
FAO Nutrition Meetings
Report Series No. 40A,B,C
WHO/Food Add./67.29
TOXICOLOGICAL EVALUATION OF SOME
ANTIMICROBIALS, ANTIOXIDANTS, EMULSIFIERS,
STABILIZERS, FLOUR-TREATMENT AGENTS, ACIDS AND BASES
The content of this document is the result of the deliberations of the
Joint FAO/WHO Expert Committee on Food Additives which met at Rome,
13-20 December, 19651 Geneva, 11-18 October, 19662
1 Ninth Report of the Joint FAO/WHO Expert Committee on Food
Additives, FAO Nutrition Meetings Report Series, 1966 No. 40;
Wld Hlth Org. techn. Rep. Ser., 1966, 339
2 Tenth Report of the Joint FAO/WHO Expert Committee on Food
Additives, FAO Nutrition Meetings Report Series, 1967, in press;
Food and Agriculture Organization of the United Nations
World Health Organization
1967
BUTYLATED HYDROXYTOLUENE
Synonyms BHT
Chemical Name 2,6-ditertiarybutyl-p-cresol;
4-,methyl-2,6-ditertiarybutylphenol
Empirical formula C15H24O
Structural formula
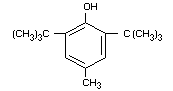 Molecular weight 220.36
Definition Butylated hydroxytoluene contains not
less than 99 per cent. of C15H24O.
Description Butylated hydroxytoluene is a white,
crystalline or flaked solid, having a
characteristic faint aromatic odour.
Use As an antioxidant.
Biological Data
The document entitled "Specifications for Identity and Purity and
Toxicological Evaluation of some Antimicrobials and Antioxidants"
(FAO/WHO, 1965) contains a monograph on BHT summarizing the data
available at the end of 1964. Since then a substantial amount of new
information has become available for consideration. This and other
data are summarized in the present monograph.
Biochemical aspects
Excretion and tissue distribution
The metabolism of butylated hydroxytoluene (BHT) administered to
rabbits orally in single doses of 500 mg/kg body-weight was studied.
The metabolites 2,6-di-tertbutyle-4-hydroxymethylphenol (BHT-alc),
3,5-di-tert-butyl-4-hydroxybenzoic acid (BHT-acid) and
4,4'-ethylene-bis-(2,6-di-tert-butylphenol) were identified. The
urinary metabolites of BHT comprised 37.5 per cent. as glucoronides,
16.7 per cent. as ethereal sulfates and 6.8 per cent. as free phenols;
unchanged BHT was present only in the faeces (Akagi & Aoki, 1962a);
3,5-di-tert-butyl-4-hydroxybenzaldehyde (BHT-ald) was also isolated
from rabbit urine (Aoki, 1962). The main metabolic pathway was
confirmed by administering BHT-alc to rabbits and isolating BHT-ald,
BHT-acid, the ethylene-bis derative and unchanged BHT-alc in the
urine (Akagi & Aoki, 1962b).
The fate in the body of 14C-labelled BHT has been elucidated.
The relatively slow excretion of BHT Is probably attributable to
enterohepatic circulation rather than to tissue retention. Rats were
given single oral doses (1-100 mg/rat) of BHT-14C and approximately
80-90 per cent. of the dose was recovered in 4 days in the urine and
faeces. Of the total radioactivity, 40 per cent. appeared in the urine
of females and 25 per cent. in males. After 4 days approximately 3.8
per cent. of the dose was retained mainly in the alimentary tract. A
substantial portion of the radioactivity was found in the bile
collected from two rats (1 male, 1 female) over a period of 40 hours
(Daniel & Gage, 1965).
Groups of two rats (1 male, 1 female) were given 1-5 oral doses
of 44 mg/kg body-weight BHT on alternate days and each group killed 24
hours after the final dose. The range of the total dose accounted for
was 92-103.5 per cent. in males and 92.6-98.6 per cent. in females.
There was an indication of sex difference in the route of excretion,
females excreting 19-43 per cent. of the radioactivity in urine and
males only 3-15 per cent. Eight days after administration of 5 doses
92 per cent. of the radioactivity had been excreted by males and 97
per cent. by females. Subcutaneous administration of graded doses of
BHT to female rats revealed substantial faecal excretion but the rate
of excretion decreased with increasing dose. There was no evidence of
accumulation of BHT-14C in the body under the conditions of repeated
oral dosage (Tye at al., 1965).
The BHT content of fat and liver of rats given diets containing
0.5 and 1.0 per cent. BHT for periods up to 35 and 50 days
respectively: with 0.5 per cent. BHT in the diet, a level of
approximately 30 ppm in the fat was reached in males and 45 ppm in
females, with approximately 1-3 ppm in the liver, while with 1.0 per
cent. BHT the level in the fat was 50 ppm in males and 30 ppm in
females. On cessation of treatment, the level of BHT in fat fell with
a half-life of 7-10 days (Daniel & Gage, 1965). The level of BHT in
the fat reached a plateau at approximately 100 ppm after 3-4 days when
daily doses of 500 mg/kg body-weight were given by intubation; 200
mg/kg body-weight/day for 1 week produced a level of about 50 ppm
(Gilbert & Golberg, 1965).
When feed containing 500 ppm BHT was given to laying hens, 20 ppm
was found in the fat fraction of eggs; 100 ppm in the feed resulted in
residues of less than 5 ppm. In the broiler chicken, over a period of
21 weeks, the residues in body fat were 55 ppm on the 500 ppm diet and
less than 5 ppm on the 100 ppm diet (Van Stratum & Vos, 1965).
One-day-old chicks were given 14C-BHT at a level of 200 ppm in
the feed for 10 weeks. At broiler age, edible portions had residues
amounting to 1-3 ppm of BHT and metabolites. Similar diets given to
laying hens produced residues in eggs of 2 ppm after 7 days, the level
thereafter remaining constant (Frawley et al., 1965a).
Stimulation of liver microsomal processing enzymes
Rats given BHT by daily intubation showed increased activity of
some liver microsomal enzymes. Stimulation of enzyme activity
correlated with an increase in relative liver weight, the threshold
dose for these changes in enzyme activity in female rats being below
25-75 mg BHT/kg body-weight/day. The storage of BHT in fat appeared to
be influenced by the activity of the processing enzymes. In rats given
500 mg/kg body-weight daily the level of BHT in fat attained values of
230 ppm in females and 162 ppm in males by the second day, by which
time the relative liver weight and processing enzyme activities had
become elevated. Thereafter, liver weight and enzyme activities
continued to rise but the BHT content of fat fell to a plateau of
about 100 ppm in both sexes (Gilbert & Golberg, 1965).
In further work with rats it was found that increased output of
urinary ascorbic acid paralleled liver enlargement induced by BHA or
BHT in onset, degree and duration, being rapid but transient with BHA
and slower in onset but more prolonged with BHT (Gaunt et al., 1965a).
The parallelism between stimulation of processing enzyme activity,
increase in urinary ascorbic acid output, and increase in relative
liver weight brought about by BHT was unaffected by 14 days of dietary
restriction, and all these changes except liver weight were reversible
during 14 days' recovery on normal diet (Gaunt et al., 1965b).
Special studies
Diets containing 0.1 or 0.5 per cent. BHT together with two
dietary levels of lard (10 and 20 per cent.) were given to mice. The
0.5 per cent. level of BHT produced slight but significant reduction
in mean pup weight and total litter weight at 22 days of age. The 0.1
per cent. level of BHT had no such effect. Out. of 7754 mice born,
none showed anophthalmia, although 12 out of the 144 mothers were
selected from an established anophthalmic strain (Johnson, 1965).
In a study on the embryotoxicity of BHT three dosing schedules
were employed: single doses (1000 mg/kg body-weight) on a specific day
of gestation, repeated daily doses (250-500 mg/kg body-weight) from
the time of mating throughout pregnancy and daily doses (250-500 mg/kg
body-weight for mice and 500 and 700 mg/kg body-weight for rats)
during a 7-10 week period before mating, continuing throughout mating
and gestation up to the time the animals were killed. No significant
embryotoxic effects were observed on examination of the skeletal and
soft tissue of the fully developed foetuses as well as by other
criteria. Reproduction and post-natal development were also
unaffected (Clegg, 1965)
Acute toxicity
Animal Route LD50 Reference
(mg/kg
body-weight)
Rat oral 2 450 Karplyuk, 1960
Mouse oral 2 000 Karplyuk, 1960
Short-term studies
Rat. BHT (2000 ppm) incorporated in a diet containing 19.9 per
cent. casein was administered to a group of 8 weeks; a further group
of 8 rats served as controls. The experiment was repeated with 16.6
per cent. casein in the diet of further groups for 4 weeks and again
with 9.6 per cent. casein (and no added choline) for 7 weeks. In all 3
instances BHT caused stimulation of growth and improved protein
efficiency. The N content of the liver was, however, greatly reduced
in BHT-treated animals, except when the level of BHT was reduced to
200 ppm. Recovery of hepatic protein after fasting (details not given)
was also impared in rats on 2000 ppm BHT. Liver lipid content was
increased with 2000 ppm but not with 200 ppm BHT. A dietary level of
2000 ppm BHT also increased the adrenal weight and ascorbic acid
content, although if recalculated on the basis of weight of gland,
there was no significant difference. The increase in adrenal ascorbic
acid is interpreted as indicating a stress imposed on the organism by
BHT (Sporn & Schöbesch, 1961).
Groups of 48 weanling rats (24 of each sex) were given diets
containing 1000 ppm BHT for periods of up to 16 weeks. A group of 48
rats served as controls. Measurements of growth rate, food
consumption, weight and micropathological examination of organs at
autopsy revealed no difference from untreated rats. However, increase
in relative liver weight and in the weight of the adrenals was
produced without histopathological evidence of damage. Biochemical
measurement and histochemical assessments of liver glucose
-phosphatase and glucose 6-phosphate dehydrogenase activities revealed
no difference from the control group (Gaunt at al., 1965a).
Multigeneration study
Weanling rats (16 of each sex) were fed a diet containing 20 per
cent. lard and 0, 300, 1000 or 3000 ppm BHT and mated at 100 days of
age (79 days on test). Ten days after weaning of the first litter the
animals were again mated to produce a second litter. The offspring (16
females and 8 males) ware mated at 100 days of age. Numerous function
and clinical tests including serum cholesterols and lipids were
performed on the parents and the first filial generation up to 28
weeks and gross and microscopical examination at 42 weeks. At the 3000
ppm dietary level a 10-20 per cent. reduction in growth rate of
parents and offspring was observed. A 20 per cent. elevation of serum
cholesterol levels was observed after 28 weeks, but no cholesterol
elevation after 10 weeks. A 10-20 per cent. increase in relative liver
weight was also observed upon killing after 42 weeks on diet. All
other observations at 3000 ppm and all observations at 1000 ppm and
300 ppm were comparable with control. All criteria of reproduction
were normal. No teratogenic effects ware detected (Frawley et al.,
1965b). Similar results to those obtained with the parental and first
filial generations were also obtained with the second filial
generation. Examination of two litters obtained from the latter at 100
days of age revealed no effects except a reduction of mean body-weight
at the 3000 ppm level. The offspring were examined for: litter size,
mean body-weight, occurrence of still-birth, survival rate and gross
and microscopic pathology (Frawley, 1967).
Comments
After single and repeated oral doses BHT is eliminated almost
completely in four days in the urine and faeces. The relatively slower
rate of excretion than that shown by BHA could be due to enterohepatic
circulation. Results of the analysis of tissues of rats suggest that
unchanged BHT does not tend to accumulate in the body.
The browning, thinning and loss of hair reported in one strain of
rat has not been reproduced in other strains. Reproduction studies
carried out on several strains of rats and mice fail to reveal
anophthalmia in the offspring reported earlier in a different strain
of rats.
Diet containing 20 per cent. lard and 1000 ppm BHT has no effect
on serum cholesterol and lipid levels, relative liver and other organ
weights, liver lipids and growth rate.
Metabolic studies in man are desirable.
Evaluation
Level causing no toxicological effect
Rat. 1000 ppm in the diet, equivalent to 50 mg/kg
body-weight/day.
Estimate of acceptable daily intake for man
mg/kg body-weight1
Unconditional acceptance 0-0.5
Conditional acceptance 0.5-2
REFERENCES
Akagi, M. & Aoki, I. (1962a) Chem, Pharm. Bull. (Tokyo), 10, 101
Akagi, M. & Aoki, I. (1962b) Chem. Pharm, Bull. (Tokyo), 10, 200
Aoki, I. (1962) Chem. Pharm. Bull. (Tokyo), 10, 105
Clegg, D. J. (1965)Fd Cosmet Toxicol., 3, 387
Daniel, J. W. & Gage, J. C. (1965) Fd Cosmet. Toxicol., 3, 405
FAO/WHO (1965) FAO Nutrition Meetings Report Series, No. 38;
WHO/Food Add./24.65
Frawley, J. P. (1967) Unpublished report submitted to WHO
Frawley, J. P., Kay, J. M. & Calandra, J. C. (1965a) Fd Cosmet.
Toxicol., 3, 471
Frawley, J. P., Kohn, F.E. Kay, J. H. & Calandra, J. C. (1965b)
Fd Cosmet. Toxicol., 3, 377
Gaunt. I. F., Feuer, G., Fairweather, F. A. & Gilbert, D. (1965a)
Fd Cosmet.Toxicol., 3, 433
Gaunt, I. F., Gilbert, D. & Martin, D. (1965b) Fd Cosmet. Toxicol.,
3, 445
Gilbert, D. & Golberg, L. (1965) Fd Cosmet. Toxicol., 3, 417
1 As sum of BHA and BHT.
Johnson, A. R. (1965) Fd Cosmet. Toxicol., 3, 417
Sporn, A. & Schöbesch, O. (1961) Igiena (Bucharest), 9, 113
van Stratum, D. G. C. & Vos, H. J. (1965) Fd Cosmet. Toxicol., 3,
475
Tye, R., Engel, J. D. & Rapion, I. (1965) Fd Cosmet. Toxicol., 3,
475
Molecular weight 220.36
Definition Butylated hydroxytoluene contains not
less than 99 per cent. of C15H24O.
Description Butylated hydroxytoluene is a white,
crystalline or flaked solid, having a
characteristic faint aromatic odour.
Use As an antioxidant.
Biological Data
The document entitled "Specifications for Identity and Purity and
Toxicological Evaluation of some Antimicrobials and Antioxidants"
(FAO/WHO, 1965) contains a monograph on BHT summarizing the data
available at the end of 1964. Since then a substantial amount of new
information has become available for consideration. This and other
data are summarized in the present monograph.
Biochemical aspects
Excretion and tissue distribution
The metabolism of butylated hydroxytoluene (BHT) administered to
rabbits orally in single doses of 500 mg/kg body-weight was studied.
The metabolites 2,6-di-tertbutyle-4-hydroxymethylphenol (BHT-alc),
3,5-di-tert-butyl-4-hydroxybenzoic acid (BHT-acid) and
4,4'-ethylene-bis-(2,6-di-tert-butylphenol) were identified. The
urinary metabolites of BHT comprised 37.5 per cent. as glucoronides,
16.7 per cent. as ethereal sulfates and 6.8 per cent. as free phenols;
unchanged BHT was present only in the faeces (Akagi & Aoki, 1962a);
3,5-di-tert-butyl-4-hydroxybenzaldehyde (BHT-ald) was also isolated
from rabbit urine (Aoki, 1962). The main metabolic pathway was
confirmed by administering BHT-alc to rabbits and isolating BHT-ald,
BHT-acid, the ethylene-bis derative and unchanged BHT-alc in the
urine (Akagi & Aoki, 1962b).
The fate in the body of 14C-labelled BHT has been elucidated.
The relatively slow excretion of BHT Is probably attributable to
enterohepatic circulation rather than to tissue retention. Rats were
given single oral doses (1-100 mg/rat) of BHT-14C and approximately
80-90 per cent. of the dose was recovered in 4 days in the urine and
faeces. Of the total radioactivity, 40 per cent. appeared in the urine
of females and 25 per cent. in males. After 4 days approximately 3.8
per cent. of the dose was retained mainly in the alimentary tract. A
substantial portion of the radioactivity was found in the bile
collected from two rats (1 male, 1 female) over a period of 40 hours
(Daniel & Gage, 1965).
Groups of two rats (1 male, 1 female) were given 1-5 oral doses
of 44 mg/kg body-weight BHT on alternate days and each group killed 24
hours after the final dose. The range of the total dose accounted for
was 92-103.5 per cent. in males and 92.6-98.6 per cent. in females.
There was an indication of sex difference in the route of excretion,
females excreting 19-43 per cent. of the radioactivity in urine and
males only 3-15 per cent. Eight days after administration of 5 doses
92 per cent. of the radioactivity had been excreted by males and 97
per cent. by females. Subcutaneous administration of graded doses of
BHT to female rats revealed substantial faecal excretion but the rate
of excretion decreased with increasing dose. There was no evidence of
accumulation of BHT-14C in the body under the conditions of repeated
oral dosage (Tye at al., 1965).
The BHT content of fat and liver of rats given diets containing
0.5 and 1.0 per cent. BHT for periods up to 35 and 50 days
respectively: with 0.5 per cent. BHT in the diet, a level of
approximately 30 ppm in the fat was reached in males and 45 ppm in
females, with approximately 1-3 ppm in the liver, while with 1.0 per
cent. BHT the level in the fat was 50 ppm in males and 30 ppm in
females. On cessation of treatment, the level of BHT in fat fell with
a half-life of 7-10 days (Daniel & Gage, 1965). The level of BHT in
the fat reached a plateau at approximately 100 ppm after 3-4 days when
daily doses of 500 mg/kg body-weight were given by intubation; 200
mg/kg body-weight/day for 1 week produced a level of about 50 ppm
(Gilbert & Golberg, 1965).
When feed containing 500 ppm BHT was given to laying hens, 20 ppm
was found in the fat fraction of eggs; 100 ppm in the feed resulted in
residues of less than 5 ppm. In the broiler chicken, over a period of
21 weeks, the residues in body fat were 55 ppm on the 500 ppm diet and
less than 5 ppm on the 100 ppm diet (Van Stratum & Vos, 1965).
One-day-old chicks were given 14C-BHT at a level of 200 ppm in
the feed for 10 weeks. At broiler age, edible portions had residues
amounting to 1-3 ppm of BHT and metabolites. Similar diets given to
laying hens produced residues in eggs of 2 ppm after 7 days, the level
thereafter remaining constant (Frawley et al., 1965a).
Stimulation of liver microsomal processing enzymes
Rats given BHT by daily intubation showed increased activity of
some liver microsomal enzymes. Stimulation of enzyme activity
correlated with an increase in relative liver weight, the threshold
dose for these changes in enzyme activity in female rats being below
25-75 mg BHT/kg body-weight/day. The storage of BHT in fat appeared to
be influenced by the activity of the processing enzymes. In rats given
500 mg/kg body-weight daily the level of BHT in fat attained values of
230 ppm in females and 162 ppm in males by the second day, by which
time the relative liver weight and processing enzyme activities had
become elevated. Thereafter, liver weight and enzyme activities
continued to rise but the BHT content of fat fell to a plateau of
about 100 ppm in both sexes (Gilbert & Golberg, 1965).
In further work with rats it was found that increased output of
urinary ascorbic acid paralleled liver enlargement induced by BHA or
BHT in onset, degree and duration, being rapid but transient with BHA
and slower in onset but more prolonged with BHT (Gaunt et al., 1965a).
The parallelism between stimulation of processing enzyme activity,
increase in urinary ascorbic acid output, and increase in relative
liver weight brought about by BHT was unaffected by 14 days of dietary
restriction, and all these changes except liver weight were reversible
during 14 days' recovery on normal diet (Gaunt et al., 1965b).
Special studies
Diets containing 0.1 or 0.5 per cent. BHT together with two
dietary levels of lard (10 and 20 per cent.) were given to mice. The
0.5 per cent. level of BHT produced slight but significant reduction
in mean pup weight and total litter weight at 22 days of age. The 0.1
per cent. level of BHT had no such effect. Out. of 7754 mice born,
none showed anophthalmia, although 12 out of the 144 mothers were
selected from an established anophthalmic strain (Johnson, 1965).
In a study on the embryotoxicity of BHT three dosing schedules
were employed: single doses (1000 mg/kg body-weight) on a specific day
of gestation, repeated daily doses (250-500 mg/kg body-weight) from
the time of mating throughout pregnancy and daily doses (250-500 mg/kg
body-weight for mice and 500 and 700 mg/kg body-weight for rats)
during a 7-10 week period before mating, continuing throughout mating
and gestation up to the time the animals were killed. No significant
embryotoxic effects were observed on examination of the skeletal and
soft tissue of the fully developed foetuses as well as by other
criteria. Reproduction and post-natal development were also
unaffected (Clegg, 1965)
Acute toxicity
Animal Route LD50 Reference
(mg/kg
body-weight)
Rat oral 2 450 Karplyuk, 1960
Mouse oral 2 000 Karplyuk, 1960
Short-term studies
Rat. BHT (2000 ppm) incorporated in a diet containing 19.9 per
cent. casein was administered to a group of 8 weeks; a further group
of 8 rats served as controls. The experiment was repeated with 16.6
per cent. casein in the diet of further groups for 4 weeks and again
with 9.6 per cent. casein (and no added choline) for 7 weeks. In all 3
instances BHT caused stimulation of growth and improved protein
efficiency. The N content of the liver was, however, greatly reduced
in BHT-treated animals, except when the level of BHT was reduced to
200 ppm. Recovery of hepatic protein after fasting (details not given)
was also impared in rats on 2000 ppm BHT. Liver lipid content was
increased with 2000 ppm but not with 200 ppm BHT. A dietary level of
2000 ppm BHT also increased the adrenal weight and ascorbic acid
content, although if recalculated on the basis of weight of gland,
there was no significant difference. The increase in adrenal ascorbic
acid is interpreted as indicating a stress imposed on the organism by
BHT (Sporn & Schöbesch, 1961).
Groups of 48 weanling rats (24 of each sex) were given diets
containing 1000 ppm BHT for periods of up to 16 weeks. A group of 48
rats served as controls. Measurements of growth rate, food
consumption, weight and micropathological examination of organs at
autopsy revealed no difference from untreated rats. However, increase
in relative liver weight and in the weight of the adrenals was
produced without histopathological evidence of damage. Biochemical
measurement and histochemical assessments of liver glucose
-phosphatase and glucose 6-phosphate dehydrogenase activities revealed
no difference from the control group (Gaunt at al., 1965a).
Multigeneration study
Weanling rats (16 of each sex) were fed a diet containing 20 per
cent. lard and 0, 300, 1000 or 3000 ppm BHT and mated at 100 days of
age (79 days on test). Ten days after weaning of the first litter the
animals were again mated to produce a second litter. The offspring (16
females and 8 males) ware mated at 100 days of age. Numerous function
and clinical tests including serum cholesterols and lipids were
performed on the parents and the first filial generation up to 28
weeks and gross and microscopical examination at 42 weeks. At the 3000
ppm dietary level a 10-20 per cent. reduction in growth rate of
parents and offspring was observed. A 20 per cent. elevation of serum
cholesterol levels was observed after 28 weeks, but no cholesterol
elevation after 10 weeks. A 10-20 per cent. increase in relative liver
weight was also observed upon killing after 42 weeks on diet. All
other observations at 3000 ppm and all observations at 1000 ppm and
300 ppm were comparable with control. All criteria of reproduction
were normal. No teratogenic effects ware detected (Frawley et al.,
1965b). Similar results to those obtained with the parental and first
filial generations were also obtained with the second filial
generation. Examination of two litters obtained from the latter at 100
days of age revealed no effects except a reduction of mean body-weight
at the 3000 ppm level. The offspring were examined for: litter size,
mean body-weight, occurrence of still-birth, survival rate and gross
and microscopic pathology (Frawley, 1967).
Comments
After single and repeated oral doses BHT is eliminated almost
completely in four days in the urine and faeces. The relatively slower
rate of excretion than that shown by BHA could be due to enterohepatic
circulation. Results of the analysis of tissues of rats suggest that
unchanged BHT does not tend to accumulate in the body.
The browning, thinning and loss of hair reported in one strain of
rat has not been reproduced in other strains. Reproduction studies
carried out on several strains of rats and mice fail to reveal
anophthalmia in the offspring reported earlier in a different strain
of rats.
Diet containing 20 per cent. lard and 1000 ppm BHT has no effect
on serum cholesterol and lipid levels, relative liver and other organ
weights, liver lipids and growth rate.
Metabolic studies in man are desirable.
Evaluation
Level causing no toxicological effect
Rat. 1000 ppm in the diet, equivalent to 50 mg/kg
body-weight/day.
Estimate of acceptable daily intake for man
mg/kg body-weight1
Unconditional acceptance 0-0.5
Conditional acceptance 0.5-2
REFERENCES
Akagi, M. & Aoki, I. (1962a) Chem, Pharm. Bull. (Tokyo), 10, 101
Akagi, M. & Aoki, I. (1962b) Chem. Pharm, Bull. (Tokyo), 10, 200
Aoki, I. (1962) Chem. Pharm. Bull. (Tokyo), 10, 105
Clegg, D. J. (1965)Fd Cosmet Toxicol., 3, 387
Daniel, J. W. & Gage, J. C. (1965) Fd Cosmet. Toxicol., 3, 405
FAO/WHO (1965) FAO Nutrition Meetings Report Series, No. 38;
WHO/Food Add./24.65
Frawley, J. P. (1967) Unpublished report submitted to WHO
Frawley, J. P., Kay, J. M. & Calandra, J. C. (1965a) Fd Cosmet.
Toxicol., 3, 471
Frawley, J. P., Kohn, F.E. Kay, J. H. & Calandra, J. C. (1965b)
Fd Cosmet. Toxicol., 3, 377
Gaunt. I. F., Feuer, G., Fairweather, F. A. & Gilbert, D. (1965a)
Fd Cosmet.Toxicol., 3, 433
Gaunt, I. F., Gilbert, D. & Martin, D. (1965b) Fd Cosmet. Toxicol.,
3, 445
Gilbert, D. & Golberg, L. (1965) Fd Cosmet. Toxicol., 3, 417
1 As sum of BHA and BHT.
Johnson, A. R. (1965) Fd Cosmet. Toxicol., 3, 417
Sporn, A. & Schöbesch, O. (1961) Igiena (Bucharest), 9, 113
van Stratum, D. G. C. & Vos, H. J. (1965) Fd Cosmet. Toxicol., 3,
475
Tye, R., Engel, J. D. & Rapion, I. (1965) Fd Cosmet. Toxicol., 3,
475
 Molecular weight 220.36
Definition Butylated hydroxytoluene contains not
less than 99 per cent. of C15H24O.
Description Butylated hydroxytoluene is a white,
crystalline or flaked solid, having a
characteristic faint aromatic odour.
Use As an antioxidant.
Biological Data
The document entitled "Specifications for Identity and Purity and
Toxicological Evaluation of some Antimicrobials and Antioxidants"
(FAO/WHO, 1965) contains a monograph on BHT summarizing the data
available at the end of 1964. Since then a substantial amount of new
information has become available for consideration. This and other
data are summarized in the present monograph.
Biochemical aspects
Excretion and tissue distribution
The metabolism of butylated hydroxytoluene (BHT) administered to
rabbits orally in single doses of 500 mg/kg body-weight was studied.
The metabolites 2,6-di-tertbutyle-4-hydroxymethylphenol (BHT-alc),
3,5-di-tert-butyl-4-hydroxybenzoic acid (BHT-acid) and
4,4'-ethylene-bis-(2,6-di-tert-butylphenol) were identified. The
urinary metabolites of BHT comprised 37.5 per cent. as glucoronides,
16.7 per cent. as ethereal sulfates and 6.8 per cent. as free phenols;
unchanged BHT was present only in the faeces (Akagi & Aoki, 1962a);
3,5-di-tert-butyl-4-hydroxybenzaldehyde (BHT-ald) was also isolated
from rabbit urine (Aoki, 1962). The main metabolic pathway was
confirmed by administering BHT-alc to rabbits and isolating BHT-ald,
BHT-acid, the ethylene-bis derative and unchanged BHT-alc in the
urine (Akagi & Aoki, 1962b).
The fate in the body of 14C-labelled BHT has been elucidated.
The relatively slow excretion of BHT Is probably attributable to
enterohepatic circulation rather than to tissue retention. Rats were
given single oral doses (1-100 mg/rat) of BHT-14C and approximately
80-90 per cent. of the dose was recovered in 4 days in the urine and
faeces. Of the total radioactivity, 40 per cent. appeared in the urine
of females and 25 per cent. in males. After 4 days approximately 3.8
per cent. of the dose was retained mainly in the alimentary tract. A
substantial portion of the radioactivity was found in the bile
collected from two rats (1 male, 1 female) over a period of 40 hours
(Daniel & Gage, 1965).
Groups of two rats (1 male, 1 female) were given 1-5 oral doses
of 44 mg/kg body-weight BHT on alternate days and each group killed 24
hours after the final dose. The range of the total dose accounted for
was 92-103.5 per cent. in males and 92.6-98.6 per cent. in females.
There was an indication of sex difference in the route of excretion,
females excreting 19-43 per cent. of the radioactivity in urine and
males only 3-15 per cent. Eight days after administration of 5 doses
92 per cent. of the radioactivity had been excreted by males and 97
per cent. by females. Subcutaneous administration of graded doses of
BHT to female rats revealed substantial faecal excretion but the rate
of excretion decreased with increasing dose. There was no evidence of
accumulation of BHT-14C in the body under the conditions of repeated
oral dosage (Tye at al., 1965).
The BHT content of fat and liver of rats given diets containing
0.5 and 1.0 per cent. BHT for periods up to 35 and 50 days
respectively: with 0.5 per cent. BHT in the diet, a level of
approximately 30 ppm in the fat was reached in males and 45 ppm in
females, with approximately 1-3 ppm in the liver, while with 1.0 per
cent. BHT the level in the fat was 50 ppm in males and 30 ppm in
females. On cessation of treatment, the level of BHT in fat fell with
a half-life of 7-10 days (Daniel & Gage, 1965). The level of BHT in
the fat reached a plateau at approximately 100 ppm after 3-4 days when
daily doses of 500 mg/kg body-weight were given by intubation; 200
mg/kg body-weight/day for 1 week produced a level of about 50 ppm
(Gilbert & Golberg, 1965).
When feed containing 500 ppm BHT was given to laying hens, 20 ppm
was found in the fat fraction of eggs; 100 ppm in the feed resulted in
residues of less than 5 ppm. In the broiler chicken, over a period of
21 weeks, the residues in body fat were 55 ppm on the 500 ppm diet and
less than 5 ppm on the 100 ppm diet (Van Stratum & Vos, 1965).
One-day-old chicks were given 14C-BHT at a level of 200 ppm in
the feed for 10 weeks. At broiler age, edible portions had residues
amounting to 1-3 ppm of BHT and metabolites. Similar diets given to
laying hens produced residues in eggs of 2 ppm after 7 days, the level
thereafter remaining constant (Frawley et al., 1965a).
Stimulation of liver microsomal processing enzymes
Rats given BHT by daily intubation showed increased activity of
some liver microsomal enzymes. Stimulation of enzyme activity
correlated with an increase in relative liver weight, the threshold
dose for these changes in enzyme activity in female rats being below
25-75 mg BHT/kg body-weight/day. The storage of BHT in fat appeared to
be influenced by the activity of the processing enzymes. In rats given
500 mg/kg body-weight daily the level of BHT in fat attained values of
230 ppm in females and 162 ppm in males by the second day, by which
time the relative liver weight and processing enzyme activities had
become elevated. Thereafter, liver weight and enzyme activities
continued to rise but the BHT content of fat fell to a plateau of
about 100 ppm in both sexes (Gilbert & Golberg, 1965).
In further work with rats it was found that increased output of
urinary ascorbic acid paralleled liver enlargement induced by BHA or
BHT in onset, degree and duration, being rapid but transient with BHA
and slower in onset but more prolonged with BHT (Gaunt et al., 1965a).
The parallelism between stimulation of processing enzyme activity,
increase in urinary ascorbic acid output, and increase in relative
liver weight brought about by BHT was unaffected by 14 days of dietary
restriction, and all these changes except liver weight were reversible
during 14 days' recovery on normal diet (Gaunt et al., 1965b).
Special studies
Diets containing 0.1 or 0.5 per cent. BHT together with two
dietary levels of lard (10 and 20 per cent.) were given to mice. The
0.5 per cent. level of BHT produced slight but significant reduction
in mean pup weight and total litter weight at 22 days of age. The 0.1
per cent. level of BHT had no such effect. Out. of 7754 mice born,
none showed anophthalmia, although 12 out of the 144 mothers were
selected from an established anophthalmic strain (Johnson, 1965).
In a study on the embryotoxicity of BHT three dosing schedules
were employed: single doses (1000 mg/kg body-weight) on a specific day
of gestation, repeated daily doses (250-500 mg/kg body-weight) from
the time of mating throughout pregnancy and daily doses (250-500 mg/kg
body-weight for mice and 500 and 700 mg/kg body-weight for rats)
during a 7-10 week period before mating, continuing throughout mating
and gestation up to the time the animals were killed. No significant
embryotoxic effects were observed on examination of the skeletal and
soft tissue of the fully developed foetuses as well as by other
criteria. Reproduction and post-natal development were also
unaffected (Clegg, 1965)
Acute toxicity
Animal Route LD50 Reference
(mg/kg
body-weight)
Rat oral 2 450 Karplyuk, 1960
Mouse oral 2 000 Karplyuk, 1960
Short-term studies
Rat. BHT (2000 ppm) incorporated in a diet containing 19.9 per
cent. casein was administered to a group of 8 weeks; a further group
of 8 rats served as controls. The experiment was repeated with 16.6
per cent. casein in the diet of further groups for 4 weeks and again
with 9.6 per cent. casein (and no added choline) for 7 weeks. In all 3
instances BHT caused stimulation of growth and improved protein
efficiency. The N content of the liver was, however, greatly reduced
in BHT-treated animals, except when the level of BHT was reduced to
200 ppm. Recovery of hepatic protein after fasting (details not given)
was also impared in rats on 2000 ppm BHT. Liver lipid content was
increased with 2000 ppm but not with 200 ppm BHT. A dietary level of
2000 ppm BHT also increased the adrenal weight and ascorbic acid
content, although if recalculated on the basis of weight of gland,
there was no significant difference. The increase in adrenal ascorbic
acid is interpreted as indicating a stress imposed on the organism by
BHT (Sporn & Schöbesch, 1961).
Groups of 48 weanling rats (24 of each sex) were given diets
containing 1000 ppm BHT for periods of up to 16 weeks. A group of 48
rats served as controls. Measurements of growth rate, food
consumption, weight and micropathological examination of organs at
autopsy revealed no difference from untreated rats. However, increase
in relative liver weight and in the weight of the adrenals was
produced without histopathological evidence of damage. Biochemical
measurement and histochemical assessments of liver glucose
-phosphatase and glucose 6-phosphate dehydrogenase activities revealed
no difference from the control group (Gaunt at al., 1965a).
Multigeneration study
Weanling rats (16 of each sex) were fed a diet containing 20 per
cent. lard and 0, 300, 1000 or 3000 ppm BHT and mated at 100 days of
age (79 days on test). Ten days after weaning of the first litter the
animals were again mated to produce a second litter. The offspring (16
females and 8 males) ware mated at 100 days of age. Numerous function
and clinical tests including serum cholesterols and lipids were
performed on the parents and the first filial generation up to 28
weeks and gross and microscopical examination at 42 weeks. At the 3000
ppm dietary level a 10-20 per cent. reduction in growth rate of
parents and offspring was observed. A 20 per cent. elevation of serum
cholesterol levels was observed after 28 weeks, but no cholesterol
elevation after 10 weeks. A 10-20 per cent. increase in relative liver
weight was also observed upon killing after 42 weeks on diet. All
other observations at 3000 ppm and all observations at 1000 ppm and
300 ppm were comparable with control. All criteria of reproduction
were normal. No teratogenic effects ware detected (Frawley et al.,
1965b). Similar results to those obtained with the parental and first
filial generations were also obtained with the second filial
generation. Examination of two litters obtained from the latter at 100
days of age revealed no effects except a reduction of mean body-weight
at the 3000 ppm level. The offspring were examined for: litter size,
mean body-weight, occurrence of still-birth, survival rate and gross
and microscopic pathology (Frawley, 1967).
Comments
After single and repeated oral doses BHT is eliminated almost
completely in four days in the urine and faeces. The relatively slower
rate of excretion than that shown by BHA could be due to enterohepatic
circulation. Results of the analysis of tissues of rats suggest that
unchanged BHT does not tend to accumulate in the body.
The browning, thinning and loss of hair reported in one strain of
rat has not been reproduced in other strains. Reproduction studies
carried out on several strains of rats and mice fail to reveal
anophthalmia in the offspring reported earlier in a different strain
of rats.
Diet containing 20 per cent. lard and 1000 ppm BHT has no effect
on serum cholesterol and lipid levels, relative liver and other organ
weights, liver lipids and growth rate.
Metabolic studies in man are desirable.
Evaluation
Level causing no toxicological effect
Rat. 1000 ppm in the diet, equivalent to 50 mg/kg
body-weight/day.
Estimate of acceptable daily intake for man
mg/kg body-weight1
Unconditional acceptance 0-0.5
Conditional acceptance 0.5-2
REFERENCES
Akagi, M. & Aoki, I. (1962a) Chem, Pharm. Bull. (Tokyo), 10, 101
Akagi, M. & Aoki, I. (1962b) Chem. Pharm, Bull. (Tokyo), 10, 200
Aoki, I. (1962) Chem. Pharm. Bull. (Tokyo), 10, 105
Clegg, D. J. (1965)Fd Cosmet Toxicol., 3, 387
Daniel, J. W. & Gage, J. C. (1965) Fd Cosmet. Toxicol., 3, 405
FAO/WHO (1965) FAO Nutrition Meetings Report Series, No. 38;
WHO/Food Add./24.65
Frawley, J. P. (1967) Unpublished report submitted to WHO
Frawley, J. P., Kay, J. M. & Calandra, J. C. (1965a) Fd Cosmet.
Toxicol., 3, 471
Frawley, J. P., Kohn, F.E. Kay, J. H. & Calandra, J. C. (1965b)
Fd Cosmet. Toxicol., 3, 377
Gaunt. I. F., Feuer, G., Fairweather, F. A. & Gilbert, D. (1965a)
Fd Cosmet.Toxicol., 3, 433
Gaunt, I. F., Gilbert, D. & Martin, D. (1965b) Fd Cosmet. Toxicol.,
3, 445
Gilbert, D. & Golberg, L. (1965) Fd Cosmet. Toxicol., 3, 417
1 As sum of BHA and BHT.
Johnson, A. R. (1965) Fd Cosmet. Toxicol., 3, 417
Sporn, A. & Schöbesch, O. (1961) Igiena (Bucharest), 9, 113
van Stratum, D. G. C. & Vos, H. J. (1965) Fd Cosmet. Toxicol., 3,
475
Tye, R., Engel, J. D. & Rapion, I. (1965) Fd Cosmet. Toxicol., 3,
475
Molecular weight 220.36
Definition Butylated hydroxytoluene contains not
less than 99 per cent. of C15H24O.
Description Butylated hydroxytoluene is a white,
crystalline or flaked solid, having a
characteristic faint aromatic odour.
Use As an antioxidant.
Biological Data
The document entitled "Specifications for Identity and Purity and
Toxicological Evaluation of some Antimicrobials and Antioxidants"
(FAO/WHO, 1965) contains a monograph on BHT summarizing the data
available at the end of 1964. Since then a substantial amount of new
information has become available for consideration. This and other
data are summarized in the present monograph.
Biochemical aspects
Excretion and tissue distribution
The metabolism of butylated hydroxytoluene (BHT) administered to
rabbits orally in single doses of 500 mg/kg body-weight was studied.
The metabolites 2,6-di-tertbutyle-4-hydroxymethylphenol (BHT-alc),
3,5-di-tert-butyl-4-hydroxybenzoic acid (BHT-acid) and
4,4'-ethylene-bis-(2,6-di-tert-butylphenol) were identified. The
urinary metabolites of BHT comprised 37.5 per cent. as glucoronides,
16.7 per cent. as ethereal sulfates and 6.8 per cent. as free phenols;
unchanged BHT was present only in the faeces (Akagi & Aoki, 1962a);
3,5-di-tert-butyl-4-hydroxybenzaldehyde (BHT-ald) was also isolated
from rabbit urine (Aoki, 1962). The main metabolic pathway was
confirmed by administering BHT-alc to rabbits and isolating BHT-ald,
BHT-acid, the ethylene-bis derative and unchanged BHT-alc in the
urine (Akagi & Aoki, 1962b).
The fate in the body of 14C-labelled BHT has been elucidated.
The relatively slow excretion of BHT Is probably attributable to
enterohepatic circulation rather than to tissue retention. Rats were
given single oral doses (1-100 mg/rat) of BHT-14C and approximately
80-90 per cent. of the dose was recovered in 4 days in the urine and
faeces. Of the total radioactivity, 40 per cent. appeared in the urine
of females and 25 per cent. in males. After 4 days approximately 3.8
per cent. of the dose was retained mainly in the alimentary tract. A
substantial portion of the radioactivity was found in the bile
collected from two rats (1 male, 1 female) over a period of 40 hours
(Daniel & Gage, 1965).
Groups of two rats (1 male, 1 female) were given 1-5 oral doses
of 44 mg/kg body-weight BHT on alternate days and each group killed 24
hours after the final dose. The range of the total dose accounted for
was 92-103.5 per cent. in males and 92.6-98.6 per cent. in females.
There was an indication of sex difference in the route of excretion,
females excreting 19-43 per cent. of the radioactivity in urine and
males only 3-15 per cent. Eight days after administration of 5 doses
92 per cent. of the radioactivity had been excreted by males and 97
per cent. by females. Subcutaneous administration of graded doses of
BHT to female rats revealed substantial faecal excretion but the rate
of excretion decreased with increasing dose. There was no evidence of
accumulation of BHT-14C in the body under the conditions of repeated
oral dosage (Tye at al., 1965).
The BHT content of fat and liver of rats given diets containing
0.5 and 1.0 per cent. BHT for periods up to 35 and 50 days
respectively: with 0.5 per cent. BHT in the diet, a level of
approximately 30 ppm in the fat was reached in males and 45 ppm in
females, with approximately 1-3 ppm in the liver, while with 1.0 per
cent. BHT the level in the fat was 50 ppm in males and 30 ppm in
females. On cessation of treatment, the level of BHT in fat fell with
a half-life of 7-10 days (Daniel & Gage, 1965). The level of BHT in
the fat reached a plateau at approximately 100 ppm after 3-4 days when
daily doses of 500 mg/kg body-weight were given by intubation; 200
mg/kg body-weight/day for 1 week produced a level of about 50 ppm
(Gilbert & Golberg, 1965).
When feed containing 500 ppm BHT was given to laying hens, 20 ppm
was found in the fat fraction of eggs; 100 ppm in the feed resulted in
residues of less than 5 ppm. In the broiler chicken, over a period of
21 weeks, the residues in body fat were 55 ppm on the 500 ppm diet and
less than 5 ppm on the 100 ppm diet (Van Stratum & Vos, 1965).
One-day-old chicks were given 14C-BHT at a level of 200 ppm in
the feed for 10 weeks. At broiler age, edible portions had residues
amounting to 1-3 ppm of BHT and metabolites. Similar diets given to
laying hens produced residues in eggs of 2 ppm after 7 days, the level
thereafter remaining constant (Frawley et al., 1965a).
Stimulation of liver microsomal processing enzymes
Rats given BHT by daily intubation showed increased activity of
some liver microsomal enzymes. Stimulation of enzyme activity
correlated with an increase in relative liver weight, the threshold
dose for these changes in enzyme activity in female rats being below
25-75 mg BHT/kg body-weight/day. The storage of BHT in fat appeared to
be influenced by the activity of the processing enzymes. In rats given
500 mg/kg body-weight daily the level of BHT in fat attained values of
230 ppm in females and 162 ppm in males by the second day, by which
time the relative liver weight and processing enzyme activities had
become elevated. Thereafter, liver weight and enzyme activities
continued to rise but the BHT content of fat fell to a plateau of
about 100 ppm in both sexes (Gilbert & Golberg, 1965).
In further work with rats it was found that increased output of
urinary ascorbic acid paralleled liver enlargement induced by BHA or
BHT in onset, degree and duration, being rapid but transient with BHA
and slower in onset but more prolonged with BHT (Gaunt et al., 1965a).
The parallelism between stimulation of processing enzyme activity,
increase in urinary ascorbic acid output, and increase in relative
liver weight brought about by BHT was unaffected by 14 days of dietary
restriction, and all these changes except liver weight were reversible
during 14 days' recovery on normal diet (Gaunt et al., 1965b).
Special studies
Diets containing 0.1 or 0.5 per cent. BHT together with two
dietary levels of lard (10 and 20 per cent.) were given to mice. The
0.5 per cent. level of BHT produced slight but significant reduction
in mean pup weight and total litter weight at 22 days of age. The 0.1
per cent. level of BHT had no such effect. Out. of 7754 mice born,
none showed anophthalmia, although 12 out of the 144 mothers were
selected from an established anophthalmic strain (Johnson, 1965).
In a study on the embryotoxicity of BHT three dosing schedules
were employed: single doses (1000 mg/kg body-weight) on a specific day
of gestation, repeated daily doses (250-500 mg/kg body-weight) from
the time of mating throughout pregnancy and daily doses (250-500 mg/kg
body-weight for mice and 500 and 700 mg/kg body-weight for rats)
during a 7-10 week period before mating, continuing throughout mating
and gestation up to the time the animals were killed. No significant
embryotoxic effects were observed on examination of the skeletal and
soft tissue of the fully developed foetuses as well as by other
criteria. Reproduction and post-natal development were also
unaffected (Clegg, 1965)
Acute toxicity
Animal Route LD50 Reference
(mg/kg
body-weight)
Rat oral 2 450 Karplyuk, 1960
Mouse oral 2 000 Karplyuk, 1960
Short-term studies
Rat. BHT (2000 ppm) incorporated in a diet containing 19.9 per
cent. casein was administered to a group of 8 weeks; a further group
of 8 rats served as controls. The experiment was repeated with 16.6
per cent. casein in the diet of further groups for 4 weeks and again
with 9.6 per cent. casein (and no added choline) for 7 weeks. In all 3
instances BHT caused stimulation of growth and improved protein
efficiency. The N content of the liver was, however, greatly reduced
in BHT-treated animals, except when the level of BHT was reduced to
200 ppm. Recovery of hepatic protein after fasting (details not given)
was also impared in rats on 2000 ppm BHT. Liver lipid content was
increased with 2000 ppm but not with 200 ppm BHT. A dietary level of
2000 ppm BHT also increased the adrenal weight and ascorbic acid
content, although if recalculated on the basis of weight of gland,
there was no significant difference. The increase in adrenal ascorbic
acid is interpreted as indicating a stress imposed on the organism by
BHT (Sporn & Schöbesch, 1961).
Groups of 48 weanling rats (24 of each sex) were given diets
containing 1000 ppm BHT for periods of up to 16 weeks. A group of 48
rats served as controls. Measurements of growth rate, food
consumption, weight and micropathological examination of organs at
autopsy revealed no difference from untreated rats. However, increase
in relative liver weight and in the weight of the adrenals was
produced without histopathological evidence of damage. Biochemical
measurement and histochemical assessments of liver glucose
-phosphatase and glucose 6-phosphate dehydrogenase activities revealed
no difference from the control group (Gaunt at al., 1965a).
Multigeneration study
Weanling rats (16 of each sex) were fed a diet containing 20 per
cent. lard and 0, 300, 1000 or 3000 ppm BHT and mated at 100 days of
age (79 days on test). Ten days after weaning of the first litter the
animals were again mated to produce a second litter. The offspring (16
females and 8 males) ware mated at 100 days of age. Numerous function
and clinical tests including serum cholesterols and lipids were
performed on the parents and the first filial generation up to 28
weeks and gross and microscopical examination at 42 weeks. At the 3000
ppm dietary level a 10-20 per cent. reduction in growth rate of
parents and offspring was observed. A 20 per cent. elevation of serum
cholesterol levels was observed after 28 weeks, but no cholesterol
elevation after 10 weeks. A 10-20 per cent. increase in relative liver
weight was also observed upon killing after 42 weeks on diet. All
other observations at 3000 ppm and all observations at 1000 ppm and
300 ppm were comparable with control. All criteria of reproduction
were normal. No teratogenic effects ware detected (Frawley et al.,
1965b). Similar results to those obtained with the parental and first
filial generations were also obtained with the second filial
generation. Examination of two litters obtained from the latter at 100
days of age revealed no effects except a reduction of mean body-weight
at the 3000 ppm level. The offspring were examined for: litter size,
mean body-weight, occurrence of still-birth, survival rate and gross
and microscopic pathology (Frawley, 1967).
Comments
After single and repeated oral doses BHT is eliminated almost
completely in four days in the urine and faeces. The relatively slower
rate of excretion than that shown by BHA could be due to enterohepatic
circulation. Results of the analysis of tissues of rats suggest that
unchanged BHT does not tend to accumulate in the body.
The browning, thinning and loss of hair reported in one strain of
rat has not been reproduced in other strains. Reproduction studies
carried out on several strains of rats and mice fail to reveal
anophthalmia in the offspring reported earlier in a different strain
of rats.
Diet containing 20 per cent. lard and 1000 ppm BHT has no effect
on serum cholesterol and lipid levels, relative liver and other organ
weights, liver lipids and growth rate.
Metabolic studies in man are desirable.
Evaluation
Level causing no toxicological effect
Rat. 1000 ppm in the diet, equivalent to 50 mg/kg
body-weight/day.
Estimate of acceptable daily intake for man
mg/kg body-weight1
Unconditional acceptance 0-0.5
Conditional acceptance 0.5-2
REFERENCES
Akagi, M. & Aoki, I. (1962a) Chem, Pharm. Bull. (Tokyo), 10, 101
Akagi, M. & Aoki, I. (1962b) Chem. Pharm, Bull. (Tokyo), 10, 200
Aoki, I. (1962) Chem. Pharm. Bull. (Tokyo), 10, 105
Clegg, D. J. (1965)Fd Cosmet Toxicol., 3, 387
Daniel, J. W. & Gage, J. C. (1965) Fd Cosmet. Toxicol., 3, 405
FAO/WHO (1965) FAO Nutrition Meetings Report Series, No. 38;
WHO/Food Add./24.65
Frawley, J. P. (1967) Unpublished report submitted to WHO
Frawley, J. P., Kay, J. M. & Calandra, J. C. (1965a) Fd Cosmet.
Toxicol., 3, 471
Frawley, J. P., Kohn, F.E. Kay, J. H. & Calandra, J. C. (1965b)
Fd Cosmet. Toxicol., 3, 377
Gaunt. I. F., Feuer, G., Fairweather, F. A. & Gilbert, D. (1965a)
Fd Cosmet.Toxicol., 3, 433
Gaunt, I. F., Gilbert, D. & Martin, D. (1965b) Fd Cosmet. Toxicol.,
3, 445
Gilbert, D. & Golberg, L. (1965) Fd Cosmet. Toxicol., 3, 417
1 As sum of BHA and BHT.
Johnson, A. R. (1965) Fd Cosmet. Toxicol., 3, 417
Sporn, A. & Schöbesch, O. (1961) Igiena (Bucharest), 9, 113
van Stratum, D. G. C. & Vos, H. J. (1965) Fd Cosmet. Toxicol., 3,
475
Tye, R., Engel, J. D. & Rapion, I. (1965) Fd Cosmet. Toxicol., 3,
475
