
FAO Nutrition Meetings
Report Series No. 40A,B,C
WHO/Food Add./67.29
TOXICOLOGICAL EVALUATION OF SOME
ANTIMICROBIALS, ANTIOXIDANTS, EMULSIFIERS,
STABILIZERS, FLOUR-TREATMENT AGENTS, ACIDS AND BASES
The content of this document is the result of the deliberations of the
Joint FAO/WHO Expert Committee on Food Additives which met at Rome,
13-20 December, 19651 Geneva, 11-18 October, 19662
1 Ninth Report of the Joint FAO/WHO Expert Committee on Food
Additives, FAO Nutrition Meetings Report Series, 1966 No. 40;
Wld Hlth Org. techn. Rep. Ser., 1966, 339
2 Tenth Report of the Joint FAO/WHO Expert Committee on Food
Additives, FAO Nutrition Meetings Report Series, 1967, in press;
Food and Agriculture Organization of the United Nations
World Health Organization
1967
CHOLIC ACID
Chemical name 3,7,12-trihydroxycholanic acid
Empirical formula C24H40O5
Structural formula
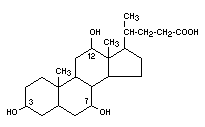 Molecular weight 408.58
Definition Cholic acid after drying contains not
less than 98 per cent. C24H40O5
Description Colourless plates or a white,
crystalline powder having a bitter taste
with a sweetish after-taste.
Uses As a foam stabilizer.
Biological Data
(See Desoxycholic Acid)
DESOXYCHOLIC ACID
Chemical name 3,12-dihydroxycholanic acid
Empirical formula C24H40O4
Structural formula
Molecular weight 408.58
Definition Cholic acid after drying contains not
less than 98 per cent. C24H40O5
Description Colourless plates or a white,
crystalline powder having a bitter taste
with a sweetish after-taste.
Uses As a foam stabilizer.
Biological Data
(See Desoxycholic Acid)
DESOXYCHOLIC ACID
Chemical name 3,12-dihydroxycholanic acid
Empirical formula C24H40O4
Structural formula
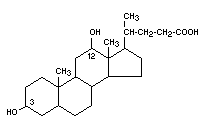 Molecular weight 392.56
Definition Desoxycholic acid after drying contains
not less than 98 per cent. and not more
than the equivalent of 102 per cent.
C24H40O4
Description White, crystalline powder
Uses As foam stabilizer
Biological Data
Biochemical aspects
These bile acids and their salts are found as natural
constituents of the bile. The nucleus of the bile acids is closely
related to cholesterol, from which they are formed in the liver, and
this conversion depends on their relative concentrations. Between 90
and 95 per cent. of bile acids are reabsorbed, mainly from the lower
half of the small intestine, and undergo enterohepatic circulation;
small quantities occur in the stools and very little is normally
excreted in the urine. Bile salts affect the absorption of fats,
fat-soluble vitamins, and various ions. In normal individuals,
additional administration of moderate quantities of bile acids or
salts by mouth has no demonstrable effect, since there are enough bile
salts present in the intestinal lumen to carry out all the absorptive
functions. If there is a deficiency of bile salts, administration may
be beneficial. Bile salts stimulate excretion of bile and hence tend
to hasten their own elimination. Bile salts may also cause some
stimulation of intestinal movement.
The daily output from a fistula in man ranged from 1-2.3 g daily;
when stimulated by bile, the daily output was increased fourfold
(Josephson, 1941).
Acute toxicity
Animal Route LD50 References
(mg/kg
body-weight)
CHOLIC ACID
Rabbit i.v. 50 (Na salt) Gillert, 1926
DESOXYCHOLIC ACID
Rabbit i.v. 15 (Na salt) Gillert, 1926
In general, bile acids and salts have only a minor toxic
potential when given by mouth. In large doses, they are likely to have
the same effects as saponins; the main action is likely to be
irritation of mucous membranes. Parenterally they are much more toxic
and may cause haemolysis, a digitalis-like action on the heart and
effects on the central nervous system. Toxic effects that may be
attributable to accumulation of bile acids or salts are also seen in
obstructive jaundice, since they disappear if cholestyramine, which
adsorbs bile acids in the intestinal lumen and prevents their
reabsorption, is administered (van Itallie et al., 1961).
Short-term studies
Man. Two grams of ox bile salts were administered daily to a
child of 5 years who had a congenital deficiency of bile salts, for a
period of 11 months with consequent improvement in fat absorption and
no evidence of ill effects (Rose et al., 1955).
Comments
No long-term animal studies are reported. They are not necessary,
since every healthy person circulates about 1 g of bile salts or more
daily throughout life. Adequate specifications are needed to ensure
that the substances used closely conform to the naturally occurring
bile salts.
Evaluation
Because of their bitter taste, bile acids and their salts tend to
be limited in use.
A 5 per cent. variation in the daily output of bile is equivalent
to 75 mg bile salts daily (equivalent to 1.25 mg/kg body-weight/day).
This amount is unlikely to affect the normal equilibrium of bile acid
metabolism.
Estimate of acceptable daily intake for man
mg/kg body-weight
Unconditional acceptance 0-1.25
REFERENCES
Gillert, E. (1926) Z. Ges. exp. Med., 52, 779
van Itallie, T. B., Hashim, S. A., Crampton, R. S. & Tennent, D. M.
(1961) New England J. Med., 265, 469
Josephson, B. (1941) Physiol. Rev., 21, 435
Ross, C. A. C., Frazer, A. C., French, J. M., Gerrard, J. W., Sammons,
H. G. & Smellie, J. M. (l955) Lancet, i, 1087
Molecular weight 392.56
Definition Desoxycholic acid after drying contains
not less than 98 per cent. and not more
than the equivalent of 102 per cent.
C24H40O4
Description White, crystalline powder
Uses As foam stabilizer
Biological Data
Biochemical aspects
These bile acids and their salts are found as natural
constituents of the bile. The nucleus of the bile acids is closely
related to cholesterol, from which they are formed in the liver, and
this conversion depends on their relative concentrations. Between 90
and 95 per cent. of bile acids are reabsorbed, mainly from the lower
half of the small intestine, and undergo enterohepatic circulation;
small quantities occur in the stools and very little is normally
excreted in the urine. Bile salts affect the absorption of fats,
fat-soluble vitamins, and various ions. In normal individuals,
additional administration of moderate quantities of bile acids or
salts by mouth has no demonstrable effect, since there are enough bile
salts present in the intestinal lumen to carry out all the absorptive
functions. If there is a deficiency of bile salts, administration may
be beneficial. Bile salts stimulate excretion of bile and hence tend
to hasten their own elimination. Bile salts may also cause some
stimulation of intestinal movement.
The daily output from a fistula in man ranged from 1-2.3 g daily;
when stimulated by bile, the daily output was increased fourfold
(Josephson, 1941).
Acute toxicity
Animal Route LD50 References
(mg/kg
body-weight)
CHOLIC ACID
Rabbit i.v. 50 (Na salt) Gillert, 1926
DESOXYCHOLIC ACID
Rabbit i.v. 15 (Na salt) Gillert, 1926
In general, bile acids and salts have only a minor toxic
potential when given by mouth. In large doses, they are likely to have
the same effects as saponins; the main action is likely to be
irritation of mucous membranes. Parenterally they are much more toxic
and may cause haemolysis, a digitalis-like action on the heart and
effects on the central nervous system. Toxic effects that may be
attributable to accumulation of bile acids or salts are also seen in
obstructive jaundice, since they disappear if cholestyramine, which
adsorbs bile acids in the intestinal lumen and prevents their
reabsorption, is administered (van Itallie et al., 1961).
Short-term studies
Man. Two grams of ox bile salts were administered daily to a
child of 5 years who had a congenital deficiency of bile salts, for a
period of 11 months with consequent improvement in fat absorption and
no evidence of ill effects (Rose et al., 1955).
Comments
No long-term animal studies are reported. They are not necessary,
since every healthy person circulates about 1 g of bile salts or more
daily throughout life. Adequate specifications are needed to ensure
that the substances used closely conform to the naturally occurring
bile salts.
Evaluation
Because of their bitter taste, bile acids and their salts tend to
be limited in use.
A 5 per cent. variation in the daily output of bile is equivalent
to 75 mg bile salts daily (equivalent to 1.25 mg/kg body-weight/day).
This amount is unlikely to affect the normal equilibrium of bile acid
metabolism.
Estimate of acceptable daily intake for man
mg/kg body-weight
Unconditional acceptance 0-1.25
REFERENCES
Gillert, E. (1926) Z. Ges. exp. Med., 52, 779
van Itallie, T. B., Hashim, S. A., Crampton, R. S. & Tennent, D. M.
(1961) New England J. Med., 265, 469
Josephson, B. (1941) Physiol. Rev., 21, 435
Ross, C. A. C., Frazer, A. C., French, J. M., Gerrard, J. W., Sammons,
H. G. & Smellie, J. M. (l955) Lancet, i, 1087
 Molecular weight 408.58
Definition Cholic acid after drying contains not
less than 98 per cent. C24H40O5
Description Colourless plates or a white,
crystalline powder having a bitter taste
with a sweetish after-taste.
Uses As a foam stabilizer.
Biological Data
(See Desoxycholic Acid)
DESOXYCHOLIC ACID
Chemical name 3,12-dihydroxycholanic acid
Empirical formula C24H40O4
Structural formula
Molecular weight 408.58
Definition Cholic acid after drying contains not
less than 98 per cent. C24H40O5
Description Colourless plates or a white,
crystalline powder having a bitter taste
with a sweetish after-taste.
Uses As a foam stabilizer.
Biological Data
(See Desoxycholic Acid)
DESOXYCHOLIC ACID
Chemical name 3,12-dihydroxycholanic acid
Empirical formula C24H40O4
Structural formula
 Molecular weight 392.56
Definition Desoxycholic acid after drying contains
not less than 98 per cent. and not more
than the equivalent of 102 per cent.
C24H40O4
Description White, crystalline powder
Uses As foam stabilizer
Biological Data
Biochemical aspects
These bile acids and their salts are found as natural
constituents of the bile. The nucleus of the bile acids is closely
related to cholesterol, from which they are formed in the liver, and
this conversion depends on their relative concentrations. Between 90
and 95 per cent. of bile acids are reabsorbed, mainly from the lower
half of the small intestine, and undergo enterohepatic circulation;
small quantities occur in the stools and very little is normally
excreted in the urine. Bile salts affect the absorption of fats,
fat-soluble vitamins, and various ions. In normal individuals,
additional administration of moderate quantities of bile acids or
salts by mouth has no demonstrable effect, since there are enough bile
salts present in the intestinal lumen to carry out all the absorptive
functions. If there is a deficiency of bile salts, administration may
be beneficial. Bile salts stimulate excretion of bile and hence tend
to hasten their own elimination. Bile salts may also cause some
stimulation of intestinal movement.
The daily output from a fistula in man ranged from 1-2.3 g daily;
when stimulated by bile, the daily output was increased fourfold
(Josephson, 1941).
Acute toxicity
Animal Route LD50 References
(mg/kg
body-weight)
CHOLIC ACID
Rabbit i.v. 50 (Na salt) Gillert, 1926
DESOXYCHOLIC ACID
Rabbit i.v. 15 (Na salt) Gillert, 1926
In general, bile acids and salts have only a minor toxic
potential when given by mouth. In large doses, they are likely to have
the same effects as saponins; the main action is likely to be
irritation of mucous membranes. Parenterally they are much more toxic
and may cause haemolysis, a digitalis-like action on the heart and
effects on the central nervous system. Toxic effects that may be
attributable to accumulation of bile acids or salts are also seen in
obstructive jaundice, since they disappear if cholestyramine, which
adsorbs bile acids in the intestinal lumen and prevents their
reabsorption, is administered (van Itallie et al., 1961).
Short-term studies
Man. Two grams of ox bile salts were administered daily to a
child of 5 years who had a congenital deficiency of bile salts, for a
period of 11 months with consequent improvement in fat absorption and
no evidence of ill effects (Rose et al., 1955).
Comments
No long-term animal studies are reported. They are not necessary,
since every healthy person circulates about 1 g of bile salts or more
daily throughout life. Adequate specifications are needed to ensure
that the substances used closely conform to the naturally occurring
bile salts.
Evaluation
Because of their bitter taste, bile acids and their salts tend to
be limited in use.
A 5 per cent. variation in the daily output of bile is equivalent
to 75 mg bile salts daily (equivalent to 1.25 mg/kg body-weight/day).
This amount is unlikely to affect the normal equilibrium of bile acid
metabolism.
Estimate of acceptable daily intake for man
mg/kg body-weight
Unconditional acceptance 0-1.25
REFERENCES
Gillert, E. (1926) Z. Ges. exp. Med., 52, 779
van Itallie, T. B., Hashim, S. A., Crampton, R. S. & Tennent, D. M.
(1961) New England J. Med., 265, 469
Josephson, B. (1941) Physiol. Rev., 21, 435
Ross, C. A. C., Frazer, A. C., French, J. M., Gerrard, J. W., Sammons,
H. G. & Smellie, J. M. (l955) Lancet, i, 1087
Molecular weight 392.56
Definition Desoxycholic acid after drying contains
not less than 98 per cent. and not more
than the equivalent of 102 per cent.
C24H40O4
Description White, crystalline powder
Uses As foam stabilizer
Biological Data
Biochemical aspects
These bile acids and their salts are found as natural
constituents of the bile. The nucleus of the bile acids is closely
related to cholesterol, from which they are formed in the liver, and
this conversion depends on their relative concentrations. Between 90
and 95 per cent. of bile acids are reabsorbed, mainly from the lower
half of the small intestine, and undergo enterohepatic circulation;
small quantities occur in the stools and very little is normally
excreted in the urine. Bile salts affect the absorption of fats,
fat-soluble vitamins, and various ions. In normal individuals,
additional administration of moderate quantities of bile acids or
salts by mouth has no demonstrable effect, since there are enough bile
salts present in the intestinal lumen to carry out all the absorptive
functions. If there is a deficiency of bile salts, administration may
be beneficial. Bile salts stimulate excretion of bile and hence tend
to hasten their own elimination. Bile salts may also cause some
stimulation of intestinal movement.
The daily output from a fistula in man ranged from 1-2.3 g daily;
when stimulated by bile, the daily output was increased fourfold
(Josephson, 1941).
Acute toxicity
Animal Route LD50 References
(mg/kg
body-weight)
CHOLIC ACID
Rabbit i.v. 50 (Na salt) Gillert, 1926
DESOXYCHOLIC ACID
Rabbit i.v. 15 (Na salt) Gillert, 1926
In general, bile acids and salts have only a minor toxic
potential when given by mouth. In large doses, they are likely to have
the same effects as saponins; the main action is likely to be
irritation of mucous membranes. Parenterally they are much more toxic
and may cause haemolysis, a digitalis-like action on the heart and
effects on the central nervous system. Toxic effects that may be
attributable to accumulation of bile acids or salts are also seen in
obstructive jaundice, since they disappear if cholestyramine, which
adsorbs bile acids in the intestinal lumen and prevents their
reabsorption, is administered (van Itallie et al., 1961).
Short-term studies
Man. Two grams of ox bile salts were administered daily to a
child of 5 years who had a congenital deficiency of bile salts, for a
period of 11 months with consequent improvement in fat absorption and
no evidence of ill effects (Rose et al., 1955).
Comments
No long-term animal studies are reported. They are not necessary,
since every healthy person circulates about 1 g of bile salts or more
daily throughout life. Adequate specifications are needed to ensure
that the substances used closely conform to the naturally occurring
bile salts.
Evaluation
Because of their bitter taste, bile acids and their salts tend to
be limited in use.
A 5 per cent. variation in the daily output of bile is equivalent
to 75 mg bile salts daily (equivalent to 1.25 mg/kg body-weight/day).
This amount is unlikely to affect the normal equilibrium of bile acid
metabolism.
Estimate of acceptable daily intake for man
mg/kg body-weight
Unconditional acceptance 0-1.25
REFERENCES
Gillert, E. (1926) Z. Ges. exp. Med., 52, 779
van Itallie, T. B., Hashim, S. A., Crampton, R. S. & Tennent, D. M.
(1961) New England J. Med., 265, 469
Josephson, B. (1941) Physiol. Rev., 21, 435
Ross, C. A. C., Frazer, A. C., French, J. M., Gerrard, J. W., Sammons,
H. G. & Smellie, J. M. (l955) Lancet, i, 1087
