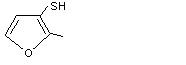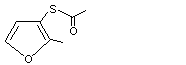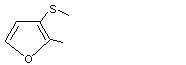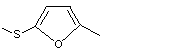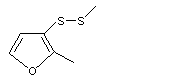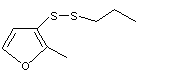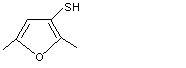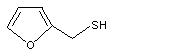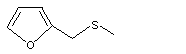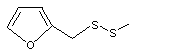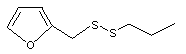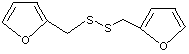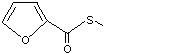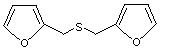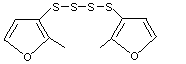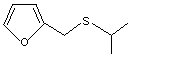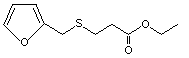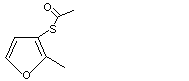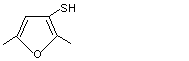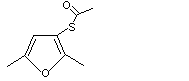WHO FOOD ADDITIVES SERIES: 50
Sulfur-substituted Furan Derivatives
First draft prepared by
Dr A. Mattia
Division of Petition Review, Office of Food Additive Safety, Center for Food Safety and Applied Nutrition, Food and Drug Administration, College Park, Maryland, USA
and Professor A.G. Renwick
Clinical Pharmacology Group, University of Southampton, Southampton, England
1. EVALUATION
1.1 Introduction
The Committee evaluated a group of 33 flavouring agents that includes thiofuran and thiofurfuryl derivatives (see Table 1), using the Procedure for the Safety Evaluation of Flavouring Agents (see Figure 1). The Committee has not previously evaluated any member of the group. Eighteen of the 33 flavouring agents in this group have been reported to occur naturally in foods (Stofberg & Grundschober, 1987; Maarse et al., 1999). Thiofurfuryl and thiofuran derivatives have been detected mainly in coffee and cooked meats.
Table 1. Summary of results of safety evaluations of sulfur-substituted furan derivatives used as flavouring agentsa
|
Flavouring agent |
No. |
CAS No. and structure |
Step B3a,b
Does intake
exceed threshold for human intake? |
Step B4
Adequate margin of safety for the flavouring agent or related chemical? |
Conclusion based on current intake |
|
Structural class II |
|
|
|
|
|
|
2-Methyl-3-furanthiol |
1060 |
28588-74-1
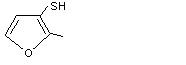 |
No
Europe: 0.6
USA: 0.9 |
Yes. The NOEL of 5 mg/kg bw per day is > 100 000 times the estimated intake of 2-methyl-3-furanthiol when used as a flavouring agent. |
No safety concern |
|
Ethanoic acid, s-(2-methyl-3-furanyl) ester |
1069 |
55764-25-5
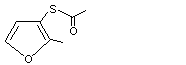 |
No
Europe: 0.01
USA: 0.07 |
Yes. The NOEL of 5 mg/kg bw per day for the related substance No. 1060 is > 1 million times the estimated intake of S-ethanoic acid when used as a flavouring agent. |
No safety concern |
|
2-Methyl-3-(methylthio)furan |
1061 |
63012-97-5
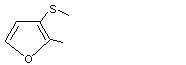 |
No
Europe: 1
USA: 0.1 |
Yes. The NOEL of 1.3 mg/kg bw per day for the related substance No. 1077 is > 10 000 times the estimated intake of 2-methyl-3-(methylthio)furan when used as a flavouring agent. |
No safety concern |
|
2-Methyl-5-(methylthio)furan |
1062 |
13678-59-6
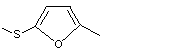 |
No
Europe: 1
USA: 0.02 |
Yes. The NOEL of 1.3 mg/kg bw per day for the related substance No. 1077 is > 10 000 times the estimated intake of 2-methyl-5-(methylthio)furan when used as a flavouring agent. |
No safety concern |
|
Methyl-2-methyl-3-furyl disulfide |
1064 |
65505-17-1
 |
No
Europe: 0.9
USA: 0.05 |
Yes. The NOEL of 1.2 mg/kg bw per day is > 10 000 times the estimated intake of methyl-2-methyl-3-furyl disulfide when used as a flavouring agent. |
No safety concern |
|
Propyl-2-methyl-3-furyl disulfide |
1065 |
61197-09-9
 |
No
Europe: ND
USA: 0.7 |
Yes. The NOEL of 1.2 mg/kg bw per day for the related substance No. 1064 is > 100 000 times the estimated intake of propyl-2-methyl-3-furyl disulfide when used as a flavouring agent. |
No safety concern |
|
Bis(2-methyl-3-furyl) disulfide |
1066 |
28588-75-2
 |
No
Europe: 0.3
USA: 0.7 |
Yes. The NOEL of 0.45 mg/kg bw per day is > 10 000 times the estimated intake of bis(2-methyl-3-furyl) disulfide when used as a flavouring agent. |
No safety concern |
|
2,5-Dimethyl-3-furanthiol |
1063 |
55764-23-3
 |
No
Europe: 0.03
USA: 0.7 |
Yes. The NOEL of 5 mg/kg bw per day for the related substance No. 1060 is > 100 000 times the estimated intake of 2,5-dimethyl-3-furanthiol when used as a flavouring agent. |
No safety concern |
|
Furfuryl mercaptan |
1072 |
98-02-2
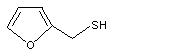 |
No
Europe: 40
USA: 11 |
Yes. The NOEL of 3 mg/kg bw per day is > 1000 times the estimated intake of furfuryl mercaptan when used as a flavouring agent. |
No safety concern |
|
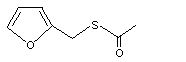
Furfuryl thioacetate |
1074 |
13678-68-7
 |
No
Europe: 0.5
USA: 0.05 |
Yes. The NOEL of 0.83 mg/kg bw per day for the related substance No. 1089 is > 100 000 times the estimated intake of furfuryl thioacetate when used as a flavouring agent. |
No safety concern |
|
Furfuryl methyl sulfide |
1076 |
1438-91-1
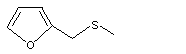 |
No
Europe: 1
USA: 0.1 |
Yes. The NOEL of 1.3 mg/kg bw per day for the related substance No. 1077 is > 10 000 times the estimated intake of furfuryl methyl sulfide when used as a flavouring agent. |
No safety concern |
|
Methyl furfuryl disulfide |
1078 |
57500-00-2
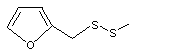 |
No
Europe: 1
USA: 0.04 |
Yes. The NOEL of 1.2 mg/kg bw per day for the related substance No. 1064 is > 10 000 times the estimated intake of methyl furfuryl disulfide when used as a flavouring agent. |
No safety concern |
|
Furfuryl propyl disulfide |
1079 |
2527366-36-0
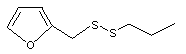 |
No
Europe: ND
USA: 3 |
Yes. The NOEL of 1.2 mg/kg bw per day for the related substance No. 1064 is > 10 000 times the estimated intake of furfuryl propyl disulfide when used as a flavouring agent. |
No safety concern |
|
2,2'-(Dithiodimethylene) difuran |
1081 |
4437-20-1
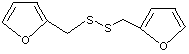 |
No
Europe: 4
USA: 0.7 |
Yes. The NOEL of 3 mg/kg bw per day for the related substance No. 1072 is > 10 000 times the estimated intake of 2,2'-(dithiodimethylene) difuran when used as a flavouring agent. |
No safety concern |
|
Methyl thiofuroate |
1083 |
13679-61-3
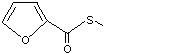 |
No
Europe: 0.4
USA: 0.1 |
Yes. The flavouring agent is expected to hydrolyse to methanethiol and furoic acid. The NOEL of 0.74 mg/kg bw per day for the related substance No. 1071, which also hydrolyses to furoic acid, is > 100 000 times the estimated intake of methyl thiofuroate when used as a flavouring agent. |
No safety concern |
|
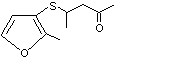
4-[(2-Methyl-3-furyl)thio]-2-pentanone |
1084 |
180031-78-1
 |
No
Europe: ND
USA: 0.6 |
Yes. The NOEL of 3.8 mg/kg bw per day for the related substance No. 1085 is > 100 000 times, respectively, the estimated intake of 4-[(2-methyl-3-furyl)thio]-2-pentanone when used as a flavouring agent. |
No safety concern |
|
2-Methyl-3-tetrahydrofuranthiol |
1090 |
57124-87-5
 |
No
Europe: 4
USA: 0.7 |
Yes. The NOEL of 8.3 mg/kg bw per day for the related substance No. 1089 is > 100 000 times the estimated intake of 2-methyl-3-tetrahydrofuranthiol when used as a flavouring agent. |
No safety concern |
|
2,2'-(Thiodimethylene) difuran |
1080 |
13678-67-6
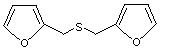 |
No
Europe: 0.9
USA: 0.005 |
Yes. The NOEL of 10 mg/kg bw per day is > 100 000 times the estimated intake of 2,2'-(thiodimethylene) difuran when used as a flavouring agent. |
No safety concern |
|
Structural class III |
|
|
|
|
|
|
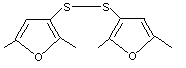
Bis(2,5-dimethyl-3-furyl)disulfide |
1067 |
28588-73-0
 |
No
Europe: 0.01
USA: 0.7 |
Yes. The NOEL of 0.45 mg/kg bw per day for the related substance No. 1066 is > 10 000 times the estimated intake of bis(2,5-dimethyl-3-furyl)disulfide when used as a flavouring agent. |
No safety concern |
|
Bis(2-methyl-3-furyl) tetrasulfide |
1068 |
28588-76-3
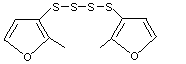 |
No
Europe: ND
USA: 0.7 |
Yes. The NOEL of 0.56 mg/kg bw per day is > 10 000 times the estimated intake of bis(2-methyl-3-furyl) tetrasulfide when used as a flavouring agent. |
No safety concern |
|
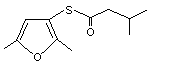
2,5-Dimethyl-3-furan thioisovalerate |
1070 |
55764-28-8
 |
No
Europe: ND
USA: 0.7 |
Yes. The NOEL of 0.73 mg/kg bw per day is > 10 000 times the estimated intake of 2,5-dimethyl-3-furan thioisovalerate when used as a flavouring agent. |
No safety concern |
|
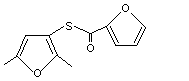
S-2,5-Dimethyl-3-thiofuroyl furan |
1071 |
65505-16-0
 |
No
Europe: ND
USA: 0.01 |
Yes. The NOEL of 0.74 mg/kg bw per day is > 1 million times the estimated intake of S-2,5-dimethyl-3-thiofuroyl furan when used as a flavouring agent. |
No safety concern |
|
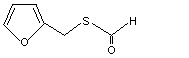
Furfuryl thioformate |
1073 |
59020-90-5
 |
No
Europe: 2
USA: 0.02 |
Yes. The NOELs of 3 and 0.83 mg/kg bw per day for the related substances Nos. 1072 and 1074 are > 1 million and > 10 000 times, respectively, the estimated intake of furfuryl thioformate when used as a flavouring agent. |
No safety concern |
|
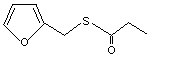
Furfuryl thiopropionate |
1075 |
59020-85-8
 |
No
Europe: 0.01
USA: 0.005 |
Yes. The NOELs of 3 and 0.83 mg/kg bw per day for the related substances Nos. 1072 and 1074 are > 1 million times the estimated intake of furfuryl thiopropionate when used as a flavouring agent. |
No safety concern |
|
Furfuryl isopropyl sulfide |
1077 |
1883-78-9
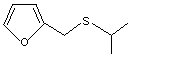 |
No
Europe: 0.001
USA: 0.1 |
Yes. The NOEL of 1.3 mg/kg bw per day is > 100 000 times the estimated intake of furfuryl isopropyl sulfide when used as a flavouring agent. |
No safety concern |
|
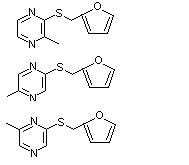
2-Methyl-3-, 5- or 6-(furfurylthio)pyrazine |
1082 |
65530-53-2
 |
No
Europe: 0.4
USA: 0.7 |
Yes. The NOEL of 1.7 mg/kg bw per day is > 100 000 times the estimated intake of 2-methyl-3-, 5- or 6-(furfurylthio)pyrazine when used as a flavouring agent. |
No safety concern |
|
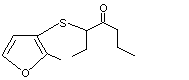
3-[(2-Methyl-3-furyl)thio]-4-heptanone |
1085 |
61295-41-8
 |
No
Europe: ND
USA: 0.7 |
Yes. The NOEL of 3.8 mg/kg bw per day is > 100 000 times the estimated intake of 3-[(2-methyl-3-furyl)thio]-4-heptanone when used as a flavouring agent. |
No safety concern |
|
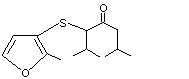
2,6-Dimethyl-3-[(2-methyl-3-furyl)thio]-4-heptanone |
1086 |
61295-51-0
 |
No
Europe: ND
USA: 0.7 |
Yes. The NOEL of 3.8 mg/kg bw per day for the related substance No. 1085 is > 100 000 times the estimated intake of 2,6-dimethyl-3-[(2-methyl-3-furyl)thio]-4-heptanone when used as a flavouring agent. |
No safety concern |
|
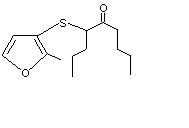
4-[(2-Methyl-3-furyl)thio]-5-nonanone |
1087 |
61295-50-9
 |
No
Europe: ND
USA: 0.7 |
Yes. The NOEL of 3.8 mg/kg bw per day for the related substance No. 1085 is > 100 000 times the estimated intake of 4-[(2-methyl-3-furyl)thio]-5-nonanone when used as a flavouring agent. |
No safety concern |
|
Ethyl 3-(furfurylthio) propionate |
1088 |
94278-27-0
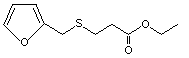 |
No
Europe: 0.01
USA: 0.2 |
Yes. The NOEL of 17 mg/kg bw per day is > 1 million times the estimated intake of ethyl 3-(furfurylthio) propionate when used as a flavouring agent. |
No safety concern |
|
2-Methyl-3-thioacetoxy-4,5-dihydrofuran |
1089 |
26486-14-6
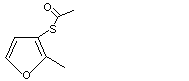 |
No
Europe: ND
USA: 0.7 |
Yes. The NOEL of 8.3 mg/kg bw per day is > 100 000 times the estimated intake of 2-methyl-3-thioacetoxy-4,5-dihydrofuran when used as a flavouring agent. |
No safety concern |
|
cis- and trans-2,5-Dimethyl-3-tetrahydrofuranthiol |
1091 |
26486-21-5
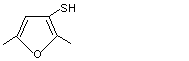 |
No
Europe: ND
USA: 0.9 |
Yes. The NOEL of 8.3 mg/kg bw per day for the related substance No. 1089 is > 100 000 times the estimated intake of cis- and trans-2,5-dimethyl-3-tetrahydrofuranthiol when used as a flavouring agent. |
No safety concern |
|
cis- and trans-2,5-Dimethyltetrahydro-3-furyl thioacetate |
1092 |
252736-39-3
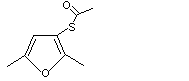 |
No
Europe: ND
USA: 2 |
Yes. The NOEL of 8.3 mg/kg bw per day for the related substance No. 1089 is > 100 000 times the estimated intake of cis- and trans-2,5-dimethyltetrahydro-3-furyl thioacetate when used as a flavouring agent. |
No safety concern |
|
CAS: Chemical Abstracts Service; ND: no intake data reported |
|
a |
Step 2: None of the flavouring agents in this group was predicted to be metabolized to innocuous products. |
|
b |
The human intake threshold is 540 µg/person per day for class II and 90 µg/person per day for class III. All intake levels are expressed in µg/person per day. The combined intake of flavouring agents in class II is 56 and 20 µg/person per day in Europe and the USA, respectively. The combined intake of flavouring agents in class III is 2.4 and 8.8 µg/person per day in Europe and the USA, respectively. |
1.2 Estimated daily intake
The total annual volume of production of the 33 sulfur-substituted furan derivatives is approximately 370 kg in Europe (international Organization of the Falvor Industry, 1995) and 210 kg in the USA (Lucas et al., 1999). Approximately 65% of the total annual volume in Europe and 40% of that in the USA arises from use of one substance, furfuryl mercaptan (No. 1072), with annual volumes of 240 kg in Europe and 82 kg in the USA. The estimated per capita intake of furfuryl mercaptan is 40 µg/day in Europe and 11 µg/day in the USA. The estimated daily intakes of each of the other 32 flavouring agents in this group are less than 5 µg per day in Europe and in the USA.
1.3 Absorption, distribution, metabolism and elimination
No data on the metabolism of these flavouring agents were available; however, all the members of the group contain a reactive divalent sulfur atom attached to the heteroaromatic or heterocyclic ring. Therefore, these flavouring agents are probably metabolized via reactions of the divalent sulfur, like other heteroaromatic ring systems containing sulfur as a side-chain substituent. However, in the absence of data on the disposition of these substances, the metabolic fate of this group of flavouring agents is unknown.
1.4 Application of the Procedure for the Safety Evaluation of Flavouring Agents
|
Step 1. |
Twenty-nine of the 33 flavouring agents in this group are aromatic heterocyclic compounds bearing ring substituents. Twenty-two of the 29 heteroaromatic S-substituted furan derivatives containing only one aromatic ring were assigned to structural class II (Cramer et al., 1978) if they were naturally occurring or to structural class III if they did not occur naturally in foods. The seven remaining heteroaromatic substances contain two aromatic rings. Of these, five agents (Nos 1066, 1067, 1068, 1071 and 1081) would be hydrolysed (thioester) or readily reduced (di- or tetra-sulfides) to mononuclear residues. They were assigned to structural class II or III depending on their natural occurrence. The two remaining substances (Nos 1080 and 1082), which are not readily converted to mononuclear residues, were placed in structural class III. |
| |
The remaining four flavouring agents are heterocyclic but not heteroaromatic (Nos 1089–1092). One agent, 2-methyl-3-tetrahydrofuran-thiol (No. 1090), occurs naturally in foods and was placed in structural class II. The other three agents were placed in structural class III. |
| |
In summary, 18 of the 33 flavouring agents were assigned to structural class II because they are common components of food, while 15 that are not common components of food were assigned to structural class III. |
|
Step 2. |
None of the flavouring agents in this group was predicted to be metabolized to innocuous products. The evaluation of these agents therefore proceeded down the B (right-hand) side of the decision tree. |
|
Step B3. |
The current estimated daily per capita intakes of each of the 33 flavouring agents in this group is below the threshold for human intake for the respective structural classes (540 mg/day for structural class II and 90 mg/day for structural class III). Accordingly, the evaluation of all 33 substances in the group proceeded to step B4. |
|
Step B4. |
The NOEL for 2-methyl-3-furanthiol (No. 1060) in a 90-day dietary study in rats was 5 mg/kg bw per day (Oser, 1970a). This NOEL is appropriate for ethanoic acid, S-(2-methyl-3-furanyl) ester (No. 1069) because the acetate ester would be hydrolysed to 2-methyl-3-furanthiol (No. 1060). This NOEL is also appropriate for the closely structurally related agent, 2,5-dimethyl-3-furanthiol (No. 1063). |
The NOEL for furfuryl isopropyl sulfide (No. 1077) in a 90-day dietary study in rats was 1.3 mg/kg bw per day (Posternak et al., 1969). This NOEL is also appropriate for three structurally related sulfides, 2-methyl-3-(methylthio)furan (No. 1061), 2- methyl-5-(methylthio)furan (No. 1062) and furfuryl methyl sulfide (No. 1076), which would be expected to participate in the same metabolic pathways as furfuryl isopropyl sulfide.
The NOEL for methyl 2-methyl-3-furyl disulfide (No. 1064) in a 90-day dietary study in rats was 1.2 mg/kg bw per day (Gallo et al., 1976a). This NOEL is also appropriate for three structurally related disulfides, propyl 2-methyl-3-furyl disulfide (No. 1065), methyl furfuryl disulfide (No. 1078) and furfuryl propyl disulfide (No. 1079).
The NOEL for bis(2-methyl-3-furyl) disulfide (No. 1066) in a 90-day dietary study in rats was 0.45 mg/kg bw per day (Morgareidge & oser, 1970a). This NOEL is also appropriate for a structurally related bis-disulfide, bis(2,5-dimethyl-3-furyl) disulfide (No. 1067).
The NOEL for furfuryl mercaptan (No. 1072) in a multiple-dose, 13-week study in rats treated by gavage was 3 mg/kg bw per day (Phillips et al., 1977); and the NOEL for furfuryl thioacetate (No. 1074) in a 90-day dietary study in rats was 0.83 mg/kg bw per day (Posternak et al., 1969). These NOELs are also appropriate for the esters furfuryl thioformate (No. 1073) and furfuryl thiopropionate (No. 1075), because they are either close structural relatives of furfuryl thioacetate (No. 1074) or are expected to be hydrolysed to furfuryl mercaptan (No. 1072). The NOEL of 3 mg/kg bw per day for furfuryl mercaptan (No. 1072) is also appropriate for 2,2'-(dithiodimethylene) difuran (No. 1081), because this chemical is anticipated to be readily reduced to furfuryl mercaptan.
The NOEL for 2,5-dimethyl-3-thiofuroyl furan (No. 1071) in a 90-day dietary study in rats was 0.74 mg/kg bw per day (Morgareidge et al., 1974b). This NOEL is appropriate for methyl thiofuroate (No. 1083), because both 2,5-dimethyl-3-thiofuroyl furan and methyl thiofuroate would be hydrolysed to furoic acid.
The NOEL for 3-[(2-methyl-3-furyl)thio]-4-heptanone (No. 1085) in a 90- day dietary study in rats was 3.8 mg/kg bw per day (Gallo et al., 1976b). This NOEL is also appropriate for three structurally related thioketones, 4-[(2-methyl-3-furyl)thio]-2-pentanone (No. 1084), 2,6-dimethyl-3-[(2- methyl-3-furyl)thio]-4-heptanone (No. 1086) and 4-[(2-methyl-3-furyl)thio]-5- nonanone (No. 1087).
The NOEL for 2,2'-(thiodimethylene) difuran (No. 1080) in a 14-day study in rats treated in the diet was 10 mg/kg bw per day (Gill & van Miller, 1987); that for bis(2-methyl-3-furyl) tetrasulfide (No. 1068) in a 90-day study was 0.56 mg/kg bw per day (Morgareidge & Oser, 1970b); that for 2,5-dimethyl-3-furan thioisovalerate (No. 1070) in a 90-day study was 0.73 mg/kg bw per day (Morgareidge et al., 1974a); that for 2-methyl-3-, 5- or 6-(furfurylthio)pyrazine (No. 1082) in a 90-day study was 1.7 mg/kg bw per day (Posternak et al., 1975); and that for ethyl 3-(furfurylthio)propionate (No. 1088) in a 90-day study was 17 mg/kg bw per day (Bio-Research Laboratory, 1980).
The NOEL for 2-methylthioacetoxy-4,5-dihydrofuran (No. 1089) in a 1-year dietary study in rats was 8.3 mg/kg bw per day (Munday & Gellatly, 1974a). This NOEL is also appropriate for three structurally related analogues, 2-methyl-3-tetrahydrofuranthiol (No. 1090), cis- and trans-2,5-dimethyl-3-tetrahydrofuranthiol (No. 1091) and cis-and trans-2,5-dimethyltetrahydro-3-furyl thioacetate (No. 1092), because these chemicals are expected to be hydrolysed to the corresponding dihydro- or tetrahydrofuranthiol.
The NOEL(s) used to evaluate each flavouring agent and the margins of safety they provide, on the basis of current intake levels, are summarized in Table 2.
Table 2. No-observed-effect levels and margins of safety for sulfur-substituted furan derivatives used as flavouring agents
|
Flavouring agent |
No. |
Related flavouring agent(s) |
No. |
NOEL (mg/kg bw per day) |
Reference |
Highest estimated intake
(mg/kg bw per day) |
Margin of safety |
|
Methyl-3-furanthiol |
1060 |
|
|
5a |
Oser (1970a) |
0.015 |
> 100 000 |
|
Ethanoic acid, S-(2-methyl-3-furanyl)ester |
1069 |
|
|
0.001 |
> 1 000 000 |
|
2,5-Dimethyl-3-furanthiol |
1063 |
|
|
0.01 |
> 100 000 |
|
Methyl 2-methyl-3-furyl disulfide |
1064 |
|
|
1.2 a |
Gallo et al. (1976a) |
0.014 |
> 10 000 |
|
Propyl 2-methyl-3-furyl disulfide |
1065 |
|
|
0.01 |
> 100 000 |
|
Methyl furfuryl disulfide |
1078 |
|
|
0.017 |
> 10 000 |
|
Furfuryl propyl disulfide |
1079 |
|
|
0.04 |
> 10 000 |
|
Bis(2-methyl-3-furyl) disulfide |
1066 |
|
|
0.45 a |
Morgareidge & Oser(1970a) |
0.01 |
> 10 000 |
|
Bis(2,5-dimethyl-3-furyl) disulfide |
1067 |
|
|
0.01 |
> 10 000 |
|
Furfuryl mercaptan |
1072 |
|
|
3 a |
Phillips et al. (1977) |
0.58 |
> 1 000 |
|
2,2’-(Dithiodimethylene) difuran |
1081 |
|
|
0.064 |
> 10 000 |
|
Furfuryl thioformate |
1073 |
|
|
0.026 |
> 100 000 |
|
Furfuryl thiopropionate |
1075 |
|
|
0.0002 |
> 1 000 000 |
|
Furfuryl thioacetate |
1074 |
|
|
0.83 a |
Posternak et al. (1969) |
0.008 |
> 100 000 |
|
Furfuryl thioformate |
1073 |
|
|
0.026 |
> 10 000 |
|
Furfuryl thiopropionate |
1075 |
|
|
0.0002 |
> 1 000 000 |
|
4-[(2-Furanmethyl)thio]-2-pentanone |
1084 |
|
|
54b, c |
Wnorowski (1997) |
0.01 |
> 1 000 000 |
|
2-Methyl-3-tetrahydrofuranthiol |
1090 |
12 b, c |
Rush (1991) |
0.07 |
> 100 000 |
|
2,2'-(Thiodimethylene) difuran |
1080 |
|
|
10 b |
Gill & Van Miller, (1987) |
0.014 |
> 100 000 |
|
Bis(2-methyl-3-furyl) tetrasulfide |
1068 |
|
|
0.56 a |
Morgareidge & Oser (1970b) |
0.01 |
> 10 000 |
|
2,5-Dimethyl-3-furan thio-isovalerate |
1070 |
|
|
0.73 a |
Morgareidge et al. (1974a) |
0.01 |
> 10 000 |
|
2,5-Dimethyl-3-thiofuroylfuran |
1071 |
|
|
0.74 a |
Morgareidge et al. (1974b) |
0.0002 |
> 1 000 000 |
| |
|
Methyl thiofuroate |
1083 |
|
|
0.002 |
> 100 000 |
|
Furfuryl isopropyl sulfide |
1077 |
|
|
1.3 a |
Posternak et al. (1969) |
0.002 |
> 100 000 |
| |
|
2-Methyl-3-(methylthio)-furan |
1061 |
|
|
0.024 |
> 10 000 |
| |
|
2-Methyl-5-(methylthio)furan |
1062 |
|
|
0.021 |
> 10 000 |
| |
|
Furfuryl methyl sulfide |
1076 |
|
|
0.02 |
> 10 000 |
|
2-Methyl-3-, 5-, or 6-(furfurylthio)pyrazine |
1082 |
|
1.7 a |
Posternak & Vodoz (1975) |
0.01 |
> 100 000 |
|
|
3-[(2-Methyl-3-furyl)thio]-4-heptanone |
1085 |
|
3.8 a |
Gallo et al. (1976b) |
0.01 |
> 100 000 |
|
| |
|
4-[(2-Methyl-3-furyl)thiol]-2-pentanone |
1084 |
|
|
0.01 |
> 100 000 |
| |
|
2,6-Dimethyl-3-[(2-methyl-3-furyl)thio]-4-heptanone |
1086 |
|
|
0.01 |
> 100 000 |
| |
|
4-[(2-Methyl-3-furyl)thio]-5-nonanone |
1087 |
|
|
0.01 |
> 100 000 |
|
Ethyl 3-(furfurylthio) propionate |
1088 |
|
|
17 a |
Bio-Research Laboratory (1980) |
0.003 |
> 1 000 000 |
|
2-Methyl-3-thioacetoxy-4,5-dihydrofuran |
1089 |
|
|
8.3d |
Munday & Gellatly (1974a) |
0.01 |
> 100 000 |
| |
|
2-Methyl-3-tetrahydro-furanthiol |
1090 |
|
|
0.07 |
> 100 000 |
| |
|
cis- and trans-2,5-Dimethyl-3-tetrahydrofuranthiol |
1091 |
|
|
0.02 |
> 100 000 |
| |
|
cis- and trans-2,5-Dimethyl-tetrahydrofuryl thioacetate |
1092 |
|
|
0.03 |
> 100 000 |
a 90-day study
b 14-day study
c This study was not used in the evaluation as a 90-day study on a structurally related flavouring agent was available.
d 1-year study
1.5 Consideration of combined intake from use as flavouring agents
The hydrolysis of furfuryl thioesters (Nos 1073, 1074 and 1075) or reduction of the disulfides (Nos 1078, 1079 and 1081) in this group would yield the common metabolite furfuryl mercaptan (No. 1072). In the unlikely event that these seven flavouring agents were to be consumed simultaneously on a daily basis, the estimated combined intakes (48 and 15 µg per person per day in Europe and the USA, respectively) would not exceed the threshold for daily human intake of compounds in structural class II (540 µg per person) or III (90 µg per person). Likewise, hydrolysis of 2-methyl-3-furan thioester (No. 1069) or reduction of the corresponding disulfides (Nos 1064, 1065 and 1066) or tetrasulfide (No. 1068) would yield the common metabolite 2-methyl-3-furanthiol (No. 1060). In the unlikely event that these six flavouring agents were to be consumed simultaneously on a daily basis, the estimated combined daily intake would be less than 5 µg per person in Europe and the USA, which would not exceed the threshold for human daily intake of compounds in structural class II (540 µg per person) or III (90 µg per person). On the basis of the evaluation of the collective data, the Committee identified no safety concern from combined intake.
1.6 Conclusions
The Committee concluded that use of flavouring agents in this group of 33 thiofurfuryl and thiofuran derivatives at current levels of intake would not present a safety concern. In the Procedure, data on toxicity were required for all the evaluations. The Committee noted the absence of data on the metabolic fate of these substances but considered that their potential metabolism via the reactive divalent sulfur atom and their very low levels of use as flavouring agents were consistent with the outcome of the evaluations.
2. Relevant background information
2.1 Explanation
The background information provides key data relevant to the safety evaluation of 33 thiofuran derivatives used as flavouring agents. The S-containing functional groups in this review include thiols, mono-, di- and tetra-sulfides and thioesters. Fifteen are either 2- or 3-thiofuran derivatives, 14 are furfurylthio derivatives, and four are dihydro or tetrahydro-3-thiofuran derivatives.
All the substances in the group contain a divalent S atom. They are expected to be metabolized by biotransformations at the reactive S atom. They participate in common routes of absorption, distribution and metabolic detoxication and have similar toxicological end-points. Saturation of metabolic pathways is unlikely, given the extremely low levels of intake of thiofurans from use as flavouring agents or from natural occurrence in food (see section 2.2).
2.2 Additional considerations on intake
Annual use volumes and intake values for each of the sulfur-substituted furan derivatives used as flavouring agents in this group are reported in Table 3. As noted, more than 65% of the total annual volume for flavouring agents in this group in Europe and 40% in the USA comes solely from the use of furfuryl mercaptan (No. 1072) as a flavouring agent (i.e., 244/374 kg in Europe and 82/207 kg in the USA). It is the only substance in this group for which the estimated daily per capita intake1 ("eaters only") from use as a flavouring agent is > 0.1 µg/kg bw. In four national surveys conducted in the USA in 1970, 1975, 1982 and 1987 (National Academy of Sciences, 1989), the highest annual volume of use of furfuryl mercaptan was 160 kg, indicating that the results of surveys for furfuryl mercaptan have been consistent over time.
Table 3. Annual volumes of use of S-substituted furan derivatives used as flavouring agents in Europe and the USA
|
Substance (No.) |
Most recent annual volume (kg)a |
Intakeb |
Annual volume in naturally occurring foods (kg)c |
Consumption ratiod |
|
|
µg/day |
µg/kg bw
per day |
|
|
Structural class II |
|
2-Methyl-3-furanthiol (1060) |
|
Europe |
4.3 |
0.6 |
0.010 |
+ |
NA |
|
USA |
6.8 |
0.9 |
0.015 |
|
|
|
Ethanoic acid, S-(2-methyl-3-furanyl) ester (1069) |
|
Europe |
0.1 |
0.01 |
0.0002 |
+ |
NA |
|
USA |
0.5 |
0.07 |
0.001 |
|
|
|
2-Methyl-3-(methylthio)furan (1061) |
|
Europe |
10 |
1.4 |
0.024 |
6700 |
670 |
|
USA |
0.8 |
0.1 |
0.002 |
|
>10 000 |
|
2-Methyl-5-(methylthio)furan (1062) |
|
Europe |
9 |
1.3 |
0.021 |
+ |
|
|
USA |
0.14 |
0.02 |
0.0001 |
|
NA |
|
Methyl 2-methyl-3-furyl disulfide (1064) |
|
Europe |
6 |
0.9 |
0.014 |
11 |
1.4 |
|
USA |
0.4 |
0.05 |
0.0009 |
|
28 |
|
Propyl 2-methyl-3-furyl disulfide (1065) |
|
Europe |
NR |
NA |
NA |
+ |
NA |
|
USA |
4 |
0.7 |
0.01 |
|
|
|
Bis(2-methyl-3-furyl) disulfide (1066) |
|
Europe |
2.2 |
0.31 |
0.005 |
+ |
NA |
|
USA |
4 |
0.7 |
0.01 |
|
|
|
2,5-Dimethyl-3-furanthiol (1063) |
|
Europe |
0.2 |
0.029 |
0.0005 |
+ |
NA |
|
USA |
4 |
0.7 |
0.01 |
|
|
|
Furfuryl mercaptan (1072) |
|
Europe |
240 |
35 |
0.6 |
1700 |
7 |
|
USA |
82 |
11 |
0.18 |
|
21 |
|
Furfuryl thioacetate (1074) |
|
Europe |
3.5 |
0.5 |
0.008 |
+ |
NA |
|
USA |
0.4 |
0.05 |
0.0009 |
|
|
|
Furfuryl methyl sulfide (1076) |
|
Europe |
8 |
1.1 |
0.019 |
1800 |
230 |
|
USA |
0.9 |
0.11 |
0.002 |
|
2000 |
|
Methyl furfuryl disulfide (1078) |
|
Europe |
7 |
1.0 |
0.017 |
430 |
61 |
|
USA |
0.28 |
0.037 |
0.0006 |
|
1500 |
|
Furfuryl propyl disulfide (1079) |
|
Europe |
NR |
NA |
NA |
+ |
NA |
|
USA |
20 |
2.6 |
0.04 |
|
|
|
2,2'-(Dithiodimethylene)difuran (1081) |
|
Europe |
27 |
3.9 |
0.064 |
+ |
NA |
|
USA |
5 |
0.66 |
0.011 |
|
|
|
Methyl thiofuroate (1083) |
|
Europe |
3 |
0.43 |
0.007 |
+ |
NA |
|
USA |
0.9 |
0.11 |
0.002 |
|
|
|
4-[(2-Furanmethyl)thio]-2-pentanone (1084) |
|
Europe |
NR |
NA |
NA |
+ |
NA |
|
USA |
4.5 |
0.6 |
0.01 |
|
|
|
2-Methyl-3-tetrahydrofuranthiol (1090) |
|
Europe |
29 |
4.1 |
0.07 |
+ |
NA |
|
USA |
4 |
0.7 |
0.01 |
|
|
|
2,2'-(Thiodimethylene)difuran (1080) |
|
Europe |
6 |
0.86 |
0.014 |
+ |
NA |
|
USA |
0.04 |
0.0053 |
0.0001 |
|
|
|
Structural class III |
|
Bis(2,5-dimethyl-3-furyl) disulfide (1067) |
|
Europe |
0.1 |
0.01 |
0.0002 |
– |
NA |
|
USA |
4 |
0.7 |
0.01 |
|
|
|
Bis(2-methyl-3-furyl) tetrasulfide (1068) |
|
Europe |
NR |
NA |
NA |
– |
NA |
|
USA |
4 |
0.7 |
0.01 |
|
|
|
2,5-Dimethyl-3-furyl thioisovalerate (1070) |
|
Europe |
NR |
NA |
NA |
– |
NA |
|
USA |
4 |
0.7 |
0.01 |
|
|
|
2,5-Dimethyl-3-thiofuroylfuran (1071) |
|
Europe |
NR |
NA |
NA |
– |
NA |
|
USA |
0.1 |
0.013 |
0.0002 |
|
|
|
Furfuryl thioformate (1073) |
|
Europe |
11 |
1.6 |
0.026 |
– |
NA |
|
USA |
0.14 |
0.02 |
0.0003 |
|
|
|
Furfuryl thiopropionate (1075) |
|
Europe |
0.1 |
0.014 |
0.0002 |
– |
NA |
|
USA |
0.04 |
0.005 |
0.0001 |
|
|
|
Furfuryl isopropyl sulfide (1077) |
|
Europe |
0.01 |
0.0014 |
0.00002 |
– |
NA |
|
USA |
1 |
0.1 |
0.002 |
|
|
|
2-Methyl-3-, 5- or 6-(furfurylthio)pyrazine (1082) |
|
Europe |
3 |
0.43 |
0.007 |
– |
NA |
|
USA |
4 |
0.7 |
0.01 |
|
|
|
3-[(2-Methyl-3-furyl)thio]-4-heptanone (1085) |
|
Europe |
NR |
NA |
NA |
– |
NA |
|
USA |
4 |
0.7 |
0.01 |
|
|
|
2,6-Dimethyl-3-[(2-methyl-3-furyl)thio]-4-heptanone (1086) |
|
Europe |
NR |
NA |
NA |
– |
NA |
|
USA |
4 |
0.7 |
0.01 |
|
|
|
4-[(2-Methyl-3-furyl)thio]-5-nonanone (1087) |
|
Europe |
NR |
NA |
NA |
– |
NA |
|
USA |
4 |
0.7 |
0.01 |
|
|
|
Ethyl 3-(furfurylthio)propionate (1088) |
|
Europe |
0.1 |
0.01 |
0.0002 |
– |
NA |
|
USA |
1.4 |
0.2 |
0.0031 |
|
|
|
2-Methyl-3-thioacetoxy-4,5-dihydrofuran (1089) |
|
Europe |
NR |
NA |
NA |
– |
NA |
|
USA |
4 |
0.7 |
0.01 |
|
|
|
cis- and trans-2,5-Dimethyl-3-tetrahydrofuranthiol (1091) |
|
Europe |
NR |
NA |
NA |
– |
NA |
|
USA |
7 |
0.9 |
0.02 |
|
|
|
cis- and trans-2,5-Dimethyltetrahydro-3-furyl thioacetate (1092) |
|
Europe |
NR |
NA |
NA |
– |
NA |
|
USA |
15 |
2 |
0.03 |
|
|
|
Total |
|
Europe |
370 |
53 |
0.9 |
|
NA |
|
USA |
210 |
27 |
0.49 |
|
|
|
NR, no data reported; NA, not applicable; +, reported to occur naturally in foods (Maarse et al., 1999) but no quantitative data available; –, not reported to occur naturally in foods |
|
a |
From International Organization of the Flavor Industry (1995) and Lucas et al. (1999) |
|
b |
Intake (µg/person per day) was calculated as follows: [(annual volume, kg) × (1 × 109 µg/kg)/(population × survey correction factor × 365 days)], where population (10%, ‘eaters only’) = 32 × 106 for Europe and 26 × 106 for the USA. The correction factor = 0.6 for Europe and 0.8 for the USA, representing the assumption that only 60% and 80% of the annual production volume of the flavour, resepctively, was reported in the poundage surveys. Intake (µg/kg bw per day) calculated as follows: [(µg/person per day)/body weight], where body weight = 60 kg. Slight variations may occur from rounding. |
|
c |
Quantitative data from Stofberg & Grundschober (1987) |
|
d |
Calculated as follows: (annual consumption in food, kg)/(most recently reported volume as a flavouring agent, kg) |
|
e |
Anticipated annual volume in the USA as reported by the Flavor and Extract Manufacturers Association |
Quantitative data on natural occurrence and consumption ratios have been reported for five substances in the group. They are consumed predominantly from coffee (i.e., consumption ratio > 1) (Stofberg & Kirschman, 1985; Stofberg & Grundschober, 1987) (see Table 3). On the basis of the mean daily per capita consumption of coffee in the USA (Department of Agriculture, 1997), the estimated daily per capita intake of furfuryl mercaptan or furfuryl methyl sulfide from consumption of coffee by men aged > 20 is 0.81 mg. By comparison, the estimated daily per capita intake ("eaters only’) of furfuryl mercaptan from its use as a flavouring agent in the USA is 0.011 mg. Therefore, intake of furfuryl mercaptan as a result of consumption of coffee is approximately 80 times its consumption from use as a flavouring agent (Stofberg & Kirschman, 1985; Stofberg & Grundschober, 1987).
Thiofuran derivatives are known for their powerful aromas in cooked, browned and roasted foods. Their strong organoleptic properties make a significant contribution to the taste and smell of various foods, even when they are present at extremely low concentrations. For instance, at 100 ppm, furfuryl mercaptan (No. 1072) has an extremely powerful, diffusive odor. After dilution to < 2 ppm, it contributes an agreeable, coffee-like, burnt caramel, sweet aroma. The thresholds of aroma detection for thiofurans range from 0.01 ppm for 2-methyl-3-(methylthio)furan (No. 1061) to 0.1 ppm for ethyl 3-(furfurylthio)propionate (No. 1088). In sensory tolerance tests, most panelists reported that atmospheres containing thiofurans were intolerable in the range 0.5–200 ppm (Flavor and Extract Manufacturers’ Association, 1996).
A direct result of their low odour thresholds is that thiolfurans and sulfides have a compelling, self-limiting effect in food. In the vast majority of food categories, < 1 ppm is used. Even in foods in which substantial evaporation of the sulfur derivative occurs during processing (e.g., hard sweets and baked goods), use is predominantly < 20 ppm. The low levels of use in food are reflected in the low reported annual volume of use of thiofurans as flavouring agents.
2.3 Biological data
2.3.1 Biochemical data
Studies have been reported on the hydrolysis in vitro of two substances in this group. Approximately 96% ester hydrolysis was observed when ethyl 3-(furfurylthio) propionate (No. 1088) was incubated in simulated intestinal fluid for 1 h (Bio-Research Laboratory, 1980). When 2-methyl-3-tetrahydrofuranthiol (No. 1090) was incubated in intestinal fluid for 4 h, 18% bis(2-methyl-3-tetrahydrofuryl)disulfide was formed (Salzer, 1991)
2.3.2 Biotransformation
Although no studies in vivo are available, thiofurans and thiofurfuryl derivatives are probably metabolized via reactions of the divalent sulfur atom, like other heteroaromatic ring systems containing a sulfur substituent (Damani, 1987). Thiols are highly reactive in vivo mainly because most exist in the ionized form at physiological pH. Thiols are oxidized to unstable sulfenic acids (RSOH), which are further oxidized to the corresponding sulfinic (RSO2H) and sulfonic acids (RSO3H). Methylation of thiols, primarily by S-adenosyl methionine, yields methyl sulfides, which are then readily oxidized to sulfoxides and sulfones. Thiols may react with physiological thiols to form mixed disulfides or conjugate with glucuronic acid. Oxidation of the alpha-carbon results in desulfuration and formation of an aldehyde, which oxidizes to the corresponding acid (McBain & Menn, 1969; Dutton & Illing, 1972; Maiorino et al., 1989; Richardson et al., 1991).
The labile nature of the S–S bond also presents di- and tetrasulfides with a variety of metabolic options for detoxication, as the disulfide bond is rapidly reduced to the corresponding thiol (i.e. mercaptan) in a reversible reaction in vivo. Thiol–disulfide exchange reactions occur in vivo, resulting from nucleophilic substitution by sulfur. Such reactions with cellular concentrations of endogenous thiols (GSH) and disulfides (GSSG) produce mixed disulfides, which may also undergo reduction (Brigelius, 1985; Sies et al., 1987; Cotgreave et al., 1989).
On the basis of the metabolism of simple aliphatic and aromatic monosulfides, furfuryl and furan monosulfides are expected to undergo oxidation, mainly to the sulfoxide and sulfone. Sulfoxides and sulfones are physiologically stable and are excreted unchanged in the urine (McBain & Menn, 1969; Nickson & Mitchell, 1994; Nickson et al., 1995; Nnane & Damani 1995).
2.4 Toxicological studies
2.4.1 Acute toxicity
LD50 values after oral administration have been reported for 13 of the 33 sulfur compounds in this group, and ranged from 100 mg/kg bw for 2-methyl-3-furanthiol (No. 1060) to > 5000 mg/kg bw for ethyl 3-(furfurylthio)propionate (No. 1088) (see Table 4). Most values were in the range 100–500 mg/kg bw.
Table 4. Studies of the acute toxicity of S-substituted furan derivatives used as flavouring agents used as flavouring agents
|
No. |
Agent |
Species |
Sexa |
Route |
LD50
(mg/kg bw) |
Reference |
|
1060 |
2-Methyl-3-furanthiol |
Mouse |
M,F |
Gavage |
100 |
Oser (1969a) |
|
1060 |
2-Methyl-3-furanthiol |
Mouse |
M,F |
Gavage |
100 |
Moran et al. (1980) |
|
1064 |
Methyl 2-methyl-3-furyl disulfide |
Mouse |
M,F |
Gavage |
140 |
Moran et al. (1980) |
|
1065 |
Propyl 2-methyl-3-furyl disulfide |
Mouse |
M,F |
Gavage |
280 |
Moran et al. (1980) |
|
1066 |
Bis(2-methyl-3-furyl)disulfide |
Mouse |
M,F |
Gavage |
110 |
Oser (1969b) |
|
1063 |
2,5-Dimethyl-3-furanthiol |
Mouse |
M,F |
Gavage |
< 540 |
Fogleman & Suppers (1973c) |
|
1063 |
2,5-Dimethyl-3-furanthiol |
Mouse |
M,F |
Gavage |
360 |
Moran et al.(1980) |
|
1072 |
Furfuryl mercaptan |
Mouse |
M |
Intraperitoneal |
100–200 |
Doull et al.(1962) |
|
1090 |
2-Methyl-3-tetrahydrofuranthiol |
Mouse |
NR |
Gavage |
1900 |
Oser (1970b) |
|
1068 |
Bis(2-methyl-3-furyl)tetrasulfide |
Mouse |
M,F |
Gavage |
220 |
Oser (1970b) |
|
1068 |
Bis(2-methyl-3-furyl)tetrasulfide |
Mouse |
NR |
Gavage |
220 |
Moran et al. (1980) |
|
1070 |
2,5-Dimethyl-3-furanthioisovalerate |
Mouse |
M,F |
Gavage |
580 |
Fogleman & Suppers (1974a) |
|
1070 |
2,5-Dimethyl-3-furanthioisovalerate |
Mouse |
M,F |
Gavage |
720 |
Fogleman & Suppers (1973b) |
|
1071 |
2,5-Dimethyl-3-thiofuroylfuran |
Mouse |
M,F |
Gavage |
620 |
Fogleman & Suppers (1973a,b) |
|
1082 |
2-Methyl-3-, 5- or 6-(furfurylthio)pyrazine |
Rat |
M,F |
Oral |
1000 |
Posternak et al. (1975) |
|
1085 |
3-[(2-Methyl-3-furyl)thio]-4-heptanone |
Mouse |
M,F |
Gavage |
420 |
Moran et al. (1980) |
|
1088 |
Ethyl 3-(furfurylthio)propionate |
Mouse |
M,F |
Oral |
> 5000 |
Griffiths & Babish (1977) |
a M, male; F, female; NR, not reported
2.4.2 Short-term studies of toxicity
Short-term studies (lasting 1 year or less) on toxicity have been performed with 16 thiofuran derivatives, and are summarized in Table 5. Studies on representative thiols (Nos 1060, 1072 and 1090), monosulfides (Nos 1077, 1080, 1082, 1084, 1086 and 1088), disulfides (Nos 1064 and 1066), a tetrasulfide (No. 1068) and thioesters (Nos 1070, 1071, 1074 and 1089) are described below. Most of the studies were carried out in two laboratories, each using a standardized test protocol. The protocol and results are discussed below.
Table 5. Results of short-term studies of the toxicity of S-substituted furan derivatives used as flavouring agents
|
No. |
Substance |
Species, sex |
No. test groupsa/no. per groupb |
Route |
Duration |
NOEL (mg/kg bw per day) |
Reference |
|
1060 |
2-Methyl-3-furanthiol |
Rat, M, F |
1/30 |
Diet |
90 days |
5c |
Oser (1970a) |
|
1064 |
Methyl 2-methyl-3-furyl disulfide |
Rat, M, F |
1/30 |
Diet |
90 days |
1.2c |
Gallo et al. (1976a) |
|
1066 |
Bis(2-methyl-3-furyl) disulfide |
Rat, M, F |
1/30 |
Diet |
90 days |
0.45c |
Morgareidge & Oser (1970a) |
|
1066 |
Bis(2-methyl-3-furyl) disulfide |
Rat, M, F |
1/30 |
Diet |
90 days |
< 4.0 |
Oser (1970b) |
|
1072 |
Furfuryl mercaptan |
Rat, M, F |
3/30 |
Gavage |
91 days |
3 |
Phillips et al. (1977) |
|
1074 |
Furfuryl thioacetate |
Rat, M, F |
1/32 |
Diet |
90 days |
0.81 (F)c |
Posternak et al. (1969) |
|
0.83 (M)c |
|
1084 |
4-[(2-Furanmethyl)thio]-2-pentanone |
Rats, M,F |
1/10 |
Diet |
14 days |
51 (F)c |
Wnorowski (1997) |
|
54 (M) |
|
1090 |
2-Methyl-3-tetrahydrofuranthiol |
Rat, M, F |
1/10 |
Diet |
14 days |
11 (F) |
Rush (1991) |
|
12 (M) |
|
1080 |
2,2'-(Thiodimethylene)difuran |
Rat, M, F |
1/10 |
Diet |
14 days |
10 |
Gill & Van Miller (1987) |
|
1068 |
Bis(2-methyl-3-furyl) tetrasulfide |
Rat, M, F |
1/30 |
Diet |
90 days |
0.56c |
Morgareidge & Oser (1970b) |
|
1070 |
2,5-Dimethyl-3-furan thioisovalerate |
Rat, M, F |
1/30 |
Diet |
90 days |
0.73c |
Morgareidge et al. (1974a) |
|
1071 |
2,5-Dimethyl-3-thiofuroylfuran |
Rat, M, F |
1/30 |
Diet |
90 days |
0.74c |
Morgareidge et al. (1974b) |
|
1077 |
Furfuryl isopropyl sulfide |
Rat, M, F |
1/32 |
Diet |
90 days |
1.3 (F)c |
Posternak et al. (1969) |
|
1.3 (M)c |
|
1082 |
2-Methyl-3-, 5-, or 6-(furfurylthio)-pyrazine |
Rat, M, F |
1/32 |
Diet |
90 days |
1.6 (F)c |
Posternak et al. (1975) |
|
1.7 (M)c |
|
1085 |
3-[(2-Methyl-3-furyl)thio]-4-heptanone |
Rat, M, F |
1/30 |
Diet |
90 days |
3.8c |
Gallo et al. (1976b) |
|
1088 |
Ethyl 3-(furfurylthio)propionate |
Rats, M,F |
2/20 |
Diet |
90 days |
17c |
Bio-Research Laboratory (1980) |
|
1089 |
2-Methyl-3-thioacetoxy-4,5-dihydrofuran |
Rat, M |
1/6 |
Diet |
7 days |
50 |
Munday & Gellatly (1972) |
|
1089 |
2-Methyl-3-thioacetoxy-4,5-dihydrofuran |
Rats, M,F |
6/6 |
Diet |
3-weeks |
2.5 |
Munday & Gellatly (1972) |
|
1089 |
2-Methyl-3-thioacetoxy-4,5-dihydrofuran |
Rats, M,F |
6/16 |
Diet |
13 weeks |
1.4 |
Munday & Gellatly (1973a) |
|
1089 |
2-Methyl-3-thioacetoxy-4,5-dihydrofuran |
Rats, M,F |
2/16 |
Diet |
16 weeks |
< 11 |
Munday & Gellatly (1973b) |
|
1089 |
2-Methyl-3-thioacetoxy-4,5-dihydrofuran |
Rats, M,F |
4/16 |
Diet |
1 year |
8.3c,d |
Munday & Gellatly (1974a) |
|
a |
Does not include control animals |
|
b |
Includes both male and female animals |
|
c |
Performed at either a single or multiple doses that produced no adverse effects. Therefore, this dose level is not a true NOEL but the highest dose tested that produced no adverse effects. The actual NOEL would be higher. |
|
d |
For the first 16 weeks, animals were maintained on diets containing lower doses of 1.4, 2.8, 5.5 or 8.3 mg/kg bw of test material. |
(a) 2-Methyl-3-furanthiol (No. 1060), methyl 2-methyl-3-furyl disulfide (No. 1064), bis(2-methyl-3-furyl) disulfide (No. 1066), bis(2-methyl-3- furyl)tetrasulfide (No. 1068), 2,5-dimethyl-3-furan thioisovalerate (No. 1070), 2,5-dimethyl-3-thiofuroyl furan (No. 1071) and 3-[(2-methyl-3-furyl)thio]-4-heptanone (No. 1085)
A series of 90-day studies was performed with seven of the substances in this group, given at a single target level of intake that was 100 times the estimated possible average daily intake from use of the substance as a flavouring agent. The possible average daily intake was determined by multiplying usual use levels in each of 33 food categories (e.g., baked goods and meat products) by the average amount of that food category consumed daily and summing the intake over all 33 food categories (Department of Agriculture, 1965). The possible average daily intake of the vast majority of flavouring agents with low reported annual volumes of use (National Academy of Sciences, 1987; Lucas et al., 1999) is a gross exaggeration of the average daily intake. The calculation is based on the assumption that all foods in a category always contain the substance and that the food category is consumed each day (Oser & Hall, 1977). Therefore, the dietary concentrations in these studies were many orders of magnitude greater than actual levels of intake of thiofurans derivatives used as flavouring agents.
Each study was performed according to the protocol described below. The results are summarized in Table 5. The only study that showed effects (decreased growth rates in both sexes and decreased food use efficiency and increased liver and kidney weights in males) was conducted with bis(2-methyl-3-furyl) disulfide (No. 1066) at 4 mg/kg bw per day (Morgareidge & Oser, 1970b) and was repeated at a lower level of intake (Morgareidge & Oser, 1970a). This study is discussed separately.
In each study, groups of 15 male and 15 female weanling Wistar rats were individually housed and given their respective diet and tap water ad libitum. The substances were dissolved in acetone and blended into a basal laboratory diet to yield the required daily intake for each substance. Controls received the basal laboratory diet admixed with acetone. The acetone was evaporated from the diets before presentation to the animals. Samples of each diet were taken weekly to assess the stability and concentration of the test material.
Daily observations of appearance, behaviour, appetite, elimination, gross signs of adverse effects and mortality showed no differences among test and control animals. Weekly measurement of body weight and food consumption also showed no significant differences in any of the seven studies. Haematological, blood chemical and urine analyses performed during weeks 6 and 12 on eight males and eight females from each group gave normal values. No differences in absolute or relative weightsof the liver or kidneys were found at necropsy. No evidence of gross or histological alteration was seen in tissues from major organs of eight male and eight female rats or in the livers and kidneys of the remaining seven animals. The levels of intake of these substances that had no effect are listed in Table 5.
In the only study in which effects were found, bis(2-methyl-3-furyl) disulfide (No. 1066) was added to the diet of groups of 15 male and 15 female Wistar rats at a concentration providing a dose of 4 mg/kg bw per day, for 90 days. The body-weight gain of treated males was 37% less than that of controls, and treated males had significantly less efficient food use than control males. Females gained 15% less weight but showed no difference in food use efficiency . The absolute weights of the liver and kidney were lower in treated males, but the relative weights of these organs were significantly greater than in control males. Haematological, blood chemical and urine analyses and gross and histopathological examination showed no observable difference between test and control animals. The authors concluded that treatment was associated with retarded growth rates in animals of each sex, accompanied by decreased food use efficiency and increased relative liver and kidney weights in males. No effects were observed in a 90-day study with the same protocol but at a lower dose (0.45 mg/kg bw per day) (Morgareidge & Oser, 1970a).
(b) Furfuryl mercaptan (No. 1072)
Furfuryl mercaptan is the flavouring agent with the highest reported annual volume of use in Europe and the USA. Groups of 15 weanling Wistar rats of each sex were given a dose of 0, 1, 3 or 30 mg/kg bw per day in corn oil by stomach tube for 13 weeks. Additional groups of five rats of each sex were given 0, 3 or 30 mg/kg bw per day for 2 or 6 weeks. Clinical observations were made daily. Body weights were measured initially on days 1, 6 and 9 and then weekly up to week 12. Food and water intake was measured 1 day before the weight measurements.
At the highest dose, a decrease in body-weight gain associated with reduced food intake was observed, and many differences in organ weights were found at the end of the study. Significant differences in the absolute and relative weights of the brain, kidneys and stomach of males and the brain, heart, liver, kidneys and stomach of females were reported at 30 mg/kg bw per day at 13 weeks; these were considered to be associated with the reduced body weights. Increased relative heart weight in males and reduced absolute and relative kidney weight in females were reported in the group at 3 mg/kg bw per day that was terminated at 6 weeks. These changes were not present in the group at 3 mg/kg bw per day terminated at 13 weeks and were considered not to be related to treatment. Urine analysis, including concentration tests, performed at 6 and 13 weeks revealed no significant differences between test and control groups. Haematological examinations at study termination showed increases in haemoglobin concentration and packed cell volume at the highest dose, but these were not considered to be adverse effects. At termination, macroscopic and microscopic examination showed no evidence of abnormalities related to treatment. As the changes in organ weights were seen only at the highest dose, the authors concluded that 3 mg/kg bw per day had no untoward effects. No NOEL could be identified (Phillips et al., 1977).
(c) Furfuryl thioacetate (No. 1074), furfuryl isopropyl sulfide (No. 1077) and 2-methyl-3-,5- or 6-(furfurylthio)pyrazine (No. 1082)
Three flavouring agents were studied with a similar 90-day protocol. A control and a test group, each consisting of 16 male and 16 female Charles River CD rats, were housed in pairs of the same sex and given access to water and food ad libitum. The concentration of the test material in the diet was adjusted during the study to maintain a constant intake of furfuryl thioacetate of about 0.83 mg/kg bw per day for males and 0.81 mg/kg bw per day for females (Posternak et al., 1969), furfuryl isopropyl sulfide at 1.3 mg/kg bw per day for both males and females (Posternak et al., 1969) and 2-methyl-3-,5- or 6-(furfurylthio)pyrazine at 1.7 mg/kg bw per day for males and 1.6 mg/kg bw per day for females (Posternak et al., 1975). The doses were calculated to be > 100 times the possible average daily intakes. Clinical observations were recorded daily, and food consumption and body weights were determined weekly. During weeks 7 and 13 of the study, haematological and clinical chemical(blood urea) determinations were conducted on 50% of the animals. After 90 days, all animals were killed and subjected to a detailed necropsy, and the liver and kidneys were weighed. A wide range of tissues and organs from each animal were preserved, and major organs and tissues were examined histologically. No differences were observed between groups of test and control animals
(d) 2,2'-(Thiodimethylene)difuran (No. 1080)
Groups of five Fischer 344 rats of each sex were given 2,2'-(thiodimethylene)-difuran at a dose of 10 mg/kg bw per day for 14 days and observed for deaths, body weight, toxic signs and food intake; each underwent gross necropsy, and the liver and kidney were examined microscopically. No biologically significant physical signs of toxicity were seen, and no differences were found in absolute body weight or food consumption, gross observations at necropsy or histological appearance of the liver or kidneys. The treated group showed a statistically significant decrease in body-weight gain, and all male rats showed decreased absolute and relative weights of the liver and decreased absolute kidney weight. The authors considered that the biological significance of the decreases seen was questionable in the absence of other treatment-related effects (Gill & Van Miller, 1987).
(e) 4-[(2-Methyl-3-furyl)thio]-2-pentanone (No. 1084)
In a 14-day study, groups of five Sprague-Dawley rats of each sex were maintained on diets containing 630 ppm of 4-[(2-furanylmethyl)thio]-2-pentanone for 7 days; the concentration was reduced to 530 ppm during the second week to achieve a targeted intake of 41 mg/kg bw per day. Weekly measurement of body weight and food consumption indicated that the average daily intake was 54 mg/kg bw for males and 51 mg/kg bw for females. Animals were observed daily for gross signs of toxicity. On day 15, gross necropsies were performed on all animals, and the kidneys and liver of each animal were removed, weighed and prepared for histological evaluation.
All animals survived and appeared healthy. Although a statistically significant (p < 0.05) increase in relative liver weight was reported in males, no pathological changes were seen microscopically. A significant (p < 0.05) reduction in food consumption was reported for treated males, which was presumed to be related to the poor palatability of the sulfide-containing diet. The change in food consumption was not accompanied by a significant difference in body-weight gain between treated and control groups. Gross necropsy and microscopic examination of the kidneys and liver revealed no effects related to treatment (Wnorowski, 1997).
(f) Ethyl 3-(furfurylthio) propionate (No. 1088)
Ethyl 3-(furfurylthio) propionate was added to the diet of groups of 10 male and 10 female rats (strain not given) at concentrations calculated to result in an average daily intake of 5.8 or 17 mg/kg bw for 90 days. Body weight, food intake and food use efficiency were recorded weekly. No significant difference was found between treated and control animals at either dose. Haematological, blood chemical and urine analyses conducted at weeks 7 and 13 gave normal values. The weights of the liver and kidneys at necropsy were normal, and gross and microscopic examination revealed no dose-related effects (Bio-Research Laboratory, 1980).
(g) 2-Methyl-3-thioacetoxy-4,5-dihydrofuran (No. 1089)
Groups of three male and three female Wistar rats received diets containing 2-methyl-3-thioacetoxy-4,5-dihydrofuran spray-dried in maltodextrin at a concentration of 50, 130, 250, 500, 750 or 1000 ppm for 3 weeks, corresponding to calculated intakes of 2.5, 6.5, 12, 25, 38 and 50 mg/kg bw per day (Food and Drug Administration, 1993). Five male and six female rats were fed a control diet.. The test diets also contained a heat-reaction flavour prepared by heating 4-hydroxy-5-methyl-2,3-dihydrofuran-3-one and 1-cysteine in aqueous solution. The corresponding dietary concentrations of the reaction flavour were 4000, 10 000, 20 000, 40 000, 60 000 and 80 000 ppm. Body-weight gain, food intake, food use efficiency, water intake, activities of serum alanine and aminotransferases, lactic dehydrogenase and alkaline phosphatase, and serum sodium, potassium, chloride, urea nitrogen, creatinine and total protein were measured; haematological parameters (haemoglobin concentration, erythrocyte volume fraction, total leukocyte count and blood film examination), organ weights (heart, liver, spleen, kidneys, brain, testes, adrenals, pituitary and thyroid), and macroscopic and microscopic appearance post mortem were recorded.
Haemolysis of red blood cells occurred at doses > 6.5 mg/kg bw per day, as evidenced by a reduced haemoglobin concentration and erythrocyte volume fraction, increased spleen weights and microscopic changes in the spleen and liver. No other biological effects could be attributed to feeding of the test diets, but it was not possible to determine whether 2-methyl-3-thioacetoxy-4,5-dihydrofuran or the heat-reaction flavour was the haemolytic agent (Munday & Gellatly, 1972).
Groups of eight male and eight female Wistar rats were fed diets containing 28, 56, 110, 170, 280 or 550 ppm of 2-methyl-3-thioacetoxy-4,5-dihydrofuran spray -dried with maltodextrin for 13 weeks, corresponding to calculated intakes of 1.4, 2.8, 5.6, 8.3, 14 and 28 mg/kg bw per day (Food and Drug Administration, 1993). Sixteen male and 16 female rats were fed a control diet, and groups of six rats of each sex per dose and a control group of 12 rats of each sex were fed the same diets for 6 weeks. The parameters studied were the same as those in the 3-week study (Munday & Gallatly, 1972). Haemolysis of red blood cells, as evidenced by reduced erythrocyte volume fraction, generally accompanied by decreased haemoglobin concentrations, increased spleen weights and microscopic changes in the spleen and liver, were observed. The NOEL was 1.4 mg/kg bw per day (Munday & Gellatly, 1973a).
Groups of eight male and eight female Wistar rats were fed diets containing 230 or 550 ppm of 2-methyl-3 thioacetoxy-4,5-dihydrofuran for 16 weeks and then transferred to a control diet for a recovery period of 12 weeks. These concentrations corresponded to calculated intakes of 11 and 28 mg/kg bw per day (Food and Drug Administration, 1993). Sixteen male and 16 female rats were fed the control diet. Half the animals were killed after a further 6 weeks and the remainder after the 12-week recovery period to investigate the reversibility of the haemolytic effects. At necropsy, the spleen, liver and kidneys were weighed and examined histologically. All the changes in the spleen and liver associated with the breakdown and production of red blood cells were reversible after cessation of treatment, except for the levels of splenic haemosiderin, which remained elevated. Haemosiderin, an iron-containing breakdown product of haemoglobin, is a normal constituent of the mammalian organism. Rapid elimination of this material cannot therefore be expected, particularly from the spleen, which is a major depot of haemosiderin in healthy animals (Munday & Gellatly, 1973b).
In the 13-week study of Munday & Gallatly (1973a), an additional eight male and eight female rats were maintained on diets containing 2-methyl-3-thioacetoxy-4,5-dihydrofuran at a concentration of 28, 56, 110 or 170 ppm for the 13 weeks of the study, plus an additional 3 weeks while the results of the 13-week study were being evaluated. All rats were then given the diet containing 170 ppm for a further 36 weeks, for a total duration of 1 year. Observation of general condition and survival, body-weight gain, haematological end-points and macroscopic and microscopic examination at necropsy showed no adverse effects. In particular, there was no evidence of the accelerated splenic erythropoiesis that had been reported in the 13-week study (Munday & Gellatly, 1974).
The haemolysis observed in the study of Munday & Gellatly (1973a) was a result of oxidative stress due to active oxygen species formed via intra-erythrocytic redox cycling of disulfides. Disulfides are reduced to their corresponding thiols by thiol–disulfide exchange with glutathione. Although generally recognized as anti-oxidants, thiols act as pro-oxidants after one electron oxidation to a thyil radical. In erythrocytes, such oxidation is mediated by oxyhaemoglobin and results in the formation of methaemoglobin, hydrogen peroxide and a thiolate anion. The hydrogen peroxide and the more active oxygen species formed from the thyil radical are responsible for initiating the cellular damage that leads to haemolysis (Munday & Mann, 1994). Haemolysis was observed only when very high doses of disulfides were administered. The authors noted that human erythrocytes are usually resistant to oxidative damage, although certain individuals are susceptible because of hereditary deficiencies (Munday et al., 1976, 1990; Munday & Mann, 1994).
(h) 2-Methyl-3-tetrahydrofuranthiol (No. 1090)
2-Methyl-3-tetrahydrofuranthiol was added to the diet of groups of five male and five female Sprague-Dawley rats for 14 days at a concentration calculated to provide a target dose of 10 mg/kg bw per day. Weekly measurements of body weight and food consumption indicated that the average daily intake was 12 mg/kg for males and 11 mg/kg for females. The kidney and liver weights at termination on day 15 didi not differ significant between treated and control groups. Gross necropsy on day 15 and histopathological examination of the kidneys, liver and gross lesions revealed no dose-related adverse effects (Rush, 1991).
3. REFERENCES
Bio-Research Laboratory (1980) Ethyl-beta-furfural-alpha-thiopropionate. Unpublished report. Submitted to WHO by Flavor and Extract Manufacturers’ Association of the United States.
Brigelius, R. (1985) Mixed disulfides: Biological function and increase in oxidative stress. In: Sies, H., ed., Oxidative Stress, New York, Academic Press, pp. 243–271.
Cotgreave, I.A., Atzori, L. & Moldéus, N. (1989) Thiol–disulphide exchange: Physiological and toxicological aspects. In: Damani, L.A., ed., Sulphur-containing Drugs and Related Organic Compounds. Chemistry, Biochemistry and Toxicology, Vol. 2, Part B, Analytical, Biochemical and Toxicological Aspects of Sulphur Xenobiochemistry, New York: John Wiley & Sons, pp. 101–119.
Damani, L.A. (1987) Metabolism of sulphur-containing drugs. In: Benford, D.J., Bridges, J.W. & Gibson, G.G., eds, Drug Metabolism—From Molecules to Man, New York, Taylor & Francis, pp. 581–603.
Department of Agriculture (1965) Food Intake and Nutritive Value of the Diets of Men, Women, and Children in the United States, Spring 1965, Preliminary report, Washington DC: Consumer and Food Economics Research Division, Agricultural Research Service.
Department of Agriculture (1997) Data Tables: Results from USDA’s 1994–96 Continuing Survey of Food Intakes by Individuals and 1994–96 Diet and Health Knowledge Survey, Beltsville, Maryland: Food Surveys Research Group.
Doull, J., Plzak, V. & Brois, S.J. (1962) A Survey of Compounds for Radiation Protection, Arlington, Virginia: United States Air Force, Armed Services Technical Information Agency.
Dutton, G.J. & Illing, H.P.A. (1972) Mechanism of biosynthesis of thio-beta-d-glucosides. Biochem. J., 129, 539–550.
Flavor and Extract Manufacturers’ Association of the United States (1996) Sensory tolerance test protocol. Private communication. Submitted to WHO by Flavor and Extract Manufacturers’ Association of the United States.
Fogleman, R.W. & Suppers, L. (1973a) Acute oral toxicity in mice with 2,5-dimethyl-3-thiofuroylfuran. Private communication. Submitted to WHO by Flavor and Extract Manufacturers’ Association of the United States.
Fogleman, R.W. & Suppers, L. (1973b) Acute oral toxicity in mice with 2,5-dimethyl-3-thioisovalerate. Private communication. Submitted to WHO by Flavor and Extract Manufacturers’ Association of the United States.
Fogleman, R.W. & Suppers, L. (1973c) Acute oral toxicity in mice with 2,5-dimethyl-3-furanthiol. Private communication. Submitted to WHO by Flavor and Extract Manufacturers’ Association of the United States.
Fogleman, R.W. & Suppers, L. (1974a) Acute oral toxicity in mice with 2,5-dimethyl-3-thioisovalerate. Private communication. Submitted to WHO by Flavor and Extract Manufacturers’ Association of the United States.
Fogleman, R.W. & Suppers, L. (1974b) Acute oral toxicity in mice with 2,5-dimethyl-3-thiofuroylfuran. Private communication. Submitted to WHO by Flavor and Extract Manufacturers’ Association of the United States.
Food and Drug Administration (1993) Priority-based Assessment of Food Additives (PAFA) Database, Washington DC: Center for Food Safety and Applied Nutrition, p. 58.
Gallo, M.A., Cox, G.E. & Babish, J.G. (1976a) 90-day feeding studies in rats with compound 75-31065 (methyl benzyl disulfide). Unpublished report from Food and Drug Research Laboratories, Inc. Submitted to WHO by Flavor and Extract Manufacturers’ Association of the United States.
Gallo, M.A., Cox, G.E. & Babish, J.G. (1976b) 90-day feeding study in rats with compound 75-15963 (3-[(2-methyl-3-furyl)-thio]-4-heptanone). Unpublished report from Food and Drug Research Laboratories, Inc. Submitted to WHO by Flavor and Extract Manufacturers’ Association of the United States.
Gill, M.W. & Van Miller, J.P. (1987) Fourteen-day dietary minimum toxicity screen (MTS) in albino rats. Unpublished report No. 50-527 from Bushy Run Research Center, Export, Pennsylvania, USA. Submitted to WHO by Flavor and Extract Manufacturers’ Association of the United States.
Griffiths, J. & Babish, J.G. (1977) Acute oral toxicity (LD50) of ethyl furfuryl alpha-thiopropionate in mice. Private communication. Submitted to WHO by Flavor and Extract Manufacturers’ Association of the United States.
International Organization of the Flavor Industry (1995) European inquiry on volume of use. Private communication. Submitted to WHO by Flavor and Extract Manufacturers’ Association of the United States.
Lucas, C.D., Putnam, J.M. & Hallagan, J.B. (1999) Flavor and Extract Manufacturers Association of the United States 1995 Poundage and Technical Effects Update Survey, Washington DC: Flavor and Extract Manufacturers’ Association of the United States.
Maarse, H., Visscher, C.A., Willemsens, L.C. & Boelens, M.H. (1999) Volatile Components in Food—Qualitative and Quantitative Data, Zeist: Centraal Instituut Voor Voedingsonderzioek TNO.
Maiorino, R.M., Bruce, D.C. & Aposhian, H.V. (1989) Determination and metabolism of dithiol chelating agents. VI. Isolation and identification of the mixed disulfides of meso-2,3-dimercaptosuccinic acid with l-cysteine in human urine. Toxicol. Appl. Pharmacol., 97, 338.
McBain, D.A. & Menn, J.J. (1969) S-Methylation, oxidation, hydroxylation, and conjugation of thiophenol in the rat. Biochem. Pharmacol., 18, 2282–2285.
Moran, E.J., Easterday, O.D. & Oser, B.L. (1980) Acute oral toxicity of selected flavor chemicals. Drug Chem. Toxicol., 3, 249–258.
Morgareidge, K. & Oser, B.L. (1970a) 90-day feeding studies in rats with bis-(2-methyl-3-furyl)-disulfide. Unpublished report. Submitted to WHO by Flavor and Extract Manufacturers’ Association of the United States.
Morgareidge, K. & Oser, B.L. (1970b) 90-day feeding studies in rats with bis-(2-methyl-3-furyl)-tetrasulfide. Unpublished report. Submitted to WHO by Flavor and Extract Manufacturers’ Association of the United States.
Morgareidge, K., Cox, G.E. & Bailey, D.E. (1974a) 90-day feeding study in rats with 2,5-dimethyl-3-thioisovalerylfuran. Unpbulished report. Submitted to WHO by Flavor and Extract Manufacturers’ Association of the United States.
Morgareidge, K., Cox, G.E. & Bailey, D.E. (1974b) 90-day feeding study in rats with 2,5-dimethyl-3-thiofuroylfuran. Unpbulished report. Submitted to WHO by Flavor and Extract Manufacturers’ Association of the United States.
Munday, R. & Gellatly, J.B. (1972) Biological evaluation of dusmelline and DUS-5. I. Palatability study in rats. Unpublished report. Submitted to WHO by Flavor and Extract Manufacturers’ Association of the United States.
Munday, R. & Gellatly, J.B. (1973a) Biological evaluation of feeding trial with DUS-5. 4. 13-week rat. Unpublished report. Submitted to WHO by Flavor and Extract Manufacturers’ Association of the United States.
Munday, R. & Gellatly, J.B. (1973b) Biological evaluation of DUS-5. Part 5. Reversibility of toxic effects induced by feeding of DUS-5. Unpublished report. Submitted to WHO by Flavor and Extract Manufacturers’ Association of the United States.
Munday, R. & Gellatly, J.B. (1974) Biological evaluation of DUS-5. Part 7. 1-year test with DUS-5 in rats. Unpublished report. Submitted to WHO by Flavor and Extract Manufacturers’ Association of the United States.
Munday, R. & Manns, E. (1994) Comparative toxicity of prop(en)yl disulfides derived from Alliaceae. Possible involvement of 1-propenyl disulfides in onion-induced hemolytic anemia. J. Agric. Food Chem., 42, 959–962.
Munday, R., Robinson, J.A., Salmond, G.W. & Gellatly, J.B. (1976) Biochemical effects of haemolytic thiols. Unpublished report. Submitted to WHO by Flavor and Extract Manufacturers’ Association of the United States.
Munday, R., Manns, E. & Fowke, E.A. (1990) Steric effects on the haemolytic activity of aromatic disulphides in rats. Food Chem. Toxicol., 28, 561–566.
National Academy of Sciences (1989) 1987 Poundage and Technical Effects Update of Substances Added to Food, Washington DC.
Nickson, R.M. & Mitchell, S.C. (1994) Fate of dipropyl sulfide and dipropyl sulfoxide in rat. Xenobiotica, 24, 157–168.
Nickson, R.M., Mitchell, S.C. & Zhang, A.Q. (1995) Fate of dipropyl sulfone in rat. Xenobiotica, 25, 1391–1398.
Nnane, P. & Damani, L.A. (1995) The involvement of rat liver CYP2B1 and CYP2D1 in the microsomal sulphoxidation of 4-chlorophenyl methyl sulphide. ISSX Proc. , 8, 110.
Oser, B.L. (1969a) The acute oral toxicity (LD50) to mice of 2-methyl-3-furanthiol. Unpublished report. Submitted to WHO by Flavor and Extract Manufacturers’ Association of the United States.
Oser, B.L. (1969b) The acute oral toxicity (LD50) to mice of bis-(2-methyl-3-furyl) disulfide. Unpublished report. Submitted to WHO by Flavor and Extract Manufacturers’ Association of the United States.
Oser, B.L. (1970a) 90-day feeding studies with 2-methyl-3-furanthiol in rats. Unpublished report. Submitted to WHO by Flavor and Extract Manufacturers’ Association of the United States.
Oser, B.L. (1970b) The acute oral toxicity (LD50) to mice of ten compounds. Unpublished report. Submitted to WHO by Flavor and Extract Manufacturers’ Association of the United States.
Oser, B.L. & Hall, R.L. (1977) Criteria employed by the expert panel of Flavor and Extract Manufacturers’ Association of the United States for the GRAS evaluation of flavoring substances. Food Cosmet. Toxicol., 15, 457–466.
Phillips, J.C., Gaunt, I.F., Hardy, J., Kiss, I.S., Gangolli, S.D. & Butterworth, K.R. (1977) Short-term toxicity of furfuryl mercaptan in rats. Food Cosmet. Toxicol., 15, 383–387.
Posternak, J.M., Linder, A. & Vodoz, C.A. (1969) Summaries of toxicological data. Toxicological tests on flavoring matters. Food Cosmet. Toxicol., 7, 405–407.
Posternak, J.M., Dufour, J.J., Rogg, C. & Vodoz, C.A. (1975) Toxicology tests on flavouring materials. II. Pyrazines and other compounds. Food Cosmet. Toxicol., 13, 487–490.
Richardson, K.A., Edward, V.T., Jones, B.C. & Hutson, D.H. (1991) Metabolism in the rat of a model xenobiotic plant metabolite S-benzyl-N-malonyl-l-cysteine. Xenobiotica, 21, 371.
Rush, R.E. (1991) 2-Methyl-3-tetrahydrofuranthiol. Unpublished report. Submitted to WHO by Flavor and Extract Manufacturers’ Association of the United States.
Salzer, U. (1991) In vitro hydrolysis test for 2-methyl-3-tetrahydrofuranthiol. Unpublished report. Submitted to WHO by Flavor and Extract Manufacturers’ Association of the United States.
Sies, H., Brigelius, R. & Graf, P. (1987) Hormones, glutathione status and protein S-thiolation. Adv. Enzyme Regul., 26, 175–184.
Stofberg, J. & Grundschober, F. (1987) Consumption ratio and food predominance of flavoring materials. Perfumer Flavorist, 12, 27.
Stofberg, J. & Kirschman, J.C. (1985) The consumption ratio of flavoring materials: A mechanism for setting priorities for safety evaluation. Food Chem. Toxicol., 23, 857–860.
Wnorowski, G. (1997) 14-day dietary study in rats. Study No. 5212. Unpublished report No. 088816-3206 from Product Safety Labs, East Brunswick, New Jersey, USA. Submitted to WHO by Flavor and Extract Manufacturers’ Association of the United States.