
IPCS INTERNATIONAL PROGRAMME ON CHEMICAL SAFETY
Health and Safety Guide No. 15
PROPYLENE OXIDE
HEALTH AND SAFETY GUIDE
UNITED NATIONS ENVIRONMENT PROGRAMME
INTERNATIONAL LABOUR ORGANISATION
WORLD HEALTH ORGANIZATION
WORLD HEALTH ORGANIZATION, GENEVA
This is a companion volume to Environmental Health Criteria 56:
Propylene Oxide
Published by the World Health Organization for the International
Programme on Chemical Safety (a collaborative programme of the
United Nations Environment Programme, the International Labour
Organisation, and the World Health Organization)
ISBN 92 4 154338 8
ISSN 0259 - 7268
(c) World Health Organization 1988
Publications of the World Health Organization enjoy copyright
protection in accordance with the provisions of Protocol 2 of the
Universal Copyright Convention. For rights of reproduction or
translation of WHO publications, in part or in toto, application
should be made to the Office of Publications, World Health
Organization, Geneva, Switzerland. The World Health Organization
welcomes such applications.
The designations employed and the presentation of the material in
this publication do not imply the expression of any opinion
whatsoever on the part of the Secretariat of the World Health
Organization concerning the legal status of any country, territory,
city or area or of its authorities, or concerning the delimitation
of its frontiers or boundaries.
The mention of specific companies or of certain manufacturers'
products does not imply that they are endorsed or recommended by the
World Health Organization in preference to others of a similar
nature that are not mentioned. Errors and omissions excepted, the
names of proprietary products are distinguished by initial capital
letters.
CONTENTS
INTRODUCTION
1. PRODUCT IDENTITY AND USES
1.1. Identity
1.2. Physical and chemical properties
1.3. Composition
1.4. Production and uses
2. SUMMARY AND EVALUATION
2.1. Human exposure
2.2. Fate in the environment
2.3. Uptake, metabolism, and excretion
2.4. Effects on organisms in the environment
2.5. Effects on animals
2.6. Effects on human beings
3. CONCLUSIONS AND RECOMMENDATIONS
4. HUMAN HEALTH HAZARDS, PREVENTION AND PROTECTION, EMERGENCY ACTION
4.1. Main human health hazards, prevention and protection, first aid
4.1.1. Advice to physicians
4.1.2. Health surveillance advice
4.2. Explosion and fire hazards
4.2.1. Explosion hazards
4.2.2. Fire hazards
4.2.2.1 Prevention
4.2.2.2 Fire-extinguishing agents
4.3. Storage
4.4. Transport
4.5. Spillage and disposal
4.5.1. Spillage
4.5.2. Disposal
5. HAZARDS FOR THE ENVIRONMENT AND THEIR PREVENTION
6. INTERNATIONAL CHEMICAL SAFETY CARD
7. CURRENT REGULATIONS, GUIDELINES, AND STANDARDS
7.1. Previous evaluations by international bodies
7.2. Exposure limit values
7.3. Specific restrictions
7.4. Labelling, packaging, and transport
7.5. Waste disposal
7.6. Other measures
BIBLIOGRAPHY
INTRODUCTION
The Environmental Health Criteria (EHC) documents produced by the
International Programme on Chemical Safety, include an assessment of
the effects on the environment and on human health of exposure to a
chemical or combination of chemicals, or physical or biological
agents. They also provide guidelines for setting exposure limits.
The purpose of a Health and Safety Guide is to facilitate the
application of these guidelines in national chemical safety
programmes. The first three sections of a Health and Safety Guide
highlight the relevant technical information in the corresponding
EHC. Section 4 includes advice on preventive and protective measures
and emergency action; health workers should be thoroughly familiar
with the medical information to ensure that they can act efficiently
in an emergency. Within the Guide is an International Chemical
Safety Card which should be readily available, and should be clearly
explained, to all who could come into contact with the chemical. The
section on Regulatory information has been extracted from the legal
file of the International Register of Potentially Toxic Chemicals
(IRPTC) and from other United Nations sources.
The target readership includes occupational health services, those
in ministries, governmental agencies, industry, and trade unions,
who are involved in the safe use of chemicals and the avoidance of
environmental health hazards and those wanting more information on
this topic. An attempt has been made to use only terms that will be
familiar to the intended user. However, it is inevitable that
sections 1 and 2 contain technical terms. A bibliography has been
included for readers who require further background information.
Revision of the information in this Guide will take place in due
course, and the eventual aim is to use standardized terminology.
Comments on any difficulties encountered in using the Guide would be
very helpful and should be addressed to:
The Manager
International Programme on Chemical Safety
Division of Environmental Health
World Health Organization
1211 Geneva 27
Switzerland
THE INFORMATION IN THIS GUIDE SHOULD BE CONSIDERED AS A STARTING
POINT TO A COMPREHENSIVE HEALTH AND SAFETY PROGRAMME
1. PRODUCT IDENTITY AND USES
1.1 Identity
Common name: Propylene oxide
Chemical formula: C3H6O
Chemical structure:
H O
' / \
H - C - C - C - H
' ' '
H H H
Common synonyms: 1,2-epoxypropane, methyl ethylene oxide,
methyl oxirane, propene oxide, propylene
epoxide, 1,2-propylene oxide
CAS registry number: 75-56-9
RTECS registry number: TZ2975000
UN number: 1280
Conversion factors: 1 ppm = 2.37 mg/m3
1 mg/m3 = 0.42 ppm at
25°C and 101.3 kPa (760 mmHg)
1.2 Physical and Chemical Properties
Propylene oxide is a colourless, highly volatile liquid at room
temperature and normal atmospheric pressure. It has an ethereal
odour with reported odour thresholds of 20 mg/m3 for perception
and 80-470 mg/m3 for recognition.
The liquid is highly flammable and the vapour forms an explosive
mixture with air. Propylene oxide may polymerize violently. It is
very reactive, particularly with chlorine, ammonia, strong oxidizing
agents, and acids. Some physical and chemical properties of
propylene oxide are listed on the International Chemical Safety Card
(pp. 20-23).
1.3 Composition
Common impurities that may be present in small amounts include
water, acetic acid, chloride, and aldehydes. Small amounts of
monochloroacetone, 1,2-dichloro-3-propanol, and propylene dichloride
can occur in the end product, when propylene oxide is manufactured
by the chlorohydrin process.
1.4 Production and Uses
World production of propylene oxide exceeds 2 million tonnes per
year. Large volume producers are found in Japan, the USA, and
Western Europe. Most of the compound is used as a chemical
intermediate. Small amounts are used for the sterilization of
medical equipment and for the fumigation of foodstuffs.
2. SUMMARY AND EVALUATION
2.1 Human Exposure
The main route of human exposure is through inhalation at the work
place. No published data have been found on ambient levels at a
distance from point sources. Eight-hour time-weighted average
occupational exposure levels are normally below 5 mg/m3, however,
peak exposures of up to 9000 mg/m3 have been recorded. Data are
insufficient to estimate the levels of exposure to propylene oxide
through residues in food after fumigation and sterilization. The
main conversion products in foodstuffs are chloropropanols and
1,2-propanediol, which are more persistent than the parent compound.
Analysis of fumigated foodstuffs revealed that chloropropanols and
1,2-propanediol were present at levels ranging from 4 to 47 mg/kg
and 29 to 2000 mg/kg, respectively. However, in pure fats, where
degradation is minimal, levels of propylene oxide of over 4000mg/kg
have been determined. Levels of up to 6260 mg/kg have been found in
wrapping materials.
2.2 Fate in the Environment
Propylene oxide enters the environment mainly through evaporation
and in vented gases during production, handling, storage, transport,
and use. The compound is removed from the atmosphere by slow
oxidation and by rain washout.
It is highly soluble in water, but is likely to evaporate to a great
extent. In neutral fresh water, propylene oxide is converted to
1,2-propanediol and, in marine waters, to halopropanols, but, even
in the presence of micro-organisms, these processes are slow.
Because of its limited lipid solubility, propylene oxide is unlikely
to bioaccumulate.
2.3 Uptake, Metabolism, and Excretion
Inhaled propylene oxide is readily absorbed, distributed throughout
the body, and rapidly metabolized. The half-life in the rat has been
estimated to be 40 min. Data are not available on skin absorption.
From in vitro studies, it would appear that propylene oxide is
metabolized by glutathione epoxide transferase to S-(2-hydroxy-
1-propyl)glutathione. It is converted to 1,2-propanediol by epoxide
hydrolase and non-enzymic hydrolysis, but both of these reactions
are slow. The diol can be oxidized to lactic and pyruvic acid.
2.4 Effects on Organisms in the Environment
The toxicity of propylene oxide for aquatic organisms is low.
Concentrations lethal for half the number of fish in populations
tested for 1-4 days were approximately 90 mg/litre (LC50) or
higher. The probability of adverse effects on the aquatic
environment is considered to be low.
2.5 Effects on Animals
Single oral doses of 520-1140 mg/kg body weight (LD50) or vapour
concentrations of 9500 mg/m3 (LC50) for 4 h have been shown to be
lethal for half the number of exposed rats. The compound is thus
slightly toxic in acute exposures, according to the scale of Hodge &
Sterner. Damage to the stomach mucosa and liver was observed after
lethal oral exposure, and severe eye and nose irritation, laboured
breathing, and central nervous system depression were observed after
lethal vapour exposure. Dogs exposed through inhalation, once for
4h, to a concentration of 3230-5880 mg/m3 showed salivation,
lachrymation, nasal discharge, and vomiting. Deaths were observed at
4750-5880 mg/m3, but not at 3230 mg/m3.
When various animal species were exposed to propylene oxide vapour,
for 7 h per day, 5 days per week, for 112-218 days, at
concentrations of 0, 240, 460, or 1080 mg/m3, rabbits and monkeys
did not show any adverse effects on appearance, mortality, growth,
and histopathology of internal organs. Rats and guinea-pigs showed
irritation of the respiratory passages and histological lung damage
at 1080 mg/m3. An increase in lung weight was observed in female
guinea-pigs at concentrations of 460 mg/m3 or more. No effects
were observed in any species at a concentration of 240 mg/m3. A
concentration of 3400 mg/m3 administered over 2 weeks, for 6 h per
day, 5 days a week, resulted in dyspnoea and death in rats. In the
same study, mice showed dyspnoea at 460 and 1150 mg/m3. Reduced
activity was observed in both species, and irregular limb movement
was seen in rats.
Rats and mice exposed for 2 years showed dose-related increased
incidences of inflammatory and proliferative lesions of the nasal
epithelium at an exposure level of 470 mg/m3. In rats, the
inflammatory lesions and hyperplasia were also observed at an
exposure level of 240 mg/m3, but not at 70 mg/m3.
Depression of the central nervous system, the severity of which
increased with increasing level and length of exposure, was observed
when rats and mice were exposed through inhalation to single high
concentrations of propylene oxide.
An aqueous solution of propylene oxide (100 or 200 g/litre) was
irritating to rabbit skin, when applied under occlusive cover.
Propylene oxide administered via inhalation for 2 years produced
ovarian atrophy in mice at 940 mg/m3. Testicular atrophy was found
at 240 and 720 mg/m3 in rats.
No sperm head abnormalities were detected in mice after exposure to
720 mg propylene oxide/m3 for 7 h/day, over 5 days, or in
cynomolgus monkeys exposed to 240 or 710 mg/m3 for 7 h/day and 5
days/week, over 2 years, but sperm count and motility were reduced,
and sperm drive range (time to traverse a linear path) increased in
the monkeys. Reduced sperm motility and damage to spermatocytes were
observed in male rats treated with an oral LD50 (520 mg/kg body
weight) dose. Reduced fertility was observed when these males were
mated with normal females between 2 and 10 weeks after exposure.
Reduced fetal ossification of vertebrae and ribs and wavy ribs were
observed when pregnant Sprague Dawley rats were exposed to 1190 mg
propylene oxide/m3, for 7 h/day, on days 1-16 of gestation. When
rats were additionally exposed for 3 weeks before mating, the
numbers of corpora lutea, implantations per dam, and live fetuses
were decreased. No teratogenic or fetotoxic effects were found in
New Zealand rabbits exposed to propylene oxide at 1190 mg/m3.
Propylene oxide is an alkylating agent. It is mutagenic in micro-
organisms and insects and produces DNA damage, mutations, and
chromosomal aberrations in mammalian cells in vitro. It produced
micronuclei in mouse erythrocytes after parenteral injection of 2
doses of 300 mg/kg body weight, but not after oral exposure. No
chromosomal aberrations or sister chromatid exchanges occurred in
monkeys exposed through inhalation to 237 or 717 mg propylene
oxide/m3, for 7 h per day, 5 days per week, for 2 years. No
dominant lethal effects were observed in rats or mice following
inhalation or oral exposure, respectively.
In long-term studies on the carcinogenicity of propylene oxide in
rats and mice, malignant tumours were mainly observed at the site of
entry into the body and included: squamous cell carcinomas of the
forestomach in rats administered oral doses of 15 or 60 mg/kg body
weight; local sarcomas in mice at subcutaneous doses of 1 and 2.5 mg
per mouse; nasal adenomas in rats exposed through inhalation at 940
or 717 mg/m3 (but not at 470 mg/m3); and mainly nasal
haemangiomas and haemangiosarcomas in mice at 940 mg/m3. At the
dose levels at which these tumours were induced, propylene oxide
produced localized tissue damage. In rats, the incidences of both
phaeochromocytomas in the adrenals and peritoneal mesotheliomas
increased following inhalation exposure to propylene oxide. It also
induced an increase in the incidence of mammary adenocarcinomas in
female rats following inhalation exposure at 940 mg/m3.
2.6 Effects on Human Beings
Corneal and conjunctival damage and allergic contact dermatitis have
been reported following accidental exposure to propylene oxide
solution.
Adequate data on chromosomal effects in human beings are not
available.
Adequate epidemiological studies on cancer incidence in exposed
populations have not been carried out.
Taking into account the available data, i.e., the alkylating nature
of propylene oxide, the formation of DNA adducts, the positive
responses in both in vitro and in vivo mutagenesis assays, the
carcinogenic effects on animals, predominantly at the sites of entry
into the body, and the absence of adequate data on cancer in human
beings, propylene oxide should be considered as a probable human
carcinogen, and levels in the environment should be kept as low as
feasible.
3. CONCLUSIONS AND RECOMMENDATIONS
On the basis of available data, propylene oxide should be
considered, for practical purposes, as a probable human carcinogen,
and exposure should be kept as low as possible.
The predominant long-term effects of inhalation may include
irritation of the nose and the other respiratory passages, lung
damage, and dyspnoea. Propylene oxide exhibits fetotoxicity in rats
and affects the spermatogenic function in monkeys. Common effects
following exposures to high vapour concentrations include irritation
of the eyes and respiratory tract, nasal discharge, dyspnoea, and
central nervous system depression. Damage to stomach mucosa and the
liver were observed after high oral exposures. A solution of
propylene oxide in water is irritating to the skin and may produce
allergic contact dermatitis.
The risk for the health of the general population from exposure to
ambient air concentrations of propylene oxide is likely to be
negligible.
4. HUMAN HEALTH HAZARDS, PREVENTION AND PROTECTION, EMERGENCY ACTION
4.1 Main Human Health Hazards, Prevention and Protection, First Aid
The human health hazards associated with certain types of exposure
to propylene oxide, preventive and protective measures, and first
aid recommendations are listed in the International Chemical Safety
Card on pp. 20-23.
4.1.1 Advice to Physicians
No specific antidote is known. Treat symptomatically. If skin
sensitization has developed because of exposure to propylene oxide,
further exposure should not be allowed. Be aware of the possibility
of delayed pulmonary oedema.
4.1.2 Health Surveillance Advice
Human beings potentially exposed to propylene oxide should undergo
periodic medical examination with particular emphasis given to the
respiratory tract, the reproductive system, skin, and eyes.
Physicians should be aware of the fact that propylene oxide is
carcinogenic in experimental animals and that it should be regarded,
for practical purposes, as if it represented a carcinogenic risk for
human beings.
Exposure to propylene oxide should be checked regularly by general
and/or personal monitoring.
4.2 Explosion and Fire Hazards
4.2.1 Explosion hazards
Propylene oxide-air mixtures containing 2-37% by volume of propylene
oxide may be explosive at or above -37°C and can be ignited by
sources of heat or ignition. The vapour, which is heavier than air,
may travel along the ground and be ignited from a distance.
The compound may polymerize violently. It reacts violently with many
compounds including chlorine and other oxidizing agents, ammonia,
acids, and chlorosulfonic acid.
4.2.2 Fire hazards
Propylene oxide is a highly flammable liquid.
4.2.2.1 Prevention
Use closed systems, ventilation, explosion-proof electrical
equipment and lighting, and spark-proof hand tools. Do not use
propylene oxide near sources of heat or ignition. Do not smoke. Do
not use compressed air for filling, discharging, or handling. Avoid
contact between the compound and incompatible materials (section
4.2.1).
In the case of fire, shut off the supply. If this is impossible and
there is no risk to the surroundings, let the fire burn itself out.
Firefighters should be equipped with self-contained breathing
apparatus, eye protection, and full protective clothing.
4.2.2.2 Fire-extinguishing agents
Use powder, alcohol-resistant foam, large amounts of water, halons,
or carbon dioxide.
4.3 Storage
Propylene oxide should be well-labelled and stored in a cool,
fire-roof, well-ventilated room. Keep away from any sources of
ignition or heat, and incompatible materials (section 4.2.1). It
should not be stored where food can become contaminated.
4.4 Transport
In case of accident, stop the engine. Remove all sources of
ignition. Keep bystanders at a distance and mark the roads. Keep
upwind. Evacuate the area endangered by poison gas. Wear full
protective clothing and self-contained breathing apparatus. In case
of spillage or fire, use the methods advised in sections 4.5 and
4.2, respectively. In case of poisoning, follow the advice in
section 4.1. Notify the police and the fire brigade immediately.
4.5 Spillage and Disposal
4.5.1 Spillage
Remove all ignition sources and evacuate the danger area; collect
leaking liquid in sealable containers; absorb spilled liquid in sand
or other inert absorbent and remove to a safe place. Ensure personal
protection by use of self-contained breathing apparatus and full
protective clothing.
4.5.2 Disposal
Discharge concentrated liquid wastes that do not contain peroxides,
at a controlled rate, near a pilot flame. For concentrated liquid
wastes that do contain peroxides, perforate the container from a
safe distance and burn the waste in an open area. Open burning and
evaporation are recommended for small amounts. Incineration may be
difficult in view of the high volatility of the compound.
5. HAZARDS FOR THE ENVIRONMENT AND THEIR PREVENTION
Propylene oxide will evaporate from water. Degradation in the
atmosphere and in water is slow. Bioaccumulation of propylene oxide
and its conversion products is unlikely.
The toxicity of propylene oxide for aquatic organisms is low. The
probability of an adverse impact on the aquatic environment is
considered low.
Contamination of soil, water, and the atmosphere can be avoided by
proper methods of storage, transport, handling, and waste disposal.
In the case of spillage, apply the methods recommended in section
4.5.1.
6. INTERNATIONAL CHEMICAL SAFETY CARD
This card should be easily available to all health workers
concerned with, and users of, propylene oxide. It should be
displayed at, or near, entrances to areas where there is potential
exposure to propylene oxide, and on processing equipment and
containers. The card should be translated into the appropriate
language(s). All persons potentially exposed to the chemical
should also have the instructions on the chemical safety card
clearly explained.
Space is available on the card for insertion of the National
Occupational Exposure Limit, the address and telephone number of
the National Poison Control Centre, and for local trade names.
PROPYLENE OXIDE
(PO, 1,2-epoxypropane, methyl oxirane, propene oxide)
PHYSICAL PROPERTIES OTHER CHARACTERISTICS
Relative molecular mass 58.08 Colourless liquid with ethereal odour; the vapour is
Appearance colourless liquid heavier than air, may travel along the ground and be
Odour ethereal ignited from a distance; the liquid may polymerize
Odour perception threshold 20 mg/m3 violently; propylene oxide reacts violently with
Melting point (°C) -112.1 many compounds including chlorine and other oxidizing
Boiling point (°C) 37.8 agents, ammonia, and chlorosulfonic acid; it
Solubility in water (20°C) (g/litre) 405can induce adverse effects well below the odour
Density (20°C) 0.83 g/ml threshold
Relative vapour density 2
Vapour pressure (20°C) 59 kPa
Flash point (open cup) -37°C
Flammable (explosive) limits (%) 2-37 %
log n-octanol/water partition
coefficient -0.13
HAZARDS/SYMPTOMS PREVENTION AND PROTECTION FIRST AID
SKIN: Severe irritation by Wear clean protective impervious Remove contaminated clothing and shoes
solutions in water gloves and boots, and clean immediately; rinse with water for at least
impermeable body-covering clothing 15 min; seek medical attention
EYES: Severe irritation by Wear safety face shield or eye Rinse opened eyes with water for
vapour and aqueous solutions protection in combination with at least 15 min; seek medical attention
breathing protection
INHALATION: Irritation of Use explosion-proof general or Remove victim to fresh air; keep victim
respiratory tract; dyspnoea; exhaust ventilation and, for quiet and warm; if breathing has stopped,
central nervous system depression non-routine activities, use a apply artificial respiration; transport
self-contained breathing apparatus to hospital
INGESTION: Irritation of Do not eat, drink, or smoke during Rinse mouth; give plenty of water to
stomach; effects on liver; work drink; do not induce vomiting;
abdominal pain transport to hospital
REPEATED EXPOSURE: Probable Exposure should be kept as low as
human mutagen and carcinogen; feasible
may pose a reproductive hazard
ENVIRONMENT: low toxicity Use proper methods of storage,
to aquatic life transport, waste disposal and
handling of spills (see below)
SPILLAGE STORAGEFIRE AND EXPLOSION
Remove all ignition sources; Store in a cool, well-labelled, Flammable; vapour-air mixtures may be
evacuate area; collect fire-proof, well-ventilated room; explosive; use closed systems, ventilation,
leaking liquid in sealable store away from foodstuffs and explosion-proof equipment; do
containers; absorb spilled liquid and incompatible materials not use compressed air for filling,
in sand or other inert discharging, or handling; no sources of
absorbent; ensure personal ignition or heat; extinguish fires with
protection by use of a self- powder, alcohol-resistant foam, lots of
contained breathing apparatus and water, halons, carbon dioxide
full protective clothing
WASTE DISPOSAL
Burn after ignition from a National Occupational Exposure UN: 1280
safe distance; evaporation Limit:
in an open area
National Poison Control Centre:
Local trade names
 7. CURRENT REGULATIONS, GUIDELINES, AND STANDARDS
The information given in this section has been extracted from the
International Register of Potentially Toxic Chemicals (IRPTC) legal
file and other United Nations sources. Its intention is to give the
reader a representative but non-exhaustive overview of current
regulations, guidelines, and standards.
The reader should be aware that regulatory decisions about chemicals
taken in a certain country can only be fully understood in the
framework of the legislation of that country.a
7.1 Previous Evaluations by International Bodies
An International Agency for Research on Cancer Working Group (IARC,
1985) evaluated the carcinogenicity of propylene oxide and concluded
that:
"There is sufficient evidence for the carcinogenicity of propylene
oxide to experimental animals; there is inadequate evidence for its
carcinogenicity to humans. It is noted that, in the absence of
adequate data in humans, it is reasonable, for practical purposes,
to regard chemicals for which there is sufficient evidence of
carcinogenicity in experimental animals as if they probably
represented a carcinogenic risk to humans." This conclusion was
confirmed in 1987.
7.2 Exposure Limit Values
Some exposure limit values are given in the table on pp. 28-29.
When no effective date appears in the IRPTC legal file, the year of
the reference from which the data are taken is indicated by (r).
7.3 Specific Restrictions
In the United Kingdom, it is prohibited to undertake any activity
that entails the use of 5 or more tons of propylene oxide on any
site or in any pipeline, unless the health and safety executive has
been notified at least 3 months before commencing activity.
Specified changes in activity of this type should be re-notified as
if it were a new activity (1983).
a The regulations and guidelines of all countries are subject to
change and should always be verified with appropriate regulatory
authorities before application.
In the Federal Republic of Germany, handling of propylene oxide is
prohibited or restricted for adolescents and pregnant or nursing
women (1980).
In Kenya, the compound is permitted as a starch-modifying agent. The
food products in or on which it is permitted and maximum levels of
use are listed (1982 (r)).
In the USA, the compound may be used to modify the food additive
"modified food starch" with certain restrictions, while residual
propylene chlorohydrin may not exceed 5 mg/kg (1983 (r)). The
substance is further exempted from tolerance requirements in plant,
meat, and animal products when used according to good agricultural
practice as an inert (or occasionally active) ingredient of
pesticides applied to growing crops, raw agricultural products after
harvest, or animals, for some specified purposes (1983 (r)).
7.4 Labelling, Packaging, and Transport
The European Community legislation requires labelling of propylene
oxide as a dangerous substance using the symbols:
7. CURRENT REGULATIONS, GUIDELINES, AND STANDARDS
The information given in this section has been extracted from the
International Register of Potentially Toxic Chemicals (IRPTC) legal
file and other United Nations sources. Its intention is to give the
reader a representative but non-exhaustive overview of current
regulations, guidelines, and standards.
The reader should be aware that regulatory decisions about chemicals
taken in a certain country can only be fully understood in the
framework of the legislation of that country.a
7.1 Previous Evaluations by International Bodies
An International Agency for Research on Cancer Working Group (IARC,
1985) evaluated the carcinogenicity of propylene oxide and concluded
that:
"There is sufficient evidence for the carcinogenicity of propylene
oxide to experimental animals; there is inadequate evidence for its
carcinogenicity to humans. It is noted that, in the absence of
adequate data in humans, it is reasonable, for practical purposes,
to regard chemicals for which there is sufficient evidence of
carcinogenicity in experimental animals as if they probably
represented a carcinogenic risk to humans." This conclusion was
confirmed in 1987.
7.2 Exposure Limit Values
Some exposure limit values are given in the table on pp. 28-29.
When no effective date appears in the IRPTC legal file, the year of
the reference from which the data are taken is indicated by (r).
7.3 Specific Restrictions
In the United Kingdom, it is prohibited to undertake any activity
that entails the use of 5 or more tons of propylene oxide on any
site or in any pipeline, unless the health and safety executive has
been notified at least 3 months before commencing activity.
Specified changes in activity of this type should be re-notified as
if it were a new activity (1983).
a The regulations and guidelines of all countries are subject to
change and should always be verified with appropriate regulatory
authorities before application.
In the Federal Republic of Germany, handling of propylene oxide is
prohibited or restricted for adolescents and pregnant or nursing
women (1980).
In Kenya, the compound is permitted as a starch-modifying agent. The
food products in or on which it is permitted and maximum levels of
use are listed (1982 (r)).
In the USA, the compound may be used to modify the food additive
"modified food starch" with certain restrictions, while residual
propylene chlorohydrin may not exceed 5 mg/kg (1983 (r)). The
substance is further exempted from tolerance requirements in plant,
meat, and animal products when used according to good agricultural
practice as an inert (or occasionally active) ingredient of
pesticides applied to growing crops, raw agricultural products after
harvest, or animals, for some specified purposes (1983 (r)).
7.4 Labelling, Packaging, and Transport
The European Community legislation requires labelling of propylene
oxide as a dangerous substance using the symbols:
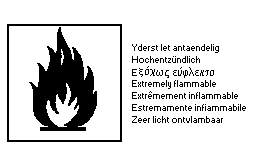
 The label must read:
extremely flammable; harmful by inhalation, in contact with
skin, and if swallowed; keep container in a well ventilated
place; keep away from sources of ignition - no smoking; in case
of contact with eyes, rinse immediately with plenty of water
and seek medical advice; do not empty into drains (1983).
The International Maritime Organisation and the United Nations
Committee of Experts on the Transportation of Dangerous Goods
classify propylene oxide as a flammable liquid (Hazard Class 3.1 and
3, respectively) and a very dangerous substance for packing purposes
(Packing Group I). Packing, transport methods, and label are
recommended (1982 (r)). The recommended symbol on the label is:
The label must read:
extremely flammable; harmful by inhalation, in contact with
skin, and if swallowed; keep container in a well ventilated
place; keep away from sources of ignition - no smoking; in case
of contact with eyes, rinse immediately with plenty of water
and seek medical advice; do not empty into drains (1983).
The International Maritime Organisation and the United Nations
Committee of Experts on the Transportation of Dangerous Goods
classify propylene oxide as a flammable liquid (Hazard Class 3.1 and
3, respectively) and a very dangerous substance for packing purposes
(Packing Group I). Packing, transport methods, and label are
recommended (1982 (r)). The recommended symbol on the label is:
 In the USA, propylene oxide, when carried in bulk, is classified as
a "cargo of particular hazard" for the purpose of regulations
governing handling of dangerous cargoes in or contiguous to water
front facilities. A permit is required for handling such cargo (1983
(r)).
7.5 Waste Disposal
In the Federal Republic of Germany, the air emissions of organic
compounds in Class III, carcinogens, which includes propylene oxide,
must not exceed (as the sum of all compounds in one class) a mass
concentration of 5 mg/m3 at a mass flow of greater than 25 kg/h
(1986 (r)).
In the USA, the substance is designated as a hazardous substance for
purposes of discharge (including spillage, leaking, etc.) under the
water pollution control act. A permit is required for discharge from
any point source into water in the USA. Under the Comprehensive
Environmental Response, Compensation, and Liability Act of 1980
(CERCLA), unless in compliance with a specified permit or procedure,
owners/operators of vessels or on- or offshore facilities must
notify the USA government (National Response Center) of any release
of the hazardous substance in or on navigable waters, adjoining
shorelines, the contiguous zone, or beyond the contiguous zone or to
any other environmental media (air, land or ground water) in an
amount equal to or more than 45.4 kg in any 24-h period (1985 (r)).
7.6 Other Measures
The European Community legislation concerning the major accident
hazards of certain industrial activities foresees that the
manufacturer must take all necessary measures to prevent accidents
and to limit their consequences for man and the environment when
processing propylene oxide, or when storing it in quantities equal
to or over 50 tonnes.
Furthermore, when propylene oxide is processed in quantities equal
to or over 50 tonnes or is stored in quantities equal to or over
300 tonnes, notification has to be made to the competent authorities
including information on the substance, on the installation,
information on possible major accident situations and emergency
plans (1984).
EXPOSURE LIMIT VALUES
Medium Specification Country/ Exposure limit descriptiona Value Effective
organization Date
AIR Occupational Australia Threshold limit value (TWA) 240 mg/m3 1985 (r)
Denmark Threshold limit value (TWA) 12 mg/m3 1985
Finland Exposure limit value (TWA) 240 mg/m3 1981
Germany, Maximum work-site concentration withdrawn 1986 (r)
Federal (carcinogenicity)
Republic of
Netherlands Maximum limit (TWA) 240 mg/m3 1985 (r)
Romania Maximum permissible concentration (TWA) 100 mg/m3 1985 (r)
Ceiling value 200 mg/m3 1985 (r)
Sweden Threshold limit value (TWA) 12 mg/m3 1985
Sweden Short-term exposure limit 25 mg/m3 1985
(15-min TWA)
United Kingdom Threshold limit value (TWA) 50 mg/m3b 1985 (r)
Short-term exposure limit 240 mg/m3b 1985 (r)
(10-min TWA)
USA (OSHA) Permissible exposure limit 240 mg/m3 1974
USA (AUEIH) Threshold limit value (TWA) 50 mg/m3 1976
USSR Ceiling value 1 mg/m3 1977
Medium Specification Country/ Exposure limit descriptiona Value Effective
organization Date
AIR Ambient USSR Maximum allowable concentration 0.08 mg/m3 1984
(once per day)
WATER Surface USSR Maximum allowable concentration 0.01 mg/litre 1983
Fishing USSR Maximum allowable concentration 0.005 mg/litre 1982 (r)
FOOD Specified USA Residue tolerance 300-700 mg/kg 1985 (r)
a TWA = time-weighted average over one working day (usually 8 h)
b Intended change.
BIBLIOGRAPHY
BRETHERICK L. (1981) Hazards in the chemical laboratory., 3rd ed.
London, The Royal Society of Chemistry.
CEC (1984) Classification and labelling of dangerous substances.
Brussels, Commission of the European Communities.
DUTCH CHEMICAL INDUSTRY ASSOCIATION (1980) Handling chemicals
safely., 2nd ed. Dutch Association of Safety Experts, Dutch Safety
Institute.
GOSSELIN R.E., SMITH R.P., ODGE H.C., & BRADDOCK; J.E. (1976)
Clinical toxicology of commercial products, 5th ed. Baltimore,
Maryland, Williams and Wilkins Company.
HOMMEL, G. (1987) [Handbook of dangerous goods.] 2nd ed. Berlin,
Springer Verlag (in German).
IARC (1972-present) IARC monographs on the evaluation of
carcinogenic risk of chemicals to man. Lyons, International Agency
for Research on Cancer.
IRPTC (1985) IRPTC file on treatment and disposal methods for waste
chemicals. Geneva, International Register for Potentially Toxic
Chemicals, United Nations Environment Programme.
IRPTC (1987) IRPTC legal file 1986. Geneva, International Register
of Potentially Toxic Chemicals, United Nations Environment
Programme.
IRPTC Data profile on individual chemical substances. Geneva,
International Register of Potentially Toxic Chemicals, United
Nations Environment Programme (unpublished file).
SAX, N.I. (1984) Dangerous properties of industrial materials. New
York, Van Nostrand Reinhold Company, Inc.
UNITED NATIONS (1986) Recommendations on the transport of dangerous
goods. 4th ed., New York, United Nations.
US NIOSH Current intelligence bulletins. Washington, DC, US
Department of Health, Education and Welfare.
US NIOSH (1976) A guide to industrial respiratory protection. US
National Institute for Occupational Safety and Health, pp. 76-189.
US NIOSH/OSHA (1985) Pocket guide to chemical hazards. Washington
DC, US Department of Health, Education and Welfare, US Department of
Labor (Publication No. 85.114).
WHO (1985) EHC No. 56: Propylene Oxide. Geneva, World Health
Organization.
In the USA, propylene oxide, when carried in bulk, is classified as
a "cargo of particular hazard" for the purpose of regulations
governing handling of dangerous cargoes in or contiguous to water
front facilities. A permit is required for handling such cargo (1983
(r)).
7.5 Waste Disposal
In the Federal Republic of Germany, the air emissions of organic
compounds in Class III, carcinogens, which includes propylene oxide,
must not exceed (as the sum of all compounds in one class) a mass
concentration of 5 mg/m3 at a mass flow of greater than 25 kg/h
(1986 (r)).
In the USA, the substance is designated as a hazardous substance for
purposes of discharge (including spillage, leaking, etc.) under the
water pollution control act. A permit is required for discharge from
any point source into water in the USA. Under the Comprehensive
Environmental Response, Compensation, and Liability Act of 1980
(CERCLA), unless in compliance with a specified permit or procedure,
owners/operators of vessels or on- or offshore facilities must
notify the USA government (National Response Center) of any release
of the hazardous substance in or on navigable waters, adjoining
shorelines, the contiguous zone, or beyond the contiguous zone or to
any other environmental media (air, land or ground water) in an
amount equal to or more than 45.4 kg in any 24-h period (1985 (r)).
7.6 Other Measures
The European Community legislation concerning the major accident
hazards of certain industrial activities foresees that the
manufacturer must take all necessary measures to prevent accidents
and to limit their consequences for man and the environment when
processing propylene oxide, or when storing it in quantities equal
to or over 50 tonnes.
Furthermore, when propylene oxide is processed in quantities equal
to or over 50 tonnes or is stored in quantities equal to or over
300 tonnes, notification has to be made to the competent authorities
including information on the substance, on the installation,
information on possible major accident situations and emergency
plans (1984).
EXPOSURE LIMIT VALUES
Medium Specification Country/ Exposure limit descriptiona Value Effective
organization Date
AIR Occupational Australia Threshold limit value (TWA) 240 mg/m3 1985 (r)
Denmark Threshold limit value (TWA) 12 mg/m3 1985
Finland Exposure limit value (TWA) 240 mg/m3 1981
Germany, Maximum work-site concentration withdrawn 1986 (r)
Federal (carcinogenicity)
Republic of
Netherlands Maximum limit (TWA) 240 mg/m3 1985 (r)
Romania Maximum permissible concentration (TWA) 100 mg/m3 1985 (r)
Ceiling value 200 mg/m3 1985 (r)
Sweden Threshold limit value (TWA) 12 mg/m3 1985
Sweden Short-term exposure limit 25 mg/m3 1985
(15-min TWA)
United Kingdom Threshold limit value (TWA) 50 mg/m3b 1985 (r)
Short-term exposure limit 240 mg/m3b 1985 (r)
(10-min TWA)
USA (OSHA) Permissible exposure limit 240 mg/m3 1974
USA (AUEIH) Threshold limit value (TWA) 50 mg/m3 1976
USSR Ceiling value 1 mg/m3 1977
Medium Specification Country/ Exposure limit descriptiona Value Effective
organization Date
AIR Ambient USSR Maximum allowable concentration 0.08 mg/m3 1984
(once per day)
WATER Surface USSR Maximum allowable concentration 0.01 mg/litre 1983
Fishing USSR Maximum allowable concentration 0.005 mg/litre 1982 (r)
FOOD Specified USA Residue tolerance 300-700 mg/kg 1985 (r)
a TWA = time-weighted average over one working day (usually 8 h)
b Intended change.
BIBLIOGRAPHY
BRETHERICK L. (1981) Hazards in the chemical laboratory., 3rd ed.
London, The Royal Society of Chemistry.
CEC (1984) Classification and labelling of dangerous substances.
Brussels, Commission of the European Communities.
DUTCH CHEMICAL INDUSTRY ASSOCIATION (1980) Handling chemicals
safely., 2nd ed. Dutch Association of Safety Experts, Dutch Safety
Institute.
GOSSELIN R.E., SMITH R.P., ODGE H.C., & BRADDOCK; J.E. (1976)
Clinical toxicology of commercial products, 5th ed. Baltimore,
Maryland, Williams and Wilkins Company.
HOMMEL, G. (1987) [Handbook of dangerous goods.] 2nd ed. Berlin,
Springer Verlag (in German).
IARC (1972-present) IARC monographs on the evaluation of
carcinogenic risk of chemicals to man. Lyons, International Agency
for Research on Cancer.
IRPTC (1985) IRPTC file on treatment and disposal methods for waste
chemicals. Geneva, International Register for Potentially Toxic
Chemicals, United Nations Environment Programme.
IRPTC (1987) IRPTC legal file 1986. Geneva, International Register
of Potentially Toxic Chemicals, United Nations Environment
Programme.
IRPTC Data profile on individual chemical substances. Geneva,
International Register of Potentially Toxic Chemicals, United
Nations Environment Programme (unpublished file).
SAX, N.I. (1984) Dangerous properties of industrial materials. New
York, Van Nostrand Reinhold Company, Inc.
UNITED NATIONS (1986) Recommendations on the transport of dangerous
goods. 4th ed., New York, United Nations.
US NIOSH Current intelligence bulletins. Washington, DC, US
Department of Health, Education and Welfare.
US NIOSH (1976) A guide to industrial respiratory protection. US
National Institute for Occupational Safety and Health, pp. 76-189.
US NIOSH/OSHA (1985) Pocket guide to chemical hazards. Washington
DC, US Department of Health, Education and Welfare, US Department of
Labor (Publication No. 85.114).
WHO (1985) EHC No. 56: Propylene Oxide. Geneva, World Health
Organization.
 7. CURRENT REGULATIONS, GUIDELINES, AND STANDARDS
The information given in this section has been extracted from the
International Register of Potentially Toxic Chemicals (IRPTC) legal
file and other United Nations sources. Its intention is to give the
reader a representative but non-exhaustive overview of current
regulations, guidelines, and standards.
The reader should be aware that regulatory decisions about chemicals
taken in a certain country can only be fully understood in the
framework of the legislation of that country.a
7.1 Previous Evaluations by International Bodies
An International Agency for Research on Cancer Working Group (IARC,
1985) evaluated the carcinogenicity of propylene oxide and concluded
that:
"There is sufficient evidence for the carcinogenicity of propylene
oxide to experimental animals; there is inadequate evidence for its
carcinogenicity to humans. It is noted that, in the absence of
adequate data in humans, it is reasonable, for practical purposes,
to regard chemicals for which there is sufficient evidence of
carcinogenicity in experimental animals as if they probably
represented a carcinogenic risk to humans." This conclusion was
confirmed in 1987.
7.2 Exposure Limit Values
Some exposure limit values are given in the table on pp. 28-29.
When no effective date appears in the IRPTC legal file, the year of
the reference from which the data are taken is indicated by (r).
7.3 Specific Restrictions
In the United Kingdom, it is prohibited to undertake any activity
that entails the use of 5 or more tons of propylene oxide on any
site or in any pipeline, unless the health and safety executive has
been notified at least 3 months before commencing activity.
Specified changes in activity of this type should be re-notified as
if it were a new activity (1983).
a The regulations and guidelines of all countries are subject to
change and should always be verified with appropriate regulatory
authorities before application.
In the Federal Republic of Germany, handling of propylene oxide is
prohibited or restricted for adolescents and pregnant or nursing
women (1980).
In Kenya, the compound is permitted as a starch-modifying agent. The
food products in or on which it is permitted and maximum levels of
use are listed (1982 (r)).
In the USA, the compound may be used to modify the food additive
"modified food starch" with certain restrictions, while residual
propylene chlorohydrin may not exceed 5 mg/kg (1983 (r)). The
substance is further exempted from tolerance requirements in plant,
meat, and animal products when used according to good agricultural
practice as an inert (or occasionally active) ingredient of
pesticides applied to growing crops, raw agricultural products after
harvest, or animals, for some specified purposes (1983 (r)).
7.4 Labelling, Packaging, and Transport
The European Community legislation requires labelling of propylene
oxide as a dangerous substance using the symbols:
7. CURRENT REGULATIONS, GUIDELINES, AND STANDARDS
The information given in this section has been extracted from the
International Register of Potentially Toxic Chemicals (IRPTC) legal
file and other United Nations sources. Its intention is to give the
reader a representative but non-exhaustive overview of current
regulations, guidelines, and standards.
The reader should be aware that regulatory decisions about chemicals
taken in a certain country can only be fully understood in the
framework of the legislation of that country.a
7.1 Previous Evaluations by International Bodies
An International Agency for Research on Cancer Working Group (IARC,
1985) evaluated the carcinogenicity of propylene oxide and concluded
that:
"There is sufficient evidence for the carcinogenicity of propylene
oxide to experimental animals; there is inadequate evidence for its
carcinogenicity to humans. It is noted that, in the absence of
adequate data in humans, it is reasonable, for practical purposes,
to regard chemicals for which there is sufficient evidence of
carcinogenicity in experimental animals as if they probably
represented a carcinogenic risk to humans." This conclusion was
confirmed in 1987.
7.2 Exposure Limit Values
Some exposure limit values are given in the table on pp. 28-29.
When no effective date appears in the IRPTC legal file, the year of
the reference from which the data are taken is indicated by (r).
7.3 Specific Restrictions
In the United Kingdom, it is prohibited to undertake any activity
that entails the use of 5 or more tons of propylene oxide on any
site or in any pipeline, unless the health and safety executive has
been notified at least 3 months before commencing activity.
Specified changes in activity of this type should be re-notified as
if it were a new activity (1983).
a The regulations and guidelines of all countries are subject to
change and should always be verified with appropriate regulatory
authorities before application.
In the Federal Republic of Germany, handling of propylene oxide is
prohibited or restricted for adolescents and pregnant or nursing
women (1980).
In Kenya, the compound is permitted as a starch-modifying agent. The
food products in or on which it is permitted and maximum levels of
use are listed (1982 (r)).
In the USA, the compound may be used to modify the food additive
"modified food starch" with certain restrictions, while residual
propylene chlorohydrin may not exceed 5 mg/kg (1983 (r)). The
substance is further exempted from tolerance requirements in plant,
meat, and animal products when used according to good agricultural
practice as an inert (or occasionally active) ingredient of
pesticides applied to growing crops, raw agricultural products after
harvest, or animals, for some specified purposes (1983 (r)).
7.4 Labelling, Packaging, and Transport
The European Community legislation requires labelling of propylene
oxide as a dangerous substance using the symbols:

 The label must read:
extremely flammable; harmful by inhalation, in contact with
skin, and if swallowed; keep container in a well ventilated
place; keep away from sources of ignition - no smoking; in case
of contact with eyes, rinse immediately with plenty of water
and seek medical advice; do not empty into drains (1983).
The International Maritime Organisation and the United Nations
Committee of Experts on the Transportation of Dangerous Goods
classify propylene oxide as a flammable liquid (Hazard Class 3.1 and
3, respectively) and a very dangerous substance for packing purposes
(Packing Group I). Packing, transport methods, and label are
recommended (1982 (r)). The recommended symbol on the label is:
The label must read:
extremely flammable; harmful by inhalation, in contact with
skin, and if swallowed; keep container in a well ventilated
place; keep away from sources of ignition - no smoking; in case
of contact with eyes, rinse immediately with plenty of water
and seek medical advice; do not empty into drains (1983).
The International Maritime Organisation and the United Nations
Committee of Experts on the Transportation of Dangerous Goods
classify propylene oxide as a flammable liquid (Hazard Class 3.1 and
3, respectively) and a very dangerous substance for packing purposes
(Packing Group I). Packing, transport methods, and label are
recommended (1982 (r)). The recommended symbol on the label is:
 In the USA, propylene oxide, when carried in bulk, is classified as
a "cargo of particular hazard" for the purpose of regulations
governing handling of dangerous cargoes in or contiguous to water
front facilities. A permit is required for handling such cargo (1983
(r)).
7.5 Waste Disposal
In the Federal Republic of Germany, the air emissions of organic
compounds in Class III, carcinogens, which includes propylene oxide,
must not exceed (as the sum of all compounds in one class) a mass
concentration of 5 mg/m3 at a mass flow of greater than 25 kg/h
(1986 (r)).
In the USA, the substance is designated as a hazardous substance for
purposes of discharge (including spillage, leaking, etc.) under the
water pollution control act. A permit is required for discharge from
any point source into water in the USA. Under the Comprehensive
Environmental Response, Compensation, and Liability Act of 1980
(CERCLA), unless in compliance with a specified permit or procedure,
owners/operators of vessels or on- or offshore facilities must
notify the USA government (National Response Center) of any release
of the hazardous substance in or on navigable waters, adjoining
shorelines, the contiguous zone, or beyond the contiguous zone or to
any other environmental media (air, land or ground water) in an
amount equal to or more than 45.4 kg in any 24-h period (1985 (r)).
7.6 Other Measures
The European Community legislation concerning the major accident
hazards of certain industrial activities foresees that the
manufacturer must take all necessary measures to prevent accidents
and to limit their consequences for man and the environment when
processing propylene oxide, or when storing it in quantities equal
to or over 50 tonnes.
Furthermore, when propylene oxide is processed in quantities equal
to or over 50 tonnes or is stored in quantities equal to or over
300 tonnes, notification has to be made to the competent authorities
including information on the substance, on the installation,
information on possible major accident situations and emergency
plans (1984).
EXPOSURE LIMIT VALUES
Medium Specification Country/ Exposure limit descriptiona Value Effective
organization Date
AIR Occupational Australia Threshold limit value (TWA) 240 mg/m3 1985 (r)
Denmark Threshold limit value (TWA) 12 mg/m3 1985
Finland Exposure limit value (TWA) 240 mg/m3 1981
Germany, Maximum work-site concentration withdrawn 1986 (r)
Federal (carcinogenicity)
Republic of
Netherlands Maximum limit (TWA) 240 mg/m3 1985 (r)
Romania Maximum permissible concentration (TWA) 100 mg/m3 1985 (r)
Ceiling value 200 mg/m3 1985 (r)
Sweden Threshold limit value (TWA) 12 mg/m3 1985
Sweden Short-term exposure limit 25 mg/m3 1985
(15-min TWA)
United Kingdom Threshold limit value (TWA) 50 mg/m3b 1985 (r)
Short-term exposure limit 240 mg/m3b 1985 (r)
(10-min TWA)
USA (OSHA) Permissible exposure limit 240 mg/m3 1974
USA (AUEIH) Threshold limit value (TWA) 50 mg/m3 1976
USSR Ceiling value 1 mg/m3 1977
Medium Specification Country/ Exposure limit descriptiona Value Effective
organization Date
AIR Ambient USSR Maximum allowable concentration 0.08 mg/m3 1984
(once per day)
WATER Surface USSR Maximum allowable concentration 0.01 mg/litre 1983
Fishing USSR Maximum allowable concentration 0.005 mg/litre 1982 (r)
FOOD Specified USA Residue tolerance 300-700 mg/kg 1985 (r)
a TWA = time-weighted average over one working day (usually 8 h)
b Intended change.
BIBLIOGRAPHY
BRETHERICK L. (1981) Hazards in the chemical laboratory., 3rd ed.
London, The Royal Society of Chemistry.
CEC (1984) Classification and labelling of dangerous substances.
Brussels, Commission of the European Communities.
DUTCH CHEMICAL INDUSTRY ASSOCIATION (1980) Handling chemicals
safely., 2nd ed. Dutch Association of Safety Experts, Dutch Safety
Institute.
GOSSELIN R.E., SMITH R.P., ODGE H.C., & BRADDOCK; J.E. (1976)
Clinical toxicology of commercial products, 5th ed. Baltimore,
Maryland, Williams and Wilkins Company.
HOMMEL, G. (1987) [Handbook of dangerous goods.] 2nd ed. Berlin,
Springer Verlag (in German).
IARC (1972-present) IARC monographs on the evaluation of
carcinogenic risk of chemicals to man. Lyons, International Agency
for Research on Cancer.
IRPTC (1985) IRPTC file on treatment and disposal methods for waste
chemicals. Geneva, International Register for Potentially Toxic
Chemicals, United Nations Environment Programme.
IRPTC (1987) IRPTC legal file 1986. Geneva, International Register
of Potentially Toxic Chemicals, United Nations Environment
Programme.
IRPTC Data profile on individual chemical substances. Geneva,
International Register of Potentially Toxic Chemicals, United
Nations Environment Programme (unpublished file).
SAX, N.I. (1984) Dangerous properties of industrial materials. New
York, Van Nostrand Reinhold Company, Inc.
UNITED NATIONS (1986) Recommendations on the transport of dangerous
goods. 4th ed., New York, United Nations.
US NIOSH Current intelligence bulletins. Washington, DC, US
Department of Health, Education and Welfare.
US NIOSH (1976) A guide to industrial respiratory protection. US
National Institute for Occupational Safety and Health, pp. 76-189.
US NIOSH/OSHA (1985) Pocket guide to chemical hazards. Washington
DC, US Department of Health, Education and Welfare, US Department of
Labor (Publication No. 85.114).
WHO (1985) EHC No. 56: Propylene Oxide. Geneva, World Health
Organization.
In the USA, propylene oxide, when carried in bulk, is classified as
a "cargo of particular hazard" for the purpose of regulations
governing handling of dangerous cargoes in or contiguous to water
front facilities. A permit is required for handling such cargo (1983
(r)).
7.5 Waste Disposal
In the Federal Republic of Germany, the air emissions of organic
compounds in Class III, carcinogens, which includes propylene oxide,
must not exceed (as the sum of all compounds in one class) a mass
concentration of 5 mg/m3 at a mass flow of greater than 25 kg/h
(1986 (r)).
In the USA, the substance is designated as a hazardous substance for
purposes of discharge (including spillage, leaking, etc.) under the
water pollution control act. A permit is required for discharge from
any point source into water in the USA. Under the Comprehensive
Environmental Response, Compensation, and Liability Act of 1980
(CERCLA), unless in compliance with a specified permit or procedure,
owners/operators of vessels or on- or offshore facilities must
notify the USA government (National Response Center) of any release
of the hazardous substance in or on navigable waters, adjoining
shorelines, the contiguous zone, or beyond the contiguous zone or to
any other environmental media (air, land or ground water) in an
amount equal to or more than 45.4 kg in any 24-h period (1985 (r)).
7.6 Other Measures
The European Community legislation concerning the major accident
hazards of certain industrial activities foresees that the
manufacturer must take all necessary measures to prevent accidents
and to limit their consequences for man and the environment when
processing propylene oxide, or when storing it in quantities equal
to or over 50 tonnes.
Furthermore, when propylene oxide is processed in quantities equal
to or over 50 tonnes or is stored in quantities equal to or over
300 tonnes, notification has to be made to the competent authorities
including information on the substance, on the installation,
information on possible major accident situations and emergency
plans (1984).
EXPOSURE LIMIT VALUES
Medium Specification Country/ Exposure limit descriptiona Value Effective
organization Date
AIR Occupational Australia Threshold limit value (TWA) 240 mg/m3 1985 (r)
Denmark Threshold limit value (TWA) 12 mg/m3 1985
Finland Exposure limit value (TWA) 240 mg/m3 1981
Germany, Maximum work-site concentration withdrawn 1986 (r)
Federal (carcinogenicity)
Republic of
Netherlands Maximum limit (TWA) 240 mg/m3 1985 (r)
Romania Maximum permissible concentration (TWA) 100 mg/m3 1985 (r)
Ceiling value 200 mg/m3 1985 (r)
Sweden Threshold limit value (TWA) 12 mg/m3 1985
Sweden Short-term exposure limit 25 mg/m3 1985
(15-min TWA)
United Kingdom Threshold limit value (TWA) 50 mg/m3b 1985 (r)
Short-term exposure limit 240 mg/m3b 1985 (r)
(10-min TWA)
USA (OSHA) Permissible exposure limit 240 mg/m3 1974
USA (AUEIH) Threshold limit value (TWA) 50 mg/m3 1976
USSR Ceiling value 1 mg/m3 1977
Medium Specification Country/ Exposure limit descriptiona Value Effective
organization Date
AIR Ambient USSR Maximum allowable concentration 0.08 mg/m3 1984
(once per day)
WATER Surface USSR Maximum allowable concentration 0.01 mg/litre 1983
Fishing USSR Maximum allowable concentration 0.005 mg/litre 1982 (r)
FOOD Specified USA Residue tolerance 300-700 mg/kg 1985 (r)
a TWA = time-weighted average over one working day (usually 8 h)
b Intended change.
BIBLIOGRAPHY
BRETHERICK L. (1981) Hazards in the chemical laboratory., 3rd ed.
London, The Royal Society of Chemistry.
CEC (1984) Classification and labelling of dangerous substances.
Brussels, Commission of the European Communities.
DUTCH CHEMICAL INDUSTRY ASSOCIATION (1980) Handling chemicals
safely., 2nd ed. Dutch Association of Safety Experts, Dutch Safety
Institute.
GOSSELIN R.E., SMITH R.P., ODGE H.C., & BRADDOCK; J.E. (1976)
Clinical toxicology of commercial products, 5th ed. Baltimore,
Maryland, Williams and Wilkins Company.
HOMMEL, G. (1987) [Handbook of dangerous goods.] 2nd ed. Berlin,
Springer Verlag (in German).
IARC (1972-present) IARC monographs on the evaluation of
carcinogenic risk of chemicals to man. Lyons, International Agency
for Research on Cancer.
IRPTC (1985) IRPTC file on treatment and disposal methods for waste
chemicals. Geneva, International Register for Potentially Toxic
Chemicals, United Nations Environment Programme.
IRPTC (1987) IRPTC legal file 1986. Geneva, International Register
of Potentially Toxic Chemicals, United Nations Environment
Programme.
IRPTC Data profile on individual chemical substances. Geneva,
International Register of Potentially Toxic Chemicals, United
Nations Environment Programme (unpublished file).
SAX, N.I. (1984) Dangerous properties of industrial materials. New
York, Van Nostrand Reinhold Company, Inc.
UNITED NATIONS (1986) Recommendations on the transport of dangerous
goods. 4th ed., New York, United Nations.
US NIOSH Current intelligence bulletins. Washington, DC, US
Department of Health, Education and Welfare.
US NIOSH (1976) A guide to industrial respiratory protection. US
National Institute for Occupational Safety and Health, pp. 76-189.
US NIOSH/OSHA (1985) Pocket guide to chemical hazards. Washington
DC, US Department of Health, Education and Welfare, US Department of
Labor (Publication No. 85.114).
WHO (1985) EHC No. 56: Propylene Oxide. Geneva, World Health
Organization.
