
ETOFENPROX
First draft prepared by
P.J.C.M. Janssen, P.H. Arentzen, E.M. den Tonkelaar
National Institute of Public Health and Environmental Protection,
Bilthoven, Netherlands
EXPLANATION
Etofenprox is an insecticide with an action similar to the
pyrethroids. It is active against a wide range of insect pests and
is effective against strains of rice green leafhopper and
planthoppers resistant to organophosphorus and carbamate
insecticides. It is also used to control public health pests, and
on livestock (Agrochemicals Handbook, 1991). Etofenprox was
considered for the first time by the present Meeting.
EVALUATION FOR ACCEPTABLE DAILY INTAKE
BIOLOGICAL DATA
Biochemical aspects
Absorption, distribution and excretion
Rats
Groups of 5 Charles River CD rats/sex received a single oral
dose by intubation of 30 or 180 mg/kg bw of 14C-etofenprox (a 1:1
mixture of [1-14C-propyl] etofenprox and [alpha-14C-
benzyl]etofenprox dissolved in PEG 400). Separate groups were used
to determine excretion of radioactivity in urine and faeces,
excretion in bile (bile-cannulated rats) and concentrations in
blood. Tissue radioactivity concentrations were measured in all
groups. Excretion in faeces over the 5 days post-dosing period were
about 87% and 90%, and in urine 9% and 7% at the low and high dose,
respectively. No radioactivity was found in expired air. Mean
plasma radioactivity concentrations reached peak levels (5.1
mg/litre at the low dose and 16.9 mg/litre at the high dose) after 3
to 5 hours. Elimination from plasma was biphasic with half-lives of
7-8 hours for the rapid phase (for both dose levels) and 18 hours
and 35 hours for the slow phase, for the low and high dose,
respectively. Total retention in the tissues at 5 days from
treatment was 3-4% for both doses with highest concentrations being
present in fat (mean of 14 mg/kg at the low dose and 92 mg/kg at the
high dose). Concentrations in other tissues were markedly lower
(0.04-1.7 mg/kg). In rats with cannulated bile ducts, mean
excretion in bile amounted to 15-30% (low dose) and 10% (high dose).
Based on the study results and the biotransformation pattern
identified in excreta, total estimated gastrointestinal absorption
was between 65-93% (low dose) and 48-77% (high dose) (Hawkins et
al., 1985a).
A group of 25 Charles River CD rats/sex was dosed orally with
14C-etofenprox (the same mixture as above) at a dose level of 30
mg/kg bw/day for 7 days. At 4 hours, 1, 2, 5 and 10 days after the
last dose, 5 rats/sex were killed and tissue concentrations were
determined. At 4 hours from the last dose, highest mean tissue
concentrations were present in fat (98 mg/kg), adrenals (42 mg/kg),
ovaries (24 mg/kg), liver (26 mg/kg), thyroid (16 mg/kg) and kidneys
(8.8 mg/kg). Tissue concentrations declined progressively with
time, but fat concentrations declined relatively slowly in a linear
fashion with approximate half-lives of 5 days for males and 8.5 days
for females (Hawkins et al., 1985a).
A further part of the study of Hawkins et al. (1985a) was
focused on transplacental transfer and transfer into milk. This
involved oral dosing with 14C-etofenprox (mixture as above) at 30
mg/kg bw/day to pregnant Charles River CD rats from day 10 through
day 16 of gestation and to pregnant/nursing rats (same strain) from
day 18 of pregnancy until about day 9 after parturition. Groups of
2 pregnant rats were killed before parturition at 4 hours, 1, 2, 3
or 5 days after the final dose. Pups were allowed to suckle the
nursing rats for 1 hour, at which time the pups were killed and
their stomach content was analyzed for radioactivity. Total
radioactivity and elimination patterns in tissues of pregnant rats
were similar to those seen in non-pregnant rats. At 4 hours after
the final dose, radioactivity concentrations were low in placentae
(4.7 mg/kg) and fetuses (1.6 mg/kg) compared to other tissues such
as adrenals (62 mg/kg) and liver (27 mg/kg). Highest maternal tissue
concentrations were present in mammary glands (87 mg/kg); this level
declined with a half-life of about 3.5 days. Secretion of the test
compound into milk was observed. Pup stomach contents contained
radioactivity concentrations of 41-88 mg/kg (compared to maternal
plasma concentrations of 1.9-3.6 mg/litre). After cessation of
exposure this secretion dropped markedly (Hawkins et al., 1985a).
Dogs
Groups of 2 beagle dogs/sex received a single oral dose by
intubation of 30 mg/kg bw of 14C-etofenprox (a 1:1 mixture of [1-
14C-propyl]etofenprox and [alpha-14C-benzyl]etofenprox dissolved in
PEG 400). Radioactivity in blood, urine and faeces was monitored up
to day 5 after treatment. Concentrations in tissues and bile were
determined in two additional groups (2 dogs/sex) given the same dose
and sacrificed after 2 or 4 hours. Total excretion in faeces and
urine was about 90% and 6% of the dose, respectively. In blood
plasma, peak concentrations of 4.4-7.2 mg/litre were reached at
0.25-3 hours after dosing. Blood levels showed absorption to be
more rapid in females. After reaching the peak, radioactivity
concentrations declined with half-lives in the range of
8.6-17 hours. In tissues, highest concentrations were found in
liver (3.1-9.6 mg/kg), followed by kidneys (1.0-3.3 mg/kg) and fat
(0.4-4.1 mg/kg); lowest levels were found in muscle (0.3-0.6 mg/kg).
Concentrations in bile were very high (815-1036 mg/kg), indicating
that this is an important excretion route of absorbed radioactivity.
Based on the study results the pattern of metabolites identified in
excreta, the total estimated gastrointestinal absorption was 14-51%
(Hawkins et al., 1985b).
Biotransformation
Rats
Hawkins et al. (1985a) identified metabolites in their oral
study in Charles River CD rats. Metabolite identification was
carried out in faeces and urine in the single dose groups, in bile
in the bile-cannulated rats (given a single dose of 30 mg/kg bw), in
liver and fat in the repeated-dose group (at 4 hours after the last
dose) and in pup stomach contents in the group dosed during
lactation. Faecal radioactivity was present as unchanged etofenprox
(7-30% of the dose), 2-(4-hydroxyphenyl)-2-methylpropyl-3-
phenoxybenzyl ether (19-25%) and 2-(4-ethoxyphenyl)-2-methylpropyl-
3-(4-hydroxyphenoxy)benzyl ether (7.7-13%). The percentage that was
present as unchanged compound was slightly higher in the high-dose
group compared to the low-dose group and in females compared to
males. The same metabolites were present (as glucuronide or sulfate
conjugates) in liver, bile and, in small amounts (< 2% of the
dose), in urine. No unchanged compound was present in bile. In fat
and milk, radioactivity was almost entirely present as unchanged
etofenprox.
In an additional study, the presence or absence in rats (SD
strain) of 2-(4-ethoxyphenyl)-2-methylpropyl-3-phenoxybenzoate (MTI-
500 alpha-CO, a major metabolite in plants and soil and a
photodegradation product) was determined after administration of a
single dose of 30 mg/kg bw [alpha-14C-benzyl]etofenprox. The
metabolite MTI-500 alpha-CO could be detected in faeces and urine in
trace amounts only (< 0.002% of the dose). The metabolite 3-
phenoxybenzoic acid, the hydrolysis product of MTI-500 alpha-CO, was
found in faeces in larger amounts (4% of the dose). In addition 3-
(4-hydroxyphenoxy)benzoic acid was present (as conjugate) in faeces
(0.47%) and urine (0.006%) (Tomoda et al., 1986).
From these studies in rats the proposed metabolism scheme may
be derived by combining the results of Hawkins et al. (1985a) with
the scheme as proposed by Tomoda et al. (1986). The resulting
metabolism scheme is presented in Figure 1. The metabolite MTI-500
alpha-CO is probably only formed in rats as an intermediate which is
rapidly metabolized further into 3-phenoxybenzoic acid.
Dogs
Hawkins et al. (1985b) identified metabolites in their oral
study in beagle dogs. In blood plasma radioactivity was present as
unchanged etofenprox, 2-(4-hydroxyphenyl)-2-methylpropyl-3-
phenoxybenzyl ether and as 2-(4-ethoxyphenyl)-2-methylpropyl-3-(4-
hydroxyphenoxy)benzyl ether. Faecal radioactivity was present as
unchanged etofenprox (48 or 59% of the dose) and as the two above
metabolites (together 2.9 or 3.5% of the dose). These metabolites
were also present (as glucuronide or sulphate conjugates) in liver,
bile and, in small amounts (together 1.6% of the dose), in urine.
Unchanged compound was also found in the liver. No unchanged
compound was present in bile. Radioactivity in fat was almost
entirely in the form of unchanged etofenprox. The major pathways of
etofenprox biotransformation in dogs derived from these studies is
given in Figure 1.
Toxicological studies
Acute toxicity studies
Results of acute toxicity studies for etofenprox are given in
Table 1. WHO has classified etofenprox as unlikely to present acute
hazard in normal use (WHO, 1992).
Additional acute studies were carried out with Trebon 20 EC, an
emulsifiable concentrate containing 20% etofenprox (+ 6.5%
emulsifier + 73.5% organic solvent). In both mice and rats the oral
LD50 value was > 5 g/kg bw (Kashima, 1985a,b). The dermal LD50 in
rats was > 2 g/kg bw (Kashima, 1985c).
The results of a series of acute studies to determine the
general pharmacological/toxicological action of etofenprox at high
dose levels are given in Table 2 (Kamiya et al. (1985).
Short-term toxicity studies
Mice
Groups of 20 CD-1 mice/sex were fed diets containing 0, 50,
500, 3000 or 15 000 ppm etofenprox (purity 96%) for 13 weeks. These
dose levels were equal to 6.1, 60, 375 or 1975 mg/kg bw/day in males
and 6.9, 71, 390 or 2192 mg/kg bw/day in females. Observations
included clinical signs, mortality, food consumption, body weight,
haematology, clinical chemistry and urinalysis. The weights of 10
organs/animal were recorded. Gross pathology and histopathology
(about 27 tissues/animal) were carried out. At 15 000 ppm the main
effects were: clinical signs (including hunched posture, lethargy,
body tremors, emaciated appearance, respiratory distress), increased
mortality, growth retardation, decreased RBC, Hb, PCV and MCHC,
increased liver weight with hepatocyte enlargement, increased kidney
weight with tubular basophilia and dilatation and microscopic
changes in the lymphoreticular system (increased cellularity of
splenic white pulp, reactive changes in lymph nodes, reduced thymic
cellularity). At 3000 ppm a slight decrease in PCV, Hb and RBC
occurred in males. The effects at 3000 ppm were considered
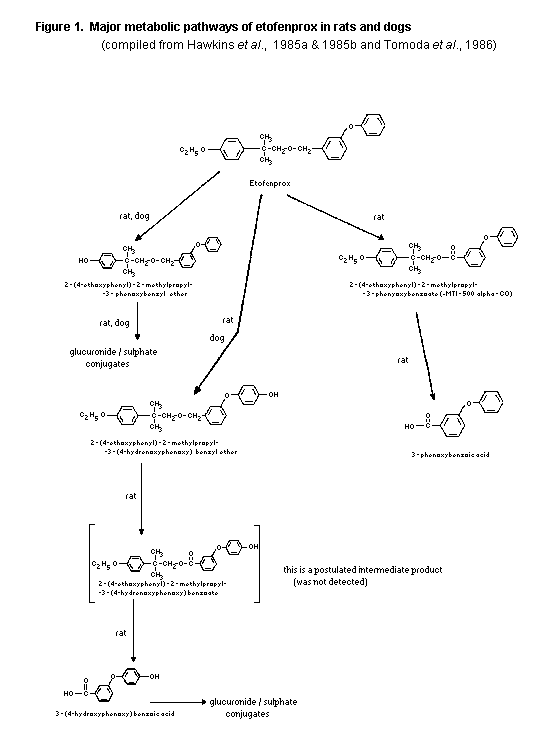 Table 1. Acute toxicity of etofenprox
Species Sex Route LD50 LC50 Purity Reference
(g/kg bw) (g/m3)
Mouse M&F oral > 107.2 96%a Hashimoto et al., 1982a
M&F dermalb > 2.14 96%a Hashimoto et al., 1982a
M i.p. > 53.6 96%a Hashimoto et al., 1982a
F i.p. 13.4 96%a Hashimoto et al., 1982a
M&F s.c. > 53.6 96%a Hashimoto et al., 1982a
Rat M&F oral > 42.88 96%a Hashimoto et al., 1982b
M&F dermalb > 2.14 96%a Hashimoto et al., 1982b
M&F i.p. > 42.88 96%a Hashimoto et al., 1982b
M&F s.c. > 32.16 96%a Hashimoto et al., 1982b
M&F inhalc > 5.9 96%d Jackson et al., 1983
Dog M&F oral > 5.0 96.3%e Harling et al., 1985a
a compound given as undiluted molten solution (40 °C)
b exposure for 24 hours
c exposure for 4 hours
d compound given as aerosol in acetone
e compound given in gelatine capsules
Table 2. Study results - pharmacological/toxicological action (Kamiya et al., 1985)
Parameter Species (sex) Route Result
Central nervous system
- spontaneous motor activity DDY mouse (M) oral dose-related decrease at 25 & 50 g/kg bw (no other
levels tested)
- thiopental-induced sleeping time DDY mouse (M) oral dose-related increase at 25 & 50 g/kg bw; no effect
at 12.5 g/kg bw
- muscle relaxation actiona DDY mouse (M) oral affected at 50 g/kg bw; no effect at 5 g/kg bw
- body (rectum) temperature DDY mouse (M) oral no effect at 25 and 50 g/kg bw
- induced tonic and clonic DDY mouse (M) oral no effect at 5 & 50 g/kg bw
convulsionsb
- general behaviour Wistar rat (M) oral no effect at 1 & 10 g/kg bw
- EEG of frontal lobe Wistar rat (M) oral dose-related effect at 1 & 10 g/kg bw (no other
levels tested)
- EEG of hippocampus Wistar rat (M) oral effect at 10 g/kg bw; no effect at 1 g/kg bw
& parietal lobe
- conscious reaction induced Wistar rat (M) oral no effect at 1 & 10 g/kg bw
by sound stimulation
- spinal reflex potential mongrel cat (M/F) i.d.c no effect at 0.125-1 g/kg bw (cumulative administration)
Effect on smooth muscle
- intestinal charcoal propulsion DDY mouse (M) oral dose-related increase at 25 & 50 g/kg bw; no effect
at 12.5 g/kg bw
- normal movement and contractile rat, in vitro - 0.01-1.0 mmol/l (cumulative administration)
- responsed of vas deferens produced no effect
- autonomous movement of gravid rat. in vitro - 0.001-1.0 mmol/l (cumulative administration)
or non-gravid uteruse produced no effect
- uterus contractabilityf rat, in vitro - no effect at 0.1 mmol/l
- spontaneous movement rabbit, in vitro - 0.01-1.0 mmol/l (cumulative administration)
& contraction of ileum produced no effect
- spontaneous movement of ileum guinea-pig, - 0.001-1.0 mmol/l (cumulative administration)
in vitro produced no effect
Table 2 (contd)
Parameter Species (sex) Route Result
neuro-muscular junction
- contraction of musculus Wistar rat (M) i.v. no effect at 12.5-100 mg/kg bw
gastrocnemiusg
autonomic ganglia
- contractions of nictitating mongrel cat (M/F) i.v. no effect at 10-100 mg/kg bw
membraneh
respiratory and circulatory system
- respiration rate, blood pressure mongrel dog (M/F) i.v. decreased respiration rate (followed by increased rate)
and ECG in combination with increased blood pressure & heart rate
at > 30 mg/kg bw; at 100 mg/kg bw severer effects; no
effect at 10 mg/kg bw
- cardiovascular response as mongrel dog (M/F) i.v. no effect at 100 mg/kg bw
induced by NE, ISP, AC, HIi
& bilateral carotid occlusion
- contractility of isolated atrium guinea-pig, in vitro -0.01-1.0 mmol/l (cumulative administration) produced no
effect
- positive & negative inotropic guinea-pig, in vitro -0.01-1.0 mmol/l (cumulative administration)
reaction of isolated atrium produced no effect on induced neg.
as induced by ISP & HI (+) or inotropy; effect on pos. inotropy
AC (-) at 1.0 mmol/l only (no effect at 0.01-0.1 mmol/l)
urinary volume & components
- measured: volume, Cl-, Na+ & K+ Wistar rat (M) oral dose-related decrease in volume, Cl- & Na+ at 10 & 20 g/kg
bw (no other dose levels tested), no effect on K+
Table 2 (contd)
Parameter Species (sex) Route Result
serum components
- standard blood biochemistry Wistar rat (M) oral increased glucose (after 1 h only) and GOT and GPT
after 1, 2, 6 & 24 hours (all sampling times) at 10 & 20 g/kg bw (no other
levels tested)
blood coagulation
- prothrombin time, partial Wistar rat (M) oral no effect at 10 & 20 g/kg bw
thromboplastin time & fibrinogen
after 1, 2, 6 & 24 hours
a measured as incidence of slipping down a smooth slope
b induced by pentetrazol, strychnine or electro-shock
c i.d. = intraduodenal
d contractile response induced by di-norepinephrine
e from rats in estrus and diestrus cycle or in pregnant stage
f induced by oxytocin
g induced by electrical stimulation of sciatic nerve and muscle
h as induced by pre- and post-ganglionic nerve stimulation
i NE, norepinephrine; ISP, isoproterenol; AC, acetylcholine; HI, histamine
compound-related since the same effects were also seen (more severe
in degree) at 15 000 ppm (the observed decreases were 8.5-11% at 15
000 ppm and 6% in males at 3000 ppm). The NOAEL in this study was
500 ppm (equal to 60 mg/kg bw/day) (Green et al., 1985a).
Rats
Groups of 20 Sprague-Dawley rats/sex were fed diets containing
0, 50, 300, 1800 or 10 800 ppm etofenprox (purity 96%) for 13 weeks.
These dose levels were equal to 3.3, 20, 120 or 734 mg/kg bw/day in
males and 3.8, 23, 142 or 820 mg/kg bw/day in females. Clinical
signs, mortality, food consumption and body weight were recorded.
Haematology, clinical chemistry (including T3 and T4) and
urinalysis were done in all groups at weeks 6 and 12. Additional
haematology consisted of the determination of PT and PTT in the 0
and 10 800 ppm groups at weeks 0 and 13. The weights of 10
organs/animal were recorded. Gross pathology and histopathology
(about 27 tissues/animal) were carried out. At 10 800 ppm the
following effects were observed: slight growth retardation,
decreased food utilization, increased thrombin time, PT and PTT,
decreased T4, increased cholesterol, increased ASAT, ALAT and LDH
(at week 6 only), increased liver weight with slight hepatocyte
enlargement and increased thyroid weight with increased incidence of
microfollicles in thyroids. These effects, except for the increase
in thrombin time (PTT and PT not determined), were also present to a
slight degree at 1800 ppm. The NOAEL in this study was 300 ppm
(equal to 20 mg/kg bw/day) (Green et al., 1985b).
Dogs
Groups of 6 beagle dogs/sex were given diets containing 0 or 10
000 ppm etofenprox (purity 96.3%) for 52 weeks. Two additional
groups of 4 dogs/sex received dietary dose levels of 100 or 1000 ppm
for 52 weeks. These dose levels were equal to 3.5, 33, or 352 mg/kg
bw/day in males and 3.2, 32 or 339 mg/kg bw/day in females. After
52 weeks, 4 dogs/sex/group were killed. The remaining dogs in the 0
and 10 000 ppm groups were killed after a recovery period of 8
weeks. Observations included clinical signs, mortality, food
consumption, body weight, haematology (including PT and PTT),
clinical chemistry and urinalysis. The weights of 10 organs/animal
were recorded. Gross pathology and histopathology (about 33
tissues/animal) were carried out. Effects observed at 10 000 ppm
were decreased PCV, Hb and RBC, increased SAP, decreased total
protein and albumin, increased kidney weight and increased liver
weight with minimal swelling of hepatocytes. After 8 weeks of
recovery none of these effects were found to persist. The NOAEL in
this study was 1000 ppm (equal to 32 mg/kg bw/day) (Harling et al.,
1985b).
Long-term toxicity/carcinogenicity studies
Mice
Groups of 52 CD-1 mice/sex were fed diets containing 0, 30,
100, 700 or 4900 ppm etofenprox (purity 96.3%) for 2 years. These
dose levels were equal to 3.1, 10, 75 or 550 mg/kg bw/day in males
and 3.6, 12, 81 or 615 mg/kg bw/day in females. Satellite groups of
24 mice/sex/group received the same dose levels and were used for
interim sacrifices after 26 weeks (10 animals/sex per group) and 52
weeks (remaining satellite animals). Observations included clinical
signs, mortality, food consumption, body weight, haematology,
clinical chemistry and urinalysis. Ophthalmoscopy was done in the 0
and 4900 ppm groups. The weights of 10 organs/animal were recorded.
Gross pathology and histopathology (about 30 tissues/animal) were
carried out. At 4900 ppm the following effects were seen: increased
mortality in males (survival at test end 10/52 versus 24/52 in
controls; renal lesions were considered contributory to death),
growth retardation, decreased Hb, RBC, MCHC and PCV in combination
with increased MCV (first year only), increased platelet counts
(second year), decreased urine specific gravity, and increased
weights of liver and spleen. Decreased MCHC and increased MCV were
seen at 700 ppm also. Histopathology showed an increased incidence
of renal changes in treated animals, mainly in males. The renal
changes were mostly tubular lesions appearing as groups of
basophilic or dilated tubules, sometimes associated with focal loss
of tubules. These lesions were scored on a grade 1 to 5 scale. The
incidences are presented in Table 3.
At 30 ppm the increase in the incidence of renal lesions was
considered marginal. No increase in tumour incidence was found.
The NOAEL in this study was 30 ppm (equal to 3.1 mg/kg bw/day)
(Green et al., 1986a).
Rats
Groups of 70 Sprague-Dawley rats/sex were fed diets containing
0, 30, 100, 700 or 4900 ppm etofenprox (purity 96.3%) for 2 years.
These dose levels were equal to 1.1, 3.7, 26 or 187 mg/kg bw/day in
males and 1.4, 4.8, 34 or 249 mg/kg bw/day in females. Interim
sacrifices were made after 26 or 52 weeks (10 animals/sex/group on
both occasions). Observations included clinical signs, mortality,
food consumption, body weight, haematology, clinical chemistry
(including T3 and T4) and urinalysis. The weights of 10
organs/animal were recorded. Gross pathology and histopathology
(about 30 tissues/animal) were carried out. At 4900 ppm, the
following effects were seen: decreased food intake or utilization,
decreased growth, slightly increased thrombin time, increased
urinary protein, increased liver weight with hepatocyte enlargement
(sometimes with vacuolization), slightly increased kidney weight and
increased thyroid weight with increased incidence of cystic
follicles in thyroid. At 700 ppm, decreased food intake, increased
thyroid weight, slightly increased kidney weight and presence of
eosinophilic hepatocytes (sometimes with vacuolization) were noted.
At 100 ppm no toxic effects were observed. Increased incidence of
thyroid follicular tumours was found (Table 4). No changes in
concentrations of T3 and T4 were found.
Statistical analysis of these incidences showed that for
thyroid follicular cell carcinomas the trend test was negative and
the intergroup comparison showed no significant effect. Intergroup
comparison showed a statistically significant increase in adenomas
at 4900 ppm in females only. For adenomas and carcinomas, the trend
test was positive in males and females. The NOAEL in this study was
100 ppm (equal to 3.7 mg/kg bw/day) (Green et al., 1986b).
Reproduction studies
Rats
In a two-generation study, groups of 24 or 28 Sprague-Dawley
rats/sex were used as parent animals, receiving dietary
concentrations of 0, 100, 700 or 4900 ppm etofenprox (purity 96.3%).
Two litters per generation were produced. Parent animals were
killed at day 21 after birth of the Fb litters. Pups were also
sacrificed at day 21 after birth except for selected F1a pups
(maintained on test diets up to day 21 after birth of F1b litters),
selected F1b pups (used as F1 parents) and selected F2b pups
(maintained on test diets up to week 13 after birth). Gross
pathology was done and the weights of selected organs were measured.
Histopathology was done in F1 parents (kidneys, thyroids, liver),
F0 parents (kidneys) and F2b adult animals (thyroid). At 4900 ppm,
the main effects were: decreased growth and increased water
consumption (parent animals), increased kidney weights with presence
of cysts, deposits, congestion, haemorrhages or inflammation in
collecting ducts or medulla (parents and young), increased liver
weights with hepatocyte enlargement (parents and young), decreased
pup weights, clinical signs (tremors, abnormal gait) in pups and
increased thyroid weight with slight increase in height of
follicular epithelium (F1 parents). At 700 ppm the effects were:
increased kidney weights in F2b adult females and increased liver
weights in the young of both generations. No effect on reproduction
parameters were observed. The NOAEL in this study was 100 ppm
(equivalent to 5 mg/kg bw/day) (Cozens et al., 1985a).
Table 3. Number of mice showing renal lesions
(Green et al., 1986a)
Dilated/basophilic Control 30 ppm 100 ppm 700 ppm 4900 ppm
cortical tubules
Malesa
grade 1 7 8 6 9 5
grade 2 0 3 4 6 4
grade 3 0 0 1 1 11
grade 4 0 0 1 1 6
grade 5 0 0 0 0 4
Femalesa
grade 1 3 0 5 6 7
grade 2 1 1 1 1 6
grade 3 0 0 0 2 6
grade 4 0 0 1 0 1
grade 5 0 0 0 0 0
a number of animals examined: 52 in all groups
Table 4. Number of rats with thyroid follicular tumours
(Green et al., 1986b)
Tumour type Control 30 ppm 100 ppm 700 ppm 4900 ppm
Malesa
adenomas 6 6 4 5 11
carcinomas 0 0 1 3 2
adenomas and/or 6 6 5 8 13
carcinomas
Femalesa
adenomas 0 3 2 0 9
carcinomas 0 0 0 2 1
adenomas and/or 0 3 2 2 9
carcinomas
a number of animals examined: 50 in all groups
Special studies on embryo/fetotoxicity
Rats
In a segment I study, groups of 24 Sprague-Dawley rats/sex were
given oral doses (by gavage) of 0, 12.5, 250 or 5000 mg/kg bw/day
etofenprox (purity 96.3%) and were mated during a 14-day mating
period. The male animals were treated for 9 weeks prior to mating
up to termination (autopsy) at week 15. Female animals were dosed
for 2 weeks prior to mating up to day 7 of gestation. At day 20 of
gestation females were sacrificed, standard litter data were
recorded and fetuses were monitored for abnormalities (externally
and standard soft tissue and skeletal examinations). No effects on
mating performance, litter parameters, incidences of soft tissue or
skeletal abnormalities were found. In parent animals at 5000 mg/kg
bw/day salivation with brown staining around the mouth,
staining/wetness around the anogenital region, crystalline
appearance of the faeces, unkempt hair coat (males only) and skin
lesions (males only) were present. Salivation with brown staining
around the mouth was also observed occasionally at 250 mg/kg bw/day
and, in a few animals, at 12.5 mg/kg bw/day. Increased salivation
is frequently observed in gavage studies and is probably due to the
method of application rather than representing a compound-related
toxic effect. The NOAEL for reproductive performance and
fetotoxicity was 5000 mg/kg bw/day. The NOAEL for parental toxicity
was 250 mg/kg bw/day (Cozens et al., 1985c).
In a segment II study, groups of 35 pregnant Sprague-Dawley
rats were given oral doses (by gavage) of 0, 12.5, 250 or 5000 mg/kg
bw/day etofenprox (purity 96.3%) from days 6 through 17 of
pregnancy. No test compound was administered at later stages of the
study. On day 20 of gestation, 21-24 dams/group were sacrificed for
a standard teratogenicity evaluation (i.e. number of corpora lutea,
number and distribution of live and dead fetuses, fetal weights,
external abnormalities, soft tissue examination, skeletal
examination). The remaining dams were allowed to deliver F1 pups.
Litter data were recorded at birth. In the 4-21 days period after
birth, a number of neuromotor parameters (including several reflexes
and eye opening) were determined. On day 21, one pup/sex/litter was
selected for use as F1 parents; pups not selected for later mating
were killed and examined internally and externally for
abnormalities. Mating was done when these animals were 12 weeks
old. Behavioural tests were performed prior to mating. At day 21
after birth of F2 pups all animals were killed and examined
internally and externally for abnormalities. At 5000 mg/kg bw/day
maternal growth was retarded. Salivation, red/brown staining around
the mouth, wet and yellow staining of the fur in the anogenital
region were seen in the 5000 mg/kg bw/day dams during the treatment
period. At 250 and 12.5 mg/kg bw/day salivation and red/brown
staining around the mouth occurred in lesser incidences; these
effects are not considered as compound-related adverse effects. No
effects on mating performance, litter parameters, incidences of soft
tissue or skeletal abnormalities were found in any group. The NOAEL
for fetotoxicity was 5000 mg/kg bw/day and the NOAEL for maternal
toxicity was 250 mg/kg bw/day (Cozens et al., 1985b).
In a segment III study, groups of 25 pregnant Sprague-Dawley
rats were given oral doses (by gavage) of 0, 12.5, 250 or 5000 mg/kg
bw/day etofenprox (purity 96.3%) from day 17 of pregnancy through
day 21 after birth of the F1 pups. No test compound was
administered at later stages of the study. Litter data were
recorded at birth of the F1 pups. On day 4, pups were culled to
8/litter (pups not selected were discarded). In the 4-21 days
period after birth a number of neuromotor parameters (including
several reflexes and eye opening) were determined. On day 21, one
pup/sex/litter was selected for later mating (parent animals and
pups not selected were killed and examined internally and
externally). Mating was done when these animals were about 12 weeks
old. Behavioural tests were performed prior to mating (i.e. general
aspects of behaviour, hole board test for mobility and
inquisitivity, inclined-plane test for motor coordination and a one-
trial avoidance test). At day 21 after birth of F2 pups all
animals were killed and examined internally and externally for
abnormalities. In dams at 5000 mg/kg bw/day, decreased growth
during the treatment phase and clinical signs (salivation,
occasional brown staining around the mouth, staining around the
urogenital region) were observed. Only salivation and brown
staining were observed at 250 mg/kg bw/day; these effects are not
considered as compound-related adverse effects. At 12.5 mg/kg
bw/day no effects in dams were found. At 5000 mg/kg bw/day only, in
F1 pups increased mortality (preceding symptoms: body tremors and
haemorrhages around the nose) and decreased growth were found in the
period up to day 21 after birth. Also at 5000 mg/kg bw/day, only in
F1 weanlings and F1 adults, enlarged kidneys and histopathological
kidney changes were observed. The NOAEL for neonatal effects and
maternal toxicity was 250 mg/kg bw/day (Cozens et al., 1985d).
Rabbits
Groups of 16 or 17 mated New Zeeland white rabbits were given
oral doses (by gavage) of 0, 10, 50 or 250 mg/kg bw/day etofenprox
(purity 96.3%) from day 6 through 18 of gestation. On day 29 all
dams were sacrificed and examined according to standard procedures
(litter data, soft tissue examination, skeletal examination).
Slight body-weight loss was seen in dams at 250 mg/kg bw/day and
growth retardation at 50 mg/kg bw/day. At 250 mg/kg bw/day, the
incidences of late abortions and early embryonic deaths were
increased. At 50 mg/kg bw/day no effect on fetuses was found. The
NOAEL for embryo/fetotoxicity was 50 mg/kg bw/day. The NOAEL for
maternal toxicity was 10 mg/kg bw/day (Bottomley et al., 1985).
Special studies on genotoxicity
Etofenprox has been examined for genotoxicity in bacteria,
mammalian cells in vitro and in mice in vivo (micronucleus
test). In addition a test for UDS in human HeLa cells in vitro
has been carried out. The results are presented in Table 5. The
results were negative in all test systems.
Special studies on the metabolite 2-(4-ethoxyphenyl)-
2-methylpropyl-3-phenoxybenzoate (MTI-500 alpha-CO)
The oral LD50 of MTI-500 alpha-CO in male and female Charles
River CD rats was >5000 mg/kg bw (Cummins & Gardner, 1985).
Groups of 10 Sprague-Dawley rats/sex were fed diets containing
0, 50, 700 or 10 000 ppm MTI-500 alpha-CO (purity 97.6%) for 13
weeks. These dose levels were equal to 3.8, 54 or 800 mg/kg bw/day
in males and 4.7, 64 or 930 mg/kg bw/day in females. Clinical
signs, mortality, food consumption and body weights were recorded.
Haematology, clinical chemistry, T3, T4 and urinalysis were done
in all groups at week 13. At termination the weights of 10
organs/animal were recorded. Gross pathology was done in all
animals. Histopathology was done in about 27 tissues/animal in the
0 and 10 000 ppm groups and in lungs, liver and kidneys of the
intermediate groups. At 10 000 ppm the following effects were
observed: growth retardation, increased total WBC (females only),
increased SAP and SGOT, decreases in T4, serum cholesterol and
serum globulin, increased relative weights of liver (females only),
kidneys and thymus (females only), histopathological changes in
kidneys (hypertrophy of the tubular epithelium of the pars recta) in
4/10 females. At the lower dose levels no effects were observed.
The NOAEL in this study was 700 ppm (equal to 54 mg/kg bw/day)
(Powell et al., 1988).
The mutagenic potential of MTI-500 alpha-CO (purity 99.6%) was
examined in the Ames test using Salmonella typhimurium tester
strains TA 98, 100, 97a, 1535, 1537 and 102 without and with
metabolic activation. In addition, Escherichia coli strain WP2
uvra was also used as test organism (without and with activation).
The concentration range was 50-5000 µg/plate. The results were
negative in all test organisms (Bootman & May, 1985).
Table 5. Results of genotoxicity assays with etofenprox
Test system Test object Concentration Purity Results References
of etofenprox (%)
In vitro
Ames test, without S. typhimurium 200-3200 µg/plate; 96.3 negative Foster, 1985
and with activation TA100, TA98, TA1535, DMSO as solvent
TA1537, TA1538
HGPRT (gene Chinese hamster 9.75-156 µg/ml; 96.3 negative Seeberg, 1985a
mutations), without V79 cells DMSO as solvent
and with activation
Chromosome aberrations human lymphocytes 12.5-50 µg/ml; 96.3 negativea Bootman et al.,
test, without and with DMSO as solvent 1985a
activation
Unscheduled DNA human cells HeLa S3 9.75-156 µg/ml 96.3 negative Seeberg, 1985b
synthesis, without (without act.) or
and with activation 2.44-39 µg/ml (with
act.); solvent DMSO
In vivo
Bone marrow CD-1 mouse 80, 400 and 2000 96.3 negative Bootman et al.,
micronucleus test mg/kg bw, p.o. (single 1985b
dose); sacrifice at 24,
48 and 72 h
a Both the test without activation and the test with activation showed a treatment-related reduction in mitotic index.
Special study on sensitization
Etofenprox (purity not reported) was tested for sensitizing
potential in a group of 20 male English Hartley guinea-pigs using
the maximization test according to Magnusson & Kligman. Treatment
in the induction phase was done with a 20% solution in corn oil.
The second induction application was preceded by a treatment with
10% sodium lauryl sulphate in petrolatum ointment to produce an
irritative response in the induction phase. Etofenprox did not
produce sensitization (Kobayashi, 1985).
Special studies on skin and eye irritation
Skin irritation of etofenprox was studied using a group of 6
Japanese white rabbits. A quantity of 0.5 ml of undiluted
etofenprox (purity 96.3%) was applied as a molten solution (melting
point of compound: 35-38 °C) to the shaven intact skin under
occlusion for 4 hours. Skin observations were made up to day 14
after treatment. Very slight erythema was seen in 1/6 animals up to
day 7. The classification (Draize) based on this result is not
irritating (Kashima, 1985d, 1991a).
Eye irritation of etofenprox was studied using a group of 6
Japanese white rabbits. A quantity of 0.1 ml of undiluted
etofenprox (purity 96.3%) was applied as a molten solution (melting
point of compound: 35-38 °C) through instillation into the
conjunctival sac of the right eye. No washing was done.
Observations were made up to 72 hours after treatment. Mild
conjunctival redness was observed in 6/6 animals; after 72 hours
this effect was no longer present. Mild conjunctival edema was seen
in 1/6 animals, which disappeared after 24 hours. The
classification (Draize) based on this result is not irritating
(Kashima, 1985e; 1991b).
Observation in humans
Health assessments were carried out once or twice each year on
a group of 21 operators engaged in the production of technical
etofenprox for periods ranging from 1.5 to 5.5 years. The
examinations included determination of blood pressure, X-ray
examination, haematology, blood biochemistry, limited urinalysis,
electrocardiography and interview for subjective complaints. No
compound-related effects were observed. The report did not include
data on the exposure concentrations (Yamazaki, 1992).
COMMENTS
After oral administration to rats, total excretion in faeces
was 85-90% and 7-9% in urine. Excretion in bile was found to be 10%
to 30%. Unchanged etofenprox was not found in the bile. Total
retention in the body after 5 days was 3-4%. In the
gastrointestinal tract from 48 to 93% was absorbed. Absorption
tended to be dose-dependent. Tissue concentrations were highest in
fat; this residue was present as unchanged parent compound.
Etofenprox was secreted (as the unchanged compound) in milk. The
major biotransformation routes involve O-de-ethylation of the
ethylphenyl moiety and hydroxylation of the phenoxybenzyl moiety
followed by conjugation with glucuronide or sulfate. Oxidation of
the alpha-CH2 group followed by hydrolysis represents an additional
route. The available results for dogs indicate a lower
gastrointestinal absorption rate than in rats. The major
biotransformation routes were the same as in rats.
Etofenprox has a low acute oral toxicity in mice, rats and
dogs. WHO has classified etofenprox as unlikely to present acute
hazard in normal use.
In a 13-week study in mice, using dietary concentrations of 0,
50, 500, 3000 or 15 000 ppm, the NOAEL was 500 ppm, equal to 60
mg/kg bw/day. The main effects seen were mortality, growth
retardation, increased weights of liver (with enlarged hepatocytes)
and kidneys (with tubular basophilia and dilatation) and decreases
in red blood cell parameters.
In a 13-week study in rats using dietary concentrations of 0,
50, 300, 1800 or 10 800 ppm, the NOAEL was 300 ppm, equal to 20
mg/kg bw/day, based on effects on growth and the liver. In
addition, increased thyroid weight with increased incidence of
microfollicles in this organ was observed.
In a 52-week study in dogs using dietary concentrations of 0,
100, 1000 or 10 000 ppm, the NOAEL was 1000 ppm (equal to 32 mg/kg
bw/day) based on decreased red blood cell parameters, increased
serum alkaline phosphatase and increased liver weight (with swelling
of hepatocytes).
In a two-year toxicity/carcinogenicity study in mice using
dietary concentrations of 0, 30, 100, 700 or 4900 ppm, the NOAEL was
30 ppm (equal to 3.1 mg/kg bw/day) based on an increased incidence
of tubular lesions in the kidneys at > 100 ppm. There was no
evidence of carcinogenicity.
A two-year toxicity/carcinogenicity study in rats also used
dietary concentrations of 0, 30, 100, 700 or 4900 ppm. The NOAEL
was 100 ppm (equal to 3.7 mg/kg bw/day), based on increased weights
of thyroid and kidneys and microscopic liver changes at >700 ppm.
The incidence of cystic follicles in the thyroid was increased at
4900 ppm only. There was an increased incidence of thyroid
follicular adenomas among the 4900 ppm animals which was
statistically significantly increased in females only. The absence
of genotoxicity of etofenprox (see below) in combination with the
observed activation of the thyroid gland, which might be related to
the effects on the liver (the latter probably leading to increased
breakdown of thyroid hormones), is a strong indication for a non-
genotoxic mechanism of induction of the thyroid tumours.
In a two-generation study in rats using dietary concentrations
of 0, 100, 700 or 4900 ppm, the NOAEL was 100 ppm (equivalent to 5
mg/kg bw/day). No effects on reproduction were observed in this
study. Main effects seen in parents as well as young were decreased
growth and effects on the weights and histopathology of liver and
kidneys. The effects on the offspring were consistent with exposure
to unchanged etofenprox via milk.
Embryo/fetotoxicity and teratogenicity were studied in rats (3
studies, segment I, II & III, respectively) and rabbits (1 study).
In the three studies in the rat, etofenprox was administered by
gavage at dose levels of 0, 12.5, 250 or 5000 mg/kg bw/day. In each
of the studies, dose-related maternal toxicity (clinical signs,
growth retardation) was observed at 5000 mg/kg bw/day. In the two
studies in rats with dosing before or during pregnancy, no effects
on offspring/fetuses were seen. In the study in rats with dosing
during lactation (segment III), toxic effects developed in the
offspring, most likely as a result of exposure to etofenprox via
milk. In none of the studies in rats were irreversible structural
malformations found. The NOAEL for maternal or parental toxicity in
each of these studies was 250 mg/kg bw/day. For fetotoxicity, the
NOAEL was 5000 mg/kg bw/day in the segment I and II studies. The
NOAEL for neonatal effects in the segment III study was 250 mg/kg
bw/day. In the rabbit study, the NOAEL for maternal toxicity was 10
mg/kg bw/day, based on decreased growth at 50 and 250 mg/kg bw/day.
Incidences of late abortions and early-embryonal mortality were
increased at 250 mg/kg bw/day only. The NOAEL for embryo/
fetotoxicity was 50 mg/kg bw/day. No irreversible structural
malformations were noted in this study.
Based on the results of the available in vitro and in vivo
genotoxicity data there was no evidence that etofenprox is
genotoxic.
The most sensitive species in the animal studies presently
available appear to be rodents, with NOAELs of 3.1 and 3.7 mg/kg
bw/day for mice and rats, respectively, in the long-term studies.
The ADI was based on the long-term study in mice, using a 100-fold
safety factor.
TOXICOLOGICAL EVALUATION
Level causing no toxicological effects
Mouse: 30 ppm, equal to 3.1 mg/kg bw/day (two-year study)
Rat: 100 ppm, equal to 3.7 mg/kg bw/day (two-year study)
Rabbit: 10 mg/kg bw/day (maternal toxicity in a
teratogenicity study)
Dog: 1000 ppm, equal to 32 mg/kg bw/day (52-week study).
Estimate of acceptable daily intake for humans
0-0.03 mg/kg bw.
Studies which will provide information valuable in the continued
evaluation of the compound
Clarification of the dose-response for thyroid effects in the
rat, including evaluation of T3, T4, TSH and other relevant
parameters.
Observations in humans with adequate information on exposure
levels.
REFERENCES
Agrochemicals Handbook (1991) The Agrochemicals Handbook - Third
Edition. Royal Society of Chemistry -Information Services,
Cambridge, England.
Bootman, J., Hodson-Walker, G. & Dance, C.A. (1985a). In vitro
assessment of the clastogenic activity of MTI-500, ethofenprox, in
cultured human peripheral lymphocytes. Life Science Research
Limited, England; report no. 85/MT0017/430, dated 17 July 1985.
Submitted to WHO by Mitsui Toatsu Chemicals, Inc., Tokyo, Japan.
Bootman, J., Hodson-Walker, G. & Dance, C.A. (1985b). MTI-500,
etofenprox: assessment of clastogenic action on bone marrow
erythrocytes in the micronucleus test. Life Science Research
Limited, England; report no. 85/MT0016/406, dated 3 July 1985.
Submitted to WHO by Mitsui Toatsu Chemicals, Inc., Tokyo, Japan.
Bootman, J. & May, K. (1985). MTI-500alpha-CO: assessment of its
mutagenic potential using amino-acid auxotrophs of Salmonella
typhimurium and Escherichia coli. Life Science Research Limited,
England; report no. 85/MT0020/433, dated 19 July 1985. Submitted to
WHO by Mitsui Toatsu Chemicals, Inc., Tokyo, Japan.
Bottomley, A.M., Barton, S.J., Masters, R.E., Offer, J.M., Parker,
C.A., Anderson, A. & Dawe, I.S. (1985). Effect of etofenprox (MTI-
500) on pregnancy of the rabbit. Huntingdon Research Centre Ltd.,
England; report no. MTC 85(84)/85444, dated 20 December 1985.
Submitted to WHO by Mitsui Toatsu Chemicals, Inc., Tokyo, Japan.
Cozens, D.D., Barton, S.J., Offer, J.M., Parker, C.A. & Anderson, A.
(1985a). Effect of etofenprox (MTI-500) on multiple generations of
the rat. Huntingdon Research Centre Ltd., England; report no. MTC
67/85706, dated 29 October 1985. Submitted to WHO by Mitsui Toatsu
Chemicals, Inc., Tokyo, Japan.
Cozens, D.D., Hughes, E.W. & Anderson, A. (1985b). Effect of
etofenprox (MTI-500) on pregnancy of the rat with rearing to
maturation of the F1 generation. Huntingdon Research Centre Ltd.,
England; report no. MTC 64/85422, dated 28 October 1985. Submitted
to WHO by Mitsui Toatsu Chemicals, Inc., Tokyo, Japan.
Cozens, D.D., Hughes, E.W., Clark, R. & Anderson, A. (1985c). Effect
of etofenprox (MTI-500) on fertility and pregnancy of the rat.
Huntingdon Research Centre Ltd., England; report no. MTC 66/84668,
dated 29 October 1985. Submitted to WHO by Mitsui Toatsu Chemicals,
Inc., Tokyo, Japan.
Cozens, D.D., Hughes, E.W., Offer, J. & Anderson, A. (1985d). Effect
of etofenprox (MTI-500) on the peri and post natal period of the rat
with rearing to maturation of the F1 offspring. Huntingdon Research
Centre Ltd., England; report no. MTC 65/85423, dated 29 October
1985. Submitted to WHO by Mitsui Toatsu Chemicals, Inc., Tokyo,
Japan.
Cummins, H.A. & Gardner, J.R. (1985) MTI-500 alpha-CO: Acute oral
toxicity in the rat. Life Science Research Ltd., England - study no.
85/MT0018/474; report dated 2 August 1985. Submitted to WHO by
Mitsui Toatsu Chemicals, Inc., Tokyo, Japan.
Foster, R. (1985). Reverse mutation in Salmonella typhimurium. Test
substance MTI-500. Life Science Research Roma Toxicology Centre,
Italy; report no. 162001-M-06185, dated 22/8/1985. Submitted to WHO
by Mitsui Toatsu Chemicals, Inc., Tokyo, Japan.
Green, O.P., Heywood, R., Street, A.E., Gopinath, & Almond, R.H.
(1985a). Assessment of the toxicity of MTI-500 to mice by dietary
administration for 13 weeks. Huntingdon Research Centre Ltd.,
England; report no. MTC 55/821112, dated 8 July 1983, re-issued 31
October 1985. Submitted to WHO by Mitsui Toatsu Chemicals, Inc.,
Tokyo, Japan.
Green, O.P., Street, A.E., Heywood, R., Gopinath, C. & Almond, R.H.
(1985b). Assessment of the toxicity of MTI-500 in rats during
dietary administration for 13 weeks. Huntingdon Research Centre
Ltd., England; report no. MTC 56/821067, dated 7 July 1983, re-
issued 18 December 1985. Submitted to WHO by Mitsui Toatsu
Chemicals, Inc., Tokyo, Japan.
Green, O.P., Heaps, C.J., Heywood, R., Street, A.E., Gopinath, C.,
Imm, S. & Gibson, W.A. (1986a). Ethofenprox (MTI-500) potential
tumorigenic and toxic effects in prolonged dietary administration to
mice. Huntingdon Research Centre Ltd., England; report no. MTC
58/85582, dated 6 January 1986, re-issued 24 February 1986.
Submitted to WHO by Mitsui Toatsu Chemicals, Inc., Tokyo, Japan.
Green, O.P., Heaps, C.J., Heywood, R., Street, A.E., Gopinath, C.,
Sing, H. & Gibson, W.A. (1986b). Ethofenprox (MTI-500) potential
tumorigenic and toxic effects in prolonged dietary administration to
rats. Huntingdon Research Centre Ltd., England; report no. MTC
59/85581, dated 24 January 1986. Submitted to WHO by Mitsui Toatsu
Chemicals, Inc., Tokyo, Japan.
Harling, R.J., Burford, P. & Heywood, R. (1985a) Ethofenprox (MTI-
500) Acute limit test of toxicity to dogs following a single oral
administration. Huntingdon Research Centre Ltd., England; report no.
MTC 101/851185, dated 24 October 1985. Submitted to WHO by Mitsui
Toatsu Chemicals, Inc., Tokyo, Japan.
Harling, R.J., Burford, P., Street, A.E., Heywood, R., Majeed, S.R.
& Gopinath, C. (1985b). Ethofenprox (MTI-500) toxicity to dogs by
repeated dietary administration for 52 weeks followed by a recovery
period of 8 weeks. Huntingdon Research Centre Ltd., England; report
no. MTC 71/85234, dated 25 October 1985, re-issued 8 November 1985.
Submitted to WHO by Mitsui Toatsu Chemicals, Inc., Tokyo, Japan.
Hashimoto, K., Ohtaki, T., Yamaguchi, K., Nishijima, Y., Imai, K. &
Azegami, J. (1982a). Report on acute toxicity study of MTI-500
(ethofenprox) in mice. Hatano Research Institute, Japan; report no.
A-82-35~42, dated 9 November 1982. Submitted to WHO by Mitsui Toatsu
Chemicals, Inc., Tokyo, Japan.
Hashimoto, K., Ohtaki, T., Yamada, S., Watanabe, C., Imai, K. &
Azegami, J. (1982b). Report on acute toxicity study of MTI-500
(ethofenprox) in rats. Hatano Research Institute, Japan; report no.
A-82-27~34, dated 9 November 1982. Submitted to WHO by Mitsui Toatsu
Chemicals, Inc., Tokyo, Japan.
Hawkins, D.R., Kirkpatrick, D., Ewen, B., Midgley, I., Biggs, S.R. &
Whitby, B.R. (1985a). The biokinetics and metabolism of 14C-
ethofenprox in the rat. Huntingdon Research Centre Ltd., England;
report no. HRC/MTC 68/84610, dated 1 August 1985. Submitted to WHO
by Mitsui Toatsu Chemicals, Inc., Tokyo, Japan.
Hawkins, D.R., Kirkpatrick, D., Ewen, B., Midgley, I. & Biggs, S.R.
(1985b). The metabolism of 14C-ethofenprox in dogs. Huntingdon
Research Centre Ltd., England; report no. HRC/MTC 69/84583, dated 11
October 1985. Submitted to WHO by Mitsui Toatsu Chemicals, Inc.,
Tokyo, Japan.
Jackson, G.C., Hardy, C.J., Clark, G.C., Gregson, R.L., Lewis, D.J.
& Gopinath, C. (1983). MTI-500 acute inhalation toxicity in rats 4
hour exposure. Huntingdon Research Centre Ltd., England; report no.
MTC 60/821079, dated 19 April 1983 Submitted to WHO by Mitsui Toatsu
Chemicals, Inc., Tokyo, Japan.
Kamiya, J., Yoshiwara, K., Saito, S., Takahashi, Y. Oseki, K.,
Shimizu, H., Kawazura, H., Shiga, Y., Yoshida, M., Hayakawa, M. &
Tanaka Y. (1985). General pharmacology of MTI-500. Institute of
Biological Sciences - Mitsui Pharmaceuticals Inc., [A translation of
an original Japanese report published in Japanese, in: Pharmacology
and Therapeutics, vol.13 no.11, pp. 229-244, November 1985].
Translated by T. Hirakawa.
Kashima, M. (1985a). Acute oral toxicity of MTI-500 emulsion in
mice. Nippon Experimental Medical Research Institute, Ltd., Japan;
report no. NEMRI-H-85-50, dated 2 September 1985. Submitted to WHO
by Mitsui Toatsu Chemicals, Inc., Tokyo, Japan.
Kashima, M. (1985b). Acute oral toxicity of MTI-500 emulsion in
rats. Nippon Experimental Medical Research Institute, Ltd., Japan;
report no. NEMRI-H-85-48, dated 2 September 1985. Submitted to WHO
by Mitsui Toatsu Chemicals, Inc., Tokyo, Japan.
Kashima, M. (1985c). Acute dermal toxicity of MTI-500 emulsion in
rats. Nippon Experimental Medical Research Institute, Ltd., Japan;
report no. NEMRI-H-85-49, dated 2 September 1985. Submitted to WHO
by Mitsui Toatsu Chemicals, Inc., Tokyo, Japan.
Kashima, M. (1985d). MTI-500 primary skin stimulation test in
rabbits. Nippon Experimental Medical Research Institute, Ltd, Japan;
report no. NEMRI-H-85-5, dated 23 August 1985. Submitted to WHO by
Mitsui Toatsu Chemicals, Inc., Tokyo, Japan.
Kashima, M. (1985e). MTI-500 primary ophthalmic stimulation test in
rabbits. Nippon Experimental Medical Research Institute, Ltd, Japan;
report no. NEMRI-H-85-55, dated 24 October 1985. Submitted to WHO by
Mitsui Toatsu Chemicals, Inc., Tokyo, Japan.
Kashima (1991a) Final report modification (I) - study name: MTI-500
primary skin stimulation test in rabbits. Nippon Experimental
Medical Research Institute, Ltd, Japan; project no. NEMRI-H-85-5.
Modification d.d. 28 October 1991. Submitted to WHO by Mitsui Toatsu
Chemicals, Inc., Tokyo, Japan.
Kashima (1991b) Final report modification (I) - study name: MTI-500
primary ophthalmic stimulation test in rabbits. Nippon Experimental
Medical Research Institute, Ltd, Japan; project no. NEMRI-H-85-55.
Modification d.d. 28 October 1991. Submitted to WHO by Mitsui Toatsu
Chemicals, Inc., Tokyo, Japan.
Kobayashi, K. (1985). MTI-500 skin sensitization test in guinea-
pigs. Nippon Experimental Medical Research Institute, Ltd, Japan;
report dated 31 October 1985. Submitted to WHO by Mitsui Toatsu
Chemicals, Inc., Tokyo, Japan.
Powell, A.J., Coleman, M., Crook, D., Gopinath, C., Gobson, W.A.,
Read, R.M. & Anderson, A. (1988) MTI-500 alpha-CO: Toxicity to rats
by dietary administration for 13 weeks (Final report). Huntingdon
Research Centre Ltd., England; report no. MTC 141/871448, dated 15
March 1988. Submitted to WHO by Mitsui Toatsu Chemicals, Inc.,
Tokyo, Japan.
Seeberg, A.H. (1985a). Gene mutation in Chinese Hamster V79 cells.
Test substance MTI-500. Life Science Research Roma Toxicology
Centre, Italy; report no. 162002-M-06985, dated 22/8/1985. Submitted
to WHO by Mitsui Toatsu Chemicals, Inc., Tokyo, Japan.
Seeberg, A.H. (1985b). Unscheduled DNA synthesis in human cells,
cell line: Hela S3. Test substance MTI-500. Life Science Research
Roma Toxicology Centre, Italy; report no. 162003-M-05785, dated
30/7/1985. Submitted to WHO by Mitsui Toatsu Chemicals, Inc., Tokyo,
Japan.
Tomoda, K., Arai, M. & Ohnuma, K. (1986). Metabolism study of
ethofenprox (MTI-500). 6. Metabolism in rat. Chiba Branch
Laboratory, Agrochemicals Laboratory - Mitsui Toatsu Chemicals,
Inc., Japan; report dated July 1986. Submitted to WHO by Mitsui
Toatsu Chemicals, Inc., Tokyo, Japan.
WHO (1992). The WHO recommended classification of pesticides by
hazard and guidelines to classification 1992-1993 (WHO/PCS/92.14).
Available from the International Programme on Chemical Safety, World
Health Organization, Geneva, Switzerland.
Yamazaki, Y. (1992). Health report from the Industrial Hygiene
Section, Ohmuta Factory - Mitsui Toatsu Chemicals, Inc., dated 17
April 1992. Submitted to WHO by Mitsui Toatsu Chemicals, Inc.,
Tokyo, Japan.
Table 1. Acute toxicity of etofenprox
Species Sex Route LD50 LC50 Purity Reference
(g/kg bw) (g/m3)
Mouse M&F oral > 107.2 96%a Hashimoto et al., 1982a
M&F dermalb > 2.14 96%a Hashimoto et al., 1982a
M i.p. > 53.6 96%a Hashimoto et al., 1982a
F i.p. 13.4 96%a Hashimoto et al., 1982a
M&F s.c. > 53.6 96%a Hashimoto et al., 1982a
Rat M&F oral > 42.88 96%a Hashimoto et al., 1982b
M&F dermalb > 2.14 96%a Hashimoto et al., 1982b
M&F i.p. > 42.88 96%a Hashimoto et al., 1982b
M&F s.c. > 32.16 96%a Hashimoto et al., 1982b
M&F inhalc > 5.9 96%d Jackson et al., 1983
Dog M&F oral > 5.0 96.3%e Harling et al., 1985a
a compound given as undiluted molten solution (40 °C)
b exposure for 24 hours
c exposure for 4 hours
d compound given as aerosol in acetone
e compound given in gelatine capsules
Table 2. Study results - pharmacological/toxicological action (Kamiya et al., 1985)
Parameter Species (sex) Route Result
Central nervous system
- spontaneous motor activity DDY mouse (M) oral dose-related decrease at 25 & 50 g/kg bw (no other
levels tested)
- thiopental-induced sleeping time DDY mouse (M) oral dose-related increase at 25 & 50 g/kg bw; no effect
at 12.5 g/kg bw
- muscle relaxation actiona DDY mouse (M) oral affected at 50 g/kg bw; no effect at 5 g/kg bw
- body (rectum) temperature DDY mouse (M) oral no effect at 25 and 50 g/kg bw
- induced tonic and clonic DDY mouse (M) oral no effect at 5 & 50 g/kg bw
convulsionsb
- general behaviour Wistar rat (M) oral no effect at 1 & 10 g/kg bw
- EEG of frontal lobe Wistar rat (M) oral dose-related effect at 1 & 10 g/kg bw (no other
levels tested)
- EEG of hippocampus Wistar rat (M) oral effect at 10 g/kg bw; no effect at 1 g/kg bw
& parietal lobe
- conscious reaction induced Wistar rat (M) oral no effect at 1 & 10 g/kg bw
by sound stimulation
- spinal reflex potential mongrel cat (M/F) i.d.c no effect at 0.125-1 g/kg bw (cumulative administration)
Effect on smooth muscle
- intestinal charcoal propulsion DDY mouse (M) oral dose-related increase at 25 & 50 g/kg bw; no effect
at 12.5 g/kg bw
- normal movement and contractile rat, in vitro - 0.01-1.0 mmol/l (cumulative administration)
- responsed of vas deferens produced no effect
- autonomous movement of gravid rat. in vitro - 0.001-1.0 mmol/l (cumulative administration)
or non-gravid uteruse produced no effect
- uterus contractabilityf rat, in vitro - no effect at 0.1 mmol/l
- spontaneous movement rabbit, in vitro - 0.01-1.0 mmol/l (cumulative administration)
& contraction of ileum produced no effect
- spontaneous movement of ileum guinea-pig, - 0.001-1.0 mmol/l (cumulative administration)
in vitro produced no effect
Table 2 (contd)
Parameter Species (sex) Route Result
neuro-muscular junction
- contraction of musculus Wistar rat (M) i.v. no effect at 12.5-100 mg/kg bw
gastrocnemiusg
autonomic ganglia
- contractions of nictitating mongrel cat (M/F) i.v. no effect at 10-100 mg/kg bw
membraneh
respiratory and circulatory system
- respiration rate, blood pressure mongrel dog (M/F) i.v. decreased respiration rate (followed by increased rate)
and ECG in combination with increased blood pressure & heart rate
at > 30 mg/kg bw; at 100 mg/kg bw severer effects; no
effect at 10 mg/kg bw
- cardiovascular response as mongrel dog (M/F) i.v. no effect at 100 mg/kg bw
induced by NE, ISP, AC, HIi
& bilateral carotid occlusion
- contractility of isolated atrium guinea-pig, in vitro -0.01-1.0 mmol/l (cumulative administration) produced no
effect
- positive & negative inotropic guinea-pig, in vitro -0.01-1.0 mmol/l (cumulative administration)
reaction of isolated atrium produced no effect on induced neg.
as induced by ISP & HI (+) or inotropy; effect on pos. inotropy
AC (-) at 1.0 mmol/l only (no effect at 0.01-0.1 mmol/l)
urinary volume & components
- measured: volume, Cl-, Na+ & K+ Wistar rat (M) oral dose-related decrease in volume, Cl- & Na+ at 10 & 20 g/kg
bw (no other dose levels tested), no effect on K+
Table 2 (contd)
Parameter Species (sex) Route Result
serum components
- standard blood biochemistry Wistar rat (M) oral increased glucose (after 1 h only) and GOT and GPT
after 1, 2, 6 & 24 hours (all sampling times) at 10 & 20 g/kg bw (no other
levels tested)
blood coagulation
- prothrombin time, partial Wistar rat (M) oral no effect at 10 & 20 g/kg bw
thromboplastin time & fibrinogen
after 1, 2, 6 & 24 hours
a measured as incidence of slipping down a smooth slope
b induced by pentetrazol, strychnine or electro-shock
c i.d. = intraduodenal
d contractile response induced by di-norepinephrine
e from rats in estrus and diestrus cycle or in pregnant stage
f induced by oxytocin
g induced by electrical stimulation of sciatic nerve and muscle
h as induced by pre- and post-ganglionic nerve stimulation
i NE, norepinephrine; ISP, isoproterenol; AC, acetylcholine; HI, histamine
compound-related since the same effects were also seen (more severe
in degree) at 15 000 ppm (the observed decreases were 8.5-11% at 15
000 ppm and 6% in males at 3000 ppm). The NOAEL in this study was
500 ppm (equal to 60 mg/kg bw/day) (Green et al., 1985a).
Rats
Groups of 20 Sprague-Dawley rats/sex were fed diets containing
0, 50, 300, 1800 or 10 800 ppm etofenprox (purity 96%) for 13 weeks.
These dose levels were equal to 3.3, 20, 120 or 734 mg/kg bw/day in
males and 3.8, 23, 142 or 820 mg/kg bw/day in females. Clinical
signs, mortality, food consumption and body weight were recorded.
Haematology, clinical chemistry (including T3 and T4) and
urinalysis were done in all groups at weeks 6 and 12. Additional
haematology consisted of the determination of PT and PTT in the 0
and 10 800 ppm groups at weeks 0 and 13. The weights of 10
organs/animal were recorded. Gross pathology and histopathology
(about 27 tissues/animal) were carried out. At 10 800 ppm the
following effects were observed: slight growth retardation,
decreased food utilization, increased thrombin time, PT and PTT,
decreased T4, increased cholesterol, increased ASAT, ALAT and LDH
(at week 6 only), increased liver weight with slight hepatocyte
enlargement and increased thyroid weight with increased incidence of
microfollicles in thyroids. These effects, except for the increase
in thrombin time (PTT and PT not determined), were also present to a
slight degree at 1800 ppm. The NOAEL in this study was 300 ppm
(equal to 20 mg/kg bw/day) (Green et al., 1985b).
Dogs
Groups of 6 beagle dogs/sex were given diets containing 0 or 10
000 ppm etofenprox (purity 96.3%) for 52 weeks. Two additional
groups of 4 dogs/sex received dietary dose levels of 100 or 1000 ppm
for 52 weeks. These dose levels were equal to 3.5, 33, or 352 mg/kg
bw/day in males and 3.2, 32 or 339 mg/kg bw/day in females. After
52 weeks, 4 dogs/sex/group were killed. The remaining dogs in the 0
and 10 000 ppm groups were killed after a recovery period of 8
weeks. Observations included clinical signs, mortality, food
consumption, body weight, haematology (including PT and PTT),
clinical chemistry and urinalysis. The weights of 10 organs/animal
were recorded. Gross pathology and histopathology (about 33
tissues/animal) were carried out. Effects observed at 10 000 ppm
were decreased PCV, Hb and RBC, increased SAP, decreased total
protein and albumin, increased kidney weight and increased liver
weight with minimal swelling of hepatocytes. After 8 weeks of
recovery none of these effects were found to persist. The NOAEL in
this study was 1000 ppm (equal to 32 mg/kg bw/day) (Harling et al.,
1985b).
Long-term toxicity/carcinogenicity studies
Mice
Groups of 52 CD-1 mice/sex were fed diets containing 0, 30,
100, 700 or 4900 ppm etofenprox (purity 96.3%) for 2 years. These
dose levels were equal to 3.1, 10, 75 or 550 mg/kg bw/day in males
and 3.6, 12, 81 or 615 mg/kg bw/day in females. Satellite groups of
24 mice/sex/group received the same dose levels and were used for
interim sacrifices after 26 weeks (10 animals/sex per group) and 52
weeks (remaining satellite animals). Observations included clinical
signs, mortality, food consumption, body weight, haematology,
clinical chemistry and urinalysis. Ophthalmoscopy was done in the 0
and 4900 ppm groups. The weights of 10 organs/animal were recorded.
Gross pathology and histopathology (about 30 tissues/animal) were
carried out. At 4900 ppm the following effects were seen: increased
mortality in males (survival at test end 10/52 versus 24/52 in
controls; renal lesions were considered contributory to death),
growth retardation, decreased Hb, RBC, MCHC and PCV in combination
with increased MCV (first year only), increased platelet counts
(second year), decreased urine specific gravity, and increased
weights of liver and spleen. Decreased MCHC and increased MCV were
seen at 700 ppm also. Histopathology showed an increased incidence
of renal changes in treated animals, mainly in males. The renal
changes were mostly tubular lesions appearing as groups of
basophilic or dilated tubules, sometimes associated with focal loss
of tubules. These lesions were scored on a grade 1 to 5 scale. The
incidences are presented in Table 3.
At 30 ppm the increase in the incidence of renal lesions was
considered marginal. No increase in tumour incidence was found.
The NOAEL in this study was 30 ppm (equal to 3.1 mg/kg bw/day)
(Green et al., 1986a).
Rats
Groups of 70 Sprague-Dawley rats/sex were fed diets containing
0, 30, 100, 700 or 4900 ppm etofenprox (purity 96.3%) for 2 years.
These dose levels were equal to 1.1, 3.7, 26 or 187 mg/kg bw/day in
males and 1.4, 4.8, 34 or 249 mg/kg bw/day in females. Interim
sacrifices were made after 26 or 52 weeks (10 animals/sex/group on
both occasions). Observations included clinical signs, mortality,
food consumption, body weight, haematology, clinical chemistry
(including T3 and T4) and urinalysis. The weights of 10
organs/animal were recorded. Gross pathology and histopathology
(about 30 tissues/animal) were carried out. At 4900 ppm, the
following effects were seen: decreased food intake or utilization,
decreased growth, slightly increased thrombin time, increased
urinary protein, increased liver weight with hepatocyte enlargement
(sometimes with vacuolization), slightly increased kidney weight and
increased thyroid weight with increased incidence of cystic
follicles in thyroid. At 700 ppm, decreased food intake, increased
thyroid weight, slightly increased kidney weight and presence of
eosinophilic hepatocytes (sometimes with vacuolization) were noted.
At 100 ppm no toxic effects were observed. Increased incidence of
thyroid follicular tumours was found (Table 4). No changes in
concentrations of T3 and T4 were found.
Statistical analysis of these incidences showed that for
thyroid follicular cell carcinomas the trend test was negative and
the intergroup comparison showed no significant effect. Intergroup
comparison showed a statistically significant increase in adenomas
at 4900 ppm in females only. For adenomas and carcinomas, the trend
test was positive in males and females. The NOAEL in this study was
100 ppm (equal to 3.7 mg/kg bw/day) (Green et al., 1986b).
Reproduction studies
Rats
In a two-generation study, groups of 24 or 28 Sprague-Dawley
rats/sex were used as parent animals, receiving dietary
concentrations of 0, 100, 700 or 4900 ppm etofenprox (purity 96.3%).
Two litters per generation were produced. Parent animals were
killed at day 21 after birth of the Fb litters. Pups were also
sacrificed at day 21 after birth except for selected F1a pups
(maintained on test diets up to day 21 after birth of F1b litters),
selected F1b pups (used as F1 parents) and selected F2b pups
(maintained on test diets up to week 13 after birth). Gross
pathology was done and the weights of selected organs were measured.
Histopathology was done in F1 parents (kidneys, thyroids, liver),
F0 parents (kidneys) and F2b adult animals (thyroid). At 4900 ppm,
the main effects were: decreased growth and increased water
consumption (parent animals), increased kidney weights with presence
of cysts, deposits, congestion, haemorrhages or inflammation in
collecting ducts or medulla (parents and young), increased liver
weights with hepatocyte enlargement (parents and young), decreased
pup weights, clinical signs (tremors, abnormal gait) in pups and
increased thyroid weight with slight increase in height of
follicular epithelium (F1 parents). At 700 ppm the effects were:
increased kidney weights in F2b adult females and increased liver
weights in the young of both generations. No effect on reproduction
parameters were observed. The NOAEL in this study was 100 ppm
(equivalent to 5 mg/kg bw/day) (Cozens et al., 1985a).
Table 3. Number of mice showing renal lesions
(Green et al., 1986a)
Dilated/basophilic Control 30 ppm 100 ppm 700 ppm 4900 ppm
cortical tubules
Malesa
grade 1 7 8 6 9 5
grade 2 0 3 4 6 4
grade 3 0 0 1 1 11
grade 4 0 0 1 1 6
grade 5 0 0 0 0 4
Femalesa
grade 1 3 0 5 6 7
grade 2 1 1 1 1 6
grade 3 0 0 0 2 6
grade 4 0 0 1 0 1
grade 5 0 0 0 0 0
a number of animals examined: 52 in all groups
Table 4. Number of rats with thyroid follicular tumours
(Green et al., 1986b)
Tumour type Control 30 ppm 100 ppm 700 ppm 4900 ppm
Malesa
adenomas 6 6 4 5 11
carcinomas 0 0 1 3 2
adenomas and/or 6 6 5 8 13
carcinomas
Femalesa
adenomas 0 3 2 0 9
carcinomas 0 0 0 2 1
adenomas and/or 0 3 2 2 9
carcinomas
a number of animals examined: 50 in all groups
Special studies on embryo/fetotoxicity
Rats
In a segment I study, groups of 24 Sprague-Dawley rats/sex were
given oral doses (by gavage) of 0, 12.5, 250 or 5000 mg/kg bw/day
etofenprox (purity 96.3%) and were mated during a 14-day mating
period. The male animals were treated for 9 weeks prior to mating
up to termination (autopsy) at week 15. Female animals were dosed
for 2 weeks prior to mating up to day 7 of gestation. At day 20 of
gestation females were sacrificed, standard litter data were
recorded and fetuses were monitored for abnormalities (externally
and standard soft tissue and skeletal examinations). No effects on
mating performance, litter parameters, incidences of soft tissue or
skeletal abnormalities were found. In parent animals at 5000 mg/kg
bw/day salivation with brown staining around the mouth,
staining/wetness around the anogenital region, crystalline
appearance of the faeces, unkempt hair coat (males only) and skin
lesions (males only) were present. Salivation with brown staining
around the mouth was also observed occasionally at 250 mg/kg bw/day
and, in a few animals, at 12.5 mg/kg bw/day. Increased salivation
is frequently observed in gavage studies and is probably due to the
method of application rather than representing a compound-related
toxic effect. The NOAEL for reproductive performance and
fetotoxicity was 5000 mg/kg bw/day. The NOAEL for parental toxicity
was 250 mg/kg bw/day (Cozens et al., 1985c).
In a segment II study, groups of 35 pregnant Sprague-Dawley
rats were given oral doses (by gavage) of 0, 12.5, 250 or 5000 mg/kg
bw/day etofenprox (purity 96.3%) from days 6 through 17 of
pregnancy. No test compound was administered at later stages of the
study. On day 20 of gestation, 21-24 dams/group were sacrificed for
a standard teratogenicity evaluation (i.e. number of corpora lutea,
number and distribution of live and dead fetuses, fetal weights,
external abnormalities, soft tissue examination, skeletal
examination). The remaining dams were allowed to deliver F1 pups.
Litter data were recorded at birth. In the 4-21 days period after
birth, a number of neuromotor parameters (including several reflexes
and eye opening) were determined. On day 21, one pup/sex/litter was
selected for use as F1 parents; pups not selected for later mating
were killed and examined internally and externally for
abnormalities. Mating was done when these animals were 12 weeks
old. Behavioural tests were performed prior to mating. At day 21
after birth of F2 pups all animals were killed and examined
internally and externally for abnormalities. At 5000 mg/kg bw/day
maternal growth was retarded. Salivation, red/brown staining around
the mouth, wet and yellow staining of the fur in the anogenital
region were seen in the 5000 mg/kg bw/day dams during the treatment
period. At 250 and 12.5 mg/kg bw/day salivation and red/brown
staining around the mouth occurred in lesser incidences; these
effects are not considered as compound-related adverse effects. No
effects on mating performance, litter parameters, incidences of soft
tissue or skeletal abnormalities were found in any group. The NOAEL
for fetotoxicity was 5000 mg/kg bw/day and the NOAEL for maternal
toxicity was 250 mg/kg bw/day (Cozens et al., 1985b).
In a segment III study, groups of 25 pregnant Sprague-Dawley
rats were given oral doses (by gavage) of 0, 12.5, 250 or 5000 mg/kg
bw/day etofenprox (purity 96.3%) from day 17 of pregnancy through
day 21 after birth of the F1 pups. No test compound was
administered at later stages of the study. Litter data were
recorded at birth of the F1 pups. On day 4, pups were culled to
8/litter (pups not selected were discarded). In the 4-21 days
period after birth a number of neuromotor parameters (including
several reflexes and eye opening) were determined. On day 21, one
pup/sex/litter was selected for later mating (parent animals and
pups not selected were killed and examined internally and
externally). Mating was done when these animals were about 12 weeks
old. Behavioural tests were performed prior to mating (i.e. general
aspects of behaviour, hole board test for mobility and
inquisitivity, inclined-plane test for motor coordination and a one-
trial avoidance test). At day 21 after birth of F2 pups all
animals were killed and examined internally and externally for
abnormalities. In dams at 5000 mg/kg bw/day, decreased growth
during the treatment phase and clinical signs (salivation,
occasional brown staining around the mouth, staining around the
urogenital region) were observed. Only salivation and brown
staining were observed at 250 mg/kg bw/day; these effects are not
considered as compound-related adverse effects. At 12.5 mg/kg
bw/day no effects in dams were found. At 5000 mg/kg bw/day only, in
F1 pups increased mortality (preceding symptoms: body tremors and
haemorrhages around the nose) and decreased growth were found in the
period up to day 21 after birth. Also at 5000 mg/kg bw/day, only in
F1 weanlings and F1 adults, enlarged kidneys and histopathological
kidney changes were observed. The NOAEL for neonatal effects and
maternal toxicity was 250 mg/kg bw/day (Cozens et al., 1985d).
Rabbits
Groups of 16 or 17 mated New Zeeland white rabbits were given
oral doses (by gavage) of 0, 10, 50 or 250 mg/kg bw/day etofenprox
(purity 96.3%) from day 6 through 18 of gestation. On day 29 all
dams were sacrificed and examined according to standard procedures
(litter data, soft tissue examination, skeletal examination).
Slight body-weight loss was seen in dams at 250 mg/kg bw/day and
growth retardation at 50 mg/kg bw/day. At 250 mg/kg bw/day, the
incidences of late abortions and early embryonic deaths were
increased. At 50 mg/kg bw/day no effect on fetuses was found. The
NOAEL for embryo/fetotoxicity was 50 mg/kg bw/day. The NOAEL for
maternal toxicity was 10 mg/kg bw/day (Bottomley et al., 1985).
Special studies on genotoxicity
Etofenprox has been examined for genotoxicity in bacteria,
mammalian cells in vitro and in mice in vivo (micronucleus
test). In addition a test for UDS in human HeLa cells in vitro
has been carried out. The results are presented in Table 5. The
results were negative in all test systems.
Special studies on the metabolite 2-(4-ethoxyphenyl)-
2-methylpropyl-3-phenoxybenzoate (MTI-500 alpha-CO)
The oral LD50 of MTI-500 alpha-CO in male and female Charles
River CD rats was >5000 mg/kg bw (Cummins & Gardner, 1985).
Groups of 10 Sprague-Dawley rats/sex were fed diets containing
0, 50, 700 or 10 000 ppm MTI-500 alpha-CO (purity 97.6%) for 13
weeks. These dose levels were equal to 3.8, 54 or 800 mg/kg bw/day
in males and 4.7, 64 or 930 mg/kg bw/day in females. Clinical
signs, mortality, food consumption and body weights were recorded.
Haematology, clinical chemistry, T3, T4 and urinalysis were done
in all groups at week 13. At termination the weights of 10
organs/animal were recorded. Gross pathology was done in all
animals. Histopathology was done in about 27 tissues/animal in the
0 and 10 000 ppm groups and in lungs, liver and kidneys of the
intermediate groups. At 10 000 ppm the following effects were
observed: growth retardation, increased total WBC (females only),
increased SAP and SGOT, decreases in T4, serum cholesterol and
serum globulin, increased relative weights of liver (females only),
kidneys and thymus (females only), histopathological changes in
kidneys (hypertrophy of the tubular epithelium of the pars recta) in
4/10 females. At the lower dose levels no effects were observed.
The NOAEL in this study was 700 ppm (equal to 54 mg/kg bw/day)
(Powell et al., 1988).
The mutagenic potential of MTI-500 alpha-CO (purity 99.6%) was
examined in the Ames test using Salmonella typhimurium tester
strains TA 98, 100, 97a, 1535, 1537 and 102 without and with
metabolic activation. In addition, Escherichia coli strain WP2
uvra was also used as test organism (without and with activation).
The concentration range was 50-5000 µg/plate. The results were
negative in all test organisms (Bootman & May, 1985).
Table 5. Results of genotoxicity assays with etofenprox
Test system Test object Concentration Purity Results References
of etofenprox (%)
In vitro
Ames test, without S. typhimurium 200-3200 µg/plate; 96.3 negative Foster, 1985
and with activation TA100, TA98, TA1535, DMSO as solvent
TA1537, TA1538
HGPRT (gene Chinese hamster 9.75-156 µg/ml; 96.3 negative Seeberg, 1985a
mutations), without V79 cells DMSO as solvent
and with activation
Chromosome aberrations human lymphocytes 12.5-50 µg/ml; 96.3 negativea Bootman et al.,
test, without and with DMSO as solvent 1985a
activation
Unscheduled DNA human cells HeLa S3 9.75-156 µg/ml 96.3 negative Seeberg, 1985b
synthesis, without (without act.) or
and with activation 2.44-39 µg/ml (with
act.); solvent DMSO
In vivo
Bone marrow CD-1 mouse 80, 400 and 2000 96.3 negative Bootman et al.,
micronucleus test mg/kg bw, p.o. (single 1985b
dose); sacrifice at 24,
48 and 72 h
a Both the test without activation and the test with activation showed a treatment-related reduction in mitotic index.
Special study on sensitization
Etofenprox (purity not reported) was tested for sensitizing
potential in a group of 20 male English Hartley guinea-pigs using
the maximization test according to Magnusson & Kligman. Treatment
in the induction phase was done with a 20% solution in corn oil.
The second induction application was preceded by a treatment with
10% sodium lauryl sulphate in petrolatum ointment to produce an
irritative response in the induction phase. Etofenprox did not
produce sensitization (Kobayashi, 1985).
Special studies on skin and eye irritation
Skin irritation of etofenprox was studied using a group of 6
Japanese white rabbits. A quantity of 0.5 ml of undiluted
etofenprox (purity 96.3%) was applied as a molten solution (melting
point of compound: 35-38 °C) to the shaven intact skin under
occlusion for 4 hours. Skin observations were made up to day 14
after treatment. Very slight erythema was seen in 1/6 animals up to
day 7. The classification (Draize) based on this result is not
irritating (Kashima, 1985d, 1991a).
Eye irritation of etofenprox was studied using a group of 6
Japanese white rabbits. A quantity of 0.1 ml of undiluted
etofenprox (purity 96.3%) was applied as a molten solution (melting
point of compound: 35-38 °C) through instillation into the
conjunctival sac of the right eye. No washing was done.
Observations were made up to 72 hours after treatment. Mild
conjunctival redness was observed in 6/6 animals; after 72 hours
this effect was no longer present. Mild conjunctival edema was seen
in 1/6 animals, which disappeared after 24 hours. The
classification (Draize) based on this result is not irritating
(Kashima, 1985e; 1991b).
Observation in humans
Health assessments were carried out once or twice each year on
a group of 21 operators engaged in the production of technical
etofenprox for periods ranging from 1.5 to 5.5 years. The
examinations included determination of blood pressure, X-ray
examination, haematology, blood biochemistry, limited urinalysis,
electrocardiography and interview for subjective complaints. No
compound-related effects were observed. The report did not include
data on the exposure concentrations (Yamazaki, 1992).
COMMENTS
After oral administration to rats, total excretion in faeces
was 85-90% and 7-9% in urine. Excretion in bile was found to be 10%
to 30%. Unchanged etofenprox was not found in the bile. Total
retention in the body after 5 days was 3-4%. In the
gastrointestinal tract from 48 to 93% was absorbed. Absorption
tended to be dose-dependent. Tissue concentrations were highest in
fat; this residue was present as unchanged parent compound.
Etofenprox was secreted (as the unchanged compound) in milk. The
major biotransformation routes involve O-de-ethylation of the
ethylphenyl moiety and hydroxylation of the phenoxybenzyl moiety
followed by conjugation with glucuronide or sulfate. Oxidation of
the alpha-CH2 group followed by hydrolysis represents an additional
route. The available results for dogs indicate a lower
gastrointestinal absorption rate than in rats. The major
biotransformation routes were the same as in rats.
Etofenprox has a low acute oral toxicity in mice, rats and
dogs. WHO has classified etofenprox as unlikely to present acute
hazard in normal use.
In a 13-week study in mice, using dietary concentrations of 0,
50, 500, 3000 or 15 000 ppm, the NOAEL was 500 ppm, equal to 60
mg/kg bw/day. The main effects seen were mortality, growth
retardation, increased weights of liver (with enlarged hepatocytes)
and kidneys (with tubular basophilia and dilatation) and decreases
in red blood cell parameters.
In a 13-week study in rats using dietary concentrations of 0,
50, 300, 1800 or 10 800 ppm, the NOAEL was 300 ppm, equal to 20
mg/kg bw/day, based on effects on growth and the liver. In
addition, increased thyroid weight with increased incidence of
microfollicles in this organ was observed.
In a 52-week study in dogs using dietary concentrations of 0,
100, 1000 or 10 000 ppm, the NOAEL was 1000 ppm (equal to 32 mg/kg
bw/day) based on decreased red blood cell parameters, increased
serum alkaline phosphatase and increased liver weight (with swelling
of hepatocytes).
In a two-year toxicity/carcinogenicity study in mice using
dietary concentrations of 0, 30, 100, 700 or 4900 ppm, the NOAEL was
30 ppm (equal to 3.1 mg/kg bw/day) based on an increased incidence
of tubular lesions in the kidneys at > 100 ppm. There was no
evidence of carcinogenicity.
A two-year toxicity/carcinogenicity study in rats also used
dietary concentrations of 0, 30, 100, 700 or 4900 ppm. The NOAEL
was 100 ppm (equal to 3.7 mg/kg bw/day), based on increased weights
of thyroid and kidneys and microscopic liver changes at >700 ppm.
The incidence of cystic follicles in the thyroid was increased at
4900 ppm only. There was an increased incidence of thyroid
follicular adenomas among the 4900 ppm animals which was
statistically significantly increased in females only. The absence
of genotoxicity of etofenprox (see below) in combination with the
observed activation of the thyroid gland, which might be related to
the effects on the liver (the latter probably leading to increased
breakdown of thyroid hormones), is a strong indication for a non-
genotoxic mechanism of induction of the thyroid tumours.
In a two-generation study in rats using dietary concentrations
of 0, 100, 700 or 4900 ppm, the NOAEL was 100 ppm (equivalent to 5
mg/kg bw/day). No effects on reproduction were observed in this
study. Main effects seen in parents as well as young were decreased
growth and effects on the weights and histopathology of liver and
kidneys. The effects on the offspring were consistent with exposure
to unchanged etofenprox via milk.
Embryo/fetotoxicity and teratogenicity were studied in rats (3
studies, segment I, II & III, respectively) and rabbits (1 study).
In the three studies in the rat, etofenprox was administered by
gavage at dose levels of 0, 12.5, 250 or 5000 mg/kg bw/day. In each
of the studies, dose-related maternal toxicity (clinical signs,
growth retardation) was observed at 5000 mg/kg bw/day. In the two
studies in rats with dosing before or during pregnancy, no effects
on offspring/fetuses were seen. In the study in rats with dosing
during lactation (segment III), toxic effects developed in the
offspring, most likely as a result of exposure to etofenprox via
milk. In none of the studies in rats were irreversible structural
malformations found. The NOAEL for maternal or parental toxicity in
each of these studies was 250 mg/kg bw/day. For fetotoxicity, the
NOAEL was 5000 mg/kg bw/day in the segment I and II studies. The
NOAEL for neonatal effects in the segment III study was 250 mg/kg
bw/day. In the rabbit study, the NOAEL for maternal toxicity was 10
mg/kg bw/day, based on decreased growth at 50 and 250 mg/kg bw/day.
Incidences of late abortions and early-embryonal mortality were
increased at 250 mg/kg bw/day only. The NOAEL for embryo/
fetotoxicity was 50 mg/kg bw/day. No irreversible structural
malformations were noted in this study.
Based on the results of the available in vitro and in vivo
genotoxicity data there was no evidence that etofenprox is
genotoxic.
The most sensitive species in the animal studies presently
available appear to be rodents, with NOAELs of 3.1 and 3.7 mg/kg
bw/day for mice and rats, respectively, in the long-term studies.
The ADI was based on the long-term study in mice, using a 100-fold
safety factor.
TOXICOLOGICAL EVALUATION
Level causing no toxicological effects
Mouse: 30 ppm, equal to 3.1 mg/kg bw/day (two-year study)
Rat: 100 ppm, equal to 3.7 mg/kg bw/day (two-year study)
Rabbit: 10 mg/kg bw/day (maternal toxicity in a
teratogenicity study)
Dog: 1000 ppm, equal to 32 mg/kg bw/day (52-week study).
Estimate of acceptable daily intake for humans
0-0.03 mg/kg bw.
Studies which will provide information valuable in the continued
evaluation of the compound
Clarification of the dose-response for thyroid effects in the
rat, including evaluation of T3, T4, TSH and other relevant
parameters.
Observations in humans with adequate information on exposure
levels.
REFERENCES
Agrochemicals Handbook (1991) The Agrochemicals Handbook - Third
Edition. Royal Society of Chemistry -Information Services,
Cambridge, England.
Bootman, J., Hodson-Walker, G. & Dance, C.A. (1985a). In vitro
assessment of the clastogenic activity of MTI-500, ethofenprox, in
cultured human peripheral lymphocytes. Life Science Research
Limited, England; report no. 85/MT0017/430, dated 17 July 1985.
Submitted to WHO by Mitsui Toatsu Chemicals, Inc., Tokyo, Japan.
Bootman, J., Hodson-Walker, G. & Dance, C.A. (1985b). MTI-500,
etofenprox: assessment of clastogenic action on bone marrow
erythrocytes in the micronucleus test. Life Science Research
Limited, England; report no. 85/MT0016/406, dated 3 July 1985.
Submitted to WHO by Mitsui Toatsu Chemicals, Inc., Tokyo, Japan.
Bootman, J. & May, K. (1985). MTI-500alpha-CO: assessment of its
mutagenic potential using amino-acid auxotrophs of Salmonella
typhimurium and Escherichia coli. Life Science Research Limited,
England; report no. 85/MT0020/433, dated 19 July 1985. Submitted to
WHO by Mitsui Toatsu Chemicals, Inc., Tokyo, Japan.
Bottomley, A.M., Barton, S.J., Masters, R.E., Offer, J.M., Parker,
C.A., Anderson, A. & Dawe, I.S. (1985). Effect of etofenprox (MTI-
500) on pregnancy of the rabbit. Huntingdon Research Centre Ltd.,
England; report no. MTC 85(84)/85444, dated 20 December 1985.
Submitted to WHO by Mitsui Toatsu Chemicals, Inc., Tokyo, Japan.
Cozens, D.D., Barton, S.J., Offer, J.M., Parker, C.A. & Anderson, A.
(1985a). Effect of etofenprox (MTI-500) on multiple generations of
the rat. Huntingdon Research Centre Ltd., England; report no. MTC
67/85706, dated 29 October 1985. Submitted to WHO by Mitsui Toatsu
Chemicals, Inc., Tokyo, Japan.
Cozens, D.D., Hughes, E.W. & Anderson, A. (1985b). Effect of
etofenprox (MTI-500) on pregnancy of the rat with rearing to
maturation of the F1 generation. Huntingdon Research Centre Ltd.,
England; report no. MTC 64/85422, dated 28 October 1985. Submitted
to WHO by Mitsui Toatsu Chemicals, Inc., Tokyo, Japan.
Cozens, D.D., Hughes, E.W., Clark, R. & Anderson, A. (1985c). Effect
of etofenprox (MTI-500) on fertility and pregnancy of the rat.
Huntingdon Research Centre Ltd., England; report no. MTC 66/84668,
dated 29 October 1985. Submitted to WHO by Mitsui Toatsu Chemicals,
Inc., Tokyo, Japan.
Cozens, D.D., Hughes, E.W., Offer, J. & Anderson, A. (1985d). Effect
of etofenprox (MTI-500) on the peri and post natal period of the rat
with rearing to maturation of the F1 offspring. Huntingdon Research
Centre Ltd., England; report no. MTC 65/85423, dated 29 October
1985. Submitted to WHO by Mitsui Toatsu Chemicals, Inc., Tokyo,
Japan.
Cummins, H.A. & Gardner, J.R. (1985) MTI-500 alpha-CO: Acute oral
toxicity in the rat. Life Science Research Ltd., England - study no.
85/MT0018/474; report dated 2 August 1985. Submitted to WHO by
Mitsui Toatsu Chemicals, Inc., Tokyo, Japan.
Foster, R. (1985). Reverse mutation in Salmonella typhimurium. Test
substance MTI-500. Life Science Research Roma Toxicology Centre,
Italy; report no. 162001-M-06185, dated 22/8/1985. Submitted to WHO
by Mitsui Toatsu Chemicals, Inc., Tokyo, Japan.
Green, O.P., Heywood, R., Street, A.E., Gopinath, & Almond, R.H.
(1985a). Assessment of the toxicity of MTI-500 to mice by dietary
administration for 13 weeks. Huntingdon Research Centre Ltd.,
England; report no. MTC 55/821112, dated 8 July 1983, re-issued 31
October 1985. Submitted to WHO by Mitsui Toatsu Chemicals, Inc.,
Tokyo, Japan.
Green, O.P., Street, A.E., Heywood, R., Gopinath, C. & Almond, R.H.
(1985b). Assessment of the toxicity of MTI-500 in rats during
dietary administration for 13 weeks. Huntingdon Research Centre
Ltd., England; report no. MTC 56/821067, dated 7 July 1983, re-
issued 18 December 1985. Submitted to WHO by Mitsui Toatsu
Chemicals, Inc., Tokyo, Japan.
Green, O.P., Heaps, C.J., Heywood, R., Street, A.E., Gopinath, C.,
Imm, S. & Gibson, W.A. (1986a). Ethofenprox (MTI-500) potential
tumorigenic and toxic effects in prolonged dietary administration to
mice. Huntingdon Research Centre Ltd., England; report no. MTC
58/85582, dated 6 January 1986, re-issued 24 February 1986.
Submitted to WHO by Mitsui Toatsu Chemicals, Inc., Tokyo, Japan.
Green, O.P., Heaps, C.J., Heywood, R., Street, A.E., Gopinath, C.,
Sing, H. & Gibson, W.A. (1986b). Ethofenprox (MTI-500) potential
tumorigenic and toxic effects in prolonged dietary administration to
rats. Huntingdon Research Centre Ltd., England; report no. MTC
59/85581, dated 24 January 1986. Submitted to WHO by Mitsui Toatsu
Chemicals, Inc., Tokyo, Japan.
Harling, R.J., Burford, P. & Heywood, R. (1985a) Ethofenprox (MTI-
500) Acute limit test of toxicity to dogs following a single oral
administration. Huntingdon Research Centre Ltd., England; report no.
MTC 101/851185, dated 24 October 1985. Submitted to WHO by Mitsui
Toatsu Chemicals, Inc., Tokyo, Japan.
Harling, R.J., Burford, P., Street, A.E., Heywood, R., Majeed, S.R.
& Gopinath, C. (1985b). Ethofenprox (MTI-500) toxicity to dogs by
repeated dietary administration for 52 weeks followed by a recovery
period of 8 weeks. Huntingdon Research Centre Ltd., England; report
no. MTC 71/85234, dated 25 October 1985, re-issued 8 November 1985.
Submitted to WHO by Mitsui Toatsu Chemicals, Inc., Tokyo, Japan.
Hashimoto, K., Ohtaki, T., Yamaguchi, K., Nishijima, Y., Imai, K. &
Azegami, J. (1982a). Report on acute toxicity study of MTI-500
(ethofenprox) in mice. Hatano Research Institute, Japan; report no.
A-82-35~42, dated 9 November 1982. Submitted to WHO by Mitsui Toatsu
Chemicals, Inc., Tokyo, Japan.
Hashimoto, K., Ohtaki, T., Yamada, S., Watanabe, C., Imai, K. &
Azegami, J. (1982b). Report on acute toxicity study of MTI-500
(ethofenprox) in rats. Hatano Research Institute, Japan; report no.
A-82-27~34, dated 9 November 1982. Submitted to WHO by Mitsui Toatsu
Chemicals, Inc., Tokyo, Japan.
Hawkins, D.R., Kirkpatrick, D., Ewen, B., Midgley, I., Biggs, S.R. &
Whitby, B.R. (1985a). The biokinetics and metabolism of 14C-
ethofenprox in the rat. Huntingdon Research Centre Ltd., England;
report no. HRC/MTC 68/84610, dated 1 August 1985. Submitted to WHO
by Mitsui Toatsu Chemicals, Inc., Tokyo, Japan.
Hawkins, D.R., Kirkpatrick, D., Ewen, B., Midgley, I. & Biggs, S.R.
(1985b). The metabolism of 14C-ethofenprox in dogs. Huntingdon
Research Centre Ltd., England; report no. HRC/MTC 69/84583, dated 11
October 1985. Submitted to WHO by Mitsui Toatsu Chemicals, Inc.,
Tokyo, Japan.
Jackson, G.C., Hardy, C.J., Clark, G.C., Gregson, R.L., Lewis, D.J.
& Gopinath, C. (1983). MTI-500 acute inhalation toxicity in rats 4
hour exposure. Huntingdon Research Centre Ltd., England; report no.
MTC 60/821079, dated 19 April 1983 Submitted to WHO by Mitsui Toatsu
Chemicals, Inc., Tokyo, Japan.
Kamiya, J., Yoshiwara, K., Saito, S., Takahashi, Y. Oseki, K.,
Shimizu, H., Kawazura, H., Shiga, Y., Yoshida, M., Hayakawa, M. &
Tanaka Y. (1985). General pharmacology of MTI-500. Institute of
Biological Sciences - Mitsui Pharmaceuticals Inc., [A translation of
an original Japanese report published in Japanese, in: Pharmacology
and Therapeutics, vol.13 no.11, pp. 229-244, November 1985].
Translated by T. Hirakawa.
Kashima, M. (1985a). Acute oral toxicity of MTI-500 emulsion in
mice. Nippon Experimental Medical Research Institute, Ltd., Japan;
report no. NEMRI-H-85-50, dated 2 September 1985. Submitted to WHO
by Mitsui Toatsu Chemicals, Inc., Tokyo, Japan.
Kashima, M. (1985b). Acute oral toxicity of MTI-500 emulsion in
rats. Nippon Experimental Medical Research Institute, Ltd., Japan;
report no. NEMRI-H-85-48, dated 2 September 1985. Submitted to WHO
by Mitsui Toatsu Chemicals, Inc., Tokyo, Japan.
Kashima, M. (1985c). Acute dermal toxicity of MTI-500 emulsion in
rats. Nippon Experimental Medical Research Institute, Ltd., Japan;
report no. NEMRI-H-85-49, dated 2 September 1985. Submitted to WHO
by Mitsui Toatsu Chemicals, Inc., Tokyo, Japan.
Kashima, M. (1985d). MTI-500 primary skin stimulation test in
rabbits. Nippon Experimental Medical Research Institute, Ltd, Japan;
report no. NEMRI-H-85-5, dated 23 August 1985. Submitted to WHO by
Mitsui Toatsu Chemicals, Inc., Tokyo, Japan.
Kashima, M. (1985e). MTI-500 primary ophthalmic stimulation test in
rabbits. Nippon Experimental Medical Research Institute, Ltd, Japan;
report no. NEMRI-H-85-55, dated 24 October 1985. Submitted to WHO by
Mitsui Toatsu Chemicals, Inc., Tokyo, Japan.
Kashima (1991a) Final report modification (I) - study name: MTI-500
primary skin stimulation test in rabbits. Nippon Experimental
Medical Research Institute, Ltd, Japan; project no. NEMRI-H-85-5.
Modification d.d. 28 October 1991. Submitted to WHO by Mitsui Toatsu
Chemicals, Inc., Tokyo, Japan.
Kashima (1991b) Final report modification (I) - study name: MTI-500
primary ophthalmic stimulation test in rabbits. Nippon Experimental
Medical Research Institute, Ltd, Japan; project no. NEMRI-H-85-55.
Modification d.d. 28 October 1991. Submitted to WHO by Mitsui Toatsu
Chemicals, Inc., Tokyo, Japan.
Kobayashi, K. (1985). MTI-500 skin sensitization test in guinea-
pigs. Nippon Experimental Medical Research Institute, Ltd, Japan;
report dated 31 October 1985. Submitted to WHO by Mitsui Toatsu
Chemicals, Inc., Tokyo, Japan.
Powell, A.J., Coleman, M., Crook, D., Gopinath, C., Gobson, W.A.,
Read, R.M. & Anderson, A. (1988) MTI-500 alpha-CO: Toxicity to rats
by dietary administration for 13 weeks (Final report). Huntingdon
Research Centre Ltd., England; report no. MTC 141/871448, dated 15
March 1988. Submitted to WHO by Mitsui Toatsu Chemicals, Inc.,
Tokyo, Japan.
Seeberg, A.H. (1985a). Gene mutation in Chinese Hamster V79 cells.
Test substance MTI-500. Life Science Research Roma Toxicology
Centre, Italy; report no. 162002-M-06985, dated 22/8/1985. Submitted
to WHO by Mitsui Toatsu Chemicals, Inc., Tokyo, Japan.
Seeberg, A.H. (1985b). Unscheduled DNA synthesis in human cells,
cell line: Hela S3. Test substance MTI-500. Life Science Research
Roma Toxicology Centre, Italy; report no. 162003-M-05785, dated
30/7/1985. Submitted to WHO by Mitsui Toatsu Chemicals, Inc., Tokyo,
Japan.
Tomoda, K., Arai, M. & Ohnuma, K. (1986). Metabolism study of
ethofenprox (MTI-500). 6. Metabolism in rat. Chiba Branch
Laboratory, Agrochemicals Laboratory - Mitsui Toatsu Chemicals,
Inc., Japan; report dated July 1986. Submitted to WHO by Mitsui
Toatsu Chemicals, Inc., Tokyo, Japan.
WHO (1992). The WHO recommended classification of pesticides by
hazard and guidelines to classification 1992-1993 (WHO/PCS/92.14).
Available from the International Programme on Chemical Safety, World
Health Organization, Geneva, Switzerland.
Yamazaki, Y. (1992). Health report from the Industrial Hygiene
Section, Ohmuta Factory - Mitsui Toatsu Chemicals, Inc., dated 17
April 1992. Submitted to WHO by Mitsui Toatsu Chemicals, Inc.,
Tokyo, Japan.
 Table 1. Acute toxicity of etofenprox
Species Sex Route LD50 LC50 Purity Reference
(g/kg bw) (g/m3)
Mouse M&F oral > 107.2 96%a Hashimoto et al., 1982a
M&F dermalb > 2.14 96%a Hashimoto et al., 1982a
M i.p. > 53.6 96%a Hashimoto et al., 1982a
F i.p. 13.4 96%a Hashimoto et al., 1982a
M&F s.c. > 53.6 96%a Hashimoto et al., 1982a
Rat M&F oral > 42.88 96%a Hashimoto et al., 1982b
M&F dermalb > 2.14 96%a Hashimoto et al., 1982b
M&F i.p. > 42.88 96%a Hashimoto et al., 1982b
M&F s.c. > 32.16 96%a Hashimoto et al., 1982b
M&F inhalc > 5.9 96%d Jackson et al., 1983
Dog M&F oral > 5.0 96.3%e Harling et al., 1985a
a compound given as undiluted molten solution (40 °C)
b exposure for 24 hours
c exposure for 4 hours
d compound given as aerosol in acetone
e compound given in gelatine capsules
Table 2. Study results - pharmacological/toxicological action (Kamiya et al., 1985)
Parameter Species (sex) Route Result
Central nervous system
- spontaneous motor activity DDY mouse (M) oral dose-related decrease at 25 & 50 g/kg bw (no other
levels tested)
- thiopental-induced sleeping time DDY mouse (M) oral dose-related increase at 25 & 50 g/kg bw; no effect
at 12.5 g/kg bw
- muscle relaxation actiona DDY mouse (M) oral affected at 50 g/kg bw; no effect at 5 g/kg bw
- body (rectum) temperature DDY mouse (M) oral no effect at 25 and 50 g/kg bw
- induced tonic and clonic DDY mouse (M) oral no effect at 5 & 50 g/kg bw
convulsionsb
- general behaviour Wistar rat (M) oral no effect at 1 & 10 g/kg bw
- EEG of frontal lobe Wistar rat (M) oral dose-related effect at 1 & 10 g/kg bw (no other
levels tested)
- EEG of hippocampus Wistar rat (M) oral effect at 10 g/kg bw; no effect at 1 g/kg bw
& parietal lobe
- conscious reaction induced Wistar rat (M) oral no effect at 1 & 10 g/kg bw
by sound stimulation
- spinal reflex potential mongrel cat (M/F) i.d.c no effect at 0.125-1 g/kg bw (cumulative administration)
Effect on smooth muscle
- intestinal charcoal propulsion DDY mouse (M) oral dose-related increase at 25 & 50 g/kg bw; no effect
at 12.5 g/kg bw
- normal movement and contractile rat, in vitro - 0.01-1.0 mmol/l (cumulative administration)
- responsed of vas deferens produced no effect
- autonomous movement of gravid rat. in vitro - 0.001-1.0 mmol/l (cumulative administration)
or non-gravid uteruse produced no effect
- uterus contractabilityf rat, in vitro - no effect at 0.1 mmol/l
- spontaneous movement rabbit, in vitro - 0.01-1.0 mmol/l (cumulative administration)
& contraction of ileum produced no effect
- spontaneous movement of ileum guinea-pig, - 0.001-1.0 mmol/l (cumulative administration)
in vitro produced no effect
Table 2 (contd)
Parameter Species (sex) Route Result
neuro-muscular junction
- contraction of musculus Wistar rat (M) i.v. no effect at 12.5-100 mg/kg bw
gastrocnemiusg
autonomic ganglia
- contractions of nictitating mongrel cat (M/F) i.v. no effect at 10-100 mg/kg bw
membraneh
respiratory and circulatory system
- respiration rate, blood pressure mongrel dog (M/F) i.v. decreased respiration rate (followed by increased rate)
and ECG in combination with increased blood pressure & heart rate
at > 30 mg/kg bw; at 100 mg/kg bw severer effects; no
effect at 10 mg/kg bw
- cardiovascular response as mongrel dog (M/F) i.v. no effect at 100 mg/kg bw
induced by NE, ISP, AC, HIi
& bilateral carotid occlusion
- contractility of isolated atrium guinea-pig, in vitro -0.01-1.0 mmol/l (cumulative administration) produced no
effect
- positive & negative inotropic guinea-pig, in vitro -0.01-1.0 mmol/l (cumulative administration)
reaction of isolated atrium produced no effect on induced neg.
as induced by ISP & HI (+) or inotropy; effect on pos. inotropy
AC (-) at 1.0 mmol/l only (no effect at 0.01-0.1 mmol/l)
urinary volume & components
- measured: volume, Cl-, Na+ & K+ Wistar rat (M) oral dose-related decrease in volume, Cl- & Na+ at 10 & 20 g/kg
bw (no other dose levels tested), no effect on K+
Table 2 (contd)
Parameter Species (sex) Route Result
serum components
- standard blood biochemistry Wistar rat (M) oral increased glucose (after 1 h only) and GOT and GPT
after 1, 2, 6 & 24 hours (all sampling times) at 10 & 20 g/kg bw (no other
levels tested)
blood coagulation
- prothrombin time, partial Wistar rat (M) oral no effect at 10 & 20 g/kg bw
thromboplastin time & fibrinogen
after 1, 2, 6 & 24 hours
a measured as incidence of slipping down a smooth slope
b induced by pentetrazol, strychnine or electro-shock
c i.d. = intraduodenal
d contractile response induced by di-norepinephrine
e from rats in estrus and diestrus cycle or in pregnant stage
f induced by oxytocin
g induced by electrical stimulation of sciatic nerve and muscle
h as induced by pre- and post-ganglionic nerve stimulation
i NE, norepinephrine; ISP, isoproterenol; AC, acetylcholine; HI, histamine
compound-related since the same effects were also seen (more severe
in degree) at 15 000 ppm (the observed decreases were 8.5-11% at 15
000 ppm and 6% in males at 3000 ppm). The NOAEL in this study was
500 ppm (equal to 60 mg/kg bw/day) (Green et al., 1985a).
Rats
Groups of 20 Sprague-Dawley rats/sex were fed diets containing
0, 50, 300, 1800 or 10 800 ppm etofenprox (purity 96%) for 13 weeks.
These dose levels were equal to 3.3, 20, 120 or 734 mg/kg bw/day in
males and 3.8, 23, 142 or 820 mg/kg bw/day in females. Clinical
signs, mortality, food consumption and body weight were recorded.
Haematology, clinical chemistry (including T3 and T4) and
urinalysis were done in all groups at weeks 6 and 12. Additional
haematology consisted of the determination of PT and PTT in the 0
and 10 800 ppm groups at weeks 0 and 13. The weights of 10
organs/animal were recorded. Gross pathology and histopathology
(about 27 tissues/animal) were carried out. At 10 800 ppm the
following effects were observed: slight growth retardation,
decreased food utilization, increased thrombin time, PT and PTT,
decreased T4, increased cholesterol, increased ASAT, ALAT and LDH
(at week 6 only), increased liver weight with slight hepatocyte
enlargement and increased thyroid weight with increased incidence of
microfollicles in thyroids. These effects, except for the increase
in thrombin time (PTT and PT not determined), were also present to a
slight degree at 1800 ppm. The NOAEL in this study was 300 ppm
(equal to 20 mg/kg bw/day) (Green et al., 1985b).
Dogs
Groups of 6 beagle dogs/sex were given diets containing 0 or 10
000 ppm etofenprox (purity 96.3%) for 52 weeks. Two additional
groups of 4 dogs/sex received dietary dose levels of 100 or 1000 ppm
for 52 weeks. These dose levels were equal to 3.5, 33, or 352 mg/kg
bw/day in males and 3.2, 32 or 339 mg/kg bw/day in females. After
52 weeks, 4 dogs/sex/group were killed. The remaining dogs in the 0
and 10 000 ppm groups were killed after a recovery period of 8
weeks. Observations included clinical signs, mortality, food
consumption, body weight, haematology (including PT and PTT),
clinical chemistry and urinalysis. The weights of 10 organs/animal
were recorded. Gross pathology and histopathology (about 33
tissues/animal) were carried out. Effects observed at 10 000 ppm
were decreased PCV, Hb and RBC, increased SAP, decreased total
protein and albumin, increased kidney weight and increased liver
weight with minimal swelling of hepatocytes. After 8 weeks of
recovery none of these effects were found to persist. The NOAEL in
this study was 1000 ppm (equal to 32 mg/kg bw/day) (Harling et al.,
1985b).
Long-term toxicity/carcinogenicity studies
Mice
Groups of 52 CD-1 mice/sex were fed diets containing 0, 30,
100, 700 or 4900 ppm etofenprox (purity 96.3%) for 2 years. These
dose levels were equal to 3.1, 10, 75 or 550 mg/kg bw/day in males
and 3.6, 12, 81 or 615 mg/kg bw/day in females. Satellite groups of
24 mice/sex/group received the same dose levels and were used for
interim sacrifices after 26 weeks (10 animals/sex per group) and 52
weeks (remaining satellite animals). Observations included clinical
signs, mortality, food consumption, body weight, haematology,
clinical chemistry and urinalysis. Ophthalmoscopy was done in the 0
and 4900 ppm groups. The weights of 10 organs/animal were recorded.
Gross pathology and histopathology (about 30 tissues/animal) were
carried out. At 4900 ppm the following effects were seen: increased
mortality in males (survival at test end 10/52 versus 24/52 in
controls; renal lesions were considered contributory to death),
growth retardation, decreased Hb, RBC, MCHC and PCV in combination
with increased MCV (first year only), increased platelet counts
(second year), decreased urine specific gravity, and increased
weights of liver and spleen. Decreased MCHC and increased MCV were
seen at 700 ppm also. Histopathology showed an increased incidence
of renal changes in treated animals, mainly in males. The renal
changes were mostly tubular lesions appearing as groups of
basophilic or dilated tubules, sometimes associated with focal loss
of tubules. These lesions were scored on a grade 1 to 5 scale. The
incidences are presented in Table 3.
At 30 ppm the increase in the incidence of renal lesions was
considered marginal. No increase in tumour incidence was found.
The NOAEL in this study was 30 ppm (equal to 3.1 mg/kg bw/day)
(Green et al., 1986a).
Rats
Groups of 70 Sprague-Dawley rats/sex were fed diets containing
0, 30, 100, 700 or 4900 ppm etofenprox (purity 96.3%) for 2 years.
These dose levels were equal to 1.1, 3.7, 26 or 187 mg/kg bw/day in
males and 1.4, 4.8, 34 or 249 mg/kg bw/day in females. Interim
sacrifices were made after 26 or 52 weeks (10 animals/sex/group on
both occasions). Observations included clinical signs, mortality,
food consumption, body weight, haematology, clinical chemistry
(including T3 and T4) and urinalysis. The weights of 10
organs/animal were recorded. Gross pathology and histopathology
(about 30 tissues/animal) were carried out. At 4900 ppm, the
following effects were seen: decreased food intake or utilization,
decreased growth, slightly increased thrombin time, increased
urinary protein, increased liver weight with hepatocyte enlargement
(sometimes with vacuolization), slightly increased kidney weight and
increased thyroid weight with increased incidence of cystic
follicles in thyroid. At 700 ppm, decreased food intake, increased
thyroid weight, slightly increased kidney weight and presence of
eosinophilic hepatocytes (sometimes with vacuolization) were noted.
At 100 ppm no toxic effects were observed. Increased incidence of
thyroid follicular tumours was found (Table 4). No changes in
concentrations of T3 and T4 were found.
Statistical analysis of these incidences showed that for
thyroid follicular cell carcinomas the trend test was negative and
the intergroup comparison showed no significant effect. Intergroup
comparison showed a statistically significant increase in adenomas
at 4900 ppm in females only. For adenomas and carcinomas, the trend
test was positive in males and females. The NOAEL in this study was
100 ppm (equal to 3.7 mg/kg bw/day) (Green et al., 1986b).
Reproduction studies
Rats
In a two-generation study, groups of 24 or 28 Sprague-Dawley
rats/sex were used as parent animals, receiving dietary
concentrations of 0, 100, 700 or 4900 ppm etofenprox (purity 96.3%).
Two litters per generation were produced. Parent animals were
killed at day 21 after birth of the Fb litters. Pups were also
sacrificed at day 21 after birth except for selected F1a pups
(maintained on test diets up to day 21 after birth of F1b litters),
selected F1b pups (used as F1 parents) and selected F2b pups
(maintained on test diets up to week 13 after birth). Gross
pathology was done and the weights of selected organs were measured.
Histopathology was done in F1 parents (kidneys, thyroids, liver),
F0 parents (kidneys) and F2b adult animals (thyroid). At 4900 ppm,
the main effects were: decreased growth and increased water
consumption (parent animals), increased kidney weights with presence
of cysts, deposits, congestion, haemorrhages or inflammation in
collecting ducts or medulla (parents and young), increased liver
weights with hepatocyte enlargement (parents and young), decreased
pup weights, clinical signs (tremors, abnormal gait) in pups and
increased thyroid weight with slight increase in height of
follicular epithelium (F1 parents). At 700 ppm the effects were:
increased kidney weights in F2b adult females and increased liver
weights in the young of both generations. No effect on reproduction
parameters were observed. The NOAEL in this study was 100 ppm
(equivalent to 5 mg/kg bw/day) (Cozens et al., 1985a).
Table 3. Number of mice showing renal lesions
(Green et al., 1986a)
Dilated/basophilic Control 30 ppm 100 ppm 700 ppm 4900 ppm
cortical tubules
Malesa
grade 1 7 8 6 9 5
grade 2 0 3 4 6 4
grade 3 0 0 1 1 11
grade 4 0 0 1 1 6
grade 5 0 0 0 0 4
Femalesa
grade 1 3 0 5 6 7
grade 2 1 1 1 1 6
grade 3 0 0 0 2 6
grade 4 0 0 1 0 1
grade 5 0 0 0 0 0
a number of animals examined: 52 in all groups
Table 4. Number of rats with thyroid follicular tumours
(Green et al., 1986b)
Tumour type Control 30 ppm 100 ppm 700 ppm 4900 ppm
Malesa
adenomas 6 6 4 5 11
carcinomas 0 0 1 3 2
adenomas and/or 6 6 5 8 13
carcinomas
Femalesa
adenomas 0 3 2 0 9
carcinomas 0 0 0 2 1
adenomas and/or 0 3 2 2 9
carcinomas
a number of animals examined: 50 in all groups
Special studies on embryo/fetotoxicity
Rats
In a segment I study, groups of 24 Sprague-Dawley rats/sex were
given oral doses (by gavage) of 0, 12.5, 250 or 5000 mg/kg bw/day
etofenprox (purity 96.3%) and were mated during a 14-day mating
period. The male animals were treated for 9 weeks prior to mating
up to termination (autopsy) at week 15. Female animals were dosed
for 2 weeks prior to mating up to day 7 of gestation. At day 20 of
gestation females were sacrificed, standard litter data were
recorded and fetuses were monitored for abnormalities (externally
and standard soft tissue and skeletal examinations). No effects on
mating performance, litter parameters, incidences of soft tissue or
skeletal abnormalities were found. In parent animals at 5000 mg/kg
bw/day salivation with brown staining around the mouth,
staining/wetness around the anogenital region, crystalline
appearance of the faeces, unkempt hair coat (males only) and skin
lesions (males only) were present. Salivation with brown staining
around the mouth was also observed occasionally at 250 mg/kg bw/day
and, in a few animals, at 12.5 mg/kg bw/day. Increased salivation
is frequently observed in gavage studies and is probably due to the
method of application rather than representing a compound-related
toxic effect. The NOAEL for reproductive performance and
fetotoxicity was 5000 mg/kg bw/day. The NOAEL for parental toxicity
was 250 mg/kg bw/day (Cozens et al., 1985c).
In a segment II study, groups of 35 pregnant Sprague-Dawley
rats were given oral doses (by gavage) of 0, 12.5, 250 or 5000 mg/kg
bw/day etofenprox (purity 96.3%) from days 6 through 17 of
pregnancy. No test compound was administered at later stages of the
study. On day 20 of gestation, 21-24 dams/group were sacrificed for
a standard teratogenicity evaluation (i.e. number of corpora lutea,
number and distribution of live and dead fetuses, fetal weights,
external abnormalities, soft tissue examination, skeletal
examination). The remaining dams were allowed to deliver F1 pups.
Litter data were recorded at birth. In the 4-21 days period after
birth, a number of neuromotor parameters (including several reflexes
and eye opening) were determined. On day 21, one pup/sex/litter was
selected for use as F1 parents; pups not selected for later mating
were killed and examined internally and externally for
abnormalities. Mating was done when these animals were 12 weeks
old. Behavioural tests were performed prior to mating. At day 21
after birth of F2 pups all animals were killed and examined
internally and externally for abnormalities. At 5000 mg/kg bw/day
maternal growth was retarded. Salivation, red/brown staining around
the mouth, wet and yellow staining of the fur in the anogenital
region were seen in the 5000 mg/kg bw/day dams during the treatment
period. At 250 and 12.5 mg/kg bw/day salivation and red/brown
staining around the mouth occurred in lesser incidences; these
effects are not considered as compound-related adverse effects. No
effects on mating performance, litter parameters, incidences of soft
tissue or skeletal abnormalities were found in any group. The NOAEL
for fetotoxicity was 5000 mg/kg bw/day and the NOAEL for maternal
toxicity was 250 mg/kg bw/day (Cozens et al., 1985b).
In a segment III study, groups of 25 pregnant Sprague-Dawley
rats were given oral doses (by gavage) of 0, 12.5, 250 or 5000 mg/kg
bw/day etofenprox (purity 96.3%) from day 17 of pregnancy through
day 21 after birth of the F1 pups. No test compound was
administered at later stages of the study. Litter data were
recorded at birth of the F1 pups. On day 4, pups were culled to
8/litter (pups not selected were discarded). In the 4-21 days
period after birth a number of neuromotor parameters (including
several reflexes and eye opening) were determined. On day 21, one
pup/sex/litter was selected for later mating (parent animals and
pups not selected were killed and examined internally and
externally). Mating was done when these animals were about 12 weeks
old. Behavioural tests were performed prior to mating (i.e. general
aspects of behaviour, hole board test for mobility and
inquisitivity, inclined-plane test for motor coordination and a one-
trial avoidance test). At day 21 after birth of F2 pups all
animals were killed and examined internally and externally for
abnormalities. In dams at 5000 mg/kg bw/day, decreased growth
during the treatment phase and clinical signs (salivation,
occasional brown staining around the mouth, staining around the
urogenital region) were observed. Only salivation and brown
staining were observed at 250 mg/kg bw/day; these effects are not
considered as compound-related adverse effects. At 12.5 mg/kg
bw/day no effects in dams were found. At 5000 mg/kg bw/day only, in
F1 pups increased mortality (preceding symptoms: body tremors and
haemorrhages around the nose) and decreased growth were found in the
period up to day 21 after birth. Also at 5000 mg/kg bw/day, only in
F1 weanlings and F1 adults, enlarged kidneys and histopathological
kidney changes were observed. The NOAEL for neonatal effects and
maternal toxicity was 250 mg/kg bw/day (Cozens et al., 1985d).
Rabbits
Groups of 16 or 17 mated New Zeeland white rabbits were given
oral doses (by gavage) of 0, 10, 50 or 250 mg/kg bw/day etofenprox
(purity 96.3%) from day 6 through 18 of gestation. On day 29 all
dams were sacrificed and examined according to standard procedures
(litter data, soft tissue examination, skeletal examination).
Slight body-weight loss was seen in dams at 250 mg/kg bw/day and
growth retardation at 50 mg/kg bw/day. At 250 mg/kg bw/day, the
incidences of late abortions and early embryonic deaths were
increased. At 50 mg/kg bw/day no effect on fetuses was found. The
NOAEL for embryo/fetotoxicity was 50 mg/kg bw/day. The NOAEL for
maternal toxicity was 10 mg/kg bw/day (Bottomley et al., 1985).
Special studies on genotoxicity
Etofenprox has been examined for genotoxicity in bacteria,
mammalian cells in vitro and in mice in vivo (micronucleus
test). In addition a test for UDS in human HeLa cells in vitro
has been carried out. The results are presented in Table 5. The
results were negative in all test systems.
Special studies on the metabolite 2-(4-ethoxyphenyl)-
2-methylpropyl-3-phenoxybenzoate (MTI-500 alpha-CO)
The oral LD50 of MTI-500 alpha-CO in male and female Charles
River CD rats was >5000 mg/kg bw (Cummins & Gardner, 1985).
Groups of 10 Sprague-Dawley rats/sex were fed diets containing
0, 50, 700 or 10 000 ppm MTI-500 alpha-CO (purity 97.6%) for 13
weeks. These dose levels were equal to 3.8, 54 or 800 mg/kg bw/day
in males and 4.7, 64 or 930 mg/kg bw/day in females. Clinical
signs, mortality, food consumption and body weights were recorded.
Haematology, clinical chemistry, T3, T4 and urinalysis were done
in all groups at week 13. At termination the weights of 10
organs/animal were recorded. Gross pathology was done in all
animals. Histopathology was done in about 27 tissues/animal in the
0 and 10 000 ppm groups and in lungs, liver and kidneys of the
intermediate groups. At 10 000 ppm the following effects were
observed: growth retardation, increased total WBC (females only),
increased SAP and SGOT, decreases in T4, serum cholesterol and
serum globulin, increased relative weights of liver (females only),
kidneys and thymus (females only), histopathological changes in
kidneys (hypertrophy of the tubular epithelium of the pars recta) in
4/10 females. At the lower dose levels no effects were observed.
The NOAEL in this study was 700 ppm (equal to 54 mg/kg bw/day)
(Powell et al., 1988).
The mutagenic potential of MTI-500 alpha-CO (purity 99.6%) was
examined in the Ames test using Salmonella typhimurium tester
strains TA 98, 100, 97a, 1535, 1537 and 102 without and with
metabolic activation. In addition, Escherichia coli strain WP2
uvra was also used as test organism (without and with activation).
The concentration range was 50-5000 µg/plate. The results were
negative in all test organisms (Bootman & May, 1985).
Table 5. Results of genotoxicity assays with etofenprox
Test system Test object Concentration Purity Results References
of etofenprox (%)
In vitro
Ames test, without S. typhimurium 200-3200 µg/plate; 96.3 negative Foster, 1985
and with activation TA100, TA98, TA1535, DMSO as solvent
TA1537, TA1538
HGPRT (gene Chinese hamster 9.75-156 µg/ml; 96.3 negative Seeberg, 1985a
mutations), without V79 cells DMSO as solvent
and with activation
Chromosome aberrations human lymphocytes 12.5-50 µg/ml; 96.3 negativea Bootman et al.,
test, without and with DMSO as solvent 1985a
activation
Unscheduled DNA human cells HeLa S3 9.75-156 µg/ml 96.3 negative Seeberg, 1985b
synthesis, without (without act.) or
and with activation 2.44-39 µg/ml (with
act.); solvent DMSO
In vivo
Bone marrow CD-1 mouse 80, 400 and 2000 96.3 negative Bootman et al.,
micronucleus test mg/kg bw, p.o. (single 1985b
dose); sacrifice at 24,
48 and 72 h
a Both the test without activation and the test with activation showed a treatment-related reduction in mitotic index.
Special study on sensitization
Etofenprox (purity not reported) was tested for sensitizing
potential in a group of 20 male English Hartley guinea-pigs using
the maximization test according to Magnusson & Kligman. Treatment
in the induction phase was done with a 20% solution in corn oil.
The second induction application was preceded by a treatment with
10% sodium lauryl sulphate in petrolatum ointment to produce an
irritative response in the induction phase. Etofenprox did not
produce sensitization (Kobayashi, 1985).
Special studies on skin and eye irritation
Skin irritation of etofenprox was studied using a group of 6
Japanese white rabbits. A quantity of 0.5 ml of undiluted
etofenprox (purity 96.3%) was applied as a molten solution (melting
point of compound: 35-38 °C) to the shaven intact skin under
occlusion for 4 hours. Skin observations were made up to day 14
after treatment. Very slight erythema was seen in 1/6 animals up to
day 7. The classification (Draize) based on this result is not
irritating (Kashima, 1985d, 1991a).
Eye irritation of etofenprox was studied using a group of 6
Japanese white rabbits. A quantity of 0.1 ml of undiluted
etofenprox (purity 96.3%) was applied as a molten solution (melting
point of compound: 35-38 °C) through instillation into the
conjunctival sac of the right eye. No washing was done.
Observations were made up to 72 hours after treatment. Mild
conjunctival redness was observed in 6/6 animals; after 72 hours
this effect was no longer present. Mild conjunctival edema was seen
in 1/6 animals, which disappeared after 24 hours. The
classification (Draize) based on this result is not irritating
(Kashima, 1985e; 1991b).
Observation in humans
Health assessments were carried out once or twice each year on
a group of 21 operators engaged in the production of technical
etofenprox for periods ranging from 1.5 to 5.5 years. The
examinations included determination of blood pressure, X-ray
examination, haematology, blood biochemistry, limited urinalysis,
electrocardiography and interview for subjective complaints. No
compound-related effects were observed. The report did not include
data on the exposure concentrations (Yamazaki, 1992).
COMMENTS
After oral administration to rats, total excretion in faeces
was 85-90% and 7-9% in urine. Excretion in bile was found to be 10%
to 30%. Unchanged etofenprox was not found in the bile. Total
retention in the body after 5 days was 3-4%. In the
gastrointestinal tract from 48 to 93% was absorbed. Absorption
tended to be dose-dependent. Tissue concentrations were highest in
fat; this residue was present as unchanged parent compound.
Etofenprox was secreted (as the unchanged compound) in milk. The
major biotransformation routes involve O-de-ethylation of the
ethylphenyl moiety and hydroxylation of the phenoxybenzyl moiety
followed by conjugation with glucuronide or sulfate. Oxidation of
the alpha-CH2 group followed by hydrolysis represents an additional
route. The available results for dogs indicate a lower
gastrointestinal absorption rate than in rats. The major
biotransformation routes were the same as in rats.
Etofenprox has a low acute oral toxicity in mice, rats and
dogs. WHO has classified etofenprox as unlikely to present acute
hazard in normal use.
In a 13-week study in mice, using dietary concentrations of 0,
50, 500, 3000 or 15 000 ppm, the NOAEL was 500 ppm, equal to 60
mg/kg bw/day. The main effects seen were mortality, growth
retardation, increased weights of liver (with enlarged hepatocytes)
and kidneys (with tubular basophilia and dilatation) and decreases
in red blood cell parameters.
In a 13-week study in rats using dietary concentrations of 0,
50, 300, 1800 or 10 800 ppm, the NOAEL was 300 ppm, equal to 20
mg/kg bw/day, based on effects on growth and the liver. In
addition, increased thyroid weight with increased incidence of
microfollicles in this organ was observed.
In a 52-week study in dogs using dietary concentrations of 0,
100, 1000 or 10 000 ppm, the NOAEL was 1000 ppm (equal to 32 mg/kg
bw/day) based on decreased red blood cell parameters, increased
serum alkaline phosphatase and increased liver weight (with swelling
of hepatocytes).
In a two-year toxicity/carcinogenicity study in mice using
dietary concentrations of 0, 30, 100, 700 or 4900 ppm, the NOAEL was
30 ppm (equal to 3.1 mg/kg bw/day) based on an increased incidence
of tubular lesions in the kidneys at > 100 ppm. There was no
evidence of carcinogenicity.
A two-year toxicity/carcinogenicity study in rats also used
dietary concentrations of 0, 30, 100, 700 or 4900 ppm. The NOAEL
was 100 ppm (equal to 3.7 mg/kg bw/day), based on increased weights
of thyroid and kidneys and microscopic liver changes at >700 ppm.
The incidence of cystic follicles in the thyroid was increased at
4900 ppm only. There was an increased incidence of thyroid
follicular adenomas among the 4900 ppm animals which was
statistically significantly increased in females only. The absence
of genotoxicity of etofenprox (see below) in combination with the
observed activation of the thyroid gland, which might be related to
the effects on the liver (the latter probably leading to increased
breakdown of thyroid hormones), is a strong indication for a non-
genotoxic mechanism of induction of the thyroid tumours.
In a two-generation study in rats using dietary concentrations
of 0, 100, 700 or 4900 ppm, the NOAEL was 100 ppm (equivalent to 5
mg/kg bw/day). No effects on reproduction were observed in this
study. Main effects seen in parents as well as young were decreased
growth and effects on the weights and histopathology of liver and
kidneys. The effects on the offspring were consistent with exposure
to unchanged etofenprox via milk.
Embryo/fetotoxicity and teratogenicity were studied in rats (3
studies, segment I, II & III, respectively) and rabbits (1 study).
In the three studies in the rat, etofenprox was administered by
gavage at dose levels of 0, 12.5, 250 or 5000 mg/kg bw/day. In each
of the studies, dose-related maternal toxicity (clinical signs,
growth retardation) was observed at 5000 mg/kg bw/day. In the two
studies in rats with dosing before or during pregnancy, no effects
on offspring/fetuses were seen. In the study in rats with dosing
during lactation (segment III), toxic effects developed in the
offspring, most likely as a result of exposure to etofenprox via
milk. In none of the studies in rats were irreversible structural
malformations found. The NOAEL for maternal or parental toxicity in
each of these studies was 250 mg/kg bw/day. For fetotoxicity, the
NOAEL was 5000 mg/kg bw/day in the segment I and II studies. The
NOAEL for neonatal effects in the segment III study was 250 mg/kg
bw/day. In the rabbit study, the NOAEL for maternal toxicity was 10
mg/kg bw/day, based on decreased growth at 50 and 250 mg/kg bw/day.
Incidences of late abortions and early-embryonal mortality were
increased at 250 mg/kg bw/day only. The NOAEL for embryo/
fetotoxicity was 50 mg/kg bw/day. No irreversible structural
malformations were noted in this study.
Based on the results of the available in vitro and in vivo
genotoxicity data there was no evidence that etofenprox is
genotoxic.
The most sensitive species in the animal studies presently
available appear to be rodents, with NOAELs of 3.1 and 3.7 mg/kg
bw/day for mice and rats, respectively, in the long-term studies.
The ADI was based on the long-term study in mice, using a 100-fold
safety factor.
TOXICOLOGICAL EVALUATION
Level causing no toxicological effects
Mouse: 30 ppm, equal to 3.1 mg/kg bw/day (two-year study)
Rat: 100 ppm, equal to 3.7 mg/kg bw/day (two-year study)
Rabbit: 10 mg/kg bw/day (maternal toxicity in a
teratogenicity study)
Dog: 1000 ppm, equal to 32 mg/kg bw/day (52-week study).
Estimate of acceptable daily intake for humans
0-0.03 mg/kg bw.
Studies which will provide information valuable in the continued
evaluation of the compound
Clarification of the dose-response for thyroid effects in the
rat, including evaluation of T3, T4, TSH and other relevant
parameters.
Observations in humans with adequate information on exposure
levels.
REFERENCES
Agrochemicals Handbook (1991) The Agrochemicals Handbook - Third
Edition. Royal Society of Chemistry -Information Services,
Cambridge, England.
Bootman, J., Hodson-Walker, G. & Dance, C.A. (1985a). In vitro
assessment of the clastogenic activity of MTI-500, ethofenprox, in
cultured human peripheral lymphocytes. Life Science Research
Limited, England; report no. 85/MT0017/430, dated 17 July 1985.
Submitted to WHO by Mitsui Toatsu Chemicals, Inc., Tokyo, Japan.
Bootman, J., Hodson-Walker, G. & Dance, C.A. (1985b). MTI-500,
etofenprox: assessment of clastogenic action on bone marrow
erythrocytes in the micronucleus test. Life Science Research
Limited, England; report no. 85/MT0016/406, dated 3 July 1985.
Submitted to WHO by Mitsui Toatsu Chemicals, Inc., Tokyo, Japan.
Bootman, J. & May, K. (1985). MTI-500alpha-CO: assessment of its
mutagenic potential using amino-acid auxotrophs of Salmonella
typhimurium and Escherichia coli. Life Science Research Limited,
England; report no. 85/MT0020/433, dated 19 July 1985. Submitted to
WHO by Mitsui Toatsu Chemicals, Inc., Tokyo, Japan.
Bottomley, A.M., Barton, S.J., Masters, R.E., Offer, J.M., Parker,
C.A., Anderson, A. & Dawe, I.S. (1985). Effect of etofenprox (MTI-
500) on pregnancy of the rabbit. Huntingdon Research Centre Ltd.,
England; report no. MTC 85(84)/85444, dated 20 December 1985.
Submitted to WHO by Mitsui Toatsu Chemicals, Inc., Tokyo, Japan.
Cozens, D.D., Barton, S.J., Offer, J.M., Parker, C.A. & Anderson, A.
(1985a). Effect of etofenprox (MTI-500) on multiple generations of
the rat. Huntingdon Research Centre Ltd., England; report no. MTC
67/85706, dated 29 October 1985. Submitted to WHO by Mitsui Toatsu
Chemicals, Inc., Tokyo, Japan.
Cozens, D.D., Hughes, E.W. & Anderson, A. (1985b). Effect of
etofenprox (MTI-500) on pregnancy of the rat with rearing to
maturation of the F1 generation. Huntingdon Research Centre Ltd.,
England; report no. MTC 64/85422, dated 28 October 1985. Submitted
to WHO by Mitsui Toatsu Chemicals, Inc., Tokyo, Japan.
Cozens, D.D., Hughes, E.W., Clark, R. & Anderson, A. (1985c). Effect
of etofenprox (MTI-500) on fertility and pregnancy of the rat.
Huntingdon Research Centre Ltd., England; report no. MTC 66/84668,
dated 29 October 1985. Submitted to WHO by Mitsui Toatsu Chemicals,
Inc., Tokyo, Japan.
Cozens, D.D., Hughes, E.W., Offer, J. & Anderson, A. (1985d). Effect
of etofenprox (MTI-500) on the peri and post natal period of the rat
with rearing to maturation of the F1 offspring. Huntingdon Research
Centre Ltd., England; report no. MTC 65/85423, dated 29 October
1985. Submitted to WHO by Mitsui Toatsu Chemicals, Inc., Tokyo,
Japan.
Cummins, H.A. & Gardner, J.R. (1985) MTI-500 alpha-CO: Acute oral
toxicity in the rat. Life Science Research Ltd., England - study no.
85/MT0018/474; report dated 2 August 1985. Submitted to WHO by
Mitsui Toatsu Chemicals, Inc., Tokyo, Japan.
Foster, R. (1985). Reverse mutation in Salmonella typhimurium. Test
substance MTI-500. Life Science Research Roma Toxicology Centre,
Italy; report no. 162001-M-06185, dated 22/8/1985. Submitted to WHO
by Mitsui Toatsu Chemicals, Inc., Tokyo, Japan.
Green, O.P., Heywood, R., Street, A.E., Gopinath, & Almond, R.H.
(1985a). Assessment of the toxicity of MTI-500 to mice by dietary
administration for 13 weeks. Huntingdon Research Centre Ltd.,
England; report no. MTC 55/821112, dated 8 July 1983, re-issued 31
October 1985. Submitted to WHO by Mitsui Toatsu Chemicals, Inc.,
Tokyo, Japan.
Green, O.P., Street, A.E., Heywood, R., Gopinath, C. & Almond, R.H.
(1985b). Assessment of the toxicity of MTI-500 in rats during
dietary administration for 13 weeks. Huntingdon Research Centre
Ltd., England; report no. MTC 56/821067, dated 7 July 1983, re-
issued 18 December 1985. Submitted to WHO by Mitsui Toatsu
Chemicals, Inc., Tokyo, Japan.
Green, O.P., Heaps, C.J., Heywood, R., Street, A.E., Gopinath, C.,
Imm, S. & Gibson, W.A. (1986a). Ethofenprox (MTI-500) potential
tumorigenic and toxic effects in prolonged dietary administration to
mice. Huntingdon Research Centre Ltd., England; report no. MTC
58/85582, dated 6 January 1986, re-issued 24 February 1986.
Submitted to WHO by Mitsui Toatsu Chemicals, Inc., Tokyo, Japan.
Green, O.P., Heaps, C.J., Heywood, R., Street, A.E., Gopinath, C.,
Sing, H. & Gibson, W.A. (1986b). Ethofenprox (MTI-500) potential
tumorigenic and toxic effects in prolonged dietary administration to
rats. Huntingdon Research Centre Ltd., England; report no. MTC
59/85581, dated 24 January 1986. Submitted to WHO by Mitsui Toatsu
Chemicals, Inc., Tokyo, Japan.
Harling, R.J., Burford, P. & Heywood, R. (1985a) Ethofenprox (MTI-
500) Acute limit test of toxicity to dogs following a single oral
administration. Huntingdon Research Centre Ltd., England; report no.
MTC 101/851185, dated 24 October 1985. Submitted to WHO by Mitsui
Toatsu Chemicals, Inc., Tokyo, Japan.
Harling, R.J., Burford, P., Street, A.E., Heywood, R., Majeed, S.R.
& Gopinath, C. (1985b). Ethofenprox (MTI-500) toxicity to dogs by
repeated dietary administration for 52 weeks followed by a recovery
period of 8 weeks. Huntingdon Research Centre Ltd., England; report
no. MTC 71/85234, dated 25 October 1985, re-issued 8 November 1985.
Submitted to WHO by Mitsui Toatsu Chemicals, Inc., Tokyo, Japan.
Hashimoto, K., Ohtaki, T., Yamaguchi, K., Nishijima, Y., Imai, K. &
Azegami, J. (1982a). Report on acute toxicity study of MTI-500
(ethofenprox) in mice. Hatano Research Institute, Japan; report no.
A-82-35~42, dated 9 November 1982. Submitted to WHO by Mitsui Toatsu
Chemicals, Inc., Tokyo, Japan.
Hashimoto, K., Ohtaki, T., Yamada, S., Watanabe, C., Imai, K. &
Azegami, J. (1982b). Report on acute toxicity study of MTI-500
(ethofenprox) in rats. Hatano Research Institute, Japan; report no.
A-82-27~34, dated 9 November 1982. Submitted to WHO by Mitsui Toatsu
Chemicals, Inc., Tokyo, Japan.
Hawkins, D.R., Kirkpatrick, D., Ewen, B., Midgley, I., Biggs, S.R. &
Whitby, B.R. (1985a). The biokinetics and metabolism of 14C-
ethofenprox in the rat. Huntingdon Research Centre Ltd., England;
report no. HRC/MTC 68/84610, dated 1 August 1985. Submitted to WHO
by Mitsui Toatsu Chemicals, Inc., Tokyo, Japan.
Hawkins, D.R., Kirkpatrick, D., Ewen, B., Midgley, I. & Biggs, S.R.
(1985b). The metabolism of 14C-ethofenprox in dogs. Huntingdon
Research Centre Ltd., England; report no. HRC/MTC 69/84583, dated 11
October 1985. Submitted to WHO by Mitsui Toatsu Chemicals, Inc.,
Tokyo, Japan.
Jackson, G.C., Hardy, C.J., Clark, G.C., Gregson, R.L., Lewis, D.J.
& Gopinath, C. (1983). MTI-500 acute inhalation toxicity in rats 4
hour exposure. Huntingdon Research Centre Ltd., England; report no.
MTC 60/821079, dated 19 April 1983 Submitted to WHO by Mitsui Toatsu
Chemicals, Inc., Tokyo, Japan.
Kamiya, J., Yoshiwara, K., Saito, S., Takahashi, Y. Oseki, K.,
Shimizu, H., Kawazura, H., Shiga, Y., Yoshida, M., Hayakawa, M. &
Tanaka Y. (1985). General pharmacology of MTI-500. Institute of
Biological Sciences - Mitsui Pharmaceuticals Inc., [A translation of
an original Japanese report published in Japanese, in: Pharmacology
and Therapeutics, vol.13 no.11, pp. 229-244, November 1985].
Translated by T. Hirakawa.
Kashima, M. (1985a). Acute oral toxicity of MTI-500 emulsion in
mice. Nippon Experimental Medical Research Institute, Ltd., Japan;
report no. NEMRI-H-85-50, dated 2 September 1985. Submitted to WHO
by Mitsui Toatsu Chemicals, Inc., Tokyo, Japan.
Kashima, M. (1985b). Acute oral toxicity of MTI-500 emulsion in
rats. Nippon Experimental Medical Research Institute, Ltd., Japan;
report no. NEMRI-H-85-48, dated 2 September 1985. Submitted to WHO
by Mitsui Toatsu Chemicals, Inc., Tokyo, Japan.
Kashima, M. (1985c). Acute dermal toxicity of MTI-500 emulsion in
rats. Nippon Experimental Medical Research Institute, Ltd., Japan;
report no. NEMRI-H-85-49, dated 2 September 1985. Submitted to WHO
by Mitsui Toatsu Chemicals, Inc., Tokyo, Japan.
Kashima, M. (1985d). MTI-500 primary skin stimulation test in
rabbits. Nippon Experimental Medical Research Institute, Ltd, Japan;
report no. NEMRI-H-85-5, dated 23 August 1985. Submitted to WHO by
Mitsui Toatsu Chemicals, Inc., Tokyo, Japan.
Kashima, M. (1985e). MTI-500 primary ophthalmic stimulation test in
rabbits. Nippon Experimental Medical Research Institute, Ltd, Japan;
report no. NEMRI-H-85-55, dated 24 October 1985. Submitted to WHO by
Mitsui Toatsu Chemicals, Inc., Tokyo, Japan.
Kashima (1991a) Final report modification (I) - study name: MTI-500
primary skin stimulation test in rabbits. Nippon Experimental
Medical Research Institute, Ltd, Japan; project no. NEMRI-H-85-5.
Modification d.d. 28 October 1991. Submitted to WHO by Mitsui Toatsu
Chemicals, Inc., Tokyo, Japan.
Kashima (1991b) Final report modification (I) - study name: MTI-500
primary ophthalmic stimulation test in rabbits. Nippon Experimental
Medical Research Institute, Ltd, Japan; project no. NEMRI-H-85-55.
Modification d.d. 28 October 1991. Submitted to WHO by Mitsui Toatsu
Chemicals, Inc., Tokyo, Japan.
Kobayashi, K. (1985). MTI-500 skin sensitization test in guinea-
pigs. Nippon Experimental Medical Research Institute, Ltd, Japan;
report dated 31 October 1985. Submitted to WHO by Mitsui Toatsu
Chemicals, Inc., Tokyo, Japan.
Powell, A.J., Coleman, M., Crook, D., Gopinath, C., Gobson, W.A.,
Read, R.M. & Anderson, A. (1988) MTI-500 alpha-CO: Toxicity to rats
by dietary administration for 13 weeks (Final report). Huntingdon
Research Centre Ltd., England; report no. MTC 141/871448, dated 15
March 1988. Submitted to WHO by Mitsui Toatsu Chemicals, Inc.,
Tokyo, Japan.
Seeberg, A.H. (1985a). Gene mutation in Chinese Hamster V79 cells.
Test substance MTI-500. Life Science Research Roma Toxicology
Centre, Italy; report no. 162002-M-06985, dated 22/8/1985. Submitted
to WHO by Mitsui Toatsu Chemicals, Inc., Tokyo, Japan.
Seeberg, A.H. (1985b). Unscheduled DNA synthesis in human cells,
cell line: Hela S3. Test substance MTI-500. Life Science Research
Roma Toxicology Centre, Italy; report no. 162003-M-05785, dated
30/7/1985. Submitted to WHO by Mitsui Toatsu Chemicals, Inc., Tokyo,
Japan.
Tomoda, K., Arai, M. & Ohnuma, K. (1986). Metabolism study of
ethofenprox (MTI-500). 6. Metabolism in rat. Chiba Branch
Laboratory, Agrochemicals Laboratory - Mitsui Toatsu Chemicals,
Inc., Japan; report dated July 1986. Submitted to WHO by Mitsui
Toatsu Chemicals, Inc., Tokyo, Japan.
WHO (1992). The WHO recommended classification of pesticides by
hazard and guidelines to classification 1992-1993 (WHO/PCS/92.14).
Available from the International Programme on Chemical Safety, World
Health Organization, Geneva, Switzerland.
Yamazaki, Y. (1992). Health report from the Industrial Hygiene
Section, Ohmuta Factory - Mitsui Toatsu Chemicals, Inc., dated 17
April 1992. Submitted to WHO by Mitsui Toatsu Chemicals, Inc.,
Tokyo, Japan.
Table 1. Acute toxicity of etofenprox
Species Sex Route LD50 LC50 Purity Reference
(g/kg bw) (g/m3)
Mouse M&F oral > 107.2 96%a Hashimoto et al., 1982a
M&F dermalb > 2.14 96%a Hashimoto et al., 1982a
M i.p. > 53.6 96%a Hashimoto et al., 1982a
F i.p. 13.4 96%a Hashimoto et al., 1982a
M&F s.c. > 53.6 96%a Hashimoto et al., 1982a
Rat M&F oral > 42.88 96%a Hashimoto et al., 1982b
M&F dermalb > 2.14 96%a Hashimoto et al., 1982b
M&F i.p. > 42.88 96%a Hashimoto et al., 1982b
M&F s.c. > 32.16 96%a Hashimoto et al., 1982b
M&F inhalc > 5.9 96%d Jackson et al., 1983
Dog M&F oral > 5.0 96.3%e Harling et al., 1985a
a compound given as undiluted molten solution (40 °C)
b exposure for 24 hours
c exposure for 4 hours
d compound given as aerosol in acetone
e compound given in gelatine capsules
Table 2. Study results - pharmacological/toxicological action (Kamiya et al., 1985)
Parameter Species (sex) Route Result
Central nervous system
- spontaneous motor activity DDY mouse (M) oral dose-related decrease at 25 & 50 g/kg bw (no other
levels tested)
- thiopental-induced sleeping time DDY mouse (M) oral dose-related increase at 25 & 50 g/kg bw; no effect
at 12.5 g/kg bw
- muscle relaxation actiona DDY mouse (M) oral affected at 50 g/kg bw; no effect at 5 g/kg bw
- body (rectum) temperature DDY mouse (M) oral no effect at 25 and 50 g/kg bw
- induced tonic and clonic DDY mouse (M) oral no effect at 5 & 50 g/kg bw
convulsionsb
- general behaviour Wistar rat (M) oral no effect at 1 & 10 g/kg bw
- EEG of frontal lobe Wistar rat (M) oral dose-related effect at 1 & 10 g/kg bw (no other
levels tested)
- EEG of hippocampus Wistar rat (M) oral effect at 10 g/kg bw; no effect at 1 g/kg bw
& parietal lobe
- conscious reaction induced Wistar rat (M) oral no effect at 1 & 10 g/kg bw
by sound stimulation
- spinal reflex potential mongrel cat (M/F) i.d.c no effect at 0.125-1 g/kg bw (cumulative administration)
Effect on smooth muscle
- intestinal charcoal propulsion DDY mouse (M) oral dose-related increase at 25 & 50 g/kg bw; no effect
at 12.5 g/kg bw
- normal movement and contractile rat, in vitro - 0.01-1.0 mmol/l (cumulative administration)
- responsed of vas deferens produced no effect
- autonomous movement of gravid rat. in vitro - 0.001-1.0 mmol/l (cumulative administration)
or non-gravid uteruse produced no effect
- uterus contractabilityf rat, in vitro - no effect at 0.1 mmol/l
- spontaneous movement rabbit, in vitro - 0.01-1.0 mmol/l (cumulative administration)
& contraction of ileum produced no effect
- spontaneous movement of ileum guinea-pig, - 0.001-1.0 mmol/l (cumulative administration)
in vitro produced no effect
Table 2 (contd)
Parameter Species (sex) Route Result
neuro-muscular junction
- contraction of musculus Wistar rat (M) i.v. no effect at 12.5-100 mg/kg bw
gastrocnemiusg
autonomic ganglia
- contractions of nictitating mongrel cat (M/F) i.v. no effect at 10-100 mg/kg bw
membraneh
respiratory and circulatory system
- respiration rate, blood pressure mongrel dog (M/F) i.v. decreased respiration rate (followed by increased rate)
and ECG in combination with increased blood pressure & heart rate
at > 30 mg/kg bw; at 100 mg/kg bw severer effects; no
effect at 10 mg/kg bw
- cardiovascular response as mongrel dog (M/F) i.v. no effect at 100 mg/kg bw
induced by NE, ISP, AC, HIi
& bilateral carotid occlusion
- contractility of isolated atrium guinea-pig, in vitro -0.01-1.0 mmol/l (cumulative administration) produced no
effect
- positive & negative inotropic guinea-pig, in vitro -0.01-1.0 mmol/l (cumulative administration)
reaction of isolated atrium produced no effect on induced neg.
as induced by ISP & HI (+) or inotropy; effect on pos. inotropy
AC (-) at 1.0 mmol/l only (no effect at 0.01-0.1 mmol/l)
urinary volume & components
- measured: volume, Cl-, Na+ & K+ Wistar rat (M) oral dose-related decrease in volume, Cl- & Na+ at 10 & 20 g/kg
bw (no other dose levels tested), no effect on K+
Table 2 (contd)
Parameter Species (sex) Route Result
serum components
- standard blood biochemistry Wistar rat (M) oral increased glucose (after 1 h only) and GOT and GPT
after 1, 2, 6 & 24 hours (all sampling times) at 10 & 20 g/kg bw (no other
levels tested)
blood coagulation
- prothrombin time, partial Wistar rat (M) oral no effect at 10 & 20 g/kg bw
thromboplastin time & fibrinogen
after 1, 2, 6 & 24 hours
a measured as incidence of slipping down a smooth slope
b induced by pentetrazol, strychnine or electro-shock
c i.d. = intraduodenal
d contractile response induced by di-norepinephrine
e from rats in estrus and diestrus cycle or in pregnant stage
f induced by oxytocin
g induced by electrical stimulation of sciatic nerve and muscle
h as induced by pre- and post-ganglionic nerve stimulation
i NE, norepinephrine; ISP, isoproterenol; AC, acetylcholine; HI, histamine
compound-related since the same effects were also seen (more severe
in degree) at 15 000 ppm (the observed decreases were 8.5-11% at 15
000 ppm and 6% in males at 3000 ppm). The NOAEL in this study was
500 ppm (equal to 60 mg/kg bw/day) (Green et al., 1985a).
Rats
Groups of 20 Sprague-Dawley rats/sex were fed diets containing
0, 50, 300, 1800 or 10 800 ppm etofenprox (purity 96%) for 13 weeks.
These dose levels were equal to 3.3, 20, 120 or 734 mg/kg bw/day in
males and 3.8, 23, 142 or 820 mg/kg bw/day in females. Clinical
signs, mortality, food consumption and body weight were recorded.
Haematology, clinical chemistry (including T3 and T4) and
urinalysis were done in all groups at weeks 6 and 12. Additional
haematology consisted of the determination of PT and PTT in the 0
and 10 800 ppm groups at weeks 0 and 13. The weights of 10
organs/animal were recorded. Gross pathology and histopathology
(about 27 tissues/animal) were carried out. At 10 800 ppm the
following effects were observed: slight growth retardation,
decreased food utilization, increased thrombin time, PT and PTT,
decreased T4, increased cholesterol, increased ASAT, ALAT and LDH
(at week 6 only), increased liver weight with slight hepatocyte
enlargement and increased thyroid weight with increased incidence of
microfollicles in thyroids. These effects, except for the increase
in thrombin time (PTT and PT not determined), were also present to a
slight degree at 1800 ppm. The NOAEL in this study was 300 ppm
(equal to 20 mg/kg bw/day) (Green et al., 1985b).
Dogs
Groups of 6 beagle dogs/sex were given diets containing 0 or 10
000 ppm etofenprox (purity 96.3%) for 52 weeks. Two additional
groups of 4 dogs/sex received dietary dose levels of 100 or 1000 ppm
for 52 weeks. These dose levels were equal to 3.5, 33, or 352 mg/kg
bw/day in males and 3.2, 32 or 339 mg/kg bw/day in females. After
52 weeks, 4 dogs/sex/group were killed. The remaining dogs in the 0
and 10 000 ppm groups were killed after a recovery period of 8
weeks. Observations included clinical signs, mortality, food
consumption, body weight, haematology (including PT and PTT),
clinical chemistry and urinalysis. The weights of 10 organs/animal
were recorded. Gross pathology and histopathology (about 33
tissues/animal) were carried out. Effects observed at 10 000 ppm
were decreased PCV, Hb and RBC, increased SAP, decreased total
protein and albumin, increased kidney weight and increased liver
weight with minimal swelling of hepatocytes. After 8 weeks of
recovery none of these effects were found to persist. The NOAEL in
this study was 1000 ppm (equal to 32 mg/kg bw/day) (Harling et al.,
1985b).
Long-term toxicity/carcinogenicity studies
Mice
Groups of 52 CD-1 mice/sex were fed diets containing 0, 30,
100, 700 or 4900 ppm etofenprox (purity 96.3%) for 2 years. These
dose levels were equal to 3.1, 10, 75 or 550 mg/kg bw/day in males
and 3.6, 12, 81 or 615 mg/kg bw/day in females. Satellite groups of
24 mice/sex/group received the same dose levels and were used for
interim sacrifices after 26 weeks (10 animals/sex per group) and 52
weeks (remaining satellite animals). Observations included clinical
signs, mortality, food consumption, body weight, haematology,
clinical chemistry and urinalysis. Ophthalmoscopy was done in the 0
and 4900 ppm groups. The weights of 10 organs/animal were recorded.
Gross pathology and histopathology (about 30 tissues/animal) were
carried out. At 4900 ppm the following effects were seen: increased
mortality in males (survival at test end 10/52 versus 24/52 in
controls; renal lesions were considered contributory to death),
growth retardation, decreased Hb, RBC, MCHC and PCV in combination
with increased MCV (first year only), increased platelet counts
(second year), decreased urine specific gravity, and increased
weights of liver and spleen. Decreased MCHC and increased MCV were
seen at 700 ppm also. Histopathology showed an increased incidence
of renal changes in treated animals, mainly in males. The renal
changes were mostly tubular lesions appearing as groups of
basophilic or dilated tubules, sometimes associated with focal loss
of tubules. These lesions were scored on a grade 1 to 5 scale. The
incidences are presented in Table 3.
At 30 ppm the increase in the incidence of renal lesions was
considered marginal. No increase in tumour incidence was found.
The NOAEL in this study was 30 ppm (equal to 3.1 mg/kg bw/day)
(Green et al., 1986a).
Rats
Groups of 70 Sprague-Dawley rats/sex were fed diets containing
0, 30, 100, 700 or 4900 ppm etofenprox (purity 96.3%) for 2 years.
These dose levels were equal to 1.1, 3.7, 26 or 187 mg/kg bw/day in
males and 1.4, 4.8, 34 or 249 mg/kg bw/day in females. Interim
sacrifices were made after 26 or 52 weeks (10 animals/sex/group on
both occasions). Observations included clinical signs, mortality,
food consumption, body weight, haematology, clinical chemistry
(including T3 and T4) and urinalysis. The weights of 10
organs/animal were recorded. Gross pathology and histopathology
(about 30 tissues/animal) were carried out. At 4900 ppm, the
following effects were seen: decreased food intake or utilization,
decreased growth, slightly increased thrombin time, increased
urinary protein, increased liver weight with hepatocyte enlargement
(sometimes with vacuolization), slightly increased kidney weight and
increased thyroid weight with increased incidence of cystic
follicles in thyroid. At 700 ppm, decreased food intake, increased
thyroid weight, slightly increased kidney weight and presence of
eosinophilic hepatocytes (sometimes with vacuolization) were noted.
At 100 ppm no toxic effects were observed. Increased incidence of
thyroid follicular tumours was found (Table 4). No changes in
concentrations of T3 and T4 were found.
Statistical analysis of these incidences showed that for
thyroid follicular cell carcinomas the trend test was negative and
the intergroup comparison showed no significant effect. Intergroup
comparison showed a statistically significant increase in adenomas
at 4900 ppm in females only. For adenomas and carcinomas, the trend
test was positive in males and females. The NOAEL in this study was
100 ppm (equal to 3.7 mg/kg bw/day) (Green et al., 1986b).
Reproduction studies
Rats
In a two-generation study, groups of 24 or 28 Sprague-Dawley
rats/sex were used as parent animals, receiving dietary
concentrations of 0, 100, 700 or 4900 ppm etofenprox (purity 96.3%).
Two litters per generation were produced. Parent animals were
killed at day 21 after birth of the Fb litters. Pups were also
sacrificed at day 21 after birth except for selected F1a pups
(maintained on test diets up to day 21 after birth of F1b litters),
selected F1b pups (used as F1 parents) and selected F2b pups
(maintained on test diets up to week 13 after birth). Gross
pathology was done and the weights of selected organs were measured.
Histopathology was done in F1 parents (kidneys, thyroids, liver),
F0 parents (kidneys) and F2b adult animals (thyroid). At 4900 ppm,
the main effects were: decreased growth and increased water
consumption (parent animals), increased kidney weights with presence
of cysts, deposits, congestion, haemorrhages or inflammation in
collecting ducts or medulla (parents and young), increased liver
weights with hepatocyte enlargement (parents and young), decreased
pup weights, clinical signs (tremors, abnormal gait) in pups and
increased thyroid weight with slight increase in height of
follicular epithelium (F1 parents). At 700 ppm the effects were:
increased kidney weights in F2b adult females and increased liver
weights in the young of both generations. No effect on reproduction
parameters were observed. The NOAEL in this study was 100 ppm
(equivalent to 5 mg/kg bw/day) (Cozens et al., 1985a).
Table 3. Number of mice showing renal lesions
(Green et al., 1986a)
Dilated/basophilic Control 30 ppm 100 ppm 700 ppm 4900 ppm
cortical tubules
Malesa
grade 1 7 8 6 9 5
grade 2 0 3 4 6 4
grade 3 0 0 1 1 11
grade 4 0 0 1 1 6
grade 5 0 0 0 0 4
Femalesa
grade 1 3 0 5 6 7
grade 2 1 1 1 1 6
grade 3 0 0 0 2 6
grade 4 0 0 1 0 1
grade 5 0 0 0 0 0
a number of animals examined: 52 in all groups
Table 4. Number of rats with thyroid follicular tumours
(Green et al., 1986b)
Tumour type Control 30 ppm 100 ppm 700 ppm 4900 ppm
Malesa
adenomas 6 6 4 5 11
carcinomas 0 0 1 3 2
adenomas and/or 6 6 5 8 13
carcinomas
Femalesa
adenomas 0 3 2 0 9
carcinomas 0 0 0 2 1
adenomas and/or 0 3 2 2 9
carcinomas
a number of animals examined: 50 in all groups
Special studies on embryo/fetotoxicity
Rats
In a segment I study, groups of 24 Sprague-Dawley rats/sex were
given oral doses (by gavage) of 0, 12.5, 250 or 5000 mg/kg bw/day
etofenprox (purity 96.3%) and were mated during a 14-day mating
period. The male animals were treated for 9 weeks prior to mating
up to termination (autopsy) at week 15. Female animals were dosed
for 2 weeks prior to mating up to day 7 of gestation. At day 20 of
gestation females were sacrificed, standard litter data were
recorded and fetuses were monitored for abnormalities (externally
and standard soft tissue and skeletal examinations). No effects on
mating performance, litter parameters, incidences of soft tissue or
skeletal abnormalities were found. In parent animals at 5000 mg/kg
bw/day salivation with brown staining around the mouth,
staining/wetness around the anogenital region, crystalline
appearance of the faeces, unkempt hair coat (males only) and skin
lesions (males only) were present. Salivation with brown staining
around the mouth was also observed occasionally at 250 mg/kg bw/day
and, in a few animals, at 12.5 mg/kg bw/day. Increased salivation
is frequently observed in gavage studies and is probably due to the
method of application rather than representing a compound-related
toxic effect. The NOAEL for reproductive performance and
fetotoxicity was 5000 mg/kg bw/day. The NOAEL for parental toxicity
was 250 mg/kg bw/day (Cozens et al., 1985c).
In a segment II study, groups of 35 pregnant Sprague-Dawley
rats were given oral doses (by gavage) of 0, 12.5, 250 or 5000 mg/kg
bw/day etofenprox (purity 96.3%) from days 6 through 17 of
pregnancy. No test compound was administered at later stages of the
study. On day 20 of gestation, 21-24 dams/group were sacrificed for
a standard teratogenicity evaluation (i.e. number of corpora lutea,
number and distribution of live and dead fetuses, fetal weights,
external abnormalities, soft tissue examination, skeletal
examination). The remaining dams were allowed to deliver F1 pups.
Litter data were recorded at birth. In the 4-21 days period after
birth, a number of neuromotor parameters (including several reflexes
and eye opening) were determined. On day 21, one pup/sex/litter was
selected for use as F1 parents; pups not selected for later mating
were killed and examined internally and externally for
abnormalities. Mating was done when these animals were 12 weeks
old. Behavioural tests were performed prior to mating. At day 21
after birth of F2 pups all animals were killed and examined
internally and externally for abnormalities. At 5000 mg/kg bw/day
maternal growth was retarded. Salivation, red/brown staining around
the mouth, wet and yellow staining of the fur in the anogenital
region were seen in the 5000 mg/kg bw/day dams during the treatment
period. At 250 and 12.5 mg/kg bw/day salivation and red/brown
staining around the mouth occurred in lesser incidences; these
effects are not considered as compound-related adverse effects. No
effects on mating performance, litter parameters, incidences of soft
tissue or skeletal abnormalities were found in any group. The NOAEL
for fetotoxicity was 5000 mg/kg bw/day and the NOAEL for maternal
toxicity was 250 mg/kg bw/day (Cozens et al., 1985b).
In a segment III study, groups of 25 pregnant Sprague-Dawley
rats were given oral doses (by gavage) of 0, 12.5, 250 or 5000 mg/kg
bw/day etofenprox (purity 96.3%) from day 17 of pregnancy through
day 21 after birth of the F1 pups. No test compound was
administered at later stages of the study. Litter data were
recorded at birth of the F1 pups. On day 4, pups were culled to
8/litter (pups not selected were discarded). In the 4-21 days
period after birth a number of neuromotor parameters (including
several reflexes and eye opening) were determined. On day 21, one
pup/sex/litter was selected for later mating (parent animals and
pups not selected were killed and examined internally and
externally). Mating was done when these animals were about 12 weeks
old. Behavioural tests were performed prior to mating (i.e. general
aspects of behaviour, hole board test for mobility and
inquisitivity, inclined-plane test for motor coordination and a one-
trial avoidance test). At day 21 after birth of F2 pups all
animals were killed and examined internally and externally for
abnormalities. In dams at 5000 mg/kg bw/day, decreased growth
during the treatment phase and clinical signs (salivation,
occasional brown staining around the mouth, staining around the
urogenital region) were observed. Only salivation and brown
staining were observed at 250 mg/kg bw/day; these effects are not
considered as compound-related adverse effects. At 12.5 mg/kg
bw/day no effects in dams were found. At 5000 mg/kg bw/day only, in
F1 pups increased mortality (preceding symptoms: body tremors and
haemorrhages around the nose) and decreased growth were found in the
period up to day 21 after birth. Also at 5000 mg/kg bw/day, only in
F1 weanlings and F1 adults, enlarged kidneys and histopathological
kidney changes were observed. The NOAEL for neonatal effects and
maternal toxicity was 250 mg/kg bw/day (Cozens et al., 1985d).
Rabbits
Groups of 16 or 17 mated New Zeeland white rabbits were given
oral doses (by gavage) of 0, 10, 50 or 250 mg/kg bw/day etofenprox
(purity 96.3%) from day 6 through 18 of gestation. On day 29 all
dams were sacrificed and examined according to standard procedures
(litter data, soft tissue examination, skeletal examination).
Slight body-weight loss was seen in dams at 250 mg/kg bw/day and
growth retardation at 50 mg/kg bw/day. At 250 mg/kg bw/day, the
incidences of late abortions and early embryonic deaths were
increased. At 50 mg/kg bw/day no effect on fetuses was found. The
NOAEL for embryo/fetotoxicity was 50 mg/kg bw/day. The NOAEL for
maternal toxicity was 10 mg/kg bw/day (Bottomley et al., 1985).
Special studies on genotoxicity
Etofenprox has been examined for genotoxicity in bacteria,
mammalian cells in vitro and in mice in vivo (micronucleus
test). In addition a test for UDS in human HeLa cells in vitro
has been carried out. The results are presented in Table 5. The
results were negative in all test systems.
Special studies on the metabolite 2-(4-ethoxyphenyl)-
2-methylpropyl-3-phenoxybenzoate (MTI-500 alpha-CO)
The oral LD50 of MTI-500 alpha-CO in male and female Charles
River CD rats was >5000 mg/kg bw (Cummins & Gardner, 1985).
Groups of 10 Sprague-Dawley rats/sex were fed diets containing
0, 50, 700 or 10 000 ppm MTI-500 alpha-CO (purity 97.6%) for 13
weeks. These dose levels were equal to 3.8, 54 or 800 mg/kg bw/day
in males and 4.7, 64 or 930 mg/kg bw/day in females. Clinical
signs, mortality, food consumption and body weights were recorded.
Haematology, clinical chemistry, T3, T4 and urinalysis were done
in all groups at week 13. At termination the weights of 10
organs/animal were recorded. Gross pathology was done in all
animals. Histopathology was done in about 27 tissues/animal in the
0 and 10 000 ppm groups and in lungs, liver and kidneys of the
intermediate groups. At 10 000 ppm the following effects were
observed: growth retardation, increased total WBC (females only),
increased SAP and SGOT, decreases in T4, serum cholesterol and
serum globulin, increased relative weights of liver (females only),
kidneys and thymus (females only), histopathological changes in
kidneys (hypertrophy of the tubular epithelium of the pars recta) in
4/10 females. At the lower dose levels no effects were observed.
The NOAEL in this study was 700 ppm (equal to 54 mg/kg bw/day)
(Powell et al., 1988).
The mutagenic potential of MTI-500 alpha-CO (purity 99.6%) was
examined in the Ames test using Salmonella typhimurium tester
strains TA 98, 100, 97a, 1535, 1537 and 102 without and with
metabolic activation. In addition, Escherichia coli strain WP2
uvra was also used as test organism (without and with activation).
The concentration range was 50-5000 µg/plate. The results were
negative in all test organisms (Bootman & May, 1985).
Table 5. Results of genotoxicity assays with etofenprox
Test system Test object Concentration Purity Results References
of etofenprox (%)
In vitro
Ames test, without S. typhimurium 200-3200 µg/plate; 96.3 negative Foster, 1985
and with activation TA100, TA98, TA1535, DMSO as solvent
TA1537, TA1538
HGPRT (gene Chinese hamster 9.75-156 µg/ml; 96.3 negative Seeberg, 1985a
mutations), without V79 cells DMSO as solvent
and with activation
Chromosome aberrations human lymphocytes 12.5-50 µg/ml; 96.3 negativea Bootman et al.,
test, without and with DMSO as solvent 1985a
activation
Unscheduled DNA human cells HeLa S3 9.75-156 µg/ml 96.3 negative Seeberg, 1985b
synthesis, without (without act.) or
and with activation 2.44-39 µg/ml (with
act.); solvent DMSO
In vivo
Bone marrow CD-1 mouse 80, 400 and 2000 96.3 negative Bootman et al.,
micronucleus test mg/kg bw, p.o. (single 1985b
dose); sacrifice at 24,
48 and 72 h
a Both the test without activation and the test with activation showed a treatment-related reduction in mitotic index.
Special study on sensitization
Etofenprox (purity not reported) was tested for sensitizing
potential in a group of 20 male English Hartley guinea-pigs using
the maximization test according to Magnusson & Kligman. Treatment
in the induction phase was done with a 20% solution in corn oil.
The second induction application was preceded by a treatment with
10% sodium lauryl sulphate in petrolatum ointment to produce an
irritative response in the induction phase. Etofenprox did not
produce sensitization (Kobayashi, 1985).
Special studies on skin and eye irritation
Skin irritation of etofenprox was studied using a group of 6
Japanese white rabbits. A quantity of 0.5 ml of undiluted
etofenprox (purity 96.3%) was applied as a molten solution (melting
point of compound: 35-38 °C) to the shaven intact skin under
occlusion for 4 hours. Skin observations were made up to day 14
after treatment. Very slight erythema was seen in 1/6 animals up to
day 7. The classification (Draize) based on this result is not
irritating (Kashima, 1985d, 1991a).
Eye irritation of etofenprox was studied using a group of 6
Japanese white rabbits. A quantity of 0.1 ml of undiluted
etofenprox (purity 96.3%) was applied as a molten solution (melting
point of compound: 35-38 °C) through instillation into the
conjunctival sac of the right eye. No washing was done.
Observations were made up to 72 hours after treatment. Mild
conjunctival redness was observed in 6/6 animals; after 72 hours
this effect was no longer present. Mild conjunctival edema was seen
in 1/6 animals, which disappeared after 24 hours. The
classification (Draize) based on this result is not irritating
(Kashima, 1985e; 1991b).
Observation in humans
Health assessments were carried out once or twice each year on
a group of 21 operators engaged in the production of technical
etofenprox for periods ranging from 1.5 to 5.5 years. The
examinations included determination of blood pressure, X-ray
examination, haematology, blood biochemistry, limited urinalysis,
electrocardiography and interview for subjective complaints. No
compound-related effects were observed. The report did not include
data on the exposure concentrations (Yamazaki, 1992).
COMMENTS
After oral administration to rats, total excretion in faeces
was 85-90% and 7-9% in urine. Excretion in bile was found to be 10%
to 30%. Unchanged etofenprox was not found in the bile. Total
retention in the body after 5 days was 3-4%. In the
gastrointestinal tract from 48 to 93% was absorbed. Absorption
tended to be dose-dependent. Tissue concentrations were highest in
fat; this residue was present as unchanged parent compound.
Etofenprox was secreted (as the unchanged compound) in milk. The
major biotransformation routes involve O-de-ethylation of the
ethylphenyl moiety and hydroxylation of the phenoxybenzyl moiety
followed by conjugation with glucuronide or sulfate. Oxidation of
the alpha-CH2 group followed by hydrolysis represents an additional
route. The available results for dogs indicate a lower
gastrointestinal absorption rate than in rats. The major
biotransformation routes were the same as in rats.
Etofenprox has a low acute oral toxicity in mice, rats and
dogs. WHO has classified etofenprox as unlikely to present acute
hazard in normal use.
In a 13-week study in mice, using dietary concentrations of 0,
50, 500, 3000 or 15 000 ppm, the NOAEL was 500 ppm, equal to 60
mg/kg bw/day. The main effects seen were mortality, growth
retardation, increased weights of liver (with enlarged hepatocytes)
and kidneys (with tubular basophilia and dilatation) and decreases
in red blood cell parameters.
In a 13-week study in rats using dietary concentrations of 0,
50, 300, 1800 or 10 800 ppm, the NOAEL was 300 ppm, equal to 20
mg/kg bw/day, based on effects on growth and the liver. In
addition, increased thyroid weight with increased incidence of
microfollicles in this organ was observed.
In a 52-week study in dogs using dietary concentrations of 0,
100, 1000 or 10 000 ppm, the NOAEL was 1000 ppm (equal to 32 mg/kg
bw/day) based on decreased red blood cell parameters, increased
serum alkaline phosphatase and increased liver weight (with swelling
of hepatocytes).
In a two-year toxicity/carcinogenicity study in mice using
dietary concentrations of 0, 30, 100, 700 or 4900 ppm, the NOAEL was
30 ppm (equal to 3.1 mg/kg bw/day) based on an increased incidence
of tubular lesions in the kidneys at > 100 ppm. There was no
evidence of carcinogenicity.
A two-year toxicity/carcinogenicity study in rats also used
dietary concentrations of 0, 30, 100, 700 or 4900 ppm. The NOAEL
was 100 ppm (equal to 3.7 mg/kg bw/day), based on increased weights
of thyroid and kidneys and microscopic liver changes at >700 ppm.
The incidence of cystic follicles in the thyroid was increased at
4900 ppm only. There was an increased incidence of thyroid
follicular adenomas among the 4900 ppm animals which was
statistically significantly increased in females only. The absence
of genotoxicity of etofenprox (see below) in combination with the
observed activation of the thyroid gland, which might be related to
the effects on the liver (the latter probably leading to increased
breakdown of thyroid hormones), is a strong indication for a non-
genotoxic mechanism of induction of the thyroid tumours.
In a two-generation study in rats using dietary concentrations
of 0, 100, 700 or 4900 ppm, the NOAEL was 100 ppm (equivalent to 5
mg/kg bw/day). No effects on reproduction were observed in this
study. Main effects seen in parents as well as young were decreased
growth and effects on the weights and histopathology of liver and
kidneys. The effects on the offspring were consistent with exposure
to unchanged etofenprox via milk.
Embryo/fetotoxicity and teratogenicity were studied in rats (3
studies, segment I, II & III, respectively) and rabbits (1 study).
In the three studies in the rat, etofenprox was administered by
gavage at dose levels of 0, 12.5, 250 or 5000 mg/kg bw/day. In each
of the studies, dose-related maternal toxicity (clinical signs,
growth retardation) was observed at 5000 mg/kg bw/day. In the two
studies in rats with dosing before or during pregnancy, no effects
on offspring/fetuses were seen. In the study in rats with dosing
during lactation (segment III), toxic effects developed in the
offspring, most likely as a result of exposure to etofenprox via
milk. In none of the studies in rats were irreversible structural
malformations found. The NOAEL for maternal or parental toxicity in
each of these studies was 250 mg/kg bw/day. For fetotoxicity, the
NOAEL was 5000 mg/kg bw/day in the segment I and II studies. The
NOAEL for neonatal effects in the segment III study was 250 mg/kg
bw/day. In the rabbit study, the NOAEL for maternal toxicity was 10
mg/kg bw/day, based on decreased growth at 50 and 250 mg/kg bw/day.
Incidences of late abortions and early-embryonal mortality were
increased at 250 mg/kg bw/day only. The NOAEL for embryo/
fetotoxicity was 50 mg/kg bw/day. No irreversible structural
malformations were noted in this study.
Based on the results of the available in vitro and in vivo
genotoxicity data there was no evidence that etofenprox is
genotoxic.
The most sensitive species in the animal studies presently
available appear to be rodents, with NOAELs of 3.1 and 3.7 mg/kg
bw/day for mice and rats, respectively, in the long-term studies.
The ADI was based on the long-term study in mice, using a 100-fold
safety factor.
TOXICOLOGICAL EVALUATION
Level causing no toxicological effects
Mouse: 30 ppm, equal to 3.1 mg/kg bw/day (two-year study)
Rat: 100 ppm, equal to 3.7 mg/kg bw/day (two-year study)
Rabbit: 10 mg/kg bw/day (maternal toxicity in a
teratogenicity study)
Dog: 1000 ppm, equal to 32 mg/kg bw/day (52-week study).
Estimate of acceptable daily intake for humans
0-0.03 mg/kg bw.
Studies which will provide information valuable in the continued
evaluation of the compound
Clarification of the dose-response for thyroid effects in the
rat, including evaluation of T3, T4, TSH and other relevant
parameters.
Observations in humans with adequate information on exposure
levels.
REFERENCES
Agrochemicals Handbook (1991) The Agrochemicals Handbook - Third
Edition. Royal Society of Chemistry -Information Services,
Cambridge, England.
Bootman, J., Hodson-Walker, G. & Dance, C.A. (1985a). In vitro
assessment of the clastogenic activity of MTI-500, ethofenprox, in
cultured human peripheral lymphocytes. Life Science Research
Limited, England; report no. 85/MT0017/430, dated 17 July 1985.
Submitted to WHO by Mitsui Toatsu Chemicals, Inc., Tokyo, Japan.
Bootman, J., Hodson-Walker, G. & Dance, C.A. (1985b). MTI-500,
etofenprox: assessment of clastogenic action on bone marrow
erythrocytes in the micronucleus test. Life Science Research
Limited, England; report no. 85/MT0016/406, dated 3 July 1985.
Submitted to WHO by Mitsui Toatsu Chemicals, Inc., Tokyo, Japan.
Bootman, J. & May, K. (1985). MTI-500alpha-CO: assessment of its
mutagenic potential using amino-acid auxotrophs of Salmonella
typhimurium and Escherichia coli. Life Science Research Limited,
England; report no. 85/MT0020/433, dated 19 July 1985. Submitted to
WHO by Mitsui Toatsu Chemicals, Inc., Tokyo, Japan.
Bottomley, A.M., Barton, S.J., Masters, R.E., Offer, J.M., Parker,
C.A., Anderson, A. & Dawe, I.S. (1985). Effect of etofenprox (MTI-
500) on pregnancy of the rabbit. Huntingdon Research Centre Ltd.,
England; report no. MTC 85(84)/85444, dated 20 December 1985.
Submitted to WHO by Mitsui Toatsu Chemicals, Inc., Tokyo, Japan.
Cozens, D.D., Barton, S.J., Offer, J.M., Parker, C.A. & Anderson, A.
(1985a). Effect of etofenprox (MTI-500) on multiple generations of
the rat. Huntingdon Research Centre Ltd., England; report no. MTC
67/85706, dated 29 October 1985. Submitted to WHO by Mitsui Toatsu
Chemicals, Inc., Tokyo, Japan.
Cozens, D.D., Hughes, E.W. & Anderson, A. (1985b). Effect of
etofenprox (MTI-500) on pregnancy of the rat with rearing to
maturation of the F1 generation. Huntingdon Research Centre Ltd.,
England; report no. MTC 64/85422, dated 28 October 1985. Submitted
to WHO by Mitsui Toatsu Chemicals, Inc., Tokyo, Japan.
Cozens, D.D., Hughes, E.W., Clark, R. & Anderson, A. (1985c). Effect
of etofenprox (MTI-500) on fertility and pregnancy of the rat.
Huntingdon Research Centre Ltd., England; report no. MTC 66/84668,
dated 29 October 1985. Submitted to WHO by Mitsui Toatsu Chemicals,
Inc., Tokyo, Japan.
Cozens, D.D., Hughes, E.W., Offer, J. & Anderson, A. (1985d). Effect
of etofenprox (MTI-500) on the peri and post natal period of the rat
with rearing to maturation of the F1 offspring. Huntingdon Research
Centre Ltd., England; report no. MTC 65/85423, dated 29 October
1985. Submitted to WHO by Mitsui Toatsu Chemicals, Inc., Tokyo,
Japan.
Cummins, H.A. & Gardner, J.R. (1985) MTI-500 alpha-CO: Acute oral
toxicity in the rat. Life Science Research Ltd., England - study no.
85/MT0018/474; report dated 2 August 1985. Submitted to WHO by
Mitsui Toatsu Chemicals, Inc., Tokyo, Japan.
Foster, R. (1985). Reverse mutation in Salmonella typhimurium. Test
substance MTI-500. Life Science Research Roma Toxicology Centre,
Italy; report no. 162001-M-06185, dated 22/8/1985. Submitted to WHO
by Mitsui Toatsu Chemicals, Inc., Tokyo, Japan.
Green, O.P., Heywood, R., Street, A.E., Gopinath, & Almond, R.H.
(1985a). Assessment of the toxicity of MTI-500 to mice by dietary
administration for 13 weeks. Huntingdon Research Centre Ltd.,
England; report no. MTC 55/821112, dated 8 July 1983, re-issued 31
October 1985. Submitted to WHO by Mitsui Toatsu Chemicals, Inc.,
Tokyo, Japan.
Green, O.P., Street, A.E., Heywood, R., Gopinath, C. & Almond, R.H.
(1985b). Assessment of the toxicity of MTI-500 in rats during
dietary administration for 13 weeks. Huntingdon Research Centre
Ltd., England; report no. MTC 56/821067, dated 7 July 1983, re-
issued 18 December 1985. Submitted to WHO by Mitsui Toatsu
Chemicals, Inc., Tokyo, Japan.
Green, O.P., Heaps, C.J., Heywood, R., Street, A.E., Gopinath, C.,
Imm, S. & Gibson, W.A. (1986a). Ethofenprox (MTI-500) potential
tumorigenic and toxic effects in prolonged dietary administration to
mice. Huntingdon Research Centre Ltd., England; report no. MTC
58/85582, dated 6 January 1986, re-issued 24 February 1986.
Submitted to WHO by Mitsui Toatsu Chemicals, Inc., Tokyo, Japan.
Green, O.P., Heaps, C.J., Heywood, R., Street, A.E., Gopinath, C.,
Sing, H. & Gibson, W.A. (1986b). Ethofenprox (MTI-500) potential
tumorigenic and toxic effects in prolonged dietary administration to
rats. Huntingdon Research Centre Ltd., England; report no. MTC
59/85581, dated 24 January 1986. Submitted to WHO by Mitsui Toatsu
Chemicals, Inc., Tokyo, Japan.
Harling, R.J., Burford, P. & Heywood, R. (1985a) Ethofenprox (MTI-
500) Acute limit test of toxicity to dogs following a single oral
administration. Huntingdon Research Centre Ltd., England; report no.
MTC 101/851185, dated 24 October 1985. Submitted to WHO by Mitsui
Toatsu Chemicals, Inc., Tokyo, Japan.
Harling, R.J., Burford, P., Street, A.E., Heywood, R., Majeed, S.R.
& Gopinath, C. (1985b). Ethofenprox (MTI-500) toxicity to dogs by
repeated dietary administration for 52 weeks followed by a recovery
period of 8 weeks. Huntingdon Research Centre Ltd., England; report
no. MTC 71/85234, dated 25 October 1985, re-issued 8 November 1985.
Submitted to WHO by Mitsui Toatsu Chemicals, Inc., Tokyo, Japan.
Hashimoto, K., Ohtaki, T., Yamaguchi, K., Nishijima, Y., Imai, K. &
Azegami, J. (1982a). Report on acute toxicity study of MTI-500
(ethofenprox) in mice. Hatano Research Institute, Japan; report no.
A-82-35~42, dated 9 November 1982. Submitted to WHO by Mitsui Toatsu
Chemicals, Inc., Tokyo, Japan.
Hashimoto, K., Ohtaki, T., Yamada, S., Watanabe, C., Imai, K. &
Azegami, J. (1982b). Report on acute toxicity study of MTI-500
(ethofenprox) in rats. Hatano Research Institute, Japan; report no.
A-82-27~34, dated 9 November 1982. Submitted to WHO by Mitsui Toatsu
Chemicals, Inc., Tokyo, Japan.
Hawkins, D.R., Kirkpatrick, D., Ewen, B., Midgley, I., Biggs, S.R. &
Whitby, B.R. (1985a). The biokinetics and metabolism of 14C-
ethofenprox in the rat. Huntingdon Research Centre Ltd., England;
report no. HRC/MTC 68/84610, dated 1 August 1985. Submitted to WHO
by Mitsui Toatsu Chemicals, Inc., Tokyo, Japan.
Hawkins, D.R., Kirkpatrick, D., Ewen, B., Midgley, I. & Biggs, S.R.
(1985b). The metabolism of 14C-ethofenprox in dogs. Huntingdon
Research Centre Ltd., England; report no. HRC/MTC 69/84583, dated 11
October 1985. Submitted to WHO by Mitsui Toatsu Chemicals, Inc.,
Tokyo, Japan.
Jackson, G.C., Hardy, C.J., Clark, G.C., Gregson, R.L., Lewis, D.J.
& Gopinath, C. (1983). MTI-500 acute inhalation toxicity in rats 4
hour exposure. Huntingdon Research Centre Ltd., England; report no.
MTC 60/821079, dated 19 April 1983 Submitted to WHO by Mitsui Toatsu
Chemicals, Inc., Tokyo, Japan.
Kamiya, J., Yoshiwara, K., Saito, S., Takahashi, Y. Oseki, K.,
Shimizu, H., Kawazura, H., Shiga, Y., Yoshida, M., Hayakawa, M. &
Tanaka Y. (1985). General pharmacology of MTI-500. Institute of
Biological Sciences - Mitsui Pharmaceuticals Inc., [A translation of
an original Japanese report published in Japanese, in: Pharmacology
and Therapeutics, vol.13 no.11, pp. 229-244, November 1985].
Translated by T. Hirakawa.
Kashima, M. (1985a). Acute oral toxicity of MTI-500 emulsion in
mice. Nippon Experimental Medical Research Institute, Ltd., Japan;
report no. NEMRI-H-85-50, dated 2 September 1985. Submitted to WHO
by Mitsui Toatsu Chemicals, Inc., Tokyo, Japan.
Kashima, M. (1985b). Acute oral toxicity of MTI-500 emulsion in
rats. Nippon Experimental Medical Research Institute, Ltd., Japan;
report no. NEMRI-H-85-48, dated 2 September 1985. Submitted to WHO
by Mitsui Toatsu Chemicals, Inc., Tokyo, Japan.
Kashima, M. (1985c). Acute dermal toxicity of MTI-500 emulsion in
rats. Nippon Experimental Medical Research Institute, Ltd., Japan;
report no. NEMRI-H-85-49, dated 2 September 1985. Submitted to WHO
by Mitsui Toatsu Chemicals, Inc., Tokyo, Japan.
Kashima, M. (1985d). MTI-500 primary skin stimulation test in
rabbits. Nippon Experimental Medical Research Institute, Ltd, Japan;
report no. NEMRI-H-85-5, dated 23 August 1985. Submitted to WHO by
Mitsui Toatsu Chemicals, Inc., Tokyo, Japan.
Kashima, M. (1985e). MTI-500 primary ophthalmic stimulation test in
rabbits. Nippon Experimental Medical Research Institute, Ltd, Japan;
report no. NEMRI-H-85-55, dated 24 October 1985. Submitted to WHO by
Mitsui Toatsu Chemicals, Inc., Tokyo, Japan.
Kashima (1991a) Final report modification (I) - study name: MTI-500
primary skin stimulation test in rabbits. Nippon Experimental
Medical Research Institute, Ltd, Japan; project no. NEMRI-H-85-5.
Modification d.d. 28 October 1991. Submitted to WHO by Mitsui Toatsu
Chemicals, Inc., Tokyo, Japan.
Kashima (1991b) Final report modification (I) - study name: MTI-500
primary ophthalmic stimulation test in rabbits. Nippon Experimental
Medical Research Institute, Ltd, Japan; project no. NEMRI-H-85-55.
Modification d.d. 28 October 1991. Submitted to WHO by Mitsui Toatsu
Chemicals, Inc., Tokyo, Japan.
Kobayashi, K. (1985). MTI-500 skin sensitization test in guinea-
pigs. Nippon Experimental Medical Research Institute, Ltd, Japan;
report dated 31 October 1985. Submitted to WHO by Mitsui Toatsu
Chemicals, Inc., Tokyo, Japan.
Powell, A.J., Coleman, M., Crook, D., Gopinath, C., Gobson, W.A.,
Read, R.M. & Anderson, A. (1988) MTI-500 alpha-CO: Toxicity to rats
by dietary administration for 13 weeks (Final report). Huntingdon
Research Centre Ltd., England; report no. MTC 141/871448, dated 15
March 1988. Submitted to WHO by Mitsui Toatsu Chemicals, Inc.,
Tokyo, Japan.
Seeberg, A.H. (1985a). Gene mutation in Chinese Hamster V79 cells.
Test substance MTI-500. Life Science Research Roma Toxicology
Centre, Italy; report no. 162002-M-06985, dated 22/8/1985. Submitted
to WHO by Mitsui Toatsu Chemicals, Inc., Tokyo, Japan.
Seeberg, A.H. (1985b). Unscheduled DNA synthesis in human cells,
cell line: Hela S3. Test substance MTI-500. Life Science Research
Roma Toxicology Centre, Italy; report no. 162003-M-05785, dated
30/7/1985. Submitted to WHO by Mitsui Toatsu Chemicals, Inc., Tokyo,
Japan.
Tomoda, K., Arai, M. & Ohnuma, K. (1986). Metabolism study of
ethofenprox (MTI-500). 6. Metabolism in rat. Chiba Branch
Laboratory, Agrochemicals Laboratory - Mitsui Toatsu Chemicals,
Inc., Japan; report dated July 1986. Submitted to WHO by Mitsui
Toatsu Chemicals, Inc., Tokyo, Japan.
WHO (1992). The WHO recommended classification of pesticides by
hazard and guidelines to classification 1992-1993 (WHO/PCS/92.14).
Available from the International Programme on Chemical Safety, World
Health Organization, Geneva, Switzerland.
Yamazaki, Y. (1992). Health report from the Industrial Hygiene
Section, Ohmuta Factory - Mitsui Toatsu Chemicals, Inc., dated 17
April 1992. Submitted to WHO by Mitsui Toatsu Chemicals, Inc.,
Tokyo, Japan.
