
This report contains the collective views of an international group of experts and does not necessarily represent the decisions or the stated policy of the United Nations Environment Programme, the International Labour Organization, or the World Health Organization.
Concise International Chemical Assessment Document 56
First draft prepared by Drs J. Kielhorn, G. Könnecker, C. Pohlenz-Michel, S. Schmidt, and I. Mangelsdorf, Fraunhofer Institute of Toxicology and Aerosol Research, Drug Research and Clinical Inhalation, Hanover, Germany
Published under the joint sponsorship of the United Nations Environment Programme, the International Labour Organization, and the World Health Organization, and produced within the framework of the Inter-Organization Programme for the Sound Management of Chemicals.
World Health Organization
Geneva, 2003
The International Programme on Chemical Safety (IPCS), established in 1980, is a joint venture of the United Nations Environment Programme (UNEP), the International Labour Organization (ILO), and the World Health Organization (WHO). The overall objectives of the IPCS are to establish the scientific basis for assessment of the risk to human health and the environment from exposure to chemicals, through international peer review processes, as a prerequisite for the promotion of chemical safety, and to provide technical assistance in strengthening national capacities for the sound management of chemicals.
The Inter-Organization Programme for the Sound Management of Chemicals (IOMC) was established in 1995 by UNEP, ILO, the Food and Agriculture Organization of the United Nations, WHO, the United Nations Industrial Development Organization, the United Nations Institute for Training and Research, and the Organisation for Economic Co-operation and Development (Participating Organizations), following recommendations made by the 1992 UN Conference on Environment and Development to strengthen cooperation and increase coordination in the field of chemical safety. The purpose of the IOMC is to promote coordination of the policies and activities pursued by the Participating Organizations, jointly or separately, to achieve the sound management of chemicals in relation to human health and the environment.
WHO Library Cataloguing-in-Publication Data
1,2,3-Trichloropropane.
(Concise international chemical assessment document ; 56)
1.Propane - toxicity 2.Propane - adverse effects 3.Risk assessment 4.Environmental
exposure I.International Programme on Chemical Safety II.Series
ISBN 92 4 153056 1 (LC/NLM Classification: QD 305.H6)
ISSN 1020-6167
©World Health Organization 2003
All rights reserved. Publications of the World Health Organization can be obtained from Marketing and Dissemination, World Health Organization, 20 Avenue Appia, 1211 Geneva 27, Switzerland (tel: +41 22 791 2476; fax: +41 22 791 4857; email: bookorders@who.int). Requests for permission to reproduce or translate WHO publications — whether for sale or for noncommercial distribution — should be addressed to Publications, at the above address (fax: +41 22 791 4806; email: permissions@who.int).
The designations employed and the presentation of the material in this publication do not imply the expression of any opinion whatsoever on the part of the World Health Organization concerning the legal status of any country, territory, city or area or of its authorities, or concerning the delimitation of its frontiers or boundaries. Dotted lines on maps represent approximate border lines for which there may not yet be full agreement.
The mention of specific companies or of certain manufacturers’ products does not imply that they are endorsed or recommended by the World Health Organization in preference to others of a similar nature that are not mentioned. Errors and omissions excepted, the names of proprietary products are distinguished by initial capital letters.
The World Health Organization does not warrant that the information contained in this publication is complete and correct and shall not be liable for any damages incurred as a result of its use.
The Federal Ministry for the Environment, Nature Conservation and Nuclear Safety, Germany, provided financial support for the printing of this publication.
|
5. ENVIRONMENTAL TRANSPORT, DISTRIBUTION, AND TRANSFORMATION |
|
7. COMPARATIVE KINETICS AND METABOLISM IN LABORATORY ANIMALS AND HUMANS |
|
11.2.3 Uncertainties in the evaluation of environmental effects |
Concise International Chemical Assessment Documents (CICADs) are the latest in a family of publications from the International Programme on Chemical Safety (IPCS) — a cooperative programme of the World Health Organization (WHO), the International Labour Organization (ILO), and the United Nations Environment Programme (UNEP). CICADs join the Environmental Health Criteria documents (EHCs) as authoritative documents on the risk assessment of chemicals.
International Chemical Safety Cards on the relevant chemical(s) are attached at the end of the CICAD, to provide the reader with concise information on the protection of human health and on emergency action. They are produced in a separate peer-reviewed procedure at IPCS. They may be complemented by information from IPCS Poison Information Monographs (PIM), similarly produced separately from the CICAD process.
CICADs are concise documents that provide summaries of the relevant scientific information concerning the potential effects of chemicals upon human health and/or the environment. They are based on selected national or regional evaluation documents or on existing EHCs. Before acceptance for publication as CICADs by IPCS, these documents undergo extensive peer review by internationally selected experts to ensure their completeness, accuracy in the way in which the original data are represented, and the validity of the conclusions drawn.
The primary objective of CICADs is characterization of hazard and dose–response from exposure to a chemical. CICADs are not a summary of all available data on a particular chemical; rather, they include only that information considered critical for characterization of the risk posed by the chemical. The critical studies are, however, presented in sufficient detail to support the conclusions drawn. For additional information, the reader should consult the identified source documents upon which the CICAD has been based.
Risks to human health and the environment will vary considerably depending upon the type and extent of exposure. Responsible authorities are strongly encouraged to characterize risk on the basis of locally measured or predicted exposure scenarios. To assist the reader, examples of exposure estimation and risk characterization are provided in CICADs, whenever possible. These examples cannot be considered as representing all possible exposure situations, but are provided as guidance only. The reader is referred to EHC 170.1
While every effort is made to ensure that CICADs represent the current status of knowledge, new information is being developed constantly. Unless otherwise stated, CICADs are based on a search of the scientific literature to the date shown in the executive summary. In the event that a reader becomes aware of new information that would change the conclusions drawn in a CICAD, the reader is requested to contact IPCS to inform it of the new information.
Procedures
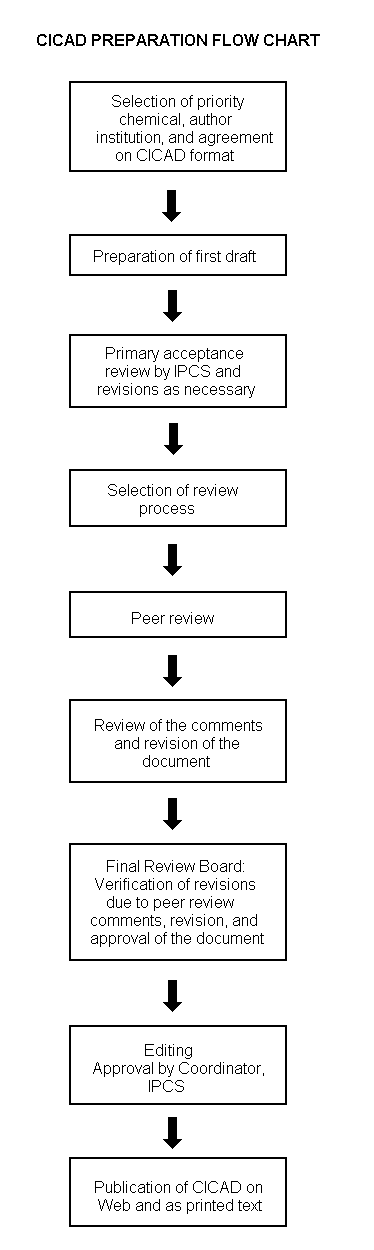
The flow chart on page 2 shows the procedures followed to produce a CICAD. These procedures are designed to take advantage of the expertise that exists around the world — expertise that is required to produce the high-quality evaluations of toxicological, exposure, and other data that are necessary for assessing risks to human health and/or the environment. The IPCS Risk Assessment Steering Group advises the Coordinator, IPCS, on the selection of chemicals for an IPCS risk assessment based on the following criteria:
Thus, it is typical of a priority chemical that
The Steering Group will also advise IPCS on the appropriate form of the document (i.e., EHC or CICAD) and which institution bears the responsibility of the document production, as well as on the type and extent of the international peer review.
The first draft is based on an existing national, regional, or international review. Authors of the first draft are usually, but not necessarily, from the institution that developed the original review. A standard outline has been developed to encourage consistency in form. The first draft undergoes primary review by IPCS to ensure that it meets the specified criteria for CICADs.

|
Advice from Risk Assessment Steering Group Criteria of priority:
Thus, it is typical of a priority chemical that
Special emphasis is placed on avoiding duplication of effort by WHO and other international organizations. A prerequisite of the production of a CICAD is the availability of a recent high-quality national/regional risk assessment document = source document. The source document and the CICAD may be produced in parallel. If the source document does not contain an environmental section, this may be produced de novo, provided it is not controversial. If no source document is available, IPCS may produce a de novo risk assessment document if the cost is justified. Depending on the complexity and extent of controversy of the issues involved, the steering group may advise on different levels of peer review:
|
The second stage involves international peer review by scientists known for their particular expertise and by scientists selected from an international roster compiled by IPCS through recommendations from IPCS national Contact Points and from IPCS Participating Institutions. Adequate time is allowed for the selected experts to undertake a thorough review. Authors are required to take reviewers’ comments into account and revise their draft, if necessary. The resulting second draft is submitted to a Final Review Board together with the reviewers’ comments. At any stage in the international review process, a consultative group may be necessary to address specific areas of the science.
The CICAD Final Review Board has several important functions:
Board members serve in their personal capacity, not as representatives of any organization, government, or industry. They are selected because of their expertise in human and environmental toxicology or because of their experience in the regulation of chemicals. Boards are chosen according to the range of expertise required for a meeting and the need for balanced geographic representation.
Board members, authors, reviewers, consultants, and advisers who participate in the preparation of a CICAD are required to declare any real or potential conflict of interest in relation to the subjects under discussion at any stage of the process. Representatives of nongovernmental organizations may be invited to observe the proceedings of the Final Review Board. Observers may participate in Board discussions only at the invitation of the Chairperson, and they may not participate in the final decision-making process.
This CICAD on 1,2,3-trichloropropane was prepared by the Fraunhofer Institute of Toxicology and Aerosol Research, Drug Research and Clinical Inhalation, Hanover, Germany. It is based on reports compiled by the German Advisory Committee on Existing Chemicals of Environmental Relevance (BUA, 1993) and the German Commission for the Investigation of Health Hazards of Chemical Compounds in the Work Area (MAK, 1993). A comprehensive literature search of relevant databases was conducted in November 2001 for health effects and in September 2002 for environmental effects to identify any relevant references published subsequent to those incorporated in these reports. Information on the preparation and peer review of the source documents is presented in Appendix 1. Information on the peer review of this CICAD is presented in Appendix 2. This CICAD was approved as an international assessment at a meeting of the Final Review Board, held in Monks Wood, United Kingdom, on 16–19 September 2002. Participants at the Final Review Board meeting are listed in Appendix 3. The International Chemical Safety Card for 1,2,3-trichloropropane (ICSC 0683), produced by the International Programme on Chemical Safety (IPCS, 1999), has also been reproduced in this document.
1,2,3-Trichloropropane (CAS No.
The predominant target compartment for 1,2,3-trichloropropane in the environment is air (about 85%), followed by water (about 11%). Reported concentrations of 1,2,3-trichloropropane in ambient air in the USA and Europe range from not detected to 0.4 µg/m3. In European rivers, concentrations range from not detected to 2.2 µg/litre.
1,2,3-Trichloropropane released into the environment is only poorly converted by abiotic processes (e.g., transformation by photochemically produced hydroxyl radicals) and might therefore remain present for extended periods. However, it can be removed from aquatic systems by evaporation and might be leached from soil into groundwater, due to the low soil sorption coefficients (Koc) reported for this compound. It is not readily biodegraded and is only slowly transformed by bacteria under aerobic and anaerobic conditions. The data available on the bioconcentration of this chlorinated alkane indicate that it is unlikely to bioaccumulate.
The main routes of exposure to 1,2,3-trichloropropane are via inhalation of contaminated air and ingestion of contaminated drinking-water. The chemical can be absorbed dermally to a more limited extent.
In animal studies, 1,2,3-trichloropropane is rapidly absorbed from the gastrointestinal tract, metabolized, and excreted. Orally applied doses were excreted within 60 h via urine (50–65%), faeces (15–20%), and breath as carbon dioxide (20%). 1,2,3-Trichloropropane appears to be metabolized faster in mice than in rats.
In rats, after oral administration, the major urinary metabolite (40% of urinary radioactivity) identified after 6 h is a mercapturic acid conjugate, N-acetyl-S-(3-chloro-2-hydroxypropyl)-L-cysteine. In the 24-h urine, a second metabolite, a cysteine conjugate (S-(3-chloro-2-hydroxypropyl)-L-cysteine), was determined. 14C activity in target organs (i.e., liver, kidney, and forestomach) was still found 60 h after oral administration of [14C]1,2,3-trichloropropane. After intravenous injection of 1,2,3-trichloropropane to rats, one major metabolite in the bile was 2-(S-glutathionyl)malonic acid. In mice, the urinary metabolite spectrum is more complex.
The isolation of the above metabolites suggests that biotransformation of 1,2,3-trichloropropane involves both conjugation with glutathione (GSH) and oxidation. One proposed pathway in the liver is the mixed-function oxidase-catalysed oxygenation of 1,2,3-trichloropropane on a terminal carbon to yield a chlorohydrin, followed by additional reactions that result in formation of the observed metabolites. A second pathway in the liver may involve the GSH transferase-catalysed formation of GSH conjugates, which either undergo additional biotransformation in the liver or are excreted in bile or plasma.
1,2,3-Trichloropropane is of moderate acute toxicity, with oral LD50 values in the rat ranging from 150 to 500 mg/kg body weight. Dermal toxicity is lower, with reported LD50 values of 836 mg/kg body weight in a single rat study and values ranging from 384 to 2457 mg/kg body weight in rabbits. A 4-h LC50 of about 3000 mg 1,2,3-trichloropropane/m3 was determined for rats and mice. The prominent toxic effect is irritation of the mucosa of eyes and nose and liver and kidney damage.
1,2,3-Trichloropropane is an irritant to skin and mucous membranes. In various tests with guinea-pigs, 1,2,3-trichloropropane showed no or only a very slight sensitizing effect.
The primary effect of repeated exposure of F344 rats and B6C3F1 mice to up to 780 mg 1,2,3-trichloropropane/m3 for 9 days by inhalation was a microscopic degenerative and inflammatory change in the nasal olfactory mucosa. In mice, testes weights were significantly reduced at the highest dose, but without associated histopathological changes. There were no other findings except for changes in liver weight in the highest dose group. In a repeated-dose experiment with doses up to 61 mg/m3, the overall no-observed-adverse-effect level (NOAEL) was 6 mg/m3 for rats and 18 mg/m3 for mice for changes in the olfactory epithelium detectable by histopathological examination.
In another study in which CD rats were exposed to up to 300 mg/m3 for 13 weeks, toxic effects in the upper respiratory tract, lung, and liver were observed. A follow-up study with doses up to 9.2 mg/m3 reported signs of irritation of mucous membranes (increase of lacrimal discharge), even at the lowest concentration of 3.1 mg/m3. The only systemic effects were changes in haematological parameters and increases in lung and ovary weights without corresponding microscopic findings.
After medium-term oral exposure of F344 rats to 1,2,3-trichloropropane, the principal toxic lesions occurred in liver, kidney, and nasal turbinates, with males showing a greater sensitivity. Haematological changes at 16 mg/kg body weight per day and higher dosages were interpreted as non-regenerative anaemia, possibly associated with a depression in erythropoiesis. The lowest-observed-adverse-effect level (LOAEL) for a 17-week treatment by gavage was 8 mg/kg body weight per day in male rats and 16 mg/kg body weight per day in female rats (increase in absolute liver weight). In B6C3F1 mice, the main targets of toxic action were lung, liver, and forestomach. Mice tolerated higher doses than rats, with LOAELs (hyperplasia of bronchiolar epithelium and hyperplasia and hyperkeratosis of forestomach) of 63 mg/kg body weight per day in female mice and 125 mg/kg body weight per day in male mice. A cardiotoxic effect discussed by one working group was not confirmed by these studies, which covered comparable or longer treatment periods. Gavage as a bolus application produced more severe effects than continuous application via drinking-water at a comparable dose level. When 1,2,3-trichloropropane was administered to Sprague-Dawley rats via drinking-water for 13 weeks, the LOAELs for increased relative liver and kidney weights were 17.6 mg/kg body weight per day for female rats and 113 mg/kg body weight per day for male rats.
From a variety of in vitro genotoxicity tests (e.g., gene mutations in bacterial and mammalian cells, gene conversions in Saccharomyces cerevisiae, induction of sister chromatid exchange, chromosomal aberrations, and micronuclei), a genotoxic potential of 1,2,3-trichloropropane in the presence of metabolic activation systems is evident. Single data on a direct genotoxic action of 1,2,3-trichloropropane seem questionable. In vivo, DNA single strand breaks were detectable by alkaline elution, whereas genotoxic action in a dominant lethal test was absent.
The major DNA adduct, S-[l-(hydroxymethyl)-2-(N7-guanyl)ethyl]glutathione, and other DNA adducts were identified in preneoplastic and neoplastic lesions of target organs.
The results of a two-generation study in Swiss mice showed that 1,2,3-trichloropropane impaired fertility and reproduction at a dose level of 120 mg/kg body weight per day (administered via gavage) in both the parent and the offspring generations in the presence of only slight systemic toxicity. A cross-mating trial suggested greater toxicity for the female than for the male reproductive system. The average estrous cycle length was significantly increased for all 1,2,3-trichloropropane-exposed females of the F1 generation, with the lowest dose being 30 mg/kg body weight per day.
1,2,3-Trichloropropane is a multisite carcinogen with corresponding preneoplastic lesions in both rats and mice after long-term application by gavage. Main targets of carcinogenic action are the forestomach and the oral mucosa in rats of both sexes, the mammary gland in female rats, pancreas and kidney in male rats, as well as preputial gland and clitoral gland, as homologous organs in male and female rats, respectively. Mice responded with neoplasms of the forestomach, the liver, and the Harderian gland. Uncommon tumour types were reported, such as carcinomas of the Zymbal’s gland and adenomatous polyps or adenocarcinomas of the intestine in rats and uterine neoplasms in mice. Considering the very high incidences of forestomach neoplasms in low-dose groups of rats (33–66%) and mice (nearly 100%), this carcinogenic activity might have been detected even at lower doses, and the LOAELs for significantly increased tumour incidences will be well below 3 mg/kg body weight per day in rats and 6 mg/kg body weight per day in mice.
1,2,3-Trichloropropane is carcinogenic in rats and mice, inducing a wide range of tumours in both sexes. Data on mode of action, including studies on metabolism, genotoxicity, and determination of DNA adduct formation, suggest that the mechanism of induction of tumours involves direct interaction of an active metabolite with genetic material. Hence, exposure to 1,2,3-trichloropropane should be avoided.
The acute toxicity of 1,2,3-trichloropropane was tested using a variety of aquatic species from different trophic levels.
A sample risk characterization according to the European Commission with respect to the aquatic environment was performed by calculating the ratio between a local predicted environmental concentration (PEC), based on measured data, and a corresponding predicted no-effect concentration (PNEC).
A PNEC for surface water was estimated from the lowest EC50 value from tests carried out in closed systems to minimize 1,2,3-trichloropropane losses. The 48-h EC50 value for Daphnia magna immobilization (20 mg/litre) was used to derive the PNEC, together with an uncertainty factor of 1000: PNEC = 20 mg/litre ÷ 1000 = 0.02 mg/litre. Although a lower value was obtained for Ceriodaphnia cf. dubia (48-h EC50 of 4.1 mg/litre), this test was based on nominal concentrations only and therefore was not used in the risk characterization.
Using the highest recently measured concentration of 1,2,3-trichloropropane in surface water (2.2 µg/litre) as the PEC, the hazard quotient (PEC/PNEC) becomes 2.2 µg/litre ÷ 20 µg/litre = 0.11. Because this is less than 1, no further information, testing, or risk reduction measures are required.
No data were identified on the toxic effects of this chemical on terrestrial invertebrates or higher plants. For the terrestrial compartment, the available toxicity studies measuring the inhibition of soil-bound microbial activity by 1,2,3-trichloropropane cannot be regarded as sufficient to support a quantitative risk characterization.
1,2,3-Trichloropropane (CAS No.
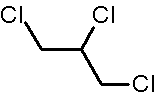
1,2,3-trichloropropane
The purity of the commercially available compound is >98–99.9%. Impurities greater than 0.1% have been identified as isomers of chlorohexane and chlorohexadiene, and there are several unidentifiable impurities (each <0.1%) (NTP, 1993).
Table 1: Physicochemical properties of 1,2,3-trichloropropane.
|
Property |
Value |
Reference |
|
Relative molecular mass |
147.43 |
|
|
Density (g/cm3) |
1.38 (20 °C) |
Rassaerts & Witzel (1975) |
|
Melting point (°C) |
–14.7 |
Lide (1995) |
|
Boiling point (°C) |
156 |
Miermans et al. (2000) |
|
Vapour pressure (kPa) |
0.492 (25 °C) |
Lide (1995) |
|
Air saturation (g/m3) |
16 (20 °C) |
Verschueren (1996) |
|
n -Octanol/water partition coefficient(log Kow) |
2.54 (calculated) |
Ruelle (2000) |
|
Water solubility (g/litre) |
1.75 (25 °C) |
Albanese et al. (1987) |
|
Henry’s law constant |
||
|
Pa·m3/mol |
31.82 (25 °C, calculated) |
Dilling (1977) |
|
Pa·m3/mol |
22.83 (25 °C, measured) |
Tancrède & Yanagisawa (1990) |
Conversion factors2 for 1,2,3-trichloropropane in air at 20 °C and 101.3 kPa are as follows:
1 mg/m3 = 0.16 ppm
1 ppm = 6.1 mg/m3
Selected methods for the detection and quantification of 1,2,3-trichloropropane in different matrices are given here. Additional and more detailed information is available in BUA (1993), IARC (1995), and references cited therein.
Analysis of 1,2,3-trichloropropane in air usually involves sorption of the chlorinated alkane onto a solid matrix, followed by thermal or solvent desorption prior to gas chromatography/flame ionization detection (GC/FID), gas chromatography/photoionization detection/electrolytic conductivity detection (GC/PID/ELCD), or gas chromatography/mass spectrometry (GC/MS). Yamamoto et al. (1998), using GC with ELCD, reported a detection limit of about 2 mg/m3. Peng & Batterman (2000) as well as Bonvalot et al. (2000), employing GC/MS, reported a method detection limit of 0.04 µg/m3. Pankow et al. (1998), also using GC/MS, reported a detection limit of about 30 µg/m3. Both Brock & Carroll (1985) and Bouhamra et al. (1997) analysed air samples by similar techniques, but without specifying detection limits.
For water, purge and trap techniques followed by gas chromatography/electron capture detection (GC/ECD) or GC/MS are generally employed. Bauer (1981a) reported a detection limit of 0.07 µg/litre, Zebarth et al. (1998) determined a limit of quantification of 0.1 µg/litre, and Miermans et al. (2000) obtained a limit of detection of 0.0004 µg/litre. Yoshikawa et al. (1998) employed a similar system, although detection limits were not specified.
Sediment was analysed by GC/ECD after nitrogen blow-out, adsorption, and solvent elution, with a detection limit of 1 µg/kg (LWA, 1989). For the detection of 1,2,3-trichloropropane in sediments, Kawata et al. (1997) used GC/MS for head space analysis after thermal equilibration, obtaining a detection limit of 1 ng/g, while Zebarth et al. (1998) employed head space GC/ELCD, with a limit of detection of 0.2 µg/kg.
Bauer (1981a) determined the presence of 1,2,3-trichloropropane in human tissue samples after blow-out and sorption techniques by GC/ECD, with a detection limit of 13 µg/kg.
According to BUA (1993), there are no known natural sources of 1,2,3-trichloropropane.
Not only is 1,2,3-trichloropropane manufactured itself, but it is also produced in significant quantities as a by-product of the production of other chlorinated compounds, including epichlorohydrin (NTP, 2000).
Moorman et al. (2000) cited an annual production of 9000–14 000 tonnes of 1,2,3-trichloropropane for the USA. Less than about 50 000 tonnes of 1,2,3-trichloropropane is produced annually, globally, as a by-product of epichlorohydrin production. There are about 20–30 epichlorohydrin-producing facilities in North America, Europe, and Asia (The Society of the Plastics Industry [SPI] Epichlorohydrin Task Group, personal communication, 2002).
1,2,3-Trichloropropane is used as an intermediate in the synthesis of other chemicals (e.g., pesticides) in closed systems and as a crosslinking agent in the production of polymers, such as polysulfides and hexafluoropropylene (SPI Epichlorohydrin Task Group, personal communication, 2002). Older reports identified 1,2,3-trichloropropane as a solvent for hydrophobic compounds and resins, as a paint and varnish remover, and as a degreasing agent (Johnson, 1968; Ellerstein & Bertozzi, 1982; Lewis, 1992; BUA, 1993; IARC, 1995; and references cited therein), but it is probably not marketed in consumer products today. The majority (>80%) of the 1,2,3-trichloropropane produced as a by-product of epichlorohydrin production is incinerated on-site (SPI Epichlorohydrin Task Group, personal communication, 2002).
According to data from the US EPA (1999), in 1999, about 11.9 tonnes of 1,2,3-trichloropropane were released into the environment both on-site and off-site, about 5.74 tonnes entered the atmosphere, 0.92 tonnes were discharged into surface water, and about 3.37 tonnes were released into the geosphere.
In general, 1,2,3-trichloropropane may be released during its manufacture, during the manufacture of other industrial chemicals (e.g., epichlorohydrin), which forms 1,2,3-trichloropropane as an unwanted by-product, or by its application or the application of products containing 1,2,3-trichloropropane as an impurity. For example, the presence of 1,2,3-trichloropropane in pesticides and nematicides employed in soil fumigation (Telone, for example, reportedly contains up to 0.17% 1,2,3-trichloropropane by weight; Zebarth et al., 1998) has been identified as a potential source for the release of this chlorinated compound into the environment (Zebarth et al., 1998; City of Shafter, 2000). In addition, 1,2,3-trichloropropane can be present in formulations employed as a well drilling aid and, as such, can lead to contamination of drinking-water (Health Canada, 2000).
The predominant target compartment for 1,2,3-trichloropropane in the environment is air (about 85%), followed by water (about 11%; Level I calculation, six-compartment model; Mackay et al., 1993). Due to its experimentally determined water solubility of 1.75 g/litre, a significant proportion of this compound should be removed from the atmosphere by washout. According to Thomas (1990), the measured Henry’s law constant of 22.83 Pa·m3/mol (Tancrède & Yanagisawa, 1990) indicates moderate volatility from the aqueous phase. In fact, Dilling (1977) determined a half-life of 56 min for the stripping of 1,2,3-trichloropropane from water, while Albanese et al. (1987) found half-lives of 92 min (fresh water) and 93 min (seawater). Tancrède et al. (1992), when employing 1,2,3-trichloropropane-spiked tap water, found an evaporation of >83% at flow rates from 9.7 to 13.6 ml/min and at three different temperatures (25, 33, and 42 °C).
Using sandy and silty loam, Anderson et al. (1991) showed the abiotic loss of 1,2,3-trichloropropane from soil, with half-lives of 2.2–3.5 days. Although this depletion was probably due to evaporation, 1,2,3-trichloropropane will, under environmental conditions, be prone to washout from soil, as its Koc values in the range of 77–95 (measured using a silty and a sandy loam; Walton et al., 1992) indicate a high mobility in soil (Swann et al., 1983; Blume, 1990). Experimental results showing the appearance of 1,2,3-trichloropropane in groundwater (Baier et al., 1987; Oki & Giambelluca, 1989) after the application of nematicides containing 1,2,3-trichloropropane prove the potential of this non-polar chlorinated alkane to contaminate groundwater.
The measured bioconcentration factors (determined according to OECD guideline 305C) in the range of 3–13 (MITI, 1992) indicate no significant bioaccumulation potential for 1,2,3-trichloropropane.
Using the calculated hydroxyl radical reaction rate constant (KOH) of Atkinson (1987) and assuming a hydroxyl radical concentration of 5 × 105 molecules/cm3, a half-life of 27.2 days was calculated for 1,2,3-trichloropropane in the atmosphere (BUA, 1993). When applying the US Environmental Protection Agency (EPA) modelling program AOPWIN (Version 1.9), a half-life of about 30.5 days can be calculated, using a rate constant for the hydrogen abstraction (KOH) of 0.3511 × 10–12 cm3/s per molecule and a hydroxyl radical concentration of 1.5 × 106 molecules/cm3. Hence, 1,2,3-trichloropropane released into the atmosphere might undergo a very slow degradation in this environmental compartment in the presence of a sufficient concentration of photochemically produced hydroxyl radicals. Hydrolysis of 1,2,3-trichloropropane seems to be of minor importance, with calculated half-lives of 44 and 74 years (Ellington et al., 1987; Milano et al., 1988).
In aerobic biodegradation tests performed according to OECD guideline 301C, 1,2,3-trichloropropane was not readily biodegradable (biological oxygen demand 0% of theoretical oxygen demand, incubation for 28 days; MITI, 1992). In a preliminary study (Vannelli et al., 1990), the co-oxidative transformation of 1,2,3-trichloropropane by the ammonia-oxidizing bacterium Nitrosomonas europaea was shown. In the experiments described, the chlorinated substrate (at a concentration of about 6.8 µmol/litre) was reduced to a residual amount of 91% (in the absence of the energy source ammonia) and to 77% (in the presence of ammonia) after 24 h of incubation. More recent studies employing the methanotroph Methylosinus trichosporium demonstrated that the recalcitrant 1,2,3-trichloropropane is co-metabolically converted to a range of different products, such as chlorinated propanols (Bosma & Janssen, 1998). To date, all attempts to isolate cultures able to utilize 1,2,3-trichloropropane as a sole source of carbon and energy have failed. Even though a modified strain of Agrobacterium radiobacter (expressing an efficient haloalkane dehalogenase from a Rhodococcus sp.) did utilize the analogous 1,2,3-tribromopropane for growth, 1,2,3-trichloropropane was not used and only slowly converted (Bosma et al., 1999). Peijnenburg et al. (1998) observed the reductive transformation of 1,2,3-trichloropropane by using anaerobic sediments and determined that reductive dehalogenation was the sole reaction to take place. They calculated a zero-order reaction constant of 0.71 mmol/litre per day. Hauck & Hegemann (2000) described an anaerobic bioreactor system inoculated from river sediment, which converted 1,2,3-trichloropropane. However, neither analytical nor kinetic data were presented for 1,2,3-trichloropropane. Nevertheless, although results reported by Anderson et al. (1991) indicated a lack of biodegradation of 1,2,3-trichloropropane in clay loam, reductive dehalogenation of this compound might take place in anaerobic sediments.
In Bochum, Germany, concentrations of 1,2,3-trichloropropane not exceeding 0.4 µg/m3 were detected (Bauer, 1981b). Bonvalot et al. (2000) reported the presence of 1,2,3-trichloropropane in ambient air sampled at the Rivière des Prairies, Montreal, Canada, station at levels of up to 0.21 µg/m3. Pankow et al. (1998) were not able to detect 1,2,3-trichloropropane in New Jersey, USA, air using sampling sites ranging from a low to high degree of urbanization and traffic. Similarly, while Yamamoto et al. (1998) did not detect 1,2,3-trichloropropane in the urban air of Yokohama, Japan, Peng & Batterman (2000) failed to detect 1,2,3-trichloropropane in ambient air sampled along roads at "rush hour" periods in Detroit, USA. However, Bouhamra et al. (1997) reported a mean value of 491 µg 1,2,3-trichloropropane/m3 in ambient air sampled at 19 different locations in Kuwait.
Peng & Batterman (2000) failed to detect 1,2,3-trichloropropane in air from an office building sampled in an indoor air quality study in Ann Arbor, USA. Bouhamra et al. (1997) found a maximum concentration of 34.3 mg/m3 in indoor air sampled in residences in Kuwait. However, they were not able to correlate this to a specific source. Using their data, they calculated an average indoor/outdoor concentration ratio for 1,2,3-trichloropropane of 5.06 for Kuwait. There is no obvious reason for these exceptionally high values for ambient and indoor air in Kuwait, and the reports have yet to be substantiated.
The presence of 1,2,3-trichloropropane in European rivers has been frequently reported. In a river monitoring program, 1,2,3-trichloropropane was detected in Dutch surface water of the rivers Rhein, Meuse, and Westerscheldt and the Northern Delta Area, with the highest concentration up to 2.2 µg/litre (Miermans et al., 2000). Liska et al. (1996) identified 1,2,3-trichloropropane at five sampling points along the river Nitra, Slovakia, in a European river monitoring program, although no concentrations were given. In a follow-up study by Frischenschlager et al. (1997), 1,2,3-trichloropropane was detected at a concentration of 1.6 µg/litre at one sampling site. An extensive compilation of monitoring data summarizing the appearance of 1,2,3-trichloropropane in selected rivers, such as Rhein, Emscher, Elbe, and Weser, with sampling sites in Germany and the Netherlands, showed maximum concentrations not exceeding 0.6 µg/litre in the years 1981–1989 (BUA, 1993, and references cited therein). Yamamoto et al. (1997) detected 1,2,3-trichloropropane in 18 of 28 samples of water from rivers and estuaries in Osaka City, Japan; concentrations ranged from 0.18 (detection limit) to about 100 µg/litre. The authors found that 1,2,3-trichloropropane was not completely eliminated by the sewage treatment process, with a maximum concentration of approximately 90 µg/litre found in effluent discharges. Yoshikawa et al. (1998) detected 1,2,3-trichloropropane in seawater sampled near Kawasaki City, Japan, but concentrations were not given.
Groundwater samples from the Netherlands and the USA were shown to contain 1,2,3-trichloropropane due to the application of impure nematicides. Accordingly, Lagas et al. (1989) reported 1,2,3-trichloropropane concentrations of up to 5.6 µg/litre in groundwater samples from two potato plantations in the Netherlands. In the USA, both Oki & Giambelluca (1989) and Baier et al. (1987) reported 1,2,3-trichloropropane in groundwater, with maximum concentrations of about 2 µg/litre (Hawaii) and >100 µg/litre (New York State). More recently, Zebarth et al. (1998) reported a maximum value of 0.86 µg/litre in water sampled from a contaminated aquifer in British Columbia, Canada, with levels up to 0.92 µg/kg in the corresponding sediment.
Drinking-water sampled from 100 German cities did not exceed a maximum concentration of 0.1 µg 1,2,3-trichloropropane/litre (Bauer, 1981b). Gelover et al. (2000) detected 1,2,3-trichloropropane in drinking-water supplies in Mexico but failed to quantify the amounts present. However, more recently, 1,2,3-trichloropropane was detected in drinking-water in the USA (Puhi, Kaua’i, Hawaii; and City of Shafter, California) at concentrations of 0.1 µg/litre (Kaua’i Department of Water, 2001) and up to 0.24 µg/litre (City of Shafter, 2000).
The most likely routes of exposure of the general population to 1,2,3-trichloropropane are via air and water. This is due to the fact that 1,2,3-trichloropropane will preferably partition into air and water (Mackay et al., 1993). Human exposure will therefore occur by inhalation of contaminated air or by ingestion of 1,2,3-trichloropropane-containing water. In view of this, it is important to consider that indoor air is more likely to contain higher concentrations of 1,2,3-trichloropropane than outdoor air, due to the greater number of potential sources and lower rates of ventilation (Bouhamra et al., 1997). 1,2,3-Trichloropropane might be taken up as well via food, as von Düszeln et al. (1982) estimated (using the 1976 German Nutrition Report consumption patterns) an average daily intake of 7.4 µg per person, but gave no further details.
Assuming a daily intake of 7.4 µg per person via food, a daily consumption of 2 litres of drinking-water containing 0.1 µg 1,2,3-trichloropropane/litre, a daily intake of 20 m3 air with an average 1,2,3-trichloropropane concentration of 0.1 µg/m3, and a body weight of 64 kg, an estimated daily intake of 150 ng/kg body weight can be calculated. Omitting the amount of 1,2,3-trichloropropane taken up via food lowers the daily intake to about 34 ng/kg body weight. Using the above assumptions but the reported much higher values for air in Kuwait (outdoor air 491 µg/m3, indoor air 2480 µg/m3 [i.e., 5.06 × 491]) and assuming that 50% of the time is spent outdoors and 50% indoors, one can calculate a daily intake of about 464 µg/kg body weight. Omitting the amount of 1,2,3-trichloropropane taken up via food has no effect on this daily intake.
Occupational exposure is by inhalation and dermal exposure through the industrial production and use of 1,2,3-trichloropropane. However, there is a lack of information on dermal exposure and intake.
The only data available on inhalation exposure are those relating to 1,2,3-trichloropropane-containing air (Brock & Carroll, 1985). Brock & Carroll (1985) reported a maximum short-term exposure concentration of 17 mg 1,2,3-trichloropropane/m3 for maintenance personnel at a chemical plant in the USA. However, exposure concentrations in other workplaces did not usually exceed 0.61 mg/m3. These workplace concentrations (workday situation: daily consumption of 20 m3 air, accrued 1-h peak concentration with 17 mg/m3, 7-h on-site ambient air with <0.61 mg/m3, remaining time ambient air with 0.1 µg/m3; intake via food and drinking-water not accounted for) might lead to a daily intake of up to 277 µg/kg body weight.
Only one report is available on 1,2,3-trichloropropane detected in human tissues. Random human pathology samples (i.e., from lung, liver, muscle, kidney from 3 females and 12 males, age 31–78) were analysed; only in adipose tissue of the fibrous capsule of the kidney was 1,2,3-trichloropropane found, at an average concentration of 3.1 µg/kg fresh weight (with a stated maximum value of 15.1 µg/kg) (Bauer, 1981b).
Following a single administration of [14C]1,2,3-trichloropropane in corn oil by gavage to male and female F344 rats (30 mg/kg body weight) and male B6C3F1 mice (30 and 60 mg/kg body weight), 1,2,3-trichloropropane was rapidly absorbed, metabolized, and excreted. The major route of excretion was in the urine. By 60 h post-dosing, rats had excreted 50% and mice 65% by this route. Excretion in the faeces and exhalation as 14CO2 each accounted for 20% of the total dose in 60 h in rats and for 15 and 20%, respectively, in mice. At 60 h, in both rats and male mice, 14C activity was mainly concentrated in liver, kidney, and forestomach, the majority of the tissue 14C activity being not extractable and apparently covalently bound to protein. In rats, no apparent sex-related differences were observed in the ability to excrete 1,2,3-trichloropropane-derived radioactivity. Male mice eliminated 1,2,3-trichloropropane-derived radioactivity more rapidly than rats, and lower concentrations of radioactivity were found in tissues 60 h after dosing in mice (Mahmood et al., 1991).
Pharmacokinetic studies in male F344/N rats after intravenous administration of 3.6 mg 1,2,3-trichloropropane/kg body weight showed that 1,2,3-trichloropropane is rapidly distributed and eliminated (Volp et al., 1984), with higher elimination from blood than from tissue. This conclusion is based on the observation that the radiolabel had longer half-lives than 1,2,3-trichloropropane in muscle, skin, adipose tissue, liver, and kidney. The biphasic elimination kinetics are characterized by values of t½(1) of 0.3–1.8 h and t½(2) of 30–45 h for 1,2,3-trichloropropane and values of t½(1) of 2.1–5.3 h and t½(2) of 87–182 h for the radiolabel (Volp et al., 1984). In bile, 30% of the dose appeared within 6 h, presumably as GSH conjugates. As faecal excretion accounted for only 18% of the dose, a significant amount of the dose seemed to be reabsorbed from the intestines. The highest tissue 14C concentrations were found in adipose tissue, liver, and kidney after 1, 4, and 24 h, respectively.
Results of acute toxicity studies (see section 8.1) show that 1,2,3-trichloropropane can be dermally absorbed, although to a more limited extent than via the gastrointestinal tract.
Evidence suggests that 1,2,3-trichloropropane can be metabolized by two major pathways in rats and mice (see Figure 1). One proposed pathway in the liver involves mixed-function oxidase-catalysed oxygenation of 1,2,3-trichloropropane on a terminal carbon to yield a chlorohydrin, followed by additional reactions that result in the formation of the observed metabolites.
A second pathway in the liver involves GSH transferase-catalysed formation of GSH conjugates, which either undergo additional biotransformation in the liver or are excreted in bile or plasma. However, neither these metabolic pathways nor the metabolites are well characterized.
In rats, the major urinary metabolite (40% of urinary radioactivity) identified after 6 h is a mercapturic acid conjugate, N-acetyl-S-(3-chloro-2-hydroxypropyl)-L-cysteine, or ACPC. In the 24-h urine, a second metabolite, a cysteine conjugate (S-(3-chloro-2-hydroxypropyl)-L-cysteine), or CPC, was determined. After intravenous injection of 1,2,3-trichloropropane to rats, one of the three major metabolites in the bile was (2-(S-glutathionyl)malonic acid), or GMA (Mahmood et al., 1991). The metabolic profile in female rats was similar to that in males, but the metabolites were present at lower levels. In male mice, the urinary metabolite spectrum is more complex, and ACPC was only a minor component (only 3% of urinary radioactivity). Several unidentified metabolites are excreted in higher amounts. Female mice were not studied.
In vitro studies showed the NADPH-dependent formation of 1,3-dichloroacetone (DCA, a direct-acting mutagen; Merrick et al., 1987) from 1,2,3-trichloropropane in the presence of human and rat microsomes. DCA was formed at a rate of 0.27 nmol/min per milligram protein with microsomes from rat liver and at a rate of 0.03 nmol/min per milligram protein with microsomes from a human liver sample. An increase in the rate of DCA formation was observable after phenobarbital- and dexamethasone-mediated induction of cytochrome P-450. In the presence of alcohol dehydrogenase and NADH, 1,3-dichloro-2-propanol and 2,3-dichloropropanol were detected as secondary metabolites of DCA and 2,3-dichloropropanal (Weber & Sipes, 1992).
Covalent binding to hepatic macromolecules was investigated after intraperitoneal administration of 30 mg 1,2,3-trichloropropane/kg body weight to rats. Four hours post-administration, the extent of covalent binding to protein, RNA, and DNA was 418, 432, and 244 pmol 1,2,3-trichloropropane equivalents/mg, respectively. Whereas binding to protein reached a maximum at 4 h and then decreased significantly to the next measured time point (at 24 h post-administration), the maximum DNA binding was reached at 24 h post-administration. The binding to both hepatic protein and DNA was cumulative for up to three dosages given 24 h apart. GSH may play a dual role in covalent binding: supporting the binding of 1,2,3-trichloropropane to hepatic DNA and inhibiting the binding to protein. It was assumed that several metabolic pathways take part in the activation and covalent binding of 1,2,3-trichloropropane (Weber & Sipes, 1990).
Significant DNA adduct levels were measured in several target and non-target tissues of tumorigenesis after single gavage dosages of 3 and 30 mg/kg body weight in male Fischer 344 rats and 6 and 60 mg/kg body weight in male B6C3F1 mice (see section 8.4.2).
In the DNA of rat liver after intraperitoneal administration of 300 mg 1,2,3-trichloropropane/kg body weight, one major adduct, S-[l-(hydroxymethyl)-2-(N7-guanyl)ethyl]glutathione, was identified (La et al., 1995), confirming the role of GSH in adduct formation. Pretreatment of rats with phenobarbital (induction of cytochrome P-450) as well as GSH depletion caused a decrease in covalent DNA binding (Weber & Sipes, 1990, 1992). S-[l-(hydroxymethyl)-2-(N7-guanyl)ethyl]glutathione was formed in forestomach, liver, glandular stomach, and kidney after repeated dosing either by gavage or by drinking-water (see section 8.4.2) (La et al., 1996).
1,2,3-Trichloropropane is of moderate acute toxicity.
A 4-h LC50 of about 3000 mg 1,2,3-trichloropropane/m3 was determined for rats and mice from a compilation of mortality rates after single inhalation exposure to high concentrations for 0.5–4 h. Symptoms of intoxication included prostration, hypoactivity, ataxia, sedation, dyspnoea, convulsions, lacrimation, salivation, irritation of the mucosa of eyes and nose, and liver and kidney damage. Immediate respiratory depression was a frequent cause of death. Delayed deaths (after 7–10 days) were ascribed to liver damage (MAK, 1993). Significant increases in the serum enzymes glutamic–oxaloacetic transaminase, glutamic–pyruvic transaminase, and ornithine carbamyl transferase up to 48 h after a single 4-h inhalation exposure to 3100 mg 1,2,3-tri-chloropropane were indicative of hepatic damage in rats (Drew et al., 1978).
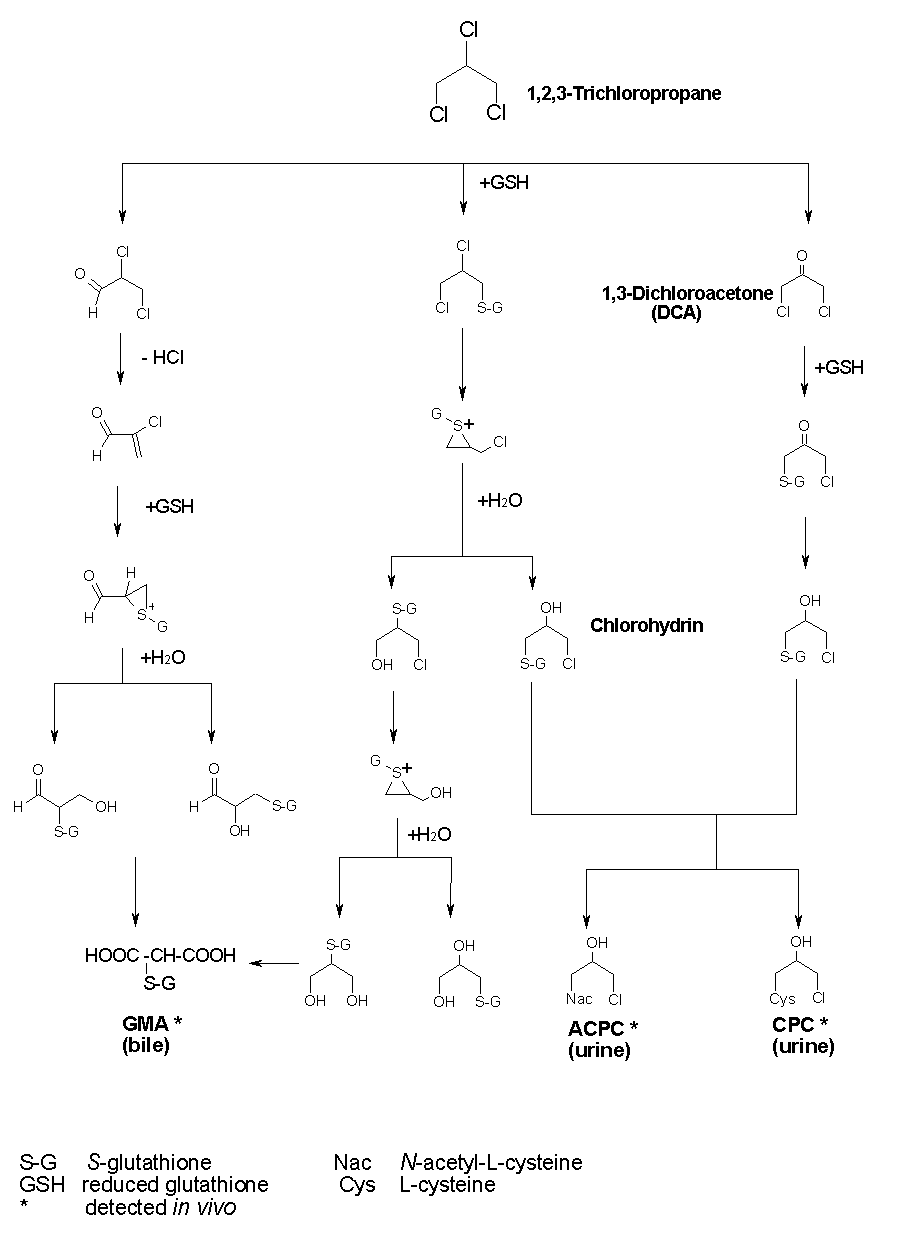
Fig. 1: Possible metabolic pathways in rats (Mahmood et al., 1991)
[ACPC = N-acetyl-S-(3-chloro-2-hydroxypropyl)-L-cysteine; CPC = S-(3-chloro-2-hydroxypropyl)-L-cysteine;
GMA = 2-(S-glutathionyl)malonic acid]
Oral LD50 values in the rat range from 150 to 500 mg/kg body weight (MAK, 1993).
Dermal toxicity is lower, with reported LD50 values of 836 mg/kg body weight in rats and 384–2457 mg/kg body weight in rabbits (MAK, 1993).
In comparison with other tested chloro-, bromo-, and mixed chlorobromopropanes, 1,2,3-trichloropropane showed a very low potency as a nephrotoxicant after a single intraperitoneal application of 1, 2, or 3 mmol/kg body weight (147, 294, or 441 mg/kg body weight). 1,2,3-Tribromopropane was the most potent. In the mid- and high-dose groups exposed to 1,2,3-trichloropropane, 1/5 and 3/5 male Wistar rats died, respectively. Forty-eight hours after application, relative kidney weight and urea excretion were dose-dependently increased (no comment on significance), and histopathological examination detected moderate kidney necrosis in one of the two surviving animals of the high-dose group. Significant DNA damage (DNA single strand breaks determined by alkaline elution) was reported starting from dosages of 0.375 mmol/kg body weight (Låg et al., 1991).
A dose of 0.5 ml of 1,2,3-trichloropropane applied to the intact skin of rabbits (5 males, 1 female) for 4 h under semi-occlusive and for 24 h under occlusive dressings produced mild reversible irritation, with a primary irritation index of 2.5 for the 24-h application (maximum possible value 8.0) (Bio/dynamics Inc., 1985a). Comparable results were reported for intact (12 rabbits, sex not given) and abraded skin of rabbits (3 males, 3 females), with a primary irritation index of 1.63 (maximum possible value 8.0) (Albert, 1982). In contrast to these test results, another study classified 1,2,3-trichloropropane as severely irritating, based on irritation scores from a Draize test (0.5 ml 1,2,3-trichloropropane, 24-h occlusive application) on intact and abraded skin of rabbits (3 males, 11 females). The mean scores were from 1.6 to 3.0 on a standard scale ranging from 0 to 4 (Clark, 1977). Therefore, direct contact of 1,2,3-trichloropropane with the skin causes slight irritation or strong irritation if confined to the skin.
After instillation of 0.1 ml of undiluted 1,2,3-trichloropropane, mild to moderate irritation of rabbit eyes was observed (conjunctival irritation, conjunctival necrosis, clouding of the cornea, iris damage), with all effects being reversible within 2–7 days (Bio/dynamics Inc., 1985b). Another eye irritation test (Draize test) reported less severe effects, leading to a classification of 1,2,3-trichloropropane as mildly irritating to rabbit eyes after 1–2 h (Clark, 1977). With a maximal irritation score of 20.0 out of a possible 110.0 at 6 h after instillation, undiluted 1,2,3-trichloropropane was classified as moderately irritating to the eye (Albert, 1982).
In the guinea-pig maximization test (Magnusson and Kligman test), only a very mild sensitizing activity of 1,2,3-trichloropropane was found (2/20 guinea-pigs with a positive reaction, 1/20 with a weak reaction; effects entirely reversible within 48 h). The applied concentrations of 1,2,3-trichloropropane were a 0.1% solution in corn oil for intradermal induction, 50% in corn oil for topical induction, and 25% for topical challenge. Results were scored on removal of the challenge patch and 24 and 48 h later (Clark, 1977; MAK, 1993).
A lack of sensitizing effects of 1,2,3-trichloropropane was reported in groups of five male and five female Dunkin-Hartley guinea-pigs. Induction was by weekly topical treatment with 0.5 ml undiluted 1,2,3-trichloropropane under occlusive bandages for 6 h over a 3-week period, followed by one challenge application after a 2-week rest period. Further groups were treated with corn oil as negative control and 2,4-dinitrochlorobenzene as positive control. Results were scored on removal of the challenge patch and 24 and 48 h later. One male and one female exposed to 1,2,3-trichloropropane were found dead on day 3 and day 11, respectively (Albert, 1982). The absence of any dermal response to undiluted 1,2,3-trichloropropane was also observed in Hartley albino guinea-pigs in a sensitization study conducted according to the Buehler method (induction: nine 6-h occlusive applications in 3 weeks followed by challenge after 2 weeks, readings after 24 and 48 h) (Bio/dynamics Inc., 1985c).
F344 rats and B6C3F1 mice were exposed to concentrations of 0, 80, 240, or 780 mg 1,2,3-trichloropropane/m3, 6 h/day, for a total of 9 days during an 11-day interval (Miller et al., 1986a). A significant decrease in body weight was noted only in rats, at all doses, while the body weights of mice were unaffected, even though both species had decreased abdominal fat at all doses. Reduced food consumption was observed at the highest concentration only. There was a significant increase in absolute and relative liver weights in the 780 mg/m3 groups of rats and mice. There were no microscopic changes in the liver indicative of severe hepatoxicity, nor were there any changes in serum enzymes that were diagnostic of liver injury. There were no gross or histopathological changes in kidneys of either rats or mice. In mice, testes weights were significantly reduced at 780 mg/m3, but without associated histopathological changes (Miller et al., 1986a).
The primary effect of exposure to 1,2,3-trichloro-propane in rats and mice in the same study was a microscopic degenerative and inflammatory change in the nasal olfactory mucosa (Miller et al., 1986a). Besides nasal exudate at the lowest dose in both species, degeneration of the epithelium was observed in rats at 80 mg/m3 and in mice at 240 mg/m3. Further nasal exostosis and fibrotic changes were detectable in rats at 780 mg/m3, whereas mice showed nasal exostosis from 240 mg/m3. Concentration-dependent adverse effects on nose tissue were more severe in rats than in mice.
Follow-up studies in F344 rats and B6C3F1 mice using lower concentrations of 1,2,3-trichloropropane (6, 18, and 61 mg/m3) for the same period were performed to derive NOAELs for the most sensitive end-point, changes in the olfactory epithelium detectable by histopathological examination of nose tissue (Miller et al., 1986b). The LOAEL for decreased thickness and inflammatory changes of the olfactory epithelium was 18 mg/m3 for rats and 61 mg/m3 for mice; the NOAEL was 6 mg/m3 for rats and 18 mg/m3 for mice. Neither signs of intoxication nor systemic effects were reported at any of these concentrations (Miller et al., 1986b).
Groups of CD rats (15 males and 15 females per group) were exposed by inhalation to 1,2,3-trichloropropane (purity 98.9%) for 13 weeks (6 h/day, 5 days/week) at concentrations of 0, 28, 92, and 300 mg/m3. Body weight decreases were noted in mid- and high-dose females only. Increases in liver weights were observed in males in all dose groups and in females in the mid- and high-dose groups. Signs of distinct respiratory tract irritation (i.e., red nasal discharge and excessive lacrimation) were reported at 92 mg/m3 and higher. Hepatocellular hypertrophy was observed in male rats at all doses. Dose-related focal peribronchial lymphoid hyperplasia was found primarily in males, whereas splenic extramedullary haematopoiesis was observed only in females at all 1,2,3-trichloropropane concentrations (Bio/dynamics Inc., 1979; Johannsen et al., 1988).
A follow-up 13-week study with lower 1,2,3-trichloropropane concentrations (0, 3.1, and 9.2 mg/m3) reported signs of irritation of mucous membranes (increase of lacrimal discharge), even at the lowest concentration of 3.1 mg/m3. However, histopathological examination of the nasal epithelium showed no treatment-related effects at any concentration. The only systemic effects were changes of haematological parameters and increases in lung and ovary weights without corresponding microscopic findings (Bio/dynamics Inc., 1983; Johannsen et al., 1988).
1,2,3-Trichloropropane (99% in 0.5% Emulphor; 1, 10, 100, and 1000 mg/litre) was administered for 13 weeks via drinking-water to male and female Sprague-Dawley rats (10 males and 10 females per group). Significantly decreased water intake was observed by females exposed to 100 and 1000 mg/litre and by males exposed to 1000 mg/litre and subsequently to reduced intake of 1,2,3-trichloropropane. Dosages were specified only for these three dose groups: i.e., 17.6 and 149 mg/kg body weight per day for females (100 and 1000 mg/litre) and 113 mg/kg body weight per day for males (1000 mg/litre). For the highest dosage, body weight gain was significantly reduced in both males and females. The only effects reported for the 100 mg/litre exposure were increased relative liver and kidney weights of females. Weight increases in these organs were found in both sexes for the highest exposure level and were accompanied by minor histological changes in these tissues. Changes of activities of hepatic enzymes (an elevation in serum cholesterol in female rats and induced hepatic aminopyrine demethylase and aniline hydroxylase in male rats) were observed at the highest dose (Villeneuve et al., 1985). The LOAELs for increased relative liver and kidney weights were 17.6 mg/kg body weight per day for female rats and 113 mg/kg body weight per day for male rats when 1,2,3-trichloropropane was administered via drinking-water. The NOAEL is about 2 mg/kg body weight per day (although the authors give the NOAEL as 15–20 mg/kg body weight, in spite of the increased liver weights in females).
F344 rats and B6C3F1 mice were administered 1,2,3-trichloropropane by gavage (>99% purity; in corn oil) at 0, 8, 16, 32, 63, 125, and 250 mg/kg body weight per day, 5 days/week, for up to 17 weeks, with an interim sacrifice at 8 weeks (Hazleton Laboratories America Inc., 1983a, 1983b; NTP, 1993; Irwin et al., 1995). In rats, principal toxic lesions occurred in liver, kidney, and nasal turbinates. After 17 weeks of exposure, absolute liver weight was increased in males of all dose groups and in females from 16 mg/kg body weight per day; kidney weights were increased from 32 mg/kg body weight per day in males and 63 mg/kg body weight per day in females, accompanied by regenerative hyperplasia (observed at interim sacrifice). General toxicity was obvious from decreased body weight of males at 63 mg/kg body weight per day and of both sexes at 125 mg/kg body weight per day. All female rats receiving 250 mg 1,2,3-trichloropropane/kg body weight per day died by week 2, and all males receiving the same dose died by week 5. At 125 mg/kg body weight per day, one male died by the end of week 5, and four females died during the studies. At the two highest dosages (125 and 250 mg/kg body weight per day), histopathology revealed changes indicative of extensive cellular damage in liver, kidneys, and nasal turbinates. Changes in liver enzymes, pseudocholinesterase, urea nitrogen, creatinine, and bilirubin also pointed to hepatocellular damage. Haematological changes at 16 mg/kg body weight per day and higher dosages were interpreted as non-regenerative anaemia, possibly associated with a depression in erythropoiesis (Hazleton Laboratories America Inc., 1983a). The LOAEL for a 17-week treatment by gavage was 8 mg/kg body weight per day in male rats and 16 mg/kg body weight per day in female rats.
In mice in the same study, main targets of toxic action were lung, liver, and forestomach. Hyperplasia of bronchiolar epithelium and hyperplasia and hyperkeratosis of forestomach were induced by a dosage of 63 mg/kg body weight per day and higher in females. From 125 mg/kg body weight per day, liver weights were significantly increased, accompanied by severe histological changes of the liver at the higher dosage in both sexes. The 250 mg/kg body weight per day dose was distinctly toxic, with increased mortality (16/20 males and 7/20 females) and necrosis of liver and lung tissue (Hazleton Laboratories America Inc., 1983b). Mice tolerated higher doses than rats, with LOAELs of 63 mg/kg body weight per day in female mice and 125 mg/kg body weight per day in male mice.
1,2,3-Trichloropropane in corn oil was administered to male and female Sprague-Dawley rats at doses of 0.01, 0.05, 0.2, and 0.8 mmol/kg body weight per day (1.5, 7.4, 29, and 118 mg/kg body weight per day) for 10 consecutive days and 0.01, 0.05, 0.1, and 0.4 mmol/kg body weight per day (1.5, 7.4, 15, and 59 mg/kg body weight per day) for 90 days (Merrick et al., 1991). Weight gain suppression occurred at a dose of 118 mg/kg body weight per day after 10 days. Liver and kidney weights were increased relative to control in the two highest groups after 10- and 90-day exposures. Serum chemistries and histopathology indicated a mild hepatotoxic response to 1,2,3-trichloropropane in the highest group of each study, but there was no renal toxicity.
In the same study, proliferative or neoplastic lesions in liver, lungs, mammary gland, and forestomach were reported in groups of 10 male and 10 female Sprague-Dawley rats administered 1,2,3-trichloropropane in corn oil by gavage in daily dosages of 0.05 or 0.4 mmol/kg body weight per day (7.4 or 59 mg/kg body weight per day) at the end of the 90-day treatment period (Merrick et al., 1991).
In the same study, myocardial necrosis and degeneration occurred in a diffuse pattern with marked eosinophila of affected cells. Male and female animals showed a cardiopathic response only at a dose of 118 mg/kg body weight per day after the 10-day exposure. In the 90-day study, the incidence of cardiopathy was higher in males than in females. Adverse effects on myocardium were reported at the lowest dosage of 1.5 mg/kg body weight per day (inflammation: 3/10 males, 1/10 females; necrosis: 2/10 males), but a progression to distinctly higher incidences was observed only for the highest dosage of 58.8 mg/kg body weight per day (inflammation: 8/10 males and females; degeneration: 5/10 males, 8/10 females; necrosis: 6/10 males, 7/10 females) (Merrick et al., 1991).
The cardiotoxic effect could not be confirmed in the National Toxicology Program (NTP) studies with F344/N rats covering a comparable treatment period and in longer-term studies (NTP, 1993). Gavage application in dosages up to 30 mg/kg body weight per day induced no dose-dependent increases in incidence of cardiomyopathy in male rats, and control animals already had high incidences. Likewise, treated B6C3F1 mice in the NTP study did not show any increased cardiotoxicity (NTP, 1993; Irwin et al., 1995).
1,2,3-Trichloropropane (>99% purity) in corn oil was administered by gavage to F344/N rats and B6C3F1 mice of both sexes (60 per group) at doses of 3, 10, and 30 mg/kg body weight per day and 6, 20, and 60 mg/kg body weight per day, respectively, for 5 days/week for an intended 104 weeks (NTP, 1993; Irwin et al., 1995; see Tables 2–5). Up to 10 animals per group were scheduled for interim evaluation at 15 months. Because of reduced survival associated with the development of chemical-related neoplasms in rats in the lowest dose groups, rats receiving 30 mg/kg body weight per day were terminated at 65 (females) or 76 weeks (males), and mice receiving 60 mg/kg body weight per day were terminated at 73 (females) or 79 weeks (males); mice receiving 20 mg/kg body weight per day were terminated at 88 weeks (NTP, 1993; Irwin et al., 1995).
Rats showed significantly decreased body weight at 30 mg/kg body weight per day from week 15 in males and from week 53 in females. In male rats, a significant increase in hyperplasia was evident in the forestomach and pancreas at >3 mg/kg body weight per day and in the kidney at >10 mg/kg body weight per day. In female rats, the incidence of hyperplasia in the forestomach and pancreas was statistically significant at >3 mg/kg body weight per day and in the kidney at >30 mg/kg body weight per day. In all dose groups of mice, eosinophilic and basophilic liver foci were noted. Focal hyperplasia of squamous epithelium of the forestomach was significantly increased in male mice of all dose groups, but only in high-dose female mice. Pathological findings were dominated by preneoplastic and neoplastic lesions (NTP, 1993; Irwin et al., 1995).
Table 2: Incidence of non-neoplastic lesions and neoplasms after administration of 1,2,3-trichloropropane by gavage to male rats.a
|
Control |
3 mg/kg body weight per day |
10 mg/kg body weight per day |
30 mg/kg body weight per day |
|
|
Animals surviving to 104 weeksb (main study) |
34/50 |
32/50 |
14/49 |
0/52 |
|
Mean survival (days) |
647 |
661 |
596 |
465 |
|
Incidences of lesions and neoplasms |
||||
|
Oral mucosa |
60 |
60 |
59 |
60 |
|
Squamous cell papilloma |
0 |
4 |
10** |
22** |
|
Squamous cell carcinoma |
1 |
0 |
11** |
25** |
|
Forestomach |
60 |
60 |
59 |
60 |
|
Hyperplasia, basal cell |
0 |
7* |
12** |
9** |
|
Hyperplasia, squamous |
3 |
28** |
13* |
6 |
|
Squamous cell papilloma |
0 |
31** |
36** |
46** |
|
Squamous cell carcinoma |
0 |
9** |
28** |
14** |
|
Pancreas |
60 |
60 |
59 |
60 |
|
Hyperplasia |
28 |
48** |
53** |
56** |
|
Adenoma |
5 |
21** |
37** |
31** |
|
Adenocarcinoma |
0 |
0 |
2 |
1 |
|
Kidney |
60 |
60 |
59 |
60 |
|
Hyperplasia |
0 |
1 |
23** |
35** |
|
Adenoma |
0 |
2 |
20** |
26** |
|
Preputial gland |
59 |
57 |
59 |
58 |
|
Adenoma |
5 |
3 |
6 |
11* |
|
Carcinoma |
0 |
3 |
3 |
6 |
|
Adenoma or carcinoma |
5 |
6 |
9 |
17** |
|
a |
From NTP (1993); Irwin et al. (1995). |
|
b |
The reduction in life span of the animals was caused by tumours induced by 1,2,3-trichloropropane. |
|
c |
Significantly different (* P < 0.05; ** P < 0.01) from controls by the life table test (squamous cell carcinoma) or by logistic regression (all other lesions). |
|
d |
Number of rats per group necropsied (oral mucosa) or with tissue examined microscopically (forestomach, pancreas, kidney, preputial gland) given in italics. |
|
e |
Number of rats with lesion or neoplasm given in bold. |
Data from the NTP study (NTP, 1993; Irwin et al., 1995) show 1,2,3-trichloropropane to be a multisite carcinogen in both rats and mice, even at the lowest dose. Main targets of tumorigenesis were squamous epithelium cells of forestomach and oral mucosa. Tumour incidences were significantly elevated in oral mucosa in the mid- and high-dose groups of rats, but only in high-dose female mice. Forestomach tumours were noted in all dose groups of both species.
Benign neoplasms of pancreas and kidney were also observed in male rats (significant at low and mid doses, respectively), together with a morphological continuum of hyperplasia, which was regarded as a preneoplastic lesion. Incidences of hyperplasia were also significantly increased in females in these tissues without simultaneous increases in incidence of neoplasms. The preputial gland and the clitoral gland, as homologous organs in male and female rats, respectively, showed a significant increase in the combined incidences of adenomas or carcinomas for high-dose males and mid- and high-dose females. In female rats, adenocarcinomas of the mammary gland were dose-dependently increased with a significant effect for the mid and high doses, whereas the incidence of fibroadenomas decreased with increasing dose, even below control incidences (NTP, 1993; Irwin et al., 1995).
Tumour types uncommon in F344 rats were also observed. Carcinomas of the Zymbal’s gland appeared in one female in the low-dose group and in three males and four females in high-dose groups (including interim sacrifice; significant effect). Adenomatous polyps or adenocarcinomas of the intestine occurred in two males and one female of the mid-dose group and three males and two females of the high-dose group. In view of the reduced number of animals at risk, both tumour types were considered to be possibly related to administration of 1,2,3-trichloropropane (NTP, 1993; Irwin et al., 1995).
Table 3: Incidence of non-neoplastic lesions and neoplasms after administration of 1,2,3-trichloropropane by gavage to female rats.a
|
Control |
3 mg/kg body weight per day |
10 mg/kg body weight per day |
30 mg/kg body weight per day |
|
|
Animals surviving to 104 weeksb (main study) |
31/50 |
30/49 |
8/52 |
0/52 |
|
Mean survival (days) |
649 |
654 |
580 |
366 |
|
Incidences of lesions and neoplasms |
||||
|
Oral mucosa |
60 |
59 |
60 |
60 |
|
Squamous cell papilloma |
1 |
5 |
10** |
21** |
|
Squamous cell carcinoma |
0 |
1 |
21** |
23** |
|
Forestomach |
60 |
59 |
59 |
60 |
|
Hyperplasia, basal cell |
0 |
10** |
5* |
9** |
|
Hyperplasia, squamous |
1 |
26** |
15** |
16** |
|
Squamous cell papilloma |
0 |
14** |
37** |
24** |
|
Squamous cell carcinoma |
0 |
3 |
9** |
6** |
|
Pancreas |
60 |
59 |
60 |
60 |
|
Hyperplasia |
5 |
15* |
24** |
11** |
|
Adenoma |
0 |
0 |
2 |
0 |
|
Kidney |
60 |
57 |
60 |
59 |
|
Hyperplasia |
0 |
2 |
3 |
12** |
|
Adenocarcinoma |
0 |
0 |
0 |
1 |
|
Clitoral gland |
56 |
56 |
58 |
59 |
|
Adenoma |
5 |
11 |
14** |
12* |
|
Carcinoma |
0 |
0 |
4 |
6 |
|
Adenoma or carcinoma |
5 |
11 |
18** |
17* |
|
Mammary gland |
60 |
59 |
60 |
60 |
|
Fibroadenoma or adenoma |
16 |
23 |
22* |
2 |
|
Adenocarcinoma |
1 |
6 |
12** |
22** |
|
a |
From NTP (1993); Irwin et al. (1995). |
|
b |
The reduction in life span of the animals was caused by tumours induced by 1,2,3-trichloropropane. |
|
c |
Significantly different (* P < 0.05; ** P < 0.01) from controls by the life table test (squamous cell carcinoma and adenocarcinoma) or by logistic regression test (all other lesions). |
|
d |
Number of rats per group necropsied (oral mucosa or mammary gland) or with tissue examined microscopically (forestomach, pancreas, kidney, clitoral gland) given in italics. |
|
e |
Number of rats with lesion or neoplasm given in bold. |
Hepatocellular neoplasms were significantly increased in mid-dose (adenomas or carcinomas combined) and high-dose males and females (adenomas), accompanied by the induction of eosinophilic or basophilic liver foci. Incidences of Harderian gland adenomas of male and female mice as well as incidences of uterine stromal polyps, adenomas, or adenocarcinomas exceeding historical incidences were considered treatment-related (NTP, 1993; Irwin et al., 1995).
Among 140 carcinogenic chemicals tested in long-term animal studies, 1,2,3-trichloropropane was among the seven producing uterine neoplasms in mice (spontaneous tumour rate 0.3%) (Griesemer & Eustis, 1994). In a database comprising 536 carcinogens, only 12 substances, including 1,2,3-trichloropropane, produced uterine neoplasms in mice (Benigni & Pino, 1998).
The lower survival of certain study groups may explain the incongruities in certain dose–responses of tumour incidences between males and females and mid and high doses, e.g., for oral mucosa and forestomach neoplasms in rats. Reduced survival also reduced the number of animals at risk for the development of certain late-appearing tumour types (NTP, 1993; Irwin et al., 1995; La et al., 1996).
A detailed description of in vitro and in vivo genotoxicity studies is given in MAK (1993) and IARC (1995). 1,2,3-Trichloropropane was mutagenic to bacteria. Gene mutation, sister chromatid exchange, and chromosomal aberrations, but not DNA damage, were induced in rodent cells in vitro. In single studies, DNA binding and induction of DNA breaks, but not of dominant lethal mutations, were reported in rodents treated in vivo (IARC, 1995).
1,2,3-Trichloropropane is mutagenic in bacterial test systems with Salmonella typhimurium strains TA 97, TA 100, and TA 1535 and E. coli WP2 uvr A in the presence of metabolic activation (Dean & Brooks, 1979; Stolzenberg & Hine, 1980; Kier, 1982; Haworth et al., 1983; Ratpan & Plaumann, 1988; NTP, 1993; Låg et al., 1994). Some of these studies also reported an increase of revertants in Salmonella strains TA 98 (Kier, 1982; NTP, 1993) and TA 1537 (Dean & Brooks, 1979), while others found negative test results with one or both of these strains (Dean & Brooks, 1979; Kier, 1982; Haworth et al., 1983; Ratpan & Plaumann, 1988; NTP, 1993) as well as with TA 1538 (Dean & Brooks, 1979; Kier, 1982; Ratpan & Plaumann, 1988). However, only a single publication points to a dose-dependent weak direct mutagenic activity of 1,2,3-trichloropropane in S. typhimurium TA 1535 (Dean & Brooks, 1979) in contrast to all other studies, despite comparable test conditions. The results demonstrate that 1,2,3-trichloropropane is a substance that, after metabolic activation, can cause both base pair substitution and frame shift mutation.
Table 4: Incidence of non-neoplastic lesions and neoplasms after administration of 1,2,3-trichloropropane by gavage to male mice.a
|
Control |
6 mg/kg body weight per day |
20 mg/kg body weight per day |
60 mg/kg body weight per day |
|
|
Animals surviving to 104 weeksb (main study) |
42/51 |
18/51 |
0/54 |
0/56 |
|
Mean survival (days) |
655 |
617 |
531 |
470 |
|
Incidences of lesions and neoplasms |
||||
|
Oral mucosa |
60 |
59 |
60 |
60 |
|
Squamous cell papilloma |
0 |
0 |
0 |
2 |
|
Forestomach |
60 |
59 |
60 |
60 |
|
Hyperplasia, squamous |
8 |
37** |
32** |
38** |
|
Squamous cell papilloma |
3 |
35** |
25** |
35** |
|
Squamous cell carcinoma |
0 |
41** |
54** |
55** |
|
Liver |
60 |
59 |
60 |
60 |
|
Hepatocellular adenoma |
12 |
18 |
21* |
31** |
|
Hepatocellular carcinoma |
4 |
11* |
6 |
3 |
|
Harderian gland |
60 |
59 |
60 |
60 |
|
Adenoma |
1 |
2 |
10** |
11** |
|
a |
From NTP (1993); Irwin et al. (1995). |
|
b |
The reduction in life span of the animals was caused by tumours induced by 1,2,3-trichloropropane. |
|
c |
Significantly different (* P < 0.05; ** P < 0.01) from controls by the life table test (squamous cell carcinoma) or the logistic regression test (all other lesions). |
|
d |
Number of mice per group necropsied (oral mucosa and Harderian gland) or with tissue examined microscopically (forestomach and liver) given in italics. |
|
e |
Number of mice with lesion or neoplasm given in bold. |
Table 5: Incidence of non-neoplastic lesions and neoplasms after administration of 1,2,3-trichloropropane by gavage to female mice.a
|
Control |
6 mg/kg body weight per day |
20 mg/kg body weight per day |
60 mg/kg body weight per day |
|
|
Animals surviving to 104 weeksb (main study) |
41/50 |
13/50 |
0/51 |
0/55 |
|
Mean survival (days) |
661 |
601 |
515 |
453 |
|
Incidences of lesions and neoplasms |
||||
|
Oral mucosa |
60 |
60 |
60 |
60 |
|
Squamous cell papilloma |
1 |
0 |
1 |
0 |
|
Squamous cell carcinoma |
0 |
0 |
1 |
5** |
|
Forestomach |
60 |
60 |
60 |
60 |
|
Hyperplasia, squamous |
11 |
25** |
23** |
36** |
|
Squamous cell papilloma |
0 |
28** |
27** |
33** |
|
Squamous cell carcinoma |
0 |
47** |
55** |
51** |
|
Liver |
60 |
60 |
60 |
60 |
|
Hepatocellular adenoma |
7 |
9 |
9 |
36** |
|
Hepatocellular carcinoma |
1 |
3 |
0 |
2 |
|
Harderian gland |
60 |
60 |
60 |
60 |
|
Adenoma |
3 |
6 |
7 |
10* |
|
Uterus |
60 |
60 |
60 |
59 |
|
Stromal polyp |
0 |
2 |
2 |
7** |
|
Adenoma |
0 |
1 |
0 |
4 |
|
Carcinoma |
0 |
4** |
3** |
8** |
|
Adenoma or carcinoma |
0 |
5** |
3** |
12** |
|
a |
From NTP (1993); Irwin et al. (1995). |
|
b |
The reduction in life span of the animals was caused by tumours induced by 1,2,3-trichloropropane. |
|
c |
Significantly different (* P < 0.05; ** P < 0.01) from controls by the life table test (squamous cell carcinoma and uterine adenocarcinoma) or the logistic regression test (all other lesions). |
|
d |
Number of mice necropsied (oral mucosa, Harderian gland, or uterus) or with tissue examined microscopically (forestomach and liver) given in italics. |
|
e |
Number of mice with lesion or neoplasm given in bold. |
1,2,3-Trichloropropane (2 and 4 mmol/litre = 294 and 588 mg/litre) showed DNA breakage in human lymphocytes detected by a comet assay in both the presence and absence of S9 mix, but was negative in the micronucleus assay (2–8 mmol/litre = 294–1176 mg/litre) (Tafazoli et al., 1996, 1998; Anderson et al., 1998).
Studies in primary hepatocytes yielded negative test results for both the induction of DNA strand breaks (30–100 µmol/litre = 5–15 mg/litre; Holme et al., 1991) and unscheduled DNA synthesis (0.001% = 10 mg/litre; Williams et al., 1989).
Metabolic activation of 1,2,3-trichloropropane by S9 mix was a prerequisite to the induction of gene mutations in mouse lymphoma cells (Sawin & Hass, 1982; NTP, 1993), gene conversions in Saccharomyces cerevisiae (Dean & Brooks, 1979), sister chromatid exchange in CHO and V79 cells (von der Hude et al., 1987; NTP, 1993), chromosomal aberrations in CHO cells (NTP, 1993), and micronuclei in metabolically competent human lymphoblastoid cell lines (AHH-1, H2E1, and MCL-5) (Doherty et al., 1996; Parry et al., 1996). A chromosomal aberration test was negative with RL1 rat liver cells (Dean & Brooks, 1979).
DNA single strand breaks were detectable by alkaline elution as early as 60 min after one intraperitoneal injection of 1,2,3-trichloropropane, with LOAELs (significant effects) of 55 mg 1,2,3-trichloropropane/kg body weight in rat kidney (Låg et al., 1991) and 30 mg 1,2,3-trichloropropane/kg body weight in rat liver (Weber & Sipes, 1991).
A dominant lethal test demonstrated the absence of genotoxic activity in this special test (Saito-Suzuki et al., 1982).
Negative test results for a micronucleus test in mouse bone marrow (Douglas et al., 1985) and unscheduled DNA synthesis in rat hepatocytes in vivo (Mirsalis et al., 1983) mentioned in two abstracts cannot be validated because of lack of documentation (e.g., dose, test conditions).
Exposure of rats to 800 mg 1,2,3-trichloropropane/m3 for 1 week resulted in a disturbance of hepatocyte mitosis. In a comparison of hepatocytes from control and exposed rats, there was a significant shift from binuclear cells to mononuclear polyploid cells. While binuclear diploid and tetraploid cells were significantly reduced in treated animals, mononuclear tetraploid and octaploid cells showed a significant corresponding increase, and cells with a ploidy of 16n appeared (Belyaeva et al., 1974).
Significant DNA adduct levels (as 14C equivalents of 1,2,3-trichloropropane) were measured in several target and non-target tissues 6 h after a single administration by gavage (La et al., 1995). One major adduct seemed to be formed. Dosages were 3 and 30 mg/kg body weight in male Fischer 344 rats and 6 and 60 mg/kg body weight in male B6C3F1 mice (low and high dosing of the carcinogenicity study [NTP, 1993]). In the rat, adduct levels ranged from 0.8 to 6.6 µmol/mol guanine and from 7.1 to 47.6 µmol/mol guanine for the low and high doses, respectively. The order of tissue adduct levels was kidney, liver, and pancreas > tongue and glandular stomach > forestomach > spleen. Adducts were not detectable in rat preputial gland or palate. In the mouse, adduct yields ranged from 0.32 to 28.1 µmol/mol guanine for the low dose and from 12.2 to 208.1 µmol/mol guanine for the high dose with the following ranking: glandular stomach > liver and forestomach > kidney > lung and spleen > brain, heart, and testes. Generally, there was a correlation between adduct formation and targets of tumorigenesis (see NTP, 1993). Targets for both end-points were forestomach and liver in mice and kidney, liver, pancreas, tongue, and forestomach in rats. Notable exceptions were relatively high DNA adduct concentrations in mouse glandular stomach and rat liver without corresponding tumours. Despite high tumour rates in palate and preputial gland of rats, no adducts were detectable, which was attributed to a lack of sensitivity of detection.
As the yields of DNA adducts from animals exposed to [14C]1,2,3-trichloropropane were not enough for structural characterization, 300 mg/kg body weight of the radiolabelled compound was administered intraperitoneally to rats. In the liver, the major DNA adduct was identified as S-[l-(hydroxymethyl)-2-(N7-guanyl)ethyl]glutathione (La et al., 1995), confirming the role of GSH for adduct formation (Weber & Sipes, 1990, 1992). This N7-guanyl adduct may play a crucial role in the genotoxic activity of 1,2,3-trichloropropane, as corresponding adducts of other haloethyl GSH conjugates induced mutations at specific G:C base pairs in Salmonella typhimurium. Furthermore, the subsequent formation of abasic sites and reactive episulfonium ions is possible from such adducts (La et al., 1996).
Further investigations concentrated on the formation of this major adduct, S-[l-(hydroxymethyl)-2-(N7-guanyl)ethyl]glutathione, and the induction of cell proliferation in male B6C3F1 mice in two target (forestomach and liver) and two non-target organs (glandular stomach and kidney) after repeated dosing of 6 mg 1,2,3-trichloropropane/kg body weight per day for 5 days either by gavage or by drinking-water. In comparison with the drinking-water exposure, the gavage bolus dose induced elevated concentrations of the DNA adduct (determination 24 h after last administration) in forestomach, liver, and kidney (significant effect for liver and kidney, approximately 2-fold), but not in glandular stomach. After a 2-week exposure (5 days/week), cell proliferation (BrdU incorporation in DNA) was increased in all four tissues relative to vehicle-treated control groups after the gavage exposure, but not after the drinking-water exposure. Significant differences between the two exposure routes were noted in glandular stomach at 18 h post-administration only, in kidney and liver at 30 h post-administration only, and in forestomach at both time points (La et al., 1996). It appears that the high local concentrations to be expected from the gavage bolus dose led to significant adduct formation and cell proliferation in contrast to the continuous but lower local concentrations resulting from drinking-water exposure (Swenberg et al., 1995; La & Swenberg, 1996; La et al., 1996).
In a study under the NTP’s Continuous Breeding Protocol, 30, 60, or 120 mg 1,2,3-trichloropropane/kg body weight per day (doses chosen from results of a dose finding study: Task 1) was administered by gavage in corn oil to Swiss CD-1 mice (Gulati et al., 1990; Chapin et al., 1997). Twenty pairs per dose group and 40 pairs per control group were continuously exposed for a 7-day precohabitation and a 98-day cohabitation period to evaluate adverse effects on the F0 generation and their litters (continuous breeding phase: Task 2). Subsequently, F0 mice from control and high-dose groups were used in a cross-over mating trial to determine the sex affected by 1,2,3-trichloropropane treatment (Task 3: mating procedure control males × control females; control males × high-dose females; high-dose males × control females). For assessment of the fertility of the F1 generation, Task 4 comprised a mating trial of F1 mice from the last litters of Task 2, which had continuously been exposed to 1,2,3-trichloropropane during gestation, via lactation, and by gavage to the same concentrations as their parents from weaning to sexual maturity (about 74 days of age) and during a 7-day cohabitation period until delivery (duration of total study 28–30 weeks).
1,2,3-Trichloropropane treatment caused dose-related impairment of fertility in the absence of gross general toxicity. In the high-dose group (120 mg 1,2,3-trichloropropane/kg body weight per day), fewer pairs of the F0 generation delivered third, fourth, and fifth litters, and the litters had fewer live pups. Parental body weights were not decreased. In both male and female F0 mice, liver weights were increased; female kidney and ovary weights were reduced; and epididymal weight was slightly reduced in the high-dose group. Testis weight and sperm parameters were unchanged. In the cross-over mating trial (Task 3), treated females mated to control males produced fewer live pups; no effect on fertility was found with treated males mated to control females. These data suggest an impairment of female fertility. The fertility index in the second-generation pups fed 1,2,3-trichloropropane was also significantly reduced. The number of pups trended lower at the high dose. In this generation (F1), in contrast to the F0 generation, the estrous cycle length was also increased at all three dose levels. The only conspicuous finding of the histopathological examination of selected organs in Task 3 was four cases of amyloidosis of the ovary among 10 treated females compared with 0/13 in the control group.
As only slight systemic toxicity was present, it can be concluded that 1,2,3-trichloropropane impairs fertility and reproduction at a dose level of 120 mg/kg body weight per day in both the parent and the offspring generations (Gulati et al., 1990). The cross-mating trial suggests slightly greater toxicity for the female than for the male reproductive system. As average estrous cycle lengths were significantly increased for all 1,2,3-trichloropropane-exposed females of the F1 generation, the LOAEL for reproductive toxicity is 30 mg/kg body weight per day.
Other more limited studies that had been performed previously showed mostly negative results. This may be due to poor study design or problems during the study.
In a one-generation reproduction/fertility study, CD rats (10 males and 20 females per group) were exposed to 0, 3.1, or 9.2 mg/m3 (Johannsen et al., 1988) for 6 h/day, 5 days/week, over a premating period of 10 weeks, the mating period of up to 40 days for both sexes, and the gestation period of 14 days (days 0 through 14) for females only. Females of the F0 generation and offspring were evaluated during a 21-day lactation period. No treatment-related effects on the parent generation or on reproductive parameters were observed (Johannsen et al., 1988). In a previous study (Bio/dynamics Inc., 1980; Johannsen et al., 1988) using 0, 27.5, and 92 mg 1,2,3-trichloropropane/m3, the weights and the histopathological examination of gonads of the F0 generation revealed no adverse effects of 1,2,3-trichloropropane exposure on testes, epididymides, or ovaries; however, in view of the poor mating performance, only limited conclusions on 1,2,3-trichloropropane-related effects on fertility and reproduction are possible.
Fifteen male Sprague-Dawley rats treated with 80 mg 1,2,3-trichloropropane/kg body weight per day for 5 days by gastric intubation (vehicle olive oil) during a dominant lethal study showed no impairment of mating performance and no meaningful changes in the indices measured, such as numbers of implants, live embryos, and dead implants (Saito-Suzuki et al., 1982).
Male rats administered 1,2,3-trichloropropane by gavage at a dosage of 125 mg/kg body weight per day for 120 days showed significant increases in relative testes weights after 8 and 17 weeks of administration and a significant decrease in relative epididymis weight at the 8-week time point only. However, no effects on sperm count or sperm morphology and no histomorphological changes of testes or epididymis were observed (Hazleton Laboratories America Inc., 1983a). Similarly treated mice showed differences in numbers of sperm per milligram cauda of epididymis, which were considered to be of questionable significance in view of the absence of any significant findings on weights of testes or epididymis or any histopathological findings (Hazleton Laboratories America Inc., 1983b).
Sprague-Dawley rats were exposed to 37 mg 1,2,3-trichloropropane/kg body weight in corn oil (determined as the maximum tolerable dose in dose–response studies) by daily intraperitoneal injection from gestation days 1 to 15 and sacrificed on gestational day 21. Maternal toxicity was apparent in the absence of any fetotoxic or teratogenic effects. These results cannot be validated, as details of the test results are not specified in the review article (Hardin et al., 1981).
In addition to these data, reproduction studies by the inhalation route (Johannsen et al., 1988) or by the oral route (Gulati et al., 1990; Chapin et al., 1997) found no external malformation or fetal toxicity effects on the F0 offspring.
The endogenous formation of reactive intermediates from 1,2,3-trichloropropane plays a crucial role in the organotoxic, genotoxic, and carcinogenic action of 1,2,3-trichloropropane. The postulated formation of alkylating species is consistent with the presence of covalently bound radioactivity in liver and kidney and in target tissues of carcinogenic action after gavage administration of [14C]1,2,3-trichloropropane. Significant DNA adduct levels (as 14C equivalents of 1,2,3-trichloropropane) were found in forestomach and liver in mice and in kidney, liver, pancreas, tongue, and forestomach in rats (Weber & Sipes, 1990; La et al., 1995). A predominant role was assigned to episulfonium ions formed in situ during the metabolism of 1,2,3-trichloropropane in the liver and from processing of GSH conjugates of 1,2,3-trichloropropane in the kidney, leading to hepatotoxic and nephrotoxic lesions (NTP, 1993).
A further publication, in abstract form only without detailed documentation of test conditions and results, presented a new hypothesis for the formation of forestomach tumours (Ito et al., 1996). According to these investigations, analysis of ras mutations in forestomach tumours of mice from the NTP (1993) study showed that the observed mutations are not consistent with the miscoding properties of the major DNA adduct S-[l-(hydroxymethyl)-2-(N7-guanyl)ethyl]glutathione that had formerly been identified in various tissues by the same working group (La et al., 1995, 1996). Preliminary studies suggested that 1,2,3-trichloropropane administration produced increases in the etheno DNA adducts 1,N6-ethenodeoxyadenosine and 3,N4-ethenodeoxycytidine, which are thought to arise endogenously from lipid oxidation. The resulting alternative hypothesis for the formation of forestomach tumours is an indirect mutagenic effect of 1,2,3-trichloropropane by GSH depletion as a consequence of the bolus application by gavage, which may induce lipid oxidation. Consequently, GSH plays a crucial role in adduct formation, being involved both in the formation of the major DNA adduct S-[l-(hydroxymethyl)-2-(N7-guanyl)ethyl]glutathione and in the initiation of lipid oxidation (Ito et al., 1996).
Studies on possible mechanisms of 1,2,3-trichloropropane-induced carcinogenesis have shown that DNA adduct formation in male B6C3F1 mice approximately doubled after administration of 6 mg 1,2,3-trichloropropane/kg body weight per day for 1 week by gavage compared with administration of the same dose via drinking-water. Further, only gavage exposure led to a significant increase in cell proliferation rate compared with administration in drinking-water (La et al., 1996). It appears that the high local concentrations to be expected from the gavage bolus dose led to significant adduct formation and cell proliferation, in contrast to the continuous but lower local concentrations resulting from drinking-water exposure. Consequently, it has to be expected that gavage exposure will overestimate the carcinogenic potency of 1,2,3-trichloropropane (Swenberg et al., 1995; La & Swenberg, 1996; La et al., 1996). A number of chemicals, including the structurally related 1,2-dibromo-3-chloropropane, are also known to induce high incidences of forestomach tumours, but only when administered via gavage (La et al., 1996).
After a 15-min exposure of 12 persons to 1,2,3-trichloropropane vapours, a concentration of 610 mg/m3 was described as an irritant to eyes and throat (insufficient documentation) (Silverman et al., 1946).
1,2,3-Trichloropropane may enter the hydrosphere due to its production, use, and application. However, it can evaporate from the aqueous phase to some extent, thus reducing the exposure concentration for aquatic species. Such loss due to evaporation of 1,2,3-trichloropropane from the aqueous phase was demonstrated in a recent study simulating a 48-h Daphnia magna open vessel test (Solvay, 2001b). Therefore, long-term toxicity tests performed with this compound should account for this potential loss by employing a test design that maintains constant exposure, thus improving the reliability of data obtained.
Several tests have been performed to establish the acute toxicity of 1,2,3-trichloropropane to freshwater biota representing different trophic levels, and these are summarized in Table 6. Most of these studies are of limited value, because initial concentrations of 1,2,3-trichloropropane were not measured and losses of 1,2,3-trichloropropane due to volatilization were not taken into account over the tests’ duration.
Use of closed test systems to reduce these losses has recently been reported for two tests with the microalga Selenastrum capricornutum and the cladoceran Daphnia magna (Solvay, 2001a, 2002).
The acute toxicity of 1,2,3-trichloropropane to Daphnia magna (end-point = immobilization after 48 h) was determined in closed glass flasks following OECD guideline 202 (Solvay, 2002). 1,2,3-Trichloropropane concentrations in the test flasks were determined by gas chromatography at the beginning and end of the test. Losses of 1,2,3-trichloropropane over the 48-h test duration ranged from 6 to 32%; therefore, the mean measured concentrations (0, 2.5, 4.7, 8.4, 15, and 27 mg/litre) were used to calculate the toxicity test end-points. No test concentrations gave partial immobility; therefore, the 48-h EC50 was estimated to be 20 mg/litre (95% confidence interval of 15–27 mg/litre), with a no-observed-effect concentration (NOEC) of 15 mg/litre.
Assays performed by Blum & Speece (1991) employing methanogens (end-point = inhibition of gas production), aerobic heterotrophic bacteria (end-point = inhibition of oxygen uptake), Nitrosomonas sp. (end-point = inhibition of ammonia consumption), and Photobacterium phosphoreum (end-point = inhibition of bioluminescence) showed that the methanogens exhibited the lowest IC50 value of 0.63 mg/litre.
The most sensitive of three algal species tested was Selenastrum capricornutum, with a 72-h EBC50 value of 49.6 mg/litre (end-point = inhibition of biomass formation) and a 72-h ERC50 value of 101 mg/litre (end-point = inhibition of growth rate; Solvay, 2001a).
For those invertebrates tested, the lowest EC50 value of 4.1 mg/litre (end-point = immobilization) — which was in fact several-fold lower than the corresponding values reported for Daphnia magna — was reported by Rose et al. (1998) for the cladoceran Ceriodaphnia cf. dubia. Possible reasons for this apparently higher sensitivity, such as differences in experimental conditions, water chemistry, or organism size to test volume ratio, were excluded by these authors. They advocate that the variation in sensitivity was probably due to an inherent sensitivity or to the dissimilar size of the different species.
From studies relating to the long-term toxicity of 1,2,3-trichloropropane and using the invertebrate Chaetogammarus marinus, Kooijman (1981) reported as the lowest value a 21-day LC50 of 20 mg/litre.
In toxicity studies conducted with several fish species, the lowest LC50 value (7-day incubation) was established for guppies (Poecilia reticulata) as 41.6 mg 1,2,3-trichloropropane/litre (Könemann, 1981). For Oncorhynchus mykiss (ABC, 1986c), the lowest estimated NOEC (96 h) value was <10 mg/litre.
Walton et al. (1989) investigated the effect of a nominal concentration of 1000 mg 1,2,3-trichloropropane/kg soil upon the microbial activity in two different soils (silty and sandy loam) by measuring soil respiration as an indicator for microbial activity. They found a reduction of carbon dioxide production of 0.09 and 0.18 µg/g soil per day after day 4 for silty and sandy loam, respectively. However, after 6 days of incubation, no significant difference between treated soils and controls was detectable. These results indicate a low toxicity potential of 1,2,3-trichloropropane for the terrestrial environment. Studies on the toxicity of 1,2,3-trichloropropane to terrestrial invertebrates or vertebrates or on effects on ecosystems are not available. However, employing ECOSAR Version 0.99g (a structure–activity relationship program provided by the US EPA that is used to predict the aquatic toxicity of chemicals based on their similarity of structure to chemicals for which the aquatic toxicity has been previously measured) and the measured physicochemical properties of 1,2,3-trichloropropane (relative molecular mass 147.43, log Kow 2.27, melting point –14.7 °C, water solubility 1750 mg/litre, ECOSAR class neutral organics), a 14-day LC50 value of about 640 mg/kg dry weight soil for earthworm toxicity can be estimated.
Table 6: Aquatic toxicity of 1,2,3-trichloropropane.
|
Species tested (reported test method) |
End-point (effect) |
Concentrationa (mg/litre) |
Reference |
|
Bacteria |
|||
|
Mixed culture of aerobic heterotrophs (inhibition of oxygen uptake) |
15-h IC50 |
290 (nominal) |
Blum & Speece (1991) |
|
Nitrosomonas sp. (inhibition of ammonia consumption) |
24-h IC50 |
30 (nominal) |
Blum & Speece (1991) |
|
Photobacterium phosphoreum (inhibition of bioluminescence) |
5-min IC50 |
19 (nominal) |
Blum & Speece (1991) |
|
Methanogens (not characterized) (inhibition of gas production) |
48-h IC50 |
0.63 (nominal) |
Blum & Speece (1991) |
|
Green algae |
|||
|
Chlorella vulgaris (inhibition of carbon dioxide uptake) |
3-h EC50 |
170 (nominal) |
Hutchinson et al. (1980) |
|
Chlamydomonas angulosa (inhibition of carbon dioxide uptake) |
3-h EC50 |
112 (nominal) |
Hutchinson et al. (1980) |
|
Selenastrum capricornutum (inhibition of biomass production and growth rate) |
72-h EBC50 |
49.6 (effectiveb) |
Solvay (2001a)c |
|
Invertebrates |
|||
|
Chaetogammarus marinus (mortality) |
48-h LC50 |
60 (not specified) |
Kooijman (1981) |
|
Daphnia magna (immobilization, static, unfed) |
48-h IC50 |
35 (nominal) |
Hermens et al. (1984) |
|
Daphnia magna (mortality and abnormal effects, static, fed) |
24-h LC50 |
41 (nominal) |
ABC (1986a) |
|
Daphnia magna (immobilization) |
48-h EC50 |
20 (effectiveb) |
Solvay (2002) |
|
Ceriodaphnia cf. dubia (immobilization) |
48-h EC50 |
4.1 (nominal) |
Rose et al. (1998) |
|
Vertebrates |
|||
|
Japanese medaka (Oryzias latipes) (mortality) |
48-h LC50 |
109 (nominal) |
MITI (1992) |
|
Bluegill sunfish (Lepomis macrochirus) (mortality, abnormal effects) |
24-h LC50 |
75 (nominal) |
ABC (1986b) |
|
Rainbow trout (Oncorhynchus mykiss) (mortality, abnormal effects) |
24-h LC50 |
75 (nominal) |
ABC (1986c) |
|
Fathead minnow (Pimephales promelas) (mortality) |
96-h LC50 |
66.5 (effectiveb) |
Geiger et al. (1990) |
|
Guppy (Poecilia reticulata) (mortality) |
7-day LC50 |
41.6 (not specified) |
Könemann (1981) |
|
a |
As 1,2,3-trichloropropane can evaporate from the aqueous phase (only recently demonstrated in studies simulating a 48-h Daphnia magna test in open vessels; Solvay, 2001b), which would therefore reduce the concentration of the test compound over time, we specified (in parentheses) whether the values given were based upon nominal (initial concentrations) or effective (loss of compound checked and accounted for) concentrations. |
|
b |
In these studies, end-points reported were based on mean measured 1,2,3-trichloropropane concentrations. |
|
c |
Performed according to OECD guideline 201. |
Data on the effects on humans are limited to the description of an irritant action on mucous membranes.
1,2,3-Trichloropropane is a multisite carcinogen in both rats and mice, even at the lowest dose, when administered in corn oil via gavage in dosages of 0, 3, 10, and 30 mg/kg body weight per day to F344/N rats and 0, 6, 20, and 60 mg/kg body weight per day to B6C3F1 mice for 5 days weekly (NTP, 1993; Irwin et al., 1995).
Main targets of carcinogenic action are the forestomach and the oral mucosa in rats of both sexes, the mammary gland in female rats, pancreas and kidney in male rats, as well as the preputial gland and clitoral gland, as homologous organs in male and female rats, respectively. Mice responded with neoplasms of the forestomach, the liver, and the Harderian gland. Uncommon tumour types were reported, such as carcinomas of the Zymbal’s gland and adenomatous polyps or adenocarcinomas of the intestine in rats and uterine neoplasms in mice (NTP, 1993; Irwin et al., 1995). Considering the very high incidences of forestomach neoplasms in low-dose groups of rats (33–66%) and mice (nearly 100%), this carcinogenic activity might have been detected even at lower doses, and the LOAELs for significantly increased tumour incidences will be well below 3 mg/kg body weight per day in rats and 6 mg/kg body weight per day in mice.
1,2,3-Trichloropropane was mutagenic to bacteria. Gene mutation, sister chromatid exchange, and chromosomal aberrations, but not DNA damage, were induced in rodent cells in vitro. In vivo, DNA single strand breaks were detectable by alkaline elution, whereas genotoxic action in a dominant lethal test was absent.
The DNA adduct S-[l-(hydroxymethyl)-2-(N7-guanyl)ethyl]glutathione was identified in preneoplastic and neoplastic lesions of target organs.
When 1,2,3-trichloropropane was administered to Sprague-Dawley rats via drinking-water for 13 weeks, the LOAELs for moderate hepatotoxic and nephrotoxic effects were 17.6 mg/kg body weight per day for female rats and 113 mg/kg body weight per day for male rats (Villeneuve et al., 1985).
Repeated inhalation exposure of F344 rats and B6C3F1 mice to 1,2,3-trichloropropane resulted in cytotoxic action in the upper respiratory tract, lung, and liver. Based on the most sensitive end-point — i.e., changes in the olfactory epithelium detectable by histopathological examination of nose tissue — the overall NOAEL was 6 mg/m3 for rats and 18 mg/m3 for mice derived from an 11-day inhalation study (Miller et al., 1986b).
The results of a two-generation study in Swiss mice showed that 1,2,3-trichloropropane impaired fertility and reproduction at a dose level of 120 mg/kg body weight per day (administered via gavage) in both the parent and the offspring generations in the presence of only slight systemic toxicity (Gulati et al., 1990; Chapin et al., 1997). A cross-mating trial suggested greater toxicity for the female than for the male reproductive system. The average estrous cycle length was significantly increased for all 1,2,3-trichloropropane-exposed females of the F1 generation, with the lowest dose being 30 mg/kg body weight per day. Developmental toxicity has not been reported, but the data are very limited.
Based on studies in animals, critical effects of 1,2,3-trichloropropane are irritation (dermal, inhalation) and cancer. Following inhalation exposure, respiratory tract irritation has been observed in rats at concentrations in the range of a few milligrams per cubic metre.
1,2,3-Trichloropropane is carcinogenic in rats and mice, inducing a wide range of tumours in both sexes. In the NTP gavage carcinogenicity studies, the reduction in life span of the animals was caused by tumours induced by 1,2,3-trichloropropane. Data on mode of action, including studies on metabolism, genotoxicity, and determination of DNA adduct formation, suggest that the mechanism of induction of tumours involves direct interaction of an active metabolite with genetic material. Hence, exposure to 1,2,3-trichloropropane should be avoided.3
While a tolerable concentration or tolerable intake has not been derived here, it should be noted that the tumorigenic potency of this compound is high, with tumours being observed at doses as low as 3 mg/kg body weight per day in rats following gavage administration.
The NTP gavage studies were chosen as key studies, as the drinking-water study was only of 13 weeks’ duration and relatively moderate changes were observed.
There is a lack of information on present-day uses of 1,2,3-trichloropropane. Older reports suggest that it has a use as a solvent and extractive agent, paint and varnish remover, and cleaning and degreasing agent. If so, then there could be widespread dermal and inhalation exposure for both occupational and general populations.
There are no data from such exposures, so estimations have been made from the scarce data available on concentrations in ambient and indoor air and drinking-water. The data on high exposures in air in Kuwait cannot be ignored, but there is no obvious explanation for them. Likewise, the value given for daily intake of 1,2,3-trichloropropane from food has not been substantiated.
Assuming a daily intake of 7.4 µg in food, a daily consumption of 2 litres of drinking-water containing 0.1 µg 1,2,3-trichloropropane/litre, a daily intake of 20 m3 air with an average load of 0.1 µg 1,2,3-trichloropropane/m3, and a body weight of 64 kg, a daily intake of 150 ng/kg body weight can be estimated (34 ng/kg body weight if the questionable food intake is not included).
Using the above assumptions but the reported much higher values for air in Kuwait (outdoor air 491 µg/m3; indoor air 2480 µg/m3) and assuming that 50% of the time is spent outdoors and 50% indoors, one can calculate a daily uptake of about 464 µg/kg body weight (nearly 0.5 mg/kg body weight per day).
Data on occupational exposure (Brock & Carroll, 1985) suggest a maximum short-term maintenance personnel exposure concentration of 17 mg 1,2,3-trichloropropane/m3 for a chemical plant in the USA. However, exposure concentrations in other workplaces did not usually exceed 0.61 mg/m3.
These workplace concentrations (workday situation: daily consumption of 20 m3 air, accrued 1-h peak concentration with 17 mg/m3, 7-h on-site ambient air with <0.61 mg/m3, remaining time ambient air with 0.1 µg/m3; intake via food and drinking-water not accounted for) give an intake of up to 277 µg/kg body weight per day, i.e., nearly 0.3 mg/kg body weight per day (compared with 3 mg/kg body weight per day for the rat carcinogenicity study).
Tumours occurred in rats and mice, even at the lowest dose; therefore, we do not have a no-effect level to compare with human population exposure. This makes a sample risk assessment problematic. However, it can be said that the mechanics for occupationally exposed persons above (0.3 mg/kg body weight per day) and for non-occupationally exposed persons exposed to concentrations as supposed by the Kuwaiti data (nearly 0.5 mg/kg body weight per day) suggest that these populations may have a higher risk of cancer than those exposed only to 1,2,3-trichloropropane in drinking-water and ambient air levels other than those cited in Kuwait (34 ng/kg body weight per day).
There is a lack of information on present uses and therefore on a general population exposure scenario.
There was only one report on occupational exposure to 1,2,3-trichloropropane, and this was not recent and could not be substantiated by other data.
Although there is likely to be widespread human exposure to 1,2,3-trichloropropane in polymer manufacture and when the chemical is used as a solvent for degreasing and paint stripping, there is a lack of information on dermal exposure.
Risk assessment may be affected by differences in the routes of exposure. Marked differences in toxicity have been reported between rats administered 1,2,3-trichloropropane in drinking-water (Villeneuve et al., 1985) and rats exposed by gavage (NTP, 1993). Hepatotoxicity, nephrotoxicity, and nasal and respiratory toxicity were observed after gavage administration (125 mg/kg body weight per day for 17 weeks), whereas increases in liver weight and mild histological lesions were observed when the chemical was administered in drinking-water (up to 113 mg/kg body weight per day for 9 days). However, it should be noted that the drinking-water study was only for 9 days’ administration. A 2-year drinking-water study was not available. However, 1,2,3-trichloropropane was found to induce up to 2.4 times more DNA adducts in mice via gavage than by drinking-water in a comparable exposure (6 mg/kg body weight per day) (La et al., 1996).
It is not known how relevant forestomach tumours after gavage administration in rats are for human risk assessment, as humans do not have this organ. However, there were tumours at other sites.
There is some uncertainty as to the relevance of changes in olfactory epithelium in rats. Rodents are obligate nasal breathers, so the significance of these changes remains unclear when extrapolating to humans and other species. However, the olfactory epithelium was also a target of the cytotoxic action of 1,2,3-trichloropropane after gavage application to rats for 17 weeks.
Further research is necessary to determine if 1,2,3-trichloropropane exposure is selecting the sex of offspring in favour of females (Gulati et al., 1990; Chapin et al., 1997).
The main environmental target compartments of 1,2,3-trichloropropane are air and water, where this compound is only slowly transformed by abiotic reactions and poorly transformed aerobically by microorganisms. Therefore, conditions not accelerating biotic or abiotic removal might lead to the presence of detectable concentrations of this compound in these two compartments. However, a reliable quantification of 1,2,3-trichloropropane currently released from all industrial sources is impossible with the data available.
The experimentally determined bioconcentration data as well as the measured log Kow value indicate a low bioaccumulation potential for 1,2,3-trichloropropane.
A sample risk characterization according to the European Commission (EC, 1996) with respect to the aquatic environment might be performed by calculating the ratio between a local PEC (based on measured data) and a corresponding PNEC.
Although a quantification of 1,2,3-trichloropropane released into the environment is not possible with data currently available, recent monitoring data from northern Europe might be regarded as representative for an industrialized region. The highest reported value for Dutch surface water of the rivers Rhein, Meuse, and Westerscheldt and the Northern Delta Area is 2.2 µg/litre, which can be employed as the local PEC. A corresponding PNEC for surface water might be estimated from the lowest EC50 value from tests carried out in closed systems to minimize 1,2,3-trichloropropane losses. The 48-h EC50 value for Daphnia magna immobilization (20 mg/litre) was used to derive the PNEC, together with an uncertainty factor of 1000 (EC, 1996): PNEC = 20 mg/litre ÷ 1000 = 0.02 mg/litre. Although a lower value was obtained for Ceriodaphnia cf. dubia (48-h EC50 of 4.1 mg/litre), this test was based on nominal concentrations only and therefore was not used in the risk characterization.
Using the highest recently measured concentration of 1,2,3-trichloropropane in surface water, the hazard quotient (PEC/PNEC) becomes 2.2 µg/litre ÷ 20 µg/litre = 0.11. Because this is less than 1, no further information, testing, or risk reduction measures are required.
1,2,3-Trichloropropane will preferably partition into air (about 85%) and water (about 11%) and only to a limited extent into soil (the remaining 4%) (Mackay et al., 1993).
The determined soil sorption coefficients indicate a low potential for soil sorption. Therefore, under conditions not promoting abiotic or biotic transformation, leaching of 1,2,3-trichloropropane into groundwater can take place.
As only inhibition tests on soil-bound microbial activity are available, it does not appear appropriate to employ these limited data for a risk characterization. Valid studies on the toxicity of 1,2,3-trichloropropane to terrestrial invertebrates or vertebrates are not available.
The acute toxicity of 1,2,3-trichloropropane was tested using a variety of species from different trophic levels. However, the number of data is still limited. Because 1,2,3-trichloropropane is volatile, only acute toxicity data based on measured concentrations were used in the risk characterization. However, it is likely that the true concentrations in the other tests were much lower than the nominal concentrations, so that the reported NOEC and LC50/EC50 values are too high (i.e., the toxicity of 1,2,3-trichloropropane is greater than that estimated from the acute toxicity tests). If the true PNEC is lower based on measured concentrations, then the calculated PEC:PNEC ratio may be 1 or higher, indicating that the risk is higher than that determined in section 11.2.1. Until further testing is carried out with additional species using closed test systems, the results of the risk characterization outlined here should be interpreted with caution.
For the terrestrial compartment, the available toxicity studies measuring the inhibition of soil-bound microbial activity by 1,2,3-trichloropropane cannot be regarded as sufficient to support a quantitative risk characterization.
There is inadequate evidence in humans for the carcinogenicity of 1,2,3-trichloropropane. There is sufficient evidence in experimental animals for the carcinogenicity of 1,2,3-trichloropropane. IARC (1995) classifies 1,2,3-trichloropropane as a Group 2A carcinogen: probably carcinogenic to humans.
ABC (1986a) Acute toxicity of trichloropropane to Daphnia magna. St. Louis, MO, Monsanto Agricultural Products Company, Analytical Bio Chemistry Laboratory, pp. 1–8 (NTIS/OTS 0000815).
ABC (1986b) Acute toxicity of trichloropropane to bluegill sunfish (Lepomis macrochirus). St. Louis, MO, Monsanto Agricultural Products Company, Analytical Bio Chemistry Laboratory, pp. 1–13 (NTIS/OTS 0000815).
ABC (1986c) Acute toxicity of trichloropropane to rainbow trout (Salmo gairdneri). St. Louis, MO, Monsanto Agricultural Products Company, Analytical Bio Chemistry Laboratory, pp. 1–9 (NTIS/OTS 0000815).
Albanese V, Milano JC, Vernet JL (1987) Etude de l’évaporation de quelques hydrocarbures halogénés de faible masse moléculaire dissous à l’état de traces dans l’eau. Environmental Technology Letters, 8:657–668.
Albert J (1982) Acute toxicity studies with 1,2,3-trichloropropane. Prepared by Stillmeadow, Inc. for Shell Development Co., Houston, TX; submitted to TSCA on 19 August 1987 (NTIS/PB 86-870001650).
Anderson D, Yu TW, McGregor DB (1998) Comet assay responses as indicators of carcinogen exposure. Mutagenesis, 13(6):539–555.
Anderson TA, Beauchamp JJ, Walton BT (1991) Fate of volatile and semivolatile organic chemicals in soils: Abiotic versus biotic losses. Journal of Environmental Quality, 20(2):420–424.
Atkinson R (1987) Structure–activity relationship for the estimation of rate constants for the gas-phase reactions of OH radicals with organic compounds. International Journal of Chemical Kinetics, 19:799–828.
Baier JH, Lykins BW, Fronk CA, Kramer SJ (1987) Using reverse osmosis to remove agricultural chemicals from groundwater. Journal of the American Water Works Association, 79:55–60.
Bauer U (1981a) Belastung des Menschen durch Schadstoffe in der Umwelt- Untersuchungen über leicht flüchtige organische Halogenverbindungen in Wasser, Luft, Lebensmitteln und im menschlichen Gewebe II. Mitteilung: Untersuchungsmethodik leicht flüchtiger Organohalogenverbindungen. Zentralblatt für Bakteriologie, Parasitenkunde, Infektionskrankheiten und Hygiene, Abteilung 1, Originale, Reihe B, 174:39–56.
Bauer U (1981b) Belastung des Menschen durch Schadstoffe in der Umwelt- Untersuchungen über leicht flüchtige organische Halogenverbindungen in Wasser, Luft, Lebensmitteln und im menschlichen Gewebe III. Mitteilung: Untersuchungsergebnisse. Zentralblatt für Bakteriologie, Parasitenkunde, Infektionskrankheiten und Hygiene, Abteilung 1, Originale, Reihe B, 174:200–237.
Belyaeva NN, Tsulaya VR, Marshak TL, Brodskii VY (1974) Effect of 1,2,3-trichloropropane on the ploidy of rat liver cells. Byulleten Eksperimental’noi Biologii i Meditsiny, 78(12):1414–1416.
Benigni R, Pino A (1998) Profiles of chemically-induced tumors in rodents: Quantitative relationship. Mutation Research, 421(1):93–107.
Bio/dynamics Inc. (1979) A 13 week inhalation toxicity study of 1,2,3-trichloropropane in the rat. St. Louis, MO, Monsanto Chemical Company, pp. 1–76 (NTIS/OTS 0000815, 26 November 1979).
Bio/dynamics Inc. (1980) A one-generation reproduction-fertility study of 1,2,3-trichloropropane in rats. St. Louis, MO, Monsanto Company, pp. 1–39 (NTIS/OTS 0000815, 7 April 1980).
Bio/dynamics Inc. (1983) A thirteen week inhalation toxicity study of 1,2,3-trichloropropane in the rat. St. Louis, MO, Monsanto Company, pp. 1–54 (NTIS/OTS 0000815, 17 August 1983).
Bio/dynamics Inc. (1985a) Primary dermal irritation study in rabbits (4- and 24-hour exposure). St. Louis, MO, Monsanto Company, pp. 1–31 (NTIS/OTS 0000815; 12 June 1985).
Bio/dynamics Inc. (1985b) Eye irritation study in rabbits. St. Louis, MO, Monsanto Company, pp. 1–12 (NTIS/OTS 0000815; 12 June 1985).
Bio/dynamics Inc. (1985c) A closed-patch repeated insult dermal sensitization study in guinea pigs (Buehler method). St. Louis, MO, Monsanto Company, pp. 1–30 (NTIS/OTS 0000815).
Blum DJW, Speece RE (1991) A database of chemical toxicity to environmental bacteria and its use in interspecies comparisons and correlations. Journal of the Water Pollution Control Federation, 63(3):198–207.
Blume H (1990) Handbuch des Bodenschutzes. Landsberg/Lech, Ecomed, pp. 580–581.
Bonvalot Y, Gagnon C, Benjamin M, Germain A, Dann T (2000) Sampling program for residential wood combustion: Winter of 1998–99 study report. Montreal, Quebec, Public Works and Government Services Canada, pp. iii–80.
Bosma T, Janssen DB (1998) Conversion of chlorinated propanes by Methylosinus trichosporium OB3b expressing soluble methane monooxygenase. Applied Microbiology and Biotechnology, 50:105–112.
Bosma T, Kruizinga E, DeBruin EJ, Poelarends GJ, Janssen DB (1999) Utilization of trihalogenated propanes by Agrobacterium radiobacter AD1 through heterologous expression of the haloalkane dehalogenase from Rhodococcus sp. strain m15-3. Applied Environmental Microbiology, 65(10):4575–4581.
Bouhamra WS, Buhamra SS, Thomson MS (1997) Determination of volatile organic compounds in indoor and ambient air of residences in Kuwait. Environment International, 23:197–204.
Brock M, Carroll J (1985) 1984 industrial hygiene monitoring for 1,2,3-trichloropropane, 2,3-dichloropropene, 1,3-dichloropropene, and isopropyl chloride at Glycerine I, Dow Chemical USA. Freeport, TX, Dow Chemical USA, pp. 1–10 (NTIS/PB86-870002354).
BUA (1993) 1,2,3-Trichloropropane. German Chemical Society (GDCh) Advisory Committee on Existing Chemicals of Environmental Relevance (BUA). Stuttgart, Hirzel, pp. 1–97 (BUA Report 154).
Chapin R, Gulati D, Mounce R, Russell S, Poonacha K (1997) 1,2,3-Trichloropropane. Environmental Health Perspectives, 105(Suppl. 1):361–362.
City of Shafter (2000) Consumer confidence report for water quality. Shafter, CA, City of Shafter, pp. 1–6.
Clark D (1977) Acute toxicity, skin and eye irritancy and skin sensitizing potential of 1,2,3-trichloropropane. Houston, TX, Shell Oil Company, pp. 1–16 (NTIS/PB 86-870001655, 1 September 1977).
Dean BJ, Brooks TM (1979) In vitro mutation studies with 1,2,3-trichloropropane. Houston, TX, Shell Oil Company, pp. 1–28 (NTIS/PB 86-870001651, 1 June 1979).
Dilling WL (1977) Interphase transfer processes. II. Evaporation rates of chloromethanes, ethanes, ethylenes, propanes and propylenes from dilute aqueous solutions. Comparisons with theoretical predictions. Environmental Science & Technology, 11:405–409.
Doherty AT, Ellard S, Parry EM, Parry JM (1996) An investigation into the activation and deactivation of chlorinated hydrocarbons to genotoxins in metabolically competent human cells. Mutagenesis, 11(3):247–274.
Douglas GR, Nestmann ER, Lee E, Marshall R, Heddle JA (1985) How well do in vitro tests predict in vivo genotoxicity. Environmental Mutagenesis, 7(Suppl. 3):31.
Drew RT, Patel JM, Lin FN (1978) Changes in serum enzymes in rats after inhalation of organic solvents singly and in combination. Toxicology and Applied Pharmacology, 45:809–819.
EC (1996) Technical guidance document in support of the Commission Directive 93/EEC on risk assessment for new notified substances and the Commission Regulation (EC)1488/94 on risk assessment for existing substances. Brussels, European Commission.
Ellerstein SM, Bertozzi ER (1982) Polymers containing sulfur. In: Kirk-Othmer encyclopedia of chemical technology, 3rd ed. New York, NY, John Wiley & Sons, pp. 793–831.
Ellington JJ, Stancil FE, Payne WD (1987) Measurement of hydrolysis rate constants for evaluation of hazardous waste land disposal. Vol. 1. Data on 32 chemicals. Athens, GA, US Environmental Protection Agency, Environmental Research Laboratory, pp. 1–25 (US EPA/600/3-86/043; PB 87-140349/GAR).
Frischenschlager H, Mittermayr C, Peck M, Rosenberg E, Grasserbauer M (1997) The potential of gas chromatography with microwave-induced plasma atomic emission detection (GC-MIP-AED) as a complementary analytical technique in environmental screening analysis of aqueous samples. Fresenius’ Journal of Analytical Chemistry, 359:213–221.
Geiger DL, Brooke LT, Call DJ (1990) Acute toxicities of organic chemicals to fathead minnows (Pimephales promelas), Vol. 5. Superior, WI, University of Wisconsin-Superior, Center for Lake Superior Environmental Studies, pp. 2–22, 40, 49–50, 70, 80.
Gelover S, Bandala ER, Leal-Ascencio T, Perez S, Martinez E (2000) GC-MS determination of volatile organic compounds in drinking water supplies in Mexico. Environmental Toxicology, 15(2):131–139.
Griesemer RA, Eustis SL (1994) Gender differences in animal bioassays for carcinogenicity. Journal of Occupational and Environmental Medicine, 36(8):855–859.
Gulati D, Mounce RC, Russel S, Poonacha KB, Chapin RE (1990) 1,2,3-Trichloropropane reproduction and fertility assessment in Swiss CD-1 mice when administered via gavage — final report. Prepared by Environmental Health Research and Testing, Inc., Lexington, KY, for National Toxicology Program, Research Triangle Park, NC, 226 pp. (NTIS/PB91-129676; NTP-90-209).
Hardin BD, Bond GP, Sikov MR, Andrew FD, Beliles RP, Niemeier RN (1981) Testing of selected workplace chemicals for teratogenic potential. Scandinavian Journal of Work, Environment & Health, 7:66–75.
Hauck R, Hegemann W (2000) Anaerobic degradation of 1,2-dichloropropane in batch and continuous culture. Water Science and Technology, 41(12):7–13.
Haworth S, Lawlor T, Mortelmans K, Speck W, Zeiger E (1983) Salmonella mutagenicity test results for 250 chemicals. Environmental Mutagenesis, Supplement 1:3–142.
Hazleton Laboratories America Inc. (1983a) 120-day toxicity gavage study of 1,2,3-trichloropropane in Fischer 344 rats. Prepared by Hazleton Laboratories America Inc., Vienna, VA, for Shell Oil Company, Houston, TX (PB-86-870001644; submitted to NTIS/TSCA on 19 August 1987).
Hazleton Laboratories America Inc. (1983b) 120 day gavage toxicity study in B6C3F1 mice: 1,2,3-trichloropropane (final report) with attachments. Prepared by Hazleton Laboratories America Inc., Vienna, VA, for Shell Oil Company, Houston, TX (PB-86-870001577; submitted to NTIS/TSCA).
Health Canada (2000) Review of contaminant occurrences in drinking water treatment chemicals. Ottawa, Ontario, Health Canada, Health Protection Branch, pp. 1–10.
Hermens J, Canton H, Janssen P, De Jong R (1984) Quantitative structure–activity relationships and toxicity studies of mixtures of chemicals with anaesthetic potency: Acute lethal and sublethal toxicity to Daphnia magna. Aquatic Toxicology, 5:143–154.
Holme JA, Soderlund EJ, Brunborg G, Lag M, Nelson SD, Dybing E (1991) DNA damage and cell death induced by 1,2-dibromo-3-chloropropane (DBCP) and structural analogs in monolayer culture of rat hepatocytes: 3-Aminobenzamide inhibits the toxicity of DBCP. Cell Biology and Toxicology, 7:413–432.
Hutchinson TC, Hellebust JA, Tam D, Mackay D, Mascarenhas RA, Shiu WY (1980) The correlation of the toxicity to algae of hydrocarbons and halogenated hydrocarbons with their physical-chemical properties. In: Afghan BK, Mackay D, eds. Hydrocarbons and halogenated hydrocarbons in the aquatic environment. New York, NY, Plenum Press, pp. 577–586.
IARC (1995) 1,2,3-Trichloropropane. Lyon, International Agency for Research on Cancer, pp. 223–244 (IARC Monographs on the Evaluation of the Carcinogenic Risk of Chemicals to Humans, Volume 63).
IPCS (1999) International Chemical Safety Card — 1,2,3-Trichloropropane. Geneva, World Health Organization, International Programme on Chemical Safety (ICSC 0683).
Irwin RD, Haseman JK, Eustis SL (1995) 1,2,3-Trichloropropane: A multisite carcinogen in rats and mice. Fundamental and Applied Toxicology, 25:241–252.
Ito N, La DK, Holt S, Craft TR, Sills RC (1996) Analysis of ras mutations in forestomach tumors from B6C3F1 mice exposed to 1,2,3-trichloropropane. Proceedings of the American Association for Cancer Research, 37:137.
Johannsen F, Levinskas G, Rusch G, Terrill J, Schroeder R (1988) Evaluation of the subchronic and reproductive effects of a series of chlorinated propanes in the rat. I. Toxicity of 1,2,3-trichloropropane. Journal of Toxicology and Environmental Health, 25:299–315.
Johnson R (1968) Polymers containing sulfur. Polysulfides. In: Kirk-Othmer encyclopedia of chemical technology, 2nd ed. New York, NY, John Wiley & Sons, pp. 253–254, 259–262.
Kaua’i Department of Water (2001) Puhi water system 2001, water quality report. Kaua’i, Kaua’i Department of Water, pp. 1–6.
Kawata K, Tanabe A, Saito S, Sakai M, Yasuhara A (1997) Screening of volatile organic compounds in river sediment. Bulletin of Environmental Contamination and Toxicology, 58(6):893–900.
Kier L (1982) Ames/Salmonella mutagenicity assays of 1,2,3-trichloropropane, 1,2,2,3-tetrachloropropane and 1,1,2,2,3-pentachloropropane. Prepared by Environmental Health Laboratory, Monsanto Company, St. Louis, MO, for US Environmental Protection Agency, pp. 1–19 (NTIS/OTS 0000815, 19 August 1982).
Könemann H (1981) Quantitative structure–activity relationships in fish toxicity studies. 1. Relationship for 50 industrial pollutants. Toxicology, 19:209–221.
Kooijman SALM (1981) Parametric analyses of mortality rates in bioassays. Water Research, 15:107–119.
La DK, Swenberg JA (1996) DNA adducts: biological markers of exposure and potential applications to risk assessment. Mutation Research, 365:129–146.
La DK, Lilly PD, Anderegg RJ, Swenberg JA (1995) DNA adduct formation in B6C3F1 mice and Fischer-344 rats exposed to 1,2,3-trichloropropane. Carcinogenesis, 16:1419–1424.
La DK, Schoonhoven R, Ito N, Swenberg JA (1996) The effects of exposure route on DNA adduct formation and cellular proliferation by 1,2,3-trichloropropane. Toxicology and Applied Pharmacology, 140(1):108–114.
Låg M, Soderlund E, Omichinski J, Brunborg G, Holme J, Dahl J, Nelson S, Dybing E (1991) Effect of bromine and chlorine positioning in the induction of renal and testicular toxicity by halogenated propanes. Chemical Research in Toxicology, 4:528–534.
Låg M, Omichinski J, Dybing E, Nelson D, Soderlund E (1994) Mutagenic activity of halogenated propanes and propenes: Effect of bromine and chlorine positioning. Chemico-Biological Interactions, 93(1):73–84.
Lagas P, Verdam B, Loch JPG (1989) Threat to groundwater quality by pesticides in the Netherlands. In: Groundwater management: quantity and quality (Proceedings of the Benidorm Symposium, October 1989). Wallingford, Oxfordshire, Centre for Ecology and Hydrology, International Association of Hydrological Sciences, pp. 171–180 (IAHS Publication No. 188).
Lewis R (1992) 1,2,3-Trichloropropane. In: Lewis R, ed. Sax’s dangerous properties of industrial materials. New York, NY, Van Nostrand Reinhold, pp. 241–264.
Lide D (1995) CRC handbook of chemistry and physics, 76th ed. Boca Raton, FL, CRC Press.
Liska I, Barceló D, Grasserbauer M (1996) Strategy for the screening of organic pollutants in a river basin — an overview of the Nitra river monitoring programme. Trends in Analytical Chemistry, 15(8):326–334.
LWA (1989) Rheingütebericht NRW ’88. Anhang. Nordrhein-Westfalen, Düsseldorf, Landesamt für Wasser und Abfall, pp. I–VII.
Mackay D, Shiu W, Ma K (1993) Illustrated handbook of physical-chemical properties and environmental fate for organic chemicals. Vol. 3. Volatile organic chemicals. Boca Raton, FL, Lewis Publishers.
Mahmood NA, Overstreet D, Burka LT (1991) Comparative disposition and metabolism of 1,2,3-trichloropropane in rats and mice. Drug Metabolism and Disposition, 19:411–418.
MAK (1993) 1,2,3-Trichloropropane. In: Greim H, ed. Occupational toxicants: critical data evaluation for MAK values and classification of carcinogens. Vol. 9. Deutsche Forschungsgemeinschaft (DFG); Commission for the Investigation of Health Hazards of Chemical Compounds in the Work Area (MAK). Weinheim, Wiley-VCH, pp. 171–192.
Merrick B, Smallwood CL, Meier JR, McKean DL, Kaylor WH, Condie LW (1987) Chemical reactivity, cytotoxicity, and mutagenicity of chloropropanones. Toxicology and Applied Pharmacology, 91:46–54.
Merrick B, Robinson M, Condie L (1991) Cardiopathic effect of 1,2,3-trichloropropane after subacute and subchronic exposure in rats. Journal of Applied Toxicology, 11:179–187.
Miermans CJH, van der Velde LE, Frintrop PCM (2000) Analysis of volatile organic compounds, using the purge and trap injector coupled to a gas chromatograph/ion-trap mass spectrometer: Review of the results in Dutch surface water of the Rhine, Meuse, Northern Delta Area and Westerscheldt, over the period 1992–1997. Chemosphere, 40(1):39–48.
Milano JC, Guibourg A, Vernet JL (1988) Evolution non biologique dans l’eau de composés organohalogénés à trois et quatre atomes de carbone: Hydrolyse et photolyse. Water Research, 22(12):1553–1562.
Miller R, Quast JF, Momany-Pfruender JJ (1986a) 1,2,3-Trichloropropane: 2-week vapor inhalation study to determine the no-adverse-effect level in rats and mice. Midland, MI, The Dow Chemical Company, pp. 1–25 (NTIS/PB 86-870002265, 13 May 1986).
Miller R, Quast JF, Gushow TS (1986b) 1,2,3-Trichloropropane: 2-week vapor inhalation study in rats and mice. Midland, MI, The Dow Chemical Company, pp. 1–43 (NTIS/PB 86-870002260; 27 May 1986).
Mirsalis J, Tyson K, Beck J, Loh F, Steinmetz K, Contreras C, Austere L, Martin S, Spalding J (1983) Induction of unscheduled DNA synthesis (UDS) in hepatocytes following in vitro and in vivo treatment. Environmental Mutagenesis, 5:482.
MITI (1992) Biodegradation and bioaccumulation: Data of existing chemicals based on the CSCL Japan. Tokyo, Ministry of International Trade & Industry, pp. 2–19.
Moorman WJ, Ahlers HW, Chapin RE, Daston GP, Foster PMD, Kavlock RJ, Morawetz JS, Schnorr TM, Schrader SM (2000) Prioritization of NTP reproductive toxicants for field studies. Reproductive Toxicology, 14(4):293–301.
NTP (1993) Toxicology and carcinogenesis studies of 1,2,3-trichloropropane (CAS No.
NTP (2000) 9th report on carcinogens. US Department of Health and Human Services, Public Health Service, National Toxicology Program, available at website http://ehp.niehs.nih.gov.
Oki DS, Giambelluca TW (1989) Groundwater contamination by nematicides: Influence of recharge timing under pineapple crop. Water Research Bulletin, 25:285–294.
Pankow JF, Luo W, Isabelle LM, Bender DA, Baker RJ (1998) Determination of a wide range of volatile organic compounds in ambient air using multisorbent adsorption/thermal desorption and gas chromatography/mass spectrometry. Analytical Chemistry, 70(24):5213–5221.
Parry JM, Parry EM, Bourner R, Doherty A, Ellard S, O’Donovan J, Hoebee B, de Stoppelaar JM, Mohn GR, Önfelt A, Renglin A, Schultz N, Söderpalm-Berndes C, Jensen K, Kirsch-Volders M, Elhajouji A, Van Hummelen P, Degrassi F, Antoccia A, Cimini D, Izzo M, Tanzarella C, Adler I-D, Kliesch U, Schriever-Schwemmer G, Gasser P, Crebelli R, Carere A, Andreoli C, Benigni R, Leopardi P, Marcon F, Zinjo Z, Natarajan AT, Boei JJWA, Kappas A, Voutsinas G, Zarani FE, Patrinelli A, Pachierotti F, Tiveron C, Hess P (1996) The detection and evaluation of aneugenic chemicals. Mutation Research, 353(1–2):11–46.
Peijnenburg W, Eriksson L, De Groot A, Sjöström M, Verboom H (1998) The kinetics of reductive dehalogenation of a set of halogenated aliphatic hydrocarbons in anaerobic slurries. Environmental Science & Pollution Research, 5(1):12–16.
Peng C, Batterman S (2000) Performance evaluation of a sorbent tube sampling method using short path thermal desorption for volatile organic compounds. Journal of Environmental Monitoring, 2(4):313–324.
Rassaerts H, Witzel D (1975) Chlorkohlenwasserstoffe, aliphatische. In: Bartholomé E, Biekert E, Hellmann H, Ley H, Weigert WM, eds. Ullmanns Encyklopädie der technischen Chemie. Weinheim/
Bergstrasse, Verlag Chemie, pp. 404–498.
Ratpan F, Plaumann H (1988) Mutagenicity of halogenated propanes and their methylated derivatives. Environmental and Molecular Mutagenesis, 12:253–259.
Rose RM, Warne MStJ, Lim RP (1998) Quantitative structure–activity relationships and volume fraction analysis for nonpolar narcotic chemicals to Australian cladoceran Ceriodaphnia cf. dubia. Archives of Environmental Contamination and Toxicology, 34:248–252.
Ruelle P (2000) The n-octanol and n-hexane/water partition coefficient of environmentally relevant chemicals predicted from the mobile order and disorder (MOD) thermodynamics. Chemosphere, 40:457–512.
Saito-Suzuki R, Teramoto S, Shirasu Y (1982) Dominant lethal studies in rats with 1,2-dibromo-3-chloropropane and its structurally related compounds. Mutation Research, 101:321–327.
Sawin VL, Hass BS (1982) Assay of 1,2,3-trichloropropane for gene mutation in mouse lymphoma cells. Houston, TX, Shell Development Company, pp. 1–26 (NTIS/PB 86-870001645, 14 January 1982).
Silverman L, Schulte HF, First MW (1946) Further studies on sensory response to certain industrial solvent vapors. Journal of Industrial Hygiene and Toxicology, 28:262–266.
Solvay (2001a) The toxicity of 1,2,3-trichloropropane to the alga Selenastrum capricornutum. Prepared for Health, Environment & Safety Department by Environmental Assessment Group Weesp, Solvay Pharmaceuticals, 24 pp. (Analysis Chemical Supplies and Environmental Development Study No. A.SOL.S.023, 27 August 2001).
Solvay (2001b) Internal memorandum: Evaporation of 1,2,3-trichloropropane during a 48-hour acute toxicity test at 20°C. Weesp, Solvay Pharmaceuticals, pp. 1–3 (Reference: AdG/AED/im/01.80, 19 July 2001).
Solvay (2002) The acute toxicity of 1,2,3-trichloropropane to the water flea (Daphnia magna). Prepared by Environmental Assessment Group Weesp, Solvay Pharmaceuticals, 17 pp. (Study No. A.SOL.S.030, September 2002).
Stolzenberg SJ, Hine CH (1980) Mutagenicity of 2- and 3-carbon halogenated compounds in the Salmonella/mammalian microsome test. Environmental Mutagenesis, 2:59–66.
Swann R, Laskowski DA, McCall PJ, Vander Kuy K, Dishburger HJ (1983) A rapid method for the estimation of the environmental parameters octanol/water partition coefficient, soil sorption constant, water to air ratio, and water solubility. Residue Reviews, 85:17–28.
Swenberg JA, La KD, Scheller NA, Wu K (1995) Dose–response relationships for carcinogens. Toxicology Letters, 82/83:751–756.
Tafazoli M, Kirsch-Volders M (1996) In vitro mutagenicity and genotoxicity study of 1,2-dichloroethylene, 1,1,2-trichloroethane, 1,3-dichloropropane, 1,2,3-trichloropropane and 1,1,3-trichloropropene, using the micronucleus test and the alkaline single cell gel electrophoresis technique (comet assay) in human lymphocytes. Mutation Research, 371:185–202.
Tafazoli M, Baeten A, Geerlings P, Kirsch-Volders M (1998) In vitro mutagenicity and genotoxicity study of a number of short-chain chlorinated hydrocarbons using the micronucleus test and the alkaline single cell gel electrophoresis technique (comet assay) in human lymphocytes: A structure–activity relationship (QSAR) analysis of the genotoxic and cytotoxic potential. Mutagenesis, 13(2):115–126.
Tancrède M, Yanagisawa Y (1990) An analytical method to determine Henry’s law constant for selected volatile organic compounds at concentrations and temperatures corresponding to tap water use. Journal of the Air & Waste Management Association, 40:1658–1663.
Tancrède M, Yanagisawa Y, Wilson R (1992) Volatilization of volatile organic compounds from showers — I. Analytical method and quantitative assessment. Atmospheric Environment, 26A:1103–1111.
Thomas R (1990) Volatilization from water. In: Lyman WJ, Reehl WF, Rosenblatt DH, eds. Handbook of chemical property estimation methods. Environmental behavior of organic compounds. New York, NY, McGraw-Hill Book Company, pp. 15.1–15.34.
US EPA (1999) Toxics Release Inventory, at US Environmental Protection Agency website http://www.epa.gov/tri/tri99/pdr/index.htm.
Vannelli T, Logan M, Arciero D, Hooper A (1990) Degradation of halogenated aliphatic compounds by the ammonia-oxidizing bacterium Nitrosomonas europaea. Applied Environmental Microbiology, 56(4):1169–1171.
Verschueren K (1996) Handbook of environmental data on organic chemicals, 3rd ed. New York, NY, John Wiley & Sons.
Villeneuve D, Chu I, Secours VE, Cote MG, Plaa GL, Valli VE (1985) Results of a 90-day toxicity study on 1,2,3- and 1,1,2-trichloropropane administered via the drinking water. The Science of the Total Environment, 47:421–426.
Volp RF, Sipes IG, Falcoz C, Carter DE, Gross JF (1984) Disposition of 1,2,3-trichloropropane in the Fischer 344 rat: Conventional and physiological pharmacokinetics. Toxicology and Applied Pharmacology, 75:8–17.
von der Hude W, Scheutwinkel M, Gramlich U, Fißler B, Basler A (1987) Genotoxicity of three-carbon compounds evaluated in the SCE test in vitro. Environmental Mutagenesis, 9(4):401–410.
von Düszeln J, Lahl U, Bätjer K, Cetinkaya M, Stachel B, Thiemann W, Gabel B, Kozicki R, Podbielski A (1982) Flüchtige Halogenkohlenwasserstoffe in Luft, Wasser und Nahrungsmitteln in der Bundesrepublik Deutschland. Analyse und Belastungsabschätzung für den Menschen. Deutsche Lebensmittel-Rundschau, 78:352–356.
Walton BT, Anderson TA, Hendricks MS, Talmage SS (1989) Physicochemical properties as predictors of organic chemical effects on soil microbial respiration. Environmental Toxicology and Chemistry, 8:53–63.
Walton BT, Hendricks MS, Anderson TA, Griest WH, Merriweather R, Beauchamp JJ, Francis CW (1992) Soil sorption of volatile and semivolatile organic compounds in a mixture. Journal of Environmental Quality, 21:552–558.
Weber GL, Sipes IG (1990) Covalent interactions of 1,2,3-trichloropropane with hepatic macromolecules: Studies in the male F-344 rat. Toxicology and Applied Pharmacology, 104:395–402.
Weber G, Sipes I (1991) Rat hepatic DNA damage induced by 1,2,3-trichloropropane. In: Witmer C, Snyder R, Jollow D, Kalf G, Kocsis J, Sipes I, eds. Biological reactive intermediates. IV. Molecular and cellular effects and their impact on human health. New York, NY, Plenum Press, pp. 853–855.
Weber GL, Sipes IG (1992) In vitro metabolism and bioactivation of 1,2,3-trichloropropane. Toxicology and Applied Pharmacology, 113(1):152–158.
Williams GM, Mori H, McQueen CA (1989) Structure–activity relationship in the rat hepatocyte DNA-repair test for 300 chemicals. Mutation Research, 221:263–286.
Yamamoto K, Fukushima M, Kakutani N, Kuroda K (1997) Volatile organic compounds in urban rivers and their estuaries in Osaka, Japan. Environmental Pollution, 95:135–143.
Yamamoto N, Okayasu H, Hiraiwa T, Murayama S, Maeda T, Morita M, Suzuki K (1998) Continuous determination of volatile organic compounds in the atmosphere by an automated gas chromatographic system. Journal of Chromatography A, 819(1–2):177–186.
Yoshikawa S, Shibata Y, Miyajima S, Kurosawa Y (1998) Screening of volatile organic compounds in river and sea water in Kawasaki city. Mizu-Kankyo-Gakkaishi, 21(12):879–883.
Zebarth BJ, Szeto SY, Hii B, Liebscher H, Grove G (1998) Groundwater contamination by chlorinated hydrocarbon impurities present in soil fumigant formulations. Water Quality Research Journal of Canada, 33(1):31–50.
BUA (1993) 1,2,3-Trichloropropane. German Chemical Society (GDCh) Advisory Committee on Existing Chemicals of Environmental Relevance (BUA). Stuttgart, Hirzel, pp. 1–97 (BUA Report 154).
For the BUA review process, the company that is in charge of writing the report (usually the largest producer in Germany) prepares a draft report using literature from an extensive literature search as well as internal company studies. This draft is subject to a peer review in several readings of a working group consisting of representatives from government agencies, the scientific community, and industry.
The English translation of this report was published in 1997.
MAK (1993) 1,2,3-Trichloropropane. In: Greim H, ed. Occupational toxicants: critical data evaluation for MAK values and classification of carcinogens. Vol. 9. Deutsche Forschungsgemeinschaft (DFG); Commission for the Investigation of Health Hazards of Chemical Compounds in the Work Area (MAK). Weinheim, Wiley-VCH, pp. 171–192.
The scientific documentations of the German Commission for the Investigation of Health Hazards of Chemical Compounds in the Work Area (MAK) are based on critical evaluations of the available toxicological and occupational medical data from extensive literature searches and from well documented industrial data. The evaluation documents involve a critical examination of the quality of the database, indicating inadequacy or doubtful validity of data and identification of data gaps. This critical evaluation and the classification of substances are the result of an extensive discussion process by the members of the Commission, proceeding from draft documentation prepared by members of the Commission, by ad hoc experts, or by the Scientific Secretariat of the Commission. Scientific expertise is guaranteed by the members of the Commission, which consists of experts from the scientific community, industry, and employer associations.
The draft CICAD on 1,2,3-trichloropropane was sent for review to IPCS national Contact Points and Participating Institutions, as well as to identified experts. Comments were received from:
J.E. Andrews, National Center for Environmental Assessment, US Environmental Protection Agency, Research Triangle Park, NC, USA
R. Benson, Drinking Water Program, US Environmental Protection Agency, Denver, CO, USA
R.S. Chhabra, National Institute of Environmental Health Sciences, Research Triangle Park, NC, USA
M. Cikrt, National Institute of Public Health, Prague, Czech Republic
C. Cowles, Health and Safety Executive, Bootle, Merseyside, United Kingdom
I. Desi, Department of Public Health, University of Szeged, Szeged, Hungary
G. Dura, Fodor Jozsef National Public Health Centre, Budapest, Hungary
L. Fishbein, Private consultant, Fairfax, VA, USA
E. Frantik, National Institute of Public Health, Prague, Czech Republic
L.R. Harris, Epichlorohydrin Task Group, The Society of the Plastics Industry, Washington, DC, USA
R.F. Hertel, Federal Institute for Health Protection of Consumers and Veterinary Medicine, Berlin, Germany
C. Hiremath, National Center for Environmental Assessment, US Environmental Protection Agency, Research Triangle Park, NC, USA
A. Hirose, National Institute of Health Sciences, Tokyo, Japan
P. Howe, Centre for Ecology and Hydrology, Monks Wood, United Kingdom
S.H. Lee, Catholic University, Seoul, Korea
D.W. Lynch, National Institute for Occupational Safety and Health, Cincinnati, OH, USA
M. Mercier, Scientific Institute of Public Health, Brussels, Belgium
E. Murono, National Institute for Occupational Safety and Health, Cincinnati, OH, USA
K. Pekari, Finnish Institute of Occupational Health, Helsinki, Finland
E. Savigny, Health and Safety Executive, Bootle, Merseyside, United Kingdom
F. Simeonova, Center of Hygiene, Medical Ecology and Nutrition, Sofia, Bulgaria
E. Soderlund, Norwegian Public Health Institute, Oslo, Norway
J. Stauber, Centre for Advanced Analytical Chemistry, Bangor, Australia
J.M.H. Temmink, Wageningen Agricultural University, Wageningen, Netherlands
K. Ziegler-Skylakakis, European Commission, Luxembourg
Monks Wood, United Kingdom
16–19 September 2002
Members
Dr R. Benson, US Environmental Protection Agency, Region VIII, Denver, CO, USA
Mr R. Cary, Health and Safety Executive, Bootle, Merseyside, United Kingdom
Dr R. Chhabra, National Institute of Environmental Health Sciences, Research Triangle Park, NC, USA
Dr S. Chou, Agency for Toxic Substances and Disease Registry (ATSDR), Atlanta, GA, USA
Dr S. Czerczak, Nofer Institute of Occupational Medicine, Lodz, Poland
Dr S. Dobson, Centre for Ecology and Hydrology, Monks Wood, Abbots Ripton, Huntingdon, Cambridgeshire, United Kingdom
Dr G. Dura, National Institute of Environmental Health, Jozsef Fodor Public Health Centre, Budapest, Hungary
Dr L. Fishbein, Fairfax, VA, USA
Dr H. Gibb, National Center for Environmental Assessment, US Environmental Protection Agency, Washington, DC, USA
Dr Y. Hayashi, Division of Chem-Bio Informatics, National Institute of Health Sciences, Ministry of Health, Labour and Welfare, Tokyo, Japan
Dr R.F. Hertel, Federal Institute for Health Protection of Consumers and Veterinary Medicine, Berlin, Germany
Dr A. Hirose, Division of Risk Assessment, National Institute of Health Sciences, Tokyo, Japan
Mr P. Howe, Centre for Ecology and Hydrology, Monks Wood, Abbots Ripton, Huntingdon, Cambridgeshire, United Kingdom
Prof. J. Jeyaratnam, Colombo, Sri Lanka
Dr J. Kielhorn, Fraunhofer Institute of Toxicology and Aerosol Research, Hanover, Germany
Prof. Y.-X. Liang, School of Public Health, Fudan University, Shanghai Medical College, Shanghai, People’s Republic of China
Dr R. Liteplo, Existing Substances Division, Environmental Contaminants Bureau, Health Canada, Ottawa, Ontario, Canada
Ms M.E. Meek, Existing Substances Division, Safe Environments Programme, Health Canada, Ottawa, Ontario, Canada
Mr F.K. Muchiri, Directorate of Occupational Health and Safety Services, Nairobi, Kenya
Dr O. Sabzevari, Department of Toxicology & Pharmacology, Faculty of Pharmacy, Tehran University of Medical Sciences, Tehran, Iran
Dr J. Sekizawa, Division of Chem-Bio Informatics, National Institute of Health Sciences, Tokyo, Japan
Dr F.P. Simeonova, Sofia, Bulgaria
Dr J. Stauber, CSIRO Energy Technology, Centre for Advanced Analytical Chemistry, Bangor, Australia
Dr M.H. Sweeney, Document Development Branch, Education and Information Division, National Institute for Occupational Safety and Health, Cincinnati, OH, USA
Dr K. Ziegler-Skylakakis, European Commission, DG Employment & Social Affairs, Luxembourg
Resource Persons
Dr C. Cowles, Health and Safety Executive, Industrial Chemicals Unit HD, Bootle, Merseyside, United Kingdom
Dr C. Elliott-Minty, Health and Safety Executive, Industrial Chemicals Unit HD, Bootle, Merseyside, United Kingdom
Dr K. Fuller, Health and Safety Executive, Industrial Chemicals Unit HD, Bootle, Merseyside, United Kingdom
Observers
Mr A.G. Berends, Solvay S.A., Brussels, Belgium; European Chemical Industry Council / European Centre for Ecotoxicology and Toxicology of Chemicals (CEFIC/ECETOC)
Mr W. Gulledge, American Chemistry Council, Arlington, VA, USA
Mr C. Newsome, Dow Chemical Company Limited, West Drayton, Middlesex, United Kingdom; European Chemical Industry Council / European Centre for Ecotoxicology and Toxicology of Chemicals (CEFIC/ECETOC)
Mr M.A. Pemberton, Wilmslow, United Kingdom; European Chemical Industry Council / European Centre for Ecotoxicology and Toxicology of Chemicals (CEFIC/ECETOC)
Mr W. Stott, Dow Chemical Company, Midland, MI, USA; European Chemical Industry Council / European Centre for Ecotoxicology and Toxicology of Chemicals (CEFIC/ECETOC)
Mr J.M. Waechter, Jr, The Dow Chemical Company, Midland, MI, USA; European Chemical Industry Council / European Centre for Ecotoxicology and Toxicology of Chemicals (CEFIC/ECETOC)
Secretariat
Dr A. Aitio, International Programme on Chemical Safety, World Health Organization, Geneva, Switzerland
Mr T. Ehara, International Programme on Chemical Safety, World Health Organization, Geneva, Switzerland
Mr H. Malcolm, Centre for Ecology and Hydrology, Monks Wood, Abbots Ripton, Huntingdon, Cambridgeshire, United Kingdom
Ms C. Vickers, International Programme on Chemical Safety, World Health Organization, Geneva, Switzerland
|
ACPC |
N-acetyl-S-(3-chloro-2-hydroxypropyl)-L-cysteine |
|
CAS |
Chemical Abstracts Service |
|
CHO |
Chinese hamster ovary |
|
CICAD |
Concise International Chemical Assessment Document |
|
CPC |
S-(3-chloro-2-hydroxypropyl)-L-cysteine |
|
DCA |
1,3-dichloroacetone |
|
DNA |
deoxyribonucleic acid |
|
EC50 |
median effective concentration |
|
ECD |
electron capture detection |
|
ELCD |
electrolytic conductivity detection |
|
EPA |
Environmental Protection Agency (US) |
|
FID |
flame ionization detection |
|
GC |
gas chromatography |
|
GMA |
2-(S-glutathionyl)malonic acid |
|
GSH |
glutathione |
|
IC50 |
median inhibitory concentration |
|
ICSC |
International Chemical Safety Card |
|
Koc |
sorption coefficient |
|
LC50 |
median lethal concentration |
|
LD50 |
median lethal dose |
|
LOAEL |
lowest-observed-adverse-effect level |
|
MS |
mass spectrometry |
|
NADH |
nicotinamide adenine dinucleotide, reduced form |
|
NADPH |
nicotinamide adenine dinucleotide phosphate, reduced form |
|
NOAEL |
no-observed-adverse-effect level |
|
NOEC |
no-observed-effect concentration |
|
NTP |
National Toxicology Program (US) |
|
OECD |
Organisation for Economic Co-operation and Development |
|
PEC |
predicted environmental concentration |
|
PID |
photoionization detection |
|
PNEC |
predicted no-effect concentration |
|
RNA |
ribonucleic acid |
|
SI |
International System of Units (Système international d’unités) |
|
SPI |
The Society of the Plastics Industry |
|
WHO |
World Health Organization |
1,2,3-TRICHLOROPROPANE ICSC:0683
Le présent CICAD consacré au 1,2,3-trichloropropane a été préparé par l’Institut Fraunhofer de toxicologie et de recherche sur les aérosols (Secteur recherche pharmaceutique et inhalation clinique), de Hanovre (Allemagne). Il s’inspire de rapports établis par le Comité consultatif de la Société allemande de chimie sur les substances chimiques d’importance écologique (BUA, 1993) et par la Commission allemande pour l’étude des risques écologiques imputables aux composés chimiques sur le lieu de travail (MAK, 1993). Il a été procédé à un dépouillement bibliographique exhaustif des bases de données correspondantes, en novembre 2001 pour les effets sanitaires et en septembre 2002 pour les effets environnementaux, afin de rechercher toute référence intéressante à des publications postérieures aux rapports en question. Des informations sur la préparation et l’examen par des pairs des sources documentaires utilisées sont données à l’appendice 1. L’appendice 2 fournit des renseignements sur l’examen par des pairs du présent CICAD. Ce CICAD a été approuvé en tant qu’évaluation internationale lors d’une réunion du Comité d’évaluation finale qui s’est tenue à Monks Wood (Royaume-Uni) du 16 au 19 septembre 2002. La liste des participants à cette réunion figure à l’appendice 3. La fiche internationale sur la sécurité chimique du 1,2,3-trichloropropane (ICSC 0683), établie par le Programme international sur la sécurité chimique (IPCS, 1999), est également reproduite dans le présent document.
Le 1,2,3-trichloropropane (No CAS
Le compartiment de l’environnement où aboutit principalement le 1,2,3-trichloropropane libéré dans le milieu est l’air atmosphérique (à hauteur d’environ 85 %), suivi par l’eau (environ 11 %). Les concentrations relevées dans l’air ambiant aux Etats-Unis et en Europe vont de non décelable à 0,4 μg/m3. Dans les cours d’eau européens, la concentration va de non décelable à 2,2 μg/litre.
Le 1,2,3-trichloropropane ne subit guère de transformation sous l’action de processus abiotiques (par exemple, attaque par les radicaux hydroxyle engendrés par voie photochimique) et il pourrait donc persister pendant de longues durées. En revanche, il peut être éliminé des systèmes aquatiques par évaporation et passer du sol aux eaux souterraines par lessivage, en raison de la valeur peu élevée de son coefficient de sorption aux particules du sol (Koc). Il est difficilement biodégradable et ne se transforme que lentement, en aérobiose ou en anaérobiose, sous l’action des bactéries. Les données relatives à la bioconcentration de cet alcane chloré montrent qu’il ne manifeste probablement pas de tendance à la bioaccumulation.
Les principales voies d’exposition au 1,2,3-trichloropropane sont l’inhalation d’air contaminé ou l’ingestion d’eau de boisson polluée.Il peut être absorbé par voie transcutanée, mais dans une proportion plus limitée.
L’expérimentation animale montre que le 1,2,3-trichloropropane est rapidement résorbé dans les voies digestives, puis tout aussi rapidement métabolisé et excrété. C’est ainsi qu’une dose administrée par voie orale est excrétée en 60 h dans les urines (à hauteur de 50 à 65 %), dans les matières fécales (à hauteur de 15 à 20 %) et dans l’air inhalé sous forme de dioxyde de carbone (à hauteur de 20 %). La métabolisation du composé se révèle plus rapide chez la souris que chez le rat.
Après administration par voie orale à des rats, le principal métabolite urinaire (correspondant à 40 % de la radioactivité urinaire) identifié au bout de 6 h est un conjugué avec l’acide mercapturique, la N-acétyl-S-(3-chloro-2-hydroxypropyl)-L-cystéine. Dans les urines de 24 heures, on trouve un second métabolite, qui est un conjugué avec la cystéine, à savoir la S-(3-chloro-2-hydroxypropyl)-L-cystéine. Soixante heures après administration par voie orale de [14C]-1,2,3-trichloropropane, la radioactivité due au carbone-14 est encore présente dans les organes cibles (comme le foie, le rein et l’estomac antérieur [portion aglandulaire]). Après injection intraveineuse de 1,2,3-trichloropropane à des rats, l’un des principaux métabolites retrouvés dans la bile s’est révélé être l’acide 2-(S-glutathionyl)malonique. Chez la souris, le spectre métabolique est plus complexe.
L’isolement de ces métabolites incite à penser que la biotransformation du 1,2,3-trichloropropane comporte une conjugaison avec le glutathion (GSH) et une oxydation. On a suggéré une voie hépatique comportant une oxygénation, catalysée par les oxydases à fonction mixte, au niveau d’un carbone terminal pour donner une chlorhydrine, après quoi d’autres réactions conduiraient à la formation des métabolites observés. Une deuxième voie métabolique intrahépatique pourrait faire intervenir la formation, catalysée par la GSH-transférase, de conjugués avec le glutathion, qui subiraient ensuite encore une biotransformation intrahépatique ou passeraient dans la bile ou le plasma.
La toxicité aiguë du 1,2,3-trichloropropane est modérée, avec une valeur de la DL50 qui va de 150 à 500 mg par kg de poids corporel chez le rat. Par voie cutanée, la toxicité est moindre, la seule étude dont on dispose chez le rat faisant état d’une DL50 de 836 mg/kg de poids corporel. Chez le lapin, la valeur de la DL50 par voie cutanée est comprise entre 384 et 2457 mg/kg de poids corporel. En ce qui concerne l’exposition dans l’air, on a obtenu une CL50 à 4 h d’environ 3000 mg/m3 chez le rat et la souris. L’effet toxique le plus marqué consiste en une irritation de la muqueuse oculaire et nasale ainsi qu’en lésions hépatiques et rénales.
Le 1,2,3-trichloropropane a un effet irritant sur la peau et les muqueuses. Les divers tests effectués sur des cobayes montre que le 1,2,3-trichloropropane n’a aucun effet sensibilisateur ou du moins un effet minime.
Le principal effet constaté chez des rats F344 et des souris B6C3F1 après des expositions répétées par la voie respiratoire à des doses de 1,2,3-trichloropropane allant jusqu’à 780 mg par m3 pendant 9 jours, a été une dégénérescence et une atteinte inflammatoire microscopiques au niveau de la muqueuse olfactive nasale. Chez la souris, le poids des testicules était sensiblement réduit à la dose la plus élevée, mais sans que l’on puisse observer d’anomalies histologiques. On n’a pas observé d’autres anomalies si ce n’est une modification du poids du foie dans le groupe exposé à la dose la plus forte. Lors d’une expérience comportant l’exposition répétée de rats et de souris à des doses allant jusqu’à 61 mg/m3, la dose sans effet nocif général observable (NOAEL) a été trouvée égale à 6 mg/m3 pour le rat et à 18 mg/m3 pour la souris, l’effet en question étant une modification de l’épithélium olfactif décelable par examen histologique.
Au cours d’une autre étude pendant laquelle des rats CD ont été exposés durant 13 semaines à des doses allant jusqu’à 300 mg/m3, on a observé des effets toxiques au niveau des voies respiratoires, des poumons et du foie. Une étude longitudinale comportant l’exposition à des doses allant jusqu’à 9,2 mg/m3 a révélé des signes d’irritation des muqueuses (augmentation de la lacrimation), même à la dose la plus faible (3,1 mg/m3). Les seuls effets généraux observés consistaient en anomalies hématologiques et en une augmentation du poids du poumon et des ovaires, mais sans anomalies histologiques correspondantes.
Après avoir administré du 1,2,3-trichloropropane par voie orale à des rats F344 pendant des périodes de durée moyenne, les principales lésions imputables à la toxicité de ce composé ont été observées au niveau du foie, du rein et des fosses nasales, les mâles présentant une plus grande sensibililité à cet égard. Les anomalies hématologiques relevées à des doses journalières supérieures où égales à 16 mg par kg de poids corporel ont été interprétées comme traduisant une anémie non régénérative, peut-être liée à une dépression de l’érythropoïèse. Une étude de 17 semaines pendant lesquelles des rats ont reçu le composé par gavage a permis d’obtenir, pour la dose la plus faible produisant un effet observable (LOAEL), la valeur de 8 mg/kg p.c. par jour dans le cas des mâles et de 16 mg/kg p.c. par jour dans le cas des femelles (augmentation du poids absolu du foie). Chez la souris B6C3F1, les principaux organes cibles de l’action toxique sont le poumon, le foie et l’estomac antérieur. La souris supporte des doses plus élevées que le rat, la LOAEL (hyperplasie de l’épithélium bronchiolaire ou hyperplasie et hyperkératose de l’estomac antérieur) étant de 63 mg/kg p.c. par jour pour la souris femelle et de 125 mg/kg p.c. par jour pour la souris mâle.L’effet cardiotoxique évoqué par un groupe de travail n’est pas confirmé par ces études, qui comportaient une durée d’administration du produit comparable, voire plus longue. Lorsque le gavage consistait dans l’administration d’un bolus, il a déterminé des effets plus graves que l’administration en continu dans l’eau de boisson à dose comparable. En administrant du 1,2,3-trichloropropane à des rats Sprague-Dawley dans leur eau de boisson pendant 13 semaines, on a obtenu pour la LOAEL (effet retenu : augmentation du poids relatif du foie et du rein) la valeur de 17,6 mg/kg p.c. par jour pour les femelles et de 113 mg/kg p.c. par jour pour les mâles.
Divers tests de génotoxicité in vitro (par exemple, des mutations géniques dans des cellules bactériennes ou mammaliennes, des conversions géniques chez Saccharomyces cerevisiae, des échanges entre chromatides soeurs, des aberrations chromosomiques et la formation de micronoyaux) indiquent à l’évidence que le 1,2,3-trichloropropane peut être génotoxique en présence d’un système d’activation métabolique. Les données isolées relatives à une action génotoxique directe du composé paraissent en revanche discutables. In vivo, l’élution en milieu alcalin permet de déceler des ruptures monocaténaires de l’ADN, mais une épreuve de létalité dominante n’a pas permis de mettre la génotoxicité du composé en évidence.
Le principal adduit avec l’ADN est le S-[1-(hydroxyméthyl)-2-(N7-guanyl)éthyl]glutathion, mais d’autres adduits avec l’ADN ont été mis en évidence dans des lésions prénéoplasiques et néoplasiques affectant les organes cibles.
Les résultats d’une étude portant sur deux générations de souris Swiss montrent que le 1,2,3-trichloropropane réduit la fécondité et perturbe la fonction de reproduction à la dose de 120 mg/kg p.c. par jour (administrée par gavage), tant dans la génération parentale que dans la progéniture, avec une toxicité générale qui reste cependant peu marquée. Un essai comportant des accouplements croisés a permis, semble-t-il, de montrer que la toxicité pour l’appareil reproducteur était plus marquée chez les femelles que chez les mâles. Chez toutes les femelles de la génération F1 exposées au 1,2,3-trichloropropane, la durée moyenne du cycle oestral était sensiblement augmentée, la dose la plus faible produisant cet effet étant égale à 30 mg/kg p.c. par jour.
Le 1,2,3-trichloropropane est un cancérogène à localisations multiples provoquant des lésions prénéoplasiques correspondantes chez le rat et la souris après administration prolongée par gavage. Les principales cibles de son action cancérogène sont l’estomac antérieur ainsi que la muqueuse buccale chez les rats des deux sexes, la glande mammaire chez les femelles, le rein et le pancréas chez les mâles ainsi que la glande préputiale et la glande clitoridienne qui sont des organes homologues chez le mâle et la femelle. Chez la souris, la réaction au traitement par le 1,2,3-trichloropropane se traduit par des néoplasmes de l’estomac antérieur, du foie et de la glande de Harder. On a également signalé des tumeurs de nature inhabituelle, comme des carcinomes de la glande de Zymbal et des polypes adénomateux ou des adénocarcinomes de l’intestin chez le rat ainsi que des cancers de l’uterus chez la souris. Compte tenu de l’incidence très élevée des néoplasmes de l’estomac antérieur dans les groupes de rats (33 à 66 %) et de souris (près de 100 %) soumis à des doses peu élevées, cette activité cancérogène aurait pu être décelée même à plus faible dose et la LOAEL correspondant à une augmentation signficative de l’incidence tumorale est largement inférieure à 3 mg/kg p.c. par jour chez le rat et à 6 mg/kg p.c. par jour chez la souris.
Le 1,2,3-trichloropropane est cancérogène pour le rat et la souris et entraîne l’apparition de tumeurs très variées chez les animaux des deux sexes. Les données relatives à son mode d’action, et notamment les études sur son métabolisme, sa génotoxicité et la formation d’adduits avec l’ADN incitent à penser que le mécanisme de cancérisation comporte l’interaction directe entre un métabolite actif et le matériel génétique. L’exposition au 1,2,3-trichloropropane doit donc être évitée.
On a procédé à une caractérisation du risque selon les directives de la Commission européenne qui portait sur l’environnement aquatique et qui a consisté à calculer le rapport de la concentration environnementale locale prédite (PEC), basée sur des mesures, à la concentration environnementale correspondante prédite sans effet (PNEC).
La PNEC pour les eaux de surface a été déterminée à partir de la valeur la plus faible de la CE50 tirée de tests en systèmes fermés afin de réduire au minimum les pertes de 1,2,3-trichloropropane. Pour obtenir la PNEC, on a utilisé la valeur de la CE50 à 48 h pour l’immobilisation de Daphnia magna (20 mg/litre) et on a appliqué au dénominateur un facteur d’incertitude de 1000 : PNEC = 20 mg/litre ÷ 1000 = 0,02 mg/litre. Bien qu’une valeur plus faible ait été obtenue pour Ceriodaphnia cf. dubia (CE50 à 48 h de 4,1 mg/litre), le test correspondant n’était basé que sur les concentrations nominales et n’a donc pas été utilisé pour la caractérisation du risque.
En prenant comme PEC la concentration la plus récemment déterminée du 1,2,3-trichloropropane dans les eaux de surface (soit 2,2 μg/litre), on trouve pour le quotient de dangerositι (PEC/PNEC) la valeur suivante : 2,2 μg/litre χ 20 μg/litre = 0,1. Comme ce chiffre est infιrieur à 1, il n’est pas nécessaire d’obtenir d’autres données, de pratiquer d’autres tests ou de prendre des mesures de réduction du risque.
On n’a pas trouvé de données relatives aux effets toxiques de ce composé chimique sur les invertébrés terrestres ou les plantes supérieures. En ce qui concerne l’environnement terrestre, les études toxicologiques dont on dispose pour mesurer l’inhibition de l’activité microbienne terricole par le 1,2,3-trichloropropane ne peuvent pas être considérées comme suffisantes en vue d’une caractérisation quantitative du risque.
El presente CICAD sobre el 1,2,3-tricloropropano, preparado por el Instituto Fraunhofer de Toxicología y de Investigación sobre los Aerosoles (Alemania), se basa en informes compilados por el Comité Consultivo Alemán sobre las Sustancias Químicas Importantes para el Medio Ambiente (BUA, 1993) y la Comisión Alemana de Investigación de los Peligros para la Salud de las Sustancias Químicas en el Entorno de Trabajo (MAK, 1993). En noviembre de 2001 se realizó una búsqueda bibliográfica amplia de bases de datos pertinentes para los efectos en la salud y en septiembre de 2002 para los efectos en el medio ambiente, a fin de localizar cualquier referencia de interés publicada después de las incorporadas a estos informes. La información relativa a la preparación y el examen colegiado de los documentos originales figura en el Apéndice 1. La información sobre el examen colegiado de este CICAD se presenta en el Apéndice 2. Este CICAD se aprobó en una reunión de la Junta de Evaluación Final celebrada en Monks Wood (Reino Unido) del 16 al 19 de septiembre de 2002. La lista de participantes en la Junta de Evaluación Final figura en el Apéndice 3. También se reproduce en este documento la Ficha internacional de seguridad química (ICSC 0683) para el 1,2,3-tricloropropano, preparada por el Programa Internacional de Seguridad de las Sustancias Químicas (IPCS, 1999).
El 1,2,3-tricloropropano (CAS Nº
El compartimento destinatario más importante del 1,2,3-tricloropropano en el medio ambiente es el aire (alrededor del 85%), seguido del agua (en torno al 11%). En los Estados Unidos y Europa se han notificado concentraciones de 1,2,3-tricloropropano en el aire ambiente que oscilan entre valores no detectados y 0,4 µg/m3. En los ríos de Europa, las concentraciones varían entre valores no detectados y 2,2 µg/l.
El 1,2,3-tricloropropano liberado en el medio ambiente apenas se degrada por procesos abióticos (por ejemplo, transformación mediante radicales hidroxilo producidos por vía fotoquímica), por lo que podría mantenerse presente durante períodos prolongados. Sin embargo, se puede eliminar de los sistemas acuáticos por evaporación y podría sufrir un proceso de lixiviación del suelo hacia el agua freática, debido a los bajos coeficientes de sorción en el suelo (Koc) notificados para este compuesto. No se degrada con facilidad y las bacterias lo transforman sólo lentamente en condiciones aerobias y anaerobias. Los datos disponibles sobre este alcano clorado indican que no es probable su bioacumulación.
Las vías principales de exposición al 1,2,3-tricloropropano son la inhalación de aire contaminado y el consumo de agua de bebida contaminada. Se puede absorber por vía cutánea en una medida más limitada.
En estudios con animales, el 1,2,3-tricloropropano se absorbe con rapidez del tracto gastrointestinal, se metaboliza y se excreta. Tras la administración oral, se excretó en un plazo de 60 horas por vía urinaria (50-65%), fecal (15-20%) y respiratoria como anhídrido carbónico (20%). Parece que los ratones metabolizan el 1,2,3-tricloropropano con mayor rapidez que las ratas.
Tras la administración oral a ratas, el principal metabolito urinario (40% de radioactividad urinaria) identificado después de seis horas fue un conjugado del ácido mercaptúrico, la N-acetil-S-(3-cloro-2-hidroxipropil)-L-cisteína. En la orina de 24 horas se detectó un segundo metabolito, un conjugado de la cisteína (S-(3-cloro-2-hidroxipropil)-L-cisteína). Incluso 60 horas después de la administración oral de [14C]1,2,3-tricloropropano se observó actividad del 14C en los órganos destinatarios (es decir, el hígado, el riñón y la región cardíaca del estómago). Tras la administración intravenosa de 1,2,3-tricloropropano a ratas, un metabolito importante en la bilis fue el ácido 2-(S-glutationil)malónico. En ratones, el espectro de los metabolitos urinarios es más complejo.
El aislamiento de los metabolitos antes mencionados parece indicar que en la biotransformación participan tanto la conjugación con el glutatión como la oxidación. Una vía propuesta en el hígado es la oxigenación de un carbono terminal del 1,2,3-tricloropropano catalizada por una oxidasa de función mixta para producir una clorohidrina, seguida de otras reacciones que dan lugar a la formación de los metabolitos observados. Una segunda vía en el hígado puede ser la formación de conjugados del glutatión por acción de la glutatión transferasa, que luego sufren una nueva biotransformación en el hígado o se excretan en la bilis o el plasma.
El 1,2,3-tricloropropano tiene una toxicidad aguda moderada, con valores de la DL50 en ratas comprendidos entre 150 y 500 mg/kg de peso corporal. La toxicidad cutánea es más baja, con valores notificados de la DL50 de 836 mg/kg de peso corporal al día de un estudio único con ratas y valores comprendidos entre 384 y 2457 mg/kg de peso corporal en conejos. En ratas y ratones se obtuvo un valor de la CL50 a las cuatro horas de unos 3000 mg de 1,2,3-tricloropropano/m3. El efecto tóxico más llamativo es la irritación de la mucosa ocular y nasal y lesiones hepáticas y renales.
El 1,2,3-tricloropropano irrita la piel y las membranas mucosas. En diversas pruebas con cobayas, se observó un efecto sensibilizador nulo o sólo muy ligero.
El efecto primordial de la exposición repetida de ratas F344 y ratones B6C3F1 por inhalación a concentraciones de hasta 780 mg de 1,2,3-tricloropropano/m3 durante nueve días fue un cambio degenerativo e inflamatorio microscópico en la mucosa olfativa nasal. En ratones, se observó una disminución importante del peso de los testículos con la dosis más alta, pero sin cambios histopatológicos asociados. No aparecieron otros fenómenos, salvo cambios en el peso del hígado en el grupo con la dosificación más alta. En un experimento con dosis repetidas de hasta 61 mg/m3, la concentración general sin efectos adversos observados (NOAEL) para los cambios detectables por examen histopatológico en el epitelio olfativo fue de 6 mg/m3 en las ratas y de 18 mg/m3 en los ratones.
En otro estudio en el que se expusieron ratas CD a concentraciones de hasta 300 mg/m3 durante 13 semanas, se observaron efectos tóxicos en las vías respiratorias superiores, los pulmones y el hígado. En un estudio complementario con dosis de hasta 9,2 mg/m3 se notificaron signos de irritación de las membranas mucosas (aumento de la descarga lacrimal), incluso con la concentración mínima de 3,1 mg/m3. Los únicos efectos sistémicos detectados fueron cambios en los parámetros hematológicos y un aumento de peso de los pulmones y los ovarios, sin las observaciones microscópicas correspondientes.
Tras una exposición oral de duración media de ratas F344 al 1,2,3-tricloropropano, las principales lesiones tóxicas se produjeron en el hígado, el riñón y los turbinatos nasales, mostrando mayor sensibilidad los machos. Los cambios hematológicos con 16 mg/kg de peso corporal al día y dosis superiores se interpretaron como anemia no regenerativa, posiblemente asociada con una depresión de la eritropoyesis. La concentración más baja con efectos adversos observados (LOAEL) para un tratamiento de 17 semanas mediante sonda fue de 8 mg/kg de peso corporal al día en ratas macho y de 16 mg/kg de peso corporal al día en ratas hembra (aumento del peso absoluto del hígado). En ratones B6C3F1, los principales órganos destinatarios de la acción tóxica fueron los pulmones, el hígado y la región cardíaca del estómago. Los ratones toleraron dosis más altas que las ratas, con valores de la LOAEL (hiperplasia del epitelio bronquiolar e hiperplasia e hiperqueratosis de la región cardíaca del estómago) de 63 mg/kg de peso corporal al día en ratas hembra y de 125 mg/kg de peso corporal al día en ratones macho. Un efecto cardiotóxico examinado por un grupo de trabajo no se confirmó en estos estudios, que abarcaron períodos de tratamiento comparables o más prolongados. La administración de un bolo mediante sonda produjo efectos más graves que la aplicación continua en el agua de bebida con una dosificación comparable. Cuando se administró a ratas Sprague-Dawley 1,2,3-tricloropropano con el agua de bebida durante 13 semanas, los valores de la LOAEL para el aumento relativo del peso del hígado y el riñón fueron de 17,6 mg/kg de peso corporal al día para las ratas hembra y de 113 mg/kg de peso corporal al día para las ratas macho.
En diversas pruebas de genotoxicidad in vitro (por ejemplo mutaciones de genes en bacterias y células de mamíferos, conversiones de genes en Saccharomyces cerevisiae, inducción del intercambio de cromátidas hermanas, aberraciones cromosómicas y micronúcleos) se puso de manifiesto un potencial genotóxico del 1,2,3-tricloropropano en presencia de sistemas de activación metabólica. Parecen cuestionables los datos aislados sobre una acción genotóxica directa del 1,2,3-tricloropropano. In vivo se detectaron roturas de cadenas sencillas de ADN mediante elución alcalina, mientras que no hubo acción genotóxica en una prueba de dominancia letal.
En lesiones preneoplásicas y neoplásicas de órganos destinatarios se identificó S-[1-(hidroximetil)-2-(N7-guanil)etil]glutatión, principal aducto de ADN, y otros aductos de ADN.
Los resultados de un estudio de dos generaciones con ratones suizos pusieron de manifiesto que el 1,2,3-tricloropropano alteraba la fecundidad y la reproducción con una dosis de 120 mg/kg de peso corporal al día (administrado mediante sonda) tanto en la generación parenteral como en las crías en presencia de una toxicidad sistémica sólo ligera. Un ensayo de apareamiento cruzado parecía indicar una mayor toxicidad para el sistema reproductivo de las hembras que de los machos. La duración media del ciclo estral fue notablemente más alta para todas las hembras de la generación F1 expuestas al 1,2,3-tricloropropano, siendo la dosis más baja de 30 mg/kg de peso corporal al día.
El 1,2,3-tricloropropano es carcinogénico para diversos órganos, con las correspondientes lesiones preneoplásicas tanto en ratas como en ratones tras su administración prolongada mediante sonda. Los puntos principales de su acción carcinogénica son la región cardíaca del estómago y la mucosa oral en ratas de ambos sexos, la glándula mamaria en las ratas hembra y el páncreas y el riñón en las ratas macho, así como las glándulas prepucial y clitorídea, como órganos homólogos en ratas macho y hembra, respectivamente. La reacción en los ratones fue de neoplasmas en la región cardíaca del estómago, el hígado y la glándula de Harder. Se notificaron tipos poco frecuentes de tumores, como carcinomas de la glándula de Zymbal y pólipos adenomatosos o adenocarcinomas del intestino en ratas y neoplasmas uterinos en ratones. Teniendo en cuenta la incidencia muy elevada de neoplasmas en la región cardíaca del estómago en grupos de ratas (33-66%) y ratones (casi el 100%) tratados con una dosis baja, esta actividad carcinogénica se podría haber detectado incluso con dosis más bajas, y la LOAEL para la incidencia de tumores significativamente elevada estará muy por encima de 3 mg/kg de peso corporal al día en ratas y 6 mg/kg de peso corporal al día en ratones.
El 1,2,3-tricloropropano es carcinogénico en ratas y ratones, induciendo una gran variedad de tumores en ambos sexos. Los datos sobre el mecanismo de acción, incluidos los estudios sobre el metabolismo, la genotoxicidad y la determinación de la formación de aductos del ADN, parecen indicar que en el mecanismo de inducción de tumores interviene la interacción directa de un metabolito activo con material genético. Por consiguiente, se debería evitar la exposición al 1,2,3-tricloropropano.
Se realizaron pruebas de toxicidad aguda del 1,2,3-tricloropropano utilizando diversas especies acuáticas de distintos niveles tróficos.
Se realizó una caracterización del riesgo de muestra conforme a la Comisión Europea con respecto al medio ambiente acuático, calculando la razón entre la concentración local prevista en el medio ambiente (PEC), basada en los datos medidos, y la correspondiente concentración prevista sin efectos (PNEC).
Se estimó una PNEC para el agua superficial a partir del valor más bajo de la CE50 obtenido en pruebas realizadas en sistemas cerrados para reducir al mínimo las pérdidas de 1,2,3-tricloropropano. Se utilizó la CL50 a las 48 horas para la inmovilización de Daphnia magna (20 mg/l) a fin de obtener la PNEC, junto con un factor de incertidumbre de 1000: PNEC = 20 mg/l ÷ 1000 = 0,02 mg/l. Aunque se obtuvo un valor más bajo para Ceriodaphnia cf. dubia (CE50 a los 48 horas de 4,1 mg/l), esta prueba sólo se basó en concentraciones nominales y, por consiguiente, no se utilizó en la caracterización del riesgo.
Utilizando la concentración más alta de 1,2,3-tricloropropano medida recientemente en el agua superficial (2,2 µg/l) como PEC, el cociente de peligro (PEC/PNEC) se convierte en 2,2 µg/l ÷ 20 µg/l = 0,1. Debido a que este valor es inferior a la unidad, no hay necesidad de ulterior información, pruebas o medidas de reducción del riesgo.
No se identificaron datos sobre los efectos tóxicos de este producto químico en invertebrados terrestres o en plantas superiores. Para el compartimento terrestre, los estudios de toxicidad disponibles, en los que se midió la inhibición por el 1,2,3-tricloropropano de la actividad microbiana ligada al suelo, no se pueden considerar suficientes para respaldar una caracterización cuantitativa del riesgo.
ENDNOTES
|
International Programme on Chemical Safety (1994) Assessing human health risks of chemicals: derivation of guidance values for health-based exposure limits. Geneva, World Health Organization (Environmental Health Criteria 170) (also available at http://www.who.int/pcs/). |
|
|
In keeping with WHO policy, which is to provide measurements in SI units, all concentrations of gaseous chemicals in air will be given in SI units in the CICAD series. Where the original study or source document has provided concentrations in SI units, these will be cited here. Where the original study or source document has provided concentrations in volumetric units, conversions will be done using the conversion factors given here, assuming a temperature of 20 °C and a pressure of 101.3 kPa. Conversions are to no more than two significant digits. |
|
|
The source document does not include derivation of a guidance value for 1,2,3-trichlororopane, a genotoxic carcinogen. In CICADs, a tolerable intake or tolerable concentration may be developed (independent of the source document) for genotoxic chemicals, to indicate the risk level at a particular dose; in this instance, however, this was not done. |
See Also:
Toxicological Abbreviations
Trichloropropane, 1,2,3- (IARC Summary & Evaluation, Volume 63, 1995)