
soil microfauna or soil microbial processes.
Diquat residues disappear rapidly from water by adsorption on aquatic
weeds and by strong adsorption on bottom mud. The toxicity of diquat
for fish is low and it does not accumulate in them. Normal
applications of diquat for aquatic weed control are not harmful for
aquatic animal life. However, in applying diquat to water containing
heavy weed growth, care should be taken to treat only a part of the
weed growth, since oxygen consumed by subsequent weed decay may
decrease dissolved oxygen levels to an extent that may be dangerous
for fish. Treated water should not be used for overhead irrigation
until a period of 10 days has elapsed following treatment.
3. CONCLUSIONS AND RECOMMENDATIONS
3.1 Conclusions
3.1.1 General population exposure
Residue levels of diquat in food and drinking-water, resulting from
its normal use, are unlikely to present a health hazard for the
general population.
Diquat has caused some fatalities following suicidal ingestion.
Occasional accidental fatalities have followed ingestion of decanted
diquat. Ill effects similar to those caused by paraquat occur, but
the characteristic fibrosis of the lungs is not a feature.
3.1.2 Occupational exposure
With reasonable work practices, including safety precautions, hygiene
measures, and proper supervision, occupational exposure during the
manufacture, formulation, and application of diquat will not cause a
hazard. However, the undiluted concentrate must be handled with great
care, because contamination of the eyes and skin (with possible
consequent dermal absorption) can result from improper work practices.
3.1.3 Environment
Diquat in soil binds rapidly and tightly to clay particles, and
residual phytotoxicity from freely available diquat is unlikely.
Under normal conditions of use, the toxicity of diquat for aquatic
animal life is low, though resulting depletion of water oxygen, due to
weed decay, may pose a problem. Diquat does not seem to present an
environmental hazard.
3.2 Recommendations
3.2.1 General
Where practical and reasonable, the availability and use of the 20%
liquid product should be limited to bona fide agriculturalists,
horticulturalists, and professional users, who work with trained
personnel, properly maintained equipment, and adequate supervision.
Every effort should be made to prevent the practice of decanting, or
rebottling, of the product into containers that have not been properly
labelled.
3.2.2 Prevention and treatment
Attention should be drawn to the fact that persons with skin lesions
(either pre-existing or following contamination with diquat) should
not be permitted to take any part in spraying procedures, until the
skin condition has resolved.
It must be stressed that treatment of persons with diquat poisoning
should be instituted as early as possible. The likelihood of recovery
from a potentially fatal dose is greatest when therapy begins within
5-6 h of poisoning.
4. HUMAN HEALTH HAZARDS, PREVENTION AND PROTECTION, EMERGENCY ACTION
4.1 Main Human Health Hazards, Prevention and Protection, First Aid
Diquat is toxic and can be fatal, if swallowed. Contact with
undiluted liquid products can cause damage to the skin or eyes, and
the utmost care must be taken to avoid exposure during handling
operations and application in the field. Where inhalation exposure to
aerosols containing diquat is likely, proper respiratory protection
equipment should be used.
The human health hazards, preventive and protective measures, and
first-aid recommendations are listed in the Summary of Chemical Safety
Information (section 6).
4.1.1 Advice to physicians
The most important measures are the immediate neutralization of
ingested diquat by 15% Fuller's earth, bentonite, or activated
charcoal, and urgent removal of the poison by vomiting or, when
possible, gastric lavage. The urgency of these measures is such that,
where transfer to hospital may involve a delay of an hour or more,
this emergency treatment may have to be given by a paramedical person,
e.g., a nurse or a medical assistant. The delay should not be more
than 4-5 h. Furthermore, Fuller's earth should be given together with
a strong purgative, such as magnesium sulfate or mannitol.
Admission to a hospital (preferably a specialized intensive care unit)
either directly, or after emergency treatment elsewhere, is essential.
When a person has swallowed a lethal dose, the most important single
determinant of survival is the early commencement of treatment.
Depending on local facilities, patients who reach hospital after the
initial treatment will have further treatment aimed at neutralizing
diquat in the gastrointestinal tract (Fuller's earth, bentonite,
activated charcoal) or its excretion in the faeces (purgatives, 10%
mannitol, gut lavage). In addition, attempts to remove absorbed
diquat from the circulation (haemoperfusion, haemodialysis) or aid its
excretion by the kidney (forced diuresis) can be instituted.
Care must be exercised in the administration of most of these
treatments, as the following serious complications may occur:
perforation of the oesophagus during gastric intubation; serious blood
chemistry disturbance when severe diarrhoea is induced; fluid overload
during forced diuresis.
In centres where facilities for analytical procedures are available,
measurement of urinary or, ideally, plasma levels of diquat may give
guidelines for the required intensity of treatment, and likely
prognosis.
It should be noted that, as with the great majority of chemicals,
there is no specific antidote.
In cases of skin and eye contamination, irrigation with water
(preferably running-water) should be commenced urgently and must be
continued uninterrupted for at least 15 minutes (timed by the clock).
Eye cases should always be taken for medical treatment. In cases of
skin contamination by the concentrate, or extensive and/or prolonged
contamination by the diluted material (particularly where signs of
skin irritation are present), the patient must be assessed at hospital
for systemic poisoning.
4.1.2 Health surveillance advice
Pre-employment and annual medical examinations should be made
available to all persons who are regularly exposed to diquat at
potentially hazardous levels. Special attention should be paid to the
condition of the skin, kidney function, urinalysis, and liver function
tests.
4.1.3 Personal protection and hygienic measures
* Avoid all contact with the skin, eyes, nose, and mouth, when
handling concentrated diquat. Wear PVC, neoprene, or butyl
rubber gloves (preferably gauntlet form), neoprene apron, rubber
boots, and face-shield;
* Take off heavily contaminated clothing immediately, and wash
underlying skin;
* Wash clothes before re-use;
* When using diquat, do not eat, drink, or smoke;
* Wash concentrate from skin or eyes, immediately;
* Do not inhale spray;
* Wash hands and exposed skin before meals and after work;
* Keep away from food, drink, and animal feed;
* Diquat for spraying should be adequately diluted;
* It should not be used by people suffering from dermatitis, or by
people with wounds, notably on the hands, until these have
healed.
4.2 Explosion and Fire Hazards
Diquat products are generally not flammable. If involved in a fire,
control with dry powder or alcohol-resistant foam. Advise the fire
service that protective clothing and self-contained breathing
apparatus should be worn, to avoid skin contamination and the
breathing of toxic fumes. Confine the use of water spray to the
cooling of unaffected stock, thus avoiding the accumulation of
polluted run-off from the site.
4.3 Storage
Store technical material and formulations away from heat, under lock
and key, and out of reach of children, animals, and unauthorized
personnel. Store in an area designated for pesticide storage,
preferably without drains. Store away from foodstuffs and animal
feed.
4.4 Transport
Before dispatch, ensure that containers are sound and that labels are
securely fixed and undamaged. Comply with local transport
regulations. Do not load together with foodstuffs or animal feed.
4.4.1 Accident procedures
Avoid exposure by the use of appropriate protective clothing, gloves,
and goggles or masks. Keep spectators away from leaking or spilled
product, and prevent smoking and the use of naked flames in the
immediate vicinity.
Extinguish fires with dry powder, carbon dioxide, alcohol-resistant
foam, sand, or earth.
Prevent liquid from spreading to other cargo, vegetation, or waterways
by containing it with the most readily available barrier material,
e.g., earth or sand.
Absorb spilled liquid, and cover contaminated areas with earth, lime,
sand, or other absorbent material; place in a secure container for
subsequent safe disposal.
4.5 Spillage and Disposal
4.5.1 Spillage
Avoid exposure by using appropriate protective clothing and
face-shields.
Empty any product remaining in damaged or leaking containers into a
clean empty drum, and label.
Absorb spillage with lime, damp sawdust, sand, or earth and dispose of
safely (see below). If spillage is large, contain it by building a
barrier of earth or sandbags.
Decontaminate empty, damaged, or leaking containers with a 10% sodium
carbonate solution, added at the rate of at least 1 litre per 20-litre
drum. Puncture or crush containers to prevent re-use.
4.5.2 Disposal
Waste containing diquat should be burnt in a proper high temperature
incinerator with effluent scrubbing.
Where no incinerator is available, contaminated absorbents or surplus
products should be decomposed by hydrolysis at pH 12 or above. A 5%
sodium hydroxide (caustic soda) solution, or saturated (7-10%) sodium
carbonate (washing soda) solution, can be used. Before disposal of
the resultant waste, the material must be analysed to ensure that the
active ingredient has been degraded to a safe level.
Diquat is rapidly inactivated by clay soil. If the above mentioned
methods are not possible, it can be buried in an approved landfill.
Never pour untreated waste or surplus products into public sewers, or
where there is any danger of run-off or seepage into streams,
water-courses, open waterways, ditches, fields with drainage systems,
or the catchment areas of boreholes, wells, springs, or ponds.
5. HAZARDS FOR THE ENVIRONMENT AND THEIR PREVENTION
Diquat is toxic for aquatic and terrestrial vegetation.
Avoid spraying over bodies of water. Do not contaminate ponds,
waterways, or ditches with product or used containers.
When using diquat for aquatic weed control, follow instructions on the
label.
6. SUMMARY OF CHEMICAL SAFETY INFORMATION
This summary should be easily available to all health workers
concerned with, and users of, diquat. It should be displayed at, or
near, entrances to areas where there is potential exposure to diquat,
and on processing equipment and containers. The summary should be
translated into the appropriate language(s). All persons potentially
exposed to the chemical should also have the instructions in the
summary clearly explained.
Space is available for insertion of the National Occupational
Exposure Limit, the address and telephone number of the National
Poison Control Centre, and for local trade names.
DIQUAT
C12H12N2Br2, 1,1'-ethylene-2,2'-bipyridylium dibromide
CAS registry no. 85-00-7
RTECS registry no. JM5690000
PHYSICAL PROPERTIES OTHER CHARACTERISTICS
Physical state crystalline powder A non-selective contact herbicide and desiccant
Colour yellow for terrestrial and aquatic applications; slightly
Odour odourless soluble in alcohol and practically insoluble
Relative molecular mass 184.2 (ion) in non-polar organic solvents; stable in neutral
Specific gravity (20°C) 1.200 and acidic solutions, but hydrolysed by alkali;
Melting point 180°C inactivated by inert clay and by anionic surfactants
Boiling point approximately 300°C
with decomposition
Solubility in water (20°C) 700 g/litre
pH of liquid formulation 6.0-7.0
Vapour pressure not measurable
HAZARDS/SYMPTOMS PREVENTION AND PROTECTION FIRST AID
SKIN: Irritating to skin; Proper application technique, proper Remove contaminated clothing; wash skin with
concentrated material may cause skin protection, including impervious soap and water; launder clothes before re-use
blisters, re-use and affect nails clothing and gloves
EYES: Severe irritant Wear face-shield or goggles Flush immediately with clean water for at least
15 minutes; seek medical advice and observe for
delayed effects
INHALATION: Irritant to Avoid inhalation of fine dust Fresh air
respiratory system and mist; use proper
respiratory protection
INGESTION: Unlikely Do not eat, drink, or smoke
occupational hazard during work; wash hands
Accidental or deliberate ingestion Obtain medical attention immediately; transport
may cause vomiting, abdominal pain, to hospital urgently; induce vomiting;
and diarrhoea, followed by burns in do not delay
mouth and throat; kidney damage and
shock may occur, but no lung damage
SPILLAGE STORAGE FIRE AND EXPLOSION
Absorb spillage with lime, damp Store in locked, well-ventilated Non-flammable and non-explosive
sawdust, sand, or earth; sweep up, storeroom, away from foodstuffs
place in closed container, and and animal feed, children,
dispose of safely; avoid and unauthorized personnel
contamination of personnel,
ponds, and waterways
WASTE DISPOSAL
Burn in high-temperature National Occupational Exposure UN No. 2782, 3015, 3016
incinerator with effluent scrubbing; Limit:
alternatively, treat with 5%
caustic soda as a hydrolysing agent; National Poison Control Centre:
comply with local regulations
Local trade names:
7. CURRENT REGULATIONS, GUIDELINES, AND STANDARDS
The information given in this section has been extracted from the
International Register of Potentially Toxic Chemicals (IRPTC) legal
file. A full reference to the original national document from which
the information was extracted can be obtained from IRPTC. When no
effective date appears in the IRPTC legal file, the year of the
reference from which the data are taken is indicated by (r).
The reader should be aware that regulatory decisions about chemicals,
taken in a certain country, can only be fully understood in the
framework of the legislation of that country. The regulations and
guidelines of all countries are subject to change and should always be
verified with appropriate regulatory authorities before application.
7.1 Previous Evaluations by International Bodies
The Joint FAO/WHO Meeting on Pesticide Residues (JMPR) reviewed
residues and toxicity data on diquat in 1970, 1972, 1976, 1977, and
1978. In 1977, it estimated that the acceptable daily intake (ADI)
for man was 0-0.008 mg/kg body weight, expressed as diquat ion.
The JMPR has also recommended maximum residue levels (tolerances) for
diquat in food commodities of plant and animal origin.
A data sheet on diquat has been prepared by WHO/FAO (1979) in a series
of "Data sheets on chemical pesticides" (No. 44). Technical diquat is
qualified as moderately hazardous in normal use (WHO, 1990).
7.2 Exposure Limit Values
Some exposure limit values are given in the table on pages 26-27.
7.3 Specific Restrictions
In some countries, diquat is only registered for certain specified
applications, or for use under certain specified conditions, e.g., in
the Federal Republic of Germany, Sweden, the United Kingdom, the USA,
and the USSR. For example, in the United Kingdom and the USA, the use
of the 20% liquid is restricted to bona fide certified professionals.
In the Federal Republic of Germany, diquat may not be handled by
adolescents and pregnant or nursing women.
EXPOSURE LIMIT VALUES
Medium Specification Country/ Exposure limit descriptiona Value Effective
organization date
AIR Work-place Australia Threshold limit value (TLV)
- Time-weighted average (TWA) 0.5 mg/m3 1983
- Short-term exposure limit (STEL) 1 mg/m3
Belgium Tolerable limit value (TLV)
- Time-weighted average (TWA) 0.5 mg/m3 1988
- Short-term exposure level (STEL) 1 mg/m3
Bulgaria Maximum permissible concentration
- Time-weighted average (TWA) 0.5 mg/m3 1987
Finland Maximum permissible concentration
- Time-weighted average (TWA) 0.5 mg/m3 1988
- Short-term exposure limit (STEL) 1.5 mg/m3
Netherlands Maximum permissible limit
- Time-weighted average (TWA) 0.5 mg/m3 1986
Switzerland Maximum work-site concentration (MAK)
- Time-weighted average (TWA) 0.5 mg/m3 1987
United Kingdom Recommended limit (RECL)
- 8-h time-weighted average (TWA) 0.5 mg/m3 1987
- Short-term exposure level (STEL) 1 mg/m3
(10-min time-weighted average)
USA (OSHA) Permissible exposure limit (PEL)
- Time-weighted average (TWA) 0.5 mg/m3 1989
USA (ACGIH) Threshold limit value (TLV)
- Time-weighted average (TWA) 0.5 mg/m3 1987
Medium Specification Country/ Exposure limit descriptiona Value Effective
organization date
USSR Preliminary safety level (vapour
and aerosol)
- Ceiling value (CLV) 0.2 mg/m3 1978
FOOD Intake from FAO/WHO Acceptable daily intake (ADI) 0-0.008 mg/kg 1977
(diquat ion) body weight
FOOD General FAO/WHO Maximum residue limit (MRL) 0.01-5 mg/kg 1982
for specified products
(diquat ion)
European Maximum levels in fruit and
Community vegetables (expressed as diquat
cation)
- vegetables 0.1 mg/kg 1989
- other products 0.05 mg/kg 1989
a TWA=time-weighted average over one working day (usually 8h).
7.4 Labelling, Packaging, and Transport
The United Nations Committee of Experts on the Transportation of
Dangerous Goods classifies diquat (at liquid concentrations of
45-100%) in:
- Hazard Class 6.1: poisonous substance;
- Packing Group III: a substance presenting a relatively low
risk of poisoning in transport.
The label should be as follows:
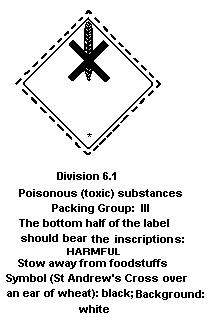 European Economic Community legislation requires labelling of diquat
as a dangerous substance using the symbol:
European Economic Community legislation requires labelling of diquat
as a dangerous substance using the symbol:
 The label must read:
Toxic in contact with skin and if swallowed; irritating to eyes,
respiratory system and skin.
The European Economic Community legislation on labelling of pesticide
preparations classifies diquat in Class 2B, for the purpose of
determining the label for preparations containing diquat and other
active ingredients.
The chemical name must be stated on the label.
The FAO specifications for plant protection products containing diquat
specify the composition and purity of its formulations and the methods
for checking this. They also specify the quality of the containers.
7.5 Waste Disposal
In the USA, diquat is regarded as a hazardous waste and permits are
required for its discharge from any point source into USA national
waters. This requirement contains detailed instructions.
BIBLIOGRAPHY
FAO (1985a) Guidelines for the packaging and storage of pesticides.
Rome, Food and Agriculture Organization of the United Nations.
FAO (1985b) Guidelines for the disposal of waste pesticides and
pesticide containers on the farm. Rome, Food and Agriculture
Organization of the United Nations.
FAO (1985c) Guidelines on good labelling practice for pesticides.
Rome, Food and Agriculture Organization of the United Nations.
FAO (1986) International code of conduct on the distribution and use
of pesticides. Rome, Food and Agriculture Organization of the United
Nations.
FAO/WHO (1986) Guide to Codex recommendations concerning pesticide
residues. Part 8. Recommendations for methods of analysis of
pesticide residues, 3rd ed. Rome, Codex Committee on Pesticide
Residues.
GIFAP (1982) Guidelines for the safe handling of pesticides during
their formulation, packing, storage and transport. Brussels,
Groupement International des Associations Nationales des Fabricants de
Produits Agrochimiques.
GIFAP (1983) Guidelines for the safe and effective use of
pesticides. Brussels, Groupement International des Associations
Nationales des Fabricants de Produits Agrochimiques.
GIFAP (1984) Guidelines for emergency measures in cases of pesticide
poisoning. Brussels, Groupement International des Associations
Nationales des Fabricants de Produits Agrochimiques.
GIFAP (1987) Guidelines for the safe transport of pesticides.
Brussels, Groupement International des Associations Nationales des
Fabricants de Produits Agrochimiques.
IARC (1972-present) IARC monographs on the evaluation of
carcinogenic risk of chemicals to man. Lyon, International Agency
for Research on Cancer.
IRPTC (1985) IRPTC file on treatment and disposal methods for waste
chemicals. Geneva, International Register for Potentially Toxic
Chemicals, United Nations Environment Programme.
IRPTC (1987) IRPTC legal file 1986. Geneva, International Register
of Potentially Toxic Chemicals, United Nations Environment Programme.
PLESTINA, R. (1984) Prevention, diagnosis, and treatment of
insecticide poisoning. Geneva, World Health Organization
(unpublished document WHO/VBC/84.889).
SAX, N.I. (1984) Dangerous properties of industrial materials. New
York, Van Nostrand Reinhold Company, Inc.
UNITED NATIONS (1986) Recommendations on the transport of dangerous
goods. 4th ed. New York, United Nations.
US NIOSH/OSHA (1981) Occupational health guidelines for chemical
hazards. 3 vol. Washington DC, US Department of Health and Human
Services, US Department of Labor (Publication No. DHSS(NIOSH) 01-123).
WHO (1984) Environmental Health Criteria 39: Paraquat and diquat.
Geneva, World Health Organization.
WHO (1990) The WHO recommended classification of pesticides by
hazard and guidelines to classification 1990/91. Geneva, World
Health Organization (unpublished document WHO/PCS/90.1).
WHO/FAO (1979) Data sheets on pesticides, No. 44: Diquat, Geneva,
World Health Organization (unpublished WHO documents).
WORTHING, C.R. & WALKER, S.B. (1987) The pesticide manual. 8th
ed. Lavenham, Lavenham Press Limited, British Crop Protection Council.
The label must read:
Toxic in contact with skin and if swallowed; irritating to eyes,
respiratory system and skin.
The European Economic Community legislation on labelling of pesticide
preparations classifies diquat in Class 2B, for the purpose of
determining the label for preparations containing diquat and other
active ingredients.
The chemical name must be stated on the label.
The FAO specifications for plant protection products containing diquat
specify the composition and purity of its formulations and the methods
for checking this. They also specify the quality of the containers.
7.5 Waste Disposal
In the USA, diquat is regarded as a hazardous waste and permits are
required for its discharge from any point source into USA national
waters. This requirement contains detailed instructions.
BIBLIOGRAPHY
FAO (1985a) Guidelines for the packaging and storage of pesticides.
Rome, Food and Agriculture Organization of the United Nations.
FAO (1985b) Guidelines for the disposal of waste pesticides and
pesticide containers on the farm. Rome, Food and Agriculture
Organization of the United Nations.
FAO (1985c) Guidelines on good labelling practice for pesticides.
Rome, Food and Agriculture Organization of the United Nations.
FAO (1986) International code of conduct on the distribution and use
of pesticides. Rome, Food and Agriculture Organization of the United
Nations.
FAO/WHO (1986) Guide to Codex recommendations concerning pesticide
residues. Part 8. Recommendations for methods of analysis of
pesticide residues, 3rd ed. Rome, Codex Committee on Pesticide
Residues.
GIFAP (1982) Guidelines for the safe handling of pesticides during
their formulation, packing, storage and transport. Brussels,
Groupement International des Associations Nationales des Fabricants de
Produits Agrochimiques.
GIFAP (1983) Guidelines for the safe and effective use of
pesticides. Brussels, Groupement International des Associations
Nationales des Fabricants de Produits Agrochimiques.
GIFAP (1984) Guidelines for emergency measures in cases of pesticide
poisoning. Brussels, Groupement International des Associations
Nationales des Fabricants de Produits Agrochimiques.
GIFAP (1987) Guidelines for the safe transport of pesticides.
Brussels, Groupement International des Associations Nationales des
Fabricants de Produits Agrochimiques.
IARC (1972-present) IARC monographs on the evaluation of
carcinogenic risk of chemicals to man. Lyon, International Agency
for Research on Cancer.
IRPTC (1985) IRPTC file on treatment and disposal methods for waste
chemicals. Geneva, International Register for Potentially Toxic
Chemicals, United Nations Environment Programme.
IRPTC (1987) IRPTC legal file 1986. Geneva, International Register
of Potentially Toxic Chemicals, United Nations Environment Programme.
PLESTINA, R. (1984) Prevention, diagnosis, and treatment of
insecticide poisoning. Geneva, World Health Organization
(unpublished document WHO/VBC/84.889).
SAX, N.I. (1984) Dangerous properties of industrial materials. New
York, Van Nostrand Reinhold Company, Inc.
UNITED NATIONS (1986) Recommendations on the transport of dangerous
goods. 4th ed. New York, United Nations.
US NIOSH/OSHA (1981) Occupational health guidelines for chemical
hazards. 3 vol. Washington DC, US Department of Health and Human
Services, US Department of Labor (Publication No. DHSS(NIOSH) 01-123).
WHO (1984) Environmental Health Criteria 39: Paraquat and diquat.
Geneva, World Health Organization.
WHO (1990) The WHO recommended classification of pesticides by
hazard and guidelines to classification 1990/91. Geneva, World
Health Organization (unpublished document WHO/PCS/90.1).
WHO/FAO (1979) Data sheets on pesticides, No. 44: Diquat, Geneva,
World Health Organization (unpublished WHO documents).
WORTHING, C.R. & WALKER, S.B. (1987) The pesticide manual. 8th
ed. Lavenham, Lavenham Press Limited, British Crop Protection Council.
 CAS chemical name: 6,7-dihydrodipyrido[1,2-a:2'1'-c]-
pyrazinediium (9CI)
Trade names (inter alia): Reglone; Aquacide; Pathclear
CAS registry number:
CAS chemical name: 6,7-dihydrodipyrido[1,2-a:2'1'-c]-
pyrazinediium (9CI)
Trade names (inter alia): Reglone; Aquacide; Pathclear
CAS registry number:  European Economic Community legislation requires labelling of diquat
as a dangerous substance using the symbol:
European Economic Community legislation requires labelling of diquat
as a dangerous substance using the symbol:
 The label must read:
Toxic in contact with skin and if swallowed; irritating to eyes,
respiratory system and skin.
The European Economic Community legislation on labelling of pesticide
preparations classifies diquat in Class 2B, for the purpose of
determining the label for preparations containing diquat and other
active ingredients.
The chemical name must be stated on the label.
The FAO specifications for plant protection products containing diquat
specify the composition and purity of its formulations and the methods
for checking this. They also specify the quality of the containers.
7.5 Waste Disposal
In the USA, diquat is regarded as a hazardous waste and permits are
required for its discharge from any point source into USA national
waters. This requirement contains detailed instructions.
BIBLIOGRAPHY
FAO (1985a) Guidelines for the packaging and storage of pesticides.
Rome, Food and Agriculture Organization of the United Nations.
FAO (1985b) Guidelines for the disposal of waste pesticides and
pesticide containers on the farm. Rome, Food and Agriculture
Organization of the United Nations.
FAO (1985c) Guidelines on good labelling practice for pesticides.
Rome, Food and Agriculture Organization of the United Nations.
FAO (1986) International code of conduct on the distribution and use
of pesticides. Rome, Food and Agriculture Organization of the United
Nations.
FAO/WHO (1986) Guide to Codex recommendations concerning pesticide
residues. Part 8. Recommendations for methods of analysis of
pesticide residues, 3rd ed. Rome, Codex Committee on Pesticide
Residues.
GIFAP (1982) Guidelines for the safe handling of pesticides during
their formulation, packing, storage and transport. Brussels,
Groupement International des Associations Nationales des Fabricants de
Produits Agrochimiques.
GIFAP (1983) Guidelines for the safe and effective use of
pesticides. Brussels, Groupement International des Associations
Nationales des Fabricants de Produits Agrochimiques.
GIFAP (1984) Guidelines for emergency measures in cases of pesticide
poisoning. Brussels, Groupement International des Associations
Nationales des Fabricants de Produits Agrochimiques.
GIFAP (1987) Guidelines for the safe transport of pesticides.
Brussels, Groupement International des Associations Nationales des
Fabricants de Produits Agrochimiques.
IARC (1972-present) IARC monographs on the evaluation of
carcinogenic risk of chemicals to man. Lyon, International Agency
for Research on Cancer.
IRPTC (1985) IRPTC file on treatment and disposal methods for waste
chemicals. Geneva, International Register for Potentially Toxic
Chemicals, United Nations Environment Programme.
IRPTC (1987) IRPTC legal file 1986. Geneva, International Register
of Potentially Toxic Chemicals, United Nations Environment Programme.
PLESTINA, R. (1984) Prevention, diagnosis, and treatment of
insecticide poisoning. Geneva, World Health Organization
(unpublished document WHO/VBC/84.889).
SAX, N.I. (1984) Dangerous properties of industrial materials. New
York, Van Nostrand Reinhold Company, Inc.
UNITED NATIONS (1986) Recommendations on the transport of dangerous
goods. 4th ed. New York, United Nations.
US NIOSH/OSHA (1981) Occupational health guidelines for chemical
hazards. 3 vol. Washington DC, US Department of Health and Human
Services, US Department of Labor (Publication No. DHSS(NIOSH) 01-123).
WHO (1984) Environmental Health Criteria 39: Paraquat and diquat.
Geneva, World Health Organization.
WHO (1990) The WHO recommended classification of pesticides by
hazard and guidelines to classification 1990/91. Geneva, World
Health Organization (unpublished document WHO/PCS/90.1).
WHO/FAO (1979) Data sheets on pesticides, No. 44: Diquat, Geneva,
World Health Organization (unpublished WHO documents).
WORTHING, C.R. & WALKER, S.B. (1987) The pesticide manual. 8th
ed. Lavenham, Lavenham Press Limited, British Crop Protection Council.
The label must read:
Toxic in contact with skin and if swallowed; irritating to eyes,
respiratory system and skin.
The European Economic Community legislation on labelling of pesticide
preparations classifies diquat in Class 2B, for the purpose of
determining the label for preparations containing diquat and other
active ingredients.
The chemical name must be stated on the label.
The FAO specifications for plant protection products containing diquat
specify the composition and purity of its formulations and the methods
for checking this. They also specify the quality of the containers.
7.5 Waste Disposal
In the USA, diquat is regarded as a hazardous waste and permits are
required for its discharge from any point source into USA national
waters. This requirement contains detailed instructions.
BIBLIOGRAPHY
FAO (1985a) Guidelines for the packaging and storage of pesticides.
Rome, Food and Agriculture Organization of the United Nations.
FAO (1985b) Guidelines for the disposal of waste pesticides and
pesticide containers on the farm. Rome, Food and Agriculture
Organization of the United Nations.
FAO (1985c) Guidelines on good labelling practice for pesticides.
Rome, Food and Agriculture Organization of the United Nations.
FAO (1986) International code of conduct on the distribution and use
of pesticides. Rome, Food and Agriculture Organization of the United
Nations.
FAO/WHO (1986) Guide to Codex recommendations concerning pesticide
residues. Part 8. Recommendations for methods of analysis of
pesticide residues, 3rd ed. Rome, Codex Committee on Pesticide
Residues.
GIFAP (1982) Guidelines for the safe handling of pesticides during
their formulation, packing, storage and transport. Brussels,
Groupement International des Associations Nationales des Fabricants de
Produits Agrochimiques.
GIFAP (1983) Guidelines for the safe and effective use of
pesticides. Brussels, Groupement International des Associations
Nationales des Fabricants de Produits Agrochimiques.
GIFAP (1984) Guidelines for emergency measures in cases of pesticide
poisoning. Brussels, Groupement International des Associations
Nationales des Fabricants de Produits Agrochimiques.
GIFAP (1987) Guidelines for the safe transport of pesticides.
Brussels, Groupement International des Associations Nationales des
Fabricants de Produits Agrochimiques.
IARC (1972-present) IARC monographs on the evaluation of
carcinogenic risk of chemicals to man. Lyon, International Agency
for Research on Cancer.
IRPTC (1985) IRPTC file on treatment and disposal methods for waste
chemicals. Geneva, International Register for Potentially Toxic
Chemicals, United Nations Environment Programme.
IRPTC (1987) IRPTC legal file 1986. Geneva, International Register
of Potentially Toxic Chemicals, United Nations Environment Programme.
PLESTINA, R. (1984) Prevention, diagnosis, and treatment of
insecticide poisoning. Geneva, World Health Organization
(unpublished document WHO/VBC/84.889).
SAX, N.I. (1984) Dangerous properties of industrial materials. New
York, Van Nostrand Reinhold Company, Inc.
UNITED NATIONS (1986) Recommendations on the transport of dangerous
goods. 4th ed. New York, United Nations.
US NIOSH/OSHA (1981) Occupational health guidelines for chemical
hazards. 3 vol. Washington DC, US Department of Health and Human
Services, US Department of Labor (Publication No. DHSS(NIOSH) 01-123).
WHO (1984) Environmental Health Criteria 39: Paraquat and diquat.
Geneva, World Health Organization.
WHO (1990) The WHO recommended classification of pesticides by
hazard and guidelines to classification 1990/91. Geneva, World
Health Organization (unpublished document WHO/PCS/90.1).
WHO/FAO (1979) Data sheets on pesticides, No. 44: Diquat, Geneva,
World Health Organization (unpublished WHO documents).
WORTHING, C.R. & WALKER, S.B. (1987) The pesticide manual. 8th
ed. Lavenham, Lavenham Press Limited, British Crop Protection Council.
