
INTERNATIONAL PROGRAMME ON CHEMICAL SAFETY
WORLD HEALTH ORGANIZATION
SAFETY EVALUATION OF CERTAIN
FOOD ADDITIVES
WHO FOOD ADDITIVES SERIES: 42
Prepared by the Fifty-first meeting of the Joint FAO/WHO
Expert Committee on Food Additives (JECFA)
World Health Organization, Geneva, 1999
IPCS - International Programme on Chemical Safety
gamma-CYCLODEXTRIN
First draft prepared by
Dr P.J. Abbott
Australia New Zealand Food Authority, Canberra, Australia
Explanation
Biological data
Biochemical aspects
Absorption, distribution, biotransformation,
and excretion
Toxicological studies
Acute toxicity
Short-term studies of toxicity
Genotoxicity
Developmental toxicity
Special studies
Ocular irritation and dermal sensitization
Cell membrane effects
Impurities
Estimate of dietary intake
Comments
Evaluation
References
1. EXPLANATION
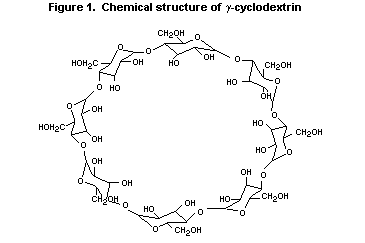
gamma-Cyclodextrin is a ring-shaped molecule made up of eight
glucose units linked by alpha-1,4-bonds. It is produced by the action
of the enzyme, cyclodextrin-glycosyl transferase (EC 2.4.1.19) on
hydrolysed starch syrups. Cyclodextrin-glycosyl transferases are
amylolytic enzymes that are produced naturally by various strains of
Bacillus and other organisms. The gene coding for the cyclodextrin-
glycosyl transferase used for the manufacture of gamma-cyclo-dextrin
was isolated from Bacillus firmus and Bacillus lentus (both of
which are considered non-pathogenic) and cloned into a strain of
Escherichia coli K12. Mixtures of alpha-, ß-, and gamma-cyclodextrin
are formed during the reaction, and the formation of
gamma-cyclodextrin is optimized by the addition of the complexant,
8-cyclohexadecen-1-one, which causes precipitation of the
gamma-cyclodextrin-8-cyclohexadecen-1-one complex, which can then be
further purified. The final product contains > 98%
gamma-cyclodextrin. Cyclodextrin-glycosyl transferase is inactivated
by heat and removed from the final gamma-cyclodextrin product. The
complexant, 8-cyclohexadecen-1-one, is extracted from the
gamma-cyclodextrin with n-decane which, in turn, is separated from
the final dry gamma-cyclodextrin product.
The circular structure of gamma-cyclodextrin (Figure 1) provides
a hydrophobic cavity which enables complexes to be formed with a
variety of organic molecules, while the hydrophilic outer surface
makes gamma-cyclodexrin water soluble. Because of these properties,
gamma-cyclodextrin can be used in a variety of ways in food, with a
potential intake in the order of grams per person per day.
 gamma-Cyclodextrin has not been reviewed previously by the
Committee; however, the structurally related ß-cyclodextrin (seven
glucose units) was evaluated at the forty-first and forty-fourth
meetings of the Committee (Annex 1, references 107 and 116).
2. BIOLOGICAL DATA
2.1 Biochemical aspects
2.1.1 Absorption, distribution, biotransformation, and excretion
Early experiments provided some evidence that gamma-cyclodextrin
(unlike alpha- or ß-cyclodextrin) is hydrolysed by salivary amylase
(French, 1957). More recent studies have confirmed that both human
salivary amylase and human or porcine pancreatic amylase can readily
hydrolyse gamma-cyclodextrin, yielding mainly maltose, some
maltotriose, and smaller amounts of glucose (Abdullah et al., 1966;
Marshall & Miwa, 1981; Kondo et al., 1990).
The fate of intravenously administered gamma-cyclodextrin was
examined in three male and two female rabbits and one male dog. The
rabbits were each given a dose of 7.5 g in saline (30 ml of a 25% w/v
solution), while the dog was given a dose of 25 g in saline (500 ml of
a 5% w/v solution at 8.3 ml/min). Blood and urine samples were
collected and examined by high-performance liquid chromatography
(HPLC) for gamma-cyclodextrin over 4 h. The half-life of
gamma-cyclo-dextrin in blood was about 50 min in rabbits and 30 min in
the dog. In rabbits, about 77% of the injected dose was recovered as
unchanged gamma-cyclodextrin in the urine within 24 h, while in the
dog, nearly 80% of the injected dose was recovered as unchanged
gamma-cyclodextrin in the urine within the first 4 h (Matsuda et al.,
1985).
Two male Wistar rats were given 14C-gamma-cyclodextrin (94%
radiochemical purity) as single oral or intravenous doses. After an
oral dose of 14C-gamma-cyclo-dextrin (25 µCi/kg bw diluted to 200
mg/kg bw), 50% of the radiolabel was excreted in the exhaled air
within 24 h, while 2% was excreted in the urine and 5% in the faeces;
35% of the radiolabel was retained in the body after 24 h. No
detectable gamma-cyclodextrin was found in blood during 8 h after
administration, suggesting metabolism in the gastrointestinal tract.
After the intravenous dose of 14C-gamma-cyclodextrin (25 µCi/kg
bw diluted to 100 mg/kg bw), 90% of the radiolabel was excreted in the
urine within 8 h, mainly as unchanged gamma-cyclodextrin. After 8 h,
small amounts of radiolabel were excreted in exhaled air (1.1%), while
0.5% was detected in the faeces; 6% was retained in the body after 24
h (de Bie & van Ommen, 1994).
Groups of four male Wistar rats were given single oral or
intravenous doses of 14C-gamma-cyclodextrin. The oral dose was 1000
mg/kg bw, and the concentration of radiolabel was either 25 or 100
µCi/kg bw. In order to examine the role of the gut microflora in the
metabolism of gamma-cyclodextrin, a separate group of germ-free male
rats was given 25 µCi/kg bw gamma-cyclodextrin diluted to 1000 mg/kg
bw. The intravenous dose was 600 mg/kg bw, and the concentration of
radiolabel was 25 µCi/kg bw.
In the first experiment, the time-course and routes of metabolism
were examined after oral administration. After administration of
1000 mg/kg bw (25 µCi/kg bw) 14C-gamma-cyclodextrin, 62% of the
radiolabel in males and 71% in females was excreted within 48 h. The
majority was excreted in expired air (57% in males and 65% in
females), with maximum excretion between 1 and 2 h after dosing.
Little was excreted in the urine (2.1% for males and 2.5% for females)
or faeces (2.9% for males and females) during the 48-h sampling
period. Retention of radiolabel after 48 h was 27% in males and 22% in
females, with the most significant retention in the liver (11% in
males, 5.2% in females). The amounts in all other organs were low:
intestine, 0.42% for males, 0.36% for females; kidney, 0.13% for
males.
In the second experiment, the excretion kinetics and time-course
in blood over 24 h were examined after a single oral dose of
1000 mg/kg bw (100 Ci/kg bw) 14C-gamma-cyclodextrin. The major route
of excretion was exhaled air (55% for males; 53% for females) during
the 24-h sampling period. The carcass retained 35% of the radioactive
dose in males and 25% in females at 24 h after treatment. The maximum
concentration in blood was reached 40 min after treatment, but this
concentration decreased by 50% after 0.5 h in males and after 6 h in
females. The urine of animals of each sex contained a peak that eluted
before glucose at all times and a small peak that co-eluted with
glucose (6% of total radiolabel). A small amount of unchanged
gamma-cyclo-dextrin was detected in urine after 4 h (< 0.02% of the
ingested dose). HPLC analysis of the stomach contents 24 h after
dosing revealed the presence of gamma-cyclodextrin and a minor peak
that co-eluted with glucose. HPLC analysis of the blood revealed the
presence of a radioactive peak that co-eluted with glucose.
In the third experiment, the excretion kinetics and time-course
in blood over 24 h were examined after a single intravenous dose of
600 mg/kg bw (25 µCi/kg bw) 14C-gamma-cyclodextrin. The major route
of excretion was the urine (87% for males, 91% for females), and the
faeces contained about 1% and the gastrointestinal tract about 0.5% of
the radiolabel; the exhaled air contained 7.8% of the dose for males
and 3.2% for females. The carcass contained 3.9% of the dose in males
and 2.2% in females, the liver having the most residue (0.41% in
males, 0.42% in females). Radiolabel was removed rapidly from the
blood, with calculated initial half-lives of 20 min in males and 15
min in females. HPLC analysis of blood revealed predominantly
gamma-cyclodextrin.
In the fourth experiment, excretion kinetics over 24 h was
examined in germ-free rats given a single oral dose of 1000 mg/kg bw
(25 µCi/kg bw) gamma-cyclodextrin. About 73% of the radiolabel was
excreted in expired air (68% in males, 65% in females). Peak
exhalation of 14C-carbon dioxide occurred at about 90 min. Low
concentrations of radiolabel were found in faeces and urine. The
gastrointestinal tract contained 9.2% in males and 11% in females,
with the majority in the caecum. The results indicate that
gastrointestinal microflora play little part in the metabolism of
gamma-cyclodextrin.
Overall, this study indicates that gamma-cyclodextrin is
metabolized to glucose, other low-molecular-mass sugar metabolites,
and carbon dioxide by luminal and/or epithelial enzymes of the
gastrointestinal tract (de Bie & van Ommen, 1994; de Bie et al.,
1998).
2.2 Toxicological studies
2.2.1 Acute toxicity
The acute toxicity of gamma-cyclodextrin was examined in mice and
rats; the results are summarized in Table 1. In each study, groups of
10 animals of each sex were tested at three doses or served as
controls. There were no deaths at the highest doses tested (Matsuda et
al., 1983).
Table 1. Acute toxicity of gamma-cyclodextrin
Species Sex Route LD50 Reference
(mg/kg bw)
Mouse Male and female Oral >16 000 Matsuda et al. (1983)
Mouse Male and female Subcutaneous > 4 000 Matsuda et al. (1983)
Mouse Male and female Intravenous > 4 000 Matsuda et al. (1983)
Mouse Male and female Intravenous 10 000 Riebeek (1990a)
Rat Male and female Oral > 8 000 Matsuda et al. (1983)
Rat Male and female Subcutaneous > 2 400 Matsuda et al. (1983)
Rat Male and female Intravenous > 3 750 Matsuda et al. (1983)
Rat Male and female Intravenous > 3 750 Riebeek (1990b)
Rat Male Intraperitoneal > 4 600 Riebeek (1990c)
In a test for micronucleus formation (see section 2.2.3), groups
of 15 Swiss mice of each sex were given a single dose of 15 g/kg bw
gamma-cyclodextrin by gavage and were killed in groups of five at 24,
48, and 72 h. All of the animals survived until termination (Immel,
1991).
The toxicity of intravenously administered gamma-cyclodextrin was
examined in groups of five male and five female Crl:CD mice and five
male and five female Crl:W1(WU)BR rats. The mice were given
gamma-cyclodextrin (purity, > 98%) at doses of 5000, 7500, or 10 000
mg/kg bw, while the rats were given doses of 2500, 3750, or 5000 mg/kg
bw as a solution in sterile saline. The animals were observed for
clinical signs over 14 days after treatment. They were examined for
gross pathological changes on day 14 after treatment.
Both mice and rats showed dose-related signs of toxicity, such as
piloerection and sluggishness, within 1 h to a few days after
treatment. Deaths occurred within a few days in both species, but the
surviving animals recovered and appeared to be healthy at the end of
the observation period. Macroscopic examination revealed no gross
treatment-related alterations (Riebeek, 1990a,b).
The toxicity of intraperitoneally administered gamma-cyclodextrin
was examined in groups of three male rats after single doses of 2000
or 4600 mg/kg bw as a solution in sterile saline. The animals were
observed for clinical signs during 14 days after treatment and were
examined grossly for pathological changes on the last day. There were
no signs of toxicity and no deaths. Macroscopic examination revealed
no gross treatment-related alterations (Riebeek, 1990c).
2.2.2 Short-term studies of toxicity
Rats
Groups of 15 Wistar rats of each sex were given
gamma-cyclodextrin intravenously as single daily doses of 0, 200, 630,
or 2000 mg/kg bw for 30 days. A group of five animals of each sex were
allowed to recover for 27 days after treatment. The animals were
examined throughout the study, and the body weights recorded weekly.
Clinical chemical parameters were examined at the end of treatment and
after the recovery period. Ten animals from each group were killed at
the end of treatment and the remainder at the end of the recovery
period.
There were no clinical signs of toxicity throughout the study.
The body-weight gain of males at 630 and 2000 mg/kg bw per day was
slightly decreased, but the difference was not significant. Food
consumption was decreased in a dose-related manner only during the
first few days of treatment. Significantly decreased mean values for
haemoglobin concentration and haemacrit were seen in males at doses
> 630 mg/kg bw per day, and significantly decreased erythrocyte
count in males at 2000 mg/kg bw per day. In females, the erythrocyte
count, haemoglobin concentration, and haematocrit were significantly
decreased only at 2000 mg/kg bw per day. The reticulocyte counts were
significantly increased in both males and females at 2000 mg/kg bw per
day. These changes were reversed after the recovery period. The only
significant clinical chemical change was an increase in creatinine and
urea in both males and females at 2000 mg/kg bw per day, which was
reversed at the end of the recovery period. Urinalysis revealed a
significantly increased incidence of haemoglobin in the urine of rats
at 2000 mg/kg bw per day, which was reversed at the end of the
recovery period.
Pathological examination at autopsy showed that the majority of
animals at 2000 mg/kg bw per day had light-coloured and, in some
cases, irregularly coloured kidneys. A dose-dependent increase in
spleen weight was found in all groups, which was significant in males
at doses > 630 mg/kg bw per day and in females at all doses. Liver
weights were significantly increased in females at > 630 mg/kg bw
per day, and kidney weights were significantly increased at 2000 mg/kg
bw per day. Lung and adrenal weights were significantly increased in
both males and females at 2000 mg/kg bw per day. These changes were
partially reversed after the recovery period, but a significant
increase in kidney and spleen weights was still present in females at
2000 mg/kg bw per day. Histopathological examination revealed marked
vacuolation of renal epithelial cells in the proximate convoluted
tubules, which may have been related to administration of a substance
that causes hyperosmolarity. There was also extensive pulmonary
histiocytosis (massive accumulation of alveolar macrophages) in all
animals at 2000 mg/kg bw per day. These changes were seen only in some
animals at 630 mg/kg bw per day and at lesser severity. Considerable
reversibility was observed after the recovery period. The NOEL was 200
mg/kg bw per day (Fuchs et al., 1992).
Groups of 15 Wistar rats of each sex were given
gamma-cyclodextrin intravenously as single daily doses of 0, 60, 120,
or 600 mg/kg bw for 90 days. Groups of five animals of each sex were
allowed to recover at the end of the treatment period. The animals
were examined throughout the study, and body weights were recorded
weekly. Haematological and urinary parameters were examined after one
month, at the end of the study, and on day 37 (males) or 36 (females)
of recovery. Clinical chemical parameters were examined at the end of
the treatment period and after recovery. Animals were killed after
90 days, their organs were examined, and tissues were taken for
histopathological examination.
There were no clinical signs of toxicity. Body-weight gain was
slightly decreased only in males at the high dose, and food
consumption was decreased in a dose-related manner only during the
first few days of treatment. Haematological examination revealed a
slight decrease in the mean erythrocyte count, haemaglobin
concentration, and haemacrit in females at one month, and these values
were significantly decreased at three months only in females at the
high dose. The reticulocyte counts were significantly increased in
females at the high dose at three months. At the end of the recovery
period, all of these values were normal. The only clinical chemical
changes were a slight decrease in bilirubin concentration in males and
females at the high dose and a slight increase in alkaline phosphatase
activity in males at the high dose. At the end of the recovery period,
all of the values were normal. The slight changes observed in urinary
parameters were considered not to be treatment-related.
Histological examination at autopsy revealed an increased
incidence of enlarged iliac lymph nodes in males and females at the
high dose, which was probably related to inflammation at the injection
site. Increases in the relative weights of lungs, liver, and spleen
were seen in males and in the relative weights of heart, lungs,
kidneys, spleen, and adrenal glands in females at the high dose. All
of these changes were reversed after the four-week recovery period.
Histopathological examination revealed hyperplasia of the mucosa of
the urinary bladder, which was reversed after four weeks, and
pulmonary histiocytosis (macrophage aggregates), which tended to
reverse after four weeks, in males and females at the high dose. The
reversibility of the urinary bladder changes indicates that the
hyperplasia was not preneoplastic. The NOEL was 120 mg/kg bw per day
(Ehling et al., 1992).
Groups of five male Wistar rats were fed diets containing
gamma-cyclo-dextrin at concentrations of 0, 5, 10, 15, or 20%,
equivalent to 0, 2500, 5000, 7500, or 10 000 mg/kg bw per day, for 14
days. A control group was fed 20% lactose. The animals were examined
for clinical signs of toxicity, and food and water consumption was
monitored throughout the study. On day 14, the animals were killed,
and blood samples were taken for examination of clinical chemical
parameters. The animals were also examined macroscopically for
pathological changes.
There were no deaths. The incidence of soft stools was slightly
higher in treated than control animals, and the mean body weights of
treated animals were slightly lower than those of controls, although
this difference was not statistically significant. Food intake was
slightly lower in controls than in treated groups. Plasma alkaline
phosphatase activity was increased in animals at 15 and 20%, while
gamma-glutamyl transferase activity was decreased in all treated
groups. The activity of aspartame and alanine aminotransferases were
unchanged, as were the concentrations of total protein, albumin, urea,
and bilirubin. Enlargement of the caecum was seen at all doses, the
most significant effect occurring in the group fed 20% lactose. There
were no macroscopic changes attributable to treatment (Lina & Jonker,
1989; Lina & Bär, 1998).
Groups of 20 Wistar rats of each sex were fed diets containing
gamma-cyclo-dextrin at concentrations of 0, 1.5, 5, or 20% (equivalent
to 0, 750, 2500, or 10 000 mg/kg bw per day) for 13 weeks. A control
group was fed 20% lactose. In order to examine the reversibility of
any effects seen, a group of 10 animals of each sex were fed either
20% gamma-cyclodextrin or 20% lactose for 13 weeks and then control
diet for one month. As a result of a feeding error, the groups
receiving 20% gamma-cyclodextrin were discarded, and two additional
groups were fed 0 or 20% gamma-cyclodextrin for 13 weeks and 10
animals receiving the 20% dose were fed control diet for an additional
month at the end of the 13-week period. Animals were examined for
clinical signs of toxicity, and food and water consumption was
monitored throughout the study. Ophthalmoscopic examinations were
conducted in animals at the high dose and those given lactose before
treatment and at the end of the study. The animals were killed at the
end of the study, and blood samples were taken for clinical chemistry.
The animals were also examined macroscopically for pathological
changes.
There were no treatment-related deaths during the study. Soft
stools were observed in some rats at 5 or 20% gamma-cyclodextrin and
in those given lactose early in the study, but this effect disappeared
subsequently. Ophthalmoscopic examination revealed no
treatment-related effects. The body weights of males at 20%
gamma-cyclodextrin were slightly but significantly decreased
throughout treatment; a more significant body-weight decrease was seen
in the males given 20% lactose. No difference was noted in these
groups during the recovery period. Food conversion efficiency was also
slightly decreased in males at 20% lactose. None of the changes in
haematological parameters was considered to be of toxicological
significance. Some changes in clinical chemistry were seen in females
at the high dose, namely increased activity of aspartate and alanine
aminotransferases, especially in one female. The sodium concentrations
were slightly higher but within the historical control range. Urinary
parameters were similar in control and treated groups, apart from a
significant increase in the urinary calcium concentration at the end
of treatment in males given 20% gamma-cyclodextrin and rats of each
sex given 20% lactose; the effect was more pronounced in the latter
group. The calcium:creatinine ratio was significantly increased in
rats receiving 20% lactose. The increased calcium appeared to be
associated with the increased load of osmotically active substances in
the large intestine; there was no increase in calcium at the end of
the recovery period.
Analysis of organ weights revealed significant increases in both
absolute and relative caecal weights in rats at 5 and 20%
gamma-cyclodextrin and 20% lactose in comparison with controls. At the
end of the recovery period, no difference from controls was seen for
rats given 20% gamma-cyclodextrin, but the increase was still evident
in those given 20% lactose. This effect is probably related to the
retention of an osmotically active substance in the large intestine.
Relative adrenal weights were increased in males given 20%
gamma-cyclodextrin and the lactose control group at the end of the
study. A very slight increase in relative liver weight was seen in
males at 20% gamma-cyclodextrin and females given lactose. No changes
in organ weight were seen at the end of the recovery period.
There were no gross pathological changes attributable to
treatment with gamma-cyclodextrin. Microscopic examination revealed
increased cortico-medullary mineralization in the kidneys of rats at
1.5 and 5% gamma-cyclodextrin but not at the high dose. This change is
considered to be relatively common in rats and not related to
treatment with gamma-cyclodextrin. There was no evidence of
hyperplasia of the mucosa of the urinary bladder or of pulmonary
histiocytosis, which were observed after intravenous administration.
The effects observed in rats after receiving gamma-cyclodextrin
in the diet at concentrations up to 20% appeared to be largely related
to the presence of high levels of an osmotically active substance in
the large intestine and not of toxicological significance. Similar,
and generally more pronounced, effects were observed when the diet
contained 20% lactose. On the basis of this study, dietary levels of
20% gamma-cyclodextrin (equivalent to10 000 mg/kg bw per day) were
considered to be tolerated without toxicological effects (Lina, 1992a;
Lina & Bär, 1998).
Dogs
Groups of four male and four female beagle dogs were fed diets
containing gamma-cyclodextrin at doses of 0, 5, 10, or 20% (equivalent
to 0, 1250, 2500, or 5000 mg/kg bw per day). Body weight and food
consumption were recorded weekly throughout the study. Ophthalmoscopic
examinations were made at the beginning and end of the study. Blood
was collected for clinical chemical and haematological examination
before the start of treatment and at weeks 6 and 13. Urinalyses were
performed at week 13. During week 14, all animals were killed, their
organs were examined, and tissues were prepared for histopathological
examination.
There were no deaths during the study and no clinical signs of
toxicity. Diarrhoea was noted in all groups, increasing in incidence
and severity with increasing doses of gamma-cyclodextrin, and was more
severe in females than in males. This effect was attributed to the
slow digestion and intestinal absorption of gamma-cyclodextrin.
Ophthalmoscopic examination revealed no differences between treated
and control animals. Weight gain was comparable in all groups, except
for a slight, non-significant decrease in males at 20% during the last
six weeks of the study. Treated dogs had slightly decreased food
intake during the first two weeks of the study, particularly at the
high dose. This effect persisted throughout the study only in dogs at
the high dose. No differences in haematological parameters were seen,
and clinical chemical parameters were similar in treated and control
groups. Urinalysis revealed a slight decrease in urinary pH in the
group receiving 20% gamma-cyclodextrin, which was considered to be
associated with the intake of poorly digestible carbohydrates and
increased fermentation in the large intestine.
Both the absolute and relative weights of the ovary were
increased in animals at 10 and 20% gamma-cyclodextrin, but no
histopathological changes were seen, and the weight of the ovaries in
the control group was lower than that of other dogs in the same
laboratory. The changes observed are considered unlikely to be
toxicologically significant. There was no change in absolute liver
weight, although the relative liver weight was increased in males at
the high dose as a result of the slight decrease in body weight at
this dose. This change is likely to be adaptive and was considered not
to be toxicologically significant. Caecal weights were increased in
the dogs at 10 and 20% gamma-cyclodextrin but statistically
significantly only in females. This change is considered to be related
to the presence of high levels of an osmotically active substance in
the large intestine. Pathological examination at the end of the study
revealed no treatment-related change. Similarly, the histopathological
findings were unremarkable. On the basis of this study, dietary levels
of 20% gamma-cyclodextrin (equivalent to 5000 mg/kg bw per day) were
considered to be tolerated without toxicological effects (Til & van
Nesselrooij, 1992; Til & Bär, 1998).
2.2.3 Genotoxicity
The results of assays for genotoxicity with gamma-cyclodextrin
are shown in Table 2.
2.2.4 Developmental toxicity
Rats
In a study of embryotoxicity and teratogenicity, groups of 25
presumed pregnant Wistar Crl:(WI)WU BR rats were fed diets containing
gamma-cyclodextrin (purity, >98%) at concentrations of 0. 1.5, 5, 10,
or 20% (equivalent to 0, 2500, 5000, or 10 000 mg/kg bw per day) on
days 0-21 of gestation. A separate group received a diet containing
20% lactose instead of pre-gelatinized potato starch. The animals were
examined throughout the study, and body weight and food consumption
were recorded. The rats were killed on day 21 and examined for
parameters of reproductive performance. Fetuses were examined for
signs of toxicity, external malformations, and soft-tissue defects and
were stained for detection of skeletal anomalies.
No deaths occurred during the study. Maternal body-weight gain
was similar in all groups except for a slight reduction at the 20%
dose on days 0-16 of gestation. Necropsy of the dams showed no adverse
effects that could be related to treatment. The number of viable
litters, the number of corpora lutea, and the mean number of
implantation sites were similar in all groups. Fetal length and body
weight were also similar in all groups. Examination of the fetuses
revealed no treatment-related increase in gross, skeletal, or visceral
abnormalities. Under the conditions of this assay, gamma-cyclodextrin
was not teratogenic (Verhagen & Waalkens-Berendsen, 1991).
Rabbits
In a study of embryotoxicity and teratogenicity, groups of 16
presumed pregnant New Zealand white rabbits were fed diets containing
gamma-cyclodextrin (purity, > 98%) at concentrations of 0, 5, 10, or
20% (equivalent to 0, 1500, 3000, or 6000 mg/kg bw per day) on days
0-29 of gestation. The animals were examined throughout the study, and
body weights and food consumption were recorded regularly. The animals
were killed on day 29 of pregnancy, and the fetuses were examined for
signs of toxicity, external malformations, and soft-tissue defects and
were stained for detection of skeletal anomalies.
Table 2. Results of assays for the genotoxicity of gamma-cyclodextrin
End-point Test object Concentration Result Reference
Bacterial gene S. typhimurium TA1535, 2-20000 Negativea Blijleven (1991)
mutation TA1537, TA 1538, µg/plate
TA98, TA100
Chromosomal Human lymphocytes 10-5000 Negativea de Vogel & van
aberrationb µg/ml Delft (1996)
Micronucleus CD-1 mouse bone 15 g/kg bw Negative Immel (1991)
formationb marrow (single dose)
a With and without exogenous metabolic activation
b Two assays conducted: treatment times, 6, 24, and 48 h; harvest times, 24 and 48 h
No deaths occurred during the study. Diarrhoea was seen in
several treated animals. Maternal body weight was significantly
decreased at the 20% dose during the first week of treatment but not
thereafter. Necropsy of the does showed no adverse effects that could
be related to treatment. The number of viable litters, the number of
corpora lutea, and the mean number of implantation sites were similar
in all groups. One abortion occurred in the control group and one in
rabbits at the 10% dose. Fetal length and body weight were similar in
all groups. Examination of the fetuses revealed no treatment-related
increase in gross, skeletal, or visceral abnormalities. An increase in
the incidence of haemorrhagic fluid in animals at the 5 and 20% doses
was considered unlikely to be treatment-related. Under the conditions
of this assay, gamma-cyclodextrin was not teratogenic
(Waalkens-Berendsen & Smits-van Prooije, 1992).
2.2.5 Special studies
2.2.5.1 Ocular irritation and dermal sensitization
gamma-Cyclodextrin was not irritating or corrosive to the eyes of
albino rabbits (Prinsen, 1990). In a skin sensitization assay in
guinea-pigs, a 30% solution of gamma-cyclodextrin induced no signs of
sensitization (Prinsen, 1992).
2.2.5.2 Cell membrane effects
The interactions between alpha-, beta-, and gamma-cyclodextrin
and membrane phospholipids, liposomes, and human erythrocytes were
studied in vitro. gamma-Cyclodextrin and the other cyclodextrins did
not increase the permeability of dipalmityol-phosphatidylcholine
liposomes, nor did they affect the active transport of 42K+ into
erythrocytes at a concentration of 10-2 mol/L. ß-Cyclo-dextrin but
not gamma-cyclodextrin or alpha-cyclodextrin increased the release of
42K+, 86Rb+, and 137Cs+ from erythocytes by passive transport at
a concentration of 1.7 × 10-2 mol/L (Szejtli et al., 1986).
gamma-Cyclodextrin induced haemolysis of human erythrocytes
in vitro in isotonic saline after 30 min, with a threshold
concentration of 16 mmol/L (approximately 20 mg/L). A similar effect
was seen with alpha-cyclodextrin at 6 mmol/L and with ß-cyclodextrin
at 3.5 mmol/L. At 15 mmol/L, gamma-cylodextrin caused a 2% release of
cholesterol from erythrocytes, indicating that cyclodextrin-induced
haemolysis is associated with removal of membrane components from
erythrocytes (Irie et al., 1982). Other studies have confirmed that
gamma-cyclodextrin causes erythrocyte haemolysis at concentrations of
15-30 mmol/L (Okada et al., 1988; Yoshida et al., 1988; Leroy-Lechat
et al., 1994).
In a study on the differential effects of cyclodextrins on human
erythrocytes in vitro, alpha-, ß-, and gamma-cyclodextrin were
incubated at increasing concentrations with erythrocytes for 30 min.
The cyclodextrins induced a change in shape, from discocyte to
spherocyte, but with ß-cyclodextrin haemolysis occurred before the
change was complete. gamma-Cyclodextrin was less potent than either
alpha- or ß-cyclodextrin in releasing potassium and haemoglobin from
erythrocytes and in solubilizing various components of erythrocytes,
including phospholipids, cholesterol, and proteins. A concentration of
4 mmol/L gamma-cyclodextrin was required to induce release of
phospholipid, and > 20 mmol was required for release of cholesterol
or protein (Ohtani et al., 1989).
The potential cytotoxicity of the cyclodextrins to P388 murine
leukaemia cells was examined by exposing the cells to increasing
concentrations of cyclodextrins for 48 h in medium containing 10%
fetal calf serum. Initial cytotoxicity was elicited at 11 mmol/L
alpha-cyclodextrin, 2.5 mmol/L ß-cyclodextrin, and 55 mmol/L
gamma-cyclodextrin (Leroy-Lechat et al., 1994).
Potential reductions in the luminescence of a strain of E. coli
after incubation with various concentrations of cyclodextrins were
examined. Luminescence was reduced by 20 and 50% by ß-cyclodextrin at
concentrations of 0.03 and 0.5%, respectively. With
gamma-cyclodextrin, a concentration of 2% was required to reduce the
luminescence by 20%, and a 50% reduction could not be achieved even at
the highest concentration of 10% gamma-cyclodextrin (Bär & Ulitzur,
1994).
2.2.5.3 Impurities
Cyclodextrin-glycosyl transferase
The gene coding for cyclodextrin-glycosyl transferase is derived
from a strain of the Bacillus firmus-lintus complex. This is a
ubiquitous group of aerobic, gram-positive, alkalophilic
microorganisms which form sub-terminal endospores. Both B. firmus
and B. lintus are considered to be non-pathogenic. After isolation
from this strain, the gene was cloned into pUC18, isolated again and
inserted into an expression and secretion vector derived from pJF118EH
by introduction of a DNA fragment coding for an appropriate signal
peptide. The vector, pJF118EH, is derived from pBR322, a widely used
vector which is considered to be safe. All genetic modifications were
made by directed mutagenesis by standard techniques. The gene that has
been introduced for production of the signal peptide is cleaved off
during exportation of cyclodextrin-glycosyl transferase and degraged
with the E. coli cell.
A strain derived from E. coli K12 was used as the host for the
cyclodextrin-glycosyl transferase expression and secretion vector.
E. coli K12 is considered to be a non-pathogenic strain and is used
for production of enzymes for food use.
For the production of cyclodextrin-glycosyl transferase, the
recombinant E. coli strain was cultured in a standard medium. The pH
of the fermentation broth was adjusted by adding food-grade ammonia or
phosphoric acid. The E. coli was grown to a certain density, after
which enzyme production was initiated by addition of isopropyl
thiogalactoside. The bacteria were then removed by fermentation and
the supernatant filtered and concentrated to the crude
cyclodextrin-glycosyl transferase preparation.
Groups of 20 Wistar rats of each sex were given an aqueous
solution of a cyclodextrin-glycosyl transferase preparation at doses
of 0, 5, 10, or 20 ml/kg bw per day, equivalent to 0, 65, 130, or 260
mg/kg bw per day of total organic solids (TOS), by gavage for 13
weeks. The animals were examined for clinical signs of toxicity, and
food and water consumption was monitored throughout the study.
Ophthalmoscopic examinations were conducted in rats at the high dose
before treatment and at the end of the study. Haematological
parameters were examined before treatment, on days 30 and 60, and at
the end of the study. The animals were killed at the end of the study,
and blood samples were taken for examination of clinical chemical
parameters. The animals were also examined macroscopically for
pathological changes, and samples were taken for histopathological
examination.
Two animals (one control male and one female at the low dose)
died as a result of dosing accidents. There were no clinical signs of
toxicity in the treated animals which survived. Ophthalmoscopic
examination of animals at the high dose revealed no treatment-related
changes. There was no significant difference in body weight between
treated and control rats, and food consumption was unchanged in the
treated group. There were no treatment-related haematological,
clinical chemical, or urinary changes. The organ weights of control
and treated groups were similar, and there were no treatment-related
gross pathological changes. Histopathological examination revealed an
increased incidence of pulmonary changes in the treated animals which
were considered to be related to aspiration of test material. The NOEL
was 20 ml/kg bw per day, equivalent to 260 mg/kg bw per day of TOS,
the highest dose tested (Jonker, 1994).
The cyclodextrin-glycosyl transferase preparation was tested for
mutagenic activity in S. typhimurium strains TA1535, TA1537, TA98,
and TA100 with and without metabolic activation at concentrations of
19-1500 µg/plate or 62-500 µg/plate. The assay was slightly
complicated by the growth-stimulating effects of some of the
components of the crude preparation, but the overall result confirmed
that cyclodextrin-glycosyl transferase had no mutagenic activity in
these strains.
8-Cyclohexadecen-1-one
8-Cyclohexadecen-1-one is a colourless, waxy, solid product. The
commercial preparation has a purity of > 98%. The acute toxicity of
8-cyclo-hexadecen-1-one was examined in five male and five female mice
at a dose of 5 ml/kg bw (4.6 g/kg bw) and in five male and five female
rats at doses of 5 and 10 g/kg bw. The animals were observed for 14
days. Two of the five mice died during the study, but there were no
deaths among the rats. It was concluded that the LD50 was > 4.6 g/kg
bw in mice and > 10 g/kg bw in rats (Dickhaus & Heisler, 1985;
Spanjers, 1986).
Groups of five CD-1 mice of each sex were fed diets containing
8-cyclo-hexadecen-1-one at doses of 0, 500, 1500, or 7500 mg/kg diet
(equivalent to 0, 75, 230, or 1100 mg/kg bw per day) for four weeks.
The animals were examined for clinical signs of toxicity, and food and
water consumption were monitored throughout the study. Haematological
and clinical chemical parameters were examined before treatment and at
the end of the study. The animals were also examined macroscopically
for pathological changes, and samples were taken for histopathological
examination. There were no deaths and no clinical signs of toxicity.
The body-weight gain of mice at the high dose was decreased, but food
and water intake were similar in treated and control groups. There
were no treatment-related haematological changes. The only clinical
chemical change was a decrease in the albumin:globulin ratio in males
at the high dose. An increase in relative liver weight was seen in
males and females at the high dose. No treatment-related gross
abnormalities were seen, but microscopic examination revealed effects
in the cytoplasm of four males at the high dose. The NOEL was 1500
mg/kg diet, equivalent to 230 mg/kg bw per day (Lina, 1992b).
Groups of five male and five female Wistar rats were fed diets
containing 8 cyclohexadecen-1-one at doses of 0, 600, 3000, or 15 000
mg/kg diet (equivalent to 0, 30, 120, or 750 mg/kg bw per day) for
four weeks. The animals were examined for clinical signs of toxicity,
and food and water consumption was monitored throughout the study.
Haematological and clinical chemical parameters were examined before
treatment and at the end of the study; the animals were also examined
macroscopically for pathological changes, and samples were taken for
histopathological examination. There were no deaths and no clinical
signs of toxicity.
A number of effects were seen in animals at the high dose.
Body-weight gain and food intake were decreased, and the red blood
cell counts and packed cell volume were increased. Clinical chemistry
revealed increased plasma urea and creatinine concentrations in males
and decreased albumin in females, and urinalysis showed increased
urinary density in animals of each sex. The absolute and relative
liver weights were increased in animals of each sex at the high dose
and also in males at the intermediate dose. The absolute and relative
adrenal weights were decreased in females at the high dose. Gross
examination revealed no remarkable findings, but microscopic
examination showed treatment-related hepatocellular cytoplasmic
alterations in animals of each sex at the high dose. There was no
evidence of degeneration or necrosis. The NOEL was 600 mg/kg diet,
equivalent to 30 mg/kg bw per day (Lina et al., 1986).
8-Cyclohexadecen-1-one was tested for mutagenic activity in
S. typhimurium strains TA1535, TA1537, TA1538, TA98, and TA100 with
and without microsomal activation. There was no increase in the number
of revertants of any of the strains tested (Wilmer, 1986).
8-Cyclohexadecen-1-one was also tested for its capacity to induce
micronuclei in animals treated in vivo. Groups of 30 CD-1 mice of
each sex were given the compound by gavage at a dose of 3 or 5 ml/kg
bw (equivalent to 2.8 and 4.6 g/kg bw). Five animals of each sex were
killed at 24 and 48 h and two of each sex at 72 h after treatment.
Examination of the bone marrow showed no increase in the number of
normochromatic erythrocytes over that in control mice (Willems, 1986).
3. ESTIMATE OF DIETARY INTAKE
gamma-Cyclodextrin can be used as a carrier for flavours,
sweeteners, and colours, and it has been proposed for use in this
manner in dry mixes for beverages, soups, dressings, gravies, sauces,
puddings, gelatines, and fillings and also in instant coffee and
instant tea, coffee whiteners, compressed sweets, chewing gum,
breakfast cereals, savoury snacks, crackers, and spices and
seasonings. It is also proposed for use as a carrier for vitamins and
polyunsaturated fatty acids in dry food mixes and in dietary
supplements, as a flavour modifier in soya milk, and as a stabilizer
in bread spreads, frozen dairy desserts, baked goods, bread,
fruit-based fillings, fat-based fillings, processed cheese, and dairy
desserts.
The estimated daily intake of gamma-cyclodextrin from its use in
food has been calculated by the dietary survey approach (Amann et al.,
1998), which is based on food consumption data from the 1989-91
Continuing Survey of Food Intakes by Individuals, in which data were
collected from a representative sample of individuals residing in
households in the United States. Each individual was surveyed over
three successive one-day periods, and the foods consumed were coded
into one of about 6000 different categories. For the purposes of the
calculation, it was assumed that each food (or food component) that
contains gamma-cyclodextrin contains this substance at the highest
feasible concentration. When gamma-cyclodextrin was used as a
component of the food, the intake of that component was calculated
from data on food consumption. The estimated daily intake was
calculated for each food in which gamma-cyclodextrin may be used from
data on one- and three-day food intake. Intakes were calculated both
per capita and per user. Users were defined as individuals who
consumed a food of at least one of the categories concerned on at
least one occasion.
The largest amounts of gamma-cyclodextrin are consumed with soya
milk and dairy desserts. Relatively high intakes (> 2 g/day) also
result from its use in bread spreads and fruit-based fillings. The
mean one-day intake of gamma-cyclodextrin from all its food uses was
estimated to be 4.1 g. The 90th percentile user was estimated to
ingest about 8.8 g on any one day. The three-day averages are 4 g for
the mean intake and 7.5 g for the 90th pecentile consumer. The intake
of gamma-cyclodextrin from chewing gum was estimated from a separate
survey on chewing-gum intake in the United States, and the average
consumer was estimated to ingest about 0.07 g gamma-cyclodextrin per
day. These data represent a 'worst-case' scenario and are based on the
assumption that gamma-cyclodextrin is used simultaneously in all
possible applications and at the highest feasible concentrations. The
realistic average daily intake of gamma-cyclodextrin would therefore
be lower than the levels indicated above.
4. COMMENTS
The Committee noted that the close structural similarity between
gamma-cyclodextrin and ß-cyclodextrin allows some comparisons to be
made of the metabolism and the toxicity of these two compounds. The
metabolism of gamma-cyclodextrin is different from that of
ß-cyclodextrin, as demonstrated both in vitro and in vivo. In rats
in vivo, gamma-cyclodextrin (at single doses up to 1000 mg/kg bw) is
rapidly metabolized to glucose, presumably by the luminal and/or
epithelial enzymes of the gastrointestinal tract. In contrast to the
metabolism of ß-cyclodextrin, there was little involvement of the
gastrointestinal microflora. Similarly, in contrast to ß-cyclodextrin,
much less gamma-cyclodextrin was absorbed and only very low levels
could be detected in the urine. After intravenous injection,
gamma-cyclodextrin (at single doses up to 600 mg/kg bw) was rapidly
cleared from the blood and was excreted, largely unchanged, in the
urine. Although no studies of metabolism in humans in vivo were
available, gamma-cyclo-dextrin, unlike ß-cyclodextrin, can be readily
hydrolysed by human salivary and pancreatic amylases in vitro.
Short-term (28- and 90-day) studies of toxicity indicate that
gamma-cyclodextrin has little toxicity when given intravenously or
orally to rats or orally to dogs. After administration of a very high
dietary concentration (20%), caecal enlargement and associated changes
were seen in both species. This effect is likely to result from the
presence of a high concentration of an osmotically active substance in
the large intestine. This result suggests that the metabolism of
gamma-cyclodextrin is less efficient at doses higher than those used
in the studies of metabolism. The results of the studies in rats
treated intravenously indicate that gamma-cyclodextrin is well
tolerated, even when given systemically, and may be less toxic than
ß-cyclodextrin.
Studies conducted in rats and rabbits with gamma-cyclodextrin at
doses up to 20% of the diet did not indicate any teratogenic effects.
Similarly, the results of a battery of studies of genotoxicity were
negative. Long-term studies of toxicity, carcinogenicity, and
reproductive toxicity have not been conducted, but, given the rapid
metabolism of this substance to glucose and its lack of genotoxicity,
the Committee concluded that such studies were not required for an
evaluation.
In vitro, gamma-cyclodextrin, like ß-cyclodextrin, sequestered
components of the membranes of erythrocytes, causing haemolysis. The
threshold concen-tration for this effect was, however, higher than
that of ß-cyclodextrin. Furthermore, gamma-cyclodextrin was not
detected in blood after dietary administration of high doses to
animals in vivo.
It was considered unlikely that interaction of gamma-cyclodextrin
with lipophilic vitamins would impair their bioavailability, because
of the rapid digestion of gamma-cyclodextrin in vivo. Also, there
was no evidence that vitamin deficiency was induced in experimental
animals given high doses of gamma-cyclodextrin.
The Committee considered studies on the short-term toxicity and
genotoxicity of the enzyme, cyclodextrin-glycosyl transferase, used in
the production of cyclodextrins, and of the complexant,
8-cyclohexadecen-1-one, used to optimize formation of
gamma-cyclodextrin. The data indicated that these substances are
unlikely to be of toxicological concern in final preparations of
gamma-cyclodextrin that comply with the specifications. The Committee
also considered information on genetic modifications in the organism
used to produce the enzyme, which raised no concern.
Although no studies of human tolerance to gamma-cyclodextrin were
submitted to the meeting, the Committee was aware that such a study
was available. Although it was unable to review the data, the
Committee was reassured by the relatively low toxicity of this
compound in animals and the fact that it is less toxic than
ß-cyclodextrin, for which studies of human tolerance were available.
Also, its rapid metabolism to maltose, maltotriose, and glucose
in vitro by human salivary and pancreatic amylases supports the
conclusion that, as in laboratory animals, it would be metabolized to
innocuous metabolites before absorption by humans.
5. EVALUATION
The Committee concluded that there were sufficient data to
allocate a temporary ADI 'not specified' but that the study of human
tolerance known to have been conducted should be reviewed in order to
confirm the absence of adverse gastrointestinal symptoms at normal
levels of intake. These data should be submitted for consideration by
the Committee by 1999.
6. REFERENCES
Abdullah, M., French, D. & Robyt, J.F. (1966) Multiple attack by
alpha-amylases. Arch. Biochem. Biophys., 114, 595-598.
Amann, M., Douglass, J., Doyle, E. & Heimbach, J. (1998) Estimated
intake for gamma-cyclodextrin in the USA from food use. Unpublished
report from TAS-Environ, Arlington, Virginia, USA. Submitted to WHO by
Bioresco Ltd, Binningen, Switzerland.
Bär, R. & Ulitzur, S. (1994) Bacterial toxicity of cyclodextrins:
Luminuous Escherichia coli as a model. Appl. Microbiol.
Biotechnol., 41, 574-577.
de Bie, A.T.H.J. & van Ommen, B. (1994) Oral and intravenous
disposition study with [14C] gamma-cyclodextrin in rats. Unpublished
report No. V93.279 from TNO-CIVO Institutes, Zeist, Netherlands.
Submitted to WHO by Bioresco Ltd, Binningen, Switzerland.
de Bie, A.T.H.J., van Ommen, B. & Bär, A. (1998) Disposition of
14C-gamma-cyclodextrin in germfree and conventional rats. Regul.
Toxicol. Pharmacol., submitted for publication.
Blijleven, W.G.H. (1991). Examination of gamma-cyclodextrin for
mutagenic activity in the Ames test. Report No. V91.027. Unpublished
report from TNO-CIVO Institute, Zeist, Netherlands. Submitted to WHO
by Bioresco Ltd, Binningen, Switzerland.
Dickhaus, S. & Heisler, E. (1985) Acute toxicological study of
8-cyclohexadecenon after oral application on the rat. Unpublished
report from Pharmatox Beratung und Forschung GmbH, Sehnde, Germany.
Submitted to WHO by Bioresco Ltd, Binningen, Switzerland.
Ehling G., Donaubauer, H.H., Mayer D., Langer, K.H. & Schmidts, H.L.
(1992) gamma-Cyclodextrin; repeated-dose (3-month) intravenous
toxicity in rats. Unpublished report No 92.0516 from Hoechst AG,
Frankfurt a.M., Germany. Submitted to WHO by Bioresco Ltd, Biningen,
Switzerland.
French D. (1957) The Schardinger dextrins. Adv. Carbohydr. Chem.,
12, 189-260.
Frijlink, H.W., Eissens, A.C., Hefting, N.R., Poelstra, K., Lerk, C.F
& Meijer, D.K.F. (1991) The effect of parenterally administered
cyclodextrins on cholesterol levels in the rat. Pharm. Res., 8,
9-16.
Fuchs, H., Donaubauer, H.H., Mayer D., Langer K.H. & Krieg K. (1992)
gamma-Cyclodextrin; repeated-dose (1-month) intravenous toxicity study
in rats. Unpublished report No 91.1007 from Hoechst AG, Frankfurt,
Germany. Submitted to WHO by Bioresco Ltd, Biningen, Switzerland.
Immel, H.R. (1991) Examination of gamma-cyclodextrin in the
micronucleus test. Unpublished report No. V90.436 from TNO-CIVO
Institutes, Zeist, Netherlands. Submitted to WHO by Bioresco Ltd,
Binningen, Switzerland.
Irie, T., Otagiri, M., Sunada, M., Uekama, K., Ohtani, Y., Yamada, Y.
& Sugiyama, Y. (1982) Cyclodextrin-induced hemolysis and shape changes
of human erythrocytes in vitro. J. Pharm. Dyn., 5, 741-744.
Jonker, D. (1994) Subchronic (13-week) oral toxicity study with an
aqueous solution of alpha-cyclodextrin-glycosyl-transferase
(alpha-CGT) in rats. Unpublished report No. V94.332 from TNO Nutrition
and Food Research Institute, Zeist, Netherlands. Submitted to WHO by
Bioresco Ltd, Binningen, Switzerland.
Kondo, H., Nakatani, H. & Hiromi, K. (1990) In vitro action of human
and porcine alpha-amylases on cyclo-maltooligosaccharides.
Carbohydr. Res., 204, 207-213.
Leroy-Lechat, F., Wouessidjewe, D., Andreux, J.P., Puisieux, F. &
Duchne, D. (1994) Evaluation of the cytotoxicity of cyclodextrins and
hydroxypropylated derivatives. Int. J. Pharm., 101, 97-103.
Lina, B.A.R. (1992a) Subchronic (90-day) oral toxicity study,
including a recovery study, with alpha-cyclodextrin and
gamma-cyclodextrin in rats. Unpublished report No. V 90.401 from
TNO-CIVO Institutes, Zeist, Netherlands. Submitted to WHO by Bioresco
Ltd, Biningen, Switzerland.
Lina, B.A.R. (1992b) Sub-acute (28-day) oral toxicity study with
8-cyclohexadecenone in mice. Unpublished report No. V91.509 from TNO
Nutrition and Food Research Institute, Zeist, Netherlands. Submitted
to WHO by Bioresco Ltd, Binningen, Switzerland.
Lina, B.A.R. & Bär, A. (1998) Subchronic oral toxicity studies with
gamma-cyclodextrin in rats. Regul. Toxicol. Pharmacol., submitted
for publication.
Lina, B.A.R. & Jonker, D. (1989) Tentative (2-week) oral toxicity
study with gamma-CD in rats. Unpublished report No. V89.263/298033
from TNO-CIVO Institutes, Zeist, Netherlands. Submitted to WHO by
Bioresco Ltd, Binningen, Switzerland.
Lina, B.A.R., van Nesselrooij, J.H.J. & Beems, R.B. (1986) Sub-acute
(28-day) oral toxicity study with 8-cyclohexadecenone in rats.
Unpublished report No. V86.498/260392 from TNO-CIVO Institutes, Zeist,
Netherlands. Submitted to WHO by Bioresco Ltd, Binningen, Switzerland.
Marshall J.J. & Miwa I. (1981) Kinetic difference between hydrolyses
of gamma-cyclodextrin by human salivary and pancreatic alpha-amylases.
Biochem. Biophys. Acta, 661, 142-147.
Matsuda K., Mera, Y., Segawa, Y., Uchida, I., Yokomine, A. & Takagi,
K. (1983) Acute toxicity study of gamma-cyclodextrin (gamma-CD) in
mice and rats. Pharmacometrics, 26, 287-291.
Matsuda, K., Segawa, Y., Yokomine, A., Uchida, I., Mera, Y. &
Tokugawa, H. (1985) Biodisposition of gamma-cyclodextrin (gamma-CD) in
the rabbit and dog after intravenous administration. Oyo Yakuri
(Pharmacometrics), 30, 889-896.
Ohtani, Y., Irie, T., Uekama K., Fukunaga, K. & Pitha, J. (1989)
Differential effects of alpha-, ß-, and gamma-cyclodextrins on human
erythrocytes. Eur. J. Biochem., 186, 17-22.
Okada, Y., Kubota, Y., Koizumi, K., Hizukuri, S., Ohfuji, T. & Ogata,
K. (1988) Some properties and the inclusion behavior of branched
cyclodextrins. Chem. Pharm. Bull., 36, 2176-2185.
Prinsen, M.K. (1990) Acute eye irritation/corrosion study with
gamma-cyclodextrin in albino rabbits. Unpublished report No.
V90.405/200069 from TNO-CIVO Institutes, Zeist, Netherlands. Submitted
to WHO by Bioresco Ltd, Binningen, Switzerland.
Prinsen, M.K. (1992) Sensitization study with gamma-CD in guinea pigs
(maximization test). Unpublished report No. V92.575/352063 from
TNO-CIVO Institutes, Zeist, Netherlands. Submitted to WHO by Bioresco
Ltd, Binningen, Switzerland.
Riebeek, W.M. (1990a) Determination of the acute intravenous toxicity
of gamma-cyclodextrin in mice. Unpublished report No. V90.222 from
TNO-CIVO Institutes, Zeist, Netherlands. Submitted to WHO by Bioresco
Ltd, Binningen, Switzerland.
Riebeek, W.M. (1990b) Determination of the acute intravenous toxicity
of gamma-cyclodextrin in rats. Unpublished report No. V90.220 from
TNO-CIVO Institutes, Zeist, Netherlands. Submitted to WHO by Bioresco
Ltd, Binningen, Switzerland.
Riebeek, W.M. (1990c) Determination of the acute intraperitoneal
toxicity of alpha-cyclodextrin and gamma-cyclodextrin in rats.
Unpublished report No. V90.233 from TNO-CIVO Institutes, Zeist,
Netherlands. Submitted to WHO by Bioresco Ltd, Binningen, Switzerland.
Spanjers, M.T. (1986) Determination of the acute oral toxicity (limit
test) of 8-cyclohexadecenone in mice. Unpublished report No.
V86.455/260393 from TNO-CIVO Institutes, Zeist, Netherlands. Submitted
to WHO by Bioresco Ltd, Binningen, Switzerland.
Szejtli, J., Csherhti, T. & Szgyi, M. (1986) Interactions between
cyclodextrins and cell-membrane phospholipids. Carbohydr. Polymers,
6, 35-49.
Til, H.P. & Bär, A. (1998) Subchronic (13-week) oral toxicity study of
gamma-cyclodextrin in dogs. Regul. Toxicol. Pharmacol., submitted
for publication.
Til, H.P. & van Nesselrooij (1992) Subchronic (90-day) feeding study
with gamma-CD in dogs. Unpublished report No. V91.475 from TNO-CIVO
Institutes, Zeist, Netherlands. Submitted to WHO by Bioresco Ltd,
Binningen, Switzerland.
Verhagen, F.J.J. & Waalkens-Berendsen, D.H. (1991) Oral
embryotoxicity/teratogenicity study with alpha-cyclodextrin and
gamma-cyclodextrin in rats. Unpublished report No. V91.210 from TNO
Nutrition and Food Research Institute, Zeist, Netherlands. Submitted
to WHO by Bioresco Ltd, Binningen, Switzerland.
de Vogel, N. & van Delft, J.H.M. (1996) Chromosome aberration test
with gamma-cyclodextrin in cultured human lymphocytes. Unpublished
report No. V96.398 from TNO-CIVO Institutes, Zeist, Netherlands.
Submitted to WHO by Bioresco Ltd, Binningen, Switzerland.
Waalkens-Berendsen, D.H. & Smits-van Prooije, A.E. (1992) Final report
of an oral embryotoxicity/teratogenicity study with alpha-cyclodextrin
and gamma-cyclodextrin in New Zealand white rabbits. Unpublished
report No. V91.564 from TNO Nutrition and Food Research Institute,
Zeist, Netherlands. Submitted to WHO by Bioresco Ltd, Binningen,
Switzerland.
Willems, M.I. (1986) Evaluation of 8-cyclohexadecenone in the
micronucleus test. Unpublished report No. V86.456/260393 from TNO-CIVO
Institutes, Zeist, Netherlands. Submitted to WHO by Bioresco Ltd,
Binningen, Switzerland.
Wilmer, J.W.G.M. (1986) Examination of 8-cyclohexadecenone for
mutagenic activity in the Ames test. Unpublished report No.
V86.264/260064 from TNO-CIVO Institutes, Zeist, Netherlands. Submitted
to WHO by Bioresco Ltd, Binningen, Switzerland.
Yim, D.G., Sato, H.H., Park, Y.H. & Park, Y.K. (1997) Production of
cyclodextrin from starch by cyclodextrin glycosyltransferase from
Bacillus firmus and characterization of purified enzyme. J. Ind.
Microbiol. Biotechnol., 18, 402-405.
Yoshida, A., Arima, H., Uekama, K. & Pitha, J. (1988) Pharmaceutical
evaluation of hydroxyalkyl ethers of ß-cyclodextrins. Int. J.
Pharmacol., 46, 217-222.
gamma-Cyclodextrin has not been reviewed previously by the
Committee; however, the structurally related ß-cyclodextrin (seven
glucose units) was evaluated at the forty-first and forty-fourth
meetings of the Committee (Annex 1, references 107 and 116).
2. BIOLOGICAL DATA
2.1 Biochemical aspects
2.1.1 Absorption, distribution, biotransformation, and excretion
Early experiments provided some evidence that gamma-cyclodextrin
(unlike alpha- or ß-cyclodextrin) is hydrolysed by salivary amylase
(French, 1957). More recent studies have confirmed that both human
salivary amylase and human or porcine pancreatic amylase can readily
hydrolyse gamma-cyclodextrin, yielding mainly maltose, some
maltotriose, and smaller amounts of glucose (Abdullah et al., 1966;
Marshall & Miwa, 1981; Kondo et al., 1990).
The fate of intravenously administered gamma-cyclodextrin was
examined in three male and two female rabbits and one male dog. The
rabbits were each given a dose of 7.5 g in saline (30 ml of a 25% w/v
solution), while the dog was given a dose of 25 g in saline (500 ml of
a 5% w/v solution at 8.3 ml/min). Blood and urine samples were
collected and examined by high-performance liquid chromatography
(HPLC) for gamma-cyclodextrin over 4 h. The half-life of
gamma-cyclo-dextrin in blood was about 50 min in rabbits and 30 min in
the dog. In rabbits, about 77% of the injected dose was recovered as
unchanged gamma-cyclodextrin in the urine within 24 h, while in the
dog, nearly 80% of the injected dose was recovered as unchanged
gamma-cyclodextrin in the urine within the first 4 h (Matsuda et al.,
1985).
Two male Wistar rats were given 14C-gamma-cyclodextrin (94%
radiochemical purity) as single oral or intravenous doses. After an
oral dose of 14C-gamma-cyclo-dextrin (25 µCi/kg bw diluted to 200
mg/kg bw), 50% of the radiolabel was excreted in the exhaled air
within 24 h, while 2% was excreted in the urine and 5% in the faeces;
35% of the radiolabel was retained in the body after 24 h. No
detectable gamma-cyclodextrin was found in blood during 8 h after
administration, suggesting metabolism in the gastrointestinal tract.
After the intravenous dose of 14C-gamma-cyclodextrin (25 µCi/kg
bw diluted to 100 mg/kg bw), 90% of the radiolabel was excreted in the
urine within 8 h, mainly as unchanged gamma-cyclodextrin. After 8 h,
small amounts of radiolabel were excreted in exhaled air (1.1%), while
0.5% was detected in the faeces; 6% was retained in the body after 24
h (de Bie & van Ommen, 1994).
Groups of four male Wistar rats were given single oral or
intravenous doses of 14C-gamma-cyclodextrin. The oral dose was 1000
mg/kg bw, and the concentration of radiolabel was either 25 or 100
µCi/kg bw. In order to examine the role of the gut microflora in the
metabolism of gamma-cyclodextrin, a separate group of germ-free male
rats was given 25 µCi/kg bw gamma-cyclodextrin diluted to 1000 mg/kg
bw. The intravenous dose was 600 mg/kg bw, and the concentration of
radiolabel was 25 µCi/kg bw.
In the first experiment, the time-course and routes of metabolism
were examined after oral administration. After administration of
1000 mg/kg bw (25 µCi/kg bw) 14C-gamma-cyclodextrin, 62% of the
radiolabel in males and 71% in females was excreted within 48 h. The
majority was excreted in expired air (57% in males and 65% in
females), with maximum excretion between 1 and 2 h after dosing.
Little was excreted in the urine (2.1% for males and 2.5% for females)
or faeces (2.9% for males and females) during the 48-h sampling
period. Retention of radiolabel after 48 h was 27% in males and 22% in
females, with the most significant retention in the liver (11% in
males, 5.2% in females). The amounts in all other organs were low:
intestine, 0.42% for males, 0.36% for females; kidney, 0.13% for
males.
In the second experiment, the excretion kinetics and time-course
in blood over 24 h were examined after a single oral dose of
1000 mg/kg bw (100 Ci/kg bw) 14C-gamma-cyclodextrin. The major route
of excretion was exhaled air (55% for males; 53% for females) during
the 24-h sampling period. The carcass retained 35% of the radioactive
dose in males and 25% in females at 24 h after treatment. The maximum
concentration in blood was reached 40 min after treatment, but this
concentration decreased by 50% after 0.5 h in males and after 6 h in
females. The urine of animals of each sex contained a peak that eluted
before glucose at all times and a small peak that co-eluted with
glucose (6% of total radiolabel). A small amount of unchanged
gamma-cyclo-dextrin was detected in urine after 4 h (< 0.02% of the
ingested dose). HPLC analysis of the stomach contents 24 h after
dosing revealed the presence of gamma-cyclodextrin and a minor peak
that co-eluted with glucose. HPLC analysis of the blood revealed the
presence of a radioactive peak that co-eluted with glucose.
In the third experiment, the excretion kinetics and time-course
in blood over 24 h were examined after a single intravenous dose of
600 mg/kg bw (25 µCi/kg bw) 14C-gamma-cyclodextrin. The major route
of excretion was the urine (87% for males, 91% for females), and the
faeces contained about 1% and the gastrointestinal tract about 0.5% of
the radiolabel; the exhaled air contained 7.8% of the dose for males
and 3.2% for females. The carcass contained 3.9% of the dose in males
and 2.2% in females, the liver having the most residue (0.41% in
males, 0.42% in females). Radiolabel was removed rapidly from the
blood, with calculated initial half-lives of 20 min in males and 15
min in females. HPLC analysis of blood revealed predominantly
gamma-cyclodextrin.
In the fourth experiment, excretion kinetics over 24 h was
examined in germ-free rats given a single oral dose of 1000 mg/kg bw
(25 µCi/kg bw) gamma-cyclodextrin. About 73% of the radiolabel was
excreted in expired air (68% in males, 65% in females). Peak
exhalation of 14C-carbon dioxide occurred at about 90 min. Low
concentrations of radiolabel were found in faeces and urine. The
gastrointestinal tract contained 9.2% in males and 11% in females,
with the majority in the caecum. The results indicate that
gastrointestinal microflora play little part in the metabolism of
gamma-cyclodextrin.
Overall, this study indicates that gamma-cyclodextrin is
metabolized to glucose, other low-molecular-mass sugar metabolites,
and carbon dioxide by luminal and/or epithelial enzymes of the
gastrointestinal tract (de Bie & van Ommen, 1994; de Bie et al.,
1998).
2.2 Toxicological studies
2.2.1 Acute toxicity
The acute toxicity of gamma-cyclodextrin was examined in mice and
rats; the results are summarized in Table 1. In each study, groups of
10 animals of each sex were tested at three doses or served as
controls. There were no deaths at the highest doses tested (Matsuda et
al., 1983).
Table 1. Acute toxicity of gamma-cyclodextrin
Species Sex Route LD50 Reference
(mg/kg bw)
Mouse Male and female Oral >16 000 Matsuda et al. (1983)
Mouse Male and female Subcutaneous > 4 000 Matsuda et al. (1983)
Mouse Male and female Intravenous > 4 000 Matsuda et al. (1983)
Mouse Male and female Intravenous 10 000 Riebeek (1990a)
Rat Male and female Oral > 8 000 Matsuda et al. (1983)
Rat Male and female Subcutaneous > 2 400 Matsuda et al. (1983)
Rat Male and female Intravenous > 3 750 Matsuda et al. (1983)
Rat Male and female Intravenous > 3 750 Riebeek (1990b)
Rat Male Intraperitoneal > 4 600 Riebeek (1990c)
In a test for micronucleus formation (see section 2.2.3), groups
of 15 Swiss mice of each sex were given a single dose of 15 g/kg bw
gamma-cyclodextrin by gavage and were killed in groups of five at 24,
48, and 72 h. All of the animals survived until termination (Immel,
1991).
The toxicity of intravenously administered gamma-cyclodextrin was
examined in groups of five male and five female Crl:CD mice and five
male and five female Crl:W1(WU)BR rats. The mice were given
gamma-cyclodextrin (purity, > 98%) at doses of 5000, 7500, or 10 000
mg/kg bw, while the rats were given doses of 2500, 3750, or 5000 mg/kg
bw as a solution in sterile saline. The animals were observed for
clinical signs over 14 days after treatment. They were examined for
gross pathological changes on day 14 after treatment.
Both mice and rats showed dose-related signs of toxicity, such as
piloerection and sluggishness, within 1 h to a few days after
treatment. Deaths occurred within a few days in both species, but the
surviving animals recovered and appeared to be healthy at the end of
the observation period. Macroscopic examination revealed no gross
treatment-related alterations (Riebeek, 1990a,b).
The toxicity of intraperitoneally administered gamma-cyclodextrin
was examined in groups of three male rats after single doses of 2000
or 4600 mg/kg bw as a solution in sterile saline. The animals were
observed for clinical signs during 14 days after treatment and were
examined grossly for pathological changes on the last day. There were
no signs of toxicity and no deaths. Macroscopic examination revealed
no gross treatment-related alterations (Riebeek, 1990c).
2.2.2 Short-term studies of toxicity
Rats
Groups of 15 Wistar rats of each sex were given
gamma-cyclodextrin intravenously as single daily doses of 0, 200, 630,
or 2000 mg/kg bw for 30 days. A group of five animals of each sex were
allowed to recover for 27 days after treatment. The animals were
examined throughout the study, and the body weights recorded weekly.
Clinical chemical parameters were examined at the end of treatment and
after the recovery period. Ten animals from each group were killed at
the end of treatment and the remainder at the end of the recovery
period.
There were no clinical signs of toxicity throughout the study.
The body-weight gain of males at 630 and 2000 mg/kg bw per day was
slightly decreased, but the difference was not significant. Food
consumption was decreased in a dose-related manner only during the
first few days of treatment. Significantly decreased mean values for
haemoglobin concentration and haemacrit were seen in males at doses
> 630 mg/kg bw per day, and significantly decreased erythrocyte
count in males at 2000 mg/kg bw per day. In females, the erythrocyte
count, haemoglobin concentration, and haematocrit were significantly
decreased only at 2000 mg/kg bw per day. The reticulocyte counts were
significantly increased in both males and females at 2000 mg/kg bw per
day. These changes were reversed after the recovery period. The only
significant clinical chemical change was an increase in creatinine and
urea in both males and females at 2000 mg/kg bw per day, which was
reversed at the end of the recovery period. Urinalysis revealed a
significantly increased incidence of haemoglobin in the urine of rats
at 2000 mg/kg bw per day, which was reversed at the end of the
recovery period.
Pathological examination at autopsy showed that the majority of
animals at 2000 mg/kg bw per day had light-coloured and, in some
cases, irregularly coloured kidneys. A dose-dependent increase in
spleen weight was found in all groups, which was significant in males
at doses > 630 mg/kg bw per day and in females at all doses. Liver
weights were significantly increased in females at > 630 mg/kg bw
per day, and kidney weights were significantly increased at 2000 mg/kg
bw per day. Lung and adrenal weights were significantly increased in
both males and females at 2000 mg/kg bw per day. These changes were
partially reversed after the recovery period, but a significant
increase in kidney and spleen weights was still present in females at
2000 mg/kg bw per day. Histopathological examination revealed marked
vacuolation of renal epithelial cells in the proximate convoluted
tubules, which may have been related to administration of a substance
that causes hyperosmolarity. There was also extensive pulmonary
histiocytosis (massive accumulation of alveolar macrophages) in all
animals at 2000 mg/kg bw per day. These changes were seen only in some
animals at 630 mg/kg bw per day and at lesser severity. Considerable
reversibility was observed after the recovery period. The NOEL was 200
mg/kg bw per day (Fuchs et al., 1992).
Groups of 15 Wistar rats of each sex were given
gamma-cyclodextrin intravenously as single daily doses of 0, 60, 120,
or 600 mg/kg bw for 90 days. Groups of five animals of each sex were
allowed to recover at the end of the treatment period. The animals
were examined throughout the study, and body weights were recorded
weekly. Haematological and urinary parameters were examined after one
month, at the end of the study, and on day 37 (males) or 36 (females)
of recovery. Clinical chemical parameters were examined at the end of
the treatment period and after recovery. Animals were killed after
90 days, their organs were examined, and tissues were taken for
histopathological examination.
There were no clinical signs of toxicity. Body-weight gain was
slightly decreased only in males at the high dose, and food
consumption was decreased in a dose-related manner only during the
first few days of treatment. Haematological examination revealed a
slight decrease in the mean erythrocyte count, haemaglobin
concentration, and haemacrit in females at one month, and these values
were significantly decreased at three months only in females at the
high dose. The reticulocyte counts were significantly increased in
females at the high dose at three months. At the end of the recovery
period, all of these values were normal. The only clinical chemical
changes were a slight decrease in bilirubin concentration in males and
females at the high dose and a slight increase in alkaline phosphatase
activity in males at the high dose. At the end of the recovery period,
all of the values were normal. The slight changes observed in urinary
parameters were considered not to be treatment-related.
Histological examination at autopsy revealed an increased
incidence of enlarged iliac lymph nodes in males and females at the
high dose, which was probably related to inflammation at the injection
site. Increases in the relative weights of lungs, liver, and spleen
were seen in males and in the relative weights of heart, lungs,
kidneys, spleen, and adrenal glands in females at the high dose. All
of these changes were reversed after the four-week recovery period.
Histopathological examination revealed hyperplasia of the mucosa of
the urinary bladder, which was reversed after four weeks, and
pulmonary histiocytosis (macrophage aggregates), which tended to
reverse after four weeks, in males and females at the high dose. The
reversibility of the urinary bladder changes indicates that the
hyperplasia was not preneoplastic. The NOEL was 120 mg/kg bw per day
(Ehling et al., 1992).
Groups of five male Wistar rats were fed diets containing
gamma-cyclo-dextrin at concentrations of 0, 5, 10, 15, or 20%,
equivalent to 0, 2500, 5000, 7500, or 10 000 mg/kg bw per day, for 14
days. A control group was fed 20% lactose. The animals were examined
for clinical signs of toxicity, and food and water consumption was
monitored throughout the study. On day 14, the animals were killed,
and blood samples were taken for examination of clinical chemical
parameters. The animals were also examined macroscopically for
pathological changes.
There were no deaths. The incidence of soft stools was slightly
higher in treated than control animals, and the mean body weights of
treated animals were slightly lower than those of controls, although
this difference was not statistically significant. Food intake was
slightly lower in controls than in treated groups. Plasma alkaline
phosphatase activity was increased in animals at 15 and 20%, while
gamma-glutamyl transferase activity was decreased in all treated
groups. The activity of aspartame and alanine aminotransferases were
unchanged, as were the concentrations of total protein, albumin, urea,
and bilirubin. Enlargement of the caecum was seen at all doses, the
most significant effect occurring in the group fed 20% lactose. There
were no macroscopic changes attributable to treatment (Lina & Jonker,
1989; Lina & Bär, 1998).
Groups of 20 Wistar rats of each sex were fed diets containing
gamma-cyclo-dextrin at concentrations of 0, 1.5, 5, or 20% (equivalent
to 0, 750, 2500, or 10 000 mg/kg bw per day) for 13 weeks. A control
group was fed 20% lactose. In order to examine the reversibility of
any effects seen, a group of 10 animals of each sex were fed either
20% gamma-cyclodextrin or 20% lactose for 13 weeks and then control
diet for one month. As a result of a feeding error, the groups
receiving 20% gamma-cyclodextrin were discarded, and two additional
groups were fed 0 or 20% gamma-cyclodextrin for 13 weeks and 10
animals receiving the 20% dose were fed control diet for an additional
month at the end of the 13-week period. Animals were examined for
clinical signs of toxicity, and food and water consumption was
monitored throughout the study. Ophthalmoscopic examinations were
conducted in animals at the high dose and those given lactose before
treatment and at the end of the study. The animals were killed at the
end of the study, and blood samples were taken for clinical chemistry.
The animals were also examined macroscopically for pathological
changes.
There were no treatment-related deaths during the study. Soft
stools were observed in some rats at 5 or 20% gamma-cyclodextrin and
in those given lactose early in the study, but this effect disappeared
subsequently. Ophthalmoscopic examination revealed no
treatment-related effects. The body weights of males at 20%
gamma-cyclodextrin were slightly but significantly decreased
throughout treatment; a more significant body-weight decrease was seen
in the males given 20% lactose. No difference was noted in these
groups during the recovery period. Food conversion efficiency was also
slightly decreased in males at 20% lactose. None of the changes in
haematological parameters was considered to be of toxicological
significance. Some changes in clinical chemistry were seen in females
at the high dose, namely increased activity of aspartate and alanine
aminotransferases, especially in one female. The sodium concentrations
were slightly higher but within the historical control range. Urinary
parameters were similar in control and treated groups, apart from a
significant increase in the urinary calcium concentration at the end
of treatment in males given 20% gamma-cyclodextrin and rats of each
sex given 20% lactose; the effect was more pronounced in the latter
group. The calcium:creatinine ratio was significantly increased in
rats receiving 20% lactose. The increased calcium appeared to be
associated with the increased load of osmotically active substances in
the large intestine; there was no increase in calcium at the end of
the recovery period.
Analysis of organ weights revealed significant increases in both
absolute and relative caecal weights in rats at 5 and 20%
gamma-cyclodextrin and 20% lactose in comparison with controls. At the
end of the recovery period, no difference from controls was seen for
rats given 20% gamma-cyclodextrin, but the increase was still evident
in those given 20% lactose. This effect is probably related to the
retention of an osmotically active substance in the large intestine.
Relative adrenal weights were increased in males given 20%
gamma-cyclodextrin and the lactose control group at the end of the
study. A very slight increase in relative liver weight was seen in
males at 20% gamma-cyclodextrin and females given lactose. No changes
in organ weight were seen at the end of the recovery period.
There were no gross pathological changes attributable to
treatment with gamma-cyclodextrin. Microscopic examination revealed
increased cortico-medullary mineralization in the kidneys of rats at
1.5 and 5% gamma-cyclodextrin but not at the high dose. This change is
considered to be relatively common in rats and not related to
treatment with gamma-cyclodextrin. There was no evidence of
hyperplasia of the mucosa of the urinary bladder or of pulmonary
histiocytosis, which were observed after intravenous administration.
The effects observed in rats after receiving gamma-cyclodextrin
in the diet at concentrations up to 20% appeared to be largely related
to the presence of high levels of an osmotically active substance in
the large intestine and not of toxicological significance. Similar,
and generally more pronounced, effects were observed when the diet
contained 20% lactose. On the basis of this study, dietary levels of
20% gamma-cyclodextrin (equivalent to10 000 mg/kg bw per day) were
considered to be tolerated without toxicological effects (Lina, 1992a;
Lina & Bär, 1998).
Dogs
Groups of four male and four female beagle dogs were fed diets
containing gamma-cyclodextrin at doses of 0, 5, 10, or 20% (equivalent
to 0, 1250, 2500, or 5000 mg/kg bw per day). Body weight and food
consumption were recorded weekly throughout the study. Ophthalmoscopic
examinations were made at the beginning and end of the study. Blood
was collected for clinical chemical and haematological examination
before the start of treatment and at weeks 6 and 13. Urinalyses were
performed at week 13. During week 14, all animals were killed, their
organs were examined, and tissues were prepared for histopathological
examination.
There were no deaths during the study and no clinical signs of
toxicity. Diarrhoea was noted in all groups, increasing in incidence
and severity with increasing doses of gamma-cyclodextrin, and was more
severe in females than in males. This effect was attributed to the
slow digestion and intestinal absorption of gamma-cyclodextrin.
Ophthalmoscopic examination revealed no differences between treated
and control animals. Weight gain was comparable in all groups, except
for a slight, non-significant decrease in males at 20% during the last
six weeks of the study. Treated dogs had slightly decreased food
intake during the first two weeks of the study, particularly at the
high dose. This effect persisted throughout the study only in dogs at
the high dose. No differences in haematological parameters were seen,
and clinical chemical parameters were similar in treated and control
groups. Urinalysis revealed a slight decrease in urinary pH in the
group receiving 20% gamma-cyclodextrin, which was considered to be
associated with the intake of poorly digestible carbohydrates and
increased fermentation in the large intestine.
Both the absolute and relative weights of the ovary were
increased in animals at 10 and 20% gamma-cyclodextrin, but no
histopathological changes were seen, and the weight of the ovaries in
the control group was lower than that of other dogs in the same
laboratory. The changes observed are considered unlikely to be
toxicologically significant. There was no change in absolute liver
weight, although the relative liver weight was increased in males at
the high dose as a result of the slight decrease in body weight at
this dose. This change is likely to be adaptive and was considered not
to be toxicologically significant. Caecal weights were increased in
the dogs at 10 and 20% gamma-cyclodextrin but statistically
significantly only in females. This change is considered to be related
to the presence of high levels of an osmotically active substance in
the large intestine. Pathological examination at the end of the study
revealed no treatment-related change. Similarly, the histopathological
findings were unremarkable. On the basis of this study, dietary levels
of 20% gamma-cyclodextrin (equivalent to 5000 mg/kg bw per day) were
considered to be tolerated without toxicological effects (Til & van
Nesselrooij, 1992; Til & Bär, 1998).
2.2.3 Genotoxicity
The results of assays for genotoxicity with gamma-cyclodextrin
are shown in Table 2.
2.2.4 Developmental toxicity
Rats
In a study of embryotoxicity and teratogenicity, groups of 25
presumed pregnant Wistar Crl:(WI)WU BR rats were fed diets containing
gamma-cyclodextrin (purity, >98%) at concentrations of 0. 1.5, 5, 10,
or 20% (equivalent to 0, 2500, 5000, or 10 000 mg/kg bw per day) on
days 0-21 of gestation. A separate group received a diet containing
20% lactose instead of pre-gelatinized potato starch. The animals were
examined throughout the study, and body weight and food consumption
were recorded. The rats were killed on day 21 and examined for
parameters of reproductive performance. Fetuses were examined for
signs of toxicity, external malformations, and soft-tissue defects and
were stained for detection of skeletal anomalies.
No deaths occurred during the study. Maternal body-weight gain
was similar in all groups except for a slight reduction at the 20%
dose on days 0-16 of gestation. Necropsy of the dams showed no adverse
effects that could be related to treatment. The number of viable
litters, the number of corpora lutea, and the mean number of
implantation sites were similar in all groups. Fetal length and body
weight were also similar in all groups. Examination of the fetuses
revealed no treatment-related increase in gross, skeletal, or visceral
abnormalities. Under the conditions of this assay, gamma-cyclodextrin
was not teratogenic (Verhagen & Waalkens-Berendsen, 1991).
Rabbits
In a study of embryotoxicity and teratogenicity, groups of 16
presumed pregnant New Zealand white rabbits were fed diets containing
gamma-cyclodextrin (purity, > 98%) at concentrations of 0, 5, 10, or
20% (equivalent to 0, 1500, 3000, or 6000 mg/kg bw per day) on days
0-29 of gestation. The animals were examined throughout the study, and
body weights and food consumption were recorded regularly. The animals
were killed on day 29 of pregnancy, and the fetuses were examined for
signs of toxicity, external malformations, and soft-tissue defects and
were stained for detection of skeletal anomalies.
Table 2. Results of assays for the genotoxicity of gamma-cyclodextrin
End-point Test object Concentration Result Reference
Bacterial gene S. typhimurium TA1535, 2-20000 Negativea Blijleven (1991)
mutation TA1537, TA 1538, µg/plate
TA98, TA100
Chromosomal Human lymphocytes 10-5000 Negativea de Vogel & van
aberrationb µg/ml Delft (1996)
Micronucleus CD-1 mouse bone 15 g/kg bw Negative Immel (1991)
formationb marrow (single dose)
a With and without exogenous metabolic activation
b Two assays conducted: treatment times, 6, 24, and 48 h; harvest times, 24 and 48 h
No deaths occurred during the study. Diarrhoea was seen in
several treated animals. Maternal body weight was significantly
decreased at the 20% dose during the first week of treatment but not
thereafter. Necropsy of the does showed no adverse effects that could
be related to treatment. The number of viable litters, the number of
corpora lutea, and the mean number of implantation sites were similar
in all groups. One abortion occurred in the control group and one in
rabbits at the 10% dose. Fetal length and body weight were similar in
all groups. Examination of the fetuses revealed no treatment-related
increase in gross, skeletal, or visceral abnormalities. An increase in
the incidence of haemorrhagic fluid in animals at the 5 and 20% doses
was considered unlikely to be treatment-related. Under the conditions
of this assay, gamma-cyclodextrin was not teratogenic
(Waalkens-Berendsen & Smits-van Prooije, 1992).
2.2.5 Special studies
2.2.5.1 Ocular irritation and dermal sensitization
gamma-Cyclodextrin was not irritating or corrosive to the eyes of
albino rabbits (Prinsen, 1990). In a skin sensitization assay in
guinea-pigs, a 30% solution of gamma-cyclodextrin induced no signs of
sensitization (Prinsen, 1992).
2.2.5.2 Cell membrane effects
The interactions between alpha-, beta-, and gamma-cyclodextrin
and membrane phospholipids, liposomes, and human erythrocytes were
studied in vitro. gamma-Cyclodextrin and the other cyclodextrins did
not increase the permeability of dipalmityol-phosphatidylcholine
liposomes, nor did they affect the active transport of 42K+ into
erythrocytes at a concentration of 10-2 mol/L. ß-Cyclo-dextrin but
not gamma-cyclodextrin or alpha-cyclodextrin increased the release of
42K+, 86Rb+, and 137Cs+ from erythocytes by passive transport at
a concentration of 1.7 × 10-2 mol/L (Szejtli et al., 1986).
gamma-Cyclodextrin induced haemolysis of human erythrocytes
in vitro in isotonic saline after 30 min, with a threshold
concentration of 16 mmol/L (approximately 20 mg/L). A similar effect
was seen with alpha-cyclodextrin at 6 mmol/L and with ß-cyclodextrin
at 3.5 mmol/L. At 15 mmol/L, gamma-cylodextrin caused a 2% release of
cholesterol from erythrocytes, indicating that cyclodextrin-induced
haemolysis is associated with removal of membrane components from
erythrocytes (Irie et al., 1982). Other studies have confirmed that
gamma-cyclodextrin causes erythrocyte haemolysis at concentrations of
15-30 mmol/L (Okada et al., 1988; Yoshida et al., 1988; Leroy-Lechat
et al., 1994).
In a study on the differential effects of cyclodextrins on human
erythrocytes in vitro, alpha-, ß-, and gamma-cyclodextrin were
incubated at increasing concentrations with erythrocytes for 30 min.
The cyclodextrins induced a change in shape, from discocyte to
spherocyte, but with ß-cyclodextrin haemolysis occurred before the
change was complete. gamma-Cyclodextrin was less potent than either
alpha- or ß-cyclodextrin in releasing potassium and haemoglobin from
erythrocytes and in solubilizing various components of erythrocytes,
including phospholipids, cholesterol, and proteins. A concentration of
4 mmol/L gamma-cyclodextrin was required to induce release of
phospholipid, and > 20 mmol was required for release of cholesterol
or protein (Ohtani et al., 1989).
The potential cytotoxicity of the cyclodextrins to P388 murine
leukaemia cells was examined by exposing the cells to increasing
concentrations of cyclodextrins for 48 h in medium containing 10%
fetal calf serum. Initial cytotoxicity was elicited at 11 mmol/L
alpha-cyclodextrin, 2.5 mmol/L ß-cyclodextrin, and 55 mmol/L
gamma-cyclodextrin (Leroy-Lechat et al., 1994).
Potential reductions in the luminescence of a strain of E. coli
after incubation with various concentrations of cyclodextrins were
examined. Luminescence was reduced by 20 and 50% by ß-cyclodextrin at
concentrations of 0.03 and 0.5%, respectively. With
gamma-cyclodextrin, a concentration of 2% was required to reduce the
luminescence by 20%, and a 50% reduction could not be achieved even at
the highest concentration of 10% gamma-cyclodextrin (Bär & Ulitzur,
1994).
2.2.5.3 Impurities
Cyclodextrin-glycosyl transferase
The gene coding for cyclodextrin-glycosyl transferase is derived
from a strain of the Bacillus firmus-lintus complex. This is a
ubiquitous group of aerobic, gram-positive, alkalophilic
microorganisms which form sub-terminal endospores. Both B. firmus
and B. lintus are considered to be non-pathogenic. After isolation
from this strain, the gene was cloned into pUC18, isolated again and
inserted into an expression and secretion vector derived from pJF118EH
by introduction of a DNA fragment coding for an appropriate signal
peptide. The vector, pJF118EH, is derived from pBR322, a widely used
vector which is considered to be safe. All genetic modifications were
made by directed mutagenesis by standard techniques. The gene that has
been introduced for production of the signal peptide is cleaved off
during exportation of cyclodextrin-glycosyl transferase and degraged
with the E. coli cell.
A strain derived from E. coli K12 was used as the host for the
cyclodextrin-glycosyl transferase expression and secretion vector.
E. coli K12 is considered to be a non-pathogenic strain and is used
for production of enzymes for food use.
For the production of cyclodextrin-glycosyl transferase, the
recombinant E. coli strain was cultured in a standard medium. The pH
of the fermentation broth was adjusted by adding food-grade ammonia or
phosphoric acid. The E. coli was grown to a certain density, after
which enzyme production was initiated by addition of isopropyl
thiogalactoside. The bacteria were then removed by fermentation and
the supernatant filtered and concentrated to the crude
cyclodextrin-glycosyl transferase preparation.
Groups of 20 Wistar rats of each sex were given an aqueous
solution of a cyclodextrin-glycosyl transferase preparation at doses
of 0, 5, 10, or 20 ml/kg bw per day, equivalent to 0, 65, 130, or 260
mg/kg bw per day of total organic solids (TOS), by gavage for 13
weeks. The animals were examined for clinical signs of toxicity, and
food and water consumption was monitored throughout the study.
Ophthalmoscopic examinations were conducted in rats at the high dose
before treatment and at the end of the study. Haematological
parameters were examined before treatment, on days 30 and 60, and at
the end of the study. The animals were killed at the end of the study,
and blood samples were taken for examination of clinical chemical
parameters. The animals were also examined macroscopically for
pathological changes, and samples were taken for histopathological
examination.
Two animals (one control male and one female at the low dose)
died as a result of dosing accidents. There were no clinical signs of
toxicity in the treated animals which survived. Ophthalmoscopic
examination of animals at the high dose revealed no treatment-related
changes. There was no significant difference in body weight between
treated and control rats, and food consumption was unchanged in the
treated group. There were no treatment-related haematological,
clinical chemical, or urinary changes. The organ weights of control
and treated groups were similar, and there were no treatment-related
gross pathological changes. Histopathological examination revealed an
increased incidence of pulmonary changes in the treated animals which
were considered to be related to aspiration of test material. The NOEL
was 20 ml/kg bw per day, equivalent to 260 mg/kg bw per day of TOS,
the highest dose tested (Jonker, 1994).
The cyclodextrin-glycosyl transferase preparation was tested for
mutagenic activity in S. typhimurium strains TA1535, TA1537, TA98,
and TA100 with and without metabolic activation at concentrations of
19-1500 µg/plate or 62-500 µg/plate. The assay was slightly
complicated by the growth-stimulating effects of some of the
components of the crude preparation, but the overall result confirmed
that cyclodextrin-glycosyl transferase had no mutagenic activity in
these strains.
8-Cyclohexadecen-1-one
8-Cyclohexadecen-1-one is a colourless, waxy, solid product. The
commercial preparation has a purity of > 98%. The acute toxicity of
8-cyclo-hexadecen-1-one was examined in five male and five female mice
at a dose of 5 ml/kg bw (4.6 g/kg bw) and in five male and five female
rats at doses of 5 and 10 g/kg bw. The animals were observed for 14
days. Two of the five mice died during the study, but there were no
deaths among the rats. It was concluded that the LD50 was > 4.6 g/kg
bw in mice and > 10 g/kg bw in rats (Dickhaus & Heisler, 1985;
Spanjers, 1986).
Groups of five CD-1 mice of each sex were fed diets containing
8-cyclo-hexadecen-1-one at doses of 0, 500, 1500, or 7500 mg/kg diet
(equivalent to 0, 75, 230, or 1100 mg/kg bw per day) for four weeks.
The animals were examined for clinical signs of toxicity, and food and
water consumption were monitored throughout the study. Haematological
and clinical chemical parameters were examined before treatment and at
the end of the study. The animals were also examined macroscopically
for pathological changes, and samples were taken for histopathological
examination. There were no deaths and no clinical signs of toxicity.
The body-weight gain of mice at the high dose was decreased, but food
and water intake were similar in treated and control groups. There
were no treatment-related haematological changes. The only clinical
chemical change was a decrease in the albumin:globulin ratio in males
at the high dose. An increase in relative liver weight was seen in
males and females at the high dose. No treatment-related gross
abnormalities were seen, but microscopic examination revealed effects
in the cytoplasm of four males at the high dose. The NOEL was 1500
mg/kg diet, equivalent to 230 mg/kg bw per day (Lina, 1992b).
Groups of five male and five female Wistar rats were fed diets
containing 8 cyclohexadecen-1-one at doses of 0, 600, 3000, or 15 000
mg/kg diet (equivalent to 0, 30, 120, or 750 mg/kg bw per day) for
four weeks. The animals were examined for clinical signs of toxicity,
and food and water consumption was monitored throughout the study.
Haematological and clinical chemical parameters were examined before
treatment and at the end of the study; the animals were also examined
macroscopically for pathological changes, and samples were taken for
histopathological examination. There were no deaths and no clinical
signs of toxicity.
A number of effects were seen in animals at the high dose.
Body-weight gain and food intake were decreased, and the red blood
cell counts and packed cell volume were increased. Clinical chemistry
revealed increased plasma urea and creatinine concentrations in males
and decreased albumin in females, and urinalysis showed increased
urinary density in animals of each sex. The absolute and relative
liver weights were increased in animals of each sex at the high dose
and also in males at the intermediate dose. The absolute and relative
adrenal weights were decreased in females at the high dose. Gross
examination revealed no remarkable findings, but microscopic
examination showed treatment-related hepatocellular cytoplasmic
alterations in animals of each sex at the high dose. There was no
evidence of degeneration or necrosis. The NOEL was 600 mg/kg diet,
equivalent to 30 mg/kg bw per day (Lina et al., 1986).
8-Cyclohexadecen-1-one was tested for mutagenic activity in
S. typhimurium strains TA1535, TA1537, TA1538, TA98, and TA100 with
and without microsomal activation. There was no increase in the number
of revertants of any of the strains tested (Wilmer, 1986).
8-Cyclohexadecen-1-one was also tested for its capacity to induce
micronuclei in animals treated in vivo. Groups of 30 CD-1 mice of
each sex were given the compound by gavage at a dose of 3 or 5 ml/kg
bw (equivalent to 2.8 and 4.6 g/kg bw). Five animals of each sex were
killed at 24 and 48 h and two of each sex at 72 h after treatment.
Examination of the bone marrow showed no increase in the number of
normochromatic erythrocytes over that in control mice (Willems, 1986).
3. ESTIMATE OF DIETARY INTAKE
gamma-Cyclodextrin can be used as a carrier for flavours,
sweeteners, and colours, and it has been proposed for use in this
manner in dry mixes for beverages, soups, dressings, gravies, sauces,
puddings, gelatines, and fillings and also in instant coffee and
instant tea, coffee whiteners, compressed sweets, chewing gum,
breakfast cereals, savoury snacks, crackers, and spices and
seasonings. It is also proposed for use as a carrier for vitamins and
polyunsaturated fatty acids in dry food mixes and in dietary
supplements, as a flavour modifier in soya milk, and as a stabilizer
in bread spreads, frozen dairy desserts, baked goods, bread,
fruit-based fillings, fat-based fillings, processed cheese, and dairy
desserts.
The estimated daily intake of gamma-cyclodextrin from its use in
food has been calculated by the dietary survey approach (Amann et al.,
1998), which is based on food consumption data from the 1989-91
Continuing Survey of Food Intakes by Individuals, in which data were
collected from a representative sample of individuals residing in
households in the United States. Each individual was surveyed over
three successive one-day periods, and the foods consumed were coded
into one of about 6000 different categories. For the purposes of the
calculation, it was assumed that each food (or food component) that
contains gamma-cyclodextrin contains this substance at the highest
feasible concentration. When gamma-cyclodextrin was used as a
component of the food, the intake of that component was calculated
from data on food consumption. The estimated daily intake was
calculated for each food in which gamma-cyclodextrin may be used from
data on one- and three-day food intake. Intakes were calculated both
per capita and per user. Users were defined as individuals who
consumed a food of at least one of the categories concerned on at
least one occasion.
The largest amounts of gamma-cyclodextrin are consumed with soya
milk and dairy desserts. Relatively high intakes (> 2 g/day) also
result from its use in bread spreads and fruit-based fillings. The
mean one-day intake of gamma-cyclodextrin from all its food uses was
estimated to be 4.1 g. The 90th percentile user was estimated to
ingest about 8.8 g on any one day. The three-day averages are 4 g for
the mean intake and 7.5 g for the 90th pecentile consumer. The intake
of gamma-cyclodextrin from chewing gum was estimated from a separate
survey on chewing-gum intake in the United States, and the average
consumer was estimated to ingest about 0.07 g gamma-cyclodextrin per
day. These data represent a 'worst-case' scenario and are based on the
assumption that gamma-cyclodextrin is used simultaneously in all
possible applications and at the highest feasible concentrations. The
realistic average daily intake of gamma-cyclodextrin would therefore
be lower than the levels indicated above.
4. COMMENTS
The Committee noted that the close structural similarity between
gamma-cyclodextrin and ß-cyclodextrin allows some comparisons to be
made of the metabolism and the toxicity of these two compounds. The
metabolism of gamma-cyclodextrin is different from that of
ß-cyclodextrin, as demonstrated both in vitro and in vivo. In rats
in vivo, gamma-cyclodextrin (at single doses up to 1000 mg/kg bw) is
rapidly metabolized to glucose, presumably by the luminal and/or
epithelial enzymes of the gastrointestinal tract. In contrast to the
metabolism of ß-cyclodextrin, there was little involvement of the
gastrointestinal microflora. Similarly, in contrast to ß-cyclodextrin,
much less gamma-cyclodextrin was absorbed and only very low levels
could be detected in the urine. After intravenous injection,
gamma-cyclodextrin (at single doses up to 600 mg/kg bw) was rapidly
cleared from the blood and was excreted, largely unchanged, in the
urine. Although no studies of metabolism in humans in vivo were
available, gamma-cyclo-dextrin, unlike ß-cyclodextrin, can be readily
hydrolysed by human salivary and pancreatic amylases in vitro.
Short-term (28- and 90-day) studies of toxicity indicate that
gamma-cyclodextrin has little toxicity when given intravenously or
orally to rats or orally to dogs. After administration of a very high
dietary concentration (20%), caecal enlargement and associated changes
were seen in both species. This effect is likely to result from the
presence of a high concentration of an osmotically active substance in
the large intestine. This result suggests that the metabolism of
gamma-cyclodextrin is less efficient at doses higher than those used
in the studies of metabolism. The results of the studies in rats
treated intravenously indicate that gamma-cyclodextrin is well
tolerated, even when given systemically, and may be less toxic than
ß-cyclodextrin.
Studies conducted in rats and rabbits with gamma-cyclodextrin at
doses up to 20% of the diet did not indicate any teratogenic effects.
Similarly, the results of a battery of studies of genotoxicity were
negative. Long-term studies of toxicity, carcinogenicity, and
reproductive toxicity have not been conducted, but, given the rapid
metabolism of this substance to glucose and its lack of genotoxicity,
the Committee concluded that such studies were not required for an
evaluation.
In vitro, gamma-cyclodextrin, like ß-cyclodextrin, sequestered
components of the membranes of erythrocytes, causing haemolysis. The
threshold concen-tration for this effect was, however, higher than
that of ß-cyclodextrin. Furthermore, gamma-cyclodextrin was not
detected in blood after dietary administration of high doses to
animals in vivo.
It was considered unlikely that interaction of gamma-cyclodextrin
with lipophilic vitamins would impair their bioavailability, because
of the rapid digestion of gamma-cyclodextrin in vivo. Also, there
was no evidence that vitamin deficiency was induced in experimental
animals given high doses of gamma-cyclodextrin.
The Committee considered studies on the short-term toxicity and
genotoxicity of the enzyme, cyclodextrin-glycosyl transferase, used in
the production of cyclodextrins, and of the complexant,
8-cyclohexadecen-1-one, used to optimize formation of
gamma-cyclodextrin. The data indicated that these substances are
unlikely to be of toxicological concern in final preparations of
gamma-cyclodextrin that comply with the specifications. The Committee
also considered information on genetic modifications in the organism
used to produce the enzyme, which raised no concern.
Although no studies of human tolerance to gamma-cyclodextrin were
submitted to the meeting, the Committee was aware that such a study
was available. Although it was unable to review the data, the
Committee was reassured by the relatively low toxicity of this
compound in animals and the fact that it is less toxic than
ß-cyclodextrin, for which studies of human tolerance were available.
Also, its rapid metabolism to maltose, maltotriose, and glucose
in vitro by human salivary and pancreatic amylases supports the
conclusion that, as in laboratory animals, it would be metabolized to
innocuous metabolites before absorption by humans.
5. EVALUATION
The Committee concluded that there were sufficient data to
allocate a temporary ADI 'not specified' but that the study of human
tolerance known to have been conducted should be reviewed in order to
confirm the absence of adverse gastrointestinal symptoms at normal
levels of intake. These data should be submitted for consideration by
the Committee by 1999.
6. REFERENCES
Abdullah, M., French, D. & Robyt, J.F. (1966) Multiple attack by
alpha-amylases. Arch. Biochem. Biophys., 114, 595-598.
Amann, M., Douglass, J., Doyle, E. & Heimbach, J. (1998) Estimated
intake for gamma-cyclodextrin in the USA from food use. Unpublished
report from TAS-Environ, Arlington, Virginia, USA. Submitted to WHO by
Bioresco Ltd, Binningen, Switzerland.
Bär, R. & Ulitzur, S. (1994) Bacterial toxicity of cyclodextrins:
Luminuous Escherichia coli as a model. Appl. Microbiol.
Biotechnol., 41, 574-577.
de Bie, A.T.H.J. & van Ommen, B. (1994) Oral and intravenous
disposition study with [14C] gamma-cyclodextrin in rats. Unpublished
report No. V93.279 from TNO-CIVO Institutes, Zeist, Netherlands.
Submitted to WHO by Bioresco Ltd, Binningen, Switzerland.
de Bie, A.T.H.J., van Ommen, B. & Bär, A. (1998) Disposition of
14C-gamma-cyclodextrin in germfree and conventional rats. Regul.
Toxicol. Pharmacol., submitted for publication.
Blijleven, W.G.H. (1991). Examination of gamma-cyclodextrin for
mutagenic activity in the Ames test. Report No. V91.027. Unpublished
report from TNO-CIVO Institute, Zeist, Netherlands. Submitted to WHO
by Bioresco Ltd, Binningen, Switzerland.
Dickhaus, S. & Heisler, E. (1985) Acute toxicological study of
8-cyclohexadecenon after oral application on the rat. Unpublished
report from Pharmatox Beratung und Forschung GmbH, Sehnde, Germany.
Submitted to WHO by Bioresco Ltd, Binningen, Switzerland.
Ehling G., Donaubauer, H.H., Mayer D., Langer, K.H. & Schmidts, H.L.
(1992) gamma-Cyclodextrin; repeated-dose (3-month) intravenous
toxicity in rats. Unpublished report No 92.0516 from Hoechst AG,
Frankfurt a.M., Germany. Submitted to WHO by Bioresco Ltd, Biningen,
Switzerland.
French D. (1957) The Schardinger dextrins. Adv. Carbohydr. Chem.,
12, 189-260.
Frijlink, H.W., Eissens, A.C., Hefting, N.R., Poelstra, K., Lerk, C.F
& Meijer, D.K.F. (1991) The effect of parenterally administered
cyclodextrins on cholesterol levels in the rat. Pharm. Res., 8,
9-16.
Fuchs, H., Donaubauer, H.H., Mayer D., Langer K.H. & Krieg K. (1992)
gamma-Cyclodextrin; repeated-dose (1-month) intravenous toxicity study
in rats. Unpublished report No 91.1007 from Hoechst AG, Frankfurt,
Germany. Submitted to WHO by Bioresco Ltd, Biningen, Switzerland.
Immel, H.R. (1991) Examination of gamma-cyclodextrin in the
micronucleus test. Unpublished report No. V90.436 from TNO-CIVO
Institutes, Zeist, Netherlands. Submitted to WHO by Bioresco Ltd,
Binningen, Switzerland.
Irie, T., Otagiri, M., Sunada, M., Uekama, K., Ohtani, Y., Yamada, Y.
& Sugiyama, Y. (1982) Cyclodextrin-induced hemolysis and shape changes
of human erythrocytes in vitro. J. Pharm. Dyn., 5, 741-744.
Jonker, D. (1994) Subchronic (13-week) oral toxicity study with an
aqueous solution of alpha-cyclodextrin-glycosyl-transferase
(alpha-CGT) in rats. Unpublished report No. V94.332 from TNO Nutrition
and Food Research Institute, Zeist, Netherlands. Submitted to WHO by
Bioresco Ltd, Binningen, Switzerland.
Kondo, H., Nakatani, H. & Hiromi, K. (1990) In vitro action of human
and porcine alpha-amylases on cyclo-maltooligosaccharides.
Carbohydr. Res., 204, 207-213.
Leroy-Lechat, F., Wouessidjewe, D., Andreux, J.P., Puisieux, F. &
Duchne, D. (1994) Evaluation of the cytotoxicity of cyclodextrins and
hydroxypropylated derivatives. Int. J. Pharm., 101, 97-103.
Lina, B.A.R. (1992a) Subchronic (90-day) oral toxicity study,
including a recovery study, with alpha-cyclodextrin and
gamma-cyclodextrin in rats. Unpublished report No. V 90.401 from
TNO-CIVO Institutes, Zeist, Netherlands. Submitted to WHO by Bioresco
Ltd, Biningen, Switzerland.
Lina, B.A.R. (1992b) Sub-acute (28-day) oral toxicity study with
8-cyclohexadecenone in mice. Unpublished report No. V91.509 from TNO
Nutrition and Food Research Institute, Zeist, Netherlands. Submitted
to WHO by Bioresco Ltd, Binningen, Switzerland.
Lina, B.A.R. & Bär, A. (1998) Subchronic oral toxicity studies with
gamma-cyclodextrin in rats. Regul. Toxicol. Pharmacol., submitted
for publication.
Lina, B.A.R. & Jonker, D. (1989) Tentative (2-week) oral toxicity
study with gamma-CD in rats. Unpublished report No. V89.263/298033
from TNO-CIVO Institutes, Zeist, Netherlands. Submitted to WHO by
Bioresco Ltd, Binningen, Switzerland.
Lina, B.A.R., van Nesselrooij, J.H.J. & Beems, R.B. (1986) Sub-acute
(28-day) oral toxicity study with 8-cyclohexadecenone in rats.
Unpublished report No. V86.498/260392 from TNO-CIVO Institutes, Zeist,
Netherlands. Submitted to WHO by Bioresco Ltd, Binningen, Switzerland.
Marshall J.J. & Miwa I. (1981) Kinetic difference between hydrolyses
of gamma-cyclodextrin by human salivary and pancreatic alpha-amylases.
Biochem. Biophys. Acta, 661, 142-147.
Matsuda K., Mera, Y., Segawa, Y., Uchida, I., Yokomine, A. & Takagi,
K. (1983) Acute toxicity study of gamma-cyclodextrin (gamma-CD) in
mice and rats. Pharmacometrics, 26, 287-291.
Matsuda, K., Segawa, Y., Yokomine, A., Uchida, I., Mera, Y. &
Tokugawa, H. (1985) Biodisposition of gamma-cyclodextrin (gamma-CD) in
the rabbit and dog after intravenous administration. Oyo Yakuri
(Pharmacometrics), 30, 889-896.
Ohtani, Y., Irie, T., Uekama K., Fukunaga, K. & Pitha, J. (1989)
Differential effects of alpha-, ß-, and gamma-cyclodextrins on human
erythrocytes. Eur. J. Biochem., 186, 17-22.
Okada, Y., Kubota, Y., Koizumi, K., Hizukuri, S., Ohfuji, T. & Ogata,
K. (1988) Some properties and the inclusion behavior of branched
cyclodextrins. Chem. Pharm. Bull., 36, 2176-2185.
Prinsen, M.K. (1990) Acute eye irritation/corrosion study with
gamma-cyclodextrin in albino rabbits. Unpublished report No.
V90.405/200069 from TNO-CIVO Institutes, Zeist, Netherlands. Submitted
to WHO by Bioresco Ltd, Binningen, Switzerland.
Prinsen, M.K. (1992) Sensitization study with gamma-CD in guinea pigs
(maximization test). Unpublished report No. V92.575/352063 from
TNO-CIVO Institutes, Zeist, Netherlands. Submitted to WHO by Bioresco
Ltd, Binningen, Switzerland.
Riebeek, W.M. (1990a) Determination of the acute intravenous toxicity
of gamma-cyclodextrin in mice. Unpublished report No. V90.222 from
TNO-CIVO Institutes, Zeist, Netherlands. Submitted to WHO by Bioresco
Ltd, Binningen, Switzerland.
Riebeek, W.M. (1990b) Determination of the acute intravenous toxicity
of gamma-cyclodextrin in rats. Unpublished report No. V90.220 from
TNO-CIVO Institutes, Zeist, Netherlands. Submitted to WHO by Bioresco
Ltd, Binningen, Switzerland.
Riebeek, W.M. (1990c) Determination of the acute intraperitoneal
toxicity of alpha-cyclodextrin and gamma-cyclodextrin in rats.
Unpublished report No. V90.233 from TNO-CIVO Institutes, Zeist,
Netherlands. Submitted to WHO by Bioresco Ltd, Binningen, Switzerland.
Spanjers, M.T. (1986) Determination of the acute oral toxicity (limit
test) of 8-cyclohexadecenone in mice. Unpublished report No.
V86.455/260393 from TNO-CIVO Institutes, Zeist, Netherlands. Submitted
to WHO by Bioresco Ltd, Binningen, Switzerland.
Szejtli, J., Csherhti, T. & Szgyi, M. (1986) Interactions between
cyclodextrins and cell-membrane phospholipids. Carbohydr. Polymers,
6, 35-49.
Til, H.P. & Bär, A. (1998) Subchronic (13-week) oral toxicity study of
gamma-cyclodextrin in dogs. Regul. Toxicol. Pharmacol., submitted
for publication.
Til, H.P. & van Nesselrooij (1992) Subchronic (90-day) feeding study
with gamma-CD in dogs. Unpublished report No. V91.475 from TNO-CIVO
Institutes, Zeist, Netherlands. Submitted to WHO by Bioresco Ltd,
Binningen, Switzerland.
Verhagen, F.J.J. & Waalkens-Berendsen, D.H. (1991) Oral
embryotoxicity/teratogenicity study with alpha-cyclodextrin and
gamma-cyclodextrin in rats. Unpublished report No. V91.210 from TNO
Nutrition and Food Research Institute, Zeist, Netherlands. Submitted
to WHO by Bioresco Ltd, Binningen, Switzerland.
de Vogel, N. & van Delft, J.H.M. (1996) Chromosome aberration test
with gamma-cyclodextrin in cultured human lymphocytes. Unpublished
report No. V96.398 from TNO-CIVO Institutes, Zeist, Netherlands.
Submitted to WHO by Bioresco Ltd, Binningen, Switzerland.
Waalkens-Berendsen, D.H. & Smits-van Prooije, A.E. (1992) Final report
of an oral embryotoxicity/teratogenicity study with alpha-cyclodextrin
and gamma-cyclodextrin in New Zealand white rabbits. Unpublished
report No. V91.564 from TNO Nutrition and Food Research Institute,
Zeist, Netherlands. Submitted to WHO by Bioresco Ltd, Binningen,
Switzerland.
Willems, M.I. (1986) Evaluation of 8-cyclohexadecenone in the
micronucleus test. Unpublished report No. V86.456/260393 from TNO-CIVO
Institutes, Zeist, Netherlands. Submitted to WHO by Bioresco Ltd,
Binningen, Switzerland.
Wilmer, J.W.G.M. (1986) Examination of 8-cyclohexadecenone for
mutagenic activity in the Ames test. Unpublished report No.
V86.264/260064 from TNO-CIVO Institutes, Zeist, Netherlands. Submitted
to WHO by Bioresco Ltd, Binningen, Switzerland.
Yim, D.G., Sato, H.H., Park, Y.H. & Park, Y.K. (1997) Production of
cyclodextrin from starch by cyclodextrin glycosyltransferase from
Bacillus firmus and characterization of purified enzyme. J. Ind.
Microbiol. Biotechnol., 18, 402-405.
Yoshida, A., Arima, H., Uekama, K. & Pitha, J. (1988) Pharmaceutical
evaluation of hydroxyalkyl ethers of ß-cyclodextrins. Int. J.
Pharmacol., 46, 217-222.
 gamma-Cyclodextrin has not been reviewed previously by the
Committee; however, the structurally related ß-cyclodextrin (seven
glucose units) was evaluated at the forty-first and forty-fourth
meetings of the Committee (Annex 1, references 107 and 116).
2. BIOLOGICAL DATA
2.1 Biochemical aspects
2.1.1 Absorption, distribution, biotransformation, and excretion
Early experiments provided some evidence that gamma-cyclodextrin
(unlike alpha- or ß-cyclodextrin) is hydrolysed by salivary amylase
(French, 1957). More recent studies have confirmed that both human
salivary amylase and human or porcine pancreatic amylase can readily
hydrolyse gamma-cyclodextrin, yielding mainly maltose, some
maltotriose, and smaller amounts of glucose (Abdullah et al., 1966;
Marshall & Miwa, 1981; Kondo et al., 1990).
The fate of intravenously administered gamma-cyclodextrin was
examined in three male and two female rabbits and one male dog. The
rabbits were each given a dose of 7.5 g in saline (30 ml of a 25% w/v
solution), while the dog was given a dose of 25 g in saline (500 ml of
a 5% w/v solution at 8.3 ml/min). Blood and urine samples were
collected and examined by high-performance liquid chromatography
(HPLC) for gamma-cyclodextrin over 4 h. The half-life of
gamma-cyclo-dextrin in blood was about 50 min in rabbits and 30 min in
the dog. In rabbits, about 77% of the injected dose was recovered as
unchanged gamma-cyclodextrin in the urine within 24 h, while in the
dog, nearly 80% of the injected dose was recovered as unchanged
gamma-cyclodextrin in the urine within the first 4 h (Matsuda et al.,
1985).
Two male Wistar rats were given 14C-gamma-cyclodextrin (94%
radiochemical purity) as single oral or intravenous doses. After an
oral dose of 14C-gamma-cyclo-dextrin (25 µCi/kg bw diluted to 200
mg/kg bw), 50% of the radiolabel was excreted in the exhaled air
within 24 h, while 2% was excreted in the urine and 5% in the faeces;
35% of the radiolabel was retained in the body after 24 h. No
detectable gamma-cyclodextrin was found in blood during 8 h after
administration, suggesting metabolism in the gastrointestinal tract.
After the intravenous dose of 14C-gamma-cyclodextrin (25 µCi/kg
bw diluted to 100 mg/kg bw), 90% of the radiolabel was excreted in the
urine within 8 h, mainly as unchanged gamma-cyclodextrin. After 8 h,
small amounts of radiolabel were excreted in exhaled air (1.1%), while
0.5% was detected in the faeces; 6% was retained in the body after 24
h (de Bie & van Ommen, 1994).
Groups of four male Wistar rats were given single oral or
intravenous doses of 14C-gamma-cyclodextrin. The oral dose was 1000
mg/kg bw, and the concentration of radiolabel was either 25 or 100
µCi/kg bw. In order to examine the role of the gut microflora in the
metabolism of gamma-cyclodextrin, a separate group of germ-free male
rats was given 25 µCi/kg bw gamma-cyclodextrin diluted to 1000 mg/kg
bw. The intravenous dose was 600 mg/kg bw, and the concentration of
radiolabel was 25 µCi/kg bw.
In the first experiment, the time-course and routes of metabolism
were examined after oral administration. After administration of
1000 mg/kg bw (25 µCi/kg bw) 14C-gamma-cyclodextrin, 62% of the
radiolabel in males and 71% in females was excreted within 48 h. The
majority was excreted in expired air (57% in males and 65% in
females), with maximum excretion between 1 and 2 h after dosing.
Little was excreted in the urine (2.1% for males and 2.5% for females)
or faeces (2.9% for males and females) during the 48-h sampling
period. Retention of radiolabel after 48 h was 27% in males and 22% in
females, with the most significant retention in the liver (11% in
males, 5.2% in females). The amounts in all other organs were low:
intestine, 0.42% for males, 0.36% for females; kidney, 0.13% for
males.
In the second experiment, the excretion kinetics and time-course
in blood over 24 h were examined after a single oral dose of
1000 mg/kg bw (100 Ci/kg bw) 14C-gamma-cyclodextrin. The major route
of excretion was exhaled air (55% for males; 53% for females) during
the 24-h sampling period. The carcass retained 35% of the radioactive
dose in males and 25% in females at 24 h after treatment. The maximum
concentration in blood was reached 40 min after treatment, but this
concentration decreased by 50% after 0.5 h in males and after 6 h in
females. The urine of animals of each sex contained a peak that eluted
before glucose at all times and a small peak that co-eluted with
glucose (6% of total radiolabel). A small amount of unchanged
gamma-cyclo-dextrin was detected in urine after 4 h (< 0.02% of the
ingested dose). HPLC analysis of the stomach contents 24 h after
dosing revealed the presence of gamma-cyclodextrin and a minor peak
that co-eluted with glucose. HPLC analysis of the blood revealed the
presence of a radioactive peak that co-eluted with glucose.
In the third experiment, the excretion kinetics and time-course
in blood over 24 h were examined after a single intravenous dose of
600 mg/kg bw (25 µCi/kg bw) 14C-gamma-cyclodextrin. The major route
of excretion was the urine (87% for males, 91% for females), and the
faeces contained about 1% and the gastrointestinal tract about 0.5% of
the radiolabel; the exhaled air contained 7.8% of the dose for males
and 3.2% for females. The carcass contained 3.9% of the dose in males
and 2.2% in females, the liver having the most residue (0.41% in
males, 0.42% in females). Radiolabel was removed rapidly from the
blood, with calculated initial half-lives of 20 min in males and 15
min in females. HPLC analysis of blood revealed predominantly
gamma-cyclodextrin.
In the fourth experiment, excretion kinetics over 24 h was
examined in germ-free rats given a single oral dose of 1000 mg/kg bw
(25 µCi/kg bw) gamma-cyclodextrin. About 73% of the radiolabel was
excreted in expired air (68% in males, 65% in females). Peak
exhalation of 14C-carbon dioxide occurred at about 90 min. Low
concentrations of radiolabel were found in faeces and urine. The
gastrointestinal tract contained 9.2% in males and 11% in females,
with the majority in the caecum. The results indicate that
gastrointestinal microflora play little part in the metabolism of
gamma-cyclodextrin.
Overall, this study indicates that gamma-cyclodextrin is
metabolized to glucose, other low-molecular-mass sugar metabolites,
and carbon dioxide by luminal and/or epithelial enzymes of the
gastrointestinal tract (de Bie & van Ommen, 1994; de Bie et al.,
1998).
2.2 Toxicological studies
2.2.1 Acute toxicity
The acute toxicity of gamma-cyclodextrin was examined in mice and
rats; the results are summarized in Table 1. In each study, groups of
10 animals of each sex were tested at three doses or served as
controls. There were no deaths at the highest doses tested (Matsuda et
al., 1983).
Table 1. Acute toxicity of gamma-cyclodextrin
Species Sex Route LD50 Reference
(mg/kg bw)
Mouse Male and female Oral >16 000 Matsuda et al. (1983)
Mouse Male and female Subcutaneous > 4 000 Matsuda et al. (1983)
Mouse Male and female Intravenous > 4 000 Matsuda et al. (1983)
Mouse Male and female Intravenous 10 000 Riebeek (1990a)
Rat Male and female Oral > 8 000 Matsuda et al. (1983)
Rat Male and female Subcutaneous > 2 400 Matsuda et al. (1983)
Rat Male and female Intravenous > 3 750 Matsuda et al. (1983)
Rat Male and female Intravenous > 3 750 Riebeek (1990b)
Rat Male Intraperitoneal > 4 600 Riebeek (1990c)
In a test for micronucleus formation (see section 2.2.3), groups
of 15 Swiss mice of each sex were given a single dose of 15 g/kg bw
gamma-cyclodextrin by gavage and were killed in groups of five at 24,
48, and 72 h. All of the animals survived until termination (Immel,
1991).
The toxicity of intravenously administered gamma-cyclodextrin was
examined in groups of five male and five female Crl:CD mice and five
male and five female Crl:W1(WU)BR rats. The mice were given
gamma-cyclodextrin (purity, > 98%) at doses of 5000, 7500, or 10 000
mg/kg bw, while the rats were given doses of 2500, 3750, or 5000 mg/kg
bw as a solution in sterile saline. The animals were observed for
clinical signs over 14 days after treatment. They were examined for
gross pathological changes on day 14 after treatment.
Both mice and rats showed dose-related signs of toxicity, such as
piloerection and sluggishness, within 1 h to a few days after
treatment. Deaths occurred within a few days in both species, but the
surviving animals recovered and appeared to be healthy at the end of
the observation period. Macroscopic examination revealed no gross
treatment-related alterations (Riebeek, 1990a,b).
The toxicity of intraperitoneally administered gamma-cyclodextrin
was examined in groups of three male rats after single doses of 2000
or 4600 mg/kg bw as a solution in sterile saline. The animals were
observed for clinical signs during 14 days after treatment and were
examined grossly for pathological changes on the last day. There were
no signs of toxicity and no deaths. Macroscopic examination revealed
no gross treatment-related alterations (Riebeek, 1990c).
2.2.2 Short-term studies of toxicity
Rats
Groups of 15 Wistar rats of each sex were given
gamma-cyclodextrin intravenously as single daily doses of 0, 200, 630,
or 2000 mg/kg bw for 30 days. A group of five animals of each sex were
allowed to recover for 27 days after treatment. The animals were
examined throughout the study, and the body weights recorded weekly.
Clinical chemical parameters were examined at the end of treatment and
after the recovery period. Ten animals from each group were killed at
the end of treatment and the remainder at the end of the recovery
period.
There were no clinical signs of toxicity throughout the study.
The body-weight gain of males at 630 and 2000 mg/kg bw per day was
slightly decreased, but the difference was not significant. Food
consumption was decreased in a dose-related manner only during the
first few days of treatment. Significantly decreased mean values for
haemoglobin concentration and haemacrit were seen in males at doses
> 630 mg/kg bw per day, and significantly decreased erythrocyte
count in males at 2000 mg/kg bw per day. In females, the erythrocyte
count, haemoglobin concentration, and haematocrit were significantly
decreased only at 2000 mg/kg bw per day. The reticulocyte counts were
significantly increased in both males and females at 2000 mg/kg bw per
day. These changes were reversed after the recovery period. The only
significant clinical chemical change was an increase in creatinine and
urea in both males and females at 2000 mg/kg bw per day, which was
reversed at the end of the recovery period. Urinalysis revealed a
significantly increased incidence of haemoglobin in the urine of rats
at 2000 mg/kg bw per day, which was reversed at the end of the
recovery period.
Pathological examination at autopsy showed that the majority of
animals at 2000 mg/kg bw per day had light-coloured and, in some
cases, irregularly coloured kidneys. A dose-dependent increase in
spleen weight was found in all groups, which was significant in males
at doses > 630 mg/kg bw per day and in females at all doses. Liver
weights were significantly increased in females at > 630 mg/kg bw
per day, and kidney weights were significantly increased at 2000 mg/kg
bw per day. Lung and adrenal weights were significantly increased in
both males and females at 2000 mg/kg bw per day. These changes were
partially reversed after the recovery period, but a significant
increase in kidney and spleen weights was still present in females at
2000 mg/kg bw per day. Histopathological examination revealed marked
vacuolation of renal epithelial cells in the proximate convoluted
tubules, which may have been related to administration of a substance
that causes hyperosmolarity. There was also extensive pulmonary
histiocytosis (massive accumulation of alveolar macrophages) in all
animals at 2000 mg/kg bw per day. These changes were seen only in some
animals at 630 mg/kg bw per day and at lesser severity. Considerable
reversibility was observed after the recovery period. The NOEL was 200
mg/kg bw per day (Fuchs et al., 1992).
Groups of 15 Wistar rats of each sex were given
gamma-cyclodextrin intravenously as single daily doses of 0, 60, 120,
or 600 mg/kg bw for 90 days. Groups of five animals of each sex were
allowed to recover at the end of the treatment period. The animals
were examined throughout the study, and body weights were recorded
weekly. Haematological and urinary parameters were examined after one
month, at the end of the study, and on day 37 (males) or 36 (females)
of recovery. Clinical chemical parameters were examined at the end of
the treatment period and after recovery. Animals were killed after
90 days, their organs were examined, and tissues were taken for
histopathological examination.
There were no clinical signs of toxicity. Body-weight gain was
slightly decreased only in males at the high dose, and food
consumption was decreased in a dose-related manner only during the
first few days of treatment. Haematological examination revealed a
slight decrease in the mean erythrocyte count, haemaglobin
concentration, and haemacrit in females at one month, and these values
were significantly decreased at three months only in females at the
high dose. The reticulocyte counts were significantly increased in
females at the high dose at three months. At the end of the recovery
period, all of these values were normal. The only clinical chemical
changes were a slight decrease in bilirubin concentration in males and
females at the high dose and a slight increase in alkaline phosphatase
activity in males at the high dose. At the end of the recovery period,
all of the values were normal. The slight changes observed in urinary
parameters were considered not to be treatment-related.
Histological examination at autopsy revealed an increased
incidence of enlarged iliac lymph nodes in males and females at the
high dose, which was probably related to inflammation at the injection
site. Increases in the relative weights of lungs, liver, and spleen
were seen in males and in the relative weights of heart, lungs,
kidneys, spleen, and adrenal glands in females at the high dose. All
of these changes were reversed after the four-week recovery period.
Histopathological examination revealed hyperplasia of the mucosa of
the urinary bladder, which was reversed after four weeks, and
pulmonary histiocytosis (macrophage aggregates), which tended to
reverse after four weeks, in males and females at the high dose. The
reversibility of the urinary bladder changes indicates that the
hyperplasia was not preneoplastic. The NOEL was 120 mg/kg bw per day
(Ehling et al., 1992).
Groups of five male Wistar rats were fed diets containing
gamma-cyclo-dextrin at concentrations of 0, 5, 10, 15, or 20%,
equivalent to 0, 2500, 5000, 7500, or 10 000 mg/kg bw per day, for 14
days. A control group was fed 20% lactose. The animals were examined
for clinical signs of toxicity, and food and water consumption was
monitored throughout the study. On day 14, the animals were killed,
and blood samples were taken for examination of clinical chemical
parameters. The animals were also examined macroscopically for
pathological changes.
There were no deaths. The incidence of soft stools was slightly
higher in treated than control animals, and the mean body weights of
treated animals were slightly lower than those of controls, although
this difference was not statistically significant. Food intake was
slightly lower in controls than in treated groups. Plasma alkaline
phosphatase activity was increased in animals at 15 and 20%, while
gamma-glutamyl transferase activity was decreased in all treated
groups. The activity of aspartame and alanine aminotransferases were
unchanged, as were the concentrations of total protein, albumin, urea,
and bilirubin. Enlargement of the caecum was seen at all doses, the
most significant effect occurring in the group fed 20% lactose. There
were no macroscopic changes attributable to treatment (Lina & Jonker,
1989; Lina & Bär, 1998).
Groups of 20 Wistar rats of each sex were fed diets containing
gamma-cyclo-dextrin at concentrations of 0, 1.5, 5, or 20% (equivalent
to 0, 750, 2500, or 10 000 mg/kg bw per day) for 13 weeks. A control
group was fed 20% lactose. In order to examine the reversibility of
any effects seen, a group of 10 animals of each sex were fed either
20% gamma-cyclodextrin or 20% lactose for 13 weeks and then control
diet for one month. As a result of a feeding error, the groups
receiving 20% gamma-cyclodextrin were discarded, and two additional
groups were fed 0 or 20% gamma-cyclodextrin for 13 weeks and 10
animals receiving the 20% dose were fed control diet for an additional
month at the end of the 13-week period. Animals were examined for
clinical signs of toxicity, and food and water consumption was
monitored throughout the study. Ophthalmoscopic examinations were
conducted in animals at the high dose and those given lactose before
treatment and at the end of the study. The animals were killed at the
end of the study, and blood samples were taken for clinical chemistry.
The animals were also examined macroscopically for pathological
changes.
There were no treatment-related deaths during the study. Soft
stools were observed in some rats at 5 or 20% gamma-cyclodextrin and
in those given lactose early in the study, but this effect disappeared
subsequently. Ophthalmoscopic examination revealed no
treatment-related effects. The body weights of males at 20%
gamma-cyclodextrin were slightly but significantly decreased
throughout treatment; a more significant body-weight decrease was seen
in the males given 20% lactose. No difference was noted in these
groups during the recovery period. Food conversion efficiency was also
slightly decreased in males at 20% lactose. None of the changes in
haematological parameters was considered to be of toxicological
significance. Some changes in clinical chemistry were seen in females
at the high dose, namely increased activity of aspartate and alanine
aminotransferases, especially in one female. The sodium concentrations
were slightly higher but within the historical control range. Urinary
parameters were similar in control and treated groups, apart from a
significant increase in the urinary calcium concentration at the end
of treatment in males given 20% gamma-cyclodextrin and rats of each
sex given 20% lactose; the effect was more pronounced in the latter
group. The calcium:creatinine ratio was significantly increased in
rats receiving 20% lactose. The increased calcium appeared to be
associated with the increased load of osmotically active substances in
the large intestine; there was no increase in calcium at the end of
the recovery period.
Analysis of organ weights revealed significant increases in both
absolute and relative caecal weights in rats at 5 and 20%
gamma-cyclodextrin and 20% lactose in comparison with controls. At the
end of the recovery period, no difference from controls was seen for
rats given 20% gamma-cyclodextrin, but the increase was still evident
in those given 20% lactose. This effect is probably related to the
retention of an osmotically active substance in the large intestine.
Relative adrenal weights were increased in males given 20%
gamma-cyclodextrin and the lactose control group at the end of the
study. A very slight increase in relative liver weight was seen in
males at 20% gamma-cyclodextrin and females given lactose. No changes
in organ weight were seen at the end of the recovery period.
There were no gross pathological changes attributable to
treatment with gamma-cyclodextrin. Microscopic examination revealed
increased cortico-medullary mineralization in the kidneys of rats at
1.5 and 5% gamma-cyclodextrin but not at the high dose. This change is
considered to be relatively common in rats and not related to
treatment with gamma-cyclodextrin. There was no evidence of
hyperplasia of the mucosa of the urinary bladder or of pulmonary
histiocytosis, which were observed after intravenous administration.
The effects observed in rats after receiving gamma-cyclodextrin
in the diet at concentrations up to 20% appeared to be largely related
to the presence of high levels of an osmotically active substance in
the large intestine and not of toxicological significance. Similar,
and generally more pronounced, effects were observed when the diet
contained 20% lactose. On the basis of this study, dietary levels of
20% gamma-cyclodextrin (equivalent to10 000 mg/kg bw per day) were
considered to be tolerated without toxicological effects (Lina, 1992a;
Lina & Bär, 1998).
Dogs
Groups of four male and four female beagle dogs were fed diets
containing gamma-cyclodextrin at doses of 0, 5, 10, or 20% (equivalent
to 0, 1250, 2500, or 5000 mg/kg bw per day). Body weight and food
consumption were recorded weekly throughout the study. Ophthalmoscopic
examinations were made at the beginning and end of the study. Blood
was collected for clinical chemical and haematological examination
before the start of treatment and at weeks 6 and 13. Urinalyses were
performed at week 13. During week 14, all animals were killed, their
organs were examined, and tissues were prepared for histopathological
examination.
There were no deaths during the study and no clinical signs of
toxicity. Diarrhoea was noted in all groups, increasing in incidence
and severity with increasing doses of gamma-cyclodextrin, and was more
severe in females than in males. This effect was attributed to the
slow digestion and intestinal absorption of gamma-cyclodextrin.
Ophthalmoscopic examination revealed no differences between treated
and control animals. Weight gain was comparable in all groups, except
for a slight, non-significant decrease in males at 20% during the last
six weeks of the study. Treated dogs had slightly decreased food
intake during the first two weeks of the study, particularly at the
high dose. This effect persisted throughout the study only in dogs at
the high dose. No differences in haematological parameters were seen,
and clinical chemical parameters were similar in treated and control
groups. Urinalysis revealed a slight decrease in urinary pH in the
group receiving 20% gamma-cyclodextrin, which was considered to be
associated with the intake of poorly digestible carbohydrates and
increased fermentation in the large intestine.
Both the absolute and relative weights of the ovary were
increased in animals at 10 and 20% gamma-cyclodextrin, but no
histopathological changes were seen, and the weight of the ovaries in
the control group was lower than that of other dogs in the same
laboratory. The changes observed are considered unlikely to be
toxicologically significant. There was no change in absolute liver
weight, although the relative liver weight was increased in males at
the high dose as a result of the slight decrease in body weight at
this dose. This change is likely to be adaptive and was considered not
to be toxicologically significant. Caecal weights were increased in
the dogs at 10 and 20% gamma-cyclodextrin but statistically
significantly only in females. This change is considered to be related
to the presence of high levels of an osmotically active substance in
the large intestine. Pathological examination at the end of the study
revealed no treatment-related change. Similarly, the histopathological
findings were unremarkable. On the basis of this study, dietary levels
of 20% gamma-cyclodextrin (equivalent to 5000 mg/kg bw per day) were
considered to be tolerated without toxicological effects (Til & van
Nesselrooij, 1992; Til & Bär, 1998).
2.2.3 Genotoxicity
The results of assays for genotoxicity with gamma-cyclodextrin
are shown in Table 2.
2.2.4 Developmental toxicity
Rats
In a study of embryotoxicity and teratogenicity, groups of 25
presumed pregnant Wistar Crl:(WI)WU BR rats were fed diets containing
gamma-cyclodextrin (purity, >98%) at concentrations of 0. 1.5, 5, 10,
or 20% (equivalent to 0, 2500, 5000, or 10 000 mg/kg bw per day) on
days 0-21 of gestation. A separate group received a diet containing
20% lactose instead of pre-gelatinized potato starch. The animals were
examined throughout the study, and body weight and food consumption
were recorded. The rats were killed on day 21 and examined for
parameters of reproductive performance. Fetuses were examined for
signs of toxicity, external malformations, and soft-tissue defects and
were stained for detection of skeletal anomalies.
No deaths occurred during the study. Maternal body-weight gain
was similar in all groups except for a slight reduction at the 20%
dose on days 0-16 of gestation. Necropsy of the dams showed no adverse
effects that could be related to treatment. The number of viable
litters, the number of corpora lutea, and the mean number of
implantation sites were similar in all groups. Fetal length and body
weight were also similar in all groups. Examination of the fetuses
revealed no treatment-related increase in gross, skeletal, or visceral
abnormalities. Under the conditions of this assay, gamma-cyclodextrin
was not teratogenic (Verhagen & Waalkens-Berendsen, 1991).
Rabbits
In a study of embryotoxicity and teratogenicity, groups of 16
presumed pregnant New Zealand white rabbits were fed diets containing
gamma-cyclodextrin (purity, > 98%) at concentrations of 0, 5, 10, or
20% (equivalent to 0, 1500, 3000, or 6000 mg/kg bw per day) on days
0-29 of gestation. The animals were examined throughout the study, and
body weights and food consumption were recorded regularly. The animals
were killed on day 29 of pregnancy, and the fetuses were examined for
signs of toxicity, external malformations, and soft-tissue defects and
were stained for detection of skeletal anomalies.
Table 2. Results of assays for the genotoxicity of gamma-cyclodextrin
End-point Test object Concentration Result Reference
Bacterial gene S. typhimurium TA1535, 2-20000 Negativea Blijleven (1991)
mutation TA1537, TA 1538, µg/plate
TA98, TA100
Chromosomal Human lymphocytes 10-5000 Negativea de Vogel & van
aberrationb µg/ml Delft (1996)
Micronucleus CD-1 mouse bone 15 g/kg bw Negative Immel (1991)
formationb marrow (single dose)
a With and without exogenous metabolic activation
b Two assays conducted: treatment times, 6, 24, and 48 h; harvest times, 24 and 48 h
No deaths occurred during the study. Diarrhoea was seen in
several treated animals. Maternal body weight was significantly
decreased at the 20% dose during the first week of treatment but not
thereafter. Necropsy of the does showed no adverse effects that could
be related to treatment. The number of viable litters, the number of
corpora lutea, and the mean number of implantation sites were similar
in all groups. One abortion occurred in the control group and one in
rabbits at the 10% dose. Fetal length and body weight were similar in
all groups. Examination of the fetuses revealed no treatment-related
increase in gross, skeletal, or visceral abnormalities. An increase in
the incidence of haemorrhagic fluid in animals at the 5 and 20% doses
was considered unlikely to be treatment-related. Under the conditions
of this assay, gamma-cyclodextrin was not teratogenic
(Waalkens-Berendsen & Smits-van Prooije, 1992).
2.2.5 Special studies
2.2.5.1 Ocular irritation and dermal sensitization
gamma-Cyclodextrin was not irritating or corrosive to the eyes of
albino rabbits (Prinsen, 1990). In a skin sensitization assay in
guinea-pigs, a 30% solution of gamma-cyclodextrin induced no signs of
sensitization (Prinsen, 1992).
2.2.5.2 Cell membrane effects
The interactions between alpha-, beta-, and gamma-cyclodextrin
and membrane phospholipids, liposomes, and human erythrocytes were
studied in vitro. gamma-Cyclodextrin and the other cyclodextrins did
not increase the permeability of dipalmityol-phosphatidylcholine
liposomes, nor did they affect the active transport of 42K+ into
erythrocytes at a concentration of 10-2 mol/L. ß-Cyclo-dextrin but
not gamma-cyclodextrin or alpha-cyclodextrin increased the release of
42K+, 86Rb+, and 137Cs+ from erythocytes by passive transport at
a concentration of 1.7 × 10-2 mol/L (Szejtli et al., 1986).
gamma-Cyclodextrin induced haemolysis of human erythrocytes
in vitro in isotonic saline after 30 min, with a threshold
concentration of 16 mmol/L (approximately 20 mg/L). A similar effect
was seen with alpha-cyclodextrin at 6 mmol/L and with ß-cyclodextrin
at 3.5 mmol/L. At 15 mmol/L, gamma-cylodextrin caused a 2% release of
cholesterol from erythrocytes, indicating that cyclodextrin-induced
haemolysis is associated with removal of membrane components from
erythrocytes (Irie et al., 1982). Other studies have confirmed that
gamma-cyclodextrin causes erythrocyte haemolysis at concentrations of
15-30 mmol/L (Okada et al., 1988; Yoshida et al., 1988; Leroy-Lechat
et al., 1994).
In a study on the differential effects of cyclodextrins on human
erythrocytes in vitro, alpha-, ß-, and gamma-cyclodextrin were
incubated at increasing concentrations with erythrocytes for 30 min.
The cyclodextrins induced a change in shape, from discocyte to
spherocyte, but with ß-cyclodextrin haemolysis occurred before the
change was complete. gamma-Cyclodextrin was less potent than either
alpha- or ß-cyclodextrin in releasing potassium and haemoglobin from
erythrocytes and in solubilizing various components of erythrocytes,
including phospholipids, cholesterol, and proteins. A concentration of
4 mmol/L gamma-cyclodextrin was required to induce release of
phospholipid, and > 20 mmol was required for release of cholesterol
or protein (Ohtani et al., 1989).
The potential cytotoxicity of the cyclodextrins to P388 murine
leukaemia cells was examined by exposing the cells to increasing
concentrations of cyclodextrins for 48 h in medium containing 10%
fetal calf serum. Initial cytotoxicity was elicited at 11 mmol/L
alpha-cyclodextrin, 2.5 mmol/L ß-cyclodextrin, and 55 mmol/L
gamma-cyclodextrin (Leroy-Lechat et al., 1994).
Potential reductions in the luminescence of a strain of E. coli
after incubation with various concentrations of cyclodextrins were
examined. Luminescence was reduced by 20 and 50% by ß-cyclodextrin at
concentrations of 0.03 and 0.5%, respectively. With
gamma-cyclodextrin, a concentration of 2% was required to reduce the
luminescence by 20%, and a 50% reduction could not be achieved even at
the highest concentration of 10% gamma-cyclodextrin (Bär & Ulitzur,
1994).
2.2.5.3 Impurities
Cyclodextrin-glycosyl transferase
The gene coding for cyclodextrin-glycosyl transferase is derived
from a strain of the Bacillus firmus-lintus complex. This is a
ubiquitous group of aerobic, gram-positive, alkalophilic
microorganisms which form sub-terminal endospores. Both B. firmus
and B. lintus are considered to be non-pathogenic. After isolation
from this strain, the gene was cloned into pUC18, isolated again and
inserted into an expression and secretion vector derived from pJF118EH
by introduction of a DNA fragment coding for an appropriate signal
peptide. The vector, pJF118EH, is derived from pBR322, a widely used
vector which is considered to be safe. All genetic modifications were
made by directed mutagenesis by standard techniques. The gene that has
been introduced for production of the signal peptide is cleaved off
during exportation of cyclodextrin-glycosyl transferase and degraged
with the E. coli cell.
A strain derived from E. coli K12 was used as the host for the
cyclodextrin-glycosyl transferase expression and secretion vector.
E. coli K12 is considered to be a non-pathogenic strain and is used
for production of enzymes for food use.
For the production of cyclodextrin-glycosyl transferase, the
recombinant E. coli strain was cultured in a standard medium. The pH
of the fermentation broth was adjusted by adding food-grade ammonia or
phosphoric acid. The E. coli was grown to a certain density, after
which enzyme production was initiated by addition of isopropyl
thiogalactoside. The bacteria were then removed by fermentation and
the supernatant filtered and concentrated to the crude
cyclodextrin-glycosyl transferase preparation.
Groups of 20 Wistar rats of each sex were given an aqueous
solution of a cyclodextrin-glycosyl transferase preparation at doses
of 0, 5, 10, or 20 ml/kg bw per day, equivalent to 0, 65, 130, or 260
mg/kg bw per day of total organic solids (TOS), by gavage for 13
weeks. The animals were examined for clinical signs of toxicity, and
food and water consumption was monitored throughout the study.
Ophthalmoscopic examinations were conducted in rats at the high dose
before treatment and at the end of the study. Haematological
parameters were examined before treatment, on days 30 and 60, and at
the end of the study. The animals were killed at the end of the study,
and blood samples were taken for examination of clinical chemical
parameters. The animals were also examined macroscopically for
pathological changes, and samples were taken for histopathological
examination.
Two animals (one control male and one female at the low dose)
died as a result of dosing accidents. There were no clinical signs of
toxicity in the treated animals which survived. Ophthalmoscopic
examination of animals at the high dose revealed no treatment-related
changes. There was no significant difference in body weight between
treated and control rats, and food consumption was unchanged in the
treated group. There were no treatment-related haematological,
clinical chemical, or urinary changes. The organ weights of control
and treated groups were similar, and there were no treatment-related
gross pathological changes. Histopathological examination revealed an
increased incidence of pulmonary changes in the treated animals which
were considered to be related to aspiration of test material. The NOEL
was 20 ml/kg bw per day, equivalent to 260 mg/kg bw per day of TOS,
the highest dose tested (Jonker, 1994).
The cyclodextrin-glycosyl transferase preparation was tested for
mutagenic activity in S. typhimurium strains TA1535, TA1537, TA98,
and TA100 with and without metabolic activation at concentrations of
19-1500 µg/plate or 62-500 µg/plate. The assay was slightly
complicated by the growth-stimulating effects of some of the
components of the crude preparation, but the overall result confirmed
that cyclodextrin-glycosyl transferase had no mutagenic activity in
these strains.
8-Cyclohexadecen-1-one
8-Cyclohexadecen-1-one is a colourless, waxy, solid product. The
commercial preparation has a purity of > 98%. The acute toxicity of
8-cyclo-hexadecen-1-one was examined in five male and five female mice
at a dose of 5 ml/kg bw (4.6 g/kg bw) and in five male and five female
rats at doses of 5 and 10 g/kg bw. The animals were observed for 14
days. Two of the five mice died during the study, but there were no
deaths among the rats. It was concluded that the LD50 was > 4.6 g/kg
bw in mice and > 10 g/kg bw in rats (Dickhaus & Heisler, 1985;
Spanjers, 1986).
Groups of five CD-1 mice of each sex were fed diets containing
8-cyclo-hexadecen-1-one at doses of 0, 500, 1500, or 7500 mg/kg diet
(equivalent to 0, 75, 230, or 1100 mg/kg bw per day) for four weeks.
The animals were examined for clinical signs of toxicity, and food and
water consumption were monitored throughout the study. Haematological
and clinical chemical parameters were examined before treatment and at
the end of the study. The animals were also examined macroscopically
for pathological changes, and samples were taken for histopathological
examination. There were no deaths and no clinical signs of toxicity.
The body-weight gain of mice at the high dose was decreased, but food
and water intake were similar in treated and control groups. There
were no treatment-related haematological changes. The only clinical
chemical change was a decrease in the albumin:globulin ratio in males
at the high dose. An increase in relative liver weight was seen in
males and females at the high dose. No treatment-related gross
abnormalities were seen, but microscopic examination revealed effects
in the cytoplasm of four males at the high dose. The NOEL was 1500
mg/kg diet, equivalent to 230 mg/kg bw per day (Lina, 1992b).
Groups of five male and five female Wistar rats were fed diets
containing 8 cyclohexadecen-1-one at doses of 0, 600, 3000, or 15 000
mg/kg diet (equivalent to 0, 30, 120, or 750 mg/kg bw per day) for
four weeks. The animals were examined for clinical signs of toxicity,
and food and water consumption was monitored throughout the study.
Haematological and clinical chemical parameters were examined before
treatment and at the end of the study; the animals were also examined
macroscopically for pathological changes, and samples were taken for
histopathological examination. There were no deaths and no clinical
signs of toxicity.
A number of effects were seen in animals at the high dose.
Body-weight gain and food intake were decreased, and the red blood
cell counts and packed cell volume were increased. Clinical chemistry
revealed increased plasma urea and creatinine concentrations in males
and decreased albumin in females, and urinalysis showed increased
urinary density in animals of each sex. The absolute and relative
liver weights were increased in animals of each sex at the high dose
and also in males at the intermediate dose. The absolute and relative
adrenal weights were decreased in females at the high dose. Gross
examination revealed no remarkable findings, but microscopic
examination showed treatment-related hepatocellular cytoplasmic
alterations in animals of each sex at the high dose. There was no
evidence of degeneration or necrosis. The NOEL was 600 mg/kg diet,
equivalent to 30 mg/kg bw per day (Lina et al., 1986).
8-Cyclohexadecen-1-one was tested for mutagenic activity in
S. typhimurium strains TA1535, TA1537, TA1538, TA98, and TA100 with
and without microsomal activation. There was no increase in the number
of revertants of any of the strains tested (Wilmer, 1986).
8-Cyclohexadecen-1-one was also tested for its capacity to induce
micronuclei in animals treated in vivo. Groups of 30 CD-1 mice of
each sex were given the compound by gavage at a dose of 3 or 5 ml/kg
bw (equivalent to 2.8 and 4.6 g/kg bw). Five animals of each sex were
killed at 24 and 48 h and two of each sex at 72 h after treatment.
Examination of the bone marrow showed no increase in the number of
normochromatic erythrocytes over that in control mice (Willems, 1986).
3. ESTIMATE OF DIETARY INTAKE
gamma-Cyclodextrin can be used as a carrier for flavours,
sweeteners, and colours, and it has been proposed for use in this
manner in dry mixes for beverages, soups, dressings, gravies, sauces,
puddings, gelatines, and fillings and also in instant coffee and
instant tea, coffee whiteners, compressed sweets, chewing gum,
breakfast cereals, savoury snacks, crackers, and spices and
seasonings. It is also proposed for use as a carrier for vitamins and
polyunsaturated fatty acids in dry food mixes and in dietary
supplements, as a flavour modifier in soya milk, and as a stabilizer
in bread spreads, frozen dairy desserts, baked goods, bread,
fruit-based fillings, fat-based fillings, processed cheese, and dairy
desserts.
The estimated daily intake of gamma-cyclodextrin from its use in
food has been calculated by the dietary survey approach (Amann et al.,
1998), which is based on food consumption data from the 1989-91
Continuing Survey of Food Intakes by Individuals, in which data were
collected from a representative sample of individuals residing in
households in the United States. Each individual was surveyed over
three successive one-day periods, and the foods consumed were coded
into one of about 6000 different categories. For the purposes of the
calculation, it was assumed that each food (or food component) that
contains gamma-cyclodextrin contains this substance at the highest
feasible concentration. When gamma-cyclodextrin was used as a
component of the food, the intake of that component was calculated
from data on food consumption. The estimated daily intake was
calculated for each food in which gamma-cyclodextrin may be used from
data on one- and three-day food intake. Intakes were calculated both
per capita and per user. Users were defined as individuals who
consumed a food of at least one of the categories concerned on at
least one occasion.
The largest amounts of gamma-cyclodextrin are consumed with soya
milk and dairy desserts. Relatively high intakes (> 2 g/day) also
result from its use in bread spreads and fruit-based fillings. The
mean one-day intake of gamma-cyclodextrin from all its food uses was
estimated to be 4.1 g. The 90th percentile user was estimated to
ingest about 8.8 g on any one day. The three-day averages are 4 g for
the mean intake and 7.5 g for the 90th pecentile consumer. The intake
of gamma-cyclodextrin from chewing gum was estimated from a separate
survey on chewing-gum intake in the United States, and the average
consumer was estimated to ingest about 0.07 g gamma-cyclodextrin per
day. These data represent a 'worst-case' scenario and are based on the
assumption that gamma-cyclodextrin is used simultaneously in all
possible applications and at the highest feasible concentrations. The
realistic average daily intake of gamma-cyclodextrin would therefore
be lower than the levels indicated above.
4. COMMENTS
The Committee noted that the close structural similarity between
gamma-cyclodextrin and ß-cyclodextrin allows some comparisons to be
made of the metabolism and the toxicity of these two compounds. The
metabolism of gamma-cyclodextrin is different from that of
ß-cyclodextrin, as demonstrated both in vitro and in vivo. In rats
in vivo, gamma-cyclodextrin (at single doses up to 1000 mg/kg bw) is
rapidly metabolized to glucose, presumably by the luminal and/or
epithelial enzymes of the gastrointestinal tract. In contrast to the
metabolism of ß-cyclodextrin, there was little involvement of the
gastrointestinal microflora. Similarly, in contrast to ß-cyclodextrin,
much less gamma-cyclodextrin was absorbed and only very low levels
could be detected in the urine. After intravenous injection,
gamma-cyclodextrin (at single doses up to 600 mg/kg bw) was rapidly
cleared from the blood and was excreted, largely unchanged, in the
urine. Although no studies of metabolism in humans in vivo were
available, gamma-cyclo-dextrin, unlike ß-cyclodextrin, can be readily
hydrolysed by human salivary and pancreatic amylases in vitro.
Short-term (28- and 90-day) studies of toxicity indicate that
gamma-cyclodextrin has little toxicity when given intravenously or
orally to rats or orally to dogs. After administration of a very high
dietary concentration (20%), caecal enlargement and associated changes
were seen in both species. This effect is likely to result from the
presence of a high concentration of an osmotically active substance in
the large intestine. This result suggests that the metabolism of
gamma-cyclodextrin is less efficient at doses higher than those used
in the studies of metabolism. The results of the studies in rats
treated intravenously indicate that gamma-cyclodextrin is well
tolerated, even when given systemically, and may be less toxic than
ß-cyclodextrin.
Studies conducted in rats and rabbits with gamma-cyclodextrin at
doses up to 20% of the diet did not indicate any teratogenic effects.
Similarly, the results of a battery of studies of genotoxicity were
negative. Long-term studies of toxicity, carcinogenicity, and
reproductive toxicity have not been conducted, but, given the rapid
metabolism of this substance to glucose and its lack of genotoxicity,
the Committee concluded that such studies were not required for an
evaluation.
In vitro, gamma-cyclodextrin, like ß-cyclodextrin, sequestered
components of the membranes of erythrocytes, causing haemolysis. The
threshold concen-tration for this effect was, however, higher than
that of ß-cyclodextrin. Furthermore, gamma-cyclodextrin was not
detected in blood after dietary administration of high doses to
animals in vivo.
It was considered unlikely that interaction of gamma-cyclodextrin
with lipophilic vitamins would impair their bioavailability, because
of the rapid digestion of gamma-cyclodextrin in vivo. Also, there
was no evidence that vitamin deficiency was induced in experimental
animals given high doses of gamma-cyclodextrin.
The Committee considered studies on the short-term toxicity and
genotoxicity of the enzyme, cyclodextrin-glycosyl transferase, used in
the production of cyclodextrins, and of the complexant,
8-cyclohexadecen-1-one, used to optimize formation of
gamma-cyclodextrin. The data indicated that these substances are
unlikely to be of toxicological concern in final preparations of
gamma-cyclodextrin that comply with the specifications. The Committee
also considered information on genetic modifications in the organism
used to produce the enzyme, which raised no concern.
Although no studies of human tolerance to gamma-cyclodextrin were
submitted to the meeting, the Committee was aware that such a study
was available. Although it was unable to review the data, the
Committee was reassured by the relatively low toxicity of this
compound in animals and the fact that it is less toxic than
ß-cyclodextrin, for which studies of human tolerance were available.
Also, its rapid metabolism to maltose, maltotriose, and glucose
in vitro by human salivary and pancreatic amylases supports the
conclusion that, as in laboratory animals, it would be metabolized to
innocuous metabolites before absorption by humans.
5. EVALUATION
The Committee concluded that there were sufficient data to
allocate a temporary ADI 'not specified' but that the study of human
tolerance known to have been conducted should be reviewed in order to
confirm the absence of adverse gastrointestinal symptoms at normal
levels of intake. These data should be submitted for consideration by
the Committee by 1999.
6. REFERENCES
Abdullah, M., French, D. & Robyt, J.F. (1966) Multiple attack by
alpha-amylases. Arch. Biochem. Biophys., 114, 595-598.
Amann, M., Douglass, J., Doyle, E. & Heimbach, J. (1998) Estimated
intake for gamma-cyclodextrin in the USA from food use. Unpublished
report from TAS-Environ, Arlington, Virginia, USA. Submitted to WHO by
Bioresco Ltd, Binningen, Switzerland.
Bär, R. & Ulitzur, S. (1994) Bacterial toxicity of cyclodextrins:
Luminuous Escherichia coli as a model. Appl. Microbiol.
Biotechnol., 41, 574-577.
de Bie, A.T.H.J. & van Ommen, B. (1994) Oral and intravenous
disposition study with [14C] gamma-cyclodextrin in rats. Unpublished
report No. V93.279 from TNO-CIVO Institutes, Zeist, Netherlands.
Submitted to WHO by Bioresco Ltd, Binningen, Switzerland.
de Bie, A.T.H.J., van Ommen, B. & Bär, A. (1998) Disposition of
14C-gamma-cyclodextrin in germfree and conventional rats. Regul.
Toxicol. Pharmacol., submitted for publication.
Blijleven, W.G.H. (1991). Examination of gamma-cyclodextrin for
mutagenic activity in the Ames test. Report No. V91.027. Unpublished
report from TNO-CIVO Institute, Zeist, Netherlands. Submitted to WHO
by Bioresco Ltd, Binningen, Switzerland.
Dickhaus, S. & Heisler, E. (1985) Acute toxicological study of
8-cyclohexadecenon after oral application on the rat. Unpublished
report from Pharmatox Beratung und Forschung GmbH, Sehnde, Germany.
Submitted to WHO by Bioresco Ltd, Binningen, Switzerland.
Ehling G., Donaubauer, H.H., Mayer D., Langer, K.H. & Schmidts, H.L.
(1992) gamma-Cyclodextrin; repeated-dose (3-month) intravenous
toxicity in rats. Unpublished report No 92.0516 from Hoechst AG,
Frankfurt a.M., Germany. Submitted to WHO by Bioresco Ltd, Biningen,
Switzerland.
French D. (1957) The Schardinger dextrins. Adv. Carbohydr. Chem.,
12, 189-260.
Frijlink, H.W., Eissens, A.C., Hefting, N.R., Poelstra, K., Lerk, C.F
& Meijer, D.K.F. (1991) The effect of parenterally administered
cyclodextrins on cholesterol levels in the rat. Pharm. Res., 8,
9-16.
Fuchs, H., Donaubauer, H.H., Mayer D., Langer K.H. & Krieg K. (1992)
gamma-Cyclodextrin; repeated-dose (1-month) intravenous toxicity study
in rats. Unpublished report No 91.1007 from Hoechst AG, Frankfurt,
Germany. Submitted to WHO by Bioresco Ltd, Biningen, Switzerland.
Immel, H.R. (1991) Examination of gamma-cyclodextrin in the
micronucleus test. Unpublished report No. V90.436 from TNO-CIVO
Institutes, Zeist, Netherlands. Submitted to WHO by Bioresco Ltd,
Binningen, Switzerland.
Irie, T., Otagiri, M., Sunada, M., Uekama, K., Ohtani, Y., Yamada, Y.
& Sugiyama, Y. (1982) Cyclodextrin-induced hemolysis and shape changes
of human erythrocytes in vitro. J. Pharm. Dyn., 5, 741-744.
Jonker, D. (1994) Subchronic (13-week) oral toxicity study with an
aqueous solution of alpha-cyclodextrin-glycosyl-transferase
(alpha-CGT) in rats. Unpublished report No. V94.332 from TNO Nutrition
and Food Research Institute, Zeist, Netherlands. Submitted to WHO by
Bioresco Ltd, Binningen, Switzerland.
Kondo, H., Nakatani, H. & Hiromi, K. (1990) In vitro action of human
and porcine alpha-amylases on cyclo-maltooligosaccharides.
Carbohydr. Res., 204, 207-213.
Leroy-Lechat, F., Wouessidjewe, D., Andreux, J.P., Puisieux, F. &
Duchne, D. (1994) Evaluation of the cytotoxicity of cyclodextrins and
hydroxypropylated derivatives. Int. J. Pharm., 101, 97-103.
Lina, B.A.R. (1992a) Subchronic (90-day) oral toxicity study,
including a recovery study, with alpha-cyclodextrin and
gamma-cyclodextrin in rats. Unpublished report No. V 90.401 from
TNO-CIVO Institutes, Zeist, Netherlands. Submitted to WHO by Bioresco
Ltd, Biningen, Switzerland.
Lina, B.A.R. (1992b) Sub-acute (28-day) oral toxicity study with
8-cyclohexadecenone in mice. Unpublished report No. V91.509 from TNO
Nutrition and Food Research Institute, Zeist, Netherlands. Submitted
to WHO by Bioresco Ltd, Binningen, Switzerland.
Lina, B.A.R. & Bär, A. (1998) Subchronic oral toxicity studies with
gamma-cyclodextrin in rats. Regul. Toxicol. Pharmacol., submitted
for publication.
Lina, B.A.R. & Jonker, D. (1989) Tentative (2-week) oral toxicity
study with gamma-CD in rats. Unpublished report No. V89.263/298033
from TNO-CIVO Institutes, Zeist, Netherlands. Submitted to WHO by
Bioresco Ltd, Binningen, Switzerland.
Lina, B.A.R., van Nesselrooij, J.H.J. & Beems, R.B. (1986) Sub-acute
(28-day) oral toxicity study with 8-cyclohexadecenone in rats.
Unpublished report No. V86.498/260392 from TNO-CIVO Institutes, Zeist,
Netherlands. Submitted to WHO by Bioresco Ltd, Binningen, Switzerland.
Marshall J.J. & Miwa I. (1981) Kinetic difference between hydrolyses
of gamma-cyclodextrin by human salivary and pancreatic alpha-amylases.
Biochem. Biophys. Acta, 661, 142-147.
Matsuda K., Mera, Y., Segawa, Y., Uchida, I., Yokomine, A. & Takagi,
K. (1983) Acute toxicity study of gamma-cyclodextrin (gamma-CD) in
mice and rats. Pharmacometrics, 26, 287-291.
Matsuda, K., Segawa, Y., Yokomine, A., Uchida, I., Mera, Y. &
Tokugawa, H. (1985) Biodisposition of gamma-cyclodextrin (gamma-CD) in
the rabbit and dog after intravenous administration. Oyo Yakuri
(Pharmacometrics), 30, 889-896.
Ohtani, Y., Irie, T., Uekama K., Fukunaga, K. & Pitha, J. (1989)
Differential effects of alpha-, ß-, and gamma-cyclodextrins on human
erythrocytes. Eur. J. Biochem., 186, 17-22.
Okada, Y., Kubota, Y., Koizumi, K., Hizukuri, S., Ohfuji, T. & Ogata,
K. (1988) Some properties and the inclusion behavior of branched
cyclodextrins. Chem. Pharm. Bull., 36, 2176-2185.
Prinsen, M.K. (1990) Acute eye irritation/corrosion study with
gamma-cyclodextrin in albino rabbits. Unpublished report No.
V90.405/200069 from TNO-CIVO Institutes, Zeist, Netherlands. Submitted
to WHO by Bioresco Ltd, Binningen, Switzerland.
Prinsen, M.K. (1992) Sensitization study with gamma-CD in guinea pigs
(maximization test). Unpublished report No. V92.575/352063 from
TNO-CIVO Institutes, Zeist, Netherlands. Submitted to WHO by Bioresco
Ltd, Binningen, Switzerland.
Riebeek, W.M. (1990a) Determination of the acute intravenous toxicity
of gamma-cyclodextrin in mice. Unpublished report No. V90.222 from
TNO-CIVO Institutes, Zeist, Netherlands. Submitted to WHO by Bioresco
Ltd, Binningen, Switzerland.
Riebeek, W.M. (1990b) Determination of the acute intravenous toxicity
of gamma-cyclodextrin in rats. Unpublished report No. V90.220 from
TNO-CIVO Institutes, Zeist, Netherlands. Submitted to WHO by Bioresco
Ltd, Binningen, Switzerland.
Riebeek, W.M. (1990c) Determination of the acute intraperitoneal
toxicity of alpha-cyclodextrin and gamma-cyclodextrin in rats.
Unpublished report No. V90.233 from TNO-CIVO Institutes, Zeist,
Netherlands. Submitted to WHO by Bioresco Ltd, Binningen, Switzerland.
Spanjers, M.T. (1986) Determination of the acute oral toxicity (limit
test) of 8-cyclohexadecenone in mice. Unpublished report No.
V86.455/260393 from TNO-CIVO Institutes, Zeist, Netherlands. Submitted
to WHO by Bioresco Ltd, Binningen, Switzerland.
Szejtli, J., Csherhti, T. & Szgyi, M. (1986) Interactions between
cyclodextrins and cell-membrane phospholipids. Carbohydr. Polymers,
6, 35-49.
Til, H.P. & Bär, A. (1998) Subchronic (13-week) oral toxicity study of
gamma-cyclodextrin in dogs. Regul. Toxicol. Pharmacol., submitted
for publication.
Til, H.P. & van Nesselrooij (1992) Subchronic (90-day) feeding study
with gamma-CD in dogs. Unpublished report No. V91.475 from TNO-CIVO
Institutes, Zeist, Netherlands. Submitted to WHO by Bioresco Ltd,
Binningen, Switzerland.
Verhagen, F.J.J. & Waalkens-Berendsen, D.H. (1991) Oral
embryotoxicity/teratogenicity study with alpha-cyclodextrin and
gamma-cyclodextrin in rats. Unpublished report No. V91.210 from TNO
Nutrition and Food Research Institute, Zeist, Netherlands. Submitted
to WHO by Bioresco Ltd, Binningen, Switzerland.
de Vogel, N. & van Delft, J.H.M. (1996) Chromosome aberration test
with gamma-cyclodextrin in cultured human lymphocytes. Unpublished
report No. V96.398 from TNO-CIVO Institutes, Zeist, Netherlands.
Submitted to WHO by Bioresco Ltd, Binningen, Switzerland.
Waalkens-Berendsen, D.H. & Smits-van Prooije, A.E. (1992) Final report
of an oral embryotoxicity/teratogenicity study with alpha-cyclodextrin
and gamma-cyclodextrin in New Zealand white rabbits. Unpublished
report No. V91.564 from TNO Nutrition and Food Research Institute,
Zeist, Netherlands. Submitted to WHO by Bioresco Ltd, Binningen,
Switzerland.
Willems, M.I. (1986) Evaluation of 8-cyclohexadecenone in the
micronucleus test. Unpublished report No. V86.456/260393 from TNO-CIVO
Institutes, Zeist, Netherlands. Submitted to WHO by Bioresco Ltd,
Binningen, Switzerland.
Wilmer, J.W.G.M. (1986) Examination of 8-cyclohexadecenone for
mutagenic activity in the Ames test. Unpublished report No.
V86.264/260064 from TNO-CIVO Institutes, Zeist, Netherlands. Submitted
to WHO by Bioresco Ltd, Binningen, Switzerland.
Yim, D.G., Sato, H.H., Park, Y.H. & Park, Y.K. (1997) Production of
cyclodextrin from starch by cyclodextrin glycosyltransferase from
Bacillus firmus and characterization of purified enzyme. J. Ind.
Microbiol. Biotechnol., 18, 402-405.
Yoshida, A., Arima, H., Uekama, K. & Pitha, J. (1988) Pharmaceutical
evaluation of hydroxyalkyl ethers of ß-cyclodextrins. Int. J.
Pharmacol., 46, 217-222.
gamma-Cyclodextrin has not been reviewed previously by the
Committee; however, the structurally related ß-cyclodextrin (seven
glucose units) was evaluated at the forty-first and forty-fourth
meetings of the Committee (Annex 1, references 107 and 116).
2. BIOLOGICAL DATA
2.1 Biochemical aspects
2.1.1 Absorption, distribution, biotransformation, and excretion
Early experiments provided some evidence that gamma-cyclodextrin
(unlike alpha- or ß-cyclodextrin) is hydrolysed by salivary amylase
(French, 1957). More recent studies have confirmed that both human
salivary amylase and human or porcine pancreatic amylase can readily
hydrolyse gamma-cyclodextrin, yielding mainly maltose, some
maltotriose, and smaller amounts of glucose (Abdullah et al., 1966;
Marshall & Miwa, 1981; Kondo et al., 1990).
The fate of intravenously administered gamma-cyclodextrin was
examined in three male and two female rabbits and one male dog. The
rabbits were each given a dose of 7.5 g in saline (30 ml of a 25% w/v
solution), while the dog was given a dose of 25 g in saline (500 ml of
a 5% w/v solution at 8.3 ml/min). Blood and urine samples were
collected and examined by high-performance liquid chromatography
(HPLC) for gamma-cyclodextrin over 4 h. The half-life of
gamma-cyclo-dextrin in blood was about 50 min in rabbits and 30 min in
the dog. In rabbits, about 77% of the injected dose was recovered as
unchanged gamma-cyclodextrin in the urine within 24 h, while in the
dog, nearly 80% of the injected dose was recovered as unchanged
gamma-cyclodextrin in the urine within the first 4 h (Matsuda et al.,
1985).
Two male Wistar rats were given 14C-gamma-cyclodextrin (94%
radiochemical purity) as single oral or intravenous doses. After an
oral dose of 14C-gamma-cyclo-dextrin (25 µCi/kg bw diluted to 200
mg/kg bw), 50% of the radiolabel was excreted in the exhaled air
within 24 h, while 2% was excreted in the urine and 5% in the faeces;
35% of the radiolabel was retained in the body after 24 h. No
detectable gamma-cyclodextrin was found in blood during 8 h after
administration, suggesting metabolism in the gastrointestinal tract.
After the intravenous dose of 14C-gamma-cyclodextrin (25 µCi/kg
bw diluted to 100 mg/kg bw), 90% of the radiolabel was excreted in the
urine within 8 h, mainly as unchanged gamma-cyclodextrin. After 8 h,
small amounts of radiolabel were excreted in exhaled air (1.1%), while
0.5% was detected in the faeces; 6% was retained in the body after 24
h (de Bie & van Ommen, 1994).
Groups of four male Wistar rats were given single oral or
intravenous doses of 14C-gamma-cyclodextrin. The oral dose was 1000
mg/kg bw, and the concentration of radiolabel was either 25 or 100
µCi/kg bw. In order to examine the role of the gut microflora in the
metabolism of gamma-cyclodextrin, a separate group of germ-free male
rats was given 25 µCi/kg bw gamma-cyclodextrin diluted to 1000 mg/kg
bw. The intravenous dose was 600 mg/kg bw, and the concentration of
radiolabel was 25 µCi/kg bw.
In the first experiment, the time-course and routes of metabolism
were examined after oral administration. After administration of
1000 mg/kg bw (25 µCi/kg bw) 14C-gamma-cyclodextrin, 62% of the
radiolabel in males and 71% in females was excreted within 48 h. The
majority was excreted in expired air (57% in males and 65% in
females), with maximum excretion between 1 and 2 h after dosing.
Little was excreted in the urine (2.1% for males and 2.5% for females)
or faeces (2.9% for males and females) during the 48-h sampling
period. Retention of radiolabel after 48 h was 27% in males and 22% in
females, with the most significant retention in the liver (11% in
males, 5.2% in females). The amounts in all other organs were low:
intestine, 0.42% for males, 0.36% for females; kidney, 0.13% for
males.
In the second experiment, the excretion kinetics and time-course
in blood over 24 h were examined after a single oral dose of
1000 mg/kg bw (100 Ci/kg bw) 14C-gamma-cyclodextrin. The major route
of excretion was exhaled air (55% for males; 53% for females) during
the 24-h sampling period. The carcass retained 35% of the radioactive
dose in males and 25% in females at 24 h after treatment. The maximum
concentration in blood was reached 40 min after treatment, but this
concentration decreased by 50% after 0.5 h in males and after 6 h in
females. The urine of animals of each sex contained a peak that eluted
before glucose at all times and a small peak that co-eluted with
glucose (6% of total radiolabel). A small amount of unchanged
gamma-cyclo-dextrin was detected in urine after 4 h (< 0.02% of the
ingested dose). HPLC analysis of the stomach contents 24 h after
dosing revealed the presence of gamma-cyclodextrin and a minor peak
that co-eluted with glucose. HPLC analysis of the blood revealed the
presence of a radioactive peak that co-eluted with glucose.
In the third experiment, the excretion kinetics and time-course
in blood over 24 h were examined after a single intravenous dose of
600 mg/kg bw (25 µCi/kg bw) 14C-gamma-cyclodextrin. The major route
of excretion was the urine (87% for males, 91% for females), and the
faeces contained about 1% and the gastrointestinal tract about 0.5% of
the radiolabel; the exhaled air contained 7.8% of the dose for males
and 3.2% for females. The carcass contained 3.9% of the dose in males
and 2.2% in females, the liver having the most residue (0.41% in
males, 0.42% in females). Radiolabel was removed rapidly from the
blood, with calculated initial half-lives of 20 min in males and 15
min in females. HPLC analysis of blood revealed predominantly
gamma-cyclodextrin.
In the fourth experiment, excretion kinetics over 24 h was
examined in germ-free rats given a single oral dose of 1000 mg/kg bw
(25 µCi/kg bw) gamma-cyclodextrin. About 73% of the radiolabel was
excreted in expired air (68% in males, 65% in females). Peak
exhalation of 14C-carbon dioxide occurred at about 90 min. Low
concentrations of radiolabel were found in faeces and urine. The
gastrointestinal tract contained 9.2% in males and 11% in females,
with the majority in the caecum. The results indicate that
gastrointestinal microflora play little part in the metabolism of
gamma-cyclodextrin.
Overall, this study indicates that gamma-cyclodextrin is
metabolized to glucose, other low-molecular-mass sugar metabolites,
and carbon dioxide by luminal and/or epithelial enzymes of the
gastrointestinal tract (de Bie & van Ommen, 1994; de Bie et al.,
1998).
2.2 Toxicological studies
2.2.1 Acute toxicity
The acute toxicity of gamma-cyclodextrin was examined in mice and
rats; the results are summarized in Table 1. In each study, groups of
10 animals of each sex were tested at three doses or served as
controls. There were no deaths at the highest doses tested (Matsuda et
al., 1983).
Table 1. Acute toxicity of gamma-cyclodextrin
Species Sex Route LD50 Reference
(mg/kg bw)
Mouse Male and female Oral >16 000 Matsuda et al. (1983)
Mouse Male and female Subcutaneous > 4 000 Matsuda et al. (1983)
Mouse Male and female Intravenous > 4 000 Matsuda et al. (1983)
Mouse Male and female Intravenous 10 000 Riebeek (1990a)
Rat Male and female Oral > 8 000 Matsuda et al. (1983)
Rat Male and female Subcutaneous > 2 400 Matsuda et al. (1983)
Rat Male and female Intravenous > 3 750 Matsuda et al. (1983)
Rat Male and female Intravenous > 3 750 Riebeek (1990b)
Rat Male Intraperitoneal > 4 600 Riebeek (1990c)
In a test for micronucleus formation (see section 2.2.3), groups
of 15 Swiss mice of each sex were given a single dose of 15 g/kg bw
gamma-cyclodextrin by gavage and were killed in groups of five at 24,
48, and 72 h. All of the animals survived until termination (Immel,
1991).
The toxicity of intravenously administered gamma-cyclodextrin was
examined in groups of five male and five female Crl:CD mice and five
male and five female Crl:W1(WU)BR rats. The mice were given
gamma-cyclodextrin (purity, > 98%) at doses of 5000, 7500, or 10 000
mg/kg bw, while the rats were given doses of 2500, 3750, or 5000 mg/kg
bw as a solution in sterile saline. The animals were observed for
clinical signs over 14 days after treatment. They were examined for
gross pathological changes on day 14 after treatment.
Both mice and rats showed dose-related signs of toxicity, such as
piloerection and sluggishness, within 1 h to a few days after
treatment. Deaths occurred within a few days in both species, but the
surviving animals recovered and appeared to be healthy at the end of
the observation period. Macroscopic examination revealed no gross
treatment-related alterations (Riebeek, 1990a,b).
The toxicity of intraperitoneally administered gamma-cyclodextrin
was examined in groups of three male rats after single doses of 2000
or 4600 mg/kg bw as a solution in sterile saline. The animals were
observed for clinical signs during 14 days after treatment and were
examined grossly for pathological changes on the last day. There were
no signs of toxicity and no deaths. Macroscopic examination revealed
no gross treatment-related alterations (Riebeek, 1990c).
2.2.2 Short-term studies of toxicity
Rats
Groups of 15 Wistar rats of each sex were given
gamma-cyclodextrin intravenously as single daily doses of 0, 200, 630,
or 2000 mg/kg bw for 30 days. A group of five animals of each sex were
allowed to recover for 27 days after treatment. The animals were
examined throughout the study, and the body weights recorded weekly.
Clinical chemical parameters were examined at the end of treatment and
after the recovery period. Ten animals from each group were killed at
the end of treatment and the remainder at the end of the recovery
period.
There were no clinical signs of toxicity throughout the study.
The body-weight gain of males at 630 and 2000 mg/kg bw per day was
slightly decreased, but the difference was not significant. Food
consumption was decreased in a dose-related manner only during the
first few days of treatment. Significantly decreased mean values for
haemoglobin concentration and haemacrit were seen in males at doses
> 630 mg/kg bw per day, and significantly decreased erythrocyte
count in males at 2000 mg/kg bw per day. In females, the erythrocyte
count, haemoglobin concentration, and haematocrit were significantly
decreased only at 2000 mg/kg bw per day. The reticulocyte counts were
significantly increased in both males and females at 2000 mg/kg bw per
day. These changes were reversed after the recovery period. The only
significant clinical chemical change was an increase in creatinine and
urea in both males and females at 2000 mg/kg bw per day, which was
reversed at the end of the recovery period. Urinalysis revealed a
significantly increased incidence of haemoglobin in the urine of rats
at 2000 mg/kg bw per day, which was reversed at the end of the
recovery period.
Pathological examination at autopsy showed that the majority of
animals at 2000 mg/kg bw per day had light-coloured and, in some
cases, irregularly coloured kidneys. A dose-dependent increase in
spleen weight was found in all groups, which was significant in males
at doses > 630 mg/kg bw per day and in females at all doses. Liver
weights were significantly increased in females at > 630 mg/kg bw
per day, and kidney weights were significantly increased at 2000 mg/kg
bw per day. Lung and adrenal weights were significantly increased in
both males and females at 2000 mg/kg bw per day. These changes were
partially reversed after the recovery period, but a significant
increase in kidney and spleen weights was still present in females at
2000 mg/kg bw per day. Histopathological examination revealed marked
vacuolation of renal epithelial cells in the proximate convoluted
tubules, which may have been related to administration of a substance
that causes hyperosmolarity. There was also extensive pulmonary
histiocytosis (massive accumulation of alveolar macrophages) in all
animals at 2000 mg/kg bw per day. These changes were seen only in some
animals at 630 mg/kg bw per day and at lesser severity. Considerable
reversibility was observed after the recovery period. The NOEL was 200
mg/kg bw per day (Fuchs et al., 1992).
Groups of 15 Wistar rats of each sex were given
gamma-cyclodextrin intravenously as single daily doses of 0, 60, 120,
or 600 mg/kg bw for 90 days. Groups of five animals of each sex were
allowed to recover at the end of the treatment period. The animals
were examined throughout the study, and body weights were recorded
weekly. Haematological and urinary parameters were examined after one
month, at the end of the study, and on day 37 (males) or 36 (females)
of recovery. Clinical chemical parameters were examined at the end of
the treatment period and after recovery. Animals were killed after
90 days, their organs were examined, and tissues were taken for
histopathological examination.
There were no clinical signs of toxicity. Body-weight gain was
slightly decreased only in males at the high dose, and food
consumption was decreased in a dose-related manner only during the
first few days of treatment. Haematological examination revealed a
slight decrease in the mean erythrocyte count, haemaglobin
concentration, and haemacrit in females at one month, and these values
were significantly decreased at three months only in females at the
high dose. The reticulocyte counts were significantly increased in
females at the high dose at three months. At the end of the recovery
period, all of these values were normal. The only clinical chemical
changes were a slight decrease in bilirubin concentration in males and
females at the high dose and a slight increase in alkaline phosphatase
activity in males at the high dose. At the end of the recovery period,
all of the values were normal. The slight changes observed in urinary
parameters were considered not to be treatment-related.
Histological examination at autopsy revealed an increased
incidence of enlarged iliac lymph nodes in males and females at the
high dose, which was probably related to inflammation at the injection
site. Increases in the relative weights of lungs, liver, and spleen
were seen in males and in the relative weights of heart, lungs,
kidneys, spleen, and adrenal glands in females at the high dose. All
of these changes were reversed after the four-week recovery period.
Histopathological examination revealed hyperplasia of the mucosa of
the urinary bladder, which was reversed after four weeks, and
pulmonary histiocytosis (macrophage aggregates), which tended to
reverse after four weeks, in males and females at the high dose. The
reversibility of the urinary bladder changes indicates that the
hyperplasia was not preneoplastic. The NOEL was 120 mg/kg bw per day
(Ehling et al., 1992).
Groups of five male Wistar rats were fed diets containing
gamma-cyclo-dextrin at concentrations of 0, 5, 10, 15, or 20%,
equivalent to 0, 2500, 5000, 7500, or 10 000 mg/kg bw per day, for 14
days. A control group was fed 20% lactose. The animals were examined
for clinical signs of toxicity, and food and water consumption was
monitored throughout the study. On day 14, the animals were killed,
and blood samples were taken for examination of clinical chemical
parameters. The animals were also examined macroscopically for
pathological changes.
There were no deaths. The incidence of soft stools was slightly
higher in treated than control animals, and the mean body weights of
treated animals were slightly lower than those of controls, although
this difference was not statistically significant. Food intake was
slightly lower in controls than in treated groups. Plasma alkaline
phosphatase activity was increased in animals at 15 and 20%, while
gamma-glutamyl transferase activity was decreased in all treated
groups. The activity of aspartame and alanine aminotransferases were
unchanged, as were the concentrations of total protein, albumin, urea,
and bilirubin. Enlargement of the caecum was seen at all doses, the
most significant effect occurring in the group fed 20% lactose. There
were no macroscopic changes attributable to treatment (Lina & Jonker,
1989; Lina & Bär, 1998).
Groups of 20 Wistar rats of each sex were fed diets containing
gamma-cyclo-dextrin at concentrations of 0, 1.5, 5, or 20% (equivalent
to 0, 750, 2500, or 10 000 mg/kg bw per day) for 13 weeks. A control
group was fed 20% lactose. In order to examine the reversibility of
any effects seen, a group of 10 animals of each sex were fed either
20% gamma-cyclodextrin or 20% lactose for 13 weeks and then control
diet for one month. As a result of a feeding error, the groups
receiving 20% gamma-cyclodextrin were discarded, and two additional
groups were fed 0 or 20% gamma-cyclodextrin for 13 weeks and 10
animals receiving the 20% dose were fed control diet for an additional
month at the end of the 13-week period. Animals were examined for
clinical signs of toxicity, and food and water consumption was
monitored throughout the study. Ophthalmoscopic examinations were
conducted in animals at the high dose and those given lactose before
treatment and at the end of the study. The animals were killed at the
end of the study, and blood samples were taken for clinical chemistry.
The animals were also examined macroscopically for pathological
changes.
There were no treatment-related deaths during the study. Soft
stools were observed in some rats at 5 or 20% gamma-cyclodextrin and
in those given lactose early in the study, but this effect disappeared
subsequently. Ophthalmoscopic examination revealed no
treatment-related effects. The body weights of males at 20%
gamma-cyclodextrin were slightly but significantly decreased
throughout treatment; a more significant body-weight decrease was seen
in the males given 20% lactose. No difference was noted in these
groups during the recovery period. Food conversion efficiency was also
slightly decreased in males at 20% lactose. None of the changes in
haematological parameters was considered to be of toxicological
significance. Some changes in clinical chemistry were seen in females
at the high dose, namely increased activity of aspartate and alanine
aminotransferases, especially in one female. The sodium concentrations
were slightly higher but within the historical control range. Urinary
parameters were similar in control and treated groups, apart from a
significant increase in the urinary calcium concentration at the end
of treatment in males given 20% gamma-cyclodextrin and rats of each
sex given 20% lactose; the effect was more pronounced in the latter
group. The calcium:creatinine ratio was significantly increased in
rats receiving 20% lactose. The increased calcium appeared to be
associated with the increased load of osmotically active substances in
the large intestine; there was no increase in calcium at the end of
the recovery period.
Analysis of organ weights revealed significant increases in both
absolute and relative caecal weights in rats at 5 and 20%
gamma-cyclodextrin and 20% lactose in comparison with controls. At the
end of the recovery period, no difference from controls was seen for
rats given 20% gamma-cyclodextrin, but the increase was still evident
in those given 20% lactose. This effect is probably related to the
retention of an osmotically active substance in the large intestine.
Relative adrenal weights were increased in males given 20%
gamma-cyclodextrin and the lactose control group at the end of the
study. A very slight increase in relative liver weight was seen in
males at 20% gamma-cyclodextrin and females given lactose. No changes
in organ weight were seen at the end of the recovery period.
There were no gross pathological changes attributable to
treatment with gamma-cyclodextrin. Microscopic examination revealed
increased cortico-medullary mineralization in the kidneys of rats at
1.5 and 5% gamma-cyclodextrin but not at the high dose. This change is
considered to be relatively common in rats and not related to
treatment with gamma-cyclodextrin. There was no evidence of
hyperplasia of the mucosa of the urinary bladder or of pulmonary
histiocytosis, which were observed after intravenous administration.
The effects observed in rats after receiving gamma-cyclodextrin
in the diet at concentrations up to 20% appeared to be largely related
to the presence of high levels of an osmotically active substance in
the large intestine and not of toxicological significance. Similar,
and generally more pronounced, effects were observed when the diet
contained 20% lactose. On the basis of this study, dietary levels of
20% gamma-cyclodextrin (equivalent to10 000 mg/kg bw per day) were
considered to be tolerated without toxicological effects (Lina, 1992a;
Lina & Bär, 1998).
Dogs
Groups of four male and four female beagle dogs were fed diets
containing gamma-cyclodextrin at doses of 0, 5, 10, or 20% (equivalent
to 0, 1250, 2500, or 5000 mg/kg bw per day). Body weight and food
consumption were recorded weekly throughout the study. Ophthalmoscopic
examinations were made at the beginning and end of the study. Blood
was collected for clinical chemical and haematological examination
before the start of treatment and at weeks 6 and 13. Urinalyses were
performed at week 13. During week 14, all animals were killed, their
organs were examined, and tissues were prepared for histopathological
examination.
There were no deaths during the study and no clinical signs of
toxicity. Diarrhoea was noted in all groups, increasing in incidence
and severity with increasing doses of gamma-cyclodextrin, and was more
severe in females than in males. This effect was attributed to the
slow digestion and intestinal absorption of gamma-cyclodextrin.
Ophthalmoscopic examination revealed no differences between treated
and control animals. Weight gain was comparable in all groups, except
for a slight, non-significant decrease in males at 20% during the last
six weeks of the study. Treated dogs had slightly decreased food
intake during the first two weeks of the study, particularly at the
high dose. This effect persisted throughout the study only in dogs at
the high dose. No differences in haematological parameters were seen,
and clinical chemical parameters were similar in treated and control
groups. Urinalysis revealed a slight decrease in urinary pH in the
group receiving 20% gamma-cyclodextrin, which was considered to be
associated with the intake of poorly digestible carbohydrates and
increased fermentation in the large intestine.
Both the absolute and relative weights of the ovary were
increased in animals at 10 and 20% gamma-cyclodextrin, but no
histopathological changes were seen, and the weight of the ovaries in
the control group was lower than that of other dogs in the same
laboratory. The changes observed are considered unlikely to be
toxicologically significant. There was no change in absolute liver
weight, although the relative liver weight was increased in males at
the high dose as a result of the slight decrease in body weight at
this dose. This change is likely to be adaptive and was considered not
to be toxicologically significant. Caecal weights were increased in
the dogs at 10 and 20% gamma-cyclodextrin but statistically
significantly only in females. This change is considered to be related
to the presence of high levels of an osmotically active substance in
the large intestine. Pathological examination at the end of the study
revealed no treatment-related change. Similarly, the histopathological
findings were unremarkable. On the basis of this study, dietary levels
of 20% gamma-cyclodextrin (equivalent to 5000 mg/kg bw per day) were
considered to be tolerated without toxicological effects (Til & van
Nesselrooij, 1992; Til & Bär, 1998).
2.2.3 Genotoxicity
The results of assays for genotoxicity with gamma-cyclodextrin
are shown in Table 2.
2.2.4 Developmental toxicity
Rats
In a study of embryotoxicity and teratogenicity, groups of 25
presumed pregnant Wistar Crl:(WI)WU BR rats were fed diets containing
gamma-cyclodextrin (purity, >98%) at concentrations of 0. 1.5, 5, 10,
or 20% (equivalent to 0, 2500, 5000, or 10 000 mg/kg bw per day) on
days 0-21 of gestation. A separate group received a diet containing
20% lactose instead of pre-gelatinized potato starch. The animals were
examined throughout the study, and body weight and food consumption
were recorded. The rats were killed on day 21 and examined for
parameters of reproductive performance. Fetuses were examined for
signs of toxicity, external malformations, and soft-tissue defects and
were stained for detection of skeletal anomalies.
No deaths occurred during the study. Maternal body-weight gain
was similar in all groups except for a slight reduction at the 20%
dose on days 0-16 of gestation. Necropsy of the dams showed no adverse
effects that could be related to treatment. The number of viable
litters, the number of corpora lutea, and the mean number of
implantation sites were similar in all groups. Fetal length and body
weight were also similar in all groups. Examination of the fetuses
revealed no treatment-related increase in gross, skeletal, or visceral
abnormalities. Under the conditions of this assay, gamma-cyclodextrin
was not teratogenic (Verhagen & Waalkens-Berendsen, 1991).
Rabbits
In a study of embryotoxicity and teratogenicity, groups of 16
presumed pregnant New Zealand white rabbits were fed diets containing
gamma-cyclodextrin (purity, > 98%) at concentrations of 0, 5, 10, or
20% (equivalent to 0, 1500, 3000, or 6000 mg/kg bw per day) on days
0-29 of gestation. The animals were examined throughout the study, and
body weights and food consumption were recorded regularly. The animals
were killed on day 29 of pregnancy, and the fetuses were examined for
signs of toxicity, external malformations, and soft-tissue defects and
were stained for detection of skeletal anomalies.
Table 2. Results of assays for the genotoxicity of gamma-cyclodextrin
End-point Test object Concentration Result Reference
Bacterial gene S. typhimurium TA1535, 2-20000 Negativea Blijleven (1991)
mutation TA1537, TA 1538, µg/plate
TA98, TA100
Chromosomal Human lymphocytes 10-5000 Negativea de Vogel & van
aberrationb µg/ml Delft (1996)
Micronucleus CD-1 mouse bone 15 g/kg bw Negative Immel (1991)
formationb marrow (single dose)
a With and without exogenous metabolic activation
b Two assays conducted: treatment times, 6, 24, and 48 h; harvest times, 24 and 48 h
No deaths occurred during the study. Diarrhoea was seen in
several treated animals. Maternal body weight was significantly
decreased at the 20% dose during the first week of treatment but not
thereafter. Necropsy of the does showed no adverse effects that could
be related to treatment. The number of viable litters, the number of
corpora lutea, and the mean number of implantation sites were similar
in all groups. One abortion occurred in the control group and one in
rabbits at the 10% dose. Fetal length and body weight were similar in
all groups. Examination of the fetuses revealed no treatment-related
increase in gross, skeletal, or visceral abnormalities. An increase in
the incidence of haemorrhagic fluid in animals at the 5 and 20% doses
was considered unlikely to be treatment-related. Under the conditions
of this assay, gamma-cyclodextrin was not teratogenic
(Waalkens-Berendsen & Smits-van Prooije, 1992).
2.2.5 Special studies
2.2.5.1 Ocular irritation and dermal sensitization
gamma-Cyclodextrin was not irritating or corrosive to the eyes of
albino rabbits (Prinsen, 1990). In a skin sensitization assay in
guinea-pigs, a 30% solution of gamma-cyclodextrin induced no signs of
sensitization (Prinsen, 1992).
2.2.5.2 Cell membrane effects
The interactions between alpha-, beta-, and gamma-cyclodextrin
and membrane phospholipids, liposomes, and human erythrocytes were
studied in vitro. gamma-Cyclodextrin and the other cyclodextrins did
not increase the permeability of dipalmityol-phosphatidylcholine
liposomes, nor did they affect the active transport of 42K+ into
erythrocytes at a concentration of 10-2 mol/L. ß-Cyclo-dextrin but
not gamma-cyclodextrin or alpha-cyclodextrin increased the release of
42K+, 86Rb+, and 137Cs+ from erythocytes by passive transport at
a concentration of 1.7 × 10-2 mol/L (Szejtli et al., 1986).
gamma-Cyclodextrin induced haemolysis of human erythrocytes
in vitro in isotonic saline after 30 min, with a threshold
concentration of 16 mmol/L (approximately 20 mg/L). A similar effect
was seen with alpha-cyclodextrin at 6 mmol/L and with ß-cyclodextrin
at 3.5 mmol/L. At 15 mmol/L, gamma-cylodextrin caused a 2% release of
cholesterol from erythrocytes, indicating that cyclodextrin-induced
haemolysis is associated with removal of membrane components from
erythrocytes (Irie et al., 1982). Other studies have confirmed that
gamma-cyclodextrin causes erythrocyte haemolysis at concentrations of
15-30 mmol/L (Okada et al., 1988; Yoshida et al., 1988; Leroy-Lechat
et al., 1994).
In a study on the differential effects of cyclodextrins on human
erythrocytes in vitro, alpha-, ß-, and gamma-cyclodextrin were
incubated at increasing concentrations with erythrocytes for 30 min.
The cyclodextrins induced a change in shape, from discocyte to
spherocyte, but with ß-cyclodextrin haemolysis occurred before the
change was complete. gamma-Cyclodextrin was less potent than either
alpha- or ß-cyclodextrin in releasing potassium and haemoglobin from
erythrocytes and in solubilizing various components of erythrocytes,
including phospholipids, cholesterol, and proteins. A concentration of
4 mmol/L gamma-cyclodextrin was required to induce release of
phospholipid, and > 20 mmol was required for release of cholesterol
or protein (Ohtani et al., 1989).
The potential cytotoxicity of the cyclodextrins to P388 murine
leukaemia cells was examined by exposing the cells to increasing
concentrations of cyclodextrins for 48 h in medium containing 10%
fetal calf serum. Initial cytotoxicity was elicited at 11 mmol/L
alpha-cyclodextrin, 2.5 mmol/L ß-cyclodextrin, and 55 mmol/L
gamma-cyclodextrin (Leroy-Lechat et al., 1994).
Potential reductions in the luminescence of a strain of E. coli
after incubation with various concentrations of cyclodextrins were
examined. Luminescence was reduced by 20 and 50% by ß-cyclodextrin at
concentrations of 0.03 and 0.5%, respectively. With
gamma-cyclodextrin, a concentration of 2% was required to reduce the
luminescence by 20%, and a 50% reduction could not be achieved even at
the highest concentration of 10% gamma-cyclodextrin (Bär & Ulitzur,
1994).
2.2.5.3 Impurities
Cyclodextrin-glycosyl transferase
The gene coding for cyclodextrin-glycosyl transferase is derived
from a strain of the Bacillus firmus-lintus complex. This is a
ubiquitous group of aerobic, gram-positive, alkalophilic
microorganisms which form sub-terminal endospores. Both B. firmus
and B. lintus are considered to be non-pathogenic. After isolation
from this strain, the gene was cloned into pUC18, isolated again and
inserted into an expression and secretion vector derived from pJF118EH
by introduction of a DNA fragment coding for an appropriate signal
peptide. The vector, pJF118EH, is derived from pBR322, a widely used
vector which is considered to be safe. All genetic modifications were
made by directed mutagenesis by standard techniques. The gene that has
been introduced for production of the signal peptide is cleaved off
during exportation of cyclodextrin-glycosyl transferase and degraged
with the E. coli cell.
A strain derived from E. coli K12 was used as the host for the
cyclodextrin-glycosyl transferase expression and secretion vector.
E. coli K12 is considered to be a non-pathogenic strain and is used
for production of enzymes for food use.
For the production of cyclodextrin-glycosyl transferase, the
recombinant E. coli strain was cultured in a standard medium. The pH
of the fermentation broth was adjusted by adding food-grade ammonia or
phosphoric acid. The E. coli was grown to a certain density, after
which enzyme production was initiated by addition of isopropyl
thiogalactoside. The bacteria were then removed by fermentation and
the supernatant filtered and concentrated to the crude
cyclodextrin-glycosyl transferase preparation.
Groups of 20 Wistar rats of each sex were given an aqueous
solution of a cyclodextrin-glycosyl transferase preparation at doses
of 0, 5, 10, or 20 ml/kg bw per day, equivalent to 0, 65, 130, or 260
mg/kg bw per day of total organic solids (TOS), by gavage for 13
weeks. The animals were examined for clinical signs of toxicity, and
food and water consumption was monitored throughout the study.
Ophthalmoscopic examinations were conducted in rats at the high dose
before treatment and at the end of the study. Haematological
parameters were examined before treatment, on days 30 and 60, and at
the end of the study. The animals were killed at the end of the study,
and blood samples were taken for examination of clinical chemical
parameters. The animals were also examined macroscopically for
pathological changes, and samples were taken for histopathological
examination.
Two animals (one control male and one female at the low dose)
died as a result of dosing accidents. There were no clinical signs of
toxicity in the treated animals which survived. Ophthalmoscopic
examination of animals at the high dose revealed no treatment-related
changes. There was no significant difference in body weight between
treated and control rats, and food consumption was unchanged in the
treated group. There were no treatment-related haematological,
clinical chemical, or urinary changes. The organ weights of control
and treated groups were similar, and there were no treatment-related
gross pathological changes. Histopathological examination revealed an
increased incidence of pulmonary changes in the treated animals which
were considered to be related to aspiration of test material. The NOEL
was 20 ml/kg bw per day, equivalent to 260 mg/kg bw per day of TOS,
the highest dose tested (Jonker, 1994).
The cyclodextrin-glycosyl transferase preparation was tested for
mutagenic activity in S. typhimurium strains TA1535, TA1537, TA98,
and TA100 with and without metabolic activation at concentrations of
19-1500 µg/plate or 62-500 µg/plate. The assay was slightly
complicated by the growth-stimulating effects of some of the
components of the crude preparation, but the overall result confirmed
that cyclodextrin-glycosyl transferase had no mutagenic activity in
these strains.
8-Cyclohexadecen-1-one
8-Cyclohexadecen-1-one is a colourless, waxy, solid product. The
commercial preparation has a purity of > 98%. The acute toxicity of
8-cyclo-hexadecen-1-one was examined in five male and five female mice
at a dose of 5 ml/kg bw (4.6 g/kg bw) and in five male and five female
rats at doses of 5 and 10 g/kg bw. The animals were observed for 14
days. Two of the five mice died during the study, but there were no
deaths among the rats. It was concluded that the LD50 was > 4.6 g/kg
bw in mice and > 10 g/kg bw in rats (Dickhaus & Heisler, 1985;
Spanjers, 1986).
Groups of five CD-1 mice of each sex were fed diets containing
8-cyclo-hexadecen-1-one at doses of 0, 500, 1500, or 7500 mg/kg diet
(equivalent to 0, 75, 230, or 1100 mg/kg bw per day) for four weeks.
The animals were examined for clinical signs of toxicity, and food and
water consumption were monitored throughout the study. Haematological
and clinical chemical parameters were examined before treatment and at
the end of the study. The animals were also examined macroscopically
for pathological changes, and samples were taken for histopathological
examination. There were no deaths and no clinical signs of toxicity.
The body-weight gain of mice at the high dose was decreased, but food
and water intake were similar in treated and control groups. There
were no treatment-related haematological changes. The only clinical
chemical change was a decrease in the albumin:globulin ratio in males
at the high dose. An increase in relative liver weight was seen in
males and females at the high dose. No treatment-related gross
abnormalities were seen, but microscopic examination revealed effects
in the cytoplasm of four males at the high dose. The NOEL was 1500
mg/kg diet, equivalent to 230 mg/kg bw per day (Lina, 1992b).
Groups of five male and five female Wistar rats were fed diets
containing 8 cyclohexadecen-1-one at doses of 0, 600, 3000, or 15 000
mg/kg diet (equivalent to 0, 30, 120, or 750 mg/kg bw per day) for
four weeks. The animals were examined for clinical signs of toxicity,
and food and water consumption was monitored throughout the study.
Haematological and clinical chemical parameters were examined before
treatment and at the end of the study; the animals were also examined
macroscopically for pathological changes, and samples were taken for
histopathological examination. There were no deaths and no clinical
signs of toxicity.
A number of effects were seen in animals at the high dose.
Body-weight gain and food intake were decreased, and the red blood
cell counts and packed cell volume were increased. Clinical chemistry
revealed increased plasma urea and creatinine concentrations in males
and decreased albumin in females, and urinalysis showed increased
urinary density in animals of each sex. The absolute and relative
liver weights were increased in animals of each sex at the high dose
and also in males at the intermediate dose. The absolute and relative
adrenal weights were decreased in females at the high dose. Gross
examination revealed no remarkable findings, but microscopic
examination showed treatment-related hepatocellular cytoplasmic
alterations in animals of each sex at the high dose. There was no
evidence of degeneration or necrosis. The NOEL was 600 mg/kg diet,
equivalent to 30 mg/kg bw per day (Lina et al., 1986).
8-Cyclohexadecen-1-one was tested for mutagenic activity in
S. typhimurium strains TA1535, TA1537, TA1538, TA98, and TA100 with
and without microsomal activation. There was no increase in the number
of revertants of any of the strains tested (Wilmer, 1986).
8-Cyclohexadecen-1-one was also tested for its capacity to induce
micronuclei in animals treated in vivo. Groups of 30 CD-1 mice of
each sex were given the compound by gavage at a dose of 3 or 5 ml/kg
bw (equivalent to 2.8 and 4.6 g/kg bw). Five animals of each sex were
killed at 24 and 48 h and two of each sex at 72 h after treatment.
Examination of the bone marrow showed no increase in the number of
normochromatic erythrocytes over that in control mice (Willems, 1986).
3. ESTIMATE OF DIETARY INTAKE
gamma-Cyclodextrin can be used as a carrier for flavours,
sweeteners, and colours, and it has been proposed for use in this
manner in dry mixes for beverages, soups, dressings, gravies, sauces,
puddings, gelatines, and fillings and also in instant coffee and
instant tea, coffee whiteners, compressed sweets, chewing gum,
breakfast cereals, savoury snacks, crackers, and spices and
seasonings. It is also proposed for use as a carrier for vitamins and
polyunsaturated fatty acids in dry food mixes and in dietary
supplements, as a flavour modifier in soya milk, and as a stabilizer
in bread spreads, frozen dairy desserts, baked goods, bread,
fruit-based fillings, fat-based fillings, processed cheese, and dairy
desserts.
The estimated daily intake of gamma-cyclodextrin from its use in
food has been calculated by the dietary survey approach (Amann et al.,
1998), which is based on food consumption data from the 1989-91
Continuing Survey of Food Intakes by Individuals, in which data were
collected from a representative sample of individuals residing in
households in the United States. Each individual was surveyed over
three successive one-day periods, and the foods consumed were coded
into one of about 6000 different categories. For the purposes of the
calculation, it was assumed that each food (or food component) that
contains gamma-cyclodextrin contains this substance at the highest
feasible concentration. When gamma-cyclodextrin was used as a
component of the food, the intake of that component was calculated
from data on food consumption. The estimated daily intake was
calculated for each food in which gamma-cyclodextrin may be used from
data on one- and three-day food intake. Intakes were calculated both
per capita and per user. Users were defined as individuals who
consumed a food of at least one of the categories concerned on at
least one occasion.
The largest amounts of gamma-cyclodextrin are consumed with soya
milk and dairy desserts. Relatively high intakes (> 2 g/day) also
result from its use in bread spreads and fruit-based fillings. The
mean one-day intake of gamma-cyclodextrin from all its food uses was
estimated to be 4.1 g. The 90th percentile user was estimated to
ingest about 8.8 g on any one day. The three-day averages are 4 g for
the mean intake and 7.5 g for the 90th pecentile consumer. The intake
of gamma-cyclodextrin from chewing gum was estimated from a separate
survey on chewing-gum intake in the United States, and the average
consumer was estimated to ingest about 0.07 g gamma-cyclodextrin per
day. These data represent a 'worst-case' scenario and are based on the
assumption that gamma-cyclodextrin is used simultaneously in all
possible applications and at the highest feasible concentrations. The
realistic average daily intake of gamma-cyclodextrin would therefore
be lower than the levels indicated above.
4. COMMENTS
The Committee noted that the close structural similarity between
gamma-cyclodextrin and ß-cyclodextrin allows some comparisons to be
made of the metabolism and the toxicity of these two compounds. The
metabolism of gamma-cyclodextrin is different from that of
ß-cyclodextrin, as demonstrated both in vitro and in vivo. In rats
in vivo, gamma-cyclodextrin (at single doses up to 1000 mg/kg bw) is
rapidly metabolized to glucose, presumably by the luminal and/or
epithelial enzymes of the gastrointestinal tract. In contrast to the
metabolism of ß-cyclodextrin, there was little involvement of the
gastrointestinal microflora. Similarly, in contrast to ß-cyclodextrin,
much less gamma-cyclodextrin was absorbed and only very low levels
could be detected in the urine. After intravenous injection,
gamma-cyclodextrin (at single doses up to 600 mg/kg bw) was rapidly
cleared from the blood and was excreted, largely unchanged, in the
urine. Although no studies of metabolism in humans in vivo were
available, gamma-cyclo-dextrin, unlike ß-cyclodextrin, can be readily
hydrolysed by human salivary and pancreatic amylases in vitro.
Short-term (28- and 90-day) studies of toxicity indicate that
gamma-cyclodextrin has little toxicity when given intravenously or
orally to rats or orally to dogs. After administration of a very high
dietary concentration (20%), caecal enlargement and associated changes
were seen in both species. This effect is likely to result from the
presence of a high concentration of an osmotically active substance in
the large intestine. This result suggests that the metabolism of
gamma-cyclodextrin is less efficient at doses higher than those used
in the studies of metabolism. The results of the studies in rats
treated intravenously indicate that gamma-cyclodextrin is well
tolerated, even when given systemically, and may be less toxic than
ß-cyclodextrin.
Studies conducted in rats and rabbits with gamma-cyclodextrin at
doses up to 20% of the diet did not indicate any teratogenic effects.
Similarly, the results of a battery of studies of genotoxicity were
negative. Long-term studies of toxicity, carcinogenicity, and
reproductive toxicity have not been conducted, but, given the rapid
metabolism of this substance to glucose and its lack of genotoxicity,
the Committee concluded that such studies were not required for an
evaluation.
In vitro, gamma-cyclodextrin, like ß-cyclodextrin, sequestered
components of the membranes of erythrocytes, causing haemolysis. The
threshold concen-tration for this effect was, however, higher than
that of ß-cyclodextrin. Furthermore, gamma-cyclodextrin was not
detected in blood after dietary administration of high doses to
animals in vivo.
It was considered unlikely that interaction of gamma-cyclodextrin
with lipophilic vitamins would impair their bioavailability, because
of the rapid digestion of gamma-cyclodextrin in vivo. Also, there
was no evidence that vitamin deficiency was induced in experimental
animals given high doses of gamma-cyclodextrin.
The Committee considered studies on the short-term toxicity and
genotoxicity of the enzyme, cyclodextrin-glycosyl transferase, used in
the production of cyclodextrins, and of the complexant,
8-cyclohexadecen-1-one, used to optimize formation of
gamma-cyclodextrin. The data indicated that these substances are
unlikely to be of toxicological concern in final preparations of
gamma-cyclodextrin that comply with the specifications. The Committee
also considered information on genetic modifications in the organism
used to produce the enzyme, which raised no concern.
Although no studies of human tolerance to gamma-cyclodextrin were
submitted to the meeting, the Committee was aware that such a study
was available. Although it was unable to review the data, the
Committee was reassured by the relatively low toxicity of this
compound in animals and the fact that it is less toxic than
ß-cyclodextrin, for which studies of human tolerance were available.
Also, its rapid metabolism to maltose, maltotriose, and glucose
in vitro by human salivary and pancreatic amylases supports the
conclusion that, as in laboratory animals, it would be metabolized to
innocuous metabolites before absorption by humans.
5. EVALUATION
The Committee concluded that there were sufficient data to
allocate a temporary ADI 'not specified' but that the study of human
tolerance known to have been conducted should be reviewed in order to
confirm the absence of adverse gastrointestinal symptoms at normal
levels of intake. These data should be submitted for consideration by
the Committee by 1999.
6. REFERENCES
Abdullah, M., French, D. & Robyt, J.F. (1966) Multiple attack by
alpha-amylases. Arch. Biochem. Biophys., 114, 595-598.
Amann, M., Douglass, J., Doyle, E. & Heimbach, J. (1998) Estimated
intake for gamma-cyclodextrin in the USA from food use. Unpublished
report from TAS-Environ, Arlington, Virginia, USA. Submitted to WHO by
Bioresco Ltd, Binningen, Switzerland.
Bär, R. & Ulitzur, S. (1994) Bacterial toxicity of cyclodextrins:
Luminuous Escherichia coli as a model. Appl. Microbiol.
Biotechnol., 41, 574-577.
de Bie, A.T.H.J. & van Ommen, B. (1994) Oral and intravenous
disposition study with [14C] gamma-cyclodextrin in rats. Unpublished
report No. V93.279 from TNO-CIVO Institutes, Zeist, Netherlands.
Submitted to WHO by Bioresco Ltd, Binningen, Switzerland.
de Bie, A.T.H.J., van Ommen, B. & Bär, A. (1998) Disposition of
14C-gamma-cyclodextrin in germfree and conventional rats. Regul.
Toxicol. Pharmacol., submitted for publication.
Blijleven, W.G.H. (1991). Examination of gamma-cyclodextrin for
mutagenic activity in the Ames test. Report No. V91.027. Unpublished
report from TNO-CIVO Institute, Zeist, Netherlands. Submitted to WHO
by Bioresco Ltd, Binningen, Switzerland.
Dickhaus, S. & Heisler, E. (1985) Acute toxicological study of
8-cyclohexadecenon after oral application on the rat. Unpublished
report from Pharmatox Beratung und Forschung GmbH, Sehnde, Germany.
Submitted to WHO by Bioresco Ltd, Binningen, Switzerland.
Ehling G., Donaubauer, H.H., Mayer D., Langer, K.H. & Schmidts, H.L.
(1992) gamma-Cyclodextrin; repeated-dose (3-month) intravenous
toxicity in rats. Unpublished report No 92.0516 from Hoechst AG,
Frankfurt a.M., Germany. Submitted to WHO by Bioresco Ltd, Biningen,
Switzerland.
French D. (1957) The Schardinger dextrins. Adv. Carbohydr. Chem.,
12, 189-260.
Frijlink, H.W., Eissens, A.C., Hefting, N.R., Poelstra, K., Lerk, C.F
& Meijer, D.K.F. (1991) The effect of parenterally administered
cyclodextrins on cholesterol levels in the rat. Pharm. Res., 8,
9-16.
Fuchs, H., Donaubauer, H.H., Mayer D., Langer K.H. & Krieg K. (1992)
gamma-Cyclodextrin; repeated-dose (1-month) intravenous toxicity study
in rats. Unpublished report No 91.1007 from Hoechst AG, Frankfurt,
Germany. Submitted to WHO by Bioresco Ltd, Biningen, Switzerland.
Immel, H.R. (1991) Examination of gamma-cyclodextrin in the
micronucleus test. Unpublished report No. V90.436 from TNO-CIVO
Institutes, Zeist, Netherlands. Submitted to WHO by Bioresco Ltd,
Binningen, Switzerland.
Irie, T., Otagiri, M., Sunada, M., Uekama, K., Ohtani, Y., Yamada, Y.
& Sugiyama, Y. (1982) Cyclodextrin-induced hemolysis and shape changes
of human erythrocytes in vitro. J. Pharm. Dyn., 5, 741-744.
Jonker, D. (1994) Subchronic (13-week) oral toxicity study with an
aqueous solution of alpha-cyclodextrin-glycosyl-transferase
(alpha-CGT) in rats. Unpublished report No. V94.332 from TNO Nutrition
and Food Research Institute, Zeist, Netherlands. Submitted to WHO by
Bioresco Ltd, Binningen, Switzerland.
Kondo, H., Nakatani, H. & Hiromi, K. (1990) In vitro action of human
and porcine alpha-amylases on cyclo-maltooligosaccharides.
Carbohydr. Res., 204, 207-213.
Leroy-Lechat, F., Wouessidjewe, D., Andreux, J.P., Puisieux, F. &
Duchne, D. (1994) Evaluation of the cytotoxicity of cyclodextrins and
hydroxypropylated derivatives. Int. J. Pharm., 101, 97-103.
Lina, B.A.R. (1992a) Subchronic (90-day) oral toxicity study,
including a recovery study, with alpha-cyclodextrin and
gamma-cyclodextrin in rats. Unpublished report No. V 90.401 from
TNO-CIVO Institutes, Zeist, Netherlands. Submitted to WHO by Bioresco
Ltd, Biningen, Switzerland.
Lina, B.A.R. (1992b) Sub-acute (28-day) oral toxicity study with
8-cyclohexadecenone in mice. Unpublished report No. V91.509 from TNO
Nutrition and Food Research Institute, Zeist, Netherlands. Submitted
to WHO by Bioresco Ltd, Binningen, Switzerland.
Lina, B.A.R. & Bär, A. (1998) Subchronic oral toxicity studies with
gamma-cyclodextrin in rats. Regul. Toxicol. Pharmacol., submitted
for publication.
Lina, B.A.R. & Jonker, D. (1989) Tentative (2-week) oral toxicity
study with gamma-CD in rats. Unpublished report No. V89.263/298033
from TNO-CIVO Institutes, Zeist, Netherlands. Submitted to WHO by
Bioresco Ltd, Binningen, Switzerland.
Lina, B.A.R., van Nesselrooij, J.H.J. & Beems, R.B. (1986) Sub-acute
(28-day) oral toxicity study with 8-cyclohexadecenone in rats.
Unpublished report No. V86.498/260392 from TNO-CIVO Institutes, Zeist,
Netherlands. Submitted to WHO by Bioresco Ltd, Binningen, Switzerland.
Marshall J.J. & Miwa I. (1981) Kinetic difference between hydrolyses
of gamma-cyclodextrin by human salivary and pancreatic alpha-amylases.
Biochem. Biophys. Acta, 661, 142-147.
Matsuda K., Mera, Y., Segawa, Y., Uchida, I., Yokomine, A. & Takagi,
K. (1983) Acute toxicity study of gamma-cyclodextrin (gamma-CD) in
mice and rats. Pharmacometrics, 26, 287-291.
Matsuda, K., Segawa, Y., Yokomine, A., Uchida, I., Mera, Y. &
Tokugawa, H. (1985) Biodisposition of gamma-cyclodextrin (gamma-CD) in
the rabbit and dog after intravenous administration. Oyo Yakuri
(Pharmacometrics), 30, 889-896.
Ohtani, Y., Irie, T., Uekama K., Fukunaga, K. & Pitha, J. (1989)
Differential effects of alpha-, ß-, and gamma-cyclodextrins on human
erythrocytes. Eur. J. Biochem., 186, 17-22.
Okada, Y., Kubota, Y., Koizumi, K., Hizukuri, S., Ohfuji, T. & Ogata,
K. (1988) Some properties and the inclusion behavior of branched
cyclodextrins. Chem. Pharm. Bull., 36, 2176-2185.
Prinsen, M.K. (1990) Acute eye irritation/corrosion study with
gamma-cyclodextrin in albino rabbits. Unpublished report No.
V90.405/200069 from TNO-CIVO Institutes, Zeist, Netherlands. Submitted
to WHO by Bioresco Ltd, Binningen, Switzerland.
Prinsen, M.K. (1992) Sensitization study with gamma-CD in guinea pigs
(maximization test). Unpublished report No. V92.575/352063 from
TNO-CIVO Institutes, Zeist, Netherlands. Submitted to WHO by Bioresco
Ltd, Binningen, Switzerland.
Riebeek, W.M. (1990a) Determination of the acute intravenous toxicity
of gamma-cyclodextrin in mice. Unpublished report No. V90.222 from
TNO-CIVO Institutes, Zeist, Netherlands. Submitted to WHO by Bioresco
Ltd, Binningen, Switzerland.
Riebeek, W.M. (1990b) Determination of the acute intravenous toxicity
of gamma-cyclodextrin in rats. Unpublished report No. V90.220 from
TNO-CIVO Institutes, Zeist, Netherlands. Submitted to WHO by Bioresco
Ltd, Binningen, Switzerland.
Riebeek, W.M. (1990c) Determination of the acute intraperitoneal
toxicity of alpha-cyclodextrin and gamma-cyclodextrin in rats.
Unpublished report No. V90.233 from TNO-CIVO Institutes, Zeist,
Netherlands. Submitted to WHO by Bioresco Ltd, Binningen, Switzerland.
Spanjers, M.T. (1986) Determination of the acute oral toxicity (limit
test) of 8-cyclohexadecenone in mice. Unpublished report No.
V86.455/260393 from TNO-CIVO Institutes, Zeist, Netherlands. Submitted
to WHO by Bioresco Ltd, Binningen, Switzerland.
Szejtli, J., Csherhti, T. & Szgyi, M. (1986) Interactions between
cyclodextrins and cell-membrane phospholipids. Carbohydr. Polymers,
6, 35-49.
Til, H.P. & Bär, A. (1998) Subchronic (13-week) oral toxicity study of
gamma-cyclodextrin in dogs. Regul. Toxicol. Pharmacol., submitted
for publication.
Til, H.P. & van Nesselrooij (1992) Subchronic (90-day) feeding study
with gamma-CD in dogs. Unpublished report No. V91.475 from TNO-CIVO
Institutes, Zeist, Netherlands. Submitted to WHO by Bioresco Ltd,
Binningen, Switzerland.
Verhagen, F.J.J. & Waalkens-Berendsen, D.H. (1991) Oral
embryotoxicity/teratogenicity study with alpha-cyclodextrin and
gamma-cyclodextrin in rats. Unpublished report No. V91.210 from TNO
Nutrition and Food Research Institute, Zeist, Netherlands. Submitted
to WHO by Bioresco Ltd, Binningen, Switzerland.
de Vogel, N. & van Delft, J.H.M. (1996) Chromosome aberration test
with gamma-cyclodextrin in cultured human lymphocytes. Unpublished
report No. V96.398 from TNO-CIVO Institutes, Zeist, Netherlands.
Submitted to WHO by Bioresco Ltd, Binningen, Switzerland.
Waalkens-Berendsen, D.H. & Smits-van Prooije, A.E. (1992) Final report
of an oral embryotoxicity/teratogenicity study with alpha-cyclodextrin
and gamma-cyclodextrin in New Zealand white rabbits. Unpublished
report No. V91.564 from TNO Nutrition and Food Research Institute,
Zeist, Netherlands. Submitted to WHO by Bioresco Ltd, Binningen,
Switzerland.
Willems, M.I. (1986) Evaluation of 8-cyclohexadecenone in the
micronucleus test. Unpublished report No. V86.456/260393 from TNO-CIVO
Institutes, Zeist, Netherlands. Submitted to WHO by Bioresco Ltd,
Binningen, Switzerland.
Wilmer, J.W.G.M. (1986) Examination of 8-cyclohexadecenone for
mutagenic activity in the Ames test. Unpublished report No.
V86.264/260064 from TNO-CIVO Institutes, Zeist, Netherlands. Submitted
to WHO by Bioresco Ltd, Binningen, Switzerland.
Yim, D.G., Sato, H.H., Park, Y.H. & Park, Y.K. (1997) Production of
cyclodextrin from starch by cyclodextrin glycosyltransferase from
Bacillus firmus and characterization of purified enzyme. J. Ind.
Microbiol. Biotechnol., 18, 402-405.
Yoshida, A., Arima, H., Uekama, K. & Pitha, J. (1988) Pharmaceutical
evaluation of hydroxyalkyl ethers of ß-cyclodextrins. Int. J.
Pharmacol., 46, 217-222.
