
IPCS INTERNATIONAL PROGRAMME ON CHEMICAL SAFETY
Health and Safety Guide No. 84
HEXACHLOROBUTADIENE
HEALTH AND SAFETY GUIDE
UNITED NATIONS INTERNATIONAL
ENVIRONMENT PROGRAMME LABOUR ORGANISATION
WORLD HEALTH ORGANIZATION
WORLD HEALTH ORGANIZATION, GENEVA 1993
This is a companion volume to Environmental Health Criteria 156:
Hexachlorobutadiene
Published by the World Health Organization for the International
Programme on Chemical Safety (a collaborative programme of the United
Nations Environment Programme, the International Labour Organisation,
and the World Health Organization)
This report contains the collective views of an international group of
experts and does not necessarily represent the decisions or the stated
policy of the United Nations Environment Programme, the International
Labour Organisation, or the World Health Organization
WHO Library Cataloguing in Publication Data
Health and safety guide for hexachlorobutadiene.
(Health and safety guide ; no. 84)
1.Butadienes - standards I.Series
ISBN 92 4 151084 6 (NLM Classification: WA 240)
ISSN 0259-7268
The World Health Organization welcomes requests for permission to
reproduce or translate its publications, in part or in full.
Applications and enquiries should be addressed to the Office of
Publications, World Health Organization, Geneva, Switzerland, which
will be glad to provide the latest information on any changes made to
the text, plans for new editions, and reprints and translations
already available.
(c) World Health Organization 1993
Publications of the World Health Organization enjoy copyright
protection in accordance with the provisions of Protocol 2 of the
Universal Copyright Convention. All rights reserved.
The designations employed and the presentation of the material in this
publication do not imply the expression of any opinion whatsoever on
the part of the Secretariat of the World Health Organization
concerning the legal status of any country, territory, city or area or
of its authorities, or concerning the delimitation of its frontiers or
boundaries.
The mention of specific companies or of certain manufacturers'
products does not imply that they are endorsed or recommended by the
World Health Organization in preference to others of a similar nature
that are not mentioned. Errors and omissions excepted, the names of
proprietary products are distinguished by initial capital letters.
CONTENTS
INTRODUCTION
1. PRODUCT IDENTITY AND USES
1.1. Identity
1.2. Physical and chemical properties
1.3. Analysis
1.4. Production and uses
2. SUMMARY AND EVALUATION
2.1. Environmental fate
2.2. Exposure
2.3. Kinetics and metabolism
2.4. Effects on organisms in the environment
2.5. Effects animals
3. CONCLUSIONS
3.1. Human health
3.2. Environment
4. HUMAN HEALTH HAZARDS, PREVENTION AND PROTECTION, EMERGENCY ACTION
4.1. Human health hazards, prevention and protection, first aid
4.2. Advice to physicians
4.3. Health surveillance advice
4.4. Explosion and fire hazards, prevention
4.4.1. Explosion and fire hazards
4.4.2. Prevention
4.4.3. Fire extinguishing agents
4.5. Storage
4.6. Transport
4.7. Spillage and disposal
4.7.1. Spillage
4.7.2. Disposal
5. HAZARDS FOR THE ENVIRONMENT AND THEIR PREVENTION
6. SUMMARY OF CHEMICAL SAFETY INFORMATION
7. CURRENT REGULATIONS, GUIDELINES, AND STANDARDS
7.1. Exposure limit values
7.2. Specific restrictions
7.3. Labelling, packaging, and transport
7.4. Waste disposal
BIBLIOGRAPHY
INTRODUCTION
The Environmental Health Criteria (EHC) monographs produced by the
International Programme on Chemical Safety include an assessment of
the effects on the environment and on human health of exposure to a
chemical or combination of chemicals, or physical or biological
agents. They also provide guidelines for setting exposure limits.
The purpose of a Health and Safety Guide is to facilitate the
application of these guidelines in national chemical safety
programmes. The first three sections of a Health and Safety Guide
highlight the relevant technical information in the corresponding EHC.
Section 4 includes advice on preventive and protective measures and
emergency action; health workers should be thoroughly familiar with
the medical information to ensure that they can act efficiently in an
emergency. Within the Guide is a Summary of Chemical Safety
Information which should be readily available, and should be clearly
explained, to all who could come into contact with the chemical. The
section on regulatory information has been extracted from the legal
file of the International Register of Potentially Toxic Chemicals
(IRPTC) and from other United Nations sources.
The target readership includes occupational health services, those in
ministries, governmental agencies, industry, and trade unions who are
involved in the safe use of chemicals and the avoidance of
environmental health hazards, and those wanting more information on
this topic. An attempt has been made to use only terms that will be
familiar to the intended user. However, sections 1 and 2 inevitably
contain some technical terms. A bibliography has been included for
readers who require further background information.
Revision of the information in this Guide will take place in due
course, and the eventual aim is to use standardized terminology.
Comments on any difficulties encountered in using the Guide would be
very helpful and should be addressed to:
The Director
International Programme on Chemical Safety
World Health Organization
1211 Geneva 27
Switzerland
THE INFORMATION IN THIS GUIDE SHOULD BE CONSIDERED AS A STARTING POINT
TO A COMPREHENSIVE HEALTH AND SAFETY PROGRAMME
1. PRODUCT IDENTITY AND USES
1.1 Identity
Common name: hexachlorobutadiene
Chemical structure:
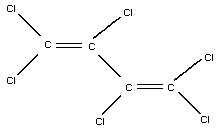 Chemical formula: C4Cl6
Common synonyms: 1,3-hexachlorobutadiene,
1,1,2,3,4,4-hexachloro-1,3-butadiene,
perchlorobutadiene
Common trade names: C-46, Dolen-pur, GP40-66: 120, UN2279
Common abbreviation: HCBD
CAS registry number: 87-68-3
RTECS registry number: EJ 0700000
Relative molecular mass: 260.8
Conversion factors: 1 ppm of hexachlorobutadiene
= 10.67 mg/m3 air, and
1 mg of hexachlorobutadiene per m3 air
= 0.094 ppm
at 25 °C and 101.3 kPa (760 mm Hg).
1.2 Physical and Chemical Properties
Hexachlorobutadiene is a non-flammable, incombustible, clear,
colourless, oily liquid at ordinary temperature and pressure. Its
odour is described as turpentine-like. The compound is poorly soluble
in water, but miscible with ether and ethanol.
Some physical and chemical data on hexachlorobutadiene are presented
in the Summary of Chemical Safety Information (section 6).
1.3 Analysis
The substance can be detected and determined quantitatively by gas
chromatographic methods. Detection limits are 0.03 µg/m3 in air,
0.001 µg/dm3 in water, 0.7 µg/kg wet weight in soil or sediment, and
0.02 µg/dm3 in blood. No odour threshold for the compound in air was
found. In water an odour threshold of 0.006 mg/dm3 was reported.
1.4 Production and Uses
Hexachlorobutadiene has not been reported to occur as a natural
product. The compound was, and is, chiefly produced as a by-product
in the manufacture of chlorinated hydrocarbons, where it occurs in
the heavy fractions (Hex-waste). The annual world production of
the compound in heavy fractions was estimated, in 1982, to be
10 000 tonnes.
The compound can be used for the recovery of chlorine-containing gas
in chlorine plants and as a wash liquor for removing certain volatile
organic compounds from gas streams. It has further been used as a
fluid in gyroscopes, as a heat transfer, transformer-insulating, and
hydraulic fluid, as a solvent for elastomers, an intermediate, and as
a fumigant.
2. SUMMARY AND EVALUATION
2.1 Environmental Fate
The main pathways of entry into the environment are emissions from
waste, and dispersive use. Intercompartmental transport occurs
chiefly by volatilization, adsorption on particulate matter, and
subsequent deposition or sedimentation. The compound does not migrate
rapidly in soil and accumulates in sediment. It is considered
persistent in water, unless there is high turbulence. Hydrolysis does
not occur. The substance seems to be biodegradable aerobically, though
biodegradability has not been investigated thoroughly. The compound
photolyzes. In addition to deposition, reaction with hydroxyl radicals
is assumed to be an important sink of hexachlorobutadiene in the
troposphere with an estimated atmospheric half-life of up to 2.3
years. The substance has a high bioaccumulating potential, as has been
confirmed by both laboratory and field observations. Average,
steady-state bioconcentration factors of 5800 and 17 000, based on wet
weight, have been experimentally determined in rainbow trout.
Biomagnification has not been observed in either the laboratory or the
field.
2.2 Exposure
Hexachlorobutadiene has been measured in urban air: in all cases,
levels have been below 0.5 µg/m3. In remote areas, concentrations
were below 1 pg/m3. In lakes and rivers in Europe, concentrations of
up to 1000 ng/dm3 have been recorded, mean levels usually being
below 100 ng/dm3. In the Great Lakes area, Canada, much lower levels
of around 1 ng/dm3 have been measured. Bottom sediment levels here
can be as high as 120 µg/kg dry weight. Older sediment layers, from
around 1960, contained higher concentrations of up to 550 µg/kg wet
weight. The sediment concentration was shown to increase with the size
of the fraction of sediment particles.
Concentrations of hexachlorobutadiene in aquatic organisms, birds, and
mammals indicate bioaccumulation, but no biomagnification. In polluted
waters, levels of over 1000 µg/kg wet weight have been measured in
several species and 120 mg/kg (lipid base) in one species. Generally,
however, present levels remain below 100 µg/kg wet weight.
The compound has been detected in the urine, blood, and tissues of
humans.
Certain food items containing a high lipid fraction were found to
contain up to about 40 µg hexachlorobutadiene/kg and, in one case,
over 1000 µg/kg.
In a single study on workers, occupational exposure levels of
1.6-16.9 mg hexachlorobutadiene/m3 were recorded.
2.3 Kinetics and Metabolism
Hexachlorobutadiene is rapidly absorbed following oral administration
to experimental animals. The rate of absorption following inhalation
or dermal exposure has not been investigated. In rats and mice, the
compound is mainly distributed to the liver, kidneys, and adipose
tissue. It is rapidly excreted. Binding to liver and kidney protein
and nucleic acids has been demonstrated.
The biotransformation of the compound in experimental animals appears
to be a saturable process, which mainly proceeds through a glutathione
mediated pathway in which hexachlorobutadiene is initially converted
to glutathione conjugates. These conjugates can be metabolized
further, especially in the brush border membrane of renal tubular
cells to a reactive sulfur metabolite, which probably accounts for the
observed nephrotoxicity, genotoxicity, and carcinogenicity.
2.4 Effects on Organisms in the Environment
Hexachlorobutadiene is moderately to very toxic for aquatic organisms.
Crustaceans and fish species were found to be most sensitive, 96-h
LC50 values ranging from 0.032 to 1.2 mg/dm3 and 0.09 to
approximately 1.7 mg/dm3 for fish and crustaceans, respectively. The
kidney was demonstrated to be an important target organ in fish.
On the basis of several long-term tests on algae and fish species, a
no-observed-effect concentration (NOEC) of 0.003 mg/dm3 could be
established, classifying the compound as very toxic for aquatic
species. End-points investigated include general toxicity,
neurotoxicity, biochemistry, haematology, pathology, and reproductive
parameters. In one 28-day early life-stage test on the fathead minnow,
reproduction was not affected at concentrations of up to
0.017 mg/dm3, whereas increased mortality and a decreased body
weight were observed at 0.013 and 0.017 mg/dm3. The NOEC was
0.0065 mg/dm3.
In a 90-day test on Japanese quail receiving a diet containing the
compound at concentrations from 0.3 up to 30 mg/kg diet, the survival
of chicks was decreased at 10 mg/kg diet only. No further reproductive
effects were found.
2.5 Effects on Animals
Hexachlorobutadiene is slightly to moderately toxic for adult rats,
moderately toxic for male weanling rats, and highly toxic for female
weanling rats following a single oral dose (based on the WHO pesticide
toxicity classification). The kidney is the major target organ and, to
a much lesser extent, the liver.
In the kidneys of rats, mice, and rabbits, hexachlorobutadiene caused
a dose-dependent necrosis of the renal proximal tubules. Young rats
and mice are more sensitive than adults, but adult female rats are
highly suceptible. The lowest, single, intraperitoneal dose at which
renal necrosis was induced in adult female rats was 25 mg/kg body
weight, and in adult male and female mice 6.3 mg/kg body weight. Other
routes have not been investigated with respect to renal toxicity.
Biochemical changes and distinct functional alterations in the kidneys
occurred at similar, or higher, doses than those at which necrosis was
observed.
In short-term oral tests, 2 reproductive studies, and 1 long-term diet
study on rats, the kidney also appeared to be the major target of
hexachlorobutadiene. Dose-related effects included a decreased
relative renal weight and tubular epithelial degeneration. The
no-observed-adverse-effect level (NOAEL) for renal toxicity in rats
and mice was 0.2 mg/kg body weight per day. In both species, adult
females were more susceptible than males.
In one short-term inhalation test, similar effects on the kidneys were
observed with exposure to a vapour concentration of 267 mg/m3,
nominally. The NOAEL was 53 mg/m3.
On the basis of animal data, the vapour of hexachlorobutadiene should
be regarded as irritating to mucous membranes and the liquid as
corrosive. The substance should also be regarded as a sensitizing
agent.
Two reproduction diet studies on rats at doses up to 20 and 75 mg/kg
body weight per day, respectively, revealed reduced pup weight at
doses of 7.5 and 20 mg/kg body weight. A highly toxic dose of 75 mg/kg
body weight per day was sufficient to prevent implantation of eggs.
Skeletal abnormalities were not observed.
In two teratogenicity tests on rats receiving 10 mg/kg body weight per
day, intraperitoneally, from days 1 to 15 of pregnancy, or exposed to
vapour concentrations of between 21 and 160 mg/m3 for 6 h per day
from days 6 to 20 of pregnancy, respectively, fetuses showed retarded
development but no malformations. The retarded development was
observed at levels that were also toxic to the dams.
Hexachlorobutadiene and, to a much greater extent, its sulfur
metabolites induced gene mutations in the Ames Salmonella test under
special conditions favouring the formation of glutathione conjugation
products. In one study of exposure to hexachlorobutadiene by
inhalation or oral administration, an increased frequency of
chromosomal aberrations was observed in mouse bone marrow cells. There
is limited evidence for the genotoxicity of hexachlorobutadiene in
mammals, but insufficient evidence in humans.
Long-term administration of hexachlorobutadiene to rats at doses of
0.02, 2, and 20 mg/kg body weight per day resulted in the induction of
renal tubular neoplasms at the highest dose level, and also marked
nephrotoxicity. The evidence for the carcinogenicity of the compound
in experimental animals is considered limited (one study in one rodent
strain) and, for humans, insufficient.
The margin of safety between the estimated no-observed-adverse-effect
level (NOAEL) and the estimated, maximum, total daily human intake is
considered sufficient to protect the general population against the
adverse effects of hexachlorobutadiene.
The maximum predicted environmental concentration (PEC), away from
point sources, is twice the extrapolated Environmental Concern Level.
Aquatic organisms therefore may be at risk in polluted surface waters.
Adverse effects on benthic organisms and fish-eating mammals cannot be
excluded.
3. CONCLUSIONS AND RECOMMENDATIONS
3.1 Human Health
Exposure of the general public mainly occurs indirectly via
drinking-water and food with a high lipid content.
No human effect data are available: the evaluation is based entirely
on experimental animal data.
On the basis of acute, oral studies on adult rats hexachlorobutadiene
is slightly to moderately toxic; on the basis of acute, oral
experiments with weanling rats, it is moderately to highly toxic.
On the basis of limited data, the vapour should be regarded as
irritating to mucous membranes of humans and the liquid should be
regarded as corrosive. The compound should be regarded as a
sensitizing agent.
Adverse reproductive effects have not been found in studies on rats.
In two teratogenicity studies, developmental toxicity was observed in
rat fetuses at levels that were also toxic to the dams.
There is limited evidence of the genotoxicity or carcinogenicity of
hexachlorobutadiene in animals, but insufficient evidence in humans.
Direct exposure is considered too low to cause adverse health effects
in the general population.
3.2 Environment
Overall, the compound can be regarded as persistent. Because of a
strong potential for adsorption on organic matter, the compound
accumulates in sediment and does not migrate rapidly in soils.
Field and laboratory data also support a high bioaccumulation
potential in aquatic and benthic organisms. No evidence has been
obtained for biomagnification.
Hexachlorobutadiene is moderately to very toxic for aquatic organisms.
Aquatic organisms may be at risk in very polluted surface waters.
Adverse effects on benthic organisms cannot be excluded. Consumption
of benthic or aquatic organisms by other species in polluted surface
water may give cause for concern.
4. HUMAN HEALTH HAZARDS, PREVENTION AND PROTECTION, EMERGENCY ACTION
4.1 Human Health Hazards, Prevention and Protection, First Aid
The human health hazards associated with certain types of exposure to
hexachlorobutadiene, together with preventive and protective measures
and first aid recommendations are shown in the Summary of Chemical
Safety Information (section 6).
4.2 Advice to Physicians
No specific antidote is known. Treat symptomatically.
4.3 Health Surveillance Advice
Human beings frequently potentially exposed to hexachlorobutadiene at
levels near, or above, the exposure limit should undergo periodic
medical examination, with emphasis on skin, eyes, and kidneys.
4.4 Explosion and Fire Hazards, Prevention
4.4.1 Explosion and fire hazards
Hexachlorobutadiene is not flammable. The autoignition temperature of
hexachlorobutadiene is 610 °C. The substance decomposes above 600 °C,
or when involved in a fire, with the formation of a noxious gas
(phosgene) and corrosive vapour (hydrochloric acid). Cylinders may
explode in the heat of a fire. The compound reacts with aluminium,
with the production of heat, and attacks rubber and certain plastics.
4.4.2 Prevention
Do not use hexachlorobutadiene near open fires and hot surfaces, or
when welding. Do not smoke. Avoid contact between the compound and
incompatible materials (see section 4.4.1).
Caution must be applied in approaching fire and applying water.
Fire-fighters should use a full-face mask, and self-contained
breathing apparatus.
4.4.3 Fire extinguishing agents
Extinguishing agents include dry chemical powder, foam, water spray,
and carbon dioxide. Water may cause frothing if it gets below the
surface of liquids, and turn to steam; water-fog, applied gently to
the surface, will cause frothing that will extinguish fire.
4.5 Storage
Hexachlorobutadiene should be stored away from foodstuffs and
incompatible materials (see section 4.4.1) in a cool, dark,
fire-proof, and well-labelled room, with ventilation across the floor.
4.6 Transport
In case of an accident, stop the engine, extinguish any naked lights,
and do not smoke. Keep bystanders at a distance and mark the roads.
Keep upwind. In case of spillage or fire, use the methods advised in
sections 4.7 and 4.4, respectively. Notify the police and the fire
service, immediately. In case of poisoning, follow the advice in
section 4.1.
4.7 Spillage and Disposal
4.7.1 Spillage
Evacuate and ventilate the area. Collect leaking liquid in a sealable
container. Do not touch spilled material. Absorb spilled liquid in
sand or an inert absorbent and remove to a safe place. Ensure personal
protection. The vapour is invisible, heavier than air, and spreads
along ground. If the substance has entered a water-course or sewer, or
has contaminated soil or vegetation, advise the police.
4.7.2 Disposal
Spray into a furnace with after-burner and alkali-scrubber.
Incineration will become easier by mixing with a flammable solvent.
5. HAZARDS FOR THE ENVIRONMENT AND THEIR PREVENTION
Overall, the compound can be regarded as persistent, with a high
bioaccumulation potential in aquatic and benthic organisms.
Hexachlorobutadiene is moderately to very toxic for aquatic organisms.
Aquatic organisms may be at risk in polluted surface waters. Adverse
effects on benthic organisms cannot be excluded. Consumption of
benthic or aquatic organisms by other species in polluted surface
water gives cause for concern.
Contamination of soil, water, and the atmosphere can be avoided by
proper methods of storage, transport, and waste disposal. In case of
spillage, apply the methods recommended in section 4.7.1. Use closed
systems where feasible.
6. SUMMARY OF CHEMICAL SAFETY INFORMATION
This summary should be easily available to all health workers
concerned with, and users of, hexachlorobutadiene. It should be
displayed at, or near, entrances to areas where there is potential
exposure to hexachlorobutadiene, and on processing equipment and
containers. The summary should be translated into the appropriate
language(s). All persons potentially exposed to the chemical should
also have the instructions in the summary clearly explained.
Space is available for insertion of the National Occupational
Exposure Limit, the address and telephone number of the National
Poison Control Centre, and for local trade names.
HEXACHLOROBUTADIENE
(perchlorobutadiene)
C4Cl6
PHYSICAL PROPERTIES OTHER CHARACTERISTICS
Relative molecular mass 260.8 Nonflammable, combustible, clear, colourless liquid with
Melting point (°C) -19 to -22 a turpentine-like odour. The substance decomposes above
Boiling point (°C) 215 600 °C, or when involved in a fire, with the formation of a
Water solubility (mg/dm3, 20 °C) 3.2 noxious gas (phosgene) and corrosive vapour (hydrochloric acid).
Specific density (20 °C) 1.5542 The vapour is heavier than air and spreads along the ground.
Relative vapour density 9.0 Reacts with aluminium, with the production of heat. Attacks
Vapour pressure (kPa)(20 °C) 0.02 rubber and certain plastics.
Autoignition temperature (°C) 610 Renal damage may develop following high, acute exposure
and following prolonged exposure. The substance should be
regarded as a possible carcinogen.
HAZARDS/SYMPTOMS PREVENTION AND PROTECTION FIRST AID
SKIN: Corrosive, burns, redness, Wear clean, impervious clothing, Remove contaminated clothing and
pain gloves, and shoes shoes instantly; wash with plenty of
water for at least 15 minutes;
obtain medical attention immediately
EYES: Corrosive, redness, pain, Wear safety goggles or face shield Rinse with plenty of water for at
lacrimation least 15 minutes; obtain medical
attention immediately
INHALATION: Irritation of all Apply ventilation, local exhaust, or Remove victim to fresh air and keep
exposed mucosae; painful sensation breathing protection by a suitable quiet and warm; if breathing has
in nose and throat; at higher respirator stopped, apply artificial
exposures pulmonary oedema respiration; obtain medical
attention immediately
INGESTION: Irritation or Do not eat, drink, chew, or smoke Rinse mouth; give large quantities
corrosion of mouth, throat, and during work; do not keep food in of water to drink; do NOT induce
gastrointestinal tract areas with potential exposure; keep vomiting; obtain medical attention
out of reach of children immediately
ENVIRONMENT: May present Contamination of water, soil,
a risk to aquatic and benthic and atmosphere can be avoided by
organisms and to species proper methods of storage, transport,
consuming these organisms and waste disposal
SPILLAGE STORAGE FIRE AND EXPLOSION
Evacuate and ventilate the area; Store away from foodstuffs and Hexachlorobutadiene decomposes above
collect leaking liquid in a sealable incompatible materials in a cool, 600 °C, or in open fires, with the
container; absorb spilt liquid in dark, fire-proof, and well labelled formation of toxic gases; cylinders
sand or inert material; ensure room, with ventilation across the may explode in the heat; do not use
personal protection floor; containers must be labelled, near open fires and hot surfaces or close
tightly closed, and kept in a good to welding sites; extinguish fires with
condition dry powder, foam, water spray, or
carbon dioxide
WASTE DISPOSAL
Incineration: spray into a furnace National occupational exposure UN 1092
with after-burner and limit: Labelling:
alkali-scrubber
National Poison Control Centre:
7. CURRENT REGULATIONS, GUIDELINES, AND STANDARDS
The information given in this section has been extracted from the
International Register of Potentially Toxic Chemicals (IRPTC) legal
file. The file contains regulatory data on chemicals from 12 countries
and recommendations by 6 international organizations. A full reference
to the original national document from which the information was
extracted can be obtained from IRPTC. When no effective date appears
in the IRPTC legal file, the year of the reference from which the data
are taken is shown, indicated by (r).
The reader should be aware that regulatory decisions about chemicals
taken in a certain country can only be fully understood in the
framework of the legislation of that country. Furthermore, the
regulations and guidelines of all countries are subject to change and
should always be verified with the appropriate regulatory authorities
before application.
7.1 Exposure Limit Values
Some values for exposure limit are shown in the table on the next
page.
7.2 Specific Restrictions
In the USSR, the substance was approved as a fungicide for
agricultural use; application dose and mode, and treatment frequency
were specified (effective date: 1981). A later regulation stipulates
that the substance is severely restricted for use as a pesticide: it
may be used on vines only for quarantine (not harvesting) purposes
(effective date: 1986 (r)).
7.3 Labelling, Packaging, and Transport
The United Nations Committee of Experts on the Transportation of
Dangerous Goods classifies hexachlorobutadiene as a poisonous
substance (Hazard Class 6.1). Hexachlorobutadiene is also classified
as a substance presenting minor danger for packing purposes (Packing
Group III). Packing methods and labels are recommended (effective
date: 1986 (r)).
In Germany, hexachlorobutadiene is classified as harmful to water
(Class 2). In order to protect water, appropriate security measures
should be taken during storage, loading, and transport (effective
date: 1985 (r)).
In Canada, the substance is classified for transport as hazardous to
the environment (Class 9.2). Special provisions for transport are
prescribed (effective date: 1987).
Table 2. Some exposure limit values
Medium Specification Country/ Exposure limit descriptiona Value Effective
organization date
Air Occupational Australia Threshold limit value (TWA) 0.24 mg/m3 b 1983 (r)
Brasil Acceptable limit (48 h/week) 1720 mg/m3 1980 (r)
Canada Threshold limit value (TWA) 0.24 mg/m3 1980
Germany Maximum worksite concentration (TWA) - c,d 1988(r)
Netherlands Maximum limit (TWA) 0.24 mg/m3 1989(r)
Switzerland Maximum worksite concentration (TWA) 0.24 mg/m3 e 1987 (r)
USA (ACGIH) Threshold limit value (TWA) 0.24 mg/m3 1987 (r)
USSR Ceiling value 5 µg/m3 1983
Air Ambient USSR Maximum allowable concentration
average per day 0.2 mg/m3 1978 (r)
once per day 1 mg/m3 1978 (r)
Water Surface USSR Maximum allowable concentration 0.01 mg/dm3 1983
Soil USSR Maximum allowable concentration 0.5 mg/kg 1981
Food USSR Maximum residue limit(specified food) 0.01 mg/kg 1983
Acceptable daily intake (calculated) 0.001 mg/kg 1983
a TWA = time-weighted-average over one working day (usually 8 h).
b Suspected carcinogen.
c Suspected carcinogen; cutaneous absorption.
d No value established because of carcinogenic potential.
e Cutaneous absorption.
7.4 Waste Disposal
In the USA, any solid waste (except domestic) containing
hexachlorobutadiene must be listed as hazardous waste, subject to
handling, transport, treatment, storage, and disposal requirements,
unless it is found the waste cannot pose a threat to human health or
the environment when improperly managed (effective date: 1980). If the
substance is a commercial chemical product, it is identified as "toxic
waste", subject to handling, transport, treatment, storage, and
disposal regulations and permit and notification requirements
(effective date: 1980). An owner or operator of a hazardous waste
incinerator must achieve 99.99% destruction and removal efficiency for
the substance, if it is designated as a principal organic hazardous
constituent in its Environmental Protection Agency (EPA) permit
(effective date: 1981).
In the USA, permits are required for discharge of hexachlorobutadiene
from any point source into USA waters (effective date: 1980). EPA is
required to set, or has set, effluent limitations and pretreatment
standards for 21 major industries regarding hexachlorobutadiene
(effective date: 1981 (r)).
BIBLIOGRAPHY
Anonymous (1989) [Chemical cards. Handling chemicals safely.] 5th
edition. The Netherlands, NIA, VNCI, Samson Tjeenk-Willink. (in Dutch)
Chemical Industry Association (1979) CEFIC TREMcards, Reference
edition. London, England, Chemical Industry Association.
INRS (1977) Fiche toxicologique no 121. Hexachlorobutadiene.
Institut National de Recherche Scientifique, Paris, France.
IRPTC (1990) Data profile for hexachlorobutadiene. Geneva,
Switzerland, International Register of Potentially Toxic Chemicals,
United Nations Environment Programme.
Chemical formula: C4Cl6
Common synonyms: 1,3-hexachlorobutadiene,
1,1,2,3,4,4-hexachloro-1,3-butadiene,
perchlorobutadiene
Common trade names: C-46, Dolen-pur, GP40-66: 120, UN2279
Common abbreviation: HCBD
CAS registry number: 87-68-3
RTECS registry number: EJ 0700000
Relative molecular mass: 260.8
Conversion factors: 1 ppm of hexachlorobutadiene
= 10.67 mg/m3 air, and
1 mg of hexachlorobutadiene per m3 air
= 0.094 ppm
at 25 °C and 101.3 kPa (760 mm Hg).
1.2 Physical and Chemical Properties
Hexachlorobutadiene is a non-flammable, incombustible, clear,
colourless, oily liquid at ordinary temperature and pressure. Its
odour is described as turpentine-like. The compound is poorly soluble
in water, but miscible with ether and ethanol.
Some physical and chemical data on hexachlorobutadiene are presented
in the Summary of Chemical Safety Information (section 6).
1.3 Analysis
The substance can be detected and determined quantitatively by gas
chromatographic methods. Detection limits are 0.03 µg/m3 in air,
0.001 µg/dm3 in water, 0.7 µg/kg wet weight in soil or sediment, and
0.02 µg/dm3 in blood. No odour threshold for the compound in air was
found. In water an odour threshold of 0.006 mg/dm3 was reported.
1.4 Production and Uses
Hexachlorobutadiene has not been reported to occur as a natural
product. The compound was, and is, chiefly produced as a by-product
in the manufacture of chlorinated hydrocarbons, where it occurs in
the heavy fractions (Hex-waste). The annual world production of
the compound in heavy fractions was estimated, in 1982, to be
10 000 tonnes.
The compound can be used for the recovery of chlorine-containing gas
in chlorine plants and as a wash liquor for removing certain volatile
organic compounds from gas streams. It has further been used as a
fluid in gyroscopes, as a heat transfer, transformer-insulating, and
hydraulic fluid, as a solvent for elastomers, an intermediate, and as
a fumigant.
2. SUMMARY AND EVALUATION
2.1 Environmental Fate
The main pathways of entry into the environment are emissions from
waste, and dispersive use. Intercompartmental transport occurs
chiefly by volatilization, adsorption on particulate matter, and
subsequent deposition or sedimentation. The compound does not migrate
rapidly in soil and accumulates in sediment. It is considered
persistent in water, unless there is high turbulence. Hydrolysis does
not occur. The substance seems to be biodegradable aerobically, though
biodegradability has not been investigated thoroughly. The compound
photolyzes. In addition to deposition, reaction with hydroxyl radicals
is assumed to be an important sink of hexachlorobutadiene in the
troposphere with an estimated atmospheric half-life of up to 2.3
years. The substance has a high bioaccumulating potential, as has been
confirmed by both laboratory and field observations. Average,
steady-state bioconcentration factors of 5800 and 17 000, based on wet
weight, have been experimentally determined in rainbow trout.
Biomagnification has not been observed in either the laboratory or the
field.
2.2 Exposure
Hexachlorobutadiene has been measured in urban air: in all cases,
levels have been below 0.5 µg/m3. In remote areas, concentrations
were below 1 pg/m3. In lakes and rivers in Europe, concentrations of
up to 1000 ng/dm3 have been recorded, mean levels usually being
below 100 ng/dm3. In the Great Lakes area, Canada, much lower levels
of around 1 ng/dm3 have been measured. Bottom sediment levels here
can be as high as 120 µg/kg dry weight. Older sediment layers, from
around 1960, contained higher concentrations of up to 550 µg/kg wet
weight. The sediment concentration was shown to increase with the size
of the fraction of sediment particles.
Concentrations of hexachlorobutadiene in aquatic organisms, birds, and
mammals indicate bioaccumulation, but no biomagnification. In polluted
waters, levels of over 1000 µg/kg wet weight have been measured in
several species and 120 mg/kg (lipid base) in one species. Generally,
however, present levels remain below 100 µg/kg wet weight.
The compound has been detected in the urine, blood, and tissues of
humans.
Certain food items containing a high lipid fraction were found to
contain up to about 40 µg hexachlorobutadiene/kg and, in one case,
over 1000 µg/kg.
In a single study on workers, occupational exposure levels of
1.6-16.9 mg hexachlorobutadiene/m3 were recorded.
2.3 Kinetics and Metabolism
Hexachlorobutadiene is rapidly absorbed following oral administration
to experimental animals. The rate of absorption following inhalation
or dermal exposure has not been investigated. In rats and mice, the
compound is mainly distributed to the liver, kidneys, and adipose
tissue. It is rapidly excreted. Binding to liver and kidney protein
and nucleic acids has been demonstrated.
The biotransformation of the compound in experimental animals appears
to be a saturable process, which mainly proceeds through a glutathione
mediated pathway in which hexachlorobutadiene is initially converted
to glutathione conjugates. These conjugates can be metabolized
further, especially in the brush border membrane of renal tubular
cells to a reactive sulfur metabolite, which probably accounts for the
observed nephrotoxicity, genotoxicity, and carcinogenicity.
2.4 Effects on Organisms in the Environment
Hexachlorobutadiene is moderately to very toxic for aquatic organisms.
Crustaceans and fish species were found to be most sensitive, 96-h
LC50 values ranging from 0.032 to 1.2 mg/dm3 and 0.09 to
approximately 1.7 mg/dm3 for fish and crustaceans, respectively. The
kidney was demonstrated to be an important target organ in fish.
On the basis of several long-term tests on algae and fish species, a
no-observed-effect concentration (NOEC) of 0.003 mg/dm3 could be
established, classifying the compound as very toxic for aquatic
species. End-points investigated include general toxicity,
neurotoxicity, biochemistry, haematology, pathology, and reproductive
parameters. In one 28-day early life-stage test on the fathead minnow,
reproduction was not affected at concentrations of up to
0.017 mg/dm3, whereas increased mortality and a decreased body
weight were observed at 0.013 and 0.017 mg/dm3. The NOEC was
0.0065 mg/dm3.
In a 90-day test on Japanese quail receiving a diet containing the
compound at concentrations from 0.3 up to 30 mg/kg diet, the survival
of chicks was decreased at 10 mg/kg diet only. No further reproductive
effects were found.
2.5 Effects on Animals
Hexachlorobutadiene is slightly to moderately toxic for adult rats,
moderately toxic for male weanling rats, and highly toxic for female
weanling rats following a single oral dose (based on the WHO pesticide
toxicity classification). The kidney is the major target organ and, to
a much lesser extent, the liver.
In the kidneys of rats, mice, and rabbits, hexachlorobutadiene caused
a dose-dependent necrosis of the renal proximal tubules. Young rats
and mice are more sensitive than adults, but adult female rats are
highly suceptible. The lowest, single, intraperitoneal dose at which
renal necrosis was induced in adult female rats was 25 mg/kg body
weight, and in adult male and female mice 6.3 mg/kg body weight. Other
routes have not been investigated with respect to renal toxicity.
Biochemical changes and distinct functional alterations in the kidneys
occurred at similar, or higher, doses than those at which necrosis was
observed.
In short-term oral tests, 2 reproductive studies, and 1 long-term diet
study on rats, the kidney also appeared to be the major target of
hexachlorobutadiene. Dose-related effects included a decreased
relative renal weight and tubular epithelial degeneration. The
no-observed-adverse-effect level (NOAEL) for renal toxicity in rats
and mice was 0.2 mg/kg body weight per day. In both species, adult
females were more susceptible than males.
In one short-term inhalation test, similar effects on the kidneys were
observed with exposure to a vapour concentration of 267 mg/m3,
nominally. The NOAEL was 53 mg/m3.
On the basis of animal data, the vapour of hexachlorobutadiene should
be regarded as irritating to mucous membranes and the liquid as
corrosive. The substance should also be regarded as a sensitizing
agent.
Two reproduction diet studies on rats at doses up to 20 and 75 mg/kg
body weight per day, respectively, revealed reduced pup weight at
doses of 7.5 and 20 mg/kg body weight. A highly toxic dose of 75 mg/kg
body weight per day was sufficient to prevent implantation of eggs.
Skeletal abnormalities were not observed.
In two teratogenicity tests on rats receiving 10 mg/kg body weight per
day, intraperitoneally, from days 1 to 15 of pregnancy, or exposed to
vapour concentrations of between 21 and 160 mg/m3 for 6 h per day
from days 6 to 20 of pregnancy, respectively, fetuses showed retarded
development but no malformations. The retarded development was
observed at levels that were also toxic to the dams.
Hexachlorobutadiene and, to a much greater extent, its sulfur
metabolites induced gene mutations in the Ames Salmonella test under
special conditions favouring the formation of glutathione conjugation
products. In one study of exposure to hexachlorobutadiene by
inhalation or oral administration, an increased frequency of
chromosomal aberrations was observed in mouse bone marrow cells. There
is limited evidence for the genotoxicity of hexachlorobutadiene in
mammals, but insufficient evidence in humans.
Long-term administration of hexachlorobutadiene to rats at doses of
0.02, 2, and 20 mg/kg body weight per day resulted in the induction of
renal tubular neoplasms at the highest dose level, and also marked
nephrotoxicity. The evidence for the carcinogenicity of the compound
in experimental animals is considered limited (one study in one rodent
strain) and, for humans, insufficient.
The margin of safety between the estimated no-observed-adverse-effect
level (NOAEL) and the estimated, maximum, total daily human intake is
considered sufficient to protect the general population against the
adverse effects of hexachlorobutadiene.
The maximum predicted environmental concentration (PEC), away from
point sources, is twice the extrapolated Environmental Concern Level.
Aquatic organisms therefore may be at risk in polluted surface waters.
Adverse effects on benthic organisms and fish-eating mammals cannot be
excluded.
3. CONCLUSIONS AND RECOMMENDATIONS
3.1 Human Health
Exposure of the general public mainly occurs indirectly via
drinking-water and food with a high lipid content.
No human effect data are available: the evaluation is based entirely
on experimental animal data.
On the basis of acute, oral studies on adult rats hexachlorobutadiene
is slightly to moderately toxic; on the basis of acute, oral
experiments with weanling rats, it is moderately to highly toxic.
On the basis of limited data, the vapour should be regarded as
irritating to mucous membranes of humans and the liquid should be
regarded as corrosive. The compound should be regarded as a
sensitizing agent.
Adverse reproductive effects have not been found in studies on rats.
In two teratogenicity studies, developmental toxicity was observed in
rat fetuses at levels that were also toxic to the dams.
There is limited evidence of the genotoxicity or carcinogenicity of
hexachlorobutadiene in animals, but insufficient evidence in humans.
Direct exposure is considered too low to cause adverse health effects
in the general population.
3.2 Environment
Overall, the compound can be regarded as persistent. Because of a
strong potential for adsorption on organic matter, the compound
accumulates in sediment and does not migrate rapidly in soils.
Field and laboratory data also support a high bioaccumulation
potential in aquatic and benthic organisms. No evidence has been
obtained for biomagnification.
Hexachlorobutadiene is moderately to very toxic for aquatic organisms.
Aquatic organisms may be at risk in very polluted surface waters.
Adverse effects on benthic organisms cannot be excluded. Consumption
of benthic or aquatic organisms by other species in polluted surface
water may give cause for concern.
4. HUMAN HEALTH HAZARDS, PREVENTION AND PROTECTION, EMERGENCY ACTION
4.1 Human Health Hazards, Prevention and Protection, First Aid
The human health hazards associated with certain types of exposure to
hexachlorobutadiene, together with preventive and protective measures
and first aid recommendations are shown in the Summary of Chemical
Safety Information (section 6).
4.2 Advice to Physicians
No specific antidote is known. Treat symptomatically.
4.3 Health Surveillance Advice
Human beings frequently potentially exposed to hexachlorobutadiene at
levels near, or above, the exposure limit should undergo periodic
medical examination, with emphasis on skin, eyes, and kidneys.
4.4 Explosion and Fire Hazards, Prevention
4.4.1 Explosion and fire hazards
Hexachlorobutadiene is not flammable. The autoignition temperature of
hexachlorobutadiene is 610 °C. The substance decomposes above 600 °C,
or when involved in a fire, with the formation of a noxious gas
(phosgene) and corrosive vapour (hydrochloric acid). Cylinders may
explode in the heat of a fire. The compound reacts with aluminium,
with the production of heat, and attacks rubber and certain plastics.
4.4.2 Prevention
Do not use hexachlorobutadiene near open fires and hot surfaces, or
when welding. Do not smoke. Avoid contact between the compound and
incompatible materials (see section 4.4.1).
Caution must be applied in approaching fire and applying water.
Fire-fighters should use a full-face mask, and self-contained
breathing apparatus.
4.4.3 Fire extinguishing agents
Extinguishing agents include dry chemical powder, foam, water spray,
and carbon dioxide. Water may cause frothing if it gets below the
surface of liquids, and turn to steam; water-fog, applied gently to
the surface, will cause frothing that will extinguish fire.
4.5 Storage
Hexachlorobutadiene should be stored away from foodstuffs and
incompatible materials (see section 4.4.1) in a cool, dark,
fire-proof, and well-labelled room, with ventilation across the floor.
4.6 Transport
In case of an accident, stop the engine, extinguish any naked lights,
and do not smoke. Keep bystanders at a distance and mark the roads.
Keep upwind. In case of spillage or fire, use the methods advised in
sections 4.7 and 4.4, respectively. Notify the police and the fire
service, immediately. In case of poisoning, follow the advice in
section 4.1.
4.7 Spillage and Disposal
4.7.1 Spillage
Evacuate and ventilate the area. Collect leaking liquid in a sealable
container. Do not touch spilled material. Absorb spilled liquid in
sand or an inert absorbent and remove to a safe place. Ensure personal
protection. The vapour is invisible, heavier than air, and spreads
along ground. If the substance has entered a water-course or sewer, or
has contaminated soil or vegetation, advise the police.
4.7.2 Disposal
Spray into a furnace with after-burner and alkali-scrubber.
Incineration will become easier by mixing with a flammable solvent.
5. HAZARDS FOR THE ENVIRONMENT AND THEIR PREVENTION
Overall, the compound can be regarded as persistent, with a high
bioaccumulation potential in aquatic and benthic organisms.
Hexachlorobutadiene is moderately to very toxic for aquatic organisms.
Aquatic organisms may be at risk in polluted surface waters. Adverse
effects on benthic organisms cannot be excluded. Consumption of
benthic or aquatic organisms by other species in polluted surface
water gives cause for concern.
Contamination of soil, water, and the atmosphere can be avoided by
proper methods of storage, transport, and waste disposal. In case of
spillage, apply the methods recommended in section 4.7.1. Use closed
systems where feasible.
6. SUMMARY OF CHEMICAL SAFETY INFORMATION
This summary should be easily available to all health workers
concerned with, and users of, hexachlorobutadiene. It should be
displayed at, or near, entrances to areas where there is potential
exposure to hexachlorobutadiene, and on processing equipment and
containers. The summary should be translated into the appropriate
language(s). All persons potentially exposed to the chemical should
also have the instructions in the summary clearly explained.
Space is available for insertion of the National Occupational
Exposure Limit, the address and telephone number of the National
Poison Control Centre, and for local trade names.
HEXACHLOROBUTADIENE
(perchlorobutadiene)
C4Cl6
PHYSICAL PROPERTIES OTHER CHARACTERISTICS
Relative molecular mass 260.8 Nonflammable, combustible, clear, colourless liquid with
Melting point (°C) -19 to -22 a turpentine-like odour. The substance decomposes above
Boiling point (°C) 215 600 °C, or when involved in a fire, with the formation of a
Water solubility (mg/dm3, 20 °C) 3.2 noxious gas (phosgene) and corrosive vapour (hydrochloric acid).
Specific density (20 °C) 1.5542 The vapour is heavier than air and spreads along the ground.
Relative vapour density 9.0 Reacts with aluminium, with the production of heat. Attacks
Vapour pressure (kPa)(20 °C) 0.02 rubber and certain plastics.
Autoignition temperature (°C) 610 Renal damage may develop following high, acute exposure
and following prolonged exposure. The substance should be
regarded as a possible carcinogen.
HAZARDS/SYMPTOMS PREVENTION AND PROTECTION FIRST AID
SKIN: Corrosive, burns, redness, Wear clean, impervious clothing, Remove contaminated clothing and
pain gloves, and shoes shoes instantly; wash with plenty of
water for at least 15 minutes;
obtain medical attention immediately
EYES: Corrosive, redness, pain, Wear safety goggles or face shield Rinse with plenty of water for at
lacrimation least 15 minutes; obtain medical
attention immediately
INHALATION: Irritation of all Apply ventilation, local exhaust, or Remove victim to fresh air and keep
exposed mucosae; painful sensation breathing protection by a suitable quiet and warm; if breathing has
in nose and throat; at higher respirator stopped, apply artificial
exposures pulmonary oedema respiration; obtain medical
attention immediately
INGESTION: Irritation or Do not eat, drink, chew, or smoke Rinse mouth; give large quantities
corrosion of mouth, throat, and during work; do not keep food in of water to drink; do NOT induce
gastrointestinal tract areas with potential exposure; keep vomiting; obtain medical attention
out of reach of children immediately
ENVIRONMENT: May present Contamination of water, soil,
a risk to aquatic and benthic and atmosphere can be avoided by
organisms and to species proper methods of storage, transport,
consuming these organisms and waste disposal
SPILLAGE STORAGE FIRE AND EXPLOSION
Evacuate and ventilate the area; Store away from foodstuffs and Hexachlorobutadiene decomposes above
collect leaking liquid in a sealable incompatible materials in a cool, 600 °C, or in open fires, with the
container; absorb spilt liquid in dark, fire-proof, and well labelled formation of toxic gases; cylinders
sand or inert material; ensure room, with ventilation across the may explode in the heat; do not use
personal protection floor; containers must be labelled, near open fires and hot surfaces or close
tightly closed, and kept in a good to welding sites; extinguish fires with
condition dry powder, foam, water spray, or
carbon dioxide
WASTE DISPOSAL
Incineration: spray into a furnace National occupational exposure UN 1092
with after-burner and limit: Labelling:
alkali-scrubber
National Poison Control Centre:
7. CURRENT REGULATIONS, GUIDELINES, AND STANDARDS
The information given in this section has been extracted from the
International Register of Potentially Toxic Chemicals (IRPTC) legal
file. The file contains regulatory data on chemicals from 12 countries
and recommendations by 6 international organizations. A full reference
to the original national document from which the information was
extracted can be obtained from IRPTC. When no effective date appears
in the IRPTC legal file, the year of the reference from which the data
are taken is shown, indicated by (r).
The reader should be aware that regulatory decisions about chemicals
taken in a certain country can only be fully understood in the
framework of the legislation of that country. Furthermore, the
regulations and guidelines of all countries are subject to change and
should always be verified with the appropriate regulatory authorities
before application.
7.1 Exposure Limit Values
Some values for exposure limit are shown in the table on the next
page.
7.2 Specific Restrictions
In the USSR, the substance was approved as a fungicide for
agricultural use; application dose and mode, and treatment frequency
were specified (effective date: 1981). A later regulation stipulates
that the substance is severely restricted for use as a pesticide: it
may be used on vines only for quarantine (not harvesting) purposes
(effective date: 1986 (r)).
7.3 Labelling, Packaging, and Transport
The United Nations Committee of Experts on the Transportation of
Dangerous Goods classifies hexachlorobutadiene as a poisonous
substance (Hazard Class 6.1). Hexachlorobutadiene is also classified
as a substance presenting minor danger for packing purposes (Packing
Group III). Packing methods and labels are recommended (effective
date: 1986 (r)).
In Germany, hexachlorobutadiene is classified as harmful to water
(Class 2). In order to protect water, appropriate security measures
should be taken during storage, loading, and transport (effective
date: 1985 (r)).
In Canada, the substance is classified for transport as hazardous to
the environment (Class 9.2). Special provisions for transport are
prescribed (effective date: 1987).
Table 2. Some exposure limit values
Medium Specification Country/ Exposure limit descriptiona Value Effective
organization date
Air Occupational Australia Threshold limit value (TWA) 0.24 mg/m3 b 1983 (r)
Brasil Acceptable limit (48 h/week) 1720 mg/m3 1980 (r)
Canada Threshold limit value (TWA) 0.24 mg/m3 1980
Germany Maximum worksite concentration (TWA) - c,d 1988(r)
Netherlands Maximum limit (TWA) 0.24 mg/m3 1989(r)
Switzerland Maximum worksite concentration (TWA) 0.24 mg/m3 e 1987 (r)
USA (ACGIH) Threshold limit value (TWA) 0.24 mg/m3 1987 (r)
USSR Ceiling value 5 µg/m3 1983
Air Ambient USSR Maximum allowable concentration
average per day 0.2 mg/m3 1978 (r)
once per day 1 mg/m3 1978 (r)
Water Surface USSR Maximum allowable concentration 0.01 mg/dm3 1983
Soil USSR Maximum allowable concentration 0.5 mg/kg 1981
Food USSR Maximum residue limit(specified food) 0.01 mg/kg 1983
Acceptable daily intake (calculated) 0.001 mg/kg 1983
a TWA = time-weighted-average over one working day (usually 8 h).
b Suspected carcinogen.
c Suspected carcinogen; cutaneous absorption.
d No value established because of carcinogenic potential.
e Cutaneous absorption.
7.4 Waste Disposal
In the USA, any solid waste (except domestic) containing
hexachlorobutadiene must be listed as hazardous waste, subject to
handling, transport, treatment, storage, and disposal requirements,
unless it is found the waste cannot pose a threat to human health or
the environment when improperly managed (effective date: 1980). If the
substance is a commercial chemical product, it is identified as "toxic
waste", subject to handling, transport, treatment, storage, and
disposal regulations and permit and notification requirements
(effective date: 1980). An owner or operator of a hazardous waste
incinerator must achieve 99.99% destruction and removal efficiency for
the substance, if it is designated as a principal organic hazardous
constituent in its Environmental Protection Agency (EPA) permit
(effective date: 1981).
In the USA, permits are required for discharge of hexachlorobutadiene
from any point source into USA waters (effective date: 1980). EPA is
required to set, or has set, effluent limitations and pretreatment
standards for 21 major industries regarding hexachlorobutadiene
(effective date: 1981 (r)).
BIBLIOGRAPHY
Anonymous (1989) [Chemical cards. Handling chemicals safely.] 5th
edition. The Netherlands, NIA, VNCI, Samson Tjeenk-Willink. (in Dutch)
Chemical Industry Association (1979) CEFIC TREMcards, Reference
edition. London, England, Chemical Industry Association.
INRS (1977) Fiche toxicologique no 121. Hexachlorobutadiene.
Institut National de Recherche Scientifique, Paris, France.
IRPTC (1990) Data profile for hexachlorobutadiene. Geneva,
Switzerland, International Register of Potentially Toxic Chemicals,
United Nations Environment Programme.
 Chemical formula: C4Cl6
Common synonyms: 1,3-hexachlorobutadiene,
1,1,2,3,4,4-hexachloro-1,3-butadiene,
perchlorobutadiene
Common trade names: C-46, Dolen-pur, GP40-66: 120, UN2279
Common abbreviation: HCBD
CAS registry number:
Chemical formula: C4Cl6
Common synonyms: 1,3-hexachlorobutadiene,
1,1,2,3,4,4-hexachloro-1,3-butadiene,
perchlorobutadiene
Common trade names: C-46, Dolen-pur, GP40-66: 120, UN2279
Common abbreviation: HCBD
CAS registry number: 