
INTERNATIONAL PROGRAMME ON CHEMICAL SAFETY
ENVIRONMENTAL HEALTH CRITERIA 165
INORGANIC LEAD
This report contains the collective views of an international group of
experts and does not necessarily represent the decisions or the stated
policy of the United Nations Environment Programme, the International
Labour Organisation, or the World Health Organization.
First draft prepared at the National Institute of Health Sciences,
Tokyo, Japan, and the Institute of Terrestrial Ecology, Monk's Wood,
United Kingdom
Published under the joint sponsorship of the United Nations
Environment Programme, the International Labour Organisation, and the
World Health Organization
World Health Organization
Geneva, 1995
The International Programme on Chemical Safety (IPCS) is a joint
venture of the United Nations Environment Programme, the International
Labour Organisation, and the World Health Organization. The main
objective of the IPCS is to carry out and disseminate evaluations of
the effects of chemicals on human health and the quality of the
environment. Supporting activities include the development of
epidemiological, experimental laboratory, and risk-assessment methods
that could produce internationally comparable results, and the
development of manpower in the field of toxicology. Other activities
carried out by the IPCS include the development of know-how for coping
with chemical accidents, coordination of laboratory testing and
epidemiological studies, and promotion of research on the mechanisms
of the biological action of chemicals.
WHO Library Cataloguing in Publication Data
Inorganic lead.
(Environmental health criteria ; 165)
1.Lead - adverse effects 2.Environmental exposure
3.Guidelines I.Series
ISBN 92 4 157165 9 (NLM Classification: QV 292)
ISSN 0250-863X
The World Health Organization welcomes requests for permission to
reproduce or translate its publications, in part or in full.
Applications and enquiries should be addressed to the Office of
Publications, World Health Organization, Geneva, Switzerland, which
will be glad to provide the latest information on any changes made to
the text, plans for new editions, and reprints and translations
already available.
(c) World Health Organization 1995
Publications of the World Health Organization enjoy copyright
protection in accordance with the provisions of Protocol 2 of the
Universal Copyright Convention. All rights reserved. The designations
employed and the presentation of the material in this publication do
not imply the expression of any opinion whatsoever on the part of the
Secretariat of the World Health Organization concerning the legal
status of any country, territory, city or area or of its authorities,
or concerning the delimitation of its frontiers or boundaries. The
mention of specific companies or of certain manufacturers' products
does not imply that they are endorsed or recommended by the World
Health Organization in preference to others of a similar nature that
are not mentioned. Errors and omissions excepted, the names of
proprietary products are distinguished by initial capital letters.
CONTENTS
ENVIROMENTAL HEALTH CRITERIA FOR INORGANIC LEAD
PREAMBLE
PREFACE
1. SUMMARY
1.1. Identity, physical and chemical properties, and analytical
methods
1.2. Sources of human exposure
1.3. Environmental transport, distribution and transformation
1.4. Environmental levels and human exposure
1.5. Kinetics and metabolism in laboratory animals and humans
1.6. Effects on laboratory animals and in vitro systems
1.7. Effects on humans
1.8. Evaluation of human health risks
2. IDENTITY, PHYSICAL AND CHEMICAL PROPERTIES, AND ANALYTICAL
METHODS
2.1. Physical and chemical properties of lead and its compounds
2.2. Analytical procedures
2.2.1. Sampling procedures
2.2.1.1 Sampling of environmental media
2.2.1.2 Sampling of biological materials
2.2.2. Analytical methods for lead
2.2.2.1 Analysis of lead in environmental
samples
2.2.2.2 Analysis of lead in biological materials
2.2.2.3 Analytical procedures for biomarkers of
lead exposure and effect
2.3. Conversion factors
3. SOURCES OF HUMAN AND ENVIRONMENTAL EXPOSURE
3.1. Natural occurrence
3.1.1. Rocks and soils
3.1.2. Sediments
3.1.3. Water
3.1.4. Air
3.1.5. Plants
3.1.6. Environmental contamination from natural sources
3.2. Anthropogenic sources
3.2.1. Lead mining
3.2.2. Smelting and refining
3.2.3. Environmental pollution from production of lead
3.3. Consumption and uses of lead and its compounds
3.4. Sources of environmental exposure
4. ENVIRONMENTAL TRANSPORT, DISTRIBUTION AND TRANSFORMATION
4.1. Transport and distribution between media
4.1.1. Atmospheric deposition
4.1.2. Transport to water and soil
4.1.3. Transport to biota
4.1.3.1 Aquatic organisms
4.1.3.2 Terrestrial organisms
4.2. Environmental transformation
4.2.1. Abiotic transformation
4.2.2. Biotransformation
5. ENVIRONMENTAL LEVELS AND HUMAN EXPOSURE
5.1. Inhalation route of exposure
5.1.1. Ambient air
5.1.1.1 Emissions from motor vehicles
5.1.1.2 Stationary sources
5.1.2. Indoor air
5.1.3. Air in the working environment
5.1.4. Smoking of tobacco
5.2. Exposure by ingestion
5.2.1. Water
5.2.2. Food and alcoholic beverages
5.2.2.1 Food
5.2.2.2 Total intake from food
5.2.2.3 Alcoholic beverages
5.2.3. Dust and surface soils
5.2.3.1 Dust
5.2.3.2 Soil
5.2.3.3 Migration of lead from food containers
5.3. Miscellaneous exposure
5.3.1. Cosmetics and medicines
5.4. General population exposure
5.5. Blood lead concentrations of various populations
5.5.1. Adult populations
5.5.2. Children
5.5.3. Remote populations
5.6. Occupational exposure
6. KINETICS AND METABOLISM IN LABORATORY ANIMALS AND HUMANS
6.1. Absorption
6.1.1. Absorption after inhalation
6.1.1.1 Animal studies
6.1.1.2 Human studies
6.1.2. Absorption of lead from the gastrointestinal tract
6.1.2.1 Animal studies
6.1.2.2 Human studies
6.1.2.3 Nutritional status and lead absorption
via gastrointestinal tract
6.1.3. Dermal absorption
6.1.3.1 Human dermal absorption
6.1.4. The relationship of external lead exposure to blood
lead concentration
6.1.4.1 Ambient air
6.1.4.2 Food
6.1.4.3 Drinking-water
6.1.4.4 Soil and dust
6.1.4.5 Total lead intake
6.2. Distribution
6.2.1. Animal studies
6.2.2. Human studies
6.2.3. Transplacental transfer
6.3. Elimination and excretion
6.4. Biological indices of lead exposure and body burden
6.4.1. Blood lead
6.4.2. Tooth lead
6.4.3. Bone lead
6.4.4. Lead in urine
6.4.5. Lead in hair
7. EFFECTS ON LABORATORY ANIMALS AND IN VITRO TEST SYSTEMS
7.1. Biochemical effects
7.1.1. Haem synthesis and haematopoiesis
7.2. Nervous system effects
7.2.1. Higher order behavioural toxicity
7.2.2. Mechanisms of lead-induced behavioural toxicity
7.2.2.1 Conclusions
7.2.3. Sensory organ toxicity
7.3. Renal system
7.4. Cardiovascular system
7.5. Reproductive system
7.6. Effects on bone
7.7. Immunological effects
7.8. Mutagenicity
7.9. Carcinogenicity
8. EFFECTS ON HUMANS
8.1. Biochemical effects of lead
8.1.1. Haem synthesis
8.1.1.1 Protoporphyrin levels
8.1.1.2 Coproporphyrin levels
8.1.1.3 delta-Aminolaevulinic acid levels in
urine and blood
8.1.1.4 Aminolaevulinic acid dehydratase levels
8.1.1.5 delta-Aminolaevulinic acid synthase
8.1.1.6 Other effects of decreased haem
synthesis
8.1.2. Vitamin D
8.1.3. Dihydrobiopterin reductase
8.1.4. Nicotinamide adenine dinucleotide synthetase
8.1.5. Nutritionally affected groups
8.2. Haematopoietic system
8.2.1. Anaemia
8.2.2. Pyrimidine-5'-nucleotidase activity
8.2.3. Erythropoietin production
8.3. Nervous system
8.3.1. Historical perspective
8.3.2. Neurotoxic effects in adults
8.3.2.1 Central nervous system
8.3.2.2 Peripheral nervous system
8.3.2.3 Autonomic nervous system
8.3.3. Neurotoxic effects in children
8.3.3.1 Historical perspective
8.3.4. Population-based cross-sectional studies on
children
8.3.4.1 Tooth lead studies
8.3.4.2 Blood lead studies
8.3.4.3 Follow-up studies
8.3.4.4 Conclusions and limitations of
cross-sectional studies
8.3.5. Prospective epidemiological studies on children
8.3.5.1 Common elements
8.3.5.2 Study descriptions
8.3.5.3 Summary of differences between studies
8.3.5.4 Results of studies
8.3.5.5 Questions prospective studies have not
answered
8.3.5.6 Attempting a consensus
8.3.6. Task group overview and interpretation of
prospective studies on children
8.3.6.1 Rationale
8.3.6.2 The prospective studies
8.3.6.3 A quantitative assessment of the
cross-sectional studies
8.3.6.4 Task group overview of cross-sectional
studies
8.3.6.5 An interpretation of the overview of
prospective and cross-sectional studies
8.3.7. Hearing impairment in children
8.4. Renal system
8.4.1. Clinical studies
8.4.2. Epidemiological studies
8.4.2.1 Occupational cohorts
8.4.2.2 General population
8.4.2.3 Cohort mortality studies
8.5. Cardiovascular system
8.5.1. Blood pressure
8.5.1.1 Studies on occupationally exposed
cohorts
8.5.1.2 Studies in the general population
8.5.2. Other cardiovascular effects
8.5.2.1 Occupational studies
8.5.2.2 Studies in the general population
8.5.3. Summary
8.6. Gastrointestinal effects
8.6.1. Occupational exposure
8.6.2. Exposure of children
8.7. Liver
8.7.1. Occupational exposure
8.7.2. Exposure of children
8.8. Reproduction
8.8.1. Female populations
8.8.2. Male populations
8.8.3. Hormonal responses
8.8.4. Postnatal growth and stature
8.9. Effects on chromosomes
8.10. Carcinogenicity
8.10.1. Occupational exposure and renal cancer
8.10.2. Conclusion
8.11. Effects on thyroid function
8.11.1. Occupational groups
8.11.2. Effects in children
8.12. Immune system
8.12.1. Occupational exposure
8.12.2. Children
8.13. Effects on bone
8.14. Biomarkers for lead effects
9. EVALUATION OF HUMAN HEALTH RISKS
9.1. Exposure assessment
9.1.1. General population exposure
9.1.2. Occupational exposures
9.2. Critical issues related to exposure evaluation
9.2.1. Sampling and analytical concerns
9.2.2. Data presentation
9.3. Relationship between exposure and dose
9.4. Surrogate measures of dose
9.4.1. Blood
9.4.2. Urine
9.4.3. Bone
9.4.4. Tooth
9.4.5. Hair
9.5. Biochemical effects of lead
9.5.1. Haem synthesis
9.5.1.1 Urinary coproporphyrin
9.5.1.2 Urinary aminolaevulinic acid in children
9.5.1.3 Urinary aminolaevulinic acid in adults
9.5.1.4 delta-Aminolaevulinic acid dehydratase
9.5.2. Vitamin D metabolism
9.5.3. Dihydrobiopterin reductase
9.5.4. Haemopoietic system
9.5.4.1 Anaemia in adults
9.5.4.2 Anaemia in children
9.5.4.3 Erythrocyte pyrimidine-5'-nucleotidase
9.6. Nervous system
9.6.1. Adults
9.6.1.1 Central nervous system
9.6.1.2 Peripheral nervous system
9.6.1.3 Autonomic nervous system
9.6.2. Children
9.6.2.1 Type of effect
9.6.2.2 Magnitude
9.6.2.3 Reversibility/persistence
9.6.2.4 Age-specific sensitivity
9.6.2.5 Interactions/subgroups
9.6.3. Animal studies
9.7. Renal system
9.8. Liver
9.9. Reproduction
9.9.1. Female
9.9.2. Male
9.10. Blood pressure
9.11. Carcinogenicity
9.12. Immune system
10. RECOMMENDATIONS FOR PROTECTION OF HUMAN HEALTH
10.1. Public health measures
10.2. Public health programmes
10.3. Screening, monitoring and assessment procedures
11. FURTHER RESEARCH
12. PREVIOUS EVALUATIONS BY INTERNATIONAL BODIES
REFERENCES
RESUME
RESUMEN
NOTE TO READERS OF THE CRITERIA MONOGRAPHS
Every effort has been made to present information in the criteria
monographs as accurately as possible without unduly delaying their
publication. In the interest of all users of the Environmental Health
Criteria monographs, readers are requested to communicate any errors
that may have occurred to the Director of the International Programme
on Chemical Safety, World Health Organization, Geneva, Switzerland, in
order that they may be included in corrigenda.
* * *
A detailed data profile and a legal file can be obtained from the
International Register of Potentially Toxic Chemicals, Case postale
356, 1219 Châtelaine, Geneva, Switzerland (Telephone No. 9799111).
* * *
This publication was made possible by grant number 5 U01
ES02617-15 from the National Institute of Environmental Health
Sciences, National Institutes of Health, USA, and by financial support
from the European Commission.
Environmental Health Criteria
PREAMBLE
Objectives
In 1973 the WHO Environmental Health Criteria Programme was
initiated with the following objectives:
(i) to assess information on the relationship between exposure to
environmental pollutants and human health, and to provide
guidelines for setting exposure limits;
(ii) to identify new or potential pollutants;
(iii) to identify gaps in knowledge concerning the health effects
of pollutants;
(iv) to promote the harmonization of toxicological and
epidemiological methods in order to have internationally
comparable results.
The first Environmental Health Criteria (EHC) monograph, on
mercury, was published in 1976 and since that time an ever-increasing
number of assessments of chemicals and of physical effects have been
produced. In addition, many EHC monographs have been devoted to
evaluating toxicological methodology, e.g., for genetic, neurotoxic,
teratogenic and nephrotoxic effects. Other publications have been
concerned with epidemiological guidelines, evaluation of short-term
tests for carcinogens, biomarkers, effects on the elderly and so
forth.
Since its inauguration the EHC Programme has widened its scope,
and the importance of environmental effects, in addition to health
effects, has been increasingly emphasized in the total evaluation of
chemicals.
The original impetus for the Programme came from World Health
Assembly resolutions and the recommendations of the 1972 UN Conference
on the Human Environment. Subsequently the work became an integral
part of the International Programme on Chemical Safety (IPCS), a
cooperative programme of UNEP, ILO and WHO. In this manner, with the
strong support of the new partners, the importance of occupational
health and environmental effects was fully recognized. The EHC
monographs have become widely established, used and recognized
throughout the world.
The recommendations of the 1992 UN Conference on Environment and
Development and the subsequent establishment of the Intergovernmental
Forum on Chemical Safety with the priorities for action in the six
programme areas of Chapter 19, Agenda 21, all lend further weight to
the need for EHC assessments of the risks of chemicals.
Scope
The criteria monographs are intended to provide critical reviews
on the effect on human health and the environment of chemicals and of
combinations of chemicals and physical and biological agents. As such,
they include and review studies that are of direct relevance for the
evaluation. However, they do not describe every study carried out.
Worldwide data are used and are quoted from original studies, not from
abstracts or reviews. Both published and unpublished reports are
considered and it is incumbent on the authors to assess all the
articles cited in the references. Preference is always given to
published data. Unpublished data are only used when relevant published
data are absent or when they are pivotal to the risk assessment. A
detailed policy statement is available that describes the procedures
used for unpublished proprietary data so that this information can be
used in the evaluation without compromising its confidential nature
(WHO (1990) Revised Guidelines for the Preparation of Environmental
Health Criteria Monographs. PCS/90.69, Geneva, World Health
Organization).
In the evaluation of human health risks, sound human data,
whenever available, are preferred to animal data. Animal and
in vitro studies provide support and are used mainly to supply
evidence missing from human studies. It is mandatory that research on
human subjects is conducted in full accord with ethical principles,
including the provisions of the Helsinki Declaration.
The EHC monographs are intended to assist national and
international authorities in making risk assessments and subsequent
risk management decisions. They represent a thorough evaluation of
risks and are not, in any sense, recommendations for regulation or
standard setting. These latter are the exclusive purview of national
and regional governments.
Content
The layout of EHC monographs for chemicals is outlined below.
* Summary - a review of the salient facts and the risk evaluation
of the chemical
* Identity - physical and chemical properties, analytical methods
* Sources of exposure
* Environmental transport, distribution and transformation
* Environmental levels and human exposure
* Kinetics and metabolism in laboratory animals and humans
* Effects on laboratory mammals and in vitro test systems
* Effects on humans
* Effects on other organisms in the laboratory and field
* Evaluation of human health risks and effects on the environment
* Conclusions and recommendations for protection of human health
and the environment
* Further research
* Previous evaluations by international bodies, e.g., IARC, JECFA,
JMPR
Selection of chemicals
Since the inception of the EHC Programme, the IPCS has organized
meetings of scientists to establish lists of priority chemicals for
subsequent evaluation. Such meetings have been held in: Ispra, Italy,
1980; Oxford, United Kingdom, 1984; Berlin, Germany, 1987; and North
Carolina, USA, 1995. The selection of chemicals has been based on the
following criteria: the existence of scientific evidence that the
substance presents a hazard to human health and/or the environment;
the possible use, persistence, accumulation or degradation of the
substance shows that there may be significant human or environmental
exposure; the size and nature of populations at risk (both human and
other species) and risks for environment; international concern, i.e.
the substance is of major interest to several countries; adequate data
on the hazards are available.
If an EHC monograph is proposed for a chemical not on the
priority list, the IPCS Secretariat consults with the Cooperating
Organizations and all the Participating Institutions before embarking
on the preparation of the monograph.
Procedures
The order of procedures that result in the publication of an EHC
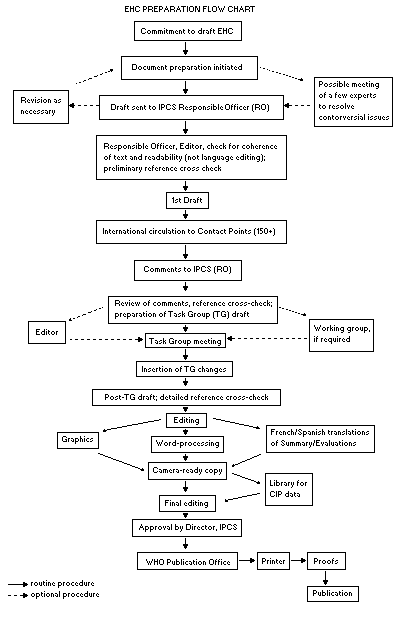
monograph is shown in the flow chart. A designated staff member of
IPCS, responsible for the scientific quality of the document, serves
as Responsible Officer (RO). The IPCS Editor is responsible for layout
and language. The first draft, prepared by consultants or, more
usually, staff from an IPCS Participating Institution, is based
initially on data provided from the International Register of
Potentially Toxic Chemicals, and reference data bases such as Medline
and Toxline.
The draft document, when received by the RO, may require an
initial review by a small panel of experts to determine its scientific
quality and objectivity. Once the RO finds the document acceptable as
a first draft, it is distributed, in its unedited form, to well over
150 EHC contact points throughout the world who are asked to comment
on its completeness and accuracy and, where necessary, provide
additional material. The contact points, usually designated by
governments, may be Participating Institutions, IPCS Focal Points, or
individual scientists known for their particular expertise. Generally
some four months are allowed before the comments are considered by the
RO and author(s). A second draft incorporating comments received and
approved by the Director, IPCS, is then distributed to Task Group
members, who carry out the peer review, at least six weeks before
their meeting.
The Task Group members serve as individual scientists, not as
representatives of any organization, government or industry. Their
function is to evaluate the accuracy, significance and relevance of
the information in the document and to assess the health and
environmental risks from exposure to the chemical. A summary and
recommendations for further research and improved safety aspects are
also required. The composition of the Task Group is dictated by the
range of expertise required for the subject of the meeting and by the
need for a balanced geographical distribution.
The three cooperating organizations of the IPCS recognize the
important role played by nongovernmental organizations.
Representatives from relevant national and international associations
may be invited to join the Task Group as observers. While observers
may provide a valuable contribution to the process, they can only
speak at the invitation of the Chairperson. Observers do not
participate in the final evaluation of the chemical; this is the sole
responsibility of the Task Group members. When the Task Group
considers it to be appropriate, it may meet in camera.
All individuals who as authors, consultants or advisers
participate in the preparation of the EHC monograph must, in addition
to serving in their personal capacity as scientists, inform the RO if
at any time a conflict of interest, whether actual or potential, could
be perceived in their work. They are required to sign a conflict of
interest statement. Such a procedure ensures the transparency and
probity of the process.
When the Task Group has completed its review and the RO is
satisfied as to the scientific correctness and completeness of the
document, it then goes for language editing, reference checking, and
preparation of camera-ready copy. After approval by the Director,
IPCS, the monograph is submitted to the WHO Office of Publications for
printing. At this time a copy of the final draft is sent to the
Chairperson and Rapporteur of the Task Group to check for any errors.
 It is accepted that the following criteria should initiate the
updating of an EHC monograph: new data are available that would
substantially change the evaluation; there is public concern for
health or environmental effects of the agent because of greater
exposure; an appreciable time period has elapsed since the last
evaluation.
All Participating Institutions are informed, through the EHC
progress report, of the authors and institutions proposed for the
drafting of the documents. A comprehensive file of all comments
received on drafts of each EHC monograph is maintained and is
available on request. The Chairpersons of Task Groups are briefed
before each meeting on their role and responsibility in ensuring that
these rules are followed.
WHO TASK GROUP ON ENVIRONMENTAL HEALTH CRITERIA FOR INORGANIC LEAD
Members
Professor S. Araki, Department of Public Health, Faculty of Medicine,
University of Tokyo, Japan
Dr P. Baghurst, Division of Human Nutrition, Commonwealth Scientific
Industrial Research Organization, Adelaide, Australia
Dr D. Bellinger, Neuroepidemiology Unit, Gardner House, Children's
Hospital, Boston, Massachusetts, USA
Dr I. Calder, Occupational and Environmental Health, South Australian
Health Commission, Adelaide, South Australia, Australia
Dr D.A. Cory-Slechta, Department of Environmental Medicine,
University of Rochester School of Medicine and Dentistry,
Rochester, New York, USA
Dr K. Dietrich, Department of Environmental Health, Division of
Biostatistics and Epidemiology, University of Cincinnati College
of Medicine, Cincinnati, Ohio, USA
Dr R.A. Goyer, Chapel Hill, North Carolina, USA (Chairman)
Dr M.R. Moore, University of Glasgow, Department of Medicine and
Therapeutics, Western Infirmary, Glasgow, Scotland
Dr C. Nam Ong, Department of Community, Occupational and Family
Medicine, National University of Singapore, National University
Hospital, Singapore
Dr S.J. Pocock, Department of Epidemiology and Population Sciences,
Medical Statistics Unit, University of London, London, England
Dr M.B. Rabinowitz, Marine Biological Laboratory, Woods Hole,
Massachusetts, USA
Dr M. Smith, Thomas Coram Research Unit, London, England
Dr G. Winneke, Medical Institute for Environmental Health,
Heinrich-Heine University, Düsseldorf, Germany (Vice-Chairman)
Observers
Dr C. Boreiko, Environmental Health, International Lead Zinc Research
Organization (ILZRO) Inc., Research Triangle Park, North
Carolina, USA
Dr N.H. Clark, Lead Industry Environment and Health Forum, Melbourne,
Victoria, Australia
Dr J.M. Davis, Environmental Criteria and Assessment Office, US
Environmental Protection Agency, Research Triangle Park, North
Carolina, USA
Professor G. Duggin, Toxicology Unit, Royal Prince Alfred Hospital,
Camperdown, Australia
Dr G.R. Neville, Queensland Health Department, Brisbane, Australia
Secretariat
Dr G.C. Becking, International Programme on Chemical Safety,
Interregional Research Unit, World Health Organization, Research
Triangle Park, North Carolina, USA (Secretary)
Dr K.R. Mahaffey, National Institute of Environmental Health
Sciences, Research Triangle Park, North Carolina, USAa
Dr A.E. Robinson, Toronto, Ontario, Canada (Rapporteur)
a Present address: US Environmental Protection Agency, Environmental
Criteria and Assessment Office, Cincinnati, Ohio, USA
ENVIRONMENTAL HEALTH CRITERIA FOR INORGANIC LEAD
A WHO Task Group on Environmental Health Criteria for Inorganic
Lead met in Brisbane, Australia, from 1 to 6 February 1993. The
meeting was sponsored by a consortium of Australian Commonwealth and
State Governments through a national Steering Committee chaired by
Dr Keith Bentley, Director, Health and Environmental Policy,
Department of Human Services and Health, Canberra. The meeting was
hosted and organized by the Queensland Department of Health,
Dr G.R. Neville being responsible for the arrangements. Dr G. Murphy,
Director of Public Health, Queensland, welcomed the participants on
behalf of the Organizers, and Dr T. Adams, Chief Commonwealth Medical
Advisor and Dr G. Johns, Parliamentary Secretary to Federal Minister
for Health, Housing and Community Services, welcomed the participants
on behalf of the Commonwealth Government. Dr Johns stressed the
importance attached to this IPCS meeting by the Commonwealth and State
Governments of Australia. Dr G.C. Becking, IPCS, welcomed the
participants on behalf of Dr M. Mercier, Director of the IPCS and the
three cooperating organizations (UNEP/ILO/WHO). The Task Group
reviewed and revised the draft criteria monograph, and made an
evaluation of the risks to human health from exposure to inorganic
lead.
The Task Group draft was prepared by Dr A.E. Robinson, Toronto,
Canada, using texts made available by Dr K.R. Mahaffeya (National
Institute of Environmental Health Sciences, Research Triangle Park,
North Carolina, USA) and Dr E. Silbergeld (University of Maryland
School of Medicine, Baltimore, Maryland, USA), and the comments
received from the IPCS contact points for environmental health
criteria monographs. The draft was revised extensively by the Task
Group taking into account the comments from the IPCS contact points.
Dr G.C. Becking (IPCS Central Unit, Interregional Research Unit)
and Dr P.G. Jenkins (IPCS Central Unit, Geneva) were responsible for
the overall scientific content and technical editing, respectively, of
this monograph.
The efforts of all who helped in the preparation and finalization
of this publication are gratefully acknowledged.
a Present address: US Environmental Protection Agency, Environmental
Criteria and Assessment Office, Cincinnati, Ohio, USA
ABBREVIATIONS
AAS atomic absorption spectrometry
AES atomic emission spectroscopy
ALA delta-aminolaevulinic acid
ALAD delta-aminolaevulinic acid dehydratase
ASV anodic stripping voltametry
EDTA ethylenediaminetetraacetic acid
FEP free erythrocyte porphyrin
GFAAS graphite furnace atomic absorption spectrometry
ICP inductively coupled plasma
IDMS isotope dilution mass spectrometry
MPb mobilization yield of lead
MSW municipal solid waste
PbB blood lead
PbT tooth lead
TML tetramethyllead
XRFS X-ray fluorescence spectroscopy
ZPP zinc protoporphyrin
PREFACE
Although many countries have initiated programmes to lower the
level of lead in the environment, human exposure to lead remains of
concern to public health officials worldwide. For over 20 years the
World Health Organization (WHO) and the International Programme on
Chemical Safety (IPCS) have been concerned about the health and
environmental effects of the levels of inorganic lead in the
environment. The evaluation of human health risks arising from
food-borne lead has been carried out by WHO on four occasions since
1972. In addition, health-based guidance values for lead in water, air
and the workplace have been developed by various Task Groups convened
by WHO. Environmental Health Criteria 3: Lead, published in 1977,
examined the effects of lead on human health and Environmental Health
Criteria 85: Lead - Environmental Aspects was published in 1989.
Since the publication of Environmental Health Criteria 3: Lead, a
large body of knowledge has accumulated concerning the effects of lead
on humans at low levels of exposure. Studies have emphasized the
effects of inorganic lead on infants and children, a high-risk
population. This monograph on inorganic lead reflects this research
emphasis; a major part of the monograph deals with the neurotoxic
effects of lead with emphasis on neurobehavioural development in
children. Less detail is presented on the health effects of the higher
levels of inorganic lead found in some workplaces, although such
exposures are still considered to pose a risk to humans in many
regions of the world.
This monograph deals only with the human health effects of
inorganic lead. No attempt has been made to evaluate the human health
effects of organo-lead compounds, although it was recognized that such
compounds when added to petrol (gasoline) are a major source of
inorganic lead in the environment. In view of the toxicity of many
organo-lead derivatives and the possible methylation of inorganic lead
in the environment, the IPCS plans to evaluate the risk to humans from
exposure to organo-lead compounds in a separate monograph.
As with all IPCS criteria monographs, no attempt has been made to
prepare an exhaustive bibliography of the extremely large amount of
lead-related literature published since 1977. Rather, an effort has
been made to review critically the studies on humans and experimental
animals that are essential for the evaluation of risks to human health
from exposure to all sources of inorganic lead.
1. SUMMARY
This monograph focuses on the risks to human health associated
with exposure to lead and inorganic lead compounds. Emphasis has been
given to data which have become available since the publication of
Environmental Health Criteria 3: Lead (IPCS, 1977). The environmental
effects of lead are discussed in Environmental Health Criteria 85:
Lead - Environmental Aspects (IPCS, 1989).
1.1 Identity, physical and chemical properties, and analytical
methods
Lead is a soft, silvery grey metal, melting at 327.5°C. It is
highly resistant to corrosion, but is soluble in nitric and hot
sulfuric acids. The usual valence state in inorganic lead compounds is
+2. Solubilities in water vary, lead sulfide and lead oxides being
poorly soluble and the nitrate, chlorate and chloride salts are
reasonably soluble in cold water. Lead also forms salts with such
organic acids as lactic and acetic acids, and stable organic compounds
such as tetraethyllead and tetramethyllead.
The most commonly used methods for the analysis of low
concentrations of lead in biological and environmental materials are
flame, graphite furnace and inductively coupled plasma atomic
absorption spectroscopy and anode stripping voltametry. Depending on
sample pretreatment, extraction techniques and analytical
instrumentation, detection limits of 0.12 µmoles lead/litre blood
(2.49 µg/dl) can be achieved. However, reliable results are obtained
only when specific procedures are followed to minimize the risk of
contamination during sample collection, storage, processing and
analysis.
1.2 Sources of human exposure
The level of lead in the earth's crust is about 20 mg/kg. Lead in
the environment may derive from either natural or anthropogenic
sources. Natural sources of atmospheric lead include geological
weathering and volcanic emissions and have been estimated at
19 000 tonnes/year, compared to an estimate of 126 000 tonnes/year
emitted to the air from the mining, smelting and consumption of over 3
million tonnes of lead per year.
Atmospheric lead concentrations of 50 pg/m3 have been found in
remote areas. Background levels of lead in soil range between 10 and
70 mg/kg and a mean level near roadways of 138 mg/kg has been
reported. Present levels of lead in water rarely exceed a few
micrograms/litre; the natural concentration of lead in surface water
has been estimated to be 0.02 µg/litre.
Lead and its compounds may enter the environment at any point
during mining, smelting, processing, use, recycling or disposal. Major
uses are in batteries, cables, pigments, petrol (gasoline) additives,
solder and steel products. Lead and lead compounds are also used in
solder applied to water distribution pipes and to seams of cans used
to store foods, in some traditional remedies, in bottle closures for
alcoholic beverages and in ceramic glazes and crystal tableware. In
countries where leaded petrol is still used, the major air emission is
from mobile and stationary sources of petrol combustion (urban
centres). Areas in the vicinity of lead mines and smelters are subject
to high levels of air emissions.
Airborne lead can be deposited on soil and water, thus reaching
humans through the food chain and in drinking-water. Atmospheric lead
is also a major source of lead in household dust.
1.3 Environmental transport, distribution and transformation
The transport and distribution of lead from fixed, mobile and
natural sources are primarily via air. Most lead emissions are
deposited near the source, although some particulate matter (< 2 µm
in diameter) is transported over long distances and results in the
contamination of remote sites such as arctic glaciers. Airborne lead
can contribute to human exposures by the contamination of food, water
and dust, as well as through direct inhalation. The removal of
airborne lead is influenced by atmospheric conditions and particulate
size. Large amounts of lead may be discharged to soil and water.
However, such material tends to remain localized because of the poor
solubility of lead compounds in water.
Lead deposited in water, whether from air or through run-off from
soils, partitions rapidly between sediment and aqueous phase,
depending upon pH, salt content, and the presence of organic chelating
agents. Above pH 5.4, hard water may contain about 30 µg lead/litre
and soft water about 500 µg lead/litre. Very little lead deposited on
soil is transported to surface or ground water except through erosion
or geochemical weathering; it is normally quite tightly bound
(chelated) to organic matter.
Airborne lead can be transferred to biota directly or through
uptake from soil. Animals can be exposed to lead directly through
grazing and soil ingestion or by inhalation. There is little
biomagnification of inorganic lead through the food chain.
1.4 Environmental levels and human exposure
In the general non-smoking adult population, the major exposure
pathway is from food and water. Airborne lead may contribute
significantly to exposure, depending upon such factors as use of
tobacco, occupation, proximity to motorways, lead smelters, etc., and
leisure activities (e.g., arts and crafts, firearm target practice).
Food, air, water and dust/soil are the major potential exposure
pathways for infants and young children. For infants up to 4 or 5
months of age, air, milk, formulae and water are the significant
sources of lead exposure.
Levels of lead found in air, food, water and soil/dust vary
widely throughout the world and depend upon the degree of industrial
development, urbanization and lifestyle factors. Ambient air levels
over 10 µg/m3 have been reported in urban areas near a smelter,
whereas lead levels below 0.2 µg/m3 have been found in cities where
leaded petrol is no longer used. Lead intake from air can, therefore,
vary from less than 4 µg/day to more than 200 µg/day.
Levels of lead in drinking-water sampled at the source are
usually below 5 µg/litre. However, water taken from taps (faucets) in
homes where lead is present in the plumbing can contain levels in
excess of 100 µg/litre, particularly after the water has been standing
in the pipes for some hours.
The level of dietary exposure to lead depends upon many lifestyle
factors, including foodstuffs consumed, processing technology, use of
lead solder, lead levels in water, and use of lead-glazed ceramics.
For infants and children, lead in dust and soil often constitutes
a major exposure pathway. Lead levels in dust depend upon such factors
as the age and condition of housing, the use of lead-based paints,
lead in petrol and urban density. The intake of lead will be
influenced by the age and behavioural characteristics of the child and
bioavailability of lead in the source material.
Inhalation is the dominant pathway for lead exposure of workers
in industries producing, refining, using or disposing of lead and lead
compounds. During an 8-h shift, workers can absorb as much as 400 µg
lead, in addition to the 20-30 µg/day absorbed from food, water and
ambient air; significant intake may occur from ingestion of large
inhaled particulate material.
1.5 Kinetics and metabolism in laboratory animals and humans
Lead is absorbed in humans and animals following inhalation or
ingestion; percutaneous absorption is minimal in humans. Depending
upon chemical speciation, particle size, and solubility in body
fluids, up to 50% of the inhaled lead compound may be absorbed. Some
inhaled particulate matter (larger than 7 µm) is swallowed following
mucociliary clearance from the respiratory tract. In experimental
animals and humans, absorption of lead from the gastrointestinal tract
is influenced by the physico-chemical nature of the ingested material,
nutritional status, and type of diet consumed. In adult humans
approximately 10% of the dietary lead is absorbed; the proportion is
higher under fasting conditions. However, in infants and young
children as much as 50% of dietary lead is absorbed, although
absorption rates for lead from dusts/soils and paint chips can be
lower depending upon the bioavailability. Diets that are deficient in
calcium, phosphate, selenium or zinc may result in increased lead
absorption. Iron and vitamin D also affect absorption of lead.
Blood lead (PbB) levels are used as a measure of body burden and
absorbed (internal) doses of lead. The relationship between blood lead
and the concentration of lead in exposure sources is curvilinear.
Once it has been absorbed, lead is not distributed homogeneously
throughout the body. There is rapid uptake into blood and soft tissue,
followed by a slower redistribution to bone. Bone accumulates lead
over much of the human life span and may serve as an endogenous source
of lead. The half-life for lead in blood and other soft tissues is
about 28-36 days, but it is much longer in the various bone
compartments. The percentage retention of lead in body stores is
higher in children than adults. Transfer of lead to the human fetus
occurs readily throughout gestation.
Blood lead is the most commonly used measure of lead exposure.
However, techniques are now available for measuring lead in teeth and
bone, although the kinetics are not fully understood.
1.6 Effects on laboratory animals and in vitro systems
In all species of experimental animals studied, including
non-human primates, lead has been shown to cause adverse effects in
several organs and organ systems, including the haematopoietic,
nervous, renal, cardiovascular, reproductive and immune systems. Lead
also affects bone and has been shown to be carcinogenic in rats and
mice.
Despite kinetic differences between experimental animal species
and humans, these studies provide strong biological support and
plausibility for the findings in humans. Impaired learning/memory
abilities have been reported in rats with PbB levels of
0.72-0.96 µmoles/litre (15-20 µg/dl) and in non-human primates at PbB
levels not exceeding 0.72 µmoles/litre (15 µg/dl). In addition, visual
and auditory impairments have been reported in experimental animal
studies.
Renal toxicity in rats appears to occur at a PbB level in excess
of 2.88 µmol/litre (60 µg/dl), a value similar to that reported to
initiate renal effects in humans. Cardiovascular effects have been
seen in rats after chronic low-level exposures resulting in PbB levels
of 0.24-1.92 µmol/litre (5-40 µg/dl). Tumours have been shown to occur
at dose levels below the maximum tolerated dose of 200 mg lead (as
lead acetate) per litre of drinking-water. This is the maximum dose
level not associated with other morphological or functional changes.
1.7 Effects on humans
In humans, lead can result in a wide range of biological effects
depending upon the level and duration of exposure. Effects at the
subcellular level, as well as effects on the overall functioning of
the body, have been noted and range from inhibition of enzymes to the
production of marked morphological changes and death. Such changes
occur over a broad range of doses, the developing human generally
being more sensitive than the adult.
Lead has been shown to have effects on many biochemical
processes; in particular, effects on haem synthesis have been studied
extensively in both adults and children. Increased levels of serum
erythrocyte protoporphyrin and increased urinary excretion of
coproporphyrin and delta-aminolaevulinic acid are observed when PbB
concentrations are elevated. Inhibition of the enzymes
delta-aminolaevulinic acid dehydratase and dihydrobiopterin reductase
are observed at lower levels.
The effects of lead on the haemopoietic system result in
decreased haemoglobin synthesis, and anaemia has been observed in
children at PbB concentrations above 1.92 µmol/litre (40 µg/dl).
For neurological, metabolic and behavioural reasons, children are
more vulnerable to the effects of lead than adults. Both prospective
and cross-sectional epidemiological studies have been conducted to
assess the extent to which environmental lead exposure affects
CNS-based psychological functions. Lead has been shown to be
associated with impaired neurobehavioural functioning in children.
Impairment of psychological and neurobehavioural functions has
been found after long-term lead exposure of workers.
Electrophysiological parameters have been shown to be useful
indicators of subclinical lead effects in the CNS.
Peripheral neuropathy has long been known to be caused by
long-term high-level lead exposure at the workplace. Slowing of nerve
conduction velocity has been found at lower levels. These effects have
often been found to be reversible after cessation of exposure,
depending on the age and duration of exposure.
The effect of lead on the heart is indirect and occurs via the
autonomic nervous system; it has no direct effect on the myocardium.
The collective evidence from population studies in adults indicates
very weak associations between PbB concentration and systolic or
diastolic blood pressure. Given the difficulties of allowing for
relevant confounding factors, a causal relationship cannot be
established from these studies. There is no evidence to suggest that
any association of PbB concentration with blood pressure is of major
health importance.
Lead is known to cause proximal renal tubular damage,
characterized by generalized aminoaciduria, hypophosphataemia with
relative hyperphosphaturia and glycosuria accompanied by nuclear
inclusion bodies, mitochondrial changes and cytomegaly of the proximal
tubular epithelial cells. Tubular effects are noted after relatively
short-term exposures and are generally reversible, whereas sclerotic
changes and interstitial fibrosis, resulting in decreased kidney
function and possible renal failure, require chronic exposure to high
lead levels. Increased risk from nephropathy was noted in workers with
a PbB level of over 3.0 µmol/litre (about 60 µg/dl). Renal effects
have recently been seen among the general population when more
sensitive indicators of function were measured.
The reproductive effects of lead in the male are limited to sperm
morphology and count. In the female, some adverse pregnancy outcomes
have been attributed to lead.
Lead does not appear to have deleterious effects on skin, muscle
or the immune system. Except in the case of the rat, lead does not
appear to be related to the development of tumours.
1.8 Evaluation of human health risks
Lead adversely affects several organs and organ systems, with
subcellular changes and neurodevelopmental effects appearing to be the
most sensitive. An association between PbB level and hypertension
(blood pressure) has been reported. Lead produces a cascade of effects
on the haem body pool and affects haem synthesis. However, some of
these effects are not considered adverse. Calcium homoeostasis is
affected, thus interfering with other cellular processes.
a) The most substantial evidence from cross-sectional and
prospective studies of populations with PbB levels generally
below 1.2 µmol/litre (25 µg/dl) relates to decrements in
intelligence quotient (IQ). It is important to note that such
observational studies cannot provide definitive evidence of a
causal relationship with lead exposure. However, the size of the
apparent IQ effect, as assessed at 4 years and above, is a
deficit between 0 and 5 points (on a scale with a standard
deviation of 15) for each 0.48 µmol/litre (10 µg/dl) increment in
PbB level, with a likely apparent effect size of between 1 and 3
points. At PbB levels above 1.2 µmol/litre (25 µg/dl), the
relationship between PbB and IQ may differ. Estimates of effect
size are group averages and only apply to the individual child in
a probabilistic manner.
Existing epidemiological studies do not provide definitive
evidence of a threshold. Below the PbB range of 0.48-0.72 µmol/
litre (10-15 µg/dl), the effects of confounding variables and
limits in the precision in analytical and psychometric
measurements increase the uncertainty attached to any estimate
of effect. However, there is some evidence of an association
below this range.
b) Animal studies provide support for a causal relationship between
lead and nervous system effects, reporting deficits in cognitive
functions at PbB levels as low as 0.53-0.72 µmol/litre
(11-15 µg/dl) which can persist well beyond the termination of
lead exposure.
c) Reduction in human peripheral nerve conduction velocity may occur
with PbB levels as low as 1.44 µmol/litre (30 µg/dl). In
addition, sensory motor function may be impaired with PbB levels
as low as about 1.92 µmol/litre (40 µg/dl), and autonomic nervous
system function (electrocardiographic R-R interval variability)
may be affected at an average PbB level of approximately
1.68 µmol/litre (35 µg/dl). The risk of lead nephropathy is
increased in workers with PbB levels above 2.88 µmol/litre
(60 µg/dl). However, recent studies using more sensitive
indicators of renal function suggest renal effects at lower
levels of lead exposure.
d) Lead exposure is associated with a small increase in blood
pressure. The likely order of magnitude is that for any two-fold
increase in PbB level (e.g., from 0.8 to 1.6 µmol/litre, i.e.
16.6 to 33.3 µg/dl), there is a mean 1 mmHg increase in systolic
blood pressure. The association with diastolic pressure is of a
similar but smaller magnitude. However, there is doubt regarding
whether these statistical associations are really due to an
effect of lead exposure or are an artifact due to confounding
factors.
e) Some but not all epidemiological studies show a dose dependent
association of pre-term delivery and some indices of fetal growth
and maturation at PbB levels of 0.72 µmol/litre (15 µg/dl) or
more.
f) The evidence for carcinogenicity of lead and several inorganic
lead compounds in humans is inadequate.
g) Effects of lead on a number of enzyme systems and biochemical
parameters have been demonstrated. The PbB levels, above which
effects are demonstrable with current techniques for the
parameters that may have clinical significance, are all greater
than 0.96 µmol/litre (20 µg/dl). Some effects on enzymes are
demonstrable at lower PbB levels, but the clinical significance
is uncertain.
2. IDENTITY, PHYSICAL AND CHEMICAL PROPERTIES, AND ANALYTICAL METHODS
2.1 Physical and chemical properties of lead and its compounds
Lead (atomic number, 82; relative atomic mass, 207.19; specific
gravity, 11.34) is a bluish or silvery grey soft metal. The melting
point is 327.5°C and the boiling point at atmospheric pressure 1740°C.
It has four naturally occurring isotopes (208, 206, 207, and 204 in
order of abundance), but the isotopic ratios for various mineral
sources may differ. This property has been exploited in
non-radioactive-tracer environmental and metabolic studies. The
physical and chemical properties of elemental lead and some lead
compounds are summarized in Table 1.
Although lead has four electrons in its valence shell, only two
ionize readily. The usual oxidation state of lead in inorganic
compounds is therefore +2 rather than +4. The inorganic salts of lead,
such as lead sulfide and the oxides of lead, are generally poorly
soluble in water. However, the nitrate, chlorate and, to a much lesser
degree, the chloride are water soluble. Some of the salts formed with
organic acids, e.g., lead oxalate, are also insoluble, but the acetate
is relatively soluble, as shown in Table 1.
Under appropriate conditions of synthesis, stable compounds are
formed in which lead is directly bound to a carbon atom. Industrially
synthesized lead-carbon compounds include tetraethyllead and
tetramethyllead, which are of importance as fuel additives and, hence,
are sources of environmental lead.
2.2 Analytical procedures
In recent years substantial advances have been made in developing
methods for the quantification of metals at low concentrations. In
order to provide improved quality assurance of such measurements,
various reference materials in different matrices have been produced
(Muramatsu & Parr, 1985). To ensure adequate quality control, the
analyst should choose a reference material that matches as closely as
possible the experimental samples to be analysed. Choices are based
upon matrix type and concentration of the element of interest. A
summary of data on 60 biological and 40 environmental (non-biological)
reference materials has been compiled by Muramatsu & Parr (1985).
With the increased interest in measuring lead in the low µg/kg
and µg/m3 range in both environmental and biological samples, there
is need for particular attention to analytical sensitivity and
reliability. As lower concentrations are measured, problems of
laboratory contamination become more significant and quality control
and quality assurance programmes are important. Because of these
concerns, all analytical results for lead should report the laboratory
Table 1. Physical and chemical data on lead and selected lead compoundsa
Name Synonym Relative atomic/ Melting point Boiling point Solubility in cold Soluble in
and formula molecular mass (°C) (°C) water (g/litre)
Lead Pb 207.19 327.502 1740 insoluble HNO3; hot concentrated
H2SO4; hot water;
glycerine; alcohol (slightly)
Lead salts
acetate Pb(C2H3O2)2 325.28 280 - 443
carbonate cerrusite PbCO3 267.20 315 (decomposes) 0.0011 acid; alkali; decomposes in
hot water
chlorate Pb(ClO3)2 374.09 230 (decomposes) very soluble alcohol
chloride cotunite PbCl2 278.10 501 950 919 NH4 salts; slightly in dilute
HCl and in NH3; hot water
(33.4 g/litre)
nitrate Pb(NO3)2 331.20 470 (decomposes) 376.5 alcohol; alkali, NH3; hot
water (1270 g/litre)
orthophosphate Pb3(PO4)2 811.51 1014 0.00014 alkali; HNO3
oxalate PbC2O4 295.21 300 (decomposes) 0.0016 HNO3
dioxide plattnerite PbO2 239.19 290 (decomposes) insoluble dilute HCl; acetic acid
(slightly)
monoxide litharge PbO 223.19 888 0.017 dilute HNO3; acetic acid
Table 1 (cont'd)
Name Synonym Relative atomic/ Melting point Boiling point Solubility in cold Soluble in
and formula molecular mass (°C) (°C) water (g/litre)
sulfate anglesite PbSO4 303.25 1170 0.0425 NH4 salts; concentrated
H2SO4 (slightly)
sulfide galena PbS 239.25 1114 0.00086 acid
a Data from Weast (1985)
performance for reference standards and for parallel blank
measurements of sample contamination for the entire analytical
process. Without these, the validity of the data should be questioned.
2.2.1 Sampling procedures
Particular attention should be paid to the cleanliness of
equipment and glassware and the purity of the chemicals to prevent
secondary contamination by lead.
For the collection of samples, standard trace element methods are
generally required (Behne, 1980) with adequate quality control
procedures (Friberg, 1988; Jorhem & Slorach, 1988, Vahter & Friberg,
1988). Quality control samples for blood, faeces, air filters and dust
have been described (Lind et al., 1988).
2.2.1.1 Sampling of environmental media
In air sampling, both high-volume samplers and low-volume
techniques have been used. It should be noted that the collection
characteristics of high-volume samplers are strongly affected by
particle size and the orientation of the sampler. For particles larger
than 5 µm in diameter the high-volume sampler system is unlikely to
collect representative samples (US EPA, 1986a). As in all sampling for
suspended particulate matter, the accuracy of volume meters should be
checked periodically. The size of the pores in filters for collecting
lead-containing particles should be small, possibly less than 0.2 µm
for glass-fibre filters (Lee & Goranson, 1972).
Depending on the purpose of sampling, care should be taken to
select the appropriate site for sampling devices and to achieve the
best possible sampling conditions by:
* estimating the amount of particulate required for analysis before
deciding on the sample volume and the sampling procedure;
* placing the sampling devices in the appropriate position (e.g.,
in the breathing zone, level with inlet tubes of house
ventilators, at window level in the case of a traffic-laden town
street, at a reasonable distance from the highway in uninhabited
zones, etc);
* taking the samples at appropriate rates and volumes (e.g., daily
breathing volumes, daily ventilating capacities of installations)
and for a sufficient time to make possible the estimation of the
average concentration (e.g., during a work-shift, or a 24-h or
longer period for general population exposure);
* taking into account the use of areas under study (cattle grazing,
recreational zones, children's playgrounds, etc).
In addition, whenever possible a procedure should be used that
makes it possible to evaluate particle-size distribution and the
physicochemical properties of the lead compounds involved, including
the shape of the particles and the state of their aggregation.
Lead may be found in water bound to particulate matter as soluble
complexes or soluble compounds. Techniques for sampling water must
take this into account. It is necessary to sample water without
fractionation (filtration) when total lead levels are required.
Because of the potential for metals from low ionic strength waters to
be adsorbed onto the surfaces of some containers, samples should be
acidified (US EPA, 1986a). Selection, cleaning, and conditioning of
storage and sample containers deserve special attention (Moody, 1982).
The preparation of soil and dust samples for lead analyses
usually involves drying (at 100°C), homogenization by grinding, and
sieving (Thornton & Webb 1975; Bolter et al., 1975). Brown & Black
(1983) have discussed the issues related to quality assurance and
quality control in the collection and analysis of soil samples. Most
reports of lead in soil provide the total elemental abundance either
by acid extraction or X-ray fluorescence. However, the leachable or
bioavailable fraction is of special interest.
For the study of the dietary intake of lead from food, two
general methods have been utilized. The advantages and disadvantages
of the "duplicate portions" technique and the equivalent composite
technique ("market basket") have been reviewed by Pekkarinen (1970).
Although the duplicate portions (duplicate diets) technique can define
variability in consumption, it is expensive, and the sampling and
analytical procedures involved are complicated and limit the number of
individuals included in any study. With the equivalent composite
technique, the economy and ease of collection must be considered in
the light of the variability of results obtained due to uncertainties
in knowledge of actual preparation techniques, including possible lead
levels in water used for processing in individual homes.
The quantity of lead likely to be leached from ceramic surfaces
by different foods and beverages may be assessed using dilute acetic
acid solutions (1 to 4%) at temperatures in the range 20 to 100°C for
times ranging from 30 min to more than 24 h (Laurs, 1976; Merwin,
1976).
Colorimetric methods are suitable for screening inorganic
materials such as pottery or paint for lead. Positive reactions
require confirmation by established quantitative methods. Spot tests
using dithizone, rhodizonate and iodide (Feigl et al., 1972) are
available.
2.2.1.2 Sampling of biological materials
The main problem in the sampling of body fluids and tissues for
lead analysis is potential secondary contamination with lead. The low
general population blood lead (PbB) levels in many regions of the
world are complicating screening efforts, requiring levels of
analytical precision and sensitivity that can be achieved only through
intensive QA/QC programmes. Issues related to such sampling have been
examined in detail by US EPA (1986a).
Special precautions are needed to ensure that all venous
blood-collecting and blood-storage materials are as free from lead as
possible (IPCS, 1977). All glass equipment involved in blood
collection and storage should be made of lead-free silicate glass,
rinsed first in mineral acid, then with copious amounts of
glass-distilled or deionized water. Polypropylene syringes have been
recommended (NAS-NRC, l972). Needles should be of stainless steel with
polypropylene hubs. Blood is often drawn directly from the needle into
vacuum tubes. It is wise to confirm periodically the absence of
significant amounts of lead in the anticoagulant used in the blood
container as well as monitoring the contamination level (blank) for
the entire analytical process.
New analytical techniques make it possible to determine lead
concentrations in microlitre quantities of blood. The trend towards
the procurement of micro-samples of blood by skin prick increases the
chance of secondary contamination of the blood. Systematic
investigation on the significance of this problem has been reported
(Mitchell et al., 1974; Mahaffey et al., 1979; DeSilva & Donnan,
1980). Mitchell et al. (l974) describe a procedure whereby sample
contamination can be reduced by spraying collodion over the cleansed
skin before lancing. The correlation between the concentration of lead
in micro-samples and in macro-samples obtained by venepuncture was
fairly good (r=0.92) over a wide range of PbB concentrations
(0.48-4.41 µmol/litre or 10-92 µg/dl whole blood). Mahaffey et al.,
(1979b) found that capillary blood levels in a comparison test were
systematically higher than corresponding venous blood levels; similar
elevations have been reported by DeSilva & Donnan (1980). Since about
1980 the requirement for reliable and accurate micro procedures has
resulted in the development of good protocols. Sinclair & Dohnt (1984)
described a procedure which resulted in the ability to collect
capillary samples with PbB levels only 3.3% higher than the presumably
correct venous value. This procedure has been used in the Port Pirie
Cohort Study (Baghurst et al., 1985, 1992) and for routine
surveillance in the Port Pirie Lead Decontamination Program (Calder et
al., 1990). Also, Lyngbye et al. (1990b) have shown that capillary
sampling without lead contamination is possible. Routine validation by
cross-comparison with venous blood samples should be undertaken on a
regular basis.
The same general precautions to avoid contamination must be taken
in the collection of urine samples as in the collection of blood
samples. Additionally, special care must be taken to prevent
precipitation during storage.
2.2.2 Analytical methods for lead
A number of analytical methods exist for determination of lead in
environmental and biological samples. These methods differ enormously
in their costs (e.g., sophisticated equipment, an adequate
infrastructure to maintain laboratory conditions and chemical
supplies) and personnel requirements (e.g., availability of skilled
personnel in adequate numbers for the work to be undertaken). Both
accuracy and precision of any of the methods can be affected greatly
by contamination of samples within the laboratory. It is important to
utilize the principles of a "clean" laboratory described by Patterson
& Settle (1976) and Everson & Patterson (1980).
It is not the purpose of this section to provide an exhaustive
description of the analytical methods that could be available to
detect and quantify lead levels in environmental and biological
samples. However, an attempt will be made to identify well-established
methods in current use and to provide information on their application
to assist in the interpretation of experimental and epidemiological
studies.
2.2.2.1 Analysis of lead in environmental samples
The most common methods used for the analysis of lead in samples
from air, water, dust, sediment, soil and foodstuffs are flame atomic
absorption spectrometry (AAS), graphite furnace atomic absorption
spectrometry (GFAAS), anodic stripping voltametry (ASV), inductively
coupled plasma-atomic emission spectroscopy (ICP-AES), and X-ray
fluorescence spectroscopy (XRFS). The reference method for the
determination of the absolute amounts of lead is by isotope dilution
mass spectrometry (IDMS) (Settle & Patterson, 1980; Grandjean & Olsen,
1984; US EPA, 1986a), but due to equipment costs and required
expertise, it is not widely used. Spectrophotometric methods, using
diphenylthiocarbazone as the colorimetric reagent, were widely used in
the past; they are less sensitive and are labour-intensive but are
still appropriate. The advantages and disadvantages were described by
Skogerboe et al. (1977).
Gould et al. (1988) utilized a citric acid solution on filter
paper to leach lead from glazed ceramic and/or enamelled metal-ware.
When treated with a lead-sensitive chromogen, there is a reaction
indicating the presence of lead on the paper. The minimal amount of
lead required to produce an observable reaction was 0.25 µg/cm2; the
maximum amount tested was 5 µg/cm2. A colorimetric test based on the
use of sodium sulfide in solution is used to estimate lead in paint
films. It is possible to determine lead concentrations greater than
1 mg/cm2 of dried paint 90% of the time when the method is used by a
trained chemical laboratory technician.
Table 2 summarizes the utility of several representative methods
for specific environmental media.
2.2.2.2 Analysis of lead in biological materials
Biological samples present special problems for the analyst
because of the low lead concentrations and matrix effects. Most
analytical techniques developed to detect and quantify lead can be
adapted to the analysis of such biological materials as blood, urine,
serum, cerebrospinal fluid, solid tissues, hair, teeth and bone.
However, certain techniques are more often used for specific matrices.
Currently, the most commonly used methods are AAS, GFAAS, ASV,
and ICP-AES. Spectrophotometric methods were commonly used in the past
and can be useful. Other specialized methods for lead analysis are
XRFS, neutron activation analysis (NAA), inductively coupled
plasma-mass spectrometry (ICP-MS), and IDMS. Table 3 summarizes the
utility of several analytical procedures applied to various biological
matrices. Included in this table are examples of the application of
XRFS (Christoffersson et al., 1986; Wielopolski et al., 1986; Nilsson
et al., 1991) for the determination in situ of the body burden of
lead.
2.2.2.3 Analytical procedures for biomarkers of lead exposure and
effect
Using standard clinical laboratory techniques, analytical
procedures have been developed: delta-aminolaevulinic acid (ALA);
delta-aminolaevulinic acid dehydratase (ALAD); urinary coproporphyrin
(CPU) and erythrocyte protoporphyrin (EP). All of these assays are
well established and reliable (Grandjean & Olsen 1984; US EPA, 1986a).
These biochemical parameters are influenced by physiological factors
other than lead. They lack the specificity and sensitivity of PbB
measurements as an index of either current lead exposures or body
stores of lead.
2.3 Conversion factors
1 µg/dl = 0.048 µmol/litre
1 µmol/litre = 20.7 µg/dl
Using the above conversion factor, blood lead concentrations are
given as µmol/litre with the equivalent µg/dl in brackets. Calculated
figures have not been rounded and added precision is not to be
inferred from the number of significant figures.
Table 2. Analytical methods for determining lead in environmental samplesa
Sample type Preparation method Analytical method Sample detection Percentage Reference
limit recovery
Air collect particulate matter on membrane ASV with mercury-graphite 0.16 µg/m3 90-110 NIOSH (1977b)
(particulate filter; wet ash with HNO3/HClO4/H2SO4; electrode (NIOSH method
lead) dissolve in acetate buffer P&CAM 191)
Air collect particulate matter on cellulose ICP-AES (NIOSH method 0.34 µg/m3 95-105 NIOSH (1981)
(particulate acetate filter; wet ash with HNO3/HClO4 P&CAM 351)
lead)
Air collect particulate matter on filter; AAS 0.1 µg/m3 93 Scott et al.
(particulate dry ash; extract with HNO3/HCl; dilute AES 0.15 µg/m3 102 (1976)
lead) with HNO3
Air sample on cellulose acetate filter; AAS 8 ng/litre 100-101 Nerin et al.
(particulate dissolve in HNO3 with heat; add HCl/H2O2 (1989)
lead) and react in hydride generator with sodium
borohydride to generate lead hydride
Air collect sample on filter; spike filter with IDMS 0.1 ng/m3 NR Volkening et
(particulate 206Pb; dissolve filter in NaOH; acidify; al. (1988)
lead) separate lead by electrodeposition; dissolve
in acid
Water digest sample with acid; heat; dilute with AAS 1.0 ng/g NR Chau et al.
(total lead) water (1979)
Soil dry sample and sieve for XRF; digest sieved XRF NR 65-98 Krueger &
sample with HNO3 and heat for AAS AAS NR 63-68 Duguay (1989)
Soil dry sample, dry ash; digest with acid AAS 2 µg/g 79-103 Beyer &
and dilute with water Cromartie (1987)
Table 2 (cont'd)
Sample type Preparation method Analytical method Sample detection Percentage Reference
limit recovery
Soil, waste, digest sample with acid; dilute with water AAS (EPA method 7420) 0.1 mg/litre NR US EPA (1986b)
and ground and filter GFAAS (EPA method 7421) 1 µg/litre NR
water
Soil, dust digest sample with hot acid; dry; redissolve AAS 12 ng/g > 80 Que Hee et al.
and paint in HNO3 (1985b)
Sediment, fish, digest sample with acid; heat; dilute with AAS 50 ng/g NR Chau et al.
vegetation water (sediment) (1980)
(total lead) 10 ng/g (fish NR
and vegetation)
Milk add 50 µl (C2H5)4NOH in ethanol to 25 µl GFAAS NR NR Michaelson &
milk; heat and dilute with water to 125 µl Sauerhoff (1974)
Evaporated dry ash sample; dissolve in HNO3 ASV 0.005 µg/g 99 Capar & Rigsby
milk (1989)
Agricultural dry ash sample with H2SO4 and HNO3; DPASV 0.4 ng/g 85-106 Satzger et al.
crops dilute with water (1982)
Grains, milk, bomb digest sample with acid; heat or GFAAS 20 µg/g (bomb) 85-107 Ellen & Van
mussels, fish digest with acid and dry ash; dissolve 5 µg/g (dry ash) 75-107 Loon (1990)
in acid; dilute with water DPASV NR 82-120
Table 2 (cont'd)
Sample type Preparation method Analytical method Sample detection Percentage Reference
limit recovery
Citrus leaves chop or pulverize sample; digest with hot ICP-AES 10-50 µg/litre 75-82 Que Hee &
and paint acid; dry; redissolve in acid (citrus Boyle (1988)
leaves)
89-96
(paint)
a AAS = atomic absorption; AES = atomic emissions spectroscopy; ASV = anode stripping voltametry; (C2H5)4NOH = tetraethylammonium
hydroxide; DPASV = differential pulse anodic stripping voltametry; EPA = US Environmental Protection Agency; GFAAS = graphite furnace
atomic absorption spectrometry; HCl = hydrochloric acid; HClO4 = perchloric acid; HNO3 = nitric acid; H2O2 = hydrogen peroxide;
H2SO4 = sulfuric acid; ICP-AES = inductively coupled plasma/atomic emission spectroscopy; IDMS = isotope dilution mass spectrometry;
NaOH = sodium hydroxide; NIOSH = National Institute for Occupational Safety and Health; NR = not reported; XRF = X-ray fluorescence
Table 3. Analytical methods for determining lead in biological materialsa
Sample Preparation method Analytical method Sample detection Percentage Reference
type limit recovery
Blood wet ash sample with acid mixtures; ASV with mercury-graphite 0.192 µmol/litre 95-105 NIOSH (1977c)
dissolve residue in dilute HClO4 electrode (NIOSH method (4 µg/dl)
P&CAM 195)
Blood wet ash sample with HNO3; dissolve GFAAS (NIOSH method 0.48 µmol/litre NR NIOSH (1977e)
residue in dilute HNO3 P&CAM 214) (10 µg/dl)
Blood dilute sample with Triton X-100(R); add GFAAS 0.011 µmol/litre 93-105 Aguilera de
nitric acid and diammonium phosphate (0.24 µg/dl) Benzo et al.
(1989)
Blood dilute sample with ammonia solution ICP-MS 0.072 µmol/litre 96-111 Delves &
containing Triton X-100(R); analyse (1.5 µg/dl) Campbell
(1988)
Blood dilute sample in 0.2% Triton X-100(R) GFAAS approx. 0.072 97-150 Que Hee et
and water; analyse µmol/litre (approx. al. (1985a)
1.5 µg/dl)
Blood and wet ash sample with HNO3, complex Spectrophotometry 0.144 µmol/litre 97 NIOSH (1977a)
urine with dephenylthiocarbazone and (NIOSH method (3.0 µg/dl) (blood);
extract with chloroform P&CAM 102) 0.0576 µmol/litre 97
(12 µg/litre) (urine)
Serum, filter sample if needed; dilute with ICP-AES 0.048-0.240 µmol/litre 85 (serum) Que Hee &
blood and acid or water (1.0-5.0 µg/dl) Boyle (1988)
urine
Urine wet ash sample with acid mixture and ASV with mercury-graphite 0.0192 µmol/litre 90-110 NIOSH (1977d)
dissolve in dilute HClO4 electrode (NIOSH method (4 µg/litre)
P&CAM 200)
Table 3 (cont'd)
Sample Preparation method Analytical method Sample detection Percentage Reference
type limit recovery
Liver, bomb digest sample with acid and heat, GFAAS 20 µg/g (bomb); 85-107 Ellen & Van
kidney, or digest with acid and dry ash; dissolve (bomb); Loon (1990)
muscle in acid; dilute with water 5 µg/g (dry ashing) 75-107 (dry
ashing)
Bone direct partially polarized photons at XRF 20 µg/g NR Christoffersson
second phalanx of left forefinger et al. (1986)
(non-invasive technique)
Bone direct partially polarized photons at XRF 20 µg/g NR Wielopolski
anteromedial skin surface of mid-tibia et al. (1986)
(non-invasive technique)
Teeth clean and section tooth; digest with ASV NR 83-114 Rabinowitz
HNO3; evaporate; redissolve in buffer et al. (1989)
solution
Teeth dry ash sample; crush; dry ash again; AAS NR 90-110 Steenhout &
dissolve in HNO3 Pourtois
(1981)
a AAS = atomic absorption spectrometry; ASV = anode stripping voltametry; GFAAS = graphite furnace atomic absorption spectrometry;
HClO4 = perchloric acid; HNO3 = nitric acid; ICP-AES = inductively coupled plasma-atomic emission spectroscopy;
ICP-MS = inductively coupled plasma-mass spectrometry; NIOSH = National Institute for Occupational Safety and Health;
NR = not reported; XRF = X-ray fluorescence
3. SOURCES OF HUMAN AND ENVIRONMENTAL EXPOSURE
3.1 Natural occurrence
Because lead is relatively abundant in the earth's crust it is
found naturally throughout the world. The major natural sources of
lead are volcanic emissions, geochemical weathering, and emissions
from sea spray. A small amount of radioisotopic lead (207Pb) is
derived from the decay of radon gas released from geological sources.
It has been estimated that the worldwide natural emission rates of
lead are of the order of 19 000 tonnes/year (Nriagu & Pacyna, 1988),
with volcanic sources accounting for 6400 tonnes/year (Nriagu, 1979).
Owing to centuries of human exploitation of lead resources, it is
difficult to determine the natural content of lead in most ecosystems.
Data on environmental levels, uses, and sources of lead have been
summarized in a recent review (OECD, 1993).
3.1.1 Rocks and soils
The average concentration of lead in the earth's crust is between
10 and 20 mg/kg (IPCS, 1989). The major geological sources of lead are
in igneous and metamorphic rocks.
The soil is the most important repository in terrestrial
ecosystems for contaminants of anthropogenic origin (Nriagu & Pacyna,
1988; Nriagu, 1989). The lead content of soils (which are for
discussion purposes distinguished here from surface dusts) is greatly
influenced by anthropogenic activities and by long- and short-range
airborne transport of lead from various sources. Both dry and wet
deposition are important routes of input.
Lead in soil may be relatively insoluble (as a sulfate, carbonate
or oxide), soluble, adsorbed onto clays, adsorbed and coprecipitated
with sesquioxides, adsorbed onto colloidal organic matter, or
complexed with organic moieties in soil (US EPA, 1986a; IPCS, 1989).
Soil pH, content of humic and fulvic acids, and amount of organic
matter influence the content and mobility of lead in soils. Since
acidic conditions favour the solubilization and leaching of lead from
the solid phase, acidic soils tend to have lower lead concentrations
when analysed as dry soil. Humic and fulvic acids can also mobilize
lead, and certain complex organic molecules can act as chelators of
lead (IPCS, 1989).
Background levels of lead in soil are in the range of 10-70 mg/kg
(GEMS, 1985). Similar results have been found in studies of mobile
source contamination near highways; soil taken at distances of
50-100 m from highways (outside the range of immediate impact from
traffic emissions) usually shows levels of lead below 40 mg/kg. In the
1985, GEMS survey of selected countries, lead concentrations in
topsoil from Malta were found to have a mean of 54 mg/kg in areas at
least 5 m from roadways; less than one metre from roadways the mean
concentration was 138 mg/kg. A 1977 report from Sweden found a mean of
16 mg/kg in non-contaminated areas (GEMS, 1985).
3.1.2 Sediments
Sediments from freshwater and marine environments have been
studied for lead content. This compartment provides a unique record of
the history of changes in global lead fluxes (Patterson, 1983). Levels
of lead in sediments dated before the onset of the industrial
revolution in Western Europe show very low levels, less than 10% of
current levels (Flegal et al., 1987). The average background level of
lead in marine sediments off southern California was reported by
Flegal et al. (1987) to be 1.3 mg/kg.
3.1.3 Water
Flegal et al. (1987) estimate that the natural concentration of
lead in surface water is about 0.02 µg/litre. In general, lead is not
found in ground or surface waters at concentrations above 10 µg/litre
(IPCS, 1989).
Data from oceans indicate very low levels of lead in sea-water
samples not affected directly by significant sources of lead. Water
samples taken from an area of the Pacific, where annual windborne-
input fluxes of lead are estimated to be 3 mg/cm2, have lead
concentrations of 3.5 ng/litre (0-100 m depth) and 0.9 ng/litre at
depths greater than 2500 m. In contrast, water samples taken from the
north Atlantic, where annual windborne-input fluxes of lead are
170 mg/cm2, contain 34 ng lead/litre at the surface and 5 ng/litre
in depths below 2500 m (Patterson, 1983). Settle & Patterson (1980)
have estimated that prehistoric oceans contained 0.5 ng/litre lead.
Flegal et al. (1987) have estimated that over 95% of the lead in
off-shore surface waters is the result of windborne inputs. However,
in coastal waters near Monterey (California, USA), higher
concentrations of lead were found in sea water, sediments and
organisms; these elevations were related to specific sources by
systematic isotope analyses (Flegal et al., 1987).
3.1.4 Air
Anthropogenic inputs of lead from a range of sources have
resulted in global dispersion of both inorganic and organic species of
lead into the air, of which 80-90% is derived from alkyllead fuel
additives (WHO, 1987). Nriagu & Pacyna (1988) estimated that a total
of 330 000 tonnes of lead is discharged directly into the atmosphere
each year. Estimations of pre-industrial levels of lead in air from
natural origins (volcanic emissions, crustal weathering, radon decay
and sea-spray releases) are in the range of 0.01-0.1 µg/m3 (US NRC,
1980). The lowest level reported since 1975 is 0.076 ng/m3 measured
at the South Pole (US EPA, 1986a).
3.1.5 Plants
Lead occurs naturally in plants and results from both deposition
and uptake. There is a positive linear relationship between lead
concentrations in plants and soil (Davies & Thornton, 1989). As with
other environmental compartments, measurement of "background" levels
of lead in plants is complicated by the general contamination of the
globe from centuries of lead use, which has included direct
application of lead-containing chemicals in agriculture (see below)
and contamination of fertilizers with lead. Lead has been measured in
superphosphate fertilizer at concentrations as high as 92 mg/kg (Lisk,
1972). Sewage sludge, used as a source of nutrients in agriculture,
may contain even higher levels of lead. The concentration of lead in
sewage sludge is typically < 1000 mg/kg. Levels as high as 26 g/kg
have been measured in the USA (Chaney et al., 1984). Soil receiving
heavy sludge applications over long periods of time (years) contained
425 mg lead/kg; the concentration in untreated soil was 47 mg/kg
(Beckett, 1979).
3.1.6 Environmental contamination from natural sources
The contribution of natural sources of lead to human exposure is
small. As a result of various breakdown processes, rocks yield lead
which is transferred to the biosphere and the atmosphere, and,
ultimately, back to the earth's crust in the form of sedimentary
rocks. Soluble lead has for thousands of years entered the oceans with
river discharges, and the rate has been estimated by Patterson (1965)
to be around 17 000 tonnes/year. Sources contributing to airborne lead
are silicate dusts, volcanic halogen aerosols, forest fires, sea salts
aerosols, meteoric and meteorite residues, and lead derived from the
decay of radon. While the lead content of most coals is relatively
low, coal fly ash is enriched in lead (Hutton et al., 1988) and is a
source of environmental contamination.
3.2 Anthropogenic sources
World lead consumption has steadily increased over the period
1965-1990 and was about 5.6 × 106 tonnes in 1990 (OECD, 1993).
Further review of the data summarized by OECD (1993) indicates a
change in consumption patterns worldwide. Although the consumption of
lead within the 24 countries of the OECD increased only slightly over
the decade from 1980 to 1990, consumption within less developed
economies (Africa and Asia) increased from 315 000 tonnes in 1970 to
844 000 tonnes in 1990.
3.2.1 Lead mining
Lead occurs in a variety of minerals, the most important of which
are galena (PbS), cerrusite (PbCO3) and anglesite (PbSO4). Galena
is by far the most important source of primary lead. It occurs mostly
in deposits associated with other minerals, particularly those
containing zinc. Mixed lead and zinc ores account for about 70% of
total primary lead supplies. Ores containing mainly lead account for
about 20% and the remaining 10% is obtained as a by-product from other
deposits, such as mixed copper-zinc deposits. The proportions of
various metals may differ in the ores of different countries. Silver
is the most important of the other metals frequently present in lead
deposits, but copper may also be present in concentrations high enough
to be commercially important. Other minor constituents of lead ores
are gold, bismuth, antimony, arsenic, cadmium, tin, gallium, thallium,
indium, germanium and tellurium.
The major countries producing lead from mining activity during
1987-1991 were the USA, Canada, Australia, Peru, the former USSR and
Mexico, as shown in Table 4. Other countries producing lead from lead
ores include China, the former Yugoslavia, Morocco, Spain, Sweden and
Tunisia. In general, the level of world production of lead from mining
activities has remained relatively constant at about 3.3 × 106
tonnes between 1988 and 1991 (ILZSG, 1992); this represents roughly
60% of the world demand for lead.
Table 4. Major countries producing lead from ore and ore
concentratesa
Country 1987 1988 1989 1990 1991
Canada 423 200 366 600 276 100 241 300 278 100
USA 318 300 395 700 419 300 495 200 483 300
Ex-USSR 510 000 520 000 500 000 490 000 --
Australia 489 200 462 000 495 000 570 000 579 000
Mexico 177 200 178 100 163 000 174 100 158 800
Peru 204 000 149 000 192 200 187 800 199 100
a From: World Bureau of Metal Statistics (1992)
3.2.2 Smelting and refining
Smelting and refining are classified as either primary or
secondary, the former producing refined lead products from ores or
concentrates (primary lead) and the latter producing lead by
recovering it from lead-bearing scrap and waste materials (secondary
lead). Secondary lead is derived from processing what is termed new
scrap arising during manufacturing processes and recycled old scrap
arising from waste materials containing lead. Most scrap is from old
sources, of which the most important are lead plates from batteries,
solder, common babbitt, soft lead, lead solders, cable coverings, type
metals, dross and other lead-containing products. There has been an
increasing contribution of secondary lead sources to the total
worldwide production of lead, as shown in Table 5 (World Bureau of
Metal Statistics, 1992). Secondary sources of lead supplied between 35
and 40% of world production during the period from 1970 to 1990.
3.2.3 Environmental pollution from production of lead
Mining operations and the smelting and refining of both primary
and secondary lead are known to cause contamination of the nearby
environment. The nature and extent of contamination depends on many
factors, including the level of production, the effectiveness of
emission controls, climate, topography and other local factors.
Concentrations are usually highest within 3 km of the point source (US
EPA, 1989). A report from China found that lead levels in ambient air,
plants and soil increased proportionally with proximity to a large
primary smelter; at 50 m from the source, the air lead level was
60 µg/m3, the lead level in plants was 29.1 mg/kg, and soil lead
level was 170 mg/kg (Wang, 1984). However, some earlier studies have
shown air pollution and soil contamination as far as 10 km from
smelters (Djuric et al., 1971; Kerin, 1973; Landrigan et al., 1975).
Table 5. Relative contribution of primary and secondary sources
relative to world lead production (1987-1990)a
1987 1988 1989 1990
Primary 422 100 3 414 200 3 286 500 3 324 500
Secondary 2 045 600 2 103 900 2 272 900 2 254 800
a From: World Bureau of Metal Statistics (1992)
The impacts of lead mining and smelting can persist for long
periods of time. A study conducted in Wales, United Kingdom, in an
area where lead mining began 2000 years ago and ended in the middle of
the 20th century, found high concentrations of lead in soils (Davies
et al., 1985). In Port Pirie, Australia, a community with one of the
world's largest and oldest primary lead smelters, lead levels in soils
were found to be grossly elevated, and the incidence of elevated blood
lead levels in pregnant women and young children was also increased
above that found in other communities in Australia (Wilson et al.,
1986).
3.3 Consumption and uses of lead and its compounds
Lead has a combination of physical and chemical properties that
have made it extremely useful industrially, i.e. high density, high
opacity to gamma and X-ray energies, low sound conductance, a low
melting point, exceptional malleability, high corrosion resistance,
and stability. In 1990, 5.627 × 106 tonnes of lead were consumed
worldwide (ILZSG, 1992). The twenty-four industrialized countries of
the OECD consumed approximately 65% of this amount, with eastern
Europe and the former USSR using 21%. Asia now utilizes about 9% of
the world's lead production.
The use patterns of refined lead vary from country to country.
The situation in 1990 in Mexico is shown in Table 6, while end-use
categories within OECD countries are summarized in Fig. 1, which
indicates the changes between 1970 and 1990 (OECD, 1993).
Table 6. Principal uses of refined lead in Mexicoa
Type of product 1988 1990
(%) (%)
Oxides 69.7 56.7
Batteries 9.2 17.9
Tetraethyllead 7.9 11.9b
Cables 4.0 1.5
Others 9.2 11.9
a ILZSG (1992)
b This does not reflect the introduction of lead-free petrol
in 1990.
From Fig. 1, it is evident that the largest use of lead within
OECD countries is for battery production, whereas there has been a
large drop in the demand for lead-containing gasoline additives.
However, this pattern is not valid worldwide, e.g., concentrations in
petrol range from zero in such countries as Japan and Thailand to
1.12 g/litre in the Virgin Islands (Octel, 1991).
In the past the use of lead in the chemical industry for the
preparation of paints, pigments and coloured inks was widespread. Many
countries have now restricted this use, and concentrations of lead
greater than 0.06% (USA) and 0.5% (New Zealand) are not permitted in
indoor paints (Albert & Badillo, 1991; OECD, 1993). In 1982, data from
the United Kingdom (UK, 1982) indicated levels of lead between 2500
and 3000 mg/kg in decorative glass paints and up to 448 g/kg in
white-lead primer. Red-leadcontaining paints, still used widely to
paint structural steel works, can contain up to 661 g lead/kg.
It is accepted that the following criteria should initiate the
updating of an EHC monograph: new data are available that would
substantially change the evaluation; there is public concern for
health or environmental effects of the agent because of greater
exposure; an appreciable time period has elapsed since the last
evaluation.
All Participating Institutions are informed, through the EHC
progress report, of the authors and institutions proposed for the
drafting of the documents. A comprehensive file of all comments
received on drafts of each EHC monograph is maintained and is
available on request. The Chairpersons of Task Groups are briefed
before each meeting on their role and responsibility in ensuring that
these rules are followed.
WHO TASK GROUP ON ENVIRONMENTAL HEALTH CRITERIA FOR INORGANIC LEAD
Members
Professor S. Araki, Department of Public Health, Faculty of Medicine,
University of Tokyo, Japan
Dr P. Baghurst, Division of Human Nutrition, Commonwealth Scientific
Industrial Research Organization, Adelaide, Australia
Dr D. Bellinger, Neuroepidemiology Unit, Gardner House, Children's
Hospital, Boston, Massachusetts, USA
Dr I. Calder, Occupational and Environmental Health, South Australian
Health Commission, Adelaide, South Australia, Australia
Dr D.A. Cory-Slechta, Department of Environmental Medicine,
University of Rochester School of Medicine and Dentistry,
Rochester, New York, USA
Dr K. Dietrich, Department of Environmental Health, Division of
Biostatistics and Epidemiology, University of Cincinnati College
of Medicine, Cincinnati, Ohio, USA
Dr R.A. Goyer, Chapel Hill, North Carolina, USA (Chairman)
Dr M.R. Moore, University of Glasgow, Department of Medicine and
Therapeutics, Western Infirmary, Glasgow, Scotland
Dr C. Nam Ong, Department of Community, Occupational and Family
Medicine, National University of Singapore, National University
Hospital, Singapore
Dr S.J. Pocock, Department of Epidemiology and Population Sciences,
Medical Statistics Unit, University of London, London, England
Dr M.B. Rabinowitz, Marine Biological Laboratory, Woods Hole,
Massachusetts, USA
Dr M. Smith, Thomas Coram Research Unit, London, England
Dr G. Winneke, Medical Institute for Environmental Health,
Heinrich-Heine University, Düsseldorf, Germany (Vice-Chairman)
Observers
Dr C. Boreiko, Environmental Health, International Lead Zinc Research
Organization (ILZRO) Inc., Research Triangle Park, North
Carolina, USA
Dr N.H. Clark, Lead Industry Environment and Health Forum, Melbourne,
Victoria, Australia
Dr J.M. Davis, Environmental Criteria and Assessment Office, US
Environmental Protection Agency, Research Triangle Park, North
Carolina, USA
Professor G. Duggin, Toxicology Unit, Royal Prince Alfred Hospital,
Camperdown, Australia
Dr G.R. Neville, Queensland Health Department, Brisbane, Australia
Secretariat
Dr G.C. Becking, International Programme on Chemical Safety,
Interregional Research Unit, World Health Organization, Research
Triangle Park, North Carolina, USA (Secretary)
Dr K.R. Mahaffey, National Institute of Environmental Health
Sciences, Research Triangle Park, North Carolina, USAa
Dr A.E. Robinson, Toronto, Ontario, Canada (Rapporteur)
a Present address: US Environmental Protection Agency, Environmental
Criteria and Assessment Office, Cincinnati, Ohio, USA
ENVIRONMENTAL HEALTH CRITERIA FOR INORGANIC LEAD
A WHO Task Group on Environmental Health Criteria for Inorganic
Lead met in Brisbane, Australia, from 1 to 6 February 1993. The
meeting was sponsored by a consortium of Australian Commonwealth and
State Governments through a national Steering Committee chaired by
Dr Keith Bentley, Director, Health and Environmental Policy,
Department of Human Services and Health, Canberra. The meeting was
hosted and organized by the Queensland Department of Health,
Dr G.R. Neville being responsible for the arrangements. Dr G. Murphy,
Director of Public Health, Queensland, welcomed the participants on
behalf of the Organizers, and Dr T. Adams, Chief Commonwealth Medical
Advisor and Dr G. Johns, Parliamentary Secretary to Federal Minister
for Health, Housing and Community Services, welcomed the participants
on behalf of the Commonwealth Government. Dr Johns stressed the
importance attached to this IPCS meeting by the Commonwealth and State
Governments of Australia. Dr G.C. Becking, IPCS, welcomed the
participants on behalf of Dr M. Mercier, Director of the IPCS and the
three cooperating organizations (UNEP/ILO/WHO). The Task Group
reviewed and revised the draft criteria monograph, and made an
evaluation of the risks to human health from exposure to inorganic
lead.
The Task Group draft was prepared by Dr A.E. Robinson, Toronto,
Canada, using texts made available by Dr K.R. Mahaffeya (National
Institute of Environmental Health Sciences, Research Triangle Park,
North Carolina, USA) and Dr E. Silbergeld (University of Maryland
School of Medicine, Baltimore, Maryland, USA), and the comments
received from the IPCS contact points for environmental health
criteria monographs. The draft was revised extensively by the Task
Group taking into account the comments from the IPCS contact points.
Dr G.C. Becking (IPCS Central Unit, Interregional Research Unit)
and Dr P.G. Jenkins (IPCS Central Unit, Geneva) were responsible for
the overall scientific content and technical editing, respectively, of
this monograph.
The efforts of all who helped in the preparation and finalization
of this publication are gratefully acknowledged.
a Present address: US Environmental Protection Agency, Environmental
Criteria and Assessment Office, Cincinnati, Ohio, USA
ABBREVIATIONS
AAS atomic absorption spectrometry
AES atomic emission spectroscopy
ALA delta-aminolaevulinic acid
ALAD delta-aminolaevulinic acid dehydratase
ASV anodic stripping voltametry
EDTA ethylenediaminetetraacetic acid
FEP free erythrocyte porphyrin
GFAAS graphite furnace atomic absorption spectrometry
ICP inductively coupled plasma
IDMS isotope dilution mass spectrometry
MPb mobilization yield of lead
MSW municipal solid waste
PbB blood lead
PbT tooth lead
TML tetramethyllead
XRFS X-ray fluorescence spectroscopy
ZPP zinc protoporphyrin
PREFACE
Although many countries have initiated programmes to lower the
level of lead in the environment, human exposure to lead remains of
concern to public health officials worldwide. For over 20 years the
World Health Organization (WHO) and the International Programme on
Chemical Safety (IPCS) have been concerned about the health and
environmental effects of the levels of inorganic lead in the
environment. The evaluation of human health risks arising from
food-borne lead has been carried out by WHO on four occasions since
1972. In addition, health-based guidance values for lead in water, air
and the workplace have been developed by various Task Groups convened
by WHO. Environmental Health Criteria 3: Lead, published in 1977,
examined the effects of lead on human health and Environmental Health
Criteria 85: Lead - Environmental Aspects was published in 1989.
Since the publication of Environmental Health Criteria 3: Lead, a
large body of knowledge has accumulated concerning the effects of lead
on humans at low levels of exposure. Studies have emphasized the
effects of inorganic lead on infants and children, a high-risk
population. This monograph on inorganic lead reflects this research
emphasis; a major part of the monograph deals with the neurotoxic
effects of lead with emphasis on neurobehavioural development in
children. Less detail is presented on the health effects of the higher
levels of inorganic lead found in some workplaces, although such
exposures are still considered to pose a risk to humans in many
regions of the world.
This monograph deals only with the human health effects of
inorganic lead. No attempt has been made to evaluate the human health
effects of organo-lead compounds, although it was recognized that such
compounds when added to petrol (gasoline) are a major source of
inorganic lead in the environment. In view of the toxicity of many
organo-lead derivatives and the possible methylation of inorganic lead
in the environment, the IPCS plans to evaluate the risk to humans from
exposure to organo-lead compounds in a separate monograph.
As with all IPCS criteria monographs, no attempt has been made to
prepare an exhaustive bibliography of the extremely large amount of
lead-related literature published since 1977. Rather, an effort has
been made to review critically the studies on humans and experimental
animals that are essential for the evaluation of risks to human health
from exposure to all sources of inorganic lead.
1. SUMMARY
This monograph focuses on the risks to human health associated
with exposure to lead and inorganic lead compounds. Emphasis has been
given to data which have become available since the publication of
Environmental Health Criteria 3: Lead (IPCS, 1977). The environmental
effects of lead are discussed in Environmental Health Criteria 85:
Lead - Environmental Aspects (IPCS, 1989).
1.1 Identity, physical and chemical properties, and analytical
methods
Lead is a soft, silvery grey metal, melting at 327.5°C. It is
highly resistant to corrosion, but is soluble in nitric and hot
sulfuric acids. The usual valence state in inorganic lead compounds is
+2. Solubilities in water vary, lead sulfide and lead oxides being
poorly soluble and the nitrate, chlorate and chloride salts are
reasonably soluble in cold water. Lead also forms salts with such
organic acids as lactic and acetic acids, and stable organic compounds
such as tetraethyllead and tetramethyllead.
The most commonly used methods for the analysis of low
concentrations of lead in biological and environmental materials are
flame, graphite furnace and inductively coupled plasma atomic
absorption spectroscopy and anode stripping voltametry. Depending on
sample pretreatment, extraction techniques and analytical
instrumentation, detection limits of 0.12 µmoles lead/litre blood
(2.49 µg/dl) can be achieved. However, reliable results are obtained
only when specific procedures are followed to minimize the risk of
contamination during sample collection, storage, processing and
analysis.
1.2 Sources of human exposure
The level of lead in the earth's crust is about 20 mg/kg. Lead in
the environment may derive from either natural or anthropogenic
sources. Natural sources of atmospheric lead include geological
weathering and volcanic emissions and have been estimated at
19 000 tonnes/year, compared to an estimate of 126 000 tonnes/year
emitted to the air from the mining, smelting and consumption of over 3
million tonnes of lead per year.
Atmospheric lead concentrations of 50 pg/m3 have been found in
remote areas. Background levels of lead in soil range between 10 and
70 mg/kg and a mean level near roadways of 138 mg/kg has been
reported. Present levels of lead in water rarely exceed a few
micrograms/litre; the natural concentration of lead in surface water
has been estimated to be 0.02 µg/litre.
Lead and its compounds may enter the environment at any point
during mining, smelting, processing, use, recycling or disposal. Major
uses are in batteries, cables, pigments, petrol (gasoline) additives,
solder and steel products. Lead and lead compounds are also used in
solder applied to water distribution pipes and to seams of cans used
to store foods, in some traditional remedies, in bottle closures for
alcoholic beverages and in ceramic glazes and crystal tableware. In
countries where leaded petrol is still used, the major air emission is
from mobile and stationary sources of petrol combustion (urban
centres). Areas in the vicinity of lead mines and smelters are subject
to high levels of air emissions.
Airborne lead can be deposited on soil and water, thus reaching
humans through the food chain and in drinking-water. Atmospheric lead
is also a major source of lead in household dust.
1.3 Environmental transport, distribution and transformation
The transport and distribution of lead from fixed, mobile and
natural sources are primarily via air. Most lead emissions are
deposited near the source, although some particulate matter (< 2 µm
in diameter) is transported over long distances and results in the
contamination of remote sites such as arctic glaciers. Airborne lead
can contribute to human exposures by the contamination of food, water
and dust, as well as through direct inhalation. The removal of
airborne lead is influenced by atmospheric conditions and particulate
size. Large amounts of lead may be discharged to soil and water.
However, such material tends to remain localized because of the poor
solubility of lead compounds in water.
Lead deposited in water, whether from air or through run-off from
soils, partitions rapidly between sediment and aqueous phase,
depending upon pH, salt content, and the presence of organic chelating
agents. Above pH 5.4, hard water may contain about 30 µg lead/litre
and soft water about 500 µg lead/litre. Very little lead deposited on
soil is transported to surface or ground water except through erosion
or geochemical weathering; it is normally quite tightly bound
(chelated) to organic matter.
Airborne lead can be transferred to biota directly or through
uptake from soil. Animals can be exposed to lead directly through
grazing and soil ingestion or by inhalation. There is little
biomagnification of inorganic lead through the food chain.
1.4 Environmental levels and human exposure
In the general non-smoking adult population, the major exposure
pathway is from food and water. Airborne lead may contribute
significantly to exposure, depending upon such factors as use of
tobacco, occupation, proximity to motorways, lead smelters, etc., and
leisure activities (e.g., arts and crafts, firearm target practice).
Food, air, water and dust/soil are the major potential exposure
pathways for infants and young children. For infants up to 4 or 5
months of age, air, milk, formulae and water are the significant
sources of lead exposure.
Levels of lead found in air, food, water and soil/dust vary
widely throughout the world and depend upon the degree of industrial
development, urbanization and lifestyle factors. Ambient air levels
over 10 µg/m3 have been reported in urban areas near a smelter,
whereas lead levels below 0.2 µg/m3 have been found in cities where
leaded petrol is no longer used. Lead intake from air can, therefore,
vary from less than 4 µg/day to more than 200 µg/day.
Levels of lead in drinking-water sampled at the source are
usually below 5 µg/litre. However, water taken from taps (faucets) in
homes where lead is present in the plumbing can contain levels in
excess of 100 µg/litre, particularly after the water has been standing
in the pipes for some hours.
The level of dietary exposure to lead depends upon many lifestyle
factors, including foodstuffs consumed, processing technology, use of
lead solder, lead levels in water, and use of lead-glazed ceramics.
For infants and children, lead in dust and soil often constitutes
a major exposure pathway. Lead levels in dust depend upon such factors
as the age and condition of housing, the use of lead-based paints,
lead in petrol and urban density. The intake of lead will be
influenced by the age and behavioural characteristics of the child and
bioavailability of lead in the source material.
Inhalation is the dominant pathway for lead exposure of workers
in industries producing, refining, using or disposing of lead and lead
compounds. During an 8-h shift, workers can absorb as much as 400 µg
lead, in addition to the 20-30 µg/day absorbed from food, water and
ambient air; significant intake may occur from ingestion of large
inhaled particulate material.
1.5 Kinetics and metabolism in laboratory animals and humans
Lead is absorbed in humans and animals following inhalation or
ingestion; percutaneous absorption is minimal in humans. Depending
upon chemical speciation, particle size, and solubility in body
fluids, up to 50% of the inhaled lead compound may be absorbed. Some
inhaled particulate matter (larger than 7 µm) is swallowed following
mucociliary clearance from the respiratory tract. In experimental
animals and humans, absorption of lead from the gastrointestinal tract
is influenced by the physico-chemical nature of the ingested material,
nutritional status, and type of diet consumed. In adult humans
approximately 10% of the dietary lead is absorbed; the proportion is
higher under fasting conditions. However, in infants and young
children as much as 50% of dietary lead is absorbed, although
absorption rates for lead from dusts/soils and paint chips can be
lower depending upon the bioavailability. Diets that are deficient in
calcium, phosphate, selenium or zinc may result in increased lead
absorption. Iron and vitamin D also affect absorption of lead.
Blood lead (PbB) levels are used as a measure of body burden and
absorbed (internal) doses of lead. The relationship between blood lead
and the concentration of lead in exposure sources is curvilinear.
Once it has been absorbed, lead is not distributed homogeneously
throughout the body. There is rapid uptake into blood and soft tissue,
followed by a slower redistribution to bone. Bone accumulates lead
over much of the human life span and may serve as an endogenous source
of lead. The half-life for lead in blood and other soft tissues is
about 28-36 days, but it is much longer in the various bone
compartments. The percentage retention of lead in body stores is
higher in children than adults. Transfer of lead to the human fetus
occurs readily throughout gestation.
Blood lead is the most commonly used measure of lead exposure.
However, techniques are now available for measuring lead in teeth and
bone, although the kinetics are not fully understood.
1.6 Effects on laboratory animals and in vitro systems
In all species of experimental animals studied, including
non-human primates, lead has been shown to cause adverse effects in
several organs and organ systems, including the haematopoietic,
nervous, renal, cardiovascular, reproductive and immune systems. Lead
also affects bone and has been shown to be carcinogenic in rats and
mice.
Despite kinetic differences between experimental animal species
and humans, these studies provide strong biological support and
plausibility for the findings in humans. Impaired learning/memory
abilities have been reported in rats with PbB levels of
0.72-0.96 µmoles/litre (15-20 µg/dl) and in non-human primates at PbB
levels not exceeding 0.72 µmoles/litre (15 µg/dl). In addition, visual
and auditory impairments have been reported in experimental animal
studies.
Renal toxicity in rats appears to occur at a PbB level in excess
of 2.88 µmol/litre (60 µg/dl), a value similar to that reported to
initiate renal effects in humans. Cardiovascular effects have been
seen in rats after chronic low-level exposures resulting in PbB levels
of 0.24-1.92 µmol/litre (5-40 µg/dl). Tumours have been shown to occur
at dose levels below the maximum tolerated dose of 200 mg lead (as
lead acetate) per litre of drinking-water. This is the maximum dose
level not associated with other morphological or functional changes.
1.7 Effects on humans
In humans, lead can result in a wide range of biological effects
depending upon the level and duration of exposure. Effects at the
subcellular level, as well as effects on the overall functioning of
the body, have been noted and range from inhibition of enzymes to the
production of marked morphological changes and death. Such changes
occur over a broad range of doses, the developing human generally
being more sensitive than the adult.
Lead has been shown to have effects on many biochemical
processes; in particular, effects on haem synthesis have been studied
extensively in both adults and children. Increased levels of serum
erythrocyte protoporphyrin and increased urinary excretion of
coproporphyrin and delta-aminolaevulinic acid are observed when PbB
concentrations are elevated. Inhibition of the enzymes
delta-aminolaevulinic acid dehydratase and dihydrobiopterin reductase
are observed at lower levels.
The effects of lead on the haemopoietic system result in
decreased haemoglobin synthesis, and anaemia has been observed in
children at PbB concentrations above 1.92 µmol/litre (40 µg/dl).
For neurological, metabolic and behavioural reasons, children are
more vulnerable to the effects of lead than adults. Both prospective
and cross-sectional epidemiological studies have been conducted to
assess the extent to which environmental lead exposure affects
CNS-based psychological functions. Lead has been shown to be
associated with impaired neurobehavioural functioning in children.
Impairment of psychological and neurobehavioural functions has
been found after long-term lead exposure of workers.
Electrophysiological parameters have been shown to be useful
indicators of subclinical lead effects in the CNS.
Peripheral neuropathy has long been known to be caused by
long-term high-level lead exposure at the workplace. Slowing of nerve
conduction velocity has been found at lower levels. These effects have
often been found to be reversible after cessation of exposure,
depending on the age and duration of exposure.
The effect of lead on the heart is indirect and occurs via the
autonomic nervous system; it has no direct effect on the myocardium.
The collective evidence from population studies in adults indicates
very weak associations between PbB concentration and systolic or
diastolic blood pressure. Given the difficulties of allowing for
relevant confounding factors, a causal relationship cannot be
established from these studies. There is no evidence to suggest that
any association of PbB concentration with blood pressure is of major
health importance.
Lead is known to cause proximal renal tubular damage,
characterized by generalized aminoaciduria, hypophosphataemia with
relative hyperphosphaturia and glycosuria accompanied by nuclear
inclusion bodies, mitochondrial changes and cytomegaly of the proximal
tubular epithelial cells. Tubular effects are noted after relatively
short-term exposures and are generally reversible, whereas sclerotic
changes and interstitial fibrosis, resulting in decreased kidney
function and possible renal failure, require chronic exposure to high
lead levels. Increased risk from nephropathy was noted in workers with
a PbB level of over 3.0 µmol/litre (about 60 µg/dl). Renal effects
have recently been seen among the general population when more
sensitive indicators of function were measured.
The reproductive effects of lead in the male are limited to sperm
morphology and count. In the female, some adverse pregnancy outcomes
have been attributed to lead.
Lead does not appear to have deleterious effects on skin, muscle
or the immune system. Except in the case of the rat, lead does not
appear to be related to the development of tumours.
1.8 Evaluation of human health risks
Lead adversely affects several organs and organ systems, with
subcellular changes and neurodevelopmental effects appearing to be the
most sensitive. An association between PbB level and hypertension
(blood pressure) has been reported. Lead produces a cascade of effects
on the haem body pool and affects haem synthesis. However, some of
these effects are not considered adverse. Calcium homoeostasis is
affected, thus interfering with other cellular processes.
a) The most substantial evidence from cross-sectional and
prospective studies of populations with PbB levels generally
below 1.2 µmol/litre (25 µg/dl) relates to decrements in
intelligence quotient (IQ). It is important to note that such
observational studies cannot provide definitive evidence of a
causal relationship with lead exposure. However, the size of the
apparent IQ effect, as assessed at 4 years and above, is a
deficit between 0 and 5 points (on a scale with a standard
deviation of 15) for each 0.48 µmol/litre (10 µg/dl) increment in
PbB level, with a likely apparent effect size of between 1 and 3
points. At PbB levels above 1.2 µmol/litre (25 µg/dl), the
relationship between PbB and IQ may differ. Estimates of effect
size are group averages and only apply to the individual child in
a probabilistic manner.
Existing epidemiological studies do not provide definitive
evidence of a threshold. Below the PbB range of 0.48-0.72 µmol/
litre (10-15 µg/dl), the effects of confounding variables and
limits in the precision in analytical and psychometric
measurements increase the uncertainty attached to any estimate
of effect. However, there is some evidence of an association
below this range.
b) Animal studies provide support for a causal relationship between
lead and nervous system effects, reporting deficits in cognitive
functions at PbB levels as low as 0.53-0.72 µmol/litre
(11-15 µg/dl) which can persist well beyond the termination of
lead exposure.
c) Reduction in human peripheral nerve conduction velocity may occur
with PbB levels as low as 1.44 µmol/litre (30 µg/dl). In
addition, sensory motor function may be impaired with PbB levels
as low as about 1.92 µmol/litre (40 µg/dl), and autonomic nervous
system function (electrocardiographic R-R interval variability)
may be affected at an average PbB level of approximately
1.68 µmol/litre (35 µg/dl). The risk of lead nephropathy is
increased in workers with PbB levels above 2.88 µmol/litre
(60 µg/dl). However, recent studies using more sensitive
indicators of renal function suggest renal effects at lower
levels of lead exposure.
d) Lead exposure is associated with a small increase in blood
pressure. The likely order of magnitude is that for any two-fold
increase in PbB level (e.g., from 0.8 to 1.6 µmol/litre, i.e.
16.6 to 33.3 µg/dl), there is a mean 1 mmHg increase in systolic
blood pressure. The association with diastolic pressure is of a
similar but smaller magnitude. However, there is doubt regarding
whether these statistical associations are really due to an
effect of lead exposure or are an artifact due to confounding
factors.
e) Some but not all epidemiological studies show a dose dependent
association of pre-term delivery and some indices of fetal growth
and maturation at PbB levels of 0.72 µmol/litre (15 µg/dl) or
more.
f) The evidence for carcinogenicity of lead and several inorganic
lead compounds in humans is inadequate.
g) Effects of lead on a number of enzyme systems and biochemical
parameters have been demonstrated. The PbB levels, above which
effects are demonstrable with current techniques for the
parameters that may have clinical significance, are all greater
than 0.96 µmol/litre (20 µg/dl). Some effects on enzymes are
demonstrable at lower PbB levels, but the clinical significance
is uncertain.
2. IDENTITY, PHYSICAL AND CHEMICAL PROPERTIES, AND ANALYTICAL METHODS
2.1 Physical and chemical properties of lead and its compounds
Lead (atomic number, 82; relative atomic mass, 207.19; specific
gravity, 11.34) is a bluish or silvery grey soft metal. The melting
point is 327.5°C and the boiling point at atmospheric pressure 1740°C.
It has four naturally occurring isotopes (208, 206, 207, and 204 in
order of abundance), but the isotopic ratios for various mineral
sources may differ. This property has been exploited in
non-radioactive-tracer environmental and metabolic studies. The
physical and chemical properties of elemental lead and some lead
compounds are summarized in Table 1.
Although lead has four electrons in its valence shell, only two
ionize readily. The usual oxidation state of lead in inorganic
compounds is therefore +2 rather than +4. The inorganic salts of lead,
such as lead sulfide and the oxides of lead, are generally poorly
soluble in water. However, the nitrate, chlorate and, to a much lesser
degree, the chloride are water soluble. Some of the salts formed with
organic acids, e.g., lead oxalate, are also insoluble, but the acetate
is relatively soluble, as shown in Table 1.
Under appropriate conditions of synthesis, stable compounds are
formed in which lead is directly bound to a carbon atom. Industrially
synthesized lead-carbon compounds include tetraethyllead and
tetramethyllead, which are of importance as fuel additives and, hence,
are sources of environmental lead.
2.2 Analytical procedures
In recent years substantial advances have been made in developing
methods for the quantification of metals at low concentrations. In
order to provide improved quality assurance of such measurements,
various reference materials in different matrices have been produced
(Muramatsu & Parr, 1985). To ensure adequate quality control, the
analyst should choose a reference material that matches as closely as
possible the experimental samples to be analysed. Choices are based
upon matrix type and concentration of the element of interest. A
summary of data on 60 biological and 40 environmental (non-biological)
reference materials has been compiled by Muramatsu & Parr (1985).
With the increased interest in measuring lead in the low µg/kg
and µg/m3 range in both environmental and biological samples, there
is need for particular attention to analytical sensitivity and
reliability. As lower concentrations are measured, problems of
laboratory contamination become more significant and quality control
and quality assurance programmes are important. Because of these
concerns, all analytical results for lead should report the laboratory
Table 1. Physical and chemical data on lead and selected lead compoundsa
Name Synonym Relative atomic/ Melting point Boiling point Solubility in cold Soluble in
and formula molecular mass (°C) (°C) water (g/litre)
Lead Pb 207.19 327.502 1740 insoluble HNO3; hot concentrated
H2SO4; hot water;
glycerine; alcohol (slightly)
Lead salts
acetate Pb(C2H3O2)2 325.28 280 - 443
carbonate cerrusite PbCO3 267.20 315 (decomposes) 0.0011 acid; alkali; decomposes in
hot water
chlorate Pb(ClO3)2 374.09 230 (decomposes) very soluble alcohol
chloride cotunite PbCl2 278.10 501 950 919 NH4 salts; slightly in dilute
HCl and in NH3; hot water
(33.4 g/litre)
nitrate Pb(NO3)2 331.20 470 (decomposes) 376.5 alcohol; alkali, NH3; hot
water (1270 g/litre)
orthophosphate Pb3(PO4)2 811.51 1014 0.00014 alkali; HNO3
oxalate PbC2O4 295.21 300 (decomposes) 0.0016 HNO3
dioxide plattnerite PbO2 239.19 290 (decomposes) insoluble dilute HCl; acetic acid
(slightly)
monoxide litharge PbO 223.19 888 0.017 dilute HNO3; acetic acid
Table 1 (cont'd)
Name Synonym Relative atomic/ Melting point Boiling point Solubility in cold Soluble in
and formula molecular mass (°C) (°C) water (g/litre)
sulfate anglesite PbSO4 303.25 1170 0.0425 NH4 salts; concentrated
H2SO4 (slightly)
sulfide galena PbS 239.25 1114 0.00086 acid
a Data from Weast (1985)
performance for reference standards and for parallel blank
measurements of sample contamination for the entire analytical
process. Without these, the validity of the data should be questioned.
2.2.1 Sampling procedures
Particular attention should be paid to the cleanliness of
equipment and glassware and the purity of the chemicals to prevent
secondary contamination by lead.
For the collection of samples, standard trace element methods are
generally required (Behne, 1980) with adequate quality control
procedures (Friberg, 1988; Jorhem & Slorach, 1988, Vahter & Friberg,
1988). Quality control samples for blood, faeces, air filters and dust
have been described (Lind et al., 1988).
2.2.1.1 Sampling of environmental media
In air sampling, both high-volume samplers and low-volume
techniques have been used. It should be noted that the collection
characteristics of high-volume samplers are strongly affected by
particle size and the orientation of the sampler. For particles larger
than 5 µm in diameter the high-volume sampler system is unlikely to
collect representative samples (US EPA, 1986a). As in all sampling for
suspended particulate matter, the accuracy of volume meters should be
checked periodically. The size of the pores in filters for collecting
lead-containing particles should be small, possibly less than 0.2 µm
for glass-fibre filters (Lee & Goranson, 1972).
Depending on the purpose of sampling, care should be taken to
select the appropriate site for sampling devices and to achieve the
best possible sampling conditions by:
* estimating the amount of particulate required for analysis before
deciding on the sample volume and the sampling procedure;
* placing the sampling devices in the appropriate position (e.g.,
in the breathing zone, level with inlet tubes of house
ventilators, at window level in the case of a traffic-laden town
street, at a reasonable distance from the highway in uninhabited
zones, etc);
* taking the samples at appropriate rates and volumes (e.g., daily
breathing volumes, daily ventilating capacities of installations)
and for a sufficient time to make possible the estimation of the
average concentration (e.g., during a work-shift, or a 24-h or
longer period for general population exposure);
* taking into account the use of areas under study (cattle grazing,
recreational zones, children's playgrounds, etc).
In addition, whenever possible a procedure should be used that
makes it possible to evaluate particle-size distribution and the
physicochemical properties of the lead compounds involved, including
the shape of the particles and the state of their aggregation.
Lead may be found in water bound to particulate matter as soluble
complexes or soluble compounds. Techniques for sampling water must
take this into account. It is necessary to sample water without
fractionation (filtration) when total lead levels are required.
Because of the potential for metals from low ionic strength waters to
be adsorbed onto the surfaces of some containers, samples should be
acidified (US EPA, 1986a). Selection, cleaning, and conditioning of
storage and sample containers deserve special attention (Moody, 1982).
The preparation of soil and dust samples for lead analyses
usually involves drying (at 100°C), homogenization by grinding, and
sieving (Thornton & Webb 1975; Bolter et al., 1975). Brown & Black
(1983) have discussed the issues related to quality assurance and
quality control in the collection and analysis of soil samples. Most
reports of lead in soil provide the total elemental abundance either
by acid extraction or X-ray fluorescence. However, the leachable or
bioavailable fraction is of special interest.
For the study of the dietary intake of lead from food, two
general methods have been utilized. The advantages and disadvantages
of the "duplicate portions" technique and the equivalent composite
technique ("market basket") have been reviewed by Pekkarinen (1970).
Although the duplicate portions (duplicate diets) technique can define
variability in consumption, it is expensive, and the sampling and
analytical procedures involved are complicated and limit the number of
individuals included in any study. With the equivalent composite
technique, the economy and ease of collection must be considered in
the light of the variability of results obtained due to uncertainties
in knowledge of actual preparation techniques, including possible lead
levels in water used for processing in individual homes.
The quantity of lead likely to be leached from ceramic surfaces
by different foods and beverages may be assessed using dilute acetic
acid solutions (1 to 4%) at temperatures in the range 20 to 100°C for
times ranging from 30 min to more than 24 h (Laurs, 1976; Merwin,
1976).
Colorimetric methods are suitable for screening inorganic
materials such as pottery or paint for lead. Positive reactions
require confirmation by established quantitative methods. Spot tests
using dithizone, rhodizonate and iodide (Feigl et al., 1972) are
available.
2.2.1.2 Sampling of biological materials
The main problem in the sampling of body fluids and tissues for
lead analysis is potential secondary contamination with lead. The low
general population blood lead (PbB) levels in many regions of the
world are complicating screening efforts, requiring levels of
analytical precision and sensitivity that can be achieved only through
intensive QA/QC programmes. Issues related to such sampling have been
examined in detail by US EPA (1986a).
Special precautions are needed to ensure that all venous
blood-collecting and blood-storage materials are as free from lead as
possible (IPCS, 1977). All glass equipment involved in blood
collection and storage should be made of lead-free silicate glass,
rinsed first in mineral acid, then with copious amounts of
glass-distilled or deionized water. Polypropylene syringes have been
recommended (NAS-NRC, l972). Needles should be of stainless steel with
polypropylene hubs. Blood is often drawn directly from the needle into
vacuum tubes. It is wise to confirm periodically the absence of
significant amounts of lead in the anticoagulant used in the blood
container as well as monitoring the contamination level (blank) for
the entire analytical process.
New analytical techniques make it possible to determine lead
concentrations in microlitre quantities of blood. The trend towards
the procurement of micro-samples of blood by skin prick increases the
chance of secondary contamination of the blood. Systematic
investigation on the significance of this problem has been reported
(Mitchell et al., 1974; Mahaffey et al., 1979; DeSilva & Donnan,
1980). Mitchell et al. (l974) describe a procedure whereby sample
contamination can be reduced by spraying collodion over the cleansed
skin before lancing. The correlation between the concentration of lead
in micro-samples and in macro-samples obtained by venepuncture was
fairly good (r=0.92) over a wide range of PbB concentrations
(0.48-4.41 µmol/litre or 10-92 µg/dl whole blood). Mahaffey et al.,
(1979b) found that capillary blood levels in a comparison test were
systematically higher than corresponding venous blood levels; similar
elevations have been reported by DeSilva & Donnan (1980). Since about
1980 the requirement for reliable and accurate micro procedures has
resulted in the development of good protocols. Sinclair & Dohnt (1984)
described a procedure which resulted in the ability to collect
capillary samples with PbB levels only 3.3% higher than the presumably
correct venous value. This procedure has been used in the Port Pirie
Cohort Study (Baghurst et al., 1985, 1992) and for routine
surveillance in the Port Pirie Lead Decontamination Program (Calder et
al., 1990). Also, Lyngbye et al. (1990b) have shown that capillary
sampling without lead contamination is possible. Routine validation by
cross-comparison with venous blood samples should be undertaken on a
regular basis.
The same general precautions to avoid contamination must be taken
in the collection of urine samples as in the collection of blood
samples. Additionally, special care must be taken to prevent
precipitation during storage.
2.2.2 Analytical methods for lead
A number of analytical methods exist for determination of lead in
environmental and biological samples. These methods differ enormously
in their costs (e.g., sophisticated equipment, an adequate
infrastructure to maintain laboratory conditions and chemical
supplies) and personnel requirements (e.g., availability of skilled
personnel in adequate numbers for the work to be undertaken). Both
accuracy and precision of any of the methods can be affected greatly
by contamination of samples within the laboratory. It is important to
utilize the principles of a "clean" laboratory described by Patterson
& Settle (1976) and Everson & Patterson (1980).
It is not the purpose of this section to provide an exhaustive
description of the analytical methods that could be available to
detect and quantify lead levels in environmental and biological
samples. However, an attempt will be made to identify well-established
methods in current use and to provide information on their application
to assist in the interpretation of experimental and epidemiological
studies.
2.2.2.1 Analysis of lead in environmental samples
The most common methods used for the analysis of lead in samples
from air, water, dust, sediment, soil and foodstuffs are flame atomic
absorption spectrometry (AAS), graphite furnace atomic absorption
spectrometry (GFAAS), anodic stripping voltametry (ASV), inductively
coupled plasma-atomic emission spectroscopy (ICP-AES), and X-ray
fluorescence spectroscopy (XRFS). The reference method for the
determination of the absolute amounts of lead is by isotope dilution
mass spectrometry (IDMS) (Settle & Patterson, 1980; Grandjean & Olsen,
1984; US EPA, 1986a), but due to equipment costs and required
expertise, it is not widely used. Spectrophotometric methods, using
diphenylthiocarbazone as the colorimetric reagent, were widely used in
the past; they are less sensitive and are labour-intensive but are
still appropriate. The advantages and disadvantages were described by
Skogerboe et al. (1977).
Gould et al. (1988) utilized a citric acid solution on filter
paper to leach lead from glazed ceramic and/or enamelled metal-ware.
When treated with a lead-sensitive chromogen, there is a reaction
indicating the presence of lead on the paper. The minimal amount of
lead required to produce an observable reaction was 0.25 µg/cm2; the
maximum amount tested was 5 µg/cm2. A colorimetric test based on the
use of sodium sulfide in solution is used to estimate lead in paint
films. It is possible to determine lead concentrations greater than
1 mg/cm2 of dried paint 90% of the time when the method is used by a
trained chemical laboratory technician.
Table 2 summarizes the utility of several representative methods
for specific environmental media.
2.2.2.2 Analysis of lead in biological materials
Biological samples present special problems for the analyst
because of the low lead concentrations and matrix effects. Most
analytical techniques developed to detect and quantify lead can be
adapted to the analysis of such biological materials as blood, urine,
serum, cerebrospinal fluid, solid tissues, hair, teeth and bone.
However, certain techniques are more often used for specific matrices.
Currently, the most commonly used methods are AAS, GFAAS, ASV,
and ICP-AES. Spectrophotometric methods were commonly used in the past
and can be useful. Other specialized methods for lead analysis are
XRFS, neutron activation analysis (NAA), inductively coupled
plasma-mass spectrometry (ICP-MS), and IDMS. Table 3 summarizes the
utility of several analytical procedures applied to various biological
matrices. Included in this table are examples of the application of
XRFS (Christoffersson et al., 1986; Wielopolski et al., 1986; Nilsson
et al., 1991) for the determination in situ of the body burden of
lead.
2.2.2.3 Analytical procedures for biomarkers of lead exposure and
effect
Using standard clinical laboratory techniques, analytical
procedures have been developed: delta-aminolaevulinic acid (ALA);
delta-aminolaevulinic acid dehydratase (ALAD); urinary coproporphyrin
(CPU) and erythrocyte protoporphyrin (EP). All of these assays are
well established and reliable (Grandjean & Olsen 1984; US EPA, 1986a).
These biochemical parameters are influenced by physiological factors
other than lead. They lack the specificity and sensitivity of PbB
measurements as an index of either current lead exposures or body
stores of lead.
2.3 Conversion factors
1 µg/dl = 0.048 µmol/litre
1 µmol/litre = 20.7 µg/dl
Using the above conversion factor, blood lead concentrations are
given as µmol/litre with the equivalent µg/dl in brackets. Calculated
figures have not been rounded and added precision is not to be
inferred from the number of significant figures.
Table 2. Analytical methods for determining lead in environmental samplesa
Sample type Preparation method Analytical method Sample detection Percentage Reference
limit recovery
Air collect particulate matter on membrane ASV with mercury-graphite 0.16 µg/m3 90-110 NIOSH (1977b)
(particulate filter; wet ash with HNO3/HClO4/H2SO4; electrode (NIOSH method
lead) dissolve in acetate buffer P&CAM 191)
Air collect particulate matter on cellulose ICP-AES (NIOSH method 0.34 µg/m3 95-105 NIOSH (1981)
(particulate acetate filter; wet ash with HNO3/HClO4 P&CAM 351)
lead)
Air collect particulate matter on filter; AAS 0.1 µg/m3 93 Scott et al.
(particulate dry ash; extract with HNO3/HCl; dilute AES 0.15 µg/m3 102 (1976)
lead) with HNO3
Air sample on cellulose acetate filter; AAS 8 ng/litre 100-101 Nerin et al.
(particulate dissolve in HNO3 with heat; add HCl/H2O2 (1989)
lead) and react in hydride generator with sodium
borohydride to generate lead hydride
Air collect sample on filter; spike filter with IDMS 0.1 ng/m3 NR Volkening et
(particulate 206Pb; dissolve filter in NaOH; acidify; al. (1988)
lead) separate lead by electrodeposition; dissolve
in acid
Water digest sample with acid; heat; dilute with AAS 1.0 ng/g NR Chau et al.
(total lead) water (1979)
Soil dry sample and sieve for XRF; digest sieved XRF NR 65-98 Krueger &
sample with HNO3 and heat for AAS AAS NR 63-68 Duguay (1989)
Soil dry sample, dry ash; digest with acid AAS 2 µg/g 79-103 Beyer &
and dilute with water Cromartie (1987)
Table 2 (cont'd)
Sample type Preparation method Analytical method Sample detection Percentage Reference
limit recovery
Soil, waste, digest sample with acid; dilute with water AAS (EPA method 7420) 0.1 mg/litre NR US EPA (1986b)
and ground and filter GFAAS (EPA method 7421) 1 µg/litre NR
water
Soil, dust digest sample with hot acid; dry; redissolve AAS 12 ng/g > 80 Que Hee et al.
and paint in HNO3 (1985b)
Sediment, fish, digest sample with acid; heat; dilute with AAS 50 ng/g NR Chau et al.
vegetation water (sediment) (1980)
(total lead) 10 ng/g (fish NR
and vegetation)
Milk add 50 µl (C2H5)4NOH in ethanol to 25 µl GFAAS NR NR Michaelson &
milk; heat and dilute with water to 125 µl Sauerhoff (1974)
Evaporated dry ash sample; dissolve in HNO3 ASV 0.005 µg/g 99 Capar & Rigsby
milk (1989)
Agricultural dry ash sample with H2SO4 and HNO3; DPASV 0.4 ng/g 85-106 Satzger et al.
crops dilute with water (1982)
Grains, milk, bomb digest sample with acid; heat or GFAAS 20 µg/g (bomb) 85-107 Ellen & Van
mussels, fish digest with acid and dry ash; dissolve 5 µg/g (dry ash) 75-107 Loon (1990)
in acid; dilute with water DPASV NR 82-120
Table 2 (cont'd)
Sample type Preparation method Analytical method Sample detection Percentage Reference
limit recovery
Citrus leaves chop or pulverize sample; digest with hot ICP-AES 10-50 µg/litre 75-82 Que Hee &
and paint acid; dry; redissolve in acid (citrus Boyle (1988)
leaves)
89-96
(paint)
a AAS = atomic absorption; AES = atomic emissions spectroscopy; ASV = anode stripping voltametry; (C2H5)4NOH = tetraethylammonium
hydroxide; DPASV = differential pulse anodic stripping voltametry; EPA = US Environmental Protection Agency; GFAAS = graphite furnace
atomic absorption spectrometry; HCl = hydrochloric acid; HClO4 = perchloric acid; HNO3 = nitric acid; H2O2 = hydrogen peroxide;
H2SO4 = sulfuric acid; ICP-AES = inductively coupled plasma/atomic emission spectroscopy; IDMS = isotope dilution mass spectrometry;
NaOH = sodium hydroxide; NIOSH = National Institute for Occupational Safety and Health; NR = not reported; XRF = X-ray fluorescence
Table 3. Analytical methods for determining lead in biological materialsa
Sample Preparation method Analytical method Sample detection Percentage Reference
type limit recovery
Blood wet ash sample with acid mixtures; ASV with mercury-graphite 0.192 µmol/litre 95-105 NIOSH (1977c)
dissolve residue in dilute HClO4 electrode (NIOSH method (4 µg/dl)
P&CAM 195)
Blood wet ash sample with HNO3; dissolve GFAAS (NIOSH method 0.48 µmol/litre NR NIOSH (1977e)
residue in dilute HNO3 P&CAM 214) (10 µg/dl)
Blood dilute sample with Triton X-100(R); add GFAAS 0.011 µmol/litre 93-105 Aguilera de
nitric acid and diammonium phosphate (0.24 µg/dl) Benzo et al.
(1989)
Blood dilute sample with ammonia solution ICP-MS 0.072 µmol/litre 96-111 Delves &
containing Triton X-100(R); analyse (1.5 µg/dl) Campbell
(1988)
Blood dilute sample in 0.2% Triton X-100(R) GFAAS approx. 0.072 97-150 Que Hee et
and water; analyse µmol/litre (approx. al. (1985a)
1.5 µg/dl)
Blood and wet ash sample with HNO3, complex Spectrophotometry 0.144 µmol/litre 97 NIOSH (1977a)
urine with dephenylthiocarbazone and (NIOSH method (3.0 µg/dl) (blood);
extract with chloroform P&CAM 102) 0.0576 µmol/litre 97
(12 µg/litre) (urine)
Serum, filter sample if needed; dilute with ICP-AES 0.048-0.240 µmol/litre 85 (serum) Que Hee &
blood and acid or water (1.0-5.0 µg/dl) Boyle (1988)
urine
Urine wet ash sample with acid mixture and ASV with mercury-graphite 0.0192 µmol/litre 90-110 NIOSH (1977d)
dissolve in dilute HClO4 electrode (NIOSH method (4 µg/litre)
P&CAM 200)
Table 3 (cont'd)
Sample Preparation method Analytical method Sample detection Percentage Reference
type limit recovery
Liver, bomb digest sample with acid and heat, GFAAS 20 µg/g (bomb); 85-107 Ellen & Van
kidney, or digest with acid and dry ash; dissolve (bomb); Loon (1990)
muscle in acid; dilute with water 5 µg/g (dry ashing) 75-107 (dry
ashing)
Bone direct partially polarized photons at XRF 20 µg/g NR Christoffersson
second phalanx of left forefinger et al. (1986)
(non-invasive technique)
Bone direct partially polarized photons at XRF 20 µg/g NR Wielopolski
anteromedial skin surface of mid-tibia et al. (1986)
(non-invasive technique)
Teeth clean and section tooth; digest with ASV NR 83-114 Rabinowitz
HNO3; evaporate; redissolve in buffer et al. (1989)
solution
Teeth dry ash sample; crush; dry ash again; AAS NR 90-110 Steenhout &
dissolve in HNO3 Pourtois
(1981)
a AAS = atomic absorption spectrometry; ASV = anode stripping voltametry; GFAAS = graphite furnace atomic absorption spectrometry;
HClO4 = perchloric acid; HNO3 = nitric acid; ICP-AES = inductively coupled plasma-atomic emission spectroscopy;
ICP-MS = inductively coupled plasma-mass spectrometry; NIOSH = National Institute for Occupational Safety and Health;
NR = not reported; XRF = X-ray fluorescence
3. SOURCES OF HUMAN AND ENVIRONMENTAL EXPOSURE
3.1 Natural occurrence
Because lead is relatively abundant in the earth's crust it is
found naturally throughout the world. The major natural sources of
lead are volcanic emissions, geochemical weathering, and emissions
from sea spray. A small amount of radioisotopic lead (207Pb) is
derived from the decay of radon gas released from geological sources.
It has been estimated that the worldwide natural emission rates of
lead are of the order of 19 000 tonnes/year (Nriagu & Pacyna, 1988),
with volcanic sources accounting for 6400 tonnes/year (Nriagu, 1979).
Owing to centuries of human exploitation of lead resources, it is
difficult to determine the natural content of lead in most ecosystems.
Data on environmental levels, uses, and sources of lead have been
summarized in a recent review (OECD, 1993).
3.1.1 Rocks and soils
The average concentration of lead in the earth's crust is between
10 and 20 mg/kg (IPCS, 1989). The major geological sources of lead are
in igneous and metamorphic rocks.
The soil is the most important repository in terrestrial
ecosystems for contaminants of anthropogenic origin (Nriagu & Pacyna,
1988; Nriagu, 1989). The lead content of soils (which are for
discussion purposes distinguished here from surface dusts) is greatly
influenced by anthropogenic activities and by long- and short-range
airborne transport of lead from various sources. Both dry and wet
deposition are important routes of input.
Lead in soil may be relatively insoluble (as a sulfate, carbonate
or oxide), soluble, adsorbed onto clays, adsorbed and coprecipitated
with sesquioxides, adsorbed onto colloidal organic matter, or
complexed with organic moieties in soil (US EPA, 1986a; IPCS, 1989).
Soil pH, content of humic and fulvic acids, and amount of organic
matter influence the content and mobility of lead in soils. Since
acidic conditions favour the solubilization and leaching of lead from
the solid phase, acidic soils tend to have lower lead concentrations
when analysed as dry soil. Humic and fulvic acids can also mobilize
lead, and certain complex organic molecules can act as chelators of
lead (IPCS, 1989).
Background levels of lead in soil are in the range of 10-70 mg/kg
(GEMS, 1985). Similar results have been found in studies of mobile
source contamination near highways; soil taken at distances of
50-100 m from highways (outside the range of immediate impact from
traffic emissions) usually shows levels of lead below 40 mg/kg. In the
1985, GEMS survey of selected countries, lead concentrations in
topsoil from Malta were found to have a mean of 54 mg/kg in areas at
least 5 m from roadways; less than one metre from roadways the mean
concentration was 138 mg/kg. A 1977 report from Sweden found a mean of
16 mg/kg in non-contaminated areas (GEMS, 1985).
3.1.2 Sediments
Sediments from freshwater and marine environments have been
studied for lead content. This compartment provides a unique record of
the history of changes in global lead fluxes (Patterson, 1983). Levels
of lead in sediments dated before the onset of the industrial
revolution in Western Europe show very low levels, less than 10% of
current levels (Flegal et al., 1987). The average background level of
lead in marine sediments off southern California was reported by
Flegal et al. (1987) to be 1.3 mg/kg.
3.1.3 Water
Flegal et al. (1987) estimate that the natural concentration of
lead in surface water is about 0.02 µg/litre. In general, lead is not
found in ground or surface waters at concentrations above 10 µg/litre
(IPCS, 1989).
Data from oceans indicate very low levels of lead in sea-water
samples not affected directly by significant sources of lead. Water
samples taken from an area of the Pacific, where annual windborne-
input fluxes of lead are estimated to be 3 mg/cm2, have lead
concentrations of 3.5 ng/litre (0-100 m depth) and 0.9 ng/litre at
depths greater than 2500 m. In contrast, water samples taken from the
north Atlantic, where annual windborne-input fluxes of lead are
170 mg/cm2, contain 34 ng lead/litre at the surface and 5 ng/litre
in depths below 2500 m (Patterson, 1983). Settle & Patterson (1980)
have estimated that prehistoric oceans contained 0.5 ng/litre lead.
Flegal et al. (1987) have estimated that over 95% of the lead in
off-shore surface waters is the result of windborne inputs. However,
in coastal waters near Monterey (California, USA), higher
concentrations of lead were found in sea water, sediments and
organisms; these elevations were related to specific sources by
systematic isotope analyses (Flegal et al., 1987).
3.1.4 Air
Anthropogenic inputs of lead from a range of sources have
resulted in global dispersion of both inorganic and organic species of
lead into the air, of which 80-90% is derived from alkyllead fuel
additives (WHO, 1987). Nriagu & Pacyna (1988) estimated that a total
of 330 000 tonnes of lead is discharged directly into the atmosphere
each year. Estimations of pre-industrial levels of lead in air from
natural origins (volcanic emissions, crustal weathering, radon decay
and sea-spray releases) are in the range of 0.01-0.1 µg/m3 (US NRC,
1980). The lowest level reported since 1975 is 0.076 ng/m3 measured
at the South Pole (US EPA, 1986a).
3.1.5 Plants
Lead occurs naturally in plants and results from both deposition
and uptake. There is a positive linear relationship between lead
concentrations in plants and soil (Davies & Thornton, 1989). As with
other environmental compartments, measurement of "background" levels
of lead in plants is complicated by the general contamination of the
globe from centuries of lead use, which has included direct
application of lead-containing chemicals in agriculture (see below)
and contamination of fertilizers with lead. Lead has been measured in
superphosphate fertilizer at concentrations as high as 92 mg/kg (Lisk,
1972). Sewage sludge, used as a source of nutrients in agriculture,
may contain even higher levels of lead. The concentration of lead in
sewage sludge is typically < 1000 mg/kg. Levels as high as 26 g/kg
have been measured in the USA (Chaney et al., 1984). Soil receiving
heavy sludge applications over long periods of time (years) contained
425 mg lead/kg; the concentration in untreated soil was 47 mg/kg
(Beckett, 1979).
3.1.6 Environmental contamination from natural sources
The contribution of natural sources of lead to human exposure is
small. As a result of various breakdown processes, rocks yield lead
which is transferred to the biosphere and the atmosphere, and,
ultimately, back to the earth's crust in the form of sedimentary
rocks. Soluble lead has for thousands of years entered the oceans with
river discharges, and the rate has been estimated by Patterson (1965)
to be around 17 000 tonnes/year. Sources contributing to airborne lead
are silicate dusts, volcanic halogen aerosols, forest fires, sea salts
aerosols, meteoric and meteorite residues, and lead derived from the
decay of radon. While the lead content of most coals is relatively
low, coal fly ash is enriched in lead (Hutton et al., 1988) and is a
source of environmental contamination.
3.2 Anthropogenic sources
World lead consumption has steadily increased over the period
1965-1990 and was about 5.6 × 106 tonnes in 1990 (OECD, 1993).
Further review of the data summarized by OECD (1993) indicates a
change in consumption patterns worldwide. Although the consumption of
lead within the 24 countries of the OECD increased only slightly over
the decade from 1980 to 1990, consumption within less developed
economies (Africa and Asia) increased from 315 000 tonnes in 1970 to
844 000 tonnes in 1990.
3.2.1 Lead mining
Lead occurs in a variety of minerals, the most important of which
are galena (PbS), cerrusite (PbCO3) and anglesite (PbSO4). Galena
is by far the most important source of primary lead. It occurs mostly
in deposits associated with other minerals, particularly those
containing zinc. Mixed lead and zinc ores account for about 70% of
total primary lead supplies. Ores containing mainly lead account for
about 20% and the remaining 10% is obtained as a by-product from other
deposits, such as mixed copper-zinc deposits. The proportions of
various metals may differ in the ores of different countries. Silver
is the most important of the other metals frequently present in lead
deposits, but copper may also be present in concentrations high enough
to be commercially important. Other minor constituents of lead ores
are gold, bismuth, antimony, arsenic, cadmium, tin, gallium, thallium,
indium, germanium and tellurium.
The major countries producing lead from mining activity during
1987-1991 were the USA, Canada, Australia, Peru, the former USSR and
Mexico, as shown in Table 4. Other countries producing lead from lead
ores include China, the former Yugoslavia, Morocco, Spain, Sweden and
Tunisia. In general, the level of world production of lead from mining
activities has remained relatively constant at about 3.3 × 106
tonnes between 1988 and 1991 (ILZSG, 1992); this represents roughly
60% of the world demand for lead.
Table 4. Major countries producing lead from ore and ore
concentratesa
Country 1987 1988 1989 1990 1991
Canada 423 200 366 600 276 100 241 300 278 100
USA 318 300 395 700 419 300 495 200 483 300
Ex-USSR 510 000 520 000 500 000 490 000 --
Australia 489 200 462 000 495 000 570 000 579 000
Mexico 177 200 178 100 163 000 174 100 158 800
Peru 204 000 149 000 192 200 187 800 199 100
a From: World Bureau of Metal Statistics (1992)
3.2.2 Smelting and refining
Smelting and refining are classified as either primary or
secondary, the former producing refined lead products from ores or
concentrates (primary lead) and the latter producing lead by
recovering it from lead-bearing scrap and waste materials (secondary
lead). Secondary lead is derived from processing what is termed new
scrap arising during manufacturing processes and recycled old scrap
arising from waste materials containing lead. Most scrap is from old
sources, of which the most important are lead plates from batteries,
solder, common babbitt, soft lead, lead solders, cable coverings, type
metals, dross and other lead-containing products. There has been an
increasing contribution of secondary lead sources to the total
worldwide production of lead, as shown in Table 5 (World Bureau of
Metal Statistics, 1992). Secondary sources of lead supplied between 35
and 40% of world production during the period from 1970 to 1990.
3.2.3 Environmental pollution from production of lead
Mining operations and the smelting and refining of both primary
and secondary lead are known to cause contamination of the nearby
environment. The nature and extent of contamination depends on many
factors, including the level of production, the effectiveness of
emission controls, climate, topography and other local factors.
Concentrations are usually highest within 3 km of the point source (US
EPA, 1989). A report from China found that lead levels in ambient air,
plants and soil increased proportionally with proximity to a large
primary smelter; at 50 m from the source, the air lead level was
60 µg/m3, the lead level in plants was 29.1 mg/kg, and soil lead
level was 170 mg/kg (Wang, 1984). However, some earlier studies have
shown air pollution and soil contamination as far as 10 km from
smelters (Djuric et al., 1971; Kerin, 1973; Landrigan et al., 1975).
Table 5. Relative contribution of primary and secondary sources
relative to world lead production (1987-1990)a
1987 1988 1989 1990
Primary 422 100 3 414 200 3 286 500 3 324 500
Secondary 2 045 600 2 103 900 2 272 900 2 254 800
a From: World Bureau of Metal Statistics (1992)
The impacts of lead mining and smelting can persist for long
periods of time. A study conducted in Wales, United Kingdom, in an
area where lead mining began 2000 years ago and ended in the middle of
the 20th century, found high concentrations of lead in soils (Davies
et al., 1985). In Port Pirie, Australia, a community with one of the
world's largest and oldest primary lead smelters, lead levels in soils
were found to be grossly elevated, and the incidence of elevated blood
lead levels in pregnant women and young children was also increased
above that found in other communities in Australia (Wilson et al.,
1986).
3.3 Consumption and uses of lead and its compounds
Lead has a combination of physical and chemical properties that
have made it extremely useful industrially, i.e. high density, high
opacity to gamma and X-ray energies, low sound conductance, a low
melting point, exceptional malleability, high corrosion resistance,
and stability. In 1990, 5.627 × 106 tonnes of lead were consumed
worldwide (ILZSG, 1992). The twenty-four industrialized countries of
the OECD consumed approximately 65% of this amount, with eastern
Europe and the former USSR using 21%. Asia now utilizes about 9% of
the world's lead production.
The use patterns of refined lead vary from country to country.
The situation in 1990 in Mexico is shown in Table 6, while end-use
categories within OECD countries are summarized in Fig. 1, which
indicates the changes between 1970 and 1990 (OECD, 1993).
Table 6. Principal uses of refined lead in Mexicoa
Type of product 1988 1990
(%) (%)
Oxides 69.7 56.7
Batteries 9.2 17.9
Tetraethyllead 7.9 11.9b
Cables 4.0 1.5
Others 9.2 11.9
a ILZSG (1992)
b This does not reflect the introduction of lead-free petrol
in 1990.
From Fig. 1, it is evident that the largest use of lead within
OECD countries is for battery production, whereas there has been a
large drop in the demand for lead-containing gasoline additives.
However, this pattern is not valid worldwide, e.g., concentrations in
petrol range from zero in such countries as Japan and Thailand to
1.12 g/litre in the Virgin Islands (Octel, 1991).
In the past the use of lead in the chemical industry for the
preparation of paints, pigments and coloured inks was widespread. Many
countries have now restricted this use, and concentrations of lead
greater than 0.06% (USA) and 0.5% (New Zealand) are not permitted in
indoor paints (Albert & Badillo, 1991; OECD, 1993). In 1982, data from
the United Kingdom (UK, 1982) indicated levels of lead between 2500
and 3000 mg/kg in decorative glass paints and up to 448 g/kg in
white-lead primer. Red-leadcontaining paints, still used widely to
paint structural steel works, can contain up to 661 g lead/kg.
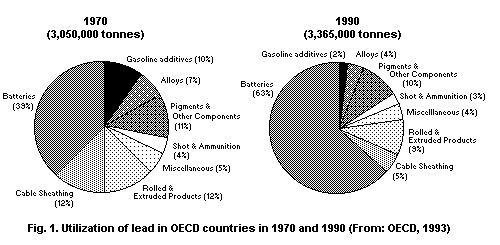 Other disperse uses of lead include lead solders (now banned in
USA for use in drinking-water systems), ammunition (Novotny et al.,
1987), foil on wine bottles (Wai et al., 1979) and cosmetics and
folk-medicines (surma in Asia, Kohl in India, and Al Kohl in Saudi
Arabia and Kuwait) (Fernando et al., 1981).
3.4 Sources of environmental exposure
As noted above, lead is a ubiquitous pollutant in the global
ecosystem, as well as occurring naturally. Its uses have resulted in
increases in soil, water and air lead levels to one to two orders of
magnitude above those estimated to have prevailed prior to rapid
industrialization in the 18th and 19th centuries (Patterson, 1983).
Whereas in specific areas point sources may contribute significant
amounts of lead to the environment, on a global scale, the combustion
of alkyllead in petrol is the predominant source of increased lead in
all compartments of the environment. This has been hypothesized based
upon mass balance studies (Nriagu, 1979) and confirmed by the changes
in environmental lead levels which have followed the significant
reductions in worldwide use of alkyllead as a gasoline additive since
the mid-1980s. For example, lead concentrations in Greenland snow
decreased by a factor of 7.5 over a 20-year period from the late 1960s
(Boutron et al., 1991).
Nriagu & Pacyna (1988) have estimated the global emissions of
lead to the atmosphere resulting from anthropogenic uses (Table 7).
Current estimates (OECD, 1993) of emissions from mobile sources would
be about 30% of the 1983 amounts. Similarly estimates of emissions of
lead to soil in 1983 were made by Nriagu & Pacyna (1988) (Table 8).
Since lead is never degraded, all lead which is shifted from
geological sources by human technology eventually enters the
environment through disposal, although this can be slowed by recycling
and recovery.
Municipal solid waste (MSW), solid waste, hazardous waste, sewage
sludge, and industrial waste-water discharges all may contain lead at
concentrations as high as 50 g/kg. Although few measurements of
environmental lead concentrations in the vicinity of disposal sites
have been conducted, analyses of fly and bottom ash from municipal
incinerators show high concentrations (up to 50 g/kg) of lead (Wadge &
Hutton, 1987), and land disposal sites which have received incinerator
ash for a number of years show high levels of lead in soil (Hutton et
al., 1988).
Dusting and flaking of lead paint from surfaces can be a source
of lead contamination in surface dust and soil near houses or
buildings as well as contributing to the concentrations of lead in
household dust. This process is a function of the type of paint and
the age and state of repair of the structure. When lead paint is
present on structures, both interior and exterior dusts have higher
concentrations than otherwise would be expected (Thornton et al.,
1985). Abatement of lead paint may be a major local source of
environmental contamination, as shown by studies near school buildings
in London (Rundle & Duggan, 1986). Removal of lead-based paints from
bridges and water towers using improper techniques can also result in
significant environmental contamination. Direct application of
lead-contaminated sludge as fertilizers, and residues of lead arsenate
from use in agriculture can lead to the contamination of soil, surface
water and ground water. In local aquatic environments, pollution can
result from leaching of lead from lead shot, shotgun cartridges and
fishing weights (IPCS, 1989). Coal contains small amounts of lead,
which can be concentrated in fly ash from coal combustion (Wadge &
Hutton, 1987) or in stack emissions (Table 8).
Table 7. Estimated worldwide anthropogenic emissions of lead to
the atmosphere (1983)a
Source category Emission rate (tonnes/year)
Coal combustion
- electric utilities 780-4650
- industry and domestic 990-9900
Oil combustion
- electric utilities 230-1740
- industry and domestic 720-2150
Pyrometallurgical non-ferrous metal production
- mining 1700-3400
- lead production 11 700-31 200
- copper-nickel production 11 000-22 100
- zinc-cadmium production 5520-11 500
Secondary non-ferrous metal production 90-1440
Steel and iron manufacturing 1070-14 200
Refuse incineration
- municipal 1400-2800
- sewage sludge 240-300
Phosphate fertilizers 60-270
Cement production 20-14 200
Wood combustion 1200-3000
Mobile sourcesb 248 030
Miscellaneous 3900-5100
Total 289 000-376 000
(median 332 000)
a Adapted from: Nriagu & Pacyna (1988), as in OECD (1993).
b Current estimates (OECD, 1993) for mobile source emissions
would be about 30% of the 1983 amounts.
Table 8. Worldwide emissions of lead into soils (1983)
Source category Emission rate (tonnes/year)
Agricultural and food wastes 1500-27 000
Animal wastes, manure 3200-20 000
Logging and other wood wastes 6600-8200
Urban refuse 18 000-62 000
Municipal sewage sludge 2800-9700
Miscellaneous organic wastes, including excreta 20-1600
Solid wastes, metal manufacturing 4100-11 000
Coal fly ash, bottom fly ash 45 000-242 000
Fertilizer 420-2300
Peat (agricultural and fuel use) 450-2000
Wastage of commercial products 195 000-390 000
Atmospheric fall-out 202 000-263 000
Total yearly input to soils 479 090-1 038 800
Mine tailings 130 000-390 000
Smelter slags and wastes 194 000-390 000
Total yearly discharge on land 803 090-1 818 800
a From: Nriagu & Pacyna (1988), adapted from OECD (1993);
many of these emissions remain localized due to the nature of
the particulate matter
4. ENVIRONMENTAL TRANSPORT, DISTRIBUTION AND TRANSFORMATION
Over the last 10-15 years, a great many studies have been
conducted on the complex interrelationships between environmental lead
emissions and their deposition on such environmental surfaces as
vegetation, soil, house dust and water. All are potential sources of
lead exposure for humans. Transport between environmental compartments
also takes place (see Fig. 2). A full discussion of the complex
physical and chemical processes controlling these pathways is beyond
the scope of this monograph and the reader is directed towards other
reviews for more details (Elias, 1985; US EPA, 1986a; IPCS, 1989).
4.1 Transport and distribution between media
4.1.1 Atmospheric deposition
From the mass balance point of view the transport and
distribution of lead from major emission sources, both fixed and
mobile, is mainly atmospheric. Most of the lead discharged to the
atmosphere is deposited near the source. However, approximately 20% is
widely dispersed (Nriagu, 1979; IPCS, 1989) and contaminates areas as
remote as glacial strata in Greenland (Settle & Patterson, 1980). The
extent of long-range transport of lead particles is dependent upon
particle size, particles > 2 µm in diameter being deposited close to
the source of emission. Between 20 and 60% of emissions from vehicles
has been reported to remain within 25 m of the roadway (ATSDR, 1991).
However, in view of the marked decrease in the concentration of lead
in cores of ice from Greenland since the decreased use of leaded
petrol (Boutron et al., 1991), it is apparent that vehicle emissions
can contribute to the levels of lead in air far from the source.
Long-range transport of lead particles was also noted by Evans &
Rigler (1985).
Lead can be removed from the atmosphere and transferred to
environmental surfaces and compartments by wet or dry deposition. Wet
deposition appears to be more important than dry deposition for the
removal of atmospheric lead. Depending upon geographical location and
the level of emissions in the area, between 40 and 70% of atmospheric
lead is removed by wet deposition (Nielsen, 1984). In most cases it is
poorly soluble and either precipitates out in soils and sediments or
is bound to organic matter in these compartments. For these reasons
lead is not readily removed and tends to accumulate in those
ecosystems where it is deposited (IPCS, 1989). Chan et al. (1986)
calculated the ratio of wet to dry deposition to be 1.63, 1.99 and
2.50 for sites in south, central, and northern Canada, respectively,
while Talbot & Andren (1983) reported that wet deposition accounted
for 80% of the total lead deposited in a semi-remote site in the USA.
Other disperse uses of lead include lead solders (now banned in
USA for use in drinking-water systems), ammunition (Novotny et al.,
1987), foil on wine bottles (Wai et al., 1979) and cosmetics and
folk-medicines (surma in Asia, Kohl in India, and Al Kohl in Saudi
Arabia and Kuwait) (Fernando et al., 1981).
3.4 Sources of environmental exposure
As noted above, lead is a ubiquitous pollutant in the global
ecosystem, as well as occurring naturally. Its uses have resulted in
increases in soil, water and air lead levels to one to two orders of
magnitude above those estimated to have prevailed prior to rapid
industrialization in the 18th and 19th centuries (Patterson, 1983).
Whereas in specific areas point sources may contribute significant
amounts of lead to the environment, on a global scale, the combustion
of alkyllead in petrol is the predominant source of increased lead in
all compartments of the environment. This has been hypothesized based
upon mass balance studies (Nriagu, 1979) and confirmed by the changes
in environmental lead levels which have followed the significant
reductions in worldwide use of alkyllead as a gasoline additive since
the mid-1980s. For example, lead concentrations in Greenland snow
decreased by a factor of 7.5 over a 20-year period from the late 1960s
(Boutron et al., 1991).
Nriagu & Pacyna (1988) have estimated the global emissions of
lead to the atmosphere resulting from anthropogenic uses (Table 7).
Current estimates (OECD, 1993) of emissions from mobile sources would
be about 30% of the 1983 amounts. Similarly estimates of emissions of
lead to soil in 1983 were made by Nriagu & Pacyna (1988) (Table 8).
Since lead is never degraded, all lead which is shifted from
geological sources by human technology eventually enters the
environment through disposal, although this can be slowed by recycling
and recovery.
Municipal solid waste (MSW), solid waste, hazardous waste, sewage
sludge, and industrial waste-water discharges all may contain lead at
concentrations as high as 50 g/kg. Although few measurements of
environmental lead concentrations in the vicinity of disposal sites
have been conducted, analyses of fly and bottom ash from municipal
incinerators show high concentrations (up to 50 g/kg) of lead (Wadge &
Hutton, 1987), and land disposal sites which have received incinerator
ash for a number of years show high levels of lead in soil (Hutton et
al., 1988).
Dusting and flaking of lead paint from surfaces can be a source
of lead contamination in surface dust and soil near houses or
buildings as well as contributing to the concentrations of lead in
household dust. This process is a function of the type of paint and
the age and state of repair of the structure. When lead paint is
present on structures, both interior and exterior dusts have higher
concentrations than otherwise would be expected (Thornton et al.,
1985). Abatement of lead paint may be a major local source of
environmental contamination, as shown by studies near school buildings
in London (Rundle & Duggan, 1986). Removal of lead-based paints from
bridges and water towers using improper techniques can also result in
significant environmental contamination. Direct application of
lead-contaminated sludge as fertilizers, and residues of lead arsenate
from use in agriculture can lead to the contamination of soil, surface
water and ground water. In local aquatic environments, pollution can
result from leaching of lead from lead shot, shotgun cartridges and
fishing weights (IPCS, 1989). Coal contains small amounts of lead,
which can be concentrated in fly ash from coal combustion (Wadge &
Hutton, 1987) or in stack emissions (Table 8).
Table 7. Estimated worldwide anthropogenic emissions of lead to
the atmosphere (1983)a
Source category Emission rate (tonnes/year)
Coal combustion
- electric utilities 780-4650
- industry and domestic 990-9900
Oil combustion
- electric utilities 230-1740
- industry and domestic 720-2150
Pyrometallurgical non-ferrous metal production
- mining 1700-3400
- lead production 11 700-31 200
- copper-nickel production 11 000-22 100
- zinc-cadmium production 5520-11 500
Secondary non-ferrous metal production 90-1440
Steel and iron manufacturing 1070-14 200
Refuse incineration
- municipal 1400-2800
- sewage sludge 240-300
Phosphate fertilizers 60-270
Cement production 20-14 200
Wood combustion 1200-3000
Mobile sourcesb 248 030
Miscellaneous 3900-5100
Total 289 000-376 000
(median 332 000)
a Adapted from: Nriagu & Pacyna (1988), as in OECD (1993).
b Current estimates (OECD, 1993) for mobile source emissions
would be about 30% of the 1983 amounts.
Table 8. Worldwide emissions of lead into soils (1983)
Source category Emission rate (tonnes/year)
Agricultural and food wastes 1500-27 000
Animal wastes, manure 3200-20 000
Logging and other wood wastes 6600-8200
Urban refuse 18 000-62 000
Municipal sewage sludge 2800-9700
Miscellaneous organic wastes, including excreta 20-1600
Solid wastes, metal manufacturing 4100-11 000
Coal fly ash, bottom fly ash 45 000-242 000
Fertilizer 420-2300
Peat (agricultural and fuel use) 450-2000
Wastage of commercial products 195 000-390 000
Atmospheric fall-out 202 000-263 000
Total yearly input to soils 479 090-1 038 800
Mine tailings 130 000-390 000
Smelter slags and wastes 194 000-390 000
Total yearly discharge on land 803 090-1 818 800
a From: Nriagu & Pacyna (1988), adapted from OECD (1993);
many of these emissions remain localized due to the nature of
the particulate matter
4. ENVIRONMENTAL TRANSPORT, DISTRIBUTION AND TRANSFORMATION
Over the last 10-15 years, a great many studies have been
conducted on the complex interrelationships between environmental lead
emissions and their deposition on such environmental surfaces as
vegetation, soil, house dust and water. All are potential sources of
lead exposure for humans. Transport between environmental compartments
also takes place (see Fig. 2). A full discussion of the complex
physical and chemical processes controlling these pathways is beyond
the scope of this monograph and the reader is directed towards other
reviews for more details (Elias, 1985; US EPA, 1986a; IPCS, 1989).
4.1 Transport and distribution between media
4.1.1 Atmospheric deposition
From the mass balance point of view the transport and
distribution of lead from major emission sources, both fixed and
mobile, is mainly atmospheric. Most of the lead discharged to the
atmosphere is deposited near the source. However, approximately 20% is
widely dispersed (Nriagu, 1979; IPCS, 1989) and contaminates areas as
remote as glacial strata in Greenland (Settle & Patterson, 1980). The
extent of long-range transport of lead particles is dependent upon
particle size, particles > 2 µm in diameter being deposited close to
the source of emission. Between 20 and 60% of emissions from vehicles
has been reported to remain within 25 m of the roadway (ATSDR, 1991).
However, in view of the marked decrease in the concentration of lead
in cores of ice from Greenland since the decreased use of leaded
petrol (Boutron et al., 1991), it is apparent that vehicle emissions
can contribute to the levels of lead in air far from the source.
Long-range transport of lead particles was also noted by Evans &
Rigler (1985).
Lead can be removed from the atmosphere and transferred to
environmental surfaces and compartments by wet or dry deposition. Wet
deposition appears to be more important than dry deposition for the
removal of atmospheric lead. Depending upon geographical location and
the level of emissions in the area, between 40 and 70% of atmospheric
lead is removed by wet deposition (Nielsen, 1984). In most cases it is
poorly soluble and either precipitates out in soils and sediments or
is bound to organic matter in these compartments. For these reasons
lead is not readily removed and tends to accumulate in those
ecosystems where it is deposited (IPCS, 1989). Chan et al. (1986)
calculated the ratio of wet to dry deposition to be 1.63, 1.99 and
2.50 for sites in south, central, and northern Canada, respectively,
while Talbot & Andren (1983) reported that wet deposition accounted
for 80% of the total lead deposited in a semi-remote site in the USA.
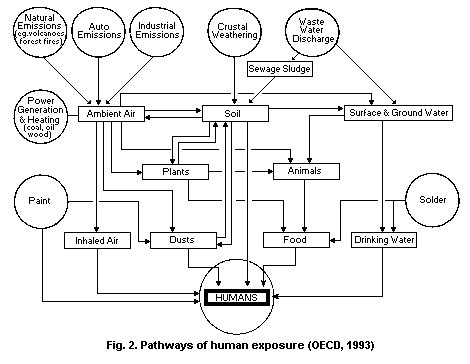 Making several assumptions regarding global atmospheric lead
concentrations, wind speed, surface area and texture, a global
deposition of approximately 410 000 tonnes/year (combined wet and dry)
was calculated by the US EPA (1986a).
4.1.2 Transport to water and soil
When deposited in water, whether from air or through run-off from
soil, lead partitions rapidly between the sediment and aqueous phase,
depending upon the salt content of the water as well as the presence
of organic complexing agents. For example, at pH > 5.4 the total
solubility of lead is about 30 µg/litre in hard water and 500 µg/litre
in soft water (Davies & Everhart 1973). In addition, the presence of
sulfate and carbonate ions can limit lead solubility, as described by
Hem & Durum (1973) in a review of the aqueous chemistry of lead.
Water-borne lead has been found to exist as soluble lead or
undissolved colloidal particles, either suspended in the aqueous phase
or carried as surface coatings on other suspended solids (Lovering,
1976). The ratio of lead in suspended solids to lead in the dissolved
form has been found to vary from 4:1 in rural areas to 27:1 in urban
streams (Getz et al., 1977).
Both natural organic compounds (humic and fulvic acids) as well
as those of anthropogenic origin (e.g., ethylenediamino-tetraacetic
and nitrilotriacetic acids) may complex lead found in surface waters
(Steelink, 1977; Reuter & Perdue, 1977; Neubecker & Allen, 1983). The
presence in water of such chelators can increase the rate of solution
of lead compounds (e.g., lead sulfide) 10 to 60 times over that of
water at the same pH without fulvates (Bondarenko, 1968; Lovering,
1976).
Lead accumulation in soils is primarily a function of the rate of
wet and dry deposition from the atmosphere. Transport within soil and
the bioavailability of lead from soil are dependent upon many factors,
including pH, mineral composition of the soil, and amount and type of
organic material, with most of the lead being bound within the upper
5 cm of soil (Reaves & Berrow 1984; Garcia-Miragaya, 1984). This
limits the amount which can be leached into water or be available for
uptake into plants. It has been shown that only 0.2% of the total lead
in soil can be released into solution by shaking (Dong et al., 1985).
However, the release of lead from organic complexes to the soluble,
and thus bioavailable, form is highly pH dependent. Within the usual
pH range for soils (4 to 6), the organic-lead complexes become more
soluble and the lead more available for plant uptake and leaching into
water (US EPA, 1986a).
4.1.3 Transport to biota
The transfer of air lead to the biota may be direct or indirect
(uptake from water, soil and vegetation). Examples of the accumulation
of lead into aquatic (wet and dry deposition) and terrestrial
organisms are given in Environmental Health Criteria 85: Lead -
Environmental Aspects (IPCS, 1989). Relevant parts of that monograph
are summarized here.
4.1.3.1 Aquatic organisms
In aquatic and aquatic/terrestrial model ecosystems, uptake by
primary producers and consumers seems to be determined by the
bioavailability of the lead. Bioavailability is generally much lower
whenever organic material, sediment or mineral particles (e.g., clay)
are present. In many organisms, it is unclear whether lead is adsorbed
onto the organism or actually taken up. Consumers take up lead from
their contaminated food, often to high concentrations but without
biomagnification.
The uptake and accumulation of lead by aquatic organisms from
water and sediment are influenced by various environmental factors
such as temperature, salinity and pH, as well as humic and alginic
acid content.
In contaminated aquatic systems, almost all of the lead is
tightly bound to sediment. Only a minor fraction is dissolved in the
water, even in the interstitial water.
The lead uptake by fish reaches equilibrium only after a number
of weeks of exposure. Lead is accumulated mostly in gill, liver,
kidney and bone.
Fish eggs show increasing lead levels with increased exposure
concentration, and there are indications that lead is present on the
egg surface but not accumulated in the embryo.
Fish accumulate lead from water as well as sediments; aquatic
uptake is influenced by the presence of cations and the oxygen content
of the water (IPCS, 1989).
4.1.3.2 Terrestrial organisms
In bacteria, the majority of lead is found in the cell wall. A
similar phenomenon is also noted in higher plants. Some lead that
passes into the plant root cell can be combined with new cell wall
material and subsequently removed from the cytoplasm to the cell wall.
Of the lead remaining in the root cell, there is evidence of very
little translocation to other parts of the plant because the
concentration of lead in shoot and leaf tissue is usually much lower
than in root. Foliar uptake of lead occurs, but only to a very limited
extent.
In animals, there is a positive correlation between tissue and
dietary lead concentrations, although tissue concentrations are almost
always lower. The distribution of lead within animals is closely
associated with calcium metabolism.
Lead shot is typically trapped in the gizzard of birds where it
is slowly ground down resulting in the release of lead.
The tetravalent organic form of lead is generally more toxic than
the divalent inorganic form, and its distribution in organisms may not
specifically follow calcium metabolism.
4.2 Environmental transformation
4.2.1 Abiotic transformation
Once released into the environment lead may be transformed from
one inorganic species or particle size to another. However, as an
element it is not subject to degradation. For example, lead-containing
particles in automobile exhaust are usually lead halides or double
salts with ammonium halides (Biggins & Harrison, 1979). Within 24 h,
over 75% of lead particulate matter is transformed to lead carbonates
and sulfates (Olson & Skogerboe, 1975).
4.2.2 Biotransformation
The transformation of inorganic lead to tetramethyllead (TML) has
been observed in aquatic systems, particularly in sediments, and
biomethylation was postulated by Wong et al. (1975) and Schmidt &
Huber (1976). However, no biological methylation of inorganic lead was
noted by Reisinger et al. (1981) in studies under many conditions
using several bacterial species known to alkylate mercury and other
heavy metals. The authors did find chemical methylation in the
presence of methylcobalamin and sulfide or aluminium ions and it was
independent of the presence of bacteria. The evidence for microbial
methylation of various compounds of lead in aquatic systems has been
reviewed by Beijer & Jernelöv (1984). It is still unclear whether the
TML formed is produced abiotically or by biotransformation.
5. ENVIRONMENTAL LEVELS AND HUMAN EXPOSURE
This chapter describes the sources of lead to which people are
exposed and quantifies that exposure. Depending on the source, the
concentration of lead and its bioavailability, the relative
contribution of each source may vary considerably. For example, men
working in ship-breaking, children exposed to deteriorating lead
paint, and people consuming soft water distributed in lead pipes have
frequently been shown to have excessive absorption of lead, leading to
clinically obvious lead poisoning.
In evaluating the exposure of the general population to lead, it
is critical to consider the interrelationships among environmental
pathways for lead and transfers across environmental media. The
general population is exposed to lead simultaneously from many sources
and through multiple pathways as shown in Fig. 2. Thus, while, for
purposes of discussion, exposure via air, water, food, dusts and
soils, and other sources are presented separately in this document,
the total exposure of the general population from all sources must be
considered. Those working in industries where lead is used or produced
may be subject to additional exposure compared with the general
population.
In addition, exposure of certain groups within the general
population may vary because of physiological, behavioural or other
factors. For example, the fetus is exposed to lead via maternal
transfer of internal and external doses, nursing infants may be
exposed to lead in breast milk, the young child is exposed more
intensively to dusts and lead on non-food items (such as lead-painted
toys), alcohol consumption and smoking increase lead exposure,
differences in diet may influence lead exposure markedly, and some
people may be exposed to lead through hobby or occupational activities
in addition to their exposure as members of the general population.
5.1 Inhalation route of exposure
5.1.1 Ambient air
Ambient air can be a major pathway of lead distribution in the
environment. Sources of lead in air include combustion products of
lead additives in petrol, and point sources such as smelters,
incinerators, and some industrial processes including the burning of
fossil fuels.
Concentrations of lead in air range from 7.6 × 10-5 µg/m3 in
remote areas such as Antarctica (Maenhaut et al., 1979) to
> 10 µg/m3 near lead smelters (Elias, 1985).
Almost all lead in air is bound to fine particles of less than
1 µm diameter, although some may be solubilized in acid aerosol
droplets. The size of these particles varies with the source and with
the age of the particle from the time of emission (US EPA, 1986a; WHO,
1987). Most lead in air is inorganic lead, and the predominant source
is from the combustion of tetraethyl- and tetramethyllead used as fuel
additives (US EPA, 1986a; WHO, 1987). A summary of lead levels on fine
airborne particles from some cities in the USA and France is given in
Table 9.
Table 9. Concentrations of lead in fine airborne particles from some
cities in the USA and France in 1984-1985a
City Population Mean lead No. of
concentration samples
(µg/m3)
Clemson (USA) 3000 0.33 15
Senonches (France) 3000 0.005 6
Orleans (France) 110 000 0.11 7
Clermont (France) 161 000 0.045 7
Akron (USA) 200 000 0.052 6
Strasbourg (France) 260 000 0.072 7
Norfolk (USA) 270 000 0.031 6
Chicago (USA) 10 000 000 0.064 5
Paris (France) 10 000 000 0.44 7
a From: Delumyea & Kalivretenos (1987)
5.1.1.1 Emissions from motor vehicles
In Europe, where leaded vehicle fuel is still used, airborne
concentrations of lead in urban areas are likely to be in the range of
0.5-3 µg/m3 (WHO, 1987). Concentrations of between 0.6 and
5.7 µg/m3 were reported in Mexico in 1982 (GEMS, 1985). Where leaded
vehicle fuel is no longer used, concentrations are likely to fall to
< 0.2 µg/m3 (Elias, 1985). In 1990, concentrations of lead in air
in urban areas of the USA had fallen to below 0.07 µg/m3 (US EPA,
1991). This decrease in airborne lead levels in the USA is shown in
Fig. 3. Reductions in lead in air have been reported from Canada,
Germany, Norway and the United Kingdom (OECD, 1993).
5.1.1.2 Stationary sources
Where emissions are largely uncontrolled, concentrations of lead
in air around stationary sources such as lead smelters range from over
10 µg/m3 50 m from the smelter to 1.5 µg/m3 one km away (Wang et
al., 1992). Where more stringent emission controls are used,
concentrations of lead are much lower (US EPA, 1991). The Port Pirie
Making several assumptions regarding global atmospheric lead
concentrations, wind speed, surface area and texture, a global
deposition of approximately 410 000 tonnes/year (combined wet and dry)
was calculated by the US EPA (1986a).
4.1.2 Transport to water and soil
When deposited in water, whether from air or through run-off from
soil, lead partitions rapidly between the sediment and aqueous phase,
depending upon the salt content of the water as well as the presence
of organic complexing agents. For example, at pH > 5.4 the total
solubility of lead is about 30 µg/litre in hard water and 500 µg/litre
in soft water (Davies & Everhart 1973). In addition, the presence of
sulfate and carbonate ions can limit lead solubility, as described by
Hem & Durum (1973) in a review of the aqueous chemistry of lead.
Water-borne lead has been found to exist as soluble lead or
undissolved colloidal particles, either suspended in the aqueous phase
or carried as surface coatings on other suspended solids (Lovering,
1976). The ratio of lead in suspended solids to lead in the dissolved
form has been found to vary from 4:1 in rural areas to 27:1 in urban
streams (Getz et al., 1977).
Both natural organic compounds (humic and fulvic acids) as well
as those of anthropogenic origin (e.g., ethylenediamino-tetraacetic
and nitrilotriacetic acids) may complex lead found in surface waters
(Steelink, 1977; Reuter & Perdue, 1977; Neubecker & Allen, 1983). The
presence in water of such chelators can increase the rate of solution
of lead compounds (e.g., lead sulfide) 10 to 60 times over that of
water at the same pH without fulvates (Bondarenko, 1968; Lovering,
1976).
Lead accumulation in soils is primarily a function of the rate of
wet and dry deposition from the atmosphere. Transport within soil and
the bioavailability of lead from soil are dependent upon many factors,
including pH, mineral composition of the soil, and amount and type of
organic material, with most of the lead being bound within the upper
5 cm of soil (Reaves & Berrow 1984; Garcia-Miragaya, 1984). This
limits the amount which can be leached into water or be available for
uptake into plants. It has been shown that only 0.2% of the total lead
in soil can be released into solution by shaking (Dong et al., 1985).
However, the release of lead from organic complexes to the soluble,
and thus bioavailable, form is highly pH dependent. Within the usual
pH range for soils (4 to 6), the organic-lead complexes become more
soluble and the lead more available for plant uptake and leaching into
water (US EPA, 1986a).
4.1.3 Transport to biota
The transfer of air lead to the biota may be direct or indirect
(uptake from water, soil and vegetation). Examples of the accumulation
of lead into aquatic (wet and dry deposition) and terrestrial
organisms are given in Environmental Health Criteria 85: Lead -
Environmental Aspects (IPCS, 1989). Relevant parts of that monograph
are summarized here.
4.1.3.1 Aquatic organisms
In aquatic and aquatic/terrestrial model ecosystems, uptake by
primary producers and consumers seems to be determined by the
bioavailability of the lead. Bioavailability is generally much lower
whenever organic material, sediment or mineral particles (e.g., clay)
are present. In many organisms, it is unclear whether lead is adsorbed
onto the organism or actually taken up. Consumers take up lead from
their contaminated food, often to high concentrations but without
biomagnification.
The uptake and accumulation of lead by aquatic organisms from
water and sediment are influenced by various environmental factors
such as temperature, salinity and pH, as well as humic and alginic
acid content.
In contaminated aquatic systems, almost all of the lead is
tightly bound to sediment. Only a minor fraction is dissolved in the
water, even in the interstitial water.
The lead uptake by fish reaches equilibrium only after a number
of weeks of exposure. Lead is accumulated mostly in gill, liver,
kidney and bone.
Fish eggs show increasing lead levels with increased exposure
concentration, and there are indications that lead is present on the
egg surface but not accumulated in the embryo.
Fish accumulate lead from water as well as sediments; aquatic
uptake is influenced by the presence of cations and the oxygen content
of the water (IPCS, 1989).
4.1.3.2 Terrestrial organisms
In bacteria, the majority of lead is found in the cell wall. A
similar phenomenon is also noted in higher plants. Some lead that
passes into the plant root cell can be combined with new cell wall
material and subsequently removed from the cytoplasm to the cell wall.
Of the lead remaining in the root cell, there is evidence of very
little translocation to other parts of the plant because the
concentration of lead in shoot and leaf tissue is usually much lower
than in root. Foliar uptake of lead occurs, but only to a very limited
extent.
In animals, there is a positive correlation between tissue and
dietary lead concentrations, although tissue concentrations are almost
always lower. The distribution of lead within animals is closely
associated with calcium metabolism.
Lead shot is typically trapped in the gizzard of birds where it
is slowly ground down resulting in the release of lead.
The tetravalent organic form of lead is generally more toxic than
the divalent inorganic form, and its distribution in organisms may not
specifically follow calcium metabolism.
4.2 Environmental transformation
4.2.1 Abiotic transformation
Once released into the environment lead may be transformed from
one inorganic species or particle size to another. However, as an
element it is not subject to degradation. For example, lead-containing
particles in automobile exhaust are usually lead halides or double
salts with ammonium halides (Biggins & Harrison, 1979). Within 24 h,
over 75% of lead particulate matter is transformed to lead carbonates
and sulfates (Olson & Skogerboe, 1975).
4.2.2 Biotransformation
The transformation of inorganic lead to tetramethyllead (TML) has
been observed in aquatic systems, particularly in sediments, and
biomethylation was postulated by Wong et al. (1975) and Schmidt &
Huber (1976). However, no biological methylation of inorganic lead was
noted by Reisinger et al. (1981) in studies under many conditions
using several bacterial species known to alkylate mercury and other
heavy metals. The authors did find chemical methylation in the
presence of methylcobalamin and sulfide or aluminium ions and it was
independent of the presence of bacteria. The evidence for microbial
methylation of various compounds of lead in aquatic systems has been
reviewed by Beijer & Jernelöv (1984). It is still unclear whether the
TML formed is produced abiotically or by biotransformation.
5. ENVIRONMENTAL LEVELS AND HUMAN EXPOSURE
This chapter describes the sources of lead to which people are
exposed and quantifies that exposure. Depending on the source, the
concentration of lead and its bioavailability, the relative
contribution of each source may vary considerably. For example, men
working in ship-breaking, children exposed to deteriorating lead
paint, and people consuming soft water distributed in lead pipes have
frequently been shown to have excessive absorption of lead, leading to
clinically obvious lead poisoning.
In evaluating the exposure of the general population to lead, it
is critical to consider the interrelationships among environmental
pathways for lead and transfers across environmental media. The
general population is exposed to lead simultaneously from many sources
and through multiple pathways as shown in Fig. 2. Thus, while, for
purposes of discussion, exposure via air, water, food, dusts and
soils, and other sources are presented separately in this document,
the total exposure of the general population from all sources must be
considered. Those working in industries where lead is used or produced
may be subject to additional exposure compared with the general
population.
In addition, exposure of certain groups within the general
population may vary because of physiological, behavioural or other
factors. For example, the fetus is exposed to lead via maternal
transfer of internal and external doses, nursing infants may be
exposed to lead in breast milk, the young child is exposed more
intensively to dusts and lead on non-food items (such as lead-painted
toys), alcohol consumption and smoking increase lead exposure,
differences in diet may influence lead exposure markedly, and some
people may be exposed to lead through hobby or occupational activities
in addition to their exposure as members of the general population.
5.1 Inhalation route of exposure
5.1.1 Ambient air
Ambient air can be a major pathway of lead distribution in the
environment. Sources of lead in air include combustion products of
lead additives in petrol, and point sources such as smelters,
incinerators, and some industrial processes including the burning of
fossil fuels.
Concentrations of lead in air range from 7.6 × 10-5 µg/m3 in
remote areas such as Antarctica (Maenhaut et al., 1979) to
> 10 µg/m3 near lead smelters (Elias, 1985).
Almost all lead in air is bound to fine particles of less than
1 µm diameter, although some may be solubilized in acid aerosol
droplets. The size of these particles varies with the source and with
the age of the particle from the time of emission (US EPA, 1986a; WHO,
1987). Most lead in air is inorganic lead, and the predominant source
is from the combustion of tetraethyl- and tetramethyllead used as fuel
additives (US EPA, 1986a; WHO, 1987). A summary of lead levels on fine
airborne particles from some cities in the USA and France is given in
Table 9.
Table 9. Concentrations of lead in fine airborne particles from some
cities in the USA and France in 1984-1985a
City Population Mean lead No. of
concentration samples
(µg/m3)
Clemson (USA) 3000 0.33 15
Senonches (France) 3000 0.005 6
Orleans (France) 110 000 0.11 7
Clermont (France) 161 000 0.045 7
Akron (USA) 200 000 0.052 6
Strasbourg (France) 260 000 0.072 7
Norfolk (USA) 270 000 0.031 6
Chicago (USA) 10 000 000 0.064 5
Paris (France) 10 000 000 0.44 7
a From: Delumyea & Kalivretenos (1987)
5.1.1.1 Emissions from motor vehicles
In Europe, where leaded vehicle fuel is still used, airborne
concentrations of lead in urban areas are likely to be in the range of
0.5-3 µg/m3 (WHO, 1987). Concentrations of between 0.6 and
5.7 µg/m3 were reported in Mexico in 1982 (GEMS, 1985). Where leaded
vehicle fuel is no longer used, concentrations are likely to fall to
< 0.2 µg/m3 (Elias, 1985). In 1990, concentrations of lead in air
in urban areas of the USA had fallen to below 0.07 µg/m3 (US EPA,
1991). This decrease in airborne lead levels in the USA is shown in
Fig. 3. Reductions in lead in air have been reported from Canada,
Germany, Norway and the United Kingdom (OECD, 1993).
5.1.1.2 Stationary sources
Where emissions are largely uncontrolled, concentrations of lead
in air around stationary sources such as lead smelters range from over
10 µg/m3 50 m from the smelter to 1.5 µg/m3 one km away (Wang et
al., 1992). Where more stringent emission controls are used,
concentrations of lead are much lower (US EPA, 1991). The Port Pirie
 smelter in South Australia is estimated to have lost 80 000 tonnes of
lead to the environment in non-stack fugitive emissions from 1889 to
1982. It is also estimated that from 1969 to 1981 this smelter
discharged 40 tonnes of lead per year into the environment. Ambient
air concentrations near the smelter were between 0.5 and 10 µg/m3
(Body et al., 1988).
5.1.2 Indoor air
Davies et al. (1987a) sampled indoor and ambient air lead levels
and found that where there was no interior lead source, such as
lead-painted surfaces, air lead concentrations inside dwellings were
highly correlated with those outside and averaged approximately 60% of
those in the external air immediately outside the house. A similar
ratio was reported in the Arnhem lead study (Diemel et al., 1981).
Indoor air lead levels are affected by the presence of smokers and
lead-painted surfaces.
Levels of airborne lead in indoor shooting ranges have been shown
to range from 2.7 to 90.5 µg/m3 depending upon the location of
sampling, which varied from the showroom to target area (Novotny et
al., 1987).
5.1.3 Air in the working environment
The diversity and extent of the industrial applications of lead
is such that it is impossible to make general statements about
exposure levels. In many instances actual exposure levels have not
been measured and often work is carried out in small enterprises which
may not be subject to workplace controls or legislated requirements.
Airborne lead concentrations in the occupational setting vary
considerably according to the type of industry and the level of
industrial hygiene practised at each plant. Occupations and operations
that may present lead hazards to workers are listed in Table 10.
Recent monitoring data (1980-1985) from Finland are summarized in
Fig. 4 (Jaakkola & Anttila, 1992).
5.1.4 Smoking of tobacco
Lead is present in tobacco. The mean content of lead in
filter-tipped cigarettes produced between 1960 and 1980 was 2.4 µg/g.
Approximately 5% of this lead may be inhaled; the remainder occurs in
the ash and side-stream smoke (Mussalo-Rauhamaa et al., 1986).
smelter in South Australia is estimated to have lost 80 000 tonnes of
lead to the environment in non-stack fugitive emissions from 1889 to
1982. It is also estimated that from 1969 to 1981 this smelter
discharged 40 tonnes of lead per year into the environment. Ambient
air concentrations near the smelter were between 0.5 and 10 µg/m3
(Body et al., 1988).
5.1.2 Indoor air
Davies et al. (1987a) sampled indoor and ambient air lead levels
and found that where there was no interior lead source, such as
lead-painted surfaces, air lead concentrations inside dwellings were
highly correlated with those outside and averaged approximately 60% of
those in the external air immediately outside the house. A similar
ratio was reported in the Arnhem lead study (Diemel et al., 1981).
Indoor air lead levels are affected by the presence of smokers and
lead-painted surfaces.
Levels of airborne lead in indoor shooting ranges have been shown
to range from 2.7 to 90.5 µg/m3 depending upon the location of
sampling, which varied from the showroom to target area (Novotny et
al., 1987).
5.1.3 Air in the working environment
The diversity and extent of the industrial applications of lead
is such that it is impossible to make general statements about
exposure levels. In many instances actual exposure levels have not
been measured and often work is carried out in small enterprises which
may not be subject to workplace controls or legislated requirements.
Airborne lead concentrations in the occupational setting vary
considerably according to the type of industry and the level of
industrial hygiene practised at each plant. Occupations and operations
that may present lead hazards to workers are listed in Table 10.
Recent monitoring data (1980-1985) from Finland are summarized in
Fig. 4 (Jaakkola & Anttila, 1992).
5.1.4 Smoking of tobacco
Lead is present in tobacco. The mean content of lead in
filter-tipped cigarettes produced between 1960 and 1980 was 2.4 µg/g.
Approximately 5% of this lead may be inhaled; the remainder occurs in
the ash and side-stream smoke (Mussalo-Rauhamaa et al., 1986).
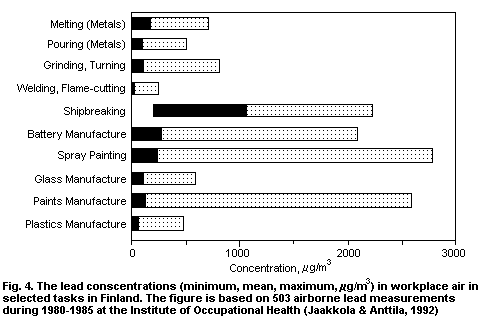 Table 10. Occupations or operations which may present lead hazards for
workersa
Primary and secondary lead smelting Lead mining
Welding and cutting of lead-painted metal Plumbing
constructions
Welding of galvanized or zinc silicate coated sheets Cable making
Shipbreaking Wire patenting
Nonferrous foundries Lead casting
Storage battery manufacture: pasting, assembling, Type founding in
welding of battery connectors printing shops
Production of lead paints Stereotype setting
Spray painting Assembling of cars
Mixing (by hand) of lead stabilizers into Automobile repair
polyvinyl chloride
Mixing (by hand) of crystal glass mass Shot making
Sanding or scraping of lead paint Welding (occasionally)
Burning of lead in enamelling workshops Lead glass blowing
Repair of automobile radiators Pottery/glass making
a Adapted from: Hernberg (1973)
5.2 Exposure by ingestion
5.2.1 Water
Exposure of humans to lead from water has been underestimated in
studies of total exposure. Due to the practice of sampling water
systems at points before entry into the distribution piping and
domestic plumbing (US EPA, 1986a; Dabeka et al., 1987), it had been
widely assumed that exposure to lead in drinking-water was not
significant.
Background or natural levels of lead in surface and ground water
are generally low. However, water with low pH and only low
concentrations of dissolved salts (referred to as aggressive) can
leach substantial quantities of lead from pipes, solder and fixtures.
Lead-lined reservoirs, cisterns and holding tanks for water (Mushak &
Crocetti, 1989) can be a major source of lead contamination of
drinking-water. For example, Wiebe et al. (1991) reported the analysis
of over 2000 water samples in Hawaii, USA, following increased
volcanic activity that resulted in the release of acid aerosols. The
lead concentration of drinking-water collected in catchment systems
ranged from < 20 to 7000 µg/litre. Sampling programmes conducted at
the tap in the USA during 1985-1988 revealed widespread elevation of
lead in drinking-water, often above the WHO guidance value of
50 µg/litre (WHO, 1984), which has now been revised to 10 µg/litre
(WHO, 1993).
The combination of acid or aggressive water and lead in plumbing
results in very high concentrations of lead in drinking-water,
particularly after it has been standing for several hours (Worth et
al., 1981; Sherlock et al., 1982; Kaminsky et al., 1988).
Surveys in Canada and the USA showed that drinking-water supplies
leaving treatment plants contain 2-3 µg lead/litre (US EPA, 1986a;
Dabeka et al., 1987). In the case of plumbosolvent water, up to 40% of
household samples may exceed 100 µg lead/litre. This has been observed
in Scotland (Sherlock et al., 1986), and reflects the contribution of
plumbing and plumbing fixtures to lead levels of drinking-water.
5.2.2 Food and alcoholic beverages
5.2.2.1 Food
The major source of lead for non-occupationally exposed adults is
food and drink. The proportion of total intake derived from food is
dependent on the concentration of lead in air, water and other
sources. Detailed data are available from several countries, including
Australia (NFA, 1991), USA (Bolger et al., 1991), Sweden (Vahter et
al., 1990) and Canada (Dabeka et al., 1987). Foods have been surveyed
from several other industrialized and developing countries
(Galal-Gorchev, 1991b). Children are exposed to additional lead from
dust and soil, and so lead from foods and beverages may not be the
predominant sources of lead for all age groups.
Lead is present in soils and is transferred to food crops growing
on the soil. Roots usually contain more lead than stems and leaves,
while seeds and fruits have the lowest concentrations.
Particulate lead present in air may adhere tenaciously to leafy
vegetables. Leaves collected in or very near urban areas have been
shown to contain substantially elevated concentrations of lead.
Quantities of lead ingested from the diet vary widely from country to
country.
Data on the lead levels of specific foodstuffs or groups of food
materials, from which one can estimate a daily dietary lead intake,
are available from several countries. In a few studies, foodstuffs
specific to infants and children have also been analysed (Kolbye et
al., 1974; Dabeka & McKenzie, 1987; Vahter et al., 1990; Bolger et
al., 1991; Albert & Badillo, 1991). Data are available for canned
foods typically consumed by young children (Capar & Rigsby, 1989). The
utilization of such data in the calculation of total intake of lead
from food is discussed in section 5.2.2.2.
An overview of the foods contributing to the dietary lead levels
in Australia is shown in Fig. 5. Similar data from other countries are
shown in Table 11 (Galal-Gorchev, 1991b). A summary of the data on
lead levels in foodstuffs from the USA is given in Table 12 and from
Canada in Table 13. More recent data from the USA have shown that
there has been a substantial reduction of lead levels in food consumed
by all age groups during the past two decades (Bolger et al., 1991)
(Fig. 6). A similar decrease in lead intake has been found in the
United Kingdom (OECD, 1993). These data should be considered as
representative of specific areas and such values can be expected to
vary elsewhere according to local agricultural and food-processing
practices, particularly in areas where lead-soldered cans are still
used. Support for this is indicated by a comparison on the one hand of
the decreased use of lead-soldered food and beverage cans in the USA
(Fig. 7) and the increased use of cans produced by alternative
technology, and on the other hand the marked decrease in food-borne
lead shown in Fig. 6. Food may represent a pathway for human lead
exposure from other media such as air and water. The use of leaded
gasoline or the proximity of industries that may produce ambient
emissions of lead can greatly influence dietary lead intake. Therefore
further caution is required when extrapolating between countries with
regards to levels of food-borne lead.
Representative levels of lead in foodstuffs from some 20
countries are given in Table 14 (Galal-Gorchev, 1991a). These results
from the GEMS/FOOD data can be compared with the levels of lead in
specific foodstuffs in the USA and Canada (Tables 12 and 13).
Table 10. Occupations or operations which may present lead hazards for
workersa
Primary and secondary lead smelting Lead mining
Welding and cutting of lead-painted metal Plumbing
constructions
Welding of galvanized or zinc silicate coated sheets Cable making
Shipbreaking Wire patenting
Nonferrous foundries Lead casting
Storage battery manufacture: pasting, assembling, Type founding in
welding of battery connectors printing shops
Production of lead paints Stereotype setting
Spray painting Assembling of cars
Mixing (by hand) of lead stabilizers into Automobile repair
polyvinyl chloride
Mixing (by hand) of crystal glass mass Shot making
Sanding or scraping of lead paint Welding (occasionally)
Burning of lead in enamelling workshops Lead glass blowing
Repair of automobile radiators Pottery/glass making
a Adapted from: Hernberg (1973)
5.2 Exposure by ingestion
5.2.1 Water
Exposure of humans to lead from water has been underestimated in
studies of total exposure. Due to the practice of sampling water
systems at points before entry into the distribution piping and
domestic plumbing (US EPA, 1986a; Dabeka et al., 1987), it had been
widely assumed that exposure to lead in drinking-water was not
significant.
Background or natural levels of lead in surface and ground water
are generally low. However, water with low pH and only low
concentrations of dissolved salts (referred to as aggressive) can
leach substantial quantities of lead from pipes, solder and fixtures.
Lead-lined reservoirs, cisterns and holding tanks for water (Mushak &
Crocetti, 1989) can be a major source of lead contamination of
drinking-water. For example, Wiebe et al. (1991) reported the analysis
of over 2000 water samples in Hawaii, USA, following increased
volcanic activity that resulted in the release of acid aerosols. The
lead concentration of drinking-water collected in catchment systems
ranged from < 20 to 7000 µg/litre. Sampling programmes conducted at
the tap in the USA during 1985-1988 revealed widespread elevation of
lead in drinking-water, often above the WHO guidance value of
50 µg/litre (WHO, 1984), which has now been revised to 10 µg/litre
(WHO, 1993).
The combination of acid or aggressive water and lead in plumbing
results in very high concentrations of lead in drinking-water,
particularly after it has been standing for several hours (Worth et
al., 1981; Sherlock et al., 1982; Kaminsky et al., 1988).
Surveys in Canada and the USA showed that drinking-water supplies
leaving treatment plants contain 2-3 µg lead/litre (US EPA, 1986a;
Dabeka et al., 1987). In the case of plumbosolvent water, up to 40% of
household samples may exceed 100 µg lead/litre. This has been observed
in Scotland (Sherlock et al., 1986), and reflects the contribution of
plumbing and plumbing fixtures to lead levels of drinking-water.
5.2.2 Food and alcoholic beverages
5.2.2.1 Food
The major source of lead for non-occupationally exposed adults is
food and drink. The proportion of total intake derived from food is
dependent on the concentration of lead in air, water and other
sources. Detailed data are available from several countries, including
Australia (NFA, 1991), USA (Bolger et al., 1991), Sweden (Vahter et
al., 1990) and Canada (Dabeka et al., 1987). Foods have been surveyed
from several other industrialized and developing countries
(Galal-Gorchev, 1991b). Children are exposed to additional lead from
dust and soil, and so lead from foods and beverages may not be the
predominant sources of lead for all age groups.
Lead is present in soils and is transferred to food crops growing
on the soil. Roots usually contain more lead than stems and leaves,
while seeds and fruits have the lowest concentrations.
Particulate lead present in air may adhere tenaciously to leafy
vegetables. Leaves collected in or very near urban areas have been
shown to contain substantially elevated concentrations of lead.
Quantities of lead ingested from the diet vary widely from country to
country.
Data on the lead levels of specific foodstuffs or groups of food
materials, from which one can estimate a daily dietary lead intake,
are available from several countries. In a few studies, foodstuffs
specific to infants and children have also been analysed (Kolbye et
al., 1974; Dabeka & McKenzie, 1987; Vahter et al., 1990; Bolger et
al., 1991; Albert & Badillo, 1991). Data are available for canned
foods typically consumed by young children (Capar & Rigsby, 1989). The
utilization of such data in the calculation of total intake of lead
from food is discussed in section 5.2.2.2.
An overview of the foods contributing to the dietary lead levels
in Australia is shown in Fig. 5. Similar data from other countries are
shown in Table 11 (Galal-Gorchev, 1991b). A summary of the data on
lead levels in foodstuffs from the USA is given in Table 12 and from
Canada in Table 13. More recent data from the USA have shown that
there has been a substantial reduction of lead levels in food consumed
by all age groups during the past two decades (Bolger et al., 1991)
(Fig. 6). A similar decrease in lead intake has been found in the
United Kingdom (OECD, 1993). These data should be considered as
representative of specific areas and such values can be expected to
vary elsewhere according to local agricultural and food-processing
practices, particularly in areas where lead-soldered cans are still
used. Support for this is indicated by a comparison on the one hand of
the decreased use of lead-soldered food and beverage cans in the USA
(Fig. 7) and the increased use of cans produced by alternative
technology, and on the other hand the marked decrease in food-borne
lead shown in Fig. 6. Food may represent a pathway for human lead
exposure from other media such as air and water. The use of leaded
gasoline or the proximity of industries that may produce ambient
emissions of lead can greatly influence dietary lead intake. Therefore
further caution is required when extrapolating between countries with
regards to levels of food-borne lead.
Representative levels of lead in foodstuffs from some 20
countries are given in Table 14 (Galal-Gorchev, 1991a). These results
from the GEMS/FOOD data can be compared with the levels of lead in
specific foodstuffs in the USA and Canada (Tables 12 and 13).
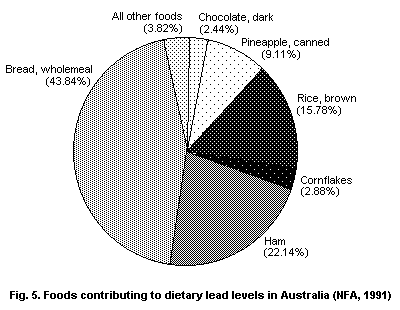
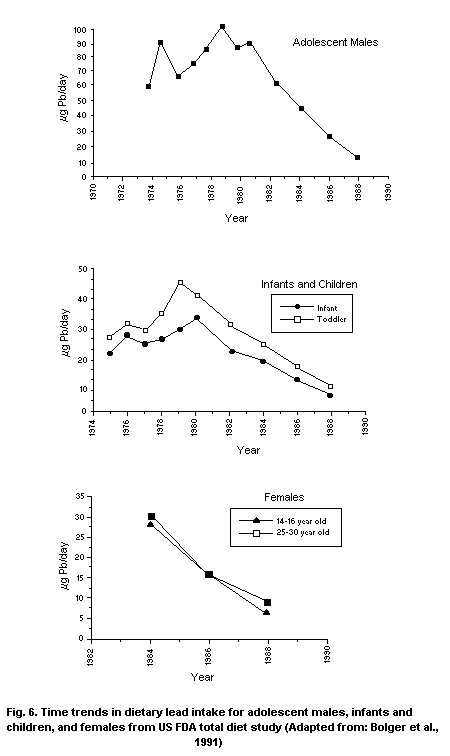
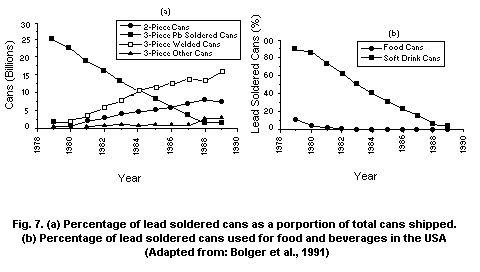 Table 11. Foods contributing to dietary lead levels in Canada,
Finland, Netherlands and the United Kingdoma
Country Food Percentage of total intake
Canada vegetables 17
meat/fish/poultry 17
beverages 15
cereals and products 15
fruits and juices 10
Finland cereals and products 24
fruits 22
beverages, sweets, etc. 20
milk and products 17
vegetables 9
Netherlands drinking-water 30
cereals and products 17
vegetables 12
wines and spirits 9
fruits 6
United Kingdom bread and cereals 15
beverages 14
potatoes 10
milk 9
canned vegetables 8
a From: Galal-Gorchev (1991b)
Table 12. Concentrations of lead in various foods in the USAa
Food group Concentration of leadb (µg/g)
Dairy products 0.003-0.083
Meat, fish and poultry 0.002-0.159
Grain and cereal products 0.002-0.136
Vegetables 0.005-0.649
Fruit and fruit juices 0.005-0.223
Oils, fats and shortenings 0.006-0.073
Sugar and adjuncts 0.006-0.073
Beverages 0.002-0.041
a From: US EPA (1986a)
b Range of concentrations shown are the lowest and highest mean
values for items within the food group and listed in Appendix 7-D
of US EPA (1986a).
Table 13. Levels of lead in various Canadian food categoriesa
Description of food category Median concentration
of lead (µg/kg)b
I Cereals (as prepared with milk, sugar, etc.), 32.4 (11.5-78.3)
bread and toast
II Water consumed directly 2.0 (0.25-71.2)
III Coffee, tea, beer, liquor, sodas, etc. (as prepared) 8.8 (< 0.05-28.9)
IV Fruit juices, fruits (canned and fresh) 7.9 (1.5-109)
V Dairy products and eggs 3.3 (1.21-81.9)
VI Starch vegetables, e.g. potatoes, rice 16.9 (5.5-83.7)
VII Other vegetables, vegetable juices and soups 31.7 (0.62-254)
VIII Meat, fish, poultry, meat-based soups 31.3 (11-121)
IX Miscellaneous (pies, puddings, nuts, snack foods) 33.1 (13.6-1381)
X Cheese (other than cottage cheese) 33.8 (27.7-6775)
a From: Dabeka et al. (1987)
b Values in parentheses are the ranges.
The lead levels in infant foods in Canada, Mexico and USA are
shown in Table 15. In 1987, Dabeka et al. (1987) found the intake of
lead by infants fed evaporated milk stored in lead-soldered cans
exceeded the Provisional Tolerable Weekly Intake of 25 µg lead/kg body
weight, set in 1993 (FAO/WHO, 1993). These values do not include lead
in water used to prepare formulae. It has been reported that infants
fed formulae prepared with water containing high levels of lead
(> 100 µg/litre) have lead intakes exceeding 25 µg/kg body weight per
week (Galal-Gorchev, 1991b).
5.2.2.2 Total intake from food
Guidelines for the determination of dietary intake of chemical
contaminants have been published (WHO, 1985). Three basic approaches
were described, namely: (i) total diet (i.e. market or shopping
basket) studies; (ii) selective studies of individual foodstuffs, and
(iii) duplicate portion (duplicate diet) studies. It is essential to
have food consumption data for the first two methods in order to
estimate a total intake. For all methods, well-designed quality
assurance and quality control programmes are essential and these have
been described by Vahter & Slorach (1990) and Vahter et al. (1991a).
Table 14. Representative levels of lead in foods from GEMS/FOOD
dataa
Commodity Typical lead levels (µg/kg)
Cereals 60
Roots and tubers 50
Fruit 50
Vegetables 50
Meat 50
Vegetable oils and fats 20
Fish 100
Pulses 40
Eggs 20
Nuts and oilseeds 40
Shellfish 20
Offal 20
Spices and herbs 30
Other foods not assessed
Drinking-water 20
Canned beverages 200
Canned foodb 200
a From: Galal-Gorchev (1991a)
b It is assumed that canned food consumption is 2% of total.
Given the differences between countries with respect to dietary
composition, the amount of specific foodstuffs consumed, the
processing technologies employed, whether consumption of water and
alcoholic beverages are included in the estimates of dietary lead, and
the number of samples taken, caution must be exercised in making
comparisons between countries.
During the late 1970s and 1980s the quantity of lead ingested as
part of diet decreased markedly in many countries. For adults not
occupationally exposed to lead, the diet remains the largest
contributor to lead intake. However, the quantities ingested are far
lower than in previous decades. In the USA, typical levels of intake
declined as shown in Fig. 6.
Table 15. Lead levels (µg/kg food) in cow's milk and infant formula
Product Canada Mexico USA
median (range)a averageb averagec
Fluid milk 1.19 (0.01-2.5) 5
Evaporated milk (canned) 71.9 (27-106) 88 10
(cardboard) -- 9
Infant formula
Ready to use lead-solder can 30.1 (1.1-122) 13 10
Ready to use lead-free can 1.6 (1.5-2) 1
Formula powder (1985) 96.6 (3.7-19)
Powdered milkd -- 21
a From: Dabeka & McKenzie (1987)
b From: Albert & Badillo (1991). Data were obtained in 1982.
c From: Bolger et al. (1991). Data were obtained in the late 1980s.
d The concentration of lead in milk consumed by the infant will be
highly dependent on the concentration of lead in water used to
dilute the powdered milk.
An overall summary of the GEMS/FOOD data for adults is given in
Fig. 8 (Galal-Gorchev, 1991a). The trends for lead intake for the
period 1980-1988 in the USA, Japan, Hungary and the United Kingdom are
shown in Fig. 9 (Galal-Gorchev, 1991b). Data for intake of lead by
infants and children in eight countries are shown in Fig. 10
(Galal-Gorchev, 1991a). Additional data are available from other
countries. Although collection methods vary, these data illustrate the
wide variations in ingestion of lead from food. Brunekreef (1986)
noted that the market basket studies tended to overestimate lead
intake when compared with duplicate diet analysis. Examples cited
included reports from the United Kingdom by Fouassin & Fondu (1980),
Buchet et al. (1983) and Sherlock et al. (1982), where market basket
surveys overestimated this intake by 2- to 3-fold.
Other studies from various countries on total lead intakes by
children and adults are summarized in Table 16. Lead contaminated
water has been shown to be a contributor to food-borne lead where
large volumes of water are used in food preparation and cooking, e.g.,
in foods prepared in boiling water.
Table 11. Foods contributing to dietary lead levels in Canada,
Finland, Netherlands and the United Kingdoma
Country Food Percentage of total intake
Canada vegetables 17
meat/fish/poultry 17
beverages 15
cereals and products 15
fruits and juices 10
Finland cereals and products 24
fruits 22
beverages, sweets, etc. 20
milk and products 17
vegetables 9
Netherlands drinking-water 30
cereals and products 17
vegetables 12
wines and spirits 9
fruits 6
United Kingdom bread and cereals 15
beverages 14
potatoes 10
milk 9
canned vegetables 8
a From: Galal-Gorchev (1991b)
Table 12. Concentrations of lead in various foods in the USAa
Food group Concentration of leadb (µg/g)
Dairy products 0.003-0.083
Meat, fish and poultry 0.002-0.159
Grain and cereal products 0.002-0.136
Vegetables 0.005-0.649
Fruit and fruit juices 0.005-0.223
Oils, fats and shortenings 0.006-0.073
Sugar and adjuncts 0.006-0.073
Beverages 0.002-0.041
a From: US EPA (1986a)
b Range of concentrations shown are the lowest and highest mean
values for items within the food group and listed in Appendix 7-D
of US EPA (1986a).
Table 13. Levels of lead in various Canadian food categoriesa
Description of food category Median concentration
of lead (µg/kg)b
I Cereals (as prepared with milk, sugar, etc.), 32.4 (11.5-78.3)
bread and toast
II Water consumed directly 2.0 (0.25-71.2)
III Coffee, tea, beer, liquor, sodas, etc. (as prepared) 8.8 (< 0.05-28.9)
IV Fruit juices, fruits (canned and fresh) 7.9 (1.5-109)
V Dairy products and eggs 3.3 (1.21-81.9)
VI Starch vegetables, e.g. potatoes, rice 16.9 (5.5-83.7)
VII Other vegetables, vegetable juices and soups 31.7 (0.62-254)
VIII Meat, fish, poultry, meat-based soups 31.3 (11-121)
IX Miscellaneous (pies, puddings, nuts, snack foods) 33.1 (13.6-1381)
X Cheese (other than cottage cheese) 33.8 (27.7-6775)
a From: Dabeka et al. (1987)
b Values in parentheses are the ranges.
The lead levels in infant foods in Canada, Mexico and USA are
shown in Table 15. In 1987, Dabeka et al. (1987) found the intake of
lead by infants fed evaporated milk stored in lead-soldered cans
exceeded the Provisional Tolerable Weekly Intake of 25 µg lead/kg body
weight, set in 1993 (FAO/WHO, 1993). These values do not include lead
in water used to prepare formulae. It has been reported that infants
fed formulae prepared with water containing high levels of lead
(> 100 µg/litre) have lead intakes exceeding 25 µg/kg body weight per
week (Galal-Gorchev, 1991b).
5.2.2.2 Total intake from food
Guidelines for the determination of dietary intake of chemical
contaminants have been published (WHO, 1985). Three basic approaches
were described, namely: (i) total diet (i.e. market or shopping
basket) studies; (ii) selective studies of individual foodstuffs, and
(iii) duplicate portion (duplicate diet) studies. It is essential to
have food consumption data for the first two methods in order to
estimate a total intake. For all methods, well-designed quality
assurance and quality control programmes are essential and these have
been described by Vahter & Slorach (1990) and Vahter et al. (1991a).
Table 14. Representative levels of lead in foods from GEMS/FOOD
dataa
Commodity Typical lead levels (µg/kg)
Cereals 60
Roots and tubers 50
Fruit 50
Vegetables 50
Meat 50
Vegetable oils and fats 20
Fish 100
Pulses 40
Eggs 20
Nuts and oilseeds 40
Shellfish 20
Offal 20
Spices and herbs 30
Other foods not assessed
Drinking-water 20
Canned beverages 200
Canned foodb 200
a From: Galal-Gorchev (1991a)
b It is assumed that canned food consumption is 2% of total.
Given the differences between countries with respect to dietary
composition, the amount of specific foodstuffs consumed, the
processing technologies employed, whether consumption of water and
alcoholic beverages are included in the estimates of dietary lead, and
the number of samples taken, caution must be exercised in making
comparisons between countries.
During the late 1970s and 1980s the quantity of lead ingested as
part of diet decreased markedly in many countries. For adults not
occupationally exposed to lead, the diet remains the largest
contributor to lead intake. However, the quantities ingested are far
lower than in previous decades. In the USA, typical levels of intake
declined as shown in Fig. 6.
Table 15. Lead levels (µg/kg food) in cow's milk and infant formula
Product Canada Mexico USA
median (range)a averageb averagec
Fluid milk 1.19 (0.01-2.5) 5
Evaporated milk (canned) 71.9 (27-106) 88 10
(cardboard) -- 9
Infant formula
Ready to use lead-solder can 30.1 (1.1-122) 13 10
Ready to use lead-free can 1.6 (1.5-2) 1
Formula powder (1985) 96.6 (3.7-19)
Powdered milkd -- 21
a From: Dabeka & McKenzie (1987)
b From: Albert & Badillo (1991). Data were obtained in 1982.
c From: Bolger et al. (1991). Data were obtained in the late 1980s.
d The concentration of lead in milk consumed by the infant will be
highly dependent on the concentration of lead in water used to
dilute the powdered milk.
An overall summary of the GEMS/FOOD data for adults is given in
Fig. 8 (Galal-Gorchev, 1991a). The trends for lead intake for the
period 1980-1988 in the USA, Japan, Hungary and the United Kingdom are
shown in Fig. 9 (Galal-Gorchev, 1991b). Data for intake of lead by
infants and children in eight countries are shown in Fig. 10
(Galal-Gorchev, 1991a). Additional data are available from other
countries. Although collection methods vary, these data illustrate the
wide variations in ingestion of lead from food. Brunekreef (1986)
noted that the market basket studies tended to overestimate lead
intake when compared with duplicate diet analysis. Examples cited
included reports from the United Kingdom by Fouassin & Fondu (1980),
Buchet et al. (1983) and Sherlock et al. (1982), where market basket
surveys overestimated this intake by 2- to 3-fold.
Other studies from various countries on total lead intakes by
children and adults are summarized in Table 16. Lead contaminated
water has been shown to be a contributor to food-borne lead where
large volumes of water are used in food preparation and cooking, e.g.,
in foods prepared in boiling water.
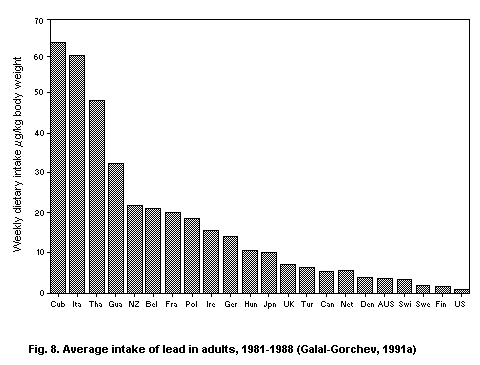
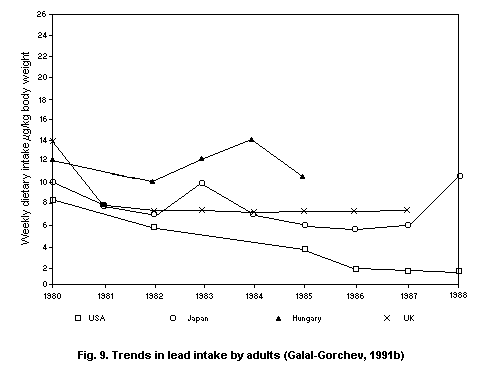
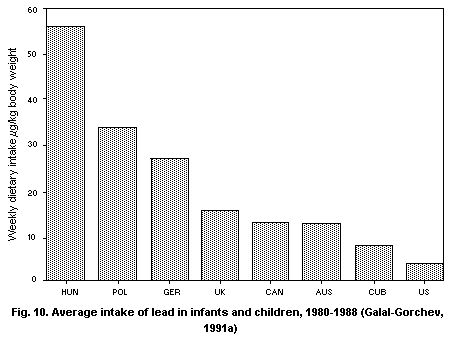 Table 16. Daily lead intake via food in adults and children
Population studied Daily intake (µg/day)a Reference
Adults, United Kingdom 110 M Brunekreef (1986)
Adults, United Kingdom 71 D Brunekreef (1986)
Adults, Belgiumb 282 M Fouassin & Fondu (1980)
Adults, Belgiumb 96 D Buchet et al. (1983)
Adults, Sweden 27 M Slorach et al. (1983)
Adults, Finland 66 M Varo & Kovistoinen (1983)
Adults, Canada 43 D Dabeka et al. (1987)
Adults, USA 82 M Gartrell et al. (1985a)
Adults (female), Japan 31 D Vahter et al. (1991b)
Adults, Germany 61 Kampe (1983)
Adults (female), Croatia 15 D Vahter et al. (1991b)
Adults, Italy 140 IAEA (1987)
Adults (female), China 46 D Vahter et al. (1991b)
Adults, Turkey 70 IAEA (1987)
Adults (female), Sweden 26 D Vahter et al. (1991b)
Children, Poland
0-1 year 225 Olejnik et al. (1985)
1-3 years 259
7-18 years 316
Adults, New Zealand 316 M Pickston et al. (1985)
Children (infant), UK 2-3 breast milk Kovar et al. (1984)
Children (< 1 year), USA 16-17 infant formula Ryu et al. (1983)
& milk
Children, USA
6 months 33.5 M Gartrell et al. (1985b)
2 years 43.4
a M = Market basket survey; D = Duplicate diet study
b Populations studied from the same region.
The relative intake of lead from various sources in 1986 and 1990
in 2-year-old infants and women of child-bearing age in the USA is
shown in Fig. 11. These data illustrate the marked decrease in lead
intake from food over a 4-year period in which there were marked
reductions in the use of lead-soldered cans and lead-containing petrol
additives in the USA (Bolger et al., 1991). Similar decreases in other
countries would no doubt occur after similar actions by public health
officials.
Table 16. Daily lead intake via food in adults and children
Population studied Daily intake (µg/day)a Reference
Adults, United Kingdom 110 M Brunekreef (1986)
Adults, United Kingdom 71 D Brunekreef (1986)
Adults, Belgiumb 282 M Fouassin & Fondu (1980)
Adults, Belgiumb 96 D Buchet et al. (1983)
Adults, Sweden 27 M Slorach et al. (1983)
Adults, Finland 66 M Varo & Kovistoinen (1983)
Adults, Canada 43 D Dabeka et al. (1987)
Adults, USA 82 M Gartrell et al. (1985a)
Adults (female), Japan 31 D Vahter et al. (1991b)
Adults, Germany 61 Kampe (1983)
Adults (female), Croatia 15 D Vahter et al. (1991b)
Adults, Italy 140 IAEA (1987)
Adults (female), China 46 D Vahter et al. (1991b)
Adults, Turkey 70 IAEA (1987)
Adults (female), Sweden 26 D Vahter et al. (1991b)
Children, Poland
0-1 year 225 Olejnik et al. (1985)
1-3 years 259
7-18 years 316
Adults, New Zealand 316 M Pickston et al. (1985)
Children (infant), UK 2-3 breast milk Kovar et al. (1984)
Children (< 1 year), USA 16-17 infant formula Ryu et al. (1983)
& milk
Children, USA
6 months 33.5 M Gartrell et al. (1985b)
2 years 43.4
a M = Market basket survey; D = Duplicate diet study
b Populations studied from the same region.
The relative intake of lead from various sources in 1986 and 1990
in 2-year-old infants and women of child-bearing age in the USA is
shown in Fig. 11. These data illustrate the marked decrease in lead
intake from food over a 4-year period in which there were marked
reductions in the use of lead-soldered cans and lead-containing petrol
additives in the USA (Bolger et al., 1991). Similar decreases in other
countries would no doubt occur after similar actions by public health
officials.
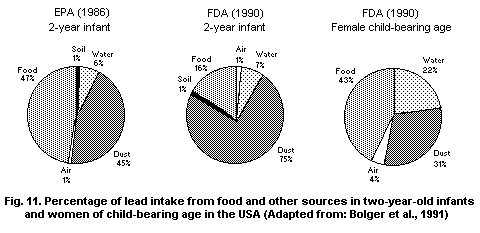 5.2.2.3 Alcoholic beverages
Contamination of alcoholic beverages with lead may occur in
several way. For example, lead solder used to repair casks or keg and
tap lines from lead capsules used as seals or from residues of lead
arsenate pesticides in soils now used to grow grapes. Alcoholic
beverages tend to be acidic and there is the possibility that large
amounts of lead can be dissolved during preparation, storage or
serving (Wai et al., 1979). Published reports on lead levels in wine
show that important variations occur from sample to sample (Jorhem et
al., 1988). The US Department of the Treasury (1991) analysed 432
table wines sold within the USA. The results are summarized in Table
17.
Sherlock et al. (1986) found that the majority of canned and
bottled beer (90 and 86% respectively) contained less than 10 µg
lead/litre. Draught beers typically contained higher lead
concentrations with 55% having lead concentrations greater than
10 µg/litre, 16% with concentrations over 20 µg/litre, and 4% with
concentrations over 100 µg/litre. The higher lead concentrations in
draught beers are considered most likely due to the draught-dispensing
equipment which sometimes contains brass or gunmetal, both of which
contain low but significant amounts of lead (Sherlock et al., 1986).
In general, alcoholic beverages do not appear to be a significant
source of lead intake for the average person.
Table 17. Distribution of lead in table wines in USAa
Range Number of Percentage of total
(µg/litre) samples samples analysedb
0-10 36 8.3
11-25 62 14.4
26-50 105 24.3
51-100 144 33.3
101-250 64 14.8
251-500 12 2.8
501-673 9 2.1
a From: US Department of the Treasury (1991)
b In all, 432 samples were analysed.
5.2.3 Dust and surface soils
5.2.3.1 Dust
Dust is a significant source of exposure to lead, particularly
for young children (see Fig. 11), as has been demonstrated in several
studies correlating children's blood lead concentrations with dust
lead levels (Rabinowitz et al., 1985; Bornschein et al., 1987; Davies
et al., 1987a; Laxen et al., 1987; Steenhout, 1987).
The major contributions to lead levels in soil and outdoor dust
are from the combustion of fossil fuels (principally leaded petrol),
stationary sources such as smelters, and peeling and flaking of
lead-based paint. Typical lead levels in road dust in the USA are
800-1300 mg/kg in rural areas to 100-5000 mg/kg in urban areas (US
EPA, 1989c).
Concentrations of lead in household dust vary greatly between
different dwellings and areas of the world. Mean concentrations of
300-2500 mg/kg have been found in the United Kingdom and USA, but
individual samples may be in the range of 10 000 to 30 000 mg/kg (Que
Hee et al., 1985b; Clark et al., 1985; Bornschein et al., 1986; Raab
et al., 1987).
Flaking lead-based paint, paint chips, and weathered powdered
paint markedly increase intake of lead from surface dust, particularly
for urban children with pica (US EPA, 1986a; Bornschein et al., 1986).
Lead-based paint chips have been found to contain 1000-5000 µg
lead/cm2 (Billick & Gray, 1978). When lead-based paint is present,
interior renovation activities greatly increase household dust lead
concentrations (Laxen et al., 1987). Improved control of dust and
surface clean-up after lead-based paint removal have been shown to
reduce lead exposure of children reoccupying affected houses (Charney
et al., 1983).
5.2.3.2 Soil
In rural and remote areas, lead in soil is derived mainly from
natural geological sources. These natural sources account for 1-30 mg
lead/kg, but where soils are derived from leadmineralized rocks,
natural concentrations may range from several hundred to several
thousand mg/kg.
Typical values for lead in rural soils in the United Kingdom are
15-106 mg/kg with a geometric mean of 42 mg/kg (Davies, 1983). A
geometric mean of 48 mg/kg for 2780 samples has also been reported
(McGrath, 1986).
Concentrations of lead in urban soil vary greatly. In the USA, a
study of city parks recorded concentrations of 200 to 3300 mg/kg (US
EPA, 1989). Concentrations of up to 10 960 mg/kg have been reported
for urban garden soils in the USA (Mielke et al., 1984), and up to 14
100 mg/kg in the United Kingdom (Culbard et al., 1988). Concentrations
can exceed 20 000 mg/kg around lead mining and processing operations
(Culbard et al., 1988). In areas where lead-based paint has been used,
soil samples taken near building foundations have been reported to be
as high as 20 000 mg/kg (Schmitt et al., 1988; Krueger & Duguay,
1989).
In general, lead concentrations in soils near roads are high
where road traffic density is high. Concentrations decrease
exponentially with distance from the road (IPCS, 1989).
Continuous application of sewage sludge results in an
accumulation of lead in soil. For example, soil receiving heavy
applications over a long period was found to contain 425 mg/kg,
compared with 47 mg/kg in an untreated soil (Beckett et al., 1979).
5.2.3.3 Migration of lead from food containers
The available data on the daily intake of lead by adults and
children indicate a general decrease in those areas where the level of
lead in petrol has decreased and a concerted effort made to avoid
lead-soldered cans for food storage (OECD, 1993). However, in many
regions of the world, lead can migrate from food storage and serving
vessels such as lead-soldered cans (see section 5.2.2.2), ceramic
dishes, pottery vessels, crystal glassware and decals on food wrap
and/or dishes. Acidic foods tend to leach more lead. However, certain
foods such as corn and beans are associated with greater release of
lead than would be predicted from their acidity alone (Bolger et al.,
1991). Oxygen appears to accelerate the release of lead from food
containers.
If foods are stored in ceramic or pottery dinnerware that was
lead-glazed and fired in a low temperature kiln, lead can migrate from
the pottery glaze into the food. The glazing process uses a flux,
which is a material that, at high temperatures, reacts with and helps
dissolve the components of the glaze. Lead oxide is a commonly used
flux. Factors that determine whether and to what extent lead will
migrate include the temperature and extent of firing of the pottery in
the manufacturing process, temperature and duration of food storage,
and the acidity of the food. It is extremely difficult to quantify the
extent of such exposures in view of the variations in the
manufacturing processes and the quality control practised in the
country of origin. However, the extent of exposure can be quite
significant, particularly among infants. Cases of lead intoxication
from this source have been reported (Wallace et al., 1985).
Lead has been found to migrate from lead crystal glass into
beverages. This problem is especially severe if beverages are stored
in lead-crystal containers, e.g., decanters or liquor bottles (de
Leacy, 1987; Graziano & Blum, 1991). This phenomenon was not observed
with borosilicate glass containers (de Leacy, 1987).
Several studies have been made of lead contamination of foods and
beverages from lead used in the manufacture or repair of metal
vessels. Recoating the inner surface of brass utensils with a mixture
of lead and tin, described as "tinning", is widely practised by
artisans in India (Vatsala & Ramakrishna, 1985). The tin-lead alloy
contains 55 to 70% lead. Water containing tamarind contained
400-500 µg lead/litre after 5 min of boiling. The practice is
considered to be widely prevalent in at least the three southern
states of India. Zhu (1984) described 344 cases of chronic lead
poisoning in Jiansu Province, China, involving people who had drunk
rain water boiled in tin kettles. Analysis of the lead content of
water showed that after boiling the water contained 0.79 to
5.34 mg lead/litre.
5.3 Miscellaneous exposure
5.3.1 Cosmetics and medicines
Some traditional medicines and customs have been found to result
in exposure to high levels of lead, most of which cannot be quantified
with any degree of accuracy. Rather than occurring as trace
ingredients or trace contaminants, various lead compounds are used as
major ingredients in traditional medicines in numerous parts of the
world (Table 18). Clinically overt lead poisoning due to traditional
cosmetics and medicines has been identified among infants (Shaltout et
al., 1981; Fernando et al., 1981; Sharma et al., 1990), children and
adults (Pontifax & Garg, 1985; Cueto et al., 1989; Mitchell-Heggs et
al., 1990; Gupta et al., 1990). There are case reports of lead
toxicity secondary to inhalation of lead from traditional remedies
(Aslam et al., 1979; Shaltout, 1981; Cueto et al., 1989; Sharma et
al., 1990; Mitchell-Heggs et al., 1990).
Often the use is not limited to adults; these may be used on
infants and young children, as well as on women. In Kuwait, the leaded
"kohl", also called "Al kohl", is traditionally applied to the raw
umbilical stump of the newborn in the erroneous belief of a beneficial
astringent action (Fernando et al., 1981). An additional use of lead
metal and lead sulfide is for inhalation of the fumes ("Bokhoor")
produced from heating on hot coals, in the mistaken belief that this
will calm irritable infants and children (Fernando et al., 1981;
Shaltout et al., 1981).
Latin-American countries also report the use of traditional
medicines with high lead concentrations. For example, the Mexican
traditional remedy "azarcon" (lead chromate) and/or "greta" (mixed
lead oxides), distributed as finely ground powders, may contain more
than 70% lead. They are used in the treatment of "empacho", a
gastrointestinal disorder considered to be due to a blockage of the
intestine (Trotter, 1990).
In addition to the potential risks of lead exposure from the use
of traditional medicines, clinical lead poisoning can result from the
lodging of lead shot in vivo (Manton & Thal, 1986).
Table 18. Sources of lead exposure in traditional medicines and cosmetics
Source of lead Comments Reference
(product)
Summa/Kohl used in Indo-Pakistan and other Muslim Aslam et al. (1979);
cultures as eyes preparation; placed on Fernando et al. (1981);
conjunctival surface or as astringent on Shaltout et al. (1981);
umbilical cord stump. Antimony originally Sharma et al. (1990)
used but lead cheaper.
Hindu folk ground seeds and roots as treatment Pontifax & Garg (1985)
medicine for diabetes (8 mg lead/g)
Bokhoor tribal custom to produce lead fumes to Shaltout et al. (1981)
ward off evil
Azarcon lead chromate and mixed lead oxides as Trotter (1990)
treatment for gastrointestinal disorders
in Mexico and southwestern USA
Skin ointments cosmetics used by Chinese actors; skin Lai (1977)
and cosmetics ointment in Europe
5.4 General population exposure
The total intake of lead by adults and children in the general
population varies greatly as to the relative contributions from
individual sources (air, water, food, soil/dust and others) and is
partly dependent on life-style and socioeconomic status. It is beyond
the scope of this review to provide comprehensive information covering
a wide range of circumstances. However, a few simplified calculations
will be given as guidance for carrying out such determinations.
Table 19 gives a summary of the total lead intake and uptake from
the general environment in adults and in children aged 1 to 5. The
assumptions made are shown and are taken from WHO (1987). Additional
intake of lead will take place in certain groups from the use of
tobacco and alcoholic beverages. The estimates given are probably on
the high side with respect to the contribution from air and dust,
since indoor and outdoor lead contributions were considered equal.
Additional intake of lead is possible due to ciliary clearance of
particles 1-5 µm in diameter with subsequent swallowing and
gastrointestinal absorption.
5.5 Blood lead concentrations of various populations
Under certain conditions, blood lead (PbB) levels are a useful
indicator of exposure and are therefore discussed here, as well as in
section 6.1.4. In general, PbB levels correlate better with recent
exposure levels (Lyngbye et al., 1990b). As is discussed in sections
5.5.1 and 5.5.2, the general trend observed in all blood lead surveys
carried out in countries engaged in risk reduction programmes over the
last 15 to 20 years is a fall in the measured levels (OECD, 1993).
PbB concentrations are the most often used estimate of general
exposure to lead. The USA, Commission of European Communities (CEC),
Australia and the WHO have carried out epidemiological surveys to
determine lead exposures in various populations. The designs of these
studies differed; some provided estimates for the country as a whole,
others compared PbB levels of people working in the same occupation in
various countries, and others surveyed general populations but were
not designed to be extrapolated as national estimates. What these
studies have in common is that they did not emphasize groups with high
lead exposures, but concentrated on typical PbB concentrations of
non-occupationally-exposed groups in the regions or countries studied.
These surveys also offer the possibility of examining the data as
distributions, especially at the high end of the distribution profile.
Such examination may identify unusual exposures and thus populations
at risk. Average PbB concentrations may disguise the risk to various
segments of the population.
In the USA, national estimates of the extent and severity of
recent human exposures to lead in the general population were based on
PbB measurements from the second National Health and Nutrition
Examination Survey of 1976-1980 (NHANES II) (Annest & Mahaffey, 1984).
The design of this study permitted extrapolation to the USA population
as a whole.
Table 19. Estimates of lead (µg/day) absorbed by adults and children
from air, dust, food and watera
Mean air Dust Source of lead (µg/day) Total
lead intake absorbed
concentration (mg/day)b (µg/day)
(µg/m3) Air Dust Food Water
Adults
0.3 N.S. 2.4 -- 10 2 14.4
0.5 N.S. 4.0 -- 10 2 16.0
1.0 N.S. 8.0 -- 10 2 20
2.0 N.S. 16.0 -- 10 2 28
Children 1-5 years
0.3 -- 0.6 -- 25 5 30.6
0.5 -- 1.0 -- 25 5 31.0
1.0 -- 2.0 -- 25 5 32.0
2.0 -- 4.0 -- 25 5 34.0
1.0 25 2 12.5 25 5 44.3
1.0 50 2 25.0 25 5 57.0
1.0 100 2 50.0 25 5 82.0
1.0 200 2 100.0 25 5 132.0
a Adapted from WHO (1987); Dust is not considered a significant
source of lead in adults, but is a significant source for workers
where hygiene practices are poor
The above estimates are based on the following assumptions:
Air: Respiratory volume in adults is 20 m3/day, and in children
5 m3/day, and the respiratory absorption is 40%.
Food: Intake of lead by adults 100 µg/day with 10% absorption and
50 µg/day for children with 50% absorption.
Water: A lead concentration of 20 µg/litre, with adult consumption
of 1 litre/day and 10% absorption and for children 0.5 litre/day
with 50% absorption.
Dust: Dust concentration of lead was 1000 µg/g and absorption was
50%.
b N.S. = Not significant
The CEC study was directed toward biological screening of the
extent of exposure to lead outside the workplace using PbB level as
the index of exposure (CEC, 1981). The object of the study was to
assess non-occupational exposure of the population to lead in the
member states; it was implemented by member states and coordinated by
the CEC. Specific populations studied were selected by member states
and 168 separate areas and population groups were investigated in
1977.
Two other major international studies were limited to urban
populations. A pilot study of human lead exposure (Friberg & Vahter,
1983), organized by the United Nations Environment Programme and WHO,
was a collaborative effort under the Global Environmental Monitoring
System (GEMS). The following countries participated in at least part
of the study: Belgium, China, India, Islamic Republic of Iran, Israel,
Japan, Mexico, Peru, USA and the former Yugoslavia. The subjects of
the study were teachers, since they comprise an occupational group not
extensively exposed to lead. An Australian study was based on urban
residents only, and included 651 subjects between 6 and 91 years of
age (Hopper et al., 1982).
Based on demographic, economic and individual variables found in
the USA study to be associated with PbB levels (Mahaffey et al.,
1982), it is clear that results from these major studies cannot be
directly compared. However, a comparison of such studies can provide
information on the exposure to lead of segments of the various
populations in various countries worldwide.
5.5.1 Adult populations
In a large health screening programme within the USA during
1976-1980 (NHANES II), over 27 000 residents aged 6 months to 74 years
were examined (Annest & Mahaffey, 1984). PbB concentrations were
determined in a subsample (9933 individuals) and yielded an arithmetic
mean value of 0.67 µmol/litre (13.9 µg/dl). In 5841 individuals aged
18-74 years, the overall mean was 0.68 µmol/litre (14.1 µg/dl)
(Roberts et al., 1985). A sex difference was noted: 0.77 µmol/litre
(16.1 µg/dl) was found in males and 0.57 µmol/litre (11.9 µg/dl) in
females. Residents in the centre of large urban areas were found to
have a PbB level of 0.72 µmol/litre (14.9 µg/dl), while rural
residents it was 0.62 µmol/litre (13.0 µg/dl). Because of the design
of the study, information on PbB levels, in relation to smoking,
alcohol consumption, and occupational status, were available. Male
workers in occupations with a high potential for lead exposure had
mean PbB levels of 0.78 µmol/litre (16.2 µg/dl) for nondrinkers/
non-smokers, 0.92 µmol/litre (19.2 µg/dl) for non-drinkers/non-smokers
and 0.95 µmol/litre (19.7 µg/dl) for drinkers/smokers. It was noted
that a true decrease of 37% in PbB level occurred during the period of
the survey (Annest et al., 1983).
Rabinowitz & Needleman (1982) reported an arithmetic mean
umbilical cord PbB concentration of 0.32 µmol/litre (6.6 µg/dl) (range
0-1.78 µmol/litre; 0-37 µg/dl) in over 11 000 samples collected
between 1979 and 1981. A decrease in PbB level of approximately 11%
was noted during the period of collection.
Friberg & Vahter (1983) reported the PbB levels of 200 teachers
from nine countries (see section 5.5). The median PbB level in this
study ranged from 0.29 µmol/litre (6 µg/dl) in Beijing and Tokyo to
1.06 µmol/litre (22 µg/dl) in Mexico City. Smokers generally had
higher PbB levels than non-smokers.
During the period 1978-1988 marked decreases (approximately 30 to
40%) in the average PbB levels of adults were noted in Belgium,
Germany, New Zealand, Sweden, the United Kingdom, and the USA (OECD,
1993).
5.5.2 Children
Results from the NHANES II survey in the USA indicated a mean PbB
level for children (6 months to 2 years old) of 0.78 µmol/litre
(16.3 µg/dl). It was noted that 18.6% of black inner city children had
PbB levels > 1.44 µmol/litre (30 µg/dl) whereas only 4.5% of white
children had such PbB concentrations. The percentage of children with
high PbB level decreased with increasing family income (Roberts et
al., 1985). The NHANES II data were analysed in detail for information
on the demographic correlates of children's PbB concentrations
(Mahaffey et al., 1982; Annest et al., 1983). The racial differences
in these proportions were significant among both boys and girls. The
proportion with elevated PbB concentrations was slightly higher among
boys than girls for both races. Among both black and white children,
the percentage with elevated PbB concentrations decreased with
increasing family income. Proportionately more young children in urban
than rural areas and in the central cities of large urban areas had
elevated blood lead concentrations.
PbB concentrations were measured in 286 Finnish children living
in the three largest cities of Finland (number of subjects = 172), in
rural areas (54 subjects) and in a lead smelter area (60 subjects)
(Taskinen et al., 1981). The mean PbB levels in urban, rural and
lead-smelter areas varied between 0.29 µmol/litre and 0.32 µmol/litre,
range 0.14-0.82 µmol/litre (6.0 and 6.7 µg/dl, range 3-17 µg/dl).
There were no significant differences between the areas of residence.
The five children who lived within 500 metres of the lead smelter
had a mean PbB concentration of 0.44 µmol/litre, range 0.24-0.62
µmol/litre (9.2 µg/dl, range 5-13 µg/dl), which was significantly
higher than the mean blood lead concentration among children living
in the rest of the country.
In a study carried out in Sweden, which included 1781 samples
obtained from children during 1978-1988, the mean PbB level decreased
from 0.29 µmol/litre (59.6 µg/litre), range 0.09-1.20 µmol/litre
(18-250 µg/litre) in 1978 to 0.16 µmol/litre (32.9 µg/litre), range
0.07-0.34 µmol/litre (15-71 µg/litre) in 1988 (Schutz et al., 1989).
In Finland, a remarkably similar decrease has been reported; the mean
PbB value was 0.14 µmol/litre (30 µg/litre), range 0.1-0.40 µmol/litre
(21-41 µg/litre) among 35 children in the Helsinki area in 1988 (Ponka
et al., 1991).
As in the USA (Annest et al., 1983), children's PbB
concentrations in Sweden have decreased in recent years. Levels were
determined each summer during the period 1978-1984 in children from
Scania in Southern Sweden (Skerfving et al., 1986). The average PbB
concentration was 0.26 µmol/litre, range 0.07-1.20 µmol/litre
(5.5 µg/dl, range 1.4-25.0 µg/dl). There was a statistically
significant decrease over time in both the rural and urban areas,
averaging about 0.019 µmol/litre (0.4 µg/dl) per year.
Decreases of 25-45% in average PbB levels in children between
1978 and 1988 have been reported in Belgium, Canada, Germany, New
Zealand, Sweden and the United Kingdom (OECD, 1993).
5.5.3 Remote populations
In contrast, a much higher mean value of 0.16 µmol/litre
(3.4 µg/dl) was obtained in Nepal upon examination of 103 children
living in the Manang District, which is also very remote and thought
to be free of anthropogenic sources of lead (Piomelli et al., 1982).
However, these people are exposed to significant amounts of natural
lead from living in dusty smoke-filled houses, and from burning pine
and yak dung. The lead level in the houses was about 0.15 µg/m3
(Davidson et al., 1981). For comparison outdoor air in Woods Hole,
Massachusetts, USA, contains 0.004 and Boston, Massachusetts, 0.01 to
0.05 µg lead/m3.
Another remote population studied was from Okapa, Eastern
Highlands, Papua New Guinea (Poole et al., 1980). Extreme care was
taken in sampling and analysis, and the precision was acceptable at
PbB levels as low as 0.24 µmol/litre (5 µg/dl). The children subsisted
on root crops and a small amount of tinned fish and meat. Villages
were typically hazy with wood smoke, natural mineral pigments were
used extensively, and tobacco smoking was common. This survey of 100
children yielded a mean PbB level of 0.25 µmol/litre (5.2 µg/dl), well
above the detection limit, and, as reported by Poole et al. (1980),
far less than the mean of 1.10 µmol/litre (23 µg/dl) for Sydney,
Australia, in 1974.
5.6 Occupational exposure
Occupational exposure to lead which results in poisoning, both
moderate and clinically symptomatic, still occurs in many countries of
the world. Although adults are mainly involved, in many countries,
especially in those with developing industries and small home-based
industries, the distinction between home and workplace is non-existent
(Verrula & Noah, 1990) and children are exposed to workplace lead.
Until recently, in the occupational setting, there was only concern
for the identification of late stage, highly symptomatic cases of lead
poisoning resulting in crippling neurological manifestations of lead
poisoning such as palsy and encephalopathy (Chakravorti & Bhar, 1978;
Wang, 1984; Davies, 1984; Lin-Fu, 1985). However, there is now concern
for lower exposures to lead.
In many countries occupational lead exposure is entirely
unregulated and no monitoring of exposures exists. Automobile battery
manufacture and repair, radiator repair, secondary smelters (including
scrap metal refiners) are found in most countries. The industries
where there is a potential for lead exposure are listed in Table 10
(section 5.1.3). Significant lead exposures are not limited to
traditional heavy industries. For example, Kaye et al. (1987)
identified exposures from leadborosilicate dust used in a capacitor
and resistor plant in the USA.
Because of transfer of lead to the fetus (in utero) and the
transport to the home of lead on clothing, etc., thereby exposing the
young child in the home, the problems of occupational exposures to
lead are not limited to the workplace per se. Cases of poisoning
among the children of lead workers have been reported (Baker et al.,
1979; Wang, 1984; Ryu et al., 1985).
Although most occupational standards are based on airborne lead
only, this route of exposure does not fully reflect the total daily
exposure of workers, also exposed to lead in food, water, alcoholic
beverages and dusts.
The potential for hazardous exposures to lead during lead
smelting and refining are well recognized, particularly where molten
lead and alloys are poured, resulting in the vaporization of metal.
This is true for both primary new metal and secondary (lead scrap)
smelters and refineries.
Small domestic versions of secondary smelters exist in a large
number of countries. These are typically located within or in close
proximity to homes. For example, in Jamaica there has been a rapid
proliferation of lead smelters, particularly illegal backyard smelters
(Rodney & Lee, 1985). The lead fumes and dust generated in such
operations pose an exceptional health hazard to children and adults
living near these operations. Rodney & Lee (1985) reported that 51% of
116 children (aged 2 to 12 years, mean 5.9 years) and 60% of 235
adults working in or living near lead smelting factories had PbB
concentrations of 1.92 µmol/litre (40 µg/dl) or more.
Other occupations where workers have been shown to be at risk
from airborne lead include electric storage battery manufacturing,
particularly where industrial hygiene is poor (Barrio & Badia, 1985);
demolition, welding and shipbreaking where lead-based paint is present
(Holness & Nethercott, 1988; Nosal & Wilhelm, 1990); pottery and
ceramic-ware production, which is often a home-based operation
involving women and children (MolinaBallesteros et al., 1983; Katagiri
et al., 1983; Kaye et al., 1987); small businesses repairing
automobile radiators (Matte et al., 1989a,b; Verrula & Noah, 1990),
and artisans producing jewellery and decorative wares. This latter
industry is of particular concern since it is predominantly carried
out at home or in non-regulated shops, often by women and children
(Behari et al., 1982). Indian silver jewellery makers were found to
have a PbB level of 5.8 µmol/litre (121 µg/dl) compared to non-exposed
controls with a PbB level of 1.3 µmol/litre (27 µg/dl) (Behari et al.,
1982).
6. KINETICS AND METABOLISM IN LABORATORY ANIMALS AND HUMANS
6.1 Absorption
The absorption of lead from environmental sources is not
dependent solely on the amount of lead presented to the portals of
entry. It is also dependent on the physical and chemical state in
which the metal is presented, and it is influenced by host factors
such as age, physiological status, nutritional condition and,
possibly, genetic factors. Men engaged in heavy work breathe more air
and eat more food than sedentary individuals of the same weight, and
on a body weight basis children eat almost as much food and breathe
almost as much air as middle-aged adults.
Considerable data from human subjects are available. Therefore,
discussion of animal studies will be limited to areas where the
information on humans is inadequate. Various models used to predict
body burden or distribution of lead have been developed (Bernard,
1977; Marcus, 1985a,b,c; Bert et al., 1989; Arnetz & Nicolich, 1990).
A full review of these models has been judged to be beyond the scope
of this document. However, some of them were used in section 6.1.4
when correlating intake and body burdens.
6.1.1 Absorption after inhalation
The absorption of lead from air to blood involves two processes:
the deposition of airborne lead particles in the respiratory tract;
and the absorption and clearance from the respiratory tract into the
circulation. Using the International Radiological Protection
Commission (IRPC) document on lung dynamics (Task Group on Lung
Dynamics, 1966), a model was developed which predicted that 35 to 50%
of inhaled lead is deposited in the respiratory tract (40-50% of
particles with a mean mass median aerodynamic diameter (MMAD) of
0.5 µm, such as are typically generated by automobiles). These are
deposited primarily in the alveolar sacs of the lung. Lead fumes and
vapours, such as those generated in operations where metals are cut or
heated, are of very small size and are respirable. Absorption after
deposition will vary according to the solubility of the lead species
(e.g., lead carbonate or lead chloride aerosols) and the inherent
toxicity to lung macrophages and cilia.
6.1.1.1 Animal studies
Limited animal studies confirm that there is almost complete
absorption of lead particles (0.1 to 0.5 µm in diameter) deposited in
the lower respiratory tract. In rats the clearance half-time from lung
is short (less than one hour) and 90 to 98% of the administered dose
is absorbed within about 48 h (US EPA, 1986a).
6.1.1.2 Human studies
The respiratory deposition of airborne lead is in the range of
30-50% and varies with particle size and ventilation rate (US EPA,
1986a). Higher deposition rates may occur with larger particles, but
this deposition takes place in the upper respiratory tract, with
eventual displacement to the gastrointestinal tract and absorption via
the ingestion route. This probably explains the observation of Kehoe
(1961) that faecal excretion of lead increased after a subject
breathed for many weeks aerosols of lead oxide (150 µg/m3) with an
MMAD of approximately 2.9 µm. In contrast, smaller particles of
inhaled lead, such as those generated by automobile exhaust,
regardless of physicochemical form, are almost (> 90%) completely
absorbed after deposition in the lower respiratory tract (Rabinowitz,
et al., 1977a; Chamberlain et al., 1978; US EPA, 1986a).
There are no quantitative data on the absorption of lead in
children after inhalation exposure. It is known that young children
weighing only one sixth of an adult inhale 40% of the daily volume of
an adult and a proportionately higher daily air volume per unit
measure (weight, body area) than do adults (Barltrop, 1972). After
controlling for weight and taking into account differences in the
anatomy of the respiratory tract between adults and children, James
(1978) calculated a rate of deposition of lead particles in children
which was 1.6 to 2.7 times that of adults.
6.1.2 Absorption of lead from the gastrointestinal tract
In the case of older children and adults without occupational
exposure, lead absorbed by the gastrointestinal tract comes from the
intake of lead in foods, beverages and soil/dust. In pre-school
children, there is concern over the intake of both food and non-food
items (e.g., toys, soil/dust). Young children may take in lead from
non-food items, via normal mouthing activity, which in the extreme, is
the behavioural trait pica, which refers to the ingestion of such
materials as soil, ash, paint chips and plaster (US EPA, 1986a). For
infants and young children, the extent of absorption of the lead in
dust/soils from the gastrointestinal tract is extremely important,
particularly for children living in urban environments.
6.1.2.1 Animal studies
The absorption of lead from the gastrointestinal tract in
experimental animals is age dependent and is modified by the level of
food intake. In 1-week-old suckling rats, an absorption rate of about
52% was reported after a single oral dose of lead chloride, compared
to 0.4% in 6-week-old animals (Kostial et al., 1978). Furthermore,
Aungst et al. (1981) reported that rat pups had higher tissue levels
of lead than adults after a single gavage dose of 1 or 10 mg lead (as
lead acetate) per kg body weight. Fasting markedly enhanced the uptake
of lead in the gastrointestinal tract of experimental animals (Garber
& Wei, 1974; Pounds et al., 1978).
In experimental animals, absorption of lead from the
gastrointestinal tract appears to be a saturable process. With
increasing doses (1 to 100 mg lead (as lead acetate) per kg body
weight), lead absorption as a percentage of dose was found to decrease
from 42% to 2% (Aungst et al., 1981; Bushnell & DeLuca, 1983) in
dietary studies. Such data are consistent with a saturable active
transport process across the gastrointestinal tract.
The low rate of absorption of lead from soluble lead salts noted
in adult rats may reflect a dietary effect. Kostial & Kello (1979)
reported less than 1% absorption in adult rats given chow diets,
whereas the absorption rate was 3-20% when rats consumed diets similar
to that of humans (milk, bread, baby foods, etc). This range more
closely resembles the human absorption rates.
The chemical form of lead can influence its bioavailability.
Stone et al. (1981) determined the biological availability of lead
intrinsically incorporated into the soft tissues of oysters. The
bioavailability to the rat was 10-30% lower than that of lead acetate
added to the basal chow diet. Henning & Cooper (1988) compared the
absorption of lead from rat milk labelled in vitro with lead-203 and
lead chloride or lead acetate solutions. Lead from soluble salts
accumulated primarily in the duodenum, to some extent in the jejunum
and minimally in the more distal small intestine. Lead from milk
accumulated only in the upper ileum. After 20 h negligible lead-203
was found in any region following administration of soluble salts, but
there was substantial retention of lead in the ileum after dosing with
milk.
Data obtained from a feeding study in rats (Dieter et al., 1993)
showed that lead uptake into rat femurs was highly dependent on the
chemical form of lead administered. Bioavailability was highest for
lead acetate, intermediate for lead oxide, and lowest for lead sulfide
and Alaskan mixed ore concentrate. This uptake was linearly related to
dose over the range studied. However, the slopes of the linear
regression equations differed according to the form of lead. They were
0.10 µg lead/g femur per kg diet for lead sulfide and 2.64 for lead
acetate.
However, results of an investigation in young swine of the
absorption of lead from mining and/or milling operation waste (LaVelle
et al., 1991) differ from those reported by Dieter et al. (1993). The
chemical species (predominantly lead sulfide) and particle size (i.e.
larger than 100 µm) were expected to result in these sources being
less bioavailable than soluble lead salts such as lead nitrate.
However, experimental data indicate that the lead in mine tailings may
be about 2-3 times more available than reagent grade lead sulfide
under the conditions of the study. Additionally, Freeman et al. (1992)
reported that the lead in mining waste soil was between 8 and 20% as
bioavailable as lead acetate. No detailed comparisons were made of the
physicochemical properties of these two mine wastes. An additional
study involving the absorption of lead from lead sulfide ores and
their oxidation products in rabbits and in vitro showed that lead
from minerals was absorbed less well than from lead acetate by a
factor of 5 (Davis et al., 1990a). The significance of these phenomena
is not clear.
6.1.2.2 Human studies
Gastrointestinal absorption of lead in humans, as in experimental
animals, is influenced by dietary factors, nutritional status and the
chemical form of the metal. Overall patterns of food intake may also
influence lead absorption. For example, lead ingested during periods
of fasting is absorbed to a much greater extent than lead ingested
with food. Chamberlain et al. (1978) reported 45% absorption of lead
chloride in fasting subjects and only 6% in feeding subjects. Using a
similar procedure, Heard & Chamberlain (1982) reported absorption of
63.3% in fasting subjects. Using multiple stable isotope lead tracers,
it was found that in adult men the gastrointestinal absorption rate
with food containing lead nitrate or lead cysteine was 6-12%. However,
when consumed under fasting conditions, lead nitrate, lead sulfide and
lead cysteine were absorbed at 16-53% (Rabinowitz et al., 1977b).
Kehoe (1961) estimated only 10% net absorption of dietary lead by
adults in long-term metabolic studies. However, with appropriate
calculation of biliary clearance as well as of urinary excretion, a
figure of 15% was estimated by Chamberlain et al. (1978). In several
studies on adult humans, absorption of lead was reported to be 14%
when it was administered with food (Chamberlain et al., 1978; Moore et
al., 1979; Rabinowitz et al., 1980).
Ziegler et al. (1978) reported that young children, aged two
weeks to two years, absorbed 42% of ingested lead at levels of intake
greater than 5 µg/kg body weight. Drill et al. (1979) estimated an
absorption rate of 17% for lead in paint chips in children aged 2 to 3
years. These authors also estimated a 30% gastrointestinal absorption
rate for lead in soil and dirt.
The amount of lead ingested by children from non-food items such
as soil, dust and paint chips through normal mouthing activity,
particularly for children with pica, is a major concern in calculating
paediatric lead exposures. Using data from Day et al. (1975) and Lepow
et al. (1974), it has been estimated that children 2 to 3 years of age
ingest about 100 mg soil per day. Using aluminium and titanium as
tracers, Clausing et al. (1987) estimated that children aged 2 to 4
years ingest between 50 and 100 mg of soil daily. The average amount
of soil ingested by young children has recently been estimated to be
between 12.5 and 21 mg/day (SAHC, 1993).
6.1.2.3 Nutritional status and lead absorption via gastrointestinal
tract
It has been known for some time that the absorption and
distribution of lead are affected by nutritional status in both
experimental animals and humans (Sobel et al., 1940). Nutritional
inadequacies can also affect the toxic response to lead (see sections
7 and 8). In view of documented nutritional inadequacies in many parts
of the world, such interrelationships become crucial in assessing the
full risk from lead exposures.
Vitamin D, calcium and phosphorus have complex and interrelated
effects on lead absorption (Fullmer, 1990). Increasing the
concentration of 1,25-dihydroxycholecalciferol, the active metabolite
of vitamin D, either exogenously or endogenously, increases
gastrointestinal absorption of lead (Fullmer, 1990). However, this
effect is dependent upon the duration of lead exposure and the
magnitude of body lead stores. This homoeostatic mechanism for calcium
and its dependence on nutritional status, as well as body burden of
lead, is complex. This may explain the divergent results of the
observed interaction in children (Rosen et al., 1980) or the lack of
association (Koo et al., 1991) of lead with vitamin D metabolism.
In experimental animals, chronic ingestion of diets with less
than adequate amounts of calcium (Quarterman & Morrison, 1975),
phosphorus (Quarterman & Morrison, 1975), iron (Mahaffey-Six & Goyer,
1972; Ragan, 1977), selenium (Stone & Soares, 1976) or zinc
(Cerklewski & Forbes, 1976) increases the fractional absorption of
lead in the gastrointestinal tract. From the work of Fullmer & Rosen
(1990), it appears that lead alters the transport of calcium and other
trace minerals by affecting carrier proteins, rather than by competing
at the mucosal surface.
The influence of iron on lead absorption in humans was assessed
in double-labelling experiments conducted by Watson et al. (1980).
Lead-203 and iron-59 were given to 28 subjects in their diet. There
were significant positive correlations between iron levels in food and
lead absorption. However, the association was not strong because 50%
of those absorbing excess lead also absorbed excess iron.
Rats fed iron-deficient diets have increased concentrations of
lead in kidney and bone when compared to rats ingesting equivalent
quantities of lead (as lead acetate) in drinking-water while being
fed an iron-adequate diet (Mahaffey-Six & Goyer, 1972). In contrast
with calcium deficiency, iron deficiency does not result in a
redistribution of lead to non-osseous tissue (Mahaffey-Six & Goyer,
1972). The degree of iron deficiency does not need to be severe to
increase retention of lead. For example, Ragan (1977) demonstrated
six-fold increases in tissue lead in rats when body iron stores were
reduced, but before frank iron deficiency developed. High levels of
dietary iron resulted in decreased kidney, femur and blood lead
concentrations in rats (Mahaffey, 1985). Iron appears to increase
absorption rather than decrease lead excretion (Barton et al., 1978).
Lead interferes with normal transferrin transport of iron, and
inhibits transferrin endocytosis and iron transport across the cell
membrane of the reticulocyte (Qian & Morgan, 1990).
6.1.3 Dermal absorption
6.1.3.1 Human dermal absorption
Moore et al. (1980) examined the uptake of lead acetate from two
hair-darkening cosmetics through the skin of eight human volunteers.
Only minute quantities of lead (0-0.3% of the applied dose) were
detectable in blood, and there was only a slight increase in
absorption when the skin was damaged. Lilley et al. (1988) and
Florence et al. (1988) have reported the dermal absorption of
inorganic lead compound leading to elevated levels of lead in human
saliva and sweat.
Dermal absorption of inorganic lead through unabraded human skin
is considered to be minimal.
6.1.4 The relationship of external lead exposure to blood lead
concentration
There are numerous environmental media that provide routes by
which humans are exposed to lead: food, water, air, soil, dust.
External exposures are the sum of the quantities of lead consumed from
all sources.
Historically there have been two lines of approach in
understanding the lead exposure and blood lead relationship. Most have
been empirical and have measured environmental lead and PbB levels
either at one time or repeatedly. With these observed correlations and
with no assumptions about how lead moves inside the body, many
reasonable predictions can be made. The validity of these predictions
is optimum when only one environmental source dominates. Some of these
rely on linear functions, while others have specified non-linearities,
especially over very wide ranges (more than a factor of 10) of lead
exposures. However, when multiple sources are considered these
predictive models have been less satisfactory.
Internal lead levels in human populations have been estimated by
analyses of a variety of biological tissues (e.g., blood, teeth, bone
and hair). Lead concentrations in each of these have particular
biological meanings with regard to external exposure to lead. Blood is
the compartment in which lead is most often measured as a marker of
exposure. However, it typically represents relatively recent
exposures, since the half-life of lead in blood is short (US EPA,
1986a) and has been estimated to be in the order of 36 days from
tracer studies (Rabinowitz et al., 1975). Lead in blood is derived
from levels in the current environment and from lead stored in tissues
(particularly bone) that re-enters the blood during tissue
mobilization (Manton, 1985). Although PbB concentration reflects
recent exposure and bears a consistent relationship to levels of lead
in the external environment, when bone mobilization is accelerated a
greater fraction of PbB will be derived from tissue stores.
To date only a few studies have utilized a multimedia approach
relating lead intake to PbB levels. The majority of studies have
attempted to correlate PbB levels and lead concentrations in specific
media.
6.1.4.1 Ambient air
a) Occupational exposure
Several studies have examined the blood lead: air lead
relationship for workers exposed to levels of airborne lead between 9
and 450 µg/m3 (King & Conchie, 1979; Gartside & Buncher, 1982;
Bishop & Hill, 1983). The results of all three studies are in general
agreement over the wide range of airborne lead studied and for PbB
levels between 0.96 and 4.32 µmol/litre (20-90 µg/dl). The blood lead:
air lead relationship in occupational settings is best described by a
curvilinear relationship having slopes between 0.00096 and
0.0038 µmol/litre (0.02 and 0.08 µg/dl) per µg/m3 air.
b) Non-occupational exposure
Both population and experimental studies have been used to
estimate the PbB: ambient air lead relationship in adults and
children. Under ambient conditions (air lead concentrations of
0.1-2.0 µg/m3) and PbB levels less than 1.44 µmol/litre
(< 30 µg/dl), the relationship has been described as linear (Colombo,
1985). The various slope estimates reported are based on the
assumption that an equilibrium level of lead in blood is achieved.
Based on three major studies (Yankel et al., 1977; Angle & McIntire,
1979; Roels et al., 1980), the median slope for children is about
0.091 µmol/litre (1.9 µg/dl) blood per µg/m3. In adult males a slope
estimate of 0.076 µmol/litre (1.6 µg/dl) blood per µg/m3 was
calculated. When one calculates the relationship between PbB and the
total contribution from air (direct inhalation plus indirect through
dust/soil), a value of about 0.14-0.24 µmol/litre (3-5 µg/dl) blood
per µg/m3 is obtained (Brunekreef, 1984; US EPA, 1986a). Also on the
basis of a linear model, Snee (1981) reported that the best estimate
of the blood lead: air lead relationship was 0.048 µmol/litre
(1 µg/dl) per µg/m3. After examining available epidemiological and
experimental data, Chamberlain (1983) concluded that most published
estimates of the slope were between 0.072 and 0.144 µmol/litre per
µg/m3. From these results, it can be concluded that airborne lead
will only be a major contributor to PbB levels in areas of high air
lead levels.
6.1.4.2 Food
The relationship of PbB to dietary intake has been estimated from
experimental (Stuik, 1974; Cools et al., 1976; Schlegel & Kufner,
1979) as well as population studies (Sherlock et al., 1982; UK, 1982;
Ryu et al., 1983). In adults, the results from both types of studies
using both linear and cube root models indicated a relationship of
between 0.0019 and 0.0028 µmol lead/litre (0.04 and 0.06 µg/dl) per µg
lead intake per day. From the study of Ryu et al. (1983) a slope of
0.0096 µmol/litre (0.2 µg/dl) per µg lead/day was obtained for infants
aged 8 to 196 days.
Currently, data are available for adults and children from
studies with careful control of important variables such as: intake of
dietary lead and of other dietary constituents, minimal exposure to
sources other than diet in the studies of infants, and intake/blood
lead measurements that can be used to estimate intake from all
sources. For infants these are studies reported by Sherlock et al.
(1982), UK (1982), Ryu et al. (1983), and Lacey et al. (1985). Studies
by the UK (1982) and by Sherlock et al. (1982) were conducted at
higher levels of lead exposure than were the studies conducted with
infants by Ryu et al. (1983). The latter studies were at exposure
levels associated with PbB concentrations under 0.96 µmol/litre
(20 µg/dl); the ratio of blood lead to ingested lead was
0.0076 µmol/litre (0.16 µg/dl) per µg lead ingested per day. Exposure
levels were low: average dietary lead intake was 17 µg/day. After four
months, the average PbB concentration was 0.29 µmol/litre (6.1 µg/dl)
whole blood. Between the ages of 4 and 6 months, 10 children remained
at a dietary lead intake of 0.76 µmol/litre (16 µg/dl). Their PbB
concentrations were quite constant at 0.35 µmol/litre (7.2 µg/dl) at
the end of the study period. The remaining seven children received
canned infant formula and/or milk during this period and their average
dietary lead intake was 61 µg/day. At the end of the study period,
their PbB concentration was 0.69 µmol/litre (14.4 µg/dl). Based on
these data, a curvilinear relationship between blood lead and total
lead intake was suggested, a 4-fold increase in lead intake resulting
in a doubling of PbB concentration. Sherlock et al. (1982) conducted a
duplicate diet study of 31 mothers and their children from Ayr,
Scotland. The slope for adults was substantially lower than for
children.
6.1.4.3 Drinking-water
There is still debate over the most appropriate model (i.e. cube
root, polynomial or logarithmic) to describe the curvilinear
relationship of waterborne lead to blood lead. The highest estimates
for the contribution of water lead to blood lead come from the cube
root and logarithmic models. Much lower estimates are obtained from a
linear model (Pocock et al., 1983; US EPA, 1986a). In a study by
Sherlock et al. (1982), a cube root relationship between lead levels
in drinking-water and blood fitted the data more closely than a linear
relationship. The curvilinear model implies that as abatement
processes lower water lead concentrations, there will be an increasing
benefit in lowering of population PbB levels (Moore, 1983).
6.1.4.4 Soil and dust
It is extremely difficult to choose the most appropriate model to
describe the soil/dust to blood lead relationship, given the many
variables involved in determining the exposure patterns of children
and the kinetics involved between the levels in the environment and
the child. A review of the available studies shows the extreme
variability in slopes obtained (0.028-0.36 µmol lead/litre
(0.6-7.6 µg/dl) blood for each 1000 µg/g soil and 0.00096-0.35 µmol
lead/litre (0.02-7.2 µg/dl) blood for each 1000 µg/g dust consumed by
children) (US EPA, 1986a). Detailed consideration has been given to
the process for the assessment of lead contamination in soil and the
derivation of soil clean-up criteria by Wixon (1991). The guideline
model uses a PbB target and slope for the soil:blood lead relationship
in the particular community in order to derive a PbB guideline. For
house dust a median value of 0.086 µmol lead/litre (1.8 µg/dl) blood
per 1000 µg lead/g dust in children appears to be based on data of
reasonable quality, as does the 0.105 µmol lead/litre (2.2 µg/dl)
blood per 1000 µg lead/g soil from the same authors.
According to Elwood (1986), studies of the association between
lead in house dust and PbB have given inconsistent results and, in
general, the only studies in which statistically significant
association has been found are those where lead levels in dust are
quite high. Landrigan et al. (1975) found a highly significant
association between PbB and dust lead in an area with a mean level of
4022 µg lead/g dust, but not in two areas with means of 922 and 816 µg
lead/g dust. The US EPA (1986a) summarized a series of studies from
which the overall relationship was judged to be 0.86 µmol/litre
(18 µg/dl) blood per 1000 µg lead/g dust. Duggan (1980) reviewed the
literature and determined that a slope of 0.24 µmol/litre (5 µg/dl)
per 1000 µg lead/g dust is reasonable.
The overall relationship between PbB and dust/soil lead depends
on the lead concentrations and bioavailability as well as on the
proximity and linkage between humans and their environment. This
relationship varies among locales.
6.1.4.5 Total lead intake
The non-linear relationship between PbB and total lead intake
(see section 5.5) is curvilinear across a broad range of PbB values,
such that the slope decreases with increasing lead levels. A number of
biological factors may explain the curvilinear relationships, such as
increased renal clearance with high PbB (Chamberlain, 1983, 1985),
distributional non-linearities due to differences in lead binding
sites in different tissues (Hammond et al., 1981; Manton, 1985;
Marcus, 1985b), or a sizeable pool of mobile lead in bone maintained
more or less independently of uptake (Rabinowitz et al., 1976;
Chamberlain, 1983).
6.2 Distribution
The initial distribution of lead in the body may depend upon the
rate of delivery of blood to various organs. However, it would appear
that distribution occurs in a similar manner regardless of the route
of absorption (Kehoe, 1987). The distribution of lead in humans under
environmental exposure conditions reflects the fact that almost all
exposures are chronic rather than acute.
6.2.1 Animal studies
Studies in rats have shown that lead is rapidly distributed into
soft tissues and subsequently redistributed into soft and mineralizing
tissues after acute and chronic exposures. After acute inhalation
(Morgan & Holmes, 1978) or oral (Aungst et al., 1981) exposure, lead
levels in rats were highest in liver, kidneys and lung, with levels
increasing in bone as those in soft tissues declined and stabilized.
Similar distribution patterns were reported for mice exposed for 12
months to 21.5 µg lead/m3, the highest levels being found in bone
and the lowest in lung (Keller & Doherty, 1980a).
Age-related differences in the distribution of lead in
experimental animals have been reported. Kostial et al. (1978) noted a
greater retention of lead in suckling rats than in adults; the levels
in the brain were also higher in the pups. A 2- to 3-fold increase in
brain lead concentration (highest in hippocampus) was noted after a
10-fold (0.1 to 1 mg/kg body weight administered by gavage) increase
in the dose given to 4- to 8-week-old rat pups (Collins et al., 1992).
Ageing has been shown to alter the pattern of distribution of
lead in rats administered lead acetate in drinking-water. Juvenile (21
days old), adult (8 months old) and elderly (16 months old) rats
received 0, 1.27 and 6.37 mg lead/kg bodyweight for 9.5 months. The
pattern of distribution, namely femur > liver > brain was similar in
all age groups. However, age-related increases in brain lead levels
were noted, along with decreases in femur lead content (Cory-Slechta,
1990a).
6.2.2 Human studies
Lead is distributed to both soft tissues (blood, liver, kidney,
etc.) and mineralizing systems (bone and teeth). Bone may be affected
adversely by lead but also serves as the body's major storage site.
Bone accumulates lead over much of the human life span, and a study of
the kinetics of distribution is important since bone can, under
appropriate conditions, pose a risk as a potential endogenous source
of lead.
Once absorbed, lead is not distributed homogeneously throughout
the body but rather into several distinct compartments. Such
biokinetic movements have been explained by Rabinowitz et al. (1976)
using a three-compartment model. This model was based on tracer and
balance data from five healthy men and has been refined by Marcus
(1985a,b,c). Three pools (blood, bone and soft tissues) were
identified, with lead having distinct half-lives in each. Blood lead
was considered the most labile compartment with a half-life of about
36 days, and bone lead the most stable with a half-life of about 27
years. Lead in soft tissue had a half-life of approximately 40 days.
The specific physiological foundations for these biokinetic
models have received much attention and are currently being refined
(O'Flaherty, 1991, 1993). A more thorough understanding of bone
growth, for example, expressed as a series of allometric equations,
should help improve models, especially when they must be applied to
growing and maturing humans.
Under steady-state conditions, about 96% of PbB is in the
erythrocyte. At PbB concentrations of < 1.92 µmol/litre (40 µg/dl),
whole blood and serum lead levels increase linearly in a positive
manner. At higher PbB concentrations a curvilinear relationship is
apparent and the serum to blood ratio increases dramatically (Manton &
Cook, 1984). Such kinetic relationships may be altered during
pregnancy. From in vitro data (Ong & Lee, 1980), fetal haemoglobin
appears to have a greater affinity for lead than adult haemoglobin.
In adults, approximately 94% of the body burden of lead is in the
bones, whereas only 73% of the body burden in children is located in
this compartment (Barry, 1975, 1981). In view of the extremely long
half-life for lead in bone, this compartment can serve as an
endogenous source of lead to other compartments long after exposure
ceases (O'Flaherty et al., 1982; Kehoe, 1987). This is due to the
labile lead compartment in bone (Rabinowitz et al., 1976) and the
ongoing bone redistribution which is subject to alteration by in vivo
metabolic processes (Parfitt, 1990). Although lead in bone generally
increases continuously with age, there is evidence that lead levels in
some bones (e.g., mid-femur and pelvic bone) plateau at middle age and
decrease with further ageing (Drasch et al., 1987). It is difficult,
however, to determine the role ageing plays in this process compared
to other biological reactions. Ageing may account in part for the
20-25% increase in PbB levels in menopausal women noted by Silbergeld
et al. (1988).
The metabolism of lead in bone has been summarized in reports by
Barry (1975, 1981), Drasch et al. (1987), Silbergeld et al. (1988),
Skerfving (1988) and Rabinowitz (1991).
Until recently, it was widely held that the human skeletal system
provided a metabolically inert depository for lead and was of little
consequence in health-risk assessment. It had been assumed that bone
lead is metabolically inert, with a half-life long enough to forestall
the risk of ready transfer back to blood. Current evidence is that
bone comprises a set of kinetically variable subcompartments for lead
deposition and is a target for toxicity. These factors complicate bone
lead kinetics as applied to long-term modelling; the mobility of bone
lead to blood is important (Rabinowitz, 1991).
Bone lead is readily mobilized to blood and the effect is most
apparent in people with a history of occupational exposure; bone lead
also appears to be a major source of blood lead in older people with
previous ambient exposures to lead. Of particular importance is
mobilization of lead from bone in pregnant women and nursing mothers
(Silbergeld, 1991). The mobilization of lead from bone to the more
bioavailable maternal blood compartment poses a risk to the fetus and
mother.
Human bone appears to have at least two, possibly three,
kinetically distinct lead compartments. Lead in trabecular (spongy)
bone appears to be more mobile than lead lodged in cortical (compact)
bone, and there appears also to be a fraction of bone lead in
equilibrium with the lead in blood (Skerfving, 1988). Trabecular bone
seems to be an important source of resorbed lead when high exposure is
reduced, e.g., through removal of medical reasons by retirement of
lead workers, or in response to chelation in adults (Shutz et al.,
1987).
6.2.3 Transplacental transfer
Lead is readily transferred from the mother to the developing
infant during pregnancy and accumulates in bone during gestation
(Barltrop, 1969). The lead concentration in cord blood is 85-90% that
of maternal blood. Moore et al. (1982) reported a geometric mean level
of 0.67 µmol/litre (14 µg/dl) in 236 pregnant women and
0.58 µmol/litre (12 µg/dl) for lead in umbilical cord blood. The mean
concentration of lead in umbilical cord blood from a sample of over
11 000 women was 0.298 µmol/litre (6.6 ± 3.2 µg/dl) (Bellinger et al.,
1987).
6.3 Elimination and excretion
In both humans and experimental animals lead is eliminated from
the body in both urine and faeces. Any dietary (including waterborne)
lead not absorbed in the gastrointestinal tract is excreted in faeces.
Airborne lead that has been swallowed and not absorbed is also
eliminated in this manner. Blood lead not retained in the body is
excreted in urine or faeces, the latter by biliary excretion. Adults
ingesting daily 0.3 to 3.0 mg lead (as lead acetate) in drinking-water
for 16 to 208 weeks excreted more than 85% of the ingested lead, 90%
of the excreted lead being in the faeces. The amount excreted through
any route is affected by age and exposure characteristics and is
species dependent (US EPA, 1986a). This section reviews only studies
of lead excretion in humans; a discussion of the results of work on
experimental animals can be found in reviews by US EPA (1986a) and
ATSDR (1993).
The age dependency of lead excretion in humans has not been
studied extensively. However, the studies of Rabinowitz et al. (1977b)
and Chamberlain et al. (1978) in adults and Ziegler et al. (1978) in
infants can be used to assess this phenomenon. Data from Ziegler et
al. (1978) and Rabinowitz et al. (1977a) are given in Table 20.
Ziegler et al. (1978) calculated a total daily retention by infants of
40 µg, which is about twice the amount calculated by Alexander et al.
(1973).
Table 20. Comparison of daily lead intake and excretion in children
and adultsa
Parameter Childrenb Adultsc
Dietary intake (µg/kg) 10.76 3.63
Fraction absorbed 0.55d 0.15
Dietary lead absorbed (µg/kg) 5.92 0.54
Air lead absorbed (µg/kg) 0.20 0.21
Total absorbed lead (µg/kg) 6.12 0.75
Urinary lead excreted (µg/kg) 1.00 0.47
Endogenous faecal lead (µg/kg) 1.56 0.24
Total excreted lead (µg/kg) 2.56 0.71
Excreted/absorbed lead 0.42 0.92
Fraction of intake retained 0.33 0.01
a Adapted from: US EPA (1986a)
b From: Ziegler et al. (1978)
c From: Rabinowitz et al. (1977a)
d Corrected for calculated endogenous faecal lead
Under conditions of relatively constant exposure to low
concentrations of lead, approximately 140 to 215 µg/day, a steady
state condition evolves in which excretion approximates intake
(Rabinowitz et al., 1976). Under these conditions urinary lead
excretion is approximately 70% of absorbed lead. Chamberlain (1985)
reported that approximately 60% of absorbed lead is retained by the
body and 40% excreted.
Chamberlain (1983, 1985) also examined the relationship between
the level of exposure and rate of lead excretion. Renal clearance at
PbB levels of between 1.2 and 3.84 µmol/litre (25 and 80 µg/dl) was
found to increase at a rate approximating the increase in plasma lead.
Chamberlain (1985) estimated endogenous faecal lead loss into the
gastrointestinal tract following administration of lead-203 via the
inhalation and parenteral routes. These estimates suggested a
clearance of approximately 0.5% of administered dose per day when PbB
concentrations were under 1.2 µmol/litre (25 µg/dl). A special form of
excretion of endogenous lead is through breast milk. Studies of breast
milk indicate that lead concentrations correlate with maternal PbB
concentrations, most studies reporting that lead secreted from breast
milk varies in concentration between 10 and 30% of the maternal PbB
concentration (Ong et al., 1985).
6.4 Biological indices of lead exposure and body burden
6.4.1 Blood lead
The relationship between levels of exposure from various
environmental media and PbB has been discussed briefly in section
6.1.4.
Due to the ease of sampling and homogeneity of the sample, blood
has been the most widely used specimen to assess the human body burden
of lead. However, in view of the relatively short half-life for lead
in blood (28-36 days) (see section 6.2.2), PbB measurements in general
reflect only recent exposures. Also, in view of the kinetics of
distribution within the body (cycling between blood, bone and soft
tissues), differentiation of low-level chronic exposure from a short
high-level exposure is not possible on the basis of a single PbB
measurement. Interpretation of PbB levels over a wide range of values
must take account of the curvilinear relationship between total intake
of lead and PbB concentrations, as well as the proportion of lead in
plasma (Manton & Cook, 1984; see also section 8.1).
A number of cohort studies have collected serial PbB measurements
for children from birth up to 7 or 10 years. For children who have not
had major changes in their environment, there is good correlation
between consecutive PbB measurements (McMichael et al., 1988;
Bellinger et al., 1992; Baghurst et al., 1992; Dietrich et al.,
1993a,b). In these extended studies it has become apparent that for
most of the children a single PbB analysis at 6 years of age gives a
reasonable assessment of the life-time lead exposure status of the
child. However, random PbB levels in samples taken before 6 years of
age can markedly underestimate the peak exposure usually seen at 2
years of age (SAHC, 1993).
A new exposure index, namely "lifetime average blood lead" or
"lifetime average integrated blood lead" has been introduced in
studies using serial blood data. It has been clearly explained in the
proceedings of a recent Workshop and this explanation is quoted
directly here. This measure, which is a reflection of the area under
the "blood lead by age curve", has been occasionally misunderstood.
Due to the shape of the longitudinal blood lead profile, the peak
blood lead level, usually observed during the second year of life is
considerably greater than the average lifetime blood lead. For
example, a five-year-old child with a lifetime average blood lead of
0.96 µmol/litre (20 µg/dl) is likely to experience blood lead levels
above 1.92 µmol/litre (40 µg/dl) during the second year of life and
may spend 3 years of life, from 12 months to 48 months, above
0.96 µmol/litre (20 µg/dl). Due to the shape of the curve, with
declining PbB beginning about 24-39 months of age, the average
lifetime blood lead for a given child decreases with increasing age.
This does not mean that the index is inappropriate but rather that
developmental blood lead profiles change over time. For example, a
lifetime average blood lead of 0.72 µmol/litre (15 µg/dl) should not
be interpreted as being equivalent to a single blood lead
determination of 0.72 µmol/litre (15 µg/dl) obtained at a single point
in life (Dietrich et al., 1991).
6.4.2 Tooth lead
Unlike blood samples, teeth are composed of several anatomically
distinct pools which form over several years. Thus, in contrast to
blood, teeth are useful tissues for assessing long-term lead
accumulation from prenatal exposures to the time of shedding of the
tooth.
The accumulation of lead in teeth (PbT) has been used as a
measure of exposure of children to lead in several epidemiological
studies (Needleman et al., 1972; Winneke et al., 1982a; Rabinowitz et
al., 1989; Fergusson et al., 1989; Hansen et al., 1989). Studies
measuring PbT and PbB, however, are few. Preliminary data from
Australia (Baghurst et al., 1992) indicate a correlation of 0.8
between whole PbT and PbB prior to tooth exfoliation, but the only
report relating lead in circumpulpal dentine and longitudinal PbB (by
Greene et al., 1992) found a correlation of only 0.5. PbT
concentrations can vary as a function of location of the tooth within
the mouth, age, and whether total PbT or dentine PbT were reported.
Therefore, the age of the tooth in the mouth and its location, the
sample (whole tooth or dentine) analysed must be considered in any
estimation of a corresponding PbB in the published studies (SAHC,
1993).
Because of the complex structure and development of teeth,
concentrations of PbT will depend on the method of sampling and
analysis, tooth type, and resorption and tooth age at exfoliation. As
a result of varying procedures used by different investigators, there
may be substantial variation in absolute values and possibly the
biological meaning of PbT levels between different studies.
Different parts of a tooth sequester lead during different stages
of development. This is especially of concern in tooth sampling
because lead is not uniformly distributed within a tooth on a
submillimeter scale. A pilot study of teeth from children with
differing histories of exposure to lead and using high precision lead
isotopic methods has shown that analyses of slices of the incisal part
of deciduous teeth give the clearest indications of the in utero
environment, and the cervical sections, the exposure from birth to
exfoliation (Gulson & Wilson, 1994).
For more detailed discussions on the measurement of PbT the
reader is referred to Grandjean et al. (1984), Purchase & Fergusson
(1986) and Fergusson et al. (1989).
6.4.3 Bone lead
The human skeleton begins to accumulate lead during fetal
development and continues to abut 60 years of age (Pounds et al.,
1991). Interest in bone lead and its measurement in vivo stems from
concern that skeletal lead is not metabolically inert (see section
6.2.2), but can be mobilized by physiological and pathological states,
for example, during pregnancy and lactation (Silbergeld, 1991) and
osteoporosis (Silbergeld et al., 1988), with possible adverse effects
in other tissues, including the fetus, as well as the desire to
develop a meaningful measure of cumulative lead exposure as a tool in
public health protection. Section 8.13 includes a brief discussion of
the skeleton as a target organ for lead toxicity.
Procedures are available to analyse bone samples for lead levels
in humans not occupationally exposed to lead (Drasch et al., 1987;
Drasch & Ott, 1988). These studies have shown a decrease in levels of
lead in bones from autopsy specimens in Germany after removal of lead
from petrol. However, full utilization of bone lead stores as
dosimeters of lead exposure in a prospective sense requires the
utilization of technologies for in vivo measurement of lead in bone
such as X-ray fluorescence analysis (Chettle et al., 1991; Rosen et
al., 1991; Todd et al., 1992).
6.4.4 Lead in urine
Although urinary lead level has been used to measure current
exposure (Robinson, 1974), its use as a biomarker of lead exposure is
questionable in view of the relatively low and variable level of lead
excreted in the urine (Jensen, 1984; Ibels & Pollock, 1986). For this
reason, and in view of technical difficulties in analysing low levels
of lead in urine, urinary lead appears to be of limited use for
general screening. However, where an elevated body burden of lead has
been estimated using PbB or other indices of lead exposure, urinary
levels of lead after administration of the chelating agent
CaNa2-EDTA is considered an excellent measure of the potentially
toxic fraction of the total body burden of lead (CDC, 1985). The
chelatable lead excreted is assumed to represent lead removal from
soft tissues and blood, as well as sub-compartments of bone (Ibels &
Pollock, 1986; Mushak, 1989). In contrast, the urinary lead excretion
associated with lead mobilization provides what is considered the best
measure of the potentially toxic fraction of the total body burden
(see CDC, 1985; US EPA, 1986a). On the basis of various in vitro
experimental and epidemiological studies (CDC, 1985; US EPA, 1986a;
Mushak, 1989), chelatable lead is assumed to be a chemical sample of
both mobile body compartments (i.e. blood and soft tissues) as well as
of sub-compartments of bone.
6.4.5 Lead in hair
Hair lead has been used as an indicator of exposure in children
(Marlowe & Errera, 1982; Wilhelm et al., 1989). However, there are
severe limitations on its use from both the methodological as well as
the metabolic perspective. Systemic variations in lead level have been
reported according to hair colour, texture, location on the body and
growth phase (Wilhelm et al., 1989). Also, it is almost impossible to
avoid external contamination, and to date no validated methods are
available for cleaning. Methods which are sufficiently vigorous to
remove superficial lead also remove lead from the hair shaft.
7. EFFECTS ON LABORATORY ANIMALS AND IN VITRO TEST SYSTEMS
Lead can affect various organ systems depending upon the level
and duration of exposure. In all species studied, adverse effects on
the nervous system have been noted at PbB concentrations lower than
for other target organs. There is particular concern for the effects
of lead on fetal development. Since the appearance of Environmental
Health Criteria 3: Lead (IPCS, 1977), there have been very many
reports on the effects of lead on in vitro and animal models. No
attempt will be made in this chapter to summarize all such studies.
Rather, emphasis has been given to those studies that relate most
directly to the understanding of the effects of lead on humans and
thus provide additional scientific support for the use of such human
studies in the assessment of risk from lead exposures.
7.1 Biochemical effects
7.1.1 Haem synthesis and haematopoiesis
The principal clinical manifestation of the effect of lead on the
haematopoietic system is anaemia but this occurs only with high levels
of exposure that are rarely seen today. Lead affects the
haematopoietic system at several levels. These include effects on haem
and globin synthesis and on erythrocyte formation and function.
Most haem synthesis is directed toward formation of haemoglobin
and the rest is used in cellular oxidative metabolism. Lead inhibits
several steps in the biosynthesis of haem. More detail can be found in
IPCS (1977), US EPA (1986a) and section 8.1.1 of this monograph.
Lead acts on steps in the synthetic pathway both inside and
outside the mitochondrion. It inhibits certain enzymes (ALA
dehydratase, ferrochelatase, coproporphyrinogen oxidase) and increases
the activity of ALA synthase (ALAS) activity as a consequence of
feed-back regulation by haem (Moore et al., 1980). The activity of
ALAS is the rate-limiting step in the haem biosynthetic pathway.
Lead affects erythrocyte formation by impairment of globin and
haem synthesis. Globin synthesis is inhibited by lead in rat bone
marrow cells at concentrations as low as 1 µmol/litre (Dresner et al.,
1982). This is thought to be secondary to decreased protein synthesis
in erythroid cells as a consequence of lead-induced inhibition of haem
synthesis. Lead also decreases erythrocyte survival through its
inhibition of membrane bound Na-K ATPase (Rhagavan et al., 1981).
It has been shown in in vitro cultures of cells from liver,
bone marrow and the nervous system that ALAS activity may be increased
by addition of only 50 µmol lead/litre and ALAD activity is decreased
by 60% at a concentration of 0.5 µmol/litre (Kusell et al., 1978;
Dresner et al., 1982). Lead (5 µmol/litre) also inhibits (20%)
porphobilinogen deaminase in red blood cell haemolysates at
5 µmol/litre (Piper & Tephly, 1974). Fowler et al. (1980) have shown
that disruption of haem synthesis results in reduction of tissue haem
levels. Increased exposure to lead decreases the content and function
of haem-dependent enzymes of the P-450 mono-oxygenase system (Meredith
& Moore, 1979). More recently it has been shown that lead induces haem
oxygenase activity thereby increasing the degradation of haemoproteins
which may adversely affect a number of cell functions such as
respiration and energy production (Maines, 1992). It has been
suggested that delay in the accumulation of haemoproteins of the
respiratory chain in brain tissue during development may result in
decreased synthesis of haem enzymes in the brain (Bull, 1980; Moore et
al., 1987). Holtzman et al. (1981), on the other hand, found no effect
of lead on brain cytochromes in rat pups with impairment of growth due
to exposure to lead.
7.2 Nervous system effects
7.2.1 Higher order behavioural toxicity
Experimental studies using animal models have demonstrated that
lead impairs learning and memory functions at virtually all stages of
the life cycle. The most significant studies have focused on
behavioural and learning impairments in experimental animals with PbB
levels below 1.44 µmol/litre (30 µg/dl). Bushnell & Levin (1983) fed
post-weaning rats drinking-water containing 10 or 100 mg lead (as lead
acetate) per litre for up to 7 weeks. PbB was not determined but has
been estimated from comparisons with similar lead exposure models to
have been less than 0.96 µmol/litre (20 µg/dl) (Davis et al., 1990b).
Brain lead averaged about 0.05 µg/g wet weight. The rats exhibited
impaired learning ability when tested for their ability to choose
between alternate arms of a radial maze. Cory-Slechta et al. (1985)
demonstrated a significant learning impairment in rats given lead
acetate in drinking-water (25 mg/litre) from weaning. The observed
outcome was a significantly higher response rate in lead-exposed rats
working under a fixed interval schedule of food reinforcement. PbB
values of lead-treated rats in that study averaged
0.72-0.96 µmol/litre (15-20 µg/dl) and brain lead levels averaged
0.07 µg/g wet weight. Similar effects have been described in older
rats (16 months of age) at steady-state PbB levels of
0.62-0.86 µmol/litre (13-18 µg/dl) (Cory-Slechta & Pokora, 1991). More
recently, Cohn et al. (1993) demonstrated selective effects of lead on
learning processes, as distinct from non-specific effects such as
motivational levels, and sensory or motor impairment using a multiple
schedule of repeated learning and performance. Exposure to lead
acetate in drinking-water (50 mg/litre) produced a PbB of
1.2 µmol/litre (25 µg/dl).
Winneke et al. (1977) exposed rats to lead while in utero,
through mother's milk and directly in drinking-water. Their learning
ability was then tested by requiring the animals to discriminate
between stimuli of either different orientation (stripes as an easy
task) or different size (discs as a difficult task). Adult rats tested
between 90 and 170 days of age with PbB levels of less than
1.44 µmol/litre (< 30 µg/dl) were slower to learn the difficult task
of size discrimination (but not the easy discrimination problem) and
tended to repeat more errors than control subjects.
Altmann et al. (1993) demonstrated deficits of both active
avoidance learning (AAL) and in vitro hippocampal long-term
potentiation (LTP) in adult rats that had received pre-weaning or pre-
and post-weaning dietary lead exposure to achieve PbB levels of
approximately 0.72 µmol/litre (15 µg/dl) and brain lead concentrations
of 0.09-0.16 µg/g wet weight. Deficit in animals with only
post-weaning exposure and blood lead levels was about 0.77 µmol/litre
(16 µg/dl) but brain lead concentrations of only 0.09 µg/g was either
absent (in the case of LTP) or markedly reduced (in the case of AAL).
Similar types of effects have been noted in studies using
non-human primates. For example, Rice (1985) reported deficits of
discrimination reversal performance in monkeys dosed orally with 0, 50
or 100 µg lead/kg body weight per day for the first 200 days of life.
At the cessation of dosing, PbB concentrations were 0.144 µmol/litre
(3 µg/dl), 0.72 µmol/litre (15 µg/dl) and 1.2 µmol/litre (25 µg/dl),
respectively. Prior to behavioural testing (3 years of age), PbB
levels were 0.144 µmol/litre (3 µg/dl), 0.528 µmol/litre (11 µg/dl)
and 0.624 µmol/litre (13 µg/dl). Additional evidence for lead-induced
changes in learning in non-human primates is provided by the study of
Lilienthal et al. (1990), which showed a dose-related increase in the
percentage of errors in a learning set formation task (discrimination)
with PbB levels in the low exposure group (350 µg lead acetate/g diet)
averaging 1.68 µmol/litre (35 µg/dl). Also, an impairment of reversal
learning was found by Bushnell & Bowman (1979b) to persist in monkeys
up to their fifth year, at which time the PbB level in treated animals
averaged 0.24 µmol/litre (5 µg/dl) compared to 0.192 µmol/litre
(4 µg/dl) in controls. In the study by Bushnell & Bowman (1979a), the
period of lead exposure was limited to the first year of life and
resulted in PbB levels averaging as high as 3.12 µmol/litre
(65 µg/dl). No description was given of quality control procedures to
ensure accuracy of PbB determinations at the lower levels reported.
Several studies on experimental animals have shown perseverative
effects (the tendency to respond repetitively and inappropriately even
though environmental conditions have changed) which may underlie many
of the lead-induced changes in learning and other higher order
behavioural processes noted in such studies. This has been clearly
demonstrated in rodents by Cohn et al. (1993) in the context of
learning impairments. In the study by Bushnell & Levin (1983)
described above, the accuracy impairment derived from a tendency of
lead-exposed rats to re-enter a previously explored arm of the maze.
Similar perseverative tendencies have been described in non-human
primate studies, e.g., on delayed matching to sample (Rice, 1984a) and
delayed alteration tasks (Levin & Bowman, 1986; Rice & Karpinski,
1988).
Experimental animal studies also reveal the importance of task
complexity in detecting lead-induced changes in behaviour, both in
non-human primates (Levin & Bowman, 1983, 1986; Gilbert & Rice, 1987)
and rodents (Winneke et al., 1977, 1982b).
Persistence of lead-induced changes in some higher order
behavioural processes is also suggested by a number of experimental
animal studies. Several reports from the longitudinal studies of
non-human primates by Bowman and his colleagues (e.g., Bushnell &
Bowman, 1979a,b; Levin & Bowman, 1983, 1986, 1989) have demonstrated
persistent neurobehavioural deficits extending up to 8 years after the
termination of exposure and long after PbB levels had declined to the
control level. In rats, Cory-Slechta & Thompson (1979) showed that the
levels of lead exposure determine the persistence of lead effects,
while Cory-Slechta (1990a,b) revealed that apparently transient
effects of lead on behaviour could re-emerge with changes in the
reward contingencies of the environment. Munoz et al. (1986) found
deficits in both spatial and visual discrimination performance of rats
at several months of age resulting from pre-weaning exposure via the
dams. The same outcome was noted by Altmann et al. (1993) in a study
where deficits in adult rats were noted following pre-weaning
exposure.
7.2.2 Mechanisms of lead-induced behavioural toxicity
While the mechanisms underlying lead-induced behavioural toxicity
have yet to be adequately determined, the experimental animal
literature provides suggestive leads. The early experimental studies
of Pentschew (1965) and Pentschew & Garro (1966) demonstrated in the
rodent model that the pathogenesis of acute high-dose encephalopathy
was secondary to increased permeability of capillaries in the brain,
leading to leakage of fluid and red blood cells. These changes are
similar to those occurring in children with acute lead encephalopathy
characterized clinically by coma, convulsions and death, and identify
the brain microvasculature as the primary target for lead in the
central nervous system following high level exposure to lead.
Changes in microvascular morphology are not evident with low
level lead exposure, but it is the vulnerability of the blood-brain
barrier that permits exposure of the brain to lead. Exposure of the
developing fetus to lead results in higher uptake of lead in the brain
than from later exposures (Rossouw et al., 1987). The development of
resistance to acute encephalopathy in rat pups during the first few
days of life is thought by Holtzman et al. (1984) to be related to
maturation of the blood-brain barrier and possibly to the ability of
the older animals to sequester lead in protein complexes. Thus
neuropathological changes may relate more to the issue of exposure
than to mechanism of effect.
More recently, the focus of mechanistic studies has involved
biochemical and neurochemical changes. Biochemical changes in synaptic
transmission are likely to be related to problems in dendrite-nerve
organization and function (Goldstein, 1990). There is continuing
reorganization of dendrite and nerve terminal connections throughout
early months of neural development. A number of biochemical and
functional studies concerning possible mechanisms of lead effect
suggest lead may disrupt this process and its function through early
childhood (e.g., Cookman et al., 1987, 1988).
Other biochemical effects from lead exposure which may be the
basis of important neurobiological mechanisms of leadinduced
behavioural toxicity include changes in protein kinases, with
implications for neurotransmitter system disturbances. At least three
protein kinases present in nerve terminals involved in modulating the
release of neurotransmitters have been shown to be affected by lead.
However, the relevance of these findings to cognitive function has not
been established (Goldstein, 1990).
From both in vivo and in vitro studies, lead exposure has
been reported to have effects on virtually all neurotransmitter
systems (US EPA, 1986a). Depending on the stage of development, these
include dopaminergic, cholinergic, serotonergic, GABAnergic,
glutamatergic and opiate systems. Probably the most extensively
investigated system has been the dopaminergic system where a number of
changes at the biochemical and receptor level have been described. One
of the difficulties, however, is determining the relationship of the
reported changes at the biochemical/receptor level to changes in
behavioural function. McIntosh et al. (1989) noted lead-induced
changes in tetrahydrobiopterin metabolism which may be related to
changes in IQ scores (Blair et al., 1982; McIntosh et al., 1985). In
further support of a functional role for lead-induced changes in
dopaminergic systems, Cory-Slechta & Widzowski (1991) reported
alterations in both D1 and D2 dopaminergic sensitivity in rats with
PbB levels of 1.2-1.44 µmol/litre (25-30 µg/dl).
More recently, studies by Altmann et al. (1993) and Cohn &
Cory-Slechta (1993) suggest a role for changes in the N-methyl-D-
aspartate (NMDA) receptor complex in lead-induced behavioural
toxicity. The NMDA receptor complex has been implicated in the
learning and memory processes.
Effects of lead on mitochondrial energy metabolism may also be
important in the pathogenesis of neurological effects. Impairment of
respiration in brain mitochondria has been observed in in vitro
preparations (Holtzman et al., 1978) and in mitochondria isolated from
brains of lead-exposed rats (Gmerek et al., 1981).
7.2.2.1 Conclusions
Given their ability to establish causal relationships between
lead exposure and biological effects, experimental animal studies can
provide evidence supportive of human epidemiological findings. Many
experimental animal studies have been carried out to characterize the
nature of lead's effects on various target organ systems as well as to
establish the underlying mechanisms of effect. Furthermore, these
studies, by their very nature, are not confounded by such co-variates
of children's IQ as parental IQ, socioeconomic status and quality of
the home environment, or complicated by nutritional inadequacies of
the study population. Earlier experimental animal studies tended to
utilize relatively high lead exposure levels, but over the past ten
years, exposure levels and protocols have been more relevant to the
human situation.
There are, of course, some limitations of experimental animal
studies that must be considered. For example, there may be differences
in species sensitivity as well as differences in pharmacokinetic and
pharmacodynamic behaviour of lead in rodents, primates and humans.
Higher external exposure levels of lead are required to produce PbB
levels corresponding to levels experienced in human populations, and
higher PbBs are necessary to induce encephalopathy in rodents than are
associated with such effects in humans. Such differences in
sensitivity obviously reflect, at least in part, differences in
kinetics of lead across species. Despite these differences, it should
be noted that corresponding levels of PbB have been associated with
neuro-behavioural toxicity in both animal models (rodents as well as
non-human primates) and human populations. This comparability of
effect levels, despite the relative insensitivity of the animal
models, suggests that the levels of concern in humans might be even
lower, although quantitative extrapolation is difficult. Although one
cannot presume that the neurotoxic effects of lead in experimental
animals necessarily predict specific effects in humans, the
similarities in neurobehavioural end-points in humans and animals are
sufficient to conclude that neurobehavioural deficits in animals are
at least qualitatively predictive of effects in humans.
7.2.3 Sensory organ toxicity
Visual and auditory functions have been shown to be affected by
lead; other sensory modalities have largely been neglected. Bushnell
et al. (1977b) studied visual acuity in infant rhesus monkeys using
different degrees of illumination. Four control animals and three
animals each in a low and a high lead group were tested. The animals
were orally dosed between postnatal days 5 and 365. Although the
dosing regimen of this group was intended to produce blood lead levels
around 4.08 µmol/litre (85 µg/dl), peak levels of between 6.58 and
14.4 µmol/litre (137 and 300 µg/dl) occurred between the fifth and the
ninth weeks of dosing. Significant impairment of scotopic vision was
observed only in the animals of the high lead group.
Using flash-evoked visual potentials (VEP), Lilienthal et al.
(1986) studied visual function in 7- to 7´-year-old rhesus monkeys
pre- and postnatally exposed to lead acetate (0, 350 and 600 mg/kg) in
their diets. Seventeen animals were tested altogether: six controls,
and five or six in each of the exposed groups. PbB levels fluctuated
around 1.92 µmol/litre (40 µg/dl) in the group fed 350 mg/kg diet
during the seven postnatal years, whereas in the group fed 600 mg/kg
they had declined from the initial average of 5.28 µmol/litre
(110 µg/dl) at age 9 months to about 2.88 µmol/litre (60 µg/dl) at 7
years of age. PbB levels in the mothers of both these groups had been
between 1.15 and 1.78 µmol/litre (24 to 37 µg/dl) during pregnancy. A
dose-related decrease of amplitudes and a similar increase of
latencies for the main VEP component were found; these were, in most
instances, significant in both the high and low lead groups.
These findings are supported by the results of studies on rodents
by Fox et al. (1977). Prolonged VEP latencies were observed following
chronic developmental lead exposure. Lower VEP amplitudes have also
been observed in rats at PbB levels exceeding 1.44 µmol/litre
(30 µg/dl) (Winneke, 1979). in vitro studies suggest that rods are
more sensitive to the effects of lead than are cones (Fox & Sillman,
1979).
Information on the impairment of auditory function by lead comes
from electrophysiological studies using brainstem auditory evoked
potentials (BAEP) in rhesus monkeys (Lilienthal et al., 1990). PbB
levels in the various groups at the time of testing averaged
0.432 µmol/litre (9 µg/dl; controls), 1.92 µmol/litre (40 µg/dl; low
lead), and 2.688 µmol/litre (56 µg/dl; high lead). Of the four peaks
discernible in the monkey, BAEP peaks II and IV exhibited
significantly prolonged latencies, which were observed only in the
animals of the high lead group.
7.3 Renal system
The renal effects of lead in animal models occur as a result of
both acute and chronic exposures. Acute lead nephrotoxicity is
characterized by decreased reabsorption of small molecular weight
compounds by the renal tubule, particularly amino acids, glucose and
phosphate (Fanconi Syndrome).
Morphological changes of chronic lead exposure include
cytomegaly, development of nuclear inclusion bodies and
ultrastructural changes in mitochondria. The basis for the cytomegaly
is not understood but it is known that there is altered homoeostasis
of water and electrolytes and cellular swelling. Inclusion bodies are
lead-protein complexes composed of acidic non-histone proteins. As
much as 90% of lead in the kidney has been shown to be contained in
the inclusion bodies, suggesting that they provide a detoxification
function. The origin of the protein is not known but Egle & Shelton
(1986) have identified the most abundant component of isolated
inclusion bodies to be a constitutive protein of the adult central
nervous system primarily in the cerebral cortex. The lead in the
inclusion bodies is chelatable with EDTA (Goyer & Wilson, 1975).
Mitochondria isolated from kidneys of lead-intoxicated rats have
impaired respiration and oxidative phosphorylation capacity, (Goyer &
Rhyne, 1973).
Kholil-Manesh et al. (1992) studied the evaluations of renal
histological and functional changes in rats continuously dosed with
high dose lead. Glomerular filtration rate (GFR) was significantly
increased at 3 months, but significantly decreased at 12 months. Lead
inclusion bodies were noted throughout the duration of the study.
Tubular atrophy and intestinal fibrosis appeared at 6 months. The
brush border of proximal tubular cells was disrupted at 1 and 3
months, but recovered later.
With continued exposure to lead, acute nephropathy may progress
to chronic interstitial nephritis that does not have any unique or
distinguishing features. There is progressive increase in interstitial
fibrosis, dilatation of tubules and formation of microcysts with
hyperplasia of tubular epithelial cells. In the rat, inclusion bodies
are reduced in number and may be entirely absent in later stages of
the nephropathy. Glomerular sclerosis occurs with onset of proteinuria
and renal failure (Goyer & Rhyne, 1973).
The sequence of morphological changes observed in experimental
models is thought to be generally true for humans. Experimental
studies in rats suggest that there may be a threshold for lead renal
toxicity in the rat at PbB levels similar to those observed for
humans. In rats exposed to different doses of lead for up to 12 weeks,
a PbB level of 2.88 µmol/litre (60 µg/dl) appears to be the threshold
for proximal renal tubular cell injury by lead (Goyer et al., 1989).
This PbB level is equivalent to kidney lead levels of about 45 mg/kg
wet weight and is the level of lead at which excretion of renal
calcium increases and ultrastructural changes occur in proximal
tubular cell mitochondria. This finding is consistent with the
observation in humans by Buchet et al. (1980) and Gennart et al.
(1992) of a threshold value of 2.88 µmol/litre (60 µg/dl) of PbB which
leads to adverse renal effects by lead.
7.4 Cardiovascular system
Early studies concerning the production of hypertension in
experimental animals, summarized in Environmental Health Criteria 3:
Lead (IPCS, 1977), were conflicting. Among rats given 70 mg of lead
acetate per day orally, only a few survived 40 days and all were
hypertensive (Griffith & Landaver, 1944). On the other hand, other
studies did not reveal a blood pressure effect from high-level lead
exposure in rats (Padilla et al., 1969) or in dogs (Fouts & Page,
1942). More recently, several experimental studies have confirmed that
lead can produce hypertension, and evidence for several plausible
mechanisms has been provided. Rats, both normotensive and
spontaneously hypertensive, exposed to lead in water supplies for up
to a year, resulting in PbB levels of up to 0.6 µmol/litre
(12.5 µg/dl), exhibited ventricular tachycardia and ventricular
fibrillation (Evis et al., 1985). Subsequent studies (Evis et al.,
1987) showed that adrenaline had a significant arrhythmogenic effect
in hypertensive animals. It was concluded that chronic exposure to
lead, when combined with high blood pressure, slightly enhances the
susceptibility of the heart to arrhythmias induced by myocardial
ischaemia. Overviews of the experimental evidence related to
hypertension and lead exposure have been published by Victery (1988)
and US EPA (1986a, 1989). Taken as a whole, these studies demonstrate
that increase in blood pressure does occur secondary to renal failure
in rats with continuous high-level exposure to lead. More recent
studies on effects of chronic low-level exposure to lead on blood
pressure in rats have shown alterations in cardiovascular parameters
in the PbB range of 0.24-1.92 µmol/litre (5-40 µg/dl), and provide
some insights as to mechanisms involved in the pathogenesis of
lead-related cardiovascular effects.
Victery et al. (1982a) exposed rats to lead in utero by giving
dams drinking-water containing 0, 5, and 25 µg lead/ml and continued
this regimen to the pups for 5 to 6 months. Although no change in
blood pressure was noted at these levels of exposure, significant
changes in the renin-angiotensin system were reported in animals given
water containing 25 µg lead/ml with a PbB level of 0.864 µmol/litre
(18 µg/dl). In a similar study (Victery et al., 1982b), exposure to
100 µg lead/ml in drinking-water (but not 500 µg/ml) produced a
significant (17 mmHg) elevation in blood pressure beginning at 3´
months of age and continuing until 6 months of age. Chai & Webb (1988)
found an elevation of 15 to 20 mmHg in the systolic blood pressure of
rats given drinking-water containing 100 µg lead/ml. These authors
suggest that alterations in the cellular mechanisms that regulate
intracellular calcium concentration may enhance pressor responsiveness
to catacholamines. Boscolo & Carmignani (1988) found raised blood
pressure in rats with exposure to drinking-water containing 0, 30 or
60 µg lead/ml for 18 months. Cardiovascular responses to blood
pressure agonists indicated that lead exposure affects the
renin-angiotensin system and induces sympathetic hyperactivity by
acting on central and peripheral sympathetic junctions, increasing the
responsiveness to stimulation of alpha-2-adrenergic receptors, and by
increasing sensitivity to stimulation of cardiac and vascular
beta-adrenergic and dopaminergic receptors.
From the experimental evidence summarized by Victery (1988), it
appears that low-level exposure to lead produces an elevation in blood
pressure. The failure to demonstrate increased blood pressure in some
studies with high-level exposure to lead suggests that the effect of
lead on blood pressure may be biphasic, i.e. a consistent effect with
low-level exposure but inconsistent effects with high-level exposure.
7.5 Reproductive system
Experimental studies on the effects of lead on the reproductive
system most often concern toxicity to either the male or female but
have addressed results of exposure to both parents. Environmental
Health Criteria 3: Lead (IPCS, 1977) identified effects on
spermatogenesis in rats exposed to lead and showed that high maternal
exposure to lead in rats can reduce numbers and size of offspring.
There may also be paternally transmitted effects resulting in
reductions of litter size, weights of offspring and in survival rate.
Few studies into the effects of lead on male sexual function have
reported PbB levels. Ivanova-Chemishanska et al. (1980) reported
changes in levels of enzymatic activity and ATP in testicular
homogenates from rats given 0.0001 or 0.01% solutions of lead acetate
as drinking-water over a 4-month period. Chowdhury et al. (1984) found
testicular atrophy along with cellular degeneration in rats with PbB
levels over 3.36 µmol/litre (70 µg/dl), but not in rats with levels of
2.59 µmol/litre (54 µg/dl). Donovan et al. (1980) found that lead
inhibited androgen binding by cytosolic receptors in mouse prostate.
Testicular homogenates from 2- to 3-week-old male offspring (PbB
levels 0.30 µmol/litre, 6.3 µg/dl) of lead-exposed female rats showed
a decreased ability to metabolize progesterone (Wiebe et al., 1982).
In an in vitro study, Wiebe et al. (1983) found a 10 to 20% decrease
in FSH binding and in the production of cyclic AMP by Sertoli cells
isolated from prepubertal rats and cultured in the presence of lead
acetate (2.64 × 10-4 mol/litre). In addition, the activity of
cellular 3ß-hydroxysteroid dehydrogenase was decreased. It was shown
by Sokol et al. (1985) that there is a dose-related suppression of
serum testosterone levels and spermatogens in adult rats (100 day of
age) given a solution of 0.3% sodium acetate as drinking-water for up
to 60 days. PbB concentrations were between 1.44 and 2.40 µmol/litre
(30 and 50 µg/dl), depending on the length of treatment. Further
studies (Sokol, 1987) supported the hypothesis that lead disrupts the
hypothalamic control of pituitary hormone secretion. However, other
evidence indicates that lead may directly or indirectly affect
testicular enzymes or may act indirectly by a reduction in testicular
binding of FSH and production of cyclic AMP (US EPA, 1986a).
Dosing mature female rats with lead in order to produce PbB
concentrations of 1.44 µmol/litre (30 µg/dl) resulted in irregular
estrous cycles. At a PbB level of 2.54 µmol/litre (53 µg/dl), animals
developed follicular cysts and there was a reduction in the number of
corpora lutea (Hilderbrand et al., 1973). Grant et al. (1980) reported
delayed vaginal opening in rats whose mothers were given drinking-
water containing 25, 50 and 250 µg lead/ml. The vaginal opening delays
in the group given 25 µg lead/ml of drinking-water occurred in the
absence of any growth retardation or other developmental delays and
were associated with median PbB levels of 0.86-1.39 µmol/litre
(18-29 µg/dl). Studies on female monkeys have shown that pre- and/or
postnatal exposure to lead can affect pubertal progression and
hypothalamicpituitary-ovarian-uterine functions. Chronic exposure of
nulliparous female monkeys to lead (PbB levels of approximately
1.68 µmol/litre, 35 µg/dl) resulted in subclinical suppression of
circulating luteinizing and follicle stimulating hormone and estradiol
without producing overt effects on general health or menstruation
(Foster, 1992).
7.6 Effects on bone
There is growing interest in lead in bone for several reasons.
Bone is a store for lead accumulated from past exposure. It has a long
biological half-life but may be mobilized and contribute to blood lead
during pregnancy. With the development of X-ray fluorescence
techniques to measure lead in bone in vivo, there is a need to
improve our understanding of bone lead metabolism and of factors that
influence lead retention and release. In addition, lead may adversely
affect bone metabolism, particularly in post-menopausal women, and
contribute to the development of osteoporosis. There has only been
limited experimental study of these concerns to date, but there are
some relevant reports in the literature.
A summary of much of the currently available literature on the
potential toxicological implications of lead in bone during pregnancy
and lactation is contained in a review by Silbergeld (1991). Toward
the latter half of pregnancy in mice, there seems to be a preferential
transfer of lead across the placenta to the fetus (Danielson et al.,
1983). In rats exposed to lead for 150 days and then not exposed for
50 days prior to mating, Buchet et al. (1977) found that there was a
substantial mobilization of lead from mother to fetus. Keller &
Doherty (1980b) found using radiotracer lead (210Pb) in female mice
that there was also a major transfer of lead to the pup during
lactation. The concomitant decrease in maternal bone lead supports the
hypothesis that bone resorption of lead occurs during lactation.
It has been shown that lead may directly and indirectly affect
various aspects of bone metabolism (Pounds et al., 1991). Lead
inhibits the renal enzyme 1-hydroxylase, reducing plasma levels of
1,2-dihydroxychole-calciferol (activated vitamin D). Lead impairs the
Haversian remodelling system in beagle dogs chronically exposed to
lead (Anderson & Danylchuk, 1977).
7.7 Immunological effects
The effects of lead on the immune system are diverse but not well
documented. Lead reduces resistance and increases mortality of
experimental animals when they are infected by a broad range of
bacterial and viral agents (Koller, 1984). Lead impairs antibody
production in animals and generally decreases immunoglobin plaque-
forming cells (Koller & Roan, 1980).
7.8 Mutagenicity
Lead is thought to have genotoxic properties. However, lead-
induced gene mutations in cultures of mammalian cells have only been
observed at concentrations toxic to the cells. Studies for point
mutations in bacterial systems have also yielded negative results (US
EPA, 1986a). Zelikoff et al. (1988) found that both insoluble lead
sulfide and more soluble lead nitrate were mutagenic when added to
Chinese hamster V79 cells. A 6-fold increase in mutation frequency was
noted at a lead nitrate level of 500 µmol/litre medium. These authors
also found that lead acetate induced morphological transformation of
Syrian hamster cells. However, they concluded that these effects may
not have been the result of direct damage to DNA but may have occurred
via indirect mechanisms including disturbances in enzyme functions
important in DNA synthesis and/or repair.
Studies on the production of chromosome aberrations, sister-
chromatid exchanges and micronuclei by lead, whether in in vitro
cultures or in vivo, have given mixed results, and summaries are
available (US EPA, 1986a; IARC, 1987a,b; ATSDR, 1991).
7.9 Carcinogenicity
There have been several experimental studies in rats and mice in
which long-term administration of a lead compound in food or
drinking-water or parental administration has produced tumours of the
kidney (Van Esch & Kroes, 1969; Moore & Meredith, 1979). These and
other studies have been discussed in detail in IARC (1980) and
summarized in US EPA (1986a). In general, methodological problems,
including dose levels, number of animals, and doses, and lack of
toxicity monitoring, make many of the studies difficult to interpret
in a quantitative manner. One study (Azar et al., 1973) addressed some
of these problems and reported an increase in the numbers of renal
tumours in rats fed diets containing 500 mg lead/kg for 2 years. PbB
levels were 3.84 µmol/litre (80 µg/dl). In all studies renal
carcinogenicity occurred against a background of proximal tubular cell
hyperplasia, cytomegaly and cellular dysplasia in response to the high
doses of lead and long exposure times (Goyer, 1985). Renal
adenocarcinoma occurred in a high percentage of lead-exposed animals
and the incidence of tumours was related to the length and severity of
exposure (Mao & Molnar, 1967). Males appear to be more susceptible to
tumours than females. The maximum dose of lead in drinking-water, not
associated with any morphological or functional evidence of renal
toxicity in rats fed a diet containing adequate levels of trace
minerals, particularly calcium, is 200 mg lead/litre (Goyer et al.,
1970). No evidence of renal tumours at doses below this value has been
reported (Azar et al., 1973; US NCI, 1979).
Several hypotheses have been proposed for the mechanism of lead
carcinogenicity in experimental animals. These include mutagenicity,
cellular proliferation, nuclear protein (inclusion bodies), promoter
activity, activation of protein kinase C, and cystic hyperplasia
(Goyer, 1993). Lead is a weak mutagen in mammalian cell systems, but
is a strong mitogen. Exposure to a single intraperitoneal injection of
lead acetate in rats stimulates a 40-fold increase in cell
proliferation as measured by autoradiography, and this is further
increased by unilateral nephrectomy (Choie & Richter, 1972). DNA
synthesis in kidneys, as measured by 3H-thymidine incorporation, is
increased 15-fold and the mitotic index 45-fold following a single
intracardiac injection of lead acetate in mice (Choie & Richter,
1974). Cell proliferation and hyperplasia are seen in the liver of
rats given a single intravenous injection of lead nitrate (Columbano
et al., 1984). In both of these studies, increases in cell
proliferation occurred in the absence of cellular necrosis, suggesting
that this was a mitogenic rather than regenerative response.
Activation of protein kinase C and formation of nuclear inclusion
bodies or lead protein complexes are additional events that may
influence regulation of cell growth and development and play a role in
the carcinogenic response in experimental animals resulting from
administration of lead (Goyer, 1993). Cystic hyperplasia, a late
morphological manifestation of chronic lead nephropathy, is a risk
factor for renal cancer (Bernstein et al., 1987). Prior to adenoma
formation in animals treated with renal carcinogens, cystic
hyperplasia was reported (Dees et al., 1980; Goyer et al., 1981).
8. EFFECTS ON HUMANS
Despite the long recognition of lead poisoning, new clinical
cases continue to be the subject of published reports. Although
important, they affect a comparatively small proportion of the
population at risk from the potential effects of exposure to
environmental lead.
Over the last 10-15 years, particular attention has been directed
towards epidemiological studies designed to evaluate the possible
neurotoxic effects of lead on the developing child, especially delayed
or impaired neurobehavioural development and performance.
In the adult population, considerable attention has been directed
towards evaluating cardiovascular effects and the implication of lead
in hypertension.
New studies have contributed to our understanding of the
biochemical effects of lead, and may facilitate early recognition of
significant change and mitigation of potentially adverse outcomes.
The effects of lead have been studied widely in both the general
population and in those exposed occupationally. Since these effects
are the same in both settings, no distinction has generally been made
in the discussion. However, it is often important to distinguish
between adults and children because of different susceptibility.
8.1 Biochemical effects of lead
Lead is known to affect a number of enzymes and physiological
systems which result in a wide variety of changes in humans. While
those affecting the haematopoietic system are well known, there are
others which need to be considered in the risk assessment process and
are considered in this section.
In considering the effects of lead on biochemical systems, it is
appropriate to discuss the form of the lead in the various body
compartments.
Blood lead (PbB) is distributed between the plasma and the
erythrocyte. There is less than 1% in the plasma for PbB levels of up
to 4.8 µmol/litre (100 µg/dl) (Manton & Cook, 1984). The curvilinear
relationship of serum lead to blood lead is shown in Fig. 12. The data
show that the erythrocytes have a capacity to bind lead up to PbB
levels of about 2.4 µmol/litre (50 µg/dl). Above this level a fairly
rapid increase in the serum levels occurs.
It should be noted that the proportion of "free" (i.e. unbound)
lead in blood is important in relation to biological activity. Lead is
bound to haemoglobin in blood and has a greater affinity for fetal
than adult haemoglobin (Ong & Lee, 1980). It may, therefore, be
5.2.2.3 Alcoholic beverages
Contamination of alcoholic beverages with lead may occur in
several way. For example, lead solder used to repair casks or keg and
tap lines from lead capsules used as seals or from residues of lead
arsenate pesticides in soils now used to grow grapes. Alcoholic
beverages tend to be acidic and there is the possibility that large
amounts of lead can be dissolved during preparation, storage or
serving (Wai et al., 1979). Published reports on lead levels in wine
show that important variations occur from sample to sample (Jorhem et
al., 1988). The US Department of the Treasury (1991) analysed 432
table wines sold within the USA. The results are summarized in Table
17.
Sherlock et al. (1986) found that the majority of canned and
bottled beer (90 and 86% respectively) contained less than 10 µg
lead/litre. Draught beers typically contained higher lead
concentrations with 55% having lead concentrations greater than
10 µg/litre, 16% with concentrations over 20 µg/litre, and 4% with
concentrations over 100 µg/litre. The higher lead concentrations in
draught beers are considered most likely due to the draught-dispensing
equipment which sometimes contains brass or gunmetal, both of which
contain low but significant amounts of lead (Sherlock et al., 1986).
In general, alcoholic beverages do not appear to be a significant
source of lead intake for the average person.
Table 17. Distribution of lead in table wines in USAa
Range Number of Percentage of total
(µg/litre) samples samples analysedb
0-10 36 8.3
11-25 62 14.4
26-50 105 24.3
51-100 144 33.3
101-250 64 14.8
251-500 12 2.8
501-673 9 2.1
a From: US Department of the Treasury (1991)
b In all, 432 samples were analysed.
5.2.3 Dust and surface soils
5.2.3.1 Dust
Dust is a significant source of exposure to lead, particularly
for young children (see Fig. 11), as has been demonstrated in several
studies correlating children's blood lead concentrations with dust
lead levels (Rabinowitz et al., 1985; Bornschein et al., 1987; Davies
et al., 1987a; Laxen et al., 1987; Steenhout, 1987).
The major contributions to lead levels in soil and outdoor dust
are from the combustion of fossil fuels (principally leaded petrol),
stationary sources such as smelters, and peeling and flaking of
lead-based paint. Typical lead levels in road dust in the USA are
800-1300 mg/kg in rural areas to 100-5000 mg/kg in urban areas (US
EPA, 1989c).
Concentrations of lead in household dust vary greatly between
different dwellings and areas of the world. Mean concentrations of
300-2500 mg/kg have been found in the United Kingdom and USA, but
individual samples may be in the range of 10 000 to 30 000 mg/kg (Que
Hee et al., 1985b; Clark et al., 1985; Bornschein et al., 1986; Raab
et al., 1987).
Flaking lead-based paint, paint chips, and weathered powdered
paint markedly increase intake of lead from surface dust, particularly
for urban children with pica (US EPA, 1986a; Bornschein et al., 1986).
Lead-based paint chips have been found to contain 1000-5000 µg
lead/cm2 (Billick & Gray, 1978). When lead-based paint is present,
interior renovation activities greatly increase household dust lead
concentrations (Laxen et al., 1987). Improved control of dust and
surface clean-up after lead-based paint removal have been shown to
reduce lead exposure of children reoccupying affected houses (Charney
et al., 1983).
5.2.3.2 Soil
In rural and remote areas, lead in soil is derived mainly from
natural geological sources. These natural sources account for 1-30 mg
lead/kg, but where soils are derived from leadmineralized rocks,
natural concentrations may range from several hundred to several
thousand mg/kg.
Typical values for lead in rural soils in the United Kingdom are
15-106 mg/kg with a geometric mean of 42 mg/kg (Davies, 1983). A
geometric mean of 48 mg/kg for 2780 samples has also been reported
(McGrath, 1986).
Concentrations of lead in urban soil vary greatly. In the USA, a
study of city parks recorded concentrations of 200 to 3300 mg/kg (US
EPA, 1989). Concentrations of up to 10 960 mg/kg have been reported
for urban garden soils in the USA (Mielke et al., 1984), and up to 14
100 mg/kg in the United Kingdom (Culbard et al., 1988). Concentrations
can exceed 20 000 mg/kg around lead mining and processing operations
(Culbard et al., 1988). In areas where lead-based paint has been used,
soil samples taken near building foundations have been reported to be
as high as 20 000 mg/kg (Schmitt et al., 1988; Krueger & Duguay,
1989).
In general, lead concentrations in soils near roads are high
where road traffic density is high. Concentrations decrease
exponentially with distance from the road (IPCS, 1989).
Continuous application of sewage sludge results in an
accumulation of lead in soil. For example, soil receiving heavy
applications over a long period was found to contain 425 mg/kg,
compared with 47 mg/kg in an untreated soil (Beckett et al., 1979).
5.2.3.3 Migration of lead from food containers
The available data on the daily intake of lead by adults and
children indicate a general decrease in those areas where the level of
lead in petrol has decreased and a concerted effort made to avoid
lead-soldered cans for food storage (OECD, 1993). However, in many
regions of the world, lead can migrate from food storage and serving
vessels such as lead-soldered cans (see section 5.2.2.2), ceramic
dishes, pottery vessels, crystal glassware and decals on food wrap
and/or dishes. Acidic foods tend to leach more lead. However, certain
foods such as corn and beans are associated with greater release of
lead than would be predicted from their acidity alone (Bolger et al.,
1991). Oxygen appears to accelerate the release of lead from food
containers.
If foods are stored in ceramic or pottery dinnerware that was
lead-glazed and fired in a low temperature kiln, lead can migrate from
the pottery glaze into the food. The glazing process uses a flux,
which is a material that, at high temperatures, reacts with and helps
dissolve the components of the glaze. Lead oxide is a commonly used
flux. Factors that determine whether and to what extent lead will
migrate include the temperature and extent of firing of the pottery in
the manufacturing process, temperature and duration of food storage,
and the acidity of the food. It is extremely difficult to quantify the
extent of such exposures in view of the variations in the
manufacturing processes and the quality control practised in the
country of origin. However, the extent of exposure can be quite
significant, particularly among infants. Cases of lead intoxication
from this source have been reported (Wallace et al., 1985).
Lead has been found to migrate from lead crystal glass into
beverages. This problem is especially severe if beverages are stored
in lead-crystal containers, e.g., decanters or liquor bottles (de
Leacy, 1987; Graziano & Blum, 1991). This phenomenon was not observed
with borosilicate glass containers (de Leacy, 1987).
Several studies have been made of lead contamination of foods and
beverages from lead used in the manufacture or repair of metal
vessels. Recoating the inner surface of brass utensils with a mixture
of lead and tin, described as "tinning", is widely practised by
artisans in India (Vatsala & Ramakrishna, 1985). The tin-lead alloy
contains 55 to 70% lead. Water containing tamarind contained
400-500 µg lead/litre after 5 min of boiling. The practice is
considered to be widely prevalent in at least the three southern
states of India. Zhu (1984) described 344 cases of chronic lead
poisoning in Jiansu Province, China, involving people who had drunk
rain water boiled in tin kettles. Analysis of the lead content of
water showed that after boiling the water contained 0.79 to
5.34 mg lead/litre.
5.3 Miscellaneous exposure
5.3.1 Cosmetics and medicines
Some traditional medicines and customs have been found to result
in exposure to high levels of lead, most of which cannot be quantified
with any degree of accuracy. Rather than occurring as trace
ingredients or trace contaminants, various lead compounds are used as
major ingredients in traditional medicines in numerous parts of the
world (Table 18). Clinically overt lead poisoning due to traditional
cosmetics and medicines has been identified among infants (Shaltout et
al., 1981; Fernando et al., 1981; Sharma et al., 1990), children and
adults (Pontifax & Garg, 1985; Cueto et al., 1989; Mitchell-Heggs et
al., 1990; Gupta et al., 1990). There are case reports of lead
toxicity secondary to inhalation of lead from traditional remedies
(Aslam et al., 1979; Shaltout, 1981; Cueto et al., 1989; Sharma et
al., 1990; Mitchell-Heggs et al., 1990).
Often the use is not limited to adults; these may be used on
infants and young children, as well as on women. In Kuwait, the leaded
"kohl", also called "Al kohl", is traditionally applied to the raw
umbilical stump of the newborn in the erroneous belief of a beneficial
astringent action (Fernando et al., 1981). An additional use of lead
metal and lead sulfide is for inhalation of the fumes ("Bokhoor")
produced from heating on hot coals, in the mistaken belief that this
will calm irritable infants and children (Fernando et al., 1981;
Shaltout et al., 1981).
Latin-American countries also report the use of traditional
medicines with high lead concentrations. For example, the Mexican
traditional remedy "azarcon" (lead chromate) and/or "greta" (mixed
lead oxides), distributed as finely ground powders, may contain more
than 70% lead. They are used in the treatment of "empacho", a
gastrointestinal disorder considered to be due to a blockage of the
intestine (Trotter, 1990).
In addition to the potential risks of lead exposure from the use
of traditional medicines, clinical lead poisoning can result from the
lodging of lead shot in vivo (Manton & Thal, 1986).
Table 18. Sources of lead exposure in traditional medicines and cosmetics
Source of lead Comments Reference
(product)
Summa/Kohl used in Indo-Pakistan and other Muslim Aslam et al. (1979);
cultures as eyes preparation; placed on Fernando et al. (1981);
conjunctival surface or as astringent on Shaltout et al. (1981);
umbilical cord stump. Antimony originally Sharma et al. (1990)
used but lead cheaper.
Hindu folk ground seeds and roots as treatment Pontifax & Garg (1985)
medicine for diabetes (8 mg lead/g)
Bokhoor tribal custom to produce lead fumes to Shaltout et al. (1981)
ward off evil
Azarcon lead chromate and mixed lead oxides as Trotter (1990)
treatment for gastrointestinal disorders
in Mexico and southwestern USA
Skin ointments cosmetics used by Chinese actors; skin Lai (1977)
and cosmetics ointment in Europe
5.4 General population exposure
The total intake of lead by adults and children in the general
population varies greatly as to the relative contributions from
individual sources (air, water, food, soil/dust and others) and is
partly dependent on life-style and socioeconomic status. It is beyond
the scope of this review to provide comprehensive information covering
a wide range of circumstances. However, a few simplified calculations
will be given as guidance for carrying out such determinations.
Table 19 gives a summary of the total lead intake and uptake from
the general environment in adults and in children aged 1 to 5. The
assumptions made are shown and are taken from WHO (1987). Additional
intake of lead will take place in certain groups from the use of
tobacco and alcoholic beverages. The estimates given are probably on
the high side with respect to the contribution from air and dust,
since indoor and outdoor lead contributions were considered equal.
Additional intake of lead is possible due to ciliary clearance of
particles 1-5 µm in diameter with subsequent swallowing and
gastrointestinal absorption.
5.5 Blood lead concentrations of various populations
Under certain conditions, blood lead (PbB) levels are a useful
indicator of exposure and are therefore discussed here, as well as in
section 6.1.4. In general, PbB levels correlate better with recent
exposure levels (Lyngbye et al., 1990b). As is discussed in sections
5.5.1 and 5.5.2, the general trend observed in all blood lead surveys
carried out in countries engaged in risk reduction programmes over the
last 15 to 20 years is a fall in the measured levels (OECD, 1993).
PbB concentrations are the most often used estimate of general
exposure to lead. The USA, Commission of European Communities (CEC),
Australia and the WHO have carried out epidemiological surveys to
determine lead exposures in various populations. The designs of these
studies differed; some provided estimates for the country as a whole,
others compared PbB levels of people working in the same occupation in
various countries, and others surveyed general populations but were
not designed to be extrapolated as national estimates. What these
studies have in common is that they did not emphasize groups with high
lead exposures, but concentrated on typical PbB concentrations of
non-occupationally-exposed groups in the regions or countries studied.
These surveys also offer the possibility of examining the data as
distributions, especially at the high end of the distribution profile.
Such examination may identify unusual exposures and thus populations
at risk. Average PbB concentrations may disguise the risk to various
segments of the population.
In the USA, national estimates of the extent and severity of
recent human exposures to lead in the general population were based on
PbB measurements from the second National Health and Nutrition
Examination Survey of 1976-1980 (NHANES II) (Annest & Mahaffey, 1984).
The design of this study permitted extrapolation to the USA population
as a whole.
Table 19. Estimates of lead (µg/day) absorbed by adults and children
from air, dust, food and watera
Mean air Dust Source of lead (µg/day) Total
lead intake absorbed
concentration (mg/day)b (µg/day)
(µg/m3) Air Dust Food Water
Adults
0.3 N.S. 2.4 -- 10 2 14.4
0.5 N.S. 4.0 -- 10 2 16.0
1.0 N.S. 8.0 -- 10 2 20
2.0 N.S. 16.0 -- 10 2 28
Children 1-5 years
0.3 -- 0.6 -- 25 5 30.6
0.5 -- 1.0 -- 25 5 31.0
1.0 -- 2.0 -- 25 5 32.0
2.0 -- 4.0 -- 25 5 34.0
1.0 25 2 12.5 25 5 44.3
1.0 50 2 25.0 25 5 57.0
1.0 100 2 50.0 25 5 82.0
1.0 200 2 100.0 25 5 132.0
a Adapted from WHO (1987); Dust is not considered a significant
source of lead in adults, but is a significant source for workers
where hygiene practices are poor
The above estimates are based on the following assumptions:
Air: Respiratory volume in adults is 20 m3/day, and in children
5 m3/day, and the respiratory absorption is 40%.
Food: Intake of lead by adults 100 µg/day with 10% absorption and
50 µg/day for children with 50% absorption.
Water: A lead concentration of 20 µg/litre, with adult consumption
of 1 litre/day and 10% absorption and for children 0.5 litre/day
with 50% absorption.
Dust: Dust concentration of lead was 1000 µg/g and absorption was
50%.
b N.S. = Not significant
The CEC study was directed toward biological screening of the
extent of exposure to lead outside the workplace using PbB level as
the index of exposure (CEC, 1981). The object of the study was to
assess non-occupational exposure of the population to lead in the
member states; it was implemented by member states and coordinated by
the CEC. Specific populations studied were selected by member states
and 168 separate areas and population groups were investigated in
1977.
Two other major international studies were limited to urban
populations. A pilot study of human lead exposure (Friberg & Vahter,
1983), organized by the United Nations Environment Programme and WHO,
was a collaborative effort under the Global Environmental Monitoring
System (GEMS). The following countries participated in at least part
of the study: Belgium, China, India, Islamic Republic of Iran, Israel,
Japan, Mexico, Peru, USA and the former Yugoslavia. The subjects of
the study were teachers, since they comprise an occupational group not
extensively exposed to lead. An Australian study was based on urban
residents only, and included 651 subjects between 6 and 91 years of
age (Hopper et al., 1982).
Based on demographic, economic and individual variables found in
the USA study to be associated with PbB levels (Mahaffey et al.,
1982), it is clear that results from these major studies cannot be
directly compared. However, a comparison of such studies can provide
information on the exposure to lead of segments of the various
populations in various countries worldwide.
5.5.1 Adult populations
In a large health screening programme within the USA during
1976-1980 (NHANES II), over 27 000 residents aged 6 months to 74 years
were examined (Annest & Mahaffey, 1984). PbB concentrations were
determined in a subsample (9933 individuals) and yielded an arithmetic
mean value of 0.67 µmol/litre (13.9 µg/dl). In 5841 individuals aged
18-74 years, the overall mean was 0.68 µmol/litre (14.1 µg/dl)
(Roberts et al., 1985). A sex difference was noted: 0.77 µmol/litre
(16.1 µg/dl) was found in males and 0.57 µmol/litre (11.9 µg/dl) in
females. Residents in the centre of large urban areas were found to
have a PbB level of 0.72 µmol/litre (14.9 µg/dl), while rural
residents it was 0.62 µmol/litre (13.0 µg/dl). Because of the design
of the study, information on PbB levels, in relation to smoking,
alcohol consumption, and occupational status, were available. Male
workers in occupations with a high potential for lead exposure had
mean PbB levels of 0.78 µmol/litre (16.2 µg/dl) for nondrinkers/
non-smokers, 0.92 µmol/litre (19.2 µg/dl) for non-drinkers/non-smokers
and 0.95 µmol/litre (19.7 µg/dl) for drinkers/smokers. It was noted
that a true decrease of 37% in PbB level occurred during the period of
the survey (Annest et al., 1983).
Rabinowitz & Needleman (1982) reported an arithmetic mean
umbilical cord PbB concentration of 0.32 µmol/litre (6.6 µg/dl) (range
0-1.78 µmol/litre; 0-37 µg/dl) in over 11 000 samples collected
between 1979 and 1981. A decrease in PbB level of approximately 11%
was noted during the period of collection.
Friberg & Vahter (1983) reported the PbB levels of 200 teachers
from nine countries (see section 5.5). The median PbB level in this
study ranged from 0.29 µmol/litre (6 µg/dl) in Beijing and Tokyo to
1.06 µmol/litre (22 µg/dl) in Mexico City. Smokers generally had
higher PbB levels than non-smokers.
During the period 1978-1988 marked decreases (approximately 30 to
40%) in the average PbB levels of adults were noted in Belgium,
Germany, New Zealand, Sweden, the United Kingdom, and the USA (OECD,
1993).
5.5.2 Children
Results from the NHANES II survey in the USA indicated a mean PbB
level for children (6 months to 2 years old) of 0.78 µmol/litre
(16.3 µg/dl). It was noted that 18.6% of black inner city children had
PbB levels > 1.44 µmol/litre (30 µg/dl) whereas only 4.5% of white
children had such PbB concentrations. The percentage of children with
high PbB level decreased with increasing family income (Roberts et
al., 1985). The NHANES II data were analysed in detail for information
on the demographic correlates of children's PbB concentrations
(Mahaffey et al., 1982; Annest et al., 1983). The racial differences
in these proportions were significant among both boys and girls. The
proportion with elevated PbB concentrations was slightly higher among
boys than girls for both races. Among both black and white children,
the percentage with elevated PbB concentrations decreased with
increasing family income. Proportionately more young children in urban
than rural areas and in the central cities of large urban areas had
elevated blood lead concentrations.
PbB concentrations were measured in 286 Finnish children living
in the three largest cities of Finland (number of subjects = 172), in
rural areas (54 subjects) and in a lead smelter area (60 subjects)
(Taskinen et al., 1981). The mean PbB levels in urban, rural and
lead-smelter areas varied between 0.29 µmol/litre and 0.32 µmol/litre,
range 0.14-0.82 µmol/litre (6.0 and 6.7 µg/dl, range 3-17 µg/dl).
There were no significant differences between the areas of residence.
The five children who lived within 500 metres of the lead smelter
had a mean PbB concentration of 0.44 µmol/litre, range 0.24-0.62
µmol/litre (9.2 µg/dl, range 5-13 µg/dl), which was significantly
higher than the mean blood lead concentration among children living
in the rest of the country.
In a study carried out in Sweden, which included 1781 samples
obtained from children during 1978-1988, the mean PbB level decreased
from 0.29 µmol/litre (59.6 µg/litre), range 0.09-1.20 µmol/litre
(18-250 µg/litre) in 1978 to 0.16 µmol/litre (32.9 µg/litre), range
0.07-0.34 µmol/litre (15-71 µg/litre) in 1988 (Schutz et al., 1989).
In Finland, a remarkably similar decrease has been reported; the mean
PbB value was 0.14 µmol/litre (30 µg/litre), range 0.1-0.40 µmol/litre
(21-41 µg/litre) among 35 children in the Helsinki area in 1988 (Ponka
et al., 1991).
As in the USA (Annest et al., 1983), children's PbB
concentrations in Sweden have decreased in recent years. Levels were
determined each summer during the period 1978-1984 in children from
Scania in Southern Sweden (Skerfving et al., 1986). The average PbB
concentration was 0.26 µmol/litre, range 0.07-1.20 µmol/litre
(5.5 µg/dl, range 1.4-25.0 µg/dl). There was a statistically
significant decrease over time in both the rural and urban areas,
averaging about 0.019 µmol/litre (0.4 µg/dl) per year.
Decreases of 25-45% in average PbB levels in children between
1978 and 1988 have been reported in Belgium, Canada, Germany, New
Zealand, Sweden and the United Kingdom (OECD, 1993).
5.5.3 Remote populations
In contrast, a much higher mean value of 0.16 µmol/litre
(3.4 µg/dl) was obtained in Nepal upon examination of 103 children
living in the Manang District, which is also very remote and thought
to be free of anthropogenic sources of lead (Piomelli et al., 1982).
However, these people are exposed to significant amounts of natural
lead from living in dusty smoke-filled houses, and from burning pine
and yak dung. The lead level in the houses was about 0.15 µg/m3
(Davidson et al., 1981). For comparison outdoor air in Woods Hole,
Massachusetts, USA, contains 0.004 and Boston, Massachusetts, 0.01 to
0.05 µg lead/m3.
Another remote population studied was from Okapa, Eastern
Highlands, Papua New Guinea (Poole et al., 1980). Extreme care was
taken in sampling and analysis, and the precision was acceptable at
PbB levels as low as 0.24 µmol/litre (5 µg/dl). The children subsisted
on root crops and a small amount of tinned fish and meat. Villages
were typically hazy with wood smoke, natural mineral pigments were
used extensively, and tobacco smoking was common. This survey of 100
children yielded a mean PbB level of 0.25 µmol/litre (5.2 µg/dl), well
above the detection limit, and, as reported by Poole et al. (1980),
far less than the mean of 1.10 µmol/litre (23 µg/dl) for Sydney,
Australia, in 1974.
5.6 Occupational exposure
Occupational exposure to lead which results in poisoning, both
moderate and clinically symptomatic, still occurs in many countries of
the world. Although adults are mainly involved, in many countries,
especially in those with developing industries and small home-based
industries, the distinction between home and workplace is non-existent
(Verrula & Noah, 1990) and children are exposed to workplace lead.
Until recently, in the occupational setting, there was only concern
for the identification of late stage, highly symptomatic cases of lead
poisoning resulting in crippling neurological manifestations of lead
poisoning such as palsy and encephalopathy (Chakravorti & Bhar, 1978;
Wang, 1984; Davies, 1984; Lin-Fu, 1985). However, there is now concern
for lower exposures to lead.
In many countries occupational lead exposure is entirely
unregulated and no monitoring of exposures exists. Automobile battery
manufacture and repair, radiator repair, secondary smelters (including
scrap metal refiners) are found in most countries. The industries
where there is a potential for lead exposure are listed in Table 10
(section 5.1.3). Significant lead exposures are not limited to
traditional heavy industries. For example, Kaye et al. (1987)
identified exposures from leadborosilicate dust used in a capacitor
and resistor plant in the USA.
Because of transfer of lead to the fetus (in utero) and the
transport to the home of lead on clothing, etc., thereby exposing the
young child in the home, the problems of occupational exposures to
lead are not limited to the workplace per se. Cases of poisoning
among the children of lead workers have been reported (Baker et al.,
1979; Wang, 1984; Ryu et al., 1985).
Although most occupational standards are based on airborne lead
only, this route of exposure does not fully reflect the total daily
exposure of workers, also exposed to lead in food, water, alcoholic
beverages and dusts.
The potential for hazardous exposures to lead during lead
smelting and refining are well recognized, particularly where molten
lead and alloys are poured, resulting in the vaporization of metal.
This is true for both primary new metal and secondary (lead scrap)
smelters and refineries.
Small domestic versions of secondary smelters exist in a large
number of countries. These are typically located within or in close
proximity to homes. For example, in Jamaica there has been a rapid
proliferation of lead smelters, particularly illegal backyard smelters
(Rodney & Lee, 1985). The lead fumes and dust generated in such
operations pose an exceptional health hazard to children and adults
living near these operations. Rodney & Lee (1985) reported that 51% of
116 children (aged 2 to 12 years, mean 5.9 years) and 60% of 235
adults working in or living near lead smelting factories had PbB
concentrations of 1.92 µmol/litre (40 µg/dl) or more.
Other occupations where workers have been shown to be at risk
from airborne lead include electric storage battery manufacturing,
particularly where industrial hygiene is poor (Barrio & Badia, 1985);
demolition, welding and shipbreaking where lead-based paint is present
(Holness & Nethercott, 1988; Nosal & Wilhelm, 1990); pottery and
ceramic-ware production, which is often a home-based operation
involving women and children (MolinaBallesteros et al., 1983; Katagiri
et al., 1983; Kaye et al., 1987); small businesses repairing
automobile radiators (Matte et al., 1989a,b; Verrula & Noah, 1990),
and artisans producing jewellery and decorative wares. This latter
industry is of particular concern since it is predominantly carried
out at home or in non-regulated shops, often by women and children
(Behari et al., 1982). Indian silver jewellery makers were found to
have a PbB level of 5.8 µmol/litre (121 µg/dl) compared to non-exposed
controls with a PbB level of 1.3 µmol/litre (27 µg/dl) (Behari et al.,
1982).
6. KINETICS AND METABOLISM IN LABORATORY ANIMALS AND HUMANS
6.1 Absorption
The absorption of lead from environmental sources is not
dependent solely on the amount of lead presented to the portals of
entry. It is also dependent on the physical and chemical state in
which the metal is presented, and it is influenced by host factors
such as age, physiological status, nutritional condition and,
possibly, genetic factors. Men engaged in heavy work breathe more air
and eat more food than sedentary individuals of the same weight, and
on a body weight basis children eat almost as much food and breathe
almost as much air as middle-aged adults.
Considerable data from human subjects are available. Therefore,
discussion of animal studies will be limited to areas where the
information on humans is inadequate. Various models used to predict
body burden or distribution of lead have been developed (Bernard,
1977; Marcus, 1985a,b,c; Bert et al., 1989; Arnetz & Nicolich, 1990).
A full review of these models has been judged to be beyond the scope
of this document. However, some of them were used in section 6.1.4
when correlating intake and body burdens.
6.1.1 Absorption after inhalation
The absorption of lead from air to blood involves two processes:
the deposition of airborne lead particles in the respiratory tract;
and the absorption and clearance from the respiratory tract into the
circulation. Using the International Radiological Protection
Commission (IRPC) document on lung dynamics (Task Group on Lung
Dynamics, 1966), a model was developed which predicted that 35 to 50%
of inhaled lead is deposited in the respiratory tract (40-50% of
particles with a mean mass median aerodynamic diameter (MMAD) of
0.5 µm, such as are typically generated by automobiles). These are
deposited primarily in the alveolar sacs of the lung. Lead fumes and
vapours, such as those generated in operations where metals are cut or
heated, are of very small size and are respirable. Absorption after
deposition will vary according to the solubility of the lead species
(e.g., lead carbonate or lead chloride aerosols) and the inherent
toxicity to lung macrophages and cilia.
6.1.1.1 Animal studies
Limited animal studies confirm that there is almost complete
absorption of lead particles (0.1 to 0.5 µm in diameter) deposited in
the lower respiratory tract. In rats the clearance half-time from lung
is short (less than one hour) and 90 to 98% of the administered dose
is absorbed within about 48 h (US EPA, 1986a).
6.1.1.2 Human studies
The respiratory deposition of airborne lead is in the range of
30-50% and varies with particle size and ventilation rate (US EPA,
1986a). Higher deposition rates may occur with larger particles, but
this deposition takes place in the upper respiratory tract, with
eventual displacement to the gastrointestinal tract and absorption via
the ingestion route. This probably explains the observation of Kehoe
(1961) that faecal excretion of lead increased after a subject
breathed for many weeks aerosols of lead oxide (150 µg/m3) with an
MMAD of approximately 2.9 µm. In contrast, smaller particles of
inhaled lead, such as those generated by automobile exhaust,
regardless of physicochemical form, are almost (> 90%) completely
absorbed after deposition in the lower respiratory tract (Rabinowitz,
et al., 1977a; Chamberlain et al., 1978; US EPA, 1986a).
There are no quantitative data on the absorption of lead in
children after inhalation exposure. It is known that young children
weighing only one sixth of an adult inhale 40% of the daily volume of
an adult and a proportionately higher daily air volume per unit
measure (weight, body area) than do adults (Barltrop, 1972). After
controlling for weight and taking into account differences in the
anatomy of the respiratory tract between adults and children, James
(1978) calculated a rate of deposition of lead particles in children
which was 1.6 to 2.7 times that of adults.
6.1.2 Absorption of lead from the gastrointestinal tract
In the case of older children and adults without occupational
exposure, lead absorbed by the gastrointestinal tract comes from the
intake of lead in foods, beverages and soil/dust. In pre-school
children, there is concern over the intake of both food and non-food
items (e.g., toys, soil/dust). Young children may take in lead from
non-food items, via normal mouthing activity, which in the extreme, is
the behavioural trait pica, which refers to the ingestion of such
materials as soil, ash, paint chips and plaster (US EPA, 1986a). For
infants and young children, the extent of absorption of the lead in
dust/soils from the gastrointestinal tract is extremely important,
particularly for children living in urban environments.
6.1.2.1 Animal studies
The absorption of lead from the gastrointestinal tract in
experimental animals is age dependent and is modified by the level of
food intake. In 1-week-old suckling rats, an absorption rate of about
52% was reported after a single oral dose of lead chloride, compared
to 0.4% in 6-week-old animals (Kostial et al., 1978). Furthermore,
Aungst et al. (1981) reported that rat pups had higher tissue levels
of lead than adults after a single gavage dose of 1 or 10 mg lead (as
lead acetate) per kg body weight. Fasting markedly enhanced the uptake
of lead in the gastrointestinal tract of experimental animals (Garber
& Wei, 1974; Pounds et al., 1978).
In experimental animals, absorption of lead from the
gastrointestinal tract appears to be a saturable process. With
increasing doses (1 to 100 mg lead (as lead acetate) per kg body
weight), lead absorption as a percentage of dose was found to decrease
from 42% to 2% (Aungst et al., 1981; Bushnell & DeLuca, 1983) in
dietary studies. Such data are consistent with a saturable active
transport process across the gastrointestinal tract.
The low rate of absorption of lead from soluble lead salts noted
in adult rats may reflect a dietary effect. Kostial & Kello (1979)
reported less than 1% absorption in adult rats given chow diets,
whereas the absorption rate was 3-20% when rats consumed diets similar
to that of humans (milk, bread, baby foods, etc). This range more
closely resembles the human absorption rates.
The chemical form of lead can influence its bioavailability.
Stone et al. (1981) determined the biological availability of lead
intrinsically incorporated into the soft tissues of oysters. The
bioavailability to the rat was 10-30% lower than that of lead acetate
added to the basal chow diet. Henning & Cooper (1988) compared the
absorption of lead from rat milk labelled in vitro with lead-203 and
lead chloride or lead acetate solutions. Lead from soluble salts
accumulated primarily in the duodenum, to some extent in the jejunum
and minimally in the more distal small intestine. Lead from milk
accumulated only in the upper ileum. After 20 h negligible lead-203
was found in any region following administration of soluble salts, but
there was substantial retention of lead in the ileum after dosing with
milk.
Data obtained from a feeding study in rats (Dieter et al., 1993)
showed that lead uptake into rat femurs was highly dependent on the
chemical form of lead administered. Bioavailability was highest for
lead acetate, intermediate for lead oxide, and lowest for lead sulfide
and Alaskan mixed ore concentrate. This uptake was linearly related to
dose over the range studied. However, the slopes of the linear
regression equations differed according to the form of lead. They were
0.10 µg lead/g femur per kg diet for lead sulfide and 2.64 for lead
acetate.
However, results of an investigation in young swine of the
absorption of lead from mining and/or milling operation waste (LaVelle
et al., 1991) differ from those reported by Dieter et al. (1993). The
chemical species (predominantly lead sulfide) and particle size (i.e.
larger than 100 µm) were expected to result in these sources being
less bioavailable than soluble lead salts such as lead nitrate.
However, experimental data indicate that the lead in mine tailings may
be about 2-3 times more available than reagent grade lead sulfide
under the conditions of the study. Additionally, Freeman et al. (1992)
reported that the lead in mining waste soil was between 8 and 20% as
bioavailable as lead acetate. No detailed comparisons were made of the
physicochemical properties of these two mine wastes. An additional
study involving the absorption of lead from lead sulfide ores and
their oxidation products in rabbits and in vitro showed that lead
from minerals was absorbed less well than from lead acetate by a
factor of 5 (Davis et al., 1990a). The significance of these phenomena
is not clear.
6.1.2.2 Human studies
Gastrointestinal absorption of lead in humans, as in experimental
animals, is influenced by dietary factors, nutritional status and the
chemical form of the metal. Overall patterns of food intake may also
influence lead absorption. For example, lead ingested during periods
of fasting is absorbed to a much greater extent than lead ingested
with food. Chamberlain et al. (1978) reported 45% absorption of lead
chloride in fasting subjects and only 6% in feeding subjects. Using a
similar procedure, Heard & Chamberlain (1982) reported absorption of
63.3% in fasting subjects. Using multiple stable isotope lead tracers,
it was found that in adult men the gastrointestinal absorption rate
with food containing lead nitrate or lead cysteine was 6-12%. However,
when consumed under fasting conditions, lead nitrate, lead sulfide and
lead cysteine were absorbed at 16-53% (Rabinowitz et al., 1977b).
Kehoe (1961) estimated only 10% net absorption of dietary lead by
adults in long-term metabolic studies. However, with appropriate
calculation of biliary clearance as well as of urinary excretion, a
figure of 15% was estimated by Chamberlain et al. (1978). In several
studies on adult humans, absorption of lead was reported to be 14%
when it was administered with food (Chamberlain et al., 1978; Moore et
al., 1979; Rabinowitz et al., 1980).
Ziegler et al. (1978) reported that young children, aged two
weeks to two years, absorbed 42% of ingested lead at levels of intake
greater than 5 µg/kg body weight. Drill et al. (1979) estimated an
absorption rate of 17% for lead in paint chips in children aged 2 to 3
years. These authors also estimated a 30% gastrointestinal absorption
rate for lead in soil and dirt.
The amount of lead ingested by children from non-food items such
as soil, dust and paint chips through normal mouthing activity,
particularly for children with pica, is a major concern in calculating
paediatric lead exposures. Using data from Day et al. (1975) and Lepow
et al. (1974), it has been estimated that children 2 to 3 years of age
ingest about 100 mg soil per day. Using aluminium and titanium as
tracers, Clausing et al. (1987) estimated that children aged 2 to 4
years ingest between 50 and 100 mg of soil daily. The average amount
of soil ingested by young children has recently been estimated to be
between 12.5 and 21 mg/day (SAHC, 1993).
6.1.2.3 Nutritional status and lead absorption via gastrointestinal
tract
It has been known for some time that the absorption and
distribution of lead are affected by nutritional status in both
experimental animals and humans (Sobel et al., 1940). Nutritional
inadequacies can also affect the toxic response to lead (see sections
7 and 8). In view of documented nutritional inadequacies in many parts
of the world, such interrelationships become crucial in assessing the
full risk from lead exposures.
Vitamin D, calcium and phosphorus have complex and interrelated
effects on lead absorption (Fullmer, 1990). Increasing the
concentration of 1,25-dihydroxycholecalciferol, the active metabolite
of vitamin D, either exogenously or endogenously, increases
gastrointestinal absorption of lead (Fullmer, 1990). However, this
effect is dependent upon the duration of lead exposure and the
magnitude of body lead stores. This homoeostatic mechanism for calcium
and its dependence on nutritional status, as well as body burden of
lead, is complex. This may explain the divergent results of the
observed interaction in children (Rosen et al., 1980) or the lack of
association (Koo et al., 1991) of lead with vitamin D metabolism.
In experimental animals, chronic ingestion of diets with less
than adequate amounts of calcium (Quarterman & Morrison, 1975),
phosphorus (Quarterman & Morrison, 1975), iron (Mahaffey-Six & Goyer,
1972; Ragan, 1977), selenium (Stone & Soares, 1976) or zinc
(Cerklewski & Forbes, 1976) increases the fractional absorption of
lead in the gastrointestinal tract. From the work of Fullmer & Rosen
(1990), it appears that lead alters the transport of calcium and other
trace minerals by affecting carrier proteins, rather than by competing
at the mucosal surface.
The influence of iron on lead absorption in humans was assessed
in double-labelling experiments conducted by Watson et al. (1980).
Lead-203 and iron-59 were given to 28 subjects in their diet. There
were significant positive correlations between iron levels in food and
lead absorption. However, the association was not strong because 50%
of those absorbing excess lead also absorbed excess iron.
Rats fed iron-deficient diets have increased concentrations of
lead in kidney and bone when compared to rats ingesting equivalent
quantities of lead (as lead acetate) in drinking-water while being
fed an iron-adequate diet (Mahaffey-Six & Goyer, 1972). In contrast
with calcium deficiency, iron deficiency does not result in a
redistribution of lead to non-osseous tissue (Mahaffey-Six & Goyer,
1972). The degree of iron deficiency does not need to be severe to
increase retention of lead. For example, Ragan (1977) demonstrated
six-fold increases in tissue lead in rats when body iron stores were
reduced, but before frank iron deficiency developed. High levels of
dietary iron resulted in decreased kidney, femur and blood lead
concentrations in rats (Mahaffey, 1985). Iron appears to increase
absorption rather than decrease lead excretion (Barton et al., 1978).
Lead interferes with normal transferrin transport of iron, and
inhibits transferrin endocytosis and iron transport across the cell
membrane of the reticulocyte (Qian & Morgan, 1990).
6.1.3 Dermal absorption
6.1.3.1 Human dermal absorption
Moore et al. (1980) examined the uptake of lead acetate from two
hair-darkening cosmetics through the skin of eight human volunteers.
Only minute quantities of lead (0-0.3% of the applied dose) were
detectable in blood, and there was only a slight increase in
absorption when the skin was damaged. Lilley et al. (1988) and
Florence et al. (1988) have reported the dermal absorption of
inorganic lead compound leading to elevated levels of lead in human
saliva and sweat.
Dermal absorption of inorganic lead through unabraded human skin
is considered to be minimal.
6.1.4 The relationship of external lead exposure to blood lead
concentration
There are numerous environmental media that provide routes by
which humans are exposed to lead: food, water, air, soil, dust.
External exposures are the sum of the quantities of lead consumed from
all sources.
Historically there have been two lines of approach in
understanding the lead exposure and blood lead relationship. Most have
been empirical and have measured environmental lead and PbB levels
either at one time or repeatedly. With these observed correlations and
with no assumptions about how lead moves inside the body, many
reasonable predictions can be made. The validity of these predictions
is optimum when only one environmental source dominates. Some of these
rely on linear functions, while others have specified non-linearities,
especially over very wide ranges (more than a factor of 10) of lead
exposures. However, when multiple sources are considered these
predictive models have been less satisfactory.
Internal lead levels in human populations have been estimated by
analyses of a variety of biological tissues (e.g., blood, teeth, bone
and hair). Lead concentrations in each of these have particular
biological meanings with regard to external exposure to lead. Blood is
the compartment in which lead is most often measured as a marker of
exposure. However, it typically represents relatively recent
exposures, since the half-life of lead in blood is short (US EPA,
1986a) and has been estimated to be in the order of 36 days from
tracer studies (Rabinowitz et al., 1975). Lead in blood is derived
from levels in the current environment and from lead stored in tissues
(particularly bone) that re-enters the blood during tissue
mobilization (Manton, 1985). Although PbB concentration reflects
recent exposure and bears a consistent relationship to levels of lead
in the external environment, when bone mobilization is accelerated a
greater fraction of PbB will be derived from tissue stores.
To date only a few studies have utilized a multimedia approach
relating lead intake to PbB levels. The majority of studies have
attempted to correlate PbB levels and lead concentrations in specific
media.
6.1.4.1 Ambient air
a) Occupational exposure
Several studies have examined the blood lead: air lead
relationship for workers exposed to levels of airborne lead between 9
and 450 µg/m3 (King & Conchie, 1979; Gartside & Buncher, 1982;
Bishop & Hill, 1983). The results of all three studies are in general
agreement over the wide range of airborne lead studied and for PbB
levels between 0.96 and 4.32 µmol/litre (20-90 µg/dl). The blood lead:
air lead relationship in occupational settings is best described by a
curvilinear relationship having slopes between 0.00096 and
0.0038 µmol/litre (0.02 and 0.08 µg/dl) per µg/m3 air.
b) Non-occupational exposure
Both population and experimental studies have been used to
estimate the PbB: ambient air lead relationship in adults and
children. Under ambient conditions (air lead concentrations of
0.1-2.0 µg/m3) and PbB levels less than 1.44 µmol/litre
(< 30 µg/dl), the relationship has been described as linear (Colombo,
1985). The various slope estimates reported are based on the
assumption that an equilibrium level of lead in blood is achieved.
Based on three major studies (Yankel et al., 1977; Angle & McIntire,
1979; Roels et al., 1980), the median slope for children is about
0.091 µmol/litre (1.9 µg/dl) blood per µg/m3. In adult males a slope
estimate of 0.076 µmol/litre (1.6 µg/dl) blood per µg/m3 was
calculated. When one calculates the relationship between PbB and the
total contribution from air (direct inhalation plus indirect through
dust/soil), a value of about 0.14-0.24 µmol/litre (3-5 µg/dl) blood
per µg/m3 is obtained (Brunekreef, 1984; US EPA, 1986a). Also on the
basis of a linear model, Snee (1981) reported that the best estimate
of the blood lead: air lead relationship was 0.048 µmol/litre
(1 µg/dl) per µg/m3. After examining available epidemiological and
experimental data, Chamberlain (1983) concluded that most published
estimates of the slope were between 0.072 and 0.144 µmol/litre per
µg/m3. From these results, it can be concluded that airborne lead
will only be a major contributor to PbB levels in areas of high air
lead levels.
6.1.4.2 Food
The relationship of PbB to dietary intake has been estimated from
experimental (Stuik, 1974; Cools et al., 1976; Schlegel & Kufner,
1979) as well as population studies (Sherlock et al., 1982; UK, 1982;
Ryu et al., 1983). In adults, the results from both types of studies
using both linear and cube root models indicated a relationship of
between 0.0019 and 0.0028 µmol lead/litre (0.04 and 0.06 µg/dl) per µg
lead intake per day. From the study of Ryu et al. (1983) a slope of
0.0096 µmol/litre (0.2 µg/dl) per µg lead/day was obtained for infants
aged 8 to 196 days.
Currently, data are available for adults and children from
studies with careful control of important variables such as: intake of
dietary lead and of other dietary constituents, minimal exposure to
sources other than diet in the studies of infants, and intake/blood
lead measurements that can be used to estimate intake from all
sources. For infants these are studies reported by Sherlock et al.
(1982), UK (1982), Ryu et al. (1983), and Lacey et al. (1985). Studies
by the UK (1982) and by Sherlock et al. (1982) were conducted at
higher levels of lead exposure than were the studies conducted with
infants by Ryu et al. (1983). The latter studies were at exposure
levels associated with PbB concentrations under 0.96 µmol/litre
(20 µg/dl); the ratio of blood lead to ingested lead was
0.0076 µmol/litre (0.16 µg/dl) per µg lead ingested per day. Exposure
levels were low: average dietary lead intake was 17 µg/day. After four
months, the average PbB concentration was 0.29 µmol/litre (6.1 µg/dl)
whole blood. Between the ages of 4 and 6 months, 10 children remained
at a dietary lead intake of 0.76 µmol/litre (16 µg/dl). Their PbB
concentrations were quite constant at 0.35 µmol/litre (7.2 µg/dl) at
the end of the study period. The remaining seven children received
canned infant formula and/or milk during this period and their average
dietary lead intake was 61 µg/day. At the end of the study period,
their PbB concentration was 0.69 µmol/litre (14.4 µg/dl). Based on
these data, a curvilinear relationship between blood lead and total
lead intake was suggested, a 4-fold increase in lead intake resulting
in a doubling of PbB concentration. Sherlock et al. (1982) conducted a
duplicate diet study of 31 mothers and their children from Ayr,
Scotland. The slope for adults was substantially lower than for
children.
6.1.4.3 Drinking-water
There is still debate over the most appropriate model (i.e. cube
root, polynomial or logarithmic) to describe the curvilinear
relationship of waterborne lead to blood lead. The highest estimates
for the contribution of water lead to blood lead come from the cube
root and logarithmic models. Much lower estimates are obtained from a
linear model (Pocock et al., 1983; US EPA, 1986a). In a study by
Sherlock et al. (1982), a cube root relationship between lead levels
in drinking-water and blood fitted the data more closely than a linear
relationship. The curvilinear model implies that as abatement
processes lower water lead concentrations, there will be an increasing
benefit in lowering of population PbB levels (Moore, 1983).
6.1.4.4 Soil and dust
It is extremely difficult to choose the most appropriate model to
describe the soil/dust to blood lead relationship, given the many
variables involved in determining the exposure patterns of children
and the kinetics involved between the levels in the environment and
the child. A review of the available studies shows the extreme
variability in slopes obtained (0.028-0.36 µmol lead/litre
(0.6-7.6 µg/dl) blood for each 1000 µg/g soil and 0.00096-0.35 µmol
lead/litre (0.02-7.2 µg/dl) blood for each 1000 µg/g dust consumed by
children) (US EPA, 1986a). Detailed consideration has been given to
the process for the assessment of lead contamination in soil and the
derivation of soil clean-up criteria by Wixon (1991). The guideline
model uses a PbB target and slope for the soil:blood lead relationship
in the particular community in order to derive a PbB guideline. For
house dust a median value of 0.086 µmol lead/litre (1.8 µg/dl) blood
per 1000 µg lead/g dust in children appears to be based on data of
reasonable quality, as does the 0.105 µmol lead/litre (2.2 µg/dl)
blood per 1000 µg lead/g soil from the same authors.
According to Elwood (1986), studies of the association between
lead in house dust and PbB have given inconsistent results and, in
general, the only studies in which statistically significant
association has been found are those where lead levels in dust are
quite high. Landrigan et al. (1975) found a highly significant
association between PbB and dust lead in an area with a mean level of
4022 µg lead/g dust, but not in two areas with means of 922 and 816 µg
lead/g dust. The US EPA (1986a) summarized a series of studies from
which the overall relationship was judged to be 0.86 µmol/litre
(18 µg/dl) blood per 1000 µg lead/g dust. Duggan (1980) reviewed the
literature and determined that a slope of 0.24 µmol/litre (5 µg/dl)
per 1000 µg lead/g dust is reasonable.
The overall relationship between PbB and dust/soil lead depends
on the lead concentrations and bioavailability as well as on the
proximity and linkage between humans and their environment. This
relationship varies among locales.
6.1.4.5 Total lead intake
The non-linear relationship between PbB and total lead intake
(see section 5.5) is curvilinear across a broad range of PbB values,
such that the slope decreases with increasing lead levels. A number of
biological factors may explain the curvilinear relationships, such as
increased renal clearance with high PbB (Chamberlain, 1983, 1985),
distributional non-linearities due to differences in lead binding
sites in different tissues (Hammond et al., 1981; Manton, 1985;
Marcus, 1985b), or a sizeable pool of mobile lead in bone maintained
more or less independently of uptake (Rabinowitz et al., 1976;
Chamberlain, 1983).
6.2 Distribution
The initial distribution of lead in the body may depend upon the
rate of delivery of blood to various organs. However, it would appear
that distribution occurs in a similar manner regardless of the route
of absorption (Kehoe, 1987). The distribution of lead in humans under
environmental exposure conditions reflects the fact that almost all
exposures are chronic rather than acute.
6.2.1 Animal studies
Studies in rats have shown that lead is rapidly distributed into
soft tissues and subsequently redistributed into soft and mineralizing
tissues after acute and chronic exposures. After acute inhalation
(Morgan & Holmes, 1978) or oral (Aungst et al., 1981) exposure, lead
levels in rats were highest in liver, kidneys and lung, with levels
increasing in bone as those in soft tissues declined and stabilized.
Similar distribution patterns were reported for mice exposed for 12
months to 21.5 µg lead/m3, the highest levels being found in bone
and the lowest in lung (Keller & Doherty, 1980a).
Age-related differences in the distribution of lead in
experimental animals have been reported. Kostial et al. (1978) noted a
greater retention of lead in suckling rats than in adults; the levels
in the brain were also higher in the pups. A 2- to 3-fold increase in
brain lead concentration (highest in hippocampus) was noted after a
10-fold (0.1 to 1 mg/kg body weight administered by gavage) increase
in the dose given to 4- to 8-week-old rat pups (Collins et al., 1992).
Ageing has been shown to alter the pattern of distribution of
lead in rats administered lead acetate in drinking-water. Juvenile (21
days old), adult (8 months old) and elderly (16 months old) rats
received 0, 1.27 and 6.37 mg lead/kg bodyweight for 9.5 months. The
pattern of distribution, namely femur > liver > brain was similar in
all age groups. However, age-related increases in brain lead levels
were noted, along with decreases in femur lead content (Cory-Slechta,
1990a).
6.2.2 Human studies
Lead is distributed to both soft tissues (blood, liver, kidney,
etc.) and mineralizing systems (bone and teeth). Bone may be affected
adversely by lead but also serves as the body's major storage site.
Bone accumulates lead over much of the human life span, and a study of
the kinetics of distribution is important since bone can, under
appropriate conditions, pose a risk as a potential endogenous source
of lead.
Once absorbed, lead is not distributed homogeneously throughout
the body but rather into several distinct compartments. Such
biokinetic movements have been explained by Rabinowitz et al. (1976)
using a three-compartment model. This model was based on tracer and
balance data from five healthy men and has been refined by Marcus
(1985a,b,c). Three pools (blood, bone and soft tissues) were
identified, with lead having distinct half-lives in each. Blood lead
was considered the most labile compartment with a half-life of about
36 days, and bone lead the most stable with a half-life of about 27
years. Lead in soft tissue had a half-life of approximately 40 days.
The specific physiological foundations for these biokinetic
models have received much attention and are currently being refined
(O'Flaherty, 1991, 1993). A more thorough understanding of bone
growth, for example, expressed as a series of allometric equations,
should help improve models, especially when they must be applied to
growing and maturing humans.
Under steady-state conditions, about 96% of PbB is in the
erythrocyte. At PbB concentrations of < 1.92 µmol/litre (40 µg/dl),
whole blood and serum lead levels increase linearly in a positive
manner. At higher PbB concentrations a curvilinear relationship is
apparent and the serum to blood ratio increases dramatically (Manton &
Cook, 1984). Such kinetic relationships may be altered during
pregnancy. From in vitro data (Ong & Lee, 1980), fetal haemoglobin
appears to have a greater affinity for lead than adult haemoglobin.
In adults, approximately 94% of the body burden of lead is in the
bones, whereas only 73% of the body burden in children is located in
this compartment (Barry, 1975, 1981). In view of the extremely long
half-life for lead in bone, this compartment can serve as an
endogenous source of lead to other compartments long after exposure
ceases (O'Flaherty et al., 1982; Kehoe, 1987). This is due to the
labile lead compartment in bone (Rabinowitz et al., 1976) and the
ongoing bone redistribution which is subject to alteration by in vivo
metabolic processes (Parfitt, 1990). Although lead in bone generally
increases continuously with age, there is evidence that lead levels in
some bones (e.g., mid-femur and pelvic bone) plateau at middle age and
decrease with further ageing (Drasch et al., 1987). It is difficult,
however, to determine the role ageing plays in this process compared
to other biological reactions. Ageing may account in part for the
20-25% increase in PbB levels in menopausal women noted by Silbergeld
et al. (1988).
The metabolism of lead in bone has been summarized in reports by
Barry (1975, 1981), Drasch et al. (1987), Silbergeld et al. (1988),
Skerfving (1988) and Rabinowitz (1991).
Until recently, it was widely held that the human skeletal system
provided a metabolically inert depository for lead and was of little
consequence in health-risk assessment. It had been assumed that bone
lead is metabolically inert, with a half-life long enough to forestall
the risk of ready transfer back to blood. Current evidence is that
bone comprises a set of kinetically variable subcompartments for lead
deposition and is a target for toxicity. These factors complicate bone
lead kinetics as applied to long-term modelling; the mobility of bone
lead to blood is important (Rabinowitz, 1991).
Bone lead is readily mobilized to blood and the effect is most
apparent in people with a history of occupational exposure; bone lead
also appears to be a major source of blood lead in older people with
previous ambient exposures to lead. Of particular importance is
mobilization of lead from bone in pregnant women and nursing mothers
(Silbergeld, 1991). The mobilization of lead from bone to the more
bioavailable maternal blood compartment poses a risk to the fetus and
mother.
Human bone appears to have at least two, possibly three,
kinetically distinct lead compartments. Lead in trabecular (spongy)
bone appears to be more mobile than lead lodged in cortical (compact)
bone, and there appears also to be a fraction of bone lead in
equilibrium with the lead in blood (Skerfving, 1988). Trabecular bone
seems to be an important source of resorbed lead when high exposure is
reduced, e.g., through removal of medical reasons by retirement of
lead workers, or in response to chelation in adults (Shutz et al.,
1987).
6.2.3 Transplacental transfer
Lead is readily transferred from the mother to the developing
infant during pregnancy and accumulates in bone during gestation
(Barltrop, 1969). The lead concentration in cord blood is 85-90% that
of maternal blood. Moore et al. (1982) reported a geometric mean level
of 0.67 µmol/litre (14 µg/dl) in 236 pregnant women and
0.58 µmol/litre (12 µg/dl) for lead in umbilical cord blood. The mean
concentration of lead in umbilical cord blood from a sample of over
11 000 women was 0.298 µmol/litre (6.6 ± 3.2 µg/dl) (Bellinger et al.,
1987).
6.3 Elimination and excretion
In both humans and experimental animals lead is eliminated from
the body in both urine and faeces. Any dietary (including waterborne)
lead not absorbed in the gastrointestinal tract is excreted in faeces.
Airborne lead that has been swallowed and not absorbed is also
eliminated in this manner. Blood lead not retained in the body is
excreted in urine or faeces, the latter by biliary excretion. Adults
ingesting daily 0.3 to 3.0 mg lead (as lead acetate) in drinking-water
for 16 to 208 weeks excreted more than 85% of the ingested lead, 90%
of the excreted lead being in the faeces. The amount excreted through
any route is affected by age and exposure characteristics and is
species dependent (US EPA, 1986a). This section reviews only studies
of lead excretion in humans; a discussion of the results of work on
experimental animals can be found in reviews by US EPA (1986a) and
ATSDR (1993).
The age dependency of lead excretion in humans has not been
studied extensively. However, the studies of Rabinowitz et al. (1977b)
and Chamberlain et al. (1978) in adults and Ziegler et al. (1978) in
infants can be used to assess this phenomenon. Data from Ziegler et
al. (1978) and Rabinowitz et al. (1977a) are given in Table 20.
Ziegler et al. (1978) calculated a total daily retention by infants of
40 µg, which is about twice the amount calculated by Alexander et al.
(1973).
Table 20. Comparison of daily lead intake and excretion in children
and adultsa
Parameter Childrenb Adultsc
Dietary intake (µg/kg) 10.76 3.63
Fraction absorbed 0.55d 0.15
Dietary lead absorbed (µg/kg) 5.92 0.54
Air lead absorbed (µg/kg) 0.20 0.21
Total absorbed lead (µg/kg) 6.12 0.75
Urinary lead excreted (µg/kg) 1.00 0.47
Endogenous faecal lead (µg/kg) 1.56 0.24
Total excreted lead (µg/kg) 2.56 0.71
Excreted/absorbed lead 0.42 0.92
Fraction of intake retained 0.33 0.01
a Adapted from: US EPA (1986a)
b From: Ziegler et al. (1978)
c From: Rabinowitz et al. (1977a)
d Corrected for calculated endogenous faecal lead
Under conditions of relatively constant exposure to low
concentrations of lead, approximately 140 to 215 µg/day, a steady
state condition evolves in which excretion approximates intake
(Rabinowitz et al., 1976). Under these conditions urinary lead
excretion is approximately 70% of absorbed lead. Chamberlain (1985)
reported that approximately 60% of absorbed lead is retained by the
body and 40% excreted.
Chamberlain (1983, 1985) also examined the relationship between
the level of exposure and rate of lead excretion. Renal clearance at
PbB levels of between 1.2 and 3.84 µmol/litre (25 and 80 µg/dl) was
found to increase at a rate approximating the increase in plasma lead.
Chamberlain (1985) estimated endogenous faecal lead loss into the
gastrointestinal tract following administration of lead-203 via the
inhalation and parenteral routes. These estimates suggested a
clearance of approximately 0.5% of administered dose per day when PbB
concentrations were under 1.2 µmol/litre (25 µg/dl). A special form of
excretion of endogenous lead is through breast milk. Studies of breast
milk indicate that lead concentrations correlate with maternal PbB
concentrations, most studies reporting that lead secreted from breast
milk varies in concentration between 10 and 30% of the maternal PbB
concentration (Ong et al., 1985).
6.4 Biological indices of lead exposure and body burden
6.4.1 Blood lead
The relationship between levels of exposure from various
environmental media and PbB has been discussed briefly in section
6.1.4.
Due to the ease of sampling and homogeneity of the sample, blood
has been the most widely used specimen to assess the human body burden
of lead. However, in view of the relatively short half-life for lead
in blood (28-36 days) (see section 6.2.2), PbB measurements in general
reflect only recent exposures. Also, in view of the kinetics of
distribution within the body (cycling between blood, bone and soft
tissues), differentiation of low-level chronic exposure from a short
high-level exposure is not possible on the basis of a single PbB
measurement. Interpretation of PbB levels over a wide range of values
must take account of the curvilinear relationship between total intake
of lead and PbB concentrations, as well as the proportion of lead in
plasma (Manton & Cook, 1984; see also section 8.1).
A number of cohort studies have collected serial PbB measurements
for children from birth up to 7 or 10 years. For children who have not
had major changes in their environment, there is good correlation
between consecutive PbB measurements (McMichael et al., 1988;
Bellinger et al., 1992; Baghurst et al., 1992; Dietrich et al.,
1993a,b). In these extended studies it has become apparent that for
most of the children a single PbB analysis at 6 years of age gives a
reasonable assessment of the life-time lead exposure status of the
child. However, random PbB levels in samples taken before 6 years of
age can markedly underestimate the peak exposure usually seen at 2
years of age (SAHC, 1993).
A new exposure index, namely "lifetime average blood lead" or
"lifetime average integrated blood lead" has been introduced in
studies using serial blood data. It has been clearly explained in the
proceedings of a recent Workshop and this explanation is quoted
directly here. This measure, which is a reflection of the area under
the "blood lead by age curve", has been occasionally misunderstood.
Due to the shape of the longitudinal blood lead profile, the peak
blood lead level, usually observed during the second year of life is
considerably greater than the average lifetime blood lead. For
example, a five-year-old child with a lifetime average blood lead of
0.96 µmol/litre (20 µg/dl) is likely to experience blood lead levels
above 1.92 µmol/litre (40 µg/dl) during the second year of life and
may spend 3 years of life, from 12 months to 48 months, above
0.96 µmol/litre (20 µg/dl). Due to the shape of the curve, with
declining PbB beginning about 24-39 months of age, the average
lifetime blood lead for a given child decreases with increasing age.
This does not mean that the index is inappropriate but rather that
developmental blood lead profiles change over time. For example, a
lifetime average blood lead of 0.72 µmol/litre (15 µg/dl) should not
be interpreted as being equivalent to a single blood lead
determination of 0.72 µmol/litre (15 µg/dl) obtained at a single point
in life (Dietrich et al., 1991).
6.4.2 Tooth lead
Unlike blood samples, teeth are composed of several anatomically
distinct pools which form over several years. Thus, in contrast to
blood, teeth are useful tissues for assessing long-term lead
accumulation from prenatal exposures to the time of shedding of the
tooth.
The accumulation of lead in teeth (PbT) has been used as a
measure of exposure of children to lead in several epidemiological
studies (Needleman et al., 1972; Winneke et al., 1982a; Rabinowitz et
al., 1989; Fergusson et al., 1989; Hansen et al., 1989). Studies
measuring PbT and PbB, however, are few. Preliminary data from
Australia (Baghurst et al., 1992) indicate a correlation of 0.8
between whole PbT and PbB prior to tooth exfoliation, but the only
report relating lead in circumpulpal dentine and longitudinal PbB (by
Greene et al., 1992) found a correlation of only 0.5. PbT
concentrations can vary as a function of location of the tooth within
the mouth, age, and whether total PbT or dentine PbT were reported.
Therefore, the age of the tooth in the mouth and its location, the
sample (whole tooth or dentine) analysed must be considered in any
estimation of a corresponding PbB in the published studies (SAHC,
1993).
Because of the complex structure and development of teeth,
concentrations of PbT will depend on the method of sampling and
analysis, tooth type, and resorption and tooth age at exfoliation. As
a result of varying procedures used by different investigators, there
may be substantial variation in absolute values and possibly the
biological meaning of PbT levels between different studies.
Different parts of a tooth sequester lead during different stages
of development. This is especially of concern in tooth sampling
because lead is not uniformly distributed within a tooth on a
submillimeter scale. A pilot study of teeth from children with
differing histories of exposure to lead and using high precision lead
isotopic methods has shown that analyses of slices of the incisal part
of deciduous teeth give the clearest indications of the in utero
environment, and the cervical sections, the exposure from birth to
exfoliation (Gulson & Wilson, 1994).
For more detailed discussions on the measurement of PbT the
reader is referred to Grandjean et al. (1984), Purchase & Fergusson
(1986) and Fergusson et al. (1989).
6.4.3 Bone lead
The human skeleton begins to accumulate lead during fetal
development and continues to abut 60 years of age (Pounds et al.,
1991). Interest in bone lead and its measurement in vivo stems from
concern that skeletal lead is not metabolically inert (see section
6.2.2), but can be mobilized by physiological and pathological states,
for example, during pregnancy and lactation (Silbergeld, 1991) and
osteoporosis (Silbergeld et al., 1988), with possible adverse effects
in other tissues, including the fetus, as well as the desire to
develop a meaningful measure of cumulative lead exposure as a tool in
public health protection. Section 8.13 includes a brief discussion of
the skeleton as a target organ for lead toxicity.
Procedures are available to analyse bone samples for lead levels
in humans not occupationally exposed to lead (Drasch et al., 1987;
Drasch & Ott, 1988). These studies have shown a decrease in levels of
lead in bones from autopsy specimens in Germany after removal of lead
from petrol. However, full utilization of bone lead stores as
dosimeters of lead exposure in a prospective sense requires the
utilization of technologies for in vivo measurement of lead in bone
such as X-ray fluorescence analysis (Chettle et al., 1991; Rosen et
al., 1991; Todd et al., 1992).
6.4.4 Lead in urine
Although urinary lead level has been used to measure current
exposure (Robinson, 1974), its use as a biomarker of lead exposure is
questionable in view of the relatively low and variable level of lead
excreted in the urine (Jensen, 1984; Ibels & Pollock, 1986). For this
reason, and in view of technical difficulties in analysing low levels
of lead in urine, urinary lead appears to be of limited use for
general screening. However, where an elevated body burden of lead has
been estimated using PbB or other indices of lead exposure, urinary
levels of lead after administration of the chelating agent
CaNa2-EDTA is considered an excellent measure of the potentially
toxic fraction of the total body burden of lead (CDC, 1985). The
chelatable lead excreted is assumed to represent lead removal from
soft tissues and blood, as well as sub-compartments of bone (Ibels &
Pollock, 1986; Mushak, 1989). In contrast, the urinary lead excretion
associated with lead mobilization provides what is considered the best
measure of the potentially toxic fraction of the total body burden
(see CDC, 1985; US EPA, 1986a). On the basis of various in vitro
experimental and epidemiological studies (CDC, 1985; US EPA, 1986a;
Mushak, 1989), chelatable lead is assumed to be a chemical sample of
both mobile body compartments (i.e. blood and soft tissues) as well as
of sub-compartments of bone.
6.4.5 Lead in hair
Hair lead has been used as an indicator of exposure in children
(Marlowe & Errera, 1982; Wilhelm et al., 1989). However, there are
severe limitations on its use from both the methodological as well as
the metabolic perspective. Systemic variations in lead level have been
reported according to hair colour, texture, location on the body and
growth phase (Wilhelm et al., 1989). Also, it is almost impossible to
avoid external contamination, and to date no validated methods are
available for cleaning. Methods which are sufficiently vigorous to
remove superficial lead also remove lead from the hair shaft.
7. EFFECTS ON LABORATORY ANIMALS AND IN VITRO TEST SYSTEMS
Lead can affect various organ systems depending upon the level
and duration of exposure. In all species studied, adverse effects on
the nervous system have been noted at PbB concentrations lower than
for other target organs. There is particular concern for the effects
of lead on fetal development. Since the appearance of Environmental
Health Criteria 3: Lead (IPCS, 1977), there have been very many
reports on the effects of lead on in vitro and animal models. No
attempt will be made in this chapter to summarize all such studies.
Rather, emphasis has been given to those studies that relate most
directly to the understanding of the effects of lead on humans and
thus provide additional scientific support for the use of such human
studies in the assessment of risk from lead exposures.
7.1 Biochemical effects
7.1.1 Haem synthesis and haematopoiesis
The principal clinical manifestation of the effect of lead on the
haematopoietic system is anaemia but this occurs only with high levels
of exposure that are rarely seen today. Lead affects the
haematopoietic system at several levels. These include effects on haem
and globin synthesis and on erythrocyte formation and function.
Most haem synthesis is directed toward formation of haemoglobin
and the rest is used in cellular oxidative metabolism. Lead inhibits
several steps in the biosynthesis of haem. More detail can be found in
IPCS (1977), US EPA (1986a) and section 8.1.1 of this monograph.
Lead acts on steps in the synthetic pathway both inside and
outside the mitochondrion. It inhibits certain enzymes (ALA
dehydratase, ferrochelatase, coproporphyrinogen oxidase) and increases
the activity of ALA synthase (ALAS) activity as a consequence of
feed-back regulation by haem (Moore et al., 1980). The activity of
ALAS is the rate-limiting step in the haem biosynthetic pathway.
Lead affects erythrocyte formation by impairment of globin and
haem synthesis. Globin synthesis is inhibited by lead in rat bone
marrow cells at concentrations as low as 1 µmol/litre (Dresner et al.,
1982). This is thought to be secondary to decreased protein synthesis
in erythroid cells as a consequence of lead-induced inhibition of haem
synthesis. Lead also decreases erythrocyte survival through its
inhibition of membrane bound Na-K ATPase (Rhagavan et al., 1981).
It has been shown in in vitro cultures of cells from liver,
bone marrow and the nervous system that ALAS activity may be increased
by addition of only 50 µmol lead/litre and ALAD activity is decreased
by 60% at a concentration of 0.5 µmol/litre (Kusell et al., 1978;
Dresner et al., 1982). Lead (5 µmol/litre) also inhibits (20%)
porphobilinogen deaminase in red blood cell haemolysates at
5 µmol/litre (Piper & Tephly, 1974). Fowler et al. (1980) have shown
that disruption of haem synthesis results in reduction of tissue haem
levels. Increased exposure to lead decreases the content and function
of haem-dependent enzymes of the P-450 mono-oxygenase system (Meredith
& Moore, 1979). More recently it has been shown that lead induces haem
oxygenase activity thereby increasing the degradation of haemoproteins
which may adversely affect a number of cell functions such as
respiration and energy production (Maines, 1992). It has been
suggested that delay in the accumulation of haemoproteins of the
respiratory chain in brain tissue during development may result in
decreased synthesis of haem enzymes in the brain (Bull, 1980; Moore et
al., 1987). Holtzman et al. (1981), on the other hand, found no effect
of lead on brain cytochromes in rat pups with impairment of growth due
to exposure to lead.
7.2 Nervous system effects
7.2.1 Higher order behavioural toxicity
Experimental studies using animal models have demonstrated that
lead impairs learning and memory functions at virtually all stages of
the life cycle. The most significant studies have focused on
behavioural and learning impairments in experimental animals with PbB
levels below 1.44 µmol/litre (30 µg/dl). Bushnell & Levin (1983) fed
post-weaning rats drinking-water containing 10 or 100 mg lead (as lead
acetate) per litre for up to 7 weeks. PbB was not determined but has
been estimated from comparisons with similar lead exposure models to
have been less than 0.96 µmol/litre (20 µg/dl) (Davis et al., 1990b).
Brain lead averaged about 0.05 µg/g wet weight. The rats exhibited
impaired learning ability when tested for their ability to choose
between alternate arms of a radial maze. Cory-Slechta et al. (1985)
demonstrated a significant learning impairment in rats given lead
acetate in drinking-water (25 mg/litre) from weaning. The observed
outcome was a significantly higher response rate in lead-exposed rats
working under a fixed interval schedule of food reinforcement. PbB
values of lead-treated rats in that study averaged
0.72-0.96 µmol/litre (15-20 µg/dl) and brain lead levels averaged
0.07 µg/g wet weight. Similar effects have been described in older
rats (16 months of age) at steady-state PbB levels of
0.62-0.86 µmol/litre (13-18 µg/dl) (Cory-Slechta & Pokora, 1991). More
recently, Cohn et al. (1993) demonstrated selective effects of lead on
learning processes, as distinct from non-specific effects such as
motivational levels, and sensory or motor impairment using a multiple
schedule of repeated learning and performance. Exposure to lead
acetate in drinking-water (50 mg/litre) produced a PbB of
1.2 µmol/litre (25 µg/dl).
Winneke et al. (1977) exposed rats to lead while in utero,
through mother's milk and directly in drinking-water. Their learning
ability was then tested by requiring the animals to discriminate
between stimuli of either different orientation (stripes as an easy
task) or different size (discs as a difficult task). Adult rats tested
between 90 and 170 days of age with PbB levels of less than
1.44 µmol/litre (< 30 µg/dl) were slower to learn the difficult task
of size discrimination (but not the easy discrimination problem) and
tended to repeat more errors than control subjects.
Altmann et al. (1993) demonstrated deficits of both active
avoidance learning (AAL) and in vitro hippocampal long-term
potentiation (LTP) in adult rats that had received pre-weaning or pre-
and post-weaning dietary lead exposure to achieve PbB levels of
approximately 0.72 µmol/litre (15 µg/dl) and brain lead concentrations
of 0.09-0.16 µg/g wet weight. Deficit in animals with only
post-weaning exposure and blood lead levels was about 0.77 µmol/litre
(16 µg/dl) but brain lead concentrations of only 0.09 µg/g was either
absent (in the case of LTP) or markedly reduced (in the case of AAL).
Similar types of effects have been noted in studies using
non-human primates. For example, Rice (1985) reported deficits of
discrimination reversal performance in monkeys dosed orally with 0, 50
or 100 µg lead/kg body weight per day for the first 200 days of life.
At the cessation of dosing, PbB concentrations were 0.144 µmol/litre
(3 µg/dl), 0.72 µmol/litre (15 µg/dl) and 1.2 µmol/litre (25 µg/dl),
respectively. Prior to behavioural testing (3 years of age), PbB
levels were 0.144 µmol/litre (3 µg/dl), 0.528 µmol/litre (11 µg/dl)
and 0.624 µmol/litre (13 µg/dl). Additional evidence for lead-induced
changes in learning in non-human primates is provided by the study of
Lilienthal et al. (1990), which showed a dose-related increase in the
percentage of errors in a learning set formation task (discrimination)
with PbB levels in the low exposure group (350 µg lead acetate/g diet)
averaging 1.68 µmol/litre (35 µg/dl). Also, an impairment of reversal
learning was found by Bushnell & Bowman (1979b) to persist in monkeys
up to their fifth year, at which time the PbB level in treated animals
averaged 0.24 µmol/litre (5 µg/dl) compared to 0.192 µmol/litre
(4 µg/dl) in controls. In the study by Bushnell & Bowman (1979a), the
period of lead exposure was limited to the first year of life and
resulted in PbB levels averaging as high as 3.12 µmol/litre
(65 µg/dl). No description was given of quality control procedures to
ensure accuracy of PbB determinations at the lower levels reported.
Several studies on experimental animals have shown perseverative
effects (the tendency to respond repetitively and inappropriately even
though environmental conditions have changed) which may underlie many
of the lead-induced changes in learning and other higher order
behavioural processes noted in such studies. This has been clearly
demonstrated in rodents by Cohn et al. (1993) in the context of
learning impairments. In the study by Bushnell & Levin (1983)
described above, the accuracy impairment derived from a tendency of
lead-exposed rats to re-enter a previously explored arm of the maze.
Similar perseverative tendencies have been described in non-human
primate studies, e.g., on delayed matching to sample (Rice, 1984a) and
delayed alteration tasks (Levin & Bowman, 1986; Rice & Karpinski,
1988).
Experimental animal studies also reveal the importance of task
complexity in detecting lead-induced changes in behaviour, both in
non-human primates (Levin & Bowman, 1983, 1986; Gilbert & Rice, 1987)
and rodents (Winneke et al., 1977, 1982b).
Persistence of lead-induced changes in some higher order
behavioural processes is also suggested by a number of experimental
animal studies. Several reports from the longitudinal studies of
non-human primates by Bowman and his colleagues (e.g., Bushnell &
Bowman, 1979a,b; Levin & Bowman, 1983, 1986, 1989) have demonstrated
persistent neurobehavioural deficits extending up to 8 years after the
termination of exposure and long after PbB levels had declined to the
control level. In rats, Cory-Slechta & Thompson (1979) showed that the
levels of lead exposure determine the persistence of lead effects,
while Cory-Slechta (1990a,b) revealed that apparently transient
effects of lead on behaviour could re-emerge with changes in the
reward contingencies of the environment. Munoz et al. (1986) found
deficits in both spatial and visual discrimination performance of rats
at several months of age resulting from pre-weaning exposure via the
dams. The same outcome was noted by Altmann et al. (1993) in a study
where deficits in adult rats were noted following pre-weaning
exposure.
7.2.2 Mechanisms of lead-induced behavioural toxicity
While the mechanisms underlying lead-induced behavioural toxicity
have yet to be adequately determined, the experimental animal
literature provides suggestive leads. The early experimental studies
of Pentschew (1965) and Pentschew & Garro (1966) demonstrated in the
rodent model that the pathogenesis of acute high-dose encephalopathy
was secondary to increased permeability of capillaries in the brain,
leading to leakage of fluid and red blood cells. These changes are
similar to those occurring in children with acute lead encephalopathy
characterized clinically by coma, convulsions and death, and identify
the brain microvasculature as the primary target for lead in the
central nervous system following high level exposure to lead.
Changes in microvascular morphology are not evident with low
level lead exposure, but it is the vulnerability of the blood-brain
barrier that permits exposure of the brain to lead. Exposure of the
developing fetus to lead results in higher uptake of lead in the brain
than from later exposures (Rossouw et al., 1987). The development of
resistance to acute encephalopathy in rat pups during the first few
days of life is thought by Holtzman et al. (1984) to be related to
maturation of the blood-brain barrier and possibly to the ability of
the older animals to sequester lead in protein complexes. Thus
neuropathological changes may relate more to the issue of exposure
than to mechanism of effect.
More recently, the focus of mechanistic studies has involved
biochemical and neurochemical changes. Biochemical changes in synaptic
transmission are likely to be related to problems in dendrite-nerve
organization and function (Goldstein, 1990). There is continuing
reorganization of dendrite and nerve terminal connections throughout
early months of neural development. A number of biochemical and
functional studies concerning possible mechanisms of lead effect
suggest lead may disrupt this process and its function through early
childhood (e.g., Cookman et al., 1987, 1988).
Other biochemical effects from lead exposure which may be the
basis of important neurobiological mechanisms of leadinduced
behavioural toxicity include changes in protein kinases, with
implications for neurotransmitter system disturbances. At least three
protein kinases present in nerve terminals involved in modulating the
release of neurotransmitters have been shown to be affected by lead.
However, the relevance of these findings to cognitive function has not
been established (Goldstein, 1990).
From both in vivo and in vitro studies, lead exposure has
been reported to have effects on virtually all neurotransmitter
systems (US EPA, 1986a). Depending on the stage of development, these
include dopaminergic, cholinergic, serotonergic, GABAnergic,
glutamatergic and opiate systems. Probably the most extensively
investigated system has been the dopaminergic system where a number of
changes at the biochemical and receptor level have been described. One
of the difficulties, however, is determining the relationship of the
reported changes at the biochemical/receptor level to changes in
behavioural function. McIntosh et al. (1989) noted lead-induced
changes in tetrahydrobiopterin metabolism which may be related to
changes in IQ scores (Blair et al., 1982; McIntosh et al., 1985). In
further support of a functional role for lead-induced changes in
dopaminergic systems, Cory-Slechta & Widzowski (1991) reported
alterations in both D1 and D2 dopaminergic sensitivity in rats with
PbB levels of 1.2-1.44 µmol/litre (25-30 µg/dl).
More recently, studies by Altmann et al. (1993) and Cohn &
Cory-Slechta (1993) suggest a role for changes in the N-methyl-D-
aspartate (NMDA) receptor complex in lead-induced behavioural
toxicity. The NMDA receptor complex has been implicated in the
learning and memory processes.
Effects of lead on mitochondrial energy metabolism may also be
important in the pathogenesis of neurological effects. Impairment of
respiration in brain mitochondria has been observed in in vitro
preparations (Holtzman et al., 1978) and in mitochondria isolated from
brains of lead-exposed rats (Gmerek et al., 1981).
7.2.2.1 Conclusions
Given their ability to establish causal relationships between
lead exposure and biological effects, experimental animal studies can
provide evidence supportive of human epidemiological findings. Many
experimental animal studies have been carried out to characterize the
nature of lead's effects on various target organ systems as well as to
establish the underlying mechanisms of effect. Furthermore, these
studies, by their very nature, are not confounded by such co-variates
of children's IQ as parental IQ, socioeconomic status and quality of
the home environment, or complicated by nutritional inadequacies of
the study population. Earlier experimental animal studies tended to
utilize relatively high lead exposure levels, but over the past ten
years, exposure levels and protocols have been more relevant to the
human situation.
There are, of course, some limitations of experimental animal
studies that must be considered. For example, there may be differences
in species sensitivity as well as differences in pharmacokinetic and
pharmacodynamic behaviour of lead in rodents, primates and humans.
Higher external exposure levels of lead are required to produce PbB
levels corresponding to levels experienced in human populations, and
higher PbBs are necessary to induce encephalopathy in rodents than are
associated with such effects in humans. Such differences in
sensitivity obviously reflect, at least in part, differences in
kinetics of lead across species. Despite these differences, it should
be noted that corresponding levels of PbB have been associated with
neuro-behavioural toxicity in both animal models (rodents as well as
non-human primates) and human populations. This comparability of
effect levels, despite the relative insensitivity of the animal
models, suggests that the levels of concern in humans might be even
lower, although quantitative extrapolation is difficult. Although one
cannot presume that the neurotoxic effects of lead in experimental
animals necessarily predict specific effects in humans, the
similarities in neurobehavioural end-points in humans and animals are
sufficient to conclude that neurobehavioural deficits in animals are
at least qualitatively predictive of effects in humans.
7.2.3 Sensory organ toxicity
Visual and auditory functions have been shown to be affected by
lead; other sensory modalities have largely been neglected. Bushnell
et al. (1977b) studied visual acuity in infant rhesus monkeys using
different degrees of illumination. Four control animals and three
animals each in a low and a high lead group were tested. The animals
were orally dosed between postnatal days 5 and 365. Although the
dosing regimen of this group was intended to produce blood lead levels
around 4.08 µmol/litre (85 µg/dl), peak levels of between 6.58 and
14.4 µmol/litre (137 and 300 µg/dl) occurred between the fifth and the
ninth weeks of dosing. Significant impairment of scotopic vision was
observed only in the animals of the high lead group.
Using flash-evoked visual potentials (VEP), Lilienthal et al.
(1986) studied visual function in 7- to 7´-year-old rhesus monkeys
pre- and postnatally exposed to lead acetate (0, 350 and 600 mg/kg) in
their diets. Seventeen animals were tested altogether: six controls,
and five or six in each of the exposed groups. PbB levels fluctuated
around 1.92 µmol/litre (40 µg/dl) in the group fed 350 mg/kg diet
during the seven postnatal years, whereas in the group fed 600 mg/kg
they had declined from the initial average of 5.28 µmol/litre
(110 µg/dl) at age 9 months to about 2.88 µmol/litre (60 µg/dl) at 7
years of age. PbB levels in the mothers of both these groups had been
between 1.15 and 1.78 µmol/litre (24 to 37 µg/dl) during pregnancy. A
dose-related decrease of amplitudes and a similar increase of
latencies for the main VEP component were found; these were, in most
instances, significant in both the high and low lead groups.
These findings are supported by the results of studies on rodents
by Fox et al. (1977). Prolonged VEP latencies were observed following
chronic developmental lead exposure. Lower VEP amplitudes have also
been observed in rats at PbB levels exceeding 1.44 µmol/litre
(30 µg/dl) (Winneke, 1979). in vitro studies suggest that rods are
more sensitive to the effects of lead than are cones (Fox & Sillman,
1979).
Information on the impairment of auditory function by lead comes
from electrophysiological studies using brainstem auditory evoked
potentials (BAEP) in rhesus monkeys (Lilienthal et al., 1990). PbB
levels in the various groups at the time of testing averaged
0.432 µmol/litre (9 µg/dl; controls), 1.92 µmol/litre (40 µg/dl; low
lead), and 2.688 µmol/litre (56 µg/dl; high lead). Of the four peaks
discernible in the monkey, BAEP peaks II and IV exhibited
significantly prolonged latencies, which were observed only in the
animals of the high lead group.
7.3 Renal system
The renal effects of lead in animal models occur as a result of
both acute and chronic exposures. Acute lead nephrotoxicity is
characterized by decreased reabsorption of small molecular weight
compounds by the renal tubule, particularly amino acids, glucose and
phosphate (Fanconi Syndrome).
Morphological changes of chronic lead exposure include
cytomegaly, development of nuclear inclusion bodies and
ultrastructural changes in mitochondria. The basis for the cytomegaly
is not understood but it is known that there is altered homoeostasis
of water and electrolytes and cellular swelling. Inclusion bodies are
lead-protein complexes composed of acidic non-histone proteins. As
much as 90% of lead in the kidney has been shown to be contained in
the inclusion bodies, suggesting that they provide a detoxification
function. The origin of the protein is not known but Egle & Shelton
(1986) have identified the most abundant component of isolated
inclusion bodies to be a constitutive protein of the adult central
nervous system primarily in the cerebral cortex. The lead in the
inclusion bodies is chelatable with EDTA (Goyer & Wilson, 1975).
Mitochondria isolated from kidneys of lead-intoxicated rats have
impaired respiration and oxidative phosphorylation capacity, (Goyer &
Rhyne, 1973).
Kholil-Manesh et al. (1992) studied the evaluations of renal
histological and functional changes in rats continuously dosed with
high dose lead. Glomerular filtration rate (GFR) was significantly
increased at 3 months, but significantly decreased at 12 months. Lead
inclusion bodies were noted throughout the duration of the study.
Tubular atrophy and intestinal fibrosis appeared at 6 months. The
brush border of proximal tubular cells was disrupted at 1 and 3
months, but recovered later.
With continued exposure to lead, acute nephropathy may progress
to chronic interstitial nephritis that does not have any unique or
distinguishing features. There is progressive increase in interstitial
fibrosis, dilatation of tubules and formation of microcysts with
hyperplasia of tubular epithelial cells. In the rat, inclusion bodies
are reduced in number and may be entirely absent in later stages of
the nephropathy. Glomerular sclerosis occurs with onset of proteinuria
and renal failure (Goyer & Rhyne, 1973).
The sequence of morphological changes observed in experimental
models is thought to be generally true for humans. Experimental
studies in rats suggest that there may be a threshold for lead renal
toxicity in the rat at PbB levels similar to those observed for
humans. In rats exposed to different doses of lead for up to 12 weeks,
a PbB level of 2.88 µmol/litre (60 µg/dl) appears to be the threshold
for proximal renal tubular cell injury by lead (Goyer et al., 1989).
This PbB level is equivalent to kidney lead levels of about 45 mg/kg
wet weight and is the level of lead at which excretion of renal
calcium increases and ultrastructural changes occur in proximal
tubular cell mitochondria. This finding is consistent with the
observation in humans by Buchet et al. (1980) and Gennart et al.
(1992) of a threshold value of 2.88 µmol/litre (60 µg/dl) of PbB which
leads to adverse renal effects by lead.
7.4 Cardiovascular system
Early studies concerning the production of hypertension in
experimental animals, summarized in Environmental Health Criteria 3:
Lead (IPCS, 1977), were conflicting. Among rats given 70 mg of lead
acetate per day orally, only a few survived 40 days and all were
hypertensive (Griffith & Landaver, 1944). On the other hand, other
studies did not reveal a blood pressure effect from high-level lead
exposure in rats (Padilla et al., 1969) or in dogs (Fouts & Page,
1942). More recently, several experimental studies have confirmed that
lead can produce hypertension, and evidence for several plausible
mechanisms has been provided. Rats, both normotensive and
spontaneously hypertensive, exposed to lead in water supplies for up
to a year, resulting in PbB levels of up to 0.6 µmol/litre
(12.5 µg/dl), exhibited ventricular tachycardia and ventricular
fibrillation (Evis et al., 1985). Subsequent studies (Evis et al.,
1987) showed that adrenaline had a significant arrhythmogenic effect
in hypertensive animals. It was concluded that chronic exposure to
lead, when combined with high blood pressure, slightly enhances the
susceptibility of the heart to arrhythmias induced by myocardial
ischaemia. Overviews of the experimental evidence related to
hypertension and lead exposure have been published by Victery (1988)
and US EPA (1986a, 1989). Taken as a whole, these studies demonstrate
that increase in blood pressure does occur secondary to renal failure
in rats with continuous high-level exposure to lead. More recent
studies on effects of chronic low-level exposure to lead on blood
pressure in rats have shown alterations in cardiovascular parameters
in the PbB range of 0.24-1.92 µmol/litre (5-40 µg/dl), and provide
some insights as to mechanisms involved in the pathogenesis of
lead-related cardiovascular effects.
Victery et al. (1982a) exposed rats to lead in utero by giving
dams drinking-water containing 0, 5, and 25 µg lead/ml and continued
this regimen to the pups for 5 to 6 months. Although no change in
blood pressure was noted at these levels of exposure, significant
changes in the renin-angiotensin system were reported in animals given
water containing 25 µg lead/ml with a PbB level of 0.864 µmol/litre
(18 µg/dl). In a similar study (Victery et al., 1982b), exposure to
100 µg lead/ml in drinking-water (but not 500 µg/ml) produced a
significant (17 mmHg) elevation in blood pressure beginning at 3´
months of age and continuing until 6 months of age. Chai & Webb (1988)
found an elevation of 15 to 20 mmHg in the systolic blood pressure of
rats given drinking-water containing 100 µg lead/ml. These authors
suggest that alterations in the cellular mechanisms that regulate
intracellular calcium concentration may enhance pressor responsiveness
to catacholamines. Boscolo & Carmignani (1988) found raised blood
pressure in rats with exposure to drinking-water containing 0, 30 or
60 µg lead/ml for 18 months. Cardiovascular responses to blood
pressure agonists indicated that lead exposure affects the
renin-angiotensin system and induces sympathetic hyperactivity by
acting on central and peripheral sympathetic junctions, increasing the
responsiveness to stimulation of alpha-2-adrenergic receptors, and by
increasing sensitivity to stimulation of cardiac and vascular
beta-adrenergic and dopaminergic receptors.
From the experimental evidence summarized by Victery (1988), it
appears that low-level exposure to lead produces an elevation in blood
pressure. The failure to demonstrate increased blood pressure in some
studies with high-level exposure to lead suggests that the effect of
lead on blood pressure may be biphasic, i.e. a consistent effect with
low-level exposure but inconsistent effects with high-level exposure.
7.5 Reproductive system
Experimental studies on the effects of lead on the reproductive
system most often concern toxicity to either the male or female but
have addressed results of exposure to both parents. Environmental
Health Criteria 3: Lead (IPCS, 1977) identified effects on
spermatogenesis in rats exposed to lead and showed that high maternal
exposure to lead in rats can reduce numbers and size of offspring.
There may also be paternally transmitted effects resulting in
reductions of litter size, weights of offspring and in survival rate.
Few studies into the effects of lead on male sexual function have
reported PbB levels. Ivanova-Chemishanska et al. (1980) reported
changes in levels of enzymatic activity and ATP in testicular
homogenates from rats given 0.0001 or 0.01% solutions of lead acetate
as drinking-water over a 4-month period. Chowdhury et al. (1984) found
testicular atrophy along with cellular degeneration in rats with PbB
levels over 3.36 µmol/litre (70 µg/dl), but not in rats with levels of
2.59 µmol/litre (54 µg/dl). Donovan et al. (1980) found that lead
inhibited androgen binding by cytosolic receptors in mouse prostate.
Testicular homogenates from 2- to 3-week-old male offspring (PbB
levels 0.30 µmol/litre, 6.3 µg/dl) of lead-exposed female rats showed
a decreased ability to metabolize progesterone (Wiebe et al., 1982).
In an in vitro study, Wiebe et al. (1983) found a 10 to 20% decrease
in FSH binding and in the production of cyclic AMP by Sertoli cells
isolated from prepubertal rats and cultured in the presence of lead
acetate (2.64 × 10-4 mol/litre). In addition, the activity of
cellular 3ß-hydroxysteroid dehydrogenase was decreased. It was shown
by Sokol et al. (1985) that there is a dose-related suppression of
serum testosterone levels and spermatogens in adult rats (100 day of
age) given a solution of 0.3% sodium acetate as drinking-water for up
to 60 days. PbB concentrations were between 1.44 and 2.40 µmol/litre
(30 and 50 µg/dl), depending on the length of treatment. Further
studies (Sokol, 1987) supported the hypothesis that lead disrupts the
hypothalamic control of pituitary hormone secretion. However, other
evidence indicates that lead may directly or indirectly affect
testicular enzymes or may act indirectly by a reduction in testicular
binding of FSH and production of cyclic AMP (US EPA, 1986a).
Dosing mature female rats with lead in order to produce PbB
concentrations of 1.44 µmol/litre (30 µg/dl) resulted in irregular
estrous cycles. At a PbB level of 2.54 µmol/litre (53 µg/dl), animals
developed follicular cysts and there was a reduction in the number of
corpora lutea (Hilderbrand et al., 1973). Grant et al. (1980) reported
delayed vaginal opening in rats whose mothers were given drinking-
water containing 25, 50 and 250 µg lead/ml. The vaginal opening delays
in the group given 25 µg lead/ml of drinking-water occurred in the
absence of any growth retardation or other developmental delays and
were associated with median PbB levels of 0.86-1.39 µmol/litre
(18-29 µg/dl). Studies on female monkeys have shown that pre- and/or
postnatal exposure to lead can affect pubertal progression and
hypothalamicpituitary-ovarian-uterine functions. Chronic exposure of
nulliparous female monkeys to lead (PbB levels of approximately
1.68 µmol/litre, 35 µg/dl) resulted in subclinical suppression of
circulating luteinizing and follicle stimulating hormone and estradiol
without producing overt effects on general health or menstruation
(Foster, 1992).
7.6 Effects on bone
There is growing interest in lead in bone for several reasons.
Bone is a store for lead accumulated from past exposure. It has a long
biological half-life but may be mobilized and contribute to blood lead
during pregnancy. With the development of X-ray fluorescence
techniques to measure lead in bone in vivo, there is a need to
improve our understanding of bone lead metabolism and of factors that
influence lead retention and release. In addition, lead may adversely
affect bone metabolism, particularly in post-menopausal women, and
contribute to the development of osteoporosis. There has only been
limited experimental study of these concerns to date, but there are
some relevant reports in the literature.
A summary of much of the currently available literature on the
potential toxicological implications of lead in bone during pregnancy
and lactation is contained in a review by Silbergeld (1991). Toward
the latter half of pregnancy in mice, there seems to be a preferential
transfer of lead across the placenta to the fetus (Danielson et al.,
1983). In rats exposed to lead for 150 days and then not exposed for
50 days prior to mating, Buchet et al. (1977) found that there was a
substantial mobilization of lead from mother to fetus. Keller &
Doherty (1980b) found using radiotracer lead (210Pb) in female mice
that there was also a major transfer of lead to the pup during
lactation. The concomitant decrease in maternal bone lead supports the
hypothesis that bone resorption of lead occurs during lactation.
It has been shown that lead may directly and indirectly affect
various aspects of bone metabolism (Pounds et al., 1991). Lead
inhibits the renal enzyme 1-hydroxylase, reducing plasma levels of
1,2-dihydroxychole-calciferol (activated vitamin D). Lead impairs the
Haversian remodelling system in beagle dogs chronically exposed to
lead (Anderson & Danylchuk, 1977).
7.7 Immunological effects
The effects of lead on the immune system are diverse but not well
documented. Lead reduces resistance and increases mortality of
experimental animals when they are infected by a broad range of
bacterial and viral agents (Koller, 1984). Lead impairs antibody
production in animals and generally decreases immunoglobin plaque-
forming cells (Koller & Roan, 1980).
7.8 Mutagenicity
Lead is thought to have genotoxic properties. However, lead-
induced gene mutations in cultures of mammalian cells have only been
observed at concentrations toxic to the cells. Studies for point
mutations in bacterial systems have also yielded negative results (US
EPA, 1986a). Zelikoff et al. (1988) found that both insoluble lead
sulfide and more soluble lead nitrate were mutagenic when added to
Chinese hamster V79 cells. A 6-fold increase in mutation frequency was
noted at a lead nitrate level of 500 µmol/litre medium. These authors
also found that lead acetate induced morphological transformation of
Syrian hamster cells. However, they concluded that these effects may
not have been the result of direct damage to DNA but may have occurred
via indirect mechanisms including disturbances in enzyme functions
important in DNA synthesis and/or repair.
Studies on the production of chromosome aberrations, sister-
chromatid exchanges and micronuclei by lead, whether in in vitro
cultures or in vivo, have given mixed results, and summaries are
available (US EPA, 1986a; IARC, 1987a,b; ATSDR, 1991).
7.9 Carcinogenicity
There have been several experimental studies in rats and mice in
which long-term administration of a lead compound in food or
drinking-water or parental administration has produced tumours of the
kidney (Van Esch & Kroes, 1969; Moore & Meredith, 1979). These and
other studies have been discussed in detail in IARC (1980) and
summarized in US EPA (1986a). In general, methodological problems,
including dose levels, number of animals, and doses, and lack of
toxicity monitoring, make many of the studies difficult to interpret
in a quantitative manner. One study (Azar et al., 1973) addressed some
of these problems and reported an increase in the numbers of renal
tumours in rats fed diets containing 500 mg lead/kg for 2 years. PbB
levels were 3.84 µmol/litre (80 µg/dl). In all studies renal
carcinogenicity occurred against a background of proximal tubular cell
hyperplasia, cytomegaly and cellular dysplasia in response to the high
doses of lead and long exposure times (Goyer, 1985). Renal
adenocarcinoma occurred in a high percentage of lead-exposed animals
and the incidence of tumours was related to the length and severity of
exposure (Mao & Molnar, 1967). Males appear to be more susceptible to
tumours than females. The maximum dose of lead in drinking-water, not
associated with any morphological or functional evidence of renal
toxicity in rats fed a diet containing adequate levels of trace
minerals, particularly calcium, is 200 mg lead/litre (Goyer et al.,
1970). No evidence of renal tumours at doses below this value has been
reported (Azar et al., 1973; US NCI, 1979).
Several hypotheses have been proposed for the mechanism of lead
carcinogenicity in experimental animals. These include mutagenicity,
cellular proliferation, nuclear protein (inclusion bodies), promoter
activity, activation of protein kinase C, and cystic hyperplasia
(Goyer, 1993). Lead is a weak mutagen in mammalian cell systems, but
is a strong mitogen. Exposure to a single intraperitoneal injection of
lead acetate in rats stimulates a 40-fold increase in cell
proliferation as measured by autoradiography, and this is further
increased by unilateral nephrectomy (Choie & Richter, 1972). DNA
synthesis in kidneys, as measured by 3H-thymidine incorporation, is
increased 15-fold and the mitotic index 45-fold following a single
intracardiac injection of lead acetate in mice (Choie & Richter,
1974). Cell proliferation and hyperplasia are seen in the liver of
rats given a single intravenous injection of lead nitrate (Columbano
et al., 1984). In both of these studies, increases in cell
proliferation occurred in the absence of cellular necrosis, suggesting
that this was a mitogenic rather than regenerative response.
Activation of protein kinase C and formation of nuclear inclusion
bodies or lead protein complexes are additional events that may
influence regulation of cell growth and development and play a role in
the carcinogenic response in experimental animals resulting from
administration of lead (Goyer, 1993). Cystic hyperplasia, a late
morphological manifestation of chronic lead nephropathy, is a risk
factor for renal cancer (Bernstein et al., 1987). Prior to adenoma
formation in animals treated with renal carcinogens, cystic
hyperplasia was reported (Dees et al., 1980; Goyer et al., 1981).
8. EFFECTS ON HUMANS
Despite the long recognition of lead poisoning, new clinical
cases continue to be the subject of published reports. Although
important, they affect a comparatively small proportion of the
population at risk from the potential effects of exposure to
environmental lead.
Over the last 10-15 years, particular attention has been directed
towards epidemiological studies designed to evaluate the possible
neurotoxic effects of lead on the developing child, especially delayed
or impaired neurobehavioural development and performance.
In the adult population, considerable attention has been directed
towards evaluating cardiovascular effects and the implication of lead
in hypertension.
New studies have contributed to our understanding of the
biochemical effects of lead, and may facilitate early recognition of
significant change and mitigation of potentially adverse outcomes.
The effects of lead have been studied widely in both the general
population and in those exposed occupationally. Since these effects
are the same in both settings, no distinction has generally been made
in the discussion. However, it is often important to distinguish
between adults and children because of different susceptibility.
8.1 Biochemical effects of lead
Lead is known to affect a number of enzymes and physiological
systems which result in a wide variety of changes in humans. While
those affecting the haematopoietic system are well known, there are
others which need to be considered in the risk assessment process and
are considered in this section.
In considering the effects of lead on biochemical systems, it is
appropriate to discuss the form of the lead in the various body
compartments.
Blood lead (PbB) is distributed between the plasma and the
erythrocyte. There is less than 1% in the plasma for PbB levels of up
to 4.8 µmol/litre (100 µg/dl) (Manton & Cook, 1984). The curvilinear
relationship of serum lead to blood lead is shown in Fig. 12. The data
show that the erythrocytes have a capacity to bind lead up to PbB
levels of about 2.4 µmol/litre (50 µg/dl). Above this level a fairly
rapid increase in the serum levels occurs.
It should be noted that the proportion of "free" (i.e. unbound)
lead in blood is important in relation to biological activity. Lead is
bound to haemoglobin in blood and has a greater affinity for fetal
than adult haemoglobin (Ong & Lee, 1980). It may, therefore, be
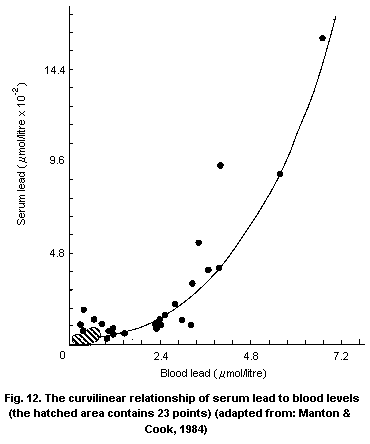 important to consider the proportion of fetal haemoglobin present in
blood samples from mothers and infants in assessing PbB concentrations
in relation to biological effects. Also, increased fetal haemoglobin
has been found in cases of human and experimental animal poisoning
(Albahary, 1972).
8.1.1 Haem synthesis
Lead is known to affect several enzymatic reactions critical in
haem synthesis, causing abnormal concentrations of haem precursors in
blood and urine. These effects of lead on haem synthesis are shown in
Fig. 13.
As shown in Fig. 13, lead inhibits the activity of three enzymes
of the biosynthetic pathway, 5-aminolaevulinate dehydratase (ALA-D),
coproporphyrinogen oxidase (COPRO-O) and ferrochelatase (FERRO-C).
This depletes haem synthesis and depresses the synthesis of the
initial and rate-limiting enzyme 5-aminolaevulinate (ALA) synthase. As
a consequence there is increased production and excretion of the
precursors ALA and coproporphyrin (COPRO) with increased circulatory
protoporphyrin (PROTO) usually bound to zinc. In the red cell,
diminished synthesis of monooxygenases (cytochromes P450) compromises
drug oxidation and lead is bound to haemoglobin.
8.1.1.1 Protoporphyrin levels
Lead interferes with the conversion of protoporphyrin to haem by
ferrochelatase. The protoporphyrin exists under these circumstances
primarily as zinc protoporphyrin, with a proportion remaining free
(Chisolm & Brown, 1979).
The relationship between protoporphyrin, either free or as the
zinc chelate, in blood and blood lead is one which could be
interpreted as showing a "threshold of effect" or as a continuum of
effect. The exact mathematical relationship is inevitably the choice
of the investigator since the uncertainties in the measurement of
blood lead and blood protoporphyrin would allow the fitting of the
data to either of these relationships. The threshold concept is
satisfactory to those that seek the "no-effect level" (Succop et al.,
1989), since it provides the start point of an analytical
identification process. The continuum of effect is, however, much more
plausible in a biological sense, since the idea of a level at which
"no effect" will occur is unlikely in a biological system.
The figures for the relationship between PbB and zinc
protoporphyrin are confused in any population by iron status (Marcus &
Schwartz, 1987), and it is thus unlikely that measurement of zinc
protoporphyrin, either alone or compared with haemoglobin status, will
provide an accurate estimation of PbB levels less than 0.96 µmol/litre
(20 µg/dl). In a study of the relationship between PbB levels and
erythrocyte protoporphyrin concentration for 2004 urban children
important to consider the proportion of fetal haemoglobin present in
blood samples from mothers and infants in assessing PbB concentrations
in relation to biological effects. Also, increased fetal haemoglobin
has been found in cases of human and experimental animal poisoning
(Albahary, 1972).
8.1.1 Haem synthesis
Lead is known to affect several enzymatic reactions critical in
haem synthesis, causing abnormal concentrations of haem precursors in
blood and urine. These effects of lead on haem synthesis are shown in
Fig. 13.
As shown in Fig. 13, lead inhibits the activity of three enzymes
of the biosynthetic pathway, 5-aminolaevulinate dehydratase (ALA-D),
coproporphyrinogen oxidase (COPRO-O) and ferrochelatase (FERRO-C).
This depletes haem synthesis and depresses the synthesis of the
initial and rate-limiting enzyme 5-aminolaevulinate (ALA) synthase. As
a consequence there is increased production and excretion of the
precursors ALA and coproporphyrin (COPRO) with increased circulatory
protoporphyrin (PROTO) usually bound to zinc. In the red cell,
diminished synthesis of monooxygenases (cytochromes P450) compromises
drug oxidation and lead is bound to haemoglobin.
8.1.1.1 Protoporphyrin levels
Lead interferes with the conversion of protoporphyrin to haem by
ferrochelatase. The protoporphyrin exists under these circumstances
primarily as zinc protoporphyrin, with a proportion remaining free
(Chisolm & Brown, 1979).
The relationship between protoporphyrin, either free or as the
zinc chelate, in blood and blood lead is one which could be
interpreted as showing a "threshold of effect" or as a continuum of
effect. The exact mathematical relationship is inevitably the choice
of the investigator since the uncertainties in the measurement of
blood lead and blood protoporphyrin would allow the fitting of the
data to either of these relationships. The threshold concept is
satisfactory to those that seek the "no-effect level" (Succop et al.,
1989), since it provides the start point of an analytical
identification process. The continuum of effect is, however, much more
plausible in a biological sense, since the idea of a level at which
"no effect" will occur is unlikely in a biological system.
The figures for the relationship between PbB and zinc
protoporphyrin are confused in any population by iron status (Marcus &
Schwartz, 1987), and it is thus unlikely that measurement of zinc
protoporphyrin, either alone or compared with haemoglobin status, will
provide an accurate estimation of PbB levels less than 0.96 µmol/litre
(20 µg/dl). In a study of the relationship between PbB levels and
erythrocyte protoporphyrin concentration for 2004 urban children
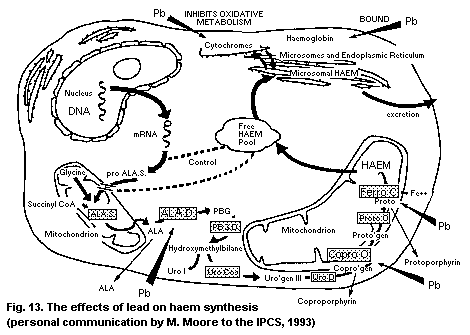 (Piomelli et al., 1982), it was concluded that a threshold is apparent
and occurs at a PbB level of between 0.72 and 0.86 µmol/litre (15 and
18 µg/dl). This is consistent with the data of Roels et al. (1976),
which show a clear discontinuity at around 1.2 µmol/litre (25 µg/dl).
Marcus & Schwartz (1987) re-analysed data from the NHANES II survey of
264 children. No positive correlation between PbB and zinc
protoporphyrin was noted below 0.96 µmol/litre (20 µg/dl).
Other reports which present relevant data are Roels et al.
(1976); Piomelli et al. (1982); Hammond et al. (1985); Rabinowitz et
al. (1986); Roels & Lauwerys (1987). In the presence of iron
deficiency the observed threshold is likely to be lower (Mahaffey &
Annest 1986; Marcus & Schwartz, 1987). At first sight it would seem
inappropriate to consider thresholds that are determined in the
presence of potential iron deficiency.
A study by Koren et al. (1990) of maternal and umbilical cord
lead and free erythrocyte porphyrin (FEP) levels for 95 mother-infant
pairs showed a correlation between maternal and cord PbB, with
maternal levels exceeding neonatal levels. Most cord PbB levels were
below 0.33 µmol/litre (7 µg/dl), and 11 were below the detection
limit. The cord blood FEP (0.86 µmol/litre) level was consistently
higher than maternal FEP (0.53 µmol/litre) but was not statistically
correlated. The elevated cord blood FEP values were attributable to
immature haem synthesis and high erythrocyte volume rather than the
presence of lead.
8.1.1.2 Coproporphyrin levels
One of the earliest observed effects of lead poisoning was a rise
in coproporphyrin excretion in the urine, due to inhibition of
coproporphyrinogen oxidase (Campbell et al., 1977). Although often
cited as a good measure of lead exposure it is clear that the
importance of the measure of current environmental levels of exposure
is small. This is because the excretion levels do not rise
significantly until the PbB excretion is greater than 1.92 µmol/litre
(40 µg/dl) (Meredith et al., 1978).
8.1.1.3 delta-Aminolaevulinic acid levels in urine and blood
Like protoporphyrin, circulating and excreted levels of ALA are
likely to be best described as a continuum of effect. Elevated levels
of this compound are of importance since neurological features of lead
exposure have been ascribed in part to increased circulating levels of
ALA (Moore et al., 1987). The rise in concentration during lead
exposure is a function first of decreased activity of ALA dehydratase
(ALAD), which is uniquely sensitive to lead toxicity, and subsequently
of increased activity of the initial and rate-limiting enzyme of haem
biosynthesis, ALA synthase (Meredith et al., 1978). It is
inappropriate to discuss haem biosynthesis control mechanisms here;
suffice it to say that blocking this pathway by lead lowers free haem
levels, which feedback to ALA synthase (Moore et al., 1987). The
special relationship between ALAD activity and lead exposure has been
best described as a negative exponential and has been used as a
measure of lead exposure in population surveys (Berlin & Schaller,
1974).
The immediate effect of the inhibition of ALAD will be an
increased level of ALA in the blood, which will then lead to increased
urinary excretion. The plasma levels of ALA are elevated in the
presence of higher lead levels. This has been seen in a number of
studies (Haeger-Aronsen, 1960; Meredith et al., 1978; O'Flaherty et
al., 1980).
Meredith et al. (1978) measured ALA metabolism in 48 male
lead-exposed workers (aged 22-56 years, PbB 4.2 ± 1.4 µmol/litre,
87 ± 29 µg/dl), who were compared with control subjects (28 male, 9
female, 18-52 years of age, PbB 1.3 ± 0.4 µmol/litre, 27 ± 3.3 µg/dl).
They found increasing levels of circulating ALA associated with PbB
that reached a plateau when the PbB level was in excess of
3 µmol/litre (62 µg/dl) and ALA exceeded 4 µmol/litre. At higher blood
ALA levels, urinary excretion of ALA increased exponentially,
consistent with decreased tubular reabsorption. The authors suggested
that there was a "critical" tissue lead concentration of around
2 µmol/litre (41 µg/dl). This study showed some continuity of the
correlation down to the lowest PbB value of the control group, namely
0.86 µmol/litre (18 µg/dl). However, these data were interpreted to
show that effects are only demonstrable above a PbB level of
1.44 µmol/litre (30 µg/dl).
Other studies show direct correlations between PbB level and
urinary ALA (Selander & Cramer, 1970; Lauwerys et al., 1974), although
these correlations are not seen as low as those in the study of
Meredith et al. (1978). The data of Selander & Cramer (1970) showed a
clear threshold effect at about 1.02 µmol/litre (40 µg/dl) in
occupational subjects.
Roels et al. (1976) reported data over a range of PbB levels from
0.24 to 1.92 µmol/litre (5 to 40 µg/dl) in children which showed
essentially no correlation with urinary ALA.
Data obtained from 39 men and 36 women in the general population
showed that increased urinary excretion of ALA occurred at PbB levels
of more than 1.68 µmol/litre (35 µg/dl) in women and more than
2.16 µmol/litre (45 µg/dl) in men (Roels & Lauwerys, 1987). The
sensitivity of the haem synthesis pathway to increased lead exposure
was in the order: children > women > men.
On balance, it would appear that lead has discernible effects on
the urine level of ALA at a PbB level of around 1.68 µmol/litre
(35 µg/dl).
8.1.1.4 delta-Aminolaevulinic acid dehydratase levels
delta-Aminolaevulinic acid dehydratase (ALAD) is commonly
measured for its activity in samples of haemolysed blood; the result
may or may not be corrected for haematocrit value. The measure of
enzyme activity will reflect the amount of enzyme present as well as
the effect of inhibition of the enzyme.
Studies in the general population have confirmed the correlation
and the apparent lack of a threshold for inhibition of ALAD in
different age groups and exposure categories (Roels et al., 1976;
Chisolm et al., 1985; Roels & Lauwerys, 1987). A negative linear
relationship between PbB and ALAD activity was found between mothers
and their newborn babies (cord blood); PbB levels ranged from 0.14 to
1.44 µmol/litre (3-30 µg/dl) (Roels et al., 1976). Roels & Lauwerys
(1987) reported a similar relationship in a population of 143 children
aged 10-13 years having PbB levels of 0.19-1.97 µmol/litre
(4.7-41 µg/dl).
In the study of Roels et al. (1976) a large number of children
were studied. Although the authors drew a regression line which
suggested that the effects continued to very low levels, the data
between 0.24-0.72 µmol/litre (5-15 µg/dl) were very scattered, and the
regression at low levels appeared to be rather speculative. It was
considered that the lead level above which an effect level is
demonstrable from these data was 0.48 µmol/litre (10 µg/dl).
8.1.1.5 delta-Aminolaevulinic acid synthase
Aminolaevulinic acid synthase (ALAS) levels are determined by a
feedback mechanism which is dependent on haem levels. There appear to
have been relatively few studies on serum ALAS, although some
consideration is given in a report by Meredith et al. (1978). However,
insufficient data were available to establish effect levels for lead
exposure.
8.1.1.6 Other effects of decreased haem synthesis
The potential impact of a reduction in the body pool of haem and
haem precursors is shown in Fig. 14. It is evident that the multiple
effects on haem metabolism from lead exposure can lead to adverse
effects in organs and systems other than the erythropoietic system.
8.1.2 Vitamin D
Formation of the most important vitamin D metabolite,
1,25-dihydroxyvitamin D, is by 1 alpha-hydroxylation of
25-hydroxyvitamin D in the kidney. This is mediated by
25-hydroxyvitamin D-1 alpla-hydroxylase, a cytochrome P450-dependent
enzyme in the mitochondria of the renal tubules. Serum concentrations
of 1,25-dihydroxyvitamin D are measured in children as an indicator of
the effects of lead on the enzyme system mediating the initial
hydroxylation. However, other factors such as dietary intake and the
physiological needs for calcium and phosphorus, and levels of
calciotropic hormones such as parathyroid hormone, can regulate the
production and circulating concentrations of 1,25-dihydroxy-vitamin D
(Rosen & Chesney, 1983).
Several studies have provided information on the effect of lead
on the circulating concentrations of 1,25-dihydroxyvitamin D. Rosen et
al. (1980) studied children with PbB levels in the range of
1.58-5.76 µmol/litre (33-120 µg/dl). While the most striking decreases
in serum 1,25-dihydroxyvitamin D occurred in children whose PbB level
was 2.97 µmol/litre (62 µg/dl), the effect was considered to be
evident in the range of 1.58-2.64 µmol/litre (33-55 µg/dl) when
compared to an age- and race-matched control group with PbB levels in
the range of 0.48-1.248 µmol/litre (10-26 µg/dl).
Mahaffey et al. (1982) measured serum 1,25-dihydroxyvitamin D in
177 subjects aged 1-16 years. PbB lead measurements were performed in
105 of these children and ranged from 0.576 to 5.76 µmol/litre (12 to
120 µg/dl). A curvilinear relationship between serum
1,25-dihydroxyvitamin D concentration and PbB was noted in children
aged 2 to 3 years. Details of the dietary intake of the subjects were
not available.
Koo et al. (1991) studied 105 children, aged 1 to 3 years and
with detailed lead exposure history from birth, to determine the
effect of chronic low to moderate lead exposure. The average lifetime
PbB concentration was 0.23-1.13 µmol/litre (4.8-23.6 µg/dl) and was
greater than 0.96 µmol/litre (> 20 µg/dl) in only three children.
With a range of concurrent PbB concentrations of 0.29-2.11 µmol/litre
(6-44 µg/dl). The children generally had adequate dietary intakes of
calcium, phosphorus and vitamin D. An effect of lead was found on
serum concentrations of calcium, phosphorus, 25-hydroxyvitamin D,
1,25-dihydroxyvitamin D and parathyroid hormone, and on bone mineral
content. In the presence of adequate nutritional status, lead exposure
at low levels (leading to a PbB level of less than 0.96 µmol/litre or
20 µg/dl) appears to have no demonstrable effect on circulating
concentrations of total and ionized calcium, magnesium, phosphorus,
calciotropic hormones including 1,25-dihydroxyvitamin D, parathyroid
hormone and calcitonin, or bone mineralization as indicated by single
photon absorptiometry. At higher levels there is a demonstrable effect
on 1,25-dihydroxyvitamin D.
8.1.3 Dihydrobiopterin reductase
This enzyme (DHBR) is part of the synthesis/salvage cycle which
controls the hydroxylation of tyrosine, tryptophan and phenylalanine.
It is thus central to the synthesis of the catecholamines. Lead has
been shown to inhibit the synthesis of tetrahydrobiopterin from
dihydrobiopterin by dihydrobiopterin reductase (Leeming & Blair, 1980;
(Piomelli et al., 1982), it was concluded that a threshold is apparent
and occurs at a PbB level of between 0.72 and 0.86 µmol/litre (15 and
18 µg/dl). This is consistent with the data of Roels et al. (1976),
which show a clear discontinuity at around 1.2 µmol/litre (25 µg/dl).
Marcus & Schwartz (1987) re-analysed data from the NHANES II survey of
264 children. No positive correlation between PbB and zinc
protoporphyrin was noted below 0.96 µmol/litre (20 µg/dl).
Other reports which present relevant data are Roels et al.
(1976); Piomelli et al. (1982); Hammond et al. (1985); Rabinowitz et
al. (1986); Roels & Lauwerys (1987). In the presence of iron
deficiency the observed threshold is likely to be lower (Mahaffey &
Annest 1986; Marcus & Schwartz, 1987). At first sight it would seem
inappropriate to consider thresholds that are determined in the
presence of potential iron deficiency.
A study by Koren et al. (1990) of maternal and umbilical cord
lead and free erythrocyte porphyrin (FEP) levels for 95 mother-infant
pairs showed a correlation between maternal and cord PbB, with
maternal levels exceeding neonatal levels. Most cord PbB levels were
below 0.33 µmol/litre (7 µg/dl), and 11 were below the detection
limit. The cord blood FEP (0.86 µmol/litre) level was consistently
higher than maternal FEP (0.53 µmol/litre) but was not statistically
correlated. The elevated cord blood FEP values were attributable to
immature haem synthesis and high erythrocyte volume rather than the
presence of lead.
8.1.1.2 Coproporphyrin levels
One of the earliest observed effects of lead poisoning was a rise
in coproporphyrin excretion in the urine, due to inhibition of
coproporphyrinogen oxidase (Campbell et al., 1977). Although often
cited as a good measure of lead exposure it is clear that the
importance of the measure of current environmental levels of exposure
is small. This is because the excretion levels do not rise
significantly until the PbB excretion is greater than 1.92 µmol/litre
(40 µg/dl) (Meredith et al., 1978).
8.1.1.3 delta-Aminolaevulinic acid levels in urine and blood
Like protoporphyrin, circulating and excreted levels of ALA are
likely to be best described as a continuum of effect. Elevated levels
of this compound are of importance since neurological features of lead
exposure have been ascribed in part to increased circulating levels of
ALA (Moore et al., 1987). The rise in concentration during lead
exposure is a function first of decreased activity of ALA dehydratase
(ALAD), which is uniquely sensitive to lead toxicity, and subsequently
of increased activity of the initial and rate-limiting enzyme of haem
biosynthesis, ALA synthase (Meredith et al., 1978). It is
inappropriate to discuss haem biosynthesis control mechanisms here;
suffice it to say that blocking this pathway by lead lowers free haem
levels, which feedback to ALA synthase (Moore et al., 1987). The
special relationship between ALAD activity and lead exposure has been
best described as a negative exponential and has been used as a
measure of lead exposure in population surveys (Berlin & Schaller,
1974).
The immediate effect of the inhibition of ALAD will be an
increased level of ALA in the blood, which will then lead to increased
urinary excretion. The plasma levels of ALA are elevated in the
presence of higher lead levels. This has been seen in a number of
studies (Haeger-Aronsen, 1960; Meredith et al., 1978; O'Flaherty et
al., 1980).
Meredith et al. (1978) measured ALA metabolism in 48 male
lead-exposed workers (aged 22-56 years, PbB 4.2 ± 1.4 µmol/litre,
87 ± 29 µg/dl), who were compared with control subjects (28 male, 9
female, 18-52 years of age, PbB 1.3 ± 0.4 µmol/litre, 27 ± 3.3 µg/dl).
They found increasing levels of circulating ALA associated with PbB
that reached a plateau when the PbB level was in excess of
3 µmol/litre (62 µg/dl) and ALA exceeded 4 µmol/litre. At higher blood
ALA levels, urinary excretion of ALA increased exponentially,
consistent with decreased tubular reabsorption. The authors suggested
that there was a "critical" tissue lead concentration of around
2 µmol/litre (41 µg/dl). This study showed some continuity of the
correlation down to the lowest PbB value of the control group, namely
0.86 µmol/litre (18 µg/dl). However, these data were interpreted to
show that effects are only demonstrable above a PbB level of
1.44 µmol/litre (30 µg/dl).
Other studies show direct correlations between PbB level and
urinary ALA (Selander & Cramer, 1970; Lauwerys et al., 1974), although
these correlations are not seen as low as those in the study of
Meredith et al. (1978). The data of Selander & Cramer (1970) showed a
clear threshold effect at about 1.02 µmol/litre (40 µg/dl) in
occupational subjects.
Roels et al. (1976) reported data over a range of PbB levels from
0.24 to 1.92 µmol/litre (5 to 40 µg/dl) in children which showed
essentially no correlation with urinary ALA.
Data obtained from 39 men and 36 women in the general population
showed that increased urinary excretion of ALA occurred at PbB levels
of more than 1.68 µmol/litre (35 µg/dl) in women and more than
2.16 µmol/litre (45 µg/dl) in men (Roels & Lauwerys, 1987). The
sensitivity of the haem synthesis pathway to increased lead exposure
was in the order: children > women > men.
On balance, it would appear that lead has discernible effects on
the urine level of ALA at a PbB level of around 1.68 µmol/litre
(35 µg/dl).
8.1.1.4 delta-Aminolaevulinic acid dehydratase levels
delta-Aminolaevulinic acid dehydratase (ALAD) is commonly
measured for its activity in samples of haemolysed blood; the result
may or may not be corrected for haematocrit value. The measure of
enzyme activity will reflect the amount of enzyme present as well as
the effect of inhibition of the enzyme.
Studies in the general population have confirmed the correlation
and the apparent lack of a threshold for inhibition of ALAD in
different age groups and exposure categories (Roels et al., 1976;
Chisolm et al., 1985; Roels & Lauwerys, 1987). A negative linear
relationship between PbB and ALAD activity was found between mothers
and their newborn babies (cord blood); PbB levels ranged from 0.14 to
1.44 µmol/litre (3-30 µg/dl) (Roels et al., 1976). Roels & Lauwerys
(1987) reported a similar relationship in a population of 143 children
aged 10-13 years having PbB levels of 0.19-1.97 µmol/litre
(4.7-41 µg/dl).
In the study of Roels et al. (1976) a large number of children
were studied. Although the authors drew a regression line which
suggested that the effects continued to very low levels, the data
between 0.24-0.72 µmol/litre (5-15 µg/dl) were very scattered, and the
regression at low levels appeared to be rather speculative. It was
considered that the lead level above which an effect level is
demonstrable from these data was 0.48 µmol/litre (10 µg/dl).
8.1.1.5 delta-Aminolaevulinic acid synthase
Aminolaevulinic acid synthase (ALAS) levels are determined by a
feedback mechanism which is dependent on haem levels. There appear to
have been relatively few studies on serum ALAS, although some
consideration is given in a report by Meredith et al. (1978). However,
insufficient data were available to establish effect levels for lead
exposure.
8.1.1.6 Other effects of decreased haem synthesis
The potential impact of a reduction in the body pool of haem and
haem precursors is shown in Fig. 14. It is evident that the multiple
effects on haem metabolism from lead exposure can lead to adverse
effects in organs and systems other than the erythropoietic system.
8.1.2 Vitamin D
Formation of the most important vitamin D metabolite,
1,25-dihydroxyvitamin D, is by 1 alpha-hydroxylation of
25-hydroxyvitamin D in the kidney. This is mediated by
25-hydroxyvitamin D-1 alpla-hydroxylase, a cytochrome P450-dependent
enzyme in the mitochondria of the renal tubules. Serum concentrations
of 1,25-dihydroxyvitamin D are measured in children as an indicator of
the effects of lead on the enzyme system mediating the initial
hydroxylation. However, other factors such as dietary intake and the
physiological needs for calcium and phosphorus, and levels of
calciotropic hormones such as parathyroid hormone, can regulate the
production and circulating concentrations of 1,25-dihydroxy-vitamin D
(Rosen & Chesney, 1983).
Several studies have provided information on the effect of lead
on the circulating concentrations of 1,25-dihydroxyvitamin D. Rosen et
al. (1980) studied children with PbB levels in the range of
1.58-5.76 µmol/litre (33-120 µg/dl). While the most striking decreases
in serum 1,25-dihydroxyvitamin D occurred in children whose PbB level
was 2.97 µmol/litre (62 µg/dl), the effect was considered to be
evident in the range of 1.58-2.64 µmol/litre (33-55 µg/dl) when
compared to an age- and race-matched control group with PbB levels in
the range of 0.48-1.248 µmol/litre (10-26 µg/dl).
Mahaffey et al. (1982) measured serum 1,25-dihydroxyvitamin D in
177 subjects aged 1-16 years. PbB lead measurements were performed in
105 of these children and ranged from 0.576 to 5.76 µmol/litre (12 to
120 µg/dl). A curvilinear relationship between serum
1,25-dihydroxyvitamin D concentration and PbB was noted in children
aged 2 to 3 years. Details of the dietary intake of the subjects were
not available.
Koo et al. (1991) studied 105 children, aged 1 to 3 years and
with detailed lead exposure history from birth, to determine the
effect of chronic low to moderate lead exposure. The average lifetime
PbB concentration was 0.23-1.13 µmol/litre (4.8-23.6 µg/dl) and was
greater than 0.96 µmol/litre (> 20 µg/dl) in only three children.
With a range of concurrent PbB concentrations of 0.29-2.11 µmol/litre
(6-44 µg/dl). The children generally had adequate dietary intakes of
calcium, phosphorus and vitamin D. An effect of lead was found on
serum concentrations of calcium, phosphorus, 25-hydroxyvitamin D,
1,25-dihydroxyvitamin D and parathyroid hormone, and on bone mineral
content. In the presence of adequate nutritional status, lead exposure
at low levels (leading to a PbB level of less than 0.96 µmol/litre or
20 µg/dl) appears to have no demonstrable effect on circulating
concentrations of total and ionized calcium, magnesium, phosphorus,
calciotropic hormones including 1,25-dihydroxyvitamin D, parathyroid
hormone and calcitonin, or bone mineralization as indicated by single
photon absorptiometry. At higher levels there is a demonstrable effect
on 1,25-dihydroxyvitamin D.
8.1.3 Dihydrobiopterin reductase
This enzyme (DHBR) is part of the synthesis/salvage cycle which
controls the hydroxylation of tyrosine, tryptophan and phenylalanine.
It is thus central to the synthesis of the catecholamines. Lead has
been shown to inhibit the synthesis of tetrahydrobiopterin from
dihydrobiopterin by dihydrobiopterin reductase (Leeming & Blair, 1980;
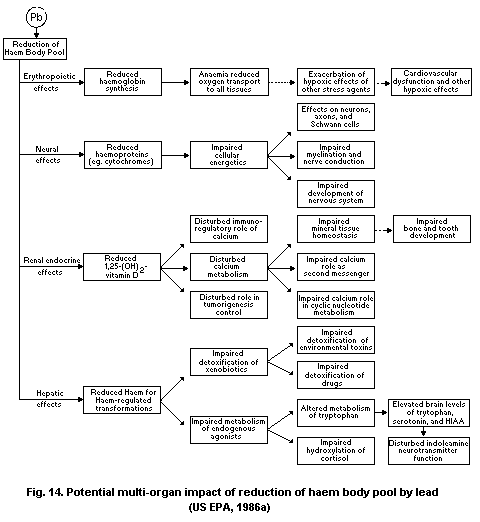 McIntosh et al., 1985) in rat brain. In humans the exact relationship
between PbB and plasma biopterins has not been properly defined.
8.1.4 Nicotinamide adenine dinucleotide synthetase
A recent study of nicotinamide adenine dinucleotide (NAD)
synthetase in erythrocytes indicates that this enzyme is sensitive to
inhibition by lead (and zinc) and is a sensitive indicator of lead
exposure. Zerez et al. (1990) found NAD synthetase activity reduced in
three subjects with elevated PbB levels in the range
1.63-3.46 µmol/litre (34-72 µg/dl).
8.1.5 Nutritionally affected groups
A body of evidence, summarized and discussed by Mahaffey (1985),
indicates that a high dietary calcium intake tends to decrease lead
absorption and retention in infants, young children and adults. Other
evidence suggests that in groups including children, with
self-selected diets, low dietary calcium intakes are associated with a
greater prevalence of elevated PbB levels (Mahaffey, 1985).
Some human data indicate that adults with low iron states have an
increased absorption of both iron and lead (Watson et al., 1980,
1986). In children, particularly those from low-income families, there
is often an association between iron deficiency and elevated PbB (Yip
& Dallman, 1984; Mahaffey, 1985). While this by itself does not
indicate a causal relationship, when considered in the context of data
on adults and animals, it suggests that iron deficiency in children
can result in increased lead absorption.
Alcoholics and people who consume excess alcohol may be at
increased risk of adverse effects from lead. In animal studies,
alcohol and lead synergistically inhibit ALAD, hepatic glutamic
oxaloacetic transaminase and glutamic pyruvic transaminase activities
(Flora & Tandon, 1987) supporting the hypothesis regarding risks to
humans.
8.2 Haematopoietic system
Lead-induced anaemia can be a direct consequence of inhibition of
haem biosynthesis; it is not necessarily associated with iron
deficiency. It may be associated with alterations of globin syntheses
(Albahary, 1972). More importantly, the synthesis of alpha- and
ß-globin chains may become asynchronous (White & Harvey, 1972).
8.2.1 Anaemia
Based on data published by Lilis et al. (1978), Baker et al.
(1979), Grandjean (1979), and earlier data, the threshold PbB level
for a decrease in haemoglobin has been estimated to be 2.40 µmol/litre
(50 µg/dl) for occupationally exposed adults (US EPA, 1986a).
Grandjean et al. (1989a) demonstrated the reduced ability of the
erythropoietic system to regenerate after blood withdrawal
(0.45 litre) in 25 lead-exposed battery workers (average PbB level
2.14 µmol/litre, 44.5 µg/dl) compared with 25 age-matched controls
(average PbB level 0.35 µmol/litre, 7 µg/dl). The haematological
parameters, except for erythropoietin were otherwise comparable for
the two groups. The effect was attributed to an effect on haem
synthesis and possible decreased erythrocyte survival in lead-exposed
workers. Lead has been found to depress serum levels of
erythropoietin, a hormone which regulates erythrocyte formation
(Graziano et al., 1991), which might also affect the reserve capacity
for erythropoiesis.
The PbB threshold for decreased haemoglobin levels in children is
estimated to be approximately 1.92 µmol/litre (40 µg/dl) (IPCS, 1977).
However, a cross-sectional epidemiological study of 579 children, aged
1-5 years in 1974, living in close proximity to a primary lead smelter
showed that adverse effects on haematocrit may occur at lower PbB
levels (Schwartz et al., 1990). Anaemia, defined as a haematocrit
below 35%, was not found at PbB levels of less than 0.92 µmol/litre
(20 µg/dl). There was a strong non-linear dose-response relationship
at higher PbB levels, which was influenced by age.
Kutbi et al. (1989) studied 200 boys aged 6-8 years. The mean PbB
was 0.33 µmol/litre (6.9 µg/dl; range 0.07-1.14 µmol/litre or
1.4-23.8 µg/dl). Subdividing the group at 0.72 µmol/litre (15 µg/dl),
they found a negative correlation between PbB level and all
haematological values for the "upper normal" (N = 7) compared with the
"normal" group (N = 193). The pattern of haematological parameters was
described as predictive of microcytic anaemia.
Anaemia has been commonly associated with the adverse effects of
occupational lead exposures. It is an effect that is easily diagnosed
clinically and is recognized as a marker of lead toxicity. Anaemia may
result from either a decrease in haemoglobin production or an increase
in the rate of destruction of erythrocytes. An analysis, made in 1974,
of the association between PbB levels and haematocrit in 579 children
(1-5 years of age) living near a primary lead smelter has recently
been presented (Schwartz et al., 1990). A haematocrit value of less
than 35% was used to indicate an adverse effect. It should be noted
that the effect of iron deficiency was not taken into account in
analysing the results. The study concluded that there was no adverse
effect of lead at PbB levels below 0.96 µmol/litre (20 µg/dl).
Furthermore, the risk of having a haematocrit value below 35% for
1-year-olds was 2% at PbB levels between 0.96 and 1.87 µmol/litre
(20 and 39 µg/dl). The degree of iron deficiency may account for a
substantial proportion of this 2%. In this study, the level at which
an effect of lead on the induction of anaemia was demonstrable was
about 1.92 µmol/litre (40 µg/dl).
8.2.2 Pyrimidine-5'-nucleotidase activity
Inhibition of erythrocyte pyrimidine-5'-nucleotidase leads to
accumulation of pyrimidine nucleotides, which has been associated with
induction of basophilic stippling.
Impaired function of erythrocyte pyrimidine-5'-nucleotidase
occurs in lead-exposed workers and as a genetically induced enzyme
deficiency. In in vitro systems the enzyme is inhibited by lead,
cadmium, mercury and zinc ions (and perhaps other metal ions) (Paglia
et al., 1975; Pagliuca et al., 1990; Ichiba & Tomokuni, 1990).
Graphs of PbB concentration against erythrocyte pyrimidine-
5'-nucleotidase activity in children have been reported for 21
children aged 2-5 years (Angle & McIntire, 1978) and for 42 aged 1-5
years (Angle et al., 1982) who participated in a preventative
programme because they had previously had PbB levels above
1.44 µmol/litre (30 µg/dl) or zinc protoporphyrin (ZPP) greater than
60 µg/dl. Both plots show considerable scatter although there was a
significant inverse logarithmic correlation. However, data relating to
the reproducibility, precision and accuracy of assessing inhibition of
erythrocyte pyrimidine-5'-nucleotidase were not presented.
This test may be capable of analytical refinement when assessment
of the sensitivity to lead (and other metals) exposure might more
reasonably be made. It does not yet appear likely to provide a routine
measure for low-lead exposure assessment.
8.2.3 Erythropoietin production
Graziano et al. (1991) found depressed serum erythropoeitin
levels in females at mid-pregnancy and at delivery associated
statistically with PbB level. Erythropoietin is a glycoprotein
produced in the renal proximal tubules which regulates both
steady-state and accelerated erythrocyte production. The study was not
adequate to define a dose-response relationship.
8.3 Nervous system
8.3.1 Historical perspective
Gross toxic effects of lead on the nervous system were reported
by ancient Greek physicians. A brief summary of such reports, and
those of Roman physicians and 18-19th century toxicologists, was
published by Kazantzis (1989). The syndrome was known as "painter's
colic", which included abdominal pain, constipation and paralysis,
symptoms now covered by the term "lead encephalopathy".
Lead colic was usually accompanied by effects on the nervous
system (lead palsy) in cases of acute and chronic poisoning, which
were often but not always fatal. Lead poisoning was well recognized as
an occupational hazard. However, it was also associated with
consumption of lead-contaminated water, wine, cider, rum from the West
Indies, and food prepared or stored in lead or lead-glazed utensils.
For example, Kazantzis (1989) described cases of lead poisoning in
girls resulting from consumption of drinking-water kept in lead-lined
cisterns, and also described the Devonshire colic attributed by Sir
George Baker in 1767 to the consumption of cider prepared in presses
made of lead or lead alloys.
The early toxicologists clearly recognized that the dose
(exposure) and form of lead was important in relation to absorption
and were able to demonstrate the presence of lead in tissues of fatal
cases of poisoning. There are many individually described, detailed
case studies in their writings.
8.3.2 Neurotoxic effects in adults
Despite the long recognition of lead poisoning, new clinical
cases continue to be the subject of published reports (Cueto et al.,
1989; Kocak et al., 1989; Zuckerman et al., 1989; Friedman &
Weinberger, 1990; Gupta et al., 1990; Mitchell-Heggs et al., 1990;
Nosal & Wilhelm, 1990; Sharma et al., 1990; Schneitzer et al., 1990;
Veerula & Noah, 1990). Although they are important, the numbers of
these poisoning cases are comparatively small compared with the
population at risk to the potential effects of exposure to lead from
environmental and dietary sources. Of particular recent concern have
been the possible neurotoxic effects on the developing child.
8.3.2.1 Central nervous system
In a study of 158 secondary lead smelter workers, Lilis et al.
(1977) found that 64% reported CNS symptoms and that there was early
occurrence of symptoms within 1 year of exposure. PbB concentrations
were elevated (about 3.36 µmol/litre or 70 µg/dl) at the time of
examination but many of the subjects had received chelation therapy.
Fischbein et al. (1979) noted CNS symptoms in 21 of 81 employees
of law enforcement agencies working in firing ranges. Symptoms
correlated with PbB levels and were present in three-quarters of
subjects having a PbB level > 2.4 µmol/litre (50 µg/dl).
Hanninen et al. (1979) reported that 49 lead-exposed workers
(including 21 male and 8 female storage battery and 16 railroad
engineering workshop employees) whose PbB level had never exceeded
3.36 µmol/litre (70 µg/dl) experienced more subjective symptoms than a
control group; excess symptoms were proportional to lead uptake.
Fischbein et al. (1980) studied 90 telephone cable splicers
having intermittent exposure to lead. They reported that 26 complained
of CNS symptoms (mean PbB level of 1.36 µmol/litre, 28.4 µg/dl, and
ZPP of 70.3 µg/dl) compared with controls (mean PbB level of
1.32 µmol/litre, 27.4 µg/dl, and ZPP of 49.3 µg/dl).
Awad El Karim et al. (1986) studied 92 lead acid battery workers
and compared them with 40 oil mill worker controls. They found CNS
symptoms in 50% of lead-exposed workers and measured PbB levels in 46
subjects working in different sections of the plant. More than 95% of
exposed workers had PbB levels above 1.92 µmol/litre (40 µg/dl),
whereas the mean PbB in the control group was 1.0 µmol/litre
(21 µg/dl).
Impairment of psychological and neuropsychological test
performance has been reported for lead-exposed workers (Hanninen et
al., 1979; Hogstedt et al., 1983; Parkinson et al., 1986; Araki et
al., 1986a,b; Huang et al., 1988a; Stollery et al., 1989, 1991).
The critical flicker fusion test has been reported to be a
sensitive indicator of CNS changes associated with exposure to lead.
Wooller & Melamed (1978) found significantly lower critical flicker
fusion in subjects with PbB levels above 2.88 µmol/litre (60 µg/dl)
than in those with levels below 0.96 µmol/litre (20 µg/dl). Williamson
& Teo (1986) found a significant decrease in mean flicker fusion in 59
lead-exposed workers (mean PbB > 2.3 µmol/litre or 48 µg/dl) compared
with matched controls. However, Gennart et al. (1992b) found no
difference between lead-exposed workers (mean PbB level
2.45 µmol/litre or 51 µg/dl, N = 98) and controls (mean PbB level
1.00 µmol/litre or 20.9 µg/dl, N = 85).
Stollery et al. (1989) studied the performance of 91 men,
occupationally exposed to inorganic lead, in a series of tests
designed to assess cognitive function. Subjects were grouped in "low"
(mean PbB 0.48 µmol/litre or 10 µg/dl, mean ZPP 8.7 µg/dl, N = 28),
"medium" (mean PbB 1.44 µmol/litre or 30.1 µg/dl, mean ZPP 22.4 µg/dl,
N = 27) and "high" (mean PbB 2.43 µmol/litre or 50.7 µg/dl, mean ZPP
58.8 µg/dl, N = 36) ranges of PbB concentrations. Results showed that
occupational exposure to lead impaired performance in a range of tests
(sensory motor reaction time, memory, attention, verbal reasoning,
spatial processing). Workers having PbB levels in excess of
1.92 µmol/litre (40 µg/dl) showed clear evidence of impairment on
tests of serial reaction time and category search. The authors
concluded that sensory motor, rather than cognitive, requirements of
many psychological tasks provide the most sensitive index of the early
effects of chronic low level lead exposure.
In a follow-up study (Stollery et al., 1991), 70 workers were
re-tested (three times within 8 months). Performance deficits in the
"high" (mean PbB 2.49 µmol/litre or 51.8 µg/dl, mean ZPP 77.4 µg/dl,
N = 22) group were not altered by practice or continued exposure. The
main deficit was a slowing of sensory motor reaction time coupled with
difficulties in remembering incidental information. There was little
evidence of impairment in workers having PbB levels less than
1.92 µmol/litre (40 µg/dl).
Finally, it appears that neuroelectrophysiological testing is a
sensitive and objective indicator of the CNS effects of lead. The
results of Araki et al. (1986b, 1987, 1992) using short-latency
somatosensory and visual evoked potentials and auditory event-related
potential (P300) indicate that subclinical electrophysiological
effects of lead occur not only in peripheral nerves but also in the
CNS.
8.3.2.2 Peripheral nervous system
Peripheral neuropathy is a common sign of chronic, high level
lead exposure, often manifesting as weakness in the upper or lower
limbs. At lower levels of lead exposure, nerve conduction velocity
(NCV) has provided a more sensitive indicator of peripheral nerve
dysfunction. More than 30 published studies have measured the
conduction velocity of electrically stimulated sensory and motor
nerves in workers exposed to lead. However, these studies have yielded
somewhat mixed results, with many showing a decrease in NCV in
relation to lead exposure (generally indexed as PbB concentration) and
a few showing no effect or occasionally even an increase in NCV
associated with lead exposure (Seppäläinen & Hernberg, 1980; Davis &
Svendsgaard, 1990).
Various reasons may underlie this lack of uniformity in NCV
findings. For example, studies may differ in methodological features,
in the characterization of lead exposure (e.g., single time-point
versus time-weighted average PbB levels) or in the handling of
confounding variables such as nerve temperature and age of subject
(Ehle, 1986). Other important factors accounting for some of the
apparent inconsistency in this area of research may be the possible
antagonistic effect of zinc to lead and differences in nerves selected
for measurement in different studies (e.g., slow versus fast fibres
(Araki et al., 1986c; Murata & Araki, 1991). A statistical
meta-analysis and critical review of 32 NCV studies by Davis &
Svendsgaard (1990) indicated that the median motor nerve shows effects
of lead more reliably than other nerves (e.g., median sensory or
ulnar).
Despite these complications, certain key well-conducted studies
provide compelling evidence of a causal relationship between lead
exposure and reduction in NCV. Araki et al. (1980) measured median
motor NCV before and after PbB levels of workers were lowered through
chelation therapy. Depending on the amount of reduction in PbB level
achieved by chelation and on a given worker's baseline NCV,
significantly improved NCV was measured in 7 out of 14 lead-exposed
workers. For all 14 workers the improvement in NCV was significantly
correlated with the decrease in PbB level (r = -0.573, P < 0.001).
In another key study, Seppäläinen et al. (1983) followed workers
prospectively from the beginning of their employment in a battery
plant, dividing them into two exposure categories (above and below the
median PbB level of 1.44 µmol/litre (30 µg/dl). Median motor NCV was
significantly reduced in the workers with blood lead levels above
1.44 µmol/litre (30 µg/dl) at 1-, 2-, and 4-year evaluation points,
despite attrition (and reduced statistical power) that resulted in
only five subjects per group after 4 years. Based on this well-
conducted prospective study, 1.44 µmol/litre (30 µg/dl) would appear
to be the lowest-observed-adverse-effect level for reduced NCV in
adults. Although another well-conducted prospective study (Spivey et
al., 1980) was unable to find an effect of lead on NCV at higher
exposure levels, NCV in the median nerve was not measured in that
study. Triebig et al. (1984) reported a dose-dependent decrease in NCV
at PbB levels above 3.36 µmol/litre, no dose-effect relationship being
detectable below this concentration.
8.3.2.3 Autonomic nervous system
There have been two reports of tests for autonomic nervous
function following lead exposure. Teruya et al. (1991) conducted a
cross-sectional survey of 172 male workers exposed to lead (mean PbB
level, 1.73 µmol/litre or 36 µg/dl, range 0.24-3.648 µmol/litre or
5-76 µg/dl), by measuring electrocardiographic R-R interval
variability (CVRR). Age-adjusted CVRR during deep breathing in
workers with PbB levels above 1.44 µmol/litre (> 30 µg/dl) was
significantly decreased as compared to those with PbB levels less than
0.96 µmol/litre (< 20 µg/dl). In addition, significant dose-response
and dose-effect relationships were observed between the PbB levels and
R-R interval variation during deep breathing among the workers with
PbB levels above 0.96 µmol/litre (> 20 µg/dl).
In a study reported by Murata & Araki (1991), the R-R interval
(CVRR) and two component coefficients of variation in the R-R
interval (C-CVRSA) were measured in 16 gun metal foundry workers
exposed to lead, zinc and copper (mean PbB level, 1.63 µmol/litre or
34 µg/dl, range 0.768-2.88 µmol/litre or 16-60 µg/dl). The CVRR and
component C-CVRSA (respiratory sinus arrhythmia) were significantly
lower in the workers than in age-matched controls, whereas the
C-CVMWSA (Mayer wave-related sinus arrhythmia) was unaffected. The
authors noted that zinc may have been antagonistic to the lead effects
in this study. Since the CVRR during deep breathing and the
component C-CVRSA of respiratory sinus arrhythmia reflect
parasympathetic activity, these two reports indicate potential
dysfunction of the autonomic nervous system (mainly, parasympathetic
activity) at average PbB levels of approximately 1.68 µmol/litre
(35 µg/dl).
8.3.3 Neurotoxic effects in children
The majority of the epidemiological research on the health
effects of lead has been focused on children because, in comparison
with adults, they are more vulnerable to lead in several respects
(Davis & Grant, 1992). For example, children typically engage in
hand-to-mouth activities (sucking fingers, putting food or other
objects in the mouth) that result in greater ingestion of lead than
adults normally experience. Also, because of their greater absorption
and retention of lead, the body burdens in children resulting from a
given external exposure level tend to be higher than adults.
Furthermore, the relatively greater exposures and body burdens of
children occur during sensitive periods of development. Finally, it
appears that children are generally more sensitive to the
toxicological effects of lead at a given internal exposure (PbB)
level. The lowest-observed-effect levels for various end-points (e.g.,
encephalopathy, anaemia, reduced haemoglobin, elevated EP, slowed NCV,
impaired neurobehavioural function) are lower in children than in
adults (US EPA, 1986a, 1990).
Section 8.3.3.2 describes findings from the main epidemiological
cross-sectional studies that have been published since 1979. This
constitutes a very substantial collection of research findings but has
certain interpretive limitations, particularly the lack of information
on exposure history.
More recent research effort has concentrated on prospective
epidemiological studies of birth cohorts repeatedly evaluated, mostly
up to the school age years. These prospective studies are described in
section 8.3.3.3.
Section 8.3.3.4 presents a quantitative overview of the
collective evidence from the prospective studies and discusses their
interpretation and the problems of inferring causality from such
observation data.
8.3.3.1 Historical perspective
While there have undoubtedly been cases of lead poisoning in
children as long as lead has been used by man, most of the attention
was previously given to cases of poisoning as a result of occupational
exposure in adults. In 1892 Gibson and colleagues in Australia
reported a case series of ten young children with lead colic. It was
not until twelve years later that peeling paint in the children's
homes was identified as the lead source (Gibson, 1904).
In 1943, Byers & Lord (1953) reported the results of an important
study, the first to suggest that there were neurological after-effects
of lead poisoning, in the absence of cerebral oedema and high
intracranial pressure, following acute lead encephalopathy. However,
in other cases it was felt that neurological manifestations
disappeared if the ingestion of lead was stopped (McKhann & Vogt,
1933). The case series of 20 school-age children had been hospitalized
for lead poisoning but without signs of acute lead encephalopathy, and
all were later discharged as cured. At follow-up, only one of the
children was making satisfactory progress at school. The children were
described as showing a variety of symptoms, including poor academic
achievement, intellectual deficits, sensory-motor deficits and
behaviour disturbance.
In the years following the findings of Byers & Lord (1953), a
number of case control studies were carried out that examined mental
retardation (Beattie et al., 1975) or hyperactivity (David et al.,
1972).
In addition, as a result of the growing concern about the dangers
of lead encephalopathy, studies were set up to investigate the
possibility that subclinical levels of lead (i.e. levels producing no
overt signs of lead encephalopathy) cause more subtle neurological
damage to children. Many of these studies investigated children
identified by screening clinics as having raised lead burdens, but
others were sited around smelters or lead works where children were
found to have elevated lead levels. The studies were important in
determining the areas of functioning which might be affected, and in
drawing attention to the methodological problems involved in carrying
out such studies. The main difference between the smelter studies or
studies around lead works, and clinical studies is that in the former
the primary source of environmental lead is known. In the hospital or
clinical studies it is likely that the ingestion of paint flakes, or
less often leaded putty, was in most cases the primary cause of the
excess lead exposure, although the exact reason for the increased lead
burden is rarely known.
Clinical and smelter studies were carried out in the United
Kingdom and USA and in general investigated children with PbB levels
above 1.92 µmol/litre (40 µg/dl), and compared their performance with
that of children with levels below this value.
Although some of the clinical and smelter studies found a
lead-associated deficit (Perino & Ernhart, 1974; Landrigan et al.,
1975), others did not (Lansdown et al., 1974). In general the studies
were methodologically flawed, with small samples lacking in
statistical power, biased ascertainment of subjects or controls, and
inadequate control for potentially confounding co-factors. The large
majority did not control for parental intelligence, which is the
variable most strongly associated with child IQ. These studies were,
however, important in suggesting an association between body lead
levels and performance, and also in demonstrating that there is an
association between social disadvantage and higher body lead levels.
8.3.4 Population-based cross-sectional studies on children
Most of the general population-based epidemiological
cross-sectional studies have, in addition to intelligence tests,
included extensive batteries of tests covering several other areas of
psychometric functioning, such as academic attainment, behaviour,
gross motor abilities and fine motor co-ordination, reaction time,
visuo-spatial skills and memory, and auditory memory. There are
differences in the tests used, and in the areas of functioning
investigated, and the results are less consistent and more difficult
to interpret than those relating to IQ tests. For that reason this
section will focus mainly on results relating to intelligence tests,
and results from other functional domains will be mentioned briefly
where appropriate.
There are many cross-sectional population studies using either
blood or teeth as the primary measure of lead body burden, and only
the more informative, in terms of providing information on risk
assessment, will be included. The majority of the studies described
used either a full or short form of the Wechsler Intelligence Scale
for Children - Revised (WISC-R or its translations) as the measure of
intelligence. Exceptions are the studies of Fulton et al. (1987),
which used the British Ability Scales (BAS), Harvey et al. (1988),
which used Wechsler Pre-school and Primary Scale of Intelligence
(WPPSI), and Schroeder et al. (1985) and Hawk et al. (1986), where the
Stanford-Binet Intelligence Scale was used.
8.3.4.1 Tooth lead studies
a) Needleman et al. (1979)
A total population of 3329 children aged 6-7 years, attending
schools in two working class towns near Boston, USA, were asked to
donate their shed deciduous teeth. Of these 70% (2335) children did
provide teeth. A 1-mm slice of tooth consisting primarily of dentine,
was split in half, and one portion was analysed by ASV. Children whose
initial tooth slice was in the highest 10th percentile (> 20 µg/g) or
the lowest 10th percentile (< 10 µg/g) were provisionally classified
into a high or low lead group. A total of 524 children were originally
classified into these lead groups, but 254 of these children were not
tested for a number of reasons, and the results for another 112 were
excluded from data analysis for medical reasons, or because a second
tooth analysis provided a discordant result. The results for 158
children (58 high lead children and 100 low lead children) were
presented. Thirty-nine non-lead variables were scaled and coded, and
those which were found to differ between the lead groups at the 10%
level were controlled as covariates. The covariates controlled were
father's socioeconomic status, mother's age at subject's birth, number
of pregnancies, mother's education and Peabody Picture Vocabulary
score. After this was done the children in the high lead group
performed significantly less well on the IQ test with a 4´ point
difference between the groups.
b) Winneke et al. (1983)
A total of 115 children aged 7-12 years living in the vicinity of
a lead/zinc smelter in Stolberg, Germany, who had provided at least
one incisor tooth, were investigated. Whole tooth lead measures ranged
from 2 to 32 µg/g, with a geometric mean of 6 µg/g. There was a
marginally significant negative association between lead level and
(short-form) verbal IQ after taking age, gender, perinatal risk
factors and social confounding factors into account. There was a
difference of 4.6 IQ points when children with tooth lead levels above
10 µg/g were compared with those with levels below 4 µg/g. Significant
negative associations were also observed between tooth lead and the
error score of Bender Gestalt shape copying test and performance on
the Vienna reaction time device.
c) Smith et al. (1983); Pocock et al. (1987)
The target population for the study by Smith et al. (1983) (also
known as the Institute of Child Health/University of Southampton
study) was 6875 children aged 6-7 years attending schools in one of
three areas in or near London. In all, 7407 teeth were donated by 4105
children. Whole tooth analysis of intact incisor teeth donated by
eligible children was carried out and children whose tooth lead level
(expressed on an ashed weight basis) was in the lowest percentile
(< 2.5 µg/g), approximately at the 50th percentile (5-5.5 µg/g) or
above the 90th percentile (> 8.0 µg/g) were invited to participate.
The sample was stratified by social grouping. Of the 432 children
selected, 403 participated in the study. Parental interviews were
carried out in the home, and information on a wide range of social and
familial variables was collected. There was a 5 point difference in IQ
scores between the high and low lead groups before controlling for any
covariates, but this was reduced to 2 points once covariates
associated with IQ score were controlled. The authors concluded that
social factors explain the differences in test performance to such a
degree that the small differences that remain may be due to other
social factors not measured. The study underlined the importance of
the social environment for the child's performance and recognized the
importance of the relationship between body lead burden and social
disadvantage.
Blood samples were taken from 93 children 3-6 months after
shedding a tooth and after psychometric testing was completed. The
range of PbB levels in this group was 0.336-1.296 µmol/litre
(7-27 µg/dl), except for one measurement of 2.064 µmol/litre
(43 µg/dl), and the mean was 0.614 µmol/litre (12.8 µg/dl). The
correlation of PbB and PbT was 0.5.
A reanalysis of this data by Pocock et al. (1989) employed
multiple regression techniques, using an "optimal" regression
strategy. The results were comparable to those previously reported.
Interactional analyses showed no evidence of any differential
association with lead in different social groups, but revealed a
significant interaction between PbT and child gender. Significant
negative association between PbT and IQ was found in boys but not in
girls.
d) Fergusson et al. (1988a,b,c)
This study was carried out in Christchurch, New Zealand, on a
sub-sample of a birth cohort of 1265 children born in 1977. Teeth were
collected from 1035 children, and a dentine "chip" was analysed to
provide a PbT estimate for 996 children. The mean dentine lead level
was 6 µg/g. Children from disadvantaged backgrounds had higher mean
lead levels than those from more advantaged backgrounds, and this
difference persisted when other social environmental factors (such as
the effects of housing, proximity to main roads, and childhood pica)
were taken into account. A WISC-R and reading test were administered
to children at ages 8 and 9 years, and results were given for 724
children at age 8 and 644 children at age 9. Small correlations
between IQ scores and dentine lead were found, but control for
confounding variables reduced these to statistical non-significance
for both ages (a correlation of -0.03 to -0.05). Parental IQ was
assessed. Control for pica as a test of reverse causality further
reduced the associations between outcomes and PbT.
e) Hansen et al. (1989); Lyngbye et al. (1990a,b); Grandjean et al.
(1991)
Children entering schools in the town of Aarhus, Denmark, were
asked to donate shed teeth. Of a potential sample of 2414 children,
1291 (54%) provided usable teeth. A sample of circumpulpal dentine was
obtained from each tooth and analysed by AAS. The mean lead level in
dentine was 10.7 µg/g, with a log normal distribution and a range from
0.40 to 168 µg/g. The correlation of whole tooth lead and circumpulpal
dentine measures were low. Average circumpulpal dentine levels were
about five times higher than average whole tooth measures. Subsequent
analysis of PbB from samples obtained two to three years later showed
an overall mean PbB level of 0.25 µmol/litre (5 µg/dl), with a
geometric mean of 0.28 µmol/litre (range 0.08-0.63 µmol per litre or
1.66-13.1 µg/dl) in children selected for the high lead group, and a
mean of 0.18 µmol/litre (range 0.08-0.70 µmol/litre or mean
3.74 µg/dl, range 1.66-14.56 µg/dl) in children selected for the low
lead group, which confirms this as a low exposure group. Children
whose level of lead in circumpulpar dentine was above 18.7 µg/g (8%,
N = 110) were selected for the high lead group, and they were matched
on gender and socioeconomic status (SES) with children whose
circumpulpar dentine levels were below 5 µg/g. A comprehensive list of
exclusions, including neurological and medical conditions, atypical
social situations, and those who did not wish to take part, reduced
the sample to 156 children for whom results were presented. Initial
analysis (by matched pair t-test) showed a difference of 6 IQ points
on full-scale WISC scores, with a difference of nearly 9 points in
verbal IQ scores. The association of IQ measures with circumpulpar
dentine lead levels was evident across different socioeconomic groups.
Confounders associated with circumpulpar dentine lead levels (such as
pregnancy variables, SES, mother's educational status and child's
gender) were entered into a stepwise regression model, and explained
only a small portion of the variance. Lead levels in the dentine
accounted for a significant (2.8%) of the total variance in full scale
IQ. Parental IQ was not controlled.
f) Rabinowitz et al. (1991)
From 764 eligible children (attending grades 1 to 3 in 7 selected
schools in Taiwan), 940 deciduous teeth were collected, the majority
being incisors. The average age of students was 6.7 years. Two of the
schools were near lead smelters. Lead levels were determined in 862
teeth from 692 children using the Boston method which analysed a
"dentine chip" (Rabinowitz et al., 1989). The mean value was 4.3 µg/g
of dentine. In all, 493 children were tested using the Ravens Coloured
Progressive Matrices Test (CPM) and the IQ equivalents were related to
tooth lead levels. Confounders were selected from a set of 40, based
on associations with lead and IQ. After correction for seven
confounding variables (social and perinatal factors), the highly
significant raw exposure-effect association (r = -0.32) was markedly
reduced for the full sample to statistical insignificance, although a
significant lead-CPM association remained for the subsample of girls
(b = -1.8; SE = 0.78; P < 0.02).
8.3.4.2 Blood lead studies
a) Winneke et al. (1985)
In a further study by this group carried out in the lead/zinc
smelter town of Nordenham, Germany, 114 out of 378 children (46%) for
whom cord PbB levels were available were tested at 6-7 years of age.
The range of cord PbB levels was 0.19-1.49 µmol/litre (4-31 µg/dl) and
for current PbB levels was 0.19-1.10 µmol/litre (4-23 µg/dl); the
means for both perinatal and current PbB levels were 0.39 µmol/litre
(8.2 µg/dl, SD = 1.6). After correction for confounders by means of a
stepwise multiple regression analysis, few significant associations
between PbB levels and performance were observed. There was a marked
influence of social background factors, but little influence on
short-form full IQ score (current PbB accounted for 0.3% additional
variance), and a borderline decrease in performance IQ (PbB accounting
for 2.4% of the variance). In general, the associations were rather
stronger with current PbB levels than with cord PbB measures. The
strongest associations were found with the error scores of the
difficult version of the Vienna reaction time task.
b) Harvey et al. (1984)
From 483 eligible children from the city of Birmingham, United
Kingdom, 187 out of 284 contacted were examined at 30 months of age.
The average PbB level from venous samples was 0.75 µmol/litre
(15.6 µg/dl, SD = 4). Cognitive ability was assessed by means of four
tests from the British Ability Scales (BAS) and three tests from the
Stanford-Binet Intelligence Scale. The raw correlation between PbB
level and IQ was -0.17 (P < 0.05). Subsequent multiple regression
analysis with a predetermined set of confounders and a sample size of
48 yielded an insignificant association between lead and IQ.
c) Harvey et al. (1988)
In a second study, 201 out of 337 eligible children, aged 5.5
years, from the inner city area of Birmingham, United Kingdom, were
studied. The mean PbB level was 0.614 µmol/litre (12.8 µg/dl, SD = 4).
No significant associations between PbB level and IQ (WPPSI) were
observed after correction for confounding factors. Apart from some
statistically significant associations between PbB level and reaction
time, none of the remaining tests showed a significant correlation
with PbB level after confounder-control.
d) Yule et al. (1981)
Children (N = 166), aged 6 to 12 years, living in the vicinity of
a leadworks in outer London were examined. PbB levels from venous
samples ranged from 0.336 to 1.58 µmol/litre (7 to 33 µg/dl, geometric
mean = 0.648 µmol/litre or 13.5 µg/dl). After controlling for social
class as the only confounder, significant negative associations were
found for IQ (WISC-R; full-scale- and verbal-, but not performance-
IQ). An average IQ difference of 7 points was found when comparing
children with PbB levels of 0.62 µmol/litre (13 µg/dl) or more with
those of 0.576 µmol/litre (12 µg/dl) or less. Significant inverse
associations were also found between PbB level and scores on tests of
attainment, i.e. reading and spelling but not mathematics.
A group of 9-year-old children from a middle class area of London
were studied. Subjects were divided into low PbB (N = 80 and PbB of
0.336-0.576 µmol/litre, 7-12 µg/dl) and high PbB (N = 82 and PbB of
0.624-1.15 µmol/litre, 13-24 µg/dl) groups for testing the association
between PbB level and IQ. No significant association between PbB level
and IQ was found before or after controlling for covariates (Lansdown
et al., 1986).
e) Schroeder et al. (1985)
One hundred and four children from a high risk population, aged
10 months to 6.5 years and with PbB levels ranging from 0.288 to
2.83 µmol/litre (6-59 µg/dl) were tested for intellectual development
using age-appropriate tests. After control for social confounding a
significant negative association was found between PbB level and IQ.
Fifty children of this cohort were reassessed 5 years later when all
PbB levels were below 1.44 µmol/litre (30 µg/dl). Neither initial nor
current PbB level was significantly related to later IQ.
f) Hawk et al. (1986)
In an effort to replicate the above-mentioned findings, 75
children from the same group, then aged 3 to 7 years, and having a
mean PbB level of 0.998 µmol/litre (20.8 µg/dl), range
0.29-2.256 µmol/litre (6-47 µg/dl), were examined using the
Stanford-Binet test. There were no significant interactions between
PbB level and factors including age, sex, maternal IQ, quality of the
care-giving environment (HOME), or socioeconomic status. There was a
statistically significant negative association between PbB level and
IQ, although the characteristics of the regression coefficient for
lead depended on which covariates were included in the model. For the
final regression model that best controlled for confounding with the
greatest precision, i.e. the most accurate and precise model,
containing lead, maternal IQ, HOME and gender, highly significant
inverse associations were found between both mean and maximum PbB
levels and IQ.
g) Fulton et al. (1987)
Five hundred and one children, aged 6-9 years, out of 1210
eligible children from 18 primary schools in central Edinburgh,
Scotland, were tested for an association between PbB level, IQ and
attainment. The sample comprised all children in the top quartile of
the PbB distribution, and a random subsample (approximately 1 in 3) of
the remainder. The geometric mean PbB level was 0.55 µmol/litre, range
0.16-1.63 µmol/litre (11.5 µg/dl, range 3.3-34.0 µg/dl). After
correction for 31 potential confounders a significant negative
association between PbB level and IQ (BAS) was found. There was a
dose-response relation with no evidence of a threshold. For purposes
of statistical analyses, children were placed into 10 groups of about
50 each on the basis of PbB level. The mean PbB level was
0.27 µmol/litre (5.6 µg/dl) in the lowest and 1.06 µmol/litre
(22.1 µg/dl) in the highest group; there was a difference of 5.8 IQ
points between the two groups. The size of the effects of lead was
small relative to that of the other factors (0.9% of a total of 45.5%
explained by the full model).
h) Hatzakis et al. (1989)
Five hundred and thirty-three children out of 1038 eligible from
four primary schools in the vicinity of the old lead-mining and
lead-smelting industrial complex of Lavrion in Greece were examined
neuropsychologically. The mean PbB level from venous samples was
1.14 µmol/litre, range 0.355-3.067 µmol/litre (23.7 µg/dl, range
7.4-63.9 µg/dl). In order to control for confounding factors, several
regression models were evaluated. Starting with a set of 24 potential
confounders, an optimal model containing 17 potentially confounding
variables was finally developed for testing the PbB/IQ association.
The continuous PbB variable was divided into five equally wide classes
from equal/below 0.715 µmol/litre (14.9 µg/dl, low) to equal/above
2.16 µmol/litre (45.0 µg/dl). After correction for confounders, a
highly significant negative association was found between PbB level
and IQ (WISC-R). A persistent decrease in IQ was only observed at PbB
concentrations above 1.2 µmol/litre (25 µg/dl). The adjusted
full-scale IQ difference between "high" and "low" PbB children was 9.1
points. Highly significant negative associations with PbB were also
found for the Bender Gestalt test and the Vienna Reaction Device,
without any indication of a threshold.
i) Silva et al. (1988)
Five hundred and seventy-nine socioeconomically advantaged
11-year-old children with an average PbB level of 0.53 µmol/litre
(11 µg/dl), range 0.19-2.4 µmol/litre (4-50 µg/dl) were tested in
Dunedin, New Zealand. Because the crude inverse association between
blood lead and IQ was not statistically significant, subsequent
multivariate analyses were not carried out.
j) European multicentre study
The results from a multicentre study in Europe were reported by
Winneke et al. (1990). The study linked eight institutions from eight
European countries; four of the individual study groups were from
areas near smelters and the others were more general populations. A
common study protocol with inherent quality assurance elements was
developed to achieve comparability between the individual studies. In
all, 1879 children, aged 6-11 years, were studied; however, for some
of the tests the sample size was reduced to only 971 children. The PbB
level ranged from 0.24 to 2.88 µmol/litre (5 to 60 µg/dl). Overall
statistical evaluation was done using a uniform predetermined
confounder model containing age, gender, occupational status of the
father, and mother's education. The inverse association between PbB
level and IQ (four subtests from the WISC-R) was of only borderline
statistical significance. An IQ decrement of about 3 points was
calculated for a PbB increase from 0.24 to 0.96 µmol/litre (5 to
20 µg/dl). The associations between the error scores on the Bender
Gestalt Test and the Vienna Reaction Device were statistically
significant and more consistent across study groups, although the
outcome-variance explained that lead never exceeded 0.8%. The data did
not allow for the identification of a threshold.
8.3.4.3 Follow-up studies
a) Bellinger et al. (1984)
A follow-up of some of the children investigated by Needleman et
al. (1979) was conducted when the children were approximately eleven
years old. Twenty-two of the original elevated lead group and 48
children from the low lead group were traced, as well as a group of 52
children who had not been tested previously, but whose lead levels in
a "dentine chip" (from teeth analysed at the time of the previous
study) fell into the mid-range between the high and low lead groups.
Children in the elevated lead group were significantly younger than
those in the other two groups. Information on IQ from group-
administered Otis Lennon Mental Ability tests, conducted 1 to 2 years
previously, was collected from school records for 15, 52 and 34
children from the elevated, mid-range and low groups, respectively.
Scores were inversely related to previously obtained lead measures,
with a 7-point difference in mean scores between the high and low lead
groups (t-ratio = 1.65, P = 0.1). No current measures of lead status
were available.
b) Winneke et al. (1989)
Out of 114 children first tested at age 6 (Winneke et al., 1985)
76 were retested at age 9. The range of PbB levels had been
0.187-1.09 µmol/litre, geometric mean = 0.39 (3.9-22.8 µg/dl,
geometric mean = 8.2) at age six and was 0.21-1.027 µmol/litre,
geometric mean 0.37 (4.4-21.4 µg/dl, geometric mean 7.8) at age 9
(r = 0.82). After correction for the same set of confounders used at
age 6 (age, gender, social background), most of the findings that had
been found to be significant at age 6, i.e. those related to reaction
performance (Vienna Reaction Device), remained virtually unchanged
three years later. The authors concluded that this observation cannot
necessarily be taken to indicate persistence, because internal
exposure had essentially remained unchanged over three years.
c) Needleman et al. (1990)
A follow-up assessment was made of 132 young adults first
investigated by Needleman et al. (1979) at the age of 6-7. Those
retested were not representative of the group of 270 tested 11 years
earlier, as they had slightly lower childhood dentine lead levels,
came from higher SES families, and were more likely to be female.
Current PbB level was assessed from 48 subjects and none was above
0.336 µmol/litre (7 µg/dl). A behavioural evaluation including a word
reading test, and a neurobehavioural assessment was administered to
each subject, and a self-report of delinquency was obtained. In
addition school records were reviewed. Multiple regressions indicated
that higher levels of dentine lead in childhood was associated with
lower reading scores, lower class rank, increased absenteeism, and
poorer performance on some of the neurological tests in young
adulthood. Comparing subjects with dentine lead levels greater than
20 µg/g with the lowest lead group, children in the high lead group
were much more likely to drop out of school (unadjusted odds ratio
4.6; adjusted odds ratio 7.4, CI 1.4-40.8) and to have a reading
disability (unadjusted odds ratio 3.9; adjusted odds ratio 5.8, CI
1.7-19.7). Adjustment for covariates made little impact on most of the
outcomes.
8.3.4.4 Conclusions and limitations of cross-sectional studies
A number of general issues are evident from looking at the
cross-sectional studies as a group. In most studies a negative
association between lead measures and IQ measures is found in
uncontrolled data. This difference is usually in the range of 4 to 6
IQ points and most marked in verbal IQ. Most studies also confirmed
the positive association between lead measures and indicators of
social disadvantage, whether this was indicated by SES, maternal
education, or other more detailed indicators of non-optimal
child-rearing environments, such as marital quality or maternal
depression. When these social and other confounding factors are
controlled, the effect has, in most cases, been to reduce the strength
of the association between lead measures and IQ, although it remains
in the same direction. When maternal intelligence has not been
controlled, the impact on the association between lead and IQ measures
of correction for covariates tends to be smaller. Where more detailed
social measures have been controlled, the impact on the magnitude of
the lead-IQ association has been greater. Associations have, in
general, been stronger with verbal IQ than with performance IQ.
The cross-sectional nature of these studies, and also the fact
that many used a single measure of current exposure, limited their
usefulness in answering questions relating to the natural history of
the association between lead exposure and outcomes, including whether
there were critical periods of exposure, and whether lead associated
deficits were persistent or reversible.
Findings from follow-up studies are difficult to interpret.
Needleman et al. (1990) interpreted their findings as indicating the
persistence of the effects of lead. Identifying persistence or
irreversibility in this context has many problems, due to the
stability of a number of other (non-lead) factors. In addition,
sampling biases, or failure to control for important covariates in the
initial or follow-up stages may lead to apparent persistence of an
effect. A further difficulty is that it is unusual to find a cohort
where exposure profiles permit easy assessment of persistence.
Neurobehavioural effects detected at age seven or later usually
persist in later follow-up studies conducted in other (non-lead) areas
of research, and for this reason it is more likely than not that
lead-associated deficits detected during childhood will be detectable
later. However, there are not enough data to conclude that this is the
case. Animal data, in which persistence of effects has been shown
after cessation of exposure, do provide support for the
irreversibility of lead-induced behavioural toxicity.
8.3.5 Prospective epidemiological studies on children
8.3.5.1 Common elements
The international prospective cohort studies shared a common
design of pre- or perinatal recruitment, with the principal measure of
prenatal exposure being the lead concentration in whole blood taken
antenatally or at birth. To facilitate later comparison of results,
meetings have been held at which common protocols (within the
constraints of local conditions) were agreed (Bornschein & Rabinowitz,
1985; Smith, 1989).
The protocols were similar in terms of the instruments of
neurobehavioural assessments, which were well standardized and
validated for the populations to which they were applied, and although
the frequency of assessment varied between studies, it was agreed that
all studies would make an assessment during infancy, in the later
pre-school period and, if possible, in the school age years.
All studies conducted several assessments of PbB concentration
but at varying frequency, and all studies participated in some form of
quality assurance/quality control (QA/QC) programme for the assessment
of lead exposure. QA/QC procedures were also used to establish
inter-examiner reliability (where more than one examiner was used) for
psychometric and covariate assessments. The importance of such
procedures was recently discussed (SAHC, 1993).
In assessing dose-effect relationships between lead exposure and
outcome variables, most studies used several indices of exposure,
including both measures of PbB concentration at particular ages and
average measures of exposure within the lifetimes of the study
subjects.
All studies used data analysis procedures which took simultaneous
account of covariates selected for statistical and substantive
considerations.
8.3.5.2 Study descriptions
a) Boston study
The study population was recruited from 11 837 infants born at
the Brigham and Women's Lying-In hospital in Boston between April 1979
and April 1981 (Bellinger et al., 1984). On the basis of cord PbB
levels of approximately 2500 children, the 90th, 50th and 10th
percentiles were identified and used as eligibility criteria for
enroling 249 children in three exposure groups: low (< 0.14 µmol/litre
or 3 µg/dl, N = 85), medium (0.288-0.336 µmol/litre or 6-7 µg/dl,
N = 88) and high (> 0.48 µmol/litre or 10 µg/dl, N = 76). No child
had a cord PbB level greater than 1.2 µmol/litre (25 µg/dl).
Exclusion criteria included birth complications or medical
conditions associated with developmental difficulties (e.g.,
gestational age < 34 weeks, Down's syndrome), a non-English speaking
family or residence in an area considered unsafe for home visitors.
The cohort consisted largely of children from intact middle and
upper-middle class families. Approximately 85% of the children were
white.
Capillary blood collected at ages 6, 12, 18 and 24 months was
analysed for lead by ASV. Venous blood collected at ages 57 months and
at 10 years was analysed for lead by AAS. In contrast to other
prospective cohorts, the mean PbB level did not rise postnatally and
indeed never exceeded 0.384 µmol/litre (8 µg/dl).
The principal psychometric tests included the Bayley Scale of
Infant Development at 6, 12, 18 and 24 months, the McCarthy Scales of
Children's Abilities at 57 months and the Wechsler Intelligence Scale
for Children - Revised and the Kaufman Test of Educational Achievement
at age 10 years. After age 2 years, the mean level of performance was
above the expected population mean (e.g., mean full scale IQ at age 10
years = 119) (Bellinger et al., 1992).
A variety of potential confounding factors was assessed including
maternal IQ, the quality of rearing environment (HOME scores at 6, 24,
57 and 120 months) and prenatal, perinatal and postnatal medical and
socio-demographic factors.
b) Cincinnati study
Women attending prenatal clinics in predesignated leadhazardous
residential areas of Cincinnati, Ohio, USA, were consecutively
recruited from 1979 to 1984 (Dietrich et al., 1987) Prenatal
exclusions were for maternal prenatal alcohol and drug abuse,
psychosis, diabetes, mental retardation; neonatal exclusions included
birth weight under 1.5 kg and gestational age (by physical
examination) below 35 weeks. Furthermore, recruited infants had to
have an Apgar score of 6 or more at 5 min postpartum and to have no
serious medical conditions. Of the mothers, 87% were single and 86%
were receiving public assistance. The final follow-up sample of 305
infants who attended their second clinic visit at 3 months of age was
85% African-American and 50% female.
Mean prenatal maternal PbB levels as assessed by ASV were
0.38 ± 0.177 µmol/litre (8.0 ± 3.7 µg/dl). Mean neonatal PbB levels
were also low at 0.221 ± 0.134 µmol/litre (4.6 ± 2.8 µg/dl). Following
the development of prewalking progression and normal hand-to-mouth
behaviours, PbB levels began to rise, peaking at around 21 months with
a mean of approximately 0.89 µmol/litre (18 ± 6 µg/dl). Approximately
35% of the sample had at least one PbB concentration equal to or
greater than 1.2 µmol/litre (25 µg/dl) sometime during the first 5
years of life, while 79% exceeded 0.72 µmol/litre (15 µg/dl) during
the same period (Dietrich et al., 1991, 1992). Virtually all children
(95%) exceeded 0.48 µmol/litre (10 µg/dl) during the first 5 years of
life. Postnatal PbB concentrations were assessed on a quarterly basis
beginning at 10 days postpartum. The vast majority of blood samples
were collected by venepuncture.
Major neurobehavioural assessments included the Bayley Scales of
Infant Development administered at 3, 6, 12 and 24 months of age. The
Kaufman Assessment Battery for Children was administered at 4 and 5
years. A comprehensive examination of gross and fine neuromotor
functions (the Bruininks-Oseretsky Scales) was administered at 6
years. Finally, performance on the Wechsler Intelligence Scale for
Children - Revised was assessed following school entry at 6.5 years.
Intellectually, this was a low functioning cohort with a mean
full-scale IQ at 6.5 years of 86.9 ± 11.3 points (Dietrich et al.,
1993a).
Covariates measured included assessments of obstetrical and
perinatal complications, birth anthropometrics, tobacco and alcohol
consumption, observational assessments of the quality of care-taking
in the home at 6, 12, 24 and 36 months, social class, iron status and
various aspects of child health likely to affect neurobehavioural
performance (e.g., otitis media, sensory deficits, allergies, seizure
disorders). Maternal IQ was assessed postnatally. All medical and
psychometric testing was conducted at a single welfare clinic located
in the heart of the recruitment area.
c) Cleveland
Five hundred and forty-three infants delivered to women at a
large inner city general hospital between February 1981 and March 1982
were considered for inclusion in this study of the joint or
independent effects of fetal alcohol exposure and lead on child
development (Ernhart et al., 1985). Due to pre-term birth or illness,
16% were excluded from further study. Also excluded were mothers
reporting the use of narcotics and those with identifiable
psychological disorders. The sample was predominantly white (65%) and
of lower SES. Approximately 50% of mothers were alcoholics.
PbB concentrations were assessed by AAS (208 mothers at delivery
and in 178 cords resulting in 142 mother-infant pairs with complete
prenatal lead exposure data). Postnatally, blood samples were analysed
for approximately half the birth cohort at ages 6 months, two years
and three years of age. At least one perinatal or postnatal PbB
concentration was available for each of 285 children with
developmental data past the neonatal period (Ernhart et al., 1987,
1989a,b).
Mean maternal PbB concentrations (N = 185) were low
(0.31 ± 0.086 µmol/litre or 6.5 ± 1.8 µg/dl) and mean cord PbB
concentrations (N = 162) were also low (0.29 ± 0.10 µmol/litre or
6.0 ± 2.1 µg/dl). Postnatally, mean PbB concentrations (N = 151) were
0.48 ± 0.16 µmol/litre (10.0 ± 3.3 µg/dl) at 6 months,
0.80 ± 0.28 µmol/litre (16.7 ± 5.9 µg/dl) at 2 years (N = 165) and
0.80 ± 5.9 µmol/litre (16.7 ± 5.9 µg/dl) at 3 years. The lowest and
highest PbB levels recorded postnatally were 0.24 and 2.016 µmol/litre
(5 and 42 µg/dl), respectively.
Neurobehavioural assessment included selected sub-scales of the
Brazelton Neonatal Behavioural Assessment Scales and Graham Rosenblith
Behavioural Examination of the Neonate at > 24 h postpartum, the Kent
Infant Development scale at 6 months, the Bayley Scales of Infant
Development at 6, 12 and 24 months, the Stanford-Binet Intelligence
Scale at 3 years, and finally the Wechsler Preschool and Primary Scale
of Intelligence at approximately 5 years of age. All assessments
beyond the neonatal period were conducted in subjects' homes.
Intellectual attainment in this high risk, socioeconomically
disadvantaged cohort was typically low as shown by a mean full-scale
IQ at approximately 5 years of 87.5 ± 16.6 points (Ernhart et al.,
1989a).
In addition to the usual assessments of obstetrical/perinatal
complications and neonatal status, other covariates measured included
the Michigan Alcoholism Screening Test, the Peabody Picture Vocabulary
Test as an assessment of maternal intelligence, and the Authoritarian
Family Ideology Scale. Women were also questioned as to their
consumption of alcohol and tobacco during pregnancy. Observational
assessments of care-taking quality in the home were conducted at 1, 2,
3 and 4 years.
d) Glasgow study
The study group for CNS outcomes consisted of 151 subjects drawn
from an initial sample of 885 families exposed to various levels of
dietary lead, principally due to a plumbosolvent water supply (Moore
et al., 1989). All subjects were born in the United Kingdom and spoke
English as a first language. The sample was divided into three groups
of approximately equal numbers (matched for social class) based on
maternal prenatal PbB levels: high (> 1.44 µmol/litre or 30 µg/dl),
medium (0.72-1.2 µmol/litre or 15-25 µg/dl) and low
(< 0.48 µmol/litre or 10 µg/dl). The SES of the sample ranged from
the chronically unemployed to the professional classes.
PbB levels were assessed postnatally at 1 and 2 years of age and
found to be 0.734 and 0.777 µmol/litre (15.3 and 16.2 µg/dl),
respectively.
Measurements of neurobehavioural outcome where the Bayley Scales
of Infant Development were used were administered at 1 and 2 years of
age.
Covariates considered in the data analysis included a measure of
obstetrical complications, birth weight, birth order, SES of the
father in postnatal follow-up years 1 and 2, and observational
assessments of care-taking quality in the first and second years of
follow-up.
e) Kosovo Study
Five groups of infants from two communities in Kosovo,
Yugoslavia, were studied (Graziano et al., 1990; Wasserman et al.,
1992). Mitrovica was the site of a lead smelter, refinery and battery
plant, and Pristina an area of minimal lead exposure. Three groups
were recruited from Mitrovica on the basis of cord PbB concentration:
(1) < 0.72 µmol/litre (< 15 µg/dl, N = 78); (2) 0.72-0.96 µmol/litre
(15-20 µg/dl, N = 99); (3) > 0.96 µmol per litre (> 20 µg/dl,
N = 217). Two groups were recruited from Pristina: (4) a group with
cord PbB levels < 0.72 µmol/litre (< 15 µg/dl) and (5) a group
matched to children in group (3) in terms of the distribution of
maternal and paternal education. Grouping was not considered in the
statistical analysis. The total cohort (N = 541) was largely Albanian
and Serbian. Infants with CNS defects, chromosomal anomalies, distant
residence and twin pairs were excluded.
PbB level and various indices of iron status (EP, Hb, serum
ferritin) were measured in venous blood collected in midpregnancy,
delivery and at postnatal ages 6, 12, 18 and 24 months. Mean PbB level
amongst children in Mitrovica exceeded 0.96 µmol/litre (20 µg/dl) at
all ages, reaching 1.68 µmol/litre (35 µg/dl) at 24 months. Amongst
children in Pristina, the mean PbB level was below 0.48 µmol/litre
(10 µg/dl) at all ages.
The Mental Scale of the Bayley Scales of Infant Development was
administered at ages 6, 12, 18 and 24 months, and the Motor Scale at 6
and 12 months. At 24 months, the mean Mental Development Index in the
cohort (n = 392) was 105.2 (SD = 18.1).
Anthropometric measurements and assessments of child and family
health, diet, and demographic status were carried out at each age. An
adaptation of the HOME Scale was administered at ages 3 and 4 years
and maternal IQ was assessed using the Raven's Standard Progressive
Matrices.
f) Port Pirie study
Port Pirie (population 16 000 in 1979) is an industrial town 200
km north-west of Adelaide, South Australia, with a large and
long-standing smelting facility (Wigg et al., 1988). Over the period
1979-1982, a cohort of 723 children from Port Pirie and surrounding
rural areas was recruited into the study. The cohort represented a 90%
sample of all children born in the area during this period.
Lifetime lead exposures were estimated from venous PbB
concentrations obtained antenatally (at 14-20 weeks of gestation, and
32 weeks), at delivery (maternal and cord blood), and from capillary
samples taken postnatally at ages 6, 15, and 24 months, and annually
thereafter. Lead concentrations in all samples were determined by AAS.
At the time that each blood sample was collected, the nurse
interviewer also conducted structured interviews to obtain information
on a range of demographic, familial, behavioural, medical and social
environmental factors. HOME scores were assessed when the child was 3
and 5 years of age. Maternal IQ was also assessed. The Bayley Scales
of Infant Development was administered at 2 years, the McCarthy Scales
of Children's Abilities at 4 years and the Wechsler Intelligence Scale
for Children - Revised version (WISC-R) at 7 years.
g) Sydney study
A total of 318 children was recruited from among infants born at
three maternal hospitals between April 1982 and March 1983 (Cooney et
al., 1989a). Exclusion criteria included prematurity, low birth
weight, severe medical problem, single or non-English speaking mother
or maternal drug or alcohol problems. All children were white and
generally represent a middle-class population.
PbB concentration was measured in maternal venous blood during
hospitalisation (geometric mean = 0.388 µmol/litre or 8.1 µg/dl,
geometric SD = 0.067 µmol/litre or 1.4 µg/dl). Additional blood
samples were collected at 6-month intervals to age 4 years and at ages
5 and 7 years. Up to 24 months of age, approximately 50% of the blood
samples were capillary, while almost all samples collected
subsequently were venous. Geometric mean PbB levels between 6 months
and 4 years were in the range of 0.48-0.72 µmol/litre (10-15 µg/dl),
falling to 0.39 and 0.37 µmol/litre (8.3 and 7.7 µg/dl) at ages 5 and
7, respectively.
The Bayley Scales of Infant Development were administered at ages
6, 12 and 24 months, the McCarthy Scales of Children's Abilities at
ages 3 (n = 215) and 5 (n = 200), and the Wechsler Intelligence Scale
for Children - Revised at 7 years (n = 175). All assessments, at least
until age 5, were conducted in the child's home. The mean Mental
Development Index and General Cognitive Index (GCI) scores were in the
intellectual normal range of 107 to 117 with a mean general cognitive
index at 4 years of 107.3 ± 14.2.
Covariates measured included maternal IQ, quality of care-taking
environment, obstetrical and postnatal factors.
8.3.5.3 Summary of differences between studies
The prospective studies differed to such an extent that it would
be surprising if they yielded identical findings. This does not mean
that the studies cannot be compared, merely that these differences and
their potential impact on study findings must be acknowledged in any
assessment of the studies as a group (Bellinger & Stiles, 1993). There
are substantial differences in the degree of confounding between lead
exposure and the other correlates of poor developmental outcomes. For
example, mean maternal IQ ranges from 121 (Boston) to 74-75
(Cleveland, Cincinnati), reflecting vast cohort differences in
socioeconomic standing and child-rearing context.
The studies also varied substantively in terms of the lead
exposure profiles observed. Site differences resulted in considerable
variability in postnatal lead exposures depending on exposure patterns
(residence near primary lead smelters, residence in deteriorated
housing, proximity to traffic, and quality of domestic
drinking-water).
Despite broad similarities in the approaches to data analysis,
there were substantial differences in the form in which the findings
were reported, impeding direct comparison of study results. Results
were diversely reported in terms of partial correlation coefficients
and standardized and unstandardized regression coefficients, and there
was variable use of raw (untransformed) and log exposure measures.
Most, but not all studies presented their results in a manner
conducive to some kind of quantification of dose-effects
relationships.
Clearly the variation in numbers of participants available to
individual studies (both initially, and as a consequence of attrition)
has a direct effect on the statistical power of each study - which is
also affected by the degree of confounding.
8.3.5.4 Results of studies
a) Infancy and early preschool assessments
The pattern of findings in infant and early preschool assessments
for which the principal instrument of assessment was the Bayley Scales
of Infant Development is inconsistent. Using different indices of
in utero exposure (e.g., prenatal, at delivery, cord or neonatal PbB
levels), three of the prospective studies found associations with
slower sensorimotor development up to 6 months or 1 year of age
(Ernhart et al., 1987; Dietrich et al., 1987, 1989) or up to 2 years
of age (Bellinger et al., 1987), but these tended to attenuate over
time (Dietrich et al., 1990; Bellinger et al., 1991a). Other studies
did not observe statistically significant adjusted relationships of
prenatal exposure with slower sensorimotor development (Moore et al.,
1989; Wigg et al., 1988; Cooney et al., 1989a,b; Wasserman et al.,
1992).
Two studies observed an inverse association between postnatal
lead exposure on mental development in later infancy (Wigg et al.,
1988; Wasserman et al., 1992), although in one study it was PbB level
at 6 months of age (Wigg et al., 1988) and in the other study it was
the level at 24 months (Wasserman et al., 1992) that was most
predictive.
b) Later preschool and school age assessment
Despite some inconsistency in the pattern of findings in infants,
there has been a convergence of positive findings on later
neurobehavioural outcomes in the prospective studies. This may
reflect:
1) the greater reliability and precision of measurement attained
with assessments of the older child; or
2) an effect of lead on abilities that cannot easily be tested
during infancy (e.g., executive, regulative and organizational
skills, higher order reasoning).
c) Boston study
Among 169 children assessed at age 57 months, the major finding
was an association between PbB level at 2 years of age and the GCI of
the McCarthy Scales. There was a decrease of 2.95 ± 1.42 GCI units for
an increase in PbB of 0.48 µmol/litre (10 µg/dl). Since the group had
an average PbB level of 0.336 µmol/litre (7.0 µg/dl) over 2 years,
this decrement was relevant to a range of approximately
0.19-0.67 µmol/litre (4-14 µg/dl) Bellinger et al., 1991a).
At 10 years of age, 148 children were reassessed. PbB
concentration at 2 years of age was inversely associated with the
WISC-R Full Scale IQ. Each 0.48 µmol/litre (10 µg/dl) increase was
associated with a 5.8 point decline in IQ (95% CI 1.7 to 9.9)
(Bellinger et al., 1992).
PbB concentration at 24 months was also inversely related to the
Battery composite score in the Kaufman Test of Educational Achievement
(Brief form) (-0.89 ± 0.24). The skills assessed were mathematics,
reading and spelling.
The point estimates associated with other postnatal ages were
generally in the same direction but only PbB level at 2 years revealed
conventional statistical significance. Other specific
neuropsychological tests did not reveal a clear pattern of
neuropsychological deficit (Stiles & Bellinger, 1993).
d) Cincinnati study
The Kaufman Assessment Battery for Children was administered to
approximately 260 children at 4 and 5 years of age (Dietrich et al.,
1991, 1992). The principal findings at 4 years were that higher
neonatal PbB levels were associated with poorer performance on all
Kaufman subscales. However, this inverse association was limited to
children from the poorer families. Following full covariate
adjustment, few statistically significant associations between
postnatal PbB levels and Kaufman scales could be found. However, the
results did suggest a weak relationship between postnatal PbB levels
and performance on a Kaufman subscale which assesses visual spatial
and visual-motor integration (adjusted regression coefficient -0.12 SE
not published).
At 5 years of age, postnatal PbB levels were associated with
poorer performance an all subscales of the Kaufman Battery. However,
after adjustment for covariates, few statistically significant
relations remained. Nevertheless, as at 4 years of age, the subscale
assessing visual-spatial and visual-motor skills was most sensitive,
with average lead exposure during the 4th year of life being
significantly associated with performance (-0.12 units per
0.048 µmol/litre or per µg/dl, SE not published).
At the age of 6.5 years, 253 children in the Cincinnati cohort
were administered the WISC-R (Dietrich et al., 1993a). The major
findings were that postnatal PbB concentrations were inversely
associated with full-scale IQ and performance IQ. Following
statistical adjustment for covariates, including maternal IQ and
quality of home care, a statistically significant relationship was
retained between postnatal PbB concentrations at nearly all ages (and
including lifetime averages) and Performance IQ. Further analysis
revealed that average lifetime PbB concentrations in excess of
0.96 µmol/litre (20 µg/dl) were associated with deficits in IQ of the
order of 7 points, when compared with children with mean
concentrations of less than 0.48 mol/litre (10 µg/dl). The regression
coefficient for Performance IQ on mean lifetime PbB level was -0.26
(± 0.12) IQ units per 0.048 µmol/litre or per µg/dl. This corresponded
to a 2.6 unit decrement associated with a 0.48 µmol/litre (10 µg/dl)
movement in lifetime average PbB level, for which the average
(interpolated graphically) is close to 0.67 µmol/litre (14 µg/dl).
At 6 years of age, the Bruininks-Oseretsky Test of Motor
Proficiency (BOTMP) was administered to 245 children in the Cincinnati
Cohort (Dietrich et al., 1993a). Following statistical adjustment for
covariates, neonatal PbB levels were associated with poorer
performance on a measure of upper-limb speed and dexterity and the
fine-motor composite. Postnatal PbB levels also remained significantly
associated with poorer scores on measures of bilateral coordination,
visual-motor control, upper-limb speed and dexterity, and the fine
motor composite.
Children having an average mean lifetime PbB level of
approximately equal to or exceeding 0.43 µmol/litre (9 µg/dl) appeared
to experience a deficit in both fine and gross motor skills relative
to children in the lowest PbB quartile. Children in the highest
average lifetime PbB quartile had scores on the gross-motor subtest
assessing bilateral coordination of approximately 0.5 standard
deviations (2.5 points) lower than their counterparts in the lowest
quartile. Children in the highest average lifetime quartile also
scored more poorly in the fine-motor functioning, having scores of
approximately 0.6 standard deviations lower (6.3 points) than those in
the lowest quartile.
e) Cleveland study
The WPPSI was administered to 242 children at the age of 4 years
and 10 months (Ernhart et al., 1989a). Statistically significant
correlations between IQ (full scale and subscales) and PbB level
measured pre- and perinatally and at ages 2 and 3 years (and a
lifetime postnatal average) became non-significant after adjustment
for covariates. No estimates for effect size were presented in the
original report (Ernhart et al., 1989a).
f) Glasgow study
No neurobehavioural assessments were carried out beyond 2 years
of age (Moore et al., 1989).
g) Kosovo study
No results beyond 2 years of age were reported (Graziano et al.,
1990; Wasserman et al., 1992).
h) Port Pirie cohort study
The principal finding at 4 years was an inverse association of
McCarthy Scales of Children Abilities (MSCA) scores (General Cognitive
Index (GCI), Perceptual Performance and Memory) with most indices of
lead exposure (McMichael et al., 1988). After adjustment for
covariates, the association of GCI was not signifi-cant at certain
specific ages. However, it remained significant for the integrated
postnatal average for which an effect size of 7.2 GCI points lost in
association with an increase of lifetime average PbB level from 0.5 to
1.5 µmol/litre (10.4-31.2 µg/dl) was estimated.
Between 7 and 8 years of age the IQ of 494 children was assessed
with the WISC-R (Baghurst et al., 1992). After adjustment for
covariates there was little association with pre- and perinatal lead
exposure assessments but significant decrements in full-scale IQ of
between 3.7 and 4.8 points (depending on age) for each (natural) log
unit increase in lifetime average PbB concentration were observed. The
estimated effect size for the lifetime average exposure up to 3 years
of age was a loss of 5.3 IQ points in association with an increase in
PbB level from 0.48 to 1.44 µmol/litre (10 to 30 µg/dl).
The Block Design subscale of the WISC-R, which tests spatial
abilities, exhibited the strongest association with lead exposure and
estimated effect sizes were stronger in girls than boys for both the
MSCA and WISC-R.
i) Sydney study
At the age of 4 years, 207 children were assessed with the
McCarthy Scales of Children Abilities (MSCA) and virtually no
significant associations with any measures of PbB were observed
(Cooney et al., 1989b).
Follow-up assessments in 175 children using WISC-R at age 7 also
yielded no associations (crude or adjusted) with blood lead history
(Cooney et al., 1991).
8.3.5.5 Questions prospective studies have not answered
A disappointment of the prospective studies was their inability
to reach any obvious consensus on the behavioural phenotype associated
with low-level lead exposure, or on age(s) of critical sensitivity.
This latter fact may reflect the phenomenon of intra-individual
"tracking", whereby an individual maintained approximately consistent
ranking with respect to his or her PbB concentration at any age, or it
may reflect the need for chronic exposure over extended periods in
order for causal effects to become apparent.
8.3.5.6 Attempting a consensus
As discussed previously, no clear delineation of an age (or age
range) of maximal sensitivity to lead exposure has emerged from the
prospective studies, and there is a diversity of reporting styles
which makes attempts to compare studies and establish a consensus of
opinion very difficult.
Nevertheless, it would appear essential to attempt some sort of
synthesis of studies performed so far, given that the social and
economic consequences of preventing an adverse health effect are
potentially enormous.
A meta-analysis by Needleman & Gatsonis (1990) concluded that the
overall pattern of results was most unlikely to have occurred by
chance. As an approach to the problem of obtaining the "best" estimate
of the effect of lead exposure on neuro-behavioural development, a
common estimate of the partial correlation between lead burden and IQ
was determined. However, in order to derive such an estimate with some
degree of certainty, it is essential to find a common outcome, and to
identify studies for which at least broadly comparable exposure
measures are available.
Schwartz (1993) has identified three prospective studies and five
cross-sectional studies that examined the association of lead exposure
as determined by PbB concentrations with full scale IQ. For the
prospective studies, the exposure measures used were 24-months PbB
level (Boston) and lifetime average up to 3 years of age (Cincinnati
and Port Pirie).
There was only one measure for each of the cross-sectional
studies. Despite the compromises necessary for such a comparison, the
estimated effect sizes for an increase in PbB level from 0.48 to
0.96 µmol/litre (10 to 20 µg/dl) for all seven studies lay between 1
and 6 IQ points lost (with five of the studies lying between 1 and 4),
and the associated 95% confidence limits only just embraced the value
zero for 3 of the 7 studies.
Thus, a broad consensus does emerge from a rough comparison. From
the estimates summarized by Schwartz (1993), a weighted mean for the
six coefficients accompanied by a standard error of 0.4 was estimated
to be 2.6 IQ units lost for an increase in PbB from 0.48 to
0.96 µmol/litre (10 to 20 µg/dl).
8.3.6 Task group overview and interpretation of prospective studies
on children
8.3.6.1 Rationale
As a complement to the narrative review, prepared by the Task
Group, a quantitative overview of the findings of the prospective
studies was considered to be a valuable means of assessing the overall
strength of evidence for an association between blood lead summary
measures and school age IQ.
While individual studies vary somewhat in their design and their
analysis strategies, their underlying objectives are closely related.
For any generally applicable conclusions on general population lead
exposure and its effect on intellectual attainment to be reached, some
consistency of evidence across studies is highly desirable.
Since any single study has considerable random error in
estimating a relationship, a quantitative overview (or meta-analysis)
can be a valuable means of defining what is the plausible magnitude of
statistical association between PbB measures and child IQ.
It seems appropriate, given the quite different types of study
design, to present two separate meta-analyses dealing with prospective
studies and cross-sectional studies, respectively.
Any such meta-analysis needs cautious interpretation. The caveats
in drawing causal inferences from statistical associations apply to
any meta-analysis just as they do to individual studies. Any increase
in statistical precision regarding the magnitude of association does
not eliminate the potential for bias in any observational study on
human populations.
8.3.6.2 The prospective studies
There are four prospective studies: Boston (Bellinger et al.,
1992); Sydney (Cooney et al., 1989a,b); Cincinnati (Dietrich et al.,
1993a); and Port Pirie (Baghurst et al., 1992) which presented
quantitative results in a form suitable for an overview (i.e. with
regression coefficients and standard errors). The Task Group
concentrated on school age assessment because:
a) the studies used the same outcome measure (WISC-R), which is
widely accepted and has been previously evaluated in cross-
sectional studies; and
b) intellectual assessment at primary school age (6-10 years) is,
perhaps, of greatest overall relevance in reaching a consensus on
the public health importance of childhood lead exposure.
Unfortunately, the Cleveland study did not publish findings in
the same quantifiable manner. The potential impact of this apparently
"negative" study on the overall evidence will be discussed in section
8.3.6.2(c).
a) Measures of exposure, outcome and association
All four studies have used blood lead as the measure of body lead
burden, but the timing and frequency of examinations has varied as has
the choice of summary measures (averages over time).
In a meta-analysis it seems appropriate to concentrate on
measures of association after adjustment for confounders. While there
is some variation in choice of confounders, the two key covariates
(mother's IQ and HOME scores) appear in all four studies.
The Port Pirie study used the logarithm of the PbB concentration
in the multiple regression analysis while the other three studies used
untransformed PbB concentration. This difference in statistical style
can be reconciled by converting the lead regression coefficients to
estimated changes in IQ for a specific increase in PbB concentration,
i.e. from 0.48 to 0.96 µmol/litre (10 to 20 µg/dl). This equals 10
times the regression coefficient (as published) for the Boston and
Cincinnati studies and log 2 times the regression coefficient for the
Port Pirie study.
It is recognized that the specific subscales of performance and
verbal IQ are of separate interest (e.g., the performance IQ has been
rather consistently shown to be more reliably associated with
postnatal lead exposure amongst the prospective studies (McMichael et
al., 1988; Bellinger et al., 1991a; Dietrich et al., 1991, 1992,
1993b). However, it appears more important to focus on full-scale IQ
as the primary outcome measure, otherwise concerns about post-hoc
selection may be raised. Such full-scale IQ was measured at age 6.5, 7
and 10 years in the Cincinnati, Port Pirie, Sydney and Boston studies,
respectively.
b) Display of individual study findings
Since most studies have not shown perinatal PbB level to be
predictive of intellectual performance, attention will be focused on
the various measures of postnatal PbB level. In fact, there are seven
such measures in each study that have been related to full-scale IQ in
the reports. Tables 21 and 22 summarize the magnitude of association
together with the standard error for all reported analyses, each after
adjustment for confounders.
Except for the earliest postnatal PbB values in the Boston and
Cincinnati studies, the adjusted associations between PbB level and IQ
are consistently negative (i.e. inverted). However, the "random noise"
in each study's analysis means that there is considerable variation in
levels of statistical significance. Hence, there is a need for a more
systematic overview combining the evidence from all studies.
c) A quantitative overview
The prime difficulty here is the different choices of PbB
summaries in the four studies. In undertaking a meta-analysis it would
have been better if all studies had essentially the same PbB measures
in their multiple regressions. Possible choices might have been (i) a
specific time point, e.g., 2 years or (ii) the means over a specific
interval, e.g., 0 to 5 years.
However, from the available analyses, some broadly similar
summaries can be chosen, as follows:
Table 21. Magnitude of association between blood lead and full-scale IQ
from four prospective studies
No. of Age at which blood lead Estimated change in
children was measured full-scale IQ a
Boston 148 year 1 0.0 (1.6)
year 1´ -1.2 (1.8)
year 2 -5.8 (2.1)
year 5 -2.6 (2.9)
year 10 -4.6 (5.2)
mean of years 2-5 -8.2 (2.8)
mean of years 2-10 -8.6 (3.4)
mean of 6 months to
10 years (unpublished) -5.7 (3.2)
Cincinnati 251 mean of year 1 0.1 (1.4)
mean of year 2 -0.2 (0.8)
mean of year 3 -1.3 (0.9)
mean of year 4 -1.5 (1.0)
mean of year 5 -2.3 (1.1)
mean of year 6 -3.3 (1.4)
mean of years 1-6 -1.3 (1.1)
Port Pirie 490 mean of first 15 months -2.8 (1.4)
mean of years 1-2 -3.2 (1.5)
mean of years 1-3 -3.3 (1.6)
mean of years 1-4 -3.2 (1.7)
mean of years 1-5 -3.0 (1.7)
mean of years 1-6 -2.8 (1.7)
mean of years 1-7 -2.6 (1.7)
Sydney 175 mean of years 1 and 2 -2.7 (2.8)
mean of years 3, 4 and 5 -1.9 (1.9)
year 7 -1.4 (1.7)
mean of years 1-7 -1.6 (2.2)
a From 0.48 to 0.96 µmol/litre (10 to 20 µg/dl) blood lead
(and its standard error) after adjustment for confounders
Table 22. Association between blood lead levels and full-scale
IQ from ten cross-sectional studies
No. of Estimated change in
subjects full-scale IQa
Lavrion, Greece 509 -2.7 (0.7)
Edinburgh, Scotland 501 -2.6 (0.9)
Greenwich, England 129 -5.6 (3.2)
European, multi-centre
Bucharest, Romania 301 -0.4 (2.6)
Budapest, Hungary 254 +0.8 (1.8)
Modena, Italy 216 +0.9 (4.2)
Sofia, Bulgaria 142 +2.2 (2.3)
Dusseldorf 1, Germany 109 -4.6 (4.5)
Dusseldorf 2, Germany 109 -3.9 (5.1)
Zagreb, Croatia 48 -1.5 (4.5)
a From 0.48 to 0.96 µmol/litre (10 to 20 µg/dl) blood lead
(and its standard error) after adjustment for confounders
i) There is some advantage in considering the mean PbB level over a
number of years, since it summarizes cumulative exposure and also
achieves a more reliable ranking of individuals than a single value.
The available long-term means are:
Boston: mean of 6 months to 10 years (D. Bellinger,
personal communication to the IPCS, 1993)
Cincinnati: mean of years 1 to 6
Port Pirie: mean of years 1 to 7
Sydney: mean of years 1 to 7
Although they differ in frequency and age range, these three
summaries should be very highly correlated with one another. The
resultant meta-analysis is displayed in Fig. 15. Each of the
individual study confidence intervals includes zero, indicating that
significance at P < 0.05 was not reached. However, the combined
evidence, weighting studies according to the inverse of their
variance, produces a weighted mean decrease in full-scale IQ of 2
points for a 0.48-µmol/litre (10-µg/dl) increase in PbB level, with
95% confidence interval from -0.3 points to -3.6 points (P = 0.01).
ii) An alternative approach is to consider PbB level at a specific
time or average over shorter intervals of time. For this approach, the
analyses of these studies are most comparable during the first three
years, as follows:
Boston: 2 year measure
Cincinnati: mean of year 3
Port Pirie: mean of years 1 to 3
Sydney: mean of years 1 and 2
This second meta-analysis is displayed in Fig. 16. The data here
support an inverse association more strongly than in Fig. 15, and the
combined evidence estimates a mean decrease of 2.6 IQ points for a
0.48-µmol/litre (10-µg/dl) increase in PbB level, with 95% confidence
interval from -1.2 points to -4.0 points (P < 0.001).
However, it should be noted that the estimates here are heavily
dependent on the choice of time points for PbB level. For instance, if
one instead chose:
Boston: 1.5 year measure
Cincinnati: mean of year 2
Port Pirie: mean of years 1 and 2
Sydney: mean of years 1 and 2
then the combined estimate is roughly halved in magnitude and is of
borderline significance.
One needs to recall that the other prospective study, carried out
in Cleveland (Ernhart et al., 1989a), has no equivalent data, but
reported no significant association. This study comprised about 150
evaluated children, and would influence the overall evidence in a less
significant direction if the data were able to be included.
8.3.6.3 A quantitative assessment of the cross-sectional studies
There have been more cross-sectional studies that have related
body burden to full-scale IQ in school-age children, but they cannot
all be included in a single meta-analysis. Here attention is focused
on the PbB studies, and it would be appropriate subsequently to
McIntosh et al., 1985) in rat brain. In humans the exact relationship
between PbB and plasma biopterins has not been properly defined.
8.1.4 Nicotinamide adenine dinucleotide synthetase
A recent study of nicotinamide adenine dinucleotide (NAD)
synthetase in erythrocytes indicates that this enzyme is sensitive to
inhibition by lead (and zinc) and is a sensitive indicator of lead
exposure. Zerez et al. (1990) found NAD synthetase activity reduced in
three subjects with elevated PbB levels in the range
1.63-3.46 µmol/litre (34-72 µg/dl).
8.1.5 Nutritionally affected groups
A body of evidence, summarized and discussed by Mahaffey (1985),
indicates that a high dietary calcium intake tends to decrease lead
absorption and retention in infants, young children and adults. Other
evidence suggests that in groups including children, with
self-selected diets, low dietary calcium intakes are associated with a
greater prevalence of elevated PbB levels (Mahaffey, 1985).
Some human data indicate that adults with low iron states have an
increased absorption of both iron and lead (Watson et al., 1980,
1986). In children, particularly those from low-income families, there
is often an association between iron deficiency and elevated PbB (Yip
& Dallman, 1984; Mahaffey, 1985). While this by itself does not
indicate a causal relationship, when considered in the context of data
on adults and animals, it suggests that iron deficiency in children
can result in increased lead absorption.
Alcoholics and people who consume excess alcohol may be at
increased risk of adverse effects from lead. In animal studies,
alcohol and lead synergistically inhibit ALAD, hepatic glutamic
oxaloacetic transaminase and glutamic pyruvic transaminase activities
(Flora & Tandon, 1987) supporting the hypothesis regarding risks to
humans.
8.2 Haematopoietic system
Lead-induced anaemia can be a direct consequence of inhibition of
haem biosynthesis; it is not necessarily associated with iron
deficiency. It may be associated with alterations of globin syntheses
(Albahary, 1972). More importantly, the synthesis of alpha- and
ß-globin chains may become asynchronous (White & Harvey, 1972).
8.2.1 Anaemia
Based on data published by Lilis et al. (1978), Baker et al.
(1979), Grandjean (1979), and earlier data, the threshold PbB level
for a decrease in haemoglobin has been estimated to be 2.40 µmol/litre
(50 µg/dl) for occupationally exposed adults (US EPA, 1986a).
Grandjean et al. (1989a) demonstrated the reduced ability of the
erythropoietic system to regenerate after blood withdrawal
(0.45 litre) in 25 lead-exposed battery workers (average PbB level
2.14 µmol/litre, 44.5 µg/dl) compared with 25 age-matched controls
(average PbB level 0.35 µmol/litre, 7 µg/dl). The haematological
parameters, except for erythropoietin were otherwise comparable for
the two groups. The effect was attributed to an effect on haem
synthesis and possible decreased erythrocyte survival in lead-exposed
workers. Lead has been found to depress serum levels of
erythropoietin, a hormone which regulates erythrocyte formation
(Graziano et al., 1991), which might also affect the reserve capacity
for erythropoiesis.
The PbB threshold for decreased haemoglobin levels in children is
estimated to be approximately 1.92 µmol/litre (40 µg/dl) (IPCS, 1977).
However, a cross-sectional epidemiological study of 579 children, aged
1-5 years in 1974, living in close proximity to a primary lead smelter
showed that adverse effects on haematocrit may occur at lower PbB
levels (Schwartz et al., 1990). Anaemia, defined as a haematocrit
below 35%, was not found at PbB levels of less than 0.92 µmol/litre
(20 µg/dl). There was a strong non-linear dose-response relationship
at higher PbB levels, which was influenced by age.
Kutbi et al. (1989) studied 200 boys aged 6-8 years. The mean PbB
was 0.33 µmol/litre (6.9 µg/dl; range 0.07-1.14 µmol/litre or
1.4-23.8 µg/dl). Subdividing the group at 0.72 µmol/litre (15 µg/dl),
they found a negative correlation between PbB level and all
haematological values for the "upper normal" (N = 7) compared with the
"normal" group (N = 193). The pattern of haematological parameters was
described as predictive of microcytic anaemia.
Anaemia has been commonly associated with the adverse effects of
occupational lead exposures. It is an effect that is easily diagnosed
clinically and is recognized as a marker of lead toxicity. Anaemia may
result from either a decrease in haemoglobin production or an increase
in the rate of destruction of erythrocytes. An analysis, made in 1974,
of the association between PbB levels and haematocrit in 579 children
(1-5 years of age) living near a primary lead smelter has recently
been presented (Schwartz et al., 1990). A haematocrit value of less
than 35% was used to indicate an adverse effect. It should be noted
that the effect of iron deficiency was not taken into account in
analysing the results. The study concluded that there was no adverse
effect of lead at PbB levels below 0.96 µmol/litre (20 µg/dl).
Furthermore, the risk of having a haematocrit value below 35% for
1-year-olds was 2% at PbB levels between 0.96 and 1.87 µmol/litre
(20 and 39 µg/dl). The degree of iron deficiency may account for a
substantial proportion of this 2%. In this study, the level at which
an effect of lead on the induction of anaemia was demonstrable was
about 1.92 µmol/litre (40 µg/dl).
8.2.2 Pyrimidine-5'-nucleotidase activity
Inhibition of erythrocyte pyrimidine-5'-nucleotidase leads to
accumulation of pyrimidine nucleotides, which has been associated with
induction of basophilic stippling.
Impaired function of erythrocyte pyrimidine-5'-nucleotidase
occurs in lead-exposed workers and as a genetically induced enzyme
deficiency. In in vitro systems the enzyme is inhibited by lead,
cadmium, mercury and zinc ions (and perhaps other metal ions) (Paglia
et al., 1975; Pagliuca et al., 1990; Ichiba & Tomokuni, 1990).
Graphs of PbB concentration against erythrocyte pyrimidine-
5'-nucleotidase activity in children have been reported for 21
children aged 2-5 years (Angle & McIntire, 1978) and for 42 aged 1-5
years (Angle et al., 1982) who participated in a preventative
programme because they had previously had PbB levels above
1.44 µmol/litre (30 µg/dl) or zinc protoporphyrin (ZPP) greater than
60 µg/dl. Both plots show considerable scatter although there was a
significant inverse logarithmic correlation. However, data relating to
the reproducibility, precision and accuracy of assessing inhibition of
erythrocyte pyrimidine-5'-nucleotidase were not presented.
This test may be capable of analytical refinement when assessment
of the sensitivity to lead (and other metals) exposure might more
reasonably be made. It does not yet appear likely to provide a routine
measure for low-lead exposure assessment.
8.2.3 Erythropoietin production
Graziano et al. (1991) found depressed serum erythropoeitin
levels in females at mid-pregnancy and at delivery associated
statistically with PbB level. Erythropoietin is a glycoprotein
produced in the renal proximal tubules which regulates both
steady-state and accelerated erythrocyte production. The study was not
adequate to define a dose-response relationship.
8.3 Nervous system
8.3.1 Historical perspective
Gross toxic effects of lead on the nervous system were reported
by ancient Greek physicians. A brief summary of such reports, and
those of Roman physicians and 18-19th century toxicologists, was
published by Kazantzis (1989). The syndrome was known as "painter's
colic", which included abdominal pain, constipation and paralysis,
symptoms now covered by the term "lead encephalopathy".
Lead colic was usually accompanied by effects on the nervous
system (lead palsy) in cases of acute and chronic poisoning, which
were often but not always fatal. Lead poisoning was well recognized as
an occupational hazard. However, it was also associated with
consumption of lead-contaminated water, wine, cider, rum from the West
Indies, and food prepared or stored in lead or lead-glazed utensils.
For example, Kazantzis (1989) described cases of lead poisoning in
girls resulting from consumption of drinking-water kept in lead-lined
cisterns, and also described the Devonshire colic attributed by Sir
George Baker in 1767 to the consumption of cider prepared in presses
made of lead or lead alloys.
The early toxicologists clearly recognized that the dose
(exposure) and form of lead was important in relation to absorption
and were able to demonstrate the presence of lead in tissues of fatal
cases of poisoning. There are many individually described, detailed
case studies in their writings.
8.3.2 Neurotoxic effects in adults
Despite the long recognition of lead poisoning, new clinical
cases continue to be the subject of published reports (Cueto et al.,
1989; Kocak et al., 1989; Zuckerman et al., 1989; Friedman &
Weinberger, 1990; Gupta et al., 1990; Mitchell-Heggs et al., 1990;
Nosal & Wilhelm, 1990; Sharma et al., 1990; Schneitzer et al., 1990;
Veerula & Noah, 1990). Although they are important, the numbers of
these poisoning cases are comparatively small compared with the
population at risk to the potential effects of exposure to lead from
environmental and dietary sources. Of particular recent concern have
been the possible neurotoxic effects on the developing child.
8.3.2.1 Central nervous system
In a study of 158 secondary lead smelter workers, Lilis et al.
(1977) found that 64% reported CNS symptoms and that there was early
occurrence of symptoms within 1 year of exposure. PbB concentrations
were elevated (about 3.36 µmol/litre or 70 µg/dl) at the time of
examination but many of the subjects had received chelation therapy.
Fischbein et al. (1979) noted CNS symptoms in 21 of 81 employees
of law enforcement agencies working in firing ranges. Symptoms
correlated with PbB levels and were present in three-quarters of
subjects having a PbB level > 2.4 µmol/litre (50 µg/dl).
Hanninen et al. (1979) reported that 49 lead-exposed workers
(including 21 male and 8 female storage battery and 16 railroad
engineering workshop employees) whose PbB level had never exceeded
3.36 µmol/litre (70 µg/dl) experienced more subjective symptoms than a
control group; excess symptoms were proportional to lead uptake.
Fischbein et al. (1980) studied 90 telephone cable splicers
having intermittent exposure to lead. They reported that 26 complained
of CNS symptoms (mean PbB level of 1.36 µmol/litre, 28.4 µg/dl, and
ZPP of 70.3 µg/dl) compared with controls (mean PbB level of
1.32 µmol/litre, 27.4 µg/dl, and ZPP of 49.3 µg/dl).
Awad El Karim et al. (1986) studied 92 lead acid battery workers
and compared them with 40 oil mill worker controls. They found CNS
symptoms in 50% of lead-exposed workers and measured PbB levels in 46
subjects working in different sections of the plant. More than 95% of
exposed workers had PbB levels above 1.92 µmol/litre (40 µg/dl),
whereas the mean PbB in the control group was 1.0 µmol/litre
(21 µg/dl).
Impairment of psychological and neuropsychological test
performance has been reported for lead-exposed workers (Hanninen et
al., 1979; Hogstedt et al., 1983; Parkinson et al., 1986; Araki et
al., 1986a,b; Huang et al., 1988a; Stollery et al., 1989, 1991).
The critical flicker fusion test has been reported to be a
sensitive indicator of CNS changes associated with exposure to lead.
Wooller & Melamed (1978) found significantly lower critical flicker
fusion in subjects with PbB levels above 2.88 µmol/litre (60 µg/dl)
than in those with levels below 0.96 µmol/litre (20 µg/dl). Williamson
& Teo (1986) found a significant decrease in mean flicker fusion in 59
lead-exposed workers (mean PbB > 2.3 µmol/litre or 48 µg/dl) compared
with matched controls. However, Gennart et al. (1992b) found no
difference between lead-exposed workers (mean PbB level
2.45 µmol/litre or 51 µg/dl, N = 98) and controls (mean PbB level
1.00 µmol/litre or 20.9 µg/dl, N = 85).
Stollery et al. (1989) studied the performance of 91 men,
occupationally exposed to inorganic lead, in a series of tests
designed to assess cognitive function. Subjects were grouped in "low"
(mean PbB 0.48 µmol/litre or 10 µg/dl, mean ZPP 8.7 µg/dl, N = 28),
"medium" (mean PbB 1.44 µmol/litre or 30.1 µg/dl, mean ZPP 22.4 µg/dl,
N = 27) and "high" (mean PbB 2.43 µmol/litre or 50.7 µg/dl, mean ZPP
58.8 µg/dl, N = 36) ranges of PbB concentrations. Results showed that
occupational exposure to lead impaired performance in a range of tests
(sensory motor reaction time, memory, attention, verbal reasoning,
spatial processing). Workers having PbB levels in excess of
1.92 µmol/litre (40 µg/dl) showed clear evidence of impairment on
tests of serial reaction time and category search. The authors
concluded that sensory motor, rather than cognitive, requirements of
many psychological tasks provide the most sensitive index of the early
effects of chronic low level lead exposure.
In a follow-up study (Stollery et al., 1991), 70 workers were
re-tested (three times within 8 months). Performance deficits in the
"high" (mean PbB 2.49 µmol/litre or 51.8 µg/dl, mean ZPP 77.4 µg/dl,
N = 22) group were not altered by practice or continued exposure. The
main deficit was a slowing of sensory motor reaction time coupled with
difficulties in remembering incidental information. There was little
evidence of impairment in workers having PbB levels less than
1.92 µmol/litre (40 µg/dl).
Finally, it appears that neuroelectrophysiological testing is a
sensitive and objective indicator of the CNS effects of lead. The
results of Araki et al. (1986b, 1987, 1992) using short-latency
somatosensory and visual evoked potentials and auditory event-related
potential (P300) indicate that subclinical electrophysiological
effects of lead occur not only in peripheral nerves but also in the
CNS.
8.3.2.2 Peripheral nervous system
Peripheral neuropathy is a common sign of chronic, high level
lead exposure, often manifesting as weakness in the upper or lower
limbs. At lower levels of lead exposure, nerve conduction velocity
(NCV) has provided a more sensitive indicator of peripheral nerve
dysfunction. More than 30 published studies have measured the
conduction velocity of electrically stimulated sensory and motor
nerves in workers exposed to lead. However, these studies have yielded
somewhat mixed results, with many showing a decrease in NCV in
relation to lead exposure (generally indexed as PbB concentration) and
a few showing no effect or occasionally even an increase in NCV
associated with lead exposure (Seppäläinen & Hernberg, 1980; Davis &
Svendsgaard, 1990).
Various reasons may underlie this lack of uniformity in NCV
findings. For example, studies may differ in methodological features,
in the characterization of lead exposure (e.g., single time-point
versus time-weighted average PbB levels) or in the handling of
confounding variables such as nerve temperature and age of subject
(Ehle, 1986). Other important factors accounting for some of the
apparent inconsistency in this area of research may be the possible
antagonistic effect of zinc to lead and differences in nerves selected
for measurement in different studies (e.g., slow versus fast fibres
(Araki et al., 1986c; Murata & Araki, 1991). A statistical
meta-analysis and critical review of 32 NCV studies by Davis &
Svendsgaard (1990) indicated that the median motor nerve shows effects
of lead more reliably than other nerves (e.g., median sensory or
ulnar).
Despite these complications, certain key well-conducted studies
provide compelling evidence of a causal relationship between lead
exposure and reduction in NCV. Araki et al. (1980) measured median
motor NCV before and after PbB levels of workers were lowered through
chelation therapy. Depending on the amount of reduction in PbB level
achieved by chelation and on a given worker's baseline NCV,
significantly improved NCV was measured in 7 out of 14 lead-exposed
workers. For all 14 workers the improvement in NCV was significantly
correlated with the decrease in PbB level (r = -0.573, P < 0.001).
In another key study, Seppäläinen et al. (1983) followed workers
prospectively from the beginning of their employment in a battery
plant, dividing them into two exposure categories (above and below the
median PbB level of 1.44 µmol/litre (30 µg/dl). Median motor NCV was
significantly reduced in the workers with blood lead levels above
1.44 µmol/litre (30 µg/dl) at 1-, 2-, and 4-year evaluation points,
despite attrition (and reduced statistical power) that resulted in
only five subjects per group after 4 years. Based on this well-
conducted prospective study, 1.44 µmol/litre (30 µg/dl) would appear
to be the lowest-observed-adverse-effect level for reduced NCV in
adults. Although another well-conducted prospective study (Spivey et
al., 1980) was unable to find an effect of lead on NCV at higher
exposure levels, NCV in the median nerve was not measured in that
study. Triebig et al. (1984) reported a dose-dependent decrease in NCV
at PbB levels above 3.36 µmol/litre, no dose-effect relationship being
detectable below this concentration.
8.3.2.3 Autonomic nervous system
There have been two reports of tests for autonomic nervous
function following lead exposure. Teruya et al. (1991) conducted a
cross-sectional survey of 172 male workers exposed to lead (mean PbB
level, 1.73 µmol/litre or 36 µg/dl, range 0.24-3.648 µmol/litre or
5-76 µg/dl), by measuring electrocardiographic R-R interval
variability (CVRR). Age-adjusted CVRR during deep breathing in
workers with PbB levels above 1.44 µmol/litre (> 30 µg/dl) was
significantly decreased as compared to those with PbB levels less than
0.96 µmol/litre (< 20 µg/dl). In addition, significant dose-response
and dose-effect relationships were observed between the PbB levels and
R-R interval variation during deep breathing among the workers with
PbB levels above 0.96 µmol/litre (> 20 µg/dl).
In a study reported by Murata & Araki (1991), the R-R interval
(CVRR) and two component coefficients of variation in the R-R
interval (C-CVRSA) were measured in 16 gun metal foundry workers
exposed to lead, zinc and copper (mean PbB level, 1.63 µmol/litre or
34 µg/dl, range 0.768-2.88 µmol/litre or 16-60 µg/dl). The CVRR and
component C-CVRSA (respiratory sinus arrhythmia) were significantly
lower in the workers than in age-matched controls, whereas the
C-CVMWSA (Mayer wave-related sinus arrhythmia) was unaffected. The
authors noted that zinc may have been antagonistic to the lead effects
in this study. Since the CVRR during deep breathing and the
component C-CVRSA of respiratory sinus arrhythmia reflect
parasympathetic activity, these two reports indicate potential
dysfunction of the autonomic nervous system (mainly, parasympathetic
activity) at average PbB levels of approximately 1.68 µmol/litre
(35 µg/dl).
8.3.3 Neurotoxic effects in children
The majority of the epidemiological research on the health
effects of lead has been focused on children because, in comparison
with adults, they are more vulnerable to lead in several respects
(Davis & Grant, 1992). For example, children typically engage in
hand-to-mouth activities (sucking fingers, putting food or other
objects in the mouth) that result in greater ingestion of lead than
adults normally experience. Also, because of their greater absorption
and retention of lead, the body burdens in children resulting from a
given external exposure level tend to be higher than adults.
Furthermore, the relatively greater exposures and body burdens of
children occur during sensitive periods of development. Finally, it
appears that children are generally more sensitive to the
toxicological effects of lead at a given internal exposure (PbB)
level. The lowest-observed-effect levels for various end-points (e.g.,
encephalopathy, anaemia, reduced haemoglobin, elevated EP, slowed NCV,
impaired neurobehavioural function) are lower in children than in
adults (US EPA, 1986a, 1990).
Section 8.3.3.2 describes findings from the main epidemiological
cross-sectional studies that have been published since 1979. This
constitutes a very substantial collection of research findings but has
certain interpretive limitations, particularly the lack of information
on exposure history.
More recent research effort has concentrated on prospective
epidemiological studies of birth cohorts repeatedly evaluated, mostly
up to the school age years. These prospective studies are described in
section 8.3.3.3.
Section 8.3.3.4 presents a quantitative overview of the
collective evidence from the prospective studies and discusses their
interpretation and the problems of inferring causality from such
observation data.
8.3.3.1 Historical perspective
While there have undoubtedly been cases of lead poisoning in
children as long as lead has been used by man, most of the attention
was previously given to cases of poisoning as a result of occupational
exposure in adults. In 1892 Gibson and colleagues in Australia
reported a case series of ten young children with lead colic. It was
not until twelve years later that peeling paint in the children's
homes was identified as the lead source (Gibson, 1904).
In 1943, Byers & Lord (1953) reported the results of an important
study, the first to suggest that there were neurological after-effects
of lead poisoning, in the absence of cerebral oedema and high
intracranial pressure, following acute lead encephalopathy. However,
in other cases it was felt that neurological manifestations
disappeared if the ingestion of lead was stopped (McKhann & Vogt,
1933). The case series of 20 school-age children had been hospitalized
for lead poisoning but without signs of acute lead encephalopathy, and
all were later discharged as cured. At follow-up, only one of the
children was making satisfactory progress at school. The children were
described as showing a variety of symptoms, including poor academic
achievement, intellectual deficits, sensory-motor deficits and
behaviour disturbance.
In the years following the findings of Byers & Lord (1953), a
number of case control studies were carried out that examined mental
retardation (Beattie et al., 1975) or hyperactivity (David et al.,
1972).
In addition, as a result of the growing concern about the dangers
of lead encephalopathy, studies were set up to investigate the
possibility that subclinical levels of lead (i.e. levels producing no
overt signs of lead encephalopathy) cause more subtle neurological
damage to children. Many of these studies investigated children
identified by screening clinics as having raised lead burdens, but
others were sited around smelters or lead works where children were
found to have elevated lead levels. The studies were important in
determining the areas of functioning which might be affected, and in
drawing attention to the methodological problems involved in carrying
out such studies. The main difference between the smelter studies or
studies around lead works, and clinical studies is that in the former
the primary source of environmental lead is known. In the hospital or
clinical studies it is likely that the ingestion of paint flakes, or
less often leaded putty, was in most cases the primary cause of the
excess lead exposure, although the exact reason for the increased lead
burden is rarely known.
Clinical and smelter studies were carried out in the United
Kingdom and USA and in general investigated children with PbB levels
above 1.92 µmol/litre (40 µg/dl), and compared their performance with
that of children with levels below this value.
Although some of the clinical and smelter studies found a
lead-associated deficit (Perino & Ernhart, 1974; Landrigan et al.,
1975), others did not (Lansdown et al., 1974). In general the studies
were methodologically flawed, with small samples lacking in
statistical power, biased ascertainment of subjects or controls, and
inadequate control for potentially confounding co-factors. The large
majority did not control for parental intelligence, which is the
variable most strongly associated with child IQ. These studies were,
however, important in suggesting an association between body lead
levels and performance, and also in demonstrating that there is an
association between social disadvantage and higher body lead levels.
8.3.4 Population-based cross-sectional studies on children
Most of the general population-based epidemiological
cross-sectional studies have, in addition to intelligence tests,
included extensive batteries of tests covering several other areas of
psychometric functioning, such as academic attainment, behaviour,
gross motor abilities and fine motor co-ordination, reaction time,
visuo-spatial skills and memory, and auditory memory. There are
differences in the tests used, and in the areas of functioning
investigated, and the results are less consistent and more difficult
to interpret than those relating to IQ tests. For that reason this
section will focus mainly on results relating to intelligence tests,
and results from other functional domains will be mentioned briefly
where appropriate.
There are many cross-sectional population studies using either
blood or teeth as the primary measure of lead body burden, and only
the more informative, in terms of providing information on risk
assessment, will be included. The majority of the studies described
used either a full or short form of the Wechsler Intelligence Scale
for Children - Revised (WISC-R or its translations) as the measure of
intelligence. Exceptions are the studies of Fulton et al. (1987),
which used the British Ability Scales (BAS), Harvey et al. (1988),
which used Wechsler Pre-school and Primary Scale of Intelligence
(WPPSI), and Schroeder et al. (1985) and Hawk et al. (1986), where the
Stanford-Binet Intelligence Scale was used.
8.3.4.1 Tooth lead studies
a) Needleman et al. (1979)
A total population of 3329 children aged 6-7 years, attending
schools in two working class towns near Boston, USA, were asked to
donate their shed deciduous teeth. Of these 70% (2335) children did
provide teeth. A 1-mm slice of tooth consisting primarily of dentine,
was split in half, and one portion was analysed by ASV. Children whose
initial tooth slice was in the highest 10th percentile (> 20 µg/g) or
the lowest 10th percentile (< 10 µg/g) were provisionally classified
into a high or low lead group. A total of 524 children were originally
classified into these lead groups, but 254 of these children were not
tested for a number of reasons, and the results for another 112 were
excluded from data analysis for medical reasons, or because a second
tooth analysis provided a discordant result. The results for 158
children (58 high lead children and 100 low lead children) were
presented. Thirty-nine non-lead variables were scaled and coded, and
those which were found to differ between the lead groups at the 10%
level were controlled as covariates. The covariates controlled were
father's socioeconomic status, mother's age at subject's birth, number
of pregnancies, mother's education and Peabody Picture Vocabulary
score. After this was done the children in the high lead group
performed significantly less well on the IQ test with a 4´ point
difference between the groups.
b) Winneke et al. (1983)
A total of 115 children aged 7-12 years living in the vicinity of
a lead/zinc smelter in Stolberg, Germany, who had provided at least
one incisor tooth, were investigated. Whole tooth lead measures ranged
from 2 to 32 µg/g, with a geometric mean of 6 µg/g. There was a
marginally significant negative association between lead level and
(short-form) verbal IQ after taking age, gender, perinatal risk
factors and social confounding factors into account. There was a
difference of 4.6 IQ points when children with tooth lead levels above
10 µg/g were compared with those with levels below 4 µg/g. Significant
negative associations were also observed between tooth lead and the
error score of Bender Gestalt shape copying test and performance on
the Vienna reaction time device.
c) Smith et al. (1983); Pocock et al. (1987)
The target population for the study by Smith et al. (1983) (also
known as the Institute of Child Health/University of Southampton
study) was 6875 children aged 6-7 years attending schools in one of
three areas in or near London. In all, 7407 teeth were donated by 4105
children. Whole tooth analysis of intact incisor teeth donated by
eligible children was carried out and children whose tooth lead level
(expressed on an ashed weight basis) was in the lowest percentile
(< 2.5 µg/g), approximately at the 50th percentile (5-5.5 µg/g) or
above the 90th percentile (> 8.0 µg/g) were invited to participate.
The sample was stratified by social grouping. Of the 432 children
selected, 403 participated in the study. Parental interviews were
carried out in the home, and information on a wide range of social and
familial variables was collected. There was a 5 point difference in IQ
scores between the high and low lead groups before controlling for any
covariates, but this was reduced to 2 points once covariates
associated with IQ score were controlled. The authors concluded that
social factors explain the differences in test performance to such a
degree that the small differences that remain may be due to other
social factors not measured. The study underlined the importance of
the social environment for the child's performance and recognized the
importance of the relationship between body lead burden and social
disadvantage.
Blood samples were taken from 93 children 3-6 months after
shedding a tooth and after psychometric testing was completed. The
range of PbB levels in this group was 0.336-1.296 µmol/litre
(7-27 µg/dl), except for one measurement of 2.064 µmol/litre
(43 µg/dl), and the mean was 0.614 µmol/litre (12.8 µg/dl). The
correlation of PbB and PbT was 0.5.
A reanalysis of this data by Pocock et al. (1989) employed
multiple regression techniques, using an "optimal" regression
strategy. The results were comparable to those previously reported.
Interactional analyses showed no evidence of any differential
association with lead in different social groups, but revealed a
significant interaction between PbT and child gender. Significant
negative association between PbT and IQ was found in boys but not in
girls.
d) Fergusson et al. (1988a,b,c)
This study was carried out in Christchurch, New Zealand, on a
sub-sample of a birth cohort of 1265 children born in 1977. Teeth were
collected from 1035 children, and a dentine "chip" was analysed to
provide a PbT estimate for 996 children. The mean dentine lead level
was 6 µg/g. Children from disadvantaged backgrounds had higher mean
lead levels than those from more advantaged backgrounds, and this
difference persisted when other social environmental factors (such as
the effects of housing, proximity to main roads, and childhood pica)
were taken into account. A WISC-R and reading test were administered
to children at ages 8 and 9 years, and results were given for 724
children at age 8 and 644 children at age 9. Small correlations
between IQ scores and dentine lead were found, but control for
confounding variables reduced these to statistical non-significance
for both ages (a correlation of -0.03 to -0.05). Parental IQ was
assessed. Control for pica as a test of reverse causality further
reduced the associations between outcomes and PbT.
e) Hansen et al. (1989); Lyngbye et al. (1990a,b); Grandjean et al.
(1991)
Children entering schools in the town of Aarhus, Denmark, were
asked to donate shed teeth. Of a potential sample of 2414 children,
1291 (54%) provided usable teeth. A sample of circumpulpal dentine was
obtained from each tooth and analysed by AAS. The mean lead level in
dentine was 10.7 µg/g, with a log normal distribution and a range from
0.40 to 168 µg/g. The correlation of whole tooth lead and circumpulpal
dentine measures were low. Average circumpulpal dentine levels were
about five times higher than average whole tooth measures. Subsequent
analysis of PbB from samples obtained two to three years later showed
an overall mean PbB level of 0.25 µmol/litre (5 µg/dl), with a
geometric mean of 0.28 µmol/litre (range 0.08-0.63 µmol per litre or
1.66-13.1 µg/dl) in children selected for the high lead group, and a
mean of 0.18 µmol/litre (range 0.08-0.70 µmol/litre or mean
3.74 µg/dl, range 1.66-14.56 µg/dl) in children selected for the low
lead group, which confirms this as a low exposure group. Children
whose level of lead in circumpulpar dentine was above 18.7 µg/g (8%,
N = 110) were selected for the high lead group, and they were matched
on gender and socioeconomic status (SES) with children whose
circumpulpar dentine levels were below 5 µg/g. A comprehensive list of
exclusions, including neurological and medical conditions, atypical
social situations, and those who did not wish to take part, reduced
the sample to 156 children for whom results were presented. Initial
analysis (by matched pair t-test) showed a difference of 6 IQ points
on full-scale WISC scores, with a difference of nearly 9 points in
verbal IQ scores. The association of IQ measures with circumpulpar
dentine lead levels was evident across different socioeconomic groups.
Confounders associated with circumpulpar dentine lead levels (such as
pregnancy variables, SES, mother's educational status and child's
gender) were entered into a stepwise regression model, and explained
only a small portion of the variance. Lead levels in the dentine
accounted for a significant (2.8%) of the total variance in full scale
IQ. Parental IQ was not controlled.
f) Rabinowitz et al. (1991)
From 764 eligible children (attending grades 1 to 3 in 7 selected
schools in Taiwan), 940 deciduous teeth were collected, the majority
being incisors. The average age of students was 6.7 years. Two of the
schools were near lead smelters. Lead levels were determined in 862
teeth from 692 children using the Boston method which analysed a
"dentine chip" (Rabinowitz et al., 1989). The mean value was 4.3 µg/g
of dentine. In all, 493 children were tested using the Ravens Coloured
Progressive Matrices Test (CPM) and the IQ equivalents were related to
tooth lead levels. Confounders were selected from a set of 40, based
on associations with lead and IQ. After correction for seven
confounding variables (social and perinatal factors), the highly
significant raw exposure-effect association (r = -0.32) was markedly
reduced for the full sample to statistical insignificance, although a
significant lead-CPM association remained for the subsample of girls
(b = -1.8; SE = 0.78; P < 0.02).
8.3.4.2 Blood lead studies
a) Winneke et al. (1985)
In a further study by this group carried out in the lead/zinc
smelter town of Nordenham, Germany, 114 out of 378 children (46%) for
whom cord PbB levels were available were tested at 6-7 years of age.
The range of cord PbB levels was 0.19-1.49 µmol/litre (4-31 µg/dl) and
for current PbB levels was 0.19-1.10 µmol/litre (4-23 µg/dl); the
means for both perinatal and current PbB levels were 0.39 µmol/litre
(8.2 µg/dl, SD = 1.6). After correction for confounders by means of a
stepwise multiple regression analysis, few significant associations
between PbB levels and performance were observed. There was a marked
influence of social background factors, but little influence on
short-form full IQ score (current PbB accounted for 0.3% additional
variance), and a borderline decrease in performance IQ (PbB accounting
for 2.4% of the variance). In general, the associations were rather
stronger with current PbB levels than with cord PbB measures. The
strongest associations were found with the error scores of the
difficult version of the Vienna reaction time task.
b) Harvey et al. (1984)
From 483 eligible children from the city of Birmingham, United
Kingdom, 187 out of 284 contacted were examined at 30 months of age.
The average PbB level from venous samples was 0.75 µmol/litre
(15.6 µg/dl, SD = 4). Cognitive ability was assessed by means of four
tests from the British Ability Scales (BAS) and three tests from the
Stanford-Binet Intelligence Scale. The raw correlation between PbB
level and IQ was -0.17 (P < 0.05). Subsequent multiple regression
analysis with a predetermined set of confounders and a sample size of
48 yielded an insignificant association between lead and IQ.
c) Harvey et al. (1988)
In a second study, 201 out of 337 eligible children, aged 5.5
years, from the inner city area of Birmingham, United Kingdom, were
studied. The mean PbB level was 0.614 µmol/litre (12.8 µg/dl, SD = 4).
No significant associations between PbB level and IQ (WPPSI) were
observed after correction for confounding factors. Apart from some
statistically significant associations between PbB level and reaction
time, none of the remaining tests showed a significant correlation
with PbB level after confounder-control.
d) Yule et al. (1981)
Children (N = 166), aged 6 to 12 years, living in the vicinity of
a leadworks in outer London were examined. PbB levels from venous
samples ranged from 0.336 to 1.58 µmol/litre (7 to 33 µg/dl, geometric
mean = 0.648 µmol/litre or 13.5 µg/dl). After controlling for social
class as the only confounder, significant negative associations were
found for IQ (WISC-R; full-scale- and verbal-, but not performance-
IQ). An average IQ difference of 7 points was found when comparing
children with PbB levels of 0.62 µmol/litre (13 µg/dl) or more with
those of 0.576 µmol/litre (12 µg/dl) or less. Significant inverse
associations were also found between PbB level and scores on tests of
attainment, i.e. reading and spelling but not mathematics.
A group of 9-year-old children from a middle class area of London
were studied. Subjects were divided into low PbB (N = 80 and PbB of
0.336-0.576 µmol/litre, 7-12 µg/dl) and high PbB (N = 82 and PbB of
0.624-1.15 µmol/litre, 13-24 µg/dl) groups for testing the association
between PbB level and IQ. No significant association between PbB level
and IQ was found before or after controlling for covariates (Lansdown
et al., 1986).
e) Schroeder et al. (1985)
One hundred and four children from a high risk population, aged
10 months to 6.5 years and with PbB levels ranging from 0.288 to
2.83 µmol/litre (6-59 µg/dl) were tested for intellectual development
using age-appropriate tests. After control for social confounding a
significant negative association was found between PbB level and IQ.
Fifty children of this cohort were reassessed 5 years later when all
PbB levels were below 1.44 µmol/litre (30 µg/dl). Neither initial nor
current PbB level was significantly related to later IQ.
f) Hawk et al. (1986)
In an effort to replicate the above-mentioned findings, 75
children from the same group, then aged 3 to 7 years, and having a
mean PbB level of 0.998 µmol/litre (20.8 µg/dl), range
0.29-2.256 µmol/litre (6-47 µg/dl), were examined using the
Stanford-Binet test. There were no significant interactions between
PbB level and factors including age, sex, maternal IQ, quality of the
care-giving environment (HOME), or socioeconomic status. There was a
statistically significant negative association between PbB level and
IQ, although the characteristics of the regression coefficient for
lead depended on which covariates were included in the model. For the
final regression model that best controlled for confounding with the
greatest precision, i.e. the most accurate and precise model,
containing lead, maternal IQ, HOME and gender, highly significant
inverse associations were found between both mean and maximum PbB
levels and IQ.
g) Fulton et al. (1987)
Five hundred and one children, aged 6-9 years, out of 1210
eligible children from 18 primary schools in central Edinburgh,
Scotland, were tested for an association between PbB level, IQ and
attainment. The sample comprised all children in the top quartile of
the PbB distribution, and a random subsample (approximately 1 in 3) of
the remainder. The geometric mean PbB level was 0.55 µmol/litre, range
0.16-1.63 µmol/litre (11.5 µg/dl, range 3.3-34.0 µg/dl). After
correction for 31 potential confounders a significant negative
association between PbB level and IQ (BAS) was found. There was a
dose-response relation with no evidence of a threshold. For purposes
of statistical analyses, children were placed into 10 groups of about
50 each on the basis of PbB level. The mean PbB level was
0.27 µmol/litre (5.6 µg/dl) in the lowest and 1.06 µmol/litre
(22.1 µg/dl) in the highest group; there was a difference of 5.8 IQ
points between the two groups. The size of the effects of lead was
small relative to that of the other factors (0.9% of a total of 45.5%
explained by the full model).
h) Hatzakis et al. (1989)
Five hundred and thirty-three children out of 1038 eligible from
four primary schools in the vicinity of the old lead-mining and
lead-smelting industrial complex of Lavrion in Greece were examined
neuropsychologically. The mean PbB level from venous samples was
1.14 µmol/litre, range 0.355-3.067 µmol/litre (23.7 µg/dl, range
7.4-63.9 µg/dl). In order to control for confounding factors, several
regression models were evaluated. Starting with a set of 24 potential
confounders, an optimal model containing 17 potentially confounding
variables was finally developed for testing the PbB/IQ association.
The continuous PbB variable was divided into five equally wide classes
from equal/below 0.715 µmol/litre (14.9 µg/dl, low) to equal/above
2.16 µmol/litre (45.0 µg/dl). After correction for confounders, a
highly significant negative association was found between PbB level
and IQ (WISC-R). A persistent decrease in IQ was only observed at PbB
concentrations above 1.2 µmol/litre (25 µg/dl). The adjusted
full-scale IQ difference between "high" and "low" PbB children was 9.1
points. Highly significant negative associations with PbB were also
found for the Bender Gestalt test and the Vienna Reaction Device,
without any indication of a threshold.
i) Silva et al. (1988)
Five hundred and seventy-nine socioeconomically advantaged
11-year-old children with an average PbB level of 0.53 µmol/litre
(11 µg/dl), range 0.19-2.4 µmol/litre (4-50 µg/dl) were tested in
Dunedin, New Zealand. Because the crude inverse association between
blood lead and IQ was not statistically significant, subsequent
multivariate analyses were not carried out.
j) European multicentre study
The results from a multicentre study in Europe were reported by
Winneke et al. (1990). The study linked eight institutions from eight
European countries; four of the individual study groups were from
areas near smelters and the others were more general populations. A
common study protocol with inherent quality assurance elements was
developed to achieve comparability between the individual studies. In
all, 1879 children, aged 6-11 years, were studied; however, for some
of the tests the sample size was reduced to only 971 children. The PbB
level ranged from 0.24 to 2.88 µmol/litre (5 to 60 µg/dl). Overall
statistical evaluation was done using a uniform predetermined
confounder model containing age, gender, occupational status of the
father, and mother's education. The inverse association between PbB
level and IQ (four subtests from the WISC-R) was of only borderline
statistical significance. An IQ decrement of about 3 points was
calculated for a PbB increase from 0.24 to 0.96 µmol/litre (5 to
20 µg/dl). The associations between the error scores on the Bender
Gestalt Test and the Vienna Reaction Device were statistically
significant and more consistent across study groups, although the
outcome-variance explained that lead never exceeded 0.8%. The data did
not allow for the identification of a threshold.
8.3.4.3 Follow-up studies
a) Bellinger et al. (1984)
A follow-up of some of the children investigated by Needleman et
al. (1979) was conducted when the children were approximately eleven
years old. Twenty-two of the original elevated lead group and 48
children from the low lead group were traced, as well as a group of 52
children who had not been tested previously, but whose lead levels in
a "dentine chip" (from teeth analysed at the time of the previous
study) fell into the mid-range between the high and low lead groups.
Children in the elevated lead group were significantly younger than
those in the other two groups. Information on IQ from group-
administered Otis Lennon Mental Ability tests, conducted 1 to 2 years
previously, was collected from school records for 15, 52 and 34
children from the elevated, mid-range and low groups, respectively.
Scores were inversely related to previously obtained lead measures,
with a 7-point difference in mean scores between the high and low lead
groups (t-ratio = 1.65, P = 0.1). No current measures of lead status
were available.
b) Winneke et al. (1989)
Out of 114 children first tested at age 6 (Winneke et al., 1985)
76 were retested at age 9. The range of PbB levels had been
0.187-1.09 µmol/litre, geometric mean = 0.39 (3.9-22.8 µg/dl,
geometric mean = 8.2) at age six and was 0.21-1.027 µmol/litre,
geometric mean 0.37 (4.4-21.4 µg/dl, geometric mean 7.8) at age 9
(r = 0.82). After correction for the same set of confounders used at
age 6 (age, gender, social background), most of the findings that had
been found to be significant at age 6, i.e. those related to reaction
performance (Vienna Reaction Device), remained virtually unchanged
three years later. The authors concluded that this observation cannot
necessarily be taken to indicate persistence, because internal
exposure had essentially remained unchanged over three years.
c) Needleman et al. (1990)
A follow-up assessment was made of 132 young adults first
investigated by Needleman et al. (1979) at the age of 6-7. Those
retested were not representative of the group of 270 tested 11 years
earlier, as they had slightly lower childhood dentine lead levels,
came from higher SES families, and were more likely to be female.
Current PbB level was assessed from 48 subjects and none was above
0.336 µmol/litre (7 µg/dl). A behavioural evaluation including a word
reading test, and a neurobehavioural assessment was administered to
each subject, and a self-report of delinquency was obtained. In
addition school records were reviewed. Multiple regressions indicated
that higher levels of dentine lead in childhood was associated with
lower reading scores, lower class rank, increased absenteeism, and
poorer performance on some of the neurological tests in young
adulthood. Comparing subjects with dentine lead levels greater than
20 µg/g with the lowest lead group, children in the high lead group
were much more likely to drop out of school (unadjusted odds ratio
4.6; adjusted odds ratio 7.4, CI 1.4-40.8) and to have a reading
disability (unadjusted odds ratio 3.9; adjusted odds ratio 5.8, CI
1.7-19.7). Adjustment for covariates made little impact on most of the
outcomes.
8.3.4.4 Conclusions and limitations of cross-sectional studies
A number of general issues are evident from looking at the
cross-sectional studies as a group. In most studies a negative
association between lead measures and IQ measures is found in
uncontrolled data. This difference is usually in the range of 4 to 6
IQ points and most marked in verbal IQ. Most studies also confirmed
the positive association between lead measures and indicators of
social disadvantage, whether this was indicated by SES, maternal
education, or other more detailed indicators of non-optimal
child-rearing environments, such as marital quality or maternal
depression. When these social and other confounding factors are
controlled, the effect has, in most cases, been to reduce the strength
of the association between lead measures and IQ, although it remains
in the same direction. When maternal intelligence has not been
controlled, the impact on the association between lead and IQ measures
of correction for covariates tends to be smaller. Where more detailed
social measures have been controlled, the impact on the magnitude of
the lead-IQ association has been greater. Associations have, in
general, been stronger with verbal IQ than with performance IQ.
The cross-sectional nature of these studies, and also the fact
that many used a single measure of current exposure, limited their
usefulness in answering questions relating to the natural history of
the association between lead exposure and outcomes, including whether
there were critical periods of exposure, and whether lead associated
deficits were persistent or reversible.
Findings from follow-up studies are difficult to interpret.
Needleman et al. (1990) interpreted their findings as indicating the
persistence of the effects of lead. Identifying persistence or
irreversibility in this context has many problems, due to the
stability of a number of other (non-lead) factors. In addition,
sampling biases, or failure to control for important covariates in the
initial or follow-up stages may lead to apparent persistence of an
effect. A further difficulty is that it is unusual to find a cohort
where exposure profiles permit easy assessment of persistence.
Neurobehavioural effects detected at age seven or later usually
persist in later follow-up studies conducted in other (non-lead) areas
of research, and for this reason it is more likely than not that
lead-associated deficits detected during childhood will be detectable
later. However, there are not enough data to conclude that this is the
case. Animal data, in which persistence of effects has been shown
after cessation of exposure, do provide support for the
irreversibility of lead-induced behavioural toxicity.
8.3.5 Prospective epidemiological studies on children
8.3.5.1 Common elements
The international prospective cohort studies shared a common
design of pre- or perinatal recruitment, with the principal measure of
prenatal exposure being the lead concentration in whole blood taken
antenatally or at birth. To facilitate later comparison of results,
meetings have been held at which common protocols (within the
constraints of local conditions) were agreed (Bornschein & Rabinowitz,
1985; Smith, 1989).
The protocols were similar in terms of the instruments of
neurobehavioural assessments, which were well standardized and
validated for the populations to which they were applied, and although
the frequency of assessment varied between studies, it was agreed that
all studies would make an assessment during infancy, in the later
pre-school period and, if possible, in the school age years.
All studies conducted several assessments of PbB concentration
but at varying frequency, and all studies participated in some form of
quality assurance/quality control (QA/QC) programme for the assessment
of lead exposure. QA/QC procedures were also used to establish
inter-examiner reliability (where more than one examiner was used) for
psychometric and covariate assessments. The importance of such
procedures was recently discussed (SAHC, 1993).
In assessing dose-effect relationships between lead exposure and
outcome variables, most studies used several indices of exposure,
including both measures of PbB concentration at particular ages and
average measures of exposure within the lifetimes of the study
subjects.
All studies used data analysis procedures which took simultaneous
account of covariates selected for statistical and substantive
considerations.
8.3.5.2 Study descriptions
a) Boston study
The study population was recruited from 11 837 infants born at
the Brigham and Women's Lying-In hospital in Boston between April 1979
and April 1981 (Bellinger et al., 1984). On the basis of cord PbB
levels of approximately 2500 children, the 90th, 50th and 10th
percentiles were identified and used as eligibility criteria for
enroling 249 children in three exposure groups: low (< 0.14 µmol/litre
or 3 µg/dl, N = 85), medium (0.288-0.336 µmol/litre or 6-7 µg/dl,
N = 88) and high (> 0.48 µmol/litre or 10 µg/dl, N = 76). No child
had a cord PbB level greater than 1.2 µmol/litre (25 µg/dl).
Exclusion criteria included birth complications or medical
conditions associated with developmental difficulties (e.g.,
gestational age < 34 weeks, Down's syndrome), a non-English speaking
family or residence in an area considered unsafe for home visitors.
The cohort consisted largely of children from intact middle and
upper-middle class families. Approximately 85% of the children were
white.
Capillary blood collected at ages 6, 12, 18 and 24 months was
analysed for lead by ASV. Venous blood collected at ages 57 months and
at 10 years was analysed for lead by AAS. In contrast to other
prospective cohorts, the mean PbB level did not rise postnatally and
indeed never exceeded 0.384 µmol/litre (8 µg/dl).
The principal psychometric tests included the Bayley Scale of
Infant Development at 6, 12, 18 and 24 months, the McCarthy Scales of
Children's Abilities at 57 months and the Wechsler Intelligence Scale
for Children - Revised and the Kaufman Test of Educational Achievement
at age 10 years. After age 2 years, the mean level of performance was
above the expected population mean (e.g., mean full scale IQ at age 10
years = 119) (Bellinger et al., 1992).
A variety of potential confounding factors was assessed including
maternal IQ, the quality of rearing environment (HOME scores at 6, 24,
57 and 120 months) and prenatal, perinatal and postnatal medical and
socio-demographic factors.
b) Cincinnati study
Women attending prenatal clinics in predesignated leadhazardous
residential areas of Cincinnati, Ohio, USA, were consecutively
recruited from 1979 to 1984 (Dietrich et al., 1987) Prenatal
exclusions were for maternal prenatal alcohol and drug abuse,
psychosis, diabetes, mental retardation; neonatal exclusions included
birth weight under 1.5 kg and gestational age (by physical
examination) below 35 weeks. Furthermore, recruited infants had to
have an Apgar score of 6 or more at 5 min postpartum and to have no
serious medical conditions. Of the mothers, 87% were single and 86%
were receiving public assistance. The final follow-up sample of 305
infants who attended their second clinic visit at 3 months of age was
85% African-American and 50% female.
Mean prenatal maternal PbB levels as assessed by ASV were
0.38 ± 0.177 µmol/litre (8.0 ± 3.7 µg/dl). Mean neonatal PbB levels
were also low at 0.221 ± 0.134 µmol/litre (4.6 ± 2.8 µg/dl). Following
the development of prewalking progression and normal hand-to-mouth
behaviours, PbB levels began to rise, peaking at around 21 months with
a mean of approximately 0.89 µmol/litre (18 ± 6 µg/dl). Approximately
35% of the sample had at least one PbB concentration equal to or
greater than 1.2 µmol/litre (25 µg/dl) sometime during the first 5
years of life, while 79% exceeded 0.72 µmol/litre (15 µg/dl) during
the same period (Dietrich et al., 1991, 1992). Virtually all children
(95%) exceeded 0.48 µmol/litre (10 µg/dl) during the first 5 years of
life. Postnatal PbB concentrations were assessed on a quarterly basis
beginning at 10 days postpartum. The vast majority of blood samples
were collected by venepuncture.
Major neurobehavioural assessments included the Bayley Scales of
Infant Development administered at 3, 6, 12 and 24 months of age. The
Kaufman Assessment Battery for Children was administered at 4 and 5
years. A comprehensive examination of gross and fine neuromotor
functions (the Bruininks-Oseretsky Scales) was administered at 6
years. Finally, performance on the Wechsler Intelligence Scale for
Children - Revised was assessed following school entry at 6.5 years.
Intellectually, this was a low functioning cohort with a mean
full-scale IQ at 6.5 years of 86.9 ± 11.3 points (Dietrich et al.,
1993a).
Covariates measured included assessments of obstetrical and
perinatal complications, birth anthropometrics, tobacco and alcohol
consumption, observational assessments of the quality of care-taking
in the home at 6, 12, 24 and 36 months, social class, iron status and
various aspects of child health likely to affect neurobehavioural
performance (e.g., otitis media, sensory deficits, allergies, seizure
disorders). Maternal IQ was assessed postnatally. All medical and
psychometric testing was conducted at a single welfare clinic located
in the heart of the recruitment area.
c) Cleveland
Five hundred and forty-three infants delivered to women at a
large inner city general hospital between February 1981 and March 1982
were considered for inclusion in this study of the joint or
independent effects of fetal alcohol exposure and lead on child
development (Ernhart et al., 1985). Due to pre-term birth or illness,
16% were excluded from further study. Also excluded were mothers
reporting the use of narcotics and those with identifiable
psychological disorders. The sample was predominantly white (65%) and
of lower SES. Approximately 50% of mothers were alcoholics.
PbB concentrations were assessed by AAS (208 mothers at delivery
and in 178 cords resulting in 142 mother-infant pairs with complete
prenatal lead exposure data). Postnatally, blood samples were analysed
for approximately half the birth cohort at ages 6 months, two years
and three years of age. At least one perinatal or postnatal PbB
concentration was available for each of 285 children with
developmental data past the neonatal period (Ernhart et al., 1987,
1989a,b).
Mean maternal PbB concentrations (N = 185) were low
(0.31 ± 0.086 µmol/litre or 6.5 ± 1.8 µg/dl) and mean cord PbB
concentrations (N = 162) were also low (0.29 ± 0.10 µmol/litre or
6.0 ± 2.1 µg/dl). Postnatally, mean PbB concentrations (N = 151) were
0.48 ± 0.16 µmol/litre (10.0 ± 3.3 µg/dl) at 6 months,
0.80 ± 0.28 µmol/litre (16.7 ± 5.9 µg/dl) at 2 years (N = 165) and
0.80 ± 5.9 µmol/litre (16.7 ± 5.9 µg/dl) at 3 years. The lowest and
highest PbB levels recorded postnatally were 0.24 and 2.016 µmol/litre
(5 and 42 µg/dl), respectively.
Neurobehavioural assessment included selected sub-scales of the
Brazelton Neonatal Behavioural Assessment Scales and Graham Rosenblith
Behavioural Examination of the Neonate at > 24 h postpartum, the Kent
Infant Development scale at 6 months, the Bayley Scales of Infant
Development at 6, 12 and 24 months, the Stanford-Binet Intelligence
Scale at 3 years, and finally the Wechsler Preschool and Primary Scale
of Intelligence at approximately 5 years of age. All assessments
beyond the neonatal period were conducted in subjects' homes.
Intellectual attainment in this high risk, socioeconomically
disadvantaged cohort was typically low as shown by a mean full-scale
IQ at approximately 5 years of 87.5 ± 16.6 points (Ernhart et al.,
1989a).
In addition to the usual assessments of obstetrical/perinatal
complications and neonatal status, other covariates measured included
the Michigan Alcoholism Screening Test, the Peabody Picture Vocabulary
Test as an assessment of maternal intelligence, and the Authoritarian
Family Ideology Scale. Women were also questioned as to their
consumption of alcohol and tobacco during pregnancy. Observational
assessments of care-taking quality in the home were conducted at 1, 2,
3 and 4 years.
d) Glasgow study
The study group for CNS outcomes consisted of 151 subjects drawn
from an initial sample of 885 families exposed to various levels of
dietary lead, principally due to a plumbosolvent water supply (Moore
et al., 1989). All subjects were born in the United Kingdom and spoke
English as a first language. The sample was divided into three groups
of approximately equal numbers (matched for social class) based on
maternal prenatal PbB levels: high (> 1.44 µmol/litre or 30 µg/dl),
medium (0.72-1.2 µmol/litre or 15-25 µg/dl) and low
(< 0.48 µmol/litre or 10 µg/dl). The SES of the sample ranged from
the chronically unemployed to the professional classes.
PbB levels were assessed postnatally at 1 and 2 years of age and
found to be 0.734 and 0.777 µmol/litre (15.3 and 16.2 µg/dl),
respectively.
Measurements of neurobehavioural outcome where the Bayley Scales
of Infant Development were used were administered at 1 and 2 years of
age.
Covariates considered in the data analysis included a measure of
obstetrical complications, birth weight, birth order, SES of the
father in postnatal follow-up years 1 and 2, and observational
assessments of care-taking quality in the first and second years of
follow-up.
e) Kosovo Study
Five groups of infants from two communities in Kosovo,
Yugoslavia, were studied (Graziano et al., 1990; Wasserman et al.,
1992). Mitrovica was the site of a lead smelter, refinery and battery
plant, and Pristina an area of minimal lead exposure. Three groups
were recruited from Mitrovica on the basis of cord PbB concentration:
(1) < 0.72 µmol/litre (< 15 µg/dl, N = 78); (2) 0.72-0.96 µmol/litre
(15-20 µg/dl, N = 99); (3) > 0.96 µmol per litre (> 20 µg/dl,
N = 217). Two groups were recruited from Pristina: (4) a group with
cord PbB levels < 0.72 µmol/litre (< 15 µg/dl) and (5) a group
matched to children in group (3) in terms of the distribution of
maternal and paternal education. Grouping was not considered in the
statistical analysis. The total cohort (N = 541) was largely Albanian
and Serbian. Infants with CNS defects, chromosomal anomalies, distant
residence and twin pairs were excluded.
PbB level and various indices of iron status (EP, Hb, serum
ferritin) were measured in venous blood collected in midpregnancy,
delivery and at postnatal ages 6, 12, 18 and 24 months. Mean PbB level
amongst children in Mitrovica exceeded 0.96 µmol/litre (20 µg/dl) at
all ages, reaching 1.68 µmol/litre (35 µg/dl) at 24 months. Amongst
children in Pristina, the mean PbB level was below 0.48 µmol/litre
(10 µg/dl) at all ages.
The Mental Scale of the Bayley Scales of Infant Development was
administered at ages 6, 12, 18 and 24 months, and the Motor Scale at 6
and 12 months. At 24 months, the mean Mental Development Index in the
cohort (n = 392) was 105.2 (SD = 18.1).
Anthropometric measurements and assessments of child and family
health, diet, and demographic status were carried out at each age. An
adaptation of the HOME Scale was administered at ages 3 and 4 years
and maternal IQ was assessed using the Raven's Standard Progressive
Matrices.
f) Port Pirie study
Port Pirie (population 16 000 in 1979) is an industrial town 200
km north-west of Adelaide, South Australia, with a large and
long-standing smelting facility (Wigg et al., 1988). Over the period
1979-1982, a cohort of 723 children from Port Pirie and surrounding
rural areas was recruited into the study. The cohort represented a 90%
sample of all children born in the area during this period.
Lifetime lead exposures were estimated from venous PbB
concentrations obtained antenatally (at 14-20 weeks of gestation, and
32 weeks), at delivery (maternal and cord blood), and from capillary
samples taken postnatally at ages 6, 15, and 24 months, and annually
thereafter. Lead concentrations in all samples were determined by AAS.
At the time that each blood sample was collected, the nurse
interviewer also conducted structured interviews to obtain information
on a range of demographic, familial, behavioural, medical and social
environmental factors. HOME scores were assessed when the child was 3
and 5 years of age. Maternal IQ was also assessed. The Bayley Scales
of Infant Development was administered at 2 years, the McCarthy Scales
of Children's Abilities at 4 years and the Wechsler Intelligence Scale
for Children - Revised version (WISC-R) at 7 years.
g) Sydney study
A total of 318 children was recruited from among infants born at
three maternal hospitals between April 1982 and March 1983 (Cooney et
al., 1989a). Exclusion criteria included prematurity, low birth
weight, severe medical problem, single or non-English speaking mother
or maternal drug or alcohol problems. All children were white and
generally represent a middle-class population.
PbB concentration was measured in maternal venous blood during
hospitalisation (geometric mean = 0.388 µmol/litre or 8.1 µg/dl,
geometric SD = 0.067 µmol/litre or 1.4 µg/dl). Additional blood
samples were collected at 6-month intervals to age 4 years and at ages
5 and 7 years. Up to 24 months of age, approximately 50% of the blood
samples were capillary, while almost all samples collected
subsequently were venous. Geometric mean PbB levels between 6 months
and 4 years were in the range of 0.48-0.72 µmol/litre (10-15 µg/dl),
falling to 0.39 and 0.37 µmol/litre (8.3 and 7.7 µg/dl) at ages 5 and
7, respectively.
The Bayley Scales of Infant Development were administered at ages
6, 12 and 24 months, the McCarthy Scales of Children's Abilities at
ages 3 (n = 215) and 5 (n = 200), and the Wechsler Intelligence Scale
for Children - Revised at 7 years (n = 175). All assessments, at least
until age 5, were conducted in the child's home. The mean Mental
Development Index and General Cognitive Index (GCI) scores were in the
intellectual normal range of 107 to 117 with a mean general cognitive
index at 4 years of 107.3 ± 14.2.
Covariates measured included maternal IQ, quality of care-taking
environment, obstetrical and postnatal factors.
8.3.5.3 Summary of differences between studies
The prospective studies differed to such an extent that it would
be surprising if they yielded identical findings. This does not mean
that the studies cannot be compared, merely that these differences and
their potential impact on study findings must be acknowledged in any
assessment of the studies as a group (Bellinger & Stiles, 1993). There
are substantial differences in the degree of confounding between lead
exposure and the other correlates of poor developmental outcomes. For
example, mean maternal IQ ranges from 121 (Boston) to 74-75
(Cleveland, Cincinnati), reflecting vast cohort differences in
socioeconomic standing and child-rearing context.
The studies also varied substantively in terms of the lead
exposure profiles observed. Site differences resulted in considerable
variability in postnatal lead exposures depending on exposure patterns
(residence near primary lead smelters, residence in deteriorated
housing, proximity to traffic, and quality of domestic
drinking-water).
Despite broad similarities in the approaches to data analysis,
there were substantial differences in the form in which the findings
were reported, impeding direct comparison of study results. Results
were diversely reported in terms of partial correlation coefficients
and standardized and unstandardized regression coefficients, and there
was variable use of raw (untransformed) and log exposure measures.
Most, but not all studies presented their results in a manner
conducive to some kind of quantification of dose-effects
relationships.
Clearly the variation in numbers of participants available to
individual studies (both initially, and as a consequence of attrition)
has a direct effect on the statistical power of each study - which is
also affected by the degree of confounding.
8.3.5.4 Results of studies
a) Infancy and early preschool assessments
The pattern of findings in infant and early preschool assessments
for which the principal instrument of assessment was the Bayley Scales
of Infant Development is inconsistent. Using different indices of
in utero exposure (e.g., prenatal, at delivery, cord or neonatal PbB
levels), three of the prospective studies found associations with
slower sensorimotor development up to 6 months or 1 year of age
(Ernhart et al., 1987; Dietrich et al., 1987, 1989) or up to 2 years
of age (Bellinger et al., 1987), but these tended to attenuate over
time (Dietrich et al., 1990; Bellinger et al., 1991a). Other studies
did not observe statistically significant adjusted relationships of
prenatal exposure with slower sensorimotor development (Moore et al.,
1989; Wigg et al., 1988; Cooney et al., 1989a,b; Wasserman et al.,
1992).
Two studies observed an inverse association between postnatal
lead exposure on mental development in later infancy (Wigg et al.,
1988; Wasserman et al., 1992), although in one study it was PbB level
at 6 months of age (Wigg et al., 1988) and in the other study it was
the level at 24 months (Wasserman et al., 1992) that was most
predictive.
b) Later preschool and school age assessment
Despite some inconsistency in the pattern of findings in infants,
there has been a convergence of positive findings on later
neurobehavioural outcomes in the prospective studies. This may
reflect:
1) the greater reliability and precision of measurement attained
with assessments of the older child; or
2) an effect of lead on abilities that cannot easily be tested
during infancy (e.g., executive, regulative and organizational
skills, higher order reasoning).
c) Boston study
Among 169 children assessed at age 57 months, the major finding
was an association between PbB level at 2 years of age and the GCI of
the McCarthy Scales. There was a decrease of 2.95 ± 1.42 GCI units for
an increase in PbB of 0.48 µmol/litre (10 µg/dl). Since the group had
an average PbB level of 0.336 µmol/litre (7.0 µg/dl) over 2 years,
this decrement was relevant to a range of approximately
0.19-0.67 µmol/litre (4-14 µg/dl) Bellinger et al., 1991a).
At 10 years of age, 148 children were reassessed. PbB
concentration at 2 years of age was inversely associated with the
WISC-R Full Scale IQ. Each 0.48 µmol/litre (10 µg/dl) increase was
associated with a 5.8 point decline in IQ (95% CI 1.7 to 9.9)
(Bellinger et al., 1992).
PbB concentration at 24 months was also inversely related to the
Battery composite score in the Kaufman Test of Educational Achievement
(Brief form) (-0.89 ± 0.24). The skills assessed were mathematics,
reading and spelling.
The point estimates associated with other postnatal ages were
generally in the same direction but only PbB level at 2 years revealed
conventional statistical significance. Other specific
neuropsychological tests did not reveal a clear pattern of
neuropsychological deficit (Stiles & Bellinger, 1993).
d) Cincinnati study
The Kaufman Assessment Battery for Children was administered to
approximately 260 children at 4 and 5 years of age (Dietrich et al.,
1991, 1992). The principal findings at 4 years were that higher
neonatal PbB levels were associated with poorer performance on all
Kaufman subscales. However, this inverse association was limited to
children from the poorer families. Following full covariate
adjustment, few statistically significant associations between
postnatal PbB levels and Kaufman scales could be found. However, the
results did suggest a weak relationship between postnatal PbB levels
and performance on a Kaufman subscale which assesses visual spatial
and visual-motor integration (adjusted regression coefficient -0.12 SE
not published).
At 5 years of age, postnatal PbB levels were associated with
poorer performance an all subscales of the Kaufman Battery. However,
after adjustment for covariates, few statistically significant
relations remained. Nevertheless, as at 4 years of age, the subscale
assessing visual-spatial and visual-motor skills was most sensitive,
with average lead exposure during the 4th year of life being
significantly associated with performance (-0.12 units per
0.048 µmol/litre or per µg/dl, SE not published).
At the age of 6.5 years, 253 children in the Cincinnati cohort
were administered the WISC-R (Dietrich et al., 1993a). The major
findings were that postnatal PbB concentrations were inversely
associated with full-scale IQ and performance IQ. Following
statistical adjustment for covariates, including maternal IQ and
quality of home care, a statistically significant relationship was
retained between postnatal PbB concentrations at nearly all ages (and
including lifetime averages) and Performance IQ. Further analysis
revealed that average lifetime PbB concentrations in excess of
0.96 µmol/litre (20 µg/dl) were associated with deficits in IQ of the
order of 7 points, when compared with children with mean
concentrations of less than 0.48 mol/litre (10 µg/dl). The regression
coefficient for Performance IQ on mean lifetime PbB level was -0.26
(± 0.12) IQ units per 0.048 µmol/litre or per µg/dl. This corresponded
to a 2.6 unit decrement associated with a 0.48 µmol/litre (10 µg/dl)
movement in lifetime average PbB level, for which the average
(interpolated graphically) is close to 0.67 µmol/litre (14 µg/dl).
At 6 years of age, the Bruininks-Oseretsky Test of Motor
Proficiency (BOTMP) was administered to 245 children in the Cincinnati
Cohort (Dietrich et al., 1993a). Following statistical adjustment for
covariates, neonatal PbB levels were associated with poorer
performance on a measure of upper-limb speed and dexterity and the
fine-motor composite. Postnatal PbB levels also remained significantly
associated with poorer scores on measures of bilateral coordination,
visual-motor control, upper-limb speed and dexterity, and the fine
motor composite.
Children having an average mean lifetime PbB level of
approximately equal to or exceeding 0.43 µmol/litre (9 µg/dl) appeared
to experience a deficit in both fine and gross motor skills relative
to children in the lowest PbB quartile. Children in the highest
average lifetime PbB quartile had scores on the gross-motor subtest
assessing bilateral coordination of approximately 0.5 standard
deviations (2.5 points) lower than their counterparts in the lowest
quartile. Children in the highest average lifetime quartile also
scored more poorly in the fine-motor functioning, having scores of
approximately 0.6 standard deviations lower (6.3 points) than those in
the lowest quartile.
e) Cleveland study
The WPPSI was administered to 242 children at the age of 4 years
and 10 months (Ernhart et al., 1989a). Statistically significant
correlations between IQ (full scale and subscales) and PbB level
measured pre- and perinatally and at ages 2 and 3 years (and a
lifetime postnatal average) became non-significant after adjustment
for covariates. No estimates for effect size were presented in the
original report (Ernhart et al., 1989a).
f) Glasgow study
No neurobehavioural assessments were carried out beyond 2 years
of age (Moore et al., 1989).
g) Kosovo study
No results beyond 2 years of age were reported (Graziano et al.,
1990; Wasserman et al., 1992).
h) Port Pirie cohort study
The principal finding at 4 years was an inverse association of
McCarthy Scales of Children Abilities (MSCA) scores (General Cognitive
Index (GCI), Perceptual Performance and Memory) with most indices of
lead exposure (McMichael et al., 1988). After adjustment for
covariates, the association of GCI was not signifi-cant at certain
specific ages. However, it remained significant for the integrated
postnatal average for which an effect size of 7.2 GCI points lost in
association with an increase of lifetime average PbB level from 0.5 to
1.5 µmol/litre (10.4-31.2 µg/dl) was estimated.
Between 7 and 8 years of age the IQ of 494 children was assessed
with the WISC-R (Baghurst et al., 1992). After adjustment for
covariates there was little association with pre- and perinatal lead
exposure assessments but significant decrements in full-scale IQ of
between 3.7 and 4.8 points (depending on age) for each (natural) log
unit increase in lifetime average PbB concentration were observed. The
estimated effect size for the lifetime average exposure up to 3 years
of age was a loss of 5.3 IQ points in association with an increase in
PbB level from 0.48 to 1.44 µmol/litre (10 to 30 µg/dl).
The Block Design subscale of the WISC-R, which tests spatial
abilities, exhibited the strongest association with lead exposure and
estimated effect sizes were stronger in girls than boys for both the
MSCA and WISC-R.
i) Sydney study
At the age of 4 years, 207 children were assessed with the
McCarthy Scales of Children Abilities (MSCA) and virtually no
significant associations with any measures of PbB were observed
(Cooney et al., 1989b).
Follow-up assessments in 175 children using WISC-R at age 7 also
yielded no associations (crude or adjusted) with blood lead history
(Cooney et al., 1991).
8.3.5.5 Questions prospective studies have not answered
A disappointment of the prospective studies was their inability
to reach any obvious consensus on the behavioural phenotype associated
with low-level lead exposure, or on age(s) of critical sensitivity.
This latter fact may reflect the phenomenon of intra-individual
"tracking", whereby an individual maintained approximately consistent
ranking with respect to his or her PbB concentration at any age, or it
may reflect the need for chronic exposure over extended periods in
order for causal effects to become apparent.
8.3.5.6 Attempting a consensus
As discussed previously, no clear delineation of an age (or age
range) of maximal sensitivity to lead exposure has emerged from the
prospective studies, and there is a diversity of reporting styles
which makes attempts to compare studies and establish a consensus of
opinion very difficult.
Nevertheless, it would appear essential to attempt some sort of
synthesis of studies performed so far, given that the social and
economic consequences of preventing an adverse health effect are
potentially enormous.
A meta-analysis by Needleman & Gatsonis (1990) concluded that the
overall pattern of results was most unlikely to have occurred by
chance. As an approach to the problem of obtaining the "best" estimate
of the effect of lead exposure on neuro-behavioural development, a
common estimate of the partial correlation between lead burden and IQ
was determined. However, in order to derive such an estimate with some
degree of certainty, it is essential to find a common outcome, and to
identify studies for which at least broadly comparable exposure
measures are available.
Schwartz (1993) has identified three prospective studies and five
cross-sectional studies that examined the association of lead exposure
as determined by PbB concentrations with full scale IQ. For the
prospective studies, the exposure measures used were 24-months PbB
level (Boston) and lifetime average up to 3 years of age (Cincinnati
and Port Pirie).
There was only one measure for each of the cross-sectional
studies. Despite the compromises necessary for such a comparison, the
estimated effect sizes for an increase in PbB level from 0.48 to
0.96 µmol/litre (10 to 20 µg/dl) for all seven studies lay between 1
and 6 IQ points lost (with five of the studies lying between 1 and 4),
and the associated 95% confidence limits only just embraced the value
zero for 3 of the 7 studies.
Thus, a broad consensus does emerge from a rough comparison. From
the estimates summarized by Schwartz (1993), a weighted mean for the
six coefficients accompanied by a standard error of 0.4 was estimated
to be 2.6 IQ units lost for an increase in PbB from 0.48 to
0.96 µmol/litre (10 to 20 µg/dl).
8.3.6 Task group overview and interpretation of prospective studies
on children
8.3.6.1 Rationale
As a complement to the narrative review, prepared by the Task
Group, a quantitative overview of the findings of the prospective
studies was considered to be a valuable means of assessing the overall
strength of evidence for an association between blood lead summary
measures and school age IQ.
While individual studies vary somewhat in their design and their
analysis strategies, their underlying objectives are closely related.
For any generally applicable conclusions on general population lead
exposure and its effect on intellectual attainment to be reached, some
consistency of evidence across studies is highly desirable.
Since any single study has considerable random error in
estimating a relationship, a quantitative overview (or meta-analysis)
can be a valuable means of defining what is the plausible magnitude of
statistical association between PbB measures and child IQ.
It seems appropriate, given the quite different types of study
design, to present two separate meta-analyses dealing with prospective
studies and cross-sectional studies, respectively.
Any such meta-analysis needs cautious interpretation. The caveats
in drawing causal inferences from statistical associations apply to
any meta-analysis just as they do to individual studies. Any increase
in statistical precision regarding the magnitude of association does
not eliminate the potential for bias in any observational study on
human populations.
8.3.6.2 The prospective studies
There are four prospective studies: Boston (Bellinger et al.,
1992); Sydney (Cooney et al., 1989a,b); Cincinnati (Dietrich et al.,
1993a); and Port Pirie (Baghurst et al., 1992) which presented
quantitative results in a form suitable for an overview (i.e. with
regression coefficients and standard errors). The Task Group
concentrated on school age assessment because:
a) the studies used the same outcome measure (WISC-R), which is
widely accepted and has been previously evaluated in cross-
sectional studies; and
b) intellectual assessment at primary school age (6-10 years) is,
perhaps, of greatest overall relevance in reaching a consensus on
the public health importance of childhood lead exposure.
Unfortunately, the Cleveland study did not publish findings in
the same quantifiable manner. The potential impact of this apparently
"negative" study on the overall evidence will be discussed in section
8.3.6.2(c).
a) Measures of exposure, outcome and association
All four studies have used blood lead as the measure of body lead
burden, but the timing and frequency of examinations has varied as has
the choice of summary measures (averages over time).
In a meta-analysis it seems appropriate to concentrate on
measures of association after adjustment for confounders. While there
is some variation in choice of confounders, the two key covariates
(mother's IQ and HOME scores) appear in all four studies.
The Port Pirie study used the logarithm of the PbB concentration
in the multiple regression analysis while the other three studies used
untransformed PbB concentration. This difference in statistical style
can be reconciled by converting the lead regression coefficients to
estimated changes in IQ for a specific increase in PbB concentration,
i.e. from 0.48 to 0.96 µmol/litre (10 to 20 µg/dl). This equals 10
times the regression coefficient (as published) for the Boston and
Cincinnati studies and log 2 times the regression coefficient for the
Port Pirie study.
It is recognized that the specific subscales of performance and
verbal IQ are of separate interest (e.g., the performance IQ has been
rather consistently shown to be more reliably associated with
postnatal lead exposure amongst the prospective studies (McMichael et
al., 1988; Bellinger et al., 1991a; Dietrich et al., 1991, 1992,
1993b). However, it appears more important to focus on full-scale IQ
as the primary outcome measure, otherwise concerns about post-hoc
selection may be raised. Such full-scale IQ was measured at age 6.5, 7
and 10 years in the Cincinnati, Port Pirie, Sydney and Boston studies,
respectively.
b) Display of individual study findings
Since most studies have not shown perinatal PbB level to be
predictive of intellectual performance, attention will be focused on
the various measures of postnatal PbB level. In fact, there are seven
such measures in each study that have been related to full-scale IQ in
the reports. Tables 21 and 22 summarize the magnitude of association
together with the standard error for all reported analyses, each after
adjustment for confounders.
Except for the earliest postnatal PbB values in the Boston and
Cincinnati studies, the adjusted associations between PbB level and IQ
are consistently negative (i.e. inverted). However, the "random noise"
in each study's analysis means that there is considerable variation in
levels of statistical significance. Hence, there is a need for a more
systematic overview combining the evidence from all studies.
c) A quantitative overview
The prime difficulty here is the different choices of PbB
summaries in the four studies. In undertaking a meta-analysis it would
have been better if all studies had essentially the same PbB measures
in their multiple regressions. Possible choices might have been (i) a
specific time point, e.g., 2 years or (ii) the means over a specific
interval, e.g., 0 to 5 years.
However, from the available analyses, some broadly similar
summaries can be chosen, as follows:
Table 21. Magnitude of association between blood lead and full-scale IQ
from four prospective studies
No. of Age at which blood lead Estimated change in
children was measured full-scale IQ a
Boston 148 year 1 0.0 (1.6)
year 1´ -1.2 (1.8)
year 2 -5.8 (2.1)
year 5 -2.6 (2.9)
year 10 -4.6 (5.2)
mean of years 2-5 -8.2 (2.8)
mean of years 2-10 -8.6 (3.4)
mean of 6 months to
10 years (unpublished) -5.7 (3.2)
Cincinnati 251 mean of year 1 0.1 (1.4)
mean of year 2 -0.2 (0.8)
mean of year 3 -1.3 (0.9)
mean of year 4 -1.5 (1.0)
mean of year 5 -2.3 (1.1)
mean of year 6 -3.3 (1.4)
mean of years 1-6 -1.3 (1.1)
Port Pirie 490 mean of first 15 months -2.8 (1.4)
mean of years 1-2 -3.2 (1.5)
mean of years 1-3 -3.3 (1.6)
mean of years 1-4 -3.2 (1.7)
mean of years 1-5 -3.0 (1.7)
mean of years 1-6 -2.8 (1.7)
mean of years 1-7 -2.6 (1.7)
Sydney 175 mean of years 1 and 2 -2.7 (2.8)
mean of years 3, 4 and 5 -1.9 (1.9)
year 7 -1.4 (1.7)
mean of years 1-7 -1.6 (2.2)
a From 0.48 to 0.96 µmol/litre (10 to 20 µg/dl) blood lead
(and its standard error) after adjustment for confounders
Table 22. Association between blood lead levels and full-scale
IQ from ten cross-sectional studies
No. of Estimated change in
subjects full-scale IQa
Lavrion, Greece 509 -2.7 (0.7)
Edinburgh, Scotland 501 -2.6 (0.9)
Greenwich, England 129 -5.6 (3.2)
European, multi-centre
Bucharest, Romania 301 -0.4 (2.6)
Budapest, Hungary 254 +0.8 (1.8)
Modena, Italy 216 +0.9 (4.2)
Sofia, Bulgaria 142 +2.2 (2.3)
Dusseldorf 1, Germany 109 -4.6 (4.5)
Dusseldorf 2, Germany 109 -3.9 (5.1)
Zagreb, Croatia 48 -1.5 (4.5)
a From 0.48 to 0.96 µmol/litre (10 to 20 µg/dl) blood lead
(and its standard error) after adjustment for confounders
i) There is some advantage in considering the mean PbB level over a
number of years, since it summarizes cumulative exposure and also
achieves a more reliable ranking of individuals than a single value.
The available long-term means are:
Boston: mean of 6 months to 10 years (D. Bellinger,
personal communication to the IPCS, 1993)
Cincinnati: mean of years 1 to 6
Port Pirie: mean of years 1 to 7
Sydney: mean of years 1 to 7
Although they differ in frequency and age range, these three
summaries should be very highly correlated with one another. The
resultant meta-analysis is displayed in Fig. 15. Each of the
individual study confidence intervals includes zero, indicating that
significance at P < 0.05 was not reached. However, the combined
evidence, weighting studies according to the inverse of their
variance, produces a weighted mean decrease in full-scale IQ of 2
points for a 0.48-µmol/litre (10-µg/dl) increase in PbB level, with
95% confidence interval from -0.3 points to -3.6 points (P = 0.01).
ii) An alternative approach is to consider PbB level at a specific
time or average over shorter intervals of time. For this approach, the
analyses of these studies are most comparable during the first three
years, as follows:
Boston: 2 year measure
Cincinnati: mean of year 3
Port Pirie: mean of years 1 to 3
Sydney: mean of years 1 and 2
This second meta-analysis is displayed in Fig. 16. The data here
support an inverse association more strongly than in Fig. 15, and the
combined evidence estimates a mean decrease of 2.6 IQ points for a
0.48-µmol/litre (10-µg/dl) increase in PbB level, with 95% confidence
interval from -1.2 points to -4.0 points (P < 0.001).
However, it should be noted that the estimates here are heavily
dependent on the choice of time points for PbB level. For instance, if
one instead chose:
Boston: 1.5 year measure
Cincinnati: mean of year 2
Port Pirie: mean of years 1 and 2
Sydney: mean of years 1 and 2
then the combined estimate is roughly halved in magnitude and is of
borderline significance.
One needs to recall that the other prospective study, carried out
in Cleveland (Ernhart et al., 1989a), has no equivalent data, but
reported no significant association. This study comprised about 150
evaluated children, and would influence the overall evidence in a less
significant direction if the data were able to be included.
8.3.6.3 A quantitative assessment of the cross-sectional studies
There have been more cross-sectional studies that have related
body burden to full-scale IQ in school-age children, but they cannot
all be included in a single meta-analysis. Here attention is focused
on the PbB studies, and it would be appropriate subsequently to
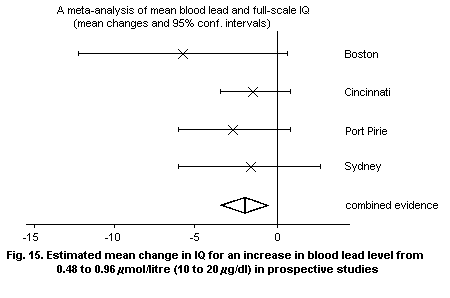
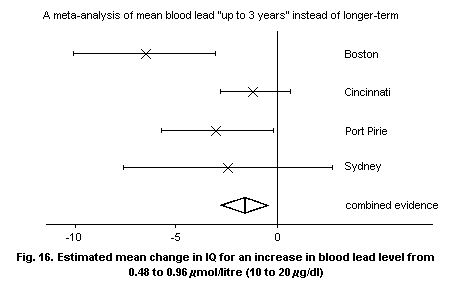 undertake an equivalent meta-analysis of the tooth lead studies. The
studies included have differed in the number and nature of covariates.
In particular, in most of the groups within the London and European
multi-centre study (MCS), maternal IQ has not been controlled. Studies
in which regression coefficients could not be obtained were excluded.
Since the studies not included in the analysis account for only a
small fraction of the total children investigated, their exclusion has
a negligible effect on the meta-analysis estimates of effect size.
The following studies are included in the meta-analysis:
Lavrion, Greece (Hatzakis et al., 1989)
Edinburgh (Fulton et al., 1987)
London (Greenwich) (Yule et al., 1981)
European multi centre study (Winneke et al., 1990)
The European multi-centre study comprises seven separate analyses
for children in Bucharest, Budapest, Modena, Sofia, Dusseldorf (two
studies) and Zagreb. The Lavrion centre in this study was separately
reported by Hatzakis et al. (1987).
As in the meta-analysis for the prospective studies, individual
cross-sectional studies varied as to whether the logarithm of the PbB
level or untransformed PbB level was used in the multiple regression
analysis. Hence, as before, all associations are expressed in terms of
the estimated changes in full-scale IQ for a change in PbB level from
0.48 to 0.96 µmol/litre (10 to 20 µg/dl), after adjustment for
confounders, as shown in Table 13.
Fig. 17 shows these estimates and their 95% confidence limits for
the 10 study samples. Only the two largest studies (Lavrion and
Edinburgh) show statistically significant inverse associations, as
indicated by confidence intervals entirely to the left of zero. The
limited statistical power of the other studies is reflected in their
wide confidence intervals. Despite the considerable variation in the
study designs (e.g., sample selection, choice of confounders), there
is no evidence of statistical heterogeneity. In other words, all the
confidence intervals overlap and the heterogeneity test is not
statistically significant.
A combination of the evidence from all of these cross-sectional
studies produces a more precise overall estimate of association, as
shown in Fig. 17. This meta-analysis of the cross-sectional studies
estimates that full-scale IQ is reduced by 2.15 points for an increase
in PbB level from 0.48 to 0.96 µmol/litre (10 to 20 µg/dl), with a 95%
confidence interval from -1.2 points to -3.1 points (P < 0.001).
undertake an equivalent meta-analysis of the tooth lead studies. The
studies included have differed in the number and nature of covariates.
In particular, in most of the groups within the London and European
multi-centre study (MCS), maternal IQ has not been controlled. Studies
in which regression coefficients could not be obtained were excluded.
Since the studies not included in the analysis account for only a
small fraction of the total children investigated, their exclusion has
a negligible effect on the meta-analysis estimates of effect size.
The following studies are included in the meta-analysis:
Lavrion, Greece (Hatzakis et al., 1989)
Edinburgh (Fulton et al., 1987)
London (Greenwich) (Yule et al., 1981)
European multi centre study (Winneke et al., 1990)
The European multi-centre study comprises seven separate analyses
for children in Bucharest, Budapest, Modena, Sofia, Dusseldorf (two
studies) and Zagreb. The Lavrion centre in this study was separately
reported by Hatzakis et al. (1987).
As in the meta-analysis for the prospective studies, individual
cross-sectional studies varied as to whether the logarithm of the PbB
level or untransformed PbB level was used in the multiple regression
analysis. Hence, as before, all associations are expressed in terms of
the estimated changes in full-scale IQ for a change in PbB level from
0.48 to 0.96 µmol/litre (10 to 20 µg/dl), after adjustment for
confounders, as shown in Table 13.
Fig. 17 shows these estimates and their 95% confidence limits for
the 10 study samples. Only the two largest studies (Lavrion and
Edinburgh) show statistically significant inverse associations, as
indicated by confidence intervals entirely to the left of zero. The
limited statistical power of the other studies is reflected in their
wide confidence intervals. Despite the considerable variation in the
study designs (e.g., sample selection, choice of confounders), there
is no evidence of statistical heterogeneity. In other words, all the
confidence intervals overlap and the heterogeneity test is not
statistically significant.
A combination of the evidence from all of these cross-sectional
studies produces a more precise overall estimate of association, as
shown in Fig. 17. This meta-analysis of the cross-sectional studies
estimates that full-scale IQ is reduced by 2.15 points for an increase
in PbB level from 0.48 to 0.96 µmol/litre (10 to 20 µg/dl), with a 95%
confidence interval from -1.2 points to -3.1 points (P < 0.001).
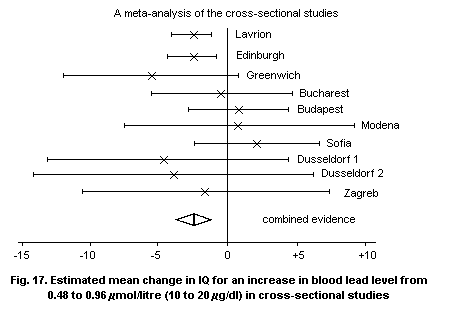 8.3.6.4 Task group overview of cross-sectional studies
a) Methods of controlling for confounders
A particular methodological concern in the cross-sectional
studies is the manner in which they have taken account of confounding
factors in deriving adjusted estimates of associations between body
lead burden and neuropsychological performance, in particular IQ.
The three aspects of the problem are:
i) Which potential confounders are measured? There is considerable
variation between studies here, suggesting that no individual study
can have fully corrected for the full range of parental and social
influences on IQ.
ii) How were confounders selected for inclusion in the analysis? Some
studies include confounders solely on the basis of their strength of
association with outcome (IQ), while others have given attention to
their association with body lead burden. It is difficult to assess the
extent to which these differences would affect the results, but the
former is more generally accepted statistical practice.
iii) Which statistical analysis strategy was employed in reaching a
final model? Most studies have adopted some form of multiple
regression technique, but have varied as to whether forward selection
(i.e. model building), backwards selection (i.e. model collapsing) or
more arbitrary choices of final model were employed. In the larger
studies, the choice between such strategies is unlikely to matter, but
in smaller studies the greater play of random variation could make the
results dependent on the statistical technique.
b) What they told us
In most studies a negative association between lead measures and
IQ measures is found in uncontrolled data. This difference is usually
in the range of 4 to 6 IQ points. Most studies also confirmed the
positive association between lead measures and indicators of social
disadvantage, whether this is indicated by SES, maternal education or
other more detailed indicators of non-optimal child-rearing
environments, such as marital quality or maternal depression. When
these social and other confounding factors are controlled, the effect
has been, in most cases, to reduce the strength of the association
between lead measures and IQ, although it remains in the same
direction. Where maternal intelligence has not been controlled, the
impact of correction for covariates tends to be smaller, and where
more detailed social measures have been controlled, the impact has
been greater.
c) What they did not tell us
The cross-sectional nature of these studies, and the fact that
many used a single measure of current exposure, limited their
usefulness in answering questions relating to the natural history of
the association between lead exposure and outcomes, including whether
there were critical periods of exposure, and whether lead-associated
deficits were persistent or reversible. Equally they were limited in
the information that could be obtained relating to the natural history
of confounders. They were also unable to answer questions of reverse
causality.
8.3.6.5 An interpretation of the overview of prospective and
cross-sectional studies
The above meta-analyses of the prospective studies and cross-
sectional studies reveal a consistency between studies which points
towards a "collectively significant" inverse association between PbB
level and full-scale IQ. Taking the results in Fig. 15 as a guideline,
there appears to be a mean decrease in full-scale IQ of the order of 2
IQ points for a change in mean PbB level from 0.48 to 0.96 µmol/litre
(10 to 20 µg/dl).
Below this range, uncertainties are increased, concerning firstly
the existence of an association and secondly estimates of the
magnitude of any putative association. The relatively limited numbers
of children in most studies with PbB levels below this range, the
strong contributions of confounding variables, and limitations in the
precision in analytical and psychometric measurements combine to lower
statistical power to detect associations and to estimate their
magnitude.
The key question is whether this statistical association is
directly attributable to the causal influence of lead on child IQ. It
is important to consider alternative explanations as follows:
a) Chance The consistency of alternative analyses and the level of
significance achieved suggest that chance cannot be a complete
explanation. However, the confidence intervals are relatively wide, so
that the magnitude of true association could be as low as a < 1-point
(rather than a 2-point) deficit, or as high as > 3 points.
b) Confounding factors The adjusted lead/IQ associations tend to be
substantially weaker than the unadjusted associations, which indicates
that confounding factors are important. Since none of the studies can
claim to have taken complete account of confounders, (e.g., father's
IQ is not included), it seems likely that some of the remaining
lead/IQ relationship, after adjustment, may still be attributable to a
degree of unexplained confounding.
c) Reverse causality One initial justification for the prospective
studies was that they could measure early exposure to lead and relate
it to later child development, thus removing the problem of reverse
causality potentially present in the cross-sectional studies. However,
it has turned out that very early lead exposure, e.g., in the
perinatal period, is not related to school-age IQ. The question arises
as to whether children of lower IQ could exhibit behaviour patterns at
earlier ages, e.g., around age 2 and older, which could enhance their
uptake of lead. Perhaps, such reverse causality remains a possibility
even in the prospective studies.
d) Selection biases The positive studies may perhaps be more likely
to report in more quantitative details, as indicated by the absence of
relevant data from Cleveland in the prospective studies. Post-hoc
selection based on the more significant PbB concen-trations may lead
to exaggerated estimates.
It is a matter of debate and conjecture as to the extent to which
these four issues should inhibit claims of a causal relationship in
the prospective studies. The essential problem is that observational
epidemiology cannot provide definitive evidence of causality when the
key statistical association is weak, the temporal relationship is
unclear and major confounders are present.
8.3.7 Hearing impairment in children
Schwartz & Otto (1987) reported that the probability of elevated
pure-tone hearing thresholds at 500, 2000 and 4000 Hz increased
significantly with increasing PbB level in 4519 subjects (4-19 years
of age) who participated in the NHANES II study. Variables included in
the backwards stepwise regression models included information derived
from medical history, clinical examinations, and assessment of
developmental milestones.
Schwartz & Otto (1991) examined data for subjects (6-19 years of
age) from the Hispanic Health and Nutrition Examination Survey
(HHANES) study which included three distinct ethnic groups. After
excluding 283 subjects with previous ear problems (discharges,
ruptured ear drums or tinnitus), data for 3262 were analysed. The
authors concluded that increasing PbB level in the range
0.36-0.86 µmol/litre (7-18 µg/dl) was associated with approximately
2 dB loss of pure-tone hearing at frequencies of 500, 1000, 2000 and
4000 Hz.
Dietrich et al. (1992) assessed the relationship between scores
on a test of sensory auditory processing (SCAN) and prenatal/postnatal
PbB concentrations in 215 subjects drawn from the Cincinnati
prospective cohort study. Higher prenatal, neonatal and postnatal PbB
concentrations were associated with more incorrect identification of
common monosyllabic words presented under conditions of muffling.
Other variables associated with impaired central auditory processing
were pure-tone audiometry results, social class, quality of caretaking
in the home, birth weight, gestational age, a measure of obstetrical
complications, and consumption of alcohol during pregnancy. Following
adjustment for these co-factors, lifetime average PbB concentration
remained significantly and inversely associated with SCAN performance.
8.4 Renal system
Acute exposure to lead is known to cause proximal renal tubular
damage, characterized by generalized aminoaciduria, hypophosphataemia
with relative hyperphosphaturia, and glycosuria (Chisolm, 1962).
Cellular structural changes include nuclear inclusion bodies,
mitochondrial changes and cytomegaly of the proximal tubular
epithelial cells (Cramer et al., 1974). Diagnosis of lead-induced
altered renal function or disease is difficult since there are no
specific indicators; blood urea nitrogen (BUN) and creatinine levels
become elevated only when twothirds of renal function has been lost
(Bernard & Becker, 1988).
8.4.1 Clinical studies
From a study of seven men occupationally exposed to lead in a
shipyard during oxy-acetylene flame cutting of lead-painted steel
hulls, Cramer et al. (1974) concluded that there is a continuum of
morphological and functional change in the pathogenesis of chronic
lead nephrotoxicity. Each subject was treated in hospital 3 or more
days after recent exposure and PbB levels exceeded 3.36 µmol/litre
(70 µg/dl) in all cases. On the basis of microscopic examination of
biopsy tissue, nuclear inclusion bodies were reported in proximal
tubular cells during the early phase but there was no impairment of
renal function. In a second phase, there was fibrosis associated with
asymptomatic azotaemia and reduced glomerular filtration rate but
without demonstrable proximal tubular dysfunction; renal failure was
not seen in the study.
Weeden et al. (1979) diagnosed lead nephropathy in 15 lead
workers, all having reduced glomerular filtration rates. Renal
biopsies of six of the subjects showed focal interstitial nephritis in
addition to non-specific changes in the proximal tubules. At the time
of examination, PbB levels for 11 of the 15 workers were within the
range 1.92-3.84 µmol/litre (40-80 µg/dl).
Baker et al. (1979) reported increased BUN and decreased
creatinine clearance in 28 workers, all of whom had relatively
prolonged, high-dose lead exposure in lead smelting or chemical
manufacturing.
A study by Maranelli & Apostoli (1987) of 60 workers, described
as "lead poisoned" and having PbB levels of 3.45 ± 0.80 µmol/litre
(71.9 ± 16.6 µg/dl), found no definitive correlation between PbB, lead
in urine after chelation, and BUN, serum creatinine and serum uric
acid.
8.4.2 Epidemiological studies
8.4.2.1 Occupational cohorts
Several recent studies of lead-exposed workers provide further
information relating to dose-effect relationships. The emphasis in
these studies was on lead and its possible association with adverse
effects on health. However, exposure to other potentially toxic
substances in the working and living environment should not be
overlooked/ignored.
For example, Buchet et al. (1980) examined 25 male lead smelter
workers and 88 male control workers. The PbB levels of the lead
workers were in the range 1.62-2.94 µmol/litre (33.8-61.3 µg/dl) for a
mean of 13.2 years (range 3.1-29.8 years) of lead exposure. The PbB
levels for the controls were in the range 0.26-1.64 µmol/litre
(5.5-34.2 µg/dl). There were no differences for parameters of renal
function between the groups and no signs of clinical renal impairment.
It was concluded that a PbB level of less than 2.98 µmol/litre
(62 µg/dl) is not associated with renal toxicity.
N-acetyl-ß-d-glucosaminidase (NAG) is a lysosomal enzyme present
in renal tubular cells. This enzyme is a sensitive but non-specific
indicator for early sub-clinical nephrotoxicity. In a study on 29
lead-exposed workers, Meyer et al. (1984) found increased NAG in
urine, but there was no correlation with PbB level. However, NAG level
was found to be normal in the 5 subjects with PbB levels exceeded
3.36 µmol/litre. These authors speculated that long-term high exposure
to lead may deplete the kidneys of NAG or render it insensitive to the
effects of lead exposure.
Verschoor et al. (1987) studied 155 male lead workers and 126
control workers who were matched for age, smoking habits,
socioeconomic status and duration of employment. The PbB levels of the
lead workers were in the range of 0.43-4.71 µmol/litre
(8.3-97.6 µg/dl) compared with the controls 0.15-0.96 µmol/litre
(3.1-18.8 µg/dl). The lead workers had elevated ZPP levels
(34-292 µmol/mol haemoglobin) compared with controls (10-35 µmol/mol
haemoglobin). No significant differences were found for various
indicators of renal function; all urinary and serum parameters were
within normal ranges. There were no differences in protein excretion
patterns and no signs of renal impairment. However, the authors found
that NAG levels in the lead-exposed workers were higher than control
values and increased with increasing PbB levels. They concluded that
lead exposure resulting in PbB levels of under 3.0 µmol/litre
(62 µg/dl) can affect renal tubular functions as measured by NAG
excretion; lead appeared to affect the tubular parameters more than
the glomerular parameters in moderately exposed workers.
Ong et al. (1987) examined 158 male and 51 female lead battery or
smelter workers and 30 control workers. The lead workers had 1-36
years exposure with an average of 10.8 ± 8.0 years and PbB levels in
the range of 0.14-3.84 µmol/litre (3-80 µg/dl); only five workers
exceed 2.88 µmol/litre (60 µg/dl). The authors found a weak but
statistically significant positive association between PbB and blood
urea nitrogen, and between PbB and serum creatinine, and that
creatinine clearance was reduced with increasing PbB level. NAG levels
in the lead-exposed workers were significantly higher than control
values and increased with increasing urine lead level when the data
were adjusted for age. These authors concluded that a relatively low
PbB level can affect renal function.
Not all investigators have found parallel association between PbB
and NAG levels. Gennart et al. (1992b) compared 98 lead-exposed lead
acid battery workers (mean PbB level 2.45 µmol/litre, 51 µg/dl,
geometric mean ZPP 10.2 µg/g Hb) with 85 controls (mean PbB level
1.00 µmol/litre, 20.9 µg/dl, geometric mean ZPP 2.84 µg/g Hb)
recruited from other departments in the same factory. None of the
indicators of renal function (retinol-binding protein,
ß2-microglobulin, albumin or NAG in urine, or creatinine or
ß2-microglobulin in serum) were correlated with PbB level, duration
of exposure or ZPP, or showed significantly different mean values
between the lead-exposed and control groups of workers.
Cardenas et al. (1993) recently evaluated 27 different
indications of renal dysfunction in 50 workers exposed to lead and 50
controls. There were significant increases in urinary NAG and sialic
acid in the lead exposed group. These changes may represent minor
cellular modifications rather than significant functional or
irreversible renal damage. There was a significant decrease in urinary
6-keto PGF1alpha and a significant increase in TXB2. These
eicosanoid changes may reflect systemic functional vascular changes
rather than an effect of lead on the kidney.
8.4.2.2 General population
An epidemiological survey on 283 persons in Scotland, from
households with water lead concentrations in excess of 100 µg/litre,
revealed a close correlation between water lead content and PbB and
serum urea concentrations (Campbell et al., 1977). The frequency of
renal dysfunction in individuals with elevated PbB concentrations
(> 2 µmol/litre or > 41 µg/dl) was significantly greater than that
of age- and sex-matched controls.
Pocock et al. (1984) measured serum creatinine, urate and urea
concentrations in 7364 British men, and 74 subjects had PbB levels
equal to or greater than 1.8 µmol/litre (37.3 µg/dl). The authors
concluded that there was no indication that exposure to lead at
concentrations commonly encountered in Britain was responsible for
impaired renal function.
Staessen et al. (1992) investigated the relationship between lead
exposure and renal function as part of a cross-sectional population
study of the health effects of environmental exposure to cadmium.
Creatinine clearance, PbB and ZPP were measured in a random population
sample of 965 men (geometric mean PbB level was 0.55 µmol/litre or
11.4 µg/dl) and 1016 women (geometric mean PbB level was
0.36 µmol/litre or 7.5 µg/dl). Creatinine clearance rate (mean was
99 ml/min in men, 80 ml/min in women) was inversely correlated with
PbB and ZPP levels before and after adjustment for age, body mass
index and diuretic treatment. Also positively correlated were serum
ß2-microglobulin and PbB level in men, serum ß2-microglobulin and ZPP
in men and women, and serum creatinine and ZPP in men. Impaired renal
function could not be explained by exposure to cadmium or by elevated
blood pressure. The authors concluded that exposure to lead may impair
renal function in the general population. However, the study could not
exclude the possibility that renal impairment may lead to an increase
in PbB level.
8.4.2.3 Cohort mortality studies
"Lead poisoning" was a diagnosis given to 241 workers employed
for 1-30 years at the Port Pirie lead smelter between 1928-1959 by a
State medical board. Death registration records for the period
1930-1977 identified 140 deaths among the group. The cause of death
profile was compared with that of 695 other male decedents,
predominantly production workers and a smaller number of office
workers. Age-standardized mortality analysis revealed a substantial
excess of deaths attributed to chronic nephritis and to cerebral
haemorrhage. The rates for lead-poisoned workers exceeded those for
the other workers, and both exceeded the rates for the Australian
general male population. The rates decreased over successive calendar
periods but excess rates persisted for chronic nephritis up until 1977
(McMichael & Johnson, 1982).
8.5 Cardiovascular system
Two persistent issues have been under intense study since
publication of Environmental Health Criteria 3: Lead (IPCS, 1977):
a) whether lead is a factor in hypertension and, if so, whether
there is a causal relationship;
b) whether lead is contributory to cardiovascular effects
influencing morbidity or mortality.
8.5.1 Blood pressure
8.5.1.1 Studies on occupationally exposed cohorts
From a study of 431 white male police officers 24-55 years of age
(Moreau et al., 1982; Orssaud et al., 1985), it was concluded that
systolic and diastolic blood pressure was related to PbB level, the
correlation being greatest for the younger subjects and decreasing
with age. Statistical adjustment was made for alcohol consumption and
body mass index, but not for smoking. The magnitude of observed
association between systolic pressure and PbB level in this small
study was somewhat greater than in the larger general population
studies cited below.
Parkinson et al. (1987) studied 270 lead battery workers and 158
non-exposed workers and compared lead exposure with systolic and
diastolic blood pressures. After controlling for age, education,
income, cigarette smoking, alcohol consumption and exercise, there was
a small and non-significant association. The average PbB level of
lead-exposed workers was 1.92 ± 0.62 µmol/litre (40 ± 13 µg/dl),
whereas in non-exposed workers it was 0.34 ± 0.24 µmol/litre
(7 ± 5 µg/dl).
8.5.1.2 Studies in the general population
The possible relationship between PbB concentration and blood
pressure has been examined in several large-scale population studies.
These include the British Regional Heart Study (BRHS), the US NHANES
II (National Health and Nutrition Examination Survey) and studies in
Wales, Denmark, Canada and Belgium.
The BRHS is a prospective study of 7735 men, initially aged 40-59
years, from 24 British towns, who were first examined in 1978-1980
(Shaper et al., 1981). The median PbB level was 0.7 µmol/litre
(14.5 µg/dl). The initial findings (Pocock et al., 1984), which
indicated a lack of association between PbB level and blood pressure,
were later re-examined to take account of potential confounding
factors such as alcohol, smoking and town of residence. Pocock et al.
(1988) noted that 95% confidence intervals for systolic and diastolic
blood pressure plotted against PbB concentrations overlapped and
showed no elevation even at the highest observed PbB concentration.
Applying statistical techniques to adjust mean systolic and diastolic
blood pressure measurements for body mass index, age, alcohol
consumption, cigarette smoking and town of residence showed signs of a
weak association, particularly for diastolic pressure and PbB level.
Multiple regression analysis showed that adjustment for personal
characteristics but not for town of residence rendered both systolic
and diastolic regressions on log PbB insignificant, chiefly because
alcohol consumption is an important confounder, being positively
related to both blood pressure and PbB level. Introducing an extra
adjustment for town of residence made the systolic and diastolic
regressions highly statistically significant, even though the
associations were weak. These cross-sectional data indicated that for
every doubling in PbB level (e.g., from 0.8 to 1.6 µmol/litre) there
are estimated mean increases of 1.45 mmHg (95% confidence interval
0.47-2.43 mmHg) in systolic blood pressure and 1.25 mmHg (95%
confidence interval 0.65-1.85 mmHg) in diastolic blood pressure.
The NHANES II study, a USA national cross-sectional survey
carried out in 1976-1980, included PbB and blood pressure measurements
in a general population sample of 5803 men and women aged 12-74.
Geometric mean PbB levels were around 0.72 µmol/litre (about 15 µg/dl)
for men and approximately 0.53 µmol/litre (11 µg/dl) for women.
Several authors have examined these data for possible PbB and blood
pressure associations.
In an analysis controlling for the confounding factors of age,
race and body mass index, Harlan et al. (1985) and Harlan (1988) found
a significant association between PbB level and blood pressure for men
but not for women. White males aged 40-59 years from the same data set
were analysed by Pirkle et al. (1985), and the correlation between PbB
level and blood pressure was confirmed. In further analysis of the
whole age range, adjustments for possible time trend and geographical
site effects did not affect the significance of the association in
males (Landis & Flegal, 1988; Schwartz, 1988), although its magnitude
did become somewhat less pronounced.
Gartside (1988) applied a method of forward stepwise regression
to PbB and blood pressure data from NHANES II and reported that the
results for white men, white women and black men were contradictory
and lacked consistency and reliability. He noted that the overall
average was too small to conclude association between PbB level and
blood pressure in this study.
Two surveys were carried out in Wales by Elwood et al. (1988a,b).
The Welsh Heart Programme carried out in 1985 provided complete PbB,
blood pressure and other data for 865 men and 856 women aged 18-64.
The geometric mean PbB level for men was 0.56 µmol/litre (11.6 µg/dl)
and for women was 0.43 µmol/litre (9.0 µg/dl). In neither sex was
there a significant correlation between PbB level and systolic or
diastolic blood pressure. The second study was in a cohort of men aged
49-65 years living in Caerphilly, Wales. The geometric mean PbB level
(N = 1137) was 0.61 µmol/litre (12.7 µg/dl). Complete data from 1137
subjects, ranking blood pressure readings according to PbB level, did
not reveal any trend in the percentage of subjects with systolic
pressure above 160 mmHg. The authors corrected only for age as a
confounding factor.
Data from a Canadian study (Neri et al., 1988) collected during
10 months in 1978-1979 for 2193 subjects aged 25-64 showed a weak but
statistically significant association between PbB level and diastolic
blood pressure.
Grandjean et al. (1989b) studied 504 men and 548 women residing
in Glostrup (Denmark) at age 40; 451 men and 410 of the women were
followed-up five years later. Average PbB levels for the men at 40 and
45 years of age were 0.62 µmol/litre (13 µg/dl) and 0.43 µmol/litre
(9 µg/dl), and for the women 0.43 µmol/litre (9 µg/dl) and
0.29 µmol/litre (6 µg/dl), respectively. All correlations found
between PbB and blood pressure became insignificant when blood
haemoglobin and alcohol intake were entered into multiple regression
analyses; an independent effect of low-level lead exposure on blood
pressure could not be distinguished.
Möller & Kristensen (1992) examined 1052 men and women in
Copenhagen, Denmark, in 1976, and re-examined them in 1981 and 1987
(men only). Initial mean PbB levels were 0.65 µmol/litre (13.6 µg/dl)
in men and 0.46 µmol/litre (9.6 µg/dl) in women, and had fallen by
around 30% 5 years later. For men, there was no significant
association between PbB level and blood pressure (or between changes
in PbB level and blood pressure) after adjustment for confounders
(tobacco, alcohol, body mass and physical activity). For women, the
association between PbB level and diastolic (but not systolic) blood
pressure remained significant on both occasions, even after
controlling for confounders.
A Belgium study (Dolenc et al., 1993) included 827 men and 821
women (mean age 25) whose mean PbB levels were 0.56 and
0.34 µmol/litre (11.6 and 7.07 µg/dl), respectively. After adjustment
for covariates (body mass index), pulse rate and serum creatinine and
serum calcium levels), systolic blood pressure was inversely
associated with PbB level in men (P < 0.05). There were no
significant PbB associations for diastolic blood pressure in men or
for either pressure in women.
A study of 398 male and 133 female civil servants (Staessen et
al., 1990) showed geometric mean PbB levels of 0.58 µmol/litre
(12.06 µg/dl) and 0.46 µmol/litre (9.56 µg/dl) in men and women,
respectively. Taking the data for both sexes together, there were
statistically significant positive associations between PbB level and
systolic and diastolic blood pressures (r = +0.11 in each case) but
these became non-significant after adjustment for confounders (sex,
age, BMI, pulse, delta-glutamyltranspeptidase (delta-GTP) and serum
calcium level).
Another cross-sectional study involving only women was performed
in Boston (Rabinowitz et al., 1987). Cord PbB levels (mean
0.33 µmol/litre, 6.9 µg/dl) among 3851 women correlated with both the
systolic (r = 0.08) and diastolic (r = 0.05) blood pressures measured
during and before delivery. Multivariate models of pregnancy
hypertension as a function of age, parity, haematocrit, diabetes,
ponderal index, and race were improved when lead was included as a
predictor. Lead appeared to have a small but demonstrable association
with pregnancy hypertension and blood pressure at delivery, but not
with eclampsia.
In an overview of the BRHS, NHANES II and Welsh studies, Pocock
et al. (1988) concluded that with overlapping confidence limits the
data provided weak but reasonably consistent evidence of lead and
blood pressure associations. They noted that the NHANES II data on
2254 men in the USA indicate a slightly stronger association between
PbB level and systolic blood pressure, whereas data from over 2000 men
in Wales did not show a statistically significant association. They
inferred that a causal relationship could not be concluded from any of
the epidemiological studies.
Staessen et al. (1994) have undertaken a more extensive
meta-analysis of nearly all of the above studies and some other
smaller ones as well. This amounted to a total of 19 studies with
28 210 subjects. They found that the association between PbB level and
blood pressure was similar in both sexes. In all studies combined, a
two-fold increase in PbB concentration was associated with a 1.0 mmHg
increase in systolic pressure (95% confidence interval (CI); 0.3 to
1.7 mmHg; P = 0.008) and a 0.7 mmHg increase in diastolic pressure
(95% CI: 0.2 to 1.3 mmHg; P = 0.02). These authors conclude that the
published evidence suggests a weak positive association between blood
pressure and lead exposure, but any such relationship may not be
causal and is unlikely to entail any public health implications
regarding hypertension.
8.5.2 Other cardiovascular effects
As recorded previously (IPCS, 1977), there is good evidence that
signs of clinical lead poisoning sometimes include evidence of toxic
action on the heart. Kopp et al. (1988) reviewed the cardiovascular
actions of lead and concluded that the degree of cardiovascular
involvement during episodes of acute lead intoxication depends on the
duration of exposure and dose. It is not known whether environmental
exposure to lead affects the electrical or mechanical activity of the
heart.
8.5.2.1 Occupational studies
There have been several studies relating to cardiovascular
effects in lead-exposed occupational groups. However, as with any
study of an occupational cohort, it is important to evaluate the
strength of the cause-effect relationship, the nature or substance of
other contributory factors and how well the workplace "exposure" has
been evaluated for other potential toxicants. Thus, the occupational
hazards associated with work in lead smelting or lead battery
operations include, but are not limited to, lead.
8.5.2.2 Studies in the general population
Pocock et al. (1988) found that after 6 years of follow-up of the
BRHS cohort of 7735 middle-aged men, 316 of the men had major
ischaemic heart disease and 66 had had a stroke. After allowance for
confounding effects of cigarette smoking and town of residence, there
was no evidence that PbB was a risk factor for these cardiovascular
events.
Möller & Kristensen (1992) studied 1052 men and women in
Copenhagen, Denmark, and related blood pressure to subsequent
cardiovascular morbidity and mortality over a 14-year period. There
were significant positive associations between PbB level and both
coronary and cardiovascular disease in univariate analysis, but these
became non-significant after controlling for confounders.
a) Clinical studies
Boscolo & Carmignani (1988) measured plasma renin activity in
hospitalized lead-exposed workers and concluded that synthesis or
release of renin is increased by short or moderate exposure to lead
and decreased with prolonged exposure.
b) Epidemiological studies
Kirkby & Gyntelberg (1985) found a significantly higher incidence
of ischaemic ECG changes in a study of lead smelter workers (20%, mean
PbB level 2.45 µmol/litre, 51 µg/dl), compared with matched
non-exposed controls (6%, mean PbB level 0.53 µmol/litre, 11 µg/dl).
There was also a slight (4-5 mmHg) increase in diastolic blood
pressure in the lead workers compared with the controls.
c) Cohort mortality studies
Two studies (Cooper, 1988 and Fanning, 1988) reported an
increased mortality rate of lead workers due to circulatory disease,
but no such relationship was found in reports of studies by
Gerhardsson et al. (1986) or Selevan et al. (1988). Gerhardsson et al.
(1986) examined a lead-exposed sub-cohort of 437 from a study cohort
of 3832 male workers first employed at a copper smelter before 1967
and followed up from 1950 to 1981. There was no excess mortality due
to ischaemic heart disease or cerebrovascular disease in the
lead-exposed worker sub-cohort. Selevan et al. (1988) found no
association between lead exposure and deaths due to hypertensive
diseases in a review of mortality in a cohort of male hourly workers
at a lead smelter. There were many confounding factors in each of
these studies.
8.5.3 Summary
Despite intensive efforts to define the relationship between body
burden of lead and blood pressure or other effects on the
cardiovascular system, no causal relationship has been demonstrated in
humans and the mechanisms remain obscure.
There is experimental evidence from animal studies indicative of
an effect of lead on blood pressure, and several mechanisms have been
proposed to explain these observations (see section 7.4).
8.6 Gastrointestinal effects
8.6.1 Occupational exposure
Colic is a well-recognized symptom of acute lead poisoning and is
still reported in groups of lead-exposed industrial workers. Of
particular concern are the reports of intoxication caused by acute
exposure (inadequate protective measures) to lead associated with
removal of lead-based paint by burning or sand-blasting and the
demolition of lead-containing industrial plants. Symptoms of colic
include abdominal pain, constipation, cramps, nausea, vomiting,
anorexia, weight loss and decreased appetite.
Symptoms in adults typically occur at PbB levels of
4.8-9.6 µmol/litre (100-200 µg/dl) but have been noted at levels as
low as 1.92 µmol/litre (40 µg/dl) (Baker et al., 1979; Haenninen et
al., 1979; Awad El Karim et al., 1986; Pollock & Ibels, 1986; Muijser
et al., 1987; Holness & Nethercott, 1988; Marino et al., 1989;
Pagliuca et al., 1990; Schneitzer et al., 1990).
Positive histories of lead colic were given by 40 workers in a
cohort of 158 secondary lead smelter workers who participated in a
clinical field survey (Lilis et al., 1977). It was reported most
frequently by those having PbB levels above 3.84 µmol/litre (80 µg/dl)
at the time of examination and was not found where ZPP was within the
normal range. Thirty percent of workers having ZPP > 200 µg/g Hb had
experienced symptoms.
In a study of a population of 585 black South African factory
workers, Irwig et al. (1978) reported that the incidence of abdominal
pain increased with increasing PbB level: it was 12% for the first
quartile (PbB < 3.28 µmol/litre or < 68 µg/dl), 23% for the
second (3.28-4.14 µmol/litre or 68-86 µg/dl), 24% for the third
(4.15-5.14 µmol/litre or 86-107 µg/dl) and 37% for the fourth
(> 5.15 µmol/litre or > 107 µg/dl).
In a cross-sectional clinical study of 90 telephone
cable-splicers by Fischbein et al. (1980), 19 (21%) workers reported
gastrointestinal symptoms (mean PbB level of 1.44 µmol/litre, or
30 µg/dl and mean ZPP of 66.6 µg/g Hb) whereas 71 (79%) reported no
such symptoms (mean PbB level of 1.3 µmol/litre or 27 µg/dl and mean
ZPP of 52.3 µg/g Hb).
8.6.2 Exposure of children
Colic is seen in children and US EPA (1986a) concluded that the
lowest-observed-adverse-effect level was in the range of
2.88-4.80 µmol/litre (60-100 µg/dl).
8.7 Liver
There appears to be no new evidence relating human body lead
burden to effects on the liver, but it has been suggested that the
effects of lead on haem synthesis may alter the functional capacity of
the hepatic cytochrome P-450 system to metabolize drugs.
8.7.1 Occupational exposure
Fischbein et al. (1977) tested five demolition workers acutely
exposed to lead for a period of 3 months prior to the study. They
found that the plasma half-life of an oral dose of antipyrine was
within the range for normal healthy volunteers; it was shorter in each
subject after chelation therapy but still within the normal range. The
plasma half-lives of phenylbutazone were also within the normal range
but were unaffected by chelation therapy.
8.7.2 Exposure of children
Saenger et al. (1984) found decreased urinary excretion of
6-ß-hydroxycortisol in 26 children with a mean PbB level of
2.11 µmol/litre (44 µg/dl); the decreased formation of the metabolite
was attributed to lead inhibition of the cytochrome P-450-dependent
mixed-function oxidases.
8.8 Reproduction
While it is generally accepted from early literature that lead
adversely affects the reproductive process in both men and women, the
evidence is mostly qualitative and dose-effect relationships have not
been established.
Most new information relates to reports of occupational cohorts
and of populations living in polluted areas near industrial plants.
There is qualitative evidence that lead is toxic to the reproduction
system in both men and women. However, there are insufficient data to
provide the basis for estimation of dose-effect relationships in
women.
8.8.1 Female populations
Nordstrom et al. (1978b) reported an increased frequency of
spontaneous abortion in women living close to a smelter in northern
Sweden. In a later report, Nordstrom et al. (1979) described the
responses to a questionnaire completed by 511 of 662 women who had
worked at the smelter and were born between 1930 and 1959. Spontaneous
abortion rates were highest in those pregnancies in which the mother
was employed during the pregnancy (13.9%) or had been employed before
the pregnancy and was living close to the smelter (17%); the frequency
rate was higher (19.4%) when the father worked at the smelter.
However, it should be noted that the smelter produced copper and lead
in addition to a number of other metallurgical and chemical products
(Nordstrom et al., 1978a) and that the effects reported may not
necessarily be attributable exclusively to lead.
A study of pregnancies in the centre and surrounding areas of the
lead smelter town of Port Pirie showed that the incidence of
miscarriages (22 or 23) and stillbirths (10 or 11) was higher in women
living close to the smelter (McMichael et al., 1986). However, in a
study of 639 Yugoslav women, the risk of spontaneous abortion was not
increased (OR = 1.1, 95% CI: 0.9-1.4) among women with mean PbB levels
of 0.77 µmol/litre (16.0 µg/dl) as compared with women with mean PbB
levels of 0.25 µmol/litre (5.2 µg/dl) (Murphy et al., 1990). Risk did
not vary with distance of residence from the smelter.
Some studies have found decreased length of gestation in women
whose PbB levels were greater than 1.09 µmol/litre (23 µg/dl) (Moore
et al., 1982), 0.58 µmol/litre (12 µg/dl) (Dietrich et al., 1986) or
0.72 µmol/litre (15 µg/dl) (McMichael et al., 1986). However, neither
Bellinger et al. (1991b) nor Graziano et al. (1990) found decreases in
gestational length or other parameters of pregnancy in women with
elevated PbB levels.
In the Cincinnati prospective study, a significant reduction in
birth weight associated with prenatal (maternal) PbB levels, after
adjustment for covariates, was reported by Bornschein et al. (1989).
In the Port Pirie prospective study of 749 pregnancies (McMichael et
al., 1986), the proportion of pregnancies resulting in low birth
weight singleton infants was more than twice as high in Port Pirie
women (whose PbB levels averaged 0.50 µmol/litre, 10.4 µg/dl) than in
women outside Port Pirie (average PbB level of 0.264 µmol/litre,
5.5 µg/dl). On the other hand, multiple regression analyses showed no
significant association between low birth weight and maternal PbB
level. In a cross-sectional study, Ward et al. (1987) reported a
significant simple relationship between placental lead concentrations
and reduced birth weight and head circumference.
Other studies have not shown a significant association between
birth weight and lead exposure. The Kosovo prospective study failed to
detect any evidence of lead-related birth weight reduction in more
than 900 births (Murphy et al., 1990), and Ernhart et al. (1986) found
no significant effect of lead on birth weight, birth length or head
circumference.
8.8.2 Male populations
Reproductive effects from occupational exposure to lead include
asthenospermia, hypospermia, teratospermia and hypogonadism (US EPA,
1986a; Braunstein et al., 1987).
In men, effects on sperm or the testes may result from chronic
exposure to lead at blood levels of the order of 1.92-2.4 µmol/litre
(40-50 µg/dl) (ATSDR, 1991).
Wildt et al. (1983) compared two groups of lead-exposed storage
battery workers and concluded that the highly exposed group (PbB of
more than 2.16 µmol/litre, 45 µg/dl) had decreased prostate/seminal
vesicle function, low semen volume and poorer functional maturity of
sperm.
Chowdhury et al. (1986) reported a significant decrease in sperm
count and motility and an increased count of abnormal spermatozoa in
lead-exposed men (average PbB level of 2.04 µmol per litre or
42.5 µg/dl) compared with controls (0.71 µmol/litre or 14.8 µg/dl).
Assenato et al. (1987) also reported decreased sperm production in 18
battery factory workers having PbB levels of 2.4-2.93 µmol/litre
(50-61 µg/dl).
In five out of six symptomatic lead intoxicated workers having
PbB levels in the range of 1.87-4.7 µmol/litre (39-98 µg/dl) at the
time of examination, Cullen et al. (1984) found defects of
spermatogenesis, including oligospermia and azoospermia.
The results of Lerda (1992) also showed significant increases in
asthenospermia and teratospermia among 38 battery workers with PbB
levels of 1.95-4.7 µmol/litre (40.5-98.0 µg/dl) compared to a control
group with PbB levels of 0.86-1.25 µmol/litre (17.6-26.0 µg/dl).
An epidemiological study by Lindholm et al. (1991) of 213 wives
of lead workers and 300 matched controls suggested an association
between paternal lead exposure and the risk of spontaneous abortion
among the wives of lead workers when the PbB level exceeded
1.5 µmol/litre (31 µg/dl) close to the time of spermatogenesis.
However, the authors acknowledged that exposure of the study subjects
to other metals and organic solvents may have influenced the results
of the study.
A weak association between paternal exposure, estimated from
occupational history, and risk of esotropia (inward deviation
strabismas) was noted in a case control study of 377 children aged 7
years (OR - 2.4, 95%, CI: 1.0-5.6) (Hakim et al., 1991). However, the
increased risk was not dose-related, and the risk of exotropia
(outward deviation) was not associated with higher paternal lead
exposure.
8.8.3 Hormonal responses
Assenato et al. (1987) found no significant differences in FSH,
testosterone, prolactin, LH and total neutral 17-ketosteroids levels
between 18 lead battery workers (PbB level, 2.4-3 µmol/litre or
50-61 µg/dl) and 18 cement workers (PbB level, 0.9-1.1 µmol/litre or
18-22 µg/dl), despite the finding of oligospermia in the lead-exposed
group.
Rodamilans et al. (1988) studied the endocrine status of 23 lead
smelters in relation to the duration of lead exposure. Five workers
exposed for less than 1 year (mean PbB level, 0.29 µmol/litre or
66 µg/dl) showed an increase in serum LH level, while that of
testosterone remained normal. Eight workers exposed for 1-5 years
(mean PbB level, 3.50 µmol/litre or 73 µg/dl) and 10 employed for more
than 5 years (mean PbB level, 3.65 µmol/litre or 76 µg/dl) showed an
increase in LH comparable with the group exposed for less than 1 year
but there was a clear reduction in serum testosterone levels.
Gustafson et al. (1989) found a lower plasma FSH level in 25
moderately exposed workers (mean PbB level, 1.82 µmol/litre or
38 µg/dl) than in 25 matched controls. A lower mean serum LH level was
reported by McGregor & Mason (1990) in 90 lead workers (mean PbB
level, 2.20 µmol/litre or 46 µg/dl) than in controls; there was also a
correlation between serum FSH and PbB levels. In an epidemiological
study involving 122 current lead workers (PbB mean level,
1.69 µmol/litre or 35.1 µg/dl) and 49 non-exposed workers (PbB mean
level, 0.4 µmol/litre or 8.3 µg/dl), Ng et al. (1991) found slightly
higher plasma LH and FSH levels among the exposed workers. The
testosterone level, however, was not significantly different between
the two groups. In contrast, Gennart et al. (1992) did not find an
effect of lead on various endocrine parameters (including FSH and LH
levels) in a study population of 221 workers having a geometric mean
PbB level of 2.45 µmol/litre (51.0 µg/dl). They suggested that the
hypothalamic-pituitary system may not be influenced by moderate
exposure to lead.
8.8.4 Postnatal growth and stature
Several reports have suggested that the physical growth and
stature of children may be reduced by exposure to lead (e.g., Mooty et
al., 1975; Johnson & Tenuta, 1979; Routh et al., 1979), but the
influence of other factors (e.g., race and diet) has often made it
difficult to isolate lead as a causal agent for such effects in human
populations. Multivariate regression analyses of NHANES data for
approximately 2700 children in the USA (Schwartz et al., 1986)
provided more convincing evidence of a significant association between
increasing PbB levels and reduced height, weight, and chest
circumference after adjusting for age, race, sex and nutritional
covariates.
8.9 Effects on chromosomes
As noted in Environmental Health Criteria 3: Lead (IPCS, 1977),
the literature remains controversial concerning induction of
chromosomal changes in human lymphocytes by in vivo exposure to
lead. Most of the studies are of small numbers of subjects in
occupational groups where lead was one of many potentially toxic
agents. However, some of the difficulties arise because of the lack of
standard procedures used for the assessment of chromosomal effects in
cultured lymphocytes and from a lack of understanding of the health
significance of chromosomal abnormalities.
Induced mitotic activity in peripheral lymphocytes and increases
in the rates of abnormal mitosis were reported by Sarto et al. (1978).
PbB levels in the range of 1.05-4.27 µmol/litre (22-89 µg/dl) were
reported to be associated with increased incidence of chromosomal
aberrations by Schmid et al. (1972) in 32 lead manufacturing workers,
by Nordenson et al. (1978) in 18 smelter workers and by Al-Hakkak et
al. (1986) in 19 lead manufacturing workers. It should be noted that
exposure of worker to potentially hazardous agents other than lead was
not documented in these studies.
Maki-Paakkanen et al. (1981) reported no change in chromosomal
aberration frequency among workers with PbB levels in the range of
1.82-5.76 µmol/litre (38-120 µg/dl); weak effects were found by Huang
et al. (1988b) when the PbB level exceeded 2.5 µmol/litre (52 µg/dl).
Qazi et al. (1980) reported increased numbers of cells with
chromosome breaks in a baby having a cord PbB level of 2.88 µmol/litre
(60 µg/dl) and a PbB level of 3.45 µmol/litre (72 µg/dl) at 2 weeks.
The effect was seen at 6 weeks and at 3 months but not later
(chelation therapy was given at 17 days and repeated at 5 months).
Bauchinger et al. (1977) tested blood taken from 38 children
selected from a school situated close to a lead plant and in whom the
PbB level was at least 1.44 µmol/litre (30 µg/dl). There was no
increase in the frequency of aberrations.
Grandjean et al. (1983) examined 10 long-term workers at a lead
storage battery plant. They found normal or slightly increased SCE
rates in workers with a PbB level of 1.4-3.6 µmol/litre (29-75 µg/dl)
and a ZPP level of 50-750 µmol/mol haemoglobin. The SCE increase was
correlated significantly with ZPP level.
Huang et al. (1988b) studied 21 lead-exposed workers from a
battery factory. They concluded that the SCE rate in workers with
long-term exposure to lead increased significantly when the mean PbB
level was 3.84 µmol/litre (80 µg/dl) or more. There were no
significant differences in SCE rates among the low- and medium-exposed
groups or the controls. These authors concluded that the effect of
chromosome damage caused by lead is not very strong.
Dalpra et al. (1983) examined blood samples taken from 19
children living in a contaminated area near a smelter and having PbB
levels in the range of 1.39-3.02 µmol/litre (29-63 µg/dl). They found
no effect on SCE frequency.
8.10 Carcinogenicity
Much has been written regarding the evidence that lead is
carcinogenic in humans (Moore & Meredith, 1979; Kazantzis, 1989;
Goyer, 1992). In several large epidemiological studies no association
was found which would associate lead with induction of cancer (Cooper
& Gaffey, 1975; Kang et al., 1980; Cooper, 1981; McMichael & Johnson,
1982). One major difficulty in many of the studies was the concurrent
exposure to potential carcinogens such as chromium (Davies, 1984), and
there has seldom been any attempt to deal with the primary etiological
agent (smoking) in the development of lung cancers associated with
lead exposure.
8.10.1 Occupational exposure and renal cancer
There are two case reports and very limited other epidemio-
logical evidence of an association between occupational exposure to
lead and renal cancer. Confounding variables, including use of tobacco
and exposure to other carcinogens, were not addressed (Baker et al.,
1980; Lilis, 1981; Selevan et al., 1985; Cantor et al., 1986).
The age-standardized mortality ratio for cancer was low in the
"lead poisoned" and other groups of workers from the Port Pirie study
by McMichael & Johnson (1982). The authors concluded that lead
poisoning did not increase the risk of cancer in humans.
8.10.2 Conclusion
Although some cohort mortality studies have indicated an
association between lead exposure and renal disease, there is no
association between renal cancer and lead in humans.
A Working Group convened by the International Agency for Research
on Cancer (IARC) in 1987 concluded that the evidence for the
carcinogenicity of lead and inorganic lead compounds in humans was
inadequate.
8.11 Effects on thyroid function
8.11.1 Occupational groups
Robins et al. (1983) found low values for serum thyroxine and
estimated free thyroxine in 7 of 12 workers having PbB levels above
2.11 µmol/litre (44 µg/dl). Both measures were correlated with PbB
level in a cross-sectional study of 47 foundry workers. Serum
thyrotropin and triiodothyronine levels were within the normal range
for both study groups.
No such relationship was found in a study by Refowitz (1984) of a
one-in-three random sample (N = 58) of male employees at a secondary
copper smelter. No thyroid abnormalities were observed and there was
no statistically significant relationship between PbB and serum
thyroxine or an estimate of free thyroxine in serum.
Tuppurainen et al. (1988) studied 176 African male workers in
Kenya and found that the duration of lead exposure correlated
negatively with serum free thyroxine and serum total thyroxine. The
correlation was strongest for the workers with the highest exposure
intensity over time. PbB data were available from periodic medical
examinations and average values (usually five determinations) were
used. The mean PbB was 2.73 µmol/litre (56.8 µg/dl), range
1.01-6.47 µmol/litre (21-135 µg/dl), and there was a mean exposure
duration of 7.6 years (range 1-20). The authors noted that current PbB
level, as a point determination, was not associated with total or free
thyroxine, triiodothyronine or thyrotropin in serum. They proposed
that long-term, highintensity exposure might be associated with
depressed thyroid function.
Gennart et al. (1992b) included assessment of thyroid function as
part of a study of lead-exposed workers. Data for serum levels of
triiodothyronine, thyroxine, free thyroxine index and thyroid-
stimulating hormone were within normal ranges for a group of 98
workers (mean PbB level, 2.45 µmol/litre or 51 µg/dl) and 85 controls
(1.00 µmol/litre or 20.9 µg/dl). The lead exposure of this study group
was considerably lower than in the workers in Kenya, and it was
suggested that thyroid function changes might not be indicators of
moderate exposure to lead.
8.11.2 Effects in children
Siegel et al. (1989) tested 68 children for thyroid function and
for PbB and found no statistically significant relationship between
lead and total or free thyroxine.
8.12 Immune system
In a review of the effects of lead on the immune responses of
experimental animals, Koller (1985) noted that there were few human
studies.
8.12.1 Occupational exposure
There is some evidence that lead workers with PbB levels in the
range of 1.0-4.10 µmol/litre (21-85 µg/dl) have increased
susceptibility to infections (colds and influenza) and have a
significant suppression of secretory IgA levels, a major factor in
defence against respiratory and gastrointestinal infections (Ewers et
al., 1982). There are also reports of impaired mitogen responses
(reflecting T-lymphocyte function) to phytohaemagglutinin (Jaremin,
1983) and of increased numbers of suppressor T-cells (Cohen et al.,
1989). However, on the basis of a study of 39 workers exposed to lead
oxide (mean PbB level of 1.84 µmol/litre or 38.5 µg/dl) and 21 control
subjects (mean PbB level of 0.57 µmol/litre or 11.8 µg/dl), Kimber et
al. (1986) concluded that chronic lead exposure in man is not
associated with the immunological changes that have been observed in
rodent studies.
Coscia et al. (1987) found increased B-lymphocyte percentage and
absolute count in workers currently exposed to lead with PbB levels
exceeding 2.4 µmol/litre (> 50 µg/dl).
8.12.2 Children
A study of 12 pre-school children (Reigart & Graber, 1976) with
PbB levels > 1.92 µmol/litre (40 µg/dl) did not reveal altered
immunity in comparison with a control group.
8.13 Effects on bone
The pharmacokinetics of the transfer of bone lead to other target
organs and blood has been discussed by Smith & Hursh (1977), Marcus
(1985a,b), Silbergeld et al. (1988) and Rabinowitz (1991) (see section
6.2.2).
Bone homoeostasis depends upon a complex interaction of its
various components, i.e. minerals, cells and the extracellular matrix
composed of collagenous and non-collagenous proteins (Marks & Popoff,
1988; Sauk & Somerman, 1991). Early work on the effects of lead on
calcium homoeostasis and calcium-mediated function was reviewed by
Pounds (1984). More recent work has been summarized by Pounds et al.
(1991), particularly the effects of lead on hormone action,
competition with calcium binding on calcium messenger systems, and the
impaired synthesis of collagen or sialoproteins. Additional evidence
for adverse effects of lead on the proteins in the mineral
compartment, thus affecting bone homoeostasis, has been reported by
Sauk & Somerman (1991).
8.14 Biomarkers for lead effects
The understanding and application of biomarkers for the
assessment of effect is becoming more widespread (IPCS, 1994).
It is desirable to identify biomarkers of lead exposure and
effect to provide easily measurable parameters that will facilitate
the risk assessment process. At present PbB levels are frequently
measured to assess both exposure and effect. Alternative biomarkers
for lead which may be easily measured are of biochemical effects,
particularly in the haem biosynthetic pathway.
However, the relationship between these effects and neurological
impairment caused by lead has not been established. Measurement of
these effects has also been used to provide biomarkers for exposure.
Anaemia has been associated with high levels of lead exposures in
the occupational setting. However, clinical tests for haemoglobin are
rather non-specific indicators of lead toxicity (Bernard & Becker,
1988). The steps within the haem biosynthetic pathway which have been
used to measure effect are: 1) inhibition of delta-aminolaevulinic
acid dehydratase (ALAD); 2) urinary excretion of delta-aminolaevulinic
acid (ALAU); (3) the accumulation of zinc protoporphyrin (ZPP) in
erythrocytes arising from the inhibition of the enzyme ferrochelatase
or the iron transport system.
There are several methodological problems related to the
measurement of ALAD, and the benefit of measuring enzyme activity in
blood in lieu of PbB determinations is not apparent (Mushak, 1989).
Early work (Roels et al., 1976) indicated enzyme inhibition at PbB
levels of 0.24 µmol/litre (> 5 µg/dl). However, the data at levels of
0.72 µmol/litre (< 15 µg/dl) appear scattered (Hernberg & Nikkanen,
1970; Granick et al., 1973).
Levels of ALA in urine have been used as an effect indicator of
lead exposure (Meredith et al., 1978). ALAU levels were found to
correlate with PbB levels as low as 0.864 µmol/litre (18 µg/dl), the
correlation becoming much stronger at PbB levels above 1.92 µmol/litre
(40 µg/dl) (Selander & Cramer, 1970). Given the present concern over
effects in children at PbB levels well below 1.92 µmol/litre
(40 µg/dl), the assay of ALAU concentrations may not be sensitive
enough to be of any value.
The effects of lead on porphyrin levels, both coproporphyrin in
urine and zinc protoporphyrin (ZPP) in blood, have been investigated
as possible biomarkers for lead. Measurement of urinary coproporphyrin
levels does not appear to be sensitive enough to be useful in
assessing exposure to lead at present environmental levels. Excretion
levels do not rise significantly until PbB levels exceed
1.92 µmol/litre (40 µg/dl) (Meredith et al., 1978). Other studies
indicate that this assay may not be sensitive enough for biological
monitoring at PbB levels between 0.96 and 1.20 µmol/litre (20 and
25 µg/dl). At present it would appear that ZPP has limited usefulness
as a biological indicator of exposure at exposures leading to PbB
levels lower than 1.20 µmol/litre (25 µg/dl) (Roels et al., 1976;
Piomelli et al., 1982; Hammond et al., 1985; Marcus & Schwartz, 1987).
The results may also be confounded by concurrent iron deficiency which
will alter the levels of ZPP.
Vitamin D metabolism involves cytochrome P-450-dependent enzymes
and thus may be affected by lead exposure. Measurement of serum levels
of 1,25-dihydroxy-vitamin-D has been proposed as a sensitive
biological monitor of lead exposure in children (Rosen et al., 1980;
Mahaffey et al., 1982; Koo et al., 1991). However, it should be noted
that dietary intakes of calcium and phosphorus, as well as circulating
levels of parathyroid hormone will regulate the production and
circulating concentrations of this vitamin D metabolite, thus making
correlations with PbB levels difficult without adequate information on
the nutritional status of the population under study (Rosen & Chisney,
1983). In populations showing adequate nutritional status, lead at
exposures leading to PbBs levels lower than 0.96 µmol/litre (20 µg/dl)
does not appear to have a demonstrable effect on circulating levels of
1,25-dihydroxy-vitamin-D.
9. EVALUATION OF HUMAN HEALTH RISKS
9.1 Exposure assessment
Lead is a ubiquitous element detected in all environmental media.
However, natural sources contribute only a small fraction of the
amounts of lead found in air, food, water and dust. The majority of
lead in these media arises from automobile and industrial emissions
and from the use of lead-containing solder and paints. Adults and
older children receive the largest proportion of lead intake from
foods, whereas dust, soil and food all make significant contributions
to the total lead intake of young children. The major contributions to
lead in soil and outdoor dust are from the combustion of fossil fuels
(principally leaded petrol), stationary sources such as smelters, and
peeling and flaking of lead-based paint.
9.1.1 General population exposure
In the absence of specific stationary sources of lead,
concentrations in ambient air are directly related to density of
traffic and whether lead is still utilized as an additive in petrol.
Reduction or elimination of lead in petrol in those countries which
have instituted regulations has resulted in a decline by as much as
eight-fold in ambient air concentrations of lead.
Levels of lead in indoor air are affected by the presence of
cigarette smoke and dust from lead-painted surfaces. Without such
sources, air lead levels indoors are about 60% of those in outdoor
air.
For most adults, the total daily exposure to lead is via food,
water and air. For infants aged up to 5 months, formula or breast milk
and water are the main sources of lead. In children, an additional
source of exposure is dust and soils. Absorption is dependent on the
chemical form of lead, type of soil and particle size
(bioavailability). Lead intake may be augmented from unusual sources
such as folk remedies, cosmetics and hobby activity. Community
contamination and workplace practices may contribute to lead exposure.
Food (including drinking-water and beverages) is the major source
of lead exposure for the general population. Infants and children may
receive an added lead burden from soil and dust. The most significant
foodstuffs will vary from country to country. In areas still utilizing
lead-soldered cans, levels of lead are substantially higher. Depending
upon lifestyles, there may be significant oral intake of lead from
some alcoholic beverages and due to the leaching of lead from low
temperature-fired ceramic containers.
Most drinking-water supplies contain lead levels lower than
5 µg/litre when they leave the treatment plant. However, where the
water is known to be plumbo-solvent, up to 40% of the samples may
exceed 100 µg/litre in homes where lead solder, lead pipes or brass
fixtures have been used.
Absorption of lead from the lung is a function of particle size
and pulmonary deposition pattern. Small particles (< 0.5 µm in
diameter) characteristic of ambient air will be deposited deeply in
the lungs with absorption rates of 90%. Larger particles, such as
those that may be encountered in occupational settings, exhibit high
deposition rates in the upper airway. Absorption of such particles
will be a function of both dissolution in the lung and particle
clearance to the gastrointestinal tract.
Human dermal absorption of inorganic lead through unabraded skin
is of limited significance.
9.1.2 Occupational exposures
In addition to exposure to lead in ambient air, water and food,
some workers may be exposed to airborne lead and dust within the
workplace. Actual levels will vary according to the engineering design
of the process equipment and the industrial hygiene practices
utilized.
9.2 Critical issues related to exposure evaluation
In view of the heterogeneity of responses to lead within human
populations, the complex interrelationships between exposure to lead
and a biological indicator for internal dose require consideration of
several key issues in order to assess human exposures.
9.2.1 Sampling and analytical concerns
Reliable comparisons of reported levels of exposure and/or dose
can only be made where authors have described the analytical and
sampling procedures in sufficient detail to allow the reader to
assess, for example, the integrity of the sampling procedures as well
as the specificity, precision and accuracy of all analytical methods.
Problems related to sampling cord blood also need to be considered.
Most studies have utilized analytical procedures of high quality.
However, consideration must be given to blood collection procedures
(finger stick versus venepuncture) when comparing results.
9.2.2 Data presentation
Inter-study comparisons of lead exposures are complicated by the
variety of methods used to present results, including median values as
well as geometric and arithmetic means. Some authors have used
log-transformed data.
In assessing exposure from data on teeth, one must know which
tooth and which compartment(s) within the tooth were sampled. In
addition, if data are to be compared between studies, authors must
state explicitly that all teeth analysed were without caries and were
shed spontaneously.
The exposure index lifetime average blood lead (PbB) level has
assisted in the assessment of cumulative exposures from serial PbB
data. It should not be interpreted as being equivalent to a single PbB
determination at a single point in life.
For some data/analytical purposes, age-specific PbB levels may be
more appropriate than a lifetime average.
9.3 Relationship between exposure and dose
The most widely used surrogate for the absorbed dose is whole PbB
concentration.
The relationship between PbB level and lead intake is curvilinear
over a wide range of PbB values. On the basis of a single study of 17
infants, the relationship between PbB level and lead intake from food
has been determined to be 0.0077 µmol lead/litre (0.16 µg/dl) per µg
lead intake per day for a median PbB level of approximately
0.48 µmol/litre (10 µg/dl).
Most studies of the relationship between PbB level and lead
exposure apply to a single environmental source, i.e. air, food, water
or soil/dust. A summary of the relationship between PbB level and lead
intake from individual media is given in Table 23.
9.4 Surrogate measures of dose
9.4.1 Blood
Whole PbB values are widely used as a measure of absorbed dose.
However, it is believed that plasma lead concentrations may better
reflect the "active" fraction of lead in blood and define the
relationship between PbB and tissue or organ accumulation (and
Table 23. Representative relationships of blood lead median level to intake of
lead for the general populationa
Population
Median Children Adults
Airb 0.09 µmol Pb/litre per µg 0.079 µmol Pb/litre per µg
Pb/m3 (1.92 µg Pb/dl) Pb/m3c (1.64 µg Pb/dl)
Water 0.003 µmol Pb/litre per µg
Pb/litre (0.06 µg Pb/dl)
Food 0.01 µmol Pb/litre per µg 0.002-0.003 µmol Pb/litre per
Pb/day (0.16 µg Pb/dl) µg Pb/day (0.04-0.06 µg Pb/dl)
Dustb 0.09 µmol Pb/litre per 1000 µg
Pb/g (1.8 µg Pb/dl)
Soilb 0.11 µmol Pb/litre per 1000 µg
Pb/g (2.2 µg Pb/dl)
a These data are provided for illustrative purposes only recognizing that the
relationships are curvilinear in nature and are broad guidelines which will
not apply at lower or higher levels of exposure.
b A value between 0.144 to 0.24 µmol Pb/litre or 3-5 µg Pb/dl per µg/m3
is obtained when one considers indirect contribution through deposition on
soil/dust.
c The air to blood lead relationship in occupational settings is best described
by a curvilinear relationship having slopes between 0.02 and 0.08 µg/m3
air. The slope is variable but lower than that found for humans in the general
environment, which is between 1.6 and 1.9 µg/m3.
effect), although there is little experimental data (because of
analytical limitations). PbB is distributed between plasma and
erythrocytes, with less than 5% being in the plasma. Most of the lead
is bound to haemoglobin.
Venous and capillary blood levels are generally equivalent,
provided that the sampling technique is adequate.
9.4.2 Urine
Urinary measurements of lead concentration are of limited value,
although they are used occasionally as a screening test for
occupational population groups.
9.4.3 Bone
Bone lead may be measured by non-invasive X-ray fluorescence, but
this technique is limited in sensitivity at present.
9.4.4 Tooth
Shed deciduous teeth have been used to provide an index of
exposure in early childhood. Interpretation of the analytical data is
dependent on the type of tooth and the part of the tooth (whole tooth,
dentine or circumpulpar dentine) analysed.
9.4.5 Hair
Hair is not useful for measurement of lead exposure.
9.5 Biochemical effects of lead
9.5.1 Haem synthesis
Evaluation of the quality of analytical data is an important
aspect in considering reports describing effects attributed to lead.
It should be noted that much of the data presented in this area has
not been as vigorously scrutinized as, for example, psychometric study
data.
An increase in erythrocyte protoporphyrin (EP) in children occurs
between PbB levels of 0.72 and 1.2 µmol/litre (15-25 µg/dl). Increases
in EP can be detected in men when the PbB level is above
1.20-1.44 µmol/litre (25-30 µg/dl), and in women when it is above
0.96-1.44 µmol/litre (20-30 µg/dl). It should be noted that the effect
of lead on haem is confounded by low iron status.
9.5.1.1 Urinary coproporphyrin
The coproporphyrin concentration in urine increases significantly
with PbB levels in excess of 1.92 µmol/litre (40 µg/dl).
9.5.1.2 Urinary aminolaevulinic acid in children
In children 1-5 years old, there is a linear relationship with
PbB in the range 1.2-3.6 µmol/litre (25-75 µg/dl). Data for children
with PbB levels of 0.24-1.92 µmol/litre (5-40 µg/dl) show essentially
no correlation with urinary aminolaevulinic acid (ALA) excretion.
Elevation of urinary ALA level is evident at a PbB level of about
1.68 µmol/litre (35 µg/dl).
9.5.1.3 Urinary aminolaevulinic acid in adults
Urinary excretion of ALA increases in men at PbB levels above
2.16 µmol/litre (45 µg/dl) and in women above 1.68 µmol/litre
(35 µg/dl).
9.5.1.4 delta-Aminolaevulinic acid dehydratase
There was a negative exponential relationship between PbB level
and delta-aminolaevulinic acid dehydratase (ALAD) activity in a
population of 10- to 13-year-old children with PbB levels in the range
of 0.19-1.97 µmol/litre (4.7-41 µg/dl). An effect was seen at a PbB
level of approximately 0.48 µmol/litre (10 µg/dl). There is an
apparent lack of a clearly defined threshold for lead inhibition of
ALAD in different age groups.
9.5.2 Vitamin D metabolism
In the presence of adequate nutritional status, PbB levels below
0.96 µmol/litre (20 µg/dl) appear to have no demonstrable effect on
circulating concentrations of 1,25-dihydroxy-vitamin-D. A PbB level
above 0.96 µmol/litre (20 µg/dl) is associated with a decrease in the
serum level of 1,25-dihydroxy-vitamin-D.
9.5.3 Dihydrobiopterin reductase
Inhibition of dihydrobiopterin reductase has been shown in humans
where the mean PbB level is as low as 0.48 µmol/litre (10 µg/dl).
A summary of some biochemical effects of lead is presented in
Table 24.
9.5.4 Haemopoietic system
9.5.4.1 Anaemia in adults
The estimated PbB associated with a decrease in haemoglobin
concentration is 2.40 µmol/litre (50 µg/dl).
9.5.4.2 Anaemia in children
Decreased haemoglobin levels in children occur at a PbB level of
approximately 1.92 µmol/litre (40 µg/dl). Anaemia, defined as a
haematocrit below 35%, is not found at a PbB level of less than
0.92 µmol/litre (20 µg/dl). The risk of having a haematocrit value
below 35% for a 1-year-old child is 2% at a PbB level of
Table 24. Biochemical effects of lead
Parameter Blood lead level above which the
biochemical effect is demonstrable with
current techniques
µmol/litre µg/dl
Protoporphyrin levels 0.96-1.44 20-30
Coproporphyrin levels 1.92 40
ALA urine levels 1.68 35
ALAD activity 0.48 10
1,25-dihydroxy-vitamin-D 0.96 20
Dihydrobiopterin reductase 0.48 10
0.96-1.87 µmol/litre (20-39 µg/dl); the contribution of iron
deficiency may account for a substantial proportion of this 2%.
Induction of anaemia is demonstrable at 1.92 µmol/litre (40 µg/dl).
9.5.4.3 Erythrocyte pyrimidine-5'-nucleotidase
A reduction of 20% or more in erythrocyte pyrimidine
-5'-nucleotidase activity is associated with PbB concentrations above
0.48 µmol/litre (10 µg/dl).
Effects of lead are demonstrable on a number of enzyme systems
and biochemical parameters. The PbB levels, above which effects are
demonstrable with current techniques for the parameters which may have
clinical significance, are all greater than 0.96 µmol/litre
(20 µg/dl). Some clinically insignificant effects on enzymes are
demonstrable at lower levels of PbB.
9.6 Nervous system
9.6.1 Adults
9.6.1.1 Central nervous system
With acute lead exposure resulting in a PbB level in excess of
3.84 µmol/litre (80 µg/dl), severe encephalopathy and/or coma may
occur. Central nervous system (CNS) symptoms are found in lead-exposed
adults when there is a history of several years of exposure to lead at
PbB levels that may not have exceeded 3.36 µmol/litre (70 µg/dl) and a
PbB level at the time of clinical assessment of at least
1.92 µmol/litre (40 µg/dl).
Impaired neurobehavioural test performance has been found in
lead-exposed workers. Changes in critical flicker fusion test have
been detected at a PbB level of about 2.4 µmol/litre (50 µg/dl).
Sensory motor function is generally more sensitive than cognitive
end-points in many neurobehavioural evaluations, the lowest-observed-
effect level being at about 1.92 µmol/litre (40 µg/dl).
It appears also that neuroelectrophysiological tests are
sensitive indicators of the CNS effects of lead. Reductions in
latencies of sensory evoked potentials and auditory event-related
potentials have been found in workers with average PbB levels of
approximately 1.92 µmol/litre (40 µg/dl).
9.6.1.2 Peripheral nervous system
Numerous studies have measured the conduction velocity of
electrically stimulated sensory and motor nerves in workers exposed to
lead. These nerve conduction velocity (NCV) studies have yielded
somewhat mixed results, with many showing a decrease in NCV in
relation to lead exposure (indexed as PbB level) and a few showing no
effect or occasionally even an increase in NCV associated with lead
exposure. Differences in the nerves evaluated, methodologies,
characterization of lead exposure, and control of confounding
variables underlie some of the variability in results across studies.
A statistical meta-analysis of 32 NCV studies has indicated that NCV
is significantly reduced in lead-exposed workers compared to reference
subjects, but that the median motor nerve shows more reliable effects
of lead than other nerves. This collective view of the evidence is
supported by key studies that provide compelling evidence of a causal
relationship between lead exposure and reductions in NCV, extending to
PbB levels as low as 1.44 µmol/litre (30 µg/dl). These effects may be
reversible depending on the duration and level of exposure.
9.6.1.3 Autonomic nervous system
Two reports examining the electrocardiographic RR interval
variability during deep breathing and the component CV of respiratory
sinus arrythmia demonstrated dysfunctions at an average PbB level of
1.68 µmol/litre (35 µg/dl). These results suggest autonomic nervous
system dysfunction, particularly the parasympathetic nervous system.
9.6.2 Children
Prospective and cross-sectional studies of children have
demonstrated associations of lead exposure, measured by various
indices, and intellectual performance. The association has been noted
across a wide range of exposure levels and in a variety of populations
before factors other than lead have been accounted for.
A key question is whether this statistical association is
directly attributable to the causal influence of lead on child IQ. It
is important to consider alternative contributory explanations, i.e.
random chance, unexplained confounding factors, reverse causality and
selection bias.
It is a matter of debate and conjecture as to the extent to which
these four issues should inhibit claims of a causal relationship in
the epidemiological studies. The essential problem is that
observational epidemiology cannot provide definitive evidence of
causality when the key statistical association is small, the temporal
relationship is unclear and major confounders are present. Animal
studies provide qualitative support for the claim of a causal role for
lead in affecting neuropsychological performance, but provide limited
assistance in establishing quantitative dose-effect relationships.
9.6.2.1 Type of effect
The clearest and most consistent associations have been found
with global measures, such as IQ, where the largest body of evidence
is available. Efforts to delineate the neuropsychological foundations
of this association with a wide variety of tests of specific
neuropsychological domains have not so far been successful.
9.6.2.2 Magnitude
Based on the evidence from cross-sectional and prospective
studies of populations with PbB levels generally below 1.2 µmol/litre
(25 µg/dl), the size of the apparent IQ effect (at ages 4 and above)
is a deficit of between 0 and 5 points (on a scale of 100 with a
standard deviation of 15) for each 0.48 µmol/litre (10 µg/dl)
increment in PbB level, with a likely apparent effect size of between
1 and 3 points. At PbB levels above 1.2 µmol/litre (25 µg/dl), the
relationship between PbB level and IQ may differ. Estimates of effect
size are group averages and only apply to the individual child in a
probabilistic manner.
Existing epidemiological studies do not provide definitive
evidence of a threshold. Below the PbB range of 0.48-0.72 µmol/litre
(10-15 µg/dl), the effect of confounding variables and limits in the
precision of analytical and psychometric measurements increases the
uncertainty attached to any estimate of effect. However, there is some
evidence of an association below this range.
9.6.2.3 Reversibility/persistence
Whilst the IPCS Task Group could not unequivocally state that
effects of early childhood exposure are persistent beyond childhood,
because the current data are too meagre, it was held that
neurobehavioural effects detected at age seven or later usually
persist. Measures in later childhood tend to be more predictive of
subsequent performance than those made earlier. It is more likely than
not that effects seen during school years are to some degree
irreversible. This has also been observed in later follow-up studies
conducted in other non-lead topics of child development research. One
of the difficulties is that there are too few studies concerning
long-term outcome in children with high early exposures and where the
sources of exposure are subsequently removed.
Virtually no useful data are available on the effects on IQ of
removing children from a "high" exposure environment to one of "low"
exposure or on reduction of body lead burden in children. This is not
to say that exposure should not be reduced when possible.
9.6.2.4 Age-specific sensitivity
From prospective studies it is not possible to determine an age
of critical sensitivity. This reflects the findings that serial PbB
measures taken at the age of 2 years and later are positively
correlated with, the individual rankings remaining approximately
constant, and this limits the ability to identify sensitive periods of
exposure.
9.6.2.5 Interactions/subgroups
The evidence is inconclusive on whether apparent effects are more
or less marked in different gender or socioeconomic status (SES)
subgroups. However, where there are suggestions of SES-related
differences, the apparent effects tend to be more marked in the lower
SES subgroups.
9.6.3 Animal studies
Experimental animal studies of CNS effects provide support for
the associations between PbB levels and neurobehavioural deficits
described in human epidemiological studies of lead. There is
supportive evidence both in terms of demonstrating causal
relationships and in the levels of PbB at which such effects are
observed, namely 0.528-0.72 µmol/litre (11-15 µg/dl). Moreover, they
provide qualitative parallels in the nature of the effects described,
as these effects include changes in learning and memory functions.
Experimental animal studies indicate that these CNS effects may depend
upon task complexity and can persist long beyond the termination of
lead exposure. These studies also provide information possibly
relevant to understanding mechanisms of effect. In addition, the
experimental animal studies provide such evidence in the absence of
the confounding factors and co-variates, such as parental IQ,
socioeconomic status, and quality of the home environment, that are
problematic to human epidemiological endeavours, and in the absence of
nutritional deficiencies that may arise in human populations.
9.7 Renal system
Renal function impairment was not associated with a PbB level
below 3.0 µmol/litre (62 µg/dl) when measured by blood urea nitrogen
and serum creatinine levels in lead workers. However, renal tubular
effects were detected in workers with a PbB level below 3.0 µmol/litre
when measured by more sensitive indicators such as NAG.
Most studies of the general population attempting to relate renal
function impairment to PbB concentration have not demonstrated an
effect with PbB levels below 1.8 µmol/litre (37.3 µg/dl). More
sensitive indicators of renal function may indicate a renal effect of
lead below this level.
9.8 Liver
Over-exposure to lead may inhibit drug metabolism in the liver.
9.9 Reproduction
9.9.1 Female
Studies on the risk of spontaneous abortion and reduced birth
weight associated with maternal PbB levels below 1.44 µmol/litre
(30 µg/dl) have yielded mixed results. Recent epidemiological studies
have shown exposure-related perturbations in the length of gestation,
significantly greater risks being associated with PbB levels of
0.72 µmol/litre (15 µg/dl) or more.
9.9.2 Male
PbB concentrations above 1.92 µmol/litre (40 µg/dl) have been
shown to affect sperm morphology and function. At present, the
reproductive consequences of these changes are unknown.
9.10 Blood pressure
A quantitative assessment of the collective evidence from all the
observational studies in adults is made difficult by the fact that
studies have adopted different policies regarding adjustment for
potential confounding factors (e.g., alcohol consumption). In
addition, quantitative findings from the two largest studies (BRHS and
NHANES II) have depended on whether adjustment was made for
geographical variations in blood pressure and blood lead.
The limited size of most observational studies has inevitably
meant that they could not consistently demonstrate a statistically
significant relationship. However, an overview of all the studies
shows that evidence is consistent with the centre-adjusted analysis of
the two main studies, i.e. there are very weak but statistically
significant associations between PbB level and both systolic and
diastolic blood pressures. The likely order of magnitude is that for
any two-fold increase in PbB level (e.g., from 0.8 to 1.6 µmol per
litre) there is a mean 1 mmHg increase in systolic blood pressure. The
association with diastolic blood pressure is of a similar magnitude.
Animal studies have provided plausible mechanisms for an effect
of lead on blood pressure. However, from such a small magnitude of
statistical associations in the presence of important confounders, one
cannot infer that low-level lead exposure is causally related to an
increase in blood pressure.
The two population studies relating PbB to cardiovascular disease
events show no statistically significant associations. Hence, there is
no clear evidence to suggest that lead has an impact of public health
importance as regards hypertension or risk of cardiovascular disease.
9.11 Carcinogenicity
Renal tumours occur in rats and mice administered high doses of
lead. However, the evidence for the carcinogenicity of lead and
inorganic lead compounds in humans is inadequate.
9.12 Immune system
There is no strong evidence in humans of an effect of lead on the
immune system.
10. RECOMMENDATIONS FOR PROTECTION OF HUMAN HEALTH
10.1 Public health measures
Public health measures should be directed towards reduction and
prevention of exposure to lead by reducing the use of lead and lead
compounds and by minimizing lead-containing emissions that result in
human exposures. This can be achieved by:
a) phasing-out any remaining uses of lead additives in motor fuels;
b) further reducing the use of lead-based paints, with the objective
of eliminating such paints;
c) development and application of methods for the safe and
economical remediation of lead-painted homes and lead
contaminated soil;
d) elimination of the use of lead in food containers (e.g., in the
seams of cans);
e) dissemination of information to assist with identification of
glazed food containers which may leach lead into food placed,
cooked or stored in the container;
f) eliminating any remaining agricultural uses of lead or lead
compounds (e.g., lead arsenate as an insecticide);
g) identifying and reducing, or preferably, eliminating lead found
as a contaminant or ingredient of folk remedies and cosmetics;
h) the use of materials and engineering practices to minimize
plumbosolvency in water treatment and water distribution systems;
i) systematic examination of processes in which lead is used or
recycled in order to identify and reduce lead exposure by means
of improved engineering design, of operators, by-standers and the
environment. Opportunities for technology transfer should be used
whenever possible.
10.2 Public health programmes
Programmes should be developed:
a) to enhance data collection and to make available to the public
information on the lead content of foodstuffs;
b) to facilitate identification of populations at high risk of
exposure to lead on the basis of monitoring data for lead in
food, air, water and soil;
c) that incorporate improved procedures for health risk assessment
of population groups at risk of exposure to lead;
d) that promote understanding and awareness concerning the effects
on human health associated with exposure to lead, while
recognizing cultural sensitivities;
e) that place emphasis on adequate nutrition, health care and
attention to socioeconomic conditions which may exacerbate the
effects of lead present in the environment.
10.3 Screening, monitoring and assessment procedures
Methods for evaluating the effects and associated risks of
exposure to lead require both improvement and further development or
research. In the short term the following measures are needed.
a) Screening
i) Blood lead measurements should be recognized as the biomarker of
choice for screening for previous exposure of children to lead.
ii) The sensitivity of the developing nervous system to the
potentially harmful effects of lead is such that other
biochemical measurements (e.g., erythrocyte protoporphyrin) are
not sufficiently sensitive for assessment of infants and
children.
b) Monitoring
i) More sensitive analytical methods should be developed for the
reliable measurement of blood lead levels below 0.72 µmol/litre
(15 µg/dl) to acceptable standards of precision and accuracy.
ii) There is a need for international analytical quality assurance
programmes utilizing reference lead-containing materials.
iii) All publications containing blood lead measurement data should
provide adequate data on current quality assurance and quality
control.
iv) Data comparisons are made more difficult by differences in units
and statistical techniques for data handling. Investigators
should be encouraged to adopt internationally agreed practices
(e.g., IUPAC units).
c) Assessment
i) Validated biomarkers are needed to define the relationship
between measures of environmental (external) exposure and
specific effects (biochemical or functional).
ii) Biomarkers indicative of deficits in cognitive performance are
needed to facilitate assessment of risk.
iii) Improved methods are needed for the definition of outcome effects
(especially IQ and neurobehavioural deficits) attributable to
lead at blood lead concentrations of about
0.48 µmol/litre (10 µg/dl) or less.
iv) Further data are required to determine whether outcome effects on
the nervous system attributed to lead are reversible or
permanent.
v) Biomarkers for the renal effects of lead are needed to link renal
damage with lead exposure and thereby improve assessment of risk
of renal damage.
11. FURTHER RESEARCH
a) Research is required to improve the process for the assessment of
risk associated with exposure to lead. Specifically this should
cover:
i) definition of the health significance of biochemical
changes associated with exposure to lead, with particular
attention to alterations associated with blood lead
concentrations of about 0.72 µmol/litre (15 µg/dl) or
less;
ii) work to define the bioavailability of lead from different
sources and to establish the relationship between exposure
(sources and speciation) and body burden;
iii) definition of the influence of host-related factors
(particularly nutrition) affecting absorption and
distribution of lead;
iv) intensification of kinetic studies of lead to provide an
improved data base for extrapolation between species;
v) elucidation of mechanisms of accumulation and mobilization
of lead from bone with particular attention to the
influence of pregnancy and ageing on kinetics;
vi) investigation of the pharmacokinetics of lead in the
pregnant female in relation to transfer of lead to the
developing embryo and fetus and factors that mitigate such
transfer;
vii) determination of the effects of pre- and post-natal
exposure to lead;
viii) improved definition of paternally mediated effects of lead
exposure on the reproductive process and outcomes.
b) More general research needs include the following:
i) rationalization of neurobehavioural tests used to assess
performance in children and animals in order to permit
comparisons of measurements and test data that use similar
biological mechanisms;
ii) follow-up research (where possible) of historical cohorts
of lead-poisoned adults to examine the sequelae;
iii) evaluation of intervention measures, such as lead
abatement from home and soil, and chelation on reduction
of blood lead levels and outcome effects on the central
nervous system.
12. PREVIOUS EVALUATIONS BY INTERNATIONAL BODIES
The carcinogenic potential of lead and lead compounds was last
evaluated by the International Agency for Research on Cancer in 1987
(IARC, 1987b). There was inadequate evidence for the carcinogenicity
of lead and inorganic lead compounds in humans, but sufficient
evidence was available to show that specified inorganic lead compounds
were carcinogenic in experimental animals. The overall evaluation
placed lead and inorganic lead compounds in Group 2B, i.e. possibly
carcinogenic to humans.
To protect workers from the adverse effects of lead on haem
synthesis and on the peripheral and central nervous system, a
health-based biological exposure limit of 1.92 µmol/litre (40 µg/dl)
was recommended by a WHO Study Group (WHO, 1980). It was further
recommended that blood lead (PbB) levels in women within the
reproductive age range should not exceed 1.44 µmol/litre (30 µg/dl).
Depending upon the background levels of PbB in the worker population,
air lead levels should not exceed 30-60 µg/m3 (WHO, 1980).
A drinking-water guideline value of 0.050 mg/litre was developed
in 1984 (WHO, 1984). This guideline value has been revised recently to
0.01 mg/litre (WHO, 1993).
Lead was evaluated by a WHO Working Group developing air quality
guidelines for Europe (WHO, 1987). Based on the assumption that PbB
levels in 98% of the population would be maintained at levels below
0.96 µmol/litre (20 µg/dl), a guideline value in the range of
0.5-1.0 µg/m3 (long-term average, such as annual mean) was
recommended.
At the 41st meeting of the Joint FAO/WHO Expert Committee on Food
Additives and Food Contaminants, a Provisional Tolerable Weekly Intake
(PTWI) of 25 µg/kg body weight was recommended (FAO/WHO, 1993). This
level refers to lead from all sources and was set to protect all
humans, including infants and children. It was based on a model
indicating daily intakes of lead between 3-4 µg/kg body weight by
infants and children and is not associated with an increase in PbB
concentrations.
Regulatory standards established by national bodies in several
countries and the European Economic Community are summarized in the
legal file of the International Register of Potentially Toxic
Chemicals (IRPTC, 1987).
REFERENCES
Aguilera de Benzo Z, Fraile R, & Carrion N (1989) Determination of
lead in whole blood by electrothermal atomization atomic absorption
spectrometry using tube and platform atomizers and dilution with Trito
X-100. J Anal At Spectrom, 4: 397-400.
Albahary C (1972) Lead and hemopoiesis: The mechanism and consequences
of the erythropathy of occupational lead poisoning. Am J Med,
52: 367-378.
Albert LA & Badillo F (1991) Environmental lead in Mexico. Rev Environ
Contamin Toxicol, 117: 1-49.
Alexander FW, Delves HT, & Clayton BE (1973) The uptake and excretion
by children of lead and other contaminants. In: Barth D, Berlin A,
Engel R, Recht P, & Smeets J ed. Environmental health aspects of lead:
Proceedings, International Symposium, Amsterdam, The Netherlands,
October 1972. Luxembourg, Commission of the European Communities,
pp 319-331.
Al-Hakkak ZS, Hamamy HA, Murad AMB, & Hussain AF (1986) Chromosome
aberrations in workers at a storage battery plant in Iraq. Mutat Res,
171: 53-60.
Altmann L, Weinsberg F, Sveinsson K, Lilienthal H, Wiegand H, &
Winneke G (1993) Impairment of long-term potentiation and learning
following chronic lead exposure. Toxicol Lett, 66(1): 105-112.
Alvares AP, Kapelner S, Sassa S, & Kappas A (1975) Drug metabolism in
normal children, lead-poisoned children, and normal adults. Clin
Pharmacol Ther, 17: 179-183.
Anderson C & Danylchuk KD (1977) The effects of chronic low level lead
intoxication on the Haversian remodeling system in dogs. Lab Invest,
37: 466-469.
Angle CR & McIntire MS (1978) Low level lead inhibition of erythrocyte
pyrimidine nucleotidase. Environ Res, 17: 296-302.
Angle CR & McIntire MS (1979) Environmental lead and children: the
Omaha study. J Toxicol Environ Health, 5: 855-870.
Angle CR, McIntire MS, & Swanson MS (1982) Erythrocyte nucleotides in
children - increased blood lead and cytidine triphosphate. Pediatr
Res, 16: 331-334.
Annest JL & Mahaffey KR (1984) Blood lead levels for persons aged 6
months-74 years: United States, 1976-1980. Hyattsville, Maryland, US
Public Health Service (Report No. 223, Series 11) (DHHS Publication
No. (PHS) 84-1683).
Annest JL, Pirkle JL, Makuc D, Neese JW, Bayse DD, & Kovar MG (1983)
Chronological trend in blood lead levels between 1976 and 1980. N Engl
J Med, 308: 1373-1377.
Araki S & Ushio K (1982) Assessment of the body burden of chelatable
lead: a model and its application to lead workers. Br J Ind Med,
39: 157-160.
Araki S, Honma T, Yanagihara S, & Ushio K (1980) Recovery of slowed
nerve conduction velocity in lead-exposed workers. Int Arch Occup
Environ Health, 46: 151-157.
Araki S, Aono H, Fukahori M, & Tabuki K (1984) Behaviour of lead and
zinc in plasma, erythrocytes, and urine and ALAD in erythrocytes
following intravenous infusion of CaEDTA in lead workers. Arch Environ
Health, 39: 363-367.
Araki S, Murata K & Aono H (1986a) Subclinical cervico-spino-bulbar
effects of lead: A study of short-latency somatosensory evoked
potentials in workers exposed to lead, zinc and copper. Am J Ind Med,
10: 163-175.
Araki S, Yokoyama K, Murata K, & Aono H (1986b) Determination of the
distribution of conduction velocities in workers exposed to lead, zinc
and copper. Br J Ind Med, 43: 321-326.
Araki S, Aono H, Murata K (1986c) Mobilisation of heavy metals into
the urine by CaEDTA: Relation to erythrocyte and plasma concentrations
and exposure indicators. Br J Ind Med, 43: 636-641.
Araki S, Murata K, & Aono H (1987) Central and peripheral nervous
system dysfunction in workers exposed to lead, zinc and copper: A
follow-up study of visual and somatosensory evoked potentials. Int
Arch Occup Environ Health, 59: 177-187.
Araki S, Murata K, Yokoyama K, & Uchida E (1992) Auditory
event-related potential (P300) in relation to peripheral nerve
conduction in workers exposed to lead, zinc, and copper: Effects of
lead on cognitive function and central nervous system. Am J Ind Med,
21: 539-547.
Arnetz BB & Nicolich MJ (1990) Modelling of environmental lead
contributors to blood lead in humans. Int Arch Occup Environ Health,
62: 397-402.
Aslam M, David SS, & Healy MA (1979) Heavy metals in some Asian
medicines and cosmetics. Publ Health (Lond), 93: 274-284.
Assenato G, Paci C, Baser M, Molinini R, Candela RG, Altamura BM, &
Giorgino R (1987) Sperm count suppression without endocrine
dysfunction in lead-exposed men. Arch Environ Health, 42: 124-127.
ATSDR (1993) Toxicological profile for lead. Atlanta, Georgia, US
Department of Health and Human Services, Agency for Toxic Substances
and Disease Registry (TP-92/12).
Aungst BJ, Docle JA, & Fung H (1981) The effect of dose on the
disposition of lead in rats after intravenous and oral administration.
Toxicol Appl Pharmacol, 61: 48-57.
Awad el Karim MA, Hamed AS, Elhaimi YAA, & Osman Y (1986) Effects of
exposure to lead among lead acid battery factory workers in Sudan.
Arch Environ Health, 41: 261-265.
Azar A, Trochimowicz HJ, & Maxfield ME (1973) Review of lead studies
carried out at Haskell Laboratory: two year feeding study and response
to hemorrhage study. In: Environmental health aspects of lead.
Proceedings of an international symposium, Amsterdam. Luxembourg,
Commission of the European Communities, pp 199-210.
Baghurst PA, McMichael AJ, Wigg N, Vimpani G, Robertson EF, Roberts
RJ, & Tong S-L (1992) Life-long exposure to environmental lead and
children's intelligence at age seven: the Port Pirie cohort study.
N Engl J Med, 327: 1269-1284.
Baker EL, Landrigan PJ, Barbour AG, Cox DH, Folland DS, Ligo RN, &
Throckmorton J (1979) Occupational lead poisoning in the United
States: clinical and biochemical findings related to blood lead
levels. Br J Ind Med, 36: 314-322.
Baker EL, Goyer RA, Fowler BA, Khettry U, Bernard DB, Adler S, White
R, Babayan R, & Feldman RG (1980) Occupational lead exposure,
nephropathy and renal cancer. Am J Ind Med, 1: 139-148.
Barrio A & Badia J (1985) [Lead encephalopathy in adults.] Rev Med
Chil, 113(6): 564-567 (in Spanish).
Barltrop D (1969) Mineral metabolism in pediatrics. Oxford, London,
Edinburgh, Melbourne, Blackwell Scientific Publishers.
Barltrop D (1972) Children and environmental lead. In: Hepple P ed.
Lead in the environment: Proceedings of a conference. London,
Institute of Petroleum, pp 52-60.
Barry PSI (1975) A comparison of concentrations of lead in human
tissues. Br J Ind Med, 32: 119-139.
Barry PSI (1981) Concentrations of lead in the tissues of children.
Br J Ind Med, 38: 61-71.
Barton JC, Conrad ME, Nuby S, & Harrison L (1978) Effects of iron on
the absorption and retention of lead. J Lab Clin Med, 92: 536-547.
Bauchinger M, Dresp J, Schmid E, Englert N, & Krause C (1977)
Chromosome analyses of children after ecological lead exposure. Mutat
Res, 56: 75-79.
Beattie AD, Moore MR, Goldberg A, Finlayson MJ, Graham JR, Mackie TM,
Main J, McLaren PA, Murdock KM, & Stewart FT (1975) Role of chronic
lead exposure in the aetiology of mental retardation. Lancet,
1: 589-592.
Beckett PH, Davis RD, & Brindley P (1979) The disposal of sewage
sludge onto farmland: the scope of the problem of toxic elements.
Water Pollut Control, 78: 419-436.
Behari LR, Singh S, Tandon SK, & Wahal AK (1982) Lead poisoning among
Indian silver jewellery makers. Ann Occup Hyg, 27: 107-109.
Behne D (1980) Problems of sampling and sample preparation for trace
element analysis in the health sciences. In: Bratter P & Schramel P
ed. Trace element analytical chemistry in medicine and biology.
Proceedings of the First International Workshop, Neuherberg, Federal
Republic of Germany. Berlin, New York, Walter de Gruyter & Co.,
pp 769-782.
Beijer K & Jernelöv A (1984) Microbial methylation of lead. In:
Grandjean P ed. Biological effects of organolead compounds. Boca
Raton, Florida, CRC Press, pp 13-19.
Bellinger DC & Stiles KM (1993) Epidemiologic approaches to assessing
the developmental toxicity of lead. Neurotoxicology, 14: 151-160.
Bellinger DC, Needleman HL, Leviton A, Waternaux C, Rabinowitz MR, &
Nichols ML (1984) Early sensory-motor development and prenatal
exposure to lead. Neurobehav Toxicol Teratol, 6: 387-402.
Bellinger DC, Leviton A, Waternaux C, Needleman H, & Rabinowitz M
(1987) Longitudinal analyses or prenatal and post-natal lead exposure
and early cognitive development. N Engl J Med, 316: 1037-1043.
Bellinger D, Sloman A, Leviton A, Rabinowitz M, Needleman H, &
Waternaux C (1991a) Low-level lead exposure and children's cognitive
function in the preschool years. Pediatrics, 87: 219-227.
Bellinger D, Leviton A, Rabinowitz M, Allred E, Needleman H,
Schoenbaum S (1991b) Weight gain and maturity in fetuses exposed to
low levels of lead. Environ Res, 54: 151-158.
Bellinger DC, Stiles KM, & Needleman HL (1992) Low-level lead
exposure, intelligence and academic achievement: A long-term follow-up
study. Pediatrics, 90: 855-861.
Berlin A & Schaller KH (1974) European standardized method for the
determination of delta-aminolevulinic acid dehydratase activity in
blood. Z Klin Chem Klin Biochem, 12: 389-390.
Bernard SR (1977) Dosimetric data and metabolic model for lead. Health
Phys, 32: 44-46.
Bernard BP & Becker CE (1988) Environmental lead exposure and the
kidney. Clin Toxicol, 26: 1-34.
Bernstein J, Evan AP, & Gardner KD (1987) Epithelial hyperplasia in
human polycystic kidney disease. Its role in pathogenesis and risk of
neoplasia. Am J Pathol, 129: 92-101.
Bert JL, van Dusen LJ, & Grace JR (1989) A generalized model for the
prediction of body lead burdens. Environ Med, 48: 117-127.
Beyer WN & Cromartie EJ (1987) A survey of Pb, Cu, Zn, Cd, Cr, As, and
Se in earthworms and soil from diverse sites. Environ Monit Assess,
8: 27-36.
Biggins PDE & Harrison RM (1979) Atmospheric chemistry of automotive
lead. Environ Sci Technol, 13: 558-565.
Billick IJ & Gray VE (1978) Lead based paint poisoning research:
review and evaluation 1971-1977. Washington, DC, US Department of
Housing and Urban Development.
Bishop L & Hill WJ (1983) A study of the relationship between blood
lead levels and occupational air lead levels. Am Stat, 37(4): 471-475.
Blair JA, Hilburn ME, Leeming RJ, McIntosh MJ, & Moore MR (1982) Lead
and tetrahydrobiopterin metabolism: possible effects on IQ. Lancet,
April 24, 964.
Body PE, Inglis GR, & Mulcahy DE (1988) Lead contamination in Port
Pirie South Australia. Adelaide, Australia, South Australia Department
of Environment and Planning (SADEP Report No. 101).
Bolger PM, Carrington CD, Capar SG, & Adams MA (1991) Reductions in
dietary lead exposure in the United States. Chem. Speciation
Bioavailab, 3(314): 31.
Bolter E, Butz T, & Arseneau JF (1975) Mobilization of heavy metals by
organic acids in the soils of a lead mining and smelting district.
In: Proceedings of the IXth Annual Conference on Trace Substances in
Environmental Health, Columbia, Missouri, 10-12 June 1975, pp 107-112.
Bondarenko GP (1968) An experimental study of the solubility of galena
in the presence of fulvic acids. Geochem Int, 5: 525-531.
Bornschein RL (1985) Influence of social factors on lead exposure and
child development. Environ Health Perspect, 62: 343-351.
Bornschein RL & Rabinowitz MB (1985) Proceedings of the Second
International Conference on Prospective Lead Studies. Environ Res,
38: 1-210.
Bornschein RL, Succop P, Dietrich KN, Clark CS, Que Hee S, & Hammond
PB (1985) The influence of social and environmental factors on dust
lead, hand lead, and blood lead levels in young children. Environ Res,
38: 108-118.
Bornschein RL, Succop PA, Krafft KM, Clark CS, Peace B, & Hammond PB
(1986) Exterior surface dust lead, interior house dust lead and
childhood lead exposure in an urban environment. In: Hemphill DD ed.
Trace substances in environmental health II. Columbia, Missouri,
University of Missouri, pp 322-332.
Bornschein RL, Succop PA, Krafft, KM, Clark CS, Peace B, & Hammond PB
(1987) Exterior surface dust lead, interior house dust lead and
childhood lead exposure in an urban environment. Trace Subst Environ
Health, 20: 322-332.
Bornschein RL, Grote J, Mitchell T, Succop PA, Dietrich KN, Krafft KM,
& Hammond PB (1989) Effects of prenatal lead exposure on infant size
at birth. In: Smith MA, Grant LD, & Sors AI ed. Lead exposure and
child development: An international assessment. Dordrecht, London,
Kluwer Academic Publishers, pp 307-319.
Boscolo P & Carmignani M (1988) Neurohumoral blood pressure regulation
in lead exposure. Environ Health Perspect, 78: 101-106.
Boutron CF, Görlach U, Candelone J-P, Bolshov MA, & Delmas RJ (1991)
Decrease in anthropogenic lead, cadmium and zinc in Greenland snows
since the late 1960s. Nature (Lond), 353: 153-156.
Braunstein GD, Dahlgren J, & Loriaux DL (1987) Hypogonadism in
chronically lead-poisoned men. Infertility, 1: 33-51.
Brown KW & Black SC (1983) Quality assurance and quality control data
validation procedures used for the Love Canal and Dallas lead soil
monitoring programs. Environ Monit Assess, 3: 113-122.
Brunekreef B (1984) The relationship between air lead and blood lead
in children: A critical review. Sci Total Environ, 38: 79-123.
Brunekreef B (1986) Childhood exposure to environmental lead. London,
United Kingdom, Monitoring and Assessment Research Centre.
Buchet J-P, Lauwerys R, Roels H, & Hubermont G (1977) Mobilization of
lead during pregnancy in rats. Int Arch Occup Environ Health,
40: 33-36.
Buchet J-P, Roels H, Bernard A, & Lauwerys R (1980) Assessment of
renal function of workers exposed to inorganic lead, cadmium, or
mercury vapour. J Occup Med, 22: 741-750.
Buchet JP, Lauwerys R, Vandervoorde A, & Pycke JM (1983) Oral daily
intake of cadmium, lead, manganese, copper, chromium, mercury,
calcium, zinc, and arsenic in Belgium: a duplicate meal study. Food
Chem Toxicol, 21: 19-24.
Bull RJ (1980) Lead and energy metabolism. In: Singhal PL & Thomas JA
ed. Lead toxicity. Baltimore, Maryland, Urban Schwarzenberg, Inc.,
pp 119-168.
Bushnell PJ & Bowman RE (1979a) Reversal learning deficits in young
monkeys exposed to lead. Pharmacol Biochem Behav, 10: 733-742.
Bushnell PJ & Bowman RE (1979b) Persistence of impaired reversal
learning in young monkeys exposed to low levels of dietary lead.
J Toxicol Environ Health, 5: 1015-1023.
Bushnell PJ & DeLuca H (1983) The effects of lactose on the absorption
and retention of dietary lead. J Nutr, 113: 365-378.
Bushnell PJ & Levin ED (1983) Effects of zinc deficiency on lead
toxicity in rats. Neurobehav Toxicol Teratology, 5: 283-288.
Bushnell G, Brockhaus A, & Baltissen R (1977a) Neurobehavioural and
system effects of long-term blood lead elevation in rats. Arch
Toxicol, 37: 247-263.
Bushnell PJ, Allan JR, & Marlar RJ (1977b) Scotopic vision deficits in
young monkeys exposed to lead. Science, 196: 333-335.
Byers RK & Lord EE (1943) Late effects of lead poisoning on mental
development. Am J Dis Child, 66: 471-494.
Calder IC, Collings MT, & Heyworth JS (1990) Evaluation of soil lead:
blood lead relationship for Port Pirie. Proceedings of the 23rd Annual
Conference of Trace Substances in Environmental Health. Environ
Geochem Health, 12(Suppl): 81-91.
Campbell BC, Beattie AD, Moore MR, Goldberg A, & Reid AG (1977) Renal
insufficiency associated with excessive lead exposure. Br Med J,
1: 482-485.
Cantor KP, Sontag JM, & Held MF (1986) Patterns of mortality among
plumbers and pipefitters. Am J Ind Med, 10: 73-89.
Capar SG & Rigsby EJ (1989) Survey of lead in canned evaporated milk.
J Assoc Off Anal Chem, 72: 378-381.
Cardenas A, Roels H, Bernard AM, Barbon R, Buchet JP, Lauwerys RR,
Rosello J, Ramis I, Mutti A, Franchini I, Fels LM, Stolte H, de Broe
ME, Nuyts GD, Taylor SA, & Price RG (1993) Markers of early renal
changes induced by industrial pollutants. II. Application to workers
exposed to lead. Br J Ind Med, 50: 28-36.
CDC (1985) Preventing lead poisoning in young children. Atlanta,
Georgia, US Department of Health and Human Services, Public Health
Service, Centers for Disease Control, pp 7-19 (Publication
No. 99-2230).
CEC (1981) Directive 77/312/CEE du Conseil du 29 mars 1977 concernant
la surveillance biologique de la population vis-à-vis du risque
saturnin. Rapport sur la mise en oeuvre de la directive. Luxembourg,
Commission des Communautés européennes, Direction Santé et Sécurité
(V/E/1/81-17/2).
Cerklewski FL & Forbes RM (1976) Influence of dietary zinc on lead
toxicity in the rat. J Nutr, 110: 689-696.
Chai S & Webb RC (1988) Effects of lead on vascular reactivity.
Environ Health Perspect, 78: 85-89.
Chakravorti S & Bhar S (1978) Chronic occupational exposure to lead:
An evaluation of environmental lead levels, lead absorption and
intoxication of storage battery workers. Bombay, Central Labour
Institute, Industrial Hygiene Division.
Chamberlain AC (1983) Effect of airborne lead on blood lead. Atmos
Environ, 17: 677-692.
Chamberlain AC (1985) Prediction of response of blood lead to airborne
and dietary lead from volunteer experiments with lead isotopes. Proc R
Soc (London), B224: 149-182.
Chamberlain AC, Heard MJ, Little P, Newton D, Wells AC, Wiffen RD
(1978) Investigations into lead from motor vehicles. Harwell, United
Kingdom Atomic Energy Authority (Report No. AERE-R9198).
Chan WH, Tang AJS, Chung DHS, & Lusis MA (1986) Concentration and
deposition of trace metals in Ontario - 1982. Water Air Soil Pollut,
29(4): 373-389.
Charney E, Kessler B, Farfel M, & Jackson D (1983) Childhood lead
poisoning: a controlled trial for the effect of dust-control measures
on blood lead levels. N Engl J Med, 309: 1089-1093.
Chau YK, Wong PTS, & Bengert GA (1979) Determination of tetraalkyl-
lead compounds in water, sediments, and fish samples. Anal Chem,
51: 186-188.
Chau YK, Wong PTS, & Kramer O (1980) Occurrence of tetraalkylead
compounds in the aquatic environment. Bull Environ Contam Toxicol,
24: 265-269.
Chettle DR, Scott MC, & Somervaille LJ (1991) Lead in bone: sampling
and quantitation using K x-rays excited by 109Cd. Environ Health
Perspect, 91: 49-55.
Chisolm JJ Jr (1962) Aminoaciduria as a manifestation of renal tubular
injury in lead intoxication and a comparison with patterns of
aminoaciduria seen in other diseases. J Pediatr, 60: 1-17.
Chisolm JJ & Brown DH (1979) Micromethod for zinc protoporphyrin in
erythrocytes: including new data on the absorptivity of zinc
protoporphyrin and new observations in neonates and sickle cell
disease. Biochem Med, 22: 214-237.
Chisolm JJ, Thomas DJ, & Hamill TG (1985) Erythrocyte porphobilinogen
synthase activity as an indicator of lead exposure to children. Clin
Chem, 31: 601-605.
Choie DD & Richter GW (1972) Cell proliferation in rat kidney induced
by lead acetate and effects of uninephrectomy on cell proliferation.
Am J Pathol, 66: 265-275.
Choie DD & Richter GW (1974) Cell proliferation in mouse kidney
induced by lead. Synthesis of deoxyribonucleic acid. Lab Invest,
30: 647-651.
Chowdhury AR, Dewan A, & Gandhi DN (1984) Toxic effect of lead on the
testes of rat. Biomed Biochem Acta, 43: 95-100.
Chowdhury AR, Chinoy NJ, & Gautam AK (1986) Effect of lead on human
semen. Adv Contracept Delivery Syst, 2: 208-211.
Christoffersson JO, Schutz A, Skerfving S, Ahlgren L, & Mattson S
(1986) Decrease of skeletal lead after end of occupational exposure.
Arch Environ Health, 41: 312-318.
Clark CS, Bornschein RL, Succop P, Que Hee SS, Hammond PB, & Peace B
(1985) Condition and type of housing as an indicator of potential
environmental lead exposure and pediatic blood lead levels. Environ
Res, 38: 46-53.
Clausing P, Brunekreef B, & van Wijnen JH (1987) A method for
estimating soil ingestion by children. Int Arch Occup Environ Health,
59: 73-82.
Cohen N, Modai D, Golik A, Weissgarten J, Peller S, Katz A, Averbukh
Z, & Shaked U (1989) Increased concanavalin A-induced suppressor cell
activity in humans with occupational lead exposure. Environ Res,
48: 1-6.
Cohn J, Cox C, & Cory-Slechta DA (1993) The effects of lead exposure
on learning in a multiple repeated acquisition and performance
schedule. Neurotoxicology, 14: 329-346.
Collins MF, Hrdina PD, & Whittle E (1992) Lead in blood and brain
regions of rats chronically exposed to low doses of the metal. Toxicol
Appl Pharmacol, 65: 314-322.
Colombo A (1985) The underdefined nature of the blood lead-air lead
relationship from biological and statistical grounds. Atmos Environ,
19: 1485-1493.
Columbano A, Ledda-Columbano GM, Lee G, Rajalakshmi S, & Sarma DSR
(1984) Inability of mitogen-induced liver hyperplasia to support
induction of enzyme-altered islands induced by liver carcinogens.
Cancer Res, 47: 5557-5559.
Cookman GR, King W, & Regan CM (1987) Chronic low-level lead exposure
impairs embryonic to adult conversion of the neural cell adhesion
molecule. J Neurochem, 49(2): 399-403.
Cookman GR, Hemmens SE, Keane GJ, King WB, & Regan CM (1988) Chronic
low level lead exposure precociously induces rat glial development
in vitro and in vivo. Neurosci Lett, 86: 33-37.
Cools A, Salle HJA, Verberk MM, & Zielhuis RL (1976) Biochemical
response of male volunteers ingesting inorganic lead for 49 days. Int
Arch Occup Environ Health, 38: 129-139.
Cooney GH, Bell A, McBride W, & Carter C (1989a) Neurobehavioural
consequences of prenatal low level exposures to lead. Neurotoxicol
Teratol, 11: 95-104.
Cooney GH, Bell A, McBride W, & Carter C (1989b) Low level exposures
to lead: the Sydney lead study. Dev Med Child Neurol, 31: 640-649.
Cooney G, Bell A, & Stavron C (1991) Low level exposures to lead and
neurobehavioural development: the Sydney study at seven years. In:
Heavy metals in the environment. Edinburgh, CEP Consultants, pp 16-19.
Cooper WC (1981) Mortality in employees of lead production facilities
and lead battery plants, 1971-1975. In: Environmental lead:
Proceedings of the Second International Symposium on Environmental
Lead Research, Cincinnati. Ohio, December 1978. New York, London, San
Francisco, pp 111-143.
Cooper WC (1988) Deaths from chronic renal disease in US battery and
lead production workers. Environ Health Perspect, 78: 61-63.
Cooper WC & Gaffey WR (1975) Mortality of lead workers. J Occup Med,
17: 100-107.
Cory-Slechta DA (1990a) Lead exposure during advanced age: alterations
in kinetics and biochemical effects. Toxicol Appl Pharmacol,
104: 67-78.
Cory-Slechta DA (1990b) Exposure duration modifies the effects of low
level lead on fixed interval performance. Neurotoxicology,
11: 427-442.
Cory-Slechta DA & Pokora MJ (1991) Behavioral manifestations of
prolonged lead exposure initiated at different stages of the life
cycle: I. Schedule-controlled responding. Neurotoxicology,
12(4): 745-760.
Cory-Slechta DA & Thompson T (1979) Behavioural toxicity of chronic
post-weaning lead exposure in the rat. Toxicol Appl Pharmacol,
47: 151-159.
Cory-Slechta DA & Widzowski DV (1991) Low level lead exposure
increases sensitivity to the stimulus properties of dopamine D1 and D2
agonists. Brain Res, 553: 65-74.
Cory-Slechta DA, Weiss B, & Cox C (1985) Performance and exposure
indices of rats exposed to low concentrations of lead. Toxicol Appl
Pharmacol, 78: 291-299.
Coscia GC, Discalzi G, & Ponzetti C (1987) Immunological aspects of
occupational lead exposure. Med Lav, 78: 360-364.
Cramer K, Goyer RA, Jagenburg OR, & Wilson MH (1974) Renal
ultrastructure, renal function and parameters of lead toxicity in
workers with different length of lead exposure. Br J Ind Med,
31: 113-127.
Cueto LM, Baer RD, & Gonzalez EM (1989) Three cases of unusual lead
poisoning. Am J Gastroenterol, 84: 1460.
Culbard EB, Thornton L, Watt JM, Wheatley M, Moorcroft S, & Thompson M
(1988) Metal contamination in British urban dusts and soils. J Environ
Qual, 17: 226-234.
Cullen MR, Kayne RD, & Robins JM (1984) Endocrine and reproductive
dysfunction in men associated with occupational inorganic lead
intoxication. Arch Environ Health, 39: 431-440.
Dabeka RW & McKenzie AD (1987) Lead, cadmium, and fluoride levels in
market milk and infant formulas in Canada. J Assoc Off Anal Chem,
70(4):754-757.
Dabeka RW, McKenzie AD, & Lacroix GMA (1987) Dietary intakes of lead,
cadmium, arsenic and fluoride by Canadian adults: a 24-hour duplicate
diet study. Food Addit Contam, 4(1): 89-102.
Dalpra L, Tibiletti MG, Nocera G, Giulotto P, Auriti L, Carnelli V, &
Simoni G (1983) SCE analysis in children exposed to lead emission from
a smelting plant. Mutat Res, 120: 249-256.
Danielson BRG, Dencker L, & Lindgren A (1983) Transplacental movement
of inorganic lead in early and late gestation in the mouse. Arch
Toxicol, 54: 97-107.
David O, Clark J, & Voller K (1972) Lead and hyperactivity. Lancet,
2: 900-903.
Davidson C, Grimm T, & Nasta M (1981) Airborne lead and other elements
derived from local fires in the Himalays. Science, 214: 1344-1346.
Davies BE (1983) A graphical estimation of the normal lead content of
some British soils. Geoderma, 29: 67-75.
Davies JM (1984) Lung cancer mortality among workers making lead
chromate and zinc chromate pigments at three English factories. Br J
Ind Med, 41: 158-169.
Davies PH & Everhart WH (1973) Effects of chemical variations in
aquatic environments: Volume 3 - Lead toxicity to rainbow trout and
testing application factor concept. Washington, DC, US Environmental
Protection Agency (EPA Report No. R373011C).
Davies BE, Elwood PC, Gallacher J, & Ginnever RC (1985) The
relationships between heavy metals in garden soils and house dusts in
an old lead mining area of North Wales, Great Britain. Environ Pollut,
B9: 255-266.
Davies DJ, Thornton I, Watt JM, Culbard EB, Harvey PG, Delves HT,
Sherlock JC, Smart GA, Thomas JFA, & Quinn MJ (1987a) Lead intake and
blood lead in 2 year olds UK urban children. Sci Total Environ,
90: 13-29.
Davies DJ, Watt JM, & Thornton I (1987b) Lead levels in Birmingham
dusts and soils. Sci Total Environ, 67: 177-185.
Davis JM & Grant LD (1992) The sensitivity of children to lead. In:
Guzelian PS, Henry CJ, & Olin S ed. Similarities and differences
between children and adults: Implications for risk assessment.
Washington, DC, ILSI Press, pp 15-27.
Davis JM & Svendsgaard DJ (1990) Nerve conduction velocity and lead: A
critical review and meta-analysis. In: Johnson BL, Anger WK, Durao A,
& Xinteras C ed. Advances in neurobehavioural toxicology: Applications
in environmental and occupational health. Chelsea, Michigan, Lewis
Publishers, pp 353-376.
Davis S, Waller P, Buschbom R, Balfour J, & White P (1990a)
Quantitative estimates of soil ingestion in normal children between
the ages of 2 and 7 years: Populations-based estimates using
aluminium, silicon, and titanium as soil tracer elements. Arch Environ
Health, 45: 112-122.
Davis JM, Otto DA, Weil DE, & Grant LD (1990b) The comparative
developmental neurotoxicity of lead in humans and animals.
Neurotoxicol Teratol, 12: 215-229.
Day JP, Hart M, & Robinson MS (1975) Lead in urban street dust. Nature
(Lond), 253: 343-345.
Dees JH, Heatfield BM, Reuben MD, & Trump BF (1980) Adenocarcinoma of
the kidney. III. Histogenesis of renal adenocarcinomas induced in rats
by N-(4-fluoro-4-biphenyl)acetamide. J Natl Cancer Inst,
64: 1537-1545.
De La Burde B & Choate MS (1972) Does asymptomatic lead exposure in
children have latent sequelae? J Pediatr, 81: 1088-1096.
De La Burde B & Choate MS (1975) Early asymptomatic lead exposure and
development at school age. J Pediatr, 87: 638-664.
De Leacy EA (1987) Lead-crystal decanters - a health risk? Med J Aust,
147: 622.
Delumyea R & Kalivretenos A (1987) Elemental carbon and lead content
of fine particles from American and French cities of comparable size
and industry, 1985. Atmos Environ, 21: 1643-1647.
Delves HT & Campbell MJ (1988) Measurements of total lead
concentrations and of lead isotope ratios in whole blood by use of
inductively coupled plasma source mass spectrometry. J Anal At
Spectrom, 3: 343-348.
Desilva PE & Donnan MB (1980) Blood lead levels in Victorian children.
Med J Aust, 2: 315-318.
Diemel JAL, Brunekreef B, Boleu JSM, Biersteker K, & Veenstra SJ
(1981) The Arnhem lead study. II. Indoor pollution, and indoor/outdoor
relationships. Environ Res, 25: 449-456.
Dieter M, Mathews HB, Jeffcoate RA, & Moseman RF (1993)
Bioavailability of lead found in mine tailings from Alaska. J Toxicol
Environ Health, 39: 79-93.
Dietrich KN, Krafft KM, Bier M, Succop P, Berger O, & Bornschein R
(1986) Early effects of fetal lead exposure: neurobehavioural findings
at six months. Int J Biosoc Res, 8: 151-168.
Dietrich KN, Krafft KM, Bornschein RL, Hammond PB, Berger OM, Succop
PA, & Bier M (1987) Low-level fetal lead exposure effect on
neurobehavioural development in early infancy. Pediatrics,
80: 721-730.
Dietrich KN, Krafft KM, Bier M, Berger O, Succop PA, & Bornschein RL
(1989) Neurobehavioural effects of foetal lead exposure: the first
year of life. In: Smith MA, Grant LD, & Sors AI ed. Lead exposure and
child development: An international assessment. Dordrecht, London,
Kluwer Academic Publishers, pp 320-331.
Dietrich KN, Succop PA, Bornschein RL, Krafft KM, Berger O, Hammond
PB, & Buncher CR (1990) Lead exposure and neurobehavioural development
in later infancy. Environ Health Perspect, 89: 13-19.
Dietrich KN, Succop PA, Berger OG, Hammond PB, & Bornschein RL (1991)
Lead exposure and cognitive development of urban, preschool children:
the Cincinnati lead cohort at age 4 years. Neurotoxicol Teratol,
13: 203-211.
Dietrich KN, Succop PA, Berger OG, & Keith RW (1992) Lead exposure and
the central auditory processing abilities and cognitive development of
urban children: the Cincinnati lead study cohort at age 5 years.
Neurotoxicol Teratol, 14: 51-56.
Dietrich K, Berger O, Succop P, Hammond P, & Bornschein R (1993a) The
developmental consequences of low to moderate prenatal and postnatal
lead exposure: Intellectual attainment in the Cincinnati Lead Study
Cohort following school entry. Neurotoxicol Teratol, 15: 37-44.
Dieterich K, Berger O, & Succop P (1993b) Lead exposure and the motor
developmental status of urban 6-year-old children in the Cincinnati
Prospective Study. Pediatrics, 91: 301-307.
Djuric D, Kerin Z, Graovac-Leposavic L, Novak L, & Kop M (1971)
Environmental contamination by lead from a mine and smelter: A
preliminary report. Arch environ Health, 23: 275-279.
Dolenc P, Staessen JA, Lauwerys RR, & Amery A (1993) Short report:
low-level lead exposure does not increase the blood pressure in the
general population. J Hypertens, 11: 589-593.
Dong A, Simsiman GV, & Chesters G (1985) Release of phosphorus and
metals from soils and sediments during dispersion. Soil Sci,
139: 97-99.
Donovan MP, Schein LG, & Thomas JA (1980) Inhibition of
androgen-receptor interaction in mouse prostate gland cytosol by
divalent metal ions. Mol Pharmacol, 17: 156-162.
Drasch GA & Ott J (1988) Lead in human bones. Investigations of an
occupationally non-exposed population in southern Bavaria (FGR). II.
Children. Sci Total Environ, 68: 61-69.
Drasch GA, Bohm J, & Baur C (1987) Lead in human bones. Investigations
of an occupationally non-exposed population in southern Bavaria (FGR).
I. Adults. Sci Total Environ, 64: 303-315.
Dresner DL, Ibrahim NG, Mascarenhas BR, & Levere RD (1982) Modulation
of bone marrow heme and protein synthesis by trace elements. Environ
Res, 28: 55-66.
Drill S, Konz J, Mahar H, & Morse M (1979) The environmental lead
problem: An assessment of lead in drinking water from a multi-media
perspective. Washington, DC, US Environmental Protection Agency
(EPA-570/9-79-003, NTIS-PB-296556).
Duggan MJ (1980) Lead in urban dust: An assessment. Water Air Soil
Pollut, 14: 309-321.
Egle P & Shelton KR (1986) Chronic lead intoxication causes a brain
specific nuclear protein to accumulate in the nuclei of cells lining
kidney tubules. J Biol Chem, 261: 2294-2298.
Elias R (1985) Lead exposures in the human environment. In: Mahaffey
KR ed. Dietary and environmental lead: human health effects.
Amsterdam, Oxford, New York, Elsevier Science Publishers, pp 79-109.
Ellen G & van Loon JW (1990) Determination of cadmium and lead in
foods by graphite furnace atomic absorption spectrometry with Zeeman
background correction: test with certified reference materials. Food
Addit Contam, 7: 265-273.
Elwood PC (1986) The sources of lead in blood: A critical review. Sci
Total Environ, 52: 1-23.
Elwood PC, Davey-Smith G, Oldham PD, & Toothill C (1988a) Two Welsh
surveys of blood lead and blood pressure. Environ Health Perspect,
78: 119-121.
Elwood PC, Yarnell JWG, Oldham PD, Catford JC, Nutbeam D, Davey-Smith
G, & Toothill C (1988b) Blood pressure and blood lead in surveys in
Wales. Am J Epidemiol, 127: 942-945.
Ernhart B & Morrow-Tlucak M (1987) The caretaking environment and
exposure of children to lead. In: Thornton I & Culbard E ed. Lead in
the home environment. Northwood, Science Reviews Ltd.
Ernhart CB, Wolf AW, Sokol RJ, Brittenham GM, & Ernhard P (1985) Fetal
lead exposure: antenatal factors. Environ Res, 38: 54-66.
Ernhart CB, Wolf WW, Kennard MJ, Erhard P, Filipovick HF, & Sokol RJ
(1986) Intrauterine exposure to low levels of lead: the status of the
neonate. Arch Environ Health, 41: 287-291.
Ernhart CB, Morrow-Tlucak M, Marler MR, & Wolf AW (1987) Low level
lead exposure in the prenatal and early preschool periods: early
preschool development. Neurotoxicol Teratol, 9: 259-270.
Ernhart CB, Morrow-Tlucak M, & Wolf AW (1988) Low level lead exposure
and intelligence in the preschool years. Sci Total Environ,
71: 453-459.
Ernhart CB, Morrow-Tlucak M, Worf AW, Super D, & Drotar D (1989a) Low
level lead exposure in the prenatal and early preschool periods:
intelligence prior to school entry. Neurotoxicol Teratol, 11: 161-170.
Ernhart CB, Brittenham G, Marler MR, & Sokol RJ (1989b) Lead related
birth defects: some methodologic issues. In: Smith MA, Grant LD, &
Sors AI ed. Lead exposure and child development: An international
assessment. Dordrecht, London, Kluwer Academic Publishers, pp 357-370.
Evans RD & Rigler FH (1985) Long distance transport of anthropogenic
lead as measured by lake sediments. Water Air Soil Pollut,
24: 141-151.
Everson J & Patterson CC (1980) "Ultra-clean" isotope dilution/mass
spectrometric analyses for lead in human blood plasma indicate that
most reported values are artificially high. Clin Chem, 26: 1603-1607.
Evis MJ, Kane KA, Moore MR, & Parratt JR (1985) The effects of chronic
low lead treatment and hypertension on the severity of cardiac
arrhythmias induced by coronary artery ligation in anaesthetized rats.
Toxicol Appl Pharmacol, 80: 234-242.
Evis MJ, Dhaliwal K, Kane KA, Moore MR, & Parratt JR (1987) The
effects of chronic lead treatment and hypertension on the severity of
cardiac arrhythmias induced by coronary artery occlusion or by
noradrenaline in anaesthetised rats. Arch Toxicol, 59: 336-340.
Ewers U, Stiller-Winkler R, & Idel H (1982) Serum immunoglobulin,
complement C3, and salivary IgA level in lead workers. Environ Res,
29: 351-357.
Fanning D (1988) A mortality study of lead workers, 1926-1985. Arch
Environ Health, 43: 247-251.
FAO/WHO (1993) Evaluation of certain food additives and contaminants.
Forty-first report of the Joint FAO/WHO Expert Committee on Food
Additives. Geneva, World Health Organization (Technical Report Series
837).
Feigl F, Anger V, & Oesper RE (1972) Spot tests in inorganic analysis.
London, Elsevier Publishing Company, pp 285-286.
Fergusson DM, Fergusson JE, Horwood LJ, & Kinzett NG (1988a) A
longitudinal study of dentine lead levels, intelligence, school
performance and behaviour. Part I. Dentine lead levels and exposure to
environmental risk factors. J Child Psychol Psychiatr, 29: 781-792.
Fergusson DM, Fergusson JE, Horwood LJ, & Kinzett NG (1988b) A
longitudinal study of dentine levels, intelligence, school performance
and behaviour. Part II. Dentine lead and cognitive ability. J Child
Psychol Psychiatr, 29: 793-809.
Fergusson DM, Fergusson JE, Horwood LJ, & Kinzett NG (1988c) A
longitudinal study of dentine lead levels, intelligence, school
performance and behaviour. Part III. Dentine lead levels and
attention/activity. J Child Psychol Psychiatr, 29: 811-824.
Fergusson JE, Fergusson DM, Horwood LJ, & Kinzett NG (1989) A
longitudinal study of dentine lead levels, intelligence, school
performance and behaviour: The measurement of dentine lead. Sci Total
Environ, 80: 229-241.
Fernando NP, Healy MA, Aslam M, Davis SS, & Hussein A (1981) Lead
poisoning and traditional practices: The consequences for world
health. A study in Kuwait. Public Health (Lond), 95: 250-260.
Fischbein A, Alvares AP, Anderson KE, Sassa S, & Kappas A (1977) Lead
intoxication among demolition workers: the effect of lead on the
hepatic cytochrome P-450 system in humans. J Toxicol Environ Health,
3: 431-437.
Fischbein A, Rice C, Sarkozi L, Kon SH, Petrocci M, & Selikoff IJ
(1979) Exposure to lead in firing ranges. J Am Med Assoc,
241: 1141-1144.
Fischbein A, Thornton J, Blumberg WE, Bernstein J, Valciukas JA, Moses
M, Davidow B, Kaul B, Sirota M, & Selikoff IJ (1980) Health status of
cable splicers with low-level exposure to lead: results of a clinical
survey. Am J Public Health, 70: 697-700.
Flegal AR, Rosman KJR, & Stephenson MD (1987) Isotope systematics of
contaminant leads in Monterey Bay. Environ Sci Technol, 21: 1075-1079.
Flora S-S & Tandon SK (1987) Effect of combined exposure to lead and
ethanol on some biochemical indices in the rat. Biochem Pharmacol,
36: 537-541.
Florence TM, Lilley SG, & Stauber JL (1988) Skin absorption of lead.
Lancet, 2: 157-158.
Fouassin A & Fondu M (1980) Evaluation of the daily intake of lead and
cadmium from food in Belgium. Arch Belg Méd Soc Hyg Méd Trav Méd Lég,
38: 453-467.
Foster WG (1992) Reproductive toxicity of chronic lead-exposure in the
female cynomolgus monkeys. J Reprod Toxicol, 16: 123-131.
Fouts PJ & Page JH (1942) The effect of chronic lead poisoning on
arterial blood pressure in dogs. Am Heart J, 24: 329-331.
Fowler BA, Kimmel CA, Woods JS, McConnell EE, & Grant LD (1980)
Chronic low-level lead toxicity in the rat: III. An integrated
assessment of long-term toxicity with special reference to the kidney.
Toxicol Appl Pharmacol, 56: 59-77.
Fox DA & Sillman AJ (1979) Heavy metals affect rod, but not cone
pholoreceptors. Science, 206: 78-80.
Fox DA, Lewkowski JP, & Cooper GP (1977) Acute and chronic effects of
neonatal lead exposure on developmental of the visual worked response
in rats. Toxicol Appl Pharmacol, 40: 449-461.
Freeman GB, Johnson JD, Killinger JM, Liao SC, Feder PI, Davis AO,
Ruby MV, Chaney RL, Lovre SC, & Bergstrom PD (1992) Relative
bioavailability of lead from mining waste soils in rats. Fundam Appl
Toxicol, 19: 388-398.
Friberg L (1988) Quality assurance. In: Clarkson TW, Friberg L,
Nordstrom GF, & Sager PR ed. Biological monitoring of toxic metals.
New York, Plenum Press, pp 103-126.
Friberg L & Vahter M (1983) Assessment of exposure to lead and cadmium
through biological monitoring: Results of a UNEP/WHO global study.
Environ Res, 30: 95-128.
Friedman JA & Weinberger HL (1990) Six children with lead poisoning. J
Dis Child, 144: 1039-1044.
Fullmer CS & Rosen JF (1990) Effect of dietary calcium and lead status
on intestinal calcium absorption. Environ Res, 51: 91-99.
Fullmer CS (1990) Intestinal lead and calcium absorption: Effect of
1,25-dihydroxy-cholecalciferol and lead status. Proc Soc Exp Biol Med,
194: 258-264.
Fulton M, Thomson G, Hunter R, Raab G, Laxen D, & Hepburn W (1987)
Influence of blood lead on the ability and attainment of children in
Edinburgh. Lancet, 30 May: 1221-1226.
Galal-Gorchev H (1991a) Dietary intake of pesticide residues, cadmium,
mercury and lead. Food Addit Contam, 8(6): 793-806.
Galal-Gorchev H (1991b) Global overview of dietary lead exposure.
Special issue incorporating the Proceedings of the Symposium on the
Bioavailability and Dietary Exposure of Lead, Chapel Hill, North
Carolina, September 1990. Chem Speciation Bioavailab, 3(3/4): 5-11.
Garber BT & Wei E (1974) Influence of dietary factors on the
gastrointestinal absorption of lead. Toxicol Appl Pharmacol,
27: 685-691.
Garcia-Miragaya J (1984) Levels, chemical fractionation, and
solubility of lead in roadside soils of Caracas, Venezuela. Soil Sci,
138: 147-152.
Gartrell MJ, Craun JC, Podrebarac DS, & Gunderson EL (1985a)
Pesticides, selected elements, and other chemicals in infant and
toddler total diet samples, October 1979 -September 1980. J Assoc Off
Anal Chem, 68: 1184-1187.
Gartrell MJ, Craun JC, Podrebarac DS, & Gunderson EL (1985b)
Pesticides, selected elements, and other chemicals in infant and
toddler total diet samples, October 1979 -September 1980. J Assoc Off
Anal Chem, 81: 1163-1183.
Gartside PS (1988) The relationship of blood lead levels and blood
pressure in NHANES II: additional calculations. Environ Health
Perspect, 78: 31-34.
Gartside PS & Buncher CR (1982) Relationship of air lead and blood
lead for workers at an automobile battery factory. Int Arch Occup
Environ Health, 50: 1-10.
GEMS (1985) Global Environmental Monitoring System: Assessment of
human exposure to lead: Comparison between Belgium, Malta, Mexico and
Sweden. Stockholm, Karolinska Institute.
Gennart JP, Bernard A, & Lauwerys R (1992) Assessment of thyroid,
testes, kidney and autonomic nervous system function in lead-exposed
workers. Int Arch Occup Environ Health, 64: 49-57.
Gerhardsson L, Lundstrom N-G, Nordbery G, & Wall S (1986) Mortality
and lead exposure: a retrospective cohort study of Swedish smelter
workers. Br J Ind Med, 43: 707-712.
Getz LL, Haney AW, Larimore RW, McNurney JW, Leland HV, Price PW,
Rolfe GL, Wortman RL, Hudson JL, Solomon RL, & Reinbold KA (1977)
Transport and distribution in a watershed ecosystem. In: Boggess WR
ed. Lead in the environment. Springfield, Virginia, National Technical
Information Service, pp 105-133 (National Science Foundation report
No. NSF/RA-770214).
Gibson JL (1904) A plea for painted railings and painted walls of
rooms as the source of lead poisoning amongst: Queensland children.
Aust Med Gaz, 23: 149.
Gilbert SG & Rice DC (1987) Low-level lifetime lead exposure produces
behavioral toxicity (spatial discrimination reversal) in adult
monkeys. Toxicol Appl Pharmacol, 91: 484-490.
Gmerek DE, McCofferty MR, O'Neill KJ, Melamed BR, & O'Neill JJ (1981)
Effect of inorganic lead on rat brain mitochondrial respiration and
energy production. J Neurochem, 36: 1109-1113.
Goldstein GW (1990) Lead poisoning and brain cell function. Environ
Health Perspect, 89: 91-94.
Gould JH, Capar SG, Jacobs RM, & Plunkett R (1988) A quick color test
to detect lead release from glazed ceramic and enameled metalware.
Anal Lett, 21: 2145-2154.
Goyer RA (1985) Renal changes associated with lead exposure. In:
Mahaffey KR ed. Dietary and environmental lead: Human health effects.
Amsterdam, Oxford, New York, Elsevier Science Publishers, pp 315-335.
Goyer RA (1992) Nephrotoxicity and carcinogenicity of lead - An update
on exposure and effects of lead. Fundam Appl Toxicol, 18: 1-16.
Goyer RA (1993) Lead toxicity: current concerns. Environ Health
Perspect, 100: 177-187.
Goyer RA & Rhyne BC (1973) Pathological effects of lead. Int Rev Exp
Pathol, 12: 1-77.
Goyer RA & Wilson MH (1975) Lead induced inclusion bodies: results of
ethylenediamine-tetra-acetic acid treatment. Lab Invest, 32: 149-156.
Goyer RA, Leonard DL, Moore JF, Rhyne B, & Krigman MR (1970) Lead
dosage and the role of the intranuclear inclusion body. Arch Environ
Health, 20: 705-711.
Goyer RA, Falk HL, Hogan M, Feldman DD, & Richter W (1981) Renal
tumors in rats given trisodium nitrilotriacetic acid in drinking water
for 2 years. J Natl Cancer Inst, 66: 869-880.
Goyer RA, Weinberg CR, Victery WM, & Miller CR (1989) Lead induced
nephrotoxicity: kidney calcium as an indicator of tubular injury. In:
Bach PH & Lock EA ed. Nephrotoxicity. New York, London, Plenum Press,
pp 11-20.
Grandjean P (1979) Occupational lead poisoning in Denmark: screening
with the haematofluorometer. Br J Ind Med, 36: 52-58.
Grandjean P & Olsen B (1984) Lead. In: Vercruysse A ed. Techniques and
instrumentation in analytical chemistry. Volume 4: Evaluation of
analytical methods in biological systems. Part B: Hazardous metals in
human toxicology. Amsterdam, Oxford, New York, Elsevier Science
Publishers, pp 153-169.
Grandjean P, Wulf HC, & Niebuhr E (1983) Sister chromatid exchange in
response to variations in occupational lead exposure. Environ Res,
32: 199-204.
Grandjean P, Hansen ON, & Lyngbye K (1984) Analysis of lead in
circumpulpal dentin of deciduous teeth. Ann Clin Lab Sci, 14: 270-275.
Grandjean P, Hansen ON, & Lyngbye K (1986) Lead concentration in
deciduous teeth; variation related to tooth type and analytical
technique. J Toxicol Environ Health, 19: 437-445.
Grandjean P, Jensen BM, Sando SH, Jorgensen PJ, & Antonsen S (1989a)
Delayed blood regeneration in lead exposure: an effect on reserve
capacity. Am J Public Health, 79: 1385-1388.
Grandjean P, Hollnagel H, Hedegaard L, Christensen JM, & Larsen S
(1989b) Blood lead-blood pressure relations: alcohol intake and
haemoglobin as confounders. Am J Epidemiol, 129: 732-739.
Grandjean P, Lyngbye T, & Hansen ON (1991) Lessons from a Danish study
on neuropsychological impairment related to lead exposure. Environ
Health Perspect, 94: 111-115.
Granick JL, Sassa S, Granick S, Levere RD, & Kappas A (1973) Studies
in lead poisoning. II. Correlation between the ratio of activated to
inactivated aminolevulinic acid dehydratase of whole blood and the
blood lead level. Biochem Med, 8: 149-159.
Grant LD & Davis JM (1989) Effects of low-level lead exposure on
paediatric neurobehavioural development: Current findings and future
directions. In: Smith MA, Grand LD, & Sors AI ed. Lead exposure and
child development: An international Assessment. Dordrecht, London,
Kluwer Academic Publishers, pp 49-115.
Grant LD, Kimmel CA, West GL, Martinez-Vargas CM, & Howard JL (1980)
Chronic low-level lead toxicity in the rat. II. Effects on postnatal
physical and behavioral development. Toxicol Appl Pharmacol, 56:
42-58.
Graziano JH & Blum C (1991) Lead exposure from lead crystal. Lancet,
337: 141-142.
Graziano JH, Popovac D, Factor-Litvak P, Shrout P, Kline J, Murphy MJ,
Zhao Y, Mehmeti A, Ahmedi X, Rajovic B, Zvicer Z, Nenezic U, Lolacono
NJ, & Steun Z (1990) Determinants of elevated blood lead during
pregnancy in a population surrounding a lead smelter in Kosova,
Yugoslavia. Environ Health Perspect, 89: 95-100.
Graziano JH, Slavkovic V, Factor-Litvak P, Popovac D, Ahmedi X, &
Mehmeti A (1991) Depressed serum erythropoietin in pregnant women with
elevated blood lead. Arch Environ Health, 46: 347-350.
Greene T, Ernhart CB, & Boyd TA (1992) Contributions of risk factors
to elevated blood and dentine levels in preschool children. Sci Total
Environ, 115: 239-260.
Griffith JQ & Landaver MA (1944) The effect of chronic lead poisoning
on arterial blood pressure in rats. Am Heart J, 28: 295-297.
Gulson B & Wilson D (1994) History of lead exposure in children
revealed from isotopic analyses of teeth. Arch Environ Health,
49X: 279-283.
Gupta AK, Sehgal SK, Mohan M, & Anand NK (1990) Cosmetic plumbism.
Indian Paediatr, 27: 760-761.
Gustafson A, Hedmer P, Schutz A, & Skerfving S (1989) Occupational
lead exposure and pituitary function. Int Arch Occup Environ Health,
61: 277-281.
Haeger-Aronsen B (1960) Studies on urinary excretion of
delta-aminolaevulic acid and other haem precursors in lead workers and
lead-intoxicated rabbits. Scand J Clin Lab Invest, 12(47): 1.
Haenninen H, Mantere P, Hernberg S, Seppalainen AM, & Kock B (1979)
Subjective symptoms in low-level exposure to lead. Neurotoxicology,
1: 333-347.
Hakim RB, Stewart WF, Canner JK, & Tielsch JM (1991) Occupational lead
exposure and strabismus in offspring: A case control study. Am J
Epidemiol, 133: 351-356.
Hammond PB, O'Flaherty EJ, & Gartside PS (1981) The impact of air-lead
on blood-lead in man: A critique of the recent literature. Food Cosmet
Toxicol, 19: 631-638.
Hammond PB, Bornschein RL, & Succop P (1985) Dose-effect and
dose-response relationships of blood lead to erythrocytic
protoporphyrin in young children. Environ Res, 38: 187-196.
Hansen ON, Trillingsgaard A, Beese I, Lyngbye T, & Grandjean P (1989)
A neuropsychological study of children with elevated dentine lead
level: assessment of the effect of lead in different socio-economic
groups. Neurotoxicol Teratol, 11: 205-213.
Harlan WR (1988) The relationship of blood lead levels to blood
pressure in the US population. Environ Health Perspect, 78: 9-13.
Harlan WR, Landis JR, Schmouder RL, Goldstein NG, & Harlan LC (1985)
Blood lead and blood pressure: relationship in the adolescent and
adult US population. J Am Med Assoc, 253: 530-534.
Harvey PG, Hamlin MW, Kumar R, & Delves HT (1984) Blood lead,
behaviour and intelligence test performance in preschool children. Sci
Total Environ, 40: 45-60.
Harvey PG, Hamlin MW, Kumar R, Morgan J, Spurgeon A, & Delves HT
(1988) Relationships between blood lead, behaviour, psychometric and
neuropsychological test performance in young children. Br J Dev
Psychol, 6: 145-156.
Hatzakis A, Kokkevi A, Maravelias C, Katsouyanni K, Salaminios F,
Kalandidi A, Koutselinis A, Stefanis C, & Trichopoulos D (1989)
Psychometric intelligence deficits in lead-exposed children. In: Smith
MA, Grand LD, & Sors AI ed. Lead exposure and child development: An
international assessment. Dordrecht, London, Kluwer Academic
Publishers, pp 211-223.
Hawk BA, Schroeder SR, Robinson G, Otto D, Mushak P, Kleinbaum D, &
Dawson G (1986) Relation of lead and social factors to IQ of low-SES
children: a partial replication. Am J. Ment Defic, 91: 178-183.
Heard MJ & Chamberlain AC (1982) Effects of minerals and food on
uptake of lead from the gastrointestinal tract in humans. Hum Toxicol,
1: 411-416.
Hem JD & Durum WH (1973) Solubility and occurrence of lead in surface
water, J An Water Works Assoc, 65: 562-568.
Henning SJ & Cooper LC (1988) Intestinal accumulation of lead salts
and milk lead by suckling rats (42645). Proc Soc Exp Biol Med,
187: 110-116.
Hernberg S (1973) Prevention of occupational poisoning from inorganic
lead. Work Environ Health, 10: 53-61.
Hernberg S & Nikkanen J (1970) Enzyme inhibition by lead under normal
urban conditions. Lancet, 1(7637): 63-64.
Hilderbrand DC, Der R, Griffin WT, & Fahim MS (1973) Effect of lead
acetate on reproduction. Am J Obstet Gynecol, 115: 1058-1065.
Hogstedt C, Hane M, Agrell A, & Bodin L (1983) Neuropsychological test
results and symptoms among workers with well-defined long term
exposure to lead. Br J Ind Med, 49: 99-105.
Holness DL & Nethercott JR (1988) Acute lead intoxication in a group
of demolition workers. Appl Ind Hyg, 3: 338-341.
Holtzman D, Shen Hgu J, & Montell P (1978) In vitro effects of
inorganic lead on isolated rat brain mitochondrial respiration.
Neurochem Res, 3: 195-206.
Holtzman D, Shen Hgu J, & Desautel M (1981) Absence of effects of lead
feedings and growth retardation on mitochondrial and microsomal
cytochrome in the developing brain. Toxicol Appl Pharmacol, 58: 48-56.
Holtzman D, Devries C, Nguyan H, Olson J, & Bensch K (1984) Maturation
of resistance to lead encephalopathy: cellular and subcellular
mechanisms. Neurotoxicology, 5: 97-124.
Hopper JL, Balderas A, & Mathews JO (1982) Analysis of variation in
blood lead levels in Melbourne families. Med J Aust, 2: 573-576.
Huang J, He F, Yigun W, & Zhang S (1988a) Observations on renal
function in workers exposed to lead. Sci Total Environ, 71: 535-537.
Huang XP, Feng ZY, Zhai WL, & Xu JH (1988b) Chromosomal aberrations
and sister chromatid exchanges in workers exposed to lead. Biomed
Environ Sci, 1: 382-387.
Huber F & Schmidt U (1976) Methylation of organolead and lead (II)
compounds to C3 4Pb by microorganisms. Nature (Lond),
259: 157-158.
Hutton M, Wadge A, & Milligan PJ (1988) Environmental levels of
cadmium and lead in the vicinity of a major refuse incinerator. Atmos
Environ, 22: 411-416.
IAEA (1987) Co-ordinated research programme on human daily dietary
intakes of nutritionally important trace elements as measured by
nuclear and other techniques. IAEA Newl, 2: 6-15.
IARC (1980) Lead and lead compounds. In: Some metals and metallic
compounds. Lyon, International Agency for Research on Cancer, pp
325-416 (IARC Monographs on the Evaluation of the Carcinogenic Risk of
Chemicals to Humans, Volume 23).
IARC (1987a) Genetic and related effects: An updating of selected IARC
Monographs from Volumes 1 to 42. Lyon, International Agency for
Research on Cancer (IARC Monographs on the Evaluation of the
Carcinogenic Risk of Chemicals to Humans, Supplement 6).
IARC (1987b) Lead and lead compounds. In: Overall evaluations of
carcinogenicity: an updating of IARC Monographs, Volumes 1 to 42.
Lyon, International Agency for Research on Cancer, pp 230-232 (IARC
Monographs on the Evaluation of the Carcinogenic Risk of Chemicals to
Humans, Supplement 7).
Ibels LS & Pollock CA (1986) Toxicology management review. Lead
intoxication. Med Toxicol, 1: 387-410.
Ichiba M & Tomokuni K (1990) Studies on erythrocyte pyrimidine
5'-nucleotidase test and its evaluation in workers occupationally
exposed to lead. Int Arch Occup Environ Health, 62: 305-310.
ILZSG (International Lead and Zinc Study Group) (1992) Lead and zinc
statistics. Int Lead Zinc Study Group Bull, 32: 3.
IPCS (1977) Environmental health criteria 3: Lead. Geneva, World
Health Organization, 160 pp.
IPCS (1989) Environmental health criteria 85: Lead - Environmental
effects. Geneva, World Health Organization, 106 pp.
IPCS (1994) Environmental health criteria 155: Biomarkers and risk
assessment: Concepts and principles. Geneva, World Health
Organization, 82 pp.
IRPC (International Radiological Protection Commission), Task Group on
Lung Dynamics (1966) Deposition and retention models for internal
dosimetry of the human respiratory trace. Health Phys, 12: 173-207.
IRPTC (1987) IRPTC legal file 1986. Geneva, International Register of
Potentially Toxic Chemicals, United Nations Environment Programme,
vol 1.
Irwig LM, Harrison WO, Rocks P, Webster I, & Andrew M (1978) Lead and
morbidity: a dose response relationship. Lancet, 2(8079): 4-7.
Jaakkola J & Anttila A (1992) [Occupational exposures to chemical
agents in Finland.] Helsinki, Institute of Occupational Health and
Finnish Work Environmental Fund (in Finnish).
James AC (1978) Lung deposition of submicron aerosols calculated as a
function of age and breathing rate. In: National Radiological
Protection Board Annual Report. Harwell, UK, National Radiological
Protection Board, pp 71-75.
Jaremin B (1983) Blast lymphocyte transformation (LTT), rosette
(E-RFC) and leukocyte migration inhibition (MIF) tests in persons
exposed to the action of lead during work. Report II. Bull Inst Marit
Trop Med (Gdynia), 34: 187-197.
Jensen AA (1984) Metabolism and toxicokinetics. In: Grandjean P ed.
Biological effects of organolead compounds. Boca Raton, Florida, CRC
Press, chapter 8, pp 97-115.
Johnson NE & Tenuta K (1979) Diets and lead blood levels of children
who practice pica. Environ Res, 18: 369-376.
Jorhem L & Slorach S (1988) Lead, chromium, tin, iron, and cadmium in
foods in welded cans. Food Addit Contam, 4: 309-316.
Jorhem L, Mattsson P, & Slorach S (1988) Lead in table wines on the
Swedish market. Food Addit Contam, 5: 645-649.
Kaminsky P, Leone J, & Duc M (1988) Incidence du saturnisme hydrique
dans un service de médecine interne en région de sols acides. Presse
Méd, 17: 419-422.
Kampe W (1983) Lead and cadmium in food - is there an actual danger?
Forum Staedte-Hyg, 34: 236-241.
Kang HK, Infante PF, & Carra JS (1980) Occupational lead exposure and
cancer (letter). Science, 207: 935-936.
Katagiri Y, Toriumi H, & Kawai M (1983) Lead exposure among 3-year-old
children and their mothers living in a pottery-producing area. Int
Arch Occup Environ Health, 52: 223-229.
Kaye WE, Novotny TE, & Tucker M (1987) New ceramics-related industry
implicated in elevated blood lead levels in children. Arch Environ
Health, 42: 161-164.
Kazantzis G (1989) Lead: ancient metal - modern menace. In: Smith MA,
Grant LD, & Sors AI ed. Lead exposure and child development: An
international assessment. Dordrecht, London, Kluwer Academic Press,
pp 119-128.
Kehoe RA (1961) The metabolism of lead in man in health and disease:
The normal metabolism of lead (The Harben Lectures, 1960). J R Inst
Public Health Hyg, 24: 81-97.
Kehoe RA (1987) Studies of lead administration and elimination in
adult volunteers under natural and experimentally induced conditions
over extended periods of time. Food Chem Toxicol, 25: 425-493.
Keller CA & Doherty RA (1980a) Distribution and excretion of lead in
young and adult female mice. Environ Res, 21: 217-228.
Keller CA & Doherty RA (1980b) Bone lead mobilization in lactating
mice and lead transfer to suckling offspring. Toxicol Appl Pharmacol,
55: 220-228.
Kerin Z (1973) Lead in new-fallen snow near a lead smelter. Arch
Environ Health, 26: 256-260.
Kholil-Manesh F, Gonich HC, Cohen AH, Alinovi R, Berganaschi E, Mutti
A, & Rosen VJ (1992) Experimental model of lead nephrotoxicity. I.
Continuous high-dose lead administration. Kidney Int, 41: 1192-1203.
Kimber I, Stonard MD, Gidlow DA, & Niewola Z (1986) Influence of
chronic low-level exposure to lead on plasma immunoglobulin
concentration and cellular immune function in man. Int Arch Occup
Environ Health, 57: 117-125.
King E & Conchie D (1979) Industrial lead absorption. Ann Occup Hyg,
22: 213-239.
Kirkby H & Gyntelberg F (1985) Blood pressure and other cardiovascular
risk factors of long term exposure to lead. Scand J Work Environ
Health, 11: 15-19.
Kocak R, Anarat A, Altintas G, & Evilyaoglu N (1989) Lead poisoning
from contaminated flour in a family of 11 members. Hum Toxicol,
8: 385-386.
Kolbye AC, Mahaffey R, Fiorino A, Corneliussen PC, & Jelinek CF (1974)
Food exposures to lead. Environ Health Perspect, 7(Exp Issue): 65-75.
Koller LD (1985) Immunological effects of lead. In: Mahaffey KR ed.
Dietary and environmental lead: Human health effects. Amsterdam,
Oxford, New York, Elsevier Science Publishers, pp 339-354.
Koller LD & Roan JG (1980) Effect of lead, cadmium and methylmercury
on immunologic memory. J Environ Pathol Toxicol, 4: 47-52.
Koo WWK, Succop PA, Bornschein RL, Krug-Wispe SK, Steichen JJ, Tsang
RC, Hammond PB, & Berger OG (1991) Serum vitamin D metabolites and
bone mineralization in young children with chronic to moderate lead
exposure. Pediatrics, 87: 680-687.
Kopp SJ, Barron JT, & Tow JP (1988) Cardiovascular actions of lead and
relationship to hypertension: a review. Environ Health Perspect,
89: 91-99.
Koren G, Chang N, Gonen R, Klein J, Weiner L, Demshar H, Pizzolato S,
Radde I, & Shime J (1990) Lead-exposure among mothers and their
newborns in Toronto. Can Med Assoc J, 142: 1241-1244.
Kostial K & Kello D (1979) Bioavailability of lead in rats fed "human"
diets. Bull Environ Contam Toxicol, 21: 312-314.
Kostial K, Kello D, & Jugo S (1978) Influence of age on metal
metabolism and toxicity. Environ Health Perspect, 25: 81-86.
Kovar IZ, Strehlow CD, Richmond J, & Thompson MG (1984) Perinatal lead
and cadmium burden in a British urban population. Arch Dis Child,
59: 36-39.
Krueger JA & Duguay KM (1989) Comparative analysis of lead in Maine
urban soils. Bull Environ Contam Toxicol, 42: 574-581.
Kusell M, Lake L, Anderson M, & Gerschenson LE (1978) Cellular and
molecular toxicology of lead. II. Effect of lead on d-aminolevulinc
acid synthelase of cultured cells. J Toxicol Environ Health,
4: 515-525.
Kutbi II, Ahmed M, & Saber A (1989) Measurement of blood-lead levels
in school children of Jeddah Saudi Arabia and assessment of sub-toxic
levels of lead on some sensitive hematological parameters. J Environ
Sci Health, A24: 943-955.
Lacey RF, Moore MR, & Richards WN (1985) Lead in water, infant diet
and blood: The Glasgow duplicate diet study. Sci Total Environ,
41: 235-257.
Lai CS (1972) Lead poisoning as an occupational hazard in Chinese
opera actors: a case report. Singap Med J, 13: 115-117.
Landis JR & Flegal KM (1988) A generalized Mantel-Haenszel analysis of
the regression of blood pressure on blood lead using NHANES II data.
Environ Health Perspect, 78: 35-41.
Landrigan PJ, Gehlbach SH, Rosenblum BF, Shoults JM, Cancelaria RM,
Barthel WF, Liddle JA, Smrek AL, Staehling NW, & Sanders JF (1975)
Epidemic lead absorption near an ore smelter; The role of particulate
lead. N Engl J Med, 292: 123-129.
Lansdown RG, Shepherd J, Clayton BE, Delves HT, Graham PJ, & Turner WC
(1974) Blood-lead levels, behaviour, and intelligence: a population
study. Lancet, 1(7857): 538-541.
Lansdown R, Yule W, Urbanowicz MA, & Hunter J (1986) The relationship
between blood-lead concentrations, intelligence, attainment and
behaviour in a school population: the second London study. Int Arch
Occup Environ Health, 57: 225-235.
Laurs AJ (1976) Review of European test methods for measuring lead and
cadmium release. In: Proceedings of the International Conference on
Ceramic Foodware Safety, Geneva, 1974. New York, Lead Industries
Association Inc., pp 27-36.
Lauwerys R, Buchet JP, Roels HA, & Materne DD (1974) Relationship
between urinary delta-aminolevulinic acid excretion and the inhibition
or red cell delta-aminolevulinate dehydratase by lead. Clin Toxicol,
7: 383-388.
Lavelle JM, Poppenga RH, Thacker BJ, Geisy JP, Weis C, Othoudt R, &
Vandervoort C (1991) Bioavailability of lead in mining wastes: An oral
intubation study in young swine. Chem Speciation Bioavailab,
3: 105-111.
Laxen DPH, Raab GM, & Fulton M (1987) Children's blood lead and
exposure to lead in household dust and water - a basis for an
environmental standard for lead in dust. Sci Total Environ,
66: 235-244.
Lee RE & Goranson S (1972) National air surveillance cascade impact or
network. I. Size distribution measurements of suspended particulate
matter in air. Environ Sci Technol, 6: 1019-1024.
Leeming RJ & Blair JA (1980) The effect of pathological and normal
physiological processes on biopterin levels in man. Clin Chim Acta,
108: 102-111.
Lepow ML, Bruckman L, Rubino RA, Markowitz S, Gillette M, & Kapish J
(1974) Role of airborne lead in increased body burden of lead in
Hartford children. Environ Health Perspect, 7: 99-102.
Lerda D (1992) Study of sperm characteristics in persons
occupationally exposed to lead. Am J Ind Med, 22: 567-571.
Levin ED & Bowman RE (1983) The effect of pre- or postnatal lead
exposure on Hamilton search task in monkeys. Neurobehav Toxicol
Teratol, 5: 391-394.
Levin ED & Bowman RE (1986) Long-term lead effects on the Hamilton
Search Test and delayed alternation in monkeys. Neurobehav Toxicol
Teratol, 8: 219-224.
Levin ED & Bowman RE (1989) Long-term effects of chronic post-natal
lead exposure on delayed spatial alternation in monkeys. Neurotoxicol
Teratol, 10: 505-510.
Lilienthal H, Winneke G, Brockhaus A, & Molik B (1986) Pre- and
postnatal lead-exposure in monkeys: Effects on activity and learning
set formation. Neurobehav Toxicol Teratol, 8: 265-272.
Lilienthal H, Winneke G, & Ewert T (1990) Effects of lead on
neurophysiological and performance measures: animal and human data.
Environ Health Perspect, 89: 21-25.
Lilis R (1981) Long-term occupational lead exposure, chronic
nephropathy, and renal cancer: a case report. Am J Ind Med,
2: 293-297.
Lilis R, Fischbein A, Eisinger J, Blumberg WE, Diamond S, Anderson HA,
Rom W, Rice C, Sarkozi L, Kon S, & Selikoff IJ (1977) Prevalence of
lead disease among secondary lead smelter workers and biological
indicators of lead exposure. Environ Res, 14: 255-285.
Lilley SG, Florence TM, & Stauber JL (1988) The use of sweat to
monitor lead absorption through the skin. Sci Total Environ,
76: 267-278.
Lind B, Vahter M, Rahnster B, & Björs U (1988) Quality control samples
for the determination of lead and cadmium in blood, feces, air
filters, and dust. Fresenius Z Anal Chem, 332: 741-743.
Lindholm ML, Sallmen M, Anttila A, Taskinen H, & Hemminki K (1991)
Paternal occupational lead exposure and spontaneous abortion. Scand J
Work Environ Health, 17(2): 95-103.
Lin-Fu JS (1985) Historical perspective on health effects of lead. In:
Mahaffey KR ed. Dietary and environmental lead: Human health effects.
Amsterdam, Oxford, New York, Elsevier Science Publishers, pp 43-64.
Lovering TG ed. (1976) Lead in the environment. Washington, DC, US
Department of the Interior, Geological Survey (Geological Survey
Professional Paper No. 957).
Lyngbye T, Hansen OL, Trillingsgaard A, Beese I, & Grandjean P (1990a)
Learning disabilities in children: significance of low-level
lead-exposure and confounding factors. Acta Paediatr Scand,
79: 352-360.
Lyngbye T, Jorgensen PJ, & Grandjean P (1990b) Validity and
interpretation of blood lead levels: a study of Danish
school-children. Scand J Clin Lab Invest, 50: 441-449.
McGrath SP (1986) The range of metal concentrations in top soils of
England and Wales in relation to soil protection guidelines. In
Hemphill DD ed. Trace substances in environmental health, vol 20.
Columbia, Missouri, University of Missouri, pp 242-252.
McGregor AJ & Mason MJ (1990) Chronic occupational lead exposure and
testicular endocrine function. Hum Exp Toxicol, 9: 371-376.
McIntosh MJ, Meredith PA, Moore MR, & Goldberg A (1985) Neurotoxic
action of lead: effect on tetrahydrobiopterin metabolism in the rat.
Comp Biochem Physiol, 81C(1): 227-231.
McIntosh MJ, Meredith PA, Moore MR, & Goldberg A (1989) Action of lead
on neurotransmission in rats. Xenobiotica, 19(1): 101-113.
McKhann CF & Vogt EC (1933) Lead poisoning in children. J Am Med
Assoc, 101: 1131-1135.
McMichael AJ & Johnson HM (1982) Long-term mortality profile of
heavily-exposed lead smelter workers. J Occup Med, 24: 375-378.
McMichael AJ, Vimpani GV, Robertson EF, Baghurst PA, & Clark PD (1986)
The Port Pirie cohort study: maternal blood lead and pregnancy
outcome. J Epidemiol Community Health, 41: 18-25.
McMichael AJ, Baghurst PA, Wigg NR, Vimpani GV, Robertson EF, &
Roberts RJ (1988) Port Pirie cohort study: environmental exposure to
lead and children's abilities at the age of four years. N Engl J Med,
319: 468-475.
Maenhaut W, Zoller WH, Duce RA, & Hoffman GL (1979) Concentration and
size distribution of particulate trace elements in the south polar
atmosphere. J Geophys Res, 84: 2421-2431.
Mahaffey KR (1985) Factors modifying susceptibility to lead toxicity.
In: Mahaffey KR ed. Dietary and environmental lead: Human health
effects. Amsterdam, Oxford, New York, Elsevier Science Publishers,
pp 373-419.
Mahaffey KR & Annest JL (1986) Association of erythrocyte
protoporphyrin with blood lead level and iron status in the Second
National Health and Nutrition Examination Survey, 1976-1980. Environ
Res, 41: 327-338.
Mahaffey-Six KR & Goyer RA (1972) The influence of iron deficiency on
tissue content and toxicity of ingested lead in the rat. J Lab Clin
Med, 79: 128-136.
Mahaffey KR, Rosen JF, Chesney RW, Peeler JT, Smith CM, & Debuca HF
(1982) Association between age, blood lead concentration, and serum
1,25-dihydroxy-cholecalciferol levels in children. Am J Clin Nutr,
35: 1327-1331.
Maines MD (1992) Haem oxygenase-clinical applications and functions.
Boca Raton, Florida, CRC Press.
Maki-Paakkanen J, Sorsa M, & Vainio H (1981) Chromosome aberrations
and sister chromatid exchanges in lead-exposed workers. Hereditas,
94: 269-275.
Manton WI (1985) Total contribution of airborne lead to blood lead. Br
J Ind Med, 42: 168-172.
Manton WI & Cook JD (1984) High accuracy (stable isotope dilution)
measurements of lead in serum and cerebrospinal fluid. Br J Ind Med,
41: 313-319.
Manton WI & Thal ER (1986) Lead poisoning from retained missiles: an
experimental study. Ann Surg, 204: 594-599.
Mao P & Molnar JJ (1967) The fine structure and histochemistry of
lead-induced renal tumors in rats. Am J Pathol, 50: 571-603.
Maranelli G & Apostoli P (1987) Assessment of renal function in lead
poisoned workers. In: Occupational and environmental chemical hazards:
Cellular and biochemical indices for monitoring toxicity. Chichester,
Ellis Horwood Ltd, chapter 42, pp 344-348.
Marcus AH (1985a) Testing alternative nonlinear kinetic models in
compartmental analysis. In: Eisenfield J & Delisi C ed. Mathematics
and computers in biomedical applications. Amsterdam, Oxford, New York,
Elsevier Science Publishers, pp 259-267.
Marcus AH (1985b) Multicompartment kinetic models for lead: Part I.
bone kinetics and variable absorption in humans without excessive lead
exposures. Environ Res, 36: 441-458.
Marcus AH (1985c) Multicompartment kinetic models for lead: Part II.
linear kinetics and variable absorption in humans without excessive
lead exposures. Environ Res, 36: 459-472.
Marcus AH & Schwartz J (1987) Dose-response curves for erythrocyte
protoporphyrin vs blood lead: effects of iron status. Environ Res,
44: 221-227.
Marino PE, Franzblau A, Lilis R, & Landrigan PJ (1989) Acute lead
poisoning in construction workers: the failure of protective
standards. Arch Environ Health, 44: 140-145.
Marks SC & Popoff SN (1988) Bone cell biology: the regulation of
development, structure, and function in the skeleton. Am J Anat,
183: 1-44.
Marlowe M & Errrera J (1982) Low lead levels and behaviour problems in
children. Behav Disord, 7: 163-172.
Matte TD, Figueroa JP, Ostrowski S, Burr G, Jackson-Hunt L, Kennylside
RA, & Baker EL (1989a) Lead poisoning among household members exposed
to lead-acid battery repair shops in Kingston, Jamaica. Int J
Epidemiol, 18: 874-881.
Matte TD, Figueroa JP, Burr G, Flesch JP, Keenlyside RA & Baker EL
(1989b) Lead exposure among lead-acid battery workers in Jamaica. Am J
Ind Med, 16: 167-177.
Meredith PA & Moore MR (1979) The influence of lead on haem
biosynthesis and biodegradation in the rat. Biochem Soc Trans,
7: 637-639.
Meredith PA, Moore MR, Campbell BC, Thompson GG, & Goldberg A (1978)
Delta-aminolevulinic acid metabolism in normal and lead-exposed
humans. Toxicology, 9: 1-9.
Merwin BW (1976) Review of US standards on test methods for lead and
cadmium release. In: Proceedings of the International Conference on
Ceramic Foodware Safety, Geneva, 1974. New York, Lead Industries
Associate Inc., pp 36-40.
Meyer BR, Fishbein A, Rosemann K, Lerman Y, Drayer DE, & Reidenberg MM
(1984) Increased urinary enzyme excretion in workers exposed to
nephrotoxic chemicals. Am J Med, 76: 989-998.
Michaelson A & Sauerhoff MW (1974) An improved model of lead induced
brain dysfunction in the suckling rat. Toxicol Appl Pharmacol,
28: 88-96.
Mielke H, Blake B, Burroughs S, & Hassinger N (1984) Urban lead levels
in Minneapolis: the case of the Hmong children. Environ Res,
34: 64-76.
Mitchell DG, Aldous KM, & Ryan FJ (1974) Mass screening for lead
poisoning, capillary blood sampling and automated Delves cup atomic
absorption analysis. NY State J Med, 74: 1599-1603.
Mitchell-Heggs CAW, Conway M, & Cassar J (1990) Herbal medicine as a
cause of combined lead and arsenic poisoning. Hum Exp Toxicol,
9: 195-196.
Molina-Ballesteros G, Zuniga-Charles MA, Ortega AC, & Solis-Camara P
(1983) Lead concentrations in the blood of children from
pottery-making families exposed to lead salts in a Mexican village.
Bull Panam Health Organ, 17: 35-41.
Möller L & Kristensen TS (1992) Blood lead as a cardiovascular risk
factor. Am J Epidemiol, 136: 1091-1100.
Moore MR (1983) Lead exposure and lead plumbosolvency. In: Rutter M &
Jones RR ed. Lead versus health. London, John Wiley and Sons,
pp 79-106.
Moore MR & Meredith PA (1979) The carcinogenicity of lead. Arch
Toxicol, 42: 87-94.
Moore MR, Meredith PA, Campbell BC, & Watson SW (1979) The
gastrointestinal absorption of lead 203 chlorine in man. In: Hemphill
DD ed. Trace substances in environmental health - XIII. Proceedings of
the 13th Annual Conference on Trace Substances in Environmental
Health, Columbia, June 1979. Columbia, Missouri, University of
Missouri, pp 368-373.
Moore MR, Meredith PA, & Goldberg A (1980) Lead and heme biosynthesis.
In: Singhal PL & Thomas JA ed. Lead toxicity. Baltimore, Maryland,
Urban and Schwarzenberg, Inc., pp 79-118.
Moore MR, Goldberg A, & Pocock SJ (1982) Some studies of maternal and
infant lead exposure in Glasgow. Scott Med J, 27: 113-122.
Moore MR, Bushnell IWR, & Goldberg A (1989) A prospective study of the
results of changes in environmental lead exposure in children in
Glasgow. In: Smith MA, Grant LD, & Sors AI ed. Lead exposure and child
development: An international assessment. Dordrecht, London, Kluwer
Academic Publishers, pp 371-378.
Moore MR, Goldberg A, & Young-Laiwah AAC (1987) Lead effects on the
haem-biosynthetic pathway. Relationship to toxicity. Ann NY Acad Sci,
514: 191-203.
Mooty J, Ferrand CF, & Harris P (1975) Relationship of diet to lead
poisoning in children. Pediatrics, 55: 636-639.
Moreau T, Orssaud G, Juguet B, & Busquet G (1982) Plombémie et
pression artérielle: premiers résultats d'une enquête transversale de
431 sujets de sexe masculin. Rev Epidemiol Santé Publique,
30: 395-397.
Morgan A & Holmes A (1978) The fate of lead in petrol-engine exhaust
particulates inhaled by the rat. Environ Res, 15: 44-56.
Muijser H, Hoogendijk EMG, Hooisma J, & Twisk DAM (1987) Lead exposure
during demolition of a steel structure coated with lead-based paints.
II. Reversible changes in the conduction velocity of the motor nerves
in transiently exposed workers. Scand J Work Environ Health,
13: 56-61.
Munoz C, Garbe K, Lilienthal H, & Winneke G (1986) Persistence of
retention deficit in rats after neonatal lead exposure.
Neurotoxicology, 7(2): 569-580.
Muramatsu Y & Parr RM (1985) Survey of currently available reference
materials for use in connection with the determination of trace
elements in biological and environmental materials. Vienna,
International Atomic Energy Agency (Report No. IAEA/RL/128).
Murata K & Araki S (1991) Autonomic nervous system dysfunction in
workers exposed to lead, zinc, and copper in relation to peripheral
nerve conduction: a study of R-R interval variability. Am J Ind Med,
20: 663-671.
Murphy MJ, Graziano JH, Popovac D, Kline JK, Mehmeti A, Factor-Litvak
P, Ahmadi G, Shrout P, Rajovic B, Nenezic DU, & Stein ZA (1990) Past
pregnancy outcomes among women living in the vicinity of a lead
smelter in Kosovo, Yugoslavia. Am J Public Health, 80: 33-35.
Mushak P (1989) Biological monitoring of lead exposure in children:
overview of selected biokinetic and toxicological issues. In: Smith
MA, Grant LD, & Sors AI ed. Lead exposure and child development: An
international assessment. Dordrecht, London, Kluwer Academic
Publishers, pp 129-145.
Mushak P & Crocetti AF (1989) Determination of numbers of lead-exposed
American children as a function of lead source: Integrated summary of
a report to the U.S. Congress on childhood lead poisoning. Environ
Res, 50: 210-299.
Mussalo-Rauhamaa H, Salmela SS, Leppanen A, & Pyysalo H (1986)
Cigarettes as a source of some trace and heavy metals and pesticides
in man. Arch Environ Health, 41: 49-55.
NAS-NRC (1972) Airborne lead in perspective. Washington, DC, National
Academy of Sciences.
Needleman HL & Gatsonis CA (1990) Low-level lead exposure and the IQ
of children: a meta-analysis of modern studies. J Am Med Assoc,
263: 673-678.
Needleman HL, Tuncay OC, & Shapiro IM (1972) Lead levels in deciduous
teeth of urban and suburban children. Nature (Lond), 235: 111-112.
Needleman HL, Gunnoe C, Leviton A, Reed R, Peresie H, Maher C, &
Barrett P (1979) Deficits in psychologic and classroom performance of
children with elevated dentine lead levels. N Engl J Med,
300: 689-695.
Needleman HL, Schell A, Bellinger D, Leviton A, & Allred EN (1990) The
long-term effects of childhood exposure to low doses of lead: An
11-year follow-up report. N Engl J Med, 322: 83-88.
Neri LC, Hewitt D, & Orser B (1988) Blood lead and blood pressure:
analysis of cross-sectional and longitudinal data from Canada. Environ
Health Perspect, 78: 123-126.
Nerin C, Olavide S, & Cacho J (1989) Determination of lead in airborne
particulate by hydride generation. Water Air Soil Pollut, 44: 339-345.
Neubecker TA & Allen HE (1983) The measurement of complexation
capacity and conditional stability constants for ligands in natural
waters. Water Res, 17: 1-14.
NFA (Australian National Food Authority, National Health and Medical
Council) (1991) The 1990 Australian market basket survey report.
Canberra, Commonwealth of Australia, Australian Government Publishing
Service.
Ng A & Patterson CC (1982) Changes of Pb and Ba with time in
California offshore basin sediments. Geochim Cosmochim Acta,
46: 2307-2322.
Ng TP, Goh HH, Ng YL, Ong CN, Chia KS, Chia SE, & Jeyaratnam J (1991)
Male endocrine functions in workers with moderate exposure to lead. Br
J Ind Med, 48: 485-491.
Nielsen T (1984) Atmospheric occurrence of organolead compounds. In:
Grandjean P ed. Biological effects of organolead compounds. Boca
Raton, Florida, CRC Press, pp 43-62.
Nilsson U, Attewell R, Christoffersson J-O, Schütz A, Ahlgren L,
Skerfving S, & Mattsson S (1991) Kinetic of lead in bone and blood
after end of occupational exposure. Pharmacol Toxicol, 69: 477-484.
NIOSH (1977a) Method No. P&CAM 102. In: Manual of analytical methods,
vol 1, 2nd ed. Cincinnati, Ohio, National Institute for Occupational
Safety and Health.
NIOSH (1977b) Method No. P&CAM 191. In: Manual of analytical methods,
vol 1, 2nd ed. Cincinnati, Ohio, National Institute for Occupational
Safety and Health, pp 191/1-191/9.
NIOSH (1977c) Method No. P&CAM 195. In: Manual of analytical methods,
vol 1, 2nd ed. Cincinnati, Ohio, National Institute for Occupational
Safety and Health.
NIOSH (1977d) Method No. P&CAM 200. In: Manual of analytical methods,
vol 1, 2nd ed. Cincinnati, Ohio, National Institute for Occupational
Safety and Health, pp 200/1-200/8.
NIOSH (1977e) Method No. P&CAM 214. In: Manual of analytical methods,
vol 1, 2nd ed. Cincinnati, Ohio, National Institute for Occupational
Safety and Health, pp 214/1-214/6.
NIOSH (1981) Method No. P&CAM 351. In: Manual of analytical methods,
vol 7. Cincinnati, Ohio, National Institute for Occupational Safety
and Health, pp 351/1-351/11.
Nordenson I, Beckman G, Beckman L, & Nordstrom S (1978) Occupational
and environmental risks in and around a smelter in northern Sweden:
IV. Chromosomal aberrations in workers exposed to lead. Hereditas,
88: 263-267.
Nordstrom S, Beckman L, & Nordenson I (1978a) Occupational and
environmental risks in and around a smelter in northern Sweden: I.
Frequencies in birth weight. Hereditas, 88: 43-46.
Nordstrom S, Beckman L, & Nordenson I (1978b) Occupational and
environmental risks in and around a smelter in northern Sweden: III.
Frequencies of spontaneous abortion. Hereditas, 88: 51-54.
Nordstrom S, Beckman L, & Nordenson I (1979) Occupational and
environmental risks around a smelter in northern Sweden: V.
Spontaneous abortion among female employees and decreased birth weight
in their offspring. Hereditas, 90: 291-296.
Nosal RM & Wilhelm WJ (1990) Lead toxicity in the shipbreaking
industry: the Ontario experience. Can J Public Health, 81: 259-262.
Novotny T, Cook M, Hughes J, & Lee SA (1987) Lead exposure in a firing
range. Am J Public Health, 77: 1225-1226.
Nriagu J (1979) Global inventory of natural and anthropogenic
emissions of trace metals to the atmosphere. Nature (Lond),
279: 409-411.
Nriagu J (1989) A global assessment of natural sources of atmospheric
trace metals. Nature (Lond), 338: 47-49.
Nriagu J & Pacyna JM (1988) Quantitative assessment of worldwide
contamination of air, water, and soils by trace metals. Nature (Lond),
333: 134-139.
OCTEL (1991) Worldwide survey of motor gasoline quality. London,
Associated Octel Company Ltd (Octel Promotional Publication No. 4).
OECD (1993) Risk Reduction Monograph No. 1: Lead background and
national experience with reducing risk. Paris, Organisation for
Economic Co-operation and Development, 277 pp (Report No.
OCDE/GD(93)67).
O'Flaherty EJ (1991) Physiologically based lead kinetics. Trace Subst
Environ Health, 24: 44-54.
O'Flaherty EJ (1993) Physiologically based models for bone-seeking
elements. IV. Kinetics of lead disposition in humans. Toxicol Appl
Pharmacol, 118: 16-29.
O'Flaherty EJ, Hammond PB, Lerner SI, Hanenson IB, & Roda SMB (1980)
The renal handling of delta-aminolevulinic acid in the rat and in the
human. Toxicol Appl Pharmacol, 55: 423-432.
O'Flaherty EJ, Hammond PB, & Lerner SI (1982) Dependence of apparent
blood lead half-life on the length of previous lead exposure in
humans. Fundam Appl Toxicol, 2: 49-54.
Olejnik D, Walkowska A, Wisnilwska J, & Ziembinski R (1985)
[Evaluation of the daily dose of mercury, lead and cadmium in the diet
of certain population groups.] Roczn Pzh, XXXVI: 9-21 (in Polish, with
English abstract).
Olson KW & Skogerboe RK (1975) Identification of soil lead compounds
from automotive sources. Environ Sci Technol, 9: 227-230.
Ong CN & Lee WR (1980) High affinity of lead for fetal hemoglobin. Br
J Ind Med, 37: 292-298.
Ong CN, Phoon WO, Law HY, Tye CY, & Lim HH (1985) Concentration of
lead in maternal blood, cord blood, and breast milk. Arch Dis Child,
60: 576-759.
Ong CN, Endo G, & Chia KS (1987) Evaluation of renal function in
workers with low blood lead levels. In: Occupational and environmental
chemical hazards: Cellular and biochemical indices for monitoring
toxicity. Chichester, Ellis Horwood Ltd, chapter 39, pp 327-333.
Orfila MP (1814) Traité des poisons. Paris, Crochard, vol 2.
Orssaud G, Claude JR, Moreau T, Lellouch J, Juguet B, & Festy B (1985)
Blood lead concentration and blood pressure. Br Med J, 290: 244.
Padilla F, Shapiro AP, & Jensen WN (1969) Effect of chronic lead
intoxication on blood pressure in the rat. Am J Med Sci, 258: 359-365.
Paglia DE, Valentine WN, & Dahlgren JG (1975) Effects of low-level
lead exposure on pyrimidine 5'-nucleotidase and other erythrocyte
enzymes: possible role of pyrimidine 5'-nucleotidase in the
pathogenesis of lead-induced anaemia. J Clin Invest, 56: 1164-1169.
Pagliuca A, Mufti GJ, Baldwin D, Lestas AN, Wallis RM, & Bellingham AJ
(1990) Lead poisoning: clinical, biochemical, and haematological
aspects of a recent outbreak. J Clin Pathol, 43: 277-281.
Parfitt AM (1990) Pharmacologic manipulation of bone remodelling and
calcium homeostasis: Calcium metabolism. Prog Basic Clin Pharmacol,
4: 1-27.
Parkinson DK, Ryan C, Brommet EJ, & Connell MM (1986) A psychiatric
epidemiologic study of occupational lead exposure. Am J Epidemiol,
123: 261-269.
Parkinson DK, Ryan C, Bromet J, & Connell MM (1987) A psychiatric
epidemiological study of occupationally exposure. Am J Epidemiol,
123: 261-269.
Patterson C (1965) Contaminated and natural lead environments of man.
Arch Environ Health, 11: 344-360.
Patterson CC (1983) Criticism of "flow of metals into the global
atmosphere". Geochim Cosmochim Acta, 47: 1163-1168.
Patterson CC & Settle DM (1976) The reduction of orders of magnitude
errors in lead analyses of biological materials and natural waters by
evaluating and controlling the extent and source of industrial lead
contamination introduced during sample collecting, handling, and
analyses. In: LaFleur PD ed. Accuracy in trace analysis: Sampling,
sample handling and analysis. Volume 1. Proceedings of the 7th
Materials Research Symposium, Gaithersburg, Maryland, October, 1974.
Washington, DC, US Department of Commerce, National Bureau of
Standards, pp 321-352 (NBS Special Publication No. 422).
Pekkarinen M (1970) Methodology in the collecting of food consumption
data. World Rev Nutr Diet, 12: 145-170.
Pentschew A (1965) Morphology and morphogenesis of lead
encephalopathy. Acta Neuropathol, 5: 133-160.
Pentschew A & Garro F (1966) Lead encephalo-myelopathy of the suckling
rat and its implications on the porphyrinopathic nervous diseases with
special reference to the permeability/disorders of the nervous
system's capillaries. Acta Neuropathol, 6: 266-278.
Perino J & Ernhart CB (1974) The relation of subclinical lead level to
cognitive and sensorimotor impairment in black preschoolers. J Learn
Disabil, 7: 616-620.
Pickston L, Brewerton HV, Drysdale JM, Hughes JT, Smith JM, Love JL,
Sutcliffe ER, & Davidson F (1985) The New Zealand diet: a survey of
elements, pesticides, colours and preservatives. N Z J Technol,
1: 81-89.
Piomelli S, Seaman C, Zullow D, Curran A, & Davidow B (1982) Threshold
for lead damage to heme synthesis in urban children. Proc Natl Acad
Sci (USA), 79: 3335-3339.
Piper W & Tephly T (1974) Differential inhibition of erythrocyte and
hepatic uroporphyrinogen. I. Synthelase activity by lead. Life Sci,
14: 873-876.
Pirkle JL, Schwartz J, Lamdis JR, & Harlan WR (1985) The relationship
between blood lead levels and blood pressure and its cardiovascular
risk implications. Am J Epidemiol, 121: 246-258.
Pocock SJ, Shaper AG, Walker M, Wale CJ, Clayton B, Delves T, Lacey
RF, Packham RF, & Powell P (1983) Effects of tap water lead, water
hardness, alcohol, and cigarettes on blood lead concentrations. J
Epidemiol Community Health, 37: 1-7.
Pocock SJ, Shaper AG, Ashby D, Delves T, & Whitehead TP (1984) Blood
lead concentration, blood pressure, and renal function. Br Med J,
289: 872-874.
Pocock S, Ashby D, & Smith MA (1987) Lead exposure and children's
intellectual performance. Int J Epidemiol, 16: 57-67.
Pocock SJ, Shaper AG, Ashby D, Delves HT, & Clayton BE (1988) The
relationship between blood lead, blood pressure, stroke and heart
attacks in middle-aged British men. Environ Health Perspect,
78: 23-30.
Pocock SJ, Ashby D, & Smith MA (1989) Lead exposure and children's
intellectual performance. The Institute of Child Health/Southampton
Study. In: Smith M, Grant LD, & Sors A ed. Lead exposure and child
development: An international assessment. Dordrecht, London, Kluwer
Academic Press, pp 149-165.
Pollock CA & Ibels LS (1986) Lead intoxication in paint removal
workers on the Sydney Harbour Bridge. Med J Aust, 145: 635-639.
Ponka A, Salminen E, & Ahonen S (1991) Lead in the ambient air and
blood specimens of children in Helsinki. Sci Total Environ, 138(1-3):
301-308.
Pontifax AH & Garg AK (1985) Lead poisoning from an Asian Indian folk
remedy. Can Med Assoc J, 133: 1227-1228.
Poole C, Smythe LE, & Alpers M (1980) Blood lead levels of Papua New
Guinea children living in a remote area. Sci Total Environ, 15: 17-24.
Pounds JG (1984) Effect of lead intoxication on calcium homeostasis
and calcium-mediated cell function: a review. Neurotoxicology,
5: 295-332.
Pounds JG, Marlar RJ, & Allen JR (1978) Metabolism of lead-210 in
juvenile and adult rhesus monkeys Macaca mulatta. Bull Environ Contam
Toxicol, 19: 684-691.
Pounds JG, Long GJ, & Rosen JF (1991) Cellular and molecular toxicity
of lead in bone. Environ Health Perspect, 91: 17-32.
Purchase NG & Fergusson JE (1986) Lead in teeth: the influence of the
tooth type and the sample within a tooth on lead levels. Sci Total
Environ, 52: 239-250.
Qazi QH, Madahar C, & Huceoglu AM (1980) Temporary increase in
chromosome breakage in an infant prenatally exposed to lead. Hum
Genet, 53: 201-203.
Qian ZM & Morgan EH (1990) Effect of lead on the transport of
transferrin-free and transferrin-bound iron into rabbit reticulocytes.
Biochem Pharmacol, 40: 1049-1054.
Quarterman J & Morrison JN (1975) The effects of dietary calcium and
phosphorus on the retention and excretion of lead in rats. Br J Nutr,
34: 351-362.
Que Hee SS & Boyle JR (1988) Simultaneous multielemental analysis of
some environmental and biological samples by inductively coupled
plasma atomic emission spectrometry. Anal Chem, 60: 1033-1042.
Que Hee SS, MacDonald TJ, & Bornschein RL (1985a) Blood lead by
furnace-Zeeman atomic absorption spectrophotometry. Microchem J,
32: 55-63.
Que Hee SS, Peace B, Clark CS, Boyle JR, Bornschein RL, & Hammond PB
(1985b) Evolution of efficient methods to sample lead sources, such as
house dust and hand dust in the homes of children. Environ Res,
38: 77-95.
Raab GM, Laxen DPH, & Fulton M (1987) Lead from dust and water as
exposure sources for children. Environ Geochem Health, 9: 80.
Rabinowitz MB (1991) Toxicokinetics of bone lead. Environ Health
Perspect, 91: 33-37.
Rabinowitz M & Needleman HL (1982) Temporal trends in the lead
concentrations of umbilical cord blood. Science, 216: 1429-1431.
Rabinowitz MB, Wetherill G, & Kopple J (1975) Absorption, storage and
excretion of lead by normal humans. In: Proceedings of the Ninth
Annual Conference on Trace Substances in Environmental Health.
Columbia, Missouri, University of Missouri, pp 361-368.
Rabinowitz MB, Wetherill GM, & Kopple JD (1976) Kinetics analysis of
lead metabolism in healthy humans. J Clin Invest, 58: 260-270.
Rabinowitz MB, Wetherill GW, & Kopple JD (1977a) Magnitude of lead
intake from respiration by normal man. J Lab Clin Med, 90: 238-248.
Rabinowitz MB, Wetherill GW, & Kopple JD (1977b) Effect of food intake
and fasting on gastrointestinal lead absorption in humans. Am J Clin
Nutr, 33: 1784-1788.
Rabinowitz MB, Kopple JD, & Wetherill GW (1980) Effect of food intake
and fasting on gastrointestinal lead absorption in humans. Am J Clin
Nutr, 33: 1784-1788.
Rabinowitz MB, Leviton A, & Needleman HL (1986) Occurrence of elevated
protoporphyrin levels in relation to lead burden in infants. Environ
Res, 39: 253-257.
Rabinowitz M, Bellinger D, Leviton A, Needleman H, & Schoenbaum S
(1987) Pregnancy hypertension, blood pressure during labor, and blood
lead levels. Hypertension, 10: 447-451.
Rabinowitz MB, Leviton A, & Bellinger D (1989) Blood lead-tooth lead
relationship among Boston children. Bull Environ Contam Toxicol,
43: 485-492.
Rabinowitz MB, Wang J-D, & Soong WT (1991) Dentine lead and child
intelligence in Taiwan. Arch Environ Health, 46: 351-360.
Ragan HA (1977) Effect of iron deficiency on the absorption of lead
and cadmium in the rat. J Lab Clin Med, 90: 700-706.
Reaves GA & Berrow ML (1984) Total lead concentrations in Scottish
soils. Geoderma, 32: 1-8.
Reisinger K, Stoeppler M, & Nurnberg HW (1981) Evidence for the
absence of biological methylation of lead in the environment. Nature
(Lond), 281: 228-230.
Reuter JH & Perdue EM (1977) Importance of heavy metal-organic matter
interactions in natural waters. Geochim Cosmochim Acta, 41: 325-334.
Rhagavan SRV, Dwight Culver B, & Gonick HC (1981) Erythrocyte
lead-binding protein after occupational exposure. II. Influence of
lead inhibition of membrane Na+, K±-adenosinetriphosphatase. J Toxicol
Environ Health, 7: 561-568.
Rice DC (1984a) Behavioural deficit (delayed matching to sample) in
monkeys exposed from birth to low level lead. Toxicol Appl Pharmacol,
75: 337-345.
Rice DC (1984b) Effect of lead on schedule-controlled behavior in the
monkey. In: Seiden LS & Balster RL ed. Behavioral pharmacology: The
current status. New York, Alan R. Liss, pp 473-486.
Rice DC (1985) Chronic low-lead exposure from both produces deficits
in discrimination reversal monkey's. Toxicol Appl Pharmacol,
77: 201-210.
Rice DC & Karpinski KF (1988) Lifetime dow level lead exposure
produces deficits in delayed alternation in adult monkeys.
Neurotoxicol Teratol, 10: 207-214.
Roberts J, Mahaffey KR, & Annest JL (1985) Blood lead levels in
general populations. In: Mahaffey KR ed. Dietary and environmental
lead: Human health effects. Amsterdam, Oxford, New York, Elsevier
Science Publishers, pp 355-372.
Robins JM, Cullen MR, Connors BB, & Kayne RD (1983) Depressed thyroid
indexes associated with occupational exposure to inorganic lead. Arch
Intern Med, 143: 220-224.
Robinson TR (1974) Delta-aminolevulinic acid and lead in urine of lead
antiknock workers. Arch Environ Health, 28: 133-138.
Rodamilans M, Osaba MJM, To-Figueras J, Fillat FR, Marques JM, Perez
P, & Corbella J (1988) Lead toxicity on endocrine testicular function
in an occupationally exposed population. Hum Toxicol, 7: 125-128.
Rodney BS & Lee MG (1985) Environmental lead hazard in a high-risk
Jamaican population. West Indian Med J, 34: 176-179.
Roels HA & Lauwerys R (1987) Evaluation of dose-effect and
dose-response relationships for lead exposure in different Belgian
population groups (foetus, child, adult men and women). Trace Elem
Med, 4: 80-87.
Roels HA, Buchet J-P, Lauwerys R, Hubermont G, Bruaux P, Claeys-
Thoreau F, Lafontaine A, & van Overschelde J (1976) Impact of air
pollution by lead on the haem biosynthetic pathway in school-age
children. Arch Environ Health, 31: 310-316.
Roels HA, Buchet JP, Lauwerys RR, Bruaux P, Claeys-Thoreau F,
Lafontaine A, & Verduyn G (1980) Exposure to lead by the oral and the
pulmonary routes of children living in the vicinity of a primary lead
smelter. Environ Res, 22: 81-94.
Rosen JF & Chesney RW (1983) Circulating calcitriol concentration in
health and disease. J Pediatr, 103: 1-7.
Rosen JF, Chesney RW, Hamstra AJ, Deluca HF, & Mahaffey KR (1980)
Reduction in 1,25-dihydroxyvitamin D in children with increased lead
absorption. N Engl J Med, 302: 1128-1131.
Rosen JF, Markowitz ME, Bijur PE, Jenks ST, Wielopolski L, Kalef-Ezra
JA, & Slatkin DN (1991) Sequential measurements of bone lead content
by L x-ray fluorescence in CaNa2EDTA-treated lead-toxic children.
Environ Health Perspect, 91: 57-62.
Rosner D & Markowitz G (1985) A "gift of God": The public health
controversy over leaded gasoline during the 1920s. Am J Public Health,
75(4): 344.
Rossouw J, Offermeier J, & van Rooyen J (1987) Apparent central
neurotransmitter receptor changes induced by low-level lead exposure
during different developmental phases in the rat. Toxicol Appl
Pharmacol, 91: 132-139.
Routh DK, Mushak P, & Boone L (1979) A new syndrome of elevated blood
lead and microcephaly. J Pediatr Psychol, 4: 67-76.
Rundle SA & Duggan MJ (1986) Lead pollution from the external
redecoration of old buildings. Sci Total Environ, 57: 181-190.
Ryu JE, Ziegler EE, Nelson SE, & Fomon SJ (1983) Dietary intake of
lead and blood lead concentration in early infancy. Am J Dis Child,
137: 886-891.
Ryu JE, Ziegler EE, Nelson SE, & Fomon SJ (1985) Dietary and
environmental exposure to lead and blood lead during early infancy.
In: Mahaffey KR ed. Dietary and environmental lead: Human health
effects. Amsterdam, Oxford, New York, Elsevier Science Publishers,
pp 187-209.
Saenger P, Markowitz MR, & Rosen JF (1984) Depressed excretion of
6ß-hydroxycortisol in lead-toxic children. J Clin Endocrinol Metab,
58: 363-367.
SAHC (1993) Proceedings of the International Meeting on
Non-Occupational Exposure to Lead, Melbourne, Australia, 5-9 October
1992. Adelaide, South Australian Health Commission, 56 pp.
Sarto F, Stella M, & Acqua A (1978) [Cytogenic studies in 20 workers
occupationally exposed to lead.] Med Lav, 69: 172-180 (in Italian).
Satzger RD, Clow CS, & Bonnin E (1982) Determination of background
levels of lead and cadmium in raw agricultural crops by using
differential pulse anodic stripping voltammetry. J Assoc Off Anal
Chem, 65: 987-991.
Sauk J & Somerman MJ (1991) Physiology of bone: mineral compartment
proteins as candidates for environmental perturbation by lead. Environ
Health Perspect, 91: 9-16.
Schlegel H & Kufner G (1979) Long-term observation of biochemical
effects of lead in human experiments. J Clin Chem Clin Biochem,
17: 225-233.
Schmid E, Bauchinger M, Pietruk S, & Hall G (1972) [Cytogenic action
of lead in human peripheral lymphocytes in vitro and in vivo.]
Mutat Res, 16: 401-406 (in German).
Schmidt U & Huber F (1976) Methylation of organolead and lead(II)
compounds (CH3)4 Pb by microorganisms. Nature (Lond),
259: 157-158.
Schmitt MDC, Trippler DL, & Wachtler JN (1988) Soil lead
concentrations in residential Minnesota as measured by ICP AES. Water
Air Soil Pollut, 39: 157-168.
Schneitzer L, Osborn HH, Bierman A, Mezey A, & Kaul B (1990) Lead
poisoning in adults from renovation of an older home. Ann Emerg Med,
19: 415-420.
Schroeder SR, Hawk B, Otto DA, Mushak P, & Hicks RE (1985) Separating
the effects of lead and social factors on IQ. Environ Res,
38: 144-154.
Schutz AS, Skerfving S, Ranstam J, & Christoffersson JO (1987)
Kinetics of lead in blood after the end of occupational exposure.
Scand J Work Environ Health, 13: 221-231.
Schutz A, Ranstam J, Skerfving S, & Tejning S (1989) Blood-lead levels
in school children in relation to industrial emission and automobile
exhausts. Ambio, 13: 115-117.
Schwartz J (1988) The relationship between blood lead and blood
pressure in the NHANES II survey. Environ Health Perspect, 78: 15-22.
Schwartz J (1993) Low-level lead exposure and children's IQ: a
meta-analysis and search for a threshold. Environ Res, 65: 42-55.
Schwartz J & Otto D (1987) Blood lead, hearing thresholds, and
neurobehavioral development in children and youth. Arch Environ
Health, 42: 153-160.
Schwartz J & Otto D (1991) Lead and minor hearing impairment. Arch
Environ Health, 46: 300-305.
Schwartz J, Angle C, & Pitcher H (1986) Relationship between childhood
blood lead levels and stature. Pediatrics, 77: 281-288.
Schwartz J, Landrigan PL, & Baker EL (1990) Lead-induced anemia:
dose-response relationships and evidence for a threshold. Am J Public
Health, 80: 165-168.
Scott DR, Hemphill DC, & Holboke LE (1976) Atomic absorption and
optical emission analysis of NASN atmospheric particulate samples for
lead. Environ Sci Technol, 9: 877-880.
Selander S & Cramer K (1970) Interrelationships between lead in blood,
lead in urine and ALA in urine during lead work. Br J Ind Med,
27: 28-39.
Selevan SG, Landrigan PJ, Stern FB, & Jones JH (1985) Mortality of
lead smelter workers. Am J Epidemiol, 122: 673-683.
Selevan SG, Landrigan PJ, Stern FB, & Jones JH (1988) Brief report:
lead and hypertension in a mortality study of lead smelter workers.
Environ Health Perspect, 78: 65-66.
Seppäläinen AM & Hernberg S (1980) Subclinical lead neuropathy. Am J
Ind Med, 1: 413-420.
Seppäläinen AM, Hernberg S, Vesanto R, & Kock B (1983) Early
neurotoxic effects of occupational lead exposure: a prospective study.
Neurotoxicology, 4(2): 181-192.
Settle DM & Patterson CC (1980) Lead in albacore: guide to lead
pollution in Americans. Science, 207: 1167-1176.
Shaltout A, Yaish SA, & Fernando N (1981) Lead encephalopathy in
infants in Kuwait. Ann Trop Paediatr (London), 1: 209-215.
Shaper AG, Pocock SJ, Walker M, Cohen NM, Wale CJ, & Thompson AG
(1981) British regional heart study: cardiovascular risk factors in
middle-aged men in 24 towns. Br Med J, 283: 179-186.
Sharma RR, Chandy MJ, & Lad SD (1990) Transient hydrocephalis and
acute lead encephalopathy in neonates and infants. Report of two
cases. Br J Neurosurg, 4: 141-146.
Sherlock J, Smart G, Forbes GI, Moore MR, Patterson WJ, Richards WN, &
Wilson TS (1982) Assessment of lead intakes and dose-response for a
population in Ayr exposed to a plumbsolvent water supply. Hum Toxicol,
1: 115-122.
Sherlock JC, Pickford CJ, & White GF (1986) Lead in alcoholic
beverages. Food Addit Contam, 3: 347-354.
Siegel M, Forsyth B, Siegel L, & Cullen MR (1989) The effect of lead
on thyroid function in children. Environ Res, 49: 190-196.
Silbergeld EK (1991) Lead in bone: implications for toxicology during
pregnancy and lactation. Environ Health Perspect, 91: 63-70.
Silbergeld EK, Schwartz J, & Mahaffey KR (1988) Lead and osteoporosis:
mobilization of lead from bone in postmenopausal women. Environ Res,
47: 79-94.
Silva PA, Hughes P, Williams S, & Faed JM (1988) Blood lead,
intelligence, reading attainment, and behaviour in eleven year old
children in Dunedin, New Zealand. J Child Psychol Psychiatry Appl
Discipl, 29: 43-52.
Sinclair DF & Dohnt BR (1984) Sampling and analysis techniques used in
a blood lead survey of 1341 children in Port Pirie, South Australia.
Clin Chem, 30(10): 1616-1619.
Skerfving S (1988) Biological monitoring of exposure to inorganic
lead. In: Clarkson TW, Friberg L, Nordberg GF, & Sager PR ed.
Biological monitoring of toxic metals. New York, Plenum Press,
pp 169-197 (Rochester Series on Environmental Toxicity).
Skerfving S, Schutz A, & Ranstam J (1986) Decreasing lead exposure in
Swedish children. Sci Total Environ, 58: 225-229.
Skogerboe RK, Hartley AM, Vogel RS, & Koirtyohann SR (1977) Monitoring
for lead in the environment. In: Boggess WR ed. Lead in the
environment. Washington, DC, National Science Foundation, pp 33-70
(NSF Report No. NSF/RA-770214; PB-278278).
Slorach SA, Gustafsson I-B, Jorhem L, & Mattsson P (1983) Intake of
lead, cadmium and certain other metals via a typical Swedish weekly
diet. Var Föda, 35(Suppl 1): 3-16.
Smith MA (1989) The effects of low-level lead exposure on children.
In: Smith MA, Grant LD, & Sors AI ed. Lead exposure and child
development: An international assessment. Dorcrecht, London, Kluwer
Academic Publishers, pp 3-47.
Smith F & Hursh J (1977) Bone storage and release. In: Field J ed.
Handbook of physiology: Reaction to environmental agents. Bethesda,
Maryland, American Physiological Society, section 9, pp 469-482.
Smith M, Delves T, Lansdown R, Clayton B, & Graham P (1983) The
effects of lead exposure on urban children: the Institute of Child
Health/Southampton Study. Dev Med Child Neurol, 47(Suppl): 1-54.
Snee RD (1981) Evaluation of studies of the relationship between blood
lead and air lead. Int Arch Occup Environ Health, 48: 219-242.
Sobel AE, Yuska H, Peters DD, & Kramer B (1940) The biochemical
behaviour of lead. I. Influence of calcium phosphorus and vitamin D on
lead in the blood and bone. J Biol Chem, 132: 239-265.
Sokol RZ (1987) Hormonal effects of lead acetate in the male rat:
Mechanism of action. Biol Reprod, 37: 1135-1138.
Sokol RZ, Madding CE, & Swerdloff RS (1985) Lead toxicity and the
hypothalamic-pituitary-testicular axis. Biol Reprod, 33: 722-728.
Spivey GH, Baloh RW, Brown CP, Browdy BL, Campion DS, Valentine JL,
Morgan DE, & Culver BD (1980) Subclinical effects of chronic increased
lead absorption - a prospective study: III. Neurologic findings at
follow-up examination. J Occup Med, 22: 607-612.
Staessen J, Yeoman WB, Fletcher AE, Markowe HLJ, Marmot MG, Rose G,
Semmence A, Shipley MJ, & Bulpitt CJ (1990) Blood lead concentration,
renal function, and blood pressure in London civil servants. Br J Ind
Med, 47: 442-447.
Staessen JA, Lauwerys RA, Buchet J-P, Bulpitt CJ, Rondia D,
Vanrenterghem Y, Amery A, & The Cadmibel Study Group (1992) Impairment
of renal function with increasing blood lead concentrations in the
general population. N Engl J Med, 327: 151-156.
Staessen JA, Christopher JB, Fagard R, Lauwerys RR, Roels H, Thijs L,
& Amery A (1994) Hypertension caused by low-level lead exposure: myth
or fact? J Cardiovasc Risk, 1: 87-97.
Steelink C (1977) Humates and other natural organic substances in the
aquatic environment. J Chem Ed, 54: 599-603.
Steenhout A (1987) How clean is clean? An exotoxicological method for
getting guidelines (air, dust, deposition, water) for lead, keeping
tooth and blood lead levels in the normal range. In: Proceedings of
the International Conference on Heavy Metals in the Environment.
Edinburgh, CEP Consultants, pp 283-285.
Steenhout A & Pourtois M (1981) Lead accumulation in teeth as a
function of age with different exposures. Br J Ind Med, 38: 297-303.
Stiles KM & Bellinger DC (1993) Neuropsychological correlates of
low-level lead exposure in school-age children: A prospective study.
Neurotoxicol Teratol, 15(1): 27-35.
Stollery BT, Banks HA, Broadbent DE, & Lee WR (1989) Cognitive
functioning in lead workers. Br J Ind Med, 46: 698-707.
Stollery BT, Broadbent DE, Banks HA, & Lee WR (1991) Short term
prospective study of cognitive functioning in lead workers. Br J Ind
Med, 48: 739-749.
Stone CH & Soares JH (1976) The effect of dietary selenium level of
lead toxicity in the Japanese quail. Poult Sci, 55: 341-349.
Stone C, Fox MRS, & Hogye KS (1981) Bioavailability of lead in oysters
fed to young Japanese quail. Environ Res, 26: 409-421.
Stuik EJ (1974) Biological response of male and female volunteers to
inorganic lead. Int Arch Arbeitsmed, 33: 83-97.
Succop PA, O'Flaherty EJ, Bornschein RL, Roba SB, & Greenland RD
(1989) Recent observations concerning the relationship of blood lead
to erythrocytic protoporphyrin. In: Smith MA, Grant LD, & Sors AE ed.
Lead exposure and child development: An international Assessment.
Dordrecht, London, Kluwer Academic Publishers, pp 477-484.
Talbot RW & Andren AW (1983) Relationships between Pb and 210Pb in
aerosol and precipitation at a semiremote site in northern Wisconsin.
J Geophys Res, 88: 6752-6760.
Taskinen H, Nordman H, Engstrom K, & Hernberg S (1981) [Blood lead
levels in Finnish children.] Duodecim, 97(10): 663-670 (in Finnish).
Teruya K, Sakurai H, Omae K, Higashi T, Muto T, & Kaneko Y (1991)
Effect of lead on cardiac parasympathetic function. Int Arch Occup
Environ Health, 62: 549-553.
Thornton I & Webb JS (1975) Trace elements in soils and surface waters
contaminated plast metaliferous mining part of England. In:
Proceedings of the IXth Annual Conference on Trace Substances in
Environmental Health, Columbia, Missouri, 10-12 June, pp 77-88.
Thornton I, Culbard E, Moorcroft S, Watt J, Wheatley M, & Thompson M
(1985) Metals in urban dusts and soils. Environ Technol Lett,
6: 137-144.
Todd AC, McNeill FE, Palethorpe JE, Peach DE, Chettle DR, Tobin MJ,
Strosko SJ, & Rosen JC (1992) In vivo x-ray fluorescence of lead in
bone using K x-ray excitation with 109Cd sources; radiation dosimetry
studies. Environ Res, 57: 117-132.
Triebig G, Weltle D, & Valentin H (1984) Investigations on
neurotoxicity of chemical substances at the workplace. V.
Determination of the motor and sensory nerve conduction velocity in
persons occupationally exposed to lead. Int Arch Occup Environ Health,
53: 189-204.
Trotter RT (1990) The cultural parameters of lead poisoning: A medical
anthropologist's view of intervention in environmental lead exposure.
Environ Health Perspect, 89: 79-84.
Tuppurainen M, Wagar G, Kurppa K, Sakari W, Wambugu A, Froseth B, Alho
J, & Nykyri E (1988) Thyroid function as assessed by routine
laboratory tests of workers with long-term lead exposure. Scand J Work
Environ Health, 14: 175-180.
UK (1982) The Glasgow duplicate diet study (1979/1980): A joint survey
for the Department of the Environment and the Ministry of Agriculture,
Fisheries and Food. London, Her Majesty's Stationery Office (Pollution
Report No. 11).
US Department of the Treasury (1991) Report of analyses of wines and
related products to determine lead content. Washington, DC, US
Department of the Treasury, Bureau of Alcohol, Tobacco and Firearms.
US EPA (1986a) Air quality criteria for lead. Washington, DC,
Environmental Protection Agency (EPA-600/8-83/028aF-dF).
US EPA (1986b) Test methods for evaluating solid waste SW-846 -
Physical/chemical methods. Method No. 7420 and 7421. Washington, DC,
US Environmental Protection Agency, Office of Solid Waste and
Emergency Response.
US EPA (1989) Evaluation of the potential carcinogenicity of lead and
lead compounds. Washington, DC, US Environmental Protection Agency,
Office of Health and Environmental Assessment (EPA/600/8-89/045A).
US EPA (1990) Supplement to the 1986 EPA air quality criteria for
lead. Volume 1, Addendum: Relationship of blood pressure to lead
exposure. Washington, DC, US Environmental Protection Agency, Office
of Health and Environmental Assessment (EPA/600/8-89/0 49F).
US EPA (1991) National air quality and emission trends report.
Research Triangle Park, North Carolina, US Environmental Protection
Agency (EPA-450/4-91-023).
US NCI (1979) Bioassay of lead dimethylditho-carbamate for possible
carcinogenicity. Washington, DC, National Cancer Institute,
Carcinogenesis Testing Program, Division of Cancer Cause and
Prevention (Technical Report Series No. 151 - NTIS PB-85-134663).
Vahter M & Friberg L (1988) Quality control in integrated human
exposure monitoring of lead and cadmium. Fresenius Z Anal Chem,
332: 726-731.
Vahter M & Slorach S (1990) GEMS - Global Environment Monitoring
System. Exposure monitoring of lead and cadmium: an international
pilot study within the WHO/UNEP Human Exposure Assessment Location
(HEAL) Programme. Nairobi, United Nations Environment Programme,
82 pp.
Vahter M, Berglund M, Friberg L, Jorhem L, Lind B, Slorach S, &
Åkesson A (1990) Dietary intake of lead and cadmium in Sweden. Var
Föda, 42(2): 1-16.
Vahter M, Berglund M, Slorach S, Friberg L, Saric M, Xingquan Z, &
Fujita M (1991a) Methods for integrated exposure monitoring of lead
and cadmium. Environ Res, 56: 78-79.
Vahter M, Berglund M, Lind B, Jorhem L, Slorach S, & Friberg L (1991b)
Personal monitoring of lead and cadmium exposure - a Swedish study
with special reference to methodological aspects. Scand J Work Environ
Health, 17: 65-74.
Van Esch GJ & Kroes R (1969) The induction of renal tumors by feeding
basic lead acetate to mice and hamsters. Br J Cancer, 23: 765-771.
Varo P & Kovistoinen P (1983) Mineral element composition of Finnish
foods. XII. General discussion and nutritional evaluation. Acta Agric
Scand, 22: 165.
Vatsala S & Ramakrishna T (1985) "Tinning" of brass utensils:
Possibility of lead poisoning. Indian J Environ Health, 27: 140-141.
Verrula GR & Noah PK (1990) Clinical manifestations of childhood lead
poisoning. J Trop Med Hyg, 93: 170-177.
Verschoor M, Wibowo A, & Herber R (1987) Influence of occupational low
level lead exposure on renal parameters. Am J Ind Med, 12: 341-351.
Victery W (1988) Evidence for effects of chronic lead exposure on
blood pressure in experimental animals: An overview. Environ Health
Perspect, 78: 71-76.
Victery W, Vander AJ, Markel H, Katzman L, Shulak JM, & Germain C
(1982a) Lead exposure, begun in-utero, decreases renin and angiotensin
II in adult rats. Proc Soc Exp Biol Med, 170: 63-67.
Victery W, Vander AJ, Shulak JM, Schoeps P, & Julius S (1982b) Lead,
hypertension and the renin-angiotensin system in rats. J Lab Clin Med,
99: 354-362.
Volkening J, Baumann H, & Heumann KG (1988) Atmospheric distribution
of particulate lead over the Atlantic Ocean from Europe to Antarctica.
Atmos Environ, 22: 1169-1174.
Wadge A & Hutton M (1987) The leachability and chemical speciation of
selected trace elements in fly ash from coal combustion and refuse
incineration. Environ Pollut, 48: 85-99.
Wai CM, Knowles CR, & Keely JF (1979) Lead caps on wine bottles and
their potential problems. Bull Environ Contam Toxicol, 21: 4-6.
Wallace DM, Kalman DA, & Bird TD (1985) Hazardous lead release from
glazed dinnerware: A cautionary note. Sci Total Environ, 44: 289-292.
Wang Y-L (1984) Industrial lead poisoning in China over the past 33
years. Ecotoxicol Environ Saf, 8: 526-530.
Wang J-D, Jang C-S, Hwang Y-H, & Chen Z-S (1992) Lead contamination
around a kindergarten near a battery recycling plant. Bull Environ
Contam Toxicol, 49: 23-30.
Ward NI, Watson R, & Bryce-Smith D (1987) Placental element levels in
relation to fetal development for obstetrically normal births. A study
of 37 elements. Evidence for the effects of cadmium, lead, and zinc on
fetal growth and for smoking as a source of cadmium. Int J Biosoc Res,
9: 63-81.
Wasserman G, Graziano JH, Factor-Litvak P, Popovac D, Morina N,
Musabegovic A, Vrenezi N, Capuni-Paracka S, Lekic V, Preteni-Redjepi
E, Hadzialijevic S, Slavkovich V, Kline J, Shrout P, & Stein Z (1992)
Independent effects of lead exposure and iron deficiency anemia on
developmental outcome at age 2 years. J Pediatr, 121(5): 695-703.
Watson WS, Hume R, & Moore MR (1980) Oral absorption of lead and iron.
Lancet, 2(8188): 236-237.
Watson WS, Morrison J, Bethel MIF, Baldwin NM, Lyon DTB, Dobson H,
Moore MR, & Hume R (1986) Food iron and lead absorption in humans. Am
J Clin Nutr, 44: 248-256.
Weast RC ed. (1985) Handbook of chemistry and physics, 66th ed. Boca
Raton, Florida, CRC Press.
Weeden RP, Mallik DK, & Batuman V (1979) Detection and treatment of
occupational lead nephropathy. Arch Intern Med, 139: 53-57.
White JM & Harvey DR (1972) Defective synthesis of alpha and ß globin
chains in lead poisoning. Nature (Lond), 236: 71-73.
WHO (1980) Recommended health-based limits in occupational exposure to
heavy metals. Report of a WHO Study Group. Geneva, World Health
Organization (Technical Report Series No. 647).
WHO (1984) Guidelines for drinking-water quality. Volume 1:
Recommendations. Geneva, World Health Organization, pp 55-56.
WHO (1985) Guidelines for the study of dietary intake of chemical
contaminants. Geneva, World Health Organization (WHO Offset
Publication No. 87).
WHO (1987) Air quality guidelines for Europe. Copenhagen, World Health
Organization, Regional Office for Europe, pp 200-209 (European Series,
No. 23).
WHO (1993) Guidelines for drinking-water quality. 2nd ed. Volume 1:
Recommendations. Geneva, World Health Organization, 188 pp.
Wiebe JP, Barr KJ, & Buckingham KD (1982) Lead administration during
pregnancy and lactation affects steriodogenesis and hormone receptors
in testes of offspring. J Toxicol Environ Health, 10: 653-666.
Wiebe JP, Salhanick AI, & Myers KI (1983) On the mechanism of action
of lead on the testes: in vitro suppression of FSH receptors, cyclic
AMP and steroidogenesis. Life Sci, 32: 1997-2005.
Wiebe RA, Anderson BS, Lehman CW, & Fu DJ (1991) Lead poisoning in
Hawaii: 1990. Hawaiian Med J, 50: 89-95.
Wielopolski L, Ellis KJ, Vaswani AN, Cohn SH, Greenberg A, Puschett
JB, Parkinson DK, Fetterolf DE, & Langrigan PJ (1986) In vivo bone
lead measurements: A rapid monitoring method for cumulative lead
exposure. Am J Ind Med, 9: 221-226.
Wigg NR, Vimpani GV, McMichael AJ, Baghurst PA, Robertson EF, &
Roberts RJ (1988) Port Pirie cohort study: childhood blood lead and
neuropsychological development at age two years. J Epidemiol Community
Health, 42: 213-219.
Wildt K, Eliasson R, & Berlin M (1983) Effects of occupational
exposure to lead on sperm and semen. In: Clarkson TW, Nordberg GF, &
Sager PR ed. Reproductive and developmental toxicity of metals. New
York, London, Plenum Press, pp 279-300.
Wilhelm M, Lombeck I, & Hafner D (1989) Hair lead levels in young
children from the FRG. J Trace Elem Electrolytes Health Dis,
3: 165-170.
Williams MK, King E, & Walford J (1969) An investigation of lead
absorption in an electric accumulator factory with the use of personal
samplers. Br J Ind Med, 26: 202-216.
Williamson AM & Teo RKC (1986) Neurobehavioural effects of
occupational lead exposure. Br J Ind Med, 43: 374-380.
Wilson D, Esterman A, Lewis M, Roder D, & Calder I (1986) Children's
blood lead levels in the lead smelting town of Port Pirie South
Australia. Arch Environ Health, 41: 245-250.
Winneke G (1979) Modification of visual evoked potentials in rats
after long-term blood lead elevation. Acta Nerv Sup (Praha),
21: 282-284.
Winneke G, Brockhaus A, & Baltissen R (1977) Neurobehavioural and
system effects of long term blood lead-elevation in rats. I.
Discrimination learning and open field-behavior. Arch Toxicol,
37: 247-263.
Winneke G, Hrdina K-G, & Brockhaus A (1982a) Neuropsychological
studies in children with elevated tooth-lead concentrations. I. A
pilot study. Int Arch Occup Environ Health, 51: 169-183.
Winneke G, Lilienthal H, & Werner W (1982b) Task dependent
neurobehavioural effects of lead in rats. Arch Toxicol, 5(Suppl):
84-93.
Winneke G, Kramer U, Brockhaus A, Ewers U, Kujanek G, Lechner H, &
Janke W (1983) Neuropsychological studies in children with elevated
tooth-lead concentrations. II. Extended study. Int Arch Occup Environ
Health, 51: 231-252.
Winneke G, Beginn U, Ewert T, Havestadt C, Kraemer U, Krause C, Thron
HL, & Wagner HM (1985) Comparing the effects of perinatal and later
childhood lead exposure on neuropsychological outcome. Environ Res,
38: 155-167.
Winneke G, Collet W, Krämer U, Brockhaus A, Ewert T, & Krause C (1989)
Follow-up studies in lead-exposed children. In: Smith MA, Grant LD, &
Sors AI ed. Lead exposure and child development: An international
assessment. Dordrecht, London, Kluwer Academic Publishers, pp 260-270.
Winneke G, Brockhaus A, Ewers U, Kramer U, & Neuf M (1990) Results
from the European multicenter study on lead neurotoxicity in children:
implications for risk assessment. Neurotoxicol Teratol, 12: 553-559.
Wong PTS, Chau YK, & Luxon PL (1975) Methylation of lead in the
environment. Nature (Lond), 253: 263-264.
Wooler K & Melamed S (1978) Simple tests for monitoring excessive lead
absorption. Med J Aust, 1: 163-169.
World Bureau of Metal Statistics (1992) World metal statistics, July,
45(7): 75-78.
Worley PF, Barabon JM, Desouza EB, & Snyder SH (1986) Mapping second
messenger systems in the brain: differential localizations of
adenylate cyclase and protein kinase. C. Proc Natl Acad Sci (USA),
83: 4053-4057.
Worth D, Matranga A, Lieberman M, Devos E, Karelekas P, Ryan C, &
Kraun G (1981) Lead in drinking water: The contribution of household
tap water to blood lead levels. In: Lynam DR, Piantanida LG, & Cole JF
ed. Environmental lead. Proceedings of the Second International
Symposium on Environmental Lead Research. New York, London, San
Francisco, Academic Press, pp 199-225.
Yankel AJ, von Lindern IH, & Walter SD (1977) The Silver Valley lead
study: The relationship between childhood blood lead levels and
environmental exposure. J Air Pollut Control Assoc, 27: 763-767.
Yip R & Dallman PR (1984) Developmental changes in erythrocyte
protoporphyrin: Roles of iron deficiency and lead toxicity. J Pediatr,
710-713.
Yule W, Lansdown R, Millar IB, & Urbanowicz M-A (1981) The
relationship between blood lead concentrations, intelligence and
attainment in a school population: a pilot study. Dev Med Child
Neurol, 23: 567-576.
Zelikoff JT, Li JH, Hartwig A, Wang XW, Costa M, & Rossman TG (1988)
Genetic toxicology of lead compounds. Carcinogenesis, 9: 1727-1732.
Zerez CR, Wong MD, & Tanaka KR (1990) Partial purification and
properties of nicotinamide adenine dinucleotide synthetase from human
erythrocytes: evidence that enzyme activity is a sensitive indicator
of lead exposure. Blood, 75: 1576-1582.
Zhu BG (1984) Study of chronic lead-poisoning from using tea kettles.
Chung-Hua Yu Fang I Hsueh Tsa Chih, 18: 328-330.
Ziegler EE, Edwards BB, Jensen RL, Mahaffey KR, & Fomon SJ (1978)
Absorption and retention of lead by infants. Pediatr Res, 12: 29-34.
Zuckerman MA, Savory D, & Rayman G (1989) Lead encephalopathy from an
imported Toby mug. Postgrad Med J, 65: 307-309.
RESUME
La présente monographie est consacrée aux risques pour la santé
humaine découlant de l'exposition au plomb et aux dérivés minéraux du
plomb. On y insiste sur les données dont on a eu connaissance depuis
la publication du No 3 de la Série des Critères de l'environnement:
Plomb (OMS, 1977). Quant aux effets du plomb au niveau de
l'environnement, ils sont étudiés dans le No 85 de cette série: Lead -
Environmental Aspects (Le plomb dans l'environnement) (WHO, 1989, en
anglais seulement).
1. Identité, propriétés physiques et chimiques et méthodes d'analyse
Le plomb est un métal mou, gris argent, qui fond à 327,5°C. Il
est très résistant à la corrosion mais se dissout à chaud dans l'acide
nitrique et l'acide sulfurique. La valence habituelle du plomb dans
ses dérivés minéraux est de +2. La solubilité dans l'eau de ses
composés est variable, le sulfure et les oxydes étant peu solubles et
le nitrate, le chlorate et le chlorure assez solubles à chaud. Le
plomb forme également des sels avec des acides organiques tels que
l'acide lactique et l'acide acétique ainsi que des composés organiques
stables tels que le plomb-tétraéthyle et le plomb-tétraméthyle.
Les méthodes d'analyse les plus couramment utilisées pour doser
le plomb à faible concentration dans les produits biologiques et dans
l'environnement sont la spectrométrie d'absorption avec atomisation en
flamme, en four à cellules de graphite et en plasma induit par haute
fréquence (ICP/PIHF), ainsi que la coulométrie avec redissolution
anodique. Selon le traitement préalable subit par l'échantillon et les
techniques d'extraction et l'instrumentation utilisées, la limite de
détection peut être de 0,12 µmoles de plomb par litre de sang
(2,49 µg/dl). Toutefois, on n'obtient de résultats fiables qu'en se
conformant à une marche à suivre bien codifiée pour réduire au minimum
le risque de contamination au cours du prélèvement des échantillons,
de leur conservation, de leur traitement et de leur analyse.
2. Sources d'exposition humaine
La proportion du plomb dans la croûte terrestre est d'environ
20 mg/kg. Le plomb présent dans l'environnement peut être d'origine
naturelle ou artificielle. Le plomb naturellement présent dans
l'atmosphère provient, entre autres, de l'altération des roches et des
éruptions volcaniques, et on l'estime à 19 000 tonnes/an, contre
126 000 tonnes/an provenant des mines et des fonderies, la
consommation annuelle totale étant supérieure à 3 millions de tonnes.
On a trouvé des concentrations de plomb atmosphérique de
50 pg/m3 dans des zones reculées. La concentration de fond du plomb
dans le sol varie de 10 à 70 mg/kg et on a relevé à proximité de
routes une teneur moyenne de 138 mg/kg. Actuellement la teneur de
l'eau en plomb dépasse rarement quelques microgrammes/litre; la
concentration naturelle du plomb dans les eaux de surface est estimée
à 0,02 µg/litre.
Le plomb et ses dérivés peuvent pénétrer dans l'environnement par
une porte d'entrée quelconque lors de l'extraction minière, des
opérations de fonte et du traitement, de l'utilisation, du recyclage
ou du rejet du plomb. Le plomb est principalement utilisé dans les
batteries, les câbles, les pigments, les additifs pour essence, la
brasure et les aciers. Le plomb et ses dérivés entrent également dans
la composition de la brasure que l'on utilise pour souder les
conduites d'eau et les boites de conserve alimentaire; il sert
également à la confection de certains remèdes traditionnels, des
capsules de bouteilles contenant des boissons alcoolisées, des vernis
pour céramique et de la verrerie de cristal. Dans les pays où l'on
utilise encore de l'essence au plomb, les principales émissions dans
l'atmosphère proviennent de sources de combustion de produits
pétroliers, sources qui peuvent être mobiles ou fixes (en
agglomération). Les émissions dans l'atmosphère sont également très
importantes à proximité des mines et des fonderies de plomb.
Le plomb en suspension dans l'air peut se déposer sur le sol et
dans l'eau et il parvient ainsi jusqu'à l'homme par l'intermédiaire de
la chaîne alimentaire et de l'eau de boisson. Le plomb atmosphérique
contribue également pour une part importante au plomb présent dans la
poussière domestique.
3. Transport, distribution et transformation dans l'environnement
Le transport et la distribution du plomb à partir de sources
fixes, mobiles ou naturelles, s'effectue principalement par
l'intermédiaire de l'air. La majeure partie du plomb émis dans l'air
se redépose à proximité de la source d'émission, même si quelques
particules (de diamètre < 2 µm) dont transportées sur de longues
distances et provoquent la contamination de sites aussi reculés que
les glaciers arctiques. Le plomb aéroporté peut contribuer à
l'exposition humaine par contamination des aliments, d l'eau et de la
poussière, ainsi que par inhalation directe. L'élimination du plomb
aéroporté dépend des conditions atmosphériques et de la taille des
particules en suspension. De grandes quantités de plomb peuvent être
déversées dans le sol et l'eau. Cependant, ces produits ont tendance à
rester sur place du fait de la médiocre solubilité dans l'eau des
dérivés du plomb.
Le plomb qui s'est déposé dans l'eau, que ce soit à partir de
l'air ou par lessivage des sols, se répartit rapidement entre les
sédiments et la phase aqueuse, selon la valeur du pH, la teneur en
sels et la présence éventuelle d'agents chélateurs organiques. Lorsque
le pH dépasse 5,4, l'eau dure peu contenir environ 30 µg de
plomb/litre et l'eau douce environ 500 µg/litre. Une très faible
quantité du plomb déposé sur le sol passe dans les eaux de surface ou
les eaux souterraines sauf en cas d'érosion ou d'altération des
roches; il est normalement fortement lié par chélation aux matières
organiques.
Le plomb en suspension dans l'air peut passer dans les êtres
vivants soit directement, soit par fixation à partir du sol. Les
animaux peuvent être exposés au plomb de manière directe par broutage
de l'herbe, ingestion de terre ou par inhalation. Le plomb minéral ne
subit qu'une faible bioamplification le long de la chaîne alimentaire.
4. Concentrations dans l'environnement et exposition humaine
Dans la population générale adulte des non-fumeurs, la principale
voie d'exposition est la consommation de nourriture et d'eau. Le plomb
est suspension dans l'air peut jouer un rôle important dans
l'exposition, en fonction de facteurs tels que le tabagisme, la
profession, la proximité d'une autoroute, d'une fonderie, etc. ou de
certains lieux de loisirs (par exemple, ateliers d'artisanat, stands
de tir, etc.). Pour les nourrissons et les enfants en bas âge, la
nourriture, l'air, l'eau et la poussière ou la terre sont les
principales voies d'exposition potentielles. Pour les nourrissons
jusqu'à quatre ou cinq mois, les principales sources d'exposition au
plomb sont l'air, le lait et les laits maternisés ainsi que l'eau.
Les concentrations de plomb observées dans l'air, les aliments,
l'eau et le sol ou la poussière varient largement d'une région à
l'autre du monde et dépendent du degré d'industrialisation,
d'urbanisation ainsi que du mode de vie. Dans l'air ambiant, des
concentrations supérieures à 10 µg/m3 ont été signalées en milieu
urbain à proximité d'une fonderie, alors que dans les villes où l'on
n'utilise plus d'essence au plomb, ces concentrations peuvent tomber
en-dessous de 0,2 µg/m3. Dans ces conditions, la dose de plomb
absorbée à partir de l'air peut varier de moins de 4 µg/jour à plus de
200 µg/jour.
Dans des échantillons d'eau potable prélevés à la source, on
trouve généralement des concentrations de plomb inférieures à
5 µg/litre. Cependant, lorsque l'eau est prélevée au robinet dans les
maisons dont les canalisations sont en plomb, les teneurs peuvent
dépasser 100 µg/litre, en particulier lorsque l'eau a séjourné dans la
tuyauterie pendant plusieurs heures.
L'exposition au plomb par la voie alimentaire dépend de nombreux
facteurs tenant au mode de vie et notamment à la nature des aliments
consommés, au mode de préparation, au fait que l'on utilise ou non de
la brasure au plomb, à la teneur de l'eau en plomb et à l'utilisation
de vaisselle recouverte d'un vernis au plomb.
Les nourrissons et les enfants peuvent souvent être très exposés
au plomb présent dans la poussière et dans la terre. La teneur de la
poussière en plomb dépend de facteurs tels que la vétusté et l'état de
la maison, l'utilisation de peintures à base de plomb, la présence de
plomb dans l'essence et la densité urbaine. La dose de plomb absorbée
dépendra également de l'âge et du comportement de l'enfant ainsi que
de la biodisponibilité du plomb dans le produit où il est présent.
Chez les ouvriers des industries où l'on produit, raffine,
utilise ou rejette du plomb ou des dérivés du plomb, la principale
voie d'exposition est l'inhalation. Au cours d'un poste de travail de
8 heures, les ouvriers peuvent en absorber des quantités atteignant
400 µg, qui s'ajoutent aux 20 ou 30 µg absorbés quotidiennement à
partir de la nourriture, de l'eau et de l'air ambiant; l'ingestion de
grosses particules peut également contribuer de façon importante à cet
apport.
5. Cinétique et métabolisme chez les animaux de laboratoire et
l'homme
L'homme et les animaux résorbent le plomb qu'ils ingèrent ou
inhalent; la résorption percutanée est minime chez l'homme. Selon sa
granulométrie, sa forme chimique et sa solubilité dans les liquides
biologiques, un dérivé du plomb peut être résorbé jusqu'à hauteur de
50% après avoir été inhalé. Certaines grosses particules (plus de
7 µm) sont avalées après avoir été rejetées des voies respiratoires
par l'action de l'ascenseur muco-ciliaire. Chez l'homme et les animaux
d'expérience, l'absorption du plomb dans les voies digestives dépend
de la nature physico-chimique du produit ingéré, de l'état
nutritionnel du sujet ainsi que du type d'aliments consommés. Chez
l'homme adulte, le plomb contenu dans les aliments est absorbé à peu
près à hauteur de 10%; la proportion est plus élevée lorsque le sujet
est à la diète. Toutefois, chez les nourrissons et les enfants en bas
âge, le plomb d'origine alimentaire peut être absorbé à hauteur de
50%, encore que le taux d'absorption du plomb provenant des poussières
ou de la terre ainsi que des écailles de peinture puisse être plus
faible, selon sa biodisponibilité. Les régimes alimentaires pauvres en
calcium, phosphates, sélénium ou zinc peuvent accroître l'absorption
du plomb. Le fer et la vitamine D influent également sur l'absorption
du plomb.
Le taux de plomb dans le sang ou plombémie est utilisé pour
évaluer la charge en plomb de l'organisme ainsi que les doses de plomb
(internes) absorbées. La relation entre la plombémie et la
concentration du plomb dans les diverses sources d'exposition n'est
pas linéaire (corrélation curviligne).
Une fois absorbé, le plomb ne se répartit pas de manière homogène
dans l'organisme. Après être rapidement passé dans le sang et les
tissus mous, il se redistribue lentement des les os. La plomb
s'accumule dans les os pendant une longue période de la vie humaine et
peut servir de source endogène de plomb. La demi-vie du plomb dans le
sang et les tissus mous est d'environ 28 à 36 jours mais elle peut
être beaucoup plus longue dans les diverses parties de l'os. Le taux
de rétention du plomb dans l'organisme est plus élevé chez l'enfant
que chez l'adulte. Pendant toute la durée de la gestation, le plomb
passe facilement de la mère au foetus.
La plombémie est la mesure la plus couramment utilisée pour
évaluer l'exposition au plomb. Toutefois, on dispose aujourd'hui de
techniques permettant de doser le plomb dans les dents et les os,
encore que la cinétique du phénomène ne soit pas parfaitement
élucidée.
6. Effets sur les animaux de laboratoire et les systèmes d'épreuves
in vitro
Chez toutes les espèces d'animaux de laboratoire étudiées, y
compris des primates non-humains, on a constaté que le plomb
produisait des effets indésirables au niveau de plusieurs organes et
systèmes, notamment le système haematopoïétique, le système nerveux,
les reins, le système cardio-vasculaire, l'appareil reproducteur et le
système immunitaire. Le plomb est également nocif pour les os et on a
montré qu'il avait des effets cancérogènes sur le rat et la souris.
Malgré les différences d'ordre cinétique qui existent entre les
animaux d'expérience et l'homme, ces études apportent des arguments
biologiques de poids à la plausibilité de tels effets chez l'homme.
Chez le rat, on a observé une diminution de la capacité
d'apprentissage et de mémorisation lorsque la plombémie était de
l'ordre de 0,72 à 0,96 µmol/litre (soit 15 à 20 µg/dl), les mêmes
effets étant observés chez des primates non-humains pour une plombémie
ne dépassant pas 0,72 µmol/litre (15 µg/dl). En outre,
l'expérimentation animale a également permis d'observer des troubles
de la vision et de l'audition.
Chez le rat, une plombémie supérieure à 2,88 µmol/litre
(60 µg/dl), soit une valeur analogue à celle qui, selon les
observations, constitue le seuil d'apparition des effets chez l'homme,
est à même de provoquer l'apparition d'effets néphrotoxiques. Des
effets cardio-vasculaires ont été également observés chez des rats
après exposition chronique à de faibles doses de plomb entraînant une
plombémie de l'ordre de 0,24 à 1,92 µmol/litre (5 à 40 µg/dl). A des
doses inférieures à la dose maximale tolérée qui est de 200 mg de
plomb (sous forme d'acétate) par litre d'eau potable, on a observé
l'apparition de tumeurs. Cette dose constitue la dose maximale qui
n'entraîne pas d'autres effets morphologiques ou fonctionnels.
7. Effets sur l'homme
Chez l'homme, le plomb peut produire des effets biologiques très
divers selon l'intensité et la durée de l'exposition. On a ainsi
observé des effets au niveau infracellulaire ainsi que des effets
s'exerçant sur les fonctions générales de l'organisme, effets qui vont
de l'inhibition de certaines enzymes jusqu'à l'apparition
d'altérations morphologiques marquées et à la mort. Ces altérations se
produisent dans de larges limites de doses, l'organisme humain en
développement étant généralement plus sensible que l'organisme adulte.
On a montré que le plomb avait des effets sur nombre de processus
biochimiques; en particulier on a largement étudié les effets qu'il
produit sur la synthèse hémique, tant chez l'adulte que chez l'enfant.
Lorsque la plombémie est élevée, on observe une augmentation du taux
de protoporphyrine érythrocytaire sérique ainsi qu'une augmentation de
l'excrétion urinaire de coproporphyrine et d'acide delta-aminolévulinique.
A des valeurs plus faibles de la plombémie, on observe l'inhibition
d'enzymes comme la delta-aminolévulinique acide-déshydratase et la
dihydrobioptérine-réductase.
Les effets du plomb sur le système haématopoïétique se traduisent
par une diminution de la synthèse de l'hémoglobine et l'on a observé
une anémie chez des enfants lorsque la plombémie dépassait
1,92 µmol/litre (40 µg/dl).
Pour des raisons d'ordre neurologique, métabolique et
compartementale, les enfants sont plus sensibles aux effets du plomb
que les adultes. Des études épidémiologiques tant prospectives que
transversales ont été effectuées pour déterminer dans quelle mesure
une exposition au plomb présent dans l'environnement affecte les
fonctions psychologiques dépendant du système nerveux central. On a
ainsi montré qu'il existait une association entre l'exposition au
plomb et les troubles des fonctions neurocomportementales chez
l'enfant.
On a également constaté des anomalies des fonctions
psychologiques et neurocomportementales chez des ouvriers longtemps
exposés au plomb. Il a été montré que les paramètres
électrophysiologiques étaient d'utiles indicateurs des effets
infracliniques du plomb sur le système nerveux central.
On sait depuis longtemps que la neuropathie périphérique est due
à une exposition prolongée à de fortes concentrations de plomb sur le
lieu de travail. A plus faibles concentrations, on a observé une
diminution de la vitesse de conduction nerveuse. Ces effets se
révèlent souvent réversibles, en fonction de l'âge et de la durée de
l'exposition, après cessation de l'exposition.
Les effets que le plomb exerce sur le coeur sont indirects et
s'opèrent par l'intermédiaire du système neuro-végétatif; il n'y a pas
d'effets directs sur le myocarde. L'ensemble des faits tirés des
études sur des populations d'adultes indiquent qu'il existe une très
faible association entre la plombémie et la tension artérielle
systolique ou diastolique. Etant donné la difficulté qu'il y a à tenir
compte des facteurs de confusion, il n'a pas été possible d'établir, à
partir des résultats de ces études, une relation de cause à effet.
Rien n'indique non plus que l'association qui pourrait exister entre
la plombémie et la tension artérielle constitue un problème médical
majeur.
On sait que le plomb peut entraîner des lésions tubulaires
proximales qui se caractérisent par une aminoacidurie généralisée, une
hypophosphatémie avec d'une hyperphosphaturie relative et une
glycosurie accompagnée d'inclusions nucléiques, d'altérations des
mitochondries et d'une hypertrophie des cellules de l'épithélium
tubulaire proximal. Ces effets sur les tubules s'observent après une
exposition relativement brève et sont généralement réversibles. En
revanche, les altérations scléreuses et les fibroses interstitielles
qu'on observe lors d'une exposition chronique à de fortes
concentrations de plomb, entraînent des problèmes fonctionnels qui
peuvent déboucher sur une insuffisance rénale. On a observé une
augmentation du risque de néphropathie chez les travailleurs dont la
plombémie dépassait 3,0 µmol/litre (soit environ 60 µg/dl). En
utilisant des indicateurs fonctionnels plus sensibles, on a récemment
découvert l'existence d'effets rénaux dans la population générale.
Les effets du plomb sur la fonction de reproduction, ne
concernent, chez l'homme, que la morphologie et le nombre des
spermatozoïdes. Chez la femme, on a attribué au plomb un certain
nombre de grossesses à issue défavorable.
Il ne semble pas que le plomb ait des effets nocifs sur la peau,
les muscles ou le système immunitaire. Si l'on excepte le cas du rat,
le plomb ne paraît pas non plus être à l'origine de l'apparition de
tumeurs.
8. Evaluation des risques pour la santé humaine
Le plomb a des effets nocifs sur plusieurs organes et systèmes,
les effets les plus sensibles étant relevés au niveau infracellulaire
ainsi que sur le développement du système nerveux. On a fait état
d'une association entre la plombémie et l'hypertension. En outre, le
plomb entraîner une cascade d'effets sur les réserves hémiques de
l'organisme et il perturbe également la synthèse hémique. Cependant,
certains de ces effets ne sont pas véritablement considérés comme
délétères. Il y a également perturbation de l'homéostase du calcium
avec des contre-coups sur d'autres processus cellulaires.
a) Les preuves les plus substantielles sont on dispose à propos de
l'action nocive du plomb proviennent d'études transversales et
prospectives sur des populations dont la plombémie est
généralement inférieure à 1,2 µmol/litre (25 µg/dl) et concernent
une diminution du quotient d'intelligence (QI). Il importe
cependant de noter que ces observations ne constituent pas une
preuve définitive d'une relation de cause à effet entre ce
phénomène et l'exposition au plomb. Cependant la mesure de ce QI
à partir de l'âge de 4 ans fait ressortir un déficit qui se situe
entre 0 et 5 points (sur une échelle avec un écart-type de 15)
pour une augmentation de la plombémie de 0,48 µmol/litre
(10 µg/dl), l'ampleur de l'effet étant vraisemblablement de 1 à 3
points. Lorsque la plombémie dépasse 1,2 µmol/litre (25 µg/dl),
on peut avoir une relation différente entre cette plombémie et le
QI. Les estimations de l'ampleur de l'effet observé sont des
moyennes calculées sur des groupes et ne représentent qu'une
probabilité pour un individu en particulier.
Les études épidémiologiques dont on dispose ne donnent pas
de preuves définitives de l'existence d'un seuil. Lorsque la
plombémie se situe en-dessous de l'intervalle
0,48-0,72 µmol/litre (10-15 µg/dl), l'incertitude qui entache
toute estimation de l'effet s'accroît, du fait des facteurs de
confusion et des limites dans la précision des dosages et des
tests psychométriques. Il n'en reste pas moins qu'en-dessous de
cet intervalle de valeurs, certains éléments incitent à penser à
l'existence d'une association.
b) L'expérimentation animale milite en faveur de l'existence d'une
relation de cause à effet entre l'exposition au plomb et certains
effets neurologiques, puisqu'elle fait ressortir des déficits
dans les fonctions cognitives pour une plombémie ne dépassant pas
0,53 à 0,72 µmol/litre (11-15 µg/dl), déficits qui peuvent
persister bien après la cessation de l'exposition au plomb.
c) Pour une plombémie ne dépassant pas 1,44 µmol/litre (30 µg/dl),
il peut y avoir réduction de la vitesse de conduction nerveuse
périphérique chez l'homme. En outre, pour des valeurs de la
plombémie de dépassant pas 1,92 µmol/litre (40 µg/dl), il peut
également y avoir perturbation des fonctions moto-sensorielles et
la fonction du système nerveux neurovégétatif (variabilité de
l'intervalle R-R sur l'électrocardiogramme) peut être affectée
pour une valeur moyenne de la plombémie d'environ 1,68 µmol/litre
(35 µg/dl). Chez les travailleurs dont la plombémie dépasse
2,88 µmol/litre (60 µg/dl), il y a accroissement du risque de
néphropathie saturnienne. Toutefois, des études récentes basées
sur des indicateurs plus sensibles de la fonction rénale incitent
à penser que des effets peuvent se produire à des valeurs plus
faibles de l'exposition au plomb.
d) Il semblerait que l'exposition au plomb soit associée à une
légère augmentation de la tension artérielle. L'ordre de grandeur
probable de cette augmentation est le suivant: pour un doublement
de la plombémie (par exemple lorsqu'elle passe de 0,8 à
1,6 µmol/litre, soit de 16,6 à 33,3 µg/dl), il y a une
augmentation moyenne de 1 mmHg de la systolique. L'association
avec la diastolique est analogue mais d'ampleur plus faible.
Toutefois, on se demande si ces associations statistiques
résultent réellement de l'exposition au plomb ou s'il s'agit d'un
artefact imputable à des facteurs de confusion.
e) Certaines études épidémiologiques - mais pas toutes - font état
d'une association liée à la dose entre les accouchements avant
terme et certains indices de la croissance et de la maturation
foetales à des valeurs de la plombémie supérieures ou égales à
0.72 µmol/litre (15 µg/dl).
f) Les données relatives à la cancérogénicité pour l'homme du plomb
et de plusieurs de ses dérivés minéraux, sont insuffisantes.
g) On a mis en évidence des effets que le plomb exerce sur un
certain nombre de systèmes enzymatiques et de paramètres
biochimiques. Les valeurs de la plombémie au-dessus desquelles on
peut mettre en évidence des effets avec les techniques actuelles,
pour ce qui est des paramètres susceptibles d'avoir une
importance clinique, sont toutes supérieures à 0,96 µmol/litre
(20 µg/dl). Certains effets sur les enzymes peuvent être mis en
évidence à des valeurs plus faibles de la plombémie, mais leur
signification clinique demeure incertaine.
RESUMEN
La presente monografía se centra en los riesgos para la salud
humana asociados a la exposición al plomo y a los compuestos
inorgánicos de plomo. Se han destacado los datos disponibles desde la
publicación de Criterios de Salud Ambiental No. 3: Plomo (OMS, 1977).
Los efectos ambientales del plomo se examinan en Environmental Health
Criteria 85: Lead - Environmental Aspects (OMS, 1989).
1. Identidad, propiedades físicas y químicas y métodos analíticos
El plomo es un metal blando, gris plateado, que se funde a
327,5°C. Es muy resistente a la corrosión, pero es soluble en ácido
nítrico y en ácido sulfúrico caliente. Su valencia corriente en los
compuestos inorgánicos es +2. Su solubilidad en agua varía; el sulfito
de plomo y los óxidos de plomo son poco solubles, mientras que las
sales de nitrato, clorato y cloruro son razonablemente solubles en
agua fría. El plomo también forma sales con ácidos orgánicos tales
como el láctico y el acético, y compuestos orgánicos estables tales
como el tetraetilo de plomo y el tetrametilo de plomo.
Los métodos utilizados más corrientemente para el análisis de
bajas concentraciones de plomo en materias biológicas y ambientales
son la llama, el horno de grafito y la espectroscopia de absorción
atómica de plasma acoplado inductivamente y la voltimetría de
separación anódica. Según sean el tratamiento previo de la muestra,
las técnicas de extracción y la instrumentación analítica, pueden
alcanzarse niveles de detección de 0,12 µmoles de plomo por litro de
sangre (2,49 µg/dl). Sin embargo, se obtienen resultados fiables sólo
cuando se siguen procedimientos específicos para reducir al mínimo el
riesgo de contaminación durante la recogida, el almacenamiento,
procesamiento y análisis de la muestra.
2. Fuentes de exposición humana
El nivel de plomo en la corteza terrestre es de aproximadamente
20 mg/kg. El plomo del medio ambiente puede provenir de fuentes
naturales o antropogénicas. Las fuentes naturales de plomo atmosférico
comprenden el desgaste geológico y las emisiones volcánicas y se han
estimado en 19 000 toneladas por año, frente a unas 126 000 toneladas
por año emitidas en el aire como resultado de la minería, la fundición
y el consumo de más de 3 millones de toneladas de plomo por año.
Se han encontrado concentraciones atmosféricas de plomo de
50 pg/m3 en zonas remotas. Los niveles básicos de plomo en el suelo
oscilan entre 10 y 70 mg/kg y se ha comunicado un nivel medio de
138 mg/kg en las proximidades de las carreteras. Los niveles de plomo
presentes en las aguas rara vez exceden de unos pocos microgramos por
litro; la concentración natural de plomo en las aguas superficiales se
ha estimado en 0,02 µg/litro.
El plomo y sus compuestos pueden entrar en el medio ambiente en
cualquier punto durante las actividades de minería, fundición,
elaboración, utilización, reciclado o eliminación. Se utiliza
principalmente en la fabricación de pilas, cables, pigmentos, aditivos
de la gasolina, productos para soldar y de acero. El plomo y los
compuestos de plomo también se utilizan para soldar las tuberías de
distribución de agua y las latas de conserva, en algunos remedios
tradicionales, en las tapas de las botellas de bebidas alcohólicas y
en los esmaltes cerámicos y la cristalería de mesa. En los países
donde todavía se utiliza gasolina con plomo, la principal emisión en
el aire proviene de fuentes móviles y estacionarias de combustión de
gasolina (centros urbanos). Las zonas próximas a las minas y funderías
de plomo están expuestas a la emisión de niveles elevados en el aire.
El plomo del aire puede depositarse en el suelo y el agua, desde
donde llega al ser humano por conducto de la cadena alimentaria y del
agua de bebida. El plomo atmosférico también es una fuente importante
del plomo presente en el polvo de las viviendas.
3. Transporte, distribución y transformación en el medio ambiente
El transporte y la distribución del plomo procedente de fuentes
fijas, móviles y naturales tienen lugar principalmente a través del
aire. La mayor parte de las emisiones de plomo se depositan cerca de
la fuente, aunque algunas partículas de materia (< 2 µm de diámetro)
recorren largas distancias y contaminan lugares remotos tales como los
glaciares árticos. El plomo del aire puede contribuir a la exposición
humana mediante la contaminación de los alimentos, del agua y del
polvo, así como por inhalación directa. La eliminación del plomo del
aire depende de las condiciones atmosféricas y del tamaño de las
partículas. Pueden descargarse grandes cantidades de plomo en el suelo
y en el agua. Sin embargo, ese material tiende a permanecer localizado
debido a la escasa solubilidad de los compuestos de plomo en el agua.
El plomo depositado en el agua, ya provenga del aire o de la
escorrentía del suelo, se distribuye rápidamente entre el sedimento y
la fase acuosa, según el pH, el contenido de sales y la presencia de
agentes quelantes orgánicos. Con un pH superior a 5,4, las aguas duras
pueden contener aproximadamente 30 µg de plomo por litro y las aguas
blandas aproximadamente 500 µg de plomo por litro. Muy poco plomo
depositado en el suelo se transporta a las aguas superficiales o a las
subterráneas, salvo mediante la erosión o el desgaste geoquímico;
normalmente está ligado a la materia orgánica de forma bastante
estrecha (por quelación).
El plomo del aire puede transferirse a la biota directamente o
por absorción del suelo. Los animales pueden encontrarse expuestos al
plomo directamente mediante la ingestión de hierba y de tierra o por
inhalación. Hay poca biomagnificación del plomo inorgánico a través de
la cadena alimentaria.
4. Niveles ambientales y exposición humana
En la población general que no fuma, la principal vía de
exposición son los alimentos y el agua. El plomo del aire puede
contribuir apreciablemente a la exposición, lo que depende de factores
tales como el consumo de tabaco, la ocupación, la proximidad de
caminos transitados por vehículos automotores, de funderías de plomo,
etc., y ciertas actividades de esparcimiento (por ejemplo, artesanía,
tiro con armas de fuego). Los alimentos, el aire, el agua y el
polvo/suelo son las principales vías potenciales de exposición de los
niños pequeños. Para los niños de hasta 4 ó 5 meses de edad, el aire,
la leche, las preparaciones para lactantes y el agua son fuentes
notables de exposición al plomo.
Los niveles de plomo presentes en el aire, los alimentos, el
agua, y el suelo/polvo varían ampliamente en el mundo y dependen del
grado de desarrollo industrial y de urbanización y de factores
relacionados con el modo de vida. Se han comunicado niveles superiores
a 10 µg/m3 presentes en el aire ambiental en zonas urbanas próximas
a funderías, mientras que en ciudades donde ha dejado de usarse la
gasolina con plomo se han detectado niveles inferiores a 0,2 µg/m3.
La absorción de plomo del aire puede, pues, variar de menos de
4 µg/día a más de 200 µg/día.
Los niveles de plomo en muestras de agua de bebida extraídas de
los manantiales suelen ser inferiores a 5 µg/litro. Sin embargo, el
agua del grifo de viviendas cuyas tuberías tienen plomo contiene
niveles que exceden de 100 µg/litro, en particular después de haber
reposado el agua en las tuberías durante algunas horas.
El nivel de exposición al plomo a través de la dieta depende de
muchos factores relacionados con el modo de vida, entre ellos los
alimentos que se consumen, la tecnología de elaboración, el empleo de
soldadura de plomo, los niveles de plomo en el agua y la utilización
de cerámica con barniz de plomo.
Para los niños, el plomo presente en el polvo y en el suelo suele
ser la principal vía de exposición. Los niveles de plomo en el polvo
dependen de factores tales como la antigüedad y el estado de la
vivienda, la utilización de pinturas a base de plomo, el plomo de la
gasolina y la densidad urbana. La absorción de plomo dependerá de la
edad y de las características comportamentales del niño así como de la
biodisponibilidad de plomo en la fuente material.
La inhalación es la vía principal de exposición al plomo para los
trabajadores de industrias que producen, refinan, utilizan o desechan
plomo y compuestos de plomo. Durante un turno de ocho horas, los
trabajadores pueden absorber nada menos que 400 µg de plomo, además de
los 20-30 µg/día que absorben de los alimentos, del agua y del aire
ambiental; puede haber una absorción notable como resultado de la
inhalación de partículas grandes.
5. Cinética y metabolismo en animales de laboratorio y en el ser
humano
Los seres humanos y los animales absorben plomo por inhalación o
por ingestión; la absorción percutánea es mínima en el ser humano.
Según la especiación química, el tamaño de las partículas y la
solubilidad de los líquidos corporales, puede absorberse hasta un 50%
de los compuestos de plomo inhalados. Algunas partículas de materia
inhaladas (de más de 7 µm) se degluten después de la eliminación
mucociliar del aparato respiratorio. En los animales experimentales y
en el ser humano, la absorción de plomo del aparato gastrointestinal
está influenciada por la naturaleza fisicoquímica del material
ingerido, el estado nutricional y el tipo de alimentación. En los
seres humanos adultos, se absorbe aproximadamente el 10% del plomo
contenido en la alimentación; la proporción es más elevada en
condiciones de ayuno. Sin embargo, los lactantes y los niños pequeños
absorben nada menos que el 50% del plomo presente en la alimentación,
pero la absorción de plomo del polvo/suelo y de desconchones de
pintura puede ser menor y depende de su biodisponibilidad. Las dietas
pobres en calcio, fosfato, selenio o zinc pueden dar lugar a una mayor
absorción de plomo. El hierro y la vitamina D también influyen en la
absorción de plomo.
Los niveles de plomo en la sangre (Pb-H) se utilizan para medir
la carga corporal y las dosis absorbidas (internas) de plomo. La
relación entre el plomo presente en la sangre y la concentración de
plomo en las fuentes de exposición es curvilínea.
Una vez absorbido, el plomo no se distribuye de manera homogénea
en todo el cuerpo. Hay una absorción rápida en la sangre y en los
tejidos blandos, seguida de una redistribución más lenta a los huesos.
Los huesos acumulan plomo durante gran parte de la vida humana y
pueden actuar como fuente endógena de plomo. La semivida del plomo en
la sangre y en otros tejidos blandos es de aproximadamente 28-36 días,
pero es mucho más larga en los diversos compartimentos óseos. La
retención porcentual de plomo en los depósitos corporales es más
elevada en los niños que en los adultos. La transferencia de plomo al
feto humano se efectúa fácilmente durante la gestación.
El nivel de plomo en la sangre es la medida más utilizada para
determinar la exposición al plomo. Sin embargo, ya se dispone de
técnicas para determinar la cantidad de plomo presente en los dientes
y en los huesos, aunque aún no se conoce del todo su cinética.
6. Efectos en los animales de laboratorio y en los sistemas in vitro
En todas las especies de animales de experimentación estudiadas,
inclusive en primates no humanos, se ha observado que el plomo tiene
efectos adversos en varios órganos y sistemas de órganos, inclusive
los sistemas hematopoyético, nervioso, renal, cardiovascular,
reproductivo e inmunitario. El plomo también afecta a los huesos y se
ha demostrado que es carcinógeno en ratas y ratones.
Pese a diferencias cinéticas entre las especies de animales
experimentales y la humana, dichos estudios apoyan firmemente y
justifican desde un punto de vista biológico los hallazgos realizados
en seres humanos. Se han comunicado deficiencias del aprendizaje y de
la memoria en ratas con niveles de Pb-H de 0,72-0,96 µmoles/litro
(15-20 µg/dl) y en primates no humanos con niveles de Pb-H de no más
de 0,72 µmoles/litro (15 µg/dl). Además, se han comunicado
deficiencias visuales y auditivas en estudios realizados en animales
de experimentación.
La toxicidad renal en las ratas parece presentarse a partir de un
nivel de Pb-H de 2,88 µmoles/litro (60 µg/dl); este valor es semejante
al que, según se ha comunicado, comienza a tener efectos renales en el
ser humano. Se han observado efectos cardio-vasculares en ratas
después de exposiciones crónicas a niveles bajos que dan lugar a
niveles de Pb-H de 0,24-1,92 µmoles/litro (5-40 µg/dl). Se ha
demostrado que aparecen tumores con dosis inferiores a la dosis máxima
tolerada, de 200 mg de plomo (como acetato de plomo) por litro de agua
de bebida. Esta es la dosis máxima no asociada a otros cambios
morfológicos o funcionales.
7. Efectos en el ser humano
En el ser humano, el plomo puede tener una amplia variedad de
efectos biológicos según el nivel y la duración de la exposición. Se
han observado efectos en el plano subcelular y efectos en el
funcionamiento general del organismo que van desde la inhibición de
las enzimas hasta la producción de acusados cambios morfológicos y la
muerte. Dichos cambios se producen a dosis muy diferentes; en general,
el ser humano que se está desarrollando es más sensible que el adulto.
Se ha mostrado que el plomo tiene efectos en muchos procesos
bioquímicos; en particular, se han estudiado mucho los efectos en la
síntesis del hemo en adultos y niños. Se observan niveles más altos de
porfirina eritrocitaria sérica y mayor excreción urinaria de
coproporfirina y de ácido delta-aminolevulínico cuando las
concentraciones de Pb-H son elevadas. Con niveles más bajos se observa
inhibición de las enzimas dehidratasa del ácido delta-aminolevulínico
y reductasa de la dihidrobiopterina.
Como resultado de los efectos del plomo en el sistema
hematopoyético disminuye la síntesis de hemoglobina y se ha observado
anemia en niños a concentraciones de Pb-H superiores a
1,92 µmoles/litro (40 µg/dl).
Por razones neurológicas, metabólicas y comportamentales, los
niños son más vulnerables a los efectos del plomo que los adultos. Se
han efectuado estudios epidemiológicos prospectivos y transversales
para evaluar la medida en que la exposición al plomo ambiental afecta
a las funciones psicológicas regidas por el sistema nervioso central
(SNC). Se ha mostrado que el plomo está asociado a deficiencias
neurocomportamentales en los niños.
Se han observado deficiencias psicológicas y
neurocomportamentales en trabajadores que habían estado expuestos al
plomo durante un tiempo prolongado. Los parámetros electrofisiológicos
han demostrado ser indicadores útiles de los efectos subclínicos del
plomo en el SNC.
Desde hace tiempo se sabe que la exposición prolongada a niveles
elevados de plomo en el medio laboral provoca neuropatías periféricas.
Con niveles más bajos se ha observado una reducción de la velocidad de
conducción nerviosa. Se ha observado a menudo que dichos efectos son
reversibles después de cesar la exposición, según la edad del sujeto y
la duración de la exposición.
Los efectos del plomo en el corazón son indirectos y se producen
por conducto del sistema nervioso autónomo; el plomo no tiene efectos
directos en el miocardio. Datos colectivos procedentes de estudios de
poblaciones adultas indican asociaciones muy débiles entre la
concentración de Pb-H y la presión arterial sistólica o diastólica.
Dada la dificultad de evaluar el influjo de los factores de confusión
pertinentes, no puede establecerse una relación causal sobre la base
de esos estudios. No hay indicios de que la relación entre la
concentración de Pb-H y la presión arterial tenga mucha importancia
para la salud.
Se sabe que el plomo provoca en los tubos proximales del riñón
lesiones que se caracterizan por aminoaciduria generalizada,
hipofosfatemia con hiperfosfaturia relativa y glucosuria acompañada de
cuerpos de inclusión nuclear, modificaciones mitocondriales y
citomegalia de las células epiteliales de los tubos proximales. Los
efectos tubulares se manifiestan después de una exposición
relativamente breve y suelen ser reversibles, mientras que los cambios
escleróticos y la fibrosis intersticial, que dan lugar a una
disminución de la función renal y a una posible insuficiencia renal,
requieren una exposición crónica a niveles elevados de plomo. Se ha
advertido un mayor riesgo de nefropatía en los trabajadores que tienen
niveles de Pb-H superiores a 3,0 µmoles/litro (aproximadamente
60 µg/dl). Recientemente se han observado efectos renales en la
población general tras haberse utilizado indicadores de función más
sensibles.
Los efectos del plomo en la función reproductora masculina se
limitan a la morfología y el número de los espermatozoides. En cuanto
a la femenina, se han atribuido al plomo algunos efectos adversos en
el embarazo.
El plomo no parece tener efectos nocivos en la piel, en los
músculos ni en el sistema inmunitario. Salvo en la rata, el plomo no
parece estar relacionado con el desarrollo de tumores.
8. Evaluación de los riesgos para la salud humana
El plomo tiene efectos adversos en varios órganos y sistemas de
órganos; los más delicados parecen ser los cambios subcelulares y los
efectos en el desarrollo del sistema nervioso. Se ha observado una
asociación entre el nivel de Pb-H y la hipertensión (presión
arterial). El plomo produce una serie de efectos en la reserva
corporal de hemo y afecta a la síntesis de éste. Sin embargo, algunos
de estos efectos no se consideran adversos. Está afectada la
homeostasia del calcio, lo que interfiere en otros procesos celulares.
a) Los datos más importantes de los estudios transversales y
prospectivos de poblaciones con niveles de Pb-H generalmente
inferiores a 1,2 µmoles/litro (25 µg/dl) se relacionan con una
disminución del coeficiente de inteligencia (CI). Es importante
señalar que esas observaciones no pueden constituir una prueba
concluyente de una relación causal con la exposición al plomo.
Sin embargo, la magnitud del efecto aparente en el CI,
determinado desde los 4 años en adelante, es un déficit de 0 a
5 puntos (en una escala con una desviación estándar de 15) por
cada 0,48 µmoles/litro (10 µg/dl) de aumento del nivel de Pb-H,
con una magnitud probable del efecto aparente de 1 a 3 puntos.
Con niveles de Pb-H superiores a 1,2 µmoles/litro (25 µg/dl), la
relación entre el Pb-H y el CI puede ser diferente. Las
estimaciones de la magnitud del efecto constituyen promedios
grupales y sólo se aplican a cada niño de manera probabilística.
Los estudios epidemiológicos existentes no prueban de modo
concluyente la existencia de un umbral. Por debajo de unos
niveles de Pb-H de 0,48-0,72 µmoles/litro (10-15 µg/dl), los
efectos de las variables de confusión y los límites de la
precisión de las mediciones analíticas y psicométricas aumentan
la incertidumbre inherente a toda estimación de un efecto. Sin
embargo, hay algunos indicios de una asociación por debajo de
dichos niveles.
b) Los estudios realizados en animales respaldan la idea de una
relación causal entre el plomo y ciertos efectos en el sistema
nervioso; se señalan deficiencias cognitivas a niveles de Pb-H de
sólo 0,53-0,72 µmoles/litro (11-15 µg/dl), deficiencias que
pueden persistir mucho después de haber terminado la exposición
al plomo.
c) Puede producirse una reducción de la velocidad de conducción
nerviosa periférica en el ser humano con niveles de Pb-H de sólo
1,44 µmoles/litro (30 µg/dl). Además, las funciones
sensitivomotrices pueden verse disminuidas con niveles de Pb-H de
sólo 1,92 µmoles/litro (40 µg/dl) aproximadamente y las funciones
del sistema nervioso autónomo (variabilidad del intervalo R-R
electrocardiográfico) pueden verse afectadas a un nivel promedio
de Pb-H de aproximadamente 1,68 µmoles por litro (35 µg/dl). El
riesgo de nefropatía plúmbica aumenta en los trabajadores que
tienen niveles de Pb-H superiores a 2,88 µmoles/litro (60 µg/dl).
Sin embargo, estudios recientes que utilizan indicadores más
sensibles de la función renal sugieren la aparición de efectos
renales a niveles más bajos de exposición al plomo.
d) La exposición al plomo está asociada a un pequeño aumento de la
presión arterial. El orden de magnitud probable es que por cada
duplicación del nivel de Pb-H (por ejemplo, de 0,8 a
1,6 µmoles/litro, es decir, de 16,6 a 33,3 µg/dl) hay un aumento
medio de 1 mmHg de presión arterial sistólica. La relación con la
presión diastólica es de una magnitud semejante pero más pequeña.
Sin embargo, no se sabe bien si estas asociaciones estadísticas
obedecen realmente a un efecto de la exposición al plomo o son un
resultado ficticio debido a factores de confusión.
e) Algunos estudios epidemiológicos, no todos, muestran una
relación, dependiente de la dosis, con el parto prematuro y
algunos índices de crecimiento y maduración fetales a niveles de
Pb-H de 0,72 µmoles/litro (15 µg/dl) o más.
f) Los indicios de carcinogenicidad del plomo y de varios compuestos
inorgánicos de plomo en el ser humano son insuficientes.
g) Se ha demostrado que el plomo tiene efectos en cierto número de
sistemas enzimáticos y de parámetros bioquímicos. Los niveles de
Pb-H por encima de los cuales las técnicas vigentes pueden
demostrar la presencia de efectos en relación con los parámetros
de importancia clínica posible son todos superiores a
0,96 µmoles/litro (20 µg/dl). Algunos efectos en enzimas pueden
demostrarse con niveles de Pb-H más bajos, pero su importancia
clínica es incierta.
8.3.6.4 Task group overview of cross-sectional studies
a) Methods of controlling for confounders
A particular methodological concern in the cross-sectional
studies is the manner in which they have taken account of confounding
factors in deriving adjusted estimates of associations between body
lead burden and neuropsychological performance, in particular IQ.
The three aspects of the problem are:
i) Which potential confounders are measured? There is considerable
variation between studies here, suggesting that no individual study
can have fully corrected for the full range of parental and social
influences on IQ.
ii) How were confounders selected for inclusion in the analysis? Some
studies include confounders solely on the basis of their strength of
association with outcome (IQ), while others have given attention to
their association with body lead burden. It is difficult to assess the
extent to which these differences would affect the results, but the
former is more generally accepted statistical practice.
iii) Which statistical analysis strategy was employed in reaching a
final model? Most studies have adopted some form of multiple
regression technique, but have varied as to whether forward selection
(i.e. model building), backwards selection (i.e. model collapsing) or
more arbitrary choices of final model were employed. In the larger
studies, the choice between such strategies is unlikely to matter, but
in smaller studies the greater play of random variation could make the
results dependent on the statistical technique.
b) What they told us
In most studies a negative association between lead measures and
IQ measures is found in uncontrolled data. This difference is usually
in the range of 4 to 6 IQ points. Most studies also confirmed the
positive association between lead measures and indicators of social
disadvantage, whether this is indicated by SES, maternal education or
other more detailed indicators of non-optimal child-rearing
environments, such as marital quality or maternal depression. When
these social and other confounding factors are controlled, the effect
has been, in most cases, to reduce the strength of the association
between lead measures and IQ, although it remains in the same
direction. Where maternal intelligence has not been controlled, the
impact of correction for covariates tends to be smaller, and where
more detailed social measures have been controlled, the impact has
been greater.
c) What they did not tell us
The cross-sectional nature of these studies, and the fact that
many used a single measure of current exposure, limited their
usefulness in answering questions relating to the natural history of
the association between lead exposure and outcomes, including whether
there were critical periods of exposure, and whether lead-associated
deficits were persistent or reversible. Equally they were limited in
the information that could be obtained relating to the natural history
of confounders. They were also unable to answer questions of reverse
causality.
8.3.6.5 An interpretation of the overview of prospective and
cross-sectional studies
The above meta-analyses of the prospective studies and cross-
sectional studies reveal a consistency between studies which points
towards a "collectively significant" inverse association between PbB
level and full-scale IQ. Taking the results in Fig. 15 as a guideline,
there appears to be a mean decrease in full-scale IQ of the order of 2
IQ points for a change in mean PbB level from 0.48 to 0.96 µmol/litre
(10 to 20 µg/dl).
Below this range, uncertainties are increased, concerning firstly
the existence of an association and secondly estimates of the
magnitude of any putative association. The relatively limited numbers
of children in most studies with PbB levels below this range, the
strong contributions of confounding variables, and limitations in the
precision in analytical and psychometric measurements combine to lower
statistical power to detect associations and to estimate their
magnitude.
The key question is whether this statistical association is
directly attributable to the causal influence of lead on child IQ. It
is important to consider alternative explanations as follows:
a) Chance The consistency of alternative analyses and the level of
significance achieved suggest that chance cannot be a complete
explanation. However, the confidence intervals are relatively wide, so
that the magnitude of true association could be as low as a < 1-point
(rather than a 2-point) deficit, or as high as > 3 points.
b) Confounding factors The adjusted lead/IQ associations tend to be
substantially weaker than the unadjusted associations, which indicates
that confounding factors are important. Since none of the studies can
claim to have taken complete account of confounders, (e.g., father's
IQ is not included), it seems likely that some of the remaining
lead/IQ relationship, after adjustment, may still be attributable to a
degree of unexplained confounding.
c) Reverse causality One initial justification for the prospective
studies was that they could measure early exposure to lead and relate
it to later child development, thus removing the problem of reverse
causality potentially present in the cross-sectional studies. However,
it has turned out that very early lead exposure, e.g., in the
perinatal period, is not related to school-age IQ. The question arises
as to whether children of lower IQ could exhibit behaviour patterns at
earlier ages, e.g., around age 2 and older, which could enhance their
uptake of lead. Perhaps, such reverse causality remains a possibility
even in the prospective studies.
d) Selection biases The positive studies may perhaps be more likely
to report in more quantitative details, as indicated by the absence of
relevant data from Cleveland in the prospective studies. Post-hoc
selection based on the more significant PbB concen-trations may lead
to exaggerated estimates.
It is a matter of debate and conjecture as to the extent to which
these four issues should inhibit claims of a causal relationship in
the prospective studies. The essential problem is that observational
epidemiology cannot provide definitive evidence of causality when the
key statistical association is weak, the temporal relationship is
unclear and major confounders are present.
8.3.7 Hearing impairment in children
Schwartz & Otto (1987) reported that the probability of elevated
pure-tone hearing thresholds at 500, 2000 and 4000 Hz increased
significantly with increasing PbB level in 4519 subjects (4-19 years
of age) who participated in the NHANES II study. Variables included in
the backwards stepwise regression models included information derived
from medical history, clinical examinations, and assessment of
developmental milestones.
Schwartz & Otto (1991) examined data for subjects (6-19 years of
age) from the Hispanic Health and Nutrition Examination Survey
(HHANES) study which included three distinct ethnic groups. After
excluding 283 subjects with previous ear problems (discharges,
ruptured ear drums or tinnitus), data for 3262 were analysed. The
authors concluded that increasing PbB level in the range
0.36-0.86 µmol/litre (7-18 µg/dl) was associated with approximately
2 dB loss of pure-tone hearing at frequencies of 500, 1000, 2000 and
4000 Hz.
Dietrich et al. (1992) assessed the relationship between scores
on a test of sensory auditory processing (SCAN) and prenatal/postnatal
PbB concentrations in 215 subjects drawn from the Cincinnati
prospective cohort study. Higher prenatal, neonatal and postnatal PbB
concentrations were associated with more incorrect identification of
common monosyllabic words presented under conditions of muffling.
Other variables associated with impaired central auditory processing
were pure-tone audiometry results, social class, quality of caretaking
in the home, birth weight, gestational age, a measure of obstetrical
complications, and consumption of alcohol during pregnancy. Following
adjustment for these co-factors, lifetime average PbB concentration
remained significantly and inversely associated with SCAN performance.
8.4 Renal system
Acute exposure to lead is known to cause proximal renal tubular
damage, characterized by generalized aminoaciduria, hypophosphataemia
with relative hyperphosphaturia, and glycosuria (Chisolm, 1962).
Cellular structural changes include nuclear inclusion bodies,
mitochondrial changes and cytomegaly of the proximal tubular
epithelial cells (Cramer et al., 1974). Diagnosis of lead-induced
altered renal function or disease is difficult since there are no
specific indicators; blood urea nitrogen (BUN) and creatinine levels
become elevated only when twothirds of renal function has been lost
(Bernard & Becker, 1988).
8.4.1 Clinical studies
From a study of seven men occupationally exposed to lead in a
shipyard during oxy-acetylene flame cutting of lead-painted steel
hulls, Cramer et al. (1974) concluded that there is a continuum of
morphological and functional change in the pathogenesis of chronic
lead nephrotoxicity. Each subject was treated in hospital 3 or more
days after recent exposure and PbB levels exceeded 3.36 µmol/litre
(70 µg/dl) in all cases. On the basis of microscopic examination of
biopsy tissue, nuclear inclusion bodies were reported in proximal
tubular cells during the early phase but there was no impairment of
renal function. In a second phase, there was fibrosis associated with
asymptomatic azotaemia and reduced glomerular filtration rate but
without demonstrable proximal tubular dysfunction; renal failure was
not seen in the study.
Weeden et al. (1979) diagnosed lead nephropathy in 15 lead
workers, all having reduced glomerular filtration rates. Renal
biopsies of six of the subjects showed focal interstitial nephritis in
addition to non-specific changes in the proximal tubules. At the time
of examination, PbB levels for 11 of the 15 workers were within the
range 1.92-3.84 µmol/litre (40-80 µg/dl).
Baker et al. (1979) reported increased BUN and decreased
creatinine clearance in 28 workers, all of whom had relatively
prolonged, high-dose lead exposure in lead smelting or chemical
manufacturing.
A study by Maranelli & Apostoli (1987) of 60 workers, described
as "lead poisoned" and having PbB levels of 3.45 ± 0.80 µmol/litre
(71.9 ± 16.6 µg/dl), found no definitive correlation between PbB, lead
in urine after chelation, and BUN, serum creatinine and serum uric
acid.
8.4.2 Epidemiological studies
8.4.2.1 Occupational cohorts
Several recent studies of lead-exposed workers provide further
information relating to dose-effect relationships. The emphasis in
these studies was on lead and its possible association with adverse
effects on health. However, exposure to other potentially toxic
substances in the working and living environment should not be
overlooked/ignored.
For example, Buchet et al. (1980) examined 25 male lead smelter
workers and 88 male control workers. The PbB levels of the lead
workers were in the range 1.62-2.94 µmol/litre (33.8-61.3 µg/dl) for a
mean of 13.2 years (range 3.1-29.8 years) of lead exposure. The PbB
levels for the controls were in the range 0.26-1.64 µmol/litre
(5.5-34.2 µg/dl). There were no differences for parameters of renal
function between the groups and no signs of clinical renal impairment.
It was concluded that a PbB level of less than 2.98 µmol/litre
(62 µg/dl) is not associated with renal toxicity.
N-acetyl-ß-d-glucosaminidase (NAG) is a lysosomal enzyme present
in renal tubular cells. This enzyme is a sensitive but non-specific
indicator for early sub-clinical nephrotoxicity. In a study on 29
lead-exposed workers, Meyer et al. (1984) found increased NAG in
urine, but there was no correlation with PbB level. However, NAG level
was found to be normal in the 5 subjects with PbB levels exceeded
3.36 µmol/litre. These authors speculated that long-term high exposure
to lead may deplete the kidneys of NAG or render it insensitive to the
effects of lead exposure.
Verschoor et al. (1987) studied 155 male lead workers and 126
control workers who were matched for age, smoking habits,
socioeconomic status and duration of employment. The PbB levels of the
lead workers were in the range of 0.43-4.71 µmol/litre
(8.3-97.6 µg/dl) compared with the controls 0.15-0.96 µmol/litre
(3.1-18.8 µg/dl). The lead workers had elevated ZPP levels
(34-292 µmol/mol haemoglobin) compared with controls (10-35 µmol/mol
haemoglobin). No significant differences were found for various
indicators of renal function; all urinary and serum parameters were
within normal ranges. There were no differences in protein excretion
patterns and no signs of renal impairment. However, the authors found
that NAG levels in the lead-exposed workers were higher than control
values and increased with increasing PbB levels. They concluded that
lead exposure resulting in PbB levels of under 3.0 µmol/litre
(62 µg/dl) can affect renal tubular functions as measured by NAG
excretion; lead appeared to affect the tubular parameters more than
the glomerular parameters in moderately exposed workers.
Ong et al. (1987) examined 158 male and 51 female lead battery or
smelter workers and 30 control workers. The lead workers had 1-36
years exposure with an average of 10.8 ± 8.0 years and PbB levels in
the range of 0.14-3.84 µmol/litre (3-80 µg/dl); only five workers
exceed 2.88 µmol/litre (60 µg/dl). The authors found a weak but
statistically significant positive association between PbB and blood
urea nitrogen, and between PbB and serum creatinine, and that
creatinine clearance was reduced with increasing PbB level. NAG levels
in the lead-exposed workers were significantly higher than control
values and increased with increasing urine lead level when the data
were adjusted for age. These authors concluded that a relatively low
PbB level can affect renal function.
Not all investigators have found parallel association between PbB
and NAG levels. Gennart et al. (1992b) compared 98 lead-exposed lead
acid battery workers (mean PbB level 2.45 µmol/litre, 51 µg/dl,
geometric mean ZPP 10.2 µg/g Hb) with 85 controls (mean PbB level
1.00 µmol/litre, 20.9 µg/dl, geometric mean ZPP 2.84 µg/g Hb)
recruited from other departments in the same factory. None of the
indicators of renal function (retinol-binding protein,
ß2-microglobulin, albumin or NAG in urine, or creatinine or
ß2-microglobulin in serum) were correlated with PbB level, duration
of exposure or ZPP, or showed significantly different mean values
between the lead-exposed and control groups of workers.
Cardenas et al. (1993) recently evaluated 27 different
indications of renal dysfunction in 50 workers exposed to lead and 50
controls. There were significant increases in urinary NAG and sialic
acid in the lead exposed group. These changes may represent minor
cellular modifications rather than significant functional or
irreversible renal damage. There was a significant decrease in urinary
6-keto PGF1alpha and a significant increase in TXB2. These
eicosanoid changes may reflect systemic functional vascular changes
rather than an effect of lead on the kidney.
8.4.2.2 General population
An epidemiological survey on 283 persons in Scotland, from
households with water lead concentrations in excess of 100 µg/litre,
revealed a close correlation between water lead content and PbB and
serum urea concentrations (Campbell et al., 1977). The frequency of
renal dysfunction in individuals with elevated PbB concentrations
(> 2 µmol/litre or > 41 µg/dl) was significantly greater than that
of age- and sex-matched controls.
Pocock et al. (1984) measured serum creatinine, urate and urea
concentrations in 7364 British men, and 74 subjects had PbB levels
equal to or greater than 1.8 µmol/litre (37.3 µg/dl). The authors
concluded that there was no indication that exposure to lead at
concentrations commonly encountered in Britain was responsible for
impaired renal function.
Staessen et al. (1992) investigated the relationship between lead
exposure and renal function as part of a cross-sectional population
study of the health effects of environmental exposure to cadmium.
Creatinine clearance, PbB and ZPP were measured in a random population
sample of 965 men (geometric mean PbB level was 0.55 µmol/litre or
11.4 µg/dl) and 1016 women (geometric mean PbB level was
0.36 µmol/litre or 7.5 µg/dl). Creatinine clearance rate (mean was
99 ml/min in men, 80 ml/min in women) was inversely correlated with
PbB and ZPP levels before and after adjustment for age, body mass
index and diuretic treatment. Also positively correlated were serum
ß2-microglobulin and PbB level in men, serum ß2-microglobulin and ZPP
in men and women, and serum creatinine and ZPP in men. Impaired renal
function could not be explained by exposure to cadmium or by elevated
blood pressure. The authors concluded that exposure to lead may impair
renal function in the general population. However, the study could not
exclude the possibility that renal impairment may lead to an increase
in PbB level.
8.4.2.3 Cohort mortality studies
"Lead poisoning" was a diagnosis given to 241 workers employed
for 1-30 years at the Port Pirie lead smelter between 1928-1959 by a
State medical board. Death registration records for the period
1930-1977 identified 140 deaths among the group. The cause of death
profile was compared with that of 695 other male decedents,
predominantly production workers and a smaller number of office
workers. Age-standardized mortality analysis revealed a substantial
excess of deaths attributed to chronic nephritis and to cerebral
haemorrhage. The rates for lead-poisoned workers exceeded those for
the other workers, and both exceeded the rates for the Australian
general male population. The rates decreased over successive calendar
periods but excess rates persisted for chronic nephritis up until 1977
(McMichael & Johnson, 1982).
8.5 Cardiovascular system
Two persistent issues have been under intense study since
publication of Environmental Health Criteria 3: Lead (IPCS, 1977):
a) whether lead is a factor in hypertension and, if so, whether
there is a causal relationship;
b) whether lead is contributory to cardiovascular effects
influencing morbidity or mortality.
8.5.1 Blood pressure
8.5.1.1 Studies on occupationally exposed cohorts
From a study of 431 white male police officers 24-55 years of age
(Moreau et al., 1982; Orssaud et al., 1985), it was concluded that
systolic and diastolic blood pressure was related to PbB level, the
correlation being greatest for the younger subjects and decreasing
with age. Statistical adjustment was made for alcohol consumption and
body mass index, but not for smoking. The magnitude of observed
association between systolic pressure and PbB level in this small
study was somewhat greater than in the larger general population
studies cited below.
Parkinson et al. (1987) studied 270 lead battery workers and 158
non-exposed workers and compared lead exposure with systolic and
diastolic blood pressures. After controlling for age, education,
income, cigarette smoking, alcohol consumption and exercise, there was
a small and non-significant association. The average PbB level of
lead-exposed workers was 1.92 ± 0.62 µmol/litre (40 ± 13 µg/dl),
whereas in non-exposed workers it was 0.34 ± 0.24 µmol/litre
(7 ± 5 µg/dl).
8.5.1.2 Studies in the general population
The possible relationship between PbB concentration and blood
pressure has been examined in several large-scale population studies.
These include the British Regional Heart Study (BRHS), the US NHANES
II (National Health and Nutrition Examination Survey) and studies in
Wales, Denmark, Canada and Belgium.
The BRHS is a prospective study of 7735 men, initially aged 40-59
years, from 24 British towns, who were first examined in 1978-1980
(Shaper et al., 1981). The median PbB level was 0.7 µmol/litre
(14.5 µg/dl). The initial findings (Pocock et al., 1984), which
indicated a lack of association between PbB level and blood pressure,
were later re-examined to take account of potential confounding
factors such as alcohol, smoking and town of residence. Pocock et al.
(1988) noted that 95% confidence intervals for systolic and diastolic
blood pressure plotted against PbB concentrations overlapped and
showed no elevation even at the highest observed PbB concentration.
Applying statistical techniques to adjust mean systolic and diastolic
blood pressure measurements for body mass index, age, alcohol
consumption, cigarette smoking and town of residence showed signs of a
weak association, particularly for diastolic pressure and PbB level.
Multiple regression analysis showed that adjustment for personal
characteristics but not for town of residence rendered both systolic
and diastolic regressions on log PbB insignificant, chiefly because
alcohol consumption is an important confounder, being positively
related to both blood pressure and PbB level. Introducing an extra
adjustment for town of residence made the systolic and diastolic
regressions highly statistically significant, even though the
associations were weak. These cross-sectional data indicated that for
every doubling in PbB level (e.g., from 0.8 to 1.6 µmol/litre) there
are estimated mean increases of 1.45 mmHg (95% confidence interval
0.47-2.43 mmHg) in systolic blood pressure and 1.25 mmHg (95%
confidence interval 0.65-1.85 mmHg) in diastolic blood pressure.
The NHANES II study, a USA national cross-sectional survey
carried out in 1976-1980, included PbB and blood pressure measurements
in a general population sample of 5803 men and women aged 12-74.
Geometric mean PbB levels were around 0.72 µmol/litre (about 15 µg/dl)
for men and approximately 0.53 µmol/litre (11 µg/dl) for women.
Several authors have examined these data for possible PbB and blood
pressure associations.
In an analysis controlling for the confounding factors of age,
race and body mass index, Harlan et al. (1985) and Harlan (1988) found
a significant association between PbB level and blood pressure for men
but not for women. White males aged 40-59 years from the same data set
were analysed by Pirkle et al. (1985), and the correlation between PbB
level and blood pressure was confirmed. In further analysis of the
whole age range, adjustments for possible time trend and geographical
site effects did not affect the significance of the association in
males (Landis & Flegal, 1988; Schwartz, 1988), although its magnitude
did become somewhat less pronounced.
Gartside (1988) applied a method of forward stepwise regression
to PbB and blood pressure data from NHANES II and reported that the
results for white men, white women and black men were contradictory
and lacked consistency and reliability. He noted that the overall
average was too small to conclude association between PbB level and
blood pressure in this study.
Two surveys were carried out in Wales by Elwood et al. (1988a,b).
The Welsh Heart Programme carried out in 1985 provided complete PbB,
blood pressure and other data for 865 men and 856 women aged 18-64.
The geometric mean PbB level for men was 0.56 µmol/litre (11.6 µg/dl)
and for women was 0.43 µmol/litre (9.0 µg/dl). In neither sex was
there a significant correlation between PbB level and systolic or
diastolic blood pressure. The second study was in a cohort of men aged
49-65 years living in Caerphilly, Wales. The geometric mean PbB level
(N = 1137) was 0.61 µmol/litre (12.7 µg/dl). Complete data from 1137
subjects, ranking blood pressure readings according to PbB level, did
not reveal any trend in the percentage of subjects with systolic
pressure above 160 mmHg. The authors corrected only for age as a
confounding factor.
Data from a Canadian study (Neri et al., 1988) collected during
10 months in 1978-1979 for 2193 subjects aged 25-64 showed a weak but
statistically significant association between PbB level and diastolic
blood pressure.
Grandjean et al. (1989b) studied 504 men and 548 women residing
in Glostrup (Denmark) at age 40; 451 men and 410 of the women were
followed-up five years later. Average PbB levels for the men at 40 and
45 years of age were 0.62 µmol/litre (13 µg/dl) and 0.43 µmol/litre
(9 µg/dl), and for the women 0.43 µmol/litre (9 µg/dl) and
0.29 µmol/litre (6 µg/dl), respectively. All correlations found
between PbB and blood pressure became insignificant when blood
haemoglobin and alcohol intake were entered into multiple regression
analyses; an independent effect of low-level lead exposure on blood
pressure could not be distinguished.
Möller & Kristensen (1992) examined 1052 men and women in
Copenhagen, Denmark, in 1976, and re-examined them in 1981 and 1987
(men only). Initial mean PbB levels were 0.65 µmol/litre (13.6 µg/dl)
in men and 0.46 µmol/litre (9.6 µg/dl) in women, and had fallen by
around 30% 5 years later. For men, there was no significant
association between PbB level and blood pressure (or between changes
in PbB level and blood pressure) after adjustment for confounders
(tobacco, alcohol, body mass and physical activity). For women, the
association between PbB level and diastolic (but not systolic) blood
pressure remained significant on both occasions, even after
controlling for confounders.
A Belgium study (Dolenc et al., 1993) included 827 men and 821
women (mean age 25) whose mean PbB levels were 0.56 and
0.34 µmol/litre (11.6 and 7.07 µg/dl), respectively. After adjustment
for covariates (body mass index), pulse rate and serum creatinine and
serum calcium levels), systolic blood pressure was inversely
associated with PbB level in men (P < 0.05). There were no
significant PbB associations for diastolic blood pressure in men or
for either pressure in women.
A study of 398 male and 133 female civil servants (Staessen et
al., 1990) showed geometric mean PbB levels of 0.58 µmol/litre
(12.06 µg/dl) and 0.46 µmol/litre (9.56 µg/dl) in men and women,
respectively. Taking the data for both sexes together, there were
statistically significant positive associations between PbB level and
systolic and diastolic blood pressures (r = +0.11 in each case) but
these became non-significant after adjustment for confounders (sex,
age, BMI, pulse, delta-glutamyltranspeptidase (delta-GTP) and serum
calcium level).
Another cross-sectional study involving only women was performed
in Boston (Rabinowitz et al., 1987). Cord PbB levels (mean
0.33 µmol/litre, 6.9 µg/dl) among 3851 women correlated with both the
systolic (r = 0.08) and diastolic (r = 0.05) blood pressures measured
during and before delivery. Multivariate models of pregnancy
hypertension as a function of age, parity, haematocrit, diabetes,
ponderal index, and race were improved when lead was included as a
predictor. Lead appeared to have a small but demonstrable association
with pregnancy hypertension and blood pressure at delivery, but not
with eclampsia.
In an overview of the BRHS, NHANES II and Welsh studies, Pocock
et al. (1988) concluded that with overlapping confidence limits the
data provided weak but reasonably consistent evidence of lead and
blood pressure associations. They noted that the NHANES II data on
2254 men in the USA indicate a slightly stronger association between
PbB level and systolic blood pressure, whereas data from over 2000 men
in Wales did not show a statistically significant association. They
inferred that a causal relationship could not be concluded from any of
the epidemiological studies.
Staessen et al. (1994) have undertaken a more extensive
meta-analysis of nearly all of the above studies and some other
smaller ones as well. This amounted to a total of 19 studies with
28 210 subjects. They found that the association between PbB level and
blood pressure was similar in both sexes. In all studies combined, a
two-fold increase in PbB concentration was associated with a 1.0 mmHg
increase in systolic pressure (95% confidence interval (CI); 0.3 to
1.7 mmHg; P = 0.008) and a 0.7 mmHg increase in diastolic pressure
(95% CI: 0.2 to 1.3 mmHg; P = 0.02). These authors conclude that the
published evidence suggests a weak positive association between blood
pressure and lead exposure, but any such relationship may not be
causal and is unlikely to entail any public health implications
regarding hypertension.
8.5.2 Other cardiovascular effects
As recorded previously (IPCS, 1977), there is good evidence that
signs of clinical lead poisoning sometimes include evidence of toxic
action on the heart. Kopp et al. (1988) reviewed the cardiovascular
actions of lead and concluded that the degree of cardiovascular
involvement during episodes of acute lead intoxication depends on the
duration of exposure and dose. It is not known whether environmental
exposure to lead affects the electrical or mechanical activity of the
heart.
8.5.2.1 Occupational studies
There have been several studies relating to cardiovascular
effects in lead-exposed occupational groups. However, as with any
study of an occupational cohort, it is important to evaluate the
strength of the cause-effect relationship, the nature or substance of
other contributory factors and how well the workplace "exposure" has
been evaluated for other potential toxicants. Thus, the occupational
hazards associated with work in lead smelting or lead battery
operations include, but are not limited to, lead.
8.5.2.2 Studies in the general population
Pocock et al. (1988) found that after 6 years of follow-up of the
BRHS cohort of 7735 middle-aged men, 316 of the men had major
ischaemic heart disease and 66 had had a stroke. After allowance for
confounding effects of cigarette smoking and town of residence, there
was no evidence that PbB was a risk factor for these cardiovascular
events.
Möller & Kristensen (1992) studied 1052 men and women in
Copenhagen, Denmark, and related blood pressure to subsequent
cardiovascular morbidity and mortality over a 14-year period. There
were significant positive associations between PbB level and both
coronary and cardiovascular disease in univariate analysis, but these
became non-significant after controlling for confounders.
a) Clinical studies
Boscolo & Carmignani (1988) measured plasma renin activity in
hospitalized lead-exposed workers and concluded that synthesis or
release of renin is increased by short or moderate exposure to lead
and decreased with prolonged exposure.
b) Epidemiological studies
Kirkby & Gyntelberg (1985) found a significantly higher incidence
of ischaemic ECG changes in a study of lead smelter workers (20%, mean
PbB level 2.45 µmol/litre, 51 µg/dl), compared with matched
non-exposed controls (6%, mean PbB level 0.53 µmol/litre, 11 µg/dl).
There was also a slight (4-5 mmHg) increase in diastolic blood
pressure in the lead workers compared with the controls.
c) Cohort mortality studies
Two studies (Cooper, 1988 and Fanning, 1988) reported an
increased mortality rate of lead workers due to circulatory disease,
but no such relationship was found in reports of studies by
Gerhardsson et al. (1986) or Selevan et al. (1988). Gerhardsson et al.
(1986) examined a lead-exposed sub-cohort of 437 from a study cohort
of 3832 male workers first employed at a copper smelter before 1967
and followed up from 1950 to 1981. There was no excess mortality due
to ischaemic heart disease or cerebrovascular disease in the
lead-exposed worker sub-cohort. Selevan et al. (1988) found no
association between lead exposure and deaths due to hypertensive
diseases in a review of mortality in a cohort of male hourly workers
at a lead smelter. There were many confounding factors in each of
these studies.
8.5.3 Summary
Despite intensive efforts to define the relationship between body
burden of lead and blood pressure or other effects on the
cardiovascular system, no causal relationship has been demonstrated in
humans and the mechanisms remain obscure.
There is experimental evidence from animal studies indicative of
an effect of lead on blood pressure, and several mechanisms have been
proposed to explain these observations (see section 7.4).
8.6 Gastrointestinal effects
8.6.1 Occupational exposure
Colic is a well-recognized symptom of acute lead poisoning and is
still reported in groups of lead-exposed industrial workers. Of
particular concern are the reports of intoxication caused by acute
exposure (inadequate protective measures) to lead associated with
removal of lead-based paint by burning or sand-blasting and the
demolition of lead-containing industrial plants. Symptoms of colic
include abdominal pain, constipation, cramps, nausea, vomiting,
anorexia, weight loss and decreased appetite.
Symptoms in adults typically occur at PbB levels of
4.8-9.6 µmol/litre (100-200 µg/dl) but have been noted at levels as
low as 1.92 µmol/litre (40 µg/dl) (Baker et al., 1979; Haenninen et
al., 1979; Awad El Karim et al., 1986; Pollock & Ibels, 1986; Muijser
et al., 1987; Holness & Nethercott, 1988; Marino et al., 1989;
Pagliuca et al., 1990; Schneitzer et al., 1990).
Positive histories of lead colic were given by 40 workers in a
cohort of 158 secondary lead smelter workers who participated in a
clinical field survey (Lilis et al., 1977). It was reported most
frequently by those having PbB levels above 3.84 µmol/litre (80 µg/dl)
at the time of examination and was not found where ZPP was within the
normal range. Thirty percent of workers having ZPP > 200 µg/g Hb had
experienced symptoms.
In a study of a population of 585 black South African factory
workers, Irwig et al. (1978) reported that the incidence of abdominal
pain increased with increasing PbB level: it was 12% for the first
quartile (PbB < 3.28 µmol/litre or < 68 µg/dl), 23% for the
second (3.28-4.14 µmol/litre or 68-86 µg/dl), 24% for the third
(4.15-5.14 µmol/litre or 86-107 µg/dl) and 37% for the fourth
(> 5.15 µmol/litre or > 107 µg/dl).
In a cross-sectional clinical study of 90 telephone
cable-splicers by Fischbein et al. (1980), 19 (21%) workers reported
gastrointestinal symptoms (mean PbB level of 1.44 µmol/litre, or
30 µg/dl and mean ZPP of 66.6 µg/g Hb) whereas 71 (79%) reported no
such symptoms (mean PbB level of 1.3 µmol/litre or 27 µg/dl and mean
ZPP of 52.3 µg/g Hb).
8.6.2 Exposure of children
Colic is seen in children and US EPA (1986a) concluded that the
lowest-observed-adverse-effect level was in the range of
2.88-4.80 µmol/litre (60-100 µg/dl).
8.7 Liver
There appears to be no new evidence relating human body lead
burden to effects on the liver, but it has been suggested that the
effects of lead on haem synthesis may alter the functional capacity of
the hepatic cytochrome P-450 system to metabolize drugs.
8.7.1 Occupational exposure
Fischbein et al. (1977) tested five demolition workers acutely
exposed to lead for a period of 3 months prior to the study. They
found that the plasma half-life of an oral dose of antipyrine was
within the range for normal healthy volunteers; it was shorter in each
subject after chelation therapy but still within the normal range. The
plasma half-lives of phenylbutazone were also within the normal range
but were unaffected by chelation therapy.
8.7.2 Exposure of children
Saenger et al. (1984) found decreased urinary excretion of
6-ß-hydroxycortisol in 26 children with a mean PbB level of
2.11 µmol/litre (44 µg/dl); the decreased formation of the metabolite
was attributed to lead inhibition of the cytochrome P-450-dependent
mixed-function oxidases.
8.8 Reproduction
While it is generally accepted from early literature that lead
adversely affects the reproductive process in both men and women, the
evidence is mostly qualitative and dose-effect relationships have not
been established.
Most new information relates to reports of occupational cohorts
and of populations living in polluted areas near industrial plants.
There is qualitative evidence that lead is toxic to the reproduction
system in both men and women. However, there are insufficient data to
provide the basis for estimation of dose-effect relationships in
women.
8.8.1 Female populations
Nordstrom et al. (1978b) reported an increased frequency of
spontaneous abortion in women living close to a smelter in northern
Sweden. In a later report, Nordstrom et al. (1979) described the
responses to a questionnaire completed by 511 of 662 women who had
worked at the smelter and were born between 1930 and 1959. Spontaneous
abortion rates were highest in those pregnancies in which the mother
was employed during the pregnancy (13.9%) or had been employed before
the pregnancy and was living close to the smelter (17%); the frequency
rate was higher (19.4%) when the father worked at the smelter.
However, it should be noted that the smelter produced copper and lead
in addition to a number of other metallurgical and chemical products
(Nordstrom et al., 1978a) and that the effects reported may not
necessarily be attributable exclusively to lead.
A study of pregnancies in the centre and surrounding areas of the
lead smelter town of Port Pirie showed that the incidence of
miscarriages (22 or 23) and stillbirths (10 or 11) was higher in women
living close to the smelter (McMichael et al., 1986). However, in a
study of 639 Yugoslav women, the risk of spontaneous abortion was not
increased (OR = 1.1, 95% CI: 0.9-1.4) among women with mean PbB levels
of 0.77 µmol/litre (16.0 µg/dl) as compared with women with mean PbB
levels of 0.25 µmol/litre (5.2 µg/dl) (Murphy et al., 1990). Risk did
not vary with distance of residence from the smelter.
Some studies have found decreased length of gestation in women
whose PbB levels were greater than 1.09 µmol/litre (23 µg/dl) (Moore
et al., 1982), 0.58 µmol/litre (12 µg/dl) (Dietrich et al., 1986) or
0.72 µmol/litre (15 µg/dl) (McMichael et al., 1986). However, neither
Bellinger et al. (1991b) nor Graziano et al. (1990) found decreases in
gestational length or other parameters of pregnancy in women with
elevated PbB levels.
In the Cincinnati prospective study, a significant reduction in
birth weight associated with prenatal (maternal) PbB levels, after
adjustment for covariates, was reported by Bornschein et al. (1989).
In the Port Pirie prospective study of 749 pregnancies (McMichael et
al., 1986), the proportion of pregnancies resulting in low birth
weight singleton infants was more than twice as high in Port Pirie
women (whose PbB levels averaged 0.50 µmol/litre, 10.4 µg/dl) than in
women outside Port Pirie (average PbB level of 0.264 µmol/litre,
5.5 µg/dl). On the other hand, multiple regression analyses showed no
significant association between low birth weight and maternal PbB
level. In a cross-sectional study, Ward et al. (1987) reported a
significant simple relationship between placental lead concentrations
and reduced birth weight and head circumference.
Other studies have not shown a significant association between
birth weight and lead exposure. The Kosovo prospective study failed to
detect any evidence of lead-related birth weight reduction in more
than 900 births (Murphy et al., 1990), and Ernhart et al. (1986) found
no significant effect of lead on birth weight, birth length or head
circumference.
8.8.2 Male populations
Reproductive effects from occupational exposure to lead include
asthenospermia, hypospermia, teratospermia and hypogonadism (US EPA,
1986a; Braunstein et al., 1987).
In men, effects on sperm or the testes may result from chronic
exposure to lead at blood levels of the order of 1.92-2.4 µmol/litre
(40-50 µg/dl) (ATSDR, 1991).
Wildt et al. (1983) compared two groups of lead-exposed storage
battery workers and concluded that the highly exposed group (PbB of
more than 2.16 µmol/litre, 45 µg/dl) had decreased prostate/seminal
vesicle function, low semen volume and poorer functional maturity of
sperm.
Chowdhury et al. (1986) reported a significant decrease in sperm
count and motility and an increased count of abnormal spermatozoa in
lead-exposed men (average PbB level of 2.04 µmol per litre or
42.5 µg/dl) compared with controls (0.71 µmol/litre or 14.8 µg/dl).
Assenato et al. (1987) also reported decreased sperm production in 18
battery factory workers having PbB levels of 2.4-2.93 µmol/litre
(50-61 µg/dl).
In five out of six symptomatic lead intoxicated workers having
PbB levels in the range of 1.87-4.7 µmol/litre (39-98 µg/dl) at the
time of examination, Cullen et al. (1984) found defects of
spermatogenesis, including oligospermia and azoospermia.
The results of Lerda (1992) also showed significant increases in
asthenospermia and teratospermia among 38 battery workers with PbB
levels of 1.95-4.7 µmol/litre (40.5-98.0 µg/dl) compared to a control
group with PbB levels of 0.86-1.25 µmol/litre (17.6-26.0 µg/dl).
An epidemiological study by Lindholm et al. (1991) of 213 wives
of lead workers and 300 matched controls suggested an association
between paternal lead exposure and the risk of spontaneous abortion
among the wives of lead workers when the PbB level exceeded
1.5 µmol/litre (31 µg/dl) close to the time of spermatogenesis.
However, the authors acknowledged that exposure of the study subjects
to other metals and organic solvents may have influenced the results
of the study.
A weak association between paternal exposure, estimated from
occupational history, and risk of esotropia (inward deviation
strabismas) was noted in a case control study of 377 children aged 7
years (OR - 2.4, 95%, CI: 1.0-5.6) (Hakim et al., 1991). However, the
increased risk was not dose-related, and the risk of exotropia
(outward deviation) was not associated with higher paternal lead
exposure.
8.8.3 Hormonal responses
Assenato et al. (1987) found no significant differences in FSH,
testosterone, prolactin, LH and total neutral 17-ketosteroids levels
between 18 lead battery workers (PbB level, 2.4-3 µmol/litre or
50-61 µg/dl) and 18 cement workers (PbB level, 0.9-1.1 µmol/litre or
18-22 µg/dl), despite the finding of oligospermia in the lead-exposed
group.
Rodamilans et al. (1988) studied the endocrine status of 23 lead
smelters in relation to the duration of lead exposure. Five workers
exposed for less than 1 year (mean PbB level, 0.29 µmol/litre or
66 µg/dl) showed an increase in serum LH level, while that of
testosterone remained normal. Eight workers exposed for 1-5 years
(mean PbB level, 3.50 µmol/litre or 73 µg/dl) and 10 employed for more
than 5 years (mean PbB level, 3.65 µmol/litre or 76 µg/dl) showed an
increase in LH comparable with the group exposed for less than 1 year
but there was a clear reduction in serum testosterone levels.
Gustafson et al. (1989) found a lower plasma FSH level in 25
moderately exposed workers (mean PbB level, 1.82 µmol/litre or
38 µg/dl) than in 25 matched controls. A lower mean serum LH level was
reported by McGregor & Mason (1990) in 90 lead workers (mean PbB
level, 2.20 µmol/litre or 46 µg/dl) than in controls; there was also a
correlation between serum FSH and PbB levels. In an epidemiological
study involving 122 current lead workers (PbB mean level,
1.69 µmol/litre or 35.1 µg/dl) and 49 non-exposed workers (PbB mean
level, 0.4 µmol/litre or 8.3 µg/dl), Ng et al. (1991) found slightly
higher plasma LH and FSH levels among the exposed workers. The
testosterone level, however, was not significantly different between
the two groups. In contrast, Gennart et al. (1992) did not find an
effect of lead on various endocrine parameters (including FSH and LH
levels) in a study population of 221 workers having a geometric mean
PbB level of 2.45 µmol/litre (51.0 µg/dl). They suggested that the
hypothalamic-pituitary system may not be influenced by moderate
exposure to lead.
8.8.4 Postnatal growth and stature
Several reports have suggested that the physical growth and
stature of children may be reduced by exposure to lead (e.g., Mooty et
al., 1975; Johnson & Tenuta, 1979; Routh et al., 1979), but the
influence of other factors (e.g., race and diet) has often made it
difficult to isolate lead as a causal agent for such effects in human
populations. Multivariate regression analyses of NHANES data for
approximately 2700 children in the USA (Schwartz et al., 1986)
provided more convincing evidence of a significant association between
increasing PbB levels and reduced height, weight, and chest
circumference after adjusting for age, race, sex and nutritional
covariates.
8.9 Effects on chromosomes
As noted in Environmental Health Criteria 3: Lead (IPCS, 1977),
the literature remains controversial concerning induction of
chromosomal changes in human lymphocytes by in vivo exposure to
lead. Most of the studies are of small numbers of subjects in
occupational groups where lead was one of many potentially toxic
agents. However, some of the difficulties arise because of the lack of
standard procedures used for the assessment of chromosomal effects in
cultured lymphocytes and from a lack of understanding of the health
significance of chromosomal abnormalities.
Induced mitotic activity in peripheral lymphocytes and increases
in the rates of abnormal mitosis were reported by Sarto et al. (1978).
PbB levels in the range of 1.05-4.27 µmol/litre (22-89 µg/dl) were
reported to be associated with increased incidence of chromosomal
aberrations by Schmid et al. (1972) in 32 lead manufacturing workers,
by Nordenson et al. (1978) in 18 smelter workers and by Al-Hakkak et
al. (1986) in 19 lead manufacturing workers. It should be noted that
exposure of worker to potentially hazardous agents other than lead was
not documented in these studies.
Maki-Paakkanen et al. (1981) reported no change in chromosomal
aberration frequency among workers with PbB levels in the range of
1.82-5.76 µmol/litre (38-120 µg/dl); weak effects were found by Huang
et al. (1988b) when the PbB level exceeded 2.5 µmol/litre (52 µg/dl).
Qazi et al. (1980) reported increased numbers of cells with
chromosome breaks in a baby having a cord PbB level of 2.88 µmol/litre
(60 µg/dl) and a PbB level of 3.45 µmol/litre (72 µg/dl) at 2 weeks.
The effect was seen at 6 weeks and at 3 months but not later
(chelation therapy was given at 17 days and repeated at 5 months).
Bauchinger et al. (1977) tested blood taken from 38 children
selected from a school situated close to a lead plant and in whom the
PbB level was at least 1.44 µmol/litre (30 µg/dl). There was no
increase in the frequency of aberrations.
Grandjean et al. (1983) examined 10 long-term workers at a lead
storage battery plant. They found normal or slightly increased SCE
rates in workers with a PbB level of 1.4-3.6 µmol/litre (29-75 µg/dl)
and a ZPP level of 50-750 µmol/mol haemoglobin. The SCE increase was
correlated significantly with ZPP level.
Huang et al. (1988b) studied 21 lead-exposed workers from a
battery factory. They concluded that the SCE rate in workers with
long-term exposure to lead increased significantly when the mean PbB
level was 3.84 µmol/litre (80 µg/dl) or more. There were no
significant differences in SCE rates among the low- and medium-exposed
groups or the controls. These authors concluded that the effect of
chromosome damage caused by lead is not very strong.
Dalpra et al. (1983) examined blood samples taken from 19
children living in a contaminated area near a smelter and having PbB
levels in the range of 1.39-3.02 µmol/litre (29-63 µg/dl). They found
no effect on SCE frequency.
8.10 Carcinogenicity
Much has been written regarding the evidence that lead is
carcinogenic in humans (Moore & Meredith, 1979; Kazantzis, 1989;
Goyer, 1992). In several large epidemiological studies no association
was found which would associate lead with induction of cancer (Cooper
& Gaffey, 1975; Kang et al., 1980; Cooper, 1981; McMichael & Johnson,
1982). One major difficulty in many of the studies was the concurrent
exposure to potential carcinogens such as chromium (Davies, 1984), and
there has seldom been any attempt to deal with the primary etiological
agent (smoking) in the development of lung cancers associated with
lead exposure.
8.10.1 Occupational exposure and renal cancer
There are two case reports and very limited other epidemio-
logical evidence of an association between occupational exposure to
lead and renal cancer. Confounding variables, including use of tobacco
and exposure to other carcinogens, were not addressed (Baker et al.,
1980; Lilis, 1981; Selevan et al., 1985; Cantor et al., 1986).
The age-standardized mortality ratio for cancer was low in the
"lead poisoned" and other groups of workers from the Port Pirie study
by McMichael & Johnson (1982). The authors concluded that lead
poisoning did not increase the risk of cancer in humans.
8.10.2 Conclusion
Although some cohort mortality studies have indicated an
association between lead exposure and renal disease, there is no
association between renal cancer and lead in humans.
A Working Group convened by the International Agency for Research
on Cancer (IARC) in 1987 concluded that the evidence for the
carcinogenicity of lead and inorganic lead compounds in humans was
inadequate.
8.11 Effects on thyroid function
8.11.1 Occupational groups
Robins et al. (1983) found low values for serum thyroxine and
estimated free thyroxine in 7 of 12 workers having PbB levels above
2.11 µmol/litre (44 µg/dl). Both measures were correlated with PbB
level in a cross-sectional study of 47 foundry workers. Serum
thyrotropin and triiodothyronine levels were within the normal range
for both study groups.
No such relationship was found in a study by Refowitz (1984) of a
one-in-three random sample (N = 58) of male employees at a secondary
copper smelter. No thyroid abnormalities were observed and there was
no statistically significant relationship between PbB and serum
thyroxine or an estimate of free thyroxine in serum.
Tuppurainen et al. (1988) studied 176 African male workers in
Kenya and found that the duration of lead exposure correlated
negatively with serum free thyroxine and serum total thyroxine. The
correlation was strongest for the workers with the highest exposure
intensity over time. PbB data were available from periodic medical
examinations and average values (usually five determinations) were
used. The mean PbB was 2.73 µmol/litre (56.8 µg/dl), range
1.01-6.47 µmol/litre (21-135 µg/dl), and there was a mean exposure
duration of 7.6 years (range 1-20). The authors noted that current PbB
level, as a point determination, was not associated with total or free
thyroxine, triiodothyronine or thyrotropin in serum. They proposed
that long-term, highintensity exposure might be associated with
depressed thyroid function.
Gennart et al. (1992b) included assessment of thyroid function as
part of a study of lead-exposed workers. Data for serum levels of
triiodothyronine, thyroxine, free thyroxine index and thyroid-
stimulating hormone were within normal ranges for a group of 98
workers (mean PbB level, 2.45 µmol/litre or 51 µg/dl) and 85 controls
(1.00 µmol/litre or 20.9 µg/dl). The lead exposure of this study group
was considerably lower than in the workers in Kenya, and it was
suggested that thyroid function changes might not be indicators of
moderate exposure to lead.
8.11.2 Effects in children
Siegel et al. (1989) tested 68 children for thyroid function and
for PbB and found no statistically significant relationship between
lead and total or free thyroxine.
8.12 Immune system
In a review of the effects of lead on the immune responses of
experimental animals, Koller (1985) noted that there were few human
studies.
8.12.1 Occupational exposure
There is some evidence that lead workers with PbB levels in the
range of 1.0-4.10 µmol/litre (21-85 µg/dl) have increased
susceptibility to infections (colds and influenza) and have a
significant suppression of secretory IgA levels, a major factor in
defence against respiratory and gastrointestinal infections (Ewers et
al., 1982). There are also reports of impaired mitogen responses
(reflecting T-lymphocyte function) to phytohaemagglutinin (Jaremin,
1983) and of increased numbers of suppressor T-cells (Cohen et al.,
1989). However, on the basis of a study of 39 workers exposed to lead
oxide (mean PbB level of 1.84 µmol/litre or 38.5 µg/dl) and 21 control
subjects (mean PbB level of 0.57 µmol/litre or 11.8 µg/dl), Kimber et
al. (1986) concluded that chronic lead exposure in man is not
associated with the immunological changes that have been observed in
rodent studies.
Coscia et al. (1987) found increased B-lymphocyte percentage and
absolute count in workers currently exposed to lead with PbB levels
exceeding 2.4 µmol/litre (> 50 µg/dl).
8.12.2 Children
A study of 12 pre-school children (Reigart & Graber, 1976) with
PbB levels > 1.92 µmol/litre (40 µg/dl) did not reveal altered
immunity in comparison with a control group.
8.13 Effects on bone
The pharmacokinetics of the transfer of bone lead to other target
organs and blood has been discussed by Smith & Hursh (1977), Marcus
(1985a,b), Silbergeld et al. (1988) and Rabinowitz (1991) (see section
6.2.2).
Bone homoeostasis depends upon a complex interaction of its
various components, i.e. minerals, cells and the extracellular matrix
composed of collagenous and non-collagenous proteins (Marks & Popoff,
1988; Sauk & Somerman, 1991). Early work on the effects of lead on
calcium homoeostasis and calcium-mediated function was reviewed by
Pounds (1984). More recent work has been summarized by Pounds et al.
(1991), particularly the effects of lead on hormone action,
competition with calcium binding on calcium messenger systems, and the
impaired synthesis of collagen or sialoproteins. Additional evidence
for adverse effects of lead on the proteins in the mineral
compartment, thus affecting bone homoeostasis, has been reported by
Sauk & Somerman (1991).
8.14 Biomarkers for lead effects
The understanding and application of biomarkers for the
assessment of effect is becoming more widespread (IPCS, 1994).
It is desirable to identify biomarkers of lead exposure and
effect to provide easily measurable parameters that will facilitate
the risk assessment process. At present PbB levels are frequently
measured to assess both exposure and effect. Alternative biomarkers
for lead which may be easily measured are of biochemical effects,
particularly in the haem biosynthetic pathway.
However, the relationship between these effects and neurological
impairment caused by lead has not been established. Measurement of
these effects has also been used to provide biomarkers for exposure.
Anaemia has been associated with high levels of lead exposures in
the occupational setting. However, clinical tests for haemoglobin are
rather non-specific indicators of lead toxicity (Bernard & Becker,
1988). The steps within the haem biosynthetic pathway which have been
used to measure effect are: 1) inhibition of delta-aminolaevulinic
acid dehydratase (ALAD); 2) urinary excretion of delta-aminolaevulinic
acid (ALAU); (3) the accumulation of zinc protoporphyrin (ZPP) in
erythrocytes arising from the inhibition of the enzyme ferrochelatase
or the iron transport system.
There are several methodological problems related to the
measurement of ALAD, and the benefit of measuring enzyme activity in
blood in lieu of PbB determinations is not apparent (Mushak, 1989).
Early work (Roels et al., 1976) indicated enzyme inhibition at PbB
levels of 0.24 µmol/litre (> 5 µg/dl). However, the data at levels of
0.72 µmol/litre (< 15 µg/dl) appear scattered (Hernberg & Nikkanen,
1970; Granick et al., 1973).
Levels of ALA in urine have been used as an effect indicator of
lead exposure (Meredith et al., 1978). ALAU levels were found to
correlate with PbB levels as low as 0.864 µmol/litre (18 µg/dl), the
correlation becoming much stronger at PbB levels above 1.92 µmol/litre
(40 µg/dl) (Selander & Cramer, 1970). Given the present concern over
effects in children at PbB levels well below 1.92 µmol/litre
(40 µg/dl), the assay of ALAU concentrations may not be sensitive
enough to be of any value.
The effects of lead on porphyrin levels, both coproporphyrin in
urine and zinc protoporphyrin (ZPP) in blood, have been investigated
as possible biomarkers for lead. Measurement of urinary coproporphyrin
levels does not appear to be sensitive enough to be useful in
assessing exposure to lead at present environmental levels. Excretion
levels do not rise significantly until PbB levels exceed
1.92 µmol/litre (40 µg/dl) (Meredith et al., 1978). Other studies
indicate that this assay may not be sensitive enough for biological
monitoring at PbB levels between 0.96 and 1.20 µmol/litre (20 and
25 µg/dl). At present it would appear that ZPP has limited usefulness
as a biological indicator of exposure at exposures leading to PbB
levels lower than 1.20 µmol/litre (25 µg/dl) (Roels et al., 1976;
Piomelli et al., 1982; Hammond et al., 1985; Marcus & Schwartz, 1987).
The results may also be confounded by concurrent iron deficiency which
will alter the levels of ZPP.
Vitamin D metabolism involves cytochrome P-450-dependent enzymes
and thus may be affected by lead exposure. Measurement of serum levels
of 1,25-dihydroxy-vitamin-D has been proposed as a sensitive
biological monitor of lead exposure in children (Rosen et al., 1980;
Mahaffey et al., 1982; Koo et al., 1991). However, it should be noted
that dietary intakes of calcium and phosphorus, as well as circulating
levels of parathyroid hormone will regulate the production and
circulating concentrations of this vitamin D metabolite, thus making
correlations with PbB levels difficult without adequate information on
the nutritional status of the population under study (Rosen & Chisney,
1983). In populations showing adequate nutritional status, lead at
exposures leading to PbBs levels lower than 0.96 µmol/litre (20 µg/dl)
does not appear to have a demonstrable effect on circulating levels of
1,25-dihydroxy-vitamin-D.
9. EVALUATION OF HUMAN HEALTH RISKS
9.1 Exposure assessment
Lead is a ubiquitous element detected in all environmental media.
However, natural sources contribute only a small fraction of the
amounts of lead found in air, food, water and dust. The majority of
lead in these media arises from automobile and industrial emissions
and from the use of lead-containing solder and paints. Adults and
older children receive the largest proportion of lead intake from
foods, whereas dust, soil and food all make significant contributions
to the total lead intake of young children. The major contributions to
lead in soil and outdoor dust are from the combustion of fossil fuels
(principally leaded petrol), stationary sources such as smelters, and
peeling and flaking of lead-based paint.
9.1.1 General population exposure
In the absence of specific stationary sources of lead,
concentrations in ambient air are directly related to density of
traffic and whether lead is still utilized as an additive in petrol.
Reduction or elimination of lead in petrol in those countries which
have instituted regulations has resulted in a decline by as much as
eight-fold in ambient air concentrations of lead.
Levels of lead in indoor air are affected by the presence of
cigarette smoke and dust from lead-painted surfaces. Without such
sources, air lead levels indoors are about 60% of those in outdoor
air.
For most adults, the total daily exposure to lead is via food,
water and air. For infants aged up to 5 months, formula or breast milk
and water are the main sources of lead. In children, an additional
source of exposure is dust and soils. Absorption is dependent on the
chemical form of lead, type of soil and particle size
(bioavailability). Lead intake may be augmented from unusual sources
such as folk remedies, cosmetics and hobby activity. Community
contamination and workplace practices may contribute to lead exposure.
Food (including drinking-water and beverages) is the major source
of lead exposure for the general population. Infants and children may
receive an added lead burden from soil and dust. The most significant
foodstuffs will vary from country to country. In areas still utilizing
lead-soldered cans, levels of lead are substantially higher. Depending
upon lifestyles, there may be significant oral intake of lead from
some alcoholic beverages and due to the leaching of lead from low
temperature-fired ceramic containers.
Most drinking-water supplies contain lead levels lower than
5 µg/litre when they leave the treatment plant. However, where the
water is known to be plumbo-solvent, up to 40% of the samples may
exceed 100 µg/litre in homes where lead solder, lead pipes or brass
fixtures have been used.
Absorption of lead from the lung is a function of particle size
and pulmonary deposition pattern. Small particles (< 0.5 µm in
diameter) characteristic of ambient air will be deposited deeply in
the lungs with absorption rates of 90%. Larger particles, such as
those that may be encountered in occupational settings, exhibit high
deposition rates in the upper airway. Absorption of such particles
will be a function of both dissolution in the lung and particle
clearance to the gastrointestinal tract.
Human dermal absorption of inorganic lead through unabraded skin
is of limited significance.
9.1.2 Occupational exposures
In addition to exposure to lead in ambient air, water and food,
some workers may be exposed to airborne lead and dust within the
workplace. Actual levels will vary according to the engineering design
of the process equipment and the industrial hygiene practices
utilized.
9.2 Critical issues related to exposure evaluation
In view of the heterogeneity of responses to lead within human
populations, the complex interrelationships between exposure to lead
and a biological indicator for internal dose require consideration of
several key issues in order to assess human exposures.
9.2.1 Sampling and analytical concerns
Reliable comparisons of reported levels of exposure and/or dose
can only be made where authors have described the analytical and
sampling procedures in sufficient detail to allow the reader to
assess, for example, the integrity of the sampling procedures as well
as the specificity, precision and accuracy of all analytical methods.
Problems related to sampling cord blood also need to be considered.
Most studies have utilized analytical procedures of high quality.
However, consideration must be given to blood collection procedures
(finger stick versus venepuncture) when comparing results.
9.2.2 Data presentation
Inter-study comparisons of lead exposures are complicated by the
variety of methods used to present results, including median values as
well as geometric and arithmetic means. Some authors have used
log-transformed data.
In assessing exposure from data on teeth, one must know which
tooth and which compartment(s) within the tooth were sampled. In
addition, if data are to be compared between studies, authors must
state explicitly that all teeth analysed were without caries and were
shed spontaneously.
The exposure index lifetime average blood lead (PbB) level has
assisted in the assessment of cumulative exposures from serial PbB
data. It should not be interpreted as being equivalent to a single PbB
determination at a single point in life.
For some data/analytical purposes, age-specific PbB levels may be
more appropriate than a lifetime average.
9.3 Relationship between exposure and dose
The most widely used surrogate for the absorbed dose is whole PbB
concentration.
The relationship between PbB level and lead intake is curvilinear
over a wide range of PbB values. On the basis of a single study of 17
infants, the relationship between PbB level and lead intake from food
has been determined to be 0.0077 µmol lead/litre (0.16 µg/dl) per µg
lead intake per day for a median PbB level of approximately
0.48 µmol/litre (10 µg/dl).
Most studies of the relationship between PbB level and lead
exposure apply to a single environmental source, i.e. air, food, water
or soil/dust. A summary of the relationship between PbB level and lead
intake from individual media is given in Table 23.
9.4 Surrogate measures of dose
9.4.1 Blood
Whole PbB values are widely used as a measure of absorbed dose.
However, it is believed that plasma lead concentrations may better
reflect the "active" fraction of lead in blood and define the
relationship between PbB and tissue or organ accumulation (and
Table 23. Representative relationships of blood lead median level to intake of
lead for the general populationa
Population
Median Children Adults
Airb 0.09 µmol Pb/litre per µg 0.079 µmol Pb/litre per µg
Pb/m3 (1.92 µg Pb/dl) Pb/m3c (1.64 µg Pb/dl)
Water 0.003 µmol Pb/litre per µg
Pb/litre (0.06 µg Pb/dl)
Food 0.01 µmol Pb/litre per µg 0.002-0.003 µmol Pb/litre per
Pb/day (0.16 µg Pb/dl) µg Pb/day (0.04-0.06 µg Pb/dl)
Dustb 0.09 µmol Pb/litre per 1000 µg
Pb/g (1.8 µg Pb/dl)
Soilb 0.11 µmol Pb/litre per 1000 µg
Pb/g (2.2 µg Pb/dl)
a These data are provided for illustrative purposes only recognizing that the
relationships are curvilinear in nature and are broad guidelines which will
not apply at lower or higher levels of exposure.
b A value between 0.144 to 0.24 µmol Pb/litre or 3-5 µg Pb/dl per µg/m3
is obtained when one considers indirect contribution through deposition on
soil/dust.
c The air to blood lead relationship in occupational settings is best described
by a curvilinear relationship having slopes between 0.02 and 0.08 µg/m3
air. The slope is variable but lower than that found for humans in the general
environment, which is between 1.6 and 1.9 µg/m3.
effect), although there is little experimental data (because of
analytical limitations). PbB is distributed between plasma and
erythrocytes, with less than 5% being in the plasma. Most of the lead
is bound to haemoglobin.
Venous and capillary blood levels are generally equivalent,
provided that the sampling technique is adequate.
9.4.2 Urine
Urinary measurements of lead concentration are of limited value,
although they are used occasionally as a screening test for
occupational population groups.
9.4.3 Bone
Bone lead may be measured by non-invasive X-ray fluorescence, but
this technique is limited in sensitivity at present.
9.4.4 Tooth
Shed deciduous teeth have been used to provide an index of
exposure in early childhood. Interpretation of the analytical data is
dependent on the type of tooth and the part of the tooth (whole tooth,
dentine or circumpulpar dentine) analysed.
9.4.5 Hair
Hair is not useful for measurement of lead exposure.
9.5 Biochemical effects of lead
9.5.1 Haem synthesis
Evaluation of the quality of analytical data is an important
aspect in considering reports describing effects attributed to lead.
It should be noted that much of the data presented in this area has
not been as vigorously scrutinized as, for example, psychometric study
data.
An increase in erythrocyte protoporphyrin (EP) in children occurs
between PbB levels of 0.72 and 1.2 µmol/litre (15-25 µg/dl). Increases
in EP can be detected in men when the PbB level is above
1.20-1.44 µmol/litre (25-30 µg/dl), and in women when it is above
0.96-1.44 µmol/litre (20-30 µg/dl). It should be noted that the effect
of lead on haem is confounded by low iron status.
9.5.1.1 Urinary coproporphyrin
The coproporphyrin concentration in urine increases significantly
with PbB levels in excess of 1.92 µmol/litre (40 µg/dl).
9.5.1.2 Urinary aminolaevulinic acid in children
In children 1-5 years old, there is a linear relationship with
PbB in the range 1.2-3.6 µmol/litre (25-75 µg/dl). Data for children
with PbB levels of 0.24-1.92 µmol/litre (5-40 µg/dl) show essentially
no correlation with urinary aminolaevulinic acid (ALA) excretion.
Elevation of urinary ALA level is evident at a PbB level of about
1.68 µmol/litre (35 µg/dl).
9.5.1.3 Urinary aminolaevulinic acid in adults
Urinary excretion of ALA increases in men at PbB levels above
2.16 µmol/litre (45 µg/dl) and in women above 1.68 µmol/litre
(35 µg/dl).
9.5.1.4 delta-Aminolaevulinic acid dehydratase
There was a negative exponential relationship between PbB level
and delta-aminolaevulinic acid dehydratase (ALAD) activity in a
population of 10- to 13-year-old children with PbB levels in the range
of 0.19-1.97 µmol/litre (4.7-41 µg/dl). An effect was seen at a PbB
level of approximately 0.48 µmol/litre (10 µg/dl). There is an
apparent lack of a clearly defined threshold for lead inhibition of
ALAD in different age groups.
9.5.2 Vitamin D metabolism
In the presence of adequate nutritional status, PbB levels below
0.96 µmol/litre (20 µg/dl) appear to have no demonstrable effect on
circulating concentrations of 1,25-dihydroxy-vitamin-D. A PbB level
above 0.96 µmol/litre (20 µg/dl) is associated with a decrease in the
serum level of 1,25-dihydroxy-vitamin-D.
9.5.3 Dihydrobiopterin reductase
Inhibition of dihydrobiopterin reductase has been shown in humans
where the mean PbB level is as low as 0.48 µmol/litre (10 µg/dl).
A summary of some biochemical effects of lead is presented in
Table 24.
9.5.4 Haemopoietic system
9.5.4.1 Anaemia in adults
The estimated PbB associated with a decrease in haemoglobin
concentration is 2.40 µmol/litre (50 µg/dl).
9.5.4.2 Anaemia in children
Decreased haemoglobin levels in children occur at a PbB level of
approximately 1.92 µmol/litre (40 µg/dl). Anaemia, defined as a
haematocrit below 35%, is not found at a PbB level of less than
0.92 µmol/litre (20 µg/dl). The risk of having a haematocrit value
below 35% for a 1-year-old child is 2% at a PbB level of
Table 24. Biochemical effects of lead
Parameter Blood lead level above which the
biochemical effect is demonstrable with
current techniques
µmol/litre µg/dl
Protoporphyrin levels 0.96-1.44 20-30
Coproporphyrin levels 1.92 40
ALA urine levels 1.68 35
ALAD activity 0.48 10
1,25-dihydroxy-vitamin-D 0.96 20
Dihydrobiopterin reductase 0.48 10
0.96-1.87 µmol/litre (20-39 µg/dl); the contribution of iron
deficiency may account for a substantial proportion of this 2%.
Induction of anaemia is demonstrable at 1.92 µmol/litre (40 µg/dl).
9.5.4.3 Erythrocyte pyrimidine-5'-nucleotidase
A reduction of 20% or more in erythrocyte pyrimidine
-5'-nucleotidase activity is associated with PbB concentrations above
0.48 µmol/litre (10 µg/dl).
Effects of lead are demonstrable on a number of enzyme systems
and biochemical parameters. The PbB levels, above which effects are
demonstrable with current techniques for the parameters which may have
clinical significance, are all greater than 0.96 µmol/litre
(20 µg/dl). Some clinically insignificant effects on enzymes are
demonstrable at lower levels of PbB.
9.6 Nervous system
9.6.1 Adults
9.6.1.1 Central nervous system
With acute lead exposure resulting in a PbB level in excess of
3.84 µmol/litre (80 µg/dl), severe encephalopathy and/or coma may
occur. Central nervous system (CNS) symptoms are found in lead-exposed
adults when there is a history of several years of exposure to lead at
PbB levels that may not have exceeded 3.36 µmol/litre (70 µg/dl) and a
PbB level at the time of clinical assessment of at least
1.92 µmol/litre (40 µg/dl).
Impaired neurobehavioural test performance has been found in
lead-exposed workers. Changes in critical flicker fusion test have
been detected at a PbB level of about 2.4 µmol/litre (50 µg/dl).
Sensory motor function is generally more sensitive than cognitive
end-points in many neurobehavioural evaluations, the lowest-observed-
effect level being at about 1.92 µmol/litre (40 µg/dl).
It appears also that neuroelectrophysiological tests are
sensitive indicators of the CNS effects of lead. Reductions in
latencies of sensory evoked potentials and auditory event-related
potentials have been found in workers with average PbB levels of
approximately 1.92 µmol/litre (40 µg/dl).
9.6.1.2 Peripheral nervous system
Numerous studies have measured the conduction velocity of
electrically stimulated sensory and motor nerves in workers exposed to
lead. These nerve conduction velocity (NCV) studies have yielded
somewhat mixed results, with many showing a decrease in NCV in
relation to lead exposure (indexed as PbB level) and a few showing no
effect or occasionally even an increase in NCV associated with lead
exposure. Differences in the nerves evaluated, methodologies,
characterization of lead exposure, and control of confounding
variables underlie some of the variability in results across studies.
A statistical meta-analysis of 32 NCV studies has indicated that NCV
is significantly reduced in lead-exposed workers compared to reference
subjects, but that the median motor nerve shows more reliable effects
of lead than other nerves. This collective view of the evidence is
supported by key studies that provide compelling evidence of a causal
relationship between lead exposure and reductions in NCV, extending to
PbB levels as low as 1.44 µmol/litre (30 µg/dl). These effects may be
reversible depending on the duration and level of exposure.
9.6.1.3 Autonomic nervous system
Two reports examining the electrocardiographic RR interval
variability during deep breathing and the component CV of respiratory
sinus arrythmia demonstrated dysfunctions at an average PbB level of
1.68 µmol/litre (35 µg/dl). These results suggest autonomic nervous
system dysfunction, particularly the parasympathetic nervous system.
9.6.2 Children
Prospective and cross-sectional studies of children have
demonstrated associations of lead exposure, measured by various
indices, and intellectual performance. The association has been noted
across a wide range of exposure levels and in a variety of populations
before factors other than lead have been accounted for.
A key question is whether this statistical association is
directly attributable to the causal influence of lead on child IQ. It
is important to consider alternative contributory explanations, i.e.
random chance, unexplained confounding factors, reverse causality and
selection bias.
It is a matter of debate and conjecture as to the extent to which
these four issues should inhibit claims of a causal relationship in
the epidemiological studies. The essential problem is that
observational epidemiology cannot provide definitive evidence of
causality when the key statistical association is small, the temporal
relationship is unclear and major confounders are present. Animal
studies provide qualitative support for the claim of a causal role for
lead in affecting neuropsychological performance, but provide limited
assistance in establishing quantitative dose-effect relationships.
9.6.2.1 Type of effect
The clearest and most consistent associations have been found
with global measures, such as IQ, where the largest body of evidence
is available. Efforts to delineate the neuropsychological foundations
of this association with a wide variety of tests of specific
neuropsychological domains have not so far been successful.
9.6.2.2 Magnitude
Based on the evidence from cross-sectional and prospective
studies of populations with PbB levels generally below 1.2 µmol/litre
(25 µg/dl), the size of the apparent IQ effect (at ages 4 and above)
is a deficit of between 0 and 5 points (on a scale of 100 with a
standard deviation of 15) for each 0.48 µmol/litre (10 µg/dl)
increment in PbB level, with a likely apparent effect size of between
1 and 3 points. At PbB levels above 1.2 µmol/litre (25 µg/dl), the
relationship between PbB level and IQ may differ. Estimates of effect
size are group averages and only apply to the individual child in a
probabilistic manner.
Existing epidemiological studies do not provide definitive
evidence of a threshold. Below the PbB range of 0.48-0.72 µmol/litre
(10-15 µg/dl), the effect of confounding variables and limits in the
precision of analytical and psychometric measurements increases the
uncertainty attached to any estimate of effect. However, there is some
evidence of an association below this range.
9.6.2.3 Reversibility/persistence
Whilst the IPCS Task Group could not unequivocally state that
effects of early childhood exposure are persistent beyond childhood,
because the current data are too meagre, it was held that
neurobehavioural effects detected at age seven or later usually
persist. Measures in later childhood tend to be more predictive of
subsequent performance than those made earlier. It is more likely than
not that effects seen during school years are to some degree
irreversible. This has also been observed in later follow-up studies
conducted in other non-lead topics of child development research. One
of the difficulties is that there are too few studies concerning
long-term outcome in children with high early exposures and where the
sources of exposure are subsequently removed.
Virtually no useful data are available on the effects on IQ of
removing children from a "high" exposure environment to one of "low"
exposure or on reduction of body lead burden in children. This is not
to say that exposure should not be reduced when possible.
9.6.2.4 Age-specific sensitivity
From prospective studies it is not possible to determine an age
of critical sensitivity. This reflects the findings that serial PbB
measures taken at the age of 2 years and later are positively
correlated with, the individual rankings remaining approximately
constant, and this limits the ability to identify sensitive periods of
exposure.
9.6.2.5 Interactions/subgroups
The evidence is inconclusive on whether apparent effects are more
or less marked in different gender or socioeconomic status (SES)
subgroups. However, where there are suggestions of SES-related
differences, the apparent effects tend to be more marked in the lower
SES subgroups.
9.6.3 Animal studies
Experimental animal studies of CNS effects provide support for
the associations between PbB levels and neurobehavioural deficits
described in human epidemiological studies of lead. There is
supportive evidence both in terms of demonstrating causal
relationships and in the levels of PbB at which such effects are
observed, namely 0.528-0.72 µmol/litre (11-15 µg/dl). Moreover, they
provide qualitative parallels in the nature of the effects described,
as these effects include changes in learning and memory functions.
Experimental animal studies indicate that these CNS effects may depend
upon task complexity and can persist long beyond the termination of
lead exposure. These studies also provide information possibly
relevant to understanding mechanisms of effect. In addition, the
experimental animal studies provide such evidence in the absence of
the confounding factors and co-variates, such as parental IQ,
socioeconomic status, and quality of the home environment, that are
problematic to human epidemiological endeavours, and in the absence of
nutritional deficiencies that may arise in human populations.
9.7 Renal system
Renal function impairment was not associated with a PbB level
below 3.0 µmol/litre (62 µg/dl) when measured by blood urea nitrogen
and serum creatinine levels in lead workers. However, renal tubular
effects were detected in workers with a PbB level below 3.0 µmol/litre
when measured by more sensitive indicators such as NAG.
Most studies of the general population attempting to relate renal
function impairment to PbB concentration have not demonstrated an
effect with PbB levels below 1.8 µmol/litre (37.3 µg/dl). More
sensitive indicators of renal function may indicate a renal effect of
lead below this level.
9.8 Liver
Over-exposure to lead may inhibit drug metabolism in the liver.
9.9 Reproduction
9.9.1 Female
Studies on the risk of spontaneous abortion and reduced birth
weight associated with maternal PbB levels below 1.44 µmol/litre
(30 µg/dl) have yielded mixed results. Recent epidemiological studies
have shown exposure-related perturbations in the length of gestation,
significantly greater risks being associated with PbB levels of
0.72 µmol/litre (15 µg/dl) or more.
9.9.2 Male
PbB concentrations above 1.92 µmol/litre (40 µg/dl) have been
shown to affect sperm morphology and function. At present, the
reproductive consequences of these changes are unknown.
9.10 Blood pressure
A quantitative assessment of the collective evidence from all the
observational studies in adults is made difficult by the fact that
studies have adopted different policies regarding adjustment for
potential confounding factors (e.g., alcohol consumption). In
addition, quantitative findings from the two largest studies (BRHS and
NHANES II) have depended on whether adjustment was made for
geographical variations in blood pressure and blood lead.
The limited size of most observational studies has inevitably
meant that they could not consistently demonstrate a statistically
significant relationship. However, an overview of all the studies
shows that evidence is consistent with the centre-adjusted analysis of
the two main studies, i.e. there are very weak but statistically
significant associations between PbB level and both systolic and
diastolic blood pressures. The likely order of magnitude is that for
any two-fold increase in PbB level (e.g., from 0.8 to 1.6 µmol per
litre) there is a mean 1 mmHg increase in systolic blood pressure. The
association with diastolic blood pressure is of a similar magnitude.
Animal studies have provided plausible mechanisms for an effect
of lead on blood pressure. However, from such a small magnitude of
statistical associations in the presence of important confounders, one
cannot infer that low-level lead exposure is causally related to an
increase in blood pressure.
The two population studies relating PbB to cardiovascular disease
events show no statistically significant associations. Hence, there is
no clear evidence to suggest that lead has an impact of public health
importance as regards hypertension or risk of cardiovascular disease.
9.11 Carcinogenicity
Renal tumours occur in rats and mice administered high doses of
lead. However, the evidence for the carcinogenicity of lead and
inorganic lead compounds in humans is inadequate.
9.12 Immune system
There is no strong evidence in humans of an effect of lead on the
immune system.
10. RECOMMENDATIONS FOR PROTECTION OF HUMAN HEALTH
10.1 Public health measures
Public health measures should be directed towards reduction and
prevention of exposure to lead by reducing the use of lead and lead
compounds and by minimizing lead-containing emissions that result in
human exposures. This can be achieved by:
a) phasing-out any remaining uses of lead additives in motor fuels;
b) further reducing the use of lead-based paints, with the objective
of eliminating such paints;
c) development and application of methods for the safe and
economical remediation of lead-painted homes and lead
contaminated soil;
d) elimination of the use of lead in food containers (e.g., in the
seams of cans);
e) dissemination of information to assist with identification of
glazed food containers which may leach lead into food placed,
cooked or stored in the container;
f) eliminating any remaining agricultural uses of lead or lead
compounds (e.g., lead arsenate as an insecticide);
g) identifying and reducing, or preferably, eliminating lead found
as a contaminant or ingredient of folk remedies and cosmetics;
h) the use of materials and engineering practices to minimize
plumbosolvency in water treatment and water distribution systems;
i) systematic examination of processes in which lead is used or
recycled in order to identify and reduce lead exposure by means
of improved engineering design, of operators, by-standers and the
environment. Opportunities for technology transfer should be used
whenever possible.
10.2 Public health programmes
Programmes should be developed:
a) to enhance data collection and to make available to the public
information on the lead content of foodstuffs;
b) to facilitate identification of populations at high risk of
exposure to lead on the basis of monitoring data for lead in
food, air, water and soil;
c) that incorporate improved procedures for health risk assessment
of population groups at risk of exposure to lead;
d) that promote understanding and awareness concerning the effects
on human health associated with exposure to lead, while
recognizing cultural sensitivities;
e) that place emphasis on adequate nutrition, health care and
attention to socioeconomic conditions which may exacerbate the
effects of lead present in the environment.
10.3 Screening, monitoring and assessment procedures
Methods for evaluating the effects and associated risks of
exposure to lead require both improvement and further development or
research. In the short term the following measures are needed.
a) Screening
i) Blood lead measurements should be recognized as the biomarker of
choice for screening for previous exposure of children to lead.
ii) The sensitivity of the developing nervous system to the
potentially harmful effects of lead is such that other
biochemical measurements (e.g., erythrocyte protoporphyrin) are
not sufficiently sensitive for assessment of infants and
children.
b) Monitoring
i) More sensitive analytical methods should be developed for the
reliable measurement of blood lead levels below 0.72 µmol/litre
(15 µg/dl) to acceptable standards of precision and accuracy.
ii) There is a need for international analytical quality assurance
programmes utilizing reference lead-containing materials.
iii) All publications containing blood lead measurement data should
provide adequate data on current quality assurance and quality
control.
iv) Data comparisons are made more difficult by differences in units
and statistical techniques for data handling. Investigators
should be encouraged to adopt internationally agreed practices
(e.g., IUPAC units).
c) Assessment
i) Validated biomarkers are needed to define the relationship
between measures of environmental (external) exposure and
specific effects (biochemical or functional).
ii) Biomarkers indicative of deficits in cognitive performance are
needed to facilitate assessment of risk.
iii) Improved methods are needed for the definition of outcome effects
(especially IQ and neurobehavioural deficits) attributable to
lead at blood lead concentrations of about
0.48 µmol/litre (10 µg/dl) or less.
iv) Further data are required to determine whether outcome effects on
the nervous system attributed to lead are reversible or
permanent.
v) Biomarkers for the renal effects of lead are needed to link renal
damage with lead exposure and thereby improve assessment of risk
of renal damage.
11. FURTHER RESEARCH
a) Research is required to improve the process for the assessment of
risk associated with exposure to lead. Specifically this should
cover:
i) definition of the health significance of biochemical
changes associated with exposure to lead, with particular
attention to alterations associated with blood lead
concentrations of about 0.72 µmol/litre (15 µg/dl) or
less;
ii) work to define the bioavailability of lead from different
sources and to establish the relationship between exposure
(sources and speciation) and body burden;
iii) definition of the influence of host-related factors
(particularly nutrition) affecting absorption and
distribution of lead;
iv) intensification of kinetic studies of lead to provide an
improved data base for extrapolation between species;
v) elucidation of mechanisms of accumulation and mobilization
of lead from bone with particular attention to the
influence of pregnancy and ageing on kinetics;
vi) investigation of the pharmacokinetics of lead in the
pregnant female in relation to transfer of lead to the
developing embryo and fetus and factors that mitigate such
transfer;
vii) determination of the effects of pre- and post-natal
exposure to lead;
viii) improved definition of paternally mediated effects of lead
exposure on the reproductive process and outcomes.
b) More general research needs include the following:
i) rationalization of neurobehavioural tests used to assess
performance in children and animals in order to permit
comparisons of measurements and test data that use similar
biological mechanisms;
ii) follow-up research (where possible) of historical cohorts
of lead-poisoned adults to examine the sequelae;
iii) evaluation of intervention measures, such as lead
abatement from home and soil, and chelation on reduction
of blood lead levels and outcome effects on the central
nervous system.
12. PREVIOUS EVALUATIONS BY INTERNATIONAL BODIES
The carcinogenic potential of lead and lead compounds was last
evaluated by the International Agency for Research on Cancer in 1987
(IARC, 1987b). There was inadequate evidence for the carcinogenicity
of lead and inorganic lead compounds in humans, but sufficient
evidence was available to show that specified inorganic lead compounds
were carcinogenic in experimental animals. The overall evaluation
placed lead and inorganic lead compounds in Group 2B, i.e. possibly
carcinogenic to humans.
To protect workers from the adverse effects of lead on haem
synthesis and on the peripheral and central nervous system, a
health-based biological exposure limit of 1.92 µmol/litre (40 µg/dl)
was recommended by a WHO Study Group (WHO, 1980). It was further
recommended that blood lead (PbB) levels in women within the
reproductive age range should not exceed 1.44 µmol/litre (30 µg/dl).
Depending upon the background levels of PbB in the worker population,
air lead levels should not exceed 30-60 µg/m3 (WHO, 1980).
A drinking-water guideline value of 0.050 mg/litre was developed
in 1984 (WHO, 1984). This guideline value has been revised recently to
0.01 mg/litre (WHO, 1993).
Lead was evaluated by a WHO Working Group developing air quality
guidelines for Europe (WHO, 1987). Based on the assumption that PbB
levels in 98% of the population would be maintained at levels below
0.96 µmol/litre (20 µg/dl), a guideline value in the range of
0.5-1.0 µg/m3 (long-term average, such as annual mean) was
recommended.
At the 41st meeting of the Joint FAO/WHO Expert Committee on Food
Additives and Food Contaminants, a Provisional Tolerable Weekly Intake
(PTWI) of 25 µg/kg body weight was recommended (FAO/WHO, 1993). This
level refers to lead from all sources and was set to protect all
humans, including infants and children. It was based on a model
indicating daily intakes of lead between 3-4 µg/kg body weight by
infants and children and is not associated with an increase in PbB
concentrations.
Regulatory standards established by national bodies in several
countries and the European Economic Community are summarized in the
legal file of the International Register of Potentially Toxic
Chemicals (IRPTC, 1987).
REFERENCES
Aguilera de Benzo Z, Fraile R, & Carrion N (1989) Determination of
lead in whole blood by electrothermal atomization atomic absorption
spectrometry using tube and platform atomizers and dilution with Trito
X-100. J Anal At Spectrom, 4: 397-400.
Albahary C (1972) Lead and hemopoiesis: The mechanism and consequences
of the erythropathy of occupational lead poisoning. Am J Med,
52: 367-378.
Albert LA & Badillo F (1991) Environmental lead in Mexico. Rev Environ
Contamin Toxicol, 117: 1-49.
Alexander FW, Delves HT, & Clayton BE (1973) The uptake and excretion
by children of lead and other contaminants. In: Barth D, Berlin A,
Engel R, Recht P, & Smeets J ed. Environmental health aspects of lead:
Proceedings, International Symposium, Amsterdam, The Netherlands,
October 1972. Luxembourg, Commission of the European Communities,
pp 319-331.
Al-Hakkak ZS, Hamamy HA, Murad AMB, & Hussain AF (1986) Chromosome
aberrations in workers at a storage battery plant in Iraq. Mutat Res,
171: 53-60.
Altmann L, Weinsberg F, Sveinsson K, Lilienthal H, Wiegand H, &
Winneke G (1993) Impairment of long-term potentiation and learning
following chronic lead exposure. Toxicol Lett, 66(1): 105-112.
Alvares AP, Kapelner S, Sassa S, & Kappas A (1975) Drug metabolism in
normal children, lead-poisoned children, and normal adults. Clin
Pharmacol Ther, 17: 179-183.
Anderson C & Danylchuk KD (1977) The effects of chronic low level lead
intoxication on the Haversian remodeling system in dogs. Lab Invest,
37: 466-469.
Angle CR & McIntire MS (1978) Low level lead inhibition of erythrocyte
pyrimidine nucleotidase. Environ Res, 17: 296-302.
Angle CR & McIntire MS (1979) Environmental lead and children: the
Omaha study. J Toxicol Environ Health, 5: 855-870.
Angle CR, McIntire MS, & Swanson MS (1982) Erythrocyte nucleotides in
children - increased blood lead and cytidine triphosphate. Pediatr
Res, 16: 331-334.
Annest JL & Mahaffey KR (1984) Blood lead levels for persons aged 6
months-74 years: United States, 1976-1980. Hyattsville, Maryland, US
Public Health Service (Report No. 223, Series 11) (DHHS Publication
No. (PHS) 84-1683).
Annest JL, Pirkle JL, Makuc D, Neese JW, Bayse DD, & Kovar MG (1983)
Chronological trend in blood lead levels between 1976 and 1980. N Engl
J Med, 308: 1373-1377.
Araki S & Ushio K (1982) Assessment of the body burden of chelatable
lead: a model and its application to lead workers. Br J Ind Med,
39: 157-160.
Araki S, Honma T, Yanagihara S, & Ushio K (1980) Recovery of slowed
nerve conduction velocity in lead-exposed workers. Int Arch Occup
Environ Health, 46: 151-157.
Araki S, Aono H, Fukahori M, & Tabuki K (1984) Behaviour of lead and
zinc in plasma, erythrocytes, and urine and ALAD in erythrocytes
following intravenous infusion of CaEDTA in lead workers. Arch Environ
Health, 39: 363-367.
Araki S, Murata K & Aono H (1986a) Subclinical cervico-spino-bulbar
effects of lead: A study of short-latency somatosensory evoked
potentials in workers exposed to lead, zinc and copper. Am J Ind Med,
10: 163-175.
Araki S, Yokoyama K, Murata K, & Aono H (1986b) Determination of the
distribution of conduction velocities in workers exposed to lead, zinc
and copper. Br J Ind Med, 43: 321-326.
Araki S, Aono H, Murata K (1986c) Mobilisation of heavy metals into
the urine by CaEDTA: Relation to erythrocyte and plasma concentrations
and exposure indicators. Br J Ind Med, 43: 636-641.
Araki S, Murata K, & Aono H (1987) Central and peripheral nervous
system dysfunction in workers exposed to lead, zinc and copper: A
follow-up study of visual and somatosensory evoked potentials. Int
Arch Occup Environ Health, 59: 177-187.
Araki S, Murata K, Yokoyama K, & Uchida E (1992) Auditory
event-related potential (P300) in relation to peripheral nerve
conduction in workers exposed to lead, zinc, and copper: Effects of
lead on cognitive function and central nervous system. Am J Ind Med,
21: 539-547.
Arnetz BB & Nicolich MJ (1990) Modelling of environmental lead
contributors to blood lead in humans. Int Arch Occup Environ Health,
62: 397-402.
Aslam M, David SS, & Healy MA (1979) Heavy metals in some Asian
medicines and cosmetics. Publ Health (Lond), 93: 274-284.
Assenato G, Paci C, Baser M, Molinini R, Candela RG, Altamura BM, &
Giorgino R (1987) Sperm count suppression without endocrine
dysfunction in lead-exposed men. Arch Environ Health, 42: 124-127.
ATSDR (1993) Toxicological profile for lead. Atlanta, Georgia, US
Department of Health and Human Services, Agency for Toxic Substances
and Disease Registry (TP-92/12).
Aungst BJ, Docle JA, & Fung H (1981) The effect of dose on the
disposition of lead in rats after intravenous and oral administration.
Toxicol Appl Pharmacol, 61: 48-57.
Awad el Karim MA, Hamed AS, Elhaimi YAA, & Osman Y (1986) Effects of
exposure to lead among lead acid battery factory workers in Sudan.
Arch Environ Health, 41: 261-265.
Azar A, Trochimowicz HJ, & Maxfield ME (1973) Review of lead studies
carried out at Haskell Laboratory: two year feeding study and response
to hemorrhage study. In: Environmental health aspects of lead.
Proceedings of an international symposium, Amsterdam. Luxembourg,
Commission of the European Communities, pp 199-210.
Baghurst PA, McMichael AJ, Wigg N, Vimpani G, Robertson EF, Roberts
RJ, & Tong S-L (1992) Life-long exposure to environmental lead and
children's intelligence at age seven: the Port Pirie cohort study.
N Engl J Med, 327: 1269-1284.
Baker EL, Landrigan PJ, Barbour AG, Cox DH, Folland DS, Ligo RN, &
Throckmorton J (1979) Occupational lead poisoning in the United
States: clinical and biochemical findings related to blood lead
levels. Br J Ind Med, 36: 314-322.
Baker EL, Goyer RA, Fowler BA, Khettry U, Bernard DB, Adler S, White
R, Babayan R, & Feldman RG (1980) Occupational lead exposure,
nephropathy and renal cancer. Am J Ind Med, 1: 139-148.
Barrio A & Badia J (1985) [Lead encephalopathy in adults.] Rev Med
Chil, 113(6): 564-567 (in Spanish).
Barltrop D (1969) Mineral metabolism in pediatrics. Oxford, London,
Edinburgh, Melbourne, Blackwell Scientific Publishers.
Barltrop D (1972) Children and environmental lead. In: Hepple P ed.
Lead in the environment: Proceedings of a conference. London,
Institute of Petroleum, pp 52-60.
Barry PSI (1975) A comparison of concentrations of lead in human
tissues. Br J Ind Med, 32: 119-139.
Barry PSI (1981) Concentrations of lead in the tissues of children.
Br J Ind Med, 38: 61-71.
Barton JC, Conrad ME, Nuby S, & Harrison L (1978) Effects of iron on
the absorption and retention of lead. J Lab Clin Med, 92: 536-547.
Bauchinger M, Dresp J, Schmid E, Englert N, & Krause C (1977)
Chromosome analyses of children after ecological lead exposure. Mutat
Res, 56: 75-79.
Beattie AD, Moore MR, Goldberg A, Finlayson MJ, Graham JR, Mackie TM,
Main J, McLaren PA, Murdock KM, & Stewart FT (1975) Role of chronic
lead exposure in the aetiology of mental retardation. Lancet,
1: 589-592.
Beckett PH, Davis RD, & Brindley P (1979) The disposal of sewage
sludge onto farmland: the scope of the problem of toxic elements.
Water Pollut Control, 78: 419-436.
Behari LR, Singh S, Tandon SK, & Wahal AK (1982) Lead poisoning among
Indian silver jewellery makers. Ann Occup Hyg, 27: 107-109.
Behne D (1980) Problems of sampling and sample preparation for trace
element analysis in the health sciences. In: Bratter P & Schramel P
ed. Trace element analytical chemistry in medicine and biology.
Proceedings of the First International Workshop, Neuherberg, Federal
Republic of Germany. Berlin, New York, Walter de Gruyter & Co.,
pp 769-782.
Beijer K & Jernelöv A (1984) Microbial methylation of lead. In:
Grandjean P ed. Biological effects of organolead compounds. Boca
Raton, Florida, CRC Press, pp 13-19.
Bellinger DC & Stiles KM (1993) Epidemiologic approaches to assessing
the developmental toxicity of lead. Neurotoxicology, 14: 151-160.
Bellinger DC, Needleman HL, Leviton A, Waternaux C, Rabinowitz MR, &
Nichols ML (1984) Early sensory-motor development and prenatal
exposure to lead. Neurobehav Toxicol Teratol, 6: 387-402.
Bellinger DC, Leviton A, Waternaux C, Needleman H, & Rabinowitz M
(1987) Longitudinal analyses or prenatal and post-natal lead exposure
and early cognitive development. N Engl J Med, 316: 1037-1043.
Bellinger D, Sloman A, Leviton A, Rabinowitz M, Needleman H, &
Waternaux C (1991a) Low-level lead exposure and children's cognitive
function in the preschool years. Pediatrics, 87: 219-227.
Bellinger D, Leviton A, Rabinowitz M, Allred E, Needleman H,
Schoenbaum S (1991b) Weight gain and maturity in fetuses exposed to
low levels of lead. Environ Res, 54: 151-158.
Bellinger DC, Stiles KM, & Needleman HL (1992) Low-level lead
exposure, intelligence and academic achievement: A long-term follow-up
study. Pediatrics, 90: 855-861.
Berlin A & Schaller KH (1974) European standardized method for the
determination of delta-aminolevulinic acid dehydratase activity in
blood. Z Klin Chem Klin Biochem, 12: 389-390.
Bernard SR (1977) Dosimetric data and metabolic model for lead. Health
Phys, 32: 44-46.
Bernard BP & Becker CE (1988) Environmental lead exposure and the
kidney. Clin Toxicol, 26: 1-34.
Bernstein J, Evan AP, & Gardner KD (1987) Epithelial hyperplasia in
human polycystic kidney disease. Its role in pathogenesis and risk of
neoplasia. Am J Pathol, 129: 92-101.
Bert JL, van Dusen LJ, & Grace JR (1989) A generalized model for the
prediction of body lead burdens. Environ Med, 48: 117-127.
Beyer WN & Cromartie EJ (1987) A survey of Pb, Cu, Zn, Cd, Cr, As, and
Se in earthworms and soil from diverse sites. Environ Monit Assess,
8: 27-36.
Biggins PDE & Harrison RM (1979) Atmospheric chemistry of automotive
lead. Environ Sci Technol, 13: 558-565.
Billick IJ & Gray VE (1978) Lead based paint poisoning research:
review and evaluation 1971-1977. Washington, DC, US Department of
Housing and Urban Development.
Bishop L & Hill WJ (1983) A study of the relationship between blood
lead levels and occupational air lead levels. Am Stat, 37(4): 471-475.
Blair JA, Hilburn ME, Leeming RJ, McIntosh MJ, & Moore MR (1982) Lead
and tetrahydrobiopterin metabolism: possible effects on IQ. Lancet,
April 24, 964.
Body PE, Inglis GR, & Mulcahy DE (1988) Lead contamination in Port
Pirie South Australia. Adelaide, Australia, South Australia Department
of Environment and Planning (SADEP Report No. 101).
Bolger PM, Carrington CD, Capar SG, & Adams MA (1991) Reductions in
dietary lead exposure in the United States. Chem. Speciation
Bioavailab, 3(314): 31.
Bolter E, Butz T, & Arseneau JF (1975) Mobilization of heavy metals by
organic acids in the soils of a lead mining and smelting district.
In: Proceedings of the IXth Annual Conference on Trace Substances in
Environmental Health, Columbia, Missouri, 10-12 June 1975, pp 107-112.
Bondarenko GP (1968) An experimental study of the solubility of galena
in the presence of fulvic acids. Geochem Int, 5: 525-531.
Bornschein RL (1985) Influence of social factors on lead exposure and
child development. Environ Health Perspect, 62: 343-351.
Bornschein RL & Rabinowitz MB (1985) Proceedings of the Second
International Conference on Prospective Lead Studies. Environ Res,
38: 1-210.
Bornschein RL, Succop P, Dietrich KN, Clark CS, Que Hee S, & Hammond
PB (1985) The influence of social and environmental factors on dust
lead, hand lead, and blood lead levels in young children. Environ Res,
38: 108-118.
Bornschein RL, Succop PA, Krafft KM, Clark CS, Peace B, & Hammond PB
(1986) Exterior surface dust lead, interior house dust lead and
childhood lead exposure in an urban environment. In: Hemphill DD ed.
Trace substances in environmental health II. Columbia, Missouri,
University of Missouri, pp 322-332.
Bornschein RL, Succop PA, Krafft, KM, Clark CS, Peace B, & Hammond PB
(1987) Exterior surface dust lead, interior house dust lead and
childhood lead exposure in an urban environment. Trace Subst Environ
Health, 20: 322-332.
Bornschein RL, Grote J, Mitchell T, Succop PA, Dietrich KN, Krafft KM,
& Hammond PB (1989) Effects of prenatal lead exposure on infant size
at birth. In: Smith MA, Grant LD, & Sors AI ed. Lead exposure and
child development: An international assessment. Dordrecht, London,
Kluwer Academic Publishers, pp 307-319.
Boscolo P & Carmignani M (1988) Neurohumoral blood pressure regulation
in lead exposure. Environ Health Perspect, 78: 101-106.
Boutron CF, Görlach U, Candelone J-P, Bolshov MA, & Delmas RJ (1991)
Decrease in anthropogenic lead, cadmium and zinc in Greenland snows
since the late 1960s. Nature (Lond), 353: 153-156.
Braunstein GD, Dahlgren J, & Loriaux DL (1987) Hypogonadism in
chronically lead-poisoned men. Infertility, 1: 33-51.
Brown KW & Black SC (1983) Quality assurance and quality control data
validation procedures used for the Love Canal and Dallas lead soil
monitoring programs. Environ Monit Assess, 3: 113-122.
Brunekreef B (1984) The relationship between air lead and blood lead
in children: A critical review. Sci Total Environ, 38: 79-123.
Brunekreef B (1986) Childhood exposure to environmental lead. London,
United Kingdom, Monitoring and Assessment Research Centre.
Buchet J-P, Lauwerys R, Roels H, & Hubermont G (1977) Mobilization of
lead during pregnancy in rats. Int Arch Occup Environ Health,
40: 33-36.
Buchet J-P, Roels H, Bernard A, & Lauwerys R (1980) Assessment of
renal function of workers exposed to inorganic lead, cadmium, or
mercury vapour. J Occup Med, 22: 741-750.
Buchet JP, Lauwerys R, Vandervoorde A, & Pycke JM (1983) Oral daily
intake of cadmium, lead, manganese, copper, chromium, mercury,
calcium, zinc, and arsenic in Belgium: a duplicate meal study. Food
Chem Toxicol, 21: 19-24.
Bull RJ (1980) Lead and energy metabolism. In: Singhal PL & Thomas JA
ed. Lead toxicity. Baltimore, Maryland, Urban Schwarzenberg, Inc.,
pp 119-168.
Bushnell PJ & Bowman RE (1979a) Reversal learning deficits in young
monkeys exposed to lead. Pharmacol Biochem Behav, 10: 733-742.
Bushnell PJ & Bowman RE (1979b) Persistence of impaired reversal
learning in young monkeys exposed to low levels of dietary lead.
J Toxicol Environ Health, 5: 1015-1023.
Bushnell PJ & DeLuca H (1983) The effects of lactose on the absorption
and retention of dietary lead. J Nutr, 113: 365-378.
Bushnell PJ & Levin ED (1983) Effects of zinc deficiency on lead
toxicity in rats. Neurobehav Toxicol Teratology, 5: 283-288.
Bushnell G, Brockhaus A, & Baltissen R (1977a) Neurobehavioural and
system effects of long-term blood lead elevation in rats. Arch
Toxicol, 37: 247-263.
Bushnell PJ, Allan JR, & Marlar RJ (1977b) Scotopic vision deficits in
young monkeys exposed to lead. Science, 196: 333-335.
Byers RK & Lord EE (1943) Late effects of lead poisoning on mental
development. Am J Dis Child, 66: 471-494.
Calder IC, Collings MT, & Heyworth JS (1990) Evaluation of soil lead:
blood lead relationship for Port Pirie. Proceedings of the 23rd Annual
Conference of Trace Substances in Environmental Health. Environ
Geochem Health, 12(Suppl): 81-91.
Campbell BC, Beattie AD, Moore MR, Goldberg A, & Reid AG (1977) Renal
insufficiency associated with excessive lead exposure. Br Med J,
1: 482-485.
Cantor KP, Sontag JM, & Held MF (1986) Patterns of mortality among
plumbers and pipefitters. Am J Ind Med, 10: 73-89.
Capar SG & Rigsby EJ (1989) Survey of lead in canned evaporated milk.
J Assoc Off Anal Chem, 72: 378-381.
Cardenas A, Roels H, Bernard AM, Barbon R, Buchet JP, Lauwerys RR,
Rosello J, Ramis I, Mutti A, Franchini I, Fels LM, Stolte H, de Broe
ME, Nuyts GD, Taylor SA, & Price RG (1993) Markers of early renal
changes induced by industrial pollutants. II. Application to workers
exposed to lead. Br J Ind Med, 50: 28-36.
CDC (1985) Preventing lead poisoning in young children. Atlanta,
Georgia, US Department of Health and Human Services, Public Health
Service, Centers for Disease Control, pp 7-19 (Publication
No. 99-2230).
CEC (1981) Directive 77/312/CEE du Conseil du 29 mars 1977 concernant
la surveillance biologique de la population vis-à-vis du risque
saturnin. Rapport sur la mise en oeuvre de la directive. Luxembourg,
Commission des Communautés européennes, Direction Santé et Sécurité
(V/E/1/81-17/2).
Cerklewski FL & Forbes RM (1976) Influence of dietary zinc on lead
toxicity in the rat. J Nutr, 110: 689-696.
Chai S & Webb RC (1988) Effects of lead on vascular reactivity.
Environ Health Perspect, 78: 85-89.
Chakravorti S & Bhar S (1978) Chronic occupational exposure to lead:
An evaluation of environmental lead levels, lead absorption and
intoxication of storage battery workers. Bombay, Central Labour
Institute, Industrial Hygiene Division.
Chamberlain AC (1983) Effect of airborne lead on blood lead. Atmos
Environ, 17: 677-692.
Chamberlain AC (1985) Prediction of response of blood lead to airborne
and dietary lead from volunteer experiments with lead isotopes. Proc R
Soc (London), B224: 149-182.
Chamberlain AC, Heard MJ, Little P, Newton D, Wells AC, Wiffen RD
(1978) Investigations into lead from motor vehicles. Harwell, United
Kingdom Atomic Energy Authority (Report No. AERE-R9198).
Chan WH, Tang AJS, Chung DHS, & Lusis MA (1986) Concentration and
deposition of trace metals in Ontario - 1982. Water Air Soil Pollut,
29(4): 373-389.
Charney E, Kessler B, Farfel M, & Jackson D (1983) Childhood lead
poisoning: a controlled trial for the effect of dust-control measures
on blood lead levels. N Engl J Med, 309: 1089-1093.
Chau YK, Wong PTS, & Bengert GA (1979) Determination of tetraalkyl-
lead compounds in water, sediments, and fish samples. Anal Chem,
51: 186-188.
Chau YK, Wong PTS, & Kramer O (1980) Occurrence of tetraalkylead
compounds in the aquatic environment. Bull Environ Contam Toxicol,
24: 265-269.
Chettle DR, Scott MC, & Somervaille LJ (1991) Lead in bone: sampling
and quantitation using K x-rays excited by 109Cd. Environ Health
Perspect, 91: 49-55.
Chisolm JJ Jr (1962) Aminoaciduria as a manifestation of renal tubular
injury in lead intoxication and a comparison with patterns of
aminoaciduria seen in other diseases. J Pediatr, 60: 1-17.
Chisolm JJ & Brown DH (1979) Micromethod for zinc protoporphyrin in
erythrocytes: including new data on the absorptivity of zinc
protoporphyrin and new observations in neonates and sickle cell
disease. Biochem Med, 22: 214-237.
Chisolm JJ, Thomas DJ, & Hamill TG (1985) Erythrocyte porphobilinogen
synthase activity as an indicator of lead exposure to children. Clin
Chem, 31: 601-605.
Choie DD & Richter GW (1972) Cell proliferation in rat kidney induced
by lead acetate and effects of uninephrectomy on cell proliferation.
Am J Pathol, 66: 265-275.
Choie DD & Richter GW (1974) Cell proliferation in mouse kidney
induced by lead. Synthesis of deoxyribonucleic acid. Lab Invest,
30: 647-651.
Chowdhury AR, Dewan A, & Gandhi DN (1984) Toxic effect of lead on the
testes of rat. Biomed Biochem Acta, 43: 95-100.
Chowdhury AR, Chinoy NJ, & Gautam AK (1986) Effect of lead on human
semen. Adv Contracept Delivery Syst, 2: 208-211.
Christoffersson JO, Schutz A, Skerfving S, Ahlgren L, & Mattson S
(1986) Decrease of skeletal lead after end of occupational exposure.
Arch Environ Health, 41: 312-318.
Clark CS, Bornschein RL, Succop P, Que Hee SS, Hammond PB, & Peace B
(1985) Condition and type of housing as an indicator of potential
environmental lead exposure and pediatic blood lead levels. Environ
Res, 38: 46-53.
Clausing P, Brunekreef B, & van Wijnen JH (1987) A method for
estimating soil ingestion by children. Int Arch Occup Environ Health,
59: 73-82.
Cohen N, Modai D, Golik A, Weissgarten J, Peller S, Katz A, Averbukh
Z, & Shaked U (1989) Increased concanavalin A-induced suppressor cell
activity in humans with occupational lead exposure. Environ Res,
48: 1-6.
Cohn J, Cox C, & Cory-Slechta DA (1993) The effects of lead exposure
on learning in a multiple repeated acquisition and performance
schedule. Neurotoxicology, 14: 329-346.
Collins MF, Hrdina PD, & Whittle E (1992) Lead in blood and brain
regions of rats chronically exposed to low doses of the metal. Toxicol
Appl Pharmacol, 65: 314-322.
Colombo A (1985) The underdefined nature of the blood lead-air lead
relationship from biological and statistical grounds. Atmos Environ,
19: 1485-1493.
Columbano A, Ledda-Columbano GM, Lee G, Rajalakshmi S, & Sarma DSR
(1984) Inability of mitogen-induced liver hyperplasia to support
induction of enzyme-altered islands induced by liver carcinogens.
Cancer Res, 47: 5557-5559.
Cookman GR, King W, & Regan CM (1987) Chronic low-level lead exposure
impairs embryonic to adult conversion of the neural cell adhesion
molecule. J Neurochem, 49(2): 399-403.
Cookman GR, Hemmens SE, Keane GJ, King WB, & Regan CM (1988) Chronic
low level lead exposure precociously induces rat glial development
in vitro and in vivo. Neurosci Lett, 86: 33-37.
Cools A, Salle HJA, Verberk MM, & Zielhuis RL (1976) Biochemical
response of male volunteers ingesting inorganic lead for 49 days. Int
Arch Occup Environ Health, 38: 129-139.
Cooney GH, Bell A, McBride W, & Carter C (1989a) Neurobehavioural
consequences of prenatal low level exposures to lead. Neurotoxicol
Teratol, 11: 95-104.
Cooney GH, Bell A, McBride W, & Carter C (1989b) Low level exposures
to lead: the Sydney lead study. Dev Med Child Neurol, 31: 640-649.
Cooney G, Bell A, & Stavron C (1991) Low level exposures to lead and
neurobehavioural development: the Sydney study at seven years. In:
Heavy metals in the environment. Edinburgh, CEP Consultants, pp 16-19.
Cooper WC (1981) Mortality in employees of lead production facilities
and lead battery plants, 1971-1975. In: Environmental lead:
Proceedings of the Second International Symposium on Environmental
Lead Research, Cincinnati. Ohio, December 1978. New York, London, San
Francisco, pp 111-143.
Cooper WC (1988) Deaths from chronic renal disease in US battery and
lead production workers. Environ Health Perspect, 78: 61-63.
Cooper WC & Gaffey WR (1975) Mortality of lead workers. J Occup Med,
17: 100-107.
Cory-Slechta DA (1990a) Lead exposure during advanced age: alterations
in kinetics and biochemical effects. Toxicol Appl Pharmacol,
104: 67-78.
Cory-Slechta DA (1990b) Exposure duration modifies the effects of low
level lead on fixed interval performance. Neurotoxicology,
11: 427-442.
Cory-Slechta DA & Pokora MJ (1991) Behavioral manifestations of
prolonged lead exposure initiated at different stages of the life
cycle: I. Schedule-controlled responding. Neurotoxicology,
12(4): 745-760.
Cory-Slechta DA & Thompson T (1979) Behavioural toxicity of chronic
post-weaning lead exposure in the rat. Toxicol Appl Pharmacol,
47: 151-159.
Cory-Slechta DA & Widzowski DV (1991) Low level lead exposure
increases sensitivity to the stimulus properties of dopamine D1 and D2
agonists. Brain Res, 553: 65-74.
Cory-Slechta DA, Weiss B, & Cox C (1985) Performance and exposure
indices of rats exposed to low concentrations of lead. Toxicol Appl
Pharmacol, 78: 291-299.
Coscia GC, Discalzi G, & Ponzetti C (1987) Immunological aspects of
occupational lead exposure. Med Lav, 78: 360-364.
Cramer K, Goyer RA, Jagenburg OR, & Wilson MH (1974) Renal
ultrastructure, renal function and parameters of lead toxicity in
workers with different length of lead exposure. Br J Ind Med,
31: 113-127.
Cueto LM, Baer RD, & Gonzalez EM (1989) Three cases of unusual lead
poisoning. Am J Gastroenterol, 84: 1460.
Culbard EB, Thornton L, Watt JM, Wheatley M, Moorcroft S, & Thompson M
(1988) Metal contamination in British urban dusts and soils. J Environ
Qual, 17: 226-234.
Cullen MR, Kayne RD, & Robins JM (1984) Endocrine and reproductive
dysfunction in men associated with occupational inorganic lead
intoxication. Arch Environ Health, 39: 431-440.
Dabeka RW & McKenzie AD (1987) Lead, cadmium, and fluoride levels in
market milk and infant formulas in Canada. J Assoc Off Anal Chem,
70(4):754-757.
Dabeka RW, McKenzie AD, & Lacroix GMA (1987) Dietary intakes of lead,
cadmium, arsenic and fluoride by Canadian adults: a 24-hour duplicate
diet study. Food Addit Contam, 4(1): 89-102.
Dalpra L, Tibiletti MG, Nocera G, Giulotto P, Auriti L, Carnelli V, &
Simoni G (1983) SCE analysis in children exposed to lead emission from
a smelting plant. Mutat Res, 120: 249-256.
Danielson BRG, Dencker L, & Lindgren A (1983) Transplacental movement
of inorganic lead in early and late gestation in the mouse. Arch
Toxicol, 54: 97-107.
David O, Clark J, & Voller K (1972) Lead and hyperactivity. Lancet,
2: 900-903.
Davidson C, Grimm T, & Nasta M (1981) Airborne lead and other elements
derived from local fires in the Himalays. Science, 214: 1344-1346.
Davies BE (1983) A graphical estimation of the normal lead content of
some British soils. Geoderma, 29: 67-75.
Davies JM (1984) Lung cancer mortality among workers making lead
chromate and zinc chromate pigments at three English factories. Br J
Ind Med, 41: 158-169.
Davies PH & Everhart WH (1973) Effects of chemical variations in
aquatic environments: Volume 3 - Lead toxicity to rainbow trout and
testing application factor concept. Washington, DC, US Environmental
Protection Agency (EPA Report No. R373011C).
Davies BE, Elwood PC, Gallacher J, & Ginnever RC (1985) The
relationships between heavy metals in garden soils and house dusts in
an old lead mining area of North Wales, Great Britain. Environ Pollut,
B9: 255-266.
Davies DJ, Thornton I, Watt JM, Culbard EB, Harvey PG, Delves HT,
Sherlock JC, Smart GA, Thomas JFA, & Quinn MJ (1987a) Lead intake and
blood lead in 2 year olds UK urban children. Sci Total Environ,
90: 13-29.
Davies DJ, Watt JM, & Thornton I (1987b) Lead levels in Birmingham
dusts and soils. Sci Total Environ, 67: 177-185.
Davis JM & Grant LD (1992) The sensitivity of children to lead. In:
Guzelian PS, Henry CJ, & Olin S ed. Similarities and differences
between children and adults: Implications for risk assessment.
Washington, DC, ILSI Press, pp 15-27.
Davis JM & Svendsgaard DJ (1990) Nerve conduction velocity and lead: A
critical review and meta-analysis. In: Johnson BL, Anger WK, Durao A,
& Xinteras C ed. Advances in neurobehavioural toxicology: Applications
in environmental and occupational health. Chelsea, Michigan, Lewis
Publishers, pp 353-376.
Davis S, Waller P, Buschbom R, Balfour J, & White P (1990a)
Quantitative estimates of soil ingestion in normal children between
the ages of 2 and 7 years: Populations-based estimates using
aluminium, silicon, and titanium as soil tracer elements. Arch Environ
Health, 45: 112-122.
Davis JM, Otto DA, Weil DE, & Grant LD (1990b) The comparative
developmental neurotoxicity of lead in humans and animals.
Neurotoxicol Teratol, 12: 215-229.
Day JP, Hart M, & Robinson MS (1975) Lead in urban street dust. Nature
(Lond), 253: 343-345.
Dees JH, Heatfield BM, Reuben MD, & Trump BF (1980) Adenocarcinoma of
the kidney. III. Histogenesis of renal adenocarcinomas induced in rats
by N-(4-fluoro-4-biphenyl)acetamide. J Natl Cancer Inst,
64: 1537-1545.
De La Burde B & Choate MS (1972) Does asymptomatic lead exposure in
children have latent sequelae? J Pediatr, 81: 1088-1096.
De La Burde B & Choate MS (1975) Early asymptomatic lead exposure and
development at school age. J Pediatr, 87: 638-664.
De Leacy EA (1987) Lead-crystal decanters - a health risk? Med J Aust,
147: 622.
Delumyea R & Kalivretenos A (1987) Elemental carbon and lead content
of fine particles from American and French cities of comparable size
and industry, 1985. Atmos Environ, 21: 1643-1647.
Delves HT & Campbell MJ (1988) Measurements of total lead
concentrations and of lead isotope ratios in whole blood by use of
inductively coupled plasma source mass spectrometry. J Anal At
Spectrom, 3: 343-348.
Desilva PE & Donnan MB (1980) Blood lead levels in Victorian children.
Med J Aust, 2: 315-318.
Diemel JAL, Brunekreef B, Boleu JSM, Biersteker K, & Veenstra SJ
(1981) The Arnhem lead study. II. Indoor pollution, and indoor/outdoor
relationships. Environ Res, 25: 449-456.
Dieter M, Mathews HB, Jeffcoate RA, & Moseman RF (1993)
Bioavailability of lead found in mine tailings from Alaska. J Toxicol
Environ Health, 39: 79-93.
Dietrich KN, Krafft KM, Bier M, Succop P, Berger O, & Bornschein R
(1986) Early effects of fetal lead exposure: neurobehavioural findings
at six months. Int J Biosoc Res, 8: 151-168.
Dietrich KN, Krafft KM, Bornschein RL, Hammond PB, Berger OM, Succop
PA, & Bier M (1987) Low-level fetal lead exposure effect on
neurobehavioural development in early infancy. Pediatrics,
80: 721-730.
Dietrich KN, Krafft KM, Bier M, Berger O, Succop PA, & Bornschein RL
(1989) Neurobehavioural effects of foetal lead exposure: the first
year of life. In: Smith MA, Grant LD, & Sors AI ed. Lead exposure and
child development: An international assessment. Dordrecht, London,
Kluwer Academic Publishers, pp 320-331.
Dietrich KN, Succop PA, Bornschein RL, Krafft KM, Berger O, Hammond
PB, & Buncher CR (1990) Lead exposure and neurobehavioural development
in later infancy. Environ Health Perspect, 89: 13-19.
Dietrich KN, Succop PA, Berger OG, Hammond PB, & Bornschein RL (1991)
Lead exposure and cognitive development of urban, preschool children:
the Cincinnati lead cohort at age 4 years. Neurotoxicol Teratol,
13: 203-211.
Dietrich KN, Succop PA, Berger OG, & Keith RW (1992) Lead exposure and
the central auditory processing abilities and cognitive development of
urban children: the Cincinnati lead study cohort at age 5 years.
Neurotoxicol Teratol, 14: 51-56.
Dietrich K, Berger O, Succop P, Hammond P, & Bornschein R (1993a) The
developmental consequences of low to moderate prenatal and postnatal
lead exposure: Intellectual attainment in the Cincinnati Lead Study
Cohort following school entry. Neurotoxicol Teratol, 15: 37-44.
Dieterich K, Berger O, & Succop P (1993b) Lead exposure and the motor
developmental status of urban 6-year-old children in the Cincinnati
Prospective Study. Pediatrics, 91: 301-307.
Djuric D, Kerin Z, Graovac-Leposavic L, Novak L, & Kop M (1971)
Environmental contamination by lead from a mine and smelter: A
preliminary report. Arch environ Health, 23: 275-279.
Dolenc P, Staessen JA, Lauwerys RR, & Amery A (1993) Short report:
low-level lead exposure does not increase the blood pressure in the
general population. J Hypertens, 11: 589-593.
Dong A, Simsiman GV, & Chesters G (1985) Release of phosphorus and
metals from soils and sediments during dispersion. Soil Sci,
139: 97-99.
Donovan MP, Schein LG, & Thomas JA (1980) Inhibition of
androgen-receptor interaction in mouse prostate gland cytosol by
divalent metal ions. Mol Pharmacol, 17: 156-162.
Drasch GA & Ott J (1988) Lead in human bones. Investigations of an
occupationally non-exposed population in southern Bavaria (FGR). II.
Children. Sci Total Environ, 68: 61-69.
Drasch GA, Bohm J, & Baur C (1987) Lead in human bones. Investigations
of an occupationally non-exposed population in southern Bavaria (FGR).
I. Adults. Sci Total Environ, 64: 303-315.
Dresner DL, Ibrahim NG, Mascarenhas BR, & Levere RD (1982) Modulation
of bone marrow heme and protein synthesis by trace elements. Environ
Res, 28: 55-66.
Drill S, Konz J, Mahar H, & Morse M (1979) The environmental lead
problem: An assessment of lead in drinking water from a multi-media
perspective. Washington, DC, US Environmental Protection Agency
(EPA-570/9-79-003, NTIS-PB-296556).
Duggan MJ (1980) Lead in urban dust: An assessment. Water Air Soil
Pollut, 14: 309-321.
Egle P & Shelton KR (1986) Chronic lead intoxication causes a brain
specific nuclear protein to accumulate in the nuclei of cells lining
kidney tubules. J Biol Chem, 261: 2294-2298.
Elias R (1985) Lead exposures in the human environment. In: Mahaffey
KR ed. Dietary and environmental lead: human health effects.
Amsterdam, Oxford, New York, Elsevier Science Publishers, pp 79-109.
Ellen G & van Loon JW (1990) Determination of cadmium and lead in
foods by graphite furnace atomic absorption spectrometry with Zeeman
background correction: test with certified reference materials. Food
Addit Contam, 7: 265-273.
Elwood PC (1986) The sources of lead in blood: A critical review. Sci
Total Environ, 52: 1-23.
Elwood PC, Davey-Smith G, Oldham PD, & Toothill C (1988a) Two Welsh
surveys of blood lead and blood pressure. Environ Health Perspect,
78: 119-121.
Elwood PC, Yarnell JWG, Oldham PD, Catford JC, Nutbeam D, Davey-Smith
G, & Toothill C (1988b) Blood pressure and blood lead in surveys in
Wales. Am J Epidemiol, 127: 942-945.
Ernhart B & Morrow-Tlucak M (1987) The caretaking environment and
exposure of children to lead. In: Thornton I & Culbard E ed. Lead in
the home environment. Northwood, Science Reviews Ltd.
Ernhart CB, Wolf AW, Sokol RJ, Brittenham GM, & Ernhard P (1985) Fetal
lead exposure: antenatal factors. Environ Res, 38: 54-66.
Ernhart CB, Wolf WW, Kennard MJ, Erhard P, Filipovick HF, & Sokol RJ
(1986) Intrauterine exposure to low levels of lead: the status of the
neonate. Arch Environ Health, 41: 287-291.
Ernhart CB, Morrow-Tlucak M, Marler MR, & Wolf AW (1987) Low level
lead exposure in the prenatal and early preschool periods: early
preschool development. Neurotoxicol Teratol, 9: 259-270.
Ernhart CB, Morrow-Tlucak M, & Wolf AW (1988) Low level lead exposure
and intelligence in the preschool years. Sci Total Environ,
71: 453-459.
Ernhart CB, Morrow-Tlucak M, Worf AW, Super D, & Drotar D (1989a) Low
level lead exposure in the prenatal and early preschool periods:
intelligence prior to school entry. Neurotoxicol Teratol, 11: 161-170.
Ernhart CB, Brittenham G, Marler MR, & Sokol RJ (1989b) Lead related
birth defects: some methodologic issues. In: Smith MA, Grant LD, &
Sors AI ed. Lead exposure and child development: An international
assessment. Dordrecht, London, Kluwer Academic Publishers, pp 357-370.
Evans RD & Rigler FH (1985) Long distance transport of anthropogenic
lead as measured by lake sediments. Water Air Soil Pollut,
24: 141-151.
Everson J & Patterson CC (1980) "Ultra-clean" isotope dilution/mass
spectrometric analyses for lead in human blood plasma indicate that
most reported values are artificially high. Clin Chem, 26: 1603-1607.
Evis MJ, Kane KA, Moore MR, & Parratt JR (1985) The effects of chronic
low lead treatment and hypertension on the severity of cardiac
arrhythmias induced by coronary artery ligation in anaesthetized rats.
Toxicol Appl Pharmacol, 80: 234-242.
Evis MJ, Dhaliwal K, Kane KA, Moore MR, & Parratt JR (1987) The
effects of chronic lead treatment and hypertension on the severity of
cardiac arrhythmias induced by coronary artery occlusion or by
noradrenaline in anaesthetised rats. Arch Toxicol, 59: 336-340.
Ewers U, Stiller-Winkler R, & Idel H (1982) Serum immunoglobulin,
complement C3, and salivary IgA level in lead workers. Environ Res,
29: 351-357.
Fanning D (1988) A mortality study of lead workers, 1926-1985. Arch
Environ Health, 43: 247-251.
FAO/WHO (1993) Evaluation of certain food additives and contaminants.
Forty-first report of the Joint FAO/WHO Expert Committee on Food
Additives. Geneva, World Health Organization (Technical Report Series
837).
Feigl F, Anger V, & Oesper RE (1972) Spot tests in inorganic analysis.
London, Elsevier Publishing Company, pp 285-286.
Fergusson DM, Fergusson JE, Horwood LJ, & Kinzett NG (1988a) A
longitudinal study of dentine lead levels, intelligence, school
performance and behaviour. Part I. Dentine lead levels and exposure to
environmental risk factors. J Child Psychol Psychiatr, 29: 781-792.
Fergusson DM, Fergusson JE, Horwood LJ, & Kinzett NG (1988b) A
longitudinal study of dentine levels, intelligence, school performance
and behaviour. Part II. Dentine lead and cognitive ability. J Child
Psychol Psychiatr, 29: 793-809.
Fergusson DM, Fergusson JE, Horwood LJ, & Kinzett NG (1988c) A
longitudinal study of dentine lead levels, intelligence, school
performance and behaviour. Part III. Dentine lead levels and
attention/activity. J Child Psychol Psychiatr, 29: 811-824.
Fergusson JE, Fergusson DM, Horwood LJ, & Kinzett NG (1989) A
longitudinal study of dentine lead levels, intelligence, school
performance and behaviour: The measurement of dentine lead. Sci Total
Environ, 80: 229-241.
Fernando NP, Healy MA, Aslam M, Davis SS, & Hussein A (1981) Lead
poisoning and traditional practices: The consequences for world
health. A study in Kuwait. Public Health (Lond), 95: 250-260.
Fischbein A, Alvares AP, Anderson KE, Sassa S, & Kappas A (1977) Lead
intoxication among demolition workers: the effect of lead on the
hepatic cytochrome P-450 system in humans. J Toxicol Environ Health,
3: 431-437.
Fischbein A, Rice C, Sarkozi L, Kon SH, Petrocci M, & Selikoff IJ
(1979) Exposure to lead in firing ranges. J Am Med Assoc,
241: 1141-1144.
Fischbein A, Thornton J, Blumberg WE, Bernstein J, Valciukas JA, Moses
M, Davidow B, Kaul B, Sirota M, & Selikoff IJ (1980) Health status of
cable splicers with low-level exposure to lead: results of a clinical
survey. Am J Public Health, 70: 697-700.
Flegal AR, Rosman KJR, & Stephenson MD (1987) Isotope systematics of
contaminant leads in Monterey Bay. Environ Sci Technol, 21: 1075-1079.
Flora S-S & Tandon SK (1987) Effect of combined exposure to lead and
ethanol on some biochemical indices in the rat. Biochem Pharmacol,
36: 537-541.
Florence TM, Lilley SG, & Stauber JL (1988) Skin absorption of lead.
Lancet, 2: 157-158.
Fouassin A & Fondu M (1980) Evaluation of the daily intake of lead and
cadmium from food in Belgium. Arch Belg Méd Soc Hyg Méd Trav Méd Lég,
38: 453-467.
Foster WG (1992) Reproductive toxicity of chronic lead-exposure in the
female cynomolgus monkeys. J Reprod Toxicol, 16: 123-131.
Fouts PJ & Page JH (1942) The effect of chronic lead poisoning on
arterial blood pressure in dogs. Am Heart J, 24: 329-331.
Fowler BA, Kimmel CA, Woods JS, McConnell EE, & Grant LD (1980)
Chronic low-level lead toxicity in the rat: III. An integrated
assessment of long-term toxicity with special reference to the kidney.
Toxicol Appl Pharmacol, 56: 59-77.
Fox DA & Sillman AJ (1979) Heavy metals affect rod, but not cone
pholoreceptors. Science, 206: 78-80.
Fox DA, Lewkowski JP, & Cooper GP (1977) Acute and chronic effects of
neonatal lead exposure on developmental of the visual worked response
in rats. Toxicol Appl Pharmacol, 40: 449-461.
Freeman GB, Johnson JD, Killinger JM, Liao SC, Feder PI, Davis AO,
Ruby MV, Chaney RL, Lovre SC, & Bergstrom PD (1992) Relative
bioavailability of lead from mining waste soils in rats. Fundam Appl
Toxicol, 19: 388-398.
Friberg L (1988) Quality assurance. In: Clarkson TW, Friberg L,
Nordstrom GF, & Sager PR ed. Biological monitoring of toxic metals.
New York, Plenum Press, pp 103-126.
Friberg L & Vahter M (1983) Assessment of exposure to lead and cadmium
through biological monitoring: Results of a UNEP/WHO global study.
Environ Res, 30: 95-128.
Friedman JA & Weinberger HL (1990) Six children with lead poisoning. J
Dis Child, 144: 1039-1044.
Fullmer CS & Rosen JF (1990) Effect of dietary calcium and lead status
on intestinal calcium absorption. Environ Res, 51: 91-99.
Fullmer CS (1990) Intestinal lead and calcium absorption: Effect of
1,25-dihydroxy-cholecalciferol and lead status. Proc Soc Exp Biol Med,
194: 258-264.
Fulton M, Thomson G, Hunter R, Raab G, Laxen D, & Hepburn W (1987)
Influence of blood lead on the ability and attainment of children in
Edinburgh. Lancet, 30 May: 1221-1226.
Galal-Gorchev H (1991a) Dietary intake of pesticide residues, cadmium,
mercury and lead. Food Addit Contam, 8(6): 793-806.
Galal-Gorchev H (1991b) Global overview of dietary lead exposure.
Special issue incorporating the Proceedings of the Symposium on the
Bioavailability and Dietary Exposure of Lead, Chapel Hill, North
Carolina, September 1990. Chem Speciation Bioavailab, 3(3/4): 5-11.
Garber BT & Wei E (1974) Influence of dietary factors on the
gastrointestinal absorption of lead. Toxicol Appl Pharmacol,
27: 685-691.
Garcia-Miragaya J (1984) Levels, chemical fractionation, and
solubility of lead in roadside soils of Caracas, Venezuela. Soil Sci,
138: 147-152.
Gartrell MJ, Craun JC, Podrebarac DS, & Gunderson EL (1985a)
Pesticides, selected elements, and other chemicals in infant and
toddler total diet samples, October 1979 -September 1980. J Assoc Off
Anal Chem, 68: 1184-1187.
Gartrell MJ, Craun JC, Podrebarac DS, & Gunderson EL (1985b)
Pesticides, selected elements, and other chemicals in infant and
toddler total diet samples, October 1979 -September 1980. J Assoc Off
Anal Chem, 81: 1163-1183.
Gartside PS (1988) The relationship of blood lead levels and blood
pressure in NHANES II: additional calculations. Environ Health
Perspect, 78: 31-34.
Gartside PS & Buncher CR (1982) Relationship of air lead and blood
lead for workers at an automobile battery factory. Int Arch Occup
Environ Health, 50: 1-10.
GEMS (1985) Global Environmental Monitoring System: Assessment of
human exposure to lead: Comparison between Belgium, Malta, Mexico and
Sweden. Stockholm, Karolinska Institute.
Gennart JP, Bernard A, & Lauwerys R (1992) Assessment of thyroid,
testes, kidney and autonomic nervous system function in lead-exposed
workers. Int Arch Occup Environ Health, 64: 49-57.
Gerhardsson L, Lundstrom N-G, Nordbery G, & Wall S (1986) Mortality
and lead exposure: a retrospective cohort study of Swedish smelter
workers. Br J Ind Med, 43: 707-712.
Getz LL, Haney AW, Larimore RW, McNurney JW, Leland HV, Price PW,
Rolfe GL, Wortman RL, Hudson JL, Solomon RL, & Reinbold KA (1977)
Transport and distribution in a watershed ecosystem. In: Boggess WR
ed. Lead in the environment. Springfield, Virginia, National Technical
Information Service, pp 105-133 (National Science Foundation report
No. NSF/RA-770214).
Gibson JL (1904) A plea for painted railings and painted walls of
rooms as the source of lead poisoning amongst: Queensland children.
Aust Med Gaz, 23: 149.
Gilbert SG & Rice DC (1987) Low-level lifetime lead exposure produces
behavioral toxicity (spatial discrimination reversal) in adult
monkeys. Toxicol Appl Pharmacol, 91: 484-490.
Gmerek DE, McCofferty MR, O'Neill KJ, Melamed BR, & O'Neill JJ (1981)
Effect of inorganic lead on rat brain mitochondrial respiration and
energy production. J Neurochem, 36: 1109-1113.
Goldstein GW (1990) Lead poisoning and brain cell function. Environ
Health Perspect, 89: 91-94.
Gould JH, Capar SG, Jacobs RM, & Plunkett R (1988) A quick color test
to detect lead release from glazed ceramic and enameled metalware.
Anal Lett, 21: 2145-2154.
Goyer RA (1985) Renal changes associated with lead exposure. In:
Mahaffey KR ed. Dietary and environmental lead: Human health effects.
Amsterdam, Oxford, New York, Elsevier Science Publishers, pp 315-335.
Goyer RA (1992) Nephrotoxicity and carcinogenicity of lead - An update
on exposure and effects of lead. Fundam Appl Toxicol, 18: 1-16.
Goyer RA (1993) Lead toxicity: current concerns. Environ Health
Perspect, 100: 177-187.
Goyer RA & Rhyne BC (1973) Pathological effects of lead. Int Rev Exp
Pathol, 12: 1-77.
Goyer RA & Wilson MH (1975) Lead induced inclusion bodies: results of
ethylenediamine-tetra-acetic acid treatment. Lab Invest, 32: 149-156.
Goyer RA, Leonard DL, Moore JF, Rhyne B, & Krigman MR (1970) Lead
dosage and the role of the intranuclear inclusion body. Arch Environ
Health, 20: 705-711.
Goyer RA, Falk HL, Hogan M, Feldman DD, & Richter W (1981) Renal
tumors in rats given trisodium nitrilotriacetic acid in drinking water
for 2 years. J Natl Cancer Inst, 66: 869-880.
Goyer RA, Weinberg CR, Victery WM, & Miller CR (1989) Lead induced
nephrotoxicity: kidney calcium as an indicator of tubular injury. In:
Bach PH & Lock EA ed. Nephrotoxicity. New York, London, Plenum Press,
pp 11-20.
Grandjean P (1979) Occupational lead poisoning in Denmark: screening
with the haematofluorometer. Br J Ind Med, 36: 52-58.
Grandjean P & Olsen B (1984) Lead. In: Vercruysse A ed. Techniques and
instrumentation in analytical chemistry. Volume 4: Evaluation of
analytical methods in biological systems. Part B: Hazardous metals in
human toxicology. Amsterdam, Oxford, New York, Elsevier Science
Publishers, pp 153-169.
Grandjean P, Wulf HC, & Niebuhr E (1983) Sister chromatid exchange in
response to variations in occupational lead exposure. Environ Res,
32: 199-204.
Grandjean P, Hansen ON, & Lyngbye K (1984) Analysis of lead in
circumpulpal dentin of deciduous teeth. Ann Clin Lab Sci, 14: 270-275.
Grandjean P, Hansen ON, & Lyngbye K (1986) Lead concentration in
deciduous teeth; variation related to tooth type and analytical
technique. J Toxicol Environ Health, 19: 437-445.
Grandjean P, Jensen BM, Sando SH, Jorgensen PJ, & Antonsen S (1989a)
Delayed blood regeneration in lead exposure: an effect on reserve
capacity. Am J Public Health, 79: 1385-1388.
Grandjean P, Hollnagel H, Hedegaard L, Christensen JM, & Larsen S
(1989b) Blood lead-blood pressure relations: alcohol intake and
haemoglobin as confounders. Am J Epidemiol, 129: 732-739.
Grandjean P, Lyngbye T, & Hansen ON (1991) Lessons from a Danish study
on neuropsychological impairment related to lead exposure. Environ
Health Perspect, 94: 111-115.
Granick JL, Sassa S, Granick S, Levere RD, & Kappas A (1973) Studies
in lead poisoning. II. Correlation between the ratio of activated to
inactivated aminolevulinic acid dehydratase of whole blood and the
blood lead level. Biochem Med, 8: 149-159.
Grant LD & Davis JM (1989) Effects of low-level lead exposure on
paediatric neurobehavioural development: Current findings and future
directions. In: Smith MA, Grand LD, & Sors AI ed. Lead exposure and
child development: An international Assessment. Dordrecht, London,
Kluwer Academic Publishers, pp 49-115.
Grant LD, Kimmel CA, West GL, Martinez-Vargas CM, & Howard JL (1980)
Chronic low-level lead toxicity in the rat. II. Effects on postnatal
physical and behavioral development. Toxicol Appl Pharmacol, 56:
42-58.
Graziano JH & Blum C (1991) Lead exposure from lead crystal. Lancet,
337: 141-142.
Graziano JH, Popovac D, Factor-Litvak P, Shrout P, Kline J, Murphy MJ,
Zhao Y, Mehmeti A, Ahmedi X, Rajovic B, Zvicer Z, Nenezic U, Lolacono
NJ, & Steun Z (1990) Determinants of elevated blood lead during
pregnancy in a population surrounding a lead smelter in Kosova,
Yugoslavia. Environ Health Perspect, 89: 95-100.
Graziano JH, Slavkovic V, Factor-Litvak P, Popovac D, Ahmedi X, &
Mehmeti A (1991) Depressed serum erythropoietin in pregnant women with
elevated blood lead. Arch Environ Health, 46: 347-350.
Greene T, Ernhart CB, & Boyd TA (1992) Contributions of risk factors
to elevated blood and dentine levels in preschool children. Sci Total
Environ, 115: 239-260.
Griffith JQ & Landaver MA (1944) The effect of chronic lead poisoning
on arterial blood pressure in rats. Am Heart J, 28: 295-297.
Gulson B & Wilson D (1994) History of lead exposure in children
revealed from isotopic analyses of teeth. Arch Environ Health,
49X: 279-283.
Gupta AK, Sehgal SK, Mohan M, & Anand NK (1990) Cosmetic plumbism.
Indian Paediatr, 27: 760-761.
Gustafson A, Hedmer P, Schutz A, & Skerfving S (1989) Occupational
lead exposure and pituitary function. Int Arch Occup Environ Health,
61: 277-281.
Haeger-Aronsen B (1960) Studies on urinary excretion of
delta-aminolaevulic acid and other haem precursors in lead workers and
lead-intoxicated rabbits. Scand J Clin Lab Invest, 12(47): 1.
Haenninen H, Mantere P, Hernberg S, Seppalainen AM, & Kock B (1979)
Subjective symptoms in low-level exposure to lead. Neurotoxicology,
1: 333-347.
Hakim RB, Stewart WF, Canner JK, & Tielsch JM (1991) Occupational lead
exposure and strabismus in offspring: A case control study. Am J
Epidemiol, 133: 351-356.
Hammond PB, O'Flaherty EJ, & Gartside PS (1981) The impact of air-lead
on blood-lead in man: A critique of the recent literature. Food Cosmet
Toxicol, 19: 631-638.
Hammond PB, Bornschein RL, & Succop P (1985) Dose-effect and
dose-response relationships of blood lead to erythrocytic
protoporphyrin in young children. Environ Res, 38: 187-196.
Hansen ON, Trillingsgaard A, Beese I, Lyngbye T, & Grandjean P (1989)
A neuropsychological study of children with elevated dentine lead
level: assessment of the effect of lead in different socio-economic
groups. Neurotoxicol Teratol, 11: 205-213.
Harlan WR (1988) The relationship of blood lead levels to blood
pressure in the US population. Environ Health Perspect, 78: 9-13.
Harlan WR, Landis JR, Schmouder RL, Goldstein NG, & Harlan LC (1985)
Blood lead and blood pressure: relationship in the adolescent and
adult US population. J Am Med Assoc, 253: 530-534.
Harvey PG, Hamlin MW, Kumar R, & Delves HT (1984) Blood lead,
behaviour and intelligence test performance in preschool children. Sci
Total Environ, 40: 45-60.
Harvey PG, Hamlin MW, Kumar R, Morgan J, Spurgeon A, & Delves HT
(1988) Relationships between blood lead, behaviour, psychometric and
neuropsychological test performance in young children. Br J Dev
Psychol, 6: 145-156.
Hatzakis A, Kokkevi A, Maravelias C, Katsouyanni K, Salaminios F,
Kalandidi A, Koutselinis A, Stefanis C, & Trichopoulos D (1989)
Psychometric intelligence deficits in lead-exposed children. In: Smith
MA, Grand LD, & Sors AI ed. Lead exposure and child development: An
international assessment. Dordrecht, London, Kluwer Academic
Publishers, pp 211-223.
Hawk BA, Schroeder SR, Robinson G, Otto D, Mushak P, Kleinbaum D, &
Dawson G (1986) Relation of lead and social factors to IQ of low-SES
children: a partial replication. Am J. Ment Defic, 91: 178-183.
Heard MJ & Chamberlain AC (1982) Effects of minerals and food on
uptake of lead from the gastrointestinal tract in humans. Hum Toxicol,
1: 411-416.
Hem JD & Durum WH (1973) Solubility and occurrence of lead in surface
water, J An Water Works Assoc, 65: 562-568.
Henning SJ & Cooper LC (1988) Intestinal accumulation of lead salts
and milk lead by suckling rats (42645). Proc Soc Exp Biol Med,
187: 110-116.
Hernberg S (1973) Prevention of occupational poisoning from inorganic
lead. Work Environ Health, 10: 53-61.
Hernberg S & Nikkanen J (1970) Enzyme inhibition by lead under normal
urban conditions. Lancet, 1(7637): 63-64.
Hilderbrand DC, Der R, Griffin WT, & Fahim MS (1973) Effect of lead
acetate on reproduction. Am J Obstet Gynecol, 115: 1058-1065.
Hogstedt C, Hane M, Agrell A, & Bodin L (1983) Neuropsychological test
results and symptoms among workers with well-defined long term
exposure to lead. Br J Ind Med, 49: 99-105.
Holness DL & Nethercott JR (1988) Acute lead intoxication in a group
of demolition workers. Appl Ind Hyg, 3: 338-341.
Holtzman D, Shen Hgu J, & Montell P (1978) In vitro effects of
inorganic lead on isolated rat brain mitochondrial respiration.
Neurochem Res, 3: 195-206.
Holtzman D, Shen Hgu J, & Desautel M (1981) Absence of effects of lead
feedings and growth retardation on mitochondrial and microsomal
cytochrome in the developing brain. Toxicol Appl Pharmacol, 58: 48-56.
Holtzman D, Devries C, Nguyan H, Olson J, & Bensch K (1984) Maturation
of resistance to lead encephalopathy: cellular and subcellular
mechanisms. Neurotoxicology, 5: 97-124.
Hopper JL, Balderas A, & Mathews JO (1982) Analysis of variation in
blood lead levels in Melbourne families. Med J Aust, 2: 573-576.
Huang J, He F, Yigun W, & Zhang S (1988a) Observations on renal
function in workers exposed to lead. Sci Total Environ, 71: 535-537.
Huang XP, Feng ZY, Zhai WL, & Xu JH (1988b) Chromosomal aberrations
and sister chromatid exchanges in workers exposed to lead. Biomed
Environ Sci, 1: 382-387.
Huber F & Schmidt U (1976) Methylation of organolead and lead (II)
compounds to C3 4Pb by microorganisms. Nature (Lond),
259: 157-158.
Hutton M, Wadge A, & Milligan PJ (1988) Environmental levels of
cadmium and lead in the vicinity of a major refuse incinerator. Atmos
Environ, 22: 411-416.
IAEA (1987) Co-ordinated research programme on human daily dietary
intakes of nutritionally important trace elements as measured by
nuclear and other techniques. IAEA Newl, 2: 6-15.
IARC (1980) Lead and lead compounds. In: Some metals and metallic
compounds. Lyon, International Agency for Research on Cancer, pp
325-416 (IARC Monographs on the Evaluation of the Carcinogenic Risk of
Chemicals to Humans, Volume 23).
IARC (1987a) Genetic and related effects: An updating of selected IARC
Monographs from Volumes 1 to 42. Lyon, International Agency for
Research on Cancer (IARC Monographs on the Evaluation of the
Carcinogenic Risk of Chemicals to Humans, Supplement 6).
IARC (1987b) Lead and lead compounds. In: Overall evaluations of
carcinogenicity: an updating of IARC Monographs, Volumes 1 to 42.
Lyon, International Agency for Research on Cancer, pp 230-232 (IARC
Monographs on the Evaluation of the Carcinogenic Risk of Chemicals to
Humans, Supplement 7).
Ibels LS & Pollock CA (1986) Toxicology management review. Lead
intoxication. Med Toxicol, 1: 387-410.
Ichiba M & Tomokuni K (1990) Studies on erythrocyte pyrimidine
5'-nucleotidase test and its evaluation in workers occupationally
exposed to lead. Int Arch Occup Environ Health, 62: 305-310.
ILZSG (International Lead and Zinc Study Group) (1992) Lead and zinc
statistics. Int Lead Zinc Study Group Bull, 32: 3.
IPCS (1977) Environmental health criteria 3: Lead. Geneva, World
Health Organization, 160 pp.
IPCS (1989) Environmental health criteria 85: Lead - Environmental
effects. Geneva, World Health Organization, 106 pp.
IPCS (1994) Environmental health criteria 155: Biomarkers and risk
assessment: Concepts and principles. Geneva, World Health
Organization, 82 pp.
IRPC (International Radiological Protection Commission), Task Group on
Lung Dynamics (1966) Deposition and retention models for internal
dosimetry of the human respiratory trace. Health Phys, 12: 173-207.
IRPTC (1987) IRPTC legal file 1986. Geneva, International Register of
Potentially Toxic Chemicals, United Nations Environment Programme,
vol 1.
Irwig LM, Harrison WO, Rocks P, Webster I, & Andrew M (1978) Lead and
morbidity: a dose response relationship. Lancet, 2(8079): 4-7.
Jaakkola J & Anttila A (1992) [Occupational exposures to chemical
agents in Finland.] Helsinki, Institute of Occupational Health and
Finnish Work Environmental Fund (in Finnish).
James AC (1978) Lung deposition of submicron aerosols calculated as a
function of age and breathing rate. In: National Radiological
Protection Board Annual Report. Harwell, UK, National Radiological
Protection Board, pp 71-75.
Jaremin B (1983) Blast lymphocyte transformation (LTT), rosette
(E-RFC) and leukocyte migration inhibition (MIF) tests in persons
exposed to the action of lead during work. Report II. Bull Inst Marit
Trop Med (Gdynia), 34: 187-197.
Jensen AA (1984) Metabolism and toxicokinetics. In: Grandjean P ed.
Biological effects of organolead compounds. Boca Raton, Florida, CRC
Press, chapter 8, pp 97-115.
Johnson NE & Tenuta K (1979) Diets and lead blood levels of children
who practice pica. Environ Res, 18: 369-376.
Jorhem L & Slorach S (1988) Lead, chromium, tin, iron, and cadmium in
foods in welded cans. Food Addit Contam, 4: 309-316.
Jorhem L, Mattsson P, & Slorach S (1988) Lead in table wines on the
Swedish market. Food Addit Contam, 5: 645-649.
Kaminsky P, Leone J, & Duc M (1988) Incidence du saturnisme hydrique
dans un service de médecine interne en région de sols acides. Presse
Méd, 17: 419-422.
Kampe W (1983) Lead and cadmium in food - is there an actual danger?
Forum Staedte-Hyg, 34: 236-241.
Kang HK, Infante PF, & Carra JS (1980) Occupational lead exposure and
cancer (letter). Science, 207: 935-936.
Katagiri Y, Toriumi H, & Kawai M (1983) Lead exposure among 3-year-old
children and their mothers living in a pottery-producing area. Int
Arch Occup Environ Health, 52: 223-229.
Kaye WE, Novotny TE, & Tucker M (1987) New ceramics-related industry
implicated in elevated blood lead levels in children. Arch Environ
Health, 42: 161-164.
Kazantzis G (1989) Lead: ancient metal - modern menace. In: Smith MA,
Grant LD, & Sors AI ed. Lead exposure and child development: An
international assessment. Dordrecht, London, Kluwer Academic Press,
pp 119-128.
Kehoe RA (1961) The metabolism of lead in man in health and disease:
The normal metabolism of lead (The Harben Lectures, 1960). J R Inst
Public Health Hyg, 24: 81-97.
Kehoe RA (1987) Studies of lead administration and elimination in
adult volunteers under natural and experimentally induced conditions
over extended periods of time. Food Chem Toxicol, 25: 425-493.
Keller CA & Doherty RA (1980a) Distribution and excretion of lead in
young and adult female mice. Environ Res, 21: 217-228.
Keller CA & Doherty RA (1980b) Bone lead mobilization in lactating
mice and lead transfer to suckling offspring. Toxicol Appl Pharmacol,
55: 220-228.
Kerin Z (1973) Lead in new-fallen snow near a lead smelter. Arch
Environ Health, 26: 256-260.
Kholil-Manesh F, Gonich HC, Cohen AH, Alinovi R, Berganaschi E, Mutti
A, & Rosen VJ (1992) Experimental model of lead nephrotoxicity. I.
Continuous high-dose lead administration. Kidney Int, 41: 1192-1203.
Kimber I, Stonard MD, Gidlow DA, & Niewola Z (1986) Influence of
chronic low-level exposure to lead on plasma immunoglobulin
concentration and cellular immune function in man. Int Arch Occup
Environ Health, 57: 117-125.
King E & Conchie D (1979) Industrial lead absorption. Ann Occup Hyg,
22: 213-239.
Kirkby H & Gyntelberg F (1985) Blood pressure and other cardiovascular
risk factors of long term exposure to lead. Scand J Work Environ
Health, 11: 15-19.
Kocak R, Anarat A, Altintas G, & Evilyaoglu N (1989) Lead poisoning
from contaminated flour in a family of 11 members. Hum Toxicol,
8: 385-386.
Kolbye AC, Mahaffey R, Fiorino A, Corneliussen PC, & Jelinek CF (1974)
Food exposures to lead. Environ Health Perspect, 7(Exp Issue): 65-75.
Koller LD (1985) Immunological effects of lead. In: Mahaffey KR ed.
Dietary and environmental lead: Human health effects. Amsterdam,
Oxford, New York, Elsevier Science Publishers, pp 339-354.
Koller LD & Roan JG (1980) Effect of lead, cadmium and methylmercury
on immunologic memory. J Environ Pathol Toxicol, 4: 47-52.
Koo WWK, Succop PA, Bornschein RL, Krug-Wispe SK, Steichen JJ, Tsang
RC, Hammond PB, & Berger OG (1991) Serum vitamin D metabolites and
bone mineralization in young children with chronic to moderate lead
exposure. Pediatrics, 87: 680-687.
Kopp SJ, Barron JT, & Tow JP (1988) Cardiovascular actions of lead and
relationship to hypertension: a review. Environ Health Perspect,
89: 91-99.
Koren G, Chang N, Gonen R, Klein J, Weiner L, Demshar H, Pizzolato S,
Radde I, & Shime J (1990) Lead-exposure among mothers and their
newborns in Toronto. Can Med Assoc J, 142: 1241-1244.
Kostial K & Kello D (1979) Bioavailability of lead in rats fed "human"
diets. Bull Environ Contam Toxicol, 21: 312-314.
Kostial K, Kello D, & Jugo S (1978) Influence of age on metal
metabolism and toxicity. Environ Health Perspect, 25: 81-86.
Kovar IZ, Strehlow CD, Richmond J, & Thompson MG (1984) Perinatal lead
and cadmium burden in a British urban population. Arch Dis Child,
59: 36-39.
Krueger JA & Duguay KM (1989) Comparative analysis of lead in Maine
urban soils. Bull Environ Contam Toxicol, 42: 574-581.
Kusell M, Lake L, Anderson M, & Gerschenson LE (1978) Cellular and
molecular toxicology of lead. II. Effect of lead on d-aminolevulinc
acid synthelase of cultured cells. J Toxicol Environ Health,
4: 515-525.
Kutbi II, Ahmed M, & Saber A (1989) Measurement of blood-lead levels
in school children of Jeddah Saudi Arabia and assessment of sub-toxic
levels of lead on some sensitive hematological parameters. J Environ
Sci Health, A24: 943-955.
Lacey RF, Moore MR, & Richards WN (1985) Lead in water, infant diet
and blood: The Glasgow duplicate diet study. Sci Total Environ,
41: 235-257.
Lai CS (1972) Lead poisoning as an occupational hazard in Chinese
opera actors: a case report. Singap Med J, 13: 115-117.
Landis JR & Flegal KM (1988) A generalized Mantel-Haenszel analysis of
the regression of blood pressure on blood lead using NHANES II data.
Environ Health Perspect, 78: 35-41.
Landrigan PJ, Gehlbach SH, Rosenblum BF, Shoults JM, Cancelaria RM,
Barthel WF, Liddle JA, Smrek AL, Staehling NW, & Sanders JF (1975)
Epidemic lead absorption near an ore smelter; The role of particulate
lead. N Engl J Med, 292: 123-129.
Lansdown RG, Shepherd J, Clayton BE, Delves HT, Graham PJ, & Turner WC
(1974) Blood-lead levels, behaviour, and intelligence: a population
study. Lancet, 1(7857): 538-541.
Lansdown R, Yule W, Urbanowicz MA, & Hunter J (1986) The relationship
between blood-lead concentrations, intelligence, attainment and
behaviour in a school population: the second London study. Int Arch
Occup Environ Health, 57: 225-235.
Laurs AJ (1976) Review of European test methods for measuring lead and
cadmium release. In: Proceedings of the International Conference on
Ceramic Foodware Safety, Geneva, 1974. New York, Lead Industries
Association Inc., pp 27-36.
Lauwerys R, Buchet JP, Roels HA, & Materne DD (1974) Relationship
between urinary delta-aminolevulinic acid excretion and the inhibition
or red cell delta-aminolevulinate dehydratase by lead. Clin Toxicol,
7: 383-388.
Lavelle JM, Poppenga RH, Thacker BJ, Geisy JP, Weis C, Othoudt R, &
Vandervoort C (1991) Bioavailability of lead in mining wastes: An oral
intubation study in young swine. Chem Speciation Bioavailab,
3: 105-111.
Laxen DPH, Raab GM, & Fulton M (1987) Children's blood lead and
exposure to lead in household dust and water - a basis for an
environmental standard for lead in dust. Sci Total Environ,
66: 235-244.
Lee RE & Goranson S (1972) National air surveillance cascade impact or
network. I. Size distribution measurements of suspended particulate
matter in air. Environ Sci Technol, 6: 1019-1024.
Leeming RJ & Blair JA (1980) The effect of pathological and normal
physiological processes on biopterin levels in man. Clin Chim Acta,
108: 102-111.
Lepow ML, Bruckman L, Rubino RA, Markowitz S, Gillette M, & Kapish J
(1974) Role of airborne lead in increased body burden of lead in
Hartford children. Environ Health Perspect, 7: 99-102.
Lerda D (1992) Study of sperm characteristics in persons
occupationally exposed to lead. Am J Ind Med, 22: 567-571.
Levin ED & Bowman RE (1983) The effect of pre- or postnatal lead
exposure on Hamilton search task in monkeys. Neurobehav Toxicol
Teratol, 5: 391-394.
Levin ED & Bowman RE (1986) Long-term lead effects on the Hamilton
Search Test and delayed alternation in monkeys. Neurobehav Toxicol
Teratol, 8: 219-224.
Levin ED & Bowman RE (1989) Long-term effects of chronic post-natal
lead exposure on delayed spatial alternation in monkeys. Neurotoxicol
Teratol, 10: 505-510.
Lilienthal H, Winneke G, Brockhaus A, & Molik B (1986) Pre- and
postnatal lead-exposure in monkeys: Effects on activity and learning
set formation. Neurobehav Toxicol Teratol, 8: 265-272.
Lilienthal H, Winneke G, & Ewert T (1990) Effects of lead on
neurophysiological and performance measures: animal and human data.
Environ Health Perspect, 89: 21-25.
Lilis R (1981) Long-term occupational lead exposure, chronic
nephropathy, and renal cancer: a case report. Am J Ind Med,
2: 293-297.
Lilis R, Fischbein A, Eisinger J, Blumberg WE, Diamond S, Anderson HA,
Rom W, Rice C, Sarkozi L, Kon S, & Selikoff IJ (1977) Prevalence of
lead disease among secondary lead smelter workers and biological
indicators of lead exposure. Environ Res, 14: 255-285.
Lilley SG, Florence TM, & Stauber JL (1988) The use of sweat to
monitor lead absorption through the skin. Sci Total Environ,
76: 267-278.
Lind B, Vahter M, Rahnster B, & Björs U (1988) Quality control samples
for the determination of lead and cadmium in blood, feces, air
filters, and dust. Fresenius Z Anal Chem, 332: 741-743.
Lindholm ML, Sallmen M, Anttila A, Taskinen H, & Hemminki K (1991)
Paternal occupational lead exposure and spontaneous abortion. Scand J
Work Environ Health, 17(2): 95-103.
Lin-Fu JS (1985) Historical perspective on health effects of lead. In:
Mahaffey KR ed. Dietary and environmental lead: Human health effects.
Amsterdam, Oxford, New York, Elsevier Science Publishers, pp 43-64.
Lovering TG ed. (1976) Lead in the environment. Washington, DC, US
Department of the Interior, Geological Survey (Geological Survey
Professional Paper No. 957).
Lyngbye T, Hansen OL, Trillingsgaard A, Beese I, & Grandjean P (1990a)
Learning disabilities in children: significance of low-level
lead-exposure and confounding factors. Acta Paediatr Scand,
79: 352-360.
Lyngbye T, Jorgensen PJ, & Grandjean P (1990b) Validity and
interpretation of blood lead levels: a study of Danish
school-children. Scand J Clin Lab Invest, 50: 441-449.
McGrath SP (1986) The range of metal concentrations in top soils of
England and Wales in relation to soil protection guidelines. In
Hemphill DD ed. Trace substances in environmental health, vol 20.
Columbia, Missouri, University of Missouri, pp 242-252.
McGregor AJ & Mason MJ (1990) Chronic occupational lead exposure and
testicular endocrine function. Hum Exp Toxicol, 9: 371-376.
McIntosh MJ, Meredith PA, Moore MR, & Goldberg A (1985) Neurotoxic
action of lead: effect on tetrahydrobiopterin metabolism in the rat.
Comp Biochem Physiol, 81C(1): 227-231.
McIntosh MJ, Meredith PA, Moore MR, & Goldberg A (1989) Action of lead
on neurotransmission in rats. Xenobiotica, 19(1): 101-113.
McKhann CF & Vogt EC (1933) Lead poisoning in children. J Am Med
Assoc, 101: 1131-1135.
McMichael AJ & Johnson HM (1982) Long-term mortality profile of
heavily-exposed lead smelter workers. J Occup Med, 24: 375-378.
McMichael AJ, Vimpani GV, Robertson EF, Baghurst PA, & Clark PD (1986)
The Port Pirie cohort study: maternal blood lead and pregnancy
outcome. J Epidemiol Community Health, 41: 18-25.
McMichael AJ, Baghurst PA, Wigg NR, Vimpani GV, Robertson EF, &
Roberts RJ (1988) Port Pirie cohort study: environmental exposure to
lead and children's abilities at the age of four years. N Engl J Med,
319: 468-475.
Maenhaut W, Zoller WH, Duce RA, & Hoffman GL (1979) Concentration and
size distribution of particulate trace elements in the south polar
atmosphere. J Geophys Res, 84: 2421-2431.
Mahaffey KR (1985) Factors modifying susceptibility to lead toxicity.
In: Mahaffey KR ed. Dietary and environmental lead: Human health
effects. Amsterdam, Oxford, New York, Elsevier Science Publishers,
pp 373-419.
Mahaffey KR & Annest JL (1986) Association of erythrocyte
protoporphyrin with blood lead level and iron status in the Second
National Health and Nutrition Examination Survey, 1976-1980. Environ
Res, 41: 327-338.
Mahaffey-Six KR & Goyer RA (1972) The influence of iron deficiency on
tissue content and toxicity of ingested lead in the rat. J Lab Clin
Med, 79: 128-136.
Mahaffey KR, Rosen JF, Chesney RW, Peeler JT, Smith CM, & Debuca HF
(1982) Association between age, blood lead concentration, and serum
1,25-dihydroxy-cholecalciferol levels in children. Am J Clin Nutr,
35: 1327-1331.
Maines MD (1992) Haem oxygenase-clinical applications and functions.
Boca Raton, Florida, CRC Press.
Maki-Paakkanen J, Sorsa M, & Vainio H (1981) Chromosome aberrations
and sister chromatid exchanges in lead-exposed workers. Hereditas,
94: 269-275.
Manton WI (1985) Total contribution of airborne lead to blood lead. Br
J Ind Med, 42: 168-172.
Manton WI & Cook JD (1984) High accuracy (stable isotope dilution)
measurements of lead in serum and cerebrospinal fluid. Br J Ind Med,
41: 313-319.
Manton WI & Thal ER (1986) Lead poisoning from retained missiles: an
experimental study. Ann Surg, 204: 594-599.
Mao P & Molnar JJ (1967) The fine structure and histochemistry of
lead-induced renal tumors in rats. Am J Pathol, 50: 571-603.
Maranelli G & Apostoli P (1987) Assessment of renal function in lead
poisoned workers. In: Occupational and environmental chemical hazards:
Cellular and biochemical indices for monitoring toxicity. Chichester,
Ellis Horwood Ltd, chapter 42, pp 344-348.
Marcus AH (1985a) Testing alternative nonlinear kinetic models in
compartmental analysis. In: Eisenfield J & Delisi C ed. Mathematics
and computers in biomedical applications. Amsterdam, Oxford, New York,
Elsevier Science Publishers, pp 259-267.
Marcus AH (1985b) Multicompartment kinetic models for lead: Part I.
bone kinetics and variable absorption in humans without excessive lead
exposures. Environ Res, 36: 441-458.
Marcus AH (1985c) Multicompartment kinetic models for lead: Part II.
linear kinetics and variable absorption in humans without excessive
lead exposures. Environ Res, 36: 459-472.
Marcus AH & Schwartz J (1987) Dose-response curves for erythrocyte
protoporphyrin vs blood lead: effects of iron status. Environ Res,
44: 221-227.
Marino PE, Franzblau A, Lilis R, & Landrigan PJ (1989) Acute lead
poisoning in construction workers: the failure of protective
standards. Arch Environ Health, 44: 140-145.
Marks SC & Popoff SN (1988) Bone cell biology: the regulation of
development, structure, and function in the skeleton. Am J Anat,
183: 1-44.
Marlowe M & Errrera J (1982) Low lead levels and behaviour problems in
children. Behav Disord, 7: 163-172.
Matte TD, Figueroa JP, Ostrowski S, Burr G, Jackson-Hunt L, Kennylside
RA, & Baker EL (1989a) Lead poisoning among household members exposed
to lead-acid battery repair shops in Kingston, Jamaica. Int J
Epidemiol, 18: 874-881.
Matte TD, Figueroa JP, Burr G, Flesch JP, Keenlyside RA & Baker EL
(1989b) Lead exposure among lead-acid battery workers in Jamaica. Am J
Ind Med, 16: 167-177.
Meredith PA & Moore MR (1979) The influence of lead on haem
biosynthesis and biodegradation in the rat. Biochem Soc Trans,
7: 637-639.
Meredith PA, Moore MR, Campbell BC, Thompson GG, & Goldberg A (1978)
Delta-aminolevulinic acid metabolism in normal and lead-exposed
humans. Toxicology, 9: 1-9.
Merwin BW (1976) Review of US standards on test methods for lead and
cadmium release. In: Proceedings of the International Conference on
Ceramic Foodware Safety, Geneva, 1974. New York, Lead Industries
Associate Inc., pp 36-40.
Meyer BR, Fishbein A, Rosemann K, Lerman Y, Drayer DE, & Reidenberg MM
(1984) Increased urinary enzyme excretion in workers exposed to
nephrotoxic chemicals. Am J Med, 76: 989-998.
Michaelson A & Sauerhoff MW (1974) An improved model of lead induced
brain dysfunction in the suckling rat. Toxicol Appl Pharmacol,
28: 88-96.
Mielke H, Blake B, Burroughs S, & Hassinger N (1984) Urban lead levels
in Minneapolis: the case of the Hmong children. Environ Res,
34: 64-76.
Mitchell DG, Aldous KM, & Ryan FJ (1974) Mass screening for lead
poisoning, capillary blood sampling and automated Delves cup atomic
absorption analysis. NY State J Med, 74: 1599-1603.
Mitchell-Heggs CAW, Conway M, & Cassar J (1990) Herbal medicine as a
cause of combined lead and arsenic poisoning. Hum Exp Toxicol,
9: 195-196.
Molina-Ballesteros G, Zuniga-Charles MA, Ortega AC, & Solis-Camara P
(1983) Lead concentrations in the blood of children from
pottery-making families exposed to lead salts in a Mexican village.
Bull Panam Health Organ, 17: 35-41.
Möller L & Kristensen TS (1992) Blood lead as a cardiovascular risk
factor. Am J Epidemiol, 136: 1091-1100.
Moore MR (1983) Lead exposure and lead plumbosolvency. In: Rutter M &
Jones RR ed. Lead versus health. London, John Wiley and Sons,
pp 79-106.
Moore MR & Meredith PA (1979) The carcinogenicity of lead. Arch
Toxicol, 42: 87-94.
Moore MR, Meredith PA, Campbell BC, & Watson SW (1979) The
gastrointestinal absorption of lead 203 chlorine in man. In: Hemphill
DD ed. Trace substances in environmental health - XIII. Proceedings of
the 13th Annual Conference on Trace Substances in Environmental
Health, Columbia, June 1979. Columbia, Missouri, University of
Missouri, pp 368-373.
Moore MR, Meredith PA, & Goldberg A (1980) Lead and heme biosynthesis.
In: Singhal PL & Thomas JA ed. Lead toxicity. Baltimore, Maryland,
Urban and Schwarzenberg, Inc., pp 79-118.
Moore MR, Goldberg A, & Pocock SJ (1982) Some studies of maternal and
infant lead exposure in Glasgow. Scott Med J, 27: 113-122.
Moore MR, Bushnell IWR, & Goldberg A (1989) A prospective study of the
results of changes in environmental lead exposure in children in
Glasgow. In: Smith MA, Grant LD, & Sors AI ed. Lead exposure and child
development: An international assessment. Dordrecht, London, Kluwer
Academic Publishers, pp 371-378.
Moore MR, Goldberg A, & Young-Laiwah AAC (1987) Lead effects on the
haem-biosynthetic pathway. Relationship to toxicity. Ann NY Acad Sci,
514: 191-203.
Mooty J, Ferrand CF, & Harris P (1975) Relationship of diet to lead
poisoning in children. Pediatrics, 55: 636-639.
Moreau T, Orssaud G, Juguet B, & Busquet G (1982) Plombémie et
pression artérielle: premiers résultats d'une enquête transversale de
431 sujets de sexe masculin. Rev Epidemiol Santé Publique,
30: 395-397.
Morgan A & Holmes A (1978) The fate of lead in petrol-engine exhaust
particulates inhaled by the rat. Environ Res, 15: 44-56.
Muijser H, Hoogendijk EMG, Hooisma J, & Twisk DAM (1987) Lead exposure
during demolition of a steel structure coated with lead-based paints.
II. Reversible changes in the conduction velocity of the motor nerves
in transiently exposed workers. Scand J Work Environ Health,
13: 56-61.
Munoz C, Garbe K, Lilienthal H, & Winneke G (1986) Persistence of
retention deficit in rats after neonatal lead exposure.
Neurotoxicology, 7(2): 569-580.
Muramatsu Y & Parr RM (1985) Survey of currently available reference
materials for use in connection with the determination of trace
elements in biological and environmental materials. Vienna,
International Atomic Energy Agency (Report No. IAEA/RL/128).
Murata K & Araki S (1991) Autonomic nervous system dysfunction in
workers exposed to lead, zinc, and copper in relation to peripheral
nerve conduction: a study of R-R interval variability. Am J Ind Med,
20: 663-671.
Murphy MJ, Graziano JH, Popovac D, Kline JK, Mehmeti A, Factor-Litvak
P, Ahmadi G, Shrout P, Rajovic B, Nenezic DU, & Stein ZA (1990) Past
pregnancy outcomes among women living in the vicinity of a lead
smelter in Kosovo, Yugoslavia. Am J Public Health, 80: 33-35.
Mushak P (1989) Biological monitoring of lead exposure in children:
overview of selected biokinetic and toxicological issues. In: Smith
MA, Grant LD, & Sors AI ed. Lead exposure and child development: An
international assessment. Dordrecht, London, Kluwer Academic
Publishers, pp 129-145.
Mushak P & Crocetti AF (1989) Determination of numbers of lead-exposed
American children as a function of lead source: Integrated summary of
a report to the U.S. Congress on childhood lead poisoning. Environ
Res, 50: 210-299.
Mussalo-Rauhamaa H, Salmela SS, Leppanen A, & Pyysalo H (1986)
Cigarettes as a source of some trace and heavy metals and pesticides
in man. Arch Environ Health, 41: 49-55.
NAS-NRC (1972) Airborne lead in perspective. Washington, DC, National
Academy of Sciences.
Needleman HL & Gatsonis CA (1990) Low-level lead exposure and the IQ
of children: a meta-analysis of modern studies. J Am Med Assoc,
263: 673-678.
Needleman HL, Tuncay OC, & Shapiro IM (1972) Lead levels in deciduous
teeth of urban and suburban children. Nature (Lond), 235: 111-112.
Needleman HL, Gunnoe C, Leviton A, Reed R, Peresie H, Maher C, &
Barrett P (1979) Deficits in psychologic and classroom performance of
children with elevated dentine lead levels. N Engl J Med,
300: 689-695.
Needleman HL, Schell A, Bellinger D, Leviton A, & Allred EN (1990) The
long-term effects of childhood exposure to low doses of lead: An
11-year follow-up report. N Engl J Med, 322: 83-88.
Neri LC, Hewitt D, & Orser B (1988) Blood lead and blood pressure:
analysis of cross-sectional and longitudinal data from Canada. Environ
Health Perspect, 78: 123-126.
Nerin C, Olavide S, & Cacho J (1989) Determination of lead in airborne
particulate by hydride generation. Water Air Soil Pollut, 44: 339-345.
Neubecker TA & Allen HE (1983) The measurement of complexation
capacity and conditional stability constants for ligands in natural
waters. Water Res, 17: 1-14.
NFA (Australian National Food Authority, National Health and Medical
Council) (1991) The 1990 Australian market basket survey report.
Canberra, Commonwealth of Australia, Australian Government Publishing
Service.
Ng A & Patterson CC (1982) Changes of Pb and Ba with time in
California offshore basin sediments. Geochim Cosmochim Acta,
46: 2307-2322.
Ng TP, Goh HH, Ng YL, Ong CN, Chia KS, Chia SE, & Jeyaratnam J (1991)
Male endocrine functions in workers with moderate exposure to lead. Br
J Ind Med, 48: 485-491.
Nielsen T (1984) Atmospheric occurrence of organolead compounds. In:
Grandjean P ed. Biological effects of organolead compounds. Boca
Raton, Florida, CRC Press, pp 43-62.
Nilsson U, Attewell R, Christoffersson J-O, Schütz A, Ahlgren L,
Skerfving S, & Mattsson S (1991) Kinetic of lead in bone and blood
after end of occupational exposure. Pharmacol Toxicol, 69: 477-484.
NIOSH (1977a) Method No. P&CAM 102. In: Manual of analytical methods,
vol 1, 2nd ed. Cincinnati, Ohio, National Institute for Occupational
Safety and Health.
NIOSH (1977b) Method No. P&CAM 191. In: Manual of analytical methods,
vol 1, 2nd ed. Cincinnati, Ohio, National Institute for Occupational
Safety and Health, pp 191/1-191/9.
NIOSH (1977c) Method No. P&CAM 195. In: Manual of analytical methods,
vol 1, 2nd ed. Cincinnati, Ohio, National Institute for Occupational
Safety and Health.
NIOSH (1977d) Method No. P&CAM 200. In: Manual of analytical methods,
vol 1, 2nd ed. Cincinnati, Ohio, National Institute for Occupational
Safety and Health, pp 200/1-200/8.
NIOSH (1977e) Method No. P&CAM 214. In: Manual of analytical methods,
vol 1, 2nd ed. Cincinnati, Ohio, National Institute for Occupational
Safety and Health, pp 214/1-214/6.
NIOSH (1981) Method No. P&CAM 351. In: Manual of analytical methods,
vol 7. Cincinnati, Ohio, National Institute for Occupational Safety
and Health, pp 351/1-351/11.
Nordenson I, Beckman G, Beckman L, & Nordstrom S (1978) Occupational
and environmental risks in and around a smelter in northern Sweden:
IV. Chromosomal aberrations in workers exposed to lead. Hereditas,
88: 263-267.
Nordstrom S, Beckman L, & Nordenson I (1978a) Occupational and
environmental risks in and around a smelter in northern Sweden: I.
Frequencies in birth weight. Hereditas, 88: 43-46.
Nordstrom S, Beckman L, & Nordenson I (1978b) Occupational and
environmental risks in and around a smelter in northern Sweden: III.
Frequencies of spontaneous abortion. Hereditas, 88: 51-54.
Nordstrom S, Beckman L, & Nordenson I (1979) Occupational and
environmental risks around a smelter in northern Sweden: V.
Spontaneous abortion among female employees and decreased birth weight
in their offspring. Hereditas, 90: 291-296.
Nosal RM & Wilhelm WJ (1990) Lead toxicity in the shipbreaking
industry: the Ontario experience. Can J Public Health, 81: 259-262.
Novotny T, Cook M, Hughes J, & Lee SA (1987) Lead exposure in a firing
range. Am J Public Health, 77: 1225-1226.
Nriagu J (1979) Global inventory of natural and anthropogenic
emissions of trace metals to the atmosphere. Nature (Lond),
279: 409-411.
Nriagu J (1989) A global assessment of natural sources of atmospheric
trace metals. Nature (Lond), 338: 47-49.
Nriagu J & Pacyna JM (1988) Quantitative assessment of worldwide
contamination of air, water, and soils by trace metals. Nature (Lond),
333: 134-139.
OCTEL (1991) Worldwide survey of motor gasoline quality. London,
Associated Octel Company Ltd (Octel Promotional Publication No. 4).
OECD (1993) Risk Reduction Monograph No. 1: Lead background and
national experience with reducing risk. Paris, Organisation for
Economic Co-operation and Development, 277 pp (Report No.
OCDE/GD(93)67).
O'Flaherty EJ (1991) Physiologically based lead kinetics. Trace Subst
Environ Health, 24: 44-54.
O'Flaherty EJ (1993) Physiologically based models for bone-seeking
elements. IV. Kinetics of lead disposition in humans. Toxicol Appl
Pharmacol, 118: 16-29.
O'Flaherty EJ, Hammond PB, Lerner SI, Hanenson IB, & Roda SMB (1980)
The renal handling of delta-aminolevulinic acid in the rat and in the
human. Toxicol Appl Pharmacol, 55: 423-432.
O'Flaherty EJ, Hammond PB, & Lerner SI (1982) Dependence of apparent
blood lead half-life on the length of previous lead exposure in
humans. Fundam Appl Toxicol, 2: 49-54.
Olejnik D, Walkowska A, Wisnilwska J, & Ziembinski R (1985)
[Evaluation of the daily dose of mercury, lead and cadmium in the diet
of certain population groups.] Roczn Pzh, XXXVI: 9-21 (in Polish, with
English abstract).
Olson KW & Skogerboe RK (1975) Identification of soil lead compounds
from automotive sources. Environ Sci Technol, 9: 227-230.
Ong CN & Lee WR (1980) High affinity of lead for fetal hemoglobin. Br
J Ind Med, 37: 292-298.
Ong CN, Phoon WO, Law HY, Tye CY, & Lim HH (1985) Concentration of
lead in maternal blood, cord blood, and breast milk. Arch Dis Child,
60: 576-759.
Ong CN, Endo G, & Chia KS (1987) Evaluation of renal function in
workers with low blood lead levels. In: Occupational and environmental
chemical hazards: Cellular and biochemical indices for monitoring
toxicity. Chichester, Ellis Horwood Ltd, chapter 39, pp 327-333.
Orfila MP (1814) Traité des poisons. Paris, Crochard, vol 2.
Orssaud G, Claude JR, Moreau T, Lellouch J, Juguet B, & Festy B (1985)
Blood lead concentration and blood pressure. Br Med J, 290: 244.
Padilla F, Shapiro AP, & Jensen WN (1969) Effect of chronic lead
intoxication on blood pressure in the rat. Am J Med Sci, 258: 359-365.
Paglia DE, Valentine WN, & Dahlgren JG (1975) Effects of low-level
lead exposure on pyrimidine 5'-nucleotidase and other erythrocyte
enzymes: possible role of pyrimidine 5'-nucleotidase in the
pathogenesis of lead-induced anaemia. J Clin Invest, 56: 1164-1169.
Pagliuca A, Mufti GJ, Baldwin D, Lestas AN, Wallis RM, & Bellingham AJ
(1990) Lead poisoning: clinical, biochemical, and haematological
aspects of a recent outbreak. J Clin Pathol, 43: 277-281.
Parfitt AM (1990) Pharmacologic manipulation of bone remodelling and
calcium homeostasis: Calcium metabolism. Prog Basic Clin Pharmacol,
4: 1-27.
Parkinson DK, Ryan C, Brommet EJ, & Connell MM (1986) A psychiatric
epidemiologic study of occupational lead exposure. Am J Epidemiol,
123: 261-269.
Parkinson DK, Ryan C, Bromet J, & Connell MM (1987) A psychiatric
epidemiological study of occupationally exposure. Am J Epidemiol,
123: 261-269.
Patterson C (1965) Contaminated and natural lead environments of man.
Arch Environ Health, 11: 344-360.
Patterson CC (1983) Criticism of "flow of metals into the global
atmosphere". Geochim Cosmochim Acta, 47: 1163-1168.
Patterson CC & Settle DM (1976) The reduction of orders of magnitude
errors in lead analyses of biological materials and natural waters by
evaluating and controlling the extent and source of industrial lead
contamination introduced during sample collecting, handling, and
analyses. In: LaFleur PD ed. Accuracy in trace analysis: Sampling,
sample handling and analysis. Volume 1. Proceedings of the 7th
Materials Research Symposium, Gaithersburg, Maryland, October, 1974.
Washington, DC, US Department of Commerce, National Bureau of
Standards, pp 321-352 (NBS Special Publication No. 422).
Pekkarinen M (1970) Methodology in the collecting of food consumption
data. World Rev Nutr Diet, 12: 145-170.
Pentschew A (1965) Morphology and morphogenesis of lead
encephalopathy. Acta Neuropathol, 5: 133-160.
Pentschew A & Garro F (1966) Lead encephalo-myelopathy of the suckling
rat and its implications on the porphyrinopathic nervous diseases with
special reference to the permeability/disorders of the nervous
system's capillaries. Acta Neuropathol, 6: 266-278.
Perino J & Ernhart CB (1974) The relation of subclinical lead level to
cognitive and sensorimotor impairment in black preschoolers. J Learn
Disabil, 7: 616-620.
Pickston L, Brewerton HV, Drysdale JM, Hughes JT, Smith JM, Love JL,
Sutcliffe ER, & Davidson F (1985) The New Zealand diet: a survey of
elements, pesticides, colours and preservatives. N Z J Technol,
1: 81-89.
Piomelli S, Seaman C, Zullow D, Curran A, & Davidow B (1982) Threshold
for lead damage to heme synthesis in urban children. Proc Natl Acad
Sci (USA), 79: 3335-3339.
Piper W & Tephly T (1974) Differential inhibition of erythrocyte and
hepatic uroporphyrinogen. I. Synthelase activity by lead. Life Sci,
14: 873-876.
Pirkle JL, Schwartz J, Lamdis JR, & Harlan WR (1985) The relationship
between blood lead levels and blood pressure and its cardiovascular
risk implications. Am J Epidemiol, 121: 246-258.
Pocock SJ, Shaper AG, Walker M, Wale CJ, Clayton B, Delves T, Lacey
RF, Packham RF, & Powell P (1983) Effects of tap water lead, water
hardness, alcohol, and cigarettes on blood lead concentrations. J
Epidemiol Community Health, 37: 1-7.
Pocock SJ, Shaper AG, Ashby D, Delves T, & Whitehead TP (1984) Blood
lead concentration, blood pressure, and renal function. Br Med J,
289: 872-874.
Pocock S, Ashby D, & Smith MA (1987) Lead exposure and children's
intellectual performance. Int J Epidemiol, 16: 57-67.
Pocock SJ, Shaper AG, Ashby D, Delves HT, & Clayton BE (1988) The
relationship between blood lead, blood pressure, stroke and heart
attacks in middle-aged British men. Environ Health Perspect,
78: 23-30.
Pocock SJ, Ashby D, & Smith MA (1989) Lead exposure and children's
intellectual performance. The Institute of Child Health/Southampton
Study. In: Smith M, Grant LD, & Sors A ed. Lead exposure and child
development: An international assessment. Dordrecht, London, Kluwer
Academic Press, pp 149-165.
Pollock CA & Ibels LS (1986) Lead intoxication in paint removal
workers on the Sydney Harbour Bridge. Med J Aust, 145: 635-639.
Ponka A, Salminen E, & Ahonen S (1991) Lead in the ambient air and
blood specimens of children in Helsinki. Sci Total Environ, 138(1-3):
301-308.
Pontifax AH & Garg AK (1985) Lead poisoning from an Asian Indian folk
remedy. Can Med Assoc J, 133: 1227-1228.
Poole C, Smythe LE, & Alpers M (1980) Blood lead levels of Papua New
Guinea children living in a remote area. Sci Total Environ, 15: 17-24.
Pounds JG (1984) Effect of lead intoxication on calcium homeostasis
and calcium-mediated cell function: a review. Neurotoxicology,
5: 295-332.
Pounds JG, Marlar RJ, & Allen JR (1978) Metabolism of lead-210 in
juvenile and adult rhesus monkeys Macaca mulatta. Bull Environ Contam
Toxicol, 19: 684-691.
Pounds JG, Long GJ, & Rosen JF (1991) Cellular and molecular toxicity
of lead in bone. Environ Health Perspect, 91: 17-32.
Purchase NG & Fergusson JE (1986) Lead in teeth: the influence of the
tooth type and the sample within a tooth on lead levels. Sci Total
Environ, 52: 239-250.
Qazi QH, Madahar C, & Huceoglu AM (1980) Temporary increase in
chromosome breakage in an infant prenatally exposed to lead. Hum
Genet, 53: 201-203.
Qian ZM & Morgan EH (1990) Effect of lead on the transport of
transferrin-free and transferrin-bound iron into rabbit reticulocytes.
Biochem Pharmacol, 40: 1049-1054.
Quarterman J & Morrison JN (1975) The effects of dietary calcium and
phosphorus on the retention and excretion of lead in rats. Br J Nutr,
34: 351-362.
Que Hee SS & Boyle JR (1988) Simultaneous multielemental analysis of
some environmental and biological samples by inductively coupled
plasma atomic emission spectrometry. Anal Chem, 60: 1033-1042.
Que Hee SS, MacDonald TJ, & Bornschein RL (1985a) Blood lead by
furnace-Zeeman atomic absorption spectrophotometry. Microchem J,
32: 55-63.
Que Hee SS, Peace B, Clark CS, Boyle JR, Bornschein RL, & Hammond PB
(1985b) Evolution of efficient methods to sample lead sources, such as
house dust and hand dust in the homes of children. Environ Res,
38: 77-95.
Raab GM, Laxen DPH, & Fulton M (1987) Lead from dust and water as
exposure sources for children. Environ Geochem Health, 9: 80.
Rabinowitz MB (1991) Toxicokinetics of bone lead. Environ Health
Perspect, 91: 33-37.
Rabinowitz M & Needleman HL (1982) Temporal trends in the lead
concentrations of umbilical cord blood. Science, 216: 1429-1431.
Rabinowitz MB, Wetherill G, & Kopple J (1975) Absorption, storage and
excretion of lead by normal humans. In: Proceedings of the Ninth
Annual Conference on Trace Substances in Environmental Health.
Columbia, Missouri, University of Missouri, pp 361-368.
Rabinowitz MB, Wetherill GM, & Kopple JD (1976) Kinetics analysis of
lead metabolism in healthy humans. J Clin Invest, 58: 260-270.
Rabinowitz MB, Wetherill GW, & Kopple JD (1977a) Magnitude of lead
intake from respiration by normal man. J Lab Clin Med, 90: 238-248.
Rabinowitz MB, Wetherill GW, & Kopple JD (1977b) Effect of food intake
and fasting on gastrointestinal lead absorption in humans. Am J Clin
Nutr, 33: 1784-1788.
Rabinowitz MB, Kopple JD, & Wetherill GW (1980) Effect of food intake
and fasting on gastrointestinal lead absorption in humans. Am J Clin
Nutr, 33: 1784-1788.
Rabinowitz MB, Leviton A, & Needleman HL (1986) Occurrence of elevated
protoporphyrin levels in relation to lead burden in infants. Environ
Res, 39: 253-257.
Rabinowitz M, Bellinger D, Leviton A, Needleman H, & Schoenbaum S
(1987) Pregnancy hypertension, blood pressure during labor, and blood
lead levels. Hypertension, 10: 447-451.
Rabinowitz MB, Leviton A, & Bellinger D (1989) Blood lead-tooth lead
relationship among Boston children. Bull Environ Contam Toxicol,
43: 485-492.
Rabinowitz MB, Wang J-D, & Soong WT (1991) Dentine lead and child
intelligence in Taiwan. Arch Environ Health, 46: 351-360.
Ragan HA (1977) Effect of iron deficiency on the absorption of lead
and cadmium in the rat. J Lab Clin Med, 90: 700-706.
Reaves GA & Berrow ML (1984) Total lead concentrations in Scottish
soils. Geoderma, 32: 1-8.
Reisinger K, Stoeppler M, & Nurnberg HW (1981) Evidence for the
absence of biological methylation of lead in the environment. Nature
(Lond), 281: 228-230.
Reuter JH & Perdue EM (1977) Importance of heavy metal-organic matter
interactions in natural waters. Geochim Cosmochim Acta, 41: 325-334.
Rhagavan SRV, Dwight Culver B, & Gonick HC (1981) Erythrocyte
lead-binding protein after occupational exposure. II. Influence of
lead inhibition of membrane Na+, K±-adenosinetriphosphatase. J Toxicol
Environ Health, 7: 561-568.
Rice DC (1984a) Behavioural deficit (delayed matching to sample) in
monkeys exposed from birth to low level lead. Toxicol Appl Pharmacol,
75: 337-345.
Rice DC (1984b) Effect of lead on schedule-controlled behavior in the
monkey. In: Seiden LS & Balster RL ed. Behavioral pharmacology: The
current status. New York, Alan R. Liss, pp 473-486.
Rice DC (1985) Chronic low-lead exposure from both produces deficits
in discrimination reversal monkey's. Toxicol Appl Pharmacol,
77: 201-210.
Rice DC & Karpinski KF (1988) Lifetime dow level lead exposure
produces deficits in delayed alternation in adult monkeys.
Neurotoxicol Teratol, 10: 207-214.
Roberts J, Mahaffey KR, & Annest JL (1985) Blood lead levels in
general populations. In: Mahaffey KR ed. Dietary and environmental
lead: Human health effects. Amsterdam, Oxford, New York, Elsevier
Science Publishers, pp 355-372.
Robins JM, Cullen MR, Connors BB, & Kayne RD (1983) Depressed thyroid
indexes associated with occupational exposure to inorganic lead. Arch
Intern Med, 143: 220-224.
Robinson TR (1974) Delta-aminolevulinic acid and lead in urine of lead
antiknock workers. Arch Environ Health, 28: 133-138.
Rodamilans M, Osaba MJM, To-Figueras J, Fillat FR, Marques JM, Perez
P, & Corbella J (1988) Lead toxicity on endocrine testicular function
in an occupationally exposed population. Hum Toxicol, 7: 125-128.
Rodney BS & Lee MG (1985) Environmental lead hazard in a high-risk
Jamaican population. West Indian Med J, 34: 176-179.
Roels HA & Lauwerys R (1987) Evaluation of dose-effect and
dose-response relationships for lead exposure in different Belgian
population groups (foetus, child, adult men and women). Trace Elem
Med, 4: 80-87.
Roels HA, Buchet J-P, Lauwerys R, Hubermont G, Bruaux P, Claeys-
Thoreau F, Lafontaine A, & van Overschelde J (1976) Impact of air
pollution by lead on the haem biosynthetic pathway in school-age
children. Arch Environ Health, 31: 310-316.
Roels HA, Buchet JP, Lauwerys RR, Bruaux P, Claeys-Thoreau F,
Lafontaine A, & Verduyn G (1980) Exposure to lead by the oral and the
pulmonary routes of children living in the vicinity of a primary lead
smelter. Environ Res, 22: 81-94.
Rosen JF & Chesney RW (1983) Circulating calcitriol concentration in
health and disease. J Pediatr, 103: 1-7.
Rosen JF, Chesney RW, Hamstra AJ, Deluca HF, & Mahaffey KR (1980)
Reduction in 1,25-dihydroxyvitamin D in children with increased lead
absorption. N Engl J Med, 302: 1128-1131.
Rosen JF, Markowitz ME, Bijur PE, Jenks ST, Wielopolski L, Kalef-Ezra
JA, & Slatkin DN (1991) Sequential measurements of bone lead content
by L x-ray fluorescence in CaNa2EDTA-treated lead-toxic children.
Environ Health Perspect, 91: 57-62.
Rosner D & Markowitz G (1985) A "gift of God": The public health
controversy over leaded gasoline during the 1920s. Am J Public Health,
75(4): 344.
Rossouw J, Offermeier J, & van Rooyen J (1987) Apparent central
neurotransmitter receptor changes induced by low-level lead exposure
during different developmental phases in the rat. Toxicol Appl
Pharmacol, 91: 132-139.
Routh DK, Mushak P, & Boone L (1979) A new syndrome of elevated blood
lead and microcephaly. J Pediatr Psychol, 4: 67-76.
Rundle SA & Duggan MJ (1986) Lead pollution from the external
redecoration of old buildings. Sci Total Environ, 57: 181-190.
Ryu JE, Ziegler EE, Nelson SE, & Fomon SJ (1983) Dietary intake of
lead and blood lead concentration in early infancy. Am J Dis Child,
137: 886-891.
Ryu JE, Ziegler EE, Nelson SE, & Fomon SJ (1985) Dietary and
environmental exposure to lead and blood lead during early infancy.
In: Mahaffey KR ed. Dietary and environmental lead: Human health
effects. Amsterdam, Oxford, New York, Elsevier Science Publishers,
pp 187-209.
Saenger P, Markowitz MR, & Rosen JF (1984) Depressed excretion of
6ß-hydroxycortisol in lead-toxic children. J Clin Endocrinol Metab,
58: 363-367.
SAHC (1993) Proceedings of the International Meeting on
Non-Occupational Exposure to Lead, Melbourne, Australia, 5-9 October
1992. Adelaide, South Australian Health Commission, 56 pp.
Sarto F, Stella M, & Acqua A (1978) [Cytogenic studies in 20 workers
occupationally exposed to lead.] Med Lav, 69: 172-180 (in Italian).
Satzger RD, Clow CS, & Bonnin E (1982) Determination of background
levels of lead and cadmium in raw agricultural crops by using
differential pulse anodic stripping voltammetry. J Assoc Off Anal
Chem, 65: 987-991.
Sauk J & Somerman MJ (1991) Physiology of bone: mineral compartment
proteins as candidates for environmental perturbation by lead. Environ
Health Perspect, 91: 9-16.
Schlegel H & Kufner G (1979) Long-term observation of biochemical
effects of lead in human experiments. J Clin Chem Clin Biochem,
17: 225-233.
Schmid E, Bauchinger M, Pietruk S, & Hall G (1972) [Cytogenic action
of lead in human peripheral lymphocytes in vitro and in vivo.]
Mutat Res, 16: 401-406 (in German).
Schmidt U & Huber F (1976) Methylation of organolead and lead(II)
compounds (CH3)4 Pb by microorganisms. Nature (Lond),
259: 157-158.
Schmitt MDC, Trippler DL, & Wachtler JN (1988) Soil lead
concentrations in residential Minnesota as measured by ICP AES. Water
Air Soil Pollut, 39: 157-168.
Schneitzer L, Osborn HH, Bierman A, Mezey A, & Kaul B (1990) Lead
poisoning in adults from renovation of an older home. Ann Emerg Med,
19: 415-420.
Schroeder SR, Hawk B, Otto DA, Mushak P, & Hicks RE (1985) Separating
the effects of lead and social factors on IQ. Environ Res,
38: 144-154.
Schutz AS, Skerfving S, Ranstam J, & Christoffersson JO (1987)
Kinetics of lead in blood after the end of occupational exposure.
Scand J Work Environ Health, 13: 221-231.
Schutz A, Ranstam J, Skerfving S, & Tejning S (1989) Blood-lead levels
in school children in relation to industrial emission and automobile
exhausts. Ambio, 13: 115-117.
Schwartz J (1988) The relationship between blood lead and blood
pressure in the NHANES II survey. Environ Health Perspect, 78: 15-22.
Schwartz J (1993) Low-level lead exposure and children's IQ: a
meta-analysis and search for a threshold. Environ Res, 65: 42-55.
Schwartz J & Otto D (1987) Blood lead, hearing thresholds, and
neurobehavioral development in children and youth. Arch Environ
Health, 42: 153-160.
Schwartz J & Otto D (1991) Lead and minor hearing impairment. Arch
Environ Health, 46: 300-305.
Schwartz J, Angle C, & Pitcher H (1986) Relationship between childhood
blood lead levels and stature. Pediatrics, 77: 281-288.
Schwartz J, Landrigan PL, & Baker EL (1990) Lead-induced anemia:
dose-response relationships and evidence for a threshold. Am J Public
Health, 80: 165-168.
Scott DR, Hemphill DC, & Holboke LE (1976) Atomic absorption and
optical emission analysis of NASN atmospheric particulate samples for
lead. Environ Sci Technol, 9: 877-880.
Selander S & Cramer K (1970) Interrelationships between lead in blood,
lead in urine and ALA in urine during lead work. Br J Ind Med,
27: 28-39.
Selevan SG, Landrigan PJ, Stern FB, & Jones JH (1985) Mortality of
lead smelter workers. Am J Epidemiol, 122: 673-683.
Selevan SG, Landrigan PJ, Stern FB, & Jones JH (1988) Brief report:
lead and hypertension in a mortality study of lead smelter workers.
Environ Health Perspect, 78: 65-66.
Seppäläinen AM & Hernberg S (1980) Subclinical lead neuropathy. Am J
Ind Med, 1: 413-420.
Seppäläinen AM, Hernberg S, Vesanto R, & Kock B (1983) Early
neurotoxic effects of occupational lead exposure: a prospective study.
Neurotoxicology, 4(2): 181-192.
Settle DM & Patterson CC (1980) Lead in albacore: guide to lead
pollution in Americans. Science, 207: 1167-1176.
Shaltout A, Yaish SA, & Fernando N (1981) Lead encephalopathy in
infants in Kuwait. Ann Trop Paediatr (London), 1: 209-215.
Shaper AG, Pocock SJ, Walker M, Cohen NM, Wale CJ, & Thompson AG
(1981) British regional heart study: cardiovascular risk factors in
middle-aged men in 24 towns. Br Med J, 283: 179-186.
Sharma RR, Chandy MJ, & Lad SD (1990) Transient hydrocephalis and
acute lead encephalopathy in neonates and infants. Report of two
cases. Br J Neurosurg, 4: 141-146.
Sherlock J, Smart G, Forbes GI, Moore MR, Patterson WJ, Richards WN, &
Wilson TS (1982) Assessment of lead intakes and dose-response for a
population in Ayr exposed to a plumbsolvent water supply. Hum Toxicol,
1: 115-122.
Sherlock JC, Pickford CJ, & White GF (1986) Lead in alcoholic
beverages. Food Addit Contam, 3: 347-354.
Siegel M, Forsyth B, Siegel L, & Cullen MR (1989) The effect of lead
on thyroid function in children. Environ Res, 49: 190-196.
Silbergeld EK (1991) Lead in bone: implications for toxicology during
pregnancy and lactation. Environ Health Perspect, 91: 63-70.
Silbergeld EK, Schwartz J, & Mahaffey KR (1988) Lead and osteoporosis:
mobilization of lead from bone in postmenopausal women. Environ Res,
47: 79-94.
Silva PA, Hughes P, Williams S, & Faed JM (1988) Blood lead,
intelligence, reading attainment, and behaviour in eleven year old
children in Dunedin, New Zealand. J Child Psychol Psychiatry Appl
Discipl, 29: 43-52.
Sinclair DF & Dohnt BR (1984) Sampling and analysis techniques used in
a blood lead survey of 1341 children in Port Pirie, South Australia.
Clin Chem, 30(10): 1616-1619.
Skerfving S (1988) Biological monitoring of exposure to inorganic
lead. In: Clarkson TW, Friberg L, Nordberg GF, & Sager PR ed.
Biological monitoring of toxic metals. New York, Plenum Press,
pp 169-197 (Rochester Series on Environmental Toxicity).
Skerfving S, Schutz A, & Ranstam J (1986) Decreasing lead exposure in
Swedish children. Sci Total Environ, 58: 225-229.
Skogerboe RK, Hartley AM, Vogel RS, & Koirtyohann SR (1977) Monitoring
for lead in the environment. In: Boggess WR ed. Lead in the
environment. Washington, DC, National Science Foundation, pp 33-70
(NSF Report No. NSF/RA-770214; PB-278278).
Slorach SA, Gustafsson I-B, Jorhem L, & Mattsson P (1983) Intake of
lead, cadmium and certain other metals via a typical Swedish weekly
diet. Var Föda, 35(Suppl 1): 3-16.
Smith MA (1989) The effects of low-level lead exposure on children.
In: Smith MA, Grant LD, & Sors AI ed. Lead exposure and child
development: An international assessment. Dorcrecht, London, Kluwer
Academic Publishers, pp 3-47.
Smith F & Hursh J (1977) Bone storage and release. In: Field J ed.
Handbook of physiology: Reaction to environmental agents. Bethesda,
Maryland, American Physiological Society, section 9, pp 469-482.
Smith M, Delves T, Lansdown R, Clayton B, & Graham P (1983) The
effects of lead exposure on urban children: the Institute of Child
Health/Southampton Study. Dev Med Child Neurol, 47(Suppl): 1-54.
Snee RD (1981) Evaluation of studies of the relationship between blood
lead and air lead. Int Arch Occup Environ Health, 48: 219-242.
Sobel AE, Yuska H, Peters DD, & Kramer B (1940) The biochemical
behaviour of lead. I. Influence of calcium phosphorus and vitamin D on
lead in the blood and bone. J Biol Chem, 132: 239-265.
Sokol RZ (1987) Hormonal effects of lead acetate in the male rat:
Mechanism of action. Biol Reprod, 37: 1135-1138.
Sokol RZ, Madding CE, & Swerdloff RS (1985) Lead toxicity and the
hypothalamic-pituitary-testicular axis. Biol Reprod, 33: 722-728.
Spivey GH, Baloh RW, Brown CP, Browdy BL, Campion DS, Valentine JL,
Morgan DE, & Culver BD (1980) Subclinical effects of chronic increased
lead absorption - a prospective study: III. Neurologic findings at
follow-up examination. J Occup Med, 22: 607-612.
Staessen J, Yeoman WB, Fletcher AE, Markowe HLJ, Marmot MG, Rose G,
Semmence A, Shipley MJ, & Bulpitt CJ (1990) Blood lead concentration,
renal function, and blood pressure in London civil servants. Br J Ind
Med, 47: 442-447.
Staessen JA, Lauwerys RA, Buchet J-P, Bulpitt CJ, Rondia D,
Vanrenterghem Y, Amery A, & The Cadmibel Study Group (1992) Impairment
of renal function with increasing blood lead concentrations in the
general population. N Engl J Med, 327: 151-156.
Staessen JA, Christopher JB, Fagard R, Lauwerys RR, Roels H, Thijs L,
& Amery A (1994) Hypertension caused by low-level lead exposure: myth
or fact? J Cardiovasc Risk, 1: 87-97.
Steelink C (1977) Humates and other natural organic substances in the
aquatic environment. J Chem Ed, 54: 599-603.
Steenhout A (1987) How clean is clean? An exotoxicological method for
getting guidelines (air, dust, deposition, water) for lead, keeping
tooth and blood lead levels in the normal range. In: Proceedings of
the International Conference on Heavy Metals in the Environment.
Edinburgh, CEP Consultants, pp 283-285.
Steenhout A & Pourtois M (1981) Lead accumulation in teeth as a
function of age with different exposures. Br J Ind Med, 38: 297-303.
Stiles KM & Bellinger DC (1993) Neuropsychological correlates of
low-level lead exposure in school-age children: A prospective study.
Neurotoxicol Teratol, 15(1): 27-35.
Stollery BT, Banks HA, Broadbent DE, & Lee WR (1989) Cognitive
functioning in lead workers. Br J Ind Med, 46: 698-707.
Stollery BT, Broadbent DE, Banks HA, & Lee WR (1991) Short term
prospective study of cognitive functioning in lead workers. Br J Ind
Med, 48: 739-749.
Stone CH & Soares JH (1976) The effect of dietary selenium level of
lead toxicity in the Japanese quail. Poult Sci, 55: 341-349.
Stone C, Fox MRS, & Hogye KS (1981) Bioavailability of lead in oysters
fed to young Japanese quail. Environ Res, 26: 409-421.
Stuik EJ (1974) Biological response of male and female volunteers to
inorganic lead. Int Arch Arbeitsmed, 33: 83-97.
Succop PA, O'Flaherty EJ, Bornschein RL, Roba SB, & Greenland RD
(1989) Recent observations concerning the relationship of blood lead
to erythrocytic protoporphyrin. In: Smith MA, Grant LD, & Sors AE ed.
Lead exposure and child development: An international Assessment.
Dordrecht, London, Kluwer Academic Publishers, pp 477-484.
Talbot RW & Andren AW (1983) Relationships between Pb and 210Pb in
aerosol and precipitation at a semiremote site in northern Wisconsin.
J Geophys Res, 88: 6752-6760.
Taskinen H, Nordman H, Engstrom K, & Hernberg S (1981) [Blood lead
levels in Finnish children.] Duodecim, 97(10): 663-670 (in Finnish).
Teruya K, Sakurai H, Omae K, Higashi T, Muto T, & Kaneko Y (1991)
Effect of lead on cardiac parasympathetic function. Int Arch Occup
Environ Health, 62: 549-553.
Thornton I & Webb JS (1975) Trace elements in soils and surface waters
contaminated plast metaliferous mining part of England. In:
Proceedings of the IXth Annual Conference on Trace Substances in
Environmental Health, Columbia, Missouri, 10-12 June, pp 77-88.
Thornton I, Culbard E, Moorcroft S, Watt J, Wheatley M, & Thompson M
(1985) Metals in urban dusts and soils. Environ Technol Lett,
6: 137-144.
Todd AC, McNeill FE, Palethorpe JE, Peach DE, Chettle DR, Tobin MJ,
Strosko SJ, & Rosen JC (1992) In vivo x-ray fluorescence of lead in
bone using K x-ray excitation with 109Cd sources; radiation dosimetry
studies. Environ Res, 57: 117-132.
Triebig G, Weltle D, & Valentin H (1984) Investigations on
neurotoxicity of chemical substances at the workplace. V.
Determination of the motor and sensory nerve conduction velocity in
persons occupationally exposed to lead. Int Arch Occup Environ Health,
53: 189-204.
Trotter RT (1990) The cultural parameters of lead poisoning: A medical
anthropologist's view of intervention in environmental lead exposure.
Environ Health Perspect, 89: 79-84.
Tuppurainen M, Wagar G, Kurppa K, Sakari W, Wambugu A, Froseth B, Alho
J, & Nykyri E (1988) Thyroid function as assessed by routine
laboratory tests of workers with long-term lead exposure. Scand J Work
Environ Health, 14: 175-180.
UK (1982) The Glasgow duplicate diet study (1979/1980): A joint survey
for the Department of the Environment and the Ministry of Agriculture,
Fisheries and Food. London, Her Majesty's Stationery Office (Pollution
Report No. 11).
US Department of the Treasury (1991) Report of analyses of wines and
related products to determine lead content. Washington, DC, US
Department of the Treasury, Bureau of Alcohol, Tobacco and Firearms.
US EPA (1986a) Air quality criteria for lead. Washington, DC,
Environmental Protection Agency (EPA-600/8-83/028aF-dF).
US EPA (1986b) Test methods for evaluating solid waste SW-846 -
Physical/chemical methods. Method No. 7420 and 7421. Washington, DC,
US Environmental Protection Agency, Office of Solid Waste and
Emergency Response.
US EPA (1989) Evaluation of the potential carcinogenicity of lead and
lead compounds. Washington, DC, US Environmental Protection Agency,
Office of Health and Environmental Assessment (EPA/600/8-89/045A).
US EPA (1990) Supplement to the 1986 EPA air quality criteria for
lead. Volume 1, Addendum: Relationship of blood pressure to lead
exposure. Washington, DC, US Environmental Protection Agency, Office
of Health and Environmental Assessment (EPA/600/8-89/0 49F).
US EPA (1991) National air quality and emission trends report.
Research Triangle Park, North Carolina, US Environmental Protection
Agency (EPA-450/4-91-023).
US NCI (1979) Bioassay of lead dimethylditho-carbamate for possible
carcinogenicity. Washington, DC, National Cancer Institute,
Carcinogenesis Testing Program, Division of Cancer Cause and
Prevention (Technical Report Series No. 151 - NTIS PB-85-134663).
Vahter M & Friberg L (1988) Quality control in integrated human
exposure monitoring of lead and cadmium. Fresenius Z Anal Chem,
332: 726-731.
Vahter M & Slorach S (1990) GEMS - Global Environment Monitoring
System. Exposure monitoring of lead and cadmium: an international
pilot study within the WHO/UNEP Human Exposure Assessment Location
(HEAL) Programme. Nairobi, United Nations Environment Programme,
82 pp.
Vahter M, Berglund M, Friberg L, Jorhem L, Lind B, Slorach S, &
Åkesson A (1990) Dietary intake of lead and cadmium in Sweden. Var
Föda, 42(2): 1-16.
Vahter M, Berglund M, Slorach S, Friberg L, Saric M, Xingquan Z, &
Fujita M (1991a) Methods for integrated exposure monitoring of lead
and cadmium. Environ Res, 56: 78-79.
Vahter M, Berglund M, Lind B, Jorhem L, Slorach S, & Friberg L (1991b)
Personal monitoring of lead and cadmium exposure - a Swedish study
with special reference to methodological aspects. Scand J Work Environ
Health, 17: 65-74.
Van Esch GJ & Kroes R (1969) The induction of renal tumors by feeding
basic lead acetate to mice and hamsters. Br J Cancer, 23: 765-771.
Varo P & Kovistoinen P (1983) Mineral element composition of Finnish
foods. XII. General discussion and nutritional evaluation. Acta Agric
Scand, 22: 165.
Vatsala S & Ramakrishna T (1985) "Tinning" of brass utensils:
Possibility of lead poisoning. Indian J Environ Health, 27: 140-141.
Verrula GR & Noah PK (1990) Clinical manifestations of childhood lead
poisoning. J Trop Med Hyg, 93: 170-177.
Verschoor M, Wibowo A, & Herber R (1987) Influence of occupational low
level lead exposure on renal parameters. Am J Ind Med, 12: 341-351.
Victery W (1988) Evidence for effects of chronic lead exposure on
blood pressure in experimental animals: An overview. Environ Health
Perspect, 78: 71-76.
Victery W, Vander AJ, Markel H, Katzman L, Shulak JM, & Germain C
(1982a) Lead exposure, begun in-utero, decreases renin and angiotensin
II in adult rats. Proc Soc Exp Biol Med, 170: 63-67.
Victery W, Vander AJ, Shulak JM, Schoeps P, & Julius S (1982b) Lead,
hypertension and the renin-angiotensin system in rats. J Lab Clin Med,
99: 354-362.
Volkening J, Baumann H, & Heumann KG (1988) Atmospheric distribution
of particulate lead over the Atlantic Ocean from Europe to Antarctica.
Atmos Environ, 22: 1169-1174.
Wadge A & Hutton M (1987) The leachability and chemical speciation of
selected trace elements in fly ash from coal combustion and refuse
incineration. Environ Pollut, 48: 85-99.
Wai CM, Knowles CR, & Keely JF (1979) Lead caps on wine bottles and
their potential problems. Bull Environ Contam Toxicol, 21: 4-6.
Wallace DM, Kalman DA, & Bird TD (1985) Hazardous lead release from
glazed dinnerware: A cautionary note. Sci Total Environ, 44: 289-292.
Wang Y-L (1984) Industrial lead poisoning in China over the past 33
years. Ecotoxicol Environ Saf, 8: 526-530.
Wang J-D, Jang C-S, Hwang Y-H, & Chen Z-S (1992) Lead contamination
around a kindergarten near a battery recycling plant. Bull Environ
Contam Toxicol, 49: 23-30.
Ward NI, Watson R, & Bryce-Smith D (1987) Placental element levels in
relation to fetal development for obstetrically normal births. A study
of 37 elements. Evidence for the effects of cadmium, lead, and zinc on
fetal growth and for smoking as a source of cadmium. Int J Biosoc Res,
9: 63-81.
Wasserman G, Graziano JH, Factor-Litvak P, Popovac D, Morina N,
Musabegovic A, Vrenezi N, Capuni-Paracka S, Lekic V, Preteni-Redjepi
E, Hadzialijevic S, Slavkovich V, Kline J, Shrout P, & Stein Z (1992)
Independent effects of lead exposure and iron deficiency anemia on
developmental outcome at age 2 years. J Pediatr, 121(5): 695-703.
Watson WS, Hume R, & Moore MR (1980) Oral absorption of lead and iron.
Lancet, 2(8188): 236-237.
Watson WS, Morrison J, Bethel MIF, Baldwin NM, Lyon DTB, Dobson H,
Moore MR, & Hume R (1986) Food iron and lead absorption in humans. Am
J Clin Nutr, 44: 248-256.
Weast RC ed. (1985) Handbook of chemistry and physics, 66th ed. Boca
Raton, Florida, CRC Press.
Weeden RP, Mallik DK, & Batuman V (1979) Detection and treatment of
occupational lead nephropathy. Arch Intern Med, 139: 53-57.
White JM & Harvey DR (1972) Defective synthesis of alpha and ß globin
chains in lead poisoning. Nature (Lond), 236: 71-73.
WHO (1980) Recommended health-based limits in occupational exposure to
heavy metals. Report of a WHO Study Group. Geneva, World Health
Organization (Technical Report Series No. 647).
WHO (1984) Guidelines for drinking-water quality. Volume 1:
Recommendations. Geneva, World Health Organization, pp 55-56.
WHO (1985) Guidelines for the study of dietary intake of chemical
contaminants. Geneva, World Health Organization (WHO Offset
Publication No. 87).
WHO (1987) Air quality guidelines for Europe. Copenhagen, World Health
Organization, Regional Office for Europe, pp 200-209 (European Series,
No. 23).
WHO (1993) Guidelines for drinking-water quality. 2nd ed. Volume 1:
Recommendations. Geneva, World Health Organization, 188 pp.
Wiebe JP, Barr KJ, & Buckingham KD (1982) Lead administration during
pregnancy and lactation affects steriodogenesis and hormone receptors
in testes of offspring. J Toxicol Environ Health, 10: 653-666.
Wiebe JP, Salhanick AI, & Myers KI (1983) On the mechanism of action
of lead on the testes: in vitro suppression of FSH receptors, cyclic
AMP and steroidogenesis. Life Sci, 32: 1997-2005.
Wiebe RA, Anderson BS, Lehman CW, & Fu DJ (1991) Lead poisoning in
Hawaii: 1990. Hawaiian Med J, 50: 89-95.
Wielopolski L, Ellis KJ, Vaswani AN, Cohn SH, Greenberg A, Puschett
JB, Parkinson DK, Fetterolf DE, & Langrigan PJ (1986) In vivo bone
lead measurements: A rapid monitoring method for cumulative lead
exposure. Am J Ind Med, 9: 221-226.
Wigg NR, Vimpani GV, McMichael AJ, Baghurst PA, Robertson EF, &
Roberts RJ (1988) Port Pirie cohort study: childhood blood lead and
neuropsychological development at age two years. J Epidemiol Community
Health, 42: 213-219.
Wildt K, Eliasson R, & Berlin M (1983) Effects of occupational
exposure to lead on sperm and semen. In: Clarkson TW, Nordberg GF, &
Sager PR ed. Reproductive and developmental toxicity of metals. New
York, London, Plenum Press, pp 279-300.
Wilhelm M, Lombeck I, & Hafner D (1989) Hair lead levels in young
children from the FRG. J Trace Elem Electrolytes Health Dis,
3: 165-170.
Williams MK, King E, & Walford J (1969) An investigation of lead
absorption in an electric accumulator factory with the use of personal
samplers. Br J Ind Med, 26: 202-216.
Williamson AM & Teo RKC (1986) Neurobehavioural effects of
occupational lead exposure. Br J Ind Med, 43: 374-380.
Wilson D, Esterman A, Lewis M, Roder D, & Calder I (1986) Children's
blood lead levels in the lead smelting town of Port Pirie South
Australia. Arch Environ Health, 41: 245-250.
Winneke G (1979) Modification of visual evoked potentials in rats
after long-term blood lead elevation. Acta Nerv Sup (Praha),
21: 282-284.
Winneke G, Brockhaus A, & Baltissen R (1977) Neurobehavioural and
system effects of long term blood lead-elevation in rats. I.
Discrimination learning and open field-behavior. Arch Toxicol,
37: 247-263.
Winneke G, Hrdina K-G, & Brockhaus A (1982a) Neuropsychological
studies in children with elevated tooth-lead concentrations. I. A
pilot study. Int Arch Occup Environ Health, 51: 169-183.
Winneke G, Lilienthal H, & Werner W (1982b) Task dependent
neurobehavioural effects of lead in rats. Arch Toxicol, 5(Suppl):
84-93.
Winneke G, Kramer U, Brockhaus A, Ewers U, Kujanek G, Lechner H, &
Janke W (1983) Neuropsychological studies in children with elevated
tooth-lead concentrations. II. Extended study. Int Arch Occup Environ
Health, 51: 231-252.
Winneke G, Beginn U, Ewert T, Havestadt C, Kraemer U, Krause C, Thron
HL, & Wagner HM (1985) Comparing the effects of perinatal and later
childhood lead exposure on neuropsychological outcome. Environ Res,
38: 155-167.
Winneke G, Collet W, Krämer U, Brockhaus A, Ewert T, & Krause C (1989)
Follow-up studies in lead-exposed children. In: Smith MA, Grant LD, &
Sors AI ed. Lead exposure and child development: An international
assessment. Dordrecht, London, Kluwer Academic Publishers, pp 260-270.
Winneke G, Brockhaus A, Ewers U, Kramer U, & Neuf M (1990) Results
from the European multicenter study on lead neurotoxicity in children:
implications for risk assessment. Neurotoxicol Teratol, 12: 553-559.
Wong PTS, Chau YK, & Luxon PL (1975) Methylation of lead in the
environment. Nature (Lond), 253: 263-264.
Wooler K & Melamed S (1978) Simple tests for monitoring excessive lead
absorption. Med J Aust, 1: 163-169.
World Bureau of Metal Statistics (1992) World metal statistics, July,
45(7): 75-78.
Worley PF, Barabon JM, Desouza EB, & Snyder SH (1986) Mapping second
messenger systems in the brain: differential localizations of
adenylate cyclase and protein kinase. C. Proc Natl Acad Sci (USA),
83: 4053-4057.
Worth D, Matranga A, Lieberman M, Devos E, Karelekas P, Ryan C, &
Kraun G (1981) Lead in drinking water: The contribution of household
tap water to blood lead levels. In: Lynam DR, Piantanida LG, & Cole JF
ed. Environmental lead. Proceedings of the Second International
Symposium on Environmental Lead Research. New York, London, San
Francisco, Academic Press, pp 199-225.
Yankel AJ, von Lindern IH, & Walter SD (1977) The Silver Valley lead
study: The relationship between childhood blood lead levels and
environmental exposure. J Air Pollut Control Assoc, 27: 763-767.
Yip R & Dallman PR (1984) Developmental changes in erythrocyte
protoporphyrin: Roles of iron deficiency and lead toxicity. J Pediatr,
710-713.
Yule W, Lansdown R, Millar IB, & Urbanowicz M-A (1981) The
relationship between blood lead concentrations, intelligence and
attainment in a school population: a pilot study. Dev Med Child
Neurol, 23: 567-576.
Zelikoff JT, Li JH, Hartwig A, Wang XW, Costa M, & Rossman TG (1988)
Genetic toxicology of lead compounds. Carcinogenesis, 9: 1727-1732.
Zerez CR, Wong MD, & Tanaka KR (1990) Partial purification and
properties of nicotinamide adenine dinucleotide synthetase from human
erythrocytes: evidence that enzyme activity is a sensitive indicator
of lead exposure. Blood, 75: 1576-1582.
Zhu BG (1984) Study of chronic lead-poisoning from using tea kettles.
Chung-Hua Yu Fang I Hsueh Tsa Chih, 18: 328-330.
Ziegler EE, Edwards BB, Jensen RL, Mahaffey KR, & Fomon SJ (1978)
Absorption and retention of lead by infants. Pediatr Res, 12: 29-34.
Zuckerman MA, Savory D, & Rayman G (1989) Lead encephalopathy from an
imported Toby mug. Postgrad Med J, 65: 307-309.
RESUME
La présente monographie est consacrée aux risques pour la santé
humaine découlant de l'exposition au plomb et aux dérivés minéraux du
plomb. On y insiste sur les données dont on a eu connaissance depuis
la publication du No 3 de la Série des Critères de l'environnement:
Plomb (OMS, 1977). Quant aux effets du plomb au niveau de
l'environnement, ils sont étudiés dans le No 85 de cette série: Lead -
Environmental Aspects (Le plomb dans l'environnement) (WHO, 1989, en
anglais seulement).
1. Identité, propriétés physiques et chimiques et méthodes d'analyse
Le plomb est un métal mou, gris argent, qui fond à 327,5°C. Il
est très résistant à la corrosion mais se dissout à chaud dans l'acide
nitrique et l'acide sulfurique. La valence habituelle du plomb dans
ses dérivés minéraux est de +2. La solubilité dans l'eau de ses
composés est variable, le sulfure et les oxydes étant peu solubles et
le nitrate, le chlorate et le chlorure assez solubles à chaud. Le
plomb forme également des sels avec des acides organiques tels que
l'acide lactique et l'acide acétique ainsi que des composés organiques
stables tels que le plomb-tétraéthyle et le plomb-tétraméthyle.
Les méthodes d'analyse les plus couramment utilisées pour doser
le plomb à faible concentration dans les produits biologiques et dans
l'environnement sont la spectrométrie d'absorption avec atomisation en
flamme, en four à cellules de graphite et en plasma induit par haute
fréquence (ICP/PIHF), ainsi que la coulométrie avec redissolution
anodique. Selon le traitement préalable subit par l'échantillon et les
techniques d'extraction et l'instrumentation utilisées, la limite de
détection peut être de 0,12 µmoles de plomb par litre de sang
(2,49 µg/dl). Toutefois, on n'obtient de résultats fiables qu'en se
conformant à une marche à suivre bien codifiée pour réduire au minimum
le risque de contamination au cours du prélèvement des échantillons,
de leur conservation, de leur traitement et de leur analyse.
2. Sources d'exposition humaine
La proportion du plomb dans la croûte terrestre est d'environ
20 mg/kg. Le plomb présent dans l'environnement peut être d'origine
naturelle ou artificielle. Le plomb naturellement présent dans
l'atmosphère provient, entre autres, de l'altération des roches et des
éruptions volcaniques, et on l'estime à 19 000 tonnes/an, contre
126 000 tonnes/an provenant des mines et des fonderies, la
consommation annuelle totale étant supérieure à 3 millions de tonnes.
On a trouvé des concentrations de plomb atmosphérique de
50 pg/m3 dans des zones reculées. La concentration de fond du plomb
dans le sol varie de 10 à 70 mg/kg et on a relevé à proximité de
routes une teneur moyenne de 138 mg/kg. Actuellement la teneur de
l'eau en plomb dépasse rarement quelques microgrammes/litre; la
concentration naturelle du plomb dans les eaux de surface est estimée
à 0,02 µg/litre.
Le plomb et ses dérivés peuvent pénétrer dans l'environnement par
une porte d'entrée quelconque lors de l'extraction minière, des
opérations de fonte et du traitement, de l'utilisation, du recyclage
ou du rejet du plomb. Le plomb est principalement utilisé dans les
batteries, les câbles, les pigments, les additifs pour essence, la
brasure et les aciers. Le plomb et ses dérivés entrent également dans
la composition de la brasure que l'on utilise pour souder les
conduites d'eau et les boites de conserve alimentaire; il sert
également à la confection de certains remèdes traditionnels, des
capsules de bouteilles contenant des boissons alcoolisées, des vernis
pour céramique et de la verrerie de cristal. Dans les pays où l'on
utilise encore de l'essence au plomb, les principales émissions dans
l'atmosphère proviennent de sources de combustion de produits
pétroliers, sources qui peuvent être mobiles ou fixes (en
agglomération). Les émissions dans l'atmosphère sont également très
importantes à proximité des mines et des fonderies de plomb.
Le plomb en suspension dans l'air peut se déposer sur le sol et
dans l'eau et il parvient ainsi jusqu'à l'homme par l'intermédiaire de
la chaîne alimentaire et de l'eau de boisson. Le plomb atmosphérique
contribue également pour une part importante au plomb présent dans la
poussière domestique.
3. Transport, distribution et transformation dans l'environnement
Le transport et la distribution du plomb à partir de sources
fixes, mobiles ou naturelles, s'effectue principalement par
l'intermédiaire de l'air. La majeure partie du plomb émis dans l'air
se redépose à proximité de la source d'émission, même si quelques
particules (de diamètre < 2 µm) dont transportées sur de longues
distances et provoquent la contamination de sites aussi reculés que
les glaciers arctiques. Le plomb aéroporté peut contribuer à
l'exposition humaine par contamination des aliments, d l'eau et de la
poussière, ainsi que par inhalation directe. L'élimination du plomb
aéroporté dépend des conditions atmosphériques et de la taille des
particules en suspension. De grandes quantités de plomb peuvent être
déversées dans le sol et l'eau. Cependant, ces produits ont tendance à
rester sur place du fait de la médiocre solubilité dans l'eau des
dérivés du plomb.
Le plomb qui s'est déposé dans l'eau, que ce soit à partir de
l'air ou par lessivage des sols, se répartit rapidement entre les
sédiments et la phase aqueuse, selon la valeur du pH, la teneur en
sels et la présence éventuelle d'agents chélateurs organiques. Lorsque
le pH dépasse 5,4, l'eau dure peu contenir environ 30 µg de
plomb/litre et l'eau douce environ 500 µg/litre. Une très faible
quantité du plomb déposé sur le sol passe dans les eaux de surface ou
les eaux souterraines sauf en cas d'érosion ou d'altération des
roches; il est normalement fortement lié par chélation aux matières
organiques.
Le plomb en suspension dans l'air peut passer dans les êtres
vivants soit directement, soit par fixation à partir du sol. Les
animaux peuvent être exposés au plomb de manière directe par broutage
de l'herbe, ingestion de terre ou par inhalation. Le plomb minéral ne
subit qu'une faible bioamplification le long de la chaîne alimentaire.
4. Concentrations dans l'environnement et exposition humaine
Dans la population générale adulte des non-fumeurs, la principale
voie d'exposition est la consommation de nourriture et d'eau. Le plomb
est suspension dans l'air peut jouer un rôle important dans
l'exposition, en fonction de facteurs tels que le tabagisme, la
profession, la proximité d'une autoroute, d'une fonderie, etc. ou de
certains lieux de loisirs (par exemple, ateliers d'artisanat, stands
de tir, etc.). Pour les nourrissons et les enfants en bas âge, la
nourriture, l'air, l'eau et la poussière ou la terre sont les
principales voies d'exposition potentielles. Pour les nourrissons
jusqu'à quatre ou cinq mois, les principales sources d'exposition au
plomb sont l'air, le lait et les laits maternisés ainsi que l'eau.
Les concentrations de plomb observées dans l'air, les aliments,
l'eau et le sol ou la poussière varient largement d'une région à
l'autre du monde et dépendent du degré d'industrialisation,
d'urbanisation ainsi que du mode de vie. Dans l'air ambiant, des
concentrations supérieures à 10 µg/m3 ont été signalées en milieu
urbain à proximité d'une fonderie, alors que dans les villes où l'on
n'utilise plus d'essence au plomb, ces concentrations peuvent tomber
en-dessous de 0,2 µg/m3. Dans ces conditions, la dose de plomb
absorbée à partir de l'air peut varier de moins de 4 µg/jour à plus de
200 µg/jour.
Dans des échantillons d'eau potable prélevés à la source, on
trouve généralement des concentrations de plomb inférieures à
5 µg/litre. Cependant, lorsque l'eau est prélevée au robinet dans les
maisons dont les canalisations sont en plomb, les teneurs peuvent
dépasser 100 µg/litre, en particulier lorsque l'eau a séjourné dans la
tuyauterie pendant plusieurs heures.
L'exposition au plomb par la voie alimentaire dépend de nombreux
facteurs tenant au mode de vie et notamment à la nature des aliments
consommés, au mode de préparation, au fait que l'on utilise ou non de
la brasure au plomb, à la teneur de l'eau en plomb et à l'utilisation
de vaisselle recouverte d'un vernis au plomb.
Les nourrissons et les enfants peuvent souvent être très exposés
au plomb présent dans la poussière et dans la terre. La teneur de la
poussière en plomb dépend de facteurs tels que la vétusté et l'état de
la maison, l'utilisation de peintures à base de plomb, la présence de
plomb dans l'essence et la densité urbaine. La dose de plomb absorbée
dépendra également de l'âge et du comportement de l'enfant ainsi que
de la biodisponibilité du plomb dans le produit où il est présent.
Chez les ouvriers des industries où l'on produit, raffine,
utilise ou rejette du plomb ou des dérivés du plomb, la principale
voie d'exposition est l'inhalation. Au cours d'un poste de travail de
8 heures, les ouvriers peuvent en absorber des quantités atteignant
400 µg, qui s'ajoutent aux 20 ou 30 µg absorbés quotidiennement à
partir de la nourriture, de l'eau et de l'air ambiant; l'ingestion de
grosses particules peut également contribuer de façon importante à cet
apport.
5. Cinétique et métabolisme chez les animaux de laboratoire et
l'homme
L'homme et les animaux résorbent le plomb qu'ils ingèrent ou
inhalent; la résorption percutanée est minime chez l'homme. Selon sa
granulométrie, sa forme chimique et sa solubilité dans les liquides
biologiques, un dérivé du plomb peut être résorbé jusqu'à hauteur de
50% après avoir été inhalé. Certaines grosses particules (plus de
7 µm) sont avalées après avoir été rejetées des voies respiratoires
par l'action de l'ascenseur muco-ciliaire. Chez l'homme et les animaux
d'expérience, l'absorption du plomb dans les voies digestives dépend
de la nature physico-chimique du produit ingéré, de l'état
nutritionnel du sujet ainsi que du type d'aliments consommés. Chez
l'homme adulte, le plomb contenu dans les aliments est absorbé à peu
près à hauteur de 10%; la proportion est plus élevée lorsque le sujet
est à la diète. Toutefois, chez les nourrissons et les enfants en bas
âge, le plomb d'origine alimentaire peut être absorbé à hauteur de
50%, encore que le taux d'absorption du plomb provenant des poussières
ou de la terre ainsi que des écailles de peinture puisse être plus
faible, selon sa biodisponibilité. Les régimes alimentaires pauvres en
calcium, phosphates, sélénium ou zinc peuvent accroître l'absorption
du plomb. Le fer et la vitamine D influent également sur l'absorption
du plomb.
Le taux de plomb dans le sang ou plombémie est utilisé pour
évaluer la charge en plomb de l'organisme ainsi que les doses de plomb
(internes) absorbées. La relation entre la plombémie et la
concentration du plomb dans les diverses sources d'exposition n'est
pas linéaire (corrélation curviligne).
Une fois absorbé, le plomb ne se répartit pas de manière homogène
dans l'organisme. Après être rapidement passé dans le sang et les
tissus mous, il se redistribue lentement des les os. La plomb
s'accumule dans les os pendant une longue période de la vie humaine et
peut servir de source endogène de plomb. La demi-vie du plomb dans le
sang et les tissus mous est d'environ 28 à 36 jours mais elle peut
être beaucoup plus longue dans les diverses parties de l'os. Le taux
de rétention du plomb dans l'organisme est plus élevé chez l'enfant
que chez l'adulte. Pendant toute la durée de la gestation, le plomb
passe facilement de la mère au foetus.
La plombémie est la mesure la plus couramment utilisée pour
évaluer l'exposition au plomb. Toutefois, on dispose aujourd'hui de
techniques permettant de doser le plomb dans les dents et les os,
encore que la cinétique du phénomène ne soit pas parfaitement
élucidée.
6. Effets sur les animaux de laboratoire et les systèmes d'épreuves
in vitro
Chez toutes les espèces d'animaux de laboratoire étudiées, y
compris des primates non-humains, on a constaté que le plomb
produisait des effets indésirables au niveau de plusieurs organes et
systèmes, notamment le système haematopoïétique, le système nerveux,
les reins, le système cardio-vasculaire, l'appareil reproducteur et le
système immunitaire. Le plomb est également nocif pour les os et on a
montré qu'il avait des effets cancérogènes sur le rat et la souris.
Malgré les différences d'ordre cinétique qui existent entre les
animaux d'expérience et l'homme, ces études apportent des arguments
biologiques de poids à la plausibilité de tels effets chez l'homme.
Chez le rat, on a observé une diminution de la capacité
d'apprentissage et de mémorisation lorsque la plombémie était de
l'ordre de 0,72 à 0,96 µmol/litre (soit 15 à 20 µg/dl), les mêmes
effets étant observés chez des primates non-humains pour une plombémie
ne dépassant pas 0,72 µmol/litre (15 µg/dl). En outre,
l'expérimentation animale a également permis d'observer des troubles
de la vision et de l'audition.
Chez le rat, une plombémie supérieure à 2,88 µmol/litre
(60 µg/dl), soit une valeur analogue à celle qui, selon les
observations, constitue le seuil d'apparition des effets chez l'homme,
est à même de provoquer l'apparition d'effets néphrotoxiques. Des
effets cardio-vasculaires ont été également observés chez des rats
après exposition chronique à de faibles doses de plomb entraînant une
plombémie de l'ordre de 0,24 à 1,92 µmol/litre (5 à 40 µg/dl). A des
doses inférieures à la dose maximale tolérée qui est de 200 mg de
plomb (sous forme d'acétate) par litre d'eau potable, on a observé
l'apparition de tumeurs. Cette dose constitue la dose maximale qui
n'entraîne pas d'autres effets morphologiques ou fonctionnels.
7. Effets sur l'homme
Chez l'homme, le plomb peut produire des effets biologiques très
divers selon l'intensité et la durée de l'exposition. On a ainsi
observé des effets au niveau infracellulaire ainsi que des effets
s'exerçant sur les fonctions générales de l'organisme, effets qui vont
de l'inhibition de certaines enzymes jusqu'à l'apparition
d'altérations morphologiques marquées et à la mort. Ces altérations se
produisent dans de larges limites de doses, l'organisme humain en
développement étant généralement plus sensible que l'organisme adulte.
On a montré que le plomb avait des effets sur nombre de processus
biochimiques; en particulier on a largement étudié les effets qu'il
produit sur la synthèse hémique, tant chez l'adulte que chez l'enfant.
Lorsque la plombémie est élevée, on observe une augmentation du taux
de protoporphyrine érythrocytaire sérique ainsi qu'une augmentation de
l'excrétion urinaire de coproporphyrine et d'acide delta-aminolévulinique.
A des valeurs plus faibles de la plombémie, on observe l'inhibition
d'enzymes comme la delta-aminolévulinique acide-déshydratase et la
dihydrobioptérine-réductase.
Les effets du plomb sur le système haématopoïétique se traduisent
par une diminution de la synthèse de l'hémoglobine et l'on a observé
une anémie chez des enfants lorsque la plombémie dépassait
1,92 µmol/litre (40 µg/dl).
Pour des raisons d'ordre neurologique, métabolique et
compartementale, les enfants sont plus sensibles aux effets du plomb
que les adultes. Des études épidémiologiques tant prospectives que
transversales ont été effectuées pour déterminer dans quelle mesure
une exposition au plomb présent dans l'environnement affecte les
fonctions psychologiques dépendant du système nerveux central. On a
ainsi montré qu'il existait une association entre l'exposition au
plomb et les troubles des fonctions neurocomportementales chez
l'enfant.
On a également constaté des anomalies des fonctions
psychologiques et neurocomportementales chez des ouvriers longtemps
exposés au plomb. Il a été montré que les paramètres
électrophysiologiques étaient d'utiles indicateurs des effets
infracliniques du plomb sur le système nerveux central.
On sait depuis longtemps que la neuropathie périphérique est due
à une exposition prolongée à de fortes concentrations de plomb sur le
lieu de travail. A plus faibles concentrations, on a observé une
diminution de la vitesse de conduction nerveuse. Ces effets se
révèlent souvent réversibles, en fonction de l'âge et de la durée de
l'exposition, après cessation de l'exposition.
Les effets que le plomb exerce sur le coeur sont indirects et
s'opèrent par l'intermédiaire du système neuro-végétatif; il n'y a pas
d'effets directs sur le myocarde. L'ensemble des faits tirés des
études sur des populations d'adultes indiquent qu'il existe une très
faible association entre la plombémie et la tension artérielle
systolique ou diastolique. Etant donné la difficulté qu'il y a à tenir
compte des facteurs de confusion, il n'a pas été possible d'établir, à
partir des résultats de ces études, une relation de cause à effet.
Rien n'indique non plus que l'association qui pourrait exister entre
la plombémie et la tension artérielle constitue un problème médical
majeur.
On sait que le plomb peut entraîner des lésions tubulaires
proximales qui se caractérisent par une aminoacidurie généralisée, une
hypophosphatémie avec d'une hyperphosphaturie relative et une
glycosurie accompagnée d'inclusions nucléiques, d'altérations des
mitochondries et d'une hypertrophie des cellules de l'épithélium
tubulaire proximal. Ces effets sur les tubules s'observent après une
exposition relativement brève et sont généralement réversibles. En
revanche, les altérations scléreuses et les fibroses interstitielles
qu'on observe lors d'une exposition chronique à de fortes
concentrations de plomb, entraînent des problèmes fonctionnels qui
peuvent déboucher sur une insuffisance rénale. On a observé une
augmentation du risque de néphropathie chez les travailleurs dont la
plombémie dépassait 3,0 µmol/litre (soit environ 60 µg/dl). En
utilisant des indicateurs fonctionnels plus sensibles, on a récemment
découvert l'existence d'effets rénaux dans la population générale.
Les effets du plomb sur la fonction de reproduction, ne
concernent, chez l'homme, que la morphologie et le nombre des
spermatozoïdes. Chez la femme, on a attribué au plomb un certain
nombre de grossesses à issue défavorable.
Il ne semble pas que le plomb ait des effets nocifs sur la peau,
les muscles ou le système immunitaire. Si l'on excepte le cas du rat,
le plomb ne paraît pas non plus être à l'origine de l'apparition de
tumeurs.
8. Evaluation des risques pour la santé humaine
Le plomb a des effets nocifs sur plusieurs organes et systèmes,
les effets les plus sensibles étant relevés au niveau infracellulaire
ainsi que sur le développement du système nerveux. On a fait état
d'une association entre la plombémie et l'hypertension. En outre, le
plomb entraîner une cascade d'effets sur les réserves hémiques de
l'organisme et il perturbe également la synthèse hémique. Cependant,
certains de ces effets ne sont pas véritablement considérés comme
délétères. Il y a également perturbation de l'homéostase du calcium
avec des contre-coups sur d'autres processus cellulaires.
a) Les preuves les plus substantielles sont on dispose à propos de
l'action nocive du plomb proviennent d'études transversales et
prospectives sur des populations dont la plombémie est
généralement inférieure à 1,2 µmol/litre (25 µg/dl) et concernent
une diminution du quotient d'intelligence (QI). Il importe
cependant de noter que ces observations ne constituent pas une
preuve définitive d'une relation de cause à effet entre ce
phénomène et l'exposition au plomb. Cependant la mesure de ce QI
à partir de l'âge de 4 ans fait ressortir un déficit qui se situe
entre 0 et 5 points (sur une échelle avec un écart-type de 15)
pour une augmentation de la plombémie de 0,48 µmol/litre
(10 µg/dl), l'ampleur de l'effet étant vraisemblablement de 1 à 3
points. Lorsque la plombémie dépasse 1,2 µmol/litre (25 µg/dl),
on peut avoir une relation différente entre cette plombémie et le
QI. Les estimations de l'ampleur de l'effet observé sont des
moyennes calculées sur des groupes et ne représentent qu'une
probabilité pour un individu en particulier.
Les études épidémiologiques dont on dispose ne donnent pas
de preuves définitives de l'existence d'un seuil. Lorsque la
plombémie se situe en-dessous de l'intervalle
0,48-0,72 µmol/litre (10-15 µg/dl), l'incertitude qui entache
toute estimation de l'effet s'accroît, du fait des facteurs de
confusion et des limites dans la précision des dosages et des
tests psychométriques. Il n'en reste pas moins qu'en-dessous de
cet intervalle de valeurs, certains éléments incitent à penser à
l'existence d'une association.
b) L'expérimentation animale milite en faveur de l'existence d'une
relation de cause à effet entre l'exposition au plomb et certains
effets neurologiques, puisqu'elle fait ressortir des déficits
dans les fonctions cognitives pour une plombémie ne dépassant pas
0,53 à 0,72 µmol/litre (11-15 µg/dl), déficits qui peuvent
persister bien après la cessation de l'exposition au plomb.
c) Pour une plombémie ne dépassant pas 1,44 µmol/litre (30 µg/dl),
il peut y avoir réduction de la vitesse de conduction nerveuse
périphérique chez l'homme. En outre, pour des valeurs de la
plombémie de dépassant pas 1,92 µmol/litre (40 µg/dl), il peut
également y avoir perturbation des fonctions moto-sensorielles et
la fonction du système nerveux neurovégétatif (variabilité de
l'intervalle R-R sur l'électrocardiogramme) peut être affectée
pour une valeur moyenne de la plombémie d'environ 1,68 µmol/litre
(35 µg/dl). Chez les travailleurs dont la plombémie dépasse
2,88 µmol/litre (60 µg/dl), il y a accroissement du risque de
néphropathie saturnienne. Toutefois, des études récentes basées
sur des indicateurs plus sensibles de la fonction rénale incitent
à penser que des effets peuvent se produire à des valeurs plus
faibles de l'exposition au plomb.
d) Il semblerait que l'exposition au plomb soit associée à une
légère augmentation de la tension artérielle. L'ordre de grandeur
probable de cette augmentation est le suivant: pour un doublement
de la plombémie (par exemple lorsqu'elle passe de 0,8 à
1,6 µmol/litre, soit de 16,6 à 33,3 µg/dl), il y a une
augmentation moyenne de 1 mmHg de la systolique. L'association
avec la diastolique est analogue mais d'ampleur plus faible.
Toutefois, on se demande si ces associations statistiques
résultent réellement de l'exposition au plomb ou s'il s'agit d'un
artefact imputable à des facteurs de confusion.
e) Certaines études épidémiologiques - mais pas toutes - font état
d'une association liée à la dose entre les accouchements avant
terme et certains indices de la croissance et de la maturation
foetales à des valeurs de la plombémie supérieures ou égales à
0.72 µmol/litre (15 µg/dl).
f) Les données relatives à la cancérogénicité pour l'homme du plomb
et de plusieurs de ses dérivés minéraux, sont insuffisantes.
g) On a mis en évidence des effets que le plomb exerce sur un
certain nombre de systèmes enzymatiques et de paramètres
biochimiques. Les valeurs de la plombémie au-dessus desquelles on
peut mettre en évidence des effets avec les techniques actuelles,
pour ce qui est des paramètres susceptibles d'avoir une
importance clinique, sont toutes supérieures à 0,96 µmol/litre
(20 µg/dl). Certains effets sur les enzymes peuvent être mis en
évidence à des valeurs plus faibles de la plombémie, mais leur
signification clinique demeure incertaine.
RESUMEN
La presente monografía se centra en los riesgos para la salud
humana asociados a la exposición al plomo y a los compuestos
inorgánicos de plomo. Se han destacado los datos disponibles desde la
publicación de Criterios de Salud Ambiental No. 3: Plomo (OMS, 1977).
Los efectos ambientales del plomo se examinan en Environmental Health
Criteria 85: Lead - Environmental Aspects (OMS, 1989).
1. Identidad, propiedades físicas y químicas y métodos analíticos
El plomo es un metal blando, gris plateado, que se funde a
327,5°C. Es muy resistente a la corrosión, pero es soluble en ácido
nítrico y en ácido sulfúrico caliente. Su valencia corriente en los
compuestos inorgánicos es +2. Su solubilidad en agua varía; el sulfito
de plomo y los óxidos de plomo son poco solubles, mientras que las
sales de nitrato, clorato y cloruro son razonablemente solubles en
agua fría. El plomo también forma sales con ácidos orgánicos tales
como el láctico y el acético, y compuestos orgánicos estables tales
como el tetraetilo de plomo y el tetrametilo de plomo.
Los métodos utilizados más corrientemente para el análisis de
bajas concentraciones de plomo en materias biológicas y ambientales
son la llama, el horno de grafito y la espectroscopia de absorción
atómica de plasma acoplado inductivamente y la voltimetría de
separación anódica. Según sean el tratamiento previo de la muestra,
las técnicas de extracción y la instrumentación analítica, pueden
alcanzarse niveles de detección de 0,12 µmoles de plomo por litro de
sangre (2,49 µg/dl). Sin embargo, se obtienen resultados fiables sólo
cuando se siguen procedimientos específicos para reducir al mínimo el
riesgo de contaminación durante la recogida, el almacenamiento,
procesamiento y análisis de la muestra.
2. Fuentes de exposición humana
El nivel de plomo en la corteza terrestre es de aproximadamente
20 mg/kg. El plomo del medio ambiente puede provenir de fuentes
naturales o antropogénicas. Las fuentes naturales de plomo atmosférico
comprenden el desgaste geológico y las emisiones volcánicas y se han
estimado en 19 000 toneladas por año, frente a unas 126 000 toneladas
por año emitidas en el aire como resultado de la minería, la fundición
y el consumo de más de 3 millones de toneladas de plomo por año.
Se han encontrado concentraciones atmosféricas de plomo de
50 pg/m3 en zonas remotas. Los niveles básicos de plomo en el suelo
oscilan entre 10 y 70 mg/kg y se ha comunicado un nivel medio de
138 mg/kg en las proximidades de las carreteras. Los niveles de plomo
presentes en las aguas rara vez exceden de unos pocos microgramos por
litro; la concentración natural de plomo en las aguas superficiales se
ha estimado en 0,02 µg/litro.
El plomo y sus compuestos pueden entrar en el medio ambiente en
cualquier punto durante las actividades de minería, fundición,
elaboración, utilización, reciclado o eliminación. Se utiliza
principalmente en la fabricación de pilas, cables, pigmentos, aditivos
de la gasolina, productos para soldar y de acero. El plomo y los
compuestos de plomo también se utilizan para soldar las tuberías de
distribución de agua y las latas de conserva, en algunos remedios
tradicionales, en las tapas de las botellas de bebidas alcohólicas y
en los esmaltes cerámicos y la cristalería de mesa. En los países
donde todavía se utiliza gasolina con plomo, la principal emisión en
el aire proviene de fuentes móviles y estacionarias de combustión de
gasolina (centros urbanos). Las zonas próximas a las minas y funderías
de plomo están expuestas a la emisión de niveles elevados en el aire.
El plomo del aire puede depositarse en el suelo y el agua, desde
donde llega al ser humano por conducto de la cadena alimentaria y del
agua de bebida. El plomo atmosférico también es una fuente importante
del plomo presente en el polvo de las viviendas.
3. Transporte, distribución y transformación en el medio ambiente
El transporte y la distribución del plomo procedente de fuentes
fijas, móviles y naturales tienen lugar principalmente a través del
aire. La mayor parte de las emisiones de plomo se depositan cerca de
la fuente, aunque algunas partículas de materia (< 2 µm de diámetro)
recorren largas distancias y contaminan lugares remotos tales como los
glaciares árticos. El plomo del aire puede contribuir a la exposición
humana mediante la contaminación de los alimentos, del agua y del
polvo, así como por inhalación directa. La eliminación del plomo del
aire depende de las condiciones atmosféricas y del tamaño de las
partículas. Pueden descargarse grandes cantidades de plomo en el suelo
y en el agua. Sin embargo, ese material tiende a permanecer localizado
debido a la escasa solubilidad de los compuestos de plomo en el agua.
El plomo depositado en el agua, ya provenga del aire o de la
escorrentía del suelo, se distribuye rápidamente entre el sedimento y
la fase acuosa, según el pH, el contenido de sales y la presencia de
agentes quelantes orgánicos. Con un pH superior a 5,4, las aguas duras
pueden contener aproximadamente 30 µg de plomo por litro y las aguas
blandas aproximadamente 500 µg de plomo por litro. Muy poco plomo
depositado en el suelo se transporta a las aguas superficiales o a las
subterráneas, salvo mediante la erosión o el desgaste geoquímico;
normalmente está ligado a la materia orgánica de forma bastante
estrecha (por quelación).
El plomo del aire puede transferirse a la biota directamente o
por absorción del suelo. Los animales pueden encontrarse expuestos al
plomo directamente mediante la ingestión de hierba y de tierra o por
inhalación. Hay poca biomagnificación del plomo inorgánico a través de
la cadena alimentaria.
4. Niveles ambientales y exposición humana
En la población general que no fuma, la principal vía de
exposición son los alimentos y el agua. El plomo del aire puede
contribuir apreciablemente a la exposición, lo que depende de factores
tales como el consumo de tabaco, la ocupación, la proximidad de
caminos transitados por vehículos automotores, de funderías de plomo,
etc., y ciertas actividades de esparcimiento (por ejemplo, artesanía,
tiro con armas de fuego). Los alimentos, el aire, el agua y el
polvo/suelo son las principales vías potenciales de exposición de los
niños pequeños. Para los niños de hasta 4 ó 5 meses de edad, el aire,
la leche, las preparaciones para lactantes y el agua son fuentes
notables de exposición al plomo.
Los niveles de plomo presentes en el aire, los alimentos, el
agua, y el suelo/polvo varían ampliamente en el mundo y dependen del
grado de desarrollo industrial y de urbanización y de factores
relacionados con el modo de vida. Se han comunicado niveles superiores
a 10 µg/m3 presentes en el aire ambiental en zonas urbanas próximas
a funderías, mientras que en ciudades donde ha dejado de usarse la
gasolina con plomo se han detectado niveles inferiores a 0,2 µg/m3.
La absorción de plomo del aire puede, pues, variar de menos de
4 µg/día a más de 200 µg/día.
Los niveles de plomo en muestras de agua de bebida extraídas de
los manantiales suelen ser inferiores a 5 µg/litro. Sin embargo, el
agua del grifo de viviendas cuyas tuberías tienen plomo contiene
niveles que exceden de 100 µg/litro, en particular después de haber
reposado el agua en las tuberías durante algunas horas.
El nivel de exposición al plomo a través de la dieta depende de
muchos factores relacionados con el modo de vida, entre ellos los
alimentos que se consumen, la tecnología de elaboración, el empleo de
soldadura de plomo, los niveles de plomo en el agua y la utilización
de cerámica con barniz de plomo.
Para los niños, el plomo presente en el polvo y en el suelo suele
ser la principal vía de exposición. Los niveles de plomo en el polvo
dependen de factores tales como la antigüedad y el estado de la
vivienda, la utilización de pinturas a base de plomo, el plomo de la
gasolina y la densidad urbana. La absorción de plomo dependerá de la
edad y de las características comportamentales del niño así como de la
biodisponibilidad de plomo en la fuente material.
La inhalación es la vía principal de exposición al plomo para los
trabajadores de industrias que producen, refinan, utilizan o desechan
plomo y compuestos de plomo. Durante un turno de ocho horas, los
trabajadores pueden absorber nada menos que 400 µg de plomo, además de
los 20-30 µg/día que absorben de los alimentos, del agua y del aire
ambiental; puede haber una absorción notable como resultado de la
inhalación de partículas grandes.
5. Cinética y metabolismo en animales de laboratorio y en el ser
humano
Los seres humanos y los animales absorben plomo por inhalación o
por ingestión; la absorción percutánea es mínima en el ser humano.
Según la especiación química, el tamaño de las partículas y la
solubilidad de los líquidos corporales, puede absorberse hasta un 50%
de los compuestos de plomo inhalados. Algunas partículas de materia
inhaladas (de más de 7 µm) se degluten después de la eliminación
mucociliar del aparato respiratorio. En los animales experimentales y
en el ser humano, la absorción de plomo del aparato gastrointestinal
está influenciada por la naturaleza fisicoquímica del material
ingerido, el estado nutricional y el tipo de alimentación. En los
seres humanos adultos, se absorbe aproximadamente el 10% del plomo
contenido en la alimentación; la proporción es más elevada en
condiciones de ayuno. Sin embargo, los lactantes y los niños pequeños
absorben nada menos que el 50% del plomo presente en la alimentación,
pero la absorción de plomo del polvo/suelo y de desconchones de
pintura puede ser menor y depende de su biodisponibilidad. Las dietas
pobres en calcio, fosfato, selenio o zinc pueden dar lugar a una mayor
absorción de plomo. El hierro y la vitamina D también influyen en la
absorción de plomo.
Los niveles de plomo en la sangre (Pb-H) se utilizan para medir
la carga corporal y las dosis absorbidas (internas) de plomo. La
relación entre el plomo presente en la sangre y la concentración de
plomo en las fuentes de exposición es curvilínea.
Una vez absorbido, el plomo no se distribuye de manera homogénea
en todo el cuerpo. Hay una absorción rápida en la sangre y en los
tejidos blandos, seguida de una redistribución más lenta a los huesos.
Los huesos acumulan plomo durante gran parte de la vida humana y
pueden actuar como fuente endógena de plomo. La semivida del plomo en
la sangre y en otros tejidos blandos es de aproximadamente 28-36 días,
pero es mucho más larga en los diversos compartimentos óseos. La
retención porcentual de plomo en los depósitos corporales es más
elevada en los niños que en los adultos. La transferencia de plomo al
feto humano se efectúa fácilmente durante la gestación.
El nivel de plomo en la sangre es la medida más utilizada para
determinar la exposición al plomo. Sin embargo, ya se dispone de
técnicas para determinar la cantidad de plomo presente en los dientes
y en los huesos, aunque aún no se conoce del todo su cinética.
6. Efectos en los animales de laboratorio y en los sistemas in vitro
En todas las especies de animales de experimentación estudiadas,
inclusive en primates no humanos, se ha observado que el plomo tiene
efectos adversos en varios órganos y sistemas de órganos, inclusive
los sistemas hematopoyético, nervioso, renal, cardiovascular,
reproductivo e inmunitario. El plomo también afecta a los huesos y se
ha demostrado que es carcinógeno en ratas y ratones.
Pese a diferencias cinéticas entre las especies de animales
experimentales y la humana, dichos estudios apoyan firmemente y
justifican desde un punto de vista biológico los hallazgos realizados
en seres humanos. Se han comunicado deficiencias del aprendizaje y de
la memoria en ratas con niveles de Pb-H de 0,72-0,96 µmoles/litro
(15-20 µg/dl) y en primates no humanos con niveles de Pb-H de no más
de 0,72 µmoles/litro (15 µg/dl). Además, se han comunicado
deficiencias visuales y auditivas en estudios realizados en animales
de experimentación.
La toxicidad renal en las ratas parece presentarse a partir de un
nivel de Pb-H de 2,88 µmoles/litro (60 µg/dl); este valor es semejante
al que, según se ha comunicado, comienza a tener efectos renales en el
ser humano. Se han observado efectos cardio-vasculares en ratas
después de exposiciones crónicas a niveles bajos que dan lugar a
niveles de Pb-H de 0,24-1,92 µmoles/litro (5-40 µg/dl). Se ha
demostrado que aparecen tumores con dosis inferiores a la dosis máxima
tolerada, de 200 mg de plomo (como acetato de plomo) por litro de agua
de bebida. Esta es la dosis máxima no asociada a otros cambios
morfológicos o funcionales.
7. Efectos en el ser humano
En el ser humano, el plomo puede tener una amplia variedad de
efectos biológicos según el nivel y la duración de la exposición. Se
han observado efectos en el plano subcelular y efectos en el
funcionamiento general del organismo que van desde la inhibición de
las enzimas hasta la producción de acusados cambios morfológicos y la
muerte. Dichos cambios se producen a dosis muy diferentes; en general,
el ser humano que se está desarrollando es más sensible que el adulto.
Se ha mostrado que el plomo tiene efectos en muchos procesos
bioquímicos; en particular, se han estudiado mucho los efectos en la
síntesis del hemo en adultos y niños. Se observan niveles más altos de
porfirina eritrocitaria sérica y mayor excreción urinaria de
coproporfirina y de ácido delta-aminolevulínico cuando las
concentraciones de Pb-H son elevadas. Con niveles más bajos se observa
inhibición de las enzimas dehidratasa del ácido delta-aminolevulínico
y reductasa de la dihidrobiopterina.
Como resultado de los efectos del plomo en el sistema
hematopoyético disminuye la síntesis de hemoglobina y se ha observado
anemia en niños a concentraciones de Pb-H superiores a
1,92 µmoles/litro (40 µg/dl).
Por razones neurológicas, metabólicas y comportamentales, los
niños son más vulnerables a los efectos del plomo que los adultos. Se
han efectuado estudios epidemiológicos prospectivos y transversales
para evaluar la medida en que la exposición al plomo ambiental afecta
a las funciones psicológicas regidas por el sistema nervioso central
(SNC). Se ha mostrado que el plomo está asociado a deficiencias
neurocomportamentales en los niños.
Se han observado deficiencias psicológicas y
neurocomportamentales en trabajadores que habían estado expuestos al
plomo durante un tiempo prolongado. Los parámetros electrofisiológicos
han demostrado ser indicadores útiles de los efectos subclínicos del
plomo en el SNC.
Desde hace tiempo se sabe que la exposición prolongada a niveles
elevados de plomo en el medio laboral provoca neuropatías periféricas.
Con niveles más bajos se ha observado una reducción de la velocidad de
conducción nerviosa. Se ha observado a menudo que dichos efectos son
reversibles después de cesar la exposición, según la edad del sujeto y
la duración de la exposición.
Los efectos del plomo en el corazón son indirectos y se producen
por conducto del sistema nervioso autónomo; el plomo no tiene efectos
directos en el miocardio. Datos colectivos procedentes de estudios de
poblaciones adultas indican asociaciones muy débiles entre la
concentración de Pb-H y la presión arterial sistólica o diastólica.
Dada la dificultad de evaluar el influjo de los factores de confusión
pertinentes, no puede establecerse una relación causal sobre la base
de esos estudios. No hay indicios de que la relación entre la
concentración de Pb-H y la presión arterial tenga mucha importancia
para la salud.
Se sabe que el plomo provoca en los tubos proximales del riñón
lesiones que se caracterizan por aminoaciduria generalizada,
hipofosfatemia con hiperfosfaturia relativa y glucosuria acompañada de
cuerpos de inclusión nuclear, modificaciones mitocondriales y
citomegalia de las células epiteliales de los tubos proximales. Los
efectos tubulares se manifiestan después de una exposición
relativamente breve y suelen ser reversibles, mientras que los cambios
escleróticos y la fibrosis intersticial, que dan lugar a una
disminución de la función renal y a una posible insuficiencia renal,
requieren una exposición crónica a niveles elevados de plomo. Se ha
advertido un mayor riesgo de nefropatía en los trabajadores que tienen
niveles de Pb-H superiores a 3,0 µmoles/litro (aproximadamente
60 µg/dl). Recientemente se han observado efectos renales en la
población general tras haberse utilizado indicadores de función más
sensibles.
Los efectos del plomo en la función reproductora masculina se
limitan a la morfología y el número de los espermatozoides. En cuanto
a la femenina, se han atribuido al plomo algunos efectos adversos en
el embarazo.
El plomo no parece tener efectos nocivos en la piel, en los
músculos ni en el sistema inmunitario. Salvo en la rata, el plomo no
parece estar relacionado con el desarrollo de tumores.
8. Evaluación de los riesgos para la salud humana
El plomo tiene efectos adversos en varios órganos y sistemas de
órganos; los más delicados parecen ser los cambios subcelulares y los
efectos en el desarrollo del sistema nervioso. Se ha observado una
asociación entre el nivel de Pb-H y la hipertensión (presión
arterial). El plomo produce una serie de efectos en la reserva
corporal de hemo y afecta a la síntesis de éste. Sin embargo, algunos
de estos efectos no se consideran adversos. Está afectada la
homeostasia del calcio, lo que interfiere en otros procesos celulares.
a) Los datos más importantes de los estudios transversales y
prospectivos de poblaciones con niveles de Pb-H generalmente
inferiores a 1,2 µmoles/litro (25 µg/dl) se relacionan con una
disminución del coeficiente de inteligencia (CI). Es importante
señalar que esas observaciones no pueden constituir una prueba
concluyente de una relación causal con la exposición al plomo.
Sin embargo, la magnitud del efecto aparente en el CI,
determinado desde los 4 años en adelante, es un déficit de 0 a
5 puntos (en una escala con una desviación estándar de 15) por
cada 0,48 µmoles/litro (10 µg/dl) de aumento del nivel de Pb-H,
con una magnitud probable del efecto aparente de 1 a 3 puntos.
Con niveles de Pb-H superiores a 1,2 µmoles/litro (25 µg/dl), la
relación entre el Pb-H y el CI puede ser diferente. Las
estimaciones de la magnitud del efecto constituyen promedios
grupales y sólo se aplican a cada niño de manera probabilística.
Los estudios epidemiológicos existentes no prueban de modo
concluyente la existencia de un umbral. Por debajo de unos
niveles de Pb-H de 0,48-0,72 µmoles/litro (10-15 µg/dl), los
efectos de las variables de confusión y los límites de la
precisión de las mediciones analíticas y psicométricas aumentan
la incertidumbre inherente a toda estimación de un efecto. Sin
embargo, hay algunos indicios de una asociación por debajo de
dichos niveles.
b) Los estudios realizados en animales respaldan la idea de una
relación causal entre el plomo y ciertos efectos en el sistema
nervioso; se señalan deficiencias cognitivas a niveles de Pb-H de
sólo 0,53-0,72 µmoles/litro (11-15 µg/dl), deficiencias que
pueden persistir mucho después de haber terminado la exposición
al plomo.
c) Puede producirse una reducción de la velocidad de conducción
nerviosa periférica en el ser humano con niveles de Pb-H de sólo
1,44 µmoles/litro (30 µg/dl). Además, las funciones
sensitivomotrices pueden verse disminuidas con niveles de Pb-H de
sólo 1,92 µmoles/litro (40 µg/dl) aproximadamente y las funciones
del sistema nervioso autónomo (variabilidad del intervalo R-R
electrocardiográfico) pueden verse afectadas a un nivel promedio
de Pb-H de aproximadamente 1,68 µmoles por litro (35 µg/dl). El
riesgo de nefropatía plúmbica aumenta en los trabajadores que
tienen niveles de Pb-H superiores a 2,88 µmoles/litro (60 µg/dl).
Sin embargo, estudios recientes que utilizan indicadores más
sensibles de la función renal sugieren la aparición de efectos
renales a niveles más bajos de exposición al plomo.
d) La exposición al plomo está asociada a un pequeño aumento de la
presión arterial. El orden de magnitud probable es que por cada
duplicación del nivel de Pb-H (por ejemplo, de 0,8 a
1,6 µmoles/litro, es decir, de 16,6 a 33,3 µg/dl) hay un aumento
medio de 1 mmHg de presión arterial sistólica. La relación con la
presión diastólica es de una magnitud semejante pero más pequeña.
Sin embargo, no se sabe bien si estas asociaciones estadísticas
obedecen realmente a un efecto de la exposición al plomo o son un
resultado ficticio debido a factores de confusión.
e) Algunos estudios epidemiológicos, no todos, muestran una
relación, dependiente de la dosis, con el parto prematuro y
algunos índices de crecimiento y maduración fetales a niveles de
Pb-H de 0,72 µmoles/litro (15 µg/dl) o más.
f) Los indicios de carcinogenicidad del plomo y de varios compuestos
inorgánicos de plomo en el ser humano son insuficientes.
g) Se ha demostrado que el plomo tiene efectos en cierto número de
sistemas enzimáticos y de parámetros bioquímicos. Los niveles de
Pb-H por encima de los cuales las técnicas vigentes pueden
demostrar la presencia de efectos en relación con los parámetros
de importancia clínica posible son todos superiores a
0,96 µmoles/litro (20 µg/dl). Algunos efectos en enzimas pueden
demostrarse con niveles de Pb-H más bajos, pero su importancia
clínica es incierta.
 It is accepted that the following criteria should initiate the
updating of an EHC monograph: new data are available that would
substantially change the evaluation; there is public concern for
health or environmental effects of the agent because of greater
exposure; an appreciable time period has elapsed since the last
evaluation.
All Participating Institutions are informed, through the EHC
progress report, of the authors and institutions proposed for the
drafting of the documents. A comprehensive file of all comments
received on drafts of each EHC monograph is maintained and is
available on request. The Chairpersons of Task Groups are briefed
before each meeting on their role and responsibility in ensuring that
these rules are followed.
WHO TASK GROUP ON ENVIRONMENTAL HEALTH CRITERIA FOR INORGANIC LEAD
Members
Professor S. Araki, Department of Public Health, Faculty of Medicine,
University of Tokyo, Japan
Dr P. Baghurst, Division of Human Nutrition, Commonwealth Scientific
Industrial Research Organization, Adelaide, Australia
Dr D. Bellinger, Neuroepidemiology Unit, Gardner House, Children's
Hospital, Boston, Massachusetts, USA
Dr I. Calder, Occupational and Environmental Health, South Australian
Health Commission, Adelaide, South Australia, Australia
Dr D.A. Cory-Slechta, Department of Environmental Medicine,
University of Rochester School of Medicine and Dentistry,
Rochester, New York, USA
Dr K. Dietrich, Department of Environmental Health, Division of
Biostatistics and Epidemiology, University of Cincinnati College
of Medicine, Cincinnati, Ohio, USA
Dr R.A. Goyer, Chapel Hill, North Carolina, USA (Chairman)
Dr M.R. Moore, University of Glasgow, Department of Medicine and
Therapeutics, Western Infirmary, Glasgow, Scotland
Dr C. Nam Ong, Department of Community, Occupational and Family
Medicine, National University of Singapore, National University
Hospital, Singapore
Dr S.J. Pocock, Department of Epidemiology and Population Sciences,
Medical Statistics Unit, University of London, London, England
Dr M.B. Rabinowitz, Marine Biological Laboratory, Woods Hole,
Massachusetts, USA
Dr M. Smith, Thomas Coram Research Unit, London, England
Dr G. Winneke, Medical Institute for Environmental Health,
Heinrich-Heine University, Düsseldorf, Germany (Vice-Chairman)
Observers
Dr C. Boreiko, Environmental Health, International Lead Zinc Research
Organization (ILZRO) Inc., Research Triangle Park, North
Carolina, USA
Dr N.H. Clark, Lead Industry Environment and Health Forum, Melbourne,
Victoria, Australia
Dr J.M. Davis, Environmental Criteria and Assessment Office, US
Environmental Protection Agency, Research Triangle Park, North
Carolina, USA
Professor G. Duggin, Toxicology Unit, Royal Prince Alfred Hospital,
Camperdown, Australia
Dr G.R. Neville, Queensland Health Department, Brisbane, Australia
Secretariat
Dr G.C. Becking, International Programme on Chemical Safety,
Interregional Research Unit, World Health Organization, Research
Triangle Park, North Carolina, USA (Secretary)
Dr K.R. Mahaffey, National Institute of Environmental Health
Sciences, Research Triangle Park, North Carolina, USAa
Dr A.E. Robinson, Toronto, Ontario, Canada (Rapporteur)
a Present address: US Environmental Protection Agency, Environmental
Criteria and Assessment Office, Cincinnati, Ohio, USA
ENVIRONMENTAL HEALTH CRITERIA FOR INORGANIC LEAD
A WHO Task Group on Environmental Health Criteria for Inorganic
Lead met in Brisbane, Australia, from 1 to 6 February 1993. The
meeting was sponsored by a consortium of Australian Commonwealth and
State Governments through a national Steering Committee chaired by
Dr Keith Bentley, Director, Health and Environmental Policy,
Department of Human Services and Health, Canberra. The meeting was
hosted and organized by the Queensland Department of Health,
Dr G.R. Neville being responsible for the arrangements. Dr G. Murphy,
Director of Public Health, Queensland, welcomed the participants on
behalf of the Organizers, and Dr T. Adams, Chief Commonwealth Medical
Advisor and Dr G. Johns, Parliamentary Secretary to Federal Minister
for Health, Housing and Community Services, welcomed the participants
on behalf of the Commonwealth Government. Dr Johns stressed the
importance attached to this IPCS meeting by the Commonwealth and State
Governments of Australia. Dr G.C. Becking, IPCS, welcomed the
participants on behalf of Dr M. Mercier, Director of the IPCS and the
three cooperating organizations (UNEP/ILO/WHO). The Task Group
reviewed and revised the draft criteria monograph, and made an
evaluation of the risks to human health from exposure to inorganic
lead.
The Task Group draft was prepared by Dr A.E. Robinson, Toronto,
Canada, using texts made available by Dr K.R. Mahaffeya (National
Institute of Environmental Health Sciences, Research Triangle Park,
North Carolina, USA) and Dr E. Silbergeld (University of Maryland
School of Medicine, Baltimore, Maryland, USA), and the comments
received from the IPCS contact points for environmental health
criteria monographs. The draft was revised extensively by the Task
Group taking into account the comments from the IPCS contact points.
Dr G.C. Becking (IPCS Central Unit, Interregional Research Unit)
and Dr P.G. Jenkins (IPCS Central Unit, Geneva) were responsible for
the overall scientific content and technical editing, respectively, of
this monograph.
The efforts of all who helped in the preparation and finalization
of this publication are gratefully acknowledged.
a Present address: US Environmental Protection Agency, Environmental
Criteria and Assessment Office, Cincinnati, Ohio, USA
ABBREVIATIONS
AAS atomic absorption spectrometry
AES atomic emission spectroscopy
ALA delta-aminolaevulinic acid
ALAD delta-aminolaevulinic acid dehydratase
ASV anodic stripping voltametry
EDTA ethylenediaminetetraacetic acid
FEP free erythrocyte porphyrin
GFAAS graphite furnace atomic absorption spectrometry
ICP inductively coupled plasma
IDMS isotope dilution mass spectrometry
MPb mobilization yield of lead
MSW municipal solid waste
PbB blood lead
PbT tooth lead
TML tetramethyllead
XRFS X-ray fluorescence spectroscopy
ZPP zinc protoporphyrin
PREFACE
Although many countries have initiated programmes to lower the
level of lead in the environment, human exposure to lead remains of
concern to public health officials worldwide. For over 20 years the
World Health Organization (WHO) and the International Programme on
Chemical Safety (IPCS) have been concerned about the health and
environmental effects of the levels of inorganic lead in the
environment. The evaluation of human health risks arising from
food-borne lead has been carried out by WHO on four occasions since
1972. In addition, health-based guidance values for lead in water, air
and the workplace have been developed by various Task Groups convened
by WHO. Environmental Health Criteria 3: Lead, published in 1977,
examined the effects of lead on human health and Environmental Health
Criteria 85: Lead - Environmental Aspects was published in 1989.
Since the publication of Environmental Health Criteria 3: Lead, a
large body of knowledge has accumulated concerning the effects of lead
on humans at low levels of exposure. Studies have emphasized the
effects of inorganic lead on infants and children, a high-risk
population. This monograph on inorganic lead reflects this research
emphasis; a major part of the monograph deals with the neurotoxic
effects of lead with emphasis on neurobehavioural development in
children. Less detail is presented on the health effects of the higher
levels of inorganic lead found in some workplaces, although such
exposures are still considered to pose a risk to humans in many
regions of the world.
This monograph deals only with the human health effects of
inorganic lead. No attempt has been made to evaluate the human health
effects of organo-lead compounds, although it was recognized that such
compounds when added to petrol (gasoline) are a major source of
inorganic lead in the environment. In view of the toxicity of many
organo-lead derivatives and the possible methylation of inorganic lead
in the environment, the IPCS plans to evaluate the risk to humans from
exposure to organo-lead compounds in a separate monograph.
As with all IPCS criteria monographs, no attempt has been made to
prepare an exhaustive bibliography of the extremely large amount of
lead-related literature published since 1977. Rather, an effort has
been made to review critically the studies on humans and experimental
animals that are essential for the evaluation of risks to human health
from exposure to all sources of inorganic lead.
1. SUMMARY
This monograph focuses on the risks to human health associated
with exposure to lead and inorganic lead compounds. Emphasis has been
given to data which have become available since the publication of
Environmental Health Criteria 3: Lead (IPCS, 1977). The environmental
effects of lead are discussed in Environmental Health Criteria 85:
Lead - Environmental Aspects (IPCS, 1989).
1.1 Identity, physical and chemical properties, and analytical
methods
Lead is a soft, silvery grey metal, melting at 327.5°C. It is
highly resistant to corrosion, but is soluble in nitric and hot
sulfuric acids. The usual valence state in inorganic lead compounds is
+2. Solubilities in water vary, lead sulfide and lead oxides being
poorly soluble and the nitrate, chlorate and chloride salts are
reasonably soluble in cold water. Lead also forms salts with such
organic acids as lactic and acetic acids, and stable organic compounds
such as tetraethyllead and tetramethyllead.
The most commonly used methods for the analysis of low
concentrations of lead in biological and environmental materials are
flame, graphite furnace and inductively coupled plasma atomic
absorption spectroscopy and anode stripping voltametry. Depending on
sample pretreatment, extraction techniques and analytical
instrumentation, detection limits of 0.12 µmoles lead/litre blood
(2.49 µg/dl) can be achieved. However, reliable results are obtained
only when specific procedures are followed to minimize the risk of
contamination during sample collection, storage, processing and
analysis.
1.2 Sources of human exposure
The level of lead in the earth's crust is about 20 mg/kg. Lead in
the environment may derive from either natural or anthropogenic
sources. Natural sources of atmospheric lead include geological
weathering and volcanic emissions and have been estimated at
19 000 tonnes/year, compared to an estimate of 126 000 tonnes/year
emitted to the air from the mining, smelting and consumption of over 3
million tonnes of lead per year.
Atmospheric lead concentrations of 50 pg/m3 have been found in
remote areas. Background levels of lead in soil range between 10 and
70 mg/kg and a mean level near roadways of 138 mg/kg has been
reported. Present levels of lead in water rarely exceed a few
micrograms/litre; the natural concentration of lead in surface water
has been estimated to be 0.02 µg/litre.
Lead and its compounds may enter the environment at any point
during mining, smelting, processing, use, recycling or disposal. Major
uses are in batteries, cables, pigments, petrol (gasoline) additives,
solder and steel products. Lead and lead compounds are also used in
solder applied to water distribution pipes and to seams of cans used
to store foods, in some traditional remedies, in bottle closures for
alcoholic beverages and in ceramic glazes and crystal tableware. In
countries where leaded petrol is still used, the major air emission is
from mobile and stationary sources of petrol combustion (urban
centres). Areas in the vicinity of lead mines and smelters are subject
to high levels of air emissions.
Airborne lead can be deposited on soil and water, thus reaching
humans through the food chain and in drinking-water. Atmospheric lead
is also a major source of lead in household dust.
1.3 Environmental transport, distribution and transformation
The transport and distribution of lead from fixed, mobile and
natural sources are primarily via air. Most lead emissions are
deposited near the source, although some particulate matter (< 2 µm
in diameter) is transported over long distances and results in the
contamination of remote sites such as arctic glaciers. Airborne lead
can contribute to human exposures by the contamination of food, water
and dust, as well as through direct inhalation. The removal of
airborne lead is influenced by atmospheric conditions and particulate
size. Large amounts of lead may be discharged to soil and water.
However, such material tends to remain localized because of the poor
solubility of lead compounds in water.
Lead deposited in water, whether from air or through run-off from
soils, partitions rapidly between sediment and aqueous phase,
depending upon pH, salt content, and the presence of organic chelating
agents. Above pH 5.4, hard water may contain about 30 µg lead/litre
and soft water about 500 µg lead/litre. Very little lead deposited on
soil is transported to surface or ground water except through erosion
or geochemical weathering; it is normally quite tightly bound
(chelated) to organic matter.
Airborne lead can be transferred to biota directly or through
uptake from soil. Animals can be exposed to lead directly through
grazing and soil ingestion or by inhalation. There is little
biomagnification of inorganic lead through the food chain.
1.4 Environmental levels and human exposure
In the general non-smoking adult population, the major exposure
pathway is from food and water. Airborne lead may contribute
significantly to exposure, depending upon such factors as use of
tobacco, occupation, proximity to motorways, lead smelters, etc., and
leisure activities (e.g., arts and crafts, firearm target practice).
Food, air, water and dust/soil are the major potential exposure
pathways for infants and young children. For infants up to 4 or 5
months of age, air, milk, formulae and water are the significant
sources of lead exposure.
Levels of lead found in air, food, water and soil/dust vary
widely throughout the world and depend upon the degree of industrial
development, urbanization and lifestyle factors. Ambient air levels
over 10 µg/m3 have been reported in urban areas near a smelter,
whereas lead levels below 0.2 µg/m3 have been found in cities where
leaded petrol is no longer used. Lead intake from air can, therefore,
vary from less than 4 µg/day to more than 200 µg/day.
Levels of lead in drinking-water sampled at the source are
usually below 5 µg/litre. However, water taken from taps (faucets) in
homes where lead is present in the plumbing can contain levels in
excess of 100 µg/litre, particularly after the water has been standing
in the pipes for some hours.
The level of dietary exposure to lead depends upon many lifestyle
factors, including foodstuffs consumed, processing technology, use of
lead solder, lead levels in water, and use of lead-glazed ceramics.
For infants and children, lead in dust and soil often constitutes
a major exposure pathway. Lead levels in dust depend upon such factors
as the age and condition of housing, the use of lead-based paints,
lead in petrol and urban density. The intake of lead will be
influenced by the age and behavioural characteristics of the child and
bioavailability of lead in the source material.
Inhalation is the dominant pathway for lead exposure of workers
in industries producing, refining, using or disposing of lead and lead
compounds. During an 8-h shift, workers can absorb as much as 400 µg
lead, in addition to the 20-30 µg/day absorbed from food, water and
ambient air; significant intake may occur from ingestion of large
inhaled particulate material.
1.5 Kinetics and metabolism in laboratory animals and humans
Lead is absorbed in humans and animals following inhalation or
ingestion; percutaneous absorption is minimal in humans. Depending
upon chemical speciation, particle size, and solubility in body
fluids, up to 50% of the inhaled lead compound may be absorbed. Some
inhaled particulate matter (larger than 7 µm) is swallowed following
mucociliary clearance from the respiratory tract. In experimental
animals and humans, absorption of lead from the gastrointestinal tract
is influenced by the physico-chemical nature of the ingested material,
nutritional status, and type of diet consumed. In adult humans
approximately 10% of the dietary lead is absorbed; the proportion is
higher under fasting conditions. However, in infants and young
children as much as 50% of dietary lead is absorbed, although
absorption rates for lead from dusts/soils and paint chips can be
lower depending upon the bioavailability. Diets that are deficient in
calcium, phosphate, selenium or zinc may result in increased lead
absorption. Iron and vitamin D also affect absorption of lead.
Blood lead (PbB) levels are used as a measure of body burden and
absorbed (internal) doses of lead. The relationship between blood lead
and the concentration of lead in exposure sources is curvilinear.
Once it has been absorbed, lead is not distributed homogeneously
throughout the body. There is rapid uptake into blood and soft tissue,
followed by a slower redistribution to bone. Bone accumulates lead
over much of the human life span and may serve as an endogenous source
of lead. The half-life for lead in blood and other soft tissues is
about 28-36 days, but it is much longer in the various bone
compartments. The percentage retention of lead in body stores is
higher in children than adults. Transfer of lead to the human fetus
occurs readily throughout gestation.
Blood lead is the most commonly used measure of lead exposure.
However, techniques are now available for measuring lead in teeth and
bone, although the kinetics are not fully understood.
1.6 Effects on laboratory animals and in vitro systems
In all species of experimental animals studied, including
non-human primates, lead has been shown to cause adverse effects in
several organs and organ systems, including the haematopoietic,
nervous, renal, cardiovascular, reproductive and immune systems. Lead
also affects bone and has been shown to be carcinogenic in rats and
mice.
Despite kinetic differences between experimental animal species
and humans, these studies provide strong biological support and
plausibility for the findings in humans. Impaired learning/memory
abilities have been reported in rats with PbB levels of
0.72-0.96 µmoles/litre (15-20 µg/dl) and in non-human primates at PbB
levels not exceeding 0.72 µmoles/litre (15 µg/dl). In addition, visual
and auditory impairments have been reported in experimental animal
studies.
Renal toxicity in rats appears to occur at a PbB level in excess
of 2.88 µmol/litre (60 µg/dl), a value similar to that reported to
initiate renal effects in humans. Cardiovascular effects have been
seen in rats after chronic low-level exposures resulting in PbB levels
of 0.24-1.92 µmol/litre (5-40 µg/dl). Tumours have been shown to occur
at dose levels below the maximum tolerated dose of 200 mg lead (as
lead acetate) per litre of drinking-water. This is the maximum dose
level not associated with other morphological or functional changes.
1.7 Effects on humans
In humans, lead can result in a wide range of biological effects
depending upon the level and duration of exposure. Effects at the
subcellular level, as well as effects on the overall functioning of
the body, have been noted and range from inhibition of enzymes to the
production of marked morphological changes and death. Such changes
occur over a broad range of doses, the developing human generally
being more sensitive than the adult.
Lead has been shown to have effects on many biochemical
processes; in particular, effects on haem synthesis have been studied
extensively in both adults and children. Increased levels of serum
erythrocyte protoporphyrin and increased urinary excretion of
coproporphyrin and delta-aminolaevulinic acid are observed when PbB
concentrations are elevated. Inhibition of the enzymes
delta-aminolaevulinic acid dehydratase and dihydrobiopterin reductase
are observed at lower levels.
The effects of lead on the haemopoietic system result in
decreased haemoglobin synthesis, and anaemia has been observed in
children at PbB concentrations above 1.92 µmol/litre (40 µg/dl).
For neurological, metabolic and behavioural reasons, children are
more vulnerable to the effects of lead than adults. Both prospective
and cross-sectional epidemiological studies have been conducted to
assess the extent to which environmental lead exposure affects
CNS-based psychological functions. Lead has been shown to be
associated with impaired neurobehavioural functioning in children.
Impairment of psychological and neurobehavioural functions has
been found after long-term lead exposure of workers.
Electrophysiological parameters have been shown to be useful
indicators of subclinical lead effects in the CNS.
Peripheral neuropathy has long been known to be caused by
long-term high-level lead exposure at the workplace. Slowing of nerve
conduction velocity has been found at lower levels. These effects have
often been found to be reversible after cessation of exposure,
depending on the age and duration of exposure.
The effect of lead on the heart is indirect and occurs via the
autonomic nervous system; it has no direct effect on the myocardium.
The collective evidence from population studies in adults indicates
very weak associations between PbB concentration and systolic or
diastolic blood pressure. Given the difficulties of allowing for
relevant confounding factors, a causal relationship cannot be
established from these studies. There is no evidence to suggest that
any association of PbB concentration with blood pressure is of major
health importance.
Lead is known to cause proximal renal tubular damage,
characterized by generalized aminoaciduria, hypophosphataemia with
relative hyperphosphaturia and glycosuria accompanied by nuclear
inclusion bodies, mitochondrial changes and cytomegaly of the proximal
tubular epithelial cells. Tubular effects are noted after relatively
short-term exposures and are generally reversible, whereas sclerotic
changes and interstitial fibrosis, resulting in decreased kidney
function and possible renal failure, require chronic exposure to high
lead levels. Increased risk from nephropathy was noted in workers with
a PbB level of over 3.0 µmol/litre (about 60 µg/dl). Renal effects
have recently been seen among the general population when more
sensitive indicators of function were measured.
The reproductive effects of lead in the male are limited to sperm
morphology and count. In the female, some adverse pregnancy outcomes
have been attributed to lead.
Lead does not appear to have deleterious effects on skin, muscle
or the immune system. Except in the case of the rat, lead does not
appear to be related to the development of tumours.
1.8 Evaluation of human health risks
Lead adversely affects several organs and organ systems, with
subcellular changes and neurodevelopmental effects appearing to be the
most sensitive. An association between PbB level and hypertension
(blood pressure) has been reported. Lead produces a cascade of effects
on the haem body pool and affects haem synthesis. However, some of
these effects are not considered adverse. Calcium homoeostasis is
affected, thus interfering with other cellular processes.
a) The most substantial evidence from cross-sectional and
prospective studies of populations with PbB levels generally
below 1.2 µmol/litre (25 µg/dl) relates to decrements in
intelligence quotient (IQ). It is important to note that such
observational studies cannot provide definitive evidence of a
causal relationship with lead exposure. However, the size of the
apparent IQ effect, as assessed at 4 years and above, is a
deficit between 0 and 5 points (on a scale with a standard
deviation of 15) for each 0.48 µmol/litre (10 µg/dl) increment in
PbB level, with a likely apparent effect size of between 1 and 3
points. At PbB levels above 1.2 µmol/litre (25 µg/dl), the
relationship between PbB and IQ may differ. Estimates of effect
size are group averages and only apply to the individual child in
a probabilistic manner.
Existing epidemiological studies do not provide definitive
evidence of a threshold. Below the PbB range of 0.48-0.72 µmol/
litre (10-15 µg/dl), the effects of confounding variables and
limits in the precision in analytical and psychometric
measurements increase the uncertainty attached to any estimate
of effect. However, there is some evidence of an association
below this range.
b) Animal studies provide support for a causal relationship between
lead and nervous system effects, reporting deficits in cognitive
functions at PbB levels as low as 0.53-0.72 µmol/litre
(11-15 µg/dl) which can persist well beyond the termination of
lead exposure.
c) Reduction in human peripheral nerve conduction velocity may occur
with PbB levels as low as 1.44 µmol/litre (30 µg/dl). In
addition, sensory motor function may be impaired with PbB levels
as low as about 1.92 µmol/litre (40 µg/dl), and autonomic nervous
system function (electrocardiographic R-R interval variability)
may be affected at an average PbB level of approximately
1.68 µmol/litre (35 µg/dl). The risk of lead nephropathy is
increased in workers with PbB levels above 2.88 µmol/litre
(60 µg/dl). However, recent studies using more sensitive
indicators of renal function suggest renal effects at lower
levels of lead exposure.
d) Lead exposure is associated with a small increase in blood
pressure. The likely order of magnitude is that for any two-fold
increase in PbB level (e.g., from 0.8 to 1.6 µmol/litre, i.e.
16.6 to 33.3 µg/dl), there is a mean 1 mmHg increase in systolic
blood pressure. The association with diastolic pressure is of a
similar but smaller magnitude. However, there is doubt regarding
whether these statistical associations are really due to an
effect of lead exposure or are an artifact due to confounding
factors.
e) Some but not all epidemiological studies show a dose dependent
association of pre-term delivery and some indices of fetal growth
and maturation at PbB levels of 0.72 µmol/litre (15 µg/dl) or
more.
f) The evidence for carcinogenicity of lead and several inorganic
lead compounds in humans is inadequate.
g) Effects of lead on a number of enzyme systems and biochemical
parameters have been demonstrated. The PbB levels, above which
effects are demonstrable with current techniques for the
parameters that may have clinical significance, are all greater
than 0.96 µmol/litre (20 µg/dl). Some effects on enzymes are
demonstrable at lower PbB levels, but the clinical significance
is uncertain.
2. IDENTITY, PHYSICAL AND CHEMICAL PROPERTIES, AND ANALYTICAL METHODS
2.1 Physical and chemical properties of lead and its compounds
Lead (atomic number, 82; relative atomic mass, 207.19; specific
gravity, 11.34) is a bluish or silvery grey soft metal. The melting
point is 327.5°C and the boiling point at atmospheric pressure 1740°C.
It has four naturally occurring isotopes (208, 206, 207, and 204 in
order of abundance), but the isotopic ratios for various mineral
sources may differ. This property has been exploited in
non-radioactive-tracer environmental and metabolic studies. The
physical and chemical properties of elemental lead and some lead
compounds are summarized in Table 1.
Although lead has four electrons in its valence shell, only two
ionize readily. The usual oxidation state of lead in inorganic
compounds is therefore +2 rather than +4. The inorganic salts of lead,
such as lead sulfide and the oxides of lead, are generally poorly
soluble in water. However, the nitrate, chlorate and, to a much lesser
degree, the chloride are water soluble. Some of the salts formed with
organic acids, e.g., lead oxalate, are also insoluble, but the acetate
is relatively soluble, as shown in Table 1.
Under appropriate conditions of synthesis, stable compounds are
formed in which lead is directly bound to a carbon atom. Industrially
synthesized lead-carbon compounds include tetraethyllead and
tetramethyllead, which are of importance as fuel additives and, hence,
are sources of environmental lead.
2.2 Analytical procedures
In recent years substantial advances have been made in developing
methods for the quantification of metals at low concentrations. In
order to provide improved quality assurance of such measurements,
various reference materials in different matrices have been produced
(Muramatsu & Parr, 1985). To ensure adequate quality control, the
analyst should choose a reference material that matches as closely as
possible the experimental samples to be analysed. Choices are based
upon matrix type and concentration of the element of interest. A
summary of data on 60 biological and 40 environmental (non-biological)
reference materials has been compiled by Muramatsu & Parr (1985).
With the increased interest in measuring lead in the low µg/kg
and µg/m3 range in both environmental and biological samples, there
is need for particular attention to analytical sensitivity and
reliability. As lower concentrations are measured, problems of
laboratory contamination become more significant and quality control
and quality assurance programmes are important. Because of these
concerns, all analytical results for lead should report the laboratory
Table 1. Physical and chemical data on lead and selected lead compoundsa
Name Synonym Relative atomic/ Melting point Boiling point Solubility in cold Soluble in
and formula molecular mass (°C) (°C) water (g/litre)
Lead Pb 207.19 327.502 1740 insoluble HNO3; hot concentrated
H2SO4; hot water;
glycerine; alcohol (slightly)
Lead salts
acetate Pb(C2H3O2)2 325.28 280 - 443
carbonate cerrusite PbCO3 267.20 315 (decomposes) 0.0011 acid; alkali; decomposes in
hot water
chlorate Pb(ClO3)2 374.09 230 (decomposes) very soluble alcohol
chloride cotunite PbCl2 278.10 501 950 919 NH4 salts; slightly in dilute
HCl and in NH3; hot water
(33.4 g/litre)
nitrate Pb(NO3)2 331.20 470 (decomposes) 376.5 alcohol; alkali, NH3; hot
water (1270 g/litre)
orthophosphate Pb3(PO4)2 811.51 1014 0.00014 alkali; HNO3
oxalate PbC2O4 295.21 300 (decomposes) 0.0016 HNO3
dioxide plattnerite PbO2 239.19 290 (decomposes) insoluble dilute HCl; acetic acid
(slightly)
monoxide litharge PbO 223.19 888 0.017 dilute HNO3; acetic acid
Table 1 (cont'd)
Name Synonym Relative atomic/ Melting point Boiling point Solubility in cold Soluble in
and formula molecular mass (°C) (°C) water (g/litre)
sulfate anglesite PbSO4 303.25 1170 0.0425 NH4 salts; concentrated
H2SO4 (slightly)
sulfide galena PbS 239.25 1114 0.00086 acid
a Data from Weast (1985)
performance for reference standards and for parallel blank
measurements of sample contamination for the entire analytical
process. Without these, the validity of the data should be questioned.
2.2.1 Sampling procedures
Particular attention should be paid to the cleanliness of
equipment and glassware and the purity of the chemicals to prevent
secondary contamination by lead.
For the collection of samples, standard trace element methods are
generally required (Behne, 1980) with adequate quality control
procedures (Friberg, 1988; Jorhem & Slorach, 1988, Vahter & Friberg,
1988). Quality control samples for blood, faeces, air filters and dust
have been described (Lind et al., 1988).
2.2.1.1 Sampling of environmental media
In air sampling, both high-volume samplers and low-volume
techniques have been used. It should be noted that the collection
characteristics of high-volume samplers are strongly affected by
particle size and the orientation of the sampler. For particles larger
than 5 µm in diameter the high-volume sampler system is unlikely to
collect representative samples (US EPA, 1986a). As in all sampling for
suspended particulate matter, the accuracy of volume meters should be
checked periodically. The size of the pores in filters for collecting
lead-containing particles should be small, possibly less than 0.2 µm
for glass-fibre filters (Lee & Goranson, 1972).
Depending on the purpose of sampling, care should be taken to
select the appropriate site for sampling devices and to achieve the
best possible sampling conditions by:
* estimating the amount of particulate required for analysis before
deciding on the sample volume and the sampling procedure;
* placing the sampling devices in the appropriate position (e.g.,
in the breathing zone, level with inlet tubes of house
ventilators, at window level in the case of a traffic-laden town
street, at a reasonable distance from the highway in uninhabited
zones, etc);
* taking the samples at appropriate rates and volumes (e.g., daily
breathing volumes, daily ventilating capacities of installations)
and for a sufficient time to make possible the estimation of the
average concentration (e.g., during a work-shift, or a 24-h or
longer period for general population exposure);
* taking into account the use of areas under study (cattle grazing,
recreational zones, children's playgrounds, etc).
In addition, whenever possible a procedure should be used that
makes it possible to evaluate particle-size distribution and the
physicochemical properties of the lead compounds involved, including
the shape of the particles and the state of their aggregation.
Lead may be found in water bound to particulate matter as soluble
complexes or soluble compounds. Techniques for sampling water must
take this into account. It is necessary to sample water without
fractionation (filtration) when total lead levels are required.
Because of the potential for metals from low ionic strength waters to
be adsorbed onto the surfaces of some containers, samples should be
acidified (US EPA, 1986a). Selection, cleaning, and conditioning of
storage and sample containers deserve special attention (Moody, 1982).
The preparation of soil and dust samples for lead analyses
usually involves drying (at 100°C), homogenization by grinding, and
sieving (Thornton & Webb 1975; Bolter et al., 1975). Brown & Black
(1983) have discussed the issues related to quality assurance and
quality control in the collection and analysis of soil samples. Most
reports of lead in soil provide the total elemental abundance either
by acid extraction or X-ray fluorescence. However, the leachable or
bioavailable fraction is of special interest.
For the study of the dietary intake of lead from food, two
general methods have been utilized. The advantages and disadvantages
of the "duplicate portions" technique and the equivalent composite
technique ("market basket") have been reviewed by Pekkarinen (1970).
Although the duplicate portions (duplicate diets) technique can define
variability in consumption, it is expensive, and the sampling and
analytical procedures involved are complicated and limit the number of
individuals included in any study. With the equivalent composite
technique, the economy and ease of collection must be considered in
the light of the variability of results obtained due to uncertainties
in knowledge of actual preparation techniques, including possible lead
levels in water used for processing in individual homes.
The quantity of lead likely to be leached from ceramic surfaces
by different foods and beverages may be assessed using dilute acetic
acid solutions (1 to 4%) at temperatures in the range 20 to 100°C for
times ranging from 30 min to more than 24 h (Laurs, 1976; Merwin,
1976).
Colorimetric methods are suitable for screening inorganic
materials such as pottery or paint for lead. Positive reactions
require confirmation by established quantitative methods. Spot tests
using dithizone, rhodizonate and iodide (Feigl et al., 1972) are
available.
2.2.1.2 Sampling of biological materials
The main problem in the sampling of body fluids and tissues for
lead analysis is potential secondary contamination with lead. The low
general population blood lead (PbB) levels in many regions of the
world are complicating screening efforts, requiring levels of
analytical precision and sensitivity that can be achieved only through
intensive QA/QC programmes. Issues related to such sampling have been
examined in detail by US EPA (1986a).
Special precautions are needed to ensure that all venous
blood-collecting and blood-storage materials are as free from lead as
possible (IPCS, 1977). All glass equipment involved in blood
collection and storage should be made of lead-free silicate glass,
rinsed first in mineral acid, then with copious amounts of
glass-distilled or deionized water. Polypropylene syringes have been
recommended (NAS-NRC, l972). Needles should be of stainless steel with
polypropylene hubs. Blood is often drawn directly from the needle into
vacuum tubes. It is wise to confirm periodically the absence of
significant amounts of lead in the anticoagulant used in the blood
container as well as monitoring the contamination level (blank) for
the entire analytical process.
New analytical techniques make it possible to determine lead
concentrations in microlitre quantities of blood. The trend towards
the procurement of micro-samples of blood by skin prick increases the
chance of secondary contamination of the blood. Systematic
investigation on the significance of this problem has been reported
(Mitchell et al., 1974; Mahaffey et al., 1979; DeSilva & Donnan,
1980). Mitchell et al. (l974) describe a procedure whereby sample
contamination can be reduced by spraying collodion over the cleansed
skin before lancing. The correlation between the concentration of lead
in micro-samples and in macro-samples obtained by venepuncture was
fairly good (r=0.92) over a wide range of PbB concentrations
(0.48-4.41 µmol/litre or 10-92 µg/dl whole blood). Mahaffey et al.,
(1979b) found that capillary blood levels in a comparison test were
systematically higher than corresponding venous blood levels; similar
elevations have been reported by DeSilva & Donnan (1980). Since about
1980 the requirement for reliable and accurate micro procedures has
resulted in the development of good protocols. Sinclair & Dohnt (1984)
described a procedure which resulted in the ability to collect
capillary samples with PbB levels only 3.3% higher than the presumably
correct venous value. This procedure has been used in the Port Pirie
Cohort Study (Baghurst et al., 1985, 1992) and for routine
surveillance in the Port Pirie Lead Decontamination Program (Calder et
al., 1990). Also, Lyngbye et al. (1990b) have shown that capillary
sampling without lead contamination is possible. Routine validation by
cross-comparison with venous blood samples should be undertaken on a
regular basis.
The same general precautions to avoid contamination must be taken
in the collection of urine samples as in the collection of blood
samples. Additionally, special care must be taken to prevent
precipitation during storage.
2.2.2 Analytical methods for lead
A number of analytical methods exist for determination of lead in
environmental and biological samples. These methods differ enormously
in their costs (e.g., sophisticated equipment, an adequate
infrastructure to maintain laboratory conditions and chemical
supplies) and personnel requirements (e.g., availability of skilled
personnel in adequate numbers for the work to be undertaken). Both
accuracy and precision of any of the methods can be affected greatly
by contamination of samples within the laboratory. It is important to
utilize the principles of a "clean" laboratory described by Patterson
& Settle (1976) and Everson & Patterson (1980).
It is not the purpose of this section to provide an exhaustive
description of the analytical methods that could be available to
detect and quantify lead levels in environmental and biological
samples. However, an attempt will be made to identify well-established
methods in current use and to provide information on their application
to assist in the interpretation of experimental and epidemiological
studies.
2.2.2.1 Analysis of lead in environmental samples
The most common methods used for the analysis of lead in samples
from air, water, dust, sediment, soil and foodstuffs are flame atomic
absorption spectrometry (AAS), graphite furnace atomic absorption
spectrometry (GFAAS), anodic stripping voltametry (ASV), inductively
coupled plasma-atomic emission spectroscopy (ICP-AES), and X-ray
fluorescence spectroscopy (XRFS). The reference method for the
determination of the absolute amounts of lead is by isotope dilution
mass spectrometry (IDMS) (Settle & Patterson, 1980; Grandjean & Olsen,
1984; US EPA, 1986a), but due to equipment costs and required
expertise, it is not widely used. Spectrophotometric methods, using
diphenylthiocarbazone as the colorimetric reagent, were widely used in
the past; they are less sensitive and are labour-intensive but are
still appropriate. The advantages and disadvantages were described by
Skogerboe et al. (1977).
Gould et al. (1988) utilized a citric acid solution on filter
paper to leach lead from glazed ceramic and/or enamelled metal-ware.
When treated with a lead-sensitive chromogen, there is a reaction
indicating the presence of lead on the paper. The minimal amount of
lead required to produce an observable reaction was 0.25 µg/cm2; the
maximum amount tested was 5 µg/cm2. A colorimetric test based on the
use of sodium sulfide in solution is used to estimate lead in paint
films. It is possible to determine lead concentrations greater than
1 mg/cm2 of dried paint 90% of the time when the method is used by a
trained chemical laboratory technician.
Table 2 summarizes the utility of several representative methods
for specific environmental media.
2.2.2.2 Analysis of lead in biological materials
Biological samples present special problems for the analyst
because of the low lead concentrations and matrix effects. Most
analytical techniques developed to detect and quantify lead can be
adapted to the analysis of such biological materials as blood, urine,
serum, cerebrospinal fluid, solid tissues, hair, teeth and bone.
However, certain techniques are more often used for specific matrices.
Currently, the most commonly used methods are AAS, GFAAS, ASV,
and ICP-AES. Spectrophotometric methods were commonly used in the past
and can be useful. Other specialized methods for lead analysis are
XRFS, neutron activation analysis (NAA), inductively coupled
plasma-mass spectrometry (ICP-MS), and IDMS. Table 3 summarizes the
utility of several analytical procedures applied to various biological
matrices. Included in this table are examples of the application of
XRFS (Christoffersson et al., 1986; Wielopolski et al., 1986; Nilsson
et al., 1991) for the determination in situ of the body burden of
lead.
2.2.2.3 Analytical procedures for biomarkers of lead exposure and
effect
Using standard clinical laboratory techniques, analytical
procedures have been developed: delta-aminolaevulinic acid (ALA);
delta-aminolaevulinic acid dehydratase (ALAD); urinary coproporphyrin
(CPU) and erythrocyte protoporphyrin (EP). All of these assays are
well established and reliable (Grandjean & Olsen 1984; US EPA, 1986a).
These biochemical parameters are influenced by physiological factors
other than lead. They lack the specificity and sensitivity of PbB
measurements as an index of either current lead exposures or body
stores of lead.
2.3 Conversion factors
1 µg/dl = 0.048 µmol/litre
1 µmol/litre = 20.7 µg/dl
Using the above conversion factor, blood lead concentrations are
given as µmol/litre with the equivalent µg/dl in brackets. Calculated
figures have not been rounded and added precision is not to be
inferred from the number of significant figures.
Table 2. Analytical methods for determining lead in environmental samplesa
Sample type Preparation method Analytical method Sample detection Percentage Reference
limit recovery
Air collect particulate matter on membrane ASV with mercury-graphite 0.16 µg/m3 90-110 NIOSH (1977b)
(particulate filter; wet ash with HNO3/HClO4/H2SO4; electrode (NIOSH method
lead) dissolve in acetate buffer P&CAM 191)
Air collect particulate matter on cellulose ICP-AES (NIOSH method 0.34 µg/m3 95-105 NIOSH (1981)
(particulate acetate filter; wet ash with HNO3/HClO4 P&CAM 351)
lead)
Air collect particulate matter on filter; AAS 0.1 µg/m3 93 Scott et al.
(particulate dry ash; extract with HNO3/HCl; dilute AES 0.15 µg/m3 102 (1976)
lead) with HNO3
Air sample on cellulose acetate filter; AAS 8 ng/litre 100-101 Nerin et al.
(particulate dissolve in HNO3 with heat; add HCl/H2O2 (1989)
lead) and react in hydride generator with sodium
borohydride to generate lead hydride
Air collect sample on filter; spike filter with IDMS 0.1 ng/m3 NR Volkening et
(particulate 206Pb; dissolve filter in NaOH; acidify; al. (1988)
lead) separate lead by electrodeposition; dissolve
in acid
Water digest sample with acid; heat; dilute with AAS 1.0 ng/g NR Chau et al.
(total lead) water (1979)
Soil dry sample and sieve for XRF; digest sieved XRF NR 65-98 Krueger &
sample with HNO3 and heat for AAS AAS NR 63-68 Duguay (1989)
Soil dry sample, dry ash; digest with acid AAS 2 µg/g 79-103 Beyer &
and dilute with water Cromartie (1987)
Table 2 (cont'd)
Sample type Preparation method Analytical method Sample detection Percentage Reference
limit recovery
Soil, waste, digest sample with acid; dilute with water AAS (EPA method 7420) 0.1 mg/litre NR US EPA (1986b)
and ground and filter GFAAS (EPA method 7421) 1 µg/litre NR
water
Soil, dust digest sample with hot acid; dry; redissolve AAS 12 ng/g > 80 Que Hee et al.
and paint in HNO3 (1985b)
Sediment, fish, digest sample with acid; heat; dilute with AAS 50 ng/g NR Chau et al.
vegetation water (sediment) (1980)
(total lead) 10 ng/g (fish NR
and vegetation)
Milk add 50 µl (C2H5)4NOH in ethanol to 25 µl GFAAS NR NR Michaelson &
milk; heat and dilute with water to 125 µl Sauerhoff (1974)
Evaporated dry ash sample; dissolve in HNO3 ASV 0.005 µg/g 99 Capar & Rigsby
milk (1989)
Agricultural dry ash sample with H2SO4 and HNO3; DPASV 0.4 ng/g 85-106 Satzger et al.
crops dilute with water (1982)
Grains, milk, bomb digest sample with acid; heat or GFAAS 20 µg/g (bomb) 85-107 Ellen & Van
mussels, fish digest with acid and dry ash; dissolve 5 µg/g (dry ash) 75-107 Loon (1990)
in acid; dilute with water DPASV NR 82-120
Table 2 (cont'd)
Sample type Preparation method Analytical method Sample detection Percentage Reference
limit recovery
Citrus leaves chop or pulverize sample; digest with hot ICP-AES 10-50 µg/litre 75-82 Que Hee &
and paint acid; dry; redissolve in acid (citrus Boyle (1988)
leaves)
89-96
(paint)
a AAS = atomic absorption; AES = atomic emissions spectroscopy; ASV = anode stripping voltametry; (C2H5)4NOH = tetraethylammonium
hydroxide; DPASV = differential pulse anodic stripping voltametry; EPA = US Environmental Protection Agency; GFAAS = graphite furnace
atomic absorption spectrometry; HCl = hydrochloric acid; HClO4 = perchloric acid; HNO3 = nitric acid; H2O2 = hydrogen peroxide;
H2SO4 = sulfuric acid; ICP-AES = inductively coupled plasma/atomic emission spectroscopy; IDMS = isotope dilution mass spectrometry;
NaOH = sodium hydroxide; NIOSH = National Institute for Occupational Safety and Health; NR = not reported; XRF = X-ray fluorescence
Table 3. Analytical methods for determining lead in biological materialsa
Sample Preparation method Analytical method Sample detection Percentage Reference
type limit recovery
Blood wet ash sample with acid mixtures; ASV with mercury-graphite 0.192 µmol/litre 95-105 NIOSH (1977c)
dissolve residue in dilute HClO4 electrode (NIOSH method (4 µg/dl)
P&CAM 195)
Blood wet ash sample with HNO3; dissolve GFAAS (NIOSH method 0.48 µmol/litre NR NIOSH (1977e)
residue in dilute HNO3 P&CAM 214) (10 µg/dl)
Blood dilute sample with Triton X-100(R); add GFAAS 0.011 µmol/litre 93-105 Aguilera de
nitric acid and diammonium phosphate (0.24 µg/dl) Benzo et al.
(1989)
Blood dilute sample with ammonia solution ICP-MS 0.072 µmol/litre 96-111 Delves &
containing Triton X-100(R); analyse (1.5 µg/dl) Campbell
(1988)
Blood dilute sample in 0.2% Triton X-100(R) GFAAS approx. 0.072 97-150 Que Hee et
and water; analyse µmol/litre (approx. al. (1985a)
1.5 µg/dl)
Blood and wet ash sample with HNO3, complex Spectrophotometry 0.144 µmol/litre 97 NIOSH (1977a)
urine with dephenylthiocarbazone and (NIOSH method (3.0 µg/dl) (blood);
extract with chloroform P&CAM 102) 0.0576 µmol/litre 97
(12 µg/litre) (urine)
Serum, filter sample if needed; dilute with ICP-AES 0.048-0.240 µmol/litre 85 (serum) Que Hee &
blood and acid or water (1.0-5.0 µg/dl) Boyle (1988)
urine
Urine wet ash sample with acid mixture and ASV with mercury-graphite 0.0192 µmol/litre 90-110 NIOSH (1977d)
dissolve in dilute HClO4 electrode (NIOSH method (4 µg/litre)
P&CAM 200)
Table 3 (cont'd)
Sample Preparation method Analytical method Sample detection Percentage Reference
type limit recovery
Liver, bomb digest sample with acid and heat, GFAAS 20 µg/g (bomb); 85-107 Ellen & Van
kidney, or digest with acid and dry ash; dissolve (bomb); Loon (1990)
muscle in acid; dilute with water 5 µg/g (dry ashing) 75-107 (dry
ashing)
Bone direct partially polarized photons at XRF 20 µg/g NR Christoffersson
second phalanx of left forefinger et al. (1986)
(non-invasive technique)
Bone direct partially polarized photons at XRF 20 µg/g NR Wielopolski
anteromedial skin surface of mid-tibia et al. (1986)
(non-invasive technique)
Teeth clean and section tooth; digest with ASV NR 83-114 Rabinowitz
HNO3; evaporate; redissolve in buffer et al. (1989)
solution
Teeth dry ash sample; crush; dry ash again; AAS NR 90-110 Steenhout &
dissolve in HNO3 Pourtois
(1981)
a AAS = atomic absorption spectrometry; ASV = anode stripping voltametry; GFAAS = graphite furnace atomic absorption spectrometry;
HClO4 = perchloric acid; HNO3 = nitric acid; ICP-AES = inductively coupled plasma-atomic emission spectroscopy;
ICP-MS = inductively coupled plasma-mass spectrometry; NIOSH = National Institute for Occupational Safety and Health;
NR = not reported; XRF = X-ray fluorescence
3. SOURCES OF HUMAN AND ENVIRONMENTAL EXPOSURE
3.1 Natural occurrence
Because lead is relatively abundant in the earth's crust it is
found naturally throughout the world. The major natural sources of
lead are volcanic emissions, geochemical weathering, and emissions
from sea spray. A small amount of radioisotopic lead (207Pb) is
derived from the decay of radon gas released from geological sources.
It has been estimated that the worldwide natural emission rates of
lead are of the order of 19 000 tonnes/year (Nriagu & Pacyna, 1988),
with volcanic sources accounting for 6400 tonnes/year (Nriagu, 1979).
Owing to centuries of human exploitation of lead resources, it is
difficult to determine the natural content of lead in most ecosystems.
Data on environmental levels, uses, and sources of lead have been
summarized in a recent review (OECD, 1993).
3.1.1 Rocks and soils
The average concentration of lead in the earth's crust is between
10 and 20 mg/kg (IPCS, 1989). The major geological sources of lead are
in igneous and metamorphic rocks.
The soil is the most important repository in terrestrial
ecosystems for contaminants of anthropogenic origin (Nriagu & Pacyna,
1988; Nriagu, 1989). The lead content of soils (which are for
discussion purposes distinguished here from surface dusts) is greatly
influenced by anthropogenic activities and by long- and short-range
airborne transport of lead from various sources. Both dry and wet
deposition are important routes of input.
Lead in soil may be relatively insoluble (as a sulfate, carbonate
or oxide), soluble, adsorbed onto clays, adsorbed and coprecipitated
with sesquioxides, adsorbed onto colloidal organic matter, or
complexed with organic moieties in soil (US EPA, 1986a; IPCS, 1989).
Soil pH, content of humic and fulvic acids, and amount of organic
matter influence the content and mobility of lead in soils. Since
acidic conditions favour the solubilization and leaching of lead from
the solid phase, acidic soils tend to have lower lead concentrations
when analysed as dry soil. Humic and fulvic acids can also mobilize
lead, and certain complex organic molecules can act as chelators of
lead (IPCS, 1989).
Background levels of lead in soil are in the range of 10-70 mg/kg
(GEMS, 1985). Similar results have been found in studies of mobile
source contamination near highways; soil taken at distances of
50-100 m from highways (outside the range of immediate impact from
traffic emissions) usually shows levels of lead below 40 mg/kg. In the
1985, GEMS survey of selected countries, lead concentrations in
topsoil from Malta were found to have a mean of 54 mg/kg in areas at
least 5 m from roadways; less than one metre from roadways the mean
concentration was 138 mg/kg. A 1977 report from Sweden found a mean of
16 mg/kg in non-contaminated areas (GEMS, 1985).
3.1.2 Sediments
Sediments from freshwater and marine environments have been
studied for lead content. This compartment provides a unique record of
the history of changes in global lead fluxes (Patterson, 1983). Levels
of lead in sediments dated before the onset of the industrial
revolution in Western Europe show very low levels, less than 10% of
current levels (Flegal et al., 1987). The average background level of
lead in marine sediments off southern California was reported by
Flegal et al. (1987) to be 1.3 mg/kg.
3.1.3 Water
Flegal et al. (1987) estimate that the natural concentration of
lead in surface water is about 0.02 µg/litre. In general, lead is not
found in ground or surface waters at concentrations above 10 µg/litre
(IPCS, 1989).
Data from oceans indicate very low levels of lead in sea-water
samples not affected directly by significant sources of lead. Water
samples taken from an area of the Pacific, where annual windborne-
input fluxes of lead are estimated to be 3 mg/cm2, have lead
concentrations of 3.5 ng/litre (0-100 m depth) and 0.9 ng/litre at
depths greater than 2500 m. In contrast, water samples taken from the
north Atlantic, where annual windborne-input fluxes of lead are
170 mg/cm2, contain 34 ng lead/litre at the surface and 5 ng/litre
in depths below 2500 m (Patterson, 1983). Settle & Patterson (1980)
have estimated that prehistoric oceans contained 0.5 ng/litre lead.
Flegal et al. (1987) have estimated that over 95% of the lead in
off-shore surface waters is the result of windborne inputs. However,
in coastal waters near Monterey (California, USA), higher
concentrations of lead were found in sea water, sediments and
organisms; these elevations were related to specific sources by
systematic isotope analyses (Flegal et al., 1987).
3.1.4 Air
Anthropogenic inputs of lead from a range of sources have
resulted in global dispersion of both inorganic and organic species of
lead into the air, of which 80-90% is derived from alkyllead fuel
additives (WHO, 1987). Nriagu & Pacyna (1988) estimated that a total
of 330 000 tonnes of lead is discharged directly into the atmosphere
each year. Estimations of pre-industrial levels of lead in air from
natural origins (volcanic emissions, crustal weathering, radon decay
and sea-spray releases) are in the range of 0.01-0.1 µg/m3 (US NRC,
1980). The lowest level reported since 1975 is 0.076 ng/m3 measured
at the South Pole (US EPA, 1986a).
3.1.5 Plants
Lead occurs naturally in plants and results from both deposition
and uptake. There is a positive linear relationship between lead
concentrations in plants and soil (Davies & Thornton, 1989). As with
other environmental compartments, measurement of "background" levels
of lead in plants is complicated by the general contamination of the
globe from centuries of lead use, which has included direct
application of lead-containing chemicals in agriculture (see below)
and contamination of fertilizers with lead. Lead has been measured in
superphosphate fertilizer at concentrations as high as 92 mg/kg (Lisk,
1972). Sewage sludge, used as a source of nutrients in agriculture,
may contain even higher levels of lead. The concentration of lead in
sewage sludge is typically < 1000 mg/kg. Levels as high as 26 g/kg
have been measured in the USA (Chaney et al., 1984). Soil receiving
heavy sludge applications over long periods of time (years) contained
425 mg lead/kg; the concentration in untreated soil was 47 mg/kg
(Beckett, 1979).
3.1.6 Environmental contamination from natural sources
The contribution of natural sources of lead to human exposure is
small. As a result of various breakdown processes, rocks yield lead
which is transferred to the biosphere and the atmosphere, and,
ultimately, back to the earth's crust in the form of sedimentary
rocks. Soluble lead has for thousands of years entered the oceans with
river discharges, and the rate has been estimated by Patterson (1965)
to be around 17 000 tonnes/year. Sources contributing to airborne lead
are silicate dusts, volcanic halogen aerosols, forest fires, sea salts
aerosols, meteoric and meteorite residues, and lead derived from the
decay of radon. While the lead content of most coals is relatively
low, coal fly ash is enriched in lead (Hutton et al., 1988) and is a
source of environmental contamination.
3.2 Anthropogenic sources
World lead consumption has steadily increased over the period
1965-1990 and was about 5.6 × 106 tonnes in 1990 (OECD, 1993).
Further review of the data summarized by OECD (1993) indicates a
change in consumption patterns worldwide. Although the consumption of
lead within the 24 countries of the OECD increased only slightly over
the decade from 1980 to 1990, consumption within less developed
economies (Africa and Asia) increased from 315 000 tonnes in 1970 to
844 000 tonnes in 1990.
3.2.1 Lead mining
Lead occurs in a variety of minerals, the most important of which
are galena (PbS), cerrusite (PbCO3) and anglesite (PbSO4). Galena
is by far the most important source of primary lead. It occurs mostly
in deposits associated with other minerals, particularly those
containing zinc. Mixed lead and zinc ores account for about 70% of
total primary lead supplies. Ores containing mainly lead account for
about 20% and the remaining 10% is obtained as a by-product from other
deposits, such as mixed copper-zinc deposits. The proportions of
various metals may differ in the ores of different countries. Silver
is the most important of the other metals frequently present in lead
deposits, but copper may also be present in concentrations high enough
to be commercially important. Other minor constituents of lead ores
are gold, bismuth, antimony, arsenic, cadmium, tin, gallium, thallium,
indium, germanium and tellurium.
The major countries producing lead from mining activity during
1987-1991 were the USA, Canada, Australia, Peru, the former USSR and
Mexico, as shown in Table 4. Other countries producing lead from lead
ores include China, the former Yugoslavia, Morocco, Spain, Sweden and
Tunisia. In general, the level of world production of lead from mining
activities has remained relatively constant at about 3.3 × 106
tonnes between 1988 and 1991 (ILZSG, 1992); this represents roughly
60% of the world demand for lead.
Table 4. Major countries producing lead from ore and ore
concentratesa
Country 1987 1988 1989 1990 1991
Canada 423 200 366 600 276 100 241 300 278 100
USA 318 300 395 700 419 300 495 200 483 300
Ex-USSR 510 000 520 000 500 000 490 000 --
Australia 489 200 462 000 495 000 570 000 579 000
Mexico 177 200 178 100 163 000 174 100 158 800
Peru 204 000 149 000 192 200 187 800 199 100
a From: World Bureau of Metal Statistics (1992)
3.2.2 Smelting and refining
Smelting and refining are classified as either primary or
secondary, the former producing refined lead products from ores or
concentrates (primary lead) and the latter producing lead by
recovering it from lead-bearing scrap and waste materials (secondary
lead). Secondary lead is derived from processing what is termed new
scrap arising during manufacturing processes and recycled old scrap
arising from waste materials containing lead. Most scrap is from old
sources, of which the most important are lead plates from batteries,
solder, common babbitt, soft lead, lead solders, cable coverings, type
metals, dross and other lead-containing products. There has been an
increasing contribution of secondary lead sources to the total
worldwide production of lead, as shown in Table 5 (World Bureau of
Metal Statistics, 1992). Secondary sources of lead supplied between 35
and 40% of world production during the period from 1970 to 1990.
3.2.3 Environmental pollution from production of lead
Mining operations and the smelting and refining of both primary
and secondary lead are known to cause contamination of the nearby
environment. The nature and extent of contamination depends on many
factors, including the level of production, the effectiveness of
emission controls, climate, topography and other local factors.
Concentrations are usually highest within 3 km of the point source (US
EPA, 1989). A report from China found that lead levels in ambient air,
plants and soil increased proportionally with proximity to a large
primary smelter; at 50 m from the source, the air lead level was
60 µg/m3, the lead level in plants was 29.1 mg/kg, and soil lead
level was 170 mg/kg (Wang, 1984). However, some earlier studies have
shown air pollution and soil contamination as far as 10 km from
smelters (Djuric et al., 1971; Kerin, 1973; Landrigan et al., 1975).
Table 5. Relative contribution of primary and secondary sources
relative to world lead production (1987-1990)a
1987 1988 1989 1990
Primary 422 100 3 414 200 3 286 500 3 324 500
Secondary 2 045 600 2 103 900 2 272 900 2 254 800
a From: World Bureau of Metal Statistics (1992)
The impacts of lead mining and smelting can persist for long
periods of time. A study conducted in Wales, United Kingdom, in an
area where lead mining began 2000 years ago and ended in the middle of
the 20th century, found high concentrations of lead in soils (Davies
et al., 1985). In Port Pirie, Australia, a community with one of the
world's largest and oldest primary lead smelters, lead levels in soils
were found to be grossly elevated, and the incidence of elevated blood
lead levels in pregnant women and young children was also increased
above that found in other communities in Australia (Wilson et al.,
1986).
3.3 Consumption and uses of lead and its compounds
Lead has a combination of physical and chemical properties that
have made it extremely useful industrially, i.e. high density, high
opacity to gamma and X-ray energies, low sound conductance, a low
melting point, exceptional malleability, high corrosion resistance,
and stability. In 1990, 5.627 × 106 tonnes of lead were consumed
worldwide (ILZSG, 1992). The twenty-four industrialized countries of
the OECD consumed approximately 65% of this amount, with eastern
Europe and the former USSR using 21%. Asia now utilizes about 9% of
the world's lead production.
The use patterns of refined lead vary from country to country.
The situation in 1990 in Mexico is shown in Table 6, while end-use
categories within OECD countries are summarized in Fig. 1, which
indicates the changes between 1970 and 1990 (OECD, 1993).
Table 6. Principal uses of refined lead in Mexicoa
Type of product 1988 1990
(%) (%)
Oxides 69.7 56.7
Batteries 9.2 17.9
Tetraethyllead 7.9 11.9b
Cables 4.0 1.5
Others 9.2 11.9
a ILZSG (1992)
b This does not reflect the introduction of lead-free petrol
in 1990.
From Fig. 1, it is evident that the largest use of lead within
OECD countries is for battery production, whereas there has been a
large drop in the demand for lead-containing gasoline additives.
However, this pattern is not valid worldwide, e.g., concentrations in
petrol range from zero in such countries as Japan and Thailand to
1.12 g/litre in the Virgin Islands (Octel, 1991).
In the past the use of lead in the chemical industry for the
preparation of paints, pigments and coloured inks was widespread. Many
countries have now restricted this use, and concentrations of lead
greater than 0.06% (USA) and 0.5% (New Zealand) are not permitted in
indoor paints (Albert & Badillo, 1991; OECD, 1993). In 1982, data from
the United Kingdom (UK, 1982) indicated levels of lead between 2500
and 3000 mg/kg in decorative glass paints and up to 448 g/kg in
white-lead primer. Red-leadcontaining paints, still used widely to
paint structural steel works, can contain up to 661 g lead/kg.
It is accepted that the following criteria should initiate the
updating of an EHC monograph: new data are available that would
substantially change the evaluation; there is public concern for
health or environmental effects of the agent because of greater
exposure; an appreciable time period has elapsed since the last
evaluation.
All Participating Institutions are informed, through the EHC
progress report, of the authors and institutions proposed for the
drafting of the documents. A comprehensive file of all comments
received on drafts of each EHC monograph is maintained and is
available on request. The Chairpersons of Task Groups are briefed
before each meeting on their role and responsibility in ensuring that
these rules are followed.
WHO TASK GROUP ON ENVIRONMENTAL HEALTH CRITERIA FOR INORGANIC LEAD
Members
Professor S. Araki, Department of Public Health, Faculty of Medicine,
University of Tokyo, Japan
Dr P. Baghurst, Division of Human Nutrition, Commonwealth Scientific
Industrial Research Organization, Adelaide, Australia
Dr D. Bellinger, Neuroepidemiology Unit, Gardner House, Children's
Hospital, Boston, Massachusetts, USA
Dr I. Calder, Occupational and Environmental Health, South Australian
Health Commission, Adelaide, South Australia, Australia
Dr D.A. Cory-Slechta, Department of Environmental Medicine,
University of Rochester School of Medicine and Dentistry,
Rochester, New York, USA
Dr K. Dietrich, Department of Environmental Health, Division of
Biostatistics and Epidemiology, University of Cincinnati College
of Medicine, Cincinnati, Ohio, USA
Dr R.A. Goyer, Chapel Hill, North Carolina, USA (Chairman)
Dr M.R. Moore, University of Glasgow, Department of Medicine and
Therapeutics, Western Infirmary, Glasgow, Scotland
Dr C. Nam Ong, Department of Community, Occupational and Family
Medicine, National University of Singapore, National University
Hospital, Singapore
Dr S.J. Pocock, Department of Epidemiology and Population Sciences,
Medical Statistics Unit, University of London, London, England
Dr M.B. Rabinowitz, Marine Biological Laboratory, Woods Hole,
Massachusetts, USA
Dr M. Smith, Thomas Coram Research Unit, London, England
Dr G. Winneke, Medical Institute for Environmental Health,
Heinrich-Heine University, Düsseldorf, Germany (Vice-Chairman)
Observers
Dr C. Boreiko, Environmental Health, International Lead Zinc Research
Organization (ILZRO) Inc., Research Triangle Park, North
Carolina, USA
Dr N.H. Clark, Lead Industry Environment and Health Forum, Melbourne,
Victoria, Australia
Dr J.M. Davis, Environmental Criteria and Assessment Office, US
Environmental Protection Agency, Research Triangle Park, North
Carolina, USA
Professor G. Duggin, Toxicology Unit, Royal Prince Alfred Hospital,
Camperdown, Australia
Dr G.R. Neville, Queensland Health Department, Brisbane, Australia
Secretariat
Dr G.C. Becking, International Programme on Chemical Safety,
Interregional Research Unit, World Health Organization, Research
Triangle Park, North Carolina, USA (Secretary)
Dr K.R. Mahaffey, National Institute of Environmental Health
Sciences, Research Triangle Park, North Carolina, USAa
Dr A.E. Robinson, Toronto, Ontario, Canada (Rapporteur)
a Present address: US Environmental Protection Agency, Environmental
Criteria and Assessment Office, Cincinnati, Ohio, USA
ENVIRONMENTAL HEALTH CRITERIA FOR INORGANIC LEAD
A WHO Task Group on Environmental Health Criteria for Inorganic
Lead met in Brisbane, Australia, from 1 to 6 February 1993. The
meeting was sponsored by a consortium of Australian Commonwealth and
State Governments through a national Steering Committee chaired by
Dr Keith Bentley, Director, Health and Environmental Policy,
Department of Human Services and Health, Canberra. The meeting was
hosted and organized by the Queensland Department of Health,
Dr G.R. Neville being responsible for the arrangements. Dr G. Murphy,
Director of Public Health, Queensland, welcomed the participants on
behalf of the Organizers, and Dr T. Adams, Chief Commonwealth Medical
Advisor and Dr G. Johns, Parliamentary Secretary to Federal Minister
for Health, Housing and Community Services, welcomed the participants
on behalf of the Commonwealth Government. Dr Johns stressed the
importance attached to this IPCS meeting by the Commonwealth and State
Governments of Australia. Dr G.C. Becking, IPCS, welcomed the
participants on behalf of Dr M. Mercier, Director of the IPCS and the
three cooperating organizations (UNEP/ILO/WHO). The Task Group
reviewed and revised the draft criteria monograph, and made an
evaluation of the risks to human health from exposure to inorganic
lead.
The Task Group draft was prepared by Dr A.E. Robinson, Toronto,
Canada, using texts made available by Dr K.R. Mahaffeya (National
Institute of Environmental Health Sciences, Research Triangle Park,
North Carolina, USA) and Dr E. Silbergeld (University of Maryland
School of Medicine, Baltimore, Maryland, USA), and the comments
received from the IPCS contact points for environmental health
criteria monographs. The draft was revised extensively by the Task
Group taking into account the comments from the IPCS contact points.
Dr G.C. Becking (IPCS Central Unit, Interregional Research Unit)
and Dr P.G. Jenkins (IPCS Central Unit, Geneva) were responsible for
the overall scientific content and technical editing, respectively, of
this monograph.
The efforts of all who helped in the preparation and finalization
of this publication are gratefully acknowledged.
a Present address: US Environmental Protection Agency, Environmental
Criteria and Assessment Office, Cincinnati, Ohio, USA
ABBREVIATIONS
AAS atomic absorption spectrometry
AES atomic emission spectroscopy
ALA delta-aminolaevulinic acid
ALAD delta-aminolaevulinic acid dehydratase
ASV anodic stripping voltametry
EDTA ethylenediaminetetraacetic acid
FEP free erythrocyte porphyrin
GFAAS graphite furnace atomic absorption spectrometry
ICP inductively coupled plasma
IDMS isotope dilution mass spectrometry
MPb mobilization yield of lead
MSW municipal solid waste
PbB blood lead
PbT tooth lead
TML tetramethyllead
XRFS X-ray fluorescence spectroscopy
ZPP zinc protoporphyrin
PREFACE
Although many countries have initiated programmes to lower the
level of lead in the environment, human exposure to lead remains of
concern to public health officials worldwide. For over 20 years the
World Health Organization (WHO) and the International Programme on
Chemical Safety (IPCS) have been concerned about the health and
environmental effects of the levels of inorganic lead in the
environment. The evaluation of human health risks arising from
food-borne lead has been carried out by WHO on four occasions since
1972. In addition, health-based guidance values for lead in water, air
and the workplace have been developed by various Task Groups convened
by WHO. Environmental Health Criteria 3: Lead, published in 1977,
examined the effects of lead on human health and Environmental Health
Criteria 85: Lead - Environmental Aspects was published in 1989.
Since the publication of Environmental Health Criteria 3: Lead, a
large body of knowledge has accumulated concerning the effects of lead
on humans at low levels of exposure. Studies have emphasized the
effects of inorganic lead on infants and children, a high-risk
population. This monograph on inorganic lead reflects this research
emphasis; a major part of the monograph deals with the neurotoxic
effects of lead with emphasis on neurobehavioural development in
children. Less detail is presented on the health effects of the higher
levels of inorganic lead found in some workplaces, although such
exposures are still considered to pose a risk to humans in many
regions of the world.
This monograph deals only with the human health effects of
inorganic lead. No attempt has been made to evaluate the human health
effects of organo-lead compounds, although it was recognized that such
compounds when added to petrol (gasoline) are a major source of
inorganic lead in the environment. In view of the toxicity of many
organo-lead derivatives and the possible methylation of inorganic lead
in the environment, the IPCS plans to evaluate the risk to humans from
exposure to organo-lead compounds in a separate monograph.
As with all IPCS criteria monographs, no attempt has been made to
prepare an exhaustive bibliography of the extremely large amount of
lead-related literature published since 1977. Rather, an effort has
been made to review critically the studies on humans and experimental
animals that are essential for the evaluation of risks to human health
from exposure to all sources of inorganic lead.
1. SUMMARY
This monograph focuses on the risks to human health associated
with exposure to lead and inorganic lead compounds. Emphasis has been
given to data which have become available since the publication of
Environmental Health Criteria 3: Lead (IPCS, 1977). The environmental
effects of lead are discussed in Environmental Health Criteria 85:
Lead - Environmental Aspects (IPCS, 1989).
1.1 Identity, physical and chemical properties, and analytical
methods
Lead is a soft, silvery grey metal, melting at 327.5°C. It is
highly resistant to corrosion, but is soluble in nitric and hot
sulfuric acids. The usual valence state in inorganic lead compounds is
+2. Solubilities in water vary, lead sulfide and lead oxides being
poorly soluble and the nitrate, chlorate and chloride salts are
reasonably soluble in cold water. Lead also forms salts with such
organic acids as lactic and acetic acids, and stable organic compounds
such as tetraethyllead and tetramethyllead.
The most commonly used methods for the analysis of low
concentrations of lead in biological and environmental materials are
flame, graphite furnace and inductively coupled plasma atomic
absorption spectroscopy and anode stripping voltametry. Depending on
sample pretreatment, extraction techniques and analytical
instrumentation, detection limits of 0.12 µmoles lead/litre blood
(2.49 µg/dl) can be achieved. However, reliable results are obtained
only when specific procedures are followed to minimize the risk of
contamination during sample collection, storage, processing and
analysis.
1.2 Sources of human exposure
The level of lead in the earth's crust is about 20 mg/kg. Lead in
the environment may derive from either natural or anthropogenic
sources. Natural sources of atmospheric lead include geological
weathering and volcanic emissions and have been estimated at
19 000 tonnes/year, compared to an estimate of 126 000 tonnes/year
emitted to the air from the mining, smelting and consumption of over 3
million tonnes of lead per year.
Atmospheric lead concentrations of 50 pg/m3 have been found in
remote areas. Background levels of lead in soil range between 10 and
70 mg/kg and a mean level near roadways of 138 mg/kg has been
reported. Present levels of lead in water rarely exceed a few
micrograms/litre; the natural concentration of lead in surface water
has been estimated to be 0.02 µg/litre.
Lead and its compounds may enter the environment at any point
during mining, smelting, processing, use, recycling or disposal. Major
uses are in batteries, cables, pigments, petrol (gasoline) additives,
solder and steel products. Lead and lead compounds are also used in
solder applied to water distribution pipes and to seams of cans used
to store foods, in some traditional remedies, in bottle closures for
alcoholic beverages and in ceramic glazes and crystal tableware. In
countries where leaded petrol is still used, the major air emission is
from mobile and stationary sources of petrol combustion (urban
centres). Areas in the vicinity of lead mines and smelters are subject
to high levels of air emissions.
Airborne lead can be deposited on soil and water, thus reaching
humans through the food chain and in drinking-water. Atmospheric lead
is also a major source of lead in household dust.
1.3 Environmental transport, distribution and transformation
The transport and distribution of lead from fixed, mobile and
natural sources are primarily via air. Most lead emissions are
deposited near the source, although some particulate matter (< 2 µm
in diameter) is transported over long distances and results in the
contamination of remote sites such as arctic glaciers. Airborne lead
can contribute to human exposures by the contamination of food, water
and dust, as well as through direct inhalation. The removal of
airborne lead is influenced by atmospheric conditions and particulate
size. Large amounts of lead may be discharged to soil and water.
However, such material tends to remain localized because of the poor
solubility of lead compounds in water.
Lead deposited in water, whether from air or through run-off from
soils, partitions rapidly between sediment and aqueous phase,
depending upon pH, salt content, and the presence of organic chelating
agents. Above pH 5.4, hard water may contain about 30 µg lead/litre
and soft water about 500 µg lead/litre. Very little lead deposited on
soil is transported to surface or ground water except through erosion
or geochemical weathering; it is normally quite tightly bound
(chelated) to organic matter.
Airborne lead can be transferred to biota directly or through
uptake from soil. Animals can be exposed to lead directly through
grazing and soil ingestion or by inhalation. There is little
biomagnification of inorganic lead through the food chain.
1.4 Environmental levels and human exposure
In the general non-smoking adult population, the major exposure
pathway is from food and water. Airborne lead may contribute
significantly to exposure, depending upon such factors as use of
tobacco, occupation, proximity to motorways, lead smelters, etc., and
leisure activities (e.g., arts and crafts, firearm target practice).
Food, air, water and dust/soil are the major potential exposure
pathways for infants and young children. For infants up to 4 or 5
months of age, air, milk, formulae and water are the significant
sources of lead exposure.
Levels of lead found in air, food, water and soil/dust vary
widely throughout the world and depend upon the degree of industrial
development, urbanization and lifestyle factors. Ambient air levels
over 10 µg/m3 have been reported in urban areas near a smelter,
whereas lead levels below 0.2 µg/m3 have been found in cities where
leaded petrol is no longer used. Lead intake from air can, therefore,
vary from less than 4 µg/day to more than 200 µg/day.
Levels of lead in drinking-water sampled at the source are
usually below 5 µg/litre. However, water taken from taps (faucets) in
homes where lead is present in the plumbing can contain levels in
excess of 100 µg/litre, particularly after the water has been standing
in the pipes for some hours.
The level of dietary exposure to lead depends upon many lifestyle
factors, including foodstuffs consumed, processing technology, use of
lead solder, lead levels in water, and use of lead-glazed ceramics.
For infants and children, lead in dust and soil often constitutes
a major exposure pathway. Lead levels in dust depend upon such factors
as the age and condition of housing, the use of lead-based paints,
lead in petrol and urban density. The intake of lead will be
influenced by the age and behavioural characteristics of the child and
bioavailability of lead in the source material.
Inhalation is the dominant pathway for lead exposure of workers
in industries producing, refining, using or disposing of lead and lead
compounds. During an 8-h shift, workers can absorb as much as 400 µg
lead, in addition to the 20-30 µg/day absorbed from food, water and
ambient air; significant intake may occur from ingestion of large
inhaled particulate material.
1.5 Kinetics and metabolism in laboratory animals and humans
Lead is absorbed in humans and animals following inhalation or
ingestion; percutaneous absorption is minimal in humans. Depending
upon chemical speciation, particle size, and solubility in body
fluids, up to 50% of the inhaled lead compound may be absorbed. Some
inhaled particulate matter (larger than 7 µm) is swallowed following
mucociliary clearance from the respiratory tract. In experimental
animals and humans, absorption of lead from the gastrointestinal tract
is influenced by the physico-chemical nature of the ingested material,
nutritional status, and type of diet consumed. In adult humans
approximately 10% of the dietary lead is absorbed; the proportion is
higher under fasting conditions. However, in infants and young
children as much as 50% of dietary lead is absorbed, although
absorption rates for lead from dusts/soils and paint chips can be
lower depending upon the bioavailability. Diets that are deficient in
calcium, phosphate, selenium or zinc may result in increased lead
absorption. Iron and vitamin D also affect absorption of lead.
Blood lead (PbB) levels are used as a measure of body burden and
absorbed (internal) doses of lead. The relationship between blood lead
and the concentration of lead in exposure sources is curvilinear.
Once it has been absorbed, lead is not distributed homogeneously
throughout the body. There is rapid uptake into blood and soft tissue,
followed by a slower redistribution to bone. Bone accumulates lead
over much of the human life span and may serve as an endogenous source
of lead. The half-life for lead in blood and other soft tissues is
about 28-36 days, but it is much longer in the various bone
compartments. The percentage retention of lead in body stores is
higher in children than adults. Transfer of lead to the human fetus
occurs readily throughout gestation.
Blood lead is the most commonly used measure of lead exposure.
However, techniques are now available for measuring lead in teeth and
bone, although the kinetics are not fully understood.
1.6 Effects on laboratory animals and in vitro systems
In all species of experimental animals studied, including
non-human primates, lead has been shown to cause adverse effects in
several organs and organ systems, including the haematopoietic,
nervous, renal, cardiovascular, reproductive and immune systems. Lead
also affects bone and has been shown to be carcinogenic in rats and
mice.
Despite kinetic differences between experimental animal species
and humans, these studies provide strong biological support and
plausibility for the findings in humans. Impaired learning/memory
abilities have been reported in rats with PbB levels of
0.72-0.96 µmoles/litre (15-20 µg/dl) and in non-human primates at PbB
levels not exceeding 0.72 µmoles/litre (15 µg/dl). In addition, visual
and auditory impairments have been reported in experimental animal
studies.
Renal toxicity in rats appears to occur at a PbB level in excess
of 2.88 µmol/litre (60 µg/dl), a value similar to that reported to
initiate renal effects in humans. Cardiovascular effects have been
seen in rats after chronic low-level exposures resulting in PbB levels
of 0.24-1.92 µmol/litre (5-40 µg/dl). Tumours have been shown to occur
at dose levels below the maximum tolerated dose of 200 mg lead (as
lead acetate) per litre of drinking-water. This is the maximum dose
level not associated with other morphological or functional changes.
1.7 Effects on humans
In humans, lead can result in a wide range of biological effects
depending upon the level and duration of exposure. Effects at the
subcellular level, as well as effects on the overall functioning of
the body, have been noted and range from inhibition of enzymes to the
production of marked morphological changes and death. Such changes
occur over a broad range of doses, the developing human generally
being more sensitive than the adult.
Lead has been shown to have effects on many biochemical
processes; in particular, effects on haem synthesis have been studied
extensively in both adults and children. Increased levels of serum
erythrocyte protoporphyrin and increased urinary excretion of
coproporphyrin and delta-aminolaevulinic acid are observed when PbB
concentrations are elevated. Inhibition of the enzymes
delta-aminolaevulinic acid dehydratase and dihydrobiopterin reductase
are observed at lower levels.
The effects of lead on the haemopoietic system result in
decreased haemoglobin synthesis, and anaemia has been observed in
children at PbB concentrations above 1.92 µmol/litre (40 µg/dl).
For neurological, metabolic and behavioural reasons, children are
more vulnerable to the effects of lead than adults. Both prospective
and cross-sectional epidemiological studies have been conducted to
assess the extent to which environmental lead exposure affects
CNS-based psychological functions. Lead has been shown to be
associated with impaired neurobehavioural functioning in children.
Impairment of psychological and neurobehavioural functions has
been found after long-term lead exposure of workers.
Electrophysiological parameters have been shown to be useful
indicators of subclinical lead effects in the CNS.
Peripheral neuropathy has long been known to be caused by
long-term high-level lead exposure at the workplace. Slowing of nerve
conduction velocity has been found at lower levels. These effects have
often been found to be reversible after cessation of exposure,
depending on the age and duration of exposure.
The effect of lead on the heart is indirect and occurs via the
autonomic nervous system; it has no direct effect on the myocardium.
The collective evidence from population studies in adults indicates
very weak associations between PbB concentration and systolic or
diastolic blood pressure. Given the difficulties of allowing for
relevant confounding factors, a causal relationship cannot be
established from these studies. There is no evidence to suggest that
any association of PbB concentration with blood pressure is of major
health importance.
Lead is known to cause proximal renal tubular damage,
characterized by generalized aminoaciduria, hypophosphataemia with
relative hyperphosphaturia and glycosuria accompanied by nuclear
inclusion bodies, mitochondrial changes and cytomegaly of the proximal
tubular epithelial cells. Tubular effects are noted after relatively
short-term exposures and are generally reversible, whereas sclerotic
changes and interstitial fibrosis, resulting in decreased kidney
function and possible renal failure, require chronic exposure to high
lead levels. Increased risk from nephropathy was noted in workers with
a PbB level of over 3.0 µmol/litre (about 60 µg/dl). Renal effects
have recently been seen among the general population when more
sensitive indicators of function were measured.
The reproductive effects of lead in the male are limited to sperm
morphology and count. In the female, some adverse pregnancy outcomes
have been attributed to lead.
Lead does not appear to have deleterious effects on skin, muscle
or the immune system. Except in the case of the rat, lead does not
appear to be related to the development of tumours.
1.8 Evaluation of human health risks
Lead adversely affects several organs and organ systems, with
subcellular changes and neurodevelopmental effects appearing to be the
most sensitive. An association between PbB level and hypertension
(blood pressure) has been reported. Lead produces a cascade of effects
on the haem body pool and affects haem synthesis. However, some of
these effects are not considered adverse. Calcium homoeostasis is
affected, thus interfering with other cellular processes.
a) The most substantial evidence from cross-sectional and
prospective studies of populations with PbB levels generally
below 1.2 µmol/litre (25 µg/dl) relates to decrements in
intelligence quotient (IQ). It is important to note that such
observational studies cannot provide definitive evidence of a
causal relationship with lead exposure. However, the size of the
apparent IQ effect, as assessed at 4 years and above, is a
deficit between 0 and 5 points (on a scale with a standard
deviation of 15) for each 0.48 µmol/litre (10 µg/dl) increment in
PbB level, with a likely apparent effect size of between 1 and 3
points. At PbB levels above 1.2 µmol/litre (25 µg/dl), the
relationship between PbB and IQ may differ. Estimates of effect
size are group averages and only apply to the individual child in
a probabilistic manner.
Existing epidemiological studies do not provide definitive
evidence of a threshold. Below the PbB range of 0.48-0.72 µmol/
litre (10-15 µg/dl), the effects of confounding variables and
limits in the precision in analytical and psychometric
measurements increase the uncertainty attached to any estimate
of effect. However, there is some evidence of an association
below this range.
b) Animal studies provide support for a causal relationship between
lead and nervous system effects, reporting deficits in cognitive
functions at PbB levels as low as 0.53-0.72 µmol/litre
(11-15 µg/dl) which can persist well beyond the termination of
lead exposure.
c) Reduction in human peripheral nerve conduction velocity may occur
with PbB levels as low as 1.44 µmol/litre (30 µg/dl). In
addition, sensory motor function may be impaired with PbB levels
as low as about 1.92 µmol/litre (40 µg/dl), and autonomic nervous
system function (electrocardiographic R-R interval variability)
may be affected at an average PbB level of approximately
1.68 µmol/litre (35 µg/dl). The risk of lead nephropathy is
increased in workers with PbB levels above 2.88 µmol/litre
(60 µg/dl). However, recent studies using more sensitive
indicators of renal function suggest renal effects at lower
levels of lead exposure.
d) Lead exposure is associated with a small increase in blood
pressure. The likely order of magnitude is that for any two-fold
increase in PbB level (e.g., from 0.8 to 1.6 µmol/litre, i.e.
16.6 to 33.3 µg/dl), there is a mean 1 mmHg increase in systolic
blood pressure. The association with diastolic pressure is of a
similar but smaller magnitude. However, there is doubt regarding
whether these statistical associations are really due to an
effect of lead exposure or are an artifact due to confounding
factors.
e) Some but not all epidemiological studies show a dose dependent
association of pre-term delivery and some indices of fetal growth
and maturation at PbB levels of 0.72 µmol/litre (15 µg/dl) or
more.
f) The evidence for carcinogenicity of lead and several inorganic
lead compounds in humans is inadequate.
g) Effects of lead on a number of enzyme systems and biochemical
parameters have been demonstrated. The PbB levels, above which
effects are demonstrable with current techniques for the
parameters that may have clinical significance, are all greater
than 0.96 µmol/litre (20 µg/dl). Some effects on enzymes are
demonstrable at lower PbB levels, but the clinical significance
is uncertain.
2. IDENTITY, PHYSICAL AND CHEMICAL PROPERTIES, AND ANALYTICAL METHODS
2.1 Physical and chemical properties of lead and its compounds
Lead (atomic number, 82; relative atomic mass, 207.19; specific
gravity, 11.34) is a bluish or silvery grey soft metal. The melting
point is 327.5°C and the boiling point at atmospheric pressure 1740°C.
It has four naturally occurring isotopes (208, 206, 207, and 204 in
order of abundance), but the isotopic ratios for various mineral
sources may differ. This property has been exploited in
non-radioactive-tracer environmental and metabolic studies. The
physical and chemical properties of elemental lead and some lead
compounds are summarized in Table 1.
Although lead has four electrons in its valence shell, only two
ionize readily. The usual oxidation state of lead in inorganic
compounds is therefore +2 rather than +4. The inorganic salts of lead,
such as lead sulfide and the oxides of lead, are generally poorly
soluble in water. However, the nitrate, chlorate and, to a much lesser
degree, the chloride are water soluble. Some of the salts formed with
organic acids, e.g., lead oxalate, are also insoluble, but the acetate
is relatively soluble, as shown in Table 1.
Under appropriate conditions of synthesis, stable compounds are
formed in which lead is directly bound to a carbon atom. Industrially
synthesized lead-carbon compounds include tetraethyllead and
tetramethyllead, which are of importance as fuel additives and, hence,
are sources of environmental lead.
2.2 Analytical procedures
In recent years substantial advances have been made in developing
methods for the quantification of metals at low concentrations. In
order to provide improved quality assurance of such measurements,
various reference materials in different matrices have been produced
(Muramatsu & Parr, 1985). To ensure adequate quality control, the
analyst should choose a reference material that matches as closely as
possible the experimental samples to be analysed. Choices are based
upon matrix type and concentration of the element of interest. A
summary of data on 60 biological and 40 environmental (non-biological)
reference materials has been compiled by Muramatsu & Parr (1985).
With the increased interest in measuring lead in the low µg/kg
and µg/m3 range in both environmental and biological samples, there
is need for particular attention to analytical sensitivity and
reliability. As lower concentrations are measured, problems of
laboratory contamination become more significant and quality control
and quality assurance programmes are important. Because of these
concerns, all analytical results for lead should report the laboratory
Table 1. Physical and chemical data on lead and selected lead compoundsa
Name Synonym Relative atomic/ Melting point Boiling point Solubility in cold Soluble in
and formula molecular mass (°C) (°C) water (g/litre)
Lead Pb 207.19 327.502 1740 insoluble HNO3; hot concentrated
H2SO4; hot water;
glycerine; alcohol (slightly)
Lead salts
acetate Pb(C2H3O2)2 325.28 280 - 443
carbonate cerrusite PbCO3 267.20 315 (decomposes) 0.0011 acid; alkali; decomposes in
hot water
chlorate Pb(ClO3)2 374.09 230 (decomposes) very soluble alcohol
chloride cotunite PbCl2 278.10 501 950 919 NH4 salts; slightly in dilute
HCl and in NH3; hot water
(33.4 g/litre)
nitrate Pb(NO3)2 331.20 470 (decomposes) 376.5 alcohol; alkali, NH3; hot
water (1270 g/litre)
orthophosphate Pb3(PO4)2 811.51 1014 0.00014 alkali; HNO3
oxalate PbC2O4 295.21 300 (decomposes) 0.0016 HNO3
dioxide plattnerite PbO2 239.19 290 (decomposes) insoluble dilute HCl; acetic acid
(slightly)
monoxide litharge PbO 223.19 888 0.017 dilute HNO3; acetic acid
Table 1 (cont'd)
Name Synonym Relative atomic/ Melting point Boiling point Solubility in cold Soluble in
and formula molecular mass (°C) (°C) water (g/litre)
sulfate anglesite PbSO4 303.25 1170 0.0425 NH4 salts; concentrated
H2SO4 (slightly)
sulfide galena PbS 239.25 1114 0.00086 acid
a Data from Weast (1985)
performance for reference standards and for parallel blank
measurements of sample contamination for the entire analytical
process. Without these, the validity of the data should be questioned.
2.2.1 Sampling procedures
Particular attention should be paid to the cleanliness of
equipment and glassware and the purity of the chemicals to prevent
secondary contamination by lead.
For the collection of samples, standard trace element methods are
generally required (Behne, 1980) with adequate quality control
procedures (Friberg, 1988; Jorhem & Slorach, 1988, Vahter & Friberg,
1988). Quality control samples for blood, faeces, air filters and dust
have been described (Lind et al., 1988).
2.2.1.1 Sampling of environmental media
In air sampling, both high-volume samplers and low-volume
techniques have been used. It should be noted that the collection
characteristics of high-volume samplers are strongly affected by
particle size and the orientation of the sampler. For particles larger
than 5 µm in diameter the high-volume sampler system is unlikely to
collect representative samples (US EPA, 1986a). As in all sampling for
suspended particulate matter, the accuracy of volume meters should be
checked periodically. The size of the pores in filters for collecting
lead-containing particles should be small, possibly less than 0.2 µm
for glass-fibre filters (Lee & Goranson, 1972).
Depending on the purpose of sampling, care should be taken to
select the appropriate site for sampling devices and to achieve the
best possible sampling conditions by:
* estimating the amount of particulate required for analysis before
deciding on the sample volume and the sampling procedure;
* placing the sampling devices in the appropriate position (e.g.,
in the breathing zone, level with inlet tubes of house
ventilators, at window level in the case of a traffic-laden town
street, at a reasonable distance from the highway in uninhabited
zones, etc);
* taking the samples at appropriate rates and volumes (e.g., daily
breathing volumes, daily ventilating capacities of installations)
and for a sufficient time to make possible the estimation of the
average concentration (e.g., during a work-shift, or a 24-h or
longer period for general population exposure);
* taking into account the use of areas under study (cattle grazing,
recreational zones, children's playgrounds, etc).
In addition, whenever possible a procedure should be used that
makes it possible to evaluate particle-size distribution and the
physicochemical properties of the lead compounds involved, including
the shape of the particles and the state of their aggregation.
Lead may be found in water bound to particulate matter as soluble
complexes or soluble compounds. Techniques for sampling water must
take this into account. It is necessary to sample water without
fractionation (filtration) when total lead levels are required.
Because of the potential for metals from low ionic strength waters to
be adsorbed onto the surfaces of some containers, samples should be
acidified (US EPA, 1986a). Selection, cleaning, and conditioning of
storage and sample containers deserve special attention (Moody, 1982).
The preparation of soil and dust samples for lead analyses
usually involves drying (at 100°C), homogenization by grinding, and
sieving (Thornton & Webb 1975; Bolter et al., 1975). Brown & Black
(1983) have discussed the issues related to quality assurance and
quality control in the collection and analysis of soil samples. Most
reports of lead in soil provide the total elemental abundance either
by acid extraction or X-ray fluorescence. However, the leachable or
bioavailable fraction is of special interest.
For the study of the dietary intake of lead from food, two
general methods have been utilized. The advantages and disadvantages
of the "duplicate portions" technique and the equivalent composite
technique ("market basket") have been reviewed by Pekkarinen (1970).
Although the duplicate portions (duplicate diets) technique can define
variability in consumption, it is expensive, and the sampling and
analytical procedures involved are complicated and limit the number of
individuals included in any study. With the equivalent composite
technique, the economy and ease of collection must be considered in
the light of the variability of results obtained due to uncertainties
in knowledge of actual preparation techniques, including possible lead
levels in water used for processing in individual homes.
The quantity of lead likely to be leached from ceramic surfaces
by different foods and beverages may be assessed using dilute acetic
acid solutions (1 to 4%) at temperatures in the range 20 to 100°C for
times ranging from 30 min to more than 24 h (Laurs, 1976; Merwin,
1976).
Colorimetric methods are suitable for screening inorganic
materials such as pottery or paint for lead. Positive reactions
require confirmation by established quantitative methods. Spot tests
using dithizone, rhodizonate and iodide (Feigl et al., 1972) are
available.
2.2.1.2 Sampling of biological materials
The main problem in the sampling of body fluids and tissues for
lead analysis is potential secondary contamination with lead. The low
general population blood lead (PbB) levels in many regions of the
world are complicating screening efforts, requiring levels of
analytical precision and sensitivity that can be achieved only through
intensive QA/QC programmes. Issues related to such sampling have been
examined in detail by US EPA (1986a).
Special precautions are needed to ensure that all venous
blood-collecting and blood-storage materials are as free from lead as
possible (IPCS, 1977). All glass equipment involved in blood
collection and storage should be made of lead-free silicate glass,
rinsed first in mineral acid, then with copious amounts of
glass-distilled or deionized water. Polypropylene syringes have been
recommended (NAS-NRC, l972). Needles should be of stainless steel with
polypropylene hubs. Blood is often drawn directly from the needle into
vacuum tubes. It is wise to confirm periodically the absence of
significant amounts of lead in the anticoagulant used in the blood
container as well as monitoring the contamination level (blank) for
the entire analytical process.
New analytical techniques make it possible to determine lead
concentrations in microlitre quantities of blood. The trend towards
the procurement of micro-samples of blood by skin prick increases the
chance of secondary contamination of the blood. Systematic
investigation on the significance of this problem has been reported
(Mitchell et al., 1974; Mahaffey et al., 1979; DeSilva & Donnan,
1980). Mitchell et al. (l974) describe a procedure whereby sample
contamination can be reduced by spraying collodion over the cleansed
skin before lancing. The correlation between the concentration of lead
in micro-samples and in macro-samples obtained by venepuncture was
fairly good (r=0.92) over a wide range of PbB concentrations
(0.48-4.41 µmol/litre or 10-92 µg/dl whole blood). Mahaffey et al.,
(1979b) found that capillary blood levels in a comparison test were
systematically higher than corresponding venous blood levels; similar
elevations have been reported by DeSilva & Donnan (1980). Since about
1980 the requirement for reliable and accurate micro procedures has
resulted in the development of good protocols. Sinclair & Dohnt (1984)
described a procedure which resulted in the ability to collect
capillary samples with PbB levels only 3.3% higher than the presumably
correct venous value. This procedure has been used in the Port Pirie
Cohort Study (Baghurst et al., 1985, 1992) and for routine
surveillance in the Port Pirie Lead Decontamination Program (Calder et
al., 1990). Also, Lyngbye et al. (1990b) have shown that capillary
sampling without lead contamination is possible. Routine validation by
cross-comparison with venous blood samples should be undertaken on a
regular basis.
The same general precautions to avoid contamination must be taken
in the collection of urine samples as in the collection of blood
samples. Additionally, special care must be taken to prevent
precipitation during storage.
2.2.2 Analytical methods for lead
A number of analytical methods exist for determination of lead in
environmental and biological samples. These methods differ enormously
in their costs (e.g., sophisticated equipment, an adequate
infrastructure to maintain laboratory conditions and chemical
supplies) and personnel requirements (e.g., availability of skilled
personnel in adequate numbers for the work to be undertaken). Both
accuracy and precision of any of the methods can be affected greatly
by contamination of samples within the laboratory. It is important to
utilize the principles of a "clean" laboratory described by Patterson
& Settle (1976) and Everson & Patterson (1980).
It is not the purpose of this section to provide an exhaustive
description of the analytical methods that could be available to
detect and quantify lead levels in environmental and biological
samples. However, an attempt will be made to identify well-established
methods in current use and to provide information on their application
to assist in the interpretation of experimental and epidemiological
studies.
2.2.2.1 Analysis of lead in environmental samples
The most common methods used for the analysis of lead in samples
from air, water, dust, sediment, soil and foodstuffs are flame atomic
absorption spectrometry (AAS), graphite furnace atomic absorption
spectrometry (GFAAS), anodic stripping voltametry (ASV), inductively
coupled plasma-atomic emission spectroscopy (ICP-AES), and X-ray
fluorescence spectroscopy (XRFS). The reference method for the
determination of the absolute amounts of lead is by isotope dilution
mass spectrometry (IDMS) (Settle & Patterson, 1980; Grandjean & Olsen,
1984; US EPA, 1986a), but due to equipment costs and required
expertise, it is not widely used. Spectrophotometric methods, using
diphenylthiocarbazone as the colorimetric reagent, were widely used in
the past; they are less sensitive and are labour-intensive but are
still appropriate. The advantages and disadvantages were described by
Skogerboe et al. (1977).
Gould et al. (1988) utilized a citric acid solution on filter
paper to leach lead from glazed ceramic and/or enamelled metal-ware.
When treated with a lead-sensitive chromogen, there is a reaction
indicating the presence of lead on the paper. The minimal amount of
lead required to produce an observable reaction was 0.25 µg/cm2; the
maximum amount tested was 5 µg/cm2. A colorimetric test based on the
use of sodium sulfide in solution is used to estimate lead in paint
films. It is possible to determine lead concentrations greater than
1 mg/cm2 of dried paint 90% of the time when the method is used by a
trained chemical laboratory technician.
Table 2 summarizes the utility of several representative methods
for specific environmental media.
2.2.2.2 Analysis of lead in biological materials
Biological samples present special problems for the analyst
because of the low lead concentrations and matrix effects. Most
analytical techniques developed to detect and quantify lead can be
adapted to the analysis of such biological materials as blood, urine,
serum, cerebrospinal fluid, solid tissues, hair, teeth and bone.
However, certain techniques are more often used for specific matrices.
Currently, the most commonly used methods are AAS, GFAAS, ASV,
and ICP-AES. Spectrophotometric methods were commonly used in the past
and can be useful. Other specialized methods for lead analysis are
XRFS, neutron activation analysis (NAA), inductively coupled
plasma-mass spectrometry (ICP-MS), and IDMS. Table 3 summarizes the
utility of several analytical procedures applied to various biological
matrices. Included in this table are examples of the application of
XRFS (Christoffersson et al., 1986; Wielopolski et al., 1986; Nilsson
et al., 1991) for the determination in situ of the body burden of
lead.
2.2.2.3 Analytical procedures for biomarkers of lead exposure and
effect
Using standard clinical laboratory techniques, analytical
procedures have been developed: delta-aminolaevulinic acid (ALA);
delta-aminolaevulinic acid dehydratase (ALAD); urinary coproporphyrin
(CPU) and erythrocyte protoporphyrin (EP). All of these assays are
well established and reliable (Grandjean & Olsen 1984; US EPA, 1986a).
These biochemical parameters are influenced by physiological factors
other than lead. They lack the specificity and sensitivity of PbB
measurements as an index of either current lead exposures or body
stores of lead.
2.3 Conversion factors
1 µg/dl = 0.048 µmol/litre
1 µmol/litre = 20.7 µg/dl
Using the above conversion factor, blood lead concentrations are
given as µmol/litre with the equivalent µg/dl in brackets. Calculated
figures have not been rounded and added precision is not to be
inferred from the number of significant figures.
Table 2. Analytical methods for determining lead in environmental samplesa
Sample type Preparation method Analytical method Sample detection Percentage Reference
limit recovery
Air collect particulate matter on membrane ASV with mercury-graphite 0.16 µg/m3 90-110 NIOSH (1977b)
(particulate filter; wet ash with HNO3/HClO4/H2SO4; electrode (NIOSH method
lead) dissolve in acetate buffer P&CAM 191)
Air collect particulate matter on cellulose ICP-AES (NIOSH method 0.34 µg/m3 95-105 NIOSH (1981)
(particulate acetate filter; wet ash with HNO3/HClO4 P&CAM 351)
lead)
Air collect particulate matter on filter; AAS 0.1 µg/m3 93 Scott et al.
(particulate dry ash; extract with HNO3/HCl; dilute AES 0.15 µg/m3 102 (1976)
lead) with HNO3
Air sample on cellulose acetate filter; AAS 8 ng/litre 100-101 Nerin et al.
(particulate dissolve in HNO3 with heat; add HCl/H2O2 (1989)
lead) and react in hydride generator with sodium
borohydride to generate lead hydride
Air collect sample on filter; spike filter with IDMS 0.1 ng/m3 NR Volkening et
(particulate 206Pb; dissolve filter in NaOH; acidify; al. (1988)
lead) separate lead by electrodeposition; dissolve
in acid
Water digest sample with acid; heat; dilute with AAS 1.0 ng/g NR Chau et al.
(total lead) water (1979)
Soil dry sample and sieve for XRF; digest sieved XRF NR 65-98 Krueger &
sample with HNO3 and heat for AAS AAS NR 63-68 Duguay (1989)
Soil dry sample, dry ash; digest with acid AAS 2 µg/g 79-103 Beyer &
and dilute with water Cromartie (1987)
Table 2 (cont'd)
Sample type Preparation method Analytical method Sample detection Percentage Reference
limit recovery
Soil, waste, digest sample with acid; dilute with water AAS (EPA method 7420) 0.1 mg/litre NR US EPA (1986b)
and ground and filter GFAAS (EPA method 7421) 1 µg/litre NR
water
Soil, dust digest sample with hot acid; dry; redissolve AAS 12 ng/g > 80 Que Hee et al.
and paint in HNO3 (1985b)
Sediment, fish, digest sample with acid; heat; dilute with AAS 50 ng/g NR Chau et al.
vegetation water (sediment) (1980)
(total lead) 10 ng/g (fish NR
and vegetation)
Milk add 50 µl (C2H5)4NOH in ethanol to 25 µl GFAAS NR NR Michaelson &
milk; heat and dilute with water to 125 µl Sauerhoff (1974)
Evaporated dry ash sample; dissolve in HNO3 ASV 0.005 µg/g 99 Capar & Rigsby
milk (1989)
Agricultural dry ash sample with H2SO4 and HNO3; DPASV 0.4 ng/g 85-106 Satzger et al.
crops dilute with water (1982)
Grains, milk, bomb digest sample with acid; heat or GFAAS 20 µg/g (bomb) 85-107 Ellen & Van
mussels, fish digest with acid and dry ash; dissolve 5 µg/g (dry ash) 75-107 Loon (1990)
in acid; dilute with water DPASV NR 82-120
Table 2 (cont'd)
Sample type Preparation method Analytical method Sample detection Percentage Reference
limit recovery
Citrus leaves chop or pulverize sample; digest with hot ICP-AES 10-50 µg/litre 75-82 Que Hee &
and paint acid; dry; redissolve in acid (citrus Boyle (1988)
leaves)
89-96
(paint)
a AAS = atomic absorption; AES = atomic emissions spectroscopy; ASV = anode stripping voltametry; (C2H5)4NOH = tetraethylammonium
hydroxide; DPASV = differential pulse anodic stripping voltametry; EPA = US Environmental Protection Agency; GFAAS = graphite furnace
atomic absorption spectrometry; HCl = hydrochloric acid; HClO4 = perchloric acid; HNO3 = nitric acid; H2O2 = hydrogen peroxide;
H2SO4 = sulfuric acid; ICP-AES = inductively coupled plasma/atomic emission spectroscopy; IDMS = isotope dilution mass spectrometry;
NaOH = sodium hydroxide; NIOSH = National Institute for Occupational Safety and Health; NR = not reported; XRF = X-ray fluorescence
Table 3. Analytical methods for determining lead in biological materialsa
Sample Preparation method Analytical method Sample detection Percentage Reference
type limit recovery
Blood wet ash sample with acid mixtures; ASV with mercury-graphite 0.192 µmol/litre 95-105 NIOSH (1977c)
dissolve residue in dilute HClO4 electrode (NIOSH method (4 µg/dl)
P&CAM 195)
Blood wet ash sample with HNO3; dissolve GFAAS (NIOSH method 0.48 µmol/litre NR NIOSH (1977e)
residue in dilute HNO3 P&CAM 214) (10 µg/dl)
Blood dilute sample with Triton X-100(R); add GFAAS 0.011 µmol/litre 93-105 Aguilera de
nitric acid and diammonium phosphate (0.24 µg/dl) Benzo et al.
(1989)
Blood dilute sample with ammonia solution ICP-MS 0.072 µmol/litre 96-111 Delves &
containing Triton X-100(R); analyse (1.5 µg/dl) Campbell
(1988)
Blood dilute sample in 0.2% Triton X-100(R) GFAAS approx. 0.072 97-150 Que Hee et
and water; analyse µmol/litre (approx. al. (1985a)
1.5 µg/dl)
Blood and wet ash sample with HNO3, complex Spectrophotometry 0.144 µmol/litre 97 NIOSH (1977a)
urine with dephenylthiocarbazone and (NIOSH method (3.0 µg/dl) (blood);
extract with chloroform P&CAM 102) 0.0576 µmol/litre 97
(12 µg/litre) (urine)
Serum, filter sample if needed; dilute with ICP-AES 0.048-0.240 µmol/litre 85 (serum) Que Hee &
blood and acid or water (1.0-5.0 µg/dl) Boyle (1988)
urine
Urine wet ash sample with acid mixture and ASV with mercury-graphite 0.0192 µmol/litre 90-110 NIOSH (1977d)
dissolve in dilute HClO4 electrode (NIOSH method (4 µg/litre)
P&CAM 200)
Table 3 (cont'd)
Sample Preparation method Analytical method Sample detection Percentage Reference
type limit recovery
Liver, bomb digest sample with acid and heat, GFAAS 20 µg/g (bomb); 85-107 Ellen & Van
kidney, or digest with acid and dry ash; dissolve (bomb); Loon (1990)
muscle in acid; dilute with water 5 µg/g (dry ashing) 75-107 (dry
ashing)
Bone direct partially polarized photons at XRF 20 µg/g NR Christoffersson
second phalanx of left forefinger et al. (1986)
(non-invasive technique)
Bone direct partially polarized photons at XRF 20 µg/g NR Wielopolski
anteromedial skin surface of mid-tibia et al. (1986)
(non-invasive technique)
Teeth clean and section tooth; digest with ASV NR 83-114 Rabinowitz
HNO3; evaporate; redissolve in buffer et al. (1989)
solution
Teeth dry ash sample; crush; dry ash again; AAS NR 90-110 Steenhout &
dissolve in HNO3 Pourtois
(1981)
a AAS = atomic absorption spectrometry; ASV = anode stripping voltametry; GFAAS = graphite furnace atomic absorption spectrometry;
HClO4 = perchloric acid; HNO3 = nitric acid; ICP-AES = inductively coupled plasma-atomic emission spectroscopy;
ICP-MS = inductively coupled plasma-mass spectrometry; NIOSH = National Institute for Occupational Safety and Health;
NR = not reported; XRF = X-ray fluorescence
3. SOURCES OF HUMAN AND ENVIRONMENTAL EXPOSURE
3.1 Natural occurrence
Because lead is relatively abundant in the earth's crust it is
found naturally throughout the world. The major natural sources of
lead are volcanic emissions, geochemical weathering, and emissions
from sea spray. A small amount of radioisotopic lead (207Pb) is
derived from the decay of radon gas released from geological sources.
It has been estimated that the worldwide natural emission rates of
lead are of the order of 19 000 tonnes/year (Nriagu & Pacyna, 1988),
with volcanic sources accounting for 6400 tonnes/year (Nriagu, 1979).
Owing to centuries of human exploitation of lead resources, it is
difficult to determine the natural content of lead in most ecosystems.
Data on environmental levels, uses, and sources of lead have been
summarized in a recent review (OECD, 1993).
3.1.1 Rocks and soils
The average concentration of lead in the earth's crust is between
10 and 20 mg/kg (IPCS, 1989). The major geological sources of lead are
in igneous and metamorphic rocks.
The soil is the most important repository in terrestrial
ecosystems for contaminants of anthropogenic origin (Nriagu & Pacyna,
1988; Nriagu, 1989). The lead content of soils (which are for
discussion purposes distinguished here from surface dusts) is greatly
influenced by anthropogenic activities and by long- and short-range
airborne transport of lead from various sources. Both dry and wet
deposition are important routes of input.
Lead in soil may be relatively insoluble (as a sulfate, carbonate
or oxide), soluble, adsorbed onto clays, adsorbed and coprecipitated
with sesquioxides, adsorbed onto colloidal organic matter, or
complexed with organic moieties in soil (US EPA, 1986a; IPCS, 1989).
Soil pH, content of humic and fulvic acids, and amount of organic
matter influence the content and mobility of lead in soils. Since
acidic conditions favour the solubilization and leaching of lead from
the solid phase, acidic soils tend to have lower lead concentrations
when analysed as dry soil. Humic and fulvic acids can also mobilize
lead, and certain complex organic molecules can act as chelators of
lead (IPCS, 1989).
Background levels of lead in soil are in the range of 10-70 mg/kg
(GEMS, 1985). Similar results have been found in studies of mobile
source contamination near highways; soil taken at distances of
50-100 m from highways (outside the range of immediate impact from
traffic emissions) usually shows levels of lead below 40 mg/kg. In the
1985, GEMS survey of selected countries, lead concentrations in
topsoil from Malta were found to have a mean of 54 mg/kg in areas at
least 5 m from roadways; less than one metre from roadways the mean
concentration was 138 mg/kg. A 1977 report from Sweden found a mean of
16 mg/kg in non-contaminated areas (GEMS, 1985).
3.1.2 Sediments
Sediments from freshwater and marine environments have been
studied for lead content. This compartment provides a unique record of
the history of changes in global lead fluxes (Patterson, 1983). Levels
of lead in sediments dated before the onset of the industrial
revolution in Western Europe show very low levels, less than 10% of
current levels (Flegal et al., 1987). The average background level of
lead in marine sediments off southern California was reported by
Flegal et al. (1987) to be 1.3 mg/kg.
3.1.3 Water
Flegal et al. (1987) estimate that the natural concentration of
lead in surface water is about 0.02 µg/litre. In general, lead is not
found in ground or surface waters at concentrations above 10 µg/litre
(IPCS, 1989).
Data from oceans indicate very low levels of lead in sea-water
samples not affected directly by significant sources of lead. Water
samples taken from an area of the Pacific, where annual windborne-
input fluxes of lead are estimated to be 3 mg/cm2, have lead
concentrations of 3.5 ng/litre (0-100 m depth) and 0.9 ng/litre at
depths greater than 2500 m. In contrast, water samples taken from the
north Atlantic, where annual windborne-input fluxes of lead are
170 mg/cm2, contain 34 ng lead/litre at the surface and 5 ng/litre
in depths below 2500 m (Patterson, 1983). Settle & Patterson (1980)
have estimated that prehistoric oceans contained 0.5 ng/litre lead.
Flegal et al. (1987) have estimated that over 95% of the lead in
off-shore surface waters is the result of windborne inputs. However,
in coastal waters near Monterey (California, USA), higher
concentrations of lead were found in sea water, sediments and
organisms; these elevations were related to specific sources by
systematic isotope analyses (Flegal et al., 1987).
3.1.4 Air
Anthropogenic inputs of lead from a range of sources have
resulted in global dispersion of both inorganic and organic species of
lead into the air, of which 80-90% is derived from alkyllead fuel
additives (WHO, 1987). Nriagu & Pacyna (1988) estimated that a total
of 330 000 tonnes of lead is discharged directly into the atmosphere
each year. Estimations of pre-industrial levels of lead in air from
natural origins (volcanic emissions, crustal weathering, radon decay
and sea-spray releases) are in the range of 0.01-0.1 µg/m3 (US NRC,
1980). The lowest level reported since 1975 is 0.076 ng/m3 measured
at the South Pole (US EPA, 1986a).
3.1.5 Plants
Lead occurs naturally in plants and results from both deposition
and uptake. There is a positive linear relationship between lead
concentrations in plants and soil (Davies & Thornton, 1989). As with
other environmental compartments, measurement of "background" levels
of lead in plants is complicated by the general contamination of the
globe from centuries of lead use, which has included direct
application of lead-containing chemicals in agriculture (see below)
and contamination of fertilizers with lead. Lead has been measured in
superphosphate fertilizer at concentrations as high as 92 mg/kg (Lisk,
1972). Sewage sludge, used as a source of nutrients in agriculture,
may contain even higher levels of lead. The concentration of lead in
sewage sludge is typically < 1000 mg/kg. Levels as high as 26 g/kg
have been measured in the USA (Chaney et al., 1984). Soil receiving
heavy sludge applications over long periods of time (years) contained
425 mg lead/kg; the concentration in untreated soil was 47 mg/kg
(Beckett, 1979).
3.1.6 Environmental contamination from natural sources
The contribution of natural sources of lead to human exposure is
small. As a result of various breakdown processes, rocks yield lead
which is transferred to the biosphere and the atmosphere, and,
ultimately, back to the earth's crust in the form of sedimentary
rocks. Soluble lead has for thousands of years entered the oceans with
river discharges, and the rate has been estimated by Patterson (1965)
to be around 17 000 tonnes/year. Sources contributing to airborne lead
are silicate dusts, volcanic halogen aerosols, forest fires, sea salts
aerosols, meteoric and meteorite residues, and lead derived from the
decay of radon. While the lead content of most coals is relatively
low, coal fly ash is enriched in lead (Hutton et al., 1988) and is a
source of environmental contamination.
3.2 Anthropogenic sources
World lead consumption has steadily increased over the period
1965-1990 and was about 5.6 × 106 tonnes in 1990 (OECD, 1993).
Further review of the data summarized by OECD (1993) indicates a
change in consumption patterns worldwide. Although the consumption of
lead within the 24 countries of the OECD increased only slightly over
the decade from 1980 to 1990, consumption within less developed
economies (Africa and Asia) increased from 315 000 tonnes in 1970 to
844 000 tonnes in 1990.
3.2.1 Lead mining
Lead occurs in a variety of minerals, the most important of which
are galena (PbS), cerrusite (PbCO3) and anglesite (PbSO4). Galena
is by far the most important source of primary lead. It occurs mostly
in deposits associated with other minerals, particularly those
containing zinc. Mixed lead and zinc ores account for about 70% of
total primary lead supplies. Ores containing mainly lead account for
about 20% and the remaining 10% is obtained as a by-product from other
deposits, such as mixed copper-zinc deposits. The proportions of
various metals may differ in the ores of different countries. Silver
is the most important of the other metals frequently present in lead
deposits, but copper may also be present in concentrations high enough
to be commercially important. Other minor constituents of lead ores
are gold, bismuth, antimony, arsenic, cadmium, tin, gallium, thallium,
indium, germanium and tellurium.
The major countries producing lead from mining activity during
1987-1991 were the USA, Canada, Australia, Peru, the former USSR and
Mexico, as shown in Table 4. Other countries producing lead from lead
ores include China, the former Yugoslavia, Morocco, Spain, Sweden and
Tunisia. In general, the level of world production of lead from mining
activities has remained relatively constant at about 3.3 × 106
tonnes between 1988 and 1991 (ILZSG, 1992); this represents roughly
60% of the world demand for lead.
Table 4. Major countries producing lead from ore and ore
concentratesa
Country 1987 1988 1989 1990 1991
Canada 423 200 366 600 276 100 241 300 278 100
USA 318 300 395 700 419 300 495 200 483 300
Ex-USSR 510 000 520 000 500 000 490 000 --
Australia 489 200 462 000 495 000 570 000 579 000
Mexico 177 200 178 100 163 000 174 100 158 800
Peru 204 000 149 000 192 200 187 800 199 100
a From: World Bureau of Metal Statistics (1992)
3.2.2 Smelting and refining
Smelting and refining are classified as either primary or
secondary, the former producing refined lead products from ores or
concentrates (primary lead) and the latter producing lead by
recovering it from lead-bearing scrap and waste materials (secondary
lead). Secondary lead is derived from processing what is termed new
scrap arising during manufacturing processes and recycled old scrap
arising from waste materials containing lead. Most scrap is from old
sources, of which the most important are lead plates from batteries,
solder, common babbitt, soft lead, lead solders, cable coverings, type
metals, dross and other lead-containing products. There has been an
increasing contribution of secondary lead sources to the total
worldwide production of lead, as shown in Table 5 (World Bureau of
Metal Statistics, 1992). Secondary sources of lead supplied between 35
and 40% of world production during the period from 1970 to 1990.
3.2.3 Environmental pollution from production of lead
Mining operations and the smelting and refining of both primary
and secondary lead are known to cause contamination of the nearby
environment. The nature and extent of contamination depends on many
factors, including the level of production, the effectiveness of
emission controls, climate, topography and other local factors.
Concentrations are usually highest within 3 km of the point source (US
EPA, 1989). A report from China found that lead levels in ambient air,
plants and soil increased proportionally with proximity to a large
primary smelter; at 50 m from the source, the air lead level was
60 µg/m3, the lead level in plants was 29.1 mg/kg, and soil lead
level was 170 mg/kg (Wang, 1984). However, some earlier studies have
shown air pollution and soil contamination as far as 10 km from
smelters (Djuric et al., 1971; Kerin, 1973; Landrigan et al., 1975).
Table 5. Relative contribution of primary and secondary sources
relative to world lead production (1987-1990)a
1987 1988 1989 1990
Primary 422 100 3 414 200 3 286 500 3 324 500
Secondary 2 045 600 2 103 900 2 272 900 2 254 800
a From: World Bureau of Metal Statistics (1992)
The impacts of lead mining and smelting can persist for long
periods of time. A study conducted in Wales, United Kingdom, in an
area where lead mining began 2000 years ago and ended in the middle of
the 20th century, found high concentrations of lead in soils (Davies
et al., 1985). In Port Pirie, Australia, a community with one of the
world's largest and oldest primary lead smelters, lead levels in soils
were found to be grossly elevated, and the incidence of elevated blood
lead levels in pregnant women and young children was also increased
above that found in other communities in Australia (Wilson et al.,
1986).
3.3 Consumption and uses of lead and its compounds
Lead has a combination of physical and chemical properties that
have made it extremely useful industrially, i.e. high density, high
opacity to gamma and X-ray energies, low sound conductance, a low
melting point, exceptional malleability, high corrosion resistance,
and stability. In 1990, 5.627 × 106 tonnes of lead were consumed
worldwide (ILZSG, 1992). The twenty-four industrialized countries of
the OECD consumed approximately 65% of this amount, with eastern
Europe and the former USSR using 21%. Asia now utilizes about 9% of
the world's lead production.
The use patterns of refined lead vary from country to country.
The situation in 1990 in Mexico is shown in Table 6, while end-use
categories within OECD countries are summarized in Fig. 1, which
indicates the changes between 1970 and 1990 (OECD, 1993).
Table 6. Principal uses of refined lead in Mexicoa
Type of product 1988 1990
(%) (%)
Oxides 69.7 56.7
Batteries 9.2 17.9
Tetraethyllead 7.9 11.9b
Cables 4.0 1.5
Others 9.2 11.9
a ILZSG (1992)
b This does not reflect the introduction of lead-free petrol
in 1990.
From Fig. 1, it is evident that the largest use of lead within
OECD countries is for battery production, whereas there has been a
large drop in the demand for lead-containing gasoline additives.
However, this pattern is not valid worldwide, e.g., concentrations in
petrol range from zero in such countries as Japan and Thailand to
1.12 g/litre in the Virgin Islands (Octel, 1991).
In the past the use of lead in the chemical industry for the
preparation of paints, pigments and coloured inks was widespread. Many
countries have now restricted this use, and concentrations of lead
greater than 0.06% (USA) and 0.5% (New Zealand) are not permitted in
indoor paints (Albert & Badillo, 1991; OECD, 1993). In 1982, data from
the United Kingdom (UK, 1982) indicated levels of lead between 2500
and 3000 mg/kg in decorative glass paints and up to 448 g/kg in
white-lead primer. Red-leadcontaining paints, still used widely to
paint structural steel works, can contain up to 661 g lead/kg.
 Other disperse uses of lead include lead solders (now banned in
USA for use in drinking-water systems), ammunition (Novotny et al.,
1987), foil on wine bottles (Wai et al., 1979) and cosmetics and
folk-medicines (surma in Asia, Kohl in India, and Al Kohl in Saudi
Arabia and Kuwait) (Fernando et al., 1981).
3.4 Sources of environmental exposure
As noted above, lead is a ubiquitous pollutant in the global
ecosystem, as well as occurring naturally. Its uses have resulted in
increases in soil, water and air lead levels to one to two orders of
magnitude above those estimated to have prevailed prior to rapid
industrialization in the 18th and 19th centuries (Patterson, 1983).
Whereas in specific areas point sources may contribute significant
amounts of lead to the environment, on a global scale, the combustion
of alkyllead in petrol is the predominant source of increased lead in
all compartments of the environment. This has been hypothesized based
upon mass balance studies (Nriagu, 1979) and confirmed by the changes
in environmental lead levels which have followed the significant
reductions in worldwide use of alkyllead as a gasoline additive since
the mid-1980s. For example, lead concentrations in Greenland snow
decreased by a factor of 7.5 over a 20-year period from the late 1960s
(Boutron et al., 1991).
Nriagu & Pacyna (1988) have estimated the global emissions of
lead to the atmosphere resulting from anthropogenic uses (Table 7).
Current estimates (OECD, 1993) of emissions from mobile sources would
be about 30% of the 1983 amounts. Similarly estimates of emissions of
lead to soil in 1983 were made by Nriagu & Pacyna (1988) (Table 8).
Since lead is never degraded, all lead which is shifted from
geological sources by human technology eventually enters the
environment through disposal, although this can be slowed by recycling
and recovery.
Municipal solid waste (MSW), solid waste, hazardous waste, sewage
sludge, and industrial waste-water discharges all may contain lead at
concentrations as high as 50 g/kg. Although few measurements of
environmental lead concentrations in the vicinity of disposal sites
have been conducted, analyses of fly and bottom ash from municipal
incinerators show high concentrations (up to 50 g/kg) of lead (Wadge &
Hutton, 1987), and land disposal sites which have received incinerator
ash for a number of years show high levels of lead in soil (Hutton et
al., 1988).
Dusting and flaking of lead paint from surfaces can be a source
of lead contamination in surface dust and soil near houses or
buildings as well as contributing to the concentrations of lead in
household dust. This process is a function of the type of paint and
the age and state of repair of the structure. When lead paint is
present on structures, both interior and exterior dusts have higher
concentrations than otherwise would be expected (Thornton et al.,
1985). Abatement of lead paint may be a major local source of
environmental contamination, as shown by studies near school buildings
in London (Rundle & Duggan, 1986). Removal of lead-based paints from
bridges and water towers using improper techniques can also result in
significant environmental contamination. Direct application of
lead-contaminated sludge as fertilizers, and residues of lead arsenate
from use in agriculture can lead to the contamination of soil, surface
water and ground water. In local aquatic environments, pollution can
result from leaching of lead from lead shot, shotgun cartridges and
fishing weights (IPCS, 1989). Coal contains small amounts of lead,
which can be concentrated in fly ash from coal combustion (Wadge &
Hutton, 1987) or in stack emissions (Table 8).
Table 7. Estimated worldwide anthropogenic emissions of lead to
the atmosphere (1983)a
Source category Emission rate (tonnes/year)
Coal combustion
- electric utilities 780-4650
- industry and domestic 990-9900
Oil combustion
- electric utilities 230-1740
- industry and domestic 720-2150
Pyrometallurgical non-ferrous metal production
- mining 1700-3400
- lead production 11 700-31 200
- copper-nickel production 11 000-22 100
- zinc-cadmium production 5520-11 500
Secondary non-ferrous metal production 90-1440
Steel and iron manufacturing 1070-14 200
Refuse incineration
- municipal 1400-2800
- sewage sludge 240-300
Phosphate fertilizers 60-270
Cement production 20-14 200
Wood combustion 1200-3000
Mobile sourcesb 248 030
Miscellaneous 3900-5100
Total 289 000-376 000
(median 332 000)
a Adapted from: Nriagu & Pacyna (1988), as in OECD (1993).
b Current estimates (OECD, 1993) for mobile source emissions
would be about 30% of the 1983 amounts.
Table 8. Worldwide emissions of lead into soils (1983)
Source category Emission rate (tonnes/year)
Agricultural and food wastes 1500-27 000
Animal wastes, manure 3200-20 000
Logging and other wood wastes 6600-8200
Urban refuse 18 000-62 000
Municipal sewage sludge 2800-9700
Miscellaneous organic wastes, including excreta 20-1600
Solid wastes, metal manufacturing 4100-11 000
Coal fly ash, bottom fly ash 45 000-242 000
Fertilizer 420-2300
Peat (agricultural and fuel use) 450-2000
Wastage of commercial products 195 000-390 000
Atmospheric fall-out 202 000-263 000
Total yearly input to soils 479 090-1 038 800
Mine tailings 130 000-390 000
Smelter slags and wastes 194 000-390 000
Total yearly discharge on land 803 090-1 818 800
a From: Nriagu & Pacyna (1988), adapted from OECD (1993);
many of these emissions remain localized due to the nature of
the particulate matter
4. ENVIRONMENTAL TRANSPORT, DISTRIBUTION AND TRANSFORMATION
Over the last 10-15 years, a great many studies have been
conducted on the complex interrelationships between environmental lead
emissions and their deposition on such environmental surfaces as
vegetation, soil, house dust and water. All are potential sources of
lead exposure for humans. Transport between environmental compartments
also takes place (see Fig. 2). A full discussion of the complex
physical and chemical processes controlling these pathways is beyond
the scope of this monograph and the reader is directed towards other
reviews for more details (Elias, 1985; US EPA, 1986a; IPCS, 1989).
4.1 Transport and distribution between media
4.1.1 Atmospheric deposition
From the mass balance point of view the transport and
distribution of lead from major emission sources, both fixed and
mobile, is mainly atmospheric. Most of the lead discharged to the
atmosphere is deposited near the source. However, approximately 20% is
widely dispersed (Nriagu, 1979; IPCS, 1989) and contaminates areas as
remote as glacial strata in Greenland (Settle & Patterson, 1980). The
extent of long-range transport of lead particles is dependent upon
particle size, particles > 2 µm in diameter being deposited close to
the source of emission. Between 20 and 60% of emissions from vehicles
has been reported to remain within 25 m of the roadway (ATSDR, 1991).
However, in view of the marked decrease in the concentration of lead
in cores of ice from Greenland since the decreased use of leaded
petrol (Boutron et al., 1991), it is apparent that vehicle emissions
can contribute to the levels of lead in air far from the source.
Long-range transport of lead particles was also noted by Evans &
Rigler (1985).
Lead can be removed from the atmosphere and transferred to
environmental surfaces and compartments by wet or dry deposition. Wet
deposition appears to be more important than dry deposition for the
removal of atmospheric lead. Depending upon geographical location and
the level of emissions in the area, between 40 and 70% of atmospheric
lead is removed by wet deposition (Nielsen, 1984). In most cases it is
poorly soluble and either precipitates out in soils and sediments or
is bound to organic matter in these compartments. For these reasons
lead is not readily removed and tends to accumulate in those
ecosystems where it is deposited (IPCS, 1989). Chan et al. (1986)
calculated the ratio of wet to dry deposition to be 1.63, 1.99 and
2.50 for sites in south, central, and northern Canada, respectively,
while Talbot & Andren (1983) reported that wet deposition accounted
for 80% of the total lead deposited in a semi-remote site in the USA.
Other disperse uses of lead include lead solders (now banned in
USA for use in drinking-water systems), ammunition (Novotny et al.,
1987), foil on wine bottles (Wai et al., 1979) and cosmetics and
folk-medicines (surma in Asia, Kohl in India, and Al Kohl in Saudi
Arabia and Kuwait) (Fernando et al., 1981).
3.4 Sources of environmental exposure
As noted above, lead is a ubiquitous pollutant in the global
ecosystem, as well as occurring naturally. Its uses have resulted in
increases in soil, water and air lead levels to one to two orders of
magnitude above those estimated to have prevailed prior to rapid
industrialization in the 18th and 19th centuries (Patterson, 1983).
Whereas in specific areas point sources may contribute significant
amounts of lead to the environment, on a global scale, the combustion
of alkyllead in petrol is the predominant source of increased lead in
all compartments of the environment. This has been hypothesized based
upon mass balance studies (Nriagu, 1979) and confirmed by the changes
in environmental lead levels which have followed the significant
reductions in worldwide use of alkyllead as a gasoline additive since
the mid-1980s. For example, lead concentrations in Greenland snow
decreased by a factor of 7.5 over a 20-year period from the late 1960s
(Boutron et al., 1991).
Nriagu & Pacyna (1988) have estimated the global emissions of
lead to the atmosphere resulting from anthropogenic uses (Table 7).
Current estimates (OECD, 1993) of emissions from mobile sources would
be about 30% of the 1983 amounts. Similarly estimates of emissions of
lead to soil in 1983 were made by Nriagu & Pacyna (1988) (Table 8).
Since lead is never degraded, all lead which is shifted from
geological sources by human technology eventually enters the
environment through disposal, although this can be slowed by recycling
and recovery.
Municipal solid waste (MSW), solid waste, hazardous waste, sewage
sludge, and industrial waste-water discharges all may contain lead at
concentrations as high as 50 g/kg. Although few measurements of
environmental lead concentrations in the vicinity of disposal sites
have been conducted, analyses of fly and bottom ash from municipal
incinerators show high concentrations (up to 50 g/kg) of lead (Wadge &
Hutton, 1987), and land disposal sites which have received incinerator
ash for a number of years show high levels of lead in soil (Hutton et
al., 1988).
Dusting and flaking of lead paint from surfaces can be a source
of lead contamination in surface dust and soil near houses or
buildings as well as contributing to the concentrations of lead in
household dust. This process is a function of the type of paint and
the age and state of repair of the structure. When lead paint is
present on structures, both interior and exterior dusts have higher
concentrations than otherwise would be expected (Thornton et al.,
1985). Abatement of lead paint may be a major local source of
environmental contamination, as shown by studies near school buildings
in London (Rundle & Duggan, 1986). Removal of lead-based paints from
bridges and water towers using improper techniques can also result in
significant environmental contamination. Direct application of
lead-contaminated sludge as fertilizers, and residues of lead arsenate
from use in agriculture can lead to the contamination of soil, surface
water and ground water. In local aquatic environments, pollution can
result from leaching of lead from lead shot, shotgun cartridges and
fishing weights (IPCS, 1989). Coal contains small amounts of lead,
which can be concentrated in fly ash from coal combustion (Wadge &
Hutton, 1987) or in stack emissions (Table 8).
Table 7. Estimated worldwide anthropogenic emissions of lead to
the atmosphere (1983)a
Source category Emission rate (tonnes/year)
Coal combustion
- electric utilities 780-4650
- industry and domestic 990-9900
Oil combustion
- electric utilities 230-1740
- industry and domestic 720-2150
Pyrometallurgical non-ferrous metal production
- mining 1700-3400
- lead production 11 700-31 200
- copper-nickel production 11 000-22 100
- zinc-cadmium production 5520-11 500
Secondary non-ferrous metal production 90-1440
Steel and iron manufacturing 1070-14 200
Refuse incineration
- municipal 1400-2800
- sewage sludge 240-300
Phosphate fertilizers 60-270
Cement production 20-14 200
Wood combustion 1200-3000
Mobile sourcesb 248 030
Miscellaneous 3900-5100
Total 289 000-376 000
(median 332 000)
a Adapted from: Nriagu & Pacyna (1988), as in OECD (1993).
b Current estimates (OECD, 1993) for mobile source emissions
would be about 30% of the 1983 amounts.
Table 8. Worldwide emissions of lead into soils (1983)
Source category Emission rate (tonnes/year)
Agricultural and food wastes 1500-27 000
Animal wastes, manure 3200-20 000
Logging and other wood wastes 6600-8200
Urban refuse 18 000-62 000
Municipal sewage sludge 2800-9700
Miscellaneous organic wastes, including excreta 20-1600
Solid wastes, metal manufacturing 4100-11 000
Coal fly ash, bottom fly ash 45 000-242 000
Fertilizer 420-2300
Peat (agricultural and fuel use) 450-2000
Wastage of commercial products 195 000-390 000
Atmospheric fall-out 202 000-263 000
Total yearly input to soils 479 090-1 038 800
Mine tailings 130 000-390 000
Smelter slags and wastes 194 000-390 000
Total yearly discharge on land 803 090-1 818 800
a From: Nriagu & Pacyna (1988), adapted from OECD (1993);
many of these emissions remain localized due to the nature of
the particulate matter
4. ENVIRONMENTAL TRANSPORT, DISTRIBUTION AND TRANSFORMATION
Over the last 10-15 years, a great many studies have been
conducted on the complex interrelationships between environmental lead
emissions and their deposition on such environmental surfaces as
vegetation, soil, house dust and water. All are potential sources of
lead exposure for humans. Transport between environmental compartments
also takes place (see Fig. 2). A full discussion of the complex
physical and chemical processes controlling these pathways is beyond
the scope of this monograph and the reader is directed towards other
reviews for more details (Elias, 1985; US EPA, 1986a; IPCS, 1989).
4.1 Transport and distribution between media
4.1.1 Atmospheric deposition
From the mass balance point of view the transport and
distribution of lead from major emission sources, both fixed and
mobile, is mainly atmospheric. Most of the lead discharged to the
atmosphere is deposited near the source. However, approximately 20% is
widely dispersed (Nriagu, 1979; IPCS, 1989) and contaminates areas as
remote as glacial strata in Greenland (Settle & Patterson, 1980). The
extent of long-range transport of lead particles is dependent upon
particle size, particles > 2 µm in diameter being deposited close to
the source of emission. Between 20 and 60% of emissions from vehicles
has been reported to remain within 25 m of the roadway (ATSDR, 1991).
However, in view of the marked decrease in the concentration of lead
in cores of ice from Greenland since the decreased use of leaded
petrol (Boutron et al., 1991), it is apparent that vehicle emissions
can contribute to the levels of lead in air far from the source.
Long-range transport of lead particles was also noted by Evans &
Rigler (1985).
Lead can be removed from the atmosphere and transferred to
environmental surfaces and compartments by wet or dry deposition. Wet
deposition appears to be more important than dry deposition for the
removal of atmospheric lead. Depending upon geographical location and
the level of emissions in the area, between 40 and 70% of atmospheric
lead is removed by wet deposition (Nielsen, 1984). In most cases it is
poorly soluble and either precipitates out in soils and sediments or
is bound to organic matter in these compartments. For these reasons
lead is not readily removed and tends to accumulate in those
ecosystems where it is deposited (IPCS, 1989). Chan et al. (1986)
calculated the ratio of wet to dry deposition to be 1.63, 1.99 and
2.50 for sites in south, central, and northern Canada, respectively,
while Talbot & Andren (1983) reported that wet deposition accounted
for 80% of the total lead deposited in a semi-remote site in the USA.
 Making several assumptions regarding global atmospheric lead
concentrations, wind speed, surface area and texture, a global
deposition of approximately 410 000 tonnes/year (combined wet and dry)
was calculated by the US EPA (1986a).
4.1.2 Transport to water and soil
When deposited in water, whether from air or through run-off from
soil, lead partitions rapidly between the sediment and aqueous phase,
depending upon the salt content of the water as well as the presence
of organic complexing agents. For example, at pH > 5.4 the total
solubility of lead is about 30 µg/litre in hard water and 500 µg/litre
in soft water (Davies & Everhart 1973). In addition, the presence of
sulfate and carbonate ions can limit lead solubility, as described by
Hem & Durum (1973) in a review of the aqueous chemistry of lead.
Water-borne lead has been found to exist as soluble lead or
undissolved colloidal particles, either suspended in the aqueous phase
or carried as surface coatings on other suspended solids (Lovering,
1976). The ratio of lead in suspended solids to lead in the dissolved
form has been found to vary from 4:1 in rural areas to 27:1 in urban
streams (Getz et al., 1977).
Both natural organic compounds (humic and fulvic acids) as well
as those of anthropogenic origin (e.g., ethylenediamino-tetraacetic
and nitrilotriacetic acids) may complex lead found in surface waters
(Steelink, 1977; Reuter & Perdue, 1977; Neubecker & Allen, 1983). The
presence in water of such chelators can increase the rate of solution
of lead compounds (e.g., lead sulfide) 10 to 60 times over that of
water at the same pH without fulvates (Bondarenko, 1968; Lovering,
1976).
Lead accumulation in soils is primarily a function of the rate of
wet and dry deposition from the atmosphere. Transport within soil and
the bioavailability of lead from soil are dependent upon many factors,
including pH, mineral composition of the soil, and amount and type of
organic material, with most of the lead being bound within the upper
5 cm of soil (Reaves & Berrow 1984; Garcia-Miragaya, 1984). This
limits the amount which can be leached into water or be available for
uptake into plants. It has been shown that only 0.2% of the total lead
in soil can be released into solution by shaking (Dong et al., 1985).
However, the release of lead from organic complexes to the soluble,
and thus bioavailable, form is highly pH dependent. Within the usual
pH range for soils (4 to 6), the organic-lead complexes become more
soluble and the lead more available for plant uptake and leaching into
water (US EPA, 1986a).
4.1.3 Transport to biota
The transfer of air lead to the biota may be direct or indirect
(uptake from water, soil and vegetation). Examples of the accumulation
of lead into aquatic (wet and dry deposition) and terrestrial
organisms are given in Environmental Health Criteria 85: Lead -
Environmental Aspects (IPCS, 1989). Relevant parts of that monograph
are summarized here.
4.1.3.1 Aquatic organisms
In aquatic and aquatic/terrestrial model ecosystems, uptake by
primary producers and consumers seems to be determined by the
bioavailability of the lead. Bioavailability is generally much lower
whenever organic material, sediment or mineral particles (e.g., clay)
are present. In many organisms, it is unclear whether lead is adsorbed
onto the organism or actually taken up. Consumers take up lead from
their contaminated food, often to high concentrations but without
biomagnification.
The uptake and accumulation of lead by aquatic organisms from
water and sediment are influenced by various environmental factors
such as temperature, salinity and pH, as well as humic and alginic
acid content.
In contaminated aquatic systems, almost all of the lead is
tightly bound to sediment. Only a minor fraction is dissolved in the
water, even in the interstitial water.
The lead uptake by fish reaches equilibrium only after a number
of weeks of exposure. Lead is accumulated mostly in gill, liver,
kidney and bone.
Fish eggs show increasing lead levels with increased exposure
concentration, and there are indications that lead is present on the
egg surface but not accumulated in the embryo.
Fish accumulate lead from water as well as sediments; aquatic
uptake is influenced by the presence of cations and the oxygen content
of the water (IPCS, 1989).
4.1.3.2 Terrestrial organisms
In bacteria, the majority of lead is found in the cell wall. A
similar phenomenon is also noted in higher plants. Some lead that
passes into the plant root cell can be combined with new cell wall
material and subsequently removed from the cytoplasm to the cell wall.
Of the lead remaining in the root cell, there is evidence of very
little translocation to other parts of the plant because the
concentration of lead in shoot and leaf tissue is usually much lower
than in root. Foliar uptake of lead occurs, but only to a very limited
extent.
In animals, there is a positive correlation between tissue and
dietary lead concentrations, although tissue concentrations are almost
always lower. The distribution of lead within animals is closely
associated with calcium metabolism.
Lead shot is typically trapped in the gizzard of birds where it
is slowly ground down resulting in the release of lead.
The tetravalent organic form of lead is generally more toxic than
the divalent inorganic form, and its distribution in organisms may not
specifically follow calcium metabolism.
4.2 Environmental transformation
4.2.1 Abiotic transformation
Once released into the environment lead may be transformed from
one inorganic species or particle size to another. However, as an
element it is not subject to degradation. For example, lead-containing
particles in automobile exhaust are usually lead halides or double
salts with ammonium halides (Biggins & Harrison, 1979). Within 24 h,
over 75% of lead particulate matter is transformed to lead carbonates
and sulfates (Olson & Skogerboe, 1975).
4.2.2 Biotransformation
The transformation of inorganic lead to tetramethyllead (TML) has
been observed in aquatic systems, particularly in sediments, and
biomethylation was postulated by Wong et al. (1975) and Schmidt &
Huber (1976). However, no biological methylation of inorganic lead was
noted by Reisinger et al. (1981) in studies under many conditions
using several bacterial species known to alkylate mercury and other
heavy metals. The authors did find chemical methylation in the
presence of methylcobalamin and sulfide or aluminium ions and it was
independent of the presence of bacteria. The evidence for microbial
methylation of various compounds of lead in aquatic systems has been
reviewed by Beijer & Jernelöv (1984). It is still unclear whether the
TML formed is produced abiotically or by biotransformation.
5. ENVIRONMENTAL LEVELS AND HUMAN EXPOSURE
This chapter describes the sources of lead to which people are
exposed and quantifies that exposure. Depending on the source, the
concentration of lead and its bioavailability, the relative
contribution of each source may vary considerably. For example, men
working in ship-breaking, children exposed to deteriorating lead
paint, and people consuming soft water distributed in lead pipes have
frequently been shown to have excessive absorption of lead, leading to
clinically obvious lead poisoning.
In evaluating the exposure of the general population to lead, it
is critical to consider the interrelationships among environmental
pathways for lead and transfers across environmental media. The
general population is exposed to lead simultaneously from many sources
and through multiple pathways as shown in Fig. 2. Thus, while, for
purposes of discussion, exposure via air, water, food, dusts and
soils, and other sources are presented separately in this document,
the total exposure of the general population from all sources must be
considered. Those working in industries where lead is used or produced
may be subject to additional exposure compared with the general
population.
In addition, exposure of certain groups within the general
population may vary because of physiological, behavioural or other
factors. For example, the fetus is exposed to lead via maternal
transfer of internal and external doses, nursing infants may be
exposed to lead in breast milk, the young child is exposed more
intensively to dusts and lead on non-food items (such as lead-painted
toys), alcohol consumption and smoking increase lead exposure,
differences in diet may influence lead exposure markedly, and some
people may be exposed to lead through hobby or occupational activities
in addition to their exposure as members of the general population.
5.1 Inhalation route of exposure
5.1.1 Ambient air
Ambient air can be a major pathway of lead distribution in the
environment. Sources of lead in air include combustion products of
lead additives in petrol, and point sources such as smelters,
incinerators, and some industrial processes including the burning of
fossil fuels.
Concentrations of lead in air range from 7.6 × 10-5 µg/m3 in
remote areas such as Antarctica (Maenhaut et al., 1979) to
> 10 µg/m3 near lead smelters (Elias, 1985).
Almost all lead in air is bound to fine particles of less than
1 µm diameter, although some may be solubilized in acid aerosol
droplets. The size of these particles varies with the source and with
the age of the particle from the time of emission (US EPA, 1986a; WHO,
1987). Most lead in air is inorganic lead, and the predominant source
is from the combustion of tetraethyl- and tetramethyllead used as fuel
additives (US EPA, 1986a; WHO, 1987). A summary of lead levels on fine
airborne particles from some cities in the USA and France is given in
Table 9.
Table 9. Concentrations of lead in fine airborne particles from some
cities in the USA and France in 1984-1985a
City Population Mean lead No. of
concentration samples
(µg/m3)
Clemson (USA) 3000 0.33 15
Senonches (France) 3000 0.005 6
Orleans (France) 110 000 0.11 7
Clermont (France) 161 000 0.045 7
Akron (USA) 200 000 0.052 6
Strasbourg (France) 260 000 0.072 7
Norfolk (USA) 270 000 0.031 6
Chicago (USA) 10 000 000 0.064 5
Paris (France) 10 000 000 0.44 7
a From: Delumyea & Kalivretenos (1987)
5.1.1.1 Emissions from motor vehicles
In Europe, where leaded vehicle fuel is still used, airborne
concentrations of lead in urban areas are likely to be in the range of
0.5-3 µg/m3 (WHO, 1987). Concentrations of between 0.6 and
5.7 µg/m3 were reported in Mexico in 1982 (GEMS, 1985). Where leaded
vehicle fuel is no longer used, concentrations are likely to fall to
< 0.2 µg/m3 (Elias, 1985). In 1990, concentrations of lead in air
in urban areas of the USA had fallen to below 0.07 µg/m3 (US EPA,
1991). This decrease in airborne lead levels in the USA is shown in
Fig. 3. Reductions in lead in air have been reported from Canada,
Germany, Norway and the United Kingdom (OECD, 1993).
5.1.1.2 Stationary sources
Where emissions are largely uncontrolled, concentrations of lead
in air around stationary sources such as lead smelters range from over
10 µg/m3 50 m from the smelter to 1.5 µg/m3 one km away (Wang et
al., 1992). Where more stringent emission controls are used,
concentrations of lead are much lower (US EPA, 1991). The Port Pirie
Making several assumptions regarding global atmospheric lead
concentrations, wind speed, surface area and texture, a global
deposition of approximately 410 000 tonnes/year (combined wet and dry)
was calculated by the US EPA (1986a).
4.1.2 Transport to water and soil
When deposited in water, whether from air or through run-off from
soil, lead partitions rapidly between the sediment and aqueous phase,
depending upon the salt content of the water as well as the presence
of organic complexing agents. For example, at pH > 5.4 the total
solubility of lead is about 30 µg/litre in hard water and 500 µg/litre
in soft water (Davies & Everhart 1973). In addition, the presence of
sulfate and carbonate ions can limit lead solubility, as described by
Hem & Durum (1973) in a review of the aqueous chemistry of lead.
Water-borne lead has been found to exist as soluble lead or
undissolved colloidal particles, either suspended in the aqueous phase
or carried as surface coatings on other suspended solids (Lovering,
1976). The ratio of lead in suspended solids to lead in the dissolved
form has been found to vary from 4:1 in rural areas to 27:1 in urban
streams (Getz et al., 1977).
Both natural organic compounds (humic and fulvic acids) as well
as those of anthropogenic origin (e.g., ethylenediamino-tetraacetic
and nitrilotriacetic acids) may complex lead found in surface waters
(Steelink, 1977; Reuter & Perdue, 1977; Neubecker & Allen, 1983). The
presence in water of such chelators can increase the rate of solution
of lead compounds (e.g., lead sulfide) 10 to 60 times over that of
water at the same pH without fulvates (Bondarenko, 1968; Lovering,
1976).
Lead accumulation in soils is primarily a function of the rate of
wet and dry deposition from the atmosphere. Transport within soil and
the bioavailability of lead from soil are dependent upon many factors,
including pH, mineral composition of the soil, and amount and type of
organic material, with most of the lead being bound within the upper
5 cm of soil (Reaves & Berrow 1984; Garcia-Miragaya, 1984). This
limits the amount which can be leached into water or be available for
uptake into plants. It has been shown that only 0.2% of the total lead
in soil can be released into solution by shaking (Dong et al., 1985).
However, the release of lead from organic complexes to the soluble,
and thus bioavailable, form is highly pH dependent. Within the usual
pH range for soils (4 to 6), the organic-lead complexes become more
soluble and the lead more available for plant uptake and leaching into
water (US EPA, 1986a).
4.1.3 Transport to biota
The transfer of air lead to the biota may be direct or indirect
(uptake from water, soil and vegetation). Examples of the accumulation
of lead into aquatic (wet and dry deposition) and terrestrial
organisms are given in Environmental Health Criteria 85: Lead -
Environmental Aspects (IPCS, 1989). Relevant parts of that monograph
are summarized here.
4.1.3.1 Aquatic organisms
In aquatic and aquatic/terrestrial model ecosystems, uptake by
primary producers and consumers seems to be determined by the
bioavailability of the lead. Bioavailability is generally much lower
whenever organic material, sediment or mineral particles (e.g., clay)
are present. In many organisms, it is unclear whether lead is adsorbed
onto the organism or actually taken up. Consumers take up lead from
their contaminated food, often to high concentrations but without
biomagnification.
The uptake and accumulation of lead by aquatic organisms from
water and sediment are influenced by various environmental factors
such as temperature, salinity and pH, as well as humic and alginic
acid content.
In contaminated aquatic systems, almost all of the lead is
tightly bound to sediment. Only a minor fraction is dissolved in the
water, even in the interstitial water.
The lead uptake by fish reaches equilibrium only after a number
of weeks of exposure. Lead is accumulated mostly in gill, liver,
kidney and bone.
Fish eggs show increasing lead levels with increased exposure
concentration, and there are indications that lead is present on the
egg surface but not accumulated in the embryo.
Fish accumulate lead from water as well as sediments; aquatic
uptake is influenced by the presence of cations and the oxygen content
of the water (IPCS, 1989).
4.1.3.2 Terrestrial organisms
In bacteria, the majority of lead is found in the cell wall. A
similar phenomenon is also noted in higher plants. Some lead that
passes into the plant root cell can be combined with new cell wall
material and subsequently removed from the cytoplasm to the cell wall.
Of the lead remaining in the root cell, there is evidence of very
little translocation to other parts of the plant because the
concentration of lead in shoot and leaf tissue is usually much lower
than in root. Foliar uptake of lead occurs, but only to a very limited
extent.
In animals, there is a positive correlation between tissue and
dietary lead concentrations, although tissue concentrations are almost
always lower. The distribution of lead within animals is closely
associated with calcium metabolism.
Lead shot is typically trapped in the gizzard of birds where it
is slowly ground down resulting in the release of lead.
The tetravalent organic form of lead is generally more toxic than
the divalent inorganic form, and its distribution in organisms may not
specifically follow calcium metabolism.
4.2 Environmental transformation
4.2.1 Abiotic transformation
Once released into the environment lead may be transformed from
one inorganic species or particle size to another. However, as an
element it is not subject to degradation. For example, lead-containing
particles in automobile exhaust are usually lead halides or double
salts with ammonium halides (Biggins & Harrison, 1979). Within 24 h,
over 75% of lead particulate matter is transformed to lead carbonates
and sulfates (Olson & Skogerboe, 1975).
4.2.2 Biotransformation
The transformation of inorganic lead to tetramethyllead (TML) has
been observed in aquatic systems, particularly in sediments, and
biomethylation was postulated by Wong et al. (1975) and Schmidt &
Huber (1976). However, no biological methylation of inorganic lead was
noted by Reisinger et al. (1981) in studies under many conditions
using several bacterial species known to alkylate mercury and other
heavy metals. The authors did find chemical methylation in the
presence of methylcobalamin and sulfide or aluminium ions and it was
independent of the presence of bacteria. The evidence for microbial
methylation of various compounds of lead in aquatic systems has been
reviewed by Beijer & Jernelöv (1984). It is still unclear whether the
TML formed is produced abiotically or by biotransformation.
5. ENVIRONMENTAL LEVELS AND HUMAN EXPOSURE
This chapter describes the sources of lead to which people are
exposed and quantifies that exposure. Depending on the source, the
concentration of lead and its bioavailability, the relative
contribution of each source may vary considerably. For example, men
working in ship-breaking, children exposed to deteriorating lead
paint, and people consuming soft water distributed in lead pipes have
frequently been shown to have excessive absorption of lead, leading to
clinically obvious lead poisoning.
In evaluating the exposure of the general population to lead, it
is critical to consider the interrelationships among environmental
pathways for lead and transfers across environmental media. The
general population is exposed to lead simultaneously from many sources
and through multiple pathways as shown in Fig. 2. Thus, while, for
purposes of discussion, exposure via air, water, food, dusts and
soils, and other sources are presented separately in this document,
the total exposure of the general population from all sources must be
considered. Those working in industries where lead is used or produced
may be subject to additional exposure compared with the general
population.
In addition, exposure of certain groups within the general
population may vary because of physiological, behavioural or other
factors. For example, the fetus is exposed to lead via maternal
transfer of internal and external doses, nursing infants may be
exposed to lead in breast milk, the young child is exposed more
intensively to dusts and lead on non-food items (such as lead-painted
toys), alcohol consumption and smoking increase lead exposure,
differences in diet may influence lead exposure markedly, and some
people may be exposed to lead through hobby or occupational activities
in addition to their exposure as members of the general population.
5.1 Inhalation route of exposure
5.1.1 Ambient air
Ambient air can be a major pathway of lead distribution in the
environment. Sources of lead in air include combustion products of
lead additives in petrol, and point sources such as smelters,
incinerators, and some industrial processes including the burning of
fossil fuels.
Concentrations of lead in air range from 7.6 × 10-5 µg/m3 in
remote areas such as Antarctica (Maenhaut et al., 1979) to
> 10 µg/m3 near lead smelters (Elias, 1985).
Almost all lead in air is bound to fine particles of less than
1 µm diameter, although some may be solubilized in acid aerosol
droplets. The size of these particles varies with the source and with
the age of the particle from the time of emission (US EPA, 1986a; WHO,
1987). Most lead in air is inorganic lead, and the predominant source
is from the combustion of tetraethyl- and tetramethyllead used as fuel
additives (US EPA, 1986a; WHO, 1987). A summary of lead levels on fine
airborne particles from some cities in the USA and France is given in
Table 9.
Table 9. Concentrations of lead in fine airborne particles from some
cities in the USA and France in 1984-1985a
City Population Mean lead No. of
concentration samples
(µg/m3)
Clemson (USA) 3000 0.33 15
Senonches (France) 3000 0.005 6
Orleans (France) 110 000 0.11 7
Clermont (France) 161 000 0.045 7
Akron (USA) 200 000 0.052 6
Strasbourg (France) 260 000 0.072 7
Norfolk (USA) 270 000 0.031 6
Chicago (USA) 10 000 000 0.064 5
Paris (France) 10 000 000 0.44 7
a From: Delumyea & Kalivretenos (1987)
5.1.1.1 Emissions from motor vehicles
In Europe, where leaded vehicle fuel is still used, airborne
concentrations of lead in urban areas are likely to be in the range of
0.5-3 µg/m3 (WHO, 1987). Concentrations of between 0.6 and
5.7 µg/m3 were reported in Mexico in 1982 (GEMS, 1985). Where leaded
vehicle fuel is no longer used, concentrations are likely to fall to
< 0.2 µg/m3 (Elias, 1985). In 1990, concentrations of lead in air
in urban areas of the USA had fallen to below 0.07 µg/m3 (US EPA,
1991). This decrease in airborne lead levels in the USA is shown in
Fig. 3. Reductions in lead in air have been reported from Canada,
Germany, Norway and the United Kingdom (OECD, 1993).
5.1.1.2 Stationary sources
Where emissions are largely uncontrolled, concentrations of lead
in air around stationary sources such as lead smelters range from over
10 µg/m3 50 m from the smelter to 1.5 µg/m3 one km away (Wang et
al., 1992). Where more stringent emission controls are used,
concentrations of lead are much lower (US EPA, 1991). The Port Pirie
 smelter in South Australia is estimated to have lost 80 000 tonnes of
lead to the environment in non-stack fugitive emissions from 1889 to
1982. It is also estimated that from 1969 to 1981 this smelter
discharged 40 tonnes of lead per year into the environment. Ambient
air concentrations near the smelter were between 0.5 and 10 µg/m3
(Body et al., 1988).
5.1.2 Indoor air
Davies et al. (1987a) sampled indoor and ambient air lead levels
and found that where there was no interior lead source, such as
lead-painted surfaces, air lead concentrations inside dwellings were
highly correlated with those outside and averaged approximately 60% of
those in the external air immediately outside the house. A similar
ratio was reported in the Arnhem lead study (Diemel et al., 1981).
Indoor air lead levels are affected by the presence of smokers and
lead-painted surfaces.
Levels of airborne lead in indoor shooting ranges have been shown
to range from 2.7 to 90.5 µg/m3 depending upon the location of
sampling, which varied from the showroom to target area (Novotny et
al., 1987).
5.1.3 Air in the working environment
The diversity and extent of the industrial applications of lead
is such that it is impossible to make general statements about
exposure levels. In many instances actual exposure levels have not
been measured and often work is carried out in small enterprises which
may not be subject to workplace controls or legislated requirements.
Airborne lead concentrations in the occupational setting vary
considerably according to the type of industry and the level of
industrial hygiene practised at each plant. Occupations and operations
that may present lead hazards to workers are listed in Table 10.
Recent monitoring data (1980-1985) from Finland are summarized in
Fig. 4 (Jaakkola & Anttila, 1992).
5.1.4 Smoking of tobacco
Lead is present in tobacco. The mean content of lead in
filter-tipped cigarettes produced between 1960 and 1980 was 2.4 µg/g.
Approximately 5% of this lead may be inhaled; the remainder occurs in
the ash and side-stream smoke (Mussalo-Rauhamaa et al., 1986).
smelter in South Australia is estimated to have lost 80 000 tonnes of
lead to the environment in non-stack fugitive emissions from 1889 to
1982. It is also estimated that from 1969 to 1981 this smelter
discharged 40 tonnes of lead per year into the environment. Ambient
air concentrations near the smelter were between 0.5 and 10 µg/m3
(Body et al., 1988).
5.1.2 Indoor air
Davies et al. (1987a) sampled indoor and ambient air lead levels
and found that where there was no interior lead source, such as
lead-painted surfaces, air lead concentrations inside dwellings were
highly correlated with those outside and averaged approximately 60% of
those in the external air immediately outside the house. A similar
ratio was reported in the Arnhem lead study (Diemel et al., 1981).
Indoor air lead levels are affected by the presence of smokers and
lead-painted surfaces.
Levels of airborne lead in indoor shooting ranges have been shown
to range from 2.7 to 90.5 µg/m3 depending upon the location of
sampling, which varied from the showroom to target area (Novotny et
al., 1987).
5.1.3 Air in the working environment
The diversity and extent of the industrial applications of lead
is such that it is impossible to make general statements about
exposure levels. In many instances actual exposure levels have not
been measured and often work is carried out in small enterprises which
may not be subject to workplace controls or legislated requirements.
Airborne lead concentrations in the occupational setting vary
considerably according to the type of industry and the level of
industrial hygiene practised at each plant. Occupations and operations
that may present lead hazards to workers are listed in Table 10.
Recent monitoring data (1980-1985) from Finland are summarized in
Fig. 4 (Jaakkola & Anttila, 1992).
5.1.4 Smoking of tobacco
Lead is present in tobacco. The mean content of lead in
filter-tipped cigarettes produced between 1960 and 1980 was 2.4 µg/g.
Approximately 5% of this lead may be inhaled; the remainder occurs in
the ash and side-stream smoke (Mussalo-Rauhamaa et al., 1986).
 Table 10. Occupations or operations which may present lead hazards for
workersa
Primary and secondary lead smelting Lead mining
Welding and cutting of lead-painted metal Plumbing
constructions
Welding of galvanized or zinc silicate coated sheets Cable making
Shipbreaking Wire patenting
Nonferrous foundries Lead casting
Storage battery manufacture: pasting, assembling, Type founding in
welding of battery connectors printing shops
Production of lead paints Stereotype setting
Spray painting Assembling of cars
Mixing (by hand) of lead stabilizers into Automobile repair
polyvinyl chloride
Mixing (by hand) of crystal glass mass Shot making
Sanding or scraping of lead paint Welding (occasionally)
Burning of lead in enamelling workshops Lead glass blowing
Repair of automobile radiators Pottery/glass making
a Adapted from: Hernberg (1973)
5.2 Exposure by ingestion
5.2.1 Water
Exposure of humans to lead from water has been underestimated in
studies of total exposure. Due to the practice of sampling water
systems at points before entry into the distribution piping and
domestic plumbing (US EPA, 1986a; Dabeka et al., 1987), it had been
widely assumed that exposure to lead in drinking-water was not
significant.
Background or natural levels of lead in surface and ground water
are generally low. However, water with low pH and only low
concentrations of dissolved salts (referred to as aggressive) can
leach substantial quantities of lead from pipes, solder and fixtures.
Lead-lined reservoirs, cisterns and holding tanks for water (Mushak &
Crocetti, 1989) can be a major source of lead contamination of
drinking-water. For example, Wiebe et al. (1991) reported the analysis
of over 2000 water samples in Hawaii, USA, following increased
volcanic activity that resulted in the release of acid aerosols. The
lead concentration of drinking-water collected in catchment systems
ranged from < 20 to 7000 µg/litre. Sampling programmes conducted at
the tap in the USA during 1985-1988 revealed widespread elevation of
lead in drinking-water, often above the WHO guidance value of
50 µg/litre (WHO, 1984), which has now been revised to 10 µg/litre
(WHO, 1993).
The combination of acid or aggressive water and lead in plumbing
results in very high concentrations of lead in drinking-water,
particularly after it has been standing for several hours (Worth et
al., 1981; Sherlock et al., 1982; Kaminsky et al., 1988).
Surveys in Canada and the USA showed that drinking-water supplies
leaving treatment plants contain 2-3 µg lead/litre (US EPA, 1986a;
Dabeka et al., 1987). In the case of plumbosolvent water, up to 40% of
household samples may exceed 100 µg lead/litre. This has been observed
in Scotland (Sherlock et al., 1986), and reflects the contribution of
plumbing and plumbing fixtures to lead levels of drinking-water.
5.2.2 Food and alcoholic beverages
5.2.2.1 Food
The major source of lead for non-occupationally exposed adults is
food and drink. The proportion of total intake derived from food is
dependent on the concentration of lead in air, water and other
sources. Detailed data are available from several countries, including
Australia (NFA, 1991), USA (Bolger et al., 1991), Sweden (Vahter et
al., 1990) and Canada (Dabeka et al., 1987). Foods have been surveyed
from several other industrialized and developing countries
(Galal-Gorchev, 1991b). Children are exposed to additional lead from
dust and soil, and so lead from foods and beverages may not be the
predominant sources of lead for all age groups.
Lead is present in soils and is transferred to food crops growing
on the soil. Roots usually contain more lead than stems and leaves,
while seeds and fruits have the lowest concentrations.
Particulate lead present in air may adhere tenaciously to leafy
vegetables. Leaves collected in or very near urban areas have been
shown to contain substantially elevated concentrations of lead.
Quantities of lead ingested from the diet vary widely from country to
country.
Data on the lead levels of specific foodstuffs or groups of food
materials, from which one can estimate a daily dietary lead intake,
are available from several countries. In a few studies, foodstuffs
specific to infants and children have also been analysed (Kolbye et
al., 1974; Dabeka & McKenzie, 1987; Vahter et al., 1990; Bolger et
al., 1991; Albert & Badillo, 1991). Data are available for canned
foods typically consumed by young children (Capar & Rigsby, 1989). The
utilization of such data in the calculation of total intake of lead
from food is discussed in section 5.2.2.2.
An overview of the foods contributing to the dietary lead levels
in Australia is shown in Fig. 5. Similar data from other countries are
shown in Table 11 (Galal-Gorchev, 1991b). A summary of the data on
lead levels in foodstuffs from the USA is given in Table 12 and from
Canada in Table 13. More recent data from the USA have shown that
there has been a substantial reduction of lead levels in food consumed
by all age groups during the past two decades (Bolger et al., 1991)
(Fig. 6). A similar decrease in lead intake has been found in the
United Kingdom (OECD, 1993). These data should be considered as
representative of specific areas and such values can be expected to
vary elsewhere according to local agricultural and food-processing
practices, particularly in areas where lead-soldered cans are still
used. Support for this is indicated by a comparison on the one hand of
the decreased use of lead-soldered food and beverage cans in the USA
(Fig. 7) and the increased use of cans produced by alternative
technology, and on the other hand the marked decrease in food-borne
lead shown in Fig. 6. Food may represent a pathway for human lead
exposure from other media such as air and water. The use of leaded
gasoline or the proximity of industries that may produce ambient
emissions of lead can greatly influence dietary lead intake. Therefore
further caution is required when extrapolating between countries with
regards to levels of food-borne lead.
Representative levels of lead in foodstuffs from some 20
countries are given in Table 14 (Galal-Gorchev, 1991a). These results
from the GEMS/FOOD data can be compared with the levels of lead in
specific foodstuffs in the USA and Canada (Tables 12 and 13).
Table 10. Occupations or operations which may present lead hazards for
workersa
Primary and secondary lead smelting Lead mining
Welding and cutting of lead-painted metal Plumbing
constructions
Welding of galvanized or zinc silicate coated sheets Cable making
Shipbreaking Wire patenting
Nonferrous foundries Lead casting
Storage battery manufacture: pasting, assembling, Type founding in
welding of battery connectors printing shops
Production of lead paints Stereotype setting
Spray painting Assembling of cars
Mixing (by hand) of lead stabilizers into Automobile repair
polyvinyl chloride
Mixing (by hand) of crystal glass mass Shot making
Sanding or scraping of lead paint Welding (occasionally)
Burning of lead in enamelling workshops Lead glass blowing
Repair of automobile radiators Pottery/glass making
a Adapted from: Hernberg (1973)
5.2 Exposure by ingestion
5.2.1 Water
Exposure of humans to lead from water has been underestimated in
studies of total exposure. Due to the practice of sampling water
systems at points before entry into the distribution piping and
domestic plumbing (US EPA, 1986a; Dabeka et al., 1987), it had been
widely assumed that exposure to lead in drinking-water was not
significant.
Background or natural levels of lead in surface and ground water
are generally low. However, water with low pH and only low
concentrations of dissolved salts (referred to as aggressive) can
leach substantial quantities of lead from pipes, solder and fixtures.
Lead-lined reservoirs, cisterns and holding tanks for water (Mushak &
Crocetti, 1989) can be a major source of lead contamination of
drinking-water. For example, Wiebe et al. (1991) reported the analysis
of over 2000 water samples in Hawaii, USA, following increased
volcanic activity that resulted in the release of acid aerosols. The
lead concentration of drinking-water collected in catchment systems
ranged from < 20 to 7000 µg/litre. Sampling programmes conducted at
the tap in the USA during 1985-1988 revealed widespread elevation of
lead in drinking-water, often above the WHO guidance value of
50 µg/litre (WHO, 1984), which has now been revised to 10 µg/litre
(WHO, 1993).
The combination of acid or aggressive water and lead in plumbing
results in very high concentrations of lead in drinking-water,
particularly after it has been standing for several hours (Worth et
al., 1981; Sherlock et al., 1982; Kaminsky et al., 1988).
Surveys in Canada and the USA showed that drinking-water supplies
leaving treatment plants contain 2-3 µg lead/litre (US EPA, 1986a;
Dabeka et al., 1987). In the case of plumbosolvent water, up to 40% of
household samples may exceed 100 µg lead/litre. This has been observed
in Scotland (Sherlock et al., 1986), and reflects the contribution of
plumbing and plumbing fixtures to lead levels of drinking-water.
5.2.2 Food and alcoholic beverages
5.2.2.1 Food
The major source of lead for non-occupationally exposed adults is
food and drink. The proportion of total intake derived from food is
dependent on the concentration of lead in air, water and other
sources. Detailed data are available from several countries, including
Australia (NFA, 1991), USA (Bolger et al., 1991), Sweden (Vahter et
al., 1990) and Canada (Dabeka et al., 1987). Foods have been surveyed
from several other industrialized and developing countries
(Galal-Gorchev, 1991b). Children are exposed to additional lead from
dust and soil, and so lead from foods and beverages may not be the
predominant sources of lead for all age groups.
Lead is present in soils and is transferred to food crops growing
on the soil. Roots usually contain more lead than stems and leaves,
while seeds and fruits have the lowest concentrations.
Particulate lead present in air may adhere tenaciously to leafy
vegetables. Leaves collected in or very near urban areas have been
shown to contain substantially elevated concentrations of lead.
Quantities of lead ingested from the diet vary widely from country to
country.
Data on the lead levels of specific foodstuffs or groups of food
materials, from which one can estimate a daily dietary lead intake,
are available from several countries. In a few studies, foodstuffs
specific to infants and children have also been analysed (Kolbye et
al., 1974; Dabeka & McKenzie, 1987; Vahter et al., 1990; Bolger et
al., 1991; Albert & Badillo, 1991). Data are available for canned
foods typically consumed by young children (Capar & Rigsby, 1989). The
utilization of such data in the calculation of total intake of lead
from food is discussed in section 5.2.2.2.
An overview of the foods contributing to the dietary lead levels
in Australia is shown in Fig. 5. Similar data from other countries are
shown in Table 11 (Galal-Gorchev, 1991b). A summary of the data on
lead levels in foodstuffs from the USA is given in Table 12 and from
Canada in Table 13. More recent data from the USA have shown that
there has been a substantial reduction of lead levels in food consumed
by all age groups during the past two decades (Bolger et al., 1991)
(Fig. 6). A similar decrease in lead intake has been found in the
United Kingdom (OECD, 1993). These data should be considered as
representative of specific areas and such values can be expected to
vary elsewhere according to local agricultural and food-processing
practices, particularly in areas where lead-soldered cans are still
used. Support for this is indicated by a comparison on the one hand of
the decreased use of lead-soldered food and beverage cans in the USA
(Fig. 7) and the increased use of cans produced by alternative
technology, and on the other hand the marked decrease in food-borne
lead shown in Fig. 6. Food may represent a pathway for human lead
exposure from other media such as air and water. The use of leaded
gasoline or the proximity of industries that may produce ambient
emissions of lead can greatly influence dietary lead intake. Therefore
further caution is required when extrapolating between countries with
regards to levels of food-borne lead.
Representative levels of lead in foodstuffs from some 20
countries are given in Table 14 (Galal-Gorchev, 1991a). These results
from the GEMS/FOOD data can be compared with the levels of lead in
specific foodstuffs in the USA and Canada (Tables 12 and 13).


 Table 11. Foods contributing to dietary lead levels in Canada,
Finland, Netherlands and the United Kingdoma
Country Food Percentage of total intake
Canada vegetables 17
meat/fish/poultry 17
beverages 15
cereals and products 15
fruits and juices 10
Finland cereals and products 24
fruits 22
beverages, sweets, etc. 20
milk and products 17
vegetables 9
Netherlands drinking-water 30
cereals and products 17
vegetables 12
wines and spirits 9
fruits 6
United Kingdom bread and cereals 15
beverages 14
potatoes 10
milk 9
canned vegetables 8
a From: Galal-Gorchev (1991b)
Table 12. Concentrations of lead in various foods in the USAa
Food group Concentration of leadb (µg/g)
Dairy products 0.003-0.083
Meat, fish and poultry 0.002-0.159
Grain and cereal products 0.002-0.136
Vegetables 0.005-0.649
Fruit and fruit juices 0.005-0.223
Oils, fats and shortenings 0.006-0.073
Sugar and adjuncts 0.006-0.073
Beverages 0.002-0.041
a From: US EPA (1986a)
b Range of concentrations shown are the lowest and highest mean
values for items within the food group and listed in Appendix 7-D
of US EPA (1986a).
Table 13. Levels of lead in various Canadian food categoriesa
Description of food category Median concentration
of lead (µg/kg)b
I Cereals (as prepared with milk, sugar, etc.), 32.4 (11.5-78.3)
bread and toast
II Water consumed directly 2.0 (0.25-71.2)
III Coffee, tea, beer, liquor, sodas, etc. (as prepared) 8.8 (< 0.05-28.9)
IV Fruit juices, fruits (canned and fresh) 7.9 (1.5-109)
V Dairy products and eggs 3.3 (1.21-81.9)
VI Starch vegetables, e.g. potatoes, rice 16.9 (5.5-83.7)
VII Other vegetables, vegetable juices and soups 31.7 (0.62-254)
VIII Meat, fish, poultry, meat-based soups 31.3 (11-121)
IX Miscellaneous (pies, puddings, nuts, snack foods) 33.1 (13.6-1381)
X Cheese (other than cottage cheese) 33.8 (27.7-6775)
a From: Dabeka et al. (1987)
b Values in parentheses are the ranges.
The lead levels in infant foods in Canada, Mexico and USA are
shown in Table 15. In 1987, Dabeka et al. (1987) found the intake of
lead by infants fed evaporated milk stored in lead-soldered cans
exceeded the Provisional Tolerable Weekly Intake of 25 µg lead/kg body
weight, set in 1993 (FAO/WHO, 1993). These values do not include lead
in water used to prepare formulae. It has been reported that infants
fed formulae prepared with water containing high levels of lead
(> 100 µg/litre) have lead intakes exceeding 25 µg/kg body weight per
week (Galal-Gorchev, 1991b).
5.2.2.2 Total intake from food
Guidelines for the determination of dietary intake of chemical
contaminants have been published (WHO, 1985). Three basic approaches
were described, namely: (i) total diet (i.e. market or shopping
basket) studies; (ii) selective studies of individual foodstuffs, and
(iii) duplicate portion (duplicate diet) studies. It is essential to
have food consumption data for the first two methods in order to
estimate a total intake. For all methods, well-designed quality
assurance and quality control programmes are essential and these have
been described by Vahter & Slorach (1990) and Vahter et al. (1991a).
Table 14. Representative levels of lead in foods from GEMS/FOOD
dataa
Commodity Typical lead levels (µg/kg)
Cereals 60
Roots and tubers 50
Fruit 50
Vegetables 50
Meat 50
Vegetable oils and fats 20
Fish 100
Pulses 40
Eggs 20
Nuts and oilseeds 40
Shellfish 20
Offal 20
Spices and herbs 30
Other foods not assessed
Drinking-water 20
Canned beverages 200
Canned foodb 200
a From: Galal-Gorchev (1991a)
b It is assumed that canned food consumption is 2% of total.
Given the differences between countries with respect to dietary
composition, the amount of specific foodstuffs consumed, the
processing technologies employed, whether consumption of water and
alcoholic beverages are included in the estimates of dietary lead, and
the number of samples taken, caution must be exercised in making
comparisons between countries.
During the late 1970s and 1980s the quantity of lead ingested as
part of diet decreased markedly in many countries. For adults not
occupationally exposed to lead, the diet remains the largest
contributor to lead intake. However, the quantities ingested are far
lower than in previous decades. In the USA, typical levels of intake
declined as shown in Fig. 6.
Table 15. Lead levels (µg/kg food) in cow's milk and infant formula
Product Canada Mexico USA
median (range)a averageb averagec
Fluid milk 1.19 (0.01-2.5) 5
Evaporated milk (canned) 71.9 (27-106) 88 10
(cardboard) -- 9
Infant formula
Ready to use lead-solder can 30.1 (1.1-122) 13 10
Ready to use lead-free can 1.6 (1.5-2) 1
Formula powder (1985) 96.6 (3.7-19)
Powdered milkd -- 21
a From: Dabeka & McKenzie (1987)
b From: Albert & Badillo (1991). Data were obtained in 1982.
c From: Bolger et al. (1991). Data were obtained in the late 1980s.
d The concentration of lead in milk consumed by the infant will be
highly dependent on the concentration of lead in water used to
dilute the powdered milk.
An overall summary of the GEMS/FOOD data for adults is given in
Fig. 8 (Galal-Gorchev, 1991a). The trends for lead intake for the
period 1980-1988 in the USA, Japan, Hungary and the United Kingdom are
shown in Fig. 9 (Galal-Gorchev, 1991b). Data for intake of lead by
infants and children in eight countries are shown in Fig. 10
(Galal-Gorchev, 1991a). Additional data are available from other
countries. Although collection methods vary, these data illustrate the
wide variations in ingestion of lead from food. Brunekreef (1986)
noted that the market basket studies tended to overestimate lead
intake when compared with duplicate diet analysis. Examples cited
included reports from the United Kingdom by Fouassin & Fondu (1980),
Buchet et al. (1983) and Sherlock et al. (1982), where market basket
surveys overestimated this intake by 2- to 3-fold.
Other studies from various countries on total lead intakes by
children and adults are summarized in Table 16. Lead contaminated
water has been shown to be a contributor to food-borne lead where
large volumes of water are used in food preparation and cooking, e.g.,
in foods prepared in boiling water.
Table 11. Foods contributing to dietary lead levels in Canada,
Finland, Netherlands and the United Kingdoma
Country Food Percentage of total intake
Canada vegetables 17
meat/fish/poultry 17
beverages 15
cereals and products 15
fruits and juices 10
Finland cereals and products 24
fruits 22
beverages, sweets, etc. 20
milk and products 17
vegetables 9
Netherlands drinking-water 30
cereals and products 17
vegetables 12
wines and spirits 9
fruits 6
United Kingdom bread and cereals 15
beverages 14
potatoes 10
milk 9
canned vegetables 8
a From: Galal-Gorchev (1991b)
Table 12. Concentrations of lead in various foods in the USAa
Food group Concentration of leadb (µg/g)
Dairy products 0.003-0.083
Meat, fish and poultry 0.002-0.159
Grain and cereal products 0.002-0.136
Vegetables 0.005-0.649
Fruit and fruit juices 0.005-0.223
Oils, fats and shortenings 0.006-0.073
Sugar and adjuncts 0.006-0.073
Beverages 0.002-0.041
a From: US EPA (1986a)
b Range of concentrations shown are the lowest and highest mean
values for items within the food group and listed in Appendix 7-D
of US EPA (1986a).
Table 13. Levels of lead in various Canadian food categoriesa
Description of food category Median concentration
of lead (µg/kg)b
I Cereals (as prepared with milk, sugar, etc.), 32.4 (11.5-78.3)
bread and toast
II Water consumed directly 2.0 (0.25-71.2)
III Coffee, tea, beer, liquor, sodas, etc. (as prepared) 8.8 (< 0.05-28.9)
IV Fruit juices, fruits (canned and fresh) 7.9 (1.5-109)
V Dairy products and eggs 3.3 (1.21-81.9)
VI Starch vegetables, e.g. potatoes, rice 16.9 (5.5-83.7)
VII Other vegetables, vegetable juices and soups 31.7 (0.62-254)
VIII Meat, fish, poultry, meat-based soups 31.3 (11-121)
IX Miscellaneous (pies, puddings, nuts, snack foods) 33.1 (13.6-1381)
X Cheese (other than cottage cheese) 33.8 (27.7-6775)
a From: Dabeka et al. (1987)
b Values in parentheses are the ranges.
The lead levels in infant foods in Canada, Mexico and USA are
shown in Table 15. In 1987, Dabeka et al. (1987) found the intake of
lead by infants fed evaporated milk stored in lead-soldered cans
exceeded the Provisional Tolerable Weekly Intake of 25 µg lead/kg body
weight, set in 1993 (FAO/WHO, 1993). These values do not include lead
in water used to prepare formulae. It has been reported that infants
fed formulae prepared with water containing high levels of lead
(> 100 µg/litre) have lead intakes exceeding 25 µg/kg body weight per
week (Galal-Gorchev, 1991b).
5.2.2.2 Total intake from food
Guidelines for the determination of dietary intake of chemical
contaminants have been published (WHO, 1985). Three basic approaches
were described, namely: (i) total diet (i.e. market or shopping
basket) studies; (ii) selective studies of individual foodstuffs, and
(iii) duplicate portion (duplicate diet) studies. It is essential to
have food consumption data for the first two methods in order to
estimate a total intake. For all methods, well-designed quality
assurance and quality control programmes are essential and these have
been described by Vahter & Slorach (1990) and Vahter et al. (1991a).
Table 14. Representative levels of lead in foods from GEMS/FOOD
dataa
Commodity Typical lead levels (µg/kg)
Cereals 60
Roots and tubers 50
Fruit 50
Vegetables 50
Meat 50
Vegetable oils and fats 20
Fish 100
Pulses 40
Eggs 20
Nuts and oilseeds 40
Shellfish 20
Offal 20
Spices and herbs 30
Other foods not assessed
Drinking-water 20
Canned beverages 200
Canned foodb 200
a From: Galal-Gorchev (1991a)
b It is assumed that canned food consumption is 2% of total.
Given the differences between countries with respect to dietary
composition, the amount of specific foodstuffs consumed, the
processing technologies employed, whether consumption of water and
alcoholic beverages are included in the estimates of dietary lead, and
the number of samples taken, caution must be exercised in making
comparisons between countries.
During the late 1970s and 1980s the quantity of lead ingested as
part of diet decreased markedly in many countries. For adults not
occupationally exposed to lead, the diet remains the largest
contributor to lead intake. However, the quantities ingested are far
lower than in previous decades. In the USA, typical levels of intake
declined as shown in Fig. 6.
Table 15. Lead levels (µg/kg food) in cow's milk and infant formula
Product Canada Mexico USA
median (range)a averageb averagec
Fluid milk 1.19 (0.01-2.5) 5
Evaporated milk (canned) 71.9 (27-106) 88 10
(cardboard) -- 9
Infant formula
Ready to use lead-solder can 30.1 (1.1-122) 13 10
Ready to use lead-free can 1.6 (1.5-2) 1
Formula powder (1985) 96.6 (3.7-19)
Powdered milkd -- 21
a From: Dabeka & McKenzie (1987)
b From: Albert & Badillo (1991). Data were obtained in 1982.
c From: Bolger et al. (1991). Data were obtained in the late 1980s.
d The concentration of lead in milk consumed by the infant will be
highly dependent on the concentration of lead in water used to
dilute the powdered milk.
An overall summary of the GEMS/FOOD data for adults is given in
Fig. 8 (Galal-Gorchev, 1991a). The trends for lead intake for the
period 1980-1988 in the USA, Japan, Hungary and the United Kingdom are
shown in Fig. 9 (Galal-Gorchev, 1991b). Data for intake of lead by
infants and children in eight countries are shown in Fig. 10
(Galal-Gorchev, 1991a). Additional data are available from other
countries. Although collection methods vary, these data illustrate the
wide variations in ingestion of lead from food. Brunekreef (1986)
noted that the market basket studies tended to overestimate lead
intake when compared with duplicate diet analysis. Examples cited
included reports from the United Kingdom by Fouassin & Fondu (1980),
Buchet et al. (1983) and Sherlock et al. (1982), where market basket
surveys overestimated this intake by 2- to 3-fold.
Other studies from various countries on total lead intakes by
children and adults are summarized in Table 16. Lead contaminated
water has been shown to be a contributor to food-borne lead where
large volumes of water are used in food preparation and cooking, e.g.,
in foods prepared in boiling water.


 Table 16. Daily lead intake via food in adults and children
Population studied Daily intake (µg/day)a Reference
Adults, United Kingdom 110 M Brunekreef (1986)
Adults, United Kingdom 71 D Brunekreef (1986)
Adults, Belgiumb 282 M Fouassin & Fondu (1980)
Adults, Belgiumb 96 D Buchet et al. (1983)
Adults, Sweden 27 M Slorach et al. (1983)
Adults, Finland 66 M Varo & Kovistoinen (1983)
Adults, Canada 43 D Dabeka et al. (1987)
Adults, USA 82 M Gartrell et al. (1985a)
Adults (female), Japan 31 D Vahter et al. (1991b)
Adults, Germany 61 Kampe (1983)
Adults (female), Croatia 15 D Vahter et al. (1991b)
Adults, Italy 140 IAEA (1987)
Adults (female), China 46 D Vahter et al. (1991b)
Adults, Turkey 70 IAEA (1987)
Adults (female), Sweden 26 D Vahter et al. (1991b)
Children, Poland
0-1 year 225 Olejnik et al. (1985)
1-3 years 259
7-18 years 316
Adults, New Zealand 316 M Pickston et al. (1985)
Children (infant), UK 2-3 breast milk Kovar et al. (1984)
Children (< 1 year), USA 16-17 infant formula Ryu et al. (1983)
& milk
Children, USA
6 months 33.5 M Gartrell et al. (1985b)
2 years 43.4
a M = Market basket survey; D = Duplicate diet study
b Populations studied from the same region.
The relative intake of lead from various sources in 1986 and 1990
in 2-year-old infants and women of child-bearing age in the USA is
shown in Fig. 11. These data illustrate the marked decrease in lead
intake from food over a 4-year period in which there were marked
reductions in the use of lead-soldered cans and lead-containing petrol
additives in the USA (Bolger et al., 1991). Similar decreases in other
countries would no doubt occur after similar actions by public health
officials.
Table 16. Daily lead intake via food in adults and children
Population studied Daily intake (µg/day)a Reference
Adults, United Kingdom 110 M Brunekreef (1986)
Adults, United Kingdom 71 D Brunekreef (1986)
Adults, Belgiumb 282 M Fouassin & Fondu (1980)
Adults, Belgiumb 96 D Buchet et al. (1983)
Adults, Sweden 27 M Slorach et al. (1983)
Adults, Finland 66 M Varo & Kovistoinen (1983)
Adults, Canada 43 D Dabeka et al. (1987)
Adults, USA 82 M Gartrell et al. (1985a)
Adults (female), Japan 31 D Vahter et al. (1991b)
Adults, Germany 61 Kampe (1983)
Adults (female), Croatia 15 D Vahter et al. (1991b)
Adults, Italy 140 IAEA (1987)
Adults (female), China 46 D Vahter et al. (1991b)
Adults, Turkey 70 IAEA (1987)
Adults (female), Sweden 26 D Vahter et al. (1991b)
Children, Poland
0-1 year 225 Olejnik et al. (1985)
1-3 years 259
7-18 years 316
Adults, New Zealand 316 M Pickston et al. (1985)
Children (infant), UK 2-3 breast milk Kovar et al. (1984)
Children (< 1 year), USA 16-17 infant formula Ryu et al. (1983)
& milk
Children, USA
6 months 33.5 M Gartrell et al. (1985b)
2 years 43.4
a M = Market basket survey; D = Duplicate diet study
b Populations studied from the same region.
The relative intake of lead from various sources in 1986 and 1990
in 2-year-old infants and women of child-bearing age in the USA is
shown in Fig. 11. These data illustrate the marked decrease in lead
intake from food over a 4-year period in which there were marked
reductions in the use of lead-soldered cans and lead-containing petrol
additives in the USA (Bolger et al., 1991). Similar decreases in other
countries would no doubt occur after similar actions by public health
officials.
 5.2.2.3 Alcoholic beverages
Contamination of alcoholic beverages with lead may occur in
several way. For example, lead solder used to repair casks or keg and
tap lines from lead capsules used as seals or from residues of lead
arsenate pesticides in soils now used to grow grapes. Alcoholic
beverages tend to be acidic and there is the possibility that large
amounts of lead can be dissolved during preparation, storage or
serving (Wai et al., 1979). Published reports on lead levels in wine
show that important variations occur from sample to sample (Jorhem et
al., 1988). The US Department of the Treasury (1991) analysed 432
table wines sold within the USA. The results are summarized in Table
17.
Sherlock et al. (1986) found that the majority of canned and
bottled beer (90 and 86% respectively) contained less than 10 µg
lead/litre. Draught beers typically contained higher lead
concentrations with 55% having lead concentrations greater than
10 µg/litre, 16% with concentrations over 20 µg/litre, and 4% with
concentrations over 100 µg/litre. The higher lead concentrations in
draught beers are considered most likely due to the draught-dispensing
equipment which sometimes contains brass or gunmetal, both of which
contain low but significant amounts of lead (Sherlock et al., 1986).
In general, alcoholic beverages do not appear to be a significant
source of lead intake for the average person.
Table 17. Distribution of lead in table wines in USAa
Range Number of Percentage of total
(µg/litre) samples samples analysedb
0-10 36 8.3
11-25 62 14.4
26-50 105 24.3
51-100 144 33.3
101-250 64 14.8
251-500 12 2.8
501-673 9 2.1
a From: US Department of the Treasury (1991)
b In all, 432 samples were analysed.
5.2.3 Dust and surface soils
5.2.3.1 Dust
Dust is a significant source of exposure to lead, particularly
for young children (see Fig. 11), as has been demonstrated in several
studies correlating children's blood lead concentrations with dust
lead levels (Rabinowitz et al., 1985; Bornschein et al., 1987; Davies
et al., 1987a; Laxen et al., 1987; Steenhout, 1987).
The major contributions to lead levels in soil and outdoor dust
are from the combustion of fossil fuels (principally leaded petrol),
stationary sources such as smelters, and peeling and flaking of
lead-based paint. Typical lead levels in road dust in the USA are
800-1300 mg/kg in rural areas to 100-5000 mg/kg in urban areas (US
EPA, 1989c).
Concentrations of lead in household dust vary greatly between
different dwellings and areas of the world. Mean concentrations of
300-2500 mg/kg have been found in the United Kingdom and USA, but
individual samples may be in the range of 10 000 to 30 000 mg/kg (Que
Hee et al., 1985b; Clark et al., 1985; Bornschein et al., 1986; Raab
et al., 1987).
Flaking lead-based paint, paint chips, and weathered powdered
paint markedly increase intake of lead from surface dust, particularly
for urban children with pica (US EPA, 1986a; Bornschein et al., 1986).
Lead-based paint chips have been found to contain 1000-5000 µg
lead/cm2 (Billick & Gray, 1978). When lead-based paint is present,
interior renovation activities greatly increase household dust lead
concentrations (Laxen et al., 1987). Improved control of dust and
surface clean-up after lead-based paint removal have been shown to
reduce lead exposure of children reoccupying affected houses (Charney
et al., 1983).
5.2.3.2 Soil
In rural and remote areas, lead in soil is derived mainly from
natural geological sources. These natural sources account for 1-30 mg
lead/kg, but where soils are derived from leadmineralized rocks,
natural concentrations may range from several hundred to several
thousand mg/kg.
Typical values for lead in rural soils in the United Kingdom are
15-106 mg/kg with a geometric mean of 42 mg/kg (Davies, 1983). A
geometric mean of 48 mg/kg for 2780 samples has also been reported
(McGrath, 1986).
Concentrations of lead in urban soil vary greatly. In the USA, a
study of city parks recorded concentrations of 200 to 3300 mg/kg (US
EPA, 1989). Concentrations of up to 10 960 mg/kg have been reported
for urban garden soils in the USA (Mielke et al., 1984), and up to 14
100 mg/kg in the United Kingdom (Culbard et al., 1988). Concentrations
can exceed 20 000 mg/kg around lead mining and processing operations
(Culbard et al., 1988). In areas where lead-based paint has been used,
soil samples taken near building foundations have been reported to be
as high as 20 000 mg/kg (Schmitt et al., 1988; Krueger & Duguay,
1989).
In general, lead concentrations in soils near roads are high
where road traffic density is high. Concentrations decrease
exponentially with distance from the road (IPCS, 1989).
Continuous application of sewage sludge results in an
accumulation of lead in soil. For example, soil receiving heavy
applications over a long period was found to contain 425 mg/kg,
compared with 47 mg/kg in an untreated soil (Beckett et al., 1979).
5.2.3.3 Migration of lead from food containers
The available data on the daily intake of lead by adults and
children indicate a general decrease in those areas where the level of
lead in petrol has decreased and a concerted effort made to avoid
lead-soldered cans for food storage (OECD, 1993). However, in many
regions of the world, lead can migrate from food storage and serving
vessels such as lead-soldered cans (see section 5.2.2.2), ceramic
dishes, pottery vessels, crystal glassware and decals on food wrap
and/or dishes. Acidic foods tend to leach more lead. However, certain
foods such as corn and beans are associated with greater release of
lead than would be predicted from their acidity alone (Bolger et al.,
1991). Oxygen appears to accelerate the release of lead from food
containers.
If foods are stored in ceramic or pottery dinnerware that was
lead-glazed and fired in a low temperature kiln, lead can migrate from
the pottery glaze into the food. The glazing process uses a flux,
which is a material that, at high temperatures, reacts with and helps
dissolve the components of the glaze. Lead oxide is a commonly used
flux. Factors that determine whether and to what extent lead will
migrate include the temperature and extent of firing of the pottery in
the manufacturing process, temperature and duration of food storage,
and the acidity of the food. It is extremely difficult to quantify the
extent of such exposures in view of the variations in the
manufacturing processes and the quality control practised in the
country of origin. However, the extent of exposure can be quite
significant, particularly among infants. Cases of lead intoxication
from this source have been reported (Wallace et al., 1985).
Lead has been found to migrate from lead crystal glass into
beverages. This problem is especially severe if beverages are stored
in lead-crystal containers, e.g., decanters or liquor bottles (de
Leacy, 1987; Graziano & Blum, 1991). This phenomenon was not observed
with borosilicate glass containers (de Leacy, 1987).
Several studies have been made of lead contamination of foods and
beverages from lead used in the manufacture or repair of metal
vessels. Recoating the inner surface of brass utensils with a mixture
of lead and tin, described as "tinning", is widely practised by
artisans in India (Vatsala & Ramakrishna, 1985). The tin-lead alloy
contains 55 to 70% lead. Water containing tamarind contained
400-500 µg lead/litre after 5 min of boiling. The practice is
considered to be widely prevalent in at least the three southern
states of India. Zhu (1984) described 344 cases of chronic lead
poisoning in Jiansu Province, China, involving people who had drunk
rain water boiled in tin kettles. Analysis of the lead content of
water showed that after boiling the water contained 0.79 to
5.34 mg lead/litre.
5.3 Miscellaneous exposure
5.3.1 Cosmetics and medicines
Some traditional medicines and customs have been found to result
in exposure to high levels of lead, most of which cannot be quantified
with any degree of accuracy. Rather than occurring as trace
ingredients or trace contaminants, various lead compounds are used as
major ingredients in traditional medicines in numerous parts of the
world (Table 18). Clinically overt lead poisoning due to traditional
cosmetics and medicines has been identified among infants (Shaltout et
al., 1981; Fernando et al., 1981; Sharma et al., 1990), children and
adults (Pontifax & Garg, 1985; Cueto et al., 1989; Mitchell-Heggs et
al., 1990; Gupta et al., 1990). There are case reports of lead
toxicity secondary to inhalation of lead from traditional remedies
(Aslam et al., 1979; Shaltout, 1981; Cueto et al., 1989; Sharma et
al., 1990; Mitchell-Heggs et al., 1990).
Often the use is not limited to adults; these may be used on
infants and young children, as well as on women. In Kuwait, the leaded
"kohl", also called "Al kohl", is traditionally applied to the raw
umbilical stump of the newborn in the erroneous belief of a beneficial
astringent action (Fernando et al., 1981). An additional use of lead
metal and lead sulfide is for inhalation of the fumes ("Bokhoor")
produced from heating on hot coals, in the mistaken belief that this
will calm irritable infants and children (Fernando et al., 1981;
Shaltout et al., 1981).
Latin-American countries also report the use of traditional
medicines with high lead concentrations. For example, the Mexican
traditional remedy "azarcon" (lead chromate) and/or "greta" (mixed
lead oxides), distributed as finely ground powders, may contain more
than 70% lead. They are used in the treatment of "empacho", a
gastrointestinal disorder considered to be due to a blockage of the
intestine (Trotter, 1990).
In addition to the potential risks of lead exposure from the use
of traditional medicines, clinical lead poisoning can result from the
lodging of lead shot in vivo (Manton & Thal, 1986).
Table 18. Sources of lead exposure in traditional medicines and cosmetics
Source of lead Comments Reference
(product)
Summa/Kohl used in Indo-Pakistan and other Muslim Aslam et al. (1979);
cultures as eyes preparation; placed on Fernando et al. (1981);
conjunctival surface or as astringent on Shaltout et al. (1981);
umbilical cord stump. Antimony originally Sharma et al. (1990)
used but lead cheaper.
Hindu folk ground seeds and roots as treatment Pontifax & Garg (1985)
medicine for diabetes (8 mg lead/g)
Bokhoor tribal custom to produce lead fumes to Shaltout et al. (1981)
ward off evil
Azarcon lead chromate and mixed lead oxides as Trotter (1990)
treatment for gastrointestinal disorders
in Mexico and southwestern USA
Skin ointments cosmetics used by Chinese actors; skin Lai (1977)
and cosmetics ointment in Europe
5.4 General population exposure
The total intake of lead by adults and children in the general
population varies greatly as to the relative contributions from
individual sources (air, water, food, soil/dust and others) and is
partly dependent on life-style and socioeconomic status. It is beyond
the scope of this review to provide comprehensive information covering
a wide range of circumstances. However, a few simplified calculations
will be given as guidance for carrying out such determinations.
Table 19 gives a summary of the total lead intake and uptake from
the general environment in adults and in children aged 1 to 5. The
assumptions made are shown and are taken from WHO (1987). Additional
intake of lead will take place in certain groups from the use of
tobacco and alcoholic beverages. The estimates given are probably on
the high side with respect to the contribution from air and dust,
since indoor and outdoor lead contributions were considered equal.
Additional intake of lead is possible due to ciliary clearance of
particles 1-5 µm in diameter with subsequent swallowing and
gastrointestinal absorption.
5.5 Blood lead concentrations of various populations
Under certain conditions, blood lead (PbB) levels are a useful
indicator of exposure and are therefore discussed here, as well as in
section 6.1.4. In general, PbB levels correlate better with recent
exposure levels (Lyngbye et al., 1990b). As is discussed in sections
5.5.1 and 5.5.2, the general trend observed in all blood lead surveys
carried out in countries engaged in risk reduction programmes over the
last 15 to 20 years is a fall in the measured levels (OECD, 1993).
PbB concentrations are the most often used estimate of general
exposure to lead. The USA, Commission of European Communities (CEC),
Australia and the WHO have carried out epidemiological surveys to
determine lead exposures in various populations. The designs of these
studies differed; some provided estimates for the country as a whole,
others compared PbB levels of people working in the same occupation in
various countries, and others surveyed general populations but were
not designed to be extrapolated as national estimates. What these
studies have in common is that they did not emphasize groups with high
lead exposures, but concentrated on typical PbB concentrations of
non-occupationally-exposed groups in the regions or countries studied.
These surveys also offer the possibility of examining the data as
distributions, especially at the high end of the distribution profile.
Such examination may identify unusual exposures and thus populations
at risk. Average PbB concentrations may disguise the risk to various
segments of the population.
In the USA, national estimates of the extent and severity of
recent human exposures to lead in the general population were based on
PbB measurements from the second National Health and Nutrition
Examination Survey of 1976-1980 (NHANES II) (Annest & Mahaffey, 1984).
The design of this study permitted extrapolation to the USA population
as a whole.
Table 19. Estimates of lead (µg/day) absorbed by adults and children
from air, dust, food and watera
Mean air Dust Source of lead (µg/day) Total
lead intake absorbed
concentration (mg/day)b (µg/day)
(µg/m3) Air Dust Food Water
Adults
0.3 N.S. 2.4 -- 10 2 14.4
0.5 N.S. 4.0 -- 10 2 16.0
1.0 N.S. 8.0 -- 10 2 20
2.0 N.S. 16.0 -- 10 2 28
Children 1-5 years
0.3 -- 0.6 -- 25 5 30.6
0.5 -- 1.0 -- 25 5 31.0
1.0 -- 2.0 -- 25 5 32.0
2.0 -- 4.0 -- 25 5 34.0
1.0 25 2 12.5 25 5 44.3
1.0 50 2 25.0 25 5 57.0
1.0 100 2 50.0 25 5 82.0
1.0 200 2 100.0 25 5 132.0
a Adapted from WHO (1987); Dust is not considered a significant
source of lead in adults, but is a significant source for workers
where hygiene practices are poor
The above estimates are based on the following assumptions:
Air: Respiratory volume in adults is 20 m3/day, and in children
5 m3/day, and the respiratory absorption is 40%.
Food: Intake of lead by adults 100 µg/day with 10% absorption and
50 µg/day for children with 50% absorption.
Water: A lead concentration of 20 µg/litre, with adult consumption
of 1 litre/day and 10% absorption and for children 0.5 litre/day
with 50% absorption.
Dust: Dust concentration of lead was 1000 µg/g and absorption was
50%.
b N.S. = Not significant
The CEC study was directed toward biological screening of the
extent of exposure to lead outside the workplace using PbB level as
the index of exposure (CEC, 1981). The object of the study was to
assess non-occupational exposure of the population to lead in the
member states; it was implemented by member states and coordinated by
the CEC. Specific populations studied were selected by member states
and 168 separate areas and population groups were investigated in
1977.
Two other major international studies were limited to urban
populations. A pilot study of human lead exposure (Friberg & Vahter,
1983), organized by the United Nations Environment Programme and WHO,
was a collaborative effort under the Global Environmental Monitoring
System (GEMS). The following countries participated in at least part
of the study: Belgium, China, India, Islamic Republic of Iran, Israel,
Japan, Mexico, Peru, USA and the former Yugoslavia. The subjects of
the study were teachers, since they comprise an occupational group not
extensively exposed to lead. An Australian study was based on urban
residents only, and included 651 subjects between 6 and 91 years of
age (Hopper et al., 1982).
Based on demographic, economic and individual variables found in
the USA study to be associated with PbB levels (Mahaffey et al.,
1982), it is clear that results from these major studies cannot be
directly compared. However, a comparison of such studies can provide
information on the exposure to lead of segments of the various
populations in various countries worldwide.
5.5.1 Adult populations
In a large health screening programme within the USA during
1976-1980 (NHANES II), over 27 000 residents aged 6 months to 74 years
were examined (Annest & Mahaffey, 1984). PbB concentrations were
determined in a subsample (9933 individuals) and yielded an arithmetic
mean value of 0.67 µmol/litre (13.9 µg/dl). In 5841 individuals aged
18-74 years, the overall mean was 0.68 µmol/litre (14.1 µg/dl)
(Roberts et al., 1985). A sex difference was noted: 0.77 µmol/litre
(16.1 µg/dl) was found in males and 0.57 µmol/litre (11.9 µg/dl) in
females. Residents in the centre of large urban areas were found to
have a PbB level of 0.72 µmol/litre (14.9 µg/dl), while rural
residents it was 0.62 µmol/litre (13.0 µg/dl). Because of the design
of the study, information on PbB levels, in relation to smoking,
alcohol consumption, and occupational status, were available. Male
workers in occupations with a high potential for lead exposure had
mean PbB levels of 0.78 µmol/litre (16.2 µg/dl) for nondrinkers/
non-smokers, 0.92 µmol/litre (19.2 µg/dl) for non-drinkers/non-smokers
and 0.95 µmol/litre (19.7 µg/dl) for drinkers/smokers. It was noted
that a true decrease of 37% in PbB level occurred during the period of
the survey (Annest et al., 1983).
Rabinowitz & Needleman (1982) reported an arithmetic mean
umbilical cord PbB concentration of 0.32 µmol/litre (6.6 µg/dl) (range
0-1.78 µmol/litre; 0-37 µg/dl) in over 11 000 samples collected
between 1979 and 1981. A decrease in PbB level of approximately 11%
was noted during the period of collection.
Friberg & Vahter (1983) reported the PbB levels of 200 teachers
from nine countries (see section 5.5). The median PbB level in this
study ranged from 0.29 µmol/litre (6 µg/dl) in Beijing and Tokyo to
1.06 µmol/litre (22 µg/dl) in Mexico City. Smokers generally had
higher PbB levels than non-smokers.
During the period 1978-1988 marked decreases (approximately 30 to
40%) in the average PbB levels of adults were noted in Belgium,
Germany, New Zealand, Sweden, the United Kingdom, and the USA (OECD,
1993).
5.5.2 Children
Results from the NHANES II survey in the USA indicated a mean PbB
level for children (6 months to 2 years old) of 0.78 µmol/litre
(16.3 µg/dl). It was noted that 18.6% of black inner city children had
PbB levels > 1.44 µmol/litre (30 µg/dl) whereas only 4.5% of white
children had such PbB concentrations. The percentage of children with
high PbB level decreased with increasing family income (Roberts et
al., 1985). The NHANES II data were analysed in detail for information
on the demographic correlates of children's PbB concentrations
(Mahaffey et al., 1982; Annest et al., 1983). The racial differences
in these proportions were significant among both boys and girls. The
proportion with elevated PbB concentrations was slightly higher among
boys than girls for both races. Among both black and white children,
the percentage with elevated PbB concentrations decreased with
increasing family income. Proportionately more young children in urban
than rural areas and in the central cities of large urban areas had
elevated blood lead concentrations.
PbB concentrations were measured in 286 Finnish children living
in the three largest cities of Finland (number of subjects = 172), in
rural areas (54 subjects) and in a lead smelter area (60 subjects)
(Taskinen et al., 1981). The mean PbB levels in urban, rural and
lead-smelter areas varied between 0.29 µmol/litre and 0.32 µmol/litre,
range 0.14-0.82 µmol/litre (6.0 and 6.7 µg/dl, range 3-17 µg/dl).
There were no significant differences between the areas of residence.
The five children who lived within 500 metres of the lead smelter
had a mean PbB concentration of 0.44 µmol/litre, range 0.24-0.62
µmol/litre (9.2 µg/dl, range 5-13 µg/dl), which was significantly
higher than the mean blood lead concentration among children living
in the rest of the country.
In a study carried out in Sweden, which included 1781 samples
obtained from children during 1978-1988, the mean PbB level decreased
from 0.29 µmol/litre (59.6 µg/litre), range 0.09-1.20 µmol/litre
(18-250 µg/litre) in 1978 to 0.16 µmol/litre (32.9 µg/litre), range
0.07-0.34 µmol/litre (15-71 µg/litre) in 1988 (Schutz et al., 1989).
In Finland, a remarkably similar decrease has been reported; the mean
PbB value was 0.14 µmol/litre (30 µg/litre), range 0.1-0.40 µmol/litre
(21-41 µg/litre) among 35 children in the Helsinki area in 1988 (Ponka
et al., 1991).
As in the USA (Annest et al., 1983), children's PbB
concentrations in Sweden have decreased in recent years. Levels were
determined each summer during the period 1978-1984 in children from
Scania in Southern Sweden (Skerfving et al., 1986). The average PbB
concentration was 0.26 µmol/litre, range 0.07-1.20 µmol/litre
(5.5 µg/dl, range 1.4-25.0 µg/dl). There was a statistically
significant decrease over time in both the rural and urban areas,
averaging about 0.019 µmol/litre (0.4 µg/dl) per year.
Decreases of 25-45% in average PbB levels in children between
1978 and 1988 have been reported in Belgium, Canada, Germany, New
Zealand, Sweden and the United Kingdom (OECD, 1993).
5.5.3 Remote populations
In contrast, a much higher mean value of 0.16 µmol/litre
(3.4 µg/dl) was obtained in Nepal upon examination of 103 children
living in the Manang District, which is also very remote and thought
to be free of anthropogenic sources of lead (Piomelli et al., 1982).
However, these people are exposed to significant amounts of natural
lead from living in dusty smoke-filled houses, and from burning pine
and yak dung. The lead level in the houses was about 0.15 µg/m3
(Davidson et al., 1981). For comparison outdoor air in Woods Hole,
Massachusetts, USA, contains 0.004 and Boston, Massachusetts, 0.01 to
0.05 µg lead/m3.
Another remote population studied was from Okapa, Eastern
Highlands, Papua New Guinea (Poole et al., 1980). Extreme care was
taken in sampling and analysis, and the precision was acceptable at
PbB levels as low as 0.24 µmol/litre (5 µg/dl). The children subsisted
on root crops and a small amount of tinned fish and meat. Villages
were typically hazy with wood smoke, natural mineral pigments were
used extensively, and tobacco smoking was common. This survey of 100
children yielded a mean PbB level of 0.25 µmol/litre (5.2 µg/dl), well
above the detection limit, and, as reported by Poole et al. (1980),
far less than the mean of 1.10 µmol/litre (23 µg/dl) for Sydney,
Australia, in 1974.
5.6 Occupational exposure
Occupational exposure to lead which results in poisoning, both
moderate and clinically symptomatic, still occurs in many countries of
the world. Although adults are mainly involved, in many countries,
especially in those with developing industries and small home-based
industries, the distinction between home and workplace is non-existent
(Verrula & Noah, 1990) and children are exposed to workplace lead.
Until recently, in the occupational setting, there was only concern
for the identification of late stage, highly symptomatic cases of lead
poisoning resulting in crippling neurological manifestations of lead
poisoning such as palsy and encephalopathy (Chakravorti & Bhar, 1978;
Wang, 1984; Davies, 1984; Lin-Fu, 1985). However, there is now concern
for lower exposures to lead.
In many countries occupational lead exposure is entirely
unregulated and no monitoring of exposures exists. Automobile battery
manufacture and repair, radiator repair, secondary smelters (including
scrap metal refiners) are found in most countries. The industries
where there is a potential for lead exposure are listed in Table 10
(section 5.1.3). Significant lead exposures are not limited to
traditional heavy industries. For example, Kaye et al. (1987)
identified exposures from leadborosilicate dust used in a capacitor
and resistor plant in the USA.
Because of transfer of lead to the fetus (in utero) and the
transport to the home of lead on clothing, etc., thereby exposing the
young child in the home, the problems of occupational exposures to
lead are not limited to the workplace per se. Cases of poisoning
among the children of lead workers have been reported (Baker et al.,
1979; Wang, 1984; Ryu et al., 1985).
Although most occupational standards are based on airborne lead
only, this route of exposure does not fully reflect the total daily
exposure of workers, also exposed to lead in food, water, alcoholic
beverages and dusts.
The potential for hazardous exposures to lead during lead
smelting and refining are well recognized, particularly where molten
lead and alloys are poured, resulting in the vaporization of metal.
This is true for both primary new metal and secondary (lead scrap)
smelters and refineries.
Small domestic versions of secondary smelters exist in a large
number of countries. These are typically located within or in close
proximity to homes. For example, in Jamaica there has been a rapid
proliferation of lead smelters, particularly illegal backyard smelters
(Rodney & Lee, 1985). The lead fumes and dust generated in such
operations pose an exceptional health hazard to children and adults
living near these operations. Rodney & Lee (1985) reported that 51% of
116 children (aged 2 to 12 years, mean 5.9 years) and 60% of 235
adults working in or living near lead smelting factories had PbB
concentrations of 1.92 µmol/litre (40 µg/dl) or more.
Other occupations where workers have been shown to be at risk
from airborne lead include electric storage battery manufacturing,
particularly where industrial hygiene is poor (Barrio & Badia, 1985);
demolition, welding and shipbreaking where lead-based paint is present
(Holness & Nethercott, 1988; Nosal & Wilhelm, 1990); pottery and
ceramic-ware production, which is often a home-based operation
involving women and children (MolinaBallesteros et al., 1983; Katagiri
et al., 1983; Kaye et al., 1987); small businesses repairing
automobile radiators (Matte et al., 1989a,b; Verrula & Noah, 1990),
and artisans producing jewellery and decorative wares. This latter
industry is of particular concern since it is predominantly carried
out at home or in non-regulated shops, often by women and children
(Behari et al., 1982). Indian silver jewellery makers were found to
have a PbB level of 5.8 µmol/litre (121 µg/dl) compared to non-exposed
controls with a PbB level of 1.3 µmol/litre (27 µg/dl) (Behari et al.,
1982).
6. KINETICS AND METABOLISM IN LABORATORY ANIMALS AND HUMANS
6.1 Absorption
The absorption of lead from environmental sources is not
dependent solely on the amount of lead presented to the portals of
entry. It is also dependent on the physical and chemical state in
which the metal is presented, and it is influenced by host factors
such as age, physiological status, nutritional condition and,
possibly, genetic factors. Men engaged in heavy work breathe more air
and eat more food than sedentary individuals of the same weight, and
on a body weight basis children eat almost as much food and breathe
almost as much air as middle-aged adults.
Considerable data from human subjects are available. Therefore,
discussion of animal studies will be limited to areas where the
information on humans is inadequate. Various models used to predict
body burden or distribution of lead have been developed (Bernard,
1977; Marcus, 1985a,b,c; Bert et al., 1989; Arnetz & Nicolich, 1990).
A full review of these models has been judged to be beyond the scope
of this document. However, some of them were used in section 6.1.4
when correlating intake and body burdens.
6.1.1 Absorption after inhalation
The absorption of lead from air to blood involves two processes:
the deposition of airborne lead particles in the respiratory tract;
and the absorption and clearance from the respiratory tract into the
circulation. Using the International Radiological Protection
Commission (IRPC) document on lung dynamics (Task Group on Lung
Dynamics, 1966), a model was developed which predicted that 35 to 50%
of inhaled lead is deposited in the respiratory tract (40-50% of
particles with a mean mass median aerodynamic diameter (MMAD) of
0.5 µm, such as are typically generated by automobiles). These are
deposited primarily in the alveolar sacs of the lung. Lead fumes and
vapours, such as those generated in operations where metals are cut or
heated, are of very small size and are respirable. Absorption after
deposition will vary according to the solubility of the lead species
(e.g., lead carbonate or lead chloride aerosols) and the inherent
toxicity to lung macrophages and cilia.
6.1.1.1 Animal studies
Limited animal studies confirm that there is almost complete
absorption of lead particles (0.1 to 0.5 µm in diameter) deposited in
the lower respiratory tract. In rats the clearance half-time from lung
is short (less than one hour) and 90 to 98% of the administered dose
is absorbed within about 48 h (US EPA, 1986a).
6.1.1.2 Human studies
The respiratory deposition of airborne lead is in the range of
30-50% and varies with particle size and ventilation rate (US EPA,
1986a). Higher deposition rates may occur with larger particles, but
this deposition takes place in the upper respiratory tract, with
eventual displacement to the gastrointestinal tract and absorption via
the ingestion route. This probably explains the observation of Kehoe
(1961) that faecal excretion of lead increased after a subject
breathed for many weeks aerosols of lead oxide (150 µg/m3) with an
MMAD of approximately 2.9 µm. In contrast, smaller particles of
inhaled lead, such as those generated by automobile exhaust,
regardless of physicochemical form, are almost (> 90%) completely
absorbed after deposition in the lower respiratory tract (Rabinowitz,
et al., 1977a; Chamberlain et al., 1978; US EPA, 1986a).
There are no quantitative data on the absorption of lead in
children after inhalation exposure. It is known that young children
weighing only one sixth of an adult inhale 40% of the daily volume of
an adult and a proportionately higher daily air volume per unit
measure (weight, body area) than do adults (Barltrop, 1972). After
controlling for weight and taking into account differences in the
anatomy of the respiratory tract between adults and children, James
(1978) calculated a rate of deposition of lead particles in children
which was 1.6 to 2.7 times that of adults.
6.1.2 Absorption of lead from the gastrointestinal tract
In the case of older children and adults without occupational
exposure, lead absorbed by the gastrointestinal tract comes from the
intake of lead in foods, beverages and soil/dust. In pre-school
children, there is concern over the intake of both food and non-food
items (e.g., toys, soil/dust). Young children may take in lead from
non-food items, via normal mouthing activity, which in the extreme, is
the behavioural trait pica, which refers to the ingestion of such
materials as soil, ash, paint chips and plaster (US EPA, 1986a). For
infants and young children, the extent of absorption of the lead in
dust/soils from the gastrointestinal tract is extremely important,
particularly for children living in urban environments.
6.1.2.1 Animal studies
The absorption of lead from the gastrointestinal tract in
experimental animals is age dependent and is modified by the level of
food intake. In 1-week-old suckling rats, an absorption rate of about
52% was reported after a single oral dose of lead chloride, compared
to 0.4% in 6-week-old animals (Kostial et al., 1978). Furthermore,
Aungst et al. (1981) reported that rat pups had higher tissue levels
of lead than adults after a single gavage dose of 1 or 10 mg lead (as
lead acetate) per kg body weight. Fasting markedly enhanced the uptake
of lead in the gastrointestinal tract of experimental animals (Garber
& Wei, 1974; Pounds et al., 1978).
In experimental animals, absorption of lead from the
gastrointestinal tract appears to be a saturable process. With
increasing doses (1 to 100 mg lead (as lead acetate) per kg body
weight), lead absorption as a percentage of dose was found to decrease
from 42% to 2% (Aungst et al., 1981; Bushnell & DeLuca, 1983) in
dietary studies. Such data are consistent with a saturable active
transport process across the gastrointestinal tract.
The low rate of absorption of lead from soluble lead salts noted
in adult rats may reflect a dietary effect. Kostial & Kello (1979)
reported less than 1% absorption in adult rats given chow diets,
whereas the absorption rate was 3-20% when rats consumed diets similar
to that of humans (milk, bread, baby foods, etc). This range more
closely resembles the human absorption rates.
The chemical form of lead can influence its bioavailability.
Stone et al. (1981) determined the biological availability of lead
intrinsically incorporated into the soft tissues of oysters. The
bioavailability to the rat was 10-30% lower than that of lead acetate
added to the basal chow diet. Henning & Cooper (1988) compared the
absorption of lead from rat milk labelled in vitro with lead-203 and
lead chloride or lead acetate solutions. Lead from soluble salts
accumulated primarily in the duodenum, to some extent in the jejunum
and minimally in the more distal small intestine. Lead from milk
accumulated only in the upper ileum. After 20 h negligible lead-203
was found in any region following administration of soluble salts, but
there was substantial retention of lead in the ileum after dosing with
milk.
Data obtained from a feeding study in rats (Dieter et al., 1993)
showed that lead uptake into rat femurs was highly dependent on the
chemical form of lead administered. Bioavailability was highest for
lead acetate, intermediate for lead oxide, and lowest for lead sulfide
and Alaskan mixed ore concentrate. This uptake was linearly related to
dose over the range studied. However, the slopes of the linear
regression equations differed according to the form of lead. They were
0.10 µg lead/g femur per kg diet for lead sulfide and 2.64 for lead
acetate.
However, results of an investigation in young swine of the
absorption of lead from mining and/or milling operation waste (LaVelle
et al., 1991) differ from those reported by Dieter et al. (1993). The
chemical species (predominantly lead sulfide) and particle size (i.e.
larger than 100 µm) were expected to result in these sources being
less bioavailable than soluble lead salts such as lead nitrate.
However, experimental data indicate that the lead in mine tailings may
be about 2-3 times more available than reagent grade lead sulfide
under the conditions of the study. Additionally, Freeman et al. (1992)
reported that the lead in mining waste soil was between 8 and 20% as
bioavailable as lead acetate. No detailed comparisons were made of the
physicochemical properties of these two mine wastes. An additional
study involving the absorption of lead from lead sulfide ores and
their oxidation products in rabbits and in vitro showed that lead
from minerals was absorbed less well than from lead acetate by a
factor of 5 (Davis et al., 1990a). The significance of these phenomena
is not clear.
6.1.2.2 Human studies
Gastrointestinal absorption of lead in humans, as in experimental
animals, is influenced by dietary factors, nutritional status and the
chemical form of the metal. Overall patterns of food intake may also
influence lead absorption. For example, lead ingested during periods
of fasting is absorbed to a much greater extent than lead ingested
with food. Chamberlain et al. (1978) reported 45% absorption of lead
chloride in fasting subjects and only 6% in feeding subjects. Using a
similar procedure, Heard & Chamberlain (1982) reported absorption of
63.3% in fasting subjects. Using multiple stable isotope lead tracers,
it was found that in adult men the gastrointestinal absorption rate
with food containing lead nitrate or lead cysteine was 6-12%. However,
when consumed under fasting conditions, lead nitrate, lead sulfide and
lead cysteine were absorbed at 16-53% (Rabinowitz et al., 1977b).
Kehoe (1961) estimated only 10% net absorption of dietary lead by
adults in long-term metabolic studies. However, with appropriate
calculation of biliary clearance as well as of urinary excretion, a
figure of 15% was estimated by Chamberlain et al. (1978). In several
studies on adult humans, absorption of lead was reported to be 14%
when it was administered with food (Chamberlain et al., 1978; Moore et
al., 1979; Rabinowitz et al., 1980).
Ziegler et al. (1978) reported that young children, aged two
weeks to two years, absorbed 42% of ingested lead at levels of intake
greater than 5 µg/kg body weight. Drill et al. (1979) estimated an
absorption rate of 17% for lead in paint chips in children aged 2 to 3
years. These authors also estimated a 30% gastrointestinal absorption
rate for lead in soil and dirt.
The amount of lead ingested by children from non-food items such
as soil, dust and paint chips through normal mouthing activity,
particularly for children with pica, is a major concern in calculating
paediatric lead exposures. Using data from Day et al. (1975) and Lepow
et al. (1974), it has been estimated that children 2 to 3 years of age
ingest about 100 mg soil per day. Using aluminium and titanium as
tracers, Clausing et al. (1987) estimated that children aged 2 to 4
years ingest between 50 and 100 mg of soil daily. The average amount
of soil ingested by young children has recently been estimated to be
between 12.5 and 21 mg/day (SAHC, 1993).
6.1.2.3 Nutritional status and lead absorption via gastrointestinal
tract
It has been known for some time that the absorption and
distribution of lead are affected by nutritional status in both
experimental animals and humans (Sobel et al., 1940). Nutritional
inadequacies can also affect the toxic response to lead (see sections
7 and 8). In view of documented nutritional inadequacies in many parts
of the world, such interrelationships become crucial in assessing the
full risk from lead exposures.
Vitamin D, calcium and phosphorus have complex and interrelated
effects on lead absorption (Fullmer, 1990). Increasing the
concentration of 1,25-dihydroxycholecalciferol, the active metabolite
of vitamin D, either exogenously or endogenously, increases
gastrointestinal absorption of lead (Fullmer, 1990). However, this
effect is dependent upon the duration of lead exposure and the
magnitude of body lead stores. This homoeostatic mechanism for calcium
and its dependence on nutritional status, as well as body burden of
lead, is complex. This may explain the divergent results of the
observed interaction in children (Rosen et al., 1980) or the lack of
association (Koo et al., 1991) of lead with vitamin D metabolism.
In experimental animals, chronic ingestion of diets with less
than adequate amounts of calcium (Quarterman & Morrison, 1975),
phosphorus (Quarterman & Morrison, 1975), iron (Mahaffey-Six & Goyer,
1972; Ragan, 1977), selenium (Stone & Soares, 1976) or zinc
(Cerklewski & Forbes, 1976) increases the fractional absorption of
lead in the gastrointestinal tract. From the work of Fullmer & Rosen
(1990), it appears that lead alters the transport of calcium and other
trace minerals by affecting carrier proteins, rather than by competing
at the mucosal surface.
The influence of iron on lead absorption in humans was assessed
in double-labelling experiments conducted by Watson et al. (1980).
Lead-203 and iron-59 were given to 28 subjects in their diet. There
were significant positive correlations between iron levels in food and
lead absorption. However, the association was not strong because 50%
of those absorbing excess lead also absorbed excess iron.
Rats fed iron-deficient diets have increased concentrations of
lead in kidney and bone when compared to rats ingesting equivalent
quantities of lead (as lead acetate) in drinking-water while being
fed an iron-adequate diet (Mahaffey-Six & Goyer, 1972). In contrast
with calcium deficiency, iron deficiency does not result in a
redistribution of lead to non-osseous tissue (Mahaffey-Six & Goyer,
1972). The degree of iron deficiency does not need to be severe to
increase retention of lead. For example, Ragan (1977) demonstrated
six-fold increases in tissue lead in rats when body iron stores were
reduced, but before frank iron deficiency developed. High levels of
dietary iron resulted in decreased kidney, femur and blood lead
concentrations in rats (Mahaffey, 1985). Iron appears to increase
absorption rather than decrease lead excretion (Barton et al., 1978).
Lead interferes with normal transferrin transport of iron, and
inhibits transferrin endocytosis and iron transport across the cell
membrane of the reticulocyte (Qian & Morgan, 1990).
6.1.3 Dermal absorption
6.1.3.1 Human dermal absorption
Moore et al. (1980) examined the uptake of lead acetate from two
hair-darkening cosmetics through the skin of eight human volunteers.
Only minute quantities of lead (0-0.3% of the applied dose) were
detectable in blood, and there was only a slight increase in
absorption when the skin was damaged. Lilley et al. (1988) and
Florence et al. (1988) have reported the dermal absorption of
inorganic lead compound leading to elevated levels of lead in human
saliva and sweat.
Dermal absorption of inorganic lead through unabraded human skin
is considered to be minimal.
6.1.4 The relationship of external lead exposure to blood lead
concentration
There are numerous environmental media that provide routes by
which humans are exposed to lead: food, water, air, soil, dust.
External exposures are the sum of the quantities of lead consumed from
all sources.
Historically there have been two lines of approach in
understanding the lead exposure and blood lead relationship. Most have
been empirical and have measured environmental lead and PbB levels
either at one time or repeatedly. With these observed correlations and
with no assumptions about how lead moves inside the body, many
reasonable predictions can be made. The validity of these predictions
is optimum when only one environmental source dominates. Some of these
rely on linear functions, while others have specified non-linearities,
especially over very wide ranges (more than a factor of 10) of lead
exposures. However, when multiple sources are considered these
predictive models have been less satisfactory.
Internal lead levels in human populations have been estimated by
analyses of a variety of biological tissues (e.g., blood, teeth, bone
and hair). Lead concentrations in each of these have particular
biological meanings with regard to external exposure to lead. Blood is
the compartment in which lead is most often measured as a marker of
exposure. However, it typically represents relatively recent
exposures, since the half-life of lead in blood is short (US EPA,
1986a) and has been estimated to be in the order of 36 days from
tracer studies (Rabinowitz et al., 1975). Lead in blood is derived
from levels in the current environment and from lead stored in tissues
(particularly bone) that re-enters the blood during tissue
mobilization (Manton, 1985). Although PbB concentration reflects
recent exposure and bears a consistent relationship to levels of lead
in the external environment, when bone mobilization is accelerated a
greater fraction of PbB will be derived from tissue stores.
To date only a few studies have utilized a multimedia approach
relating lead intake to PbB levels. The majority of studies have
attempted to correlate PbB levels and lead concentrations in specific
media.
6.1.4.1 Ambient air
a) Occupational exposure
Several studies have examined the blood lead: air lead
relationship for workers exposed to levels of airborne lead between 9
and 450 µg/m3 (King & Conchie, 1979; Gartside & Buncher, 1982;
Bishop & Hill, 1983). The results of all three studies are in general
agreement over the wide range of airborne lead studied and for PbB
levels between 0.96 and 4.32 µmol/litre (20-90 µg/dl). The blood lead:
air lead relationship in occupational settings is best described by a
curvilinear relationship having slopes between 0.00096 and
0.0038 µmol/litre (0.02 and 0.08 µg/dl) per µg/m3 air.
b) Non-occupational exposure
Both population and experimental studies have been used to
estimate the PbB: ambient air lead relationship in adults and
children. Under ambient conditions (air lead concentrations of
0.1-2.0 µg/m3) and PbB levels less than 1.44 µmol/litre
(< 30 µg/dl), the relationship has been described as linear (Colombo,
1985). The various slope estimates reported are based on the
assumption that an equilibrium level of lead in blood is achieved.
Based on three major studies (Yankel et al., 1977; Angle & McIntire,
1979; Roels et al., 1980), the median slope for children is about
0.091 µmol/litre (1.9 µg/dl) blood per µg/m3. In adult males a slope
estimate of 0.076 µmol/litre (1.6 µg/dl) blood per µg/m3 was
calculated. When one calculates the relationship between PbB and the
total contribution from air (direct inhalation plus indirect through
dust/soil), a value of about 0.14-0.24 µmol/litre (3-5 µg/dl) blood
per µg/m3 is obtained (Brunekreef, 1984; US EPA, 1986a). Also on the
basis of a linear model, Snee (1981) reported that the best estimate
of the blood lead: air lead relationship was 0.048 µmol/litre
(1 µg/dl) per µg/m3. After examining available epidemiological and
experimental data, Chamberlain (1983) concluded that most published
estimates of the slope were between 0.072 and 0.144 µmol/litre per
µg/m3. From these results, it can be concluded that airborne lead
will only be a major contributor to PbB levels in areas of high air
lead levels.
6.1.4.2 Food
The relationship of PbB to dietary intake has been estimated from
experimental (Stuik, 1974; Cools et al., 1976; Schlegel & Kufner,
1979) as well as population studies (Sherlock et al., 1982; UK, 1982;
Ryu et al., 1983). In adults, the results from both types of studies
using both linear and cube root models indicated a relationship of
between 0.0019 and 0.0028 µmol lead/litre (0.04 and 0.06 µg/dl) per µg
lead intake per day. From the study of Ryu et al. (1983) a slope of
0.0096 µmol/litre (0.2 µg/dl) per µg lead/day was obtained for infants
aged 8 to 196 days.
Currently, data are available for adults and children from
studies with careful control of important variables such as: intake of
dietary lead and of other dietary constituents, minimal exposure to
sources other than diet in the studies of infants, and intake/blood
lead measurements that can be used to estimate intake from all
sources. For infants these are studies reported by Sherlock et al.
(1982), UK (1982), Ryu et al. (1983), and Lacey et al. (1985). Studies
by the UK (1982) and by Sherlock et al. (1982) were conducted at
higher levels of lead exposure than were the studies conducted with
infants by Ryu et al. (1983). The latter studies were at exposure
levels associated with PbB concentrations under 0.96 µmol/litre
(20 µg/dl); the ratio of blood lead to ingested lead was
0.0076 µmol/litre (0.16 µg/dl) per µg lead ingested per day. Exposure
levels were low: average dietary lead intake was 17 µg/day. After four
months, the average PbB concentration was 0.29 µmol/litre (6.1 µg/dl)
whole blood. Between the ages of 4 and 6 months, 10 children remained
at a dietary lead intake of 0.76 µmol/litre (16 µg/dl). Their PbB
concentrations were quite constant at 0.35 µmol/litre (7.2 µg/dl) at
the end of the study period. The remaining seven children received
canned infant formula and/or milk during this period and their average
dietary lead intake was 61 µg/day. At the end of the study period,
their PbB concentration was 0.69 µmol/litre (14.4 µg/dl). Based on
these data, a curvilinear relationship between blood lead and total
lead intake was suggested, a 4-fold increase in lead intake resulting
in a doubling of PbB concentration. Sherlock et al. (1982) conducted a
duplicate diet study of 31 mothers and their children from Ayr,
Scotland. The slope for adults was substantially lower than for
children.
6.1.4.3 Drinking-water
There is still debate over the most appropriate model (i.e. cube
root, polynomial or logarithmic) to describe the curvilinear
relationship of waterborne lead to blood lead. The highest estimates
for the contribution of water lead to blood lead come from the cube
root and logarithmic models. Much lower estimates are obtained from a
linear model (Pocock et al., 1983; US EPA, 1986a). In a study by
Sherlock et al. (1982), a cube root relationship between lead levels
in drinking-water and blood fitted the data more closely than a linear
relationship. The curvilinear model implies that as abatement
processes lower water lead concentrations, there will be an increasing
benefit in lowering of population PbB levels (Moore, 1983).
6.1.4.4 Soil and dust
It is extremely difficult to choose the most appropriate model to
describe the soil/dust to blood lead relationship, given the many
variables involved in determining the exposure patterns of children
and the kinetics involved between the levels in the environment and
the child. A review of the available studies shows the extreme
variability in slopes obtained (0.028-0.36 µmol lead/litre
(0.6-7.6 µg/dl) blood for each 1000 µg/g soil and 0.00096-0.35 µmol
lead/litre (0.02-7.2 µg/dl) blood for each 1000 µg/g dust consumed by
children) (US EPA, 1986a). Detailed consideration has been given to
the process for the assessment of lead contamination in soil and the
derivation of soil clean-up criteria by Wixon (1991). The guideline
model uses a PbB target and slope for the soil:blood lead relationship
in the particular community in order to derive a PbB guideline. For
house dust a median value of 0.086 µmol lead/litre (1.8 µg/dl) blood
per 1000 µg lead/g dust in children appears to be based on data of
reasonable quality, as does the 0.105 µmol lead/litre (2.2 µg/dl)
blood per 1000 µg lead/g soil from the same authors.
According to Elwood (1986), studies of the association between
lead in house dust and PbB have given inconsistent results and, in
general, the only studies in which statistically significant
association has been found are those where lead levels in dust are
quite high. Landrigan et al. (1975) found a highly significant
association between PbB and dust lead in an area with a mean level of
4022 µg lead/g dust, but not in two areas with means of 922 and 816 µg
lead/g dust. The US EPA (1986a) summarized a series of studies from
which the overall relationship was judged to be 0.86 µmol/litre
(18 µg/dl) blood per 1000 µg lead/g dust. Duggan (1980) reviewed the
literature and determined that a slope of 0.24 µmol/litre (5 µg/dl)
per 1000 µg lead/g dust is reasonable.
The overall relationship between PbB and dust/soil lead depends
on the lead concentrations and bioavailability as well as on the
proximity and linkage between humans and their environment. This
relationship varies among locales.
6.1.4.5 Total lead intake
The non-linear relationship between PbB and total lead intake
(see section 5.5) is curvilinear across a broad range of PbB values,
such that the slope decreases with increasing lead levels. A number of
biological factors may explain the curvilinear relationships, such as
increased renal clearance with high PbB (Chamberlain, 1983, 1985),
distributional non-linearities due to differences in lead binding
sites in different tissues (Hammond et al., 1981; Manton, 1985;
Marcus, 1985b), or a sizeable pool of mobile lead in bone maintained
more or less independently of uptake (Rabinowitz et al., 1976;
Chamberlain, 1983).
6.2 Distribution
The initial distribution of lead in the body may depend upon the
rate of delivery of blood to various organs. However, it would appear
that distribution occurs in a similar manner regardless of the route
of absorption (Kehoe, 1987). The distribution of lead in humans under
environmental exposure conditions reflects the fact that almost all
exposures are chronic rather than acute.
6.2.1 Animal studies
Studies in rats have shown that lead is rapidly distributed into
soft tissues and subsequently redistributed into soft and mineralizing
tissues after acute and chronic exposures. After acute inhalation
(Morgan & Holmes, 1978) or oral (Aungst et al., 1981) exposure, lead
levels in rats were highest in liver, kidneys and lung, with levels
increasing in bone as those in soft tissues declined and stabilized.
Similar distribution patterns were reported for mice exposed for 12
months to 21.5 µg lead/m3, the highest levels being found in bone
and the lowest in lung (Keller & Doherty, 1980a).
Age-related differences in the distribution of lead in
experimental animals have been reported. Kostial et al. (1978) noted a
greater retention of lead in suckling rats than in adults; the levels
in the brain were also higher in the pups. A 2- to 3-fold increase in
brain lead concentration (highest in hippocampus) was noted after a
10-fold (0.1 to 1 mg/kg body weight administered by gavage) increase
in the dose given to 4- to 8-week-old rat pups (Collins et al., 1992).
Ageing has been shown to alter the pattern of distribution of
lead in rats administered lead acetate in drinking-water. Juvenile (21
days old), adult (8 months old) and elderly (16 months old) rats
received 0, 1.27 and 6.37 mg lead/kg bodyweight for 9.5 months. The
pattern of distribution, namely femur > liver > brain was similar in
all age groups. However, age-related increases in brain lead levels
were noted, along with decreases in femur lead content (Cory-Slechta,
1990a).
6.2.2 Human studies
Lead is distributed to both soft tissues (blood, liver, kidney,
etc.) and mineralizing systems (bone and teeth). Bone may be affected
adversely by lead but also serves as the body's major storage site.
Bone accumulates lead over much of the human life span, and a study of
the kinetics of distribution is important since bone can, under
appropriate conditions, pose a risk as a potential endogenous source
of lead.
Once absorbed, lead is not distributed homogeneously throughout
the body but rather into several distinct compartments. Such
biokinetic movements have been explained by Rabinowitz et al. (1976)
using a three-compartment model. This model was based on tracer and
balance data from five healthy men and has been refined by Marcus
(1985a,b,c). Three pools (blood, bone and soft tissues) were
identified, with lead having distinct half-lives in each. Blood lead
was considered the most labile compartment with a half-life of about
36 days, and bone lead the most stable with a half-life of about 27
years. Lead in soft tissue had a half-life of approximately 40 days.
The specific physiological foundations for these biokinetic
models have received much attention and are currently being refined
(O'Flaherty, 1991, 1993). A more thorough understanding of bone
growth, for example, expressed as a series of allometric equations,
should help improve models, especially when they must be applied to
growing and maturing humans.
Under steady-state conditions, about 96% of PbB is in the
erythrocyte. At PbB concentrations of < 1.92 µmol/litre (40 µg/dl),
whole blood and serum lead levels increase linearly in a positive
manner. At higher PbB concentrations a curvilinear relationship is
apparent and the serum to blood ratio increases dramatically (Manton &
Cook, 1984). Such kinetic relationships may be altered during
pregnancy. From in vitro data (Ong & Lee, 1980), fetal haemoglobin
appears to have a greater affinity for lead than adult haemoglobin.
In adults, approximately 94% of the body burden of lead is in the
bones, whereas only 73% of the body burden in children is located in
this compartment (Barry, 1975, 1981). In view of the extremely long
half-life for lead in bone, this compartment can serve as an
endogenous source of lead to other compartments long after exposure
ceases (O'Flaherty et al., 1982; Kehoe, 1987). This is due to the
labile lead compartment in bone (Rabinowitz et al., 1976) and the
ongoing bone redistribution which is subject to alteration by in vivo
metabolic processes (Parfitt, 1990). Although lead in bone generally
increases continuously with age, there is evidence that lead levels in
some bones (e.g., mid-femur and pelvic bone) plateau at middle age and
decrease with further ageing (Drasch et al., 1987). It is difficult,
however, to determine the role ageing plays in this process compared
to other biological reactions. Ageing may account in part for the
20-25% increase in PbB levels in menopausal women noted by Silbergeld
et al. (1988).
The metabolism of lead in bone has been summarized in reports by
Barry (1975, 1981), Drasch et al. (1987), Silbergeld et al. (1988),
Skerfving (1988) and Rabinowitz (1991).
Until recently, it was widely held that the human skeletal system
provided a metabolically inert depository for lead and was of little
consequence in health-risk assessment. It had been assumed that bone
lead is metabolically inert, with a half-life long enough to forestall
the risk of ready transfer back to blood. Current evidence is that
bone comprises a set of kinetically variable subcompartments for lead
deposition and is a target for toxicity. These factors complicate bone
lead kinetics as applied to long-term modelling; the mobility of bone
lead to blood is important (Rabinowitz, 1991).
Bone lead is readily mobilized to blood and the effect is most
apparent in people with a history of occupational exposure; bone lead
also appears to be a major source of blood lead in older people with
previous ambient exposures to lead. Of particular importance is
mobilization of lead from bone in pregnant women and nursing mothers
(Silbergeld, 1991). The mobilization of lead from bone to the more
bioavailable maternal blood compartment poses a risk to the fetus and
mother.
Human bone appears to have at least two, possibly three,
kinetically distinct lead compartments. Lead in trabecular (spongy)
bone appears to be more mobile than lead lodged in cortical (compact)
bone, and there appears also to be a fraction of bone lead in
equilibrium with the lead in blood (Skerfving, 1988). Trabecular bone
seems to be an important source of resorbed lead when high exposure is
reduced, e.g., through removal of medical reasons by retirement of
lead workers, or in response to chelation in adults (Shutz et al.,
1987).
6.2.3 Transplacental transfer
Lead is readily transferred from the mother to the developing
infant during pregnancy and accumulates in bone during gestation
(Barltrop, 1969). The lead concentration in cord blood is 85-90% that
of maternal blood. Moore et al. (1982) reported a geometric mean level
of 0.67 µmol/litre (14 µg/dl) in 236 pregnant women and
0.58 µmol/litre (12 µg/dl) for lead in umbilical cord blood. The mean
concentration of lead in umbilical cord blood from a sample of over
11 000 women was 0.298 µmol/litre (6.6 ± 3.2 µg/dl) (Bellinger et al.,
1987).
6.3 Elimination and excretion
In both humans and experimental animals lead is eliminated from
the body in both urine and faeces. Any dietary (including waterborne)
lead not absorbed in the gastrointestinal tract is excreted in faeces.
Airborne lead that has been swallowed and not absorbed is also
eliminated in this manner. Blood lead not retained in the body is
excreted in urine or faeces, the latter by biliary excretion. Adults
ingesting daily 0.3 to 3.0 mg lead (as lead acetate) in drinking-water
for 16 to 208 weeks excreted more than 85% of the ingested lead, 90%
of the excreted lead being in the faeces. The amount excreted through
any route is affected by age and exposure characteristics and is
species dependent (US EPA, 1986a). This section reviews only studies
of lead excretion in humans; a discussion of the results of work on
experimental animals can be found in reviews by US EPA (1986a) and
ATSDR (1993).
The age dependency of lead excretion in humans has not been
studied extensively. However, the studies of Rabinowitz et al. (1977b)
and Chamberlain et al. (1978) in adults and Ziegler et al. (1978) in
infants can be used to assess this phenomenon. Data from Ziegler et
al. (1978) and Rabinowitz et al. (1977a) are given in Table 20.
Ziegler et al. (1978) calculated a total daily retention by infants of
40 µg, which is about twice the amount calculated by Alexander et al.
(1973).
Table 20. Comparison of daily lead intake and excretion in children
and adultsa
Parameter Childrenb Adultsc
Dietary intake (µg/kg) 10.76 3.63
Fraction absorbed 0.55d 0.15
Dietary lead absorbed (µg/kg) 5.92 0.54
Air lead absorbed (µg/kg) 0.20 0.21
Total absorbed lead (µg/kg) 6.12 0.75
Urinary lead excreted (µg/kg) 1.00 0.47
Endogenous faecal lead (µg/kg) 1.56 0.24
Total excreted lead (µg/kg) 2.56 0.71
Excreted/absorbed lead 0.42 0.92
Fraction of intake retained 0.33 0.01
a Adapted from: US EPA (1986a)
b From: Ziegler et al. (1978)
c From: Rabinowitz et al. (1977a)
d Corrected for calculated endogenous faecal lead
Under conditions of relatively constant exposure to low
concentrations of lead, approximately 140 to 215 µg/day, a steady
state condition evolves in which excretion approximates intake
(Rabinowitz et al., 1976). Under these conditions urinary lead
excretion is approximately 70% of absorbed lead. Chamberlain (1985)
reported that approximately 60% of absorbed lead is retained by the
body and 40% excreted.
Chamberlain (1983, 1985) also examined the relationship between
the level of exposure and rate of lead excretion. Renal clearance at
PbB levels of between 1.2 and 3.84 µmol/litre (25 and 80 µg/dl) was
found to increase at a rate approximating the increase in plasma lead.
Chamberlain (1985) estimated endogenous faecal lead loss into the
gastrointestinal tract following administration of lead-203 via the
inhalation and parenteral routes. These estimates suggested a
clearance of approximately 0.5% of administered dose per day when PbB
concentrations were under 1.2 µmol/litre (25 µg/dl). A special form of
excretion of endogenous lead is through breast milk. Studies of breast
milk indicate that lead concentrations correlate with maternal PbB
concentrations, most studies reporting that lead secreted from breast
milk varies in concentration between 10 and 30% of the maternal PbB
concentration (Ong et al., 1985).
6.4 Biological indices of lead exposure and body burden
6.4.1 Blood lead
The relationship between levels of exposure from various
environmental media and PbB has been discussed briefly in section
6.1.4.
Due to the ease of sampling and homogeneity of the sample, blood
has been the most widely used specimen to assess the human body burden
of lead. However, in view of the relatively short half-life for lead
in blood (28-36 days) (see section 6.2.2), PbB measurements in general
reflect only recent exposures. Also, in view of the kinetics of
distribution within the body (cycling between blood, bone and soft
tissues), differentiation of low-level chronic exposure from a short
high-level exposure is not possible on the basis of a single PbB
measurement. Interpretation of PbB levels over a wide range of values
must take account of the curvilinear relationship between total intake
of lead and PbB concentrations, as well as the proportion of lead in
plasma (Manton & Cook, 1984; see also section 8.1).
A number of cohort studies have collected serial PbB measurements
for children from birth up to 7 or 10 years. For children who have not
had major changes in their environment, there is good correlation
between consecutive PbB measurements (McMichael et al., 1988;
Bellinger et al., 1992; Baghurst et al., 1992; Dietrich et al.,
1993a,b). In these extended studies it has become apparent that for
most of the children a single PbB analysis at 6 years of age gives a
reasonable assessment of the life-time lead exposure status of the
child. However, random PbB levels in samples taken before 6 years of
age can markedly underestimate the peak exposure usually seen at 2
years of age (SAHC, 1993).
A new exposure index, namely "lifetime average blood lead" or
"lifetime average integrated blood lead" has been introduced in
studies using serial blood data. It has been clearly explained in the
proceedings of a recent Workshop and this explanation is quoted
directly here. This measure, which is a reflection of the area under
the "blood lead by age curve", has been occasionally misunderstood.
Due to the shape of the longitudinal blood lead profile, the peak
blood lead level, usually observed during the second year of life is
considerably greater than the average lifetime blood lead. For
example, a five-year-old child with a lifetime average blood lead of
0.96 µmol/litre (20 µg/dl) is likely to experience blood lead levels
above 1.92 µmol/litre (40 µg/dl) during the second year of life and
may spend 3 years of life, from 12 months to 48 months, above
0.96 µmol/litre (20 µg/dl). Due to the shape of the curve, with
declining PbB beginning about 24-39 months of age, the average
lifetime blood lead for a given child decreases with increasing age.
This does not mean that the index is inappropriate but rather that
developmental blood lead profiles change over time. For example, a
lifetime average blood lead of 0.72 µmol/litre (15 µg/dl) should not
be interpreted as being equivalent to a single blood lead
determination of 0.72 µmol/litre (15 µg/dl) obtained at a single point
in life (Dietrich et al., 1991).
6.4.2 Tooth lead
Unlike blood samples, teeth are composed of several anatomically
distinct pools which form over several years. Thus, in contrast to
blood, teeth are useful tissues for assessing long-term lead
accumulation from prenatal exposures to the time of shedding of the
tooth.
The accumulation of lead in teeth (PbT) has been used as a
measure of exposure of children to lead in several epidemiological
studies (Needleman et al., 1972; Winneke et al., 1982a; Rabinowitz et
al., 1989; Fergusson et al., 1989; Hansen et al., 1989). Studies
measuring PbT and PbB, however, are few. Preliminary data from
Australia (Baghurst et al., 1992) indicate a correlation of 0.8
between whole PbT and PbB prior to tooth exfoliation, but the only
report relating lead in circumpulpal dentine and longitudinal PbB (by
Greene et al., 1992) found a correlation of only 0.5. PbT
concentrations can vary as a function of location of the tooth within
the mouth, age, and whether total PbT or dentine PbT were reported.
Therefore, the age of the tooth in the mouth and its location, the
sample (whole tooth or dentine) analysed must be considered in any
estimation of a corresponding PbB in the published studies (SAHC,
1993).
Because of the complex structure and development of teeth,
concentrations of PbT will depend on the method of sampling and
analysis, tooth type, and resorption and tooth age at exfoliation. As
a result of varying procedures used by different investigators, there
may be substantial variation in absolute values and possibly the
biological meaning of PbT levels between different studies.
Different parts of a tooth sequester lead during different stages
of development. This is especially of concern in tooth sampling
because lead is not uniformly distributed within a tooth on a
submillimeter scale. A pilot study of teeth from children with
differing histories of exposure to lead and using high precision lead
isotopic methods has shown that analyses of slices of the incisal part
of deciduous teeth give the clearest indications of the in utero
environment, and the cervical sections, the exposure from birth to
exfoliation (Gulson & Wilson, 1994).
For more detailed discussions on the measurement of PbT the
reader is referred to Grandjean et al. (1984), Purchase & Fergusson
(1986) and Fergusson et al. (1989).
6.4.3 Bone lead
The human skeleton begins to accumulate lead during fetal
development and continues to abut 60 years of age (Pounds et al.,
1991). Interest in bone lead and its measurement in vivo stems from
concern that skeletal lead is not metabolically inert (see section
6.2.2), but can be mobilized by physiological and pathological states,
for example, during pregnancy and lactation (Silbergeld, 1991) and
osteoporosis (Silbergeld et al., 1988), with possible adverse effects
in other tissues, including the fetus, as well as the desire to
develop a meaningful measure of cumulative lead exposure as a tool in
public health protection. Section 8.13 includes a brief discussion of
the skeleton as a target organ for lead toxicity.
Procedures are available to analyse bone samples for lead levels
in humans not occupationally exposed to lead (Drasch et al., 1987;
Drasch & Ott, 1988). These studies have shown a decrease in levels of
lead in bones from autopsy specimens in Germany after removal of lead
from petrol. However, full utilization of bone lead stores as
dosimeters of lead exposure in a prospective sense requires the
utilization of technologies for in vivo measurement of lead in bone
such as X-ray fluorescence analysis (Chettle et al., 1991; Rosen et
al., 1991; Todd et al., 1992).
6.4.4 Lead in urine
Although urinary lead level has been used to measure current
exposure (Robinson, 1974), its use as a biomarker of lead exposure is
questionable in view of the relatively low and variable level of lead
excreted in the urine (Jensen, 1984; Ibels & Pollock, 1986). For this
reason, and in view of technical difficulties in analysing low levels
of lead in urine, urinary lead appears to be of limited use for
general screening. However, where an elevated body burden of lead has
been estimated using PbB or other indices of lead exposure, urinary
levels of lead after administration of the chelating agent
CaNa2-EDTA is considered an excellent measure of the potentially
toxic fraction of the total body burden of lead (CDC, 1985). The
chelatable lead excreted is assumed to represent lead removal from
soft tissues and blood, as well as sub-compartments of bone (Ibels &
Pollock, 1986; Mushak, 1989). In contrast, the urinary lead excretion
associated with lead mobilization provides what is considered the best
measure of the potentially toxic fraction of the total body burden
(see CDC, 1985; US EPA, 1986a). On the basis of various in vitro
experimental and epidemiological studies (CDC, 1985; US EPA, 1986a;
Mushak, 1989), chelatable lead is assumed to be a chemical sample of
both mobile body compartments (i.e. blood and soft tissues) as well as
of sub-compartments of bone.
6.4.5 Lead in hair
Hair lead has been used as an indicator of exposure in children
(Marlowe & Errera, 1982; Wilhelm et al., 1989). However, there are
severe limitations on its use from both the methodological as well as
the metabolic perspective. Systemic variations in lead level have been
reported according to hair colour, texture, location on the body and
growth phase (Wilhelm et al., 1989). Also, it is almost impossible to
avoid external contamination, and to date no validated methods are
available for cleaning. Methods which are sufficiently vigorous to
remove superficial lead also remove lead from the hair shaft.
7. EFFECTS ON LABORATORY ANIMALS AND IN VITRO TEST SYSTEMS
Lead can affect various organ systems depending upon the level
and duration of exposure. In all species studied, adverse effects on
the nervous system have been noted at PbB concentrations lower than
for other target organs. There is particular concern for the effects
of lead on fetal development. Since the appearance of Environmental
Health Criteria 3: Lead (IPCS, 1977), there have been very many
reports on the effects of lead on in vitro and animal models. No
attempt will be made in this chapter to summarize all such studies.
Rather, emphasis has been given to those studies that relate most
directly to the understanding of the effects of lead on humans and
thus provide additional scientific support for the use of such human
studies in the assessment of risk from lead exposures.
7.1 Biochemical effects
7.1.1 Haem synthesis and haematopoiesis
The principal clinical manifestation of the effect of lead on the
haematopoietic system is anaemia but this occurs only with high levels
of exposure that are rarely seen today. Lead affects the
haematopoietic system at several levels. These include effects on haem
and globin synthesis and on erythrocyte formation and function.
Most haem synthesis is directed toward formation of haemoglobin
and the rest is used in cellular oxidative metabolism. Lead inhibits
several steps in the biosynthesis of haem. More detail can be found in
IPCS (1977), US EPA (1986a) and section 8.1.1 of this monograph.
Lead acts on steps in the synthetic pathway both inside and
outside the mitochondrion. It inhibits certain enzymes (ALA
dehydratase, ferrochelatase, coproporphyrinogen oxidase) and increases
the activity of ALA synthase (ALAS) activity as a consequence of
feed-back regulation by haem (Moore et al., 1980). The activity of
ALAS is the rate-limiting step in the haem biosynthetic pathway.
Lead affects erythrocyte formation by impairment of globin and
haem synthesis. Globin synthesis is inhibited by lead in rat bone
marrow cells at concentrations as low as 1 µmol/litre (Dresner et al.,
1982). This is thought to be secondary to decreased protein synthesis
in erythroid cells as a consequence of lead-induced inhibition of haem
synthesis. Lead also decreases erythrocyte survival through its
inhibition of membrane bound Na-K ATPase (Rhagavan et al., 1981).
It has been shown in in vitro cultures of cells from liver,
bone marrow and the nervous system that ALAS activity may be increased
by addition of only 50 µmol lead/litre and ALAD activity is decreased
by 60% at a concentration of 0.5 µmol/litre (Kusell et al., 1978;
Dresner et al., 1982). Lead (5 µmol/litre) also inhibits (20%)
porphobilinogen deaminase in red blood cell haemolysates at
5 µmol/litre (Piper & Tephly, 1974). Fowler et al. (1980) have shown
that disruption of haem synthesis results in reduction of tissue haem
levels. Increased exposure to lead decreases the content and function
of haem-dependent enzymes of the P-450 mono-oxygenase system (Meredith
& Moore, 1979). More recently it has been shown that lead induces haem
oxygenase activity thereby increasing the degradation of haemoproteins
which may adversely affect a number of cell functions such as
respiration and energy production (Maines, 1992). It has been
suggested that delay in the accumulation of haemoproteins of the
respiratory chain in brain tissue during development may result in
decreased synthesis of haem enzymes in the brain (Bull, 1980; Moore et
al., 1987). Holtzman et al. (1981), on the other hand, found no effect
of lead on brain cytochromes in rat pups with impairment of growth due
to exposure to lead.
7.2 Nervous system effects
7.2.1 Higher order behavioural toxicity
Experimental studies using animal models have demonstrated that
lead impairs learning and memory functions at virtually all stages of
the life cycle. The most significant studies have focused on
behavioural and learning impairments in experimental animals with PbB
levels below 1.44 µmol/litre (30 µg/dl). Bushnell & Levin (1983) fed
post-weaning rats drinking-water containing 10 or 100 mg lead (as lead
acetate) per litre for up to 7 weeks. PbB was not determined but has
been estimated from comparisons with similar lead exposure models to
have been less than 0.96 µmol/litre (20 µg/dl) (Davis et al., 1990b).
Brain lead averaged about 0.05 µg/g wet weight. The rats exhibited
impaired learning ability when tested for their ability to choose
between alternate arms of a radial maze. Cory-Slechta et al. (1985)
demonstrated a significant learning impairment in rats given lead
acetate in drinking-water (25 mg/litre) from weaning. The observed
outcome was a significantly higher response rate in lead-exposed rats
working under a fixed interval schedule of food reinforcement. PbB
values of lead-treated rats in that study averaged
0.72-0.96 µmol/litre (15-20 µg/dl) and brain lead levels averaged
0.07 µg/g wet weight. Similar effects have been described in older
rats (16 months of age) at steady-state PbB levels of
0.62-0.86 µmol/litre (13-18 µg/dl) (Cory-Slechta & Pokora, 1991). More
recently, Cohn et al. (1993) demonstrated selective effects of lead on
learning processes, as distinct from non-specific effects such as
motivational levels, and sensory or motor impairment using a multiple
schedule of repeated learning and performance. Exposure to lead
acetate in drinking-water (50 mg/litre) produced a PbB of
1.2 µmol/litre (25 µg/dl).
Winneke et al. (1977) exposed rats to lead while in utero,
through mother's milk and directly in drinking-water. Their learning
ability was then tested by requiring the animals to discriminate
between stimuli of either different orientation (stripes as an easy
task) or different size (discs as a difficult task). Adult rats tested
between 90 and 170 days of age with PbB levels of less than
1.44 µmol/litre (< 30 µg/dl) were slower to learn the difficult task
of size discrimination (but not the easy discrimination problem) and
tended to repeat more errors than control subjects.
Altmann et al. (1993) demonstrated deficits of both active
avoidance learning (AAL) and in vitro hippocampal long-term
potentiation (LTP) in adult rats that had received pre-weaning or pre-
and post-weaning dietary lead exposure to achieve PbB levels of
approximately 0.72 µmol/litre (15 µg/dl) and brain lead concentrations
of 0.09-0.16 µg/g wet weight. Deficit in animals with only
post-weaning exposure and blood lead levels was about 0.77 µmol/litre
(16 µg/dl) but brain lead concentrations of only 0.09 µg/g was either
absent (in the case of LTP) or markedly reduced (in the case of AAL).
Similar types of effects have been noted in studies using
non-human primates. For example, Rice (1985) reported deficits of
discrimination reversal performance in monkeys dosed orally with 0, 50
or 100 µg lead/kg body weight per day for the first 200 days of life.
At the cessation of dosing, PbB concentrations were 0.144 µmol/litre
(3 µg/dl), 0.72 µmol/litre (15 µg/dl) and 1.2 µmol/litre (25 µg/dl),
respectively. Prior to behavioural testing (3 years of age), PbB
levels were 0.144 µmol/litre (3 µg/dl), 0.528 µmol/litre (11 µg/dl)
and 0.624 µmol/litre (13 µg/dl). Additional evidence for lead-induced
changes in learning in non-human primates is provided by the study of
Lilienthal et al. (1990), which showed a dose-related increase in the
percentage of errors in a learning set formation task (discrimination)
with PbB levels in the low exposure group (350 µg lead acetate/g diet)
averaging 1.68 µmol/litre (35 µg/dl). Also, an impairment of reversal
learning was found by Bushnell & Bowman (1979b) to persist in monkeys
up to their fifth year, at which time the PbB level in treated animals
averaged 0.24 µmol/litre (5 µg/dl) compared to 0.192 µmol/litre
(4 µg/dl) in controls. In the study by Bushnell & Bowman (1979a), the
period of lead exposure was limited to the first year of life and
resulted in PbB levels averaging as high as 3.12 µmol/litre
(65 µg/dl). No description was given of quality control procedures to
ensure accuracy of PbB determinations at the lower levels reported.
Several studies on experimental animals have shown perseverative
effects (the tendency to respond repetitively and inappropriately even
though environmental conditions have changed) which may underlie many
of the lead-induced changes in learning and other higher order
behavioural processes noted in such studies. This has been clearly
demonstrated in rodents by Cohn et al. (1993) in the context of
learning impairments. In the study by Bushnell & Levin (1983)
described above, the accuracy impairment derived from a tendency of
lead-exposed rats to re-enter a previously explored arm of the maze.
Similar perseverative tendencies have been described in non-human
primate studies, e.g., on delayed matching to sample (Rice, 1984a) and
delayed alteration tasks (Levin & Bowman, 1986; Rice & Karpinski,
1988).
Experimental animal studies also reveal the importance of task
complexity in detecting lead-induced changes in behaviour, both in
non-human primates (Levin & Bowman, 1983, 1986; Gilbert & Rice, 1987)
and rodents (Winneke et al., 1977, 1982b).
Persistence of lead-induced changes in some higher order
behavioural processes is also suggested by a number of experimental
animal studies. Several reports from the longitudinal studies of
non-human primates by Bowman and his colleagues (e.g., Bushnell &
Bowman, 1979a,b; Levin & Bowman, 1983, 1986, 1989) have demonstrated
persistent neurobehavioural deficits extending up to 8 years after the
termination of exposure and long after PbB levels had declined to the
control level. In rats, Cory-Slechta & Thompson (1979) showed that the
levels of lead exposure determine the persistence of lead effects,
while Cory-Slechta (1990a,b) revealed that apparently transient
effects of lead on behaviour could re-emerge with changes in the
reward contingencies of the environment. Munoz et al. (1986) found
deficits in both spatial and visual discrimination performance of rats
at several months of age resulting from pre-weaning exposure via the
dams. The same outcome was noted by Altmann et al. (1993) in a study
where deficits in adult rats were noted following pre-weaning
exposure.
7.2.2 Mechanisms of lead-induced behavioural toxicity
While the mechanisms underlying lead-induced behavioural toxicity
have yet to be adequately determined, the experimental animal
literature provides suggestive leads. The early experimental studies
of Pentschew (1965) and Pentschew & Garro (1966) demonstrated in the
rodent model that the pathogenesis of acute high-dose encephalopathy
was secondary to increased permeability of capillaries in the brain,
leading to leakage of fluid and red blood cells. These changes are
similar to those occurring in children with acute lead encephalopathy
characterized clinically by coma, convulsions and death, and identify
the brain microvasculature as the primary target for lead in the
central nervous system following high level exposure to lead.
Changes in microvascular morphology are not evident with low
level lead exposure, but it is the vulnerability of the blood-brain
barrier that permits exposure of the brain to lead. Exposure of the
developing fetus to lead results in higher uptake of lead in the brain
than from later exposures (Rossouw et al., 1987). The development of
resistance to acute encephalopathy in rat pups during the first few
days of life is thought by Holtzman et al. (1984) to be related to
maturation of the blood-brain barrier and possibly to the ability of
the older animals to sequester lead in protein complexes. Thus
neuropathological changes may relate more to the issue of exposure
than to mechanism of effect.
More recently, the focus of mechanistic studies has involved
biochemical and neurochemical changes. Biochemical changes in synaptic
transmission are likely to be related to problems in dendrite-nerve
organization and function (Goldstein, 1990). There is continuing
reorganization of dendrite and nerve terminal connections throughout
early months of neural development. A number of biochemical and
functional studies concerning possible mechanisms of lead effect
suggest lead may disrupt this process and its function through early
childhood (e.g., Cookman et al., 1987, 1988).
Other biochemical effects from lead exposure which may be the
basis of important neurobiological mechanisms of leadinduced
behavioural toxicity include changes in protein kinases, with
implications for neurotransmitter system disturbances. At least three
protein kinases present in nerve terminals involved in modulating the
release of neurotransmitters have been shown to be affected by lead.
However, the relevance of these findings to cognitive function has not
been established (Goldstein, 1990).
From both in vivo and in vitro studies, lead exposure has
been reported to have effects on virtually all neurotransmitter
systems (US EPA, 1986a). Depending on the stage of development, these
include dopaminergic, cholinergic, serotonergic, GABAnergic,
glutamatergic and opiate systems. Probably the most extensively
investigated system has been the dopaminergic system where a number of
changes at the biochemical and receptor level have been described. One
of the difficulties, however, is determining the relationship of the
reported changes at the biochemical/receptor level to changes in
behavioural function. McIntosh et al. (1989) noted lead-induced
changes in tetrahydrobiopterin metabolism which may be related to
changes in IQ scores (Blair et al., 1982; McIntosh et al., 1985). In
further support of a functional role for lead-induced changes in
dopaminergic systems, Cory-Slechta & Widzowski (1991) reported
alterations in both D1 and D2 dopaminergic sensitivity in rats with
PbB levels of 1.2-1.44 µmol/litre (25-30 µg/dl).
More recently, studies by Altmann et al. (1993) and Cohn &
Cory-Slechta (1993) suggest a role for changes in the N-methyl-D-
aspartate (NMDA) receptor complex in lead-induced behavioural
toxicity. The NMDA receptor complex has been implicated in the
learning and memory processes.
Effects of lead on mitochondrial energy metabolism may also be
important in the pathogenesis of neurological effects. Impairment of
respiration in brain mitochondria has been observed in in vitro
preparations (Holtzman et al., 1978) and in mitochondria isolated from
brains of lead-exposed rats (Gmerek et al., 1981).
7.2.2.1 Conclusions
Given their ability to establish causal relationships between
lead exposure and biological effects, experimental animal studies can
provide evidence supportive of human epidemiological findings. Many
experimental animal studies have been carried out to characterize the
nature of lead's effects on various target organ systems as well as to
establish the underlying mechanisms of effect. Furthermore, these
studies, by their very nature, are not confounded by such co-variates
of children's IQ as parental IQ, socioeconomic status and quality of
the home environment, or complicated by nutritional inadequacies of
the study population. Earlier experimental animal studies tended to
utilize relatively high lead exposure levels, but over the past ten
years, exposure levels and protocols have been more relevant to the
human situation.
There are, of course, some limitations of experimental animal
studies that must be considered. For example, there may be differences
in species sensitivity as well as differences in pharmacokinetic and
pharmacodynamic behaviour of lead in rodents, primates and humans.
Higher external exposure levels of lead are required to produce PbB
levels corresponding to levels experienced in human populations, and
higher PbBs are necessary to induce encephalopathy in rodents than are
associated with such effects in humans. Such differences in
sensitivity obviously reflect, at least in part, differences in
kinetics of lead across species. Despite these differences, it should
be noted that corresponding levels of PbB have been associated with
neuro-behavioural toxicity in both animal models (rodents as well as
non-human primates) and human populations. This comparability of
effect levels, despite the relative insensitivity of the animal
models, suggests that the levels of concern in humans might be even
lower, although quantitative extrapolation is difficult. Although one
cannot presume that the neurotoxic effects of lead in experimental
animals necessarily predict specific effects in humans, the
similarities in neurobehavioural end-points in humans and animals are
sufficient to conclude that neurobehavioural deficits in animals are
at least qualitatively predictive of effects in humans.
7.2.3 Sensory organ toxicity
Visual and auditory functions have been shown to be affected by
lead; other sensory modalities have largely been neglected. Bushnell
et al. (1977b) studied visual acuity in infant rhesus monkeys using
different degrees of illumination. Four control animals and three
animals each in a low and a high lead group were tested. The animals
were orally dosed between postnatal days 5 and 365. Although the
dosing regimen of this group was intended to produce blood lead levels
around 4.08 µmol/litre (85 µg/dl), peak levels of between 6.58 and
14.4 µmol/litre (137 and 300 µg/dl) occurred between the fifth and the
ninth weeks of dosing. Significant impairment of scotopic vision was
observed only in the animals of the high lead group.
Using flash-evoked visual potentials (VEP), Lilienthal et al.
(1986) studied visual function in 7- to 7´-year-old rhesus monkeys
pre- and postnatally exposed to lead acetate (0, 350 and 600 mg/kg) in
their diets. Seventeen animals were tested altogether: six controls,
and five or six in each of the exposed groups. PbB levels fluctuated
around 1.92 µmol/litre (40 µg/dl) in the group fed 350 mg/kg diet
during the seven postnatal years, whereas in the group fed 600 mg/kg
they had declined from the initial average of 5.28 µmol/litre
(110 µg/dl) at age 9 months to about 2.88 µmol/litre (60 µg/dl) at 7
years of age. PbB levels in the mothers of both these groups had been
between 1.15 and 1.78 µmol/litre (24 to 37 µg/dl) during pregnancy. A
dose-related decrease of amplitudes and a similar increase of
latencies for the main VEP component were found; these were, in most
instances, significant in both the high and low lead groups.
These findings are supported by the results of studies on rodents
by Fox et al. (1977). Prolonged VEP latencies were observed following
chronic developmental lead exposure. Lower VEP amplitudes have also
been observed in rats at PbB levels exceeding 1.44 µmol/litre
(30 µg/dl) (Winneke, 1979). in vitro studies suggest that rods are
more sensitive to the effects of lead than are cones (Fox & Sillman,
1979).
Information on the impairment of auditory function by lead comes
from electrophysiological studies using brainstem auditory evoked
potentials (BAEP) in rhesus monkeys (Lilienthal et al., 1990). PbB
levels in the various groups at the time of testing averaged
0.432 µmol/litre (9 µg/dl; controls), 1.92 µmol/litre (40 µg/dl; low
lead), and 2.688 µmol/litre (56 µg/dl; high lead). Of the four peaks
discernible in the monkey, BAEP peaks II and IV exhibited
significantly prolonged latencies, which were observed only in the
animals of the high lead group.
7.3 Renal system
The renal effects of lead in animal models occur as a result of
both acute and chronic exposures. Acute lead nephrotoxicity is
characterized by decreased reabsorption of small molecular weight
compounds by the renal tubule, particularly amino acids, glucose and
phosphate (Fanconi Syndrome).
Morphological changes of chronic lead exposure include
cytomegaly, development of nuclear inclusion bodies and
ultrastructural changes in mitochondria. The basis for the cytomegaly
is not understood but it is known that there is altered homoeostasis
of water and electrolytes and cellular swelling. Inclusion bodies are
lead-protein complexes composed of acidic non-histone proteins. As
much as 90% of lead in the kidney has been shown to be contained in
the inclusion bodies, suggesting that they provide a detoxification
function. The origin of the protein is not known but Egle & Shelton
(1986) have identified the most abundant component of isolated
inclusion bodies to be a constitutive protein of the adult central
nervous system primarily in the cerebral cortex. The lead in the
inclusion bodies is chelatable with EDTA (Goyer & Wilson, 1975).
Mitochondria isolated from kidneys of lead-intoxicated rats have
impaired respiration and oxidative phosphorylation capacity, (Goyer &
Rhyne, 1973).
Kholil-Manesh et al. (1992) studied the evaluations of renal
histological and functional changes in rats continuously dosed with
high dose lead. Glomerular filtration rate (GFR) was significantly
increased at 3 months, but significantly decreased at 12 months. Lead
inclusion bodies were noted throughout the duration of the study.
Tubular atrophy and intestinal fibrosis appeared at 6 months. The
brush border of proximal tubular cells was disrupted at 1 and 3
months, but recovered later.
With continued exposure to lead, acute nephropathy may progress
to chronic interstitial nephritis that does not have any unique or
distinguishing features. There is progressive increase in interstitial
fibrosis, dilatation of tubules and formation of microcysts with
hyperplasia of tubular epithelial cells. In the rat, inclusion bodies
are reduced in number and may be entirely absent in later stages of
the nephropathy. Glomerular sclerosis occurs with onset of proteinuria
and renal failure (Goyer & Rhyne, 1973).
The sequence of morphological changes observed in experimental
models is thought to be generally true for humans. Experimental
studies in rats suggest that there may be a threshold for lead renal
toxicity in the rat at PbB levels similar to those observed for
humans. In rats exposed to different doses of lead for up to 12 weeks,
a PbB level of 2.88 µmol/litre (60 µg/dl) appears to be the threshold
for proximal renal tubular cell injury by lead (Goyer et al., 1989).
This PbB level is equivalent to kidney lead levels of about 45 mg/kg
wet weight and is the level of lead at which excretion of renal
calcium increases and ultrastructural changes occur in proximal
tubular cell mitochondria. This finding is consistent with the
observation in humans by Buchet et al. (1980) and Gennart et al.
(1992) of a threshold value of 2.88 µmol/litre (60 µg/dl) of PbB which
leads to adverse renal effects by lead.
7.4 Cardiovascular system
Early studies concerning the production of hypertension in
experimental animals, summarized in Environmental Health Criteria 3:
Lead (IPCS, 1977), were conflicting. Among rats given 70 mg of lead
acetate per day orally, only a few survived 40 days and all were
hypertensive (Griffith & Landaver, 1944). On the other hand, other
studies did not reveal a blood pressure effect from high-level lead
exposure in rats (Padilla et al., 1969) or in dogs (Fouts & Page,
1942). More recently, several experimental studies have confirmed that
lead can produce hypertension, and evidence for several plausible
mechanisms has been provided. Rats, both normotensive and
spontaneously hypertensive, exposed to lead in water supplies for up
to a year, resulting in PbB levels of up to 0.6 µmol/litre
(12.5 µg/dl), exhibited ventricular tachycardia and ventricular
fibrillation (Evis et al., 1985). Subsequent studies (Evis et al.,
1987) showed that adrenaline had a significant arrhythmogenic effect
in hypertensive animals. It was concluded that chronic exposure to
lead, when combined with high blood pressure, slightly enhances the
susceptibility of the heart to arrhythmias induced by myocardial
ischaemia. Overviews of the experimental evidence related to
hypertension and lead exposure have been published by Victery (1988)
and US EPA (1986a, 1989). Taken as a whole, these studies demonstrate
that increase in blood pressure does occur secondary to renal failure
in rats with continuous high-level exposure to lead. More recent
studies on effects of chronic low-level exposure to lead on blood
pressure in rats have shown alterations in cardiovascular parameters
in the PbB range of 0.24-1.92 µmol/litre (5-40 µg/dl), and provide
some insights as to mechanisms involved in the pathogenesis of
lead-related cardiovascular effects.
Victery et al. (1982a) exposed rats to lead in utero by giving
dams drinking-water containing 0, 5, and 25 µg lead/ml and continued
this regimen to the pups for 5 to 6 months. Although no change in
blood pressure was noted at these levels of exposure, significant
changes in the renin-angiotensin system were reported in animals given
water containing 25 µg lead/ml with a PbB level of 0.864 µmol/litre
(18 µg/dl). In a similar study (Victery et al., 1982b), exposure to
100 µg lead/ml in drinking-water (but not 500 µg/ml) produced a
significant (17 mmHg) elevation in blood pressure beginning at 3´
months of age and continuing until 6 months of age. Chai & Webb (1988)
found an elevation of 15 to 20 mmHg in the systolic blood pressure of
rats given drinking-water containing 100 µg lead/ml. These authors
suggest that alterations in the cellular mechanisms that regulate
intracellular calcium concentration may enhance pressor responsiveness
to catacholamines. Boscolo & Carmignani (1988) found raised blood
pressure in rats with exposure to drinking-water containing 0, 30 or
60 µg lead/ml for 18 months. Cardiovascular responses to blood
pressure agonists indicated that lead exposure affects the
renin-angiotensin system and induces sympathetic hyperactivity by
acting on central and peripheral sympathetic junctions, increasing the
responsiveness to stimulation of alpha-2-adrenergic receptors, and by
increasing sensitivity to stimulation of cardiac and vascular
beta-adrenergic and dopaminergic receptors.
From the experimental evidence summarized by Victery (1988), it
appears that low-level exposure to lead produces an elevation in blood
pressure. The failure to demonstrate increased blood pressure in some
studies with high-level exposure to lead suggests that the effect of
lead on blood pressure may be biphasic, i.e. a consistent effect with
low-level exposure but inconsistent effects with high-level exposure.
7.5 Reproductive system
Experimental studies on the effects of lead on the reproductive
system most often concern toxicity to either the male or female but
have addressed results of exposure to both parents. Environmental
Health Criteria 3: Lead (IPCS, 1977) identified effects on
spermatogenesis in rats exposed to lead and showed that high maternal
exposure to lead in rats can reduce numbers and size of offspring.
There may also be paternally transmitted effects resulting in
reductions of litter size, weights of offspring and in survival rate.
Few studies into the effects of lead on male sexual function have
reported PbB levels. Ivanova-Chemishanska et al. (1980) reported
changes in levels of enzymatic activity and ATP in testicular
homogenates from rats given 0.0001 or 0.01% solutions of lead acetate
as drinking-water over a 4-month period. Chowdhury et al. (1984) found
testicular atrophy along with cellular degeneration in rats with PbB
levels over 3.36 µmol/litre (70 µg/dl), but not in rats with levels of
2.59 µmol/litre (54 µg/dl). Donovan et al. (1980) found that lead
inhibited androgen binding by cytosolic receptors in mouse prostate.
Testicular homogenates from 2- to 3-week-old male offspring (PbB
levels 0.30 µmol/litre, 6.3 µg/dl) of lead-exposed female rats showed
a decreased ability to metabolize progesterone (Wiebe et al., 1982).
In an in vitro study, Wiebe et al. (1983) found a 10 to 20% decrease
in FSH binding and in the production of cyclic AMP by Sertoli cells
isolated from prepubertal rats and cultured in the presence of lead
acetate (2.64 × 10-4 mol/litre). In addition, the activity of
cellular 3ß-hydroxysteroid dehydrogenase was decreased. It was shown
by Sokol et al. (1985) that there is a dose-related suppression of
serum testosterone levels and spermatogens in adult rats (100 day of
age) given a solution of 0.3% sodium acetate as drinking-water for up
to 60 days. PbB concentrations were between 1.44 and 2.40 µmol/litre
(30 and 50 µg/dl), depending on the length of treatment. Further
studies (Sokol, 1987) supported the hypothesis that lead disrupts the
hypothalamic control of pituitary hormone secretion. However, other
evidence indicates that lead may directly or indirectly affect
testicular enzymes or may act indirectly by a reduction in testicular
binding of FSH and production of cyclic AMP (US EPA, 1986a).
Dosing mature female rats with lead in order to produce PbB
concentrations of 1.44 µmol/litre (30 µg/dl) resulted in irregular
estrous cycles. At a PbB level of 2.54 µmol/litre (53 µg/dl), animals
developed follicular cysts and there was a reduction in the number of
corpora lutea (Hilderbrand et al., 1973). Grant et al. (1980) reported
delayed vaginal opening in rats whose mothers were given drinking-
water containing 25, 50 and 250 µg lead/ml. The vaginal opening delays
in the group given 25 µg lead/ml of drinking-water occurred in the
absence of any growth retardation or other developmental delays and
were associated with median PbB levels of 0.86-1.39 µmol/litre
(18-29 µg/dl). Studies on female monkeys have shown that pre- and/or
postnatal exposure to lead can affect pubertal progression and
hypothalamicpituitary-ovarian-uterine functions. Chronic exposure of
nulliparous female monkeys to lead (PbB levels of approximately
1.68 µmol/litre, 35 µg/dl) resulted in subclinical suppression of
circulating luteinizing and follicle stimulating hormone and estradiol
without producing overt effects on general health or menstruation
(Foster, 1992).
7.6 Effects on bone
There is growing interest in lead in bone for several reasons.
Bone is a store for lead accumulated from past exposure. It has a long
biological half-life but may be mobilized and contribute to blood lead
during pregnancy. With the development of X-ray fluorescence
techniques to measure lead in bone in vivo, there is a need to
improve our understanding of bone lead metabolism and of factors that
influence lead retention and release. In addition, lead may adversely
affect bone metabolism, particularly in post-menopausal women, and
contribute to the development of osteoporosis. There has only been
limited experimental study of these concerns to date, but there are
some relevant reports in the literature.
A summary of much of the currently available literature on the
potential toxicological implications of lead in bone during pregnancy
and lactation is contained in a review by Silbergeld (1991). Toward
the latter half of pregnancy in mice, there seems to be a preferential
transfer of lead across the placenta to the fetus (Danielson et al.,
1983). In rats exposed to lead for 150 days and then not exposed for
50 days prior to mating, Buchet et al. (1977) found that there was a
substantial mobilization of lead from mother to fetus. Keller &
Doherty (1980b) found using radiotracer lead (210Pb) in female mice
that there was also a major transfer of lead to the pup during
lactation. The concomitant decrease in maternal bone lead supports the
hypothesis that bone resorption of lead occurs during lactation.
It has been shown that lead may directly and indirectly affect
various aspects of bone metabolism (Pounds et al., 1991). Lead
inhibits the renal enzyme 1-hydroxylase, reducing plasma levels of
1,2-dihydroxychole-calciferol (activated vitamin D). Lead impairs the
Haversian remodelling system in beagle dogs chronically exposed to
lead (Anderson & Danylchuk, 1977).
7.7 Immunological effects
The effects of lead on the immune system are diverse but not well
documented. Lead reduces resistance and increases mortality of
experimental animals when they are infected by a broad range of
bacterial and viral agents (Koller, 1984). Lead impairs antibody
production in animals and generally decreases immunoglobin plaque-
forming cells (Koller & Roan, 1980).
7.8 Mutagenicity
Lead is thought to have genotoxic properties. However, lead-
induced gene mutations in cultures of mammalian cells have only been
observed at concentrations toxic to the cells. Studies for point
mutations in bacterial systems have also yielded negative results (US
EPA, 1986a). Zelikoff et al. (1988) found that both insoluble lead
sulfide and more soluble lead nitrate were mutagenic when added to
Chinese hamster V79 cells. A 6-fold increase in mutation frequency was
noted at a lead nitrate level of 500 µmol/litre medium. These authors
also found that lead acetate induced morphological transformation of
Syrian hamster cells. However, they concluded that these effects may
not have been the result of direct damage to DNA but may have occurred
via indirect mechanisms including disturbances in enzyme functions
important in DNA synthesis and/or repair.
Studies on the production of chromosome aberrations, sister-
chromatid exchanges and micronuclei by lead, whether in in vitro
cultures or in vivo, have given mixed results, and summaries are
available (US EPA, 1986a; IARC, 1987a,b; ATSDR, 1991).
7.9 Carcinogenicity
There have been several experimental studies in rats and mice in
which long-term administration of a lead compound in food or
drinking-water or parental administration has produced tumours of the
kidney (Van Esch & Kroes, 1969; Moore & Meredith, 1979). These and
other studies have been discussed in detail in IARC (1980) and
summarized in US EPA (1986a). In general, methodological problems,
including dose levels, number of animals, and doses, and lack of
toxicity monitoring, make many of the studies difficult to interpret
in a quantitative manner. One study (Azar et al., 1973) addressed some
of these problems and reported an increase in the numbers of renal
tumours in rats fed diets containing 500 mg lead/kg for 2 years. PbB
levels were 3.84 µmol/litre (80 µg/dl). In all studies renal
carcinogenicity occurred against a background of proximal tubular cell
hyperplasia, cytomegaly and cellular dysplasia in response to the high
doses of lead and long exposure times (Goyer, 1985). Renal
adenocarcinoma occurred in a high percentage of lead-exposed animals
and the incidence of tumours was related to the length and severity of
exposure (Mao & Molnar, 1967). Males appear to be more susceptible to
tumours than females. The maximum dose of lead in drinking-water, not
associated with any morphological or functional evidence of renal
toxicity in rats fed a diet containing adequate levels of trace
minerals, particularly calcium, is 200 mg lead/litre (Goyer et al.,
1970). No evidence of renal tumours at doses below this value has been
reported (Azar et al., 1973; US NCI, 1979).
Several hypotheses have been proposed for the mechanism of lead
carcinogenicity in experimental animals. These include mutagenicity,
cellular proliferation, nuclear protein (inclusion bodies), promoter
activity, activation of protein kinase C, and cystic hyperplasia
(Goyer, 1993). Lead is a weak mutagen in mammalian cell systems, but
is a strong mitogen. Exposure to a single intraperitoneal injection of
lead acetate in rats stimulates a 40-fold increase in cell
proliferation as measured by autoradiography, and this is further
increased by unilateral nephrectomy (Choie & Richter, 1972). DNA
synthesis in kidneys, as measured by 3H-thymidine incorporation, is
increased 15-fold and the mitotic index 45-fold following a single
intracardiac injection of lead acetate in mice (Choie & Richter,
1974). Cell proliferation and hyperplasia are seen in the liver of
rats given a single intravenous injection of lead nitrate (Columbano
et al., 1984). In both of these studies, increases in cell
proliferation occurred in the absence of cellular necrosis, suggesting
that this was a mitogenic rather than regenerative response.
Activation of protein kinase C and formation of nuclear inclusion
bodies or lead protein complexes are additional events that may
influence regulation of cell growth and development and play a role in
the carcinogenic response in experimental animals resulting from
administration of lead (Goyer, 1993). Cystic hyperplasia, a late
morphological manifestation of chronic lead nephropathy, is a risk
factor for renal cancer (Bernstein et al., 1987). Prior to adenoma
formation in animals treated with renal carcinogens, cystic
hyperplasia was reported (Dees et al., 1980; Goyer et al., 1981).
8. EFFECTS ON HUMANS
Despite the long recognition of lead poisoning, new clinical
cases continue to be the subject of published reports. Although
important, they affect a comparatively small proportion of the
population at risk from the potential effects of exposure to
environmental lead.
Over the last 10-15 years, particular attention has been directed
towards epidemiological studies designed to evaluate the possible
neurotoxic effects of lead on the developing child, especially delayed
or impaired neurobehavioural development and performance.
In the adult population, considerable attention has been directed
towards evaluating cardiovascular effects and the implication of lead
in hypertension.
New studies have contributed to our understanding of the
biochemical effects of lead, and may facilitate early recognition of
significant change and mitigation of potentially adverse outcomes.
The effects of lead have been studied widely in both the general
population and in those exposed occupationally. Since these effects
are the same in both settings, no distinction has generally been made
in the discussion. However, it is often important to distinguish
between adults and children because of different susceptibility.
8.1 Biochemical effects of lead
Lead is known to affect a number of enzymes and physiological
systems which result in a wide variety of changes in humans. While
those affecting the haematopoietic system are well known, there are
others which need to be considered in the risk assessment process and
are considered in this section.
In considering the effects of lead on biochemical systems, it is
appropriate to discuss the form of the lead in the various body
compartments.
Blood lead (PbB) is distributed between the plasma and the
erythrocyte. There is less than 1% in the plasma for PbB levels of up
to 4.8 µmol/litre (100 µg/dl) (Manton & Cook, 1984). The curvilinear
relationship of serum lead to blood lead is shown in Fig. 12. The data
show that the erythrocytes have a capacity to bind lead up to PbB
levels of about 2.4 µmol/litre (50 µg/dl). Above this level a fairly
rapid increase in the serum levels occurs.
It should be noted that the proportion of "free" (i.e. unbound)
lead in blood is important in relation to biological activity. Lead is
bound to haemoglobin in blood and has a greater affinity for fetal
than adult haemoglobin (Ong & Lee, 1980). It may, therefore, be
5.2.2.3 Alcoholic beverages
Contamination of alcoholic beverages with lead may occur in
several way. For example, lead solder used to repair casks or keg and
tap lines from lead capsules used as seals or from residues of lead
arsenate pesticides in soils now used to grow grapes. Alcoholic
beverages tend to be acidic and there is the possibility that large
amounts of lead can be dissolved during preparation, storage or
serving (Wai et al., 1979). Published reports on lead levels in wine
show that important variations occur from sample to sample (Jorhem et
al., 1988). The US Department of the Treasury (1991) analysed 432
table wines sold within the USA. The results are summarized in Table
17.
Sherlock et al. (1986) found that the majority of canned and
bottled beer (90 and 86% respectively) contained less than 10 µg
lead/litre. Draught beers typically contained higher lead
concentrations with 55% having lead concentrations greater than
10 µg/litre, 16% with concentrations over 20 µg/litre, and 4% with
concentrations over 100 µg/litre. The higher lead concentrations in
draught beers are considered most likely due to the draught-dispensing
equipment which sometimes contains brass or gunmetal, both of which
contain low but significant amounts of lead (Sherlock et al., 1986).
In general, alcoholic beverages do not appear to be a significant
source of lead intake for the average person.
Table 17. Distribution of lead in table wines in USAa
Range Number of Percentage of total
(µg/litre) samples samples analysedb
0-10 36 8.3
11-25 62 14.4
26-50 105 24.3
51-100 144 33.3
101-250 64 14.8
251-500 12 2.8
501-673 9 2.1
a From: US Department of the Treasury (1991)
b In all, 432 samples were analysed.
5.2.3 Dust and surface soils
5.2.3.1 Dust
Dust is a significant source of exposure to lead, particularly
for young children (see Fig. 11), as has been demonstrated in several
studies correlating children's blood lead concentrations with dust
lead levels (Rabinowitz et al., 1985; Bornschein et al., 1987; Davies
et al., 1987a; Laxen et al., 1987; Steenhout, 1987).
The major contributions to lead levels in soil and outdoor dust
are from the combustion of fossil fuels (principally leaded petrol),
stationary sources such as smelters, and peeling and flaking of
lead-based paint. Typical lead levels in road dust in the USA are
800-1300 mg/kg in rural areas to 100-5000 mg/kg in urban areas (US
EPA, 1989c).
Concentrations of lead in household dust vary greatly between
different dwellings and areas of the world. Mean concentrations of
300-2500 mg/kg have been found in the United Kingdom and USA, but
individual samples may be in the range of 10 000 to 30 000 mg/kg (Que
Hee et al., 1985b; Clark et al., 1985; Bornschein et al., 1986; Raab
et al., 1987).
Flaking lead-based paint, paint chips, and weathered powdered
paint markedly increase intake of lead from surface dust, particularly
for urban children with pica (US EPA, 1986a; Bornschein et al., 1986).
Lead-based paint chips have been found to contain 1000-5000 µg
lead/cm2 (Billick & Gray, 1978). When lead-based paint is present,
interior renovation activities greatly increase household dust lead
concentrations (Laxen et al., 1987). Improved control of dust and
surface clean-up after lead-based paint removal have been shown to
reduce lead exposure of children reoccupying affected houses (Charney
et al., 1983).
5.2.3.2 Soil
In rural and remote areas, lead in soil is derived mainly from
natural geological sources. These natural sources account for 1-30 mg
lead/kg, but where soils are derived from leadmineralized rocks,
natural concentrations may range from several hundred to several
thousand mg/kg.
Typical values for lead in rural soils in the United Kingdom are
15-106 mg/kg with a geometric mean of 42 mg/kg (Davies, 1983). A
geometric mean of 48 mg/kg for 2780 samples has also been reported
(McGrath, 1986).
Concentrations of lead in urban soil vary greatly. In the USA, a
study of city parks recorded concentrations of 200 to 3300 mg/kg (US
EPA, 1989). Concentrations of up to 10 960 mg/kg have been reported
for urban garden soils in the USA (Mielke et al., 1984), and up to 14
100 mg/kg in the United Kingdom (Culbard et al., 1988). Concentrations
can exceed 20 000 mg/kg around lead mining and processing operations
(Culbard et al., 1988). In areas where lead-based paint has been used,
soil samples taken near building foundations have been reported to be
as high as 20 000 mg/kg (Schmitt et al., 1988; Krueger & Duguay,
1989).
In general, lead concentrations in soils near roads are high
where road traffic density is high. Concentrations decrease
exponentially with distance from the road (IPCS, 1989).
Continuous application of sewage sludge results in an
accumulation of lead in soil. For example, soil receiving heavy
applications over a long period was found to contain 425 mg/kg,
compared with 47 mg/kg in an untreated soil (Beckett et al., 1979).
5.2.3.3 Migration of lead from food containers
The available data on the daily intake of lead by adults and
children indicate a general decrease in those areas where the level of
lead in petrol has decreased and a concerted effort made to avoid
lead-soldered cans for food storage (OECD, 1993). However, in many
regions of the world, lead can migrate from food storage and serving
vessels such as lead-soldered cans (see section 5.2.2.2), ceramic
dishes, pottery vessels, crystal glassware and decals on food wrap
and/or dishes. Acidic foods tend to leach more lead. However, certain
foods such as corn and beans are associated with greater release of
lead than would be predicted from their acidity alone (Bolger et al.,
1991). Oxygen appears to accelerate the release of lead from food
containers.
If foods are stored in ceramic or pottery dinnerware that was
lead-glazed and fired in a low temperature kiln, lead can migrate from
the pottery glaze into the food. The glazing process uses a flux,
which is a material that, at high temperatures, reacts with and helps
dissolve the components of the glaze. Lead oxide is a commonly used
flux. Factors that determine whether and to what extent lead will
migrate include the temperature and extent of firing of the pottery in
the manufacturing process, temperature and duration of food storage,
and the acidity of the food. It is extremely difficult to quantify the
extent of such exposures in view of the variations in the
manufacturing processes and the quality control practised in the
country of origin. However, the extent of exposure can be quite
significant, particularly among infants. Cases of lead intoxication
from this source have been reported (Wallace et al., 1985).
Lead has been found to migrate from lead crystal glass into
beverages. This problem is especially severe if beverages are stored
in lead-crystal containers, e.g., decanters or liquor bottles (de
Leacy, 1987; Graziano & Blum, 1991). This phenomenon was not observed
with borosilicate glass containers (de Leacy, 1987).
Several studies have been made of lead contamination of foods and
beverages from lead used in the manufacture or repair of metal
vessels. Recoating the inner surface of brass utensils with a mixture
of lead and tin, described as "tinning", is widely practised by
artisans in India (Vatsala & Ramakrishna, 1985). The tin-lead alloy
contains 55 to 70% lead. Water containing tamarind contained
400-500 µg lead/litre after 5 min of boiling. The practice is
considered to be widely prevalent in at least the three southern
states of India. Zhu (1984) described 344 cases of chronic lead
poisoning in Jiansu Province, China, involving people who had drunk
rain water boiled in tin kettles. Analysis of the lead content of
water showed that after boiling the water contained 0.79 to
5.34 mg lead/litre.
5.3 Miscellaneous exposure
5.3.1 Cosmetics and medicines
Some traditional medicines and customs have been found to result
in exposure to high levels of lead, most of which cannot be quantified
with any degree of accuracy. Rather than occurring as trace
ingredients or trace contaminants, various lead compounds are used as
major ingredients in traditional medicines in numerous parts of the
world (Table 18). Clinically overt lead poisoning due to traditional
cosmetics and medicines has been identified among infants (Shaltout et
al., 1981; Fernando et al., 1981; Sharma et al., 1990), children and
adults (Pontifax & Garg, 1985; Cueto et al., 1989; Mitchell-Heggs et
al., 1990; Gupta et al., 1990). There are case reports of lead
toxicity secondary to inhalation of lead from traditional remedies
(Aslam et al., 1979; Shaltout, 1981; Cueto et al., 1989; Sharma et
al., 1990; Mitchell-Heggs et al., 1990).
Often the use is not limited to adults; these may be used on
infants and young children, as well as on women. In Kuwait, the leaded
"kohl", also called "Al kohl", is traditionally applied to the raw
umbilical stump of the newborn in the erroneous belief of a beneficial
astringent action (Fernando et al., 1981). An additional use of lead
metal and lead sulfide is for inhalation of the fumes ("Bokhoor")
produced from heating on hot coals, in the mistaken belief that this
will calm irritable infants and children (Fernando et al., 1981;
Shaltout et al., 1981).
Latin-American countries also report the use of traditional
medicines with high lead concentrations. For example, the Mexican
traditional remedy "azarcon" (lead chromate) and/or "greta" (mixed
lead oxides), distributed as finely ground powders, may contain more
than 70% lead. They are used in the treatment of "empacho", a
gastrointestinal disorder considered to be due to a blockage of the
intestine (Trotter, 1990).
In addition to the potential risks of lead exposure from the use
of traditional medicines, clinical lead poisoning can result from the
lodging of lead shot in vivo (Manton & Thal, 1986).
Table 18. Sources of lead exposure in traditional medicines and cosmetics
Source of lead Comments Reference
(product)
Summa/Kohl used in Indo-Pakistan and other Muslim Aslam et al. (1979);
cultures as eyes preparation; placed on Fernando et al. (1981);
conjunctival surface or as astringent on Shaltout et al. (1981);
umbilical cord stump. Antimony originally Sharma et al. (1990)
used but lead cheaper.
Hindu folk ground seeds and roots as treatment Pontifax & Garg (1985)
medicine for diabetes (8 mg lead/g)
Bokhoor tribal custom to produce lead fumes to Shaltout et al. (1981)
ward off evil
Azarcon lead chromate and mixed lead oxides as Trotter (1990)
treatment for gastrointestinal disorders
in Mexico and southwestern USA
Skin ointments cosmetics used by Chinese actors; skin Lai (1977)
and cosmetics ointment in Europe
5.4 General population exposure
The total intake of lead by adults and children in the general
population varies greatly as to the relative contributions from
individual sources (air, water, food, soil/dust and others) and is
partly dependent on life-style and socioeconomic status. It is beyond
the scope of this review to provide comprehensive information covering
a wide range of circumstances. However, a few simplified calculations
will be given as guidance for carrying out such determinations.
Table 19 gives a summary of the total lead intake and uptake from
the general environment in adults and in children aged 1 to 5. The
assumptions made are shown and are taken from WHO (1987). Additional
intake of lead will take place in certain groups from the use of
tobacco and alcoholic beverages. The estimates given are probably on
the high side with respect to the contribution from air and dust,
since indoor and outdoor lead contributions were considered equal.
Additional intake of lead is possible due to ciliary clearance of
particles 1-5 µm in diameter with subsequent swallowing and
gastrointestinal absorption.
5.5 Blood lead concentrations of various populations
Under certain conditions, blood lead (PbB) levels are a useful
indicator of exposure and are therefore discussed here, as well as in
section 6.1.4. In general, PbB levels correlate better with recent
exposure levels (Lyngbye et al., 1990b). As is discussed in sections
5.5.1 and 5.5.2, the general trend observed in all blood lead surveys
carried out in countries engaged in risk reduction programmes over the
last 15 to 20 years is a fall in the measured levels (OECD, 1993).
PbB concentrations are the most often used estimate of general
exposure to lead. The USA, Commission of European Communities (CEC),
Australia and the WHO have carried out epidemiological surveys to
determine lead exposures in various populations. The designs of these
studies differed; some provided estimates for the country as a whole,
others compared PbB levels of people working in the same occupation in
various countries, and others surveyed general populations but were
not designed to be extrapolated as national estimates. What these
studies have in common is that they did not emphasize groups with high
lead exposures, but concentrated on typical PbB concentrations of
non-occupationally-exposed groups in the regions or countries studied.
These surveys also offer the possibility of examining the data as
distributions, especially at the high end of the distribution profile.
Such examination may identify unusual exposures and thus populations
at risk. Average PbB concentrations may disguise the risk to various
segments of the population.
In the USA, national estimates of the extent and severity of
recent human exposures to lead in the general population were based on
PbB measurements from the second National Health and Nutrition
Examination Survey of 1976-1980 (NHANES II) (Annest & Mahaffey, 1984).
The design of this study permitted extrapolation to the USA population
as a whole.
Table 19. Estimates of lead (µg/day) absorbed by adults and children
from air, dust, food and watera
Mean air Dust Source of lead (µg/day) Total
lead intake absorbed
concentration (mg/day)b (µg/day)
(µg/m3) Air Dust Food Water
Adults
0.3 N.S. 2.4 -- 10 2 14.4
0.5 N.S. 4.0 -- 10 2 16.0
1.0 N.S. 8.0 -- 10 2 20
2.0 N.S. 16.0 -- 10 2 28
Children 1-5 years
0.3 -- 0.6 -- 25 5 30.6
0.5 -- 1.0 -- 25 5 31.0
1.0 -- 2.0 -- 25 5 32.0
2.0 -- 4.0 -- 25 5 34.0
1.0 25 2 12.5 25 5 44.3
1.0 50 2 25.0 25 5 57.0
1.0 100 2 50.0 25 5 82.0
1.0 200 2 100.0 25 5 132.0
a Adapted from WHO (1987); Dust is not considered a significant
source of lead in adults, but is a significant source for workers
where hygiene practices are poor
The above estimates are based on the following assumptions:
Air: Respiratory volume in adults is 20 m3/day, and in children
5 m3/day, and the respiratory absorption is 40%.
Food: Intake of lead by adults 100 µg/day with 10% absorption and
50 µg/day for children with 50% absorption.
Water: A lead concentration of 20 µg/litre, with adult consumption
of 1 litre/day and 10% absorption and for children 0.5 litre/day
with 50% absorption.
Dust: Dust concentration of lead was 1000 µg/g and absorption was
50%.
b N.S. = Not significant
The CEC study was directed toward biological screening of the
extent of exposure to lead outside the workplace using PbB level as
the index of exposure (CEC, 1981). The object of the study was to
assess non-occupational exposure of the population to lead in the
member states; it was implemented by member states and coordinated by
the CEC. Specific populations studied were selected by member states
and 168 separate areas and population groups were investigated in
1977.
Two other major international studies were limited to urban
populations. A pilot study of human lead exposure (Friberg & Vahter,
1983), organized by the United Nations Environment Programme and WHO,
was a collaborative effort under the Global Environmental Monitoring
System (GEMS). The following countries participated in at least part
of the study: Belgium, China, India, Islamic Republic of Iran, Israel,
Japan, Mexico, Peru, USA and the former Yugoslavia. The subjects of
the study were teachers, since they comprise an occupational group not
extensively exposed to lead. An Australian study was based on urban
residents only, and included 651 subjects between 6 and 91 years of
age (Hopper et al., 1982).
Based on demographic, economic and individual variables found in
the USA study to be associated with PbB levels (Mahaffey et al.,
1982), it is clear that results from these major studies cannot be
directly compared. However, a comparison of such studies can provide
information on the exposure to lead of segments of the various
populations in various countries worldwide.
5.5.1 Adult populations
In a large health screening programme within the USA during
1976-1980 (NHANES II), over 27 000 residents aged 6 months to 74 years
were examined (Annest & Mahaffey, 1984). PbB concentrations were
determined in a subsample (9933 individuals) and yielded an arithmetic
mean value of 0.67 µmol/litre (13.9 µg/dl). In 5841 individuals aged
18-74 years, the overall mean was 0.68 µmol/litre (14.1 µg/dl)
(Roberts et al., 1985). A sex difference was noted: 0.77 µmol/litre
(16.1 µg/dl) was found in males and 0.57 µmol/litre (11.9 µg/dl) in
females. Residents in the centre of large urban areas were found to
have a PbB level of 0.72 µmol/litre (14.9 µg/dl), while rural
residents it was 0.62 µmol/litre (13.0 µg/dl). Because of the design
of the study, information on PbB levels, in relation to smoking,
alcohol consumption, and occupational status, were available. Male
workers in occupations with a high potential for lead exposure had
mean PbB levels of 0.78 µmol/litre (16.2 µg/dl) for nondrinkers/
non-smokers, 0.92 µmol/litre (19.2 µg/dl) for non-drinkers/non-smokers
and 0.95 µmol/litre (19.7 µg/dl) for drinkers/smokers. It was noted
that a true decrease of 37% in PbB level occurred during the period of
the survey (Annest et al., 1983).
Rabinowitz & Needleman (1982) reported an arithmetic mean
umbilical cord PbB concentration of 0.32 µmol/litre (6.6 µg/dl) (range
0-1.78 µmol/litre; 0-37 µg/dl) in over 11 000 samples collected
between 1979 and 1981. A decrease in PbB level of approximately 11%
was noted during the period of collection.
Friberg & Vahter (1983) reported the PbB levels of 200 teachers
from nine countries (see section 5.5). The median PbB level in this
study ranged from 0.29 µmol/litre (6 µg/dl) in Beijing and Tokyo to
1.06 µmol/litre (22 µg/dl) in Mexico City. Smokers generally had
higher PbB levels than non-smokers.
During the period 1978-1988 marked decreases (approximately 30 to
40%) in the average PbB levels of adults were noted in Belgium,
Germany, New Zealand, Sweden, the United Kingdom, and the USA (OECD,
1993).
5.5.2 Children
Results from the NHANES II survey in the USA indicated a mean PbB
level for children (6 months to 2 years old) of 0.78 µmol/litre
(16.3 µg/dl). It was noted that 18.6% of black inner city children had
PbB levels > 1.44 µmol/litre (30 µg/dl) whereas only 4.5% of white
children had such PbB concentrations. The percentage of children with
high PbB level decreased with increasing family income (Roberts et
al., 1985). The NHANES II data were analysed in detail for information
on the demographic correlates of children's PbB concentrations
(Mahaffey et al., 1982; Annest et al., 1983). The racial differences
in these proportions were significant among both boys and girls. The
proportion with elevated PbB concentrations was slightly higher among
boys than girls for both races. Among both black and white children,
the percentage with elevated PbB concentrations decreased with
increasing family income. Proportionately more young children in urban
than rural areas and in the central cities of large urban areas had
elevated blood lead concentrations.
PbB concentrations were measured in 286 Finnish children living
in the three largest cities of Finland (number of subjects = 172), in
rural areas (54 subjects) and in a lead smelter area (60 subjects)
(Taskinen et al., 1981). The mean PbB levels in urban, rural and
lead-smelter areas varied between 0.29 µmol/litre and 0.32 µmol/litre,
range 0.14-0.82 µmol/litre (6.0 and 6.7 µg/dl, range 3-17 µg/dl).
There were no significant differences between the areas of residence.
The five children who lived within 500 metres of the lead smelter
had a mean PbB concentration of 0.44 µmol/litre, range 0.24-0.62
µmol/litre (9.2 µg/dl, range 5-13 µg/dl), which was significantly
higher than the mean blood lead concentration among children living
in the rest of the country.
In a study carried out in Sweden, which included 1781 samples
obtained from children during 1978-1988, the mean PbB level decreased
from 0.29 µmol/litre (59.6 µg/litre), range 0.09-1.20 µmol/litre
(18-250 µg/litre) in 1978 to 0.16 µmol/litre (32.9 µg/litre), range
0.07-0.34 µmol/litre (15-71 µg/litre) in 1988 (Schutz et al., 1989).
In Finland, a remarkably similar decrease has been reported; the mean
PbB value was 0.14 µmol/litre (30 µg/litre), range 0.1-0.40 µmol/litre
(21-41 µg/litre) among 35 children in the Helsinki area in 1988 (Ponka
et al., 1991).
As in the USA (Annest et al., 1983), children's PbB
concentrations in Sweden have decreased in recent years. Levels were
determined each summer during the period 1978-1984 in children from
Scania in Southern Sweden (Skerfving et al., 1986). The average PbB
concentration was 0.26 µmol/litre, range 0.07-1.20 µmol/litre
(5.5 µg/dl, range 1.4-25.0 µg/dl). There was a statistically
significant decrease over time in both the rural and urban areas,
averaging about 0.019 µmol/litre (0.4 µg/dl) per year.
Decreases of 25-45% in average PbB levels in children between
1978 and 1988 have been reported in Belgium, Canada, Germany, New
Zealand, Sweden and the United Kingdom (OECD, 1993).
5.5.3 Remote populations
In contrast, a much higher mean value of 0.16 µmol/litre
(3.4 µg/dl) was obtained in Nepal upon examination of 103 children
living in the Manang District, which is also very remote and thought
to be free of anthropogenic sources of lead (Piomelli et al., 1982).
However, these people are exposed to significant amounts of natural
lead from living in dusty smoke-filled houses, and from burning pine
and yak dung. The lead level in the houses was about 0.15 µg/m3
(Davidson et al., 1981). For comparison outdoor air in Woods Hole,
Massachusetts, USA, contains 0.004 and Boston, Massachusetts, 0.01 to
0.05 µg lead/m3.
Another remote population studied was from Okapa, Eastern
Highlands, Papua New Guinea (Poole et al., 1980). Extreme care was
taken in sampling and analysis, and the precision was acceptable at
PbB levels as low as 0.24 µmol/litre (5 µg/dl). The children subsisted
on root crops and a small amount of tinned fish and meat. Villages
were typically hazy with wood smoke, natural mineral pigments were
used extensively, and tobacco smoking was common. This survey of 100
children yielded a mean PbB level of 0.25 µmol/litre (5.2 µg/dl), well
above the detection limit, and, as reported by Poole et al. (1980),
far less than the mean of 1.10 µmol/litre (23 µg/dl) for Sydney,
Australia, in 1974.
5.6 Occupational exposure
Occupational exposure to lead which results in poisoning, both
moderate and clinically symptomatic, still occurs in many countries of
the world. Although adults are mainly involved, in many countries,
especially in those with developing industries and small home-based
industries, the distinction between home and workplace is non-existent
(Verrula & Noah, 1990) and children are exposed to workplace lead.
Until recently, in the occupational setting, there was only concern
for the identification of late stage, highly symptomatic cases of lead
poisoning resulting in crippling neurological manifestations of lead
poisoning such as palsy and encephalopathy (Chakravorti & Bhar, 1978;
Wang, 1984; Davies, 1984; Lin-Fu, 1985). However, there is now concern
for lower exposures to lead.
In many countries occupational lead exposure is entirely
unregulated and no monitoring of exposures exists. Automobile battery
manufacture and repair, radiator repair, secondary smelters (including
scrap metal refiners) are found in most countries. The industries
where there is a potential for lead exposure are listed in Table 10
(section 5.1.3). Significant lead exposures are not limited to
traditional heavy industries. For example, Kaye et al. (1987)
identified exposures from leadborosilicate dust used in a capacitor
and resistor plant in the USA.
Because of transfer of lead to the fetus (in utero) and the
transport to the home of lead on clothing, etc., thereby exposing the
young child in the home, the problems of occupational exposures to
lead are not limited to the workplace per se. Cases of poisoning
among the children of lead workers have been reported (Baker et al.,
1979; Wang, 1984; Ryu et al., 1985).
Although most occupational standards are based on airborne lead
only, this route of exposure does not fully reflect the total daily
exposure of workers, also exposed to lead in food, water, alcoholic
beverages and dusts.
The potential for hazardous exposures to lead during lead
smelting and refining are well recognized, particularly where molten
lead and alloys are poured, resulting in the vaporization of metal.
This is true for both primary new metal and secondary (lead scrap)
smelters and refineries.
Small domestic versions of secondary smelters exist in a large
number of countries. These are typically located within or in close
proximity to homes. For example, in Jamaica there has been a rapid
proliferation of lead smelters, particularly illegal backyard smelters
(Rodney & Lee, 1985). The lead fumes and dust generated in such
operations pose an exceptional health hazard to children and adults
living near these operations. Rodney & Lee (1985) reported that 51% of
116 children (aged 2 to 12 years, mean 5.9 years) and 60% of 235
adults working in or living near lead smelting factories had PbB
concentrations of 1.92 µmol/litre (40 µg/dl) or more.
Other occupations where workers have been shown to be at risk
from airborne lead include electric storage battery manufacturing,
particularly where industrial hygiene is poor (Barrio & Badia, 1985);
demolition, welding and shipbreaking where lead-based paint is present
(Holness & Nethercott, 1988; Nosal & Wilhelm, 1990); pottery and
ceramic-ware production, which is often a home-based operation
involving women and children (MolinaBallesteros et al., 1983; Katagiri
et al., 1983; Kaye et al., 1987); small businesses repairing
automobile radiators (Matte et al., 1989a,b; Verrula & Noah, 1990),
and artisans producing jewellery and decorative wares. This latter
industry is of particular concern since it is predominantly carried
out at home or in non-regulated shops, often by women and children
(Behari et al., 1982). Indian silver jewellery makers were found to
have a PbB level of 5.8 µmol/litre (121 µg/dl) compared to non-exposed
controls with a PbB level of 1.3 µmol/litre (27 µg/dl) (Behari et al.,
1982).
6. KINETICS AND METABOLISM IN LABORATORY ANIMALS AND HUMANS
6.1 Absorption
The absorption of lead from environmental sources is not
dependent solely on the amount of lead presented to the portals of
entry. It is also dependent on the physical and chemical state in
which the metal is presented, and it is influenced by host factors
such as age, physiological status, nutritional condition and,
possibly, genetic factors. Men engaged in heavy work breathe more air
and eat more food than sedentary individuals of the same weight, and
on a body weight basis children eat almost as much food and breathe
almost as much air as middle-aged adults.
Considerable data from human subjects are available. Therefore,
discussion of animal studies will be limited to areas where the
information on humans is inadequate. Various models used to predict
body burden or distribution of lead have been developed (Bernard,
1977; Marcus, 1985a,b,c; Bert et al., 1989; Arnetz & Nicolich, 1990).
A full review of these models has been judged to be beyond the scope
of this document. However, some of them were used in section 6.1.4
when correlating intake and body burdens.
6.1.1 Absorption after inhalation
The absorption of lead from air to blood involves two processes:
the deposition of airborne lead particles in the respiratory tract;
and the absorption and clearance from the respiratory tract into the
circulation. Using the International Radiological Protection
Commission (IRPC) document on lung dynamics (Task Group on Lung
Dynamics, 1966), a model was developed which predicted that 35 to 50%
of inhaled lead is deposited in the respiratory tract (40-50% of
particles with a mean mass median aerodynamic diameter (MMAD) of
0.5 µm, such as are typically generated by automobiles). These are
deposited primarily in the alveolar sacs of the lung. Lead fumes and
vapours, such as those generated in operations where metals are cut or
heated, are of very small size and are respirable. Absorption after
deposition will vary according to the solubility of the lead species
(e.g., lead carbonate or lead chloride aerosols) and the inherent
toxicity to lung macrophages and cilia.
6.1.1.1 Animal studies
Limited animal studies confirm that there is almost complete
absorption of lead particles (0.1 to 0.5 µm in diameter) deposited in
the lower respiratory tract. In rats the clearance half-time from lung
is short (less than one hour) and 90 to 98% of the administered dose
is absorbed within about 48 h (US EPA, 1986a).
6.1.1.2 Human studies
The respiratory deposition of airborne lead is in the range of
30-50% and varies with particle size and ventilation rate (US EPA,
1986a). Higher deposition rates may occur with larger particles, but
this deposition takes place in the upper respiratory tract, with
eventual displacement to the gastrointestinal tract and absorption via
the ingestion route. This probably explains the observation of Kehoe
(1961) that faecal excretion of lead increased after a subject
breathed for many weeks aerosols of lead oxide (150 µg/m3) with an
MMAD of approximately 2.9 µm. In contrast, smaller particles of
inhaled lead, such as those generated by automobile exhaust,
regardless of physicochemical form, are almost (> 90%) completely
absorbed after deposition in the lower respiratory tract (Rabinowitz,
et al., 1977a; Chamberlain et al., 1978; US EPA, 1986a).
There are no quantitative data on the absorption of lead in
children after inhalation exposure. It is known that young children
weighing only one sixth of an adult inhale 40% of the daily volume of
an adult and a proportionately higher daily air volume per unit
measure (weight, body area) than do adults (Barltrop, 1972). After
controlling for weight and taking into account differences in the
anatomy of the respiratory tract between adults and children, James
(1978) calculated a rate of deposition of lead particles in children
which was 1.6 to 2.7 times that of adults.
6.1.2 Absorption of lead from the gastrointestinal tract
In the case of older children and adults without occupational
exposure, lead absorbed by the gastrointestinal tract comes from the
intake of lead in foods, beverages and soil/dust. In pre-school
children, there is concern over the intake of both food and non-food
items (e.g., toys, soil/dust). Young children may take in lead from
non-food items, via normal mouthing activity, which in the extreme, is
the behavioural trait pica, which refers to the ingestion of such
materials as soil, ash, paint chips and plaster (US EPA, 1986a). For
infants and young children, the extent of absorption of the lead in
dust/soils from the gastrointestinal tract is extremely important,
particularly for children living in urban environments.
6.1.2.1 Animal studies
The absorption of lead from the gastrointestinal tract in
experimental animals is age dependent and is modified by the level of
food intake. In 1-week-old suckling rats, an absorption rate of about
52% was reported after a single oral dose of lead chloride, compared
to 0.4% in 6-week-old animals (Kostial et al., 1978). Furthermore,
Aungst et al. (1981) reported that rat pups had higher tissue levels
of lead than adults after a single gavage dose of 1 or 10 mg lead (as
lead acetate) per kg body weight. Fasting markedly enhanced the uptake
of lead in the gastrointestinal tract of experimental animals (Garber
& Wei, 1974; Pounds et al., 1978).
In experimental animals, absorption of lead from the
gastrointestinal tract appears to be a saturable process. With
increasing doses (1 to 100 mg lead (as lead acetate) per kg body
weight), lead absorption as a percentage of dose was found to decrease
from 42% to 2% (Aungst et al., 1981; Bushnell & DeLuca, 1983) in
dietary studies. Such data are consistent with a saturable active
transport process across the gastrointestinal tract.
The low rate of absorption of lead from soluble lead salts noted
in adult rats may reflect a dietary effect. Kostial & Kello (1979)
reported less than 1% absorption in adult rats given chow diets,
whereas the absorption rate was 3-20% when rats consumed diets similar
to that of humans (milk, bread, baby foods, etc). This range more
closely resembles the human absorption rates.
The chemical form of lead can influence its bioavailability.
Stone et al. (1981) determined the biological availability of lead
intrinsically incorporated into the soft tissues of oysters. The
bioavailability to the rat was 10-30% lower than that of lead acetate
added to the basal chow diet. Henning & Cooper (1988) compared the
absorption of lead from rat milk labelled in vitro with lead-203 and
lead chloride or lead acetate solutions. Lead from soluble salts
accumulated primarily in the duodenum, to some extent in the jejunum
and minimally in the more distal small intestine. Lead from milk
accumulated only in the upper ileum. After 20 h negligible lead-203
was found in any region following administration of soluble salts, but
there was substantial retention of lead in the ileum after dosing with
milk.
Data obtained from a feeding study in rats (Dieter et al., 1993)
showed that lead uptake into rat femurs was highly dependent on the
chemical form of lead administered. Bioavailability was highest for
lead acetate, intermediate for lead oxide, and lowest for lead sulfide
and Alaskan mixed ore concentrate. This uptake was linearly related to
dose over the range studied. However, the slopes of the linear
regression equations differed according to the form of lead. They were
0.10 µg lead/g femur per kg diet for lead sulfide and 2.64 for lead
acetate.
However, results of an investigation in young swine of the
absorption of lead from mining and/or milling operation waste (LaVelle
et al., 1991) differ from those reported by Dieter et al. (1993). The
chemical species (predominantly lead sulfide) and particle size (i.e.
larger than 100 µm) were expected to result in these sources being
less bioavailable than soluble lead salts such as lead nitrate.
However, experimental data indicate that the lead in mine tailings may
be about 2-3 times more available than reagent grade lead sulfide
under the conditions of the study. Additionally, Freeman et al. (1992)
reported that the lead in mining waste soil was between 8 and 20% as
bioavailable as lead acetate. No detailed comparisons were made of the
physicochemical properties of these two mine wastes. An additional
study involving the absorption of lead from lead sulfide ores and
their oxidation products in rabbits and in vitro showed that lead
from minerals was absorbed less well than from lead acetate by a
factor of 5 (Davis et al., 1990a). The significance of these phenomena
is not clear.
6.1.2.2 Human studies
Gastrointestinal absorption of lead in humans, as in experimental
animals, is influenced by dietary factors, nutritional status and the
chemical form of the metal. Overall patterns of food intake may also
influence lead absorption. For example, lead ingested during periods
of fasting is absorbed to a much greater extent than lead ingested
with food. Chamberlain et al. (1978) reported 45% absorption of lead
chloride in fasting subjects and only 6% in feeding subjects. Using a
similar procedure, Heard & Chamberlain (1982) reported absorption of
63.3% in fasting subjects. Using multiple stable isotope lead tracers,
it was found that in adult men the gastrointestinal absorption rate
with food containing lead nitrate or lead cysteine was 6-12%. However,
when consumed under fasting conditions, lead nitrate, lead sulfide and
lead cysteine were absorbed at 16-53% (Rabinowitz et al., 1977b).
Kehoe (1961) estimated only 10% net absorption of dietary lead by
adults in long-term metabolic studies. However, with appropriate
calculation of biliary clearance as well as of urinary excretion, a
figure of 15% was estimated by Chamberlain et al. (1978). In several
studies on adult humans, absorption of lead was reported to be 14%
when it was administered with food (Chamberlain et al., 1978; Moore et
al., 1979; Rabinowitz et al., 1980).
Ziegler et al. (1978) reported that young children, aged two
weeks to two years, absorbed 42% of ingested lead at levels of intake
greater than 5 µg/kg body weight. Drill et al. (1979) estimated an
absorption rate of 17% for lead in paint chips in children aged 2 to 3
years. These authors also estimated a 30% gastrointestinal absorption
rate for lead in soil and dirt.
The amount of lead ingested by children from non-food items such
as soil, dust and paint chips through normal mouthing activity,
particularly for children with pica, is a major concern in calculating
paediatric lead exposures. Using data from Day et al. (1975) and Lepow
et al. (1974), it has been estimated that children 2 to 3 years of age
ingest about 100 mg soil per day. Using aluminium and titanium as
tracers, Clausing et al. (1987) estimated that children aged 2 to 4
years ingest between 50 and 100 mg of soil daily. The average amount
of soil ingested by young children has recently been estimated to be
between 12.5 and 21 mg/day (SAHC, 1993).
6.1.2.3 Nutritional status and lead absorption via gastrointestinal
tract
It has been known for some time that the absorption and
distribution of lead are affected by nutritional status in both
experimental animals and humans (Sobel et al., 1940). Nutritional
inadequacies can also affect the toxic response to lead (see sections
7 and 8). In view of documented nutritional inadequacies in many parts
of the world, such interrelationships become crucial in assessing the
full risk from lead exposures.
Vitamin D, calcium and phosphorus have complex and interrelated
effects on lead absorption (Fullmer, 1990). Increasing the
concentration of 1,25-dihydroxycholecalciferol, the active metabolite
of vitamin D, either exogenously or endogenously, increases
gastrointestinal absorption of lead (Fullmer, 1990). However, this
effect is dependent upon the duration of lead exposure and the
magnitude of body lead stores. This homoeostatic mechanism for calcium
and its dependence on nutritional status, as well as body burden of
lead, is complex. This may explain the divergent results of the
observed interaction in children (Rosen et al., 1980) or the lack of
association (Koo et al., 1991) of lead with vitamin D metabolism.
In experimental animals, chronic ingestion of diets with less
than adequate amounts of calcium (Quarterman & Morrison, 1975),
phosphorus (Quarterman & Morrison, 1975), iron (Mahaffey-Six & Goyer,
1972; Ragan, 1977), selenium (Stone & Soares, 1976) or zinc
(Cerklewski & Forbes, 1976) increases the fractional absorption of
lead in the gastrointestinal tract. From the work of Fullmer & Rosen
(1990), it appears that lead alters the transport of calcium and other
trace minerals by affecting carrier proteins, rather than by competing
at the mucosal surface.
The influence of iron on lead absorption in humans was assessed
in double-labelling experiments conducted by Watson et al. (1980).
Lead-203 and iron-59 were given to 28 subjects in their diet. There
were significant positive correlations between iron levels in food and
lead absorption. However, the association was not strong because 50%
of those absorbing excess lead also absorbed excess iron.
Rats fed iron-deficient diets have increased concentrations of
lead in kidney and bone when compared to rats ingesting equivalent
quantities of lead (as lead acetate) in drinking-water while being
fed an iron-adequate diet (Mahaffey-Six & Goyer, 1972). In contrast
with calcium deficiency, iron deficiency does not result in a
redistribution of lead to non-osseous tissue (Mahaffey-Six & Goyer,
1972). The degree of iron deficiency does not need to be severe to
increase retention of lead. For example, Ragan (1977) demonstrated
six-fold increases in tissue lead in rats when body iron stores were
reduced, but before frank iron deficiency developed. High levels of
dietary iron resulted in decreased kidney, femur and blood lead
concentrations in rats (Mahaffey, 1985). Iron appears to increase
absorption rather than decrease lead excretion (Barton et al., 1978).
Lead interferes with normal transferrin transport of iron, and
inhibits transferrin endocytosis and iron transport across the cell
membrane of the reticulocyte (Qian & Morgan, 1990).
6.1.3 Dermal absorption
6.1.3.1 Human dermal absorption
Moore et al. (1980) examined the uptake of lead acetate from two
hair-darkening cosmetics through the skin of eight human volunteers.
Only minute quantities of lead (0-0.3% of the applied dose) were
detectable in blood, and there was only a slight increase in
absorption when the skin was damaged. Lilley et al. (1988) and
Florence et al. (1988) have reported the dermal absorption of
inorganic lead compound leading to elevated levels of lead in human
saliva and sweat.
Dermal absorption of inorganic lead through unabraded human skin
is considered to be minimal.
6.1.4 The relationship of external lead exposure to blood lead
concentration
There are numerous environmental media that provide routes by
which humans are exposed to lead: food, water, air, soil, dust.
External exposures are the sum of the quantities of lead consumed from
all sources.
Historically there have been two lines of approach in
understanding the lead exposure and blood lead relationship. Most have
been empirical and have measured environmental lead and PbB levels
either at one time or repeatedly. With these observed correlations and
with no assumptions about how lead moves inside the body, many
reasonable predictions can be made. The validity of these predictions
is optimum when only one environmental source dominates. Some of these
rely on linear functions, while others have specified non-linearities,
especially over very wide ranges (more than a factor of 10) of lead
exposures. However, when multiple sources are considered these
predictive models have been less satisfactory.
Internal lead levels in human populations have been estimated by
analyses of a variety of biological tissues (e.g., blood, teeth, bone
and hair). Lead concentrations in each of these have particular
biological meanings with regard to external exposure to lead. Blood is
the compartment in which lead is most often measured as a marker of
exposure. However, it typically represents relatively recent
exposures, since the half-life of lead in blood is short (US EPA,
1986a) and has been estimated to be in the order of 36 days from
tracer studies (Rabinowitz et al., 1975). Lead in blood is derived
from levels in the current environment and from lead stored in tissues
(particularly bone) that re-enters the blood during tissue
mobilization (Manton, 1985). Although PbB concentration reflects
recent exposure and bears a consistent relationship to levels of lead
in the external environment, when bone mobilization is accelerated a
greater fraction of PbB will be derived from tissue stores.
To date only a few studies have utilized a multimedia approach
relating lead intake to PbB levels. The majority of studies have
attempted to correlate PbB levels and lead concentrations in specific
media.
6.1.4.1 Ambient air
a) Occupational exposure
Several studies have examined the blood lead: air lead
relationship for workers exposed to levels of airborne lead between 9
and 450 µg/m3 (King & Conchie, 1979; Gartside & Buncher, 1982;
Bishop & Hill, 1983). The results of all three studies are in general
agreement over the wide range of airborne lead studied and for PbB
levels between 0.96 and 4.32 µmol/litre (20-90 µg/dl). The blood lead:
air lead relationship in occupational settings is best described by a
curvilinear relationship having slopes between 0.00096 and
0.0038 µmol/litre (0.02 and 0.08 µg/dl) per µg/m3 air.
b) Non-occupational exposure
Both population and experimental studies have been used to
estimate the PbB: ambient air lead relationship in adults and
children. Under ambient conditions (air lead concentrations of
0.1-2.0 µg/m3) and PbB levels less than 1.44 µmol/litre
(< 30 µg/dl), the relationship has been described as linear (Colombo,
1985). The various slope estimates reported are based on the
assumption that an equilibrium level of lead in blood is achieved.
Based on three major studies (Yankel et al., 1977; Angle & McIntire,
1979; Roels et al., 1980), the median slope for children is about
0.091 µmol/litre (1.9 µg/dl) blood per µg/m3. In adult males a slope
estimate of 0.076 µmol/litre (1.6 µg/dl) blood per µg/m3 was
calculated. When one calculates the relationship between PbB and the
total contribution from air (direct inhalation plus indirect through
dust/soil), a value of about 0.14-0.24 µmol/litre (3-5 µg/dl) blood
per µg/m3 is obtained (Brunekreef, 1984; US EPA, 1986a). Also on the
basis of a linear model, Snee (1981) reported that the best estimate
of the blood lead: air lead relationship was 0.048 µmol/litre
(1 µg/dl) per µg/m3. After examining available epidemiological and
experimental data, Chamberlain (1983) concluded that most published
estimates of the slope were between 0.072 and 0.144 µmol/litre per
µg/m3. From these results, it can be concluded that airborne lead
will only be a major contributor to PbB levels in areas of high air
lead levels.
6.1.4.2 Food
The relationship of PbB to dietary intake has been estimated from
experimental (Stuik, 1974; Cools et al., 1976; Schlegel & Kufner,
1979) as well as population studies (Sherlock et al., 1982; UK, 1982;
Ryu et al., 1983). In adults, the results from both types of studies
using both linear and cube root models indicated a relationship of
between 0.0019 and 0.0028 µmol lead/litre (0.04 and 0.06 µg/dl) per µg
lead intake per day. From the study of Ryu et al. (1983) a slope of
0.0096 µmol/litre (0.2 µg/dl) per µg lead/day was obtained for infants
aged 8 to 196 days.
Currently, data are available for adults and children from
studies with careful control of important variables such as: intake of
dietary lead and of other dietary constituents, minimal exposure to
sources other than diet in the studies of infants, and intake/blood
lead measurements that can be used to estimate intake from all
sources. For infants these are studies reported by Sherlock et al.
(1982), UK (1982), Ryu et al. (1983), and Lacey et al. (1985). Studies
by the UK (1982) and by Sherlock et al. (1982) were conducted at
higher levels of lead exposure than were the studies conducted with
infants by Ryu et al. (1983). The latter studies were at exposure
levels associated with PbB concentrations under 0.96 µmol/litre
(20 µg/dl); the ratio of blood lead to ingested lead was
0.0076 µmol/litre (0.16 µg/dl) per µg lead ingested per day. Exposure
levels were low: average dietary lead intake was 17 µg/day. After four
months, the average PbB concentration was 0.29 µmol/litre (6.1 µg/dl)
whole blood. Between the ages of 4 and 6 months, 10 children remained
at a dietary lead intake of 0.76 µmol/litre (16 µg/dl). Their PbB
concentrations were quite constant at 0.35 µmol/litre (7.2 µg/dl) at
the end of the study period. The remaining seven children received
canned infant formula and/or milk during this period and their average
dietary lead intake was 61 µg/day. At the end of the study period,
their PbB concentration was 0.69 µmol/litre (14.4 µg/dl). Based on
these data, a curvilinear relationship between blood lead and total
lead intake was suggested, a 4-fold increase in lead intake resulting
in a doubling of PbB concentration. Sherlock et al. (1982) conducted a
duplicate diet study of 31 mothers and their children from Ayr,
Scotland. The slope for adults was substantially lower than for
children.
6.1.4.3 Drinking-water
There is still debate over the most appropriate model (i.e. cube
root, polynomial or logarithmic) to describe the curvilinear
relationship of waterborne lead to blood lead. The highest estimates
for the contribution of water lead to blood lead come from the cube
root and logarithmic models. Much lower estimates are obtained from a
linear model (Pocock et al., 1983; US EPA, 1986a). In a study by
Sherlock et al. (1982), a cube root relationship between lead levels
in drinking-water and blood fitted the data more closely than a linear
relationship. The curvilinear model implies that as abatement
processes lower water lead concentrations, there will be an increasing
benefit in lowering of population PbB levels (Moore, 1983).
6.1.4.4 Soil and dust
It is extremely difficult to choose the most appropriate model to
describe the soil/dust to blood lead relationship, given the many
variables involved in determining the exposure patterns of children
and the kinetics involved between the levels in the environment and
the child. A review of the available studies shows the extreme
variability in slopes obtained (0.028-0.36 µmol lead/litre
(0.6-7.6 µg/dl) blood for each 1000 µg/g soil and 0.00096-0.35 µmol
lead/litre (0.02-7.2 µg/dl) blood for each 1000 µg/g dust consumed by
children) (US EPA, 1986a). Detailed consideration has been given to
the process for the assessment of lead contamination in soil and the
derivation of soil clean-up criteria by Wixon (1991). The guideline
model uses a PbB target and slope for the soil:blood lead relationship
in the particular community in order to derive a PbB guideline. For
house dust a median value of 0.086 µmol lead/litre (1.8 µg/dl) blood
per 1000 µg lead/g dust in children appears to be based on data of
reasonable quality, as does the 0.105 µmol lead/litre (2.2 µg/dl)
blood per 1000 µg lead/g soil from the same authors.
According to Elwood (1986), studies of the association between
lead in house dust and PbB have given inconsistent results and, in
general, the only studies in which statistically significant
association has been found are those where lead levels in dust are
quite high. Landrigan et al. (1975) found a highly significant
association between PbB and dust lead in an area with a mean level of
4022 µg lead/g dust, but not in two areas with means of 922 and 816 µg
lead/g dust. The US EPA (1986a) summarized a series of studies from
which the overall relationship was judged to be 0.86 µmol/litre
(18 µg/dl) blood per 1000 µg lead/g dust. Duggan (1980) reviewed the
literature and determined that a slope of 0.24 µmol/litre (5 µg/dl)
per 1000 µg lead/g dust is reasonable.
The overall relationship between PbB and dust/soil lead depends
on the lead concentrations and bioavailability as well as on the
proximity and linkage between humans and their environment. This
relationship varies among locales.
6.1.4.5 Total lead intake
The non-linear relationship between PbB and total lead intake
(see section 5.5) is curvilinear across a broad range of PbB values,
such that the slope decreases with increasing lead levels. A number of
biological factors may explain the curvilinear relationships, such as
increased renal clearance with high PbB (Chamberlain, 1983, 1985),
distributional non-linearities due to differences in lead binding
sites in different tissues (Hammond et al., 1981; Manton, 1985;
Marcus, 1985b), or a sizeable pool of mobile lead in bone maintained
more or less independently of uptake (Rabinowitz et al., 1976;
Chamberlain, 1983).
6.2 Distribution
The initial distribution of lead in the body may depend upon the
rate of delivery of blood to various organs. However, it would appear
that distribution occurs in a similar manner regardless of the route
of absorption (Kehoe, 1987). The distribution of lead in humans under
environmental exposure conditions reflects the fact that almost all
exposures are chronic rather than acute.
6.2.1 Animal studies
Studies in rats have shown that lead is rapidly distributed into
soft tissues and subsequently redistributed into soft and mineralizing
tissues after acute and chronic exposures. After acute inhalation
(Morgan & Holmes, 1978) or oral (Aungst et al., 1981) exposure, lead
levels in rats were highest in liver, kidneys and lung, with levels
increasing in bone as those in soft tissues declined and stabilized.
Similar distribution patterns were reported for mice exposed for 12
months to 21.5 µg lead/m3, the highest levels being found in bone
and the lowest in lung (Keller & Doherty, 1980a).
Age-related differences in the distribution of lead in
experimental animals have been reported. Kostial et al. (1978) noted a
greater retention of lead in suckling rats than in adults; the levels
in the brain were also higher in the pups. A 2- to 3-fold increase in
brain lead concentration (highest in hippocampus) was noted after a
10-fold (0.1 to 1 mg/kg body weight administered by gavage) increase
in the dose given to 4- to 8-week-old rat pups (Collins et al., 1992).
Ageing has been shown to alter the pattern of distribution of
lead in rats administered lead acetate in drinking-water. Juvenile (21
days old), adult (8 months old) and elderly (16 months old) rats
received 0, 1.27 and 6.37 mg lead/kg bodyweight for 9.5 months. The
pattern of distribution, namely femur > liver > brain was similar in
all age groups. However, age-related increases in brain lead levels
were noted, along with decreases in femur lead content (Cory-Slechta,
1990a).
6.2.2 Human studies
Lead is distributed to both soft tissues (blood, liver, kidney,
etc.) and mineralizing systems (bone and teeth). Bone may be affected
adversely by lead but also serves as the body's major storage site.
Bone accumulates lead over much of the human life span, and a study of
the kinetics of distribution is important since bone can, under
appropriate conditions, pose a risk as a potential endogenous source
of lead.
Once absorbed, lead is not distributed homogeneously throughout
the body but rather into several distinct compartments. Such
biokinetic movements have been explained by Rabinowitz et al. (1976)
using a three-compartment model. This model was based on tracer and
balance data from five healthy men and has been refined by Marcus
(1985a,b,c). Three pools (blood, bone and soft tissues) were
identified, with lead having distinct half-lives in each. Blood lead
was considered the most labile compartment with a half-life of about
36 days, and bone lead the most stable with a half-life of about 27
years. Lead in soft tissue had a half-life of approximately 40 days.
The specific physiological foundations for these biokinetic
models have received much attention and are currently being refined
(O'Flaherty, 1991, 1993). A more thorough understanding of bone
growth, for example, expressed as a series of allometric equations,
should help improve models, especially when they must be applied to
growing and maturing humans.
Under steady-state conditions, about 96% of PbB is in the
erythrocyte. At PbB concentrations of < 1.92 µmol/litre (40 µg/dl),
whole blood and serum lead levels increase linearly in a positive
manner. At higher PbB concentrations a curvilinear relationship is
apparent and the serum to blood ratio increases dramatically (Manton &
Cook, 1984). Such kinetic relationships may be altered during
pregnancy. From in vitro data (Ong & Lee, 1980), fetal haemoglobin
appears to have a greater affinity for lead than adult haemoglobin.
In adults, approximately 94% of the body burden of lead is in the
bones, whereas only 73% of the body burden in children is located in
this compartment (Barry, 1975, 1981). In view of the extremely long
half-life for lead in bone, this compartment can serve as an
endogenous source of lead to other compartments long after exposure
ceases (O'Flaherty et al., 1982; Kehoe, 1987). This is due to the
labile lead compartment in bone (Rabinowitz et al., 1976) and the
ongoing bone redistribution which is subject to alteration by in vivo
metabolic processes (Parfitt, 1990). Although lead in bone generally
increases continuously with age, there is evidence that lead levels in
some bones (e.g., mid-femur and pelvic bone) plateau at middle age and
decrease with further ageing (Drasch et al., 1987). It is difficult,
however, to determine the role ageing plays in this process compared
to other biological reactions. Ageing may account in part for the
20-25% increase in PbB levels in menopausal women noted by Silbergeld
et al. (1988).
The metabolism of lead in bone has been summarized in reports by
Barry (1975, 1981), Drasch et al. (1987), Silbergeld et al. (1988),
Skerfving (1988) and Rabinowitz (1991).
Until recently, it was widely held that the human skeletal system
provided a metabolically inert depository for lead and was of little
consequence in health-risk assessment. It had been assumed that bone
lead is metabolically inert, with a half-life long enough to forestall
the risk of ready transfer back to blood. Current evidence is that
bone comprises a set of kinetically variable subcompartments for lead
deposition and is a target for toxicity. These factors complicate bone
lead kinetics as applied to long-term modelling; the mobility of bone
lead to blood is important (Rabinowitz, 1991).
Bone lead is readily mobilized to blood and the effect is most
apparent in people with a history of occupational exposure; bone lead
also appears to be a major source of blood lead in older people with
previous ambient exposures to lead. Of particular importance is
mobilization of lead from bone in pregnant women and nursing mothers
(Silbergeld, 1991). The mobilization of lead from bone to the more
bioavailable maternal blood compartment poses a risk to the fetus and
mother.
Human bone appears to have at least two, possibly three,
kinetically distinct lead compartments. Lead in trabecular (spongy)
bone appears to be more mobile than lead lodged in cortical (compact)
bone, and there appears also to be a fraction of bone lead in
equilibrium with the lead in blood (Skerfving, 1988). Trabecular bone
seems to be an important source of resorbed lead when high exposure is
reduced, e.g., through removal of medical reasons by retirement of
lead workers, or in response to chelation in adults (Shutz et al.,
1987).
6.2.3 Transplacental transfer
Lead is readily transferred from the mother to the developing
infant during pregnancy and accumulates in bone during gestation
(Barltrop, 1969). The lead concentration in cord blood is 85-90% that
of maternal blood. Moore et al. (1982) reported a geometric mean level
of 0.67 µmol/litre (14 µg/dl) in 236 pregnant women and
0.58 µmol/litre (12 µg/dl) for lead in umbilical cord blood. The mean
concentration of lead in umbilical cord blood from a sample of over
11 000 women was 0.298 µmol/litre (6.6 ± 3.2 µg/dl) (Bellinger et al.,
1987).
6.3 Elimination and excretion
In both humans and experimental animals lead is eliminated from
the body in both urine and faeces. Any dietary (including waterborne)
lead not absorbed in the gastrointestinal tract is excreted in faeces.
Airborne lead that has been swallowed and not absorbed is also
eliminated in this manner. Blood lead not retained in the body is
excreted in urine or faeces, the latter by biliary excretion. Adults
ingesting daily 0.3 to 3.0 mg lead (as lead acetate) in drinking-water
for 16 to 208 weeks excreted more than 85% of the ingested lead, 90%
of the excreted lead being in the faeces. The amount excreted through
any route is affected by age and exposure characteristics and is
species dependent (US EPA, 1986a). This section reviews only studies
of lead excretion in humans; a discussion of the results of work on
experimental animals can be found in reviews by US EPA (1986a) and
ATSDR (1993).
The age dependency of lead excretion in humans has not been
studied extensively. However, the studies of Rabinowitz et al. (1977b)
and Chamberlain et al. (1978) in adults and Ziegler et al. (1978) in
infants can be used to assess this phenomenon. Data from Ziegler et
al. (1978) and Rabinowitz et al. (1977a) are given in Table 20.
Ziegler et al. (1978) calculated a total daily retention by infants of
40 µg, which is about twice the amount calculated by Alexander et al.
(1973).
Table 20. Comparison of daily lead intake and excretion in children
and adultsa
Parameter Childrenb Adultsc
Dietary intake (µg/kg) 10.76 3.63
Fraction absorbed 0.55d 0.15
Dietary lead absorbed (µg/kg) 5.92 0.54
Air lead absorbed (µg/kg) 0.20 0.21
Total absorbed lead (µg/kg) 6.12 0.75
Urinary lead excreted (µg/kg) 1.00 0.47
Endogenous faecal lead (µg/kg) 1.56 0.24
Total excreted lead (µg/kg) 2.56 0.71
Excreted/absorbed lead 0.42 0.92
Fraction of intake retained 0.33 0.01
a Adapted from: US EPA (1986a)
b From: Ziegler et al. (1978)
c From: Rabinowitz et al. (1977a)
d Corrected for calculated endogenous faecal lead
Under conditions of relatively constant exposure to low
concentrations of lead, approximately 140 to 215 µg/day, a steady
state condition evolves in which excretion approximates intake
(Rabinowitz et al., 1976). Under these conditions urinary lead
excretion is approximately 70% of absorbed lead. Chamberlain (1985)
reported that approximately 60% of absorbed lead is retained by the
body and 40% excreted.
Chamberlain (1983, 1985) also examined the relationship between
the level of exposure and rate of lead excretion. Renal clearance at
PbB levels of between 1.2 and 3.84 µmol/litre (25 and 80 µg/dl) was
found to increase at a rate approximating the increase in plasma lead.
Chamberlain (1985) estimated endogenous faecal lead loss into the
gastrointestinal tract following administration of lead-203 via the
inhalation and parenteral routes. These estimates suggested a
clearance of approximately 0.5% of administered dose per day when PbB
concentrations were under 1.2 µmol/litre (25 µg/dl). A special form of
excretion of endogenous lead is through breast milk. Studies of breast
milk indicate that lead concentrations correlate with maternal PbB
concentrations, most studies reporting that lead secreted from breast
milk varies in concentration between 10 and 30% of the maternal PbB
concentration (Ong et al., 1985).
6.4 Biological indices of lead exposure and body burden
6.4.1 Blood lead
The relationship between levels of exposure from various
environmental media and PbB has been discussed briefly in section
6.1.4.
Due to the ease of sampling and homogeneity of the sample, blood
has been the most widely used specimen to assess the human body burden
of lead. However, in view of the relatively short half-life for lead
in blood (28-36 days) (see section 6.2.2), PbB measurements in general
reflect only recent exposures. Also, in view of the kinetics of
distribution within the body (cycling between blood, bone and soft
tissues), differentiation of low-level chronic exposure from a short
high-level exposure is not possible on the basis of a single PbB
measurement. Interpretation of PbB levels over a wide range of values
must take account of the curvilinear relationship between total intake
of lead and PbB concentrations, as well as the proportion of lead in
plasma (Manton & Cook, 1984; see also section 8.1).
A number of cohort studies have collected serial PbB measurements
for children from birth up to 7 or 10 years. For children who have not
had major changes in their environment, there is good correlation
between consecutive PbB measurements (McMichael et al., 1988;
Bellinger et al., 1992; Baghurst et al., 1992; Dietrich et al.,
1993a,b). In these extended studies it has become apparent that for
most of the children a single PbB analysis at 6 years of age gives a
reasonable assessment of the life-time lead exposure status of the
child. However, random PbB levels in samples taken before 6 years of
age can markedly underestimate the peak exposure usually seen at 2
years of age (SAHC, 1993).
A new exposure index, namely "lifetime average blood lead" or
"lifetime average integrated blood lead" has been introduced in
studies using serial blood data. It has been clearly explained in the
proceedings of a recent Workshop and this explanation is quoted
directly here. This measure, which is a reflection of the area under
the "blood lead by age curve", has been occasionally misunderstood.
Due to the shape of the longitudinal blood lead profile, the peak
blood lead level, usually observed during the second year of life is
considerably greater than the average lifetime blood lead. For
example, a five-year-old child with a lifetime average blood lead of
0.96 µmol/litre (20 µg/dl) is likely to experience blood lead levels
above 1.92 µmol/litre (40 µg/dl) during the second year of life and
may spend 3 years of life, from 12 months to 48 months, above
0.96 µmol/litre (20 µg/dl). Due to the shape of the curve, with
declining PbB beginning about 24-39 months of age, the average
lifetime blood lead for a given child decreases with increasing age.
This does not mean that the index is inappropriate but rather that
developmental blood lead profiles change over time. For example, a
lifetime average blood lead of 0.72 µmol/litre (15 µg/dl) should not
be interpreted as being equivalent to a single blood lead
determination of 0.72 µmol/litre (15 µg/dl) obtained at a single point
in life (Dietrich et al., 1991).
6.4.2 Tooth lead
Unlike blood samples, teeth are composed of several anatomically
distinct pools which form over several years. Thus, in contrast to
blood, teeth are useful tissues for assessing long-term lead
accumulation from prenatal exposures to the time of shedding of the
tooth.
The accumulation of lead in teeth (PbT) has been used as a
measure of exposure of children to lead in several epidemiological
studies (Needleman et al., 1972; Winneke et al., 1982a; Rabinowitz et
al., 1989; Fergusson et al., 1989; Hansen et al., 1989). Studies
measuring PbT and PbB, however, are few. Preliminary data from
Australia (Baghurst et al., 1992) indicate a correlation of 0.8
between whole PbT and PbB prior to tooth exfoliation, but the only
report relating lead in circumpulpal dentine and longitudinal PbB (by
Greene et al., 1992) found a correlation of only 0.5. PbT
concentrations can vary as a function of location of the tooth within
the mouth, age, and whether total PbT or dentine PbT were reported.
Therefore, the age of the tooth in the mouth and its location, the
sample (whole tooth or dentine) analysed must be considered in any
estimation of a corresponding PbB in the published studies (SAHC,
1993).
Because of the complex structure and development of teeth,
concentrations of PbT will depend on the method of sampling and
analysis, tooth type, and resorption and tooth age at exfoliation. As
a result of varying procedures used by different investigators, there
may be substantial variation in absolute values and possibly the
biological meaning of PbT levels between different studies.
Different parts of a tooth sequester lead during different stages
of development. This is especially of concern in tooth sampling
because lead is not uniformly distributed within a tooth on a
submillimeter scale. A pilot study of teeth from children with
differing histories of exposure to lead and using high precision lead
isotopic methods has shown that analyses of slices of the incisal part
of deciduous teeth give the clearest indications of the in utero
environment, and the cervical sections, the exposure from birth to
exfoliation (Gulson & Wilson, 1994).
For more detailed discussions on the measurement of PbT the
reader is referred to Grandjean et al. (1984), Purchase & Fergusson
(1986) and Fergusson et al. (1989).
6.4.3 Bone lead
The human skeleton begins to accumulate lead during fetal
development and continues to abut 60 years of age (Pounds et al.,
1991). Interest in bone lead and its measurement in vivo stems from
concern that skeletal lead is not metabolically inert (see section
6.2.2), but can be mobilized by physiological and pathological states,
for example, during pregnancy and lactation (Silbergeld, 1991) and
osteoporosis (Silbergeld et al., 1988), with possible adverse effects
in other tissues, including the fetus, as well as the desire to
develop a meaningful measure of cumulative lead exposure as a tool in
public health protection. Section 8.13 includes a brief discussion of
the skeleton as a target organ for lead toxicity.
Procedures are available to analyse bone samples for lead levels
in humans not occupationally exposed to lead (Drasch et al., 1987;
Drasch & Ott, 1988). These studies have shown a decrease in levels of
lead in bones from autopsy specimens in Germany after removal of lead
from petrol. However, full utilization of bone lead stores as
dosimeters of lead exposure in a prospective sense requires the
utilization of technologies for in vivo measurement of lead in bone
such as X-ray fluorescence analysis (Chettle et al., 1991; Rosen et
al., 1991; Todd et al., 1992).
6.4.4 Lead in urine
Although urinary lead level has been used to measure current
exposure (Robinson, 1974), its use as a biomarker of lead exposure is
questionable in view of the relatively low and variable level of lead
excreted in the urine (Jensen, 1984; Ibels & Pollock, 1986). For this
reason, and in view of technical difficulties in analysing low levels
of lead in urine, urinary lead appears to be of limited use for
general screening. However, where an elevated body burden of lead has
been estimated using PbB or other indices of lead exposure, urinary
levels of lead after administration of the chelating agent
CaNa2-EDTA is considered an excellent measure of the potentially
toxic fraction of the total body burden of lead (CDC, 1985). The
chelatable lead excreted is assumed to represent lead removal from
soft tissues and blood, as well as sub-compartments of bone (Ibels &
Pollock, 1986; Mushak, 1989). In contrast, the urinary lead excretion
associated with lead mobilization provides what is considered the best
measure of the potentially toxic fraction of the total body burden
(see CDC, 1985; US EPA, 1986a). On the basis of various in vitro
experimental and epidemiological studies (CDC, 1985; US EPA, 1986a;
Mushak, 1989), chelatable lead is assumed to be a chemical sample of
both mobile body compartments (i.e. blood and soft tissues) as well as
of sub-compartments of bone.
6.4.5 Lead in hair
Hair lead has been used as an indicator of exposure in children
(Marlowe & Errera, 1982; Wilhelm et al., 1989). However, there are
severe limitations on its use from both the methodological as well as
the metabolic perspective. Systemic variations in lead level have been
reported according to hair colour, texture, location on the body and
growth phase (Wilhelm et al., 1989). Also, it is almost impossible to
avoid external contamination, and to date no validated methods are
available for cleaning. Methods which are sufficiently vigorous to
remove superficial lead also remove lead from the hair shaft.
7. EFFECTS ON LABORATORY ANIMALS AND IN VITRO TEST SYSTEMS
Lead can affect various organ systems depending upon the level
and duration of exposure. In all species studied, adverse effects on
the nervous system have been noted at PbB concentrations lower than
for other target organs. There is particular concern for the effects
of lead on fetal development. Since the appearance of Environmental
Health Criteria 3: Lead (IPCS, 1977), there have been very many
reports on the effects of lead on in vitro and animal models. No
attempt will be made in this chapter to summarize all such studies.
Rather, emphasis has been given to those studies that relate most
directly to the understanding of the effects of lead on humans and
thus provide additional scientific support for the use of such human
studies in the assessment of risk from lead exposures.
7.1 Biochemical effects
7.1.1 Haem synthesis and haematopoiesis
The principal clinical manifestation of the effect of lead on the
haematopoietic system is anaemia but this occurs only with high levels
of exposure that are rarely seen today. Lead affects the
haematopoietic system at several levels. These include effects on haem
and globin synthesis and on erythrocyte formation and function.
Most haem synthesis is directed toward formation of haemoglobin
and the rest is used in cellular oxidative metabolism. Lead inhibits
several steps in the biosynthesis of haem. More detail can be found in
IPCS (1977), US EPA (1986a) and section 8.1.1 of this monograph.
Lead acts on steps in the synthetic pathway both inside and
outside the mitochondrion. It inhibits certain enzymes (ALA
dehydratase, ferrochelatase, coproporphyrinogen oxidase) and increases
the activity of ALA synthase (ALAS) activity as a consequence of
feed-back regulation by haem (Moore et al., 1980). The activity of
ALAS is the rate-limiting step in the haem biosynthetic pathway.
Lead affects erythrocyte formation by impairment of globin and
haem synthesis. Globin synthesis is inhibited by lead in rat bone
marrow cells at concentrations as low as 1 µmol/litre (Dresner et al.,
1982). This is thought to be secondary to decreased protein synthesis
in erythroid cells as a consequence of lead-induced inhibition of haem
synthesis. Lead also decreases erythrocyte survival through its
inhibition of membrane bound Na-K ATPase (Rhagavan et al., 1981).
It has been shown in in vitro cultures of cells from liver,
bone marrow and the nervous system that ALAS activity may be increased
by addition of only 50 µmol lead/litre and ALAD activity is decreased
by 60% at a concentration of 0.5 µmol/litre (Kusell et al., 1978;
Dresner et al., 1982). Lead (5 µmol/litre) also inhibits (20%)
porphobilinogen deaminase in red blood cell haemolysates at
5 µmol/litre (Piper & Tephly, 1974). Fowler et al. (1980) have shown
that disruption of haem synthesis results in reduction of tissue haem
levels. Increased exposure to lead decreases the content and function
of haem-dependent enzymes of the P-450 mono-oxygenase system (Meredith
& Moore, 1979). More recently it has been shown that lead induces haem
oxygenase activity thereby increasing the degradation of haemoproteins
which may adversely affect a number of cell functions such as
respiration and energy production (Maines, 1992). It has been
suggested that delay in the accumulation of haemoproteins of the
respiratory chain in brain tissue during development may result in
decreased synthesis of haem enzymes in the brain (Bull, 1980; Moore et
al., 1987). Holtzman et al. (1981), on the other hand, found no effect
of lead on brain cytochromes in rat pups with impairment of growth due
to exposure to lead.
7.2 Nervous system effects
7.2.1 Higher order behavioural toxicity
Experimental studies using animal models have demonstrated that
lead impairs learning and memory functions at virtually all stages of
the life cycle. The most significant studies have focused on
behavioural and learning impairments in experimental animals with PbB
levels below 1.44 µmol/litre (30 µg/dl). Bushnell & Levin (1983) fed
post-weaning rats drinking-water containing 10 or 100 mg lead (as lead
acetate) per litre for up to 7 weeks. PbB was not determined but has
been estimated from comparisons with similar lead exposure models to
have been less than 0.96 µmol/litre (20 µg/dl) (Davis et al., 1990b).
Brain lead averaged about 0.05 µg/g wet weight. The rats exhibited
impaired learning ability when tested for their ability to choose
between alternate arms of a radial maze. Cory-Slechta et al. (1985)
demonstrated a significant learning impairment in rats given lead
acetate in drinking-water (25 mg/litre) from weaning. The observed
outcome was a significantly higher response rate in lead-exposed rats
working under a fixed interval schedule of food reinforcement. PbB
values of lead-treated rats in that study averaged
0.72-0.96 µmol/litre (15-20 µg/dl) and brain lead levels averaged
0.07 µg/g wet weight. Similar effects have been described in older
rats (16 months of age) at steady-state PbB levels of
0.62-0.86 µmol/litre (13-18 µg/dl) (Cory-Slechta & Pokora, 1991). More
recently, Cohn et al. (1993) demonstrated selective effects of lead on
learning processes, as distinct from non-specific effects such as
motivational levels, and sensory or motor impairment using a multiple
schedule of repeated learning and performance. Exposure to lead
acetate in drinking-water (50 mg/litre) produced a PbB of
1.2 µmol/litre (25 µg/dl).
Winneke et al. (1977) exposed rats to lead while in utero,
through mother's milk and directly in drinking-water. Their learning
ability was then tested by requiring the animals to discriminate
between stimuli of either different orientation (stripes as an easy
task) or different size (discs as a difficult task). Adult rats tested
between 90 and 170 days of age with PbB levels of less than
1.44 µmol/litre (< 30 µg/dl) were slower to learn the difficult task
of size discrimination (but not the easy discrimination problem) and
tended to repeat more errors than control subjects.
Altmann et al. (1993) demonstrated deficits of both active
avoidance learning (AAL) and in vitro hippocampal long-term
potentiation (LTP) in adult rats that had received pre-weaning or pre-
and post-weaning dietary lead exposure to achieve PbB levels of
approximately 0.72 µmol/litre (15 µg/dl) and brain lead concentrations
of 0.09-0.16 µg/g wet weight. Deficit in animals with only
post-weaning exposure and blood lead levels was about 0.77 µmol/litre
(16 µg/dl) but brain lead concentrations of only 0.09 µg/g was either
absent (in the case of LTP) or markedly reduced (in the case of AAL).
Similar types of effects have been noted in studies using
non-human primates. For example, Rice (1985) reported deficits of
discrimination reversal performance in monkeys dosed orally with 0, 50
or 100 µg lead/kg body weight per day for the first 200 days of life.
At the cessation of dosing, PbB concentrations were 0.144 µmol/litre
(3 µg/dl), 0.72 µmol/litre (15 µg/dl) and 1.2 µmol/litre (25 µg/dl),
respectively. Prior to behavioural testing (3 years of age), PbB
levels were 0.144 µmol/litre (3 µg/dl), 0.528 µmol/litre (11 µg/dl)
and 0.624 µmol/litre (13 µg/dl). Additional evidence for lead-induced
changes in learning in non-human primates is provided by the study of
Lilienthal et al. (1990), which showed a dose-related increase in the
percentage of errors in a learning set formation task (discrimination)
with PbB levels in the low exposure group (350 µg lead acetate/g diet)
averaging 1.68 µmol/litre (35 µg/dl). Also, an impairment of reversal
learning was found by Bushnell & Bowman (1979b) to persist in monkeys
up to their fifth year, at which time the PbB level in treated animals
averaged 0.24 µmol/litre (5 µg/dl) compared to 0.192 µmol/litre
(4 µg/dl) in controls. In the study by Bushnell & Bowman (1979a), the
period of lead exposure was limited to the first year of life and
resulted in PbB levels averaging as high as 3.12 µmol/litre
(65 µg/dl). No description was given of quality control procedures to
ensure accuracy of PbB determinations at the lower levels reported.
Several studies on experimental animals have shown perseverative
effects (the tendency to respond repetitively and inappropriately even
though environmental conditions have changed) which may underlie many
of the lead-induced changes in learning and other higher order
behavioural processes noted in such studies. This has been clearly
demonstrated in rodents by Cohn et al. (1993) in the context of
learning impairments. In the study by Bushnell & Levin (1983)
described above, the accuracy impairment derived from a tendency of
lead-exposed rats to re-enter a previously explored arm of the maze.
Similar perseverative tendencies have been described in non-human
primate studies, e.g., on delayed matching to sample (Rice, 1984a) and
delayed alteration tasks (Levin & Bowman, 1986; Rice & Karpinski,
1988).
Experimental animal studies also reveal the importance of task
complexity in detecting lead-induced changes in behaviour, both in
non-human primates (Levin & Bowman, 1983, 1986; Gilbert & Rice, 1987)
and rodents (Winneke et al., 1977, 1982b).
Persistence of lead-induced changes in some higher order
behavioural processes is also suggested by a number of experimental
animal studies. Several reports from the longitudinal studies of
non-human primates by Bowman and his colleagues (e.g., Bushnell &
Bowman, 1979a,b; Levin & Bowman, 1983, 1986, 1989) have demonstrated
persistent neurobehavioural deficits extending up to 8 years after the
termination of exposure and long after PbB levels had declined to the
control level. In rats, Cory-Slechta & Thompson (1979) showed that the
levels of lead exposure determine the persistence of lead effects,
while Cory-Slechta (1990a,b) revealed that apparently transient
effects of lead on behaviour could re-emerge with changes in the
reward contingencies of the environment. Munoz et al. (1986) found
deficits in both spatial and visual discrimination performance of rats
at several months of age resulting from pre-weaning exposure via the
dams. The same outcome was noted by Altmann et al. (1993) in a study
where deficits in adult rats were noted following pre-weaning
exposure.
7.2.2 Mechanisms of lead-induced behavioural toxicity
While the mechanisms underlying lead-induced behavioural toxicity
have yet to be adequately determined, the experimental animal
literature provides suggestive leads. The early experimental studies
of Pentschew (1965) and Pentschew & Garro (1966) demonstrated in the
rodent model that the pathogenesis of acute high-dose encephalopathy
was secondary to increased permeability of capillaries in the brain,
leading to leakage of fluid and red blood cells. These changes are
similar to those occurring in children with acute lead encephalopathy
characterized clinically by coma, convulsions and death, and identify
the brain microvasculature as the primary target for lead in the
central nervous system following high level exposure to lead.
Changes in microvascular morphology are not evident with low
level lead exposure, but it is the vulnerability of the blood-brain
barrier that permits exposure of the brain to lead. Exposure of the
developing fetus to lead results in higher uptake of lead in the brain
than from later exposures (Rossouw et al., 1987). The development of
resistance to acute encephalopathy in rat pups during the first few
days of life is thought by Holtzman et al. (1984) to be related to
maturation of the blood-brain barrier and possibly to the ability of
the older animals to sequester lead in protein complexes. Thus
neuropathological changes may relate more to the issue of exposure
than to mechanism of effect.
More recently, the focus of mechanistic studies has involved
biochemical and neurochemical changes. Biochemical changes in synaptic
transmission are likely to be related to problems in dendrite-nerve
organization and function (Goldstein, 1990). There is continuing
reorganization of dendrite and nerve terminal connections throughout
early months of neural development. A number of biochemical and
functional studies concerning possible mechanisms of lead effect
suggest lead may disrupt this process and its function through early
childhood (e.g., Cookman et al., 1987, 1988).
Other biochemical effects from lead exposure which may be the
basis of important neurobiological mechanisms of leadinduced
behavioural toxicity include changes in protein kinases, with
implications for neurotransmitter system disturbances. At least three
protein kinases present in nerve terminals involved in modulating the
release of neurotransmitters have been shown to be affected by lead.
However, the relevance of these findings to cognitive function has not
been established (Goldstein, 1990).
From both in vivo and in vitro studies, lead exposure has
been reported to have effects on virtually all neurotransmitter
systems (US EPA, 1986a). Depending on the stage of development, these
include dopaminergic, cholinergic, serotonergic, GABAnergic,
glutamatergic and opiate systems. Probably the most extensively
investigated system has been the dopaminergic system where a number of
changes at the biochemical and receptor level have been described. One
of the difficulties, however, is determining the relationship of the
reported changes at the biochemical/receptor level to changes in
behavioural function. McIntosh et al. (1989) noted lead-induced
changes in tetrahydrobiopterin metabolism which may be related to
changes in IQ scores (Blair et al., 1982; McIntosh et al., 1985). In
further support of a functional role for lead-induced changes in
dopaminergic systems, Cory-Slechta & Widzowski (1991) reported
alterations in both D1 and D2 dopaminergic sensitivity in rats with
PbB levels of 1.2-1.44 µmol/litre (25-30 µg/dl).
More recently, studies by Altmann et al. (1993) and Cohn &
Cory-Slechta (1993) suggest a role for changes in the N-methyl-D-
aspartate (NMDA) receptor complex in lead-induced behavioural
toxicity. The NMDA receptor complex has been implicated in the
learning and memory processes.
Effects of lead on mitochondrial energy metabolism may also be
important in the pathogenesis of neurological effects. Impairment of
respiration in brain mitochondria has been observed in in vitro
preparations (Holtzman et al., 1978) and in mitochondria isolated from
brains of lead-exposed rats (Gmerek et al., 1981).
7.2.2.1 Conclusions
Given their ability to establish causal relationships between
lead exposure and biological effects, experimental animal studies can
provide evidence supportive of human epidemiological findings. Many
experimental animal studies have been carried out to characterize the
nature of lead's effects on various target organ systems as well as to
establish the underlying mechanisms of effect. Furthermore, these
studies, by their very nature, are not confounded by such co-variates
of children's IQ as parental IQ, socioeconomic status and quality of
the home environment, or complicated by nutritional inadequacies of
the study population. Earlier experimental animal studies tended to
utilize relatively high lead exposure levels, but over the past ten
years, exposure levels and protocols have been more relevant to the
human situation.
There are, of course, some limitations of experimental animal
studies that must be considered. For example, there may be differences
in species sensitivity as well as differences in pharmacokinetic and
pharmacodynamic behaviour of lead in rodents, primates and humans.
Higher external exposure levels of lead are required to produce PbB
levels corresponding to levels experienced in human populations, and
higher PbBs are necessary to induce encephalopathy in rodents than are
associated with such effects in humans. Such differences in
sensitivity obviously reflect, at least in part, differences in
kinetics of lead across species. Despite these differences, it should
be noted that corresponding levels of PbB have been associated with
neuro-behavioural toxicity in both animal models (rodents as well as
non-human primates) and human populations. This comparability of
effect levels, despite the relative insensitivity of the animal
models, suggests that the levels of concern in humans might be even
lower, although quantitative extrapolation is difficult. Although one
cannot presume that the neurotoxic effects of lead in experimental
animals necessarily predict specific effects in humans, the
similarities in neurobehavioural end-points in humans and animals are
sufficient to conclude that neurobehavioural deficits in animals are
at least qualitatively predictive of effects in humans.
7.2.3 Sensory organ toxicity
Visual and auditory functions have been shown to be affected by
lead; other sensory modalities have largely been neglected. Bushnell
et al. (1977b) studied visual acuity in infant rhesus monkeys using
different degrees of illumination. Four control animals and three
animals each in a low and a high lead group were tested. The animals
were orally dosed between postnatal days 5 and 365. Although the
dosing regimen of this group was intended to produce blood lead levels
around 4.08 µmol/litre (85 µg/dl), peak levels of between 6.58 and
14.4 µmol/litre (137 and 300 µg/dl) occurred between the fifth and the
ninth weeks of dosing. Significant impairment of scotopic vision was
observed only in the animals of the high lead group.
Using flash-evoked visual potentials (VEP), Lilienthal et al.
(1986) studied visual function in 7- to 7´-year-old rhesus monkeys
pre- and postnatally exposed to lead acetate (0, 350 and 600 mg/kg) in
their diets. Seventeen animals were tested altogether: six controls,
and five or six in each of the exposed groups. PbB levels fluctuated
around 1.92 µmol/litre (40 µg/dl) in the group fed 350 mg/kg diet
during the seven postnatal years, whereas in the group fed 600 mg/kg
they had declined from the initial average of 5.28 µmol/litre
(110 µg/dl) at age 9 months to about 2.88 µmol/litre (60 µg/dl) at 7
years of age. PbB levels in the mothers of both these groups had been
between 1.15 and 1.78 µmol/litre (24 to 37 µg/dl) during pregnancy. A
dose-related decrease of amplitudes and a similar increase of
latencies for the main VEP component were found; these were, in most
instances, significant in both the high and low lead groups.
These findings are supported by the results of studies on rodents
by Fox et al. (1977). Prolonged VEP latencies were observed following
chronic developmental lead exposure. Lower VEP amplitudes have also
been observed in rats at PbB levels exceeding 1.44 µmol/litre
(30 µg/dl) (Winneke, 1979). in vitro studies suggest that rods are
more sensitive to the effects of lead than are cones (Fox & Sillman,
1979).
Information on the impairment of auditory function by lead comes
from electrophysiological studies using brainstem auditory evoked
potentials (BAEP) in rhesus monkeys (Lilienthal et al., 1990). PbB
levels in the various groups at the time of testing averaged
0.432 µmol/litre (9 µg/dl; controls), 1.92 µmol/litre (40 µg/dl; low
lead), and 2.688 µmol/litre (56 µg/dl; high lead). Of the four peaks
discernible in the monkey, BAEP peaks II and IV exhibited
significantly prolonged latencies, which were observed only in the
animals of the high lead group.
7.3 Renal system
The renal effects of lead in animal models occur as a result of
both acute and chronic exposures. Acute lead nephrotoxicity is
characterized by decreased reabsorption of small molecular weight
compounds by the renal tubule, particularly amino acids, glucose and
phosphate (Fanconi Syndrome).
Morphological changes of chronic lead exposure include
cytomegaly, development of nuclear inclusion bodies and
ultrastructural changes in mitochondria. The basis for the cytomegaly
is not understood but it is known that there is altered homoeostasis
of water and electrolytes and cellular swelling. Inclusion bodies are
lead-protein complexes composed of acidic non-histone proteins. As
much as 90% of lead in the kidney has been shown to be contained in
the inclusion bodies, suggesting that they provide a detoxification
function. The origin of the protein is not known but Egle & Shelton
(1986) have identified the most abundant component of isolated
inclusion bodies to be a constitutive protein of the adult central
nervous system primarily in the cerebral cortex. The lead in the
inclusion bodies is chelatable with EDTA (Goyer & Wilson, 1975).
Mitochondria isolated from kidneys of lead-intoxicated rats have
impaired respiration and oxidative phosphorylation capacity, (Goyer &
Rhyne, 1973).
Kholil-Manesh et al. (1992) studied the evaluations of renal
histological and functional changes in rats continuously dosed with
high dose lead. Glomerular filtration rate (GFR) was significantly
increased at 3 months, but significantly decreased at 12 months. Lead
inclusion bodies were noted throughout the duration of the study.
Tubular atrophy and intestinal fibrosis appeared at 6 months. The
brush border of proximal tubular cells was disrupted at 1 and 3
months, but recovered later.
With continued exposure to lead, acute nephropathy may progress
to chronic interstitial nephritis that does not have any unique or
distinguishing features. There is progressive increase in interstitial
fibrosis, dilatation of tubules and formation of microcysts with
hyperplasia of tubular epithelial cells. In the rat, inclusion bodies
are reduced in number and may be entirely absent in later stages of
the nephropathy. Glomerular sclerosis occurs with onset of proteinuria
and renal failure (Goyer & Rhyne, 1973).
The sequence of morphological changes observed in experimental
models is thought to be generally true for humans. Experimental
studies in rats suggest that there may be a threshold for lead renal
toxicity in the rat at PbB levels similar to those observed for
humans. In rats exposed to different doses of lead for up to 12 weeks,
a PbB level of 2.88 µmol/litre (60 µg/dl) appears to be the threshold
for proximal renal tubular cell injury by lead (Goyer et al., 1989).
This PbB level is equivalent to kidney lead levels of about 45 mg/kg
wet weight and is the level of lead at which excretion of renal
calcium increases and ultrastructural changes occur in proximal
tubular cell mitochondria. This finding is consistent with the
observation in humans by Buchet et al. (1980) and Gennart et al.
(1992) of a threshold value of 2.88 µmol/litre (60 µg/dl) of PbB which
leads to adverse renal effects by lead.
7.4 Cardiovascular system
Early studies concerning the production of hypertension in
experimental animals, summarized in Environmental Health Criteria 3:
Lead (IPCS, 1977), were conflicting. Among rats given 70 mg of lead
acetate per day orally, only a few survived 40 days and all were
hypertensive (Griffith & Landaver, 1944). On the other hand, other
studies did not reveal a blood pressure effect from high-level lead
exposure in rats (Padilla et al., 1969) or in dogs (Fouts & Page,
1942). More recently, several experimental studies have confirmed that
lead can produce hypertension, and evidence for several plausible
mechanisms has been provided. Rats, both normotensive and
spontaneously hypertensive, exposed to lead in water supplies for up
to a year, resulting in PbB levels of up to 0.6 µmol/litre
(12.5 µg/dl), exhibited ventricular tachycardia and ventricular
fibrillation (Evis et al., 1985). Subsequent studies (Evis et al.,
1987) showed that adrenaline had a significant arrhythmogenic effect
in hypertensive animals. It was concluded that chronic exposure to
lead, when combined with high blood pressure, slightly enhances the
susceptibility of the heart to arrhythmias induced by myocardial
ischaemia. Overviews of the experimental evidence related to
hypertension and lead exposure have been published by Victery (1988)
and US EPA (1986a, 1989). Taken as a whole, these studies demonstrate
that increase in blood pressure does occur secondary to renal failure
in rats with continuous high-level exposure to lead. More recent
studies on effects of chronic low-level exposure to lead on blood
pressure in rats have shown alterations in cardiovascular parameters
in the PbB range of 0.24-1.92 µmol/litre (5-40 µg/dl), and provide
some insights as to mechanisms involved in the pathogenesis of
lead-related cardiovascular effects.
Victery et al. (1982a) exposed rats to lead in utero by giving
dams drinking-water containing 0, 5, and 25 µg lead/ml and continued
this regimen to the pups for 5 to 6 months. Although no change in
blood pressure was noted at these levels of exposure, significant
changes in the renin-angiotensin system were reported in animals given
water containing 25 µg lead/ml with a PbB level of 0.864 µmol/litre
(18 µg/dl). In a similar study (Victery et al., 1982b), exposure to
100 µg lead/ml in drinking-water (but not 500 µg/ml) produced a
significant (17 mmHg) elevation in blood pressure beginning at 3´
months of age and continuing until 6 months of age. Chai & Webb (1988)
found an elevation of 15 to 20 mmHg in the systolic blood pressure of
rats given drinking-water containing 100 µg lead/ml. These authors
suggest that alterations in the cellular mechanisms that regulate
intracellular calcium concentration may enhance pressor responsiveness
to catacholamines. Boscolo & Carmignani (1988) found raised blood
pressure in rats with exposure to drinking-water containing 0, 30 or
60 µg lead/ml for 18 months. Cardiovascular responses to blood
pressure agonists indicated that lead exposure affects the
renin-angiotensin system and induces sympathetic hyperactivity by
acting on central and peripheral sympathetic junctions, increasing the
responsiveness to stimulation of alpha-2-adrenergic receptors, and by
increasing sensitivity to stimulation of cardiac and vascular
beta-adrenergic and dopaminergic receptors.
From the experimental evidence summarized by Victery (1988), it
appears that low-level exposure to lead produces an elevation in blood
pressure. The failure to demonstrate increased blood pressure in some
studies with high-level exposure to lead suggests that the effect of
lead on blood pressure may be biphasic, i.e. a consistent effect with
low-level exposure but inconsistent effects with high-level exposure.
7.5 Reproductive system
Experimental studies on the effects of lead on the reproductive
system most often concern toxicity to either the male or female but
have addressed results of exposure to both parents. Environmental
Health Criteria 3: Lead (IPCS, 1977) identified effects on
spermatogenesis in rats exposed to lead and showed that high maternal
exposure to lead in rats can reduce numbers and size of offspring.
There may also be paternally transmitted effects resulting in
reductions of litter size, weights of offspring and in survival rate.
Few studies into the effects of lead on male sexual function have
reported PbB levels. Ivanova-Chemishanska et al. (1980) reported
changes in levels of enzymatic activity and ATP in testicular
homogenates from rats given 0.0001 or 0.01% solutions of lead acetate
as drinking-water over a 4-month period. Chowdhury et al. (1984) found
testicular atrophy along with cellular degeneration in rats with PbB
levels over 3.36 µmol/litre (70 µg/dl), but not in rats with levels of
2.59 µmol/litre (54 µg/dl). Donovan et al. (1980) found that lead
inhibited androgen binding by cytosolic receptors in mouse prostate.
Testicular homogenates from 2- to 3-week-old male offspring (PbB
levels 0.30 µmol/litre, 6.3 µg/dl) of lead-exposed female rats showed
a decreased ability to metabolize progesterone (Wiebe et al., 1982).
In an in vitro study, Wiebe et al. (1983) found a 10 to 20% decrease
in FSH binding and in the production of cyclic AMP by Sertoli cells
isolated from prepubertal rats and cultured in the presence of lead
acetate (2.64 × 10-4 mol/litre). In addition, the activity of
cellular 3ß-hydroxysteroid dehydrogenase was decreased. It was shown
by Sokol et al. (1985) that there is a dose-related suppression of
serum testosterone levels and spermatogens in adult rats (100 day of
age) given a solution of 0.3% sodium acetate as drinking-water for up
to 60 days. PbB concentrations were between 1.44 and 2.40 µmol/litre
(30 and 50 µg/dl), depending on the length of treatment. Further
studies (Sokol, 1987) supported the hypothesis that lead disrupts the
hypothalamic control of pituitary hormone secretion. However, other
evidence indicates that lead may directly or indirectly affect
testicular enzymes or may act indirectly by a reduction in testicular
binding of FSH and production of cyclic AMP (US EPA, 1986a).
Dosing mature female rats with lead in order to produce PbB
concentrations of 1.44 µmol/litre (30 µg/dl) resulted in irregular
estrous cycles. At a PbB level of 2.54 µmol/litre (53 µg/dl), animals
developed follicular cysts and there was a reduction in the number of
corpora lutea (Hilderbrand et al., 1973). Grant et al. (1980) reported
delayed vaginal opening in rats whose mothers were given drinking-
water containing 25, 50 and 250 µg lead/ml. The vaginal opening delays
in the group given 25 µg lead/ml of drinking-water occurred in the
absence of any growth retardation or other developmental delays and
were associated with median PbB levels of 0.86-1.39 µmol/litre
(18-29 µg/dl). Studies on female monkeys have shown that pre- and/or
postnatal exposure to lead can affect pubertal progression and
hypothalamicpituitary-ovarian-uterine functions. Chronic exposure of
nulliparous female monkeys to lead (PbB levels of approximately
1.68 µmol/litre, 35 µg/dl) resulted in subclinical suppression of
circulating luteinizing and follicle stimulating hormone and estradiol
without producing overt effects on general health or menstruation
(Foster, 1992).
7.6 Effects on bone
There is growing interest in lead in bone for several reasons.
Bone is a store for lead accumulated from past exposure. It has a long
biological half-life but may be mobilized and contribute to blood lead
during pregnancy. With the development of X-ray fluorescence
techniques to measure lead in bone in vivo, there is a need to
improve our understanding of bone lead metabolism and of factors that
influence lead retention and release. In addition, lead may adversely
affect bone metabolism, particularly in post-menopausal women, and
contribute to the development of osteoporosis. There has only been
limited experimental study of these concerns to date, but there are
some relevant reports in the literature.
A summary of much of the currently available literature on the
potential toxicological implications of lead in bone during pregnancy
and lactation is contained in a review by Silbergeld (1991). Toward
the latter half of pregnancy in mice, there seems to be a preferential
transfer of lead across the placenta to the fetus (Danielson et al.,
1983). In rats exposed to lead for 150 days and then not exposed for
50 days prior to mating, Buchet et al. (1977) found that there was a
substantial mobilization of lead from mother to fetus. Keller &
Doherty (1980b) found using radiotracer lead (210Pb) in female mice
that there was also a major transfer of lead to the pup during
lactation. The concomitant decrease in maternal bone lead supports the
hypothesis that bone resorption of lead occurs during lactation.
It has been shown that lead may directly and indirectly affect
various aspects of bone metabolism (Pounds et al., 1991). Lead
inhibits the renal enzyme 1-hydroxylase, reducing plasma levels of
1,2-dihydroxychole-calciferol (activated vitamin D). Lead impairs the
Haversian remodelling system in beagle dogs chronically exposed to
lead (Anderson & Danylchuk, 1977).
7.7 Immunological effects
The effects of lead on the immune system are diverse but not well
documented. Lead reduces resistance and increases mortality of
experimental animals when they are infected by a broad range of
bacterial and viral agents (Koller, 1984). Lead impairs antibody
production in animals and generally decreases immunoglobin plaque-
forming cells (Koller & Roan, 1980).
7.8 Mutagenicity
Lead is thought to have genotoxic properties. However, lead-
induced gene mutations in cultures of mammalian cells have only been
observed at concentrations toxic to the cells. Studies for point
mutations in bacterial systems have also yielded negative results (US
EPA, 1986a). Zelikoff et al. (1988) found that both insoluble lead
sulfide and more soluble lead nitrate were mutagenic when added to
Chinese hamster V79 cells. A 6-fold increase in mutation frequency was
noted at a lead nitrate level of 500 µmol/litre medium. These authors
also found that lead acetate induced morphological transformation of
Syrian hamster cells. However, they concluded that these effects may
not have been the result of direct damage to DNA but may have occurred
via indirect mechanisms including disturbances in enzyme functions
important in DNA synthesis and/or repair.
Studies on the production of chromosome aberrations, sister-
chromatid exchanges and micronuclei by lead, whether in in vitro
cultures or in vivo, have given mixed results, and summaries are
available (US EPA, 1986a; IARC, 1987a,b; ATSDR, 1991).
7.9 Carcinogenicity
There have been several experimental studies in rats and mice in
which long-term administration of a lead compound in food or
drinking-water or parental administration has produced tumours of the
kidney (Van Esch & Kroes, 1969; Moore & Meredith, 1979). These and
other studies have been discussed in detail in IARC (1980) and
summarized in US EPA (1986a). In general, methodological problems,
including dose levels, number of animals, and doses, and lack of
toxicity monitoring, make many of the studies difficult to interpret
in a quantitative manner. One study (Azar et al., 1973) addressed some
of these problems and reported an increase in the numbers of renal
tumours in rats fed diets containing 500 mg lead/kg for 2 years. PbB
levels were 3.84 µmol/litre (80 µg/dl). In all studies renal
carcinogenicity occurred against a background of proximal tubular cell
hyperplasia, cytomegaly and cellular dysplasia in response to the high
doses of lead and long exposure times (Goyer, 1985). Renal
adenocarcinoma occurred in a high percentage of lead-exposed animals
and the incidence of tumours was related to the length and severity of
exposure (Mao & Molnar, 1967). Males appear to be more susceptible to
tumours than females. The maximum dose of lead in drinking-water, not
associated with any morphological or functional evidence of renal
toxicity in rats fed a diet containing adequate levels of trace
minerals, particularly calcium, is 200 mg lead/litre (Goyer et al.,
1970). No evidence of renal tumours at doses below this value has been
reported (Azar et al., 1973; US NCI, 1979).
Several hypotheses have been proposed for the mechanism of lead
carcinogenicity in experimental animals. These include mutagenicity,
cellular proliferation, nuclear protein (inclusion bodies), promoter
activity, activation of protein kinase C, and cystic hyperplasia
(Goyer, 1993). Lead is a weak mutagen in mammalian cell systems, but
is a strong mitogen. Exposure to a single intraperitoneal injection of
lead acetate in rats stimulates a 40-fold increase in cell
proliferation as measured by autoradiography, and this is further
increased by unilateral nephrectomy (Choie & Richter, 1972). DNA
synthesis in kidneys, as measured by 3H-thymidine incorporation, is
increased 15-fold and the mitotic index 45-fold following a single
intracardiac injection of lead acetate in mice (Choie & Richter,
1974). Cell proliferation and hyperplasia are seen in the liver of
rats given a single intravenous injection of lead nitrate (Columbano
et al., 1984). In both of these studies, increases in cell
proliferation occurred in the absence of cellular necrosis, suggesting
that this was a mitogenic rather than regenerative response.
Activation of protein kinase C and formation of nuclear inclusion
bodies or lead protein complexes are additional events that may
influence regulation of cell growth and development and play a role in
the carcinogenic response in experimental animals resulting from
administration of lead (Goyer, 1993). Cystic hyperplasia, a late
morphological manifestation of chronic lead nephropathy, is a risk
factor for renal cancer (Bernstein et al., 1987). Prior to adenoma
formation in animals treated with renal carcinogens, cystic
hyperplasia was reported (Dees et al., 1980; Goyer et al., 1981).
8. EFFECTS ON HUMANS
Despite the long recognition of lead poisoning, new clinical
cases continue to be the subject of published reports. Although
important, they affect a comparatively small proportion of the
population at risk from the potential effects of exposure to
environmental lead.
Over the last 10-15 years, particular attention has been directed
towards epidemiological studies designed to evaluate the possible
neurotoxic effects of lead on the developing child, especially delayed
or impaired neurobehavioural development and performance.
In the adult population, considerable attention has been directed
towards evaluating cardiovascular effects and the implication of lead
in hypertension.
New studies have contributed to our understanding of the
biochemical effects of lead, and may facilitate early recognition of
significant change and mitigation of potentially adverse outcomes.
The effects of lead have been studied widely in both the general
population and in those exposed occupationally. Since these effects
are the same in both settings, no distinction has generally been made
in the discussion. However, it is often important to distinguish
between adults and children because of different susceptibility.
8.1 Biochemical effects of lead
Lead is known to affect a number of enzymes and physiological
systems which result in a wide variety of changes in humans. While
those affecting the haematopoietic system are well known, there are
others which need to be considered in the risk assessment process and
are considered in this section.
In considering the effects of lead on biochemical systems, it is
appropriate to discuss the form of the lead in the various body
compartments.
Blood lead (PbB) is distributed between the plasma and the
erythrocyte. There is less than 1% in the plasma for PbB levels of up
to 4.8 µmol/litre (100 µg/dl) (Manton & Cook, 1984). The curvilinear
relationship of serum lead to blood lead is shown in Fig. 12. The data
show that the erythrocytes have a capacity to bind lead up to PbB
levels of about 2.4 µmol/litre (50 µg/dl). Above this level a fairly
rapid increase in the serum levels occurs.
It should be noted that the proportion of "free" (i.e. unbound)
lead in blood is important in relation to biological activity. Lead is
bound to haemoglobin in blood and has a greater affinity for fetal
than adult haemoglobin (Ong & Lee, 1980). It may, therefore, be
 important to consider the proportion of fetal haemoglobin present in
blood samples from mothers and infants in assessing PbB concentrations
in relation to biological effects. Also, increased fetal haemoglobin
has been found in cases of human and experimental animal poisoning
(Albahary, 1972).
8.1.1 Haem synthesis
Lead is known to affect several enzymatic reactions critical in
haem synthesis, causing abnormal concentrations of haem precursors in
blood and urine. These effects of lead on haem synthesis are shown in
Fig. 13.
As shown in Fig. 13, lead inhibits the activity of three enzymes
of the biosynthetic pathway, 5-aminolaevulinate dehydratase (ALA-D),
coproporphyrinogen oxidase (COPRO-O) and ferrochelatase (FERRO-C).
This depletes haem synthesis and depresses the synthesis of the
initial and rate-limiting enzyme 5-aminolaevulinate (ALA) synthase. As
a consequence there is increased production and excretion of the
precursors ALA and coproporphyrin (COPRO) with increased circulatory
protoporphyrin (PROTO) usually bound to zinc. In the red cell,
diminished synthesis of monooxygenases (cytochromes P450) compromises
drug oxidation and lead is bound to haemoglobin.
8.1.1.1 Protoporphyrin levels
Lead interferes with the conversion of protoporphyrin to haem by
ferrochelatase. The protoporphyrin exists under these circumstances
primarily as zinc protoporphyrin, with a proportion remaining free
(Chisolm & Brown, 1979).
The relationship between protoporphyrin, either free or as the
zinc chelate, in blood and blood lead is one which could be
interpreted as showing a "threshold of effect" or as a continuum of
effect. The exact mathematical relationship is inevitably the choice
of the investigator since the uncertainties in the measurement of
blood lead and blood protoporphyrin would allow the fitting of the
data to either of these relationships. The threshold concept is
satisfactory to those that seek the "no-effect level" (Succop et al.,
1989), since it provides the start point of an analytical
identification process. The continuum of effect is, however, much more
plausible in a biological sense, since the idea of a level at which
"no effect" will occur is unlikely in a biological system.
The figures for the relationship between PbB and zinc
protoporphyrin are confused in any population by iron status (Marcus &
Schwartz, 1987), and it is thus unlikely that measurement of zinc
protoporphyrin, either alone or compared with haemoglobin status, will
provide an accurate estimation of PbB levels less than 0.96 µmol/litre
(20 µg/dl). In a study of the relationship between PbB levels and
erythrocyte protoporphyrin concentration for 2004 urban children
important to consider the proportion of fetal haemoglobin present in
blood samples from mothers and infants in assessing PbB concentrations
in relation to biological effects. Also, increased fetal haemoglobin
has been found in cases of human and experimental animal poisoning
(Albahary, 1972).
8.1.1 Haem synthesis
Lead is known to affect several enzymatic reactions critical in
haem synthesis, causing abnormal concentrations of haem precursors in
blood and urine. These effects of lead on haem synthesis are shown in
Fig. 13.
As shown in Fig. 13, lead inhibits the activity of three enzymes
of the biosynthetic pathway, 5-aminolaevulinate dehydratase (ALA-D),
coproporphyrinogen oxidase (COPRO-O) and ferrochelatase (FERRO-C).
This depletes haem synthesis and depresses the synthesis of the
initial and rate-limiting enzyme 5-aminolaevulinate (ALA) synthase. As
a consequence there is increased production and excretion of the
precursors ALA and coproporphyrin (COPRO) with increased circulatory
protoporphyrin (PROTO) usually bound to zinc. In the red cell,
diminished synthesis of monooxygenases (cytochromes P450) compromises
drug oxidation and lead is bound to haemoglobin.
8.1.1.1 Protoporphyrin levels
Lead interferes with the conversion of protoporphyrin to haem by
ferrochelatase. The protoporphyrin exists under these circumstances
primarily as zinc protoporphyrin, with a proportion remaining free
(Chisolm & Brown, 1979).
The relationship between protoporphyrin, either free or as the
zinc chelate, in blood and blood lead is one which could be
interpreted as showing a "threshold of effect" or as a continuum of
effect. The exact mathematical relationship is inevitably the choice
of the investigator since the uncertainties in the measurement of
blood lead and blood protoporphyrin would allow the fitting of the
data to either of these relationships. The threshold concept is
satisfactory to those that seek the "no-effect level" (Succop et al.,
1989), since it provides the start point of an analytical
identification process. The continuum of effect is, however, much more
plausible in a biological sense, since the idea of a level at which
"no effect" will occur is unlikely in a biological system.
The figures for the relationship between PbB and zinc
protoporphyrin are confused in any population by iron status (Marcus &
Schwartz, 1987), and it is thus unlikely that measurement of zinc
protoporphyrin, either alone or compared with haemoglobin status, will
provide an accurate estimation of PbB levels less than 0.96 µmol/litre
(20 µg/dl). In a study of the relationship between PbB levels and
erythrocyte protoporphyrin concentration for 2004 urban children
 (Piomelli et al., 1982), it was concluded that a threshold is apparent
and occurs at a PbB level of between 0.72 and 0.86 µmol/litre (15 and
18 µg/dl). This is consistent with the data of Roels et al. (1976),
which show a clear discontinuity at around 1.2 µmol/litre (25 µg/dl).
Marcus & Schwartz (1987) re-analysed data from the NHANES II survey of
264 children. No positive correlation between PbB and zinc
protoporphyrin was noted below 0.96 µmol/litre (20 µg/dl).
Other reports which present relevant data are Roels et al.
(1976); Piomelli et al. (1982); Hammond et al. (1985); Rabinowitz et
al. (1986); Roels & Lauwerys (1987). In the presence of iron
deficiency the observed threshold is likely to be lower (Mahaffey &
Annest 1986; Marcus & Schwartz, 1987). At first sight it would seem
inappropriate to consider thresholds that are determined in the
presence of potential iron deficiency.
A study by Koren et al. (1990) of maternal and umbilical cord
lead and free erythrocyte porphyrin (FEP) levels for 95 mother-infant
pairs showed a correlation between maternal and cord PbB, with
maternal levels exceeding neonatal levels. Most cord PbB levels were
below 0.33 µmol/litre (7 µg/dl), and 11 were below the detection
limit. The cord blood FEP (0.86 µmol/litre) level was consistently
higher than maternal FEP (0.53 µmol/litre) but was not statistically
correlated. The elevated cord blood FEP values were attributable to
immature haem synthesis and high erythrocyte volume rather than the
presence of lead.
8.1.1.2 Coproporphyrin levels
One of the earliest observed effects of lead poisoning was a rise
in coproporphyrin excretion in the urine, due to inhibition of
coproporphyrinogen oxidase (Campbell et al., 1977). Although often
cited as a good measure of lead exposure it is clear that the
importance of the measure of current environmental levels of exposure
is small. This is because the excretion levels do not rise
significantly until the PbB excretion is greater than 1.92 µmol/litre
(40 µg/dl) (Meredith et al., 1978).
8.1.1.3 delta-Aminolaevulinic acid levels in urine and blood
Like protoporphyrin, circulating and excreted levels of ALA are
likely to be best described as a continuum of effect. Elevated levels
of this compound are of importance since neurological features of lead
exposure have been ascribed in part to increased circulating levels of
ALA (Moore et al., 1987). The rise in concentration during lead
exposure is a function first of decreased activity of ALA dehydratase
(ALAD), which is uniquely sensitive to lead toxicity, and subsequently
of increased activity of the initial and rate-limiting enzyme of haem
biosynthesis, ALA synthase (Meredith et al., 1978). It is
inappropriate to discuss haem biosynthesis control mechanisms here;
suffice it to say that blocking this pathway by lead lowers free haem
levels, which feedback to ALA synthase (Moore et al., 1987). The
special relationship between ALAD activity and lead exposure has been
best described as a negative exponential and has been used as a
measure of lead exposure in population surveys (Berlin & Schaller,
1974).
The immediate effect of the inhibition of ALAD will be an
increased level of ALA in the blood, which will then lead to increased
urinary excretion. The plasma levels of ALA are elevated in the
presence of higher lead levels. This has been seen in a number of
studies (Haeger-Aronsen, 1960; Meredith et al., 1978; O'Flaherty et
al., 1980).
Meredith et al. (1978) measured ALA metabolism in 48 male
lead-exposed workers (aged 22-56 years, PbB 4.2 ± 1.4 µmol/litre,
87 ± 29 µg/dl), who were compared with control subjects (28 male, 9
female, 18-52 years of age, PbB 1.3 ± 0.4 µmol/litre, 27 ± 3.3 µg/dl).
They found increasing levels of circulating ALA associated with PbB
that reached a plateau when the PbB level was in excess of
3 µmol/litre (62 µg/dl) and ALA exceeded 4 µmol/litre. At higher blood
ALA levels, urinary excretion of ALA increased exponentially,
consistent with decreased tubular reabsorption. The authors suggested
that there was a "critical" tissue lead concentration of around
2 µmol/litre (41 µg/dl). This study showed some continuity of the
correlation down to the lowest PbB value of the control group, namely
0.86 µmol/litre (18 µg/dl). However, these data were interpreted to
show that effects are only demonstrable above a PbB level of
1.44 µmol/litre (30 µg/dl).
Other studies show direct correlations between PbB level and
urinary ALA (Selander & Cramer, 1970; Lauwerys et al., 1974), although
these correlations are not seen as low as those in the study of
Meredith et al. (1978). The data of Selander & Cramer (1970) showed a
clear threshold effect at about 1.02 µmol/litre (40 µg/dl) in
occupational subjects.
Roels et al. (1976) reported data over a range of PbB levels from
0.24 to 1.92 µmol/litre (5 to 40 µg/dl) in children which showed
essentially no correlation with urinary ALA.
Data obtained from 39 men and 36 women in the general population
showed that increased urinary excretion of ALA occurred at PbB levels
of more than 1.68 µmol/litre (35 µg/dl) in women and more than
2.16 µmol/litre (45 µg/dl) in men (Roels & Lauwerys, 1987). The
sensitivity of the haem synthesis pathway to increased lead exposure
was in the order: children > women > men.
On balance, it would appear that lead has discernible effects on
the urine level of ALA at a PbB level of around 1.68 µmol/litre
(35 µg/dl).
8.1.1.4 delta-Aminolaevulinic acid dehydratase levels
delta-Aminolaevulinic acid dehydratase (ALAD) is commonly
measured for its activity in samples of haemolysed blood; the result
may or may not be corrected for haematocrit value. The measure of
enzyme activity will reflect the amount of enzyme present as well as
the effect of inhibition of the enzyme.
Studies in the general population have confirmed the correlation
and the apparent lack of a threshold for inhibition of ALAD in
different age groups and exposure categories (Roels et al., 1976;
Chisolm et al., 1985; Roels & Lauwerys, 1987). A negative linear
relationship between PbB and ALAD activity was found between mothers
and their newborn babies (cord blood); PbB levels ranged from 0.14 to
1.44 µmol/litre (3-30 µg/dl) (Roels et al., 1976). Roels & Lauwerys
(1987) reported a similar relationship in a population of 143 children
aged 10-13 years having PbB levels of 0.19-1.97 µmol/litre
(4.7-41 µg/dl).
In the study of Roels et al. (1976) a large number of children
were studied. Although the authors drew a regression line which
suggested that the effects continued to very low levels, the data
between 0.24-0.72 µmol/litre (5-15 µg/dl) were very scattered, and the
regression at low levels appeared to be rather speculative. It was
considered that the lead level above which an effect level is
demonstrable from these data was 0.48 µmol/litre (10 µg/dl).
8.1.1.5 delta-Aminolaevulinic acid synthase
Aminolaevulinic acid synthase (ALAS) levels are determined by a
feedback mechanism which is dependent on haem levels. There appear to
have been relatively few studies on serum ALAS, although some
consideration is given in a report by Meredith et al. (1978). However,
insufficient data were available to establish effect levels for lead
exposure.
8.1.1.6 Other effects of decreased haem synthesis
The potential impact of a reduction in the body pool of haem and
haem precursors is shown in Fig. 14. It is evident that the multiple
effects on haem metabolism from lead exposure can lead to adverse
effects in organs and systems other than the erythropoietic system.
8.1.2 Vitamin D
Formation of the most important vitamin D metabolite,
1,25-dihydroxyvitamin D, is by 1 alpha-hydroxylation of
25-hydroxyvitamin D in the kidney. This is mediated by
25-hydroxyvitamin D-1 alpla-hydroxylase, a cytochrome P450-dependent
enzyme in the mitochondria of the renal tubules. Serum concentrations
of 1,25-dihydroxyvitamin D are measured in children as an indicator of
the effects of lead on the enzyme system mediating the initial
hydroxylation. However, other factors such as dietary intake and the
physiological needs for calcium and phosphorus, and levels of
calciotropic hormones such as parathyroid hormone, can regulate the
production and circulating concentrations of 1,25-dihydroxy-vitamin D
(Rosen & Chesney, 1983).
Several studies have provided information on the effect of lead
on the circulating concentrations of 1,25-dihydroxyvitamin D. Rosen et
al. (1980) studied children with PbB levels in the range of
1.58-5.76 µmol/litre (33-120 µg/dl). While the most striking decreases
in serum 1,25-dihydroxyvitamin D occurred in children whose PbB level
was 2.97 µmol/litre (62 µg/dl), the effect was considered to be
evident in the range of 1.58-2.64 µmol/litre (33-55 µg/dl) when
compared to an age- and race-matched control group with PbB levels in
the range of 0.48-1.248 µmol/litre (10-26 µg/dl).
Mahaffey et al. (1982) measured serum 1,25-dihydroxyvitamin D in
177 subjects aged 1-16 years. PbB lead measurements were performed in
105 of these children and ranged from 0.576 to 5.76 µmol/litre (12 to
120 µg/dl). A curvilinear relationship between serum
1,25-dihydroxyvitamin D concentration and PbB was noted in children
aged 2 to 3 years. Details of the dietary intake of the subjects were
not available.
Koo et al. (1991) studied 105 children, aged 1 to 3 years and
with detailed lead exposure history from birth, to determine the
effect of chronic low to moderate lead exposure. The average lifetime
PbB concentration was 0.23-1.13 µmol/litre (4.8-23.6 µg/dl) and was
greater than 0.96 µmol/litre (> 20 µg/dl) in only three children.
With a range of concurrent PbB concentrations of 0.29-2.11 µmol/litre
(6-44 µg/dl). The children generally had adequate dietary intakes of
calcium, phosphorus and vitamin D. An effect of lead was found on
serum concentrations of calcium, phosphorus, 25-hydroxyvitamin D,
1,25-dihydroxyvitamin D and parathyroid hormone, and on bone mineral
content. In the presence of adequate nutritional status, lead exposure
at low levels (leading to a PbB level of less than 0.96 µmol/litre or
20 µg/dl) appears to have no demonstrable effect on circulating
concentrations of total and ionized calcium, magnesium, phosphorus,
calciotropic hormones including 1,25-dihydroxyvitamin D, parathyroid
hormone and calcitonin, or bone mineralization as indicated by single
photon absorptiometry. At higher levels there is a demonstrable effect
on 1,25-dihydroxyvitamin D.
8.1.3 Dihydrobiopterin reductase
This enzyme (DHBR) is part of the synthesis/salvage cycle which
controls the hydroxylation of tyrosine, tryptophan and phenylalanine.
It is thus central to the synthesis of the catecholamines. Lead has
been shown to inhibit the synthesis of tetrahydrobiopterin from
dihydrobiopterin by dihydrobiopterin reductase (Leeming & Blair, 1980;
(Piomelli et al., 1982), it was concluded that a threshold is apparent
and occurs at a PbB level of between 0.72 and 0.86 µmol/litre (15 and
18 µg/dl). This is consistent with the data of Roels et al. (1976),
which show a clear discontinuity at around 1.2 µmol/litre (25 µg/dl).
Marcus & Schwartz (1987) re-analysed data from the NHANES II survey of
264 children. No positive correlation between PbB and zinc
protoporphyrin was noted below 0.96 µmol/litre (20 µg/dl).
Other reports which present relevant data are Roels et al.
(1976); Piomelli et al. (1982); Hammond et al. (1985); Rabinowitz et
al. (1986); Roels & Lauwerys (1987). In the presence of iron
deficiency the observed threshold is likely to be lower (Mahaffey &
Annest 1986; Marcus & Schwartz, 1987). At first sight it would seem
inappropriate to consider thresholds that are determined in the
presence of potential iron deficiency.
A study by Koren et al. (1990) of maternal and umbilical cord
lead and free erythrocyte porphyrin (FEP) levels for 95 mother-infant
pairs showed a correlation between maternal and cord PbB, with
maternal levels exceeding neonatal levels. Most cord PbB levels were
below 0.33 µmol/litre (7 µg/dl), and 11 were below the detection
limit. The cord blood FEP (0.86 µmol/litre) level was consistently
higher than maternal FEP (0.53 µmol/litre) but was not statistically
correlated. The elevated cord blood FEP values were attributable to
immature haem synthesis and high erythrocyte volume rather than the
presence of lead.
8.1.1.2 Coproporphyrin levels
One of the earliest observed effects of lead poisoning was a rise
in coproporphyrin excretion in the urine, due to inhibition of
coproporphyrinogen oxidase (Campbell et al., 1977). Although often
cited as a good measure of lead exposure it is clear that the
importance of the measure of current environmental levels of exposure
is small. This is because the excretion levels do not rise
significantly until the PbB excretion is greater than 1.92 µmol/litre
(40 µg/dl) (Meredith et al., 1978).
8.1.1.3 delta-Aminolaevulinic acid levels in urine and blood
Like protoporphyrin, circulating and excreted levels of ALA are
likely to be best described as a continuum of effect. Elevated levels
of this compound are of importance since neurological features of lead
exposure have been ascribed in part to increased circulating levels of
ALA (Moore et al., 1987). The rise in concentration during lead
exposure is a function first of decreased activity of ALA dehydratase
(ALAD), which is uniquely sensitive to lead toxicity, and subsequently
of increased activity of the initial and rate-limiting enzyme of haem
biosynthesis, ALA synthase (Meredith et al., 1978). It is
inappropriate to discuss haem biosynthesis control mechanisms here;
suffice it to say that blocking this pathway by lead lowers free haem
levels, which feedback to ALA synthase (Moore et al., 1987). The
special relationship between ALAD activity and lead exposure has been
best described as a negative exponential and has been used as a
measure of lead exposure in population surveys (Berlin & Schaller,
1974).
The immediate effect of the inhibition of ALAD will be an
increased level of ALA in the blood, which will then lead to increased
urinary excretion. The plasma levels of ALA are elevated in the
presence of higher lead levels. This has been seen in a number of
studies (Haeger-Aronsen, 1960; Meredith et al., 1978; O'Flaherty et
al., 1980).
Meredith et al. (1978) measured ALA metabolism in 48 male
lead-exposed workers (aged 22-56 years, PbB 4.2 ± 1.4 µmol/litre,
87 ± 29 µg/dl), who were compared with control subjects (28 male, 9
female, 18-52 years of age, PbB 1.3 ± 0.4 µmol/litre, 27 ± 3.3 µg/dl).
They found increasing levels of circulating ALA associated with PbB
that reached a plateau when the PbB level was in excess of
3 µmol/litre (62 µg/dl) and ALA exceeded 4 µmol/litre. At higher blood
ALA levels, urinary excretion of ALA increased exponentially,
consistent with decreased tubular reabsorption. The authors suggested
that there was a "critical" tissue lead concentration of around
2 µmol/litre (41 µg/dl). This study showed some continuity of the
correlation down to the lowest PbB value of the control group, namely
0.86 µmol/litre (18 µg/dl). However, these data were interpreted to
show that effects are only demonstrable above a PbB level of
1.44 µmol/litre (30 µg/dl).
Other studies show direct correlations between PbB level and
urinary ALA (Selander & Cramer, 1970; Lauwerys et al., 1974), although
these correlations are not seen as low as those in the study of
Meredith et al. (1978). The data of Selander & Cramer (1970) showed a
clear threshold effect at about 1.02 µmol/litre (40 µg/dl) in
occupational subjects.
Roels et al. (1976) reported data over a range of PbB levels from
0.24 to 1.92 µmol/litre (5 to 40 µg/dl) in children which showed
essentially no correlation with urinary ALA.
Data obtained from 39 men and 36 women in the general population
showed that increased urinary excretion of ALA occurred at PbB levels
of more than 1.68 µmol/litre (35 µg/dl) in women and more than
2.16 µmol/litre (45 µg/dl) in men (Roels & Lauwerys, 1987). The
sensitivity of the haem synthesis pathway to increased lead exposure
was in the order: children > women > men.
On balance, it would appear that lead has discernible effects on
the urine level of ALA at a PbB level of around 1.68 µmol/litre
(35 µg/dl).
8.1.1.4 delta-Aminolaevulinic acid dehydratase levels
delta-Aminolaevulinic acid dehydratase (ALAD) is commonly
measured for its activity in samples of haemolysed blood; the result
may or may not be corrected for haematocrit value. The measure of
enzyme activity will reflect the amount of enzyme present as well as
the effect of inhibition of the enzyme.
Studies in the general population have confirmed the correlation
and the apparent lack of a threshold for inhibition of ALAD in
different age groups and exposure categories (Roels et al., 1976;
Chisolm et al., 1985; Roels & Lauwerys, 1987). A negative linear
relationship between PbB and ALAD activity was found between mothers
and their newborn babies (cord blood); PbB levels ranged from 0.14 to
1.44 µmol/litre (3-30 µg/dl) (Roels et al., 1976). Roels & Lauwerys
(1987) reported a similar relationship in a population of 143 children
aged 10-13 years having PbB levels of 0.19-1.97 µmol/litre
(4.7-41 µg/dl).
In the study of Roels et al. (1976) a large number of children
were studied. Although the authors drew a regression line which
suggested that the effects continued to very low levels, the data
between 0.24-0.72 µmol/litre (5-15 µg/dl) were very scattered, and the
regression at low levels appeared to be rather speculative. It was
considered that the lead level above which an effect level is
demonstrable from these data was 0.48 µmol/litre (10 µg/dl).
8.1.1.5 delta-Aminolaevulinic acid synthase
Aminolaevulinic acid synthase (ALAS) levels are determined by a
feedback mechanism which is dependent on haem levels. There appear to
have been relatively few studies on serum ALAS, although some
consideration is given in a report by Meredith et al. (1978). However,
insufficient data were available to establish effect levels for lead
exposure.
8.1.1.6 Other effects of decreased haem synthesis
The potential impact of a reduction in the body pool of haem and
haem precursors is shown in Fig. 14. It is evident that the multiple
effects on haem metabolism from lead exposure can lead to adverse
effects in organs and systems other than the erythropoietic system.
8.1.2 Vitamin D
Formation of the most important vitamin D metabolite,
1,25-dihydroxyvitamin D, is by 1 alpha-hydroxylation of
25-hydroxyvitamin D in the kidney. This is mediated by
25-hydroxyvitamin D-1 alpla-hydroxylase, a cytochrome P450-dependent
enzyme in the mitochondria of the renal tubules. Serum concentrations
of 1,25-dihydroxyvitamin D are measured in children as an indicator of
the effects of lead on the enzyme system mediating the initial
hydroxylation. However, other factors such as dietary intake and the
physiological needs for calcium and phosphorus, and levels of
calciotropic hormones such as parathyroid hormone, can regulate the
production and circulating concentrations of 1,25-dihydroxy-vitamin D
(Rosen & Chesney, 1983).
Several studies have provided information on the effect of lead
on the circulating concentrations of 1,25-dihydroxyvitamin D. Rosen et
al. (1980) studied children with PbB levels in the range of
1.58-5.76 µmol/litre (33-120 µg/dl). While the most striking decreases
in serum 1,25-dihydroxyvitamin D occurred in children whose PbB level
was 2.97 µmol/litre (62 µg/dl), the effect was considered to be
evident in the range of 1.58-2.64 µmol/litre (33-55 µg/dl) when
compared to an age- and race-matched control group with PbB levels in
the range of 0.48-1.248 µmol/litre (10-26 µg/dl).
Mahaffey et al. (1982) measured serum 1,25-dihydroxyvitamin D in
177 subjects aged 1-16 years. PbB lead measurements were performed in
105 of these children and ranged from 0.576 to 5.76 µmol/litre (12 to
120 µg/dl). A curvilinear relationship between serum
1,25-dihydroxyvitamin D concentration and PbB was noted in children
aged 2 to 3 years. Details of the dietary intake of the subjects were
not available.
Koo et al. (1991) studied 105 children, aged 1 to 3 years and
with detailed lead exposure history from birth, to determine the
effect of chronic low to moderate lead exposure. The average lifetime
PbB concentration was 0.23-1.13 µmol/litre (4.8-23.6 µg/dl) and was
greater than 0.96 µmol/litre (> 20 µg/dl) in only three children.
With a range of concurrent PbB concentrations of 0.29-2.11 µmol/litre
(6-44 µg/dl). The children generally had adequate dietary intakes of
calcium, phosphorus and vitamin D. An effect of lead was found on
serum concentrations of calcium, phosphorus, 25-hydroxyvitamin D,
1,25-dihydroxyvitamin D and parathyroid hormone, and on bone mineral
content. In the presence of adequate nutritional status, lead exposure
at low levels (leading to a PbB level of less than 0.96 µmol/litre or
20 µg/dl) appears to have no demonstrable effect on circulating
concentrations of total and ionized calcium, magnesium, phosphorus,
calciotropic hormones including 1,25-dihydroxyvitamin D, parathyroid
hormone and calcitonin, or bone mineralization as indicated by single
photon absorptiometry. At higher levels there is a demonstrable effect
on 1,25-dihydroxyvitamin D.
8.1.3 Dihydrobiopterin reductase
This enzyme (DHBR) is part of the synthesis/salvage cycle which
controls the hydroxylation of tyrosine, tryptophan and phenylalanine.
It is thus central to the synthesis of the catecholamines. Lead has
been shown to inhibit the synthesis of tetrahydrobiopterin from
dihydrobiopterin by dihydrobiopterin reductase (Leeming & Blair, 1980;
 McIntosh et al., 1985) in rat brain. In humans the exact relationship
between PbB and plasma biopterins has not been properly defined.
8.1.4 Nicotinamide adenine dinucleotide synthetase
A recent study of nicotinamide adenine dinucleotide (NAD)
synthetase in erythrocytes indicates that this enzyme is sensitive to
inhibition by lead (and zinc) and is a sensitive indicator of lead
exposure. Zerez et al. (1990) found NAD synthetase activity reduced in
three subjects with elevated PbB levels in the range
1.63-3.46 µmol/litre (34-72 µg/dl).
8.1.5 Nutritionally affected groups
A body of evidence, summarized and discussed by Mahaffey (1985),
indicates that a high dietary calcium intake tends to decrease lead
absorption and retention in infants, young children and adults. Other
evidence suggests that in groups including children, with
self-selected diets, low dietary calcium intakes are associated with a
greater prevalence of elevated PbB levels (Mahaffey, 1985).
Some human data indicate that adults with low iron states have an
increased absorption of both iron and lead (Watson et al., 1980,
1986). In children, particularly those from low-income families, there
is often an association between iron deficiency and elevated PbB (Yip
& Dallman, 1984; Mahaffey, 1985). While this by itself does not
indicate a causal relationship, when considered in the context of data
on adults and animals, it suggests that iron deficiency in children
can result in increased lead absorption.
Alcoholics and people who consume excess alcohol may be at
increased risk of adverse effects from lead. In animal studies,
alcohol and lead synergistically inhibit ALAD, hepatic glutamic
oxaloacetic transaminase and glutamic pyruvic transaminase activities
(Flora & Tandon, 1987) supporting the hypothesis regarding risks to
humans.
8.2 Haematopoietic system
Lead-induced anaemia can be a direct consequence of inhibition of
haem biosynthesis; it is not necessarily associated with iron
deficiency. It may be associated with alterations of globin syntheses
(Albahary, 1972). More importantly, the synthesis of alpha- and
ß-globin chains may become asynchronous (White & Harvey, 1972).
8.2.1 Anaemia
Based on data published by Lilis et al. (1978), Baker et al.
(1979), Grandjean (1979), and earlier data, the threshold PbB level
for a decrease in haemoglobin has been estimated to be 2.40 µmol/litre
(50 µg/dl) for occupationally exposed adults (US EPA, 1986a).
Grandjean et al. (1989a) demonstrated the reduced ability of the
erythropoietic system to regenerate after blood withdrawal
(0.45 litre) in 25 lead-exposed battery workers (average PbB level
2.14 µmol/litre, 44.5 µg/dl) compared with 25 age-matched controls
(average PbB level 0.35 µmol/litre, 7 µg/dl). The haematological
parameters, except for erythropoietin were otherwise comparable for
the two groups. The effect was attributed to an effect on haem
synthesis and possible decreased erythrocyte survival in lead-exposed
workers. Lead has been found to depress serum levels of
erythropoietin, a hormone which regulates erythrocyte formation
(Graziano et al., 1991), which might also affect the reserve capacity
for erythropoiesis.
The PbB threshold for decreased haemoglobin levels in children is
estimated to be approximately 1.92 µmol/litre (40 µg/dl) (IPCS, 1977).
However, a cross-sectional epidemiological study of 579 children, aged
1-5 years in 1974, living in close proximity to a primary lead smelter
showed that adverse effects on haematocrit may occur at lower PbB
levels (Schwartz et al., 1990). Anaemia, defined as a haematocrit
below 35%, was not found at PbB levels of less than 0.92 µmol/litre
(20 µg/dl). There was a strong non-linear dose-response relationship
at higher PbB levels, which was influenced by age.
Kutbi et al. (1989) studied 200 boys aged 6-8 years. The mean PbB
was 0.33 µmol/litre (6.9 µg/dl; range 0.07-1.14 µmol/litre or
1.4-23.8 µg/dl). Subdividing the group at 0.72 µmol/litre (15 µg/dl),
they found a negative correlation between PbB level and all
haematological values for the "upper normal" (N = 7) compared with the
"normal" group (N = 193). The pattern of haematological parameters was
described as predictive of microcytic anaemia.
Anaemia has been commonly associated with the adverse effects of
occupational lead exposures. It is an effect that is easily diagnosed
clinically and is recognized as a marker of lead toxicity. Anaemia may
result from either a decrease in haemoglobin production or an increase
in the rate of destruction of erythrocytes. An analysis, made in 1974,
of the association between PbB levels and haematocrit in 579 children
(1-5 years of age) living near a primary lead smelter has recently
been presented (Schwartz et al., 1990). A haematocrit value of less
than 35% was used to indicate an adverse effect. It should be noted
that the effect of iron deficiency was not taken into account in
analysing the results. The study concluded that there was no adverse
effect of lead at PbB levels below 0.96 µmol/litre (20 µg/dl).
Furthermore, the risk of having a haematocrit value below 35% for
1-year-olds was 2% at PbB levels between 0.96 and 1.87 µmol/litre
(20 and 39 µg/dl). The degree of iron deficiency may account for a
substantial proportion of this 2%. In this study, the level at which
an effect of lead on the induction of anaemia was demonstrable was
about 1.92 µmol/litre (40 µg/dl).
8.2.2 Pyrimidine-5'-nucleotidase activity
Inhibition of erythrocyte pyrimidine-5'-nucleotidase leads to
accumulation of pyrimidine nucleotides, which has been associated with
induction of basophilic stippling.
Impaired function of erythrocyte pyrimidine-5'-nucleotidase
occurs in lead-exposed workers and as a genetically induced enzyme
deficiency. In in vitro systems the enzyme is inhibited by lead,
cadmium, mercury and zinc ions (and perhaps other metal ions) (Paglia
et al., 1975; Pagliuca et al., 1990; Ichiba & Tomokuni, 1990).
Graphs of PbB concentration against erythrocyte pyrimidine-
5'-nucleotidase activity in children have been reported for 21
children aged 2-5 years (Angle & McIntire, 1978) and for 42 aged 1-5
years (Angle et al., 1982) who participated in a preventative
programme because they had previously had PbB levels above
1.44 µmol/litre (30 µg/dl) or zinc protoporphyrin (ZPP) greater than
60 µg/dl. Both plots show considerable scatter although there was a
significant inverse logarithmic correlation. However, data relating to
the reproducibility, precision and accuracy of assessing inhibition of
erythrocyte pyrimidine-5'-nucleotidase were not presented.
This test may be capable of analytical refinement when assessment
of the sensitivity to lead (and other metals) exposure might more
reasonably be made. It does not yet appear likely to provide a routine
measure for low-lead exposure assessment.
8.2.3 Erythropoietin production
Graziano et al. (1991) found depressed serum erythropoeitin
levels in females at mid-pregnancy and at delivery associated
statistically with PbB level. Erythropoietin is a glycoprotein
produced in the renal proximal tubules which regulates both
steady-state and accelerated erythrocyte production. The study was not
adequate to define a dose-response relationship.
8.3 Nervous system
8.3.1 Historical perspective
Gross toxic effects of lead on the nervous system were reported
by ancient Greek physicians. A brief summary of such reports, and
those of Roman physicians and 18-19th century toxicologists, was
published by Kazantzis (1989). The syndrome was known as "painter's
colic", which included abdominal pain, constipation and paralysis,
symptoms now covered by the term "lead encephalopathy".
Lead colic was usually accompanied by effects on the nervous
system (lead palsy) in cases of acute and chronic poisoning, which
were often but not always fatal. Lead poisoning was well recognized as
an occupational hazard. However, it was also associated with
consumption of lead-contaminated water, wine, cider, rum from the West
Indies, and food prepared or stored in lead or lead-glazed utensils.
For example, Kazantzis (1989) described cases of lead poisoning in
girls resulting from consumption of drinking-water kept in lead-lined
cisterns, and also described the Devonshire colic attributed by Sir
George Baker in 1767 to the consumption of cider prepared in presses
made of lead or lead alloys.
The early toxicologists clearly recognized that the dose
(exposure) and form of lead was important in relation to absorption
and were able to demonstrate the presence of lead in tissues of fatal
cases of poisoning. There are many individually described, detailed
case studies in their writings.
8.3.2 Neurotoxic effects in adults
Despite the long recognition of lead poisoning, new clinical
cases continue to be the subject of published reports (Cueto et al.,
1989; Kocak et al., 1989; Zuckerman et al., 1989; Friedman &
Weinberger, 1990; Gupta et al., 1990; Mitchell-Heggs et al., 1990;
Nosal & Wilhelm, 1990; Sharma et al., 1990; Schneitzer et al., 1990;
Veerula & Noah, 1990). Although they are important, the numbers of
these poisoning cases are comparatively small compared with the
population at risk to the potential effects of exposure to lead from
environmental and dietary sources. Of particular recent concern have
been the possible neurotoxic effects on the developing child.
8.3.2.1 Central nervous system
In a study of 158 secondary lead smelter workers, Lilis et al.
(1977) found that 64% reported CNS symptoms and that there was early
occurrence of symptoms within 1 year of exposure. PbB concentrations
were elevated (about 3.36 µmol/litre or 70 µg/dl) at the time of
examination but many of the subjects had received chelation therapy.
Fischbein et al. (1979) noted CNS symptoms in 21 of 81 employees
of law enforcement agencies working in firing ranges. Symptoms
correlated with PbB levels and were present in three-quarters of
subjects having a PbB level > 2.4 µmol/litre (50 µg/dl).
Hanninen et al. (1979) reported that 49 lead-exposed workers
(including 21 male and 8 female storage battery and 16 railroad
engineering workshop employees) whose PbB level had never exceeded
3.36 µmol/litre (70 µg/dl) experienced more subjective symptoms than a
control group; excess symptoms were proportional to lead uptake.
Fischbein et al. (1980) studied 90 telephone cable splicers
having intermittent exposure to lead. They reported that 26 complained
of CNS symptoms (mean PbB level of 1.36 µmol/litre, 28.4 µg/dl, and
ZPP of 70.3 µg/dl) compared with controls (mean PbB level of
1.32 µmol/litre, 27.4 µg/dl, and ZPP of 49.3 µg/dl).
Awad El Karim et al. (1986) studied 92 lead acid battery workers
and compared them with 40 oil mill worker controls. They found CNS
symptoms in 50% of lead-exposed workers and measured PbB levels in 46
subjects working in different sections of the plant. More than 95% of
exposed workers had PbB levels above 1.92 µmol/litre (40 µg/dl),
whereas the mean PbB in the control group was 1.0 µmol/litre
(21 µg/dl).
Impairment of psychological and neuropsychological test
performance has been reported for lead-exposed workers (Hanninen et
al., 1979; Hogstedt et al., 1983; Parkinson et al., 1986; Araki et
al., 1986a,b; Huang et al., 1988a; Stollery et al., 1989, 1991).
The critical flicker fusion test has been reported to be a
sensitive indicator of CNS changes associated with exposure to lead.
Wooller & Melamed (1978) found significantly lower critical flicker
fusion in subjects with PbB levels above 2.88 µmol/litre (60 µg/dl)
than in those with levels below 0.96 µmol/litre (20 µg/dl). Williamson
& Teo (1986) found a significant decrease in mean flicker fusion in 59
lead-exposed workers (mean PbB > 2.3 µmol/litre or 48 µg/dl) compared
with matched controls. However, Gennart et al. (1992b) found no
difference between lead-exposed workers (mean PbB level
2.45 µmol/litre or 51 µg/dl, N = 98) and controls (mean PbB level
1.00 µmol/litre or 20.9 µg/dl, N = 85).
Stollery et al. (1989) studied the performance of 91 men,
occupationally exposed to inorganic lead, in a series of tests
designed to assess cognitive function. Subjects were grouped in "low"
(mean PbB 0.48 µmol/litre or 10 µg/dl, mean ZPP 8.7 µg/dl, N = 28),
"medium" (mean PbB 1.44 µmol/litre or 30.1 µg/dl, mean ZPP 22.4 µg/dl,
N = 27) and "high" (mean PbB 2.43 µmol/litre or 50.7 µg/dl, mean ZPP
58.8 µg/dl, N = 36) ranges of PbB concentrations. Results showed that
occupational exposure to lead impaired performance in a range of tests
(sensory motor reaction time, memory, attention, verbal reasoning,
spatial processing). Workers having PbB levels in excess of
1.92 µmol/litre (40 µg/dl) showed clear evidence of impairment on
tests of serial reaction time and category search. The authors
concluded that sensory motor, rather than cognitive, requirements of
many psychological tasks provide the most sensitive index of the early
effects of chronic low level lead exposure.
In a follow-up study (Stollery et al., 1991), 70 workers were
re-tested (three times within 8 months). Performance deficits in the
"high" (mean PbB 2.49 µmol/litre or 51.8 µg/dl, mean ZPP 77.4 µg/dl,
N = 22) group were not altered by practice or continued exposure. The
main deficit was a slowing of sensory motor reaction time coupled with
difficulties in remembering incidental information. There was little
evidence of impairment in workers having PbB levels less than
1.92 µmol/litre (40 µg/dl).
Finally, it appears that neuroelectrophysiological testing is a
sensitive and objective indicator of the CNS effects of lead. The
results of Araki et al. (1986b, 1987, 1992) using short-latency
somatosensory and visual evoked potentials and auditory event-related
potential (P300) indicate that subclinical electrophysiological
effects of lead occur not only in peripheral nerves but also in the
CNS.
8.3.2.2 Peripheral nervous system
Peripheral neuropathy is a common sign of chronic, high level
lead exposure, often manifesting as weakness in the upper or lower
limbs. At lower levels of lead exposure, nerve conduction velocity
(NCV) has provided a more sensitive indicator of peripheral nerve
dysfunction. More than 30 published studies have measured the
conduction velocity of electrically stimulated sensory and motor
nerves in workers exposed to lead. However, these studies have yielded
somewhat mixed results, with many showing a decrease in NCV in
relation to lead exposure (generally indexed as PbB concentration) and
a few showing no effect or occasionally even an increase in NCV
associated with lead exposure (Seppäläinen & Hernberg, 1980; Davis &
Svendsgaard, 1990).
Various reasons may underlie this lack of uniformity in NCV
findings. For example, studies may differ in methodological features,
in the characterization of lead exposure (e.g., single time-point
versus time-weighted average PbB levels) or in the handling of
confounding variables such as nerve temperature and age of subject
(Ehle, 1986). Other important factors accounting for some of the
apparent inconsistency in this area of research may be the possible
antagonistic effect of zinc to lead and differences in nerves selected
for measurement in different studies (e.g., slow versus fast fibres
(Araki et al., 1986c; Murata & Araki, 1991). A statistical
meta-analysis and critical review of 32 NCV studies by Davis &
Svendsgaard (1990) indicated that the median motor nerve shows effects
of lead more reliably than other nerves (e.g., median sensory or
ulnar).
Despite these complications, certain key well-conducted studies
provide compelling evidence of a causal relationship between lead
exposure and reduction in NCV. Araki et al. (1980) measured median
motor NCV before and after PbB levels of workers were lowered through
chelation therapy. Depending on the amount of reduction in PbB level
achieved by chelation and on a given worker's baseline NCV,
significantly improved NCV was measured in 7 out of 14 lead-exposed
workers. For all 14 workers the improvement in NCV was significantly
correlated with the decrease in PbB level (r = -0.573, P < 0.001).
In another key study, Seppäläinen et al. (1983) followed workers
prospectively from the beginning of their employment in a battery
plant, dividing them into two exposure categories (above and below the
median PbB level of 1.44 µmol/litre (30 µg/dl). Median motor NCV was
significantly reduced in the workers with blood lead levels above
1.44 µmol/litre (30 µg/dl) at 1-, 2-, and 4-year evaluation points,
despite attrition (and reduced statistical power) that resulted in
only five subjects per group after 4 years. Based on this well-
conducted prospective study, 1.44 µmol/litre (30 µg/dl) would appear
to be the lowest-observed-adverse-effect level for reduced NCV in
adults. Although another well-conducted prospective study (Spivey et
al., 1980) was unable to find an effect of lead on NCV at higher
exposure levels, NCV in the median nerve was not measured in that
study. Triebig et al. (1984) reported a dose-dependent decrease in NCV
at PbB levels above 3.36 µmol/litre, no dose-effect relationship being
detectable below this concentration.
8.3.2.3 Autonomic nervous system
There have been two reports of tests for autonomic nervous
function following lead exposure. Teruya et al. (1991) conducted a
cross-sectional survey of 172 male workers exposed to lead (mean PbB
level, 1.73 µmol/litre or 36 µg/dl, range 0.24-3.648 µmol/litre or
5-76 µg/dl), by measuring electrocardiographic R-R interval
variability (CVRR). Age-adjusted CVRR during deep breathing in
workers with PbB levels above 1.44 µmol/litre (> 30 µg/dl) was
significantly decreased as compared to those with PbB levels less than
0.96 µmol/litre (< 20 µg/dl). In addition, significant dose-response
and dose-effect relationships were observed between the PbB levels and
R-R interval variation during deep breathing among the workers with
PbB levels above 0.96 µmol/litre (> 20 µg/dl).
In a study reported by Murata & Araki (1991), the R-R interval
(CVRR) and two component coefficients of variation in the R-R
interval (C-CVRSA) were measured in 16 gun metal foundry workers
exposed to lead, zinc and copper (mean PbB level, 1.63 µmol/litre or
34 µg/dl, range 0.768-2.88 µmol/litre or 16-60 µg/dl). The CVRR and
component C-CVRSA (respiratory sinus arrhythmia) were significantly
lower in the workers than in age-matched controls, whereas the
C-CVMWSA (Mayer wave-related sinus arrhythmia) was unaffected. The
authors noted that zinc may have been antagonistic to the lead effects
in this study. Since the CVRR during deep breathing and the
component C-CVRSA of respiratory sinus arrhythmia reflect
parasympathetic activity, these two reports indicate potential
dysfunction of the autonomic nervous system (mainly, parasympathetic
activity) at average PbB levels of approximately 1.68 µmol/litre
(35 µg/dl).
8.3.3 Neurotoxic effects in children
The majority of the epidemiological research on the health
effects of lead has been focused on children because, in comparison
with adults, they are more vulnerable to lead in several respects
(Davis & Grant, 1992). For example, children typically engage in
hand-to-mouth activities (sucking fingers, putting food or other
objects in the mouth) that result in greater ingestion of lead than
adults normally experience. Also, because of their greater absorption
and retention of lead, the body burdens in children resulting from a
given external exposure level tend to be higher than adults.
Furthermore, the relatively greater exposures and body burdens of
children occur during sensitive periods of development. Finally, it
appears that children are generally more sensitive to the
toxicological effects of lead at a given internal exposure (PbB)
level. The lowest-observed-effect levels for various end-points (e.g.,
encephalopathy, anaemia, reduced haemoglobin, elevated EP, slowed NCV,
impaired neurobehavioural function) are lower in children than in
adults (US EPA, 1986a, 1990).
Section 8.3.3.2 describes findings from the main epidemiological
cross-sectional studies that have been published since 1979. This
constitutes a very substantial collection of research findings but has
certain interpretive limitations, particularly the lack of information
on exposure history.
More recent research effort has concentrated on prospective
epidemiological studies of birth cohorts repeatedly evaluated, mostly
up to the school age years. These prospective studies are described in
section 8.3.3.3.
Section 8.3.3.4 presents a quantitative overview of the
collective evidence from the prospective studies and discusses their
interpretation and the problems of inferring causality from such
observation data.
8.3.3.1 Historical perspective
While there have undoubtedly been cases of lead poisoning in
children as long as lead has been used by man, most of the attention
was previously given to cases of poisoning as a result of occupational
exposure in adults. In 1892 Gibson and colleagues in Australia
reported a case series of ten young children with lead colic. It was
not until twelve years later that peeling paint in the children's
homes was identified as the lead source (Gibson, 1904).
In 1943, Byers & Lord (1953) reported the results of an important
study, the first to suggest that there were neurological after-effects
of lead poisoning, in the absence of cerebral oedema and high
intracranial pressure, following acute lead encephalopathy. However,
in other cases it was felt that neurological manifestations
disappeared if the ingestion of lead was stopped (McKhann & Vogt,
1933). The case series of 20 school-age children had been hospitalized
for lead poisoning but without signs of acute lead encephalopathy, and
all were later discharged as cured. At follow-up, only one of the
children was making satisfactory progress at school. The children were
described as showing a variety of symptoms, including poor academic
achievement, intellectual deficits, sensory-motor deficits and
behaviour disturbance.
In the years following the findings of Byers & Lord (1953), a
number of case control studies were carried out that examined mental
retardation (Beattie et al., 1975) or hyperactivity (David et al.,
1972).
In addition, as a result of the growing concern about the dangers
of lead encephalopathy, studies were set up to investigate the
possibility that subclinical levels of lead (i.e. levels producing no
overt signs of lead encephalopathy) cause more subtle neurological
damage to children. Many of these studies investigated children
identified by screening clinics as having raised lead burdens, but
others were sited around smelters or lead works where children were
found to have elevated lead levels. The studies were important in
determining the areas of functioning which might be affected, and in
drawing attention to the methodological problems involved in carrying
out such studies. The main difference between the smelter studies or
studies around lead works, and clinical studies is that in the former
the primary source of environmental lead is known. In the hospital or
clinical studies it is likely that the ingestion of paint flakes, or
less often leaded putty, was in most cases the primary cause of the
excess lead exposure, although the exact reason for the increased lead
burden is rarely known.
Clinical and smelter studies were carried out in the United
Kingdom and USA and in general investigated children with PbB levels
above 1.92 µmol/litre (40 µg/dl), and compared their performance with
that of children with levels below this value.
Although some of the clinical and smelter studies found a
lead-associated deficit (Perino & Ernhart, 1974; Landrigan et al.,
1975), others did not (Lansdown et al., 1974). In general the studies
were methodologically flawed, with small samples lacking in
statistical power, biased ascertainment of subjects or controls, and
inadequate control for potentially confounding co-factors. The large
majority did not control for parental intelligence, which is the
variable most strongly associated with child IQ. These studies were,
however, important in suggesting an association between body lead
levels and performance, and also in demonstrating that there is an
association between social disadvantage and higher body lead levels.
8.3.4 Population-based cross-sectional studies on children
Most of the general population-based epidemiological
cross-sectional studies have, in addition to intelligence tests,
included extensive batteries of tests covering several other areas of
psychometric functioning, such as academic attainment, behaviour,
gross motor abilities and fine motor co-ordination, reaction time,
visuo-spatial skills and memory, and auditory memory. There are
differences in the tests used, and in the areas of functioning
investigated, and the results are less consistent and more difficult
to interpret than those relating to IQ tests. For that reason this
section will focus mainly on results relating to intelligence tests,
and results from other functional domains will be mentioned briefly
where appropriate.
There are many cross-sectional population studies using either
blood or teeth as the primary measure of lead body burden, and only
the more informative, in terms of providing information on risk
assessment, will be included. The majority of the studies described
used either a full or short form of the Wechsler Intelligence Scale
for Children - Revised (WISC-R or its translations) as the measure of
intelligence. Exceptions are the studies of Fulton et al. (1987),
which used the British Ability Scales (BAS), Harvey et al. (1988),
which used Wechsler Pre-school and Primary Scale of Intelligence
(WPPSI), and Schroeder et al. (1985) and Hawk et al. (1986), where the
Stanford-Binet Intelligence Scale was used.
8.3.4.1 Tooth lead studies
a) Needleman et al. (1979)
A total population of 3329 children aged 6-7 years, attending
schools in two working class towns near Boston, USA, were asked to
donate their shed deciduous teeth. Of these 70% (2335) children did
provide teeth. A 1-mm slice of tooth consisting primarily of dentine,
was split in half, and one portion was analysed by ASV. Children whose
initial tooth slice was in the highest 10th percentile (> 20 µg/g) or
the lowest 10th percentile (< 10 µg/g) were provisionally classified
into a high or low lead group. A total of 524 children were originally
classified into these lead groups, but 254 of these children were not
tested for a number of reasons, and the results for another 112 were
excluded from data analysis for medical reasons, or because a second
tooth analysis provided a discordant result. The results for 158
children (58 high lead children and 100 low lead children) were
presented. Thirty-nine non-lead variables were scaled and coded, and
those which were found to differ between the lead groups at the 10%
level were controlled as covariates. The covariates controlled were
father's socioeconomic status, mother's age at subject's birth, number
of pregnancies, mother's education and Peabody Picture Vocabulary
score. After this was done the children in the high lead group
performed significantly less well on the IQ test with a 4´ point
difference between the groups.
b) Winneke et al. (1983)
A total of 115 children aged 7-12 years living in the vicinity of
a lead/zinc smelter in Stolberg, Germany, who had provided at least
one incisor tooth, were investigated. Whole tooth lead measures ranged
from 2 to 32 µg/g, with a geometric mean of 6 µg/g. There was a
marginally significant negative association between lead level and
(short-form) verbal IQ after taking age, gender, perinatal risk
factors and social confounding factors into account. There was a
difference of 4.6 IQ points when children with tooth lead levels above
10 µg/g were compared with those with levels below 4 µg/g. Significant
negative associations were also observed between tooth lead and the
error score of Bender Gestalt shape copying test and performance on
the Vienna reaction time device.
c) Smith et al. (1983); Pocock et al. (1987)
The target population for the study by Smith et al. (1983) (also
known as the Institute of Child Health/University of Southampton
study) was 6875 children aged 6-7 years attending schools in one of
three areas in or near London. In all, 7407 teeth were donated by 4105
children. Whole tooth analysis of intact incisor teeth donated by
eligible children was carried out and children whose tooth lead level
(expressed on an ashed weight basis) was in the lowest percentile
(< 2.5 µg/g), approximately at the 50th percentile (5-5.5 µg/g) or
above the 90th percentile (> 8.0 µg/g) were invited to participate.
The sample was stratified by social grouping. Of the 432 children
selected, 403 participated in the study. Parental interviews were
carried out in the home, and information on a wide range of social and
familial variables was collected. There was a 5 point difference in IQ
scores between the high and low lead groups before controlling for any
covariates, but this was reduced to 2 points once covariates
associated with IQ score were controlled. The authors concluded that
social factors explain the differences in test performance to such a
degree that the small differences that remain may be due to other
social factors not measured. The study underlined the importance of
the social environment for the child's performance and recognized the
importance of the relationship between body lead burden and social
disadvantage.
Blood samples were taken from 93 children 3-6 months after
shedding a tooth and after psychometric testing was completed. The
range of PbB levels in this group was 0.336-1.296 µmol/litre
(7-27 µg/dl), except for one measurement of 2.064 µmol/litre
(43 µg/dl), and the mean was 0.614 µmol/litre (12.8 µg/dl). The
correlation of PbB and PbT was 0.5.
A reanalysis of this data by Pocock et al. (1989) employed
multiple regression techniques, using an "optimal" regression
strategy. The results were comparable to those previously reported.
Interactional analyses showed no evidence of any differential
association with lead in different social groups, but revealed a
significant interaction between PbT and child gender. Significant
negative association between PbT and IQ was found in boys but not in
girls.
d) Fergusson et al. (1988a,b,c)
This study was carried out in Christchurch, New Zealand, on a
sub-sample of a birth cohort of 1265 children born in 1977. Teeth were
collected from 1035 children, and a dentine "chip" was analysed to
provide a PbT estimate for 996 children. The mean dentine lead level
was 6 µg/g. Children from disadvantaged backgrounds had higher mean
lead levels than those from more advantaged backgrounds, and this
difference persisted when other social environmental factors (such as
the effects of housing, proximity to main roads, and childhood pica)
were taken into account. A WISC-R and reading test were administered
to children at ages 8 and 9 years, and results were given for 724
children at age 8 and 644 children at age 9. Small correlations
between IQ scores and dentine lead were found, but control for
confounding variables reduced these to statistical non-significance
for both ages (a correlation of -0.03 to -0.05). Parental IQ was
assessed. Control for pica as a test of reverse causality further
reduced the associations between outcomes and PbT.
e) Hansen et al. (1989); Lyngbye et al. (1990a,b); Grandjean et al.
(1991)
Children entering schools in the town of Aarhus, Denmark, were
asked to donate shed teeth. Of a potential sample of 2414 children,
1291 (54%) provided usable teeth. A sample of circumpulpal dentine was
obtained from each tooth and analysed by AAS. The mean lead level in
dentine was 10.7 µg/g, with a log normal distribution and a range from
0.40 to 168 µg/g. The correlation of whole tooth lead and circumpulpal
dentine measures were low. Average circumpulpal dentine levels were
about five times higher than average whole tooth measures. Subsequent
analysis of PbB from samples obtained two to three years later showed
an overall mean PbB level of 0.25 µmol/litre (5 µg/dl), with a
geometric mean of 0.28 µmol/litre (range 0.08-0.63 µmol per litre or
1.66-13.1 µg/dl) in children selected for the high lead group, and a
mean of 0.18 µmol/litre (range 0.08-0.70 µmol/litre or mean
3.74 µg/dl, range 1.66-14.56 µg/dl) in children selected for the low
lead group, which confirms this as a low exposure group. Children
whose level of lead in circumpulpar dentine was above 18.7 µg/g (8%,
N = 110) were selected for the high lead group, and they were matched
on gender and socioeconomic status (SES) with children whose
circumpulpar dentine levels were below 5 µg/g. A comprehensive list of
exclusions, including neurological and medical conditions, atypical
social situations, and those who did not wish to take part, reduced
the sample to 156 children for whom results were presented. Initial
analysis (by matched pair t-test) showed a difference of 6 IQ points
on full-scale WISC scores, with a difference of nearly 9 points in
verbal IQ scores. The association of IQ measures with circumpulpar
dentine lead levels was evident across different socioeconomic groups.
Confounders associated with circumpulpar dentine lead levels (such as
pregnancy variables, SES, mother's educational status and child's
gender) were entered into a stepwise regression model, and explained
only a small portion of the variance. Lead levels in the dentine
accounted for a significant (2.8%) of the total variance in full scale
IQ. Parental IQ was not controlled.
f) Rabinowitz et al. (1991)
From 764 eligible children (attending grades 1 to 3 in 7 selected
schools in Taiwan), 940 deciduous teeth were collected, the majority
being incisors. The average age of students was 6.7 years. Two of the
schools were near lead smelters. Lead levels were determined in 862
teeth from 692 children using the Boston method which analysed a
"dentine chip" (Rabinowitz et al., 1989). The mean value was 4.3 µg/g
of dentine. In all, 493 children were tested using the Ravens Coloured
Progressive Matrices Test (CPM) and the IQ equivalents were related to
tooth lead levels. Confounders were selected from a set of 40, based
on associations with lead and IQ. After correction for seven
confounding variables (social and perinatal factors), the highly
significant raw exposure-effect association (r = -0.32) was markedly
reduced for the full sample to statistical insignificance, although a
significant lead-CPM association remained for the subsample of girls
(b = -1.8; SE = 0.78; P < 0.02).
8.3.4.2 Blood lead studies
a) Winneke et al. (1985)
In a further study by this group carried out in the lead/zinc
smelter town of Nordenham, Germany, 114 out of 378 children (46%) for
whom cord PbB levels were available were tested at 6-7 years of age.
The range of cord PbB levels was 0.19-1.49 µmol/litre (4-31 µg/dl) and
for current PbB levels was 0.19-1.10 µmol/litre (4-23 µg/dl); the
means for both perinatal and current PbB levels were 0.39 µmol/litre
(8.2 µg/dl, SD = 1.6). After correction for confounders by means of a
stepwise multiple regression analysis, few significant associations
between PbB levels and performance were observed. There was a marked
influence of social background factors, but little influence on
short-form full IQ score (current PbB accounted for 0.3% additional
variance), and a borderline decrease in performance IQ (PbB accounting
for 2.4% of the variance). In general, the associations were rather
stronger with current PbB levels than with cord PbB measures. The
strongest associations were found with the error scores of the
difficult version of the Vienna reaction time task.
b) Harvey et al. (1984)
From 483 eligible children from the city of Birmingham, United
Kingdom, 187 out of 284 contacted were examined at 30 months of age.
The average PbB level from venous samples was 0.75 µmol/litre
(15.6 µg/dl, SD = 4). Cognitive ability was assessed by means of four
tests from the British Ability Scales (BAS) and three tests from the
Stanford-Binet Intelligence Scale. The raw correlation between PbB
level and IQ was -0.17 (P < 0.05). Subsequent multiple regression
analysis with a predetermined set of confounders and a sample size of
48 yielded an insignificant association between lead and IQ.
c) Harvey et al. (1988)
In a second study, 201 out of 337 eligible children, aged 5.5
years, from the inner city area of Birmingham, United Kingdom, were
studied. The mean PbB level was 0.614 µmol/litre (12.8 µg/dl, SD = 4).
No significant associations between PbB level and IQ (WPPSI) were
observed after correction for confounding factors. Apart from some
statistically significant associations between PbB level and reaction
time, none of the remaining tests showed a significant correlation
with PbB level after confounder-control.
d) Yule et al. (1981)
Children (N = 166), aged 6 to 12 years, living in the vicinity of
a leadworks in outer London were examined. PbB levels from venous
samples ranged from 0.336 to 1.58 µmol/litre (7 to 33 µg/dl, geometric
mean = 0.648 µmol/litre or 13.5 µg/dl). After controlling for social
class as the only confounder, significant negative associations were
found for IQ (WISC-R; full-scale- and verbal-, but not performance-
IQ). An average IQ difference of 7 points was found when comparing
children with PbB levels of 0.62 µmol/litre (13 µg/dl) or more with
those of 0.576 µmol/litre (12 µg/dl) or less. Significant inverse
associations were also found between PbB level and scores on tests of
attainment, i.e. reading and spelling but not mathematics.
A group of 9-year-old children from a middle class area of London
were studied. Subjects were divided into low PbB (N = 80 and PbB of
0.336-0.576 µmol/litre, 7-12 µg/dl) and high PbB (N = 82 and PbB of
0.624-1.15 µmol/litre, 13-24 µg/dl) groups for testing the association
between PbB level and IQ. No significant association between PbB level
and IQ was found before or after controlling for covariates (Lansdown
et al., 1986).
e) Schroeder et al. (1985)
One hundred and four children from a high risk population, aged
10 months to 6.5 years and with PbB levels ranging from 0.288 to
2.83 µmol/litre (6-59 µg/dl) were tested for intellectual development
using age-appropriate tests. After control for social confounding a
significant negative association was found between PbB level and IQ.
Fifty children of this cohort were reassessed 5 years later when all
PbB levels were below 1.44 µmol/litre (30 µg/dl). Neither initial nor
current PbB level was significantly related to later IQ.
f) Hawk et al. (1986)
In an effort to replicate the above-mentioned findings, 75
children from the same group, then aged 3 to 7 years, and having a
mean PbB level of 0.998 µmol/litre (20.8 µg/dl), range
0.29-2.256 µmol/litre (6-47 µg/dl), were examined using the
Stanford-Binet test. There were no significant interactions between
PbB level and factors including age, sex, maternal IQ, quality of the
care-giving environment (HOME), or socioeconomic status. There was a
statistically significant negative association between PbB level and
IQ, although the characteristics of the regression coefficient for
lead depended on which covariates were included in the model. For the
final regression model that best controlled for confounding with the
greatest precision, i.e. the most accurate and precise model,
containing lead, maternal IQ, HOME and gender, highly significant
inverse associations were found between both mean and maximum PbB
levels and IQ.
g) Fulton et al. (1987)
Five hundred and one children, aged 6-9 years, out of 1210
eligible children from 18 primary schools in central Edinburgh,
Scotland, were tested for an association between PbB level, IQ and
attainment. The sample comprised all children in the top quartile of
the PbB distribution, and a random subsample (approximately 1 in 3) of
the remainder. The geometric mean PbB level was 0.55 µmol/litre, range
0.16-1.63 µmol/litre (11.5 µg/dl, range 3.3-34.0 µg/dl). After
correction for 31 potential confounders a significant negative
association between PbB level and IQ (BAS) was found. There was a
dose-response relation with no evidence of a threshold. For purposes
of statistical analyses, children were placed into 10 groups of about
50 each on the basis of PbB level. The mean PbB level was
0.27 µmol/litre (5.6 µg/dl) in the lowest and 1.06 µmol/litre
(22.1 µg/dl) in the highest group; there was a difference of 5.8 IQ
points between the two groups. The size of the effects of lead was
small relative to that of the other factors (0.9% of a total of 45.5%
explained by the full model).
h) Hatzakis et al. (1989)
Five hundred and thirty-three children out of 1038 eligible from
four primary schools in the vicinity of the old lead-mining and
lead-smelting industrial complex of Lavrion in Greece were examined
neuropsychologically. The mean PbB level from venous samples was
1.14 µmol/litre, range 0.355-3.067 µmol/litre (23.7 µg/dl, range
7.4-63.9 µg/dl). In order to control for confounding factors, several
regression models were evaluated. Starting with a set of 24 potential
confounders, an optimal model containing 17 potentially confounding
variables was finally developed for testing the PbB/IQ association.
The continuous PbB variable was divided into five equally wide classes
from equal/below 0.715 µmol/litre (14.9 µg/dl, low) to equal/above
2.16 µmol/litre (45.0 µg/dl). After correction for confounders, a
highly significant negative association was found between PbB level
and IQ (WISC-R). A persistent decrease in IQ was only observed at PbB
concentrations above 1.2 µmol/litre (25 µg/dl). The adjusted
full-scale IQ difference between "high" and "low" PbB children was 9.1
points. Highly significant negative associations with PbB were also
found for the Bender Gestalt test and the Vienna Reaction Device,
without any indication of a threshold.
i) Silva et al. (1988)
Five hundred and seventy-nine socioeconomically advantaged
11-year-old children with an average PbB level of 0.53 µmol/litre
(11 µg/dl), range 0.19-2.4 µmol/litre (4-50 µg/dl) were tested in
Dunedin, New Zealand. Because the crude inverse association between
blood lead and IQ was not statistically significant, subsequent
multivariate analyses were not carried out.
j) European multicentre study
The results from a multicentre study in Europe were reported by
Winneke et al. (1990). The study linked eight institutions from eight
European countries; four of the individual study groups were from
areas near smelters and the others were more general populations. A
common study protocol with inherent quality assurance elements was
developed to achieve comparability between the individual studies. In
all, 1879 children, aged 6-11 years, were studied; however, for some
of the tests the sample size was reduced to only 971 children. The PbB
level ranged from 0.24 to 2.88 µmol/litre (5 to 60 µg/dl). Overall
statistical evaluation was done using a uniform predetermined
confounder model containing age, gender, occupational status of the
father, and mother's education. The inverse association between PbB
level and IQ (four subtests from the WISC-R) was of only borderline
statistical significance. An IQ decrement of about 3 points was
calculated for a PbB increase from 0.24 to 0.96 µmol/litre (5 to
20 µg/dl). The associations between the error scores on the Bender
Gestalt Test and the Vienna Reaction Device were statistically
significant and more consistent across study groups, although the
outcome-variance explained that lead never exceeded 0.8%. The data did
not allow for the identification of a threshold.
8.3.4.3 Follow-up studies
a) Bellinger et al. (1984)
A follow-up of some of the children investigated by Needleman et
al. (1979) was conducted when the children were approximately eleven
years old. Twenty-two of the original elevated lead group and 48
children from the low lead group were traced, as well as a group of 52
children who had not been tested previously, but whose lead levels in
a "dentine chip" (from teeth analysed at the time of the previous
study) fell into the mid-range between the high and low lead groups.
Children in the elevated lead group were significantly younger than
those in the other two groups. Information on IQ from group-
administered Otis Lennon Mental Ability tests, conducted 1 to 2 years
previously, was collected from school records for 15, 52 and 34
children from the elevated, mid-range and low groups, respectively.
Scores were inversely related to previously obtained lead measures,
with a 7-point difference in mean scores between the high and low lead
groups (t-ratio = 1.65, P = 0.1). No current measures of lead status
were available.
b) Winneke et al. (1989)
Out of 114 children first tested at age 6 (Winneke et al., 1985)
76 were retested at age 9. The range of PbB levels had been
0.187-1.09 µmol/litre, geometric mean = 0.39 (3.9-22.8 µg/dl,
geometric mean = 8.2) at age six and was 0.21-1.027 µmol/litre,
geometric mean 0.37 (4.4-21.4 µg/dl, geometric mean 7.8) at age 9
(r = 0.82). After correction for the same set of confounders used at
age 6 (age, gender, social background), most of the findings that had
been found to be significant at age 6, i.e. those related to reaction
performance (Vienna Reaction Device), remained virtually unchanged
three years later. The authors concluded that this observation cannot
necessarily be taken to indicate persistence, because internal
exposure had essentially remained unchanged over three years.
c) Needleman et al. (1990)
A follow-up assessment was made of 132 young adults first
investigated by Needleman et al. (1979) at the age of 6-7. Those
retested were not representative of the group of 270 tested 11 years
earlier, as they had slightly lower childhood dentine lead levels,
came from higher SES families, and were more likely to be female.
Current PbB level was assessed from 48 subjects and none was above
0.336 µmol/litre (7 µg/dl). A behavioural evaluation including a word
reading test, and a neurobehavioural assessment was administered to
each subject, and a self-report of delinquency was obtained. In
addition school records were reviewed. Multiple regressions indicated
that higher levels of dentine lead in childhood was associated with
lower reading scores, lower class rank, increased absenteeism, and
poorer performance on some of the neurological tests in young
adulthood. Comparing subjects with dentine lead levels greater than
20 µg/g with the lowest lead group, children in the high lead group
were much more likely to drop out of school (unadjusted odds ratio
4.6; adjusted odds ratio 7.4, CI 1.4-40.8) and to have a reading
disability (unadjusted odds ratio 3.9; adjusted odds ratio 5.8, CI
1.7-19.7). Adjustment for covariates made little impact on most of the
outcomes.
8.3.4.4 Conclusions and limitations of cross-sectional studies
A number of general issues are evident from looking at the
cross-sectional studies as a group. In most studies a negative
association between lead measures and IQ measures is found in
uncontrolled data. This difference is usually in the range of 4 to 6
IQ points and most marked in verbal IQ. Most studies also confirmed
the positive association between lead measures and indicators of
social disadvantage, whether this was indicated by SES, maternal
education, or other more detailed indicators of non-optimal
child-rearing environments, such as marital quality or maternal
depression. When these social and other confounding factors are
controlled, the effect has, in most cases, been to reduce the strength
of the association between lead measures and IQ, although it remains
in the same direction. When maternal intelligence has not been
controlled, the impact on the association between lead and IQ measures
of correction for covariates tends to be smaller. Where more detailed
social measures have been controlled, the impact on the magnitude of
the lead-IQ association has been greater. Associations have, in
general, been stronger with verbal IQ than with performance IQ.
The cross-sectional nature of these studies, and also the fact
that many used a single measure of current exposure, limited their
usefulness in answering questions relating to the natural history of
the association between lead exposure and outcomes, including whether
there were critical periods of exposure, and whether lead associated
deficits were persistent or reversible.
Findings from follow-up studies are difficult to interpret.
Needleman et al. (1990) interpreted their findings as indicating the
persistence of the effects of lead. Identifying persistence or
irreversibility in this context has many problems, due to the
stability of a number of other (non-lead) factors. In addition,
sampling biases, or failure to control for important covariates in the
initial or follow-up stages may lead to apparent persistence of an
effect. A further difficulty is that it is unusual to find a cohort
where exposure profiles permit easy assessment of persistence.
Neurobehavioural effects detected at age seven or later usually
persist in later follow-up studies conducted in other (non-lead) areas
of research, and for this reason it is more likely than not that
lead-associated deficits detected during childhood will be detectable
later. However, there are not enough data to conclude that this is the
case. Animal data, in which persistence of effects has been shown
after cessation of exposure, do provide support for the
irreversibility of lead-induced behavioural toxicity.
8.3.5 Prospective epidemiological studies on children
8.3.5.1 Common elements
The international prospective cohort studies shared a common
design of pre- or perinatal recruitment, with the principal measure of
prenatal exposure being the lead concentration in whole blood taken
antenatally or at birth. To facilitate later comparison of results,
meetings have been held at which common protocols (within the
constraints of local conditions) were agreed (Bornschein & Rabinowitz,
1985; Smith, 1989).
The protocols were similar in terms of the instruments of
neurobehavioural assessments, which were well standardized and
validated for the populations to which they were applied, and although
the frequency of assessment varied between studies, it was agreed that
all studies would make an assessment during infancy, in the later
pre-school period and, if possible, in the school age years.
All studies conducted several assessments of PbB concentration
but at varying frequency, and all studies participated in some form of
quality assurance/quality control (QA/QC) programme for the assessment
of lead exposure. QA/QC procedures were also used to establish
inter-examiner reliability (where more than one examiner was used) for
psychometric and covariate assessments. The importance of such
procedures was recently discussed (SAHC, 1993).
In assessing dose-effect relationships between lead exposure and
outcome variables, most studies used several indices of exposure,
including both measures of PbB concentration at particular ages and
average measures of exposure within the lifetimes of the study
subjects.
All studies used data analysis procedures which took simultaneous
account of covariates selected for statistical and substantive
considerations.
8.3.5.2 Study descriptions
a) Boston study
The study population was recruited from 11 837 infants born at
the Brigham and Women's Lying-In hospital in Boston between April 1979
and April 1981 (Bellinger et al., 1984). On the basis of cord PbB
levels of approximately 2500 children, the 90th, 50th and 10th
percentiles were identified and used as eligibility criteria for
enroling 249 children in three exposure groups: low (< 0.14 µmol/litre
or 3 µg/dl, N = 85), medium (0.288-0.336 µmol/litre or 6-7 µg/dl,
N = 88) and high (> 0.48 µmol/litre or 10 µg/dl, N = 76). No child
had a cord PbB level greater than 1.2 µmol/litre (25 µg/dl).
Exclusion criteria included birth complications or medical
conditions associated with developmental difficulties (e.g.,
gestational age < 34 weeks, Down's syndrome), a non-English speaking
family or residence in an area considered unsafe for home visitors.
The cohort consisted largely of children from intact middle and
upper-middle class families. Approximately 85% of the children were
white.
Capillary blood collected at ages 6, 12, 18 and 24 months was
analysed for lead by ASV. Venous blood collected at ages 57 months and
at 10 years was analysed for lead by AAS. In contrast to other
prospective cohorts, the mean PbB level did not rise postnatally and
indeed never exceeded 0.384 µmol/litre (8 µg/dl).
The principal psychometric tests included the Bayley Scale of
Infant Development at 6, 12, 18 and 24 months, the McCarthy Scales of
Children's Abilities at 57 months and the Wechsler Intelligence Scale
for Children - Revised and the Kaufman Test of Educational Achievement
at age 10 years. After age 2 years, the mean level of performance was
above the expected population mean (e.g., mean full scale IQ at age 10
years = 119) (Bellinger et al., 1992).
A variety of potential confounding factors was assessed including
maternal IQ, the quality of rearing environment (HOME scores at 6, 24,
57 and 120 months) and prenatal, perinatal and postnatal medical and
socio-demographic factors.
b) Cincinnati study
Women attending prenatal clinics in predesignated leadhazardous
residential areas of Cincinnati, Ohio, USA, were consecutively
recruited from 1979 to 1984 (Dietrich et al., 1987) Prenatal
exclusions were for maternal prenatal alcohol and drug abuse,
psychosis, diabetes, mental retardation; neonatal exclusions included
birth weight under 1.5 kg and gestational age (by physical
examination) below 35 weeks. Furthermore, recruited infants had to
have an Apgar score of 6 or more at 5 min postpartum and to have no
serious medical conditions. Of the mothers, 87% were single and 86%
were receiving public assistance. The final follow-up sample of 305
infants who attended their second clinic visit at 3 months of age was
85% African-American and 50% female.
Mean prenatal maternal PbB levels as assessed by ASV were
0.38 ± 0.177 µmol/litre (8.0 ± 3.7 µg/dl). Mean neonatal PbB levels
were also low at 0.221 ± 0.134 µmol/litre (4.6 ± 2.8 µg/dl). Following
the development of prewalking progression and normal hand-to-mouth
behaviours, PbB levels began to rise, peaking at around 21 months with
a mean of approximately 0.89 µmol/litre (18 ± 6 µg/dl). Approximately
35% of the sample had at least one PbB concentration equal to or
greater than 1.2 µmol/litre (25 µg/dl) sometime during the first 5
years of life, while 79% exceeded 0.72 µmol/litre (15 µg/dl) during
the same period (Dietrich et al., 1991, 1992). Virtually all children
(95%) exceeded 0.48 µmol/litre (10 µg/dl) during the first 5 years of
life. Postnatal PbB concentrations were assessed on a quarterly basis
beginning at 10 days postpartum. The vast majority of blood samples
were collected by venepuncture.
Major neurobehavioural assessments included the Bayley Scales of
Infant Development administered at 3, 6, 12 and 24 months of age. The
Kaufman Assessment Battery for Children was administered at 4 and 5
years. A comprehensive examination of gross and fine neuromotor
functions (the Bruininks-Oseretsky Scales) was administered at 6
years. Finally, performance on the Wechsler Intelligence Scale for
Children - Revised was assessed following school entry at 6.5 years.
Intellectually, this was a low functioning cohort with a mean
full-scale IQ at 6.5 years of 86.9 ± 11.3 points (Dietrich et al.,
1993a).
Covariates measured included assessments of obstetrical and
perinatal complications, birth anthropometrics, tobacco and alcohol
consumption, observational assessments of the quality of care-taking
in the home at 6, 12, 24 and 36 months, social class, iron status and
various aspects of child health likely to affect neurobehavioural
performance (e.g., otitis media, sensory deficits, allergies, seizure
disorders). Maternal IQ was assessed postnatally. All medical and
psychometric testing was conducted at a single welfare clinic located
in the heart of the recruitment area.
c) Cleveland
Five hundred and forty-three infants delivered to women at a
large inner city general hospital between February 1981 and March 1982
were considered for inclusion in this study of the joint or
independent effects of fetal alcohol exposure and lead on child
development (Ernhart et al., 1985). Due to pre-term birth or illness,
16% were excluded from further study. Also excluded were mothers
reporting the use of narcotics and those with identifiable
psychological disorders. The sample was predominantly white (65%) and
of lower SES. Approximately 50% of mothers were alcoholics.
PbB concentrations were assessed by AAS (208 mothers at delivery
and in 178 cords resulting in 142 mother-infant pairs with complete
prenatal lead exposure data). Postnatally, blood samples were analysed
for approximately half the birth cohort at ages 6 months, two years
and three years of age. At least one perinatal or postnatal PbB
concentration was available for each of 285 children with
developmental data past the neonatal period (Ernhart et al., 1987,
1989a,b).
Mean maternal PbB concentrations (N = 185) were low
(0.31 ± 0.086 µmol/litre or 6.5 ± 1.8 µg/dl) and mean cord PbB
concentrations (N = 162) were also low (0.29 ± 0.10 µmol/litre or
6.0 ± 2.1 µg/dl). Postnatally, mean PbB concentrations (N = 151) were
0.48 ± 0.16 µmol/litre (10.0 ± 3.3 µg/dl) at 6 months,
0.80 ± 0.28 µmol/litre (16.7 ± 5.9 µg/dl) at 2 years (N = 165) and
0.80 ± 5.9 µmol/litre (16.7 ± 5.9 µg/dl) at 3 years. The lowest and
highest PbB levels recorded postnatally were 0.24 and 2.016 µmol/litre
(5 and 42 µg/dl), respectively.
Neurobehavioural assessment included selected sub-scales of the
Brazelton Neonatal Behavioural Assessment Scales and Graham Rosenblith
Behavioural Examination of the Neonate at > 24 h postpartum, the Kent
Infant Development scale at 6 months, the Bayley Scales of Infant
Development at 6, 12 and 24 months, the Stanford-Binet Intelligence
Scale at 3 years, and finally the Wechsler Preschool and Primary Scale
of Intelligence at approximately 5 years of age. All assessments
beyond the neonatal period were conducted in subjects' homes.
Intellectual attainment in this high risk, socioeconomically
disadvantaged cohort was typically low as shown by a mean full-scale
IQ at approximately 5 years of 87.5 ± 16.6 points (Ernhart et al.,
1989a).
In addition to the usual assessments of obstetrical/perinatal
complications and neonatal status, other covariates measured included
the Michigan Alcoholism Screening Test, the Peabody Picture Vocabulary
Test as an assessment of maternal intelligence, and the Authoritarian
Family Ideology Scale. Women were also questioned as to their
consumption of alcohol and tobacco during pregnancy. Observational
assessments of care-taking quality in the home were conducted at 1, 2,
3 and 4 years.
d) Glasgow study
The study group for CNS outcomes consisted of 151 subjects drawn
from an initial sample of 885 families exposed to various levels of
dietary lead, principally due to a plumbosolvent water supply (Moore
et al., 1989). All subjects were born in the United Kingdom and spoke
English as a first language. The sample was divided into three groups
of approximately equal numbers (matched for social class) based on
maternal prenatal PbB levels: high (> 1.44 µmol/litre or 30 µg/dl),
medium (0.72-1.2 µmol/litre or 15-25 µg/dl) and low
(< 0.48 µmol/litre or 10 µg/dl). The SES of the sample ranged from
the chronically unemployed to the professional classes.
PbB levels were assessed postnatally at 1 and 2 years of age and
found to be 0.734 and 0.777 µmol/litre (15.3 and 16.2 µg/dl),
respectively.
Measurements of neurobehavioural outcome where the Bayley Scales
of Infant Development were used were administered at 1 and 2 years of
age.
Covariates considered in the data analysis included a measure of
obstetrical complications, birth weight, birth order, SES of the
father in postnatal follow-up years 1 and 2, and observational
assessments of care-taking quality in the first and second years of
follow-up.
e) Kosovo Study
Five groups of infants from two communities in Kosovo,
Yugoslavia, were studied (Graziano et al., 1990; Wasserman et al.,
1992). Mitrovica was the site of a lead smelter, refinery and battery
plant, and Pristina an area of minimal lead exposure. Three groups
were recruited from Mitrovica on the basis of cord PbB concentration:
(1) < 0.72 µmol/litre (< 15 µg/dl, N = 78); (2) 0.72-0.96 µmol/litre
(15-20 µg/dl, N = 99); (3) > 0.96 µmol per litre (> 20 µg/dl,
N = 217). Two groups were recruited from Pristina: (4) a group with
cord PbB levels < 0.72 µmol/litre (< 15 µg/dl) and (5) a group
matched to children in group (3) in terms of the distribution of
maternal and paternal education. Grouping was not considered in the
statistical analysis. The total cohort (N = 541) was largely Albanian
and Serbian. Infants with CNS defects, chromosomal anomalies, distant
residence and twin pairs were excluded.
PbB level and various indices of iron status (EP, Hb, serum
ferritin) were measured in venous blood collected in midpregnancy,
delivery and at postnatal ages 6, 12, 18 and 24 months. Mean PbB level
amongst children in Mitrovica exceeded 0.96 µmol/litre (20 µg/dl) at
all ages, reaching 1.68 µmol/litre (35 µg/dl) at 24 months. Amongst
children in Pristina, the mean PbB level was below 0.48 µmol/litre
(10 µg/dl) at all ages.
The Mental Scale of the Bayley Scales of Infant Development was
administered at ages 6, 12, 18 and 24 months, and the Motor Scale at 6
and 12 months. At 24 months, the mean Mental Development Index in the
cohort (n = 392) was 105.2 (SD = 18.1).
Anthropometric measurements and assessments of child and family
health, diet, and demographic status were carried out at each age. An
adaptation of the HOME Scale was administered at ages 3 and 4 years
and maternal IQ was assessed using the Raven's Standard Progressive
Matrices.
f) Port Pirie study
Port Pirie (population 16 000 in 1979) is an industrial town 200
km north-west of Adelaide, South Australia, with a large and
long-standing smelting facility (Wigg et al., 1988). Over the period
1979-1982, a cohort of 723 children from Port Pirie and surrounding
rural areas was recruited into the study. The cohort represented a 90%
sample of all children born in the area during this period.
Lifetime lead exposures were estimated from venous PbB
concentrations obtained antenatally (at 14-20 weeks of gestation, and
32 weeks), at delivery (maternal and cord blood), and from capillary
samples taken postnatally at ages 6, 15, and 24 months, and annually
thereafter. Lead concentrations in all samples were determined by AAS.
At the time that each blood sample was collected, the nurse
interviewer also conducted structured interviews to obtain information
on a range of demographic, familial, behavioural, medical and social
environmental factors. HOME scores were assessed when the child was 3
and 5 years of age. Maternal IQ was also assessed. The Bayley Scales
of Infant Development was administered at 2 years, the McCarthy Scales
of Children's Abilities at 4 years and the Wechsler Intelligence Scale
for Children - Revised version (WISC-R) at 7 years.
g) Sydney study
A total of 318 children was recruited from among infants born at
three maternal hospitals between April 1982 and March 1983 (Cooney et
al., 1989a). Exclusion criteria included prematurity, low birth
weight, severe medical problem, single or non-English speaking mother
or maternal drug or alcohol problems. All children were white and
generally represent a middle-class population.
PbB concentration was measured in maternal venous blood during
hospitalisation (geometric mean = 0.388 µmol/litre or 8.1 µg/dl,
geometric SD = 0.067 µmol/litre or 1.4 µg/dl). Additional blood
samples were collected at 6-month intervals to age 4 years and at ages
5 and 7 years. Up to 24 months of age, approximately 50% of the blood
samples were capillary, while almost all samples collected
subsequently were venous. Geometric mean PbB levels between 6 months
and 4 years were in the range of 0.48-0.72 µmol/litre (10-15 µg/dl),
falling to 0.39 and 0.37 µmol/litre (8.3 and 7.7 µg/dl) at ages 5 and
7, respectively.
The Bayley Scales of Infant Development were administered at ages
6, 12 and 24 months, the McCarthy Scales of Children's Abilities at
ages 3 (n = 215) and 5 (n = 200), and the Wechsler Intelligence Scale
for Children - Revised at 7 years (n = 175). All assessments, at least
until age 5, were conducted in the child's home. The mean Mental
Development Index and General Cognitive Index (GCI) scores were in the
intellectual normal range of 107 to 117 with a mean general cognitive
index at 4 years of 107.3 ± 14.2.
Covariates measured included maternal IQ, quality of care-taking
environment, obstetrical and postnatal factors.
8.3.5.3 Summary of differences between studies
The prospective studies differed to such an extent that it would
be surprising if they yielded identical findings. This does not mean
that the studies cannot be compared, merely that these differences and
their potential impact on study findings must be acknowledged in any
assessment of the studies as a group (Bellinger & Stiles, 1993). There
are substantial differences in the degree of confounding between lead
exposure and the other correlates of poor developmental outcomes. For
example, mean maternal IQ ranges from 121 (Boston) to 74-75
(Cleveland, Cincinnati), reflecting vast cohort differences in
socioeconomic standing and child-rearing context.
The studies also varied substantively in terms of the lead
exposure profiles observed. Site differences resulted in considerable
variability in postnatal lead exposures depending on exposure patterns
(residence near primary lead smelters, residence in deteriorated
housing, proximity to traffic, and quality of domestic
drinking-water).
Despite broad similarities in the approaches to data analysis,
there were substantial differences in the form in which the findings
were reported, impeding direct comparison of study results. Results
were diversely reported in terms of partial correlation coefficients
and standardized and unstandardized regression coefficients, and there
was variable use of raw (untransformed) and log exposure measures.
Most, but not all studies presented their results in a manner
conducive to some kind of quantification of dose-effects
relationships.
Clearly the variation in numbers of participants available to
individual studies (both initially, and as a consequence of attrition)
has a direct effect on the statistical power of each study - which is
also affected by the degree of confounding.
8.3.5.4 Results of studies
a) Infancy and early preschool assessments
The pattern of findings in infant and early preschool assessments
for which the principal instrument of assessment was the Bayley Scales
of Infant Development is inconsistent. Using different indices of
in utero exposure (e.g., prenatal, at delivery, cord or neonatal PbB
levels), three of the prospective studies found associations with
slower sensorimotor development up to 6 months or 1 year of age
(Ernhart et al., 1987; Dietrich et al., 1987, 1989) or up to 2 years
of age (Bellinger et al., 1987), but these tended to attenuate over
time (Dietrich et al., 1990; Bellinger et al., 1991a). Other studies
did not observe statistically significant adjusted relationships of
prenatal exposure with slower sensorimotor development (Moore et al.,
1989; Wigg et al., 1988; Cooney et al., 1989a,b; Wasserman et al.,
1992).
Two studies observed an inverse association between postnatal
lead exposure on mental development in later infancy (Wigg et al.,
1988; Wasserman et al., 1992), although in one study it was PbB level
at 6 months of age (Wigg et al., 1988) and in the other study it was
the level at 24 months (Wasserman et al., 1992) that was most
predictive.
b) Later preschool and school age assessment
Despite some inconsistency in the pattern of findings in infants,
there has been a convergence of positive findings on later
neurobehavioural outcomes in the prospective studies. This may
reflect:
1) the greater reliability and precision of measurement attained
with assessments of the older child; or
2) an effect of lead on abilities that cannot easily be tested
during infancy (e.g., executive, regulative and organizational
skills, higher order reasoning).
c) Boston study
Among 169 children assessed at age 57 months, the major finding
was an association between PbB level at 2 years of age and the GCI of
the McCarthy Scales. There was a decrease of 2.95 ± 1.42 GCI units for
an increase in PbB of 0.48 µmol/litre (10 µg/dl). Since the group had
an average PbB level of 0.336 µmol/litre (7.0 µg/dl) over 2 years,
this decrement was relevant to a range of approximately
0.19-0.67 µmol/litre (4-14 µg/dl) Bellinger et al., 1991a).
At 10 years of age, 148 children were reassessed. PbB
concentration at 2 years of age was inversely associated with the
WISC-R Full Scale IQ. Each 0.48 µmol/litre (10 µg/dl) increase was
associated with a 5.8 point decline in IQ (95% CI 1.7 to 9.9)
(Bellinger et al., 1992).
PbB concentration at 24 months was also inversely related to the
Battery composite score in the Kaufman Test of Educational Achievement
(Brief form) (-0.89 ± 0.24). The skills assessed were mathematics,
reading and spelling.
The point estimates associated with other postnatal ages were
generally in the same direction but only PbB level at 2 years revealed
conventional statistical significance. Other specific
neuropsychological tests did not reveal a clear pattern of
neuropsychological deficit (Stiles & Bellinger, 1993).
d) Cincinnati study
The Kaufman Assessment Battery for Children was administered to
approximately 260 children at 4 and 5 years of age (Dietrich et al.,
1991, 1992). The principal findings at 4 years were that higher
neonatal PbB levels were associated with poorer performance on all
Kaufman subscales. However, this inverse association was limited to
children from the poorer families. Following full covariate
adjustment, few statistically significant associations between
postnatal PbB levels and Kaufman scales could be found. However, the
results did suggest a weak relationship between postnatal PbB levels
and performance on a Kaufman subscale which assesses visual spatial
and visual-motor integration (adjusted regression coefficient -0.12 SE
not published).
At 5 years of age, postnatal PbB levels were associated with
poorer performance an all subscales of the Kaufman Battery. However,
after adjustment for covariates, few statistically significant
relations remained. Nevertheless, as at 4 years of age, the subscale
assessing visual-spatial and visual-motor skills was most sensitive,
with average lead exposure during the 4th year of life being
significantly associated with performance (-0.12 units per
0.048 µmol/litre or per µg/dl, SE not published).
At the age of 6.5 years, 253 children in the Cincinnati cohort
were administered the WISC-R (Dietrich et al., 1993a). The major
findings were that postnatal PbB concentrations were inversely
associated with full-scale IQ and performance IQ. Following
statistical adjustment for covariates, including maternal IQ and
quality of home care, a statistically significant relationship was
retained between postnatal PbB concentrations at nearly all ages (and
including lifetime averages) and Performance IQ. Further analysis
revealed that average lifetime PbB concentrations in excess of
0.96 µmol/litre (20 µg/dl) were associated with deficits in IQ of the
order of 7 points, when compared with children with mean
concentrations of less than 0.48 mol/litre (10 µg/dl). The regression
coefficient for Performance IQ on mean lifetime PbB level was -0.26
(± 0.12) IQ units per 0.048 µmol/litre or per µg/dl. This corresponded
to a 2.6 unit decrement associated with a 0.48 µmol/litre (10 µg/dl)
movement in lifetime average PbB level, for which the average
(interpolated graphically) is close to 0.67 µmol/litre (14 µg/dl).
At 6 years of age, the Bruininks-Oseretsky Test of Motor
Proficiency (BOTMP) was administered to 245 children in the Cincinnati
Cohort (Dietrich et al., 1993a). Following statistical adjustment for
covariates, neonatal PbB levels were associated with poorer
performance on a measure of upper-limb speed and dexterity and the
fine-motor composite. Postnatal PbB levels also remained significantly
associated with poorer scores on measures of bilateral coordination,
visual-motor control, upper-limb speed and dexterity, and the fine
motor composite.
Children having an average mean lifetime PbB level of
approximately equal to or exceeding 0.43 µmol/litre (9 µg/dl) appeared
to experience a deficit in both fine and gross motor skills relative
to children in the lowest PbB quartile. Children in the highest
average lifetime PbB quartile had scores on the gross-motor subtest
assessing bilateral coordination of approximately 0.5 standard
deviations (2.5 points) lower than their counterparts in the lowest
quartile. Children in the highest average lifetime quartile also
scored more poorly in the fine-motor functioning, having scores of
approximately 0.6 standard deviations lower (6.3 points) than those in
the lowest quartile.
e) Cleveland study
The WPPSI was administered to 242 children at the age of 4 years
and 10 months (Ernhart et al., 1989a). Statistically significant
correlations between IQ (full scale and subscales) and PbB level
measured pre- and perinatally and at ages 2 and 3 years (and a
lifetime postnatal average) became non-significant after adjustment
for covariates. No estimates for effect size were presented in the
original report (Ernhart et al., 1989a).
f) Glasgow study
No neurobehavioural assessments were carried out beyond 2 years
of age (Moore et al., 1989).
g) Kosovo study
No results beyond 2 years of age were reported (Graziano et al.,
1990; Wasserman et al., 1992).
h) Port Pirie cohort study
The principal finding at 4 years was an inverse association of
McCarthy Scales of Children Abilities (MSCA) scores (General Cognitive
Index (GCI), Perceptual Performance and Memory) with most indices of
lead exposure (McMichael et al., 1988). After adjustment for
covariates, the association of GCI was not signifi-cant at certain
specific ages. However, it remained significant for the integrated
postnatal average for which an effect size of 7.2 GCI points lost in
association with an increase of lifetime average PbB level from 0.5 to
1.5 µmol/litre (10.4-31.2 µg/dl) was estimated.
Between 7 and 8 years of age the IQ of 494 children was assessed
with the WISC-R (Baghurst et al., 1992). After adjustment for
covariates there was little association with pre- and perinatal lead
exposure assessments but significant decrements in full-scale IQ of
between 3.7 and 4.8 points (depending on age) for each (natural) log
unit increase in lifetime average PbB concentration were observed. The
estimated effect size for the lifetime average exposure up to 3 years
of age was a loss of 5.3 IQ points in association with an increase in
PbB level from 0.48 to 1.44 µmol/litre (10 to 30 µg/dl).
The Block Design subscale of the WISC-R, which tests spatial
abilities, exhibited the strongest association with lead exposure and
estimated effect sizes were stronger in girls than boys for both the
MSCA and WISC-R.
i) Sydney study
At the age of 4 years, 207 children were assessed with the
McCarthy Scales of Children Abilities (MSCA) and virtually no
significant associations with any measures of PbB were observed
(Cooney et al., 1989b).
Follow-up assessments in 175 children using WISC-R at age 7 also
yielded no associations (crude or adjusted) with blood lead history
(Cooney et al., 1991).
8.3.5.5 Questions prospective studies have not answered
A disappointment of the prospective studies was their inability
to reach any obvious consensus on the behavioural phenotype associated
with low-level lead exposure, or on age(s) of critical sensitivity.
This latter fact may reflect the phenomenon of intra-individual
"tracking", whereby an individual maintained approximately consistent
ranking with respect to his or her PbB concentration at any age, or it
may reflect the need for chronic exposure over extended periods in
order for causal effects to become apparent.
8.3.5.6 Attempting a consensus
As discussed previously, no clear delineation of an age (or age
range) of maximal sensitivity to lead exposure has emerged from the
prospective studies, and there is a diversity of reporting styles
which makes attempts to compare studies and establish a consensus of
opinion very difficult.
Nevertheless, it would appear essential to attempt some sort of
synthesis of studies performed so far, given that the social and
economic consequences of preventing an adverse health effect are
potentially enormous.
A meta-analysis by Needleman & Gatsonis (1990) concluded that the
overall pattern of results was most unlikely to have occurred by
chance. As an approach to the problem of obtaining the "best" estimate
of the effect of lead exposure on neuro-behavioural development, a
common estimate of the partial correlation between lead burden and IQ
was determined. However, in order to derive such an estimate with some
degree of certainty, it is essential to find a common outcome, and to
identify studies for which at least broadly comparable exposure
measures are available.
Schwartz (1993) has identified three prospective studies and five
cross-sectional studies that examined the association of lead exposure
as determined by PbB concentrations with full scale IQ. For the
prospective studies, the exposure measures used were 24-months PbB
level (Boston) and lifetime average up to 3 years of age (Cincinnati
and Port Pirie).
There was only one measure for each of the cross-sectional
studies. Despite the compromises necessary for such a comparison, the
estimated effect sizes for an increase in PbB level from 0.48 to
0.96 µmol/litre (10 to 20 µg/dl) for all seven studies lay between 1
and 6 IQ points lost (with five of the studies lying between 1 and 4),
and the associated 95% confidence limits only just embraced the value
zero for 3 of the 7 studies.
Thus, a broad consensus does emerge from a rough comparison. From
the estimates summarized by Schwartz (1993), a weighted mean for the
six coefficients accompanied by a standard error of 0.4 was estimated
to be 2.6 IQ units lost for an increase in PbB from 0.48 to
0.96 µmol/litre (10 to 20 µg/dl).
8.3.6 Task group overview and interpretation of prospective studies
on children
8.3.6.1 Rationale
As a complement to the narrative review, prepared by the Task
Group, a quantitative overview of the findings of the prospective
studies was considered to be a valuable means of assessing the overall
strength of evidence for an association between blood lead summary
measures and school age IQ.
While individual studies vary somewhat in their design and their
analysis strategies, their underlying objectives are closely related.
For any generally applicable conclusions on general population lead
exposure and its effect on intellectual attainment to be reached, some
consistency of evidence across studies is highly desirable.
Since any single study has considerable random error in
estimating a relationship, a quantitative overview (or meta-analysis)
can be a valuable means of defining what is the plausible magnitude of
statistical association between PbB measures and child IQ.
It seems appropriate, given the quite different types of study
design, to present two separate meta-analyses dealing with prospective
studies and cross-sectional studies, respectively.
Any such meta-analysis needs cautious interpretation. The caveats
in drawing causal inferences from statistical associations apply to
any meta-analysis just as they do to individual studies. Any increase
in statistical precision regarding the magnitude of association does
not eliminate the potential for bias in any observational study on
human populations.
8.3.6.2 The prospective studies
There are four prospective studies: Boston (Bellinger et al.,
1992); Sydney (Cooney et al., 1989a,b); Cincinnati (Dietrich et al.,
1993a); and Port Pirie (Baghurst et al., 1992) which presented
quantitative results in a form suitable for an overview (i.e. with
regression coefficients and standard errors). The Task Group
concentrated on school age assessment because:
a) the studies used the same outcome measure (WISC-R), which is
widely accepted and has been previously evaluated in cross-
sectional studies; and
b) intellectual assessment at primary school age (6-10 years) is,
perhaps, of greatest overall relevance in reaching a consensus on
the public health importance of childhood lead exposure.
Unfortunately, the Cleveland study did not publish findings in
the same quantifiable manner. The potential impact of this apparently
"negative" study on the overall evidence will be discussed in section
8.3.6.2(c).
a) Measures of exposure, outcome and association
All four studies have used blood lead as the measure of body lead
burden, but the timing and frequency of examinations has varied as has
the choice of summary measures (averages over time).
In a meta-analysis it seems appropriate to concentrate on
measures of association after adjustment for confounders. While there
is some variation in choice of confounders, the two key covariates
(mother's IQ and HOME scores) appear in all four studies.
The Port Pirie study used the logarithm of the PbB concentration
in the multiple regression analysis while the other three studies used
untransformed PbB concentration. This difference in statistical style
can be reconciled by converting the lead regression coefficients to
estimated changes in IQ for a specific increase in PbB concentration,
i.e. from 0.48 to 0.96 µmol/litre (10 to 20 µg/dl). This equals 10
times the regression coefficient (as published) for the Boston and
Cincinnati studies and log 2 times the regression coefficient for the
Port Pirie study.
It is recognized that the specific subscales of performance and
verbal IQ are of separate interest (e.g., the performance IQ has been
rather consistently shown to be more reliably associated with
postnatal lead exposure amongst the prospective studies (McMichael et
al., 1988; Bellinger et al., 1991a; Dietrich et al., 1991, 1992,
1993b). However, it appears more important to focus on full-scale IQ
as the primary outcome measure, otherwise concerns about post-hoc
selection may be raised. Such full-scale IQ was measured at age 6.5, 7
and 10 years in the Cincinnati, Port Pirie, Sydney and Boston studies,
respectively.
b) Display of individual study findings
Since most studies have not shown perinatal PbB level to be
predictive of intellectual performance, attention will be focused on
the various measures of postnatal PbB level. In fact, there are seven
such measures in each study that have been related to full-scale IQ in
the reports. Tables 21 and 22 summarize the magnitude of association
together with the standard error for all reported analyses, each after
adjustment for confounders.
Except for the earliest postnatal PbB values in the Boston and
Cincinnati studies, the adjusted associations between PbB level and IQ
are consistently negative (i.e. inverted). However, the "random noise"
in each study's analysis means that there is considerable variation in
levels of statistical significance. Hence, there is a need for a more
systematic overview combining the evidence from all studies.
c) A quantitative overview
The prime difficulty here is the different choices of PbB
summaries in the four studies. In undertaking a meta-analysis it would
have been better if all studies had essentially the same PbB measures
in their multiple regressions. Possible choices might have been (i) a
specific time point, e.g., 2 years or (ii) the means over a specific
interval, e.g., 0 to 5 years.
However, from the available analyses, some broadly similar
summaries can be chosen, as follows:
Table 21. Magnitude of association between blood lead and full-scale IQ
from four prospective studies
No. of Age at which blood lead Estimated change in
children was measured full-scale IQ a
Boston 148 year 1 0.0 (1.6)
year 1´ -1.2 (1.8)
year 2 -5.8 (2.1)
year 5 -2.6 (2.9)
year 10 -4.6 (5.2)
mean of years 2-5 -8.2 (2.8)
mean of years 2-10 -8.6 (3.4)
mean of 6 months to
10 years (unpublished) -5.7 (3.2)
Cincinnati 251 mean of year 1 0.1 (1.4)
mean of year 2 -0.2 (0.8)
mean of year 3 -1.3 (0.9)
mean of year 4 -1.5 (1.0)
mean of year 5 -2.3 (1.1)
mean of year 6 -3.3 (1.4)
mean of years 1-6 -1.3 (1.1)
Port Pirie 490 mean of first 15 months -2.8 (1.4)
mean of years 1-2 -3.2 (1.5)
mean of years 1-3 -3.3 (1.6)
mean of years 1-4 -3.2 (1.7)
mean of years 1-5 -3.0 (1.7)
mean of years 1-6 -2.8 (1.7)
mean of years 1-7 -2.6 (1.7)
Sydney 175 mean of years 1 and 2 -2.7 (2.8)
mean of years 3, 4 and 5 -1.9 (1.9)
year 7 -1.4 (1.7)
mean of years 1-7 -1.6 (2.2)
a From 0.48 to 0.96 µmol/litre (10 to 20 µg/dl) blood lead
(and its standard error) after adjustment for confounders
Table 22. Association between blood lead levels and full-scale
IQ from ten cross-sectional studies
No. of Estimated change in
subjects full-scale IQa
Lavrion, Greece 509 -2.7 (0.7)
Edinburgh, Scotland 501 -2.6 (0.9)
Greenwich, England 129 -5.6 (3.2)
European, multi-centre
Bucharest, Romania 301 -0.4 (2.6)
Budapest, Hungary 254 +0.8 (1.8)
Modena, Italy 216 +0.9 (4.2)
Sofia, Bulgaria 142 +2.2 (2.3)
Dusseldorf 1, Germany 109 -4.6 (4.5)
Dusseldorf 2, Germany 109 -3.9 (5.1)
Zagreb, Croatia 48 -1.5 (4.5)
a From 0.48 to 0.96 µmol/litre (10 to 20 µg/dl) blood lead
(and its standard error) after adjustment for confounders
i) There is some advantage in considering the mean PbB level over a
number of years, since it summarizes cumulative exposure and also
achieves a more reliable ranking of individuals than a single value.
The available long-term means are:
Boston: mean of 6 months to 10 years (D. Bellinger,
personal communication to the IPCS, 1993)
Cincinnati: mean of years 1 to 6
Port Pirie: mean of years 1 to 7
Sydney: mean of years 1 to 7
Although they differ in frequency and age range, these three
summaries should be very highly correlated with one another. The
resultant meta-analysis is displayed in Fig. 15. Each of the
individual study confidence intervals includes zero, indicating that
significance at P < 0.05 was not reached. However, the combined
evidence, weighting studies according to the inverse of their
variance, produces a weighted mean decrease in full-scale IQ of 2
points for a 0.48-µmol/litre (10-µg/dl) increase in PbB level, with
95% confidence interval from -0.3 points to -3.6 points (P = 0.01).
ii) An alternative approach is to consider PbB level at a specific
time or average over shorter intervals of time. For this approach, the
analyses of these studies are most comparable during the first three
years, as follows:
Boston: 2 year measure
Cincinnati: mean of year 3
Port Pirie: mean of years 1 to 3
Sydney: mean of years 1 and 2
This second meta-analysis is displayed in Fig. 16. The data here
support an inverse association more strongly than in Fig. 15, and the
combined evidence estimates a mean decrease of 2.6 IQ points for a
0.48-µmol/litre (10-µg/dl) increase in PbB level, with 95% confidence
interval from -1.2 points to -4.0 points (P < 0.001).
However, it should be noted that the estimates here are heavily
dependent on the choice of time points for PbB level. For instance, if
one instead chose:
Boston: 1.5 year measure
Cincinnati: mean of year 2
Port Pirie: mean of years 1 and 2
Sydney: mean of years 1 and 2
then the combined estimate is roughly halved in magnitude and is of
borderline significance.
One needs to recall that the other prospective study, carried out
in Cleveland (Ernhart et al., 1989a), has no equivalent data, but
reported no significant association. This study comprised about 150
evaluated children, and would influence the overall evidence in a less
significant direction if the data were able to be included.
8.3.6.3 A quantitative assessment of the cross-sectional studies
There have been more cross-sectional studies that have related
body burden to full-scale IQ in school-age children, but they cannot
all be included in a single meta-analysis. Here attention is focused
on the PbB studies, and it would be appropriate subsequently to
McIntosh et al., 1985) in rat brain. In humans the exact relationship
between PbB and plasma biopterins has not been properly defined.
8.1.4 Nicotinamide adenine dinucleotide synthetase
A recent study of nicotinamide adenine dinucleotide (NAD)
synthetase in erythrocytes indicates that this enzyme is sensitive to
inhibition by lead (and zinc) and is a sensitive indicator of lead
exposure. Zerez et al. (1990) found NAD synthetase activity reduced in
three subjects with elevated PbB levels in the range
1.63-3.46 µmol/litre (34-72 µg/dl).
8.1.5 Nutritionally affected groups
A body of evidence, summarized and discussed by Mahaffey (1985),
indicates that a high dietary calcium intake tends to decrease lead
absorption and retention in infants, young children and adults. Other
evidence suggests that in groups including children, with
self-selected diets, low dietary calcium intakes are associated with a
greater prevalence of elevated PbB levels (Mahaffey, 1985).
Some human data indicate that adults with low iron states have an
increased absorption of both iron and lead (Watson et al., 1980,
1986). In children, particularly those from low-income families, there
is often an association between iron deficiency and elevated PbB (Yip
& Dallman, 1984; Mahaffey, 1985). While this by itself does not
indicate a causal relationship, when considered in the context of data
on adults and animals, it suggests that iron deficiency in children
can result in increased lead absorption.
Alcoholics and people who consume excess alcohol may be at
increased risk of adverse effects from lead. In animal studies,
alcohol and lead synergistically inhibit ALAD, hepatic glutamic
oxaloacetic transaminase and glutamic pyruvic transaminase activities
(Flora & Tandon, 1987) supporting the hypothesis regarding risks to
humans.
8.2 Haematopoietic system
Lead-induced anaemia can be a direct consequence of inhibition of
haem biosynthesis; it is not necessarily associated with iron
deficiency. It may be associated with alterations of globin syntheses
(Albahary, 1972). More importantly, the synthesis of alpha- and
ß-globin chains may become asynchronous (White & Harvey, 1972).
8.2.1 Anaemia
Based on data published by Lilis et al. (1978), Baker et al.
(1979), Grandjean (1979), and earlier data, the threshold PbB level
for a decrease in haemoglobin has been estimated to be 2.40 µmol/litre
(50 µg/dl) for occupationally exposed adults (US EPA, 1986a).
Grandjean et al. (1989a) demonstrated the reduced ability of the
erythropoietic system to regenerate after blood withdrawal
(0.45 litre) in 25 lead-exposed battery workers (average PbB level
2.14 µmol/litre, 44.5 µg/dl) compared with 25 age-matched controls
(average PbB level 0.35 µmol/litre, 7 µg/dl). The haematological
parameters, except for erythropoietin were otherwise comparable for
the two groups. The effect was attributed to an effect on haem
synthesis and possible decreased erythrocyte survival in lead-exposed
workers. Lead has been found to depress serum levels of
erythropoietin, a hormone which regulates erythrocyte formation
(Graziano et al., 1991), which might also affect the reserve capacity
for erythropoiesis.
The PbB threshold for decreased haemoglobin levels in children is
estimated to be approximately 1.92 µmol/litre (40 µg/dl) (IPCS, 1977).
However, a cross-sectional epidemiological study of 579 children, aged
1-5 years in 1974, living in close proximity to a primary lead smelter
showed that adverse effects on haematocrit may occur at lower PbB
levels (Schwartz et al., 1990). Anaemia, defined as a haematocrit
below 35%, was not found at PbB levels of less than 0.92 µmol/litre
(20 µg/dl). There was a strong non-linear dose-response relationship
at higher PbB levels, which was influenced by age.
Kutbi et al. (1989) studied 200 boys aged 6-8 years. The mean PbB
was 0.33 µmol/litre (6.9 µg/dl; range 0.07-1.14 µmol/litre or
1.4-23.8 µg/dl). Subdividing the group at 0.72 µmol/litre (15 µg/dl),
they found a negative correlation between PbB level and all
haematological values for the "upper normal" (N = 7) compared with the
"normal" group (N = 193). The pattern of haematological parameters was
described as predictive of microcytic anaemia.
Anaemia has been commonly associated with the adverse effects of
occupational lead exposures. It is an effect that is easily diagnosed
clinically and is recognized as a marker of lead toxicity. Anaemia may
result from either a decrease in haemoglobin production or an increase
in the rate of destruction of erythrocytes. An analysis, made in 1974,
of the association between PbB levels and haematocrit in 579 children
(1-5 years of age) living near a primary lead smelter has recently
been presented (Schwartz et al., 1990). A haematocrit value of less
than 35% was used to indicate an adverse effect. It should be noted
that the effect of iron deficiency was not taken into account in
analysing the results. The study concluded that there was no adverse
effect of lead at PbB levels below 0.96 µmol/litre (20 µg/dl).
Furthermore, the risk of having a haematocrit value below 35% for
1-year-olds was 2% at PbB levels between 0.96 and 1.87 µmol/litre
(20 and 39 µg/dl). The degree of iron deficiency may account for a
substantial proportion of this 2%. In this study, the level at which
an effect of lead on the induction of anaemia was demonstrable was
about 1.92 µmol/litre (40 µg/dl).
8.2.2 Pyrimidine-5'-nucleotidase activity
Inhibition of erythrocyte pyrimidine-5'-nucleotidase leads to
accumulation of pyrimidine nucleotides, which has been associated with
induction of basophilic stippling.
Impaired function of erythrocyte pyrimidine-5'-nucleotidase
occurs in lead-exposed workers and as a genetically induced enzyme
deficiency. In in vitro systems the enzyme is inhibited by lead,
cadmium, mercury and zinc ions (and perhaps other metal ions) (Paglia
et al., 1975; Pagliuca et al., 1990; Ichiba & Tomokuni, 1990).
Graphs of PbB concentration against erythrocyte pyrimidine-
5'-nucleotidase activity in children have been reported for 21
children aged 2-5 years (Angle & McIntire, 1978) and for 42 aged 1-5
years (Angle et al., 1982) who participated in a preventative
programme because they had previously had PbB levels above
1.44 µmol/litre (30 µg/dl) or zinc protoporphyrin (ZPP) greater than
60 µg/dl. Both plots show considerable scatter although there was a
significant inverse logarithmic correlation. However, data relating to
the reproducibility, precision and accuracy of assessing inhibition of
erythrocyte pyrimidine-5'-nucleotidase were not presented.
This test may be capable of analytical refinement when assessment
of the sensitivity to lead (and other metals) exposure might more
reasonably be made. It does not yet appear likely to provide a routine
measure for low-lead exposure assessment.
8.2.3 Erythropoietin production
Graziano et al. (1991) found depressed serum erythropoeitin
levels in females at mid-pregnancy and at delivery associated
statistically with PbB level. Erythropoietin is a glycoprotein
produced in the renal proximal tubules which regulates both
steady-state and accelerated erythrocyte production. The study was not
adequate to define a dose-response relationship.
8.3 Nervous system
8.3.1 Historical perspective
Gross toxic effects of lead on the nervous system were reported
by ancient Greek physicians. A brief summary of such reports, and
those of Roman physicians and 18-19th century toxicologists, was
published by Kazantzis (1989). The syndrome was known as "painter's
colic", which included abdominal pain, constipation and paralysis,
symptoms now covered by the term "lead encephalopathy".
Lead colic was usually accompanied by effects on the nervous
system (lead palsy) in cases of acute and chronic poisoning, which
were often but not always fatal. Lead poisoning was well recognized as
an occupational hazard. However, it was also associated with
consumption of lead-contaminated water, wine, cider, rum from the West
Indies, and food prepared or stored in lead or lead-glazed utensils.
For example, Kazantzis (1989) described cases of lead poisoning in
girls resulting from consumption of drinking-water kept in lead-lined
cisterns, and also described the Devonshire colic attributed by Sir
George Baker in 1767 to the consumption of cider prepared in presses
made of lead or lead alloys.
The early toxicologists clearly recognized that the dose
(exposure) and form of lead was important in relation to absorption
and were able to demonstrate the presence of lead in tissues of fatal
cases of poisoning. There are many individually described, detailed
case studies in their writings.
8.3.2 Neurotoxic effects in adults
Despite the long recognition of lead poisoning, new clinical
cases continue to be the subject of published reports (Cueto et al.,
1989; Kocak et al., 1989; Zuckerman et al., 1989; Friedman &
Weinberger, 1990; Gupta et al., 1990; Mitchell-Heggs et al., 1990;
Nosal & Wilhelm, 1990; Sharma et al., 1990; Schneitzer et al., 1990;
Veerula & Noah, 1990). Although they are important, the numbers of
these poisoning cases are comparatively small compared with the
population at risk to the potential effects of exposure to lead from
environmental and dietary sources. Of particular recent concern have
been the possible neurotoxic effects on the developing child.
8.3.2.1 Central nervous system
In a study of 158 secondary lead smelter workers, Lilis et al.
(1977) found that 64% reported CNS symptoms and that there was early
occurrence of symptoms within 1 year of exposure. PbB concentrations
were elevated (about 3.36 µmol/litre or 70 µg/dl) at the time of
examination but many of the subjects had received chelation therapy.
Fischbein et al. (1979) noted CNS symptoms in 21 of 81 employees
of law enforcement agencies working in firing ranges. Symptoms
correlated with PbB levels and were present in three-quarters of
subjects having a PbB level > 2.4 µmol/litre (50 µg/dl).
Hanninen et al. (1979) reported that 49 lead-exposed workers
(including 21 male and 8 female storage battery and 16 railroad
engineering workshop employees) whose PbB level had never exceeded
3.36 µmol/litre (70 µg/dl) experienced more subjective symptoms than a
control group; excess symptoms were proportional to lead uptake.
Fischbein et al. (1980) studied 90 telephone cable splicers
having intermittent exposure to lead. They reported that 26 complained
of CNS symptoms (mean PbB level of 1.36 µmol/litre, 28.4 µg/dl, and
ZPP of 70.3 µg/dl) compared with controls (mean PbB level of
1.32 µmol/litre, 27.4 µg/dl, and ZPP of 49.3 µg/dl).
Awad El Karim et al. (1986) studied 92 lead acid battery workers
and compared them with 40 oil mill worker controls. They found CNS
symptoms in 50% of lead-exposed workers and measured PbB levels in 46
subjects working in different sections of the plant. More than 95% of
exposed workers had PbB levels above 1.92 µmol/litre (40 µg/dl),
whereas the mean PbB in the control group was 1.0 µmol/litre
(21 µg/dl).
Impairment of psychological and neuropsychological test
performance has been reported for lead-exposed workers (Hanninen et
al., 1979; Hogstedt et al., 1983; Parkinson et al., 1986; Araki et
al., 1986a,b; Huang et al., 1988a; Stollery et al., 1989, 1991).
The critical flicker fusion test has been reported to be a
sensitive indicator of CNS changes associated with exposure to lead.
Wooller & Melamed (1978) found significantly lower critical flicker
fusion in subjects with PbB levels above 2.88 µmol/litre (60 µg/dl)
than in those with levels below 0.96 µmol/litre (20 µg/dl). Williamson
& Teo (1986) found a significant decrease in mean flicker fusion in 59
lead-exposed workers (mean PbB > 2.3 µmol/litre or 48 µg/dl) compared
with matched controls. However, Gennart et al. (1992b) found no
difference between lead-exposed workers (mean PbB level
2.45 µmol/litre or 51 µg/dl, N = 98) and controls (mean PbB level
1.00 µmol/litre or 20.9 µg/dl, N = 85).
Stollery et al. (1989) studied the performance of 91 men,
occupationally exposed to inorganic lead, in a series of tests
designed to assess cognitive function. Subjects were grouped in "low"
(mean PbB 0.48 µmol/litre or 10 µg/dl, mean ZPP 8.7 µg/dl, N = 28),
"medium" (mean PbB 1.44 µmol/litre or 30.1 µg/dl, mean ZPP 22.4 µg/dl,
N = 27) and "high" (mean PbB 2.43 µmol/litre or 50.7 µg/dl, mean ZPP
58.8 µg/dl, N = 36) ranges of PbB concentrations. Results showed that
occupational exposure to lead impaired performance in a range of tests
(sensory motor reaction time, memory, attention, verbal reasoning,
spatial processing). Workers having PbB levels in excess of
1.92 µmol/litre (40 µg/dl) showed clear evidence of impairment on
tests of serial reaction time and category search. The authors
concluded that sensory motor, rather than cognitive, requirements of
many psychological tasks provide the most sensitive index of the early
effects of chronic low level lead exposure.
In a follow-up study (Stollery et al., 1991), 70 workers were
re-tested (three times within 8 months). Performance deficits in the
"high" (mean PbB 2.49 µmol/litre or 51.8 µg/dl, mean ZPP 77.4 µg/dl,
N = 22) group were not altered by practice or continued exposure. The
main deficit was a slowing of sensory motor reaction time coupled with
difficulties in remembering incidental information. There was little
evidence of impairment in workers having PbB levels less than
1.92 µmol/litre (40 µg/dl).
Finally, it appears that neuroelectrophysiological testing is a
sensitive and objective indicator of the CNS effects of lead. The
results of Araki et al. (1986b, 1987, 1992) using short-latency
somatosensory and visual evoked potentials and auditory event-related
potential (P300) indicate that subclinical electrophysiological
effects of lead occur not only in peripheral nerves but also in the
CNS.
8.3.2.2 Peripheral nervous system
Peripheral neuropathy is a common sign of chronic, high level
lead exposure, often manifesting as weakness in the upper or lower
limbs. At lower levels of lead exposure, nerve conduction velocity
(NCV) has provided a more sensitive indicator of peripheral nerve
dysfunction. More than 30 published studies have measured the
conduction velocity of electrically stimulated sensory and motor
nerves in workers exposed to lead. However, these studies have yielded
somewhat mixed results, with many showing a decrease in NCV in
relation to lead exposure (generally indexed as PbB concentration) and
a few showing no effect or occasionally even an increase in NCV
associated with lead exposure (Seppäläinen & Hernberg, 1980; Davis &
Svendsgaard, 1990).
Various reasons may underlie this lack of uniformity in NCV
findings. For example, studies may differ in methodological features,
in the characterization of lead exposure (e.g., single time-point
versus time-weighted average PbB levels) or in the handling of
confounding variables such as nerve temperature and age of subject
(Ehle, 1986). Other important factors accounting for some of the
apparent inconsistency in this area of research may be the possible
antagonistic effect of zinc to lead and differences in nerves selected
for measurement in different studies (e.g., slow versus fast fibres
(Araki et al., 1986c; Murata & Araki, 1991). A statistical
meta-analysis and critical review of 32 NCV studies by Davis &
Svendsgaard (1990) indicated that the median motor nerve shows effects
of lead more reliably than other nerves (e.g., median sensory or
ulnar).
Despite these complications, certain key well-conducted studies
provide compelling evidence of a causal relationship between lead
exposure and reduction in NCV. Araki et al. (1980) measured median
motor NCV before and after PbB levels of workers were lowered through
chelation therapy. Depending on the amount of reduction in PbB level
achieved by chelation and on a given worker's baseline NCV,
significantly improved NCV was measured in 7 out of 14 lead-exposed
workers. For all 14 workers the improvement in NCV was significantly
correlated with the decrease in PbB level (r = -0.573, P < 0.001).
In another key study, Seppäläinen et al. (1983) followed workers
prospectively from the beginning of their employment in a battery
plant, dividing them into two exposure categories (above and below the
median PbB level of 1.44 µmol/litre (30 µg/dl). Median motor NCV was
significantly reduced in the workers with blood lead levels above
1.44 µmol/litre (30 µg/dl) at 1-, 2-, and 4-year evaluation points,
despite attrition (and reduced statistical power) that resulted in
only five subjects per group after 4 years. Based on this well-
conducted prospective study, 1.44 µmol/litre (30 µg/dl) would appear
to be the lowest-observed-adverse-effect level for reduced NCV in
adults. Although another well-conducted prospective study (Spivey et
al., 1980) was unable to find an effect of lead on NCV at higher
exposure levels, NCV in the median nerve was not measured in that
study. Triebig et al. (1984) reported a dose-dependent decrease in NCV
at PbB levels above 3.36 µmol/litre, no dose-effect relationship being
detectable below this concentration.
8.3.2.3 Autonomic nervous system
There have been two reports of tests for autonomic nervous
function following lead exposure. Teruya et al. (1991) conducted a
cross-sectional survey of 172 male workers exposed to lead (mean PbB
level, 1.73 µmol/litre or 36 µg/dl, range 0.24-3.648 µmol/litre or
5-76 µg/dl), by measuring electrocardiographic R-R interval
variability (CVRR). Age-adjusted CVRR during deep breathing in
workers with PbB levels above 1.44 µmol/litre (> 30 µg/dl) was
significantly decreased as compared to those with PbB levels less than
0.96 µmol/litre (< 20 µg/dl). In addition, significant dose-response
and dose-effect relationships were observed between the PbB levels and
R-R interval variation during deep breathing among the workers with
PbB levels above 0.96 µmol/litre (> 20 µg/dl).
In a study reported by Murata & Araki (1991), the R-R interval
(CVRR) and two component coefficients of variation in the R-R
interval (C-CVRSA) were measured in 16 gun metal foundry workers
exposed to lead, zinc and copper (mean PbB level, 1.63 µmol/litre or
34 µg/dl, range 0.768-2.88 µmol/litre or 16-60 µg/dl). The CVRR and
component C-CVRSA (respiratory sinus arrhythmia) were significantly
lower in the workers than in age-matched controls, whereas the
C-CVMWSA (Mayer wave-related sinus arrhythmia) was unaffected. The
authors noted that zinc may have been antagonistic to the lead effects
in this study. Since the CVRR during deep breathing and the
component C-CVRSA of respiratory sinus arrhythmia reflect
parasympathetic activity, these two reports indicate potential
dysfunction of the autonomic nervous system (mainly, parasympathetic
activity) at average PbB levels of approximately 1.68 µmol/litre
(35 µg/dl).
8.3.3 Neurotoxic effects in children
The majority of the epidemiological research on the health
effects of lead has been focused on children because, in comparison
with adults, they are more vulnerable to lead in several respects
(Davis & Grant, 1992). For example, children typically engage in
hand-to-mouth activities (sucking fingers, putting food or other
objects in the mouth) that result in greater ingestion of lead than
adults normally experience. Also, because of their greater absorption
and retention of lead, the body burdens in children resulting from a
given external exposure level tend to be higher than adults.
Furthermore, the relatively greater exposures and body burdens of
children occur during sensitive periods of development. Finally, it
appears that children are generally more sensitive to the
toxicological effects of lead at a given internal exposure (PbB)
level. The lowest-observed-effect levels for various end-points (e.g.,
encephalopathy, anaemia, reduced haemoglobin, elevated EP, slowed NCV,
impaired neurobehavioural function) are lower in children than in
adults (US EPA, 1986a, 1990).
Section 8.3.3.2 describes findings from the main epidemiological
cross-sectional studies that have been published since 1979. This
constitutes a very substantial collection of research findings but has
certain interpretive limitations, particularly the lack of information
on exposure history.
More recent research effort has concentrated on prospective
epidemiological studies of birth cohorts repeatedly evaluated, mostly
up to the school age years. These prospective studies are described in
section 8.3.3.3.
Section 8.3.3.4 presents a quantitative overview of the
collective evidence from the prospective studies and discusses their
interpretation and the problems of inferring causality from such
observation data.
8.3.3.1 Historical perspective
While there have undoubtedly been cases of lead poisoning in
children as long as lead has been used by man, most of the attention
was previously given to cases of poisoning as a result of occupational
exposure in adults. In 1892 Gibson and colleagues in Australia
reported a case series of ten young children with lead colic. It was
not until twelve years later that peeling paint in the children's
homes was identified as the lead source (Gibson, 1904).
In 1943, Byers & Lord (1953) reported the results of an important
study, the first to suggest that there were neurological after-effects
of lead poisoning, in the absence of cerebral oedema and high
intracranial pressure, following acute lead encephalopathy. However,
in other cases it was felt that neurological manifestations
disappeared if the ingestion of lead was stopped (McKhann & Vogt,
1933). The case series of 20 school-age children had been hospitalized
for lead poisoning but without signs of acute lead encephalopathy, and
all were later discharged as cured. At follow-up, only one of the
children was making satisfactory progress at school. The children were
described as showing a variety of symptoms, including poor academic
achievement, intellectual deficits, sensory-motor deficits and
behaviour disturbance.
In the years following the findings of Byers & Lord (1953), a
number of case control studies were carried out that examined mental
retardation (Beattie et al., 1975) or hyperactivity (David et al.,
1972).
In addition, as a result of the growing concern about the dangers
of lead encephalopathy, studies were set up to investigate the
possibility that subclinical levels of lead (i.e. levels producing no
overt signs of lead encephalopathy) cause more subtle neurological
damage to children. Many of these studies investigated children
identified by screening clinics as having raised lead burdens, but
others were sited around smelters or lead works where children were
found to have elevated lead levels. The studies were important in
determining the areas of functioning which might be affected, and in
drawing attention to the methodological problems involved in carrying
out such studies. The main difference between the smelter studies or
studies around lead works, and clinical studies is that in the former
the primary source of environmental lead is known. In the hospital or
clinical studies it is likely that the ingestion of paint flakes, or
less often leaded putty, was in most cases the primary cause of the
excess lead exposure, although the exact reason for the increased lead
burden is rarely known.
Clinical and smelter studies were carried out in the United
Kingdom and USA and in general investigated children with PbB levels
above 1.92 µmol/litre (40 µg/dl), and compared their performance with
that of children with levels below this value.
Although some of the clinical and smelter studies found a
lead-associated deficit (Perino & Ernhart, 1974; Landrigan et al.,
1975), others did not (Lansdown et al., 1974). In general the studies
were methodologically flawed, with small samples lacking in
statistical power, biased ascertainment of subjects or controls, and
inadequate control for potentially confounding co-factors. The large
majority did not control for parental intelligence, which is the
variable most strongly associated with child IQ. These studies were,
however, important in suggesting an association between body lead
levels and performance, and also in demonstrating that there is an
association between social disadvantage and higher body lead levels.
8.3.4 Population-based cross-sectional studies on children
Most of the general population-based epidemiological
cross-sectional studies have, in addition to intelligence tests,
included extensive batteries of tests covering several other areas of
psychometric functioning, such as academic attainment, behaviour,
gross motor abilities and fine motor co-ordination, reaction time,
visuo-spatial skills and memory, and auditory memory. There are
differences in the tests used, and in the areas of functioning
investigated, and the results are less consistent and more difficult
to interpret than those relating to IQ tests. For that reason this
section will focus mainly on results relating to intelligence tests,
and results from other functional domains will be mentioned briefly
where appropriate.
There are many cross-sectional population studies using either
blood or teeth as the primary measure of lead body burden, and only
the more informative, in terms of providing information on risk
assessment, will be included. The majority of the studies described
used either a full or short form of the Wechsler Intelligence Scale
for Children - Revised (WISC-R or its translations) as the measure of
intelligence. Exceptions are the studies of Fulton et al. (1987),
which used the British Ability Scales (BAS), Harvey et al. (1988),
which used Wechsler Pre-school and Primary Scale of Intelligence
(WPPSI), and Schroeder et al. (1985) and Hawk et al. (1986), where the
Stanford-Binet Intelligence Scale was used.
8.3.4.1 Tooth lead studies
a) Needleman et al. (1979)
A total population of 3329 children aged 6-7 years, attending
schools in two working class towns near Boston, USA, were asked to
donate their shed deciduous teeth. Of these 70% (2335) children did
provide teeth. A 1-mm slice of tooth consisting primarily of dentine,
was split in half, and one portion was analysed by ASV. Children whose
initial tooth slice was in the highest 10th percentile (> 20 µg/g) or
the lowest 10th percentile (< 10 µg/g) were provisionally classified
into a high or low lead group. A total of 524 children were originally
classified into these lead groups, but 254 of these children were not
tested for a number of reasons, and the results for another 112 were
excluded from data analysis for medical reasons, or because a second
tooth analysis provided a discordant result. The results for 158
children (58 high lead children and 100 low lead children) were
presented. Thirty-nine non-lead variables were scaled and coded, and
those which were found to differ between the lead groups at the 10%
level were controlled as covariates. The covariates controlled were
father's socioeconomic status, mother's age at subject's birth, number
of pregnancies, mother's education and Peabody Picture Vocabulary
score. After this was done the children in the high lead group
performed significantly less well on the IQ test with a 4´ point
difference between the groups.
b) Winneke et al. (1983)
A total of 115 children aged 7-12 years living in the vicinity of
a lead/zinc smelter in Stolberg, Germany, who had provided at least
one incisor tooth, were investigated. Whole tooth lead measures ranged
from 2 to 32 µg/g, with a geometric mean of 6 µg/g. There was a
marginally significant negative association between lead level and
(short-form) verbal IQ after taking age, gender, perinatal risk
factors and social confounding factors into account. There was a
difference of 4.6 IQ points when children with tooth lead levels above
10 µg/g were compared with those with levels below 4 µg/g. Significant
negative associations were also observed between tooth lead and the
error score of Bender Gestalt shape copying test and performance on
the Vienna reaction time device.
c) Smith et al. (1983); Pocock et al. (1987)
The target population for the study by Smith et al. (1983) (also
known as the Institute of Child Health/University of Southampton
study) was 6875 children aged 6-7 years attending schools in one of
three areas in or near London. In all, 7407 teeth were donated by 4105
children. Whole tooth analysis of intact incisor teeth donated by
eligible children was carried out and children whose tooth lead level
(expressed on an ashed weight basis) was in the lowest percentile
(< 2.5 µg/g), approximately at the 50th percentile (5-5.5 µg/g) or
above the 90th percentile (> 8.0 µg/g) were invited to participate.
The sample was stratified by social grouping. Of the 432 children
selected, 403 participated in the study. Parental interviews were
carried out in the home, and information on a wide range of social and
familial variables was collected. There was a 5 point difference in IQ
scores between the high and low lead groups before controlling for any
covariates, but this was reduced to 2 points once covariates
associated with IQ score were controlled. The authors concluded that
social factors explain the differences in test performance to such a
degree that the small differences that remain may be due to other
social factors not measured. The study underlined the importance of
the social environment for the child's performance and recognized the
importance of the relationship between body lead burden and social
disadvantage.
Blood samples were taken from 93 children 3-6 months after
shedding a tooth and after psychometric testing was completed. The
range of PbB levels in this group was 0.336-1.296 µmol/litre
(7-27 µg/dl), except for one measurement of 2.064 µmol/litre
(43 µg/dl), and the mean was 0.614 µmol/litre (12.8 µg/dl). The
correlation of PbB and PbT was 0.5.
A reanalysis of this data by Pocock et al. (1989) employed
multiple regression techniques, using an "optimal" regression
strategy. The results were comparable to those previously reported.
Interactional analyses showed no evidence of any differential
association with lead in different social groups, but revealed a
significant interaction between PbT and child gender. Significant
negative association between PbT and IQ was found in boys but not in
girls.
d) Fergusson et al. (1988a,b,c)
This study was carried out in Christchurch, New Zealand, on a
sub-sample of a birth cohort of 1265 children born in 1977. Teeth were
collected from 1035 children, and a dentine "chip" was analysed to
provide a PbT estimate for 996 children. The mean dentine lead level
was 6 µg/g. Children from disadvantaged backgrounds had higher mean
lead levels than those from more advantaged backgrounds, and this
difference persisted when other social environmental factors (such as
the effects of housing, proximity to main roads, and childhood pica)
were taken into account. A WISC-R and reading test were administered
to children at ages 8 and 9 years, and results were given for 724
children at age 8 and 644 children at age 9. Small correlations
between IQ scores and dentine lead were found, but control for
confounding variables reduced these to statistical non-significance
for both ages (a correlation of -0.03 to -0.05). Parental IQ was
assessed. Control for pica as a test of reverse causality further
reduced the associations between outcomes and PbT.
e) Hansen et al. (1989); Lyngbye et al. (1990a,b); Grandjean et al.
(1991)
Children entering schools in the town of Aarhus, Denmark, were
asked to donate shed teeth. Of a potential sample of 2414 children,
1291 (54%) provided usable teeth. A sample of circumpulpal dentine was
obtained from each tooth and analysed by AAS. The mean lead level in
dentine was 10.7 µg/g, with a log normal distribution and a range from
0.40 to 168 µg/g. The correlation of whole tooth lead and circumpulpal
dentine measures were low. Average circumpulpal dentine levels were
about five times higher than average whole tooth measures. Subsequent
analysis of PbB from samples obtained two to three years later showed
an overall mean PbB level of 0.25 µmol/litre (5 µg/dl), with a
geometric mean of 0.28 µmol/litre (range 0.08-0.63 µmol per litre or
1.66-13.1 µg/dl) in children selected for the high lead group, and a
mean of 0.18 µmol/litre (range 0.08-0.70 µmol/litre or mean
3.74 µg/dl, range 1.66-14.56 µg/dl) in children selected for the low
lead group, which confirms this as a low exposure group. Children
whose level of lead in circumpulpar dentine was above 18.7 µg/g (8%,
N = 110) were selected for the high lead group, and they were matched
on gender and socioeconomic status (SES) with children whose
circumpulpar dentine levels were below 5 µg/g. A comprehensive list of
exclusions, including neurological and medical conditions, atypical
social situations, and those who did not wish to take part, reduced
the sample to 156 children for whom results were presented. Initial
analysis (by matched pair t-test) showed a difference of 6 IQ points
on full-scale WISC scores, with a difference of nearly 9 points in
verbal IQ scores. The association of IQ measures with circumpulpar
dentine lead levels was evident across different socioeconomic groups.
Confounders associated with circumpulpar dentine lead levels (such as
pregnancy variables, SES, mother's educational status and child's
gender) were entered into a stepwise regression model, and explained
only a small portion of the variance. Lead levels in the dentine
accounted for a significant (2.8%) of the total variance in full scale
IQ. Parental IQ was not controlled.
f) Rabinowitz et al. (1991)
From 764 eligible children (attending grades 1 to 3 in 7 selected
schools in Taiwan), 940 deciduous teeth were collected, the majority
being incisors. The average age of students was 6.7 years. Two of the
schools were near lead smelters. Lead levels were determined in 862
teeth from 692 children using the Boston method which analysed a
"dentine chip" (Rabinowitz et al., 1989). The mean value was 4.3 µg/g
of dentine. In all, 493 children were tested using the Ravens Coloured
Progressive Matrices Test (CPM) and the IQ equivalents were related to
tooth lead levels. Confounders were selected from a set of 40, based
on associations with lead and IQ. After correction for seven
confounding variables (social and perinatal factors), the highly
significant raw exposure-effect association (r = -0.32) was markedly
reduced for the full sample to statistical insignificance, although a
significant lead-CPM association remained for the subsample of girls
(b = -1.8; SE = 0.78; P < 0.02).
8.3.4.2 Blood lead studies
a) Winneke et al. (1985)
In a further study by this group carried out in the lead/zinc
smelter town of Nordenham, Germany, 114 out of 378 children (46%) for
whom cord PbB levels were available were tested at 6-7 years of age.
The range of cord PbB levels was 0.19-1.49 µmol/litre (4-31 µg/dl) and
for current PbB levels was 0.19-1.10 µmol/litre (4-23 µg/dl); the
means for both perinatal and current PbB levels were 0.39 µmol/litre
(8.2 µg/dl, SD = 1.6). After correction for confounders by means of a
stepwise multiple regression analysis, few significant associations
between PbB levels and performance were observed. There was a marked
influence of social background factors, but little influence on
short-form full IQ score (current PbB accounted for 0.3% additional
variance), and a borderline decrease in performance IQ (PbB accounting
for 2.4% of the variance). In general, the associations were rather
stronger with current PbB levels than with cord PbB measures. The
strongest associations were found with the error scores of the
difficult version of the Vienna reaction time task.
b) Harvey et al. (1984)
From 483 eligible children from the city of Birmingham, United
Kingdom, 187 out of 284 contacted were examined at 30 months of age.
The average PbB level from venous samples was 0.75 µmol/litre
(15.6 µg/dl, SD = 4). Cognitive ability was assessed by means of four
tests from the British Ability Scales (BAS) and three tests from the
Stanford-Binet Intelligence Scale. The raw correlation between PbB
level and IQ was -0.17 (P < 0.05). Subsequent multiple regression
analysis with a predetermined set of confounders and a sample size of
48 yielded an insignificant association between lead and IQ.
c) Harvey et al. (1988)
In a second study, 201 out of 337 eligible children, aged 5.5
years, from the inner city area of Birmingham, United Kingdom, were
studied. The mean PbB level was 0.614 µmol/litre (12.8 µg/dl, SD = 4).
No significant associations between PbB level and IQ (WPPSI) were
observed after correction for confounding factors. Apart from some
statistically significant associations between PbB level and reaction
time, none of the remaining tests showed a significant correlation
with PbB level after confounder-control.
d) Yule et al. (1981)
Children (N = 166), aged 6 to 12 years, living in the vicinity of
a leadworks in outer London were examined. PbB levels from venous
samples ranged from 0.336 to 1.58 µmol/litre (7 to 33 µg/dl, geometric
mean = 0.648 µmol/litre or 13.5 µg/dl). After controlling for social
class as the only confounder, significant negative associations were
found for IQ (WISC-R; full-scale- and verbal-, but not performance-
IQ). An average IQ difference of 7 points was found when comparing
children with PbB levels of 0.62 µmol/litre (13 µg/dl) or more with
those of 0.576 µmol/litre (12 µg/dl) or less. Significant inverse
associations were also found between PbB level and scores on tests of
attainment, i.e. reading and spelling but not mathematics.
A group of 9-year-old children from a middle class area of London
were studied. Subjects were divided into low PbB (N = 80 and PbB of
0.336-0.576 µmol/litre, 7-12 µg/dl) and high PbB (N = 82 and PbB of
0.624-1.15 µmol/litre, 13-24 µg/dl) groups for testing the association
between PbB level and IQ. No significant association between PbB level
and IQ was found before or after controlling for covariates (Lansdown
et al., 1986).
e) Schroeder et al. (1985)
One hundred and four children from a high risk population, aged
10 months to 6.5 years and with PbB levels ranging from 0.288 to
2.83 µmol/litre (6-59 µg/dl) were tested for intellectual development
using age-appropriate tests. After control for social confounding a
significant negative association was found between PbB level and IQ.
Fifty children of this cohort were reassessed 5 years later when all
PbB levels were below 1.44 µmol/litre (30 µg/dl). Neither initial nor
current PbB level was significantly related to later IQ.
f) Hawk et al. (1986)
In an effort to replicate the above-mentioned findings, 75
children from the same group, then aged 3 to 7 years, and having a
mean PbB level of 0.998 µmol/litre (20.8 µg/dl), range
0.29-2.256 µmol/litre (6-47 µg/dl), were examined using the
Stanford-Binet test. There were no significant interactions between
PbB level and factors including age, sex, maternal IQ, quality of the
care-giving environment (HOME), or socioeconomic status. There was a
statistically significant negative association between PbB level and
IQ, although the characteristics of the regression coefficient for
lead depended on which covariates were included in the model. For the
final regression model that best controlled for confounding with the
greatest precision, i.e. the most accurate and precise model,
containing lead, maternal IQ, HOME and gender, highly significant
inverse associations were found between both mean and maximum PbB
levels and IQ.
g) Fulton et al. (1987)
Five hundred and one children, aged 6-9 years, out of 1210
eligible children from 18 primary schools in central Edinburgh,
Scotland, were tested for an association between PbB level, IQ and
attainment. The sample comprised all children in the top quartile of
the PbB distribution, and a random subsample (approximately 1 in 3) of
the remainder. The geometric mean PbB level was 0.55 µmol/litre, range
0.16-1.63 µmol/litre (11.5 µg/dl, range 3.3-34.0 µg/dl). After
correction for 31 potential confounders a significant negative
association between PbB level and IQ (BAS) was found. There was a
dose-response relation with no evidence of a threshold. For purposes
of statistical analyses, children were placed into 10 groups of about
50 each on the basis of PbB level. The mean PbB level was
0.27 µmol/litre (5.6 µg/dl) in the lowest and 1.06 µmol/litre
(22.1 µg/dl) in the highest group; there was a difference of 5.8 IQ
points between the two groups. The size of the effects of lead was
small relative to that of the other factors (0.9% of a total of 45.5%
explained by the full model).
h) Hatzakis et al. (1989)
Five hundred and thirty-three children out of 1038 eligible from
four primary schools in the vicinity of the old lead-mining and
lead-smelting industrial complex of Lavrion in Greece were examined
neuropsychologically. The mean PbB level from venous samples was
1.14 µmol/litre, range 0.355-3.067 µmol/litre (23.7 µg/dl, range
7.4-63.9 µg/dl). In order to control for confounding factors, several
regression models were evaluated. Starting with a set of 24 potential
confounders, an optimal model containing 17 potentially confounding
variables was finally developed for testing the PbB/IQ association.
The continuous PbB variable was divided into five equally wide classes
from equal/below 0.715 µmol/litre (14.9 µg/dl, low) to equal/above
2.16 µmol/litre (45.0 µg/dl). After correction for confounders, a
highly significant negative association was found between PbB level
and IQ (WISC-R). A persistent decrease in IQ was only observed at PbB
concentrations above 1.2 µmol/litre (25 µg/dl). The adjusted
full-scale IQ difference between "high" and "low" PbB children was 9.1
points. Highly significant negative associations with PbB were also
found for the Bender Gestalt test and the Vienna Reaction Device,
without any indication of a threshold.
i) Silva et al. (1988)
Five hundred and seventy-nine socioeconomically advantaged
11-year-old children with an average PbB level of 0.53 µmol/litre
(11 µg/dl), range 0.19-2.4 µmol/litre (4-50 µg/dl) were tested in
Dunedin, New Zealand. Because the crude inverse association between
blood lead and IQ was not statistically significant, subsequent
multivariate analyses were not carried out.
j) European multicentre study
The results from a multicentre study in Europe were reported by
Winneke et al. (1990). The study linked eight institutions from eight
European countries; four of the individual study groups were from
areas near smelters and the others were more general populations. A
common study protocol with inherent quality assurance elements was
developed to achieve comparability between the individual studies. In
all, 1879 children, aged 6-11 years, were studied; however, for some
of the tests the sample size was reduced to only 971 children. The PbB
level ranged from 0.24 to 2.88 µmol/litre (5 to 60 µg/dl). Overall
statistical evaluation was done using a uniform predetermined
confounder model containing age, gender, occupational status of the
father, and mother's education. The inverse association between PbB
level and IQ (four subtests from the WISC-R) was of only borderline
statistical significance. An IQ decrement of about 3 points was
calculated for a PbB increase from 0.24 to 0.96 µmol/litre (5 to
20 µg/dl). The associations between the error scores on the Bender
Gestalt Test and the Vienna Reaction Device were statistically
significant and more consistent across study groups, although the
outcome-variance explained that lead never exceeded 0.8%. The data did
not allow for the identification of a threshold.
8.3.4.3 Follow-up studies
a) Bellinger et al. (1984)
A follow-up of some of the children investigated by Needleman et
al. (1979) was conducted when the children were approximately eleven
years old. Twenty-two of the original elevated lead group and 48
children from the low lead group were traced, as well as a group of 52
children who had not been tested previously, but whose lead levels in
a "dentine chip" (from teeth analysed at the time of the previous
study) fell into the mid-range between the high and low lead groups.
Children in the elevated lead group were significantly younger than
those in the other two groups. Information on IQ from group-
administered Otis Lennon Mental Ability tests, conducted 1 to 2 years
previously, was collected from school records for 15, 52 and 34
children from the elevated, mid-range and low groups, respectively.
Scores were inversely related to previously obtained lead measures,
with a 7-point difference in mean scores between the high and low lead
groups (t-ratio = 1.65, P = 0.1). No current measures of lead status
were available.
b) Winneke et al. (1989)
Out of 114 children first tested at age 6 (Winneke et al., 1985)
76 were retested at age 9. The range of PbB levels had been
0.187-1.09 µmol/litre, geometric mean = 0.39 (3.9-22.8 µg/dl,
geometric mean = 8.2) at age six and was 0.21-1.027 µmol/litre,
geometric mean 0.37 (4.4-21.4 µg/dl, geometric mean 7.8) at age 9
(r = 0.82). After correction for the same set of confounders used at
age 6 (age, gender, social background), most of the findings that had
been found to be significant at age 6, i.e. those related to reaction
performance (Vienna Reaction Device), remained virtually unchanged
three years later. The authors concluded that this observation cannot
necessarily be taken to indicate persistence, because internal
exposure had essentially remained unchanged over three years.
c) Needleman et al. (1990)
A follow-up assessment was made of 132 young adults first
investigated by Needleman et al. (1979) at the age of 6-7. Those
retested were not representative of the group of 270 tested 11 years
earlier, as they had slightly lower childhood dentine lead levels,
came from higher SES families, and were more likely to be female.
Current PbB level was assessed from 48 subjects and none was above
0.336 µmol/litre (7 µg/dl). A behavioural evaluation including a word
reading test, and a neurobehavioural assessment was administered to
each subject, and a self-report of delinquency was obtained. In
addition school records were reviewed. Multiple regressions indicated
that higher levels of dentine lead in childhood was associated with
lower reading scores, lower class rank, increased absenteeism, and
poorer performance on some of the neurological tests in young
adulthood. Comparing subjects with dentine lead levels greater than
20 µg/g with the lowest lead group, children in the high lead group
were much more likely to drop out of school (unadjusted odds ratio
4.6; adjusted odds ratio 7.4, CI 1.4-40.8) and to have a reading
disability (unadjusted odds ratio 3.9; adjusted odds ratio 5.8, CI
1.7-19.7). Adjustment for covariates made little impact on most of the
outcomes.
8.3.4.4 Conclusions and limitations of cross-sectional studies
A number of general issues are evident from looking at the
cross-sectional studies as a group. In most studies a negative
association between lead measures and IQ measures is found in
uncontrolled data. This difference is usually in the range of 4 to 6
IQ points and most marked in verbal IQ. Most studies also confirmed
the positive association between lead measures and indicators of
social disadvantage, whether this was indicated by SES, maternal
education, or other more detailed indicators of non-optimal
child-rearing environments, such as marital quality or maternal
depression. When these social and other confounding factors are
controlled, the effect has, in most cases, been to reduce the strength
of the association between lead measures and IQ, although it remains
in the same direction. When maternal intelligence has not been
controlled, the impact on the association between lead and IQ measures
of correction for covariates tends to be smaller. Where more detailed
social measures have been controlled, the impact on the magnitude of
the lead-IQ association has been greater. Associations have, in
general, been stronger with verbal IQ than with performance IQ.
The cross-sectional nature of these studies, and also the fact
that many used a single measure of current exposure, limited their
usefulness in answering questions relating to the natural history of
the association between lead exposure and outcomes, including whether
there were critical periods of exposure, and whether lead associated
deficits were persistent or reversible.
Findings from follow-up studies are difficult to interpret.
Needleman et al. (1990) interpreted their findings as indicating the
persistence of the effects of lead. Identifying persistence or
irreversibility in this context has many problems, due to the
stability of a number of other (non-lead) factors. In addition,
sampling biases, or failure to control for important covariates in the
initial or follow-up stages may lead to apparent persistence of an
effect. A further difficulty is that it is unusual to find a cohort
where exposure profiles permit easy assessment of persistence.
Neurobehavioural effects detected at age seven or later usually
persist in later follow-up studies conducted in other (non-lead) areas
of research, and for this reason it is more likely than not that
lead-associated deficits detected during childhood will be detectable
later. However, there are not enough data to conclude that this is the
case. Animal data, in which persistence of effects has been shown
after cessation of exposure, do provide support for the
irreversibility of lead-induced behavioural toxicity.
8.3.5 Prospective epidemiological studies on children
8.3.5.1 Common elements
The international prospective cohort studies shared a common
design of pre- or perinatal recruitment, with the principal measure of
prenatal exposure being the lead concentration in whole blood taken
antenatally or at birth. To facilitate later comparison of results,
meetings have been held at which common protocols (within the
constraints of local conditions) were agreed (Bornschein & Rabinowitz,
1985; Smith, 1989).
The protocols were similar in terms of the instruments of
neurobehavioural assessments, which were well standardized and
validated for the populations to which they were applied, and although
the frequency of assessment varied between studies, it was agreed that
all studies would make an assessment during infancy, in the later
pre-school period and, if possible, in the school age years.
All studies conducted several assessments of PbB concentration
but at varying frequency, and all studies participated in some form of
quality assurance/quality control (QA/QC) programme for the assessment
of lead exposure. QA/QC procedures were also used to establish
inter-examiner reliability (where more than one examiner was used) for
psychometric and covariate assessments. The importance of such
procedures was recently discussed (SAHC, 1993).
In assessing dose-effect relationships between lead exposure and
outcome variables, most studies used several indices of exposure,
including both measures of PbB concentration at particular ages and
average measures of exposure within the lifetimes of the study
subjects.
All studies used data analysis procedures which took simultaneous
account of covariates selected for statistical and substantive
considerations.
8.3.5.2 Study descriptions
a) Boston study
The study population was recruited from 11 837 infants born at
the Brigham and Women's Lying-In hospital in Boston between April 1979
and April 1981 (Bellinger et al., 1984). On the basis of cord PbB
levels of approximately 2500 children, the 90th, 50th and 10th
percentiles were identified and used as eligibility criteria for
enroling 249 children in three exposure groups: low (< 0.14 µmol/litre
or 3 µg/dl, N = 85), medium (0.288-0.336 µmol/litre or 6-7 µg/dl,
N = 88) and high (> 0.48 µmol/litre or 10 µg/dl, N = 76). No child
had a cord PbB level greater than 1.2 µmol/litre (25 µg/dl).
Exclusion criteria included birth complications or medical
conditions associated with developmental difficulties (e.g.,
gestational age < 34 weeks, Down's syndrome), a non-English speaking
family or residence in an area considered unsafe for home visitors.
The cohort consisted largely of children from intact middle and
upper-middle class families. Approximately 85% of the children were
white.
Capillary blood collected at ages 6, 12, 18 and 24 months was
analysed for lead by ASV. Venous blood collected at ages 57 months and
at 10 years was analysed for lead by AAS. In contrast to other
prospective cohorts, the mean PbB level did not rise postnatally and
indeed never exceeded 0.384 µmol/litre (8 µg/dl).
The principal psychometric tests included the Bayley Scale of
Infant Development at 6, 12, 18 and 24 months, the McCarthy Scales of
Children's Abilities at 57 months and the Wechsler Intelligence Scale
for Children - Revised and the Kaufman Test of Educational Achievement
at age 10 years. After age 2 years, the mean level of performance was
above the expected population mean (e.g., mean full scale IQ at age 10
years = 119) (Bellinger et al., 1992).
A variety of potential confounding factors was assessed including
maternal IQ, the quality of rearing environment (HOME scores at 6, 24,
57 and 120 months) and prenatal, perinatal and postnatal medical and
socio-demographic factors.
b) Cincinnati study
Women attending prenatal clinics in predesignated leadhazardous
residential areas of Cincinnati, Ohio, USA, were consecutively
recruited from 1979 to 1984 (Dietrich et al., 1987) Prenatal
exclusions were for maternal prenatal alcohol and drug abuse,
psychosis, diabetes, mental retardation; neonatal exclusions included
birth weight under 1.5 kg and gestational age (by physical
examination) below 35 weeks. Furthermore, recruited infants had to
have an Apgar score of 6 or more at 5 min postpartum and to have no
serious medical conditions. Of the mothers, 87% were single and 86%
were receiving public assistance. The final follow-up sample of 305
infants who attended their second clinic visit at 3 months of age was
85% African-American and 50% female.
Mean prenatal maternal PbB levels as assessed by ASV were
0.38 ± 0.177 µmol/litre (8.0 ± 3.7 µg/dl). Mean neonatal PbB levels
were also low at 0.221 ± 0.134 µmol/litre (4.6 ± 2.8 µg/dl). Following
the development of prewalking progression and normal hand-to-mouth
behaviours, PbB levels began to rise, peaking at around 21 months with
a mean of approximately 0.89 µmol/litre (18 ± 6 µg/dl). Approximately
35% of the sample had at least one PbB concentration equal to or
greater than 1.2 µmol/litre (25 µg/dl) sometime during the first 5
years of life, while 79% exceeded 0.72 µmol/litre (15 µg/dl) during
the same period (Dietrich et al., 1991, 1992). Virtually all children
(95%) exceeded 0.48 µmol/litre (10 µg/dl) during the first 5 years of
life. Postnatal PbB concentrations were assessed on a quarterly basis
beginning at 10 days postpartum. The vast majority of blood samples
were collected by venepuncture.
Major neurobehavioural assessments included the Bayley Scales of
Infant Development administered at 3, 6, 12 and 24 months of age. The
Kaufman Assessment Battery for Children was administered at 4 and 5
years. A comprehensive examination of gross and fine neuromotor
functions (the Bruininks-Oseretsky Scales) was administered at 6
years. Finally, performance on the Wechsler Intelligence Scale for
Children - Revised was assessed following school entry at 6.5 years.
Intellectually, this was a low functioning cohort with a mean
full-scale IQ at 6.5 years of 86.9 ± 11.3 points (Dietrich et al.,
1993a).
Covariates measured included assessments of obstetrical and
perinatal complications, birth anthropometrics, tobacco and alcohol
consumption, observational assessments of the quality of care-taking
in the home at 6, 12, 24 and 36 months, social class, iron status and
various aspects of child health likely to affect neurobehavioural
performance (e.g., otitis media, sensory deficits, allergies, seizure
disorders). Maternal IQ was assessed postnatally. All medical and
psychometric testing was conducted at a single welfare clinic located
in the heart of the recruitment area.
c) Cleveland
Five hundred and forty-three infants delivered to women at a
large inner city general hospital between February 1981 and March 1982
were considered for inclusion in this study of the joint or
independent effects of fetal alcohol exposure and lead on child
development (Ernhart et al., 1985). Due to pre-term birth or illness,
16% were excluded from further study. Also excluded were mothers
reporting the use of narcotics and those with identifiable
psychological disorders. The sample was predominantly white (65%) and
of lower SES. Approximately 50% of mothers were alcoholics.
PbB concentrations were assessed by AAS (208 mothers at delivery
and in 178 cords resulting in 142 mother-infant pairs with complete
prenatal lead exposure data). Postnatally, blood samples were analysed
for approximately half the birth cohort at ages 6 months, two years
and three years of age. At least one perinatal or postnatal PbB
concentration was available for each of 285 children with
developmental data past the neonatal period (Ernhart et al., 1987,
1989a,b).
Mean maternal PbB concentrations (N = 185) were low
(0.31 ± 0.086 µmol/litre or 6.5 ± 1.8 µg/dl) and mean cord PbB
concentrations (N = 162) were also low (0.29 ± 0.10 µmol/litre or
6.0 ± 2.1 µg/dl). Postnatally, mean PbB concentrations (N = 151) were
0.48 ± 0.16 µmol/litre (10.0 ± 3.3 µg/dl) at 6 months,
0.80 ± 0.28 µmol/litre (16.7 ± 5.9 µg/dl) at 2 years (N = 165) and
0.80 ± 5.9 µmol/litre (16.7 ± 5.9 µg/dl) at 3 years. The lowest and
highest PbB levels recorded postnatally were 0.24 and 2.016 µmol/litre
(5 and 42 µg/dl), respectively.
Neurobehavioural assessment included selected sub-scales of the
Brazelton Neonatal Behavioural Assessment Scales and Graham Rosenblith
Behavioural Examination of the Neonate at > 24 h postpartum, the Kent
Infant Development scale at 6 months, the Bayley Scales of Infant
Development at 6, 12 and 24 months, the Stanford-Binet Intelligence
Scale at 3 years, and finally the Wechsler Preschool and Primary Scale
of Intelligence at approximately 5 years of age. All assessments
beyond the neonatal period were conducted in subjects' homes.
Intellectual attainment in this high risk, socioeconomically
disadvantaged cohort was typically low as shown by a mean full-scale
IQ at approximately 5 years of 87.5 ± 16.6 points (Ernhart et al.,
1989a).
In addition to the usual assessments of obstetrical/perinatal
complications and neonatal status, other covariates measured included
the Michigan Alcoholism Screening Test, the Peabody Picture Vocabulary
Test as an assessment of maternal intelligence, and the Authoritarian
Family Ideology Scale. Women were also questioned as to their
consumption of alcohol and tobacco during pregnancy. Observational
assessments of care-taking quality in the home were conducted at 1, 2,
3 and 4 years.
d) Glasgow study
The study group for CNS outcomes consisted of 151 subjects drawn
from an initial sample of 885 families exposed to various levels of
dietary lead, principally due to a plumbosolvent water supply (Moore
et al., 1989). All subjects were born in the United Kingdom and spoke
English as a first language. The sample was divided into three groups
of approximately equal numbers (matched for social class) based on
maternal prenatal PbB levels: high (> 1.44 µmol/litre or 30 µg/dl),
medium (0.72-1.2 µmol/litre or 15-25 µg/dl) and low
(< 0.48 µmol/litre or 10 µg/dl). The SES of the sample ranged from
the chronically unemployed to the professional classes.
PbB levels were assessed postnatally at 1 and 2 years of age and
found to be 0.734 and 0.777 µmol/litre (15.3 and 16.2 µg/dl),
respectively.
Measurements of neurobehavioural outcome where the Bayley Scales
of Infant Development were used were administered at 1 and 2 years of
age.
Covariates considered in the data analysis included a measure of
obstetrical complications, birth weight, birth order, SES of the
father in postnatal follow-up years 1 and 2, and observational
assessments of care-taking quality in the first and second years of
follow-up.
e) Kosovo Study
Five groups of infants from two communities in Kosovo,
Yugoslavia, were studied (Graziano et al., 1990; Wasserman et al.,
1992). Mitrovica was the site of a lead smelter, refinery and battery
plant, and Pristina an area of minimal lead exposure. Three groups
were recruited from Mitrovica on the basis of cord PbB concentration:
(1) < 0.72 µmol/litre (< 15 µg/dl, N = 78); (2) 0.72-0.96 µmol/litre
(15-20 µg/dl, N = 99); (3) > 0.96 µmol per litre (> 20 µg/dl,
N = 217). Two groups were recruited from Pristina: (4) a group with
cord PbB levels < 0.72 µmol/litre (< 15 µg/dl) and (5) a group
matched to children in group (3) in terms of the distribution of
maternal and paternal education. Grouping was not considered in the
statistical analysis. The total cohort (N = 541) was largely Albanian
and Serbian. Infants with CNS defects, chromosomal anomalies, distant
residence and twin pairs were excluded.
PbB level and various indices of iron status (EP, Hb, serum
ferritin) were measured in venous blood collected in midpregnancy,
delivery and at postnatal ages 6, 12, 18 and 24 months. Mean PbB level
amongst children in Mitrovica exceeded 0.96 µmol/litre (20 µg/dl) at
all ages, reaching 1.68 µmol/litre (35 µg/dl) at 24 months. Amongst
children in Pristina, the mean PbB level was below 0.48 µmol/litre
(10 µg/dl) at all ages.
The Mental Scale of the Bayley Scales of Infant Development was
administered at ages 6, 12, 18 and 24 months, and the Motor Scale at 6
and 12 months. At 24 months, the mean Mental Development Index in the
cohort (n = 392) was 105.2 (SD = 18.1).
Anthropometric measurements and assessments of child and family
health, diet, and demographic status were carried out at each age. An
adaptation of the HOME Scale was administered at ages 3 and 4 years
and maternal IQ was assessed using the Raven's Standard Progressive
Matrices.
f) Port Pirie study
Port Pirie (population 16 000 in 1979) is an industrial town 200
km north-west of Adelaide, South Australia, with a large and
long-standing smelting facility (Wigg et al., 1988). Over the period
1979-1982, a cohort of 723 children from Port Pirie and surrounding
rural areas was recruited into the study. The cohort represented a 90%
sample of all children born in the area during this period.
Lifetime lead exposures were estimated from venous PbB
concentrations obtained antenatally (at 14-20 weeks of gestation, and
32 weeks), at delivery (maternal and cord blood), and from capillary
samples taken postnatally at ages 6, 15, and 24 months, and annually
thereafter. Lead concentrations in all samples were determined by AAS.
At the time that each blood sample was collected, the nurse
interviewer also conducted structured interviews to obtain information
on a range of demographic, familial, behavioural, medical and social
environmental factors. HOME scores were assessed when the child was 3
and 5 years of age. Maternal IQ was also assessed. The Bayley Scales
of Infant Development was administered at 2 years, the McCarthy Scales
of Children's Abilities at 4 years and the Wechsler Intelligence Scale
for Children - Revised version (WISC-R) at 7 years.
g) Sydney study
A total of 318 children was recruited from among infants born at
three maternal hospitals between April 1982 and March 1983 (Cooney et
al., 1989a). Exclusion criteria included prematurity, low birth
weight, severe medical problem, single or non-English speaking mother
or maternal drug or alcohol problems. All children were white and
generally represent a middle-class population.
PbB concentration was measured in maternal venous blood during
hospitalisation (geometric mean = 0.388 µmol/litre or 8.1 µg/dl,
geometric SD = 0.067 µmol/litre or 1.4 µg/dl). Additional blood
samples were collected at 6-month intervals to age 4 years and at ages
5 and 7 years. Up to 24 months of age, approximately 50% of the blood
samples were capillary, while almost all samples collected
subsequently were venous. Geometric mean PbB levels between 6 months
and 4 years were in the range of 0.48-0.72 µmol/litre (10-15 µg/dl),
falling to 0.39 and 0.37 µmol/litre (8.3 and 7.7 µg/dl) at ages 5 and
7, respectively.
The Bayley Scales of Infant Development were administered at ages
6, 12 and 24 months, the McCarthy Scales of Children's Abilities at
ages 3 (n = 215) and 5 (n = 200), and the Wechsler Intelligence Scale
for Children - Revised at 7 years (n = 175). All assessments, at least
until age 5, were conducted in the child's home. The mean Mental
Development Index and General Cognitive Index (GCI) scores were in the
intellectual normal range of 107 to 117 with a mean general cognitive
index at 4 years of 107.3 ± 14.2.
Covariates measured included maternal IQ, quality of care-taking
environment, obstetrical and postnatal factors.
8.3.5.3 Summary of differences between studies
The prospective studies differed to such an extent that it would
be surprising if they yielded identical findings. This does not mean
that the studies cannot be compared, merely that these differences and
their potential impact on study findings must be acknowledged in any
assessment of the studies as a group (Bellinger & Stiles, 1993). There
are substantial differences in the degree of confounding between lead
exposure and the other correlates of poor developmental outcomes. For
example, mean maternal IQ ranges from 121 (Boston) to 74-75
(Cleveland, Cincinnati), reflecting vast cohort differences in
socioeconomic standing and child-rearing context.
The studies also varied substantively in terms of the lead
exposure profiles observed. Site differences resulted in considerable
variability in postnatal lead exposures depending on exposure patterns
(residence near primary lead smelters, residence in deteriorated
housing, proximity to traffic, and quality of domestic
drinking-water).
Despite broad similarities in the approaches to data analysis,
there were substantial differences in the form in which the findings
were reported, impeding direct comparison of study results. Results
were diversely reported in terms of partial correlation coefficients
and standardized and unstandardized regression coefficients, and there
was variable use of raw (untransformed) and log exposure measures.
Most, but not all studies presented their results in a manner
conducive to some kind of quantification of dose-effects
relationships.
Clearly the variation in numbers of participants available to
individual studies (both initially, and as a consequence of attrition)
has a direct effect on the statistical power of each study - which is
also affected by the degree of confounding.
8.3.5.4 Results of studies
a) Infancy and early preschool assessments
The pattern of findings in infant and early preschool assessments
for which the principal instrument of assessment was the Bayley Scales
of Infant Development is inconsistent. Using different indices of
in utero exposure (e.g., prenatal, at delivery, cord or neonatal PbB
levels), three of the prospective studies found associations with
slower sensorimotor development up to 6 months or 1 year of age
(Ernhart et al., 1987; Dietrich et al., 1987, 1989) or up to 2 years
of age (Bellinger et al., 1987), but these tended to attenuate over
time (Dietrich et al., 1990; Bellinger et al., 1991a). Other studies
did not observe statistically significant adjusted relationships of
prenatal exposure with slower sensorimotor development (Moore et al.,
1989; Wigg et al., 1988; Cooney et al., 1989a,b; Wasserman et al.,
1992).
Two studies observed an inverse association between postnatal
lead exposure on mental development in later infancy (Wigg et al.,
1988; Wasserman et al., 1992), although in one study it was PbB level
at 6 months of age (Wigg et al., 1988) and in the other study it was
the level at 24 months (Wasserman et al., 1992) that was most
predictive.
b) Later preschool and school age assessment
Despite some inconsistency in the pattern of findings in infants,
there has been a convergence of positive findings on later
neurobehavioural outcomes in the prospective studies. This may
reflect:
1) the greater reliability and precision of measurement attained
with assessments of the older child; or
2) an effect of lead on abilities that cannot easily be tested
during infancy (e.g., executive, regulative and organizational
skills, higher order reasoning).
c) Boston study
Among 169 children assessed at age 57 months, the major finding
was an association between PbB level at 2 years of age and the GCI of
the McCarthy Scales. There was a decrease of 2.95 ± 1.42 GCI units for
an increase in PbB of 0.48 µmol/litre (10 µg/dl). Since the group had
an average PbB level of 0.336 µmol/litre (7.0 µg/dl) over 2 years,
this decrement was relevant to a range of approximately
0.19-0.67 µmol/litre (4-14 µg/dl) Bellinger et al., 1991a).
At 10 years of age, 148 children were reassessed. PbB
concentration at 2 years of age was inversely associated with the
WISC-R Full Scale IQ. Each 0.48 µmol/litre (10 µg/dl) increase was
associated with a 5.8 point decline in IQ (95% CI 1.7 to 9.9)
(Bellinger et al., 1992).
PbB concentration at 24 months was also inversely related to the
Battery composite score in the Kaufman Test of Educational Achievement
(Brief form) (-0.89 ± 0.24). The skills assessed were mathematics,
reading and spelling.
The point estimates associated with other postnatal ages were
generally in the same direction but only PbB level at 2 years revealed
conventional statistical significance. Other specific
neuropsychological tests did not reveal a clear pattern of
neuropsychological deficit (Stiles & Bellinger, 1993).
d) Cincinnati study
The Kaufman Assessment Battery for Children was administered to
approximately 260 children at 4 and 5 years of age (Dietrich et al.,
1991, 1992). The principal findings at 4 years were that higher
neonatal PbB levels were associated with poorer performance on all
Kaufman subscales. However, this inverse association was limited to
children from the poorer families. Following full covariate
adjustment, few statistically significant associations between
postnatal PbB levels and Kaufman scales could be found. However, the
results did suggest a weak relationship between postnatal PbB levels
and performance on a Kaufman subscale which assesses visual spatial
and visual-motor integration (adjusted regression coefficient -0.12 SE
not published).
At 5 years of age, postnatal PbB levels were associated with
poorer performance an all subscales of the Kaufman Battery. However,
after adjustment for covariates, few statistically significant
relations remained. Nevertheless, as at 4 years of age, the subscale
assessing visual-spatial and visual-motor skills was most sensitive,
with average lead exposure during the 4th year of life being
significantly associated with performance (-0.12 units per
0.048 µmol/litre or per µg/dl, SE not published).
At the age of 6.5 years, 253 children in the Cincinnati cohort
were administered the WISC-R (Dietrich et al., 1993a). The major
findings were that postnatal PbB concentrations were inversely
associated with full-scale IQ and performance IQ. Following
statistical adjustment for covariates, including maternal IQ and
quality of home care, a statistically significant relationship was
retained between postnatal PbB concentrations at nearly all ages (and
including lifetime averages) and Performance IQ. Further analysis
revealed that average lifetime PbB concentrations in excess of
0.96 µmol/litre (20 µg/dl) were associated with deficits in IQ of the
order of 7 points, when compared with children with mean
concentrations of less than 0.48 mol/litre (10 µg/dl). The regression
coefficient for Performance IQ on mean lifetime PbB level was -0.26
(± 0.12) IQ units per 0.048 µmol/litre or per µg/dl. This corresponded
to a 2.6 unit decrement associated with a 0.48 µmol/litre (10 µg/dl)
movement in lifetime average PbB level, for which the average
(interpolated graphically) is close to 0.67 µmol/litre (14 µg/dl).
At 6 years of age, the Bruininks-Oseretsky Test of Motor
Proficiency (BOTMP) was administered to 245 children in the Cincinnati
Cohort (Dietrich et al., 1993a). Following statistical adjustment for
covariates, neonatal PbB levels were associated with poorer
performance on a measure of upper-limb speed and dexterity and the
fine-motor composite. Postnatal PbB levels also remained significantly
associated with poorer scores on measures of bilateral coordination,
visual-motor control, upper-limb speed and dexterity, and the fine
motor composite.
Children having an average mean lifetime PbB level of
approximately equal to or exceeding 0.43 µmol/litre (9 µg/dl) appeared
to experience a deficit in both fine and gross motor skills relative
to children in the lowest PbB quartile. Children in the highest
average lifetime PbB quartile had scores on the gross-motor subtest
assessing bilateral coordination of approximately 0.5 standard
deviations (2.5 points) lower than their counterparts in the lowest
quartile. Children in the highest average lifetime quartile also
scored more poorly in the fine-motor functioning, having scores of
approximately 0.6 standard deviations lower (6.3 points) than those in
the lowest quartile.
e) Cleveland study
The WPPSI was administered to 242 children at the age of 4 years
and 10 months (Ernhart et al., 1989a). Statistically significant
correlations between IQ (full scale and subscales) and PbB level
measured pre- and perinatally and at ages 2 and 3 years (and a
lifetime postnatal average) became non-significant after adjustment
for covariates. No estimates for effect size were presented in the
original report (Ernhart et al., 1989a).
f) Glasgow study
No neurobehavioural assessments were carried out beyond 2 years
of age (Moore et al., 1989).
g) Kosovo study
No results beyond 2 years of age were reported (Graziano et al.,
1990; Wasserman et al., 1992).
h) Port Pirie cohort study
The principal finding at 4 years was an inverse association of
McCarthy Scales of Children Abilities (MSCA) scores (General Cognitive
Index (GCI), Perceptual Performance and Memory) with most indices of
lead exposure (McMichael et al., 1988). After adjustment for
covariates, the association of GCI was not signifi-cant at certain
specific ages. However, it remained significant for the integrated
postnatal average for which an effect size of 7.2 GCI points lost in
association with an increase of lifetime average PbB level from 0.5 to
1.5 µmol/litre (10.4-31.2 µg/dl) was estimated.
Between 7 and 8 years of age the IQ of 494 children was assessed
with the WISC-R (Baghurst et al., 1992). After adjustment for
covariates there was little association with pre- and perinatal lead
exposure assessments but significant decrements in full-scale IQ of
between 3.7 and 4.8 points (depending on age) for each (natural) log
unit increase in lifetime average PbB concentration were observed. The
estimated effect size for the lifetime average exposure up to 3 years
of age was a loss of 5.3 IQ points in association with an increase in
PbB level from 0.48 to 1.44 µmol/litre (10 to 30 µg/dl).
The Block Design subscale of the WISC-R, which tests spatial
abilities, exhibited the strongest association with lead exposure and
estimated effect sizes were stronger in girls than boys for both the
MSCA and WISC-R.
i) Sydney study
At the age of 4 years, 207 children were assessed with the
McCarthy Scales of Children Abilities (MSCA) and virtually no
significant associations with any measures of PbB were observed
(Cooney et al., 1989b).
Follow-up assessments in 175 children using WISC-R at age 7 also
yielded no associations (crude or adjusted) with blood lead history
(Cooney et al., 1991).
8.3.5.5 Questions prospective studies have not answered
A disappointment of the prospective studies was their inability
to reach any obvious consensus on the behavioural phenotype associated
with low-level lead exposure, or on age(s) of critical sensitivity.
This latter fact may reflect the phenomenon of intra-individual
"tracking", whereby an individual maintained approximately consistent
ranking with respect to his or her PbB concentration at any age, or it
may reflect the need for chronic exposure over extended periods in
order for causal effects to become apparent.
8.3.5.6 Attempting a consensus
As discussed previously, no clear delineation of an age (or age
range) of maximal sensitivity to lead exposure has emerged from the
prospective studies, and there is a diversity of reporting styles
which makes attempts to compare studies and establish a consensus of
opinion very difficult.
Nevertheless, it would appear essential to attempt some sort of
synthesis of studies performed so far, given that the social and
economic consequences of preventing an adverse health effect are
potentially enormous.
A meta-analysis by Needleman & Gatsonis (1990) concluded that the
overall pattern of results was most unlikely to have occurred by
chance. As an approach to the problem of obtaining the "best" estimate
of the effect of lead exposure on neuro-behavioural development, a
common estimate of the partial correlation between lead burden and IQ
was determined. However, in order to derive such an estimate with some
degree of certainty, it is essential to find a common outcome, and to
identify studies for which at least broadly comparable exposure
measures are available.
Schwartz (1993) has identified three prospective studies and five
cross-sectional studies that examined the association of lead exposure
as determined by PbB concentrations with full scale IQ. For the
prospective studies, the exposure measures used were 24-months PbB
level (Boston) and lifetime average up to 3 years of age (Cincinnati
and Port Pirie).
There was only one measure for each of the cross-sectional
studies. Despite the compromises necessary for such a comparison, the
estimated effect sizes for an increase in PbB level from 0.48 to
0.96 µmol/litre (10 to 20 µg/dl) for all seven studies lay between 1
and 6 IQ points lost (with five of the studies lying between 1 and 4),
and the associated 95% confidence limits only just embraced the value
zero for 3 of the 7 studies.
Thus, a broad consensus does emerge from a rough comparison. From
the estimates summarized by Schwartz (1993), a weighted mean for the
six coefficients accompanied by a standard error of 0.4 was estimated
to be 2.6 IQ units lost for an increase in PbB from 0.48 to
0.96 µmol/litre (10 to 20 µg/dl).
8.3.6 Task group overview and interpretation of prospective studies
on children
8.3.6.1 Rationale
As a complement to the narrative review, prepared by the Task
Group, a quantitative overview of the findings of the prospective
studies was considered to be a valuable means of assessing the overall
strength of evidence for an association between blood lead summary
measures and school age IQ.
While individual studies vary somewhat in their design and their
analysis strategies, their underlying objectives are closely related.
For any generally applicable conclusions on general population lead
exposure and its effect on intellectual attainment to be reached, some
consistency of evidence across studies is highly desirable.
Since any single study has considerable random error in
estimating a relationship, a quantitative overview (or meta-analysis)
can be a valuable means of defining what is the plausible magnitude of
statistical association between PbB measures and child IQ.
It seems appropriate, given the quite different types of study
design, to present two separate meta-analyses dealing with prospective
studies and cross-sectional studies, respectively.
Any such meta-analysis needs cautious interpretation. The caveats
in drawing causal inferences from statistical associations apply to
any meta-analysis just as they do to individual studies. Any increase
in statistical precision regarding the magnitude of association does
not eliminate the potential for bias in any observational study on
human populations.
8.3.6.2 The prospective studies
There are four prospective studies: Boston (Bellinger et al.,
1992); Sydney (Cooney et al., 1989a,b); Cincinnati (Dietrich et al.,
1993a); and Port Pirie (Baghurst et al., 1992) which presented
quantitative results in a form suitable for an overview (i.e. with
regression coefficients and standard errors). The Task Group
concentrated on school age assessment because:
a) the studies used the same outcome measure (WISC-R), which is
widely accepted and has been previously evaluated in cross-
sectional studies; and
b) intellectual assessment at primary school age (6-10 years) is,
perhaps, of greatest overall relevance in reaching a consensus on
the public health importance of childhood lead exposure.
Unfortunately, the Cleveland study did not publish findings in
the same quantifiable manner. The potential impact of this apparently
"negative" study on the overall evidence will be discussed in section
8.3.6.2(c).
a) Measures of exposure, outcome and association
All four studies have used blood lead as the measure of body lead
burden, but the timing and frequency of examinations has varied as has
the choice of summary measures (averages over time).
In a meta-analysis it seems appropriate to concentrate on
measures of association after adjustment for confounders. While there
is some variation in choice of confounders, the two key covariates
(mother's IQ and HOME scores) appear in all four studies.
The Port Pirie study used the logarithm of the PbB concentration
in the multiple regression analysis while the other three studies used
untransformed PbB concentration. This difference in statistical style
can be reconciled by converting the lead regression coefficients to
estimated changes in IQ for a specific increase in PbB concentration,
i.e. from 0.48 to 0.96 µmol/litre (10 to 20 µg/dl). This equals 10
times the regression coefficient (as published) for the Boston and
Cincinnati studies and log 2 times the regression coefficient for the
Port Pirie study.
It is recognized that the specific subscales of performance and
verbal IQ are of separate interest (e.g., the performance IQ has been
rather consistently shown to be more reliably associated with
postnatal lead exposure amongst the prospective studies (McMichael et
al., 1988; Bellinger et al., 1991a; Dietrich et al., 1991, 1992,
1993b). However, it appears more important to focus on full-scale IQ
as the primary outcome measure, otherwise concerns about post-hoc
selection may be raised. Such full-scale IQ was measured at age 6.5, 7
and 10 years in the Cincinnati, Port Pirie, Sydney and Boston studies,
respectively.
b) Display of individual study findings
Since most studies have not shown perinatal PbB level to be
predictive of intellectual performance, attention will be focused on
the various measures of postnatal PbB level. In fact, there are seven
such measures in each study that have been related to full-scale IQ in
the reports. Tables 21 and 22 summarize the magnitude of association
together with the standard error for all reported analyses, each after
adjustment for confounders.
Except for the earliest postnatal PbB values in the Boston and
Cincinnati studies, the adjusted associations between PbB level and IQ
are consistently negative (i.e. inverted). However, the "random noise"
in each study's analysis means that there is considerable variation in
levels of statistical significance. Hence, there is a need for a more
systematic overview combining the evidence from all studies.
c) A quantitative overview
The prime difficulty here is the different choices of PbB
summaries in the four studies. In undertaking a meta-analysis it would
have been better if all studies had essentially the same PbB measures
in their multiple regressions. Possible choices might have been (i) a
specific time point, e.g., 2 years or (ii) the means over a specific
interval, e.g., 0 to 5 years.
However, from the available analyses, some broadly similar
summaries can be chosen, as follows:
Table 21. Magnitude of association between blood lead and full-scale IQ
from four prospective studies
No. of Age at which blood lead Estimated change in
children was measured full-scale IQ a
Boston 148 year 1 0.0 (1.6)
year 1´ -1.2 (1.8)
year 2 -5.8 (2.1)
year 5 -2.6 (2.9)
year 10 -4.6 (5.2)
mean of years 2-5 -8.2 (2.8)
mean of years 2-10 -8.6 (3.4)
mean of 6 months to
10 years (unpublished) -5.7 (3.2)
Cincinnati 251 mean of year 1 0.1 (1.4)
mean of year 2 -0.2 (0.8)
mean of year 3 -1.3 (0.9)
mean of year 4 -1.5 (1.0)
mean of year 5 -2.3 (1.1)
mean of year 6 -3.3 (1.4)
mean of years 1-6 -1.3 (1.1)
Port Pirie 490 mean of first 15 months -2.8 (1.4)
mean of years 1-2 -3.2 (1.5)
mean of years 1-3 -3.3 (1.6)
mean of years 1-4 -3.2 (1.7)
mean of years 1-5 -3.0 (1.7)
mean of years 1-6 -2.8 (1.7)
mean of years 1-7 -2.6 (1.7)
Sydney 175 mean of years 1 and 2 -2.7 (2.8)
mean of years 3, 4 and 5 -1.9 (1.9)
year 7 -1.4 (1.7)
mean of years 1-7 -1.6 (2.2)
a From 0.48 to 0.96 µmol/litre (10 to 20 µg/dl) blood lead
(and its standard error) after adjustment for confounders
Table 22. Association between blood lead levels and full-scale
IQ from ten cross-sectional studies
No. of Estimated change in
subjects full-scale IQa
Lavrion, Greece 509 -2.7 (0.7)
Edinburgh, Scotland 501 -2.6 (0.9)
Greenwich, England 129 -5.6 (3.2)
European, multi-centre
Bucharest, Romania 301 -0.4 (2.6)
Budapest, Hungary 254 +0.8 (1.8)
Modena, Italy 216 +0.9 (4.2)
Sofia, Bulgaria 142 +2.2 (2.3)
Dusseldorf 1, Germany 109 -4.6 (4.5)
Dusseldorf 2, Germany 109 -3.9 (5.1)
Zagreb, Croatia 48 -1.5 (4.5)
a From 0.48 to 0.96 µmol/litre (10 to 20 µg/dl) blood lead
(and its standard error) after adjustment for confounders
i) There is some advantage in considering the mean PbB level over a
number of years, since it summarizes cumulative exposure and also
achieves a more reliable ranking of individuals than a single value.
The available long-term means are:
Boston: mean of 6 months to 10 years (D. Bellinger,
personal communication to the IPCS, 1993)
Cincinnati: mean of years 1 to 6
Port Pirie: mean of years 1 to 7
Sydney: mean of years 1 to 7
Although they differ in frequency and age range, these three
summaries should be very highly correlated with one another. The
resultant meta-analysis is displayed in Fig. 15. Each of the
individual study confidence intervals includes zero, indicating that
significance at P < 0.05 was not reached. However, the combined
evidence, weighting studies according to the inverse of their
variance, produces a weighted mean decrease in full-scale IQ of 2
points for a 0.48-µmol/litre (10-µg/dl) increase in PbB level, with
95% confidence interval from -0.3 points to -3.6 points (P = 0.01).
ii) An alternative approach is to consider PbB level at a specific
time or average over shorter intervals of time. For this approach, the
analyses of these studies are most comparable during the first three
years, as follows:
Boston: 2 year measure
Cincinnati: mean of year 3
Port Pirie: mean of years 1 to 3
Sydney: mean of years 1 and 2
This second meta-analysis is displayed in Fig. 16. The data here
support an inverse association more strongly than in Fig. 15, and the
combined evidence estimates a mean decrease of 2.6 IQ points for a
0.48-µmol/litre (10-µg/dl) increase in PbB level, with 95% confidence
interval from -1.2 points to -4.0 points (P < 0.001).
However, it should be noted that the estimates here are heavily
dependent on the choice of time points for PbB level. For instance, if
one instead chose:
Boston: 1.5 year measure
Cincinnati: mean of year 2
Port Pirie: mean of years 1 and 2
Sydney: mean of years 1 and 2
then the combined estimate is roughly halved in magnitude and is of
borderline significance.
One needs to recall that the other prospective study, carried out
in Cleveland (Ernhart et al., 1989a), has no equivalent data, but
reported no significant association. This study comprised about 150
evaluated children, and would influence the overall evidence in a less
significant direction if the data were able to be included.
8.3.6.3 A quantitative assessment of the cross-sectional studies
There have been more cross-sectional studies that have related
body burden to full-scale IQ in school-age children, but they cannot
all be included in a single meta-analysis. Here attention is focused
on the PbB studies, and it would be appropriate subsequently to

 undertake an equivalent meta-analysis of the tooth lead studies. The
studies included have differed in the number and nature of covariates.
In particular, in most of the groups within the London and European
multi-centre study (MCS), maternal IQ has not been controlled. Studies
in which regression coefficients could not be obtained were excluded.
Since the studies not included in the analysis account for only a
small fraction of the total children investigated, their exclusion has
a negligible effect on the meta-analysis estimates of effect size.
The following studies are included in the meta-analysis:
Lavrion, Greece (Hatzakis et al., 1989)
Edinburgh (Fulton et al., 1987)
London (Greenwich) (Yule et al., 1981)
European multi centre study (Winneke et al., 1990)
The European multi-centre study comprises seven separate analyses
for children in Bucharest, Budapest, Modena, Sofia, Dusseldorf (two
studies) and Zagreb. The Lavrion centre in this study was separately
reported by Hatzakis et al. (1987).
As in the meta-analysis for the prospective studies, individual
cross-sectional studies varied as to whether the logarithm of the PbB
level or untransformed PbB level was used in the multiple regression
analysis. Hence, as before, all associations are expressed in terms of
the estimated changes in full-scale IQ for a change in PbB level from
0.48 to 0.96 µmol/litre (10 to 20 µg/dl), after adjustment for
confounders, as shown in Table 13.
Fig. 17 shows these estimates and their 95% confidence limits for
the 10 study samples. Only the two largest studies (Lavrion and
Edinburgh) show statistically significant inverse associations, as
indicated by confidence intervals entirely to the left of zero. The
limited statistical power of the other studies is reflected in their
wide confidence intervals. Despite the considerable variation in the
study designs (e.g., sample selection, choice of confounders), there
is no evidence of statistical heterogeneity. In other words, all the
confidence intervals overlap and the heterogeneity test is not
statistically significant.
A combination of the evidence from all of these cross-sectional
studies produces a more precise overall estimate of association, as
shown in Fig. 17. This meta-analysis of the cross-sectional studies
estimates that full-scale IQ is reduced by 2.15 points for an increase
in PbB level from 0.48 to 0.96 µmol/litre (10 to 20 µg/dl), with a 95%
confidence interval from -1.2 points to -3.1 points (P < 0.001).
undertake an equivalent meta-analysis of the tooth lead studies. The
studies included have differed in the number and nature of covariates.
In particular, in most of the groups within the London and European
multi-centre study (MCS), maternal IQ has not been controlled. Studies
in which regression coefficients could not be obtained were excluded.
Since the studies not included in the analysis account for only a
small fraction of the total children investigated, their exclusion has
a negligible effect on the meta-analysis estimates of effect size.
The following studies are included in the meta-analysis:
Lavrion, Greece (Hatzakis et al., 1989)
Edinburgh (Fulton et al., 1987)
London (Greenwich) (Yule et al., 1981)
European multi centre study (Winneke et al., 1990)
The European multi-centre study comprises seven separate analyses
for children in Bucharest, Budapest, Modena, Sofia, Dusseldorf (two
studies) and Zagreb. The Lavrion centre in this study was separately
reported by Hatzakis et al. (1987).
As in the meta-analysis for the prospective studies, individual
cross-sectional studies varied as to whether the logarithm of the PbB
level or untransformed PbB level was used in the multiple regression
analysis. Hence, as before, all associations are expressed in terms of
the estimated changes in full-scale IQ for a change in PbB level from
0.48 to 0.96 µmol/litre (10 to 20 µg/dl), after adjustment for
confounders, as shown in Table 13.
Fig. 17 shows these estimates and their 95% confidence limits for
the 10 study samples. Only the two largest studies (Lavrion and
Edinburgh) show statistically significant inverse associations, as
indicated by confidence intervals entirely to the left of zero. The
limited statistical power of the other studies is reflected in their
wide confidence intervals. Despite the considerable variation in the
study designs (e.g., sample selection, choice of confounders), there
is no evidence of statistical heterogeneity. In other words, all the
confidence intervals overlap and the heterogeneity test is not
statistically significant.
A combination of the evidence from all of these cross-sectional
studies produces a more precise overall estimate of association, as
shown in Fig. 17. This meta-analysis of the cross-sectional studies
estimates that full-scale IQ is reduced by 2.15 points for an increase
in PbB level from 0.48 to 0.96 µmol/litre (10 to 20 µg/dl), with a 95%
confidence interval from -1.2 points to -3.1 points (P < 0.001).
 8.3.6.4 Task group overview of cross-sectional studies
a) Methods of controlling for confounders
A particular methodological concern in the cross-sectional
studies is the manner in which they have taken account of confounding
factors in deriving adjusted estimates of associations between body
lead burden and neuropsychological performance, in particular IQ.
The three aspects of the problem are:
i) Which potential confounders are measured? There is considerable
variation between studies here, suggesting that no individual study
can have fully corrected for the full range of parental and social
influences on IQ.
ii) How were confounders selected for inclusion in the analysis? Some
studies include confounders solely on the basis of their strength of
association with outcome (IQ), while others have given attention to
their association with body lead burden. It is difficult to assess the
extent to which these differences would affect the results, but the
former is more generally accepted statistical practice.
iii) Which statistical analysis strategy was employed in reaching a
final model? Most studies have adopted some form of multiple
regression technique, but have varied as to whether forward selection
(i.e. model building), backwards selection (i.e. model collapsing) or
more arbitrary choices of final model were employed. In the larger
studies, the choice between such strategies is unlikely to matter, but
in smaller studies the greater play of random variation could make the
results dependent on the statistical technique.
b) What they told us
In most studies a negative association between lead measures and
IQ measures is found in uncontrolled data. This difference is usually
in the range of 4 to 6 IQ points. Most studies also confirmed the
positive association between lead measures and indicators of social
disadvantage, whether this is indicated by SES, maternal education or
other more detailed indicators of non-optimal child-rearing
environments, such as marital quality or maternal depression. When
these social and other confounding factors are controlled, the effect
has been, in most cases, to reduce the strength of the association
between lead measures and IQ, although it remains in the same
direction. Where maternal intelligence has not been controlled, the
impact of correction for covariates tends to be smaller, and where
more detailed social measures have been controlled, the impact has
been greater.
c) What they did not tell us
The cross-sectional nature of these studies, and the fact that
many used a single measure of current exposure, limited their
usefulness in answering questions relating to the natural history of
the association between lead exposure and outcomes, including whether
there were critical periods of exposure, and whether lead-associated
deficits were persistent or reversible. Equally they were limited in
the information that could be obtained relating to the natural history
of confounders. They were also unable to answer questions of reverse
causality.
8.3.6.5 An interpretation of the overview of prospective and
cross-sectional studies
The above meta-analyses of the prospective studies and cross-
sectional studies reveal a consistency between studies which points
towards a "collectively significant" inverse association between PbB
level and full-scale IQ. Taking the results in Fig. 15 as a guideline,
there appears to be a mean decrease in full-scale IQ of the order of 2
IQ points for a change in mean PbB level from 0.48 to 0.96 µmol/litre
(10 to 20 µg/dl).
Below this range, uncertainties are increased, concerning firstly
the existence of an association and secondly estimates of the
magnitude of any putative association. The relatively limited numbers
of children in most studies with PbB levels below this range, the
strong contributions of confounding variables, and limitations in the
precision in analytical and psychometric measurements combine to lower
statistical power to detect associations and to estimate their
magnitude.
The key question is whether this statistical association is
directly attributable to the causal influence of lead on child IQ. It
is important to consider alternative explanations as follows:
a) Chance The consistency of alternative analyses and the level of
significance achieved suggest that chance cannot be a complete
explanation. However, the confidence intervals are relatively wide, so
that the magnitude of true association could be as low as a < 1-point
(rather than a 2-point) deficit, or as high as > 3 points.
b) Confounding factors The adjusted lead/IQ associations tend to be
substantially weaker than the unadjusted associations, which indicates
that confounding factors are important. Since none of the studies can
claim to have taken complete account of confounders, (e.g., father's
IQ is not included), it seems likely that some of the remaining
lead/IQ relationship, after adjustment, may still be attributable to a
degree of unexplained confounding.
c) Reverse causality One initial justification for the prospective
studies was that they could measure early exposure to lead and relate
it to later child development, thus removing the problem of reverse
causality potentially present in the cross-sectional studies. However,
it has turned out that very early lead exposure, e.g., in the
perinatal period, is not related to school-age IQ. The question arises
as to whether children of lower IQ could exhibit behaviour patterns at
earlier ages, e.g., around age 2 and older, which could enhance their
uptake of lead. Perhaps, such reverse causality remains a possibility
even in the prospective studies.
d) Selection biases The positive studies may perhaps be more likely
to report in more quantitative details, as indicated by the absence of
relevant data from Cleveland in the prospective studies. Post-hoc
selection based on the more significant PbB concen-trations may lead
to exaggerated estimates.
It is a matter of debate and conjecture as to the extent to which
these four issues should inhibit claims of a causal relationship in
the prospective studies. The essential problem is that observational
epidemiology cannot provide definitive evidence of causality when the
key statistical association is weak, the temporal relationship is
unclear and major confounders are present.
8.3.7 Hearing impairment in children
Schwartz & Otto (1987) reported that the probability of elevated
pure-tone hearing thresholds at 500, 2000 and 4000 Hz increased
significantly with increasing PbB level in 4519 subjects (4-19 years
of age) who participated in the NHANES II study. Variables included in
the backwards stepwise regression models included information derived
from medical history, clinical examinations, and assessment of
developmental milestones.
Schwartz & Otto (1991) examined data for subjects (6-19 years of
age) from the Hispanic Health and Nutrition Examination Survey
(HHANES) study which included three distinct ethnic groups. After
excluding 283 subjects with previous ear problems (discharges,
ruptured ear drums or tinnitus), data for 3262 were analysed. The
authors concluded that increasing PbB level in the range
0.36-0.86 µmol/litre (7-18 µg/dl) was associated with approximately
2 dB loss of pure-tone hearing at frequencies of 500, 1000, 2000 and
4000 Hz.
Dietrich et al. (1992) assessed the relationship between scores
on a test of sensory auditory processing (SCAN) and prenatal/postnatal
PbB concentrations in 215 subjects drawn from the Cincinnati
prospective cohort study. Higher prenatal, neonatal and postnatal PbB
concentrations were associated with more incorrect identification of
common monosyllabic words presented under conditions of muffling.
Other variables associated with impaired central auditory processing
were pure-tone audiometry results, social class, quality of caretaking
in the home, birth weight, gestational age, a measure of obstetrical
complications, and consumption of alcohol during pregnancy. Following
adjustment for these co-factors, lifetime average PbB concentration
remained significantly and inversely associated with SCAN performance.
8.4 Renal system
Acute exposure to lead is known to cause proximal renal tubular
damage, characterized by generalized aminoaciduria, hypophosphataemia
with relative hyperphosphaturia, and glycosuria (Chisolm, 1962).
Cellular structural changes include nuclear inclusion bodies,
mitochondrial changes and cytomegaly of the proximal tubular
epithelial cells (Cramer et al., 1974). Diagnosis of lead-induced
altered renal function or disease is difficult since there are no
specific indicators; blood urea nitrogen (BUN) and creatinine levels
become elevated only when twothirds of renal function has been lost
(Bernard & Becker, 1988).
8.4.1 Clinical studies
From a study of seven men occupationally exposed to lead in a
shipyard during oxy-acetylene flame cutting of lead-painted steel
hulls, Cramer et al. (1974) concluded that there is a continuum of
morphological and functional change in the pathogenesis of chronic
lead nephrotoxicity. Each subject was treated in hospital 3 or more
days after recent exposure and PbB levels exceeded 3.36 µmol/litre
(70 µg/dl) in all cases. On the basis of microscopic examination of
biopsy tissue, nuclear inclusion bodies were reported in proximal
tubular cells during the early phase but there was no impairment of
renal function. In a second phase, there was fibrosis associated with
asymptomatic azotaemia and reduced glomerular filtration rate but
without demonstrable proximal tubular dysfunction; renal failure was
not seen in the study.
Weeden et al. (1979) diagnosed lead nephropathy in 15 lead
workers, all having reduced glomerular filtration rates. Renal
biopsies of six of the subjects showed focal interstitial nephritis in
addition to non-specific changes in the proximal tubules. At the time
of examination, PbB levels for 11 of the 15 workers were within the
range 1.92-3.84 µmol/litre (40-80 µg/dl).
Baker et al. (1979) reported increased BUN and decreased
creatinine clearance in 28 workers, all of whom had relatively
prolonged, high-dose lead exposure in lead smelting or chemical
manufacturing.
A study by Maranelli & Apostoli (1987) of 60 workers, described
as "lead poisoned" and having PbB levels of 3.45 ± 0.80 µmol/litre
(71.9 ± 16.6 µg/dl), found no definitive correlation between PbB, lead
in urine after chelation, and BUN, serum creatinine and serum uric
acid.
8.4.2 Epidemiological studies
8.4.2.1 Occupational cohorts
Several recent studies of lead-exposed workers provide further
information relating to dose-effect relationships. The emphasis in
these studies was on lead and its possible association with adverse
effects on health. However, exposure to other potentially toxic
substances in the working and living environment should not be
overlooked/ignored.
For example, Buchet et al. (1980) examined 25 male lead smelter
workers and 88 male control workers. The PbB levels of the lead
workers were in the range 1.62-2.94 µmol/litre (33.8-61.3 µg/dl) for a
mean of 13.2 years (range 3.1-29.8 years) of lead exposure. The PbB
levels for the controls were in the range 0.26-1.64 µmol/litre
(5.5-34.2 µg/dl). There were no differences for parameters of renal
function between the groups and no signs of clinical renal impairment.
It was concluded that a PbB level of less than 2.98 µmol/litre
(62 µg/dl) is not associated with renal toxicity.
N-acetyl-ß-d-glucosaminidase (NAG) is a lysosomal enzyme present
in renal tubular cells. This enzyme is a sensitive but non-specific
indicator for early sub-clinical nephrotoxicity. In a study on 29
lead-exposed workers, Meyer et al. (1984) found increased NAG in
urine, but there was no correlation with PbB level. However, NAG level
was found to be normal in the 5 subjects with PbB levels exceeded
3.36 µmol/litre. These authors speculated that long-term high exposure
to lead may deplete the kidneys of NAG or render it insensitive to the
effects of lead exposure.
Verschoor et al. (1987) studied 155 male lead workers and 126
control workers who were matched for age, smoking habits,
socioeconomic status and duration of employment. The PbB levels of the
lead workers were in the range of 0.43-4.71 µmol/litre
(8.3-97.6 µg/dl) compared with the controls 0.15-0.96 µmol/litre
(3.1-18.8 µg/dl). The lead workers had elevated ZPP levels
(34-292 µmol/mol haemoglobin) compared with controls (10-35 µmol/mol
haemoglobin). No significant differences were found for various
indicators of renal function; all urinary and serum parameters were
within normal ranges. There were no differences in protein excretion
patterns and no signs of renal impairment. However, the authors found
that NAG levels in the lead-exposed workers were higher than control
values and increased with increasing PbB levels. They concluded that
lead exposure resulting in PbB levels of under 3.0 µmol/litre
(62 µg/dl) can affect renal tubular functions as measured by NAG
excretion; lead appeared to affect the tubular parameters more than
the glomerular parameters in moderately exposed workers.
Ong et al. (1987) examined 158 male and 51 female lead battery or
smelter workers and 30 control workers. The lead workers had 1-36
years exposure with an average of 10.8 ± 8.0 years and PbB levels in
the range of 0.14-3.84 µmol/litre (3-80 µg/dl); only five workers
exceed 2.88 µmol/litre (60 µg/dl). The authors found a weak but
statistically significant positive association between PbB and blood
urea nitrogen, and between PbB and serum creatinine, and that
creatinine clearance was reduced with increasing PbB level. NAG levels
in the lead-exposed workers were significantly higher than control
values and increased with increasing urine lead level when the data
were adjusted for age. These authors concluded that a relatively low
PbB level can affect renal function.
Not all investigators have found parallel association between PbB
and NAG levels. Gennart et al. (1992b) compared 98 lead-exposed lead
acid battery workers (mean PbB level 2.45 µmol/litre, 51 µg/dl,
geometric mean ZPP 10.2 µg/g Hb) with 85 controls (mean PbB level
1.00 µmol/litre, 20.9 µg/dl, geometric mean ZPP 2.84 µg/g Hb)
recruited from other departments in the same factory. None of the
indicators of renal function (retinol-binding protein,
ß2-microglobulin, albumin or NAG in urine, or creatinine or
ß2-microglobulin in serum) were correlated with PbB level, duration
of exposure or ZPP, or showed significantly different mean values
between the lead-exposed and control groups of workers.
Cardenas et al. (1993) recently evaluated 27 different
indications of renal dysfunction in 50 workers exposed to lead and 50
controls. There were significant increases in urinary NAG and sialic
acid in the lead exposed group. These changes may represent minor
cellular modifications rather than significant functional or
irreversible renal damage. There was a significant decrease in urinary
6-keto PGF1alpha and a significant increase in TXB2. These
eicosanoid changes may reflect systemic functional vascular changes
rather than an effect of lead on the kidney.
8.4.2.2 General population
An epidemiological survey on 283 persons in Scotland, from
households with water lead concentrations in excess of 100 µg/litre,
revealed a close correlation between water lead content and PbB and
serum urea concentrations (Campbell et al., 1977). The frequency of
renal dysfunction in individuals with elevated PbB concentrations
(> 2 µmol/litre or > 41 µg/dl) was significantly greater than that
of age- and sex-matched controls.
Pocock et al. (1984) measured serum creatinine, urate and urea
concentrations in 7364 British men, and 74 subjects had PbB levels
equal to or greater than 1.8 µmol/litre (37.3 µg/dl). The authors
concluded that there was no indication that exposure to lead at
concentrations commonly encountered in Britain was responsible for
impaired renal function.
Staessen et al. (1992) investigated the relationship between lead
exposure and renal function as part of a cross-sectional population
study of the health effects of environmental exposure to cadmium.
Creatinine clearance, PbB and ZPP were measured in a random population
sample of 965 men (geometric mean PbB level was 0.55 µmol/litre or
11.4 µg/dl) and 1016 women (geometric mean PbB level was
0.36 µmol/litre or 7.5 µg/dl). Creatinine clearance rate (mean was
99 ml/min in men, 80 ml/min in women) was inversely correlated with
PbB and ZPP levels before and after adjustment for age, body mass
index and diuretic treatment. Also positively correlated were serum
ß2-microglobulin and PbB level in men, serum ß2-microglobulin and ZPP
in men and women, and serum creatinine and ZPP in men. Impaired renal
function could not be explained by exposure to cadmium or by elevated
blood pressure. The authors concluded that exposure to lead may impair
renal function in the general population. However, the study could not
exclude the possibility that renal impairment may lead to an increase
in PbB level.
8.4.2.3 Cohort mortality studies
"Lead poisoning" was a diagnosis given to 241 workers employed
for 1-30 years at the Port Pirie lead smelter between 1928-1959 by a
State medical board. Death registration records for the period
1930-1977 identified 140 deaths among the group. The cause of death
profile was compared with that of 695 other male decedents,
predominantly production workers and a smaller number of office
workers. Age-standardized mortality analysis revealed a substantial
excess of deaths attributed to chronic nephritis and to cerebral
haemorrhage. The rates for lead-poisoned workers exceeded those for
the other workers, and both exceeded the rates for the Australian
general male population. The rates decreased over successive calendar
periods but excess rates persisted for chronic nephritis up until 1977
(McMichael & Johnson, 1982).
8.5 Cardiovascular system
Two persistent issues have been under intense study since
publication of Environmental Health Criteria 3: Lead (IPCS, 1977):
a) whether lead is a factor in hypertension and, if so, whether
there is a causal relationship;
b) whether lead is contributory to cardiovascular effects
influencing morbidity or mortality.
8.5.1 Blood pressure
8.5.1.1 Studies on occupationally exposed cohorts
From a study of 431 white male police officers 24-55 years of age
(Moreau et al., 1982; Orssaud et al., 1985), it was concluded that
systolic and diastolic blood pressure was related to PbB level, the
correlation being greatest for the younger subjects and decreasing
with age. Statistical adjustment was made for alcohol consumption and
body mass index, but not for smoking. The magnitude of observed
association between systolic pressure and PbB level in this small
study was somewhat greater than in the larger general population
studies cited below.
Parkinson et al. (1987) studied 270 lead battery workers and 158
non-exposed workers and compared lead exposure with systolic and
diastolic blood pressures. After controlling for age, education,
income, cigarette smoking, alcohol consumption and exercise, there was
a small and non-significant association. The average PbB level of
lead-exposed workers was 1.92 ± 0.62 µmol/litre (40 ± 13 µg/dl),
whereas in non-exposed workers it was 0.34 ± 0.24 µmol/litre
(7 ± 5 µg/dl).
8.5.1.2 Studies in the general population
The possible relationship between PbB concentration and blood
pressure has been examined in several large-scale population studies.
These include the British Regional Heart Study (BRHS), the US NHANES
II (National Health and Nutrition Examination Survey) and studies in
Wales, Denmark, Canada and Belgium.
The BRHS is a prospective study of 7735 men, initially aged 40-59
years, from 24 British towns, who were first examined in 1978-1980
(Shaper et al., 1981). The median PbB level was 0.7 µmol/litre
(14.5 µg/dl). The initial findings (Pocock et al., 1984), which
indicated a lack of association between PbB level and blood pressure,
were later re-examined to take account of potential confounding
factors such as alcohol, smoking and town of residence. Pocock et al.
(1988) noted that 95% confidence intervals for systolic and diastolic
blood pressure plotted against PbB concentrations overlapped and
showed no elevation even at the highest observed PbB concentration.
Applying statistical techniques to adjust mean systolic and diastolic
blood pressure measurements for body mass index, age, alcohol
consumption, cigarette smoking and town of residence showed signs of a
weak association, particularly for diastolic pressure and PbB level.
Multiple regression analysis showed that adjustment for personal
characteristics but not for town of residence rendered both systolic
and diastolic regressions on log PbB insignificant, chiefly because
alcohol consumption is an important confounder, being positively
related to both blood pressure and PbB level. Introducing an extra
adjustment for town of residence made the systolic and diastolic
regressions highly statistically significant, even though the
associations were weak. These cross-sectional data indicated that for
every doubling in PbB level (e.g., from 0.8 to 1.6 µmol/litre) there
are estimated mean increases of 1.45 mmHg (95% confidence interval
0.47-2.43 mmHg) in systolic blood pressure and 1.25 mmHg (95%
confidence interval 0.65-1.85 mmHg) in diastolic blood pressure.
The NHANES II study, a USA national cross-sectional survey
carried out in 1976-1980, included PbB and blood pressure measurements
in a general population sample of 5803 men and women aged 12-74.
Geometric mean PbB levels were around 0.72 µmol/litre (about 15 µg/dl)
for men and approximately 0.53 µmol/litre (11 µg/dl) for women.
Several authors have examined these data for possible PbB and blood
pressure associations.
In an analysis controlling for the confounding factors of age,
race and body mass index, Harlan et al. (1985) and Harlan (1988) found
a significant association between PbB level and blood pressure for men
but not for women. White males aged 40-59 years from the same data set
were analysed by Pirkle et al. (1985), and the correlation between PbB
level and blood pressure was confirmed. In further analysis of the
whole age range, adjustments for possible time trend and geographical
site effects did not affect the significance of the association in
males (Landis & Flegal, 1988; Schwartz, 1988), although its magnitude
did become somewhat less pronounced.
Gartside (1988) applied a method of forward stepwise regression
to PbB and blood pressure data from NHANES II and reported that the
results for white men, white women and black men were contradictory
and lacked consistency and reliability. He noted that the overall
average was too small to conclude association between PbB level and
blood pressure in this study.
Two surveys were carried out in Wales by Elwood et al. (1988a,b).
The Welsh Heart Programme carried out in 1985 provided complete PbB,
blood pressure and other data for 865 men and 856 women aged 18-64.
The geometric mean PbB level for men was 0.56 µmol/litre (11.6 µg/dl)
and for women was 0.43 µmol/litre (9.0 µg/dl). In neither sex was
there a significant correlation between PbB level and systolic or
diastolic blood pressure. The second study was in a cohort of men aged
49-65 years living in Caerphilly, Wales. The geometric mean PbB level
(N = 1137) was 0.61 µmol/litre (12.7 µg/dl). Complete data from 1137
subjects, ranking blood pressure readings according to PbB level, did
not reveal any trend in the percentage of subjects with systolic
pressure above 160 mmHg. The authors corrected only for age as a
confounding factor.
Data from a Canadian study (Neri et al., 1988) collected during
10 months in 1978-1979 for 2193 subjects aged 25-64 showed a weak but
statistically significant association between PbB level and diastolic
blood pressure.
Grandjean et al. (1989b) studied 504 men and 548 women residing
in Glostrup (Denmark) at age 40; 451 men and 410 of the women were
followed-up five years later. Average PbB levels for the men at 40 and
45 years of age were 0.62 µmol/litre (13 µg/dl) and 0.43 µmol/litre
(9 µg/dl), and for the women 0.43 µmol/litre (9 µg/dl) and
0.29 µmol/litre (6 µg/dl), respectively. All correlations found
between PbB and blood pressure became insignificant when blood
haemoglobin and alcohol intake were entered into multiple regression
analyses; an independent effect of low-level lead exposure on blood
pressure could not be distinguished.
Möller & Kristensen (1992) examined 1052 men and women in
Copenhagen, Denmark, in 1976, and re-examined them in 1981 and 1987
(men only). Initial mean PbB levels were 0.65 µmol/litre (13.6 µg/dl)
in men and 0.46 µmol/litre (9.6 µg/dl) in women, and had fallen by
around 30% 5 years later. For men, there was no significant
association between PbB level and blood pressure (or between changes
in PbB level and blood pressure) after adjustment for confounders
(tobacco, alcohol, body mass and physical activity). For women, the
association between PbB level and diastolic (but not systolic) blood
pressure remained significant on both occasions, even after
controlling for confounders.
A Belgium study (Dolenc et al., 1993) included 827 men and 821
women (mean age 25) whose mean PbB levels were 0.56 and
0.34 µmol/litre (11.6 and 7.07 µg/dl), respectively. After adjustment
for covariates (body mass index), pulse rate and serum creatinine and
serum calcium levels), systolic blood pressure was inversely
associated with PbB level in men (P < 0.05). There were no
significant PbB associations for diastolic blood pressure in men or
for either pressure in women.
A study of 398 male and 133 female civil servants (Staessen et
al., 1990) showed geometric mean PbB levels of 0.58 µmol/litre
(12.06 µg/dl) and 0.46 µmol/litre (9.56 µg/dl) in men and women,
respectively. Taking the data for both sexes together, there were
statistically significant positive associations between PbB level and
systolic and diastolic blood pressures (r = +0.11 in each case) but
these became non-significant after adjustment for confounders (sex,
age, BMI, pulse, delta-glutamyltranspeptidase (delta-GTP) and serum
calcium level).
Another cross-sectional study involving only women was performed
in Boston (Rabinowitz et al., 1987). Cord PbB levels (mean
0.33 µmol/litre, 6.9 µg/dl) among 3851 women correlated with both the
systolic (r = 0.08) and diastolic (r = 0.05) blood pressures measured
during and before delivery. Multivariate models of pregnancy
hypertension as a function of age, parity, haematocrit, diabetes,
ponderal index, and race were improved when lead was included as a
predictor. Lead appeared to have a small but demonstrable association
with pregnancy hypertension and blood pressure at delivery, but not
with eclampsia.
In an overview of the BRHS, NHANES II and Welsh studies, Pocock
et al. (1988) concluded that with overlapping confidence limits the
data provided weak but reasonably consistent evidence of lead and
blood pressure associations. They noted that the NHANES II data on
2254 men in the USA indicate a slightly stronger association between
PbB level and systolic blood pressure, whereas data from over 2000 men
in Wales did not show a statistically significant association. They
inferred that a causal relationship could not be concluded from any of
the epidemiological studies.
Staessen et al. (1994) have undertaken a more extensive
meta-analysis of nearly all of the above studies and some other
smaller ones as well. This amounted to a total of 19 studies with
28 210 subjects. They found that the association between PbB level and
blood pressure was similar in both sexes. In all studies combined, a
two-fold increase in PbB concentration was associated with a 1.0 mmHg
increase in systolic pressure (95% confidence interval (CI); 0.3 to
1.7 mmHg; P = 0.008) and a 0.7 mmHg increase in diastolic pressure
(95% CI: 0.2 to 1.3 mmHg; P = 0.02). These authors conclude that the
published evidence suggests a weak positive association between blood
pressure and lead exposure, but any such relationship may not be
causal and is unlikely to entail any public health implications
regarding hypertension.
8.5.2 Other cardiovascular effects
As recorded previously (IPCS, 1977), there is good evidence that
signs of clinical lead poisoning sometimes include evidence of toxic
action on the heart. Kopp et al. (1988) reviewed the cardiovascular
actions of lead and concluded that the degree of cardiovascular
involvement during episodes of acute lead intoxication depends on the
duration of exposure and dose. It is not known whether environmental
exposure to lead affects the electrical or mechanical activity of the
heart.
8.5.2.1 Occupational studies
There have been several studies relating to cardiovascular
effects in lead-exposed occupational groups. However, as with any
study of an occupational cohort, it is important to evaluate the
strength of the cause-effect relationship, the nature or substance of
other contributory factors and how well the workplace "exposure" has
been evaluated for other potential toxicants. Thus, the occupational
hazards associated with work in lead smelting or lead battery
operations include, but are not limited to, lead.
8.5.2.2 Studies in the general population
Pocock et al. (1988) found that after 6 years of follow-up of the
BRHS cohort of 7735 middle-aged men, 316 of the men had major
ischaemic heart disease and 66 had had a stroke. After allowance for
confounding effects of cigarette smoking and town of residence, there
was no evidence that PbB was a risk factor for these cardiovascular
events.
Möller & Kristensen (1992) studied 1052 men and women in
Copenhagen, Denmark, and related blood pressure to subsequent
cardiovascular morbidity and mortality over a 14-year period. There
were significant positive associations between PbB level and both
coronary and cardiovascular disease in univariate analysis, but these
became non-significant after controlling for confounders.
a) Clinical studies
Boscolo & Carmignani (1988) measured plasma renin activity in
hospitalized lead-exposed workers and concluded that synthesis or
release of renin is increased by short or moderate exposure to lead
and decreased with prolonged exposure.
b) Epidemiological studies
Kirkby & Gyntelberg (1985) found a significantly higher incidence
of ischaemic ECG changes in a study of lead smelter workers (20%, mean
PbB level 2.45 µmol/litre, 51 µg/dl), compared with matched
non-exposed controls (6%, mean PbB level 0.53 µmol/litre, 11 µg/dl).
There was also a slight (4-5 mmHg) increase in diastolic blood
pressure in the lead workers compared with the controls.
c) Cohort mortality studies
Two studies (Cooper, 1988 and Fanning, 1988) reported an
increased mortality rate of lead workers due to circulatory disease,
but no such relationship was found in reports of studies by
Gerhardsson et al. (1986) or Selevan et al. (1988). Gerhardsson et al.
(1986) examined a lead-exposed sub-cohort of 437 from a study cohort
of 3832 male workers first employed at a copper smelter before 1967
and followed up from 1950 to 1981. There was no excess mortality due
to ischaemic heart disease or cerebrovascular disease in the
lead-exposed worker sub-cohort. Selevan et al. (1988) found no
association between lead exposure and deaths due to hypertensive
diseases in a review of mortality in a cohort of male hourly workers
at a lead smelter. There were many confounding factors in each of
these studies.
8.5.3 Summary
Despite intensive efforts to define the relationship between body
burden of lead and blood pressure or other effects on the
cardiovascular system, no causal relationship has been demonstrated in
humans and the mechanisms remain obscure.
There is experimental evidence from animal studies indicative of
an effect of lead on blood pressure, and several mechanisms have been
proposed to explain these observations (see section 7.4).
8.6 Gastrointestinal effects
8.6.1 Occupational exposure
Colic is a well-recognized symptom of acute lead poisoning and is
still reported in groups of lead-exposed industrial workers. Of
particular concern are the reports of intoxication caused by acute
exposure (inadequate protective measures) to lead associated with
removal of lead-based paint by burning or sand-blasting and the
demolition of lead-containing industrial plants. Symptoms of colic
include abdominal pain, constipation, cramps, nausea, vomiting,
anorexia, weight loss and decreased appetite.
Symptoms in adults typically occur at PbB levels of
4.8-9.6 µmol/litre (100-200 µg/dl) but have been noted at levels as
low as 1.92 µmol/litre (40 µg/dl) (Baker et al., 1979; Haenninen et
al., 1979; Awad El Karim et al., 1986; Pollock & Ibels, 1986; Muijser
et al., 1987; Holness & Nethercott, 1988; Marino et al., 1989;
Pagliuca et al., 1990; Schneitzer et al., 1990).
Positive histories of lead colic were given by 40 workers in a
cohort of 158 secondary lead smelter workers who participated in a
clinical field survey (Lilis et al., 1977). It was reported most
frequently by those having PbB levels above 3.84 µmol/litre (80 µg/dl)
at the time of examination and was not found where ZPP was within the
normal range. Thirty percent of workers having ZPP > 200 µg/g Hb had
experienced symptoms.
In a study of a population of 585 black South African factory
workers, Irwig et al. (1978) reported that the incidence of abdominal
pain increased with increasing PbB level: it was 12% for the first
quartile (PbB < 3.28 µmol/litre or < 68 µg/dl), 23% for the
second (3.28-4.14 µmol/litre or 68-86 µg/dl), 24% for the third
(4.15-5.14 µmol/litre or 86-107 µg/dl) and 37% for the fourth
(> 5.15 µmol/litre or > 107 µg/dl).
In a cross-sectional clinical study of 90 telephone
cable-splicers by Fischbein et al. (1980), 19 (21%) workers reported
gastrointestinal symptoms (mean PbB level of 1.44 µmol/litre, or
30 µg/dl and mean ZPP of 66.6 µg/g Hb) whereas 71 (79%) reported no
such symptoms (mean PbB level of 1.3 µmol/litre or 27 µg/dl and mean
ZPP of 52.3 µg/g Hb).
8.6.2 Exposure of children
Colic is seen in children and US EPA (1986a) concluded that the
lowest-observed-adverse-effect level was in the range of
2.88-4.80 µmol/litre (60-100 µg/dl).
8.7 Liver
There appears to be no new evidence relating human body lead
burden to effects on the liver, but it has been suggested that the
effects of lead on haem synthesis may alter the functional capacity of
the hepatic cytochrome P-450 system to metabolize drugs.
8.7.1 Occupational exposure
Fischbein et al. (1977) tested five demolition workers acutely
exposed to lead for a period of 3 months prior to the study. They
found that the plasma half-life of an oral dose of antipyrine was
within the range for normal healthy volunteers; it was shorter in each
subject after chelation therapy but still within the normal range. The
plasma half-lives of phenylbutazone were also within the normal range
but were unaffected by chelation therapy.
8.7.2 Exposure of children
Saenger et al. (1984) found decreased urinary excretion of
6-ß-hydroxycortisol in 26 children with a mean PbB level of
2.11 µmol/litre (44 µg/dl); the decreased formation of the metabolite
was attributed to lead inhibition of the cytochrome P-450-dependent
mixed-function oxidases.
8.8 Reproduction
While it is generally accepted from early literature that lead
adversely affects the reproductive process in both men and women, the
evidence is mostly qualitative and dose-effect relationships have not
been established.
Most new information relates to reports of occupational cohorts
and of populations living in polluted areas near industrial plants.
There is qualitative evidence that lead is toxic to the reproduction
system in both men and women. However, there are insufficient data to
provide the basis for estimation of dose-effect relationships in
women.
8.8.1 Female populations
Nordstrom et al. (1978b) reported an increased frequency of
spontaneous abortion in women living close to a smelter in northern
Sweden. In a later report, Nordstrom et al. (1979) described the
responses to a questionnaire completed by 511 of 662 women who had
worked at the smelter and were born between 1930 and 1959. Spontaneous
abortion rates were highest in those pregnancies in which the mother
was employed during the pregnancy (13.9%) or had been employed before
the pregnancy and was living close to the smelter (17%); the frequency
rate was higher (19.4%) when the father worked at the smelter.
However, it should be noted that the smelter produced copper and lead
in addition to a number of other metallurgical and chemical products
(Nordstrom et al., 1978a) and that the effects reported may not
necessarily be attributable exclusively to lead.
A study of pregnancies in the centre and surrounding areas of the
lead smelter town of Port Pirie showed that the incidence of
miscarriages (22 or 23) and stillbirths (10 or 11) was higher in women
living close to the smelter (McMichael et al., 1986). However, in a
study of 639 Yugoslav women, the risk of spontaneous abortion was not
increased (OR = 1.1, 95% CI: 0.9-1.4) among women with mean PbB levels
of 0.77 µmol/litre (16.0 µg/dl) as compared with women with mean PbB
levels of 0.25 µmol/litre (5.2 µg/dl) (Murphy et al., 1990). Risk did
not vary with distance of residence from the smelter.
Some studies have found decreased length of gestation in women
whose PbB levels were greater than 1.09 µmol/litre (23 µg/dl) (Moore
et al., 1982), 0.58 µmol/litre (12 µg/dl) (Dietrich et al., 1986) or
0.72 µmol/litre (15 µg/dl) (McMichael et al., 1986). However, neither
Bellinger et al. (1991b) nor Graziano et al. (1990) found decreases in
gestational length or other parameters of pregnancy in women with
elevated PbB levels.
In the Cincinnati prospective study, a significant reduction in
birth weight associated with prenatal (maternal) PbB levels, after
adjustment for covariates, was reported by Bornschein et al. (1989).
In the Port Pirie prospective study of 749 pregnancies (McMichael et
al., 1986), the proportion of pregnancies resulting in low birth
weight singleton infants was more than twice as high in Port Pirie
women (whose PbB levels averaged 0.50 µmol/litre, 10.4 µg/dl) than in
women outside Port Pirie (average PbB level of 0.264 µmol/litre,
5.5 µg/dl). On the other hand, multiple regression analyses showed no
significant association between low birth weight and maternal PbB
level. In a cross-sectional study, Ward et al. (1987) reported a
significant simple relationship between placental lead concentrations
and reduced birth weight and head circumference.
Other studies have not shown a significant association between
birth weight and lead exposure. The Kosovo prospective study failed to
detect any evidence of lead-related birth weight reduction in more
than 900 births (Murphy et al., 1990), and Ernhart et al. (1986) found
no significant effect of lead on birth weight, birth length or head
circumference.
8.8.2 Male populations
Reproductive effects from occupational exposure to lead include
asthenospermia, hypospermia, teratospermia and hypogonadism (US EPA,
1986a; Braunstein et al., 1987).
In men, effects on sperm or the testes may result from chronic
exposure to lead at blood levels of the order of 1.92-2.4 µmol/litre
(40-50 µg/dl) (ATSDR, 1991).
Wildt et al. (1983) compared two groups of lead-exposed storage
battery workers and concluded that the highly exposed group (PbB of
more than 2.16 µmol/litre, 45 µg/dl) had decreased prostate/seminal
vesicle function, low semen volume and poorer functional maturity of
sperm.
Chowdhury et al. (1986) reported a significant decrease in sperm
count and motility and an increased count of abnormal spermatozoa in
lead-exposed men (average PbB level of 2.04 µmol per litre or
42.5 µg/dl) compared with controls (0.71 µmol/litre or 14.8 µg/dl).
Assenato et al. (1987) also reported decreased sperm production in 18
battery factory workers having PbB levels of 2.4-2.93 µmol/litre
(50-61 µg/dl).
In five out of six symptomatic lead intoxicated workers having
PbB levels in the range of 1.87-4.7 µmol/litre (39-98 µg/dl) at the
time of examination, Cullen et al. (1984) found defects of
spermatogenesis, including oligospermia and azoospermia.
The results of Lerda (1992) also showed significant increases in
asthenospermia and teratospermia among 38 battery workers with PbB
levels of 1.95-4.7 µmol/litre (40.5-98.0 µg/dl) compared to a control
group with PbB levels of 0.86-1.25 µmol/litre (17.6-26.0 µg/dl).
An epidemiological study by Lindholm et al. (1991) of 213 wives
of lead workers and 300 matched controls suggested an association
between paternal lead exposure and the risk of spontaneous abortion
among the wives of lead workers when the PbB level exceeded
1.5 µmol/litre (31 µg/dl) close to the time of spermatogenesis.
However, the authors acknowledged that exposure of the study subjects
to other metals and organic solvents may have influenced the results
of the study.
A weak association between paternal exposure, estimated from
occupational history, and risk of esotropia (inward deviation
strabismas) was noted in a case control study of 377 children aged 7
years (OR - 2.4, 95%, CI: 1.0-5.6) (Hakim et al., 1991). However, the
increased risk was not dose-related, and the risk of exotropia
(outward deviation) was not associated with higher paternal lead
exposure.
8.8.3 Hormonal responses
Assenato et al. (1987) found no significant differences in FSH,
testosterone, prolactin, LH and total neutral 17-ketosteroids levels
between 18 lead battery workers (PbB level, 2.4-3 µmol/litre or
50-61 µg/dl) and 18 cement workers (PbB level, 0.9-1.1 µmol/litre or
18-22 µg/dl), despite the finding of oligospermia in the lead-exposed
group.
Rodamilans et al. (1988) studied the endocrine status of 23 lead
smelters in relation to the duration of lead exposure. Five workers
exposed for less than 1 year (mean PbB level, 0.29 µmol/litre or
66 µg/dl) showed an increase in serum LH level, while that of
testosterone remained normal. Eight workers exposed for 1-5 years
(mean PbB level, 3.50 µmol/litre or 73 µg/dl) and 10 employed for more
than 5 years (mean PbB level, 3.65 µmol/litre or 76 µg/dl) showed an
increase in LH comparable with the group exposed for less than 1 year
but there was a clear reduction in serum testosterone levels.
Gustafson et al. (1989) found a lower plasma FSH level in 25
moderately exposed workers (mean PbB level, 1.82 µmol/litre or
38 µg/dl) than in 25 matched controls. A lower mean serum LH level was
reported by McGregor & Mason (1990) in 90 lead workers (mean PbB
level, 2.20 µmol/litre or 46 µg/dl) than in controls; there was also a
correlation between serum FSH and PbB levels. In an epidemiological
study involving 122 current lead workers (PbB mean level,
1.69 µmol/litre or 35.1 µg/dl) and 49 non-exposed workers (PbB mean
level, 0.4 µmol/litre or 8.3 µg/dl), Ng et al. (1991) found slightly
higher plasma LH and FSH levels among the exposed workers. The
testosterone level, however, was not significantly different between
the two groups. In contrast, Gennart et al. (1992) did not find an
effect of lead on various endocrine parameters (including FSH and LH
levels) in a study population of 221 workers having a geometric mean
PbB level of 2.45 µmol/litre (51.0 µg/dl). They suggested that the
hypothalamic-pituitary system may not be influenced by moderate
exposure to lead.
8.8.4 Postnatal growth and stature
Several reports have suggested that the physical growth and
stature of children may be reduced by exposure to lead (e.g., Mooty et
al., 1975; Johnson & Tenuta, 1979; Routh et al., 1979), but the
influence of other factors (e.g., race and diet) has often made it
difficult to isolate lead as a causal agent for such effects in human
populations. Multivariate regression analyses of NHANES data for
approximately 2700 children in the USA (Schwartz et al., 1986)
provided more convincing evidence of a significant association between
increasing PbB levels and reduced height, weight, and chest
circumference after adjusting for age, race, sex and nutritional
covariates.
8.9 Effects on chromosomes
As noted in Environmental Health Criteria 3: Lead (IPCS, 1977),
the literature remains controversial concerning induction of
chromosomal changes in human lymphocytes by in vivo exposure to
lead. Most of the studies are of small numbers of subjects in
occupational groups where lead was one of many potentially toxic
agents. However, some of the difficulties arise because of the lack of
standard procedures used for the assessment of chromosomal effects in
cultured lymphocytes and from a lack of understanding of the health
significance of chromosomal abnormalities.
Induced mitotic activity in peripheral lymphocytes and increases
in the rates of abnormal mitosis were reported by Sarto et al. (1978).
PbB levels in the range of 1.05-4.27 µmol/litre (22-89 µg/dl) were
reported to be associated with increased incidence of chromosomal
aberrations by Schmid et al. (1972) in 32 lead manufacturing workers,
by Nordenson et al. (1978) in 18 smelter workers and by Al-Hakkak et
al. (1986) in 19 lead manufacturing workers. It should be noted that
exposure of worker to potentially hazardous agents other than lead was
not documented in these studies.
Maki-Paakkanen et al. (1981) reported no change in chromosomal
aberration frequency among workers with PbB levels in the range of
1.82-5.76 µmol/litre (38-120 µg/dl); weak effects were found by Huang
et al. (1988b) when the PbB level exceeded 2.5 µmol/litre (52 µg/dl).
Qazi et al. (1980) reported increased numbers of cells with
chromosome breaks in a baby having a cord PbB level of 2.88 µmol/litre
(60 µg/dl) and a PbB level of 3.45 µmol/litre (72 µg/dl) at 2 weeks.
The effect was seen at 6 weeks and at 3 months but not later
(chelation therapy was given at 17 days and repeated at 5 months).
Bauchinger et al. (1977) tested blood taken from 38 children
selected from a school situated close to a lead plant and in whom the
PbB level was at least 1.44 µmol/litre (30 µg/dl). There was no
increase in the frequency of aberrations.
Grandjean et al. (1983) examined 10 long-term workers at a lead
storage battery plant. They found normal or slightly increased SCE
rates in workers with a PbB level of 1.4-3.6 µmol/litre (29-75 µg/dl)
and a ZPP level of 50-750 µmol/mol haemoglobin. The SCE increase was
correlated significantly with ZPP level.
Huang et al. (1988b) studied 21 lead-exposed workers from a
battery factory. They concluded that the SCE rate in workers with
long-term exposure to lead increased significantly when the mean PbB
level was 3.84 µmol/litre (80 µg/dl) or more. There were no
significant differences in SCE rates among the low- and medium-exposed
groups or the controls. These authors concluded that the effect of
chromosome damage caused by lead is not very strong.
Dalpra et al. (1983) examined blood samples taken from 19
children living in a contaminated area near a smelter and having PbB
levels in the range of 1.39-3.02 µmol/litre (29-63 µg/dl). They found
no effect on SCE frequency.
8.10 Carcinogenicity
Much has been written regarding the evidence that lead is
carcinogenic in humans (Moore & Meredith, 1979; Kazantzis, 1989;
Goyer, 1992). In several large epidemiological studies no association
was found which would associate lead with induction of cancer (Cooper
& Gaffey, 1975; Kang et al., 1980; Cooper, 1981; McMichael & Johnson,
1982). One major difficulty in many of the studies was the concurrent
exposure to potential carcinogens such as chromium (Davies, 1984), and
there has seldom been any attempt to deal with the primary etiological
agent (smoking) in the development of lung cancers associated with
lead exposure.
8.10.1 Occupational exposure and renal cancer
There are two case reports and very limited other epidemio-
logical evidence of an association between occupational exposure to
lead and renal cancer. Confounding variables, including use of tobacco
and exposure to other carcinogens, were not addressed (Baker et al.,
1980; Lilis, 1981; Selevan et al., 1985; Cantor et al., 1986).
The age-standardized mortality ratio for cancer was low in the
"lead poisoned" and other groups of workers from the Port Pirie study
by McMichael & Johnson (1982). The authors concluded that lead
poisoning did not increase the risk of cancer in humans.
8.10.2 Conclusion
Although some cohort mortality studies have indicated an
association between lead exposure and renal disease, there is no
association between renal cancer and lead in humans.
A Working Group convened by the International Agency for Research
on Cancer (IARC) in 1987 concluded that the evidence for the
carcinogenicity of lead and inorganic lead compounds in humans was
inadequate.
8.11 Effects on thyroid function
8.11.1 Occupational groups
Robins et al. (1983) found low values for serum thyroxine and
estimated free thyroxine in 7 of 12 workers having PbB levels above
2.11 µmol/litre (44 µg/dl). Both measures were correlated with PbB
level in a cross-sectional study of 47 foundry workers. Serum
thyrotropin and triiodothyronine levels were within the normal range
for both study groups.
No such relationship was found in a study by Refowitz (1984) of a
one-in-three random sample (N = 58) of male employees at a secondary
copper smelter. No thyroid abnormalities were observed and there was
no statistically significant relationship between PbB and serum
thyroxine or an estimate of free thyroxine in serum.
Tuppurainen et al. (1988) studied 176 African male workers in
Kenya and found that the duration of lead exposure correlated
negatively with serum free thyroxine and serum total thyroxine. The
correlation was strongest for the workers with the highest exposure
intensity over time. PbB data were available from periodic medical
examinations and average values (usually five determinations) were
used. The mean PbB was 2.73 µmol/litre (56.8 µg/dl), range
1.01-6.47 µmol/litre (21-135 µg/dl), and there was a mean exposure
duration of 7.6 years (range 1-20). The authors noted that current PbB
level, as a point determination, was not associated with total or free
thyroxine, triiodothyronine or thyrotropin in serum. They proposed
that long-term, highintensity exposure might be associated with
depressed thyroid function.
Gennart et al. (1992b) included assessment of thyroid function as
part of a study of lead-exposed workers. Data for serum levels of
triiodothyronine, thyroxine, free thyroxine index and thyroid-
stimulating hormone were within normal ranges for a group of 98
workers (mean PbB level, 2.45 µmol/litre or 51 µg/dl) and 85 controls
(1.00 µmol/litre or 20.9 µg/dl). The lead exposure of this study group
was considerably lower than in the workers in Kenya, and it was
suggested that thyroid function changes might not be indicators of
moderate exposure to lead.
8.11.2 Effects in children
Siegel et al. (1989) tested 68 children for thyroid function and
for PbB and found no statistically significant relationship between
lead and total or free thyroxine.
8.12 Immune system
In a review of the effects of lead on the immune responses of
experimental animals, Koller (1985) noted that there were few human
studies.
8.12.1 Occupational exposure
There is some evidence that lead workers with PbB levels in the
range of 1.0-4.10 µmol/litre (21-85 µg/dl) have increased
susceptibility to infections (colds and influenza) and have a
significant suppression of secretory IgA levels, a major factor in
defence against respiratory and gastrointestinal infections (Ewers et
al., 1982). There are also reports of impaired mitogen responses
(reflecting T-lymphocyte function) to phytohaemagglutinin (Jaremin,
1983) and of increased numbers of suppressor T-cells (Cohen et al.,
1989). However, on the basis of a study of 39 workers exposed to lead
oxide (mean PbB level of 1.84 µmol/litre or 38.5 µg/dl) and 21 control
subjects (mean PbB level of 0.57 µmol/litre or 11.8 µg/dl), Kimber et
al. (1986) concluded that chronic lead exposure in man is not
associated with the immunological changes that have been observed in
rodent studies.
Coscia et al. (1987) found increased B-lymphocyte percentage and
absolute count in workers currently exposed to lead with PbB levels
exceeding 2.4 µmol/litre (> 50 µg/dl).
8.12.2 Children
A study of 12 pre-school children (Reigart & Graber, 1976) with
PbB levels > 1.92 µmol/litre (40 µg/dl) did not reveal altered
immunity in comparison with a control group.
8.13 Effects on bone
The pharmacokinetics of the transfer of bone lead to other target
organs and blood has been discussed by Smith & Hursh (1977), Marcus
(1985a,b), Silbergeld et al. (1988) and Rabinowitz (1991) (see section
6.2.2).
Bone homoeostasis depends upon a complex interaction of its
various components, i.e. minerals, cells and the extracellular matrix
composed of collagenous and non-collagenous proteins (Marks & Popoff,
1988; Sauk & Somerman, 1991). Early work on the effects of lead on
calcium homoeostasis and calcium-mediated function was reviewed by
Pounds (1984). More recent work has been summarized by Pounds et al.
(1991), particularly the effects of lead on hormone action,
competition with calcium binding on calcium messenger systems, and the
impaired synthesis of collagen or sialoproteins. Additional evidence
for adverse effects of lead on the proteins in the mineral
compartment, thus affecting bone homoeostasis, has been reported by
Sauk & Somerman (1991).
8.14 Biomarkers for lead effects
The understanding and application of biomarkers for the
assessment of effect is becoming more widespread (IPCS, 1994).
It is desirable to identify biomarkers of lead exposure and
effect to provide easily measurable parameters that will facilitate
the risk assessment process. At present PbB levels are frequently
measured to assess both exposure and effect. Alternative biomarkers
for lead which may be easily measured are of biochemical effects,
particularly in the haem biosynthetic pathway.
However, the relationship between these effects and neurological
impairment caused by lead has not been established. Measurement of
these effects has also been used to provide biomarkers for exposure.
Anaemia has been associated with high levels of lead exposures in
the occupational setting. However, clinical tests for haemoglobin are
rather non-specific indicators of lead toxicity (Bernard & Becker,
1988). The steps within the haem biosynthetic pathway which have been
used to measure effect are: 1) inhibition of delta-aminolaevulinic
acid dehydratase (ALAD); 2) urinary excretion of delta-aminolaevulinic
acid (ALAU); (3) the accumulation of zinc protoporphyrin (ZPP) in
erythrocytes arising from the inhibition of the enzyme ferrochelatase
or the iron transport system.
There are several methodological problems related to the
measurement of ALAD, and the benefit of measuring enzyme activity in
blood in lieu of PbB determinations is not apparent (Mushak, 1989).
Early work (Roels et al., 1976) indicated enzyme inhibition at PbB
levels of 0.24 µmol/litre (> 5 µg/dl). However, the data at levels of
0.72 µmol/litre (< 15 µg/dl) appear scattered (Hernberg & Nikkanen,
1970; Granick et al., 1973).
Levels of ALA in urine have been used as an effect indicator of
lead exposure (Meredith et al., 1978). ALAU levels were found to
correlate with PbB levels as low as 0.864 µmol/litre (18 µg/dl), the
correlation becoming much stronger at PbB levels above 1.92 µmol/litre
(40 µg/dl) (Selander & Cramer, 1970). Given the present concern over
effects in children at PbB levels well below 1.92 µmol/litre
(40 µg/dl), the assay of ALAU concentrations may not be sensitive
enough to be of any value.
The effects of lead on porphyrin levels, both coproporphyrin in
urine and zinc protoporphyrin (ZPP) in blood, have been investigated
as possible biomarkers for lead. Measurement of urinary coproporphyrin
levels does not appear to be sensitive enough to be useful in
assessing exposure to lead at present environmental levels. Excretion
levels do not rise significantly until PbB levels exceed
1.92 µmol/litre (40 µg/dl) (Meredith et al., 1978). Other studies
indicate that this assay may not be sensitive enough for biological
monitoring at PbB levels between 0.96 and 1.20 µmol/litre (20 and
25 µg/dl). At present it would appear that ZPP has limited usefulness
as a biological indicator of exposure at exposures leading to PbB
levels lower than 1.20 µmol/litre (25 µg/dl) (Roels et al., 1976;
Piomelli et al., 1982; Hammond et al., 1985; Marcus & Schwartz, 1987).
The results may also be confounded by concurrent iron deficiency which
will alter the levels of ZPP.
Vitamin D metabolism involves cytochrome P-450-dependent enzymes
and thus may be affected by lead exposure. Measurement of serum levels
of 1,25-dihydroxy-vitamin-D has been proposed as a sensitive
biological monitor of lead exposure in children (Rosen et al., 1980;
Mahaffey et al., 1982; Koo et al., 1991). However, it should be noted
that dietary intakes of calcium and phosphorus, as well as circulating
levels of parathyroid hormone will regulate the production and
circulating concentrations of this vitamin D metabolite, thus making
correlations with PbB levels difficult without adequate information on
the nutritional status of the population under study (Rosen & Chisney,
1983). In populations showing adequate nutritional status, lead at
exposures leading to PbBs levels lower than 0.96 µmol/litre (20 µg/dl)
does not appear to have a demonstrable effect on circulating levels of
1,25-dihydroxy-vitamin-D.
9. EVALUATION OF HUMAN HEALTH RISKS
9.1 Exposure assessment
Lead is a ubiquitous element detected in all environmental media.
However, natural sources contribute only a small fraction of the
amounts of lead found in air, food, water and dust. The majority of
lead in these media arises from automobile and industrial emissions
and from the use of lead-containing solder and paints. Adults and
older children receive the largest proportion of lead intake from
foods, whereas dust, soil and food all make significant contributions
to the total lead intake of young children. The major contributions to
lead in soil and outdoor dust are from the combustion of fossil fuels
(principally leaded petrol), stationary sources such as smelters, and
peeling and flaking of lead-based paint.
9.1.1 General population exposure
In the absence of specific stationary sources of lead,
concentrations in ambient air are directly related to density of
traffic and whether lead is still utilized as an additive in petrol.
Reduction or elimination of lead in petrol in those countries which
have instituted regulations has resulted in a decline by as much as
eight-fold in ambient air concentrations of lead.
Levels of lead in indoor air are affected by the presence of
cigarette smoke and dust from lead-painted surfaces. Without such
sources, air lead levels indoors are about 60% of those in outdoor
air.
For most adults, the total daily exposure to lead is via food,
water and air. For infants aged up to 5 months, formula or breast milk
and water are the main sources of lead. In children, an additional
source of exposure is dust and soils. Absorption is dependent on the
chemical form of lead, type of soil and particle size
(bioavailability). Lead intake may be augmented from unusual sources
such as folk remedies, cosmetics and hobby activity. Community
contamination and workplace practices may contribute to lead exposure.
Food (including drinking-water and beverages) is the major source
of lead exposure for the general population. Infants and children may
receive an added lead burden from soil and dust. The most significant
foodstuffs will vary from country to country. In areas still utilizing
lead-soldered cans, levels of lead are substantially higher. Depending
upon lifestyles, there may be significant oral intake of lead from
some alcoholic beverages and due to the leaching of lead from low
temperature-fired ceramic containers.
Most drinking-water supplies contain lead levels lower than
5 µg/litre when they leave the treatment plant. However, where the
water is known to be plumbo-solvent, up to 40% of the samples may
exceed 100 µg/litre in homes where lead solder, lead pipes or brass
fixtures have been used.
Absorption of lead from the lung is a function of particle size
and pulmonary deposition pattern. Small particles (< 0.5 µm in
diameter) characteristic of ambient air will be deposited deeply in
the lungs with absorption rates of 90%. Larger particles, such as
those that may be encountered in occupational settings, exhibit high
deposition rates in the upper airway. Absorption of such particles
will be a function of both dissolution in the lung and particle
clearance to the gastrointestinal tract.
Human dermal absorption of inorganic lead through unabraded skin
is of limited significance.
9.1.2 Occupational exposures
In addition to exposure to lead in ambient air, water and food,
some workers may be exposed to airborne lead and dust within the
workplace. Actual levels will vary according to the engineering design
of the process equipment and the industrial hygiene practices
utilized.
9.2 Critical issues related to exposure evaluation
In view of the heterogeneity of responses to lead within human
populations, the complex interrelationships between exposure to lead
and a biological indicator for internal dose require consideration of
several key issues in order to assess human exposures.
9.2.1 Sampling and analytical concerns
Reliable comparisons of reported levels of exposure and/or dose
can only be made where authors have described the analytical and
sampling procedures in sufficient detail to allow the reader to
assess, for example, the integrity of the sampling procedures as well
as the specificity, precision and accuracy of all analytical methods.
Problems related to sampling cord blood also need to be considered.
Most studies have utilized analytical procedures of high quality.
However, consideration must be given to blood collection procedures
(finger stick versus venepuncture) when comparing results.
9.2.2 Data presentation
Inter-study comparisons of lead exposures are complicated by the
variety of methods used to present results, including median values as
well as geometric and arithmetic means. Some authors have used
log-transformed data.
In assessing exposure from data on teeth, one must know which
tooth and which compartment(s) within the tooth were sampled. In
addition, if data are to be compared between studies, authors must
state explicitly that all teeth analysed were without caries and were
shed spontaneously.
The exposure index lifetime average blood lead (PbB) level has
assisted in the assessment of cumulative exposures from serial PbB
data. It should not be interpreted as being equivalent to a single PbB
determination at a single point in life.
For some data/analytical purposes, age-specific PbB levels may be
more appropriate than a lifetime average.
9.3 Relationship between exposure and dose
The most widely used surrogate for the absorbed dose is whole PbB
concentration.
The relationship between PbB level and lead intake is curvilinear
over a wide range of PbB values. On the basis of a single study of 17
infants, the relationship between PbB level and lead intake from food
has been determined to be 0.0077 µmol lead/litre (0.16 µg/dl) per µg
lead intake per day for a median PbB level of approximately
0.48 µmol/litre (10 µg/dl).
Most studies of the relationship between PbB level and lead
exposure apply to a single environmental source, i.e. air, food, water
or soil/dust. A summary of the relationship between PbB level and lead
intake from individual media is given in Table 23.
9.4 Surrogate measures of dose
9.4.1 Blood
Whole PbB values are widely used as a measure of absorbed dose.
However, it is believed that plasma lead concentrations may better
reflect the "active" fraction of lead in blood and define the
relationship between PbB and tissue or organ accumulation (and
Table 23. Representative relationships of blood lead median level to intake of
lead for the general populationa
Population
Median Children Adults
Airb 0.09 µmol Pb/litre per µg 0.079 µmol Pb/litre per µg
Pb/m3 (1.92 µg Pb/dl) Pb/m3c (1.64 µg Pb/dl)
Water 0.003 µmol Pb/litre per µg
Pb/litre (0.06 µg Pb/dl)
Food 0.01 µmol Pb/litre per µg 0.002-0.003 µmol Pb/litre per
Pb/day (0.16 µg Pb/dl) µg Pb/day (0.04-0.06 µg Pb/dl)
Dustb 0.09 µmol Pb/litre per 1000 µg
Pb/g (1.8 µg Pb/dl)
Soilb 0.11 µmol Pb/litre per 1000 µg
Pb/g (2.2 µg Pb/dl)
a These data are provided for illustrative purposes only recognizing that the
relationships are curvilinear in nature and are broad guidelines which will
not apply at lower or higher levels of exposure.
b A value between 0.144 to 0.24 µmol Pb/litre or 3-5 µg Pb/dl per µg/m3
is obtained when one considers indirect contribution through deposition on
soil/dust.
c The air to blood lead relationship in occupational settings is best described
by a curvilinear relationship having slopes between 0.02 and 0.08 µg/m3
air. The slope is variable but lower than that found for humans in the general
environment, which is between 1.6 and 1.9 µg/m3.
effect), although there is little experimental data (because of
analytical limitations). PbB is distributed between plasma and
erythrocytes, with less than 5% being in the plasma. Most of the lead
is bound to haemoglobin.
Venous and capillary blood levels are generally equivalent,
provided that the sampling technique is adequate.
9.4.2 Urine
Urinary measurements of lead concentration are of limited value,
although they are used occasionally as a screening test for
occupational population groups.
9.4.3 Bone
Bone lead may be measured by non-invasive X-ray fluorescence, but
this technique is limited in sensitivity at present.
9.4.4 Tooth
Shed deciduous teeth have been used to provide an index of
exposure in early childhood. Interpretation of the analytical data is
dependent on the type of tooth and the part of the tooth (whole tooth,
dentine or circumpulpar dentine) analysed.
9.4.5 Hair
Hair is not useful for measurement of lead exposure.
9.5 Biochemical effects of lead
9.5.1 Haem synthesis
Evaluation of the quality of analytical data is an important
aspect in considering reports describing effects attributed to lead.
It should be noted that much of the data presented in this area has
not been as vigorously scrutinized as, for example, psychometric study
data.
An increase in erythrocyte protoporphyrin (EP) in children occurs
between PbB levels of 0.72 and 1.2 µmol/litre (15-25 µg/dl). Increases
in EP can be detected in men when the PbB level is above
1.20-1.44 µmol/litre (25-30 µg/dl), and in women when it is above
0.96-1.44 µmol/litre (20-30 µg/dl). It should be noted that the effect
of lead on haem is confounded by low iron status.
9.5.1.1 Urinary coproporphyrin
The coproporphyrin concentration in urine increases significantly
with PbB levels in excess of 1.92 µmol/litre (40 µg/dl).
9.5.1.2 Urinary aminolaevulinic acid in children
In children 1-5 years old, there is a linear relationship with
PbB in the range 1.2-3.6 µmol/litre (25-75 µg/dl). Data for children
with PbB levels of 0.24-1.92 µmol/litre (5-40 µg/dl) show essentially
no correlation with urinary aminolaevulinic acid (ALA) excretion.
Elevation of urinary ALA level is evident at a PbB level of about
1.68 µmol/litre (35 µg/dl).
9.5.1.3 Urinary aminolaevulinic acid in adults
Urinary excretion of ALA increases in men at PbB levels above
2.16 µmol/litre (45 µg/dl) and in women above 1.68 µmol/litre
(35 µg/dl).
9.5.1.4 delta-Aminolaevulinic acid dehydratase
There was a negative exponential relationship between PbB level
and delta-aminolaevulinic acid dehydratase (ALAD) activity in a
population of 10- to 13-year-old children with PbB levels in the range
of 0.19-1.97 µmol/litre (4.7-41 µg/dl). An effect was seen at a PbB
level of approximately 0.48 µmol/litre (10 µg/dl). There is an
apparent lack of a clearly defined threshold for lead inhibition of
ALAD in different age groups.
9.5.2 Vitamin D metabolism
In the presence of adequate nutritional status, PbB levels below
0.96 µmol/litre (20 µg/dl) appear to have no demonstrable effect on
circulating concentrations of 1,25-dihydroxy-vitamin-D. A PbB level
above 0.96 µmol/litre (20 µg/dl) is associated with a decrease in the
serum level of 1,25-dihydroxy-vitamin-D.
9.5.3 Dihydrobiopterin reductase
Inhibition of dihydrobiopterin reductase has been shown in humans
where the mean PbB level is as low as 0.48 µmol/litre (10 µg/dl).
A summary of some biochemical effects of lead is presented in
Table 24.
9.5.4 Haemopoietic system
9.5.4.1 Anaemia in adults
The estimated PbB associated with a decrease in haemoglobin
concentration is 2.40 µmol/litre (50 µg/dl).
9.5.4.2 Anaemia in children
Decreased haemoglobin levels in children occur at a PbB level of
approximately 1.92 µmol/litre (40 µg/dl). Anaemia, defined as a
haematocrit below 35%, is not found at a PbB level of less than
0.92 µmol/litre (20 µg/dl). The risk of having a haematocrit value
below 35% for a 1-year-old child is 2% at a PbB level of
Table 24. Biochemical effects of lead
Parameter Blood lead level above which the
biochemical effect is demonstrable with
current techniques
µmol/litre µg/dl
Protoporphyrin levels 0.96-1.44 20-30
Coproporphyrin levels 1.92 40
ALA urine levels 1.68 35
ALAD activity 0.48 10
1,25-dihydroxy-vitamin-D 0.96 20
Dihydrobiopterin reductase 0.48 10
0.96-1.87 µmol/litre (20-39 µg/dl); the contribution of iron
deficiency may account for a substantial proportion of this 2%.
Induction of anaemia is demonstrable at 1.92 µmol/litre (40 µg/dl).
9.5.4.3 Erythrocyte pyrimidine-5'-nucleotidase
A reduction of 20% or more in erythrocyte pyrimidine
-5'-nucleotidase activity is associated with PbB concentrations above
0.48 µmol/litre (10 µg/dl).
Effects of lead are demonstrable on a number of enzyme systems
and biochemical parameters. The PbB levels, above which effects are
demonstrable with current techniques for the parameters which may have
clinical significance, are all greater than 0.96 µmol/litre
(20 µg/dl). Some clinically insignificant effects on enzymes are
demonstrable at lower levels of PbB.
9.6 Nervous system
9.6.1 Adults
9.6.1.1 Central nervous system
With acute lead exposure resulting in a PbB level in excess of
3.84 µmol/litre (80 µg/dl), severe encephalopathy and/or coma may
occur. Central nervous system (CNS) symptoms are found in lead-exposed
adults when there is a history of several years of exposure to lead at
PbB levels that may not have exceeded 3.36 µmol/litre (70 µg/dl) and a
PbB level at the time of clinical assessment of at least
1.92 µmol/litre (40 µg/dl).
Impaired neurobehavioural test performance has been found in
lead-exposed workers. Changes in critical flicker fusion test have
been detected at a PbB level of about 2.4 µmol/litre (50 µg/dl).
Sensory motor function is generally more sensitive than cognitive
end-points in many neurobehavioural evaluations, the lowest-observed-
effect level being at about 1.92 µmol/litre (40 µg/dl).
It appears also that neuroelectrophysiological tests are
sensitive indicators of the CNS effects of lead. Reductions in
latencies of sensory evoked potentials and auditory event-related
potentials have been found in workers with average PbB levels of
approximately 1.92 µmol/litre (40 µg/dl).
9.6.1.2 Peripheral nervous system
Numerous studies have measured the conduction velocity of
electrically stimulated sensory and motor nerves in workers exposed to
lead. These nerve conduction velocity (NCV) studies have yielded
somewhat mixed results, with many showing a decrease in NCV in
relation to lead exposure (indexed as PbB level) and a few showing no
effect or occasionally even an increase in NCV associated with lead
exposure. Differences in the nerves evaluated, methodologies,
characterization of lead exposure, and control of confounding
variables underlie some of the variability in results across studies.
A statistical meta-analysis of 32 NCV studies has indicated that NCV
is significantly reduced in lead-exposed workers compared to reference
subjects, but that the median motor nerve shows more reliable effects
of lead than other nerves. This collective view of the evidence is
supported by key studies that provide compelling evidence of a causal
relationship between lead exposure and reductions in NCV, extending to
PbB levels as low as 1.44 µmol/litre (30 µg/dl). These effects may be
reversible depending on the duration and level of exposure.
9.6.1.3 Autonomic nervous system
Two reports examining the electrocardiographic RR interval
variability during deep breathing and the component CV of respiratory
sinus arrythmia demonstrated dysfunctions at an average PbB level of
1.68 µmol/litre (35 µg/dl). These results suggest autonomic nervous
system dysfunction, particularly the parasympathetic nervous system.
9.6.2 Children
Prospective and cross-sectional studies of children have
demonstrated associations of lead exposure, measured by various
indices, and intellectual performance. The association has been noted
across a wide range of exposure levels and in a variety of populations
before factors other than lead have been accounted for.
A key question is whether this statistical association is
directly attributable to the causal influence of lead on child IQ. It
is important to consider alternative contributory explanations, i.e.
random chance, unexplained confounding factors, reverse causality and
selection bias.
It is a matter of debate and conjecture as to the extent to which
these four issues should inhibit claims of a causal relationship in
the epidemiological studies. The essential problem is that
observational epidemiology cannot provide definitive evidence of
causality when the key statistical association is small, the temporal
relationship is unclear and major confounders are present. Animal
studies provide qualitative support for the claim of a causal role for
lead in affecting neuropsychological performance, but provide limited
assistance in establishing quantitative dose-effect relationships.
9.6.2.1 Type of effect
The clearest and most consistent associations have been found
with global measures, such as IQ, where the largest body of evidence
is available. Efforts to delineate the neuropsychological foundations
of this association with a wide variety of tests of specific
neuropsychological domains have not so far been successful.
9.6.2.2 Magnitude
Based on the evidence from cross-sectional and prospective
studies of populations with PbB levels generally below 1.2 µmol/litre
(25 µg/dl), the size of the apparent IQ effect (at ages 4 and above)
is a deficit of between 0 and 5 points (on a scale of 100 with a
standard deviation of 15) for each 0.48 µmol/litre (10 µg/dl)
increment in PbB level, with a likely apparent effect size of between
1 and 3 points. At PbB levels above 1.2 µmol/litre (25 µg/dl), the
relationship between PbB level and IQ may differ. Estimates of effect
size are group averages and only apply to the individual child in a
probabilistic manner.
Existing epidemiological studies do not provide definitive
evidence of a threshold. Below the PbB range of 0.48-0.72 µmol/litre
(10-15 µg/dl), the effect of confounding variables and limits in the
precision of analytical and psychometric measurements increases the
uncertainty attached to any estimate of effect. However, there is some
evidence of an association below this range.
9.6.2.3 Reversibility/persistence
Whilst the IPCS Task Group could not unequivocally state that
effects of early childhood exposure are persistent beyond childhood,
because the current data are too meagre, it was held that
neurobehavioural effects detected at age seven or later usually
persist. Measures in later childhood tend to be more predictive of
subsequent performance than those made earlier. It is more likely than
not that effects seen during school years are to some degree
irreversible. This has also been observed in later follow-up studies
conducted in other non-lead topics of child development research. One
of the difficulties is that there are too few studies concerning
long-term outcome in children with high early exposures and where the
sources of exposure are subsequently removed.
Virtually no useful data are available on the effects on IQ of
removing children from a "high" exposure environment to one of "low"
exposure or on reduction of body lead burden in children. This is not
to say that exposure should not be reduced when possible.
9.6.2.4 Age-specific sensitivity
From prospective studies it is not possible to determine an age
of critical sensitivity. This reflects the findings that serial PbB
measures taken at the age of 2 years and later are positively
correlated with, the individual rankings remaining approximately
constant, and this limits the ability to identify sensitive periods of
exposure.
9.6.2.5 Interactions/subgroups
The evidence is inconclusive on whether apparent effects are more
or less marked in different gender or socioeconomic status (SES)
subgroups. However, where there are suggestions of SES-related
differences, the apparent effects tend to be more marked in the lower
SES subgroups.
9.6.3 Animal studies
Experimental animal studies of CNS effects provide support for
the associations between PbB levels and neurobehavioural deficits
described in human epidemiological studies of lead. There is
supportive evidence both in terms of demonstrating causal
relationships and in the levels of PbB at which such effects are
observed, namely 0.528-0.72 µmol/litre (11-15 µg/dl). Moreover, they
provide qualitative parallels in the nature of the effects described,
as these effects include changes in learning and memory functions.
Experimental animal studies indicate that these CNS effects may depend
upon task complexity and can persist long beyond the termination of
lead exposure. These studies also provide information possibly
relevant to understanding mechanisms of effect. In addition, the
experimental animal studies provide such evidence in the absence of
the confounding factors and co-variates, such as parental IQ,
socioeconomic status, and quality of the home environment, that are
problematic to human epidemiological endeavours, and in the absence of
nutritional deficiencies that may arise in human populations.
9.7 Renal system
Renal function impairment was not associated with a PbB level
below 3.0 µmol/litre (62 µg/dl) when measured by blood urea nitrogen
and serum creatinine levels in lead workers. However, renal tubular
effects were detected in workers with a PbB level below 3.0 µmol/litre
when measured by more sensitive indicators such as NAG.
Most studies of the general population attempting to relate renal
function impairment to PbB concentration have not demonstrated an
effect with PbB levels below 1.8 µmol/litre (37.3 µg/dl). More
sensitive indicators of renal function may indicate a renal effect of
lead below this level.
9.8 Liver
Over-exposure to lead may inhibit drug metabolism in the liver.
9.9 Reproduction
9.9.1 Female
Studies on the risk of spontaneous abortion and reduced birth
weight associated with maternal PbB levels below 1.44 µmol/litre
(30 µg/dl) have yielded mixed results. Recent epidemiological studies
have shown exposure-related perturbations in the length of gestation,
significantly greater risks being associated with PbB levels of
0.72 µmol/litre (15 µg/dl) or more.
9.9.2 Male
PbB concentrations above 1.92 µmol/litre (40 µg/dl) have been
shown to affect sperm morphology and function. At present, the
reproductive consequences of these changes are unknown.
9.10 Blood pressure
A quantitative assessment of the collective evidence from all the
observational studies in adults is made difficult by the fact that
studies have adopted different policies regarding adjustment for
potential confounding factors (e.g., alcohol consumption). In
addition, quantitative findings from the two largest studies (BRHS and
NHANES II) have depended on whether adjustment was made for
geographical variations in blood pressure and blood lead.
The limited size of most observational studies has inevitably
meant that they could not consistently demonstrate a statistically
significant relationship. However, an overview of all the studies
shows that evidence is consistent with the centre-adjusted analysis of
the two main studies, i.e. there are very weak but statistically
significant associations between PbB level and both systolic and
diastolic blood pressures. The likely order of magnitude is that for
any two-fold increase in PbB level (e.g., from 0.8 to 1.6 µmol per
litre) there is a mean 1 mmHg increase in systolic blood pressure. The
association with diastolic blood pressure is of a similar magnitude.
Animal studies have provided plausible mechanisms for an effect
of lead on blood pressure. However, from such a small magnitude of
statistical associations in the presence of important confounders, one
cannot infer that low-level lead exposure is causally related to an
increase in blood pressure.
The two population studies relating PbB to cardiovascular disease
events show no statistically significant associations. Hence, there is
no clear evidence to suggest that lead has an impact of public health
importance as regards hypertension or risk of cardiovascular disease.
9.11 Carcinogenicity
Renal tumours occur in rats and mice administered high doses of
lead. However, the evidence for the carcinogenicity of lead and
inorganic lead compounds in humans is inadequate.
9.12 Immune system
There is no strong evidence in humans of an effect of lead on the
immune system.
10. RECOMMENDATIONS FOR PROTECTION OF HUMAN HEALTH
10.1 Public health measures
Public health measures should be directed towards reduction and
prevention of exposure to lead by reducing the use of lead and lead
compounds and by minimizing lead-containing emissions that result in
human exposures. This can be achieved by:
a) phasing-out any remaining uses of lead additives in motor fuels;
b) further reducing the use of lead-based paints, with the objective
of eliminating such paints;
c) development and application of methods for the safe and
economical remediation of lead-painted homes and lead
contaminated soil;
d) elimination of the use of lead in food containers (e.g., in the
seams of cans);
e) dissemination of information to assist with identification of
glazed food containers which may leach lead into food placed,
cooked or stored in the container;
f) eliminating any remaining agricultural uses of lead or lead
compounds (e.g., lead arsenate as an insecticide);
g) identifying and reducing, or preferably, eliminating lead found
as a contaminant or ingredient of folk remedies and cosmetics;
h) the use of materials and engineering practices to minimize
plumbosolvency in water treatment and water distribution systems;
i) systematic examination of processes in which lead is used or
recycled in order to identify and reduce lead exposure by means
of improved engineering design, of operators, by-standers and the
environment. Opportunities for technology transfer should be used
whenever possible.
10.2 Public health programmes
Programmes should be developed:
a) to enhance data collection and to make available to the public
information on the lead content of foodstuffs;
b) to facilitate identification of populations at high risk of
exposure to lead on the basis of monitoring data for lead in
food, air, water and soil;
c) that incorporate improved procedures for health risk assessment
of population groups at risk of exposure to lead;
d) that promote understanding and awareness concerning the effects
on human health associated with exposure to lead, while
recognizing cultural sensitivities;
e) that place emphasis on adequate nutrition, health care and
attention to socioeconomic conditions which may exacerbate the
effects of lead present in the environment.
10.3 Screening, monitoring and assessment procedures
Methods for evaluating the effects and associated risks of
exposure to lead require both improvement and further development or
research. In the short term the following measures are needed.
a) Screening
i) Blood lead measurements should be recognized as the biomarker of
choice for screening for previous exposure of children to lead.
ii) The sensitivity of the developing nervous system to the
potentially harmful effects of lead is such that other
biochemical measurements (e.g., erythrocyte protoporphyrin) are
not sufficiently sensitive for assessment of infants and
children.
b) Monitoring
i) More sensitive analytical methods should be developed for the
reliable measurement of blood lead levels below 0.72 µmol/litre
(15 µg/dl) to acceptable standards of precision and accuracy.
ii) There is a need for international analytical quality assurance
programmes utilizing reference lead-containing materials.
iii) All publications containing blood lead measurement data should
provide adequate data on current quality assurance and quality
control.
iv) Data comparisons are made more difficult by differences in units
and statistical techniques for data handling. Investigators
should be encouraged to adopt internationally agreed practices
(e.g., IUPAC units).
c) Assessment
i) Validated biomarkers are needed to define the relationship
between measures of environmental (external) exposure and
specific effects (biochemical or functional).
ii) Biomarkers indicative of deficits in cognitive performance are
needed to facilitate assessment of risk.
iii) Improved methods are needed for the definition of outcome effects
(especially IQ and neurobehavioural deficits) attributable to
lead at blood lead concentrations of about
0.48 µmol/litre (10 µg/dl) or less.
iv) Further data are required to determine whether outcome effects on
the nervous system attributed to lead are reversible or
permanent.
v) Biomarkers for the renal effects of lead are needed to link renal
damage with lead exposure and thereby improve assessment of risk
of renal damage.
11. FURTHER RESEARCH
a) Research is required to improve the process for the assessment of
risk associated with exposure to lead. Specifically this should
cover:
i) definition of the health significance of biochemical
changes associated with exposure to lead, with particular
attention to alterations associated with blood lead
concentrations of about 0.72 µmol/litre (15 µg/dl) or
less;
ii) work to define the bioavailability of lead from different
sources and to establish the relationship between exposure
(sources and speciation) and body burden;
iii) definition of the influence of host-related factors
(particularly nutrition) affecting absorption and
distribution of lead;
iv) intensification of kinetic studies of lead to provide an
improved data base for extrapolation between species;
v) elucidation of mechanisms of accumulation and mobilization
of lead from bone with particular attention to the
influence of pregnancy and ageing on kinetics;
vi) investigation of the pharmacokinetics of lead in the
pregnant female in relation to transfer of lead to the
developing embryo and fetus and factors that mitigate such
transfer;
vii) determination of the effects of pre- and post-natal
exposure to lead;
viii) improved definition of paternally mediated effects of lead
exposure on the reproductive process and outcomes.
b) More general research needs include the following:
i) rationalization of neurobehavioural tests used to assess
performance in children and animals in order to permit
comparisons of measurements and test data that use similar
biological mechanisms;
ii) follow-up research (where possible) of historical cohorts
of lead-poisoned adults to examine the sequelae;
iii) evaluation of intervention measures, such as lead
abatement from home and soil, and chelation on reduction
of blood lead levels and outcome effects on the central
nervous system.
12. PREVIOUS EVALUATIONS BY INTERNATIONAL BODIES
The carcinogenic potential of lead and lead compounds was last
evaluated by the International Agency for Research on Cancer in 1987
(IARC, 1987b). There was inadequate evidence for the carcinogenicity
of lead and inorganic lead compounds in humans, but sufficient
evidence was available to show that specified inorganic lead compounds
were carcinogenic in experimental animals. The overall evaluation
placed lead and inorganic lead compounds in Group 2B, i.e. possibly
carcinogenic to humans.
To protect workers from the adverse effects of lead on haem
synthesis and on the peripheral and central nervous system, a
health-based biological exposure limit of 1.92 µmol/litre (40 µg/dl)
was recommended by a WHO Study Group (WHO, 1980). It was further
recommended that blood lead (PbB) levels in women within the
reproductive age range should not exceed 1.44 µmol/litre (30 µg/dl).
Depending upon the background levels of PbB in the worker population,
air lead levels should not exceed 30-60 µg/m3 (WHO, 1980).
A drinking-water guideline value of 0.050 mg/litre was developed
in 1984 (WHO, 1984). This guideline value has been revised recently to
0.01 mg/litre (WHO, 1993).
Lead was evaluated by a WHO Working Group developing air quality
guidelines for Europe (WHO, 1987). Based on the assumption that PbB
levels in 98% of the population would be maintained at levels below
0.96 µmol/litre (20 µg/dl), a guideline value in the range of
0.5-1.0 µg/m3 (long-term average, such as annual mean) was
recommended.
At the 41st meeting of the Joint FAO/WHO Expert Committee on Food
Additives and Food Contaminants, a Provisional Tolerable Weekly Intake
(PTWI) of 25 µg/kg body weight was recommended (FAO/WHO, 1993). This
level refers to lead from all sources and was set to protect all
humans, including infants and children. It was based on a model
indicating daily intakes of lead between 3-4 µg/kg body weight by
infants and children and is not associated with an increase in PbB
concentrations.
Regulatory standards established by national bodies in several
countries and the European Economic Community are summarized in the
legal file of the International Register of Potentially Toxic
Chemicals (IRPTC, 1987).
REFERENCES
Aguilera de Benzo Z, Fraile R, & Carrion N (1989) Determination of
lead in whole blood by electrothermal atomization atomic absorption
spectrometry using tube and platform atomizers and dilution with Trito
X-100. J Anal At Spectrom, 4: 397-400.
Albahary C (1972) Lead and hemopoiesis: The mechanism and consequences
of the erythropathy of occupational lead poisoning. Am J Med,
52: 367-378.
Albert LA & Badillo F (1991) Environmental lead in Mexico. Rev Environ
Contamin Toxicol, 117: 1-49.
Alexander FW, Delves HT, & Clayton BE (1973) The uptake and excretion
by children of lead and other contaminants. In: Barth D, Berlin A,
Engel R, Recht P, & Smeets J ed. Environmental health aspects of lead:
Proceedings, International Symposium, Amsterdam, The Netherlands,
October 1972. Luxembourg, Commission of the European Communities,
pp 319-331.
Al-Hakkak ZS, Hamamy HA, Murad AMB, & Hussain AF (1986) Chromosome
aberrations in workers at a storage battery plant in Iraq. Mutat Res,
171: 53-60.
Altmann L, Weinsberg F, Sveinsson K, Lilienthal H, Wiegand H, &
Winneke G (1993) Impairment of long-term potentiation and learning
following chronic lead exposure. Toxicol Lett, 66(1): 105-112.
Alvares AP, Kapelner S, Sassa S, & Kappas A (1975) Drug metabolism in
normal children, lead-poisoned children, and normal adults. Clin
Pharmacol Ther, 17: 179-183.
Anderson C & Danylchuk KD (1977) The effects of chronic low level lead
intoxication on the Haversian remodeling system in dogs. Lab Invest,
37: 466-469.
Angle CR & McIntire MS (1978) Low level lead inhibition of erythrocyte
pyrimidine nucleotidase. Environ Res, 17: 296-302.
Angle CR & McIntire MS (1979) Environmental lead and children: the
Omaha study. J Toxicol Environ Health, 5: 855-870.
Angle CR, McIntire MS, & Swanson MS (1982) Erythrocyte nucleotides in
children - increased blood lead and cytidine triphosphate. Pediatr
Res, 16: 331-334.
Annest JL & Mahaffey KR (1984) Blood lead levels for persons aged 6
months-74 years: United States, 1976-1980. Hyattsville, Maryland, US
Public Health Service (Report No. 223, Series 11) (DHHS Publication
No. (PHS) 84-1683).
Annest JL, Pirkle JL, Makuc D, Neese JW, Bayse DD, & Kovar MG (1983)
Chronological trend in blood lead levels between 1976 and 1980. N Engl
J Med, 308: 1373-1377.
Araki S & Ushio K (1982) Assessment of the body burden of chelatable
lead: a model and its application to lead workers. Br J Ind Med,
39: 157-160.
Araki S, Honma T, Yanagihara S, & Ushio K (1980) Recovery of slowed
nerve conduction velocity in lead-exposed workers. Int Arch Occup
Environ Health, 46: 151-157.
Araki S, Aono H, Fukahori M, & Tabuki K (1984) Behaviour of lead and
zinc in plasma, erythrocytes, and urine and ALAD in erythrocytes
following intravenous infusion of CaEDTA in lead workers. Arch Environ
Health, 39: 363-367.
Araki S, Murata K & Aono H (1986a) Subclinical cervico-spino-bulbar
effects of lead: A study of short-latency somatosensory evoked
potentials in workers exposed to lead, zinc and copper. Am J Ind Med,
10: 163-175.
Araki S, Yokoyama K, Murata K, & Aono H (1986b) Determination of the
distribution of conduction velocities in workers exposed to lead, zinc
and copper. Br J Ind Med, 43: 321-326.
Araki S, Aono H, Murata K (1986c) Mobilisation of heavy metals into
the urine by CaEDTA: Relation to erythrocyte and plasma concentrations
and exposure indicators. Br J Ind Med, 43: 636-641.
Araki S, Murata K, & Aono H (1987) Central and peripheral nervous
system dysfunction in workers exposed to lead, zinc and copper: A
follow-up study of visual and somatosensory evoked potentials. Int
Arch Occup Environ Health, 59: 177-187.
Araki S, Murata K, Yokoyama K, & Uchida E (1992) Auditory
event-related potential (P300) in relation to peripheral nerve
conduction in workers exposed to lead, zinc, and copper: Effects of
lead on cognitive function and central nervous system. Am J Ind Med,
21: 539-547.
Arnetz BB & Nicolich MJ (1990) Modelling of environmental lead
contributors to blood lead in humans. Int Arch Occup Environ Health,
62: 397-402.
Aslam M, David SS, & Healy MA (1979) Heavy metals in some Asian
medicines and cosmetics. Publ Health (Lond), 93: 274-284.
Assenato G, Paci C, Baser M, Molinini R, Candela RG, Altamura BM, &
Giorgino R (1987) Sperm count suppression without endocrine
dysfunction in lead-exposed men. Arch Environ Health, 42: 124-127.
ATSDR (1993) Toxicological profile for lead. Atlanta, Georgia, US
Department of Health and Human Services, Agency for Toxic Substances
and Disease Registry (TP-92/12).
Aungst BJ, Docle JA, & Fung H (1981) The effect of dose on the
disposition of lead in rats after intravenous and oral administration.
Toxicol Appl Pharmacol, 61: 48-57.
Awad el Karim MA, Hamed AS, Elhaimi YAA, & Osman Y (1986) Effects of
exposure to lead among lead acid battery factory workers in Sudan.
Arch Environ Health, 41: 261-265.
Azar A, Trochimowicz HJ, & Maxfield ME (1973) Review of lead studies
carried out at Haskell Laboratory: two year feeding study and response
to hemorrhage study. In: Environmental health aspects of lead.
Proceedings of an international symposium, Amsterdam. Luxembourg,
Commission of the European Communities, pp 199-210.
Baghurst PA, McMichael AJ, Wigg N, Vimpani G, Robertson EF, Roberts
RJ, & Tong S-L (1992) Life-long exposure to environmental lead and
children's intelligence at age seven: the Port Pirie cohort study.
N Engl J Med, 327: 1269-1284.
Baker EL, Landrigan PJ, Barbour AG, Cox DH, Folland DS, Ligo RN, &
Throckmorton J (1979) Occupational lead poisoning in the United
States: clinical and biochemical findings related to blood lead
levels. Br J Ind Med, 36: 314-322.
Baker EL, Goyer RA, Fowler BA, Khettry U, Bernard DB, Adler S, White
R, Babayan R, & Feldman RG (1980) Occupational lead exposure,
nephropathy and renal cancer. Am J Ind Med, 1: 139-148.
Barrio A & Badia J (1985) [Lead encephalopathy in adults.] Rev Med
Chil, 113(6): 564-567 (in Spanish).
Barltrop D (1969) Mineral metabolism in pediatrics. Oxford, London,
Edinburgh, Melbourne, Blackwell Scientific Publishers.
Barltrop D (1972) Children and environmental lead. In: Hepple P ed.
Lead in the environment: Proceedings of a conference. London,
Institute of Petroleum, pp 52-60.
Barry PSI (1975) A comparison of concentrations of lead in human
tissues. Br J Ind Med, 32: 119-139.
Barry PSI (1981) Concentrations of lead in the tissues of children.
Br J Ind Med, 38: 61-71.
Barton JC, Conrad ME, Nuby S, & Harrison L (1978) Effects of iron on
the absorption and retention of lead. J Lab Clin Med, 92: 536-547.
Bauchinger M, Dresp J, Schmid E, Englert N, & Krause C (1977)
Chromosome analyses of children after ecological lead exposure. Mutat
Res, 56: 75-79.
Beattie AD, Moore MR, Goldberg A, Finlayson MJ, Graham JR, Mackie TM,
Main J, McLaren PA, Murdock KM, & Stewart FT (1975) Role of chronic
lead exposure in the aetiology of mental retardation. Lancet,
1: 589-592.
Beckett PH, Davis RD, & Brindley P (1979) The disposal of sewage
sludge onto farmland: the scope of the problem of toxic elements.
Water Pollut Control, 78: 419-436.
Behari LR, Singh S, Tandon SK, & Wahal AK (1982) Lead poisoning among
Indian silver jewellery makers. Ann Occup Hyg, 27: 107-109.
Behne D (1980) Problems of sampling and sample preparation for trace
element analysis in the health sciences. In: Bratter P & Schramel P
ed. Trace element analytical chemistry in medicine and biology.
Proceedings of the First International Workshop, Neuherberg, Federal
Republic of Germany. Berlin, New York, Walter de Gruyter & Co.,
pp 769-782.
Beijer K & Jernelöv A (1984) Microbial methylation of lead. In:
Grandjean P ed. Biological effects of organolead compounds. Boca
Raton, Florida, CRC Press, pp 13-19.
Bellinger DC & Stiles KM (1993) Epidemiologic approaches to assessing
the developmental toxicity of lead. Neurotoxicology, 14: 151-160.
Bellinger DC, Needleman HL, Leviton A, Waternaux C, Rabinowitz MR, &
Nichols ML (1984) Early sensory-motor development and prenatal
exposure to lead. Neurobehav Toxicol Teratol, 6: 387-402.
Bellinger DC, Leviton A, Waternaux C, Needleman H, & Rabinowitz M
(1987) Longitudinal analyses or prenatal and post-natal lead exposure
and early cognitive development. N Engl J Med, 316: 1037-1043.
Bellinger D, Sloman A, Leviton A, Rabinowitz M, Needleman H, &
Waternaux C (1991a) Low-level lead exposure and children's cognitive
function in the preschool years. Pediatrics, 87: 219-227.
Bellinger D, Leviton A, Rabinowitz M, Allred E, Needleman H,
Schoenbaum S (1991b) Weight gain and maturity in fetuses exposed to
low levels of lead. Environ Res, 54: 151-158.
Bellinger DC, Stiles KM, & Needleman HL (1992) Low-level lead
exposure, intelligence and academic achievement: A long-term follow-up
study. Pediatrics, 90: 855-861.
Berlin A & Schaller KH (1974) European standardized method for the
determination of delta-aminolevulinic acid dehydratase activity in
blood. Z Klin Chem Klin Biochem, 12: 389-390.
Bernard SR (1977) Dosimetric data and metabolic model for lead. Health
Phys, 32: 44-46.
Bernard BP & Becker CE (1988) Environmental lead exposure and the
kidney. Clin Toxicol, 26: 1-34.
Bernstein J, Evan AP, & Gardner KD (1987) Epithelial hyperplasia in
human polycystic kidney disease. Its role in pathogenesis and risk of
neoplasia. Am J Pathol, 129: 92-101.
Bert JL, van Dusen LJ, & Grace JR (1989) A generalized model for the
prediction of body lead burdens. Environ Med, 48: 117-127.
Beyer WN & Cromartie EJ (1987) A survey of Pb, Cu, Zn, Cd, Cr, As, and
Se in earthworms and soil from diverse sites. Environ Monit Assess,
8: 27-36.
Biggins PDE & Harrison RM (1979) Atmospheric chemistry of automotive
lead. Environ Sci Technol, 13: 558-565.
Billick IJ & Gray VE (1978) Lead based paint poisoning research:
review and evaluation 1971-1977. Washington, DC, US Department of
Housing and Urban Development.
Bishop L & Hill WJ (1983) A study of the relationship between blood
lead levels and occupational air lead levels. Am Stat, 37(4): 471-475.
Blair JA, Hilburn ME, Leeming RJ, McIntosh MJ, & Moore MR (1982) Lead
and tetrahydrobiopterin metabolism: possible effects on IQ. Lancet,
April 24, 964.
Body PE, Inglis GR, & Mulcahy DE (1988) Lead contamination in Port
Pirie South Australia. Adelaide, Australia, South Australia Department
of Environment and Planning (SADEP Report No. 101).
Bolger PM, Carrington CD, Capar SG, & Adams MA (1991) Reductions in
dietary lead exposure in the United States. Chem. Speciation
Bioavailab, 3(314): 31.
Bolter E, Butz T, & Arseneau JF (1975) Mobilization of heavy metals by
organic acids in the soils of a lead mining and smelting district.
In: Proceedings of the IXth Annual Conference on Trace Substances in
Environmental Health, Columbia, Missouri, 10-12 June 1975, pp 107-112.
Bondarenko GP (1968) An experimental study of the solubility of galena
in the presence of fulvic acids. Geochem Int, 5: 525-531.
Bornschein RL (1985) Influence of social factors on lead exposure and
child development. Environ Health Perspect, 62: 343-351.
Bornschein RL & Rabinowitz MB (1985) Proceedings of the Second
International Conference on Prospective Lead Studies. Environ Res,
38: 1-210.
Bornschein RL, Succop P, Dietrich KN, Clark CS, Que Hee S, & Hammond
PB (1985) The influence of social and environmental factors on dust
lead, hand lead, and blood lead levels in young children. Environ Res,
38: 108-118.
Bornschein RL, Succop PA, Krafft KM, Clark CS, Peace B, & Hammond PB
(1986) Exterior surface dust lead, interior house dust lead and
childhood lead exposure in an urban environment. In: Hemphill DD ed.
Trace substances in environmental health II. Columbia, Missouri,
University of Missouri, pp 322-332.
Bornschein RL, Succop PA, Krafft, KM, Clark CS, Peace B, & Hammond PB
(1987) Exterior surface dust lead, interior house dust lead and
childhood lead exposure in an urban environment. Trace Subst Environ
Health, 20: 322-332.
Bornschein RL, Grote J, Mitchell T, Succop PA, Dietrich KN, Krafft KM,
& Hammond PB (1989) Effects of prenatal lead exposure on infant size
at birth. In: Smith MA, Grant LD, & Sors AI ed. Lead exposure and
child development: An international assessment. Dordrecht, London,
Kluwer Academic Publishers, pp 307-319.
Boscolo P & Carmignani M (1988) Neurohumoral blood pressure regulation
in lead exposure. Environ Health Perspect, 78: 101-106.
Boutron CF, Görlach U, Candelone J-P, Bolshov MA, & Delmas RJ (1991)
Decrease in anthropogenic lead, cadmium and zinc in Greenland snows
since the late 1960s. Nature (Lond), 353: 153-156.
Braunstein GD, Dahlgren J, & Loriaux DL (1987) Hypogonadism in
chronically lead-poisoned men. Infertility, 1: 33-51.
Brown KW & Black SC (1983) Quality assurance and quality control data
validation procedures used for the Love Canal and Dallas lead soil
monitoring programs. Environ Monit Assess, 3: 113-122.
Brunekreef B (1984) The relationship between air lead and blood lead
in children: A critical review. Sci Total Environ, 38: 79-123.
Brunekreef B (1986) Childhood exposure to environmental lead. London,
United Kingdom, Monitoring and Assessment Research Centre.
Buchet J-P, Lauwerys R, Roels H, & Hubermont G (1977) Mobilization of
lead during pregnancy in rats. Int Arch Occup Environ Health,
40: 33-36.
Buchet J-P, Roels H, Bernard A, & Lauwerys R (1980) Assessment of
renal function of workers exposed to inorganic lead, cadmium, or
mercury vapour. J Occup Med, 22: 741-750.
Buchet JP, Lauwerys R, Vandervoorde A, & Pycke JM (1983) Oral daily
intake of cadmium, lead, manganese, copper, chromium, mercury,
calcium, zinc, and arsenic in Belgium: a duplicate meal study. Food
Chem Toxicol, 21: 19-24.
Bull RJ (1980) Lead and energy metabolism. In: Singhal PL & Thomas JA
ed. Lead toxicity. Baltimore, Maryland, Urban Schwarzenberg, Inc.,
pp 119-168.
Bushnell PJ & Bowman RE (1979a) Reversal learning deficits in young
monkeys exposed to lead. Pharmacol Biochem Behav, 10: 733-742.
Bushnell PJ & Bowman RE (1979b) Persistence of impaired reversal
learning in young monkeys exposed to low levels of dietary lead.
J Toxicol Environ Health, 5: 1015-1023.
Bushnell PJ & DeLuca H (1983) The effects of lactose on the absorption
and retention of dietary lead. J Nutr, 113: 365-378.
Bushnell PJ & Levin ED (1983) Effects of zinc deficiency on lead
toxicity in rats. Neurobehav Toxicol Teratology, 5: 283-288.
Bushnell G, Brockhaus A, & Baltissen R (1977a) Neurobehavioural and
system effects of long-term blood lead elevation in rats. Arch
Toxicol, 37: 247-263.
Bushnell PJ, Allan JR, & Marlar RJ (1977b) Scotopic vision deficits in
young monkeys exposed to lead. Science, 196: 333-335.
Byers RK & Lord EE (1943) Late effects of lead poisoning on mental
development. Am J Dis Child, 66: 471-494.
Calder IC, Collings MT, & Heyworth JS (1990) Evaluation of soil lead:
blood lead relationship for Port Pirie. Proceedings of the 23rd Annual
Conference of Trace Substances in Environmental Health. Environ
Geochem Health, 12(Suppl): 81-91.
Campbell BC, Beattie AD, Moore MR, Goldberg A, & Reid AG (1977) Renal
insufficiency associated with excessive lead exposure. Br Med J,
1: 482-485.
Cantor KP, Sontag JM, & Held MF (1986) Patterns of mortality among
plumbers and pipefitters. Am J Ind Med, 10: 73-89.
Capar SG & Rigsby EJ (1989) Survey of lead in canned evaporated milk.
J Assoc Off Anal Chem, 72: 378-381.
Cardenas A, Roels H, Bernard AM, Barbon R, Buchet JP, Lauwerys RR,
Rosello J, Ramis I, Mutti A, Franchini I, Fels LM, Stolte H, de Broe
ME, Nuyts GD, Taylor SA, & Price RG (1993) Markers of early renal
changes induced by industrial pollutants. II. Application to workers
exposed to lead. Br J Ind Med, 50: 28-36.
CDC (1985) Preventing lead poisoning in young children. Atlanta,
Georgia, US Department of Health and Human Services, Public Health
Service, Centers for Disease Control, pp 7-19 (Publication
No. 99-2230).
CEC (1981) Directive 77/312/CEE du Conseil du 29 mars 1977 concernant
la surveillance biologique de la population vis-à-vis du risque
saturnin. Rapport sur la mise en oeuvre de la directive. Luxembourg,
Commission des Communautés européennes, Direction Santé et Sécurité
(V/E/1/81-17/2).
Cerklewski FL & Forbes RM (1976) Influence of dietary zinc on lead
toxicity in the rat. J Nutr, 110: 689-696.
Chai S & Webb RC (1988) Effects of lead on vascular reactivity.
Environ Health Perspect, 78: 85-89.
Chakravorti S & Bhar S (1978) Chronic occupational exposure to lead:
An evaluation of environmental lead levels, lead absorption and
intoxication of storage battery workers. Bombay, Central Labour
Institute, Industrial Hygiene Division.
Chamberlain AC (1983) Effect of airborne lead on blood lead. Atmos
Environ, 17: 677-692.
Chamberlain AC (1985) Prediction of response of blood lead to airborne
and dietary lead from volunteer experiments with lead isotopes. Proc R
Soc (London), B224: 149-182.
Chamberlain AC, Heard MJ, Little P, Newton D, Wells AC, Wiffen RD
(1978) Investigations into lead from motor vehicles. Harwell, United
Kingdom Atomic Energy Authority (Report No. AERE-R9198).
Chan WH, Tang AJS, Chung DHS, & Lusis MA (1986) Concentration and
deposition of trace metals in Ontario - 1982. Water Air Soil Pollut,
29(4): 373-389.
Charney E, Kessler B, Farfel M, & Jackson D (1983) Childhood lead
poisoning: a controlled trial for the effect of dust-control measures
on blood lead levels. N Engl J Med, 309: 1089-1093.
Chau YK, Wong PTS, & Bengert GA (1979) Determination of tetraalkyl-
lead compounds in water, sediments, and fish samples. Anal Chem,
51: 186-188.
Chau YK, Wong PTS, & Kramer O (1980) Occurrence of tetraalkylead
compounds in the aquatic environment. Bull Environ Contam Toxicol,
24: 265-269.
Chettle DR, Scott MC, & Somervaille LJ (1991) Lead in bone: sampling
and quantitation using K x-rays excited by 109Cd. Environ Health
Perspect, 91: 49-55.
Chisolm JJ Jr (1962) Aminoaciduria as a manifestation of renal tubular
injury in lead intoxication and a comparison with patterns of
aminoaciduria seen in other diseases. J Pediatr, 60: 1-17.
Chisolm JJ & Brown DH (1979) Micromethod for zinc protoporphyrin in
erythrocytes: including new data on the absorptivity of zinc
protoporphyrin and new observations in neonates and sickle cell
disease. Biochem Med, 22: 214-237.
Chisolm JJ, Thomas DJ, & Hamill TG (1985) Erythrocyte porphobilinogen
synthase activity as an indicator of lead exposure to children. Clin
Chem, 31: 601-605.
Choie DD & Richter GW (1972) Cell proliferation in rat kidney induced
by lead acetate and effects of uninephrectomy on cell proliferation.
Am J Pathol, 66: 265-275.
Choie DD & Richter GW (1974) Cell proliferation in mouse kidney
induced by lead. Synthesis of deoxyribonucleic acid. Lab Invest,
30: 647-651.
Chowdhury AR, Dewan A, & Gandhi DN (1984) Toxic effect of lead on the
testes of rat. Biomed Biochem Acta, 43: 95-100.
Chowdhury AR, Chinoy NJ, & Gautam AK (1986) Effect of lead on human
semen. Adv Contracept Delivery Syst, 2: 208-211.
Christoffersson JO, Schutz A, Skerfving S, Ahlgren L, & Mattson S
(1986) Decrease of skeletal lead after end of occupational exposure.
Arch Environ Health, 41: 312-318.
Clark CS, Bornschein RL, Succop P, Que Hee SS, Hammond PB, & Peace B
(1985) Condition and type of housing as an indicator of potential
environmental lead exposure and pediatic blood lead levels. Environ
Res, 38: 46-53.
Clausing P, Brunekreef B, & van Wijnen JH (1987) A method for
estimating soil ingestion by children. Int Arch Occup Environ Health,
59: 73-82.
Cohen N, Modai D, Golik A, Weissgarten J, Peller S, Katz A, Averbukh
Z, & Shaked U (1989) Increased concanavalin A-induced suppressor cell
activity in humans with occupational lead exposure. Environ Res,
48: 1-6.
Cohn J, Cox C, & Cory-Slechta DA (1993) The effects of lead exposure
on learning in a multiple repeated acquisition and performance
schedule. Neurotoxicology, 14: 329-346.
Collins MF, Hrdina PD, & Whittle E (1992) Lead in blood and brain
regions of rats chronically exposed to low doses of the metal. Toxicol
Appl Pharmacol, 65: 314-322.
Colombo A (1985) The underdefined nature of the blood lead-air lead
relationship from biological and statistical grounds. Atmos Environ,
19: 1485-1493.
Columbano A, Ledda-Columbano GM, Lee G, Rajalakshmi S, & Sarma DSR
(1984) Inability of mitogen-induced liver hyperplasia to support
induction of enzyme-altered islands induced by liver carcinogens.
Cancer Res, 47: 5557-5559.
Cookman GR, King W, & Regan CM (1987) Chronic low-level lead exposure
impairs embryonic to adult conversion of the neural cell adhesion
molecule. J Neurochem, 49(2): 399-403.
Cookman GR, Hemmens SE, Keane GJ, King WB, & Regan CM (1988) Chronic
low level lead exposure precociously induces rat glial development
in vitro and in vivo. Neurosci Lett, 86: 33-37.
Cools A, Salle HJA, Verberk MM, & Zielhuis RL (1976) Biochemical
response of male volunteers ingesting inorganic lead for 49 days. Int
Arch Occup Environ Health, 38: 129-139.
Cooney GH, Bell A, McBride W, & Carter C (1989a) Neurobehavioural
consequences of prenatal low level exposures to lead. Neurotoxicol
Teratol, 11: 95-104.
Cooney GH, Bell A, McBride W, & Carter C (1989b) Low level exposures
to lead: the Sydney lead study. Dev Med Child Neurol, 31: 640-649.
Cooney G, Bell A, & Stavron C (1991) Low level exposures to lead and
neurobehavioural development: the Sydney study at seven years. In:
Heavy metals in the environment. Edinburgh, CEP Consultants, pp 16-19.
Cooper WC (1981) Mortality in employees of lead production facilities
and lead battery plants, 1971-1975. In: Environmental lead:
Proceedings of the Second International Symposium on Environmental
Lead Research, Cincinnati. Ohio, December 1978. New York, London, San
Francisco, pp 111-143.
Cooper WC (1988) Deaths from chronic renal disease in US battery and
lead production workers. Environ Health Perspect, 78: 61-63.
Cooper WC & Gaffey WR (1975) Mortality of lead workers. J Occup Med,
17: 100-107.
Cory-Slechta DA (1990a) Lead exposure during advanced age: alterations
in kinetics and biochemical effects. Toxicol Appl Pharmacol,
104: 67-78.
Cory-Slechta DA (1990b) Exposure duration modifies the effects of low
level lead on fixed interval performance. Neurotoxicology,
11: 427-442.
Cory-Slechta DA & Pokora MJ (1991) Behavioral manifestations of
prolonged lead exposure initiated at different stages of the life
cycle: I. Schedule-controlled responding. Neurotoxicology,
12(4): 745-760.
Cory-Slechta DA & Thompson T (1979) Behavioural toxicity of chronic
post-weaning lead exposure in the rat. Toxicol Appl Pharmacol,
47: 151-159.
Cory-Slechta DA & Widzowski DV (1991) Low level lead exposure
increases sensitivity to the stimulus properties of dopamine D1 and D2
agonists. Brain Res, 553: 65-74.
Cory-Slechta DA, Weiss B, & Cox C (1985) Performance and exposure
indices of rats exposed to low concentrations of lead. Toxicol Appl
Pharmacol, 78: 291-299.
Coscia GC, Discalzi G, & Ponzetti C (1987) Immunological aspects of
occupational lead exposure. Med Lav, 78: 360-364.
Cramer K, Goyer RA, Jagenburg OR, & Wilson MH (1974) Renal
ultrastructure, renal function and parameters of lead toxicity in
workers with different length of lead exposure. Br J Ind Med,
31: 113-127.
Cueto LM, Baer RD, & Gonzalez EM (1989) Three cases of unusual lead
poisoning. Am J Gastroenterol, 84: 1460.
Culbard EB, Thornton L, Watt JM, Wheatley M, Moorcroft S, & Thompson M
(1988) Metal contamination in British urban dusts and soils. J Environ
Qual, 17: 226-234.
Cullen MR, Kayne RD, & Robins JM (1984) Endocrine and reproductive
dysfunction in men associated with occupational inorganic lead
intoxication. Arch Environ Health, 39: 431-440.
Dabeka RW & McKenzie AD (1987) Lead, cadmium, and fluoride levels in
market milk and infant formulas in Canada. J Assoc Off Anal Chem,
70(4):754-757.
Dabeka RW, McKenzie AD, & Lacroix GMA (1987) Dietary intakes of lead,
cadmium, arsenic and fluoride by Canadian adults: a 24-hour duplicate
diet study. Food Addit Contam, 4(1): 89-102.
Dalpra L, Tibiletti MG, Nocera G, Giulotto P, Auriti L, Carnelli V, &
Simoni G (1983) SCE analysis in children exposed to lead emission from
a smelting plant. Mutat Res, 120: 249-256.
Danielson BRG, Dencker L, & Lindgren A (1983) Transplacental movement
of inorganic lead in early and late gestation in the mouse. Arch
Toxicol, 54: 97-107.
David O, Clark J, & Voller K (1972) Lead and hyperactivity. Lancet,
2: 900-903.
Davidson C, Grimm T, & Nasta M (1981) Airborne lead and other elements
derived from local fires in the Himalays. Science, 214: 1344-1346.
Davies BE (1983) A graphical estimation of the normal lead content of
some British soils. Geoderma, 29: 67-75.
Davies JM (1984) Lung cancer mortality among workers making lead
chromate and zinc chromate pigments at three English factories. Br J
Ind Med, 41: 158-169.
Davies PH & Everhart WH (1973) Effects of chemical variations in
aquatic environments: Volume 3 - Lead toxicity to rainbow trout and
testing application factor concept. Washington, DC, US Environmental
Protection Agency (EPA Report No. R373011C).
Davies BE, Elwood PC, Gallacher J, & Ginnever RC (1985) The
relationships between heavy metals in garden soils and house dusts in
an old lead mining area of North Wales, Great Britain. Environ Pollut,
B9: 255-266.
Davies DJ, Thornton I, Watt JM, Culbard EB, Harvey PG, Delves HT,
Sherlock JC, Smart GA, Thomas JFA, & Quinn MJ (1987a) Lead intake and
blood lead in 2 year olds UK urban children. Sci Total Environ,
90: 13-29.
Davies DJ, Watt JM, & Thornton I (1987b) Lead levels in Birmingham
dusts and soils. Sci Total Environ, 67: 177-185.
Davis JM & Grant LD (1992) The sensitivity of children to lead. In:
Guzelian PS, Henry CJ, & Olin S ed. Similarities and differences
between children and adults: Implications for risk assessment.
Washington, DC, ILSI Press, pp 15-27.
Davis JM & Svendsgaard DJ (1990) Nerve conduction velocity and lead: A
critical review and meta-analysis. In: Johnson BL, Anger WK, Durao A,
& Xinteras C ed. Advances in neurobehavioural toxicology: Applications
in environmental and occupational health. Chelsea, Michigan, Lewis
Publishers, pp 353-376.
Davis S, Waller P, Buschbom R, Balfour J, & White P (1990a)
Quantitative estimates of soil ingestion in normal children between
the ages of 2 and 7 years: Populations-based estimates using
aluminium, silicon, and titanium as soil tracer elements. Arch Environ
Health, 45: 112-122.
Davis JM, Otto DA, Weil DE, & Grant LD (1990b) The comparative
developmental neurotoxicity of lead in humans and animals.
Neurotoxicol Teratol, 12: 215-229.
Day JP, Hart M, & Robinson MS (1975) Lead in urban street dust. Nature
(Lond), 253: 343-345.
Dees JH, Heatfield BM, Reuben MD, & Trump BF (1980) Adenocarcinoma of
the kidney. III. Histogenesis of renal adenocarcinomas induced in rats
by N-(4-fluoro-4-biphenyl)acetamide. J Natl Cancer Inst,
64: 1537-1545.
De La Burde B & Choate MS (1972) Does asymptomatic lead exposure in
children have latent sequelae? J Pediatr, 81: 1088-1096.
De La Burde B & Choate MS (1975) Early asymptomatic lead exposure and
development at school age. J Pediatr, 87: 638-664.
De Leacy EA (1987) Lead-crystal decanters - a health risk? Med J Aust,
147: 622.
Delumyea R & Kalivretenos A (1987) Elemental carbon and lead content
of fine particles from American and French cities of comparable size
and industry, 1985. Atmos Environ, 21: 1643-1647.
Delves HT & Campbell MJ (1988) Measurements of total lead
concentrations and of lead isotope ratios in whole blood by use of
inductively coupled plasma source mass spectrometry. J Anal At
Spectrom, 3: 343-348.
Desilva PE & Donnan MB (1980) Blood lead levels in Victorian children.
Med J Aust, 2: 315-318.
Diemel JAL, Brunekreef B, Boleu JSM, Biersteker K, & Veenstra SJ
(1981) The Arnhem lead study. II. Indoor pollution, and indoor/outdoor
relationships. Environ Res, 25: 449-456.
Dieter M, Mathews HB, Jeffcoate RA, & Moseman RF (1993)
Bioavailability of lead found in mine tailings from Alaska. J Toxicol
Environ Health, 39: 79-93.
Dietrich KN, Krafft KM, Bier M, Succop P, Berger O, & Bornschein R
(1986) Early effects of fetal lead exposure: neurobehavioural findings
at six months. Int J Biosoc Res, 8: 151-168.
Dietrich KN, Krafft KM, Bornschein RL, Hammond PB, Berger OM, Succop
PA, & Bier M (1987) Low-level fetal lead exposure effect on
neurobehavioural development in early infancy. Pediatrics,
80: 721-730.
Dietrich KN, Krafft KM, Bier M, Berger O, Succop PA, & Bornschein RL
(1989) Neurobehavioural effects of foetal lead exposure: the first
year of life. In: Smith MA, Grant LD, & Sors AI ed. Lead exposure and
child development: An international assessment. Dordrecht, London,
Kluwer Academic Publishers, pp 320-331.
Dietrich KN, Succop PA, Bornschein RL, Krafft KM, Berger O, Hammond
PB, & Buncher CR (1990) Lead exposure and neurobehavioural development
in later infancy. Environ Health Perspect, 89: 13-19.
Dietrich KN, Succop PA, Berger OG, Hammond PB, & Bornschein RL (1991)
Lead exposure and cognitive development of urban, preschool children:
the Cincinnati lead cohort at age 4 years. Neurotoxicol Teratol,
13: 203-211.
Dietrich KN, Succop PA, Berger OG, & Keith RW (1992) Lead exposure and
the central auditory processing abilities and cognitive development of
urban children: the Cincinnati lead study cohort at age 5 years.
Neurotoxicol Teratol, 14: 51-56.
Dietrich K, Berger O, Succop P, Hammond P, & Bornschein R (1993a) The
developmental consequences of low to moderate prenatal and postnatal
lead exposure: Intellectual attainment in the Cincinnati Lead Study
Cohort following school entry. Neurotoxicol Teratol, 15: 37-44.
Dieterich K, Berger O, & Succop P (1993b) Lead exposure and the motor
developmental status of urban 6-year-old children in the Cincinnati
Prospective Study. Pediatrics, 91: 301-307.
Djuric D, Kerin Z, Graovac-Leposavic L, Novak L, & Kop M (1971)
Environmental contamination by lead from a mine and smelter: A
preliminary report. Arch environ Health, 23: 275-279.
Dolenc P, Staessen JA, Lauwerys RR, & Amery A (1993) Short report:
low-level lead exposure does not increase the blood pressure in the
general population. J Hypertens, 11: 589-593.
Dong A, Simsiman GV, & Chesters G (1985) Release of phosphorus and
metals from soils and sediments during dispersion. Soil Sci,
139: 97-99.
Donovan MP, Schein LG, & Thomas JA (1980) Inhibition of
androgen-receptor interaction in mouse prostate gland cytosol by
divalent metal ions. Mol Pharmacol, 17: 156-162.
Drasch GA & Ott J (1988) Lead in human bones. Investigations of an
occupationally non-exposed population in southern Bavaria (FGR). II.
Children. Sci Total Environ, 68: 61-69.
Drasch GA, Bohm J, & Baur C (1987) Lead in human bones. Investigations
of an occupationally non-exposed population in southern Bavaria (FGR).
I. Adults. Sci Total Environ, 64: 303-315.
Dresner DL, Ibrahim NG, Mascarenhas BR, & Levere RD (1982) Modulation
of bone marrow heme and protein synthesis by trace elements. Environ
Res, 28: 55-66.
Drill S, Konz J, Mahar H, & Morse M (1979) The environmental lead
problem: An assessment of lead in drinking water from a multi-media
perspective. Washington, DC, US Environmental Protection Agency
(EPA-570/9-79-003, NTIS-PB-296556).
Duggan MJ (1980) Lead in urban dust: An assessment. Water Air Soil
Pollut, 14: 309-321.
Egle P & Shelton KR (1986) Chronic lead intoxication causes a brain
specific nuclear protein to accumulate in the nuclei of cells lining
kidney tubules. J Biol Chem, 261: 2294-2298.
Elias R (1985) Lead exposures in the human environment. In: Mahaffey
KR ed. Dietary and environmental lead: human health effects.
Amsterdam, Oxford, New York, Elsevier Science Publishers, pp 79-109.
Ellen G & van Loon JW (1990) Determination of cadmium and lead in
foods by graphite furnace atomic absorption spectrometry with Zeeman
background correction: test with certified reference materials. Food
Addit Contam, 7: 265-273.
Elwood PC (1986) The sources of lead in blood: A critical review. Sci
Total Environ, 52: 1-23.
Elwood PC, Davey-Smith G, Oldham PD, & Toothill C (1988a) Two Welsh
surveys of blood lead and blood pressure. Environ Health Perspect,
78: 119-121.
Elwood PC, Yarnell JWG, Oldham PD, Catford JC, Nutbeam D, Davey-Smith
G, & Toothill C (1988b) Blood pressure and blood lead in surveys in
Wales. Am J Epidemiol, 127: 942-945.
Ernhart B & Morrow-Tlucak M (1987) The caretaking environment and
exposure of children to lead. In: Thornton I & Culbard E ed. Lead in
the home environment. Northwood, Science Reviews Ltd.
Ernhart CB, Wolf AW, Sokol RJ, Brittenham GM, & Ernhard P (1985) Fetal
lead exposure: antenatal factors. Environ Res, 38: 54-66.
Ernhart CB, Wolf WW, Kennard MJ, Erhard P, Filipovick HF, & Sokol RJ
(1986) Intrauterine exposure to low levels of lead: the status of the
neonate. Arch Environ Health, 41: 287-291.
Ernhart CB, Morrow-Tlucak M, Marler MR, & Wolf AW (1987) Low level
lead exposure in the prenatal and early preschool periods: early
preschool development. Neurotoxicol Teratol, 9: 259-270.
Ernhart CB, Morrow-Tlucak M, & Wolf AW (1988) Low level lead exposure
and intelligence in the preschool years. Sci Total Environ,
71: 453-459.
Ernhart CB, Morrow-Tlucak M, Worf AW, Super D, & Drotar D (1989a) Low
level lead exposure in the prenatal and early preschool periods:
intelligence prior to school entry. Neurotoxicol Teratol, 11: 161-170.
Ernhart CB, Brittenham G, Marler MR, & Sokol RJ (1989b) Lead related
birth defects: some methodologic issues. In: Smith MA, Grant LD, &
Sors AI ed. Lead exposure and child development: An international
assessment. Dordrecht, London, Kluwer Academic Publishers, pp 357-370.
Evans RD & Rigler FH (1985) Long distance transport of anthropogenic
lead as measured by lake sediments. Water Air Soil Pollut,
24: 141-151.
Everson J & Patterson CC (1980) "Ultra-clean" isotope dilution/mass
spectrometric analyses for lead in human blood plasma indicate that
most reported values are artificially high. Clin Chem, 26: 1603-1607.
Evis MJ, Kane KA, Moore MR, & Parratt JR (1985) The effects of chronic
low lead treatment and hypertension on the severity of cardiac
arrhythmias induced by coronary artery ligation in anaesthetized rats.
Toxicol Appl Pharmacol, 80: 234-242.
Evis MJ, Dhaliwal K, Kane KA, Moore MR, & Parratt JR (1987) The
effects of chronic lead treatment and hypertension on the severity of
cardiac arrhythmias induced by coronary artery occlusion or by
noradrenaline in anaesthetised rats. Arch Toxicol, 59: 336-340.
Ewers U, Stiller-Winkler R, & Idel H (1982) Serum immunoglobulin,
complement C3, and salivary IgA level in lead workers. Environ Res,
29: 351-357.
Fanning D (1988) A mortality study of lead workers, 1926-1985. Arch
Environ Health, 43: 247-251.
FAO/WHO (1993) Evaluation of certain food additives and contaminants.
Forty-first report of the Joint FAO/WHO Expert Committee on Food
Additives. Geneva, World Health Organization (Technical Report Series
837).
Feigl F, Anger V, & Oesper RE (1972) Spot tests in inorganic analysis.
London, Elsevier Publishing Company, pp 285-286.
Fergusson DM, Fergusson JE, Horwood LJ, & Kinzett NG (1988a) A
longitudinal study of dentine lead levels, intelligence, school
performance and behaviour. Part I. Dentine lead levels and exposure to
environmental risk factors. J Child Psychol Psychiatr, 29: 781-792.
Fergusson DM, Fergusson JE, Horwood LJ, & Kinzett NG (1988b) A
longitudinal study of dentine levels, intelligence, school performance
and behaviour. Part II. Dentine lead and cognitive ability. J Child
Psychol Psychiatr, 29: 793-809.
Fergusson DM, Fergusson JE, Horwood LJ, & Kinzett NG (1988c) A
longitudinal study of dentine lead levels, intelligence, school
performance and behaviour. Part III. Dentine lead levels and
attention/activity. J Child Psychol Psychiatr, 29: 811-824.
Fergusson JE, Fergusson DM, Horwood LJ, & Kinzett NG (1989) A
longitudinal study of dentine lead levels, intelligence, school
performance and behaviour: The measurement of dentine lead. Sci Total
Environ, 80: 229-241.
Fernando NP, Healy MA, Aslam M, Davis SS, & Hussein A (1981) Lead
poisoning and traditional practices: The consequences for world
health. A study in Kuwait. Public Health (Lond), 95: 250-260.
Fischbein A, Alvares AP, Anderson KE, Sassa S, & Kappas A (1977) Lead
intoxication among demolition workers: the effect of lead on the
hepatic cytochrome P-450 system in humans. J Toxicol Environ Health,
3: 431-437.
Fischbein A, Rice C, Sarkozi L, Kon SH, Petrocci M, & Selikoff IJ
(1979) Exposure to lead in firing ranges. J Am Med Assoc,
241: 1141-1144.
Fischbein A, Thornton J, Blumberg WE, Bernstein J, Valciukas JA, Moses
M, Davidow B, Kaul B, Sirota M, & Selikoff IJ (1980) Health status of
cable splicers with low-level exposure to lead: results of a clinical
survey. Am J Public Health, 70: 697-700.
Flegal AR, Rosman KJR, & Stephenson MD (1987) Isotope systematics of
contaminant leads in Monterey Bay. Environ Sci Technol, 21: 1075-1079.
Flora S-S & Tandon SK (1987) Effect of combined exposure to lead and
ethanol on some biochemical indices in the rat. Biochem Pharmacol,
36: 537-541.
Florence TM, Lilley SG, & Stauber JL (1988) Skin absorption of lead.
Lancet, 2: 157-158.
Fouassin A & Fondu M (1980) Evaluation of the daily intake of lead and
cadmium from food in Belgium. Arch Belg Méd Soc Hyg Méd Trav Méd Lég,
38: 453-467.
Foster WG (1992) Reproductive toxicity of chronic lead-exposure in the
female cynomolgus monkeys. J Reprod Toxicol, 16: 123-131.
Fouts PJ & Page JH (1942) The effect of chronic lead poisoning on
arterial blood pressure in dogs. Am Heart J, 24: 329-331.
Fowler BA, Kimmel CA, Woods JS, McConnell EE, & Grant LD (1980)
Chronic low-level lead toxicity in the rat: III. An integrated
assessment of long-term toxicity with special reference to the kidney.
Toxicol Appl Pharmacol, 56: 59-77.
Fox DA & Sillman AJ (1979) Heavy metals affect rod, but not cone
pholoreceptors. Science, 206: 78-80.
Fox DA, Lewkowski JP, & Cooper GP (1977) Acute and chronic effects of
neonatal lead exposure on developmental of the visual worked response
in rats. Toxicol Appl Pharmacol, 40: 449-461.
Freeman GB, Johnson JD, Killinger JM, Liao SC, Feder PI, Davis AO,
Ruby MV, Chaney RL, Lovre SC, & Bergstrom PD (1992) Relative
bioavailability of lead from mining waste soils in rats. Fundam Appl
Toxicol, 19: 388-398.
Friberg L (1988) Quality assurance. In: Clarkson TW, Friberg L,
Nordstrom GF, & Sager PR ed. Biological monitoring of toxic metals.
New York, Plenum Press, pp 103-126.
Friberg L & Vahter M (1983) Assessment of exposure to lead and cadmium
through biological monitoring: Results of a UNEP/WHO global study.
Environ Res, 30: 95-128.
Friedman JA & Weinberger HL (1990) Six children with lead poisoning. J
Dis Child, 144: 1039-1044.
Fullmer CS & Rosen JF (1990) Effect of dietary calcium and lead status
on intestinal calcium absorption. Environ Res, 51: 91-99.
Fullmer CS (1990) Intestinal lead and calcium absorption: Effect of
1,25-dihydroxy-cholecalciferol and lead status. Proc Soc Exp Biol Med,
194: 258-264.
Fulton M, Thomson G, Hunter R, Raab G, Laxen D, & Hepburn W (1987)
Influence of blood lead on the ability and attainment of children in
Edinburgh. Lancet, 30 May: 1221-1226.
Galal-Gorchev H (1991a) Dietary intake of pesticide residues, cadmium,
mercury and lead. Food Addit Contam, 8(6): 793-806.
Galal-Gorchev H (1991b) Global overview of dietary lead exposure.
Special issue incorporating the Proceedings of the Symposium on the
Bioavailability and Dietary Exposure of Lead, Chapel Hill, North
Carolina, September 1990. Chem Speciation Bioavailab, 3(3/4): 5-11.
Garber BT & Wei E (1974) Influence of dietary factors on the
gastrointestinal absorption of lead. Toxicol Appl Pharmacol,
27: 685-691.
Garcia-Miragaya J (1984) Levels, chemical fractionation, and
solubility of lead in roadside soils of Caracas, Venezuela. Soil Sci,
138: 147-152.
Gartrell MJ, Craun JC, Podrebarac DS, & Gunderson EL (1985a)
Pesticides, selected elements, and other chemicals in infant and
toddler total diet samples, October 1979 -September 1980. J Assoc Off
Anal Chem, 68: 1184-1187.
Gartrell MJ, Craun JC, Podrebarac DS, & Gunderson EL (1985b)
Pesticides, selected elements, and other chemicals in infant and
toddler total diet samples, October 1979 -September 1980. J Assoc Off
Anal Chem, 81: 1163-1183.
Gartside PS (1988) The relationship of blood lead levels and blood
pressure in NHANES II: additional calculations. Environ Health
Perspect, 78: 31-34.
Gartside PS & Buncher CR (1982) Relationship of air lead and blood
lead for workers at an automobile battery factory. Int Arch Occup
Environ Health, 50: 1-10.
GEMS (1985) Global Environmental Monitoring System: Assessment of
human exposure to lead: Comparison between Belgium, Malta, Mexico and
Sweden. Stockholm, Karolinska Institute.
Gennart JP, Bernard A, & Lauwerys R (1992) Assessment of thyroid,
testes, kidney and autonomic nervous system function in lead-exposed
workers. Int Arch Occup Environ Health, 64: 49-57.
Gerhardsson L, Lundstrom N-G, Nordbery G, & Wall S (1986) Mortality
and lead exposure: a retrospective cohort study of Swedish smelter
workers. Br J Ind Med, 43: 707-712.
Getz LL, Haney AW, Larimore RW, McNurney JW, Leland HV, Price PW,
Rolfe GL, Wortman RL, Hudson JL, Solomon RL, & Reinbold KA (1977)
Transport and distribution in a watershed ecosystem. In: Boggess WR
ed. Lead in the environment. Springfield, Virginia, National Technical
Information Service, pp 105-133 (National Science Foundation report
No. NSF/RA-770214).
Gibson JL (1904) A plea for painted railings and painted walls of
rooms as the source of lead poisoning amongst: Queensland children.
Aust Med Gaz, 23: 149.
Gilbert SG & Rice DC (1987) Low-level lifetime lead exposure produces
behavioral toxicity (spatial discrimination reversal) in adult
monkeys. Toxicol Appl Pharmacol, 91: 484-490.
Gmerek DE, McCofferty MR, O'Neill KJ, Melamed BR, & O'Neill JJ (1981)
Effect of inorganic lead on rat brain mitochondrial respiration and
energy production. J Neurochem, 36: 1109-1113.
Goldstein GW (1990) Lead poisoning and brain cell function. Environ
Health Perspect, 89: 91-94.
Gould JH, Capar SG, Jacobs RM, & Plunkett R (1988) A quick color test
to detect lead release from glazed ceramic and enameled metalware.
Anal Lett, 21: 2145-2154.
Goyer RA (1985) Renal changes associated with lead exposure. In:
Mahaffey KR ed. Dietary and environmental lead: Human health effects.
Amsterdam, Oxford, New York, Elsevier Science Publishers, pp 315-335.
Goyer RA (1992) Nephrotoxicity and carcinogenicity of lead - An update
on exposure and effects of lead. Fundam Appl Toxicol, 18: 1-16.
Goyer RA (1993) Lead toxicity: current concerns. Environ Health
Perspect, 100: 177-187.
Goyer RA & Rhyne BC (1973) Pathological effects of lead. Int Rev Exp
Pathol, 12: 1-77.
Goyer RA & Wilson MH (1975) Lead induced inclusion bodies: results of
ethylenediamine-tetra-acetic acid treatment. Lab Invest, 32: 149-156.
Goyer RA, Leonard DL, Moore JF, Rhyne B, & Krigman MR (1970) Lead
dosage and the role of the intranuclear inclusion body. Arch Environ
Health, 20: 705-711.
Goyer RA, Falk HL, Hogan M, Feldman DD, & Richter W (1981) Renal
tumors in rats given trisodium nitrilotriacetic acid in drinking water
for 2 years. J Natl Cancer Inst, 66: 869-880.
Goyer RA, Weinberg CR, Victery WM, & Miller CR (1989) Lead induced
nephrotoxicity: kidney calcium as an indicator of tubular injury. In:
Bach PH & Lock EA ed. Nephrotoxicity. New York, London, Plenum Press,
pp 11-20.
Grandjean P (1979) Occupational lead poisoning in Denmark: screening
with the haematofluorometer. Br J Ind Med, 36: 52-58.
Grandjean P & Olsen B (1984) Lead. In: Vercruysse A ed. Techniques and
instrumentation in analytical chemistry. Volume 4: Evaluation of
analytical methods in biological systems. Part B: Hazardous metals in
human toxicology. Amsterdam, Oxford, New York, Elsevier Science
Publishers, pp 153-169.
Grandjean P, Wulf HC, & Niebuhr E (1983) Sister chromatid exchange in
response to variations in occupational lead exposure. Environ Res,
32: 199-204.
Grandjean P, Hansen ON, & Lyngbye K (1984) Analysis of lead in
circumpulpal dentin of deciduous teeth. Ann Clin Lab Sci, 14: 270-275.
Grandjean P, Hansen ON, & Lyngbye K (1986) Lead concentration in
deciduous teeth; variation related to tooth type and analytical
technique. J Toxicol Environ Health, 19: 437-445.
Grandjean P, Jensen BM, Sando SH, Jorgensen PJ, & Antonsen S (1989a)
Delayed blood regeneration in lead exposure: an effect on reserve
capacity. Am J Public Health, 79: 1385-1388.
Grandjean P, Hollnagel H, Hedegaard L, Christensen JM, & Larsen S
(1989b) Blood lead-blood pressure relations: alcohol intake and
haemoglobin as confounders. Am J Epidemiol, 129: 732-739.
Grandjean P, Lyngbye T, & Hansen ON (1991) Lessons from a Danish study
on neuropsychological impairment related to lead exposure. Environ
Health Perspect, 94: 111-115.
Granick JL, Sassa S, Granick S, Levere RD, & Kappas A (1973) Studies
in lead poisoning. II. Correlation between the ratio of activated to
inactivated aminolevulinic acid dehydratase of whole blood and the
blood lead level. Biochem Med, 8: 149-159.
Grant LD & Davis JM (1989) Effects of low-level lead exposure on
paediatric neurobehavioural development: Current findings and future
directions. In: Smith MA, Grand LD, & Sors AI ed. Lead exposure and
child development: An international Assessment. Dordrecht, London,
Kluwer Academic Publishers, pp 49-115.
Grant LD, Kimmel CA, West GL, Martinez-Vargas CM, & Howard JL (1980)
Chronic low-level lead toxicity in the rat. II. Effects on postnatal
physical and behavioral development. Toxicol Appl Pharmacol, 56:
42-58.
Graziano JH & Blum C (1991) Lead exposure from lead crystal. Lancet,
337: 141-142.
Graziano JH, Popovac D, Factor-Litvak P, Shrout P, Kline J, Murphy MJ,
Zhao Y, Mehmeti A, Ahmedi X, Rajovic B, Zvicer Z, Nenezic U, Lolacono
NJ, & Steun Z (1990) Determinants of elevated blood lead during
pregnancy in a population surrounding a lead smelter in Kosova,
Yugoslavia. Environ Health Perspect, 89: 95-100.
Graziano JH, Slavkovic V, Factor-Litvak P, Popovac D, Ahmedi X, &
Mehmeti A (1991) Depressed serum erythropoietin in pregnant women with
elevated blood lead. Arch Environ Health, 46: 347-350.
Greene T, Ernhart CB, & Boyd TA (1992) Contributions of risk factors
to elevated blood and dentine levels in preschool children. Sci Total
Environ, 115: 239-260.
Griffith JQ & Landaver MA (1944) The effect of chronic lead poisoning
on arterial blood pressure in rats. Am Heart J, 28: 295-297.
Gulson B & Wilson D (1994) History of lead exposure in children
revealed from isotopic analyses of teeth. Arch Environ Health,
49X: 279-283.
Gupta AK, Sehgal SK, Mohan M, & Anand NK (1990) Cosmetic plumbism.
Indian Paediatr, 27: 760-761.
Gustafson A, Hedmer P, Schutz A, & Skerfving S (1989) Occupational
lead exposure and pituitary function. Int Arch Occup Environ Health,
61: 277-281.
Haeger-Aronsen B (1960) Studies on urinary excretion of
delta-aminolaevulic acid and other haem precursors in lead workers and
lead-intoxicated rabbits. Scand J Clin Lab Invest, 12(47): 1.
Haenninen H, Mantere P, Hernberg S, Seppalainen AM, & Kock B (1979)
Subjective symptoms in low-level exposure to lead. Neurotoxicology,
1: 333-347.
Hakim RB, Stewart WF, Canner JK, & Tielsch JM (1991) Occupational lead
exposure and strabismus in offspring: A case control study. Am J
Epidemiol, 133: 351-356.
Hammond PB, O'Flaherty EJ, & Gartside PS (1981) The impact of air-lead
on blood-lead in man: A critique of the recent literature. Food Cosmet
Toxicol, 19: 631-638.
Hammond PB, Bornschein RL, & Succop P (1985) Dose-effect and
dose-response relationships of blood lead to erythrocytic
protoporphyrin in young children. Environ Res, 38: 187-196.
Hansen ON, Trillingsgaard A, Beese I, Lyngbye T, & Grandjean P (1989)
A neuropsychological study of children with elevated dentine lead
level: assessment of the effect of lead in different socio-economic
groups. Neurotoxicol Teratol, 11: 205-213.
Harlan WR (1988) The relationship of blood lead levels to blood
pressure in the US population. Environ Health Perspect, 78: 9-13.
Harlan WR, Landis JR, Schmouder RL, Goldstein NG, & Harlan LC (1985)
Blood lead and blood pressure: relationship in the adolescent and
adult US population. J Am Med Assoc, 253: 530-534.
Harvey PG, Hamlin MW, Kumar R, & Delves HT (1984) Blood lead,
behaviour and intelligence test performance in preschool children. Sci
Total Environ, 40: 45-60.
Harvey PG, Hamlin MW, Kumar R, Morgan J, Spurgeon A, & Delves HT
(1988) Relationships between blood lead, behaviour, psychometric and
neuropsychological test performance in young children. Br J Dev
Psychol, 6: 145-156.
Hatzakis A, Kokkevi A, Maravelias C, Katsouyanni K, Salaminios F,
Kalandidi A, Koutselinis A, Stefanis C, & Trichopoulos D (1989)
Psychometric intelligence deficits in lead-exposed children. In: Smith
MA, Grand LD, & Sors AI ed. Lead exposure and child development: An
international assessment. Dordrecht, London, Kluwer Academic
Publishers, pp 211-223.
Hawk BA, Schroeder SR, Robinson G, Otto D, Mushak P, Kleinbaum D, &
Dawson G (1986) Relation of lead and social factors to IQ of low-SES
children: a partial replication. Am J. Ment Defic, 91: 178-183.
Heard MJ & Chamberlain AC (1982) Effects of minerals and food on
uptake of lead from the gastrointestinal tract in humans. Hum Toxicol,
1: 411-416.
Hem JD & Durum WH (1973) Solubility and occurrence of lead in surface
water, J An Water Works Assoc, 65: 562-568.
Henning SJ & Cooper LC (1988) Intestinal accumulation of lead salts
and milk lead by suckling rats (42645). Proc Soc Exp Biol Med,
187: 110-116.
Hernberg S (1973) Prevention of occupational poisoning from inorganic
lead. Work Environ Health, 10: 53-61.
Hernberg S & Nikkanen J (1970) Enzyme inhibition by lead under normal
urban conditions. Lancet, 1(7637): 63-64.
Hilderbrand DC, Der R, Griffin WT, & Fahim MS (1973) Effect of lead
acetate on reproduction. Am J Obstet Gynecol, 115: 1058-1065.
Hogstedt C, Hane M, Agrell A, & Bodin L (1983) Neuropsychological test
results and symptoms among workers with well-defined long term
exposure to lead. Br J Ind Med, 49: 99-105.
Holness DL & Nethercott JR (1988) Acute lead intoxication in a group
of demolition workers. Appl Ind Hyg, 3: 338-341.
Holtzman D, Shen Hgu J, & Montell P (1978) In vitro effects of
inorganic lead on isolated rat brain mitochondrial respiration.
Neurochem Res, 3: 195-206.
Holtzman D, Shen Hgu J, & Desautel M (1981) Absence of effects of lead
feedings and growth retardation on mitochondrial and microsomal
cytochrome in the developing brain. Toxicol Appl Pharmacol, 58: 48-56.
Holtzman D, Devries C, Nguyan H, Olson J, & Bensch K (1984) Maturation
of resistance to lead encephalopathy: cellular and subcellular
mechanisms. Neurotoxicology, 5: 97-124.
Hopper JL, Balderas A, & Mathews JO (1982) Analysis of variation in
blood lead levels in Melbourne families. Med J Aust, 2: 573-576.
Huang J, He F, Yigun W, & Zhang S (1988a) Observations on renal
function in workers exposed to lead. Sci Total Environ, 71: 535-537.
Huang XP, Feng ZY, Zhai WL, & Xu JH (1988b) Chromosomal aberrations
and sister chromatid exchanges in workers exposed to lead. Biomed
Environ Sci, 1: 382-387.
Huber F & Schmidt U (1976) Methylation of organolead and lead (II)
compounds to C3 4Pb by microorganisms. Nature (Lond),
259: 157-158.
Hutton M, Wadge A, & Milligan PJ (1988) Environmental levels of
cadmium and lead in the vicinity of a major refuse incinerator. Atmos
Environ, 22: 411-416.
IAEA (1987) Co-ordinated research programme on human daily dietary
intakes of nutritionally important trace elements as measured by
nuclear and other techniques. IAEA Newl, 2: 6-15.
IARC (1980) Lead and lead compounds. In: Some metals and metallic
compounds. Lyon, International Agency for Research on Cancer, pp
325-416 (IARC Monographs on the Evaluation of the Carcinogenic Risk of
Chemicals to Humans, Volume 23).
IARC (1987a) Genetic and related effects: An updating of selected IARC
Monographs from Volumes 1 to 42. Lyon, International Agency for
Research on Cancer (IARC Monographs on the Evaluation of the
Carcinogenic Risk of Chemicals to Humans, Supplement 6).
IARC (1987b) Lead and lead compounds. In: Overall evaluations of
carcinogenicity: an updating of IARC Monographs, Volumes 1 to 42.
Lyon, International Agency for Research on Cancer, pp 230-232 (IARC
Monographs on the Evaluation of the Carcinogenic Risk of Chemicals to
Humans, Supplement 7).
Ibels LS & Pollock CA (1986) Toxicology management review. Lead
intoxication. Med Toxicol, 1: 387-410.
Ichiba M & Tomokuni K (1990) Studies on erythrocyte pyrimidine
5'-nucleotidase test and its evaluation in workers occupationally
exposed to lead. Int Arch Occup Environ Health, 62: 305-310.
ILZSG (International Lead and Zinc Study Group) (1992) Lead and zinc
statistics. Int Lead Zinc Study Group Bull, 32: 3.
IPCS (1977) Environmental health criteria 3: Lead. Geneva, World
Health Organization, 160 pp.
IPCS (1989) Environmental health criteria 85: Lead - Environmental
effects. Geneva, World Health Organization, 106 pp.
IPCS (1994) Environmental health criteria 155: Biomarkers and risk
assessment: Concepts and principles. Geneva, World Health
Organization, 82 pp.
IRPC (International Radiological Protection Commission), Task Group on
Lung Dynamics (1966) Deposition and retention models for internal
dosimetry of the human respiratory trace. Health Phys, 12: 173-207.
IRPTC (1987) IRPTC legal file 1986. Geneva, International Register of
Potentially Toxic Chemicals, United Nations Environment Programme,
vol 1.
Irwig LM, Harrison WO, Rocks P, Webster I, & Andrew M (1978) Lead and
morbidity: a dose response relationship. Lancet, 2(8079): 4-7.
Jaakkola J & Anttila A (1992) [Occupational exposures to chemical
agents in Finland.] Helsinki, Institute of Occupational Health and
Finnish Work Environmental Fund (in Finnish).
James AC (1978) Lung deposition of submicron aerosols calculated as a
function of age and breathing rate. In: National Radiological
Protection Board Annual Report. Harwell, UK, National Radiological
Protection Board, pp 71-75.
Jaremin B (1983) Blast lymphocyte transformation (LTT), rosette
(E-RFC) and leukocyte migration inhibition (MIF) tests in persons
exposed to the action of lead during work. Report II. Bull Inst Marit
Trop Med (Gdynia), 34: 187-197.
Jensen AA (1984) Metabolism and toxicokinetics. In: Grandjean P ed.
Biological effects of organolead compounds. Boca Raton, Florida, CRC
Press, chapter 8, pp 97-115.
Johnson NE & Tenuta K (1979) Diets and lead blood levels of children
who practice pica. Environ Res, 18: 369-376.
Jorhem L & Slorach S (1988) Lead, chromium, tin, iron, and cadmium in
foods in welded cans. Food Addit Contam, 4: 309-316.
Jorhem L, Mattsson P, & Slorach S (1988) Lead in table wines on the
Swedish market. Food Addit Contam, 5: 645-649.
Kaminsky P, Leone J, & Duc M (1988) Incidence du saturnisme hydrique
dans un service de médecine interne en région de sols acides. Presse
Méd, 17: 419-422.
Kampe W (1983) Lead and cadmium in food - is there an actual danger?
Forum Staedte-Hyg, 34: 236-241.
Kang HK, Infante PF, & Carra JS (1980) Occupational lead exposure and
cancer (letter). Science, 207: 935-936.
Katagiri Y, Toriumi H, & Kawai M (1983) Lead exposure among 3-year-old
children and their mothers living in a pottery-producing area. Int
Arch Occup Environ Health, 52: 223-229.
Kaye WE, Novotny TE, & Tucker M (1987) New ceramics-related industry
implicated in elevated blood lead levels in children. Arch Environ
Health, 42: 161-164.
Kazantzis G (1989) Lead: ancient metal - modern menace. In: Smith MA,
Grant LD, & Sors AI ed. Lead exposure and child development: An
international assessment. Dordrecht, London, Kluwer Academic Press,
pp 119-128.
Kehoe RA (1961) The metabolism of lead in man in health and disease:
The normal metabolism of lead (The Harben Lectures, 1960). J R Inst
Public Health Hyg, 24: 81-97.
Kehoe RA (1987) Studies of lead administration and elimination in
adult volunteers under natural and experimentally induced conditions
over extended periods of time. Food Chem Toxicol, 25: 425-493.
Keller CA & Doherty RA (1980a) Distribution and excretion of lead in
young and adult female mice. Environ Res, 21: 217-228.
Keller CA & Doherty RA (1980b) Bone lead mobilization in lactating
mice and lead transfer to suckling offspring. Toxicol Appl Pharmacol,
55: 220-228.
Kerin Z (1973) Lead in new-fallen snow near a lead smelter. Arch
Environ Health, 26: 256-260.
Kholil-Manesh F, Gonich HC, Cohen AH, Alinovi R, Berganaschi E, Mutti
A, & Rosen VJ (1992) Experimental model of lead nephrotoxicity. I.
Continuous high-dose lead administration. Kidney Int, 41: 1192-1203.
Kimber I, Stonard MD, Gidlow DA, & Niewola Z (1986) Influence of
chronic low-level exposure to lead on plasma immunoglobulin
concentration and cellular immune function in man. Int Arch Occup
Environ Health, 57: 117-125.
King E & Conchie D (1979) Industrial lead absorption. Ann Occup Hyg,
22: 213-239.
Kirkby H & Gyntelberg F (1985) Blood pressure and other cardiovascular
risk factors of long term exposure to lead. Scand J Work Environ
Health, 11: 15-19.
Kocak R, Anarat A, Altintas G, & Evilyaoglu N (1989) Lead poisoning
from contaminated flour in a family of 11 members. Hum Toxicol,
8: 385-386.
Kolbye AC, Mahaffey R, Fiorino A, Corneliussen PC, & Jelinek CF (1974)
Food exposures to lead. Environ Health Perspect, 7(Exp Issue): 65-75.
Koller LD (1985) Immunological effects of lead. In: Mahaffey KR ed.
Dietary and environmental lead: Human health effects. Amsterdam,
Oxford, New York, Elsevier Science Publishers, pp 339-354.
Koller LD & Roan JG (1980) Effect of lead, cadmium and methylmercury
on immunologic memory. J Environ Pathol Toxicol, 4: 47-52.
Koo WWK, Succop PA, Bornschein RL, Krug-Wispe SK, Steichen JJ, Tsang
RC, Hammond PB, & Berger OG (1991) Serum vitamin D metabolites and
bone mineralization in young children with chronic to moderate lead
exposure. Pediatrics, 87: 680-687.
Kopp SJ, Barron JT, & Tow JP (1988) Cardiovascular actions of lead and
relationship to hypertension: a review. Environ Health Perspect,
89: 91-99.
Koren G, Chang N, Gonen R, Klein J, Weiner L, Demshar H, Pizzolato S,
Radde I, & Shime J (1990) Lead-exposure among mothers and their
newborns in Toronto. Can Med Assoc J, 142: 1241-1244.
Kostial K & Kello D (1979) Bioavailability of lead in rats fed "human"
diets. Bull Environ Contam Toxicol, 21: 312-314.
Kostial K, Kello D, & Jugo S (1978) Influence of age on metal
metabolism and toxicity. Environ Health Perspect, 25: 81-86.
Kovar IZ, Strehlow CD, Richmond J, & Thompson MG (1984) Perinatal lead
and cadmium burden in a British urban population. Arch Dis Child,
59: 36-39.
Krueger JA & Duguay KM (1989) Comparative analysis of lead in Maine
urban soils. Bull Environ Contam Toxicol, 42: 574-581.
Kusell M, Lake L, Anderson M, & Gerschenson LE (1978) Cellular and
molecular toxicology of lead. II. Effect of lead on d-aminolevulinc
acid synthelase of cultured cells. J Toxicol Environ Health,
4: 515-525.
Kutbi II, Ahmed M, & Saber A (1989) Measurement of blood-lead levels
in school children of Jeddah Saudi Arabia and assessment of sub-toxic
levels of lead on some sensitive hematological parameters. J Environ
Sci Health, A24: 943-955.
Lacey RF, Moore MR, & Richards WN (1985) Lead in water, infant diet
and blood: The Glasgow duplicate diet study. Sci Total Environ,
41: 235-257.
Lai CS (1972) Lead poisoning as an occupational hazard in Chinese
opera actors: a case report. Singap Med J, 13: 115-117.
Landis JR & Flegal KM (1988) A generalized Mantel-Haenszel analysis of
the regression of blood pressure on blood lead using NHANES II data.
Environ Health Perspect, 78: 35-41.
Landrigan PJ, Gehlbach SH, Rosenblum BF, Shoults JM, Cancelaria RM,
Barthel WF, Liddle JA, Smrek AL, Staehling NW, & Sanders JF (1975)
Epidemic lead absorption near an ore smelter; The role of particulate
lead. N Engl J Med, 292: 123-129.
Lansdown RG, Shepherd J, Clayton BE, Delves HT, Graham PJ, & Turner WC
(1974) Blood-lead levels, behaviour, and intelligence: a population
study. Lancet, 1(7857): 538-541.
Lansdown R, Yule W, Urbanowicz MA, & Hunter J (1986) The relationship
between blood-lead concentrations, intelligence, attainment and
behaviour in a school population: the second London study. Int Arch
Occup Environ Health, 57: 225-235.
Laurs AJ (1976) Review of European test methods for measuring lead and
cadmium release. In: Proceedings of the International Conference on
Ceramic Foodware Safety, Geneva, 1974. New York, Lead Industries
Association Inc., pp 27-36.
Lauwerys R, Buchet JP, Roels HA, & Materne DD (1974) Relationship
between urinary delta-aminolevulinic acid excretion and the inhibition
or red cell delta-aminolevulinate dehydratase by lead. Clin Toxicol,
7: 383-388.
Lavelle JM, Poppenga RH, Thacker BJ, Geisy JP, Weis C, Othoudt R, &
Vandervoort C (1991) Bioavailability of lead in mining wastes: An oral
intubation study in young swine. Chem Speciation Bioavailab,
3: 105-111.
Laxen DPH, Raab GM, & Fulton M (1987) Children's blood lead and
exposure to lead in household dust and water - a basis for an
environmental standard for lead in dust. Sci Total Environ,
66: 235-244.
Lee RE & Goranson S (1972) National air surveillance cascade impact or
network. I. Size distribution measurements of suspended particulate
matter in air. Environ Sci Technol, 6: 1019-1024.
Leeming RJ & Blair JA (1980) The effect of pathological and normal
physiological processes on biopterin levels in man. Clin Chim Acta,
108: 102-111.
Lepow ML, Bruckman L, Rubino RA, Markowitz S, Gillette M, & Kapish J
(1974) Role of airborne lead in increased body burden of lead in
Hartford children. Environ Health Perspect, 7: 99-102.
Lerda D (1992) Study of sperm characteristics in persons
occupationally exposed to lead. Am J Ind Med, 22: 567-571.
Levin ED & Bowman RE (1983) The effect of pre- or postnatal lead
exposure on Hamilton search task in monkeys. Neurobehav Toxicol
Teratol, 5: 391-394.
Levin ED & Bowman RE (1986) Long-term lead effects on the Hamilton
Search Test and delayed alternation in monkeys. Neurobehav Toxicol
Teratol, 8: 219-224.
Levin ED & Bowman RE (1989) Long-term effects of chronic post-natal
lead exposure on delayed spatial alternation in monkeys. Neurotoxicol
Teratol, 10: 505-510.
Lilienthal H, Winneke G, Brockhaus A, & Molik B (1986) Pre- and
postnatal lead-exposure in monkeys: Effects on activity and learning
set formation. Neurobehav Toxicol Teratol, 8: 265-272.
Lilienthal H, Winneke G, & Ewert T (1990) Effects of lead on
neurophysiological and performance measures: animal and human data.
Environ Health Perspect, 89: 21-25.
Lilis R (1981) Long-term occupational lead exposure, chronic
nephropathy, and renal cancer: a case report. Am J Ind Med,
2: 293-297.
Lilis R, Fischbein A, Eisinger J, Blumberg WE, Diamond S, Anderson HA,
Rom W, Rice C, Sarkozi L, Kon S, & Selikoff IJ (1977) Prevalence of
lead disease among secondary lead smelter workers and biological
indicators of lead exposure. Environ Res, 14: 255-285.
Lilley SG, Florence TM, & Stauber JL (1988) The use of sweat to
monitor lead absorption through the skin. Sci Total Environ,
76: 267-278.
Lind B, Vahter M, Rahnster B, & Björs U (1988) Quality control samples
for the determination of lead and cadmium in blood, feces, air
filters, and dust. Fresenius Z Anal Chem, 332: 741-743.
Lindholm ML, Sallmen M, Anttila A, Taskinen H, & Hemminki K (1991)
Paternal occupational lead exposure and spontaneous abortion. Scand J
Work Environ Health, 17(2): 95-103.
Lin-Fu JS (1985) Historical perspective on health effects of lead. In:
Mahaffey KR ed. Dietary and environmental lead: Human health effects.
Amsterdam, Oxford, New York, Elsevier Science Publishers, pp 43-64.
Lovering TG ed. (1976) Lead in the environment. Washington, DC, US
Department of the Interior, Geological Survey (Geological Survey
Professional Paper No. 957).
Lyngbye T, Hansen OL, Trillingsgaard A, Beese I, & Grandjean P (1990a)
Learning disabilities in children: significance of low-level
lead-exposure and confounding factors. Acta Paediatr Scand,
79: 352-360.
Lyngbye T, Jorgensen PJ, & Grandjean P (1990b) Validity and
interpretation of blood lead levels: a study of Danish
school-children. Scand J Clin Lab Invest, 50: 441-449.
McGrath SP (1986) The range of metal concentrations in top soils of
England and Wales in relation to soil protection guidelines. In
Hemphill DD ed. Trace substances in environmental health, vol 20.
Columbia, Missouri, University of Missouri, pp 242-252.
McGregor AJ & Mason MJ (1990) Chronic occupational lead exposure and
testicular endocrine function. Hum Exp Toxicol, 9: 371-376.
McIntosh MJ, Meredith PA, Moore MR, & Goldberg A (1985) Neurotoxic
action of lead: effect on tetrahydrobiopterin metabolism in the rat.
Comp Biochem Physiol, 81C(1): 227-231.
McIntosh MJ, Meredith PA, Moore MR, & Goldberg A (1989) Action of lead
on neurotransmission in rats. Xenobiotica, 19(1): 101-113.
McKhann CF & Vogt EC (1933) Lead poisoning in children. J Am Med
Assoc, 101: 1131-1135.
McMichael AJ & Johnson HM (1982) Long-term mortality profile of
heavily-exposed lead smelter workers. J Occup Med, 24: 375-378.
McMichael AJ, Vimpani GV, Robertson EF, Baghurst PA, & Clark PD (1986)
The Port Pirie cohort study: maternal blood lead and pregnancy
outcome. J Epidemiol Community Health, 41: 18-25.
McMichael AJ, Baghurst PA, Wigg NR, Vimpani GV, Robertson EF, &
Roberts RJ (1988) Port Pirie cohort study: environmental exposure to
lead and children's abilities at the age of four years. N Engl J Med,
319: 468-475.
Maenhaut W, Zoller WH, Duce RA, & Hoffman GL (1979) Concentration and
size distribution of particulate trace elements in the south polar
atmosphere. J Geophys Res, 84: 2421-2431.
Mahaffey KR (1985) Factors modifying susceptibility to lead toxicity.
In: Mahaffey KR ed. Dietary and environmental lead: Human health
effects. Amsterdam, Oxford, New York, Elsevier Science Publishers,
pp 373-419.
Mahaffey KR & Annest JL (1986) Association of erythrocyte
protoporphyrin with blood lead level and iron status in the Second
National Health and Nutrition Examination Survey, 1976-1980. Environ
Res, 41: 327-338.
Mahaffey-Six KR & Goyer RA (1972) The influence of iron deficiency on
tissue content and toxicity of ingested lead in the rat. J Lab Clin
Med, 79: 128-136.
Mahaffey KR, Rosen JF, Chesney RW, Peeler JT, Smith CM, & Debuca HF
(1982) Association between age, blood lead concentration, and serum
1,25-dihydroxy-cholecalciferol levels in children. Am J Clin Nutr,
35: 1327-1331.
Maines MD (1992) Haem oxygenase-clinical applications and functions.
Boca Raton, Florida, CRC Press.
Maki-Paakkanen J, Sorsa M, & Vainio H (1981) Chromosome aberrations
and sister chromatid exchanges in lead-exposed workers. Hereditas,
94: 269-275.
Manton WI (1985) Total contribution of airborne lead to blood lead. Br
J Ind Med, 42: 168-172.
Manton WI & Cook JD (1984) High accuracy (stable isotope dilution)
measurements of lead in serum and cerebrospinal fluid. Br J Ind Med,
41: 313-319.
Manton WI & Thal ER (1986) Lead poisoning from retained missiles: an
experimental study. Ann Surg, 204: 594-599.
Mao P & Molnar JJ (1967) The fine structure and histochemistry of
lead-induced renal tumors in rats. Am J Pathol, 50: 571-603.
Maranelli G & Apostoli P (1987) Assessment of renal function in lead
poisoned workers. In: Occupational and environmental chemical hazards:
Cellular and biochemical indices for monitoring toxicity. Chichester,
Ellis Horwood Ltd, chapter 42, pp 344-348.
Marcus AH (1985a) Testing alternative nonlinear kinetic models in
compartmental analysis. In: Eisenfield J & Delisi C ed. Mathematics
and computers in biomedical applications. Amsterdam, Oxford, New York,
Elsevier Science Publishers, pp 259-267.
Marcus AH (1985b) Multicompartment kinetic models for lead: Part I.
bone kinetics and variable absorption in humans without excessive lead
exposures. Environ Res, 36: 441-458.
Marcus AH (1985c) Multicompartment kinetic models for lead: Part II.
linear kinetics and variable absorption in humans without excessive
lead exposures. Environ Res, 36: 459-472.
Marcus AH & Schwartz J (1987) Dose-response curves for erythrocyte
protoporphyrin vs blood lead: effects of iron status. Environ Res,
44: 221-227.
Marino PE, Franzblau A, Lilis R, & Landrigan PJ (1989) Acute lead
poisoning in construction workers: the failure of protective
standards. Arch Environ Health, 44: 140-145.
Marks SC & Popoff SN (1988) Bone cell biology: the regulation of
development, structure, and function in the skeleton. Am J Anat,
183: 1-44.
Marlowe M & Errrera J (1982) Low lead levels and behaviour problems in
children. Behav Disord, 7: 163-172.
Matte TD, Figueroa JP, Ostrowski S, Burr G, Jackson-Hunt L, Kennylside
RA, & Baker EL (1989a) Lead poisoning among household members exposed
to lead-acid battery repair shops in Kingston, Jamaica. Int J
Epidemiol, 18: 874-881.
Matte TD, Figueroa JP, Burr G, Flesch JP, Keenlyside RA & Baker EL
(1989b) Lead exposure among lead-acid battery workers in Jamaica. Am J
Ind Med, 16: 167-177.
Meredith PA & Moore MR (1979) The influence of lead on haem
biosynthesis and biodegradation in the rat. Biochem Soc Trans,
7: 637-639.
Meredith PA, Moore MR, Campbell BC, Thompson GG, & Goldberg A (1978)
Delta-aminolevulinic acid metabolism in normal and lead-exposed
humans. Toxicology, 9: 1-9.
Merwin BW (1976) Review of US standards on test methods for lead and
cadmium release. In: Proceedings of the International Conference on
Ceramic Foodware Safety, Geneva, 1974. New York, Lead Industries
Associate Inc., pp 36-40.
Meyer BR, Fishbein A, Rosemann K, Lerman Y, Drayer DE, & Reidenberg MM
(1984) Increased urinary enzyme excretion in workers exposed to
nephrotoxic chemicals. Am J Med, 76: 989-998.
Michaelson A & Sauerhoff MW (1974) An improved model of lead induced
brain dysfunction in the suckling rat. Toxicol Appl Pharmacol,
28: 88-96.
Mielke H, Blake B, Burroughs S, & Hassinger N (1984) Urban lead levels
in Minneapolis: the case of the Hmong children. Environ Res,
34: 64-76.
Mitchell DG, Aldous KM, & Ryan FJ (1974) Mass screening for lead
poisoning, capillary blood sampling and automated Delves cup atomic
absorption analysis. NY State J Med, 74: 1599-1603.
Mitchell-Heggs CAW, Conway M, & Cassar J (1990) Herbal medicine as a
cause of combined lead and arsenic poisoning. Hum Exp Toxicol,
9: 195-196.
Molina-Ballesteros G, Zuniga-Charles MA, Ortega AC, & Solis-Camara P
(1983) Lead concentrations in the blood of children from
pottery-making families exposed to lead salts in a Mexican village.
Bull Panam Health Organ, 17: 35-41.
Möller L & Kristensen TS (1992) Blood lead as a cardiovascular risk
factor. Am J Epidemiol, 136: 1091-1100.
Moore MR (1983) Lead exposure and lead plumbosolvency. In: Rutter M &
Jones RR ed. Lead versus health. London, John Wiley and Sons,
pp 79-106.
Moore MR & Meredith PA (1979) The carcinogenicity of lead. Arch
Toxicol, 42: 87-94.
Moore MR, Meredith PA, Campbell BC, & Watson SW (1979) The
gastrointestinal absorption of lead 203 chlorine in man. In: Hemphill
DD ed. Trace substances in environmental health - XIII. Proceedings of
the 13th Annual Conference on Trace Substances in Environmental
Health, Columbia, June 1979. Columbia, Missouri, University of
Missouri, pp 368-373.
Moore MR, Meredith PA, & Goldberg A (1980) Lead and heme biosynthesis.
In: Singhal PL & Thomas JA ed. Lead toxicity. Baltimore, Maryland,
Urban and Schwarzenberg, Inc., pp 79-118.
Moore MR, Goldberg A, & Pocock SJ (1982) Some studies of maternal and
infant lead exposure in Glasgow. Scott Med J, 27: 113-122.
Moore MR, Bushnell IWR, & Goldberg A (1989) A prospective study of the
results of changes in environmental lead exposure in children in
Glasgow. In: Smith MA, Grant LD, & Sors AI ed. Lead exposure and child
development: An international assessment. Dordrecht, London, Kluwer
Academic Publishers, pp 371-378.
Moore MR, Goldberg A, & Young-Laiwah AAC (1987) Lead effects on the
haem-biosynthetic pathway. Relationship to toxicity. Ann NY Acad Sci,
514: 191-203.
Mooty J, Ferrand CF, & Harris P (1975) Relationship of diet to lead
poisoning in children. Pediatrics, 55: 636-639.
Moreau T, Orssaud G, Juguet B, & Busquet G (1982) Plombémie et
pression artérielle: premiers résultats d'une enquête transversale de
431 sujets de sexe masculin. Rev Epidemiol Santé Publique,
30: 395-397.
Morgan A & Holmes A (1978) The fate of lead in petrol-engine exhaust
particulates inhaled by the rat. Environ Res, 15: 44-56.
Muijser H, Hoogendijk EMG, Hooisma J, & Twisk DAM (1987) Lead exposure
during demolition of a steel structure coated with lead-based paints.
II. Reversible changes in the conduction velocity of the motor nerves
in transiently exposed workers. Scand J Work Environ Health,
13: 56-61.
Munoz C, Garbe K, Lilienthal H, & Winneke G (1986) Persistence of
retention deficit in rats after neonatal lead exposure.
Neurotoxicology, 7(2): 569-580.
Muramatsu Y & Parr RM (1985) Survey of currently available reference
materials for use in connection with the determination of trace
elements in biological and environmental materials. Vienna,
International Atomic Energy Agency (Report No. IAEA/RL/128).
Murata K & Araki S (1991) Autonomic nervous system dysfunction in
workers exposed to lead, zinc, and copper in relation to peripheral
nerve conduction: a study of R-R interval variability. Am J Ind Med,
20: 663-671.
Murphy MJ, Graziano JH, Popovac D, Kline JK, Mehmeti A, Factor-Litvak
P, Ahmadi G, Shrout P, Rajovic B, Nenezic DU, & Stein ZA (1990) Past
pregnancy outcomes among women living in the vicinity of a lead
smelter in Kosovo, Yugoslavia. Am J Public Health, 80: 33-35.
Mushak P (1989) Biological monitoring of lead exposure in children:
overview of selected biokinetic and toxicological issues. In: Smith
MA, Grant LD, & Sors AI ed. Lead exposure and child development: An
international assessment. Dordrecht, London, Kluwer Academic
Publishers, pp 129-145.
Mushak P & Crocetti AF (1989) Determination of numbers of lead-exposed
American children as a function of lead source: Integrated summary of
a report to the U.S. Congress on childhood lead poisoning. Environ
Res, 50: 210-299.
Mussalo-Rauhamaa H, Salmela SS, Leppanen A, & Pyysalo H (1986)
Cigarettes as a source of some trace and heavy metals and pesticides
in man. Arch Environ Health, 41: 49-55.
NAS-NRC (1972) Airborne lead in perspective. Washington, DC, National
Academy of Sciences.
Needleman HL & Gatsonis CA (1990) Low-level lead exposure and the IQ
of children: a meta-analysis of modern studies. J Am Med Assoc,
263: 673-678.
Needleman HL, Tuncay OC, & Shapiro IM (1972) Lead levels in deciduous
teeth of urban and suburban children. Nature (Lond), 235: 111-112.
Needleman HL, Gunnoe C, Leviton A, Reed R, Peresie H, Maher C, &
Barrett P (1979) Deficits in psychologic and classroom performance of
children with elevated dentine lead levels. N Engl J Med,
300: 689-695.
Needleman HL, Schell A, Bellinger D, Leviton A, & Allred EN (1990) The
long-term effects of childhood exposure to low doses of lead: An
11-year follow-up report. N Engl J Med, 322: 83-88.
Neri LC, Hewitt D, & Orser B (1988) Blood lead and blood pressure:
analysis of cross-sectional and longitudinal data from Canada. Environ
Health Perspect, 78: 123-126.
Nerin C, Olavide S, & Cacho J (1989) Determination of lead in airborne
particulate by hydride generation. Water Air Soil Pollut, 44: 339-345.
Neubecker TA & Allen HE (1983) The measurement of complexation
capacity and conditional stability constants for ligands in natural
waters. Water Res, 17: 1-14.
NFA (Australian National Food Authority, National Health and Medical
Council) (1991) The 1990 Australian market basket survey report.
Canberra, Commonwealth of Australia, Australian Government Publishing
Service.
Ng A & Patterson CC (1982) Changes of Pb and Ba with time in
California offshore basin sediments. Geochim Cosmochim Acta,
46: 2307-2322.
Ng TP, Goh HH, Ng YL, Ong CN, Chia KS, Chia SE, & Jeyaratnam J (1991)
Male endocrine functions in workers with moderate exposure to lead. Br
J Ind Med, 48: 485-491.
Nielsen T (1984) Atmospheric occurrence of organolead compounds. In:
Grandjean P ed. Biological effects of organolead compounds. Boca
Raton, Florida, CRC Press, pp 43-62.
Nilsson U, Attewell R, Christoffersson J-O, Schütz A, Ahlgren L,
Skerfving S, & Mattsson S (1991) Kinetic of lead in bone and blood
after end of occupational exposure. Pharmacol Toxicol, 69: 477-484.
NIOSH (1977a) Method No. P&CAM 102. In: Manual of analytical methods,
vol 1, 2nd ed. Cincinnati, Ohio, National Institute for Occupational
Safety and Health.
NIOSH (1977b) Method No. P&CAM 191. In: Manual of analytical methods,
vol 1, 2nd ed. Cincinnati, Ohio, National Institute for Occupational
Safety and Health, pp 191/1-191/9.
NIOSH (1977c) Method No. P&CAM 195. In: Manual of analytical methods,
vol 1, 2nd ed. Cincinnati, Ohio, National Institute for Occupational
Safety and Health.
NIOSH (1977d) Method No. P&CAM 200. In: Manual of analytical methods,
vol 1, 2nd ed. Cincinnati, Ohio, National Institute for Occupational
Safety and Health, pp 200/1-200/8.
NIOSH (1977e) Method No. P&CAM 214. In: Manual of analytical methods,
vol 1, 2nd ed. Cincinnati, Ohio, National Institute for Occupational
Safety and Health, pp 214/1-214/6.
NIOSH (1981) Method No. P&CAM 351. In: Manual of analytical methods,
vol 7. Cincinnati, Ohio, National Institute for Occupational Safety
and Health, pp 351/1-351/11.
Nordenson I, Beckman G, Beckman L, & Nordstrom S (1978) Occupational
and environmental risks in and around a smelter in northern Sweden:
IV. Chromosomal aberrations in workers exposed to lead. Hereditas,
88: 263-267.
Nordstrom S, Beckman L, & Nordenson I (1978a) Occupational and
environmental risks in and around a smelter in northern Sweden: I.
Frequencies in birth weight. Hereditas, 88: 43-46.
Nordstrom S, Beckman L, & Nordenson I (1978b) Occupational and
environmental risks in and around a smelter in northern Sweden: III.
Frequencies of spontaneous abortion. Hereditas, 88: 51-54.
Nordstrom S, Beckman L, & Nordenson I (1979) Occupational and
environmental risks around a smelter in northern Sweden: V.
Spontaneous abortion among female employees and decreased birth weight
in their offspring. Hereditas, 90: 291-296.
Nosal RM & Wilhelm WJ (1990) Lead toxicity in the shipbreaking
industry: the Ontario experience. Can J Public Health, 81: 259-262.
Novotny T, Cook M, Hughes J, & Lee SA (1987) Lead exposure in a firing
range. Am J Public Health, 77: 1225-1226.
Nriagu J (1979) Global inventory of natural and anthropogenic
emissions of trace metals to the atmosphere. Nature (Lond),
279: 409-411.
Nriagu J (1989) A global assessment of natural sources of atmospheric
trace metals. Nature (Lond), 338: 47-49.
Nriagu J & Pacyna JM (1988) Quantitative assessment of worldwide
contamination of air, water, and soils by trace metals. Nature (Lond),
333: 134-139.
OCTEL (1991) Worldwide survey of motor gasoline quality. London,
Associated Octel Company Ltd (Octel Promotional Publication No. 4).
OECD (1993) Risk Reduction Monograph No. 1: Lead background and
national experience with reducing risk. Paris, Organisation for
Economic Co-operation and Development, 277 pp (Report No.
OCDE/GD(93)67).
O'Flaherty EJ (1991) Physiologically based lead kinetics. Trace Subst
Environ Health, 24: 44-54.
O'Flaherty EJ (1993) Physiologically based models for bone-seeking
elements. IV. Kinetics of lead disposition in humans. Toxicol Appl
Pharmacol, 118: 16-29.
O'Flaherty EJ, Hammond PB, Lerner SI, Hanenson IB, & Roda SMB (1980)
The renal handling of delta-aminolevulinic acid in the rat and in the
human. Toxicol Appl Pharmacol, 55: 423-432.
O'Flaherty EJ, Hammond PB, & Lerner SI (1982) Dependence of apparent
blood lead half-life on the length of previous lead exposure in
humans. Fundam Appl Toxicol, 2: 49-54.
Olejnik D, Walkowska A, Wisnilwska J, & Ziembinski R (1985)
[Evaluation of the daily dose of mercury, lead and cadmium in the diet
of certain population groups.] Roczn Pzh, XXXVI: 9-21 (in Polish, with
English abstract).
Olson KW & Skogerboe RK (1975) Identification of soil lead compounds
from automotive sources. Environ Sci Technol, 9: 227-230.
Ong CN & Lee WR (1980) High affinity of lead for fetal hemoglobin. Br
J Ind Med, 37: 292-298.
Ong CN, Phoon WO, Law HY, Tye CY, & Lim HH (1985) Concentration of
lead in maternal blood, cord blood, and breast milk. Arch Dis Child,
60: 576-759.
Ong CN, Endo G, & Chia KS (1987) Evaluation of renal function in
workers with low blood lead levels. In: Occupational and environmental
chemical hazards: Cellular and biochemical indices for monitoring
toxicity. Chichester, Ellis Horwood Ltd, chapter 39, pp 327-333.
Orfila MP (1814) Traité des poisons. Paris, Crochard, vol 2.
Orssaud G, Claude JR, Moreau T, Lellouch J, Juguet B, & Festy B (1985)
Blood lead concentration and blood pressure. Br Med J, 290: 244.
Padilla F, Shapiro AP, & Jensen WN (1969) Effect of chronic lead
intoxication on blood pressure in the rat. Am J Med Sci, 258: 359-365.
Paglia DE, Valentine WN, & Dahlgren JG (1975) Effects of low-level
lead exposure on pyrimidine 5'-nucleotidase and other erythrocyte
enzymes: possible role of pyrimidine 5'-nucleotidase in the
pathogenesis of lead-induced anaemia. J Clin Invest, 56: 1164-1169.
Pagliuca A, Mufti GJ, Baldwin D, Lestas AN, Wallis RM, & Bellingham AJ
(1990) Lead poisoning: clinical, biochemical, and haematological
aspects of a recent outbreak. J Clin Pathol, 43: 277-281.
Parfitt AM (1990) Pharmacologic manipulation of bone remodelling and
calcium homeostasis: Calcium metabolism. Prog Basic Clin Pharmacol,
4: 1-27.
Parkinson DK, Ryan C, Brommet EJ, & Connell MM (1986) A psychiatric
epidemiologic study of occupational lead exposure. Am J Epidemiol,
123: 261-269.
Parkinson DK, Ryan C, Bromet J, & Connell MM (1987) A psychiatric
epidemiological study of occupationally exposure. Am J Epidemiol,
123: 261-269.
Patterson C (1965) Contaminated and natural lead environments of man.
Arch Environ Health, 11: 344-360.
Patterson CC (1983) Criticism of "flow of metals into the global
atmosphere". Geochim Cosmochim Acta, 47: 1163-1168.
Patterson CC & Settle DM (1976) The reduction of orders of magnitude
errors in lead analyses of biological materials and natural waters by
evaluating and controlling the extent and source of industrial lead
contamination introduced during sample collecting, handling, and
analyses. In: LaFleur PD ed. Accuracy in trace analysis: Sampling,
sample handling and analysis. Volume 1. Proceedings of the 7th
Materials Research Symposium, Gaithersburg, Maryland, October, 1974.
Washington, DC, US Department of Commerce, National Bureau of
Standards, pp 321-352 (NBS Special Publication No. 422).
Pekkarinen M (1970) Methodology in the collecting of food consumption
data. World Rev Nutr Diet, 12: 145-170.
Pentschew A (1965) Morphology and morphogenesis of lead
encephalopathy. Acta Neuropathol, 5: 133-160.
Pentschew A & Garro F (1966) Lead encephalo-myelopathy of the suckling
rat and its implications on the porphyrinopathic nervous diseases with
special reference to the permeability/disorders of the nervous
system's capillaries. Acta Neuropathol, 6: 266-278.
Perino J & Ernhart CB (1974) The relation of subclinical lead level to
cognitive and sensorimotor impairment in black preschoolers. J Learn
Disabil, 7: 616-620.
Pickston L, Brewerton HV, Drysdale JM, Hughes JT, Smith JM, Love JL,
Sutcliffe ER, & Davidson F (1985) The New Zealand diet: a survey of
elements, pesticides, colours and preservatives. N Z J Technol,
1: 81-89.
Piomelli S, Seaman C, Zullow D, Curran A, & Davidow B (1982) Threshold
for lead damage to heme synthesis in urban children. Proc Natl Acad
Sci (USA), 79: 3335-3339.
Piper W & Tephly T (1974) Differential inhibition of erythrocyte and
hepatic uroporphyrinogen. I. Synthelase activity by lead. Life Sci,
14: 873-876.
Pirkle JL, Schwartz J, Lamdis JR, & Harlan WR (1985) The relationship
between blood lead levels and blood pressure and its cardiovascular
risk implications. Am J Epidemiol, 121: 246-258.
Pocock SJ, Shaper AG, Walker M, Wale CJ, Clayton B, Delves T, Lacey
RF, Packham RF, & Powell P (1983) Effects of tap water lead, water
hardness, alcohol, and cigarettes on blood lead concentrations. J
Epidemiol Community Health, 37: 1-7.
Pocock SJ, Shaper AG, Ashby D, Delves T, & Whitehead TP (1984) Blood
lead concentration, blood pressure, and renal function. Br Med J,
289: 872-874.
Pocock S, Ashby D, & Smith MA (1987) Lead exposure and children's
intellectual performance. Int J Epidemiol, 16: 57-67.
Pocock SJ, Shaper AG, Ashby D, Delves HT, & Clayton BE (1988) The
relationship between blood lead, blood pressure, stroke and heart
attacks in middle-aged British men. Environ Health Perspect,
78: 23-30.
Pocock SJ, Ashby D, & Smith MA (1989) Lead exposure and children's
intellectual performance. The Institute of Child Health/Southampton
Study. In: Smith M, Grant LD, & Sors A ed. Lead exposure and child
development: An international assessment. Dordrecht, London, Kluwer
Academic Press, pp 149-165.
Pollock CA & Ibels LS (1986) Lead intoxication in paint removal
workers on the Sydney Harbour Bridge. Med J Aust, 145: 635-639.
Ponka A, Salminen E, & Ahonen S (1991) Lead in the ambient air and
blood specimens of children in Helsinki. Sci Total Environ, 138(1-3):
301-308.
Pontifax AH & Garg AK (1985) Lead poisoning from an Asian Indian folk
remedy. Can Med Assoc J, 133: 1227-1228.
Poole C, Smythe LE, & Alpers M (1980) Blood lead levels of Papua New
Guinea children living in a remote area. Sci Total Environ, 15: 17-24.
Pounds JG (1984) Effect of lead intoxication on calcium homeostasis
and calcium-mediated cell function: a review. Neurotoxicology,
5: 295-332.
Pounds JG, Marlar RJ, & Allen JR (1978) Metabolism of lead-210 in
juvenile and adult rhesus monkeys Macaca mulatta. Bull Environ Contam
Toxicol, 19: 684-691.
Pounds JG, Long GJ, & Rosen JF (1991) Cellular and molecular toxicity
of lead in bone. Environ Health Perspect, 91: 17-32.
Purchase NG & Fergusson JE (1986) Lead in teeth: the influence of the
tooth type and the sample within a tooth on lead levels. Sci Total
Environ, 52: 239-250.
Qazi QH, Madahar C, & Huceoglu AM (1980) Temporary increase in
chromosome breakage in an infant prenatally exposed to lead. Hum
Genet, 53: 201-203.
Qian ZM & Morgan EH (1990) Effect of lead on the transport of
transferrin-free and transferrin-bound iron into rabbit reticulocytes.
Biochem Pharmacol, 40: 1049-1054.
Quarterman J & Morrison JN (1975) The effects of dietary calcium and
phosphorus on the retention and excretion of lead in rats. Br J Nutr,
34: 351-362.
Que Hee SS & Boyle JR (1988) Simultaneous multielemental analysis of
some environmental and biological samples by inductively coupled
plasma atomic emission spectrometry. Anal Chem, 60: 1033-1042.
Que Hee SS, MacDonald TJ, & Bornschein RL (1985a) Blood lead by
furnace-Zeeman atomic absorption spectrophotometry. Microchem J,
32: 55-63.
Que Hee SS, Peace B, Clark CS, Boyle JR, Bornschein RL, & Hammond PB
(1985b) Evolution of efficient methods to sample lead sources, such as
house dust and hand dust in the homes of children. Environ Res,
38: 77-95.
Raab GM, Laxen DPH, & Fulton M (1987) Lead from dust and water as
exposure sources for children. Environ Geochem Health, 9: 80.
Rabinowitz MB (1991) Toxicokinetics of bone lead. Environ Health
Perspect, 91: 33-37.
Rabinowitz M & Needleman HL (1982) Temporal trends in the lead
concentrations of umbilical cord blood. Science, 216: 1429-1431.
Rabinowitz MB, Wetherill G, & Kopple J (1975) Absorption, storage and
excretion of lead by normal humans. In: Proceedings of the Ninth
Annual Conference on Trace Substances in Environmental Health.
Columbia, Missouri, University of Missouri, pp 361-368.
Rabinowitz MB, Wetherill GM, & Kopple JD (1976) Kinetics analysis of
lead metabolism in healthy humans. J Clin Invest, 58: 260-270.
Rabinowitz MB, Wetherill GW, & Kopple JD (1977a) Magnitude of lead
intake from respiration by normal man. J Lab Clin Med, 90: 238-248.
Rabinowitz MB, Wetherill GW, & Kopple JD (1977b) Effect of food intake
and fasting on gastrointestinal lead absorption in humans. Am J Clin
Nutr, 33: 1784-1788.
Rabinowitz MB, Kopple JD, & Wetherill GW (1980) Effect of food intake
and fasting on gastrointestinal lead absorption in humans. Am J Clin
Nutr, 33: 1784-1788.
Rabinowitz MB, Leviton A, & Needleman HL (1986) Occurrence of elevated
protoporphyrin levels in relation to lead burden in infants. Environ
Res, 39: 253-257.
Rabinowitz M, Bellinger D, Leviton A, Needleman H, & Schoenbaum S
(1987) Pregnancy hypertension, blood pressure during labor, and blood
lead levels. Hypertension, 10: 447-451.
Rabinowitz MB, Leviton A, & Bellinger D (1989) Blood lead-tooth lead
relationship among Boston children. Bull Environ Contam Toxicol,
43: 485-492.
Rabinowitz MB, Wang J-D, & Soong WT (1991) Dentine lead and child
intelligence in Taiwan. Arch Environ Health, 46: 351-360.
Ragan HA (1977) Effect of iron deficiency on the absorption of lead
and cadmium in the rat. J Lab Clin Med, 90: 700-706.
Reaves GA & Berrow ML (1984) Total lead concentrations in Scottish
soils. Geoderma, 32: 1-8.
Reisinger K, Stoeppler M, & Nurnberg HW (1981) Evidence for the
absence of biological methylation of lead in the environment. Nature
(Lond), 281: 228-230.
Reuter JH & Perdue EM (1977) Importance of heavy metal-organic matter
interactions in natural waters. Geochim Cosmochim Acta, 41: 325-334.
Rhagavan SRV, Dwight Culver B, & Gonick HC (1981) Erythrocyte
lead-binding protein after occupational exposure. II. Influence of
lead inhibition of membrane Na+, K±-adenosinetriphosphatase. J Toxicol
Environ Health, 7: 561-568.
Rice DC (1984a) Behavioural deficit (delayed matching to sample) in
monkeys exposed from birth to low level lead. Toxicol Appl Pharmacol,
75: 337-345.
Rice DC (1984b) Effect of lead on schedule-controlled behavior in the
monkey. In: Seiden LS & Balster RL ed. Behavioral pharmacology: The
current status. New York, Alan R. Liss, pp 473-486.
Rice DC (1985) Chronic low-lead exposure from both produces deficits
in discrimination reversal monkey's. Toxicol Appl Pharmacol,
77: 201-210.
Rice DC & Karpinski KF (1988) Lifetime dow level lead exposure
produces deficits in delayed alternation in adult monkeys.
Neurotoxicol Teratol, 10: 207-214.
Roberts J, Mahaffey KR, & Annest JL (1985) Blood lead levels in
general populations. In: Mahaffey KR ed. Dietary and environmental
lead: Human health effects. Amsterdam, Oxford, New York, Elsevier
Science Publishers, pp 355-372.
Robins JM, Cullen MR, Connors BB, & Kayne RD (1983) Depressed thyroid
indexes associated with occupational exposure to inorganic lead. Arch
Intern Med, 143: 220-224.
Robinson TR (1974) Delta-aminolevulinic acid and lead in urine of lead
antiknock workers. Arch Environ Health, 28: 133-138.
Rodamilans M, Osaba MJM, To-Figueras J, Fillat FR, Marques JM, Perez
P, & Corbella J (1988) Lead toxicity on endocrine testicular function
in an occupationally exposed population. Hum Toxicol, 7: 125-128.
Rodney BS & Lee MG (1985) Environmental lead hazard in a high-risk
Jamaican population. West Indian Med J, 34: 176-179.
Roels HA & Lauwerys R (1987) Evaluation of dose-effect and
dose-response relationships for lead exposure in different Belgian
population groups (foetus, child, adult men and women). Trace Elem
Med, 4: 80-87.
Roels HA, Buchet J-P, Lauwerys R, Hubermont G, Bruaux P, Claeys-
Thoreau F, Lafontaine A, & van Overschelde J (1976) Impact of air
pollution by lead on the haem biosynthetic pathway in school-age
children. Arch Environ Health, 31: 310-316.
Roels HA, Buchet JP, Lauwerys RR, Bruaux P, Claeys-Thoreau F,
Lafontaine A, & Verduyn G (1980) Exposure to lead by the oral and the
pulmonary routes of children living in the vicinity of a primary lead
smelter. Environ Res, 22: 81-94.
Rosen JF & Chesney RW (1983) Circulating calcitriol concentration in
health and disease. J Pediatr, 103: 1-7.
Rosen JF, Chesney RW, Hamstra AJ, Deluca HF, & Mahaffey KR (1980)
Reduction in 1,25-dihydroxyvitamin D in children with increased lead
absorption. N Engl J Med, 302: 1128-1131.
Rosen JF, Markowitz ME, Bijur PE, Jenks ST, Wielopolski L, Kalef-Ezra
JA, & Slatkin DN (1991) Sequential measurements of bone lead content
by L x-ray fluorescence in CaNa2EDTA-treated lead-toxic children.
Environ Health Perspect, 91: 57-62.
Rosner D & Markowitz G (1985) A "gift of God": The public health
controversy over leaded gasoline during the 1920s. Am J Public Health,
75(4): 344.
Rossouw J, Offermeier J, & van Rooyen J (1987) Apparent central
neurotransmitter receptor changes induced by low-level lead exposure
during different developmental phases in the rat. Toxicol Appl
Pharmacol, 91: 132-139.
Routh DK, Mushak P, & Boone L (1979) A new syndrome of elevated blood
lead and microcephaly. J Pediatr Psychol, 4: 67-76.
Rundle SA & Duggan MJ (1986) Lead pollution from the external
redecoration of old buildings. Sci Total Environ, 57: 181-190.
Ryu JE, Ziegler EE, Nelson SE, & Fomon SJ (1983) Dietary intake of
lead and blood lead concentration in early infancy. Am J Dis Child,
137: 886-891.
Ryu JE, Ziegler EE, Nelson SE, & Fomon SJ (1985) Dietary and
environmental exposure to lead and blood lead during early infancy.
In: Mahaffey KR ed. Dietary and environmental lead: Human health
effects. Amsterdam, Oxford, New York, Elsevier Science Publishers,
pp 187-209.
Saenger P, Markowitz MR, & Rosen JF (1984) Depressed excretion of
6ß-hydroxycortisol in lead-toxic children. J Clin Endocrinol Metab,
58: 363-367.
SAHC (1993) Proceedings of the International Meeting on
Non-Occupational Exposure to Lead, Melbourne, Australia, 5-9 October
1992. Adelaide, South Australian Health Commission, 56 pp.
Sarto F, Stella M, & Acqua A (1978) [Cytogenic studies in 20 workers
occupationally exposed to lead.] Med Lav, 69: 172-180 (in Italian).
Satzger RD, Clow CS, & Bonnin E (1982) Determination of background
levels of lead and cadmium in raw agricultural crops by using
differential pulse anodic stripping voltammetry. J Assoc Off Anal
Chem, 65: 987-991.
Sauk J & Somerman MJ (1991) Physiology of bone: mineral compartment
proteins as candidates for environmental perturbation by lead. Environ
Health Perspect, 91: 9-16.
Schlegel H & Kufner G (1979) Long-term observation of biochemical
effects of lead in human experiments. J Clin Chem Clin Biochem,
17: 225-233.
Schmid E, Bauchinger M, Pietruk S, & Hall G (1972) [Cytogenic action
of lead in human peripheral lymphocytes in vitro and in vivo.]
Mutat Res, 16: 401-406 (in German).
Schmidt U & Huber F (1976) Methylation of organolead and lead(II)
compounds (CH3)4 Pb by microorganisms. Nature (Lond),
259: 157-158.
Schmitt MDC, Trippler DL, & Wachtler JN (1988) Soil lead
concentrations in residential Minnesota as measured by ICP AES. Water
Air Soil Pollut, 39: 157-168.
Schneitzer L, Osborn HH, Bierman A, Mezey A, & Kaul B (1990) Lead
poisoning in adults from renovation of an older home. Ann Emerg Med,
19: 415-420.
Schroeder SR, Hawk B, Otto DA, Mushak P, & Hicks RE (1985) Separating
the effects of lead and social factors on IQ. Environ Res,
38: 144-154.
Schutz AS, Skerfving S, Ranstam J, & Christoffersson JO (1987)
Kinetics of lead in blood after the end of occupational exposure.
Scand J Work Environ Health, 13: 221-231.
Schutz A, Ranstam J, Skerfving S, & Tejning S (1989) Blood-lead levels
in school children in relation to industrial emission and automobile
exhausts. Ambio, 13: 115-117.
Schwartz J (1988) The relationship between blood lead and blood
pressure in the NHANES II survey. Environ Health Perspect, 78: 15-22.
Schwartz J (1993) Low-level lead exposure and children's IQ: a
meta-analysis and search for a threshold. Environ Res, 65: 42-55.
Schwartz J & Otto D (1987) Blood lead, hearing thresholds, and
neurobehavioral development in children and youth. Arch Environ
Health, 42: 153-160.
Schwartz J & Otto D (1991) Lead and minor hearing impairment. Arch
Environ Health, 46: 300-305.
Schwartz J, Angle C, & Pitcher H (1986) Relationship between childhood
blood lead levels and stature. Pediatrics, 77: 281-288.
Schwartz J, Landrigan PL, & Baker EL (1990) Lead-induced anemia:
dose-response relationships and evidence for a threshold. Am J Public
Health, 80: 165-168.
Scott DR, Hemphill DC, & Holboke LE (1976) Atomic absorption and
optical emission analysis of NASN atmospheric particulate samples for
lead. Environ Sci Technol, 9: 877-880.
Selander S & Cramer K (1970) Interrelationships between lead in blood,
lead in urine and ALA in urine during lead work. Br J Ind Med,
27: 28-39.
Selevan SG, Landrigan PJ, Stern FB, & Jones JH (1985) Mortality of
lead smelter workers. Am J Epidemiol, 122: 673-683.
Selevan SG, Landrigan PJ, Stern FB, & Jones JH (1988) Brief report:
lead and hypertension in a mortality study of lead smelter workers.
Environ Health Perspect, 78: 65-66.
Seppäläinen AM & Hernberg S (1980) Subclinical lead neuropathy. Am J
Ind Med, 1: 413-420.
Seppäläinen AM, Hernberg S, Vesanto R, & Kock B (1983) Early
neurotoxic effects of occupational lead exposure: a prospective study.
Neurotoxicology, 4(2): 181-192.
Settle DM & Patterson CC (1980) Lead in albacore: guide to lead
pollution in Americans. Science, 207: 1167-1176.
Shaltout A, Yaish SA, & Fernando N (1981) Lead encephalopathy in
infants in Kuwait. Ann Trop Paediatr (London), 1: 209-215.
Shaper AG, Pocock SJ, Walker M, Cohen NM, Wale CJ, & Thompson AG
(1981) British regional heart study: cardiovascular risk factors in
middle-aged men in 24 towns. Br Med J, 283: 179-186.
Sharma RR, Chandy MJ, & Lad SD (1990) Transient hydrocephalis and
acute lead encephalopathy in neonates and infants. Report of two
cases. Br J Neurosurg, 4: 141-146.
Sherlock J, Smart G, Forbes GI, Moore MR, Patterson WJ, Richards WN, &
Wilson TS (1982) Assessment of lead intakes and dose-response for a
population in Ayr exposed to a plumbsolvent water supply. Hum Toxicol,
1: 115-122.
Sherlock JC, Pickford CJ, & White GF (1986) Lead in alcoholic
beverages. Food Addit Contam, 3: 347-354.
Siegel M, Forsyth B, Siegel L, & Cullen MR (1989) The effect of lead
on thyroid function in children. Environ Res, 49: 190-196.
Silbergeld EK (1991) Lead in bone: implications for toxicology during
pregnancy and lactation. Environ Health Perspect, 91: 63-70.
Silbergeld EK, Schwartz J, & Mahaffey KR (1988) Lead and osteoporosis:
mobilization of lead from bone in postmenopausal women. Environ Res,
47: 79-94.
Silva PA, Hughes P, Williams S, & Faed JM (1988) Blood lead,
intelligence, reading attainment, and behaviour in eleven year old
children in Dunedin, New Zealand. J Child Psychol Psychiatry Appl
Discipl, 29: 43-52.
Sinclair DF & Dohnt BR (1984) Sampling and analysis techniques used in
a blood lead survey of 1341 children in Port Pirie, South Australia.
Clin Chem, 30(10): 1616-1619.
Skerfving S (1988) Biological monitoring of exposure to inorganic
lead. In: Clarkson TW, Friberg L, Nordberg GF, & Sager PR ed.
Biological monitoring of toxic metals. New York, Plenum Press,
pp 169-197 (Rochester Series on Environmental Toxicity).
Skerfving S, Schutz A, & Ranstam J (1986) Decreasing lead exposure in
Swedish children. Sci Total Environ, 58: 225-229.
Skogerboe RK, Hartley AM, Vogel RS, & Koirtyohann SR (1977) Monitoring
for lead in the environment. In: Boggess WR ed. Lead in the
environment. Washington, DC, National Science Foundation, pp 33-70
(NSF Report No. NSF/RA-770214; PB-278278).
Slorach SA, Gustafsson I-B, Jorhem L, & Mattsson P (1983) Intake of
lead, cadmium and certain other metals via a typical Swedish weekly
diet. Var Föda, 35(Suppl 1): 3-16.
Smith MA (1989) The effects of low-level lead exposure on children.
In: Smith MA, Grant LD, & Sors AI ed. Lead exposure and child
development: An international assessment. Dorcrecht, London, Kluwer
Academic Publishers, pp 3-47.
Smith F & Hursh J (1977) Bone storage and release. In: Field J ed.
Handbook of physiology: Reaction to environmental agents. Bethesda,
Maryland, American Physiological Society, section 9, pp 469-482.
Smith M, Delves T, Lansdown R, Clayton B, & Graham P (1983) The
effects of lead exposure on urban children: the Institute of Child
Health/Southampton Study. Dev Med Child Neurol, 47(Suppl): 1-54.
Snee RD (1981) Evaluation of studies of the relationship between blood
lead and air lead. Int Arch Occup Environ Health, 48: 219-242.
Sobel AE, Yuska H, Peters DD, & Kramer B (1940) The biochemical
behaviour of lead. I. Influence of calcium phosphorus and vitamin D on
lead in the blood and bone. J Biol Chem, 132: 239-265.
Sokol RZ (1987) Hormonal effects of lead acetate in the male rat:
Mechanism of action. Biol Reprod, 37: 1135-1138.
Sokol RZ, Madding CE, & Swerdloff RS (1985) Lead toxicity and the
hypothalamic-pituitary-testicular axis. Biol Reprod, 33: 722-728.
Spivey GH, Baloh RW, Brown CP, Browdy BL, Campion DS, Valentine JL,
Morgan DE, & Culver BD (1980) Subclinical effects of chronic increased
lead absorption - a prospective study: III. Neurologic findings at
follow-up examination. J Occup Med, 22: 607-612.
Staessen J, Yeoman WB, Fletcher AE, Markowe HLJ, Marmot MG, Rose G,
Semmence A, Shipley MJ, & Bulpitt CJ (1990) Blood lead concentration,
renal function, and blood pressure in London civil servants. Br J Ind
Med, 47: 442-447.
Staessen JA, Lauwerys RA, Buchet J-P, Bulpitt CJ, Rondia D,
Vanrenterghem Y, Amery A, & The Cadmibel Study Group (1992) Impairment
of renal function with increasing blood lead concentrations in the
general population. N Engl J Med, 327: 151-156.
Staessen JA, Christopher JB, Fagard R, Lauwerys RR, Roels H, Thijs L,
& Amery A (1994) Hypertension caused by low-level lead exposure: myth
or fact? J Cardiovasc Risk, 1: 87-97.
Steelink C (1977) Humates and other natural organic substances in the
aquatic environment. J Chem Ed, 54: 599-603.
Steenhout A (1987) How clean is clean? An exotoxicological method for
getting guidelines (air, dust, deposition, water) for lead, keeping
tooth and blood lead levels in the normal range. In: Proceedings of
the International Conference on Heavy Metals in the Environment.
Edinburgh, CEP Consultants, pp 283-285.
Steenhout A & Pourtois M (1981) Lead accumulation in teeth as a
function of age with different exposures. Br J Ind Med, 38: 297-303.
Stiles KM & Bellinger DC (1993) Neuropsychological correlates of
low-level lead exposure in school-age children: A prospective study.
Neurotoxicol Teratol, 15(1): 27-35.
Stollery BT, Banks HA, Broadbent DE, & Lee WR (1989) Cognitive
functioning in lead workers. Br J Ind Med, 46: 698-707.
Stollery BT, Broadbent DE, Banks HA, & Lee WR (1991) Short term
prospective study of cognitive functioning in lead workers. Br J Ind
Med, 48: 739-749.
Stone CH & Soares JH (1976) The effect of dietary selenium level of
lead toxicity in the Japanese quail. Poult Sci, 55: 341-349.
Stone C, Fox MRS, & Hogye KS (1981) Bioavailability of lead in oysters
fed to young Japanese quail. Environ Res, 26: 409-421.
Stuik EJ (1974) Biological response of male and female volunteers to
inorganic lead. Int Arch Arbeitsmed, 33: 83-97.
Succop PA, O'Flaherty EJ, Bornschein RL, Roba SB, & Greenland RD
(1989) Recent observations concerning the relationship of blood lead
to erythrocytic protoporphyrin. In: Smith MA, Grant LD, & Sors AE ed.
Lead exposure and child development: An international Assessment.
Dordrecht, London, Kluwer Academic Publishers, pp 477-484.
Talbot RW & Andren AW (1983) Relationships between Pb and 210Pb in
aerosol and precipitation at a semiremote site in northern Wisconsin.
J Geophys Res, 88: 6752-6760.
Taskinen H, Nordman H, Engstrom K, & Hernberg S (1981) [Blood lead
levels in Finnish children.] Duodecim, 97(10): 663-670 (in Finnish).
Teruya K, Sakurai H, Omae K, Higashi T, Muto T, & Kaneko Y (1991)
Effect of lead on cardiac parasympathetic function. Int Arch Occup
Environ Health, 62: 549-553.
Thornton I & Webb JS (1975) Trace elements in soils and surface waters
contaminated plast metaliferous mining part of England. In:
Proceedings of the IXth Annual Conference on Trace Substances in
Environmental Health, Columbia, Missouri, 10-12 June, pp 77-88.
Thornton I, Culbard E, Moorcroft S, Watt J, Wheatley M, & Thompson M
(1985) Metals in urban dusts and soils. Environ Technol Lett,
6: 137-144.
Todd AC, McNeill FE, Palethorpe JE, Peach DE, Chettle DR, Tobin MJ,
Strosko SJ, & Rosen JC (1992) In vivo x-ray fluorescence of lead in
bone using K x-ray excitation with 109Cd sources; radiation dosimetry
studies. Environ Res, 57: 117-132.
Triebig G, Weltle D, & Valentin H (1984) Investigations on
neurotoxicity of chemical substances at the workplace. V.
Determination of the motor and sensory nerve conduction velocity in
persons occupationally exposed to lead. Int Arch Occup Environ Health,
53: 189-204.
Trotter RT (1990) The cultural parameters of lead poisoning: A medical
anthropologist's view of intervention in environmental lead exposure.
Environ Health Perspect, 89: 79-84.
Tuppurainen M, Wagar G, Kurppa K, Sakari W, Wambugu A, Froseth B, Alho
J, & Nykyri E (1988) Thyroid function as assessed by routine
laboratory tests of workers with long-term lead exposure. Scand J Work
Environ Health, 14: 175-180.
UK (1982) The Glasgow duplicate diet study (1979/1980): A joint survey
for the Department of the Environment and the Ministry of Agriculture,
Fisheries and Food. London, Her Majesty's Stationery Office (Pollution
Report No. 11).
US Department of the Treasury (1991) Report of analyses of wines and
related products to determine lead content. Washington, DC, US
Department of the Treasury, Bureau of Alcohol, Tobacco and Firearms.
US EPA (1986a) Air quality criteria for lead. Washington, DC,
Environmental Protection Agency (EPA-600/8-83/028aF-dF).
US EPA (1986b) Test methods for evaluating solid waste SW-846 -
Physical/chemical methods. Method No. 7420 and 7421. Washington, DC,
US Environmental Protection Agency, Office of Solid Waste and
Emergency Response.
US EPA (1989) Evaluation of the potential carcinogenicity of lead and
lead compounds. Washington, DC, US Environmental Protection Agency,
Office of Health and Environmental Assessment (EPA/600/8-89/045A).
US EPA (1990) Supplement to the 1986 EPA air quality criteria for
lead. Volume 1, Addendum: Relationship of blood pressure to lead
exposure. Washington, DC, US Environmental Protection Agency, Office
of Health and Environmental Assessment (EPA/600/8-89/0 49F).
US EPA (1991) National air quality and emission trends report.
Research Triangle Park, North Carolina, US Environmental Protection
Agency (EPA-450/4-91-023).
US NCI (1979) Bioassay of lead dimethylditho-carbamate for possible
carcinogenicity. Washington, DC, National Cancer Institute,
Carcinogenesis Testing Program, Division of Cancer Cause and
Prevention (Technical Report Series No. 151 - NTIS PB-85-134663).
Vahter M & Friberg L (1988) Quality control in integrated human
exposure monitoring of lead and cadmium. Fresenius Z Anal Chem,
332: 726-731.
Vahter M & Slorach S (1990) GEMS - Global Environment Monitoring
System. Exposure monitoring of lead and cadmium: an international
pilot study within the WHO/UNEP Human Exposure Assessment Location
(HEAL) Programme. Nairobi, United Nations Environment Programme,
82 pp.
Vahter M, Berglund M, Friberg L, Jorhem L, Lind B, Slorach S, &
Åkesson A (1990) Dietary intake of lead and cadmium in Sweden. Var
Föda, 42(2): 1-16.
Vahter M, Berglund M, Slorach S, Friberg L, Saric M, Xingquan Z, &
Fujita M (1991a) Methods for integrated exposure monitoring of lead
and cadmium. Environ Res, 56: 78-79.
Vahter M, Berglund M, Lind B, Jorhem L, Slorach S, & Friberg L (1991b)
Personal monitoring of lead and cadmium exposure - a Swedish study
with special reference to methodological aspects. Scand J Work Environ
Health, 17: 65-74.
Van Esch GJ & Kroes R (1969) The induction of renal tumors by feeding
basic lead acetate to mice and hamsters. Br J Cancer, 23: 765-771.
Varo P & Kovistoinen P (1983) Mineral element composition of Finnish
foods. XII. General discussion and nutritional evaluation. Acta Agric
Scand, 22: 165.
Vatsala S & Ramakrishna T (1985) "Tinning" of brass utensils:
Possibility of lead poisoning. Indian J Environ Health, 27: 140-141.
Verrula GR & Noah PK (1990) Clinical manifestations of childhood lead
poisoning. J Trop Med Hyg, 93: 170-177.
Verschoor M, Wibowo A, & Herber R (1987) Influence of occupational low
level lead exposure on renal parameters. Am J Ind Med, 12: 341-351.
Victery W (1988) Evidence for effects of chronic lead exposure on
blood pressure in experimental animals: An overview. Environ Health
Perspect, 78: 71-76.
Victery W, Vander AJ, Markel H, Katzman L, Shulak JM, & Germain C
(1982a) Lead exposure, begun in-utero, decreases renin and angiotensin
II in adult rats. Proc Soc Exp Biol Med, 170: 63-67.
Victery W, Vander AJ, Shulak JM, Schoeps P, & Julius S (1982b) Lead,
hypertension and the renin-angiotensin system in rats. J Lab Clin Med,
99: 354-362.
Volkening J, Baumann H, & Heumann KG (1988) Atmospheric distribution
of particulate lead over the Atlantic Ocean from Europe to Antarctica.
Atmos Environ, 22: 1169-1174.
Wadge A & Hutton M (1987) The leachability and chemical speciation of
selected trace elements in fly ash from coal combustion and refuse
incineration. Environ Pollut, 48: 85-99.
Wai CM, Knowles CR, & Keely JF (1979) Lead caps on wine bottles and
their potential problems. Bull Environ Contam Toxicol, 21: 4-6.
Wallace DM, Kalman DA, & Bird TD (1985) Hazardous lead release from
glazed dinnerware: A cautionary note. Sci Total Environ, 44: 289-292.
Wang Y-L (1984) Industrial lead poisoning in China over the past 33
years. Ecotoxicol Environ Saf, 8: 526-530.
Wang J-D, Jang C-S, Hwang Y-H, & Chen Z-S (1992) Lead contamination
around a kindergarten near a battery recycling plant. Bull Environ
Contam Toxicol, 49: 23-30.
Ward NI, Watson R, & Bryce-Smith D (1987) Placental element levels in
relation to fetal development for obstetrically normal births. A study
of 37 elements. Evidence for the effects of cadmium, lead, and zinc on
fetal growth and for smoking as a source of cadmium. Int J Biosoc Res,
9: 63-81.
Wasserman G, Graziano JH, Factor-Litvak P, Popovac D, Morina N,
Musabegovic A, Vrenezi N, Capuni-Paracka S, Lekic V, Preteni-Redjepi
E, Hadzialijevic S, Slavkovich V, Kline J, Shrout P, & Stein Z (1992)
Independent effects of lead exposure and iron deficiency anemia on
developmental outcome at age 2 years. J Pediatr, 121(5): 695-703.
Watson WS, Hume R, & Moore MR (1980) Oral absorption of lead and iron.
Lancet, 2(8188): 236-237.
Watson WS, Morrison J, Bethel MIF, Baldwin NM, Lyon DTB, Dobson H,
Moore MR, & Hume R (1986) Food iron and lead absorption in humans. Am
J Clin Nutr, 44: 248-256.
Weast RC ed. (1985) Handbook of chemistry and physics, 66th ed. Boca
Raton, Florida, CRC Press.
Weeden RP, Mallik DK, & Batuman V (1979) Detection and treatment of
occupational lead nephropathy. Arch Intern Med, 139: 53-57.
White JM & Harvey DR (1972) Defective synthesis of alpha and ß globin
chains in lead poisoning. Nature (Lond), 236: 71-73.
WHO (1980) Recommended health-based limits in occupational exposure to
heavy metals. Report of a WHO Study Group. Geneva, World Health
Organization (Technical Report Series No. 647).
WHO (1984) Guidelines for drinking-water quality. Volume 1:
Recommendations. Geneva, World Health Organization, pp 55-56.
WHO (1985) Guidelines for the study of dietary intake of chemical
contaminants. Geneva, World Health Organization (WHO Offset
Publication No. 87).
WHO (1987) Air quality guidelines for Europe. Copenhagen, World Health
Organization, Regional Office for Europe, pp 200-209 (European Series,
No. 23).
WHO (1993) Guidelines for drinking-water quality. 2nd ed. Volume 1:
Recommendations. Geneva, World Health Organization, 188 pp.
Wiebe JP, Barr KJ, & Buckingham KD (1982) Lead administration during
pregnancy and lactation affects steriodogenesis and hormone receptors
in testes of offspring. J Toxicol Environ Health, 10: 653-666.
Wiebe JP, Salhanick AI, & Myers KI (1983) On the mechanism of action
of lead on the testes: in vitro suppression of FSH receptors, cyclic
AMP and steroidogenesis. Life Sci, 32: 1997-2005.
Wiebe RA, Anderson BS, Lehman CW, & Fu DJ (1991) Lead poisoning in
Hawaii: 1990. Hawaiian Med J, 50: 89-95.
Wielopolski L, Ellis KJ, Vaswani AN, Cohn SH, Greenberg A, Puschett
JB, Parkinson DK, Fetterolf DE, & Langrigan PJ (1986) In vivo bone
lead measurements: A rapid monitoring method for cumulative lead
exposure. Am J Ind Med, 9: 221-226.
Wigg NR, Vimpani GV, McMichael AJ, Baghurst PA, Robertson EF, &
Roberts RJ (1988) Port Pirie cohort study: childhood blood lead and
neuropsychological development at age two years. J Epidemiol Community
Health, 42: 213-219.
Wildt K, Eliasson R, & Berlin M (1983) Effects of occupational
exposure to lead on sperm and semen. In: Clarkson TW, Nordberg GF, &
Sager PR ed. Reproductive and developmental toxicity of metals. New
York, London, Plenum Press, pp 279-300.
Wilhelm M, Lombeck I, & Hafner D (1989) Hair lead levels in young
children from the FRG. J Trace Elem Electrolytes Health Dis,
3: 165-170.
Williams MK, King E, & Walford J (1969) An investigation of lead
absorption in an electric accumulator factory with the use of personal
samplers. Br J Ind Med, 26: 202-216.
Williamson AM & Teo RKC (1986) Neurobehavioural effects of
occupational lead exposure. Br J Ind Med, 43: 374-380.
Wilson D, Esterman A, Lewis M, Roder D, & Calder I (1986) Children's
blood lead levels in the lead smelting town of Port Pirie South
Australia. Arch Environ Health, 41: 245-250.
Winneke G (1979) Modification of visual evoked potentials in rats
after long-term blood lead elevation. Acta Nerv Sup (Praha),
21: 282-284.
Winneke G, Brockhaus A, & Baltissen R (1977) Neurobehavioural and
system effects of long term blood lead-elevation in rats. I.
Discrimination learning and open field-behavior. Arch Toxicol,
37: 247-263.
Winneke G, Hrdina K-G, & Brockhaus A (1982a) Neuropsychological
studies in children with elevated tooth-lead concentrations. I. A
pilot study. Int Arch Occup Environ Health, 51: 169-183.
Winneke G, Lilienthal H, & Werner W (1982b) Task dependent
neurobehavioural effects of lead in rats. Arch Toxicol, 5(Suppl):
84-93.
Winneke G, Kramer U, Brockhaus A, Ewers U, Kujanek G, Lechner H, &
Janke W (1983) Neuropsychological studies in children with elevated
tooth-lead concentrations. II. Extended study. Int Arch Occup Environ
Health, 51: 231-252.
Winneke G, Beginn U, Ewert T, Havestadt C, Kraemer U, Krause C, Thron
HL, & Wagner HM (1985) Comparing the effects of perinatal and later
childhood lead exposure on neuropsychological outcome. Environ Res,
38: 155-167.
Winneke G, Collet W, Krämer U, Brockhaus A, Ewert T, & Krause C (1989)
Follow-up studies in lead-exposed children. In: Smith MA, Grant LD, &
Sors AI ed. Lead exposure and child development: An international
assessment. Dordrecht, London, Kluwer Academic Publishers, pp 260-270.
Winneke G, Brockhaus A, Ewers U, Kramer U, & Neuf M (1990) Results
from the European multicenter study on lead neurotoxicity in children:
implications for risk assessment. Neurotoxicol Teratol, 12: 553-559.
Wong PTS, Chau YK, & Luxon PL (1975) Methylation of lead in the
environment. Nature (Lond), 253: 263-264.
Wooler K & Melamed S (1978) Simple tests for monitoring excessive lead
absorption. Med J Aust, 1: 163-169.
World Bureau of Metal Statistics (1992) World metal statistics, July,
45(7): 75-78.
Worley PF, Barabon JM, Desouza EB, & Snyder SH (1986) Mapping second
messenger systems in the brain: differential localizations of
adenylate cyclase and protein kinase. C. Proc Natl Acad Sci (USA),
83: 4053-4057.
Worth D, Matranga A, Lieberman M, Devos E, Karelekas P, Ryan C, &
Kraun G (1981) Lead in drinking water: The contribution of household
tap water to blood lead levels. In: Lynam DR, Piantanida LG, & Cole JF
ed. Environmental lead. Proceedings of the Second International
Symposium on Environmental Lead Research. New York, London, San
Francisco, Academic Press, pp 199-225.
Yankel AJ, von Lindern IH, & Walter SD (1977) The Silver Valley lead
study: The relationship between childhood blood lead levels and
environmental exposure. J Air Pollut Control Assoc, 27: 763-767.
Yip R & Dallman PR (1984) Developmental changes in erythrocyte
protoporphyrin: Roles of iron deficiency and lead toxicity. J Pediatr,
710-713.
Yule W, Lansdown R, Millar IB, & Urbanowicz M-A (1981) The
relationship between blood lead concentrations, intelligence and
attainment in a school population: a pilot study. Dev Med Child
Neurol, 23: 567-576.
Zelikoff JT, Li JH, Hartwig A, Wang XW, Costa M, & Rossman TG (1988)
Genetic toxicology of lead compounds. Carcinogenesis, 9: 1727-1732.
Zerez CR, Wong MD, & Tanaka KR (1990) Partial purification and
properties of nicotinamide adenine dinucleotide synthetase from human
erythrocytes: evidence that enzyme activity is a sensitive indicator
of lead exposure. Blood, 75: 1576-1582.
Zhu BG (1984) Study of chronic lead-poisoning from using tea kettles.
Chung-Hua Yu Fang I Hsueh Tsa Chih, 18: 328-330.
Ziegler EE, Edwards BB, Jensen RL, Mahaffey KR, & Fomon SJ (1978)
Absorption and retention of lead by infants. Pediatr Res, 12: 29-34.
Zuckerman MA, Savory D, & Rayman G (1989) Lead encephalopathy from an
imported Toby mug. Postgrad Med J, 65: 307-309.
RESUME
La présente monographie est consacrée aux risques pour la santé
humaine découlant de l'exposition au plomb et aux dérivés minéraux du
plomb. On y insiste sur les données dont on a eu connaissance depuis
la publication du No 3 de la Série des Critères de l'environnement:
Plomb (OMS, 1977). Quant aux effets du plomb au niveau de
l'environnement, ils sont étudiés dans le No 85 de cette série: Lead -
Environmental Aspects (Le plomb dans l'environnement) (WHO, 1989, en
anglais seulement).
1. Identité, propriétés physiques et chimiques et méthodes d'analyse
Le plomb est un métal mou, gris argent, qui fond à 327,5°C. Il
est très résistant à la corrosion mais se dissout à chaud dans l'acide
nitrique et l'acide sulfurique. La valence habituelle du plomb dans
ses dérivés minéraux est de +2. La solubilité dans l'eau de ses
composés est variable, le sulfure et les oxydes étant peu solubles et
le nitrate, le chlorate et le chlorure assez solubles à chaud. Le
plomb forme également des sels avec des acides organiques tels que
l'acide lactique et l'acide acétique ainsi que des composés organiques
stables tels que le plomb-tétraéthyle et le plomb-tétraméthyle.
Les méthodes d'analyse les plus couramment utilisées pour doser
le plomb à faible concentration dans les produits biologiques et dans
l'environnement sont la spectrométrie d'absorption avec atomisation en
flamme, en four à cellules de graphite et en plasma induit par haute
fréquence (ICP/PIHF), ainsi que la coulométrie avec redissolution
anodique. Selon le traitement préalable subit par l'échantillon et les
techniques d'extraction et l'instrumentation utilisées, la limite de
détection peut être de 0,12 µmoles de plomb par litre de sang
(2,49 µg/dl). Toutefois, on n'obtient de résultats fiables qu'en se
conformant à une marche à suivre bien codifiée pour réduire au minimum
le risque de contamination au cours du prélèvement des échantillons,
de leur conservation, de leur traitement et de leur analyse.
2. Sources d'exposition humaine
La proportion du plomb dans la croûte terrestre est d'environ
20 mg/kg. Le plomb présent dans l'environnement peut être d'origine
naturelle ou artificielle. Le plomb naturellement présent dans
l'atmosphère provient, entre autres, de l'altération des roches et des
éruptions volcaniques, et on l'estime à 19 000 tonnes/an, contre
126 000 tonnes/an provenant des mines et des fonderies, la
consommation annuelle totale étant supérieure à 3 millions de tonnes.
On a trouvé des concentrations de plomb atmosphérique de
50 pg/m3 dans des zones reculées. La concentration de fond du plomb
dans le sol varie de 10 à 70 mg/kg et on a relevé à proximité de
routes une teneur moyenne de 138 mg/kg. Actuellement la teneur de
l'eau en plomb dépasse rarement quelques microgrammes/litre; la
concentration naturelle du plomb dans les eaux de surface est estimée
à 0,02 µg/litre.
Le plomb et ses dérivés peuvent pénétrer dans l'environnement par
une porte d'entrée quelconque lors de l'extraction minière, des
opérations de fonte et du traitement, de l'utilisation, du recyclage
ou du rejet du plomb. Le plomb est principalement utilisé dans les
batteries, les câbles, les pigments, les additifs pour essence, la
brasure et les aciers. Le plomb et ses dérivés entrent également dans
la composition de la brasure que l'on utilise pour souder les
conduites d'eau et les boites de conserve alimentaire; il sert
également à la confection de certains remèdes traditionnels, des
capsules de bouteilles contenant des boissons alcoolisées, des vernis
pour céramique et de la verrerie de cristal. Dans les pays où l'on
utilise encore de l'essence au plomb, les principales émissions dans
l'atmosphère proviennent de sources de combustion de produits
pétroliers, sources qui peuvent être mobiles ou fixes (en
agglomération). Les émissions dans l'atmosphère sont également très
importantes à proximité des mines et des fonderies de plomb.
Le plomb en suspension dans l'air peut se déposer sur le sol et
dans l'eau et il parvient ainsi jusqu'à l'homme par l'intermédiaire de
la chaîne alimentaire et de l'eau de boisson. Le plomb atmosphérique
contribue également pour une part importante au plomb présent dans la
poussière domestique.
3. Transport, distribution et transformation dans l'environnement
Le transport et la distribution du plomb à partir de sources
fixes, mobiles ou naturelles, s'effectue principalement par
l'intermédiaire de l'air. La majeure partie du plomb émis dans l'air
se redépose à proximité de la source d'émission, même si quelques
particules (de diamètre < 2 µm) dont transportées sur de longues
distances et provoquent la contamination de sites aussi reculés que
les glaciers arctiques. Le plomb aéroporté peut contribuer à
l'exposition humaine par contamination des aliments, d l'eau et de la
poussière, ainsi que par inhalation directe. L'élimination du plomb
aéroporté dépend des conditions atmosphériques et de la taille des
particules en suspension. De grandes quantités de plomb peuvent être
déversées dans le sol et l'eau. Cependant, ces produits ont tendance à
rester sur place du fait de la médiocre solubilité dans l'eau des
dérivés du plomb.
Le plomb qui s'est déposé dans l'eau, que ce soit à partir de
l'air ou par lessivage des sols, se répartit rapidement entre les
sédiments et la phase aqueuse, selon la valeur du pH, la teneur en
sels et la présence éventuelle d'agents chélateurs organiques. Lorsque
le pH dépasse 5,4, l'eau dure peu contenir environ 30 µg de
plomb/litre et l'eau douce environ 500 µg/litre. Une très faible
quantité du plomb déposé sur le sol passe dans les eaux de surface ou
les eaux souterraines sauf en cas d'érosion ou d'altération des
roches; il est normalement fortement lié par chélation aux matières
organiques.
Le plomb en suspension dans l'air peut passer dans les êtres
vivants soit directement, soit par fixation à partir du sol. Les
animaux peuvent être exposés au plomb de manière directe par broutage
de l'herbe, ingestion de terre ou par inhalation. Le plomb minéral ne
subit qu'une faible bioamplification le long de la chaîne alimentaire.
4. Concentrations dans l'environnement et exposition humaine
Dans la population générale adulte des non-fumeurs, la principale
voie d'exposition est la consommation de nourriture et d'eau. Le plomb
est suspension dans l'air peut jouer un rôle important dans
l'exposition, en fonction de facteurs tels que le tabagisme, la
profession, la proximité d'une autoroute, d'une fonderie, etc. ou de
certains lieux de loisirs (par exemple, ateliers d'artisanat, stands
de tir, etc.). Pour les nourrissons et les enfants en bas âge, la
nourriture, l'air, l'eau et la poussière ou la terre sont les
principales voies d'exposition potentielles. Pour les nourrissons
jusqu'à quatre ou cinq mois, les principales sources d'exposition au
plomb sont l'air, le lait et les laits maternisés ainsi que l'eau.
Les concentrations de plomb observées dans l'air, les aliments,
l'eau et le sol ou la poussière varient largement d'une région à
l'autre du monde et dépendent du degré d'industrialisation,
d'urbanisation ainsi que du mode de vie. Dans l'air ambiant, des
concentrations supérieures à 10 µg/m3 ont été signalées en milieu
urbain à proximité d'une fonderie, alors que dans les villes où l'on
n'utilise plus d'essence au plomb, ces concentrations peuvent tomber
en-dessous de 0,2 µg/m3. Dans ces conditions, la dose de plomb
absorbée à partir de l'air peut varier de moins de 4 µg/jour à plus de
200 µg/jour.
Dans des échantillons d'eau potable prélevés à la source, on
trouve généralement des concentrations de plomb inférieures à
5 µg/litre. Cependant, lorsque l'eau est prélevée au robinet dans les
maisons dont les canalisations sont en plomb, les teneurs peuvent
dépasser 100 µg/litre, en particulier lorsque l'eau a séjourné dans la
tuyauterie pendant plusieurs heures.
L'exposition au plomb par la voie alimentaire dépend de nombreux
facteurs tenant au mode de vie et notamment à la nature des aliments
consommés, au mode de préparation, au fait que l'on utilise ou non de
la brasure au plomb, à la teneur de l'eau en plomb et à l'utilisation
de vaisselle recouverte d'un vernis au plomb.
Les nourrissons et les enfants peuvent souvent être très exposés
au plomb présent dans la poussière et dans la terre. La teneur de la
poussière en plomb dépend de facteurs tels que la vétusté et l'état de
la maison, l'utilisation de peintures à base de plomb, la présence de
plomb dans l'essence et la densité urbaine. La dose de plomb absorbée
dépendra également de l'âge et du comportement de l'enfant ainsi que
de la biodisponibilité du plomb dans le produit où il est présent.
Chez les ouvriers des industries où l'on produit, raffine,
utilise ou rejette du plomb ou des dérivés du plomb, la principale
voie d'exposition est l'inhalation. Au cours d'un poste de travail de
8 heures, les ouvriers peuvent en absorber des quantités atteignant
400 µg, qui s'ajoutent aux 20 ou 30 µg absorbés quotidiennement à
partir de la nourriture, de l'eau et de l'air ambiant; l'ingestion de
grosses particules peut également contribuer de façon importante à cet
apport.
5. Cinétique et métabolisme chez les animaux de laboratoire et
l'homme
L'homme et les animaux résorbent le plomb qu'ils ingèrent ou
inhalent; la résorption percutanée est minime chez l'homme. Selon sa
granulométrie, sa forme chimique et sa solubilité dans les liquides
biologiques, un dérivé du plomb peut être résorbé jusqu'à hauteur de
50% après avoir été inhalé. Certaines grosses particules (plus de
7 µm) sont avalées après avoir été rejetées des voies respiratoires
par l'action de l'ascenseur muco-ciliaire. Chez l'homme et les animaux
d'expérience, l'absorption du plomb dans les voies digestives dépend
de la nature physico-chimique du produit ingéré, de l'état
nutritionnel du sujet ainsi que du type d'aliments consommés. Chez
l'homme adulte, le plomb contenu dans les aliments est absorbé à peu
près à hauteur de 10%; la proportion est plus élevée lorsque le sujet
est à la diète. Toutefois, chez les nourrissons et les enfants en bas
âge, le plomb d'origine alimentaire peut être absorbé à hauteur de
50%, encore que le taux d'absorption du plomb provenant des poussières
ou de la terre ainsi que des écailles de peinture puisse être plus
faible, selon sa biodisponibilité. Les régimes alimentaires pauvres en
calcium, phosphates, sélénium ou zinc peuvent accroître l'absorption
du plomb. Le fer et la vitamine D influent également sur l'absorption
du plomb.
Le taux de plomb dans le sang ou plombémie est utilisé pour
évaluer la charge en plomb de l'organisme ainsi que les doses de plomb
(internes) absorbées. La relation entre la plombémie et la
concentration du plomb dans les diverses sources d'exposition n'est
pas linéaire (corrélation curviligne).
Une fois absorbé, le plomb ne se répartit pas de manière homogène
dans l'organisme. Après être rapidement passé dans le sang et les
tissus mous, il se redistribue lentement des les os. La plomb
s'accumule dans les os pendant une longue période de la vie humaine et
peut servir de source endogène de plomb. La demi-vie du plomb dans le
sang et les tissus mous est d'environ 28 à 36 jours mais elle peut
être beaucoup plus longue dans les diverses parties de l'os. Le taux
de rétention du plomb dans l'organisme est plus élevé chez l'enfant
que chez l'adulte. Pendant toute la durée de la gestation, le plomb
passe facilement de la mère au foetus.
La plombémie est la mesure la plus couramment utilisée pour
évaluer l'exposition au plomb. Toutefois, on dispose aujourd'hui de
techniques permettant de doser le plomb dans les dents et les os,
encore que la cinétique du phénomène ne soit pas parfaitement
élucidée.
6. Effets sur les animaux de laboratoire et les systèmes d'épreuves
in vitro
Chez toutes les espèces d'animaux de laboratoire étudiées, y
compris des primates non-humains, on a constaté que le plomb
produisait des effets indésirables au niveau de plusieurs organes et
systèmes, notamment le système haematopoïétique, le système nerveux,
les reins, le système cardio-vasculaire, l'appareil reproducteur et le
système immunitaire. Le plomb est également nocif pour les os et on a
montré qu'il avait des effets cancérogènes sur le rat et la souris.
Malgré les différences d'ordre cinétique qui existent entre les
animaux d'expérience et l'homme, ces études apportent des arguments
biologiques de poids à la plausibilité de tels effets chez l'homme.
Chez le rat, on a observé une diminution de la capacité
d'apprentissage et de mémorisation lorsque la plombémie était de
l'ordre de 0,72 à 0,96 µmol/litre (soit 15 à 20 µg/dl), les mêmes
effets étant observés chez des primates non-humains pour une plombémie
ne dépassant pas 0,72 µmol/litre (15 µg/dl). En outre,
l'expérimentation animale a également permis d'observer des troubles
de la vision et de l'audition.
Chez le rat, une plombémie supérieure à 2,88 µmol/litre
(60 µg/dl), soit une valeur analogue à celle qui, selon les
observations, constitue le seuil d'apparition des effets chez l'homme,
est à même de provoquer l'apparition d'effets néphrotoxiques. Des
effets cardio-vasculaires ont été également observés chez des rats
après exposition chronique à de faibles doses de plomb entraînant une
plombémie de l'ordre de 0,24 à 1,92 µmol/litre (5 à 40 µg/dl). A des
doses inférieures à la dose maximale tolérée qui est de 200 mg de
plomb (sous forme d'acétate) par litre d'eau potable, on a observé
l'apparition de tumeurs. Cette dose constitue la dose maximale qui
n'entraîne pas d'autres effets morphologiques ou fonctionnels.
7. Effets sur l'homme
Chez l'homme, le plomb peut produire des effets biologiques très
divers selon l'intensité et la durée de l'exposition. On a ainsi
observé des effets au niveau infracellulaire ainsi que des effets
s'exerçant sur les fonctions générales de l'organisme, effets qui vont
de l'inhibition de certaines enzymes jusqu'à l'apparition
d'altérations morphologiques marquées et à la mort. Ces altérations se
produisent dans de larges limites de doses, l'organisme humain en
développement étant généralement plus sensible que l'organisme adulte.
On a montré que le plomb avait des effets sur nombre de processus
biochimiques; en particulier on a largement étudié les effets qu'il
produit sur la synthèse hémique, tant chez l'adulte que chez l'enfant.
Lorsque la plombémie est élevée, on observe une augmentation du taux
de protoporphyrine érythrocytaire sérique ainsi qu'une augmentation de
l'excrétion urinaire de coproporphyrine et d'acide delta-aminolévulinique.
A des valeurs plus faibles de la plombémie, on observe l'inhibition
d'enzymes comme la delta-aminolévulinique acide-déshydratase et la
dihydrobioptérine-réductase.
Les effets du plomb sur le système haématopoïétique se traduisent
par une diminution de la synthèse de l'hémoglobine et l'on a observé
une anémie chez des enfants lorsque la plombémie dépassait
1,92 µmol/litre (40 µg/dl).
Pour des raisons d'ordre neurologique, métabolique et
compartementale, les enfants sont plus sensibles aux effets du plomb
que les adultes. Des études épidémiologiques tant prospectives que
transversales ont été effectuées pour déterminer dans quelle mesure
une exposition au plomb présent dans l'environnement affecte les
fonctions psychologiques dépendant du système nerveux central. On a
ainsi montré qu'il existait une association entre l'exposition au
plomb et les troubles des fonctions neurocomportementales chez
l'enfant.
On a également constaté des anomalies des fonctions
psychologiques et neurocomportementales chez des ouvriers longtemps
exposés au plomb. Il a été montré que les paramètres
électrophysiologiques étaient d'utiles indicateurs des effets
infracliniques du plomb sur le système nerveux central.
On sait depuis longtemps que la neuropathie périphérique est due
à une exposition prolongée à de fortes concentrations de plomb sur le
lieu de travail. A plus faibles concentrations, on a observé une
diminution de la vitesse de conduction nerveuse. Ces effets se
révèlent souvent réversibles, en fonction de l'âge et de la durée de
l'exposition, après cessation de l'exposition.
Les effets que le plomb exerce sur le coeur sont indirects et
s'opèrent par l'intermédiaire du système neuro-végétatif; il n'y a pas
d'effets directs sur le myocarde. L'ensemble des faits tirés des
études sur des populations d'adultes indiquent qu'il existe une très
faible association entre la plombémie et la tension artérielle
systolique ou diastolique. Etant donné la difficulté qu'il y a à tenir
compte des facteurs de confusion, il n'a pas été possible d'établir, à
partir des résultats de ces études, une relation de cause à effet.
Rien n'indique non plus que l'association qui pourrait exister entre
la plombémie et la tension artérielle constitue un problème médical
majeur.
On sait que le plomb peut entraîner des lésions tubulaires
proximales qui se caractérisent par une aminoacidurie généralisée, une
hypophosphatémie avec d'une hyperphosphaturie relative et une
glycosurie accompagnée d'inclusions nucléiques, d'altérations des
mitochondries et d'une hypertrophie des cellules de l'épithélium
tubulaire proximal. Ces effets sur les tubules s'observent après une
exposition relativement brève et sont généralement réversibles. En
revanche, les altérations scléreuses et les fibroses interstitielles
qu'on observe lors d'une exposition chronique à de fortes
concentrations de plomb, entraînent des problèmes fonctionnels qui
peuvent déboucher sur une insuffisance rénale. On a observé une
augmentation du risque de néphropathie chez les travailleurs dont la
plombémie dépassait 3,0 µmol/litre (soit environ 60 µg/dl). En
utilisant des indicateurs fonctionnels plus sensibles, on a récemment
découvert l'existence d'effets rénaux dans la population générale.
Les effets du plomb sur la fonction de reproduction, ne
concernent, chez l'homme, que la morphologie et le nombre des
spermatozoïdes. Chez la femme, on a attribué au plomb un certain
nombre de grossesses à issue défavorable.
Il ne semble pas que le plomb ait des effets nocifs sur la peau,
les muscles ou le système immunitaire. Si l'on excepte le cas du rat,
le plomb ne paraît pas non plus être à l'origine de l'apparition de
tumeurs.
8. Evaluation des risques pour la santé humaine
Le plomb a des effets nocifs sur plusieurs organes et systèmes,
les effets les plus sensibles étant relevés au niveau infracellulaire
ainsi que sur le développement du système nerveux. On a fait état
d'une association entre la plombémie et l'hypertension. En outre, le
plomb entraîner une cascade d'effets sur les réserves hémiques de
l'organisme et il perturbe également la synthèse hémique. Cependant,
certains de ces effets ne sont pas véritablement considérés comme
délétères. Il y a également perturbation de l'homéostase du calcium
avec des contre-coups sur d'autres processus cellulaires.
a) Les preuves les plus substantielles sont on dispose à propos de
l'action nocive du plomb proviennent d'études transversales et
prospectives sur des populations dont la plombémie est
généralement inférieure à 1,2 µmol/litre (25 µg/dl) et concernent
une diminution du quotient d'intelligence (QI). Il importe
cependant de noter que ces observations ne constituent pas une
preuve définitive d'une relation de cause à effet entre ce
phénomène et l'exposition au plomb. Cependant la mesure de ce QI
à partir de l'âge de 4 ans fait ressortir un déficit qui se situe
entre 0 et 5 points (sur une échelle avec un écart-type de 15)
pour une augmentation de la plombémie de 0,48 µmol/litre
(10 µg/dl), l'ampleur de l'effet étant vraisemblablement de 1 à 3
points. Lorsque la plombémie dépasse 1,2 µmol/litre (25 µg/dl),
on peut avoir une relation différente entre cette plombémie et le
QI. Les estimations de l'ampleur de l'effet observé sont des
moyennes calculées sur des groupes et ne représentent qu'une
probabilité pour un individu en particulier.
Les études épidémiologiques dont on dispose ne donnent pas
de preuves définitives de l'existence d'un seuil. Lorsque la
plombémie se situe en-dessous de l'intervalle
0,48-0,72 µmol/litre (10-15 µg/dl), l'incertitude qui entache
toute estimation de l'effet s'accroît, du fait des facteurs de
confusion et des limites dans la précision des dosages et des
tests psychométriques. Il n'en reste pas moins qu'en-dessous de
cet intervalle de valeurs, certains éléments incitent à penser à
l'existence d'une association.
b) L'expérimentation animale milite en faveur de l'existence d'une
relation de cause à effet entre l'exposition au plomb et certains
effets neurologiques, puisqu'elle fait ressortir des déficits
dans les fonctions cognitives pour une plombémie ne dépassant pas
0,53 à 0,72 µmol/litre (11-15 µg/dl), déficits qui peuvent
persister bien après la cessation de l'exposition au plomb.
c) Pour une plombémie ne dépassant pas 1,44 µmol/litre (30 µg/dl),
il peut y avoir réduction de la vitesse de conduction nerveuse
périphérique chez l'homme. En outre, pour des valeurs de la
plombémie de dépassant pas 1,92 µmol/litre (40 µg/dl), il peut
également y avoir perturbation des fonctions moto-sensorielles et
la fonction du système nerveux neurovégétatif (variabilité de
l'intervalle R-R sur l'électrocardiogramme) peut être affectée
pour une valeur moyenne de la plombémie d'environ 1,68 µmol/litre
(35 µg/dl). Chez les travailleurs dont la plombémie dépasse
2,88 µmol/litre (60 µg/dl), il y a accroissement du risque de
néphropathie saturnienne. Toutefois, des études récentes basées
sur des indicateurs plus sensibles de la fonction rénale incitent
à penser que des effets peuvent se produire à des valeurs plus
faibles de l'exposition au plomb.
d) Il semblerait que l'exposition au plomb soit associée à une
légère augmentation de la tension artérielle. L'ordre de grandeur
probable de cette augmentation est le suivant: pour un doublement
de la plombémie (par exemple lorsqu'elle passe de 0,8 à
1,6 µmol/litre, soit de 16,6 à 33,3 µg/dl), il y a une
augmentation moyenne de 1 mmHg de la systolique. L'association
avec la diastolique est analogue mais d'ampleur plus faible.
Toutefois, on se demande si ces associations statistiques
résultent réellement de l'exposition au plomb ou s'il s'agit d'un
artefact imputable à des facteurs de confusion.
e) Certaines études épidémiologiques - mais pas toutes - font état
d'une association liée à la dose entre les accouchements avant
terme et certains indices de la croissance et de la maturation
foetales à des valeurs de la plombémie supérieures ou égales à
0.72 µmol/litre (15 µg/dl).
f) Les données relatives à la cancérogénicité pour l'homme du plomb
et de plusieurs de ses dérivés minéraux, sont insuffisantes.
g) On a mis en évidence des effets que le plomb exerce sur un
certain nombre de systèmes enzymatiques et de paramètres
biochimiques. Les valeurs de la plombémie au-dessus desquelles on
peut mettre en évidence des effets avec les techniques actuelles,
pour ce qui est des paramètres susceptibles d'avoir une
importance clinique, sont toutes supérieures à 0,96 µmol/litre
(20 µg/dl). Certains effets sur les enzymes peuvent être mis en
évidence à des valeurs plus faibles de la plombémie, mais leur
signification clinique demeure incertaine.
RESUMEN
La presente monografía se centra en los riesgos para la salud
humana asociados a la exposición al plomo y a los compuestos
inorgánicos de plomo. Se han destacado los datos disponibles desde la
publicación de Criterios de Salud Ambiental No. 3: Plomo (OMS, 1977).
Los efectos ambientales del plomo se examinan en Environmental Health
Criteria 85: Lead - Environmental Aspects (OMS, 1989).
1. Identidad, propiedades físicas y químicas y métodos analíticos
El plomo es un metal blando, gris plateado, que se funde a
327,5°C. Es muy resistente a la corrosión, pero es soluble en ácido
nítrico y en ácido sulfúrico caliente. Su valencia corriente en los
compuestos inorgánicos es +2. Su solubilidad en agua varía; el sulfito
de plomo y los óxidos de plomo son poco solubles, mientras que las
sales de nitrato, clorato y cloruro son razonablemente solubles en
agua fría. El plomo también forma sales con ácidos orgánicos tales
como el láctico y el acético, y compuestos orgánicos estables tales
como el tetraetilo de plomo y el tetrametilo de plomo.
Los métodos utilizados más corrientemente para el análisis de
bajas concentraciones de plomo en materias biológicas y ambientales
son la llama, el horno de grafito y la espectroscopia de absorción
atómica de plasma acoplado inductivamente y la voltimetría de
separación anódica. Según sean el tratamiento previo de la muestra,
las técnicas de extracción y la instrumentación analítica, pueden
alcanzarse niveles de detección de 0,12 µmoles de plomo por litro de
sangre (2,49 µg/dl). Sin embargo, se obtienen resultados fiables sólo
cuando se siguen procedimientos específicos para reducir al mínimo el
riesgo de contaminación durante la recogida, el almacenamiento,
procesamiento y análisis de la muestra.
2. Fuentes de exposición humana
El nivel de plomo en la corteza terrestre es de aproximadamente
20 mg/kg. El plomo del medio ambiente puede provenir de fuentes
naturales o antropogénicas. Las fuentes naturales de plomo atmosférico
comprenden el desgaste geológico y las emisiones volcánicas y se han
estimado en 19 000 toneladas por año, frente a unas 126 000 toneladas
por año emitidas en el aire como resultado de la minería, la fundición
y el consumo de más de 3 millones de toneladas de plomo por año.
Se han encontrado concentraciones atmosféricas de plomo de
50 pg/m3 en zonas remotas. Los niveles básicos de plomo en el suelo
oscilan entre 10 y 70 mg/kg y se ha comunicado un nivel medio de
138 mg/kg en las proximidades de las carreteras. Los niveles de plomo
presentes en las aguas rara vez exceden de unos pocos microgramos por
litro; la concentración natural de plomo en las aguas superficiales se
ha estimado en 0,02 µg/litro.
El plomo y sus compuestos pueden entrar en el medio ambiente en
cualquier punto durante las actividades de minería, fundición,
elaboración, utilización, reciclado o eliminación. Se utiliza
principalmente en la fabricación de pilas, cables, pigmentos, aditivos
de la gasolina, productos para soldar y de acero. El plomo y los
compuestos de plomo también se utilizan para soldar las tuberías de
distribución de agua y las latas de conserva, en algunos remedios
tradicionales, en las tapas de las botellas de bebidas alcohólicas y
en los esmaltes cerámicos y la cristalería de mesa. En los países
donde todavía se utiliza gasolina con plomo, la principal emisión en
el aire proviene de fuentes móviles y estacionarias de combustión de
gasolina (centros urbanos). Las zonas próximas a las minas y funderías
de plomo están expuestas a la emisión de niveles elevados en el aire.
El plomo del aire puede depositarse en el suelo y el agua, desde
donde llega al ser humano por conducto de la cadena alimentaria y del
agua de bebida. El plomo atmosférico también es una fuente importante
del plomo presente en el polvo de las viviendas.
3. Transporte, distribución y transformación en el medio ambiente
El transporte y la distribución del plomo procedente de fuentes
fijas, móviles y naturales tienen lugar principalmente a través del
aire. La mayor parte de las emisiones de plomo se depositan cerca de
la fuente, aunque algunas partículas de materia (< 2 µm de diámetro)
recorren largas distancias y contaminan lugares remotos tales como los
glaciares árticos. El plomo del aire puede contribuir a la exposición
humana mediante la contaminación de los alimentos, del agua y del
polvo, así como por inhalación directa. La eliminación del plomo del
aire depende de las condiciones atmosféricas y del tamaño de las
partículas. Pueden descargarse grandes cantidades de plomo en el suelo
y en el agua. Sin embargo, ese material tiende a permanecer localizado
debido a la escasa solubilidad de los compuestos de plomo en el agua.
El plomo depositado en el agua, ya provenga del aire o de la
escorrentía del suelo, se distribuye rápidamente entre el sedimento y
la fase acuosa, según el pH, el contenido de sales y la presencia de
agentes quelantes orgánicos. Con un pH superior a 5,4, las aguas duras
pueden contener aproximadamente 30 µg de plomo por litro y las aguas
blandas aproximadamente 500 µg de plomo por litro. Muy poco plomo
depositado en el suelo se transporta a las aguas superficiales o a las
subterráneas, salvo mediante la erosión o el desgaste geoquímico;
normalmente está ligado a la materia orgánica de forma bastante
estrecha (por quelación).
El plomo del aire puede transferirse a la biota directamente o
por absorción del suelo. Los animales pueden encontrarse expuestos al
plomo directamente mediante la ingestión de hierba y de tierra o por
inhalación. Hay poca biomagnificación del plomo inorgánico a través de
la cadena alimentaria.
4. Niveles ambientales y exposición humana
En la población general que no fuma, la principal vía de
exposición son los alimentos y el agua. El plomo del aire puede
contribuir apreciablemente a la exposición, lo que depende de factores
tales como el consumo de tabaco, la ocupación, la proximidad de
caminos transitados por vehículos automotores, de funderías de plomo,
etc., y ciertas actividades de esparcimiento (por ejemplo, artesanía,
tiro con armas de fuego). Los alimentos, el aire, el agua y el
polvo/suelo son las principales vías potenciales de exposición de los
niños pequeños. Para los niños de hasta 4 ó 5 meses de edad, el aire,
la leche, las preparaciones para lactantes y el agua son fuentes
notables de exposición al plomo.
Los niveles de plomo presentes en el aire, los alimentos, el
agua, y el suelo/polvo varían ampliamente en el mundo y dependen del
grado de desarrollo industrial y de urbanización y de factores
relacionados con el modo de vida. Se han comunicado niveles superiores
a 10 µg/m3 presentes en el aire ambiental en zonas urbanas próximas
a funderías, mientras que en ciudades donde ha dejado de usarse la
gasolina con plomo se han detectado niveles inferiores a 0,2 µg/m3.
La absorción de plomo del aire puede, pues, variar de menos de
4 µg/día a más de 200 µg/día.
Los niveles de plomo en muestras de agua de bebida extraídas de
los manantiales suelen ser inferiores a 5 µg/litro. Sin embargo, el
agua del grifo de viviendas cuyas tuberías tienen plomo contiene
niveles que exceden de 100 µg/litro, en particular después de haber
reposado el agua en las tuberías durante algunas horas.
El nivel de exposición al plomo a través de la dieta depende de
muchos factores relacionados con el modo de vida, entre ellos los
alimentos que se consumen, la tecnología de elaboración, el empleo de
soldadura de plomo, los niveles de plomo en el agua y la utilización
de cerámica con barniz de plomo.
Para los niños, el plomo presente en el polvo y en el suelo suele
ser la principal vía de exposición. Los niveles de plomo en el polvo
dependen de factores tales como la antigüedad y el estado de la
vivienda, la utilización de pinturas a base de plomo, el plomo de la
gasolina y la densidad urbana. La absorción de plomo dependerá de la
edad y de las características comportamentales del niño así como de la
biodisponibilidad de plomo en la fuente material.
La inhalación es la vía principal de exposición al plomo para los
trabajadores de industrias que producen, refinan, utilizan o desechan
plomo y compuestos de plomo. Durante un turno de ocho horas, los
trabajadores pueden absorber nada menos que 400 µg de plomo, además de
los 20-30 µg/día que absorben de los alimentos, del agua y del aire
ambiental; puede haber una absorción notable como resultado de la
inhalación de partículas grandes.
5. Cinética y metabolismo en animales de laboratorio y en el ser
humano
Los seres humanos y los animales absorben plomo por inhalación o
por ingestión; la absorción percutánea es mínima en el ser humano.
Según la especiación química, el tamaño de las partículas y la
solubilidad de los líquidos corporales, puede absorberse hasta un 50%
de los compuestos de plomo inhalados. Algunas partículas de materia
inhaladas (de más de 7 µm) se degluten después de la eliminación
mucociliar del aparato respiratorio. En los animales experimentales y
en el ser humano, la absorción de plomo del aparato gastrointestinal
está influenciada por la naturaleza fisicoquímica del material
ingerido, el estado nutricional y el tipo de alimentación. En los
seres humanos adultos, se absorbe aproximadamente el 10% del plomo
contenido en la alimentación; la proporción es más elevada en
condiciones de ayuno. Sin embargo, los lactantes y los niños pequeños
absorben nada menos que el 50% del plomo presente en la alimentación,
pero la absorción de plomo del polvo/suelo y de desconchones de
pintura puede ser menor y depende de su biodisponibilidad. Las dietas
pobres en calcio, fosfato, selenio o zinc pueden dar lugar a una mayor
absorción de plomo. El hierro y la vitamina D también influyen en la
absorción de plomo.
Los niveles de plomo en la sangre (Pb-H) se utilizan para medir
la carga corporal y las dosis absorbidas (internas) de plomo. La
relación entre el plomo presente en la sangre y la concentración de
plomo en las fuentes de exposición es curvilínea.
Una vez absorbido, el plomo no se distribuye de manera homogénea
en todo el cuerpo. Hay una absorción rápida en la sangre y en los
tejidos blandos, seguida de una redistribución más lenta a los huesos.
Los huesos acumulan plomo durante gran parte de la vida humana y
pueden actuar como fuente endógena de plomo. La semivida del plomo en
la sangre y en otros tejidos blandos es de aproximadamente 28-36 días,
pero es mucho más larga en los diversos compartimentos óseos. La
retención porcentual de plomo en los depósitos corporales es más
elevada en los niños que en los adultos. La transferencia de plomo al
feto humano se efectúa fácilmente durante la gestación.
El nivel de plomo en la sangre es la medida más utilizada para
determinar la exposición al plomo. Sin embargo, ya se dispone de
técnicas para determinar la cantidad de plomo presente en los dientes
y en los huesos, aunque aún no se conoce del todo su cinética.
6. Efectos en los animales de laboratorio y en los sistemas in vitro
En todas las especies de animales de experimentación estudiadas,
inclusive en primates no humanos, se ha observado que el plomo tiene
efectos adversos en varios órganos y sistemas de órganos, inclusive
los sistemas hematopoyético, nervioso, renal, cardiovascular,
reproductivo e inmunitario. El plomo también afecta a los huesos y se
ha demostrado que es carcinógeno en ratas y ratones.
Pese a diferencias cinéticas entre las especies de animales
experimentales y la humana, dichos estudios apoyan firmemente y
justifican desde un punto de vista biológico los hallazgos realizados
en seres humanos. Se han comunicado deficiencias del aprendizaje y de
la memoria en ratas con niveles de Pb-H de 0,72-0,96 µmoles/litro
(15-20 µg/dl) y en primates no humanos con niveles de Pb-H de no más
de 0,72 µmoles/litro (15 µg/dl). Además, se han comunicado
deficiencias visuales y auditivas en estudios realizados en animales
de experimentación.
La toxicidad renal en las ratas parece presentarse a partir de un
nivel de Pb-H de 2,88 µmoles/litro (60 µg/dl); este valor es semejante
al que, según se ha comunicado, comienza a tener efectos renales en el
ser humano. Se han observado efectos cardio-vasculares en ratas
después de exposiciones crónicas a niveles bajos que dan lugar a
niveles de Pb-H de 0,24-1,92 µmoles/litro (5-40 µg/dl). Se ha
demostrado que aparecen tumores con dosis inferiores a la dosis máxima
tolerada, de 200 mg de plomo (como acetato de plomo) por litro de agua
de bebida. Esta es la dosis máxima no asociada a otros cambios
morfológicos o funcionales.
7. Efectos en el ser humano
En el ser humano, el plomo puede tener una amplia variedad de
efectos biológicos según el nivel y la duración de la exposición. Se
han observado efectos en el plano subcelular y efectos en el
funcionamiento general del organismo que van desde la inhibición de
las enzimas hasta la producción de acusados cambios morfológicos y la
muerte. Dichos cambios se producen a dosis muy diferentes; en general,
el ser humano que se está desarrollando es más sensible que el adulto.
Se ha mostrado que el plomo tiene efectos en muchos procesos
bioquímicos; en particular, se han estudiado mucho los efectos en la
síntesis del hemo en adultos y niños. Se observan niveles más altos de
porfirina eritrocitaria sérica y mayor excreción urinaria de
coproporfirina y de ácido delta-aminolevulínico cuando las
concentraciones de Pb-H son elevadas. Con niveles más bajos se observa
inhibición de las enzimas dehidratasa del ácido delta-aminolevulínico
y reductasa de la dihidrobiopterina.
Como resultado de los efectos del plomo en el sistema
hematopoyético disminuye la síntesis de hemoglobina y se ha observado
anemia en niños a concentraciones de Pb-H superiores a
1,92 µmoles/litro (40 µg/dl).
Por razones neurológicas, metabólicas y comportamentales, los
niños son más vulnerables a los efectos del plomo que los adultos. Se
han efectuado estudios epidemiológicos prospectivos y transversales
para evaluar la medida en que la exposición al plomo ambiental afecta
a las funciones psicológicas regidas por el sistema nervioso central
(SNC). Se ha mostrado que el plomo está asociado a deficiencias
neurocomportamentales en los niños.
Se han observado deficiencias psicológicas y
neurocomportamentales en trabajadores que habían estado expuestos al
plomo durante un tiempo prolongado. Los parámetros electrofisiológicos
han demostrado ser indicadores útiles de los efectos subclínicos del
plomo en el SNC.
Desde hace tiempo se sabe que la exposición prolongada a niveles
elevados de plomo en el medio laboral provoca neuropatías periféricas.
Con niveles más bajos se ha observado una reducción de la velocidad de
conducción nerviosa. Se ha observado a menudo que dichos efectos son
reversibles después de cesar la exposición, según la edad del sujeto y
la duración de la exposición.
Los efectos del plomo en el corazón son indirectos y se producen
por conducto del sistema nervioso autónomo; el plomo no tiene efectos
directos en el miocardio. Datos colectivos procedentes de estudios de
poblaciones adultas indican asociaciones muy débiles entre la
concentración de Pb-H y la presión arterial sistólica o diastólica.
Dada la dificultad de evaluar el influjo de los factores de confusión
pertinentes, no puede establecerse una relación causal sobre la base
de esos estudios. No hay indicios de que la relación entre la
concentración de Pb-H y la presión arterial tenga mucha importancia
para la salud.
Se sabe que el plomo provoca en los tubos proximales del riñón
lesiones que se caracterizan por aminoaciduria generalizada,
hipofosfatemia con hiperfosfaturia relativa y glucosuria acompañada de
cuerpos de inclusión nuclear, modificaciones mitocondriales y
citomegalia de las células epiteliales de los tubos proximales. Los
efectos tubulares se manifiestan después de una exposición
relativamente breve y suelen ser reversibles, mientras que los cambios
escleróticos y la fibrosis intersticial, que dan lugar a una
disminución de la función renal y a una posible insuficiencia renal,
requieren una exposición crónica a niveles elevados de plomo. Se ha
advertido un mayor riesgo de nefropatía en los trabajadores que tienen
niveles de Pb-H superiores a 3,0 µmoles/litro (aproximadamente
60 µg/dl). Recientemente se han observado efectos renales en la
población general tras haberse utilizado indicadores de función más
sensibles.
Los efectos del plomo en la función reproductora masculina se
limitan a la morfología y el número de los espermatozoides. En cuanto
a la femenina, se han atribuido al plomo algunos efectos adversos en
el embarazo.
El plomo no parece tener efectos nocivos en la piel, en los
músculos ni en el sistema inmunitario. Salvo en la rata, el plomo no
parece estar relacionado con el desarrollo de tumores.
8. Evaluación de los riesgos para la salud humana
El plomo tiene efectos adversos en varios órganos y sistemas de
órganos; los más delicados parecen ser los cambios subcelulares y los
efectos en el desarrollo del sistema nervioso. Se ha observado una
asociación entre el nivel de Pb-H y la hipertensión (presión
arterial). El plomo produce una serie de efectos en la reserva
corporal de hemo y afecta a la síntesis de éste. Sin embargo, algunos
de estos efectos no se consideran adversos. Está afectada la
homeostasia del calcio, lo que interfiere en otros procesos celulares.
a) Los datos más importantes de los estudios transversales y
prospectivos de poblaciones con niveles de Pb-H generalmente
inferiores a 1,2 µmoles/litro (25 µg/dl) se relacionan con una
disminución del coeficiente de inteligencia (CI). Es importante
señalar que esas observaciones no pueden constituir una prueba
concluyente de una relación causal con la exposición al plomo.
Sin embargo, la magnitud del efecto aparente en el CI,
determinado desde los 4 años en adelante, es un déficit de 0 a
5 puntos (en una escala con una desviación estándar de 15) por
cada 0,48 µmoles/litro (10 µg/dl) de aumento del nivel de Pb-H,
con una magnitud probable del efecto aparente de 1 a 3 puntos.
Con niveles de Pb-H superiores a 1,2 µmoles/litro (25 µg/dl), la
relación entre el Pb-H y el CI puede ser diferente. Las
estimaciones de la magnitud del efecto constituyen promedios
grupales y sólo se aplican a cada niño de manera probabilística.
Los estudios epidemiológicos existentes no prueban de modo
concluyente la existencia de un umbral. Por debajo de unos
niveles de Pb-H de 0,48-0,72 µmoles/litro (10-15 µg/dl), los
efectos de las variables de confusión y los límites de la
precisión de las mediciones analíticas y psicométricas aumentan
la incertidumbre inherente a toda estimación de un efecto. Sin
embargo, hay algunos indicios de una asociación por debajo de
dichos niveles.
b) Los estudios realizados en animales respaldan la idea de una
relación causal entre el plomo y ciertos efectos en el sistema
nervioso; se señalan deficiencias cognitivas a niveles de Pb-H de
sólo 0,53-0,72 µmoles/litro (11-15 µg/dl), deficiencias que
pueden persistir mucho después de haber terminado la exposición
al plomo.
c) Puede producirse una reducción de la velocidad de conducción
nerviosa periférica en el ser humano con niveles de Pb-H de sólo
1,44 µmoles/litro (30 µg/dl). Además, las funciones
sensitivomotrices pueden verse disminuidas con niveles de Pb-H de
sólo 1,92 µmoles/litro (40 µg/dl) aproximadamente y las funciones
del sistema nervioso autónomo (variabilidad del intervalo R-R
electrocardiográfico) pueden verse afectadas a un nivel promedio
de Pb-H de aproximadamente 1,68 µmoles por litro (35 µg/dl). El
riesgo de nefropatía plúmbica aumenta en los trabajadores que
tienen niveles de Pb-H superiores a 2,88 µmoles/litro (60 µg/dl).
Sin embargo, estudios recientes que utilizan indicadores más
sensibles de la función renal sugieren la aparición de efectos
renales a niveles más bajos de exposición al plomo.
d) La exposición al plomo está asociada a un pequeño aumento de la
presión arterial. El orden de magnitud probable es que por cada
duplicación del nivel de Pb-H (por ejemplo, de 0,8 a
1,6 µmoles/litro, es decir, de 16,6 a 33,3 µg/dl) hay un aumento
medio de 1 mmHg de presión arterial sistólica. La relación con la
presión diastólica es de una magnitud semejante pero más pequeña.
Sin embargo, no se sabe bien si estas asociaciones estadísticas
obedecen realmente a un efecto de la exposición al plomo o son un
resultado ficticio debido a factores de confusión.
e) Algunos estudios epidemiológicos, no todos, muestran una
relación, dependiente de la dosis, con el parto prematuro y
algunos índices de crecimiento y maduración fetales a niveles de
Pb-H de 0,72 µmoles/litro (15 µg/dl) o más.
f) Los indicios de carcinogenicidad del plomo y de varios compuestos
inorgánicos de plomo en el ser humano son insuficientes.
g) Se ha demostrado que el plomo tiene efectos en cierto número de
sistemas enzimáticos y de parámetros bioquímicos. Los niveles de
Pb-H por encima de los cuales las técnicas vigentes pueden
demostrar la presencia de efectos en relación con los parámetros
de importancia clínica posible son todos superiores a
0,96 µmoles/litro (20 µg/dl). Algunos efectos en enzimas pueden
demostrarse con niveles de Pb-H más bajos, pero su importancia
clínica es incierta.
8.3.6.4 Task group overview of cross-sectional studies
a) Methods of controlling for confounders
A particular methodological concern in the cross-sectional
studies is the manner in which they have taken account of confounding
factors in deriving adjusted estimates of associations between body
lead burden and neuropsychological performance, in particular IQ.
The three aspects of the problem are:
i) Which potential confounders are measured? There is considerable
variation between studies here, suggesting that no individual study
can have fully corrected for the full range of parental and social
influences on IQ.
ii) How were confounders selected for inclusion in the analysis? Some
studies include confounders solely on the basis of their strength of
association with outcome (IQ), while others have given attention to
their association with body lead burden. It is difficult to assess the
extent to which these differences would affect the results, but the
former is more generally accepted statistical practice.
iii) Which statistical analysis strategy was employed in reaching a
final model? Most studies have adopted some form of multiple
regression technique, but have varied as to whether forward selection
(i.e. model building), backwards selection (i.e. model collapsing) or
more arbitrary choices of final model were employed. In the larger
studies, the choice between such strategies is unlikely to matter, but
in smaller studies the greater play of random variation could make the
results dependent on the statistical technique.
b) What they told us
In most studies a negative association between lead measures and
IQ measures is found in uncontrolled data. This difference is usually
in the range of 4 to 6 IQ points. Most studies also confirmed the
positive association between lead measures and indicators of social
disadvantage, whether this is indicated by SES, maternal education or
other more detailed indicators of non-optimal child-rearing
environments, such as marital quality or maternal depression. When
these social and other confounding factors are controlled, the effect
has been, in most cases, to reduce the strength of the association
between lead measures and IQ, although it remains in the same
direction. Where maternal intelligence has not been controlled, the
impact of correction for covariates tends to be smaller, and where
more detailed social measures have been controlled, the impact has
been greater.
c) What they did not tell us
The cross-sectional nature of these studies, and the fact that
many used a single measure of current exposure, limited their
usefulness in answering questions relating to the natural history of
the association between lead exposure and outcomes, including whether
there were critical periods of exposure, and whether lead-associated
deficits were persistent or reversible. Equally they were limited in
the information that could be obtained relating to the natural history
of confounders. They were also unable to answer questions of reverse
causality.
8.3.6.5 An interpretation of the overview of prospective and
cross-sectional studies
The above meta-analyses of the prospective studies and cross-
sectional studies reveal a consistency between studies which points
towards a "collectively significant" inverse association between PbB
level and full-scale IQ. Taking the results in Fig. 15 as a guideline,
there appears to be a mean decrease in full-scale IQ of the order of 2
IQ points for a change in mean PbB level from 0.48 to 0.96 µmol/litre
(10 to 20 µg/dl).
Below this range, uncertainties are increased, concerning firstly
the existence of an association and secondly estimates of the
magnitude of any putative association. The relatively limited numbers
of children in most studies with PbB levels below this range, the
strong contributions of confounding variables, and limitations in the
precision in analytical and psychometric measurements combine to lower
statistical power to detect associations and to estimate their
magnitude.
The key question is whether this statistical association is
directly attributable to the causal influence of lead on child IQ. It
is important to consider alternative explanations as follows:
a) Chance The consistency of alternative analyses and the level of
significance achieved suggest that chance cannot be a complete
explanation. However, the confidence intervals are relatively wide, so
that the magnitude of true association could be as low as a < 1-point
(rather than a 2-point) deficit, or as high as > 3 points.
b) Confounding factors The adjusted lead/IQ associations tend to be
substantially weaker than the unadjusted associations, which indicates
that confounding factors are important. Since none of the studies can
claim to have taken complete account of confounders, (e.g., father's
IQ is not included), it seems likely that some of the remaining
lead/IQ relationship, after adjustment, may still be attributable to a
degree of unexplained confounding.
c) Reverse causality One initial justification for the prospective
studies was that they could measure early exposure to lead and relate
it to later child development, thus removing the problem of reverse
causality potentially present in the cross-sectional studies. However,
it has turned out that very early lead exposure, e.g., in the
perinatal period, is not related to school-age IQ. The question arises
as to whether children of lower IQ could exhibit behaviour patterns at
earlier ages, e.g., around age 2 and older, which could enhance their
uptake of lead. Perhaps, such reverse causality remains a possibility
even in the prospective studies.
d) Selection biases The positive studies may perhaps be more likely
to report in more quantitative details, as indicated by the absence of
relevant data from Cleveland in the prospective studies. Post-hoc
selection based on the more significant PbB concen-trations may lead
to exaggerated estimates.
It is a matter of debate and conjecture as to the extent to which
these four issues should inhibit claims of a causal relationship in
the prospective studies. The essential problem is that observational
epidemiology cannot provide definitive evidence of causality when the
key statistical association is weak, the temporal relationship is
unclear and major confounders are present.
8.3.7 Hearing impairment in children
Schwartz & Otto (1987) reported that the probability of elevated
pure-tone hearing thresholds at 500, 2000 and 4000 Hz increased
significantly with increasing PbB level in 4519 subjects (4-19 years
of age) who participated in the NHANES II study. Variables included in
the backwards stepwise regression models included information derived
from medical history, clinical examinations, and assessment of
developmental milestones.
Schwartz & Otto (1991) examined data for subjects (6-19 years of
age) from the Hispanic Health and Nutrition Examination Survey
(HHANES) study which included three distinct ethnic groups. After
excluding 283 subjects with previous ear problems (discharges,
ruptured ear drums or tinnitus), data for 3262 were analysed. The
authors concluded that increasing PbB level in the range
0.36-0.86 µmol/litre (7-18 µg/dl) was associated with approximately
2 dB loss of pure-tone hearing at frequencies of 500, 1000, 2000 and
4000 Hz.
Dietrich et al. (1992) assessed the relationship between scores
on a test of sensory auditory processing (SCAN) and prenatal/postnatal
PbB concentrations in 215 subjects drawn from the Cincinnati
prospective cohort study. Higher prenatal, neonatal and postnatal PbB
concentrations were associated with more incorrect identification of
common monosyllabic words presented under conditions of muffling.
Other variables associated with impaired central auditory processing
were pure-tone audiometry results, social class, quality of caretaking
in the home, birth weight, gestational age, a measure of obstetrical
complications, and consumption of alcohol during pregnancy. Following
adjustment for these co-factors, lifetime average PbB concentration
remained significantly and inversely associated with SCAN performance.
8.4 Renal system
Acute exposure to lead is known to cause proximal renal tubular
damage, characterized by generalized aminoaciduria, hypophosphataemia
with relative hyperphosphaturia, and glycosuria (Chisolm, 1962).
Cellular structural changes include nuclear inclusion bodies,
mitochondrial changes and cytomegaly of the proximal tubular
epithelial cells (Cramer et al., 1974). Diagnosis of lead-induced
altered renal function or disease is difficult since there are no
specific indicators; blood urea nitrogen (BUN) and creatinine levels
become elevated only when twothirds of renal function has been lost
(Bernard & Becker, 1988).
8.4.1 Clinical studies
From a study of seven men occupationally exposed to lead in a
shipyard during oxy-acetylene flame cutting of lead-painted steel
hulls, Cramer et al. (1974) concluded that there is a continuum of
morphological and functional change in the pathogenesis of chronic
lead nephrotoxicity. Each subject was treated in hospital 3 or more
days after recent exposure and PbB levels exceeded 3.36 µmol/litre
(70 µg/dl) in all cases. On the basis of microscopic examination of
biopsy tissue, nuclear inclusion bodies were reported in proximal
tubular cells during the early phase but there was no impairment of
renal function. In a second phase, there was fibrosis associated with
asymptomatic azotaemia and reduced glomerular filtration rate but
without demonstrable proximal tubular dysfunction; renal failure was
not seen in the study.
Weeden et al. (1979) diagnosed lead nephropathy in 15 lead
workers, all having reduced glomerular filtration rates. Renal
biopsies of six of the subjects showed focal interstitial nephritis in
addition to non-specific changes in the proximal tubules. At the time
of examination, PbB levels for 11 of the 15 workers were within the
range 1.92-3.84 µmol/litre (40-80 µg/dl).
Baker et al. (1979) reported increased BUN and decreased
creatinine clearance in 28 workers, all of whom had relatively
prolonged, high-dose lead exposure in lead smelting or chemical
manufacturing.
A study by Maranelli & Apostoli (1987) of 60 workers, described
as "lead poisoned" and having PbB levels of 3.45 ± 0.80 µmol/litre
(71.9 ± 16.6 µg/dl), found no definitive correlation between PbB, lead
in urine after chelation, and BUN, serum creatinine and serum uric
acid.
8.4.2 Epidemiological studies
8.4.2.1 Occupational cohorts
Several recent studies of lead-exposed workers provide further
information relating to dose-effect relationships. The emphasis in
these studies was on lead and its possible association with adverse
effects on health. However, exposure to other potentially toxic
substances in the working and living environment should not be
overlooked/ignored.
For example, Buchet et al. (1980) examined 25 male lead smelter
workers and 88 male control workers. The PbB levels of the lead
workers were in the range 1.62-2.94 µmol/litre (33.8-61.3 µg/dl) for a
mean of 13.2 years (range 3.1-29.8 years) of lead exposure. The PbB
levels for the controls were in the range 0.26-1.64 µmol/litre
(5.5-34.2 µg/dl). There were no differences for parameters of renal
function between the groups and no signs of clinical renal impairment.
It was concluded that a PbB level of less than 2.98 µmol/litre
(62 µg/dl) is not associated with renal toxicity.
N-acetyl-ß-d-glucosaminidase (NAG) is a lysosomal enzyme present
in renal tubular cells. This enzyme is a sensitive but non-specific
indicator for early sub-clinical nephrotoxicity. In a study on 29
lead-exposed workers, Meyer et al. (1984) found increased NAG in
urine, but there was no correlation with PbB level. However, NAG level
was found to be normal in the 5 subjects with PbB levels exceeded
3.36 µmol/litre. These authors speculated that long-term high exposure
to lead may deplete the kidneys of NAG or render it insensitive to the
effects of lead exposure.
Verschoor et al. (1987) studied 155 male lead workers and 126
control workers who were matched for age, smoking habits,
socioeconomic status and duration of employment. The PbB levels of the
lead workers were in the range of 0.43-4.71 µmol/litre
(8.3-97.6 µg/dl) compared with the controls 0.15-0.96 µmol/litre
(3.1-18.8 µg/dl). The lead workers had elevated ZPP levels
(34-292 µmol/mol haemoglobin) compared with controls (10-35 µmol/mol
haemoglobin). No significant differences were found for various
indicators of renal function; all urinary and serum parameters were
within normal ranges. There were no differences in protein excretion
patterns and no signs of renal impairment. However, the authors found
that NAG levels in the lead-exposed workers were higher than control
values and increased with increasing PbB levels. They concluded that
lead exposure resulting in PbB levels of under 3.0 µmol/litre
(62 µg/dl) can affect renal tubular functions as measured by NAG
excretion; lead appeared to affect the tubular parameters more than
the glomerular parameters in moderately exposed workers.
Ong et al. (1987) examined 158 male and 51 female lead battery or
smelter workers and 30 control workers. The lead workers had 1-36
years exposure with an average of 10.8 ± 8.0 years and PbB levels in
the range of 0.14-3.84 µmol/litre (3-80 µg/dl); only five workers
exceed 2.88 µmol/litre (60 µg/dl). The authors found a weak but
statistically significant positive association between PbB and blood
urea nitrogen, and between PbB and serum creatinine, and that
creatinine clearance was reduced with increasing PbB level. NAG levels
in the lead-exposed workers were significantly higher than control
values and increased with increasing urine lead level when the data
were adjusted for age. These authors concluded that a relatively low
PbB level can affect renal function.
Not all investigators have found parallel association between PbB
and NAG levels. Gennart et al. (1992b) compared 98 lead-exposed lead
acid battery workers (mean PbB level 2.45 µmol/litre, 51 µg/dl,
geometric mean ZPP 10.2 µg/g Hb) with 85 controls (mean PbB level
1.00 µmol/litre, 20.9 µg/dl, geometric mean ZPP 2.84 µg/g Hb)
recruited from other departments in the same factory. None of the
indicators of renal function (retinol-binding protein,
ß2-microglobulin, albumin or NAG in urine, or creatinine or
ß2-microglobulin in serum) were correlated with PbB level, duration
of exposure or ZPP, or showed significantly different mean values
between the lead-exposed and control groups of workers.
Cardenas et al. (1993) recently evaluated 27 different
indications of renal dysfunction in 50 workers exposed to lead and 50
controls. There were significant increases in urinary NAG and sialic
acid in the lead exposed group. These changes may represent minor
cellular modifications rather than significant functional or
irreversible renal damage. There was a significant decrease in urinary
6-keto PGF1alpha and a significant increase in TXB2. These
eicosanoid changes may reflect systemic functional vascular changes
rather than an effect of lead on the kidney.
8.4.2.2 General population
An epidemiological survey on 283 persons in Scotland, from
households with water lead concentrations in excess of 100 µg/litre,
revealed a close correlation between water lead content and PbB and
serum urea concentrations (Campbell et al., 1977). The frequency of
renal dysfunction in individuals with elevated PbB concentrations
(> 2 µmol/litre or > 41 µg/dl) was significantly greater than that
of age- and sex-matched controls.
Pocock et al. (1984) measured serum creatinine, urate and urea
concentrations in 7364 British men, and 74 subjects had PbB levels
equal to or greater than 1.8 µmol/litre (37.3 µg/dl). The authors
concluded that there was no indication that exposure to lead at
concentrations commonly encountered in Britain was responsible for
impaired renal function.
Staessen et al. (1992) investigated the relationship between lead
exposure and renal function as part of a cross-sectional population
study of the health effects of environmental exposure to cadmium.
Creatinine clearance, PbB and ZPP were measured in a random population
sample of 965 men (geometric mean PbB level was 0.55 µmol/litre or
11.4 µg/dl) and 1016 women (geometric mean PbB level was
0.36 µmol/litre or 7.5 µg/dl). Creatinine clearance rate (mean was
99 ml/min in men, 80 ml/min in women) was inversely correlated with
PbB and ZPP levels before and after adjustment for age, body mass
index and diuretic treatment. Also positively correlated were serum
ß2-microglobulin and PbB level in men, serum ß2-microglobulin and ZPP
in men and women, and serum creatinine and ZPP in men. Impaired renal
function could not be explained by exposure to cadmium or by elevated
blood pressure. The authors concluded that exposure to lead may impair
renal function in the general population. However, the study could not
exclude the possibility that renal impairment may lead to an increase
in PbB level.
8.4.2.3 Cohort mortality studies
"Lead poisoning" was a diagnosis given to 241 workers employed
for 1-30 years at the Port Pirie lead smelter between 1928-1959 by a
State medical board. Death registration records for the period
1930-1977 identified 140 deaths among the group. The cause of death
profile was compared with that of 695 other male decedents,
predominantly production workers and a smaller number of office
workers. Age-standardized mortality analysis revealed a substantial
excess of deaths attributed to chronic nephritis and to cerebral
haemorrhage. The rates for lead-poisoned workers exceeded those for
the other workers, and both exceeded the rates for the Australian
general male population. The rates decreased over successive calendar
periods but excess rates persisted for chronic nephritis up until 1977
(McMichael & Johnson, 1982).
8.5 Cardiovascular system
Two persistent issues have been under intense study since
publication of Environmental Health Criteria 3: Lead (IPCS, 1977):
a) whether lead is a factor in hypertension and, if so, whether
there is a causal relationship;
b) whether lead is contributory to cardiovascular effects
influencing morbidity or mortality.
8.5.1 Blood pressure
8.5.1.1 Studies on occupationally exposed cohorts
From a study of 431 white male police officers 24-55 years of age
(Moreau et al., 1982; Orssaud et al., 1985), it was concluded that
systolic and diastolic blood pressure was related to PbB level, the
correlation being greatest for the younger subjects and decreasing
with age. Statistical adjustment was made for alcohol consumption and
body mass index, but not for smoking. The magnitude of observed
association between systolic pressure and PbB level in this small
study was somewhat greater than in the larger general population
studies cited below.
Parkinson et al. (1987) studied 270 lead battery workers and 158
non-exposed workers and compared lead exposure with systolic and
diastolic blood pressures. After controlling for age, education,
income, cigarette smoking, alcohol consumption and exercise, there was
a small and non-significant association. The average PbB level of
lead-exposed workers was 1.92 ± 0.62 µmol/litre (40 ± 13 µg/dl),
whereas in non-exposed workers it was 0.34 ± 0.24 µmol/litre
(7 ± 5 µg/dl).
8.5.1.2 Studies in the general population
The possible relationship between PbB concentration and blood
pressure has been examined in several large-scale population studies.
These include the British Regional Heart Study (BRHS), the US NHANES
II (National Health and Nutrition Examination Survey) and studies in
Wales, Denmark, Canada and Belgium.
The BRHS is a prospective study of 7735 men, initially aged 40-59
years, from 24 British towns, who were first examined in 1978-1980
(Shaper et al., 1981). The median PbB level was 0.7 µmol/litre
(14.5 µg/dl). The initial findings (Pocock et al., 1984), which
indicated a lack of association between PbB level and blood pressure,
were later re-examined to take account of potential confounding
factors such as alcohol, smoking and town of residence. Pocock et al.
(1988) noted that 95% confidence intervals for systolic and diastolic
blood pressure plotted against PbB concentrations overlapped and
showed no elevation even at the highest observed PbB concentration.
Applying statistical techniques to adjust mean systolic and diastolic
blood pressure measurements for body mass index, age, alcohol
consumption, cigarette smoking and town of residence showed signs of a
weak association, particularly for diastolic pressure and PbB level.
Multiple regression analysis showed that adjustment for personal
characteristics but not for town of residence rendered both systolic
and diastolic regressions on log PbB insignificant, chiefly because
alcohol consumption is an important confounder, being positively
related to both blood pressure and PbB level. Introducing an extra
adjustment for town of residence made the systolic and diastolic
regressions highly statistically significant, even though the
associations were weak. These cross-sectional data indicated that for
every doubling in PbB level (e.g., from 0.8 to 1.6 µmol/litre) there
are estimated mean increases of 1.45 mmHg (95% confidence interval
0.47-2.43 mmHg) in systolic blood pressure and 1.25 mmHg (95%
confidence interval 0.65-1.85 mmHg) in diastolic blood pressure.
The NHANES II study, a USA national cross-sectional survey
carried out in 1976-1980, included PbB and blood pressure measurements
in a general population sample of 5803 men and women aged 12-74.
Geometric mean PbB levels were around 0.72 µmol/litre (about 15 µg/dl)
for men and approximately 0.53 µmol/litre (11 µg/dl) for women.
Several authors have examined these data for possible PbB and blood
pressure associations.
In an analysis controlling for the confounding factors of age,
race and body mass index, Harlan et al. (1985) and Harlan (1988) found
a significant association between PbB level and blood pressure for men
but not for women. White males aged 40-59 years from the same data set
were analysed by Pirkle et al. (1985), and the correlation between PbB
level and blood pressure was confirmed. In further analysis of the
whole age range, adjustments for possible time trend and geographical
site effects did not affect the significance of the association in
males (Landis & Flegal, 1988; Schwartz, 1988), although its magnitude
did become somewhat less pronounced.
Gartside (1988) applied a method of forward stepwise regression
to PbB and blood pressure data from NHANES II and reported that the
results for white men, white women and black men were contradictory
and lacked consistency and reliability. He noted that the overall
average was too small to conclude association between PbB level and
blood pressure in this study.
Two surveys were carried out in Wales by Elwood et al. (1988a,b).
The Welsh Heart Programme carried out in 1985 provided complete PbB,
blood pressure and other data for 865 men and 856 women aged 18-64.
The geometric mean PbB level for men was 0.56 µmol/litre (11.6 µg/dl)
and for women was 0.43 µmol/litre (9.0 µg/dl). In neither sex was
there a significant correlation between PbB level and systolic or
diastolic blood pressure. The second study was in a cohort of men aged
49-65 years living in Caerphilly, Wales. The geometric mean PbB level
(N = 1137) was 0.61 µmol/litre (12.7 µg/dl). Complete data from 1137
subjects, ranking blood pressure readings according to PbB level, did
not reveal any trend in the percentage of subjects with systolic
pressure above 160 mmHg. The authors corrected only for age as a
confounding factor.
Data from a Canadian study (Neri et al., 1988) collected during
10 months in 1978-1979 for 2193 subjects aged 25-64 showed a weak but
statistically significant association between PbB level and diastolic
blood pressure.
Grandjean et al. (1989b) studied 504 men and 548 women residing
in Glostrup (Denmark) at age 40; 451 men and 410 of the women were
followed-up five years later. Average PbB levels for the men at 40 and
45 years of age were 0.62 µmol/litre (13 µg/dl) and 0.43 µmol/litre
(9 µg/dl), and for the women 0.43 µmol/litre (9 µg/dl) and
0.29 µmol/litre (6 µg/dl), respectively. All correlations found
between PbB and blood pressure became insignificant when blood
haemoglobin and alcohol intake were entered into multiple regression
analyses; an independent effect of low-level lead exposure on blood
pressure could not be distinguished.
Möller & Kristensen (1992) examined 1052 men and women in
Copenhagen, Denmark, in 1976, and re-examined them in 1981 and 1987
(men only). Initial mean PbB levels were 0.65 µmol/litre (13.6 µg/dl)
in men and 0.46 µmol/litre (9.6 µg/dl) in women, and had fallen by
around 30% 5 years later. For men, there was no significant
association between PbB level and blood pressure (or between changes
in PbB level and blood pressure) after adjustment for confounders
(tobacco, alcohol, body mass and physical activity). For women, the
association between PbB level and diastolic (but not systolic) blood
pressure remained significant on both occasions, even after
controlling for confounders.
A Belgium study (Dolenc et al., 1993) included 827 men and 821
women (mean age 25) whose mean PbB levels were 0.56 and
0.34 µmol/litre (11.6 and 7.07 µg/dl), respectively. After adjustment
for covariates (body mass index), pulse rate and serum creatinine and
serum calcium levels), systolic blood pressure was inversely
associated with PbB level in men (P < 0.05). There were no
significant PbB associations for diastolic blood pressure in men or
for either pressure in women.
A study of 398 male and 133 female civil servants (Staessen et
al., 1990) showed geometric mean PbB levels of 0.58 µmol/litre
(12.06 µg/dl) and 0.46 µmol/litre (9.56 µg/dl) in men and women,
respectively. Taking the data for both sexes together, there were
statistically significant positive associations between PbB level and
systolic and diastolic blood pressures (r = +0.11 in each case) but
these became non-significant after adjustment for confounders (sex,
age, BMI, pulse, delta-glutamyltranspeptidase (delta-GTP) and serum
calcium level).
Another cross-sectional study involving only women was performed
in Boston (Rabinowitz et al., 1987). Cord PbB levels (mean
0.33 µmol/litre, 6.9 µg/dl) among 3851 women correlated with both the
systolic (r = 0.08) and diastolic (r = 0.05) blood pressures measured
during and before delivery. Multivariate models of pregnancy
hypertension as a function of age, parity, haematocrit, diabetes,
ponderal index, and race were improved when lead was included as a
predictor. Lead appeared to have a small but demonstrable association
with pregnancy hypertension and blood pressure at delivery, but not
with eclampsia.
In an overview of the BRHS, NHANES II and Welsh studies, Pocock
et al. (1988) concluded that with overlapping confidence limits the
data provided weak but reasonably consistent evidence of lead and
blood pressure associations. They noted that the NHANES II data on
2254 men in the USA indicate a slightly stronger association between
PbB level and systolic blood pressure, whereas data from over 2000 men
in Wales did not show a statistically significant association. They
inferred that a causal relationship could not be concluded from any of
the epidemiological studies.
Staessen et al. (1994) have undertaken a more extensive
meta-analysis of nearly all of the above studies and some other
smaller ones as well. This amounted to a total of 19 studies with
28 210 subjects. They found that the association between PbB level and
blood pressure was similar in both sexes. In all studies combined, a
two-fold increase in PbB concentration was associated with a 1.0 mmHg
increase in systolic pressure (95% confidence interval (CI); 0.3 to
1.7 mmHg; P = 0.008) and a 0.7 mmHg increase in diastolic pressure
(95% CI: 0.2 to 1.3 mmHg; P = 0.02). These authors conclude that the
published evidence suggests a weak positive association between blood
pressure and lead exposure, but any such relationship may not be
causal and is unlikely to entail any public health implications
regarding hypertension.
8.5.2 Other cardiovascular effects
As recorded previously (IPCS, 1977), there is good evidence that
signs of clinical lead poisoning sometimes include evidence of toxic
action on the heart. Kopp et al. (1988) reviewed the cardiovascular
actions of lead and concluded that the degree of cardiovascular
involvement during episodes of acute lead intoxication depends on the
duration of exposure and dose. It is not known whether environmental
exposure to lead affects the electrical or mechanical activity of the
heart.
8.5.2.1 Occupational studies
There have been several studies relating to cardiovascular
effects in lead-exposed occupational groups. However, as with any
study of an occupational cohort, it is important to evaluate the
strength of the cause-effect relationship, the nature or substance of
other contributory factors and how well the workplace "exposure" has
been evaluated for other potential toxicants. Thus, the occupational
hazards associated with work in lead smelting or lead battery
operations include, but are not limited to, lead.
8.5.2.2 Studies in the general population
Pocock et al. (1988) found that after 6 years of follow-up of the
BRHS cohort of 7735 middle-aged men, 316 of the men had major
ischaemic heart disease and 66 had had a stroke. After allowance for
confounding effects of cigarette smoking and town of residence, there
was no evidence that PbB was a risk factor for these cardiovascular
events.
Möller & Kristensen (1992) studied 1052 men and women in
Copenhagen, Denmark, and related blood pressure to subsequent
cardiovascular morbidity and mortality over a 14-year period. There
were significant positive associations between PbB level and both
coronary and cardiovascular disease in univariate analysis, but these
became non-significant after controlling for confounders.
a) Clinical studies
Boscolo & Carmignani (1988) measured plasma renin activity in
hospitalized lead-exposed workers and concluded that synthesis or
release of renin is increased by short or moderate exposure to lead
and decreased with prolonged exposure.
b) Epidemiological studies
Kirkby & Gyntelberg (1985) found a significantly higher incidence
of ischaemic ECG changes in a study of lead smelter workers (20%, mean
PbB level 2.45 µmol/litre, 51 µg/dl), compared with matched
non-exposed controls (6%, mean PbB level 0.53 µmol/litre, 11 µg/dl).
There was also a slight (4-5 mmHg) increase in diastolic blood
pressure in the lead workers compared with the controls.
c) Cohort mortality studies
Two studies (Cooper, 1988 and Fanning, 1988) reported an
increased mortality rate of lead workers due to circulatory disease,
but no such relationship was found in reports of studies by
Gerhardsson et al. (1986) or Selevan et al. (1988). Gerhardsson et al.
(1986) examined a lead-exposed sub-cohort of 437 from a study cohort
of 3832 male workers first employed at a copper smelter before 1967
and followed up from 1950 to 1981. There was no excess mortality due
to ischaemic heart disease or cerebrovascular disease in the
lead-exposed worker sub-cohort. Selevan et al. (1988) found no
association between lead exposure and deaths due to hypertensive
diseases in a review of mortality in a cohort of male hourly workers
at a lead smelter. There were many confounding factors in each of
these studies.
8.5.3 Summary
Despite intensive efforts to define the relationship between body
burden of lead and blood pressure or other effects on the
cardiovascular system, no causal relationship has been demonstrated in
humans and the mechanisms remain obscure.
There is experimental evidence from animal studies indicative of
an effect of lead on blood pressure, and several mechanisms have been
proposed to explain these observations (see section 7.4).
8.6 Gastrointestinal effects
8.6.1 Occupational exposure
Colic is a well-recognized symptom of acute lead poisoning and is
still reported in groups of lead-exposed industrial workers. Of
particular concern are the reports of intoxication caused by acute
exposure (inadequate protective measures) to lead associated with
removal of lead-based paint by burning or sand-blasting and the
demolition of lead-containing industrial plants. Symptoms of colic
include abdominal pain, constipation, cramps, nausea, vomiting,
anorexia, weight loss and decreased appetite.
Symptoms in adults typically occur at PbB levels of
4.8-9.6 µmol/litre (100-200 µg/dl) but have been noted at levels as
low as 1.92 µmol/litre (40 µg/dl) (Baker et al., 1979; Haenninen et
al., 1979; Awad El Karim et al., 1986; Pollock & Ibels, 1986; Muijser
et al., 1987; Holness & Nethercott, 1988; Marino et al., 1989;
Pagliuca et al., 1990; Schneitzer et al., 1990).
Positive histories of lead colic were given by 40 workers in a
cohort of 158 secondary lead smelter workers who participated in a
clinical field survey (Lilis et al., 1977). It was reported most
frequently by those having PbB levels above 3.84 µmol/litre (80 µg/dl)
at the time of examination and was not found where ZPP was within the
normal range. Thirty percent of workers having ZPP > 200 µg/g Hb had
experienced symptoms.
In a study of a population of 585 black South African factory
workers, Irwig et al. (1978) reported that the incidence of abdominal
pain increased with increasing PbB level: it was 12% for the first
quartile (PbB < 3.28 µmol/litre or < 68 µg/dl), 23% for the
second (3.28-4.14 µmol/litre or 68-86 µg/dl), 24% for the third
(4.15-5.14 µmol/litre or 86-107 µg/dl) and 37% for the fourth
(> 5.15 µmol/litre or > 107 µg/dl).
In a cross-sectional clinical study of 90 telephone
cable-splicers by Fischbein et al. (1980), 19 (21%) workers reported
gastrointestinal symptoms (mean PbB level of 1.44 µmol/litre, or
30 µg/dl and mean ZPP of 66.6 µg/g Hb) whereas 71 (79%) reported no
such symptoms (mean PbB level of 1.3 µmol/litre or 27 µg/dl and mean
ZPP of 52.3 µg/g Hb).
8.6.2 Exposure of children
Colic is seen in children and US EPA (1986a) concluded that the
lowest-observed-adverse-effect level was in the range of
2.88-4.80 µmol/litre (60-100 µg/dl).
8.7 Liver
There appears to be no new evidence relating human body lead
burden to effects on the liver, but it has been suggested that the
effects of lead on haem synthesis may alter the functional capacity of
the hepatic cytochrome P-450 system to metabolize drugs.
8.7.1 Occupational exposure
Fischbein et al. (1977) tested five demolition workers acutely
exposed to lead for a period of 3 months prior to the study. They
found that the plasma half-life of an oral dose of antipyrine was
within the range for normal healthy volunteers; it was shorter in each
subject after chelation therapy but still within the normal range. The
plasma half-lives of phenylbutazone were also within the normal range
but were unaffected by chelation therapy.
8.7.2 Exposure of children
Saenger et al. (1984) found decreased urinary excretion of
6-ß-hydroxycortisol in 26 children with a mean PbB level of
2.11 µmol/litre (44 µg/dl); the decreased formation of the metabolite
was attributed to lead inhibition of the cytochrome P-450-dependent
mixed-function oxidases.
8.8 Reproduction
While it is generally accepted from early literature that lead
adversely affects the reproductive process in both men and women, the
evidence is mostly qualitative and dose-effect relationships have not
been established.
Most new information relates to reports of occupational cohorts
and of populations living in polluted areas near industrial plants.
There is qualitative evidence that lead is toxic to the reproduction
system in both men and women. However, there are insufficient data to
provide the basis for estimation of dose-effect relationships in
women.
8.8.1 Female populations
Nordstrom et al. (1978b) reported an increased frequency of
spontaneous abortion in women living close to a smelter in northern
Sweden. In a later report, Nordstrom et al. (1979) described the
responses to a questionnaire completed by 511 of 662 women who had
worked at the smelter and were born between 1930 and 1959. Spontaneous
abortion rates were highest in those pregnancies in which the mother
was employed during the pregnancy (13.9%) or had been employed before
the pregnancy and was living close to the smelter (17%); the frequency
rate was higher (19.4%) when the father worked at the smelter.
However, it should be noted that the smelter produced copper and lead
in addition to a number of other metallurgical and chemical products
(Nordstrom et al., 1978a) and that the effects reported may not
necessarily be attributable exclusively to lead.
A study of pregnancies in the centre and surrounding areas of the
lead smelter town of Port Pirie showed that the incidence of
miscarriages (22 or 23) and stillbirths (10 or 11) was higher in women
living close to the smelter (McMichael et al., 1986). However, in a
study of 639 Yugoslav women, the risk of spontaneous abortion was not
increased (OR = 1.1, 95% CI: 0.9-1.4) among women with mean PbB levels
of 0.77 µmol/litre (16.0 µg/dl) as compared with women with mean PbB
levels of 0.25 µmol/litre (5.2 µg/dl) (Murphy et al., 1990). Risk did
not vary with distance of residence from the smelter.
Some studies have found decreased length of gestation in women
whose PbB levels were greater than 1.09 µmol/litre (23 µg/dl) (Moore
et al., 1982), 0.58 µmol/litre (12 µg/dl) (Dietrich et al., 1986) or
0.72 µmol/litre (15 µg/dl) (McMichael et al., 1986). However, neither
Bellinger et al. (1991b) nor Graziano et al. (1990) found decreases in
gestational length or other parameters of pregnancy in women with
elevated PbB levels.
In the Cincinnati prospective study, a significant reduction in
birth weight associated with prenatal (maternal) PbB levels, after
adjustment for covariates, was reported by Bornschein et al. (1989).
In the Port Pirie prospective study of 749 pregnancies (McMichael et
al., 1986), the proportion of pregnancies resulting in low birth
weight singleton infants was more than twice as high in Port Pirie
women (whose PbB levels averaged 0.50 µmol/litre, 10.4 µg/dl) than in
women outside Port Pirie (average PbB level of 0.264 µmol/litre,
5.5 µg/dl). On the other hand, multiple regression analyses showed no
significant association between low birth weight and maternal PbB
level. In a cross-sectional study, Ward et al. (1987) reported a
significant simple relationship between placental lead concentrations
and reduced birth weight and head circumference.
Other studies have not shown a significant association between
birth weight and lead exposure. The Kosovo prospective study failed to
detect any evidence of lead-related birth weight reduction in more
than 900 births (Murphy et al., 1990), and Ernhart et al. (1986) found
no significant effect of lead on birth weight, birth length or head
circumference.
8.8.2 Male populations
Reproductive effects from occupational exposure to lead include
asthenospermia, hypospermia, teratospermia and hypogonadism (US EPA,
1986a; Braunstein et al., 1987).
In men, effects on sperm or the testes may result from chronic
exposure to lead at blood levels of the order of 1.92-2.4 µmol/litre
(40-50 µg/dl) (ATSDR, 1991).
Wildt et al. (1983) compared two groups of lead-exposed storage
battery workers and concluded that the highly exposed group (PbB of
more than 2.16 µmol/litre, 45 µg/dl) had decreased prostate/seminal
vesicle function, low semen volume and poorer functional maturity of
sperm.
Chowdhury et al. (1986) reported a significant decrease in sperm
count and motility and an increased count of abnormal spermatozoa in
lead-exposed men (average PbB level of 2.04 µmol per litre or
42.5 µg/dl) compared with controls (0.71 µmol/litre or 14.8 µg/dl).
Assenato et al. (1987) also reported decreased sperm production in 18
battery factory workers having PbB levels of 2.4-2.93 µmol/litre
(50-61 µg/dl).
In five out of six symptomatic lead intoxicated workers having
PbB levels in the range of 1.87-4.7 µmol/litre (39-98 µg/dl) at the
time of examination, Cullen et al. (1984) found defects of
spermatogenesis, including oligospermia and azoospermia.
The results of Lerda (1992) also showed significant increases in
asthenospermia and teratospermia among 38 battery workers with PbB
levels of 1.95-4.7 µmol/litre (40.5-98.0 µg/dl) compared to a control
group with PbB levels of 0.86-1.25 µmol/litre (17.6-26.0 µg/dl).
An epidemiological study by Lindholm et al. (1991) of 213 wives
of lead workers and 300 matched controls suggested an association
between paternal lead exposure and the risk of spontaneous abortion
among the wives of lead workers when the PbB level exceeded
1.5 µmol/litre (31 µg/dl) close to the time of spermatogenesis.
However, the authors acknowledged that exposure of the study subjects
to other metals and organic solvents may have influenced the results
of the study.
A weak association between paternal exposure, estimated from
occupational history, and risk of esotropia (inward deviation
strabismas) was noted in a case control study of 377 children aged 7
years (OR - 2.4, 95%, CI: 1.0-5.6) (Hakim et al., 1991). However, the
increased risk was not dose-related, and the risk of exotropia
(outward deviation) was not associated with higher paternal lead
exposure.
8.8.3 Hormonal responses
Assenato et al. (1987) found no significant differences in FSH,
testosterone, prolactin, LH and total neutral 17-ketosteroids levels
between 18 lead battery workers (PbB level, 2.4-3 µmol/litre or
50-61 µg/dl) and 18 cement workers (PbB level, 0.9-1.1 µmol/litre or
18-22 µg/dl), despite the finding of oligospermia in the lead-exposed
group.
Rodamilans et al. (1988) studied the endocrine status of 23 lead
smelters in relation to the duration of lead exposure. Five workers
exposed for less than 1 year (mean PbB level, 0.29 µmol/litre or
66 µg/dl) showed an increase in serum LH level, while that of
testosterone remained normal. Eight workers exposed for 1-5 years
(mean PbB level, 3.50 µmol/litre or 73 µg/dl) and 10 employed for more
than 5 years (mean PbB level, 3.65 µmol/litre or 76 µg/dl) showed an
increase in LH comparable with the group exposed for less than 1 year
but there was a clear reduction in serum testosterone levels.
Gustafson et al. (1989) found a lower plasma FSH level in 25
moderately exposed workers (mean PbB level, 1.82 µmol/litre or
38 µg/dl) than in 25 matched controls. A lower mean serum LH level was
reported by McGregor & Mason (1990) in 90 lead workers (mean PbB
level, 2.20 µmol/litre or 46 µg/dl) than in controls; there was also a
correlation between serum FSH and PbB levels. In an epidemiological
study involving 122 current lead workers (PbB mean level,
1.69 µmol/litre or 35.1 µg/dl) and 49 non-exposed workers (PbB mean
level, 0.4 µmol/litre or 8.3 µg/dl), Ng et al. (1991) found slightly
higher plasma LH and FSH levels among the exposed workers. The
testosterone level, however, was not significantly different between
the two groups. In contrast, Gennart et al. (1992) did not find an
effect of lead on various endocrine parameters (including FSH and LH
levels) in a study population of 221 workers having a geometric mean
PbB level of 2.45 µmol/litre (51.0 µg/dl). They suggested that the
hypothalamic-pituitary system may not be influenced by moderate
exposure to lead.
8.8.4 Postnatal growth and stature
Several reports have suggested that the physical growth and
stature of children may be reduced by exposure to lead (e.g., Mooty et
al., 1975; Johnson & Tenuta, 1979; Routh et al., 1979), but the
influence of other factors (e.g., race and diet) has often made it
difficult to isolate lead as a causal agent for such effects in human
populations. Multivariate regression analyses of NHANES data for
approximately 2700 children in the USA (Schwartz et al., 1986)
provided more convincing evidence of a significant association between
increasing PbB levels and reduced height, weight, and chest
circumference after adjusting for age, race, sex and nutritional
covariates.
8.9 Effects on chromosomes
As noted in Environmental Health Criteria 3: Lead (IPCS, 1977),
the literature remains controversial concerning induction of
chromosomal changes in human lymphocytes by in vivo exposure to
lead. Most of the studies are of small numbers of subjects in
occupational groups where lead was one of many potentially toxic
agents. However, some of the difficulties arise because of the lack of
standard procedures used for the assessment of chromosomal effects in
cultured lymphocytes and from a lack of understanding of the health
significance of chromosomal abnormalities.
Induced mitotic activity in peripheral lymphocytes and increases
in the rates of abnormal mitosis were reported by Sarto et al. (1978).
PbB levels in the range of 1.05-4.27 µmol/litre (22-89 µg/dl) were
reported to be associated with increased incidence of chromosomal
aberrations by Schmid et al. (1972) in 32 lead manufacturing workers,
by Nordenson et al. (1978) in 18 smelter workers and by Al-Hakkak et
al. (1986) in 19 lead manufacturing workers. It should be noted that
exposure of worker to potentially hazardous agents other than lead was
not documented in these studies.
Maki-Paakkanen et al. (1981) reported no change in chromosomal
aberration frequency among workers with PbB levels in the range of
1.82-5.76 µmol/litre (38-120 µg/dl); weak effects were found by Huang
et al. (1988b) when the PbB level exceeded 2.5 µmol/litre (52 µg/dl).
Qazi et al. (1980) reported increased numbers of cells with
chromosome breaks in a baby having a cord PbB level of 2.88 µmol/litre
(60 µg/dl) and a PbB level of 3.45 µmol/litre (72 µg/dl) at 2 weeks.
The effect was seen at 6 weeks and at 3 months but not later
(chelation therapy was given at 17 days and repeated at 5 months).
Bauchinger et al. (1977) tested blood taken from 38 children
selected from a school situated close to a lead plant and in whom the
PbB level was at least 1.44 µmol/litre (30 µg/dl). There was no
increase in the frequency of aberrations.
Grandjean et al. (1983) examined 10 long-term workers at a lead
storage battery plant. They found normal or slightly increased SCE
rates in workers with a PbB level of 1.4-3.6 µmol/litre (29-75 µg/dl)
and a ZPP level of 50-750 µmol/mol haemoglobin. The SCE increase was
correlated significantly with ZPP level.
Huang et al. (1988b) studied 21 lead-exposed workers from a
battery factory. They concluded that the SCE rate in workers with
long-term exposure to lead increased significantly when the mean PbB
level was 3.84 µmol/litre (80 µg/dl) or more. There were no
significant differences in SCE rates among the low- and medium-exposed
groups or the controls. These authors concluded that the effect of
chromosome damage caused by lead is not very strong.
Dalpra et al. (1983) examined blood samples taken from 19
children living in a contaminated area near a smelter and having PbB
levels in the range of 1.39-3.02 µmol/litre (29-63 µg/dl). They found
no effect on SCE frequency.
8.10 Carcinogenicity
Much has been written regarding the evidence that lead is
carcinogenic in humans (Moore & Meredith, 1979; Kazantzis, 1989;
Goyer, 1992). In several large epidemiological studies no association
was found which would associate lead with induction of cancer (Cooper
& Gaffey, 1975; Kang et al., 1980; Cooper, 1981; McMichael & Johnson,
1982). One major difficulty in many of the studies was the concurrent
exposure to potential carcinogens such as chromium (Davies, 1984), and
there has seldom been any attempt to deal with the primary etiological
agent (smoking) in the development of lung cancers associated with
lead exposure.
8.10.1 Occupational exposure and renal cancer
There are two case reports and very limited other epidemio-
logical evidence of an association between occupational exposure to
lead and renal cancer. Confounding variables, including use of tobacco
and exposure to other carcinogens, were not addressed (Baker et al.,
1980; Lilis, 1981; Selevan et al., 1985; Cantor et al., 1986).
The age-standardized mortality ratio for cancer was low in the
"lead poisoned" and other groups of workers from the Port Pirie study
by McMichael & Johnson (1982). The authors concluded that lead
poisoning did not increase the risk of cancer in humans.
8.10.2 Conclusion
Although some cohort mortality studies have indicated an
association between lead exposure and renal disease, there is no
association between renal cancer and lead in humans.
A Working Group convened by the International Agency for Research
on Cancer (IARC) in 1987 concluded that the evidence for the
carcinogenicity of lead and inorganic lead compounds in humans was
inadequate.
8.11 Effects on thyroid function
8.11.1 Occupational groups
Robins et al. (1983) found low values for serum thyroxine and
estimated free thyroxine in 7 of 12 workers having PbB levels above
2.11 µmol/litre (44 µg/dl). Both measures were correlated with PbB
level in a cross-sectional study of 47 foundry workers. Serum
thyrotropin and triiodothyronine levels were within the normal range
for both study groups.
No such relationship was found in a study by Refowitz (1984) of a
one-in-three random sample (N = 58) of male employees at a secondary
copper smelter. No thyroid abnormalities were observed and there was
no statistically significant relationship between PbB and serum
thyroxine or an estimate of free thyroxine in serum.
Tuppurainen et al. (1988) studied 176 African male workers in
Kenya and found that the duration of lead exposure correlated
negatively with serum free thyroxine and serum total thyroxine. The
correlation was strongest for the workers with the highest exposure
intensity over time. PbB data were available from periodic medical
examinations and average values (usually five determinations) were
used. The mean PbB was 2.73 µmol/litre (56.8 µg/dl), range
1.01-6.47 µmol/litre (21-135 µg/dl), and there was a mean exposure
duration of 7.6 years (range 1-20). The authors noted that current PbB
level, as a point determination, was not associated with total or free
thyroxine, triiodothyronine or thyrotropin in serum. They proposed
that long-term, highintensity exposure might be associated with
depressed thyroid function.
Gennart et al. (1992b) included assessment of thyroid function as
part of a study of lead-exposed workers. Data for serum levels of
triiodothyronine, thyroxine, free thyroxine index and thyroid-
stimulating hormone were within normal ranges for a group of 98
workers (mean PbB level, 2.45 µmol/litre or 51 µg/dl) and 85 controls
(1.00 µmol/litre or 20.9 µg/dl). The lead exposure of this study group
was considerably lower than in the workers in Kenya, and it was
suggested that thyroid function changes might not be indicators of
moderate exposure to lead.
8.11.2 Effects in children
Siegel et al. (1989) tested 68 children for thyroid function and
for PbB and found no statistically significant relationship between
lead and total or free thyroxine.
8.12 Immune system
In a review of the effects of lead on the immune responses of
experimental animals, Koller (1985) noted that there were few human
studies.
8.12.1 Occupational exposure
There is some evidence that lead workers with PbB levels in the
range of 1.0-4.10 µmol/litre (21-85 µg/dl) have increased
susceptibility to infections (colds and influenza) and have a
significant suppression of secretory IgA levels, a major factor in
defence against respiratory and gastrointestinal infections (Ewers et
al., 1982). There are also reports of impaired mitogen responses
(reflecting T-lymphocyte function) to phytohaemagglutinin (Jaremin,
1983) and of increased numbers of suppressor T-cells (Cohen et al.,
1989). However, on the basis of a study of 39 workers exposed to lead
oxide (mean PbB level of 1.84 µmol/litre or 38.5 µg/dl) and 21 control
subjects (mean PbB level of 0.57 µmol/litre or 11.8 µg/dl), Kimber et
al. (1986) concluded that chronic lead exposure in man is not
associated with the immunological changes that have been observed in
rodent studies.
Coscia et al. (1987) found increased B-lymphocyte percentage and
absolute count in workers currently exposed to lead with PbB levels
exceeding 2.4 µmol/litre (> 50 µg/dl).
8.12.2 Children
A study of 12 pre-school children (Reigart & Graber, 1976) with
PbB levels > 1.92 µmol/litre (40 µg/dl) did not reveal altered
immunity in comparison with a control group.
8.13 Effects on bone
The pharmacokinetics of the transfer of bone lead to other target
organs and blood has been discussed by Smith & Hursh (1977), Marcus
(1985a,b), Silbergeld et al. (1988) and Rabinowitz (1991) (see section
6.2.2).
Bone homoeostasis depends upon a complex interaction of its
various components, i.e. minerals, cells and the extracellular matrix
composed of collagenous and non-collagenous proteins (Marks & Popoff,
1988; Sauk & Somerman, 1991). Early work on the effects of lead on
calcium homoeostasis and calcium-mediated function was reviewed by
Pounds (1984). More recent work has been summarized by Pounds et al.
(1991), particularly the effects of lead on hormone action,
competition with calcium binding on calcium messenger systems, and the
impaired synthesis of collagen or sialoproteins. Additional evidence
for adverse effects of lead on the proteins in the mineral
compartment, thus affecting bone homoeostasis, has been reported by
Sauk & Somerman (1991).
8.14 Biomarkers for lead effects
The understanding and application of biomarkers for the
assessment of effect is becoming more widespread (IPCS, 1994).
It is desirable to identify biomarkers of lead exposure and
effect to provide easily measurable parameters that will facilitate
the risk assessment process. At present PbB levels are frequently
measured to assess both exposure and effect. Alternative biomarkers
for lead which may be easily measured are of biochemical effects,
particularly in the haem biosynthetic pathway.
However, the relationship between these effects and neurological
impairment caused by lead has not been established. Measurement of
these effects has also been used to provide biomarkers for exposure.
Anaemia has been associated with high levels of lead exposures in
the occupational setting. However, clinical tests for haemoglobin are
rather non-specific indicators of lead toxicity (Bernard & Becker,
1988). The steps within the haem biosynthetic pathway which have been
used to measure effect are: 1) inhibition of delta-aminolaevulinic
acid dehydratase (ALAD); 2) urinary excretion of delta-aminolaevulinic
acid (ALAU); (3) the accumulation of zinc protoporphyrin (ZPP) in
erythrocytes arising from the inhibition of the enzyme ferrochelatase
or the iron transport system.
There are several methodological problems related to the
measurement of ALAD, and the benefit of measuring enzyme activity in
blood in lieu of PbB determinations is not apparent (Mushak, 1989).
Early work (Roels et al., 1976) indicated enzyme inhibition at PbB
levels of 0.24 µmol/litre (> 5 µg/dl). However, the data at levels of
0.72 µmol/litre (< 15 µg/dl) appear scattered (Hernberg & Nikkanen,
1970; Granick et al., 1973).
Levels of ALA in urine have been used as an effect indicator of
lead exposure (Meredith et al., 1978). ALAU levels were found to
correlate with PbB levels as low as 0.864 µmol/litre (18 µg/dl), the
correlation becoming much stronger at PbB levels above 1.92 µmol/litre
(40 µg/dl) (Selander & Cramer, 1970). Given the present concern over
effects in children at PbB levels well below 1.92 µmol/litre
(40 µg/dl), the assay of ALAU concentrations may not be sensitive
enough to be of any value.
The effects of lead on porphyrin levels, both coproporphyrin in
urine and zinc protoporphyrin (ZPP) in blood, have been investigated
as possible biomarkers for lead. Measurement of urinary coproporphyrin
levels does not appear to be sensitive enough to be useful in
assessing exposure to lead at present environmental levels. Excretion
levels do not rise significantly until PbB levels exceed
1.92 µmol/litre (40 µg/dl) (Meredith et al., 1978). Other studies
indicate that this assay may not be sensitive enough for biological
monitoring at PbB levels between 0.96 and 1.20 µmol/litre (20 and
25 µg/dl). At present it would appear that ZPP has limited usefulness
as a biological indicator of exposure at exposures leading to PbB
levels lower than 1.20 µmol/litre (25 µg/dl) (Roels et al., 1976;
Piomelli et al., 1982; Hammond et al., 1985; Marcus & Schwartz, 1987).
The results may also be confounded by concurrent iron deficiency which
will alter the levels of ZPP.
Vitamin D metabolism involves cytochrome P-450-dependent enzymes
and thus may be affected by lead exposure. Measurement of serum levels
of 1,25-dihydroxy-vitamin-D has been proposed as a sensitive
biological monitor of lead exposure in children (Rosen et al., 1980;
Mahaffey et al., 1982; Koo et al., 1991). However, it should be noted
that dietary intakes of calcium and phosphorus, as well as circulating
levels of parathyroid hormone will regulate the production and
circulating concentrations of this vitamin D metabolite, thus making
correlations with PbB levels difficult without adequate information on
the nutritional status of the population under study (Rosen & Chisney,
1983). In populations showing adequate nutritional status, lead at
exposures leading to PbBs levels lower than 0.96 µmol/litre (20 µg/dl)
does not appear to have a demonstrable effect on circulating levels of
1,25-dihydroxy-vitamin-D.
9. EVALUATION OF HUMAN HEALTH RISKS
9.1 Exposure assessment
Lead is a ubiquitous element detected in all environmental media.
However, natural sources contribute only a small fraction of the
amounts of lead found in air, food, water and dust. The majority of
lead in these media arises from automobile and industrial emissions
and from the use of lead-containing solder and paints. Adults and
older children receive the largest proportion of lead intake from
foods, whereas dust, soil and food all make significant contributions
to the total lead intake of young children. The major contributions to
lead in soil and outdoor dust are from the combustion of fossil fuels
(principally leaded petrol), stationary sources such as smelters, and
peeling and flaking of lead-based paint.
9.1.1 General population exposure
In the absence of specific stationary sources of lead,
concentrations in ambient air are directly related to density of
traffic and whether lead is still utilized as an additive in petrol.
Reduction or elimination of lead in petrol in those countries which
have instituted regulations has resulted in a decline by as much as
eight-fold in ambient air concentrations of lead.
Levels of lead in indoor air are affected by the presence of
cigarette smoke and dust from lead-painted surfaces. Without such
sources, air lead levels indoors are about 60% of those in outdoor
air.
For most adults, the total daily exposure to lead is via food,
water and air. For infants aged up to 5 months, formula or breast milk
and water are the main sources of lead. In children, an additional
source of exposure is dust and soils. Absorption is dependent on the
chemical form of lead, type of soil and particle size
(bioavailability). Lead intake may be augmented from unusual sources
such as folk remedies, cosmetics and hobby activity. Community
contamination and workplace practices may contribute to lead exposure.
Food (including drinking-water and beverages) is the major source
of lead exposure for the general population. Infants and children may
receive an added lead burden from soil and dust. The most significant
foodstuffs will vary from country to country. In areas still utilizing
lead-soldered cans, levels of lead are substantially higher. Depending
upon lifestyles, there may be significant oral intake of lead from
some alcoholic beverages and due to the leaching of lead from low
temperature-fired ceramic containers.
Most drinking-water supplies contain lead levels lower than
5 µg/litre when they leave the treatment plant. However, where the
water is known to be plumbo-solvent, up to 40% of the samples may
exceed 100 µg/litre in homes where lead solder, lead pipes or brass
fixtures have been used.
Absorption of lead from the lung is a function of particle size
and pulmonary deposition pattern. Small particles (< 0.5 µm in
diameter) characteristic of ambient air will be deposited deeply in
the lungs with absorption rates of 90%. Larger particles, such as
those that may be encountered in occupational settings, exhibit high
deposition rates in the upper airway. Absorption of such particles
will be a function of both dissolution in the lung and particle
clearance to the gastrointestinal tract.
Human dermal absorption of inorganic lead through unabraded skin
is of limited significance.
9.1.2 Occupational exposures
In addition to exposure to lead in ambient air, water and food,
some workers may be exposed to airborne lead and dust within the
workplace. Actual levels will vary according to the engineering design
of the process equipment and the industrial hygiene practices
utilized.
9.2 Critical issues related to exposure evaluation
In view of the heterogeneity of responses to lead within human
populations, the complex interrelationships between exposure to lead
and a biological indicator for internal dose require consideration of
several key issues in order to assess human exposures.
9.2.1 Sampling and analytical concerns
Reliable comparisons of reported levels of exposure and/or dose
can only be made where authors have described the analytical and
sampling procedures in sufficient detail to allow the reader to
assess, for example, the integrity of the sampling procedures as well
as the specificity, precision and accuracy of all analytical methods.
Problems related to sampling cord blood also need to be considered.
Most studies have utilized analytical procedures of high quality.
However, consideration must be given to blood collection procedures
(finger stick versus venepuncture) when comparing results.
9.2.2 Data presentation
Inter-study comparisons of lead exposures are complicated by the
variety of methods used to present results, including median values as
well as geometric and arithmetic means. Some authors have used
log-transformed data.
In assessing exposure from data on teeth, one must know which
tooth and which compartment(s) within the tooth were sampled. In
addition, if data are to be compared between studies, authors must
state explicitly that all teeth analysed were without caries and were
shed spontaneously.
The exposure index lifetime average blood lead (PbB) level has
assisted in the assessment of cumulative exposures from serial PbB
data. It should not be interpreted as being equivalent to a single PbB
determination at a single point in life.
For some data/analytical purposes, age-specific PbB levels may be
more appropriate than a lifetime average.
9.3 Relationship between exposure and dose
The most widely used surrogate for the absorbed dose is whole PbB
concentration.
The relationship between PbB level and lead intake is curvilinear
over a wide range of PbB values. On the basis of a single study of 17
infants, the relationship between PbB level and lead intake from food
has been determined to be 0.0077 µmol lead/litre (0.16 µg/dl) per µg
lead intake per day for a median PbB level of approximately
0.48 µmol/litre (10 µg/dl).
Most studies of the relationship between PbB level and lead
exposure apply to a single environmental source, i.e. air, food, water
or soil/dust. A summary of the relationship between PbB level and lead
intake from individual media is given in Table 23.
9.4 Surrogate measures of dose
9.4.1 Blood
Whole PbB values are widely used as a measure of absorbed dose.
However, it is believed that plasma lead concentrations may better
reflect the "active" fraction of lead in blood and define the
relationship between PbB and tissue or organ accumulation (and
Table 23. Representative relationships of blood lead median level to intake of
lead for the general populationa
Population
Median Children Adults
Airb 0.09 µmol Pb/litre per µg 0.079 µmol Pb/litre per µg
Pb/m3 (1.92 µg Pb/dl) Pb/m3c (1.64 µg Pb/dl)
Water 0.003 µmol Pb/litre per µg
Pb/litre (0.06 µg Pb/dl)
Food 0.01 µmol Pb/litre per µg 0.002-0.003 µmol Pb/litre per
Pb/day (0.16 µg Pb/dl) µg Pb/day (0.04-0.06 µg Pb/dl)
Dustb 0.09 µmol Pb/litre per 1000 µg
Pb/g (1.8 µg Pb/dl)
Soilb 0.11 µmol Pb/litre per 1000 µg
Pb/g (2.2 µg Pb/dl)
a These data are provided for illustrative purposes only recognizing that the
relationships are curvilinear in nature and are broad guidelines which will
not apply at lower or higher levels of exposure.
b A value between 0.144 to 0.24 µmol Pb/litre or 3-5 µg Pb/dl per µg/m3
is obtained when one considers indirect contribution through deposition on
soil/dust.
c The air to blood lead relationship in occupational settings is best described
by a curvilinear relationship having slopes between 0.02 and 0.08 µg/m3
air. The slope is variable but lower than that found for humans in the general
environment, which is between 1.6 and 1.9 µg/m3.
effect), although there is little experimental data (because of
analytical limitations). PbB is distributed between plasma and
erythrocytes, with less than 5% being in the plasma. Most of the lead
is bound to haemoglobin.
Venous and capillary blood levels are generally equivalent,
provided that the sampling technique is adequate.
9.4.2 Urine
Urinary measurements of lead concentration are of limited value,
although they are used occasionally as a screening test for
occupational population groups.
9.4.3 Bone
Bone lead may be measured by non-invasive X-ray fluorescence, but
this technique is limited in sensitivity at present.
9.4.4 Tooth
Shed deciduous teeth have been used to provide an index of
exposure in early childhood. Interpretation of the analytical data is
dependent on the type of tooth and the part of the tooth (whole tooth,
dentine or circumpulpar dentine) analysed.
9.4.5 Hair
Hair is not useful for measurement of lead exposure.
9.5 Biochemical effects of lead
9.5.1 Haem synthesis
Evaluation of the quality of analytical data is an important
aspect in considering reports describing effects attributed to lead.
It should be noted that much of the data presented in this area has
not been as vigorously scrutinized as, for example, psychometric study
data.
An increase in erythrocyte protoporphyrin (EP) in children occurs
between PbB levels of 0.72 and 1.2 µmol/litre (15-25 µg/dl). Increases
in EP can be detected in men when the PbB level is above
1.20-1.44 µmol/litre (25-30 µg/dl), and in women when it is above
0.96-1.44 µmol/litre (20-30 µg/dl). It should be noted that the effect
of lead on haem is confounded by low iron status.
9.5.1.1 Urinary coproporphyrin
The coproporphyrin concentration in urine increases significantly
with PbB levels in excess of 1.92 µmol/litre (40 µg/dl).
9.5.1.2 Urinary aminolaevulinic acid in children
In children 1-5 years old, there is a linear relationship with
PbB in the range 1.2-3.6 µmol/litre (25-75 µg/dl). Data for children
with PbB levels of 0.24-1.92 µmol/litre (5-40 µg/dl) show essentially
no correlation with urinary aminolaevulinic acid (ALA) excretion.
Elevation of urinary ALA level is evident at a PbB level of about
1.68 µmol/litre (35 µg/dl).
9.5.1.3 Urinary aminolaevulinic acid in adults
Urinary excretion of ALA increases in men at PbB levels above
2.16 µmol/litre (45 µg/dl) and in women above 1.68 µmol/litre
(35 µg/dl).
9.5.1.4 delta-Aminolaevulinic acid dehydratase
There was a negative exponential relationship between PbB level
and delta-aminolaevulinic acid dehydratase (ALAD) activity in a
population of 10- to 13-year-old children with PbB levels in the range
of 0.19-1.97 µmol/litre (4.7-41 µg/dl). An effect was seen at a PbB
level of approximately 0.48 µmol/litre (10 µg/dl). There is an
apparent lack of a clearly defined threshold for lead inhibition of
ALAD in different age groups.
9.5.2 Vitamin D metabolism
In the presence of adequate nutritional status, PbB levels below
0.96 µmol/litre (20 µg/dl) appear to have no demonstrable effect on
circulating concentrations of 1,25-dihydroxy-vitamin-D. A PbB level
above 0.96 µmol/litre (20 µg/dl) is associated with a decrease in the
serum level of 1,25-dihydroxy-vitamin-D.
9.5.3 Dihydrobiopterin reductase
Inhibition of dihydrobiopterin reductase has been shown in humans
where the mean PbB level is as low as 0.48 µmol/litre (10 µg/dl).
A summary of some biochemical effects of lead is presented in
Table 24.
9.5.4 Haemopoietic system
9.5.4.1 Anaemia in adults
The estimated PbB associated with a decrease in haemoglobin
concentration is 2.40 µmol/litre (50 µg/dl).
9.5.4.2 Anaemia in children
Decreased haemoglobin levels in children occur at a PbB level of
approximately 1.92 µmol/litre (40 µg/dl). Anaemia, defined as a
haematocrit below 35%, is not found at a PbB level of less than
0.92 µmol/litre (20 µg/dl). The risk of having a haematocrit value
below 35% for a 1-year-old child is 2% at a PbB level of
Table 24. Biochemical effects of lead
Parameter Blood lead level above which the
biochemical effect is demonstrable with
current techniques
µmol/litre µg/dl
Protoporphyrin levels 0.96-1.44 20-30
Coproporphyrin levels 1.92 40
ALA urine levels 1.68 35
ALAD activity 0.48 10
1,25-dihydroxy-vitamin-D 0.96 20
Dihydrobiopterin reductase 0.48 10
0.96-1.87 µmol/litre (20-39 µg/dl); the contribution of iron
deficiency may account for a substantial proportion of this 2%.
Induction of anaemia is demonstrable at 1.92 µmol/litre (40 µg/dl).
9.5.4.3 Erythrocyte pyrimidine-5'-nucleotidase
A reduction of 20% or more in erythrocyte pyrimidine
-5'-nucleotidase activity is associated with PbB concentrations above
0.48 µmol/litre (10 µg/dl).
Effects of lead are demonstrable on a number of enzyme systems
and biochemical parameters. The PbB levels, above which effects are
demonstrable with current techniques for the parameters which may have
clinical significance, are all greater than 0.96 µmol/litre
(20 µg/dl). Some clinically insignificant effects on enzymes are
demonstrable at lower levels of PbB.
9.6 Nervous system
9.6.1 Adults
9.6.1.1 Central nervous system
With acute lead exposure resulting in a PbB level in excess of
3.84 µmol/litre (80 µg/dl), severe encephalopathy and/or coma may
occur. Central nervous system (CNS) symptoms are found in lead-exposed
adults when there is a history of several years of exposure to lead at
PbB levels that may not have exceeded 3.36 µmol/litre (70 µg/dl) and a
PbB level at the time of clinical assessment of at least
1.92 µmol/litre (40 µg/dl).
Impaired neurobehavioural test performance has been found in
lead-exposed workers. Changes in critical flicker fusion test have
been detected at a PbB level of about 2.4 µmol/litre (50 µg/dl).
Sensory motor function is generally more sensitive than cognitive
end-points in many neurobehavioural evaluations, the lowest-observed-
effect level being at about 1.92 µmol/litre (40 µg/dl).
It appears also that neuroelectrophysiological tests are
sensitive indicators of the CNS effects of lead. Reductions in
latencies of sensory evoked potentials and auditory event-related
potentials have been found in workers with average PbB levels of
approximately 1.92 µmol/litre (40 µg/dl).
9.6.1.2 Peripheral nervous system
Numerous studies have measured the conduction velocity of
electrically stimulated sensory and motor nerves in workers exposed to
lead. These nerve conduction velocity (NCV) studies have yielded
somewhat mixed results, with many showing a decrease in NCV in
relation to lead exposure (indexed as PbB level) and a few showing no
effect or occasionally even an increase in NCV associated with lead
exposure. Differences in the nerves evaluated, methodologies,
characterization of lead exposure, and control of confounding
variables underlie some of the variability in results across studies.
A statistical meta-analysis of 32 NCV studies has indicated that NCV
is significantly reduced in lead-exposed workers compared to reference
subjects, but that the median motor nerve shows more reliable effects
of lead than other nerves. This collective view of the evidence is
supported by key studies that provide compelling evidence of a causal
relationship between lead exposure and reductions in NCV, extending to
PbB levels as low as 1.44 µmol/litre (30 µg/dl). These effects may be
reversible depending on the duration and level of exposure.
9.6.1.3 Autonomic nervous system
Two reports examining the electrocardiographic RR interval
variability during deep breathing and the component CV of respiratory
sinus arrythmia demonstrated dysfunctions at an average PbB level of
1.68 µmol/litre (35 µg/dl). These results suggest autonomic nervous
system dysfunction, particularly the parasympathetic nervous system.
9.6.2 Children
Prospective and cross-sectional studies of children have
demonstrated associations of lead exposure, measured by various
indices, and intellectual performance. The association has been noted
across a wide range of exposure levels and in a variety of populations
before factors other than lead have been accounted for.
A key question is whether this statistical association is
directly attributable to the causal influence of lead on child IQ. It
is important to consider alternative contributory explanations, i.e.
random chance, unexplained confounding factors, reverse causality and
selection bias.
It is a matter of debate and conjecture as to the extent to which
these four issues should inhibit claims of a causal relationship in
the epidemiological studies. The essential problem is that
observational epidemiology cannot provide definitive evidence of
causality when the key statistical association is small, the temporal
relationship is unclear and major confounders are present. Animal
studies provide qualitative support for the claim of a causal role for
lead in affecting neuropsychological performance, but provide limited
assistance in establishing quantitative dose-effect relationships.
9.6.2.1 Type of effect
The clearest and most consistent associations have been found
with global measures, such as IQ, where the largest body of evidence
is available. Efforts to delineate the neuropsychological foundations
of this association with a wide variety of tests of specific
neuropsychological domains have not so far been successful.
9.6.2.2 Magnitude
Based on the evidence from cross-sectional and prospective
studies of populations with PbB levels generally below 1.2 µmol/litre
(25 µg/dl), the size of the apparent IQ effect (at ages 4 and above)
is a deficit of between 0 and 5 points (on a scale of 100 with a
standard deviation of 15) for each 0.48 µmol/litre (10 µg/dl)
increment in PbB level, with a likely apparent effect size of between
1 and 3 points. At PbB levels above 1.2 µmol/litre (25 µg/dl), the
relationship between PbB level and IQ may differ. Estimates of effect
size are group averages and only apply to the individual child in a
probabilistic manner.
Existing epidemiological studies do not provide definitive
evidence of a threshold. Below the PbB range of 0.48-0.72 µmol/litre
(10-15 µg/dl), the effect of confounding variables and limits in the
precision of analytical and psychometric measurements increases the
uncertainty attached to any estimate of effect. However, there is some
evidence of an association below this range.
9.6.2.3 Reversibility/persistence
Whilst the IPCS Task Group could not unequivocally state that
effects of early childhood exposure are persistent beyond childhood,
because the current data are too meagre, it was held that
neurobehavioural effects detected at age seven or later usually
persist. Measures in later childhood tend to be more predictive of
subsequent performance than those made earlier. It is more likely than
not that effects seen during school years are to some degree
irreversible. This has also been observed in later follow-up studies
conducted in other non-lead topics of child development research. One
of the difficulties is that there are too few studies concerning
long-term outcome in children with high early exposures and where the
sources of exposure are subsequently removed.
Virtually no useful data are available on the effects on IQ of
removing children from a "high" exposure environment to one of "low"
exposure or on reduction of body lead burden in children. This is not
to say that exposure should not be reduced when possible.
9.6.2.4 Age-specific sensitivity
From prospective studies it is not possible to determine an age
of critical sensitivity. This reflects the findings that serial PbB
measures taken at the age of 2 years and later are positively
correlated with, the individual rankings remaining approximately
constant, and this limits the ability to identify sensitive periods of
exposure.
9.6.2.5 Interactions/subgroups
The evidence is inconclusive on whether apparent effects are more
or less marked in different gender or socioeconomic status (SES)
subgroups. However, where there are suggestions of SES-related
differences, the apparent effects tend to be more marked in the lower
SES subgroups.
9.6.3 Animal studies
Experimental animal studies of CNS effects provide support for
the associations between PbB levels and neurobehavioural deficits
described in human epidemiological studies of lead. There is
supportive evidence both in terms of demonstrating causal
relationships and in the levels of PbB at which such effects are
observed, namely 0.528-0.72 µmol/litre (11-15 µg/dl). Moreover, they
provide qualitative parallels in the nature of the effects described,
as these effects include changes in learning and memory functions.
Experimental animal studies indicate that these CNS effects may depend
upon task complexity and can persist long beyond the termination of
lead exposure. These studies also provide information possibly
relevant to understanding mechanisms of effect. In addition, the
experimental animal studies provide such evidence in the absence of
the confounding factors and co-variates, such as parental IQ,
socioeconomic status, and quality of the home environment, that are
problematic to human epidemiological endeavours, and in the absence of
nutritional deficiencies that may arise in human populations.
9.7 Renal system
Renal function impairment was not associated with a PbB level
below 3.0 µmol/litre (62 µg/dl) when measured by blood urea nitrogen
and serum creatinine levels in lead workers. However, renal tubular
effects were detected in workers with a PbB level below 3.0 µmol/litre
when measured by more sensitive indicators such as NAG.
Most studies of the general population attempting to relate renal
function impairment to PbB concentration have not demonstrated an
effect with PbB levels below 1.8 µmol/litre (37.3 µg/dl). More
sensitive indicators of renal function may indicate a renal effect of
lead below this level.
9.8 Liver
Over-exposure to lead may inhibit drug metabolism in the liver.
9.9 Reproduction
9.9.1 Female
Studies on the risk of spontaneous abortion and reduced birth
weight associated with maternal PbB levels below 1.44 µmol/litre
(30 µg/dl) have yielded mixed results. Recent epidemiological studies
have shown exposure-related perturbations in the length of gestation,
significantly greater risks being associated with PbB levels of
0.72 µmol/litre (15 µg/dl) or more.
9.9.2 Male
PbB concentrations above 1.92 µmol/litre (40 µg/dl) have been
shown to affect sperm morphology and function. At present, the
reproductive consequences of these changes are unknown.
9.10 Blood pressure
A quantitative assessment of the collective evidence from all the
observational studies in adults is made difficult by the fact that
studies have adopted different policies regarding adjustment for
potential confounding factors (e.g., alcohol consumption). In
addition, quantitative findings from the two largest studies (BRHS and
NHANES II) have depended on whether adjustment was made for
geographical variations in blood pressure and blood lead.
The limited size of most observational studies has inevitably
meant that they could not consistently demonstrate a statistically
significant relationship. However, an overview of all the studies
shows that evidence is consistent with the centre-adjusted analysis of
the two main studies, i.e. there are very weak but statistically
significant associations between PbB level and both systolic and
diastolic blood pressures. The likely order of magnitude is that for
any two-fold increase in PbB level (e.g., from 0.8 to 1.6 µmol per
litre) there is a mean 1 mmHg increase in systolic blood pressure. The
association with diastolic blood pressure is of a similar magnitude.
Animal studies have provided plausible mechanisms for an effect
of lead on blood pressure. However, from such a small magnitude of
statistical associations in the presence of important confounders, one
cannot infer that low-level lead exposure is causally related to an
increase in blood pressure.
The two population studies relating PbB to cardiovascular disease
events show no statistically significant associations. Hence, there is
no clear evidence to suggest that lead has an impact of public health
importance as regards hypertension or risk of cardiovascular disease.
9.11 Carcinogenicity
Renal tumours occur in rats and mice administered high doses of
lead. However, the evidence for the carcinogenicity of lead and
inorganic lead compounds in humans is inadequate.
9.12 Immune system
There is no strong evidence in humans of an effect of lead on the
immune system.
10. RECOMMENDATIONS FOR PROTECTION OF HUMAN HEALTH
10.1 Public health measures
Public health measures should be directed towards reduction and
prevention of exposure to lead by reducing the use of lead and lead
compounds and by minimizing lead-containing emissions that result in
human exposures. This can be achieved by:
a) phasing-out any remaining uses of lead additives in motor fuels;
b) further reducing the use of lead-based paints, with the objective
of eliminating such paints;
c) development and application of methods for the safe and
economical remediation of lead-painted homes and lead
contaminated soil;
d) elimination of the use of lead in food containers (e.g., in the
seams of cans);
e) dissemination of information to assist with identification of
glazed food containers which may leach lead into food placed,
cooked or stored in the container;
f) eliminating any remaining agricultural uses of lead or lead
compounds (e.g., lead arsenate as an insecticide);
g) identifying and reducing, or preferably, eliminating lead found
as a contaminant or ingredient of folk remedies and cosmetics;
h) the use of materials and engineering practices to minimize
plumbosolvency in water treatment and water distribution systems;
i) systematic examination of processes in which lead is used or
recycled in order to identify and reduce lead exposure by means
of improved engineering design, of operators, by-standers and the
environment. Opportunities for technology transfer should be used
whenever possible.
10.2 Public health programmes
Programmes should be developed:
a) to enhance data collection and to make available to the public
information on the lead content of foodstuffs;
b) to facilitate identification of populations at high risk of
exposure to lead on the basis of monitoring data for lead in
food, air, water and soil;
c) that incorporate improved procedures for health risk assessment
of population groups at risk of exposure to lead;
d) that promote understanding and awareness concerning the effects
on human health associated with exposure to lead, while
recognizing cultural sensitivities;
e) that place emphasis on adequate nutrition, health care and
attention to socioeconomic conditions which may exacerbate the
effects of lead present in the environment.
10.3 Screening, monitoring and assessment procedures
Methods for evaluating the effects and associated risks of
exposure to lead require both improvement and further development or
research. In the short term the following measures are needed.
a) Screening
i) Blood lead measurements should be recognized as the biomarker of
choice for screening for previous exposure of children to lead.
ii) The sensitivity of the developing nervous system to the
potentially harmful effects of lead is such that other
biochemical measurements (e.g., erythrocyte protoporphyrin) are
not sufficiently sensitive for assessment of infants and
children.
b) Monitoring
i) More sensitive analytical methods should be developed for the
reliable measurement of blood lead levels below 0.72 µmol/litre
(15 µg/dl) to acceptable standards of precision and accuracy.
ii) There is a need for international analytical quality assurance
programmes utilizing reference lead-containing materials.
iii) All publications containing blood lead measurement data should
provide adequate data on current quality assurance and quality
control.
iv) Data comparisons are made more difficult by differences in units
and statistical techniques for data handling. Investigators
should be encouraged to adopt internationally agreed practices
(e.g., IUPAC units).
c) Assessment
i) Validated biomarkers are needed to define the relationship
between measures of environmental (external) exposure and
specific effects (biochemical or functional).
ii) Biomarkers indicative of deficits in cognitive performance are
needed to facilitate assessment of risk.
iii) Improved methods are needed for the definition of outcome effects
(especially IQ and neurobehavioural deficits) attributable to
lead at blood lead concentrations of about
0.48 µmol/litre (10 µg/dl) or less.
iv) Further data are required to determine whether outcome effects on
the nervous system attributed to lead are reversible or
permanent.
v) Biomarkers for the renal effects of lead are needed to link renal
damage with lead exposure and thereby improve assessment of risk
of renal damage.
11. FURTHER RESEARCH
a) Research is required to improve the process for the assessment of
risk associated with exposure to lead. Specifically this should
cover:
i) definition of the health significance of biochemical
changes associated with exposure to lead, with particular
attention to alterations associated with blood lead
concentrations of about 0.72 µmol/litre (15 µg/dl) or
less;
ii) work to define the bioavailability of lead from different
sources and to establish the relationship between exposure
(sources and speciation) and body burden;
iii) definition of the influence of host-related factors
(particularly nutrition) affecting absorption and
distribution of lead;
iv) intensification of kinetic studies of lead to provide an
improved data base for extrapolation between species;
v) elucidation of mechanisms of accumulation and mobilization
of lead from bone with particular attention to the
influence of pregnancy and ageing on kinetics;
vi) investigation of the pharmacokinetics of lead in the
pregnant female in relation to transfer of lead to the
developing embryo and fetus and factors that mitigate such
transfer;
vii) determination of the effects of pre- and post-natal
exposure to lead;
viii) improved definition of paternally mediated effects of lead
exposure on the reproductive process and outcomes.
b) More general research needs include the following:
i) rationalization of neurobehavioural tests used to assess
performance in children and animals in order to permit
comparisons of measurements and test data that use similar
biological mechanisms;
ii) follow-up research (where possible) of historical cohorts
of lead-poisoned adults to examine the sequelae;
iii) evaluation of intervention measures, such as lead
abatement from home and soil, and chelation on reduction
of blood lead levels and outcome effects on the central
nervous system.
12. PREVIOUS EVALUATIONS BY INTERNATIONAL BODIES
The carcinogenic potential of lead and lead compounds was last
evaluated by the International Agency for Research on Cancer in 1987
(IARC, 1987b). There was inadequate evidence for the carcinogenicity
of lead and inorganic lead compounds in humans, but sufficient
evidence was available to show that specified inorganic lead compounds
were carcinogenic in experimental animals. The overall evaluation
placed lead and inorganic lead compounds in Group 2B, i.e. possibly
carcinogenic to humans.
To protect workers from the adverse effects of lead on haem
synthesis and on the peripheral and central nervous system, a
health-based biological exposure limit of 1.92 µmol/litre (40 µg/dl)
was recommended by a WHO Study Group (WHO, 1980). It was further
recommended that blood lead (PbB) levels in women within the
reproductive age range should not exceed 1.44 µmol/litre (30 µg/dl).
Depending upon the background levels of PbB in the worker population,
air lead levels should not exceed 30-60 µg/m3 (WHO, 1980).
A drinking-water guideline value of 0.050 mg/litre was developed
in 1984 (WHO, 1984). This guideline value has been revised recently to
0.01 mg/litre (WHO, 1993).
Lead was evaluated by a WHO Working Group developing air quality
guidelines for Europe (WHO, 1987). Based on the assumption that PbB
levels in 98% of the population would be maintained at levels below
0.96 µmol/litre (20 µg/dl), a guideline value in the range of
0.5-1.0 µg/m3 (long-term average, such as annual mean) was
recommended.
At the 41st meeting of the Joint FAO/WHO Expert Committee on Food
Additives and Food Contaminants, a Provisional Tolerable Weekly Intake
(PTWI) of 25 µg/kg body weight was recommended (FAO/WHO, 1993). This
level refers to lead from all sources and was set to protect all
humans, including infants and children. It was based on a model
indicating daily intakes of lead between 3-4 µg/kg body weight by
infants and children and is not associated with an increase in PbB
concentrations.
Regulatory standards established by national bodies in several
countries and the European Economic Community are summarized in the
legal file of the International Register of Potentially Toxic
Chemicals (IRPTC, 1987).
REFERENCES
Aguilera de Benzo Z, Fraile R, & Carrion N (1989) Determination of
lead in whole blood by electrothermal atomization atomic absorption
spectrometry using tube and platform atomizers and dilution with Trito
X-100. J Anal At Spectrom, 4: 397-400.
Albahary C (1972) Lead and hemopoiesis: The mechanism and consequences
of the erythropathy of occupational lead poisoning. Am J Med,
52: 367-378.
Albert LA & Badillo F (1991) Environmental lead in Mexico. Rev Environ
Contamin Toxicol, 117: 1-49.
Alexander FW, Delves HT, & Clayton BE (1973) The uptake and excretion
by children of lead and other contaminants. In: Barth D, Berlin A,
Engel R, Recht P, & Smeets J ed. Environmental health aspects of lead:
Proceedings, International Symposium, Amsterdam, The Netherlands,
October 1972. Luxembourg, Commission of the European Communities,
pp 319-331.
Al-Hakkak ZS, Hamamy HA, Murad AMB, & Hussain AF (1986) Chromosome
aberrations in workers at a storage battery plant in Iraq. Mutat Res,
171: 53-60.
Altmann L, Weinsberg F, Sveinsson K, Lilienthal H, Wiegand H, &
Winneke G (1993) Impairment of long-term potentiation and learning
following chronic lead exposure. Toxicol Lett, 66(1): 105-112.
Alvares AP, Kapelner S, Sassa S, & Kappas A (1975) Drug metabolism in
normal children, lead-poisoned children, and normal adults. Clin
Pharmacol Ther, 17: 179-183.
Anderson C & Danylchuk KD (1977) The effects of chronic low level lead
intoxication on the Haversian remodeling system in dogs. Lab Invest,
37: 466-469.
Angle CR & McIntire MS (1978) Low level lead inhibition of erythrocyte
pyrimidine nucleotidase. Environ Res, 17: 296-302.
Angle CR & McIntire MS (1979) Environmental lead and children: the
Omaha study. J Toxicol Environ Health, 5: 855-870.
Angle CR, McIntire MS, & Swanson MS (1982) Erythrocyte nucleotides in
children - increased blood lead and cytidine triphosphate. Pediatr
Res, 16: 331-334.
Annest JL & Mahaffey KR (1984) Blood lead levels for persons aged 6
months-74 years: United States, 1976-1980. Hyattsville, Maryland, US
Public Health Service (Report No. 223, Series 11) (DHHS Publication
No. (PHS) 84-1683).
Annest JL, Pirkle JL, Makuc D, Neese JW, Bayse DD, & Kovar MG (1983)
Chronological trend in blood lead levels between 1976 and 1980. N Engl
J Med, 308: 1373-1377.
Araki S & Ushio K (1982) Assessment of the body burden of chelatable
lead: a model and its application to lead workers. Br J Ind Med,
39: 157-160.
Araki S, Honma T, Yanagihara S, & Ushio K (1980) Recovery of slowed
nerve conduction velocity in lead-exposed workers. Int Arch Occup
Environ Health, 46: 151-157.
Araki S, Aono H, Fukahori M, & Tabuki K (1984) Behaviour of lead and
zinc in plasma, erythrocytes, and urine and ALAD in erythrocytes
following intravenous infusion of CaEDTA in lead workers. Arch Environ
Health, 39: 363-367.
Araki S, Murata K & Aono H (1986a) Subclinical cervico-spino-bulbar
effects of lead: A study of short-latency somatosensory evoked
potentials in workers exposed to lead, zinc and copper. Am J Ind Med,
10: 163-175.
Araki S, Yokoyama K, Murata K, & Aono H (1986b) Determination of the
distribution of conduction velocities in workers exposed to lead, zinc
and copper. Br J Ind Med, 43: 321-326.
Araki S, Aono H, Murata K (1986c) Mobilisation of heavy metals into
the urine by CaEDTA: Relation to erythrocyte and plasma concentrations
and exposure indicators. Br J Ind Med, 43: 636-641.
Araki S, Murata K, & Aono H (1987) Central and peripheral nervous
system dysfunction in workers exposed to lead, zinc and copper: A
follow-up study of visual and somatosensory evoked potentials. Int
Arch Occup Environ Health, 59: 177-187.
Araki S, Murata K, Yokoyama K, & Uchida E (1992) Auditory
event-related potential (P300) in relation to peripheral nerve
conduction in workers exposed to lead, zinc, and copper: Effects of
lead on cognitive function and central nervous system. Am J Ind Med,
21: 539-547.
Arnetz BB & Nicolich MJ (1990) Modelling of environmental lead
contributors to blood lead in humans. Int Arch Occup Environ Health,
62: 397-402.
Aslam M, David SS, & Healy MA (1979) Heavy metals in some Asian
medicines and cosmetics. Publ Health (Lond), 93: 274-284.
Assenato G, Paci C, Baser M, Molinini R, Candela RG, Altamura BM, &
Giorgino R (1987) Sperm count suppression without endocrine
dysfunction in lead-exposed men. Arch Environ Health, 42: 124-127.
ATSDR (1993) Toxicological profile for lead. Atlanta, Georgia, US
Department of Health and Human Services, Agency for Toxic Substances
and Disease Registry (TP-92/12).
Aungst BJ, Docle JA, & Fung H (1981) The effect of dose on the
disposition of lead in rats after intravenous and oral administration.
Toxicol Appl Pharmacol, 61: 48-57.
Awad el Karim MA, Hamed AS, Elhaimi YAA, & Osman Y (1986) Effects of
exposure to lead among lead acid battery factory workers in Sudan.
Arch Environ Health, 41: 261-265.
Azar A, Trochimowicz HJ, & Maxfield ME (1973) Review of lead studies
carried out at Haskell Laboratory: two year feeding study and response
to hemorrhage study. In: Environmental health aspects of lead.
Proceedings of an international symposium, Amsterdam. Luxembourg,
Commission of the European Communities, pp 199-210.
Baghurst PA, McMichael AJ, Wigg N, Vimpani G, Robertson EF, Roberts
RJ, & Tong S-L (1992) Life-long exposure to environmental lead and
children's intelligence at age seven: the Port Pirie cohort study.
N Engl J Med, 327: 1269-1284.
Baker EL, Landrigan PJ, Barbour AG, Cox DH, Folland DS, Ligo RN, &
Throckmorton J (1979) Occupational lead poisoning in the United
States: clinical and biochemical findings related to blood lead
levels. Br J Ind Med, 36: 314-322.
Baker EL, Goyer RA, Fowler BA, Khettry U, Bernard DB, Adler S, White
R, Babayan R, & Feldman RG (1980) Occupational lead exposure,
nephropathy and renal cancer. Am J Ind Med, 1: 139-148.
Barrio A & Badia J (1985) [Lead encephalopathy in adults.] Rev Med
Chil, 113(6): 564-567 (in Spanish).
Barltrop D (1969) Mineral metabolism in pediatrics. Oxford, London,
Edinburgh, Melbourne, Blackwell Scientific Publishers.
Barltrop D (1972) Children and environmental lead. In: Hepple P ed.
Lead in the environment: Proceedings of a conference. London,
Institute of Petroleum, pp 52-60.
Barry PSI (1975) A comparison of concentrations of lead in human
tissues. Br J Ind Med, 32: 119-139.
Barry PSI (1981) Concentrations of lead in the tissues of children.
Br J Ind Med, 38: 61-71.
Barton JC, Conrad ME, Nuby S, & Harrison L (1978) Effects of iron on
the absorption and retention of lead. J Lab Clin Med, 92: 536-547.
Bauchinger M, Dresp J, Schmid E, Englert N, & Krause C (1977)
Chromosome analyses of children after ecological lead exposure. Mutat
Res, 56: 75-79.
Beattie AD, Moore MR, Goldberg A, Finlayson MJ, Graham JR, Mackie TM,
Main J, McLaren PA, Murdock KM, & Stewart FT (1975) Role of chronic
lead exposure in the aetiology of mental retardation. Lancet,
1: 589-592.
Beckett PH, Davis RD, & Brindley P (1979) The disposal of sewage
sludge onto farmland: the scope of the problem of toxic elements.
Water Pollut Control, 78: 419-436.
Behari LR, Singh S, Tandon SK, & Wahal AK (1982) Lead poisoning among
Indian silver jewellery makers. Ann Occup Hyg, 27: 107-109.
Behne D (1980) Problems of sampling and sample preparation for trace
element analysis in the health sciences. In: Bratter P & Schramel P
ed. Trace element analytical chemistry in medicine and biology.
Proceedings of the First International Workshop, Neuherberg, Federal
Republic of Germany. Berlin, New York, Walter de Gruyter & Co.,
pp 769-782.
Beijer K & Jernelöv A (1984) Microbial methylation of lead. In:
Grandjean P ed. Biological effects of organolead compounds. Boca
Raton, Florida, CRC Press, pp 13-19.
Bellinger DC & Stiles KM (1993) Epidemiologic approaches to assessing
the developmental toxicity of lead. Neurotoxicology, 14: 151-160.
Bellinger DC, Needleman HL, Leviton A, Waternaux C, Rabinowitz MR, &
Nichols ML (1984) Early sensory-motor development and prenatal
exposure to lead. Neurobehav Toxicol Teratol, 6: 387-402.
Bellinger DC, Leviton A, Waternaux C, Needleman H, & Rabinowitz M
(1987) Longitudinal analyses or prenatal and post-natal lead exposure
and early cognitive development. N Engl J Med, 316: 1037-1043.
Bellinger D, Sloman A, Leviton A, Rabinowitz M, Needleman H, &
Waternaux C (1991a) Low-level lead exposure and children's cognitive
function in the preschool years. Pediatrics, 87: 219-227.
Bellinger D, Leviton A, Rabinowitz M, Allred E, Needleman H,
Schoenbaum S (1991b) Weight gain and maturity in fetuses exposed to
low levels of lead. Environ Res, 54: 151-158.
Bellinger DC, Stiles KM, & Needleman HL (1992) Low-level lead
exposure, intelligence and academic achievement: A long-term follow-up
study. Pediatrics, 90: 855-861.
Berlin A & Schaller KH (1974) European standardized method for the
determination of delta-aminolevulinic acid dehydratase activity in
blood. Z Klin Chem Klin Biochem, 12: 389-390.
Bernard SR (1977) Dosimetric data and metabolic model for lead. Health
Phys, 32: 44-46.
Bernard BP & Becker CE (1988) Environmental lead exposure and the
kidney. Clin Toxicol, 26: 1-34.
Bernstein J, Evan AP, & Gardner KD (1987) Epithelial hyperplasia in
human polycystic kidney disease. Its role in pathogenesis and risk of
neoplasia. Am J Pathol, 129: 92-101.
Bert JL, van Dusen LJ, & Grace JR (1989) A generalized model for the
prediction of body lead burdens. Environ Med, 48: 117-127.
Beyer WN & Cromartie EJ (1987) A survey of Pb, Cu, Zn, Cd, Cr, As, and
Se in earthworms and soil from diverse sites. Environ Monit Assess,
8: 27-36.
Biggins PDE & Harrison RM (1979) Atmospheric chemistry of automotive
lead. Environ Sci Technol, 13: 558-565.
Billick IJ & Gray VE (1978) Lead based paint poisoning research:
review and evaluation 1971-1977. Washington, DC, US Department of
Housing and Urban Development.
Bishop L & Hill WJ (1983) A study of the relationship between blood
lead levels and occupational air lead levels. Am Stat, 37(4): 471-475.
Blair JA, Hilburn ME, Leeming RJ, McIntosh MJ, & Moore MR (1982) Lead
and tetrahydrobiopterin metabolism: possible effects on IQ. Lancet,
April 24, 964.
Body PE, Inglis GR, & Mulcahy DE (1988) Lead contamination in Port
Pirie South Australia. Adelaide, Australia, South Australia Department
of Environment and Planning (SADEP Report No. 101).
Bolger PM, Carrington CD, Capar SG, & Adams MA (1991) Reductions in
dietary lead exposure in the United States. Chem. Speciation
Bioavailab, 3(314): 31.
Bolter E, Butz T, & Arseneau JF (1975) Mobilization of heavy metals by
organic acids in the soils of a lead mining and smelting district.
In: Proceedings of the IXth Annual Conference on Trace Substances in
Environmental Health, Columbia, Missouri, 10-12 June 1975, pp 107-112.
Bondarenko GP (1968) An experimental study of the solubility of galena
in the presence of fulvic acids. Geochem Int, 5: 525-531.
Bornschein RL (1985) Influence of social factors on lead exposure and
child development. Environ Health Perspect, 62: 343-351.
Bornschein RL & Rabinowitz MB (1985) Proceedings of the Second
International Conference on Prospective Lead Studies. Environ Res,
38: 1-210.
Bornschein RL, Succop P, Dietrich KN, Clark CS, Que Hee S, & Hammond
PB (1985) The influence of social and environmental factors on dust
lead, hand lead, and blood lead levels in young children. Environ Res,
38: 108-118.
Bornschein RL, Succop PA, Krafft KM, Clark CS, Peace B, & Hammond PB
(1986) Exterior surface dust lead, interior house dust lead and
childhood lead exposure in an urban environment. In: Hemphill DD ed.
Trace substances in environmental health II. Columbia, Missouri,
University of Missouri, pp 322-332.
Bornschein RL, Succop PA, Krafft, KM, Clark CS, Peace B, & Hammond PB
(1987) Exterior surface dust lead, interior house dust lead and
childhood lead exposure in an urban environment. Trace Subst Environ
Health, 20: 322-332.
Bornschein RL, Grote J, Mitchell T, Succop PA, Dietrich KN, Krafft KM,
& Hammond PB (1989) Effects of prenatal lead exposure on infant size
at birth. In: Smith MA, Grant LD, & Sors AI ed. Lead exposure and
child development: An international assessment. Dordrecht, London,
Kluwer Academic Publishers, pp 307-319.
Boscolo P & Carmignani M (1988) Neurohumoral blood pressure regulation
in lead exposure. Environ Health Perspect, 78: 101-106.
Boutron CF, Görlach U, Candelone J-P, Bolshov MA, & Delmas RJ (1991)
Decrease in anthropogenic lead, cadmium and zinc in Greenland snows
since the late 1960s. Nature (Lond), 353: 153-156.
Braunstein GD, Dahlgren J, & Loriaux DL (1987) Hypogonadism in
chronically lead-poisoned men. Infertility, 1: 33-51.
Brown KW & Black SC (1983) Quality assurance and quality control data
validation procedures used for the Love Canal and Dallas lead soil
monitoring programs. Environ Monit Assess, 3: 113-122.
Brunekreef B (1984) The relationship between air lead and blood lead
in children: A critical review. Sci Total Environ, 38: 79-123.
Brunekreef B (1986) Childhood exposure to environmental lead. London,
United Kingdom, Monitoring and Assessment Research Centre.
Buchet J-P, Lauwerys R, Roels H, & Hubermont G (1977) Mobilization of
lead during pregnancy in rats. Int Arch Occup Environ Health,
40: 33-36.
Buchet J-P, Roels H, Bernard A, & Lauwerys R (1980) Assessment of
renal function of workers exposed to inorganic lead, cadmium, or
mercury vapour. J Occup Med, 22: 741-750.
Buchet JP, Lauwerys R, Vandervoorde A, & Pycke JM (1983) Oral daily
intake of cadmium, lead, manganese, copper, chromium, mercury,
calcium, zinc, and arsenic in Belgium: a duplicate meal study. Food
Chem Toxicol, 21: 19-24.
Bull RJ (1980) Lead and energy metabolism. In: Singhal PL & Thomas JA
ed. Lead toxicity. Baltimore, Maryland, Urban Schwarzenberg, Inc.,
pp 119-168.
Bushnell PJ & Bowman RE (1979a) Reversal learning deficits in young
monkeys exposed to lead. Pharmacol Biochem Behav, 10: 733-742.
Bushnell PJ & Bowman RE (1979b) Persistence of impaired reversal
learning in young monkeys exposed to low levels of dietary lead.
J Toxicol Environ Health, 5: 1015-1023.
Bushnell PJ & DeLuca H (1983) The effects of lactose on the absorption
and retention of dietary lead. J Nutr, 113: 365-378.
Bushnell PJ & Levin ED (1983) Effects of zinc deficiency on lead
toxicity in rats. Neurobehav Toxicol Teratology, 5: 283-288.
Bushnell G, Brockhaus A, & Baltissen R (1977a) Neurobehavioural and
system effects of long-term blood lead elevation in rats. Arch
Toxicol, 37: 247-263.
Bushnell PJ, Allan JR, & Marlar RJ (1977b) Scotopic vision deficits in
young monkeys exposed to lead. Science, 196: 333-335.
Byers RK & Lord EE (1943) Late effects of lead poisoning on mental
development. Am J Dis Child, 66: 471-494.
Calder IC, Collings MT, & Heyworth JS (1990) Evaluation of soil lead:
blood lead relationship for Port Pirie. Proceedings of the 23rd Annual
Conference of Trace Substances in Environmental Health. Environ
Geochem Health, 12(Suppl): 81-91.
Campbell BC, Beattie AD, Moore MR, Goldberg A, & Reid AG (1977) Renal
insufficiency associated with excessive lead exposure. Br Med J,
1: 482-485.
Cantor KP, Sontag JM, & Held MF (1986) Patterns of mortality among
plumbers and pipefitters. Am J Ind Med, 10: 73-89.
Capar SG & Rigsby EJ (1989) Survey of lead in canned evaporated milk.
J Assoc Off Anal Chem, 72: 378-381.
Cardenas A, Roels H, Bernard AM, Barbon R, Buchet JP, Lauwerys RR,
Rosello J, Ramis I, Mutti A, Franchini I, Fels LM, Stolte H, de Broe
ME, Nuyts GD, Taylor SA, & Price RG (1993) Markers of early renal
changes induced by industrial pollutants. II. Application to workers
exposed to lead. Br J Ind Med, 50: 28-36.
CDC (1985) Preventing lead poisoning in young children. Atlanta,
Georgia, US Department of Health and Human Services, Public Health
Service, Centers for Disease Control, pp 7-19 (Publication
No. 99-2230).
CEC (1981) Directive 77/312/CEE du Conseil du 29 mars 1977 concernant
la surveillance biologique de la population vis-à-vis du risque
saturnin. Rapport sur la mise en oeuvre de la directive. Luxembourg,
Commission des Communautés européennes, Direction Santé et Sécurité
(V/E/1/81-17/2).
Cerklewski FL & Forbes RM (1976) Influence of dietary zinc on lead
toxicity in the rat. J Nutr, 110: 689-696.
Chai S & Webb RC (1988) Effects of lead on vascular reactivity.
Environ Health Perspect, 78: 85-89.
Chakravorti S & Bhar S (1978) Chronic occupational exposure to lead:
An evaluation of environmental lead levels, lead absorption and
intoxication of storage battery workers. Bombay, Central Labour
Institute, Industrial Hygiene Division.
Chamberlain AC (1983) Effect of airborne lead on blood lead. Atmos
Environ, 17: 677-692.
Chamberlain AC (1985) Prediction of response of blood lead to airborne
and dietary lead from volunteer experiments with lead isotopes. Proc R
Soc (London), B224: 149-182.
Chamberlain AC, Heard MJ, Little P, Newton D, Wells AC, Wiffen RD
(1978) Investigations into lead from motor vehicles. Harwell, United
Kingdom Atomic Energy Authority (Report No. AERE-R9198).
Chan WH, Tang AJS, Chung DHS, & Lusis MA (1986) Concentration and
deposition of trace metals in Ontario - 1982. Water Air Soil Pollut,
29(4): 373-389.
Charney E, Kessler B, Farfel M, & Jackson D (1983) Childhood lead
poisoning: a controlled trial for the effect of dust-control measures
on blood lead levels. N Engl J Med, 309: 1089-1093.
Chau YK, Wong PTS, & Bengert GA (1979) Determination of tetraalkyl-
lead compounds in water, sediments, and fish samples. Anal Chem,
51: 186-188.
Chau YK, Wong PTS, & Kramer O (1980) Occurrence of tetraalkylead
compounds in the aquatic environment. Bull Environ Contam Toxicol,
24: 265-269.
Chettle DR, Scott MC, & Somervaille LJ (1991) Lead in bone: sampling
and quantitation using K x-rays excited by 109Cd. Environ Health
Perspect, 91: 49-55.
Chisolm JJ Jr (1962) Aminoaciduria as a manifestation of renal tubular
injury in lead intoxication and a comparison with patterns of
aminoaciduria seen in other diseases. J Pediatr, 60: 1-17.
Chisolm JJ & Brown DH (1979) Micromethod for zinc protoporphyrin in
erythrocytes: including new data on the absorptivity of zinc
protoporphyrin and new observations in neonates and sickle cell
disease. Biochem Med, 22: 214-237.
Chisolm JJ, Thomas DJ, & Hamill TG (1985) Erythrocyte porphobilinogen
synthase activity as an indicator of lead exposure to children. Clin
Chem, 31: 601-605.
Choie DD & Richter GW (1972) Cell proliferation in rat kidney induced
by lead acetate and effects of uninephrectomy on cell proliferation.
Am J Pathol, 66: 265-275.
Choie DD & Richter GW (1974) Cell proliferation in mouse kidney
induced by lead. Synthesis of deoxyribonucleic acid. Lab Invest,
30: 647-651.
Chowdhury AR, Dewan A, & Gandhi DN (1984) Toxic effect of lead on the
testes of rat. Biomed Biochem Acta, 43: 95-100.
Chowdhury AR, Chinoy NJ, & Gautam AK (1986) Effect of lead on human
semen. Adv Contracept Delivery Syst, 2: 208-211.
Christoffersson JO, Schutz A, Skerfving S, Ahlgren L, & Mattson S
(1986) Decrease of skeletal lead after end of occupational exposure.
Arch Environ Health, 41: 312-318.
Clark CS, Bornschein RL, Succop P, Que Hee SS, Hammond PB, & Peace B
(1985) Condition and type of housing as an indicator of potential
environmental lead exposure and pediatic blood lead levels. Environ
Res, 38: 46-53.
Clausing P, Brunekreef B, & van Wijnen JH (1987) A method for
estimating soil ingestion by children. Int Arch Occup Environ Health,
59: 73-82.
Cohen N, Modai D, Golik A, Weissgarten J, Peller S, Katz A, Averbukh
Z, & Shaked U (1989) Increased concanavalin A-induced suppressor cell
activity in humans with occupational lead exposure. Environ Res,
48: 1-6.
Cohn J, Cox C, & Cory-Slechta DA (1993) The effects of lead exposure
on learning in a multiple repeated acquisition and performance
schedule. Neurotoxicology, 14: 329-346.
Collins MF, Hrdina PD, & Whittle E (1992) Lead in blood and brain
regions of rats chronically exposed to low doses of the metal. Toxicol
Appl Pharmacol, 65: 314-322.
Colombo A (1985) The underdefined nature of the blood lead-air lead
relationship from biological and statistical grounds. Atmos Environ,
19: 1485-1493.
Columbano A, Ledda-Columbano GM, Lee G, Rajalakshmi S, & Sarma DSR
(1984) Inability of mitogen-induced liver hyperplasia to support
induction of enzyme-altered islands induced by liver carcinogens.
Cancer Res, 47: 5557-5559.
Cookman GR, King W, & Regan CM (1987) Chronic low-level lead exposure
impairs embryonic to adult conversion of the neural cell adhesion
molecule. J Neurochem, 49(2): 399-403.
Cookman GR, Hemmens SE, Keane GJ, King WB, & Regan CM (1988) Chronic
low level lead exposure precociously induces rat glial development
in vitro and in vivo. Neurosci Lett, 86: 33-37.
Cools A, Salle HJA, Verberk MM, & Zielhuis RL (1976) Biochemical
response of male volunteers ingesting inorganic lead for 49 days. Int
Arch Occup Environ Health, 38: 129-139.
Cooney GH, Bell A, McBride W, & Carter C (1989a) Neurobehavioural
consequences of prenatal low level exposures to lead. Neurotoxicol
Teratol, 11: 95-104.
Cooney GH, Bell A, McBride W, & Carter C (1989b) Low level exposures
to lead: the Sydney lead study. Dev Med Child Neurol, 31: 640-649.
Cooney G, Bell A, & Stavron C (1991) Low level exposures to lead and
neurobehavioural development: the Sydney study at seven years. In:
Heavy metals in the environment. Edinburgh, CEP Consultants, pp 16-19.
Cooper WC (1981) Mortality in employees of lead production facilities
and lead battery plants, 1971-1975. In: Environmental lead:
Proceedings of the Second International Symposium on Environmental
Lead Research, Cincinnati. Ohio, December 1978. New York, London, San
Francisco, pp 111-143.
Cooper WC (1988) Deaths from chronic renal disease in US battery and
lead production workers. Environ Health Perspect, 78: 61-63.
Cooper WC & Gaffey WR (1975) Mortality of lead workers. J Occup Med,
17: 100-107.
Cory-Slechta DA (1990a) Lead exposure during advanced age: alterations
in kinetics and biochemical effects. Toxicol Appl Pharmacol,
104: 67-78.
Cory-Slechta DA (1990b) Exposure duration modifies the effects of low
level lead on fixed interval performance. Neurotoxicology,
11: 427-442.
Cory-Slechta DA & Pokora MJ (1991) Behavioral manifestations of
prolonged lead exposure initiated at different stages of the life
cycle: I. Schedule-controlled responding. Neurotoxicology,
12(4): 745-760.
Cory-Slechta DA & Thompson T (1979) Behavioural toxicity of chronic
post-weaning lead exposure in the rat. Toxicol Appl Pharmacol,
47: 151-159.
Cory-Slechta DA & Widzowski DV (1991) Low level lead exposure
increases sensitivity to the stimulus properties of dopamine D1 and D2
agonists. Brain Res, 553: 65-74.
Cory-Slechta DA, Weiss B, & Cox C (1985) Performance and exposure
indices of rats exposed to low concentrations of lead. Toxicol Appl
Pharmacol, 78: 291-299.
Coscia GC, Discalzi G, & Ponzetti C (1987) Immunological aspects of
occupational lead exposure. Med Lav, 78: 360-364.
Cramer K, Goyer RA, Jagenburg OR, & Wilson MH (1974) Renal
ultrastructure, renal function and parameters of lead toxicity in
workers with different length of lead exposure. Br J Ind Med,
31: 113-127.
Cueto LM, Baer RD, & Gonzalez EM (1989) Three cases of unusual lead
poisoning. Am J Gastroenterol, 84: 1460.
Culbard EB, Thornton L, Watt JM, Wheatley M, Moorcroft S, & Thompson M
(1988) Metal contamination in British urban dusts and soils. J Environ
Qual, 17: 226-234.
Cullen MR, Kayne RD, & Robins JM (1984) Endocrine and reproductive
dysfunction in men associated with occupational inorganic lead
intoxication. Arch Environ Health, 39: 431-440.
Dabeka RW & McKenzie AD (1987) Lead, cadmium, and fluoride levels in
market milk and infant formulas in Canada. J Assoc Off Anal Chem,
70(4):754-757.
Dabeka RW, McKenzie AD, & Lacroix GMA (1987) Dietary intakes of lead,
cadmium, arsenic and fluoride by Canadian adults: a 24-hour duplicate
diet study. Food Addit Contam, 4(1): 89-102.
Dalpra L, Tibiletti MG, Nocera G, Giulotto P, Auriti L, Carnelli V, &
Simoni G (1983) SCE analysis in children exposed to lead emission from
a smelting plant. Mutat Res, 120: 249-256.
Danielson BRG, Dencker L, & Lindgren A (1983) Transplacental movement
of inorganic lead in early and late gestation in the mouse. Arch
Toxicol, 54: 97-107.
David O, Clark J, & Voller K (1972) Lead and hyperactivity. Lancet,
2: 900-903.
Davidson C, Grimm T, & Nasta M (1981) Airborne lead and other elements
derived from local fires in the Himalays. Science, 214: 1344-1346.
Davies BE (1983) A graphical estimation of the normal lead content of
some British soils. Geoderma, 29: 67-75.
Davies JM (1984) Lung cancer mortality among workers making lead
chromate and zinc chromate pigments at three English factories. Br J
Ind Med, 41: 158-169.
Davies PH & Everhart WH (1973) Effects of chemical variations in
aquatic environments: Volume 3 - Lead toxicity to rainbow trout and
testing application factor concept. Washington, DC, US Environmental
Protection Agency (EPA Report No. R373011C).
Davies BE, Elwood PC, Gallacher J, & Ginnever RC (1985) The
relationships between heavy metals in garden soils and house dusts in
an old lead mining area of North Wales, Great Britain. Environ Pollut,
B9: 255-266.
Davies DJ, Thornton I, Watt JM, Culbard EB, Harvey PG, Delves HT,
Sherlock JC, Smart GA, Thomas JFA, & Quinn MJ (1987a) Lead intake and
blood lead in 2 year olds UK urban children. Sci Total Environ,
90: 13-29.
Davies DJ, Watt JM, & Thornton I (1987b) Lead levels in Birmingham
dusts and soils. Sci Total Environ, 67: 177-185.
Davis JM & Grant LD (1992) The sensitivity of children to lead. In:
Guzelian PS, Henry CJ, & Olin S ed. Similarities and differences
between children and adults: Implications for risk assessment.
Washington, DC, ILSI Press, pp 15-27.
Davis JM & Svendsgaard DJ (1990) Nerve conduction velocity and lead: A
critical review and meta-analysis. In: Johnson BL, Anger WK, Durao A,
& Xinteras C ed. Advances in neurobehavioural toxicology: Applications
in environmental and occupational health. Chelsea, Michigan, Lewis
Publishers, pp 353-376.
Davis S, Waller P, Buschbom R, Balfour J, & White P (1990a)
Quantitative estimates of soil ingestion in normal children between
the ages of 2 and 7 years: Populations-based estimates using
aluminium, silicon, and titanium as soil tracer elements. Arch Environ
Health, 45: 112-122.
Davis JM, Otto DA, Weil DE, & Grant LD (1990b) The comparative
developmental neurotoxicity of lead in humans and animals.
Neurotoxicol Teratol, 12: 215-229.
Day JP, Hart M, & Robinson MS (1975) Lead in urban street dust. Nature
(Lond), 253: 343-345.
Dees JH, Heatfield BM, Reuben MD, & Trump BF (1980) Adenocarcinoma of
the kidney. III. Histogenesis of renal adenocarcinomas induced in rats
by N-(4-fluoro-4-biphenyl)acetamide. J Natl Cancer Inst,
64: 1537-1545.
De La Burde B & Choate MS (1972) Does asymptomatic lead exposure in
children have latent sequelae? J Pediatr, 81: 1088-1096.
De La Burde B & Choate MS (1975) Early asymptomatic lead exposure and
development at school age. J Pediatr, 87: 638-664.
De Leacy EA (1987) Lead-crystal decanters - a health risk? Med J Aust,
147: 622.
Delumyea R & Kalivretenos A (1987) Elemental carbon and lead content
of fine particles from American and French cities of comparable size
and industry, 1985. Atmos Environ, 21: 1643-1647.
Delves HT & Campbell MJ (1988) Measurements of total lead
concentrations and of lead isotope ratios in whole blood by use of
inductively coupled plasma source mass spectrometry. J Anal At
Spectrom, 3: 343-348.
Desilva PE & Donnan MB (1980) Blood lead levels in Victorian children.
Med J Aust, 2: 315-318.
Diemel JAL, Brunekreef B, Boleu JSM, Biersteker K, & Veenstra SJ
(1981) The Arnhem lead study. II. Indoor pollution, and indoor/outdoor
relationships. Environ Res, 25: 449-456.
Dieter M, Mathews HB, Jeffcoate RA, & Moseman RF (1993)
Bioavailability of lead found in mine tailings from Alaska. J Toxicol
Environ Health, 39: 79-93.
Dietrich KN, Krafft KM, Bier M, Succop P, Berger O, & Bornschein R
(1986) Early effects of fetal lead exposure: neurobehavioural findings
at six months. Int J Biosoc Res, 8: 151-168.
Dietrich KN, Krafft KM, Bornschein RL, Hammond PB, Berger OM, Succop
PA, & Bier M (1987) Low-level fetal lead exposure effect on
neurobehavioural development in early infancy. Pediatrics,
80: 721-730.
Dietrich KN, Krafft KM, Bier M, Berger O, Succop PA, & Bornschein RL
(1989) Neurobehavioural effects of foetal lead exposure: the first
year of life. In: Smith MA, Grant LD, & Sors AI ed. Lead exposure and
child development: An international assessment. Dordrecht, London,
Kluwer Academic Publishers, pp 320-331.
Dietrich KN, Succop PA, Bornschein RL, Krafft KM, Berger O, Hammond
PB, & Buncher CR (1990) Lead exposure and neurobehavioural development
in later infancy. Environ Health Perspect, 89: 13-19.
Dietrich KN, Succop PA, Berger OG, Hammond PB, & Bornschein RL (1991)
Lead exposure and cognitive development of urban, preschool children:
the Cincinnati lead cohort at age 4 years. Neurotoxicol Teratol,
13: 203-211.
Dietrich KN, Succop PA, Berger OG, & Keith RW (1992) Lead exposure and
the central auditory processing abilities and cognitive development of
urban children: the Cincinnati lead study cohort at age 5 years.
Neurotoxicol Teratol, 14: 51-56.
Dietrich K, Berger O, Succop P, Hammond P, & Bornschein R (1993a) The
developmental consequences of low to moderate prenatal and postnatal
lead exposure: Intellectual attainment in the Cincinnati Lead Study
Cohort following school entry. Neurotoxicol Teratol, 15: 37-44.
Dieterich K, Berger O, & Succop P (1993b) Lead exposure and the motor
developmental status of urban 6-year-old children in the Cincinnati
Prospective Study. Pediatrics, 91: 301-307.
Djuric D, Kerin Z, Graovac-Leposavic L, Novak L, & Kop M (1971)
Environmental contamination by lead from a mine and smelter: A
preliminary report. Arch environ Health, 23: 275-279.
Dolenc P, Staessen JA, Lauwerys RR, & Amery A (1993) Short report:
low-level lead exposure does not increase the blood pressure in the
general population. J Hypertens, 11: 589-593.
Dong A, Simsiman GV, & Chesters G (1985) Release of phosphorus and
metals from soils and sediments during dispersion. Soil Sci,
139: 97-99.
Donovan MP, Schein LG, & Thomas JA (1980) Inhibition of
androgen-receptor interaction in mouse prostate gland cytosol by
divalent metal ions. Mol Pharmacol, 17: 156-162.
Drasch GA & Ott J (1988) Lead in human bones. Investigations of an
occupationally non-exposed population in southern Bavaria (FGR). II.
Children. Sci Total Environ, 68: 61-69.
Drasch GA, Bohm J, & Baur C (1987) Lead in human bones. Investigations
of an occupationally non-exposed population in southern Bavaria (FGR).
I. Adults. Sci Total Environ, 64: 303-315.
Dresner DL, Ibrahim NG, Mascarenhas BR, & Levere RD (1982) Modulation
of bone marrow heme and protein synthesis by trace elements. Environ
Res, 28: 55-66.
Drill S, Konz J, Mahar H, & Morse M (1979) The environmental lead
problem: An assessment of lead in drinking water from a multi-media
perspective. Washington, DC, US Environmental Protection Agency
(EPA-570/9-79-003, NTIS-PB-296556).
Duggan MJ (1980) Lead in urban dust: An assessment. Water Air Soil
Pollut, 14: 309-321.
Egle P & Shelton KR (1986) Chronic lead intoxication causes a brain
specific nuclear protein to accumulate in the nuclei of cells lining
kidney tubules. J Biol Chem, 261: 2294-2298.
Elias R (1985) Lead exposures in the human environment. In: Mahaffey
KR ed. Dietary and environmental lead: human health effects.
Amsterdam, Oxford, New York, Elsevier Science Publishers, pp 79-109.
Ellen G & van Loon JW (1990) Determination of cadmium and lead in
foods by graphite furnace atomic absorption spectrometry with Zeeman
background correction: test with certified reference materials. Food
Addit Contam, 7: 265-273.
Elwood PC (1986) The sources of lead in blood: A critical review. Sci
Total Environ, 52: 1-23.
Elwood PC, Davey-Smith G, Oldham PD, & Toothill C (1988a) Two Welsh
surveys of blood lead and blood pressure. Environ Health Perspect,
78: 119-121.
Elwood PC, Yarnell JWG, Oldham PD, Catford JC, Nutbeam D, Davey-Smith
G, & Toothill C (1988b) Blood pressure and blood lead in surveys in
Wales. Am J Epidemiol, 127: 942-945.
Ernhart B & Morrow-Tlucak M (1987) The caretaking environment and
exposure of children to lead. In: Thornton I & Culbard E ed. Lead in
the home environment. Northwood, Science Reviews Ltd.
Ernhart CB, Wolf AW, Sokol RJ, Brittenham GM, & Ernhard P (1985) Fetal
lead exposure: antenatal factors. Environ Res, 38: 54-66.
Ernhart CB, Wolf WW, Kennard MJ, Erhard P, Filipovick HF, & Sokol RJ
(1986) Intrauterine exposure to low levels of lead: the status of the
neonate. Arch Environ Health, 41: 287-291.
Ernhart CB, Morrow-Tlucak M, Marler MR, & Wolf AW (1987) Low level
lead exposure in the prenatal and early preschool periods: early
preschool development. Neurotoxicol Teratol, 9: 259-270.
Ernhart CB, Morrow-Tlucak M, & Wolf AW (1988) Low level lead exposure
and intelligence in the preschool years. Sci Total Environ,
71: 453-459.
Ernhart CB, Morrow-Tlucak M, Worf AW, Super D, & Drotar D (1989a) Low
level lead exposure in the prenatal and early preschool periods:
intelligence prior to school entry. Neurotoxicol Teratol, 11: 161-170.
Ernhart CB, Brittenham G, Marler MR, & Sokol RJ (1989b) Lead related
birth defects: some methodologic issues. In: Smith MA, Grant LD, &
Sors AI ed. Lead exposure and child development: An international
assessment. Dordrecht, London, Kluwer Academic Publishers, pp 357-370.
Evans RD & Rigler FH (1985) Long distance transport of anthropogenic
lead as measured by lake sediments. Water Air Soil Pollut,
24: 141-151.
Everson J & Patterson CC (1980) "Ultra-clean" isotope dilution/mass
spectrometric analyses for lead in human blood plasma indicate that
most reported values are artificially high. Clin Chem, 26: 1603-1607.
Evis MJ, Kane KA, Moore MR, & Parratt JR (1985) The effects of chronic
low lead treatment and hypertension on the severity of cardiac
arrhythmias induced by coronary artery ligation in anaesthetized rats.
Toxicol Appl Pharmacol, 80: 234-242.
Evis MJ, Dhaliwal K, Kane KA, Moore MR, & Parratt JR (1987) The
effects of chronic lead treatment and hypertension on the severity of
cardiac arrhythmias induced by coronary artery occlusion or by
noradrenaline in anaesthetised rats. Arch Toxicol, 59: 336-340.
Ewers U, Stiller-Winkler R, & Idel H (1982) Serum immunoglobulin,
complement C3, and salivary IgA level in lead workers. Environ Res,
29: 351-357.
Fanning D (1988) A mortality study of lead workers, 1926-1985. Arch
Environ Health, 43: 247-251.
FAO/WHO (1993) Evaluation of certain food additives and contaminants.
Forty-first report of the Joint FAO/WHO Expert Committee on Food
Additives. Geneva, World Health Organization (Technical Report Series
837).
Feigl F, Anger V, & Oesper RE (1972) Spot tests in inorganic analysis.
London, Elsevier Publishing Company, pp 285-286.
Fergusson DM, Fergusson JE, Horwood LJ, & Kinzett NG (1988a) A
longitudinal study of dentine lead levels, intelligence, school
performance and behaviour. Part I. Dentine lead levels and exposure to
environmental risk factors. J Child Psychol Psychiatr, 29: 781-792.
Fergusson DM, Fergusson JE, Horwood LJ, & Kinzett NG (1988b) A
longitudinal study of dentine levels, intelligence, school performance
and behaviour. Part II. Dentine lead and cognitive ability. J Child
Psychol Psychiatr, 29: 793-809.
Fergusson DM, Fergusson JE, Horwood LJ, & Kinzett NG (1988c) A
longitudinal study of dentine lead levels, intelligence, school
performance and behaviour. Part III. Dentine lead levels and
attention/activity. J Child Psychol Psychiatr, 29: 811-824.
Fergusson JE, Fergusson DM, Horwood LJ, & Kinzett NG (1989) A
longitudinal study of dentine lead levels, intelligence, school
performance and behaviour: The measurement of dentine lead. Sci Total
Environ, 80: 229-241.
Fernando NP, Healy MA, Aslam M, Davis SS, & Hussein A (1981) Lead
poisoning and traditional practices: The consequences for world
health. A study in Kuwait. Public Health (Lond), 95: 250-260.
Fischbein A, Alvares AP, Anderson KE, Sassa S, & Kappas A (1977) Lead
intoxication among demolition workers: the effect of lead on the
hepatic cytochrome P-450 system in humans. J Toxicol Environ Health,
3: 431-437.
Fischbein A, Rice C, Sarkozi L, Kon SH, Petrocci M, & Selikoff IJ
(1979) Exposure to lead in firing ranges. J Am Med Assoc,
241: 1141-1144.
Fischbein A, Thornton J, Blumberg WE, Bernstein J, Valciukas JA, Moses
M, Davidow B, Kaul B, Sirota M, & Selikoff IJ (1980) Health status of
cable splicers with low-level exposure to lead: results of a clinical
survey. Am J Public Health, 70: 697-700.
Flegal AR, Rosman KJR, & Stephenson MD (1987) Isotope systematics of
contaminant leads in Monterey Bay. Environ Sci Technol, 21: 1075-1079.
Flora S-S & Tandon SK (1987) Effect of combined exposure to lead and
ethanol on some biochemical indices in the rat. Biochem Pharmacol,
36: 537-541.
Florence TM, Lilley SG, & Stauber JL (1988) Skin absorption of lead.
Lancet, 2: 157-158.
Fouassin A & Fondu M (1980) Evaluation of the daily intake of lead and
cadmium from food in Belgium. Arch Belg Méd Soc Hyg Méd Trav Méd Lég,
38: 453-467.
Foster WG (1992) Reproductive toxicity of chronic lead-exposure in the
female cynomolgus monkeys. J Reprod Toxicol, 16: 123-131.
Fouts PJ & Page JH (1942) The effect of chronic lead poisoning on
arterial blood pressure in dogs. Am Heart J, 24: 329-331.
Fowler BA, Kimmel CA, Woods JS, McConnell EE, & Grant LD (1980)
Chronic low-level lead toxicity in the rat: III. An integrated
assessment of long-term toxicity with special reference to the kidney.
Toxicol Appl Pharmacol, 56: 59-77.
Fox DA & Sillman AJ (1979) Heavy metals affect rod, but not cone
pholoreceptors. Science, 206: 78-80.
Fox DA, Lewkowski JP, & Cooper GP (1977) Acute and chronic effects of
neonatal lead exposure on developmental of the visual worked response
in rats. Toxicol Appl Pharmacol, 40: 449-461.
Freeman GB, Johnson JD, Killinger JM, Liao SC, Feder PI, Davis AO,
Ruby MV, Chaney RL, Lovre SC, & Bergstrom PD (1992) Relative
bioavailability of lead from mining waste soils in rats. Fundam Appl
Toxicol, 19: 388-398.
Friberg L (1988) Quality assurance. In: Clarkson TW, Friberg L,
Nordstrom GF, & Sager PR ed. Biological monitoring of toxic metals.
New York, Plenum Press, pp 103-126.
Friberg L & Vahter M (1983) Assessment of exposure to lead and cadmium
through biological monitoring: Results of a UNEP/WHO global study.
Environ Res, 30: 95-128.
Friedman JA & Weinberger HL (1990) Six children with lead poisoning. J
Dis Child, 144: 1039-1044.
Fullmer CS & Rosen JF (1990) Effect of dietary calcium and lead status
on intestinal calcium absorption. Environ Res, 51: 91-99.
Fullmer CS (1990) Intestinal lead and calcium absorption: Effect of
1,25-dihydroxy-cholecalciferol and lead status. Proc Soc Exp Biol Med,
194: 258-264.
Fulton M, Thomson G, Hunter R, Raab G, Laxen D, & Hepburn W (1987)
Influence of blood lead on the ability and attainment of children in
Edinburgh. Lancet, 30 May: 1221-1226.
Galal-Gorchev H (1991a) Dietary intake of pesticide residues, cadmium,
mercury and lead. Food Addit Contam, 8(6): 793-806.
Galal-Gorchev H (1991b) Global overview of dietary lead exposure.
Special issue incorporating the Proceedings of the Symposium on the
Bioavailability and Dietary Exposure of Lead, Chapel Hill, North
Carolina, September 1990. Chem Speciation Bioavailab, 3(3/4): 5-11.
Garber BT & Wei E (1974) Influence of dietary factors on the
gastrointestinal absorption of lead. Toxicol Appl Pharmacol,
27: 685-691.
Garcia-Miragaya J (1984) Levels, chemical fractionation, and
solubility of lead in roadside soils of Caracas, Venezuela. Soil Sci,
138: 147-152.
Gartrell MJ, Craun JC, Podrebarac DS, & Gunderson EL (1985a)
Pesticides, selected elements, and other chemicals in infant and
toddler total diet samples, October 1979 -September 1980. J Assoc Off
Anal Chem, 68: 1184-1187.
Gartrell MJ, Craun JC, Podrebarac DS, & Gunderson EL (1985b)
Pesticides, selected elements, and other chemicals in infant and
toddler total diet samples, October 1979 -September 1980. J Assoc Off
Anal Chem, 81: 1163-1183.
Gartside PS (1988) The relationship of blood lead levels and blood
pressure in NHANES II: additional calculations. Environ Health
Perspect, 78: 31-34.
Gartside PS & Buncher CR (1982) Relationship of air lead and blood
lead for workers at an automobile battery factory. Int Arch Occup
Environ Health, 50: 1-10.
GEMS (1985) Global Environmental Monitoring System: Assessment of
human exposure to lead: Comparison between Belgium, Malta, Mexico and
Sweden. Stockholm, Karolinska Institute.
Gennart JP, Bernard A, & Lauwerys R (1992) Assessment of thyroid,
testes, kidney and autonomic nervous system function in lead-exposed
workers. Int Arch Occup Environ Health, 64: 49-57.
Gerhardsson L, Lundstrom N-G, Nordbery G, & Wall S (1986) Mortality
and lead exposure: a retrospective cohort study of Swedish smelter
workers. Br J Ind Med, 43: 707-712.
Getz LL, Haney AW, Larimore RW, McNurney JW, Leland HV, Price PW,
Rolfe GL, Wortman RL, Hudson JL, Solomon RL, & Reinbold KA (1977)
Transport and distribution in a watershed ecosystem. In: Boggess WR
ed. Lead in the environment. Springfield, Virginia, National Technical
Information Service, pp 105-133 (National Science Foundation report
No. NSF/RA-770214).
Gibson JL (1904) A plea for painted railings and painted walls of
rooms as the source of lead poisoning amongst: Queensland children.
Aust Med Gaz, 23: 149.
Gilbert SG & Rice DC (1987) Low-level lifetime lead exposure produces
behavioral toxicity (spatial discrimination reversal) in adult
monkeys. Toxicol Appl Pharmacol, 91: 484-490.
Gmerek DE, McCofferty MR, O'Neill KJ, Melamed BR, & O'Neill JJ (1981)
Effect of inorganic lead on rat brain mitochondrial respiration and
energy production. J Neurochem, 36: 1109-1113.
Goldstein GW (1990) Lead poisoning and brain cell function. Environ
Health Perspect, 89: 91-94.
Gould JH, Capar SG, Jacobs RM, & Plunkett R (1988) A quick color test
to detect lead release from glazed ceramic and enameled metalware.
Anal Lett, 21: 2145-2154.
Goyer RA (1985) Renal changes associated with lead exposure. In:
Mahaffey KR ed. Dietary and environmental lead: Human health effects.
Amsterdam, Oxford, New York, Elsevier Science Publishers, pp 315-335.
Goyer RA (1992) Nephrotoxicity and carcinogenicity of lead - An update
on exposure and effects of lead. Fundam Appl Toxicol, 18: 1-16.
Goyer RA (1993) Lead toxicity: current concerns. Environ Health
Perspect, 100: 177-187.
Goyer RA & Rhyne BC (1973) Pathological effects of lead. Int Rev Exp
Pathol, 12: 1-77.
Goyer RA & Wilson MH (1975) Lead induced inclusion bodies: results of
ethylenediamine-tetra-acetic acid treatment. Lab Invest, 32: 149-156.
Goyer RA, Leonard DL, Moore JF, Rhyne B, & Krigman MR (1970) Lead
dosage and the role of the intranuclear inclusion body. Arch Environ
Health, 20: 705-711.
Goyer RA, Falk HL, Hogan M, Feldman DD, & Richter W (1981) Renal
tumors in rats given trisodium nitrilotriacetic acid in drinking water
for 2 years. J Natl Cancer Inst, 66: 869-880.
Goyer RA, Weinberg CR, Victery WM, & Miller CR (1989) Lead induced
nephrotoxicity: kidney calcium as an indicator of tubular injury. In:
Bach PH & Lock EA ed. Nephrotoxicity. New York, London, Plenum Press,
pp 11-20.
Grandjean P (1979) Occupational lead poisoning in Denmark: screening
with the haematofluorometer. Br J Ind Med, 36: 52-58.
Grandjean P & Olsen B (1984) Lead. In: Vercruysse A ed. Techniques and
instrumentation in analytical chemistry. Volume 4: Evaluation of
analytical methods in biological systems. Part B: Hazardous metals in
human toxicology. Amsterdam, Oxford, New York, Elsevier Science
Publishers, pp 153-169.
Grandjean P, Wulf HC, & Niebuhr E (1983) Sister chromatid exchange in
response to variations in occupational lead exposure. Environ Res,
32: 199-204.
Grandjean P, Hansen ON, & Lyngbye K (1984) Analysis of lead in
circumpulpal dentin of deciduous teeth. Ann Clin Lab Sci, 14: 270-275.
Grandjean P, Hansen ON, & Lyngbye K (1986) Lead concentration in
deciduous teeth; variation related to tooth type and analytical
technique. J Toxicol Environ Health, 19: 437-445.
Grandjean P, Jensen BM, Sando SH, Jorgensen PJ, & Antonsen S (1989a)
Delayed blood regeneration in lead exposure: an effect on reserve
capacity. Am J Public Health, 79: 1385-1388.
Grandjean P, Hollnagel H, Hedegaard L, Christensen JM, & Larsen S
(1989b) Blood lead-blood pressure relations: alcohol intake and
haemoglobin as confounders. Am J Epidemiol, 129: 732-739.
Grandjean P, Lyngbye T, & Hansen ON (1991) Lessons from a Danish study
on neuropsychological impairment related to lead exposure. Environ
Health Perspect, 94: 111-115.
Granick JL, Sassa S, Granick S, Levere RD, & Kappas A (1973) Studies
in lead poisoning. II. Correlation between the ratio of activated to
inactivated aminolevulinic acid dehydratase of whole blood and the
blood lead level. Biochem Med, 8: 149-159.
Grant LD & Davis JM (1989) Effects of low-level lead exposure on
paediatric neurobehavioural development: Current findings and future
directions. In: Smith MA, Grand LD, & Sors AI ed. Lead exposure and
child development: An international Assessment. Dordrecht, London,
Kluwer Academic Publishers, pp 49-115.
Grant LD, Kimmel CA, West GL, Martinez-Vargas CM, & Howard JL (1980)
Chronic low-level lead toxicity in the rat. II. Effects on postnatal
physical and behavioral development. Toxicol Appl Pharmacol, 56:
42-58.
Graziano JH & Blum C (1991) Lead exposure from lead crystal. Lancet,
337: 141-142.
Graziano JH, Popovac D, Factor-Litvak P, Shrout P, Kline J, Murphy MJ,
Zhao Y, Mehmeti A, Ahmedi X, Rajovic B, Zvicer Z, Nenezic U, Lolacono
NJ, & Steun Z (1990) Determinants of elevated blood lead during
pregnancy in a population surrounding a lead smelter in Kosova,
Yugoslavia. Environ Health Perspect, 89: 95-100.
Graziano JH, Slavkovic V, Factor-Litvak P, Popovac D, Ahmedi X, &
Mehmeti A (1991) Depressed serum erythropoietin in pregnant women with
elevated blood lead. Arch Environ Health, 46: 347-350.
Greene T, Ernhart CB, & Boyd TA (1992) Contributions of risk factors
to elevated blood and dentine levels in preschool children. Sci Total
Environ, 115: 239-260.
Griffith JQ & Landaver MA (1944) The effect of chronic lead poisoning
on arterial blood pressure in rats. Am Heart J, 28: 295-297.
Gulson B & Wilson D (1994) History of lead exposure in children
revealed from isotopic analyses of teeth. Arch Environ Health,
49X: 279-283.
Gupta AK, Sehgal SK, Mohan M, & Anand NK (1990) Cosmetic plumbism.
Indian Paediatr, 27: 760-761.
Gustafson A, Hedmer P, Schutz A, & Skerfving S (1989) Occupational
lead exposure and pituitary function. Int Arch Occup Environ Health,
61: 277-281.
Haeger-Aronsen B (1960) Studies on urinary excretion of
delta-aminolaevulic acid and other haem precursors in lead workers and
lead-intoxicated rabbits. Scand J Clin Lab Invest, 12(47): 1.
Haenninen H, Mantere P, Hernberg S, Seppalainen AM, & Kock B (1979)
Subjective symptoms in low-level exposure to lead. Neurotoxicology,
1: 333-347.
Hakim RB, Stewart WF, Canner JK, & Tielsch JM (1991) Occupational lead
exposure and strabismus in offspring: A case control study. Am J
Epidemiol, 133: 351-356.
Hammond PB, O'Flaherty EJ, & Gartside PS (1981) The impact of air-lead
on blood-lead in man: A critique of the recent literature. Food Cosmet
Toxicol, 19: 631-638.
Hammond PB, Bornschein RL, & Succop P (1985) Dose-effect and
dose-response relationships of blood lead to erythrocytic
protoporphyrin in young children. Environ Res, 38: 187-196.
Hansen ON, Trillingsgaard A, Beese I, Lyngbye T, & Grandjean P (1989)
A neuropsychological study of children with elevated dentine lead
level: assessment of the effect of lead in different socio-economic
groups. Neurotoxicol Teratol, 11: 205-213.
Harlan WR (1988) The relationship of blood lead levels to blood
pressure in the US population. Environ Health Perspect, 78: 9-13.
Harlan WR, Landis JR, Schmouder RL, Goldstein NG, & Harlan LC (1985)
Blood lead and blood pressure: relationship in the adolescent and
adult US population. J Am Med Assoc, 253: 530-534.
Harvey PG, Hamlin MW, Kumar R, & Delves HT (1984) Blood lead,
behaviour and intelligence test performance in preschool children. Sci
Total Environ, 40: 45-60.
Harvey PG, Hamlin MW, Kumar R, Morgan J, Spurgeon A, & Delves HT
(1988) Relationships between blood lead, behaviour, psychometric and
neuropsychological test performance in young children. Br J Dev
Psychol, 6: 145-156.
Hatzakis A, Kokkevi A, Maravelias C, Katsouyanni K, Salaminios F,
Kalandidi A, Koutselinis A, Stefanis C, & Trichopoulos D (1989)
Psychometric intelligence deficits in lead-exposed children. In: Smith
MA, Grand LD, & Sors AI ed. Lead exposure and child development: An
international assessment. Dordrecht, London, Kluwer Academic
Publishers, pp 211-223.
Hawk BA, Schroeder SR, Robinson G, Otto D, Mushak P, Kleinbaum D, &
Dawson G (1986) Relation of lead and social factors to IQ of low-SES
children: a partial replication. Am J. Ment Defic, 91: 178-183.
Heard MJ & Chamberlain AC (1982) Effects of minerals and food on
uptake of lead from the gastrointestinal tract in humans. Hum Toxicol,
1: 411-416.
Hem JD & Durum WH (1973) Solubility and occurrence of lead in surface
water, J An Water Works Assoc, 65: 562-568.
Henning SJ & Cooper LC (1988) Intestinal accumulation of lead salts
and milk lead by suckling rats (42645). Proc Soc Exp Biol Med,
187: 110-116.
Hernberg S (1973) Prevention of occupational poisoning from inorganic
lead. Work Environ Health, 10: 53-61.
Hernberg S & Nikkanen J (1970) Enzyme inhibition by lead under normal
urban conditions. Lancet, 1(7637): 63-64.
Hilderbrand DC, Der R, Griffin WT, & Fahim MS (1973) Effect of lead
acetate on reproduction. Am J Obstet Gynecol, 115: 1058-1065.
Hogstedt C, Hane M, Agrell A, & Bodin L (1983) Neuropsychological test
results and symptoms among workers with well-defined long term
exposure to lead. Br J Ind Med, 49: 99-105.
Holness DL & Nethercott JR (1988) Acute lead intoxication in a group
of demolition workers. Appl Ind Hyg, 3: 338-341.
Holtzman D, Shen Hgu J, & Montell P (1978) In vitro effects of
inorganic lead on isolated rat brain mitochondrial respiration.
Neurochem Res, 3: 195-206.
Holtzman D, Shen Hgu J, & Desautel M (1981) Absence of effects of lead
feedings and growth retardation on mitochondrial and microsomal
cytochrome in the developing brain. Toxicol Appl Pharmacol, 58: 48-56.
Holtzman D, Devries C, Nguyan H, Olson J, & Bensch K (1984) Maturation
of resistance to lead encephalopathy: cellular and subcellular
mechanisms. Neurotoxicology, 5: 97-124.
Hopper JL, Balderas A, & Mathews JO (1982) Analysis of variation in
blood lead levels in Melbourne families. Med J Aust, 2: 573-576.
Huang J, He F, Yigun W, & Zhang S (1988a) Observations on renal
function in workers exposed to lead. Sci Total Environ, 71: 535-537.
Huang XP, Feng ZY, Zhai WL, & Xu JH (1988b) Chromosomal aberrations
and sister chromatid exchanges in workers exposed to lead. Biomed
Environ Sci, 1: 382-387.
Huber F & Schmidt U (1976) Methylation of organolead and lead (II)
compounds to C3 4Pb by microorganisms. Nature (Lond),
259: 157-158.
Hutton M, Wadge A, & Milligan PJ (1988) Environmental levels of
cadmium and lead in the vicinity of a major refuse incinerator. Atmos
Environ, 22: 411-416.
IAEA (1987) Co-ordinated research programme on human daily dietary
intakes of nutritionally important trace elements as measured by
nuclear and other techniques. IAEA Newl, 2: 6-15.
IARC (1980) Lead and lead compounds. In: Some metals and metallic
compounds. Lyon, International Agency for Research on Cancer, pp
325-416 (IARC Monographs on the Evaluation of the Carcinogenic Risk of
Chemicals to Humans, Volume 23).
IARC (1987a) Genetic and related effects: An updating of selected IARC
Monographs from Volumes 1 to 42. Lyon, International Agency for
Research on Cancer (IARC Monographs on the Evaluation of the
Carcinogenic Risk of Chemicals to Humans, Supplement 6).
IARC (1987b) Lead and lead compounds. In: Overall evaluations of
carcinogenicity: an updating of IARC Monographs, Volumes 1 to 42.
Lyon, International Agency for Research on Cancer, pp 230-232 (IARC
Monographs on the Evaluation of the Carcinogenic Risk of Chemicals to
Humans, Supplement 7).
Ibels LS & Pollock CA (1986) Toxicology management review. Lead
intoxication. Med Toxicol, 1: 387-410.
Ichiba M & Tomokuni K (1990) Studies on erythrocyte pyrimidine
5'-nucleotidase test and its evaluation in workers occupationally
exposed to lead. Int Arch Occup Environ Health, 62: 305-310.
ILZSG (International Lead and Zinc Study Group) (1992) Lead and zinc
statistics. Int Lead Zinc Study Group Bull, 32: 3.
IPCS (1977) Environmental health criteria 3: Lead. Geneva, World
Health Organization, 160 pp.
IPCS (1989) Environmental health criteria 85: Lead - Environmental
effects. Geneva, World Health Organization, 106 pp.
IPCS (1994) Environmental health criteria 155: Biomarkers and risk
assessment: Concepts and principles. Geneva, World Health
Organization, 82 pp.
IRPC (International Radiological Protection Commission), Task Group on
Lung Dynamics (1966) Deposition and retention models for internal
dosimetry of the human respiratory trace. Health Phys, 12: 173-207.
IRPTC (1987) IRPTC legal file 1986. Geneva, International Register of
Potentially Toxic Chemicals, United Nations Environment Programme,
vol 1.
Irwig LM, Harrison WO, Rocks P, Webster I, & Andrew M (1978) Lead and
morbidity: a dose response relationship. Lancet, 2(8079): 4-7.
Jaakkola J & Anttila A (1992) [Occupational exposures to chemical
agents in Finland.] Helsinki, Institute of Occupational Health and
Finnish Work Environmental Fund (in Finnish).
James AC (1978) Lung deposition of submicron aerosols calculated as a
function of age and breathing rate. In: National Radiological
Protection Board Annual Report. Harwell, UK, National Radiological
Protection Board, pp 71-75.
Jaremin B (1983) Blast lymphocyte transformation (LTT), rosette
(E-RFC) and leukocyte migration inhibition (MIF) tests in persons
exposed to the action of lead during work. Report II. Bull Inst Marit
Trop Med (Gdynia), 34: 187-197.
Jensen AA (1984) Metabolism and toxicokinetics. In: Grandjean P ed.
Biological effects of organolead compounds. Boca Raton, Florida, CRC
Press, chapter 8, pp 97-115.
Johnson NE & Tenuta K (1979) Diets and lead blood levels of children
who practice pica. Environ Res, 18: 369-376.
Jorhem L & Slorach S (1988) Lead, chromium, tin, iron, and cadmium in
foods in welded cans. Food Addit Contam, 4: 309-316.
Jorhem L, Mattsson P, & Slorach S (1988) Lead in table wines on the
Swedish market. Food Addit Contam, 5: 645-649.
Kaminsky P, Leone J, & Duc M (1988) Incidence du saturnisme hydrique
dans un service de médecine interne en région de sols acides. Presse
Méd, 17: 419-422.
Kampe W (1983) Lead and cadmium in food - is there an actual danger?
Forum Staedte-Hyg, 34: 236-241.
Kang HK, Infante PF, & Carra JS (1980) Occupational lead exposure and
cancer (letter). Science, 207: 935-936.
Katagiri Y, Toriumi H, & Kawai M (1983) Lead exposure among 3-year-old
children and their mothers living in a pottery-producing area. Int
Arch Occup Environ Health, 52: 223-229.
Kaye WE, Novotny TE, & Tucker M (1987) New ceramics-related industry
implicated in elevated blood lead levels in children. Arch Environ
Health, 42: 161-164.
Kazantzis G (1989) Lead: ancient metal - modern menace. In: Smith MA,
Grant LD, & Sors AI ed. Lead exposure and child development: An
international assessment. Dordrecht, London, Kluwer Academic Press,
pp 119-128.
Kehoe RA (1961) The metabolism of lead in man in health and disease:
The normal metabolism of lead (The Harben Lectures, 1960). J R Inst
Public Health Hyg, 24: 81-97.
Kehoe RA (1987) Studies of lead administration and elimination in
adult volunteers under natural and experimentally induced conditions
over extended periods of time. Food Chem Toxicol, 25: 425-493.
Keller CA & Doherty RA (1980a) Distribution and excretion of lead in
young and adult female mice. Environ Res, 21: 217-228.
Keller CA & Doherty RA (1980b) Bone lead mobilization in lactating
mice and lead transfer to suckling offspring. Toxicol Appl Pharmacol,
55: 220-228.
Kerin Z (1973) Lead in new-fallen snow near a lead smelter. Arch
Environ Health, 26: 256-260.
Kholil-Manesh F, Gonich HC, Cohen AH, Alinovi R, Berganaschi E, Mutti
A, & Rosen VJ (1992) Experimental model of lead nephrotoxicity. I.
Continuous high-dose lead administration. Kidney Int, 41: 1192-1203.
Kimber I, Stonard MD, Gidlow DA, & Niewola Z (1986) Influence of
chronic low-level exposure to lead on plasma immunoglobulin
concentration and cellular immune function in man. Int Arch Occup
Environ Health, 57: 117-125.
King E & Conchie D (1979) Industrial lead absorption. Ann Occup Hyg,
22: 213-239.
Kirkby H & Gyntelberg F (1985) Blood pressure and other cardiovascular
risk factors of long term exposure to lead. Scand J Work Environ
Health, 11: 15-19.
Kocak R, Anarat A, Altintas G, & Evilyaoglu N (1989) Lead poisoning
from contaminated flour in a family of 11 members. Hum Toxicol,
8: 385-386.
Kolbye AC, Mahaffey R, Fiorino A, Corneliussen PC, & Jelinek CF (1974)
Food exposures to lead. Environ Health Perspect, 7(Exp Issue): 65-75.
Koller LD (1985) Immunological effects of lead. In: Mahaffey KR ed.
Dietary and environmental lead: Human health effects. Amsterdam,
Oxford, New York, Elsevier Science Publishers, pp 339-354.
Koller LD & Roan JG (1980) Effect of lead, cadmium and methylmercury
on immunologic memory. J Environ Pathol Toxicol, 4: 47-52.
Koo WWK, Succop PA, Bornschein RL, Krug-Wispe SK, Steichen JJ, Tsang
RC, Hammond PB, & Berger OG (1991) Serum vitamin D metabolites and
bone mineralization in young children with chronic to moderate lead
exposure. Pediatrics, 87: 680-687.
Kopp SJ, Barron JT, & Tow JP (1988) Cardiovascular actions of lead and
relationship to hypertension: a review. Environ Health Perspect,
89: 91-99.
Koren G, Chang N, Gonen R, Klein J, Weiner L, Demshar H, Pizzolato S,
Radde I, & Shime J (1990) Lead-exposure among mothers and their
newborns in Toronto. Can Med Assoc J, 142: 1241-1244.
Kostial K & Kello D (1979) Bioavailability of lead in rats fed "human"
diets. Bull Environ Contam Toxicol, 21: 312-314.
Kostial K, Kello D, & Jugo S (1978) Influence of age on metal
metabolism and toxicity. Environ Health Perspect, 25: 81-86.
Kovar IZ, Strehlow CD, Richmond J, & Thompson MG (1984) Perinatal lead
and cadmium burden in a British urban population. Arch Dis Child,
59: 36-39.
Krueger JA & Duguay KM (1989) Comparative analysis of lead in Maine
urban soils. Bull Environ Contam Toxicol, 42: 574-581.
Kusell M, Lake L, Anderson M, & Gerschenson LE (1978) Cellular and
molecular toxicology of lead. II. Effect of lead on d-aminolevulinc
acid synthelase of cultured cells. J Toxicol Environ Health,
4: 515-525.
Kutbi II, Ahmed M, & Saber A (1989) Measurement of blood-lead levels
in school children of Jeddah Saudi Arabia and assessment of sub-toxic
levels of lead on some sensitive hematological parameters. J Environ
Sci Health, A24: 943-955.
Lacey RF, Moore MR, & Richards WN (1985) Lead in water, infant diet
and blood: The Glasgow duplicate diet study. Sci Total Environ,
41: 235-257.
Lai CS (1972) Lead poisoning as an occupational hazard in Chinese
opera actors: a case report. Singap Med J, 13: 115-117.
Landis JR & Flegal KM (1988) A generalized Mantel-Haenszel analysis of
the regression of blood pressure on blood lead using NHANES II data.
Environ Health Perspect, 78: 35-41.
Landrigan PJ, Gehlbach SH, Rosenblum BF, Shoults JM, Cancelaria RM,
Barthel WF, Liddle JA, Smrek AL, Staehling NW, & Sanders JF (1975)
Epidemic lead absorption near an ore smelter; The role of particulate
lead. N Engl J Med, 292: 123-129.
Lansdown RG, Shepherd J, Clayton BE, Delves HT, Graham PJ, & Turner WC
(1974) Blood-lead levels, behaviour, and intelligence: a population
study. Lancet, 1(7857): 538-541.
Lansdown R, Yule W, Urbanowicz MA, & Hunter J (1986) The relationship
between blood-lead concentrations, intelligence, attainment and
behaviour in a school population: the second London study. Int Arch
Occup Environ Health, 57: 225-235.
Laurs AJ (1976) Review of European test methods for measuring lead and
cadmium release. In: Proceedings of the International Conference on
Ceramic Foodware Safety, Geneva, 1974. New York, Lead Industries
Association Inc., pp 27-36.
Lauwerys R, Buchet JP, Roels HA, & Materne DD (1974) Relationship
between urinary delta-aminolevulinic acid excretion and the inhibition
or red cell delta-aminolevulinate dehydratase by lead. Clin Toxicol,
7: 383-388.
Lavelle JM, Poppenga RH, Thacker BJ, Geisy JP, Weis C, Othoudt R, &
Vandervoort C (1991) Bioavailability of lead in mining wastes: An oral
intubation study in young swine. Chem Speciation Bioavailab,
3: 105-111.
Laxen DPH, Raab GM, & Fulton M (1987) Children's blood lead and
exposure to lead in household dust and water - a basis for an
environmental standard for lead in dust. Sci Total Environ,
66: 235-244.
Lee RE & Goranson S (1972) National air surveillance cascade impact or
network. I. Size distribution measurements of suspended particulate
matter in air. Environ Sci Technol, 6: 1019-1024.
Leeming RJ & Blair JA (1980) The effect of pathological and normal
physiological processes on biopterin levels in man. Clin Chim Acta,
108: 102-111.
Lepow ML, Bruckman L, Rubino RA, Markowitz S, Gillette M, & Kapish J
(1974) Role of airborne lead in increased body burden of lead in
Hartford children. Environ Health Perspect, 7: 99-102.
Lerda D (1992) Study of sperm characteristics in persons
occupationally exposed to lead. Am J Ind Med, 22: 567-571.
Levin ED & Bowman RE (1983) The effect of pre- or postnatal lead
exposure on Hamilton search task in monkeys. Neurobehav Toxicol
Teratol, 5: 391-394.
Levin ED & Bowman RE (1986) Long-term lead effects on the Hamilton
Search Test and delayed alternation in monkeys. Neurobehav Toxicol
Teratol, 8: 219-224.
Levin ED & Bowman RE (1989) Long-term effects of chronic post-natal
lead exposure on delayed spatial alternation in monkeys. Neurotoxicol
Teratol, 10: 505-510.
Lilienthal H, Winneke G, Brockhaus A, & Molik B (1986) Pre- and
postnatal lead-exposure in monkeys: Effects on activity and learning
set formation. Neurobehav Toxicol Teratol, 8: 265-272.
Lilienthal H, Winneke G, & Ewert T (1990) Effects of lead on
neurophysiological and performance measures: animal and human data.
Environ Health Perspect, 89: 21-25.
Lilis R (1981) Long-term occupational lead exposure, chronic
nephropathy, and renal cancer: a case report. Am J Ind Med,
2: 293-297.
Lilis R, Fischbein A, Eisinger J, Blumberg WE, Diamond S, Anderson HA,
Rom W, Rice C, Sarkozi L, Kon S, & Selikoff IJ (1977) Prevalence of
lead disease among secondary lead smelter workers and biological
indicators of lead exposure. Environ Res, 14: 255-285.
Lilley SG, Florence TM, & Stauber JL (1988) The use of sweat to
monitor lead absorption through the skin. Sci Total Environ,
76: 267-278.
Lind B, Vahter M, Rahnster B, & Björs U (1988) Quality control samples
for the determination of lead and cadmium in blood, feces, air
filters, and dust. Fresenius Z Anal Chem, 332: 741-743.
Lindholm ML, Sallmen M, Anttila A, Taskinen H, & Hemminki K (1991)
Paternal occupational lead exposure and spontaneous abortion. Scand J
Work Environ Health, 17(2): 95-103.
Lin-Fu JS (1985) Historical perspective on health effects of lead. In:
Mahaffey KR ed. Dietary and environmental lead: Human health effects.
Amsterdam, Oxford, New York, Elsevier Science Publishers, pp 43-64.
Lovering TG ed. (1976) Lead in the environment. Washington, DC, US
Department of the Interior, Geological Survey (Geological Survey
Professional Paper No. 957).
Lyngbye T, Hansen OL, Trillingsgaard A, Beese I, & Grandjean P (1990a)
Learning disabilities in children: significance of low-level
lead-exposure and confounding factors. Acta Paediatr Scand,
79: 352-360.
Lyngbye T, Jorgensen PJ, & Grandjean P (1990b) Validity and
interpretation of blood lead levels: a study of Danish
school-children. Scand J Clin Lab Invest, 50: 441-449.
McGrath SP (1986) The range of metal concentrations in top soils of
England and Wales in relation to soil protection guidelines. In
Hemphill DD ed. Trace substances in environmental health, vol 20.
Columbia, Missouri, University of Missouri, pp 242-252.
McGregor AJ & Mason MJ (1990) Chronic occupational lead exposure and
testicular endocrine function. Hum Exp Toxicol, 9: 371-376.
McIntosh MJ, Meredith PA, Moore MR, & Goldberg A (1985) Neurotoxic
action of lead: effect on tetrahydrobiopterin metabolism in the rat.
Comp Biochem Physiol, 81C(1): 227-231.
McIntosh MJ, Meredith PA, Moore MR, & Goldberg A (1989) Action of lead
on neurotransmission in rats. Xenobiotica, 19(1): 101-113.
McKhann CF & Vogt EC (1933) Lead poisoning in children. J Am Med
Assoc, 101: 1131-1135.
McMichael AJ & Johnson HM (1982) Long-term mortality profile of
heavily-exposed lead smelter workers. J Occup Med, 24: 375-378.
McMichael AJ, Vimpani GV, Robertson EF, Baghurst PA, & Clark PD (1986)
The Port Pirie cohort study: maternal blood lead and pregnancy
outcome. J Epidemiol Community Health, 41: 18-25.
McMichael AJ, Baghurst PA, Wigg NR, Vimpani GV, Robertson EF, &
Roberts RJ (1988) Port Pirie cohort study: environmental exposure to
lead and children's abilities at the age of four years. N Engl J Med,
319: 468-475.
Maenhaut W, Zoller WH, Duce RA, & Hoffman GL (1979) Concentration and
size distribution of particulate trace elements in the south polar
atmosphere. J Geophys Res, 84: 2421-2431.
Mahaffey KR (1985) Factors modifying susceptibility to lead toxicity.
In: Mahaffey KR ed. Dietary and environmental lead: Human health
effects. Amsterdam, Oxford, New York, Elsevier Science Publishers,
pp 373-419.
Mahaffey KR & Annest JL (1986) Association of erythrocyte
protoporphyrin with blood lead level and iron status in the Second
National Health and Nutrition Examination Survey, 1976-1980. Environ
Res, 41: 327-338.
Mahaffey-Six KR & Goyer RA (1972) The influence of iron deficiency on
tissue content and toxicity of ingested lead in the rat. J Lab Clin
Med, 79: 128-136.
Mahaffey KR, Rosen JF, Chesney RW, Peeler JT, Smith CM, & Debuca HF
(1982) Association between age, blood lead concentration, and serum
1,25-dihydroxy-cholecalciferol levels in children. Am J Clin Nutr,
35: 1327-1331.
Maines MD (1992) Haem oxygenase-clinical applications and functions.
Boca Raton, Florida, CRC Press.
Maki-Paakkanen J, Sorsa M, & Vainio H (1981) Chromosome aberrations
and sister chromatid exchanges in lead-exposed workers. Hereditas,
94: 269-275.
Manton WI (1985) Total contribution of airborne lead to blood lead. Br
J Ind Med, 42: 168-172.
Manton WI & Cook JD (1984) High accuracy (stable isotope dilution)
measurements of lead in serum and cerebrospinal fluid. Br J Ind Med,
41: 313-319.
Manton WI & Thal ER (1986) Lead poisoning from retained missiles: an
experimental study. Ann Surg, 204: 594-599.
Mao P & Molnar JJ (1967) The fine structure and histochemistry of
lead-induced renal tumors in rats. Am J Pathol, 50: 571-603.
Maranelli G & Apostoli P (1987) Assessment of renal function in lead
poisoned workers. In: Occupational and environmental chemical hazards:
Cellular and biochemical indices for monitoring toxicity. Chichester,
Ellis Horwood Ltd, chapter 42, pp 344-348.
Marcus AH (1985a) Testing alternative nonlinear kinetic models in
compartmental analysis. In: Eisenfield J & Delisi C ed. Mathematics
and computers in biomedical applications. Amsterdam, Oxford, New York,
Elsevier Science Publishers, pp 259-267.
Marcus AH (1985b) Multicompartment kinetic models for lead: Part I.
bone kinetics and variable absorption in humans without excessive lead
exposures. Environ Res, 36: 441-458.
Marcus AH (1985c) Multicompartment kinetic models for lead: Part II.
linear kinetics and variable absorption in humans without excessive
lead exposures. Environ Res, 36: 459-472.
Marcus AH & Schwartz J (1987) Dose-response curves for erythrocyte
protoporphyrin vs blood lead: effects of iron status. Environ Res,
44: 221-227.
Marino PE, Franzblau A, Lilis R, & Landrigan PJ (1989) Acute lead
poisoning in construction workers: the failure of protective
standards. Arch Environ Health, 44: 140-145.
Marks SC & Popoff SN (1988) Bone cell biology: the regulation of
development, structure, and function in the skeleton. Am J Anat,
183: 1-44.
Marlowe M & Errrera J (1982) Low lead levels and behaviour problems in
children. Behav Disord, 7: 163-172.
Matte TD, Figueroa JP, Ostrowski S, Burr G, Jackson-Hunt L, Kennylside
RA, & Baker EL (1989a) Lead poisoning among household members exposed
to lead-acid battery repair shops in Kingston, Jamaica. Int J
Epidemiol, 18: 874-881.
Matte TD, Figueroa JP, Burr G, Flesch JP, Keenlyside RA & Baker EL
(1989b) Lead exposure among lead-acid battery workers in Jamaica. Am J
Ind Med, 16: 167-177.
Meredith PA & Moore MR (1979) The influence of lead on haem
biosynthesis and biodegradation in the rat. Biochem Soc Trans,
7: 637-639.
Meredith PA, Moore MR, Campbell BC, Thompson GG, & Goldberg A (1978)
Delta-aminolevulinic acid metabolism in normal and lead-exposed
humans. Toxicology, 9: 1-9.
Merwin BW (1976) Review of US standards on test methods for lead and
cadmium release. In: Proceedings of the International Conference on
Ceramic Foodware Safety, Geneva, 1974. New York, Lead Industries
Associate Inc., pp 36-40.
Meyer BR, Fishbein A, Rosemann K, Lerman Y, Drayer DE, & Reidenberg MM
(1984) Increased urinary enzyme excretion in workers exposed to
nephrotoxic chemicals. Am J Med, 76: 989-998.
Michaelson A & Sauerhoff MW (1974) An improved model of lead induced
brain dysfunction in the suckling rat. Toxicol Appl Pharmacol,
28: 88-96.
Mielke H, Blake B, Burroughs S, & Hassinger N (1984) Urban lead levels
in Minneapolis: the case of the Hmong children. Environ Res,
34: 64-76.
Mitchell DG, Aldous KM, & Ryan FJ (1974) Mass screening for lead
poisoning, capillary blood sampling and automated Delves cup atomic
absorption analysis. NY State J Med, 74: 1599-1603.
Mitchell-Heggs CAW, Conway M, & Cassar J (1990) Herbal medicine as a
cause of combined lead and arsenic poisoning. Hum Exp Toxicol,
9: 195-196.
Molina-Ballesteros G, Zuniga-Charles MA, Ortega AC, & Solis-Camara P
(1983) Lead concentrations in the blood of children from
pottery-making families exposed to lead salts in a Mexican village.
Bull Panam Health Organ, 17: 35-41.
Möller L & Kristensen TS (1992) Blood lead as a cardiovascular risk
factor. Am J Epidemiol, 136: 1091-1100.
Moore MR (1983) Lead exposure and lead plumbosolvency. In: Rutter M &
Jones RR ed. Lead versus health. London, John Wiley and Sons,
pp 79-106.
Moore MR & Meredith PA (1979) The carcinogenicity of lead. Arch
Toxicol, 42: 87-94.
Moore MR, Meredith PA, Campbell BC, & Watson SW (1979) The
gastrointestinal absorption of lead 203 chlorine in man. In: Hemphill
DD ed. Trace substances in environmental health - XIII. Proceedings of
the 13th Annual Conference on Trace Substances in Environmental
Health, Columbia, June 1979. Columbia, Missouri, University of
Missouri, pp 368-373.
Moore MR, Meredith PA, & Goldberg A (1980) Lead and heme biosynthesis.
In: Singhal PL & Thomas JA ed. Lead toxicity. Baltimore, Maryland,
Urban and Schwarzenberg, Inc., pp 79-118.
Moore MR, Goldberg A, & Pocock SJ (1982) Some studies of maternal and
infant lead exposure in Glasgow. Scott Med J, 27: 113-122.
Moore MR, Bushnell IWR, & Goldberg A (1989) A prospective study of the
results of changes in environmental lead exposure in children in
Glasgow. In: Smith MA, Grant LD, & Sors AI ed. Lead exposure and child
development: An international assessment. Dordrecht, London, Kluwer
Academic Publishers, pp 371-378.
Moore MR, Goldberg A, & Young-Laiwah AAC (1987) Lead effects on the
haem-biosynthetic pathway. Relationship to toxicity. Ann NY Acad Sci,
514: 191-203.
Mooty J, Ferrand CF, & Harris P (1975) Relationship of diet to lead
poisoning in children. Pediatrics, 55: 636-639.
Moreau T, Orssaud G, Juguet B, & Busquet G (1982) Plombémie et
pression artérielle: premiers résultats d'une enquête transversale de
431 sujets de sexe masculin. Rev Epidemiol Santé Publique,
30: 395-397.
Morgan A & Holmes A (1978) The fate of lead in petrol-engine exhaust
particulates inhaled by the rat. Environ Res, 15: 44-56.
Muijser H, Hoogendijk EMG, Hooisma J, & Twisk DAM (1987) Lead exposure
during demolition of a steel structure coated with lead-based paints.
II. Reversible changes in the conduction velocity of the motor nerves
in transiently exposed workers. Scand J Work Environ Health,
13: 56-61.
Munoz C, Garbe K, Lilienthal H, & Winneke G (1986) Persistence of
retention deficit in rats after neonatal lead exposure.
Neurotoxicology, 7(2): 569-580.
Muramatsu Y & Parr RM (1985) Survey of currently available reference
materials for use in connection with the determination of trace
elements in biological and environmental materials. Vienna,
International Atomic Energy Agency (Report No. IAEA/RL/128).
Murata K & Araki S (1991) Autonomic nervous system dysfunction in
workers exposed to lead, zinc, and copper in relation to peripheral
nerve conduction: a study of R-R interval variability. Am J Ind Med,
20: 663-671.
Murphy MJ, Graziano JH, Popovac D, Kline JK, Mehmeti A, Factor-Litvak
P, Ahmadi G, Shrout P, Rajovic B, Nenezic DU, & Stein ZA (1990) Past
pregnancy outcomes among women living in the vicinity of a lead
smelter in Kosovo, Yugoslavia. Am J Public Health, 80: 33-35.
Mushak P (1989) Biological monitoring of lead exposure in children:
overview of selected biokinetic and toxicological issues. In: Smith
MA, Grant LD, & Sors AI ed. Lead exposure and child development: An
international assessment. Dordrecht, London, Kluwer Academic
Publishers, pp 129-145.
Mushak P & Crocetti AF (1989) Determination of numbers of lead-exposed
American children as a function of lead source: Integrated summary of
a report to the U.S. Congress on childhood lead poisoning. Environ
Res, 50: 210-299.
Mussalo-Rauhamaa H, Salmela SS, Leppanen A, & Pyysalo H (1986)
Cigarettes as a source of some trace and heavy metals and pesticides
in man. Arch Environ Health, 41: 49-55.
NAS-NRC (1972) Airborne lead in perspective. Washington, DC, National
Academy of Sciences.
Needleman HL & Gatsonis CA (1990) Low-level lead exposure and the IQ
of children: a meta-analysis of modern studies. J Am Med Assoc,
263: 673-678.
Needleman HL, Tuncay OC, & Shapiro IM (1972) Lead levels in deciduous
teeth of urban and suburban children. Nature (Lond), 235: 111-112.
Needleman HL, Gunnoe C, Leviton A, Reed R, Peresie H, Maher C, &
Barrett P (1979) Deficits in psychologic and classroom performance of
children with elevated dentine lead levels. N Engl J Med,
300: 689-695.
Needleman HL, Schell A, Bellinger D, Leviton A, & Allred EN (1990) The
long-term effects of childhood exposure to low doses of lead: An
11-year follow-up report. N Engl J Med, 322: 83-88.
Neri LC, Hewitt D, & Orser B (1988) Blood lead and blood pressure:
analysis of cross-sectional and longitudinal data from Canada. Environ
Health Perspect, 78: 123-126.
Nerin C, Olavide S, & Cacho J (1989) Determination of lead in airborne
particulate by hydride generation. Water Air Soil Pollut, 44: 339-345.
Neubecker TA & Allen HE (1983) The measurement of complexation
capacity and conditional stability constants for ligands in natural
waters. Water Res, 17: 1-14.
NFA (Australian National Food Authority, National Health and Medical
Council) (1991) The 1990 Australian market basket survey report.
Canberra, Commonwealth of Australia, Australian Government Publishing
Service.
Ng A & Patterson CC (1982) Changes of Pb and Ba with time in
California offshore basin sediments. Geochim Cosmochim Acta,
46: 2307-2322.
Ng TP, Goh HH, Ng YL, Ong CN, Chia KS, Chia SE, & Jeyaratnam J (1991)
Male endocrine functions in workers with moderate exposure to lead. Br
J Ind Med, 48: 485-491.
Nielsen T (1984) Atmospheric occurrence of organolead compounds. In:
Grandjean P ed. Biological effects of organolead compounds. Boca
Raton, Florida, CRC Press, pp 43-62.
Nilsson U, Attewell R, Christoffersson J-O, Schütz A, Ahlgren L,
Skerfving S, & Mattsson S (1991) Kinetic of lead in bone and blood
after end of occupational exposure. Pharmacol Toxicol, 69: 477-484.
NIOSH (1977a) Method No. P&CAM 102. In: Manual of analytical methods,
vol 1, 2nd ed. Cincinnati, Ohio, National Institute for Occupational
Safety and Health.
NIOSH (1977b) Method No. P&CAM 191. In: Manual of analytical methods,
vol 1, 2nd ed. Cincinnati, Ohio, National Institute for Occupational
Safety and Health, pp 191/1-191/9.
NIOSH (1977c) Method No. P&CAM 195. In: Manual of analytical methods,
vol 1, 2nd ed. Cincinnati, Ohio, National Institute for Occupational
Safety and Health.
NIOSH (1977d) Method No. P&CAM 200. In: Manual of analytical methods,
vol 1, 2nd ed. Cincinnati, Ohio, National Institute for Occupational
Safety and Health, pp 200/1-200/8.
NIOSH (1977e) Method No. P&CAM 214. In: Manual of analytical methods,
vol 1, 2nd ed. Cincinnati, Ohio, National Institute for Occupational
Safety and Health, pp 214/1-214/6.
NIOSH (1981) Method No. P&CAM 351. In: Manual of analytical methods,
vol 7. Cincinnati, Ohio, National Institute for Occupational Safety
and Health, pp 351/1-351/11.
Nordenson I, Beckman G, Beckman L, & Nordstrom S (1978) Occupational
and environmental risks in and around a smelter in northern Sweden:
IV. Chromosomal aberrations in workers exposed to lead. Hereditas,
88: 263-267.
Nordstrom S, Beckman L, & Nordenson I (1978a) Occupational and
environmental risks in and around a smelter in northern Sweden: I.
Frequencies in birth weight. Hereditas, 88: 43-46.
Nordstrom S, Beckman L, & Nordenson I (1978b) Occupational and
environmental risks in and around a smelter in northern Sweden: III.
Frequencies of spontaneous abortion. Hereditas, 88: 51-54.
Nordstrom S, Beckman L, & Nordenson I (1979) Occupational and
environmental risks around a smelter in northern Sweden: V.
Spontaneous abortion among female employees and decreased birth weight
in their offspring. Hereditas, 90: 291-296.
Nosal RM & Wilhelm WJ (1990) Lead toxicity in the shipbreaking
industry: the Ontario experience. Can J Public Health, 81: 259-262.
Novotny T, Cook M, Hughes J, & Lee SA (1987) Lead exposure in a firing
range. Am J Public Health, 77: 1225-1226.
Nriagu J (1979) Global inventory of natural and anthropogenic
emissions of trace metals to the atmosphere. Nature (Lond),
279: 409-411.
Nriagu J (1989) A global assessment of natural sources of atmospheric
trace metals. Nature (Lond), 338: 47-49.
Nriagu J & Pacyna JM (1988) Quantitative assessment of worldwide
contamination of air, water, and soils by trace metals. Nature (Lond),
333: 134-139.
OCTEL (1991) Worldwide survey of motor gasoline quality. London,
Associated Octel Company Ltd (Octel Promotional Publication No. 4).
OECD (1993) Risk Reduction Monograph No. 1: Lead background and
national experience with reducing risk. Paris, Organisation for
Economic Co-operation and Development, 277 pp (Report No.
OCDE/GD(93)67).
O'Flaherty EJ (1991) Physiologically based lead kinetics. Trace Subst
Environ Health, 24: 44-54.
O'Flaherty EJ (1993) Physiologically based models for bone-seeking
elements. IV. Kinetics of lead disposition in humans. Toxicol Appl
Pharmacol, 118: 16-29.
O'Flaherty EJ, Hammond PB, Lerner SI, Hanenson IB, & Roda SMB (1980)
The renal handling of delta-aminolevulinic acid in the rat and in the
human. Toxicol Appl Pharmacol, 55: 423-432.
O'Flaherty EJ, Hammond PB, & Lerner SI (1982) Dependence of apparent
blood lead half-life on the length of previous lead exposure in
humans. Fundam Appl Toxicol, 2: 49-54.
Olejnik D, Walkowska A, Wisnilwska J, & Ziembinski R (1985)
[Evaluation of the daily dose of mercury, lead and cadmium in the diet
of certain population groups.] Roczn Pzh, XXXVI: 9-21 (in Polish, with
English abstract).
Olson KW & Skogerboe RK (1975) Identification of soil lead compounds
from automotive sources. Environ Sci Technol, 9: 227-230.
Ong CN & Lee WR (1980) High affinity of lead for fetal hemoglobin. Br
J Ind Med, 37: 292-298.
Ong CN, Phoon WO, Law HY, Tye CY, & Lim HH (1985) Concentration of
lead in maternal blood, cord blood, and breast milk. Arch Dis Child,
60: 576-759.
Ong CN, Endo G, & Chia KS (1987) Evaluation of renal function in
workers with low blood lead levels. In: Occupational and environmental
chemical hazards: Cellular and biochemical indices for monitoring
toxicity. Chichester, Ellis Horwood Ltd, chapter 39, pp 327-333.
Orfila MP (1814) Traité des poisons. Paris, Crochard, vol 2.
Orssaud G, Claude JR, Moreau T, Lellouch J, Juguet B, & Festy B (1985)
Blood lead concentration and blood pressure. Br Med J, 290: 244.
Padilla F, Shapiro AP, & Jensen WN (1969) Effect of chronic lead
intoxication on blood pressure in the rat. Am J Med Sci, 258: 359-365.
Paglia DE, Valentine WN, & Dahlgren JG (1975) Effects of low-level
lead exposure on pyrimidine 5'-nucleotidase and other erythrocyte
enzymes: possible role of pyrimidine 5'-nucleotidase in the
pathogenesis of lead-induced anaemia. J Clin Invest, 56: 1164-1169.
Pagliuca A, Mufti GJ, Baldwin D, Lestas AN, Wallis RM, & Bellingham AJ
(1990) Lead poisoning: clinical, biochemical, and haematological
aspects of a recent outbreak. J Clin Pathol, 43: 277-281.
Parfitt AM (1990) Pharmacologic manipulation of bone remodelling and
calcium homeostasis: Calcium metabolism. Prog Basic Clin Pharmacol,
4: 1-27.
Parkinson DK, Ryan C, Brommet EJ, & Connell MM (1986) A psychiatric
epidemiologic study of occupational lead exposure. Am J Epidemiol,
123: 261-269.
Parkinson DK, Ryan C, Bromet J, & Connell MM (1987) A psychiatric
epidemiological study of occupationally exposure. Am J Epidemiol,
123: 261-269.
Patterson C (1965) Contaminated and natural lead environments of man.
Arch Environ Health, 11: 344-360.
Patterson CC (1983) Criticism of "flow of metals into the global
atmosphere". Geochim Cosmochim Acta, 47: 1163-1168.
Patterson CC & Settle DM (1976) The reduction of orders of magnitude
errors in lead analyses of biological materials and natural waters by
evaluating and controlling the extent and source of industrial lead
contamination introduced during sample collecting, handling, and
analyses. In: LaFleur PD ed. Accuracy in trace analysis: Sampling,
sample handling and analysis. Volume 1. Proceedings of the 7th
Materials Research Symposium, Gaithersburg, Maryland, October, 1974.
Washington, DC, US Department of Commerce, National Bureau of
Standards, pp 321-352 (NBS Special Publication No. 422).
Pekkarinen M (1970) Methodology in the collecting of food consumption
data. World Rev Nutr Diet, 12: 145-170.
Pentschew A (1965) Morphology and morphogenesis of lead
encephalopathy. Acta Neuropathol, 5: 133-160.
Pentschew A & Garro F (1966) Lead encephalo-myelopathy of the suckling
rat and its implications on the porphyrinopathic nervous diseases with
special reference to the permeability/disorders of the nervous
system's capillaries. Acta Neuropathol, 6: 266-278.
Perino J & Ernhart CB (1974) The relation of subclinical lead level to
cognitive and sensorimotor impairment in black preschoolers. J Learn
Disabil, 7: 616-620.
Pickston L, Brewerton HV, Drysdale JM, Hughes JT, Smith JM, Love JL,
Sutcliffe ER, & Davidson F (1985) The New Zealand diet: a survey of
elements, pesticides, colours and preservatives. N Z J Technol,
1: 81-89.
Piomelli S, Seaman C, Zullow D, Curran A, & Davidow B (1982) Threshold
for lead damage to heme synthesis in urban children. Proc Natl Acad
Sci (USA), 79: 3335-3339.
Piper W & Tephly T (1974) Differential inhibition of erythrocyte and
hepatic uroporphyrinogen. I. Synthelase activity by lead. Life Sci,
14: 873-876.
Pirkle JL, Schwartz J, Lamdis JR, & Harlan WR (1985) The relationship
between blood lead levels and blood pressure and its cardiovascular
risk implications. Am J Epidemiol, 121: 246-258.
Pocock SJ, Shaper AG, Walker M, Wale CJ, Clayton B, Delves T, Lacey
RF, Packham RF, & Powell P (1983) Effects of tap water lead, water
hardness, alcohol, and cigarettes on blood lead concentrations. J
Epidemiol Community Health, 37: 1-7.
Pocock SJ, Shaper AG, Ashby D, Delves T, & Whitehead TP (1984) Blood
lead concentration, blood pressure, and renal function. Br Med J,
289: 872-874.
Pocock S, Ashby D, & Smith MA (1987) Lead exposure and children's
intellectual performance. Int J Epidemiol, 16: 57-67.
Pocock SJ, Shaper AG, Ashby D, Delves HT, & Clayton BE (1988) The
relationship between blood lead, blood pressure, stroke and heart
attacks in middle-aged British men. Environ Health Perspect,
78: 23-30.
Pocock SJ, Ashby D, & Smith MA (1989) Lead exposure and children's
intellectual performance. The Institute of Child Health/Southampton
Study. In: Smith M, Grant LD, & Sors A ed. Lead exposure and child
development: An international assessment. Dordrecht, London, Kluwer
Academic Press, pp 149-165.
Pollock CA & Ibels LS (1986) Lead intoxication in paint removal
workers on the Sydney Harbour Bridge. Med J Aust, 145: 635-639.
Ponka A, Salminen E, & Ahonen S (1991) Lead in the ambient air and
blood specimens of children in Helsinki. Sci Total Environ, 138(1-3):
301-308.
Pontifax AH & Garg AK (1985) Lead poisoning from an Asian Indian folk
remedy. Can Med Assoc J, 133: 1227-1228.
Poole C, Smythe LE, & Alpers M (1980) Blood lead levels of Papua New
Guinea children living in a remote area. Sci Total Environ, 15: 17-24.
Pounds JG (1984) Effect of lead intoxication on calcium homeostasis
and calcium-mediated cell function: a review. Neurotoxicology,
5: 295-332.
Pounds JG, Marlar RJ, & Allen JR (1978) Metabolism of lead-210 in
juvenile and adult rhesus monkeys Macaca mulatta. Bull Environ Contam
Toxicol, 19: 684-691.
Pounds JG, Long GJ, & Rosen JF (1991) Cellular and molecular toxicity
of lead in bone. Environ Health Perspect, 91: 17-32.
Purchase NG & Fergusson JE (1986) Lead in teeth: the influence of the
tooth type and the sample within a tooth on lead levels. Sci Total
Environ, 52: 239-250.
Qazi QH, Madahar C, & Huceoglu AM (1980) Temporary increase in
chromosome breakage in an infant prenatally exposed to lead. Hum
Genet, 53: 201-203.
Qian ZM & Morgan EH (1990) Effect of lead on the transport of
transferrin-free and transferrin-bound iron into rabbit reticulocytes.
Biochem Pharmacol, 40: 1049-1054.
Quarterman J & Morrison JN (1975) The effects of dietary calcium and
phosphorus on the retention and excretion of lead in rats. Br J Nutr,
34: 351-362.
Que Hee SS & Boyle JR (1988) Simultaneous multielemental analysis of
some environmental and biological samples by inductively coupled
plasma atomic emission spectrometry. Anal Chem, 60: 1033-1042.
Que Hee SS, MacDonald TJ, & Bornschein RL (1985a) Blood lead by
furnace-Zeeman atomic absorption spectrophotometry. Microchem J,
32: 55-63.
Que Hee SS, Peace B, Clark CS, Boyle JR, Bornschein RL, & Hammond PB
(1985b) Evolution of efficient methods to sample lead sources, such as
house dust and hand dust in the homes of children. Environ Res,
38: 77-95.
Raab GM, Laxen DPH, & Fulton M (1987) Lead from dust and water as
exposure sources for children. Environ Geochem Health, 9: 80.
Rabinowitz MB (1991) Toxicokinetics of bone lead. Environ Health
Perspect, 91: 33-37.
Rabinowitz M & Needleman HL (1982) Temporal trends in the lead
concentrations of umbilical cord blood. Science, 216: 1429-1431.
Rabinowitz MB, Wetherill G, & Kopple J (1975) Absorption, storage and
excretion of lead by normal humans. In: Proceedings of the Ninth
Annual Conference on Trace Substances in Environmental Health.
Columbia, Missouri, University of Missouri, pp 361-368.
Rabinowitz MB, Wetherill GM, & Kopple JD (1976) Kinetics analysis of
lead metabolism in healthy humans. J Clin Invest, 58: 260-270.
Rabinowitz MB, Wetherill GW, & Kopple JD (1977a) Magnitude of lead
intake from respiration by normal man. J Lab Clin Med, 90: 238-248.
Rabinowitz MB, Wetherill GW, & Kopple JD (1977b) Effect of food intake
and fasting on gastrointestinal lead absorption in humans. Am J Clin
Nutr, 33: 1784-1788.
Rabinowitz MB, Kopple JD, & Wetherill GW (1980) Effect of food intake
and fasting on gastrointestinal lead absorption in humans. Am J Clin
Nutr, 33: 1784-1788.
Rabinowitz MB, Leviton A, & Needleman HL (1986) Occurrence of elevated
protoporphyrin levels in relation to lead burden in infants. Environ
Res, 39: 253-257.
Rabinowitz M, Bellinger D, Leviton A, Needleman H, & Schoenbaum S
(1987) Pregnancy hypertension, blood pressure during labor, and blood
lead levels. Hypertension, 10: 447-451.
Rabinowitz MB, Leviton A, & Bellinger D (1989) Blood lead-tooth lead
relationship among Boston children. Bull Environ Contam Toxicol,
43: 485-492.
Rabinowitz MB, Wang J-D, & Soong WT (1991) Dentine lead and child
intelligence in Taiwan. Arch Environ Health, 46: 351-360.
Ragan HA (1977) Effect of iron deficiency on the absorption of lead
and cadmium in the rat. J Lab Clin Med, 90: 700-706.
Reaves GA & Berrow ML (1984) Total lead concentrations in Scottish
soils. Geoderma, 32: 1-8.
Reisinger K, Stoeppler M, & Nurnberg HW (1981) Evidence for the
absence of biological methylation of lead in the environment. Nature
(Lond), 281: 228-230.
Reuter JH & Perdue EM (1977) Importance of heavy metal-organic matter
interactions in natural waters. Geochim Cosmochim Acta, 41: 325-334.
Rhagavan SRV, Dwight Culver B, & Gonick HC (1981) Erythrocyte
lead-binding protein after occupational exposure. II. Influence of
lead inhibition of membrane Na+, K±-adenosinetriphosphatase. J Toxicol
Environ Health, 7: 561-568.
Rice DC (1984a) Behavioural deficit (delayed matching to sample) in
monkeys exposed from birth to low level lead. Toxicol Appl Pharmacol,
75: 337-345.
Rice DC (1984b) Effect of lead on schedule-controlled behavior in the
monkey. In: Seiden LS & Balster RL ed. Behavioral pharmacology: The
current status. New York, Alan R. Liss, pp 473-486.
Rice DC (1985) Chronic low-lead exposure from both produces deficits
in discrimination reversal monkey's. Toxicol Appl Pharmacol,
77: 201-210.
Rice DC & Karpinski KF (1988) Lifetime dow level lead exposure
produces deficits in delayed alternation in adult monkeys.
Neurotoxicol Teratol, 10: 207-214.
Roberts J, Mahaffey KR, & Annest JL (1985) Blood lead levels in
general populations. In: Mahaffey KR ed. Dietary and environmental
lead: Human health effects. Amsterdam, Oxford, New York, Elsevier
Science Publishers, pp 355-372.
Robins JM, Cullen MR, Connors BB, & Kayne RD (1983) Depressed thyroid
indexes associated with occupational exposure to inorganic lead. Arch
Intern Med, 143: 220-224.
Robinson TR (1974) Delta-aminolevulinic acid and lead in urine of lead
antiknock workers. Arch Environ Health, 28: 133-138.
Rodamilans M, Osaba MJM, To-Figueras J, Fillat FR, Marques JM, Perez
P, & Corbella J (1988) Lead toxicity on endocrine testicular function
in an occupationally exposed population. Hum Toxicol, 7: 125-128.
Rodney BS & Lee MG (1985) Environmental lead hazard in a high-risk
Jamaican population. West Indian Med J, 34: 176-179.
Roels HA & Lauwerys R (1987) Evaluation of dose-effect and
dose-response relationships for lead exposure in different Belgian
population groups (foetus, child, adult men and women). Trace Elem
Med, 4: 80-87.
Roels HA, Buchet J-P, Lauwerys R, Hubermont G, Bruaux P, Claeys-
Thoreau F, Lafontaine A, & van Overschelde J (1976) Impact of air
pollution by lead on the haem biosynthetic pathway in school-age
children. Arch Environ Health, 31: 310-316.
Roels HA, Buchet JP, Lauwerys RR, Bruaux P, Claeys-Thoreau F,
Lafontaine A, & Verduyn G (1980) Exposure to lead by the oral and the
pulmonary routes of children living in the vicinity of a primary lead
smelter. Environ Res, 22: 81-94.
Rosen JF & Chesney RW (1983) Circulating calcitriol concentration in
health and disease. J Pediatr, 103: 1-7.
Rosen JF, Chesney RW, Hamstra AJ, Deluca HF, & Mahaffey KR (1980)
Reduction in 1,25-dihydroxyvitamin D in children with increased lead
absorption. N Engl J Med, 302: 1128-1131.
Rosen JF, Markowitz ME, Bijur PE, Jenks ST, Wielopolski L, Kalef-Ezra
JA, & Slatkin DN (1991) Sequential measurements of bone lead content
by L x-ray fluorescence in CaNa2EDTA-treated lead-toxic children.
Environ Health Perspect, 91: 57-62.
Rosner D & Markowitz G (1985) A "gift of God": The public health
controversy over leaded gasoline during the 1920s. Am J Public Health,
75(4): 344.
Rossouw J, Offermeier J, & van Rooyen J (1987) Apparent central
neurotransmitter receptor changes induced by low-level lead exposure
during different developmental phases in the rat. Toxicol Appl
Pharmacol, 91: 132-139.
Routh DK, Mushak P, & Boone L (1979) A new syndrome of elevated blood
lead and microcephaly. J Pediatr Psychol, 4: 67-76.
Rundle SA & Duggan MJ (1986) Lead pollution from the external
redecoration of old buildings. Sci Total Environ, 57: 181-190.
Ryu JE, Ziegler EE, Nelson SE, & Fomon SJ (1983) Dietary intake of
lead and blood lead concentration in early infancy. Am J Dis Child,
137: 886-891.
Ryu JE, Ziegler EE, Nelson SE, & Fomon SJ (1985) Dietary and
environmental exposure to lead and blood lead during early infancy.
In: Mahaffey KR ed. Dietary and environmental lead: Human health
effects. Amsterdam, Oxford, New York, Elsevier Science Publishers,
pp 187-209.
Saenger P, Markowitz MR, & Rosen JF (1984) Depressed excretion of
6ß-hydroxycortisol in lead-toxic children. J Clin Endocrinol Metab,
58: 363-367.
SAHC (1993) Proceedings of the International Meeting on
Non-Occupational Exposure to Lead, Melbourne, Australia, 5-9 October
1992. Adelaide, South Australian Health Commission, 56 pp.
Sarto F, Stella M, & Acqua A (1978) [Cytogenic studies in 20 workers
occupationally exposed to lead.] Med Lav, 69: 172-180 (in Italian).
Satzger RD, Clow CS, & Bonnin E (1982) Determination of background
levels of lead and cadmium in raw agricultural crops by using
differential pulse anodic stripping voltammetry. J Assoc Off Anal
Chem, 65: 987-991.
Sauk J & Somerman MJ (1991) Physiology of bone: mineral compartment
proteins as candidates for environmental perturbation by lead. Environ
Health Perspect, 91: 9-16.
Schlegel H & Kufner G (1979) Long-term observation of biochemical
effects of lead in human experiments. J Clin Chem Clin Biochem,
17: 225-233.
Schmid E, Bauchinger M, Pietruk S, & Hall G (1972) [Cytogenic action
of lead in human peripheral lymphocytes in vitro and in vivo.]
Mutat Res, 16: 401-406 (in German).
Schmidt U & Huber F (1976) Methylation of organolead and lead(II)
compounds (CH3)4 Pb by microorganisms. Nature (Lond),
259: 157-158.
Schmitt MDC, Trippler DL, & Wachtler JN (1988) Soil lead
concentrations in residential Minnesota as measured by ICP AES. Water
Air Soil Pollut, 39: 157-168.
Schneitzer L, Osborn HH, Bierman A, Mezey A, & Kaul B (1990) Lead
poisoning in adults from renovation of an older home. Ann Emerg Med,
19: 415-420.
Schroeder SR, Hawk B, Otto DA, Mushak P, & Hicks RE (1985) Separating
the effects of lead and social factors on IQ. Environ Res,
38: 144-154.
Schutz AS, Skerfving S, Ranstam J, & Christoffersson JO (1987)
Kinetics of lead in blood after the end of occupational exposure.
Scand J Work Environ Health, 13: 221-231.
Schutz A, Ranstam J, Skerfving S, & Tejning S (1989) Blood-lead levels
in school children in relation to industrial emission and automobile
exhausts. Ambio, 13: 115-117.
Schwartz J (1988) The relationship between blood lead and blood
pressure in the NHANES II survey. Environ Health Perspect, 78: 15-22.
Schwartz J (1993) Low-level lead exposure and children's IQ: a
meta-analysis and search for a threshold. Environ Res, 65: 42-55.
Schwartz J & Otto D (1987) Blood lead, hearing thresholds, and
neurobehavioral development in children and youth. Arch Environ
Health, 42: 153-160.
Schwartz J & Otto D (1991) Lead and minor hearing impairment. Arch
Environ Health, 46: 300-305.
Schwartz J, Angle C, & Pitcher H (1986) Relationship between childhood
blood lead levels and stature. Pediatrics, 77: 281-288.
Schwartz J, Landrigan PL, & Baker EL (1990) Lead-induced anemia:
dose-response relationships and evidence for a threshold. Am J Public
Health, 80: 165-168.
Scott DR, Hemphill DC, & Holboke LE (1976) Atomic absorption and
optical emission analysis of NASN atmospheric particulate samples for
lead. Environ Sci Technol, 9: 877-880.
Selander S & Cramer K (1970) Interrelationships between lead in blood,
lead in urine and ALA in urine during lead work. Br J Ind Med,
27: 28-39.
Selevan SG, Landrigan PJ, Stern FB, & Jones JH (1985) Mortality of
lead smelter workers. Am J Epidemiol, 122: 673-683.
Selevan SG, Landrigan PJ, Stern FB, & Jones JH (1988) Brief report:
lead and hypertension in a mortality study of lead smelter workers.
Environ Health Perspect, 78: 65-66.
Seppäläinen AM & Hernberg S (1980) Subclinical lead neuropathy. Am J
Ind Med, 1: 413-420.
Seppäläinen AM, Hernberg S, Vesanto R, & Kock B (1983) Early
neurotoxic effects of occupational lead exposure: a prospective study.
Neurotoxicology, 4(2): 181-192.
Settle DM & Patterson CC (1980) Lead in albacore: guide to lead
pollution in Americans. Science, 207: 1167-1176.
Shaltout A, Yaish SA, & Fernando N (1981) Lead encephalopathy in
infants in Kuwait. Ann Trop Paediatr (London), 1: 209-215.
Shaper AG, Pocock SJ, Walker M, Cohen NM, Wale CJ, & Thompson AG
(1981) British regional heart study: cardiovascular risk factors in
middle-aged men in 24 towns. Br Med J, 283: 179-186.
Sharma RR, Chandy MJ, & Lad SD (1990) Transient hydrocephalis and
acute lead encephalopathy in neonates and infants. Report of two
cases. Br J Neurosurg, 4: 141-146.
Sherlock J, Smart G, Forbes GI, Moore MR, Patterson WJ, Richards WN, &
Wilson TS (1982) Assessment of lead intakes and dose-response for a
population in Ayr exposed to a plumbsolvent water supply. Hum Toxicol,
1: 115-122.
Sherlock JC, Pickford CJ, & White GF (1986) Lead in alcoholic
beverages. Food Addit Contam, 3: 347-354.
Siegel M, Forsyth B, Siegel L, & Cullen MR (1989) The effect of lead
on thyroid function in children. Environ Res, 49: 190-196.
Silbergeld EK (1991) Lead in bone: implications for toxicology during
pregnancy and lactation. Environ Health Perspect, 91: 63-70.
Silbergeld EK, Schwartz J, & Mahaffey KR (1988) Lead and osteoporosis:
mobilization of lead from bone in postmenopausal women. Environ Res,
47: 79-94.
Silva PA, Hughes P, Williams S, & Faed JM (1988) Blood lead,
intelligence, reading attainment, and behaviour in eleven year old
children in Dunedin, New Zealand. J Child Psychol Psychiatry Appl
Discipl, 29: 43-52.
Sinclair DF & Dohnt BR (1984) Sampling and analysis techniques used in
a blood lead survey of 1341 children in Port Pirie, South Australia.
Clin Chem, 30(10): 1616-1619.
Skerfving S (1988) Biological monitoring of exposure to inorganic
lead. In: Clarkson TW, Friberg L, Nordberg GF, & Sager PR ed.
Biological monitoring of toxic metals. New York, Plenum Press,
pp 169-197 (Rochester Series on Environmental Toxicity).
Skerfving S, Schutz A, & Ranstam J (1986) Decreasing lead exposure in
Swedish children. Sci Total Environ, 58: 225-229.
Skogerboe RK, Hartley AM, Vogel RS, & Koirtyohann SR (1977) Monitoring
for lead in the environment. In: Boggess WR ed. Lead in the
environment. Washington, DC, National Science Foundation, pp 33-70
(NSF Report No. NSF/RA-770214; PB-278278).
Slorach SA, Gustafsson I-B, Jorhem L, & Mattsson P (1983) Intake of
lead, cadmium and certain other metals via a typical Swedish weekly
diet. Var Föda, 35(Suppl 1): 3-16.
Smith MA (1989) The effects of low-level lead exposure on children.
In: Smith MA, Grant LD, & Sors AI ed. Lead exposure and child
development: An international assessment. Dorcrecht, London, Kluwer
Academic Publishers, pp 3-47.
Smith F & Hursh J (1977) Bone storage and release. In: Field J ed.
Handbook of physiology: Reaction to environmental agents. Bethesda,
Maryland, American Physiological Society, section 9, pp 469-482.
Smith M, Delves T, Lansdown R, Clayton B, & Graham P (1983) The
effects of lead exposure on urban children: the Institute of Child
Health/Southampton Study. Dev Med Child Neurol, 47(Suppl): 1-54.
Snee RD (1981) Evaluation of studies of the relationship between blood
lead and air lead. Int Arch Occup Environ Health, 48: 219-242.
Sobel AE, Yuska H, Peters DD, & Kramer B (1940) The biochemical
behaviour of lead. I. Influence of calcium phosphorus and vitamin D on
lead in the blood and bone. J Biol Chem, 132: 239-265.
Sokol RZ (1987) Hormonal effects of lead acetate in the male rat:
Mechanism of action. Biol Reprod, 37: 1135-1138.
Sokol RZ, Madding CE, & Swerdloff RS (1985) Lead toxicity and the
hypothalamic-pituitary-testicular axis. Biol Reprod, 33: 722-728.
Spivey GH, Baloh RW, Brown CP, Browdy BL, Campion DS, Valentine JL,
Morgan DE, & Culver BD (1980) Subclinical effects of chronic increased
lead absorption - a prospective study: III. Neurologic findings at
follow-up examination. J Occup Med, 22: 607-612.
Staessen J, Yeoman WB, Fletcher AE, Markowe HLJ, Marmot MG, Rose G,
Semmence A, Shipley MJ, & Bulpitt CJ (1990) Blood lead concentration,
renal function, and blood pressure in London civil servants. Br J Ind
Med, 47: 442-447.
Staessen JA, Lauwerys RA, Buchet J-P, Bulpitt CJ, Rondia D,
Vanrenterghem Y, Amery A, & The Cadmibel Study Group (1992) Impairment
of renal function with increasing blood lead concentrations in the
general population. N Engl J Med, 327: 151-156.
Staessen JA, Christopher JB, Fagard R, Lauwerys RR, Roels H, Thijs L,
& Amery A (1994) Hypertension caused by low-level lead exposure: myth
or fact? J Cardiovasc Risk, 1: 87-97.
Steelink C (1977) Humates and other natural organic substances in the
aquatic environment. J Chem Ed, 54: 599-603.
Steenhout A (1987) How clean is clean? An exotoxicological method for
getting guidelines (air, dust, deposition, water) for lead, keeping
tooth and blood lead levels in the normal range. In: Proceedings of
the International Conference on Heavy Metals in the Environment.
Edinburgh, CEP Consultants, pp 283-285.
Steenhout A & Pourtois M (1981) Lead accumulation in teeth as a
function of age with different exposures. Br J Ind Med, 38: 297-303.
Stiles KM & Bellinger DC (1993) Neuropsychological correlates of
low-level lead exposure in school-age children: A prospective study.
Neurotoxicol Teratol, 15(1): 27-35.
Stollery BT, Banks HA, Broadbent DE, & Lee WR (1989) Cognitive
functioning in lead workers. Br J Ind Med, 46: 698-707.
Stollery BT, Broadbent DE, Banks HA, & Lee WR (1991) Short term
prospective study of cognitive functioning in lead workers. Br J Ind
Med, 48: 739-749.
Stone CH & Soares JH (1976) The effect of dietary selenium level of
lead toxicity in the Japanese quail. Poult Sci, 55: 341-349.
Stone C, Fox MRS, & Hogye KS (1981) Bioavailability of lead in oysters
fed to young Japanese quail. Environ Res, 26: 409-421.
Stuik EJ (1974) Biological response of male and female volunteers to
inorganic lead. Int Arch Arbeitsmed, 33: 83-97.
Succop PA, O'Flaherty EJ, Bornschein RL, Roba SB, & Greenland RD
(1989) Recent observations concerning the relationship of blood lead
to erythrocytic protoporphyrin. In: Smith MA, Grant LD, & Sors AE ed.
Lead exposure and child development: An international Assessment.
Dordrecht, London, Kluwer Academic Publishers, pp 477-484.
Talbot RW & Andren AW (1983) Relationships between Pb and 210Pb in
aerosol and precipitation at a semiremote site in northern Wisconsin.
J Geophys Res, 88: 6752-6760.
Taskinen H, Nordman H, Engstrom K, & Hernberg S (1981) [Blood lead
levels in Finnish children.] Duodecim, 97(10): 663-670 (in Finnish).
Teruya K, Sakurai H, Omae K, Higashi T, Muto T, & Kaneko Y (1991)
Effect of lead on cardiac parasympathetic function. Int Arch Occup
Environ Health, 62: 549-553.
Thornton I & Webb JS (1975) Trace elements in soils and surface waters
contaminated plast metaliferous mining part of England. In:
Proceedings of the IXth Annual Conference on Trace Substances in
Environmental Health, Columbia, Missouri, 10-12 June, pp 77-88.
Thornton I, Culbard E, Moorcroft S, Watt J, Wheatley M, & Thompson M
(1985) Metals in urban dusts and soils. Environ Technol Lett,
6: 137-144.
Todd AC, McNeill FE, Palethorpe JE, Peach DE, Chettle DR, Tobin MJ,
Strosko SJ, & Rosen JC (1992) In vivo x-ray fluorescence of lead in
bone using K x-ray excitation with 109Cd sources; radiation dosimetry
studies. Environ Res, 57: 117-132.
Triebig G, Weltle D, & Valentin H (1984) Investigations on
neurotoxicity of chemical substances at the workplace. V.
Determination of the motor and sensory nerve conduction velocity in
persons occupationally exposed to lead. Int Arch Occup Environ Health,
53: 189-204.
Trotter RT (1990) The cultural parameters of lead poisoning: A medical
anthropologist's view of intervention in environmental lead exposure.
Environ Health Perspect, 89: 79-84.
Tuppurainen M, Wagar G, Kurppa K, Sakari W, Wambugu A, Froseth B, Alho
J, & Nykyri E (1988) Thyroid function as assessed by routine
laboratory tests of workers with long-term lead exposure. Scand J Work
Environ Health, 14: 175-180.
UK (1982) The Glasgow duplicate diet study (1979/1980): A joint survey
for the Department of the Environment and the Ministry of Agriculture,
Fisheries and Food. London, Her Majesty's Stationery Office (Pollution
Report No. 11).
US Department of the Treasury (1991) Report of analyses of wines and
related products to determine lead content. Washington, DC, US
Department of the Treasury, Bureau of Alcohol, Tobacco and Firearms.
US EPA (1986a) Air quality criteria for lead. Washington, DC,
Environmental Protection Agency (EPA-600/8-83/028aF-dF).
US EPA (1986b) Test methods for evaluating solid waste SW-846 -
Physical/chemical methods. Method No. 7420 and 7421. Washington, DC,
US Environmental Protection Agency, Office of Solid Waste and
Emergency Response.
US EPA (1989) Evaluation of the potential carcinogenicity of lead and
lead compounds. Washington, DC, US Environmental Protection Agency,
Office of Health and Environmental Assessment (EPA/600/8-89/045A).
US EPA (1990) Supplement to the 1986 EPA air quality criteria for
lead. Volume 1, Addendum: Relationship of blood pressure to lead
exposure. Washington, DC, US Environmental Protection Agency, Office
of Health and Environmental Assessment (EPA/600/8-89/0 49F).
US EPA (1991) National air quality and emission trends report.
Research Triangle Park, North Carolina, US Environmental Protection
Agency (EPA-450/4-91-023).
US NCI (1979) Bioassay of lead dimethylditho-carbamate for possible
carcinogenicity. Washington, DC, National Cancer Institute,
Carcinogenesis Testing Program, Division of Cancer Cause and
Prevention (Technical Report Series No. 151 - NTIS PB-85-134663).
Vahter M & Friberg L (1988) Quality control in integrated human
exposure monitoring of lead and cadmium. Fresenius Z Anal Chem,
332: 726-731.
Vahter M & Slorach S (1990) GEMS - Global Environment Monitoring
System. Exposure monitoring of lead and cadmium: an international
pilot study within the WHO/UNEP Human Exposure Assessment Location
(HEAL) Programme. Nairobi, United Nations Environment Programme,
82 pp.
Vahter M, Berglund M, Friberg L, Jorhem L, Lind B, Slorach S, &
Åkesson A (1990) Dietary intake of lead and cadmium in Sweden. Var
Föda, 42(2): 1-16.
Vahter M, Berglund M, Slorach S, Friberg L, Saric M, Xingquan Z, &
Fujita M (1991a) Methods for integrated exposure monitoring of lead
and cadmium. Environ Res, 56: 78-79.
Vahter M, Berglund M, Lind B, Jorhem L, Slorach S, & Friberg L (1991b)
Personal monitoring of lead and cadmium exposure - a Swedish study
with special reference to methodological aspects. Scand J Work Environ
Health, 17: 65-74.
Van Esch GJ & Kroes R (1969) The induction of renal tumors by feeding
basic lead acetate to mice and hamsters. Br J Cancer, 23: 765-771.
Varo P & Kovistoinen P (1983) Mineral element composition of Finnish
foods. XII. General discussion and nutritional evaluation. Acta Agric
Scand, 22: 165.
Vatsala S & Ramakrishna T (1985) "Tinning" of brass utensils:
Possibility of lead poisoning. Indian J Environ Health, 27: 140-141.
Verrula GR & Noah PK (1990) Clinical manifestations of childhood lead
poisoning. J Trop Med Hyg, 93: 170-177.
Verschoor M, Wibowo A, & Herber R (1987) Influence of occupational low
level lead exposure on renal parameters. Am J Ind Med, 12: 341-351.
Victery W (1988) Evidence for effects of chronic lead exposure on
blood pressure in experimental animals: An overview. Environ Health
Perspect, 78: 71-76.
Victery W, Vander AJ, Markel H, Katzman L, Shulak JM, & Germain C
(1982a) Lead exposure, begun in-utero, decreases renin and angiotensin
II in adult rats. Proc Soc Exp Biol Med, 170: 63-67.
Victery W, Vander AJ, Shulak JM, Schoeps P, & Julius S (1982b) Lead,
hypertension and the renin-angiotensin system in rats. J Lab Clin Med,
99: 354-362.
Volkening J, Baumann H, & Heumann KG (1988) Atmospheric distribution
of particulate lead over the Atlantic Ocean from Europe to Antarctica.
Atmos Environ, 22: 1169-1174.
Wadge A & Hutton M (1987) The leachability and chemical speciation of
selected trace elements in fly ash from coal combustion and refuse
incineration. Environ Pollut, 48: 85-99.
Wai CM, Knowles CR, & Keely JF (1979) Lead caps on wine bottles and
their potential problems. Bull Environ Contam Toxicol, 21: 4-6.
Wallace DM, Kalman DA, & Bird TD (1985) Hazardous lead release from
glazed dinnerware: A cautionary note. Sci Total Environ, 44: 289-292.
Wang Y-L (1984) Industrial lead poisoning in China over the past 33
years. Ecotoxicol Environ Saf, 8: 526-530.
Wang J-D, Jang C-S, Hwang Y-H, & Chen Z-S (1992) Lead contamination
around a kindergarten near a battery recycling plant. Bull Environ
Contam Toxicol, 49: 23-30.
Ward NI, Watson R, & Bryce-Smith D (1987) Placental element levels in
relation to fetal development for obstetrically normal births. A study
of 37 elements. Evidence for the effects of cadmium, lead, and zinc on
fetal growth and for smoking as a source of cadmium. Int J Biosoc Res,
9: 63-81.
Wasserman G, Graziano JH, Factor-Litvak P, Popovac D, Morina N,
Musabegovic A, Vrenezi N, Capuni-Paracka S, Lekic V, Preteni-Redjepi
E, Hadzialijevic S, Slavkovich V, Kline J, Shrout P, & Stein Z (1992)
Independent effects of lead exposure and iron deficiency anemia on
developmental outcome at age 2 years. J Pediatr, 121(5): 695-703.
Watson WS, Hume R, & Moore MR (1980) Oral absorption of lead and iron.
Lancet, 2(8188): 236-237.
Watson WS, Morrison J, Bethel MIF, Baldwin NM, Lyon DTB, Dobson H,
Moore MR, & Hume R (1986) Food iron and lead absorption in humans. Am
J Clin Nutr, 44: 248-256.
Weast RC ed. (1985) Handbook of chemistry and physics, 66th ed. Boca
Raton, Florida, CRC Press.
Weeden RP, Mallik DK, & Batuman V (1979) Detection and treatment of
occupational lead nephropathy. Arch Intern Med, 139: 53-57.
White JM & Harvey DR (1972) Defective synthesis of alpha and ß globin
chains in lead poisoning. Nature (Lond), 236: 71-73.
WHO (1980) Recommended health-based limits in occupational exposure to
heavy metals. Report of a WHO Study Group. Geneva, World Health
Organization (Technical Report Series No. 647).
WHO (1984) Guidelines for drinking-water quality. Volume 1:
Recommendations. Geneva, World Health Organization, pp 55-56.
WHO (1985) Guidelines for the study of dietary intake of chemical
contaminants. Geneva, World Health Organization (WHO Offset
Publication No. 87).
WHO (1987) Air quality guidelines for Europe. Copenhagen, World Health
Organization, Regional Office for Europe, pp 200-209 (European Series,
No. 23).
WHO (1993) Guidelines for drinking-water quality. 2nd ed. Volume 1:
Recommendations. Geneva, World Health Organization, 188 pp.
Wiebe JP, Barr KJ, & Buckingham KD (1982) Lead administration during
pregnancy and lactation affects steriodogenesis and hormone receptors
in testes of offspring. J Toxicol Environ Health, 10: 653-666.
Wiebe JP, Salhanick AI, & Myers KI (1983) On the mechanism of action
of lead on the testes: in vitro suppression of FSH receptors, cyclic
AMP and steroidogenesis. Life Sci, 32: 1997-2005.
Wiebe RA, Anderson BS, Lehman CW, & Fu DJ (1991) Lead poisoning in
Hawaii: 1990. Hawaiian Med J, 50: 89-95.
Wielopolski L, Ellis KJ, Vaswani AN, Cohn SH, Greenberg A, Puschett
JB, Parkinson DK, Fetterolf DE, & Langrigan PJ (1986) In vivo bone
lead measurements: A rapid monitoring method for cumulative lead
exposure. Am J Ind Med, 9: 221-226.
Wigg NR, Vimpani GV, McMichael AJ, Baghurst PA, Robertson EF, &
Roberts RJ (1988) Port Pirie cohort study: childhood blood lead and
neuropsychological development at age two years. J Epidemiol Community
Health, 42: 213-219.
Wildt K, Eliasson R, & Berlin M (1983) Effects of occupational
exposure to lead on sperm and semen. In: Clarkson TW, Nordberg GF, &
Sager PR ed. Reproductive and developmental toxicity of metals. New
York, London, Plenum Press, pp 279-300.
Wilhelm M, Lombeck I, & Hafner D (1989) Hair lead levels in young
children from the FRG. J Trace Elem Electrolytes Health Dis,
3: 165-170.
Williams MK, King E, & Walford J (1969) An investigation of lead
absorption in an electric accumulator factory with the use of personal
samplers. Br J Ind Med, 26: 202-216.
Williamson AM & Teo RKC (1986) Neurobehavioural effects of
occupational lead exposure. Br J Ind Med, 43: 374-380.
Wilson D, Esterman A, Lewis M, Roder D, & Calder I (1986) Children's
blood lead levels in the lead smelting town of Port Pirie South
Australia. Arch Environ Health, 41: 245-250.
Winneke G (1979) Modification of visual evoked potentials in rats
after long-term blood lead elevation. Acta Nerv Sup (Praha),
21: 282-284.
Winneke G, Brockhaus A, & Baltissen R (1977) Neurobehavioural and
system effects of long term blood lead-elevation in rats. I.
Discrimination learning and open field-behavior. Arch Toxicol,
37: 247-263.
Winneke G, Hrdina K-G, & Brockhaus A (1982a) Neuropsychological
studies in children with elevated tooth-lead concentrations. I. A
pilot study. Int Arch Occup Environ Health, 51: 169-183.
Winneke G, Lilienthal H, & Werner W (1982b) Task dependent
neurobehavioural effects of lead in rats. Arch Toxicol, 5(Suppl):
84-93.
Winneke G, Kramer U, Brockhaus A, Ewers U, Kujanek G, Lechner H, &
Janke W (1983) Neuropsychological studies in children with elevated
tooth-lead concentrations. II. Extended study. Int Arch Occup Environ
Health, 51: 231-252.
Winneke G, Beginn U, Ewert T, Havestadt C, Kraemer U, Krause C, Thron
HL, & Wagner HM (1985) Comparing the effects of perinatal and later
childhood lead exposure on neuropsychological outcome. Environ Res,
38: 155-167.
Winneke G, Collet W, Krämer U, Brockhaus A, Ewert T, & Krause C (1989)
Follow-up studies in lead-exposed children. In: Smith MA, Grant LD, &
Sors AI ed. Lead exposure and child development: An international
assessment. Dordrecht, London, Kluwer Academic Publishers, pp 260-270.
Winneke G, Brockhaus A, Ewers U, Kramer U, & Neuf M (1990) Results
from the European multicenter study on lead neurotoxicity in children:
implications for risk assessment. Neurotoxicol Teratol, 12: 553-559.
Wong PTS, Chau YK, & Luxon PL (1975) Methylation of lead in the
environment. Nature (Lond), 253: 263-264.
Wooler K & Melamed S (1978) Simple tests for monitoring excessive lead
absorption. Med J Aust, 1: 163-169.
World Bureau of Metal Statistics (1992) World metal statistics, July,
45(7): 75-78.
Worley PF, Barabon JM, Desouza EB, & Snyder SH (1986) Mapping second
messenger systems in the brain: differential localizations of
adenylate cyclase and protein kinase. C. Proc Natl Acad Sci (USA),
83: 4053-4057.
Worth D, Matranga A, Lieberman M, Devos E, Karelekas P, Ryan C, &
Kraun G (1981) Lead in drinking water: The contribution of household
tap water to blood lead levels. In: Lynam DR, Piantanida LG, & Cole JF
ed. Environmental lead. Proceedings of the Second International
Symposium on Environmental Lead Research. New York, London, San
Francisco, Academic Press, pp 199-225.
Yankel AJ, von Lindern IH, & Walter SD (1977) The Silver Valley lead
study: The relationship between childhood blood lead levels and
environmental exposure. J Air Pollut Control Assoc, 27: 763-767.
Yip R & Dallman PR (1984) Developmental changes in erythrocyte
protoporphyrin: Roles of iron deficiency and lead toxicity. J Pediatr,
710-713.
Yule W, Lansdown R, Millar IB, & Urbanowicz M-A (1981) The
relationship between blood lead concentrations, intelligence and
attainment in a school population: a pilot study. Dev Med Child
Neurol, 23: 567-576.
Zelikoff JT, Li JH, Hartwig A, Wang XW, Costa M, & Rossman TG (1988)
Genetic toxicology of lead compounds. Carcinogenesis, 9: 1727-1732.
Zerez CR, Wong MD, & Tanaka KR (1990) Partial purification and
properties of nicotinamide adenine dinucleotide synthetase from human
erythrocytes: evidence that enzyme activity is a sensitive indicator
of lead exposure. Blood, 75: 1576-1582.
Zhu BG (1984) Study of chronic lead-poisoning from using tea kettles.
Chung-Hua Yu Fang I Hsueh Tsa Chih, 18: 328-330.
Ziegler EE, Edwards BB, Jensen RL, Mahaffey KR, & Fomon SJ (1978)
Absorption and retention of lead by infants. Pediatr Res, 12: 29-34.
Zuckerman MA, Savory D, & Rayman G (1989) Lead encephalopathy from an
imported Toby mug. Postgrad Med J, 65: 307-309.
RESUME
La présente monographie est consacrée aux risques pour la santé
humaine découlant de l'exposition au plomb et aux dérivés minéraux du
plomb. On y insiste sur les données dont on a eu connaissance depuis
la publication du No 3 de la Série des Critères de l'environnement:
Plomb (OMS, 1977). Quant aux effets du plomb au niveau de
l'environnement, ils sont étudiés dans le No 85 de cette série: Lead -
Environmental Aspects (Le plomb dans l'environnement) (WHO, 1989, en
anglais seulement).
1. Identité, propriétés physiques et chimiques et méthodes d'analyse
Le plomb est un métal mou, gris argent, qui fond à 327,5°C. Il
est très résistant à la corrosion mais se dissout à chaud dans l'acide
nitrique et l'acide sulfurique. La valence habituelle du plomb dans
ses dérivés minéraux est de +2. La solubilité dans l'eau de ses
composés est variable, le sulfure et les oxydes étant peu solubles et
le nitrate, le chlorate et le chlorure assez solubles à chaud. Le
plomb forme également des sels avec des acides organiques tels que
l'acide lactique et l'acide acétique ainsi que des composés organiques
stables tels que le plomb-tétraéthyle et le plomb-tétraméthyle.
Les méthodes d'analyse les plus couramment utilisées pour doser
le plomb à faible concentration dans les produits biologiques et dans
l'environnement sont la spectrométrie d'absorption avec atomisation en
flamme, en four à cellules de graphite et en plasma induit par haute
fréquence (ICP/PIHF), ainsi que la coulométrie avec redissolution
anodique. Selon le traitement préalable subit par l'échantillon et les
techniques d'extraction et l'instrumentation utilisées, la limite de
détection peut être de 0,12 µmoles de plomb par litre de sang
(2,49 µg/dl). Toutefois, on n'obtient de résultats fiables qu'en se
conformant à une marche à suivre bien codifiée pour réduire au minimum
le risque de contamination au cours du prélèvement des échantillons,
de leur conservation, de leur traitement et de leur analyse.
2. Sources d'exposition humaine
La proportion du plomb dans la croûte terrestre est d'environ
20 mg/kg. Le plomb présent dans l'environnement peut être d'origine
naturelle ou artificielle. Le plomb naturellement présent dans
l'atmosphère provient, entre autres, de l'altération des roches et des
éruptions volcaniques, et on l'estime à 19 000 tonnes/an, contre
126 000 tonnes/an provenant des mines et des fonderies, la
consommation annuelle totale étant supérieure à 3 millions de tonnes.
On a trouvé des concentrations de plomb atmosphérique de
50 pg/m3 dans des zones reculées. La concentration de fond du plomb
dans le sol varie de 10 à 70 mg/kg et on a relevé à proximité de
routes une teneur moyenne de 138 mg/kg. Actuellement la teneur de
l'eau en plomb dépasse rarement quelques microgrammes/litre; la
concentration naturelle du plomb dans les eaux de surface est estimée
à 0,02 µg/litre.
Le plomb et ses dérivés peuvent pénétrer dans l'environnement par
une porte d'entrée quelconque lors de l'extraction minière, des
opérations de fonte et du traitement, de l'utilisation, du recyclage
ou du rejet du plomb. Le plomb est principalement utilisé dans les
batteries, les câbles, les pigments, les additifs pour essence, la
brasure et les aciers. Le plomb et ses dérivés entrent également dans
la composition de la brasure que l'on utilise pour souder les
conduites d'eau et les boites de conserve alimentaire; il sert
également à la confection de certains remèdes traditionnels, des
capsules de bouteilles contenant des boissons alcoolisées, des vernis
pour céramique et de la verrerie de cristal. Dans les pays où l'on
utilise encore de l'essence au plomb, les principales émissions dans
l'atmosphère proviennent de sources de combustion de produits
pétroliers, sources qui peuvent être mobiles ou fixes (en
agglomération). Les émissions dans l'atmosphère sont également très
importantes à proximité des mines et des fonderies de plomb.
Le plomb en suspension dans l'air peut se déposer sur le sol et
dans l'eau et il parvient ainsi jusqu'à l'homme par l'intermédiaire de
la chaîne alimentaire et de l'eau de boisson. Le plomb atmosphérique
contribue également pour une part importante au plomb présent dans la
poussière domestique.
3. Transport, distribution et transformation dans l'environnement
Le transport et la distribution du plomb à partir de sources
fixes, mobiles ou naturelles, s'effectue principalement par
l'intermédiaire de l'air. La majeure partie du plomb émis dans l'air
se redépose à proximité de la source d'émission, même si quelques
particules (de diamètre < 2 µm) dont transportées sur de longues
distances et provoquent la contamination de sites aussi reculés que
les glaciers arctiques. Le plomb aéroporté peut contribuer à
l'exposition humaine par contamination des aliments, d l'eau et de la
poussière, ainsi que par inhalation directe. L'élimination du plomb
aéroporté dépend des conditions atmosphériques et de la taille des
particules en suspension. De grandes quantités de plomb peuvent être
déversées dans le sol et l'eau. Cependant, ces produits ont tendance à
rester sur place du fait de la médiocre solubilité dans l'eau des
dérivés du plomb.
Le plomb qui s'est déposé dans l'eau, que ce soit à partir de
l'air ou par lessivage des sols, se répartit rapidement entre les
sédiments et la phase aqueuse, selon la valeur du pH, la teneur en
sels et la présence éventuelle d'agents chélateurs organiques. Lorsque
le pH dépasse 5,4, l'eau dure peu contenir environ 30 µg de
plomb/litre et l'eau douce environ 500 µg/litre. Une très faible
quantité du plomb déposé sur le sol passe dans les eaux de surface ou
les eaux souterraines sauf en cas d'érosion ou d'altération des
roches; il est normalement fortement lié par chélation aux matières
organiques.
Le plomb en suspension dans l'air peut passer dans les êtres
vivants soit directement, soit par fixation à partir du sol. Les
animaux peuvent être exposés au plomb de manière directe par broutage
de l'herbe, ingestion de terre ou par inhalation. Le plomb minéral ne
subit qu'une faible bioamplification le long de la chaîne alimentaire.
4. Concentrations dans l'environnement et exposition humaine
Dans la population générale adulte des non-fumeurs, la principale
voie d'exposition est la consommation de nourriture et d'eau. Le plomb
est suspension dans l'air peut jouer un rôle important dans
l'exposition, en fonction de facteurs tels que le tabagisme, la
profession, la proximité d'une autoroute, d'une fonderie, etc. ou de
certains lieux de loisirs (par exemple, ateliers d'artisanat, stands
de tir, etc.). Pour les nourrissons et les enfants en bas âge, la
nourriture, l'air, l'eau et la poussière ou la terre sont les
principales voies d'exposition potentielles. Pour les nourrissons
jusqu'à quatre ou cinq mois, les principales sources d'exposition au
plomb sont l'air, le lait et les laits maternisés ainsi que l'eau.
Les concentrations de plomb observées dans l'air, les aliments,
l'eau et le sol ou la poussière varient largement d'une région à
l'autre du monde et dépendent du degré d'industrialisation,
d'urbanisation ainsi que du mode de vie. Dans l'air ambiant, des
concentrations supérieures à 10 µg/m3 ont été signalées en milieu
urbain à proximité d'une fonderie, alors que dans les villes où l'on
n'utilise plus d'essence au plomb, ces concentrations peuvent tomber
en-dessous de 0,2 µg/m3. Dans ces conditions, la dose de plomb
absorbée à partir de l'air peut varier de moins de 4 µg/jour à plus de
200 µg/jour.
Dans des échantillons d'eau potable prélevés à la source, on
trouve généralement des concentrations de plomb inférieures à
5 µg/litre. Cependant, lorsque l'eau est prélevée au robinet dans les
maisons dont les canalisations sont en plomb, les teneurs peuvent
dépasser 100 µg/litre, en particulier lorsque l'eau a séjourné dans la
tuyauterie pendant plusieurs heures.
L'exposition au plomb par la voie alimentaire dépend de nombreux
facteurs tenant au mode de vie et notamment à la nature des aliments
consommés, au mode de préparation, au fait que l'on utilise ou non de
la brasure au plomb, à la teneur de l'eau en plomb et à l'utilisation
de vaisselle recouverte d'un vernis au plomb.
Les nourrissons et les enfants peuvent souvent être très exposés
au plomb présent dans la poussière et dans la terre. La teneur de la
poussière en plomb dépend de facteurs tels que la vétusté et l'état de
la maison, l'utilisation de peintures à base de plomb, la présence de
plomb dans l'essence et la densité urbaine. La dose de plomb absorbée
dépendra également de l'âge et du comportement de l'enfant ainsi que
de la biodisponibilité du plomb dans le produit où il est présent.
Chez les ouvriers des industries où l'on produit, raffine,
utilise ou rejette du plomb ou des dérivés du plomb, la principale
voie d'exposition est l'inhalation. Au cours d'un poste de travail de
8 heures, les ouvriers peuvent en absorber des quantités atteignant
400 µg, qui s'ajoutent aux 20 ou 30 µg absorbés quotidiennement à
partir de la nourriture, de l'eau et de l'air ambiant; l'ingestion de
grosses particules peut également contribuer de façon importante à cet
apport.
5. Cinétique et métabolisme chez les animaux de laboratoire et
l'homme
L'homme et les animaux résorbent le plomb qu'ils ingèrent ou
inhalent; la résorption percutanée est minime chez l'homme. Selon sa
granulométrie, sa forme chimique et sa solubilité dans les liquides
biologiques, un dérivé du plomb peut être résorbé jusqu'à hauteur de
50% après avoir été inhalé. Certaines grosses particules (plus de
7 µm) sont avalées après avoir été rejetées des voies respiratoires
par l'action de l'ascenseur muco-ciliaire. Chez l'homme et les animaux
d'expérience, l'absorption du plomb dans les voies digestives dépend
de la nature physico-chimique du produit ingéré, de l'état
nutritionnel du sujet ainsi que du type d'aliments consommés. Chez
l'homme adulte, le plomb contenu dans les aliments est absorbé à peu
près à hauteur de 10%; la proportion est plus élevée lorsque le sujet
est à la diète. Toutefois, chez les nourrissons et les enfants en bas
âge, le plomb d'origine alimentaire peut être absorbé à hauteur de
50%, encore que le taux d'absorption du plomb provenant des poussières
ou de la terre ainsi que des écailles de peinture puisse être plus
faible, selon sa biodisponibilité. Les régimes alimentaires pauvres en
calcium, phosphates, sélénium ou zinc peuvent accroître l'absorption
du plomb. Le fer et la vitamine D influent également sur l'absorption
du plomb.
Le taux de plomb dans le sang ou plombémie est utilisé pour
évaluer la charge en plomb de l'organisme ainsi que les doses de plomb
(internes) absorbées. La relation entre la plombémie et la
concentration du plomb dans les diverses sources d'exposition n'est
pas linéaire (corrélation curviligne).
Une fois absorbé, le plomb ne se répartit pas de manière homogène
dans l'organisme. Après être rapidement passé dans le sang et les
tissus mous, il se redistribue lentement des les os. La plomb
s'accumule dans les os pendant une longue période de la vie humaine et
peut servir de source endogène de plomb. La demi-vie du plomb dans le
sang et les tissus mous est d'environ 28 à 36 jours mais elle peut
être beaucoup plus longue dans les diverses parties de l'os. Le taux
de rétention du plomb dans l'organisme est plus élevé chez l'enfant
que chez l'adulte. Pendant toute la durée de la gestation, le plomb
passe facilement de la mère au foetus.
La plombémie est la mesure la plus couramment utilisée pour
évaluer l'exposition au plomb. Toutefois, on dispose aujourd'hui de
techniques permettant de doser le plomb dans les dents et les os,
encore que la cinétique du phénomène ne soit pas parfaitement
élucidée.
6. Effets sur les animaux de laboratoire et les systèmes d'épreuves
in vitro
Chez toutes les espèces d'animaux de laboratoire étudiées, y
compris des primates non-humains, on a constaté que le plomb
produisait des effets indésirables au niveau de plusieurs organes et
systèmes, notamment le système haematopoïétique, le système nerveux,
les reins, le système cardio-vasculaire, l'appareil reproducteur et le
système immunitaire. Le plomb est également nocif pour les os et on a
montré qu'il avait des effets cancérogènes sur le rat et la souris.
Malgré les différences d'ordre cinétique qui existent entre les
animaux d'expérience et l'homme, ces études apportent des arguments
biologiques de poids à la plausibilité de tels effets chez l'homme.
Chez le rat, on a observé une diminution de la capacité
d'apprentissage et de mémorisation lorsque la plombémie était de
l'ordre de 0,72 à 0,96 µmol/litre (soit 15 à 20 µg/dl), les mêmes
effets étant observés chez des primates non-humains pour une plombémie
ne dépassant pas 0,72 µmol/litre (15 µg/dl). En outre,
l'expérimentation animale a également permis d'observer des troubles
de la vision et de l'audition.
Chez le rat, une plombémie supérieure à 2,88 µmol/litre
(60 µg/dl), soit une valeur analogue à celle qui, selon les
observations, constitue le seuil d'apparition des effets chez l'homme,
est à même de provoquer l'apparition d'effets néphrotoxiques. Des
effets cardio-vasculaires ont été également observés chez des rats
après exposition chronique à de faibles doses de plomb entraînant une
plombémie de l'ordre de 0,24 à 1,92 µmol/litre (5 à 40 µg/dl). A des
doses inférieures à la dose maximale tolérée qui est de 200 mg de
plomb (sous forme d'acétate) par litre d'eau potable, on a observé
l'apparition de tumeurs. Cette dose constitue la dose maximale qui
n'entraîne pas d'autres effets morphologiques ou fonctionnels.
7. Effets sur l'homme
Chez l'homme, le plomb peut produire des effets biologiques très
divers selon l'intensité et la durée de l'exposition. On a ainsi
observé des effets au niveau infracellulaire ainsi que des effets
s'exerçant sur les fonctions générales de l'organisme, effets qui vont
de l'inhibition de certaines enzymes jusqu'à l'apparition
d'altérations morphologiques marquées et à la mort. Ces altérations se
produisent dans de larges limites de doses, l'organisme humain en
développement étant généralement plus sensible que l'organisme adulte.
On a montré que le plomb avait des effets sur nombre de processus
biochimiques; en particulier on a largement étudié les effets qu'il
produit sur la synthèse hémique, tant chez l'adulte que chez l'enfant.
Lorsque la plombémie est élevée, on observe une augmentation du taux
de protoporphyrine érythrocytaire sérique ainsi qu'une augmentation de
l'excrétion urinaire de coproporphyrine et d'acide delta-aminolévulinique.
A des valeurs plus faibles de la plombémie, on observe l'inhibition
d'enzymes comme la delta-aminolévulinique acide-déshydratase et la
dihydrobioptérine-réductase.
Les effets du plomb sur le système haématopoïétique se traduisent
par une diminution de la synthèse de l'hémoglobine et l'on a observé
une anémie chez des enfants lorsque la plombémie dépassait
1,92 µmol/litre (40 µg/dl).
Pour des raisons d'ordre neurologique, métabolique et
compartementale, les enfants sont plus sensibles aux effets du plomb
que les adultes. Des études épidémiologiques tant prospectives que
transversales ont été effectuées pour déterminer dans quelle mesure
une exposition au plomb présent dans l'environnement affecte les
fonctions psychologiques dépendant du système nerveux central. On a
ainsi montré qu'il existait une association entre l'exposition au
plomb et les troubles des fonctions neurocomportementales chez
l'enfant.
On a également constaté des anomalies des fonctions
psychologiques et neurocomportementales chez des ouvriers longtemps
exposés au plomb. Il a été montré que les paramètres
électrophysiologiques étaient d'utiles indicateurs des effets
infracliniques du plomb sur le système nerveux central.
On sait depuis longtemps que la neuropathie périphérique est due
à une exposition prolongée à de fortes concentrations de plomb sur le
lieu de travail. A plus faibles concentrations, on a observé une
diminution de la vitesse de conduction nerveuse. Ces effets se
révèlent souvent réversibles, en fonction de l'âge et de la durée de
l'exposition, après cessation de l'exposition.
Les effets que le plomb exerce sur le coeur sont indirects et
s'opèrent par l'intermédiaire du système neuro-végétatif; il n'y a pas
d'effets directs sur le myocarde. L'ensemble des faits tirés des
études sur des populations d'adultes indiquent qu'il existe une très
faible association entre la plombémie et la tension artérielle
systolique ou diastolique. Etant donné la difficulté qu'il y a à tenir
compte des facteurs de confusion, il n'a pas été possible d'établir, à
partir des résultats de ces études, une relation de cause à effet.
Rien n'indique non plus que l'association qui pourrait exister entre
la plombémie et la tension artérielle constitue un problème médical
majeur.
On sait que le plomb peut entraîner des lésions tubulaires
proximales qui se caractérisent par une aminoacidurie généralisée, une
hypophosphatémie avec d'une hyperphosphaturie relative et une
glycosurie accompagnée d'inclusions nucléiques, d'altérations des
mitochondries et d'une hypertrophie des cellules de l'épithélium
tubulaire proximal. Ces effets sur les tubules s'observent après une
exposition relativement brève et sont généralement réversibles. En
revanche, les altérations scléreuses et les fibroses interstitielles
qu'on observe lors d'une exposition chronique à de fortes
concentrations de plomb, entraînent des problèmes fonctionnels qui
peuvent déboucher sur une insuffisance rénale. On a observé une
augmentation du risque de néphropathie chez les travailleurs dont la
plombémie dépassait 3,0 µmol/litre (soit environ 60 µg/dl). En
utilisant des indicateurs fonctionnels plus sensibles, on a récemment
découvert l'existence d'effets rénaux dans la population générale.
Les effets du plomb sur la fonction de reproduction, ne
concernent, chez l'homme, que la morphologie et le nombre des
spermatozoïdes. Chez la femme, on a attribué au plomb un certain
nombre de grossesses à issue défavorable.
Il ne semble pas que le plomb ait des effets nocifs sur la peau,
les muscles ou le système immunitaire. Si l'on excepte le cas du rat,
le plomb ne paraît pas non plus être à l'origine de l'apparition de
tumeurs.
8. Evaluation des risques pour la santé humaine
Le plomb a des effets nocifs sur plusieurs organes et systèmes,
les effets les plus sensibles étant relevés au niveau infracellulaire
ainsi que sur le développement du système nerveux. On a fait état
d'une association entre la plombémie et l'hypertension. En outre, le
plomb entraîner une cascade d'effets sur les réserves hémiques de
l'organisme et il perturbe également la synthèse hémique. Cependant,
certains de ces effets ne sont pas véritablement considérés comme
délétères. Il y a également perturbation de l'homéostase du calcium
avec des contre-coups sur d'autres processus cellulaires.
a) Les preuves les plus substantielles sont on dispose à propos de
l'action nocive du plomb proviennent d'études transversales et
prospectives sur des populations dont la plombémie est
généralement inférieure à 1,2 µmol/litre (25 µg/dl) et concernent
une diminution du quotient d'intelligence (QI). Il importe
cependant de noter que ces observations ne constituent pas une
preuve définitive d'une relation de cause à effet entre ce
phénomène et l'exposition au plomb. Cependant la mesure de ce QI
à partir de l'âge de 4 ans fait ressortir un déficit qui se situe
entre 0 et 5 points (sur une échelle avec un écart-type de 15)
pour une augmentation de la plombémie de 0,48 µmol/litre
(10 µg/dl), l'ampleur de l'effet étant vraisemblablement de 1 à 3
points. Lorsque la plombémie dépasse 1,2 µmol/litre (25 µg/dl),
on peut avoir une relation différente entre cette plombémie et le
QI. Les estimations de l'ampleur de l'effet observé sont des
moyennes calculées sur des groupes et ne représentent qu'une
probabilité pour un individu en particulier.
Les études épidémiologiques dont on dispose ne donnent pas
de preuves définitives de l'existence d'un seuil. Lorsque la
plombémie se situe en-dessous de l'intervalle
0,48-0,72 µmol/litre (10-15 µg/dl), l'incertitude qui entache
toute estimation de l'effet s'accroît, du fait des facteurs de
confusion et des limites dans la précision des dosages et des
tests psychométriques. Il n'en reste pas moins qu'en-dessous de
cet intervalle de valeurs, certains éléments incitent à penser à
l'existence d'une association.
b) L'expérimentation animale milite en faveur de l'existence d'une
relation de cause à effet entre l'exposition au plomb et certains
effets neurologiques, puisqu'elle fait ressortir des déficits
dans les fonctions cognitives pour une plombémie ne dépassant pas
0,53 à 0,72 µmol/litre (11-15 µg/dl), déficits qui peuvent
persister bien après la cessation de l'exposition au plomb.
c) Pour une plombémie ne dépassant pas 1,44 µmol/litre (30 µg/dl),
il peut y avoir réduction de la vitesse de conduction nerveuse
périphérique chez l'homme. En outre, pour des valeurs de la
plombémie de dépassant pas 1,92 µmol/litre (40 µg/dl), il peut
également y avoir perturbation des fonctions moto-sensorielles et
la fonction du système nerveux neurovégétatif (variabilité de
l'intervalle R-R sur l'électrocardiogramme) peut être affectée
pour une valeur moyenne de la plombémie d'environ 1,68 µmol/litre
(35 µg/dl). Chez les travailleurs dont la plombémie dépasse
2,88 µmol/litre (60 µg/dl), il y a accroissement du risque de
néphropathie saturnienne. Toutefois, des études récentes basées
sur des indicateurs plus sensibles de la fonction rénale incitent
à penser que des effets peuvent se produire à des valeurs plus
faibles de l'exposition au plomb.
d) Il semblerait que l'exposition au plomb soit associée à une
légère augmentation de la tension artérielle. L'ordre de grandeur
probable de cette augmentation est le suivant: pour un doublement
de la plombémie (par exemple lorsqu'elle passe de 0,8 à
1,6 µmol/litre, soit de 16,6 à 33,3 µg/dl), il y a une
augmentation moyenne de 1 mmHg de la systolique. L'association
avec la diastolique est analogue mais d'ampleur plus faible.
Toutefois, on se demande si ces associations statistiques
résultent réellement de l'exposition au plomb ou s'il s'agit d'un
artefact imputable à des facteurs de confusion.
e) Certaines études épidémiologiques - mais pas toutes - font état
d'une association liée à la dose entre les accouchements avant
terme et certains indices de la croissance et de la maturation
foetales à des valeurs de la plombémie supérieures ou égales à
0.72 µmol/litre (15 µg/dl).
f) Les données relatives à la cancérogénicité pour l'homme du plomb
et de plusieurs de ses dérivés minéraux, sont insuffisantes.
g) On a mis en évidence des effets que le plomb exerce sur un
certain nombre de systèmes enzymatiques et de paramètres
biochimiques. Les valeurs de la plombémie au-dessus desquelles on
peut mettre en évidence des effets avec les techniques actuelles,
pour ce qui est des paramètres susceptibles d'avoir une
importance clinique, sont toutes supérieures à 0,96 µmol/litre
(20 µg/dl). Certains effets sur les enzymes peuvent être mis en
évidence à des valeurs plus faibles de la plombémie, mais leur
signification clinique demeure incertaine.
RESUMEN
La presente monografía se centra en los riesgos para la salud
humana asociados a la exposición al plomo y a los compuestos
inorgánicos de plomo. Se han destacado los datos disponibles desde la
publicación de Criterios de Salud Ambiental No. 3: Plomo (OMS, 1977).
Los efectos ambientales del plomo se examinan en Environmental Health
Criteria 85: Lead - Environmental Aspects (OMS, 1989).
1. Identidad, propiedades físicas y químicas y métodos analíticos
El plomo es un metal blando, gris plateado, que se funde a
327,5°C. Es muy resistente a la corrosión, pero es soluble en ácido
nítrico y en ácido sulfúrico caliente. Su valencia corriente en los
compuestos inorgánicos es +2. Su solubilidad en agua varía; el sulfito
de plomo y los óxidos de plomo son poco solubles, mientras que las
sales de nitrato, clorato y cloruro son razonablemente solubles en
agua fría. El plomo también forma sales con ácidos orgánicos tales
como el láctico y el acético, y compuestos orgánicos estables tales
como el tetraetilo de plomo y el tetrametilo de plomo.
Los métodos utilizados más corrientemente para el análisis de
bajas concentraciones de plomo en materias biológicas y ambientales
son la llama, el horno de grafito y la espectroscopia de absorción
atómica de plasma acoplado inductivamente y la voltimetría de
separación anódica. Según sean el tratamiento previo de la muestra,
las técnicas de extracción y la instrumentación analítica, pueden
alcanzarse niveles de detección de 0,12 µmoles de plomo por litro de
sangre (2,49 µg/dl). Sin embargo, se obtienen resultados fiables sólo
cuando se siguen procedimientos específicos para reducir al mínimo el
riesgo de contaminación durante la recogida, el almacenamiento,
procesamiento y análisis de la muestra.
2. Fuentes de exposición humana
El nivel de plomo en la corteza terrestre es de aproximadamente
20 mg/kg. El plomo del medio ambiente puede provenir de fuentes
naturales o antropogénicas. Las fuentes naturales de plomo atmosférico
comprenden el desgaste geológico y las emisiones volcánicas y se han
estimado en 19 000 toneladas por año, frente a unas 126 000 toneladas
por año emitidas en el aire como resultado de la minería, la fundición
y el consumo de más de 3 millones de toneladas de plomo por año.
Se han encontrado concentraciones atmosféricas de plomo de
50 pg/m3 en zonas remotas. Los niveles básicos de plomo en el suelo
oscilan entre 10 y 70 mg/kg y se ha comunicado un nivel medio de
138 mg/kg en las proximidades de las carreteras. Los niveles de plomo
presentes en las aguas rara vez exceden de unos pocos microgramos por
litro; la concentración natural de plomo en las aguas superficiales se
ha estimado en 0,02 µg/litro.
El plomo y sus compuestos pueden entrar en el medio ambiente en
cualquier punto durante las actividades de minería, fundición,
elaboración, utilización, reciclado o eliminación. Se utiliza
principalmente en la fabricación de pilas, cables, pigmentos, aditivos
de la gasolina, productos para soldar y de acero. El plomo y los
compuestos de plomo también se utilizan para soldar las tuberías de
distribución de agua y las latas de conserva, en algunos remedios
tradicionales, en las tapas de las botellas de bebidas alcohólicas y
en los esmaltes cerámicos y la cristalería de mesa. En los países
donde todavía se utiliza gasolina con plomo, la principal emisión en
el aire proviene de fuentes móviles y estacionarias de combustión de
gasolina (centros urbanos). Las zonas próximas a las minas y funderías
de plomo están expuestas a la emisión de niveles elevados en el aire.
El plomo del aire puede depositarse en el suelo y el agua, desde
donde llega al ser humano por conducto de la cadena alimentaria y del
agua de bebida. El plomo atmosférico también es una fuente importante
del plomo presente en el polvo de las viviendas.
3. Transporte, distribución y transformación en el medio ambiente
El transporte y la distribución del plomo procedente de fuentes
fijas, móviles y naturales tienen lugar principalmente a través del
aire. La mayor parte de las emisiones de plomo se depositan cerca de
la fuente, aunque algunas partículas de materia (< 2 µm de diámetro)
recorren largas distancias y contaminan lugares remotos tales como los
glaciares árticos. El plomo del aire puede contribuir a la exposición
humana mediante la contaminación de los alimentos, del agua y del
polvo, así como por inhalación directa. La eliminación del plomo del
aire depende de las condiciones atmosféricas y del tamaño de las
partículas. Pueden descargarse grandes cantidades de plomo en el suelo
y en el agua. Sin embargo, ese material tiende a permanecer localizado
debido a la escasa solubilidad de los compuestos de plomo en el agua.
El plomo depositado en el agua, ya provenga del aire o de la
escorrentía del suelo, se distribuye rápidamente entre el sedimento y
la fase acuosa, según el pH, el contenido de sales y la presencia de
agentes quelantes orgánicos. Con un pH superior a 5,4, las aguas duras
pueden contener aproximadamente 30 µg de plomo por litro y las aguas
blandas aproximadamente 500 µg de plomo por litro. Muy poco plomo
depositado en el suelo se transporta a las aguas superficiales o a las
subterráneas, salvo mediante la erosión o el desgaste geoquímico;
normalmente está ligado a la materia orgánica de forma bastante
estrecha (por quelación).
El plomo del aire puede transferirse a la biota directamente o
por absorción del suelo. Los animales pueden encontrarse expuestos al
plomo directamente mediante la ingestión de hierba y de tierra o por
inhalación. Hay poca biomagnificación del plomo inorgánico a través de
la cadena alimentaria.
4. Niveles ambientales y exposición humana
En la población general que no fuma, la principal vía de
exposición son los alimentos y el agua. El plomo del aire puede
contribuir apreciablemente a la exposición, lo que depende de factores
tales como el consumo de tabaco, la ocupación, la proximidad de
caminos transitados por vehículos automotores, de funderías de plomo,
etc., y ciertas actividades de esparcimiento (por ejemplo, artesanía,
tiro con armas de fuego). Los alimentos, el aire, el agua y el
polvo/suelo son las principales vías potenciales de exposición de los
niños pequeños. Para los niños de hasta 4 ó 5 meses de edad, el aire,
la leche, las preparaciones para lactantes y el agua son fuentes
notables de exposición al plomo.
Los niveles de plomo presentes en el aire, los alimentos, el
agua, y el suelo/polvo varían ampliamente en el mundo y dependen del
grado de desarrollo industrial y de urbanización y de factores
relacionados con el modo de vida. Se han comunicado niveles superiores
a 10 µg/m3 presentes en el aire ambiental en zonas urbanas próximas
a funderías, mientras que en ciudades donde ha dejado de usarse la
gasolina con plomo se han detectado niveles inferiores a 0,2 µg/m3.
La absorción de plomo del aire puede, pues, variar de menos de
4 µg/día a más de 200 µg/día.
Los niveles de plomo en muestras de agua de bebida extraídas de
los manantiales suelen ser inferiores a 5 µg/litro. Sin embargo, el
agua del grifo de viviendas cuyas tuberías tienen plomo contiene
niveles que exceden de 100 µg/litro, en particular después de haber
reposado el agua en las tuberías durante algunas horas.
El nivel de exposición al plomo a través de la dieta depende de
muchos factores relacionados con el modo de vida, entre ellos los
alimentos que se consumen, la tecnología de elaboración, el empleo de
soldadura de plomo, los niveles de plomo en el agua y la utilización
de cerámica con barniz de plomo.
Para los niños, el plomo presente en el polvo y en el suelo suele
ser la principal vía de exposición. Los niveles de plomo en el polvo
dependen de factores tales como la antigüedad y el estado de la
vivienda, la utilización de pinturas a base de plomo, el plomo de la
gasolina y la densidad urbana. La absorción de plomo dependerá de la
edad y de las características comportamentales del niño así como de la
biodisponibilidad de plomo en la fuente material.
La inhalación es la vía principal de exposición al plomo para los
trabajadores de industrias que producen, refinan, utilizan o desechan
plomo y compuestos de plomo. Durante un turno de ocho horas, los
trabajadores pueden absorber nada menos que 400 µg de plomo, además de
los 20-30 µg/día que absorben de los alimentos, del agua y del aire
ambiental; puede haber una absorción notable como resultado de la
inhalación de partículas grandes.
5. Cinética y metabolismo en animales de laboratorio y en el ser
humano
Los seres humanos y los animales absorben plomo por inhalación o
por ingestión; la absorción percutánea es mínima en el ser humano.
Según la especiación química, el tamaño de las partículas y la
solubilidad de los líquidos corporales, puede absorberse hasta un 50%
de los compuestos de plomo inhalados. Algunas partículas de materia
inhaladas (de más de 7 µm) se degluten después de la eliminación
mucociliar del aparato respiratorio. En los animales experimentales y
en el ser humano, la absorción de plomo del aparato gastrointestinal
está influenciada por la naturaleza fisicoquímica del material
ingerido, el estado nutricional y el tipo de alimentación. En los
seres humanos adultos, se absorbe aproximadamente el 10% del plomo
contenido en la alimentación; la proporción es más elevada en
condiciones de ayuno. Sin embargo, los lactantes y los niños pequeños
absorben nada menos que el 50% del plomo presente en la alimentación,
pero la absorción de plomo del polvo/suelo y de desconchones de
pintura puede ser menor y depende de su biodisponibilidad. Las dietas
pobres en calcio, fosfato, selenio o zinc pueden dar lugar a una mayor
absorción de plomo. El hierro y la vitamina D también influyen en la
absorción de plomo.
Los niveles de plomo en la sangre (Pb-H) se utilizan para medir
la carga corporal y las dosis absorbidas (internas) de plomo. La
relación entre el plomo presente en la sangre y la concentración de
plomo en las fuentes de exposición es curvilínea.
Una vez absorbido, el plomo no se distribuye de manera homogénea
en todo el cuerpo. Hay una absorción rápida en la sangre y en los
tejidos blandos, seguida de una redistribución más lenta a los huesos.
Los huesos acumulan plomo durante gran parte de la vida humana y
pueden actuar como fuente endógena de plomo. La semivida del plomo en
la sangre y en otros tejidos blandos es de aproximadamente 28-36 días,
pero es mucho más larga en los diversos compartimentos óseos. La
retención porcentual de plomo en los depósitos corporales es más
elevada en los niños que en los adultos. La transferencia de plomo al
feto humano se efectúa fácilmente durante la gestación.
El nivel de plomo en la sangre es la medida más utilizada para
determinar la exposición al plomo. Sin embargo, ya se dispone de
técnicas para determinar la cantidad de plomo presente en los dientes
y en los huesos, aunque aún no se conoce del todo su cinética.
6. Efectos en los animales de laboratorio y en los sistemas in vitro
En todas las especies de animales de experimentación estudiadas,
inclusive en primates no humanos, se ha observado que el plomo tiene
efectos adversos en varios órganos y sistemas de órganos, inclusive
los sistemas hematopoyético, nervioso, renal, cardiovascular,
reproductivo e inmunitario. El plomo también afecta a los huesos y se
ha demostrado que es carcinógeno en ratas y ratones.
Pese a diferencias cinéticas entre las especies de animales
experimentales y la humana, dichos estudios apoyan firmemente y
justifican desde un punto de vista biológico los hallazgos realizados
en seres humanos. Se han comunicado deficiencias del aprendizaje y de
la memoria en ratas con niveles de Pb-H de 0,72-0,96 µmoles/litro
(15-20 µg/dl) y en primates no humanos con niveles de Pb-H de no más
de 0,72 µmoles/litro (15 µg/dl). Además, se han comunicado
deficiencias visuales y auditivas en estudios realizados en animales
de experimentación.
La toxicidad renal en las ratas parece presentarse a partir de un
nivel de Pb-H de 2,88 µmoles/litro (60 µg/dl); este valor es semejante
al que, según se ha comunicado, comienza a tener efectos renales en el
ser humano. Se han observado efectos cardio-vasculares en ratas
después de exposiciones crónicas a niveles bajos que dan lugar a
niveles de Pb-H de 0,24-1,92 µmoles/litro (5-40 µg/dl). Se ha
demostrado que aparecen tumores con dosis inferiores a la dosis máxima
tolerada, de 200 mg de plomo (como acetato de plomo) por litro de agua
de bebida. Esta es la dosis máxima no asociada a otros cambios
morfológicos o funcionales.
7. Efectos en el ser humano
En el ser humano, el plomo puede tener una amplia variedad de
efectos biológicos según el nivel y la duración de la exposición. Se
han observado efectos en el plano subcelular y efectos en el
funcionamiento general del organismo que van desde la inhibición de
las enzimas hasta la producción de acusados cambios morfológicos y la
muerte. Dichos cambios se producen a dosis muy diferentes; en general,
el ser humano que se está desarrollando es más sensible que el adulto.
Se ha mostrado que el plomo tiene efectos en muchos procesos
bioquímicos; en particular, se han estudiado mucho los efectos en la
síntesis del hemo en adultos y niños. Se observan niveles más altos de
porfirina eritrocitaria sérica y mayor excreción urinaria de
coproporfirina y de ácido delta-aminolevulínico cuando las
concentraciones de Pb-H son elevadas. Con niveles más bajos se observa
inhibición de las enzimas dehidratasa del ácido delta-aminolevulínico
y reductasa de la dihidrobiopterina.
Como resultado de los efectos del plomo en el sistema
hematopoyético disminuye la síntesis de hemoglobina y se ha observado
anemia en niños a concentraciones de Pb-H superiores a
1,92 µmoles/litro (40 µg/dl).
Por razones neurológicas, metabólicas y comportamentales, los
niños son más vulnerables a los efectos del plomo que los adultos. Se
han efectuado estudios epidemiológicos prospectivos y transversales
para evaluar la medida en que la exposición al plomo ambiental afecta
a las funciones psicológicas regidas por el sistema nervioso central
(SNC). Se ha mostrado que el plomo está asociado a deficiencias
neurocomportamentales en los niños.
Se han observado deficiencias psicológicas y
neurocomportamentales en trabajadores que habían estado expuestos al
plomo durante un tiempo prolongado. Los parámetros electrofisiológicos
han demostrado ser indicadores útiles de los efectos subclínicos del
plomo en el SNC.
Desde hace tiempo se sabe que la exposición prolongada a niveles
elevados de plomo en el medio laboral provoca neuropatías periféricas.
Con niveles más bajos se ha observado una reducción de la velocidad de
conducción nerviosa. Se ha observado a menudo que dichos efectos son
reversibles después de cesar la exposición, según la edad del sujeto y
la duración de la exposición.
Los efectos del plomo en el corazón son indirectos y se producen
por conducto del sistema nervioso autónomo; el plomo no tiene efectos
directos en el miocardio. Datos colectivos procedentes de estudios de
poblaciones adultas indican asociaciones muy débiles entre la
concentración de Pb-H y la presión arterial sistólica o diastólica.
Dada la dificultad de evaluar el influjo de los factores de confusión
pertinentes, no puede establecerse una relación causal sobre la base
de esos estudios. No hay indicios de que la relación entre la
concentración de Pb-H y la presión arterial tenga mucha importancia
para la salud.
Se sabe que el plomo provoca en los tubos proximales del riñón
lesiones que se caracterizan por aminoaciduria generalizada,
hipofosfatemia con hiperfosfaturia relativa y glucosuria acompañada de
cuerpos de inclusión nuclear, modificaciones mitocondriales y
citomegalia de las células epiteliales de los tubos proximales. Los
efectos tubulares se manifiestan después de una exposición
relativamente breve y suelen ser reversibles, mientras que los cambios
escleróticos y la fibrosis intersticial, que dan lugar a una
disminución de la función renal y a una posible insuficiencia renal,
requieren una exposición crónica a niveles elevados de plomo. Se ha
advertido un mayor riesgo de nefropatía en los trabajadores que tienen
niveles de Pb-H superiores a 3,0 µmoles/litro (aproximadamente
60 µg/dl). Recientemente se han observado efectos renales en la
población general tras haberse utilizado indicadores de función más
sensibles.
Los efectos del plomo en la función reproductora masculina se
limitan a la morfología y el número de los espermatozoides. En cuanto
a la femenina, se han atribuido al plomo algunos efectos adversos en
el embarazo.
El plomo no parece tener efectos nocivos en la piel, en los
músculos ni en el sistema inmunitario. Salvo en la rata, el plomo no
parece estar relacionado con el desarrollo de tumores.
8. Evaluación de los riesgos para la salud humana
El plomo tiene efectos adversos en varios órganos y sistemas de
órganos; los más delicados parecen ser los cambios subcelulares y los
efectos en el desarrollo del sistema nervioso. Se ha observado una
asociación entre el nivel de Pb-H y la hipertensión (presión
arterial). El plomo produce una serie de efectos en la reserva
corporal de hemo y afecta a la síntesis de éste. Sin embargo, algunos
de estos efectos no se consideran adversos. Está afectada la
homeostasia del calcio, lo que interfiere en otros procesos celulares.
a) Los datos más importantes de los estudios transversales y
prospectivos de poblaciones con niveles de Pb-H generalmente
inferiores a 1,2 µmoles/litro (25 µg/dl) se relacionan con una
disminución del coeficiente de inteligencia (CI). Es importante
señalar que esas observaciones no pueden constituir una prueba
concluyente de una relación causal con la exposición al plomo.
Sin embargo, la magnitud del efecto aparente en el CI,
determinado desde los 4 años en adelante, es un déficit de 0 a
5 puntos (en una escala con una desviación estándar de 15) por
cada 0,48 µmoles/litro (10 µg/dl) de aumento del nivel de Pb-H,
con una magnitud probable del efecto aparente de 1 a 3 puntos.
Con niveles de Pb-H superiores a 1,2 µmoles/litro (25 µg/dl), la
relación entre el Pb-H y el CI puede ser diferente. Las
estimaciones de la magnitud del efecto constituyen promedios
grupales y sólo se aplican a cada niño de manera probabilística.
Los estudios epidemiológicos existentes no prueban de modo
concluyente la existencia de un umbral. Por debajo de unos
niveles de Pb-H de 0,48-0,72 µmoles/litro (10-15 µg/dl), los
efectos de las variables de confusión y los límites de la
precisión de las mediciones analíticas y psicométricas aumentan
la incertidumbre inherente a toda estimación de un efecto. Sin
embargo, hay algunos indicios de una asociación por debajo de
dichos niveles.
b) Los estudios realizados en animales respaldan la idea de una
relación causal entre el plomo y ciertos efectos en el sistema
nervioso; se señalan deficiencias cognitivas a niveles de Pb-H de
sólo 0,53-0,72 µmoles/litro (11-15 µg/dl), deficiencias que
pueden persistir mucho después de haber terminado la exposición
al plomo.
c) Puede producirse una reducción de la velocidad de conducción
nerviosa periférica en el ser humano con niveles de Pb-H de sólo
1,44 µmoles/litro (30 µg/dl). Además, las funciones
sensitivomotrices pueden verse disminuidas con niveles de Pb-H de
sólo 1,92 µmoles/litro (40 µg/dl) aproximadamente y las funciones
del sistema nervioso autónomo (variabilidad del intervalo R-R
electrocardiográfico) pueden verse afectadas a un nivel promedio
de Pb-H de aproximadamente 1,68 µmoles por litro (35 µg/dl). El
riesgo de nefropatía plúmbica aumenta en los trabajadores que
tienen niveles de Pb-H superiores a 2,88 µmoles/litro (60 µg/dl).
Sin embargo, estudios recientes que utilizan indicadores más
sensibles de la función renal sugieren la aparición de efectos
renales a niveles más bajos de exposición al plomo.
d) La exposición al plomo está asociada a un pequeño aumento de la
presión arterial. El orden de magnitud probable es que por cada
duplicación del nivel de Pb-H (por ejemplo, de 0,8 a
1,6 µmoles/litro, es decir, de 16,6 a 33,3 µg/dl) hay un aumento
medio de 1 mmHg de presión arterial sistólica. La relación con la
presión diastólica es de una magnitud semejante pero más pequeña.
Sin embargo, no se sabe bien si estas asociaciones estadísticas
obedecen realmente a un efecto de la exposición al plomo o son un
resultado ficticio debido a factores de confusión.
e) Algunos estudios epidemiológicos, no todos, muestran una
relación, dependiente de la dosis, con el parto prematuro y
algunos índices de crecimiento y maduración fetales a niveles de
Pb-H de 0,72 µmoles/litro (15 µg/dl) o más.
f) Los indicios de carcinogenicidad del plomo y de varios compuestos
inorgánicos de plomo en el ser humano son insuficientes.
g) Se ha demostrado que el plomo tiene efectos en cierto número de
sistemas enzimáticos y de parámetros bioquímicos. Los niveles de
Pb-H por encima de los cuales las técnicas vigentes pueden
demostrar la presencia de efectos en relación con los parámetros
de importancia clínica posible son todos superiores a
0,96 µmoles/litro (20 µg/dl). Algunos efectos en enzimas pueden
demostrarse con niveles de Pb-H más bajos, pero su importancia
clínica es incierta.
