
INSECTICIDES
CYPERMETHRIN & alpha-CYPERMETHRIN
First draft prepared by
Mrs Ir. M.E.J. Pronk,
Dr G.J.A. Speijers,
Mrs M.F.A. Wouters
Toxicology Advisory Centre
National Institute of Public Health and Environmental Protection
Bilthoven, Netherlands
Dr L. Ritter
Canadian Network of Toxicology Centres
University of Guelph
Guelph, Ontario, Canada
1. Explanation
2. Biological data
2.1 Biochemical aspects
2.1.1 Absorption, distribution and excretion
2.1.2 Effects on liver enzymes
2.2 Toxicological data
2.2.1 Acute toxicity
2.2.2 Short-term toxicity studies
2.2.3 Long-term toxicity/carcinogenicity studies
2.2.4 Reproductive toxicity studies
2.2.5 Special studies on embryotoxicity and
teratogenicity
2.2.6 Special studies on genotoxicity
2.2.7 Special studies on neurotoxicity
2.2.8 Special studies on biochemistry and
electrophysiology
2.2.9 Special studies on sensitization
2.2.10 Special studies on skin and eye irritation
2.3 Observations in humans
3. Comments
4. Evaluation
5. References
1. EXPLANATION
Cypermethrin and alpha-cypermethrin are highly active synthetic
pyrethroid insecticides, effective against a wide range of pests in
agriculture, public health and animal husbandry. Cypermethrin has been
widely used throughout the world since the late 1970s while
alpha-cypermethrin has been available commercially since the mid
1980s.
Cypermethrin and alpha-cypermethrin are neuropoisons acting on
the axons in the peripheral and central nervous system by interacting
with sodium channels in mammals and insects.
Cypermethrin and alpha-cypermethrin have not been previously
evaluated by the Committee. However, cypermethrin was reviewed by the
Joint FAO/WHO Meeting on Pesticide Residues (JMPR) in 1979 and 1981
(FAO, 1980, 1982); an ADI of 0-0.05 mg/kg bw was established at the
1981 meeting. Studies reviewed in 1979 and 1981, which were not
available at the present meeting were considered in this evaluation on
the basis of the JMPR summaries.
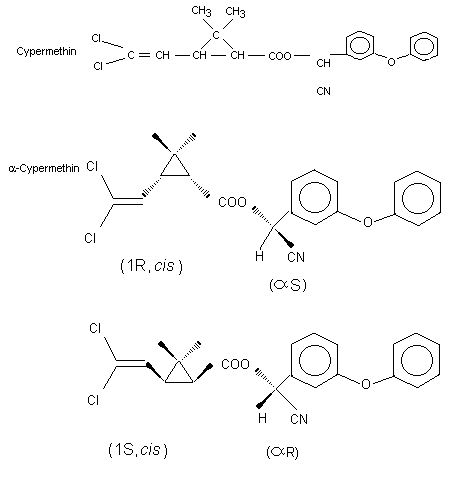
Cypermethrin consists of eight isomers, four cis and four trans
isomers, the cis isomers being the more biologically active. Depending
on the manufacturing source, the cis:trans ratio varies from 40:60 to
80:20. In the studies submitted for evaluation the cis:trans ratio was
in the range of 48:52 to 50:50. The purity varied between 92-95.1%.
alpha-Cypermethrin contains more than 90% of the insecticidally
most active enantiomer pair of the four cis isomers of cypermethrin as
a racemic mixture.
 2. BIOLOGICAL DATA
2.1 Biochemical aspects
2.1.1 Absorption, distribution and excretion
According to the JMPR (FAO, 1980, 1982) cypermethrin is readily
absorbed and rapidly eliminated via urine and faeces of mice, rats,
dogs, sheep and cows. Absorption from the gastrointestinal tract is
more rapid with the trans isomer than with the cis isomer. The highest
mean concentrations are found in body fat, liver, kidney, muscle, skin
and milk. The clearance rate from adipose tissue is slow and the
elimination half-life in rats and mice ranges from 10 to 20 and from
20 to 30 days, respectively. The data suggest a potential for
bioaccumulation in the body following continuous exposure. With
respect to the chemical and especially the isomeric complexity of the
molecule, the metabolic profile due to all of its isomers is extremely
complex. Cypermethrin is readily cleaved at the ester linkage to
produce the cyclopropane carboxylic acid and a 3-phenoxybenzoyl moiety
that is further metabolised by oxidation at the 4'position. The
resultant phenol is almost totally conjugated with sulfate. The
4'-hydroxy sulfate forms the major aryl metabolite (16%), and
3-phenoxybenzoic acid is the second most important (5%). The other
identified aryl metabolites are 3-(4-hydroxyphenoxy)benzoic acid (1%)
and the glycine conjugate, N-(3-phenoxybenzoyl) glycine (1%).
2.1.1.1 Rats
Eight female Wistar rats were orally dosed with 2.5 mg
14C-benzyl-labelled cis-cypermethrin/kg bw. They were killed in
groups of two at 8, 14, 25 and 42 days after dosing, and the
radioactivity in fat, liver and kidney was measured. Selected fat
samples were analysed for parent compound. Between days 14 and 42,
radioactivity was eliminated from the fat with a half-life of 20-25
days. Residues in liver and kidney were 30 to 40 times lower than
those in fat, but were eliminated at a similar rate. In two pooled fat
samples taken 8 and 24 days after dosing, 90-100% of the radioactivity
was in the form of the parent compound (Crawford & Hutson, 1978).
2.1.1.2 Chickens
Six Warren laying hens were treated daily for 14 days with 10 mg
14C-phenoxy-labelled cypermethrin/kg food (equivalent to 1.25 mg/kg
bw per day) in gelatin capsules in the diet. Eggs and excreta were
collected dally. The hens were killed 4.5 hours after the last dose,
and liver, muscle and fat samples were collected. Of the total
radioactivity, 95.2% was recovered. Radioactivity in eggs plateaued 8
days after the start of dosing and reached 0.05 mg/kg. Most
radioactivity was found in the yolk and was a mixture of parent
compound and material that was closely associated with neutral lipids
and phosphatidyl cholines. In tissues, highest radioactivity was found
in the liver (0.37 µg/g). This radioactivity was composed of parent
compound (0.05 µg/g) and a mixture of very polar metabolites that were
not hydrolysed to significant amounts of 3-phenoxybenzoic acid or its
4-hydroxy derivative. Fat contained 0.08 µg/g and about 60% of the
residue was present as parent compound. Residues in muscle
(0.01-0.02 µ/g) were too low for characterization (Hutson & Stoydin,
1987).
2.1.1.3 Sheep
Two male sheep were each treated dermally (single dose) with a
mixture of 962 mg 14C-cypermethrin (both cyclopropyl- and
benzyl-labelled), equivalent to 21.9 mg/kg bw in acetone. After 24
hours (sheep 1) and 6 days (sheep 2) the animals were killed, and
radioactivity in fat, muscle, liver, kidney and skin (application
area) was measured. Urine and faeces were collected up to the time the
sheep were killed. One sheep treated with acetone served as a control.
Another sheep was treated orally (single dose) with 177 mg 14C-
cypermethrin mixture in a gelatin capsule (equivalent to 3.9 mg/kg
bw). Urine and faeces were collected for 2 days and tissues were
assayed. When applied dermally radioactivity was slowly absorbed
and eliminated. Less than 0.5% was excreted in urine within 24 hours
and only 2% over a 6-day period. Faecal elimination was also slow,
0.5% of the dose being eliminated in 6 days. About 30% of the applied
radioactivity was recovered from the application areas of both sheep.
Very little radioactivity was found in the tissues. In liver, kidney,
renal fat and subcutaneous fat (other than the application area),
residues ranged from 0.09 to 0.3 µg/g and muscle samples contained
0.03 to 0.06 µg/g.
Following oral treatment, the elimination of radioactivity was
rapid, 61% of the administered radioactivity being eliminated within
48 hours. Urinary elimination comprised 41% of the dose and faecal
elimination 20.5%. Residues found in tissues were comparable to those
found after dermal application. Most of the radioactivity in the fat
samples of all sheep was found to be parent compound. In muscle, liver
and kidney samples, only a small percentage of the total tissue
radioactivity was identified as parent compound (Crawford & Hutson,
1977).
2.1.1.4 Cattle
a) Cypermethrin
Two lactating Friesian cows were fed twice daily a diet
containing 0.2 mg 14C-benzyl-labelled cypermethrin/kg feed. Cow I
was treated for 20 days and cow II for 21 days. A control cow was
maintained under identical conditions. The radioactivity in milk,
urine and faeces was measured daily and after 20-21 days of dosing the
cows were sacrificed and blood, major organs and tissues were analysed
for radioactivity. Total recovery of the radioactivity was 97.8%. The
major route of excretion was via the urine (54%) and faeces (43%).
Milk contained 0.5% of the dose. The residues in muscle, blood and
brain were less than 0.005 µg/g. Liver contained 0.006 µg/g, kidney
0.004 µg/g, bile 0.027 µg/g, renal fat 0.011 µg/g and subcutaneous fat
0.009 µg/g.
The urinary metabolites were tentatively identified as
N-(3-phenoxy-benzoyl)glutamic acid, the major metabolite, and
3-(4-hydroxyphenoxy) benzoic acid O-sulphate, the minor metabolite
(ratio 4:1). In faeces 36% was eliminated as unchanged parent compound
(Hutson & Stoydin, 1976).
Two lactating Friesian cows were fed twice daily for 7 days a
diet containing 5 mg 14C-cypermethrin (cyclopropyl-labelled)/kg
feed. A third cow was treated similarly with 14C-benzyl-labelled
cypermethrin. A control cow was maintained under identical conditions.
The radioactivity in the milk, urine and faeces was measured daily.
After 7 days of dosing the cows were killed and their blood, major
organs and tissues were analysed for radioactivity. Total recovery of
radioactivity was 92.4% for the cows dosed with cyclopropyl-labelled
cypermethrin and 89.6% for the cow dosed with benzyl-labelled
cypermethrin. The major route of excretion was similar for both label
compounds. Urinary excretion accounted for about 49% of the dose while
faecal excretion accounted for about 38%. An equilibrium between
ingestion and excretion was reached after 3-4 days. Very low levels of
radioactivity were determined in milk (0.003-0.013 µg/g), tissues
(muscle, blood, brain 0.07 µg/g, liver, kidney 0.13 µg/g and renal and
subcutaneous fat 0.10 µg/g). The urinary metabolites included the
glutamic acid conjugate of 3-phenoxybenzoic acid (68%), 3-phenoxy-
benzoylglycine (16%) and 3-phenoxybenzoic acid (9%). 3-(4-Hydroxy-
phenoxy)benzoic acid and its O-sulfate conjugate appeared to be
present in only small amounts (1%) (Crawford, 1978).
One lactating cow was administered a diet containing 10 mg
14C-benzyl-labelled cypermethrin/kg feed twice daily for 7 days.
Milk, urine and faeces were collected daily for radioassay. The cow
was killed 16 hours after the last dose and samples of fat, muscle,
liver and kidney were analysed. An untreated cow was held as control.
Total recovery was 93%. Radioactivity was rapidly eliminated in equal
proportions in the urine and faeces. Radioactivity in milk was
< 0.2%. Analysis of milk revealed that the radioactivity was due to
unchanged cypermethrin, which was located mostly in the lipophilic
components (cream or butterfat). Radioactivity in tissues was
generally in the order: liver (0.21 µg/g) > kidney (0.11 µg/g) > fat
(0.1 µg/g) > blood (0.04 µg/g) > muscle (0.01 µg/g). The residue in
fat was largely unchanged cypermethrin. Radioactivity in liver and
kidney was due mainly to N-(3-phenoxybenzoyl)glutamic acid. The
liver and kidney metabolites were hydrolysed in hot acid to 3-phenoxy-
benzoic acid and its 4'-hydroxy derivative (Croucher et al., 1980).
b) alpha-Cypermethrin
One lactating cow received orally 125 mg 14C-alpha-cypermethrin
(benzyl labelled)/dose twice daily for 4 consecutive days (target dose
250 mg/day). Another cow received the same dose of unlabelled alpha-
cypermethrin. (The overall calculated daily dietary concentrations
were 19 and 14 mg/kg, respectively). The animals were killed 6 hours
after the last dose. Urine, faeces, milk, kidneys, liver, fat and
muscle were analysed for radioactivity.
The major route of excretion of radioactivity was via the faeces,
accounting for 34% of the total administered dose. Urinary excretion
accounted for 23% of the dose and milk less than 1%. Total radioactive
residues in milk accounted for 0.014 mg/kg on day 2 and rose to
0.2 mg/kg by day 4 of dosing. The major proportion (93%) of the milk
residues was confined to the cream fraction. Residues in tissues were
highest in liver, renal fat, omental fat, subcutaneous fat and kidney
(0.56, 0.48, 0.43, 0.39 and 0.22 mg/kg, respectively). Plasma
contained 0.08 mg/kg and muscle samples contained < 0.03 mg/kg
residues.
The liver and kidney contained a range of components. The liver
extract contained at least eight metabolites with a broad range of
polarities. One component (16%) had similar chromatographic properties
to alpha-cypermethrin. The kidney extract contained at least nine
metabolites with a broad range of polarities, one component (20% of
the profile) had similar chromatographic properties to alpha-
cypermethrin. Muscle, fat and milk contained mainly a single
component (muscle 85%, fat 91% and milk 97% of the exact profile),
which in each case had similar chromatographic properties to
alpha-cypermethrin. Urinary metabolites were analysed using HPLC.
The two major components (44% and 20%) had identical chromatographic
properties to N-(3-phenoxybenzoyl)glutamic acid and
N-(3-phenoxybenzoyl)glycine, respectively. A minor component
(3%) had identical chromatographic properties to 3-phenoxybenzoic
acid (Dunsire & Gifford, 1993; Morrison & Richardson, 1994).
2.1.2 Effects on liver enzymes
Six Wistar rats/sex were randomly selected from each of the
control and 1000 mg cypermethrin/kg food group at termination of a
2-year feeding study. The activity of hepatic p-nitroanisole
o-demethylase (PNOD) was determined in each of these rats. In male
rats treated with cypermethrin, PNOD activity, expressed as per gram
of liver, was significantly increased (38%). The total PNOD activity
of liver was also increased (30%), but, owing to great inter-animal
variation, statistical significance was not reached. Treated females
exhibited increases in PNOD activity when expressed per gram of liver
(21%) and per whole liver (39%), but only the latter was significant
(Potter & McAusland, 1980).
2.2 Toxicological data
2.2.1 Acute toxicity
Acute toxicity studies of cypermethrin and alpha-cypermethrin are
summarized in Tables 1 and 2.
Cypermethrin-induced signs of toxicity were typical of
cyano-containing pyrethroid intoxication. After oral administration
signs of intoxication included sedation, ataxia, splayed gait, tip-toe
walking, with occasional tremors and convulsions. These signs of
toxicity appeared within a few hours following dosing, and survivors
recovered within 3 days. The acute toxicity after dermal
administration is of a low order (Coombs et al., 1976).
alpha-Cypermethrin-induced clinical signs, like those of
cypermethrin, are typical of cyano-containing pyrethroid. The observed
signs included ataxia, abasia, gait abnormalities, choreoathetosis,
tip-toe walking, and increased salivation, lacrimation, piloerection,
tremor and clonic convulsions. Surviving animals recovered within 7
days (Rose, 1982b, 1983).
2.2.2 Short-term toxicity studies
2.2.2.1 Mice
Groups of eight mice (CD-1)/sex received diets containing 0, 200,
400, 800, 1200 or 1600 mg alpha-cypermethrin/kg feed for 29 days. One
male fed 1600 mg/kg and one female fed 1200 mg/kg were killed in a
moribund condition. These animals showed neurological disturbances.
Mice at 1200 and 1600 mg/kg developed ungroomed coats, ataxia/abnormal
gait, over-activity or hunched posture. At 800 mg/kg some animals
had ungroomed coats and abnormal gait. Body weight (gain) was
dose-relatedly decreased in mice at 1200 and 1600 mg/kg and in females
at 800 mg/kg. A similar, but less marked effect was apparent in males
receiving 800 mg/kg and females receiving 400 mg/kg. Food consumption
was lowered in rats at 1200 and 1600 mg/kg during the first 2 weeks. A
depression in lymphocyte numbers was noted in males receiving 1600
mg/kg. ALAT and ASAT levels were increased in males at 1600 mg/kg.
Urea levels were increased in all dosed males and females, without
clear dose relation. Plasma albumin and A/G ratio were decreased in
males at 1600 mg/kg. Relative kidney weight was increased in males at
800, 1200 and 1600 mg/kg. Relative lung weight was increased in males
at 1600 mg/kg. No macroscopic or microscopic effects were seen. The
NOEL was 400 mg/kg feed, equal to 57 mg/kg bw per day (Green, 1993).
Groups of 12 mice (CD-1)/sex received diets containing 0, 50, 250
or 1000 mg alpha-cypermethrin/kg feed for 13 weeks. Four males at
1000 mg/kg died during week 12 probably due to stress of treatment and
refusal of food and water. The relative kidney weights of these
animals appeared to be slightly higher. One mouse at 250 mg/kg died.
Clinical signs included thin build, ungroomed coat, hair loss and
encrustations of the dorsal body surface in animals receiving
1000 mg/kg, ungroomed haircoat in mice at 250 mg/kg, hair loss and
encrustations in males at 250 mg/kg and hair loss in two males at
50 mg/kg. Body weight gain was markedly lower in mice at 1000 mg/kg
and lower in mice at 250 mg/kg. Food consumption was slightly higher
in mice at 1000 mg/kg. Overall food conversion efficiency of animals
receiving 1000 mg/kg (and to a lesser extent 250 mg/kg) was lower than
that of controls. In males at 1000 mg/kg, PVC, Hb, RBC, total WBC and
leucocyte counts were decreased. ASAT was increased in a dose-
dependent manner in males at 250 and 1000 mg/kg, and glucose was
decreased in males at 1000 mg/kg. Serum AP was increased in females at
1000 mg/kg. Urinary specific gravity was increased in mice at the
highest dose level. In males at 1000 mg/kg relative brain, adrenal,
heart, kidney, liver, spleen, lung and testes weight were increased.
In females relative liver weight was increased at 250 and 1000 mg/kg
and relative brain and spleen weight at 1000 mg/kg. Two males and
eleven females treated at 1000 mg/kg were considered to be emaciated
at necropsy. No histological changes were observed. Owing to the hair
loss seen at 50 mg/kg, a NOEL was not identified (Amyes et al.,
1994).
2.2.2.2 Rats
a) Cypermethrin
In a 5-week feeding study, groups of six rats/sex (Charles River)
received diets containing 0, 25, 100, 250, 750 or 1500 mg cyper-
methrin/kg feed. No effects were found in haematological, macroscopic
or microscopic examinations. In the highest dose group, rats developed
piloerection, nervousness and incoordinated movement from week
2 onwards. Body weight gain, food intake and terminal body weight
were all reduced for both sexes at 1500 mg/kg. Relative liver
weight and blood urea levels were increased at the highest dose level
(only a summary available) (Coombs et al., 1976).
Table 1. Acute toxicity of cypermethrln in animals
Species1 Route Purity2 LD50 Remark Reference
mg/kg bw
Mouse oral 98.1% 88 5% in corn oil Rose, 1982a
Mouse oral ? 82 5% in corn oil Coombs et al., 1976
Mouse oral 98.1% 1126 40% in DMSO Rose, 1982a
Mouse oral 98.1% 657 50% aq. susp. Rose, 1982a
Mouse percutan. 98.1% > 100 5% in corn oil Rose, 1982a
Rat oral ? 251 5% in corn oil Coombs et al., 1976
Rat oral 98.1% 4000 40% in DMSO Rose, 1982a
Rat oral 98.1% 3423 50% aq. susp. Rose, 1982a
Rat oral ? > 2000 techn. conc. Price, 1984
Rat percutan. ? > 2000 techn. conc. Price, 1984
Rat percutan. ? > 1600 40% in xylene Coombs et al., 1976
Rat i.p. ? approx. 40% in corn oil Coombs et al., 1976
2600
Hamster oral ? 203 5% in corn oil Coombs et al., 1976
Guinea-pig oral ? approx. 20% in corn oil Coombs et al., 1976
500
1 Both male and female animals were used in all of the studies
2 cis:trans isomer ratio 51:49; ? - unknown
Table 2. Acute toxicity of alpha-cypermethrin in animals
Species1 Route Purity LD50 Remark Reference
(%) mg/kg bw
or mg/m3
Mouse oral 96.6 35 5% in corn oil Rose, 1982b
Mouse oral 96.6 762 40% in DMSO Rose, 1982b
Mouse oral 96.6 798 50% aq. suspension Rose, 1982b
Mouse percut. 96.6 > 100 5% in corn oil Rose, 1982b
Rat oral techn. 64 50% aq. CMC Gardner, 1993
Rat oral 96.6 4000 40% in DMSO Rose, 1982b
Rat oral 96.6 > 5000 50% aq. suspension Rose, 1982b
Rat dermal techn. > 2000 undiluted Gardner, 1993
Rat inhal 95.6 1590 as aerosol Jackson, 1993
(MMAD 9 µm)
1 Both male and female animals were used in all of the studies
Groups of 12 rats/sex (Wistar) were fed diets containing 0 (24
rats/sex), 25, 100, 400 or 1600 mg cypermethrin/kg feed for 91-95
days. There were no deaths, no clinical signs and no macroscopic or
microscopic changes. In the highest dose group a reduced body weight
gain was observed. During the first week reduced food consumption was
observed in both sexes at 1600 mg/kg. This could be attributed to the
palatability of the diet. During week 13, females at 1600 mg/kg had
reduced food intake. Males at 1600 mg/kg showed decreases in Hb, MCV
and eosinophil counts, and an increase in prothrombin time. In males
at 400 mg/kg there was also a decrease in eosinophil numbers. The mean
plasma urea concentration in the 1600 mg/kg groups was increased.
Relative liver weight was increased in males and females at 1600 mg/kg
and in males at 400 mg/kg. Relative kidney weight was increased in
males at 1600 mg/kg. The NOEL was 100 mg/kg feed, equivalent to
5 mg/kg bw per day (Pickering, 1981).
b) alpha-Cypermethrin
Groups of 10 Wistar rats/sex received diets containing 0 (20
rats/sex), 20, 100, 200, 400 or 800 mg alpha-cypermethrin/kg feed for
5 weeks. Observations included mortality, clinical signs, body weight
and food consumption, haematology, clinical chemistry, urinalysis,
organ weight, and macroscopic and microscopic lesions.
One male at 800 mg/kg died unrelated to treatment and two males
at 800 mg/kg were killed, owing to signs of severe neurological
disturbance. Both sexes at 800 mg/kg produced abnormal gait and
increased sensitivity to noise. Abnormal gait was seen in one male at
400 mg/kg. The mean body weights and food intake of both males and
females fed 400 or 800 mg/kg were significantly lower than those of
controls. In both males and females at 800 mg/kg and in males at
400 mg/kg prothrombin time was increased. In males at 800 mg/kg the
kaolin-cephalin coagulation time was increased and the percentage of
polymorphic neutrophils was increased at 800 mg/kg. In females
platelet count, total leucocyte count and absolute values of
polymorphic neutrophils and lymphocytes were increased, but the Hb and
haematocrit values were decreased. The MCV was decreased at 200, 400
and 800 mg/kg but not in a dose-related manner. Relative brain, liver
and kidney weights were increased in males fed 800 mg/kg and relative
brain and liver weights were increased in males fed 400 mg/kg. In
females at 800 mg/kg relative brain, liver and kidney weights were
increased. One male at 800 mg/kg that was removed from the study
showed scanty axonal lesions of the sciatic nerves. The NOEL was
200 mg/kg feed, equivalent to 10 mg/kg bw per day (Thorpe, 1982).
In a range finding study groups of five male and five female rats
were fed diets containing a-cypermethrin at concentrations of 0, 50,
200, 800 or 1200 mg/kg feed for up to 6 weeks. No effects were found
on food consumption, haematology and clinical chemistry and
microscopy. All rats at 1200 mg/kg and all males at 800 mg/kg were
killed during weeks 2 to 4 because of severe clinical signs. These
signs included high stepping, splayed gait, abasia and hyper-
sensitivity. Cachexia was seen in extreme cases. There were no
significant changes in fore or hind limb grip strength or hind limb
landing foot splay. Female rats fed 800 mg/kg had a lower mean body
weight and food intake compared to controls. These females also had a
lower leucocyte count. Microscopic examination revealed lymphocytolysis
and iymphocyte depletion of the cortical region of the thymus in males
fed 800 mg/kg and males and females at 1200 mg/kg (Fokkema, 1994a).
Groups of 30 male and 30 female Wistar rats received diets
containing 0 (60 rats/sex), 20, 60, 180 or 540 mg alpha-
cypermethrin/kg feed for 90 days. After 6 weeks 10 rats/sex
(controls 20/sex) were sacrificed for interim examinations. Three
males at 540 mg/kg developed an abnormal gait consisting of splayed
hind limbs. Skin sores and fur loss were observed in all males with
the highest incidence at 540 mg/kg and in females at 0 and 540 mg/kg.
Two males, one from controls and one at 540 mg/kg were removed because
of severe skin lesions. Reduced body weight and reduced food
consumption were seen in rats at 540 mg/kg. During the second part of
the study, body weight gain was also reduced in males at 60 and
180 mg/kg, but not in a clearly dose-related manner. Food consumption
was lower at 60 mg/kg than at 180 mg/kg. At termination the Hb value
was decreased in males and females at 540 mg/kg. MCV and MCHC were
decreased in females at 540 mg/kg. Platelet counts were increased in
males and females at 540 mg/kg. The number of lymphocytes was
increased and the number of eosinophils was decreased in males at
540 mg/kg. Urea concentration was increased in females at 540 mg/kg.
AP was decreased in females at 180 and 540 mg/kg. Urinary volume was
decreased in females at 540 mg/kg and specific gravity was increased
in males and females at 540 mg/kg. In females fed 540 mg/kg, relative
spleen, heart and brain weights were increased. In both sexes at
540 mg/kg relative kidney weight was increased and relative liver
weight was increased in both sexes at 180 and 540 mg/kg. Relative
testis weight was increased in males at 540 mg/kg. Histopathological
investigations showed a solitary form of axonal degeneration,
affecting the fibre only, in the sciatic nerve, in two males fed
540 mg/kg, but there were no clinical signs of toxicity. The NOEL was
60 mg/kg feed, equivalent to 3 mg/kg bw per day (Clark, 1982).
2.2.2.3 Rabbits
Occluded dermal applications of 0, 2, 20 or 200 mg cyper-
methrin/kg bw in PEG 300 were made to abraded and intact skin of
groups of 10 male and 10 female NZW rabbits/sex. The applications were
made for 6 h/day, 5 days/week for 3 weeks. No effects were observed on
haematology, clinical chemistry or following macroscopic or micro-
scopic examination. Slight to severe skin irritation was observed
in rabbits at 200 mg/kg bw and slight to moderate skin irritation was
observed in rabbits receiving 2 and 20 mg/kg bw. Decreased food
consumption and body weight gain was observed in rabbits treated with
200 mg/kg bw. Absolute and relative gonad weights were reduced in male
rabbits at 200 mg/kg bw. The NOEL was 20 mg/kg bw (Henderson &
Parkinson, 1981).
2.2.2.4 Dogs
a) Cypermethrin
In a 5-week feeding study, groups of three beagle dogs/sex
received diets containing 0, 15, 150 or 1500 mg cypermethrin/kg feed.
No effects were found on body weight, food consumption, ophthalmo-
scopy, haematology, clinical chemistry, macroscopy, liver weight or
following microscopic examination. In the highest dose group dogs
developed signs of intoxication, including apprehension, diarrhoea
and vomiting, licking and chewing of the paws, whole body tremors, a
stiff exaggerated hind leg gait, ataxia, inappetence, and decreased
body weight gain. Relative thyroid weight and blood urea levels were
increased and blood glucose levels were decreased (only a summary
available) (Coombs et al., 1976).
Groups of beagle dogs (four/sex) were fed cypermethrin (purity
98%) in the diet at concentrations of 0, 5, 50, 500 or 1500 mg/kg feed
for 13 weeks. No mortality occurred. Dogs at 1500 mg/kg developed
diarrhoea, anorexia and tremors as well as ataxia, incoordination and
hyperaesthesia. Also a reduced food intake and body weight gain was
seen. Due to the clinical signs two dogs/sex at 1500 mg/kg had to be
killed. Minor variations in haematology were observed; the kaolin-
cephalin clotting time was consistently lower throughout the study in
female dogs at 500 mg/kg. However, because of the lack of a dose-
response relationship, the variability of this parameter throughout
the study, and the fact that there were no changes in the prothrombin
time, this finding is not considered to be relevant. No effects were
seen on clinical chemistry parameters or on organ weights. Microscopic
examination of tissues and organs revealed a non-specific focal
bronchopneumonia in the lungs of the dogs at 1500 mg/kg. A pink colour
of the optic disc was noted in ophthalmological examinations. The NOEL
was 500 mg/kg feed, equivalent to 12.5 mg/kg bw per day (Buckwell &
Butterworth, 1977).
A further experiment was undertaken to determine the cause of the
pink discoloration, which occurred in all groups. Cypermethrin was fed
to two groups of three male dogs at concentrations of 0 or 500 mg/kg
food for 13 weeks. Specific ophthalmological examinations were made to
evaluate the degree of coloration of the optic disc. At the end of 13
weeks, there were no consistent differences between the colour of the
optic discs of the treated dogs and the controls (Buckwell &
Butterworth, 1977).
b) alpha-Cypermethrin
This study was conducted in two parts.
In the first part one male and one female beagle dog were fed
alpha-cypermethrin in the diet at concentrations of 200 mg/kg feed for
7 days, 400 mg/kg feed for 2 days and 300 mg/kg feed for 7 days. Due
to severe intoxication of the animals receiving 400 mg/kg, dosing was
discontinued after 2 days. The animals were fed the control diet and
in week 3 treatment was commenced at 300 mg/kg. After the third week
the dogs were killed.
Clinical signs including ataxia, body tremors, subdued behaviour,
head nodding, food regurgitation, diminished response to stimuli and
inflammation of gums and tongue were obtained when dogs were dosed
with 300 and 400 mg/kg. Body weight loss was observed at 300 mg/kg.
Examination of the cellular composition of blood and the chemical
components of plasma showed no abnormalities, and no macroscopic
changes were observed.
In the second part one male and one female beagle dog were fed
alpha-cypermethrin in the diet at 300 mg/kg food for 3 days (male dog)
or 4 days (female dog), and then 250 mg/kg food for 7 days. At
300 mg/kg the same effects were obtained as above. At 250 mg/kg only
the female dog developed the clinical signs shown by the dogs treated
with 300 and 400 mg/kg (Greenough & Goburdhun, 1984).
Groups of four beagle dogs/sex received diets containing dose
levels of 0, 30, 90 or 270 (six dogs/sex) mg alpha-cypermethrin/kg
feed for 13 weeks. No effects on mortality, body weight, food
consumption, ophthalmoscopy, haematology, clinical chemistry,
macroscopy, organ weights or microscopy were seen. One female at
270 mg/kg was killed, because of severe head and body tremors, ataxia,
poor limb coordination, inflamed gums and elevated temperature. All
dogs at 270 mg/kg developed marked clinical signs, including body
tremors, head nodding, lip licking, subduedness, ataxia, agitation and
high stepping gait. The NOEL was 90 mg/kg, equivalent to 2.25 mg/kg bw
per day (Greenough et al., 1984).
Four groups of beagle dogs (four/sex) received via the diet 0,
60, 120 or 240 mg alpha-cypermethrin/kg feed dally for 52 weeks. No
effects were observed on body weight, food consumption, ophthalmo-
scopy, haematology, clinical chemistry, urinalysis or organ weights,
and no changes were observed in macroscopic or microscopic examinations.
Two males at 240 mg/kg developed skin reddening on the tail. Abdominal
skin reddening and alopecia were seen in another male at 240 mg/kg and
one female at 120 mg/kg. The tail reddening caused obvious irritation
and resulted in ulceration and necrosis in one male leading to
amputation of part of the tail. The NOEL was 60 mg/kg feed, equivalent
to 1.5 mg/kg bw per day (Dean & Jackson, 1995).
2.2.3 Long-term toxicity/carcinogenicity studies
2.2.3.1 Mice
Groups of mice (70/sex, SPF-Swiss-derived) received diets
containing 0 (2 groups), 100, 400 or 1600 mg cypermethrin/kg feed for
up to 101 weeks. Ten mice/sex were killed after 52 weeks for interim
necropsy. Observations included mortality, clinical signs, body weight
gain, food consumption, haematology, clinical chemistry, organ
weights, and macroscopy and microscopy. Body weight gain of both males
and females at 1600 mg/kg was reduced when compared to the combined
control groups. Several haematological changes, consistent with mild
anaemia, were found in the 1600 mg/kg group at the interim kill, but
not at termination. At interim kill and at termination, an increase in
thrombocytosis and absolute and relative liver weight was seen in
males at 1600 mg/kg. An increase in the incidence of benign
alveologenic tumours was observed in females at 1600 mg/kg, but was
within historical control incidence. In this study the NOEL was
400 mg/kg feed, equal to 57 mg/kg bw per day (Lindsay et al., 1982).
2.2.3.2 Rats
Groups of 48 Wistar rats/sex received diets containing 1, 10, 100
or 1000 mg cypermethrin/kg feed for 2 years. The control group
consisted of 96 rats/sex and was fed untreated diet for 2 years. After
6 and 12 months six rats/sex were sacrificed and after 18 months 12
rats/sex were sacrificed (controls, respectively, 12 and 24 rats/sex).
The only effects observed were reductions in body weight and food
consumption in males and females at 1000 mg/kg. No dose-related
effects were observed on mortality, clinical chemistry, haematology,
clinical chemistry, organ weights, macroscopy or microscopy. No
difference between control and treated groups were found in the
sciatic nerves. There was no increase in compound-related tumours. The
NOEL was 100 mg/kg feed, equivalent to 5 mg/kg bw per day (McAusland
et al., 1978).
2.2.3.3 Dogs
Groups of four beagle dogs/sex (5´-7 months of age) were fed
diets containing 0, 3, 30, 300 or 1000 mg cypermethrin/kg food for 2
years. Groups of four dogs/sex were allocated to a satellite study and
received a diet containing 0, 300 or 1000 mg/kg cypermethrin (data
concerning this satellite group have only been included in the report
to aid interpretation of the results of the main study). Due to severe
signs of intoxication observed at the 1000 mg/kg level, the
concentration was reduced to 750 mg/kg at week 4 of the study and,
when signs of intoxication persisted during weeks 6-8, animals in the
high-dose group were fed a control diet for 10 days to allow them to
recover. Following the 10 days of control diet, the dogs were fed
cypermethrin at a concentration of 600 mg/kg food for the remainder of
the study. No effects were observed on ophthalmoscopy, clinical
chemistry, organ weights, macroscopy or microscopy. No abnormalities
were found in the sciatic nerves, brain or spinal cord. One male dog
in the satellite group fed 1000 mg/kg convulsed and died. Signs of
intoxication appeared within 24 hours of the initiation of dosing and
consisted of licking and chewing of the paws, a stiff high stepping
gait, whole body tremors, head shaking, incoordination, ataxia and, in
some cases, convulsions. These signs were observed at 1000 mg/kg and
to a lesser extent at 750 mg/kg, but not at 600 mg/kg. The body
weights of male dogs in the highest dose group were significantly
lower than the controls, probably due to initial reduction in food
consumption observed at 1000 mg/kg. In the satellite group no effect
on body weight gain was seen. Apart from some occasionally decreased
sodium levels in the males given the highest dose, no consistent
haematological changes were seen. The NOEL was 300 mg/kg feed,
equivalent to 7.5 mg/kg bw per day (Buckwell, 1981).
2.2.4 Reproductive toxicity studies
Groups of 30 rats/sex (Wistar) received diets containing 0, 10,
100 and 500 mg cypermethrin/kg feed for 5 weeks prior to mating and
then throughout pregnancy and lactation for three successive
generations. Two litters were bred per generation. The first litters
were discarded at weaning. Males and females from the second litter
were randomly selected to breed the next generation. A significant
reduction in body weight gain was seen in the male and female parent
rats receiving 500 mg/kg in all three generations. This was correlated
with a reduction in food consumption. Litter size was reduced at
500 mg/kg in the F1a litter at birth and after 7 and 21 days. Litter
weights were reduced at 500 mg/kg in the F1a, litters on days 7, 14
and 21 of lactation. No other effects on fertility or reproduction
parameters were found. The NOEL for maternal and reproduction toxicity
was 100 mg/kg feed, equivalent to 5 mg/kg bw per day (Hend et al.,
1978; Fish, 1979; Thorpe, 1985).
2.2.5 Special studies on embryotoxicity and teratogenicity
2.2.5.1 Rats
a) Cypermethrin
Groups of 25 pregnant female rats (Sprague-Dawley) received by
gavage 0, 17.5, 35 or 70 mg cypermethrin/kg bw per day in corn oil
during days 6 to 15 of gestation. The females were sacrificed on day
21 of gestation for examination of their uterine contents. One female
at 70 mg/kg bw per day was found dead and one female at 70 mg/kg bw
per day was killed for ethical reasons following severe convulsions.
Eleven out of 25 females at the 70 mg/kg bw per day group showed
neurological disturbances (ataxia, convulsions, hypersensitivity to
noise). A dose-related reduction in body weight gain was observed in
the groups given 35 and 70 mg/kg bw per day. There were no indications
of any embryotoxic or teratogenic effects. The NOEL for maternal
toxicity was 17.5 mg/kg bw per day and the NOEL for embryotoxicity was
70 mg/kg bw per day (Tesh et al., 1978).
b) alpha-Cypermethrin
In a range-finding study, five pregnant female Sprague-Dawley
rats received daily, by gavage, 0, 3, 9, 15 or 18 mg alpha-cyper-
methrin/kg bw in corn oil during days 6-15 of gestation. Maternal
body weights, food consumption and clinical observations were
recorded. On day 20 of gestation the females were killed and the
fetuses were weighed, sexed and externally examined. Four dams at
18 mg/kg bw per day and one dam at 15 mg/kg bw per day showed hindlimb
splay and unsteady gait during dosing. Mean body weight gain was
reduced in a dose-related manner at 9, 15 and 18 mg/kg bw per day and
at 15 and 18 mg/kg bw per day food consumption was reduced. No other
treatment-related abnormalities were observed (Irvine & Twomey, 1994).
In another study, groups of 24 pregnant female Sprague-Dawley
rats received by gavage 0, 3, 9 or 18 mg alpha-cypermethrin (purity
95.6%)/kg bw per day in corn oil during days 6-15 of gestation.
Following marked clinical signs of toxicity the dose level of 18 mg/kg
bw per day was lowered to 15 mg/kg bw per day on day 10 of gestation.
Clinical signs, body weights and food consumption were recorded. On
day 20 of gestation the females were killed and necropsied. The
fetuses were weighed, sexed and examined for external, visceral and
skeletal abnormalities.
Females at 18 mg/kg bw per day showed unsteady gait,
piloerection, limb splay and hypersensitivity to sound. After
reduction of the dose level the signs were similar but less marked.
After treatment with 18/15 mg/kg bw per day a lowered body weight gain
and food consumption was seen. At 9 mg/kg bw per day a slight body
weight reduction was seen. Mean fetal weights were slightly reduced at
18/15 mg/kg bw per day. There was no indication for teratogenicity.
The NOEL for maternal and fetal toxicity was 9 mg/kg bw per day
(Irvine, 1994c).
2.2.5.2 Rabbits
a) Cypermethrin
In a range-finding study, groups of four female pregnant rabbits
(NZW) received during days 6 to 18 of gestation by gavage 0, 25, 50,
100 or 120 mg cypermethrin/kg bw per day in corn oil. The dams were
sacrificed on day 29 of gestation. No adverse effects were seen in the
mothers and fetuses (Tesh et al., 1984a).
Groups of 16 pregnant NZW rabbits received by gavage 0, 20, 50 or
120 mg cypermethrin/kg bw per day in corn oil during days 6 to 18 of
gestation. The dams were killed on day 29 of gestation. One control
female, three females receiving 20 mg/kg bw per day and two females in
each of the groups receiving 50 and 120 mg/kg bw per day were killed
for ethical reasons. Necropsy revealed evidence of respiratory tract
infection and/or gastrointestinal tract infection not related to the
substance. Two females at 20 mg/kg bw per day and two females at
120 mg/kg bw per day aborted during the post-treatment phase of the
investigation. The number of implantations, live young and
resorptions, pre- and post-implantation losses, and fetal and
placental weights were unaffected by treatment. There was no
indication for embryotoxicity or teratogenicity. The NOEL for
embryo-toxicity was 120 mg/kg bw per day (Tesh et al., 1984b, 1988).
Groups of pregnant rabbits (20 rabbits/group, 30 rabbits were
used as an additional control group) were administered cypermethrin
dissolved in corn oil at dose levels of 0, 3, 10 or 30 mg/kg bw per
day orally from days 6 to 18 of gestation. On day 28 of gestation the
rabbits were killed and examination was made of live fetuses, dead
fetuses, resorption sites and corpora lutea. Live fetuses were
maintained for 24 hours to assess viability. Fetuses were also
examined for gross somatic and skeletal deformities. There was no
significant mortality or difference in weight gain during the period
of gestation. There were no significant differences between control
and test groups with respect to pregnancy, fetal death and survival.
Although a wide range of skeletal and visceral abnormalities was found
in the course of the study, there were no differences between control
and test groups with respect to abnormalities. It was concluded that
oral dosing up to 30 mg/kg bw during the major period of organogenesis
resulted in no teratogenic effects in offspring (FAO, 1980).
b) alpha-Cypermethrin
In a range finding study groups of five mated female NZW rabbits
received by gavage 0, 5, 15, 25 or 30 mg a-cypermethrin/kg bw per day
as solutions in corn oil, during days 7-19 of gestation. On day 28 of
pregnancy the females were killed and a necropsy was performed. The
fetuses were weighed, sexed and externally examined. One female each
at 15 and 25 mg/kg bw per day was killed prematurely. At 25 and
30 mg/kg bw per day marked reductions in body weight and food
consumption were seen. There was no indication for either
embryotoxicity or teratogenicity (Irvine, 1994a).
In another study, groups of 16 pregnant NZW rabbits received by
gavage 0, 3, 15 or 30 mg alpha-cypermethrin/kg bw per day in corn oil
during days 7-19 of gestation. Maternal clinical signs, body weight
and food consumption were recorded. The females were killed on day 28
of pregnancy. The uterus was weighed and the numbers of corpora lutea,
implantations and live fetuses were counted. The fetuses were weighed,
sexed and examined for external, visceral and skeletal abnormalities.
Two control females, three at 15 mg/kg bw per day and two at
30 mg/kg bw per day were killed, because of severe weight loss and low
food consumption. One female at 15 mg/kg bw aborted on day 28. In all
groups, including controls, there was a similar mean body weight loss
after the onset of dosing, which continued until day 11. At 30 mg/kg
bw per day there was a further reduction in mean body weight gain
towards the end of the dosing period. Food consumption reflected the
changes in mean body weight gain. The NOEL for maternal toxicity was
3 mg/kg bw per day and the NOEL for embryotoxicity was 30 mg/kg bw.
There was no indication for teratogenicity (Irvine, 1994b).
2.2.6 Special studies on genotoxicity
Results of genotoxicity studies carried out cypermethrin and
alpha-cypermethrin are summarized in Tables 3 and 4.
2.2.7 Special studies on neurotoxicity
2.2.7.1 Rats
a) Cypermethrin
Groups of 6 or 12 rats/sex were administered single oral doses of
100, 200 or 400 mg cypermethrin/kg bw (purity 97%) as a 5% dispersion
in corn oil. The observation period was 9 days. The rats were then
killed and examined histologically. All rats showed signs of
intoxication. At 400 mg/kg bw, within 4 hours of dosing, rats
developed signs of intoxication, including coarse tremors, spasmodic
movements of the body and tail and bleeding from the nose. Tip-toe
walking was also seen in some rats. All animals, except one, were
killed, owing to the severity of the signs. Histological examination
revealed swelling of the myelin sheaths and breaks of some of the
axons of the sciatic nerves. At 200 mg/kg bw similar effects were
observed; eight rats of each sex died or were killed within 48 h of
dosing. The remaining four rats survived the observation period. At
100 mg/kg bw all animals survived the 9 days. One female out of 12
showed minimal lesions in the sciatic nerve in this group. A NOEL
could not be determined (Carter & Butterworth, 1976).
In a neuromuscular dysfunction test, Wistar rats (10/sex) were
treated by gavage with 0, 25, 50, 100, 150 or 200 mg cypermethrin/kg
bw per day in DMSO for 7 consecutive days. The rats were killed 3-4
weeks after the start of dosing, and right and left sciatic/posterior
tibial nerves were analysed. Neuromuscular function was assessed by
means of the inclined plane test and peripheral nerve damage by
reference to ß-glucuronidase and ß-galactosidase activity increases in
nerve tissue homogenates.
Table 3. Results of mutagenicity assays on cypermethrin
Test system Test object Concentration Purity Results References
(%)
In vitro
Gene mutations S. typhimurium 0.2-2000 93.5 negative2 Brooks,
assay TA1525, TA100, µg/plate1 1980
TA1538, TA98, Dean,
TA1537 1981
Gene mutations E.coli WP2 or 0.2-2000 93.5 negative2 Dean,
assay WP2 uvrA µ/plate1 1981
Mitotic gene Saccharomyces 0.01-5.0 93.5 negative Dean,
conversions cerevisiae JD1 mg/litre1 1981
assay
Host-mediated S. cerevisiae 25 and 50 93.5 negative Brooks,
assay JD1 mg/kg bw 1980
in mice
Cell BHK 21/C113 31.25-250 93.5 negative Dean,
transformation cells µg/ml1 1981
assay
Chromosomal RL4 liver cells 7.5-30 µg/ml 93.5 negative Dean,
aberrations 1981
assay
Table 3. Results of mutagenicity assays on cypermethrin (cont'd).
Test system Test object Concentration Purity Results References
(%)
In vivo
Chromosome Chinese hamster 2x oral dose of ? negative Dean,
aberration 20 or 40 mg/kg 1977
assay in bone bw (2 successive
marrow days)
Dominant mouse single oral dose ? negative Dean
lethal assay of 6.25, 12.5 or et al.,
25 mg/kg bw or 1977
5 daily doses of
2.5 or 5 mg/kg
bw per day
Host-mediated mouse/ orally 0, 25, 50 ? negative JMPR,
assay S. cerevisiae mg/kg 1979
DNA damage CD rats m 100 mg/kg, f 150 ? negative Creedy &
assay mg/kg; 1, 4, 16 Wooder,
hour exposure 1977
Wistar rats m 300 mg/kg; 1, 4, negative Creedy &
16 hour exposure Wooder,
f 450 mg/kg, 1 hour 1977
exposure f 337.5
mg/kg, 4 and 16
hour exposure
1 with and without metabolic activation
2 at 200 and 2000 µg/plate, formation of visible droplets in the top agar was seen
Table 4. Results of mutagenicity assays on alpha-cypermethrin
Test system Test object Concentration Purity Results References
(%)
In vitro
Gene mutations S. typhimurium 31.5-5000 95.6 negative Brooks &
assay1 TA98, TA100, µg/ml (no toxicity) Wiggins,
TA1535, TA1537 1992
TA1538
E. coli WP2 uvrA
Gene mutations S. cerevisiae 31.25-4000 95.8 negative Brooks,
assay1 XV185-14C µg/ml 1984
Gene mutations1 L5178Y mouse 3.3-50 µg/ml 95.4 negative Vanderwaart,
assay lymphoma cells 1994
Chromosomal human peripheral -act: 93.75-1000 95.6 negative Brooks &
aberrations lymphocytes µg/ml precipitation Wiggins,
assay1 +act: 125-1000 was seen 1993
µg/ml
In vivo
Chromosomal rat femoral single oral dose 95.8 negative Clare &
aberrations bone marrow 2-8 mg/kg3 Wiggins,
1984
Table 4. Results of mutagenicity assays on alpha-cypermethrin (cont'd).
Test system Test object Concentration Purity Results References
(%)
Micronucleus mouse single oral dose 95.4 negative Vanderwaart,
assay 1-10 mg/kg bw 1995
Alkaline rat single oral dose 96.5 negative Wooder,
elution 40 mg/kg, 6 1981
analysis hours exposure
assay2
1 With and without metabolic activation
2 The effect of alpha-cypermethrin on the integrity of rat liver DNA was investigated
by this method
3 Initially doses of 10, 20 or 40 mg/kg were used, but the animals exhibited severe
signs of toxicity and the experiment was terminated at these doses. Surviving
females were evaluated for chromosomal damage and none was observed
At 200 mg/kg bw per day 50% of the males and 62.5% of the females
died, at 150 mg/kg bw per day two females and one male died and at
100 mg/kg bw per day one female died but no males. No mortalities
occurred in the other groups. A dose-related (in severity and
duration) increase was seen in clinical signs at doses > 100 mg/kg bw.
These signs included salivation, ataxia, splayed hind limb gait,
hyperexcitability to auditory stimuli, tremor and choreoathetosis. At
the same dose levels body weight gain was also reduced.
A dose-related transient functional impairment was found in rats
treated with cypermethrin in the inclined plane test. This effect was
maximal at the end of the 7-day subacute dosing regimen. At doses
which caused mortality, significant increases in ß-glucuronidase and
ß-galactosidase were found 3-4 weeks after the start of dosing in the
distal portion of the sciatic/posterior tibial nerves. There was no
direct correlation between the time course of the neuromuscular
function and the neurobiochemical changes. The NOEL was 50 mg/kg bw
per day (Rose & Dewar, 1983).
The effects of some pyrethroids, including cypermethrin, on
amplitude and pre-pulse inhibition of the acoustic startle reflex were
studied in male Wistar rats. The pyrethroids were suspended in corn
oil. Cypermethrin was given orally at dose levels of 0, 0.5, 1 or
2 mg/kg bw to 12 males. Each animal received all doses of cyper-
methrin. The intersession interval was one week. Animal behaviour
was observed by the experimenter before and after the test session for
a period of 10 minutes and the effects of the pyrethroids on overt
behaviour were measured by scoring the presence of pawing and
salivation, burrowing, hyperactivity, hyperreactivity to an external
stimulus, fine tremors of low intensity and fluid loss. Neither
cypermethrin nor the other pyrethroids tested affected the amplitude
or the latency of the startle reflex (Hijzen et al., 1988).
b) alpha-Cypermethrin
The acute neurotoxicity of alpha-cypermethrin was studied in
Crl:CD:BR rats in two separate acute studies, each using four groups
of ten animals/sex or five rats per sex (additional study). The groups
received a single dose of 0, 4, 20 or 40 mg alpha-cypermethrin/kg bw
in corn oil. During the 14-day observation period, clinical signs and
body weight were analysed. In the main study a detailed clinical
assessment for neurotoxicological effects was performed. This included
a functional observational battery (FOB) and measurements of fore and
hind limb grip strength, hindlimb landing foot splay and motor
activity. In each study five rats/sex were killed on day 15 and brain,
eyes, muscle, nerves, spinal cord and spinal ganglia were analysed.
One male rat in each of the 20 and 40 mg/kg bw groups of the
additional study was found dead on the day after dosing. Clinical
signs were seen in male rats dosed with 20 and 40 mg/kg bw. The signs
(similar in both studies) developed between 3 to 8 hours after dosing
and resolved by three days after dosing. The signs included abnormal/
splayed gait, thrashing, prostration, vocalization, piloerection,
hunched posture, unkempt appearance, soiled/stained body areas and
diarrhoea. The signs in females were similar but lower in frequency.
In addition to these signs there were also isolated cases of
twitching, tremors, abasia, hypersensitivity, pale eyes, soft faeces
and thinning of the fur.
During FOB conducted 5 hours after dosing, gait abnormalities and
clinical signs of increased reactivity were seen in most male rats
dosed with 20 and 40 mg/kg bw. In females the signs were less
frequent. In the 20 and 40 mg/kg bw groups there was a increase in
very slight to slight sporadic fibre degeneration in the sciatic
nerve. The changes were more frequent in the proximal than in the
distal part of the nerve. The NOEL was 4 mg/kg bw (Fokkema, 1994b).
c) Cypermethrin and alpha-cypermethrin
This experiment was performed in two phases. The first phase was
conducted to determine the time course for neurochemical changes in
Wistar rats occurring in the sciatic/posterior tibial nerve (SPTN),
trigeminal nerve and trigeminal ganglion following treatment with
cypermethrin for 5 days/week for 4 weeks. Cypermethrin was
administered at 150 mg/kg bw per day in DMSO (reduced to 100 mg/kg bw
per day in arachis oil, after 10 doses, because of mortality),
alpha-Cypermethrin was dosed at 37.5 mg/kg bw per day in DMSO (also
reduced after 10 days to 25 mg/kg bw per day in arachis oil). Five
animals per sex, treated with either cypermethrin and alpha-cyper-
methrin, were killed at 2, 3, 4, 5, 6, 8, 10 or 12 weeks and examined.
Dosing resulted in the death of 56% of the cypermethrin-treated
animals and 21% of the alpha-cypermethrin-treated animals. The most
frequent signs of intoxication included abnormal gait, ataxia,
lethargy, chromodacryorrhoea, salivation and hypersensitivity to
sensory stimuli. The ß-glucuronidase and ß-galactosidase activities in
the SPTN were increased at 5, 6 and 8 weeks, when compared to
controls. The increase was maximal after 5 weeks, and after 12 weeks
was comparable to controls. No significant enzyme changes were found
in the trigeminal ganglia and trigeminal nerve of treated animals.
Phase 2 was conducted to establish the dose level which did not
cause peripheral nerve degeneration in the SPTN, trigeminal nerve and
ganglia. Groups of 10 rats/sex were dosed with 37.5, 75 or 150 mg
cypermethrin/kg bw per day in DMSO or 10, 20 or 40 mg alpha-cyper-
methrin/kg bw per day in DMSO, 5 days/week for 4 weeks. A control
group of 10 animals was used.
Signs of intoxication similar to those reported in phase 1 were
seen at the highest dose levels. A large increase in ß-glucuronidase
and ß-galactosidase activities in the SPTN was seen at 150 mg/kg bw
per day cypermethrin and 40 mg/kg bw per day alpha-cypermethrin. In
the groups administered 75 mg/kg bw per day cypermethrin or 20 mg/kg
bw per day alpha-cypermethrin a small increase in ß-galactosidase was
found in both the distal and proximal sections of the SPTN. The
magnitude of the enzyme changes was similar to those of phase 1.
Significant enzyme changes were also found in the trigeminal ganglia
and to a lesser extent in the trigeminal nerve of the groups
administered 75 or 150 mg/kg bw per day cypermethrin and 20 or
40 mg/kg bw per day alpha-cypermethrin. The NOELs were 37.5 mg/kg bw
cypermethrin and 10 mg/kg bw alpha-cypermethrin (Rose, 1983).
2.2.7.2. Chickens
In a delayed neurotoxicity study, six adult domestic hens
received 1000 mg cypermethrin/kg bw per day in DMSO for 5 days. After
3 weeks the dosing regime was repeated and a further three weeks later
the birds were killed. A positive (tri- ortho-tolyl phosphate) and
negative control (not dosed) group were used.
None of the cypermethrin-treated hens developed any signs of
intoxication. Histological examination of the nervous system revealed
no lesions. All birds receiving the positive control developed
clinical signs of neurological damage within 15 days and became
progressively more unsteady and ataxic thereafter. Histological
examination of these animals showed lesions in the cerebellum, sciatic
nerve and spinal cord, including axonal and myelin degeneration
(Owen & Butterworth, 1977).
2.2.7.3 Hamsters
The 1979 JMPR (FAO, 1980) evaluated some neurotoxicity studies
with cypermethrin in hamsters. At doses of > 794 mg/kg bw, all
treated hamsters showed clinical signs of poisoning, including
tremors, abnormal irregular movements and an unusual gait. As in the
cases of rats, axon and myelin degeneration was noted in all groups
treated. The lesions included swelling and breaks in the axons and
clumping of myelin.
Hamsters treated orally with a single dose of 40 mg/kg bw,
followed by four doses of 20 mg/kg bw, developed weight loss and
sometimes mortality. There was loss of fur and dermal ulceration.
There was no effect in the mean slip angle experiment, and a marginal
increase in ß-galactosidase was observed in peripheral nerve.
Hamsters treated orally with doses of 5, 10 or 20 mg/kg bw per
day for 5 days showed no mortality. Lower body weight gain was
observed at 20 mg/kg bw per day. One female (out of 5) at the highest
dose developed hyperexcitability. There was a significant deficit in
the mean slip angle test, females showing an earlier dose-related
deficit than noted in males. ß-Galactosidase activity was increased at
all dose levels 3 weeks after the onset of the experiment. This effect
was significant at the two highest dose levels. In this experiment
dermal irritation and fur loss were noted.
In another experiment hamsters were orally treated with
30 mg/kg bw per day for 5 days. There was no mortality and there were
no differences in weight gain. There was some transient skin
irritation accompanied by skin ulceration. One male (out of 16) had an
unusual gait. There was a slight deficit in the inclined plane test
which was noted in the early parts of the experiment. Increases in
both ß-glucuronidase and ß-galactosidase were evident in peripheral
nerve tissue.
2.2.8 Special studies on biochemistry and electrophysiology
In three independent experiments with Wistar rats the effects of
varying doses of cypermethrin (purity 98%, 10% in DMSO) on the
trigeminal ganglion and three sections of the maxillary branch of the
trigeminal nerve (proximal, distal and endings) were determined.
Increases in ß-galactosidase activity in these tissues were taken as
evidence of axonal degeneration.
The three studies involved repeated oral administration of
cypermethrin at 150 mg/kg bw per day for 5 or 7 days and 0, 25, 50,
100 or 200 mg/kg bw per day for 5 or 7 days. Mortality occurred in
animals receiving 100 mg/kg bw per day or more. A dose-related
transient functional impairment, assessed by means of the inclined
plane test was found in the first week. Significant increases in
ß-glucuronidase and ß-galactosidase activity of the sciatic, tibial or
trigeminal nerves only occurred with 5 or 7 doses of 150 or
200 mg/kg bw per day. Increased activity of the enzymes in the distal
portion of nerves was found, but even in the most severely intoxicated
animals the magnitude of this increase was less than that induced by
the known neurotoxic agent methylmercury chloride (Dewar & Moffett,
1978).
Cypermethrin (1:1 cis:trans) was administered to male and female
rats at dose levels ranging from 25 to 200 mg/kg bw per day for 5
consecutive days by oral intubation as a 10% w/v solution in DMSO. A
dose-related functional deficit was observed when the mean slip angle
test and the landing foot spread test were applied to the animals. The
deficit was maximal from days 6 to 14 after the beginning of
treatment, and complete functional recovery occurred within 4 weeks.
Substantial variation in data from the landing foot spread test was
noted. Data were inconsistent over the course of the study.
ß-Glucuronidase activity was increased in a dose-dependent fashion in
both males and females. The results suggested that cypermethrin
produced a primary axonal degeneration, readily measurable 28 days
after treatment as an increase in ß-glucuronidase activity and in
deficits in specific behavioural-function testing of rats (FAO, 1980).
Electrophysiological studies were performed to determine whether
acute or subacute intoxication with cypermethrin produced changes in
the conduction velocity of slower fibres in peripheral nerves or
alterations in the maximal motor conduction velocity. There was no
evidence to suggest that cypermethrin, at doses that induced severe
clinical signs of intoxication, including ataxia, had any effect on
maximal motor conduction velocity or conduction velocity of the slower
motor fibres in peripheral nerves. Doses used in the study ranged from
a single dose of 200 mg/kg bw to 7 consecutive doses of 150 mg/kg bw
followed by two doses of 400 mg/kg bw. At near-lethal doses there were
no effects on conduction velocity even in the presence of clinical
signs of acute intoxication and at dose levels where previous studies
had shown functional degeneration. These electro-physiological
findings are reflective of motor function, which would suggest that
the physiological and functional deficits observed as a result of
acute intoxication are primarily sensory in nature (FAO, 1980).
2.2.9 Special studies on sensitization
2.2.9.1 Cypermethrin
Two out of 20 guinea-pigs developed a positive reaction to
cypermethrin in the Magnusson Kligman test, indicating that
cypermethrin is not a sensitizer (Coombs et al., 1976).
2.2.9.2 alpha-Cypermethrin
No positive reactions were obtained in a Magnusson-Kligman test
performed with guinea-pigs (Gardner, 1993).
2.2.10 Special studies on skin and eye irritation
2.2.10.1 Cypermethrin
A single application of undiluted technical cypermethrin was
moderately irritant to occluded rabbit skin (Coombs et al., 1976).
A single application of undiluted technical cypermethrin to
rabbit eyes produced a mild transient conjunctivitis and blepharism
lasting 2 days (Coombs et al., 1976).
2.2.10.2 alpha-Cypermethrin
Six NZW rabbits receiving a semi-occlusive topical application
with 500 mg alpha-cypermethrin technical developed very slight
erythema in two animals up to 72 hours after removal of the dressings.
There were no other dermal reactions (Gardner, 1993).
Six NZW rabbits receiving an instillation of 0.1 ml (equivalent
to 45 mg) alpha-cypermethrin technical developed slight conjunctival
redness and ocular discharge up to 72 hours after treatment. No cornea
or iris irritation was observed (Gardner, 1993).
2.3 Observations in humans
The symptoms and signs of acute poisoning resulting from
pyrethroids are very similar. Apart from the irritative symptoms of
the skin and respiratory tract (or digestive tract in ingestive
poisoning), acute pyrethroid poisoning is clinically characterized by
abnormalities of nervous excitability.
Occupationally exposed people had abnormal skin sensations
described as burning, itching or tingling, which could be exacerbated
by sweating or washing and readily disappeared after several hours.
Systemic symptoms included dizziness, headache, nausea, anorexia and
fatigue. Vomiting was more prominent in patients with ingestive
poisoning than in occupational poisoning cases. Other symptoms such as
chest tightness, paraesthesia, palpitation, blurred vision and
increased sweating were less frequently seen. The more serious cases
developed coarse muscular fasciculations in large muscles or
extremities (Van den Bercken & Vijverberg, 1989; He et al., 1989).
Urine obtained from operators spraying cypermethrin in
experimental trials was analysed for the presence of the chlorinated
cyclopropane carboxylic acid metabolite. This metabolite was observed
in the urine of exposed workers at levels up to 0.4 µg/ml (the limit
of detection was estimated to be 0.05 µg/ml).
Cypermethrin sprayers were found to have residues on the exposed
parts of their bodies. The rate of dermal exposure of the operators
during spraying ranged from 1.5 to 46.1 mg/hour. There was a
reasonable relationship between the total cypermethrin deposited
dermally and the excretion in urine. The levels of the cyclopropane
carboxylic acid metabolite in the 24-hour urine were between <0.05
and 0.32 mg (not specified). This, together with the finding of 0.6 mg
(not specified) of this metabolite in 72-hour urine from one man, led
to the estimation that approximately 3% of the total dermal dose was
absorbed and rapidly excreted by the operators (FAO, 1980).
In a controlled experiment with sprayers, no abnormalities were
found in clinical and neurological examinations, blood chemistry or
peripheral nerve function tests (including the trigeminal nerve). In
some electroneurophysiological tests (motor conduction velocity, slow
fibre conduction velocity and cornea reflex), a significant change
within the normal range appeared to exist for the group sprayers
between pre- and post-exposure measurements. These changes probably
reflect seasonal variations (FAO, 1982).
3. COMMENTS
The Committee considered toxicological data on cypermethrin and
alpha-cypermethrin, including the results of acute, short-term, and
reproductive studies, and studies on pharmacokinetics and metabolism,
genotoxicity, long-term toxicity/carcinogenicity and neurotoxicity.
Results of effects in humans were also considered.
Cypermethrin is a mixture of four cis and four trans isomers. The
cis isomers are more biologically active and more persistent than the
trans isomers, alpha-Cypermethrin is a mixture of the two most active
cis isomers. A typical cypermethrin sample contains 25% alpha-
cypermethrin. Cypermethrin and alpha-cypermethrin are alpha-cyano or
type II pyrethroids that cause neurotoxicity in mammals and insects.
They affect nerve membrane sodium channels, causing a long-lasting
prolongation of the normally transient increase in sodium permeability
of the membrane during excitation. At high dose levels, these type II
pyrethroids induce salivation and tremors that progress to
characteristic clonic-tonic convulsions (choreoathetosis and
salivation syndrome).
After oral administration, cypermethrin is readily absorbed,
distributed and excreted in rats, chickens, sheep and cattle.
Cypermethrin is primarily eliminated in urine and faeces in about
equal proportions. Less than 1% is excreted in milk. When cypermethrin
was applied dermally to sheep, 2.5% was eliminated in urine and faeces
within 6 days and after an oral dose about 60% was eliminated within 2
days.
Studies in cattle indicated that absorption, distribution and
excretion were comparable for cypermethrin and alpha-cypermethrin. The
major metabolic route for both cypermethrin and its isomers, including
alpha-cypermethrin, is cleavage of the ester bond followed by
hydroxylation and conjugation of the cyclopropyl and phenoxybenzyl
portions of the molecule. The data suggest that there is no isomeric
interconversion during metabolism.
The acute oral toxicity of cypermethrin and alpha-cypermethrin is
moderate to high. WHO has classified these substances as "moderately
hazardous" (WHO, 1996). In rats and mice, the oral LD50 ranges from
82 to 4000 mg/kg bw for cypermethrin and from 35 to > 5000 mg/kg bw
for alpha-cypermethrin, depending on the vehicle used. At lethal or
near lethal doses the signs are typical of type-II pyrethroids and
include salivation, ataxia, gait abnormalities and convulsions.
Several oral short-term toxicity studies with cypermethrin were
available. Cypermethrin has been tested in rats (5 weeks and 90 days)
and dogs (5 weeks and 13 weeks) at dose levels ranging from 25 to
1600 mg/kg feed (equivalent to 1.25-80 mg/kg bw per day) and 5 to
1500 mg/kg feed (equivalent to 0.125-37.5 mg/kg bw per day),
respectively. In these studies, the clinical signs included ataxia,
abnormal gait, nervousness, and, particularly in dogs, inappetence,
diarrhoea, vomiting and hyperaesthesia. In both rats and dogs,
cypermethrin caused decreases in body weight gain, food intake, and a
number of haematological parameters, increases in some organ weights
and plasma urea levels, and, at lethal or near-lethal doses, effects
on the nervous system. For cypermethrin the lowest NOEL in short-term
studies was in a 90-day study with rats administered 25, 100, 400 or
1600 mg/kg feed (equivalent to 1.25-80 mg/kg bw per day). Male rats
given 1600 mg/kg feed showed decreases in haemoglobin concentration,
mean corpuscular volume and eosinophil numbers, and increases in
prothrombin time, plasma urea levels and relative liver and kidney
weights. The decrease in eosinophil numbers and increase in relative
liver weight were also observed in males at 400 mg/kg feed. In female
rats, reduced food intake and increased relative liver weight were
noted in rats given 1600 mg/kg feed. The NOEL in this study was
100 mg/kg feed, equivalent to 5 mg/kg bw per day.
alpha-Cypermethrin was tested in oral short-term toxicity studies
with mice (29 days and 13 weeks), rats (5 weeks, 6 weeks and 90 days)
and dogs (13 weeks and 52 weeks) at dose levels ranging from 50 to
1600 mg/kg feed (equivalent to 7-240 mg/kg bw per day), 20 to
1200 mg/kg feed (equivalent to 1.25-60 mg/kg bw per day) and 30 to
270 mg/kg feed (equivalent to 0.75-6.75 mg/kg bw per day),
respectively. In these studies, alpha-cypermethrin caused the same
effects as described for cypermethrin in the short-term studies. The
signs of toxicity included ataxia, abnormal gait, increased
sensitivity to noise, hyperactivity, hunched posture and, as
demonstrated histologically, axonal degeneration of the sciatic
nerves. For alpha-cypermethrin the lowest NOEL was in a 52-week study
with dogs. In this study a diet containing 60, 120 or 240 mg/kg feed
(equivalent to 1.5 to 6 mg/kg bw per day) was administered. Dogs given
120 and 240 mg/kg feed showed skin reddening, ulceration and necrosis.
The NOEL in this study was 1.5 mg/kg bw per day.
A three-generation reproductive toxicity study with cypermethrin
was performed in rats at dose levels of 10, 100 or 500 mg/kg feed
(equivalent to 0.5-25 mg/kg bw per day). At the highest dose a
reduction in body weight gain and food consumption and a concomitant
reduction in litter size and weight were seen in the F1a progeny
only. No other effects on fertility or reproduction parameters were
observed. The NOEL was 100 mg/kg feed, equivalent to 5 mg/kg bw per
day.
Cypermethrin did not cause embryotoxicity or teratogenicity in
rats at doses up to 70 mg/kg bw per day or in rabbits at doses up to
120 mg/kg bw per day. alpha-Cypermethrin did not cause embryotoxicity
or teratogenicity in rats at doses up to 9 mg/kg bw per day or in
rabbits up to 30 mg/kg bw per day. The NOELs for maternal toxicity in
rats were 17.5 and 9 mg/kg bw per day for cypermethrin and
alpha-cypermethrin, respectively, while the NOELs for maternal
toxicity in rabbits were 30 and 3 mg/kg bw per day for cypermethrin
and alpha-cypermethrin, respectively.
Cypermethrin and alpha-cypermethrin have been tested in a wide
variety of in vitro and in vivo genotoxicity studies. All of the
results were negative.
Two long-term toxicity/carcinogenicity studies with mice and rats
were available on cypermethrin. Mice received a diet containing 100,
400 or 1600 mg/kg feed (equal to 14-228 mg/kg bw per day) for 101
weeks. At 1600 mg/kg feed reduced body weight gain, changes in
haematological parameters and increased liver weight were observed. No
effects were observed at 400 mg/kg feed, equal to 57 mg/kg bw per day.
In a study in which rats received diets containing 1, 10, 100 or
1000 mg/kg feed (equivalent to 0.05-50 mg/kg bw per day) for 2 years,
the only effects observed were reductions in body weight and food
consumption at 1000 mg/kg feed. The NOEL was 100 mg/kg feed,
equivalent to 5 mg/kg bw per day.
In a two-year toxicity study, dogs received diets containing
3, 30, 300 or 1000 mg cypermethrin/kg feed (equivalent to
0.075-25 mg/kg bw per day). The dose level of 1000 mg/kg feed was
reduced to 600 mg/kg feed owing to severe intoxication. At 300 mg/kg
feed, equivalent to 7.5 mg/kg bw per day, no effects were seen. The
Committee concluded that cypermethrin was not carcinogenic in these
studies.
Long-term toxicity/carcinogenicity or reproductive toxicity
studies were not available on alpha-cypermethrin. The Committee noted
the absence of reproductive toxicity and carcinogenicity associated
with administration of cypermethrin, which contains 25% alpha-
cypermethrin. The Committee also noted the absence of genotoxicity
for either cypermethrin or alpha-cypermethrin, the absence of
carcinogenicity associated with compounds of similar structure and
the similar metabolism and disposition of the two compounds. In view
of the foregoing, the Committee concluded that it was unnecessary to
request the results of long-term toxicity/carcinogenicity or
reproductive toxicity studies on alpha-cypermethrin.
Several studies on the neurotoxicity of cypermethrin and
alpha-cypermethrin in rats were available. In these studies high oral
doses of cypermethrin and alpha-cypermethrin caused clinical signs
that included coarse tremor and spasmodic body and tail movements.
Evidence of axonal damage in the sciatic/posterior tibial nerves and
the trigeminal nerve and ganglion was indicated by significant
increases in ß-glucuronidase and ß-galactosidase in nerve tissue
homogenates, in addition to abnormal neuromuscular function tests. In
the inclined plane test, cypermethrin (in DMSO) caused transient
functional impairment. The lowest NOEL for neurotoxicity was
37.5 mg/kg bw per day for cypermethrin (in DMSO) and 4 mg/kg bw per
day for alpha-cypermethrin (in corn oil), indicating that the toxicity
may be influenced by the vehicle used.
Humans occupationally exposed to cypermethrin developed skin
sensation as a first reaction, followed by systemic effects such as
dizziness, headache, nausea, paraesthesia and increased sweating. In
more serious cases, muscular fasciculations developed in large muscles
or in the extremities. In experiments with operators spraying
cypermethrin, no clinical nervous system abnormalities were observed.
However, exposure levels were not measured.
4. EVALUATION
The Committee established an ADI of 0-50 µg/kg bw for
cypermethrin on the basis of the NOEL of 5 mg/kg bw per day in 90-day,
2-year and reproductive toxicity studies in rats and the application
of a safety factor of 100. The Committee established an ADI of
0-20 µg/kg bw for alpha-cypermethrin on the basis of the NOEL of
1.5 mg/kg bw per day in a 52-week study in dogs and the application of
a safety factor of 100.
5. REFERENCES
Amyes, S.J., Holmes, P., Irving, E.M., Green, C.F., Virgo, D.M., &
Sparrow, S. (1994). Alpha-cypermethrin: preliminary toxicity study by
dietary administration to CD-1 mice for 13 weeks. Unpublished report
No. 92/SHL009/0849 from Pharmaco-LSR Ltd, Suffolk, England. Submitted
to the WHO by Cyanamid, Wayne, NJ. USA.
Brooks, T.M. (1980). Toxicity studies with agricultural chemicals:
Mutagenicity studies with RIPCORD in microorganisms in vitro and in
the host-mediated assay (Experiment Nos 1846 and 1847). Unpublished
report TLGR.80.059 from Shell Toxicology Laboratory (Tunstall).
Submitted to the WHO by Cyanamid, Wayne, NJ, USA.
Brooks, T.M. (1984). Genotoxicity studies with Fastac: the induction
of gene mutation in the yeast Saccharomyces cerevisiae. Unpublished
report No. SBGR.84.117 from Shell Research Limited, Sittingbourne
Research Centre. Submitted to the WHO by Cyanamid, Wayne, NJ, USA.
Brooks, T.M. & Wiggins, D.E. (1992). FASTAC TM: Bacterial mutagenicity
studies. Unpublished report No. SBTR.92.022 from Shell Research
Limited, Sittingbourne Research Centre. Submitted to the WHO by
Cyanamid, Wayne, NJ, USA.
Brooks, T.M. & Wiggins, D.E. (1993). FASTAC TM: In vitro chromosome
studies using cultured human lymphocytes. Unpublished report
No. SBTR.93.007 from Shell Research Limited, Sittingbourne Research
Centre. Submitted to the WHO by Cyanamid, Wayne, NJ, USA.
Buckwell, A.C. (1981). A 2-year feeding study in dogs on WL 43467
(Experiment No. 1412). Unpublished report No. SBGR.81.126 (plus two
addenda) from Shell Toxicology Laboratory (Tunstall). Submitted to the
WHO by Cyanamid, Wayne, NJ, USA.
Buckwell, A.C. & Butterworth, S. (1977). Toxicology studies on the
pyrethroid insecticide WL 43467: a 13-week feeding study in dogs
(Experiment No. 1112). Unpublished report No. TLGR 0127.77 (including
addendum and corrigendum) from Shell Toxicology Laboratory (Tunstall).
Submitted to the WHO by Cyanamid, Wayne, NJ, USA.
Carter, B.I. & Butterworth, S.T.G. (1976). Toxicity of insecticides:
The acute oral toxicity and neuropathological effects of WL 43467 to
rats. Unpublished report No. TLGR.0055.76 from Shell Research,
Sittingbourne Research Centre. Submitted to the WHO by Cyanamid,
Wayne, NJ, USA.
Clare, M.G. & Wiggins, D.E. (1984). Genotoxicity studies with fastac:
in vivo cytogenetic test using rat bone marrow. Unpublished report
No. SBGR.84.120 from Shell Research Limited, Sittingbourne Research
Centre. Submitted to the WHO by Cyanamid, Wayne, NJ, USA.
Clark, D.G. (1982). WL85871: A 90-day feeding study in rats.
Unpublished report No. SBGR.81.293 from Shell Research Limited,
Sittingbourne Research Centre. Submitted to the WHO by Cyanamid,
Wayne, NJ, USA.
Coombs, A.D., Carter, B.I., Hend, R.W., Butterworth, S.G., & Buckwell,
A.C. (1976) Toxicity studies on the insecticide WL 43467: Summary of
results of preliminary experiments. Unpublished report
No. TLGR.0104.76 from Shell Toxicology Laboratory (Tunstall).
Submitted to the WHO by Cyanamid, Wayne, NJ, USA.
Crawford, M.J. (1978). The excretion and residues of radioactivity in
cows treated orally with 14C-labelled WL 43467. Unpublished report
No. TLGR.0029.78 from Shell Toxicology Laboratory (Tunstall).
Submitted to the WHO by Cyanamid, Wayne, NJ, USA.
Crawford, M.J. & Hutson, D.H. (1977). The elimination and retention of
WL 43467 when administered dermally or orally to sheep. Unpublished
report No. TLGR.0098.77 from Shell Toxicology Laboratory (Tunstall).
Submitted to the WHO by Cyanamid, Wayne, NJ, USA.
Crawford, M.J. & Hutson, D.H. (1978). The elimination of residues from
the fat of rats following the oral administration of [14C-benzyl]
WL 43381 (cis-WL 43467). Unpublished report No. TLGR.0078.78 from
Shell Toxicology Laboratory (Tunstall). Submitted to the WHO by
Cyanamid, Wayne, NJ, USA.
Creedy, C.L. & Wooder, M.F. (1977) Studies on the effect of WL 43467
upon the integrity of rat liver cell DNA in vivo. Unpublished report
No. TLGR.0043.77 from Shell Toxicology Laboratory (Tunstall).
Submitted to the WHO by Cyanamid, Wayne, NJ, USA.
Croucher, A., Stoydin, G., Cheeseman, M.E., Buckwell, A.C., &
Gellatly, J.B.M. (1980). The metabolic fate of cypermethrin in the
cow: elimination and residues derived from 14C-benzyl label.
Unpublished report No. TLGR.80.121 from Shell Toxicology Laboratory
(Tunstall). Submitted to the WHO by Cyanamid, Wayne, NJ, USA.
Dean, B.J. (1977). Toxicity studies with WL 43467: Chromosome studies
on bone marrow cells of Chinese hamsters after two daily oral doses of
WL 43467. Unpublished report No. TLGR.0136.77 from Shell Toxicology
Laboratory (Tunstall). Submitted to the WHO by Cyanamid,
Wayne, NJ, USA.
Dean, B.J. (1981). Toxicity studies with agricultural chemicals:
In vitro genotoxicity studies with RIPCORD (Experiment
No. AIR-1820). Unpublished report No. TLGR.80.116 from Shell
Toxicology Laboratory (Tunstall). Submitted to the WHO by Cyanamid,
Wayne, NJ, USA.
Dean, I. & Jackson, F. (1995). WL85871: 52-week oral (dietary)
toxicity study in dogs (IRI Project No. 652238). Unpublished report
No. IRI/11110 from Inveresk Research International, Scotland.
Submitted to the WHO by Cyanamid, Wayne, NJ, USA.
Dean, B.J., Van der Pauw, C.L., & Butterworth S.T.G. (1977). Toxicity
studies with WL 43467: Dominant lethal assay in male mice after single
oral doses of WL 43467. Unpublished report No. TLGR.0042.77 from Shell
Toxicology Laboratory (Tunstall). Submitted to the WHO by Cyanamid,
Wayne, NJ, USA.
Dewar, A.J. & Moffett, B.J. (1978). Toxicity studies on the
insecticide WL 43467: Biochemical studies on the effect of WL 43467 on
the rat trigeminal nerve and ganglion. Unpublished report No.
TLGR.0162.77 from Shell Toxicology Laboratory (Tunstall). Submitted to
the WHO by Cyanamid, Wayne, NJ, USA.
Dunsire, J.P. & Gifford, L.J. (1993). The distribution of
[benzyl-14C]WL85871 (FASTAC) in the lactating cow (nature of residue
study to EPA guidelines - Live phase) (IRI Project No. 153667).
Unpublished report No. IRI/9879 from Inveresk Research International,
Scotland. Submitted to the WHO by Cyanamid, Wayne, NJ, USA.
FAO (1980). Pesticide residues in food-1979. Report of the Joint
Meeting of the FAO Panel of Experts on Pesticide Residues in Food and
the Environment and the WHO Expert Group on Pesticide Residues. Rome,
Food and Agriculture Organization of the United Nations (FAO Plant
Production and Protection Paper, No. 20).
FAO (1982). Pesticide residues in food-1981. Report of the Joint
Meeting of the FAO Panel of Experts on Pesticide Residues in Food and
the Environment and the WHO Expert Group on Pesticide Residues. Rome,
Food and Agriculture Organization of the United Nations (FAO Plant
Production and Protection Paper, No. 37).
Fish, A. (1979). Corrigendum I in Group Research Report
No. TLGR.0188.78. Unpublished data from Shell Toxicology Laboratory
(Tunstall). Submitted to the WHO by Cyanamid, Wayne, NJ, USA.
Fokkema, G.N. (1994a). WL85871 (FASTAC): A 6-week range finding
feeding study in the rat. Unpublished report No. SBTR.93.002 from
Shell Research Limited, Sittingbourne Research Centre. Submitted to
the WHO by Cyanamid, Wayne, NJ, USA.
Fokkema, G.N. (1994b). WL85871 (FASTAC): An acute oral (gavage)
neurotoxicity study in the rat. Unpublished report No. SBTR.92.027
from Shell Research Limited, Sittingbourne Research Centre. Submitted
to the WHO by Cyanamid, Wayne, NJ, USA.
Gardner, J.R. (1993). FASTAC technical: Acute oral and dermal toxicity
in rat, skin and eye irritancy in rabbit and skin sensitisation
potential in guinea pig. Unpublished report No. SBTR.92.033 from Shell
Research Limited, Sittingbourne Research Centre. Submitted to the WHO
by Cyanamid, Wayne, NJ, USA.
Green, C.F. (1993). Alphacypermethrin: Preliminary toxicity study by
dietary administration to CD-1 mice for four weeks. Unpublished report
No. LSR 92/0346 from Life Science Research Limited. Submitted to the
WHO by Cyanamid, Wayne, NJ, USA.
Greenough, R.J. & Goburdhun, R. (1984). WL 85871: Oral (dietary)
maximum tolerated dose study in dogs (IRI Project No. 631087).
Unpublished report No. IRI/3107 from Inveresk Research International,
Scotland. Submitted to the WHO by Cyanamid, Wayne, NJ, USA.
Greenough, R.J., Cockrill, J.B., & Goburdhun, R. (1984).
WL 85871: 13-week oral (dietary) toxicity study in dogs (IRI Project
No. 631092). Unpublished report No. IRI/3197 from Inveresk Research
International, Scotland. Submitted to the WHO by Cyanamid,
Wayne, NJ, USA.
He, F., Wang, S., Liu, L., Chen, S., Zhang, Z. & Sun, J. (1989).
Clinical manifestations and diagnosis of acute pyrethroid poisoning.
Arch. Toxicol., 63, 54-58.
Hend, R.W., Hendy, R. & Fleming, D.J. (1978). Toxicity studies on the
insecticide WL 43367: a three generation reproduction study in rats.
Unpublished report No. TLGR.0188.78 from Shell Toxicology Laboratory
(Tunstall). Submitted to the WHO by Cyanamid, Wayne, NJ, USA.
Henderson, C. & Parkinson, G.R. (1981). Cypermethrin technical:
subacute dermal toxicity study in rabbits (CTL Study No. LB0019).
Unpublished report No. CTL/P/588 from ICI Central Toxicology
Laboratory. Submitted to the WHO by Cyanamid, Wayne, NJ, USA.
Hijzen, T.H., de Beun, R., & Slanger, J.L. (1988). Effects of
pyrethroids on the acoustic startle reflex in the rat. Toxicology,
49, 271-276.
Hutson, D.H. & Stoydin, G. (1976). The excretion of radioactivity from
cows fed with radioactively labelled WL 43467. Unpublished report
No. TLGR.0075.76 from Shell Toxicology Laboratory (Tunstall).
Submitted to the WHO by Cyanamid, Wayne, NJ, USA.
Hutson, D.H. & Stoydin, G. (1987). Excretion and residues of the
pyrethroid insecticide cypermethrin in laying hens. Pestic. Sci.,
18, 157-168.
IPCS (1996) The WHO recommended classification of pesticides by hazard
and guidelines to classification 1996-1997. World Health Organization,
Geneva, p. 23 (Document WHO/PCS/96.3).
Irvine, L.F.H (1994a). Oral (gavage) rabbit developmental toxicity
dose ranging study. Unpublished report No. SLN/3/92 from Toxicol
Laboratories Limited, Ledbury, England. Submitted to the WHO by
Cyanamid, Wayne, NJ, USA.
Irvine, L.F.H. (1994b). Oral (gavage) rabbit developmental toxicity
(teratogenicity) study. Unpublished report No. SLN/4/93 from Toxicol
Laboratories Limited, Ledbury, England. Submitted to the WHO by
Cyanamid, Wayne, NJ, USA.
Irvine, L.H.F. (1994c). Alphacypermethrin. Oral (gavage) rat
developmental toxicity (teratogenicity) study. Unpublished report
No. SLN/2/92 from Toxicol Laboratories Limited, Ledbury, England.
Submitted to the WHO by Cyanamid, Wayne, NJ, USA.
Irvine, L.F.H & Twomey, K. (1994). Alphacypermethrin. Oral (gavage)
rat developmental study. Unpublished report No. SLN/1/92 from Toxicol
Laboratories Limited, Ledbury, England. Submitted to the WHO by
Cyanamid, Wayne, NJ, USA.
Jackson, G.C. (1993). Alphacypermethrin: Acute inhalation toxicity in
rats, 4-hour exposure. Unpublished report No. SLL 266/930770 from
Huntingdon Research Centre, England. Submitted to the WHO by Cyanamid,
Wayne, NJ, USA.
Lindsay, S., Banham, P.B., Chart, I.S., Chalmers, D.T., Godley, M.J.,
& Taylor, K. (1982). Cypermethrin: Lifetime feeding study in mice
(CTL Study No. PM0366). Unpublished report No. CTL/P/687 (including 1
supplement) from ICI Central Toxicology Laboratory. Submitted to the
WHO by Cyanamid, Wayne, NJ, USA.
McAusland, H.E., Butterworth, S.T.G., & Hunt, P.F. (1978). Toxicity
studies on the insecticide WL 43467: A two-year feeding study in rats.
Unpublished report No. TLGR.0189.78 (including 4 corrigenda/addenda)
from Shell Toxicology Laboratory (Tunstall). Submitted to the WHO by
Cyanamid, Wayne, NJ, USA.
Morrison, B.J. & Richardson, K.A. (1994). WL85871 (alphacypermethrin,
FASTAC): The metabolism of 14C-WL85871 after repeated oral dosing in
the lactating cow; in-life phase and metabolite profiling (Experiment
No. 6046). Unpublished report No. SBTR.93.063 from Sittingbourne
Research Centre, Sittingbourne, England. Submitted to the WHO by
Cyanamid, Wayne, NJ, USA.
Owen, D.E. & Butterworth, S.T.G. (1977). Toxicity of pyrethroid
insecticides: Investigation of the neurotoxic potential of WL 43467 to
adult domestic hens. Unpublished report No. TLGR.0134.77 from Shell
Toxicology Laboratory (Tunstall). Submitted to the WHO by Cyanamid,
Wayne, NJ, USA.
Pickering, R.G. (1981). A 90-day feeding study of WL 43467 in rats
(Experiment No. 1806). Unpublished report No. TLGR.80.143 from Shell
Toxicology Laboratory (Tunstall). Submitted to the WHO by Cyanamid,
Wayne, NJ, USA.
Potter, D. & McAusland, H.E. (1980). Toxicity studies on the
insecticide WL43467: A study of liver microsomal enzyme activity in
rats fed WL 43467 for 2 years (Experiment No. 1105). Unpublished
report from Shell Toxicology Laboratory (Tunstall). Submitted to the
WHO by Cyanamid, Wayne, NJ, USA.
Price, J.B. (1984). Toxicology of pyrethroids: the acute oral and
percutaneous toxicity of Ripcord (WL-43467). Technical concentrate
(Project No. SRCAIR84). Unpublished report No. SBGR.84.299 from Shell
Research Limited, Sittingbourne Research Centre. Submitted to the WHO
by Cyanamid, Wayne, NJ, USA.
Rose, G.P. (1982a). Toxicology of pyrethroids: the acute oral and
percutaneous toxicity of WL 85871 (cis-2-Ripcord) in comparison with
Ripcord. Unpublished report No. SBGR.82.130 from Shell Research
Limited, Sittingbourne Research Centre. Submitted to the WHO by
Cyanamid, Wayne, NJ, USA.
Rose, G.P. (1982b). Toxicology of pyrethroids: the acute oral and
percutaneous toxicity of WL 85871 (cis-2-RIPCORD) comparison with
RIPCORD. Unpublished report No. SBGR.82.130 from Shell Research
Limited, Sittingbourne Research Centre. Submitted to the WHO by
Cyanamid, Wayne, NJ, USA.
Rose, G.P. (1983). Neurotoxicity of WL85871. Comparison with WL43467:
The effect of twenty oral doses of WL85871 or WL43467 over a period of
4 weeks on the rat sciatic/posterior tibia nerve, trigeminal nerve and
trigeminal ganglion. Unpublished report No. SBGR.83.185 from Shell
Research Limited, Sittingbourne Research Centre. Submitted to the WHO
by Cyanamid, Wayne, NJ, USA.
Rose, G.P. & Dewar, A.J. (1983). Intoxication with four synthetic
pyrethroids fails to show any correlation between neuromuscular
dysfunction and neurobiochemical abnormalities in rats.
Arch. Toxicol., 53, 297-316.
Tesh, J.M., Tesh, S.A., & Davies, W. (1978). WL 43467: Effects upon
the progress and outcome of pregnancy in the rat. Unpublished report
of Life Science Research. Submitted to the WHO by Cyanamid,
Wayne, NJ, USA.
Tesh, J.M., Ross, F.W., & Wightman, T.J. (1984a). WL 43467: Effects of
oral administration upon pregnancy in the rabbit. 1. Dosage range
finding study. Unpublished test report No. 84/SHL003/014 from of Life
Science Research. Submitted to the WHO by Cyanamid, Wayne, NJ, USA.
Tesh, J.M., Ross, F.W., & Wightman, T.J. (1984b). WL 43467: Effects of
oral administration upon pregnancy in the rabbit. 2. Main study.
Unpublished test report No. 84/SHL004/043 from Life Science Research.
Submitted to the WHO by Cyanamid, Wayne, NJ, USA.
Tesh, J.M., Ross, F.W., & Wightman, T.J. (1988). Supplementary report
to LSR report No. 84/SHL004/043. Unpublished report No. 88/SHL004/775
from Life Science Research. Submitted to the WHO by Cyanamid,
Wayne, NJ, USA.
Thorpe, E. (1982). A 5-week feeding study with WL 85871 in rats
(Experiment No. 2095). Unpublished report No. SBGR.81.212 from Shell
Research Limited, Sittingbourne Research Centre. Submitted to the WHO
by Cyanamid, Wayne, NJ, USA.
Thorpe, E. (1985). Fourth corrigendum/addendum to TLGR.0188.78.
Unpublished report from Shell Research Limited, Sittingbourne Research
Centre (Four volumes). Submitted to the WHO by Cyanamid,
Wayne, NJ, USA.
Van der Bercken, J. & Vijverberg, H.P.M. (1989). Neurotoxicological
effects of pyrethroid insecticides. Unpublished report from the
Department of Veterinary Pharmacology, Pharmacy and Toxicology.
University of Utrecht, The Netherlands. Submitted to the WHO by
Cyanamid, Wayne, NJ, USA.
Vanderwaart, E.J. (1994). Evaluation of the mutagenic activity of
Fastac technical in a in vitro mammalian cell gene mutation test
with L5178Y mouse lymphoma cells (with independent repeat).
Unpublished report No. 087367 from Notox B.V., The Netherlands.
Submitted to the WHO by Cyanamid, Wayne, NJ, USA.
Vanderwaart, E.J. (1995). Micronucleus test in bone marrow cells of
the mouse with Fastac technical. Unpublished report No. 087378 from
Notox B.V., The Netherlands. Submitted to the WHO by Cyanamid,
Wayne, NJ, USA.
WHO (1996). The WHO recommended classification of pesticides by hazard
and guidelines to classification 1996-1997 (WHO/PCS/96.3). Available
from the International Programme on Chemical Safety, World Health
Organization, Geneva, Switzerland.
Wooder, M.F. (1981). Studies on the effect of WL 85871 on the
integrity of rat liver DNO in vivo (Experiment No. 2103).
Unpublished report No. SBGR.81.225 from Shell Toxicology Laboratory
(Tunstall). Submitted to the WHO by Cyanamid, Wayne, NJ, USA.
2. BIOLOGICAL DATA
2.1 Biochemical aspects
2.1.1 Absorption, distribution and excretion
According to the JMPR (FAO, 1980, 1982) cypermethrin is readily
absorbed and rapidly eliminated via urine and faeces of mice, rats,
dogs, sheep and cows. Absorption from the gastrointestinal tract is
more rapid with the trans isomer than with the cis isomer. The highest
mean concentrations are found in body fat, liver, kidney, muscle, skin
and milk. The clearance rate from adipose tissue is slow and the
elimination half-life in rats and mice ranges from 10 to 20 and from
20 to 30 days, respectively. The data suggest a potential for
bioaccumulation in the body following continuous exposure. With
respect to the chemical and especially the isomeric complexity of the
molecule, the metabolic profile due to all of its isomers is extremely
complex. Cypermethrin is readily cleaved at the ester linkage to
produce the cyclopropane carboxylic acid and a 3-phenoxybenzoyl moiety
that is further metabolised by oxidation at the 4'position. The
resultant phenol is almost totally conjugated with sulfate. The
4'-hydroxy sulfate forms the major aryl metabolite (16%), and
3-phenoxybenzoic acid is the second most important (5%). The other
identified aryl metabolites are 3-(4-hydroxyphenoxy)benzoic acid (1%)
and the glycine conjugate, N-(3-phenoxybenzoyl) glycine (1%).
2.1.1.1 Rats
Eight female Wistar rats were orally dosed with 2.5 mg
14C-benzyl-labelled cis-cypermethrin/kg bw. They were killed in
groups of two at 8, 14, 25 and 42 days after dosing, and the
radioactivity in fat, liver and kidney was measured. Selected fat
samples were analysed for parent compound. Between days 14 and 42,
radioactivity was eliminated from the fat with a half-life of 20-25
days. Residues in liver and kidney were 30 to 40 times lower than
those in fat, but were eliminated at a similar rate. In two pooled fat
samples taken 8 and 24 days after dosing, 90-100% of the radioactivity
was in the form of the parent compound (Crawford & Hutson, 1978).
2.1.1.2 Chickens
Six Warren laying hens were treated daily for 14 days with 10 mg
14C-phenoxy-labelled cypermethrin/kg food (equivalent to 1.25 mg/kg
bw per day) in gelatin capsules in the diet. Eggs and excreta were
collected dally. The hens were killed 4.5 hours after the last dose,
and liver, muscle and fat samples were collected. Of the total
radioactivity, 95.2% was recovered. Radioactivity in eggs plateaued 8
days after the start of dosing and reached 0.05 mg/kg. Most
radioactivity was found in the yolk and was a mixture of parent
compound and material that was closely associated with neutral lipids
and phosphatidyl cholines. In tissues, highest radioactivity was found
in the liver (0.37 µg/g). This radioactivity was composed of parent
compound (0.05 µg/g) and a mixture of very polar metabolites that were
not hydrolysed to significant amounts of 3-phenoxybenzoic acid or its
4-hydroxy derivative. Fat contained 0.08 µg/g and about 60% of the
residue was present as parent compound. Residues in muscle
(0.01-0.02 µ/g) were too low for characterization (Hutson & Stoydin,
1987).
2.1.1.3 Sheep
Two male sheep were each treated dermally (single dose) with a
mixture of 962 mg 14C-cypermethrin (both cyclopropyl- and
benzyl-labelled), equivalent to 21.9 mg/kg bw in acetone. After 24
hours (sheep 1) and 6 days (sheep 2) the animals were killed, and
radioactivity in fat, muscle, liver, kidney and skin (application
area) was measured. Urine and faeces were collected up to the time the
sheep were killed. One sheep treated with acetone served as a control.
Another sheep was treated orally (single dose) with 177 mg 14C-
cypermethrin mixture in a gelatin capsule (equivalent to 3.9 mg/kg
bw). Urine and faeces were collected for 2 days and tissues were
assayed. When applied dermally radioactivity was slowly absorbed
and eliminated. Less than 0.5% was excreted in urine within 24 hours
and only 2% over a 6-day period. Faecal elimination was also slow,
0.5% of the dose being eliminated in 6 days. About 30% of the applied
radioactivity was recovered from the application areas of both sheep.
Very little radioactivity was found in the tissues. In liver, kidney,
renal fat and subcutaneous fat (other than the application area),
residues ranged from 0.09 to 0.3 µg/g and muscle samples contained
0.03 to 0.06 µg/g.
Following oral treatment, the elimination of radioactivity was
rapid, 61% of the administered radioactivity being eliminated within
48 hours. Urinary elimination comprised 41% of the dose and faecal
elimination 20.5%. Residues found in tissues were comparable to those
found after dermal application. Most of the radioactivity in the fat
samples of all sheep was found to be parent compound. In muscle, liver
and kidney samples, only a small percentage of the total tissue
radioactivity was identified as parent compound (Crawford & Hutson,
1977).
2.1.1.4 Cattle
a) Cypermethrin
Two lactating Friesian cows were fed twice daily a diet
containing 0.2 mg 14C-benzyl-labelled cypermethrin/kg feed. Cow I
was treated for 20 days and cow II for 21 days. A control cow was
maintained under identical conditions. The radioactivity in milk,
urine and faeces was measured daily and after 20-21 days of dosing the
cows were sacrificed and blood, major organs and tissues were analysed
for radioactivity. Total recovery of the radioactivity was 97.8%. The
major route of excretion was via the urine (54%) and faeces (43%).
Milk contained 0.5% of the dose. The residues in muscle, blood and
brain were less than 0.005 µg/g. Liver contained 0.006 µg/g, kidney
0.004 µg/g, bile 0.027 µg/g, renal fat 0.011 µg/g and subcutaneous fat
0.009 µg/g.
The urinary metabolites were tentatively identified as
N-(3-phenoxy-benzoyl)glutamic acid, the major metabolite, and
3-(4-hydroxyphenoxy) benzoic acid O-sulphate, the minor metabolite
(ratio 4:1). In faeces 36% was eliminated as unchanged parent compound
(Hutson & Stoydin, 1976).
Two lactating Friesian cows were fed twice daily for 7 days a
diet containing 5 mg 14C-cypermethrin (cyclopropyl-labelled)/kg
feed. A third cow was treated similarly with 14C-benzyl-labelled
cypermethrin. A control cow was maintained under identical conditions.
The radioactivity in the milk, urine and faeces was measured daily.
After 7 days of dosing the cows were killed and their blood, major
organs and tissues were analysed for radioactivity. Total recovery of
radioactivity was 92.4% for the cows dosed with cyclopropyl-labelled
cypermethrin and 89.6% for the cow dosed with benzyl-labelled
cypermethrin. The major route of excretion was similar for both label
compounds. Urinary excretion accounted for about 49% of the dose while
faecal excretion accounted for about 38%. An equilibrium between
ingestion and excretion was reached after 3-4 days. Very low levels of
radioactivity were determined in milk (0.003-0.013 µg/g), tissues
(muscle, blood, brain 0.07 µg/g, liver, kidney 0.13 µg/g and renal and
subcutaneous fat 0.10 µg/g). The urinary metabolites included the
glutamic acid conjugate of 3-phenoxybenzoic acid (68%), 3-phenoxy-
benzoylglycine (16%) and 3-phenoxybenzoic acid (9%). 3-(4-Hydroxy-
phenoxy)benzoic acid and its O-sulfate conjugate appeared to be
present in only small amounts (1%) (Crawford, 1978).
One lactating cow was administered a diet containing 10 mg
14C-benzyl-labelled cypermethrin/kg feed twice daily for 7 days.
Milk, urine and faeces were collected daily for radioassay. The cow
was killed 16 hours after the last dose and samples of fat, muscle,
liver and kidney were analysed. An untreated cow was held as control.
Total recovery was 93%. Radioactivity was rapidly eliminated in equal
proportions in the urine and faeces. Radioactivity in milk was
< 0.2%. Analysis of milk revealed that the radioactivity was due to
unchanged cypermethrin, which was located mostly in the lipophilic
components (cream or butterfat). Radioactivity in tissues was
generally in the order: liver (0.21 µg/g) > kidney (0.11 µg/g) > fat
(0.1 µg/g) > blood (0.04 µg/g) > muscle (0.01 µg/g). The residue in
fat was largely unchanged cypermethrin. Radioactivity in liver and
kidney was due mainly to N-(3-phenoxybenzoyl)glutamic acid. The
liver and kidney metabolites were hydrolysed in hot acid to 3-phenoxy-
benzoic acid and its 4'-hydroxy derivative (Croucher et al., 1980).
b) alpha-Cypermethrin
One lactating cow received orally 125 mg 14C-alpha-cypermethrin
(benzyl labelled)/dose twice daily for 4 consecutive days (target dose
250 mg/day). Another cow received the same dose of unlabelled alpha-
cypermethrin. (The overall calculated daily dietary concentrations
were 19 and 14 mg/kg, respectively). The animals were killed 6 hours
after the last dose. Urine, faeces, milk, kidneys, liver, fat and
muscle were analysed for radioactivity.
The major route of excretion of radioactivity was via the faeces,
accounting for 34% of the total administered dose. Urinary excretion
accounted for 23% of the dose and milk less than 1%. Total radioactive
residues in milk accounted for 0.014 mg/kg on day 2 and rose to
0.2 mg/kg by day 4 of dosing. The major proportion (93%) of the milk
residues was confined to the cream fraction. Residues in tissues were
highest in liver, renal fat, omental fat, subcutaneous fat and kidney
(0.56, 0.48, 0.43, 0.39 and 0.22 mg/kg, respectively). Plasma
contained 0.08 mg/kg and muscle samples contained < 0.03 mg/kg
residues.
The liver and kidney contained a range of components. The liver
extract contained at least eight metabolites with a broad range of
polarities. One component (16%) had similar chromatographic properties
to alpha-cypermethrin. The kidney extract contained at least nine
metabolites with a broad range of polarities, one component (20% of
the profile) had similar chromatographic properties to alpha-
cypermethrin. Muscle, fat and milk contained mainly a single
component (muscle 85%, fat 91% and milk 97% of the exact profile),
which in each case had similar chromatographic properties to
alpha-cypermethrin. Urinary metabolites were analysed using HPLC.
The two major components (44% and 20%) had identical chromatographic
properties to N-(3-phenoxybenzoyl)glutamic acid and
N-(3-phenoxybenzoyl)glycine, respectively. A minor component
(3%) had identical chromatographic properties to 3-phenoxybenzoic
acid (Dunsire & Gifford, 1993; Morrison & Richardson, 1994).
2.1.2 Effects on liver enzymes
Six Wistar rats/sex were randomly selected from each of the
control and 1000 mg cypermethrin/kg food group at termination of a
2-year feeding study. The activity of hepatic p-nitroanisole
o-demethylase (PNOD) was determined in each of these rats. In male
rats treated with cypermethrin, PNOD activity, expressed as per gram
of liver, was significantly increased (38%). The total PNOD activity
of liver was also increased (30%), but, owing to great inter-animal
variation, statistical significance was not reached. Treated females
exhibited increases in PNOD activity when expressed per gram of liver
(21%) and per whole liver (39%), but only the latter was significant
(Potter & McAusland, 1980).
2.2 Toxicological data
2.2.1 Acute toxicity
Acute toxicity studies of cypermethrin and alpha-cypermethrin are
summarized in Tables 1 and 2.
Cypermethrin-induced signs of toxicity were typical of
cyano-containing pyrethroid intoxication. After oral administration
signs of intoxication included sedation, ataxia, splayed gait, tip-toe
walking, with occasional tremors and convulsions. These signs of
toxicity appeared within a few hours following dosing, and survivors
recovered within 3 days. The acute toxicity after dermal
administration is of a low order (Coombs et al., 1976).
alpha-Cypermethrin-induced clinical signs, like those of
cypermethrin, are typical of cyano-containing pyrethroid. The observed
signs included ataxia, abasia, gait abnormalities, choreoathetosis,
tip-toe walking, and increased salivation, lacrimation, piloerection,
tremor and clonic convulsions. Surviving animals recovered within 7
days (Rose, 1982b, 1983).
2.2.2 Short-term toxicity studies
2.2.2.1 Mice
Groups of eight mice (CD-1)/sex received diets containing 0, 200,
400, 800, 1200 or 1600 mg alpha-cypermethrin/kg feed for 29 days. One
male fed 1600 mg/kg and one female fed 1200 mg/kg were killed in a
moribund condition. These animals showed neurological disturbances.
Mice at 1200 and 1600 mg/kg developed ungroomed coats, ataxia/abnormal
gait, over-activity or hunched posture. At 800 mg/kg some animals
had ungroomed coats and abnormal gait. Body weight (gain) was
dose-relatedly decreased in mice at 1200 and 1600 mg/kg and in females
at 800 mg/kg. A similar, but less marked effect was apparent in males
receiving 800 mg/kg and females receiving 400 mg/kg. Food consumption
was lowered in rats at 1200 and 1600 mg/kg during the first 2 weeks. A
depression in lymphocyte numbers was noted in males receiving 1600
mg/kg. ALAT and ASAT levels were increased in males at 1600 mg/kg.
Urea levels were increased in all dosed males and females, without
clear dose relation. Plasma albumin and A/G ratio were decreased in
males at 1600 mg/kg. Relative kidney weight was increased in males at
800, 1200 and 1600 mg/kg. Relative lung weight was increased in males
at 1600 mg/kg. No macroscopic or microscopic effects were seen. The
NOEL was 400 mg/kg feed, equal to 57 mg/kg bw per day (Green, 1993).
Groups of 12 mice (CD-1)/sex received diets containing 0, 50, 250
or 1000 mg alpha-cypermethrin/kg feed for 13 weeks. Four males at
1000 mg/kg died during week 12 probably due to stress of treatment and
refusal of food and water. The relative kidney weights of these
animals appeared to be slightly higher. One mouse at 250 mg/kg died.
Clinical signs included thin build, ungroomed coat, hair loss and
encrustations of the dorsal body surface in animals receiving
1000 mg/kg, ungroomed haircoat in mice at 250 mg/kg, hair loss and
encrustations in males at 250 mg/kg and hair loss in two males at
50 mg/kg. Body weight gain was markedly lower in mice at 1000 mg/kg
and lower in mice at 250 mg/kg. Food consumption was slightly higher
in mice at 1000 mg/kg. Overall food conversion efficiency of animals
receiving 1000 mg/kg (and to a lesser extent 250 mg/kg) was lower than
that of controls. In males at 1000 mg/kg, PVC, Hb, RBC, total WBC and
leucocyte counts were decreased. ASAT was increased in a dose-
dependent manner in males at 250 and 1000 mg/kg, and glucose was
decreased in males at 1000 mg/kg. Serum AP was increased in females at
1000 mg/kg. Urinary specific gravity was increased in mice at the
highest dose level. In males at 1000 mg/kg relative brain, adrenal,
heart, kidney, liver, spleen, lung and testes weight were increased.
In females relative liver weight was increased at 250 and 1000 mg/kg
and relative brain and spleen weight at 1000 mg/kg. Two males and
eleven females treated at 1000 mg/kg were considered to be emaciated
at necropsy. No histological changes were observed. Owing to the hair
loss seen at 50 mg/kg, a NOEL was not identified (Amyes et al.,
1994).
2.2.2.2 Rats
a) Cypermethrin
In a 5-week feeding study, groups of six rats/sex (Charles River)
received diets containing 0, 25, 100, 250, 750 or 1500 mg cyper-
methrin/kg feed. No effects were found in haematological, macroscopic
or microscopic examinations. In the highest dose group, rats developed
piloerection, nervousness and incoordinated movement from week
2 onwards. Body weight gain, food intake and terminal body weight
were all reduced for both sexes at 1500 mg/kg. Relative liver
weight and blood urea levels were increased at the highest dose level
(only a summary available) (Coombs et al., 1976).
Table 1. Acute toxicity of cypermethrln in animals
Species1 Route Purity2 LD50 Remark Reference
mg/kg bw
Mouse oral 98.1% 88 5% in corn oil Rose, 1982a
Mouse oral ? 82 5% in corn oil Coombs et al., 1976
Mouse oral 98.1% 1126 40% in DMSO Rose, 1982a
Mouse oral 98.1% 657 50% aq. susp. Rose, 1982a
Mouse percutan. 98.1% > 100 5% in corn oil Rose, 1982a
Rat oral ? 251 5% in corn oil Coombs et al., 1976
Rat oral 98.1% 4000 40% in DMSO Rose, 1982a
Rat oral 98.1% 3423 50% aq. susp. Rose, 1982a
Rat oral ? > 2000 techn. conc. Price, 1984
Rat percutan. ? > 2000 techn. conc. Price, 1984
Rat percutan. ? > 1600 40% in xylene Coombs et al., 1976
Rat i.p. ? approx. 40% in corn oil Coombs et al., 1976
2600
Hamster oral ? 203 5% in corn oil Coombs et al., 1976
Guinea-pig oral ? approx. 20% in corn oil Coombs et al., 1976
500
1 Both male and female animals were used in all of the studies
2 cis:trans isomer ratio 51:49; ? - unknown
Table 2. Acute toxicity of alpha-cypermethrin in animals
Species1 Route Purity LD50 Remark Reference
(%) mg/kg bw
or mg/m3
Mouse oral 96.6 35 5% in corn oil Rose, 1982b
Mouse oral 96.6 762 40% in DMSO Rose, 1982b
Mouse oral 96.6 798 50% aq. suspension Rose, 1982b
Mouse percut. 96.6 > 100 5% in corn oil Rose, 1982b
Rat oral techn. 64 50% aq. CMC Gardner, 1993
Rat oral 96.6 4000 40% in DMSO Rose, 1982b
Rat oral 96.6 > 5000 50% aq. suspension Rose, 1982b
Rat dermal techn. > 2000 undiluted Gardner, 1993
Rat inhal 95.6 1590 as aerosol Jackson, 1993
(MMAD 9 µm)
1 Both male and female animals were used in all of the studies
Groups of 12 rats/sex (Wistar) were fed diets containing 0 (24
rats/sex), 25, 100, 400 or 1600 mg cypermethrin/kg feed for 91-95
days. There were no deaths, no clinical signs and no macroscopic or
microscopic changes. In the highest dose group a reduced body weight
gain was observed. During the first week reduced food consumption was
observed in both sexes at 1600 mg/kg. This could be attributed to the
palatability of the diet. During week 13, females at 1600 mg/kg had
reduced food intake. Males at 1600 mg/kg showed decreases in Hb, MCV
and eosinophil counts, and an increase in prothrombin time. In males
at 400 mg/kg there was also a decrease in eosinophil numbers. The mean
plasma urea concentration in the 1600 mg/kg groups was increased.
Relative liver weight was increased in males and females at 1600 mg/kg
and in males at 400 mg/kg. Relative kidney weight was increased in
males at 1600 mg/kg. The NOEL was 100 mg/kg feed, equivalent to
5 mg/kg bw per day (Pickering, 1981).
b) alpha-Cypermethrin
Groups of 10 Wistar rats/sex received diets containing 0 (20
rats/sex), 20, 100, 200, 400 or 800 mg alpha-cypermethrin/kg feed for
5 weeks. Observations included mortality, clinical signs, body weight
and food consumption, haematology, clinical chemistry, urinalysis,
organ weight, and macroscopic and microscopic lesions.
One male at 800 mg/kg died unrelated to treatment and two males
at 800 mg/kg were killed, owing to signs of severe neurological
disturbance. Both sexes at 800 mg/kg produced abnormal gait and
increased sensitivity to noise. Abnormal gait was seen in one male at
400 mg/kg. The mean body weights and food intake of both males and
females fed 400 or 800 mg/kg were significantly lower than those of
controls. In both males and females at 800 mg/kg and in males at
400 mg/kg prothrombin time was increased. In males at 800 mg/kg the
kaolin-cephalin coagulation time was increased and the percentage of
polymorphic neutrophils was increased at 800 mg/kg. In females
platelet count, total leucocyte count and absolute values of
polymorphic neutrophils and lymphocytes were increased, but the Hb and
haematocrit values were decreased. The MCV was decreased at 200, 400
and 800 mg/kg but not in a dose-related manner. Relative brain, liver
and kidney weights were increased in males fed 800 mg/kg and relative
brain and liver weights were increased in males fed 400 mg/kg. In
females at 800 mg/kg relative brain, liver and kidney weights were
increased. One male at 800 mg/kg that was removed from the study
showed scanty axonal lesions of the sciatic nerves. The NOEL was
200 mg/kg feed, equivalent to 10 mg/kg bw per day (Thorpe, 1982).
In a range finding study groups of five male and five female rats
were fed diets containing a-cypermethrin at concentrations of 0, 50,
200, 800 or 1200 mg/kg feed for up to 6 weeks. No effects were found
on food consumption, haematology and clinical chemistry and
microscopy. All rats at 1200 mg/kg and all males at 800 mg/kg were
killed during weeks 2 to 4 because of severe clinical signs. These
signs included high stepping, splayed gait, abasia and hyper-
sensitivity. Cachexia was seen in extreme cases. There were no
significant changes in fore or hind limb grip strength or hind limb
landing foot splay. Female rats fed 800 mg/kg had a lower mean body
weight and food intake compared to controls. These females also had a
lower leucocyte count. Microscopic examination revealed lymphocytolysis
and iymphocyte depletion of the cortical region of the thymus in males
fed 800 mg/kg and males and females at 1200 mg/kg (Fokkema, 1994a).
Groups of 30 male and 30 female Wistar rats received diets
containing 0 (60 rats/sex), 20, 60, 180 or 540 mg alpha-
cypermethrin/kg feed for 90 days. After 6 weeks 10 rats/sex
(controls 20/sex) were sacrificed for interim examinations. Three
males at 540 mg/kg developed an abnormal gait consisting of splayed
hind limbs. Skin sores and fur loss were observed in all males with
the highest incidence at 540 mg/kg and in females at 0 and 540 mg/kg.
Two males, one from controls and one at 540 mg/kg were removed because
of severe skin lesions. Reduced body weight and reduced food
consumption were seen in rats at 540 mg/kg. During the second part of
the study, body weight gain was also reduced in males at 60 and
180 mg/kg, but not in a clearly dose-related manner. Food consumption
was lower at 60 mg/kg than at 180 mg/kg. At termination the Hb value
was decreased in males and females at 540 mg/kg. MCV and MCHC were
decreased in females at 540 mg/kg. Platelet counts were increased in
males and females at 540 mg/kg. The number of lymphocytes was
increased and the number of eosinophils was decreased in males at
540 mg/kg. Urea concentration was increased in females at 540 mg/kg.
AP was decreased in females at 180 and 540 mg/kg. Urinary volume was
decreased in females at 540 mg/kg and specific gravity was increased
in males and females at 540 mg/kg. In females fed 540 mg/kg, relative
spleen, heart and brain weights were increased. In both sexes at
540 mg/kg relative kidney weight was increased and relative liver
weight was increased in both sexes at 180 and 540 mg/kg. Relative
testis weight was increased in males at 540 mg/kg. Histopathological
investigations showed a solitary form of axonal degeneration,
affecting the fibre only, in the sciatic nerve, in two males fed
540 mg/kg, but there were no clinical signs of toxicity. The NOEL was
60 mg/kg feed, equivalent to 3 mg/kg bw per day (Clark, 1982).
2.2.2.3 Rabbits
Occluded dermal applications of 0, 2, 20 or 200 mg cyper-
methrin/kg bw in PEG 300 were made to abraded and intact skin of
groups of 10 male and 10 female NZW rabbits/sex. The applications were
made for 6 h/day, 5 days/week for 3 weeks. No effects were observed on
haematology, clinical chemistry or following macroscopic or micro-
scopic examination. Slight to severe skin irritation was observed
in rabbits at 200 mg/kg bw and slight to moderate skin irritation was
observed in rabbits receiving 2 and 20 mg/kg bw. Decreased food
consumption and body weight gain was observed in rabbits treated with
200 mg/kg bw. Absolute and relative gonad weights were reduced in male
rabbits at 200 mg/kg bw. The NOEL was 20 mg/kg bw (Henderson &
Parkinson, 1981).
2.2.2.4 Dogs
a) Cypermethrin
In a 5-week feeding study, groups of three beagle dogs/sex
received diets containing 0, 15, 150 or 1500 mg cypermethrin/kg feed.
No effects were found on body weight, food consumption, ophthalmo-
scopy, haematology, clinical chemistry, macroscopy, liver weight or
following microscopic examination. In the highest dose group dogs
developed signs of intoxication, including apprehension, diarrhoea
and vomiting, licking and chewing of the paws, whole body tremors, a
stiff exaggerated hind leg gait, ataxia, inappetence, and decreased
body weight gain. Relative thyroid weight and blood urea levels were
increased and blood glucose levels were decreased (only a summary
available) (Coombs et al., 1976).
Groups of beagle dogs (four/sex) were fed cypermethrin (purity
98%) in the diet at concentrations of 0, 5, 50, 500 or 1500 mg/kg feed
for 13 weeks. No mortality occurred. Dogs at 1500 mg/kg developed
diarrhoea, anorexia and tremors as well as ataxia, incoordination and
hyperaesthesia. Also a reduced food intake and body weight gain was
seen. Due to the clinical signs two dogs/sex at 1500 mg/kg had to be
killed. Minor variations in haematology were observed; the kaolin-
cephalin clotting time was consistently lower throughout the study in
female dogs at 500 mg/kg. However, because of the lack of a dose-
response relationship, the variability of this parameter throughout
the study, and the fact that there were no changes in the prothrombin
time, this finding is not considered to be relevant. No effects were
seen on clinical chemistry parameters or on organ weights. Microscopic
examination of tissues and organs revealed a non-specific focal
bronchopneumonia in the lungs of the dogs at 1500 mg/kg. A pink colour
of the optic disc was noted in ophthalmological examinations. The NOEL
was 500 mg/kg feed, equivalent to 12.5 mg/kg bw per day (Buckwell &
Butterworth, 1977).
A further experiment was undertaken to determine the cause of the
pink discoloration, which occurred in all groups. Cypermethrin was fed
to two groups of three male dogs at concentrations of 0 or 500 mg/kg
food for 13 weeks. Specific ophthalmological examinations were made to
evaluate the degree of coloration of the optic disc. At the end of 13
weeks, there were no consistent differences between the colour of the
optic discs of the treated dogs and the controls (Buckwell &
Butterworth, 1977).
b) alpha-Cypermethrin
This study was conducted in two parts.
In the first part one male and one female beagle dog were fed
alpha-cypermethrin in the diet at concentrations of 200 mg/kg feed for
7 days, 400 mg/kg feed for 2 days and 300 mg/kg feed for 7 days. Due
to severe intoxication of the animals receiving 400 mg/kg, dosing was
discontinued after 2 days. The animals were fed the control diet and
in week 3 treatment was commenced at 300 mg/kg. After the third week
the dogs were killed.
Clinical signs including ataxia, body tremors, subdued behaviour,
head nodding, food regurgitation, diminished response to stimuli and
inflammation of gums and tongue were obtained when dogs were dosed
with 300 and 400 mg/kg. Body weight loss was observed at 300 mg/kg.
Examination of the cellular composition of blood and the chemical
components of plasma showed no abnormalities, and no macroscopic
changes were observed.
In the second part one male and one female beagle dog were fed
alpha-cypermethrin in the diet at 300 mg/kg food for 3 days (male dog)
or 4 days (female dog), and then 250 mg/kg food for 7 days. At
300 mg/kg the same effects were obtained as above. At 250 mg/kg only
the female dog developed the clinical signs shown by the dogs treated
with 300 and 400 mg/kg (Greenough & Goburdhun, 1984).
Groups of four beagle dogs/sex received diets containing dose
levels of 0, 30, 90 or 270 (six dogs/sex) mg alpha-cypermethrin/kg
feed for 13 weeks. No effects on mortality, body weight, food
consumption, ophthalmoscopy, haematology, clinical chemistry,
macroscopy, organ weights or microscopy were seen. One female at
270 mg/kg was killed, because of severe head and body tremors, ataxia,
poor limb coordination, inflamed gums and elevated temperature. All
dogs at 270 mg/kg developed marked clinical signs, including body
tremors, head nodding, lip licking, subduedness, ataxia, agitation and
high stepping gait. The NOEL was 90 mg/kg, equivalent to 2.25 mg/kg bw
per day (Greenough et al., 1984).
Four groups of beagle dogs (four/sex) received via the diet 0,
60, 120 or 240 mg alpha-cypermethrin/kg feed dally for 52 weeks. No
effects were observed on body weight, food consumption, ophthalmo-
scopy, haematology, clinical chemistry, urinalysis or organ weights,
and no changes were observed in macroscopic or microscopic examinations.
Two males at 240 mg/kg developed skin reddening on the tail. Abdominal
skin reddening and alopecia were seen in another male at 240 mg/kg and
one female at 120 mg/kg. The tail reddening caused obvious irritation
and resulted in ulceration and necrosis in one male leading to
amputation of part of the tail. The NOEL was 60 mg/kg feed, equivalent
to 1.5 mg/kg bw per day (Dean & Jackson, 1995).
2.2.3 Long-term toxicity/carcinogenicity studies
2.2.3.1 Mice
Groups of mice (70/sex, SPF-Swiss-derived) received diets
containing 0 (2 groups), 100, 400 or 1600 mg cypermethrin/kg feed for
up to 101 weeks. Ten mice/sex were killed after 52 weeks for interim
necropsy. Observations included mortality, clinical signs, body weight
gain, food consumption, haematology, clinical chemistry, organ
weights, and macroscopy and microscopy. Body weight gain of both males
and females at 1600 mg/kg was reduced when compared to the combined
control groups. Several haematological changes, consistent with mild
anaemia, were found in the 1600 mg/kg group at the interim kill, but
not at termination. At interim kill and at termination, an increase in
thrombocytosis and absolute and relative liver weight was seen in
males at 1600 mg/kg. An increase in the incidence of benign
alveologenic tumours was observed in females at 1600 mg/kg, but was
within historical control incidence. In this study the NOEL was
400 mg/kg feed, equal to 57 mg/kg bw per day (Lindsay et al., 1982).
2.2.3.2 Rats
Groups of 48 Wistar rats/sex received diets containing 1, 10, 100
or 1000 mg cypermethrin/kg feed for 2 years. The control group
consisted of 96 rats/sex and was fed untreated diet for 2 years. After
6 and 12 months six rats/sex were sacrificed and after 18 months 12
rats/sex were sacrificed (controls, respectively, 12 and 24 rats/sex).
The only effects observed were reductions in body weight and food
consumption in males and females at 1000 mg/kg. No dose-related
effects were observed on mortality, clinical chemistry, haematology,
clinical chemistry, organ weights, macroscopy or microscopy. No
difference between control and treated groups were found in the
sciatic nerves. There was no increase in compound-related tumours. The
NOEL was 100 mg/kg feed, equivalent to 5 mg/kg bw per day (McAusland
et al., 1978).
2.2.3.3 Dogs
Groups of four beagle dogs/sex (5´-7 months of age) were fed
diets containing 0, 3, 30, 300 or 1000 mg cypermethrin/kg food for 2
years. Groups of four dogs/sex were allocated to a satellite study and
received a diet containing 0, 300 or 1000 mg/kg cypermethrin (data
concerning this satellite group have only been included in the report
to aid interpretation of the results of the main study). Due to severe
signs of intoxication observed at the 1000 mg/kg level, the
concentration was reduced to 750 mg/kg at week 4 of the study and,
when signs of intoxication persisted during weeks 6-8, animals in the
high-dose group were fed a control diet for 10 days to allow them to
recover. Following the 10 days of control diet, the dogs were fed
cypermethrin at a concentration of 600 mg/kg food for the remainder of
the study. No effects were observed on ophthalmoscopy, clinical
chemistry, organ weights, macroscopy or microscopy. No abnormalities
were found in the sciatic nerves, brain or spinal cord. One male dog
in the satellite group fed 1000 mg/kg convulsed and died. Signs of
intoxication appeared within 24 hours of the initiation of dosing and
consisted of licking and chewing of the paws, a stiff high stepping
gait, whole body tremors, head shaking, incoordination, ataxia and, in
some cases, convulsions. These signs were observed at 1000 mg/kg and
to a lesser extent at 750 mg/kg, but not at 600 mg/kg. The body
weights of male dogs in the highest dose group were significantly
lower than the controls, probably due to initial reduction in food
consumption observed at 1000 mg/kg. In the satellite group no effect
on body weight gain was seen. Apart from some occasionally decreased
sodium levels in the males given the highest dose, no consistent
haematological changes were seen. The NOEL was 300 mg/kg feed,
equivalent to 7.5 mg/kg bw per day (Buckwell, 1981).
2.2.4 Reproductive toxicity studies
Groups of 30 rats/sex (Wistar) received diets containing 0, 10,
100 and 500 mg cypermethrin/kg feed for 5 weeks prior to mating and
then throughout pregnancy and lactation for three successive
generations. Two litters were bred per generation. The first litters
were discarded at weaning. Males and females from the second litter
were randomly selected to breed the next generation. A significant
reduction in body weight gain was seen in the male and female parent
rats receiving 500 mg/kg in all three generations. This was correlated
with a reduction in food consumption. Litter size was reduced at
500 mg/kg in the F1a litter at birth and after 7 and 21 days. Litter
weights were reduced at 500 mg/kg in the F1a, litters on days 7, 14
and 21 of lactation. No other effects on fertility or reproduction
parameters were found. The NOEL for maternal and reproduction toxicity
was 100 mg/kg feed, equivalent to 5 mg/kg bw per day (Hend et al.,
1978; Fish, 1979; Thorpe, 1985).
2.2.5 Special studies on embryotoxicity and teratogenicity
2.2.5.1 Rats
a) Cypermethrin
Groups of 25 pregnant female rats (Sprague-Dawley) received by
gavage 0, 17.5, 35 or 70 mg cypermethrin/kg bw per day in corn oil
during days 6 to 15 of gestation. The females were sacrificed on day
21 of gestation for examination of their uterine contents. One female
at 70 mg/kg bw per day was found dead and one female at 70 mg/kg bw
per day was killed for ethical reasons following severe convulsions.
Eleven out of 25 females at the 70 mg/kg bw per day group showed
neurological disturbances (ataxia, convulsions, hypersensitivity to
noise). A dose-related reduction in body weight gain was observed in
the groups given 35 and 70 mg/kg bw per day. There were no indications
of any embryotoxic or teratogenic effects. The NOEL for maternal
toxicity was 17.5 mg/kg bw per day and the NOEL for embryotoxicity was
70 mg/kg bw per day (Tesh et al., 1978).
b) alpha-Cypermethrin
In a range-finding study, five pregnant female Sprague-Dawley
rats received daily, by gavage, 0, 3, 9, 15 or 18 mg alpha-cyper-
methrin/kg bw in corn oil during days 6-15 of gestation. Maternal
body weights, food consumption and clinical observations were
recorded. On day 20 of gestation the females were killed and the
fetuses were weighed, sexed and externally examined. Four dams at
18 mg/kg bw per day and one dam at 15 mg/kg bw per day showed hindlimb
splay and unsteady gait during dosing. Mean body weight gain was
reduced in a dose-related manner at 9, 15 and 18 mg/kg bw per day and
at 15 and 18 mg/kg bw per day food consumption was reduced. No other
treatment-related abnormalities were observed (Irvine & Twomey, 1994).
In another study, groups of 24 pregnant female Sprague-Dawley
rats received by gavage 0, 3, 9 or 18 mg alpha-cypermethrin (purity
95.6%)/kg bw per day in corn oil during days 6-15 of gestation.
Following marked clinical signs of toxicity the dose level of 18 mg/kg
bw per day was lowered to 15 mg/kg bw per day on day 10 of gestation.
Clinical signs, body weights and food consumption were recorded. On
day 20 of gestation the females were killed and necropsied. The
fetuses were weighed, sexed and examined for external, visceral and
skeletal abnormalities.
Females at 18 mg/kg bw per day showed unsteady gait,
piloerection, limb splay and hypersensitivity to sound. After
reduction of the dose level the signs were similar but less marked.
After treatment with 18/15 mg/kg bw per day a lowered body weight gain
and food consumption was seen. At 9 mg/kg bw per day a slight body
weight reduction was seen. Mean fetal weights were slightly reduced at
18/15 mg/kg bw per day. There was no indication for teratogenicity.
The NOEL for maternal and fetal toxicity was 9 mg/kg bw per day
(Irvine, 1994c).
2.2.5.2 Rabbits
a) Cypermethrin
In a range-finding study, groups of four female pregnant rabbits
(NZW) received during days 6 to 18 of gestation by gavage 0, 25, 50,
100 or 120 mg cypermethrin/kg bw per day in corn oil. The dams were
sacrificed on day 29 of gestation. No adverse effects were seen in the
mothers and fetuses (Tesh et al., 1984a).
Groups of 16 pregnant NZW rabbits received by gavage 0, 20, 50 or
120 mg cypermethrin/kg bw per day in corn oil during days 6 to 18 of
gestation. The dams were killed on day 29 of gestation. One control
female, three females receiving 20 mg/kg bw per day and two females in
each of the groups receiving 50 and 120 mg/kg bw per day were killed
for ethical reasons. Necropsy revealed evidence of respiratory tract
infection and/or gastrointestinal tract infection not related to the
substance. Two females at 20 mg/kg bw per day and two females at
120 mg/kg bw per day aborted during the post-treatment phase of the
investigation. The number of implantations, live young and
resorptions, pre- and post-implantation losses, and fetal and
placental weights were unaffected by treatment. There was no
indication for embryotoxicity or teratogenicity. The NOEL for
embryo-toxicity was 120 mg/kg bw per day (Tesh et al., 1984b, 1988).
Groups of pregnant rabbits (20 rabbits/group, 30 rabbits were
used as an additional control group) were administered cypermethrin
dissolved in corn oil at dose levels of 0, 3, 10 or 30 mg/kg bw per
day orally from days 6 to 18 of gestation. On day 28 of gestation the
rabbits were killed and examination was made of live fetuses, dead
fetuses, resorption sites and corpora lutea. Live fetuses were
maintained for 24 hours to assess viability. Fetuses were also
examined for gross somatic and skeletal deformities. There was no
significant mortality or difference in weight gain during the period
of gestation. There were no significant differences between control
and test groups with respect to pregnancy, fetal death and survival.
Although a wide range of skeletal and visceral abnormalities was found
in the course of the study, there were no differences between control
and test groups with respect to abnormalities. It was concluded that
oral dosing up to 30 mg/kg bw during the major period of organogenesis
resulted in no teratogenic effects in offspring (FAO, 1980).
b) alpha-Cypermethrin
In a range finding study groups of five mated female NZW rabbits
received by gavage 0, 5, 15, 25 or 30 mg a-cypermethrin/kg bw per day
as solutions in corn oil, during days 7-19 of gestation. On day 28 of
pregnancy the females were killed and a necropsy was performed. The
fetuses were weighed, sexed and externally examined. One female each
at 15 and 25 mg/kg bw per day was killed prematurely. At 25 and
30 mg/kg bw per day marked reductions in body weight and food
consumption were seen. There was no indication for either
embryotoxicity or teratogenicity (Irvine, 1994a).
In another study, groups of 16 pregnant NZW rabbits received by
gavage 0, 3, 15 or 30 mg alpha-cypermethrin/kg bw per day in corn oil
during days 7-19 of gestation. Maternal clinical signs, body weight
and food consumption were recorded. The females were killed on day 28
of pregnancy. The uterus was weighed and the numbers of corpora lutea,
implantations and live fetuses were counted. The fetuses were weighed,
sexed and examined for external, visceral and skeletal abnormalities.
Two control females, three at 15 mg/kg bw per day and two at
30 mg/kg bw per day were killed, because of severe weight loss and low
food consumption. One female at 15 mg/kg bw aborted on day 28. In all
groups, including controls, there was a similar mean body weight loss
after the onset of dosing, which continued until day 11. At 30 mg/kg
bw per day there was a further reduction in mean body weight gain
towards the end of the dosing period. Food consumption reflected the
changes in mean body weight gain. The NOEL for maternal toxicity was
3 mg/kg bw per day and the NOEL for embryotoxicity was 30 mg/kg bw.
There was no indication for teratogenicity (Irvine, 1994b).
2.2.6 Special studies on genotoxicity
Results of genotoxicity studies carried out cypermethrin and
alpha-cypermethrin are summarized in Tables 3 and 4.
2.2.7 Special studies on neurotoxicity
2.2.7.1 Rats
a) Cypermethrin
Groups of 6 or 12 rats/sex were administered single oral doses of
100, 200 or 400 mg cypermethrin/kg bw (purity 97%) as a 5% dispersion
in corn oil. The observation period was 9 days. The rats were then
killed and examined histologically. All rats showed signs of
intoxication. At 400 mg/kg bw, within 4 hours of dosing, rats
developed signs of intoxication, including coarse tremors, spasmodic
movements of the body and tail and bleeding from the nose. Tip-toe
walking was also seen in some rats. All animals, except one, were
killed, owing to the severity of the signs. Histological examination
revealed swelling of the myelin sheaths and breaks of some of the
axons of the sciatic nerves. At 200 mg/kg bw similar effects were
observed; eight rats of each sex died or were killed within 48 h of
dosing. The remaining four rats survived the observation period. At
100 mg/kg bw all animals survived the 9 days. One female out of 12
showed minimal lesions in the sciatic nerve in this group. A NOEL
could not be determined (Carter & Butterworth, 1976).
In a neuromuscular dysfunction test, Wistar rats (10/sex) were
treated by gavage with 0, 25, 50, 100, 150 or 200 mg cypermethrin/kg
bw per day in DMSO for 7 consecutive days. The rats were killed 3-4
weeks after the start of dosing, and right and left sciatic/posterior
tibial nerves were analysed. Neuromuscular function was assessed by
means of the inclined plane test and peripheral nerve damage by
reference to ß-glucuronidase and ß-galactosidase activity increases in
nerve tissue homogenates.
Table 3. Results of mutagenicity assays on cypermethrin
Test system Test object Concentration Purity Results References
(%)
In vitro
Gene mutations S. typhimurium 0.2-2000 93.5 negative2 Brooks,
assay TA1525, TA100, µg/plate1 1980
TA1538, TA98, Dean,
TA1537 1981
Gene mutations E.coli WP2 or 0.2-2000 93.5 negative2 Dean,
assay WP2 uvrA µ/plate1 1981
Mitotic gene Saccharomyces 0.01-5.0 93.5 negative Dean,
conversions cerevisiae JD1 mg/litre1 1981
assay
Host-mediated S. cerevisiae 25 and 50 93.5 negative Brooks,
assay JD1 mg/kg bw 1980
in mice
Cell BHK 21/C113 31.25-250 93.5 negative Dean,
transformation cells µg/ml1 1981
assay
Chromosomal RL4 liver cells 7.5-30 µg/ml 93.5 negative Dean,
aberrations 1981
assay
Table 3. Results of mutagenicity assays on cypermethrin (cont'd).
Test system Test object Concentration Purity Results References
(%)
In vivo
Chromosome Chinese hamster 2x oral dose of ? negative Dean,
aberration 20 or 40 mg/kg 1977
assay in bone bw (2 successive
marrow days)
Dominant mouse single oral dose ? negative Dean
lethal assay of 6.25, 12.5 or et al.,
25 mg/kg bw or 1977
5 daily doses of
2.5 or 5 mg/kg
bw per day
Host-mediated mouse/ orally 0, 25, 50 ? negative JMPR,
assay S. cerevisiae mg/kg 1979
DNA damage CD rats m 100 mg/kg, f 150 ? negative Creedy &
assay mg/kg; 1, 4, 16 Wooder,
hour exposure 1977
Wistar rats m 300 mg/kg; 1, 4, negative Creedy &
16 hour exposure Wooder,
f 450 mg/kg, 1 hour 1977
exposure f 337.5
mg/kg, 4 and 16
hour exposure
1 with and without metabolic activation
2 at 200 and 2000 µg/plate, formation of visible droplets in the top agar was seen
Table 4. Results of mutagenicity assays on alpha-cypermethrin
Test system Test object Concentration Purity Results References
(%)
In vitro
Gene mutations S. typhimurium 31.5-5000 95.6 negative Brooks &
assay1 TA98, TA100, µg/ml (no toxicity) Wiggins,
TA1535, TA1537 1992
TA1538
E. coli WP2 uvrA
Gene mutations S. cerevisiae 31.25-4000 95.8 negative Brooks,
assay1 XV185-14C µg/ml 1984
Gene mutations1 L5178Y mouse 3.3-50 µg/ml 95.4 negative Vanderwaart,
assay lymphoma cells 1994
Chromosomal human peripheral -act: 93.75-1000 95.6 negative Brooks &
aberrations lymphocytes µg/ml precipitation Wiggins,
assay1 +act: 125-1000 was seen 1993
µg/ml
In vivo
Chromosomal rat femoral single oral dose 95.8 negative Clare &
aberrations bone marrow 2-8 mg/kg3 Wiggins,
1984
Table 4. Results of mutagenicity assays on alpha-cypermethrin (cont'd).
Test system Test object Concentration Purity Results References
(%)
Micronucleus mouse single oral dose 95.4 negative Vanderwaart,
assay 1-10 mg/kg bw 1995
Alkaline rat single oral dose 96.5 negative Wooder,
elution 40 mg/kg, 6 1981
analysis hours exposure
assay2
1 With and without metabolic activation
2 The effect of alpha-cypermethrin on the integrity of rat liver DNA was investigated
by this method
3 Initially doses of 10, 20 or 40 mg/kg were used, but the animals exhibited severe
signs of toxicity and the experiment was terminated at these doses. Surviving
females were evaluated for chromosomal damage and none was observed
At 200 mg/kg bw per day 50% of the males and 62.5% of the females
died, at 150 mg/kg bw per day two females and one male died and at
100 mg/kg bw per day one female died but no males. No mortalities
occurred in the other groups. A dose-related (in severity and
duration) increase was seen in clinical signs at doses > 100 mg/kg bw.
These signs included salivation, ataxia, splayed hind limb gait,
hyperexcitability to auditory stimuli, tremor and choreoathetosis. At
the same dose levels body weight gain was also reduced.
A dose-related transient functional impairment was found in rats
treated with cypermethrin in the inclined plane test. This effect was
maximal at the end of the 7-day subacute dosing regimen. At doses
which caused mortality, significant increases in ß-glucuronidase and
ß-galactosidase were found 3-4 weeks after the start of dosing in the
distal portion of the sciatic/posterior tibial nerves. There was no
direct correlation between the time course of the neuromuscular
function and the neurobiochemical changes. The NOEL was 50 mg/kg bw
per day (Rose & Dewar, 1983).
The effects of some pyrethroids, including cypermethrin, on
amplitude and pre-pulse inhibition of the acoustic startle reflex were
studied in male Wistar rats. The pyrethroids were suspended in corn
oil. Cypermethrin was given orally at dose levels of 0, 0.5, 1 or
2 mg/kg bw to 12 males. Each animal received all doses of cyper-
methrin. The intersession interval was one week. Animal behaviour
was observed by the experimenter before and after the test session for
a period of 10 minutes and the effects of the pyrethroids on overt
behaviour were measured by scoring the presence of pawing and
salivation, burrowing, hyperactivity, hyperreactivity to an external
stimulus, fine tremors of low intensity and fluid loss. Neither
cypermethrin nor the other pyrethroids tested affected the amplitude
or the latency of the startle reflex (Hijzen et al., 1988).
b) alpha-Cypermethrin
The acute neurotoxicity of alpha-cypermethrin was studied in
Crl:CD:BR rats in two separate acute studies, each using four groups
of ten animals/sex or five rats per sex (additional study). The groups
received a single dose of 0, 4, 20 or 40 mg alpha-cypermethrin/kg bw
in corn oil. During the 14-day observation period, clinical signs and
body weight were analysed. In the main study a detailed clinical
assessment for neurotoxicological effects was performed. This included
a functional observational battery (FOB) and measurements of fore and
hind limb grip strength, hindlimb landing foot splay and motor
activity. In each study five rats/sex were killed on day 15 and brain,
eyes, muscle, nerves, spinal cord and spinal ganglia were analysed.
One male rat in each of the 20 and 40 mg/kg bw groups of the
additional study was found dead on the day after dosing. Clinical
signs were seen in male rats dosed with 20 and 40 mg/kg bw. The signs
(similar in both studies) developed between 3 to 8 hours after dosing
and resolved by three days after dosing. The signs included abnormal/
splayed gait, thrashing, prostration, vocalization, piloerection,
hunched posture, unkempt appearance, soiled/stained body areas and
diarrhoea. The signs in females were similar but lower in frequency.
In addition to these signs there were also isolated cases of
twitching, tremors, abasia, hypersensitivity, pale eyes, soft faeces
and thinning of the fur.
During FOB conducted 5 hours after dosing, gait abnormalities and
clinical signs of increased reactivity were seen in most male rats
dosed with 20 and 40 mg/kg bw. In females the signs were less
frequent. In the 20 and 40 mg/kg bw groups there was a increase in
very slight to slight sporadic fibre degeneration in the sciatic
nerve. The changes were more frequent in the proximal than in the
distal part of the nerve. The NOEL was 4 mg/kg bw (Fokkema, 1994b).
c) Cypermethrin and alpha-cypermethrin
This experiment was performed in two phases. The first phase was
conducted to determine the time course for neurochemical changes in
Wistar rats occurring in the sciatic/posterior tibial nerve (SPTN),
trigeminal nerve and trigeminal ganglion following treatment with
cypermethrin for 5 days/week for 4 weeks. Cypermethrin was
administered at 150 mg/kg bw per day in DMSO (reduced to 100 mg/kg bw
per day in arachis oil, after 10 doses, because of mortality),
alpha-Cypermethrin was dosed at 37.5 mg/kg bw per day in DMSO (also
reduced after 10 days to 25 mg/kg bw per day in arachis oil). Five
animals per sex, treated with either cypermethrin and alpha-cyper-
methrin, were killed at 2, 3, 4, 5, 6, 8, 10 or 12 weeks and examined.
Dosing resulted in the death of 56% of the cypermethrin-treated
animals and 21% of the alpha-cypermethrin-treated animals. The most
frequent signs of intoxication included abnormal gait, ataxia,
lethargy, chromodacryorrhoea, salivation and hypersensitivity to
sensory stimuli. The ß-glucuronidase and ß-galactosidase activities in
the SPTN were increased at 5, 6 and 8 weeks, when compared to
controls. The increase was maximal after 5 weeks, and after 12 weeks
was comparable to controls. No significant enzyme changes were found
in the trigeminal ganglia and trigeminal nerve of treated animals.
Phase 2 was conducted to establish the dose level which did not
cause peripheral nerve degeneration in the SPTN, trigeminal nerve and
ganglia. Groups of 10 rats/sex were dosed with 37.5, 75 or 150 mg
cypermethrin/kg bw per day in DMSO or 10, 20 or 40 mg alpha-cyper-
methrin/kg bw per day in DMSO, 5 days/week for 4 weeks. A control
group of 10 animals was used.
Signs of intoxication similar to those reported in phase 1 were
seen at the highest dose levels. A large increase in ß-glucuronidase
and ß-galactosidase activities in the SPTN was seen at 150 mg/kg bw
per day cypermethrin and 40 mg/kg bw per day alpha-cypermethrin. In
the groups administered 75 mg/kg bw per day cypermethrin or 20 mg/kg
bw per day alpha-cypermethrin a small increase in ß-galactosidase was
found in both the distal and proximal sections of the SPTN. The
magnitude of the enzyme changes was similar to those of phase 1.
Significant enzyme changes were also found in the trigeminal ganglia
and to a lesser extent in the trigeminal nerve of the groups
administered 75 or 150 mg/kg bw per day cypermethrin and 20 or
40 mg/kg bw per day alpha-cypermethrin. The NOELs were 37.5 mg/kg bw
cypermethrin and 10 mg/kg bw alpha-cypermethrin (Rose, 1983).
2.2.7.2. Chickens
In a delayed neurotoxicity study, six adult domestic hens
received 1000 mg cypermethrin/kg bw per day in DMSO for 5 days. After
3 weeks the dosing regime was repeated and a further three weeks later
the birds were killed. A positive (tri- ortho-tolyl phosphate) and
negative control (not dosed) group were used.
None of the cypermethrin-treated hens developed any signs of
intoxication. Histological examination of the nervous system revealed
no lesions. All birds receiving the positive control developed
clinical signs of neurological damage within 15 days and became
progressively more unsteady and ataxic thereafter. Histological
examination of these animals showed lesions in the cerebellum, sciatic
nerve and spinal cord, including axonal and myelin degeneration
(Owen & Butterworth, 1977).
2.2.7.3 Hamsters
The 1979 JMPR (FAO, 1980) evaluated some neurotoxicity studies
with cypermethrin in hamsters. At doses of > 794 mg/kg bw, all
treated hamsters showed clinical signs of poisoning, including
tremors, abnormal irregular movements and an unusual gait. As in the
cases of rats, axon and myelin degeneration was noted in all groups
treated. The lesions included swelling and breaks in the axons and
clumping of myelin.
Hamsters treated orally with a single dose of 40 mg/kg bw,
followed by four doses of 20 mg/kg bw, developed weight loss and
sometimes mortality. There was loss of fur and dermal ulceration.
There was no effect in the mean slip angle experiment, and a marginal
increase in ß-galactosidase was observed in peripheral nerve.
Hamsters treated orally with doses of 5, 10 or 20 mg/kg bw per
day for 5 days showed no mortality. Lower body weight gain was
observed at 20 mg/kg bw per day. One female (out of 5) at the highest
dose developed hyperexcitability. There was a significant deficit in
the mean slip angle test, females showing an earlier dose-related
deficit than noted in males. ß-Galactosidase activity was increased at
all dose levels 3 weeks after the onset of the experiment. This effect
was significant at the two highest dose levels. In this experiment
dermal irritation and fur loss were noted.
In another experiment hamsters were orally treated with
30 mg/kg bw per day for 5 days. There was no mortality and there were
no differences in weight gain. There was some transient skin
irritation accompanied by skin ulceration. One male (out of 16) had an
unusual gait. There was a slight deficit in the inclined plane test
which was noted in the early parts of the experiment. Increases in
both ß-glucuronidase and ß-galactosidase were evident in peripheral
nerve tissue.
2.2.8 Special studies on biochemistry and electrophysiology
In three independent experiments with Wistar rats the effects of
varying doses of cypermethrin (purity 98%, 10% in DMSO) on the
trigeminal ganglion and three sections of the maxillary branch of the
trigeminal nerve (proximal, distal and endings) were determined.
Increases in ß-galactosidase activity in these tissues were taken as
evidence of axonal degeneration.
The three studies involved repeated oral administration of
cypermethrin at 150 mg/kg bw per day for 5 or 7 days and 0, 25, 50,
100 or 200 mg/kg bw per day for 5 or 7 days. Mortality occurred in
animals receiving 100 mg/kg bw per day or more. A dose-related
transient functional impairment, assessed by means of the inclined
plane test was found in the first week. Significant increases in
ß-glucuronidase and ß-galactosidase activity of the sciatic, tibial or
trigeminal nerves only occurred with 5 or 7 doses of 150 or
200 mg/kg bw per day. Increased activity of the enzymes in the distal
portion of nerves was found, but even in the most severely intoxicated
animals the magnitude of this increase was less than that induced by
the known neurotoxic agent methylmercury chloride (Dewar & Moffett,
1978).
Cypermethrin (1:1 cis:trans) was administered to male and female
rats at dose levels ranging from 25 to 200 mg/kg bw per day for 5
consecutive days by oral intubation as a 10% w/v solution in DMSO. A
dose-related functional deficit was observed when the mean slip angle
test and the landing foot spread test were applied to the animals. The
deficit was maximal from days 6 to 14 after the beginning of
treatment, and complete functional recovery occurred within 4 weeks.
Substantial variation in data from the landing foot spread test was
noted. Data were inconsistent over the course of the study.
ß-Glucuronidase activity was increased in a dose-dependent fashion in
both males and females. The results suggested that cypermethrin
produced a primary axonal degeneration, readily measurable 28 days
after treatment as an increase in ß-glucuronidase activity and in
deficits in specific behavioural-function testing of rats (FAO, 1980).
Electrophysiological studies were performed to determine whether
acute or subacute intoxication with cypermethrin produced changes in
the conduction velocity of slower fibres in peripheral nerves or
alterations in the maximal motor conduction velocity. There was no
evidence to suggest that cypermethrin, at doses that induced severe
clinical signs of intoxication, including ataxia, had any effect on
maximal motor conduction velocity or conduction velocity of the slower
motor fibres in peripheral nerves. Doses used in the study ranged from
a single dose of 200 mg/kg bw to 7 consecutive doses of 150 mg/kg bw
followed by two doses of 400 mg/kg bw. At near-lethal doses there were
no effects on conduction velocity even in the presence of clinical
signs of acute intoxication and at dose levels where previous studies
had shown functional degeneration. These electro-physiological
findings are reflective of motor function, which would suggest that
the physiological and functional deficits observed as a result of
acute intoxication are primarily sensory in nature (FAO, 1980).
2.2.9 Special studies on sensitization
2.2.9.1 Cypermethrin
Two out of 20 guinea-pigs developed a positive reaction to
cypermethrin in the Magnusson Kligman test, indicating that
cypermethrin is not a sensitizer (Coombs et al., 1976).
2.2.9.2 alpha-Cypermethrin
No positive reactions were obtained in a Magnusson-Kligman test
performed with guinea-pigs (Gardner, 1993).
2.2.10 Special studies on skin and eye irritation
2.2.10.1 Cypermethrin
A single application of undiluted technical cypermethrin was
moderately irritant to occluded rabbit skin (Coombs et al., 1976).
A single application of undiluted technical cypermethrin to
rabbit eyes produced a mild transient conjunctivitis and blepharism
lasting 2 days (Coombs et al., 1976).
2.2.10.2 alpha-Cypermethrin
Six NZW rabbits receiving a semi-occlusive topical application
with 500 mg alpha-cypermethrin technical developed very slight
erythema in two animals up to 72 hours after removal of the dressings.
There were no other dermal reactions (Gardner, 1993).
Six NZW rabbits receiving an instillation of 0.1 ml (equivalent
to 45 mg) alpha-cypermethrin technical developed slight conjunctival
redness and ocular discharge up to 72 hours after treatment. No cornea
or iris irritation was observed (Gardner, 1993).
2.3 Observations in humans
The symptoms and signs of acute poisoning resulting from
pyrethroids are very similar. Apart from the irritative symptoms of
the skin and respiratory tract (or digestive tract in ingestive
poisoning), acute pyrethroid poisoning is clinically characterized by
abnormalities of nervous excitability.
Occupationally exposed people had abnormal skin sensations
described as burning, itching or tingling, which could be exacerbated
by sweating or washing and readily disappeared after several hours.
Systemic symptoms included dizziness, headache, nausea, anorexia and
fatigue. Vomiting was more prominent in patients with ingestive
poisoning than in occupational poisoning cases. Other symptoms such as
chest tightness, paraesthesia, palpitation, blurred vision and
increased sweating were less frequently seen. The more serious cases
developed coarse muscular fasciculations in large muscles or
extremities (Van den Bercken & Vijverberg, 1989; He et al., 1989).
Urine obtained from operators spraying cypermethrin in
experimental trials was analysed for the presence of the chlorinated
cyclopropane carboxylic acid metabolite. This metabolite was observed
in the urine of exposed workers at levels up to 0.4 µg/ml (the limit
of detection was estimated to be 0.05 µg/ml).
Cypermethrin sprayers were found to have residues on the exposed
parts of their bodies. The rate of dermal exposure of the operators
during spraying ranged from 1.5 to 46.1 mg/hour. There was a
reasonable relationship between the total cypermethrin deposited
dermally and the excretion in urine. The levels of the cyclopropane
carboxylic acid metabolite in the 24-hour urine were between <0.05
and 0.32 mg (not specified). This, together with the finding of 0.6 mg
(not specified) of this metabolite in 72-hour urine from one man, led
to the estimation that approximately 3% of the total dermal dose was
absorbed and rapidly excreted by the operators (FAO, 1980).
In a controlled experiment with sprayers, no abnormalities were
found in clinical and neurological examinations, blood chemistry or
peripheral nerve function tests (including the trigeminal nerve). In
some electroneurophysiological tests (motor conduction velocity, slow
fibre conduction velocity and cornea reflex), a significant change
within the normal range appeared to exist for the group sprayers
between pre- and post-exposure measurements. These changes probably
reflect seasonal variations (FAO, 1982).
3. COMMENTS
The Committee considered toxicological data on cypermethrin and
alpha-cypermethrin, including the results of acute, short-term, and
reproductive studies, and studies on pharmacokinetics and metabolism,
genotoxicity, long-term toxicity/carcinogenicity and neurotoxicity.
Results of effects in humans were also considered.
Cypermethrin is a mixture of four cis and four trans isomers. The
cis isomers are more biologically active and more persistent than the
trans isomers, alpha-Cypermethrin is a mixture of the two most active
cis isomers. A typical cypermethrin sample contains 25% alpha-
cypermethrin. Cypermethrin and alpha-cypermethrin are alpha-cyano or
type II pyrethroids that cause neurotoxicity in mammals and insects.
They affect nerve membrane sodium channels, causing a long-lasting
prolongation of the normally transient increase in sodium permeability
of the membrane during excitation. At high dose levels, these type II
pyrethroids induce salivation and tremors that progress to
characteristic clonic-tonic convulsions (choreoathetosis and
salivation syndrome).
After oral administration, cypermethrin is readily absorbed,
distributed and excreted in rats, chickens, sheep and cattle.
Cypermethrin is primarily eliminated in urine and faeces in about
equal proportions. Less than 1% is excreted in milk. When cypermethrin
was applied dermally to sheep, 2.5% was eliminated in urine and faeces
within 6 days and after an oral dose about 60% was eliminated within 2
days.
Studies in cattle indicated that absorption, distribution and
excretion were comparable for cypermethrin and alpha-cypermethrin. The
major metabolic route for both cypermethrin and its isomers, including
alpha-cypermethrin, is cleavage of the ester bond followed by
hydroxylation and conjugation of the cyclopropyl and phenoxybenzyl
portions of the molecule. The data suggest that there is no isomeric
interconversion during metabolism.
The acute oral toxicity of cypermethrin and alpha-cypermethrin is
moderate to high. WHO has classified these substances as "moderately
hazardous" (WHO, 1996). In rats and mice, the oral LD50 ranges from
82 to 4000 mg/kg bw for cypermethrin and from 35 to > 5000 mg/kg bw
for alpha-cypermethrin, depending on the vehicle used. At lethal or
near lethal doses the signs are typical of type-II pyrethroids and
include salivation, ataxia, gait abnormalities and convulsions.
Several oral short-term toxicity studies with cypermethrin were
available. Cypermethrin has been tested in rats (5 weeks and 90 days)
and dogs (5 weeks and 13 weeks) at dose levels ranging from 25 to
1600 mg/kg feed (equivalent to 1.25-80 mg/kg bw per day) and 5 to
1500 mg/kg feed (equivalent to 0.125-37.5 mg/kg bw per day),
respectively. In these studies, the clinical signs included ataxia,
abnormal gait, nervousness, and, particularly in dogs, inappetence,
diarrhoea, vomiting and hyperaesthesia. In both rats and dogs,
cypermethrin caused decreases in body weight gain, food intake, and a
number of haematological parameters, increases in some organ weights
and plasma urea levels, and, at lethal or near-lethal doses, effects
on the nervous system. For cypermethrin the lowest NOEL in short-term
studies was in a 90-day study with rats administered 25, 100, 400 or
1600 mg/kg feed (equivalent to 1.25-80 mg/kg bw per day). Male rats
given 1600 mg/kg feed showed decreases in haemoglobin concentration,
mean corpuscular volume and eosinophil numbers, and increases in
prothrombin time, plasma urea levels and relative liver and kidney
weights. The decrease in eosinophil numbers and increase in relative
liver weight were also observed in males at 400 mg/kg feed. In female
rats, reduced food intake and increased relative liver weight were
noted in rats given 1600 mg/kg feed. The NOEL in this study was
100 mg/kg feed, equivalent to 5 mg/kg bw per day.
alpha-Cypermethrin was tested in oral short-term toxicity studies
with mice (29 days and 13 weeks), rats (5 weeks, 6 weeks and 90 days)
and dogs (13 weeks and 52 weeks) at dose levels ranging from 50 to
1600 mg/kg feed (equivalent to 7-240 mg/kg bw per day), 20 to
1200 mg/kg feed (equivalent to 1.25-60 mg/kg bw per day) and 30 to
270 mg/kg feed (equivalent to 0.75-6.75 mg/kg bw per day),
respectively. In these studies, alpha-cypermethrin caused the same
effects as described for cypermethrin in the short-term studies. The
signs of toxicity included ataxia, abnormal gait, increased
sensitivity to noise, hyperactivity, hunched posture and, as
demonstrated histologically, axonal degeneration of the sciatic
nerves. For alpha-cypermethrin the lowest NOEL was in a 52-week study
with dogs. In this study a diet containing 60, 120 or 240 mg/kg feed
(equivalent to 1.5 to 6 mg/kg bw per day) was administered. Dogs given
120 and 240 mg/kg feed showed skin reddening, ulceration and necrosis.
The NOEL in this study was 1.5 mg/kg bw per day.
A three-generation reproductive toxicity study with cypermethrin
was performed in rats at dose levels of 10, 100 or 500 mg/kg feed
(equivalent to 0.5-25 mg/kg bw per day). At the highest dose a
reduction in body weight gain and food consumption and a concomitant
reduction in litter size and weight were seen in the F1a progeny
only. No other effects on fertility or reproduction parameters were
observed. The NOEL was 100 mg/kg feed, equivalent to 5 mg/kg bw per
day.
Cypermethrin did not cause embryotoxicity or teratogenicity in
rats at doses up to 70 mg/kg bw per day or in rabbits at doses up to
120 mg/kg bw per day. alpha-Cypermethrin did not cause embryotoxicity
or teratogenicity in rats at doses up to 9 mg/kg bw per day or in
rabbits up to 30 mg/kg bw per day. The NOELs for maternal toxicity in
rats were 17.5 and 9 mg/kg bw per day for cypermethrin and
alpha-cypermethrin, respectively, while the NOELs for maternal
toxicity in rabbits were 30 and 3 mg/kg bw per day for cypermethrin
and alpha-cypermethrin, respectively.
Cypermethrin and alpha-cypermethrin have been tested in a wide
variety of in vitro and in vivo genotoxicity studies. All of the
results were negative.
Two long-term toxicity/carcinogenicity studies with mice and rats
were available on cypermethrin. Mice received a diet containing 100,
400 or 1600 mg/kg feed (equal to 14-228 mg/kg bw per day) for 101
weeks. At 1600 mg/kg feed reduced body weight gain, changes in
haematological parameters and increased liver weight were observed. No
effects were observed at 400 mg/kg feed, equal to 57 mg/kg bw per day.
In a study in which rats received diets containing 1, 10, 100 or
1000 mg/kg feed (equivalent to 0.05-50 mg/kg bw per day) for 2 years,
the only effects observed were reductions in body weight and food
consumption at 1000 mg/kg feed. The NOEL was 100 mg/kg feed,
equivalent to 5 mg/kg bw per day.
In a two-year toxicity study, dogs received diets containing
3, 30, 300 or 1000 mg cypermethrin/kg feed (equivalent to
0.075-25 mg/kg bw per day). The dose level of 1000 mg/kg feed was
reduced to 600 mg/kg feed owing to severe intoxication. At 300 mg/kg
feed, equivalent to 7.5 mg/kg bw per day, no effects were seen. The
Committee concluded that cypermethrin was not carcinogenic in these
studies.
Long-term toxicity/carcinogenicity or reproductive toxicity
studies were not available on alpha-cypermethrin. The Committee noted
the absence of reproductive toxicity and carcinogenicity associated
with administration of cypermethrin, which contains 25% alpha-
cypermethrin. The Committee also noted the absence of genotoxicity
for either cypermethrin or alpha-cypermethrin, the absence of
carcinogenicity associated with compounds of similar structure and
the similar metabolism and disposition of the two compounds. In view
of the foregoing, the Committee concluded that it was unnecessary to
request the results of long-term toxicity/carcinogenicity or
reproductive toxicity studies on alpha-cypermethrin.
Several studies on the neurotoxicity of cypermethrin and
alpha-cypermethrin in rats were available. In these studies high oral
doses of cypermethrin and alpha-cypermethrin caused clinical signs
that included coarse tremor and spasmodic body and tail movements.
Evidence of axonal damage in the sciatic/posterior tibial nerves and
the trigeminal nerve and ganglion was indicated by significant
increases in ß-glucuronidase and ß-galactosidase in nerve tissue
homogenates, in addition to abnormal neuromuscular function tests. In
the inclined plane test, cypermethrin (in DMSO) caused transient
functional impairment. The lowest NOEL for neurotoxicity was
37.5 mg/kg bw per day for cypermethrin (in DMSO) and 4 mg/kg bw per
day for alpha-cypermethrin (in corn oil), indicating that the toxicity
may be influenced by the vehicle used.
Humans occupationally exposed to cypermethrin developed skin
sensation as a first reaction, followed by systemic effects such as
dizziness, headache, nausea, paraesthesia and increased sweating. In
more serious cases, muscular fasciculations developed in large muscles
or in the extremities. In experiments with operators spraying
cypermethrin, no clinical nervous system abnormalities were observed.
However, exposure levels were not measured.
4. EVALUATION
The Committee established an ADI of 0-50 µg/kg bw for
cypermethrin on the basis of the NOEL of 5 mg/kg bw per day in 90-day,
2-year and reproductive toxicity studies in rats and the application
of a safety factor of 100. The Committee established an ADI of
0-20 µg/kg bw for alpha-cypermethrin on the basis of the NOEL of
1.5 mg/kg bw per day in a 52-week study in dogs and the application of
a safety factor of 100.
5. REFERENCES
Amyes, S.J., Holmes, P., Irving, E.M., Green, C.F., Virgo, D.M., &
Sparrow, S. (1994). Alpha-cypermethrin: preliminary toxicity study by
dietary administration to CD-1 mice for 13 weeks. Unpublished report
No. 92/SHL009/0849 from Pharmaco-LSR Ltd, Suffolk, England. Submitted
to the WHO by Cyanamid, Wayne, NJ. USA.
Brooks, T.M. (1980). Toxicity studies with agricultural chemicals:
Mutagenicity studies with RIPCORD in microorganisms in vitro and in
the host-mediated assay (Experiment Nos 1846 and 1847). Unpublished
report TLGR.80.059 from Shell Toxicology Laboratory (Tunstall).
Submitted to the WHO by Cyanamid, Wayne, NJ, USA.
Brooks, T.M. (1984). Genotoxicity studies with Fastac: the induction
of gene mutation in the yeast Saccharomyces cerevisiae. Unpublished
report No. SBGR.84.117 from Shell Research Limited, Sittingbourne
Research Centre. Submitted to the WHO by Cyanamid, Wayne, NJ, USA.
Brooks, T.M. & Wiggins, D.E. (1992). FASTAC TM: Bacterial mutagenicity
studies. Unpublished report No. SBTR.92.022 from Shell Research
Limited, Sittingbourne Research Centre. Submitted to the WHO by
Cyanamid, Wayne, NJ, USA.
Brooks, T.M. & Wiggins, D.E. (1993). FASTAC TM: In vitro chromosome
studies using cultured human lymphocytes. Unpublished report
No. SBTR.93.007 from Shell Research Limited, Sittingbourne Research
Centre. Submitted to the WHO by Cyanamid, Wayne, NJ, USA.
Buckwell, A.C. (1981). A 2-year feeding study in dogs on WL 43467
(Experiment No. 1412). Unpublished report No. SBGR.81.126 (plus two
addenda) from Shell Toxicology Laboratory (Tunstall). Submitted to the
WHO by Cyanamid, Wayne, NJ, USA.
Buckwell, A.C. & Butterworth, S. (1977). Toxicology studies on the
pyrethroid insecticide WL 43467: a 13-week feeding study in dogs
(Experiment No. 1112). Unpublished report No. TLGR 0127.77 (including
addendum and corrigendum) from Shell Toxicology Laboratory (Tunstall).
Submitted to the WHO by Cyanamid, Wayne, NJ, USA.
Carter, B.I. & Butterworth, S.T.G. (1976). Toxicity of insecticides:
The acute oral toxicity and neuropathological effects of WL 43467 to
rats. Unpublished report No. TLGR.0055.76 from Shell Research,
Sittingbourne Research Centre. Submitted to the WHO by Cyanamid,
Wayne, NJ, USA.
Clare, M.G. & Wiggins, D.E. (1984). Genotoxicity studies with fastac:
in vivo cytogenetic test using rat bone marrow. Unpublished report
No. SBGR.84.120 from Shell Research Limited, Sittingbourne Research
Centre. Submitted to the WHO by Cyanamid, Wayne, NJ, USA.
Clark, D.G. (1982). WL85871: A 90-day feeding study in rats.
Unpublished report No. SBGR.81.293 from Shell Research Limited,
Sittingbourne Research Centre. Submitted to the WHO by Cyanamid,
Wayne, NJ, USA.
Coombs, A.D., Carter, B.I., Hend, R.W., Butterworth, S.G., & Buckwell,
A.C. (1976) Toxicity studies on the insecticide WL 43467: Summary of
results of preliminary experiments. Unpublished report
No. TLGR.0104.76 from Shell Toxicology Laboratory (Tunstall).
Submitted to the WHO by Cyanamid, Wayne, NJ, USA.
Crawford, M.J. (1978). The excretion and residues of radioactivity in
cows treated orally with 14C-labelled WL 43467. Unpublished report
No. TLGR.0029.78 from Shell Toxicology Laboratory (Tunstall).
Submitted to the WHO by Cyanamid, Wayne, NJ, USA.
Crawford, M.J. & Hutson, D.H. (1977). The elimination and retention of
WL 43467 when administered dermally or orally to sheep. Unpublished
report No. TLGR.0098.77 from Shell Toxicology Laboratory (Tunstall).
Submitted to the WHO by Cyanamid, Wayne, NJ, USA.
Crawford, M.J. & Hutson, D.H. (1978). The elimination of residues from
the fat of rats following the oral administration of [14C-benzyl]
WL 43381 (cis-WL 43467). Unpublished report No. TLGR.0078.78 from
Shell Toxicology Laboratory (Tunstall). Submitted to the WHO by
Cyanamid, Wayne, NJ, USA.
Creedy, C.L. & Wooder, M.F. (1977) Studies on the effect of WL 43467
upon the integrity of rat liver cell DNA in vivo. Unpublished report
No. TLGR.0043.77 from Shell Toxicology Laboratory (Tunstall).
Submitted to the WHO by Cyanamid, Wayne, NJ, USA.
Croucher, A., Stoydin, G., Cheeseman, M.E., Buckwell, A.C., &
Gellatly, J.B.M. (1980). The metabolic fate of cypermethrin in the
cow: elimination and residues derived from 14C-benzyl label.
Unpublished report No. TLGR.80.121 from Shell Toxicology Laboratory
(Tunstall). Submitted to the WHO by Cyanamid, Wayne, NJ, USA.
Dean, B.J. (1977). Toxicity studies with WL 43467: Chromosome studies
on bone marrow cells of Chinese hamsters after two daily oral doses of
WL 43467. Unpublished report No. TLGR.0136.77 from Shell Toxicology
Laboratory (Tunstall). Submitted to the WHO by Cyanamid,
Wayne, NJ, USA.
Dean, B.J. (1981). Toxicity studies with agricultural chemicals:
In vitro genotoxicity studies with RIPCORD (Experiment
No. AIR-1820). Unpublished report No. TLGR.80.116 from Shell
Toxicology Laboratory (Tunstall). Submitted to the WHO by Cyanamid,
Wayne, NJ, USA.
Dean, I. & Jackson, F. (1995). WL85871: 52-week oral (dietary)
toxicity study in dogs (IRI Project No. 652238). Unpublished report
No. IRI/11110 from Inveresk Research International, Scotland.
Submitted to the WHO by Cyanamid, Wayne, NJ, USA.
Dean, B.J., Van der Pauw, C.L., & Butterworth S.T.G. (1977). Toxicity
studies with WL 43467: Dominant lethal assay in male mice after single
oral doses of WL 43467. Unpublished report No. TLGR.0042.77 from Shell
Toxicology Laboratory (Tunstall). Submitted to the WHO by Cyanamid,
Wayne, NJ, USA.
Dewar, A.J. & Moffett, B.J. (1978). Toxicity studies on the
insecticide WL 43467: Biochemical studies on the effect of WL 43467 on
the rat trigeminal nerve and ganglion. Unpublished report No.
TLGR.0162.77 from Shell Toxicology Laboratory (Tunstall). Submitted to
the WHO by Cyanamid, Wayne, NJ, USA.
Dunsire, J.P. & Gifford, L.J. (1993). The distribution of
[benzyl-14C]WL85871 (FASTAC) in the lactating cow (nature of residue
study to EPA guidelines - Live phase) (IRI Project No. 153667).
Unpublished report No. IRI/9879 from Inveresk Research International,
Scotland. Submitted to the WHO by Cyanamid, Wayne, NJ, USA.
FAO (1980). Pesticide residues in food-1979. Report of the Joint
Meeting of the FAO Panel of Experts on Pesticide Residues in Food and
the Environment and the WHO Expert Group on Pesticide Residues. Rome,
Food and Agriculture Organization of the United Nations (FAO Plant
Production and Protection Paper, No. 20).
FAO (1982). Pesticide residues in food-1981. Report of the Joint
Meeting of the FAO Panel of Experts on Pesticide Residues in Food and
the Environment and the WHO Expert Group on Pesticide Residues. Rome,
Food and Agriculture Organization of the United Nations (FAO Plant
Production and Protection Paper, No. 37).
Fish, A. (1979). Corrigendum I in Group Research Report
No. TLGR.0188.78. Unpublished data from Shell Toxicology Laboratory
(Tunstall). Submitted to the WHO by Cyanamid, Wayne, NJ, USA.
Fokkema, G.N. (1994a). WL85871 (FASTAC): A 6-week range finding
feeding study in the rat. Unpublished report No. SBTR.93.002 from
Shell Research Limited, Sittingbourne Research Centre. Submitted to
the WHO by Cyanamid, Wayne, NJ, USA.
Fokkema, G.N. (1994b). WL85871 (FASTAC): An acute oral (gavage)
neurotoxicity study in the rat. Unpublished report No. SBTR.92.027
from Shell Research Limited, Sittingbourne Research Centre. Submitted
to the WHO by Cyanamid, Wayne, NJ, USA.
Gardner, J.R. (1993). FASTAC technical: Acute oral and dermal toxicity
in rat, skin and eye irritancy in rabbit and skin sensitisation
potential in guinea pig. Unpublished report No. SBTR.92.033 from Shell
Research Limited, Sittingbourne Research Centre. Submitted to the WHO
by Cyanamid, Wayne, NJ, USA.
Green, C.F. (1993). Alphacypermethrin: Preliminary toxicity study by
dietary administration to CD-1 mice for four weeks. Unpublished report
No. LSR 92/0346 from Life Science Research Limited. Submitted to the
WHO by Cyanamid, Wayne, NJ, USA.
Greenough, R.J. & Goburdhun, R. (1984). WL 85871: Oral (dietary)
maximum tolerated dose study in dogs (IRI Project No. 631087).
Unpublished report No. IRI/3107 from Inveresk Research International,
Scotland. Submitted to the WHO by Cyanamid, Wayne, NJ, USA.
Greenough, R.J., Cockrill, J.B., & Goburdhun, R. (1984).
WL 85871: 13-week oral (dietary) toxicity study in dogs (IRI Project
No. 631092). Unpublished report No. IRI/3197 from Inveresk Research
International, Scotland. Submitted to the WHO by Cyanamid,
Wayne, NJ, USA.
He, F., Wang, S., Liu, L., Chen, S., Zhang, Z. & Sun, J. (1989).
Clinical manifestations and diagnosis of acute pyrethroid poisoning.
Arch. Toxicol., 63, 54-58.
Hend, R.W., Hendy, R. & Fleming, D.J. (1978). Toxicity studies on the
insecticide WL 43367: a three generation reproduction study in rats.
Unpublished report No. TLGR.0188.78 from Shell Toxicology Laboratory
(Tunstall). Submitted to the WHO by Cyanamid, Wayne, NJ, USA.
Henderson, C. & Parkinson, G.R. (1981). Cypermethrin technical:
subacute dermal toxicity study in rabbits (CTL Study No. LB0019).
Unpublished report No. CTL/P/588 from ICI Central Toxicology
Laboratory. Submitted to the WHO by Cyanamid, Wayne, NJ, USA.
Hijzen, T.H., de Beun, R., & Slanger, J.L. (1988). Effects of
pyrethroids on the acoustic startle reflex in the rat. Toxicology,
49, 271-276.
Hutson, D.H. & Stoydin, G. (1976). The excretion of radioactivity from
cows fed with radioactively labelled WL 43467. Unpublished report
No. TLGR.0075.76 from Shell Toxicology Laboratory (Tunstall).
Submitted to the WHO by Cyanamid, Wayne, NJ, USA.
Hutson, D.H. & Stoydin, G. (1987). Excretion and residues of the
pyrethroid insecticide cypermethrin in laying hens. Pestic. Sci.,
18, 157-168.
IPCS (1996) The WHO recommended classification of pesticides by hazard
and guidelines to classification 1996-1997. World Health Organization,
Geneva, p. 23 (Document WHO/PCS/96.3).
Irvine, L.F.H (1994a). Oral (gavage) rabbit developmental toxicity
dose ranging study. Unpublished report No. SLN/3/92 from Toxicol
Laboratories Limited, Ledbury, England. Submitted to the WHO by
Cyanamid, Wayne, NJ, USA.
Irvine, L.F.H. (1994b). Oral (gavage) rabbit developmental toxicity
(teratogenicity) study. Unpublished report No. SLN/4/93 from Toxicol
Laboratories Limited, Ledbury, England. Submitted to the WHO by
Cyanamid, Wayne, NJ, USA.
Irvine, L.H.F. (1994c). Alphacypermethrin. Oral (gavage) rat
developmental toxicity (teratogenicity) study. Unpublished report
No. SLN/2/92 from Toxicol Laboratories Limited, Ledbury, England.
Submitted to the WHO by Cyanamid, Wayne, NJ, USA.
Irvine, L.F.H & Twomey, K. (1994). Alphacypermethrin. Oral (gavage)
rat developmental study. Unpublished report No. SLN/1/92 from Toxicol
Laboratories Limited, Ledbury, England. Submitted to the WHO by
Cyanamid, Wayne, NJ, USA.
Jackson, G.C. (1993). Alphacypermethrin: Acute inhalation toxicity in
rats, 4-hour exposure. Unpublished report No. SLL 266/930770 from
Huntingdon Research Centre, England. Submitted to the WHO by Cyanamid,
Wayne, NJ, USA.
Lindsay, S., Banham, P.B., Chart, I.S., Chalmers, D.T., Godley, M.J.,
& Taylor, K. (1982). Cypermethrin: Lifetime feeding study in mice
(CTL Study No. PM0366). Unpublished report No. CTL/P/687 (including 1
supplement) from ICI Central Toxicology Laboratory. Submitted to the
WHO by Cyanamid, Wayne, NJ, USA.
McAusland, H.E., Butterworth, S.T.G., & Hunt, P.F. (1978). Toxicity
studies on the insecticide WL 43467: A two-year feeding study in rats.
Unpublished report No. TLGR.0189.78 (including 4 corrigenda/addenda)
from Shell Toxicology Laboratory (Tunstall). Submitted to the WHO by
Cyanamid, Wayne, NJ, USA.
Morrison, B.J. & Richardson, K.A. (1994). WL85871 (alphacypermethrin,
FASTAC): The metabolism of 14C-WL85871 after repeated oral dosing in
the lactating cow; in-life phase and metabolite profiling (Experiment
No. 6046). Unpublished report No. SBTR.93.063 from Sittingbourne
Research Centre, Sittingbourne, England. Submitted to the WHO by
Cyanamid, Wayne, NJ, USA.
Owen, D.E. & Butterworth, S.T.G. (1977). Toxicity of pyrethroid
insecticides: Investigation of the neurotoxic potential of WL 43467 to
adult domestic hens. Unpublished report No. TLGR.0134.77 from Shell
Toxicology Laboratory (Tunstall). Submitted to the WHO by Cyanamid,
Wayne, NJ, USA.
Pickering, R.G. (1981). A 90-day feeding study of WL 43467 in rats
(Experiment No. 1806). Unpublished report No. TLGR.80.143 from Shell
Toxicology Laboratory (Tunstall). Submitted to the WHO by Cyanamid,
Wayne, NJ, USA.
Potter, D. & McAusland, H.E. (1980). Toxicity studies on the
insecticide WL43467: A study of liver microsomal enzyme activity in
rats fed WL 43467 for 2 years (Experiment No. 1105). Unpublished
report from Shell Toxicology Laboratory (Tunstall). Submitted to the
WHO by Cyanamid, Wayne, NJ, USA.
Price, J.B. (1984). Toxicology of pyrethroids: the acute oral and
percutaneous toxicity of Ripcord (WL-43467). Technical concentrate
(Project No. SRCAIR84). Unpublished report No. SBGR.84.299 from Shell
Research Limited, Sittingbourne Research Centre. Submitted to the WHO
by Cyanamid, Wayne, NJ, USA.
Rose, G.P. (1982a). Toxicology of pyrethroids: the acute oral and
percutaneous toxicity of WL 85871 (cis-2-Ripcord) in comparison with
Ripcord. Unpublished report No. SBGR.82.130 from Shell Research
Limited, Sittingbourne Research Centre. Submitted to the WHO by
Cyanamid, Wayne, NJ, USA.
Rose, G.P. (1982b). Toxicology of pyrethroids: the acute oral and
percutaneous toxicity of WL 85871 (cis-2-RIPCORD) comparison with
RIPCORD. Unpublished report No. SBGR.82.130 from Shell Research
Limited, Sittingbourne Research Centre. Submitted to the WHO by
Cyanamid, Wayne, NJ, USA.
Rose, G.P. (1983). Neurotoxicity of WL85871. Comparison with WL43467:
The effect of twenty oral doses of WL85871 or WL43467 over a period of
4 weeks on the rat sciatic/posterior tibia nerve, trigeminal nerve and
trigeminal ganglion. Unpublished report No. SBGR.83.185 from Shell
Research Limited, Sittingbourne Research Centre. Submitted to the WHO
by Cyanamid, Wayne, NJ, USA.
Rose, G.P. & Dewar, A.J. (1983). Intoxication with four synthetic
pyrethroids fails to show any correlation between neuromuscular
dysfunction and neurobiochemical abnormalities in rats.
Arch. Toxicol., 53, 297-316.
Tesh, J.M., Tesh, S.A., & Davies, W. (1978). WL 43467: Effects upon
the progress and outcome of pregnancy in the rat. Unpublished report
of Life Science Research. Submitted to the WHO by Cyanamid,
Wayne, NJ, USA.
Tesh, J.M., Ross, F.W., & Wightman, T.J. (1984a). WL 43467: Effects of
oral administration upon pregnancy in the rabbit. 1. Dosage range
finding study. Unpublished test report No. 84/SHL003/014 from of Life
Science Research. Submitted to the WHO by Cyanamid, Wayne, NJ, USA.
Tesh, J.M., Ross, F.W., & Wightman, T.J. (1984b). WL 43467: Effects of
oral administration upon pregnancy in the rabbit. 2. Main study.
Unpublished test report No. 84/SHL004/043 from Life Science Research.
Submitted to the WHO by Cyanamid, Wayne, NJ, USA.
Tesh, J.M., Ross, F.W., & Wightman, T.J. (1988). Supplementary report
to LSR report No. 84/SHL004/043. Unpublished report No. 88/SHL004/775
from Life Science Research. Submitted to the WHO by Cyanamid,
Wayne, NJ, USA.
Thorpe, E. (1982). A 5-week feeding study with WL 85871 in rats
(Experiment No. 2095). Unpublished report No. SBGR.81.212 from Shell
Research Limited, Sittingbourne Research Centre. Submitted to the WHO
by Cyanamid, Wayne, NJ, USA.
Thorpe, E. (1985). Fourth corrigendum/addendum to TLGR.0188.78.
Unpublished report from Shell Research Limited, Sittingbourne Research
Centre (Four volumes). Submitted to the WHO by Cyanamid,
Wayne, NJ, USA.
Van der Bercken, J. & Vijverberg, H.P.M. (1989). Neurotoxicological
effects of pyrethroid insecticides. Unpublished report from the
Department of Veterinary Pharmacology, Pharmacy and Toxicology.
University of Utrecht, The Netherlands. Submitted to the WHO by
Cyanamid, Wayne, NJ, USA.
Vanderwaart, E.J. (1994). Evaluation of the mutagenic activity of
Fastac technical in a in vitro mammalian cell gene mutation test
with L5178Y mouse lymphoma cells (with independent repeat).
Unpublished report No. 087367 from Notox B.V., The Netherlands.
Submitted to the WHO by Cyanamid, Wayne, NJ, USA.
Vanderwaart, E.J. (1995). Micronucleus test in bone marrow cells of
the mouse with Fastac technical. Unpublished report No. 087378 from
Notox B.V., The Netherlands. Submitted to the WHO by Cyanamid,
Wayne, NJ, USA.
WHO (1996). The WHO recommended classification of pesticides by hazard
and guidelines to classification 1996-1997 (WHO/PCS/96.3). Available
from the International Programme on Chemical Safety, World Health
Organization, Geneva, Switzerland.
Wooder, M.F. (1981). Studies on the effect of WL 85871 on the
integrity of rat liver DNO in vivo (Experiment No. 2103).
Unpublished report No. SBGR.81.225 from Shell Toxicology Laboratory
(Tunstall). Submitted to the WHO by Cyanamid, Wayne, NJ, USA.
 2. BIOLOGICAL DATA
2.1 Biochemical aspects
2.1.1 Absorption, distribution and excretion
According to the JMPR (FAO, 1980, 1982) cypermethrin is readily
absorbed and rapidly eliminated via urine and faeces of mice, rats,
dogs, sheep and cows. Absorption from the gastrointestinal tract is
more rapid with the trans isomer than with the cis isomer. The highest
mean concentrations are found in body fat, liver, kidney, muscle, skin
and milk. The clearance rate from adipose tissue is slow and the
elimination half-life in rats and mice ranges from 10 to 20 and from
20 to 30 days, respectively. The data suggest a potential for
bioaccumulation in the body following continuous exposure. With
respect to the chemical and especially the isomeric complexity of the
molecule, the metabolic profile due to all of its isomers is extremely
complex. Cypermethrin is readily cleaved at the ester linkage to
produce the cyclopropane carboxylic acid and a 3-phenoxybenzoyl moiety
that is further metabolised by oxidation at the 4'position. The
resultant phenol is almost totally conjugated with sulfate. The
4'-hydroxy sulfate forms the major aryl metabolite (16%), and
3-phenoxybenzoic acid is the second most important (5%). The other
identified aryl metabolites are 3-(4-hydroxyphenoxy)benzoic acid (1%)
and the glycine conjugate, N-(3-phenoxybenzoyl) glycine (1%).
2.1.1.1 Rats
Eight female Wistar rats were orally dosed with 2.5 mg
14C-benzyl-labelled cis-cypermethrin/kg bw. They were killed in
groups of two at 8, 14, 25 and 42 days after dosing, and the
radioactivity in fat, liver and kidney was measured. Selected fat
samples were analysed for parent compound. Between days 14 and 42,
radioactivity was eliminated from the fat with a half-life of 20-25
days. Residues in liver and kidney were 30 to 40 times lower than
those in fat, but were eliminated at a similar rate. In two pooled fat
samples taken 8 and 24 days after dosing, 90-100% of the radioactivity
was in the form of the parent compound (Crawford & Hutson, 1978).
2.1.1.2 Chickens
Six Warren laying hens were treated daily for 14 days with 10 mg
14C-phenoxy-labelled cypermethrin/kg food (equivalent to 1.25 mg/kg
bw per day) in gelatin capsules in the diet. Eggs and excreta were
collected dally. The hens were killed 4.5 hours after the last dose,
and liver, muscle and fat samples were collected. Of the total
radioactivity, 95.2% was recovered. Radioactivity in eggs plateaued 8
days after the start of dosing and reached 0.05 mg/kg. Most
radioactivity was found in the yolk and was a mixture of parent
compound and material that was closely associated with neutral lipids
and phosphatidyl cholines. In tissues, highest radioactivity was found
in the liver (0.37 µg/g). This radioactivity was composed of parent
compound (0.05 µg/g) and a mixture of very polar metabolites that were
not hydrolysed to significant amounts of 3-phenoxybenzoic acid or its
4-hydroxy derivative. Fat contained 0.08 µg/g and about 60% of the
residue was present as parent compound. Residues in muscle
(0.01-0.02 µ/g) were too low for characterization (Hutson & Stoydin,
1987).
2.1.1.3 Sheep
Two male sheep were each treated dermally (single dose) with a
mixture of 962 mg 14C-cypermethrin (both cyclopropyl- and
benzyl-labelled), equivalent to 21.9 mg/kg bw in acetone. After 24
hours (sheep 1) and 6 days (sheep 2) the animals were killed, and
radioactivity in fat, muscle, liver, kidney and skin (application
area) was measured. Urine and faeces were collected up to the time the
sheep were killed. One sheep treated with acetone served as a control.
Another sheep was treated orally (single dose) with 177 mg 14C-
cypermethrin mixture in a gelatin capsule (equivalent to 3.9 mg/kg
bw). Urine and faeces were collected for 2 days and tissues were
assayed. When applied dermally radioactivity was slowly absorbed
and eliminated. Less than 0.5% was excreted in urine within 24 hours
and only 2% over a 6-day period. Faecal elimination was also slow,
0.5% of the dose being eliminated in 6 days. About 30% of the applied
radioactivity was recovered from the application areas of both sheep.
Very little radioactivity was found in the tissues. In liver, kidney,
renal fat and subcutaneous fat (other than the application area),
residues ranged from 0.09 to 0.3 µg/g and muscle samples contained
0.03 to 0.06 µg/g.
Following oral treatment, the elimination of radioactivity was
rapid, 61% of the administered radioactivity being eliminated within
48 hours. Urinary elimination comprised 41% of the dose and faecal
elimination 20.5%. Residues found in tissues were comparable to those
found after dermal application. Most of the radioactivity in the fat
samples of all sheep was found to be parent compound. In muscle, liver
and kidney samples, only a small percentage of the total tissue
radioactivity was identified as parent compound (Crawford & Hutson,
1977).
2.1.1.4 Cattle
a) Cypermethrin
Two lactating Friesian cows were fed twice daily a diet
containing 0.2 mg 14C-benzyl-labelled cypermethrin/kg feed. Cow I
was treated for 20 days and cow II for 21 days. A control cow was
maintained under identical conditions. The radioactivity in milk,
urine and faeces was measured daily and after 20-21 days of dosing the
cows were sacrificed and blood, major organs and tissues were analysed
for radioactivity. Total recovery of the radioactivity was 97.8%. The
major route of excretion was via the urine (54%) and faeces (43%).
Milk contained 0.5% of the dose. The residues in muscle, blood and
brain were less than 0.005 µg/g. Liver contained 0.006 µg/g, kidney
0.004 µg/g, bile 0.027 µg/g, renal fat 0.011 µg/g and subcutaneous fat
0.009 µg/g.
The urinary metabolites were tentatively identified as
N-(3-phenoxy-benzoyl)glutamic acid, the major metabolite, and
3-(4-hydroxyphenoxy) benzoic acid O-sulphate, the minor metabolite
(ratio 4:1). In faeces 36% was eliminated as unchanged parent compound
(Hutson & Stoydin, 1976).
Two lactating Friesian cows were fed twice daily for 7 days a
diet containing 5 mg 14C-cypermethrin (cyclopropyl-labelled)/kg
feed. A third cow was treated similarly with 14C-benzyl-labelled
cypermethrin. A control cow was maintained under identical conditions.
The radioactivity in the milk, urine and faeces was measured daily.
After 7 days of dosing the cows were killed and their blood, major
organs and tissues were analysed for radioactivity. Total recovery of
radioactivity was 92.4% for the cows dosed with cyclopropyl-labelled
cypermethrin and 89.6% for the cow dosed with benzyl-labelled
cypermethrin. The major route of excretion was similar for both label
compounds. Urinary excretion accounted for about 49% of the dose while
faecal excretion accounted for about 38%. An equilibrium between
ingestion and excretion was reached after 3-4 days. Very low levels of
radioactivity were determined in milk (0.003-0.013 µg/g), tissues
(muscle, blood, brain 0.07 µg/g, liver, kidney 0.13 µg/g and renal and
subcutaneous fat 0.10 µg/g). The urinary metabolites included the
glutamic acid conjugate of 3-phenoxybenzoic acid (68%), 3-phenoxy-
benzoylglycine (16%) and 3-phenoxybenzoic acid (9%). 3-(4-Hydroxy-
phenoxy)benzoic acid and its O-sulfate conjugate appeared to be
present in only small amounts (1%) (Crawford, 1978).
One lactating cow was administered a diet containing 10 mg
14C-benzyl-labelled cypermethrin/kg feed twice daily for 7 days.
Milk, urine and faeces were collected daily for radioassay. The cow
was killed 16 hours after the last dose and samples of fat, muscle,
liver and kidney were analysed. An untreated cow was held as control.
Total recovery was 93%. Radioactivity was rapidly eliminated in equal
proportions in the urine and faeces. Radioactivity in milk was
< 0.2%. Analysis of milk revealed that the radioactivity was due to
unchanged cypermethrin, which was located mostly in the lipophilic
components (cream or butterfat). Radioactivity in tissues was
generally in the order: liver (0.21 µg/g) > kidney (0.11 µg/g) > fat
(0.1 µg/g) > blood (0.04 µg/g) > muscle (0.01 µg/g). The residue in
fat was largely unchanged cypermethrin. Radioactivity in liver and
kidney was due mainly to N-(3-phenoxybenzoyl)glutamic acid. The
liver and kidney metabolites were hydrolysed in hot acid to 3-phenoxy-
benzoic acid and its 4'-hydroxy derivative (Croucher et al., 1980).
b) alpha-Cypermethrin
One lactating cow received orally 125 mg 14C-alpha-cypermethrin
(benzyl labelled)/dose twice daily for 4 consecutive days (target dose
250 mg/day). Another cow received the same dose of unlabelled alpha-
cypermethrin. (The overall calculated daily dietary concentrations
were 19 and 14 mg/kg, respectively). The animals were killed 6 hours
after the last dose. Urine, faeces, milk, kidneys, liver, fat and
muscle were analysed for radioactivity.
The major route of excretion of radioactivity was via the faeces,
accounting for 34% of the total administered dose. Urinary excretion
accounted for 23% of the dose and milk less than 1%. Total radioactive
residues in milk accounted for 0.014 mg/kg on day 2 and rose to
0.2 mg/kg by day 4 of dosing. The major proportion (93%) of the milk
residues was confined to the cream fraction. Residues in tissues were
highest in liver, renal fat, omental fat, subcutaneous fat and kidney
(0.56, 0.48, 0.43, 0.39 and 0.22 mg/kg, respectively). Plasma
contained 0.08 mg/kg and muscle samples contained < 0.03 mg/kg
residues.
The liver and kidney contained a range of components. The liver
extract contained at least eight metabolites with a broad range of
polarities. One component (16%) had similar chromatographic properties
to alpha-cypermethrin. The kidney extract contained at least nine
metabolites with a broad range of polarities, one component (20% of
the profile) had similar chromatographic properties to alpha-
cypermethrin. Muscle, fat and milk contained mainly a single
component (muscle 85%, fat 91% and milk 97% of the exact profile),
which in each case had similar chromatographic properties to
alpha-cypermethrin. Urinary metabolites were analysed using HPLC.
The two major components (44% and 20%) had identical chromatographic
properties to N-(3-phenoxybenzoyl)glutamic acid and
N-(3-phenoxybenzoyl)glycine, respectively. A minor component
(3%) had identical chromatographic properties to 3-phenoxybenzoic
acid (Dunsire & Gifford, 1993; Morrison & Richardson, 1994).
2.1.2 Effects on liver enzymes
Six Wistar rats/sex were randomly selected from each of the
control and 1000 mg cypermethrin/kg food group at termination of a
2-year feeding study. The activity of hepatic p-nitroanisole
o-demethylase (PNOD) was determined in each of these rats. In male
rats treated with cypermethrin, PNOD activity, expressed as per gram
of liver, was significantly increased (38%). The total PNOD activity
of liver was also increased (30%), but, owing to great inter-animal
variation, statistical significance was not reached. Treated females
exhibited increases in PNOD activity when expressed per gram of liver
(21%) and per whole liver (39%), but only the latter was significant
(Potter & McAusland, 1980).
2.2 Toxicological data
2.2.1 Acute toxicity
Acute toxicity studies of cypermethrin and alpha-cypermethrin are
summarized in Tables 1 and 2.
Cypermethrin-induced signs of toxicity were typical of
cyano-containing pyrethroid intoxication. After oral administration
signs of intoxication included sedation, ataxia, splayed gait, tip-toe
walking, with occasional tremors and convulsions. These signs of
toxicity appeared within a few hours following dosing, and survivors
recovered within 3 days. The acute toxicity after dermal
administration is of a low order (Coombs et al., 1976).
alpha-Cypermethrin-induced clinical signs, like those of
cypermethrin, are typical of cyano-containing pyrethroid. The observed
signs included ataxia, abasia, gait abnormalities, choreoathetosis,
tip-toe walking, and increased salivation, lacrimation, piloerection,
tremor and clonic convulsions. Surviving animals recovered within 7
days (Rose, 1982b, 1983).
2.2.2 Short-term toxicity studies
2.2.2.1 Mice
Groups of eight mice (CD-1)/sex received diets containing 0, 200,
400, 800, 1200 or 1600 mg alpha-cypermethrin/kg feed for 29 days. One
male fed 1600 mg/kg and one female fed 1200 mg/kg were killed in a
moribund condition. These animals showed neurological disturbances.
Mice at 1200 and 1600 mg/kg developed ungroomed coats, ataxia/abnormal
gait, over-activity or hunched posture. At 800 mg/kg some animals
had ungroomed coats and abnormal gait. Body weight (gain) was
dose-relatedly decreased in mice at 1200 and 1600 mg/kg and in females
at 800 mg/kg. A similar, but less marked effect was apparent in males
receiving 800 mg/kg and females receiving 400 mg/kg. Food consumption
was lowered in rats at 1200 and 1600 mg/kg during the first 2 weeks. A
depression in lymphocyte numbers was noted in males receiving 1600
mg/kg. ALAT and ASAT levels were increased in males at 1600 mg/kg.
Urea levels were increased in all dosed males and females, without
clear dose relation. Plasma albumin and A/G ratio were decreased in
males at 1600 mg/kg. Relative kidney weight was increased in males at
800, 1200 and 1600 mg/kg. Relative lung weight was increased in males
at 1600 mg/kg. No macroscopic or microscopic effects were seen. The
NOEL was 400 mg/kg feed, equal to 57 mg/kg bw per day (Green, 1993).
Groups of 12 mice (CD-1)/sex received diets containing 0, 50, 250
or 1000 mg alpha-cypermethrin/kg feed for 13 weeks. Four males at
1000 mg/kg died during week 12 probably due to stress of treatment and
refusal of food and water. The relative kidney weights of these
animals appeared to be slightly higher. One mouse at 250 mg/kg died.
Clinical signs included thin build, ungroomed coat, hair loss and
encrustations of the dorsal body surface in animals receiving
1000 mg/kg, ungroomed haircoat in mice at 250 mg/kg, hair loss and
encrustations in males at 250 mg/kg and hair loss in two males at
50 mg/kg. Body weight gain was markedly lower in mice at 1000 mg/kg
and lower in mice at 250 mg/kg. Food consumption was slightly higher
in mice at 1000 mg/kg. Overall food conversion efficiency of animals
receiving 1000 mg/kg (and to a lesser extent 250 mg/kg) was lower than
that of controls. In males at 1000 mg/kg, PVC, Hb, RBC, total WBC and
leucocyte counts were decreased. ASAT was increased in a dose-
dependent manner in males at 250 and 1000 mg/kg, and glucose was
decreased in males at 1000 mg/kg. Serum AP was increased in females at
1000 mg/kg. Urinary specific gravity was increased in mice at the
highest dose level. In males at 1000 mg/kg relative brain, adrenal,
heart, kidney, liver, spleen, lung and testes weight were increased.
In females relative liver weight was increased at 250 and 1000 mg/kg
and relative brain and spleen weight at 1000 mg/kg. Two males and
eleven females treated at 1000 mg/kg were considered to be emaciated
at necropsy. No histological changes were observed. Owing to the hair
loss seen at 50 mg/kg, a NOEL was not identified (Amyes et al.,
1994).
2.2.2.2 Rats
a) Cypermethrin
In a 5-week feeding study, groups of six rats/sex (Charles River)
received diets containing 0, 25, 100, 250, 750 or 1500 mg cyper-
methrin/kg feed. No effects were found in haematological, macroscopic
or microscopic examinations. In the highest dose group, rats developed
piloerection, nervousness and incoordinated movement from week
2 onwards. Body weight gain, food intake and terminal body weight
were all reduced for both sexes at 1500 mg/kg. Relative liver
weight and blood urea levels were increased at the highest dose level
(only a summary available) (Coombs et al., 1976).
Table 1. Acute toxicity of cypermethrln in animals
Species1 Route Purity2 LD50 Remark Reference
mg/kg bw
Mouse oral 98.1% 88 5% in corn oil Rose, 1982a
Mouse oral ? 82 5% in corn oil Coombs et al., 1976
Mouse oral 98.1% 1126 40% in DMSO Rose, 1982a
Mouse oral 98.1% 657 50% aq. susp. Rose, 1982a
Mouse percutan. 98.1% > 100 5% in corn oil Rose, 1982a
Rat oral ? 251 5% in corn oil Coombs et al., 1976
Rat oral 98.1% 4000 40% in DMSO Rose, 1982a
Rat oral 98.1% 3423 50% aq. susp. Rose, 1982a
Rat oral ? > 2000 techn. conc. Price, 1984
Rat percutan. ? > 2000 techn. conc. Price, 1984
Rat percutan. ? > 1600 40% in xylene Coombs et al., 1976
Rat i.p. ? approx. 40% in corn oil Coombs et al., 1976
2600
Hamster oral ? 203 5% in corn oil Coombs et al., 1976
Guinea-pig oral ? approx. 20% in corn oil Coombs et al., 1976
500
1 Both male and female animals were used in all of the studies
2 cis:trans isomer ratio 51:49; ? - unknown
Table 2. Acute toxicity of alpha-cypermethrin in animals
Species1 Route Purity LD50 Remark Reference
(%) mg/kg bw
or mg/m3
Mouse oral 96.6 35 5% in corn oil Rose, 1982b
Mouse oral 96.6 762 40% in DMSO Rose, 1982b
Mouse oral 96.6 798 50% aq. suspension Rose, 1982b
Mouse percut. 96.6 > 100 5% in corn oil Rose, 1982b
Rat oral techn. 64 50% aq. CMC Gardner, 1993
Rat oral 96.6 4000 40% in DMSO Rose, 1982b
Rat oral 96.6 > 5000 50% aq. suspension Rose, 1982b
Rat dermal techn. > 2000 undiluted Gardner, 1993
Rat inhal 95.6 1590 as aerosol Jackson, 1993
(MMAD 9 µm)
1 Both male and female animals were used in all of the studies
Groups of 12 rats/sex (Wistar) were fed diets containing 0 (24
rats/sex), 25, 100, 400 or 1600 mg cypermethrin/kg feed for 91-95
days. There were no deaths, no clinical signs and no macroscopic or
microscopic changes. In the highest dose group a reduced body weight
gain was observed. During the first week reduced food consumption was
observed in both sexes at 1600 mg/kg. This could be attributed to the
palatability of the diet. During week 13, females at 1600 mg/kg had
reduced food intake. Males at 1600 mg/kg showed decreases in Hb, MCV
and eosinophil counts, and an increase in prothrombin time. In males
at 400 mg/kg there was also a decrease in eosinophil numbers. The mean
plasma urea concentration in the 1600 mg/kg groups was increased.
Relative liver weight was increased in males and females at 1600 mg/kg
and in males at 400 mg/kg. Relative kidney weight was increased in
males at 1600 mg/kg. The NOEL was 100 mg/kg feed, equivalent to
5 mg/kg bw per day (Pickering, 1981).
b) alpha-Cypermethrin
Groups of 10 Wistar rats/sex received diets containing 0 (20
rats/sex), 20, 100, 200, 400 or 800 mg alpha-cypermethrin/kg feed for
5 weeks. Observations included mortality, clinical signs, body weight
and food consumption, haematology, clinical chemistry, urinalysis,
organ weight, and macroscopic and microscopic lesions.
One male at 800 mg/kg died unrelated to treatment and two males
at 800 mg/kg were killed, owing to signs of severe neurological
disturbance. Both sexes at 800 mg/kg produced abnormal gait and
increased sensitivity to noise. Abnormal gait was seen in one male at
400 mg/kg. The mean body weights and food intake of both males and
females fed 400 or 800 mg/kg were significantly lower than those of
controls. In both males and females at 800 mg/kg and in males at
400 mg/kg prothrombin time was increased. In males at 800 mg/kg the
kaolin-cephalin coagulation time was increased and the percentage of
polymorphic neutrophils was increased at 800 mg/kg. In females
platelet count, total leucocyte count and absolute values of
polymorphic neutrophils and lymphocytes were increased, but the Hb and
haematocrit values were decreased. The MCV was decreased at 200, 400
and 800 mg/kg but not in a dose-related manner. Relative brain, liver
and kidney weights were increased in males fed 800 mg/kg and relative
brain and liver weights were increased in males fed 400 mg/kg. In
females at 800 mg/kg relative brain, liver and kidney weights were
increased. One male at 800 mg/kg that was removed from the study
showed scanty axonal lesions of the sciatic nerves. The NOEL was
200 mg/kg feed, equivalent to 10 mg/kg bw per day (Thorpe, 1982).
In a range finding study groups of five male and five female rats
were fed diets containing a-cypermethrin at concentrations of 0, 50,
200, 800 or 1200 mg/kg feed for up to 6 weeks. No effects were found
on food consumption, haematology and clinical chemistry and
microscopy. All rats at 1200 mg/kg and all males at 800 mg/kg were
killed during weeks 2 to 4 because of severe clinical signs. These
signs included high stepping, splayed gait, abasia and hyper-
sensitivity. Cachexia was seen in extreme cases. There were no
significant changes in fore or hind limb grip strength or hind limb
landing foot splay. Female rats fed 800 mg/kg had a lower mean body
weight and food intake compared to controls. These females also had a
lower leucocyte count. Microscopic examination revealed lymphocytolysis
and iymphocyte depletion of the cortical region of the thymus in males
fed 800 mg/kg and males and females at 1200 mg/kg (Fokkema, 1994a).
Groups of 30 male and 30 female Wistar rats received diets
containing 0 (60 rats/sex), 20, 60, 180 or 540 mg alpha-
cypermethrin/kg feed for 90 days. After 6 weeks 10 rats/sex
(controls 20/sex) were sacrificed for interim examinations. Three
males at 540 mg/kg developed an abnormal gait consisting of splayed
hind limbs. Skin sores and fur loss were observed in all males with
the highest incidence at 540 mg/kg and in females at 0 and 540 mg/kg.
Two males, one from controls and one at 540 mg/kg were removed because
of severe skin lesions. Reduced body weight and reduced food
consumption were seen in rats at 540 mg/kg. During the second part of
the study, body weight gain was also reduced in males at 60 and
180 mg/kg, but not in a clearly dose-related manner. Food consumption
was lower at 60 mg/kg than at 180 mg/kg. At termination the Hb value
was decreased in males and females at 540 mg/kg. MCV and MCHC were
decreased in females at 540 mg/kg. Platelet counts were increased in
males and females at 540 mg/kg. The number of lymphocytes was
increased and the number of eosinophils was decreased in males at
540 mg/kg. Urea concentration was increased in females at 540 mg/kg.
AP was decreased in females at 180 and 540 mg/kg. Urinary volume was
decreased in females at 540 mg/kg and specific gravity was increased
in males and females at 540 mg/kg. In females fed 540 mg/kg, relative
spleen, heart and brain weights were increased. In both sexes at
540 mg/kg relative kidney weight was increased and relative liver
weight was increased in both sexes at 180 and 540 mg/kg. Relative
testis weight was increased in males at 540 mg/kg. Histopathological
investigations showed a solitary form of axonal degeneration,
affecting the fibre only, in the sciatic nerve, in two males fed
540 mg/kg, but there were no clinical signs of toxicity. The NOEL was
60 mg/kg feed, equivalent to 3 mg/kg bw per day (Clark, 1982).
2.2.2.3 Rabbits
Occluded dermal applications of 0, 2, 20 or 200 mg cyper-
methrin/kg bw in PEG 300 were made to abraded and intact skin of
groups of 10 male and 10 female NZW rabbits/sex. The applications were
made for 6 h/day, 5 days/week for 3 weeks. No effects were observed on
haematology, clinical chemistry or following macroscopic or micro-
scopic examination. Slight to severe skin irritation was observed
in rabbits at 200 mg/kg bw and slight to moderate skin irritation was
observed in rabbits receiving 2 and 20 mg/kg bw. Decreased food
consumption and body weight gain was observed in rabbits treated with
200 mg/kg bw. Absolute and relative gonad weights were reduced in male
rabbits at 200 mg/kg bw. The NOEL was 20 mg/kg bw (Henderson &
Parkinson, 1981).
2.2.2.4 Dogs
a) Cypermethrin
In a 5-week feeding study, groups of three beagle dogs/sex
received diets containing 0, 15, 150 or 1500 mg cypermethrin/kg feed.
No effects were found on body weight, food consumption, ophthalmo-
scopy, haematology, clinical chemistry, macroscopy, liver weight or
following microscopic examination. In the highest dose group dogs
developed signs of intoxication, including apprehension, diarrhoea
and vomiting, licking and chewing of the paws, whole body tremors, a
stiff exaggerated hind leg gait, ataxia, inappetence, and decreased
body weight gain. Relative thyroid weight and blood urea levels were
increased and blood glucose levels were decreased (only a summary
available) (Coombs et al., 1976).
Groups of beagle dogs (four/sex) were fed cypermethrin (purity
98%) in the diet at concentrations of 0, 5, 50, 500 or 1500 mg/kg feed
for 13 weeks. No mortality occurred. Dogs at 1500 mg/kg developed
diarrhoea, anorexia and tremors as well as ataxia, incoordination and
hyperaesthesia. Also a reduced food intake and body weight gain was
seen. Due to the clinical signs two dogs/sex at 1500 mg/kg had to be
killed. Minor variations in haematology were observed; the kaolin-
cephalin clotting time was consistently lower throughout the study in
female dogs at 500 mg/kg. However, because of the lack of a dose-
response relationship, the variability of this parameter throughout
the study, and the fact that there were no changes in the prothrombin
time, this finding is not considered to be relevant. No effects were
seen on clinical chemistry parameters or on organ weights. Microscopic
examination of tissues and organs revealed a non-specific focal
bronchopneumonia in the lungs of the dogs at 1500 mg/kg. A pink colour
of the optic disc was noted in ophthalmological examinations. The NOEL
was 500 mg/kg feed, equivalent to 12.5 mg/kg bw per day (Buckwell &
Butterworth, 1977).
A further experiment was undertaken to determine the cause of the
pink discoloration, which occurred in all groups. Cypermethrin was fed
to two groups of three male dogs at concentrations of 0 or 500 mg/kg
food for 13 weeks. Specific ophthalmological examinations were made to
evaluate the degree of coloration of the optic disc. At the end of 13
weeks, there were no consistent differences between the colour of the
optic discs of the treated dogs and the controls (Buckwell &
Butterworth, 1977).
b) alpha-Cypermethrin
This study was conducted in two parts.
In the first part one male and one female beagle dog were fed
alpha-cypermethrin in the diet at concentrations of 200 mg/kg feed for
7 days, 400 mg/kg feed for 2 days and 300 mg/kg feed for 7 days. Due
to severe intoxication of the animals receiving 400 mg/kg, dosing was
discontinued after 2 days. The animals were fed the control diet and
in week 3 treatment was commenced at 300 mg/kg. After the third week
the dogs were killed.
Clinical signs including ataxia, body tremors, subdued behaviour,
head nodding, food regurgitation, diminished response to stimuli and
inflammation of gums and tongue were obtained when dogs were dosed
with 300 and 400 mg/kg. Body weight loss was observed at 300 mg/kg.
Examination of the cellular composition of blood and the chemical
components of plasma showed no abnormalities, and no macroscopic
changes were observed.
In the second part one male and one female beagle dog were fed
alpha-cypermethrin in the diet at 300 mg/kg food for 3 days (male dog)
or 4 days (female dog), and then 250 mg/kg food for 7 days. At
300 mg/kg the same effects were obtained as above. At 250 mg/kg only
the female dog developed the clinical signs shown by the dogs treated
with 300 and 400 mg/kg (Greenough & Goburdhun, 1984).
Groups of four beagle dogs/sex received diets containing dose
levels of 0, 30, 90 or 270 (six dogs/sex) mg alpha-cypermethrin/kg
feed for 13 weeks. No effects on mortality, body weight, food
consumption, ophthalmoscopy, haematology, clinical chemistry,
macroscopy, organ weights or microscopy were seen. One female at
270 mg/kg was killed, because of severe head and body tremors, ataxia,
poor limb coordination, inflamed gums and elevated temperature. All
dogs at 270 mg/kg developed marked clinical signs, including body
tremors, head nodding, lip licking, subduedness, ataxia, agitation and
high stepping gait. The NOEL was 90 mg/kg, equivalent to 2.25 mg/kg bw
per day (Greenough et al., 1984).
Four groups of beagle dogs (four/sex) received via the diet 0,
60, 120 or 240 mg alpha-cypermethrin/kg feed dally for 52 weeks. No
effects were observed on body weight, food consumption, ophthalmo-
scopy, haematology, clinical chemistry, urinalysis or organ weights,
and no changes were observed in macroscopic or microscopic examinations.
Two males at 240 mg/kg developed skin reddening on the tail. Abdominal
skin reddening and alopecia were seen in another male at 240 mg/kg and
one female at 120 mg/kg. The tail reddening caused obvious irritation
and resulted in ulceration and necrosis in one male leading to
amputation of part of the tail. The NOEL was 60 mg/kg feed, equivalent
to 1.5 mg/kg bw per day (Dean & Jackson, 1995).
2.2.3 Long-term toxicity/carcinogenicity studies
2.2.3.1 Mice
Groups of mice (70/sex, SPF-Swiss-derived) received diets
containing 0 (2 groups), 100, 400 or 1600 mg cypermethrin/kg feed for
up to 101 weeks. Ten mice/sex were killed after 52 weeks for interim
necropsy. Observations included mortality, clinical signs, body weight
gain, food consumption, haematology, clinical chemistry, organ
weights, and macroscopy and microscopy. Body weight gain of both males
and females at 1600 mg/kg was reduced when compared to the combined
control groups. Several haematological changes, consistent with mild
anaemia, were found in the 1600 mg/kg group at the interim kill, but
not at termination. At interim kill and at termination, an increase in
thrombocytosis and absolute and relative liver weight was seen in
males at 1600 mg/kg. An increase in the incidence of benign
alveologenic tumours was observed in females at 1600 mg/kg, but was
within historical control incidence. In this study the NOEL was
400 mg/kg feed, equal to 57 mg/kg bw per day (Lindsay et al., 1982).
2.2.3.2 Rats
Groups of 48 Wistar rats/sex received diets containing 1, 10, 100
or 1000 mg cypermethrin/kg feed for 2 years. The control group
consisted of 96 rats/sex and was fed untreated diet for 2 years. After
6 and 12 months six rats/sex were sacrificed and after 18 months 12
rats/sex were sacrificed (controls, respectively, 12 and 24 rats/sex).
The only effects observed were reductions in body weight and food
consumption in males and females at 1000 mg/kg. No dose-related
effects were observed on mortality, clinical chemistry, haematology,
clinical chemistry, organ weights, macroscopy or microscopy. No
difference between control and treated groups were found in the
sciatic nerves. There was no increase in compound-related tumours. The
NOEL was 100 mg/kg feed, equivalent to 5 mg/kg bw per day (McAusland
et al., 1978).
2.2.3.3 Dogs
Groups of four beagle dogs/sex (5´-7 months of age) were fed
diets containing 0, 3, 30, 300 or 1000 mg cypermethrin/kg food for 2
years. Groups of four dogs/sex were allocated to a satellite study and
received a diet containing 0, 300 or 1000 mg/kg cypermethrin (data
concerning this satellite group have only been included in the report
to aid interpretation of the results of the main study). Due to severe
signs of intoxication observed at the 1000 mg/kg level, the
concentration was reduced to 750 mg/kg at week 4 of the study and,
when signs of intoxication persisted during weeks 6-8, animals in the
high-dose group were fed a control diet for 10 days to allow them to
recover. Following the 10 days of control diet, the dogs were fed
cypermethrin at a concentration of 600 mg/kg food for the remainder of
the study. No effects were observed on ophthalmoscopy, clinical
chemistry, organ weights, macroscopy or microscopy. No abnormalities
were found in the sciatic nerves, brain or spinal cord. One male dog
in the satellite group fed 1000 mg/kg convulsed and died. Signs of
intoxication appeared within 24 hours of the initiation of dosing and
consisted of licking and chewing of the paws, a stiff high stepping
gait, whole body tremors, head shaking, incoordination, ataxia and, in
some cases, convulsions. These signs were observed at 1000 mg/kg and
to a lesser extent at 750 mg/kg, but not at 600 mg/kg. The body
weights of male dogs in the highest dose group were significantly
lower than the controls, probably due to initial reduction in food
consumption observed at 1000 mg/kg. In the satellite group no effect
on body weight gain was seen. Apart from some occasionally decreased
sodium levels in the males given the highest dose, no consistent
haematological changes were seen. The NOEL was 300 mg/kg feed,
equivalent to 7.5 mg/kg bw per day (Buckwell, 1981).
2.2.4 Reproductive toxicity studies
Groups of 30 rats/sex (Wistar) received diets containing 0, 10,
100 and 500 mg cypermethrin/kg feed for 5 weeks prior to mating and
then throughout pregnancy and lactation for three successive
generations. Two litters were bred per generation. The first litters
were discarded at weaning. Males and females from the second litter
were randomly selected to breed the next generation. A significant
reduction in body weight gain was seen in the male and female parent
rats receiving 500 mg/kg in all three generations. This was correlated
with a reduction in food consumption. Litter size was reduced at
500 mg/kg in the F1a litter at birth and after 7 and 21 days. Litter
weights were reduced at 500 mg/kg in the F1a, litters on days 7, 14
and 21 of lactation. No other effects on fertility or reproduction
parameters were found. The NOEL for maternal and reproduction toxicity
was 100 mg/kg feed, equivalent to 5 mg/kg bw per day (Hend et al.,
1978; Fish, 1979; Thorpe, 1985).
2.2.5 Special studies on embryotoxicity and teratogenicity
2.2.5.1 Rats
a) Cypermethrin
Groups of 25 pregnant female rats (Sprague-Dawley) received by
gavage 0, 17.5, 35 or 70 mg cypermethrin/kg bw per day in corn oil
during days 6 to 15 of gestation. The females were sacrificed on day
21 of gestation for examination of their uterine contents. One female
at 70 mg/kg bw per day was found dead and one female at 70 mg/kg bw
per day was killed for ethical reasons following severe convulsions.
Eleven out of 25 females at the 70 mg/kg bw per day group showed
neurological disturbances (ataxia, convulsions, hypersensitivity to
noise). A dose-related reduction in body weight gain was observed in
the groups given 35 and 70 mg/kg bw per day. There were no indications
of any embryotoxic or teratogenic effects. The NOEL for maternal
toxicity was 17.5 mg/kg bw per day and the NOEL for embryotoxicity was
70 mg/kg bw per day (Tesh et al., 1978).
b) alpha-Cypermethrin
In a range-finding study, five pregnant female Sprague-Dawley
rats received daily, by gavage, 0, 3, 9, 15 or 18 mg alpha-cyper-
methrin/kg bw in corn oil during days 6-15 of gestation. Maternal
body weights, food consumption and clinical observations were
recorded. On day 20 of gestation the females were killed and the
fetuses were weighed, sexed and externally examined. Four dams at
18 mg/kg bw per day and one dam at 15 mg/kg bw per day showed hindlimb
splay and unsteady gait during dosing. Mean body weight gain was
reduced in a dose-related manner at 9, 15 and 18 mg/kg bw per day and
at 15 and 18 mg/kg bw per day food consumption was reduced. No other
treatment-related abnormalities were observed (Irvine & Twomey, 1994).
In another study, groups of 24 pregnant female Sprague-Dawley
rats received by gavage 0, 3, 9 or 18 mg alpha-cypermethrin (purity
95.6%)/kg bw per day in corn oil during days 6-15 of gestation.
Following marked clinical signs of toxicity the dose level of 18 mg/kg
bw per day was lowered to 15 mg/kg bw per day on day 10 of gestation.
Clinical signs, body weights and food consumption were recorded. On
day 20 of gestation the females were killed and necropsied. The
fetuses were weighed, sexed and examined for external, visceral and
skeletal abnormalities.
Females at 18 mg/kg bw per day showed unsteady gait,
piloerection, limb splay and hypersensitivity to sound. After
reduction of the dose level the signs were similar but less marked.
After treatment with 18/15 mg/kg bw per day a lowered body weight gain
and food consumption was seen. At 9 mg/kg bw per day a slight body
weight reduction was seen. Mean fetal weights were slightly reduced at
18/15 mg/kg bw per day. There was no indication for teratogenicity.
The NOEL for maternal and fetal toxicity was 9 mg/kg bw per day
(Irvine, 1994c).
2.2.5.2 Rabbits
a) Cypermethrin
In a range-finding study, groups of four female pregnant rabbits
(NZW) received during days 6 to 18 of gestation by gavage 0, 25, 50,
100 or 120 mg cypermethrin/kg bw per day in corn oil. The dams were
sacrificed on day 29 of gestation. No adverse effects were seen in the
mothers and fetuses (Tesh et al., 1984a).
Groups of 16 pregnant NZW rabbits received by gavage 0, 20, 50 or
120 mg cypermethrin/kg bw per day in corn oil during days 6 to 18 of
gestation. The dams were killed on day 29 of gestation. One control
female, three females receiving 20 mg/kg bw per day and two females in
each of the groups receiving 50 and 120 mg/kg bw per day were killed
for ethical reasons. Necropsy revealed evidence of respiratory tract
infection and/or gastrointestinal tract infection not related to the
substance. Two females at 20 mg/kg bw per day and two females at
120 mg/kg bw per day aborted during the post-treatment phase of the
investigation. The number of implantations, live young and
resorptions, pre- and post-implantation losses, and fetal and
placental weights were unaffected by treatment. There was no
indication for embryotoxicity or teratogenicity. The NOEL for
embryo-toxicity was 120 mg/kg bw per day (Tesh et al., 1984b, 1988).
Groups of pregnant rabbits (20 rabbits/group, 30 rabbits were
used as an additional control group) were administered cypermethrin
dissolved in corn oil at dose levels of 0, 3, 10 or 30 mg/kg bw per
day orally from days 6 to 18 of gestation. On day 28 of gestation the
rabbits were killed and examination was made of live fetuses, dead
fetuses, resorption sites and corpora lutea. Live fetuses were
maintained for 24 hours to assess viability. Fetuses were also
examined for gross somatic and skeletal deformities. There was no
significant mortality or difference in weight gain during the period
of gestation. There were no significant differences between control
and test groups with respect to pregnancy, fetal death and survival.
Although a wide range of skeletal and visceral abnormalities was found
in the course of the study, there were no differences between control
and test groups with respect to abnormalities. It was concluded that
oral dosing up to 30 mg/kg bw during the major period of organogenesis
resulted in no teratogenic effects in offspring (FAO, 1980).
b) alpha-Cypermethrin
In a range finding study groups of five mated female NZW rabbits
received by gavage 0, 5, 15, 25 or 30 mg a-cypermethrin/kg bw per day
as solutions in corn oil, during days 7-19 of gestation. On day 28 of
pregnancy the females were killed and a necropsy was performed. The
fetuses were weighed, sexed and externally examined. One female each
at 15 and 25 mg/kg bw per day was killed prematurely. At 25 and
30 mg/kg bw per day marked reductions in body weight and food
consumption were seen. There was no indication for either
embryotoxicity or teratogenicity (Irvine, 1994a).
In another study, groups of 16 pregnant NZW rabbits received by
gavage 0, 3, 15 or 30 mg alpha-cypermethrin/kg bw per day in corn oil
during days 7-19 of gestation. Maternal clinical signs, body weight
and food consumption were recorded. The females were killed on day 28
of pregnancy. The uterus was weighed and the numbers of corpora lutea,
implantations and live fetuses were counted. The fetuses were weighed,
sexed and examined for external, visceral and skeletal abnormalities.
Two control females, three at 15 mg/kg bw per day and two at
30 mg/kg bw per day were killed, because of severe weight loss and low
food consumption. One female at 15 mg/kg bw aborted on day 28. In all
groups, including controls, there was a similar mean body weight loss
after the onset of dosing, which continued until day 11. At 30 mg/kg
bw per day there was a further reduction in mean body weight gain
towards the end of the dosing period. Food consumption reflected the
changes in mean body weight gain. The NOEL for maternal toxicity was
3 mg/kg bw per day and the NOEL for embryotoxicity was 30 mg/kg bw.
There was no indication for teratogenicity (Irvine, 1994b).
2.2.6 Special studies on genotoxicity
Results of genotoxicity studies carried out cypermethrin and
alpha-cypermethrin are summarized in Tables 3 and 4.
2.2.7 Special studies on neurotoxicity
2.2.7.1 Rats
a) Cypermethrin
Groups of 6 or 12 rats/sex were administered single oral doses of
100, 200 or 400 mg cypermethrin/kg bw (purity 97%) as a 5% dispersion
in corn oil. The observation period was 9 days. The rats were then
killed and examined histologically. All rats showed signs of
intoxication. At 400 mg/kg bw, within 4 hours of dosing, rats
developed signs of intoxication, including coarse tremors, spasmodic
movements of the body and tail and bleeding from the nose. Tip-toe
walking was also seen in some rats. All animals, except one, were
killed, owing to the severity of the signs. Histological examination
revealed swelling of the myelin sheaths and breaks of some of the
axons of the sciatic nerves. At 200 mg/kg bw similar effects were
observed; eight rats of each sex died or were killed within 48 h of
dosing. The remaining four rats survived the observation period. At
100 mg/kg bw all animals survived the 9 days. One female out of 12
showed minimal lesions in the sciatic nerve in this group. A NOEL
could not be determined (Carter & Butterworth, 1976).
In a neuromuscular dysfunction test, Wistar rats (10/sex) were
treated by gavage with 0, 25, 50, 100, 150 or 200 mg cypermethrin/kg
bw per day in DMSO for 7 consecutive days. The rats were killed 3-4
weeks after the start of dosing, and right and left sciatic/posterior
tibial nerves were analysed. Neuromuscular function was assessed by
means of the inclined plane test and peripheral nerve damage by
reference to ß-glucuronidase and ß-galactosidase activity increases in
nerve tissue homogenates.
Table 3. Results of mutagenicity assays on cypermethrin
Test system Test object Concentration Purity Results References
(%)
In vitro
Gene mutations S. typhimurium 0.2-2000 93.5 negative2 Brooks,
assay TA1525, TA100, µg/plate1 1980
TA1538, TA98, Dean,
TA1537 1981
Gene mutations E.coli WP2 or 0.2-2000 93.5 negative2 Dean,
assay WP2 uvrA µ/plate1 1981
Mitotic gene Saccharomyces 0.01-5.0 93.5 negative Dean,
conversions cerevisiae JD1 mg/litre1 1981
assay
Host-mediated S. cerevisiae 25 and 50 93.5 negative Brooks,
assay JD1 mg/kg bw 1980
in mice
Cell BHK 21/C113 31.25-250 93.5 negative Dean,
transformation cells µg/ml1 1981
assay
Chromosomal RL4 liver cells 7.5-30 µg/ml 93.5 negative Dean,
aberrations 1981
assay
Table 3. Results of mutagenicity assays on cypermethrin (cont'd).
Test system Test object Concentration Purity Results References
(%)
In vivo
Chromosome Chinese hamster 2x oral dose of ? negative Dean,
aberration 20 or 40 mg/kg 1977
assay in bone bw (2 successive
marrow days)
Dominant mouse single oral dose ? negative Dean
lethal assay of 6.25, 12.5 or et al.,
25 mg/kg bw or 1977
5 daily doses of
2.5 or 5 mg/kg
bw per day
Host-mediated mouse/ orally 0, 25, 50 ? negative JMPR,
assay S. cerevisiae mg/kg 1979
DNA damage CD rats m 100 mg/kg, f 150 ? negative Creedy &
assay mg/kg; 1, 4, 16 Wooder,
hour exposure 1977
Wistar rats m 300 mg/kg; 1, 4, negative Creedy &
16 hour exposure Wooder,
f 450 mg/kg, 1 hour 1977
exposure f 337.5
mg/kg, 4 and 16
hour exposure
1 with and without metabolic activation
2 at 200 and 2000 µg/plate, formation of visible droplets in the top agar was seen
Table 4. Results of mutagenicity assays on alpha-cypermethrin
Test system Test object Concentration Purity Results References
(%)
In vitro
Gene mutations S. typhimurium 31.5-5000 95.6 negative Brooks &
assay1 TA98, TA100, µg/ml (no toxicity) Wiggins,
TA1535, TA1537 1992
TA1538
E. coli WP2 uvrA
Gene mutations S. cerevisiae 31.25-4000 95.8 negative Brooks,
assay1 XV185-14C µg/ml 1984
Gene mutations1 L5178Y mouse 3.3-50 µg/ml 95.4 negative Vanderwaart,
assay lymphoma cells 1994
Chromosomal human peripheral -act: 93.75-1000 95.6 negative Brooks &
aberrations lymphocytes µg/ml precipitation Wiggins,
assay1 +act: 125-1000 was seen 1993
µg/ml
In vivo
Chromosomal rat femoral single oral dose 95.8 negative Clare &
aberrations bone marrow 2-8 mg/kg3 Wiggins,
1984
Table 4. Results of mutagenicity assays on alpha-cypermethrin (cont'd).
Test system Test object Concentration Purity Results References
(%)
Micronucleus mouse single oral dose 95.4 negative Vanderwaart,
assay 1-10 mg/kg bw 1995
Alkaline rat single oral dose 96.5 negative Wooder,
elution 40 mg/kg, 6 1981
analysis hours exposure
assay2
1 With and without metabolic activation
2 The effect of alpha-cypermethrin on the integrity of rat liver DNA was investigated
by this method
3 Initially doses of 10, 20 or 40 mg/kg were used, but the animals exhibited severe
signs of toxicity and the experiment was terminated at these doses. Surviving
females were evaluated for chromosomal damage and none was observed
At 200 mg/kg bw per day 50% of the males and 62.5% of the females
died, at 150 mg/kg bw per day two females and one male died and at
100 mg/kg bw per day one female died but no males. No mortalities
occurred in the other groups. A dose-related (in severity and
duration) increase was seen in clinical signs at doses > 100 mg/kg bw.
These signs included salivation, ataxia, splayed hind limb gait,
hyperexcitability to auditory stimuli, tremor and choreoathetosis. At
the same dose levels body weight gain was also reduced.
A dose-related transient functional impairment was found in rats
treated with cypermethrin in the inclined plane test. This effect was
maximal at the end of the 7-day subacute dosing regimen. At doses
which caused mortality, significant increases in ß-glucuronidase and
ß-galactosidase were found 3-4 weeks after the start of dosing in the
distal portion of the sciatic/posterior tibial nerves. There was no
direct correlation between the time course of the neuromuscular
function and the neurobiochemical changes. The NOEL was 50 mg/kg bw
per day (Rose & Dewar, 1983).
The effects of some pyrethroids, including cypermethrin, on
amplitude and pre-pulse inhibition of the acoustic startle reflex were
studied in male Wistar rats. The pyrethroids were suspended in corn
oil. Cypermethrin was given orally at dose levels of 0, 0.5, 1 or
2 mg/kg bw to 12 males. Each animal received all doses of cyper-
methrin. The intersession interval was one week. Animal behaviour
was observed by the experimenter before and after the test session for
a period of 10 minutes and the effects of the pyrethroids on overt
behaviour were measured by scoring the presence of pawing and
salivation, burrowing, hyperactivity, hyperreactivity to an external
stimulus, fine tremors of low intensity and fluid loss. Neither
cypermethrin nor the other pyrethroids tested affected the amplitude
or the latency of the startle reflex (Hijzen et al., 1988).
b) alpha-Cypermethrin
The acute neurotoxicity of alpha-cypermethrin was studied in
Crl:CD:BR rats in two separate acute studies, each using four groups
of ten animals/sex or five rats per sex (additional study). The groups
received a single dose of 0, 4, 20 or 40 mg alpha-cypermethrin/kg bw
in corn oil. During the 14-day observation period, clinical signs and
body weight were analysed. In the main study a detailed clinical
assessment for neurotoxicological effects was performed. This included
a functional observational battery (FOB) and measurements of fore and
hind limb grip strength, hindlimb landing foot splay and motor
activity. In each study five rats/sex were killed on day 15 and brain,
eyes, muscle, nerves, spinal cord and spinal ganglia were analysed.
One male rat in each of the 20 and 40 mg/kg bw groups of the
additional study was found dead on the day after dosing. Clinical
signs were seen in male rats dosed with 20 and 40 mg/kg bw. The signs
(similar in both studies) developed between 3 to 8 hours after dosing
and resolved by three days after dosing. The signs included abnormal/
splayed gait, thrashing, prostration, vocalization, piloerection,
hunched posture, unkempt appearance, soiled/stained body areas and
diarrhoea. The signs in females were similar but lower in frequency.
In addition to these signs there were also isolated cases of
twitching, tremors, abasia, hypersensitivity, pale eyes, soft faeces
and thinning of the fur.
During FOB conducted 5 hours after dosing, gait abnormalities and
clinical signs of increased reactivity were seen in most male rats
dosed with 20 and 40 mg/kg bw. In females the signs were less
frequent. In the 20 and 40 mg/kg bw groups there was a increase in
very slight to slight sporadic fibre degeneration in the sciatic
nerve. The changes were more frequent in the proximal than in the
distal part of the nerve. The NOEL was 4 mg/kg bw (Fokkema, 1994b).
c) Cypermethrin and alpha-cypermethrin
This experiment was performed in two phases. The first phase was
conducted to determine the time course for neurochemical changes in
Wistar rats occurring in the sciatic/posterior tibial nerve (SPTN),
trigeminal nerve and trigeminal ganglion following treatment with
cypermethrin for 5 days/week for 4 weeks. Cypermethrin was
administered at 150 mg/kg bw per day in DMSO (reduced to 100 mg/kg bw
per day in arachis oil, after 10 doses, because of mortality),
alpha-Cypermethrin was dosed at 37.5 mg/kg bw per day in DMSO (also
reduced after 10 days to 25 mg/kg bw per day in arachis oil). Five
animals per sex, treated with either cypermethrin and alpha-cyper-
methrin, were killed at 2, 3, 4, 5, 6, 8, 10 or 12 weeks and examined.
Dosing resulted in the death of 56% of the cypermethrin-treated
animals and 21% of the alpha-cypermethrin-treated animals. The most
frequent signs of intoxication included abnormal gait, ataxia,
lethargy, chromodacryorrhoea, salivation and hypersensitivity to
sensory stimuli. The ß-glucuronidase and ß-galactosidase activities in
the SPTN were increased at 5, 6 and 8 weeks, when compared to
controls. The increase was maximal after 5 weeks, and after 12 weeks
was comparable to controls. No significant enzyme changes were found
in the trigeminal ganglia and trigeminal nerve of treated animals.
Phase 2 was conducted to establish the dose level which did not
cause peripheral nerve degeneration in the SPTN, trigeminal nerve and
ganglia. Groups of 10 rats/sex were dosed with 37.5, 75 or 150 mg
cypermethrin/kg bw per day in DMSO or 10, 20 or 40 mg alpha-cyper-
methrin/kg bw per day in DMSO, 5 days/week for 4 weeks. A control
group of 10 animals was used.
Signs of intoxication similar to those reported in phase 1 were
seen at the highest dose levels. A large increase in ß-glucuronidase
and ß-galactosidase activities in the SPTN was seen at 150 mg/kg bw
per day cypermethrin and 40 mg/kg bw per day alpha-cypermethrin. In
the groups administered 75 mg/kg bw per day cypermethrin or 20 mg/kg
bw per day alpha-cypermethrin a small increase in ß-galactosidase was
found in both the distal and proximal sections of the SPTN. The
magnitude of the enzyme changes was similar to those of phase 1.
Significant enzyme changes were also found in the trigeminal ganglia
and to a lesser extent in the trigeminal nerve of the groups
administered 75 or 150 mg/kg bw per day cypermethrin and 20 or
40 mg/kg bw per day alpha-cypermethrin. The NOELs were 37.5 mg/kg bw
cypermethrin and 10 mg/kg bw alpha-cypermethrin (Rose, 1983).
2.2.7.2. Chickens
In a delayed neurotoxicity study, six adult domestic hens
received 1000 mg cypermethrin/kg bw per day in DMSO for 5 days. After
3 weeks the dosing regime was repeated and a further three weeks later
the birds were killed. A positive (tri- ortho-tolyl phosphate) and
negative control (not dosed) group were used.
None of the cypermethrin-treated hens developed any signs of
intoxication. Histological examination of the nervous system revealed
no lesions. All birds receiving the positive control developed
clinical signs of neurological damage within 15 days and became
progressively more unsteady and ataxic thereafter. Histological
examination of these animals showed lesions in the cerebellum, sciatic
nerve and spinal cord, including axonal and myelin degeneration
(Owen & Butterworth, 1977).
2.2.7.3 Hamsters
The 1979 JMPR (FAO, 1980) evaluated some neurotoxicity studies
with cypermethrin in hamsters. At doses of > 794 mg/kg bw, all
treated hamsters showed clinical signs of poisoning, including
tremors, abnormal irregular movements and an unusual gait. As in the
cases of rats, axon and myelin degeneration was noted in all groups
treated. The lesions included swelling and breaks in the axons and
clumping of myelin.
Hamsters treated orally with a single dose of 40 mg/kg bw,
followed by four doses of 20 mg/kg bw, developed weight loss and
sometimes mortality. There was loss of fur and dermal ulceration.
There was no effect in the mean slip angle experiment, and a marginal
increase in ß-galactosidase was observed in peripheral nerve.
Hamsters treated orally with doses of 5, 10 or 20 mg/kg bw per
day for 5 days showed no mortality. Lower body weight gain was
observed at 20 mg/kg bw per day. One female (out of 5) at the highest
dose developed hyperexcitability. There was a significant deficit in
the mean slip angle test, females showing an earlier dose-related
deficit than noted in males. ß-Galactosidase activity was increased at
all dose levels 3 weeks after the onset of the experiment. This effect
was significant at the two highest dose levels. In this experiment
dermal irritation and fur loss were noted.
In another experiment hamsters were orally treated with
30 mg/kg bw per day for 5 days. There was no mortality and there were
no differences in weight gain. There was some transient skin
irritation accompanied by skin ulceration. One male (out of 16) had an
unusual gait. There was a slight deficit in the inclined plane test
which was noted in the early parts of the experiment. Increases in
both ß-glucuronidase and ß-galactosidase were evident in peripheral
nerve tissue.
2.2.8 Special studies on biochemistry and electrophysiology
In three independent experiments with Wistar rats the effects of
varying doses of cypermethrin (purity 98%, 10% in DMSO) on the
trigeminal ganglion and three sections of the maxillary branch of the
trigeminal nerve (proximal, distal and endings) were determined.
Increases in ß-galactosidase activity in these tissues were taken as
evidence of axonal degeneration.
The three studies involved repeated oral administration of
cypermethrin at 150 mg/kg bw per day for 5 or 7 days and 0, 25, 50,
100 or 200 mg/kg bw per day for 5 or 7 days. Mortality occurred in
animals receiving 100 mg/kg bw per day or more. A dose-related
transient functional impairment, assessed by means of the inclined
plane test was found in the first week. Significant increases in
ß-glucuronidase and ß-galactosidase activity of the sciatic, tibial or
trigeminal nerves only occurred with 5 or 7 doses of 150 or
200 mg/kg bw per day. Increased activity of the enzymes in the distal
portion of nerves was found, but even in the most severely intoxicated
animals the magnitude of this increase was less than that induced by
the known neurotoxic agent methylmercury chloride (Dewar & Moffett,
1978).
Cypermethrin (1:1 cis:trans) was administered to male and female
rats at dose levels ranging from 25 to 200 mg/kg bw per day for 5
consecutive days by oral intubation as a 10% w/v solution in DMSO. A
dose-related functional deficit was observed when the mean slip angle
test and the landing foot spread test were applied to the animals. The
deficit was maximal from days 6 to 14 after the beginning of
treatment, and complete functional recovery occurred within 4 weeks.
Substantial variation in data from the landing foot spread test was
noted. Data were inconsistent over the course of the study.
ß-Glucuronidase activity was increased in a dose-dependent fashion in
both males and females. The results suggested that cypermethrin
produced a primary axonal degeneration, readily measurable 28 days
after treatment as an increase in ß-glucuronidase activity and in
deficits in specific behavioural-function testing of rats (FAO, 1980).
Electrophysiological studies were performed to determine whether
acute or subacute intoxication with cypermethrin produced changes in
the conduction velocity of slower fibres in peripheral nerves or
alterations in the maximal motor conduction velocity. There was no
evidence to suggest that cypermethrin, at doses that induced severe
clinical signs of intoxication, including ataxia, had any effect on
maximal motor conduction velocity or conduction velocity of the slower
motor fibres in peripheral nerves. Doses used in the study ranged from
a single dose of 200 mg/kg bw to 7 consecutive doses of 150 mg/kg bw
followed by two doses of 400 mg/kg bw. At near-lethal doses there were
no effects on conduction velocity even in the presence of clinical
signs of acute intoxication and at dose levels where previous studies
had shown functional degeneration. These electro-physiological
findings are reflective of motor function, which would suggest that
the physiological and functional deficits observed as a result of
acute intoxication are primarily sensory in nature (FAO, 1980).
2.2.9 Special studies on sensitization
2.2.9.1 Cypermethrin
Two out of 20 guinea-pigs developed a positive reaction to
cypermethrin in the Magnusson Kligman test, indicating that
cypermethrin is not a sensitizer (Coombs et al., 1976).
2.2.9.2 alpha-Cypermethrin
No positive reactions were obtained in a Magnusson-Kligman test
performed with guinea-pigs (Gardner, 1993).
2.2.10 Special studies on skin and eye irritation
2.2.10.1 Cypermethrin
A single application of undiluted technical cypermethrin was
moderately irritant to occluded rabbit skin (Coombs et al., 1976).
A single application of undiluted technical cypermethrin to
rabbit eyes produced a mild transient conjunctivitis and blepharism
lasting 2 days (Coombs et al., 1976).
2.2.10.2 alpha-Cypermethrin
Six NZW rabbits receiving a semi-occlusive topical application
with 500 mg alpha-cypermethrin technical developed very slight
erythema in two animals up to 72 hours after removal of the dressings.
There were no other dermal reactions (Gardner, 1993).
Six NZW rabbits receiving an instillation of 0.1 ml (equivalent
to 45 mg) alpha-cypermethrin technical developed slight conjunctival
redness and ocular discharge up to 72 hours after treatment. No cornea
or iris irritation was observed (Gardner, 1993).
2.3 Observations in humans
The symptoms and signs of acute poisoning resulting from
pyrethroids are very similar. Apart from the irritative symptoms of
the skin and respiratory tract (or digestive tract in ingestive
poisoning), acute pyrethroid poisoning is clinically characterized by
abnormalities of nervous excitability.
Occupationally exposed people had abnormal skin sensations
described as burning, itching or tingling, which could be exacerbated
by sweating or washing and readily disappeared after several hours.
Systemic symptoms included dizziness, headache, nausea, anorexia and
fatigue. Vomiting was more prominent in patients with ingestive
poisoning than in occupational poisoning cases. Other symptoms such as
chest tightness, paraesthesia, palpitation, blurred vision and
increased sweating were less frequently seen. The more serious cases
developed coarse muscular fasciculations in large muscles or
extremities (Van den Bercken & Vijverberg, 1989; He et al., 1989).
Urine obtained from operators spraying cypermethrin in
experimental trials was analysed for the presence of the chlorinated
cyclopropane carboxylic acid metabolite. This metabolite was observed
in the urine of exposed workers at levels up to 0.4 µg/ml (the limit
of detection was estimated to be 0.05 µg/ml).
Cypermethrin sprayers were found to have residues on the exposed
parts of their bodies. The rate of dermal exposure of the operators
during spraying ranged from 1.5 to 46.1 mg/hour. There was a
reasonable relationship between the total cypermethrin deposited
dermally and the excretion in urine. The levels of the cyclopropane
carboxylic acid metabolite in the 24-hour urine were between <0.05
and 0.32 mg (not specified). This, together with the finding of 0.6 mg
(not specified) of this metabolite in 72-hour urine from one man, led
to the estimation that approximately 3% of the total dermal dose was
absorbed and rapidly excreted by the operators (FAO, 1980).
In a controlled experiment with sprayers, no abnormalities were
found in clinical and neurological examinations, blood chemistry or
peripheral nerve function tests (including the trigeminal nerve). In
some electroneurophysiological tests (motor conduction velocity, slow
fibre conduction velocity and cornea reflex), a significant change
within the normal range appeared to exist for the group sprayers
between pre- and post-exposure measurements. These changes probably
reflect seasonal variations (FAO, 1982).
3. COMMENTS
The Committee considered toxicological data on cypermethrin and
alpha-cypermethrin, including the results of acute, short-term, and
reproductive studies, and studies on pharmacokinetics and metabolism,
genotoxicity, long-term toxicity/carcinogenicity and neurotoxicity.
Results of effects in humans were also considered.
Cypermethrin is a mixture of four cis and four trans isomers. The
cis isomers are more biologically active and more persistent than the
trans isomers, alpha-Cypermethrin is a mixture of the two most active
cis isomers. A typical cypermethrin sample contains 25% alpha-
cypermethrin. Cypermethrin and alpha-cypermethrin are alpha-cyano or
type II pyrethroids that cause neurotoxicity in mammals and insects.
They affect nerve membrane sodium channels, causing a long-lasting
prolongation of the normally transient increase in sodium permeability
of the membrane during excitation. At high dose levels, these type II
pyrethroids induce salivation and tremors that progress to
characteristic clonic-tonic convulsions (choreoathetosis and
salivation syndrome).
After oral administration, cypermethrin is readily absorbed,
distributed and excreted in rats, chickens, sheep and cattle.
Cypermethrin is primarily eliminated in urine and faeces in about
equal proportions. Less than 1% is excreted in milk. When cypermethrin
was applied dermally to sheep, 2.5% was eliminated in urine and faeces
within 6 days and after an oral dose about 60% was eliminated within 2
days.
Studies in cattle indicated that absorption, distribution and
excretion were comparable for cypermethrin and alpha-cypermethrin. The
major metabolic route for both cypermethrin and its isomers, including
alpha-cypermethrin, is cleavage of the ester bond followed by
hydroxylation and conjugation of the cyclopropyl and phenoxybenzyl
portions of the molecule. The data suggest that there is no isomeric
interconversion during metabolism.
The acute oral toxicity of cypermethrin and alpha-cypermethrin is
moderate to high. WHO has classified these substances as "moderately
hazardous" (WHO, 1996). In rats and mice, the oral LD50 ranges from
82 to 4000 mg/kg bw for cypermethrin and from 35 to > 5000 mg/kg bw
for alpha-cypermethrin, depending on the vehicle used. At lethal or
near lethal doses the signs are typical of type-II pyrethroids and
include salivation, ataxia, gait abnormalities and convulsions.
Several oral short-term toxicity studies with cypermethrin were
available. Cypermethrin has been tested in rats (5 weeks and 90 days)
and dogs (5 weeks and 13 weeks) at dose levels ranging from 25 to
1600 mg/kg feed (equivalent to 1.25-80 mg/kg bw per day) and 5 to
1500 mg/kg feed (equivalent to 0.125-37.5 mg/kg bw per day),
respectively. In these studies, the clinical signs included ataxia,
abnormal gait, nervousness, and, particularly in dogs, inappetence,
diarrhoea, vomiting and hyperaesthesia. In both rats and dogs,
cypermethrin caused decreases in body weight gain, food intake, and a
number of haematological parameters, increases in some organ weights
and plasma urea levels, and, at lethal or near-lethal doses, effects
on the nervous system. For cypermethrin the lowest NOEL in short-term
studies was in a 90-day study with rats administered 25, 100, 400 or
1600 mg/kg feed (equivalent to 1.25-80 mg/kg bw per day). Male rats
given 1600 mg/kg feed showed decreases in haemoglobin concentration,
mean corpuscular volume and eosinophil numbers, and increases in
prothrombin time, plasma urea levels and relative liver and kidney
weights. The decrease in eosinophil numbers and increase in relative
liver weight were also observed in males at 400 mg/kg feed. In female
rats, reduced food intake and increased relative liver weight were
noted in rats given 1600 mg/kg feed. The NOEL in this study was
100 mg/kg feed, equivalent to 5 mg/kg bw per day.
alpha-Cypermethrin was tested in oral short-term toxicity studies
with mice (29 days and 13 weeks), rats (5 weeks, 6 weeks and 90 days)
and dogs (13 weeks and 52 weeks) at dose levels ranging from 50 to
1600 mg/kg feed (equivalent to 7-240 mg/kg bw per day), 20 to
1200 mg/kg feed (equivalent to 1.25-60 mg/kg bw per day) and 30 to
270 mg/kg feed (equivalent to 0.75-6.75 mg/kg bw per day),
respectively. In these studies, alpha-cypermethrin caused the same
effects as described for cypermethrin in the short-term studies. The
signs of toxicity included ataxia, abnormal gait, increased
sensitivity to noise, hyperactivity, hunched posture and, as
demonstrated histologically, axonal degeneration of the sciatic
nerves. For alpha-cypermethrin the lowest NOEL was in a 52-week study
with dogs. In this study a diet containing 60, 120 or 240 mg/kg feed
(equivalent to 1.5 to 6 mg/kg bw per day) was administered. Dogs given
120 and 240 mg/kg feed showed skin reddening, ulceration and necrosis.
The NOEL in this study was 1.5 mg/kg bw per day.
A three-generation reproductive toxicity study with cypermethrin
was performed in rats at dose levels of 10, 100 or 500 mg/kg feed
(equivalent to 0.5-25 mg/kg bw per day). At the highest dose a
reduction in body weight gain and food consumption and a concomitant
reduction in litter size and weight were seen in the F1a progeny
only. No other effects on fertility or reproduction parameters were
observed. The NOEL was 100 mg/kg feed, equivalent to 5 mg/kg bw per
day.
Cypermethrin did not cause embryotoxicity or teratogenicity in
rats at doses up to 70 mg/kg bw per day or in rabbits at doses up to
120 mg/kg bw per day. alpha-Cypermethrin did not cause embryotoxicity
or teratogenicity in rats at doses up to 9 mg/kg bw per day or in
rabbits up to 30 mg/kg bw per day. The NOELs for maternal toxicity in
rats were 17.5 and 9 mg/kg bw per day for cypermethrin and
alpha-cypermethrin, respectively, while the NOELs for maternal
toxicity in rabbits were 30 and 3 mg/kg bw per day for cypermethrin
and alpha-cypermethrin, respectively.
Cypermethrin and alpha-cypermethrin have been tested in a wide
variety of in vitro and in vivo genotoxicity studies. All of the
results were negative.
Two long-term toxicity/carcinogenicity studies with mice and rats
were available on cypermethrin. Mice received a diet containing 100,
400 or 1600 mg/kg feed (equal to 14-228 mg/kg bw per day) for 101
weeks. At 1600 mg/kg feed reduced body weight gain, changes in
haematological parameters and increased liver weight were observed. No
effects were observed at 400 mg/kg feed, equal to 57 mg/kg bw per day.
In a study in which rats received diets containing 1, 10, 100 or
1000 mg/kg feed (equivalent to 0.05-50 mg/kg bw per day) for 2 years,
the only effects observed were reductions in body weight and food
consumption at 1000 mg/kg feed. The NOEL was 100 mg/kg feed,
equivalent to 5 mg/kg bw per day.
In a two-year toxicity study, dogs received diets containing
3, 30, 300 or 1000 mg cypermethrin/kg feed (equivalent to
0.075-25 mg/kg bw per day). The dose level of 1000 mg/kg feed was
reduced to 600 mg/kg feed owing to severe intoxication. At 300 mg/kg
feed, equivalent to 7.5 mg/kg bw per day, no effects were seen. The
Committee concluded that cypermethrin was not carcinogenic in these
studies.
Long-term toxicity/carcinogenicity or reproductive toxicity
studies were not available on alpha-cypermethrin. The Committee noted
the absence of reproductive toxicity and carcinogenicity associated
with administration of cypermethrin, which contains 25% alpha-
cypermethrin. The Committee also noted the absence of genotoxicity
for either cypermethrin or alpha-cypermethrin, the absence of
carcinogenicity associated with compounds of similar structure and
the similar metabolism and disposition of the two compounds. In view
of the foregoing, the Committee concluded that it was unnecessary to
request the results of long-term toxicity/carcinogenicity or
reproductive toxicity studies on alpha-cypermethrin.
Several studies on the neurotoxicity of cypermethrin and
alpha-cypermethrin in rats were available. In these studies high oral
doses of cypermethrin and alpha-cypermethrin caused clinical signs
that included coarse tremor and spasmodic body and tail movements.
Evidence of axonal damage in the sciatic/posterior tibial nerves and
the trigeminal nerve and ganglion was indicated by significant
increases in ß-glucuronidase and ß-galactosidase in nerve tissue
homogenates, in addition to abnormal neuromuscular function tests. In
the inclined plane test, cypermethrin (in DMSO) caused transient
functional impairment. The lowest NOEL for neurotoxicity was
37.5 mg/kg bw per day for cypermethrin (in DMSO) and 4 mg/kg bw per
day for alpha-cypermethrin (in corn oil), indicating that the toxicity
may be influenced by the vehicle used.
Humans occupationally exposed to cypermethrin developed skin
sensation as a first reaction, followed by systemic effects such as
dizziness, headache, nausea, paraesthesia and increased sweating. In
more serious cases, muscular fasciculations developed in large muscles
or in the extremities. In experiments with operators spraying
cypermethrin, no clinical nervous system abnormalities were observed.
However, exposure levels were not measured.
4. EVALUATION
The Committee established an ADI of 0-50 µg/kg bw for
cypermethrin on the basis of the NOEL of 5 mg/kg bw per day in 90-day,
2-year and reproductive toxicity studies in rats and the application
of a safety factor of 100. The Committee established an ADI of
0-20 µg/kg bw for alpha-cypermethrin on the basis of the NOEL of
1.5 mg/kg bw per day in a 52-week study in dogs and the application of
a safety factor of 100.
5. REFERENCES
Amyes, S.J., Holmes, P., Irving, E.M., Green, C.F., Virgo, D.M., &
Sparrow, S. (1994). Alpha-cypermethrin: preliminary toxicity study by
dietary administration to CD-1 mice for 13 weeks. Unpublished report
No. 92/SHL009/0849 from Pharmaco-LSR Ltd, Suffolk, England. Submitted
to the WHO by Cyanamid, Wayne, NJ. USA.
Brooks, T.M. (1980). Toxicity studies with agricultural chemicals:
Mutagenicity studies with RIPCORD in microorganisms in vitro and in
the host-mediated assay (Experiment Nos 1846 and 1847). Unpublished
report TLGR.80.059 from Shell Toxicology Laboratory (Tunstall).
Submitted to the WHO by Cyanamid, Wayne, NJ, USA.
Brooks, T.M. (1984). Genotoxicity studies with Fastac: the induction
of gene mutation in the yeast Saccharomyces cerevisiae. Unpublished
report No. SBGR.84.117 from Shell Research Limited, Sittingbourne
Research Centre. Submitted to the WHO by Cyanamid, Wayne, NJ, USA.
Brooks, T.M. & Wiggins, D.E. (1992). FASTAC TM: Bacterial mutagenicity
studies. Unpublished report No. SBTR.92.022 from Shell Research
Limited, Sittingbourne Research Centre. Submitted to the WHO by
Cyanamid, Wayne, NJ, USA.
Brooks, T.M. & Wiggins, D.E. (1993). FASTAC TM: In vitro chromosome
studies using cultured human lymphocytes. Unpublished report
No. SBTR.93.007 from Shell Research Limited, Sittingbourne Research
Centre. Submitted to the WHO by Cyanamid, Wayne, NJ, USA.
Buckwell, A.C. (1981). A 2-year feeding study in dogs on WL 43467
(Experiment No. 1412). Unpublished report No. SBGR.81.126 (plus two
addenda) from Shell Toxicology Laboratory (Tunstall). Submitted to the
WHO by Cyanamid, Wayne, NJ, USA.
Buckwell, A.C. & Butterworth, S. (1977). Toxicology studies on the
pyrethroid insecticide WL 43467: a 13-week feeding study in dogs
(Experiment No. 1112). Unpublished report No. TLGR 0127.77 (including
addendum and corrigendum) from Shell Toxicology Laboratory (Tunstall).
Submitted to the WHO by Cyanamid, Wayne, NJ, USA.
Carter, B.I. & Butterworth, S.T.G. (1976). Toxicity of insecticides:
The acute oral toxicity and neuropathological effects of WL 43467 to
rats. Unpublished report No. TLGR.0055.76 from Shell Research,
Sittingbourne Research Centre. Submitted to the WHO by Cyanamid,
Wayne, NJ, USA.
Clare, M.G. & Wiggins, D.E. (1984). Genotoxicity studies with fastac:
in vivo cytogenetic test using rat bone marrow. Unpublished report
No. SBGR.84.120 from Shell Research Limited, Sittingbourne Research
Centre. Submitted to the WHO by Cyanamid, Wayne, NJ, USA.
Clark, D.G. (1982). WL85871: A 90-day feeding study in rats.
Unpublished report No. SBGR.81.293 from Shell Research Limited,
Sittingbourne Research Centre. Submitted to the WHO by Cyanamid,
Wayne, NJ, USA.
Coombs, A.D., Carter, B.I., Hend, R.W., Butterworth, S.G., & Buckwell,
A.C. (1976) Toxicity studies on the insecticide WL 43467: Summary of
results of preliminary experiments. Unpublished report
No. TLGR.0104.76 from Shell Toxicology Laboratory (Tunstall).
Submitted to the WHO by Cyanamid, Wayne, NJ, USA.
Crawford, M.J. (1978). The excretion and residues of radioactivity in
cows treated orally with 14C-labelled WL 43467. Unpublished report
No. TLGR.0029.78 from Shell Toxicology Laboratory (Tunstall).
Submitted to the WHO by Cyanamid, Wayne, NJ, USA.
Crawford, M.J. & Hutson, D.H. (1977). The elimination and retention of
WL 43467 when administered dermally or orally to sheep. Unpublished
report No. TLGR.0098.77 from Shell Toxicology Laboratory (Tunstall).
Submitted to the WHO by Cyanamid, Wayne, NJ, USA.
Crawford, M.J. & Hutson, D.H. (1978). The elimination of residues from
the fat of rats following the oral administration of [14C-benzyl]
WL 43381 (cis-WL 43467). Unpublished report No. TLGR.0078.78 from
Shell Toxicology Laboratory (Tunstall). Submitted to the WHO by
Cyanamid, Wayne, NJ, USA.
Creedy, C.L. & Wooder, M.F. (1977) Studies on the effect of WL 43467
upon the integrity of rat liver cell DNA in vivo. Unpublished report
No. TLGR.0043.77 from Shell Toxicology Laboratory (Tunstall).
Submitted to the WHO by Cyanamid, Wayne, NJ, USA.
Croucher, A., Stoydin, G., Cheeseman, M.E., Buckwell, A.C., &
Gellatly, J.B.M. (1980). The metabolic fate of cypermethrin in the
cow: elimination and residues derived from 14C-benzyl label.
Unpublished report No. TLGR.80.121 from Shell Toxicology Laboratory
(Tunstall). Submitted to the WHO by Cyanamid, Wayne, NJ, USA.
Dean, B.J. (1977). Toxicity studies with WL 43467: Chromosome studies
on bone marrow cells of Chinese hamsters after two daily oral doses of
WL 43467. Unpublished report No. TLGR.0136.77 from Shell Toxicology
Laboratory (Tunstall). Submitted to the WHO by Cyanamid,
Wayne, NJ, USA.
Dean, B.J. (1981). Toxicity studies with agricultural chemicals:
In vitro genotoxicity studies with RIPCORD (Experiment
No. AIR-1820). Unpublished report No. TLGR.80.116 from Shell
Toxicology Laboratory (Tunstall). Submitted to the WHO by Cyanamid,
Wayne, NJ, USA.
Dean, I. & Jackson, F. (1995). WL85871: 52-week oral (dietary)
toxicity study in dogs (IRI Project No. 652238). Unpublished report
No. IRI/11110 from Inveresk Research International, Scotland.
Submitted to the WHO by Cyanamid, Wayne, NJ, USA.
Dean, B.J., Van der Pauw, C.L., & Butterworth S.T.G. (1977). Toxicity
studies with WL 43467: Dominant lethal assay in male mice after single
oral doses of WL 43467. Unpublished report No. TLGR.0042.77 from Shell
Toxicology Laboratory (Tunstall). Submitted to the WHO by Cyanamid,
Wayne, NJ, USA.
Dewar, A.J. & Moffett, B.J. (1978). Toxicity studies on the
insecticide WL 43467: Biochemical studies on the effect of WL 43467 on
the rat trigeminal nerve and ganglion. Unpublished report No.
TLGR.0162.77 from Shell Toxicology Laboratory (Tunstall). Submitted to
the WHO by Cyanamid, Wayne, NJ, USA.
Dunsire, J.P. & Gifford, L.J. (1993). The distribution of
[benzyl-14C]WL85871 (FASTAC) in the lactating cow (nature of residue
study to EPA guidelines - Live phase) (IRI Project No. 153667).
Unpublished report No. IRI/9879 from Inveresk Research International,
Scotland. Submitted to the WHO by Cyanamid, Wayne, NJ, USA.
FAO (1980). Pesticide residues in food-1979. Report of the Joint
Meeting of the FAO Panel of Experts on Pesticide Residues in Food and
the Environment and the WHO Expert Group on Pesticide Residues. Rome,
Food and Agriculture Organization of the United Nations (FAO Plant
Production and Protection Paper, No. 20).
FAO (1982). Pesticide residues in food-1981. Report of the Joint
Meeting of the FAO Panel of Experts on Pesticide Residues in Food and
the Environment and the WHO Expert Group on Pesticide Residues. Rome,
Food and Agriculture Organization of the United Nations (FAO Plant
Production and Protection Paper, No. 37).
Fish, A. (1979). Corrigendum I in Group Research Report
No. TLGR.0188.78. Unpublished data from Shell Toxicology Laboratory
(Tunstall). Submitted to the WHO by Cyanamid, Wayne, NJ, USA.
Fokkema, G.N. (1994a). WL85871 (FASTAC): A 6-week range finding
feeding study in the rat. Unpublished report No. SBTR.93.002 from
Shell Research Limited, Sittingbourne Research Centre. Submitted to
the WHO by Cyanamid, Wayne, NJ, USA.
Fokkema, G.N. (1994b). WL85871 (FASTAC): An acute oral (gavage)
neurotoxicity study in the rat. Unpublished report No. SBTR.92.027
from Shell Research Limited, Sittingbourne Research Centre. Submitted
to the WHO by Cyanamid, Wayne, NJ, USA.
Gardner, J.R. (1993). FASTAC technical: Acute oral and dermal toxicity
in rat, skin and eye irritancy in rabbit and skin sensitisation
potential in guinea pig. Unpublished report No. SBTR.92.033 from Shell
Research Limited, Sittingbourne Research Centre. Submitted to the WHO
by Cyanamid, Wayne, NJ, USA.
Green, C.F. (1993). Alphacypermethrin: Preliminary toxicity study by
dietary administration to CD-1 mice for four weeks. Unpublished report
No. LSR 92/0346 from Life Science Research Limited. Submitted to the
WHO by Cyanamid, Wayne, NJ, USA.
Greenough, R.J. & Goburdhun, R. (1984). WL 85871: Oral (dietary)
maximum tolerated dose study in dogs (IRI Project No. 631087).
Unpublished report No. IRI/3107 from Inveresk Research International,
Scotland. Submitted to the WHO by Cyanamid, Wayne, NJ, USA.
Greenough, R.J., Cockrill, J.B., & Goburdhun, R. (1984).
WL 85871: 13-week oral (dietary) toxicity study in dogs (IRI Project
No. 631092). Unpublished report No. IRI/3197 from Inveresk Research
International, Scotland. Submitted to the WHO by Cyanamid,
Wayne, NJ, USA.
He, F., Wang, S., Liu, L., Chen, S., Zhang, Z. & Sun, J. (1989).
Clinical manifestations and diagnosis of acute pyrethroid poisoning.
Arch. Toxicol., 63, 54-58.
Hend, R.W., Hendy, R. & Fleming, D.J. (1978). Toxicity studies on the
insecticide WL 43367: a three generation reproduction study in rats.
Unpublished report No. TLGR.0188.78 from Shell Toxicology Laboratory
(Tunstall). Submitted to the WHO by Cyanamid, Wayne, NJ, USA.
Henderson, C. & Parkinson, G.R. (1981). Cypermethrin technical:
subacute dermal toxicity study in rabbits (CTL Study No. LB0019).
Unpublished report No. CTL/P/588 from ICI Central Toxicology
Laboratory. Submitted to the WHO by Cyanamid, Wayne, NJ, USA.
Hijzen, T.H., de Beun, R., & Slanger, J.L. (1988). Effects of
pyrethroids on the acoustic startle reflex in the rat. Toxicology,
49, 271-276.
Hutson, D.H. & Stoydin, G. (1976). The excretion of radioactivity from
cows fed with radioactively labelled WL 43467. Unpublished report
No. TLGR.0075.76 from Shell Toxicology Laboratory (Tunstall).
Submitted to the WHO by Cyanamid, Wayne, NJ, USA.
Hutson, D.H. & Stoydin, G. (1987). Excretion and residues of the
pyrethroid insecticide cypermethrin in laying hens. Pestic. Sci.,
18, 157-168.
IPCS (1996) The WHO recommended classification of pesticides by hazard
and guidelines to classification 1996-1997. World Health Organization,
Geneva, p. 23 (Document WHO/PCS/96.3).
Irvine, L.F.H (1994a). Oral (gavage) rabbit developmental toxicity
dose ranging study. Unpublished report No. SLN/3/92 from Toxicol
Laboratories Limited, Ledbury, England. Submitted to the WHO by
Cyanamid, Wayne, NJ, USA.
Irvine, L.F.H. (1994b). Oral (gavage) rabbit developmental toxicity
(teratogenicity) study. Unpublished report No. SLN/4/93 from Toxicol
Laboratories Limited, Ledbury, England. Submitted to the WHO by
Cyanamid, Wayne, NJ, USA.
Irvine, L.H.F. (1994c). Alphacypermethrin. Oral (gavage) rat
developmental toxicity (teratogenicity) study. Unpublished report
No. SLN/2/92 from Toxicol Laboratories Limited, Ledbury, England.
Submitted to the WHO by Cyanamid, Wayne, NJ, USA.
Irvine, L.F.H & Twomey, K. (1994). Alphacypermethrin. Oral (gavage)
rat developmental study. Unpublished report No. SLN/1/92 from Toxicol
Laboratories Limited, Ledbury, England. Submitted to the WHO by
Cyanamid, Wayne, NJ, USA.
Jackson, G.C. (1993). Alphacypermethrin: Acute inhalation toxicity in
rats, 4-hour exposure. Unpublished report No. SLL 266/930770 from
Huntingdon Research Centre, England. Submitted to the WHO by Cyanamid,
Wayne, NJ, USA.
Lindsay, S., Banham, P.B., Chart, I.S., Chalmers, D.T., Godley, M.J.,
& Taylor, K. (1982). Cypermethrin: Lifetime feeding study in mice
(CTL Study No. PM0366). Unpublished report No. CTL/P/687 (including 1
supplement) from ICI Central Toxicology Laboratory. Submitted to the
WHO by Cyanamid, Wayne, NJ, USA.
McAusland, H.E., Butterworth, S.T.G., & Hunt, P.F. (1978). Toxicity
studies on the insecticide WL 43467: A two-year feeding study in rats.
Unpublished report No. TLGR.0189.78 (including 4 corrigenda/addenda)
from Shell Toxicology Laboratory (Tunstall). Submitted to the WHO by
Cyanamid, Wayne, NJ, USA.
Morrison, B.J. & Richardson, K.A. (1994). WL85871 (alphacypermethrin,
FASTAC): The metabolism of 14C-WL85871 after repeated oral dosing in
the lactating cow; in-life phase and metabolite profiling (Experiment
No. 6046). Unpublished report No. SBTR.93.063 from Sittingbourne
Research Centre, Sittingbourne, England. Submitted to the WHO by
Cyanamid, Wayne, NJ, USA.
Owen, D.E. & Butterworth, S.T.G. (1977). Toxicity of pyrethroid
insecticides: Investigation of the neurotoxic potential of WL 43467 to
adult domestic hens. Unpublished report No. TLGR.0134.77 from Shell
Toxicology Laboratory (Tunstall). Submitted to the WHO by Cyanamid,
Wayne, NJ, USA.
Pickering, R.G. (1981). A 90-day feeding study of WL 43467 in rats
(Experiment No. 1806). Unpublished report No. TLGR.80.143 from Shell
Toxicology Laboratory (Tunstall). Submitted to the WHO by Cyanamid,
Wayne, NJ, USA.
Potter, D. & McAusland, H.E. (1980). Toxicity studies on the
insecticide WL43467: A study of liver microsomal enzyme activity in
rats fed WL 43467 for 2 years (Experiment No. 1105). Unpublished
report from Shell Toxicology Laboratory (Tunstall). Submitted to the
WHO by Cyanamid, Wayne, NJ, USA.
Price, J.B. (1984). Toxicology of pyrethroids: the acute oral and
percutaneous toxicity of Ripcord (WL-43467). Technical concentrate
(Project No. SRCAIR84). Unpublished report No. SBGR.84.299 from Shell
Research Limited, Sittingbourne Research Centre. Submitted to the WHO
by Cyanamid, Wayne, NJ, USA.
Rose, G.P. (1982a). Toxicology of pyrethroids: the acute oral and
percutaneous toxicity of WL 85871 (cis-2-Ripcord) in comparison with
Ripcord. Unpublished report No. SBGR.82.130 from Shell Research
Limited, Sittingbourne Research Centre. Submitted to the WHO by
Cyanamid, Wayne, NJ, USA.
Rose, G.P. (1982b). Toxicology of pyrethroids: the acute oral and
percutaneous toxicity of WL 85871 (cis-2-RIPCORD) comparison with
RIPCORD. Unpublished report No. SBGR.82.130 from Shell Research
Limited, Sittingbourne Research Centre. Submitted to the WHO by
Cyanamid, Wayne, NJ, USA.
Rose, G.P. (1983). Neurotoxicity of WL85871. Comparison with WL43467:
The effect of twenty oral doses of WL85871 or WL43467 over a period of
4 weeks on the rat sciatic/posterior tibia nerve, trigeminal nerve and
trigeminal ganglion. Unpublished report No. SBGR.83.185 from Shell
Research Limited, Sittingbourne Research Centre. Submitted to the WHO
by Cyanamid, Wayne, NJ, USA.
Rose, G.P. & Dewar, A.J. (1983). Intoxication with four synthetic
pyrethroids fails to show any correlation between neuromuscular
dysfunction and neurobiochemical abnormalities in rats.
Arch. Toxicol., 53, 297-316.
Tesh, J.M., Tesh, S.A., & Davies, W. (1978). WL 43467: Effects upon
the progress and outcome of pregnancy in the rat. Unpublished report
of Life Science Research. Submitted to the WHO by Cyanamid,
Wayne, NJ, USA.
Tesh, J.M., Ross, F.W., & Wightman, T.J. (1984a). WL 43467: Effects of
oral administration upon pregnancy in the rabbit. 1. Dosage range
finding study. Unpublished test report No. 84/SHL003/014 from of Life
Science Research. Submitted to the WHO by Cyanamid, Wayne, NJ, USA.
Tesh, J.M., Ross, F.W., & Wightman, T.J. (1984b). WL 43467: Effects of
oral administration upon pregnancy in the rabbit. 2. Main study.
Unpublished test report No. 84/SHL004/043 from Life Science Research.
Submitted to the WHO by Cyanamid, Wayne, NJ, USA.
Tesh, J.M., Ross, F.W., & Wightman, T.J. (1988). Supplementary report
to LSR report No. 84/SHL004/043. Unpublished report No. 88/SHL004/775
from Life Science Research. Submitted to the WHO by Cyanamid,
Wayne, NJ, USA.
Thorpe, E. (1982). A 5-week feeding study with WL 85871 in rats
(Experiment No. 2095). Unpublished report No. SBGR.81.212 from Shell
Research Limited, Sittingbourne Research Centre. Submitted to the WHO
by Cyanamid, Wayne, NJ, USA.
Thorpe, E. (1985). Fourth corrigendum/addendum to TLGR.0188.78.
Unpublished report from Shell Research Limited, Sittingbourne Research
Centre (Four volumes). Submitted to the WHO by Cyanamid,
Wayne, NJ, USA.
Van der Bercken, J. & Vijverberg, H.P.M. (1989). Neurotoxicological
effects of pyrethroid insecticides. Unpublished report from the
Department of Veterinary Pharmacology, Pharmacy and Toxicology.
University of Utrecht, The Netherlands. Submitted to the WHO by
Cyanamid, Wayne, NJ, USA.
Vanderwaart, E.J. (1994). Evaluation of the mutagenic activity of
Fastac technical in a in vitro mammalian cell gene mutation test
with L5178Y mouse lymphoma cells (with independent repeat).
Unpublished report No. 087367 from Notox B.V., The Netherlands.
Submitted to the WHO by Cyanamid, Wayne, NJ, USA.
Vanderwaart, E.J. (1995). Micronucleus test in bone marrow cells of
the mouse with Fastac technical. Unpublished report No. 087378 from
Notox B.V., The Netherlands. Submitted to the WHO by Cyanamid,
Wayne, NJ, USA.
WHO (1996). The WHO recommended classification of pesticides by hazard
and guidelines to classification 1996-1997 (WHO/PCS/96.3). Available
from the International Programme on Chemical Safety, World Health
Organization, Geneva, Switzerland.
Wooder, M.F. (1981). Studies on the effect of WL 85871 on the
integrity of rat liver DNO in vivo (Experiment No. 2103).
Unpublished report No. SBGR.81.225 from Shell Toxicology Laboratory
(Tunstall). Submitted to the WHO by Cyanamid, Wayne, NJ, USA.
2. BIOLOGICAL DATA
2.1 Biochemical aspects
2.1.1 Absorption, distribution and excretion
According to the JMPR (FAO, 1980, 1982) cypermethrin is readily
absorbed and rapidly eliminated via urine and faeces of mice, rats,
dogs, sheep and cows. Absorption from the gastrointestinal tract is
more rapid with the trans isomer than with the cis isomer. The highest
mean concentrations are found in body fat, liver, kidney, muscle, skin
and milk. The clearance rate from adipose tissue is slow and the
elimination half-life in rats and mice ranges from 10 to 20 and from
20 to 30 days, respectively. The data suggest a potential for
bioaccumulation in the body following continuous exposure. With
respect to the chemical and especially the isomeric complexity of the
molecule, the metabolic profile due to all of its isomers is extremely
complex. Cypermethrin is readily cleaved at the ester linkage to
produce the cyclopropane carboxylic acid and a 3-phenoxybenzoyl moiety
that is further metabolised by oxidation at the 4'position. The
resultant phenol is almost totally conjugated with sulfate. The
4'-hydroxy sulfate forms the major aryl metabolite (16%), and
3-phenoxybenzoic acid is the second most important (5%). The other
identified aryl metabolites are 3-(4-hydroxyphenoxy)benzoic acid (1%)
and the glycine conjugate, N-(3-phenoxybenzoyl) glycine (1%).
2.1.1.1 Rats
Eight female Wistar rats were orally dosed with 2.5 mg
14C-benzyl-labelled cis-cypermethrin/kg bw. They were killed in
groups of two at 8, 14, 25 and 42 days after dosing, and the
radioactivity in fat, liver and kidney was measured. Selected fat
samples were analysed for parent compound. Between days 14 and 42,
radioactivity was eliminated from the fat with a half-life of 20-25
days. Residues in liver and kidney were 30 to 40 times lower than
those in fat, but were eliminated at a similar rate. In two pooled fat
samples taken 8 and 24 days after dosing, 90-100% of the radioactivity
was in the form of the parent compound (Crawford & Hutson, 1978).
2.1.1.2 Chickens
Six Warren laying hens were treated daily for 14 days with 10 mg
14C-phenoxy-labelled cypermethrin/kg food (equivalent to 1.25 mg/kg
bw per day) in gelatin capsules in the diet. Eggs and excreta were
collected dally. The hens were killed 4.5 hours after the last dose,
and liver, muscle and fat samples were collected. Of the total
radioactivity, 95.2% was recovered. Radioactivity in eggs plateaued 8
days after the start of dosing and reached 0.05 mg/kg. Most
radioactivity was found in the yolk and was a mixture of parent
compound and material that was closely associated with neutral lipids
and phosphatidyl cholines. In tissues, highest radioactivity was found
in the liver (0.37 µg/g). This radioactivity was composed of parent
compound (0.05 µg/g) and a mixture of very polar metabolites that were
not hydrolysed to significant amounts of 3-phenoxybenzoic acid or its
4-hydroxy derivative. Fat contained 0.08 µg/g and about 60% of the
residue was present as parent compound. Residues in muscle
(0.01-0.02 µ/g) were too low for characterization (Hutson & Stoydin,
1987).
2.1.1.3 Sheep
Two male sheep were each treated dermally (single dose) with a
mixture of 962 mg 14C-cypermethrin (both cyclopropyl- and
benzyl-labelled), equivalent to 21.9 mg/kg bw in acetone. After 24
hours (sheep 1) and 6 days (sheep 2) the animals were killed, and
radioactivity in fat, muscle, liver, kidney and skin (application
area) was measured. Urine and faeces were collected up to the time the
sheep were killed. One sheep treated with acetone served as a control.
Another sheep was treated orally (single dose) with 177 mg 14C-
cypermethrin mixture in a gelatin capsule (equivalent to 3.9 mg/kg
bw). Urine and faeces were collected for 2 days and tissues were
assayed. When applied dermally radioactivity was slowly absorbed
and eliminated. Less than 0.5% was excreted in urine within 24 hours
and only 2% over a 6-day period. Faecal elimination was also slow,
0.5% of the dose being eliminated in 6 days. About 30% of the applied
radioactivity was recovered from the application areas of both sheep.
Very little radioactivity was found in the tissues. In liver, kidney,
renal fat and subcutaneous fat (other than the application area),
residues ranged from 0.09 to 0.3 µg/g and muscle samples contained
0.03 to 0.06 µg/g.
Following oral treatment, the elimination of radioactivity was
rapid, 61% of the administered radioactivity being eliminated within
48 hours. Urinary elimination comprised 41% of the dose and faecal
elimination 20.5%. Residues found in tissues were comparable to those
found after dermal application. Most of the radioactivity in the fat
samples of all sheep was found to be parent compound. In muscle, liver
and kidney samples, only a small percentage of the total tissue
radioactivity was identified as parent compound (Crawford & Hutson,
1977).
2.1.1.4 Cattle
a) Cypermethrin
Two lactating Friesian cows were fed twice daily a diet
containing 0.2 mg 14C-benzyl-labelled cypermethrin/kg feed. Cow I
was treated for 20 days and cow II for 21 days. A control cow was
maintained under identical conditions. The radioactivity in milk,
urine and faeces was measured daily and after 20-21 days of dosing the
cows were sacrificed and blood, major organs and tissues were analysed
for radioactivity. Total recovery of the radioactivity was 97.8%. The
major route of excretion was via the urine (54%) and faeces (43%).
Milk contained 0.5% of the dose. The residues in muscle, blood and
brain were less than 0.005 µg/g. Liver contained 0.006 µg/g, kidney
0.004 µg/g, bile 0.027 µg/g, renal fat 0.011 µg/g and subcutaneous fat
0.009 µg/g.
The urinary metabolites were tentatively identified as
N-(3-phenoxy-benzoyl)glutamic acid, the major metabolite, and
3-(4-hydroxyphenoxy) benzoic acid O-sulphate, the minor metabolite
(ratio 4:1). In faeces 36% was eliminated as unchanged parent compound
(Hutson & Stoydin, 1976).
Two lactating Friesian cows were fed twice daily for 7 days a
diet containing 5 mg 14C-cypermethrin (cyclopropyl-labelled)/kg
feed. A third cow was treated similarly with 14C-benzyl-labelled
cypermethrin. A control cow was maintained under identical conditions.
The radioactivity in the milk, urine and faeces was measured daily.
After 7 days of dosing the cows were killed and their blood, major
organs and tissues were analysed for radioactivity. Total recovery of
radioactivity was 92.4% for the cows dosed with cyclopropyl-labelled
cypermethrin and 89.6% for the cow dosed with benzyl-labelled
cypermethrin. The major route of excretion was similar for both label
compounds. Urinary excretion accounted for about 49% of the dose while
faecal excretion accounted for about 38%. An equilibrium between
ingestion and excretion was reached after 3-4 days. Very low levels of
radioactivity were determined in milk (0.003-0.013 µg/g), tissues
(muscle, blood, brain 0.07 µg/g, liver, kidney 0.13 µg/g and renal and
subcutaneous fat 0.10 µg/g). The urinary metabolites included the
glutamic acid conjugate of 3-phenoxybenzoic acid (68%), 3-phenoxy-
benzoylglycine (16%) and 3-phenoxybenzoic acid (9%). 3-(4-Hydroxy-
phenoxy)benzoic acid and its O-sulfate conjugate appeared to be
present in only small amounts (1%) (Crawford, 1978).
One lactating cow was administered a diet containing 10 mg
14C-benzyl-labelled cypermethrin/kg feed twice daily for 7 days.
Milk, urine and faeces were collected daily for radioassay. The cow
was killed 16 hours after the last dose and samples of fat, muscle,
liver and kidney were analysed. An untreated cow was held as control.
Total recovery was 93%. Radioactivity was rapidly eliminated in equal
proportions in the urine and faeces. Radioactivity in milk was
< 0.2%. Analysis of milk revealed that the radioactivity was due to
unchanged cypermethrin, which was located mostly in the lipophilic
components (cream or butterfat). Radioactivity in tissues was
generally in the order: liver (0.21 µg/g) > kidney (0.11 µg/g) > fat
(0.1 µg/g) > blood (0.04 µg/g) > muscle (0.01 µg/g). The residue in
fat was largely unchanged cypermethrin. Radioactivity in liver and
kidney was due mainly to N-(3-phenoxybenzoyl)glutamic acid. The
liver and kidney metabolites were hydrolysed in hot acid to 3-phenoxy-
benzoic acid and its 4'-hydroxy derivative (Croucher et al., 1980).
b) alpha-Cypermethrin
One lactating cow received orally 125 mg 14C-alpha-cypermethrin
(benzyl labelled)/dose twice daily for 4 consecutive days (target dose
250 mg/day). Another cow received the same dose of unlabelled alpha-
cypermethrin. (The overall calculated daily dietary concentrations
were 19 and 14 mg/kg, respectively). The animals were killed 6 hours
after the last dose. Urine, faeces, milk, kidneys, liver, fat and
muscle were analysed for radioactivity.
The major route of excretion of radioactivity was via the faeces,
accounting for 34% of the total administered dose. Urinary excretion
accounted for 23% of the dose and milk less than 1%. Total radioactive
residues in milk accounted for 0.014 mg/kg on day 2 and rose to
0.2 mg/kg by day 4 of dosing. The major proportion (93%) of the milk
residues was confined to the cream fraction. Residues in tissues were
highest in liver, renal fat, omental fat, subcutaneous fat and kidney
(0.56, 0.48, 0.43, 0.39 and 0.22 mg/kg, respectively). Plasma
contained 0.08 mg/kg and muscle samples contained < 0.03 mg/kg
residues.
The liver and kidney contained a range of components. The liver
extract contained at least eight metabolites with a broad range of
polarities. One component (16%) had similar chromatographic properties
to alpha-cypermethrin. The kidney extract contained at least nine
metabolites with a broad range of polarities, one component (20% of
the profile) had similar chromatographic properties to alpha-
cypermethrin. Muscle, fat and milk contained mainly a single
component (muscle 85%, fat 91% and milk 97% of the exact profile),
which in each case had similar chromatographic properties to
alpha-cypermethrin. Urinary metabolites were analysed using HPLC.
The two major components (44% and 20%) had identical chromatographic
properties to N-(3-phenoxybenzoyl)glutamic acid and
N-(3-phenoxybenzoyl)glycine, respectively. A minor component
(3%) had identical chromatographic properties to 3-phenoxybenzoic
acid (Dunsire & Gifford, 1993; Morrison & Richardson, 1994).
2.1.2 Effects on liver enzymes
Six Wistar rats/sex were randomly selected from each of the
control and 1000 mg cypermethrin/kg food group at termination of a
2-year feeding study. The activity of hepatic p-nitroanisole
o-demethylase (PNOD) was determined in each of these rats. In male
rats treated with cypermethrin, PNOD activity, expressed as per gram
of liver, was significantly increased (38%). The total PNOD activity
of liver was also increased (30%), but, owing to great inter-animal
variation, statistical significance was not reached. Treated females
exhibited increases in PNOD activity when expressed per gram of liver
(21%) and per whole liver (39%), but only the latter was significant
(Potter & McAusland, 1980).
2.2 Toxicological data
2.2.1 Acute toxicity
Acute toxicity studies of cypermethrin and alpha-cypermethrin are
summarized in Tables 1 and 2.
Cypermethrin-induced signs of toxicity were typical of
cyano-containing pyrethroid intoxication. After oral administration
signs of intoxication included sedation, ataxia, splayed gait, tip-toe
walking, with occasional tremors and convulsions. These signs of
toxicity appeared within a few hours following dosing, and survivors
recovered within 3 days. The acute toxicity after dermal
administration is of a low order (Coombs et al., 1976).
alpha-Cypermethrin-induced clinical signs, like those of
cypermethrin, are typical of cyano-containing pyrethroid. The observed
signs included ataxia, abasia, gait abnormalities, choreoathetosis,
tip-toe walking, and increased salivation, lacrimation, piloerection,
tremor and clonic convulsions. Surviving animals recovered within 7
days (Rose, 1982b, 1983).
2.2.2 Short-term toxicity studies
2.2.2.1 Mice
Groups of eight mice (CD-1)/sex received diets containing 0, 200,
400, 800, 1200 or 1600 mg alpha-cypermethrin/kg feed for 29 days. One
male fed 1600 mg/kg and one female fed 1200 mg/kg were killed in a
moribund condition. These animals showed neurological disturbances.
Mice at 1200 and 1600 mg/kg developed ungroomed coats, ataxia/abnormal
gait, over-activity or hunched posture. At 800 mg/kg some animals
had ungroomed coats and abnormal gait. Body weight (gain) was
dose-relatedly decreased in mice at 1200 and 1600 mg/kg and in females
at 800 mg/kg. A similar, but less marked effect was apparent in males
receiving 800 mg/kg and females receiving 400 mg/kg. Food consumption
was lowered in rats at 1200 and 1600 mg/kg during the first 2 weeks. A
depression in lymphocyte numbers was noted in males receiving 1600
mg/kg. ALAT and ASAT levels were increased in males at 1600 mg/kg.
Urea levels were increased in all dosed males and females, without
clear dose relation. Plasma albumin and A/G ratio were decreased in
males at 1600 mg/kg. Relative kidney weight was increased in males at
800, 1200 and 1600 mg/kg. Relative lung weight was increased in males
at 1600 mg/kg. No macroscopic or microscopic effects were seen. The
NOEL was 400 mg/kg feed, equal to 57 mg/kg bw per day (Green, 1993).
Groups of 12 mice (CD-1)/sex received diets containing 0, 50, 250
or 1000 mg alpha-cypermethrin/kg feed for 13 weeks. Four males at
1000 mg/kg died during week 12 probably due to stress of treatment and
refusal of food and water. The relative kidney weights of these
animals appeared to be slightly higher. One mouse at 250 mg/kg died.
Clinical signs included thin build, ungroomed coat, hair loss and
encrustations of the dorsal body surface in animals receiving
1000 mg/kg, ungroomed haircoat in mice at 250 mg/kg, hair loss and
encrustations in males at 250 mg/kg and hair loss in two males at
50 mg/kg. Body weight gain was markedly lower in mice at 1000 mg/kg
and lower in mice at 250 mg/kg. Food consumption was slightly higher
in mice at 1000 mg/kg. Overall food conversion efficiency of animals
receiving 1000 mg/kg (and to a lesser extent 250 mg/kg) was lower than
that of controls. In males at 1000 mg/kg, PVC, Hb, RBC, total WBC and
leucocyte counts were decreased. ASAT was increased in a dose-
dependent manner in males at 250 and 1000 mg/kg, and glucose was
decreased in males at 1000 mg/kg. Serum AP was increased in females at
1000 mg/kg. Urinary specific gravity was increased in mice at the
highest dose level. In males at 1000 mg/kg relative brain, adrenal,
heart, kidney, liver, spleen, lung and testes weight were increased.
In females relative liver weight was increased at 250 and 1000 mg/kg
and relative brain and spleen weight at 1000 mg/kg. Two males and
eleven females treated at 1000 mg/kg were considered to be emaciated
at necropsy. No histological changes were observed. Owing to the hair
loss seen at 50 mg/kg, a NOEL was not identified (Amyes et al.,
1994).
2.2.2.2 Rats
a) Cypermethrin
In a 5-week feeding study, groups of six rats/sex (Charles River)
received diets containing 0, 25, 100, 250, 750 or 1500 mg cyper-
methrin/kg feed. No effects were found in haematological, macroscopic
or microscopic examinations. In the highest dose group, rats developed
piloerection, nervousness and incoordinated movement from week
2 onwards. Body weight gain, food intake and terminal body weight
were all reduced for both sexes at 1500 mg/kg. Relative liver
weight and blood urea levels were increased at the highest dose level
(only a summary available) (Coombs et al., 1976).
Table 1. Acute toxicity of cypermethrln in animals
Species1 Route Purity2 LD50 Remark Reference
mg/kg bw
Mouse oral 98.1% 88 5% in corn oil Rose, 1982a
Mouse oral ? 82 5% in corn oil Coombs et al., 1976
Mouse oral 98.1% 1126 40% in DMSO Rose, 1982a
Mouse oral 98.1% 657 50% aq. susp. Rose, 1982a
Mouse percutan. 98.1% > 100 5% in corn oil Rose, 1982a
Rat oral ? 251 5% in corn oil Coombs et al., 1976
Rat oral 98.1% 4000 40% in DMSO Rose, 1982a
Rat oral 98.1% 3423 50% aq. susp. Rose, 1982a
Rat oral ? > 2000 techn. conc. Price, 1984
Rat percutan. ? > 2000 techn. conc. Price, 1984
Rat percutan. ? > 1600 40% in xylene Coombs et al., 1976
Rat i.p. ? approx. 40% in corn oil Coombs et al., 1976
2600
Hamster oral ? 203 5% in corn oil Coombs et al., 1976
Guinea-pig oral ? approx. 20% in corn oil Coombs et al., 1976
500
1 Both male and female animals were used in all of the studies
2 cis:trans isomer ratio 51:49; ? - unknown
Table 2. Acute toxicity of alpha-cypermethrin in animals
Species1 Route Purity LD50 Remark Reference
(%) mg/kg bw
or mg/m3
Mouse oral 96.6 35 5% in corn oil Rose, 1982b
Mouse oral 96.6 762 40% in DMSO Rose, 1982b
Mouse oral 96.6 798 50% aq. suspension Rose, 1982b
Mouse percut. 96.6 > 100 5% in corn oil Rose, 1982b
Rat oral techn. 64 50% aq. CMC Gardner, 1993
Rat oral 96.6 4000 40% in DMSO Rose, 1982b
Rat oral 96.6 > 5000 50% aq. suspension Rose, 1982b
Rat dermal techn. > 2000 undiluted Gardner, 1993
Rat inhal 95.6 1590 as aerosol Jackson, 1993
(MMAD 9 µm)
1 Both male and female animals were used in all of the studies
Groups of 12 rats/sex (Wistar) were fed diets containing 0 (24
rats/sex), 25, 100, 400 or 1600 mg cypermethrin/kg feed for 91-95
days. There were no deaths, no clinical signs and no macroscopic or
microscopic changes. In the highest dose group a reduced body weight
gain was observed. During the first week reduced food consumption was
observed in both sexes at 1600 mg/kg. This could be attributed to the
palatability of the diet. During week 13, females at 1600 mg/kg had
reduced food intake. Males at 1600 mg/kg showed decreases in Hb, MCV
and eosinophil counts, and an increase in prothrombin time. In males
at 400 mg/kg there was also a decrease in eosinophil numbers. The mean
plasma urea concentration in the 1600 mg/kg groups was increased.
Relative liver weight was increased in males and females at 1600 mg/kg
and in males at 400 mg/kg. Relative kidney weight was increased in
males at 1600 mg/kg. The NOEL was 100 mg/kg feed, equivalent to
5 mg/kg bw per day (Pickering, 1981).
b) alpha-Cypermethrin
Groups of 10 Wistar rats/sex received diets containing 0 (20
rats/sex), 20, 100, 200, 400 or 800 mg alpha-cypermethrin/kg feed for
5 weeks. Observations included mortality, clinical signs, body weight
and food consumption, haematology, clinical chemistry, urinalysis,
organ weight, and macroscopic and microscopic lesions.
One male at 800 mg/kg died unrelated to treatment and two males
at 800 mg/kg were killed, owing to signs of severe neurological
disturbance. Both sexes at 800 mg/kg produced abnormal gait and
increased sensitivity to noise. Abnormal gait was seen in one male at
400 mg/kg. The mean body weights and food intake of both males and
females fed 400 or 800 mg/kg were significantly lower than those of
controls. In both males and females at 800 mg/kg and in males at
400 mg/kg prothrombin time was increased. In males at 800 mg/kg the
kaolin-cephalin coagulation time was increased and the percentage of
polymorphic neutrophils was increased at 800 mg/kg. In females
platelet count, total leucocyte count and absolute values of
polymorphic neutrophils and lymphocytes were increased, but the Hb and
haematocrit values were decreased. The MCV was decreased at 200, 400
and 800 mg/kg but not in a dose-related manner. Relative brain, liver
and kidney weights were increased in males fed 800 mg/kg and relative
brain and liver weights were increased in males fed 400 mg/kg. In
females at 800 mg/kg relative brain, liver and kidney weights were
increased. One male at 800 mg/kg that was removed from the study
showed scanty axonal lesions of the sciatic nerves. The NOEL was
200 mg/kg feed, equivalent to 10 mg/kg bw per day (Thorpe, 1982).
In a range finding study groups of five male and five female rats
were fed diets containing a-cypermethrin at concentrations of 0, 50,
200, 800 or 1200 mg/kg feed for up to 6 weeks. No effects were found
on food consumption, haematology and clinical chemistry and
microscopy. All rats at 1200 mg/kg and all males at 800 mg/kg were
killed during weeks 2 to 4 because of severe clinical signs. These
signs included high stepping, splayed gait, abasia and hyper-
sensitivity. Cachexia was seen in extreme cases. There were no
significant changes in fore or hind limb grip strength or hind limb
landing foot splay. Female rats fed 800 mg/kg had a lower mean body
weight and food intake compared to controls. These females also had a
lower leucocyte count. Microscopic examination revealed lymphocytolysis
and iymphocyte depletion of the cortical region of the thymus in males
fed 800 mg/kg and males and females at 1200 mg/kg (Fokkema, 1994a).
Groups of 30 male and 30 female Wistar rats received diets
containing 0 (60 rats/sex), 20, 60, 180 or 540 mg alpha-
cypermethrin/kg feed for 90 days. After 6 weeks 10 rats/sex
(controls 20/sex) were sacrificed for interim examinations. Three
males at 540 mg/kg developed an abnormal gait consisting of splayed
hind limbs. Skin sores and fur loss were observed in all males with
the highest incidence at 540 mg/kg and in females at 0 and 540 mg/kg.
Two males, one from controls and one at 540 mg/kg were removed because
of severe skin lesions. Reduced body weight and reduced food
consumption were seen in rats at 540 mg/kg. During the second part of
the study, body weight gain was also reduced in males at 60 and
180 mg/kg, but not in a clearly dose-related manner. Food consumption
was lower at 60 mg/kg than at 180 mg/kg. At termination the Hb value
was decreased in males and females at 540 mg/kg. MCV and MCHC were
decreased in females at 540 mg/kg. Platelet counts were increased in
males and females at 540 mg/kg. The number of lymphocytes was
increased and the number of eosinophils was decreased in males at
540 mg/kg. Urea concentration was increased in females at 540 mg/kg.
AP was decreased in females at 180 and 540 mg/kg. Urinary volume was
decreased in females at 540 mg/kg and specific gravity was increased
in males and females at 540 mg/kg. In females fed 540 mg/kg, relative
spleen, heart and brain weights were increased. In both sexes at
540 mg/kg relative kidney weight was increased and relative liver
weight was increased in both sexes at 180 and 540 mg/kg. Relative
testis weight was increased in males at 540 mg/kg. Histopathological
investigations showed a solitary form of axonal degeneration,
affecting the fibre only, in the sciatic nerve, in two males fed
540 mg/kg, but there were no clinical signs of toxicity. The NOEL was
60 mg/kg feed, equivalent to 3 mg/kg bw per day (Clark, 1982).
2.2.2.3 Rabbits
Occluded dermal applications of 0, 2, 20 or 200 mg cyper-
methrin/kg bw in PEG 300 were made to abraded and intact skin of
groups of 10 male and 10 female NZW rabbits/sex. The applications were
made for 6 h/day, 5 days/week for 3 weeks. No effects were observed on
haematology, clinical chemistry or following macroscopic or micro-
scopic examination. Slight to severe skin irritation was observed
in rabbits at 200 mg/kg bw and slight to moderate skin irritation was
observed in rabbits receiving 2 and 20 mg/kg bw. Decreased food
consumption and body weight gain was observed in rabbits treated with
200 mg/kg bw. Absolute and relative gonad weights were reduced in male
rabbits at 200 mg/kg bw. The NOEL was 20 mg/kg bw (Henderson &
Parkinson, 1981).
2.2.2.4 Dogs
a) Cypermethrin
In a 5-week feeding study, groups of three beagle dogs/sex
received diets containing 0, 15, 150 or 1500 mg cypermethrin/kg feed.
No effects were found on body weight, food consumption, ophthalmo-
scopy, haematology, clinical chemistry, macroscopy, liver weight or
following microscopic examination. In the highest dose group dogs
developed signs of intoxication, including apprehension, diarrhoea
and vomiting, licking and chewing of the paws, whole body tremors, a
stiff exaggerated hind leg gait, ataxia, inappetence, and decreased
body weight gain. Relative thyroid weight and blood urea levels were
increased and blood glucose levels were decreased (only a summary
available) (Coombs et al., 1976).
Groups of beagle dogs (four/sex) were fed cypermethrin (purity
98%) in the diet at concentrations of 0, 5, 50, 500 or 1500 mg/kg feed
for 13 weeks. No mortality occurred. Dogs at 1500 mg/kg developed
diarrhoea, anorexia and tremors as well as ataxia, incoordination and
hyperaesthesia. Also a reduced food intake and body weight gain was
seen. Due to the clinical signs two dogs/sex at 1500 mg/kg had to be
killed. Minor variations in haematology were observed; the kaolin-
cephalin clotting time was consistently lower throughout the study in
female dogs at 500 mg/kg. However, because of the lack of a dose-
response relationship, the variability of this parameter throughout
the study, and the fact that there were no changes in the prothrombin
time, this finding is not considered to be relevant. No effects were
seen on clinical chemistry parameters or on organ weights. Microscopic
examination of tissues and organs revealed a non-specific focal
bronchopneumonia in the lungs of the dogs at 1500 mg/kg. A pink colour
of the optic disc was noted in ophthalmological examinations. The NOEL
was 500 mg/kg feed, equivalent to 12.5 mg/kg bw per day (Buckwell &
Butterworth, 1977).
A further experiment was undertaken to determine the cause of the
pink discoloration, which occurred in all groups. Cypermethrin was fed
to two groups of three male dogs at concentrations of 0 or 500 mg/kg
food for 13 weeks. Specific ophthalmological examinations were made to
evaluate the degree of coloration of the optic disc. At the end of 13
weeks, there were no consistent differences between the colour of the
optic discs of the treated dogs and the controls (Buckwell &
Butterworth, 1977).
b) alpha-Cypermethrin
This study was conducted in two parts.
In the first part one male and one female beagle dog were fed
alpha-cypermethrin in the diet at concentrations of 200 mg/kg feed for
7 days, 400 mg/kg feed for 2 days and 300 mg/kg feed for 7 days. Due
to severe intoxication of the animals receiving 400 mg/kg, dosing was
discontinued after 2 days. The animals were fed the control diet and
in week 3 treatment was commenced at 300 mg/kg. After the third week
the dogs were killed.
Clinical signs including ataxia, body tremors, subdued behaviour,
head nodding, food regurgitation, diminished response to stimuli and
inflammation of gums and tongue were obtained when dogs were dosed
with 300 and 400 mg/kg. Body weight loss was observed at 300 mg/kg.
Examination of the cellular composition of blood and the chemical
components of plasma showed no abnormalities, and no macroscopic
changes were observed.
In the second part one male and one female beagle dog were fed
alpha-cypermethrin in the diet at 300 mg/kg food for 3 days (male dog)
or 4 days (female dog), and then 250 mg/kg food for 7 days. At
300 mg/kg the same effects were obtained as above. At 250 mg/kg only
the female dog developed the clinical signs shown by the dogs treated
with 300 and 400 mg/kg (Greenough & Goburdhun, 1984).
Groups of four beagle dogs/sex received diets containing dose
levels of 0, 30, 90 or 270 (six dogs/sex) mg alpha-cypermethrin/kg
feed for 13 weeks. No effects on mortality, body weight, food
consumption, ophthalmoscopy, haematology, clinical chemistry,
macroscopy, organ weights or microscopy were seen. One female at
270 mg/kg was killed, because of severe head and body tremors, ataxia,
poor limb coordination, inflamed gums and elevated temperature. All
dogs at 270 mg/kg developed marked clinical signs, including body
tremors, head nodding, lip licking, subduedness, ataxia, agitation and
high stepping gait. The NOEL was 90 mg/kg, equivalent to 2.25 mg/kg bw
per day (Greenough et al., 1984).
Four groups of beagle dogs (four/sex) received via the diet 0,
60, 120 or 240 mg alpha-cypermethrin/kg feed dally for 52 weeks. No
effects were observed on body weight, food consumption, ophthalmo-
scopy, haematology, clinical chemistry, urinalysis or organ weights,
and no changes were observed in macroscopic or microscopic examinations.
Two males at 240 mg/kg developed skin reddening on the tail. Abdominal
skin reddening and alopecia were seen in another male at 240 mg/kg and
one female at 120 mg/kg. The tail reddening caused obvious irritation
and resulted in ulceration and necrosis in one male leading to
amputation of part of the tail. The NOEL was 60 mg/kg feed, equivalent
to 1.5 mg/kg bw per day (Dean & Jackson, 1995).
2.2.3 Long-term toxicity/carcinogenicity studies
2.2.3.1 Mice
Groups of mice (70/sex, SPF-Swiss-derived) received diets
containing 0 (2 groups), 100, 400 or 1600 mg cypermethrin/kg feed for
up to 101 weeks. Ten mice/sex were killed after 52 weeks for interim
necropsy. Observations included mortality, clinical signs, body weight
gain, food consumption, haematology, clinical chemistry, organ
weights, and macroscopy and microscopy. Body weight gain of both males
and females at 1600 mg/kg was reduced when compared to the combined
control groups. Several haematological changes, consistent with mild
anaemia, were found in the 1600 mg/kg group at the interim kill, but
not at termination. At interim kill and at termination, an increase in
thrombocytosis and absolute and relative liver weight was seen in
males at 1600 mg/kg. An increase in the incidence of benign
alveologenic tumours was observed in females at 1600 mg/kg, but was
within historical control incidence. In this study the NOEL was
400 mg/kg feed, equal to 57 mg/kg bw per day (Lindsay et al., 1982).
2.2.3.2 Rats
Groups of 48 Wistar rats/sex received diets containing 1, 10, 100
or 1000 mg cypermethrin/kg feed for 2 years. The control group
consisted of 96 rats/sex and was fed untreated diet for 2 years. After
6 and 12 months six rats/sex were sacrificed and after 18 months 12
rats/sex were sacrificed (controls, respectively, 12 and 24 rats/sex).
The only effects observed were reductions in body weight and food
consumption in males and females at 1000 mg/kg. No dose-related
effects were observed on mortality, clinical chemistry, haematology,
clinical chemistry, organ weights, macroscopy or microscopy. No
difference between control and treated groups were found in the
sciatic nerves. There was no increase in compound-related tumours. The
NOEL was 100 mg/kg feed, equivalent to 5 mg/kg bw per day (McAusland
et al., 1978).
2.2.3.3 Dogs
Groups of four beagle dogs/sex (5´-7 months of age) were fed
diets containing 0, 3, 30, 300 or 1000 mg cypermethrin/kg food for 2
years. Groups of four dogs/sex were allocated to a satellite study and
received a diet containing 0, 300 or 1000 mg/kg cypermethrin (data
concerning this satellite group have only been included in the report
to aid interpretation of the results of the main study). Due to severe
signs of intoxication observed at the 1000 mg/kg level, the
concentration was reduced to 750 mg/kg at week 4 of the study and,
when signs of intoxication persisted during weeks 6-8, animals in the
high-dose group were fed a control diet for 10 days to allow them to
recover. Following the 10 days of control diet, the dogs were fed
cypermethrin at a concentration of 600 mg/kg food for the remainder of
the study. No effects were observed on ophthalmoscopy, clinical
chemistry, organ weights, macroscopy or microscopy. No abnormalities
were found in the sciatic nerves, brain or spinal cord. One male dog
in the satellite group fed 1000 mg/kg convulsed and died. Signs of
intoxication appeared within 24 hours of the initiation of dosing and
consisted of licking and chewing of the paws, a stiff high stepping
gait, whole body tremors, head shaking, incoordination, ataxia and, in
some cases, convulsions. These signs were observed at 1000 mg/kg and
to a lesser extent at 750 mg/kg, but not at 600 mg/kg. The body
weights of male dogs in the highest dose group were significantly
lower than the controls, probably due to initial reduction in food
consumption observed at 1000 mg/kg. In the satellite group no effect
on body weight gain was seen. Apart from some occasionally decreased
sodium levels in the males given the highest dose, no consistent
haematological changes were seen. The NOEL was 300 mg/kg feed,
equivalent to 7.5 mg/kg bw per day (Buckwell, 1981).
2.2.4 Reproductive toxicity studies
Groups of 30 rats/sex (Wistar) received diets containing 0, 10,
100 and 500 mg cypermethrin/kg feed for 5 weeks prior to mating and
then throughout pregnancy and lactation for three successive
generations. Two litters were bred per generation. The first litters
were discarded at weaning. Males and females from the second litter
were randomly selected to breed the next generation. A significant
reduction in body weight gain was seen in the male and female parent
rats receiving 500 mg/kg in all three generations. This was correlated
with a reduction in food consumption. Litter size was reduced at
500 mg/kg in the F1a litter at birth and after 7 and 21 days. Litter
weights were reduced at 500 mg/kg in the F1a, litters on days 7, 14
and 21 of lactation. No other effects on fertility or reproduction
parameters were found. The NOEL for maternal and reproduction toxicity
was 100 mg/kg feed, equivalent to 5 mg/kg bw per day (Hend et al.,
1978; Fish, 1979; Thorpe, 1985).
2.2.5 Special studies on embryotoxicity and teratogenicity
2.2.5.1 Rats
a) Cypermethrin
Groups of 25 pregnant female rats (Sprague-Dawley) received by
gavage 0, 17.5, 35 or 70 mg cypermethrin/kg bw per day in corn oil
during days 6 to 15 of gestation. The females were sacrificed on day
21 of gestation for examination of their uterine contents. One female
at 70 mg/kg bw per day was found dead and one female at 70 mg/kg bw
per day was killed for ethical reasons following severe convulsions.
Eleven out of 25 females at the 70 mg/kg bw per day group showed
neurological disturbances (ataxia, convulsions, hypersensitivity to
noise). A dose-related reduction in body weight gain was observed in
the groups given 35 and 70 mg/kg bw per day. There were no indications
of any embryotoxic or teratogenic effects. The NOEL for maternal
toxicity was 17.5 mg/kg bw per day and the NOEL for embryotoxicity was
70 mg/kg bw per day (Tesh et al., 1978).
b) alpha-Cypermethrin
In a range-finding study, five pregnant female Sprague-Dawley
rats received daily, by gavage, 0, 3, 9, 15 or 18 mg alpha-cyper-
methrin/kg bw in corn oil during days 6-15 of gestation. Maternal
body weights, food consumption and clinical observations were
recorded. On day 20 of gestation the females were killed and the
fetuses were weighed, sexed and externally examined. Four dams at
18 mg/kg bw per day and one dam at 15 mg/kg bw per day showed hindlimb
splay and unsteady gait during dosing. Mean body weight gain was
reduced in a dose-related manner at 9, 15 and 18 mg/kg bw per day and
at 15 and 18 mg/kg bw per day food consumption was reduced. No other
treatment-related abnormalities were observed (Irvine & Twomey, 1994).
In another study, groups of 24 pregnant female Sprague-Dawley
rats received by gavage 0, 3, 9 or 18 mg alpha-cypermethrin (purity
95.6%)/kg bw per day in corn oil during days 6-15 of gestation.
Following marked clinical signs of toxicity the dose level of 18 mg/kg
bw per day was lowered to 15 mg/kg bw per day on day 10 of gestation.
Clinical signs, body weights and food consumption were recorded. On
day 20 of gestation the females were killed and necropsied. The
fetuses were weighed, sexed and examined for external, visceral and
skeletal abnormalities.
Females at 18 mg/kg bw per day showed unsteady gait,
piloerection, limb splay and hypersensitivity to sound. After
reduction of the dose level the signs were similar but less marked.
After treatment with 18/15 mg/kg bw per day a lowered body weight gain
and food consumption was seen. At 9 mg/kg bw per day a slight body
weight reduction was seen. Mean fetal weights were slightly reduced at
18/15 mg/kg bw per day. There was no indication for teratogenicity.
The NOEL for maternal and fetal toxicity was 9 mg/kg bw per day
(Irvine, 1994c).
2.2.5.2 Rabbits
a) Cypermethrin
In a range-finding study, groups of four female pregnant rabbits
(NZW) received during days 6 to 18 of gestation by gavage 0, 25, 50,
100 or 120 mg cypermethrin/kg bw per day in corn oil. The dams were
sacrificed on day 29 of gestation. No adverse effects were seen in the
mothers and fetuses (Tesh et al., 1984a).
Groups of 16 pregnant NZW rabbits received by gavage 0, 20, 50 or
120 mg cypermethrin/kg bw per day in corn oil during days 6 to 18 of
gestation. The dams were killed on day 29 of gestation. One control
female, three females receiving 20 mg/kg bw per day and two females in
each of the groups receiving 50 and 120 mg/kg bw per day were killed
for ethical reasons. Necropsy revealed evidence of respiratory tract
infection and/or gastrointestinal tract infection not related to the
substance. Two females at 20 mg/kg bw per day and two females at
120 mg/kg bw per day aborted during the post-treatment phase of the
investigation. The number of implantations, live young and
resorptions, pre- and post-implantation losses, and fetal and
placental weights were unaffected by treatment. There was no
indication for embryotoxicity or teratogenicity. The NOEL for
embryo-toxicity was 120 mg/kg bw per day (Tesh et al., 1984b, 1988).
Groups of pregnant rabbits (20 rabbits/group, 30 rabbits were
used as an additional control group) were administered cypermethrin
dissolved in corn oil at dose levels of 0, 3, 10 or 30 mg/kg bw per
day orally from days 6 to 18 of gestation. On day 28 of gestation the
rabbits were killed and examination was made of live fetuses, dead
fetuses, resorption sites and corpora lutea. Live fetuses were
maintained for 24 hours to assess viability. Fetuses were also
examined for gross somatic and skeletal deformities. There was no
significant mortality or difference in weight gain during the period
of gestation. There were no significant differences between control
and test groups with respect to pregnancy, fetal death and survival.
Although a wide range of skeletal and visceral abnormalities was found
in the course of the study, there were no differences between control
and test groups with respect to abnormalities. It was concluded that
oral dosing up to 30 mg/kg bw during the major period of organogenesis
resulted in no teratogenic effects in offspring (FAO, 1980).
b) alpha-Cypermethrin
In a range finding study groups of five mated female NZW rabbits
received by gavage 0, 5, 15, 25 or 30 mg a-cypermethrin/kg bw per day
as solutions in corn oil, during days 7-19 of gestation. On day 28 of
pregnancy the females were killed and a necropsy was performed. The
fetuses were weighed, sexed and externally examined. One female each
at 15 and 25 mg/kg bw per day was killed prematurely. At 25 and
30 mg/kg bw per day marked reductions in body weight and food
consumption were seen. There was no indication for either
embryotoxicity or teratogenicity (Irvine, 1994a).
In another study, groups of 16 pregnant NZW rabbits received by
gavage 0, 3, 15 or 30 mg alpha-cypermethrin/kg bw per day in corn oil
during days 7-19 of gestation. Maternal clinical signs, body weight
and food consumption were recorded. The females were killed on day 28
of pregnancy. The uterus was weighed and the numbers of corpora lutea,
implantations and live fetuses were counted. The fetuses were weighed,
sexed and examined for external, visceral and skeletal abnormalities.
Two control females, three at 15 mg/kg bw per day and two at
30 mg/kg bw per day were killed, because of severe weight loss and low
food consumption. One female at 15 mg/kg bw aborted on day 28. In all
groups, including controls, there was a similar mean body weight loss
after the onset of dosing, which continued until day 11. At 30 mg/kg
bw per day there was a further reduction in mean body weight gain
towards the end of the dosing period. Food consumption reflected the
changes in mean body weight gain. The NOEL for maternal toxicity was
3 mg/kg bw per day and the NOEL for embryotoxicity was 30 mg/kg bw.
There was no indication for teratogenicity (Irvine, 1994b).
2.2.6 Special studies on genotoxicity
Results of genotoxicity studies carried out cypermethrin and
alpha-cypermethrin are summarized in Tables 3 and 4.
2.2.7 Special studies on neurotoxicity
2.2.7.1 Rats
a) Cypermethrin
Groups of 6 or 12 rats/sex were administered single oral doses of
100, 200 or 400 mg cypermethrin/kg bw (purity 97%) as a 5% dispersion
in corn oil. The observation period was 9 days. The rats were then
killed and examined histologically. All rats showed signs of
intoxication. At 400 mg/kg bw, within 4 hours of dosing, rats
developed signs of intoxication, including coarse tremors, spasmodic
movements of the body and tail and bleeding from the nose. Tip-toe
walking was also seen in some rats. All animals, except one, were
killed, owing to the severity of the signs. Histological examination
revealed swelling of the myelin sheaths and breaks of some of the
axons of the sciatic nerves. At 200 mg/kg bw similar effects were
observed; eight rats of each sex died or were killed within 48 h of
dosing. The remaining four rats survived the observation period. At
100 mg/kg bw all animals survived the 9 days. One female out of 12
showed minimal lesions in the sciatic nerve in this group. A NOEL
could not be determined (Carter & Butterworth, 1976).
In a neuromuscular dysfunction test, Wistar rats (10/sex) were
treated by gavage with 0, 25, 50, 100, 150 or 200 mg cypermethrin/kg
bw per day in DMSO for 7 consecutive days. The rats were killed 3-4
weeks after the start of dosing, and right and left sciatic/posterior
tibial nerves were analysed. Neuromuscular function was assessed by
means of the inclined plane test and peripheral nerve damage by
reference to ß-glucuronidase and ß-galactosidase activity increases in
nerve tissue homogenates.
Table 3. Results of mutagenicity assays on cypermethrin
Test system Test object Concentration Purity Results References
(%)
In vitro
Gene mutations S. typhimurium 0.2-2000 93.5 negative2 Brooks,
assay TA1525, TA100, µg/plate1 1980
TA1538, TA98, Dean,
TA1537 1981
Gene mutations E.coli WP2 or 0.2-2000 93.5 negative2 Dean,
assay WP2 uvrA µ/plate1 1981
Mitotic gene Saccharomyces 0.01-5.0 93.5 negative Dean,
conversions cerevisiae JD1 mg/litre1 1981
assay
Host-mediated S. cerevisiae 25 and 50 93.5 negative Brooks,
assay JD1 mg/kg bw 1980
in mice
Cell BHK 21/C113 31.25-250 93.5 negative Dean,
transformation cells µg/ml1 1981
assay
Chromosomal RL4 liver cells 7.5-30 µg/ml 93.5 negative Dean,
aberrations 1981
assay
Table 3. Results of mutagenicity assays on cypermethrin (cont'd).
Test system Test object Concentration Purity Results References
(%)
In vivo
Chromosome Chinese hamster 2x oral dose of ? negative Dean,
aberration 20 or 40 mg/kg 1977
assay in bone bw (2 successive
marrow days)
Dominant mouse single oral dose ? negative Dean
lethal assay of 6.25, 12.5 or et al.,
25 mg/kg bw or 1977
5 daily doses of
2.5 or 5 mg/kg
bw per day
Host-mediated mouse/ orally 0, 25, 50 ? negative JMPR,
assay S. cerevisiae mg/kg 1979
DNA damage CD rats m 100 mg/kg, f 150 ? negative Creedy &
assay mg/kg; 1, 4, 16 Wooder,
hour exposure 1977
Wistar rats m 300 mg/kg; 1, 4, negative Creedy &
16 hour exposure Wooder,
f 450 mg/kg, 1 hour 1977
exposure f 337.5
mg/kg, 4 and 16
hour exposure
1 with and without metabolic activation
2 at 200 and 2000 µg/plate, formation of visible droplets in the top agar was seen
Table 4. Results of mutagenicity assays on alpha-cypermethrin
Test system Test object Concentration Purity Results References
(%)
In vitro
Gene mutations S. typhimurium 31.5-5000 95.6 negative Brooks &
assay1 TA98, TA100, µg/ml (no toxicity) Wiggins,
TA1535, TA1537 1992
TA1538
E. coli WP2 uvrA
Gene mutations S. cerevisiae 31.25-4000 95.8 negative Brooks,
assay1 XV185-14C µg/ml 1984
Gene mutations1 L5178Y mouse 3.3-50 µg/ml 95.4 negative Vanderwaart,
assay lymphoma cells 1994
Chromosomal human peripheral -act: 93.75-1000 95.6 negative Brooks &
aberrations lymphocytes µg/ml precipitation Wiggins,
assay1 +act: 125-1000 was seen 1993
µg/ml
In vivo
Chromosomal rat femoral single oral dose 95.8 negative Clare &
aberrations bone marrow 2-8 mg/kg3 Wiggins,
1984
Table 4. Results of mutagenicity assays on alpha-cypermethrin (cont'd).
Test system Test object Concentration Purity Results References
(%)
Micronucleus mouse single oral dose 95.4 negative Vanderwaart,
assay 1-10 mg/kg bw 1995
Alkaline rat single oral dose 96.5 negative Wooder,
elution 40 mg/kg, 6 1981
analysis hours exposure
assay2
1 With and without metabolic activation
2 The effect of alpha-cypermethrin on the integrity of rat liver DNA was investigated
by this method
3 Initially doses of 10, 20 or 40 mg/kg were used, but the animals exhibited severe
signs of toxicity and the experiment was terminated at these doses. Surviving
females were evaluated for chromosomal damage and none was observed
At 200 mg/kg bw per day 50% of the males and 62.5% of the females
died, at 150 mg/kg bw per day two females and one male died and at
100 mg/kg bw per day one female died but no males. No mortalities
occurred in the other groups. A dose-related (in severity and
duration) increase was seen in clinical signs at doses > 100 mg/kg bw.
These signs included salivation, ataxia, splayed hind limb gait,
hyperexcitability to auditory stimuli, tremor and choreoathetosis. At
the same dose levels body weight gain was also reduced.
A dose-related transient functional impairment was found in rats
treated with cypermethrin in the inclined plane test. This effect was
maximal at the end of the 7-day subacute dosing regimen. At doses
which caused mortality, significant increases in ß-glucuronidase and
ß-galactosidase were found 3-4 weeks after the start of dosing in the
distal portion of the sciatic/posterior tibial nerves. There was no
direct correlation between the time course of the neuromuscular
function and the neurobiochemical changes. The NOEL was 50 mg/kg bw
per day (Rose & Dewar, 1983).
The effects of some pyrethroids, including cypermethrin, on
amplitude and pre-pulse inhibition of the acoustic startle reflex were
studied in male Wistar rats. The pyrethroids were suspended in corn
oil. Cypermethrin was given orally at dose levels of 0, 0.5, 1 or
2 mg/kg bw to 12 males. Each animal received all doses of cyper-
methrin. The intersession interval was one week. Animal behaviour
was observed by the experimenter before and after the test session for
a period of 10 minutes and the effects of the pyrethroids on overt
behaviour were measured by scoring the presence of pawing and
salivation, burrowing, hyperactivity, hyperreactivity to an external
stimulus, fine tremors of low intensity and fluid loss. Neither
cypermethrin nor the other pyrethroids tested affected the amplitude
or the latency of the startle reflex (Hijzen et al., 1988).
b) alpha-Cypermethrin
The acute neurotoxicity of alpha-cypermethrin was studied in
Crl:CD:BR rats in two separate acute studies, each using four groups
of ten animals/sex or five rats per sex (additional study). The groups
received a single dose of 0, 4, 20 or 40 mg alpha-cypermethrin/kg bw
in corn oil. During the 14-day observation period, clinical signs and
body weight were analysed. In the main study a detailed clinical
assessment for neurotoxicological effects was performed. This included
a functional observational battery (FOB) and measurements of fore and
hind limb grip strength, hindlimb landing foot splay and motor
activity. In each study five rats/sex were killed on day 15 and brain,
eyes, muscle, nerves, spinal cord and spinal ganglia were analysed.
One male rat in each of the 20 and 40 mg/kg bw groups of the
additional study was found dead on the day after dosing. Clinical
signs were seen in male rats dosed with 20 and 40 mg/kg bw. The signs
(similar in both studies) developed between 3 to 8 hours after dosing
and resolved by three days after dosing. The signs included abnormal/
splayed gait, thrashing, prostration, vocalization, piloerection,
hunched posture, unkempt appearance, soiled/stained body areas and
diarrhoea. The signs in females were similar but lower in frequency.
In addition to these signs there were also isolated cases of
twitching, tremors, abasia, hypersensitivity, pale eyes, soft faeces
and thinning of the fur.
During FOB conducted 5 hours after dosing, gait abnormalities and
clinical signs of increased reactivity were seen in most male rats
dosed with 20 and 40 mg/kg bw. In females the signs were less
frequent. In the 20 and 40 mg/kg bw groups there was a increase in
very slight to slight sporadic fibre degeneration in the sciatic
nerve. The changes were more frequent in the proximal than in the
distal part of the nerve. The NOEL was 4 mg/kg bw (Fokkema, 1994b).
c) Cypermethrin and alpha-cypermethrin
This experiment was performed in two phases. The first phase was
conducted to determine the time course for neurochemical changes in
Wistar rats occurring in the sciatic/posterior tibial nerve (SPTN),
trigeminal nerve and trigeminal ganglion following treatment with
cypermethrin for 5 days/week for 4 weeks. Cypermethrin was
administered at 150 mg/kg bw per day in DMSO (reduced to 100 mg/kg bw
per day in arachis oil, after 10 doses, because of mortality),
alpha-Cypermethrin was dosed at 37.5 mg/kg bw per day in DMSO (also
reduced after 10 days to 25 mg/kg bw per day in arachis oil). Five
animals per sex, treated with either cypermethrin and alpha-cyper-
methrin, were killed at 2, 3, 4, 5, 6, 8, 10 or 12 weeks and examined.
Dosing resulted in the death of 56% of the cypermethrin-treated
animals and 21% of the alpha-cypermethrin-treated animals. The most
frequent signs of intoxication included abnormal gait, ataxia,
lethargy, chromodacryorrhoea, salivation and hypersensitivity to
sensory stimuli. The ß-glucuronidase and ß-galactosidase activities in
the SPTN were increased at 5, 6 and 8 weeks, when compared to
controls. The increase was maximal after 5 weeks, and after 12 weeks
was comparable to controls. No significant enzyme changes were found
in the trigeminal ganglia and trigeminal nerve of treated animals.
Phase 2 was conducted to establish the dose level which did not
cause peripheral nerve degeneration in the SPTN, trigeminal nerve and
ganglia. Groups of 10 rats/sex were dosed with 37.5, 75 or 150 mg
cypermethrin/kg bw per day in DMSO or 10, 20 or 40 mg alpha-cyper-
methrin/kg bw per day in DMSO, 5 days/week for 4 weeks. A control
group of 10 animals was used.
Signs of intoxication similar to those reported in phase 1 were
seen at the highest dose levels. A large increase in ß-glucuronidase
and ß-galactosidase activities in the SPTN was seen at 150 mg/kg bw
per day cypermethrin and 40 mg/kg bw per day alpha-cypermethrin. In
the groups administered 75 mg/kg bw per day cypermethrin or 20 mg/kg
bw per day alpha-cypermethrin a small increase in ß-galactosidase was
found in both the distal and proximal sections of the SPTN. The
magnitude of the enzyme changes was similar to those of phase 1.
Significant enzyme changes were also found in the trigeminal ganglia
and to a lesser extent in the trigeminal nerve of the groups
administered 75 or 150 mg/kg bw per day cypermethrin and 20 or
40 mg/kg bw per day alpha-cypermethrin. The NOELs were 37.5 mg/kg bw
cypermethrin and 10 mg/kg bw alpha-cypermethrin (Rose, 1983).
2.2.7.2. Chickens
In a delayed neurotoxicity study, six adult domestic hens
received 1000 mg cypermethrin/kg bw per day in DMSO for 5 days. After
3 weeks the dosing regime was repeated and a further three weeks later
the birds were killed. A positive (tri- ortho-tolyl phosphate) and
negative control (not dosed) group were used.
None of the cypermethrin-treated hens developed any signs of
intoxication. Histological examination of the nervous system revealed
no lesions. All birds receiving the positive control developed
clinical signs of neurological damage within 15 days and became
progressively more unsteady and ataxic thereafter. Histological
examination of these animals showed lesions in the cerebellum, sciatic
nerve and spinal cord, including axonal and myelin degeneration
(Owen & Butterworth, 1977).
2.2.7.3 Hamsters
The 1979 JMPR (FAO, 1980) evaluated some neurotoxicity studies
with cypermethrin in hamsters. At doses of > 794 mg/kg bw, all
treated hamsters showed clinical signs of poisoning, including
tremors, abnormal irregular movements and an unusual gait. As in the
cases of rats, axon and myelin degeneration was noted in all groups
treated. The lesions included swelling and breaks in the axons and
clumping of myelin.
Hamsters treated orally with a single dose of 40 mg/kg bw,
followed by four doses of 20 mg/kg bw, developed weight loss and
sometimes mortality. There was loss of fur and dermal ulceration.
There was no effect in the mean slip angle experiment, and a marginal
increase in ß-galactosidase was observed in peripheral nerve.
Hamsters treated orally with doses of 5, 10 or 20 mg/kg bw per
day for 5 days showed no mortality. Lower body weight gain was
observed at 20 mg/kg bw per day. One female (out of 5) at the highest
dose developed hyperexcitability. There was a significant deficit in
the mean slip angle test, females showing an earlier dose-related
deficit than noted in males. ß-Galactosidase activity was increased at
all dose levels 3 weeks after the onset of the experiment. This effect
was significant at the two highest dose levels. In this experiment
dermal irritation and fur loss were noted.
In another experiment hamsters were orally treated with
30 mg/kg bw per day for 5 days. There was no mortality and there were
no differences in weight gain. There was some transient skin
irritation accompanied by skin ulceration. One male (out of 16) had an
unusual gait. There was a slight deficit in the inclined plane test
which was noted in the early parts of the experiment. Increases in
both ß-glucuronidase and ß-galactosidase were evident in peripheral
nerve tissue.
2.2.8 Special studies on biochemistry and electrophysiology
In three independent experiments with Wistar rats the effects of
varying doses of cypermethrin (purity 98%, 10% in DMSO) on the
trigeminal ganglion and three sections of the maxillary branch of the
trigeminal nerve (proximal, distal and endings) were determined.
Increases in ß-galactosidase activity in these tissues were taken as
evidence of axonal degeneration.
The three studies involved repeated oral administration of
cypermethrin at 150 mg/kg bw per day for 5 or 7 days and 0, 25, 50,
100 or 200 mg/kg bw per day for 5 or 7 days. Mortality occurred in
animals receiving 100 mg/kg bw per day or more. A dose-related
transient functional impairment, assessed by means of the inclined
plane test was found in the first week. Significant increases in
ß-glucuronidase and ß-galactosidase activity of the sciatic, tibial or
trigeminal nerves only occurred with 5 or 7 doses of 150 or
200 mg/kg bw per day. Increased activity of the enzymes in the distal
portion of nerves was found, but even in the most severely intoxicated
animals the magnitude of this increase was less than that induced by
the known neurotoxic agent methylmercury chloride (Dewar & Moffett,
1978).
Cypermethrin (1:1 cis:trans) was administered to male and female
rats at dose levels ranging from 25 to 200 mg/kg bw per day for 5
consecutive days by oral intubation as a 10% w/v solution in DMSO. A
dose-related functional deficit was observed when the mean slip angle
test and the landing foot spread test were applied to the animals. The
deficit was maximal from days 6 to 14 after the beginning of
treatment, and complete functional recovery occurred within 4 weeks.
Substantial variation in data from the landing foot spread test was
noted. Data were inconsistent over the course of the study.
ß-Glucuronidase activity was increased in a dose-dependent fashion in
both males and females. The results suggested that cypermethrin
produced a primary axonal degeneration, readily measurable 28 days
after treatment as an increase in ß-glucuronidase activity and in
deficits in specific behavioural-function testing of rats (FAO, 1980).
Electrophysiological studies were performed to determine whether
acute or subacute intoxication with cypermethrin produced changes in
the conduction velocity of slower fibres in peripheral nerves or
alterations in the maximal motor conduction velocity. There was no
evidence to suggest that cypermethrin, at doses that induced severe
clinical signs of intoxication, including ataxia, had any effect on
maximal motor conduction velocity or conduction velocity of the slower
motor fibres in peripheral nerves. Doses used in the study ranged from
a single dose of 200 mg/kg bw to 7 consecutive doses of 150 mg/kg bw
followed by two doses of 400 mg/kg bw. At near-lethal doses there were
no effects on conduction velocity even in the presence of clinical
signs of acute intoxication and at dose levels where previous studies
had shown functional degeneration. These electro-physiological
findings are reflective of motor function, which would suggest that
the physiological and functional deficits observed as a result of
acute intoxication are primarily sensory in nature (FAO, 1980).
2.2.9 Special studies on sensitization
2.2.9.1 Cypermethrin
Two out of 20 guinea-pigs developed a positive reaction to
cypermethrin in the Magnusson Kligman test, indicating that
cypermethrin is not a sensitizer (Coombs et al., 1976).
2.2.9.2 alpha-Cypermethrin
No positive reactions were obtained in a Magnusson-Kligman test
performed with guinea-pigs (Gardner, 1993).
2.2.10 Special studies on skin and eye irritation
2.2.10.1 Cypermethrin
A single application of undiluted technical cypermethrin was
moderately irritant to occluded rabbit skin (Coombs et al., 1976).
A single application of undiluted technical cypermethrin to
rabbit eyes produced a mild transient conjunctivitis and blepharism
lasting 2 days (Coombs et al., 1976).
2.2.10.2 alpha-Cypermethrin
Six NZW rabbits receiving a semi-occlusive topical application
with 500 mg alpha-cypermethrin technical developed very slight
erythema in two animals up to 72 hours after removal of the dressings.
There were no other dermal reactions (Gardner, 1993).
Six NZW rabbits receiving an instillation of 0.1 ml (equivalent
to 45 mg) alpha-cypermethrin technical developed slight conjunctival
redness and ocular discharge up to 72 hours after treatment. No cornea
or iris irritation was observed (Gardner, 1993).
2.3 Observations in humans
The symptoms and signs of acute poisoning resulting from
pyrethroids are very similar. Apart from the irritative symptoms of
the skin and respiratory tract (or digestive tract in ingestive
poisoning), acute pyrethroid poisoning is clinically characterized by
abnormalities of nervous excitability.
Occupationally exposed people had abnormal skin sensations
described as burning, itching or tingling, which could be exacerbated
by sweating or washing and readily disappeared after several hours.
Systemic symptoms included dizziness, headache, nausea, anorexia and
fatigue. Vomiting was more prominent in patients with ingestive
poisoning than in occupational poisoning cases. Other symptoms such as
chest tightness, paraesthesia, palpitation, blurred vision and
increased sweating were less frequently seen. The more serious cases
developed coarse muscular fasciculations in large muscles or
extremities (Van den Bercken & Vijverberg, 1989; He et al., 1989).
Urine obtained from operators spraying cypermethrin in
experimental trials was analysed for the presence of the chlorinated
cyclopropane carboxylic acid metabolite. This metabolite was observed
in the urine of exposed workers at levels up to 0.4 µg/ml (the limit
of detection was estimated to be 0.05 µg/ml).
Cypermethrin sprayers were found to have residues on the exposed
parts of their bodies. The rate of dermal exposure of the operators
during spraying ranged from 1.5 to 46.1 mg/hour. There was a
reasonable relationship between the total cypermethrin deposited
dermally and the excretion in urine. The levels of the cyclopropane
carboxylic acid metabolite in the 24-hour urine were between <0.05
and 0.32 mg (not specified). This, together with the finding of 0.6 mg
(not specified) of this metabolite in 72-hour urine from one man, led
to the estimation that approximately 3% of the total dermal dose was
absorbed and rapidly excreted by the operators (FAO, 1980).
In a controlled experiment with sprayers, no abnormalities were
found in clinical and neurological examinations, blood chemistry or
peripheral nerve function tests (including the trigeminal nerve). In
some electroneurophysiological tests (motor conduction velocity, slow
fibre conduction velocity and cornea reflex), a significant change
within the normal range appeared to exist for the group sprayers
between pre- and post-exposure measurements. These changes probably
reflect seasonal variations (FAO, 1982).
3. COMMENTS
The Committee considered toxicological data on cypermethrin and
alpha-cypermethrin, including the results of acute, short-term, and
reproductive studies, and studies on pharmacokinetics and metabolism,
genotoxicity, long-term toxicity/carcinogenicity and neurotoxicity.
Results of effects in humans were also considered.
Cypermethrin is a mixture of four cis and four trans isomers. The
cis isomers are more biologically active and more persistent than the
trans isomers, alpha-Cypermethrin is a mixture of the two most active
cis isomers. A typical cypermethrin sample contains 25% alpha-
cypermethrin. Cypermethrin and alpha-cypermethrin are alpha-cyano or
type II pyrethroids that cause neurotoxicity in mammals and insects.
They affect nerve membrane sodium channels, causing a long-lasting
prolongation of the normally transient increase in sodium permeability
of the membrane during excitation. At high dose levels, these type II
pyrethroids induce salivation and tremors that progress to
characteristic clonic-tonic convulsions (choreoathetosis and
salivation syndrome).
After oral administration, cypermethrin is readily absorbed,
distributed and excreted in rats, chickens, sheep and cattle.
Cypermethrin is primarily eliminated in urine and faeces in about
equal proportions. Less than 1% is excreted in milk. When cypermethrin
was applied dermally to sheep, 2.5% was eliminated in urine and faeces
within 6 days and after an oral dose about 60% was eliminated within 2
days.
Studies in cattle indicated that absorption, distribution and
excretion were comparable for cypermethrin and alpha-cypermethrin. The
major metabolic route for both cypermethrin and its isomers, including
alpha-cypermethrin, is cleavage of the ester bond followed by
hydroxylation and conjugation of the cyclopropyl and phenoxybenzyl
portions of the molecule. The data suggest that there is no isomeric
interconversion during metabolism.
The acute oral toxicity of cypermethrin and alpha-cypermethrin is
moderate to high. WHO has classified these substances as "moderately
hazardous" (WHO, 1996). In rats and mice, the oral LD50 ranges from
82 to 4000 mg/kg bw for cypermethrin and from 35 to > 5000 mg/kg bw
for alpha-cypermethrin, depending on the vehicle used. At lethal or
near lethal doses the signs are typical of type-II pyrethroids and
include salivation, ataxia, gait abnormalities and convulsions.
Several oral short-term toxicity studies with cypermethrin were
available. Cypermethrin has been tested in rats (5 weeks and 90 days)
and dogs (5 weeks and 13 weeks) at dose levels ranging from 25 to
1600 mg/kg feed (equivalent to 1.25-80 mg/kg bw per day) and 5 to
1500 mg/kg feed (equivalent to 0.125-37.5 mg/kg bw per day),
respectively. In these studies, the clinical signs included ataxia,
abnormal gait, nervousness, and, particularly in dogs, inappetence,
diarrhoea, vomiting and hyperaesthesia. In both rats and dogs,
cypermethrin caused decreases in body weight gain, food intake, and a
number of haematological parameters, increases in some organ weights
and plasma urea levels, and, at lethal or near-lethal doses, effects
on the nervous system. For cypermethrin the lowest NOEL in short-term
studies was in a 90-day study with rats administered 25, 100, 400 or
1600 mg/kg feed (equivalent to 1.25-80 mg/kg bw per day). Male rats
given 1600 mg/kg feed showed decreases in haemoglobin concentration,
mean corpuscular volume and eosinophil numbers, and increases in
prothrombin time, plasma urea levels and relative liver and kidney
weights. The decrease in eosinophil numbers and increase in relative
liver weight were also observed in males at 400 mg/kg feed. In female
rats, reduced food intake and increased relative liver weight were
noted in rats given 1600 mg/kg feed. The NOEL in this study was
100 mg/kg feed, equivalent to 5 mg/kg bw per day.
alpha-Cypermethrin was tested in oral short-term toxicity studies
with mice (29 days and 13 weeks), rats (5 weeks, 6 weeks and 90 days)
and dogs (13 weeks and 52 weeks) at dose levels ranging from 50 to
1600 mg/kg feed (equivalent to 7-240 mg/kg bw per day), 20 to
1200 mg/kg feed (equivalent to 1.25-60 mg/kg bw per day) and 30 to
270 mg/kg feed (equivalent to 0.75-6.75 mg/kg bw per day),
respectively. In these studies, alpha-cypermethrin caused the same
effects as described for cypermethrin in the short-term studies. The
signs of toxicity included ataxia, abnormal gait, increased
sensitivity to noise, hyperactivity, hunched posture and, as
demonstrated histologically, axonal degeneration of the sciatic
nerves. For alpha-cypermethrin the lowest NOEL was in a 52-week study
with dogs. In this study a diet containing 60, 120 or 240 mg/kg feed
(equivalent to 1.5 to 6 mg/kg bw per day) was administered. Dogs given
120 and 240 mg/kg feed showed skin reddening, ulceration and necrosis.
The NOEL in this study was 1.5 mg/kg bw per day.
A three-generation reproductive toxicity study with cypermethrin
was performed in rats at dose levels of 10, 100 or 500 mg/kg feed
(equivalent to 0.5-25 mg/kg bw per day). At the highest dose a
reduction in body weight gain and food consumption and a concomitant
reduction in litter size and weight were seen in the F1a progeny
only. No other effects on fertility or reproduction parameters were
observed. The NOEL was 100 mg/kg feed, equivalent to 5 mg/kg bw per
day.
Cypermethrin did not cause embryotoxicity or teratogenicity in
rats at doses up to 70 mg/kg bw per day or in rabbits at doses up to
120 mg/kg bw per day. alpha-Cypermethrin did not cause embryotoxicity
or teratogenicity in rats at doses up to 9 mg/kg bw per day or in
rabbits up to 30 mg/kg bw per day. The NOELs for maternal toxicity in
rats were 17.5 and 9 mg/kg bw per day for cypermethrin and
alpha-cypermethrin, respectively, while the NOELs for maternal
toxicity in rabbits were 30 and 3 mg/kg bw per day for cypermethrin
and alpha-cypermethrin, respectively.
Cypermethrin and alpha-cypermethrin have been tested in a wide
variety of in vitro and in vivo genotoxicity studies. All of the
results were negative.
Two long-term toxicity/carcinogenicity studies with mice and rats
were available on cypermethrin. Mice received a diet containing 100,
400 or 1600 mg/kg feed (equal to 14-228 mg/kg bw per day) for 101
weeks. At 1600 mg/kg feed reduced body weight gain, changes in
haematological parameters and increased liver weight were observed. No
effects were observed at 400 mg/kg feed, equal to 57 mg/kg bw per day.
In a study in which rats received diets containing 1, 10, 100 or
1000 mg/kg feed (equivalent to 0.05-50 mg/kg bw per day) for 2 years,
the only effects observed were reductions in body weight and food
consumption at 1000 mg/kg feed. The NOEL was 100 mg/kg feed,
equivalent to 5 mg/kg bw per day.
In a two-year toxicity study, dogs received diets containing
3, 30, 300 or 1000 mg cypermethrin/kg feed (equivalent to
0.075-25 mg/kg bw per day). The dose level of 1000 mg/kg feed was
reduced to 600 mg/kg feed owing to severe intoxication. At 300 mg/kg
feed, equivalent to 7.5 mg/kg bw per day, no effects were seen. The
Committee concluded that cypermethrin was not carcinogenic in these
studies.
Long-term toxicity/carcinogenicity or reproductive toxicity
studies were not available on alpha-cypermethrin. The Committee noted
the absence of reproductive toxicity and carcinogenicity associated
with administration of cypermethrin, which contains 25% alpha-
cypermethrin. The Committee also noted the absence of genotoxicity
for either cypermethrin or alpha-cypermethrin, the absence of
carcinogenicity associated with compounds of similar structure and
the similar metabolism and disposition of the two compounds. In view
of the foregoing, the Committee concluded that it was unnecessary to
request the results of long-term toxicity/carcinogenicity or
reproductive toxicity studies on alpha-cypermethrin.
Several studies on the neurotoxicity of cypermethrin and
alpha-cypermethrin in rats were available. In these studies high oral
doses of cypermethrin and alpha-cypermethrin caused clinical signs
that included coarse tremor and spasmodic body and tail movements.
Evidence of axonal damage in the sciatic/posterior tibial nerves and
the trigeminal nerve and ganglion was indicated by significant
increases in ß-glucuronidase and ß-galactosidase in nerve tissue
homogenates, in addition to abnormal neuromuscular function tests. In
the inclined plane test, cypermethrin (in DMSO) caused transient
functional impairment. The lowest NOEL for neurotoxicity was
37.5 mg/kg bw per day for cypermethrin (in DMSO) and 4 mg/kg bw per
day for alpha-cypermethrin (in corn oil), indicating that the toxicity
may be influenced by the vehicle used.
Humans occupationally exposed to cypermethrin developed skin
sensation as a first reaction, followed by systemic effects such as
dizziness, headache, nausea, paraesthesia and increased sweating. In
more serious cases, muscular fasciculations developed in large muscles
or in the extremities. In experiments with operators spraying
cypermethrin, no clinical nervous system abnormalities were observed.
However, exposure levels were not measured.
4. EVALUATION
The Committee established an ADI of 0-50 µg/kg bw for
cypermethrin on the basis of the NOEL of 5 mg/kg bw per day in 90-day,
2-year and reproductive toxicity studies in rats and the application
of a safety factor of 100. The Committee established an ADI of
0-20 µg/kg bw for alpha-cypermethrin on the basis of the NOEL of
1.5 mg/kg bw per day in a 52-week study in dogs and the application of
a safety factor of 100.
5. REFERENCES
Amyes, S.J., Holmes, P., Irving, E.M., Green, C.F., Virgo, D.M., &
Sparrow, S. (1994). Alpha-cypermethrin: preliminary toxicity study by
dietary administration to CD-1 mice for 13 weeks. Unpublished report
No. 92/SHL009/0849 from Pharmaco-LSR Ltd, Suffolk, England. Submitted
to the WHO by Cyanamid, Wayne, NJ. USA.
Brooks, T.M. (1980). Toxicity studies with agricultural chemicals:
Mutagenicity studies with RIPCORD in microorganisms in vitro and in
the host-mediated assay (Experiment Nos 1846 and 1847). Unpublished
report TLGR.80.059 from Shell Toxicology Laboratory (Tunstall).
Submitted to the WHO by Cyanamid, Wayne, NJ, USA.
Brooks, T.M. (1984). Genotoxicity studies with Fastac: the induction
of gene mutation in the yeast Saccharomyces cerevisiae. Unpublished
report No. SBGR.84.117 from Shell Research Limited, Sittingbourne
Research Centre. Submitted to the WHO by Cyanamid, Wayne, NJ, USA.
Brooks, T.M. & Wiggins, D.E. (1992). FASTAC TM: Bacterial mutagenicity
studies. Unpublished report No. SBTR.92.022 from Shell Research
Limited, Sittingbourne Research Centre. Submitted to the WHO by
Cyanamid, Wayne, NJ, USA.
Brooks, T.M. & Wiggins, D.E. (1993). FASTAC TM: In vitro chromosome
studies using cultured human lymphocytes. Unpublished report
No. SBTR.93.007 from Shell Research Limited, Sittingbourne Research
Centre. Submitted to the WHO by Cyanamid, Wayne, NJ, USA.
Buckwell, A.C. (1981). A 2-year feeding study in dogs on WL 43467
(Experiment No. 1412). Unpublished report No. SBGR.81.126 (plus two
addenda) from Shell Toxicology Laboratory (Tunstall). Submitted to the
WHO by Cyanamid, Wayne, NJ, USA.
Buckwell, A.C. & Butterworth, S. (1977). Toxicology studies on the
pyrethroid insecticide WL 43467: a 13-week feeding study in dogs
(Experiment No. 1112). Unpublished report No. TLGR 0127.77 (including
addendum and corrigendum) from Shell Toxicology Laboratory (Tunstall).
Submitted to the WHO by Cyanamid, Wayne, NJ, USA.
Carter, B.I. & Butterworth, S.T.G. (1976). Toxicity of insecticides:
The acute oral toxicity and neuropathological effects of WL 43467 to
rats. Unpublished report No. TLGR.0055.76 from Shell Research,
Sittingbourne Research Centre. Submitted to the WHO by Cyanamid,
Wayne, NJ, USA.
Clare, M.G. & Wiggins, D.E. (1984). Genotoxicity studies with fastac:
in vivo cytogenetic test using rat bone marrow. Unpublished report
No. SBGR.84.120 from Shell Research Limited, Sittingbourne Research
Centre. Submitted to the WHO by Cyanamid, Wayne, NJ, USA.
Clark, D.G. (1982). WL85871: A 90-day feeding study in rats.
Unpublished report No. SBGR.81.293 from Shell Research Limited,
Sittingbourne Research Centre. Submitted to the WHO by Cyanamid,
Wayne, NJ, USA.
Coombs, A.D., Carter, B.I., Hend, R.W., Butterworth, S.G., & Buckwell,
A.C. (1976) Toxicity studies on the insecticide WL 43467: Summary of
results of preliminary experiments. Unpublished report
No. TLGR.0104.76 from Shell Toxicology Laboratory (Tunstall).
Submitted to the WHO by Cyanamid, Wayne, NJ, USA.
Crawford, M.J. (1978). The excretion and residues of radioactivity in
cows treated orally with 14C-labelled WL 43467. Unpublished report
No. TLGR.0029.78 from Shell Toxicology Laboratory (Tunstall).
Submitted to the WHO by Cyanamid, Wayne, NJ, USA.
Crawford, M.J. & Hutson, D.H. (1977). The elimination and retention of
WL 43467 when administered dermally or orally to sheep. Unpublished
report No. TLGR.0098.77 from Shell Toxicology Laboratory (Tunstall).
Submitted to the WHO by Cyanamid, Wayne, NJ, USA.
Crawford, M.J. & Hutson, D.H. (1978). The elimination of residues from
the fat of rats following the oral administration of [14C-benzyl]
WL 43381 (cis-WL 43467). Unpublished report No. TLGR.0078.78 from
Shell Toxicology Laboratory (Tunstall). Submitted to the WHO by
Cyanamid, Wayne, NJ, USA.
Creedy, C.L. & Wooder, M.F. (1977) Studies on the effect of WL 43467
upon the integrity of rat liver cell DNA in vivo. Unpublished report
No. TLGR.0043.77 from Shell Toxicology Laboratory (Tunstall).
Submitted to the WHO by Cyanamid, Wayne, NJ, USA.
Croucher, A., Stoydin, G., Cheeseman, M.E., Buckwell, A.C., &
Gellatly, J.B.M. (1980). The metabolic fate of cypermethrin in the
cow: elimination and residues derived from 14C-benzyl label.
Unpublished report No. TLGR.80.121 from Shell Toxicology Laboratory
(Tunstall). Submitted to the WHO by Cyanamid, Wayne, NJ, USA.
Dean, B.J. (1977). Toxicity studies with WL 43467: Chromosome studies
on bone marrow cells of Chinese hamsters after two daily oral doses of
WL 43467. Unpublished report No. TLGR.0136.77 from Shell Toxicology
Laboratory (Tunstall). Submitted to the WHO by Cyanamid,
Wayne, NJ, USA.
Dean, B.J. (1981). Toxicity studies with agricultural chemicals:
In vitro genotoxicity studies with RIPCORD (Experiment
No. AIR-1820). Unpublished report No. TLGR.80.116 from Shell
Toxicology Laboratory (Tunstall). Submitted to the WHO by Cyanamid,
Wayne, NJ, USA.
Dean, I. & Jackson, F. (1995). WL85871: 52-week oral (dietary)
toxicity study in dogs (IRI Project No. 652238). Unpublished report
No. IRI/11110 from Inveresk Research International, Scotland.
Submitted to the WHO by Cyanamid, Wayne, NJ, USA.
Dean, B.J., Van der Pauw, C.L., & Butterworth S.T.G. (1977). Toxicity
studies with WL 43467: Dominant lethal assay in male mice after single
oral doses of WL 43467. Unpublished report No. TLGR.0042.77 from Shell
Toxicology Laboratory (Tunstall). Submitted to the WHO by Cyanamid,
Wayne, NJ, USA.
Dewar, A.J. & Moffett, B.J. (1978). Toxicity studies on the
insecticide WL 43467: Biochemical studies on the effect of WL 43467 on
the rat trigeminal nerve and ganglion. Unpublished report No.
TLGR.0162.77 from Shell Toxicology Laboratory (Tunstall). Submitted to
the WHO by Cyanamid, Wayne, NJ, USA.
Dunsire, J.P. & Gifford, L.J. (1993). The distribution of
[benzyl-14C]WL85871 (FASTAC) in the lactating cow (nature of residue
study to EPA guidelines - Live phase) (IRI Project No. 153667).
Unpublished report No. IRI/9879 from Inveresk Research International,
Scotland. Submitted to the WHO by Cyanamid, Wayne, NJ, USA.
FAO (1980). Pesticide residues in food-1979. Report of the Joint
Meeting of the FAO Panel of Experts on Pesticide Residues in Food and
the Environment and the WHO Expert Group on Pesticide Residues. Rome,
Food and Agriculture Organization of the United Nations (FAO Plant
Production and Protection Paper, No. 20).
FAO (1982). Pesticide residues in food-1981. Report of the Joint
Meeting of the FAO Panel of Experts on Pesticide Residues in Food and
the Environment and the WHO Expert Group on Pesticide Residues. Rome,
Food and Agriculture Organization of the United Nations (FAO Plant
Production and Protection Paper, No. 37).
Fish, A. (1979). Corrigendum I in Group Research Report
No. TLGR.0188.78. Unpublished data from Shell Toxicology Laboratory
(Tunstall). Submitted to the WHO by Cyanamid, Wayne, NJ, USA.
Fokkema, G.N. (1994a). WL85871 (FASTAC): A 6-week range finding
feeding study in the rat. Unpublished report No. SBTR.93.002 from
Shell Research Limited, Sittingbourne Research Centre. Submitted to
the WHO by Cyanamid, Wayne, NJ, USA.
Fokkema, G.N. (1994b). WL85871 (FASTAC): An acute oral (gavage)
neurotoxicity study in the rat. Unpublished report No. SBTR.92.027
from Shell Research Limited, Sittingbourne Research Centre. Submitted
to the WHO by Cyanamid, Wayne, NJ, USA.
Gardner, J.R. (1993). FASTAC technical: Acute oral and dermal toxicity
in rat, skin and eye irritancy in rabbit and skin sensitisation
potential in guinea pig. Unpublished report No. SBTR.92.033 from Shell
Research Limited, Sittingbourne Research Centre. Submitted to the WHO
by Cyanamid, Wayne, NJ, USA.
Green, C.F. (1993). Alphacypermethrin: Preliminary toxicity study by
dietary administration to CD-1 mice for four weeks. Unpublished report
No. LSR 92/0346 from Life Science Research Limited. Submitted to the
WHO by Cyanamid, Wayne, NJ, USA.
Greenough, R.J. & Goburdhun, R. (1984). WL 85871: Oral (dietary)
maximum tolerated dose study in dogs (IRI Project No. 631087).
Unpublished report No. IRI/3107 from Inveresk Research International,
Scotland. Submitted to the WHO by Cyanamid, Wayne, NJ, USA.
Greenough, R.J., Cockrill, J.B., & Goburdhun, R. (1984).
WL 85871: 13-week oral (dietary) toxicity study in dogs (IRI Project
No. 631092). Unpublished report No. IRI/3197 from Inveresk Research
International, Scotland. Submitted to the WHO by Cyanamid,
Wayne, NJ, USA.
He, F., Wang, S., Liu, L., Chen, S., Zhang, Z. & Sun, J. (1989).
Clinical manifestations and diagnosis of acute pyrethroid poisoning.
Arch. Toxicol., 63, 54-58.
Hend, R.W., Hendy, R. & Fleming, D.J. (1978). Toxicity studies on the
insecticide WL 43367: a three generation reproduction study in rats.
Unpublished report No. TLGR.0188.78 from Shell Toxicology Laboratory
(Tunstall). Submitted to the WHO by Cyanamid, Wayne, NJ, USA.
Henderson, C. & Parkinson, G.R. (1981). Cypermethrin technical:
subacute dermal toxicity study in rabbits (CTL Study No. LB0019).
Unpublished report No. CTL/P/588 from ICI Central Toxicology
Laboratory. Submitted to the WHO by Cyanamid, Wayne, NJ, USA.
Hijzen, T.H., de Beun, R., & Slanger, J.L. (1988). Effects of
pyrethroids on the acoustic startle reflex in the rat. Toxicology,
49, 271-276.
Hutson, D.H. & Stoydin, G. (1976). The excretion of radioactivity from
cows fed with radioactively labelled WL 43467. Unpublished report
No. TLGR.0075.76 from Shell Toxicology Laboratory (Tunstall).
Submitted to the WHO by Cyanamid, Wayne, NJ, USA.
Hutson, D.H. & Stoydin, G. (1987). Excretion and residues of the
pyrethroid insecticide cypermethrin in laying hens. Pestic. Sci.,
18, 157-168.
IPCS (1996) The WHO recommended classification of pesticides by hazard
and guidelines to classification 1996-1997. World Health Organization,
Geneva, p. 23 (Document WHO/PCS/96.3).
Irvine, L.F.H (1994a). Oral (gavage) rabbit developmental toxicity
dose ranging study. Unpublished report No. SLN/3/92 from Toxicol
Laboratories Limited, Ledbury, England. Submitted to the WHO by
Cyanamid, Wayne, NJ, USA.
Irvine, L.F.H. (1994b). Oral (gavage) rabbit developmental toxicity
(teratogenicity) study. Unpublished report No. SLN/4/93 from Toxicol
Laboratories Limited, Ledbury, England. Submitted to the WHO by
Cyanamid, Wayne, NJ, USA.
Irvine, L.H.F. (1994c). Alphacypermethrin. Oral (gavage) rat
developmental toxicity (teratogenicity) study. Unpublished report
No. SLN/2/92 from Toxicol Laboratories Limited, Ledbury, England.
Submitted to the WHO by Cyanamid, Wayne, NJ, USA.
Irvine, L.F.H & Twomey, K. (1994). Alphacypermethrin. Oral (gavage)
rat developmental study. Unpublished report No. SLN/1/92 from Toxicol
Laboratories Limited, Ledbury, England. Submitted to the WHO by
Cyanamid, Wayne, NJ, USA.
Jackson, G.C. (1993). Alphacypermethrin: Acute inhalation toxicity in
rats, 4-hour exposure. Unpublished report No. SLL 266/930770 from
Huntingdon Research Centre, England. Submitted to the WHO by Cyanamid,
Wayne, NJ, USA.
Lindsay, S., Banham, P.B., Chart, I.S., Chalmers, D.T., Godley, M.J.,
& Taylor, K. (1982). Cypermethrin: Lifetime feeding study in mice
(CTL Study No. PM0366). Unpublished report No. CTL/P/687 (including 1
supplement) from ICI Central Toxicology Laboratory. Submitted to the
WHO by Cyanamid, Wayne, NJ, USA.
McAusland, H.E., Butterworth, S.T.G., & Hunt, P.F. (1978). Toxicity
studies on the insecticide WL 43467: A two-year feeding study in rats.
Unpublished report No. TLGR.0189.78 (including 4 corrigenda/addenda)
from Shell Toxicology Laboratory (Tunstall). Submitted to the WHO by
Cyanamid, Wayne, NJ, USA.
Morrison, B.J. & Richardson, K.A. (1994). WL85871 (alphacypermethrin,
FASTAC): The metabolism of 14C-WL85871 after repeated oral dosing in
the lactating cow; in-life phase and metabolite profiling (Experiment
No. 6046). Unpublished report No. SBTR.93.063 from Sittingbourne
Research Centre, Sittingbourne, England. Submitted to the WHO by
Cyanamid, Wayne, NJ, USA.
Owen, D.E. & Butterworth, S.T.G. (1977). Toxicity of pyrethroid
insecticides: Investigation of the neurotoxic potential of WL 43467 to
adult domestic hens. Unpublished report No. TLGR.0134.77 from Shell
Toxicology Laboratory (Tunstall). Submitted to the WHO by Cyanamid,
Wayne, NJ, USA.
Pickering, R.G. (1981). A 90-day feeding study of WL 43467 in rats
(Experiment No. 1806). Unpublished report No. TLGR.80.143 from Shell
Toxicology Laboratory (Tunstall). Submitted to the WHO by Cyanamid,
Wayne, NJ, USA.
Potter, D. & McAusland, H.E. (1980). Toxicity studies on the
insecticide WL43467: A study of liver microsomal enzyme activity in
rats fed WL 43467 for 2 years (Experiment No. 1105). Unpublished
report from Shell Toxicology Laboratory (Tunstall). Submitted to the
WHO by Cyanamid, Wayne, NJ, USA.
Price, J.B. (1984). Toxicology of pyrethroids: the acute oral and
percutaneous toxicity of Ripcord (WL-43467). Technical concentrate
(Project No. SRCAIR84). Unpublished report No. SBGR.84.299 from Shell
Research Limited, Sittingbourne Research Centre. Submitted to the WHO
by Cyanamid, Wayne, NJ, USA.
Rose, G.P. (1982a). Toxicology of pyrethroids: the acute oral and
percutaneous toxicity of WL 85871 (cis-2-Ripcord) in comparison with
Ripcord. Unpublished report No. SBGR.82.130 from Shell Research
Limited, Sittingbourne Research Centre. Submitted to the WHO by
Cyanamid, Wayne, NJ, USA.
Rose, G.P. (1982b). Toxicology of pyrethroids: the acute oral and
percutaneous toxicity of WL 85871 (cis-2-RIPCORD) comparison with
RIPCORD. Unpublished report No. SBGR.82.130 from Shell Research
Limited, Sittingbourne Research Centre. Submitted to the WHO by
Cyanamid, Wayne, NJ, USA.
Rose, G.P. (1983). Neurotoxicity of WL85871. Comparison with WL43467:
The effect of twenty oral doses of WL85871 or WL43467 over a period of
4 weeks on the rat sciatic/posterior tibia nerve, trigeminal nerve and
trigeminal ganglion. Unpublished report No. SBGR.83.185 from Shell
Research Limited, Sittingbourne Research Centre. Submitted to the WHO
by Cyanamid, Wayne, NJ, USA.
Rose, G.P. & Dewar, A.J. (1983). Intoxication with four synthetic
pyrethroids fails to show any correlation between neuromuscular
dysfunction and neurobiochemical abnormalities in rats.
Arch. Toxicol., 53, 297-316.
Tesh, J.M., Tesh, S.A., & Davies, W. (1978). WL 43467: Effects upon
the progress and outcome of pregnancy in the rat. Unpublished report
of Life Science Research. Submitted to the WHO by Cyanamid,
Wayne, NJ, USA.
Tesh, J.M., Ross, F.W., & Wightman, T.J. (1984a). WL 43467: Effects of
oral administration upon pregnancy in the rabbit. 1. Dosage range
finding study. Unpublished test report No. 84/SHL003/014 from of Life
Science Research. Submitted to the WHO by Cyanamid, Wayne, NJ, USA.
Tesh, J.M., Ross, F.W., & Wightman, T.J. (1984b). WL 43467: Effects of
oral administration upon pregnancy in the rabbit. 2. Main study.
Unpublished test report No. 84/SHL004/043 from Life Science Research.
Submitted to the WHO by Cyanamid, Wayne, NJ, USA.
Tesh, J.M., Ross, F.W., & Wightman, T.J. (1988). Supplementary report
to LSR report No. 84/SHL004/043. Unpublished report No. 88/SHL004/775
from Life Science Research. Submitted to the WHO by Cyanamid,
Wayne, NJ, USA.
Thorpe, E. (1982). A 5-week feeding study with WL 85871 in rats
(Experiment No. 2095). Unpublished report No. SBGR.81.212 from Shell
Research Limited, Sittingbourne Research Centre. Submitted to the WHO
by Cyanamid, Wayne, NJ, USA.
Thorpe, E. (1985). Fourth corrigendum/addendum to TLGR.0188.78.
Unpublished report from Shell Research Limited, Sittingbourne Research
Centre (Four volumes). Submitted to the WHO by Cyanamid,
Wayne, NJ, USA.
Van der Bercken, J. & Vijverberg, H.P.M. (1989). Neurotoxicological
effects of pyrethroid insecticides. Unpublished report from the
Department of Veterinary Pharmacology, Pharmacy and Toxicology.
University of Utrecht, The Netherlands. Submitted to the WHO by
Cyanamid, Wayne, NJ, USA.
Vanderwaart, E.J. (1994). Evaluation of the mutagenic activity of
Fastac technical in a in vitro mammalian cell gene mutation test
with L5178Y mouse lymphoma cells (with independent repeat).
Unpublished report No. 087367 from Notox B.V., The Netherlands.
Submitted to the WHO by Cyanamid, Wayne, NJ, USA.
Vanderwaart, E.J. (1995). Micronucleus test in bone marrow cells of
the mouse with Fastac technical. Unpublished report No. 087378 from
Notox B.V., The Netherlands. Submitted to the WHO by Cyanamid,
Wayne, NJ, USA.
WHO (1996). The WHO recommended classification of pesticides by hazard
and guidelines to classification 1996-1997 (WHO/PCS/96.3). Available
from the International Programme on Chemical Safety, World Health
Organization, Geneva, Switzerland.
Wooder, M.F. (1981). Studies on the effect of WL 85871 on the
integrity of rat liver DNO in vivo (Experiment No. 2103).
Unpublished report No. SBGR.81.225 from Shell Toxicology Laboratory
(Tunstall). Submitted to the WHO by Cyanamid, Wayne, NJ, USA.
