
Pesticide residues in food - 2003 - Joint FAO/WHO Meeting on Pesticide Residues
First draft prepared by
D. Andrew, R. Shillaker & I. Dewhurst
Pesticides Safety Directorate, Department for Environment,
Food and Rural Affairs, York, England
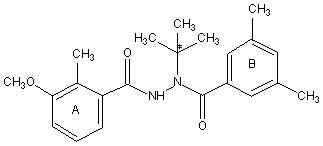
Methoxyfenozide (N-tert-butyl-N’-(3-methoxy-o-toluoy)-3,5-xylohydrazide) is a dia-cylhydrazine insecticide that acts as an ecdysone agonist. Methoxyfenozide has not been evaluated previously by the JMPR. The Meeting noted that the purity of the material tested (>98%) was higher than that of the material proposed for commercialization (97%), but concluded that the findings were applicable to the proposed technical specification and production material.
The studies of toxicity with methoxyfenozide were performed between 1994 and 1998. The purity of the methoxyfenozide used in the majority of studies (98-99.2%) was higher than that of the proposed technical specification (purity, 97%). Data supplied confirmed that the specifications of the main toxicity batch were consistent with those of the production material. Overall, the Meeting concluded that the material tested was adequately representative of the production material.
The toxicokinetics and metabolism of methoxyfenozide in rats were investigated using the following 14C-labelled test materials: (i) [methoxyphenyl ring uniform label-14C]methoxyfenozide ("A-ring label"), radiochemical purity, 98.2%; (ii) S[dimethylphenyl ring uniform label-14C]methoxyfenozide ("B-ring label"), radiochemical purity, 99.0%; (iii) t-butyl [14C]methoxyfenozide ("t label"), radiochemical purity, 96.4%.
Rats
In a study of absorption, distribution, metabolism, and excretion that was designed to comply with the pesticide assessment guidelines of the United States Environmental Protection Agency (EPA) and Japan and that complied with good laboratory practice (GLP), forty-two groups of Sprague-Dawley rats (up to five animals of each sex per group) were given either A-ring, B-ring or t-butyl-labelled [14C]methoxyfenozide at a nominal dose of 10 or 1000 mg of active substance/kg bw, by oral gavage. The test materials were prepared as suspensions in 0.5% aqueous methylcellulose. The [14C]methoxyfenozide was combined with appropriate amounts of unlabelled (12C) methoxyfenozide for dilution of radioactivity at the highest dose and with 13C-labelled (A-ring carbonyl; B-ring methyl; t-butyl carbon) methoxyfenozide for identification of metabolites.
Three basic experimental designs were used: one to determine the excretion, distribution, and mass balance of radioactivity (120 h after dosing), one to evaluate the pharmacokinetics of radioactivity in the blood (to determine the time to Cmax and ½ Cmax), and one to determine the tissue distribution of radioactivity at Cmax, ½ Cmax and 5 days after dosing. Biliary excretion was investigated in bile-duct cannulated rats given a dose of 10 mg/kg bw by gavage. In addition to the single dose experiments, one group of animals received diets containing unlabelled methoxyfenozide at a concentration of 200 mg/kg (equivalent to 20 mg/kg bw per day) for 2 weeks before receiving a single oral dose of [14C-A-ring]methoxyfenozide at 10 mg/kg. Another group received five daily oral doses of [14C-A-ring]methoxyfenozide at 10 mg/kg by gavage. The amount of radiolabel contained in exhaled air was determined using activated charcoal and sodium hydroxide traps and liquid scintillation counting (LSC) after appropriate sample preparation.

Figure 1. Structure of methoxyfenozide
The excretion of radiolabel after a single oral dose of A-ring, B-ring or t-butyl-labelled methoxyfenozide followed a similar pattern, regardless of dose (representative data for A-ring-labelled material are presented in Tables 1-3). For each dose, the results for the excretion, pharmacokinetics and tissue distribution of radioactivity were generally comparable for males and females. The overall recovery of 14C ranged from 97% to 104% of the administered dose for all experiments, including the biliary excretion experiment. Most of the radiolabel was excreted during the first 24 h after administration of a single oral dose, primarily in the faeces, which contained 58-77% of the administered dose at day 1 (86-97% after 5-7 days). Females consistently excreted a greater proportion of the administered dose in the urine. Minimal amounts of radiolabel (0.10-0.23%) remained in the tissues and carcass at 5 days after a single dose. Less than 0.2% of the administered dose was exhaled as 14CO2. These data indicate that the A-ring and B-ring remain associated during the metabolism of methoxyfenozide. Pre-treatment of animals with diets containing methoxyfenozide for 2 weeks, or with five daily doses of 14C-labelled methoxyfenozide did not appreciably alter absorption or distribution (Table 1).
Table 1. Recovery of A-ring-labelled [14C]methoxyfenozide (%) in rats
|
Dose |
Route |
Sex |
Time of sacrifice |
Recovery of the administered dose (%) |
|||||
|
Urine |
Faeces |
Bile |
Tissues |
Carcass |
Total |
||||
|
1000 |
Oral |
Male |
Day 7 |
4.89 |
89.21 |
— |
— |
0.10 |
94.20 |
|
1000 |
Oral |
Female |
Day 7 |
7.50 |
87.00 |
— |
— |
0.05 |
94.55 |
|
1000 |
Oral |
Male |
Day 5 |
5.33 |
93.49 |
— |
0.07 |
0.09 |
98.98 |
|
1000 |
Oral |
Female |
Day 5 |
8.92 |
89.81 |
— |
0.01 |
0.10 |
98.85 |
|
10 |
Oral |
Male |
Day 5 |
7.06 |
96.81 |
— |
0.08 |
0.06 |
104.01 |
|
10 |
Oral |
Female |
Day 5 |
11.95 |
92.32 |
— |
0.01 |
0.10 |
104.38 |
|
10 |
Oral (bile) |
Male |
72 h |
4.98 |
26.16 |
64 |
0.08 |
0.27 |
95.89 |
|
10 |
Oral (bile) |
Female |
72 h |
22.15 |
35.04 |
38 |
0.12 |
1.34 |
97.38 |
|
10a |
Oral + gavagea |
Male |
Day 5 |
7.62 |
90.92 |
— |
0.16 |
0.07 |
98.76 |
|
10a |
Oral + gavagea |
Female |
Day 5 |
12.17 |
90.01 |
— |
0.01 |
0.06 |
102.26 |
|
10 |
Oralb |
Male |
15 min |
5.00 |
71.54 |
— |
3.30 |
2.07 |
97.75 |
|
10 |
Oralb |
Female |
15 min |
8.47 |
66.30 |
— |
3.46 |
3.24 |
95.44 |
From Watts & Longacre, 1998
a 14 days at 200 mg/kg [12C]methoxyfenozide plus a single dose of [14C-A-ring]methoxyfenozide at 10 mg/kg bw administered by gavage
b Six daily doses administered by gavage
Table 2. Toxicokinetic half-lives (T1/2) for elimination and peak concentration of radiolabel in plasma in rats given a single oral dose of A-ring-labelled [14 C]methoxyfenozide by gavage
|
Dose |
Sex |
Plasma elimination T1/2 |
alpha-Phase |
beta-Phase |
Peak concentration |
|
1000 |
Male |
9.4 |
0.2 |
24.2 |
27.68 |
|
1000 |
Female |
10.1 |
0.5 |
22.5 |
29.74 |
|
10 |
Male |
5.0 |
0.5 |
26.4 |
0.81 |
|
10 |
Female |
4.6 |
0.2 |
19.6 |
0.59 |
From Watts & Longacre (1998)
Table 3. Mean concentration (pg equivalents/g) of radioactivity in blood, carcass, and tissues in rats given a single oral dose of A-ring-labelled [14 C]methoxyfenozide
|
Tissue |
Cmax(0.25 h) |
1/2 Cmax (2.0 h) |
5 days |
|||
|
Male |
Female |
Male |
Female |
Male |
Female |
|
|
Adrenals |
32.38 |
51.56 |
51.56 |
18.58 |
0.8300 |
0.6631 |
|
Blood |
12.61 |
37.84 |
16.39 |
16.71 |
0.4789 |
0.3530 |
|
Bone marrow |
6.24 |
14.48 |
6.84 |
5.66 |
0.6104 |
1.4821 |
|
Brain |
0.88 |
2.64 |
1.25 |
1.34 |
0.0936 |
0.0404 |
|
Carcass (residual) |
8.53 |
34.61 |
8.97 |
30.10 |
0.9230 |
1.1200 |
|
Fat |
5.30 |
13.22 |
10.38 |
12.31 |
0.4863 |
0.7756 |
|
Heart |
9.89 |
21.96 |
12.23 |
10.50 |
0.2800 |
0.1553 |
|
Intestinal tract |
791.70 |
1294.08 |
1493.84 |
1985.52 |
0.7872 |
1.3753 |
|
Kidneys |
33.77 |
61.40 |
39.28 |
30.61 |
1.3949 |
0.9565 |
|
Liver |
368.12 |
927.28 |
271.26 |
154.92 |
16.5151 |
1.8051 |
|
Lungs |
16.92 |
33.45 |
16.84 |
14.78 |
0.3483 |
0.1300 |
|
Muscle (thigh) |
6.23 |
11.61 |
6.38 |
6.33 |
0.1326 |
0.0605 |
|
Ovaries |
— |
40.20 |
— |
20.07 |
— |
0.2964 |
|
Plasma |
17.34 |
46.38 |
25.23 |
24.56 |
0.0992 |
0.0402 |
|
Spleen |
55.26 |
45.36 |
15.00 |
11.96 |
0.3702 |
0.3895 |
|
Stomach |
6051.39 |
3538.61 |
1547.58 |
4129.65 |
0.3644 |
0.1960 |
|
Testes |
1.55 |
— |
4.89 |
— |
0.0405 |
— |
|
Thyroids |
20.81 |
19.51 |
13.62 |
7.78 |
4.2711 |
2.5416 |
From Watts & Longacre (1998)
The biliary excretion of radiolabel after a single oral dose of [14C-A-ring]methoxyfenozide at 10 mg/kg was investigated in bile-duct cannulated rats (Table 1). Biliary excretion was rapid, with 22% (females) and 50% (males) of the administered dose being excreted within 12 h. Overall, 38% (females) and 64% (males) of the radiolabel was excreted in bile within 72 h. Considerable variability between individual animals was seen in cannulated female rats (bile, 13-55%; and urine, 5-43% within 72 h), but the overall amount absorbed (in bile, urine, carcass and tissues) was similar for all four females (56-67%). Taking the biliary component into account, the overall extent of oral absorption of methoxyfenozide at a dose of 10 mg/kg bw was 60-70% in both sexes.
Toxicokinetic parameters for plasma and blood were investigated after a single dose of [14C]methoxyfenozide (at a lower and higher dose for each labelling position) by gavage. Findings were similar, irrespective of labelling position. [14C]Methoxyfenozide was rapidly absorbed, with maximum concentrations of radioactivity in blood and plasma being observed 15-30min after dosing (Table 2). The maximum concentrations were similar for each radiolabel and sex at each dose, but tended to be slightly higher in males, especially at the lower dose. Peak blood concentrations were not proportional to the dose administered, i.e. peak concentrations in blood and plasma at the highest dose were only 24-68-fold higher than at the lowest dose, which is indicative of saturation of absorption. Values for the area under the curve (AUC) were not calculated. Elimination of radiolabel from the plasma followed a biphasic pattern (Table 2), with secondary peaks indicative of enterohepatic circulation (especially in females). In rats given the test compound at the higher dose, the overall elimination half-life of radiolabel from the plasma was longer than in rats given the lower dose. These results are consistent with saturation of the elimination pathway for parent compound and/or its metabolites in rats at the higher dose in both sexes, with these effects being more prominent in females.
The tissue distribution of radioactivity was investigated after a single dose of A-ring-labelled or t-labelled methoxyfenozide administered by gavage (at Cmax, ½ Cmax and at 5 days after dosing at 10 or 1000 mg/kg bw), and after a single dose of B-ring-labelled methoxyfenozide (5 days after dosing at 10 mg/kg bw). Tissue distribution was also investigated after dosing with A-ring-labelled methoxyfenozide at 10 mg/kg bw as a pulse dose (5 days after dosing) and as a repeated dose (at 0.25 h after the last dose, at about Cmax).
Similar results were seen in all experiments. The absorbed radioactivity was widely distributed, with the highest concentration of absorbed radioactivity found in the liver at 0.5-2 h after dosing (the higher concentrations found in the stomach and intestinal tract were attributed to largely unabsorbed material). Tissue distributions for the A-ring radiolabel are given in Table 3. Clearance from the body was extensive; 5 days after a single dose of 10 mg/kg bw, the highest percentage of radioactivity, representing <0.1% of the administered dose, was found in the liver (Watts & Longacre, 1998).
A proposed metabolic pathway for methoxyfenozide in rats is shown in Figure 2.
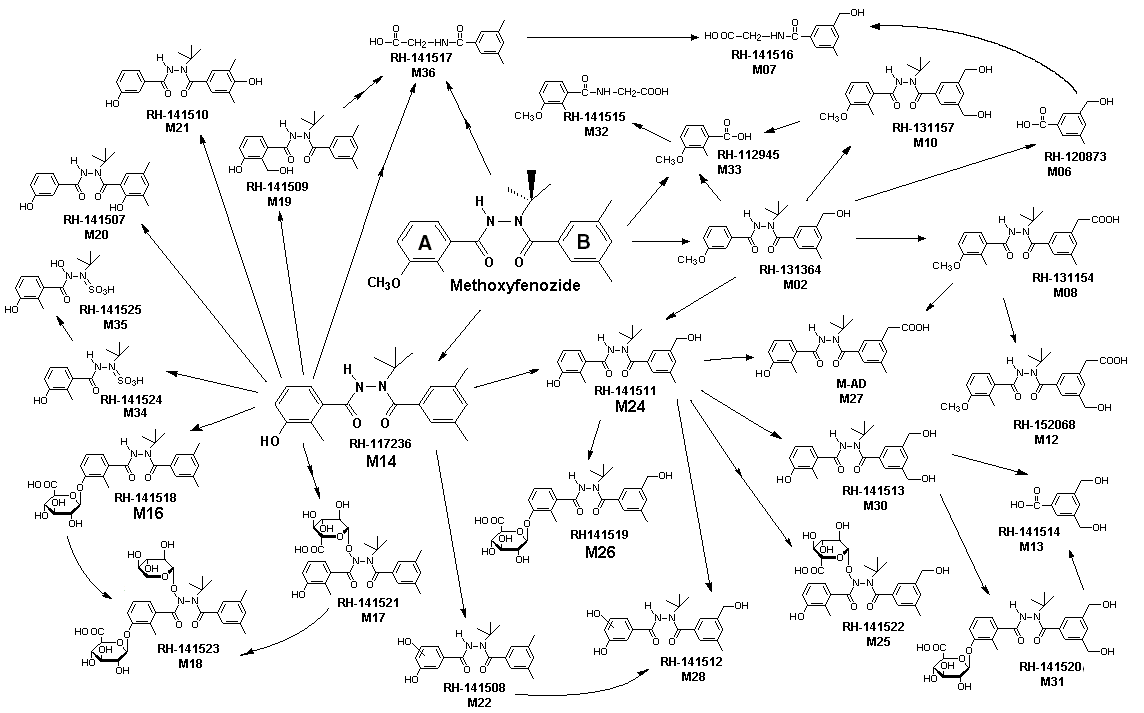
Figure 2. Proposed metabolic pathway for methoxyfenozide in rats
From Watts & Longacre (1998)
M14 and M24 = main faecal metabolites
M16 and M26 (glucuronides of M14 and M24) = main biliary metabolites
Metabolites in the urine, faecal, and bile samples from the study described above were identified and quantified. Most data were obtained for methoxyfenozide labelled with 14C in the A ring and t-butyl group. Urine samples (mostly collected up to 24 h after dosing) were analysed directly by reverse-phase high-performance liquid chromatography (HPLC) and normal-phase thin-layer chromatography (TLC). Faeces (collected up to 24-48 h after dosing) were homogenized, extracted with acetonitrile and then partitioned to yield methylene chloride and aqueous fractions (which were analysed by reverse-phase HPLC and TLC) and post-extraction solids. Bile samples were analysed directly by reverse-phase HPLC and liquid chromatography-mass spectrometry (LC-MS). Metabolites were characterized and identified by TLC, gas chromatography-mass spectrometry (GC-MS) and/or LC-MS and compared with available reference standards. Selected metabolites were also derivatized for structural confirmation.
[14C]Methoxyfenozide was metabolized extensively. A total of 31 metabolites, of which 25 were identified, were isolated from the rat urine and faeces. In addition, 24 metabolites were found in the bile and characterized; of these, 12 were identified (four were unique to bile). Table 4 summarizes the distribution of metabolites in the urine and faeces for methoxyfenozide labelled on the A-ring. Similar patterns were seen with B-ring, and t-butyl-labelled methoxyfenozide.
Table 4. Major metabolites of A-ring-labelled methoxyfenozide in rats (% of administered dose)
|
Metabolite |
Male |
Female |
||||
|
Faeces |
Urine |
Subtotal |
Faeces |
Urine |
Subtotal |
|
|
1000 mg/kg bw |
||||||
|
Active substance |
39.34 |
— |
39.34 |
36.13 |
— |
36.13 |
|
M14 |
14.59 |
0.46 |
15.05 |
15.04 |
2.92 |
17.96 |
|
M22 |
2.95 |
— |
2.95 |
1.43 |
— |
1.43 |
|
M24 |
13.46 |
0.65 |
14.1 |
16.66 |
1.31 |
17.97 |
|
M10 |
4.46 |
0.10 |
4.56 |
5.72 |
0.25 |
5.97 |
|
M28 |
2.54 |
— |
2.54 |
0.31 |
— |
0.31 |
|
M30 |
2.32 |
0.19 |
2.51 |
4.39 |
0.18 |
4.57 |
|
Unknownsb |
0.27 |
0.26 |
0.53 |
— |
0.18 |
0.18 |
|
PES |
8.58 |
NA |
8.58 |
3.65 |
NA |
3.65 |
|
Total % of administered dose identified |
89.38 |
4.13 |
93.51 |
84.45 |
6.17 |
90.62 |
|
10 mg/kg bw |
||||||
|
Active substance |
18.05 |
— |
18.05 |
20.35 |
— |
20.35 |
|
M14 |
23.57 |
0.94 |
24.51 |
26.48 |
1.89 |
28.37 |
|
M22 |
2.51 |
— |
2.51 |
2.58 |
— |
2.58 |
|
M24 |
22.69 |
0.95 |
23.64 |
21.61 |
0.81 |
22.42 |
|
M10 |
3.46 |
— |
3.46 |
5.00 |
0.08 |
5.08 |
|
M28 |
3.30 |
— |
3.30 |
1.01 |
— |
1.01 |
|
M30 |
3.54 |
0.12 |
3.66 |
5.89 |
0.17 |
5.06 |
|
Unknownsb |
1.36 |
0.50 |
1.86 |
— |
0.14 |
0.14 |
|
PES |
14.16 |
NA |
14.16 |
3.40 |
NA |
3.40 |
|
Total % of administered dose identified |
93.42 |
5.85 |
99.27 |
87.11 |
9.04 |
96.15 |
|
Pulsed dose, 10 mg/kg bw |
||||||
|
Active substance |
18.25 |
— |
18.25 |
17.16 |
— |
17.16 |
|
M14 |
10.90 |
0.18 |
11.08 |
29.28 |
2.59 |
31.83 |
|
M22 |
7.17 |
0.12 |
7.29 |
3.06 |
— |
3.06 |
|
M24 |
14.66 |
0.65 |
15.31 |
20.13 |
0.69 |
20.82 |
|
M10 |
3.57 |
— |
3.57 |
3.38 |
0.13 |
3.51 |
|
M28 |
11.96 |
— |
11.96 |
2.25 |
— |
2.25 |
|
M30 |
5.35 |
— |
5.35 |
4.29 |
— |
4.29 |
|
Unknownsb |
— |
0.44 |
0.44 |
— |
— |
— |
|
PES |
10.60 |
NA |
10.60 |
2.17 |
NA |
2.17 |
|
Total % of administered dose identified |
88.54 |
6.33 |
94.87 |
83.61 |
9.06 |
92.67 |
From Watts & Longacre (1998)
NA, Not applicable; PES, Post-extraction solids
Parent compound was found only in the faeces (not in the urine or bile) and comprised 14-26% and 30-39% of the administered dose for animals at the lower and higher doses, respectively, indicating that animals at the lower dose metabolized a greater fraction of the administered dose compared with animals at the higher dose. Seven metabolites (M10, M14, M16, M22, M24, M28, M30) were found to be present in faeces plus urine each at >2% of the administered dose. The predominant metabolites were M14 (desmethylated parent) and M24 (hydroxy methyl derivative). Parent compund plus these seven metabolites accounted for 74-90% of the administered dose (in faeces plus urine) in all groups. For each of these groups, the total of parent plus identified metabolites accounted for >83% of the administered dose, i.e. the metabolic profile of methoxyfenozide in faeces and urine was well defined. Less than 5% of the administered dose was present as metabolites formed from the cleavage of the amide bridge.
Two metabolites, M1 6 (A-ring glucuronide of M14) and M26 (A-ring glucuronide of M24), were the main metabolites in bile. M16 was present at 13% and 18% in males and females respectively, M26 was present at 5% in males and 11% in females, all other metabolites represented <3% of the administered dose. The presence of M16 and M26 at higher concentrations in bile than in faeces indicates that these two metabolites were subject to subsequent hydrolysis.
The primary pathway probably involves demethylation of the A-ring methoxy moiety to form the corresponding phenol (M14), which is conjugated with glucuronic acid to form M16. Hydroxylation on the B-ring methyl moieties is also a significant metabolic pathway. Cleavage of methoxyfenozide to release either of the rings or the t-butyl group is only a minor pathway; none of the cleaved metabolites (M06, M07, M13, M32-36) were present at >2% of the dose. In males, however, cleaved metabolites represented up to about 50% of the metabolites found in urine. There was an indication that males cleaved more of the absorbed dose than did females, on the basis of urinary metabolite patterns.
Results for the animals receiving diets containing methoxyfenozide for 14 days plus a single dose of [14C]methoxyfenozide at 10 mg/kg bw by gavage showed evidence of induction of metabolism. Concentrations of M22, M28 and M30 increased, while concentrations of M14 and M24 were reduced relative to concentrations in animals that received only a single dose of [14C]methoxyfenozide at 10 mg/kg bw (Table 4) (Watts & Longacre, 1998).
The dermal absorption in vivo of methoxyfenozide formulated as an aqueous flow-able liquid (RH-112,485 2F) or as a wettable powder (RH-112,485 280WP) was tested in rats in a study that was designed to comply with US EPA guidelines and GLP. The methoxyfenozide administered was uniformly labelled with 14C on the methoxyphenyl ring; this is acceptable given the limited cleavage seen in studies of oral metabolism. To provide data on exposure to the concentrated product and in-use-dilutions, groups of four male Crl : CD BR rats received radiolabelled methoxyfenozide at three aqueous dilutions (0.025, 0.25, or 2.5% w/v), applied in a volume of 100 µl to a shaved area of about 10 cm2 for 1, 10, or 24 h. Systemically absorbed methoxyfenozide was defined as the radiolabel found in the carcass, urine (plus urine funnel and cage washes), faeces, and whole blood.
For RH-112,485 2F, the total mean recovery of radiolabel in all groups ranged from 98% to 114%. After an exposure of 1, 10, or 24 h to 14C-labelled RH-112,485 2F formulation diluted in water to a concentration of 2.5, 0.25, or 0.025% w/v, a small amount of radiolabel (<1-4%) was systemically absorbed.
For RH-112,485 280 WP, three animals with poor recoveries were excluded from further analysis. The total mean recovery of radiolabel in all groups ranged from 85% to 110%. After an exposure of 1, 10, or 24 h to 14C-labelled RH-112,485 280 WP at a concentration of 2.5, 0.25, or 0.025% w/v, <1-2% of radiolabel was systemically absorbed.
Findings were similar for both formulations. The amount of radiolabel that was systemically absorbed did not increase linearly between the 10-h and 24-h exposure periods, indicating that most of the radiolabel that remained in or on the skin after washing was tightly bound and was not available for systemic absorption. This study shows that methoxyfenozide is poorly absorbed (<4%) after dermal exposure to either of the formulated products or in-use dilutions. The low rate of dermal absorption may be attributed to very low solubility in water (3.3 mg/l at 20°C) (Watts & Frederick, 1998).
The acute toxicity of methoxyfenozide is summarized in Table 5. All studies used methoxyfenozide of 98% purity, and complied with the applicable OECD guideline and GLP. Methoxyfenozide is of low acute toxicity when administered by the oral, dermal and inhalation routes. No specific signs of toxicity were reported.
Table 5. Acute toxicity of methoxyfenozide
|
Species |
Strain |
Sex |
Route |
Vehicle |
LD50 (mg/kg bw) or LC50 |
Purity |
Reference |
|
Rat |
Crl:CD BR |
Males & females |
Oral |
Methyl-cellulose |
>5000 |
98 |
Gingrich & Parno (1995a) |
|
Mouse |
Crl:CD-1(ICR)BR |
Males & females |
Oral |
Methyl-cellulose |
>5000 |
98 |
Procopio & Parno (1995) |
|
Rat |
Crl:CD BR |
Males & females |
Dermal |
Water |
>5000 |
98 |
Parno et al. (1998b) |
|
Rat |
Crl:CD BR |
Males & females |
Inhalation |
None (dust aerosol) |
>4.3mg/l |
98 |
Bernacki & Fergusson (1995) |
MMAD, mass median aerodynamic diameter
(b) Ocular and dermal irritation and dermal sensitization
Methoxyfenozide was not irritating to rabbit skin (Gingrich & Parno, 1995b) and produced only minimal, transient irritation to rabbit eyes (Gingrich & Parno, 1995 c). Negative results were obtained in a Magnusson and Kligman maximization test for skin sensitization in guinea-pigs (Glaza, 1995).
Mice
In an initial study, groups of five female Crl:CD-1(ICR)BR mice were given diets containing methoxyfenozide (purity, 99.2%) at a concentration of 0, 50, 250, 1000, 2500 or 7000 mg/kg for 2 weeks. Limited investigations of clinical chemistry and haematology were performed in addition to routine investigations of mortality, clinical signs, body weight and food consumption. Microscopic examination was performed on the thyroid, liver, spleen, adrenals, kidney and pituitary from animals in the control group and the group receiving the highest dose. In addition, the liver from each mouse in all other treatment groups was also examined microscopically.
No clinical signs of ill health or reaction to treatment after 2 weeks of dietary exposure to methoxyfenozide were recorded. No treatment-related effects on haematology, clinical chemistry, body weight or food consumption were observed. The only findings of note were a statistically significant increase in relative liver weights at 7000 mg/kg and periportal hepatocyte hyperplasia at >2500 mg/kg. The no-observed-adverse-effect level (NOAEL) was 1000 mg/kg (equal to 217 mg/kg per day) on the basis of histopathological changes in the liver at 2500 mg/kg (Kaminski et al., 1993).
In a study that complied with GLP and the essential requirements of OECD guideline 408 (1981), groups of 10 male and 10 female Crl: CD-1(ICR)BR VAF Plus mice were given diets containing methoxyfenozide (purity, 99.2%) at a concentration of 0, 70, 700, 2500 and 7000 mg/kg for 3 months. These doses were equal to mean intakes of 0, 12, 113, 428 and 1149 mg/kg bw per day in males, and 0, 17, 165, 589, 1742 mg/kg bw per day in females. Active substance content, homogeneity and stability were satisfactorily demonstrated. All mice were observed daily for signs of ill health or reaction to treatment. Physical examinations and measurement of body weight and feed consumption were performed weekly. After 13 weeks, mice were bled for haematology (methaemoglobin formation was not investigated) and clinical chemistry analyses and were necropsied. An extensive histopathological examination was conducted for all animals in the control group and in the group receiving the highest dose. In addition, liver, lungs, kidneys and gross lesions from all animals from the other treatment groups were examined.
There were no deaths or substance-related clinical signs of toxicity. There were no significant substance-related effects on body weight or body-weight change in either sex at <2500 mg/kg. Cumulative body-weight gain after 4, 8 and 13 weeks was consistently reduced in both males (by 18-21%) and females (by 15-38%) at the highest dose. There were no notable substance-related haematological effects or changes in any clinical chemistry parameter. A statistically significant, small (4-6%) increase in mean cell volume and mean cell haemoglobin concentration for females at 7000 mg/kg was not considered to be an adverse effect, but might be an indication of a homeostatic response to effects on erythrocytes. There were no effects of toxicological concern on organ weights in either sex at any dose. A statistically significant increase in liver weight relative to body weight was not considered to be of toxicological concern because of its small magnitude (8%); this appeared to be secondary to reduced body weight (only a 3% increase in absolute liver weight) and there was no other evidence of hepatic toxicity in this study. Contrary to the previous 2-week study, no substance-related gross pathological changes or histopathological findings were observed in any tissues. The NOAEL for methoxyfenozide after 3 months of dietary feeding in mice was 2500 mg/kg (equal to 428 and 589 mg/kg bw per day for males and females, respectively) on the basis of reduced body-weight gain at 7000 mg/kg (equal to 1149 mg/kg bw per day) (Kaminski, Shuey & Gillette, 1995a).
Rats
In an initial study, groups of five male and five female Crl: CD®BR rats were given diets containing methoxyfenozide (purity, 90%) at concentrations of up to 20 000 mg/kg for 2 weeks (Table 6).
Table 6. Methoxyfenozide content of diets fed to rats for 13 weeks
|
Dietary concentration (mg/kg) |
Mixing procedure |
Mean intake (mg/kg bw per day) |
|
|
Males |
Females |
||
|
0 |
0 |
0 |
|
|
50 |
Acetone/powder mixing procedure |
5 |
5 |
|
250 |
Acetone/powder mixing procedure |
24 |
25 |
|
1 000 |
Acetone/powder mixing procedure |
101 |
99 |
|
1 000 |
Powder/powder mixing procedure |
99 |
98 |
|
5 000 |
Acetone/powder mixing procedure |
507 |
513 |
|
5 000 |
Powder/powder mixing procedure |
502 |
489 |
|
20 000 |
Acetone/powder mixing procedure |
1977 |
1999 |
From Kyle et al. (1992)
All rats were observed routinely for signs of ill health or reaction to treatment, body weight and feed consumption. After 2 weeks of treatment, blood samples were collected for measurement of haematology (including reticulocytes, but not methaemoglobin formation) and clinical chemistry parameters. Histological examination was limited to seven major organs at the highest dose and controls, except for the liver, thyroid and pituitary where there was evidence of substance-related effects at the highest dose.
There were no deaths and no effects on clinical signs, body weight or feed consumption were evident. No treatment-related adverse haematological effects were observed in either sex at any dose. No clear treatment-related clinical chemistry changes were seen in males at doses of up to 5000 mg/kg or in females at doses of up to 20 000 mg/kg. A treatment-related decrease (by 47%, statistically significant) in triglyceride concentrations was observed in males at 20 000 mg/kg. Increases in absolute and relative weights of liver (14-23%) and adrenals (up to 30%) were noted at >5000 mg/kg. Minimal to slight hepatocyte hypertrophy was observed in all rats at 20 000 mg/kg and in three females at 5000 mg/kg. There was evidence of single cell necrosis in the livers of animals receiving 20 000 mg/kg. A dose-related minimal to slight hypertrophy and/or hyperplasia of the follicular cells of the thyroid occurred in male and female rats at >1000 mg/kg. Minimal hypertrophy of the zona fasciculata of the adrenal cortex was seen in three females at 20 000 mg/kg. Isolated findings of adrenal hypertrophy in females at 1000 and 5000 mg/kg were of questionable significance. The NOAEL was 250 mg/kg (equal to 24 mg/kg per day) on the basis of follicular cell hypertrophy and/or hyperplasia of the thyroid in both sexes at 1000 mg/kg (equal to 98 mg/kg bw per day) (Kyle et al., 1992).
In a study complying with GLP and the essential requirements of OECD guideline 408 (1981), groups of 10 male and 10 female Crl:CD BR rats were given diets containing methoxyfenozide (purity, 99.2%) at a concentration of 0, 50, 250, 1000, 5000 or 20 000 mg/kg for 90 days. These doses were equal to mean intakes of 0, 3.4, 17, 69, 353 and 1369 mg/kg bw per day for males, and 0, 3.7, 19, 72, 379 and 1531 mg/kg bw per day for females. Adequate stability, homogeneity and achieved concentration were demonstrated. Body weights and food consumption were measured weekly. Blood samples from non-fasted were taken at termination for the assessment of haematological (excluding methaemoglobin formation) and clinical chemistry parameters. Urine analysis was performed before termination. Ophthalmoscopy was performed before dosing and before termination. A comprehensive list of tissues from animals in the control group and in the group receiving the highest dose was examined microscopically; liver, lungs, kidneys and gross lesions were investigated in animals at the intermediate dose.
One female at 1000 mg/kg was found dead during week 7. No clinical signs of toxicity were noted in any animal. No notable effects were seen on body weight, body-weight gain, food consumption or ophthalmoscopy. Haematological findings indicative of mild anaemia were noted at the highest dose (Table 7). Plasma aspartate transaminase, alanine transaminase and alkaline phosphatase activities were decreased in treated groups; however, these findings were not considered to be of toxicological significance. Slight changes in other clinical chemistry values suggested mild hepatotoxicity at 20 000 mg/kg (Table 7). There was an indication of a slight increase in urinary protein concentrations in females at 5000 and 20 000 mg/kg, but in the absence of any related pathological findings this was not considered to be an adverse finding. No gross treatment-related findings were seen at necropsy. liver weights were increased, and there was evidence of periportal hepatocyte hypertrophy in both sexes at doses of >5000 mg/kg (Table 7). Thyroid weights were also increased slightly in males at the highest dose, but in the absence of histopathological correlates this is of unclear toxicological significance. The NOAEL was 1000 mg/kg (equal to 69 mg/kg bw per day) on the basis of increased relative liver weights (>10%) and associated hypertrophy at >5000 mg/kg (Anderson et al., 1995).
Table 7. Findings in rats given diets containing methoxyfenozide for 90 days
|
Parameter |
Sex |
Dietary concentration (mg/kg) |
|||||
|
0 |
50 |
250 |
1000 |
5000 |
20 000 |
||
|
Mean intake (mg/kg bw per day) |
M |
— |
3.4 |
17 |
69 |
353 |
1369 |
|
F |
— |
3.7 |
19 |
72 |
379 |
1531 |
|
|
Terminal body weight (g) |
M |
587.8 |
585.1 |
557.6 |
587.0 |
573.1 |
574.2 |
|
F |
330.0 |
323.8 |
356.0 |
323.4 |
334.1 |
324.1 |
|
|
Body-weight gain (g) |
M |
479.6 |
480.0 |
449.7 |
479.5 |
468.9 |
463.9 |
|
214.3 |
214.32 |
242.3 |
206.3 |
222.8 |
207.9 |
||
|
Haematology |
|||||||
|
Erythrocytes (106/ml) |
M |
8.22 |
8.37 |
8.11 |
8.49 |
8.06 |
7.95 |
|
F |
7.48 |
7.36 |
7.09 |
7.46 |
7.27 |
6.92* |
|
|
Erythrocyte volume fraction (l/l) |
M |
0.470 |
0.472 |
0.460 |
0.479 |
0.469 |
0.458 |
|
F |
0.450 |
0.446 |
0.433 |
0.456 |
0.441 |
0.426 |
|
|
Haemoglobin (g/dl) |
M |
158 |
15.9 |
15.7 |
16.0 |
15.9 |
15.5 |
|
F |
15.7 |
15.6 |
15.3 |
15.8 |
15.3 |
14.6* |
|
|
Clinical chemistry |
|||||||
|
AST (U/l) |
M |
124 |
130 |
110 |
108 |
105 |
115 |
|
F |
123 |
110 |
97 |
101 |
94 |
82* |
|
|
ALT (U/l) |
M |
53 |
62 |
46 |
51 |
52 |
57 |
|
F |
66 |
53 |
54 |
52 |
50 |
47* |
|
|
AP (U/l) |
M |
360 |
392 |
356 |
274 |
313 |
247* |
|
F |
358 |
312 |
296 |
248 |
245* |
204* |
|
|
Triglycerides (mg/dl) |
M |
131 |
149 |
119 |
137 |
147 |
168 |
|
F |
81 |
70 |
100 |
69 |
90 |
85 |
|
|
Cholesterol (mg/dl) |
M |
67 |
70 |
63 |
62 |
67 |
64 |
|
F |
76 |
71 |
76 |
77 |
69 |
63* |
|
|
Albumin (g/dl) |
M |
3.8 |
4.0 |
3.8 |
3.9 |
4.0 |
4.2* |
|
F |
4.4 |
4.2 |
4.3 |
4.2 |
4.2 |
4.1 |
|
|
Total protein (g/dl) |
M |
5.9 |
6.2 |
6.1 |
6.2 |
6.2 |
6.4* |
|
F |
6.4 |
6.2 |
6.4 |
6.3 |
6.3 |
6.4 |
|
|
Albumin: globulin ratio |
M |
1.8 |
1.8 |
1.8 |
1.8 |
1.8 |
1.8 |
|
F |
2.2 |
2.1 |
2.1 |
2.0 |
1.9 |
1.8* |
|
|
Organ weights |
|||||||
|
Liver (g) |
M |
20.5 |
21.6 |
20.52 |
21.4 |
22.0 |
23.1 |
|
F |
11.8 |
11.3 |
12.5 |
11.2 |
12.9 |
13.0 |
|
|
Liver (% bw) |
M |
3.39 |
3.59 |
3.593 |
3.57 |
3.77* |
3.94* |
|
F |
3.53 |
3.41 |
3.41 |
3.39 |
3.78 |
4.06* |
|
|
Thyroid (g) |
M |
0.031 |
0.031 |
0.0310 |
0.0310 |
0.034 |
0.037 |
|
F |
0.023 |
0.022 |
0.023 |
0.021 |
0.024 |
0.022 |
|
|
Thyroid (% bw) × 100 |
M |
0.526 |
0.526 |
0.524 |
0.493 |
0.582 |
0.623 |
|
F |
0.684 |
0.664 |
0.642 |
0.649 |
0.705 |
0.657 |
|
|
Liver histopathology |
|||||||
|
Hypertrophy (a = slight, b = moderate) |
M |
— |
— |
— |
— |
6a; 4b |
10b |
|
F |
— |
— |
— |
— |
10a |
10b |
|
From Anderson et al. (1995)
M, male; F, female; AST, aspartate transaminase; ALT, alanine transaminase; AP, alkaline phosphatase
* p < 0.05
Dogs
Five reports of dietary studies in dogs were submitted: two 2-week studies, a 90-day study, a 1-year study and a study of reversibility of blood effects. All studies, except the first 2-week study, were conducted by the same laboratory.
In a 2-week dietary study conducted in 1992, a final report was issued in 1994 and a revised report was issued in 1995; the latter included amendments to the interpretation of the haematology, clinical chemistry and organ weight data. The present summary refers to the revised study report, which complied with GLP. Groups of two male and two female beagle dogs (aged 6-8 months at the start of dosing) were given diets containing methoxyfenozide (purity, 99.2%) at concentrations of up to 30 000 mg/kg (Table 8), as 400 g of food per day. Body weight was measured before dosing and on days 1, 2, 4, 7 and 14. Food consumption was measured daily. Blood samples for haematology (including methaemoglobin formation and reticulocytes) and clinical chemistry examination were taken after about 18 h without food before dosing and in week 2 (this might permit some recovery of levels of methaemoglobin formation). At necropsy major organs were weighed, but no histopathological investigations were performed.
Table 8. Dietary concentrations of methoxyfenozide administered in a 2-week study in dogs
|
Group |
Dietary concentration |
Mean intake (mg/kg bw per day) |
|
|
Male |
Female |
||
|
1 |
0 |
0 |
0 |
|
2 |
300 |
13 |
16 |
|
3 |
3 500 |
154 |
157 |
|
4 |
7 000 |
308 |
299 |
|
5 |
15 000 |
603 |
608 |
|
6 |
30 000 |
1225 |
1130 |
From Nuttall & Kelly (1995)
There were no deaths, no treatment-related clinical signs and no notable changes in food consumption or body-weight gain.
Treatment-related changes in erythrocyte cell parameters, indicative of haemolytic anaemia, were recorded for animals of each sex receiving methoxyfenozide at >3500 mg/kg. However, not all changes in haemolytic parameters were dose-dependent. Particularly notable was a clear dose-related increase in the mean percentage of erythrocytes with Heinz bodies: males, 0% of controls, 13% at 3500 mg/kg, rising to 41% at 30 000 mg/kg; females, 0% of controls, 17% at 3500 mg/kg, rising to 33% at 30 000 mg/kg. The mean percentage of methaemoglobin was increased, but there was no clear dose-response: males, 0.8% for controls, 1.5-3.4% at >3500 mg/kg; females, 1.0% for controls, 1.7-3.5% at >3500 mg/kg. A slight to moderate incidence of Howell-Jolly bodies was seen in all males at >3500 mg/kg and in all females at >7000 mg/kg. The only treatment-related effect was an increase in plasma total bilirubin concentrations in most dogs receiving >3500 mg/kg when compared with values before dosing and for controls. The spleen was the only organ investigated for which there were indications of a treatment-related effect on weight. Increases in relative and absolute weights were seen in both sexes at >3500 mg/kg and in males at 300 mg/kg (Table 9).
Table 9. Spleen weights in dogs given diets containing methoxyfenozide for 2 weeks
|
Dietary concentration (mg/kg) |
Body weight (g) |
Spleen weight (g) |
Spleen: body weight (%) |
Spleen: body weight |
|
Males |
||||
|
0 |
9 850 |
25.3 |
0.26 |
|
|
300 |
8 600 |
30.3 |
0.35 |
+35% |
|
3 500 |
8 680 |
58.4 |
0.67 |
+111% |
|
7 000 |
8 700 |
42.9 |
0.51 |
+98% |
|
15 000 |
8 600 |
77.0 |
0.89 |
+249% |
|
30 000 |
10 000 |
63.2 |
0.62 |
+142% |
|
Females |
||||
|
0 |
7 550 |
25.1 |
0.33 |
|
|
300 |
7 500 |
23.6 |
0.31 |
-6% |
|
3 500 |
7 725 |
40.3 |
0.52 |
+57% |
|
7 000 |
7 650 |
63.9 |
0.85 |
+154% |
|
15 000 |
7 675 |
45.9 |
0.63 |
+88% |
|
30 000 |
7 350 |
38.8 |
0.53 |
+58% |
From Nuttall & Kelly (1995)
The NOAEL was 300 mg/kg (equal to 13 mg/kg bw per day) on the basis of evidence of haemolytic anaemia (haematological changes, increased serum bilirubin, increased spleen weight), in both sexes at 3500 mg/kg. The increased spleen weight at 300 mg/kg in males is not considered to be adverse in the absence of effects on erythrocytes (Nuttall & Kelly, 1995).
Groups of two male and two female beagle dogs were given diets containing methoxyfenozide (purity, 98%) at a concentration of 0, 500, 5000, 15 000 or 30 000 mg/kg for 2 weeks. Mean intakes were 0, 18, 202, 509 and 1003 mg/kg bw per day for males, and 20, 196, 757 and 1186 mg/kg bw per day for females. The study complied with GLP. All dogs were observed routinely for signs of ill health or reaction to treatment, and body weight and food consumption were recorded. All dogs were offered 400 g of feed daily, beginning with a 2-week period before dosing. During the first week of this 2-week period and before necropsy, blood samples were collected from all dogs for haematology (including methaemoglobin formation and reticulocytes) and clinical chemistry analyses) were saved in 10% neutral buffered formalin. Liver and spleen were weighed. Histopathological evaluation was performed on liver, spleen, bone marrow and all gross lesions from all dogs.
There were no compound-related deaths, clinical signs of systemic toxicity, and no effects on body weight or feed consumption. In females at 15 000 and 30 000 mg/kg, changes seen included a decrease in erythrocyte counts (about 30%), haemoglobin concentration (about 25%) and erythrocyte volume fraction (about 25%), an increase in methaemoglobin formation (about 300%) and a significant increase in the percentage of reticulocytes (by 500-5000%). Other effects observed included Heinz bodies and nucleated erythrocytes. Minimal changes in erythrocyte counts, haemoglobin concentration, erythrocyte volume fraction and reticulocyte counts were observed in both sexes at 5000 mg/kg and in males at 15 000 and 30 000 mg/kg. Given the small group size, these latter changes are difficult to interpret, but overall there would seem to be a substance-related increase in the percentage of reticulocytes at >5000 mg/kg in females (Table 10). There was some evidence for a slight increase in total bilirubin in females at the highest dose, but the variability in values before dosing makes this an equivocal finding.
Table 10. Reticulocyte counts in dogs given diets containing methoxyfenozide for 2 weeks
|
Dietary concentration (mg/kg) |
Reticulocytes (%) |
Mean % change compared with: |
||
|
Day -12 |
Day 16 |
Before dosing |
Concurrent control |
|
|
Males |
||||
|
0 |
0.0; 0.2 |
0.7; 0.2 |
+400 |
— |
|
500 |
0.4; 0.0 |
0.8; 0.6 |
+250 |
+40 |
|
5 000 |
0.6; 0.8 |
1.5; 1.0 |
+86 |
+160 |
|
15 000 |
0.2; 0.6 |
2.0; 1.6 |
+350 |
+260 |
|
30 000 |
0.0; 0.2 |
1.0; 2.6 |
+1700 |
+260 |
|
Females |
||||
|
0 |
0.4; 0.2 |
1.2; 0.6 |
+200 |
— |
|
500 |
0.0; 0.4 |
0.4; 1.2 |
+300 |
-11 |
|
5 000 |
0.2; 0.4 |
2.8; 2.0 |
+700 |
+167 |
|
15 000 |
0.2; 0.4 |
4.6; 9.1 |
+2200 |
+667 |
|
30 000 |
0.0; 0.2 |
6.6; 3.6 |
+5000 |
+467 |
From Vandenberghe & Gillette (1995)
Relative spleen weight was increased compared with that of controls at most doses in both sexes; the lack of a clear dose-related response in either sex makes it questionable whether the response was treatment related (Table 11). Histopathological findings were limited to minimal haemosiderin accumulation in Kupffer cells in the livers of females at 15 000 and 30 000 mg/kg. There were no histopathological findings in the spleen.
Table 11. Spleen weight in dogs given diets containing methoxyfenozide for 2 weeks
|
Dietary concentration (mg/kg) |
Mean body weight (g) |
Spleen weight (g) |
Spleen: body weight (%) |
Spleen: body weight (% change versus control) |
|
Males |
||||
|
0 |
8970 |
28.500 |
0.318 |
— |
|
500 |
9200 |
47.200 |
0.530 |
+67 |
|
5 000 |
8680 |
58.350 |
0.671 |
+111 |
|
15 000 |
8800 |
40.950 |
0.469 |
+47 |
|
30 000 |
8730 |
36.600 |
0.428 |
+35 |
|
Females |
||||
|
0 |
7910 |
24.600 |
0.311 |
— |
|
500 |
7630 |
37.050 |
0.484 |
+56 |
|
5 000 |
7190 |
33.500 |
0.461 |
+48 |
|
15 000 |
7870 |
23.650 |
0.299 |
-4 |
|
30 000 |
7680 |
31.900 |
0.415 |
+33 |
From Vandenberghe & Gillette (1995)
The NOAEL was 500 mg/kg (equal to 20 mg/kg bw) on the basis of increased reticulocytes in females at 5000 mg/kg (196 mg/kg bw per day) (Vandenberghe & Gillette, 1995).
Groups of four male and four female beagle dogs (aged 5-6 months at the start of dosing) were given diets containing methoxyfenozide (purity, 99.8%) at a concentration of 0, 15, 50, 500 or 5000 mg/kg for 13 weeks. Owing to an apparent lack of substance-related effects at all doses, dogs from the group receiving 15 mg/kg were dosed for a further 2 weeks at 15 mg/kg and then for 6 weeks at 15 000 mg/kg. The mean dietary intake during dosing at the higher concentration was 422 mg/kg bw per day in males and 460 mg/kg bw per day in females. The lack of concurrent negative controls for these animals at the highest dose limits interpretation of the results, but for some parameters a comparison can be made of findings before and after the increase in dose. The study complied with GLP and with OECD guideline 409 (1998); the absence of toxicity at the highest dose was acceptable given the findings in the 2-week studies.
Stability, homogeneity and achieved dietary content were satisfactory. Mean intakes of methoxyfenozide are shown in Table 12. Dogs had access to 400 g of food per day for 2 h. Dogs were observed routinely for mortality, clinical signs, body weight and food consumption. Fasted blood samples were taken for the assessment of haematological (including methaemoglobin formation, erythrocyte morphology and reticulocyte count) and clinical chemistry parameters before the start of dosing and during weeks 7 and 13, with samples being taken from the extended dosing group after dosing at 15 000 mg/kg for 16 and 38 days. Ophthalmoscopy was performed before the start of dosing and before termination. Urine analysis was performed at termination. Weights of the adrenals, liver, spleen, kidneys, testes, brain and thyroids were recorded at necropsy. A comprehensive list of tissues from all animals was examined histopathologically.
Table 12. Findings in dogs given diets containing methoxyfenozide for 90 days
|
Parameter |
Dietary concentration (mg/kg) |
||||
|
0 |
15 |
50 |
500 |
5000 |
|
|
Males |
|||||
|
Intakes (mg/kg bw per day) |
0 |
0.6 |
2.0 |
21 |
198 |
|
Absolute kidney weight g) |
59 |
— |
54 |
53 |
50 |
|
Relative liver weight (% body weight) |
2.8 |
— |
3.0 |
3.2 |
3.2 |
|
Body-weight gain (g) |
|||||
|
Weeks 0-6 |
2000 |
1675 |
1500 |
1400 |
1775 |
|
Weeks 6-13 |
1650 |
160 |
1650 |
1800 |
1500 |
|
Platelets (103/µl) |
|||||
|
Before start of dosing |
393 |
356 |
390 |
357 |
401 |
|
Day 45 |
317 |
342 |
393 |
332 |
393 |
|
Day 87 |
301 |
302 |
348 |
323 |
366 |
|
Erythrocyte count (106/µl) |
|||||
|
Before start of dosing |
6.14 |
5.95 |
6.32 |
5.78 |
5.84 |
|
Day 45 |
5.84 |
5.72 |
5.94 |
5.38 |
5.38 |
|
Day 87 |
7.29 |
6.12* |
6.07* |
5.88* |
6.07* |
|
Haemoglobin (g/dl) |
|||||
|
Before start of dosing |
12.7 |
13.4 |
13.8 |
12.7 |
12.6 |
|
Day 45 |
12.1 |
12.8 |
13.3 |
12.1 |
12.0 |
|
Day 87 |
15.0 |
13.7 |
13.6 |
13.0* |
13.1* |
|
Erythrocyte volume fraction (l/l) |
|||||
|
Before start of dosing |
0.364 |
0.379 |
0.394 |
0.362 |
0.363 |
|
Day 45 |
0.350 |
0.366 |
0.376 |
0.343 |
0.341 |
|
Day 87 |
0.440 |
0.394 |
0.387 |
0.371* |
0.384 |
|
Methaemoglobin (%) |
|||||
|
Before start of dosing |
1.0 |
0.7 |
0.8 |
1.0 |
0.8 |
|
Day 45 |
1.0 |
0.9 |
0.9 |
1.0 |
1.0 |
|
Day 87 |
0.7 |
0.8 |
0.8 |
0.8 |
1.0* |
|
Females |
|||||
|
Intakes (mg/kg bw per day) |
0 |
0.6 |
1.9 |
20 |
209 |
|
Absolute kidney weight (g) |
44 |
— |
44 |
53* |
42 |
|
Relative liver weight (% body weight) |
2.7 |
— |
2.8 |
3.1 |
3.1 |
|
Body-weight gain (g) |
|||||
|
Weeks 0-6 |
1825 |
1650 |
130 |
1850 |
1300* |
|
Weeks 6-13 |
1150 |
1175 |
1025 |
1400 |
1125 |
|
Platelet (103/µl) |
|||||
|
Before start of dosing |
444 |
375 |
372 |
402 |
400 |
|
Day 45 |
410 |
335 |
361 |
352 |
364 |
|
Day 87 |
351 |
332 |
310 |
325 |
300 |
|
Erythrocyte count (106/µl) |
|||||
|
Before start of dosing |
6.38 |
6.02 |
6.02 |
5.81 |
6.13 |
|
Day 45 |
6.19 |
5.83 |
6.24 |
5.48 |
5.93 |
|
Day 87 |
6.90 |
6.58 |
6.77 |
6.18 |
6.35 |
|
Haemoglobin (g/dl) |
|||||
|
Before start of dosing |
13.3 |
13.5 |
13.4 |
13.2 |
13.5 |
|
Day 45 |
13.3 |
13.3 |
13.9 |
12.5 |
13.5 |
|
Day 87 |
14.7 |
14.6 |
14.9 |
13.6 |
14.2 |
|
Erythrocyte volume fraction (l/l) |
|||||
|
Before start of dosing |
0.386 |
0.387 |
0.382 |
0.374 |
0.390 |
|
Day 45 |
0.381 |
0.381 |
0.399 |
0.356 |
0.383 |
|
Day 87 |
0.426 |
0.426 |
0.429 |
0.394 |
0.403 |
|
Methaemoglobin (%) |
|||||
|
Before start of dosing |
0.9 |
0.9 |
0.9 |
1.0 |
0.9 |
|
Day 45 |
0.9 |
0.9 |
0.9 |
1.1 |
0.9 |
|
Day 87 |
0.7 |
0.8 |
0.8 |
0.9 |
0.7 |
From Kaminski, Shuey & Lomax (1995b)
a Values are means, n = 4
* Significant difference from control, p < 0.05; analysis of covariance followed by Dunnett t-test
All dogs survived to termination and no substance-related clinical signs were observed. Mean body weight and body-weight gains of females at 5000 mg/kg were lower than controls at the start of the study and attained statistical significance at a number of time-points, but body-weight gain between weeks 6 and 13 was similar to that of controls (Table 12). These findings were primarily, but not entirely, attributable to one dog that gained only 2.0 kg (control range, 2.4-3.1 kg). No effects on body weight were observed in males. Interpretation of the body weight data for dogs dosed at 15 000 mg/kg is difficult in the absence of concurrent controls. No effects were seen on food consumption by either sex during the main 90-day study. There was also no change in mean daily food consumption of animals for which the dose administered was increased from 15 mg/kg to 15 000 mg/kg.
No consistent effects were seen on haematological parameters during the main 90-day study. Although there were some statistically significant changes, none were considered to be substance-related because of the lack of change from values before the start of dosing and/or lack of convincing time- or dose-response relationship. The statistically significant reduction in erythrocyte count at all doses in males on day 87 was associated with a high value for controls and showed no dose-response relationship (Table 12). The impact of fasting on methaemoglobin concentrations is uncertain. There were no apparent haematological effects in animals for which the dose was increased from 15 mg/kg to 15 000 mg/kg. There were no notable changes in clinical chemistry parameters when variations in values before the start of dosing were taken into consideration. Urine analysis results indicated a slight increase in specific gravity and protein content in both sexes at 5000 mg/kg. Ophthalmoscopy findings were similar in all groups.
Relative liver weights were higher at 500 and 5000 mg/kg in both sexes compared with those of the controls, but there was no clear dose-response relationship (Table 12). Absolute liver weights were not increased in the satellite group receiving 15 000 mg/kg. Absolute and relative kidney weights were decreased slightly in males at 50, 500 and 5000 mg/kg (Table 12). Individual kidney weights (absolute and relative) at 5000 mg/kg were mostly below the range of values for the controls, which when taken with the urine analysis results, indicates a possible marginal effect at 5000 mg/kg. After exposure to 15 000 mg/kg, relative kidney weights for males were mostly below the range of values for controls from the main part of the study. An apparent decrease in absolute and relative spleen weight in males at 50-5000 mg/kg is attributed to one control male with a large spleen. No substance-related gross or microscopic findings were noted at necropsy of animals at the end of the main 90-day study or at the end of exposure to 15 000 mg/kg. The absence of any animals with pigment in the liver or spleen is notable.
The NOAEL was 5000 mg/kg (198-209 mg/kg bw per day). The slight effects on body weights, haematology, urine analysis and organ weights are not considered to be clearly adverse, given the absence of histopathological findings and the variability in values before the start of dosing (Kaminski, Shuey & Lomax, 1995b).
In a study that essentially complied with OECD guideline 452 (1981) and was performed according to GLP, groups of four male and four female beagle dogs (aged 5-6 months at the start of dosing) were given diets containing methoxyfenozide (purity, 98%) at a concentration of 0, 60, 300, 3000, or 30 000 mg/kg (equal to 0, 2.2, 9.8, 106 or 1152 mg/kg bw per day in males, and 0, 2.2, 12.6, 111 or 1199 mg/kg bw per day in females) for 52 weeks. Dogs had access to about 400 g of food for 2 h per day (except for one occasion when food had to be left in the cages overnight). Adequate homogeneity, stability and content of the diets were demonstrated. Dogs were observed routinely for mortality, signs of ill health, body weight and food consumption. Urine analysis was conducted for all dogs before the start of testing, and after 3, 6, and 12 months of treatment. Blood samples were collected from all (fasted) dogs for haematology (including methaemoglobin formation, but not morphology or reticulocyte count) and clinical chemistry analyses before the start of testing, and after 3, 6, and 12 months of treatment. Ophthalmology examinations were performed on all dogs before the start of testing and after 12 months of treatment. At termination, all dogs were killed and necropsied, major organs were weighed and an extensive range of tissues from all animals was examined histologically.
There were no deaths or clinical signs of toxicity. Body-weight gain was reduced at 30 000 mg/kg in males at the start of the study, but was similar to that of controls for the final 9 months of the study (Table 13). In females, body-weight gain was reduced at 30 000 mg/kg over the latter half of the study, primarily as a result of weight loss by one animal (Table 13). Food consumption was similar in all groups.
Table 13. Mean body-weight gain (range) in dogs given diets containing methoxyfenozide for 1 year
|
Dietary concentration (mg/kg) |
|||||
|
0 |
60 |
300 |
3000 |
30 000 |
|
|
Males |
|||||
|
Weeks 0-13 |
3042 |
2520 |
2928 |
2888 |
2518 |
|
(2755-3307) |
(2157-2826) |
(1912-3877) |
(2470-3383) |
(2019-3174) |
|
|
Weeks 0-52 |
4022 |
3571 |
3971 |
3752 |
3111 |
|
(3530-4218) |
(3221-3960) |
(2425-5958) |
(3511-3967) |
(2298-3627) |
|
|
Females |
|||||
|
Weeks 0-26 |
2474 |
2684 |
2227 |
2834 |
2578 |
|
(1981-2982) |
(2431-2885) |
(1303-3427) |
(1890-3483) |
(1766-3157) |
|
|
Weeks 0-52 |
2782 |
2641 |
2446 |
2635 |
2348 |
|
(2362-3077) |
(2333-3272) |
(1463-3621) |
(1985-3311) |
(1391-2791) |
|
From Morrison & Shuey (1997)
The percentage of methaemoglobin was statistically significantly increased at 30 000 mg/kg in both sexes compared with controls (Table 14). There was also evidence for a slight increase in methaemoglobin concentration at 3000 mg/kg in both sexes when compared with mean and individual values before the start of dosing. Although the maximum mean percentage of methaemoglobin (2.8%) at 30 000 mg/kg was not clearly adverse relative to the background level in humans (2-12%) (Bell et al., 1972), the use of fasted samples could mean that the peak percentage in dogs was >2.8%. Decreases were seen in erythrocyte parameters at 3000 and 30 000 mg/kg in both sexes (Table 14). Although many of the changes at 3000 mg/kg were not statistically significant when compared with controls, the changes were evident relative to values for the group before the start of testing (Table 14). There was also a very minimal increase in nucleated erythrocytes in both sexes at 30 000 mg/kg. The pattern of changes in haematology is consistent with methaemoglobin-induced anaemia. Platelet numbers were increased in both sexes at 3000 mg/kg (by about 50%) and 30 000 mg/kg (by about 85%) compared with numbers for controls. There were no effects on white blood cells. Total bilirubin in blood and urine was increased at 3000 and 30 000 mg/kg in both sexes. There was no effect on blood urea nitrogen, urine specific gravity or protein content, or on ophthalmological findings.
Table 14. Haematology results in dogs given diets containing methoxyfenozide for 1 year
|
Parameter |
Dietary concentration (mg/kg) |
||||
|
0 |
60 |
300 |
3000 |
30 000 |
|
|
Males |
|||||
|
Platelet (103/µl) |
|||||
|
Before start of dosing |
378 |
319 |
349 |
425 |
405 |
|
Day 93 |
345 |
298 |
328 |
542* |
650* |
|
Day 184 |
305 |
267 |
294 |
497* |
581* |
|
Day 366 |
309 |
255 |
309 |
468* |
605* |
|
Erythrocyte count (106/µl) |
|||||
|
Before start of dosing |
6.20 |
5.93 |
5.67 |
5.64 |
6.46 |
|
Day 93 |
6.53 |
6.25 |
6.61 |
5.60* |
5.57* |
|
Day 184 |
6.55 |
6.17 |
6.38 |
5.77 |
5.83 |
|
Day 366 |
6.81 |
6.99 |
6.92 |
6.24 |
6.52 |
|
Haemoglobin (g/dl) |
|||||
|
Before start of dosing |
13.2 |
12.8 |
12.4 |
12.8 |
13.8 |
|
Day 93 |
13.9 |
13.6 |
14.5 |
12.9 |
12.4 |
|
Day 184 |
14.0 |
13.3 |
13.9 |
13.1 |
13.2 |
|
Day 366 |
14.5 |
14.9 |
15.2 |
14.3 |
14.4 |
|
Erythrocyte volume fraction (l/l) |
|||||
|
Before start of dosing |
0.388 |
0.371 |
0.362 |
0.368 |
0.399 |
|
Day 93 |
0.407 |
0.398 |
0.425 |
0.377 |
0.362 |
|
Day 184 |
0.408 |
0.394 |
0.417 |
0.390 |
0.383 |
|
Day 366 |
0.425 |
0.422 |
0.422 |
0.416 |
0.422 |
|
Methaemoglobin (%) |
|||||
|
Before start of dosing |
0.6 |
0.7 |
0.8 |
0.6 |
0.7 |
|
Day 93 |
0.6 |
0.7 |
0.8 |
1.0 |
2.8* |
|
Day 184 |
0.6 |
0.7 |
0.6 |
1.0 |
2.2* |
|
Day 366 |
0.7 |
0.9 |
0.9 |
1.1 |
1.9* |
|
Females |
|||||
|
Platelet (103/µl) |
|||||
|
Before start of dosing |
418 |
326 |
385 |
373 |
333 |
|
Day 93 |
343 |
286 |
420 |
430 |
562* |
|
Day 184 |
332 |
298 |
388 |
430 |
560* |
|
Day 366 |
368 |
372 |
421 |
434 |
667* |
|
Erythrocyte count (106/µl) |
|||||
|
Before start of dosing |
6.62 |
5.88 |
6.30 |
5.64 |
6.06 |
|
Day 93 |
6.76 |
6.35 |
6.36 |
5.54* |
5.07* |
|
Day 184 |
6.96 |
6.20 |
6.17 |
5.54* |
5.23* |
|
Day 366 |
7.49 |
6.42 |
6.75 |
6.09* |
5.84* |
|
Haemoglobin (g/dl) |
|||||
|
Before start of dosing |
14.0 |
12.9 |
13.6 |
12.2 |
13.2 |
|
Day 93 |
14.4 |
14.2 |
13.8 |
12.3 |
12.0 |
|
Day 184 |
14.9 |
13.9 |
13.6 |
12.7* |
12.3* |
|
Day 366 |
16.0 |
14.6 |
15.0 |
13.7 |
13.8 |
|
Erythrocyte volume fraction (l/l) |
|||||
|
Before start of dosing |
0.412 |
0.383 |
0.397 |
0.357 |
0.386 |
|
Day 93 |
0.424 |
0.413 |
0.405 |
0.359 |
0.344* |
|
Day 184 |
0.437 |
0.410 |
0.400 |
0.366* |
0.363* |
|
Day 366 |
0.469 |
0.424 |
0.434 |
0.395 |
0.399 |
|
Methaemoglobin (%) |
|||||
|
Before start of dosing |
0.8 |
0.7 |
0.8 |
0.8 |
0.9 |
|
Day 93 |
0.6 |
0.8 |
0.9 |
1.1 |
2.5* |
|
Day 184 |
0.8 |
0.7 |
0.8 |
1.0 |
2.8* |
|
Day 366 |
0.8 |
0.8 |
1.4 |
1.3 |
1.9* |
From Morrison & Shuey (1997)
* p < 0.05
There were increases in mean absolute and relative liver weights (by 13 and 24%, respectively) and in mean absolute and relative thyroid weights (by 43 and 63%, respectively) of males at 30 000 mg/kg. Kidney weight was unaffected by treatment. There were no treatment-related gross pathological findings. There were no histopathological correlates to the increased liver and thyroid weights, although increased iron positive pigment was seen in splenic and hepatic macrophages in animals receiving 30 000 mg/kg. In the bone marrow, increased cellularity characterized by decreased fat vacuoles and increased haematopoietic cells (primarily erythrocytes and erythrocyte precursors) was observed at 30 000 mg/kg. The histopathological changes were consistent with the normal response to haemolytic anaemia. There were no substance-related histopathological effects on the thyroid or kidney.
The NOAEL was 300 mg/kg (equal to 9.8 mg/kg bw per day) on the basis of changes in liver hypertrophy, erythrocyte parameters and increased blood platelet count at 3000 mg/kg (equal to 106 mg/kg bw per day) (Morrison & Shuey, 1997).
A study complying with GLP was designed to investigate the reversibility of blood effects seen in the 1-year study in dogs. Groups of four male beagle dogs (aged 10-11 months at the start of dosing) were given diets containing methoxyfenozide (purity, 98%) at a concentration of 0 or 30 000 mg/kg for 4 weeks (the dosing phase). The dogs were selected from the control group and group receiving the lowest dose in a previously conducted 13-week study of dermal application with another test material. The dogs had access to about 400 g of food for 2 h per day. Acceptable stability, homogeneity and achieved content of the diet were demonstrated. The achieved intakes were 0 and 1036 mg/kg bw per day. After 4 weeks of treatment, all animals were maintained on untreated diet for 4 additional weeks (the recovery phase).
All dogs were observed routinely for mortality, clinical signs, feed consumption and body weights. Blood samples (fasting not specified) were collected from all dogs for haematology analyses (reticulocytes not counted) before the start of treatment, after 4 weeks of treatment, and after 2 and 4 weeks of recovery. At the end of the recovery phase, animals were returned to the animal care unit. No mortalities or clinical signs indicative of systemic toxicity were seen. Body-weight gain and food consumption were unaffected by treatment. Methoxyfenozide produced a range of haematological effects after 4 weeks of dietary administration at 30 000 mg/kg bw (Table 15). Clear evidence of reversibility was demonstrated after the 4-week recovery phase (Table 15) (Bannister & Morrison, 1998).
Table 15. Haematology findings in male dogs given diets containing methoxyfenozide (recovery study)
|
Parameter |
Dietary conceentration (mg/kg) |
|
|
0 |
30 000 |
|
|
Methaemoglobin (%) |
||
|
Before start of dosing phase—day 0 |
0.7 |
0.8 |
|
Dosing phase—day 28 |
0.8 |
2.2* |
|
Recovery phase—day 14 |
0.9 |
1.4 |
|
Recovery phase—day 28 |
0.9 |
1.0 |
|
Mean cell volume (µm3) |
||
|
Before start of dosing phase—day 0 |
67.8 |
68.6 |
|
Dosing phase—day 28 |
66.8 |
72.6* |
|
Recovery phase—day 14 |
66.7 |
71.6* |
|
Recovery phase—day 28 |
67.0 |
70.3 |
|
Mean cell haemoglobin (pg) |
||
|
Before start of dosing phase—day 0 |
23.5 |
23.9 |
|
Dosing phase—day 28 |
23.3 |
25.2* |
|
Recovery phase—day 14 |
23.2 |
24.8* |
|
Recovery phase—day 28 |
22.9 |
24.3 |
|
Platelets (103/µl) |
||
|
Before start of dosing phase—day 0 |
339 |
333 |
|
Dosing phase—day 28 |
338 |
487 |
|
Recovery phase—day 14 |
334 |
416 |
|
Recovery phase—day 28 |
326 |
362 |
|
Haemoglobin (g/dl) |
||
|
Before start of dosing phase—day 0 |
15.6 |
16.3 |
|
Dosing phase—day 28 |
15.8 |
14.9* |
|
Recovery phase—day 14 |
16.0 |
16.2 |
|
Recovery phase—day 28 |
16.5 |
17.0 |
|
Erythrocyte count (106/µl) |
||
|
Before start of dosing phase—day 0 |
6.63 |
6.86 |
|
Dosing phase—day 28 |
6.79 |
5.91* |
|
Recovery phase—day 14 |
6.91 |
6.54 |
|
Recovery phase—day 28 |
7.18 |
7.01 |
|
Erythrocyte volume fraction (l/l) |
||
|
Before start of dosing phase—day 0 |
0.449 |
0.470 |
|
Dosing phase—day 28 |
0.453 |
0.428* |
|
Recovery phase—day 14 |
0.461 |
0.468 |
|
Recovery phase—day 28 |
0.480 |
0.492 |
From Bannister & Morrison (1998)
*p < 0.05; Dunnett t-test
Rats
This study of toxicity exceeded the basic requirements of OECD guideline 410 (1981), notably in terms of the number of animals used and the extent of the histopathological examination. The study complied with GLP. Methoxyfenozide (purity, 98%), a powder, was moistened with tap water (1:1, w/v) and applied to the shaved intact skin (about 10% of the total body surface area) of Crl:CD®BR rats (10 of each sex per group) at a dose of 0, 75, 300 or 1000 mg/kg bw per day for 5 days per week over a 4-week period (20 exposures). The exposure site was covered with an occlusive dressing, which was removed after 6 h and the exposure site was washed with soap solution and then wiped. Animals were observed routinely for mortality, signs of ill health, reaction to treatment, body weight and food consumption. Application sites were evaluated for skin irritation before each daily treatment and at terminal sacrifice. Haematology (including methaemoglobin measurement, but not reticulocyte count) and clinical chemistry were performed at terminal sacrifice. An indirect ophthalmoscopic examination was performed before initiation of dosing and at the end of the dosing period. An extensive histopathological examination (including treated and non-treated skin) was conducted for all control animals and animals at the highest dose. In addition, kidneys, liver, lungs and all gross lesions from all animals were examined histopathologically.
All rats survived to termination. There was no evidence of substance-related effects on clinical signs, skin irritation, ocular effects, haematology, clinical chemistry, organ weight or histopathology. A reduction in food consumption (13%) in week 4 and reduced body-weight gain (16% between days 0-28) in males at 1000 mg/kg bw per day was not considered to be an effect of clear toxicological importance; there was no statistically significant decrease in body weight and no effect on food consumption or body-weight gain was seen in females. The NOAEL was 1000 mg/kg bw per day, the highest dose tested (Parno et al., 1998a).
Mice
Groups of 60 male and 60 female Crl:CD-1®(ICR)BR (VAF Plus) mice were given diets containing methoxyfenozide (purity, 98%) at a concentration of 0, 70, 2800, or 7000 mg/kg for 18 months. These concentrations resulted in mean intakes of 0, 10.0, 405 or 1020 mg/kg bw per day in males, and 0, 12.8, 529 or 1354 mg/kg bw per day in females. Mice had access to food ad libitum and were housed individually. Satisfactory stability, homogeneity and content of the test material were demonstrated. Mice were observed routinely for mortality, ill health, reaction to treatment, body weight and feed consumption. After 12 and 18 months of treatment, white blood cell differential counts were conducted for all control mice and mice at the highest dose. An extensive histopathological examination was performed for these mice, as well as on mice that died in groups receiving the other doses. Microscopic examination for survivors at the lowest and intermediate doses was limited to liver, kidneys, lungs and gross lesions. The study report included an independent peer review that confirmed the conclusions of the original pathologist. The study complied with OECD guideline 451 (1981) and GLP.
No effects on survival were observed in mice at any dose. A total of 43-48 males per group and 47-52 females per group survived to termination. There were no compound-related clinical signs indicative of systemic toxicity in any of the treatment groups. There were no substance-related effects on body weight, cumulative body-weight gain, food consumption, white blood cell counts or non-neoplastic pathology findings. A slight (8-10%) but statistically significant reduction in kidney weight in males at 2800 and 7000 mg/kg was not considered to be substance-related, in the absence of a dose-response relationship, the lack of an effect on relative kidney weight and no associated histopathological findings. A slight dose-related increase in absolute liver weight (9-13%) and relative liver weight (12-18%) in males at 2800 and 7000 mg/kg was considered to be of no toxicological importance because the increases were not statistically significant, were not seen in females and there were no related histopathological findings. A slight increase in the incidence of extramedullary haematopoiesis in the spleen was noted at the highest dose (males, 55%; females, 72%, versus 43% and 57% in controls respectively). This finding was consistent with effects on erythrocytes seen in shorter studies, but was not statistically significant and was only slightly above the high background rate in controls.
The incidence of bronchiolo-alveolar adenoma or carcinoma was slightly increased in females at 2800 and 7000 mg/kg (10% for each group) compared with controls, and 70 mg/kg (3.3% for each group) (Table 16). There was no clear evidence of an increased incidence in males. The trend in females was statistically significant, but no pair-wise comparison was statistically significant. The incidence of these adenomas (3-8%) and carcinomas (3-7%) in females at 2800 and 7000 mg/kg was within the range for historical controls for adenomas (up to 15%) and carcinomas (up to 10%) reported for female Charles River mice of this strain (Lang, 1995; no data were available from the test facility). Overall, it was considered that the lung tumours were not treatment-related.
Table 16. Incidence of lung tumours in mice given diets containing methoxyfenozide for 18 months
|
Lung tumour |
Dietary concentration (mg/kg) |
|||
|
0 |
70 |
2800 |
7000 |
|
|
Males |
||||
|
Lung, adenoma, bronchiolo-alveolar |
8/60 |
6/60 |
10/60 |
8/60 |
|
Lung, carcinoma, bronchiolo-alveolar |
1/60 |
2/60 |
3/60 |
1/60 |
|
Lung, adenoma or carcinoma, bronchiolo-alveolar |
9/60 |
8/60 |
13/60 |
8/60 |
|
Females |
||||
|
Lung, adenoma, bronchiolo-alveolar |
2/60 |
0/60 |
2/60 |
5/60 |
|
Lung, carcinoma, bronchiolo-alveolar |
0/60 |
2/60 |
4/60 |
1/60 |
|
Lung, adenoma or carcinoma, bronchiolo-alveolar |
2/60 |
2/60 |
6/60 |
6/60 |
From Robinson et al. (1998)
The NOAEL for carcinogenicity and non-neoplastic effects was 7000 mg/kg (equal to 1020 mg/kg per day), the highest dose tested (Robinson et al., 1998).
Rats
Groups of 70 male and 70 female Sprague-Dawley rats were given diets containing methoxyfenozide (purity, 98%) at a concentration of 0 (control), 200, 8000, or 20 000 mg/kg. These concentrations resulted in mean intakes of 0, 10.2, 411 and 1045 mg/kg per day in males, and 0, 11.9, 491 and 1248 mg/kg per day in females. The study complied with GLP and OECD guideline 453. Rats had access to food ad libitum and were housed individually. Satisfactory stability homogeneity and content of the test material were demonstrated. All rats were observed daily for signs of ill health or reaction to treatment. Physical examinations were performed weekly. Body weight and food consumption were monitored weekly for the first 13 weeks of dosing and once every fourth week thereafter. Ophthalmoscopic examinations were performed on all rats during the period before the start of dosing, and on all surviving rats before the termination of treatment. Urine analysis parameters were evaluated for at least 10 rats of each sex per group at 3, 6, 12 and 18 months of treatment, and again before the termination of treatment. Haematology (including methaemoglobin from 6 months) and clinical chemistry parameters were measured from 20 rats of each sex per group at 3, 6, 12 and 18 months (there was no information on fasting status). At the time of the terminal necropsies, blood samples were collected and analysed from 20 rats of each sex per group, or from all surviving animals when less than 20 animals remained in a group. The same animals were bled on each occasion when possible. White blood cell differential count was conducted for controls and animals at the highest dose only. At 12 months, 10 rats of each sex per group were necropsied and organ weights were recorded. All rats surviving to termination were necropsied and organ weights recorded. An extensive histopathological examination was performed for all controls and animals at the highest dose at sacrifice at 12 months and at termination, and on all animals not surviving to termination. At scheduled necropsy of animals at the lowest and intermediate doses, histopathological examination was limited to kidneys, liver, lungs, gross lesions and any tissues with potentially treatment-related lesions in animals at the highest dose. Mammary glands and pituitary glands of all females were examined histopathologically. An independent peer review by a second pathologist confirmed the histopathological findings.
Survival was poor in all groups, but overall this was considered not to have a significant impact on the interpretation of the results. There was an indication that time to 50% mortality was reduced at 20 000 mg/kg (Table 17). Less than 50% survival at 2 years (as recommended in the OECD guideline 453) is a recognized problem with the strain of rat used in this study (Charles River Sprague-Dawley rats, Crl:CD®BR).
Table 17. Survival data for rats given diets containing methoxyfenozide in a study of chronic toxicity and carcinogenicity
|
Dietary concentration (mg/kg) |
Weeks to 50% survival |
Duration of treatment at terminal necropsy |
Survivors at necropsy |
Survival (%) |
|
Males |
||||
|
0 |
88-89 |
99 |
16 |
27 |
|
200 |
85 |
99 |
18 |
30 |
|
8 000 |
85-86 |
99 |
20 |
33 |
|
20 000 |
82 |
89 |
17 |
28 |
|
Females |
||||
|
0 |
89-90 |
95 |
18 |
30 |
|
200 |
90 |
95 |
22 |
37 |
|
8 000 |
83 |
95 |
16 |
27 |
|
20 000 |
80 |
95 |
19 |
32 |
From Anderson & Gillette (1998)
No substance-related clinical signs of toxicity were noted at any concentration. Body weight was decreased in females at 20 000 mg/kg from week 45 onwards, which was statistically significant (p < 0.05) from week 73. Cumulative body-weight gain in males at the highest dose was reduced by 2.5% at week 53 and by 6% at termination and was not considered to be an adverse effect. There were no effects on food consumption.
Statistically significant, minimal to slight haematological changes were observed at 8000 mg/kg and 20 000 mg/kg in both sexes (Table 18). At 8000 mg/kg, erythrocyte count was decreased in both sexes, and haemoglobin concentration and erythrocyte volume fraction were decreased in males at week 14. At 20 000 mg/kg, erythrocyte counts, haemoglobin, and erythrocyte volume fraction were decreased during the first year of treatment. A slight increase in platelets and a minimal increase in methaemoglobin formation was seen at 20 000 mg/kg during the second year of treatment. The only clinical chemistry finding of note was an increase in plasma gamma-glutamyl transferase at >8000 mg/kg, consistent with the observed hepatotoxicity. Statistically significant decreases in bilirubin and alkaline phosphatase were seen at >8000 mg/kg, but were not considered to be of toxicological importance. There were no consistent effects on any urine analysis parameters. Ophthalmoscopic examination at termination of treatment indicated there were no ocular lesions associated with treatment.
Table 18. Haematology findings in rats given diets containing methoxyfenozide for up to 99 weeks
|
Parametera |
Dietary concentration (mg/kg) |
|||
|
0 |
200 |
8 000 |
20 000 |
|
|
Males |
||||
|
Erythrocyte count (106/µl) |
||||
|
Week 14s |
7.67 |
7.57 |
7.16* |
6.96* |
|
Week 54 |
8.53 |
8.19 |
8.28 |
7.93* |
|
Week 78 |
7.38 |
7.89 |
7.79 |
7.14 |
|
Termination |
7.42 |
7.02 |
7.21 |
6.99 |
|
Haemoglobin (g/dl) |
||||
|
Week 14 |
14.5 |
14.3 |
13.6* |
13.3* |
|
Week 54 |
15.4 |
14.9 |
15.2 |
14.7* |
|
Week 78 |
14.1 |
14.6 |
14.2 |
13.8 |
|
Termination |
14.2 |
13.6 |
13.4 |
13.3 |
|
Erythrocyte volume fraction (l/l) |
||||
|
Week 14 |
0.442 |
0.438 |
0.414* |
0.406* |
|
Week 54 |
0.495 |
0.480 |
0.484 |
0.467* |
|
Week 78 |
0.441 |
0.465 |
0.451 |
0.427 |
|
Termination |
0.440 |
0.420 |
0.420 |
0.413 |
|
Platelet (103/µl) |
||||
|
Week 14 |
960 |
961 |
993 |
943 |
|
Week 54 |
913 |
1005 |
941 |
930 |
|
Week 78 |
930 |
1039 |
987 |
1082 |
|
Termination |
1112 |
1258 |
1210 |
1270 |
|
Methaemoglobin (%) |
||||
|
Week 14 |
ND |
ND |
ND |
ND |
|
Week 54 |
0.7 |
0.6 |
0.6 |
0.9* |
|
Week 78 |
1.2 |
0.8 |
1.1 |
1.2 |
|
Termination |
0.7 |
0.7 |
0.9 |
0.9* |
|
Females |
||||
|
Erythrocyte count (106/µl) |
||||
|
Week 14 |
6.91 |
6.86 |
6.57* |
6.33* |
|
Week 54 |
7.52 |
7.36 |
7.40 |
7.07* |
|
Week 78 |
7.03 |
7.01 |
6.73 |
6.68 |
|
Termination |
6.84 |
6.77 |
6.73 |
6.96 |
|
Haemoglobin (g/dl) |
||||
|
Week 14 |
14.2 |
13.9 |
13.6 |
13.1* |
|
Week 54 |
15.4 |
15.0 |
15.1 |
14.3* |
|
Week 78 |
14.7 |
14.6 |
14.3 |
13.9 |
|
Termination |
14.1 |
14.1 |
13.8 |
14.1 |
|
Erythrocyte volume fraction (l/l) |
||||
|
Week 14 |
0.433 |
0.432 |
0.417 |
0.405* |
|
Week 54 |
0.480 |
0.469 |
0.475 |
0.453 |
|
Week 78 |
0.447 |
0.447 |
0.432 |
0.422 |
|
Termination |
0.429 |
0.431 |
0.423 |
0.432 |
|
Platelets (103/µl) |
||||
|
Week 14 |
897 |
903 |
904 |
948 |
|
Week 54 |
829 |
893 |
844 |
970* |
|
Week 78 |
836 |
853 |
869 |
961* |
|
Termination |
926 |
1020 |
948 |
1027 |
|
Methaemoglobin (%) |
||||
|
Week 14 |
ND |
ND |
ND |
ND |
|
Week 54 |
0.6 |
0.5 |
0.6 |
0.8 |
|
Week 78 |
1.2 |
0.9* |
1.1 |
1.2 |
|
Terminal |
0.8 |
0.8 |
0.8 |
1.1* |
From Anderson & Gillette (1998)
a Means of 20 animals per dose per time-point, except at termination, when there were 15-20 animals per dose
* Significant difference from control (p < 0.05); ANOVA, Dunnett t-test
ND, Not determined
Increases in relative and absolute liver weights were seen at >8000 mg/kg in both sexes, with an increase in female adrenal weights at 20 000 mg/kg (Table 19). No treatment-related gross changes were seen. Primary target organs were kidney (glomerular nephropathy), liver (hepatocellular hypertrophy) and thyroid (follicular hyperplasia) (Tables 20 and 21). Effects were seen consistently at >8000 mg/kg. An increase in the severity of glomerular nephropathy at 200 mg/kg did not fit the dose-response relationship and was considered to be incidental. There were also several substance-related pathological changes in females at the highest dose, which the study investigators considered were secondary to uraemia that occurred as a result of severe chronic progressive glomerulonephropathy. These included increases in mineralization of the heart, aorta and structures in the kidney; fibrous osteodystrophy of the bone. Acute to subacute inflammation and erosion/ulceration of the forestomach and mineralization and associated giant cell inflammation of the glandular stomach were also increased at 8000 mg/kg (Table 20).
Table 19. Mean liver and adrenal weights in rats given diets containing methoxyfenozide for up to 99 weeks
|
Dietary concentration (mg/kg) |
||||||||
|
0 |
200 |
8 000 |
20 000 |
|||||
|
12 months |
Termination |
12 months |
Termination |
12 months |
Termination |
12 months |
Termination |
|
|
Males |
||||||||
|
Liver |
||||||||
|
Absolute weight (g) |
22.9 |
18.9 |
26.1 |
20.0 |
27.6 |
19.9 |
32.2* |
23.2* |
|
Relative weight (%) |
3.1 |
2.6 |
3.3 |
2.5 |
3.7* |
2.8 |
4.1* |
3.05 |
|
Adrenals |
||||||||
|
Absolute weight (g) |
0.06 |
0.08 |
0.06 |
0.08 |
0.08* |
0.12 |
0.07 |
0.10 |
|
Relative weight (%) |
0.008 |
0.01 |
0.008 |
0.0110 |
0.0110 |
0.017 |
0.009 |
0.013 |
|
Females |
||||||||
|
Liver |
||||||||
|
Absolute weight (g) |
15.9 |
16.4 |
15.91 |
14.9 |
16.4 |
17.5 |
17.7 |
16.6 |
|
Relative weight (%) |
3.3 |
2.6 |
3.3 |
2.7 |
3.6 |
2.9 |
3.8* |
3.3* |
|
Adrenals |
||||||||
|
Absolute weight (g) |
0.08 |
0.120 |
0.07 |
0.14 |
0.08 |
0.12 |
0.12* |
0.15 |
|
Relative weight (%) |
0.017 |
0.016 |
0.016 |
0.025 |
0.017 |
0.021 |
0.025* |
0.032* |
From Anderson & Gillette (1998)
* Significant difference from control (p < 0.05); ANOVA, Dunnett t-test
Table 20. Histopathology findings in rats given diets containing methoxyfenozide for up to 99 weeks
|
Finding |
Dietary concentration (mg/kg) |
|||
|
0 |
200 |
8 000 |
20 000 |
|
|
At sacrifice at 12 months: |
||||
|
Males |
||||
|
Chronic progressive glomerulonephropathy |
||||
|
Minimal |
6/10 |
2/10 |
6/10 |
2/10 |
|
Slight |
0/10 |
3/10 |
4/10 |
0/10 |
|
Moderate/moderately severe |
1/10 |
3/10 |
0/10 |
8/10 |
|
Total incidence |
7/10 |
8/10 |
10/10 |
10/10 |
|
Periportal hepatocellular hypertrophy |
0/10 |
0/10 |
8/10 |
10/10 |
|
Thyroid hypertrophy, follicular |
2/10 |
0/10 |
4/10 |
1/10 |
|
Thyroid, altered colloid |
1/10 |
1/10 |
4/10 |
3/10 |
|
Females |
||||
|
Chronic progressive glomerulonephropathy |
5/10 |
2/10 |
4/10 |
8/10 |
|
Periportal hepatocellular hypertrophy |
0/10 |
0/10 |
6/10 |
8/10 |
|
Thyroid hypertrophy, follicular |
1/10 |
0/10 |
4/10 |
5/10 |
|
Thyroid, altered colloid |
1/10 |
0/10 |
0/10 |
4/10 |
|
All animals: |
||||
|
Males |
||||
|
Kidney, hyperplasia, epithelial, pelvis |
10/70 |
9/70 |
5/70 |
15/70 |
|
Periportal hepatocellular hypertrophy |
1/70 |
0/70 |
29/70*** |
42/70*** |
|
Thyroid hypertrophy, follicular |
10/70 |
5/68 |
19/70* |
19/69* |
|
Thyroid, altered colloid |
8/70 |
11/68 |
17/70* |
17/69* |
|
Females |
||||
|
Chronic progressive glomerulonephropathy |
||||
|
Moderate to severe |
8/70 |
7/70 |
6/70 |
14/70 |
|
Total incidence |
51/70 |
50/70 |
53/70 |
60/70* |
|
Kidney, hyperplasia, epithelial, pelvis |
10/70 |
17/70 |
14/70 |
23/70** |
|
Periportal hepatocellular hypertrophy (minimal to moderate) |
2/70 |
2/70 |
30/70*** |
50/70*** |
|
Thyroid hypertrophy, follicular |
10/70 |
4/70 |
14/70 |
22/70* |
|
Thyroid, altered colloid |
11/70 |
3/70 |
11/70 |
27/70** |
|
Aorta, mineralization |
0/70 |
2/70 |
1/70 |
6/69* |
|
Heart, mineralization |
0/70 |
2/70 |
1/70 |
5/70* |
|
Kidney, mineralization |
0/70 |
2/70 |
0/70 |
6/70* |
|
Bone, fibrous osteodystrophy |
0/70 |
2/70 |
1/70 |
5/70* |
|
Forestomach, inflammation |
2/70 |
2/70 |
5/70 |
9/70* |
|
Forestomach, oedema |
2/70 |
3/70 |
6/70 |
8/70* |
|
Forestomach, erosion/ulceration |
2/70 |
2/70 |
4/70 |
7/70* |
|
Glandular stomach, mineralization |
0/70 |
2/70 |
1/70 |
6/70* |
|
Glandular stomach, inflammation, giant cell |
0/70 |
1/70 |
1/70 |
5/70* |
From Anderson & Gillette (1998)
*p < 0.05; **p < 0.01; ***p < 0.001
Table 21. Incidence of neoplastic histopathology in rats given diets containing methoxyfenozide for up to 99 weeks (animals surviving to termination)
|
Finding |
Dietary concentration (mg/kg) |
|||
|
0 |
200 |
8 000 |
20 000 |
|
|
Males |
||||
|
Liver |
||||
|
Hepatocellular adenoma |
0/70 |
0/70 |
3/70 |
2/70 |
|
Hepatocellular carcinoma |
3/70 |
0/70 |
2/70 |
1/70 |
|
Thyroid |
||||
|
C-cell adenoma |
2/70 (1/16) |
2/68 (1/18) |
10/70a (7/20) |
4/69 (2/17) |
|
C-cell carcinoma |
0/70 |
0/68 |
0/70 |
0/69 |
|
Females |
||||
|
Liver |
||||
|
Hepatocellular adenoma |
0/70 (0/18) |
1/70 (1/22) |
2/70 (2/16) |
4/70b (3/19) |
|
Hepatocellular carcinoma |
0/70 |
0/70 |
0/70 |
0/70 |
|
Thyroid |
||||
|
C-cell adenoma |
2/70 |
2/70 |
4/70 |
4/70 |
|
C-cell carcinoma |
0/70 |
1/70 |
0/70 |
0/70 |
|
Mammary gland |
||||
|
Adenoma |
3/68 |
4/69 |
3/69 |
3/68 |
|
Fibroadenoma |
28/68 |
24/69 |
25/69 |
19/68 |
|
Adenocarcinoma |
6/68 (0/18) |
17/69c (1/21) |
16/69d(0/15) |
8/68 (1/19) |
|
Sum of these three tumours |
33/68 |
38/69 |
36/69 |
25/68 |
From Anderson & Gillette (1998)
a p = 0.0168 (survival-adjusted analysis)
b p = 0.0275 (survival-adjusted analysis)
c p = 0.0337 (survival-adjusted analysis)
d p = 0.0156 (survival-adjusted analysis)
There was a slightly increased incidence of hepatocellular adenoma in females at the highest dose (Table 21). The incidence in this group (5.7% of all females, 6.7% of females at final sacrifice plus early deaths) is consistent with ranges for historical controls for hepatocellular adenoma in this strain of Charles River rat (females, 0.8-13.3%, males, 1.4-8.0% (Giknis & Clifford, 2001); females, 1.4-21.7%, males, 0-16.7% (McMartin et al., 1992). These historic control data are not perfect as they are from a range of test facilities and are not specific to the laboratory that performed the main study. The adenomas observed in this study were reported to be of similar appearance and size to that of spontaneous tumours. There was no increase in mitotic figures, the incidence/severity of altered foci or hepatocellular carcinoma. There was no increase in the incidence of hepatocellular tumours in males. Overall, it is considered that although there was marked liver hypertrophy in females at the highest dose (Table 20), there was no clear substance-related increase in the incidence of hepatic tumours in this study. The increased incidence (14.3%) of thyroid C-cell adenoma in males at 8000 mg/kg bw (Table 21) was not part of a dose-response relationship and was just within the range of incidence for historic controls reported for males of this strain by Giknis & Clifford, 2001 (1.4-14.3%), but slightly exceeded that reported by McMartin et al., 1992 (2.9-8.7%). Overall, it was considered that there was no substance-related increase in the incidence of thyroid tumours in this study. Statistically significant increased incidences of mammary gland adenoma were seen in females at 200 and 8000 mg/kg bw (Table 21). This is, however, not considered to be a substance-related effect because of the lack of a dose-response relationship across groups that showed similar survival to termination. It is also notable that on the basis of the number of females with mammary gland adenocarcinoma, or the related adenoma and fibroadenoma, none of the test groups showed an increased incidence compared with that in the negative control group (Table 21). The incidence of mammary gland adenocarcinoma in females at 200 and 8000 mg/kg bw (23-25% of all females) was well within the historical ranges for females of this strain—6.7-30% (McMartin et al., 1992) and 8.6-58.3% (Giknis & Clifford, 2001).
Within the limitations of the study (reduced survival, generic historic control data) it is considered that there was no clear oncogenic response at doses of up to 20 000 mg/kg (equal to 1045 mg/kg bw per day). The overall NOAEL was 200 mg/kg (equal to 10 mg/kg bw per day) on the basis of reductions in erythrocyte parameters, increased incidence of histopathological changes in the liver (periportal hepatocyte hypertrophy) and thyroid (follicular hypertrophy and altered colloid) at 8000 mg/kg (equal to 411 mg /kg bw per day) (Anderson & Gillette, 1998).
Dogs
No long-term studies in dogs were submitted.
Methoxyfenozide has been investigated for its ability to induce gene mutations in bacteria and mammalian cells in vitro, for the induction of chromosomal aberrations in mammalian cells and in an assay for micronucleus formation in mice in vivo (Table 22). Precipitation of methoxyfenozide limited the maximum concentration that could be used. All studies gave negative results, and complied with the OECD guideline current at the time of performance and with GLP. A repeat Ames test was performed because the original test did not use strains (TA102 or E. coli WP2) regarded as being sensitive to hydrazines (methoxyfenozide is a hydrazine derivative, as are some impurities of methoxyfenozide). The purity of the material tested was greater than that of the proposed technical specification, but the impurity profile (qualitative and quantitative) of the technical material (97%) did not give rise to any significant concerns regarding genotoxicity. The results of an Ames test and assay for micronucleus formation in vivo with a flowable formulation (240SC) were negative. The Meeting concluded that methoxyfenozide (technical material) was unlikely to be genotoxic.
Table 22. Results of studies of genotoxicity with methoxyfenozide
|
End-point |
Test object |
Concentration/dose |
Purity (%) |
Results |
Reference |
|
In vitro |
|||||
|
Reverse mutation |
S. typhimurium TA98, TA100, TA1535, TA1537 |
50-5000 µg/plate ± S9 |
98 |
Negative +S9, negative -S9 |
Sames & Streelman (1995) |
|
Reverse mutation |
S. typhimurium TA98, TA100, TA102, TA1535, TA1537 |
50-5000 µg/plate ± S9 |
99.2 |
Negative +S9, negative -S9 |
Sames & Ciaccio (1998) |
|
Reverse mutation |
S. typhimurium TA98a, TA100, TA102, TA1535, TA1537 |
50-5000 µg/plate + S9 |
23.3; formulation 2F 240SC |
Negative +S9, negative -S9 |
Sames & Ciaccio (1996) |
|
Gene mutation |
Chinese hamster ovary cells, Hprt locus |
0.5-100 µg/ml ± S9b |
99.2 |
Negative +S9, negative -S9 |
Pant (1994) |
|
Chromosome aberrations |
Chinese hamster ovary cells |
13-150 µg/ml ± S9c (multiple exposure and sample times) |
99.2 |
Negative +S9, negative -S9 |
Kumaroo (1994) |
|
In vivo |
|||||
|
Micronucleus formation |
CD-1 mice (five of each sex per group), bone-marrow cells |
0, 500, 2500, 5000 mg/kg bw, by gavage, in methylcellulose |
99 |
Negative |
Sames & Black (1995) |
|
Micronucleus formation |
CD-1 mice (five of each sex per group), bone-marrow cells |
0, 1250, 2500, 5000 mg/kg bw, by gavage, in methylcellulose |
23.6; formulation 2F |
Negative |
Sames & Ciaccio (1997) |
S9, 9000 × g supernatant fraction of rodent liver
a Performed as a separate assay, owing to contamination
b Precipitation at 100 µg/ml; considerable variation in mutation rates between duplicate culture
c Precipitation and reduced mitotic index at 150 µg/ml
In a two-generation study that complied with GLP and OECD test guideline 416 (1983) and also included some additional investigations (i.e. sperm tests, estrous cycling and developmental milestones), groups of Crl:CD®BR rats (one litter per generation) were given diets containing methoxyfenozide (purity, 98.0%) at a concentration of 0, 200, 2000, or 20 000 mg/kg active substance. Satisfactory stability, content and homogeneity were demonstrated. Mean intake of methoxyfenozide is shown in Table 23. Doses were selected on the basis of the findings of the 90-day dietary study in rats.
Table 23. Mean intake of methoxyfenozide in a two-generation study in rats
|
Intake (mg/kg bw per day) |
|||
|
200 |
2 000 |
20 000 |
|
|
Males |
|||
|
P1 premating |
15.4 |
153.4 |
1551.9 |
|
P2 premating |
19.1 |
193.1 |
1956.0 |
|
Females |
|||
|
P1 premating |
17.9 |
180.7 |
1820.7 |
|
P2 premating |
20.4 |
202.7 |
2036.5 |
|
P1 gestation |
14.7 |
147.9 |
1563.7 |
|
P2 gestation |
14.1 |
143.0 |
1474.0 |
|
P1 lactation |
26.7 |
263.6 |
2 657.4 |
|
P2 lactation |
26.1 |
268.7 |
2612.6 |
From O'Hara et al. (1997)
Exposure of P1 animals (30 of each sex per dose; not littermates) began when the animals were aged approximately 6 weeks. P2 animals (30 of each sex per dose) were randomly selected from P1 offspring (designated F1) and given treated diet from weaning. P1 and P2 animals were mated after at least 10 weeks of exposure.
Body weight, food consumption, and clinical signs were monitored in parental animals throughout treatment. Estrus cycling was evaluated in females for 3 weeks before mating. Parental animals were necropsied after weaning of their offspring. Sperm evaluation was performed for all P1 and P2 males at the time of necropsy. Weights of the liver and organs of the reproductive system were determined in parental animals. Reproductive tissues, pituitary and liver were examined histopathologically in all control animals and animals at the highest dose, and in all animals found dead or sacrificed during the course of the study. Tissues in which treatment-related lesions were identified were also examined in all animals at the lowest and intermediate doses. In addition, reproductive tissues were examined in animals at the lowest and intermediate doses that were suspected to have reduced fertility. All tissues with gross pathological changes were examined microscopically. Litters were culled to eight pups (four of each sex per litter, where possible) on day 4 of lactation. Stillborn pups, pups that died during lactation, pups culled at day 4, or sacrificed at weaning were examined grossly. Sexual maturation (age at preputial separation in males, vaginal patency in females) was evaluated in F1 offspring selected as P2 parental animals, and F2 females (one per litter). Anogenital distance was measured in all F2 offspring on the day of birth.
There were no substance-related deaths or clinical signs of systemic toxicity in parental animals or in pups during the lactation period. Decreased body-weight gain was observed at 20 000 mg/kg in P1 males (about 10%) and to a lesser extent in P2 males (about 3%), beginning at week 8 of treatment and continuing throughout the remaining treatment period. There were no adverse substance-related effects on female body weight. Food consumption was similar in all groups, statistically significant increased feed consumption during gestation (by up to 12-16% in the P1 animals at the highest dose) were linked to increased body weights, showed no clear dose-related trend and are considered to be incidental findings.
There were no substance-related effects on estrous cycling, sperm parameters, mating performance, litter size, pup body weight or pup viability. An apparent increase in the number of stillborn F1 pups was reversed for the F2 generation and was not considered to be an adverse effect of treatment (Table 24). There was no consistent effect on pup sex ratio. A statistically significant reduction in the proportion of male pups in the F2 offspring at day 0 after exposure to methoxyfenozide at 20 000 mg/kg was considered to be incidental. Although the proportion of F2 male pups at 20 000 mg/kg (44.7%) was slightly below the laboratory's lowest range for historical controls (47.7-53.4%), the statistical results were skewed as the control value (56.5%) was also outside the range for historical controls (Table 24). In addition, there was no evidence of a male-specific effect in utero (no decrease in the number of pups delivered per litter) and no evidence of feminization of male offspring, as determined by anogenital distance (Table 24). There were no adverse gross findings in F1 or F2 offspring at any dietary concentration. There were no substance-related effects on sexual maturation in F1 males (age at preputial separation) at any dietary concentration.
Table 24. Litter findings (means) in rats given diets containing methoxyfenozide in a two-generation study
|
Parameter |
Intake (mg/kg of feed) |
|||
|
0 |
200 |
2 000 |
20 000 |
|
|
P1 pups stillborn |
0/259 |
2/312 |
2/373 |
7/320* |
|
(0%)b |
(0.6%) |
(0.5%) |
(2.2%) |
|
|
P2 pups stillborn |
8/311 |
2/381* |
3/360 |
2/367* |
|
(2.6%) |
(0.5%) |
(0.8%) |
(0.5%) |
|
|
P1 litter size |
11.8 |
13.6 |
13.8 |
13.9 |
|
P2 litter size |
13.0 |
13.6 |
15.0 |
13.6 |
|
F1, % male pups |
||||
|
Day 0 |
46.3% |
52.3% |
52.2% |
48.3% |
|
Day 21 |
46.9% |
48.6% |
49.6% |
48.5% |
|
F2,% male pups |
||||
|
Day 0 |
56.5% |
50.2% |
52.5% |
44.7%* |
|
Day 21 |
55.3% |
48.5% |
50.3% |
49.0% |
|
F1, pup weight, day 21 (g) |
59.7 |
60.2 |
58.3 |
59.5 |
|
F2, pup weight, day 21 (g) |
58.1 |
61.0 |
61.4 |
58.3 |
|
F1, days to vaginal patency (n = 22-25) |
30.9 ± 0.42 (range, 28-35) |
32.0 ± 0.42 |
31.6 ±0.42 |
34.0* ± 0.55 (range, 29-42) |
|
F2, days to vaginal patency (n = 21-28) |
31.8 ±0.39 (range, 29-35) |
31.9 ±0.51 |
31.6 ±0.43 |
33.3 ± 0.54 (range, 29-39) |
|
Body weight on day of vaginal patency (g)a |
125.0 |
125.3 |
124.6 |
128.3 |
|
F2 female pups, day 0, (n = 22-28) |
||||
|
Anogenital distance (mm) |
1.06 |
1.11 |
1.13* |
1.10 |
|
F2 male pups, day 0, (n = 24-28) |
||||
|
Anogenital distance (mm) |
2.50 |
2.51 |
2.55 |
2.53 |
From O'Hara et al., (1997)
*p < 0.05 (Fisher's exact test, Mann-Whitney U test or ANOVA with Dunnett t-test)
a Test laboratory's historical control data for the same strain of rat (five studies for 1995-2000): mean days to vaginal patency = 31.3-32.6 days; mean weight at day of vaginal patency =111-128 g
A slight delay in attainment of vaginal patency was noted in F1 females (statistically significant) and in F2 females (not statistically significant) at 20 000 mg/kg (Table 24), the values being outside the range for historical data for the test facility. There was no indication that this might be linked with outliers or be secondary to reduced body weight or general pup development. However, there was no effect on mating or reproductive success subsequent to this delay. There were no specific effects on the weight of the reproductive organs examined; increases in relative testes and epididymal weights were secondary to reduced body weights. There were no pathological changes in the reproductive organs examined. A significant increase in absolute and relative liver weight (13-27%), increased incidences of hepatocellular hypertrophy (periportal or midzonal) and pigmentation of Kupffer cells were seen in parental animals exposed at 20 000 mg/kg. A statistically significant but slight (7%) increase in relative liver weight seen in P1 males and mild hepatocellular hypertrophy in four out of 30 P1 females at 2000 mg/kg were not considered to be adverse effects.
The minimal effects on the liver at 2000 mg/kg in parental (P1) animals at 2000 mg/kg was not considered to be an adverse effect. The NOAEL for general systemic toxicity was therefore 2000 mg/kg (equal to 143 mg/kg bw per day) on the basis of reduced body-weight gain, increased liver weight (>10%) and histopathological changes in liver (hepatocellular hypertrophy and vacuolation, pigment in Kupffer cells) at 20 000 mg/kg.
The NOAEL for reproductive outcome was 20 000 mg/kg (equal to 1474 mg/kg bw per day), the highest dose tested.
The NOAEL for pup development was 2000 mg/kg (equal to 143 mg/kg bw per day) on the basis of a slight delay in attainment of vaginal patency in pups at the highest dose that was seen in both generations, with no subsequent effect on reproduction or general development (O'Hara et al., 1997).
Rats
Groups of 25 mated female Crl:CD BR Sprague-Dawley rats were given methoxyfenozide (purity, 99.2%) at a dose of 0, 100, 300 or 1000 mg/kg bw per day on days 6-15 of gestation (day 0 = plug observed) by gavage in aqueous carboxymethylcellulose. Dams were sacrificed on day 20 of gestation and fetuses were examined. Approximately half of the fetuses in each litter were examined for visceral abnormalities by serial sectioning, the remainder were examined for skeletal abnormalities after staining with alizarin red. This study complied with GLP and OECD guideline 414 (1981).
No animals died and no clinical signs of toxicity were observed. No treatment-related effects were seen on maternal body weights, body-weight gain or food consumption. Gross necropsy of dams did not reveal any treatment-related findings. No treatment-related effects were seen on reproductive or litter parameters (including percentage of live male fetuses). Total incidences of fetal alterations were significantly lower in treated groups. Ossification of a number of bones was found to be greater in treated groups (Table 25). The finding of a single fetus with multiple abnormalities at the highest dose was not considered to be a treatment-related finding. The incidence of bifid thoracic vertebral centra was marginally increased at the highest dose; however, values did not attain statistical significance (Table 25). The litter and fetal incidence for this lesion at the highest dose were very similar to means for historical controls reported for this strain of rat by MARTA and MTA databases (1996), (Table 25), and it was considered that the marginally increased incidence of bifid thoracic centra at the highest dose in the present study was not of clear toxicological significance.
Table 25. Developmental findings in rats given methoxyfenozide by gavage during organogenesis
|
Parameter |
Dose (mg/kg bw per day) |
|||||||
|
0 |
100 |
300 |
1000 |
|||||
|
Reproductive parameters |
||||||||
|
No. pregnant |
25 |
25 |
22 |
24 |
||||
|
Corpora lutea |
18.1 |
17.7 |
17.5 |
17.4 |
||||
|
Implantations |
15.1 |
15.1 |
14.3 |
14.9 |
||||
|
Number of litters |
25 |
25 |
22 |
24 |
||||
|
Litter parameters |
||||||||
|
Litter size |
14.7 |
14.7 |
13.6 |
14.5 |
||||
|
Fetal viability |
100% |
100% |
100% |
100% |
||||
|
Fetal weight |
3.33 |
3.38 |
3.36 |
3.36 |
||||
|
Live male fetuses/litter |
54.8% |
47.4% |
54.7% |
49.5% |
||||
|
Fetal parameters: % litter incidence (% fetal incidence) |
||||||||
|
Fetuses with alteration (total) |
44.0 |
(6.8) |
44.0 |
(4.9) |
18.2 |
(1.3**) |
20.8 |
(3.2**) |
|
*, Ossification, lumbar arch |
16.0 |
(2.2) |
0.0** |
(0.0**) |
4.5** |
(0.6**) |
0.0** |
(0.0**) |
|
*, Ossification, sternebrae |
12.0 |
(3.8) |
8.0 |
(1.0**) |
0.0 |
(1.0**) |
4.2 |
(1.1**) |
|
*, Ossification, pubes |
28.0 |
(8.1) |
28.0 |
(5.3) |
9.1 |
(1.3**) |
8.3 |
(2.2**) |
|
*, Ossification, ischia |
16.0 |
(2.7) |
8.0 |
(2.6) |
0.0 |
(0.0**) |
0.0 |
(0.0**) |
|
Bifid thoracic centraa |
4.0 |
(0.5) |
4.0 |
(0.5) |
4.5 |
(0.6) |
12.5 |
(1.6) |
From Hoberman (1994)
*, Decreased
** Significantly different to controls (p < 0.01)
a MARTA and MTA (1996): historical control data: average litter incidence, 8% (maximum value, 75%); average fetal incidence, 1.2% (maximum value, 10.7%)
No evidence of teratogenicity was seen. The NOAEL for maternal toxicity was 1000 mg/kg bw per day on the basis of the absence of toxicity at the highest dose of 1000 mg/kg bw per day (limit dose). The NOAEL for developmental toxicity was 1000mg/ kgbw per day (Hoberman, 1994).
Rabbits
Groups of 16 mated female New Zealand white rabbits were given methoxyfenozide (purity, 98%) at a dose of 0, 100, 300, or 1000 mg/kg bw per day on days 7-19 of gestation (day 0 = day of mating) in aqueous carboxymethylcellulose by gavage. Dams were sacrificed on day 29 of gestation and fetuses were examined after caesarean section. All fetuses were examined for visceral abnormalities and for skeletal abnormalities after staining with alizarin red. This study complied with OECD guideline 414 (1981) and with GLP.
One control animal died after an intubation error and one animal at 300 mg/kg bw per day aborted on day 22 of gestation. No clinical signs of toxicity were noted. Mean body-weight gain of animals at the highest dose was decreased during days 20-29, but this was seen as a correction for increased body-weight gain during the dosing period (Table 26). No treatment-related gross lesions were detected on examination of the does post mortem. No treatment-related effects on reproductive or litter parameters were seen. The slightly lower mean litter size at the highest dose reflects the lower numbers of corpora lutea and implantation sites and was not treatment related. A statistically significant decrease in the number of live male fetuses was seen at 1000 mg/kg bw per day; however, this finding was not considered to be substance-related because the value was within the range for normal variation and the control group had an unusually high number and percentage of male fetuses compared with historical controls (Table 26). No substance-related skeletal malformations were noted.
Table 26. Summary of findings in a study of developmental toxicity in rabbits
|
Parameter |
Dose (mg/kg bw per day) |
|||||||
|
0 |
100 |
300 |
1000 |
|||||
|
Reproductive parameters |
||||||||
|
Weight gain (days 0-29) |
280.3 |
364.7 |
317.4 |
292.4 |
||||
|
Weight gain (days 7-20) |
143.1 |
159.6 |
146.6 |
193.5 |
||||
|
Weight gain (days 20-29) |
184.9 |
226.6 |
188.2 |
150.8 |
||||
|
No. pregnant |
16 |
16 |
15 |
15 |
||||
|
No. of litters |
15 |
16 |
14 |
15 |
||||
|
Litter parameters |
||||||||
|
Corpora lutea |
10.4 |
10.9 |
10.3 |
9.8 |
||||
|
Implantation sites |
9.7 |
10.0 |
9.9 |
8.8 |
||||
|
Litter size |
9.5 |
9.4 |
9.9 |
8.7 |
||||
|
Fetal viability (%) |
100 |
100 |
100 |
100 |
||||
|
Litter weight (g) |
40.1 |
43.1 |
39.0 |
42.7 |
||||
|
Live females/litter |
3.5 |
4.4 |
4.7 |
4.5 |
||||
|
Live males/litter2 |
6.1 |
5.1 |
5.1 |
4.2* |
||||
|
% live males/litter3 (%) |
63.6 |
52.1 |
52.1 |
49.5 |
||||
|
Fetal findings: % fetal incidence and [% litter incidence] |
||||||||
|
Total soft tissue observations |
4.9 |
[33.3] |
6.6 |
[50.0] |
4.3 |
[21.4] |
10.7 |
[33.3] |
|
Left carotid variation1 |
0.7 |
[6.7] |
2.0 |
[12.5] |
3.6 |
[14.2] |
8.4 |
[20.0] |
|
(1)a |
(1)b |
(3)a |
(2)b |
(5)a |
(2)b |
(11)a |
(3)b |
|
|
Total skeletal retardations |
70.6 |
[100] |
66.2 |
[100] |
72.5 |
[92.9] |
63.4 |
[93.3] |
|
Unossified sternebrae |
16.1 |
[53.3] |
9.3 |
[43.8] |
7.2 |
[35.7] |
9.9 |
[33.3] |
|
Partially ossified arches of lumbar vertebrae |
43.4 |
[93.3] |
36.5 |
[75.0] |
42.8 |
[92.9] |
35.1 |
[80.0] |
From Shuey (1997)
*p < 0.05, a = fetuses,b = litters; with one or more fetuses with left carotid arising from innominate
Historical control range data for the test laboratory
1 Left carotid artery arising from innominate: 3.3-9.0% (fetal incidence); 13-46% (litter incidence)
2 Number of live males/litter (1990-1996): 4.1-4.7 (litter incidence)
3 Live males/litter (1990-1996): 50.3-57.8% (litter incidence)
Skeletal retardations were slightly decreased at the highest dose owing to the increased ossification of sternebrae and vertebrae; this finding was not considered to constitute an adverse effect. The total fetal incidence of soft tissue findings was slightly increased at the highest dose, largely due to the increased incidence of a circulatory system alteration (left carotid artery arising from the innominate artery). This finding is classified as a developmental variation. When incidences are expressed as a percentage (Table 26), there appears to be a dose-related increase, at all doses, in the fetal incidence and litter incidence of this variation. Although the number of fetuses with the finding increased from one in the controls to 11 at the highest dose, the number of litters involved was small (increasing from only one in the controls to three at the highest dose), incidences were within the range for historical controls, and analysis of the litter incidence revealed no statistically significant differences (p > 0.05 in Fisher exact test). It was considered that the apparent increased incidence of this variation in the present study was not a toxicologically significant adverse finding.
No evidence of teratogenicity was seen. The NOAEL for maternal toxicity was 1000 mg/kg bw per day on the basis of the absence of any toxicologically significant effects at the limit dose. The NOAEL for developmental toxicity was 1000 mg/kg bw per day on the basis of the absence of clear substance-related adverse findings at the highest dose of 1000 mg/kg bw per day (limit dose) (Shuey, 1997).
(a) Acute neurotoxicity in rats
In a study that complied with GLP and that was according to US EPA guidelines, groups of 10 male and 10 female Crl:CD®BR rats (aged about 6 weeks) were given a single dose of methoxyfenozide (purity, 98%) at 0, 500, 1000, or 2000 mg/kg bw by gavage suspended in 0.5% aqueous methylcellulose at a constant volume of 20 ml/kg. Analysis of suspensions showed satisfactory concentrations of active substance. All rats were observed daily for signs of ill health or reaction to treatment and were weighed weekly. A functional observational battery (FOB) was performed on each rat pre-test and motor activity assessment was performed 7 days before dosing. FOB and motor activity testing was repeated within 3 h of dosing on day 0 and on days 7 and 14 after dosing. The time of testing on day 0 was chosen to be as close as feasible to the time of peak blood concentrations of active substance and/or metabolites. On day 14, after the final FOB and motor activity assessment, rats were perfused with neutral buffered formalin and given a limited gross necropsy. Twelve randomly selected control animals (six males and six females) and 12 randomly selected animals at the highest dose (six males and six females) received a special neuropathology evaluation that included examination of the brain, spinal cord, selected ganglia, spinal root fibres and peripheral nerves of the hindlimb. Stains used were haematoxylin and eosin, and toluidine blue. The study investigators reported that studies with six positive control substances conducted at the test laboratory in 1994 gave results consistent with the published literature.
No mortalities, substance-related clinical signs of systemic toxicity or body-weight effects were observed during the study period. There were no consistent substance-related effects on motor activity or any of the FOB parameters. Hind-limb grip strength for males at 2000 mg/kg bw was reduced statistically significantly on day 0 within 3 h of dosing; the reduction was by 24% (adjusted for pre-test mean); the corresponding mean hind-limb grip strength value for females at 2000 mg/kg bw showed a slight increase (13%). There was a statistically significant increase (by 31% after adjustment for the pre-test mean) in the total number of movements shown by males at 2000 mg/kg bw on day 14. Although slight increases were seen on days 0 and 7, they were not statistically significant, nor was the slight increase in total time spent by males in movement at 2000 mg/kg bw. In females at 2000 mg/kg bw, on day 14 the total number of movements decreased slightly (not statistically significantly) and there was no change in the total time spent in movement. Overall, there was considered to be no convincing evidence for effect on grip strength or motor activity.
Gross necropsy revealed no substance-related findings. No treatment-related histopathological alterations were observed in any of the examined areas of the central or peripheral nervous systems. A low incidence of minimal axonal degeneration, consisting of one to two focally degenerating axons, in peripheral nerves (maximum incidence, one out of six) was a consistent background finding. Methoxyfenozide, when administered orally by gavage as a single dose at doses up to and including 2000 mg/kg bw, produced no evidence for neurotoxic effects in rats. The NOAEL for neurotoxicity and neuropathology was 2000 mg/kg, the limit dose tested (Anderson & Gillette, 1996).
(b) Thirteen-week study of neurotoxicity in rats
In a study that complied with GLP and that was performed according to US EPA guidelines, groups 10 male and 10 female Crl:CD®BR rats (aged 6 weeks) were given diets containing methoxyfenozide (purity, 98%) at a concentration of 0 (control), 200, 2000, or 20 000 mg/kg for 13 weeks. These doses were equal to an average intake of 0, 13, 130, and 1318 mg/kg bw per day in males, and 0, 16, 159, 1577 mg/kg bw per day in females. Satisfactory stability, homogeneity and active substance content were demonstrated. All rats were observed daily for signs of ill health or reaction to treatment. Body weight and food consumption were monitored and physical examinations were performed weekly. Neurotoxicity testing, including FOB and motor activity assessments, were performed on all rats pre-test and on weeks 4, 8, and 13. All rats surviving to necropsy were perfused with neutral buffered formalin and given a limited gross necropsy. Histopathological evaluations of the nervous system were performed for six rats of each sex per group (at the highest dose and in the control group).
There were no substance-related mortalities or clinical signs of toxicity. The study investigators considered that there were no treatment-related effects on body weight. A slight, not statistically significant, reduction in overall body weight of 5-7% in both sexes at 20 000 mg/kg was not regarded as an adverse effect. No substance-related changes were seen in either the FOB or motor activity assessments.
There were no substance-related gross or microscopic pathological findings. Minimal axonal degeneration observed in peripheral nerves, consisting of one to two degenerating axons per nerve, was a typical background finding. There was no evidence of neurotoxic or neuropathic effects in rats receiving diets containing methoxyfenozide at concentrations of up to and including 20 000 mg/kg for 13 weeks. The NOAEL was 20 000 mg/kg (equal to 1318 mg/kg bw per day), the highest dose tested (Kane & Gillette, 1996).
The acute oral toxicity and mutagenicity of metabolite N-2-3-hdroxyenzoyl-N’-3,5dimethylbenzoyl-n'-tert-butylhydrazine (M14) have been investigated. M14 was found to be of low acute oral toxicity in CD-1 (ICR) mice in a study that complied with GLP; no deaths were seen at 5000 mg/kg bw (Parno et al, 1997).
In a study that complied with GLP, no increases in mutation rates were seen in S. typhimurium strains TA98, TA100, TA102, TA1535 or TA1537 at concentrations up to the limit value of 5000 µg/plate, in the presence and absence of metabolic activation (Sames & Ciaccio, 1998).
Commercialization of methoxyfenozide has only occurred recently and data on human exposures are limited. There have been no adverse health effects reported among manufacturing workers, users or the general population.
Orally administered [14C]methoxyfenozide is absorbed rapidly in rats; 58-77% of the administered dose was excreted within 24 h. Peak plasma concentrations of radioactivity (Cmax) were seen approximately 30 min after dosing. Excretion occurs mainly via the faeces, after absorption followed by secretion in the bile. On the basis of the quantities of radioactivity excreted in the bile and urine, approximately 60-70% of an orally administered dose of 10 mg/kg bw was absorbed. Absorption and excretion profiles were similar irrespective of dose (10 or 1000 mg/kg bw), single or repeated dosing (over 14 days) or sex, the only differences being evidence of saturation at the highest dose and a slightly increased level of urinary excretion in females. Concentrations of radioactivity at Cmax were highest in the liver, with concentration in the adrenals and in the spleen also being higher than that in whole blood.
More than 30 metabolites of methoxyfenozide were identified in rat urine, faeces and bile. The primary reactions were demethylation, glucuronidation and hydroxylation. Less than 5% of the methoxyfenozide administered was cleaved at the amide bridge between the two aromatic rings. Repeated dosing at 10 mg/kg bw for 14 days altered the metabolite profile to a limited extent, with an increase in the concentration of multiple hydroxylated compounds.
Methoxyfenozide has low acute toxicity when administered by the oral, dermal or inhalation routes. In rats, the acute LD50 was > 5000 mg/kg bw after oral or dermal administration. Methoxyfenozide was not irritating to rabbit skin, and produced minimal transient irritation of the rabbit eye. Methoxyfenozide did not induce skin in a Magnusson and Kligman maximization test for sensitization in guinea-pigs.
Short-term studies in rats, mice and dogs fed with diets containing methoxyfenozide showed that these animals tolerated high concentrations of methoxyfenozide, equivalent to about 1000 mg/kg bw per day, with no marked adverse effects. Effects seen to varying degrees in all species were increased liver weight, hepatocyte hypertrophy and alterations in erythrocyte parameters consistent with a mild haemolytic effect, accompanied by formation of methaemoglobin. Findings were not always consistent between studies in the same species and comparison was also hindered to a certain extent by variations in blood sampling procedure and in the range of parameters investigated. The NOAEL in mice was 2500 mg/kg, equal to 428 mg/kg bw per day on the basis of reduced body-weight gain at 7000 mg/kg, equal to 1149 mg/kg bw per day, in the 90-day study. The NOAEL in rats was 1000 mg/kg, equal to 69 mg/kg bw per day, on the basis of increased (by >10%) relative liver weights and periportal hepatocyte hypertrophy at 5000 mg/kg, equal to 353 mg/kg bw per day, in the 90-day study. Thyroid follicular cell hypertrophy or hyperplasia seen in the 2-week study in rats receiving methoxyfenozide at 1000 mg/kg, equal to 98 mg/kg bw per day, was not reproduced in the 90-day study. In dogs, increases in the formation of methaemoglobin and abnormal erythrocyte morphology were seen in two 14-day studies, with increases in spleen weights also noted in one of these studies. The overall NOAEL in the 14-day studies in dogs was 500 mg/kg, equal to 20 mg/kg bw per day, with a lowest-observed-adverse-effect level (LOAEL) of 3500 mg/kg, equal to 154 mg/kg bw per day. No treatment-related adverse effects were seen in a 90-day study in dogs receiving doses of up to 5000 mg/kg, equal to 198 mg/kg bw per day. In a 1-year study in dogs fed diets containing methoxyfenozide at concentrations of 0-30 000 mg/kg (0, 60, 300, 3000, 30 000 mg/kg), there was evidence of haemolysis, methaemoglobinaemia, increased concentrations of bilirubin in blood and urine and increases in numbers of platelets. The presence of increased quantities of iron-positive pigment in the liver and spleen is consistent with phagocytosis of damaged erythrocytes. The NOAEL was 300 mg/kg, equal to 9.8 mg/kg bw per day, with a LOAEL of 3000 mg/kg, equal to 106 mg/kg bw per day. Extensive reversibility of haematological effects was demonstrated in dogs examined 4 weeks after the end of a 4-week exposure to 30 000 mg/kg, equal to 1036 mg/kg bw per day, of methoxyfenozide in the diet. This is consistent with the increases in reticulocytes and bone marrow hyperplasia observed in other studies and indicated that the effects on erythrocytes are not attributable to a direct effect on stem cells.
The chronic toxicity and carcinogenicity of methoxyfenozide was investigated in mice and rats given diets containing methoxyfenozide at concentrations equating to > 1000 mg/kg bw per day in the groups receiving the highest dose. There was no treatment-related increase in the incidence of any tumour type. There were no significant treatment related non-neoplastic effects. The NOAEL for carcinogenicity and non-neoplastic effects in mice was 7000 mg/kg, equal to 1020 mg/kg per day, the highest dose tested. The Meeting concluded that methoxyfenozide is not carcinogenic in mice.
In the study in rats, poor survival (<50% at week 90) in all groups resulted in the study being terminated at 99 weeks. This reduction in the duration of exposure reduced the power of the study, but the study was considered to be adequate for the assessment of carcinogenic potential in rats. Non-neoplastic findings were consistent with those of the short-term studies. Changes in erythrocyte parameters, increases in numbers of platelets, serum gamma-glutamyl transferase activity, liver weight, hepatocyte hypertrophy, glomerular nephropathy, thyroid follicular hyperplasia and erosion of the glandular stomach were seen at doses of 8000 mg/kg, equal to 411 mg/kg bw per day, and above. The NOAEL for non-neoplastic effects was 200 mg/kg, equal to 10 mg/kg bw per day. There was no treatment-related increase in the incidence of any tumour type. The NOAEL for neoplastic effects was 20 000 mg/kg, equal to 1045 mg/kg bw per day. The Meeting concluded that methoxyfenozide was not carcinogenic in rats.
Methoxyfenozide (purity, 99%) has been investigated in an adequate range of studies of genotoxicity in vitro and in vivo and was found to give negative results. The Meeting noted that the purity of the material tested was greater than that of the proposed technical specification, but that the impurity profile (qualitative and quantitative) of the technical material (97%) did not give rise to any significant concerns regarding genotoxicity. The Meeting concluded that methoxyfenozide (technical material) was unlikely to be genotoxic.
In view of the lack of genotoxicity and the absence of carcinogenicity observed in studies in rats and mice, the Meeting concluded that methoxyfenozide is not likely to pose a carcinogenic risk to humans.
A two-generation study of reproductive toxicity in rats treated with methoxyfenozide showed that there were no adverse effects on estrus cycling, sperm parameters, mating performance, litter size, pup body weight, pup viability or pup gross pathology at doses of up to 20 000 mg/kg, equal to 1474 mg/kg bw per day. A significant increase in absolute and relative liver weights and altered liver histopathology were seen in parental animals at 20 000 mg/kg. The only compound-related effect observed in pups was a slight delay in attainment of vaginal patency, noted in both generations at a dose of 20 000 mg/kg, the values being outside the range for historical data for the test facility. The developmental delay in attainment of vaginal patency did not have any impact on reproduction at the second mating. There was no evidence that developing pups or second-generation parents were especially sensitive to methoxyfenozide. The NOAEL for reproductive effects was 20 000 mg/kg, equal to 1474 mg/kg bw per day, the highest dose tested. The NOAEL for pup development was 2000 mg/kg, equal to 143 mg/kg bw per day, on the basis of delayed vaginal patency at 20 000 mg/kg. The NOAEL for parental toxicity was 2000 mg/kg on the basis of increased liver weights and histopathological changes at 20 000 mg/kg.
The developmental toxicity of methoxyfenozide was investigated in rats and rabbits. Some marginal increases in fetal alterations were noted, but these were within the range of values for historical controls and were not of toxicological concern. There was no evidence of maternal toxicity at the limit dose of 1000 mg/kg bw per day used in both studies. The overall NOAEL was 1000 mg/kg bw per day. The Meeting concluded that methoxyfenozide is not teratogenic.
Methoxyfenozide was tested in studies of neurotoxicity, although there were no signs of neurotoxicity induced by methoxyfenozide in routine studies of toxicity. No evidence of neurotoxicity or neuropathy was seen at 2000 mg/kg bw, the highest dose tested in a study of acute neurotoxicity in rats, or at 20 000 mg/kg, equal to 1318 mg/kg bw per day, the highest dose tested in a 90-day study of neurotoxicity in rats receiving repeated doses of methoxyfenozide. No haematological investigations were performed in these studies.
The animal, soil and plant metabolite, N-2,3-hydroxybenzoyl-N’-3,5-dimethylbenzoyl-N’-tert-butylhydrazine (M14) has low acute oral toxicity in mice (LD50, >5000 mg/kg bw), and was not mutagenic in an Ames test. It is expected that the metabolites of methoxyfenozide identified in rats will be of no greater toxicity than the parent compound.
Methoxyfenozide is a new compound and there has been only limited exposure of humans to this pesticide. No adverse findings have been identified during routine medical monitoring of workers and operators.
The Meeting concluded that the existing database on methoxyfenozide was adequate to characterize the potential hazards to fetuses, infants and children.
The Meeting established an acceptable daily intake (ADI) for methoxyfenozide of 0-0.1 mg/kg bw based on the NOAELs of 200 mg/kg, equal to 10 mg/kg bw per day, for effects on erythrocytes plus liver and thyroid hypertrophy in the long-term study in rats, and 300 mg/kg, equal to 9.8 mg/kg bw per day, for haematological effects in the 1-year study in dogs, and a 100-fold safety factor.
The Meeting concluded that the toxicological profile of methoxyfenozide required the derivation of an acute reference dose (RfD). The most appropriate end-point was considered to be haematotoxicity, for which the dog is the most sensitive species. In view of the fact that a 1-day study in dogs was available for the closely related compound, tebufenozide, which has a similar toxicity profile on repeated dosing, the Meeting decided to use this study to establish the acute RfD for methoxyfenozide. An acute RfD of 0.9 mg/kg bw was established, on the basis of the lack of haematological effects at 4300 mg/kg, equal to 89.4 mg/kg bw, and using a safety factor of 100. The Meeting noted that this value was likely to be conservative since tebufenozide was more potent than methoxyfenozide in producing effects on erythrocytes.
Levels relevant to risk assessment
|
Species |
Studya |
Effect |
NOAEL |
LOAEL |
|
Mouse |
78-week study of chronic toxicity and carcinogenicity |
Toxicity and carcinogenicity |
7000 mg/kgb, equal to |
— |
|
Rat |
2-year study of chronic toxicity and carcinogenicity |
Toxicity |
200 mg/kg, equal to |
8000 mg/kg, equal to 411 mg/kg bw per day |
|
Carcinogenicity |
20 000 mg/kgb, equal to |
— |
||
|
Two-generation study of reproductive toxicity |
Parental and offspring toxicity |
2000 mg/kg, equal to |
20 000 mg/kg, equal to |
|
|
Dog |
1-year study of toxicity |
Toxicity |
300 mg/kg, equal to |
3000 mg/kg, equal to |
|
Single dose study with tebufenozide |
Toxicity |
4300 mg/kgb, equal to |
— |
a All studies investigated dietary administration of methoxyfenozide
b Highest dose tested
Estimate of acceptable daily intake for humans
0-0.1 mg/kg bw
Estimate of acute reference dose
0.9 mg/kg bw
Studies that would provide information useful for continued evaluation of the compound
Observations in humans
Summary of critical end-points for methoxyfenozide
|
Absorption, distribution, excretion and metabolism in mammals |
|
|
Rate and extent of absorption: |
About 60-70% within 72 h in the rat (including biliary excretion of 40-65%) at a dose of 10 mg/kg bw |
|
Distribution: |
Widely distributed; highest absorbed concentrations after 15min-2 h in the liver |
|
Potential for accumulation |
Low potential: <0.1% in liver after 5 days |
|
Rate and extent of excretion: |
Rapid: 60-80% in 24 h, mainly in the faeces |
|
Metabolism in animals |
Extensive (no parent found in urine or bile) |
|
Little cleavage of parent |
|
|
Toxicologically significant compounds |
Methoxyfenozide |
|
Acute toxicity |
|
|
Rat, LD50, oral |
>5000 mg/kg bw |
|
Rat, LD50, dermal |
>5000 mg/kg bw |
|
Rat, LC50, inhalation |
>4.3 mg/l (4-h exposure, nose only, maximum achievable concentration) |
|
Skin sensitization |
Not sensitizing (Magnusson & Kligman test) |
|
Short-term studies of toxicity |
|
|
Target/critical effect |
Liver (hypertrophy), erythrocytes (methaemoglobin and haemolysis) |
|
Lowest relevant oral NOAEL |
10 mg/kg bw per day (1-year study in dogs) |
|
Lowest relevant dermal NOAEL |
1000 mg/kg bw per day (28-day study in rats) |
|
Lowest relevant inhalation NOAEC |
No data |
|
Genotoxicity |
Not genotoxic |
|
Long-term toxicity and carcinogenicity |
|
|
Target/critical effect |
Erythrocytes (reduced parameters), liver (hypertrophy), thyroid (hypertrophy) |
|
Lowest relevant NOAEL |
10 mg/kg bw per day (80-90 week study in rats) |
|
Carcinogenicity |
Not carcinogenic |
|
Reproductive toxicity |
|
|
Reproduction target/critical effect |
Delayed attainment of vaginal patency |
|
Parental hepatotoxicity |
|
|
Lowest relevant NOAEL for reproductive toxicity |
143 mg/kg bw per day |
|
Developmental target/critical effect |
No embryotoxicity or fetotoxicity |
|
Not teratogenic |
|
|
Lowest relevant NOAEL for developmental toxicity |
1000 mg/kg bw per day (highest dose tested in rats and rabbits) |
|
Neurotoxicity/delayed neurotoxicity |
|
|
Acute neurotoxicity |
NOAEL: >2000 mg/kg bw; no neuropathy (rat) |
|
90-day study of neurotoxicity |
NOAEL: 1318 mg/kg bw per day (highest dose tested); no neuropathy (rat) |
|
Other toxicological studies |
|
|
Tebufenozide single dose study in dogs |
No effects at 89.4 mg/kg bw (highest dose tested) |
|
Metabolite: N-2,3-hydroxybenzoyl-N'-3,5-dimethylbenzoyl-N’-tert-butylhydrazine (M14) |
Acute LD50 >5000 mg/kg bw in mice treated orally; not mutagenic in an Ames test |
|
Medical data |
No adverse effects reported but data limited (new compound) |
|
Summary |
Value |
Study |
Safety factor |
|
ADI |
0-0.1 mg/kg bw |
Rat, long-term study; and dog, 1 -year study |
100 |
|
Acute RfD |
0.9 mg/kg bw |
Dog, single dose of tebufenozide |
100 |
Anderson, D.M. & Gillette, D.M. (1996) RH-2485 technical: Acute oral (gavage) neurotoxicity study in rats. Unpublished report No. 94R-252, 91174 from Rohm and Haas Co. Submitted to WHO by Dow Agrosciences, Indianapolis, USA.
Anderson, D.M. & Gillette, D.M. (1998) RH-2485 technical: 24-month dietary chronic/oncogenicity study in rats. Unpublished report No: 94R-226, 91173 from Rohm and Haas Co. Submitted to WHO by Dow Agrosciences, Indianapolis, USA.
Anderson, D.M., Shuey, D.L. & Lomax, L.G. (1995) RH-112,485: three-month dietary toxicity study in rats. Unpublished report No: 92R-031, 91169 from Rohm and Haas Co. Submitted to WHO by Dow Agrosciences, Indianapolis, USA.
Bannister, R.M. & Morrison, R.D. (1998) RH-2485 technical: blood recovery study in dogs. Unpublished report No. 98R-123, 91177 from Rohm and Haas Co. Submitted to WHO by Dow Agrosciences, Indianapolis, USA.
Bernacki, H. J. & Ferguson, J.S. (1995) RH-112,485: Acute inhalation toxicity study in rats. Unpublished report No. 94R-250, 91165 from Rohm and Haas Co. Submitted to WHO by Dow Agrosciences, Indianapolis, USA.
Bell, G.H., Davidson, J.N. & Emslie-Smith, D. (1972) Textbook of Physiology and Biochemistry, 8th Ed., Edinburgh and London: Churchill Livingstone.
Giknis, M.L.A. & Clifford, C.B. (2001) Compilation of spontaneous neoplastic lesions and survival in Crl:CD-1(SD)BR rats from control groups. Unpublished report from Charles River Laboratories. Submitted to WHO by Dow Agrosciences, Indianapolis, USA.
Gingrich, S.L. & Parno, J.R. (1995a) RH-112,485: acute oral toxicity study in male and female rats. Unpublished report No. 94R-247, 91159 from Rohm and Haas Co. Submitted to WHO by Dow Agrosciences, Indianapolis, USA.
Gingrich, S.L. & Parno, J.R. (1995b) RH-112,485: skin irritation study in rabbit. Unpublished report No. 94R-249, 91166 from Rohm and Haas Co. Submitted to WHO by Dow Agrosciences, Indianapolis, USA.
Gingrich, S.L. & Parno, J.R. (1995c) RH-112,485: eye irritation study in rabbits. Unpublished report No. 94R-248, 91167 from Rohm and Haas Co. Submitted to WHO by Dow Agrosciences, Indianapolis, USA.
Glaza, S.M. (1995) Dermal sensitization study of RH-112,485 technical in guinea pigs—maximization test. Unpublished report No. 94RC-0251, 91168 from Rohm and Haas Co. Submitted to WHO by Dow Agrosciences, Indianapolis, USA.
Hoberman, A.M. (1994) Developmental toxicity (embryo-fetal toxicity and teratogenic potential) study of RH-112,485 administered orally via gavage to Crl:CD BR VAF/Plus presumed pregnant rats. Unpublished report No. 92RC-026, 91179 from Rohm and Haas Co. Submitted to WHO by Dow Agrosciences, Indianapolis, USA.
Kaminski, E.J., Carbone, J.P. & Lomax, L.G. (1993) RH-2485: two-week dietary toxicity screen in female mice. Unpublished report No. 96768. Submitted to WHO by Dow Agrosciences, Indianapolis, USA.
Kaminski, E.J., Shuey, D.L. & Gillette, D.M. (1995a) RH-2485: three-month dietary toxicity study in mice. Unpublished report No. 94R-046, 91170 from Rohm and Haas Co. Submitted to WHO by Dow Agrosciences, Indianapolis, USA.
Kaminski, E.J., Shuey, D.L. & Lomax, L.G. (1995b) RH-2485: three-month dietary toxicity study in dogs. Unpublished report No. 94R-056, 91171 from Rohm and Haas Co. Submitted to WHO by Dow Agrosciences, Indianapolis, USA.
Kane, W.W. & Gillette, D.M. (1996) RH-2485 technical: three-month dietary neurotoxicity study in rats. Unpublished report No. 94R-253, 91175 from Rohm and Haas Co. Submitted to WHO by Dow Agrosciences, Indianapolis, USA.
Kumaroo, P. (1994) RH-112,485: test for chemical induction of chromosome aberration using monolayer cultures of Chinese hamster ovary (CHO) cells with and without metabolic activation. Unpublished report No. 92RC-0205, 91182 from Rohm and Haas Co. Submitted to WHO by Dow Agrosciences, Indianapolis, USA.
Kyle, M.E., Kaminski, E.J. & Kulwich, B.A. (1992) RH-112,485: two-week dietary screen in rats. Unpublished report No. 96767. Submitted to WHO by Dow Agrosciences, Indianapolis, USA.
MARTA and MTA (Middle Atlantic Reproduction and Teratology Association and Midwest Teratology Association) (1996) Historical control data (1992-1994) for developmental and reproductive toxicity studies using the Crl:CD® (SD)BR rat, Charles River Laboratories.
McMartin, D. et al. (1992) Neoplasms and related proliferative lesions in control Sprague Dawley rats from carcinogenicity studies, historical data and diagnostic considerations. Toxicol. Pathol., 20, 212-225.
Morrison, R.D. & Shuey, D.L. (1997) RH-2485 technical: one-year dietary toxicity study in dogs. Unpublished report No. 94R-257, 91186 from Rohm and Haas Co. Submitted to WHO by Dow Agrosciences, Indianapolis, USA.
Nuttall, J. & Kelly, J. (1995) Revised final report. RH 112 485: 14-day (dietary administration) dose range-finding study in the dog. Unpublished report No. 96766. Submitted to WHO by Dow Agrosciences, Indianapolis, USA.
O'Hara, G.P., Romanello, A.S. & Gillette, D.M. (1997) RH-2485 technical: two-generation reproductive toxicity study in rats. Unpublished report No. 94R-254, 91187 from Rohm and Haas Co. Submitted to WHO by Dow Agrosciences, Indianapolis, USA
Pant, K. (1994) RH-112,485: test for chemical induction of gene mutation at the HGPRT locus in cultured Chinese hamster ovary (CHO) cells with and without metabolic activation. Unpublished report No. 92RC-0204, 91185 from Rohm and Haas Co. Submitted to WHO by Dow Agrosciences, Indianapolis, USA.
Parno, J.R., Craig, L.P. & Eberly, S.L. (1997) RH-117,236: acute oral toxicity study in male and female mice. Unpublished report No. 97R-151, 91162 from Rohm and Haas Co. Submitted to WHO by Dow Agrosciences, Indianapolis, USA.
Parno, J.R., Craig, L.P. & Eberly, S.L. (1998a) RH-112,485 technical: twenty-eight day dermal toxicity study in rats. Unpublished report No. 97R-027, 91172 from Rohm and Haas Co. Submitted to WHO by Dow Agrosciences, Indianapolis, USA.
Parno, J.R., Craig, L.P. & Procopio, K.R. (1998b) RH-112,485: acute dermal toxicity study in male and female rats. Unpublished report No. 97R-268, 91164 from Rohm and Haas Co. Submitted to WHO by Dow Agrosciences, Indianapolis, USA.
Procopio, K.R. & Parno, J.R. (1995) RH-112,485: acute oral toxicity study in male and female mice., Unpublished report No. 95R-036, 91161 from Rohm and Haas Co. Submitted to WHO by Dow Agrosciences, Indianapolis, USA.
Robinson, P., Anderson, D.M. & Davidson, B.F. (1998) RH-2485 technical: eighteen-month dietary oncogenicity study in mice. Unpublished report No. 94R-255, 91188 from Rohm and Haas Co. Submitted to WHO by Dow Agrosciences, Indianapolis, USA.
Sames, J.S. & Black, K.A. (1995) RH-112,485: micronucleus assay in CD-1 mouse bone marrow cells. Unpublished report No. 94R-259, 91181 from Rohm and Haas Co. Submitted to WHO by Dow Agrosciences, Indianapolis, USA.
Sames, J.L. & Ciaccio, P.J. (1996) RH-112,485 2F: Salmonella typhimurium gene mutation assay (Ames test). Unpublished report No. 96R-163, 91184 from Rohm and Haas Co. Submitted to WHO by Dow Agrosciences, Indianapolis, USA.
Sames, J.L. & Ciaccio, P.J. (1997) RH-112,485 2F: micronucleus assay in CD-1 mouse bone marrow cells. Unpublished report No. 97R-058, 91180 from Rohm and Haas Co. Submitted to WHO by Dow Agrosciences, Indianapolis, USA.
Sames, J.L. & Ciaccio, P.J. (1998) RH-117,236 technical: Salmonella typhimurium gene mutation assay (Ames test). Unpublished report No. 97R-150,91183 from Rohm and Haas Co. Submitted to WHO by Dow Agrosciences, Indianapolis, USA.
Sames, J.L. & Streelman, D.R. (1995) RH-112,485: Salmonella typhimurium gene mutation assay (Ames test). Unpublished report No. 94R-258, 91176 from Rohm and Haas Co. Submitted to WHO by Dow Agrosciences, Indianapolis, USA.
Shuey, D.L. (1997) RH-2485 technical: oral (gavage) developmental toxicity study in rabbits. Unpublished report No. 96R-047, 91178 from Rohm and Haas Co. Submitted to WHO by Dow Agrosciences, Indianapolis, USA.
Vandenberghe, Y.L. & Gillette, D.M. (1995) RH-112,485 two-week oral definitive toxicity study in dogs. Unpublished report No. 96774. Submitted to WHO by Dow Agrosciences, Indianapolis, USA.
Watts, V.S. & Frederick, C.B. (1998) 14C-RH-112,485 80WP and 14C-RH-112,485 2F formulations: dermal absorption in male rats. Unpublished report No. 97R-060 from Rohm and Haas Co. Submitted to WHO by Dow Agrosciences, Indianapolis, USA.
Watts, V.S. & Longacre, S.L. (1998) 14C-RH-112,485: pharmacokinetic study in rats. Unpublished report No. 94R-057, 91160 from Rohm and Haas Co. Submitted to WHO by Dow Agrosciences, Indianapolis, USA.
See Also:
Toxicological Abbreviations