
FAO Meeting Report No. PL/1965/10/1
WHO/Food Add./27.65
EVALUATION OF THE TOXICITY OF PESTICIDE RESIDUES IN FOOD
The content of this document is the result of the deliberations of the
Joint Meeting of the FAO Committee on Pesticides in Agriculture and
the WHO Expert Committee on Pesticide Residues, which met in Rome,
15-22 March 19651
Food and Agriculture Organization of the United Nations
World Health Organization
1965
1 Report of the second joint meeting of the FAO Committee on
Pesticides in Agriculture and the WHO Expert Committee on Pesticide
Residues, FAO Meeting Report No. PL/1965/10; WHO/Food Add./26.65
NABAM
Chemical name
Disodium ethylene-1,2-bisdithiocarbamate
Synonyms
DSE, "Parzate"
Empirical formula
C4H6N2S4Na2
Structural formula
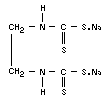 BIOLOGICAL DATA
Acute toxicity
Animal Route LD50 mg/kg References
body-weight
Rat Oral 395 Blackwell-Smith et al., 1953
Mouse Oral 580 Kligman & Rosenweig, 1948
Short-term studies
Rat. When nabam was fed over a period of 9 days to 4 female
weanling rats, increase in thyroid weight and thyroid hyperplasia was
found at levels of 1000, 1500 and 2000 ppm in the diet. A maximum
no-effect level was not determined. The action on the thyroid was
similar to that observed with thiouracil. In addition, feeding levels
of 1500 and 2000 ppm affected the growth rate of the animals, although
1000 ppm did not appear to do so (Seifter & Ehrich, 1948).
Comments on experimental studies reported
For nabam, as for most of the dithiocarbamates, short- and
long-term studies in animals have been reported, but for all of them
biochemical data are inadequate.
EVALUATION
The chemical nature of the residues of the dithiocarbamates in
or on the plant has not been ascertained. The compounds themselves
have effects on the thyroid, nervous system and blood in animals. In
the absence of information about their mode of action an acceptable
intake for man cannot be estimated.
Further work required
Determination and evaluation of toxicity of the residues
occurring in the plant. Extension of the long-term studies including
reproduction studies which should concern at least 2 species. Special
attention should be given to neurological changes, goitrogenicity and
occurrence of anaemia.
REFERENCES
Blackwell-Smith, R., jr, Finnegan, J. K., Larson, P. S., Sahyoun, P.
F., Dreyfus, M. L. & Haag, H. B. (1953) J. Pharmacol. exp. Ther.,
109, 159
Kligman, A. M. & Rosenweig, W. (1948) J. Invest. Dermatology, 10,
59
Seifter, J. & Ehrich, W. E. (1948) J. Pharmacol. exp. Ther., 92,
303
BIOLOGICAL DATA
Acute toxicity
Animal Route LD50 mg/kg References
body-weight
Rat Oral 395 Blackwell-Smith et al., 1953
Mouse Oral 580 Kligman & Rosenweig, 1948
Short-term studies
Rat. When nabam was fed over a period of 9 days to 4 female
weanling rats, increase in thyroid weight and thyroid hyperplasia was
found at levels of 1000, 1500 and 2000 ppm in the diet. A maximum
no-effect level was not determined. The action on the thyroid was
similar to that observed with thiouracil. In addition, feeding levels
of 1500 and 2000 ppm affected the growth rate of the animals, although
1000 ppm did not appear to do so (Seifter & Ehrich, 1948).
Comments on experimental studies reported
For nabam, as for most of the dithiocarbamates, short- and
long-term studies in animals have been reported, but for all of them
biochemical data are inadequate.
EVALUATION
The chemical nature of the residues of the dithiocarbamates in
or on the plant has not been ascertained. The compounds themselves
have effects on the thyroid, nervous system and blood in animals. In
the absence of information about their mode of action an acceptable
intake for man cannot be estimated.
Further work required
Determination and evaluation of toxicity of the residues
occurring in the plant. Extension of the long-term studies including
reproduction studies which should concern at least 2 species. Special
attention should be given to neurological changes, goitrogenicity and
occurrence of anaemia.
REFERENCES
Blackwell-Smith, R., jr, Finnegan, J. K., Larson, P. S., Sahyoun, P.
F., Dreyfus, M. L. & Haag, H. B. (1953) J. Pharmacol. exp. Ther.,
109, 159
Kligman, A. M. & Rosenweig, W. (1948) J. Invest. Dermatology, 10,
59
Seifter, J. & Ehrich, W. E. (1948) J. Pharmacol. exp. Ther., 92,
303
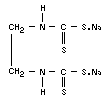 BIOLOGICAL DATA
Acute toxicity
Animal Route LD50 mg/kg References
body-weight
Rat Oral 395 Blackwell-Smith et al., 1953
Mouse Oral 580 Kligman & Rosenweig, 1948
Short-term studies
Rat. When nabam was fed over a period of 9 days to 4 female
weanling rats, increase in thyroid weight and thyroid hyperplasia was
found at levels of 1000, 1500 and 2000 ppm in the diet. A maximum
no-effect level was not determined. The action on the thyroid was
similar to that observed with thiouracil. In addition, feeding levels
of 1500 and 2000 ppm affected the growth rate of the animals, although
1000 ppm did not appear to do so (Seifter & Ehrich, 1948).
Comments on experimental studies reported
For nabam, as for most of the dithiocarbamates, short- and
long-term studies in animals have been reported, but for all of them
biochemical data are inadequate.
EVALUATION
The chemical nature of the residues of the dithiocarbamates in
or on the plant has not been ascertained. The compounds themselves
have effects on the thyroid, nervous system and blood in animals. In
the absence of information about their mode of action an acceptable
intake for man cannot be estimated.
Further work required
Determination and evaluation of toxicity of the residues
occurring in the plant. Extension of the long-term studies including
reproduction studies which should concern at least 2 species. Special
attention should be given to neurological changes, goitrogenicity and
occurrence of anaemia.
REFERENCES
Blackwell-Smith, R., jr, Finnegan, J. K., Larson, P. S., Sahyoun, P.
F., Dreyfus, M. L. & Haag, H. B. (1953) J. Pharmacol. exp. Ther.,
109, 159
Kligman, A. M. & Rosenweig, W. (1948) J. Invest. Dermatology, 10,
59
Seifter, J. & Ehrich, W. E. (1948) J. Pharmacol. exp. Ther., 92,
303
BIOLOGICAL DATA
Acute toxicity
Animal Route LD50 mg/kg References
body-weight
Rat Oral 395 Blackwell-Smith et al., 1953
Mouse Oral 580 Kligman & Rosenweig, 1948
Short-term studies
Rat. When nabam was fed over a period of 9 days to 4 female
weanling rats, increase in thyroid weight and thyroid hyperplasia was
found at levels of 1000, 1500 and 2000 ppm in the diet. A maximum
no-effect level was not determined. The action on the thyroid was
similar to that observed with thiouracil. In addition, feeding levels
of 1500 and 2000 ppm affected the growth rate of the animals, although
1000 ppm did not appear to do so (Seifter & Ehrich, 1948).
Comments on experimental studies reported
For nabam, as for most of the dithiocarbamates, short- and
long-term studies in animals have been reported, but for all of them
biochemical data are inadequate.
EVALUATION
The chemical nature of the residues of the dithiocarbamates in
or on the plant has not been ascertained. The compounds themselves
have effects on the thyroid, nervous system and blood in animals. In
the absence of information about their mode of action an acceptable
intake for man cannot be estimated.
Further work required
Determination and evaluation of toxicity of the residues
occurring in the plant. Extension of the long-term studies including
reproduction studies which should concern at least 2 species. Special
attention should be given to neurological changes, goitrogenicity and
occurrence of anaemia.
REFERENCES
Blackwell-Smith, R., jr, Finnegan, J. K., Larson, P. S., Sahyoun, P.
F., Dreyfus, M. L. & Haag, H. B. (1953) J. Pharmacol. exp. Ther.,
109, 159
Kligman, A. M. & Rosenweig, W. (1948) J. Invest. Dermatology, 10,
59
Seifter, J. & Ehrich, W. E. (1948) J. Pharmacol. exp. Ther., 92,
303
