
IPCS INTERNATIONAL PROGRAMME ON CHEMICAL SAFETY
Health and Safety Guide No. 55
1,2-DICHLOROETHANE
HEALTH AND SAFETY GUIDE
UNITED NATIONS ENVIRONMENT PROGRAMME
INTERNATIONAL LABOUR ORGANISATION
WORLD HEALTH ORGANIZATION
WORLD HEALTH ORGANIZATION, GENEVA 1991
This is a companion volume to Environmental Health Criteria 62:
1,2-Dichloroethane
Published by the World Health Organization for the International
Programme on Chemical Safety (a collaborative programme of the United
Nations Environment Programme, the International Labour Organisation,
and the World Health Organization)
This report contains the collective views of an international group of
experts and does not necessarily represent the decisions or the stated
policy of the United Nations Environment Programme, the International
Labour Organisation, or the World Health Organization
WHO Library Cataloguing in Publication Data
1,2-Dichloroethane : health and safety guide.
(Health and safety guide ; no. 55)
1. Ethylene dichlorides - standards I. Series
ISBN 92 4 151055 2 (NLM Classification: QV 633)
ISSN 0259-7268
(c) World Health Organization 1991
Publications of the World Health Organization enjoy copyright
protection in accordance with the provisions of Protocol 2 of the
Universal Copyright Convention. For rights of reproduction or
translation of WHO publications, in part or in toto, application
should be made to the Office of Publications, World Health
Organization, Geneva, Switzerland. The World Health Organization
welcomes such applications.
The designations employed and the presentation of the material in this
publication do not imply the expression of any opinion whatsoever on
the part of the Secretariat of the World Health Organization
concerning the legal status of any country, territory, city or area or
of its authorities, or concerning the delimitation of its frontiers or
boundaries.
The mention of specific companies or of certain manufacturers'
products does not imply that they are endorsed or recommended by the
World Health Organization in preference to others of a similar nature
that are not mentioned. Errors and omissions excepted, the names of
proprietary products are distinguished by initial capital letters.
CONTENTS
INTRODUCTION
1. PRODUCT IDENTITY AND USES
1.1. Identity
1.2. Physical and chemical properties
1.3. Composition
1.4. Uses
2. SUMMARY AND EVALUATION
2.1. Human exposure
2.2. Fate of 1,2-dichloroethane
2.3. Uptake, metabolism, and excretion
2.4. Effects on organisms in the environment
2.5. Effects on animals and cell systems
2.6. Effects on human beings
3. CONCLUSIONS
4. HUMAN HEALTH HAZARDS, PREVENTION AND PROTECTION, EMERGENCY ACTION
4.1. Main human health hazards, prevention and protection,
first aid
4.2. Advice to physicians
4.3. Health surveillance advice
4.4. Explosion and fire hazards
4.4.1. Explosion hazards
4.4.2. Fire hazards
4.4.3. Prevention
4.4.4. Fire-extinguishing agents
4.5. Storage
4.6. Transport
4.7. Spillage and disposal
4.7.1. Spillage
4.7.2. Disposal
5. HAZARDS FOR THE ENVIRONMENT AND THEIR PREVENTION
6. SUMMARY OF CHEMICAL SAFETY INFORMATION
7. CURRENT REGULATIONS, GUIDELINES, AND STANDARDS
7.1. Exposure limit values
7.2. Specific restrictions
7.3. Labelling, packaging, and transport
7.4. Waste disposal
BIBLIOGRAPHY
INTRODUCTION
The Environmental Health Criteria (EHC) documents produced by the
International Programme on Chemical Safety include an assessment of
the effects on the environment and on human health of exposure to a
chemical or combination of chemicals, or physical or biological
agents. They also provide guidelines for setting exposure limits.
The purpose of a Health and Safety Guide is to facilitate the
application of these guidelines in national chemical safety
programmes. The first three sections of a Health and Safety Guide
highlight the relevant technical information in the corresponding EHC.
Section 4 includes advice on preventive and protective measures and
emergency action; health workers should be thoroughly familiar with
the medical information to ensure that they can act efficiently in an
emergency. Within the Guide is a Summary of Chemical Safety
Information which should be readily available, and should be clearly
explained, to all who could come into contact with the chemical. The
section on regulatory information has been extracted from the legal
file of the International Register of Potentially Toxic Chemicals
(IRPTC) and from other United Nations sources.
The target readership includes occupational health services, those in
ministries, governmental agencies, industry, and trade unions who are
involved in the safe use of chemicals and the avoidance of
environmental health hazards, and those wanting more information on
this topic. An attempt has been made to use only terms that will be
familiar to the intended user. However, sections 1 and 2 inevitably
contain some technical terms. A bibliography has been included for
readers who require further background information.
Revision of the information in this Guide will take place in due
course, and the eventual aim is to use standardized terminology.
Comments on any difficulties encountered in using the Guide would be
very helpful and should be addressed to:
The Manager
International Programme on Chemical Safety
Division of Environmental Health
World Health Organization
1211 Geneva 27
Switzerland
THE INFORMATION IN THIS GUIDE SHOULD BE CONSIDERED AS A STARTING POINT
TO A COMPREHENSIVE HEALTH AND SAFETY PROGRAMME
1. PRODUCT IDENTITY AND USES
1.1 Identity
Common name: 1,2-dichloroethane
Molecular formula: C2H4Cl2
Chemical structure:
H H
' '
Cl - C - C - Cl
' '
H H
Common trade names: Borer-Sol; Brocide; Destruxol;
Dichlor-emulsion; Dichlor-mulsion; Dutch
Liquid; Dutch Oil; ENT 1656; Gaze
Olefiant
Common synonyms: alpha, beta-dichloroethane;
1,2-bichloroethane; ethane dichloride;
ethylene chloride; ethylene dichloride;
1,2-ethylene dichloride;
sym(metric)-dichloroethane
Abbreviation: EDC
CAS registry number: 107-06-2
RTECS registry number: KI0525000
Conversion factor 1 ppm = 4.05 mg/m3 air, and
1 mg/m3 = 0.25 ppm
at 25°C and 101.3 kPa (760 mmHg)
1.2 Physical and Chemical Properties
1,2-Dichloroethane is a colourless liquid, with a sweet taste and a
chloroform-like odour. Reported odour thresholds are 25-450 mg/m3
for perception and 162-750 mg/m3 for recognition. 1,2-Dichlorethane
is a volatile and flammable compound. When dry, it is stable at
ordinary temperatures. In the presence of air, moisture, and light,
the liquid decomposes slowly. Some physical and chemical properties
of 1,2-dichloroethane are given in the Summary of Chemical Safety
Information (section 6).
1.3 Composition
Most commercial 1,2-dichloroethane is 97-99% pure and contains
approximately 0.1%, by weight, of alkylamines, to inhibit
decomposition. Impure 1,2-dichloroethane may contain polychlorinated
ethanes, and the uninhibited product may also contain chorine and/or
hydrogen chlorine.
1.4 Uses
1,2-Dichloroethane has not been reported to occur naturally. It is
used mainly for the synthesis of vinyl chloride. Other chemicals
produced from 1,2-dichloroethane are 1,1,1-trichloroethane,
ethyleneamines, vinylidene chloride, trichloroethylene, and
tetrachloroethylene. Small quantities are used as a lead scavenger in
gasoline, and for solvent and seed fumigant applications.
2. SUMMARY AND EVALUATION
2.1 Human Exposure
Human exposure mainly occurs at, and in the vicinity of, production
facilities, through inhalation and skin contact. Further human
exposure to the vapour of this compound can occur, when it is used in
gasoline, as a solvent, or as a seed fumigant. Accidental, oral
intoxication has frequently been reported. Average concentrations
near production facilities have been below 40µg/m3. In the air of
cities, average concentrations of between 0.3 and 6.5 µg/m3, with a
reported maximum of 30 µg/m3, have been measured. Average levels in
drinking-water are generally below 1 µg/litre. There are no recent
data on occupational exposure levels.
2.2 Fate of 1,2-Dichloroethane
Approximately 0.2% of the total volume of 1,2-dichloroethane produced
is estimated to be lost to the air, water, and soil (two-thirds from
production facilities and one-third following dispersive use).
Approximately 35% of the emissions of 1,2-dichloroethane, associated
with the production of the compound itself and end-products, were
estimated to occur via disposal of heavy ends, the so-called EDC-tars,
a mixture of low- and high-boiling chlorinated hydrocarbons. In the
atmosphere, 1,2-dichloroethane is degraded by sunlight, fairly
rapidly. The products of this degradation are oxides of carbon and
hydrogen chloride. The process is rapid enough to prevent
accumulation of the compound in the atmosphere. Evaporation is the
major pathway by which 1,2-dichloroethane is lost from water.
Chemical degradation and biodegradation are relatively insignificant
processes. Bioconcentration is unlikely.
2.3 Uptake, Metabolism, and Excretion
1,2-Dichloroethane is readily absorbed via the dermal, oral, and
inhalation routes. In rodents, it is distributed widely over the
body, with a preference for adipose tissue and liver, directly after
oral exposure, and for adipose tissue alone, directly after
inhalation. Most of the absorbed chemical is excreted rapidly via the
urine, mainly as glutathione conjugates, and via the lungs, as carbon
dioxide or the unchanged compound.
In vivo, the metabolism of 1,2-dichloroethane appears to be a
saturable process in rodents. At low exposure levels, most of the
body burden is recovered in the form of urinary metabolites. As
exposure levels increase, a greater fraction of the compound is
excreted unchanged via the lungs.
Metabolism takes place through two known pathways: one via P-450-
mediated microsomal oxidation and the other via glutathione
conjugation. The former pathway involves the formation of
2-chloroacetaldehyde and 2-chloroethanol. This pathway does not
appear to be important in producing DNA damage in vivo. Reactive
intermediates, capable of interacting with DNA, are formed when
1,2-dichloroethane is metabolized via glutathione conjugation.
2.4 Effects on Organisms in the Environment
Concentrations of between 85 and 550 mg/litre water are lethal for
half the number of fish exposed for 1-4 days (LC50). The LC50
value (1 or 2 days) for Daphnia magna varies between 250 and
320 mg/litre. In a 28-day test with Daphnia magna, the
no-observed-effect levels were 11 mg/litre, on the basis of
reproduction, and 42 mg/litre on the basis of length. Bioaccumulation
in fish is unlikely. Although 1,2-dichloroethane was slightly toxic
for the aquatic species tested, it does not pose a significant hazard
for the aquatic environment, except in the case of accident or
inappropriate disposal.
1,2-Dichloroethane, at an influent concentration of 258 mg/litre, did
not affect the treatment efficiency of a bench-scale activated sludge
system. In a batch anaerobic assay, 1,2-dichloroethane, at a
concentration of 2.5 mg/litre, was slightly toxic for the digestive
process.
2.5 Effects on Animals and Cell Systems
Single oral doses of 680 and 850 mg/kg body weight and exposure to
vapour concentrations of 5100-6666 mg/m3, for 6h, have been shown to
be lethal for half the number of exposed rats in a population (LD50
and LC50, respectively). Thus, the compound is slightly toxic,
according to the scale of Hodge & Sterner. The LD50 and 6-h LC50
for mice were 413-489 mg/kg body weight and 1060 mg/m3,
respectively.
In an exposure-related inhalation study on rats, no adverse effects
were observed with a 7-h exposure to 1200 mg/m3. At higher levels,
central nervous system depression and an increased mortality rate were
noted. Liver and kidney damage was found in most animals that died.
After a single oral dose of 615-770 mg/kg body weight, liver damage,
myocardial oedema, and damage to coronary vessels were observed.
In 3 short-term inhalation studies, various species were exposed to
concentrations of between 405 and 3900 mg 1,2-dichloroethane/m3 air,
for 6 or 7 h per day, and 5 days per week. Mice and rats appeared to
be more sensitive than guinea-pigs, rabbits, monkeys, dogs, or cats.
The overall no-observed-effect level in the rat, for exposure periods
ranging from 4 to 9 months, was about 400 mg/m3. Signs of toxicity,
including central nervous system depression and death, were observed
in all species exposed to concentrations of between 1620 and
3900 mg/m3. In rats, fatty changes in the liver were observed at
concentrations from 1540 mg/m3 upwards. Repeated oral
administration of 1,2-dichloroethane, at a dose of 300 mg/kg body
weight, was lethal for rats, and produced necrosis and fatty changes
in the liver. No effects were observed in rats when the chemical was
given orally at 10 mg/kg body weight per day, for 90 days, or at
150 mg/kg body weight, 5 times per week, for 2 weeks.
1,2-Dichloroethane causes corneal damage in animals, but no gross skin
reactions occurred with a patch test on guinea-pigs. Corneal opacity
was observed in dogs, following subcutaneous injection.
1,2-Dichloroethane is weakly mutagenic in Salmonella typhimurium
T1535. A stronger response was obtained after metabolic activation by
cytosolic glutathione S-transferase. Mutagenicity also occurs in
fungi, Drosophila, and mammalian cells in vitro. In two human cell
lines, the incidence of gene mutations was found to increase with
increasing levels of glutathione S-transferase. A weak mutagenic
effect was reported in a spot test on mice. DNA damage has been
observed in bacteria, mammalian cells in vitro, and in mammals in
vivo. 1,2-Dichloroethane did not induce cell transformation in one
of two assays, and enhanced virus-induced cell transformation in the
other. Negative results were obtained in one dominant-lethal assay
and in 2 micronucleus assays on mice.
1,2-Dichloroethane was carcinogenic in B6C3F1 mice and Osborne-Mendel
rats, following oral administration of doses of 50-300 mg/kg body
weight, given, in oil, by gavage. The compound produced a
statistically-significant increase in squamous cell carcinoma of the
forestomach, haemangiosarcoma, and mammary adenocarcinoma in rats, and
mammary adenocarcinoma and hepatocellular carcinoma in mice. In
inhalation studies on Swiss mice and Sprague Dawley rats, using
concentrations of up to 607 mg/m3, no increase in tumour incidence
was reported. Taking into consideration that cancer has been produced
in two species of experimental animals and in several target organs,
it can be concluded that 1,2-dichloroethane is carcinogenic for rats
and mice, when administered by gavage.
1,2-Dichloroethane has been found in the fetuses of rats following
inhalation by the dams. Fetal toxicity was observed in two studies on
rats at low exposure levels. However, fetal toxicity could not be
observed in two other studies on rats and one on rabbits at higher
exposure levels.
2.6 Effects on Human Beings
The first symptoms of acute intoxication in human beings include
depression of the central nervous system, gastroenteritis, and
irritation of the mucous membranes of the eyes and respiratory tract.
An interval relatively free of symptoms can be experienced. The
intoxication can also lead to cardiovascular insufficiency (often the
cause of death), blood coagulation disorders, lung oedema, and
functional abnormalities of the liver and kidneys.
Quantitative exposure-effect data on human beings are limited to two
early reports on small groups of occupationally-exposed men.
Evaluation of these data indicate that repeated inhalation exposure in
the range of approximately 40-800 mg/m3 may lead to depression of
the central nervous system and gastrointestinal and liver
abnormalities. Because of the limitations of the human data base, it
is necessary to rely on the data available from animal studies, to
derive a no-observed-effect level for human beings. This is possible
because of the similarity in the spectrum of adverse effects in man
and laboratory animals. These effects include central nervous system
depression, liver, and possibly kidney, abnormalities, lung oedema,
and cardiovascular disorders. The dose-response data from animal
studies include a no-observed-effect level of 400 mg 1,2-
dichloroethane/m3 air. After considering current human exposure
levels in air and drinking-water, it can be concluded that, even in
the context of a worst-case scenario, 1,2-dichloroethane is unlikely
to present a toxic hazard for the general population, under prevailing
exposure conditions.
In the absence of human data, and taking into account that
1,2-dichloroethane produces a reactive intermediate that alkylates the
DNA, is weakly positive in a number of in vitro mutagenicity tests,
and results in the production of both rare and common tumours in rats
and mice, it would be prudent to consider it as a possible human
carcinogen. Therefore, 1,2-dichloroethane should be regarded, for
practical purposes, as if it presented a carcinogenic risk for man,
and levels in the environment should be kept as low as feasible.
Since there are no human data, it is necessary to rely on the limited
data available from laboratory animal investigations in evaluating
human reproduction hazards and teratogenicity. The weight of evidence
suggests that exposure to prevailing environmental levels does not
pose a reproductive or teratogenic hazard.
3. CONCLUSIONS AND RECOMMENDATIONS
Taking into account the body of available data, it would be prudent to
consider 1,2-dichloroethane as a possible human carcinogen and, for
practical purposes, to regard it as a carcinogenic risk for human
beings. Thus, levels in the environment should be kept as low as
feasible.
After a single oral or inhalation exposure, 1,2-dichloroethane may
irritate the mucous membranes of the eyes and gastrointestinal and
respiratory tracts, and may cause systemic changes in the central
nervous system, gastrointestinal tract, and liver in human beings.
Liquid 1,2-dichloroethane is severely irritating to the skin.
It is not possible to derive a no-observed-effect level for
non-carcinogenic effects on the basis of available human data.
However, a no-observed-effect level of 400 mg/m3 can be established
on the basis of animal toxicity data. After considering current human
exposure levels in air and in drinking-water, it can be concluded
that, under prevailing exposure conditions, 1,2-dichloroethane is
unlikely to present a toxic hazard for the general population.
There are no human data for the evaluation of the reproductive hazards
and teratogenicity. The weight of evidence from experimental animal
studies does not suggest that exposure of human beings to prevailing
environmental levels would pose a reproductive or teratogenic hazard.
1,2-Dichloroethane will not accumulate in the atmosphere.
Bioconcentration is unlikely. Although it was slightly toxic for the
aquatic species tested, 1,2-dichloroethane does not pose a significant
hazard for the aquatic environment, except in the case of accident or
inappropriate disposal.
4. HUMAN HEALTH HAZARDS, PREVENTION AND PROTECTION, EMERGENCY ACTION
4.1 Main Human Health Hazards, Prevention and Protection, First Aid
The human health hazards associated with certain types of exposure to
1,2-dichloroethane, together with preventive and protective measures
and first aid recommendations, are listed in the Summary of Chemical
Safety Information (section 6).
4.2 Advice to Physicians
No specific antidote to 1,2-dichloroethane poisoning is known. Treat
symptomatically. Be aware that, following the initial symptoms of
oral poisoning, central nervous system depression and respiratory and
circulatory failure may occur after an interval relatively free of
symptoms.
4.3 Health Surveillance Advice
Human beings potentially exposed to 1,2-dichloroethane should undergo
periodic medical examination, with emphasis on effects on the eyes,
skin, central nervous system, gastrointestinal tract, liver, kidneys,
and cardiovascular system, and on the functioning of the central
nervous system.
4.4 Explosion and Fire Hazards
4.4.1 Explosion hazards
Above 13°C, explosive vapour-air mixtures may be formed that can be
ignited by open fires, sparks, or glowing surfaces. Because of its
low electroconductivity, the compound can generate electrostatic
charges as a result of flow or agitation. The vapours are heavier
than air, may travel along the ground, and be ignited from a distance.
1,2-Dichloroethane will react violently with oxidants (e.g., nitric
acid). Mixtures with dinitrogen tetroxide (N2O4), sodium,
potassium, aluminium, magnesium, liquid ammonia, or
dimethylaminopropylamine may be explosive.
4.4.2 Fire hazards
1,2-Dichloroethane is a flammable liquid. It decomposes in a flame or
on a hot surface, to form phosgene and hydrochloric acid.
4.4.3 Prevention
Use closed systems, ventilation, and explosion-proof electrical
equipment. Make connections to earth. Do not use the compound near
sources of ignition. Do not use compressed air for filling,
discharging, or handling. In case of fire, keep drums cool by
spraying with water. Fire-fighters should use self-contained
breathing apparatus.
4.4.4 Fire-extinguishing agents
Use dry chemical, carbon dioxide, alcohol foam, halons, water sprays.
4.5 Storage
1,2-Dichloroethane should be stored in a dark, dry, fire-proof, and
properly labelled room, with ventilation across the floor. It should
be kept away from food, oxidants, and other incompatible substances
(section 4.4.1).
4.6 Transport
In case of accident, stop the engine. Remove all sources of ignition.
Keep bystanders at a distance and mark the roads. Keep upwind. In
case of spillage or fire, use the methods advised in sections 4.7 and
4.4, respectively. Notify the police and the fire brigade
immediately. In case of poisoning, follow the advice in section 4.1.
4.7 Spillage and Disposal
4.7.1 Spillage
Remove all ignition sources and evacuate the danger area. Collect
leaking liquid in a sealable container. Absorb spilled liquid in
sand, earth, or other inert absorbent. Do not allow run-off into a
sewer. Remove the inert absorbent to a safe place, then flush the
area with water. Ensure personal protection by using a self-contained
breathing apparatus.
4.7.2 Disposal (based on the IRPTC waste disposal file)
Solvent wastes may be regenerated commercially using fractional
distillation. Concentrated wastes, such as distillation residues,
spent catalysts, and complex sludges (tars), are disposed of in
special waste incinerators, since phosgene is liberated during
burning. Dilute with kerosene or fuel oil, because of high chlorine
content. Aqueous wastes that contain 1,2-dichloroethane (process
effluents) are aerated, until the volatile chlorohydrocarbon has
evaporated. Special attention should be paid to emission limits.
5. HAZARDS FOR THE ENVIRONMENT AND THEIR PREVENTION
1,2-Dichloroethane will not accumulate in the atmosphere.
Bioconcentration is unlikely. Although it was slightly toxic for the
aquatic species tested, 1,2-dichloroethane does not pose a signficant
hazard for the aquatic environment, except in case of accident or
inappropriate disposal.
Contamination of soil, water, and the atmosphere can be avoided by
proper methods of storage, transport, handling, and waste disposal.
In cases of spillage, apply methods recommended in section 4.7.1.
6. SUMMARY OF CHEMICAL SAFETY INFORMATION
This summary should be easily available to all health workers
concerned with, and users of, 1,2-dichloroethane It should be
displayed at, or near, entrances to areas where there is potential
exposure to 1,2-dichloroethane, and on processing equipment and
containers. The summary should be translated into the appropriate
language(s). All persons potentially exposed to the chemical should
also have the instructions in the summary clearly explained.
Space is available for insertion of the National Occupational
Exposure Limit, the address and telephone number of the National
Poison Control Centre, and for local trade names.
1,2-DICHLOROETHANE
(EDC, ethane dichloride, ethylene dichloride, glycol dichloride) (ClCH2-CH2Cl)
PHYSICAL PROPERTIES OTHER CHARACTERISTICS
Relative molecular mass 98.96 Colourless liquid with a sweet taste and a chloroform-like
Melting point (°C) -35 odour; can have adverse effects well below the odour
Boiling point (°C) 83 threshold; the compound can generate electrostatic charges
Water solubility (20°C) (g/litre) 8.69 through flow or agitation; it will react violently with
oxidizing
Specific density (20°C) 1.23 agents, which are mixed with dinitrogen tetroxide (N2O4),
Relative vapour density 3.42 sodium, potassium, aluminium, magnesium, liquid ammonia,
Vapour pressure (20°C) (kPa) 8.53 or dimethylaminopropylamine; may be explosive; vapours are
Flash point (°C) (closed cup) 13 heavier than air, travel along the ground, and can be ignited
Flammability (explosive) limits (Vol. %) 6-16 from a distance; the compound decomposes in contact with a
Log n-octanol/water partition coefficient 1.48 flame or on a hot surface forming phosgene and hydrochloric
acid
HAZARDS/SYMPTOMS PREVENTION AND PROTECTION FIRST AID
SKIN: irritation and degreasing; Wear clean protective gloves and Remove contaminated clothing; wash with
redness; may enter body through protective clothing water and soap
skin
EYES: irritation and inflammation; Wear face-shield Rinse with plenty of water for at least
redness; blurred vision 15 min; send to a doctor
INHALATION: irritation and Explosion-proof exhaust ventilation Remove victims to fresh air and keep
inflammation of respiratory and or, for non-routine activities, quiet; if breathing has stopped, apply
gastrointestinal tracts; central air-supplied or self-contained artificial respiration
nervous system depression; effects breathing apparatus
on liver and kidneys; cyanosis,
vomiting, diarrhoea, headache,
dizziness, dullness
GENERAL: should be regarded as
posing a carcinogenic risk for man
INGESTION: irritation and Do not eat, drink, or smoke during Rinse mouth; give plenty of water to
inflammation of gastrointestinal work drink; induce vomiting in conscious
tract; central nervous system patients
depression; effects on liver and
kidneys; effects may be delayed;
cyanosis, vomiting, diarrhoea,
headache, dizziness, dullness
ENVIRONMENT: the compound Apply proper methods of storage,
can be slightly toxic for aquatic transport, waste disposal, and
life and handling of spills
SPILLAGE STORAGE FIRE AND EXPLOSION
Remove ignition sources; evacuate Store in dark, dry, fire-proof, Flammable; vapour-air mixtures may be
area; collect leaking liquid in a and properly labelled room, with explosive; no sources of ignition; make
sealable container; do not allow ventilation across the floor; store connections to earth; do not use
run-off into a sewer; absorb spills away from foodstuffs, oxidants, compressed air for handling; use closed
in sand or other inert absorbent, and other incompatible substances; systems, ventilation, and explosion-
and remove to a safe place; ensure containers should be tightly proof electrical equipment; in case of
personal protection by using a closed and labelled fire, keep drums cool by spraying with
self-contained breathing apparatus water; extinguish fire with dry
chemical, carbon dioxide, alcohol foam,
halons, or water sprays
WASTE DISPOSAL NATIONAL INFORMATION
Disposal in special waste National Occupational Exposure Limits: UN: 1184
incinerators with dilution by
kerosene or fuel oil; aqueous
wastes should be aerated; solvent
wastes may be regenerated National Poison Control Centre:
7. CURRENT REGULATIONS, GUIDELINES, AND STANDARDS
The information given in this section has been extracted from the
International Register of Potentially Toxic Chemicals (IRPTC) legal
file. A full reference to the original national document from which
the information was extracted can be obtained from IRPTC. When no
effective date appears in the IRPTC legal file, the year of the
reference from which the data are taken is indicated by (r).
The reader should be aware that regulatory decisions about chemicals,
taken in a certain country, can only be fully understood in the
framework of the legislation of that country. The regulations and
guidelines of all countries are subject to change and should always be
verified with appropriate regulatory authorities before application.
7.1 Exposure Limit Values
Some exposure limit values are given in the table on pp. 26-28.
7.2 Specific Restrictions
The European Economic Community prohibits the use and marketing of
1,2-dichloroethane as part of the composition of cosmetic products
(effective date: 1986). Member states should ensure that dangerous
preparations (solvents) are not placed on the market, unless their
packages and fastenings and labels comply with the requirements laid
down (effective date: 1984).
In the German Democratic Republic, the use of 1,2-dichloroethane is
not permitted in the manufacture of consumer goods (effective date:
1977).
In the USSR, the compound is approved as an insecticide for
agricultural use with specifications for application, dose, mode, and
treatment frequency (effective date: 1982).
In the USA, the compound is exempted from tolerance requirements in
plant products and animal products, when used according to good
agricultural practice as an inert (or occasionally active) ingredient
of pesticides, applied for some specified purposes (effective date:
1983 (r)). It is exempted from residue tolerance requirements, when
used as a fumigant, after harvest, on certain specified grains
(effective date: 1983 (r)). It is permitted as a food additive in
feed and drinking-water for animals, with restriction on the use as a
solvent in the extraction processing of animal by-products for use in
animal feed (effective date: 1983 (r)).
Medium Specification Country/ Exposure limit descriptiona Value Effective
organization date
AIR Occupational Australia Threshold limit value (TLV) 1985 (r)
- Time-weighted average (TWA) 200 mg/m3
Brazil Acceptable limit 1982 (r)
- 48 h/week 156 mg/m3
Czechoslovakia Maximum allowable concentration 1985
- Time-weighted average (TWA) 50 mg/m3
Ceiling value 100 mg/m3 1985
Germany, Federal Maximum work-site concentration 1986 (r)
Republic of - Time-weighted average (TWA) 80 mg/m3b
Short-term exposure limit (STEL) 1986 (r)
- 30 min 160 mg/m3
Japan Maximum allowable concentration (MAC) 1984
40 mg/m3
Netherlands Maximum limit 1985 (r)
- Time-weighted average (TWA) 200 mg/m3
Poland Ceiling value 10 mg/m3c 1985 (r)
Sweden Threshold limit value (TLV) 1985
- Time-weighted average (TWA) 4 mg/m3d
Short-term exposure limit (STEL) 1985
- 15-min time-weighted average 20 mg/m3
United Kingdom Recommended threshold limit value 1985 (r)
- Time-weighted average (TWA) 40 mg/m3
Short-term exposure limit (STEL) 1985 (r)
- 10-min time-weighted average 60 mg/m3
Medium Specification Country/ Exposure limit description Value Effective
organization date
AIR Occupational USA (ACGIH) Threshold limit value (TLV) 1985 (r)
- Time-weighted average (TWA) 40 mg/m3
USA (ACGIH) Short-term exposure limit (STEL) 60 mg/m3 1985 (r)
USA (OSHA) Threshold limit value (TLV) 1974
- Time-weighted average (TWA) 202 mg/m3
Ceiling value 1974
- Acceptable 405 mg/m3
Ceiling value 1974
- 5 min/3 h 810 mg/m3
USA (NIOSH) Threshold limit value (TLV)
- Time-weighted average (TWA) 40 mg/m3
Short-term exposure limit (STEL)
- 15 min 80 mg/m3
AIR Ambient Czechoslovakia Maximum allowable concentration (MAC) 1982 (r)
- Average per day 1 mg/m3
Maximum allowable concentration (MAC) 1982 (r)
- Average per 0.5 h 3 mg/m3
WATER Surface USSR Preliminary safety level 0.21 mg/litre 1983
Drinking- WHO Guideline 0.01 mg/litre 1983 (r)
Medium Specification Country/ Exposure limit description Value Effective
organization date
FOOD FAO/WHO Allowable daily intake nonee 1983 (r)
Bread, cereal Guideline 0.1 mg/kg 1982 (r)
products
Cereal grains Guideline 50 mg/kg 1982 (r)
a TWA = time-weighted average over one working day (usually 8 h).
b Suspected potential carcinogen; the exposure limits are therefore tentatively retained.
c Skin absorption.
d Skin absorption; carcinogenic.
e The substance should be used in accordance with good manufacturing practice.
7.3 Labelling, Packaging, and Transport
The European Economic Community regulations state that the label
should read as follows (effective date: 1976 (r)):
Highly flammable; harmful by inhalation; keep container tightly
closed; keep away from sources of ignition - no smoking; do not
empty into drains; take precautionary measures against static
discharges.
The recommended labels are:
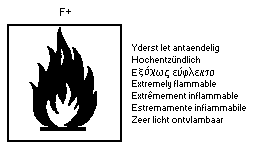
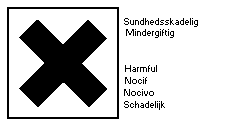 The United Nations Committee of Experts on the Transportation of
Dangerous Goods classifies 1,2-dichloroethane as a flammable liquid
(Class 3) and a poisonous substance (Subsidiary Risk Class 6.1)
presenting medium danger for packing purposes (Packing Group II).
Packing methods and labels are recommended (effective date: 1982 (r)).
The recommended labels are:
The United Nations Committee of Experts on the Transportation of
Dangerous Goods classifies 1,2-dichloroethane as a flammable liquid
(Class 3) and a poisonous substance (Subsidiary Risk Class 6.1)
presenting medium danger for packing purposes (Packing Group II).
Packing methods and labels are recommended (effective date: 1982 (r)).
The recommended labels are:

 The International Maritime Organization classifies 1,2-dichloroethane
as a flammable liquid (Class 3.1) with medium danger for packing
purposes (Packing Group II) (effective date: 1982 (r)).
7.4 Waste Disposal
In the Federal Republic of Germany, the air emissions of organic
compounds in Class I, which include 1,2-dichloroethane, must not
exceed (as the sum of all compounds in this class) a mass
concentration of 20mg/m3 at a mass flow of 0.1 kg/h. If compounds
of different classes are present, the mass concentration must not
exceed 30 mg/m3 (effective date: 1982 (r)).
In the USA, any solid waste (except domestic) that contains
chlorinated ethanes must be listed as hazardous waste (subject to
handling, transport, treatment, storage, and disposal regulations, and
permit and notification requirements), unless it is found that the
waste cannot pose a threat to human health or the environment when
improperly managed. Certain specified industrial solid wastes
containing 1,2-dichloroethane are identified as hazardous wastes. If
1,2-dichloroethane is a commercial chemical product, it is identified
as a "toxic waste", subject to handling, transport, treatment,
storage, and disposal regulations, and permit notification
requirements (effective date: 1983 (r)). An owner or operator of a
hazardous waste incinerator must achieve 99.99% destruction and
removal efficiency for the substance, if it is designated as a
principal organic hazardous constituent in its EPA permit (effective
date: 1981). Permits are required for the discharge of
1,2-dichloroethane from any point source into USA waters (effective
date: 1980). EPA is required to set, or has set, effluent limitations
and pretreatment standards for 21 major industries, regarding
chlorinated ethanes (effective date: 1983 (r)).
BIBLIOGRAPHY
ACGIH (1986) Documentation of the threshold limit values and
biological exposure indices. Cincinnati, American Conference of
Governmental Industrial Hygienists.
ACGIH (1989) Threshold limit values and biological exposure indices
for 1989-1990. Cincinnati, American Conference of Governmental
Industrial Hygienists.
CLAYTON, G.D. & CLAYTON, F.E. (1981) Patty's industrial hygiene and
toxicology. Vol. 2 C. New York, Wiley-Interscience, John Wiley &
Sons.
GOSSELIN, R.E., HODGE, H.C., SMITH R.P., & GLEASON, M.N. (1976)
Clinical toxicology of commercial products. 4th ed. Baltimore,
Maryland, The Williams and Wilkins Company.
HANDLING CHEMICALS SAFELY (1989) Handling chemicals safely. 2nd ed.
Dutch Association of Safety Experts, Dutch Chemical Industry
Association, Dutch Safety Institute.
IRPTC (1988) Data profile (legal file). Geneva, International
Register of Potentially Toxic Chemicals.
SAX, N.I. (1984) Dangerous properties of industrial materials. New
York, Van Nostrand Reinhold Company.
US NIOSH (1976) A guide to industrial respiratory protection. 3 Vol.
Cincinnati, Ohio, US National Institute for Occupational Safety and
Health. Occupational Safety and Heath Administration,
US NIOSH/OSHA (1981) Occupational health guidelines for chemical
hazards. 3 Vol. Washington, DC, US Department of Health and Human
Services, US Department of Labor (Publication No. DHHS(NIOSH) 01-123).
US NIOSH/OSHA (1985) Pocket guide to chemical hazards. Washington
DC, US National Institute for Occupational Safety and Health,
Occupational Safety and Heath Administration (Publication No. 85.114).
WHO (1987) Environmental Health Criteria 62: 1,2-Dichloroethane.
Geneva, World Health Organization.
The International Maritime Organization classifies 1,2-dichloroethane
as a flammable liquid (Class 3.1) with medium danger for packing
purposes (Packing Group II) (effective date: 1982 (r)).
7.4 Waste Disposal
In the Federal Republic of Germany, the air emissions of organic
compounds in Class I, which include 1,2-dichloroethane, must not
exceed (as the sum of all compounds in this class) a mass
concentration of 20mg/m3 at a mass flow of 0.1 kg/h. If compounds
of different classes are present, the mass concentration must not
exceed 30 mg/m3 (effective date: 1982 (r)).
In the USA, any solid waste (except domestic) that contains
chlorinated ethanes must be listed as hazardous waste (subject to
handling, transport, treatment, storage, and disposal regulations, and
permit and notification requirements), unless it is found that the
waste cannot pose a threat to human health or the environment when
improperly managed. Certain specified industrial solid wastes
containing 1,2-dichloroethane are identified as hazardous wastes. If
1,2-dichloroethane is a commercial chemical product, it is identified
as a "toxic waste", subject to handling, transport, treatment,
storage, and disposal regulations, and permit notification
requirements (effective date: 1983 (r)). An owner or operator of a
hazardous waste incinerator must achieve 99.99% destruction and
removal efficiency for the substance, if it is designated as a
principal organic hazardous constituent in its EPA permit (effective
date: 1981). Permits are required for the discharge of
1,2-dichloroethane from any point source into USA waters (effective
date: 1980). EPA is required to set, or has set, effluent limitations
and pretreatment standards for 21 major industries, regarding
chlorinated ethanes (effective date: 1983 (r)).
BIBLIOGRAPHY
ACGIH (1986) Documentation of the threshold limit values and
biological exposure indices. Cincinnati, American Conference of
Governmental Industrial Hygienists.
ACGIH (1989) Threshold limit values and biological exposure indices
for 1989-1990. Cincinnati, American Conference of Governmental
Industrial Hygienists.
CLAYTON, G.D. & CLAYTON, F.E. (1981) Patty's industrial hygiene and
toxicology. Vol. 2 C. New York, Wiley-Interscience, John Wiley &
Sons.
GOSSELIN, R.E., HODGE, H.C., SMITH R.P., & GLEASON, M.N. (1976)
Clinical toxicology of commercial products. 4th ed. Baltimore,
Maryland, The Williams and Wilkins Company.
HANDLING CHEMICALS SAFELY (1989) Handling chemicals safely. 2nd ed.
Dutch Association of Safety Experts, Dutch Chemical Industry
Association, Dutch Safety Institute.
IRPTC (1988) Data profile (legal file). Geneva, International
Register of Potentially Toxic Chemicals.
SAX, N.I. (1984) Dangerous properties of industrial materials. New
York, Van Nostrand Reinhold Company.
US NIOSH (1976) A guide to industrial respiratory protection. 3 Vol.
Cincinnati, Ohio, US National Institute for Occupational Safety and
Health. Occupational Safety and Heath Administration,
US NIOSH/OSHA (1981) Occupational health guidelines for chemical
hazards. 3 Vol. Washington, DC, US Department of Health and Human
Services, US Department of Labor (Publication No. DHHS(NIOSH) 01-123).
US NIOSH/OSHA (1985) Pocket guide to chemical hazards. Washington
DC, US National Institute for Occupational Safety and Health,
Occupational Safety and Heath Administration (Publication No. 85.114).
WHO (1987) Environmental Health Criteria 62: 1,2-Dichloroethane.
Geneva, World Health Organization.

 The United Nations Committee of Experts on the Transportation of
Dangerous Goods classifies 1,2-dichloroethane as a flammable liquid
(Class 3) and a poisonous substance (Subsidiary Risk Class 6.1)
presenting medium danger for packing purposes (Packing Group II).
Packing methods and labels are recommended (effective date: 1982 (r)).
The recommended labels are:
The United Nations Committee of Experts on the Transportation of
Dangerous Goods classifies 1,2-dichloroethane as a flammable liquid
(Class 3) and a poisonous substance (Subsidiary Risk Class 6.1)
presenting medium danger for packing purposes (Packing Group II).
Packing methods and labels are recommended (effective date: 1982 (r)).
The recommended labels are:

 The International Maritime Organization classifies 1,2-dichloroethane
as a flammable liquid (Class 3.1) with medium danger for packing
purposes (Packing Group II) (effective date: 1982 (r)).
7.4 Waste Disposal
In the Federal Republic of Germany, the air emissions of organic
compounds in Class I, which include 1,2-dichloroethane, must not
exceed (as the sum of all compounds in this class) a mass
concentration of 20mg/m3 at a mass flow of 0.1 kg/h. If compounds
of different classes are present, the mass concentration must not
exceed 30 mg/m3 (effective date: 1982 (r)).
In the USA, any solid waste (except domestic) that contains
chlorinated ethanes must be listed as hazardous waste (subject to
handling, transport, treatment, storage, and disposal regulations, and
permit and notification requirements), unless it is found that the
waste cannot pose a threat to human health or the environment when
improperly managed. Certain specified industrial solid wastes
containing 1,2-dichloroethane are identified as hazardous wastes. If
1,2-dichloroethane is a commercial chemical product, it is identified
as a "toxic waste", subject to handling, transport, treatment,
storage, and disposal regulations, and permit notification
requirements (effective date: 1983 (r)). An owner or operator of a
hazardous waste incinerator must achieve 99.99% destruction and
removal efficiency for the substance, if it is designated as a
principal organic hazardous constituent in its EPA permit (effective
date: 1981). Permits are required for the discharge of
1,2-dichloroethane from any point source into USA waters (effective
date: 1980). EPA is required to set, or has set, effluent limitations
and pretreatment standards for 21 major industries, regarding
chlorinated ethanes (effective date: 1983 (r)).
BIBLIOGRAPHY
ACGIH (1986) Documentation of the threshold limit values and
biological exposure indices. Cincinnati, American Conference of
Governmental Industrial Hygienists.
ACGIH (1989) Threshold limit values and biological exposure indices
for 1989-1990. Cincinnati, American Conference of Governmental
Industrial Hygienists.
CLAYTON, G.D. & CLAYTON, F.E. (1981) Patty's industrial hygiene and
toxicology. Vol. 2 C. New York, Wiley-Interscience, John Wiley &
Sons.
GOSSELIN, R.E., HODGE, H.C., SMITH R.P., & GLEASON, M.N. (1976)
Clinical toxicology of commercial products. 4th ed. Baltimore,
Maryland, The Williams and Wilkins Company.
HANDLING CHEMICALS SAFELY (1989) Handling chemicals safely. 2nd ed.
Dutch Association of Safety Experts, Dutch Chemical Industry
Association, Dutch Safety Institute.
IRPTC (1988) Data profile (legal file). Geneva, International
Register of Potentially Toxic Chemicals.
SAX, N.I. (1984) Dangerous properties of industrial materials. New
York, Van Nostrand Reinhold Company.
US NIOSH (1976) A guide to industrial respiratory protection. 3 Vol.
Cincinnati, Ohio, US National Institute for Occupational Safety and
Health. Occupational Safety and Heath Administration,
US NIOSH/OSHA (1981) Occupational health guidelines for chemical
hazards. 3 Vol. Washington, DC, US Department of Health and Human
Services, US Department of Labor (Publication No. DHHS(NIOSH) 01-123).
US NIOSH/OSHA (1985) Pocket guide to chemical hazards. Washington
DC, US National Institute for Occupational Safety and Health,
Occupational Safety and Heath Administration (Publication No. 85.114).
WHO (1987) Environmental Health Criteria 62: 1,2-Dichloroethane.
Geneva, World Health Organization.
The International Maritime Organization classifies 1,2-dichloroethane
as a flammable liquid (Class 3.1) with medium danger for packing
purposes (Packing Group II) (effective date: 1982 (r)).
7.4 Waste Disposal
In the Federal Republic of Germany, the air emissions of organic
compounds in Class I, which include 1,2-dichloroethane, must not
exceed (as the sum of all compounds in this class) a mass
concentration of 20mg/m3 at a mass flow of 0.1 kg/h. If compounds
of different classes are present, the mass concentration must not
exceed 30 mg/m3 (effective date: 1982 (r)).
In the USA, any solid waste (except domestic) that contains
chlorinated ethanes must be listed as hazardous waste (subject to
handling, transport, treatment, storage, and disposal regulations, and
permit and notification requirements), unless it is found that the
waste cannot pose a threat to human health or the environment when
improperly managed. Certain specified industrial solid wastes
containing 1,2-dichloroethane are identified as hazardous wastes. If
1,2-dichloroethane is a commercial chemical product, it is identified
as a "toxic waste", subject to handling, transport, treatment,
storage, and disposal regulations, and permit notification
requirements (effective date: 1983 (r)). An owner or operator of a
hazardous waste incinerator must achieve 99.99% destruction and
removal efficiency for the substance, if it is designated as a
principal organic hazardous constituent in its EPA permit (effective
date: 1981). Permits are required for the discharge of
1,2-dichloroethane from any point source into USA waters (effective
date: 1980). EPA is required to set, or has set, effluent limitations
and pretreatment standards for 21 major industries, regarding
chlorinated ethanes (effective date: 1983 (r)).
BIBLIOGRAPHY
ACGIH (1986) Documentation of the threshold limit values and
biological exposure indices. Cincinnati, American Conference of
Governmental Industrial Hygienists.
ACGIH (1989) Threshold limit values and biological exposure indices
for 1989-1990. Cincinnati, American Conference of Governmental
Industrial Hygienists.
CLAYTON, G.D. & CLAYTON, F.E. (1981) Patty's industrial hygiene and
toxicology. Vol. 2 C. New York, Wiley-Interscience, John Wiley &
Sons.
GOSSELIN, R.E., HODGE, H.C., SMITH R.P., & GLEASON, M.N. (1976)
Clinical toxicology of commercial products. 4th ed. Baltimore,
Maryland, The Williams and Wilkins Company.
HANDLING CHEMICALS SAFELY (1989) Handling chemicals safely. 2nd ed.
Dutch Association of Safety Experts, Dutch Chemical Industry
Association, Dutch Safety Institute.
IRPTC (1988) Data profile (legal file). Geneva, International
Register of Potentially Toxic Chemicals.
SAX, N.I. (1984) Dangerous properties of industrial materials. New
York, Van Nostrand Reinhold Company.
US NIOSH (1976) A guide to industrial respiratory protection. 3 Vol.
Cincinnati, Ohio, US National Institute for Occupational Safety and
Health. Occupational Safety and Heath Administration,
US NIOSH/OSHA (1981) Occupational health guidelines for chemical
hazards. 3 Vol. Washington, DC, US Department of Health and Human
Services, US Department of Labor (Publication No. DHHS(NIOSH) 01-123).
US NIOSH/OSHA (1985) Pocket guide to chemical hazards. Washington
DC, US National Institute for Occupational Safety and Health,
Occupational Safety and Heath Administration (Publication No. 85.114).
WHO (1987) Environmental Health Criteria 62: 1,2-Dichloroethane.
Geneva, World Health Organization.
