
INTERNATIONAL PROGRAMME ON CHEMICAL SAFETY
ENVIRONMENTAL HEALTH CRITERIA 97
DELTAMETHRIN
This report contains the collective views of an international group of
experts and does not necessarily represent the decisions or the stated
policy of the United Nations Environment Programme, the International
Labour Organisation, or the World Health Organization.
Published under the joint sponsorship of
the United Nations Environment Programme,
the International Labour Organisation,
and the World Health Organization
World Health Orgnization
Geneva, 1990
The International Programme on Chemical Safety (IPCS) is a
joint venture of the United Nations Environment Programme, the
International Labour Organisation, and the World Health
Organization. The main objective of the IPCS is to carry out and
disseminate evaluations of the effects of chemicals on human health
and the quality of the environment. Supporting activities include
the development of epidemiological, experimental laboratory, and
risk-assessment methods that could produce internationally
comparable results, and the development of manpower in the field of
toxicology. Other activities carried out by the IPCS include the
development of know-how for coping with chemical accidents,
coordination of laboratory testing and epidemiological studies, and
promotion of research on the mechanisms of the biological action of
chemicals.
WHO Library Cataloguing in Publication Data
Deltamethrin.
(Environmental health criteria ; 97)
1.Pyrethrins I.Series
ISBN 92 4 154297 7 (NLM Classification: WA 240)
ISSN 0250-863X
The World Health Organization welcomes requests for permission
to reproduce or translate its publications, in part or in full.
Applications and enquiries should be addressed to the Office of
Publications, World Health Organization, Geneva, Switzerland, which
will be glad to provide the latest information on any changes made
to the text, plans for new editions, and reprints and translations
already available.
(c) World Health Organization 1990
Publications of the World Health Organization enjoy copyright
protection in accordance with the provisions of Protocol 2 of the
Universal Copyright Convention. All rights reserved.
The designations employed and the presentation of the material
in this publication do not imply the expression of any opinion
whatsoever on the part of the Secretariat of the World Health
Organization concerning the legal status of any country, territory,
city or area or of its authorities, or concerning the delimitation
of its frontiers or boundaries.
The mention of specific companies or of certain manufacturers'
products does not imply that they are endorsed or recommended by the
World Health Organization in preference to others of a similar
nature that are not mentioned. Errors and omissions excepted, the
names of proprietary products are distinguished by initial capital
letters.
CONTENTS
ENVIRONMENTAL HEALTH CRITERIA FOR TETRAMETHRIN, CYHALOTHRIN, AND
DELTAMETHRIN
INTRODUCTION
1. SUMMARY AND EVALUATION, CONCLUSIONS AND RECOMMENDATIONS
1.1. Summary and evaluation
1.1.1. Identity, physical and chemical properties,
analytical methods
1.1.2. Production and uses
1.1.3. Human exposure
1.1.4. Environmental exposure and fate
1.1.5. Uptake, metabolism, and excretion
1.1.6. Effects on organisms in the environment
1.1.7. Effects on experimental animals and in vitro test
systems
1.1.8. Effects on human beings
1.2. Conclusions
1.3. Recommendations
2. IDENTITY, PHYSICAL AND CHEMICAL PROPERTIES, ANALYTICAL METHODS
2.1. Identity
2.2. Physical and chemical properties
2.3. Analytical methods
3. SOURCES OF ENVIRONMENTAL POLLUTION AND ENVIRONMENTAL LEVELS
3.1. Industrial production
3.2. Use patterns
3.3. Residues in food
3.4. Levels in the environment
4. ENVIRONMENTAL TRANSPORT, DISTRIBUTION, AND TRANSFORMATION
4.1. Transport and distribution between media
4.2. Abiotic degradation in air and water
4.3. Environmental fate
4.4. Bioaccumulation
5. KINETICS AND METABOLISM
5.1. Metabolism in experimental animals
5.2. Metabolism and fate in farm animals
5.3. Enzymatic systems for biotransformation
5.4. Metabolism in human beings
6. EFFECTS ON ORGANISMS IN THE ENVIRONMENT
6.1. Aquatic organisms
6.1.1. Acute toxicity for fish
6.1.2. Acute toxicity for other aquatic organisms
6.1.3. Field studies and community effects
6.1.4. Appraisal
6.2. Terrestrial organisms
6.2.1. Plants
6.2.2. Soil microorganisms
6.2.3. Soil fauna
6.2.3.1 Earthworms
6.2.3.2 Slugs
6.2.3.3 Soil arthropods
6.2.4. Beneficial insects
6.2.4.1 Honey-bees
6.2.4.2 Foliar insects
6.2.5. Birds
6.2.5.1 Laboratory studies
6.2.5.2 Field studies on birds
7. EFFECTS ON EXPERIMENTAL ANIMALS AND IN VITRO TEST SYSTEMS
7.1. Single exposures
7.1.1. Mouse
7.1.2. Rat
7.1.3. Rabbit
7.1.4. Dog
7.2. Irritation and sensitization
7.2.1. Skin irritation
7.2.2. Eye irritation
7.2.3. Sensitization
7.3. Short-term exposure
7.3.1. Rat
7.3.2. Dog
7.4. Long-term exposure and carcinogenicity
7.4.1. Mouse and rat
7.4.2. Dog
7.5. Mutagenicity
7.5.1. Microorganisms
7.5.2. Cultured cells
7.5.3. Mouse
7.5.4. Appraisal
7.6. Teratological and reproductive effects
7.6.1. Teratology
7.6.1.1 Mouse
7.6.1.2 Rat
7.6.1.3 Rabbit
7.6.2. Reproduction studies
7.7. Neurotoxicity and behavioural effects
7.8. Miscellaneous effects
7.9. Potentiation
7.10. Mechanism of toxicity (mode of action)
7.11. Experimental studies on antidotes
8. EFFECTS ON MAN
8.1. General population-poisoning incidents
8.2. Occupational exposure
8.2.1. Acute toxicity-poisoning incidents
8.2.2. Effects of short- and long-term exposure
8.3. Clinical studies
9. PREVIOUS EVALUATIONS BY INTERNATIONAL BODIES
REFERENCES
APPENDIX I
RESUME
RESUMEN
WHO TASK GROUP ON ENVIRONMENTAL HEALTH CRITERIA FOR TETRAMETHRIN,
CYHALOTHRIN, AND DELTAMETHRIN
Members
Dr V. Benes, Department of Toxicology & Reference Laboratory,
Institute of Hygiene and Epidemiology, Prague, Czechoslovakia
Dr A.J. Browning, Toxicology Evaluation Section, Department of
Community Services and Health, Woden, Australia
Dr S. Dobson, Institute of Terrestrial Ecology, Monks Wood
Experimental Station, Huntingdon, Cambridge, United Kingdom
(Chairman)
Dr K. Imaida, Section of Tumor Pathology, Division of Pathology,
National Institute of Hygienic Sciences, Tokyo, Japan
Dr P. Hurley, Office of Pesticide Programme, US Environmental
Protection Agency, Washington, DC, USA
Dr S.K. Kashyap, National Institute of Occupational Health,
(I.C.M.R.) Ahmedabad, India (Vice-Chairman)
Dr Yu. I. Kundiev, Research Institute of Labour, Hygiene and
Occupational Diseases, Kiev, USSR
Dr J.P. Leahey, ICI Agrochemicals, Jealotts Hill Research Station,
Bracknell, Berkshire, United Kingdom (Rapporteur)
Dr M. Matsuo, Sumitomo Chemical Company Limited, Biochemistry &
Toxicology Laboratory, Osaka, Japan
Observers
Mr M. L'Hotellier, International Group of National Associations of
Manufacturers of Agrochemical Products (GIFAP)
Dr N. Punja, International Group of National Associations of
Manufacturers of Agrochemical Products (GIFAP)
Secretariat
Dr K.W. Jager, Division of Environmental Health, International
Programme on Chemical Safety, World Health Organization, Geneva,
Switzerland (Secretary)
Dr R. Plestina, Division of Vector Biology and Control, World
Health Organization, Geneva, Switzerland
Dr J. Sekizawa, Division of Information on Chemical Safety,
National Institute of Hygienic Sciences, Tokyo, Japan (Rapporteur)
NOTE TO READERS OF THE CRITERIA DOCUMENTS
Every effort has been made to present information in the
criteria documents as accurately as possible without unduly
delaying their publication. In the interest of all users of the
environmental health criteria documents, readers are kindly
requested to communicate any errors that may have occurred to the
Manager of the International Programme on Chemical Safety, World
Health Organization, Geneva, Switzerland, in order that they may be
included in corrigenda, which will appear in subsequent volumes.
* * *
A detailed data profile and a legal file can be obtained from
the International Register of Potentially Toxic Chemicals, Palais
des Nations, 1211 Geneva 10, Switzerland (Telephone no. 7988400 -
7985850).
NOTE: The proprietary information contained in this document
cannot replace documentation for registration purposes, because the
latter has to be closely linked to the source, the manufacturing
route, and the purity/impurities of the substance to be registered.
The data should be used in accordance with para. 82-84 and
recommendations para. 90 of the 2nd FAO Government Consultation
(1982).
ENVIRONMENTAL HEALTH CRITERIA FOR TETRAMETHRIN, CYHALOTHRIN, AND
DELTAMETHRIN
A WHO Task Group on Environmental Health Criteria for
Tetramethrin, Cyhalothrin, and Deltamethrin met at the World Health
Organization, Geneva, from 24 - 28 October 1988. Dr M. Mercier,
Manager of the IPCS, welcomed the participants on behalf of the
three IPCS cooperating organizations (UNEP/ILO/WHO). The Group
reviewed and revised the draft Criteria Documents and Health and
Safety Guides and made an evaluation of the risks for human health
and the environment from exposure to tetramethrin, cyhalothrin, and
deltamethrin.
The first drafts of the documents on tetramethrin and
deltamethrin were prepared by Dr J. MIYAMOTO and Dr M. MATSUO of
Sumitomo Chemical Co. Limited. Dr J. SEKISAURA of the National
Institute of Hygienic Sciences, Tokyo, Japan, assisted in the
finalization of the drafts. The first draft of the document on
cyhalothrin was prepared by the IPCS Secretariat based on material
made available by ICI Agrochemicals, United Kingdom.
The second drafts were prepared by the IPCS Secretariat,
incorporating comments received following circulation of the first
drafts to the IPCS contact points for Environmental Health Criteria
documents.
Dr K. JAGER of the IPCS Central Unit was responsible for the
scientific content of the deltamethrin document, and Mrs M.O. HEAD
of Oxford, England, for the editing.
The fact that Sumitomo Chemical Company Limited, Japan, ICI
Agrochemicals, United Kingdom, and Roussel Uclaf SA, France, made
available to the IPCS and the Task Group their proprietary
toxicological information on their products under discussion is
gratefully acknowledged. This allowed the Task Group to make their
evaluation on a more complete data base.
The efforts of all who helped in the preparation and
finalization of the documents is gratefully acknowledged.
INTRODUCTION
Synthetic pyrethroids-a profile
During investigations to modify the chemical structures of
natural pyrethrins, a certain number of synthetic pyrethroids were
produced with improved physical and chemical properties and greater
biological activity. Several of the earlier synthetic pyrethroids
were successfully commercialized, mainly for the control of
household insects. Other more recent pyrethroids have been
introduced as agricultural insecticides because of their excellent
activity against a wide range of insect pests and their non-
persistence in the environment.
The pyrethroids constitute another group of insecticides in
addition to organochlorine, organophosphorus, carbamate, and other
compounds. Pyrethroids commercially available so far include
allethrin, resmethrin, d-phenothrin, and tetramethrin (for insects
of public health importance), and cypermethrin, deltamethrin,
fenvalerate, and permethrin (mainly for agricultural insects).
Other pyrethroids are also available, including furamethrin,
kadethrin, and tellallethrin (usually for household insects),
fenpropathrin, tralomethrin, cyhalothrin, lambda-cyhalothrin,
tefluthrin, cyfluthrin, flucythrinate, fluvalinate, and biphenate
(for agricultural insects).
Toxicological evaluations of several synthetic pyrethroids have
been performed by the FAO/WHO Joint Meeting on Pesticide Residues
(JMPR). The acceptable daily intake (ADI) has been estimated by
the JMPR for cypermethrin, deltamethrin, fenvalerate, permethrin,
d-phenothrin, cyfluthrin, cyhalothrin, and flucythrinate.
Chemically, synthetic pyrethroids are esters of specific acids
(e.g., chrysanthemic acid, halo-substituted chrysanthemic acid,
2-(4-chlorophenyl)-3-methylbutyric acid) and alcohols (e.g.,
allethrolone, 3-phenoxybenzyl alcohol). For certain pyrethroids,
the asymmetric centre(s) exist in the acid and/or alcohol moiety,
and the commercial products sometimes consist of a mixture of both
optical (1R/1S or d/l) and geometric (cis/trans) isomers. However,
most of the insecticidal activity of such products may reside in
only one or two isomers. Some of the products (e.g., d-phenothrin,
deltamethrin) consist only of such active isomer(s).
Synthetic pyrethroids are neuropoisons acting on the axons in
the peripheral and central nervous systems by interacting with
sodium channels in mammals and/or insects. A single dose produces
toxic signs in mammals, such as tremors, hyperexcitability,
salivation, choreo-athetosis, and paralysis. The signs disappear
fairly rapidly, and the animals recover, generally within a week.
At near-lethal dose levels, synthetic pyrethroids cause transient
changes in the nervous system, such as axonal swelling and/or
breaks and myelin degeneration in the sciatic nerves. They are not
considered to cause delayed neurotoxicity of the kind induced by
some organophosphorus compounds. The mechanism of toxicity of
synthetic pyrethroids, and their classification into two types, are
discussed in Appendix I.
Some pyrethroids (e.g., deltamethrin, fenvalerate,
flucythrinate, and cypermethrin) may cause a transient itching
and/or burning sensation in exposed human skin.
Synthetic pyrethroids are generally metabolized in mammals
through ester hydrolysis, oxidation, and conjugation, and there is
no tendency to accumulate in tissues. In the environment,
synthetic pyrethroids are fairly rapidly degraded in soil and in
plants. Ester hydrolysis and oxidation at various sites on the
molecule are the major degradation processes. The pyrethroids are
strongly adsorbed on soil and sediments, and are hardly eluted with
water. There is little tendency for bioaccumulation in organisms.
Because of low application rates and rapid degradation in the
environment, residues in food are generally low.
Synthetic pyrethroids have been shown to be toxic for fish,
aquatic arthropods, and honey-bees in laboratory tests. But, in
practical usage, no serious adverse effects have been noticed
because of the low rates of application and lack of persistence in
the environment. The toxicity of synthetic pyrethroids in birds
and domestic animals is low.
In addition to the evaluation documents of FAO/WHO, there are
several good reviews and books on the chemistry, metabolism,
mammalian toxicity, environmental effects, etc., of synthetic
pyrethroids, including those by Elliot (1977), Miyamoto (1981),
Miyamoto & Kearney (1983), and Leahey (1985).
1. SUMMARY AND EVALUATION, CONCLUSIONS AND RECOMMENDATIONS
1.1 Summary and Evaluation
1.1.1 Identity, physical and chemical properties, analytical
methods
Deltamethrin was synthesized in 1974, and first marketed in
1977. Chemically, it is the [1R, cis; alphaS]-isomer of 8
stereoisomeric esters of the dibromo analogue of chrysanthemic
acid, 2,2-dimethyl-3-(2,2-dibromovinyl) cyclopropanecarboxylic acid
(Br2CA) with alpha-cyano-3-phenoxybenzyl alcohol.
Technical grade deltamethrin is an odourless white powder with
a melting point of 98 - 101 °C and contains more than 98% of the
material. The vapour pressure is 2.0 x 10-6 Pa at 25 °C and it is
practically non-volatile. It is insoluble in water, but soluble in
organic solvents, such as acetone, cyclohexanone, and xylene. It
is stable to light, heat, and air, but unstable in alkaline media.
The determination of residues and analysis of environmental
samples were carried out by solvent extraction with n-hexane/
acetone, partitioning with n-hexane/acetone/water, clean-up with a
silica gel column chromatograph, and determination with a gas
chromatograph equipped with an electron capture detector with a
minimum detectable concentration of 0.01 mg/kg or less. High-
performance liquid chromatography with an UV-detector is used for
product analysis.
1.1.2 Production and uses
The consumption of deltamethrin in the world was about 250
tonnes in 1987. It is mostly used on cotton (45% of the
consumption) and on crops such as coffee, maize, cereals, fruit,
vegetables, and hops, and on stored products. Deltamethrin is also
used in animal health, in vector control, and in public health. It
is formulated as an emulsifiable concentrate, ultra-low-volume
concentrate, wettable powder, suspension concentrate, or dust
powder, alone, or in combination with other pesticides.
1.1.3 Human exposure
Exposure of the general population to deltamethrin is mainly
via dietary residues, but may also occur from its use in public
health. Residue levels in crops treated according to good
agricultural practice are generally very low, except for those
arising from post-harvest treatment. Extensive data have been
reviewed by FAO/WHO (see section 9).
Exposure of the general population is expected to be very low,
but actual data in the form of total diet studies are lacking.
1.1.4 Environmental exposure and fate
When 14C-(acid, alcohol, or cyano labelling)-deltamethrin-[1R,
cis; alphaS] was exposed to sunlight as a thin film for 4 - 8 h,
70% of it was transformed by cis/trans-isomerization to give the
[1R, trans; alphaS] and [1S, trans; alphaS] isomers, together with
ester cleavage products, including Br2CA and alpha-cyano-3-
phenoxybenzyl alcohol.
Deltamethrin was degraded in cotton plants, under glasshouse
conditions, with an initial half-life of 1.1 weeks, and the time
needed for 90% loss was 4.6 weeks.
The major metabolites were free and conjugated Br2CA, trans-
hydroxymethyl-Br2CA, and 3-(4-hydroxyphenoxy)benzoic acid formed by
ester cleavage, oxidation, and conjugation.
Deltamethrin was incubated in sand and organic soil at 28 °C
under laboratory conditions. Approximately 52% and 74% of the
applied deltamethrin was recovered from sand and organic soil,
respectively, 8 weeks after treatment.
Deltamethrin is not mobile in the environment because of its
strong adsorption on particles, its insolubility in water, and very
low rates of application.
No data are available on actual levels in the environment, but
with the current use pattern and under normal conditions of use,
environmental exposure is expected to be very low. Degradation to
less toxic products is rapid.
1.1.5 Uptake, metabolism, and excretion
Deltamethrin is readily absorbed by the oral route, but less so
dermally; the rate of absorption is strongly dependent on the
carrier or solvent. Absorbed deltamethrin is readily metabolized
and excreted.
When rats were given 14C-(acid, alcohol, or cyano labelled)-
deltamethrin orally at the rate of 0.64 - 1.60 mg/kg, the
radiocarbon from the acid and alcohol moiety was almost completely
eliminated within 2 - 4 days. Tissue residue levels were generally
very low, except in fat, where slightly higher residues occurred.
However, the cyano portion was excreted more slowly, with total
recovery of 79% in 8 days. The major metabolic reactions were
oxidation (at the trans-methyl of the cyclopropane ring and at the
2'-, 4'-, and 5-positions of the alcohol moiety), ester cleavage,
and conversion of the cyano portion to thiocyanate. The resultant
carboxylic acids and phenols were conjugated with sulfuric acid,
glycine, and glucuronic acid.
When mice were fed 14C-(acid, alcohol, or cyano labelled)-
deltamethrin orally at rates of 1.7 - 4.4 mg/kg, the excretion of
the radiocarbon was rapid and almost complete, except for the cyano
portion. The major metabolic reactions in mice were generally
similar to those in rats.
In cows and poultry, degradation pathways are very close to
those in rodents.
1.1.6 Effects on organisms in the environment
Deltamethrin is highly toxic for fish, the 96-h LC50 ranging
between 0.4 and 2.0 µg/litre. It is also highly toxic for aquatic
invertebrates; the 48-h LC50 for Daphnia is 5 µg/litre. However,
extensive field studies, in experimental ponds, and field use have
shown that this high potential toxicity is not realized. Some
kills of aquatic invertebrates occur in the field, but these are
usually compensated for rapidly.
The toxicity of deltamethrin for birds is very low with LD50
values for a single oral dose exceeding 1000 mg/kg. Under
laboratory conditions, it is highly toxic for honey-bees with a
contact LD50 of 0.051 µg/bee. Field trials and actual usage have
established that deltamethrin formulations have a repellent action,
which means that, in practice, the hazard for bees is low.
1.1.7 Effects on experimental animals and in vitro test systems
In a non-aqueous vehicle, the acute oral toxicity of
deltamethrin is high to moderate with LD50 values of 19 - 34 mg/kg
(mouse) and 31 - 139 mg/kg (rat). However, in a suspension in
water, the toxicity is much less with LD50 values exceeding
5000 mg/kg (rat). Deltamethrin is a Type II pyrethroid; clinical
signs of poisoning include tremor, salivation, and convulsion. The
onset of signs is rapid and they disappear within several days in
survivors. The electroencephalogram shows generalized spike and
wave discharges prior to choreo-athetosis.
Single applications of technical deltamethrin did not produce
any irritant effect on the intact and abraded skin of the rabbit.
However, transient irritating effects were produced in the eye of
the rabbit, with and without rinsing. Deltamethrin was not a skin
sensitizer in the guinea-pig.
When rats were dosed, by gavage, with deltamethrin levels of up
to 10.0 mg/kg body weight per day for 13 weeks, hyperexcitability
was observed at 6 weeks in males given the highest dose. Body
weight gain was lower in males given 2.5 and 10 mg/kg.
When beagle dogs were dosed orally with deltamethrin at levels
of up to 10 mg/kg body weight per day for 13 weeks, there were
various compound-related symptoms, such as vomiting, tremor,
salivation, and depressed gag-, patellar-, and flexor reflexes. In
a 2-year feeding study on dogs, 1 mg/kg body weight per day was the
no-observed-effect level (highest level tested).
When mice were fed deltamethrin at levels of up to 100 mg/kg
diet for 24 months, tumour incidence was unaffected. The no-
observed-effect level for systemic toxicity was 100 mg/kg diet.
When rats were fed deltamethrin at levels of up to 50 mg/kg
diet for 2 years, no compound-related tumours were observed. The
no-observed-effect level for systemic toxicity was 50 mg/kg diet.
Deltamethrin was not mutagenic in a variety of in vivo and in
vitro test systems, including: DNA repair, gene mutation,
chromosomal aberration, sister chromatid exchange, micronucleus
formation, and dominant lethal tests.
Teratology studies were conducted on pregnant rats and mice in
which deltamethrin was administered orally at levels of up to
10 mg/kg body weight per day during the period of major
organogenesis. There were no teratogenic or reproductive effects,
except for a dose-related decrease in mean fetal weight in the
mouse study and slightly delayed ossification in the rat study.
Rabbits received deltamethrin at levels of up to 16 mg/kg body
weight per day between days 6 and 19 of pregnancy. A decreased
average fetal weight was noted at the highest dose. No teratogenic
effects were observed in rabbits.
When rats were fed deltamethrin at levels of up to 50 mg/kg
diet in a 3-generation, 2-litter reproduction study, no effects on
reproduction were observed.
There are indications that potentiation of toxicity may occur
when deltamethrin is combined with some organophosphorus compounds.
1.1.8 Effects on human beings
Deltamethrin can induce skin sensations in exposed workers.
Several non-fatal cases of poisoning have been reported through
occupational exposure resulting from neglect of safety precautions.
Numbness, itching, tingling, and burning of the skin and vertigo
are symptoms that are frequently reported. Occasionally, a
transient papular or blotchy erythema has been described. Most of
these symptoms are transient and disappear within 5 - 7 days. No
long-term adverse effects have been reported. Three non-fatal
cases of deltamethrin poisoning have been described following
ingestion of several grams of the product.
1.2 Conclusions
General population: The exposure of the general population to
deltamethrin is expected to be very low and is not likely to
present a hazard under recommended conditions of use.
Occupational exposure: With good work practices, measures of
hygiene, and safety precautions, deltamethrin is unlikely to
present a hazard for those occupationally exposed.
Environment: It is unlikely that deltamethrin or its degradation
products will attain levels of adverse environmental significance
with recommended rates of application. Under laboratory
conditions, deltamethrin is highly toxic for fish, aquatic
arthropods, and honey-bees. However, under field conditions,
lasting adverse effects are not likely to occur under recommended
conditions of use.
1.3 Recommendations
Although dietary levels are considered to be very low following
recommended usage, confirmation of this through inclusion of
deltamethrin in monitoring studies should be considered.
Deltamethrin has been used for many years and several cases of
non-fatal poisoning and transient effects from occupational
exposure have been reported. Observations of human exposure should
be maintained.
2. IDENTITY, PHYSICAL AND CHEMICAL PROPERTIES, ANALYTICAL METHODS
2.1 Identity
Molecular formula: C22H19Br2NO3
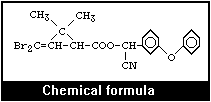
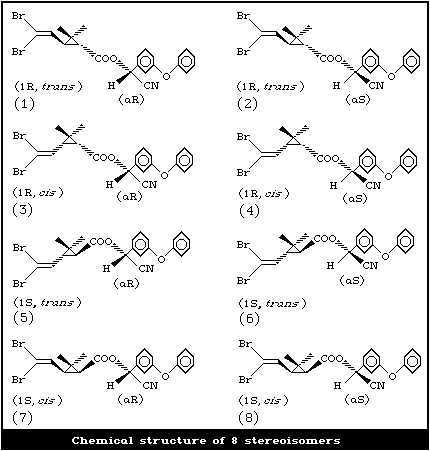 Deltamethrin is the first pyrethroid composed of a single
isomer of 8 stereoisomers selectively prepared by the
esterification of [1R, 3R or cis]-2,2-dimethyl-3-(2,2-dibromovinyl)
cyclopropanecarboxylic acid with (alphaS)- or (+)-alpha-cyano-3-
phenoxybenzyl alcohol or by selective recrystallization of the
racemic esters obtained by esterification of the (1R, 3R or cis)-
acid with the racemic or [alphaR, alphaS, or alphaRS or ±]-alcohol
(Elliott et al., 1974). Thus, its stereospecific structure (4) is
the ester of [1R, 3R or cis]-acid with (alphaS)-alcohol.
The acid is a characteristic dibromo analogue of chrysanthemic
acid.
2.2 Physical and Chemical Properties
Technical grade deltamethrin contains more than 98% deltamethrin
(FAO/WHO, 1981). It is stable to heat (6 months at 40 °C), light,
and air, but unstable in alkaline media (FAO/WHO, 1981; Meister et
al., 1983; Worthing & Walker, 1983). Some physical and chemical
properties are listed in Table 1, and the chemical composition of
various stereoisomeric mixtures is shown in Table 2.
Table 1. Some physical and chemical properties of deltamethrin
-------------------------------------------------------------------
Physical state crystalline powder
Colour colourless
Odour odourless
Density (20 °C) 0.5 g/cm3
Relative molecular mass 505.24
Melting point (°C) 98-101
Boiling point decomposes above 300 °C
Water solubility (20 °C) < 0.002 mg/litre (practically insoluble)
Solubility in organic solublea
solvents
Vapour pressure (25 °C) 2.0 x 10-6 Pa
n-Octanol-water 5.43
partition coefficient
(Log Pow)
-------------------------------------------------------------------
a Acetone (500 g/litre), ethanol (15 g/litre), cyclohexanone (750
g/litre), dioxane (900 g/litre), xylene (250 g/litre), ethyl
acetate.
2.3 Analytical Methods
Methods for the determination of deltamethrin residues and the
analysis of environmental samples, and products are summarized in
Table 3.
To analyse technical grade deltamethrin, a mixture of
deltamethrin and diphenylamine (an internal standard) was injected
in a high-performance liquid chromatograph equipped with a UV-
detector (Mourot et al., 1979).
The Joint FAO/WHO Codex Alimentarius Commission has published
recommendations for methods for the determination of deltamethrin
residues (FAO/WHO 1985b). A further review of analytical methods
for deltamethrin has been made by Vaysse et al. (1984).
Table 2. Chemical identity of deltamethrins of various stereoisomeric compositions
---------------------------------------------------------------------------------------------------------------
Common name CA Index name (9CI) Stereoisomeric Synonyms and trade names
CAS Registry No. compositionc
NIOSH Accession No.a Stereospecific nameb
---------------------------------------------------------------------------------------------------------------
Deltamethrin Cyclopropanecarboxylic acid, (4) Decamethrin, Decis,
52918-63-5 3-(2,2-dibromovinyl)-2,2-dimethyl-, K-Othrine, NRDC 161,
GZ1233000a alpha-cyano(3-phenoxyphenyl)methyl ester, WHO 1998, K-Obiol, Butox
[1R-[1 (S*), 3 R]]-, Butoflin, Cislin, FMC 45498
RU 22974
(S)-alpha-cyano-3-phenoxybenzyl
(1R, cis)-2,2-dimethyl-3-(2,2-di-
bromovinyl)cyclopropanecarboxylate
d- cis-Deltamethrin same as deltamethrin - Decamethrin, Decis
52820-00-5
GZ1240000a (S)-alpha-cyano-3-phenoxybenzyl
(d, cis)-2,2-dimethyl-3-(2,2-di-
bromovinyl)cyclopropanecarboxylate
---------------------------------------------------------------------------------------------------------------
a Registry of Toxic Effects of Chemical Substances (RTECS) (1981-82 edition).
b (1R), d, (+) or (1S), 1, (-) in the acid part of deltamethrin signifies the same stereospecific conformation,
respectively.
c The number in the parenthesis identifies the structure shown in the figures of stereoisomers.
Table 3. Analytical methods for deltamethrin
---------------------------------------------------------------------------------------------------------------------------------
Sample Extraction Sample preparation Determination: MDCb % Recovery Reference
solvent ----------------------------------- (mg/kg) (fortification
Partition Clean up GLC or HPLC level in
column elution condition; detector, mg/kg)
carrier flow, column,
temp, R.T.a
---------------------------------------------------------------------------------------------------------------------------------
RESIDUE ANALYSIS
apple n-hexane/ ext.sol.c/ silica gel CH2Cl2 ECD-GC; N2; 0.01 105(0.1), 100(1.0) 1
acetone H2O 50 ml/min; 1 m
(1/1) 3% OV-7; 235 °C
pear 0.01 125(0.1), 98(1.0)
cabbage 0.01 130(0.1), 118(1.0)
potato 0.01 126(0.1), 97(1.0)
apple, acetonitrile petroleum Florisil ether/ EDC-GC; 1.2 m 0.005 85-100(0.02-0.1) 2
peach, ether/H2O n-hexane DC-200, OV-1 or
grape, (1/4) OV-101; 245 °C,
tomato 10-12 min
wheat methanol n-hexane alumina HPLC; 235 nm; 87(2.0) 3
grain 30 cm; uBondapak;
C 18; methanol/H2O
(4/1); 2.5 ml/min
wheat n-hexane Florisil ether/ ECD-GC; N2; 91 4
petroleum 75 ml/min; 0.6 m
ether (1/9) 5% SE-30; 215 °C
meat ethyl ether/ acetonitrile gel diisopropyl ECD-GLC; N2; 0.001 90-95% at 0.01 5
petroleum permeation ether 40 ml/min; 1.8 m
ether column SE-30 1% on gas
(Styragel) Chrom. PAW
milk hexane acetonitrile Florisil + benzene/ ECD-GLC; N2; 0.01 83-87% at 0.1 5
cellulose/ hexane 40 ml/min; 1.8 m
charcoal (1/1) SE-30 1% on gas
Chrom. PAW
---------------------------------------------------------------------------------------------------------------------------------
Table 3. (contd.)
-----------------------------------------------------------------------------------------------------------------------------------
Sample Extraction Sample preparation Determination: MDCb % Recovery Reference
solvent ----------------------------------- (mg/kg) (fortification
Partition Clean up GLC or HPLC level in
column elution condition; detector, mg/kg)
carrier flow, column,
temp, R.T.a
-----------------------------------------------------------------------------------------------------------------------------------
ENVIRONMENTAL ANALYSIS
locust n-hexane Florisil ether/ ECD-GC; N2; 92 4
petroleum 75 ml/min; 0.6 m 5%
ether (1/9) SE-30; 215 °C
sea water XAD-2 ext.sol.c/ alumina ECD-GC; N2; 6
resin n-hexane 70 ml/min; 1.5 m
acetone 4% SE-30; 207 °C
water n-hexane alumina ECD-GC; N2; 6
70 ml/min; 1.5 m
4% SE-30; 207 °C
water petroleum Florisil petroleum ECD-GLC; 1 m OV 0.0001 97 at 0.010 8
ether/ ether/ 1-3% on Chromosorb
diethyl- diethyl- W.A.W. HMDS 60/80
ether (1/1) ether
(80/20)
soil acetone, acid hexane ECD-GLC; 5.2% 0.001 > 91% 9
acetone/ alumina ether OV-210 with AR/CH4
hexane (1/1) hexane
hexane (5-10%)
acetone, acid hexane/ ECD-GLC; N2; 0.0001 > 91% 5
acetone/ alumina ethyl ether 40 ml/min; 1.8 m
hexane (1/1) (90/10) SE-30 1% on gas
hexane Chrom. PAW
cotton n-hexane transesterification 7
foliage followed by ECD-GC;
(dislodgeable 31 ml/min; 0.45 m
residue) 5% SE-30; 120 °C
-----------------------------------------------------------------------------------------------------------------------------------
Table 3. (contd.)
-----------------------------------------------------------------------------------------------------------------------------------
Sample Extraction Sample preparation Determination: MDCb % Recovery Reference
solvent ----------------------------------- (mg/kg) (fortification
Partition Clean up GLC or HPLC level in
column elution condition; detector, mg/kg)
carrier flow, column,
temp, R.T.a
-----------------------------------------------------------------------------------------------------------------------------------
PRODUCT ANALYSIS
Technical HPLC, 230 nm; 15 cm 10
grade Lichrosorb Si-60;
n-hexane/diisopropyl
ether (93/7); 80 ml/h;
7.6 min
isoctane/ HPLC - UV detector 5
dioxane 254 nm (230 nm for
(80/20) conc. <0.5%) Silica-60;
100ml/h; isooctane/
dioxane (95/5)
-----------------------------------------------------------------------------------------------------------------------------------
a R.T.: retention time;
b MDC: minimum detectable concentration;
c ext .sol.: extraction solvent.
References
1. Baker & Bottomley (1982); 2. Mestres et al. (1978a); 3. Noble et al. (1982); 4. Pansu et al. (1981); 5. Vaysse et al. (1984);
6. Zitko et al. (1979); 7. Estesen et al. (1979); 8. Mestres et al. (1978b); 9. Hill (1982); 10. Mourot et al. (1979).
3. SOURCES OF ENVIRONMENTAL POLLUTION AND ENVIRONMENTAL LEVELS
3.1 Industrial Production
Deltamethrin was first marketed in 1977. Production volumes in
recent years are shown in Table 4.
Table 4. Worldwide production of deltamethrin
-------------------------------------------------
Year Production Reference
(tonnes)
-------------------------------------------------
1979 75 Wood Mackenzie (1980)
1980 100 Wood Mackenzie (1981)
1981 100 Wood Mackenzie (1982, 1983)
1982 115 Wood Mackenzie (1983)
1987 250 Information from Roussel Uclaf
-------------------------------------------------
3.2 Use Patterns
After an initial period when the product was mainly used on
cotton, several major crops were treated with deltamethrin from
1980 to 1987. Some 85% of the total production is used for crop
protection. Within this, 45% is used on cotton, 25%, on fruit and
vegetable crops, 20% on cereals, corn, and soybean, and the
remaining 10% on miscellaneous crops.
Deltamethrin is used to protect stored commodities (mainly
cereals, grains, coffee beans, dry beans), in forestry, and in
public health (e.g., Chagas disease control in South America, and
malaria control in Central America and on the African continent).
It is also used in animal facilities and against cattle
infestation.
It is formulated as an emulsifable concentrate (25 - 100
g/litre), an ultra-low-volume concentrate (1.5 - 30 g/litre), a
wettable powder (25 - 50 g/kg), a flowable powder (7.5 - 50
g/litre), or a dust powder (0.5 - 2.5 g/kg). It is also used in
combination with other pesticides and with piperonyl butoxide
(unpublished information from Roussel Uclaf to the IPCS, 1988).
3.3 Residues in Food
Supervised trials have been carried out on a wide variety of
crops and comprehensive summaries of analyses for residues in these
trials can be found in the evaluation reports of the Joint FAO/WHO
Meeting on Pesticide Residues (JMPR) (FAO/WHO 1981, 1982, 1983,
1985a, 1986a, 1986b, 1988b). A comprehensive list of maximum
residue limits (MRLs) for a large number of commodities resulted
from these evaluations (FAO/WHO, 1986c, 1988a,c) (see section 9).
Residues were determined in stored products, e.g., wheat,
maize, and coffee. The residue level in wheat grains treated with
deltamethrin at the rate of 2 mg/kg was 1.08 mg/kg after storage
for 9 months. When the wheat was subjected to milling and baking,
the residue levels in white bread were 0.11 mg/kg (Halls & Periam,
1980).
Mestres et. al. (1986) reviewed the changes in deltamethrin
residues in edible crops resulting from processing and cooking and
found that, depending on the commodity, pre- or post-harvest
residues were reduced by 20 - 98% by processing, and especially by
cooking.
When 0.27 g of 14C-(alcohol labelling)-deltamethrin was
injected intrarumenally in a lactating Jersey cow, in solution in a
sesame oil/alcohol mixture, only 0.4% of the compound was found in
whole milk. Peak residue levels of 0.045 and 0.92 mg/kg were found
in whole milk and rendered butter fat, respectively, 1 day after
administration. Residues in omental fat and leg muscle were 0.088
and 0.008 mg/kg, respectively, 2 days after treatment (Wellcome
Foundation, 1979).
3.4 Levels in the Environment
No information is available.
4. ENVIRONMENTAL TRANSPORT, DISTRIBUTION, AND TRANSFORMATION
4.1 Transport and Distribution Between Media
Using three different soils (silty clay, silty clay loam, and
loamy sand), Kaufman et al. (1981) found that deltamethrin was
practically immobile in soil columns. Approximately 96 - 97% of
the 14C activity remained in the upper 0 - 2.5 cm layer of the
columns with only 1.3 % in the 2.5 - 5.1 cm layer and no 14C in the
leachate. Soil thin layer chromatography (soil TLC) was also used
to evaluate the mobility of deltamethrin. According to the
pesticide mobility classification system developed by Helling &
Turner, deltamethrin is classified as a low-mobility to immobile
compound in soils.
The immobility of deltamethrin in soil was also studied by
Hascoet (1977) using a French Fontainebleau sand column leached
with a very high volume of water (equivalent to 1030 mm of rain).
In this experiment, approximately 97% of the applied 14C-
deltamethrin remained in the upper 0 - 2.5 cm layer and only 2% was
found in the leachate. The author concluded that deltamethrin was
unlikely to leach in cultivated soil that had a higher organic
matter content and/or higher clay contents than sand (organic
matter 0.03%), which has especially good filtration and low
adsorption properties.
The leaching of deltamethrin was also studied in three
different German soils the organic contents of which ranged from
0.8 to 2.6%. The study was carried out using the commercial
product Decis EC 25 at a rate equivalent to 1 litre/ha (i.e., 25 g
deltamethrin/ha). Each column was leached with 370 ml of water,
which was equivalent to a rainfall of 200 mm for 2 days. Under
these conditions, the amount of active ingredient (a.i.) detected
in seepage water was found to be less than 1 µg/ml, which was less
than 2% of the original applied dose (Thier & Schmidt, 1976).
The mobility of the primary deltamethrin degradation products
3-phenoxybenzoic acid (PBacid) and 3-phenoxybenzyl alcohol (PBalc)
was also investigated by Kaufman et al. (1981) using soil TLC and
soil columns. PBacid was found to be relatively mobile, whereas
PBalc was only slightly mobile. 2,2-Dimethyl-3-(2,2-dibromovinyl)
cyclopropanecarboxylic acid (Br2CA) was not studied in this
experiment, but Cl2CA, the chloride substituted analogue, was
evaluated and also found to be relatively mobile. However, these
metabolites did not accumulate in the soil to any extent, since
they were never in excess of 3% of the applied dose under the
aerobic conditions reported by Kaufman & Kayser (1979a,b). The
very significant production of 14CO2 during the incubation period
confirmed that they were further degraded.
4.2 Abiotic Degradation in Air and Water
Degradation pathways for deltamethrin are summarized in Fig. 1.
Deltamethrin is the first pyrethroid composed of a single
isomer of 8 stereoisomers selectively prepared by the
esterification of [1R, 3R or cis]-2,2-dimethyl-3-(2,2-dibromovinyl)
cyclopropanecarboxylic acid with (alphaS)- or (+)-alpha-cyano-3-
phenoxybenzyl alcohol or by selective recrystallization of the
racemic esters obtained by esterification of the (1R, 3R or cis)-
acid with the racemic or [alphaR, alphaS, or alphaRS or ±]-alcohol
(Elliott et al., 1974). Thus, its stereospecific structure (4) is
the ester of [1R, 3R or cis]-acid with (alphaS)-alcohol.
The acid is a characteristic dibromo analogue of chrysanthemic
acid.
2.2 Physical and Chemical Properties
Technical grade deltamethrin contains more than 98% deltamethrin
(FAO/WHO, 1981). It is stable to heat (6 months at 40 °C), light,
and air, but unstable in alkaline media (FAO/WHO, 1981; Meister et
al., 1983; Worthing & Walker, 1983). Some physical and chemical
properties are listed in Table 1, and the chemical composition of
various stereoisomeric mixtures is shown in Table 2.
Table 1. Some physical and chemical properties of deltamethrin
-------------------------------------------------------------------
Physical state crystalline powder
Colour colourless
Odour odourless
Density (20 °C) 0.5 g/cm3
Relative molecular mass 505.24
Melting point (°C) 98-101
Boiling point decomposes above 300 °C
Water solubility (20 °C) < 0.002 mg/litre (practically insoluble)
Solubility in organic solublea
solvents
Vapour pressure (25 °C) 2.0 x 10-6 Pa
n-Octanol-water 5.43
partition coefficient
(Log Pow)
-------------------------------------------------------------------
a Acetone (500 g/litre), ethanol (15 g/litre), cyclohexanone (750
g/litre), dioxane (900 g/litre), xylene (250 g/litre), ethyl
acetate.
2.3 Analytical Methods
Methods for the determination of deltamethrin residues and the
analysis of environmental samples, and products are summarized in
Table 3.
To analyse technical grade deltamethrin, a mixture of
deltamethrin and diphenylamine (an internal standard) was injected
in a high-performance liquid chromatograph equipped with a UV-
detector (Mourot et al., 1979).
The Joint FAO/WHO Codex Alimentarius Commission has published
recommendations for methods for the determination of deltamethrin
residues (FAO/WHO 1985b). A further review of analytical methods
for deltamethrin has been made by Vaysse et al. (1984).
Table 2. Chemical identity of deltamethrins of various stereoisomeric compositions
---------------------------------------------------------------------------------------------------------------
Common name CA Index name (9CI) Stereoisomeric Synonyms and trade names
CAS Registry No. compositionc
NIOSH Accession No.a Stereospecific nameb
---------------------------------------------------------------------------------------------------------------
Deltamethrin Cyclopropanecarboxylic acid, (4) Decamethrin, Decis,
52918-63-5 3-(2,2-dibromovinyl)-2,2-dimethyl-, K-Othrine, NRDC 161,
GZ1233000a alpha-cyano(3-phenoxyphenyl)methyl ester, WHO 1998, K-Obiol, Butox
[1R-[1 (S*), 3 R]]-, Butoflin, Cislin, FMC 45498
RU 22974
(S)-alpha-cyano-3-phenoxybenzyl
(1R, cis)-2,2-dimethyl-3-(2,2-di-
bromovinyl)cyclopropanecarboxylate
d- cis-Deltamethrin same as deltamethrin - Decamethrin, Decis
52820-00-5
GZ1240000a (S)-alpha-cyano-3-phenoxybenzyl
(d, cis)-2,2-dimethyl-3-(2,2-di-
bromovinyl)cyclopropanecarboxylate
---------------------------------------------------------------------------------------------------------------
a Registry of Toxic Effects of Chemical Substances (RTECS) (1981-82 edition).
b (1R), d, (+) or (1S), 1, (-) in the acid part of deltamethrin signifies the same stereospecific conformation,
respectively.
c The number in the parenthesis identifies the structure shown in the figures of stereoisomers.
Table 3. Analytical methods for deltamethrin
---------------------------------------------------------------------------------------------------------------------------------
Sample Extraction Sample preparation Determination: MDCb % Recovery Reference
solvent ----------------------------------- (mg/kg) (fortification
Partition Clean up GLC or HPLC level in
column elution condition; detector, mg/kg)
carrier flow, column,
temp, R.T.a
---------------------------------------------------------------------------------------------------------------------------------
RESIDUE ANALYSIS
apple n-hexane/ ext.sol.c/ silica gel CH2Cl2 ECD-GC; N2; 0.01 105(0.1), 100(1.0) 1
acetone H2O 50 ml/min; 1 m
(1/1) 3% OV-7; 235 °C
pear 0.01 125(0.1), 98(1.0)
cabbage 0.01 130(0.1), 118(1.0)
potato 0.01 126(0.1), 97(1.0)
apple, acetonitrile petroleum Florisil ether/ EDC-GC; 1.2 m 0.005 85-100(0.02-0.1) 2
peach, ether/H2O n-hexane DC-200, OV-1 or
grape, (1/4) OV-101; 245 °C,
tomato 10-12 min
wheat methanol n-hexane alumina HPLC; 235 nm; 87(2.0) 3
grain 30 cm; uBondapak;
C 18; methanol/H2O
(4/1); 2.5 ml/min
wheat n-hexane Florisil ether/ ECD-GC; N2; 91 4
petroleum 75 ml/min; 0.6 m
ether (1/9) 5% SE-30; 215 °C
meat ethyl ether/ acetonitrile gel diisopropyl ECD-GLC; N2; 0.001 90-95% at 0.01 5
petroleum permeation ether 40 ml/min; 1.8 m
ether column SE-30 1% on gas
(Styragel) Chrom. PAW
milk hexane acetonitrile Florisil + benzene/ ECD-GLC; N2; 0.01 83-87% at 0.1 5
cellulose/ hexane 40 ml/min; 1.8 m
charcoal (1/1) SE-30 1% on gas
Chrom. PAW
---------------------------------------------------------------------------------------------------------------------------------
Table 3. (contd.)
-----------------------------------------------------------------------------------------------------------------------------------
Sample Extraction Sample preparation Determination: MDCb % Recovery Reference
solvent ----------------------------------- (mg/kg) (fortification
Partition Clean up GLC or HPLC level in
column elution condition; detector, mg/kg)
carrier flow, column,
temp, R.T.a
-----------------------------------------------------------------------------------------------------------------------------------
ENVIRONMENTAL ANALYSIS
locust n-hexane Florisil ether/ ECD-GC; N2; 92 4
petroleum 75 ml/min; 0.6 m 5%
ether (1/9) SE-30; 215 °C
sea water XAD-2 ext.sol.c/ alumina ECD-GC; N2; 6
resin n-hexane 70 ml/min; 1.5 m
acetone 4% SE-30; 207 °C
water n-hexane alumina ECD-GC; N2; 6
70 ml/min; 1.5 m
4% SE-30; 207 °C
water petroleum Florisil petroleum ECD-GLC; 1 m OV 0.0001 97 at 0.010 8
ether/ ether/ 1-3% on Chromosorb
diethyl- diethyl- W.A.W. HMDS 60/80
ether (1/1) ether
(80/20)
soil acetone, acid hexane ECD-GLC; 5.2% 0.001 > 91% 9
acetone/ alumina ether OV-210 with AR/CH4
hexane (1/1) hexane
hexane (5-10%)
acetone, acid hexane/ ECD-GLC; N2; 0.0001 > 91% 5
acetone/ alumina ethyl ether 40 ml/min; 1.8 m
hexane (1/1) (90/10) SE-30 1% on gas
hexane Chrom. PAW
cotton n-hexane transesterification 7
foliage followed by ECD-GC;
(dislodgeable 31 ml/min; 0.45 m
residue) 5% SE-30; 120 °C
-----------------------------------------------------------------------------------------------------------------------------------
Table 3. (contd.)
-----------------------------------------------------------------------------------------------------------------------------------
Sample Extraction Sample preparation Determination: MDCb % Recovery Reference
solvent ----------------------------------- (mg/kg) (fortification
Partition Clean up GLC or HPLC level in
column elution condition; detector, mg/kg)
carrier flow, column,
temp, R.T.a
-----------------------------------------------------------------------------------------------------------------------------------
PRODUCT ANALYSIS
Technical HPLC, 230 nm; 15 cm 10
grade Lichrosorb Si-60;
n-hexane/diisopropyl
ether (93/7); 80 ml/h;
7.6 min
isoctane/ HPLC - UV detector 5
dioxane 254 nm (230 nm for
(80/20) conc. <0.5%) Silica-60;
100ml/h; isooctane/
dioxane (95/5)
-----------------------------------------------------------------------------------------------------------------------------------
a R.T.: retention time;
b MDC: minimum detectable concentration;
c ext .sol.: extraction solvent.
References
1. Baker & Bottomley (1982); 2. Mestres et al. (1978a); 3. Noble et al. (1982); 4. Pansu et al. (1981); 5. Vaysse et al. (1984);
6. Zitko et al. (1979); 7. Estesen et al. (1979); 8. Mestres et al. (1978b); 9. Hill (1982); 10. Mourot et al. (1979).
3. SOURCES OF ENVIRONMENTAL POLLUTION AND ENVIRONMENTAL LEVELS
3.1 Industrial Production
Deltamethrin was first marketed in 1977. Production volumes in
recent years are shown in Table 4.
Table 4. Worldwide production of deltamethrin
-------------------------------------------------
Year Production Reference
(tonnes)
-------------------------------------------------
1979 75 Wood Mackenzie (1980)
1980 100 Wood Mackenzie (1981)
1981 100 Wood Mackenzie (1982, 1983)
1982 115 Wood Mackenzie (1983)
1987 250 Information from Roussel Uclaf
-------------------------------------------------
3.2 Use Patterns
After an initial period when the product was mainly used on
cotton, several major crops were treated with deltamethrin from
1980 to 1987. Some 85% of the total production is used for crop
protection. Within this, 45% is used on cotton, 25%, on fruit and
vegetable crops, 20% on cereals, corn, and soybean, and the
remaining 10% on miscellaneous crops.
Deltamethrin is used to protect stored commodities (mainly
cereals, grains, coffee beans, dry beans), in forestry, and in
public health (e.g., Chagas disease control in South America, and
malaria control in Central America and on the African continent).
It is also used in animal facilities and against cattle
infestation.
It is formulated as an emulsifable concentrate (25 - 100
g/litre), an ultra-low-volume concentrate (1.5 - 30 g/litre), a
wettable powder (25 - 50 g/kg), a flowable powder (7.5 - 50
g/litre), or a dust powder (0.5 - 2.5 g/kg). It is also used in
combination with other pesticides and with piperonyl butoxide
(unpublished information from Roussel Uclaf to the IPCS, 1988).
3.3 Residues in Food
Supervised trials have been carried out on a wide variety of
crops and comprehensive summaries of analyses for residues in these
trials can be found in the evaluation reports of the Joint FAO/WHO
Meeting on Pesticide Residues (JMPR) (FAO/WHO 1981, 1982, 1983,
1985a, 1986a, 1986b, 1988b). A comprehensive list of maximum
residue limits (MRLs) for a large number of commodities resulted
from these evaluations (FAO/WHO, 1986c, 1988a,c) (see section 9).
Residues were determined in stored products, e.g., wheat,
maize, and coffee. The residue level in wheat grains treated with
deltamethrin at the rate of 2 mg/kg was 1.08 mg/kg after storage
for 9 months. When the wheat was subjected to milling and baking,
the residue levels in white bread were 0.11 mg/kg (Halls & Periam,
1980).
Mestres et. al. (1986) reviewed the changes in deltamethrin
residues in edible crops resulting from processing and cooking and
found that, depending on the commodity, pre- or post-harvest
residues were reduced by 20 - 98% by processing, and especially by
cooking.
When 0.27 g of 14C-(alcohol labelling)-deltamethrin was
injected intrarumenally in a lactating Jersey cow, in solution in a
sesame oil/alcohol mixture, only 0.4% of the compound was found in
whole milk. Peak residue levels of 0.045 and 0.92 mg/kg were found
in whole milk and rendered butter fat, respectively, 1 day after
administration. Residues in omental fat and leg muscle were 0.088
and 0.008 mg/kg, respectively, 2 days after treatment (Wellcome
Foundation, 1979).
3.4 Levels in the Environment
No information is available.
4. ENVIRONMENTAL TRANSPORT, DISTRIBUTION, AND TRANSFORMATION
4.1 Transport and Distribution Between Media
Using three different soils (silty clay, silty clay loam, and
loamy sand), Kaufman et al. (1981) found that deltamethrin was
practically immobile in soil columns. Approximately 96 - 97% of
the 14C activity remained in the upper 0 - 2.5 cm layer of the
columns with only 1.3 % in the 2.5 - 5.1 cm layer and no 14C in the
leachate. Soil thin layer chromatography (soil TLC) was also used
to evaluate the mobility of deltamethrin. According to the
pesticide mobility classification system developed by Helling &
Turner, deltamethrin is classified as a low-mobility to immobile
compound in soils.
The immobility of deltamethrin in soil was also studied by
Hascoet (1977) using a French Fontainebleau sand column leached
with a very high volume of water (equivalent to 1030 mm of rain).
In this experiment, approximately 97% of the applied 14C-
deltamethrin remained in the upper 0 - 2.5 cm layer and only 2% was
found in the leachate. The author concluded that deltamethrin was
unlikely to leach in cultivated soil that had a higher organic
matter content and/or higher clay contents than sand (organic
matter 0.03%), which has especially good filtration and low
adsorption properties.
The leaching of deltamethrin was also studied in three
different German soils the organic contents of which ranged from
0.8 to 2.6%. The study was carried out using the commercial
product Decis EC 25 at a rate equivalent to 1 litre/ha (i.e., 25 g
deltamethrin/ha). Each column was leached with 370 ml of water,
which was equivalent to a rainfall of 200 mm for 2 days. Under
these conditions, the amount of active ingredient (a.i.) detected
in seepage water was found to be less than 1 µg/ml, which was less
than 2% of the original applied dose (Thier & Schmidt, 1976).
The mobility of the primary deltamethrin degradation products
3-phenoxybenzoic acid (PBacid) and 3-phenoxybenzyl alcohol (PBalc)
was also investigated by Kaufman et al. (1981) using soil TLC and
soil columns. PBacid was found to be relatively mobile, whereas
PBalc was only slightly mobile. 2,2-Dimethyl-3-(2,2-dibromovinyl)
cyclopropanecarboxylic acid (Br2CA) was not studied in this
experiment, but Cl2CA, the chloride substituted analogue, was
evaluated and also found to be relatively mobile. However, these
metabolites did not accumulate in the soil to any extent, since
they were never in excess of 3% of the applied dose under the
aerobic conditions reported by Kaufman & Kayser (1979a,b). The
very significant production of 14CO2 during the incubation period
confirmed that they were further degraded.
4.2 Abiotic Degradation in Air and Water
Degradation pathways for deltamethrin are summarized in Fig. 1.
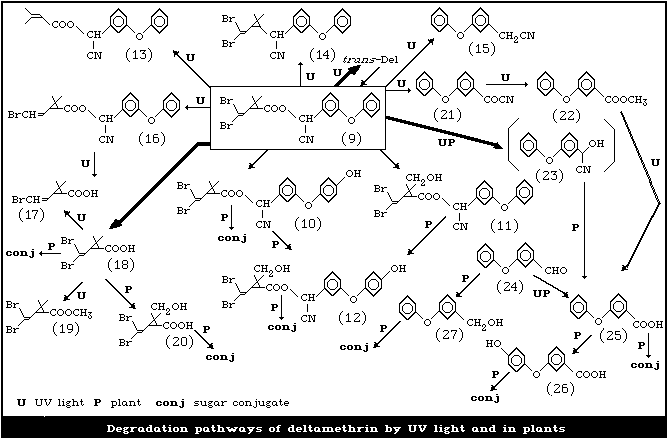 When 14C-deltamethrin-[1R, 3R; alphaS] (9) labelled at the
cyano, benzylic, or dibromo-substituted carbon was exposed to
sunlight as a thin film (40 µg/cm2) for 4 - 8 h, the trans-[1R,
3S; alphaS] and -[1S, 3R; alphaS] isomers were formed. They
accounted for approximately 70% of the applied radioactivity.
Smaller amounts of ester cleavage products including the
2,2-dimethyl-3-(2,2-dibromovinyl) cyclopropanecarboxylic acid
(Br2CA) (18) and the cyanohydrin component, and 18% of
unidentified products were also formed (Fig. 1). In a thick film (3
mg/cm2), small amounts of other products including
alpha-cyano-3-phenoxybenzyl 3,3-dimethylacrylate (13) and 3-phenoxy
2,2-dimethyl-3-(2,2- dibromovinyl)cyclopropan-1-yl-benzylcyanide
(14) (decarboxydeltamethrin) were also detected. In contrast, the
predominant products in methanol were the trans mixtures, which
amounted to approximately 35% of the applied radioactivity. Under
UV radiation (peak output 290 - 320 nm), the photodegradation rate
of deltamethrin in alcohols decreased in the order of methanol,
ethanol, and 2-propanol, as the solvent viscosity increased. The
relative photolysis rates in hexane and cyclohexane, with respective
relative viscosities of 0.33 and 1, were 1.5 and 1. There was no
difference in the extent of the reaction on flushing the hexane with
O2 or N2, while the triplet quenchers piperilene and
1,3-cyclohexadiene reduced the reaction rate in hexane.
At 30 - 50% conversion, the trans-[1R, 3S; alphaS] and -[1S,
3R; alphaS] isomers were the major photoproducts in aqueous
acetonitrile, whereas they were observed in only minor amounts in
methanol and were absent in hexane. The mono-debrominated esters
(16) were the major ester products in methanol and hexane. The
cis-acid (18) was always the major photoproduct from the acid
moiety, with smaller amounts of the two isomeric debrominated acids
(17).
Major products from the alcohol moiety were 3-phenoxybenzoic
acid (25) (PBacid) in aqueous acetonitrile, 3-phenoxybenzyl cyanide
(15) in hexane, and methyl 3-phenoxybenzoate (22) in methanol.
Photolysis of 3-phenoxybenzoyl cyanide (21) gave methyl
3-phenoxybenzoate and the methyl ester of Br2CA (19) in methanol
and PBacid in aqueous acetonitrile. Thus, it appears that the
photoproducts obtained originated from cyclopropane ring opening
and various recombinations, scission of the ester oxygen-benzyl
carbon bond, scission of the acyl-oxygen bond, and/or reductive
debromination (Ruzo et al., 1976, 1977).
A photodegradation study with 14C-deltamethrin in aqueous
solution showed that such a solution, at pH 5, is hydrolytically
stable. When exposed to simulated sunlight, degradation was
induced. The primary product observed was PBacid. A half-life of
47.7 days was calculated for the non-sensitized system, but this
was reduced to 4.03 days when sensitized with 1% acetone.
Practically no volatile degradation products were observed (Bowman
& Carpenter, 1987).
4.3 Environmental Fate
The degradation and persistence of 14C-cyano- and 14C-phenoxy-
deltamethrin was examined in a Dubbs fine sandy loam and a Memphis
silt loam under aerobic laboratory conditions at 25 °C (Kaufman &
Kayser 1979a); 14C-deltamethrin was applied at final concentrations
equivalent to 0.02 and 0.2 kg/ha. Deltamethrin degradation
occurred rapidly in both soils with 62 - 77% and 52 - 60% of the
14C-cyano- and 14C-phenoxy-labels, respectively, being evolved as
14CO2 during the 128-day incubation period. The half-life of
deltamethrin varied from 11 to 19 days in the two soil types.
The effect of soil temperature on the degradation of
deltamethrin was also examined in Dubbs fine sandy loam under
laboratory conditions using 14C-cyano- and 14C-vinyl-labelled
deltamethrin (Kaufman & Kayser 1979b). Degradation and evolution
of 14C-labelled forms of deltamethrin occurred most rapidly at
25 °C and most slowly in soils incubated at 10 °C. The half-life
of deltamethrin was 46, 13, and 27 days in soils incubated at 10,
25, and 40 °C, respectively.
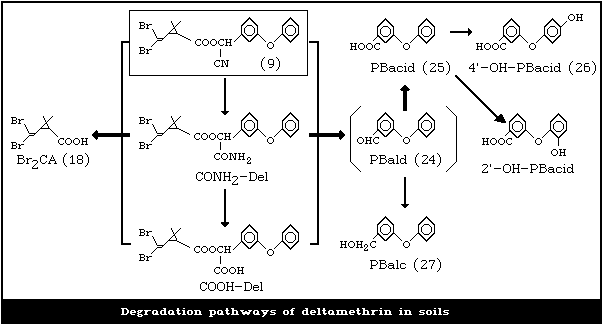
The results of these two studies indicate that deltamethrin
degradation occurs by two principal pathways (Fig. 2): hydrolysis
of the ester linkage to yield Br2CA (18) and 3-phenoxybenzoic acid;
and hydrolysis of the cyano group to yield first the amide, and
subsequently the carboxylic acid (DCOOH) analogues of deltamethrin.
Br2CA accumulated to a maximum of 5.7% of the original 14C in soil
incubated at 40 °C, whereas DCOOH accumulated at 10 °C (to a
maximum of 5.3%). However, both products decreased in
concentration by the end of the 64-day incubation period. In the
first experiment, DCOOH was also identified as the major
degradation product to reach a maximum concentration of 6 - 9% of
the original 14C. But it ultimately dissipated to less than 2% at
the end of the 128-day incubation period.
From the 14C-phenoxy label, 3-phenoxybenzoic acid (PBacid) was
identified as the main degradation product resulting from
hydrolysis of the ester bond. This product was further degraded to
yield both 3-(2-hydroxyphenoxy)-benzoic acid and 3-(4-hydroxyphenoxy)
benzoic acid. In this experiment, DCOOH was the only deltamethrin
degradation product detected in excess of 3% of the original material
applied.
Although essentially no radiolabel was detected in the leachate
from soil columns treated with 14C-deltamethrin, PBacid produced by
degradation of deltamethrin was fairly mobile in the soil columns
(Kaufman et al., 1981).
The degradation pathways are proposed in Fig. 2.
When 14C-deltamethrin-[1R, 3R; alphaS] (9) labelled at the
cyano, benzylic, or dibromo-substituted carbon was exposed to
sunlight as a thin film (40 µg/cm2) for 4 - 8 h, the trans-[1R,
3S; alphaS] and -[1S, 3R; alphaS] isomers were formed. They
accounted for approximately 70% of the applied radioactivity.
Smaller amounts of ester cleavage products including the
2,2-dimethyl-3-(2,2-dibromovinyl) cyclopropanecarboxylic acid
(Br2CA) (18) and the cyanohydrin component, and 18% of
unidentified products were also formed (Fig. 1). In a thick film (3
mg/cm2), small amounts of other products including
alpha-cyano-3-phenoxybenzyl 3,3-dimethylacrylate (13) and 3-phenoxy
2,2-dimethyl-3-(2,2- dibromovinyl)cyclopropan-1-yl-benzylcyanide
(14) (decarboxydeltamethrin) were also detected. In contrast, the
predominant products in methanol were the trans mixtures, which
amounted to approximately 35% of the applied radioactivity. Under
UV radiation (peak output 290 - 320 nm), the photodegradation rate
of deltamethrin in alcohols decreased in the order of methanol,
ethanol, and 2-propanol, as the solvent viscosity increased. The
relative photolysis rates in hexane and cyclohexane, with respective
relative viscosities of 0.33 and 1, were 1.5 and 1. There was no
difference in the extent of the reaction on flushing the hexane with
O2 or N2, while the triplet quenchers piperilene and
1,3-cyclohexadiene reduced the reaction rate in hexane.
At 30 - 50% conversion, the trans-[1R, 3S; alphaS] and -[1S,
3R; alphaS] isomers were the major photoproducts in aqueous
acetonitrile, whereas they were observed in only minor amounts in
methanol and were absent in hexane. The mono-debrominated esters
(16) were the major ester products in methanol and hexane. The
cis-acid (18) was always the major photoproduct from the acid
moiety, with smaller amounts of the two isomeric debrominated acids
(17).
Major products from the alcohol moiety were 3-phenoxybenzoic
acid (25) (PBacid) in aqueous acetonitrile, 3-phenoxybenzyl cyanide
(15) in hexane, and methyl 3-phenoxybenzoate (22) in methanol.
Photolysis of 3-phenoxybenzoyl cyanide (21) gave methyl
3-phenoxybenzoate and the methyl ester of Br2CA (19) in methanol
and PBacid in aqueous acetonitrile. Thus, it appears that the
photoproducts obtained originated from cyclopropane ring opening
and various recombinations, scission of the ester oxygen-benzyl
carbon bond, scission of the acyl-oxygen bond, and/or reductive
debromination (Ruzo et al., 1976, 1977).
A photodegradation study with 14C-deltamethrin in aqueous
solution showed that such a solution, at pH 5, is hydrolytically
stable. When exposed to simulated sunlight, degradation was
induced. The primary product observed was PBacid. A half-life of
47.7 days was calculated for the non-sensitized system, but this
was reduced to 4.03 days when sensitized with 1% acetone.
Practically no volatile degradation products were observed (Bowman
& Carpenter, 1987).
4.3 Environmental Fate
The degradation and persistence of 14C-cyano- and 14C-phenoxy-
deltamethrin was examined in a Dubbs fine sandy loam and a Memphis
silt loam under aerobic laboratory conditions at 25 °C (Kaufman &
Kayser 1979a); 14C-deltamethrin was applied at final concentrations
equivalent to 0.02 and 0.2 kg/ha. Deltamethrin degradation
occurred rapidly in both soils with 62 - 77% and 52 - 60% of the
14C-cyano- and 14C-phenoxy-labels, respectively, being evolved as
14CO2 during the 128-day incubation period. The half-life of
deltamethrin varied from 11 to 19 days in the two soil types.
The effect of soil temperature on the degradation of
deltamethrin was also examined in Dubbs fine sandy loam under
laboratory conditions using 14C-cyano- and 14C-vinyl-labelled
deltamethrin (Kaufman & Kayser 1979b). Degradation and evolution
of 14C-labelled forms of deltamethrin occurred most rapidly at
25 °C and most slowly in soils incubated at 10 °C. The half-life
of deltamethrin was 46, 13, and 27 days in soils incubated at 10,
25, and 40 °C, respectively.
The results of these two studies indicate that deltamethrin
degradation occurs by two principal pathways (Fig. 2): hydrolysis
of the ester linkage to yield Br2CA (18) and 3-phenoxybenzoic acid;
and hydrolysis of the cyano group to yield first the amide, and
subsequently the carboxylic acid (DCOOH) analogues of deltamethrin.
Br2CA accumulated to a maximum of 5.7% of the original 14C in soil
incubated at 40 °C, whereas DCOOH accumulated at 10 °C (to a
maximum of 5.3%). However, both products decreased in
concentration by the end of the 64-day incubation period. In the
first experiment, DCOOH was also identified as the major
degradation product to reach a maximum concentration of 6 - 9% of
the original 14C. But it ultimately dissipated to less than 2% at
the end of the 128-day incubation period.
From the 14C-phenoxy label, 3-phenoxybenzoic acid (PBacid) was
identified as the main degradation product resulting from
hydrolysis of the ester bond. This product was further degraded to
yield both 3-(2-hydroxyphenoxy)-benzoic acid and 3-(4-hydroxyphenoxy)
benzoic acid. In this experiment, DCOOH was the only deltamethrin
degradation product detected in excess of 3% of the original material
applied.
Although essentially no radiolabel was detected in the leachate
from soil columns treated with 14C-deltamethrin, PBacid produced by
degradation of deltamethrin was fairly mobile in the soil columns
(Kaufman et al., 1981).
The degradation pathways are proposed in Fig. 2.
 The degradation of deltamethrin was also examined under
anaerobic conditions using 14C-cyano-, 14C-phenoxy-, and
14C-vinyl-labelled materials for the tests (Kaufman & Kayser,
1980). Under anaerobic conditions, 14CO2 evolution varied
according to the 14C label position and the time of flooding.
Generally, flooding reduced or initially inhibited the rate of
14CO2 dissipation. However, after one month, 14CO2 dissipation
started again, which suggested the presence of a unique microbial
flora. It was also shown that all three carboxylic acids that
accumulate initially in flooded soils are subsequently further
degraded. Some reduction of PBacid to 3-phenoxybenzyl alcohol
(PBalc) was also observed in these flooded soils.
When deltamethrin was applied to a sandy clay loam soil at
17.5 g/ha in an indoor incubation study and in two field
experiments, the half-lives of deltamethrin were found to be 4.9
and 6.9 weeks under indoor and field conditions, respectively
(Hill, 1983). This difference in the rate of decrease in the
residue was attributed to climatic effects.
This was further confirmed by Hill & Schaalje (1985) who
pointed out a first-order dissipation, if degree-days above 0 °C
rather than days was used as the independent variable, when
deltamethrin was applied by pipette to soils. When deltamethrin
was boom-sprayed, a biphasic first-order plot was observed. A two-
compartment model that predicts an initial fast loss of residue
followed by a slower first-order degradation gave a good fit of the
data.
Chapman & Harris (1981) examined the relative persistence of
five pyrethroids, permethrin, cypermethrin, deltamethrin,
fenpropathrin, and fenvalerate, in sand and organic soil at 28 °C,
under laboratory conditions. All of the insecticides (1 mg/kg) were
degraded more rapidly in natural soils than in sterilized soils,
suggesting the importance of microbial degradation. The rate of
degradation under non-sterilized conditions decreased as follows:
fenpropathrin > permethrin > cypermethrin > fenvalerate >
deltamethrin. Amounts of approximately 52% and 74% of the
deltamethrin applied were recovered from the sand and organic soil,
respectively, 8 weeks after treatment.
It was pointed out by Chapman et al. (1981) that biological
processes played a major role in the degradation of deltamethrin in
soils.
The degradation of deltamethrin was also investigated by Zhang
et al. (1984) in an organic soil over a 180-day period. The half-
life of deltamethrin was found to be 72 days, indicating that
deltamethrin is likely to be less susceptible to degradation in
organic soils than in mineral soils. Identification of metabolites
present in the extractable phase confirmed the metabolic pathways
previously reported by Kaufman. Levels of bound 14C residues
increased with the incubation period to reach 19% of the original
14C after 180 days. Most of these bound 14C residues were in the
humic fraction. Bacterial and actinomycete populations increased
in the treated soil, but fungal populations remained relatively
stable during the incubation period.
The degradation of deltamethrin was also studied in two German
soils. Half-lives for sandy soil and sandy loam soil were 35 and
60 days, respectively (Thier & Schmidt, 1977).
All these studies demonstrate that deltamethrin is readily and
quickly degraded in the soil. The half-life of the compound
depends on the nature of the soil as well as the temperature.
Generally speaking, the half-life ranges from 11 to 72 days, under
aerobic conditions. Deltamethrin degradation is slower under
anaerobic or sterile conditions, indicating that microorganisms and
other biological processes play a very important role.
The metabolism of deltamethrin in cotton plants was studied
using material 14C-labelled at the dibromovinyl, benzylic, and
cyano carbons. Under glasshouse conditions, the initial half-life
of deltamethrin was 1.1 weeks and the time needed for 90% loss was
4.6 weeks. Conversion of deltamethrin to the trans-isomer occurred
via photochemical reactions and, after 6 weeks, the trans/cis ratio
was 0.44:1. Deltamethrin degraded more rapidly under field
conditions to give a higher proportion of trans- to cis-isomers and
large amounts of unextractable products. Trace amounts of three
deltamethrin derivatives hydroxylated either at the 4'-position
(10), or at the trans-methyl relative to the carboxy group in the
acid moiety (7), or at both sites (12) were detected with all three
14C preparations (Fig. 1). However, the major metabolites were
free and conjugated Br2CA together with small quantities of the
trans-hydroxymethyl derivatives (20) of Br2CA and 3-(4-
hydroxyphenoxy) benzoic acid (26). The above compounds were
analogues of those formed from permethrin and cypermethrin in
plants. Several types of conjugated metabolites were isolated, but
they were not fully characterized. One type was cleaved readily
with beta-glucosidase or hydrogen chloride to yield Br2CA and
PBacid. Two other types were resistant to beta-glucosidase, but
cleaved readily with hydrogen chloride to yield Br2CA (from the
dibromovinyl label) and 3-phenoxybenzoic acid, 3-phenoxybenzyl
alcohol (from the alcohol label), and alpha-cyano-3-phenoxybenzyl
alcohol (from the cyano and alcohol labels). The metabolites of
deltamethrin identified in plants were analogous to those in
mammals, except for the conjugated products.
The metabolism of deltamethrin and its degradation products in
cotton and bean leaf disks has also been studied. Limited
conversion (approximately 6%) of deltamethrin occurred to give
Br2CA and 3-phenoxybenzyl alcohol (27) (PBalc) conjugates. The
ester cleavage products used as substrates underwent more extensive
metabolism, and two to three types of glucosides were formed from
Br2CA and four from PBalc. 3-Phenoxybenzaldehyde (24),
administered directly or as the cyanohydrin (23), was reduced to
PBalc, though part was oxidized to PBacid (Ruzo & Casida, 1979).
4.4 Bioaccumulation
Bioaccumulation studies with fish, have shown that pyrethroids
have bioconcentration factors (BCFs) that are far lower than those
predicted from the correlation between the Kow partition
coefficient and BCF. The low accumulation can be attributed to
metabolism by the fish and to the reduced bioavailability to fish
of deltamethrin bound by dissolved organic carbon and suspended
colloids. Metabolic kinetics were assessed by Cary (1978) in
Ictalurus punctatus maintained for 30 days in the water of a
hydrosoil system, in which the soil was treated with a dose of
125 g a.i./ha (10 times the normal agricultural dose) and then
flooded after 31 days. During the exposure period, none of the
300 fish died or behaved abnormally despite a final deltamethrin
concentration of 2.19 µg/litre, which is more than 3 times the
acute 96-h LC50 of 0.63 µg/litre (Table 6). During a third phase,
fish were introduced into an uncontaminated liquid medium,
continuously renewed, to monitor elimination of deltamethrin or its
metabolites. The main results are given in Table 5.
Table 5. Bioaccumulation factors after exposure of Ictalurus
punctatus and depuration kineticsa
----------------------------------------------------------------
Organ Value of bioconcentration 14C elimination (%)
factor (BCF)b during after depuration of
exposure, 30 days -------------------
1 day 14 days
----------------------------------------------------------------
muscles 25 <50 77
viscera 972 67 86
carcasses 41 >50 93
body as a whole 144 >50 93
----------------------------------------------------------------
a From: Cary (1978).
b BCF: µg/kg concentration in fish/µg/litre concentration in
water.
Muir et al. (1985) monitored the fate and uptake of
14C-labelled deltamethrin in organisms in experimental ponds over
306 days. Initial concentrations of the pyrethroid ranged from 1.8
to 2.5 µg/litre. The deltamethrin rapidly became distributed in
suspended solids, plants, sediment, and air with a half-life of
2 - 4 h in the water. Aquatic plants (the floating duckweed Lemna
sp. and a submerged/floating weed (Potomageton berchtoldi)
accumulated deltamethrin at concentrations of between 253 and
1021 µg/kg, respectively, 24 h after treatment, but the compound
had all disappeared within 14 days. Fathead minnows, Pimephales
promelas, showed bioconcentration factors of 248 - 907. Although
radioactivity remained in the fish throughout the experimental
period, presumably in the fat, the levels fell steadily and no
effects were seen on the fish.
5. KINETICS AND METABOLISM
5.1 Metabolism in Experimental Animals
Metabolic pathways of deltamethrin in mammals are summarized in
Fig. 3.
The degradation of deltamethrin was also examined under
anaerobic conditions using 14C-cyano-, 14C-phenoxy-, and
14C-vinyl-labelled materials for the tests (Kaufman & Kayser,
1980). Under anaerobic conditions, 14CO2 evolution varied
according to the 14C label position and the time of flooding.
Generally, flooding reduced or initially inhibited the rate of
14CO2 dissipation. However, after one month, 14CO2 dissipation
started again, which suggested the presence of a unique microbial
flora. It was also shown that all three carboxylic acids that
accumulate initially in flooded soils are subsequently further
degraded. Some reduction of PBacid to 3-phenoxybenzyl alcohol
(PBalc) was also observed in these flooded soils.
When deltamethrin was applied to a sandy clay loam soil at
17.5 g/ha in an indoor incubation study and in two field
experiments, the half-lives of deltamethrin were found to be 4.9
and 6.9 weeks under indoor and field conditions, respectively
(Hill, 1983). This difference in the rate of decrease in the
residue was attributed to climatic effects.
This was further confirmed by Hill & Schaalje (1985) who
pointed out a first-order dissipation, if degree-days above 0 °C
rather than days was used as the independent variable, when
deltamethrin was applied by pipette to soils. When deltamethrin
was boom-sprayed, a biphasic first-order plot was observed. A two-
compartment model that predicts an initial fast loss of residue
followed by a slower first-order degradation gave a good fit of the
data.
Chapman & Harris (1981) examined the relative persistence of
five pyrethroids, permethrin, cypermethrin, deltamethrin,
fenpropathrin, and fenvalerate, in sand and organic soil at 28 °C,
under laboratory conditions. All of the insecticides (1 mg/kg) were
degraded more rapidly in natural soils than in sterilized soils,
suggesting the importance of microbial degradation. The rate of
degradation under non-sterilized conditions decreased as follows:
fenpropathrin > permethrin > cypermethrin > fenvalerate >
deltamethrin. Amounts of approximately 52% and 74% of the
deltamethrin applied were recovered from the sand and organic soil,
respectively, 8 weeks after treatment.
It was pointed out by Chapman et al. (1981) that biological
processes played a major role in the degradation of deltamethrin in
soils.
The degradation of deltamethrin was also investigated by Zhang
et al. (1984) in an organic soil over a 180-day period. The half-
life of deltamethrin was found to be 72 days, indicating that
deltamethrin is likely to be less susceptible to degradation in
organic soils than in mineral soils. Identification of metabolites
present in the extractable phase confirmed the metabolic pathways
previously reported by Kaufman. Levels of bound 14C residues
increased with the incubation period to reach 19% of the original
14C after 180 days. Most of these bound 14C residues were in the
humic fraction. Bacterial and actinomycete populations increased
in the treated soil, but fungal populations remained relatively
stable during the incubation period.
The degradation of deltamethrin was also studied in two German
soils. Half-lives for sandy soil and sandy loam soil were 35 and
60 days, respectively (Thier & Schmidt, 1977).
All these studies demonstrate that deltamethrin is readily and
quickly degraded in the soil. The half-life of the compound
depends on the nature of the soil as well as the temperature.
Generally speaking, the half-life ranges from 11 to 72 days, under
aerobic conditions. Deltamethrin degradation is slower under
anaerobic or sterile conditions, indicating that microorganisms and
other biological processes play a very important role.
The metabolism of deltamethrin in cotton plants was studied
using material 14C-labelled at the dibromovinyl, benzylic, and
cyano carbons. Under glasshouse conditions, the initial half-life
of deltamethrin was 1.1 weeks and the time needed for 90% loss was
4.6 weeks. Conversion of deltamethrin to the trans-isomer occurred
via photochemical reactions and, after 6 weeks, the trans/cis ratio
was 0.44:1. Deltamethrin degraded more rapidly under field
conditions to give a higher proportion of trans- to cis-isomers and
large amounts of unextractable products. Trace amounts of three
deltamethrin derivatives hydroxylated either at the 4'-position
(10), or at the trans-methyl relative to the carboxy group in the
acid moiety (7), or at both sites (12) were detected with all three
14C preparations (Fig. 1). However, the major metabolites were
free and conjugated Br2CA together with small quantities of the
trans-hydroxymethyl derivatives (20) of Br2CA and 3-(4-
hydroxyphenoxy) benzoic acid (26). The above compounds were
analogues of those formed from permethrin and cypermethrin in
plants. Several types of conjugated metabolites were isolated, but
they were not fully characterized. One type was cleaved readily
with beta-glucosidase or hydrogen chloride to yield Br2CA and
PBacid. Two other types were resistant to beta-glucosidase, but
cleaved readily with hydrogen chloride to yield Br2CA (from the
dibromovinyl label) and 3-phenoxybenzoic acid, 3-phenoxybenzyl
alcohol (from the alcohol label), and alpha-cyano-3-phenoxybenzyl
alcohol (from the cyano and alcohol labels). The metabolites of
deltamethrin identified in plants were analogous to those in
mammals, except for the conjugated products.
The metabolism of deltamethrin and its degradation products in
cotton and bean leaf disks has also been studied. Limited
conversion (approximately 6%) of deltamethrin occurred to give
Br2CA and 3-phenoxybenzyl alcohol (27) (PBalc) conjugates. The
ester cleavage products used as substrates underwent more extensive
metabolism, and two to three types of glucosides were formed from
Br2CA and four from PBalc. 3-Phenoxybenzaldehyde (24),
administered directly or as the cyanohydrin (23), was reduced to
PBalc, though part was oxidized to PBacid (Ruzo & Casida, 1979).
4.4 Bioaccumulation
Bioaccumulation studies with fish, have shown that pyrethroids
have bioconcentration factors (BCFs) that are far lower than those
predicted from the correlation between the Kow partition
coefficient and BCF. The low accumulation can be attributed to
metabolism by the fish and to the reduced bioavailability to fish
of deltamethrin bound by dissolved organic carbon and suspended
colloids. Metabolic kinetics were assessed by Cary (1978) in
Ictalurus punctatus maintained for 30 days in the water of a
hydrosoil system, in which the soil was treated with a dose of
125 g a.i./ha (10 times the normal agricultural dose) and then
flooded after 31 days. During the exposure period, none of the
300 fish died or behaved abnormally despite a final deltamethrin
concentration of 2.19 µg/litre, which is more than 3 times the
acute 96-h LC50 of 0.63 µg/litre (Table 6). During a third phase,
fish were introduced into an uncontaminated liquid medium,
continuously renewed, to monitor elimination of deltamethrin or its
metabolites. The main results are given in Table 5.
Table 5. Bioaccumulation factors after exposure of Ictalurus
punctatus and depuration kineticsa
----------------------------------------------------------------
Organ Value of bioconcentration 14C elimination (%)
factor (BCF)b during after depuration of
exposure, 30 days -------------------
1 day 14 days
----------------------------------------------------------------
muscles 25 <50 77
viscera 972 67 86
carcasses 41 >50 93
body as a whole 144 >50 93
----------------------------------------------------------------
a From: Cary (1978).
b BCF: µg/kg concentration in fish/µg/litre concentration in
water.
Muir et al. (1985) monitored the fate and uptake of
14C-labelled deltamethrin in organisms in experimental ponds over
306 days. Initial concentrations of the pyrethroid ranged from 1.8
to 2.5 µg/litre. The deltamethrin rapidly became distributed in
suspended solids, plants, sediment, and air with a half-life of
2 - 4 h in the water. Aquatic plants (the floating duckweed Lemna
sp. and a submerged/floating weed (Potomageton berchtoldi)
accumulated deltamethrin at concentrations of between 253 and
1021 µg/kg, respectively, 24 h after treatment, but the compound
had all disappeared within 14 days. Fathead minnows, Pimephales
promelas, showed bioconcentration factors of 248 - 907. Although
radioactivity remained in the fish throughout the experimental
period, presumably in the fat, the levels fell steadily and no
effects were seen on the fish.
5. KINETICS AND METABOLISM
5.1 Metabolism in Experimental Animals
Metabolic pathways of deltamethrin in mammals are summarized in
Fig. 3.
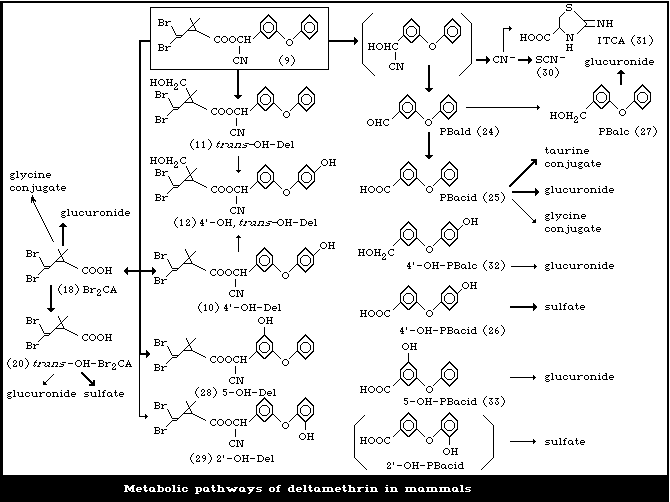 After oral administration to male rats at 0.64 - 1.60 mg/kg,
the acid and alcohol moieties of deltamethrin were almost
completely eliminated from the body within 2 - 4 days (Ruzo et al.,
1978). On the other hand, the cyano group was eliminated more
slowly, the total recovery during 8 days being 79% of the
radiocarbon dose (43% and 36% in the urine and faeces,
respectively). Tissue residues of deltamethrin labelled with 14C
at the dibromovinyl carbon in the acid moiety and the benzylic
carbon in the alcohol moiety were generally very low, whereas
residue levels in the fat were somewhat higher (0.1 - 0.2 mg/kg).
Residue levels of the radiocarbon derived from the cyano group were
relatively high, especially in the skin and stomach. Essentially,
all the radiocarbon in the stomach was thiocyanate. No noticeable
14CO2 was evolved from any of the radioactive preparations,
including the CN-labelled group, in contrast to the CN group from
fenvalerate, which yielded 14CO2 in considerable amounts.
The major metabolic reactions of deltamethrin were oxidation
(at the trans methyl relative to carbonyl group of the acid moiety
and at the 2'-, 4'-, and 5-positions of the alcohol moiety),
cleavage of the ester linkage, and conversion of the cyano portion
to thiocyanate and 2-iminothiazolidine-4-carboxylic acid (31)
(ITCA) (see Fig. 3). These carboxylic acid and phenol derivatives
were conjugated with sulfuric acid, glycine, and/or glucuronic
acid.
The major faecal metabolites were unchanged deltamethrin (9),
accounting for 13 - 21% of the dose, followed by 4'-OH- (10) and
5-OH-deltamethrin (28), and a trace amount of 2'-OH-deltamethrin
(29). Intact deltamethrin and the 4'-OH-derivative appeared not
only as the administered S-epimer, but also in parts as the
R-epimer, probably due to artefactural racemization on exchange of
the alpha-position hydrogen in methanol solution. The metabolites
from the acid moiety were mostly 3-(2,2-dibromovinyl)-2,2-
dimethylcyclopropanecarboxylic acid (18) (Br2CA) in free form
(10% of the dose), glucuronide (51%) and glycine (trace level)
conjugates, and OH-Br2CA (20) in free form and glucuronide
conjugate (<1%).
The major metabolites of the aromatic portion of the alcohol
moiety were 3-phenoxybenzoic acid (25) (PBacid) in free form (5%),
and glucuronide (13%) and glycine (4%) conjugates and its
4'-hydroxy derivative (26) (4'-OH-PBacid).
Sulfate of 4'-OH-PBacid accounted for about 50% of the dose,
together with small amounts of free (4%) and glucuronide forms
(2%). The CN group was converted mainly to thiocyanate (30) and,
in small amounts, to ITCA (31) (Ruzo et al., 1978). The trans-
isomer of deltamethrin was also rapidly metabolized and yielded
almost the same metabolites as deltamethrin, though 5-OH-derivative
was found in the cis-isomer, but not in the trans-isomer (Ruzo et
al., 1978).
When a single oral dose of 14C-(acid-, alcohol-, or cyano-
labelled) deltamethrin was administered to male mice at 1.7 - 4.4
mg/kg, the acid moiety and the aromatic portion of the alcohol
moiety were rapidly and almost completely excreted, whereas the CN
group was excreted relatively slowly (Ruzo et al., 1979).
Gray & Rickard (1982) followed the distribution of 14C-acid-,
14C-alcohol-, and 14C-cyano-labelled deltamethrin and selected
metabolites in the liver, blood, cerebrum, cerebellum, and spinal
cord after iv administration of a toxic, but non-lethal, dose
(1.75 mg/kg) to rats. Approximately 50% of the dose was cleared
from the blood within 0.7 - 0.8 min, after which the rate of
clearance decreased. 3-Phenoxybenzoic acid (PBacid) was isolated
from the blood in vivo, and was also the major metabolite when
14C-alcohol-labelled deltamethrin was incubated with blood in
vitro. Deltamethrin levels in the liver peaked at 7 - 10 nmol/g at
5 min and then decreased to 1 nmol/g by 30 min. In contrast, peak
central nervous system levels of deltamethrin were achieved within
1 min (0.5 nmol/g), decreasing to 0.2 nmol/g at 15 min, and
remaining stable until 60 min. Peak levels of deltamethrin were
not related to the severity of toxicity, though the levels of
unextractable pentane radiolabel did appear to be correlated with
signs of motor toxicity. Experiments with brain homogenates from
animals injected iv with deltamethrin failed to reproduce the
pentane-unextractable radioactivity in vitro and metabolism of the
compound was not demonstrated.
The major metabolic pathways of deltamethrin in mice were
similar to those in rats, though there were some differences. These
included the presence of more unchanged deltamethrin in mouse faeces
than in rat faeces. In mouse faeces, there were 4 monohydroxy ester
metabolites (2'-OH-, 4'-OH-, 5-OH-, and trans-OH- deltamethrin
(11)) and one dihydroxy metabolite (12) (4'-OH- trans-
OH-deltamethrin) that were not found in mouse urine. Major
metabolites from the acid moiety in mice were Br2CA,
trans-OH-Br2CA (20), and their glucuronide and sulfate
conjugates. Among them, trans-OH-Br2CA-sulfate was detected
only in mice, but not in rats. Compared with rats, much larger
amounts of trans-OH-Br2CA and its conjugates were formed in
mice. A major metabolite of the alcohol moiety in mice was the
taurine conjugate of PBacid in the urine, which was not detected in
rats. Generally, mice produced smaller amounts of phenolic
compounds compared with rats. Also, 3-phenoxybenzaldehyde (24)
(PBald), 3-phenoxybenzyl alcohol (32) (PBalc), and its glucuronide,
and glucuronides of 3-(4- hydroxyphenoxy)benzyl alcohol (33)
(4'-OH-PBalc) and 5-hydroxy-3- phenoxybenzoic acid (34)
(5-OH-PBacid) were found in mice, but not in rats. When mice were
given an ip dose of 14C-deltamethrin, with or without piperonyl
butoxide (PBO) and/or S,S,S-tributyl- phosphorotrithioate (DEF),
the same metabolites were obtained as with oral administration.
However, DEF decreased the hydrolytic products relative to the
controls, while PBO decreased the oxidation products (Ruzo et al.,
1979).
The comparison between the excreted radioactivity of
14C-deltamethrin in rats treated by the percutaneous route and iv
(controls) showed that only 3.6% of the dosage applied on the skin
was absorbed and excreted in 24 h with 1.1% excreted during the
first 6 h. Since the rat skin is more permeable than human skin,
the uptake of deltamethrin through the human skin should be
relatively weak (Pottier et al., 1982).
5.2 Metabolism and Fate in Farm Animals
In a metabolic study, 14C-deltamethrin was administered orally
to lactating dairy cows at the rate of 10 mg/kg body weight per day
for 3 consecutive days. It was poorly absorbed and mainly
eliminated in the faeces as unchanged deltamethrin. Only 4 - 6% of
the administered 14C was eliminated in the urine, and 0.42 - 1.62%
was secreted in the milk. The radiocarbon contents of various
tissues were generally very low with the exception of those of the
liver, kidney, and fat, which were higher (Akhtar et al. 1986).
Deltamethrin degradation occurred by cleavage of the ester bond, as
already reported in rats and mice (Ruzo et al. 1978, 1979). The
enzymes responsible for the ester bond cleavage were located in cow
liver homogenate, mainly in the microsomal fraction, as seen in an
in vitro study (Akhtar, 1984). Metabolites resulting from ester
bond cleavage were further metabolized and/or conjugated, resulting
in a large number of compounds excreted in the urine (see Fig. 3).
In milk, the major identifiable radiolabelled compound was
deltamethrin.
In a feeding study by Akhtar et al. (1987), deltamethrin was
administered twice daily to lactating dairy cows in portions of
their daily feed at the rate of 2 or 10 mg/kg diet for 28
consecutive days. The level of 2 mg/kg diet was the residue level
found in a recently treated pasture (Hill & Johnson, 1987), whereas
10 mg/kg diet was five times this level. Deltamethrin residues in
the milk were dose-dependent and appeared to reach a plateau
between 7 and 9 days after the start of treatment. At the high
deltamethrin intake of 10 mg/kg diet, the deltamethrin residue in
milk was about 0.025 mg/litre. Deltamethrin residues in tissues
were measured 1, 4, and 9 days after the last dose. At the
10 mg/kg diet intake, very small amounts of deltamethrin residues
were found in the liver (<0.005 mg/kg), kidney (<0.002 mg/kg),
and muscle (0.002 - 0.014 mg/kg). Residues in fat were about
0.04 mg/kg and 0.2 mg/kg for the 2 and 10 mg/kg intake,
respectively. Depletion of deltamethrin residues in milk was very
rapid (estimated half-life was about 1 day); while in fat (renal
and subcutaneous) the half-life was 7 - 9 days. Br2CA (3-(2,2-
dibromovinyl)-2,2-dimethylcyclopropanecarboxylic acid) and PBacid
(3-phenoxybenzoic acid) were the only metabolites detected in the
milk and tissues of treated cows. In all cases, they were found at
trace levels of < 0.0235 mg/litre and < 0.034 mg/litre,
respectively. These two metabolites were also previously
identified in rats and mice as the major degradation products of
deltamethrin (Ruzo et al., 1978, 1979).
The fate of 14C-deltamethrin was examined in Leghorn hens
(Akhtar et al., 1985). When laying hens were administered 7.5 mg
of 14C-labelled deltamethrin/hen per day orally for 3 consecutive
days, about 83% and 90% of the administered 14C was eliminated
during the first 24 h and 48 h after dosing, respectively. Tissue
residues were generally very low with the exception of those in the
liver and kidney. Very low levels of residues were found in eggs
obtained within the first 24 h after dosing, but levels increased
reaching a peak within 48 h of the last dose. Residue levels were
higher in the yolk (up to 0.6 mg/kg) than in the albumen (up to
0.2 mg/kg), which is probably related to the lipid content of
yolks. Metabolites were the same as those found in rats and mice.
These studies showed that feeding domestic animals on
deltamethrin-treated feed resulted in very low levels of residues
(if any) in products of animal origin and is unlikely to present a
hazard for the consumer.
5.3 Enzymatic Systems for Biotransformation
Deltamethrin (1 µg) was incubated at 37 °C for 30 min with each
of the following mouse microsome preparations; a) tetraethyl
pyrophosphate (TEPP)-treated microsomes (no esterase and oxidase
activity); b) normal microsomes (esterase activity); c) TEPP-
treated microsomes plus NADPH (oxidase activity); and d) normal
microsomes plus NADPH (esterase plus oxidase activity) (Shono et
al., 1979). Deltamethrin was more rapidly metabolized under the
oxidase system than under the esterase system. The major site of
ring hydroxylation was the 4'-position and the secondary site was
the 5-position. The trans methyl group was an important site of
hydroxylation of the esters and cis methyl oxidation was evident in
the metabolites of the cleaved acid moiety. The preferred sites of
hydroxylation were as follows; trans of dimethyl group,
4'-position in the phenol group, and cis of the dimethyl group,
which was equal to the 5-position in the phenoxy group. Cleavage
of deltamethrin to cyanohydrin may result from both esterase and
oxidase enzyme activities, since larger amounts of the cleaved
products were evident in the oxidase system.
However, at a much higher (approximately 35-fold) concentration
of deltamethrin than that in the above study, it was not detectably
hydrolysed (Miyamoto, 1976; Soderlund & Casida, 1977).
Deltamethrin was hydrolysed by esterases in the blood, brain,
kidney, and stomach of mice yielding PBald and PBacid (Ruzo et al.,
1979).
5.4 Metabolism in Human Beings
Three young male human volunteers underwent a complete medical
check-up one week prior to the morning of the study. Each of them
received a single dose of 3 mg of 14C-deltamethrin mixed in 1 g
glucose and diluted first in 10 ml PEG 300 and again in 150 ml
water. Total radioactivity was 1.8 ± 09 mBq. Samples of blood,
urine, saliva, and faeces were taken at intervals over 5 days.
Clinical and biological examinations were performed every 12 h
during the trial and one week after its termination. Radioactivity
in the biological samples was measured with a liquid scintillation
spectrometer. The clinical and biological checks did not detect
any abnormal findings. There were no signs of side effects or
intolerance reactions, either during or after the trial period.
The maximum plasma radioactivity appeared between 1 and 2 h after
administration of the product, and remained over the detection
limit (0.2 KBq/litre) during the 48 h. The apparent elimination
half-life was between 10.0 and 11.5 h. The radioactivity of blood
cells, as well as the saliva, was extremely low. Urinary excretion
was 51 - 59% of the initial radioactivity; 90% of this
radioactivity was excreted during the 24 h following absorption.
The apparent half-life of urinary excretion was 10.0 - 13.5 h,
which is consistent with the plasma data. Faecal elimination at
the end of the observation period represented 10 - 26% of the dose.
The total faecal plus urine elimination was around 64 - 77% of the
initial dose after 96 h (Papalexiou et al., 1984).
6. EFFECTS ON ORGANISMS IN THE ENVIRONMENT
6.1 Aquatic Organisms
6.1.1 Acute toxicity for fish
Acute toxicity data for deltamethrin in fish have been
summarized by L'Hotellier & Vincent (1986) (Table 6). From this,
it appears that deltamethrin is highly toxic for fish, though the
toxicity varies with the formulation tested.
Table 6. Acute toxicity of deltamethrin tested as the technical or formulated product on
fish; lethal concentrations all expressed as µg active ingredient (a.i.)/litre (96-h)
------------------------------------------------------------------------------------------
Species Systema LC50 (µg/litre) Ref. LC50 (µg/litre) Ref.
(Common name) tested as No. tested as No.
technical formulated
product productb
------------------------------------------------------------------------------------------
Alburnus alburnus S 0.69 4 82 (ULV) 4
(Bleak)
Brachydanio rerio F,S 2.0 10 -
(Zebra fish)
Cyprinodon macularius S - 0.6c (EC) 13
(Desert pupfish)
Cyprinodon S - 0.9 (EC) 19
variegatusd
(Sheepshead minnow)
Cyrpinus carpio F, S 1.84 4 0.65 (EC) 4
(Common carp) 0.86 3 210.0 (ULV)
Gambusia affinis F, S - 1.0c (EC) 13
(Mosquito fish)
Ictalurus nebulosus F, S 1.2 7 2.3 (EC) 15
(Brown bullhead)
Ictalurus punctatus F, S 0.63 8 -
(Hannel catfish)
Idus idus melanotus S - 1.2 (EC) 16
(Golden orfe)
Lebistes reticulatus F, S - 1.8 (EC) 17
(Guppy)
Lepomis gibbosus F, S 0.58 5 0.87 (EC) 14
(Pumpkinseed sunfish)
Lepomis machrochirus F 1.2 6 -
(Bluegill sunfish)
Osteochilus hasseltie S - 1.2 (EC) 20
(Nilem carp)
Puntius gonionotuse F,S - 0.87 (EC) 18
(Jawa carp)
Rhodeus sericeus S 1.12 4 140 (ULV) 4
amarus
Salmo gairdneri F, S 0.39 1 2.2 (EC) 12
(Rainbow trout)
Salmo salar 1.97 2 0.59 (EC) 2
------------------------------------------------------------------------------------------
Table 6. (contd.)
------------------------------------------------------------------------------------------
Species Systema LC50 (µg/litre) Ref. LC50 (µg/litre) Ref.
(Common name) tested as No. tested as No.
technical formulated
product productb
------------------------------------------------------------------------------------------
Salmo trutta F, S - 4.7c (EC) 11
(Brown trout)
Sarotherodon F, S 3.5 9 2.0 (EC) 9
mossambicuse
Tilapia mossambicae F, S - 0.8c (EC) 13
------------------------------------------------------------------------------------------
a F: Flow system, S: Static condition.
b EC: 25 g a.i./litre; ULV: 1 g a.i./litre; values in a.i. equivalent obtained by
calculation.
c LC50 (48-h)
d Marine fish.
e River or pond fish from tropical areas (water temperature > 24 °C).
References
(1) Knauf & Horlein (1979); (2) Zitko et al. (1979); (3) Knauf & Schulze (1977a);
(4) Gulyas & Csanyi (undated); (5) Waltersdorfer & Schulze (1976a); (6) Buccafusco et al.
(1977a); (7) Knauf & Schulze (1977b); (8) Buccafusco et al. (1977b); (9) Adeney et al.
(1980); (10) Lepailleur & Chambon (1984); (11) Lhoste et al. (1979); (12) Waltersdorfer &
Schulze (1976c); (13) Mulla et al. (1978); (14) Waltersdorfer & Schulze (1976d); (15) Knauf
& Schulze (1977b); (16) Waltersdorfer & Schulze (1976b); (17) Waltersdorfer & Schulze
(1976a); (18) Santosa & Hadi (1980); (19) Heitmuller et al. (1978); (20) Santosa (1983)
Zitko et al. (1979) established a 96-h lethal threshold for
Atlantic salmon (Salmo salar) of 1.97 µg/litre.
6.1.2 Acute toxicity for other aquatic organisms
Data on aquatic organisms other than fish are presented in
Table 7 and are of the same order as those for fish, although the
oyster (Crassostrea virginica) is somewhat more tolerant and the
Northern lobster (Homarus americanus) (96-h lethal threshold
0.0014 µg/litre) is far more sensitive (Zitko et al., 1979).
Mohsen & Mulla (1981) exposed aquatic insect larvae to
deltamethrin (as a 2.5% emulsifiable concentrate) for 1 h under
flow-through conditions, and calculated the LC50 after a 24-h
holding period. For the target species blackfly (Simulium
virgatum) an LC50 of 0.9 µg/litre was calculated. Non-target
species tested, mayfly (Baetis parvus) and caddisfly (Hydropsyche
californica), were found to be more susceptible, with LC50 values
of 0.4 µg/litre.
Varanka, (1987) investigated the effects of deltamethrin on
three species of freshwater mussels. Results presented in Table 8
show that the mussels are very insensitive to the pyrethroid.
Table 7. Acute toxicity of deltamethrin tested as technical or
formulated product on other aquatic organisms-lethal concentrations
expressed as µg active ingredient (a.i.)/litre (96-h)a
-------------------------------------------------------------------
Species LC50 (µg/litre) LC50 (µg/litre)
tested as technical tested as formulated
product productb
-------------------------------------------------------------------
Crassostrea virginica - 12.0
(Eastern oyster)
Daphnia magna 5c -
(Water flea)
Gammarus pulex - 0.03c
(Scud)
Penaeus duorarum - 0.35
(Pink shrimp)
Uca pugilatorulosus - 1.1
(Fiddler crab)
Bufo bufo (larvae) - 0.93
(Common toad)
-------------------------------------------------------------------
a Adapted from: L'Hotellier & Vincent (1986).
b EC: 25 g a.i./litre; ULV: 1 g a.i./litre; values in a.i.
equivalent obtained by calculation.
c LC50 (48-h).
Table 8. Acute toxicitya of deltamethrin formulationb in
freshwater mussels, under static conditions at 21 - 23 °Cc
-------------------------------------------------------------------
Species 24-h 48-h 72-h 96-h 7-day
-------------------------------------------------------------------
Anodonta cygnea nd nd ~24.6 12.0 7.6
Anodonta anatina nd nd nd ~23.4 10.3
Unio pictorum nd ~31.8 9.7 7.0 6.0
-------------------------------------------------------------------
a LC50 µg active ingredient (a.i.)/litre): values in a.i.
equivalent obtained by calculation.
b ULV 0.12%.
c From: Varanka (1987).
6.1.3 Field studies and community effects
Two experimental pond studies have been performed. Tooby et
al. (1981) reported that application of deltamethrin to static
water at 10 g a.i./ha did not have any lethal effects on two fish
species (Canassius auratus, Rutilus rutilus) or on molluscs.
Aquatic insects and crustaceans present were killed. Rawn et al.
(1985) applied deltamethrin at a similar rate and also reported
that no fish were killed. The half-life of deltamethrin in the
pond was 2 - 4 h for water and 2 - 14 days for bottom sediment.
Neto et al. (1983) sprayed-flooded fields in Brazil, at
intervals of 2 days, with rates of deltamethrin progressively
increased at 5, 10, 12, and 13 g a.i./ha. The expected
concentrations in water from these applications were between 3 and
7 µg/litre. No mortality was recorded in fish placed in the
sprayed area in experimental cages. Slight "agitation" was
reported after exposure to the highest dose.
Impact assessments on the use of deltamethrin on paddy fields
have been made in the field in various countries throughout the
world. The maximum normal usage rate of the compound was 6.5 g
a.i./ha. In these studies, fish ( Tilapia spp., Cyprinus carpio,
Gambusia spp.) tolerated deltamethrin up to 18.75 g a.i./ha without
any adverse effects. The compound is known to be toxic for aquatic
organisms and is not recommended for use over water under any but
exceptional circumstances. However, it has been used to control
vectors of major human diseases, i.e., mosquitos and blackfly
( Elossina spp.), where benefit outweighed potential risk. In these
cases, extensive field evaluations of the environmental impact have
been made. While there have not been any instances of fish kills
from these applications, there are reports of large numbers of
deaths of aquatic invertebrates. The populations usually recovered
rapidly and all studies have shown numbers back to normal before
the compound was applied again in the following season. It is
suggested that relatively resistant parts of the population soon
recolonize the area; immigration also occurs (Takken et al., 1978;
Smies et al., 1980; Baldry et al., 1981; Everts et al., 1983).
6.1.4 Appraisal
Notwithstanding its high toxicity for fish and crustacea, the
results of many studies, as well as the wide use of deltamethrin
for several years, have confirmed that its normal use does not
cause significant mortality in fish populations. This difference
is due to its strong adsorption on soil and its rapid breakdown,
decreasing its bioavailability under field conditions.
6.2 Terrestrial Organisms
6.2.1 Plants
Hargreaves & Cooper (1979) sprayed glasshouse-grown tomato
seedlings with 50 mg deltamethrin/litre (2.5% emulsifiable
concentrate) 3 weeks after emergence and again 7 days later. Three
days after the second application, plants were examined for damage.
No damage was found and, at this rate of use, deltamethrin was not
phytotoxic.
6.2.2 Soil microorganisms
In a study by Tu (1980) on the effects of 5 pyrethroids on
microbial populations and their activity in soil, 0.5 mg
deltamethrin/kg incorporated into sandy loams (residues under
normal use conditions would be of the order of < 0.001 mg/kg)
produced only a few transient effects. No effects were noted on
the nitrifying microorganisms and their capacity to produce nitrate
and there were no inhibitory effects on deshydrogenase or urease
activity. Deltamethrin induced an increase in oxygen consumption
because of an increase in microbial respiration (probably linked
with the microbial degradation of deltamethrin). It also
stimulated the growth of soil fungi and inhibited the development
of bacteria. Four weeks after treatment, deltamethrin-treated soil
recovered completely and microorganism activity was equal to that
in untreated soil.
6.2.3 Soil fauna
6.2.3.1 Earthworms
When deltamethrin at 12.5 g a.i./ha (high agricultural dose)
was incorporated into the soil to a depth of 1 cm, there were no
toxic effects on earthworms (Lumbricus terrestris) during an
observation period of 28 days (Bouche & Fayolle, 1979). However,
significant toxic effects on earthworms were observed at levels of
60 - 125 g a.i./ha (5 - 10 times the highest rates applied in
agriculture).
In another study with Eisenia foetida andrei, deltamethrin
incorporated in artificial soil at concentrations of 1.7 mg/kg and
10 mg/kg did not produce any lethal effects (Chambon & Lepailleur,
1984).
6.2.3.2 Slugs
Lettuce leaves treated with 4 times normal dosage rates, were
fed to slugs ( Agrolimax sp.). Leaves were quickly consumed but no
toxic effects (mortality or activity) were observed (Ricou, 1978).
6.2.3.3 Soil arthropods
Under laboratory conditions, deltamethrin, applied topically
and by immersion, was very toxic for the carabid beetle
Pterostichus melanarius (Illiger). Under natural conditions in the
field, deltamethrin applied at normal dose rates was not toxic for
these organisms (Dunning et al., 1981).
Everts et al. (1985) monitored the effects, on non-target
organisms, of various compounds when used for the control of tsetse
fly in the Ivory Coast in Africa. Deltamethrin was the most
effective compound against the tsetse and also killed non-target
musca flies. After deltamethrin spraying, Orthoptera and
Proctotrupoidea were also significantly decreased while Nematocera
increased in number. The results of this study suggest that ground
spraying of the pyrethroid had greater effects on terrestrial
arthropods than aerial applications.
Concurrent laboratory and field studies were conducted on the
effects of deltamethrin on beneficial predatory spiders in a polder
area of the Netherlands (Everts et al., 1988). During two growing
seasons, 2800 samples were taken over an area of 17 different
fields. The authors found that effects on spiders were eliminated
when it rained soon after application, since the effect of the
pyrethroid appeared to be indirect, causing the dehydration of
spiders. This different response under dry and damp conditions was
confirmed in the laboratory. However, reduction of spider numbers
in the field was much greater than predicted from laboratory tests
and recovery was more rapid in laboratory populations than in field
populations. The uptake and effects of deltamethrin were greater
through exposure to residues than through contact or oral exposure.
There was a positive correlation between temperature and the
toxicity of deltamethrin for spiders in the field. This contrasted
with reports of a negative correlation for target insects reported
in the literature. Laboratory studies showed that the negative
temperature effect only occurred when spiders could not drink. It
appeared that qualitative prediction from laboratory to field was
possible but that quantitative prediction was not.
6.2.4 Beneficial insects
6.2.4.1 Honey-bees
Single applications of deltamethrin are highly toxic for honey-
bees (Apis mellifera). Stevenson et al (1978) found a contact LD50
of 0.051 µg/bee and an oral LD50 of 0.079 µg/bee.
Arzone & Vidano (1978) did not find any difference in mortality
between controls and bees fed on sugar solutions containing 0.2 µg
deltamethrin/litre. Increased mortality was recorded at all higher
exposures reaching 100% within 1 h at a concentration of 12.5
µg/litre.
In the field, direct treatment of caged bees caused a high
mortality rate with doses of from 11.2 g/ha upwards (Atkins et al.,
1976). Rape flowers were treated at a rate of 0.75 g a.i./100
litre and 1.5 g a.i./100 litre with an emulsifiable concentrate
formulation, 25 g/litre; control plots were treated with water.
Cages (3 x 2 x 2 m) containing a small hive (2 frames + open brood)
were put over the treated flowers once the spray had dried. The
mortality of the bees was then assessed over 7 days. The average
mortalities were not significantly higher in the treated plots than
in water-sprayed control plots (Louveaux et al., 1977).
However, Bocquet et al. (1980, 1983) demonstrated, after 3
years of field experiments, that deltamethrin under field
conditions was innocuous at doses up to 12.5 g/ha. They also noted
a repellant effect by the formulating materials, which lasted for
2 - 3 h. Further studies have been reported by Florelli et al.
(1987a,b).
6.2.4.2 Foliar insects
Deltamethrin was 70 times more toxic to the tobacco budworm
(Heliothis virescens) than to its predator, green lacewing
(Chrysopa carnea), but it was only 1.25 times more toxic to the
tobacco budworm than to its parasite (Campoletis sonorensis) (Plapp
& Bull, 1978).
In an apple orchard, where deltamethrin was applied at
12.5 mg/kg, no predatory mites (Typhlodromus pyri) were found
during 10 weeks of observation, but spider mites (Paponychus ulmi)
were not affected. The elimination of the predatory mite led to a
marked increase in spider mite populations, later in the same
season (Aliniazee & Cranham, 1980).
The impact of deltamethrin used against the English grain aphid
(Sitobion avenae) was studied in 1983, 1984, and 1985 in the Paris
basin. This study was carried out on wheat with pitfall traps,
yellow water traps, suction sampling (D-vac), and sampling of ears.
Effects were noted on: S. avenae, phytophagous Diptera (Opimyza
florum, Phytomyza nigra, and Oscinella frit), Homoptera (Zyginidia
scutellaris, Metopolophium dirhodum), Thysanoptera (Limothrips
cerealium, Acolothrips intermedius), predatory Diptera (Empididae,
Dolichopodidae), and on spiders (Erigonidae, Lycosidae,
Linyphiidae, Theridiidae). The detritiphagous insects (Sciaridae,
Chironomidae), the Carabidae and Staphylinidae and most
microhymenoptera showed little or no difference after treatment.
During the 3 years, no differences were observed from year to year
as a result of field treatment, populations appearing homogeneous
at the beginning of each trial (Fischer & Chambon, 1987).
A large-scale field trial was carried out in 1984 in southern
England to investigate the side-effects of deltamethrin on non-
target arthropods in winter wheat. The insecticides were applied
in June and two methods, suction sampling (D-vac) and quadrats,
were used to sample the arthropods for up to 75 days after
treatment. During the post-treatment period, the numbers of
Carabidae and Staphylinidae adults found in D-vac samples were
reduced by 22% and 20%, respectively, compared with the controls
(Vickerman et al., 1987a).
In the same field trial, arthropods were sampled with a D-vac
for 11 weeks. Total numbers were similar in the control and
deltamethrin-treated plots. The numbers of Empididae were reduced
by deltamethrin, but Dolichopodidae were more numerous in treated
than in control plots. The numbers of Aphidius spp. were higher in
the deltamethrin-treated plots than in the control plots. The
numbers of Coccinellidae larvae were reduced (Vickerman et al.,
1987b).
6.2.5 Birds
6.2.5.1 Laboratory studies
Data on the acute toxicity of deltamethrin for birds are given
in Table 9.
Table 9. Acute toxicity of deltamethrin for birds
---------------------------------------------------------------------------
Species Sex Application LD50 (mg/kg) Reference
---------------------------------------------------------------------------
Red partridge male & oral >3000 Grolleau & Griban,
(Alectonis tufa) female 1976b
Grey partridge male & oral >1800 Grolleau & Griban,
(Perdix perdix) female 1976b
Chicken oral >1000 Grandadam, 1976
(Gallus domestica)
Hen adult oral >2500 Ross et al., 1978
female
Mallard duck oral >4640 Beavers & Fink,
(Anas platyrhynchos) 1977a
Game duck oral >4000 Grolleau & Griban,
1976a
---------------------------------------------------------------------------
The toxicity of deltamethrin for birds is very low. Both
technical grade and commercially formulated deltamethrin
administered in feed at 100 mg/kg diet was not palatable to
Japanese quail (Coturnix coturnix japonica), with strong individual
variations. Unpalatability diminished after repeated exposure and
even became reversed in the case of the purified deltamethrin,
which attracted quail already suffering from toxic effects (David,
1981).
Groups of 39 female Japanese quail (Coturnix coturnix japonica)
were given daily doses of 0, 0.2, or 1 mg technical deltamethrin
per animal, by gavage, over 34 days. No significant effects were
observed on reproduction (De Lavaur et al., 1985).
6.2.5.2 Field studies on birds
The low toxicity of deltamethrin for birds, indicated by
laboratory studies, has been confirmed in the field. In studies on
the ecological consequences of the use of the compound to control
tsetse fly (Takken et al., 1978) and blackfly (Smies et al., 1980)
in West Africa, populations of various species of insectivorous,
granivorous, and piscivorous birds were examined before and after
spraying. There were no indications of any effects on either
numbers or species diversity.
7. EFFECTS ON EXPERIMENTAL ANIMALS AND IN VITRO TEST SYSTEMS
7.1 Single Exposures
Tables 10 and 11 show the results of acute toxicity studies on
various animal species. From these tables, it is clear that the
vehicle has a great influence on the LD50, probably by influencing
absorption. Powder formulations and aqueous suspensions are
significantly less toxic than formulations in oils or organic
solvents (Pham Huu Chanh et al., 1984).
The acute oral toxicity of deltamethrin for rats produced such
symptoms as: staining of the fur, excessive grooming, salivation,
diarrhoea, drowsiness, weakness, dyspnoea, piloerection, ptosis,
difficulty in walking, general motor incoordination, hypotonia,
choreoathetosis, clonic seizures, and death (Glomot, 1979; Glomot
et al., 1979, 1981a; Kavlock et al., 1979; Ray & Cremer, 1979; Pham
Huu Chanh et al., 1984). Electroencephalogram (EEG) records showed
generalized spike discharges prior to choreoathetosis (Ray &
Cremer, 1979; Ray, 1980).
Mice presented far fewer symptoms than rats after oral dosing
at comparable levels, diarrhoea being the only reportable
observation (Glomot et al., 1980a).
Rats were injected intraperitoneally with 14C-labelled
deltamethrin at the threshold doses required to produce the motor
symptoms of toxicity of tremor and choreoathetosis. Blood and brain
samples were analysed for their total radiolabel content, and were
also extracted with ethyl acetate to determine the levels of
extractable parent deltamethrin and 3-phenoxybenzyl-derived acid and
the residual radiolabel after this extraction. There was a clear
correlation between onset of symptoms and blood and brain levels of
deltamethrin. It was found that certain threshold levels of parent
deltamethrin in the blood and brain were required for symptoms
development, and that the symptoms persisted for as long as this
threshold was maintained (Rickard & Brodie, 1985).
7.1.1 Mouse
Mice intravenously injected with deltamethrin showed intense
tremors, convulsions, and ataxia, immediately after administration.
Tachycardia and respiratory defects were also observed at higher
dosages. Surviving animals appeared normal after 4 - 5 h.
Immediately after intraperitoneal injection, jumping movements,
slight convulsions and prostration, ptosis, tail hypertonicity, and
cyanosis were observed. These toxic signs disappeared after 72 h
in surviving animals.
Animals administered deltamethrin by gavage showed muscular
stiffening and convulsions, 1 h after dosing. After 24 h,
hypermotility, stereotype movements of the head, tachycardia,
hypertonicity of the tail, and a few convulsions were observed.
Behaviour and appearance were normal again after 48 h (Glomot &
Chevalier, 1976a,c).
Table 10. Acute toxicity of technical grade deltamethrin
--------------------------------------------------------------------------------------------------------------
Species Sex Route Vehicle LD50 (mg/kg Reference
body weight)
--------------------------------------------------------------------------------------------------------------
Rat male oral sesame oil 128 Glomot & Chevalier (1976a)
female 139
male PEG 200 67
female 86
Rat male adult peanut oil 52 Kavlock et al. (1979)
female adult 31
female weanling 50
Rat male adult peanut oil 53 Gaines & Linder (1986)
female adult 30
female weanling 48
Rat male + female aqueous suspension > 5000 Audegond et al. (1981)
with carboxy- (no mortality)
methylcellulose
Rat dermal - 700 Panshina & Sasinovich (1983)
Rat male methylcellulose (1%) > 2940 Kynoch et al. (1979)
female
Rat female adult xylene > 800 Kavlock et al. (1979)
Rat male + female inhalation (6 h) dust 600 mg/m3 Coombs & Clark (1978)
Rat male adult (2 h) DMSO 10% aerosol 940 mg/m3 Kavlock et al. (1979)
female adult > 785 mg/m3
Rat male + female (1 h) micronized powder > 4620 mg/m3 Jackson & Hardy (1986)
Rat intraperitoneal - 58.8 Panshina & Sasinovich (1983)
Rat male intraperitoneal sesame oil 209 Glomot & Chevalier (1976b)
female 186
male PEG 200 24 Glomot & Chevalier (1976b)
female 25
--------------------------------------------------------------------------------------------------------------
Table 10. (contd.)
--------------------------------------------------------------------------------------------------------------
Species Sex Route Vehicle LD50 (mg/kg Reference
body weight)
--------------------------------------------------------------------------------------------------------------
Rat male intravenous PEG 200 3.3 Glomot & Chevalier, (1976c)
female 3.3
Rat female adult acetone 4 Kavlock et al. (1979)
female weanling 1.8
Mouse male oral sesame oil 33 Glomot & Chevalier (1976a)
female 34
male PEG 200 21 Glomot & Chevalier (1976a)
female 19
Mouse intraperitoneal - 33 Panshina & Sasinovich (1983)
Mouse male intraperitoneal sesame oil 171 Glomot & Chevalier (1976b)
female 166
Mouse male PEG 200 18 Glomot & Chevalier (1976b)
female 12
Mouse male PEG 200 4.1 Glomot & Chevalier (1976c)
female 4.0
Mouse male glycerol formal 5 Glomot & Chevalier (1976c)
female 5.8
Dog male + female oral in capsules >300 Glomot et al. (1977)
no mortality
Dog male + female PEG 200 2 Glomot & Chevalier (1976c)
Rabbit male dermal PEG 400 > 2000 Clair (1977)
female > 2000
--------------------------------------------------------------------------------------------------------------
Table 11. Acute toxicity of some formulations
--------------------------------------------------------------------------------------------------------------
Species Sex Route Formulation LD50 (mg/kg Reference
body weight)
--------------------------------------------------------------------------------------------------------------
Rat male, female oral 2.5% flowable formulation 22 000 Glomot et al. (1979)
Rat male, female oral 2.5% wettable powder >15 000 Glomot (1979)
Mouse male, female oral 2.5% wettable powder >15 000 Glomot et al. (1980a)
Dog male, female oral 2.5% wettable powder >10 000 Glomot et al. (1980b)
Rat male, female oral 2.5% emulsifiable concentrate 535 Coquet (1977)
Rat male, female oral 10 g/litre ULV >6 470 Coquet (1977)
Rat male, female inhalation (4 h) aerosol-2.5% wettable powder >2 800 mg/m3 Clark et al. (1980)
--------------------------------------------------------------------------------------------------------------
7.1.2 Rats
Rats intravenously injected with deltamethrin showed muscular
contractions, piloerection, respiratory defects, convulsions, and
paresis of the hind quarters, immediately following treatment.
Surviving animals showed normal behaviour after 48 h. Immediately
after intraperitoneal injection, tremor, convulsions, prostration,
and cyanosis were observed. These toxic signs disappeared after
48 h in surviving animals. Animals administered deltamethrin by
gavage showed motor incoordination, convulsions, respiratory
defects, and hypomotility, shortly after dosing. Normal behaviour
was observed after 3 days (Glomot & Chevalier, 1976c).
In an inhalation study (whole body exposure for 6 h),
hyperactivity, grooming, and irritation were observed during
exposure. The animals were hypersensitive to touch and noise and
showed uncoordinated movements. Gross pathological investigation
showed a gas-filled stomach and small intestine, and massive
haemorrhage and degeneration in the lung (Coombs & Clark, 1978).
Rats were exposed (whole body exposure) for 4 h to an aerosol
concentration of deltamethrin equal to 2.8 g/m3, the highest
attainable airborne concentration of a 2.5% wettable powder
formulation. Approximately 80% of the total aerosol had a mean
aerodynamic diameter of less than 5.5 µm. Dyspnoea and gasping
were observed in exposed rats. Relative lung weights and
macroscopic pathology were normal. There was no mortality (Clark
et al., 1980).
7.1.3 Rabbit
Rabbits (10 males and 10 females) were treated with 2 g
deltamethrin in 2 ml PEG 400 per kg body weight on 80 cm2 of
occluded shaved skin for 24 h. The animals were observed for 14
days. Two animals showed obvious erythema. No body weight changes
or abnormal behaviour were observed. On histological observation
of the liver, kidneys, and skin, small changes were observed, but
these were common for this strain of rabbit and not related to
treatment (Clair, 1977).
7.1.4 Dog
Dogs given oral doses of 100 mg deltamethrin/kg body weight or
more showed transient hyperexcitability, akinesia, vomiting, and
stiffness of the hind legs (Glomot et al., 1977).
Dogs orally dosed with 10.0 mg deltamethrin/kg body weight did
not display any clinical signs related to treatment (Glomot et al.,
1980b).
7.2 Irritation and Sensitization
7.2.1 Skin irritation
Male albino rabbits (12 per group) weighing 2.5 - 3.5 kg were
administered 0.5 g deltamethrin on either shaved intact or abraded
skin. The occlusive patch was fixed on the skin for 23 h.
Technical deltamethrin (98% purity) did not produce any irritant
effects (Coquet, 1976a).
Male albino rabbits (6) weighing 2.5 - 2.9 kg were administered
0.5 ml of formulated deltamethrin (25 g/litre flowable suspension
concentrate) to both shaved intact and abraded skin. The Primary
Irritation Index after 24 h exposure of occluded sites was 1.2,
i.e., slightly irritating (Glomot et al., 1981b).
An evaluation similar to the one described above was carried out
for a 2.5% wettable powder concentrate deltamethrin formulation.
Rabbits had a Primary Irritation Index of 2.41, i.e., moderately
irritating. Moderate erythema continued for 72 h, while the oedema
generally diminished, with the exception of scarified skin sites
(Glomot et al., 1981c).
The skin irritation potentials of Decis emulsifiable
concentrate 2.5% and Decis Flowable 2.5% were studied on rabbits
and guinea-pigs with 0.05, 0.10, 0.5, 1, and 2.5% deltamethrin.
The threshold irritative levels were 0.05% for Decis emulsifiable
concentrate and 2.5% for Decis Flowable. The intensity of
irritation depended on the relative content of organic solvents and
emulsifiers in the trade products. The water-soluble concentrate
of Decis 2.5% caused negligible risk of contact irritative
dermatitis (Bainova & Kaloyanova, 1985).
7.2.2 Eye irritation
Deltamethrin (0.1 g/animal) was administered into the
conjunctival sac of the eyes of 6 male albino rabbits, weighing
2.5 kg, with or without rinsing 60 seconds after instillation.
Deltamethrin produced transient irritating effects, both with and
without rinsing (Coquet, 1976b).
Male albino rats (9) weighing between 2 and 3 kg were
administered 0.1 ml of formulated deltamethrin (25 g/litre flowable
suspension concentrate) in the conjunctival sac. Six of the
treated eyes remained unwashed, while the remaining three were
rinsed with lukewarm water 20 - 30 seconds after instillation.
There was only transient clouding of the cornea in 2 animals 1 h
after dosing (1 washed, 1 unwashed), which cleared by day 2. Low
grade conjunctival irritation was noted among all animals
initially, which disappeared following day 2 of observations
(Glomot et al., 1981d).
A 2.5% deltamethrin formulation diluted 1/10 in distilled water
(0.1 ml per rabbit) elicited a similar pattern of initial transient
corneal clouding in 3 out of 9 rabbits examined, which cleared by
day 4. The undiluted formulation (100 mg) administered in the
conjunctival sac of rabbits produced increased involvement of the
conjunctiva, iris, and cornea in all animals, generally moderate in
severity, with low grade corneal opacity persisting in 2 rabbits
until day 7 (1 washed, 1 unwashed) (Glomot et al., 1981e).
7.2.3 Sensitization
Deltamethrin (0.5 g/animal) was applied topically to the skin
of albino guinea-pigs (10 male and female) 3 times per week, with a
2-day interval for 3 weeks, and once at the start of the fourth
week. The preparation was covered with an occlusive patch for
48 h. On days 1 and 10, the guinea-pigs received an intradermal
injection of 0.1 ml of Freund's adjuvant. The animals were
challenged 12 days after the last application with 0.5 g
deltamethrin. No sensitization was found (Guillot & Guilaine,
1977).
7.3 Short-Term Exposure
7.3.1 Rat
Male and female weanling Sprague-Dawley rats (20 of each sex
per group) were dosed (by gavage) with 0, 0.1, 1, 2.5, or 10 mg
deltamethrin in PEG 200/kg body weight per day for 13 weeks. No
treatment-related effects were observed on food and water
consumption, mortality, urinalysis, and haematology. Neurological
examinations and ophthalmoscopy did not reveal any abnormalities.
At the highest dose level, a slight hyperexcitibility was observed
among some rats in week 6. Lower body weight gain was noted in
males at 2.5 and 10 mg/kg. No clear treatment-related effects were
noted in the results of laboratory investigations or on the weights
of the organs. Gross and microscopic examination of a variety of
tissues and organs did not show any treatment-related findings.
Following the 13-week dosage period, 5 males and 5 females per
group were allowed to recover for 4 weeks. No evidence of
hyperexcitability was observed among the rats; body weight gain was
slightly higher in the treated groups than in the controls. The
no-observed-effect level was 1 mg/kg body weight (Hunter et al.,
1977).
Four groups of CD rats (8 of each sex per group) were exposed to
aerosolized deltamethrin (technical grade powder) for 6 h per day, 5
days a week, for 2 weeks, and for 4 days during a third week. Mean
aerosol concentrations were 3, 9.6, and 56.3 mg a.i./m3 with
about 87% of respirable particles (diameter lower than 5.5 µm). No
rats died as a result of exposure. Signs of irritation (agitated
grooming and ptyalism due to the powder were noted in all groups
during exposure, with more pronounced toxic signs (ataxia and
walking with arched backs) in the group receiving the highest dose
tested. Male rats also showed a reduced body weight (-5%) in all
groups. An elevation of the serum sodium ion content was noted at
the two highest doses. No increased incidence of any particular
lesion was observed in the high-dose group compared with the control
group. Irritation and weight loss were only slight at 3 mg/m3 and
this can be considered as a no-effect level (Coombs et al., 1978).
7.3.2 Dog
Male and female beagle dogs (3 - 5/sex per group), 25 weeks of
age, received a daily oral dose of 0, 0.1, 1, 2.5, or 10 mg
deltamethrin/kg body weight in PEG 200 in gelatin capsules over 13
weeks. All treated groups showed reduced body weight gain, but
this was not dose-related. Liquid faeces were associated with all
groups of treated dogs throughout the dosing period. Dilatation of
the pupils was seen in dogs receiving 2.5 and 10 mg/kg per day.
The sign was first seen 4 - 7 h after dosing and persisted
throughout the day. The incidence of vomiting increased dose-
dependently in all treated groups, except the group receiving
0.1 mg/kg. In the highest dose group, unsteadiness, body tremors,
and jerking movements were seen, particularly in males, in weeks 2,
3, and 4. Excessive salivation was seen initially and diminished
during the dosing period. After 5 and 12 weeks, depression of the
gag reflex was noted in a proportion of animals in all treated
groups. However, this was not considered to be of toxicological
significance. Exaggeration or depression of the patellar reflex
was observed in some animals in all treated groups after 5 and 12
weeks, mainly at 1, 2.5, or 10 mg/kg per day. Some animals in all
treated groups showed depression of the flexor reflex. Dose levels
of 2.5 and 10 mg deltamethrin/kg per day caused modification of the
EEG pattern in some animals, 12 weeks following administration.
Histopathological evaluations of tissues and organs, including the
nervous system and muscle tissue, did not reveal any abnormalities
that could be related to the administration of the compound.
During recovery, the gag reflex continued to be depressed, whereas
exaggeration of the patellar reflex was still seen in some dogs
that had previously received 1 mg/kg per day (Chesterman et al.,
1977).
7.4 Long-Term Exposure and Carcinogenicity
7.4.1 Mouse and rat
Male and female Charles River CD-1 mice (80 of each sex per
group) were fed dietary levels of deltamethrin of 0, 1, 5, 25, or
100 mg/kg, daily, for 24 months. There were no clear effects
related to the administration of deltamethrin on general behaviour,
mortality, body weight, and food consumption. Blood chemistry,
haematology, and urine analysis parameters were normal after 12, 18,
and 24 months (at the times of interim and terminal sacrifice).
Microscopic examination of tissues did not reveal any lesions
indicative of a compound-related effect. The tumour incidence was
unaffected by deltamethrin administration. The no-observed-effect
level was 100 mg/kg diet (Goldenthal et al., 1980a).
Deltamethrin was administered, by gavage, to C57BL/6 mice at 4
dose levels (0, 1, 4, and 8 mg/kg body weight) and to BDVI rats at
3 dose levels (0, 3, and 6 mg/kg body weight) on 5 days a week for
104 weeks. After completion of the treatment, the animals were
observed until 120 weeks of age, when all survivors were killed.
The treatment had a slight effect on body growth and survival
rates, especially in the groups of mice and rats treated with the
highest dose. In C57BL/6 mice, various types of tumours were
observed in all treated groups. An increased incidence of
lymphomas was observed in mice receiving deltamethrin at levels of
1 and 4 mg/kg body weight, but not in the group treated with
8 mg/kg body weight. No significant difference in the incidences
of lung adenomas, liver-cell tumours, or other tumours was observed
in treated groups compared with controls. In BDVI rats, an
increased incidence of pituitary, thyroid, and mammary tumours was
noted; however, no clear dose-response relationship was shown
(Cabral et al., 1986). The details of this study were not
available for evaluation.
Male and female Charles River CD rats (90 of each sex per
group) were fed with 0, 2, 20, or 50 mg deltamethrin/kg diet for
2 years. A second control group (60 of each sex) was also used.
Interim sacrifices (10 of each sex per group excluding control
group 2) were made after 6, 12, and 18 months. No changes in
general behaviour and appearance were observed in relation to
treatment. Survival rate was similar for control and treated rats
(50 - 67%). Rats in the 50 mg/kg group gained slightly less weight
than control rats, whereas the food consumption was essentially the
same. Ophthalmoscopic findings were generally similar for control
and treated rats. No haematological and biochemical parameters
were changed in a biologically significant way in relation to
treatment at any time, except for a decrease in SGPT (serum
glutamic pyruvic transaminase) activity at 6 months in the mid- and
high-dose groups. Organ weights were not affected. The
macroscopic and microscopic findings were common for the species
and the strain, except for a slightly increased incidence of axonal
degeneration in sciatic, tibial, and/or plantar nerves in the 20
and 50 mg/kg groups at 18 months, but not at termination. Thus,
this was not considered to be indicative of a compound-related
effect. The incidence of benign testicular tumours (interstitial
cell adenomas) at terminal sacrifice in this study was: control
group 1, 0/37; control group 2, 4/35; low-dose group 1/38; mid-dose
group 1/30; high-dose group 6/38. The incidence seen in the high-
dose group was considered to be spontaneous, because it was not
significantly higher than in the second control group or in
historical control groups (Goldenthal et al., 1980b; Richter &
Goldenthal, 1983).
7.4.2 Dog
Deltamethrin dissolved in maize oil was administered in the diet
to 64 beagle dogs (8 of each sex per group) at levels of 0, 1, 10,
and 40 mg/kg for 24 months. This corresponds to 0, 0.025, 0.25, and
1 mg/kg body weight, respectively. Individual body weights and food
consumption values were determined weekly. Ophthalmoscopic,
haematological, biochemical, and urinalysis examinations were
conducted during the pre-test period and at 6, 12, 18, and 24 months
of the study. Neurological examinations were conducted at
approximately 1 year and before termination. No signs of overt
toxicity were observed in any of the dogs. Body weight and food
consumption values were similar for control and treated dogs. No
compound-related effects were observed during the ophthalmoscopic
and physical examinations. Although there were some random
statistically significant differences between the control and
other dose groups in the haematological and biochemical tests,
physiologically significant changes were not observed at any
interval in the study. Two treated and two control animals died
during the study. No compound-related gross or microscopic changes
were observed in the surviving dogs that were sacrificed and
necropsied. Inflammatory, degenerative, and proliferative changes
described were spontaneous in nature, or related to the estrous
phase of the menstrual cycle, and unrelated to compound
administration. On the basis of this study, it has been concluded
that the no-observed-effect level is 40 mg/kg diet (equivalent to
1 mg/kg body weight per day) (IRDC, 1980).
7.5 Mutagenicity
7.5.1 Microorganisms
DNA repair tests in Escherichia coli were conducted at dose
levels of 1250, 2500, or 5000 µg deltamethrin/ml. Deltamethrin was
dissolved in dimethyl sulfoxide (DMSO) and 0.1 ml of the solution
was spread on a plate. Growth inhibition was compared between DNA
repair deficient mutants (p3478 and CM611) and wild types (W3110
and WP2). Partial precipitation of deltamethrin from the solution
occurred when it came into contact with the aqueous bacterial
growth medium. Deltamethrin did not have any damaging effects on
DNA (Peyre et al., 1980).
Deltamethrin was examined for its mutagenic potential in the
Ames test with 5 strains of Salmonella typhimurium (TA 1535, TA
1537, TA 1538, TA 98, and TA100) at doses of 2, 10, 50, 200, 500,
1000, or 5000 µg/plate, with and without S-9 mix (metabolic enzyme
system). It was dissolved in DMSO and precipitated out of solution
at concentrations of 200 µg/plate or more. Deltamethrin did not
have any effect on the mutation rate in any of the strains at any
of the concentrations tested (Peyre et al., 1980).
A similar Ames test was carried out at 0.2, 2, 20, 200, or
400 µg deltamethrin/plate with microsome enzymes. The compounds
did not influence the number of revertants of the 5 strains (same
as above) of S. typhimurium. Again, deltamethrin was dissolved in
DMSO and precipitated out of solution at 200 µg/plate or more
(Fouillet, 1976).
Kavlock et al. (1979) found deltamethrin not to be mutagenic in
2 assays with S. typhimurium at doses of 0 - 1000 µg/plate in DMSO,
with or without metabolic activation. They also obtained negative
results with E. coli at 10 - 1000 µg/plate as well as with
Saccharomyces cerevisiae at concentrations of 1 - 5%, in both cases
with and without metabolic activation. Deltamethrin was found not
to be mutagenic in S. typhimurium strains TA100 and TA98, in the
presence or absence of a rat liver activation system, using the
plate incorporation assay and fluctuation tests. The compound,
dissolved in DMSO, precipitated out of solution at 600 µg/plate
(Pluijmen et al., 1984).
7.5.2 Cultured cells
Deltamethrin, dissolved in a mixture of cremaphor oil and
ethanol (1:1), was applied to a culture of Chinese hamster ovary
cells (CHO) at levels of 0.04, 0.2, 1.0, or 5.0 mmol/litre, with or
without metabolic activation, and examined for chromosomal
aberrations and sister chromatid exchanges (SCE). Because of the
cytotoxic effect of cremaphor oil when combined with S-9 mix or
deltamethrin, no cells would grow in the control dish with
activation, or in the 5 mmol/litre deltamethrin dishes, either with
or without metabolic activation. A high incidence of chromosomal
aberrations and SCEs was observed in the dishes containing 1 mmol
deltamethrin/litre, with activation. However, the absence of
control values (both with and without activation) because of a
broken test tube, made the interpretation equivocal. A second
study was conducted in which deltamethrin was dissolved in DMSO and
applied to the cells at levels of 0.001, 0.01, 0.1, or 0.2
mmol/litre, with or without metabolic activation. In this study,
deltamethrin did not produce any cytotoxic effects and did not
induce either chromosomal aberrations or SCEs in CHO cells.
However, no positive controls were tested and only single plates
were prepared per dose level (Sobels et al., 1978).
Deltamethrin was found not to be mutagenic in V79 Chinese
hamster cells, in the presence or absence of hepatocytes. It is
not known which solvent was used (Pluijmen et al., 1984).
7.5.3 Mouse
An in vivo cytogenetic test was conducted on mice (3 males and
3 females per group). Mice were treated orally with deltamethrin
in sesame oil for 2 consecutive days at 5 or 10 mg/kg body weight.
The incidence of chromosomal aberrations in bone marrow cells or
micronuclei in the polychromatic erythrocytes of treated groups
was, however, comparable to that of the control groups. No
positive controls were tested (Sobels et al., 1978).
Deltamethrin was applied orally, once, at 15 mg/kg body weight
to Swiss mice. A time-related effect on the chromosomes in bone
marrow cells was observed by killing 2 animals every 3 h during
24 h. The report stated that the incidences of chromated
aberrations were low and that there were no consistent time-related
trends in the distribution of the aberrations. However, the time-
related trend of aberrations was not reported. Again, no positive
controls were tested (Sobels et al., 1978).
A dominant-lethal assay with deltamethrin was performed.
Groups of 9 - 13 male mice were dosed orally at 3 mg/kg body weight
in sesame oil for 7 days or at a single dose of 6 or 15 mg/kg body
weight in sesame oil, and mated with 6 - 18 non-treated females.
There were no effects on the rates of pre- and post-implantation
losses, while the positive control, triethylene triphosphoramide
(10 mg/kg body weight), reduced pregnancies in the second and third
weeks after treatment and increased embryonal losses (Vannier &
Glomot, 1977).
Deltamethrin in olive oil was administered orally to female
Swiss mice at single or repeated (5 times at daily intervals) doses
of 1.36, 3.4, or 6.8 mg/kg per day. Bone marrow smears were
prepared 6, 24, or 48 h after treatment. No mutagenic activity was
observed with deltamethrin, whereas the positive control,
cyclophosphamide, induced a positive response (Polàkovà & Vargovà,
1983).
In a micronucleus test, a single dose of deltamethrin in corn
oil was administered orally at 16 mg/kg body weight to Swiss CD-1
mice (5 of each sex per group). No mutagenic activity was observed
with deltamethrin, whereas the positive controls,
triethylenemelamine and dimethylbenzanthracene, both induced
positive responses (Vannier & Fournex, 1983).
7.5.4 Appraisal
Deltamethrin is not mutagenic or clastogenic in a variety of in
vitro and in vivo test systems.
7.6 Teratological and Reproductive Effects
7.6.1 Teratology
7.6.1.1 Mouse
Deltamethrin was dissolved in corn oil and administered by
gastric intubation at doses of 0, 3.0, 6.0, or 12.0 mg/kg body
weight on days 7 - 16 of gestation to groups of CD-1 mice. Mice
were sacrificed on day 18 of gestation. There was a dose-related
( P <0.001) reduction in maternal weight gain during pregnancy and
high-dose females gained 58% less weight than the controls. There
was no dose-related mortality but dams in the high- and mid-dose
groups became convulsive after dosing. Treatment did not affect
the number of implantation sites, fetal mortality, fetal weights,
or the number of sternal and caudal ossification centres.
A significant ( P <0.01) dose-related increase in the
occurrence of supernumerary ribs was observed. No other dose-
related skeletal or visceral anomalies were observed (Kavlock et
al., 1979).
Pregnant female Swiss CD-1 SPF mice (24 per group) were given
deltamethrin dissolved in sesame oil by oral intubation at dose-
levels of 0, 0.1, 1, or 10 mg/kg body weight per day on days
6 - 17 of pregnancy. The animals were necropsied on day 18 of
pregnancy. The numbers of implantation sites, fetal losses, and
viable fetuses were not affected by treatment. There was a dose-
related decrease in mean fetal weight. Apart from delayed
ossification at all dose levels, skeletal examination revealed no
abnormalities. A teratogenic effect was not observed (Glomot &
Vannier, 1977).
In a complementary teratology study, pregnant female Swiss CD-1
mice were given deltamethrin dissolved in sesame oil by oral
intubation at 0, 0.1, 1, or 10 mg/kg body weight per day from day 6
to day 17 of gestation. Females were either sacrificed on day 18
of gestation or allowed to litter for subsequent examination of
pups on days 1 or 21 of lactation. The compound caused a moderate
and transient retardation of development of the fetus at the 1 and
10 mg/kg body weight dose rate, but these effects were not observed
on days 1 or 21 post-partum. There were no teratogenic effects
related to treatment (Vannier & Glomot, 1982).
7.6.1.2 Rat
Pregnant female Sprague-Dawley rats (24 per group) received 0,
0.1, 1, or 10 mg deltamethrin/kg body weight per day by oral
intubation on days 6 - 18 of pregnancy. Apart from 12 females in
the control and 10 mg/kg groups, which were allowed to deliver, the
dams were sacrificed and examined on day 21. There were no effects
on reproduction or on the teratogenic parameters examined, except
for slightly delayed ossification at the highest dose level (Glomot
& Vannier, 1977).
Deltamethrin was dissolved in corn oil and administered by
gastric intubation at doses of 0, 1.25, 2.5, or 5.0 mg/kg body
weight on days 7 - 20 of gestation. Rats were sacrificed on day 21
of gestation. There was a dose-related reduction ( P <0.01) in
maternal weight gain during pregnancy, and dams in the high-dose
group gained only 80% of the control value. Treatment did not
affect the number of implantation sites, fetal mortality, fetal
weight, or the number of sternal and caudal ossification centres
(Kavlock et al., 1979).
7.6.1.3 Rabbit
Groups of 15 pregnant New Zealand White rabbits received
deltamethrin dissolved in sesame oil at levels of 0, 1, 4, or
16 mg/kg body weight per day during days 6 - 19 of pregnancy.
Examination was carried out on day 28 of gestation. The mean fetal
loss was not dose-related. The mean fetal weight in the highest-
dose group was decreased. Some malformations (hydrocephaly,
exencephaly, and thoracogastroschisis) were observed in 2 fetuses
of animals at the highest dose level. In a supplementary study,
pregnant rabbits were similarly dosed with 16 mg/kg body weight per
day; one fetus with spina bifida and shortened tail was detected
among 69 apparently normal fetuses. Malformations were within the
normal limits of the strain used and were not considered to be
related to the treatment, despite the occurrence at the highest
dose level only (Glomot & Vannier, 1977, 1978).
7.6.2 Reproduction studies
Groups of 10 male and 20 female Charles River rats were fed
deltamethrin in the diet at 0, 2, 20, or 50 mg/kg and mated to
begin a 3-generation, 2-litter (first generation, 3 litter)
standard reproduction study. Parental body weights and food
consumption were recorded during the study. After weaning of the
second litter, the surviving parent rats were sacrificed and
necropsied. Five male and 5 female pups of the F3b generation were
necropsied. No changes relevant to treatment were observed in
general behaviour or survival of parent rats or pups. The body
weight of F0 males of the 50 mg/kg group was decreased from week 11
onwards. There were some slight decreases in mean food consumption
of F1 male parent rats in the 50 mg/kg group. The basic
reproduction indices (fertility, gestation, lactation, viability,
and litter size) were not affected by the treatment. However, the
mean pup weight in some litters, especially in the 50 mg/kg group,
was slightly decreased in comparison to the controls on day 21 of
lactation. Gross external examination did not reveal any
abnormalities. No gross or microscopic lesions of treatment-
related significance or significant effects on the organ weights of
the F3b generation were observed (Wrenn et al., 1980).
Deltamethrin was dissolved in corn oil and administered by
gastric intubation at doses of 0, 2.5, or 5.0 mg/kg body weight to
Sprague-Dawley rats from day 7 of gestation to day 15 of lactation.
The dams were allowed to litter and rear their young: litters were
reduced at birth to 4 males and 4 females per litter. The pups
were weighed weekly and examined for the development of eye-
opening, startle reflex, and air-righting. The litters were weaned
on day 22 post-partum and the males discarded. Weekly weighing of
the females continued and at 6 weeks of age they were tested in a
circular open-field. There were no effects on parturition, litter
size, or pup viability. Weights at birth were similar for all
groups, but a dose-related depression in growth was observed during
the pre-weaning period. This early diminution in pre-weaning
weight appeared to have little effect on the morphological and
behavioural parameters measured (Kavlock et al., 1979).
7.7 Neurotoxicity and Behavioural Effects
Adult hens (10 per group) were gavaged with a single dose of 0,
500, 1250, or 5000 mg deltamethrin/kg body weight suspended in corn
oil or 0 or 100 mg/kg body weight dissolved in sesame oil. During
21 days, observations were made on mortality, health, neurotoxic
signs, and body weight. Deltamethrin did not induce any clinical,
macroscopic, or histological signs of delayed neurotoxicity (Ross
et al., 1978).
Groups of 5 male and 5 female Wistar rats were administered
25 mg deltamethrin/kg body weight in 10 mg corn oil/kg on 2
consecutive days. Controls received 10 mg corn oil/kg body weight.
A tilting plane test was performed every second day from day 4 to
day 16 of the study. Two male rats died at 25 mg/kg. No effect
was found on the slip-angle (Davies et al., 1983).
The effects of deltamethrin were studied in a rat performance
test that arranged for milk delivery after every fortieth lever
press. Deltamethrin (1 - 8 mg/kg body weight, given orally, 2 h
before the test) produced both dose-related increases in pause
duration and decreases in response rate. Deltamethrin was also
studied using a conditional flavour-aversion test. Deltamethrin-
treated, trained rats displayed an aversion to saccharin that was
greatest at 2 mg/kg (Macphail, 1981).
The neurological effects of the 4 synthetic pyrethroids,
resmethrin, permethrin, cypermethrin, and deltamethrin, have been
investigated in the rat to establish whether there is a correlation
between the clinical-functional status of the animal and peripheral
nerve damage, as measured biochemically (Rose & Dewar, 1983).
Neuromuscular dysfunction was assessed by means of the inclined
plane test and peripheral nerve damage by reference to
beta-glucuronidase and beta-galactosidase activity increases in
nerve tissue homogenates from treated and control animals. A
transient functional impairment was found in animals treated with
any one of the 4 pyrethroids tested and in all cases this was
greatest at the end of the 7-day dosing regimen (deltamethrin doses
of 5 - 20 mg/kg per day in arachis oil). Significant increases in
beta-glucuronidase and beta-galactosidase activities were found
3 - 4 weeks after the start of dosing, in the distal portion of the
sciatic/posterior tibial nerves from permethrin-, cypermethrin-,
and deltamethrin-treated animals, but no changes were found in
resmethrin-treated animals. It is concluded, therefore, that there
is no direct correlation between the time-course of the
neuromuscular dysfunction and the neurobiochemical changes. This
suggests that these pyrethroids have at least two distinct actions-
a short-term pharmacological effect at near-lethal dose levels and
a more long-term neurotoxic effect that results in sparse axonal
nerve damage.
To better characterize the behavioural toxicity of pyrethroid
insecticides, comparisons were made of the effects of cismethrin
and deltamethrin exposure on motor activity and the acoustic
startle response in male Long-Evans rats (Crofton & Reiter, 1984).
Acute dose-effect, acute time-course, and 30-day repeated-exposure
determinations of 1-h motor activity were made using figure-eight
mazes. The acoustic startle response was measured to a 13-kHz,
120-dB(A), 40-millisecond tone at each of 3 background white noise
levels (50, 65, and 80 dB). Deltamethrin (0, 2, 4, 6, or 8 mg/kg
body weight) or cismethrin (0, 6, 12, 18, or 24 mg/kg) were
administered orally in 0.2 ml/kg corn oil. Cismethrin and
deltamethrin produced similar dose-dependent decreases in motor
activity. The time course of onset and recovery for this decreased
activity was rapid (1 - 4 h). No cumulative effects on motor
activity of a 30-day exposure to 2 mg deltamethrin/kg per day or
6 mg cismethrin/kg per day were found. The effects of cismethrin
and deltamethrin on the acoustic startle response were dissimilar:
deltamethrin produced a dose-dependent decrease in amplitude and an
increase in latency, and cismethrin produced an increase in
amplitude and no change in latency. The differential effects of
cismethrin (Type I pyrethroids) and deltamethrin (Type II
pyrethroids) on the acoustic startle response may be related to the
contrasting effects previously shown with neurophysiological and/or
neurochemical techniques (see Appendix I).
7.8 Miscellaneous Effects
Analgesic effects of deltamethrin for thermic (hot plate test,
60 °C) and mechanical stimuli were investigated in mice and rats,
respectively. Deltamethrin prolonged the response-time to these
tests. Although this action was not significant at 500 mg
deltamethrin/kg body weight given orally, the reaction time was
increased at 1000 and 1500 mg/kg given orally in aqueous suspension
with 10% gum arabic (Chanh et al., 1981).
In rats, treatment with deltamethrin increased mean arterial
pressure and aortic output (Forshaw & Bradbury, 1983). The
cardiovascular effects of deltamethrin were due to both increased
catecholamine release in peripheral vascular beds, and to a direct
positive inotropic effect on the heart.
Krasnjih & Pavlova (1985) demonstrated induction of microsomal
oxygenases in rats 20 h after a single administration of 1/2 LD50.
Daily administration of 1/10th LD50 for 2 months reduced
acetylcholinesterase activity in the serum, erythrocytes, liver,
and cerebrum. It also led to some changes in the aspartate
aminotransferase activity and the urea and protein contents of
serum.
When rats were dosed orally with a single dose of 1/2 LD50 or 3
daily doses of 1/5 LD50 deltamethrin, the activities of transferrin
and ceruloplasmin in plasma, 20 h after dosing, were unchanged.
After the single dose, microsomal monooxygenase activity was
increased by 87%, and after the 3 doses, it was increased by 290%
(Kagan et al., 1986).
7.9 Potentiation
Deltamethrin was hydrolysed in vitro by esterases in blood,
brain, kidney, liver, and stomach preparations of mice.
Pretreatment of mice with the oxidase inhibitor, piperonyl butoxide
(PBO), or the esterase inhibitor, S,S,S-tributylphosphorotrithioate
(DEF), delayed metabolism of intraperitoneally administered
deltamethrin. PBO or DEF made mice more sensitive to deltamethrin
(Ruzo et al., 1979).
Plasma esterases, in addition to hepatic esterases, play a role
in the metabolism of deltamethrin in mammals and cause its rapid
detoxification by the oral route. In a potentiation study, a range
of esterase inhibitors, consisting mainly of organophosphorus
insecticides, was given to male rats in oral doses that inhibited
50% of the plasma cholinesterase. After 15 min, or 2 or 24 h, an
oral LD50 dose of deltamethrin EC formulation was given, which
showed potentiation with azinphos ethyl, omethoate, and dichlorvos.
It appears that users must handle deltamethrin in these
combinations very carefully because of their high toxicity.
Acephate, monocrotophos, phosphamidon, parathion methyl, and the 2
controls did not act as potentiators (Audegond et al., 1988).
7.10 Mechanism of Toxicity (Mode of Action)
Deltamethrin is classified as a Type II pyrethroid. For the
mode of action of pyrethroids in general see Appendix I.
The lowest concentration of deltamethrin to have an effect in
crayfish stretch receptor neurones on sodium channels was 10-12
mol/litre, but the response of the preparation to gamma-
aminobutyric acid (GABA) appeared to be unaffected by concentrations
of deltamethrin up to 10-7 mol/litre. Although 10-6 mol/litre
deltamethrin had a slight effect on the GABA response of the dactyl
abductor muscle, it appears that the majority of the effects of
cyano-pyrethroids in invertebrates could be accounted for solely by
their action on sodium channels (Chalmers et al., 1987).
Pyrethroid-induced motor symptoms, i.e., deltamethrin-induced
writhing and cismethrin-induced tremor, were studied, using a
number of pharmacological agents, in intact conscious rats and
spinal rats. The results suggest that pyrethroid-induced motor
symptoms, i.e., writhing and tremor, are mediated via a spinal site
of action, probably involving interneurones. Deltamethrin-induced
"non-motor" symptoms, i.e., increase in brain blood flow and blood
glucose may result from a supraspinal component of deltamethrin
activity. In contrast, the cardiovascular effects of deltamethrin
are mediated via a peripheral site of action (Bradbury et al.,
1983).
Tissue culture experiments have shown that the dorsal root
ganglion is more sensitive to deltamethrin than the spinal cord or
peripheral nerve fibres. The morphological alterations observed in
the neuronal bodies of the ganglia may reflect some perturbation of
the ionic equilibrium (Na+ and Ca+) (Souyri, 1985).
The results of several other, sometimes very detailed and
specialized, studies on the mode of action of deltamethrin have
been reported. Because these results do not basically influence
the present evaluation of deltamethrin, they are not reviewed in
detail. The interested reader is referred to the following
publications and a review by Bidet et al. (1988): Aldridge et al.
(1978), Duclohier & Georgescauld (1979), Gray et al. (1980),
Jacques et al. (1980), Miller & Adams (1980), Gammon et al. (1981),
Pichon (1981), Brodie & Aldridge (1982), Dyball (1982), Parkin &
LeQuesne (1982), Ray (1982), Staatz et al. (1982), Brodie (1983),
Takahashi & LeQuesne (1983), Berlin et al. (1984), Prasada Rao et
al. (1984), Bloomquist & Soderlund (1985), Brodie (1985), Brodie &
Opacka (1985), Bloomquist et al. (1986), Chinn & Narahashi (1986),
Doherty et al. (1986), Forshaw & Ray (1986), Staatz-Benson & Hosko
(1986), Brooks & Clark (1987), Forshaw et al. (1987), Leibowitz et
al. (1987), Lummis et al. (1987), Stein et al. (1987).
7.11 Experimental Studies on Antidotes
Treatments capable of counteracting acute deltamethrin
poisoning have been investigated in experimental animals. Ray &
Cremer (1979) have proposed atropine, Gammon et al. (1982),
phenobarbital and diazepam, and Bradbury et al. (1981, 1983),
mephenesin.
In order to find a therapeutically usable antagonist,
pharmacological screening was carried out by Dumont (1978), Dumont
& Chifflot (1978), and Dumont & Laurent (1979). The outcome was
that barbiturates are therapeutically active, but that the most
efficient product is ethyl carbamate. Cotonat et al. (1987) and
Fournier (1988) have confirmed that ethyl carbamate is an effective
treatment for severe deltamethrin poisoning. However, a serious
drawback is its antimitotic activity. Leclercq et al. (1986) began
by evaluating the activity of common anticonvulsants, such as
diazepam and clomethiazole. These products exhibited satisfactory
activity in rats and dogs (Thiebault et al., 1985, 1988).
Phenoprobamate and mephenesin carbamate have been shown to be
effective in the experimental treatment of deltamethrin poisoning
(Cotonat et al., 1987; Leclercq et al., 1986). A summary of the
results of the antidote studies can be found in Bleys et al.
(1986). Clinical trials based on these studies will be undertaken
(unpublished information given to the IPCS by Roussel Uclaf).
The therapeutic effects of methocarbamol have also been shown
by Hiromori et al. (1986). However, the mechanism underlying this
activity is not clear as the actual anticonvulsant activity is
weak.
It appears that for the time being barbiturates, and especially
diazepam, offer the safest symptomatic treatment in case of
deltamethrin poisoning. Advice on treatment for deltamethrin
poisoning is given in the IPCS Deltamethrin health and safety guide
(WHO, 1989).
8. EFFECTS ON MAN
8.1 General Population-Poisoning Incidents
A few cases of attempted suicides with deltamethrin
formulations (mainly EC), all non-fatal, have been reported in
anti-poison centres. Two typical cases are described below.
The first poisoning case in a 13-year-old girl who ingested
voluntarily 200 ml of a 2.5% EC formulation, (5 grams of
deltamethrin) was described by Rousselin (1983). After an unknown
time, she lost consciousness and developed generalized muscle
cramps, myosis, and tachycardia. Treatment in hospital was as
follows: gastric lavage, PAM 0.5 mg; atropine 2 mg; sodium
nitrite, 3% sodium thiosulfate; and, lastly, high doses of
diazepam. She completely recovered in 48 h.
A second poisoning case, concerning another attempted suicide
by a 23-year-old man, was reported by Foulhoux (1988). After oral
absorption of 70 cc of a 2.5% EC formulation (1.75 g pure
deltamethrin), there were no neurological signs in this patient.
Digestive and hepatic signs occurred, probably due to absorption of
the solvent, since determination of xylene in plasma was positive.
The patient was treated with haemodialysis, phenobarbital,
lidocaine, and provoked alkaline diuresis. Recovery followed
within 48 h.
8.2 Occupational Exposure
8.2.1 Acute toxicity-poisoning incidents
Rousselin (1983) described a case of poisoning in an
agricultural worker as a result of skin contamination with a liquid
containing 5 g deltamethrin/litre. He developed paraesthesia in
the legs, mouth, and tongue, and diarrhoea. Following washing of
the skin and administration of antihistamines, he still had
tingling sensations in his toes after 24 h, but was fully recovered
after 48 h.
Outbreaks of acute deltamethrin and fenvalerate poisoning
occurred in cotton growers in China in 1982 - 84. The farmers
handled the pyrethroid insecticides without taking any precautions.
Skin sensations occurred in more than 90% of the exposed workers.
After repeated spraying in the cotton fields, the mild cases
presented severe headaches, dizziness, fatigue, nausea, and
anorexia, with transient changes in the EEG. A severe case
developed muscular fasciculation, repetitive discharges in the EMG,
and frequent convulsions, which were treated with diazepam and
phenobarbital. However, in follow-up studies, all workers were
found to have made complete recovery, and the prognosis of acute
pyrethroid poisoning was found to be good (He, 1987; Tong Ying,
1988).
More recently He et al. (1989) reviewed 573 cases of acute
pyrethroid poisoning reported in the Chinese medical literature
during 1983 - 88. Among these there were 325 cases of acute
deltamethrin poisoning: 158 occupational, due to inappropriate
handling, and 167 accidental, mostly due to ingestion. Two
patients died of convulsions. All others recovered with
symptomatic and supportive treatment within 1 - 6 days. Clinical
manifestations are well reviewed (He, 1987).
8.2.2 Effects of short- and long-term exposure
Among plant workers dermally exposed to technical deltamethrin
or its formulations, cutaneous and mucuous manifestations were
observed. Initial lesions were tenacious and painful pruritus,
especially observed after exposure to hot water or perspiration,
followed by a blotchy local burning sensation with blotchy erythema
for about 2 days. Thereafter, slight and regular desquamation,
restricted to the contaminated area, occurred. Cutaneous signs
were sometimes accompanied by itching of the face (mainly around
the mouth) and/or rhinorrhoea or lachrymation (Husson, 1978).
Apart from the above-mentioned effects, no long-term or
persistent effect, or allergic diseases were reported in 70
workers, who had been exposed from 1977 - 87 in a deltamethrin-
manufacturing and -formulating plant in France (unpublished
Roussel Uclaf information supplied to the IPCS, 1988).
A field study was carried out in the United Kingdom with three
unprotected operators and one operator wearing hood, gloves, and
respirator, all of whom were involved in orchard spraying with
deltamethrin according to normal field practice. The exposure time
was 3.5 h. No changes were found in blood cell counts, total
protein, urea, alkaline phosphates, gamma-GT and SGOT in blood.
Little deltamethrin was found in the respirator pad and no residues
were found in the urine. There was no decrease in nerve conduction
velocity, but a slight tendency to the opposite reaction. None of
the operators experienced facial sensations (Hewson & Burgess, 1981).
Four operators were evaluated by the same authors during normal
field applications of deltamethrin lasting one day. Three of the
operators did not wear any protection on their heads or hands,
while one wore hood, gloves, and a respirator. Motor and sensory
nerve conduction velocities were determined as well as
haematological and biochemical parameters and urinalysis. No
changes were observed in the blood parameters measured and no
residues were found in the urine samples. Furthermore, nerve
conduction velocity did not decrease. Residues were primarily
confined to gloves and legs. None of the operators experienced
facial sensations (Hewson & Burgess, 1981).
Persons exposed to deltamethrin for 7 - 8 years in production
and formulation were subjected to clinical and haematological
examinations. Evaluations were conducted at several plants. There
were no measurable effects other than transient irritation of
cutaneous and mucous membranes, which was without sequelae.
Adequate precautionary measures, such as the wearing of gloves and
face masks, provided protection from exposure (Foulhoux, 1981).
A medical survey of agricultural workers involved in the use
and application of EC and WP formulations of deltamethrin in
Yugoslavia revealed no untoward symptoms of exposure, other than
itching and burning of the face, and nasal hypersecretion. Medical
examinations included chest X-ray, ECG, liver function tests,
neurological examinations (eye tonometry, Goldman perimetry, dark
adaptation ability), kidney function tests, and whole blood and
plasma cholinesterase activity. No adverse effects were noted.
The need for the proper use of masks and gloves, as well as good
personal hygiene (e.g., washing), was emphasized (Plestina, 1981).
Five healthy volunteers, 16 - 40 years of age, were exposed to
deltamethrin during 5 days of spraying in a cotton field in India
in 1981. A sixth volunteer was engaged in mixing and loading the
emulsion during the same period. Spraymen were exposed for 7 h
daily. No one complained about any symptoms. No clinical
abnormalities were detected, particularly with respect to
neurological examination (muscle power, coordination, tremors,
reflexes, and both light and deep sensations). No cardiovascular,
respiratory, or abdominal abnormalities were detected, and no skin,
mucous membrane, or eye lesions were observed during, and after
cessation of, exposure (Trivedi, 1981).
A health survey was carried out among spraymen exposed to 2.5%
deltamethrin emulsifiable concentrate in cotton fields in China.
The subjects were exposed to deltamethrin at concentrations of
0.022 - 24.070 µg/m3 in the air of the respiratory zone and
0.013 - 0.347 µg/cm2 of skin contact. One half of the 44 sprayers
complained of itching and burning sensations on their faces. A few
miliary red papules also appeared on the face of one of them, but
no signs of acute deltamethrin poisoning were noticed during
physical examination. There were no significant differences in the
sodium, potassium, and urea contents of the serum, the sodium,
potassium, ATPase, and serotonin contents of whole blood, and the
levels of 3-methyl-4-hydroxymandelic acid and 5-hydroxy-
indoleacetic acid in the urine between the subjects examined and
the controls. Deltamethrin in the urine of spraymen was below the
detection limit of 0.10 µg/litre (Wang et al., 1988).
Mestres et al. (1985) measured the dermal and inhalation
exposure of mixer/applicators who applied deltamethrin to vegetable
crops in greenhouses in southern France and of workers who picked
fruit from treated trees in the same area. This appeared to be
less than 0.0065% of the toxic dose per hour with a mask and
0.0017%, without a mask.
In a department producing an aerosol of the domestic Bulgarian
insecticide "Dekazol" containing 0.02, 0.04, or 0.08% deltamethrin,
severe subjective complaints of sensory irritation were found
because of the high levels of contamination of the workplace air
with deltamethrin and also dermal contamination. Skin irritation
with conjunctivitis and irritation of the respiratory system were
discovered in all 25 workers. Two of them had contact urticaria.
Patch testing with 0.03% deltamethrin showed a positive reaction in
5 out of 23 workers tested (Bainova et al., 1986).
8.3 Clinical Studies
Three formulations of deltamethrin in petroleum solvent were
patch tested on 37 human volunteers (double blind trial against
solvent control). A dose of 20 µl of a 1% suspension in water, of
a 25 g/litre emulsifiable concentrate was put on the facial skin of
each volunteer, with a randomized distribution of control and
active dilution. The duration of the irritation was short (from
some minutes to 1 h) and the severity was described as slight by
most of the volunteers. No skin damage was reported (Foulhoux et
al., 1981).
9. PREVIOUS EVALUATIONS BY INTERNATIONAL BODIES
The Joint FAO/WHO Meeting on Pesticide Residues (JMPR)
discussed and evaluated deltamethrin at its meetings in 1980, 1981,
1982, 1984, 1985, 1986, 1987, and 1988 (FAO/WHO, 1981, 1982, 1983,
1985a, 1986a, 1986b, 1988a,b,c). In 1982, an acceptable daily
intake (ADI) of 0 - 0.01 mg/kg body weight was established.
The following Maximum Residue Limits (MRLs), in mg/kg, resulted
from these evaluations:
tea 10.0
hops dry, wheat bran unprocesseda 5.0
coffee beans (post-harvest) 2.0
wheat wholemeal,a cereal grains,a 1.0
(ph) lentil (dry),a beans (dry),a
field pea (dry)a
straw and fodder (dry) of cereal 0.5
grains, legume animal feeds (dry
weight), leafy vegetables
brassica leafy vegetables,a edible 0.2
peel of fruiting vegetablesa
bulb vegetables, edible peel of 0.1
assorted fruits, legume vegetables,
oilseeds, pome fruits, wheat floura
artichokes, bananas, clementines, 0.05
coco beans, grapes, kiwi fruit,
oranges (sweet, sour), stone
fruits/strawberries
legume oilseeds, melons, 0.01
mushrooms, pineapples, root and
tuber vegetables, milksa
WHO has classified deltamethrin as a moderately hazardous
technical product in normal use (WHO, 1988). A Data Sheet on
deltamethrin (No. 50) has been issued (WHO/FAO, 1984).
-------------------------------------------------------------------
a Not yet confirmed by Codex Alimentarius Commission (FAO/WHO,
1986c, and 1988c).
REFERENCES
ADENEY, R.J., GRZYWACZ, D., & MATTHIESSEN P. (1980) The acute
toxicities of pyrethrin analogues to Sarotherodon mossambicus
(Peters), Centre for Overseas Pest Research, 6 pp. (Unpublished
proprietary data submitted to WHO by Roussel Uclaf).
AKHTAR, M.H. (1984) Metabolism of deltamethrin by cow and chicken
liver enzyme preparations. J. agric. food Chem., 32: 258-262.
AKHTAR, M.H., HAMILTON, R.M.G., & TRENHOLM, H.L. (1985)
Metabolism, distribution and excretion of deltamethrin by leghorn
hens. J. agric. food Chem., 33: 610-617.
AKHTAR, M.H., MARTIN, K.E., & TRENHOLM, H.L. (1986) Fate of
14C-deltamethrin in lactating dairy cows. J. agric. food Chem.,
34: 753-758.
AKHTAR, M.H., DANIS, C., TRENHOLM, H.L., & MARTIN, K.E. (1987)
Residues in milk and tissues of lactating dairy cows fed
deltamethrin for 28 consecutive days. J. agric. food Chem.,
ALDRIDGE, W.N., CLOTHIER, B., FORSHAW, P., & JOHNSON, M.K., PARKER,
V.H., PRICE, R.J., SKILLETER, D.N., VERSCHOYLE, R.D., STEVENS,
C. (1978) The effect of DDT and the pyrethroids cismethrin and
decamethrin on the acetyl choline and cyclic nucleotide content of
rat brain. Biochem. Pharmacol., 27: 1703-1706.
ALINIAZEE, M.T. & CRANHAM, J.E. (1980) Effects of four synthetic
pyrethroids on a predatory mite, Typhlodroums pyri, and its prey,
Panonychus ulmi, on apples in southeast England. Environ. Entomol.,
9: 436-439.
ARZONE, A. & VIDANO, C. (1978) [Azione sull'ape di etiofencaus,
decamethrin e ciexatin.] Apic. mod., 69: 157-162, (in Italian).
ATKINS, E.L., KELLUM D., & NEUMAN, K.J. (1976) Effect of
pesticides on apiculture project No. 1449: The annual report,
University of California Riverside, pp. 536-567.
AUDEGOND, L., CATEZ, D., FOULHOUX, P., FOURNEX, R., LE RUMEUR, C.,
L'HOTELLIER, M., & STEPNIEWSKI, J.P. (1988) Potentialisation de
la toxicité de la deltaméthrine par les insecticides
organophosphorés (Unpublished proprietary data submitted to WHO by
Roussel Uclaf).
AUDEGOND L., COLLAS E., & GLOMOT R. (1981) RU 22974
(deltamethrin) single administration study by oral route in the rat
(the compound is given as a suspension) (Unpublished report RU-
81239/A, submitted to WHO by Roussel Uclaf).
BAINOVA, A. & KALOYANOVA, F. (1985) [Study of allergenic and
irritating effect of synthetic pyrethroids on skin.] Hig. Zdrav.,
28(2): 19-21 (in Bulgarian).
BAINOVA, A., MIHAVSKI, M., ISMIROVA, N., & BENCHEV, V. (1986)
Specific skin irritation after contact with specific pyrethroids.
6th Congress of Bulgarian Dermatologists, Varna, 2-5 October, 1986,
(Abstract No. 74) (in Bulgarian).
BAKER, P.G. & BOTTOMLEY, P. (1982) Determination of residues of
synthetic pyrethroids in fruit and vegetables by gas-liquid and
high-performance liquid chromatography. Analyst, 107: 206-212.
BALDRY, D.A.T., EVERTS, J., ROMAN, B., BOON VON OCHSSEE, G.A., &
LAVEISSIERE, C. (1981) The experimental application of
insecticides from a helicopter for the control of riverine
populations of Glossina tachinoides in West Africa. Part VIII: The
effects of two spray applications of OMS-570 (endosulfan) and of
OMS 1998 (decamethrin) on G. tachinoides and non-target organisms
in Upper Volta. Trop. Pest Manage., 27(1): 83-110.
BEAVERS, J.B. & FINK, R. (1977a) Actue oral LD50 Mallard duck
technical DECIS final report, (Unpublished report WI77.06.06/A,
submitted to WHO by Roussel Uclaf). Wildlife International.
BERLIN, J.R., AKERA, T., BRODY, T.M., & MATSUMURA, F. (1984) The
inotropic effects of a synthetic pyrethroid decamethrin on isolated
guinea pig atrial muscle. Eur. J. Pharmacol., 98: 313-322.
BIDET, D., DUMONT, C., FOULHOUX, P., FOURNEX, R., & ROUGERON, C.
(1988) Mechanisms responsible for the toxicity of deltamethrin and
other pyrethroids. (Unpublished document, submitted to WHO by
Roussel Uclaf).
BLEYS, M., COTONAT, J., FOULHOUX, P. (1986) Lettre à l'éditeur,
J. Toxicol. clin. exp., 6(3): 211-212.
BLOOMQUIST, J.R., SODERLUND, D.M. (1985) Neurotoxic insecticides
inhibit GABA-dependent chloride uptake by mouse brain vesicles.
Biochem. biophys. Res. Commun., 133(1): 37-43.
BLOOMQUIST, J.R., ADAMS, P.M., & SODERLUND, D.M. (1986)
Inhibition of gamma-aminobutyric acid-stimulated chloride flux in
mouse brain vesicles by polychlorocycloalkane and pyrethroid
insecticides. Neurotoxicology, 7(3): 11-20.
BOCQUET, J.C., PASTRE, P., ROA, L., & BAUMEISTER, R. (1980) Etude
de l'action de la deltaméthrine sur Apis mellifera en conditions de
plein-champ. Phytiatr. Phytopharm., 29: 83-92.
BOCQUET, J.C., PASTRE, P., & BAUMEISTER, R. (1983) Bilan de cinq
années d'études de l'effet de la deltaméthrine sur abeilles en
conditions naturelles. 6ème Congrès International du Colza, Paris,
mai 1983.
BOUCHE, M.B. & FAYOLLE, L. (1979) Tests carried out with Procida
compound Decis EC 2.5 on earthworms lethal effect in relation with
time (Unpublished proprietary report No. INRA-VT-79.05.22/78.10.
10/A, submitted to WHO by Roussel Uclaf).
BOWMAN, H. & CARPENTER, M. (1987) Determination of
photodegradation of 14C-deltamethrin in aqueous solution
(Unpublished proprietary report ABC LABS 35491, submitted to WHO by
Roussel Uclaf).
BRADBURY, J.E., GRAY, A.J., & FORSHAW, P. (1981) Protection
against pyrethroid toxicity in rats with mephenesin. Toxicol.
appl. Pharmacol., 60: 382.
BRADBURY, J.E., FORSHAW, P.J., GRAY, A.J., & RAY, D.E. (1983) The
action of mephenesin and other agents on the effects produced by
two neurotoxic pyrethroids in the intact and spinal rat.
Neuropharmacology, 22(7): 907-914.
BRODIE, M.E. (1983) Correlations between cerebellar cyclic GMP
and motor effects induced by deltamethrin: independence of olivo-
cerebellar tract. Neurotoxicology, 4(4): 1-11.
BRODIE, M.E. (1985) Deltamethrin infusion into different sites in
the neuraxis of freely moving rats. Neurobehav. Toxicol. Teratol.,
7(1): 51-55.
BRODIE, M.E. & ALDRIDGE, W.N. (1982) Elevated cerebellar cyclic
GMP levels during the deltamethrin-induced motor syndrome.
Neurobehav. Toxicol. Teratol., 4: 109-113.
BRODIE, M.E. & OPACKA, J. (1985) Dissociation between circling
behaviour and striatal dopamine activity following unilateral
deltamethrin administration to rats. Naunyn-Schmiedeberg's Arch.
Pharmacol., 331: 341-346.
BROOKS, M.W. & CLARK, J.M. (1987) Enhancement of norepinephrine
release from rat brain synaptosomes by alpha cyano pyrethroids.
Pestic. Biochem. Physiol., 28: 127-139.
BUCCAFUSCO, R.J., ELLS, S.J., & CARY, G.A. (1977a) Acute toxicity
of NRDC 161 to bluegill (Lepomis macrochirus) under dynamic test
conditions, E.G. & G. Bionomics Aquatic Toxicology Laboratory, 12 pp.
(Unpublished proprietary data submitted to WHO by Roussel Uclaf).
BUCCAFUSCO R.J., ELLS, S.J., & CARY, G.A. (1977b) Acute toxicity
of NRDC 161 (Decamethrine) to Channel Catfish (Ictalurus
punctatus), E.G. & G. Bionomics Aquatic Toxicology Laboratory,
7 pp. (Unpublished proprietary data supplied to WHO by Roussel
Uclaf).
CABRAL, J.R.P., GALENDO, D., LAVAL, M., & LYANDRUT, N. (1986)
Carcinogenicity study of the pesticide deltamethrin in mice and
rats. Summary Report in Poster Session at IUPAC Meeting, Ottawa,
August 1986.
CARY, G.A. (1978) Kinetics of 14C-NRDC-161 in a model aquatic
ecosystem, E.G. & G. Bionomics Aquatic Toxicology Laboratory.
(Unpublished proprietary report BW-78-2-075, submitted to WHO by
Roussel Uclaf).
CHALMERS, A.E., MILLER, T.A, & OLSEN, R.W. (1987) Deltamethrin:
a neurophysiological study of the sites of action. Pestic.
Biochem. Physiol., 27: 36-41.
CHAMBON, A. & LEPAILLEUR, H. (1984) Etude de l'effet toxique de
la deltamethrine (produit technique > 98%) et de la formulation CE
25 g/l Decis vis-à-vis de l'espece de vers (Eisenia fetida andrei)
(Unpublished proprietary report IRCHA-84.30.07/F, submitted to WHO
by Roussel Uclaf).
CHANH, P.H., NAVARRO-DELMASURE, C., CHANH, A.P.H., LEAN, C.S.,
ZIADE, F., & SAMAHA, F. (1981) Analgesic effects of decamethrin,
Surg. Transplant., 9: 503-504.
CHAPMAN, R.A. & HARRIS, C.R. (1981) Persistence of four
pyrethroid insecticides in a mineral and organic soil. J. environ.
Sci. Health, B16: 605-615.
CHAPMAN, R.A., TU, C.M., HARRIS, C.R., & COLE, C. (1981)
Persistence of five pyrethroid insecticides in sterile and natural,
mineral and organic soil. Bull. Environ. Contam. Toxicol, 26:
513-519.
CHESTERMAN, H., HEYWOOD, R., PERKIN, C.J., BEARD, D., STREET, E., &
PRENTICE, D.E. (1977) RU 22974. Oral toxicity study in Beagle
dogs, Huntingdon, Huntingdon Research Centre, (Unpublished report
RSL 253/7751/A3, submitted to WHO by Roussel Uclaf).
CHINN, K. & NARAHASHI, T. (1986) Stabilization of sodium channel
states by deltamethrin in mouse neuroblastoma cells. J. Physiol.,
380: 191-207.
CLAIR, M. (1977) RU 22974 DECIS. Acute toxicity in the rabbit by
percutaneous administration. Joinville-le-Pont, Institut Français
de Recherches et Essais Biologique (Unpublished report IFREB-R
770257.1/A, submitted to WHO by Roussel Uclaf).
CLARK, G.C., JACKSON, G.C., & ALEXANDER, D.J. (1980) Acute
inhalation toxicity in rats, 4-hour exposure (Decis PM 2.5
percent), Huntingdon Research Centre, (Unpublished report RSL
437/80568, submitted to WHO by Roussel Uclaf).
COOMBS, D.W. & CLARK, G.C. (1978) RU 22974: Acute inhalation
toxicity in rats. 6 Hour LC50. Huntingdon, Huntingdon Research
Centre, (Unpublished report RSL 310/78453/A, submitted to WHO by
Roussel Uclaf).
COOMBS, D.W., CLARK, G.C., STREET, A.E., & GIBSON, W.A. (1978) RU
22974 inhalation toxicity study in rats 14 x 6 hour exposures over
a period of 3 weeks (Unpublished proprietary report
RSL/318/78638/A, submitted to WHO by Roussel Uclaf).
COQUET, B. (1976a) RU 22974. Test to determine primary cutaneous
irritation in the rabbit. Joinville-le-Pont, Institut Francais de
Recherches et Essais Biologiques (Unpublished report IFREB-R
761157/A, submitted to WHO by Roussel Uclaf).
COQUET, B. (1976b) RU 22974. Test to evaluate ocular irritation
in the rabbit, Joinville-le-Pont, Institut Français de Recherches
et Essais Biologiques (Unpublished report IFREB- R 761158/A,
submitted to WHO by Roussel Uclaf).
COQUET, B. (1977) RU 22974. Decis formulations - Determination of
the LD50 in the rat by oral administration, Joinville-le-Pont,
Institut Français de Recherches et Essais Biologiques (Unpublished
report 770257-A, submitted to WHO by Roussel Uclaf).
COTONAT, J., BLEYS, M., & FOULHOUX, P. (1987) Effets antagonistes
du phenprobamate et du carbamate de mephenesine sur l'intoxication
à la deltaméthrine. J. Toxicol. clin. exp., 7: 5-19.
CROFTON, K.M. & REITER, L.W. (1984) Effects of two pyrethroid
insecticides on motor activity and the acoustic startle response in
the rat. Toxicol. appl. Pharmacol., 75: 318-328
DAVID, D. (1981) Laboratory evaluation of repellent properties
against birds of the synthetic pyrethroid decamethrin. Poult. Sci.,
60: 1149-1151.
DAVIES, J.E., MUNT, P.L., & GOOR, J.L. (1983) RU 22974.
Investigation of possible neurological effects using the tilting
plane test. Huntingdon, Huntingdon Research Centre (Unpublished
proprietary report RSL603/83232/A, submitted to WHO by Roussel
Uclaf).
DE LAVAUR, E., LE SECH, J., & GROLLEAU, G. (1985) Accumulation et
élimination de la deltaméthrine chez la caille japonaise. Ann.
Fals. Exp. Chim., 835: 73-78.
DOHERTY, J.D., LAUTER, C.J., & SALEM, N., Jr (1986) Synaptic
effects of the synthetic pyrethroid resmethrin in rat brain in
vitro. Comp. Biochem. Physiol., C84(2): 373-9.
DUCLOHIER, H. & GEORGESCAULD, D. (1979) The effects of the
insecticide decamethrin on action potential and voltage-clamp
currents of myxicola giant axon. Comp. Biochem. Physiol., C62(2):
217-223.
DUMONT, C. & CHIFFLOT, L. (1978) Recherche d'un antidote contre
les effets toxiques aigus (Unpublished report RU-AE-72, submitted
to WHO by Roussel Uclaf).
DUMONT, C. & LAURENT, J. (1979) Recherche d'antagonistes des
effets neurotoxiques aigus du RU 22 974 (Unpublished report RU-AG-
35, submitted to WHO by Roussel Uclaf).
DUNNING, R.A., COOPER, J.M., WARDMAN, J.M., & WINDER, G. (1981)
Susceptibility of the carabid Pterostichus melanarius (Illiger) to
aphicide sprays applied to the sugar-beet crop. (Unpublished
proprietary report UK-SOIL-F.81/A, submitted to WHO by Roussel
Uclaf).
DYBALL, R.E.J. (1982) Inhibition by decamethrin and resmethrin of
hormone release from the isolated rat neurohypophysis - a model
mammalian neurosecretory system. Pestic. Biochem. Physiol., 17:
42-47.
ELLIOTT, M. (1977) Synthetic pyrethroids, Washington, DC,
American Chemical Society, p. 229 (ACS Symposium Series 42).
ELLIOTT, M., FARNHAN, A.W., JANES, N.F., NEEDHAM, P.H., & PULMAN,
D.A. (1974) Synthetic insecticide with a new order of activity.
Nature, 248: 710-711.
ESTESEN, B.J., BUCH, N.A., & WARE, G.W. (1979) Dislodgeable
insecticide residue on cotton foliage; permethrin, curacron,
fenvalerate, sulprofos, decis and endosulfan. Bull. environ.
Contam. Toxicol., 22: 245-248.
EVANS, M.H. (1976) End-plate potentials in frog muscle exposed to
a synthetic pyrethroid. Pestic. Biochem. Physiol., 6: 547-550.
EVERTS, J.W., VAN FRANKENHUYZEN, K., ROMAN, B., & KOEMAN, J.H.
(1983) Side effects of experimental pyrethroid applications for
the control of tsetse flies in a riverine forest habitat in Africa.
Arch. environ. Contam. Toxicol., 12: 91-97.
EVERTS, J.W., KORTENHOFF, B.A., HOOGLAND, H., VLUG, H.J., JACQUE,
R., & KOEMAN. J.H. (1985) Effects on man-target terrestrial
arthropods of synthetic pyrethroids asked for the control of the
Tsetse Fly ( Glossina spp.) in settlement areas of the Southern
Ivory Coast, Africa. Arch. environ. Contam. Toxicol., 14: 647-650.
FAO (1982) Second Government Consultation on International
Harmonization of Pesticide Registration Requirements, Rome, 11-15
October, 1982, Rome, Food and Agriculture Organization of the
United Nations.
FAO/WHO (1981) 1980 Evaluations of some pesticide residues in
food, Rome, Food and Agriculture Organization of the United Nations
(FAO Plant Production and Protection Paper No. 26 Sup).
FAO/WHO (1982) 198l Evaluations of some pesticide residues in
food, Rome, Food and Agriculture Organization of the United Nations
(FAO Plant Production and Protection Paper No. 42).
FAO/WHO (1983) 1982 Evaluations of some pesticide residues in
food, Rome, Food and Agriculture Organization of the United Nations
(FAO Plant Production and Protection Paper No. 49).
FAO/WHO (1985a) 1984 Evaluations of some pesticide residues in
food, Rome, Food and Agriculture Organization of the United Nations
(FAO Plant Production and Protection Paper No. 67).
FAO/WHO (1985b) Guide to Codex recommendations concerning
pesticide residues. Part 8. Recommendations for methods of
analysis of pesticide residues, 3rd ed., Rome, Food and Agriculture
Organization of the United Nations, Codex Committee on Pesticide
Residues.
FAO/WHO (1986a) 1985 Evaluations of some pesticide residues in
food. Part I - Residues, Rome, Food and Agriculture Organization
of the United Nations (FAO Plant Production and Protection Paper
No. 72/1).
FAO/WHO (1986b) 1986 Evaluations of some pesticide residues in
food. Part I - Residues, Rome, Food and Agriculture Organization
of the United Nations (FAO Plant Production and Protection Paper
No. 78).
FAO/WHO (1986c) Codex maximum limits for pesticide residues, 2nd
ed., Rome, Food and Agriculture Organization of the United Nations,
Codex Alimentarius Commission, CAC XIII.
FAO/WHO, (1988a) 1987 Evaluations of some pesticide residues in
food. Part I - Residues, Rome, Food and Agriculture Organization
of the United Nations (FAO Plant Production and Protection Paper
No. 86/1).
FAO/WHO, (1988b) 1988 Evaluations of some pesticide residues in
food. Part 1 - residues, Rome, Food and Agriculture Organization
of the United Nations. (FAO Plant Production and Protection Paper
93/1.
FAO/WHO, (1988c) Supplement 1 to Codex maximum limits for pesticide
residues, Rome, Food and Agriculture Organization of the United
Nations.
FISCHER, L. & CHAMBON, J.P. (1987) Faunistical inventory of
cereal arthropods after flowering and incidence of insecticide
treatments with deltamethrin, dimethoate and phosalone on the
apigeal fauna. Meded. Fac. Landbouwwet. Rijksuniv. Gent, 52(2a):
FLANNIGAN, S.A. & TUCKER, S.B. (1985) Variation in cutaneous
sensation between synthetic pyrethroid insecticides. Contact
Dermatitis, 13: 140-147.
FLORELLI, F., GARNIER, P., & ROA, L. (1987a) Bilan de 8 années
d'expérimentation sur la selectivite du Decis vis à vis des
abeilles - La défense des végétaux, 243: 8
FLORELLI, F., HELLER, J.J., GARNIER, P., & BAUMEISTER, R. (1987b)
Incidence sur les abeilles, les ruches et leur production, de
traitements aériens du colza avec la deltaméthrine - Annales ANPP -
Conférence Internationale sur les Ravageurs en Agriculture, Paris,
1-3 décembre 1987, 189-203.
FORSHAW, P.J. & BRADBURY, J.E. (1983) Pharmacological effects of
pyrethroids on the cardiovascular system of the rat. Eur. J.
Pharmacol., 91: 207-213.
FORSHAW, P.J. & RAY, D.E. (1986) The effects of two pyrethroids,
cismethrin and deltamethrin, on skeletal muscle and the trigeminal
reflex system in the rat. Pestic. Biochem. Physiol., 25: 143-151.
FORSHAW, P.J., LISTER, T., & RAV, D.E. (1987) The effects of two
types of pyrethroid on rat skeletal muscle. Eur. J. Pharmacol.,
134(1): 89-96.
FOUILLET, X. (1976) RU 22974 Mutagenicity study of various
preparations. Salmonella microsome test, Institut Français de
Recherches et Essais Biologiques (Unpublished report no. IFREB-R
761153/A, submitted to WHO by Roussel Uclaf).
FOULHOUX, P. (1981) Medical observations of personnel working on
synthesis or formulation of deltamethrin (Unpublished report RU-
81.18.06/A, submitted to WHO by Roussel Uclaf).
FOULHOUX, P. (1988) Assessment of potential adverse effects of
deltamethrin on man, Département Central de Toxicovigilance Roussel
Uclaf, 16 pp. (Unpublished report submitted to WHO by Roussel
Uclaf).
FOULHOUX, P., BOGGIN, CORNILLIET, POTTIER, RAYNAUD, STERN-VEYRIN, &
SUTTET (1981) Cutaneous irritation patch test on human volunteers
with Decis EC25, Decis flowable 25, Decis + mineral oil 25
(Unpublished report RU-81.03.02/DM/A, submitted to WHO by Roussel
Uclaf).
FOURNIER, P.E. (1988) Etude de l'activité antidote du carbamate
d'éthyl dans l'intoxication aiguë par la deltaméthrine chez le rat,
Paris, Unité de Pharmacologie Clinique, Hospital Fernand Widal
(Unpublished report submitted to WHO by Roussel Uclaf).
GAINES, T.B. & LINDER, R.E. (1986) Acute toxicity of pesticides
in adult and weanling rats, Fundam. appl. Toxicol., 7: 299-308.
GAMMON, D.W. & CASIDA, J.E. (1983) Pyrethroids of the most potent
class antagonize GABA action at the crayfish neuromuscular
junction. Neurosci. Lett., 40: 163-168.
GAMMON, D.W., BROWN, M.A., & CASIDA, J.E. (1981) Two classes of
pyrethroid action in the cockroach. Pestic. Biochem. Physiol., 15:
181-191.
GAMMON, D.W., LAWRENCE, L.J., & CASIDA, J.E. (1982) Pyrethroid
toxicology: Protective effects of diazepam and phenobarbital in
the mouse and the cockroach. Toxicol. appl. Pharmacol., 66:
290-296.
GLICKMAN, A.H. & CASIDA, J.E. (1982) Species and structural
variations affecting pyrethroid neurotoxicity. Neurobehav.
Toxicol. Teratol., 4(6): 793-799.
GLOMOT, R. (1979) Acute toxicity study by oral route in the rat
(Proprietary report RU-79803-54/A2, submitted to WHO by Roussel
Uclaf).
GLOMOT, R. & CHEVALIER, B. (1976a) RU 22974. Acute toxicity
study mouse and rat by oral route (Unpublished report TOX 76810/A,
submitted to WHO by Roussel Uclaf).
GLOMOT, R. & CHEVALIER, B. (1976b) RU 22974. Acute toxicity
study mouse and rat by intraperitoneal route (Unpublished report
TOX 76811/A, submitted to WHO by Roussel Uclaf).
GLOMOT, R. & CHEVALIER, B. (1976c) RU 22974. Acute toxicity
study mouse and rat by intravenous route (Unpublished report TOX
76812/A, submitted to WHO by Roussel Uclaf).
GLOMOT, R. & VANNIER, B. (1977) RU 22974. Teratological study in
mouse, rat and rabbit (Unpublished report TOX 76534-76536/2/A,
submitted to WHO by Roussel Uclaf).
GLOMOT, R. & VANNIER, B. (1978) RU 22974. Teratological study in
rabbit. Complementary information (Unpublished report TOX 76534-
76536/A3, submitted to WHO by Roussel Uclaf).
GLOMOT, R., AUDEGOND, L., & COLLAS, E. (1979) Acute oral toxicity
study in the rat (Decis 25 G/L Mixofluid) (Proprietary report RU
79824/A, submitted to WHO by Roussel Uclaf).
GLOMOT, R., AUDEGOND, L., & COLLAS, E. (1980a) Single
administration by oral route in the mouse (Decis Wettable Powder,
2.5%) (Proprietary report RU-80817/A, submitted to WHO by Roussel
Uclaf).
GLOMOT, R., AUDEGOND, L., & COLLAS, E. (1980b) Single
administration study by oral route in the dog (Decis Wettable
Powder, 2.5%) (Proprietary report RU 80194/A, submitted to WHO by
Roussel Uclaf).
GLOMOT, R., AUDEGOND, L., & COLLAS, E. (1981a) Single
administration study by oral route in the rat (Proprietary report
RU-81239/A, submitted WHO by Roussel Uclaf).
GLOMOT, R., AUDEGOND, L., & COLLAS, E. (1981b) Primary dermal
irritation study in the rabbit. Deltamethrin 25 G/L Mixofluid
(Proprietary report RU-79193/A, submitted to WHO by Roussel Uclaf).
GLOMOT, R., AUDEGOND, L., & COLLAS, E. (1981c) Primary dermal
irritation study in the rabbit (Deltamethrin wettable powder, 2.5%)
(Proprietary report RU-80200/A, submitted to WHO by Roussel Uclaf).
GLOMOT, R., AUDEGOND, L., & COLLAS, E. (1981d) Primary eye
irritation study in the rabbit (Deltamethrin in 25 G/L Mixofluid)
(Proprietary report RU-79192/A, submitted to WHO by Roussel Uclaf).
GLOMOT, R., AUDEGOND, L., & COLLAS., E. (1981e) Primary eye
irritation study in the rabbit (Decis Wettable Powder, 2.5%)
(Proprietary report RU-79192/A, submitted WHO by Roussel Uclaf).
GLOMOT, R., CHEVALIER, B., COLLAS, E., & AUDEGOND, L. (1977) RU
22974. Acute toxicity study by oral route in male and female
Beagle dogs (Unpublished report TOX 77804/JL-5, submitted to WHO
by Roussel Uclaf).
GOLDENTHAL, E.I., BLAIR, M., JEFFERSON, N.D., SPICER, E.J.F.,
ARCEO, R.J., & KAHN, A., III (1980a) RU 22974. Two year toxicity
and carcinogenicity study in mice, Mattawan, Michigan,
International Research and Development Corporation (Unpublished
report IRDC 406-001/A-4, submitted to WHO by Roussel Uclaf).
GOLDENTHAL, E.I., JEFFERSON, N.D., BLAIR, M., THORSTENSON, J.H.,
SPICER, E.J.F., ARCEO, R.J., & KAHN, A., III (1980b) RU 22974.
Two year oral toxicity and carcinogenicity study in rats,
Mattawan, Michigan, International Research and Development
Corporation (Unpublished report IRDC 406-002/A.4, submitted to WHO
by Roussel Uclaf).
GRANDADAM, A. (1976) Test to determine the toxicity in the
chicken by oral route (Unpublished report RU76.05.05/A, submitted
to WHO by Roussel Uclaf).
GRAY, A.J. & RICKARD, J. (1982) The toxicokinetics of
deltamethrin in rats after intravenous administration of a toxic
dose. Pestic. Biochem. Physiol.., 18: 205-215.
GRAY, A.J., CONNORS, T.A., & RICKARD, J. (1980) Mechanism of
mammalian toxicity of the pyrethroids. In: Littaner, U.Z. et al.,
ed. Neurotransmitters and their receptors, New York, John Wiley
and Sons, pp. 565-568.
GROLLEAU G. & GRIBAN, J. (1976a) Toxicity of deltamethrin or
DECIS in single ingestion in game duck Anas platyrhnchos L.,
JoyenJossas, France, Institut National de la Recherche Agronomique
(Unpublished report INRA-76.21 12/A, submitted to WHO by Roussel
Uclaf).
GROLLEAU, G. & GRIBAN, J. (1976b) Toxicity of deltamethrin or
DECIS by single ingestion in grey partridge, Perdix Perdix L. and
red partridge, Alectotis rufa L., JoyenJossas, France, Institut
National de la Recherche Agronomique (Unpublished report INRA76.28
09/A, submitted to WHO by Roussel Uclaf).
GUILLOT, J.P. & GUILAINE, J. (1977) RU 22974. Decaméthrine.
Decis technical Roussel Uclaf. Sensitization test in the guinea
pig. Joinville-le-Pont, Institut Français de Recherches et Essais
Biologique (Unpublished report no. IFREB-R 709241/A, submitted to
WHO by Roussel Uclaf).
GULYAS P. & CSANYI B. (undated) Acute fish toxicological
investigation of diverse insecticides, Hungary, Scientific Research
Centre of Economy of Water Supplies, 21 pp. (Unpublished
proprietary data supplied to WHO by Roussel Uclaf).
HALLS, G.R.H. & PERIAM, A.W. (1980) The fate of residues of NRDC
161 on wheat during storage and after milling and baking - Report
after 9 months storage, November 1980, Wellcome Research
Laboratories Report (Unpublished report HEFH 80-4, submitted to
WHO by Roussel Uclaf).
HARGREAVES, J.R. & COOPER, L.P. (1979) Phytotoxicity tests with
pyrethroid insecticides on glasshouse grown tomato seedlings.
Queensland J. Agric. anim. Sci., 36: 151-154.
HASCOET, M. (1977) Laboratory leaching soil study with
decamethrin (RU 22974), Versailles, Institut National de la
Recherche Agronomique (Unpublished proprietary report INRA-
77.20.09/A, submitted to WHO by Roussel Uclaf).
HE, F. (1987) Occupational neurotoxicology: Current problems and
trends. Paper presented at the XXII International Congress on
Occupational Health, Sydney, 27 September-2 October, 1987.
HE, F., WANG, S., LIU, L., CHEN, S., ZHANG, Z., & SUN, J. (1989)
Clinical manifestations and diagnosis of acute pyrethroid
poisoning. Arch. Toxicol., 63: 54-58.
HEHEITMULLER, T., SHUBA, P.J., & PARRISH, R. (1978) Acute
toxicity of Decis to sheepshead minnows (Cyprinodon variegatus),
E.G. & G. Bionomics Marine Laboratory, 7 pp. (Unpublished
proprietary report BR-78-12219, supplied to WHO by Roussel Uclaf).
HEWSON, R.T. & BURGESS, J.E. (1981) An operator study with
deltamethrin including measurements of nerve conduction times
(Unpublished proprietary report GB-NT-11.81A, submitted to WHO by
Roussel Uclaf).
HILL, B.D. (1982) Determination of Deltamethrin residues in
soils, Alberta, Agriculture Canada Research Station, 7 pp
(Unpublished proprietary report CAN-82.12.10/A, submitted to WHO by
Roussel Uclaf).
HILL, B.D. (1983) Persistence of deltamethrin in a Lethbridge
sandy clay loam. J. environ. Sci. Health, B18(6): 691-703.
HILL, B.D. & JOHNSON, D.L. (1987) Persistence of deltamethrin and
its isomers on pasture forage and litter. J. agric. Food Chem.,
35: 373-378.
HILL, B.D. & SCHAALJE, G.B. (1985) A two-compartment model for
the dissipation of deltamethrin on soil. J. agric. food Chem., 33:
1001-1006.
HIROMORI, T., NAKANISHI, T., KAWAGUCHI, S., SAKO, H., SUZUKI, T., &
KIYAMOTO, J. (1986) Therapeutic effects of methocarbamol on acute
intoxication by pyrethroids in rats. J. pestic. Sci., 11: 9-14.
HUNTER, B., JORDAN, J., HEYWOOD, R., HEPWORTH, P., STREET, A.E., &
PRENTICE, D.E. (1977) RU 22974. Assessment of toxicity to rats
by oral administration for 13 weeks (followed by a 4-week
withdrawal period) (Unpublished report RSL-254/76938/A3, submitted
to WHO by Roussel Uclaf).
HUSSON, J.M. (1978) Medical observations made on people working
on the manufacture and formulation of the pyrethroid insecticide
decamethrin (Unpublished report RU 78.25.08/A, submitted to WHO by
Roussel Uclaf).
IRDC (1980) Two year chronic dog feeding study, Matawan,
Michigan, International Research and Development Corporation
(Unpublished proprietary report IRDC-406-004/AI, submitted to WHO
by Roussel Uclaf).
JACKSON, G. & HARDY, C. (1986) RU 22974. Acute inhalation
toxicity in rats 1-hour exposure (Unpublished report RSL-DTM-
851457/A, submitted to WHO by Roussel Uclaf).
JACQUES, Y., ROMEY, G., CAVEY, M.T., KARTAALOVSKI, B., & LAZDUNSKI,
M. (1980) Interaction of pyrethroids with the sodium channel in
mammalian neuronal cells in culture. Biochem. Biophys. Acta, 600:
882-897.
KAGAN, YU. S., PANSHINA, T.N., & SASINOVICH, L.M. (1986)
Biochemical effects of toxic action of synthetic pyrethroids. Gig.
i Sanit., 1: 7-9.
KAUFMAN, D.D. & KAYSER, A.J. (1979a) Degradation of 14C-phenoxy-
and 14C-cyano-decamethrin in soil (Unpublished proprietary report
USDA-I-23.04.79/A2, submitted to WHO by Roussel Uclaf).
KAUFMAN, D.D. & KAYSER, A.J. (1979b) The effect of soil
temperature on the degradation of 14C-cyano-decamethrin in soil
(Unpublished proprietary report USDA-II-24.04.79/A2, submitted to
WHO by Roussel Uclaf).
KAUFMAN, D.D. & KAYSER, A.J. (1980) Degradation of 14C-cyano-,
14C-phenoxy-, and 14C-vinyl-decamethrin in flooded soil
(Unpublished proprietary report USDA-III-12.05.80/A, submitted to
WHO by Roussel Uclaf).
KAUFMAN, D.D., RUSSEL, B.A., HELLING, C.S., & KAYSER, A.J. (1981)
Movement of cypermethrin, decamethrin, permethrin and their
degradation products in soil. J. agric. food Chem., 29: 239-245.
KAVLOCK, R., CHERNOFF, N., BARON, R., LINDER, R., ROGERS, E.,
CARVER, B., DILLEY, J., & SIMMON, V. (1979) Toxicity studies with
decamethrin, a synthetic pyrethroid insecticide. J. environ.
Pathol. Toxicol., 2: 751-765.
KNAUF, W. & HORLEIN, G. (1979 The acute toxicity of decamethrine
to the Rainbow trout Salmo gairdneri, Richardson, Frankfurt,
Hoechst AG, 19 pp (Unpublished proprietary report 19/79, submitted
to WHO by Roussel Uclaf).
KNAUF, W. & SCHULZE, E.F. (1977a) Effect of decamethrine on
Cyprinus carpio, Frankfurt, Hoechst AG, 9 pp (Unpublished
proprietary report 15/77, submitted to WHO by Roussel Uclaf).
KNAUF, W. & SCHULZE, E.F. (1977b) The effect of decamethrin
6E0660 on Catfish (Ictalurus nebulosus) in a static test,
Frankfurt, Hoechst Ag, 18 pp (Unpublished proprietary report
32/79, submitted to WHO by Roussel Uclaf).
KRASNJIH, A. & PAVLOVA, L. (1985) Harmful effects of
deltamethrin. In: Levina, E., ed. Harmful substances in industry,
Moscow, Khimija Publishing House, p. 154.
KYNOCH, S.R., LLOYD, G.K., & ANDREWS, C.D. (1979) Acute
percutaneous toxicity to rats of decamethrin, Huntingdon,
Huntingdon Research Centre (Unpublished report RSL-1009 8/D,
submitted to WHO by Roussel Uclaf).
LAWRENCE, L.J. & CASIDA, J.E. (1982) Pyrethroid toxicology: Mouse
intracerebral structure-toxicity relationship. Pestic. Biochem.
Physiol., 18: 9-14.
LAWRENCE, L.J. & CASIDA, J.E. (1983) Stereospecific action of
pyrethroid insecticides on the gamma-aminobutyric acid receptor-
ionophore complex. Science, 221: 1399-1401.
LAWRENCE, L.J., GEE, K.W., & YAMAMURA, H.I. (1985) Interactions
of pyrethroid insecticides with chloride ionophore-associated
binding sites. Neurotoxicology, 6: 87-98.
LEAHEY, J.P. (1985) The pyrethroid insecticide, London, Taylor &
Francis Ltd., p. 440.
LECLERCQ, M., COTONAT, J., & FOULHOUX, P. (1986) Recherche d'un
antagoniste à l'intoxication par la deltaméthrine. J. Toxicol.
clin. exp., 6, 83-85.
LEIBOWITZ, M.D., SCHWARZ, J.R., HOLAN, G., & HILLE, B. (1987)
Electrophysiological comparison of insecticide and alkaloid
agonists of Na channels. J. gen. Physiol., 90(1): 75-93.
LEPAILLEUR, H. & CHAMBON, A. (1984) Etude de la toxicité létale
du produit technique deltaméthrine vis à vis du poisson-zebré, 5 pp
(Unpublished proprietary dated report IRCHA B.7827, submitted to
WHO by Roussel Uclaf).
LHOSTE, J., FRANCOIS, Y., & RUPAUD, Y. (1979) Ichtyotoxicité de
la decaméthrine vis à vis de Salmo trutta L. (poisson téléostéen)
en fonction de l'age et des conditions expérimentales - Congrès sur
la lutte contre les insectes en milieu tropical, Marseilles - March
13-16 1979, pp. 885-902.
L'HOTELLIER, M. & VINCENT, P. (1986) Assessment of the impact of
deltamethrin on aquatic species. In British Crop Protection
Conference - Pests and Disease: 1109-1116.
LOUVEAUX, J., MISSONNIER, J., & MESQUIDA, J. (1977) Tests de
toxicité sur abeille domestique avec le Decis CE (Unpublished
proprietary report submitted to WHO by Roussel Uclaf).
LUMMIS, S.C., CHOW, S.C., HOLAN, G., & JOHNSTON, G.A. (1987)
Gamma-aminobutyric acid receptor ionophore complexes: differential
effects of deltamethrin, dichlorodiphenyltrichloroethane, and some
novel insecticides in a rat brain membrane preparation. J.
Neurochem., 48(3): 689-94.
LUND, A.E. & NARAHASHI, T. (1983) Kinetics of sodium channel
modification as the basis for the variation in the nerve membrane
effects of pyrethroids and DDT analogs. Pestic. Biochem. Physiol.,
20: 203-216.
MACPHAIL, R.C. (1981) Behavioral effects of a synthetic
insecticide (decamethrin). Fed. Proc. Fed. Am. Soc. Exp. Biol.,
40: 678.
MEISTER, R.T., BERG, G.L., SINE, C., MEISTER, S., & POPLYK, J.
(1983) Farm chemicals handbook. Section C. Pesticide dictionary,
Willoughby, Ohio, Meister Publishing Co.
MESTRES, R., CHEVALLIER, C., ESPNOZA, C., & CORNET, R. (1978a)
Dosage des résidues de decaméthrine dans les produits végétaux.
Trav. Soc. Pharm. Montpellier, 38: 183-192.
MESTRES R., CHEVALLIER C., & ESPINOZA, C. (1978b) Analytical
method for decamethrine residue analysis, Montpellier, Faculte de
Pharmacie, 2 pp. (Proprietary report FP-78.08.07/A, dated 7/8/1978
submitted to WHO by Roussel Uclaf).
MESTRES, R., FRANCOIS, C., CAUSSE C., VIAN L., & WINNETT, G.
(1985) Survey of exposure to pesticides in greenhouses. Bull.
environ. Contam. Toxicol., 35: 750-756.
MESTRES, R., ESPINOZA, C., & CHEVALLIER, C. (1986) Effets sur les
résidus de deltaméthrine de la transformation des produits
agricoles en vue de leur consommation. Med. Nutr., XXII (3):
181-184.
MILLER, T.A. & ADAMS, M.E. (1980) Neural and behavioral
correlates of pyrethroid and DDT-type poisoning in the housefly,
Musca domestica. Pestic. Biochem. Physiol., 13: 137-147.
MIYAMOTO, J. (1976) Degradation, metabolism and toxicity of
synthetic pyrethroids. Environ. Health Perspect., 14: 15-28.
MIYAMOTO, J. (1981) The chemistry, metabolism and residue
analysis of synthetic pyrethroids. Pure appl. Chem., 53: 1967-2022.
MIYAMOTO, J. & KEARNEY, P.C. (1983) Pesticide chemistry - Human
welfare and the environment. Proceedings of the Fifth International
Congress of Pesticide Chemistry, Kyoto, Japan, 29 August - 4
September, 1982, Oxford, Pergamon Press, Vol. 1-4.
MOHSEN, Z.H. & MULLA, M.S. (1981) Toxicity of blackfly larvicidal
formulations to some aquatic insects in the laboratory. Bull.
environ. Contam. Toxicol., 26: 696-703.
MOUROT, D., DELEPINE, B., BOISSEAU, J., & GAYOT, G. (1979) High-
performance liquid chromatography of decamethrin. J. Chromatogr.,
173: 412-414.
MUIR, D.C.G., RAWN, G.P., & GRIFT, N.P., (1985) Fate of the
pyrethroid insecticide deltamethrin in small ponds: A mass balance
study. J. agric. food Chem., 33: 603-609.
MULLA, M.S., NAVVAB GOJRATI, H.A., & DARWAZEH, H.A. (1978)
Toxicity of mosquito larvicidal pyrethroids to four species of
freshwater fishes. Environ. Entomol.., 7(3): 428-430.
NETO, P.X.R., RODRIGUES, I.C.F., & FILHO, A.M (1983) Etude sur
les effets secondaires de la deltaméthrine sur l'ichtyofaune de la
region du projet "Rio Formoso" (Unpublished report submitted to WHO
by Roussel Uclaf).
NOBLE, R.M., HAMILTON, D.J., & OSBORNE, W.J. (1982) Stability of
pyrethroids on wheat in storage. Pestic. Sci., 13: 246-252.
PANSHINA T.N. & SASINOVICH, L.M. (1983) Toxicology of synthetic
pyrethroids. Khim. sel'sk. khoz., 12: 51-53.
PANSU, M., DHOUIBI, M.H., & PINTA, M. (1981) Determination of
traces of biopermethrine and decamethrine pyrethrinoids in
biological substrates by gas chromatography. Analysis, 9: 55-59.
PAPALEXIOU, Ph., BITAR, N., & STOCKIS, A. (1984) Absorption of
radiocarbon labelled deltamethrin given orally in healthy
volunteers, Charleroi, Belgium, Institut de Pharmacologie et
d'Investigations Biomédicales (Unpublished proprietary report
50/22, submitted to WHO by Roussel Uclaf).
PARKIN, P.J. & LEQUESNE, P.M. (1982) Effects of a synthetic
pyrethroid deltamethrin on excitability changes following a nerve
impulse. J. Neurol. Neurosurg. Psychiatry., 45: 337-342.
PEYRE, M., CHANTOT, J.F., GLOMOT, R., & PENASSE, L. (1980)
Detection of a mutagenic potency of decamethrin (RU 22974), in
bacterial tests (Unpublished report RU/TOX/80-2101/A, submitted to
WHO by Roussel Uclaf).
PHAM, H.C., NAVARRO-DALMASURE, C., PHAM, H.C., CLAVEL, P., VAN
HAVERBEKE, G., CHEAV, S.L. (1984) Toxicological studies of
deltamethrin. Int. J. Tissue React., 6(2): 127-33.
PICHON, Y. (1981) Pharmacological characterization of ionic
channels in unmyelinated axons. J. Physiol., 17(9): 1119-1128.
PLAPP, F.W., Jr & BULL, D.L. (1978) Toxicity and selectivity of
some insecticides to Chrysopa carnea, a predator of the tobacco
budworm. Environ. Entomol., 7: 431-434.
PLESTINA, R. (1981) An evaluation of the use of deltamethrin in
public health (Unpublished report ZAG. KOT. 81/A, submitted to WHO
by Roussel Uclaf).
PLUIJMEN, M., DREVON, C., MONTESANO, R., MALAVEILLE C.,
HAUTEFEUILLE, A., & BARTSCH, H. (1984) Lack of mutagenicity of
synthetic pyrethroids in Salmonella typhimurium strains and in V79
Chinese hamster cells. Mutat. Res., 137: 7-15.
POLAKOVA, H. & VARGOVA, M. (1983) Evaluation of the mutagenic
effects of decamethrin: cytogenetic analysis of bone marrow. Mutat.
Res., 120: 167-171.
POTTIER J., CHATELET, P., & JOUQUEY, S. (1982) Resorption
percutanée de la deltaméthrine - Etude d'un modèle animal, 10 pp.
(Unpublished proprietary report AN.34, submitted to WHO by Roussel
Uclaf).
PRASADA RAO, K.S., CHETTY, C.S., & DESAIAH, D. (1984) In vitro
effects of pyrethroids on rat brain and liver ATPase activities. J
Toxicol. environ. Health, 14(2-3): 257-65.
RAWN, G.P., MUIR, D.O.G., & GRIFT, P.G. (1985) Fate of the
pyrethroid insecticide deltamethrin in small ponds, a mass balance
study. J. agr. food Chem., 33: 603-609.
RAY, D.E. (1980) An EEG investigation of decamethrin-induced
choreathetosis in the rat. Exp. Brain Res., 38: 221-227.
RAY, D.E. (1982) Changes in brain blood flow associated with
deltamethrin-induced choreoathetosis in the rat. Exp. Brain Res.,
45: 269-276.
RAY, D.E. & CREMER, J.E. (1979) The action of decamethrin (a
synthetic pyrethroid) on the rat. Pestic. Biochem. Physiol., 10:
333-340.
RICHTER, W.R. & GOLDENTHAL, E.I. (1983) Two-year oral toxicity
and carcinogenicity study in rats. Amendment to the final report.
Matawan, Michinga, International Research and Development
Corporation (Unpublished proprietary report IRDC 406.002/A5,
submitted to WHO by Roussel Uclaf).
RICKARD, J. & BRODIE, M.E. (1985) Correlation of blood and brain
levels of the neurotoxic pyrethroid deltamethrin with the onset of
symptoms in rats. Pestic. Biochem. Physiol., 23: 143-156.
RICOU (1978) Assessment of side effects of S 276-B (Decis EC
25 g/l) towards grey slugs (Agriolimax spp.) (Unpublished
proprietary report INRA-LIM.78.10/A, submitted to WHO by Roussel
Uclaf).
ROSE, G.P. & DEWAR, A.J. (1983) Intoxication with four synthetic
pyrethroids fail to show any correlation between neuromuscular
dysfunction and neurobiochemical abnormalities in rats. Arch.
Toxicol., 53: 297-316.
ROSE, D.B., ROBERTS, N.L., CAMERON, M.McD., PRENTICE, D.E., COOKE,
L., & GIBSON, W.A. (1978) RU 22974. (Decamethrine) LD50
determination and assessment of neurotoxicity in the domestic hen,
Huntingdon, Huntingdon Research Centre (Unpublished report RSL 293-
NT/7830/A, submitted to WHO by Roussel Uclaf).
ROUSSELIN, X. (1983) Toxicité des dérivés du pyrèthre, Paris,
Faculté de Médecine Lariboisiere-Saint-Louis (Thèse de l'Université
Paris VII).
RUIGT, G.S.F. & VAN DEN BERCKEN, J. (1986) Action of pyrethroids
on a nerve-muscle preparation of the clawed frog, Xenopus laevis.
Pestic. Biochem. Physiol., 25: 176-187.
RUZO, L.O. & CASIDA, J.E. (1979) Degradation of decamethrin on
cotton plants. J. agric. food Chem., 27: 572-575.
RUZO, L.O., HOLMSTEAD, R.L., & CASIDA, J.E. (1976) Solution
photochemistry of the potent pyrethroid insecticide alpha-cyano-3-
phenoxy-benzyl cis-2,2-dimethyl-3-(2,2-dibromovinyl)
cyclopropanecarboxylate. Tetrahedron Lett., 3045-3048.
RUZO, L.O., HOLMSTEAD, R.L., & CASIDA, J.E. (1977) Pyrethroid
photochemistry: decamethrin. J. agric. food Chem., 25: 1385-1394.
RUZO, L.O., UNAI, T., & CASIDA, J.E. (1978) Decamethrin metabolism
in rats. J. agric. food Chem., 26: 918-925.
RUZO, L.O., ENGEL, J.L., & CASIDA, J.E. (1979) Decamethrin
metabolites from oxidative, hydrolytic and conjugative reactions in
mice. J. agric. food Chem., 27: 725-731.
SANTOSA, K. (1983) Toxicity of Decis 2.5 EC (Decamethrin) to
fish, Jakarta, Department of Agriculture Research and Development
Inland Fisheries Institute, 4 pp. (Unpublished proprietary data
submitted to WHO by Roussel Uclaf).
SANTOSA, K. & HADI, H. (1980) Trial results pesticide lethal
toxicity to fish, Department of Agriculture Research and
Development Inland Fisheries Institute, Jakarta, Indonesia -
Report no. 174/UI/80 dated 5 December 1980, 3 pp. (Unpublished
proprietary data submitted to WHO by Roussel Uclaf).
SMIES, M., EVERS, R.H.J., REIJNENBURG, P.H.M., & KOEMAN, J.H.
(1980) Environmental aspects of field trials with pyrethroids to
eradicate tsetse fly in Nigeria. Ecotoxicol. environ. Saf., 4:
114-128.
SOBELS, F.H., TATES, A.D., & VANNIER, B. (1978) Cytogenetic study
with RU 22974. Detection of a mutagenic potency in mammalian
cells, Leiden, The Netherlands, State University of Leiden
(Unpublished report ULN-782211/A, submitted to WHO by Roussel
Uclaf).
SODERLUND, D.M. & CASIDA, J.E. (1977) Effects of pyrethroid
structure on rates of hydrolysis and oxidation by mouse liver
microsomal enzymes. Pestic. Biochem. Physiol., 7: 391-401.
SOUYRI, F. (1985) Autoradiographic studies of (3H)-deltamethrin
binding with nerve tissue cultures. Pestic. Sci., 6(6): 701-703.
STAATZ, C.G., BLOOM, A.S., & LECH, J.J. (1982) Effects of
pyrethroids on [3H]-kainic acid binding to mouse forebrain
membranes. Toxicol. appl. Pharmacol., 64: 566-569.
STAATZ-BENSON, C.G. & HOSKO, M.J. (1986) Interaction of
pyrethroids with mammalian spinal neurons. Pestic. Biochem.
Physiol., 25: 19-30.
STEIN, E.A., WASHBURN, M., WALCZAK, C., & BLOOM, A.S. (1987)
Effects of pyrethroid insecticides on operant responding maintained
by food. Neurotoxicol. Teratol., 9(1): 27-31.
STEVENSON, J.H., NEEDHAM, P.H., & WALKER, J. (1978) Poisoning of
honey bee by pesticides: investigations of the changing pattern in
Britain over 20 years. Rothamsted Exp. Stn Rep., Part 2: 55-72.
TAKAHASHI, M. & LEQUESNE, P. ((1982) The effects of the
pyrethroids deltamethrin and cismethrin on nerve excitability in
rats. J. Neurol. Neurosurg. Psychiatry, 45: 1005-1011.
TAKKEN, W., BALK, F., JANSEN, R.C., & KOEMAN, J.H. (1978) The
experimental application of insecticides from a helicopter for the
control of riverine populations of Glossina tachinoides in West
Africa. VI. Observations on side effects. P.A.N.S., 24: 455-466.
THIEBAULT, J.J., BOST, J., & FOULHOUX, P. (1985) Intoxication
expérimentale par la deltaméthrine chez le chien et son traitement.
Toxicol. vét. Collect. Méd. lég. Toxicol. méd., 131, 47-62.
THIEBAULT, J.J., BOST, J., FOULHOUX, P., DEVAUX, P., & TILLIER, C.
(in press) Intravenous deltamethrin intoxication of dogs.
Therapeutic attempts. Veterinary and Human Toxicology.
THIER, W. & SCHMIDT, D. (1976) Laboratory leaching studies of
Decis EC 25 in three different German soils (Unpublished
proprietary report RL-77.25.05/A, submitted to WHO by Roussel
Uclaf).
THIER, W. & SCHMIDT, D. (1977) The behaviour of the pesticide in
the soil: Decamethrin, (Unpublished proprietary report A09232,
submitted to WHO by Roussel Uclaf) (in German).
TONG YING (1988) Clinical manifestations of 211 cases intoxicated
by deltamethrin and fenvalerate (Unpublished report submitted to
WHO by Roussel Uclaf).
TOOBY, T.E., THOMSON, A.N., RYCROFT, R.J., BLACK, I.A., & HEWSON,
R.T. (1981) A pond study to investigate the effects on fish and
aquatic invertebrates of deltamethrin applied directly onto water,
Fisheries Laboratory, Ministry of Agriculture, Fisheries and Food
(Unpublished report AEP-81.30.09A, submitted to WHO by Roussel
Uclaf).
TRIVEDI, K. (1981) Spray worker exposure and health survey during
decamethrin (decis) spray in the field, Sevagram, India, Mahatma
Gandhi Institute of Medical Sciences (Unpublished information
submitted to WHO by Roussel Uclaf).
TU, C.M. (1980) Influence of five pyrethroid insecticides on
microbial populations and activities in soil. Microb. Ecol., 5:
321-327.
VAN DEN BERCKEN, J. (l977) The action of allethrin on the
peripheral nervous system of the frog. Pestic. Sci., 8: 692-699.
VAN DEN BERCKEN, J. & VIJVERBERG, H.P.M. (1980) Voltage clamp
studies on the effects of allethrin and DDT on the sodium channels
in frog myelinated nerve membrane. In: Insect neurobiology and
pesticide action (Neutox 79), London, Society of Chemical Industry,
Vol. 2, pp. 79-85.
VAN DEN BERCKEN, J., AKKERMANS, L.M.A., & VAN DER ZALM, J.M.
(1973) DDT-like action of allethrin in the sensory nervous system
of Xenopus laevis. Eur. J. Pharmacol., 21: 95-106.
VAN DEN BERCKEN, J., KROESE, A.B.A., & AKKERMANS, L.M.A. (1979)
Effect of insecticides on the sensory nervous system. In:
Narahashi, T., ed. Neurotoxicology of insecticides and pheromones,
New York, London, Plenum Press, pp. 183-210.
VANNIER, B. & FOURNEX, R. (1983) Deltamethrin detection of a
mutagenic potency/micronucleus test in the mouse (Proprietary
unpublished report RU-83607/A, submitted to WHO by Roussel Uclaf).
VANNIER, B. & GLOMOT, R. (1977) RU 22974. Mutagenic study.
Dominant lethal assay in the male mouse (Unpublished report TOX
76533/DB9/A, submitted to WHO by Roussel Uclaf).
VANNIER, B. & GLOMOT, R. (1982) RU 22974 complementary
teratological study in the mouse (Unpublished report RU-82506-12/A,
submitted to WHO by Roussel Uclaf).
VARANKA, I. (1987) Effect of mosquito killer insecticides on
freshwater mussels. Comp. Biochem. Physiol. C. Comp. Pharmacol.,
86: 157-162.
VAYSSE, M., GIUDICELLI, J.C., DEVAUX, P., & L'HOTELLIER, M. (1984)
Decis, in Zweig analytical methods for pesticides and plant growth
regulators. Vol XIII, 3: 53-68, New York, Academic Press.
VERSCHOYLE, R.D. & ALDRIDGE, W.N. (1980) Structure-activity
relationship of some pyrethroids in rats. Arch. Toxicol., 45:
325-329.
VICKERMAN, G.P., COOMBES D.S., TURNER, G., MEAD-BRIGGS, M.A., &
EDWARDS, J. (1987a) The effects of pirimicarb, dimethoate and
deltamethrin on carabidae and staphylinidae in winter wheat. Meded.
Fac. Landbouwwet. Rijksuniv. Gent, 52(2a):
VICKERMAN, G.P., COOMBES, D.S., TURNER, G., MEAD-BRIGGS, M.A., &
EDWARDS, J. (1987b) The effects of pirimicarb, dimethoate and
deltamethrin on non-target arthropods in winter wheat.
International Conference on Pests in Agriculture, Paris, 1-3
December 1987, Annales ANPP, pp. 67-74.
VIJVERBERG, H.P.M., VAN DEN BECKEN, J. (1982) Action of
pyrethroid insecticides on the vertebrate nervous system.
Neuropathol. appl. Neurobiol., 8: 421-440.
VIJVERBERG, H.P.M. & VAN DEN BERCKEN, J. (1979) Frequency
dependent effects of the pyrethroid insecticide decamethrin in frog
myelinated nerve fibres. Eur. J. Pharmacol., 58: 501-504.
VIJVERBERG, H.P.M., RUIGT, G.S.F., & VAN DEN BERCKEN, J. (1982a)
Structure-related effects of pyrethroid insecticides on the
lateral-line sense organ and on peripheral nerves of the clawed
frog, Xenopus laevis. Pestic. Biochem. Physiol., 18: 315-324.
VIJVERBERG, H.P.M., VAN DER ZALM, J.M., & VAN DEN BERCKEN, J.
(1982b) Similar mode of action of pyrethroids and DDT on sodium
channel gating in myelinated nerves. Nature, (Lond.) 295: 601-603.
VIJVERBERG, H.P.M., VAN DER ZALM, J.M., VAN KLEEF, R.G.D.M., & VAN
DEN BERCKEN, J. (1983) Temperature and structure-dependent
interaction of pyrethroids with the sodium channels in frog node of
Ranvier. Biochim. Biophys. Acta, 728: 73-82.
WALTERSDORFER & SCHULZE E.F. (1976a) Decis 2.5. Effect on
Lebistes reticulatus, Francfort, Hoechst Ag, 8 pp. (Unpublished
proprietary report 42/76, submitted to WHO by Roussel Uclaf).
WALTERSDORFER & SCHULZE E.F. (1976b) Decis 2.5. Effect on Idus
melanotus, Frankfurt, Hoechst AG, 5 pp (Unpublished proprietary
report 33/76, submitted to WHO by Roussel Uclaf).
WALTERSDORFER & SCHULZE E.F. (1976c) Decis 2.5. Effect on Salmo
gairdneri (rainbow trout), Frankfurt, Hoechst AG, 7 pp (Unpublished
proprietary report 35/76, submitted to WHO by Roussel Uclaf).
WALTERSDORFER & SCHULZE E.F. (1976d) Decamethrine effect on
Lepomis gibbosus (Bluegill sunfish), Frankfurt, Hoechst AG, 6 pp.
(Unpublished proprietary report 16/77, submitted to WHO by Roussel
Uclaf).
WANG, S., ZHENG, Q., YU, L., XU, B., & LIX., Y. (1988) Health
survey among farmers exposed to deltamethrin in the cotton fields.
Ecotoxicol. environ. Saf., 15: 1-6.
WELLCOME FOUNDATION (1979) Residues in cow's milk resulting from
intrarumenal, and later, dermal administration of 14C-labelled
decamethrin (Unpublished report submitted to WHO by Roussel Uclaf).
WHO (1979) Safe use of pesticides. Third Report of the WHO
Expert Committee on Vector Biology and Control, Geneva, World
Health Organization, pp. 18-23. (WHO Technical Report Series,
No. 634).
WHO (1988) The WHO recommended classification of pesticides by
hazard and guidelines to classification 1988-1989, Geneva, World
Health Organization (Unpublished document WHO/VBC/88.953).
WHO (1989) Health and Safety Guide No. 30: Deltamethrin, Geneva,
World Health Organization.
WHO (1984) Data sheet on pesticides, No. 50: Deltamethrin, Geneva,
World Health Organization, 9 pp (Unpublished document
VBC/DS/84.50).
WOOD MACKENZIE (1980) Agrochem. Monit., 9: 12.
WOOD MACKENZIE (1981) Agrochem. Monit., 15: 12.
WOOD MACKENZIE (1982) Agrochem. Monit., 21: 13.
WOOD MACKENZIE (1983) Agrochem. Monit., 27: 3-12.
WORTHING, C.R. & WALKER, S.B. (1983) Pesticide manual, 7th ed.,
Croydon, British Crop Protection Council, p. 161.
WOUTERS, W. & VAN DEN BERCKEN, J. (1978) Action of pyrethroids.
Gen. Pharmacol., 9: 387-398.
WRENN, J.M., RODWELL, D.E., GOLDENTHAL, E.I., SPIDER, E.J.C., &
RAJASEKARAN, D. (1980) Three-generation reproduction study in
rats, Matawan, Michigan, International Research and Development
Corporation (Unpublished report IDC-406 003/A4. submitted to WHO by
Roussel Uclaf).
ZHANG, L.Z., KHAN, S.U., AKHTAR, M.H., & IVARSON, K.C. (1984)
Persistence, degradation, and distribution of Deltamethrin in an
organic soil under laboratory conditions, J. agric. food Chem., 32:
1207-1211
ZITKO, V., MCLEESE, D.W., METCALFE, C.D., & CARSON, W.G. (1979)
Toxicity of permethrin, decamethrin, and related pyrethroids to
salmon and lobster. Bull. environ. Contam. Toxicol., 21: 338-343.
APPENDIX I
On the basis of electrophysiological studies with peripheral
nerve preparations of frogs (Xenopus laevis; Rana temporaria; and
Rana esculenta), it is possible to distinguish between 2 classes of
pyrethroid insecticides: (Type I and Type II). A similar
distinction between these 2 classes of pyrethroids has been made on
the basis of the symptoms of toxicity in mammals and insects (Van
den Bercken et al., 1979; WHO, 1979; Verschoyle & Aldridge, 1980;
Glickman & Casida, 1982; Lawrence & Casida, 1982). The same
distinction was found in studies on cockroaches (Gammon et al.,
1981).
Based on the binding assay on the gamma-aminobutyric acid
(GABA) receptor-ionophore complex, synthetic pyrethroids can also
be classified into two types: the alpha-cyano-3-phenoxybenzyl
pyrethroids and the non-cyano pyrethroids (Gammon et al., 1982;
Gammon & Casida, 1983; Lawrence & Casida, 1983; Lawrence et al.,
1985).
Pyrethroids that do not contain an alpha-cyano group
(allethrin, d-phenothrin, permethrin, tetramethrin, cismethrin, and
bioresmethrin) (Type I: T-syndrome)
The pyrethroids that do not contain an alpha-cyano group give
rise to pronounced repetitive activity in sense organs and in
sensory nerve fibres (Van den Bercken et al., 1973). At room
temperature, this repetitive activity usually consists of trains of
3 - 10 impulses and occasionally up to 25 impulses. Train duration
is between 10 and 5 milliseconds.
These compounds also induce pronounced repetitive firing of the
presynaptic motor nerve terminal in the neuromuscular junction (Van
den Bercken, 1977). There was no significant effect of the
insecticide on neurotransmitter release or on the sensitivity of
the subsynaptic membrane, nor on the muscle fibre membrane.
Presynaptic repetitive firing was also observed in the sympathetic
ganglion treated with these pyrethroids.
In the lateral-line sense organ and in the motor nerve
terminal, but not in the cutaneous touch receptor or in sensory
nerve fibres, the pyrethroid-induced repetitive activity increases
dramatically as the temperature is lowered, and a decrease of 5 °C
in temperature may cause a more than 3-fold increase in the number
of repetitive impulses per train. This effect is easily reversed
by raising the temperature. The origin of this "negative
temperature coefficient" is not clear (Vijverberg et al., 1983).
Synthetic pyrethroids act directly on the axon through
interference with the sodium channel gating mechanism that
underlies the generation and conduction of each nerve impulse. The
transitional state of the sodium channel is controlled by 2
separately acting gating mechanisms, referred to as the activation
gate and the inactivation gate. Since pyrethroids only appear to
affect the sodium current during depolarization, the rapid opening
of the activation gate and the slow closing of the inactivation
gate proceed normally. However, once the sodium channel is open,
the activation gate is restrained in the open position by the
pyrethroid molecule. While all pyrethroids have essentially the
same basic mechanism of action, however, the rate of relaxation
differs substantially for the various pyrethroids (Flannigan &
Tucker, 1985).
In the isolated node of Ranvier, allethrin causes prolongation
of the transient increase in sodium permeability of the nerve
membrane during excitation (Van den Bercken & Vijverberg, 1980).
Evidence so far available indicates that allethrin selectively
slows down the closing of the activation gate of a fraction of the
sodium channels that open during depolarization of the membrane.
The time constant of closing of the activation gate in the
allethrin-affected channels is about 100 milliseconds compared with
less than 100 microseconds in the normal sodium channel, i.e., it
is slowed down by a factor of more than 100. This results in a
marked prolongation of the sodium current across the nerve membrane
during excitation, and this prolonged sodium current is directly
responsible for the repetitive activity induced by allethrin
(Vijverberg et al., 1983).
The effects of cismethrin on synaptic transmission in the frog
neuromuscular junction, as reported by Evans (1976), are almost
identical to those of allethrin, i.e., presynaptic repetitive
firing, and no significant effects on transmitter release or on the
subsynaptic membrane.
Interestingly, the action of these pyrethroids closely
resembles that of the insecticide DDT in the peripheral nervous
system of the frog. DDT also causes pronounced repetitive activity
in sense organs, in sensory nerve fibres, and in motor nerve
terminals, due to a prolongation of the transient increase in
sodium permeability of the nerve membrane during excitation.
Recently was demonstrated that allethrin and DDT have essentially
the same effect on sodium channels in frog myelinated nerve
membrane. Both compounds slow down the rate of closing of a
fraction of the sodium channels that open on depolarization of the
membrane (Van den Bercken et al., 1973, 1979; Vijverberg et al.,
1982b).
In the electrophysiological experiments using giant axons of
crayfish, the type I pyrethroids and DDT analogues retain sodium
channels in a modified open state only intermittantly, cause large
depolarizing afterpotentials, and evoke repetitive firing with
minimal effect on the resting potential (Lund & Narahashi, 1983).
These results strongly suggest that permethrin and cismethrin,
like allethrin, primarily affect the sodium channels in the nerve
membrane and cause a prolongation of the transient increase in
sodium permeability of the membrane during excitation.
The effects of pyrethroids on end-plate and muscle action
potentials were studied in the pectoralis nerve-muscle preparation
of the clawed frog (Xenopus laevis). Type I pyrethroids
(allethrin, cismethrin, bioresmethrin, and 1R, cis-phenothrin)
caused moderate presynaptic repetitive activity, resulting in the
occurrence of multiple end-plate potentials (Ruigt & Van den
Bercken, 1986).
Pyrethroids with an alpha-cyano group on the 3-phenoxybenzyl
alcohol (deltamethrin, cypermethrin, fenvalerate, and fenpropanate)
(Type II: CS-syndrome)
The pyrethroids with an alpha-cyano group cause an intense
repetitive activity in the lateral line organ in the form of long-
lasting trains of impulses (Vijverberg et al., 1982a). Such a
train may last for up to 1 min and contains thousands of impulses.
The duration of the trains and the number of impulses per train
increase markedly on lowering the temperature. Cypermethrin does
not cause repetitive activity in myelinated nerve fibres. Instead,
this pyrethroid causes a frequency-dependent depression of the
nervous impulse, brought about by a progressive depolarization of
the nerve membrane as a result of the summation of depolarizing
after-potentials during train stimulation (Vijverberg & Van den
Bercken, 1979; Vijverberg et al., 1983).
In the isolated node of Ranvier, cypermethrin, like allethrin,
specifically affects the sodium channels of the nerve membrane and
causes a long-lasting prolongation of the transient increase in
sodium permeability during excitation, presumably by slowing down
the closing of the activation gate of the sodium channel
(Vijverberg & Van den Bercken, 1979; Vijverberg et al., 1983). The
time constant of closing of the activation gate in the
cypermethrin-affected channels is prolonged to more than 100
milliseconds. Apparently, the amplitude of the prolonged sodium
current after cypermethrin is too small to induce repetitive
activity in nerve fibres, but is sufficient to cause the long-
lasting repetitive firing in the lateral-line sense organ.
These results suggest that alpha-cyano pyrethroids primarily
affect the sodium channels in the nerve membrane and cause a long-
lasting prolongation of the transient increase in sodium
permeability of the membrane during excitation.
In the electrophysiological experiments using giant axons of
cray-fish, the Type II pyrethroids retain sodium channels in a
modified continuous open state persistently, depolarize the
membrane, and block the action potential without causing repetitive
firing (Lund & Narahashi, 1983).
Diazepam, which facilitates GABA reaction, delayed the onset of
action of deltamethrin and fenvalerate, but not permethrin and
allethrin, in both the mouse and cockroach. Possible mechanisms of
the Type II pyrethroid syndrome include action at the GABA receptor
complex or a closely linked class of neuroreceptor (Gammon et al.,
1982).
The Type II syndrome of intracerebrally administered pyrethroids
closely approximates that of the convulsant picrotoxin (PTX).
Deltamethrin inhibits the binding of [3H]-dihydropicrotoxin to rat
brain synaptic membranes, whereas the non-toxic R epimer of
deltamethrin is inactive. These findings suggest a possible
relation between the Type II pyrethroid action and the GABA receptor
complex. The stereospecific correlation between the toxicity of
Type II pyrethroids and their potency to inhibit the [35S]-TBPS
binding was established using a radioligand,
[35S]- t-butyl-bicyclophosphorothionate [35S]-TBPS. Studies
with 37 pyrethroids revealed an absolute correlation, without any
false positive or negative, between mouse intracerebral toxicity and
in vitro inhibition: all toxic cyano compounds including
deltamethrin, 1R, cis-cypermethrin, 1R, trans-cypermethrin, and
[2S, alpha S]-fenvalerate were inhibitors, but their non-toxic
stereoisomers were not; non-cyano pyrethroids were much less potent
or were inactive (Lawrence & Casida, 1983).
In the [35S]-TBPS and [3H]-Ro 5-4864 (a convulsant benzo-
diazepine radioligand) binding assay, the inhibitory potencies of
pyrethroids were closely related to their mammalian toxicities.
The most toxic pyrethroids of Type II were the most potent
inhibitors of [3H]-Ro 5-4864 specific binding to rat brain
membranes. The [3H]-dihydropicrotoxin and [35S]-TBPS binding
studies with pyrethroids strongly indicated that Type II effects of
pyrethroids are mediated, at least in part, through an interaction
with a GABA-regulated chloride ionophore-associated binding site.
Moreover, studies with [3H]-Ro 5-4864 support this hypothesis and,
in addition, indicate that the pyrethroid-binding site may be very
closely related to the convulsant benzodiazepine site of action
(Lawrence et al., 1985).
The Type II pyrethroids (deltamethrin, 1R, cis-cypermethrin and
[2S, alphaS]-fenvalerate) increased the input resistance of
crayfish claw opener muscle fibres bathed in GABA. In contrast,
two non-insecticidal stereoisomers and Type I pyrethroids
(permethrin, resmethrin, allethrin) were inactive. Therefore,
cyanophenoxybenzyl pyrethroids appear to act on the GABA receptor-
ionophore complex (Gammon & Casida, 1983).
The effects of pyrethroids on end-plate and muscle action
potentials were studied in the pectoralis nerve-muscle preparation
of the clawed frog (Xenopus laevis). Type II pyrethroids
(cypermethrin and deltamethrin) induced trains of repetitive muscle
action potentials without presynaptic repetitive activity.
However, an intermediate group of pyrethroids (1R-permethrin,
cyphenothrin, and fenvalerate) caused both types of effect. Thus,
in muscle or nerve membrane the pyrethroid induced repetitive
activities due to a prolongation of the sodium current. But no
clear distinction was observed between non-cyano and alpha-cyano
pyrethroids (Ruigt & Van den Bercken, 1986).
Appraisal
In summary, the results strongly suggest that the primary
target site of pyrethroid insecticides in the vertebrate nervous
system is the sodium channel in the nerve membrane. Pyrethroids
without an alpha-cyano group (allethrin, d-phenothrin, permethrin,
and cismethrin) cause a moderate prolongation of the transient
increase in sodium permeability of the nerve membrane during
excitation. This results in relatively short trains of repetitive
nerve impulses in sense organs, sensory (afferent) nerve fibres,
and, in effect, nerve terminals. On the other hand, the alpha-
cyano pyrethroids cause a long-lasting prolongation of the
transient increase in sodium permeability of the nerve membrane
during excitation. This results in long-lasting trains of
repetitive impulses in sense organs and a frequency-dependent
depression of the nerve impulse in nerve fibres. The difference in
effects between permethrin and cypermethrin, which have identical
molecular structures except for the presence of an alpha-cyano
group on the phenoxybenzyl alcohol, indicates that it is this
alpha-cyano group that is responsible for the long-lasting
prolongation of the sodium permeability.
Since the mechanisms responsible for nerve impulse generation
and conduction are basically the same throughout the entire nervous
system, pyrethroids may also induce repetitive activity in various
parts of the brain. The difference in symptoms of poisoning by
alpha-cyano pyrethroids, compared with the classical pyrethroids,
is not necessarily due to an exclusive central site of action.
It may be related to the long-lasting repetitive activity in
sense organs and possibly in other parts of the nervous system,
which, in a more advance state of poisoning, may be accompanied by
a frequency-dependent depression of the nervous impulse.
Pyrethroids also cause pronounced repetitive activity and a
prolongation of the transient increase in sodium permeability of
the nerve membrane in insects and other invertebrates. Available
information indicates that the sodium channel in the nerve membrane
is also the most important target site of pyrethroids in the
invertebrate nervous system (Wouters & Van den Bercken, 1978; WHO,
1979).
Because of the universal character of the processes underlying
nerve excitability, the action of pyrethroids should not be
considered restricted to particular animal species, or to a certain
region of the nervous system. Although it has been established
that sense organs and nerve endings are the most vulnerable to the
action of pyrethroids, the ultimate lesion that causes death will
depend on the animal species, environmental conditions, and on the
chemical structure and physical characteristics of the pyrethroid
molecule (Vijverberg & Van den Bercken, 1982).
1. RESUME ET EVALUATION, CONCLUSIONS ET RECOMMANDATIONS
1.1 Résumé et évaluation
1.1.1 Identité, propriétés physiques et chimiques, méthodes
d'analyse
La deltaméthrine a été synthétisée en 1974 et commercialisée
pour la première fois en 1977. Sur le plan chimique, c'est un
ester de l'analogue dibromé de l'acide chrysanthémique (acide
diméthyl-2,2(dimbromo-2,2 vinyl)-3 cyclopropanecarboxylique)
(Br2CA) et de l'alcol alpha-cyano-phénoxy-3 benzylique; plus
précisément, c'est l'isomère [1R, cis;alpha S] parmi les huit
stéréoisomères que compte cet ester.
La deltaméthrine de qualité technique se présente sous la forme
d'une poudre blanche inodore dont le point de fusion est de 98 -
101 °C et qui contient plus de 98% de deltaméthrine. Sa tension de
vapeur est de 2,0 x 10-6 Pa à 25 °C; autrement dit, elle n'est
pratiquement pas volatile. Insoluble dans l'eau, elle est en
revanche soluble dans des solvants organiques tels que l'acétone, la
cyclohexanone et le xylène. Elle est stable à la lumière, à la
chaleur et à l'air, mais instable en milieu alcalin.
Pour doser les résidus et analyser des échantillons prélevés
dans l'environnement, on procède à une extraction par solvant au
moyen d'un mélange n-hexane/acétone, à un partage entre le
n-hexane, l'acétone et l'eau, suivi d'une purification par
chromatographie sur colonne de gel de silice, le dosage final
s'effectuant par chromatographie en phase gazeuse avec détection
par capture d'électrons. La concentration minimale décelable par
cette méthode est de 0,01 mg/kg, voire moins. L'analyse des
produits s'effectue par chromatographie en phase liquide à haute
performance avec détecteur UV.
1.1.2 Production et usage
En 1987, la consommation mondiale de deltaméthrine était
d'environ 250 tonnes. On l'utilise essentiellement pour traiter le
coton (45% de la consommation) ainsi que pour les cultures telles
que le café, le maïs, les céréales, les fruits, les légumes, le
houblon et les produits stockés. On l'utilise aussi pour l'hygiène
des animaux, la lutte contre les vecteurs ainsi qu'à des fins de
santé publique. On l'applique soit seule, soit associée à d'autres
pesticides en formulations telles que concentrés émulsionnables,
concentrés pour pulvérisation à très bas volume, poudres
mouillables, concentrés pour suspension, poudres pour poudrage, etc.
1.1.3 Exposition humaine
L'exposition de la population dans son ensemble provient
principalement de la présence de résidus alimentaires mais peut
également résulter de son utilisation en santé publique. Sur les
récoltes correctement traitées, le taux de résidus est généralement
très faible sauf lorsque le traitement a eu lieu après la récolte.
La FAO et l'OMS ont passé en revue de nombreuses données sur ces
questions.
L'exposition de la population dans son ensemble est vrai-
semblablement très faible, mais on manque de données effectives
découlant d'études sur la ration alimentaire totale.
1.1.4 Exposition et destinée dans l'environnement
En exposant à la lumière solaire une fine pellicule de
deltaméthrine [1R, cis; alpha S] marquée au C14 au niveau de la
fonction acide, de la fonction alcool ou du groupe cyano, pendant 4
à 8 heures, on a constaté que 70 % du produit était transformé en
isomères [1R, trans alpha S] et [1S, trans alpha S] par
isomérisation cis/trans, accompagnés des produits de clivage de
l'ester, en particulier l'acide diméthyl-2,2(dibromovinyl-2,2)-3
cyclo-propane-carboxylique et l'alcool alpha-cyano-phénoxy-3
benzylique.
La deltaméthrine présente sur des plants de coton en serre se
dégrade avec une demi-vie initiale de 1,1 semaine, le taux de
dégradation étant de 90 % au bout de 4,5 semaines.
Les principaux métabolites consistent en Br2CA libre et
conjugué, en trans-hydroxyméthyl-Br2CA et en acide (hydroxy-4
phénoxy)-3 benzoïque résultant du clivage de l'ester, d'une
oxydation et d'une conjugaison.
Après avoir fait incuber de deltaméthrine dans du sable et du
terreau à 28 °C dans les conditions du laboratoire, on en a
récupéré respectivement 52 % et 74 %, huit semaines après ce
traitement.
La deltaméthrine ne se déplace pas dans l'environnement du fait
qu'elle est fortement adsorbée sur les particules, qu'elle est
insoluble dans l'eau et qu'elle est appliquée à très faibles doses.
On ne dispose pas de données sur les concentrations effectives
dans l'environnement, mais compte tenu des modalités actuelles
d'utilisation et pour peu que l'insecticide soit utilisé
normalement, l'exposition environnementale devrait être très
faible. La deltaméthrine se dégrade rapidement en produits moins
toxiques.
1.1.5 Absorption, métabolisme et excrétion
Administrée par voie orale, la deltraméthrine est facilement
absorbée; l'absorption est moindre par la voie percutanée encore
que sa vitesse dépende fortement du véhicule ou du solvant utilisé.
Une fois absorbée, la deltaméthrine est rapidement métabolisée et
excrétée.
On a administré à des rats par voie orale, à raison de 0,64 à
1,60 mg/kg, de la deltaméthrine marquée au carbone-14 au niveau des
fonctions acide, alcool et nitrile; le radiocarbone provenant de
la fraction acide et de la fraction alcoolique a été complètement
éliminé en l'espace de 2 à 4 jours. Les taux de résidus
tissulaires étaient généralement faibles sauf dans les graisses où
ils étaient un peu plus élevés. Cependant, le reste nitrile a été
excrété plus lentement, le taux global de récupération étant de
79 % en huit jours. Les principales réactions métaboliques étaient
l'oxydation (au niveau du méthyl- trans, du cycle cyclopropane et
des positions 2', 4' et 5' du reste alcoolique), le clivage de
l'ester et la conversion du nitrile en thiocyanate. Les acides
carboxyliques et les phénols résultants étaient conjugués à l'acide
sulfurique, à la glycine et à l'acide glucuronique.
Après avoir administré par voie orale à des souris de la
deltaméthrine marquée au 14C au niveau des fonctions acide, alcool
et nitrile à raison de 1,7 et 4,4 mg/kg, on a constaté que
l'excrétion du radiocarbone s'effectuait rapidement sauf dans le
cas du reste nitrile. Les principales réactions métaboliques
étaient en général analogues chez les souris et chez les rats.
Chez les vaches et la volaille, les voies de dégradation sont
tout à fait analogues à celles des rongeurs.
1.1.6 Effets sur les êtres vivants dans leur milieu naturel
La deltaméthrine est extrêmement toxique pour les poissons, la
CL50 à 96 heures allant de 0,4 à 2,0 µg/litre. Elle est également
très toxique pour les invertébrés aquatiques; pour la daphnie, la
CL50 à 48 heures est de 5 µg/litre. Toutefois des études
approfondies sur des étangs expérimentaux ainsi que les résultats
de l'utilisation en plein champ ont montré que cette forte toxicité
potentielle était inopérante. On a observé sur le terrain une
certaine mortalité parmi les invertébrés aquatiques mais les
populations se reconstituent généralement assez vite.
pour les oiseaux, la toxicité de la deltaméthrine est très
faible, les valeurs de la DL50 pour une dose unique par voie orale
étant supérieure à 1000 mg/kg. Au laboratoire, la deltaméthrine
est très toxique pour les abeilles, avec une DL50 de contact de
0,051 µg/abeille. Les essais en situation réelle et l'expérience
acquise dans l'utilisation effective du produit ont montré que les
formulations de deltaméthrine exerçaient une action répulsive, ce
qui signifie qu'en pratique le danger pour les abeilles est très
réduit.
1.1.7 Effets sur les animaux d'expérience et systemes d'epreuve
in vitro
Dans un véhicule non aqueux, la deltaméthrine présente une
toxicité aiguë par voie orale forte à modérée avec des DL50 de 19 à
34mg/kg chez la souris et de 39 à 139 mg/kg chez le rat.
Toutefois, en suspension dans l'eau, la toxicité est bien moidre,
les valeurs de la DL50 dépassant 5000 mg/kg chez le rat. La
deltaméthrine est un pyréthroide du Type II. Les signes cliniques
d'intoxication consistent en tremblements, salivation et
convulsions. L'intoxication est rapide et chez les survivants, les
signes disparaissent en quelques jours. L'électro-encéphalogramme
présente des décharges pointes-ondes généralisées qui précèdent
la choréo-athétose.
Une seule application de deltaméthrine technique n'a pas
produit d'effets irritants sur la peau intacte ou abrasée du lapin.
Toutefois elle a produit de effets irritants passagers au niveau de
l'oeil avec ou sans rinçage. La deltaméthrine n'a pas d'effet
sensibilisateur cutané chez le cobaye.
En administrant à des rats par gavage de la deltaméthrine à des
doses quotidiennes allant jusqu'à 10 mg/kg de poids corporel
pendant 13 semaines, on a provoqué chez ces animaux une
hyperexcitabilité qui se manifestait au bout de six semaines chez
les mâles recevant la dose la plus forte. Aux doses de 2,5 et
10 mg/kg, le gain de poids a été plus faible chez les mâles.
Chez des chiens beagle qui avaient reçu par voie orale de la
deltaméthrine en doses quotidiennes allant jusqu'à 10 mg/kg de
poids corporel pendant 13 semaines, on a observé divers symptômes
liés à cette substance, tels que des vomissements, des
tremblements, de la salivation et un affaiblissement du réflexe
pharyngé, du réflexe rotulien et du réflexe de flexion. Lors d'une
étude d'alimentation de deux ans sur des chiens, on a constaté que
la dose sans effet observable se situait à 1 mg/kg de poids
corporel par jour (dose la plus forte expérimentée).
L'administration de deltraméthrine à des souris à des doses
atteingnant 100 mg/kg de nourriture pendant 24 mois n'a pas modifié
l'incidence des tumeurs. La dose sans effet observé relative à la
toxicité générale était de 100 mg/kg de nourriture.
Chez des rats à qui l'on avait administré de la deltaméthrine
en doses allant jusqu'à 50 mg/kg de nourriture pendant deux ans, on
n'a observé aucune tumeur attribuable à cette substance. La dose
sans effet observé relative à la toxicité générale était de 5 mg/kg
de nourriture.
La deltaméthrine ne s'est pas révélée mutagène dans divers
systèmes d'épreuve in vivo et in vitro; notamment la réparation de
l'ADN, la mutation génique, les aberrations chromosomiques,
l'échange entre chromatides-soeurs, la formation de micronoyaux et
la létalité dominante.
Des études de tératogénicité ont été effectuées chez des rates
et des souris gravides à qui l'on administrait par voie orale des
doses quotidiennes de deltaméthrine allant jusqu'à 1000 mg/kg
pendant la phase principale de l'organogénèse. On n'a constaté
aucun effet tératogène ni altération de la fonction de reproduction
chez ces rates et ces souris, si ce n'est une réduction, liée à la
dose, dans le poids moyen des foetus chez les souris et un léger
retard d'ossification chez les rats.
Des lapines gravides ont reçu au sixième et dix-neuvième jours
de leur grossesse des doses quotidiennes de deltaméthrine allant
jusqu'à 16 mg/kg. A la dose la plus forte, on a noté une réduction
du poids moyen des foetus. Aucun effet tératogène n'a été observé
chez les lapins.
Après administration de deltaméthrine à des rats à des doses
allant jusqu'à 5 mg/kg de nourriture, dans le but d'effectuer une
étude de reproduction sur trois générations et deux portées, on a
constaté l'absence totale de tout effet.
Il existe certains indices selon lesquels l'association de
delta- méthrine à certains composés organophosphorés pourrai
conduire à une potentialisation de la toxicité de ces produits.
1.1.8 Effets sur l'homme
La deltaméthrine peut provoquer certaines sensations cutanées
chez des travailleurs exposés. Plusieurs cas d'intoxication
professionnelle non mortelle ont été signalés qui étaient dus à
l'inobservation des mesures de sécurité. Un engourdissement, un
prurit, des fourmillements et une sensation de brûlure de la peau
ainsi que des vertiges sont des symptômes fréquemment signalés. On
a décrit occasionnellement un érythème papulaire ou une couperose à
caractère passager. La plupart de ces symptômes sont passagers et
disparaissent en cinq à sept jours. Aucun effet indésirable à long
terme n'a été signalé. On a décrit trois cas d'empoisonnement non
mortel par la deltamétrine à la suite de l'ingestion de plusieurs
grammes de ce produit.
1.2 Conclusions
Population générale: l'exposition de la population générale à la
deltaméthrine est vraisemblablement très faible et ce produit,
lorsqu'il est utilisé conformément aux recommandations, ne présente
probablement aucun risque.
Exposition professionnelle: moyennant de bonnes méthodes de
travail et l'application de mesures d'hygiène et de sécurité, la
deltaméthrine ne devrait pas présenter de danger pour les personnes
qui y sont exposées de par leur profession.
Environnement: Utilisée aux doses recommandées, il est improbable
que la deltaméthrine ou ses produits de dégradation s'accumulent au
point d'avoir des effets nocifs sur l'environnement. Au
laboratoire, la deltaméthrine est très toxique pour les poissons,
les arthropodes aquatiques et les abeilles. Toutefois, sur le
terrain, il est peu probable que le produit ait des effets nocifs
s'il est utilisé conformément aux recommandations.
1.3 Recommandations
Bien que les teneurs dans les aliments soient très faibles
lorsque la deltaméthrine est utilisée conformément aux
recommandations, il est souhaitable de s'en assurer en soumettant
la deltaméthrine à une surveillance.
La deltaméthrine est utilisée depuis de nombreuses années et
l'on signale un certain nombre de cas d'intoxications non mortelles
ainsi que des effets passagers par suite d'exposition
professionnelle. Il est souhaitable que l'exposition humaine
continue d'être surveillée.
1. RESUMEN Y EVALUACION, CONCLUSIONES Y RECOMENDACIONES
1.1 Resumen y evaluación
1.1.1 Identidad, propiedades fíisicas y químicas, métodos de
análisis
La deltametrina fue sintetizada en 1974 y se comercializó por
primera vez en 1977. Químicamente, es el isómero [1R, cis; S] de 8
ésteres estereoisoméricos del análogo dibromo del ácido
crisantémico, ácido 2,2-dimetil-3-(2,2-dibromovinil) ciclopro-
panocarboxílico (Br2CA) con alcohol alpha-ciano-3-fenoxibencílico.
La deltametrina de calidad técnica es un polvo blanco inodoro
con un punto de fusión de 98 - 101 °C y contiene más del 98% del
material. La presión del vapor es 2,0 x 10-6 Pa a 25 °C y es
prácticamente no volátil. Es insoluble en agua, pero soluble en
disolventes orgánicos como la acetona, la ciclohexanona y el
xileno. Es estable a la luz, el calor y el aire, pero inestable en
medios alcalinos.
La determinación de la presencia de residuos y el análisis de
muestras ambientales se efectuaron por extracción con los
disolventes n-hexano/acetona, partición con n-hexano/acetona/agua,
absorción con un cromatógrafo de columna de gel de sílice y
determinación con un cromatógrafo de fase gaseosa equipado con un
detector de captura de electrones con una concentración mínima
detectable de 0,01 mg/kg o menos. Para el análisis de productos se
utiliza la cromatografía de fase líquida de alto rendimiento con un
detector de rayos UV.
1.1.2 Producción y empleo
El consumo mundial de deltametrina era de 250 toneladas
aproximadamente en 1987. La deltametrina se utiliza sobre todo en
el algodón (45% del consumo), en cultivos como el café, el maíz,
los cereales, la fruta, las hortalizas y el lúpulo, y en productos
almacenados. Se emplea también en higiene animal, en la lucha
contra los vectores y en salud pública. Se fabrica como solución
concentrada emulsionable, solución concentrada de pequeñísimo
volumen, polvo humectable, solución concentrada en suspensión y
polvo para utilización en seco, sola o en combinación con otros
plaguicidas.
1.1.3 Exposición humana
La exposición de la población en general a la deltametrina
procede principalmente de residuos en la dieta, pero puede resultar
también de su empleo en salud pública. En los cultivos tratados de
acuerdo con buenas prácticas agrícolas, las concentraciones
residuales son por lo general muy bajas, excepto en caso de
tratamiento después de la recolección. La FAO y la OMS han
examinado abundantes datos al respecto.
Se cree que la exposición de la población en general es muy
baja, pero no se dispone de datos en forma de estudios de dietas
totales.
1.1.4 Exposición ambiental y evolución
Cuando la deltametrina-[1R, cis; alpha S] marcada con
C14-(compuesto irradiado ácido, alcohol o ciano) se expuso a la
luz del sol en forma de una película fina durante 4 a 8 horas, el
70% se transformó por isomerización- cis/trans produciendo los
isómeros [1R, trans; alpha S] y [1S, trans; alpha S], junto con
productos de la escisión de los ésteres, entre ellos Br2CA y
alcohol alpha-ciano-3-fenoxibencílico.
La deltametrina se degradó en las plantas de algodón, en
condiciones de invernadero, con una vida media inicial de 1,1
semanas, y el tiempo necesario para una pérdida del 90% fue de 4,6
semanas.
Los principales metabolitos fueron Br2CA libre y combinado,
trans-hidroximetil-Br2CA y ácido 3-(4-hidroxifenoxi)benzoico,
formados por la escisión, la oxidación y la combinación de los
ésteres.
La deltametrina se incubó en arena y mantillo orgánico a 28 °C
en condiciones de laboratorio. Ocho semanas después del
tratamiento, se recuperó de la arena y del mantillo orgánico
alrededor del 52% y el 74%, respectivamente, de la deltametrina
aplicada.
La deltametrina no es móvil en el medio debido a su fuerte
adsorción en las partículas, su insolubilidad en el agua y sus
tasas muy bajas de aplicación.
No se dispone de datos sobre las concentraciones existentes en
el medio pero, dadas las modalidades actuales de uso y en
condiciones normales, es de esperar que la exposición ambiental sea
muy baja. Se degrada rápidamente, convirtiéndose en productos
menos tóxicos.
1.1.5 Ingestion, metabolismo y excreción
La deltametrina se absorbe fácilmente por vía oral y con menor
facilidad por la piel, pero la tasa de absorción depende
considerablemente del portador o disolvente. La deltametrina
absorbida se metaboliza y se excreta fácilmente.
En la administración por vía oral a ratas de deltametrina
marcada con C14-(compuesto irradiado ácido, alcohol o ciano), a
tasas de 0,64 - 1,60 mg/kg, el radiocarbono de las partes ácido y
alcohol se eliminó casi por completo en 2 - 4 días. Las
concentraciones residuales en los tejidos fueron por lo general muy
bajas, excepto en la grasa, en donde fueron ligeramente más altas.
Sin embargo, la parte ciano se excretó más lentamente, con una
recuperación total del 79% en 8 días. Las principales reacciones
metabólicas fueron oxidación (en el trans-metilo del anillo de
ciclopropano y en las posiciones 2'-, 4'-, y 5 de la parte
alcohol), escisión de ésteres y conversión de la parte ciano en
tiocianato. Los ácidos carboxílicos y fenoles resultantes se
combinaron con ácido sulfúrico, glicocola y ácido glucurónico.
Cuando se administró a ratones C14-(compuesto irradiado ácido,
alcohol o ciano)-deltametrina por vía oral, a tasas de 1,7 - 4,4
mg/kg, la excreción del radiocarbono fue rápida y casi total,
excepto en la parte ciano. Las principales reacciones metabólicas
observadas en los ratones fueron en general semejantes a las
observadas en las ratas.
En el ganado vacuno y las aves de corral, las vías de
degradación son muy similares a las observadas en los roedores.
1.1.6 Efectos en los organismos presentes en el medio
La deltametrina es muy tóxica para los peces, ya que la CL50 en
96 horas oscila entre 0,4 y 2,0 µg/litro. Es también muy tóxica
para los invertebrados acuáticos: la CL50 en 48 horas para Daphnia
es de 5 µg/litro. Sin embargo, extensos estudios realizados en
estanques experimentales y el empleo sobre el terreno han
demostrado que esa elevada toxicidad potencial no llega a
concretarse. En la práctica hay algunas muertes de invertebrados
acuáticos que, por lo general, se compensan con rapidez.
La toxicidad de la deltametrina en las aves es muy baja, con
valores de la DL50 superiores a 1000 mg/kg cuando se administra una
sola dosis por vía oral. En condiciones de laboratorio, la
deltametrina es muy tóxica para las abejas, con una DL50 por
contacto de 0,051 µg/abeja. Los ensayos sobre el terreno y el
empleo no experimental han demostrado que los preparados de
deltametrina tienen una acción repelente, lo cual quiere decir que,
en la práctica, el riesgo para las abejas es escaso.
1.1.7 Efectos en animales experimentales y en sistemas de pruebas
in vitro
En un vehículo no acuoso, la toxicidad de la deltametrina
administrada por vía oral en forma aguda va de alta a moderada, con
DL50 de 19 a 34 mg/kg (ratones) y de 31 a 139 mg/kg (ratas). Sin
embargo, cuando la deltametrina está en suspensión en agua, su
toxicidad es mucho menor con DL50 superiores a 5000 mg/kg (ratas).
La deltametrina es un piretroide de tipo II; los signos clínicos
comprenden temblor, salivación y convulsiones. La intoxicación se
presenta rápidamente y, en los supervivientes, los signos
desaparecen en unos días. El electroencefalograma muestra
descargas generalizadas en picos y ondas, seguidas de
coreoatetosis.
Aplicaciones únicas de deltametrina técnica no tuvieron ningún
efecto irritante en la piel, intacta o con abrasiones, de conejos.
Sin embargo, se observaron efectos irritantes transitorios en los
ojos de conejos, con y sin lavado. La deltametrina no actuó como
sensibilizante dérmico en cobayas.
En ratas a las que se administró deltametrina con sonda, en
dosis de hasta 10,0 mg/kg de peso corporal diarios durante 13
semanas, a las 6 semanas se observó hiperexcitabilidad en los
machos que habían recibido la dosis más alta. El aumento del peso
corporal en los machos fue menor con dosis de 2,5 y 10 mg/kg.
En perros de la raza beagle a los que se administró por vía
oral deltametrina en dosis de hasta 10 mg/kg de peso corporal
diarios durante 13 semanas, se observaron varios síntomas
relacionados con el compuesto, como vómitos, temblor, salivación y
reflejos faríngeo, patelar y flexor deprimidos. En un estudio de
alimentación en perros, de dos años de duración, la dosis
desprovista de efectos fue de 1 mg/kg de peso corporal diario
(dosis más alta estudiada).
En ratones a los que se administró deltametrina en dosis de
hasta 100 mg/kg de alimentos durante 24 meses, no resultó afectada
la incidencia de tumores. En cuanto a la toxicidad general, la
dosis desprovista de efectos fue de 100 mg/kg de alimentos.
En ratas a las que se administró deltametrina a niveles de
hasta 50 mg/kg de alimentos durante dos años, no se observaron
tumores relacionados con el compuesto. La concentración
desprovista de efectos de la toxicidad general fue de 50 mg/kg de
alimentos.
No se observaron efectos mutagénicos de la deltametrina en una
multitud de sistemas de pruebas in vivo e in vitro, incluidos:
reparación del ADN, mutación génica, aberración cromosómica,
intercambio de cromátidas hermanas, formación de micronúcleos y
genes letales dominantes.
Se realizaron estudios teratológicos con ratas y ratonas
preñadas, administrando deltametrina por vía oral en dosis de hasta
10 mg/kg diarias durante el periodo de mayor organogénesis. No se
observaron efectos teratogénicos ni reproductivos en las ratas ni
en las ratonas, excepto una disminución, relacionada con la dosis,
del peso fetal medio en el estudio con ratonas y una osificación
ligeramente retrasada en el estudio con ratas.
Se administró a conejas deltametrina en dosis de hasta 16 mg/kg
diarios entre los días 6 y 19 del embarazo. Con la dosis más alta,
se registró una disminución del peso fetal medio. No se observaron
efectos teratogénicos.
Se administró a ratas deltametrina en dosis de hasta 50 mg/kg
de alimentos en un estudio sobre reproducción con tres generaciones
y dos camadas, sin que se observaran efectos sobre la reproducción.
Hay indicios de que la toxicidad puede potenciarse cuando la
deltametrina se combina con algunos compuestos organofosforados.
1.1.8 Efectos en los seres humanos
La deltametrina puede provocar sensaciones cutáneas en los
trabajadores expuestos. Se han notificado varios casos de
intoxicación no mortal debidos a exposición ocupacional por no
respetar las precauciones de seguridad. Son síntomas
frecuentemente mencionados adormecimiento, picor, hormigueo y
quemazón de la piel, y vértigo. En ocasiones se ha descrito un
eritema papular o maculoso. La mayor parte de esos síntomas son
temporales y desaparecen en 5 ó 7 días. No se han comunicado
efectos negativos a largo plazo. Se han descrito tres casos no
mortales de intoxicación por deltametrina tras la ingestión de
varios gramos del producto.
1.2 Conclusiones
Población en general: No es probable que la exposición de la
población en general a la deltametrina, que se cree muy baja,
represente un riesgo en las condiciones de empleo recomendadas.
Exposición ocupacional: Si se aplican buenas prácticas laborales,
medidas de higiene y precauciones de seguridad, no es probable que
la deltametrina represente un riesgo para las personas
ocupacionalmente expuestas a ella.
Medio ambiente: Es improbable que la deltametrina o los productos
de su degradación alcancen niveles que puedan tener efectos
negativos en el medio con las tasas de aplicación recomendadas. En
condiciones de laboratorio, la deltametrina es muy tóxica para los
peces, los artrópodos acuáticos y las abejas. No obstante, en la
práctica, no es probable que haya efectos perjudiciales duraderos
con las condiciones de empleo recomendadas.
1.3 Recomendaciones
Aunque se cree que con el uso recomendado las concentraciones
en la dieta son muy bajas, debe considerarse la conveniencia de
confirmarlo incluyendo la deltametrina en estudios de vigilancia.
La deltametrina se utiliza desde hace muchos años y se han
notificado varios casos de intoxicación no mortal y efectos
transitorios debidos a exposición ocupacional. Debe mantenerse la
observación de la exposición humana.
After oral administration to male rats at 0.64 - 1.60 mg/kg,
the acid and alcohol moieties of deltamethrin were almost
completely eliminated from the body within 2 - 4 days (Ruzo et al.,
1978). On the other hand, the cyano group was eliminated more
slowly, the total recovery during 8 days being 79% of the
radiocarbon dose (43% and 36% in the urine and faeces,
respectively). Tissue residues of deltamethrin labelled with 14C
at the dibromovinyl carbon in the acid moiety and the benzylic
carbon in the alcohol moiety were generally very low, whereas
residue levels in the fat were somewhat higher (0.1 - 0.2 mg/kg).
Residue levels of the radiocarbon derived from the cyano group were
relatively high, especially in the skin and stomach. Essentially,
all the radiocarbon in the stomach was thiocyanate. No noticeable
14CO2 was evolved from any of the radioactive preparations,
including the CN-labelled group, in contrast to the CN group from
fenvalerate, which yielded 14CO2 in considerable amounts.
The major metabolic reactions of deltamethrin were oxidation
(at the trans methyl relative to carbonyl group of the acid moiety
and at the 2'-, 4'-, and 5-positions of the alcohol moiety),
cleavage of the ester linkage, and conversion of the cyano portion
to thiocyanate and 2-iminothiazolidine-4-carboxylic acid (31)
(ITCA) (see Fig. 3). These carboxylic acid and phenol derivatives
were conjugated with sulfuric acid, glycine, and/or glucuronic
acid.
The major faecal metabolites were unchanged deltamethrin (9),
accounting for 13 - 21% of the dose, followed by 4'-OH- (10) and
5-OH-deltamethrin (28), and a trace amount of 2'-OH-deltamethrin
(29). Intact deltamethrin and the 4'-OH-derivative appeared not
only as the administered S-epimer, but also in parts as the
R-epimer, probably due to artefactural racemization on exchange of
the alpha-position hydrogen in methanol solution. The metabolites
from the acid moiety were mostly 3-(2,2-dibromovinyl)-2,2-
dimethylcyclopropanecarboxylic acid (18) (Br2CA) in free form
(10% of the dose), glucuronide (51%) and glycine (trace level)
conjugates, and OH-Br2CA (20) in free form and glucuronide
conjugate (<1%).
The major metabolites of the aromatic portion of the alcohol
moiety were 3-phenoxybenzoic acid (25) (PBacid) in free form (5%),
and glucuronide (13%) and glycine (4%) conjugates and its
4'-hydroxy derivative (26) (4'-OH-PBacid).
Sulfate of 4'-OH-PBacid accounted for about 50% of the dose,
together with small amounts of free (4%) and glucuronide forms
(2%). The CN group was converted mainly to thiocyanate (30) and,
in small amounts, to ITCA (31) (Ruzo et al., 1978). The trans-
isomer of deltamethrin was also rapidly metabolized and yielded
almost the same metabolites as deltamethrin, though 5-OH-derivative
was found in the cis-isomer, but not in the trans-isomer (Ruzo et
al., 1978).
When a single oral dose of 14C-(acid-, alcohol-, or cyano-
labelled) deltamethrin was administered to male mice at 1.7 - 4.4
mg/kg, the acid moiety and the aromatic portion of the alcohol
moiety were rapidly and almost completely excreted, whereas the CN
group was excreted relatively slowly (Ruzo et al., 1979).
Gray & Rickard (1982) followed the distribution of 14C-acid-,
14C-alcohol-, and 14C-cyano-labelled deltamethrin and selected
metabolites in the liver, blood, cerebrum, cerebellum, and spinal
cord after iv administration of a toxic, but non-lethal, dose
(1.75 mg/kg) to rats. Approximately 50% of the dose was cleared
from the blood within 0.7 - 0.8 min, after which the rate of
clearance decreased. 3-Phenoxybenzoic acid (PBacid) was isolated
from the blood in vivo, and was also the major metabolite when
14C-alcohol-labelled deltamethrin was incubated with blood in
vitro. Deltamethrin levels in the liver peaked at 7 - 10 nmol/g at
5 min and then decreased to 1 nmol/g by 30 min. In contrast, peak
central nervous system levels of deltamethrin were achieved within
1 min (0.5 nmol/g), decreasing to 0.2 nmol/g at 15 min, and
remaining stable until 60 min. Peak levels of deltamethrin were
not related to the severity of toxicity, though the levels of
unextractable pentane radiolabel did appear to be correlated with
signs of motor toxicity. Experiments with brain homogenates from
animals injected iv with deltamethrin failed to reproduce the
pentane-unextractable radioactivity in vitro and metabolism of the
compound was not demonstrated.
The major metabolic pathways of deltamethrin in mice were
similar to those in rats, though there were some differences. These
included the presence of more unchanged deltamethrin in mouse faeces
than in rat faeces. In mouse faeces, there were 4 monohydroxy ester
metabolites (2'-OH-, 4'-OH-, 5-OH-, and trans-OH- deltamethrin
(11)) and one dihydroxy metabolite (12) (4'-OH- trans-
OH-deltamethrin) that were not found in mouse urine. Major
metabolites from the acid moiety in mice were Br2CA,
trans-OH-Br2CA (20), and their glucuronide and sulfate
conjugates. Among them, trans-OH-Br2CA-sulfate was detected
only in mice, but not in rats. Compared with rats, much larger
amounts of trans-OH-Br2CA and its conjugates were formed in
mice. A major metabolite of the alcohol moiety in mice was the
taurine conjugate of PBacid in the urine, which was not detected in
rats. Generally, mice produced smaller amounts of phenolic
compounds compared with rats. Also, 3-phenoxybenzaldehyde (24)
(PBald), 3-phenoxybenzyl alcohol (32) (PBalc), and its glucuronide,
and glucuronides of 3-(4- hydroxyphenoxy)benzyl alcohol (33)
(4'-OH-PBalc) and 5-hydroxy-3- phenoxybenzoic acid (34)
(5-OH-PBacid) were found in mice, but not in rats. When mice were
given an ip dose of 14C-deltamethrin, with or without piperonyl
butoxide (PBO) and/or S,S,S-tributyl- phosphorotrithioate (DEF),
the same metabolites were obtained as with oral administration.
However, DEF decreased the hydrolytic products relative to the
controls, while PBO decreased the oxidation products (Ruzo et al.,
1979).
The comparison between the excreted radioactivity of
14C-deltamethrin in rats treated by the percutaneous route and iv
(controls) showed that only 3.6% of the dosage applied on the skin
was absorbed and excreted in 24 h with 1.1% excreted during the
first 6 h. Since the rat skin is more permeable than human skin,
the uptake of deltamethrin through the human skin should be
relatively weak (Pottier et al., 1982).
5.2 Metabolism and Fate in Farm Animals
In a metabolic study, 14C-deltamethrin was administered orally
to lactating dairy cows at the rate of 10 mg/kg body weight per day
for 3 consecutive days. It was poorly absorbed and mainly
eliminated in the faeces as unchanged deltamethrin. Only 4 - 6% of
the administered 14C was eliminated in the urine, and 0.42 - 1.62%
was secreted in the milk. The radiocarbon contents of various
tissues were generally very low with the exception of those of the
liver, kidney, and fat, which were higher (Akhtar et al. 1986).
Deltamethrin degradation occurred by cleavage of the ester bond, as
already reported in rats and mice (Ruzo et al. 1978, 1979). The
enzymes responsible for the ester bond cleavage were located in cow
liver homogenate, mainly in the microsomal fraction, as seen in an
in vitro study (Akhtar, 1984). Metabolites resulting from ester
bond cleavage were further metabolized and/or conjugated, resulting
in a large number of compounds excreted in the urine (see Fig. 3).
In milk, the major identifiable radiolabelled compound was
deltamethrin.
In a feeding study by Akhtar et al. (1987), deltamethrin was
administered twice daily to lactating dairy cows in portions of
their daily feed at the rate of 2 or 10 mg/kg diet for 28
consecutive days. The level of 2 mg/kg diet was the residue level
found in a recently treated pasture (Hill & Johnson, 1987), whereas
10 mg/kg diet was five times this level. Deltamethrin residues in
the milk were dose-dependent and appeared to reach a plateau
between 7 and 9 days after the start of treatment. At the high
deltamethrin intake of 10 mg/kg diet, the deltamethrin residue in
milk was about 0.025 mg/litre. Deltamethrin residues in tissues
were measured 1, 4, and 9 days after the last dose. At the
10 mg/kg diet intake, very small amounts of deltamethrin residues
were found in the liver (<0.005 mg/kg), kidney (<0.002 mg/kg),
and muscle (0.002 - 0.014 mg/kg). Residues in fat were about
0.04 mg/kg and 0.2 mg/kg for the 2 and 10 mg/kg intake,
respectively. Depletion of deltamethrin residues in milk was very
rapid (estimated half-life was about 1 day); while in fat (renal
and subcutaneous) the half-life was 7 - 9 days. Br2CA (3-(2,2-
dibromovinyl)-2,2-dimethylcyclopropanecarboxylic acid) and PBacid
(3-phenoxybenzoic acid) were the only metabolites detected in the
milk and tissues of treated cows. In all cases, they were found at
trace levels of < 0.0235 mg/litre and < 0.034 mg/litre,
respectively. These two metabolites were also previously
identified in rats and mice as the major degradation products of
deltamethrin (Ruzo et al., 1978, 1979).
The fate of 14C-deltamethrin was examined in Leghorn hens
(Akhtar et al., 1985). When laying hens were administered 7.5 mg
of 14C-labelled deltamethrin/hen per day orally for 3 consecutive
days, about 83% and 90% of the administered 14C was eliminated
during the first 24 h and 48 h after dosing, respectively. Tissue
residues were generally very low with the exception of those in the
liver and kidney. Very low levels of residues were found in eggs
obtained within the first 24 h after dosing, but levels increased
reaching a peak within 48 h of the last dose. Residue levels were
higher in the yolk (up to 0.6 mg/kg) than in the albumen (up to
0.2 mg/kg), which is probably related to the lipid content of
yolks. Metabolites were the same as those found in rats and mice.
These studies showed that feeding domestic animals on
deltamethrin-treated feed resulted in very low levels of residues
(if any) in products of animal origin and is unlikely to present a
hazard for the consumer.
5.3 Enzymatic Systems for Biotransformation
Deltamethrin (1 µg) was incubated at 37 °C for 30 min with each
of the following mouse microsome preparations; a) tetraethyl
pyrophosphate (TEPP)-treated microsomes (no esterase and oxidase
activity); b) normal microsomes (esterase activity); c) TEPP-
treated microsomes plus NADPH (oxidase activity); and d) normal
microsomes plus NADPH (esterase plus oxidase activity) (Shono et
al., 1979). Deltamethrin was more rapidly metabolized under the
oxidase system than under the esterase system. The major site of
ring hydroxylation was the 4'-position and the secondary site was
the 5-position. The trans methyl group was an important site of
hydroxylation of the esters and cis methyl oxidation was evident in
the metabolites of the cleaved acid moiety. The preferred sites of
hydroxylation were as follows; trans of dimethyl group,
4'-position in the phenol group, and cis of the dimethyl group,
which was equal to the 5-position in the phenoxy group. Cleavage
of deltamethrin to cyanohydrin may result from both esterase and
oxidase enzyme activities, since larger amounts of the cleaved
products were evident in the oxidase system.
However, at a much higher (approximately 35-fold) concentration
of deltamethrin than that in the above study, it was not detectably
hydrolysed (Miyamoto, 1976; Soderlund & Casida, 1977).
Deltamethrin was hydrolysed by esterases in the blood, brain,
kidney, and stomach of mice yielding PBald and PBacid (Ruzo et al.,
1979).
5.4 Metabolism in Human Beings
Three young male human volunteers underwent a complete medical
check-up one week prior to the morning of the study. Each of them
received a single dose of 3 mg of 14C-deltamethrin mixed in 1 g
glucose and diluted first in 10 ml PEG 300 and again in 150 ml
water. Total radioactivity was 1.8 ± 09 mBq. Samples of blood,
urine, saliva, and faeces were taken at intervals over 5 days.
Clinical and biological examinations were performed every 12 h
during the trial and one week after its termination. Radioactivity
in the biological samples was measured with a liquid scintillation
spectrometer. The clinical and biological checks did not detect
any abnormal findings. There were no signs of side effects or
intolerance reactions, either during or after the trial period.
The maximum plasma radioactivity appeared between 1 and 2 h after
administration of the product, and remained over the detection
limit (0.2 KBq/litre) during the 48 h. The apparent elimination
half-life was between 10.0 and 11.5 h. The radioactivity of blood
cells, as well as the saliva, was extremely low. Urinary excretion
was 51 - 59% of the initial radioactivity; 90% of this
radioactivity was excreted during the 24 h following absorption.
The apparent half-life of urinary excretion was 10.0 - 13.5 h,
which is consistent with the plasma data. Faecal elimination at
the end of the observation period represented 10 - 26% of the dose.
The total faecal plus urine elimination was around 64 - 77% of the
initial dose after 96 h (Papalexiou et al., 1984).
6. EFFECTS ON ORGANISMS IN THE ENVIRONMENT
6.1 Aquatic Organisms
6.1.1 Acute toxicity for fish
Acute toxicity data for deltamethrin in fish have been
summarized by L'Hotellier & Vincent (1986) (Table 6). From this,
it appears that deltamethrin is highly toxic for fish, though the
toxicity varies with the formulation tested.
Table 6. Acute toxicity of deltamethrin tested as the technical or formulated product on
fish; lethal concentrations all expressed as µg active ingredient (a.i.)/litre (96-h)
------------------------------------------------------------------------------------------
Species Systema LC50 (µg/litre) Ref. LC50 (µg/litre) Ref.
(Common name) tested as No. tested as No.
technical formulated
product productb
------------------------------------------------------------------------------------------
Alburnus alburnus S 0.69 4 82 (ULV) 4
(Bleak)
Brachydanio rerio F,S 2.0 10 -
(Zebra fish)
Cyprinodon macularius S - 0.6c (EC) 13
(Desert pupfish)
Cyprinodon S - 0.9 (EC) 19
variegatusd
(Sheepshead minnow)
Cyrpinus carpio F, S 1.84 4 0.65 (EC) 4
(Common carp) 0.86 3 210.0 (ULV)
Gambusia affinis F, S - 1.0c (EC) 13
(Mosquito fish)
Ictalurus nebulosus F, S 1.2 7 2.3 (EC) 15
(Brown bullhead)
Ictalurus punctatus F, S 0.63 8 -
(Hannel catfish)
Idus idus melanotus S - 1.2 (EC) 16
(Golden orfe)
Lebistes reticulatus F, S - 1.8 (EC) 17
(Guppy)
Lepomis gibbosus F, S 0.58 5 0.87 (EC) 14
(Pumpkinseed sunfish)
Lepomis machrochirus F 1.2 6 -
(Bluegill sunfish)
Osteochilus hasseltie S - 1.2 (EC) 20
(Nilem carp)
Puntius gonionotuse F,S - 0.87 (EC) 18
(Jawa carp)
Rhodeus sericeus S 1.12 4 140 (ULV) 4
amarus
Salmo gairdneri F, S 0.39 1 2.2 (EC) 12
(Rainbow trout)
Salmo salar 1.97 2 0.59 (EC) 2
------------------------------------------------------------------------------------------
Table 6. (contd.)
------------------------------------------------------------------------------------------
Species Systema LC50 (µg/litre) Ref. LC50 (µg/litre) Ref.
(Common name) tested as No. tested as No.
technical formulated
product productb
------------------------------------------------------------------------------------------
Salmo trutta F, S - 4.7c (EC) 11
(Brown trout)
Sarotherodon F, S 3.5 9 2.0 (EC) 9
mossambicuse
Tilapia mossambicae F, S - 0.8c (EC) 13
------------------------------------------------------------------------------------------
a F: Flow system, S: Static condition.
b EC: 25 g a.i./litre; ULV: 1 g a.i./litre; values in a.i. equivalent obtained by
calculation.
c LC50 (48-h)
d Marine fish.
e River or pond fish from tropical areas (water temperature > 24 °C).
References
(1) Knauf & Horlein (1979); (2) Zitko et al. (1979); (3) Knauf & Schulze (1977a);
(4) Gulyas & Csanyi (undated); (5) Waltersdorfer & Schulze (1976a); (6) Buccafusco et al.
(1977a); (7) Knauf & Schulze (1977b); (8) Buccafusco et al. (1977b); (9) Adeney et al.
(1980); (10) Lepailleur & Chambon (1984); (11) Lhoste et al. (1979); (12) Waltersdorfer &
Schulze (1976c); (13) Mulla et al. (1978); (14) Waltersdorfer & Schulze (1976d); (15) Knauf
& Schulze (1977b); (16) Waltersdorfer & Schulze (1976b); (17) Waltersdorfer & Schulze
(1976a); (18) Santosa & Hadi (1980); (19) Heitmuller et al. (1978); (20) Santosa (1983)
Zitko et al. (1979) established a 96-h lethal threshold for
Atlantic salmon (Salmo salar) of 1.97 µg/litre.
6.1.2 Acute toxicity for other aquatic organisms
Data on aquatic organisms other than fish are presented in
Table 7 and are of the same order as those for fish, although the
oyster (Crassostrea virginica) is somewhat more tolerant and the
Northern lobster (Homarus americanus) (96-h lethal threshold
0.0014 µg/litre) is far more sensitive (Zitko et al., 1979).
Mohsen & Mulla (1981) exposed aquatic insect larvae to
deltamethrin (as a 2.5% emulsifiable concentrate) for 1 h under
flow-through conditions, and calculated the LC50 after a 24-h
holding period. For the target species blackfly (Simulium
virgatum) an LC50 of 0.9 µg/litre was calculated. Non-target
species tested, mayfly (Baetis parvus) and caddisfly (Hydropsyche
californica), were found to be more susceptible, with LC50 values
of 0.4 µg/litre.
Varanka, (1987) investigated the effects of deltamethrin on
three species of freshwater mussels. Results presented in Table 8
show that the mussels are very insensitive to the pyrethroid.
Table 7. Acute toxicity of deltamethrin tested as technical or
formulated product on other aquatic organisms-lethal concentrations
expressed as µg active ingredient (a.i.)/litre (96-h)a
-------------------------------------------------------------------
Species LC50 (µg/litre) LC50 (µg/litre)
tested as technical tested as formulated
product productb
-------------------------------------------------------------------
Crassostrea virginica - 12.0
(Eastern oyster)
Daphnia magna 5c -
(Water flea)
Gammarus pulex - 0.03c
(Scud)
Penaeus duorarum - 0.35
(Pink shrimp)
Uca pugilatorulosus - 1.1
(Fiddler crab)
Bufo bufo (larvae) - 0.93
(Common toad)
-------------------------------------------------------------------
a Adapted from: L'Hotellier & Vincent (1986).
b EC: 25 g a.i./litre; ULV: 1 g a.i./litre; values in a.i.
equivalent obtained by calculation.
c LC50 (48-h).
Table 8. Acute toxicitya of deltamethrin formulationb in
freshwater mussels, under static conditions at 21 - 23 °Cc
-------------------------------------------------------------------
Species 24-h 48-h 72-h 96-h 7-day
-------------------------------------------------------------------
Anodonta cygnea nd nd ~24.6 12.0 7.6
Anodonta anatina nd nd nd ~23.4 10.3
Unio pictorum nd ~31.8 9.7 7.0 6.0
-------------------------------------------------------------------
a LC50 µg active ingredient (a.i.)/litre): values in a.i.
equivalent obtained by calculation.
b ULV 0.12%.
c From: Varanka (1987).
6.1.3 Field studies and community effects
Two experimental pond studies have been performed. Tooby et
al. (1981) reported that application of deltamethrin to static
water at 10 g a.i./ha did not have any lethal effects on two fish
species (Canassius auratus, Rutilus rutilus) or on molluscs.
Aquatic insects and crustaceans present were killed. Rawn et al.
(1985) applied deltamethrin at a similar rate and also reported
that no fish were killed. The half-life of deltamethrin in the
pond was 2 - 4 h for water and 2 - 14 days for bottom sediment.
Neto et al. (1983) sprayed-flooded fields in Brazil, at
intervals of 2 days, with rates of deltamethrin progressively
increased at 5, 10, 12, and 13 g a.i./ha. The expected
concentrations in water from these applications were between 3 and
7 µg/litre. No mortality was recorded in fish placed in the
sprayed area in experimental cages. Slight "agitation" was
reported after exposure to the highest dose.
Impact assessments on the use of deltamethrin on paddy fields
have been made in the field in various countries throughout the
world. The maximum normal usage rate of the compound was 6.5 g
a.i./ha. In these studies, fish ( Tilapia spp., Cyprinus carpio,
Gambusia spp.) tolerated deltamethrin up to 18.75 g a.i./ha without
any adverse effects. The compound is known to be toxic for aquatic
organisms and is not recommended for use over water under any but
exceptional circumstances. However, it has been used to control
vectors of major human diseases, i.e., mosquitos and blackfly
( Elossina spp.), where benefit outweighed potential risk. In these
cases, extensive field evaluations of the environmental impact have
been made. While there have not been any instances of fish kills
from these applications, there are reports of large numbers of
deaths of aquatic invertebrates. The populations usually recovered
rapidly and all studies have shown numbers back to normal before
the compound was applied again in the following season. It is
suggested that relatively resistant parts of the population soon
recolonize the area; immigration also occurs (Takken et al., 1978;
Smies et al., 1980; Baldry et al., 1981; Everts et al., 1983).
6.1.4 Appraisal
Notwithstanding its high toxicity for fish and crustacea, the
results of many studies, as well as the wide use of deltamethrin
for several years, have confirmed that its normal use does not
cause significant mortality in fish populations. This difference
is due to its strong adsorption on soil and its rapid breakdown,
decreasing its bioavailability under field conditions.
6.2 Terrestrial Organisms
6.2.1 Plants
Hargreaves & Cooper (1979) sprayed glasshouse-grown tomato
seedlings with 50 mg deltamethrin/litre (2.5% emulsifiable
concentrate) 3 weeks after emergence and again 7 days later. Three
days after the second application, plants were examined for damage.
No damage was found and, at this rate of use, deltamethrin was not
phytotoxic.
6.2.2 Soil microorganisms
In a study by Tu (1980) on the effects of 5 pyrethroids on
microbial populations and their activity in soil, 0.5 mg
deltamethrin/kg incorporated into sandy loams (residues under
normal use conditions would be of the order of < 0.001 mg/kg)
produced only a few transient effects. No effects were noted on
the nitrifying microorganisms and their capacity to produce nitrate
and there were no inhibitory effects on deshydrogenase or urease
activity. Deltamethrin induced an increase in oxygen consumption
because of an increase in microbial respiration (probably linked
with the microbial degradation of deltamethrin). It also
stimulated the growth of soil fungi and inhibited the development
of bacteria. Four weeks after treatment, deltamethrin-treated soil
recovered completely and microorganism activity was equal to that
in untreated soil.
6.2.3 Soil fauna
6.2.3.1 Earthworms
When deltamethrin at 12.5 g a.i./ha (high agricultural dose)
was incorporated into the soil to a depth of 1 cm, there were no
toxic effects on earthworms (Lumbricus terrestris) during an
observation period of 28 days (Bouche & Fayolle, 1979). However,
significant toxic effects on earthworms were observed at levels of
60 - 125 g a.i./ha (5 - 10 times the highest rates applied in
agriculture).
In another study with Eisenia foetida andrei, deltamethrin
incorporated in artificial soil at concentrations of 1.7 mg/kg and
10 mg/kg did not produce any lethal effects (Chambon & Lepailleur,
1984).
6.2.3.2 Slugs
Lettuce leaves treated with 4 times normal dosage rates, were
fed to slugs ( Agrolimax sp.). Leaves were quickly consumed but no
toxic effects (mortality or activity) were observed (Ricou, 1978).
6.2.3.3 Soil arthropods
Under laboratory conditions, deltamethrin, applied topically
and by immersion, was very toxic for the carabid beetle
Pterostichus melanarius (Illiger). Under natural conditions in the
field, deltamethrin applied at normal dose rates was not toxic for
these organisms (Dunning et al., 1981).
Everts et al. (1985) monitored the effects, on non-target
organisms, of various compounds when used for the control of tsetse
fly in the Ivory Coast in Africa. Deltamethrin was the most
effective compound against the tsetse and also killed non-target
musca flies. After deltamethrin spraying, Orthoptera and
Proctotrupoidea were also significantly decreased while Nematocera
increased in number. The results of this study suggest that ground
spraying of the pyrethroid had greater effects on terrestrial
arthropods than aerial applications.
Concurrent laboratory and field studies were conducted on the
effects of deltamethrin on beneficial predatory spiders in a polder
area of the Netherlands (Everts et al., 1988). During two growing
seasons, 2800 samples were taken over an area of 17 different
fields. The authors found that effects on spiders were eliminated
when it rained soon after application, since the effect of the
pyrethroid appeared to be indirect, causing the dehydration of
spiders. This different response under dry and damp conditions was
confirmed in the laboratory. However, reduction of spider numbers
in the field was much greater than predicted from laboratory tests
and recovery was more rapid in laboratory populations than in field
populations. The uptake and effects of deltamethrin were greater
through exposure to residues than through contact or oral exposure.
There was a positive correlation between temperature and the
toxicity of deltamethrin for spiders in the field. This contrasted
with reports of a negative correlation for target insects reported
in the literature. Laboratory studies showed that the negative
temperature effect only occurred when spiders could not drink. It
appeared that qualitative prediction from laboratory to field was
possible but that quantitative prediction was not.
6.2.4 Beneficial insects
6.2.4.1 Honey-bees
Single applications of deltamethrin are highly toxic for honey-
bees (Apis mellifera). Stevenson et al (1978) found a contact LD50
of 0.051 µg/bee and an oral LD50 of 0.079 µg/bee.
Arzone & Vidano (1978) did not find any difference in mortality
between controls and bees fed on sugar solutions containing 0.2 µg
deltamethrin/litre. Increased mortality was recorded at all higher
exposures reaching 100% within 1 h at a concentration of 12.5
µg/litre.
In the field, direct treatment of caged bees caused a high
mortality rate with doses of from 11.2 g/ha upwards (Atkins et al.,
1976). Rape flowers were treated at a rate of 0.75 g a.i./100
litre and 1.5 g a.i./100 litre with an emulsifiable concentrate
formulation, 25 g/litre; control plots were treated with water.
Cages (3 x 2 x 2 m) containing a small hive (2 frames + open brood)
were put over the treated flowers once the spray had dried. The
mortality of the bees was then assessed over 7 days. The average
mortalities were not significantly higher in the treated plots than
in water-sprayed control plots (Louveaux et al., 1977).
However, Bocquet et al. (1980, 1983) demonstrated, after 3
years of field experiments, that deltamethrin under field
conditions was innocuous at doses up to 12.5 g/ha. They also noted
a repellant effect by the formulating materials, which lasted for
2 - 3 h. Further studies have been reported by Florelli et al.
(1987a,b).
6.2.4.2 Foliar insects
Deltamethrin was 70 times more toxic to the tobacco budworm
(Heliothis virescens) than to its predator, green lacewing
(Chrysopa carnea), but it was only 1.25 times more toxic to the
tobacco budworm than to its parasite (Campoletis sonorensis) (Plapp
& Bull, 1978).
In an apple orchard, where deltamethrin was applied at
12.5 mg/kg, no predatory mites (Typhlodromus pyri) were found
during 10 weeks of observation, but spider mites (Paponychus ulmi)
were not affected. The elimination of the predatory mite led to a
marked increase in spider mite populations, later in the same
season (Aliniazee & Cranham, 1980).
The impact of deltamethrin used against the English grain aphid
(Sitobion avenae) was studied in 1983, 1984, and 1985 in the Paris
basin. This study was carried out on wheat with pitfall traps,
yellow water traps, suction sampling (D-vac), and sampling of ears.
Effects were noted on: S. avenae, phytophagous Diptera (Opimyza
florum, Phytomyza nigra, and Oscinella frit), Homoptera (Zyginidia
scutellaris, Metopolophium dirhodum), Thysanoptera (Limothrips
cerealium, Acolothrips intermedius), predatory Diptera (Empididae,
Dolichopodidae), and on spiders (Erigonidae, Lycosidae,
Linyphiidae, Theridiidae). The detritiphagous insects (Sciaridae,
Chironomidae), the Carabidae and Staphylinidae and most
microhymenoptera showed little or no difference after treatment.
During the 3 years, no differences were observed from year to year
as a result of field treatment, populations appearing homogeneous
at the beginning of each trial (Fischer & Chambon, 1987).
A large-scale field trial was carried out in 1984 in southern
England to investigate the side-effects of deltamethrin on non-
target arthropods in winter wheat. The insecticides were applied
in June and two methods, suction sampling (D-vac) and quadrats,
were used to sample the arthropods for up to 75 days after
treatment. During the post-treatment period, the numbers of
Carabidae and Staphylinidae adults found in D-vac samples were
reduced by 22% and 20%, respectively, compared with the controls
(Vickerman et al., 1987a).
In the same field trial, arthropods were sampled with a D-vac
for 11 weeks. Total numbers were similar in the control and
deltamethrin-treated plots. The numbers of Empididae were reduced
by deltamethrin, but Dolichopodidae were more numerous in treated
than in control plots. The numbers of Aphidius spp. were higher in
the deltamethrin-treated plots than in the control plots. The
numbers of Coccinellidae larvae were reduced (Vickerman et al.,
1987b).
6.2.5 Birds
6.2.5.1 Laboratory studies
Data on the acute toxicity of deltamethrin for birds are given
in Table 9.
Table 9. Acute toxicity of deltamethrin for birds
---------------------------------------------------------------------------
Species Sex Application LD50 (mg/kg) Reference
---------------------------------------------------------------------------
Red partridge male & oral >3000 Grolleau & Griban,
(Alectonis tufa) female 1976b
Grey partridge male & oral >1800 Grolleau & Griban,
(Perdix perdix) female 1976b
Chicken oral >1000 Grandadam, 1976
(Gallus domestica)
Hen adult oral >2500 Ross et al., 1978
female
Mallard duck oral >4640 Beavers & Fink,
(Anas platyrhynchos) 1977a
Game duck oral >4000 Grolleau & Griban,
1976a
---------------------------------------------------------------------------
The toxicity of deltamethrin for birds is very low. Both
technical grade and commercially formulated deltamethrin
administered in feed at 100 mg/kg diet was not palatable to
Japanese quail (Coturnix coturnix japonica), with strong individual
variations. Unpalatability diminished after repeated exposure and
even became reversed in the case of the purified deltamethrin,
which attracted quail already suffering from toxic effects (David,
1981).
Groups of 39 female Japanese quail (Coturnix coturnix japonica)
were given daily doses of 0, 0.2, or 1 mg technical deltamethrin
per animal, by gavage, over 34 days. No significant effects were
observed on reproduction (De Lavaur et al., 1985).
6.2.5.2 Field studies on birds
The low toxicity of deltamethrin for birds, indicated by
laboratory studies, has been confirmed in the field. In studies on
the ecological consequences of the use of the compound to control
tsetse fly (Takken et al., 1978) and blackfly (Smies et al., 1980)
in West Africa, populations of various species of insectivorous,
granivorous, and piscivorous birds were examined before and after
spraying. There were no indications of any effects on either
numbers or species diversity.
7. EFFECTS ON EXPERIMENTAL ANIMALS AND IN VITRO TEST SYSTEMS
7.1 Single Exposures
Tables 10 and 11 show the results of acute toxicity studies on
various animal species. From these tables, it is clear that the
vehicle has a great influence on the LD50, probably by influencing
absorption. Powder formulations and aqueous suspensions are
significantly less toxic than formulations in oils or organic
solvents (Pham Huu Chanh et al., 1984).
The acute oral toxicity of deltamethrin for rats produced such
symptoms as: staining of the fur, excessive grooming, salivation,
diarrhoea, drowsiness, weakness, dyspnoea, piloerection, ptosis,
difficulty in walking, general motor incoordination, hypotonia,
choreoathetosis, clonic seizures, and death (Glomot, 1979; Glomot
et al., 1979, 1981a; Kavlock et al., 1979; Ray & Cremer, 1979; Pham
Huu Chanh et al., 1984). Electroencephalogram (EEG) records showed
generalized spike discharges prior to choreoathetosis (Ray &
Cremer, 1979; Ray, 1980).
Mice presented far fewer symptoms than rats after oral dosing
at comparable levels, diarrhoea being the only reportable
observation (Glomot et al., 1980a).
Rats were injected intraperitoneally with 14C-labelled
deltamethrin at the threshold doses required to produce the motor
symptoms of toxicity of tremor and choreoathetosis. Blood and brain
samples were analysed for their total radiolabel content, and were
also extracted with ethyl acetate to determine the levels of
extractable parent deltamethrin and 3-phenoxybenzyl-derived acid and
the residual radiolabel after this extraction. There was a clear
correlation between onset of symptoms and blood and brain levels of
deltamethrin. It was found that certain threshold levels of parent
deltamethrin in the blood and brain were required for symptoms
development, and that the symptoms persisted for as long as this
threshold was maintained (Rickard & Brodie, 1985).
7.1.1 Mouse
Mice intravenously injected with deltamethrin showed intense
tremors, convulsions, and ataxia, immediately after administration.
Tachycardia and respiratory defects were also observed at higher
dosages. Surviving animals appeared normal after 4 - 5 h.
Immediately after intraperitoneal injection, jumping movements,
slight convulsions and prostration, ptosis, tail hypertonicity, and
cyanosis were observed. These toxic signs disappeared after 72 h
in surviving animals.
Animals administered deltamethrin by gavage showed muscular
stiffening and convulsions, 1 h after dosing. After 24 h,
hypermotility, stereotype movements of the head, tachycardia,
hypertonicity of the tail, and a few convulsions were observed.
Behaviour and appearance were normal again after 48 h (Glomot &
Chevalier, 1976a,c).
Table 10. Acute toxicity of technical grade deltamethrin
--------------------------------------------------------------------------------------------------------------
Species Sex Route Vehicle LD50 (mg/kg Reference
body weight)
--------------------------------------------------------------------------------------------------------------
Rat male oral sesame oil 128 Glomot & Chevalier (1976a)
female 139
male PEG 200 67
female 86
Rat male adult peanut oil 52 Kavlock et al. (1979)
female adult 31
female weanling 50
Rat male adult peanut oil 53 Gaines & Linder (1986)
female adult 30
female weanling 48
Rat male + female aqueous suspension > 5000 Audegond et al. (1981)
with carboxy- (no mortality)
methylcellulose
Rat dermal - 700 Panshina & Sasinovich (1983)
Rat male methylcellulose (1%) > 2940 Kynoch et al. (1979)
female
Rat female adult xylene > 800 Kavlock et al. (1979)
Rat male + female inhalation (6 h) dust 600 mg/m3 Coombs & Clark (1978)
Rat male adult (2 h) DMSO 10% aerosol 940 mg/m3 Kavlock et al. (1979)
female adult > 785 mg/m3
Rat male + female (1 h) micronized powder > 4620 mg/m3 Jackson & Hardy (1986)
Rat intraperitoneal - 58.8 Panshina & Sasinovich (1983)
Rat male intraperitoneal sesame oil 209 Glomot & Chevalier (1976b)
female 186
male PEG 200 24 Glomot & Chevalier (1976b)
female 25
--------------------------------------------------------------------------------------------------------------
Table 10. (contd.)
--------------------------------------------------------------------------------------------------------------
Species Sex Route Vehicle LD50 (mg/kg Reference
body weight)
--------------------------------------------------------------------------------------------------------------
Rat male intravenous PEG 200 3.3 Glomot & Chevalier, (1976c)
female 3.3
Rat female adult acetone 4 Kavlock et al. (1979)
female weanling 1.8
Mouse male oral sesame oil 33 Glomot & Chevalier (1976a)
female 34
male PEG 200 21 Glomot & Chevalier (1976a)
female 19
Mouse intraperitoneal - 33 Panshina & Sasinovich (1983)
Mouse male intraperitoneal sesame oil 171 Glomot & Chevalier (1976b)
female 166
Mouse male PEG 200 18 Glomot & Chevalier (1976b)
female 12
Mouse male PEG 200 4.1 Glomot & Chevalier (1976c)
female 4.0
Mouse male glycerol formal 5 Glomot & Chevalier (1976c)
female 5.8
Dog male + female oral in capsules >300 Glomot et al. (1977)
no mortality
Dog male + female PEG 200 2 Glomot & Chevalier (1976c)
Rabbit male dermal PEG 400 > 2000 Clair (1977)
female > 2000
--------------------------------------------------------------------------------------------------------------
Table 11. Acute toxicity of some formulations
--------------------------------------------------------------------------------------------------------------
Species Sex Route Formulation LD50 (mg/kg Reference
body weight)
--------------------------------------------------------------------------------------------------------------
Rat male, female oral 2.5% flowable formulation 22 000 Glomot et al. (1979)
Rat male, female oral 2.5% wettable powder >15 000 Glomot (1979)
Mouse male, female oral 2.5% wettable powder >15 000 Glomot et al. (1980a)
Dog male, female oral 2.5% wettable powder >10 000 Glomot et al. (1980b)
Rat male, female oral 2.5% emulsifiable concentrate 535 Coquet (1977)
Rat male, female oral 10 g/litre ULV >6 470 Coquet (1977)
Rat male, female inhalation (4 h) aerosol-2.5% wettable powder >2 800 mg/m3 Clark et al. (1980)
--------------------------------------------------------------------------------------------------------------
7.1.2 Rats
Rats intravenously injected with deltamethrin showed muscular
contractions, piloerection, respiratory defects, convulsions, and
paresis of the hind quarters, immediately following treatment.
Surviving animals showed normal behaviour after 48 h. Immediately
after intraperitoneal injection, tremor, convulsions, prostration,
and cyanosis were observed. These toxic signs disappeared after
48 h in surviving animals. Animals administered deltamethrin by
gavage showed motor incoordination, convulsions, respiratory
defects, and hypomotility, shortly after dosing. Normal behaviour
was observed after 3 days (Glomot & Chevalier, 1976c).
In an inhalation study (whole body exposure for 6 h),
hyperactivity, grooming, and irritation were observed during
exposure. The animals were hypersensitive to touch and noise and
showed uncoordinated movements. Gross pathological investigation
showed a gas-filled stomach and small intestine, and massive
haemorrhage and degeneration in the lung (Coombs & Clark, 1978).
Rats were exposed (whole body exposure) for 4 h to an aerosol
concentration of deltamethrin equal to 2.8 g/m3, the highest
attainable airborne concentration of a 2.5% wettable powder
formulation. Approximately 80% of the total aerosol had a mean
aerodynamic diameter of less than 5.5 µm. Dyspnoea and gasping
were observed in exposed rats. Relative lung weights and
macroscopic pathology were normal. There was no mortality (Clark
et al., 1980).
7.1.3 Rabbit
Rabbits (10 males and 10 females) were treated with 2 g
deltamethrin in 2 ml PEG 400 per kg body weight on 80 cm2 of
occluded shaved skin for 24 h. The animals were observed for 14
days. Two animals showed obvious erythema. No body weight changes
or abnormal behaviour were observed. On histological observation
of the liver, kidneys, and skin, small changes were observed, but
these were common for this strain of rabbit and not related to
treatment (Clair, 1977).
7.1.4 Dog
Dogs given oral doses of 100 mg deltamethrin/kg body weight or
more showed transient hyperexcitability, akinesia, vomiting, and
stiffness of the hind legs (Glomot et al., 1977).
Dogs orally dosed with 10.0 mg deltamethrin/kg body weight did
not display any clinical signs related to treatment (Glomot et al.,
1980b).
7.2 Irritation and Sensitization
7.2.1 Skin irritation
Male albino rabbits (12 per group) weighing 2.5 - 3.5 kg were
administered 0.5 g deltamethrin on either shaved intact or abraded
skin. The occlusive patch was fixed on the skin for 23 h.
Technical deltamethrin (98% purity) did not produce any irritant
effects (Coquet, 1976a).
Male albino rabbits (6) weighing 2.5 - 2.9 kg were administered
0.5 ml of formulated deltamethrin (25 g/litre flowable suspension
concentrate) to both shaved intact and abraded skin. The Primary
Irritation Index after 24 h exposure of occluded sites was 1.2,
i.e., slightly irritating (Glomot et al., 1981b).
An evaluation similar to the one described above was carried out
for a 2.5% wettable powder concentrate deltamethrin formulation.
Rabbits had a Primary Irritation Index of 2.41, i.e., moderately
irritating. Moderate erythema continued for 72 h, while the oedema
generally diminished, with the exception of scarified skin sites
(Glomot et al., 1981c).
The skin irritation potentials of Decis emulsifiable
concentrate 2.5% and Decis Flowable 2.5% were studied on rabbits
and guinea-pigs with 0.05, 0.10, 0.5, 1, and 2.5% deltamethrin.
The threshold irritative levels were 0.05% for Decis emulsifiable
concentrate and 2.5% for Decis Flowable. The intensity of
irritation depended on the relative content of organic solvents and
emulsifiers in the trade products. The water-soluble concentrate
of Decis 2.5% caused negligible risk of contact irritative
dermatitis (Bainova & Kaloyanova, 1985).
7.2.2 Eye irritation
Deltamethrin (0.1 g/animal) was administered into the
conjunctival sac of the eyes of 6 male albino rabbits, weighing
2.5 kg, with or without rinsing 60 seconds after instillation.
Deltamethrin produced transient irritating effects, both with and
without rinsing (Coquet, 1976b).
Male albino rats (9) weighing between 2 and 3 kg were
administered 0.1 ml of formulated deltamethrin (25 g/litre flowable
suspension concentrate) in the conjunctival sac. Six of the
treated eyes remained unwashed, while the remaining three were
rinsed with lukewarm water 20 - 30 seconds after instillation.
There was only transient clouding of the cornea in 2 animals 1 h
after dosing (1 washed, 1 unwashed), which cleared by day 2. Low
grade conjunctival irritation was noted among all animals
initially, which disappeared following day 2 of observations
(Glomot et al., 1981d).
A 2.5% deltamethrin formulation diluted 1/10 in distilled water
(0.1 ml per rabbit) elicited a similar pattern of initial transient
corneal clouding in 3 out of 9 rabbits examined, which cleared by
day 4. The undiluted formulation (100 mg) administered in the
conjunctival sac of rabbits produced increased involvement of the
conjunctiva, iris, and cornea in all animals, generally moderate in
severity, with low grade corneal opacity persisting in 2 rabbits
until day 7 (1 washed, 1 unwashed) (Glomot et al., 1981e).
7.2.3 Sensitization
Deltamethrin (0.5 g/animal) was applied topically to the skin
of albino guinea-pigs (10 male and female) 3 times per week, with a
2-day interval for 3 weeks, and once at the start of the fourth
week. The preparation was covered with an occlusive patch for
48 h. On days 1 and 10, the guinea-pigs received an intradermal
injection of 0.1 ml of Freund's adjuvant. The animals were
challenged 12 days after the last application with 0.5 g
deltamethrin. No sensitization was found (Guillot & Guilaine,
1977).
7.3 Short-Term Exposure
7.3.1 Rat
Male and female weanling Sprague-Dawley rats (20 of each sex
per group) were dosed (by gavage) with 0, 0.1, 1, 2.5, or 10 mg
deltamethrin in PEG 200/kg body weight per day for 13 weeks. No
treatment-related effects were observed on food and water
consumption, mortality, urinalysis, and haematology. Neurological
examinations and ophthalmoscopy did not reveal any abnormalities.
At the highest dose level, a slight hyperexcitibility was observed
among some rats in week 6. Lower body weight gain was noted in
males at 2.5 and 10 mg/kg. No clear treatment-related effects were
noted in the results of laboratory investigations or on the weights
of the organs. Gross and microscopic examination of a variety of
tissues and organs did not show any treatment-related findings.
Following the 13-week dosage period, 5 males and 5 females per
group were allowed to recover for 4 weeks. No evidence of
hyperexcitability was observed among the rats; body weight gain was
slightly higher in the treated groups than in the controls. The
no-observed-effect level was 1 mg/kg body weight (Hunter et al.,
1977).
Four groups of CD rats (8 of each sex per group) were exposed to
aerosolized deltamethrin (technical grade powder) for 6 h per day, 5
days a week, for 2 weeks, and for 4 days during a third week. Mean
aerosol concentrations were 3, 9.6, and 56.3 mg a.i./m3 with
about 87% of respirable particles (diameter lower than 5.5 µm). No
rats died as a result of exposure. Signs of irritation (agitated
grooming and ptyalism due to the powder were noted in all groups
during exposure, with more pronounced toxic signs (ataxia and
walking with arched backs) in the group receiving the highest dose
tested. Male rats also showed a reduced body weight (-5%) in all
groups. An elevation of the serum sodium ion content was noted at
the two highest doses. No increased incidence of any particular
lesion was observed in the high-dose group compared with the control
group. Irritation and weight loss were only slight at 3 mg/m3 and
this can be considered as a no-effect level (Coombs et al., 1978).
7.3.2 Dog
Male and female beagle dogs (3 - 5/sex per group), 25 weeks of
age, received a daily oral dose of 0, 0.1, 1, 2.5, or 10 mg
deltamethrin/kg body weight in PEG 200 in gelatin capsules over 13
weeks. All treated groups showed reduced body weight gain, but
this was not dose-related. Liquid faeces were associated with all
groups of treated dogs throughout the dosing period. Dilatation of
the pupils was seen in dogs receiving 2.5 and 10 mg/kg per day.
The sign was first seen 4 - 7 h after dosing and persisted
throughout the day. The incidence of vomiting increased dose-
dependently in all treated groups, except the group receiving
0.1 mg/kg. In the highest dose group, unsteadiness, body tremors,
and jerking movements were seen, particularly in males, in weeks 2,
3, and 4. Excessive salivation was seen initially and diminished
during the dosing period. After 5 and 12 weeks, depression of the
gag reflex was noted in a proportion of animals in all treated
groups. However, this was not considered to be of toxicological
significance. Exaggeration or depression of the patellar reflex
was observed in some animals in all treated groups after 5 and 12
weeks, mainly at 1, 2.5, or 10 mg/kg per day. Some animals in all
treated groups showed depression of the flexor reflex. Dose levels
of 2.5 and 10 mg deltamethrin/kg per day caused modification of the
EEG pattern in some animals, 12 weeks following administration.
Histopathological evaluations of tissues and organs, including the
nervous system and muscle tissue, did not reveal any abnormalities
that could be related to the administration of the compound.
During recovery, the gag reflex continued to be depressed, whereas
exaggeration of the patellar reflex was still seen in some dogs
that had previously received 1 mg/kg per day (Chesterman et al.,
1977).
7.4 Long-Term Exposure and Carcinogenicity
7.4.1 Mouse and rat
Male and female Charles River CD-1 mice (80 of each sex per
group) were fed dietary levels of deltamethrin of 0, 1, 5, 25, or
100 mg/kg, daily, for 24 months. There were no clear effects
related to the administration of deltamethrin on general behaviour,
mortality, body weight, and food consumption. Blood chemistry,
haematology, and urine analysis parameters were normal after 12, 18,
and 24 months (at the times of interim and terminal sacrifice).
Microscopic examination of tissues did not reveal any lesions
indicative of a compound-related effect. The tumour incidence was
unaffected by deltamethrin administration. The no-observed-effect
level was 100 mg/kg diet (Goldenthal et al., 1980a).
Deltamethrin was administered, by gavage, to C57BL/6 mice at 4
dose levels (0, 1, 4, and 8 mg/kg body weight) and to BDVI rats at
3 dose levels (0, 3, and 6 mg/kg body weight) on 5 days a week for
104 weeks. After completion of the treatment, the animals were
observed until 120 weeks of age, when all survivors were killed.
The treatment had a slight effect on body growth and survival
rates, especially in the groups of mice and rats treated with the
highest dose. In C57BL/6 mice, various types of tumours were
observed in all treated groups. An increased incidence of
lymphomas was observed in mice receiving deltamethrin at levels of
1 and 4 mg/kg body weight, but not in the group treated with
8 mg/kg body weight. No significant difference in the incidences
of lung adenomas, liver-cell tumours, or other tumours was observed
in treated groups compared with controls. In BDVI rats, an
increased incidence of pituitary, thyroid, and mammary tumours was
noted; however, no clear dose-response relationship was shown
(Cabral et al., 1986). The details of this study were not
available for evaluation.
Male and female Charles River CD rats (90 of each sex per
group) were fed with 0, 2, 20, or 50 mg deltamethrin/kg diet for
2 years. A second control group (60 of each sex) was also used.
Interim sacrifices (10 of each sex per group excluding control
group 2) were made after 6, 12, and 18 months. No changes in
general behaviour and appearance were observed in relation to
treatment. Survival rate was similar for control and treated rats
(50 - 67%). Rats in the 50 mg/kg group gained slightly less weight
than control rats, whereas the food consumption was essentially the
same. Ophthalmoscopic findings were generally similar for control
and treated rats. No haematological and biochemical parameters
were changed in a biologically significant way in relation to
treatment at any time, except for a decrease in SGPT (serum
glutamic pyruvic transaminase) activity at 6 months in the mid- and
high-dose groups. Organ weights were not affected. The
macroscopic and microscopic findings were common for the species
and the strain, except for a slightly increased incidence of axonal
degeneration in sciatic, tibial, and/or plantar nerves in the 20
and 50 mg/kg groups at 18 months, but not at termination. Thus,
this was not considered to be indicative of a compound-related
effect. The incidence of benign testicular tumours (interstitial
cell adenomas) at terminal sacrifice in this study was: control
group 1, 0/37; control group 2, 4/35; low-dose group 1/38; mid-dose
group 1/30; high-dose group 6/38. The incidence seen in the high-
dose group was considered to be spontaneous, because it was not
significantly higher than in the second control group or in
historical control groups (Goldenthal et al., 1980b; Richter &
Goldenthal, 1983).
7.4.2 Dog
Deltamethrin dissolved in maize oil was administered in the diet
to 64 beagle dogs (8 of each sex per group) at levels of 0, 1, 10,
and 40 mg/kg for 24 months. This corresponds to 0, 0.025, 0.25, and
1 mg/kg body weight, respectively. Individual body weights and food
consumption values were determined weekly. Ophthalmoscopic,
haematological, biochemical, and urinalysis examinations were
conducted during the pre-test period and at 6, 12, 18, and 24 months
of the study. Neurological examinations were conducted at
approximately 1 year and before termination. No signs of overt
toxicity were observed in any of the dogs. Body weight and food
consumption values were similar for control and treated dogs. No
compound-related effects were observed during the ophthalmoscopic
and physical examinations. Although there were some random
statistically significant differences between the control and
other dose groups in the haematological and biochemical tests,
physiologically significant changes were not observed at any
interval in the study. Two treated and two control animals died
during the study. No compound-related gross or microscopic changes
were observed in the surviving dogs that were sacrificed and
necropsied. Inflammatory, degenerative, and proliferative changes
described were spontaneous in nature, or related to the estrous
phase of the menstrual cycle, and unrelated to compound
administration. On the basis of this study, it has been concluded
that the no-observed-effect level is 40 mg/kg diet (equivalent to
1 mg/kg body weight per day) (IRDC, 1980).
7.5 Mutagenicity
7.5.1 Microorganisms
DNA repair tests in Escherichia coli were conducted at dose
levels of 1250, 2500, or 5000 µg deltamethrin/ml. Deltamethrin was
dissolved in dimethyl sulfoxide (DMSO) and 0.1 ml of the solution
was spread on a plate. Growth inhibition was compared between DNA
repair deficient mutants (p3478 and CM611) and wild types (W3110
and WP2). Partial precipitation of deltamethrin from the solution
occurred when it came into contact with the aqueous bacterial
growth medium. Deltamethrin did not have any damaging effects on
DNA (Peyre et al., 1980).
Deltamethrin was examined for its mutagenic potential in the
Ames test with 5 strains of Salmonella typhimurium (TA 1535, TA
1537, TA 1538, TA 98, and TA100) at doses of 2, 10, 50, 200, 500,
1000, or 5000 µg/plate, with and without S-9 mix (metabolic enzyme
system). It was dissolved in DMSO and precipitated out of solution
at concentrations of 200 µg/plate or more. Deltamethrin did not
have any effect on the mutation rate in any of the strains at any
of the concentrations tested (Peyre et al., 1980).
A similar Ames test was carried out at 0.2, 2, 20, 200, or
400 µg deltamethrin/plate with microsome enzymes. The compounds
did not influence the number of revertants of the 5 strains (same
as above) of S. typhimurium. Again, deltamethrin was dissolved in
DMSO and precipitated out of solution at 200 µg/plate or more
(Fouillet, 1976).
Kavlock et al. (1979) found deltamethrin not to be mutagenic in
2 assays with S. typhimurium at doses of 0 - 1000 µg/plate in DMSO,
with or without metabolic activation. They also obtained negative
results with E. coli at 10 - 1000 µg/plate as well as with
Saccharomyces cerevisiae at concentrations of 1 - 5%, in both cases
with and without metabolic activation. Deltamethrin was found not
to be mutagenic in S. typhimurium strains TA100 and TA98, in the
presence or absence of a rat liver activation system, using the
plate incorporation assay and fluctuation tests. The compound,
dissolved in DMSO, precipitated out of solution at 600 µg/plate
(Pluijmen et al., 1984).
7.5.2 Cultured cells
Deltamethrin, dissolved in a mixture of cremaphor oil and
ethanol (1:1), was applied to a culture of Chinese hamster ovary
cells (CHO) at levels of 0.04, 0.2, 1.0, or 5.0 mmol/litre, with or
without metabolic activation, and examined for chromosomal
aberrations and sister chromatid exchanges (SCE). Because of the
cytotoxic effect of cremaphor oil when combined with S-9 mix or
deltamethrin, no cells would grow in the control dish with
activation, or in the 5 mmol/litre deltamethrin dishes, either with
or without metabolic activation. A high incidence of chromosomal
aberrations and SCEs was observed in the dishes containing 1 mmol
deltamethrin/litre, with activation. However, the absence of
control values (both with and without activation) because of a
broken test tube, made the interpretation equivocal. A second
study was conducted in which deltamethrin was dissolved in DMSO and
applied to the cells at levels of 0.001, 0.01, 0.1, or 0.2
mmol/litre, with or without metabolic activation. In this study,
deltamethrin did not produce any cytotoxic effects and did not
induce either chromosomal aberrations or SCEs in CHO cells.
However, no positive controls were tested and only single plates
were prepared per dose level (Sobels et al., 1978).
Deltamethrin was found not to be mutagenic in V79 Chinese
hamster cells, in the presence or absence of hepatocytes. It is
not known which solvent was used (Pluijmen et al., 1984).
7.5.3 Mouse
An in vivo cytogenetic test was conducted on mice (3 males and
3 females per group). Mice were treated orally with deltamethrin
in sesame oil for 2 consecutive days at 5 or 10 mg/kg body weight.
The incidence of chromosomal aberrations in bone marrow cells or
micronuclei in the polychromatic erythrocytes of treated groups
was, however, comparable to that of the control groups. No
positive controls were tested (Sobels et al., 1978).
Deltamethrin was applied orally, once, at 15 mg/kg body weight
to Swiss mice. A time-related effect on the chromosomes in bone
marrow cells was observed by killing 2 animals every 3 h during
24 h. The report stated that the incidences of chromated
aberrations were low and that there were no consistent time-related
trends in the distribution of the aberrations. However, the time-
related trend of aberrations was not reported. Again, no positive
controls were tested (Sobels et al., 1978).
A dominant-lethal assay with deltamethrin was performed.
Groups of 9 - 13 male mice were dosed orally at 3 mg/kg body weight
in sesame oil for 7 days or at a single dose of 6 or 15 mg/kg body
weight in sesame oil, and mated with 6 - 18 non-treated females.
There were no effects on the rates of pre- and post-implantation
losses, while the positive control, triethylene triphosphoramide
(10 mg/kg body weight), reduced pregnancies in the second and third
weeks after treatment and increased embryonal losses (Vannier &
Glomot, 1977).
Deltamethrin in olive oil was administered orally to female
Swiss mice at single or repeated (5 times at daily intervals) doses
of 1.36, 3.4, or 6.8 mg/kg per day. Bone marrow smears were
prepared 6, 24, or 48 h after treatment. No mutagenic activity was
observed with deltamethrin, whereas the positive control,
cyclophosphamide, induced a positive response (Polàkovà & Vargovà,
1983).
In a micronucleus test, a single dose of deltamethrin in corn
oil was administered orally at 16 mg/kg body weight to Swiss CD-1
mice (5 of each sex per group). No mutagenic activity was observed
with deltamethrin, whereas the positive controls,
triethylenemelamine and dimethylbenzanthracene, both induced
positive responses (Vannier & Fournex, 1983).
7.5.4 Appraisal
Deltamethrin is not mutagenic or clastogenic in a variety of in
vitro and in vivo test systems.
7.6 Teratological and Reproductive Effects
7.6.1 Teratology
7.6.1.1 Mouse
Deltamethrin was dissolved in corn oil and administered by
gastric intubation at doses of 0, 3.0, 6.0, or 12.0 mg/kg body
weight on days 7 - 16 of gestation to groups of CD-1 mice. Mice
were sacrificed on day 18 of gestation. There was a dose-related
( P <0.001) reduction in maternal weight gain during pregnancy and
high-dose females gained 58% less weight than the controls. There
was no dose-related mortality but dams in the high- and mid-dose
groups became convulsive after dosing. Treatment did not affect
the number of implantation sites, fetal mortality, fetal weights,
or the number of sternal and caudal ossification centres.
A significant ( P <0.01) dose-related increase in the
occurrence of supernumerary ribs was observed. No other dose-
related skeletal or visceral anomalies were observed (Kavlock et
al., 1979).
Pregnant female Swiss CD-1 SPF mice (24 per group) were given
deltamethrin dissolved in sesame oil by oral intubation at dose-
levels of 0, 0.1, 1, or 10 mg/kg body weight per day on days
6 - 17 of pregnancy. The animals were necropsied on day 18 of
pregnancy. The numbers of implantation sites, fetal losses, and
viable fetuses were not affected by treatment. There was a dose-
related decrease in mean fetal weight. Apart from delayed
ossification at all dose levels, skeletal examination revealed no
abnormalities. A teratogenic effect was not observed (Glomot &
Vannier, 1977).
In a complementary teratology study, pregnant female Swiss CD-1
mice were given deltamethrin dissolved in sesame oil by oral
intubation at 0, 0.1, 1, or 10 mg/kg body weight per day from day 6
to day 17 of gestation. Females were either sacrificed on day 18
of gestation or allowed to litter for subsequent examination of
pups on days 1 or 21 of lactation. The compound caused a moderate
and transient retardation of development of the fetus at the 1 and
10 mg/kg body weight dose rate, but these effects were not observed
on days 1 or 21 post-partum. There were no teratogenic effects
related to treatment (Vannier & Glomot, 1982).
7.6.1.2 Rat
Pregnant female Sprague-Dawley rats (24 per group) received 0,
0.1, 1, or 10 mg deltamethrin/kg body weight per day by oral
intubation on days 6 - 18 of pregnancy. Apart from 12 females in
the control and 10 mg/kg groups, which were allowed to deliver, the
dams were sacrificed and examined on day 21. There were no effects
on reproduction or on the teratogenic parameters examined, except
for slightly delayed ossification at the highest dose level (Glomot
& Vannier, 1977).
Deltamethrin was dissolved in corn oil and administered by
gastric intubation at doses of 0, 1.25, 2.5, or 5.0 mg/kg body
weight on days 7 - 20 of gestation. Rats were sacrificed on day 21
of gestation. There was a dose-related reduction ( P <0.01) in
maternal weight gain during pregnancy, and dams in the high-dose
group gained only 80% of the control value. Treatment did not
affect the number of implantation sites, fetal mortality, fetal
weight, or the number of sternal and caudal ossification centres
(Kavlock et al., 1979).
7.6.1.3 Rabbit
Groups of 15 pregnant New Zealand White rabbits received
deltamethrin dissolved in sesame oil at levels of 0, 1, 4, or
16 mg/kg body weight per day during days 6 - 19 of pregnancy.
Examination was carried out on day 28 of gestation. The mean fetal
loss was not dose-related. The mean fetal weight in the highest-
dose group was decreased. Some malformations (hydrocephaly,
exencephaly, and thoracogastroschisis) were observed in 2 fetuses
of animals at the highest dose level. In a supplementary study,
pregnant rabbits were similarly dosed with 16 mg/kg body weight per
day; one fetus with spina bifida and shortened tail was detected
among 69 apparently normal fetuses. Malformations were within the
normal limits of the strain used and were not considered to be
related to the treatment, despite the occurrence at the highest
dose level only (Glomot & Vannier, 1977, 1978).
7.6.2 Reproduction studies
Groups of 10 male and 20 female Charles River rats were fed
deltamethrin in the diet at 0, 2, 20, or 50 mg/kg and mated to
begin a 3-generation, 2-litter (first generation, 3 litter)
standard reproduction study. Parental body weights and food
consumption were recorded during the study. After weaning of the
second litter, the surviving parent rats were sacrificed and
necropsied. Five male and 5 female pups of the F3b generation were
necropsied. No changes relevant to treatment were observed in
general behaviour or survival of parent rats or pups. The body
weight of F0 males of the 50 mg/kg group was decreased from week 11
onwards. There were some slight decreases in mean food consumption
of F1 male parent rats in the 50 mg/kg group. The basic
reproduction indices (fertility, gestation, lactation, viability,
and litter size) were not affected by the treatment. However, the
mean pup weight in some litters, especially in the 50 mg/kg group,
was slightly decreased in comparison to the controls on day 21 of
lactation. Gross external examination did not reveal any
abnormalities. No gross or microscopic lesions of treatment-
related significance or significant effects on the organ weights of
the F3b generation were observed (Wrenn et al., 1980).
Deltamethrin was dissolved in corn oil and administered by
gastric intubation at doses of 0, 2.5, or 5.0 mg/kg body weight to
Sprague-Dawley rats from day 7 of gestation to day 15 of lactation.
The dams were allowed to litter and rear their young: litters were
reduced at birth to 4 males and 4 females per litter. The pups
were weighed weekly and examined for the development of eye-
opening, startle reflex, and air-righting. The litters were weaned
on day 22 post-partum and the males discarded. Weekly weighing of
the females continued and at 6 weeks of age they were tested in a
circular open-field. There were no effects on parturition, litter
size, or pup viability. Weights at birth were similar for all
groups, but a dose-related depression in growth was observed during
the pre-weaning period. This early diminution in pre-weaning
weight appeared to have little effect on the morphological and
behavioural parameters measured (Kavlock et al., 1979).
7.7 Neurotoxicity and Behavioural Effects
Adult hens (10 per group) were gavaged with a single dose of 0,
500, 1250, or 5000 mg deltamethrin/kg body weight suspended in corn
oil or 0 or 100 mg/kg body weight dissolved in sesame oil. During
21 days, observations were made on mortality, health, neurotoxic
signs, and body weight. Deltamethrin did not induce any clinical,
macroscopic, or histological signs of delayed neurotoxicity (Ross
et al., 1978).
Groups of 5 male and 5 female Wistar rats were administered
25 mg deltamethrin/kg body weight in 10 mg corn oil/kg on 2
consecutive days. Controls received 10 mg corn oil/kg body weight.
A tilting plane test was performed every second day from day 4 to
day 16 of the study. Two male rats died at 25 mg/kg. No effect
was found on the slip-angle (Davies et al., 1983).
The effects of deltamethrin were studied in a rat performance
test that arranged for milk delivery after every fortieth lever
press. Deltamethrin (1 - 8 mg/kg body weight, given orally, 2 h
before the test) produced both dose-related increases in pause
duration and decreases in response rate. Deltamethrin was also
studied using a conditional flavour-aversion test. Deltamethrin-
treated, trained rats displayed an aversion to saccharin that was
greatest at 2 mg/kg (Macphail, 1981).
The neurological effects of the 4 synthetic pyrethroids,
resmethrin, permethrin, cypermethrin, and deltamethrin, have been
investigated in the rat to establish whether there is a correlation
between the clinical-functional status of the animal and peripheral
nerve damage, as measured biochemically (Rose & Dewar, 1983).
Neuromuscular dysfunction was assessed by means of the inclined
plane test and peripheral nerve damage by reference to
beta-glucuronidase and beta-galactosidase activity increases in
nerve tissue homogenates from treated and control animals. A
transient functional impairment was found in animals treated with
any one of the 4 pyrethroids tested and in all cases this was
greatest at the end of the 7-day dosing regimen (deltamethrin doses
of 5 - 20 mg/kg per day in arachis oil). Significant increases in
beta-glucuronidase and beta-galactosidase activities were found
3 - 4 weeks after the start of dosing, in the distal portion of the
sciatic/posterior tibial nerves from permethrin-, cypermethrin-,
and deltamethrin-treated animals, but no changes were found in
resmethrin-treated animals. It is concluded, therefore, that there
is no direct correlation between the time-course of the
neuromuscular dysfunction and the neurobiochemical changes. This
suggests that these pyrethroids have at least two distinct actions-
a short-term pharmacological effect at near-lethal dose levels and
a more long-term neurotoxic effect that results in sparse axonal
nerve damage.
To better characterize the behavioural toxicity of pyrethroid
insecticides, comparisons were made of the effects of cismethrin
and deltamethrin exposure on motor activity and the acoustic
startle response in male Long-Evans rats (Crofton & Reiter, 1984).
Acute dose-effect, acute time-course, and 30-day repeated-exposure
determinations of 1-h motor activity were made using figure-eight
mazes. The acoustic startle response was measured to a 13-kHz,
120-dB(A), 40-millisecond tone at each of 3 background white noise
levels (50, 65, and 80 dB). Deltamethrin (0, 2, 4, 6, or 8 mg/kg
body weight) or cismethrin (0, 6, 12, 18, or 24 mg/kg) were
administered orally in 0.2 ml/kg corn oil. Cismethrin and
deltamethrin produced similar dose-dependent decreases in motor
activity. The time course of onset and recovery for this decreased
activity was rapid (1 - 4 h). No cumulative effects on motor
activity of a 30-day exposure to 2 mg deltamethrin/kg per day or
6 mg cismethrin/kg per day were found. The effects of cismethrin
and deltamethrin on the acoustic startle response were dissimilar:
deltamethrin produced a dose-dependent decrease in amplitude and an
increase in latency, and cismethrin produced an increase in
amplitude and no change in latency. The differential effects of
cismethrin (Type I pyrethroids) and deltamethrin (Type II
pyrethroids) on the acoustic startle response may be related to the
contrasting effects previously shown with neurophysiological and/or
neurochemical techniques (see Appendix I).
7.8 Miscellaneous Effects
Analgesic effects of deltamethrin for thermic (hot plate test,
60 °C) and mechanical stimuli were investigated in mice and rats,
respectively. Deltamethrin prolonged the response-time to these
tests. Although this action was not significant at 500 mg
deltamethrin/kg body weight given orally, the reaction time was
increased at 1000 and 1500 mg/kg given orally in aqueous suspension
with 10% gum arabic (Chanh et al., 1981).
In rats, treatment with deltamethrin increased mean arterial
pressure and aortic output (Forshaw & Bradbury, 1983). The
cardiovascular effects of deltamethrin were due to both increased
catecholamine release in peripheral vascular beds, and to a direct
positive inotropic effect on the heart.
Krasnjih & Pavlova (1985) demonstrated induction of microsomal
oxygenases in rats 20 h after a single administration of 1/2 LD50.
Daily administration of 1/10th LD50 for 2 months reduced
acetylcholinesterase activity in the serum, erythrocytes, liver,
and cerebrum. It also led to some changes in the aspartate
aminotransferase activity and the urea and protein contents of
serum.
When rats were dosed orally with a single dose of 1/2 LD50 or 3
daily doses of 1/5 LD50 deltamethrin, the activities of transferrin
and ceruloplasmin in plasma, 20 h after dosing, were unchanged.
After the single dose, microsomal monooxygenase activity was
increased by 87%, and after the 3 doses, it was increased by 290%
(Kagan et al., 1986).
7.9 Potentiation
Deltamethrin was hydrolysed in vitro by esterases in blood,
brain, kidney, liver, and stomach preparations of mice.
Pretreatment of mice with the oxidase inhibitor, piperonyl butoxide
(PBO), or the esterase inhibitor, S,S,S-tributylphosphorotrithioate
(DEF), delayed metabolism of intraperitoneally administered
deltamethrin. PBO or DEF made mice more sensitive to deltamethrin
(Ruzo et al., 1979).
Plasma esterases, in addition to hepatic esterases, play a role
in the metabolism of deltamethrin in mammals and cause its rapid
detoxification by the oral route. In a potentiation study, a range
of esterase inhibitors, consisting mainly of organophosphorus
insecticides, was given to male rats in oral doses that inhibited
50% of the plasma cholinesterase. After 15 min, or 2 or 24 h, an
oral LD50 dose of deltamethrin EC formulation was given, which
showed potentiation with azinphos ethyl, omethoate, and dichlorvos.
It appears that users must handle deltamethrin in these
combinations very carefully because of their high toxicity.
Acephate, monocrotophos, phosphamidon, parathion methyl, and the 2
controls did not act as potentiators (Audegond et al., 1988).
7.10 Mechanism of Toxicity (Mode of Action)
Deltamethrin is classified as a Type II pyrethroid. For the
mode of action of pyrethroids in general see Appendix I.
The lowest concentration of deltamethrin to have an effect in
crayfish stretch receptor neurones on sodium channels was 10-12
mol/litre, but the response of the preparation to gamma-
aminobutyric acid (GABA) appeared to be unaffected by concentrations
of deltamethrin up to 10-7 mol/litre. Although 10-6 mol/litre
deltamethrin had a slight effect on the GABA response of the dactyl
abductor muscle, it appears that the majority of the effects of
cyano-pyrethroids in invertebrates could be accounted for solely by
their action on sodium channels (Chalmers et al., 1987).
Pyrethroid-induced motor symptoms, i.e., deltamethrin-induced
writhing and cismethrin-induced tremor, were studied, using a
number of pharmacological agents, in intact conscious rats and
spinal rats. The results suggest that pyrethroid-induced motor
symptoms, i.e., writhing and tremor, are mediated via a spinal site
of action, probably involving interneurones. Deltamethrin-induced
"non-motor" symptoms, i.e., increase in brain blood flow and blood
glucose may result from a supraspinal component of deltamethrin
activity. In contrast, the cardiovascular effects of deltamethrin
are mediated via a peripheral site of action (Bradbury et al.,
1983).
Tissue culture experiments have shown that the dorsal root
ganglion is more sensitive to deltamethrin than the spinal cord or
peripheral nerve fibres. The morphological alterations observed in
the neuronal bodies of the ganglia may reflect some perturbation of
the ionic equilibrium (Na+ and Ca+) (Souyri, 1985).
The results of several other, sometimes very detailed and
specialized, studies on the mode of action of deltamethrin have
been reported. Because these results do not basically influence
the present evaluation of deltamethrin, they are not reviewed in
detail. The interested reader is referred to the following
publications and a review by Bidet et al. (1988): Aldridge et al.
(1978), Duclohier & Georgescauld (1979), Gray et al. (1980),
Jacques et al. (1980), Miller & Adams (1980), Gammon et al. (1981),
Pichon (1981), Brodie & Aldridge (1982), Dyball (1982), Parkin &
LeQuesne (1982), Ray (1982), Staatz et al. (1982), Brodie (1983),
Takahashi & LeQuesne (1983), Berlin et al. (1984), Prasada Rao et
al. (1984), Bloomquist & Soderlund (1985), Brodie (1985), Brodie &
Opacka (1985), Bloomquist et al. (1986), Chinn & Narahashi (1986),
Doherty et al. (1986), Forshaw & Ray (1986), Staatz-Benson & Hosko
(1986), Brooks & Clark (1987), Forshaw et al. (1987), Leibowitz et
al. (1987), Lummis et al. (1987), Stein et al. (1987).
7.11 Experimental Studies on Antidotes
Treatments capable of counteracting acute deltamethrin
poisoning have been investigated in experimental animals. Ray &
Cremer (1979) have proposed atropine, Gammon et al. (1982),
phenobarbital and diazepam, and Bradbury et al. (1981, 1983),
mephenesin.
In order to find a therapeutically usable antagonist,
pharmacological screening was carried out by Dumont (1978), Dumont
& Chifflot (1978), and Dumont & Laurent (1979). The outcome was
that barbiturates are therapeutically active, but that the most
efficient product is ethyl carbamate. Cotonat et al. (1987) and
Fournier (1988) have confirmed that ethyl carbamate is an effective
treatment for severe deltamethrin poisoning. However, a serious
drawback is its antimitotic activity. Leclercq et al. (1986) began
by evaluating the activity of common anticonvulsants, such as
diazepam and clomethiazole. These products exhibited satisfactory
activity in rats and dogs (Thiebault et al., 1985, 1988).
Phenoprobamate and mephenesin carbamate have been shown to be
effective in the experimental treatment of deltamethrin poisoning
(Cotonat et al., 1987; Leclercq et al., 1986). A summary of the
results of the antidote studies can be found in Bleys et al.
(1986). Clinical trials based on these studies will be undertaken
(unpublished information given to the IPCS by Roussel Uclaf).
The therapeutic effects of methocarbamol have also been shown
by Hiromori et al. (1986). However, the mechanism underlying this
activity is not clear as the actual anticonvulsant activity is
weak.
It appears that for the time being barbiturates, and especially
diazepam, offer the safest symptomatic treatment in case of
deltamethrin poisoning. Advice on treatment for deltamethrin
poisoning is given in the IPCS Deltamethrin health and safety guide
(WHO, 1989).
8. EFFECTS ON MAN
8.1 General Population-Poisoning Incidents
A few cases of attempted suicides with deltamethrin
formulations (mainly EC), all non-fatal, have been reported in
anti-poison centres. Two typical cases are described below.
The first poisoning case in a 13-year-old girl who ingested
voluntarily 200 ml of a 2.5% EC formulation, (5 grams of
deltamethrin) was described by Rousselin (1983). After an unknown
time, she lost consciousness and developed generalized muscle
cramps, myosis, and tachycardia. Treatment in hospital was as
follows: gastric lavage, PAM 0.5 mg; atropine 2 mg; sodium
nitrite, 3% sodium thiosulfate; and, lastly, high doses of
diazepam. She completely recovered in 48 h.
A second poisoning case, concerning another attempted suicide
by a 23-year-old man, was reported by Foulhoux (1988). After oral
absorption of 70 cc of a 2.5% EC formulation (1.75 g pure
deltamethrin), there were no neurological signs in this patient.
Digestive and hepatic signs occurred, probably due to absorption of
the solvent, since determination of xylene in plasma was positive.
The patient was treated with haemodialysis, phenobarbital,
lidocaine, and provoked alkaline diuresis. Recovery followed
within 48 h.
8.2 Occupational Exposure
8.2.1 Acute toxicity-poisoning incidents
Rousselin (1983) described a case of poisoning in an
agricultural worker as a result of skin contamination with a liquid
containing 5 g deltamethrin/litre. He developed paraesthesia in
the legs, mouth, and tongue, and diarrhoea. Following washing of
the skin and administration of antihistamines, he still had
tingling sensations in his toes after 24 h, but was fully recovered
after 48 h.
Outbreaks of acute deltamethrin and fenvalerate poisoning
occurred in cotton growers in China in 1982 - 84. The farmers
handled the pyrethroid insecticides without taking any precautions.
Skin sensations occurred in more than 90% of the exposed workers.
After repeated spraying in the cotton fields, the mild cases
presented severe headaches, dizziness, fatigue, nausea, and
anorexia, with transient changes in the EEG. A severe case
developed muscular fasciculation, repetitive discharges in the EMG,
and frequent convulsions, which were treated with diazepam and
phenobarbital. However, in follow-up studies, all workers were
found to have made complete recovery, and the prognosis of acute
pyrethroid poisoning was found to be good (He, 1987; Tong Ying,
1988).
More recently He et al. (1989) reviewed 573 cases of acute
pyrethroid poisoning reported in the Chinese medical literature
during 1983 - 88. Among these there were 325 cases of acute
deltamethrin poisoning: 158 occupational, due to inappropriate
handling, and 167 accidental, mostly due to ingestion. Two
patients died of convulsions. All others recovered with
symptomatic and supportive treatment within 1 - 6 days. Clinical
manifestations are well reviewed (He, 1987).
8.2.2 Effects of short- and long-term exposure
Among plant workers dermally exposed to technical deltamethrin
or its formulations, cutaneous and mucuous manifestations were
observed. Initial lesions were tenacious and painful pruritus,
especially observed after exposure to hot water or perspiration,
followed by a blotchy local burning sensation with blotchy erythema
for about 2 days. Thereafter, slight and regular desquamation,
restricted to the contaminated area, occurred. Cutaneous signs
were sometimes accompanied by itching of the face (mainly around
the mouth) and/or rhinorrhoea or lachrymation (Husson, 1978).
Apart from the above-mentioned effects, no long-term or
persistent effect, or allergic diseases were reported in 70
workers, who had been exposed from 1977 - 87 in a deltamethrin-
manufacturing and -formulating plant in France (unpublished
Roussel Uclaf information supplied to the IPCS, 1988).
A field study was carried out in the United Kingdom with three
unprotected operators and one operator wearing hood, gloves, and
respirator, all of whom were involved in orchard spraying with
deltamethrin according to normal field practice. The exposure time
was 3.5 h. No changes were found in blood cell counts, total
protein, urea, alkaline phosphates, gamma-GT and SGOT in blood.
Little deltamethrin was found in the respirator pad and no residues
were found in the urine. There was no decrease in nerve conduction
velocity, but a slight tendency to the opposite reaction. None of
the operators experienced facial sensations (Hewson & Burgess, 1981).
Four operators were evaluated by the same authors during normal
field applications of deltamethrin lasting one day. Three of the
operators did not wear any protection on their heads or hands,
while one wore hood, gloves, and a respirator. Motor and sensory
nerve conduction velocities were determined as well as
haematological and biochemical parameters and urinalysis. No
changes were observed in the blood parameters measured and no
residues were found in the urine samples. Furthermore, nerve
conduction velocity did not decrease. Residues were primarily
confined to gloves and legs. None of the operators experienced
facial sensations (Hewson & Burgess, 1981).
Persons exposed to deltamethrin for 7 - 8 years in production
and formulation were subjected to clinical and haematological
examinations. Evaluations were conducted at several plants. There
were no measurable effects other than transient irritation of
cutaneous and mucous membranes, which was without sequelae.
Adequate precautionary measures, such as the wearing of gloves and
face masks, provided protection from exposure (Foulhoux, 1981).
A medical survey of agricultural workers involved in the use
and application of EC and WP formulations of deltamethrin in
Yugoslavia revealed no untoward symptoms of exposure, other than
itching and burning of the face, and nasal hypersecretion. Medical
examinations included chest X-ray, ECG, liver function tests,
neurological examinations (eye tonometry, Goldman perimetry, dark
adaptation ability), kidney function tests, and whole blood and
plasma cholinesterase activity. No adverse effects were noted.
The need for the proper use of masks and gloves, as well as good
personal hygiene (e.g., washing), was emphasized (Plestina, 1981).
Five healthy volunteers, 16 - 40 years of age, were exposed to
deltamethrin during 5 days of spraying in a cotton field in India
in 1981. A sixth volunteer was engaged in mixing and loading the
emulsion during the same period. Spraymen were exposed for 7 h
daily. No one complained about any symptoms. No clinical
abnormalities were detected, particularly with respect to
neurological examination (muscle power, coordination, tremors,
reflexes, and both light and deep sensations). No cardiovascular,
respiratory, or abdominal abnormalities were detected, and no skin,
mucous membrane, or eye lesions were observed during, and after
cessation of, exposure (Trivedi, 1981).
A health survey was carried out among spraymen exposed to 2.5%
deltamethrin emulsifiable concentrate in cotton fields in China.
The subjects were exposed to deltamethrin at concentrations of
0.022 - 24.070 µg/m3 in the air of the respiratory zone and
0.013 - 0.347 µg/cm2 of skin contact. One half of the 44 sprayers
complained of itching and burning sensations on their faces. A few
miliary red papules also appeared on the face of one of them, but
no signs of acute deltamethrin poisoning were noticed during
physical examination. There were no significant differences in the
sodium, potassium, and urea contents of the serum, the sodium,
potassium, ATPase, and serotonin contents of whole blood, and the
levels of 3-methyl-4-hydroxymandelic acid and 5-hydroxy-
indoleacetic acid in the urine between the subjects examined and
the controls. Deltamethrin in the urine of spraymen was below the
detection limit of 0.10 µg/litre (Wang et al., 1988).
Mestres et al. (1985) measured the dermal and inhalation
exposure of mixer/applicators who applied deltamethrin to vegetable
crops in greenhouses in southern France and of workers who picked
fruit from treated trees in the same area. This appeared to be
less than 0.0065% of the toxic dose per hour with a mask and
0.0017%, without a mask.
In a department producing an aerosol of the domestic Bulgarian
insecticide "Dekazol" containing 0.02, 0.04, or 0.08% deltamethrin,
severe subjective complaints of sensory irritation were found
because of the high levels of contamination of the workplace air
with deltamethrin and also dermal contamination. Skin irritation
with conjunctivitis and irritation of the respiratory system were
discovered in all 25 workers. Two of them had contact urticaria.
Patch testing with 0.03% deltamethrin showed a positive reaction in
5 out of 23 workers tested (Bainova et al., 1986).
8.3 Clinical Studies
Three formulations of deltamethrin in petroleum solvent were
patch tested on 37 human volunteers (double blind trial against
solvent control). A dose of 20 µl of a 1% suspension in water, of
a 25 g/litre emulsifiable concentrate was put on the facial skin of
each volunteer, with a randomized distribution of control and
active dilution. The duration of the irritation was short (from
some minutes to 1 h) and the severity was described as slight by
most of the volunteers. No skin damage was reported (Foulhoux et
al., 1981).
9. PREVIOUS EVALUATIONS BY INTERNATIONAL BODIES
The Joint FAO/WHO Meeting on Pesticide Residues (JMPR)
discussed and evaluated deltamethrin at its meetings in 1980, 1981,
1982, 1984, 1985, 1986, 1987, and 1988 (FAO/WHO, 1981, 1982, 1983,
1985a, 1986a, 1986b, 1988a,b,c). In 1982, an acceptable daily
intake (ADI) of 0 - 0.01 mg/kg body weight was established.
The following Maximum Residue Limits (MRLs), in mg/kg, resulted
from these evaluations:
tea 10.0
hops dry, wheat bran unprocesseda 5.0
coffee beans (post-harvest) 2.0
wheat wholemeal,a cereal grains,a 1.0
(ph) lentil (dry),a beans (dry),a
field pea (dry)a
straw and fodder (dry) of cereal 0.5
grains, legume animal feeds (dry
weight), leafy vegetables
brassica leafy vegetables,a edible 0.2
peel of fruiting vegetablesa
bulb vegetables, edible peel of 0.1
assorted fruits, legume vegetables,
oilseeds, pome fruits, wheat floura
artichokes, bananas, clementines, 0.05
coco beans, grapes, kiwi fruit,
oranges (sweet, sour), stone
fruits/strawberries
legume oilseeds, melons, 0.01
mushrooms, pineapples, root and
tuber vegetables, milksa
WHO has classified deltamethrin as a moderately hazardous
technical product in normal use (WHO, 1988). A Data Sheet on
deltamethrin (No. 50) has been issued (WHO/FAO, 1984).
-------------------------------------------------------------------
a Not yet confirmed by Codex Alimentarius Commission (FAO/WHO,
1986c, and 1988c).
REFERENCES
ADENEY, R.J., GRZYWACZ, D., & MATTHIESSEN P. (1980) The acute
toxicities of pyrethrin analogues to Sarotherodon mossambicus
(Peters), Centre for Overseas Pest Research, 6 pp. (Unpublished
proprietary data submitted to WHO by Roussel Uclaf).
AKHTAR, M.H. (1984) Metabolism of deltamethrin by cow and chicken
liver enzyme preparations. J. agric. food Chem., 32: 258-262.
AKHTAR, M.H., HAMILTON, R.M.G., & TRENHOLM, H.L. (1985)
Metabolism, distribution and excretion of deltamethrin by leghorn
hens. J. agric. food Chem., 33: 610-617.
AKHTAR, M.H., MARTIN, K.E., & TRENHOLM, H.L. (1986) Fate of
14C-deltamethrin in lactating dairy cows. J. agric. food Chem.,
34: 753-758.
AKHTAR, M.H., DANIS, C., TRENHOLM, H.L., & MARTIN, K.E. (1987)
Residues in milk and tissues of lactating dairy cows fed
deltamethrin for 28 consecutive days. J. agric. food Chem.,
ALDRIDGE, W.N., CLOTHIER, B., FORSHAW, P., & JOHNSON, M.K., PARKER,
V.H., PRICE, R.J., SKILLETER, D.N., VERSCHOYLE, R.D., STEVENS,
C. (1978) The effect of DDT and the pyrethroids cismethrin and
decamethrin on the acetyl choline and cyclic nucleotide content of
rat brain. Biochem. Pharmacol., 27: 1703-1706.
ALINIAZEE, M.T. & CRANHAM, J.E. (1980) Effects of four synthetic
pyrethroids on a predatory mite, Typhlodroums pyri, and its prey,
Panonychus ulmi, on apples in southeast England. Environ. Entomol.,
9: 436-439.
ARZONE, A. & VIDANO, C. (1978) [Azione sull'ape di etiofencaus,
decamethrin e ciexatin.] Apic. mod., 69: 157-162, (in Italian).
ATKINS, E.L., KELLUM D., & NEUMAN, K.J. (1976) Effect of
pesticides on apiculture project No. 1449: The annual report,
University of California Riverside, pp. 536-567.
AUDEGOND, L., CATEZ, D., FOULHOUX, P., FOURNEX, R., LE RUMEUR, C.,
L'HOTELLIER, M., & STEPNIEWSKI, J.P. (1988) Potentialisation de
la toxicité de la deltaméthrine par les insecticides
organophosphorés (Unpublished proprietary data submitted to WHO by
Roussel Uclaf).
AUDEGOND L., COLLAS E., & GLOMOT R. (1981) RU 22974
(deltamethrin) single administration study by oral route in the rat
(the compound is given as a suspension) (Unpublished report RU-
81239/A, submitted to WHO by Roussel Uclaf).
BAINOVA, A. & KALOYANOVA, F. (1985) [Study of allergenic and
irritating effect of synthetic pyrethroids on skin.] Hig. Zdrav.,
28(2): 19-21 (in Bulgarian).
BAINOVA, A., MIHAVSKI, M., ISMIROVA, N., & BENCHEV, V. (1986)
Specific skin irritation after contact with specific pyrethroids.
6th Congress of Bulgarian Dermatologists, Varna, 2-5 October, 1986,
(Abstract No. 74) (in Bulgarian).
BAKER, P.G. & BOTTOMLEY, P. (1982) Determination of residues of
synthetic pyrethroids in fruit and vegetables by gas-liquid and
high-performance liquid chromatography. Analyst, 107: 206-212.
BALDRY, D.A.T., EVERTS, J., ROMAN, B., BOON VON OCHSSEE, G.A., &
LAVEISSIERE, C. (1981) The experimental application of
insecticides from a helicopter for the control of riverine
populations of Glossina tachinoides in West Africa. Part VIII: The
effects of two spray applications of OMS-570 (endosulfan) and of
OMS 1998 (decamethrin) on G. tachinoides and non-target organisms
in Upper Volta. Trop. Pest Manage., 27(1): 83-110.
BEAVERS, J.B. & FINK, R. (1977a) Actue oral LD50 Mallard duck
technical DECIS final report, (Unpublished report WI77.06.06/A,
submitted to WHO by Roussel Uclaf). Wildlife International.
BERLIN, J.R., AKERA, T., BRODY, T.M., & MATSUMURA, F. (1984) The
inotropic effects of a synthetic pyrethroid decamethrin on isolated
guinea pig atrial muscle. Eur. J. Pharmacol., 98: 313-322.
BIDET, D., DUMONT, C., FOULHOUX, P., FOURNEX, R., & ROUGERON, C.
(1988) Mechanisms responsible for the toxicity of deltamethrin and
other pyrethroids. (Unpublished document, submitted to WHO by
Roussel Uclaf).
BLEYS, M., COTONAT, J., FOULHOUX, P. (1986) Lettre à l'éditeur,
J. Toxicol. clin. exp., 6(3): 211-212.
BLOOMQUIST, J.R., SODERLUND, D.M. (1985) Neurotoxic insecticides
inhibit GABA-dependent chloride uptake by mouse brain vesicles.
Biochem. biophys. Res. Commun., 133(1): 37-43.
BLOOMQUIST, J.R., ADAMS, P.M., & SODERLUND, D.M. (1986)
Inhibition of gamma-aminobutyric acid-stimulated chloride flux in
mouse brain vesicles by polychlorocycloalkane and pyrethroid
insecticides. Neurotoxicology, 7(3): 11-20.
BOCQUET, J.C., PASTRE, P., ROA, L., & BAUMEISTER, R. (1980) Etude
de l'action de la deltaméthrine sur Apis mellifera en conditions de
plein-champ. Phytiatr. Phytopharm., 29: 83-92.
BOCQUET, J.C., PASTRE, P., & BAUMEISTER, R. (1983) Bilan de cinq
années d'études de l'effet de la deltaméthrine sur abeilles en
conditions naturelles. 6ème Congrès International du Colza, Paris,
mai 1983.
BOUCHE, M.B. & FAYOLLE, L. (1979) Tests carried out with Procida
compound Decis EC 2.5 on earthworms lethal effect in relation with
time (Unpublished proprietary report No. INRA-VT-79.05.22/78.10.
10/A, submitted to WHO by Roussel Uclaf).
BOWMAN, H. & CARPENTER, M. (1987) Determination of
photodegradation of 14C-deltamethrin in aqueous solution
(Unpublished proprietary report ABC LABS 35491, submitted to WHO by
Roussel Uclaf).
BRADBURY, J.E., GRAY, A.J., & FORSHAW, P. (1981) Protection
against pyrethroid toxicity in rats with mephenesin. Toxicol.
appl. Pharmacol., 60: 382.
BRADBURY, J.E., FORSHAW, P.J., GRAY, A.J., & RAY, D.E. (1983) The
action of mephenesin and other agents on the effects produced by
two neurotoxic pyrethroids in the intact and spinal rat.
Neuropharmacology, 22(7): 907-914.
BRODIE, M.E. (1983) Correlations between cerebellar cyclic GMP
and motor effects induced by deltamethrin: independence of olivo-
cerebellar tract. Neurotoxicology, 4(4): 1-11.
BRODIE, M.E. (1985) Deltamethrin infusion into different sites in
the neuraxis of freely moving rats. Neurobehav. Toxicol. Teratol.,
7(1): 51-55.
BRODIE, M.E. & ALDRIDGE, W.N. (1982) Elevated cerebellar cyclic
GMP levels during the deltamethrin-induced motor syndrome.
Neurobehav. Toxicol. Teratol., 4: 109-113.
BRODIE, M.E. & OPACKA, J. (1985) Dissociation between circling
behaviour and striatal dopamine activity following unilateral
deltamethrin administration to rats. Naunyn-Schmiedeberg's Arch.
Pharmacol., 331: 341-346.
BROOKS, M.W. & CLARK, J.M. (1987) Enhancement of norepinephrine
release from rat brain synaptosomes by alpha cyano pyrethroids.
Pestic. Biochem. Physiol., 28: 127-139.
BUCCAFUSCO, R.J., ELLS, S.J., & CARY, G.A. (1977a) Acute toxicity
of NRDC 161 to bluegill (Lepomis macrochirus) under dynamic test
conditions, E.G. & G. Bionomics Aquatic Toxicology Laboratory, 12 pp.
(Unpublished proprietary data submitted to WHO by Roussel Uclaf).
BUCCAFUSCO R.J., ELLS, S.J., & CARY, G.A. (1977b) Acute toxicity
of NRDC 161 (Decamethrine) to Channel Catfish (Ictalurus
punctatus), E.G. & G. Bionomics Aquatic Toxicology Laboratory,
7 pp. (Unpublished proprietary data supplied to WHO by Roussel
Uclaf).
CABRAL, J.R.P., GALENDO, D., LAVAL, M., & LYANDRUT, N. (1986)
Carcinogenicity study of the pesticide deltamethrin in mice and
rats. Summary Report in Poster Session at IUPAC Meeting, Ottawa,
August 1986.
CARY, G.A. (1978) Kinetics of 14C-NRDC-161 in a model aquatic
ecosystem, E.G. & G. Bionomics Aquatic Toxicology Laboratory.
(Unpublished proprietary report BW-78-2-075, submitted to WHO by
Roussel Uclaf).
CHALMERS, A.E., MILLER, T.A, & OLSEN, R.W. (1987) Deltamethrin:
a neurophysiological study of the sites of action. Pestic.
Biochem. Physiol., 27: 36-41.
CHAMBON, A. & LEPAILLEUR, H. (1984) Etude de l'effet toxique de
la deltamethrine (produit technique > 98%) et de la formulation CE
25 g/l Decis vis-à-vis de l'espece de vers (Eisenia fetida andrei)
(Unpublished proprietary report IRCHA-84.30.07/F, submitted to WHO
by Roussel Uclaf).
CHANH, P.H., NAVARRO-DELMASURE, C., CHANH, A.P.H., LEAN, C.S.,
ZIADE, F., & SAMAHA, F. (1981) Analgesic effects of decamethrin,
Surg. Transplant., 9: 503-504.
CHAPMAN, R.A. & HARRIS, C.R. (1981) Persistence of four
pyrethroid insecticides in a mineral and organic soil. J. environ.
Sci. Health, B16: 605-615.
CHAPMAN, R.A., TU, C.M., HARRIS, C.R., & COLE, C. (1981)
Persistence of five pyrethroid insecticides in sterile and natural,
mineral and organic soil. Bull. Environ. Contam. Toxicol, 26:
513-519.
CHESTERMAN, H., HEYWOOD, R., PERKIN, C.J., BEARD, D., STREET, E., &
PRENTICE, D.E. (1977) RU 22974. Oral toxicity study in Beagle
dogs, Huntingdon, Huntingdon Research Centre, (Unpublished report
RSL 253/7751/A3, submitted to WHO by Roussel Uclaf).
CHINN, K. & NARAHASHI, T. (1986) Stabilization of sodium channel
states by deltamethrin in mouse neuroblastoma cells. J. Physiol.,
380: 191-207.
CLAIR, M. (1977) RU 22974 DECIS. Acute toxicity in the rabbit by
percutaneous administration. Joinville-le-Pont, Institut Français
de Recherches et Essais Biologique (Unpublished report IFREB-R
770257.1/A, submitted to WHO by Roussel Uclaf).
CLARK, G.C., JACKSON, G.C., & ALEXANDER, D.J. (1980) Acute
inhalation toxicity in rats, 4-hour exposure (Decis PM 2.5
percent), Huntingdon Research Centre, (Unpublished report RSL
437/80568, submitted to WHO by Roussel Uclaf).
COOMBS, D.W. & CLARK, G.C. (1978) RU 22974: Acute inhalation
toxicity in rats. 6 Hour LC50. Huntingdon, Huntingdon Research
Centre, (Unpublished report RSL 310/78453/A, submitted to WHO by
Roussel Uclaf).
COOMBS, D.W., CLARK, G.C., STREET, A.E., & GIBSON, W.A. (1978) RU
22974 inhalation toxicity study in rats 14 x 6 hour exposures over
a period of 3 weeks (Unpublished proprietary report
RSL/318/78638/A, submitted to WHO by Roussel Uclaf).
COQUET, B. (1976a) RU 22974. Test to determine primary cutaneous
irritation in the rabbit. Joinville-le-Pont, Institut Francais de
Recherches et Essais Biologiques (Unpublished report IFREB-R
761157/A, submitted to WHO by Roussel Uclaf).
COQUET, B. (1976b) RU 22974. Test to evaluate ocular irritation
in the rabbit, Joinville-le-Pont, Institut Français de Recherches
et Essais Biologiques (Unpublished report IFREB- R 761158/A,
submitted to WHO by Roussel Uclaf).
COQUET, B. (1977) RU 22974. Decis formulations - Determination of
the LD50 in the rat by oral administration, Joinville-le-Pont,
Institut Français de Recherches et Essais Biologiques (Unpublished
report 770257-A, submitted to WHO by Roussel Uclaf).
COTONAT, J., BLEYS, M., & FOULHOUX, P. (1987) Effets antagonistes
du phenprobamate et du carbamate de mephenesine sur l'intoxication
à la deltaméthrine. J. Toxicol. clin. exp., 7: 5-19.
CROFTON, K.M. & REITER, L.W. (1984) Effects of two pyrethroid
insecticides on motor activity and the acoustic startle response in
the rat. Toxicol. appl. Pharmacol., 75: 318-328
DAVID, D. (1981) Laboratory evaluation of repellent properties
against birds of the synthetic pyrethroid decamethrin. Poult. Sci.,
60: 1149-1151.
DAVIES, J.E., MUNT, P.L., & GOOR, J.L. (1983) RU 22974.
Investigation of possible neurological effects using the tilting
plane test. Huntingdon, Huntingdon Research Centre (Unpublished
proprietary report RSL603/83232/A, submitted to WHO by Roussel
Uclaf).
DE LAVAUR, E., LE SECH, J., & GROLLEAU, G. (1985) Accumulation et
élimination de la deltaméthrine chez la caille japonaise. Ann.
Fals. Exp. Chim., 835: 73-78.
DOHERTY, J.D., LAUTER, C.J., & SALEM, N., Jr (1986) Synaptic
effects of the synthetic pyrethroid resmethrin in rat brain in
vitro. Comp. Biochem. Physiol., C84(2): 373-9.
DUCLOHIER, H. & GEORGESCAULD, D. (1979) The effects of the
insecticide decamethrin on action potential and voltage-clamp
currents of myxicola giant axon. Comp. Biochem. Physiol., C62(2):
217-223.
DUMONT, C. & CHIFFLOT, L. (1978) Recherche d'un antidote contre
les effets toxiques aigus (Unpublished report RU-AE-72, submitted
to WHO by Roussel Uclaf).
DUMONT, C. & LAURENT, J. (1979) Recherche d'antagonistes des
effets neurotoxiques aigus du RU 22 974 (Unpublished report RU-AG-
35, submitted to WHO by Roussel Uclaf).
DUNNING, R.A., COOPER, J.M., WARDMAN, J.M., & WINDER, G. (1981)
Susceptibility of the carabid Pterostichus melanarius (Illiger) to
aphicide sprays applied to the sugar-beet crop. (Unpublished
proprietary report UK-SOIL-F.81/A, submitted to WHO by Roussel
Uclaf).
DYBALL, R.E.J. (1982) Inhibition by decamethrin and resmethrin of
hormone release from the isolated rat neurohypophysis - a model
mammalian neurosecretory system. Pestic. Biochem. Physiol., 17:
42-47.
ELLIOTT, M. (1977) Synthetic pyrethroids, Washington, DC,
American Chemical Society, p. 229 (ACS Symposium Series 42).
ELLIOTT, M., FARNHAN, A.W., JANES, N.F., NEEDHAM, P.H., & PULMAN,
D.A. (1974) Synthetic insecticide with a new order of activity.
Nature, 248: 710-711.
ESTESEN, B.J., BUCH, N.A., & WARE, G.W. (1979) Dislodgeable
insecticide residue on cotton foliage; permethrin, curacron,
fenvalerate, sulprofos, decis and endosulfan. Bull. environ.
Contam. Toxicol., 22: 245-248.
EVANS, M.H. (1976) End-plate potentials in frog muscle exposed to
a synthetic pyrethroid. Pestic. Biochem. Physiol., 6: 547-550.
EVERTS, J.W., VAN FRANKENHUYZEN, K., ROMAN, B., & KOEMAN, J.H.
(1983) Side effects of experimental pyrethroid applications for
the control of tsetse flies in a riverine forest habitat in Africa.
Arch. environ. Contam. Toxicol., 12: 91-97.
EVERTS, J.W., KORTENHOFF, B.A., HOOGLAND, H., VLUG, H.J., JACQUE,
R., & KOEMAN. J.H. (1985) Effects on man-target terrestrial
arthropods of synthetic pyrethroids asked for the control of the
Tsetse Fly ( Glossina spp.) in settlement areas of the Southern
Ivory Coast, Africa. Arch. environ. Contam. Toxicol., 14: 647-650.
FAO (1982) Second Government Consultation on International
Harmonization of Pesticide Registration Requirements, Rome, 11-15
October, 1982, Rome, Food and Agriculture Organization of the
United Nations.
FAO/WHO (1981) 1980 Evaluations of some pesticide residues in
food, Rome, Food and Agriculture Organization of the United Nations
(FAO Plant Production and Protection Paper No. 26 Sup).
FAO/WHO (1982) 198l Evaluations of some pesticide residues in
food, Rome, Food and Agriculture Organization of the United Nations
(FAO Plant Production and Protection Paper No. 42).
FAO/WHO (1983) 1982 Evaluations of some pesticide residues in
food, Rome, Food and Agriculture Organization of the United Nations
(FAO Plant Production and Protection Paper No. 49).
FAO/WHO (1985a) 1984 Evaluations of some pesticide residues in
food, Rome, Food and Agriculture Organization of the United Nations
(FAO Plant Production and Protection Paper No. 67).
FAO/WHO (1985b) Guide to Codex recommendations concerning
pesticide residues. Part 8. Recommendations for methods of
analysis of pesticide residues, 3rd ed., Rome, Food and Agriculture
Organization of the United Nations, Codex Committee on Pesticide
Residues.
FAO/WHO (1986a) 1985 Evaluations of some pesticide residues in
food. Part I - Residues, Rome, Food and Agriculture Organization
of the United Nations (FAO Plant Production and Protection Paper
No. 72/1).
FAO/WHO (1986b) 1986 Evaluations of some pesticide residues in
food. Part I - Residues, Rome, Food and Agriculture Organization
of the United Nations (FAO Plant Production and Protection Paper
No. 78).
FAO/WHO (1986c) Codex maximum limits for pesticide residues, 2nd
ed., Rome, Food and Agriculture Organization of the United Nations,
Codex Alimentarius Commission, CAC XIII.
FAO/WHO, (1988a) 1987 Evaluations of some pesticide residues in
food. Part I - Residues, Rome, Food and Agriculture Organization
of the United Nations (FAO Plant Production and Protection Paper
No. 86/1).
FAO/WHO, (1988b) 1988 Evaluations of some pesticide residues in
food. Part 1 - residues, Rome, Food and Agriculture Organization
of the United Nations. (FAO Plant Production and Protection Paper
93/1.
FAO/WHO, (1988c) Supplement 1 to Codex maximum limits for pesticide
residues, Rome, Food and Agriculture Organization of the United
Nations.
FISCHER, L. & CHAMBON, J.P. (1987) Faunistical inventory of
cereal arthropods after flowering and incidence of insecticide
treatments with deltamethrin, dimethoate and phosalone on the
apigeal fauna. Meded. Fac. Landbouwwet. Rijksuniv. Gent, 52(2a):
FLANNIGAN, S.A. & TUCKER, S.B. (1985) Variation in cutaneous
sensation between synthetic pyrethroid insecticides. Contact
Dermatitis, 13: 140-147.
FLORELLI, F., GARNIER, P., & ROA, L. (1987a) Bilan de 8 années
d'expérimentation sur la selectivite du Decis vis à vis des
abeilles - La défense des végétaux, 243: 8
FLORELLI, F., HELLER, J.J., GARNIER, P., & BAUMEISTER, R. (1987b)
Incidence sur les abeilles, les ruches et leur production, de
traitements aériens du colza avec la deltaméthrine - Annales ANPP -
Conférence Internationale sur les Ravageurs en Agriculture, Paris,
1-3 décembre 1987, 189-203.
FORSHAW, P.J. & BRADBURY, J.E. (1983) Pharmacological effects of
pyrethroids on the cardiovascular system of the rat. Eur. J.
Pharmacol., 91: 207-213.
FORSHAW, P.J. & RAY, D.E. (1986) The effects of two pyrethroids,
cismethrin and deltamethrin, on skeletal muscle and the trigeminal
reflex system in the rat. Pestic. Biochem. Physiol., 25: 143-151.
FORSHAW, P.J., LISTER, T., & RAV, D.E. (1987) The effects of two
types of pyrethroid on rat skeletal muscle. Eur. J. Pharmacol.,
134(1): 89-96.
FOUILLET, X. (1976) RU 22974 Mutagenicity study of various
preparations. Salmonella microsome test, Institut Français de
Recherches et Essais Biologiques (Unpublished report no. IFREB-R
761153/A, submitted to WHO by Roussel Uclaf).
FOULHOUX, P. (1981) Medical observations of personnel working on
synthesis or formulation of deltamethrin (Unpublished report RU-
81.18.06/A, submitted to WHO by Roussel Uclaf).
FOULHOUX, P. (1988) Assessment of potential adverse effects of
deltamethrin on man, Département Central de Toxicovigilance Roussel
Uclaf, 16 pp. (Unpublished report submitted to WHO by Roussel
Uclaf).
FOULHOUX, P., BOGGIN, CORNILLIET, POTTIER, RAYNAUD, STERN-VEYRIN, &
SUTTET (1981) Cutaneous irritation patch test on human volunteers
with Decis EC25, Decis flowable 25, Decis + mineral oil 25
(Unpublished report RU-81.03.02/DM/A, submitted to WHO by Roussel
Uclaf).
FOURNIER, P.E. (1988) Etude de l'activité antidote du carbamate
d'éthyl dans l'intoxication aiguë par la deltaméthrine chez le rat,
Paris, Unité de Pharmacologie Clinique, Hospital Fernand Widal
(Unpublished report submitted to WHO by Roussel Uclaf).
GAINES, T.B. & LINDER, R.E. (1986) Acute toxicity of pesticides
in adult and weanling rats, Fundam. appl. Toxicol., 7: 299-308.
GAMMON, D.W. & CASIDA, J.E. (1983) Pyrethroids of the most potent
class antagonize GABA action at the crayfish neuromuscular
junction. Neurosci. Lett., 40: 163-168.
GAMMON, D.W., BROWN, M.A., & CASIDA, J.E. (1981) Two classes of
pyrethroid action in the cockroach. Pestic. Biochem. Physiol., 15:
181-191.
GAMMON, D.W., LAWRENCE, L.J., & CASIDA, J.E. (1982) Pyrethroid
toxicology: Protective effects of diazepam and phenobarbital in
the mouse and the cockroach. Toxicol. appl. Pharmacol., 66:
290-296.
GLICKMAN, A.H. & CASIDA, J.E. (1982) Species and structural
variations affecting pyrethroid neurotoxicity. Neurobehav.
Toxicol. Teratol., 4(6): 793-799.
GLOMOT, R. (1979) Acute toxicity study by oral route in the rat
(Proprietary report RU-79803-54/A2, submitted to WHO by Roussel
Uclaf).
GLOMOT, R. & CHEVALIER, B. (1976a) RU 22974. Acute toxicity
study mouse and rat by oral route (Unpublished report TOX 76810/A,
submitted to WHO by Roussel Uclaf).
GLOMOT, R. & CHEVALIER, B. (1976b) RU 22974. Acute toxicity
study mouse and rat by intraperitoneal route (Unpublished report
TOX 76811/A, submitted to WHO by Roussel Uclaf).
GLOMOT, R. & CHEVALIER, B. (1976c) RU 22974. Acute toxicity
study mouse and rat by intravenous route (Unpublished report TOX
76812/A, submitted to WHO by Roussel Uclaf).
GLOMOT, R. & VANNIER, B. (1977) RU 22974. Teratological study in
mouse, rat and rabbit (Unpublished report TOX 76534-76536/2/A,
submitted to WHO by Roussel Uclaf).
GLOMOT, R. & VANNIER, B. (1978) RU 22974. Teratological study in
rabbit. Complementary information (Unpublished report TOX 76534-
76536/A3, submitted to WHO by Roussel Uclaf).
GLOMOT, R., AUDEGOND, L., & COLLAS, E. (1979) Acute oral toxicity
study in the rat (Decis 25 G/L Mixofluid) (Proprietary report RU
79824/A, submitted to WHO by Roussel Uclaf).
GLOMOT, R., AUDEGOND, L., & COLLAS, E. (1980a) Single
administration by oral route in the mouse (Decis Wettable Powder,
2.5%) (Proprietary report RU-80817/A, submitted to WHO by Roussel
Uclaf).
GLOMOT, R., AUDEGOND, L., & COLLAS, E. (1980b) Single
administration study by oral route in the dog (Decis Wettable
Powder, 2.5%) (Proprietary report RU 80194/A, submitted to WHO by
Roussel Uclaf).
GLOMOT, R., AUDEGOND, L., & COLLAS, E. (1981a) Single
administration study by oral route in the rat (Proprietary report
RU-81239/A, submitted WHO by Roussel Uclaf).
GLOMOT, R., AUDEGOND, L., & COLLAS, E. (1981b) Primary dermal
irritation study in the rabbit. Deltamethrin 25 G/L Mixofluid
(Proprietary report RU-79193/A, submitted to WHO by Roussel Uclaf).
GLOMOT, R., AUDEGOND, L., & COLLAS, E. (1981c) Primary dermal
irritation study in the rabbit (Deltamethrin wettable powder, 2.5%)
(Proprietary report RU-80200/A, submitted to WHO by Roussel Uclaf).
GLOMOT, R., AUDEGOND, L., & COLLAS, E. (1981d) Primary eye
irritation study in the rabbit (Deltamethrin in 25 G/L Mixofluid)
(Proprietary report RU-79192/A, submitted to WHO by Roussel Uclaf).
GLOMOT, R., AUDEGOND, L., & COLLAS., E. (1981e) Primary eye
irritation study in the rabbit (Decis Wettable Powder, 2.5%)
(Proprietary report RU-79192/A, submitted WHO by Roussel Uclaf).
GLOMOT, R., CHEVALIER, B., COLLAS, E., & AUDEGOND, L. (1977) RU
22974. Acute toxicity study by oral route in male and female
Beagle dogs (Unpublished report TOX 77804/JL-5, submitted to WHO
by Roussel Uclaf).
GOLDENTHAL, E.I., BLAIR, M., JEFFERSON, N.D., SPICER, E.J.F.,
ARCEO, R.J., & KAHN, A., III (1980a) RU 22974. Two year toxicity
and carcinogenicity study in mice, Mattawan, Michigan,
International Research and Development Corporation (Unpublished
report IRDC 406-001/A-4, submitted to WHO by Roussel Uclaf).
GOLDENTHAL, E.I., JEFFERSON, N.D., BLAIR, M., THORSTENSON, J.H.,
SPICER, E.J.F., ARCEO, R.J., & KAHN, A., III (1980b) RU 22974.
Two year oral toxicity and carcinogenicity study in rats,
Mattawan, Michigan, International Research and Development
Corporation (Unpublished report IRDC 406-002/A.4, submitted to WHO
by Roussel Uclaf).
GRANDADAM, A. (1976) Test to determine the toxicity in the
chicken by oral route (Unpublished report RU76.05.05/A, submitted
to WHO by Roussel Uclaf).
GRAY, A.J. & RICKARD, J. (1982) The toxicokinetics of
deltamethrin in rats after intravenous administration of a toxic
dose. Pestic. Biochem. Physiol.., 18: 205-215.
GRAY, A.J., CONNORS, T.A., & RICKARD, J. (1980) Mechanism of
mammalian toxicity of the pyrethroids. In: Littaner, U.Z. et al.,
ed. Neurotransmitters and their receptors, New York, John Wiley
and Sons, pp. 565-568.
GROLLEAU G. & GRIBAN, J. (1976a) Toxicity of deltamethrin or
DECIS in single ingestion in game duck Anas platyrhnchos L.,
JoyenJossas, France, Institut National de la Recherche Agronomique
(Unpublished report INRA-76.21 12/A, submitted to WHO by Roussel
Uclaf).
GROLLEAU, G. & GRIBAN, J. (1976b) Toxicity of deltamethrin or
DECIS by single ingestion in grey partridge, Perdix Perdix L. and
red partridge, Alectotis rufa L., JoyenJossas, France, Institut
National de la Recherche Agronomique (Unpublished report INRA76.28
09/A, submitted to WHO by Roussel Uclaf).
GUILLOT, J.P. & GUILAINE, J. (1977) RU 22974. Decaméthrine.
Decis technical Roussel Uclaf. Sensitization test in the guinea
pig. Joinville-le-Pont, Institut Français de Recherches et Essais
Biologique (Unpublished report no. IFREB-R 709241/A, submitted to
WHO by Roussel Uclaf).
GULYAS P. & CSANYI B. (undated) Acute fish toxicological
investigation of diverse insecticides, Hungary, Scientific Research
Centre of Economy of Water Supplies, 21 pp. (Unpublished
proprietary data supplied to WHO by Roussel Uclaf).
HALLS, G.R.H. & PERIAM, A.W. (1980) The fate of residues of NRDC
161 on wheat during storage and after milling and baking - Report
after 9 months storage, November 1980, Wellcome Research
Laboratories Report (Unpublished report HEFH 80-4, submitted to
WHO by Roussel Uclaf).
HARGREAVES, J.R. & COOPER, L.P. (1979) Phytotoxicity tests with
pyrethroid insecticides on glasshouse grown tomato seedlings.
Queensland J. Agric. anim. Sci., 36: 151-154.
HASCOET, M. (1977) Laboratory leaching soil study with
decamethrin (RU 22974), Versailles, Institut National de la
Recherche Agronomique (Unpublished proprietary report INRA-
77.20.09/A, submitted to WHO by Roussel Uclaf).
HE, F. (1987) Occupational neurotoxicology: Current problems and
trends. Paper presented at the XXII International Congress on
Occupational Health, Sydney, 27 September-2 October, 1987.
HE, F., WANG, S., LIU, L., CHEN, S., ZHANG, Z., & SUN, J. (1989)
Clinical manifestations and diagnosis of acute pyrethroid
poisoning. Arch. Toxicol., 63: 54-58.
HEHEITMULLER, T., SHUBA, P.J., & PARRISH, R. (1978) Acute
toxicity of Decis to sheepshead minnows (Cyprinodon variegatus),
E.G. & G. Bionomics Marine Laboratory, 7 pp. (Unpublished
proprietary report BR-78-12219, supplied to WHO by Roussel Uclaf).
HEWSON, R.T. & BURGESS, J.E. (1981) An operator study with
deltamethrin including measurements of nerve conduction times
(Unpublished proprietary report GB-NT-11.81A, submitted to WHO by
Roussel Uclaf).
HILL, B.D. (1982) Determination of Deltamethrin residues in
soils, Alberta, Agriculture Canada Research Station, 7 pp
(Unpublished proprietary report CAN-82.12.10/A, submitted to WHO by
Roussel Uclaf).
HILL, B.D. (1983) Persistence of deltamethrin in a Lethbridge
sandy clay loam. J. environ. Sci. Health, B18(6): 691-703.
HILL, B.D. & JOHNSON, D.L. (1987) Persistence of deltamethrin and
its isomers on pasture forage and litter. J. agric. Food Chem.,
35: 373-378.
HILL, B.D. & SCHAALJE, G.B. (1985) A two-compartment model for
the dissipation of deltamethrin on soil. J. agric. food Chem., 33:
1001-1006.
HIROMORI, T., NAKANISHI, T., KAWAGUCHI, S., SAKO, H., SUZUKI, T., &
KIYAMOTO, J. (1986) Therapeutic effects of methocarbamol on acute
intoxication by pyrethroids in rats. J. pestic. Sci., 11: 9-14.
HUNTER, B., JORDAN, J., HEYWOOD, R., HEPWORTH, P., STREET, A.E., &
PRENTICE, D.E. (1977) RU 22974. Assessment of toxicity to rats
by oral administration for 13 weeks (followed by a 4-week
withdrawal period) (Unpublished report RSL-254/76938/A3, submitted
to WHO by Roussel Uclaf).
HUSSON, J.M. (1978) Medical observations made on people working
on the manufacture and formulation of the pyrethroid insecticide
decamethrin (Unpublished report RU 78.25.08/A, submitted to WHO by
Roussel Uclaf).
IRDC (1980) Two year chronic dog feeding study, Matawan,
Michigan, International Research and Development Corporation
(Unpublished proprietary report IRDC-406-004/AI, submitted to WHO
by Roussel Uclaf).
JACKSON, G. & HARDY, C. (1986) RU 22974. Acute inhalation
toxicity in rats 1-hour exposure (Unpublished report RSL-DTM-
851457/A, submitted to WHO by Roussel Uclaf).
JACQUES, Y., ROMEY, G., CAVEY, M.T., KARTAALOVSKI, B., & LAZDUNSKI,
M. (1980) Interaction of pyrethroids with the sodium channel in
mammalian neuronal cells in culture. Biochem. Biophys. Acta, 600:
882-897.
KAGAN, YU. S., PANSHINA, T.N., & SASINOVICH, L.M. (1986)
Biochemical effects of toxic action of synthetic pyrethroids. Gig.
i Sanit., 1: 7-9.
KAUFMAN, D.D. & KAYSER, A.J. (1979a) Degradation of 14C-phenoxy-
and 14C-cyano-decamethrin in soil (Unpublished proprietary report
USDA-I-23.04.79/A2, submitted to WHO by Roussel Uclaf).
KAUFMAN, D.D. & KAYSER, A.J. (1979b) The effect of soil
temperature on the degradation of 14C-cyano-decamethrin in soil
(Unpublished proprietary report USDA-II-24.04.79/A2, submitted to
WHO by Roussel Uclaf).
KAUFMAN, D.D. & KAYSER, A.J. (1980) Degradation of 14C-cyano-,
14C-phenoxy-, and 14C-vinyl-decamethrin in flooded soil
(Unpublished proprietary report USDA-III-12.05.80/A, submitted to
WHO by Roussel Uclaf).
KAUFMAN, D.D., RUSSEL, B.A., HELLING, C.S., & KAYSER, A.J. (1981)
Movement of cypermethrin, decamethrin, permethrin and their
degradation products in soil. J. agric. food Chem., 29: 239-245.
KAVLOCK, R., CHERNOFF, N., BARON, R., LINDER, R., ROGERS, E.,
CARVER, B., DILLEY, J., & SIMMON, V. (1979) Toxicity studies with
decamethrin, a synthetic pyrethroid insecticide. J. environ.
Pathol. Toxicol., 2: 751-765.
KNAUF, W. & HORLEIN, G. (1979 The acute toxicity of decamethrine
to the Rainbow trout Salmo gairdneri, Richardson, Frankfurt,
Hoechst AG, 19 pp (Unpublished proprietary report 19/79, submitted
to WHO by Roussel Uclaf).
KNAUF, W. & SCHULZE, E.F. (1977a) Effect of decamethrine on
Cyprinus carpio, Frankfurt, Hoechst AG, 9 pp (Unpublished
proprietary report 15/77, submitted to WHO by Roussel Uclaf).
KNAUF, W. & SCHULZE, E.F. (1977b) The effect of decamethrin
6E0660 on Catfish (Ictalurus nebulosus) in a static test,
Frankfurt, Hoechst Ag, 18 pp (Unpublished proprietary report
32/79, submitted to WHO by Roussel Uclaf).
KRASNJIH, A. & PAVLOVA, L. (1985) Harmful effects of
deltamethrin. In: Levina, E., ed. Harmful substances in industry,
Moscow, Khimija Publishing House, p. 154.
KYNOCH, S.R., LLOYD, G.K., & ANDREWS, C.D. (1979) Acute
percutaneous toxicity to rats of decamethrin, Huntingdon,
Huntingdon Research Centre (Unpublished report RSL-1009 8/D,
submitted to WHO by Roussel Uclaf).
LAWRENCE, L.J. & CASIDA, J.E. (1982) Pyrethroid toxicology: Mouse
intracerebral structure-toxicity relationship. Pestic. Biochem.
Physiol., 18: 9-14.
LAWRENCE, L.J. & CASIDA, J.E. (1983) Stereospecific action of
pyrethroid insecticides on the gamma-aminobutyric acid receptor-
ionophore complex. Science, 221: 1399-1401.
LAWRENCE, L.J., GEE, K.W., & YAMAMURA, H.I. (1985) Interactions
of pyrethroid insecticides with chloride ionophore-associated
binding sites. Neurotoxicology, 6: 87-98.
LEAHEY, J.P. (1985) The pyrethroid insecticide, London, Taylor &
Francis Ltd., p. 440.
LECLERCQ, M., COTONAT, J., & FOULHOUX, P. (1986) Recherche d'un
antagoniste à l'intoxication par la deltaméthrine. J. Toxicol.
clin. exp., 6, 83-85.
LEIBOWITZ, M.D., SCHWARZ, J.R., HOLAN, G., & HILLE, B. (1987)
Electrophysiological comparison of insecticide and alkaloid
agonists of Na channels. J. gen. Physiol., 90(1): 75-93.
LEPAILLEUR, H. & CHAMBON, A. (1984) Etude de la toxicité létale
du produit technique deltaméthrine vis à vis du poisson-zebré, 5 pp
(Unpublished proprietary dated report IRCHA B.7827, submitted to
WHO by Roussel Uclaf).
LHOSTE, J., FRANCOIS, Y., & RUPAUD, Y. (1979) Ichtyotoxicité de
la decaméthrine vis à vis de Salmo trutta L. (poisson téléostéen)
en fonction de l'age et des conditions expérimentales - Congrès sur
la lutte contre les insectes en milieu tropical, Marseilles - March
13-16 1979, pp. 885-902.
L'HOTELLIER, M. & VINCENT, P. (1986) Assessment of the impact of
deltamethrin on aquatic species. In British Crop Protection
Conference - Pests and Disease: 1109-1116.
LOUVEAUX, J., MISSONNIER, J., & MESQUIDA, J. (1977) Tests de
toxicité sur abeille domestique avec le Decis CE (Unpublished
proprietary report submitted to WHO by Roussel Uclaf).
LUMMIS, S.C., CHOW, S.C., HOLAN, G., & JOHNSTON, G.A. (1987)
Gamma-aminobutyric acid receptor ionophore complexes: differential
effects of deltamethrin, dichlorodiphenyltrichloroethane, and some
novel insecticides in a rat brain membrane preparation. J.
Neurochem., 48(3): 689-94.
LUND, A.E. & NARAHASHI, T. (1983) Kinetics of sodium channel
modification as the basis for the variation in the nerve membrane
effects of pyrethroids and DDT analogs. Pestic. Biochem. Physiol.,
20: 203-216.
MACPHAIL, R.C. (1981) Behavioral effects of a synthetic
insecticide (decamethrin). Fed. Proc. Fed. Am. Soc. Exp. Biol.,
40: 678.
MEISTER, R.T., BERG, G.L., SINE, C., MEISTER, S., & POPLYK, J.
(1983) Farm chemicals handbook. Section C. Pesticide dictionary,
Willoughby, Ohio, Meister Publishing Co.
MESTRES, R., CHEVALLIER, C., ESPNOZA, C., & CORNET, R. (1978a)
Dosage des résidues de decaméthrine dans les produits végétaux.
Trav. Soc. Pharm. Montpellier, 38: 183-192.
MESTRES R., CHEVALLIER C., & ESPINOZA, C. (1978b) Analytical
method for decamethrine residue analysis, Montpellier, Faculte de
Pharmacie, 2 pp. (Proprietary report FP-78.08.07/A, dated 7/8/1978
submitted to WHO by Roussel Uclaf).
MESTRES, R., FRANCOIS, C., CAUSSE C., VIAN L., & WINNETT, G.
(1985) Survey of exposure to pesticides in greenhouses. Bull.
environ. Contam. Toxicol., 35: 750-756.
MESTRES, R., ESPINOZA, C., & CHEVALLIER, C. (1986) Effets sur les
résidus de deltaméthrine de la transformation des produits
agricoles en vue de leur consommation. Med. Nutr., XXII (3):
181-184.
MILLER, T.A. & ADAMS, M.E. (1980) Neural and behavioral
correlates of pyrethroid and DDT-type poisoning in the housefly,
Musca domestica. Pestic. Biochem. Physiol., 13: 137-147.
MIYAMOTO, J. (1976) Degradation, metabolism and toxicity of
synthetic pyrethroids. Environ. Health Perspect., 14: 15-28.
MIYAMOTO, J. (1981) The chemistry, metabolism and residue
analysis of synthetic pyrethroids. Pure appl. Chem., 53: 1967-2022.
MIYAMOTO, J. & KEARNEY, P.C. (1983) Pesticide chemistry - Human
welfare and the environment. Proceedings of the Fifth International
Congress of Pesticide Chemistry, Kyoto, Japan, 29 August - 4
September, 1982, Oxford, Pergamon Press, Vol. 1-4.
MOHSEN, Z.H. & MULLA, M.S. (1981) Toxicity of blackfly larvicidal
formulations to some aquatic insects in the laboratory. Bull.
environ. Contam. Toxicol., 26: 696-703.
MOUROT, D., DELEPINE, B., BOISSEAU, J., & GAYOT, G. (1979) High-
performance liquid chromatography of decamethrin. J. Chromatogr.,
173: 412-414.
MUIR, D.C.G., RAWN, G.P., & GRIFT, N.P., (1985) Fate of the
pyrethroid insecticide deltamethrin in small ponds: A mass balance
study. J. agric. food Chem., 33: 603-609.
MULLA, M.S., NAVVAB GOJRATI, H.A., & DARWAZEH, H.A. (1978)
Toxicity of mosquito larvicidal pyrethroids to four species of
freshwater fishes. Environ. Entomol.., 7(3): 428-430.
NETO, P.X.R., RODRIGUES, I.C.F., & FILHO, A.M (1983) Etude sur
les effets secondaires de la deltaméthrine sur l'ichtyofaune de la
region du projet "Rio Formoso" (Unpublished report submitted to WHO
by Roussel Uclaf).
NOBLE, R.M., HAMILTON, D.J., & OSBORNE, W.J. (1982) Stability of
pyrethroids on wheat in storage. Pestic. Sci., 13: 246-252.
PANSHINA T.N. & SASINOVICH, L.M. (1983) Toxicology of synthetic
pyrethroids. Khim. sel'sk. khoz., 12: 51-53.
PANSU, M., DHOUIBI, M.H., & PINTA, M. (1981) Determination of
traces of biopermethrine and decamethrine pyrethrinoids in
biological substrates by gas chromatography. Analysis, 9: 55-59.
PAPALEXIOU, Ph., BITAR, N., & STOCKIS, A. (1984) Absorption of
radiocarbon labelled deltamethrin given orally in healthy
volunteers, Charleroi, Belgium, Institut de Pharmacologie et
d'Investigations Biomédicales (Unpublished proprietary report
50/22, submitted to WHO by Roussel Uclaf).
PARKIN, P.J. & LEQUESNE, P.M. (1982) Effects of a synthetic
pyrethroid deltamethrin on excitability changes following a nerve
impulse. J. Neurol. Neurosurg. Psychiatry., 45: 337-342.
PEYRE, M., CHANTOT, J.F., GLOMOT, R., & PENASSE, L. (1980)
Detection of a mutagenic potency of decamethrin (RU 22974), in
bacterial tests (Unpublished report RU/TOX/80-2101/A, submitted to
WHO by Roussel Uclaf).
PHAM, H.C., NAVARRO-DALMASURE, C., PHAM, H.C., CLAVEL, P., VAN
HAVERBEKE, G., CHEAV, S.L. (1984) Toxicological studies of
deltamethrin. Int. J. Tissue React., 6(2): 127-33.
PICHON, Y. (1981) Pharmacological characterization of ionic
channels in unmyelinated axons. J. Physiol., 17(9): 1119-1128.
PLAPP, F.W., Jr & BULL, D.L. (1978) Toxicity and selectivity of
some insecticides to Chrysopa carnea, a predator of the tobacco
budworm. Environ. Entomol., 7: 431-434.
PLESTINA, R. (1981) An evaluation of the use of deltamethrin in
public health (Unpublished report ZAG. KOT. 81/A, submitted to WHO
by Roussel Uclaf).
PLUIJMEN, M., DREVON, C., MONTESANO, R., MALAVEILLE C.,
HAUTEFEUILLE, A., & BARTSCH, H. (1984) Lack of mutagenicity of
synthetic pyrethroids in Salmonella typhimurium strains and in V79
Chinese hamster cells. Mutat. Res., 137: 7-15.
POLAKOVA, H. & VARGOVA, M. (1983) Evaluation of the mutagenic
effects of decamethrin: cytogenetic analysis of bone marrow. Mutat.
Res., 120: 167-171.
POTTIER J., CHATELET, P., & JOUQUEY, S. (1982) Resorption
percutanée de la deltaméthrine - Etude d'un modèle animal, 10 pp.
(Unpublished proprietary report AN.34, submitted to WHO by Roussel
Uclaf).
PRASADA RAO, K.S., CHETTY, C.S., & DESAIAH, D. (1984) In vitro
effects of pyrethroids on rat brain and liver ATPase activities. J
Toxicol. environ. Health, 14(2-3): 257-65.
RAWN, G.P., MUIR, D.O.G., & GRIFT, P.G. (1985) Fate of the
pyrethroid insecticide deltamethrin in small ponds, a mass balance
study. J. agr. food Chem., 33: 603-609.
RAY, D.E. (1980) An EEG investigation of decamethrin-induced
choreathetosis in the rat. Exp. Brain Res., 38: 221-227.
RAY, D.E. (1982) Changes in brain blood flow associated with
deltamethrin-induced choreoathetosis in the rat. Exp. Brain Res.,
45: 269-276.
RAY, D.E. & CREMER, J.E. (1979) The action of decamethrin (a
synthetic pyrethroid) on the rat. Pestic. Biochem. Physiol., 10:
333-340.
RICHTER, W.R. & GOLDENTHAL, E.I. (1983) Two-year oral toxicity
and carcinogenicity study in rats. Amendment to the final report.
Matawan, Michinga, International Research and Development
Corporation (Unpublished proprietary report IRDC 406.002/A5,
submitted to WHO by Roussel Uclaf).
RICKARD, J. & BRODIE, M.E. (1985) Correlation of blood and brain
levels of the neurotoxic pyrethroid deltamethrin with the onset of
symptoms in rats. Pestic. Biochem. Physiol., 23: 143-156.
RICOU (1978) Assessment of side effects of S 276-B (Decis EC
25 g/l) towards grey slugs (Agriolimax spp.) (Unpublished
proprietary report INRA-LIM.78.10/A, submitted to WHO by Roussel
Uclaf).
ROSE, G.P. & DEWAR, A.J. (1983) Intoxication with four synthetic
pyrethroids fail to show any correlation between neuromuscular
dysfunction and neurobiochemical abnormalities in rats. Arch.
Toxicol., 53: 297-316.
ROSE, D.B., ROBERTS, N.L., CAMERON, M.McD., PRENTICE, D.E., COOKE,
L., & GIBSON, W.A. (1978) RU 22974. (Decamethrine) LD50
determination and assessment of neurotoxicity in the domestic hen,
Huntingdon, Huntingdon Research Centre (Unpublished report RSL 293-
NT/7830/A, submitted to WHO by Roussel Uclaf).
ROUSSELIN, X. (1983) Toxicité des dérivés du pyrèthre, Paris,
Faculté de Médecine Lariboisiere-Saint-Louis (Thèse de l'Université
Paris VII).
RUIGT, G.S.F. & VAN DEN BERCKEN, J. (1986) Action of pyrethroids
on a nerve-muscle preparation of the clawed frog, Xenopus laevis.
Pestic. Biochem. Physiol., 25: 176-187.
RUZO, L.O. & CASIDA, J.E. (1979) Degradation of decamethrin on
cotton plants. J. agric. food Chem., 27: 572-575.
RUZO, L.O., HOLMSTEAD, R.L., & CASIDA, J.E. (1976) Solution
photochemistry of the potent pyrethroid insecticide alpha-cyano-3-
phenoxy-benzyl cis-2,2-dimethyl-3-(2,2-dibromovinyl)
cyclopropanecarboxylate. Tetrahedron Lett., 3045-3048.
RUZO, L.O., HOLMSTEAD, R.L., & CASIDA, J.E. (1977) Pyrethroid
photochemistry: decamethrin. J. agric. food Chem., 25: 1385-1394.
RUZO, L.O., UNAI, T., & CASIDA, J.E. (1978) Decamethrin metabolism
in rats. J. agric. food Chem., 26: 918-925.
RUZO, L.O., ENGEL, J.L., & CASIDA, J.E. (1979) Decamethrin
metabolites from oxidative, hydrolytic and conjugative reactions in
mice. J. agric. food Chem., 27: 725-731.
SANTOSA, K. (1983) Toxicity of Decis 2.5 EC (Decamethrin) to
fish, Jakarta, Department of Agriculture Research and Development
Inland Fisheries Institute, 4 pp. (Unpublished proprietary data
submitted to WHO by Roussel Uclaf).
SANTOSA, K. & HADI, H. (1980) Trial results pesticide lethal
toxicity to fish, Department of Agriculture Research and
Development Inland Fisheries Institute, Jakarta, Indonesia -
Report no. 174/UI/80 dated 5 December 1980, 3 pp. (Unpublished
proprietary data submitted to WHO by Roussel Uclaf).
SMIES, M., EVERS, R.H.J., REIJNENBURG, P.H.M., & KOEMAN, J.H.
(1980) Environmental aspects of field trials with pyrethroids to
eradicate tsetse fly in Nigeria. Ecotoxicol. environ. Saf., 4:
114-128.
SOBELS, F.H., TATES, A.D., & VANNIER, B. (1978) Cytogenetic study
with RU 22974. Detection of a mutagenic potency in mammalian
cells, Leiden, The Netherlands, State University of Leiden
(Unpublished report ULN-782211/A, submitted to WHO by Roussel
Uclaf).
SODERLUND, D.M. & CASIDA, J.E. (1977) Effects of pyrethroid
structure on rates of hydrolysis and oxidation by mouse liver
microsomal enzymes. Pestic. Biochem. Physiol., 7: 391-401.
SOUYRI, F. (1985) Autoradiographic studies of (3H)-deltamethrin
binding with nerve tissue cultures. Pestic. Sci., 6(6): 701-703.
STAATZ, C.G., BLOOM, A.S., & LECH, J.J. (1982) Effects of
pyrethroids on [3H]-kainic acid binding to mouse forebrain
membranes. Toxicol. appl. Pharmacol., 64: 566-569.
STAATZ-BENSON, C.G. & HOSKO, M.J. (1986) Interaction of
pyrethroids with mammalian spinal neurons. Pestic. Biochem.
Physiol., 25: 19-30.
STEIN, E.A., WASHBURN, M., WALCZAK, C., & BLOOM, A.S. (1987)
Effects of pyrethroid insecticides on operant responding maintained
by food. Neurotoxicol. Teratol., 9(1): 27-31.
STEVENSON, J.H., NEEDHAM, P.H., & WALKER, J. (1978) Poisoning of
honey bee by pesticides: investigations of the changing pattern in
Britain over 20 years. Rothamsted Exp. Stn Rep., Part 2: 55-72.
TAKAHASHI, M. & LEQUESNE, P. ((1982) The effects of the
pyrethroids deltamethrin and cismethrin on nerve excitability in
rats. J. Neurol. Neurosurg. Psychiatry, 45: 1005-1011.
TAKKEN, W., BALK, F., JANSEN, R.C., & KOEMAN, J.H. (1978) The
experimental application of insecticides from a helicopter for the
control of riverine populations of Glossina tachinoides in West
Africa. VI. Observations on side effects. P.A.N.S., 24: 455-466.
THIEBAULT, J.J., BOST, J., & FOULHOUX, P. (1985) Intoxication
expérimentale par la deltaméthrine chez le chien et son traitement.
Toxicol. vét. Collect. Méd. lég. Toxicol. méd., 131, 47-62.
THIEBAULT, J.J., BOST, J., FOULHOUX, P., DEVAUX, P., & TILLIER, C.
(in press) Intravenous deltamethrin intoxication of dogs.
Therapeutic attempts. Veterinary and Human Toxicology.
THIER, W. & SCHMIDT, D. (1976) Laboratory leaching studies of
Decis EC 25 in three different German soils (Unpublished
proprietary report RL-77.25.05/A, submitted to WHO by Roussel
Uclaf).
THIER, W. & SCHMIDT, D. (1977) The behaviour of the pesticide in
the soil: Decamethrin, (Unpublished proprietary report A09232,
submitted to WHO by Roussel Uclaf) (in German).
TONG YING (1988) Clinical manifestations of 211 cases intoxicated
by deltamethrin and fenvalerate (Unpublished report submitted to
WHO by Roussel Uclaf).
TOOBY, T.E., THOMSON, A.N., RYCROFT, R.J., BLACK, I.A., & HEWSON,
R.T. (1981) A pond study to investigate the effects on fish and
aquatic invertebrates of deltamethrin applied directly onto water,
Fisheries Laboratory, Ministry of Agriculture, Fisheries and Food
(Unpublished report AEP-81.30.09A, submitted to WHO by Roussel
Uclaf).
TRIVEDI, K. (1981) Spray worker exposure and health survey during
decamethrin (decis) spray in the field, Sevagram, India, Mahatma
Gandhi Institute of Medical Sciences (Unpublished information
submitted to WHO by Roussel Uclaf).
TU, C.M. (1980) Influence of five pyrethroid insecticides on
microbial populations and activities in soil. Microb. Ecol., 5:
321-327.
VAN DEN BERCKEN, J. (l977) The action of allethrin on the
peripheral nervous system of the frog. Pestic. Sci., 8: 692-699.
VAN DEN BERCKEN, J. & VIJVERBERG, H.P.M. (1980) Voltage clamp
studies on the effects of allethrin and DDT on the sodium channels
in frog myelinated nerve membrane. In: Insect neurobiology and
pesticide action (Neutox 79), London, Society of Chemical Industry,
Vol. 2, pp. 79-85.
VAN DEN BERCKEN, J., AKKERMANS, L.M.A., & VAN DER ZALM, J.M.
(1973) DDT-like action of allethrin in the sensory nervous system
of Xenopus laevis. Eur. J. Pharmacol., 21: 95-106.
VAN DEN BERCKEN, J., KROESE, A.B.A., & AKKERMANS, L.M.A. (1979)
Effect of insecticides on the sensory nervous system. In:
Narahashi, T., ed. Neurotoxicology of insecticides and pheromones,
New York, London, Plenum Press, pp. 183-210.
VANNIER, B. & FOURNEX, R. (1983) Deltamethrin detection of a
mutagenic potency/micronucleus test in the mouse (Proprietary
unpublished report RU-83607/A, submitted to WHO by Roussel Uclaf).
VANNIER, B. & GLOMOT, R. (1977) RU 22974. Mutagenic study.
Dominant lethal assay in the male mouse (Unpublished report TOX
76533/DB9/A, submitted to WHO by Roussel Uclaf).
VANNIER, B. & GLOMOT, R. (1982) RU 22974 complementary
teratological study in the mouse (Unpublished report RU-82506-12/A,
submitted to WHO by Roussel Uclaf).
VARANKA, I. (1987) Effect of mosquito killer insecticides on
freshwater mussels. Comp. Biochem. Physiol. C. Comp. Pharmacol.,
86: 157-162.
VAYSSE, M., GIUDICELLI, J.C., DEVAUX, P., & L'HOTELLIER, M. (1984)
Decis, in Zweig analytical methods for pesticides and plant growth
regulators. Vol XIII, 3: 53-68, New York, Academic Press.
VERSCHOYLE, R.D. & ALDRIDGE, W.N. (1980) Structure-activity
relationship of some pyrethroids in rats. Arch. Toxicol., 45:
325-329.
VICKERMAN, G.P., COOMBES D.S., TURNER, G., MEAD-BRIGGS, M.A., &
EDWARDS, J. (1987a) The effects of pirimicarb, dimethoate and
deltamethrin on carabidae and staphylinidae in winter wheat. Meded.
Fac. Landbouwwet. Rijksuniv. Gent, 52(2a):
VICKERMAN, G.P., COOMBES, D.S., TURNER, G., MEAD-BRIGGS, M.A., &
EDWARDS, J. (1987b) The effects of pirimicarb, dimethoate and
deltamethrin on non-target arthropods in winter wheat.
International Conference on Pests in Agriculture, Paris, 1-3
December 1987, Annales ANPP, pp. 67-74.
VIJVERBERG, H.P.M., VAN DEN BECKEN, J. (1982) Action of
pyrethroid insecticides on the vertebrate nervous system.
Neuropathol. appl. Neurobiol., 8: 421-440.
VIJVERBERG, H.P.M. & VAN DEN BERCKEN, J. (1979) Frequency
dependent effects of the pyrethroid insecticide decamethrin in frog
myelinated nerve fibres. Eur. J. Pharmacol., 58: 501-504.
VIJVERBERG, H.P.M., RUIGT, G.S.F., & VAN DEN BERCKEN, J. (1982a)
Structure-related effects of pyrethroid insecticides on the
lateral-line sense organ and on peripheral nerves of the clawed
frog, Xenopus laevis. Pestic. Biochem. Physiol., 18: 315-324.
VIJVERBERG, H.P.M., VAN DER ZALM, J.M., & VAN DEN BERCKEN, J.
(1982b) Similar mode of action of pyrethroids and DDT on sodium
channel gating in myelinated nerves. Nature, (Lond.) 295: 601-603.
VIJVERBERG, H.P.M., VAN DER ZALM, J.M., VAN KLEEF, R.G.D.M., & VAN
DEN BERCKEN, J. (1983) Temperature and structure-dependent
interaction of pyrethroids with the sodium channels in frog node of
Ranvier. Biochim. Biophys. Acta, 728: 73-82.
WALTERSDORFER & SCHULZE E.F. (1976a) Decis 2.5. Effect on
Lebistes reticulatus, Francfort, Hoechst Ag, 8 pp. (Unpublished
proprietary report 42/76, submitted to WHO by Roussel Uclaf).
WALTERSDORFER & SCHULZE E.F. (1976b) Decis 2.5. Effect on Idus
melanotus, Frankfurt, Hoechst AG, 5 pp (Unpublished proprietary
report 33/76, submitted to WHO by Roussel Uclaf).
WALTERSDORFER & SCHULZE E.F. (1976c) Decis 2.5. Effect on Salmo
gairdneri (rainbow trout), Frankfurt, Hoechst AG, 7 pp (Unpublished
proprietary report 35/76, submitted to WHO by Roussel Uclaf).
WALTERSDORFER & SCHULZE E.F. (1976d) Decamethrine effect on
Lepomis gibbosus (Bluegill sunfish), Frankfurt, Hoechst AG, 6 pp.
(Unpublished proprietary report 16/77, submitted to WHO by Roussel
Uclaf).
WANG, S., ZHENG, Q., YU, L., XU, B., & LIX., Y. (1988) Health
survey among farmers exposed to deltamethrin in the cotton fields.
Ecotoxicol. environ. Saf., 15: 1-6.
WELLCOME FOUNDATION (1979) Residues in cow's milk resulting from
intrarumenal, and later, dermal administration of 14C-labelled
decamethrin (Unpublished report submitted to WHO by Roussel Uclaf).
WHO (1979) Safe use of pesticides. Third Report of the WHO
Expert Committee on Vector Biology and Control, Geneva, World
Health Organization, pp. 18-23. (WHO Technical Report Series,
No. 634).
WHO (1988) The WHO recommended classification of pesticides by
hazard and guidelines to classification 1988-1989, Geneva, World
Health Organization (Unpublished document WHO/VBC/88.953).
WHO (1989) Health and Safety Guide No. 30: Deltamethrin, Geneva,
World Health Organization.
WHO (1984) Data sheet on pesticides, No. 50: Deltamethrin, Geneva,
World Health Organization, 9 pp (Unpublished document
VBC/DS/84.50).
WOOD MACKENZIE (1980) Agrochem. Monit., 9: 12.
WOOD MACKENZIE (1981) Agrochem. Monit., 15: 12.
WOOD MACKENZIE (1982) Agrochem. Monit., 21: 13.
WOOD MACKENZIE (1983) Agrochem. Monit., 27: 3-12.
WORTHING, C.R. & WALKER, S.B. (1983) Pesticide manual, 7th ed.,
Croydon, British Crop Protection Council, p. 161.
WOUTERS, W. & VAN DEN BERCKEN, J. (1978) Action of pyrethroids.
Gen. Pharmacol., 9: 387-398.
WRENN, J.M., RODWELL, D.E., GOLDENTHAL, E.I., SPIDER, E.J.C., &
RAJASEKARAN, D. (1980) Three-generation reproduction study in
rats, Matawan, Michigan, International Research and Development
Corporation (Unpublished report IDC-406 003/A4. submitted to WHO by
Roussel Uclaf).
ZHANG, L.Z., KHAN, S.U., AKHTAR, M.H., & IVARSON, K.C. (1984)
Persistence, degradation, and distribution of Deltamethrin in an
organic soil under laboratory conditions, J. agric. food Chem., 32:
1207-1211
ZITKO, V., MCLEESE, D.W., METCALFE, C.D., & CARSON, W.G. (1979)
Toxicity of permethrin, decamethrin, and related pyrethroids to
salmon and lobster. Bull. environ. Contam. Toxicol., 21: 338-343.
APPENDIX I
On the basis of electrophysiological studies with peripheral
nerve preparations of frogs (Xenopus laevis; Rana temporaria; and
Rana esculenta), it is possible to distinguish between 2 classes of
pyrethroid insecticides: (Type I and Type II). A similar
distinction between these 2 classes of pyrethroids has been made on
the basis of the symptoms of toxicity in mammals and insects (Van
den Bercken et al., 1979; WHO, 1979; Verschoyle & Aldridge, 1980;
Glickman & Casida, 1982; Lawrence & Casida, 1982). The same
distinction was found in studies on cockroaches (Gammon et al.,
1981).
Based on the binding assay on the gamma-aminobutyric acid
(GABA) receptor-ionophore complex, synthetic pyrethroids can also
be classified into two types: the alpha-cyano-3-phenoxybenzyl
pyrethroids and the non-cyano pyrethroids (Gammon et al., 1982;
Gammon & Casida, 1983; Lawrence & Casida, 1983; Lawrence et al.,
1985).
Pyrethroids that do not contain an alpha-cyano group
(allethrin, d-phenothrin, permethrin, tetramethrin, cismethrin, and
bioresmethrin) (Type I: T-syndrome)
The pyrethroids that do not contain an alpha-cyano group give
rise to pronounced repetitive activity in sense organs and in
sensory nerve fibres (Van den Bercken et al., 1973). At room
temperature, this repetitive activity usually consists of trains of
3 - 10 impulses and occasionally up to 25 impulses. Train duration
is between 10 and 5 milliseconds.
These compounds also induce pronounced repetitive firing of the
presynaptic motor nerve terminal in the neuromuscular junction (Van
den Bercken, 1977). There was no significant effect of the
insecticide on neurotransmitter release or on the sensitivity of
the subsynaptic membrane, nor on the muscle fibre membrane.
Presynaptic repetitive firing was also observed in the sympathetic
ganglion treated with these pyrethroids.
In the lateral-line sense organ and in the motor nerve
terminal, but not in the cutaneous touch receptor or in sensory
nerve fibres, the pyrethroid-induced repetitive activity increases
dramatically as the temperature is lowered, and a decrease of 5 °C
in temperature may cause a more than 3-fold increase in the number
of repetitive impulses per train. This effect is easily reversed
by raising the temperature. The origin of this "negative
temperature coefficient" is not clear (Vijverberg et al., 1983).
Synthetic pyrethroids act directly on the axon through
interference with the sodium channel gating mechanism that
underlies the generation and conduction of each nerve impulse. The
transitional state of the sodium channel is controlled by 2
separately acting gating mechanisms, referred to as the activation
gate and the inactivation gate. Since pyrethroids only appear to
affect the sodium current during depolarization, the rapid opening
of the activation gate and the slow closing of the inactivation
gate proceed normally. However, once the sodium channel is open,
the activation gate is restrained in the open position by the
pyrethroid molecule. While all pyrethroids have essentially the
same basic mechanism of action, however, the rate of relaxation
differs substantially for the various pyrethroids (Flannigan &
Tucker, 1985).
In the isolated node of Ranvier, allethrin causes prolongation
of the transient increase in sodium permeability of the nerve
membrane during excitation (Van den Bercken & Vijverberg, 1980).
Evidence so far available indicates that allethrin selectively
slows down the closing of the activation gate of a fraction of the
sodium channels that open during depolarization of the membrane.
The time constant of closing of the activation gate in the
allethrin-affected channels is about 100 milliseconds compared with
less than 100 microseconds in the normal sodium channel, i.e., it
is slowed down by a factor of more than 100. This results in a
marked prolongation of the sodium current across the nerve membrane
during excitation, and this prolonged sodium current is directly
responsible for the repetitive activity induced by allethrin
(Vijverberg et al., 1983).
The effects of cismethrin on synaptic transmission in the frog
neuromuscular junction, as reported by Evans (1976), are almost
identical to those of allethrin, i.e., presynaptic repetitive
firing, and no significant effects on transmitter release or on the
subsynaptic membrane.
Interestingly, the action of these pyrethroids closely
resembles that of the insecticide DDT in the peripheral nervous
system of the frog. DDT also causes pronounced repetitive activity
in sense organs, in sensory nerve fibres, and in motor nerve
terminals, due to a prolongation of the transient increase in
sodium permeability of the nerve membrane during excitation.
Recently was demonstrated that allethrin and DDT have essentially
the same effect on sodium channels in frog myelinated nerve
membrane. Both compounds slow down the rate of closing of a
fraction of the sodium channels that open on depolarization of the
membrane (Van den Bercken et al., 1973, 1979; Vijverberg et al.,
1982b).
In the electrophysiological experiments using giant axons of
crayfish, the type I pyrethroids and DDT analogues retain sodium
channels in a modified open state only intermittantly, cause large
depolarizing afterpotentials, and evoke repetitive firing with
minimal effect on the resting potential (Lund & Narahashi, 1983).
These results strongly suggest that permethrin and cismethrin,
like allethrin, primarily affect the sodium channels in the nerve
membrane and cause a prolongation of the transient increase in
sodium permeability of the membrane during excitation.
The effects of pyrethroids on end-plate and muscle action
potentials were studied in the pectoralis nerve-muscle preparation
of the clawed frog (Xenopus laevis). Type I pyrethroids
(allethrin, cismethrin, bioresmethrin, and 1R, cis-phenothrin)
caused moderate presynaptic repetitive activity, resulting in the
occurrence of multiple end-plate potentials (Ruigt & Van den
Bercken, 1986).
Pyrethroids with an alpha-cyano group on the 3-phenoxybenzyl
alcohol (deltamethrin, cypermethrin, fenvalerate, and fenpropanate)
(Type II: CS-syndrome)
The pyrethroids with an alpha-cyano group cause an intense
repetitive activity in the lateral line organ in the form of long-
lasting trains of impulses (Vijverberg et al., 1982a). Such a
train may last for up to 1 min and contains thousands of impulses.
The duration of the trains and the number of impulses per train
increase markedly on lowering the temperature. Cypermethrin does
not cause repetitive activity in myelinated nerve fibres. Instead,
this pyrethroid causes a frequency-dependent depression of the
nervous impulse, brought about by a progressive depolarization of
the nerve membrane as a result of the summation of depolarizing
after-potentials during train stimulation (Vijverberg & Van den
Bercken, 1979; Vijverberg et al., 1983).
In the isolated node of Ranvier, cypermethrin, like allethrin,
specifically affects the sodium channels of the nerve membrane and
causes a long-lasting prolongation of the transient increase in
sodium permeability during excitation, presumably by slowing down
the closing of the activation gate of the sodium channel
(Vijverberg & Van den Bercken, 1979; Vijverberg et al., 1983). The
time constant of closing of the activation gate in the
cypermethrin-affected channels is prolonged to more than 100
milliseconds. Apparently, the amplitude of the prolonged sodium
current after cypermethrin is too small to induce repetitive
activity in nerve fibres, but is sufficient to cause the long-
lasting repetitive firing in the lateral-line sense organ.
These results suggest that alpha-cyano pyrethroids primarily
affect the sodium channels in the nerve membrane and cause a long-
lasting prolongation of the transient increase in sodium
permeability of the membrane during excitation.
In the electrophysiological experiments using giant axons of
cray-fish, the Type II pyrethroids retain sodium channels in a
modified continuous open state persistently, depolarize the
membrane, and block the action potential without causing repetitive
firing (Lund & Narahashi, 1983).
Diazepam, which facilitates GABA reaction, delayed the onset of
action of deltamethrin and fenvalerate, but not permethrin and
allethrin, in both the mouse and cockroach. Possible mechanisms of
the Type II pyrethroid syndrome include action at the GABA receptor
complex or a closely linked class of neuroreceptor (Gammon et al.,
1982).
The Type II syndrome of intracerebrally administered pyrethroids
closely approximates that of the convulsant picrotoxin (PTX).
Deltamethrin inhibits the binding of [3H]-dihydropicrotoxin to rat
brain synaptic membranes, whereas the non-toxic R epimer of
deltamethrin is inactive. These findings suggest a possible
relation between the Type II pyrethroid action and the GABA receptor
complex. The stereospecific correlation between the toxicity of
Type II pyrethroids and their potency to inhibit the [35S]-TBPS
binding was established using a radioligand,
[35S]- t-butyl-bicyclophosphorothionate [35S]-TBPS. Studies
with 37 pyrethroids revealed an absolute correlation, without any
false positive or negative, between mouse intracerebral toxicity and
in vitro inhibition: all toxic cyano compounds including
deltamethrin, 1R, cis-cypermethrin, 1R, trans-cypermethrin, and
[2S, alpha S]-fenvalerate were inhibitors, but their non-toxic
stereoisomers were not; non-cyano pyrethroids were much less potent
or were inactive (Lawrence & Casida, 1983).
In the [35S]-TBPS and [3H]-Ro 5-4864 (a convulsant benzo-
diazepine radioligand) binding assay, the inhibitory potencies of
pyrethroids were closely related to their mammalian toxicities.
The most toxic pyrethroids of Type II were the most potent
inhibitors of [3H]-Ro 5-4864 specific binding to rat brain
membranes. The [3H]-dihydropicrotoxin and [35S]-TBPS binding
studies with pyrethroids strongly indicated that Type II effects of
pyrethroids are mediated, at least in part, through an interaction
with a GABA-regulated chloride ionophore-associated binding site.
Moreover, studies with [3H]-Ro 5-4864 support this hypothesis and,
in addition, indicate that the pyrethroid-binding site may be very
closely related to the convulsant benzodiazepine site of action
(Lawrence et al., 1985).
The Type II pyrethroids (deltamethrin, 1R, cis-cypermethrin and
[2S, alphaS]-fenvalerate) increased the input resistance of
crayfish claw opener muscle fibres bathed in GABA. In contrast,
two non-insecticidal stereoisomers and Type I pyrethroids
(permethrin, resmethrin, allethrin) were inactive. Therefore,
cyanophenoxybenzyl pyrethroids appear to act on the GABA receptor-
ionophore complex (Gammon & Casida, 1983).
The effects of pyrethroids on end-plate and muscle action
potentials were studied in the pectoralis nerve-muscle preparation
of the clawed frog (Xenopus laevis). Type II pyrethroids
(cypermethrin and deltamethrin) induced trains of repetitive muscle
action potentials without presynaptic repetitive activity.
However, an intermediate group of pyrethroids (1R-permethrin,
cyphenothrin, and fenvalerate) caused both types of effect. Thus,
in muscle or nerve membrane the pyrethroid induced repetitive
activities due to a prolongation of the sodium current. But no
clear distinction was observed between non-cyano and alpha-cyano
pyrethroids (Ruigt & Van den Bercken, 1986).
Appraisal
In summary, the results strongly suggest that the primary
target site of pyrethroid insecticides in the vertebrate nervous
system is the sodium channel in the nerve membrane. Pyrethroids
without an alpha-cyano group (allethrin, d-phenothrin, permethrin,
and cismethrin) cause a moderate prolongation of the transient
increase in sodium permeability of the nerve membrane during
excitation. This results in relatively short trains of repetitive
nerve impulses in sense organs, sensory (afferent) nerve fibres,
and, in effect, nerve terminals. On the other hand, the alpha-
cyano pyrethroids cause a long-lasting prolongation of the
transient increase in sodium permeability of the nerve membrane
during excitation. This results in long-lasting trains of
repetitive impulses in sense organs and a frequency-dependent
depression of the nerve impulse in nerve fibres. The difference in
effects between permethrin and cypermethrin, which have identical
molecular structures except for the presence of an alpha-cyano
group on the phenoxybenzyl alcohol, indicates that it is this
alpha-cyano group that is responsible for the long-lasting
prolongation of the sodium permeability.
Since the mechanisms responsible for nerve impulse generation
and conduction are basically the same throughout the entire nervous
system, pyrethroids may also induce repetitive activity in various
parts of the brain. The difference in symptoms of poisoning by
alpha-cyano pyrethroids, compared with the classical pyrethroids,
is not necessarily due to an exclusive central site of action.
It may be related to the long-lasting repetitive activity in
sense organs and possibly in other parts of the nervous system,
which, in a more advance state of poisoning, may be accompanied by
a frequency-dependent depression of the nervous impulse.
Pyrethroids also cause pronounced repetitive activity and a
prolongation of the transient increase in sodium permeability of
the nerve membrane in insects and other invertebrates. Available
information indicates that the sodium channel in the nerve membrane
is also the most important target site of pyrethroids in the
invertebrate nervous system (Wouters & Van den Bercken, 1978; WHO,
1979).
Because of the universal character of the processes underlying
nerve excitability, the action of pyrethroids should not be
considered restricted to particular animal species, or to a certain
region of the nervous system. Although it has been established
that sense organs and nerve endings are the most vulnerable to the
action of pyrethroids, the ultimate lesion that causes death will
depend on the animal species, environmental conditions, and on the
chemical structure and physical characteristics of the pyrethroid
molecule (Vijverberg & Van den Bercken, 1982).
1. RESUME ET EVALUATION, CONCLUSIONS ET RECOMMANDATIONS
1.1 Résumé et évaluation
1.1.1 Identité, propriétés physiques et chimiques, méthodes
d'analyse
La deltaméthrine a été synthétisée en 1974 et commercialisée
pour la première fois en 1977. Sur le plan chimique, c'est un
ester de l'analogue dibromé de l'acide chrysanthémique (acide
diméthyl-2,2(dimbromo-2,2 vinyl)-3 cyclopropanecarboxylique)
(Br2CA) et de l'alcol alpha-cyano-phénoxy-3 benzylique; plus
précisément, c'est l'isomère [1R, cis;alpha S] parmi les huit
stéréoisomères que compte cet ester.
La deltaméthrine de qualité technique se présente sous la forme
d'une poudre blanche inodore dont le point de fusion est de 98 -
101 °C et qui contient plus de 98% de deltaméthrine. Sa tension de
vapeur est de 2,0 x 10-6 Pa à 25 °C; autrement dit, elle n'est
pratiquement pas volatile. Insoluble dans l'eau, elle est en
revanche soluble dans des solvants organiques tels que l'acétone, la
cyclohexanone et le xylène. Elle est stable à la lumière, à la
chaleur et à l'air, mais instable en milieu alcalin.
Pour doser les résidus et analyser des échantillons prélevés
dans l'environnement, on procède à une extraction par solvant au
moyen d'un mélange n-hexane/acétone, à un partage entre le
n-hexane, l'acétone et l'eau, suivi d'une purification par
chromatographie sur colonne de gel de silice, le dosage final
s'effectuant par chromatographie en phase gazeuse avec détection
par capture d'électrons. La concentration minimale décelable par
cette méthode est de 0,01 mg/kg, voire moins. L'analyse des
produits s'effectue par chromatographie en phase liquide à haute
performance avec détecteur UV.
1.1.2 Production et usage
En 1987, la consommation mondiale de deltaméthrine était
d'environ 250 tonnes. On l'utilise essentiellement pour traiter le
coton (45% de la consommation) ainsi que pour les cultures telles
que le café, le maïs, les céréales, les fruits, les légumes, le
houblon et les produits stockés. On l'utilise aussi pour l'hygiène
des animaux, la lutte contre les vecteurs ainsi qu'à des fins de
santé publique. On l'applique soit seule, soit associée à d'autres
pesticides en formulations telles que concentrés émulsionnables,
concentrés pour pulvérisation à très bas volume, poudres
mouillables, concentrés pour suspension, poudres pour poudrage, etc.
1.1.3 Exposition humaine
L'exposition de la population dans son ensemble provient
principalement de la présence de résidus alimentaires mais peut
également résulter de son utilisation en santé publique. Sur les
récoltes correctement traitées, le taux de résidus est généralement
très faible sauf lorsque le traitement a eu lieu après la récolte.
La FAO et l'OMS ont passé en revue de nombreuses données sur ces
questions.
L'exposition de la population dans son ensemble est vrai-
semblablement très faible, mais on manque de données effectives
découlant d'études sur la ration alimentaire totale.
1.1.4 Exposition et destinée dans l'environnement
En exposant à la lumière solaire une fine pellicule de
deltaméthrine [1R, cis; alpha S] marquée au C14 au niveau de la
fonction acide, de la fonction alcool ou du groupe cyano, pendant 4
à 8 heures, on a constaté que 70 % du produit était transformé en
isomères [1R, trans alpha S] et [1S, trans alpha S] par
isomérisation cis/trans, accompagnés des produits de clivage de
l'ester, en particulier l'acide diméthyl-2,2(dibromovinyl-2,2)-3
cyclo-propane-carboxylique et l'alcool alpha-cyano-phénoxy-3
benzylique.
La deltaméthrine présente sur des plants de coton en serre se
dégrade avec une demi-vie initiale de 1,1 semaine, le taux de
dégradation étant de 90 % au bout de 4,5 semaines.
Les principaux métabolites consistent en Br2CA libre et
conjugué, en trans-hydroxyméthyl-Br2CA et en acide (hydroxy-4
phénoxy)-3 benzoïque résultant du clivage de l'ester, d'une
oxydation et d'une conjugaison.
Après avoir fait incuber de deltaméthrine dans du sable et du
terreau à 28 °C dans les conditions du laboratoire, on en a
récupéré respectivement 52 % et 74 %, huit semaines après ce
traitement.
La deltaméthrine ne se déplace pas dans l'environnement du fait
qu'elle est fortement adsorbée sur les particules, qu'elle est
insoluble dans l'eau et qu'elle est appliquée à très faibles doses.
On ne dispose pas de données sur les concentrations effectives
dans l'environnement, mais compte tenu des modalités actuelles
d'utilisation et pour peu que l'insecticide soit utilisé
normalement, l'exposition environnementale devrait être très
faible. La deltaméthrine se dégrade rapidement en produits moins
toxiques.
1.1.5 Absorption, métabolisme et excrétion
Administrée par voie orale, la deltraméthrine est facilement
absorbée; l'absorption est moindre par la voie percutanée encore
que sa vitesse dépende fortement du véhicule ou du solvant utilisé.
Une fois absorbée, la deltaméthrine est rapidement métabolisée et
excrétée.
On a administré à des rats par voie orale, à raison de 0,64 à
1,60 mg/kg, de la deltaméthrine marquée au carbone-14 au niveau des
fonctions acide, alcool et nitrile; le radiocarbone provenant de
la fraction acide et de la fraction alcoolique a été complètement
éliminé en l'espace de 2 à 4 jours. Les taux de résidus
tissulaires étaient généralement faibles sauf dans les graisses où
ils étaient un peu plus élevés. Cependant, le reste nitrile a été
excrété plus lentement, le taux global de récupération étant de
79 % en huit jours. Les principales réactions métaboliques étaient
l'oxydation (au niveau du méthyl- trans, du cycle cyclopropane et
des positions 2', 4' et 5' du reste alcoolique), le clivage de
l'ester et la conversion du nitrile en thiocyanate. Les acides
carboxyliques et les phénols résultants étaient conjugués à l'acide
sulfurique, à la glycine et à l'acide glucuronique.
Après avoir administré par voie orale à des souris de la
deltaméthrine marquée au 14C au niveau des fonctions acide, alcool
et nitrile à raison de 1,7 et 4,4 mg/kg, on a constaté que
l'excrétion du radiocarbone s'effectuait rapidement sauf dans le
cas du reste nitrile. Les principales réactions métaboliques
étaient en général analogues chez les souris et chez les rats.
Chez les vaches et la volaille, les voies de dégradation sont
tout à fait analogues à celles des rongeurs.
1.1.6 Effets sur les êtres vivants dans leur milieu naturel
La deltaméthrine est extrêmement toxique pour les poissons, la
CL50 à 96 heures allant de 0,4 à 2,0 µg/litre. Elle est également
très toxique pour les invertébrés aquatiques; pour la daphnie, la
CL50 à 48 heures est de 5 µg/litre. Toutefois des études
approfondies sur des étangs expérimentaux ainsi que les résultats
de l'utilisation en plein champ ont montré que cette forte toxicité
potentielle était inopérante. On a observé sur le terrain une
certaine mortalité parmi les invertébrés aquatiques mais les
populations se reconstituent généralement assez vite.
pour les oiseaux, la toxicité de la deltaméthrine est très
faible, les valeurs de la DL50 pour une dose unique par voie orale
étant supérieure à 1000 mg/kg. Au laboratoire, la deltaméthrine
est très toxique pour les abeilles, avec une DL50 de contact de
0,051 µg/abeille. Les essais en situation réelle et l'expérience
acquise dans l'utilisation effective du produit ont montré que les
formulations de deltaméthrine exerçaient une action répulsive, ce
qui signifie qu'en pratique le danger pour les abeilles est très
réduit.
1.1.7 Effets sur les animaux d'expérience et systemes d'epreuve
in vitro
Dans un véhicule non aqueux, la deltaméthrine présente une
toxicité aiguë par voie orale forte à modérée avec des DL50 de 19 à
34mg/kg chez la souris et de 39 à 139 mg/kg chez le rat.
Toutefois, en suspension dans l'eau, la toxicité est bien moidre,
les valeurs de la DL50 dépassant 5000 mg/kg chez le rat. La
deltaméthrine est un pyréthroide du Type II. Les signes cliniques
d'intoxication consistent en tremblements, salivation et
convulsions. L'intoxication est rapide et chez les survivants, les
signes disparaissent en quelques jours. L'électro-encéphalogramme
présente des décharges pointes-ondes généralisées qui précèdent
la choréo-athétose.
Une seule application de deltaméthrine technique n'a pas
produit d'effets irritants sur la peau intacte ou abrasée du lapin.
Toutefois elle a produit de effets irritants passagers au niveau de
l'oeil avec ou sans rinçage. La deltaméthrine n'a pas d'effet
sensibilisateur cutané chez le cobaye.
En administrant à des rats par gavage de la deltaméthrine à des
doses quotidiennes allant jusqu'à 10 mg/kg de poids corporel
pendant 13 semaines, on a provoqué chez ces animaux une
hyperexcitabilité qui se manifestait au bout de six semaines chez
les mâles recevant la dose la plus forte. Aux doses de 2,5 et
10 mg/kg, le gain de poids a été plus faible chez les mâles.
Chez des chiens beagle qui avaient reçu par voie orale de la
deltaméthrine en doses quotidiennes allant jusqu'à 10 mg/kg de
poids corporel pendant 13 semaines, on a observé divers symptômes
liés à cette substance, tels que des vomissements, des
tremblements, de la salivation et un affaiblissement du réflexe
pharyngé, du réflexe rotulien et du réflexe de flexion. Lors d'une
étude d'alimentation de deux ans sur des chiens, on a constaté que
la dose sans effet observable se situait à 1 mg/kg de poids
corporel par jour (dose la plus forte expérimentée).
L'administration de deltraméthrine à des souris à des doses
atteingnant 100 mg/kg de nourriture pendant 24 mois n'a pas modifié
l'incidence des tumeurs. La dose sans effet observé relative à la
toxicité générale était de 100 mg/kg de nourriture.
Chez des rats à qui l'on avait administré de la deltaméthrine
en doses allant jusqu'à 50 mg/kg de nourriture pendant deux ans, on
n'a observé aucune tumeur attribuable à cette substance. La dose
sans effet observé relative à la toxicité générale était de 5 mg/kg
de nourriture.
La deltaméthrine ne s'est pas révélée mutagène dans divers
systèmes d'épreuve in vivo et in vitro; notamment la réparation de
l'ADN, la mutation génique, les aberrations chromosomiques,
l'échange entre chromatides-soeurs, la formation de micronoyaux et
la létalité dominante.
Des études de tératogénicité ont été effectuées chez des rates
et des souris gravides à qui l'on administrait par voie orale des
doses quotidiennes de deltaméthrine allant jusqu'à 1000 mg/kg
pendant la phase principale de l'organogénèse. On n'a constaté
aucun effet tératogène ni altération de la fonction de reproduction
chez ces rates et ces souris, si ce n'est une réduction, liée à la
dose, dans le poids moyen des foetus chez les souris et un léger
retard d'ossification chez les rats.
Des lapines gravides ont reçu au sixième et dix-neuvième jours
de leur grossesse des doses quotidiennes de deltaméthrine allant
jusqu'à 16 mg/kg. A la dose la plus forte, on a noté une réduction
du poids moyen des foetus. Aucun effet tératogène n'a été observé
chez les lapins.
Après administration de deltaméthrine à des rats à des doses
allant jusqu'à 5 mg/kg de nourriture, dans le but d'effectuer une
étude de reproduction sur trois générations et deux portées, on a
constaté l'absence totale de tout effet.
Il existe certains indices selon lesquels l'association de
delta- méthrine à certains composés organophosphorés pourrai
conduire à une potentialisation de la toxicité de ces produits.
1.1.8 Effets sur l'homme
La deltaméthrine peut provoquer certaines sensations cutanées
chez des travailleurs exposés. Plusieurs cas d'intoxication
professionnelle non mortelle ont été signalés qui étaient dus à
l'inobservation des mesures de sécurité. Un engourdissement, un
prurit, des fourmillements et une sensation de brûlure de la peau
ainsi que des vertiges sont des symptômes fréquemment signalés. On
a décrit occasionnellement un érythème papulaire ou une couperose à
caractère passager. La plupart de ces symptômes sont passagers et
disparaissent en cinq à sept jours. Aucun effet indésirable à long
terme n'a été signalé. On a décrit trois cas d'empoisonnement non
mortel par la deltamétrine à la suite de l'ingestion de plusieurs
grammes de ce produit.
1.2 Conclusions
Population générale: l'exposition de la population générale à la
deltaméthrine est vraisemblablement très faible et ce produit,
lorsqu'il est utilisé conformément aux recommandations, ne présente
probablement aucun risque.
Exposition professionnelle: moyennant de bonnes méthodes de
travail et l'application de mesures d'hygiène et de sécurité, la
deltaméthrine ne devrait pas présenter de danger pour les personnes
qui y sont exposées de par leur profession.
Environnement: Utilisée aux doses recommandées, il est improbable
que la deltaméthrine ou ses produits de dégradation s'accumulent au
point d'avoir des effets nocifs sur l'environnement. Au
laboratoire, la deltaméthrine est très toxique pour les poissons,
les arthropodes aquatiques et les abeilles. Toutefois, sur le
terrain, il est peu probable que le produit ait des effets nocifs
s'il est utilisé conformément aux recommandations.
1.3 Recommandations
Bien que les teneurs dans les aliments soient très faibles
lorsque la deltaméthrine est utilisée conformément aux
recommandations, il est souhaitable de s'en assurer en soumettant
la deltaméthrine à une surveillance.
La deltaméthrine est utilisée depuis de nombreuses années et
l'on signale un certain nombre de cas d'intoxications non mortelles
ainsi que des effets passagers par suite d'exposition
professionnelle. Il est souhaitable que l'exposition humaine
continue d'être surveillée.
1. RESUMEN Y EVALUACION, CONCLUSIONES Y RECOMENDACIONES
1.1 Resumen y evaluación
1.1.1 Identidad, propiedades fíisicas y químicas, métodos de
análisis
La deltametrina fue sintetizada en 1974 y se comercializó por
primera vez en 1977. Químicamente, es el isómero [1R, cis; S] de 8
ésteres estereoisoméricos del análogo dibromo del ácido
crisantémico, ácido 2,2-dimetil-3-(2,2-dibromovinil) ciclopro-
panocarboxílico (Br2CA) con alcohol alpha-ciano-3-fenoxibencílico.
La deltametrina de calidad técnica es un polvo blanco inodoro
con un punto de fusión de 98 - 101 °C y contiene más del 98% del
material. La presión del vapor es 2,0 x 10-6 Pa a 25 °C y es
prácticamente no volátil. Es insoluble en agua, pero soluble en
disolventes orgánicos como la acetona, la ciclohexanona y el
xileno. Es estable a la luz, el calor y el aire, pero inestable en
medios alcalinos.
La determinación de la presencia de residuos y el análisis de
muestras ambientales se efectuaron por extracción con los
disolventes n-hexano/acetona, partición con n-hexano/acetona/agua,
absorción con un cromatógrafo de columna de gel de sílice y
determinación con un cromatógrafo de fase gaseosa equipado con un
detector de captura de electrones con una concentración mínima
detectable de 0,01 mg/kg o menos. Para el análisis de productos se
utiliza la cromatografía de fase líquida de alto rendimiento con un
detector de rayos UV.
1.1.2 Producción y empleo
El consumo mundial de deltametrina era de 250 toneladas
aproximadamente en 1987. La deltametrina se utiliza sobre todo en
el algodón (45% del consumo), en cultivos como el café, el maíz,
los cereales, la fruta, las hortalizas y el lúpulo, y en productos
almacenados. Se emplea también en higiene animal, en la lucha
contra los vectores y en salud pública. Se fabrica como solución
concentrada emulsionable, solución concentrada de pequeñísimo
volumen, polvo humectable, solución concentrada en suspensión y
polvo para utilización en seco, sola o en combinación con otros
plaguicidas.
1.1.3 Exposición humana
La exposición de la población en general a la deltametrina
procede principalmente de residuos en la dieta, pero puede resultar
también de su empleo en salud pública. En los cultivos tratados de
acuerdo con buenas prácticas agrícolas, las concentraciones
residuales son por lo general muy bajas, excepto en caso de
tratamiento después de la recolección. La FAO y la OMS han
examinado abundantes datos al respecto.
Se cree que la exposición de la población en general es muy
baja, pero no se dispone de datos en forma de estudios de dietas
totales.
1.1.4 Exposición ambiental y evolución
Cuando la deltametrina-[1R, cis; alpha S] marcada con
C14-(compuesto irradiado ácido, alcohol o ciano) se expuso a la
luz del sol en forma de una película fina durante 4 a 8 horas, el
70% se transformó por isomerización- cis/trans produciendo los
isómeros [1R, trans; alpha S] y [1S, trans; alpha S], junto con
productos de la escisión de los ésteres, entre ellos Br2CA y
alcohol alpha-ciano-3-fenoxibencílico.
La deltametrina se degradó en las plantas de algodón, en
condiciones de invernadero, con una vida media inicial de 1,1
semanas, y el tiempo necesario para una pérdida del 90% fue de 4,6
semanas.
Los principales metabolitos fueron Br2CA libre y combinado,
trans-hidroximetil-Br2CA y ácido 3-(4-hidroxifenoxi)benzoico,
formados por la escisión, la oxidación y la combinación de los
ésteres.
La deltametrina se incubó en arena y mantillo orgánico a 28 °C
en condiciones de laboratorio. Ocho semanas después del
tratamiento, se recuperó de la arena y del mantillo orgánico
alrededor del 52% y el 74%, respectivamente, de la deltametrina
aplicada.
La deltametrina no es móvil en el medio debido a su fuerte
adsorción en las partículas, su insolubilidad en el agua y sus
tasas muy bajas de aplicación.
No se dispone de datos sobre las concentraciones existentes en
el medio pero, dadas las modalidades actuales de uso y en
condiciones normales, es de esperar que la exposición ambiental sea
muy baja. Se degrada rápidamente, convirtiéndose en productos
menos tóxicos.
1.1.5 Ingestion, metabolismo y excreción
La deltametrina se absorbe fácilmente por vía oral y con menor
facilidad por la piel, pero la tasa de absorción depende
considerablemente del portador o disolvente. La deltametrina
absorbida se metaboliza y se excreta fácilmente.
En la administración por vía oral a ratas de deltametrina
marcada con C14-(compuesto irradiado ácido, alcohol o ciano), a
tasas de 0,64 - 1,60 mg/kg, el radiocarbono de las partes ácido y
alcohol se eliminó casi por completo en 2 - 4 días. Las
concentraciones residuales en los tejidos fueron por lo general muy
bajas, excepto en la grasa, en donde fueron ligeramente más altas.
Sin embargo, la parte ciano se excretó más lentamente, con una
recuperación total del 79% en 8 días. Las principales reacciones
metabólicas fueron oxidación (en el trans-metilo del anillo de
ciclopropano y en las posiciones 2'-, 4'-, y 5 de la parte
alcohol), escisión de ésteres y conversión de la parte ciano en
tiocianato. Los ácidos carboxílicos y fenoles resultantes se
combinaron con ácido sulfúrico, glicocola y ácido glucurónico.
Cuando se administró a ratones C14-(compuesto irradiado ácido,
alcohol o ciano)-deltametrina por vía oral, a tasas de 1,7 - 4,4
mg/kg, la excreción del radiocarbono fue rápida y casi total,
excepto en la parte ciano. Las principales reacciones metabólicas
observadas en los ratones fueron en general semejantes a las
observadas en las ratas.
En el ganado vacuno y las aves de corral, las vías de
degradación son muy similares a las observadas en los roedores.
1.1.6 Efectos en los organismos presentes en el medio
La deltametrina es muy tóxica para los peces, ya que la CL50 en
96 horas oscila entre 0,4 y 2,0 µg/litro. Es también muy tóxica
para los invertebrados acuáticos: la CL50 en 48 horas para Daphnia
es de 5 µg/litro. Sin embargo, extensos estudios realizados en
estanques experimentales y el empleo sobre el terreno han
demostrado que esa elevada toxicidad potencial no llega a
concretarse. En la práctica hay algunas muertes de invertebrados
acuáticos que, por lo general, se compensan con rapidez.
La toxicidad de la deltametrina en las aves es muy baja, con
valores de la DL50 superiores a 1000 mg/kg cuando se administra una
sola dosis por vía oral. En condiciones de laboratorio, la
deltametrina es muy tóxica para las abejas, con una DL50 por
contacto de 0,051 µg/abeja. Los ensayos sobre el terreno y el
empleo no experimental han demostrado que los preparados de
deltametrina tienen una acción repelente, lo cual quiere decir que,
en la práctica, el riesgo para las abejas es escaso.
1.1.7 Efectos en animales experimentales y en sistemas de pruebas
in vitro
En un vehículo no acuoso, la toxicidad de la deltametrina
administrada por vía oral en forma aguda va de alta a moderada, con
DL50 de 19 a 34 mg/kg (ratones) y de 31 a 139 mg/kg (ratas). Sin
embargo, cuando la deltametrina está en suspensión en agua, su
toxicidad es mucho menor con DL50 superiores a 5000 mg/kg (ratas).
La deltametrina es un piretroide de tipo II; los signos clínicos
comprenden temblor, salivación y convulsiones. La intoxicación se
presenta rápidamente y, en los supervivientes, los signos
desaparecen en unos días. El electroencefalograma muestra
descargas generalizadas en picos y ondas, seguidas de
coreoatetosis.
Aplicaciones únicas de deltametrina técnica no tuvieron ningún
efecto irritante en la piel, intacta o con abrasiones, de conejos.
Sin embargo, se observaron efectos irritantes transitorios en los
ojos de conejos, con y sin lavado. La deltametrina no actuó como
sensibilizante dérmico en cobayas.
En ratas a las que se administró deltametrina con sonda, en
dosis de hasta 10,0 mg/kg de peso corporal diarios durante 13
semanas, a las 6 semanas se observó hiperexcitabilidad en los
machos que habían recibido la dosis más alta. El aumento del peso
corporal en los machos fue menor con dosis de 2,5 y 10 mg/kg.
En perros de la raza beagle a los que se administró por vía
oral deltametrina en dosis de hasta 10 mg/kg de peso corporal
diarios durante 13 semanas, se observaron varios síntomas
relacionados con el compuesto, como vómitos, temblor, salivación y
reflejos faríngeo, patelar y flexor deprimidos. En un estudio de
alimentación en perros, de dos años de duración, la dosis
desprovista de efectos fue de 1 mg/kg de peso corporal diario
(dosis más alta estudiada).
En ratones a los que se administró deltametrina en dosis de
hasta 100 mg/kg de alimentos durante 24 meses, no resultó afectada
la incidencia de tumores. En cuanto a la toxicidad general, la
dosis desprovista de efectos fue de 100 mg/kg de alimentos.
En ratas a las que se administró deltametrina a niveles de
hasta 50 mg/kg de alimentos durante dos años, no se observaron
tumores relacionados con el compuesto. La concentración
desprovista de efectos de la toxicidad general fue de 50 mg/kg de
alimentos.
No se observaron efectos mutagénicos de la deltametrina en una
multitud de sistemas de pruebas in vivo e in vitro, incluidos:
reparación del ADN, mutación génica, aberración cromosómica,
intercambio de cromátidas hermanas, formación de micronúcleos y
genes letales dominantes.
Se realizaron estudios teratológicos con ratas y ratonas
preñadas, administrando deltametrina por vía oral en dosis de hasta
10 mg/kg diarias durante el periodo de mayor organogénesis. No se
observaron efectos teratogénicos ni reproductivos en las ratas ni
en las ratonas, excepto una disminución, relacionada con la dosis,
del peso fetal medio en el estudio con ratonas y una osificación
ligeramente retrasada en el estudio con ratas.
Se administró a conejas deltametrina en dosis de hasta 16 mg/kg
diarios entre los días 6 y 19 del embarazo. Con la dosis más alta,
se registró una disminución del peso fetal medio. No se observaron
efectos teratogénicos.
Se administró a ratas deltametrina en dosis de hasta 50 mg/kg
de alimentos en un estudio sobre reproducción con tres generaciones
y dos camadas, sin que se observaran efectos sobre la reproducción.
Hay indicios de que la toxicidad puede potenciarse cuando la
deltametrina se combina con algunos compuestos organofosforados.
1.1.8 Efectos en los seres humanos
La deltametrina puede provocar sensaciones cutáneas en los
trabajadores expuestos. Se han notificado varios casos de
intoxicación no mortal debidos a exposición ocupacional por no
respetar las precauciones de seguridad. Son síntomas
frecuentemente mencionados adormecimiento, picor, hormigueo y
quemazón de la piel, y vértigo. En ocasiones se ha descrito un
eritema papular o maculoso. La mayor parte de esos síntomas son
temporales y desaparecen en 5 ó 7 días. No se han comunicado
efectos negativos a largo plazo. Se han descrito tres casos no
mortales de intoxicación por deltametrina tras la ingestión de
varios gramos del producto.
1.2 Conclusiones
Población en general: No es probable que la exposición de la
población en general a la deltametrina, que se cree muy baja,
represente un riesgo en las condiciones de empleo recomendadas.
Exposición ocupacional: Si se aplican buenas prácticas laborales,
medidas de higiene y precauciones de seguridad, no es probable que
la deltametrina represente un riesgo para las personas
ocupacionalmente expuestas a ella.
Medio ambiente: Es improbable que la deltametrina o los productos
de su degradación alcancen niveles que puedan tener efectos
negativos en el medio con las tasas de aplicación recomendadas. En
condiciones de laboratorio, la deltametrina es muy tóxica para los
peces, los artrópodos acuáticos y las abejas. No obstante, en la
práctica, no es probable que haya efectos perjudiciales duraderos
con las condiciones de empleo recomendadas.
1.3 Recomendaciones
Aunque se cree que con el uso recomendado las concentraciones
en la dieta son muy bajas, debe considerarse la conveniencia de
confirmarlo incluyendo la deltametrina en estudios de vigilancia.
La deltametrina se utiliza desde hace muchos años y se han
notificado varios casos de intoxicación no mortal y efectos
transitorios debidos a exposición ocupacional. Debe mantenerse la
observación de la exposición humana.

 Deltamethrin is the first pyrethroid composed of a single
isomer of 8 stereoisomers selectively prepared by the
esterification of [1R, 3R or cis]-2,2-dimethyl-3-(2,2-dibromovinyl)
cyclopropanecarboxylic acid with (alphaS)- or (+)-alpha-cyano-3-
phenoxybenzyl alcohol or by selective recrystallization of the
racemic esters obtained by esterification of the (1R, 3R or cis)-
acid with the racemic or [alphaR, alphaS, or alphaRS or ±]-alcohol
(Elliott et al., 1974). Thus, its stereospecific structure (4) is
the ester of [1R, 3R or cis]-acid with (alphaS)-alcohol.
The acid is a characteristic dibromo analogue of chrysanthemic
acid.
2.2 Physical and Chemical Properties
Technical grade deltamethrin contains more than 98% deltamethrin
(FAO/WHO, 1981). It is stable to heat (6 months at 40 °C), light,
and air, but unstable in alkaline media (FAO/WHO, 1981; Meister et
al., 1983; Worthing & Walker, 1983). Some physical and chemical
properties are listed in Table 1, and the chemical composition of
various stereoisomeric mixtures is shown in Table 2.
Table 1. Some physical and chemical properties of deltamethrin
-------------------------------------------------------------------
Physical state crystalline powder
Colour colourless
Odour odourless
Density (20 °C) 0.5 g/cm3
Relative molecular mass 505.24
Melting point (°C) 98-101
Boiling point decomposes above 300 °C
Water solubility (20 °C) < 0.002 mg/litre (practically insoluble)
Solubility in organic solublea
solvents
Vapour pressure (25 °C) 2.0 x 10-6 Pa
n-Octanol-water 5.43
partition coefficient
(Log Pow)
-------------------------------------------------------------------
a Acetone (500 g/litre), ethanol (15 g/litre), cyclohexanone (750
g/litre), dioxane (900 g/litre), xylene (250 g/litre), ethyl
acetate.
2.3 Analytical Methods
Methods for the determination of deltamethrin residues and the
analysis of environmental samples, and products are summarized in
Table 3.
To analyse technical grade deltamethrin, a mixture of
deltamethrin and diphenylamine (an internal standard) was injected
in a high-performance liquid chromatograph equipped with a UV-
detector (Mourot et al., 1979).
The Joint FAO/WHO Codex Alimentarius Commission has published
recommendations for methods for the determination of deltamethrin
residues (FAO/WHO 1985b). A further review of analytical methods
for deltamethrin has been made by Vaysse et al. (1984).
Table 2. Chemical identity of deltamethrins of various stereoisomeric compositions
---------------------------------------------------------------------------------------------------------------
Common name CA Index name (9CI) Stereoisomeric Synonyms and trade names
CAS Registry No. compositionc
NIOSH Accession No.a Stereospecific nameb
---------------------------------------------------------------------------------------------------------------
Deltamethrin Cyclopropanecarboxylic acid, (4) Decamethrin, Decis,
Deltamethrin is the first pyrethroid composed of a single
isomer of 8 stereoisomers selectively prepared by the
esterification of [1R, 3R or cis]-2,2-dimethyl-3-(2,2-dibromovinyl)
cyclopropanecarboxylic acid with (alphaS)- or (+)-alpha-cyano-3-
phenoxybenzyl alcohol or by selective recrystallization of the
racemic esters obtained by esterification of the (1R, 3R or cis)-
acid with the racemic or [alphaR, alphaS, or alphaRS or ±]-alcohol
(Elliott et al., 1974). Thus, its stereospecific structure (4) is
the ester of [1R, 3R or cis]-acid with (alphaS)-alcohol.
The acid is a characteristic dibromo analogue of chrysanthemic
acid.
2.2 Physical and Chemical Properties
Technical grade deltamethrin contains more than 98% deltamethrin
(FAO/WHO, 1981). It is stable to heat (6 months at 40 °C), light,
and air, but unstable in alkaline media (FAO/WHO, 1981; Meister et
al., 1983; Worthing & Walker, 1983). Some physical and chemical
properties are listed in Table 1, and the chemical composition of
various stereoisomeric mixtures is shown in Table 2.
Table 1. Some physical and chemical properties of deltamethrin
-------------------------------------------------------------------
Physical state crystalline powder
Colour colourless
Odour odourless
Density (20 °C) 0.5 g/cm3
Relative molecular mass 505.24
Melting point (°C) 98-101
Boiling point decomposes above 300 °C
Water solubility (20 °C) < 0.002 mg/litre (practically insoluble)
Solubility in organic solublea
solvents
Vapour pressure (25 °C) 2.0 x 10-6 Pa
n-Octanol-water 5.43
partition coefficient
(Log Pow)
-------------------------------------------------------------------
a Acetone (500 g/litre), ethanol (15 g/litre), cyclohexanone (750
g/litre), dioxane (900 g/litre), xylene (250 g/litre), ethyl
acetate.
2.3 Analytical Methods
Methods for the determination of deltamethrin residues and the
analysis of environmental samples, and products are summarized in
Table 3.
To analyse technical grade deltamethrin, a mixture of
deltamethrin and diphenylamine (an internal standard) was injected
in a high-performance liquid chromatograph equipped with a UV-
detector (Mourot et al., 1979).
The Joint FAO/WHO Codex Alimentarius Commission has published
recommendations for methods for the determination of deltamethrin
residues (FAO/WHO 1985b). A further review of analytical methods
for deltamethrin has been made by Vaysse et al. (1984).
Table 2. Chemical identity of deltamethrins of various stereoisomeric compositions
---------------------------------------------------------------------------------------------------------------
Common name CA Index name (9CI) Stereoisomeric Synonyms and trade names
CAS Registry No. compositionc
NIOSH Accession No.a Stereospecific nameb
---------------------------------------------------------------------------------------------------------------
Deltamethrin Cyclopropanecarboxylic acid, (4) Decamethrin, Decis,
 When 14C-deltamethrin-[1R, 3R; alphaS] (9) labelled at the
cyano, benzylic, or dibromo-substituted carbon was exposed to
sunlight as a thin film (40 µg/cm2) for 4 - 8 h, the trans-[1R,
3S; alphaS] and -[1S, 3R; alphaS] isomers were formed. They
accounted for approximately 70% of the applied radioactivity.
Smaller amounts of ester cleavage products including the
2,2-dimethyl-3-(2,2-dibromovinyl) cyclopropanecarboxylic acid
(Br2CA) (18) and the cyanohydrin component, and 18% of
unidentified products were also formed (Fig. 1). In a thick film (3
mg/cm2), small amounts of other products including
alpha-cyano-3-phenoxybenzyl 3,3-dimethylacrylate (13) and 3-phenoxy
2,2-dimethyl-3-(2,2- dibromovinyl)cyclopropan-1-yl-benzylcyanide
(14) (decarboxydeltamethrin) were also detected. In contrast, the
predominant products in methanol were the trans mixtures, which
amounted to approximately 35% of the applied radioactivity. Under
UV radiation (peak output 290 - 320 nm), the photodegradation rate
of deltamethrin in alcohols decreased in the order of methanol,
ethanol, and 2-propanol, as the solvent viscosity increased. The
relative photolysis rates in hexane and cyclohexane, with respective
relative viscosities of 0.33 and 1, were 1.5 and 1. There was no
difference in the extent of the reaction on flushing the hexane with
O2 or N2, while the triplet quenchers piperilene and
1,3-cyclohexadiene reduced the reaction rate in hexane.
At 30 - 50% conversion, the trans-[1R, 3S; alphaS] and -[1S,
3R; alphaS] isomers were the major photoproducts in aqueous
acetonitrile, whereas they were observed in only minor amounts in
methanol and were absent in hexane. The mono-debrominated esters
(16) were the major ester products in methanol and hexane. The
cis-acid (18) was always the major photoproduct from the acid
moiety, with smaller amounts of the two isomeric debrominated acids
(17).
Major products from the alcohol moiety were 3-phenoxybenzoic
acid (25) (PBacid) in aqueous acetonitrile, 3-phenoxybenzyl cyanide
(15) in hexane, and methyl 3-phenoxybenzoate (22) in methanol.
Photolysis of 3-phenoxybenzoyl cyanide (21) gave methyl
3-phenoxybenzoate and the methyl ester of Br2CA (19) in methanol
and PBacid in aqueous acetonitrile. Thus, it appears that the
photoproducts obtained originated from cyclopropane ring opening
and various recombinations, scission of the ester oxygen-benzyl
carbon bond, scission of the acyl-oxygen bond, and/or reductive
debromination (Ruzo et al., 1976, 1977).
A photodegradation study with 14C-deltamethrin in aqueous
solution showed that such a solution, at pH 5, is hydrolytically
stable. When exposed to simulated sunlight, degradation was
induced. The primary product observed was PBacid. A half-life of
47.7 days was calculated for the non-sensitized system, but this
was reduced to 4.03 days when sensitized with 1% acetone.
Practically no volatile degradation products were observed (Bowman
& Carpenter, 1987).
4.3 Environmental Fate
The degradation and persistence of 14C-cyano- and 14C-phenoxy-
deltamethrin was examined in a Dubbs fine sandy loam and a Memphis
silt loam under aerobic laboratory conditions at 25 °C (Kaufman &
Kayser 1979a); 14C-deltamethrin was applied at final concentrations
equivalent to 0.02 and 0.2 kg/ha. Deltamethrin degradation
occurred rapidly in both soils with 62 - 77% and 52 - 60% of the
14C-cyano- and 14C-phenoxy-labels, respectively, being evolved as
14CO2 during the 128-day incubation period. The half-life of
deltamethrin varied from 11 to 19 days in the two soil types.
The effect of soil temperature on the degradation of
deltamethrin was also examined in Dubbs fine sandy loam under
laboratory conditions using 14C-cyano- and 14C-vinyl-labelled
deltamethrin (Kaufman & Kayser 1979b). Degradation and evolution
of 14C-labelled forms of deltamethrin occurred most rapidly at
25 °C and most slowly in soils incubated at 10 °C. The half-life
of deltamethrin was 46, 13, and 27 days in soils incubated at 10,
25, and 40 °C, respectively.
The results of these two studies indicate that deltamethrin
degradation occurs by two principal pathways (Fig. 2): hydrolysis
of the ester linkage to yield Br2CA (18) and 3-phenoxybenzoic acid;
and hydrolysis of the cyano group to yield first the amide, and
subsequently the carboxylic acid (DCOOH) analogues of deltamethrin.
Br2CA accumulated to a maximum of 5.7% of the original 14C in soil
incubated at 40 °C, whereas DCOOH accumulated at 10 °C (to a
maximum of 5.3%). However, both products decreased in
concentration by the end of the 64-day incubation period. In the
first experiment, DCOOH was also identified as the major
degradation product to reach a maximum concentration of 6 - 9% of
the original 14C. But it ultimately dissipated to less than 2% at
the end of the 128-day incubation period.
From the 14C-phenoxy label, 3-phenoxybenzoic acid (PBacid) was
identified as the main degradation product resulting from
hydrolysis of the ester bond. This product was further degraded to
yield both 3-(2-hydroxyphenoxy)-benzoic acid and 3-(4-hydroxyphenoxy)
benzoic acid. In this experiment, DCOOH was the only deltamethrin
degradation product detected in excess of 3% of the original material
applied.
Although essentially no radiolabel was detected in the leachate
from soil columns treated with 14C-deltamethrin, PBacid produced by
degradation of deltamethrin was fairly mobile in the soil columns
(Kaufman et al., 1981).
The degradation pathways are proposed in Fig. 2.
When 14C-deltamethrin-[1R, 3R; alphaS] (9) labelled at the
cyano, benzylic, or dibromo-substituted carbon was exposed to
sunlight as a thin film (40 µg/cm2) for 4 - 8 h, the trans-[1R,
3S; alphaS] and -[1S, 3R; alphaS] isomers were formed. They
accounted for approximately 70% of the applied radioactivity.
Smaller amounts of ester cleavage products including the
2,2-dimethyl-3-(2,2-dibromovinyl) cyclopropanecarboxylic acid
(Br2CA) (18) and the cyanohydrin component, and 18% of
unidentified products were also formed (Fig. 1). In a thick film (3
mg/cm2), small amounts of other products including
alpha-cyano-3-phenoxybenzyl 3,3-dimethylacrylate (13) and 3-phenoxy
2,2-dimethyl-3-(2,2- dibromovinyl)cyclopropan-1-yl-benzylcyanide
(14) (decarboxydeltamethrin) were also detected. In contrast, the
predominant products in methanol were the trans mixtures, which
amounted to approximately 35% of the applied radioactivity. Under
UV radiation (peak output 290 - 320 nm), the photodegradation rate
of deltamethrin in alcohols decreased in the order of methanol,
ethanol, and 2-propanol, as the solvent viscosity increased. The
relative photolysis rates in hexane and cyclohexane, with respective
relative viscosities of 0.33 and 1, were 1.5 and 1. There was no
difference in the extent of the reaction on flushing the hexane with
O2 or N2, while the triplet quenchers piperilene and
1,3-cyclohexadiene reduced the reaction rate in hexane.
At 30 - 50% conversion, the trans-[1R, 3S; alphaS] and -[1S,
3R; alphaS] isomers were the major photoproducts in aqueous
acetonitrile, whereas they were observed in only minor amounts in
methanol and were absent in hexane. The mono-debrominated esters
(16) were the major ester products in methanol and hexane. The
cis-acid (18) was always the major photoproduct from the acid
moiety, with smaller amounts of the two isomeric debrominated acids
(17).
Major products from the alcohol moiety were 3-phenoxybenzoic
acid (25) (PBacid) in aqueous acetonitrile, 3-phenoxybenzyl cyanide
(15) in hexane, and methyl 3-phenoxybenzoate (22) in methanol.
Photolysis of 3-phenoxybenzoyl cyanide (21) gave methyl
3-phenoxybenzoate and the methyl ester of Br2CA (19) in methanol
and PBacid in aqueous acetonitrile. Thus, it appears that the
photoproducts obtained originated from cyclopropane ring opening
and various recombinations, scission of the ester oxygen-benzyl
carbon bond, scission of the acyl-oxygen bond, and/or reductive
debromination (Ruzo et al., 1976, 1977).
A photodegradation study with 14C-deltamethrin in aqueous
solution showed that such a solution, at pH 5, is hydrolytically
stable. When exposed to simulated sunlight, degradation was
induced. The primary product observed was PBacid. A half-life of
47.7 days was calculated for the non-sensitized system, but this
was reduced to 4.03 days when sensitized with 1% acetone.
Practically no volatile degradation products were observed (Bowman
& Carpenter, 1987).
4.3 Environmental Fate
The degradation and persistence of 14C-cyano- and 14C-phenoxy-
deltamethrin was examined in a Dubbs fine sandy loam and a Memphis
silt loam under aerobic laboratory conditions at 25 °C (Kaufman &
Kayser 1979a); 14C-deltamethrin was applied at final concentrations
equivalent to 0.02 and 0.2 kg/ha. Deltamethrin degradation
occurred rapidly in both soils with 62 - 77% and 52 - 60% of the
14C-cyano- and 14C-phenoxy-labels, respectively, being evolved as
14CO2 during the 128-day incubation period. The half-life of
deltamethrin varied from 11 to 19 days in the two soil types.
The effect of soil temperature on the degradation of
deltamethrin was also examined in Dubbs fine sandy loam under
laboratory conditions using 14C-cyano- and 14C-vinyl-labelled
deltamethrin (Kaufman & Kayser 1979b). Degradation and evolution
of 14C-labelled forms of deltamethrin occurred most rapidly at
25 °C and most slowly in soils incubated at 10 °C. The half-life
of deltamethrin was 46, 13, and 27 days in soils incubated at 10,
25, and 40 °C, respectively.
The results of these two studies indicate that deltamethrin
degradation occurs by two principal pathways (Fig. 2): hydrolysis
of the ester linkage to yield Br2CA (18) and 3-phenoxybenzoic acid;
and hydrolysis of the cyano group to yield first the amide, and
subsequently the carboxylic acid (DCOOH) analogues of deltamethrin.
Br2CA accumulated to a maximum of 5.7% of the original 14C in soil
incubated at 40 °C, whereas DCOOH accumulated at 10 °C (to a
maximum of 5.3%). However, both products decreased in
concentration by the end of the 64-day incubation period. In the
first experiment, DCOOH was also identified as the major
degradation product to reach a maximum concentration of 6 - 9% of
the original 14C. But it ultimately dissipated to less than 2% at
the end of the 128-day incubation period.
From the 14C-phenoxy label, 3-phenoxybenzoic acid (PBacid) was
identified as the main degradation product resulting from
hydrolysis of the ester bond. This product was further degraded to
yield both 3-(2-hydroxyphenoxy)-benzoic acid and 3-(4-hydroxyphenoxy)
benzoic acid. In this experiment, DCOOH was the only deltamethrin
degradation product detected in excess of 3% of the original material
applied.
Although essentially no radiolabel was detected in the leachate
from soil columns treated with 14C-deltamethrin, PBacid produced by
degradation of deltamethrin was fairly mobile in the soil columns
(Kaufman et al., 1981).
The degradation pathways are proposed in Fig. 2.
 The degradation of deltamethrin was also examined under
anaerobic conditions using 14C-cyano-, 14C-phenoxy-, and
14C-vinyl-labelled materials for the tests (Kaufman & Kayser,
1980). Under anaerobic conditions, 14CO2 evolution varied
according to the 14C label position and the time of flooding.
Generally, flooding reduced or initially inhibited the rate of
14CO2 dissipation. However, after one month, 14CO2 dissipation
started again, which suggested the presence of a unique microbial
flora. It was also shown that all three carboxylic acids that
accumulate initially in flooded soils are subsequently further
degraded. Some reduction of PBacid to 3-phenoxybenzyl alcohol
(PBalc) was also observed in these flooded soils.
When deltamethrin was applied to a sandy clay loam soil at
17.5 g/ha in an indoor incubation study and in two field
experiments, the half-lives of deltamethrin were found to be 4.9
and 6.9 weeks under indoor and field conditions, respectively
(Hill, 1983). This difference in the rate of decrease in the
residue was attributed to climatic effects.
This was further confirmed by Hill & Schaalje (1985) who
pointed out a first-order dissipation, if degree-days above 0 °C
rather than days was used as the independent variable, when
deltamethrin was applied by pipette to soils. When deltamethrin
was boom-sprayed, a biphasic first-order plot was observed. A two-
compartment model that predicts an initial fast loss of residue
followed by a slower first-order degradation gave a good fit of the
data.
Chapman & Harris (1981) examined the relative persistence of
five pyrethroids, permethrin, cypermethrin, deltamethrin,
fenpropathrin, and fenvalerate, in sand and organic soil at 28 °C,
under laboratory conditions. All of the insecticides (1 mg/kg) were
degraded more rapidly in natural soils than in sterilized soils,
suggesting the importance of microbial degradation. The rate of
degradation under non-sterilized conditions decreased as follows:
fenpropathrin > permethrin > cypermethrin > fenvalerate >
deltamethrin. Amounts of approximately 52% and 74% of the
deltamethrin applied were recovered from the sand and organic soil,
respectively, 8 weeks after treatment.
It was pointed out by Chapman et al. (1981) that biological
processes played a major role in the degradation of deltamethrin in
soils.
The degradation of deltamethrin was also investigated by Zhang
et al. (1984) in an organic soil over a 180-day period. The half-
life of deltamethrin was found to be 72 days, indicating that
deltamethrin is likely to be less susceptible to degradation in
organic soils than in mineral soils. Identification of metabolites
present in the extractable phase confirmed the metabolic pathways
previously reported by Kaufman. Levels of bound 14C residues
increased with the incubation period to reach 19% of the original
14C after 180 days. Most of these bound 14C residues were in the
humic fraction. Bacterial and actinomycete populations increased
in the treated soil, but fungal populations remained relatively
stable during the incubation period.
The degradation of deltamethrin was also studied in two German
soils. Half-lives for sandy soil and sandy loam soil were 35 and
60 days, respectively (Thier & Schmidt, 1977).
All these studies demonstrate that deltamethrin is readily and
quickly degraded in the soil. The half-life of the compound
depends on the nature of the soil as well as the temperature.
Generally speaking, the half-life ranges from 11 to 72 days, under
aerobic conditions. Deltamethrin degradation is slower under
anaerobic or sterile conditions, indicating that microorganisms and
other biological processes play a very important role.
The metabolism of deltamethrin in cotton plants was studied
using material 14C-labelled at the dibromovinyl, benzylic, and
cyano carbons. Under glasshouse conditions, the initial half-life
of deltamethrin was 1.1 weeks and the time needed for 90% loss was
4.6 weeks. Conversion of deltamethrin to the trans-isomer occurred
via photochemical reactions and, after 6 weeks, the trans/cis ratio
was 0.44:1. Deltamethrin degraded more rapidly under field
conditions to give a higher proportion of trans- to cis-isomers and
large amounts of unextractable products. Trace amounts of three
deltamethrin derivatives hydroxylated either at the 4'-position
(10), or at the trans-methyl relative to the carboxy group in the
acid moiety (7), or at both sites (12) were detected with all three
14C preparations (Fig. 1). However, the major metabolites were
free and conjugated Br2CA together with small quantities of the
trans-hydroxymethyl derivatives (20) of Br2CA and 3-(4-
hydroxyphenoxy) benzoic acid (26). The above compounds were
analogues of those formed from permethrin and cypermethrin in
plants. Several types of conjugated metabolites were isolated, but
they were not fully characterized. One type was cleaved readily
with beta-glucosidase or hydrogen chloride to yield Br2CA and
PBacid. Two other types were resistant to beta-glucosidase, but
cleaved readily with hydrogen chloride to yield Br2CA (from the
dibromovinyl label) and 3-phenoxybenzoic acid, 3-phenoxybenzyl
alcohol (from the alcohol label), and alpha-cyano-3-phenoxybenzyl
alcohol (from the cyano and alcohol labels). The metabolites of
deltamethrin identified in plants were analogous to those in
mammals, except for the conjugated products.
The metabolism of deltamethrin and its degradation products in
cotton and bean leaf disks has also been studied. Limited
conversion (approximately 6%) of deltamethrin occurred to give
Br2CA and 3-phenoxybenzyl alcohol (27) (PBalc) conjugates. The
ester cleavage products used as substrates underwent more extensive
metabolism, and two to three types of glucosides were formed from
Br2CA and four from PBalc. 3-Phenoxybenzaldehyde (24),
administered directly or as the cyanohydrin (23), was reduced to
PBalc, though part was oxidized to PBacid (Ruzo & Casida, 1979).
4.4 Bioaccumulation
Bioaccumulation studies with fish, have shown that pyrethroids
have bioconcentration factors (BCFs) that are far lower than those
predicted from the correlation between the Kow partition
coefficient and BCF. The low accumulation can be attributed to
metabolism by the fish and to the reduced bioavailability to fish
of deltamethrin bound by dissolved organic carbon and suspended
colloids. Metabolic kinetics were assessed by Cary (1978) in
Ictalurus punctatus maintained for 30 days in the water of a
hydrosoil system, in which the soil was treated with a dose of
125 g a.i./ha (10 times the normal agricultural dose) and then
flooded after 31 days. During the exposure period, none of the
300 fish died or behaved abnormally despite a final deltamethrin
concentration of 2.19 µg/litre, which is more than 3 times the
acute 96-h LC50 of 0.63 µg/litre (Table 6). During a third phase,
fish were introduced into an uncontaminated liquid medium,
continuously renewed, to monitor elimination of deltamethrin or its
metabolites. The main results are given in Table 5.
Table 5. Bioaccumulation factors after exposure of Ictalurus
punctatus and depuration kineticsa
----------------------------------------------------------------
Organ Value of bioconcentration 14C elimination (%)
factor (BCF)b during after depuration of
exposure, 30 days -------------------
1 day 14 days
----------------------------------------------------------------
muscles 25 <50 77
viscera 972 67 86
carcasses 41 >50 93
body as a whole 144 >50 93
----------------------------------------------------------------
a From: Cary (1978).
b BCF: µg/kg concentration in fish/µg/litre concentration in
water.
Muir et al. (1985) monitored the fate and uptake of
14C-labelled deltamethrin in organisms in experimental ponds over
306 days. Initial concentrations of the pyrethroid ranged from 1.8
to 2.5 µg/litre. The deltamethrin rapidly became distributed in
suspended solids, plants, sediment, and air with a half-life of
2 - 4 h in the water. Aquatic plants (the floating duckweed Lemna
sp. and a submerged/floating weed (Potomageton berchtoldi)
accumulated deltamethrin at concentrations of between 253 and
1021 µg/kg, respectively, 24 h after treatment, but the compound
had all disappeared within 14 days. Fathead minnows, Pimephales
promelas, showed bioconcentration factors of 248 - 907. Although
radioactivity remained in the fish throughout the experimental
period, presumably in the fat, the levels fell steadily and no
effects were seen on the fish.
5. KINETICS AND METABOLISM
5.1 Metabolism in Experimental Animals
Metabolic pathways of deltamethrin in mammals are summarized in
Fig. 3.
The degradation of deltamethrin was also examined under
anaerobic conditions using 14C-cyano-, 14C-phenoxy-, and
14C-vinyl-labelled materials for the tests (Kaufman & Kayser,
1980). Under anaerobic conditions, 14CO2 evolution varied
according to the 14C label position and the time of flooding.
Generally, flooding reduced or initially inhibited the rate of
14CO2 dissipation. However, after one month, 14CO2 dissipation
started again, which suggested the presence of a unique microbial
flora. It was also shown that all three carboxylic acids that
accumulate initially in flooded soils are subsequently further
degraded. Some reduction of PBacid to 3-phenoxybenzyl alcohol
(PBalc) was also observed in these flooded soils.
When deltamethrin was applied to a sandy clay loam soil at
17.5 g/ha in an indoor incubation study and in two field
experiments, the half-lives of deltamethrin were found to be 4.9
and 6.9 weeks under indoor and field conditions, respectively
(Hill, 1983). This difference in the rate of decrease in the
residue was attributed to climatic effects.
This was further confirmed by Hill & Schaalje (1985) who
pointed out a first-order dissipation, if degree-days above 0 °C
rather than days was used as the independent variable, when
deltamethrin was applied by pipette to soils. When deltamethrin
was boom-sprayed, a biphasic first-order plot was observed. A two-
compartment model that predicts an initial fast loss of residue
followed by a slower first-order degradation gave a good fit of the
data.
Chapman & Harris (1981) examined the relative persistence of
five pyrethroids, permethrin, cypermethrin, deltamethrin,
fenpropathrin, and fenvalerate, in sand and organic soil at 28 °C,
under laboratory conditions. All of the insecticides (1 mg/kg) were
degraded more rapidly in natural soils than in sterilized soils,
suggesting the importance of microbial degradation. The rate of
degradation under non-sterilized conditions decreased as follows:
fenpropathrin > permethrin > cypermethrin > fenvalerate >
deltamethrin. Amounts of approximately 52% and 74% of the
deltamethrin applied were recovered from the sand and organic soil,
respectively, 8 weeks after treatment.
It was pointed out by Chapman et al. (1981) that biological
processes played a major role in the degradation of deltamethrin in
soils.
The degradation of deltamethrin was also investigated by Zhang
et al. (1984) in an organic soil over a 180-day period. The half-
life of deltamethrin was found to be 72 days, indicating that
deltamethrin is likely to be less susceptible to degradation in
organic soils than in mineral soils. Identification of metabolites
present in the extractable phase confirmed the metabolic pathways
previously reported by Kaufman. Levels of bound 14C residues
increased with the incubation period to reach 19% of the original
14C after 180 days. Most of these bound 14C residues were in the
humic fraction. Bacterial and actinomycete populations increased
in the treated soil, but fungal populations remained relatively
stable during the incubation period.
The degradation of deltamethrin was also studied in two German
soils. Half-lives for sandy soil and sandy loam soil were 35 and
60 days, respectively (Thier & Schmidt, 1977).
All these studies demonstrate that deltamethrin is readily and
quickly degraded in the soil. The half-life of the compound
depends on the nature of the soil as well as the temperature.
Generally speaking, the half-life ranges from 11 to 72 days, under
aerobic conditions. Deltamethrin degradation is slower under
anaerobic or sterile conditions, indicating that microorganisms and
other biological processes play a very important role.
The metabolism of deltamethrin in cotton plants was studied
using material 14C-labelled at the dibromovinyl, benzylic, and
cyano carbons. Under glasshouse conditions, the initial half-life
of deltamethrin was 1.1 weeks and the time needed for 90% loss was
4.6 weeks. Conversion of deltamethrin to the trans-isomer occurred
via photochemical reactions and, after 6 weeks, the trans/cis ratio
was 0.44:1. Deltamethrin degraded more rapidly under field
conditions to give a higher proportion of trans- to cis-isomers and
large amounts of unextractable products. Trace amounts of three
deltamethrin derivatives hydroxylated either at the 4'-position
(10), or at the trans-methyl relative to the carboxy group in the
acid moiety (7), or at both sites (12) were detected with all three
14C preparations (Fig. 1). However, the major metabolites were
free and conjugated Br2CA together with small quantities of the
trans-hydroxymethyl derivatives (20) of Br2CA and 3-(4-
hydroxyphenoxy) benzoic acid (26). The above compounds were
analogues of those formed from permethrin and cypermethrin in
plants. Several types of conjugated metabolites were isolated, but
they were not fully characterized. One type was cleaved readily
with beta-glucosidase or hydrogen chloride to yield Br2CA and
PBacid. Two other types were resistant to beta-glucosidase, but
cleaved readily with hydrogen chloride to yield Br2CA (from the
dibromovinyl label) and 3-phenoxybenzoic acid, 3-phenoxybenzyl
alcohol (from the alcohol label), and alpha-cyano-3-phenoxybenzyl
alcohol (from the cyano and alcohol labels). The metabolites of
deltamethrin identified in plants were analogous to those in
mammals, except for the conjugated products.
The metabolism of deltamethrin and its degradation products in
cotton and bean leaf disks has also been studied. Limited
conversion (approximately 6%) of deltamethrin occurred to give
Br2CA and 3-phenoxybenzyl alcohol (27) (PBalc) conjugates. The
ester cleavage products used as substrates underwent more extensive
metabolism, and two to three types of glucosides were formed from
Br2CA and four from PBalc. 3-Phenoxybenzaldehyde (24),
administered directly or as the cyanohydrin (23), was reduced to
PBalc, though part was oxidized to PBacid (Ruzo & Casida, 1979).
4.4 Bioaccumulation
Bioaccumulation studies with fish, have shown that pyrethroids
have bioconcentration factors (BCFs) that are far lower than those
predicted from the correlation between the Kow partition
coefficient and BCF. The low accumulation can be attributed to
metabolism by the fish and to the reduced bioavailability to fish
of deltamethrin bound by dissolved organic carbon and suspended
colloids. Metabolic kinetics were assessed by Cary (1978) in
Ictalurus punctatus maintained for 30 days in the water of a
hydrosoil system, in which the soil was treated with a dose of
125 g a.i./ha (10 times the normal agricultural dose) and then
flooded after 31 days. During the exposure period, none of the
300 fish died or behaved abnormally despite a final deltamethrin
concentration of 2.19 µg/litre, which is more than 3 times the
acute 96-h LC50 of 0.63 µg/litre (Table 6). During a third phase,
fish were introduced into an uncontaminated liquid medium,
continuously renewed, to monitor elimination of deltamethrin or its
metabolites. The main results are given in Table 5.
Table 5. Bioaccumulation factors after exposure of Ictalurus
punctatus and depuration kineticsa
----------------------------------------------------------------
Organ Value of bioconcentration 14C elimination (%)
factor (BCF)b during after depuration of
exposure, 30 days -------------------
1 day 14 days
----------------------------------------------------------------
muscles 25 <50 77
viscera 972 67 86
carcasses 41 >50 93
body as a whole 144 >50 93
----------------------------------------------------------------
a From: Cary (1978).
b BCF: µg/kg concentration in fish/µg/litre concentration in
water.
Muir et al. (1985) monitored the fate and uptake of
14C-labelled deltamethrin in organisms in experimental ponds over
306 days. Initial concentrations of the pyrethroid ranged from 1.8
to 2.5 µg/litre. The deltamethrin rapidly became distributed in
suspended solids, plants, sediment, and air with a half-life of
2 - 4 h in the water. Aquatic plants (the floating duckweed Lemna
sp. and a submerged/floating weed (Potomageton berchtoldi)
accumulated deltamethrin at concentrations of between 253 and
1021 µg/kg, respectively, 24 h after treatment, but the compound
had all disappeared within 14 days. Fathead minnows, Pimephales
promelas, showed bioconcentration factors of 248 - 907. Although
radioactivity remained in the fish throughout the experimental
period, presumably in the fat, the levels fell steadily and no
effects were seen on the fish.
5. KINETICS AND METABOLISM
5.1 Metabolism in Experimental Animals
Metabolic pathways of deltamethrin in mammals are summarized in
Fig. 3.
 After oral administration to male rats at 0.64 - 1.60 mg/kg,
the acid and alcohol moieties of deltamethrin were almost
completely eliminated from the body within 2 - 4 days (Ruzo et al.,
1978). On the other hand, the cyano group was eliminated more
slowly, the total recovery during 8 days being 79% of the
radiocarbon dose (43% and 36% in the urine and faeces,
respectively). Tissue residues of deltamethrin labelled with 14C
at the dibromovinyl carbon in the acid moiety and the benzylic
carbon in the alcohol moiety were generally very low, whereas
residue levels in the fat were somewhat higher (0.1 - 0.2 mg/kg).
Residue levels of the radiocarbon derived from the cyano group were
relatively high, especially in the skin and stomach. Essentially,
all the radiocarbon in the stomach was thiocyanate. No noticeable
14CO2 was evolved from any of the radioactive preparations,
including the CN-labelled group, in contrast to the CN group from
fenvalerate, which yielded 14CO2 in considerable amounts.
The major metabolic reactions of deltamethrin were oxidation
(at the trans methyl relative to carbonyl group of the acid moiety
and at the 2'-, 4'-, and 5-positions of the alcohol moiety),
cleavage of the ester linkage, and conversion of the cyano portion
to thiocyanate and 2-iminothiazolidine-4-carboxylic acid (31)
(ITCA) (see Fig. 3). These carboxylic acid and phenol derivatives
were conjugated with sulfuric acid, glycine, and/or glucuronic
acid.
The major faecal metabolites were unchanged deltamethrin (9),
accounting for 13 - 21% of the dose, followed by 4'-OH- (10) and
5-OH-deltamethrin (28), and a trace amount of 2'-OH-deltamethrin
(29). Intact deltamethrin and the 4'-OH-derivative appeared not
only as the administered S-epimer, but also in parts as the
R-epimer, probably due to artefactural racemization on exchange of
the alpha-position hydrogen in methanol solution. The metabolites
from the acid moiety were mostly 3-(2,2-dibromovinyl)-2,2-
dimethylcyclopropanecarboxylic acid (18) (Br2CA) in free form
(10% of the dose), glucuronide (51%) and glycine (trace level)
conjugates, and OH-Br2CA (20) in free form and glucuronide
conjugate (<1%).
The major metabolites of the aromatic portion of the alcohol
moiety were 3-phenoxybenzoic acid (25) (PBacid) in free form (5%),
and glucuronide (13%) and glycine (4%) conjugates and its
4'-hydroxy derivative (26) (4'-OH-PBacid).
Sulfate of 4'-OH-PBacid accounted for about 50% of the dose,
together with small amounts of free (4%) and glucuronide forms
(2%). The CN group was converted mainly to thiocyanate (30) and,
in small amounts, to ITCA (31) (Ruzo et al., 1978). The trans-
isomer of deltamethrin was also rapidly metabolized and yielded
almost the same metabolites as deltamethrin, though 5-OH-derivative
was found in the cis-isomer, but not in the trans-isomer (Ruzo et
al., 1978).
When a single oral dose of 14C-(acid-, alcohol-, or cyano-
labelled) deltamethrin was administered to male mice at 1.7 - 4.4
mg/kg, the acid moiety and the aromatic portion of the alcohol
moiety were rapidly and almost completely excreted, whereas the CN
group was excreted relatively slowly (Ruzo et al., 1979).
Gray & Rickard (1982) followed the distribution of 14C-acid-,
14C-alcohol-, and 14C-cyano-labelled deltamethrin and selected
metabolites in the liver, blood, cerebrum, cerebellum, and spinal
cord after iv administration of a toxic, but non-lethal, dose
(1.75 mg/kg) to rats. Approximately 50% of the dose was cleared
from the blood within 0.7 - 0.8 min, after which the rate of
clearance decreased. 3-Phenoxybenzoic acid (PBacid) was isolated
from the blood in vivo, and was also the major metabolite when
14C-alcohol-labelled deltamethrin was incubated with blood in
vitro. Deltamethrin levels in the liver peaked at 7 - 10 nmol/g at
5 min and then decreased to 1 nmol/g by 30 min. In contrast, peak
central nervous system levels of deltamethrin were achieved within
1 min (0.5 nmol/g), decreasing to 0.2 nmol/g at 15 min, and
remaining stable until 60 min. Peak levels of deltamethrin were
not related to the severity of toxicity, though the levels of
unextractable pentane radiolabel did appear to be correlated with
signs of motor toxicity. Experiments with brain homogenates from
animals injected iv with deltamethrin failed to reproduce the
pentane-unextractable radioactivity in vitro and metabolism of the
compound was not demonstrated.
The major metabolic pathways of deltamethrin in mice were
similar to those in rats, though there were some differences. These
included the presence of more unchanged deltamethrin in mouse faeces
than in rat faeces. In mouse faeces, there were 4 monohydroxy ester
metabolites (2'-OH-, 4'-OH-, 5-OH-, and trans-OH- deltamethrin
(11)) and one dihydroxy metabolite (12) (4'-OH- trans-
OH-deltamethrin) that were not found in mouse urine. Major
metabolites from the acid moiety in mice were Br2CA,
trans-OH-Br2CA (20), and their glucuronide and sulfate
conjugates. Among them, trans-OH-Br2CA-sulfate was detected
only in mice, but not in rats. Compared with rats, much larger
amounts of trans-OH-Br2CA and its conjugates were formed in
mice. A major metabolite of the alcohol moiety in mice was the
taurine conjugate of PBacid in the urine, which was not detected in
rats. Generally, mice produced smaller amounts of phenolic
compounds compared with rats. Also, 3-phenoxybenzaldehyde (24)
(PBald), 3-phenoxybenzyl alcohol (32) (PBalc), and its glucuronide,
and glucuronides of 3-(4- hydroxyphenoxy)benzyl alcohol (33)
(4'-OH-PBalc) and 5-hydroxy-3- phenoxybenzoic acid (34)
(5-OH-PBacid) were found in mice, but not in rats. When mice were
given an ip dose of 14C-deltamethrin, with or without piperonyl
butoxide (PBO) and/or S,S,S-tributyl- phosphorotrithioate (DEF),
the same metabolites were obtained as with oral administration.
However, DEF decreased the hydrolytic products relative to the
controls, while PBO decreased the oxidation products (Ruzo et al.,
1979).
The comparison between the excreted radioactivity of
14C-deltamethrin in rats treated by the percutaneous route and iv
(controls) showed that only 3.6% of the dosage applied on the skin
was absorbed and excreted in 24 h with 1.1% excreted during the
first 6 h. Since the rat skin is more permeable than human skin,
the uptake of deltamethrin through the human skin should be
relatively weak (Pottier et al., 1982).
5.2 Metabolism and Fate in Farm Animals
In a metabolic study, 14C-deltamethrin was administered orally
to lactating dairy cows at the rate of 10 mg/kg body weight per day
for 3 consecutive days. It was poorly absorbed and mainly
eliminated in the faeces as unchanged deltamethrin. Only 4 - 6% of
the administered 14C was eliminated in the urine, and 0.42 - 1.62%
was secreted in the milk. The radiocarbon contents of various
tissues were generally very low with the exception of those of the
liver, kidney, and fat, which were higher (Akhtar et al. 1986).
Deltamethrin degradation occurred by cleavage of the ester bond, as
already reported in rats and mice (Ruzo et al. 1978, 1979). The
enzymes responsible for the ester bond cleavage were located in cow
liver homogenate, mainly in the microsomal fraction, as seen in an
in vitro study (Akhtar, 1984). Metabolites resulting from ester
bond cleavage were further metabolized and/or conjugated, resulting
in a large number of compounds excreted in the urine (see Fig. 3).
In milk, the major identifiable radiolabelled compound was
deltamethrin.
In a feeding study by Akhtar et al. (1987), deltamethrin was
administered twice daily to lactating dairy cows in portions of
their daily feed at the rate of 2 or 10 mg/kg diet for 28
consecutive days. The level of 2 mg/kg diet was the residue level
found in a recently treated pasture (Hill & Johnson, 1987), whereas
10 mg/kg diet was five times this level. Deltamethrin residues in
the milk were dose-dependent and appeared to reach a plateau
between 7 and 9 days after the start of treatment. At the high
deltamethrin intake of 10 mg/kg diet, the deltamethrin residue in
milk was about 0.025 mg/litre. Deltamethrin residues in tissues
were measured 1, 4, and 9 days after the last dose. At the
10 mg/kg diet intake, very small amounts of deltamethrin residues
were found in the liver (<0.005 mg/kg), kidney (<0.002 mg/kg),
and muscle (0.002 - 0.014 mg/kg). Residues in fat were about
0.04 mg/kg and 0.2 mg/kg for the 2 and 10 mg/kg intake,
respectively. Depletion of deltamethrin residues in milk was very
rapid (estimated half-life was about 1 day); while in fat (renal
and subcutaneous) the half-life was 7 - 9 days. Br2CA (3-(2,2-
dibromovinyl)-2,2-dimethylcyclopropanecarboxylic acid) and PBacid
(3-phenoxybenzoic acid) were the only metabolites detected in the
milk and tissues of treated cows. In all cases, they were found at
trace levels of < 0.0235 mg/litre and < 0.034 mg/litre,
respectively. These two metabolites were also previously
identified in rats and mice as the major degradation products of
deltamethrin (Ruzo et al., 1978, 1979).
The fate of 14C-deltamethrin was examined in Leghorn hens
(Akhtar et al., 1985). When laying hens were administered 7.5 mg
of 14C-labelled deltamethrin/hen per day orally for 3 consecutive
days, about 83% and 90% of the administered 14C was eliminated
during the first 24 h and 48 h after dosing, respectively. Tissue
residues were generally very low with the exception of those in the
liver and kidney. Very low levels of residues were found in eggs
obtained within the first 24 h after dosing, but levels increased
reaching a peak within 48 h of the last dose. Residue levels were
higher in the yolk (up to 0.6 mg/kg) than in the albumen (up to
0.2 mg/kg), which is probably related to the lipid content of
yolks. Metabolites were the same as those found in rats and mice.
These studies showed that feeding domestic animals on
deltamethrin-treated feed resulted in very low levels of residues
(if any) in products of animal origin and is unlikely to present a
hazard for the consumer.
5.3 Enzymatic Systems for Biotransformation
Deltamethrin (1 µg) was incubated at 37 °C for 30 min with each
of the following mouse microsome preparations; a) tetraethyl
pyrophosphate (TEPP)-treated microsomes (no esterase and oxidase
activity); b) normal microsomes (esterase activity); c) TEPP-
treated microsomes plus NADPH (oxidase activity); and d) normal
microsomes plus NADPH (esterase plus oxidase activity) (Shono et
al., 1979). Deltamethrin was more rapidly metabolized under the
oxidase system than under the esterase system. The major site of
ring hydroxylation was the 4'-position and the secondary site was
the 5-position. The trans methyl group was an important site of
hydroxylation of the esters and cis methyl oxidation was evident in
the metabolites of the cleaved acid moiety. The preferred sites of
hydroxylation were as follows; trans of dimethyl group,
4'-position in the phenol group, and cis of the dimethyl group,
which was equal to the 5-position in the phenoxy group. Cleavage
of deltamethrin to cyanohydrin may result from both esterase and
oxidase enzyme activities, since larger amounts of the cleaved
products were evident in the oxidase system.
However, at a much higher (approximately 35-fold) concentration
of deltamethrin than that in the above study, it was not detectably
hydrolysed (Miyamoto, 1976; Soderlund & Casida, 1977).
Deltamethrin was hydrolysed by esterases in the blood, brain,
kidney, and stomach of mice yielding PBald and PBacid (Ruzo et al.,
1979).
5.4 Metabolism in Human Beings
Three young male human volunteers underwent a complete medical
check-up one week prior to the morning of the study. Each of them
received a single dose of 3 mg of 14C-deltamethrin mixed in 1 g
glucose and diluted first in 10 ml PEG 300 and again in 150 ml
water. Total radioactivity was 1.8 ± 09 mBq. Samples of blood,
urine, saliva, and faeces were taken at intervals over 5 days.
Clinical and biological examinations were performed every 12 h
during the trial and one week after its termination. Radioactivity
in the biological samples was measured with a liquid scintillation
spectrometer. The clinical and biological checks did not detect
any abnormal findings. There were no signs of side effects or
intolerance reactions, either during or after the trial period.
The maximum plasma radioactivity appeared between 1 and 2 h after
administration of the product, and remained over the detection
limit (0.2 KBq/litre) during the 48 h. The apparent elimination
half-life was between 10.0 and 11.5 h. The radioactivity of blood
cells, as well as the saliva, was extremely low. Urinary excretion
was 51 - 59% of the initial radioactivity; 90% of this
radioactivity was excreted during the 24 h following absorption.
The apparent half-life of urinary excretion was 10.0 - 13.5 h,
which is consistent with the plasma data. Faecal elimination at
the end of the observation period represented 10 - 26% of the dose.
The total faecal plus urine elimination was around 64 - 77% of the
initial dose after 96 h (Papalexiou et al., 1984).
6. EFFECTS ON ORGANISMS IN THE ENVIRONMENT
6.1 Aquatic Organisms
6.1.1 Acute toxicity for fish
Acute toxicity data for deltamethrin in fish have been
summarized by L'Hotellier & Vincent (1986) (Table 6). From this,
it appears that deltamethrin is highly toxic for fish, though the
toxicity varies with the formulation tested.
Table 6. Acute toxicity of deltamethrin tested as the technical or formulated product on
fish; lethal concentrations all expressed as µg active ingredient (a.i.)/litre (96-h)
------------------------------------------------------------------------------------------
Species Systema LC50 (µg/litre) Ref. LC50 (µg/litre) Ref.
(Common name) tested as No. tested as No.
technical formulated
product productb
------------------------------------------------------------------------------------------
Alburnus alburnus S 0.69 4 82 (ULV) 4
(Bleak)
Brachydanio rerio F,S 2.0 10 -
(Zebra fish)
Cyprinodon macularius S - 0.6c (EC) 13
(Desert pupfish)
Cyprinodon S - 0.9 (EC) 19
variegatusd
(Sheepshead minnow)
Cyrpinus carpio F, S 1.84 4 0.65 (EC) 4
(Common carp) 0.86 3 210.0 (ULV)
Gambusia affinis F, S - 1.0c (EC) 13
(Mosquito fish)
Ictalurus nebulosus F, S 1.2 7 2.3 (EC) 15
(Brown bullhead)
Ictalurus punctatus F, S 0.63 8 -
(Hannel catfish)
Idus idus melanotus S - 1.2 (EC) 16
(Golden orfe)
Lebistes reticulatus F, S - 1.8 (EC) 17
(Guppy)
Lepomis gibbosus F, S 0.58 5 0.87 (EC) 14
(Pumpkinseed sunfish)
Lepomis machrochirus F 1.2 6 -
(Bluegill sunfish)
Osteochilus hasseltie S - 1.2 (EC) 20
(Nilem carp)
Puntius gonionotuse F,S - 0.87 (EC) 18
(Jawa carp)
Rhodeus sericeus S 1.12 4 140 (ULV) 4
amarus
Salmo gairdneri F, S 0.39 1 2.2 (EC) 12
(Rainbow trout)
Salmo salar 1.97 2 0.59 (EC) 2
------------------------------------------------------------------------------------------
Table 6. (contd.)
------------------------------------------------------------------------------------------
Species Systema LC50 (µg/litre) Ref. LC50 (µg/litre) Ref.
(Common name) tested as No. tested as No.
technical formulated
product productb
------------------------------------------------------------------------------------------
Salmo trutta F, S - 4.7c (EC) 11
(Brown trout)
Sarotherodon F, S 3.5 9 2.0 (EC) 9
mossambicuse
Tilapia mossambicae F, S - 0.8c (EC) 13
------------------------------------------------------------------------------------------
a F: Flow system, S: Static condition.
b EC: 25 g a.i./litre; ULV: 1 g a.i./litre; values in a.i. equivalent obtained by
calculation.
c LC50 (48-h)
d Marine fish.
e River or pond fish from tropical areas (water temperature > 24 °C).
References
(1) Knauf & Horlein (1979); (2) Zitko et al. (1979); (3) Knauf & Schulze (1977a);
(4) Gulyas & Csanyi (undated); (5) Waltersdorfer & Schulze (1976a); (6) Buccafusco et al.
(1977a); (7) Knauf & Schulze (1977b); (8) Buccafusco et al. (1977b); (9) Adeney et al.
(1980); (10) Lepailleur & Chambon (1984); (11) Lhoste et al. (1979); (12) Waltersdorfer &
Schulze (1976c); (13) Mulla et al. (1978); (14) Waltersdorfer & Schulze (1976d); (15) Knauf
& Schulze (1977b); (16) Waltersdorfer & Schulze (1976b); (17) Waltersdorfer & Schulze
(1976a); (18) Santosa & Hadi (1980); (19) Heitmuller et al. (1978); (20) Santosa (1983)
Zitko et al. (1979) established a 96-h lethal threshold for
Atlantic salmon (Salmo salar) of 1.97 µg/litre.
6.1.2 Acute toxicity for other aquatic organisms
Data on aquatic organisms other than fish are presented in
Table 7 and are of the same order as those for fish, although the
oyster (Crassostrea virginica) is somewhat more tolerant and the
Northern lobster (Homarus americanus) (96-h lethal threshold
0.0014 µg/litre) is far more sensitive (Zitko et al., 1979).
Mohsen & Mulla (1981) exposed aquatic insect larvae to
deltamethrin (as a 2.5% emulsifiable concentrate) for 1 h under
flow-through conditions, and calculated the LC50 after a 24-h
holding period. For the target species blackfly (Simulium
virgatum) an LC50 of 0.9 µg/litre was calculated. Non-target
species tested, mayfly (Baetis parvus) and caddisfly (Hydropsyche
californica), were found to be more susceptible, with LC50 values
of 0.4 µg/litre.
Varanka, (1987) investigated the effects of deltamethrin on
three species of freshwater mussels. Results presented in Table 8
show that the mussels are very insensitive to the pyrethroid.
Table 7. Acute toxicity of deltamethrin tested as technical or
formulated product on other aquatic organisms-lethal concentrations
expressed as µg active ingredient (a.i.)/litre (96-h)a
-------------------------------------------------------------------
Species LC50 (µg/litre) LC50 (µg/litre)
tested as technical tested as formulated
product productb
-------------------------------------------------------------------
Crassostrea virginica - 12.0
(Eastern oyster)
Daphnia magna 5c -
(Water flea)
Gammarus pulex - 0.03c
(Scud)
Penaeus duorarum - 0.35
(Pink shrimp)
Uca pugilatorulosus - 1.1
(Fiddler crab)
Bufo bufo (larvae) - 0.93
(Common toad)
-------------------------------------------------------------------
a Adapted from: L'Hotellier & Vincent (1986).
b EC: 25 g a.i./litre; ULV: 1 g a.i./litre; values in a.i.
equivalent obtained by calculation.
c LC50 (48-h).
Table 8. Acute toxicitya of deltamethrin formulationb in
freshwater mussels, under static conditions at 21 - 23 °Cc
-------------------------------------------------------------------
Species 24-h 48-h 72-h 96-h 7-day
-------------------------------------------------------------------
Anodonta cygnea nd nd ~24.6 12.0 7.6
Anodonta anatina nd nd nd ~23.4 10.3
Unio pictorum nd ~31.8 9.7 7.0 6.0
-------------------------------------------------------------------
a LC50 µg active ingredient (a.i.)/litre): values in a.i.
equivalent obtained by calculation.
b ULV 0.12%.
c From: Varanka (1987).
6.1.3 Field studies and community effects
Two experimental pond studies have been performed. Tooby et
al. (1981) reported that application of deltamethrin to static
water at 10 g a.i./ha did not have any lethal effects on two fish
species (Canassius auratus, Rutilus rutilus) or on molluscs.
Aquatic insects and crustaceans present were killed. Rawn et al.
(1985) applied deltamethrin at a similar rate and also reported
that no fish were killed. The half-life of deltamethrin in the
pond was 2 - 4 h for water and 2 - 14 days for bottom sediment.
Neto et al. (1983) sprayed-flooded fields in Brazil, at
intervals of 2 days, with rates of deltamethrin progressively
increased at 5, 10, 12, and 13 g a.i./ha. The expected
concentrations in water from these applications were between 3 and
7 µg/litre. No mortality was recorded in fish placed in the
sprayed area in experimental cages. Slight "agitation" was
reported after exposure to the highest dose.
Impact assessments on the use of deltamethrin on paddy fields
have been made in the field in various countries throughout the
world. The maximum normal usage rate of the compound was 6.5 g
a.i./ha. In these studies, fish ( Tilapia spp., Cyprinus carpio,
Gambusia spp.) tolerated deltamethrin up to 18.75 g a.i./ha without
any adverse effects. The compound is known to be toxic for aquatic
organisms and is not recommended for use over water under any but
exceptional circumstances. However, it has been used to control
vectors of major human diseases, i.e., mosquitos and blackfly
( Elossina spp.), where benefit outweighed potential risk. In these
cases, extensive field evaluations of the environmental impact have
been made. While there have not been any instances of fish kills
from these applications, there are reports of large numbers of
deaths of aquatic invertebrates. The populations usually recovered
rapidly and all studies have shown numbers back to normal before
the compound was applied again in the following season. It is
suggested that relatively resistant parts of the population soon
recolonize the area; immigration also occurs (Takken et al., 1978;
Smies et al., 1980; Baldry et al., 1981; Everts et al., 1983).
6.1.4 Appraisal
Notwithstanding its high toxicity for fish and crustacea, the
results of many studies, as well as the wide use of deltamethrin
for several years, have confirmed that its normal use does not
cause significant mortality in fish populations. This difference
is due to its strong adsorption on soil and its rapid breakdown,
decreasing its bioavailability under field conditions.
6.2 Terrestrial Organisms
6.2.1 Plants
Hargreaves & Cooper (1979) sprayed glasshouse-grown tomato
seedlings with 50 mg deltamethrin/litre (2.5% emulsifiable
concentrate) 3 weeks after emergence and again 7 days later. Three
days after the second application, plants were examined for damage.
No damage was found and, at this rate of use, deltamethrin was not
phytotoxic.
6.2.2 Soil microorganisms
In a study by Tu (1980) on the effects of 5 pyrethroids on
microbial populations and their activity in soil, 0.5 mg
deltamethrin/kg incorporated into sandy loams (residues under
normal use conditions would be of the order of < 0.001 mg/kg)
produced only a few transient effects. No effects were noted on
the nitrifying microorganisms and their capacity to produce nitrate
and there were no inhibitory effects on deshydrogenase or urease
activity. Deltamethrin induced an increase in oxygen consumption
because of an increase in microbial respiration (probably linked
with the microbial degradation of deltamethrin). It also
stimulated the growth of soil fungi and inhibited the development
of bacteria. Four weeks after treatment, deltamethrin-treated soil
recovered completely and microorganism activity was equal to that
in untreated soil.
6.2.3 Soil fauna
6.2.3.1 Earthworms
When deltamethrin at 12.5 g a.i./ha (high agricultural dose)
was incorporated into the soil to a depth of 1 cm, there were no
toxic effects on earthworms (Lumbricus terrestris) during an
observation period of 28 days (Bouche & Fayolle, 1979). However,
significant toxic effects on earthworms were observed at levels of
60 - 125 g a.i./ha (5 - 10 times the highest rates applied in
agriculture).
In another study with Eisenia foetida andrei, deltamethrin
incorporated in artificial soil at concentrations of 1.7 mg/kg and
10 mg/kg did not produce any lethal effects (Chambon & Lepailleur,
1984).
6.2.3.2 Slugs
Lettuce leaves treated with 4 times normal dosage rates, were
fed to slugs ( Agrolimax sp.). Leaves were quickly consumed but no
toxic effects (mortality or activity) were observed (Ricou, 1978).
6.2.3.3 Soil arthropods
Under laboratory conditions, deltamethrin, applied topically
and by immersion, was very toxic for the carabid beetle
Pterostichus melanarius (Illiger). Under natural conditions in the
field, deltamethrin applied at normal dose rates was not toxic for
these organisms (Dunning et al., 1981).
Everts et al. (1985) monitored the effects, on non-target
organisms, of various compounds when used for the control of tsetse
fly in the Ivory Coast in Africa. Deltamethrin was the most
effective compound against the tsetse and also killed non-target
musca flies. After deltamethrin spraying, Orthoptera and
Proctotrupoidea were also significantly decreased while Nematocera
increased in number. The results of this study suggest that ground
spraying of the pyrethroid had greater effects on terrestrial
arthropods than aerial applications.
Concurrent laboratory and field studies were conducted on the
effects of deltamethrin on beneficial predatory spiders in a polder
area of the Netherlands (Everts et al., 1988). During two growing
seasons, 2800 samples were taken over an area of 17 different
fields. The authors found that effects on spiders were eliminated
when it rained soon after application, since the effect of the
pyrethroid appeared to be indirect, causing the dehydration of
spiders. This different response under dry and damp conditions was
confirmed in the laboratory. However, reduction of spider numbers
in the field was much greater than predicted from laboratory tests
and recovery was more rapid in laboratory populations than in field
populations. The uptake and effects of deltamethrin were greater
through exposure to residues than through contact or oral exposure.
There was a positive correlation between temperature and the
toxicity of deltamethrin for spiders in the field. This contrasted
with reports of a negative correlation for target insects reported
in the literature. Laboratory studies showed that the negative
temperature effect only occurred when spiders could not drink. It
appeared that qualitative prediction from laboratory to field was
possible but that quantitative prediction was not.
6.2.4 Beneficial insects
6.2.4.1 Honey-bees
Single applications of deltamethrin are highly toxic for honey-
bees (Apis mellifera). Stevenson et al (1978) found a contact LD50
of 0.051 µg/bee and an oral LD50 of 0.079 µg/bee.
Arzone & Vidano (1978) did not find any difference in mortality
between controls and bees fed on sugar solutions containing 0.2 µg
deltamethrin/litre. Increased mortality was recorded at all higher
exposures reaching 100% within 1 h at a concentration of 12.5
µg/litre.
In the field, direct treatment of caged bees caused a high
mortality rate with doses of from 11.2 g/ha upwards (Atkins et al.,
1976). Rape flowers were treated at a rate of 0.75 g a.i./100
litre and 1.5 g a.i./100 litre with an emulsifiable concentrate
formulation, 25 g/litre; control plots were treated with water.
Cages (3 x 2 x 2 m) containing a small hive (2 frames + open brood)
were put over the treated flowers once the spray had dried. The
mortality of the bees was then assessed over 7 days. The average
mortalities were not significantly higher in the treated plots than
in water-sprayed control plots (Louveaux et al., 1977).
However, Bocquet et al. (1980, 1983) demonstrated, after 3
years of field experiments, that deltamethrin under field
conditions was innocuous at doses up to 12.5 g/ha. They also noted
a repellant effect by the formulating materials, which lasted for
2 - 3 h. Further studies have been reported by Florelli et al.
(1987a,b).
6.2.4.2 Foliar insects
Deltamethrin was 70 times more toxic to the tobacco budworm
(Heliothis virescens) than to its predator, green lacewing
(Chrysopa carnea), but it was only 1.25 times more toxic to the
tobacco budworm than to its parasite (Campoletis sonorensis) (Plapp
& Bull, 1978).
In an apple orchard, where deltamethrin was applied at
12.5 mg/kg, no predatory mites (Typhlodromus pyri) were found
during 10 weeks of observation, but spider mites (Paponychus ulmi)
were not affected. The elimination of the predatory mite led to a
marked increase in spider mite populations, later in the same
season (Aliniazee & Cranham, 1980).
The impact of deltamethrin used against the English grain aphid
(Sitobion avenae) was studied in 1983, 1984, and 1985 in the Paris
basin. This study was carried out on wheat with pitfall traps,
yellow water traps, suction sampling (D-vac), and sampling of ears.
Effects were noted on: S. avenae, phytophagous Diptera (Opimyza
florum, Phytomyza nigra, and Oscinella frit), Homoptera (Zyginidia
scutellaris, Metopolophium dirhodum), Thysanoptera (Limothrips
cerealium, Acolothrips intermedius), predatory Diptera (Empididae,
Dolichopodidae), and on spiders (Erigonidae, Lycosidae,
Linyphiidae, Theridiidae). The detritiphagous insects (Sciaridae,
Chironomidae), the Carabidae and Staphylinidae and most
microhymenoptera showed little or no difference after treatment.
During the 3 years, no differences were observed from year to year
as a result of field treatment, populations appearing homogeneous
at the beginning of each trial (Fischer & Chambon, 1987).
A large-scale field trial was carried out in 1984 in southern
England to investigate the side-effects of deltamethrin on non-
target arthropods in winter wheat. The insecticides were applied
in June and two methods, suction sampling (D-vac) and quadrats,
were used to sample the arthropods for up to 75 days after
treatment. During the post-treatment period, the numbers of
Carabidae and Staphylinidae adults found in D-vac samples were
reduced by 22% and 20%, respectively, compared with the controls
(Vickerman et al., 1987a).
In the same field trial, arthropods were sampled with a D-vac
for 11 weeks. Total numbers were similar in the control and
deltamethrin-treated plots. The numbers of Empididae were reduced
by deltamethrin, but Dolichopodidae were more numerous in treated
than in control plots. The numbers of Aphidius spp. were higher in
the deltamethrin-treated plots than in the control plots. The
numbers of Coccinellidae larvae were reduced (Vickerman et al.,
1987b).
6.2.5 Birds
6.2.5.1 Laboratory studies
Data on the acute toxicity of deltamethrin for birds are given
in Table 9.
Table 9. Acute toxicity of deltamethrin for birds
---------------------------------------------------------------------------
Species Sex Application LD50 (mg/kg) Reference
---------------------------------------------------------------------------
Red partridge male & oral >3000 Grolleau & Griban,
(Alectonis tufa) female 1976b
Grey partridge male & oral >1800 Grolleau & Griban,
(Perdix perdix) female 1976b
Chicken oral >1000 Grandadam, 1976
(Gallus domestica)
Hen adult oral >2500 Ross et al., 1978
female
Mallard duck oral >4640 Beavers & Fink,
(Anas platyrhynchos) 1977a
Game duck oral >4000 Grolleau & Griban,
1976a
---------------------------------------------------------------------------
The toxicity of deltamethrin for birds is very low. Both
technical grade and commercially formulated deltamethrin
administered in feed at 100 mg/kg diet was not palatable to
Japanese quail (Coturnix coturnix japonica), with strong individual
variations. Unpalatability diminished after repeated exposure and
even became reversed in the case of the purified deltamethrin,
which attracted quail already suffering from toxic effects (David,
1981).
Groups of 39 female Japanese quail (Coturnix coturnix japonica)
were given daily doses of 0, 0.2, or 1 mg technical deltamethrin
per animal, by gavage, over 34 days. No significant effects were
observed on reproduction (De Lavaur et al., 1985).
6.2.5.2 Field studies on birds
The low toxicity of deltamethrin for birds, indicated by
laboratory studies, has been confirmed in the field. In studies on
the ecological consequences of the use of the compound to control
tsetse fly (Takken et al., 1978) and blackfly (Smies et al., 1980)
in West Africa, populations of various species of insectivorous,
granivorous, and piscivorous birds were examined before and after
spraying. There were no indications of any effects on either
numbers or species diversity.
7. EFFECTS ON EXPERIMENTAL ANIMALS AND IN VITRO TEST SYSTEMS
7.1 Single Exposures
Tables 10 and 11 show the results of acute toxicity studies on
various animal species. From these tables, it is clear that the
vehicle has a great influence on the LD50, probably by influencing
absorption. Powder formulations and aqueous suspensions are
significantly less toxic than formulations in oils or organic
solvents (Pham Huu Chanh et al., 1984).
The acute oral toxicity of deltamethrin for rats produced such
symptoms as: staining of the fur, excessive grooming, salivation,
diarrhoea, drowsiness, weakness, dyspnoea, piloerection, ptosis,
difficulty in walking, general motor incoordination, hypotonia,
choreoathetosis, clonic seizures, and death (Glomot, 1979; Glomot
et al., 1979, 1981a; Kavlock et al., 1979; Ray & Cremer, 1979; Pham
Huu Chanh et al., 1984). Electroencephalogram (EEG) records showed
generalized spike discharges prior to choreoathetosis (Ray &
Cremer, 1979; Ray, 1980).
Mice presented far fewer symptoms than rats after oral dosing
at comparable levels, diarrhoea being the only reportable
observation (Glomot et al., 1980a).
Rats were injected intraperitoneally with 14C-labelled
deltamethrin at the threshold doses required to produce the motor
symptoms of toxicity of tremor and choreoathetosis. Blood and brain
samples were analysed for their total radiolabel content, and were
also extracted with ethyl acetate to determine the levels of
extractable parent deltamethrin and 3-phenoxybenzyl-derived acid and
the residual radiolabel after this extraction. There was a clear
correlation between onset of symptoms and blood and brain levels of
deltamethrin. It was found that certain threshold levels of parent
deltamethrin in the blood and brain were required for symptoms
development, and that the symptoms persisted for as long as this
threshold was maintained (Rickard & Brodie, 1985).
7.1.1 Mouse
Mice intravenously injected with deltamethrin showed intense
tremors, convulsions, and ataxia, immediately after administration.
Tachycardia and respiratory defects were also observed at higher
dosages. Surviving animals appeared normal after 4 - 5 h.
Immediately after intraperitoneal injection, jumping movements,
slight convulsions and prostration, ptosis, tail hypertonicity, and
cyanosis were observed. These toxic signs disappeared after 72 h
in surviving animals.
Animals administered deltamethrin by gavage showed muscular
stiffening and convulsions, 1 h after dosing. After 24 h,
hypermotility, stereotype movements of the head, tachycardia,
hypertonicity of the tail, and a few convulsions were observed.
Behaviour and appearance were normal again after 48 h (Glomot &
Chevalier, 1976a,c).
Table 10. Acute toxicity of technical grade deltamethrin
--------------------------------------------------------------------------------------------------------------
Species Sex Route Vehicle LD50 (mg/kg Reference
body weight)
--------------------------------------------------------------------------------------------------------------
Rat male oral sesame oil 128 Glomot & Chevalier (1976a)
female 139
male PEG 200 67
female 86
Rat male adult peanut oil 52 Kavlock et al. (1979)
female adult 31
female weanling 50
Rat male adult peanut oil 53 Gaines & Linder (1986)
female adult 30
female weanling 48
Rat male + female aqueous suspension > 5000 Audegond et al. (1981)
with carboxy- (no mortality)
methylcellulose
Rat dermal - 700 Panshina & Sasinovich (1983)
Rat male methylcellulose (1%) > 2940 Kynoch et al. (1979)
female
Rat female adult xylene > 800 Kavlock et al. (1979)
Rat male + female inhalation (6 h) dust 600 mg/m3 Coombs & Clark (1978)
Rat male adult (2 h) DMSO 10% aerosol 940 mg/m3 Kavlock et al. (1979)
female adult > 785 mg/m3
Rat male + female (1 h) micronized powder > 4620 mg/m3 Jackson & Hardy (1986)
Rat intraperitoneal - 58.8 Panshina & Sasinovich (1983)
Rat male intraperitoneal sesame oil 209 Glomot & Chevalier (1976b)
female 186
male PEG 200 24 Glomot & Chevalier (1976b)
female 25
--------------------------------------------------------------------------------------------------------------
Table 10. (contd.)
--------------------------------------------------------------------------------------------------------------
Species Sex Route Vehicle LD50 (mg/kg Reference
body weight)
--------------------------------------------------------------------------------------------------------------
Rat male intravenous PEG 200 3.3 Glomot & Chevalier, (1976c)
female 3.3
Rat female adult acetone 4 Kavlock et al. (1979)
female weanling 1.8
Mouse male oral sesame oil 33 Glomot & Chevalier (1976a)
female 34
male PEG 200 21 Glomot & Chevalier (1976a)
female 19
Mouse intraperitoneal - 33 Panshina & Sasinovich (1983)
Mouse male intraperitoneal sesame oil 171 Glomot & Chevalier (1976b)
female 166
Mouse male PEG 200 18 Glomot & Chevalier (1976b)
female 12
Mouse male PEG 200 4.1 Glomot & Chevalier (1976c)
female 4.0
Mouse male glycerol formal 5 Glomot & Chevalier (1976c)
female 5.8
Dog male + female oral in capsules >300 Glomot et al. (1977)
no mortality
Dog male + female PEG 200 2 Glomot & Chevalier (1976c)
Rabbit male dermal PEG 400 > 2000 Clair (1977)
female > 2000
--------------------------------------------------------------------------------------------------------------
Table 11. Acute toxicity of some formulations
--------------------------------------------------------------------------------------------------------------
Species Sex Route Formulation LD50 (mg/kg Reference
body weight)
--------------------------------------------------------------------------------------------------------------
Rat male, female oral 2.5% flowable formulation 22 000 Glomot et al. (1979)
Rat male, female oral 2.5% wettable powder >15 000 Glomot (1979)
Mouse male, female oral 2.5% wettable powder >15 000 Glomot et al. (1980a)
Dog male, female oral 2.5% wettable powder >10 000 Glomot et al. (1980b)
Rat male, female oral 2.5% emulsifiable concentrate 535 Coquet (1977)
Rat male, female oral 10 g/litre ULV >6 470 Coquet (1977)
Rat male, female inhalation (4 h) aerosol-2.5% wettable powder >2 800 mg/m3 Clark et al. (1980)
--------------------------------------------------------------------------------------------------------------
7.1.2 Rats
Rats intravenously injected with deltamethrin showed muscular
contractions, piloerection, respiratory defects, convulsions, and
paresis of the hind quarters, immediately following treatment.
Surviving animals showed normal behaviour after 48 h. Immediately
after intraperitoneal injection, tremor, convulsions, prostration,
and cyanosis were observed. These toxic signs disappeared after
48 h in surviving animals. Animals administered deltamethrin by
gavage showed motor incoordination, convulsions, respiratory
defects, and hypomotility, shortly after dosing. Normal behaviour
was observed after 3 days (Glomot & Chevalier, 1976c).
In an inhalation study (whole body exposure for 6 h),
hyperactivity, grooming, and irritation were observed during
exposure. The animals were hypersensitive to touch and noise and
showed uncoordinated movements. Gross pathological investigation
showed a gas-filled stomach and small intestine, and massive
haemorrhage and degeneration in the lung (Coombs & Clark, 1978).
Rats were exposed (whole body exposure) for 4 h to an aerosol
concentration of deltamethrin equal to 2.8 g/m3, the highest
attainable airborne concentration of a 2.5% wettable powder
formulation. Approximately 80% of the total aerosol had a mean
aerodynamic diameter of less than 5.5 µm. Dyspnoea and gasping
were observed in exposed rats. Relative lung weights and
macroscopic pathology were normal. There was no mortality (Clark
et al., 1980).
7.1.3 Rabbit
Rabbits (10 males and 10 females) were treated with 2 g
deltamethrin in 2 ml PEG 400 per kg body weight on 80 cm2 of
occluded shaved skin for 24 h. The animals were observed for 14
days. Two animals showed obvious erythema. No body weight changes
or abnormal behaviour were observed. On histological observation
of the liver, kidneys, and skin, small changes were observed, but
these were common for this strain of rabbit and not related to
treatment (Clair, 1977).
7.1.4 Dog
Dogs given oral doses of 100 mg deltamethrin/kg body weight or
more showed transient hyperexcitability, akinesia, vomiting, and
stiffness of the hind legs (Glomot et al., 1977).
Dogs orally dosed with 10.0 mg deltamethrin/kg body weight did
not display any clinical signs related to treatment (Glomot et al.,
1980b).
7.2 Irritation and Sensitization
7.2.1 Skin irritation
Male albino rabbits (12 per group) weighing 2.5 - 3.5 kg were
administered 0.5 g deltamethrin on either shaved intact or abraded
skin. The occlusive patch was fixed on the skin for 23 h.
Technical deltamethrin (98% purity) did not produce any irritant
effects (Coquet, 1976a).
Male albino rabbits (6) weighing 2.5 - 2.9 kg were administered
0.5 ml of formulated deltamethrin (25 g/litre flowable suspension
concentrate) to both shaved intact and abraded skin. The Primary
Irritation Index after 24 h exposure of occluded sites was 1.2,
i.e., slightly irritating (Glomot et al., 1981b).
An evaluation similar to the one described above was carried out
for a 2.5% wettable powder concentrate deltamethrin formulation.
Rabbits had a Primary Irritation Index of 2.41, i.e., moderately
irritating. Moderate erythema continued for 72 h, while the oedema
generally diminished, with the exception of scarified skin sites
(Glomot et al., 1981c).
The skin irritation potentials of Decis emulsifiable
concentrate 2.5% and Decis Flowable 2.5% were studied on rabbits
and guinea-pigs with 0.05, 0.10, 0.5, 1, and 2.5% deltamethrin.
The threshold irritative levels were 0.05% for Decis emulsifiable
concentrate and 2.5% for Decis Flowable. The intensity of
irritation depended on the relative content of organic solvents and
emulsifiers in the trade products. The water-soluble concentrate
of Decis 2.5% caused negligible risk of contact irritative
dermatitis (Bainova & Kaloyanova, 1985).
7.2.2 Eye irritation
Deltamethrin (0.1 g/animal) was administered into the
conjunctival sac of the eyes of 6 male albino rabbits, weighing
2.5 kg, with or without rinsing 60 seconds after instillation.
Deltamethrin produced transient irritating effects, both with and
without rinsing (Coquet, 1976b).
Male albino rats (9) weighing between 2 and 3 kg were
administered 0.1 ml of formulated deltamethrin (25 g/litre flowable
suspension concentrate) in the conjunctival sac. Six of the
treated eyes remained unwashed, while the remaining three were
rinsed with lukewarm water 20 - 30 seconds after instillation.
There was only transient clouding of the cornea in 2 animals 1 h
after dosing (1 washed, 1 unwashed), which cleared by day 2. Low
grade conjunctival irritation was noted among all animals
initially, which disappeared following day 2 of observations
(Glomot et al., 1981d).
A 2.5% deltamethrin formulation diluted 1/10 in distilled water
(0.1 ml per rabbit) elicited a similar pattern of initial transient
corneal clouding in 3 out of 9 rabbits examined, which cleared by
day 4. The undiluted formulation (100 mg) administered in the
conjunctival sac of rabbits produced increased involvement of the
conjunctiva, iris, and cornea in all animals, generally moderate in
severity, with low grade corneal opacity persisting in 2 rabbits
until day 7 (1 washed, 1 unwashed) (Glomot et al., 1981e).
7.2.3 Sensitization
Deltamethrin (0.5 g/animal) was applied topically to the skin
of albino guinea-pigs (10 male and female) 3 times per week, with a
2-day interval for 3 weeks, and once at the start of the fourth
week. The preparation was covered with an occlusive patch for
48 h. On days 1 and 10, the guinea-pigs received an intradermal
injection of 0.1 ml of Freund's adjuvant. The animals were
challenged 12 days after the last application with 0.5 g
deltamethrin. No sensitization was found (Guillot & Guilaine,
1977).
7.3 Short-Term Exposure
7.3.1 Rat
Male and female weanling Sprague-Dawley rats (20 of each sex
per group) were dosed (by gavage) with 0, 0.1, 1, 2.5, or 10 mg
deltamethrin in PEG 200/kg body weight per day for 13 weeks. No
treatment-related effects were observed on food and water
consumption, mortality, urinalysis, and haematology. Neurological
examinations and ophthalmoscopy did not reveal any abnormalities.
At the highest dose level, a slight hyperexcitibility was observed
among some rats in week 6. Lower body weight gain was noted in
males at 2.5 and 10 mg/kg. No clear treatment-related effects were
noted in the results of laboratory investigations or on the weights
of the organs. Gross and microscopic examination of a variety of
tissues and organs did not show any treatment-related findings.
Following the 13-week dosage period, 5 males and 5 females per
group were allowed to recover for 4 weeks. No evidence of
hyperexcitability was observed among the rats; body weight gain was
slightly higher in the treated groups than in the controls. The
no-observed-effect level was 1 mg/kg body weight (Hunter et al.,
1977).
Four groups of CD rats (8 of each sex per group) were exposed to
aerosolized deltamethrin (technical grade powder) for 6 h per day, 5
days a week, for 2 weeks, and for 4 days during a third week. Mean
aerosol concentrations were 3, 9.6, and 56.3 mg a.i./m3 with
about 87% of respirable particles (diameter lower than 5.5 µm). No
rats died as a result of exposure. Signs of irritation (agitated
grooming and ptyalism due to the powder were noted in all groups
during exposure, with more pronounced toxic signs (ataxia and
walking with arched backs) in the group receiving the highest dose
tested. Male rats also showed a reduced body weight (-5%) in all
groups. An elevation of the serum sodium ion content was noted at
the two highest doses. No increased incidence of any particular
lesion was observed in the high-dose group compared with the control
group. Irritation and weight loss were only slight at 3 mg/m3 and
this can be considered as a no-effect level (Coombs et al., 1978).
7.3.2 Dog
Male and female beagle dogs (3 - 5/sex per group), 25 weeks of
age, received a daily oral dose of 0, 0.1, 1, 2.5, or 10 mg
deltamethrin/kg body weight in PEG 200 in gelatin capsules over 13
weeks. All treated groups showed reduced body weight gain, but
this was not dose-related. Liquid faeces were associated with all
groups of treated dogs throughout the dosing period. Dilatation of
the pupils was seen in dogs receiving 2.5 and 10 mg/kg per day.
The sign was first seen 4 - 7 h after dosing and persisted
throughout the day. The incidence of vomiting increased dose-
dependently in all treated groups, except the group receiving
0.1 mg/kg. In the highest dose group, unsteadiness, body tremors,
and jerking movements were seen, particularly in males, in weeks 2,
3, and 4. Excessive salivation was seen initially and diminished
during the dosing period. After 5 and 12 weeks, depression of the
gag reflex was noted in a proportion of animals in all treated
groups. However, this was not considered to be of toxicological
significance. Exaggeration or depression of the patellar reflex
was observed in some animals in all treated groups after 5 and 12
weeks, mainly at 1, 2.5, or 10 mg/kg per day. Some animals in all
treated groups showed depression of the flexor reflex. Dose levels
of 2.5 and 10 mg deltamethrin/kg per day caused modification of the
EEG pattern in some animals, 12 weeks following administration.
Histopathological evaluations of tissues and organs, including the
nervous system and muscle tissue, did not reveal any abnormalities
that could be related to the administration of the compound.
During recovery, the gag reflex continued to be depressed, whereas
exaggeration of the patellar reflex was still seen in some dogs
that had previously received 1 mg/kg per day (Chesterman et al.,
1977).
7.4 Long-Term Exposure and Carcinogenicity
7.4.1 Mouse and rat
Male and female Charles River CD-1 mice (80 of each sex per
group) were fed dietary levels of deltamethrin of 0, 1, 5, 25, or
100 mg/kg, daily, for 24 months. There were no clear effects
related to the administration of deltamethrin on general behaviour,
mortality, body weight, and food consumption. Blood chemistry,
haematology, and urine analysis parameters were normal after 12, 18,
and 24 months (at the times of interim and terminal sacrifice).
Microscopic examination of tissues did not reveal any lesions
indicative of a compound-related effect. The tumour incidence was
unaffected by deltamethrin administration. The no-observed-effect
level was 100 mg/kg diet (Goldenthal et al., 1980a).
Deltamethrin was administered, by gavage, to C57BL/6 mice at 4
dose levels (0, 1, 4, and 8 mg/kg body weight) and to BDVI rats at
3 dose levels (0, 3, and 6 mg/kg body weight) on 5 days a week for
104 weeks. After completion of the treatment, the animals were
observed until 120 weeks of age, when all survivors were killed.
The treatment had a slight effect on body growth and survival
rates, especially in the groups of mice and rats treated with the
highest dose. In C57BL/6 mice, various types of tumours were
observed in all treated groups. An increased incidence of
lymphomas was observed in mice receiving deltamethrin at levels of
1 and 4 mg/kg body weight, but not in the group treated with
8 mg/kg body weight. No significant difference in the incidences
of lung adenomas, liver-cell tumours, or other tumours was observed
in treated groups compared with controls. In BDVI rats, an
increased incidence of pituitary, thyroid, and mammary tumours was
noted; however, no clear dose-response relationship was shown
(Cabral et al., 1986). The details of this study were not
available for evaluation.
Male and female Charles River CD rats (90 of each sex per
group) were fed with 0, 2, 20, or 50 mg deltamethrin/kg diet for
2 years. A second control group (60 of each sex) was also used.
Interim sacrifices (10 of each sex per group excluding control
group 2) were made after 6, 12, and 18 months. No changes in
general behaviour and appearance were observed in relation to
treatment. Survival rate was similar for control and treated rats
(50 - 67%). Rats in the 50 mg/kg group gained slightly less weight
than control rats, whereas the food consumption was essentially the
same. Ophthalmoscopic findings were generally similar for control
and treated rats. No haematological and biochemical parameters
were changed in a biologically significant way in relation to
treatment at any time, except for a decrease in SGPT (serum
glutamic pyruvic transaminase) activity at 6 months in the mid- and
high-dose groups. Organ weights were not affected. The
macroscopic and microscopic findings were common for the species
and the strain, except for a slightly increased incidence of axonal
degeneration in sciatic, tibial, and/or plantar nerves in the 20
and 50 mg/kg groups at 18 months, but not at termination. Thus,
this was not considered to be indicative of a compound-related
effect. The incidence of benign testicular tumours (interstitial
cell adenomas) at terminal sacrifice in this study was: control
group 1, 0/37; control group 2, 4/35; low-dose group 1/38; mid-dose
group 1/30; high-dose group 6/38. The incidence seen in the high-
dose group was considered to be spontaneous, because it was not
significantly higher than in the second control group or in
historical control groups (Goldenthal et al., 1980b; Richter &
Goldenthal, 1983).
7.4.2 Dog
Deltamethrin dissolved in maize oil was administered in the diet
to 64 beagle dogs (8 of each sex per group) at levels of 0, 1, 10,
and 40 mg/kg for 24 months. This corresponds to 0, 0.025, 0.25, and
1 mg/kg body weight, respectively. Individual body weights and food
consumption values were determined weekly. Ophthalmoscopic,
haematological, biochemical, and urinalysis examinations were
conducted during the pre-test period and at 6, 12, 18, and 24 months
of the study. Neurological examinations were conducted at
approximately 1 year and before termination. No signs of overt
toxicity were observed in any of the dogs. Body weight and food
consumption values were similar for control and treated dogs. No
compound-related effects were observed during the ophthalmoscopic
and physical examinations. Although there were some random
statistically significant differences between the control and
other dose groups in the haematological and biochemical tests,
physiologically significant changes were not observed at any
interval in the study. Two treated and two control animals died
during the study. No compound-related gross or microscopic changes
were observed in the surviving dogs that were sacrificed and
necropsied. Inflammatory, degenerative, and proliferative changes
described were spontaneous in nature, or related to the estrous
phase of the menstrual cycle, and unrelated to compound
administration. On the basis of this study, it has been concluded
that the no-observed-effect level is 40 mg/kg diet (equivalent to
1 mg/kg body weight per day) (IRDC, 1980).
7.5 Mutagenicity
7.5.1 Microorganisms
DNA repair tests in Escherichia coli were conducted at dose
levels of 1250, 2500, or 5000 µg deltamethrin/ml. Deltamethrin was
dissolved in dimethyl sulfoxide (DMSO) and 0.1 ml of the solution
was spread on a plate. Growth inhibition was compared between DNA
repair deficient mutants (p3478 and CM611) and wild types (W3110
and WP2). Partial precipitation of deltamethrin from the solution
occurred when it came into contact with the aqueous bacterial
growth medium. Deltamethrin did not have any damaging effects on
DNA (Peyre et al., 1980).
Deltamethrin was examined for its mutagenic potential in the
Ames test with 5 strains of Salmonella typhimurium (TA 1535, TA
1537, TA 1538, TA 98, and TA100) at doses of 2, 10, 50, 200, 500,
1000, or 5000 µg/plate, with and without S-9 mix (metabolic enzyme
system). It was dissolved in DMSO and precipitated out of solution
at concentrations of 200 µg/plate or more. Deltamethrin did not
have any effect on the mutation rate in any of the strains at any
of the concentrations tested (Peyre et al., 1980).
A similar Ames test was carried out at 0.2, 2, 20, 200, or
400 µg deltamethrin/plate with microsome enzymes. The compounds
did not influence the number of revertants of the 5 strains (same
as above) of S. typhimurium. Again, deltamethrin was dissolved in
DMSO and precipitated out of solution at 200 µg/plate or more
(Fouillet, 1976).
Kavlock et al. (1979) found deltamethrin not to be mutagenic in
2 assays with S. typhimurium at doses of 0 - 1000 µg/plate in DMSO,
with or without metabolic activation. They also obtained negative
results with E. coli at 10 - 1000 µg/plate as well as with
Saccharomyces cerevisiae at concentrations of 1 - 5%, in both cases
with and without metabolic activation. Deltamethrin was found not
to be mutagenic in S. typhimurium strains TA100 and TA98, in the
presence or absence of a rat liver activation system, using the
plate incorporation assay and fluctuation tests. The compound,
dissolved in DMSO, precipitated out of solution at 600 µg/plate
(Pluijmen et al., 1984).
7.5.2 Cultured cells
Deltamethrin, dissolved in a mixture of cremaphor oil and
ethanol (1:1), was applied to a culture of Chinese hamster ovary
cells (CHO) at levels of 0.04, 0.2, 1.0, or 5.0 mmol/litre, with or
without metabolic activation, and examined for chromosomal
aberrations and sister chromatid exchanges (SCE). Because of the
cytotoxic effect of cremaphor oil when combined with S-9 mix or
deltamethrin, no cells would grow in the control dish with
activation, or in the 5 mmol/litre deltamethrin dishes, either with
or without metabolic activation. A high incidence of chromosomal
aberrations and SCEs was observed in the dishes containing 1 mmol
deltamethrin/litre, with activation. However, the absence of
control values (both with and without activation) because of a
broken test tube, made the interpretation equivocal. A second
study was conducted in which deltamethrin was dissolved in DMSO and
applied to the cells at levels of 0.001, 0.01, 0.1, or 0.2
mmol/litre, with or without metabolic activation. In this study,
deltamethrin did not produce any cytotoxic effects and did not
induce either chromosomal aberrations or SCEs in CHO cells.
However, no positive controls were tested and only single plates
were prepared per dose level (Sobels et al., 1978).
Deltamethrin was found not to be mutagenic in V79 Chinese
hamster cells, in the presence or absence of hepatocytes. It is
not known which solvent was used (Pluijmen et al., 1984).
7.5.3 Mouse
An in vivo cytogenetic test was conducted on mice (3 males and
3 females per group). Mice were treated orally with deltamethrin
in sesame oil for 2 consecutive days at 5 or 10 mg/kg body weight.
The incidence of chromosomal aberrations in bone marrow cells or
micronuclei in the polychromatic erythrocytes of treated groups
was, however, comparable to that of the control groups. No
positive controls were tested (Sobels et al., 1978).
Deltamethrin was applied orally, once, at 15 mg/kg body weight
to Swiss mice. A time-related effect on the chromosomes in bone
marrow cells was observed by killing 2 animals every 3 h during
24 h. The report stated that the incidences of chromated
aberrations were low and that there were no consistent time-related
trends in the distribution of the aberrations. However, the time-
related trend of aberrations was not reported. Again, no positive
controls were tested (Sobels et al., 1978).
A dominant-lethal assay with deltamethrin was performed.
Groups of 9 - 13 male mice were dosed orally at 3 mg/kg body weight
in sesame oil for 7 days or at a single dose of 6 or 15 mg/kg body
weight in sesame oil, and mated with 6 - 18 non-treated females.
There were no effects on the rates of pre- and post-implantation
losses, while the positive control, triethylene triphosphoramide
(10 mg/kg body weight), reduced pregnancies in the second and third
weeks after treatment and increased embryonal losses (Vannier &
Glomot, 1977).
Deltamethrin in olive oil was administered orally to female
Swiss mice at single or repeated (5 times at daily intervals) doses
of 1.36, 3.4, or 6.8 mg/kg per day. Bone marrow smears were
prepared 6, 24, or 48 h after treatment. No mutagenic activity was
observed with deltamethrin, whereas the positive control,
cyclophosphamide, induced a positive response (Polàkovà & Vargovà,
1983).
In a micronucleus test, a single dose of deltamethrin in corn
oil was administered orally at 16 mg/kg body weight to Swiss CD-1
mice (5 of each sex per group). No mutagenic activity was observed
with deltamethrin, whereas the positive controls,
triethylenemelamine and dimethylbenzanthracene, both induced
positive responses (Vannier & Fournex, 1983).
7.5.4 Appraisal
Deltamethrin is not mutagenic or clastogenic in a variety of in
vitro and in vivo test systems.
7.6 Teratological and Reproductive Effects
7.6.1 Teratology
7.6.1.1 Mouse
Deltamethrin was dissolved in corn oil and administered by
gastric intubation at doses of 0, 3.0, 6.0, or 12.0 mg/kg body
weight on days 7 - 16 of gestation to groups of CD-1 mice. Mice
were sacrificed on day 18 of gestation. There was a dose-related
( P <0.001) reduction in maternal weight gain during pregnancy and
high-dose females gained 58% less weight than the controls. There
was no dose-related mortality but dams in the high- and mid-dose
groups became convulsive after dosing. Treatment did not affect
the number of implantation sites, fetal mortality, fetal weights,
or the number of sternal and caudal ossification centres.
A significant ( P <0.01) dose-related increase in the
occurrence of supernumerary ribs was observed. No other dose-
related skeletal or visceral anomalies were observed (Kavlock et
al., 1979).
Pregnant female Swiss CD-1 SPF mice (24 per group) were given
deltamethrin dissolved in sesame oil by oral intubation at dose-
levels of 0, 0.1, 1, or 10 mg/kg body weight per day on days
6 - 17 of pregnancy. The animals were necropsied on day 18 of
pregnancy. The numbers of implantation sites, fetal losses, and
viable fetuses were not affected by treatment. There was a dose-
related decrease in mean fetal weight. Apart from delayed
ossification at all dose levels, skeletal examination revealed no
abnormalities. A teratogenic effect was not observed (Glomot &
Vannier, 1977).
In a complementary teratology study, pregnant female Swiss CD-1
mice were given deltamethrin dissolved in sesame oil by oral
intubation at 0, 0.1, 1, or 10 mg/kg body weight per day from day 6
to day 17 of gestation. Females were either sacrificed on day 18
of gestation or allowed to litter for subsequent examination of
pups on days 1 or 21 of lactation. The compound caused a moderate
and transient retardation of development of the fetus at the 1 and
10 mg/kg body weight dose rate, but these effects were not observed
on days 1 or 21 post-partum. There were no teratogenic effects
related to treatment (Vannier & Glomot, 1982).
7.6.1.2 Rat
Pregnant female Sprague-Dawley rats (24 per group) received 0,
0.1, 1, or 10 mg deltamethrin/kg body weight per day by oral
intubation on days 6 - 18 of pregnancy. Apart from 12 females in
the control and 10 mg/kg groups, which were allowed to deliver, the
dams were sacrificed and examined on day 21. There were no effects
on reproduction or on the teratogenic parameters examined, except
for slightly delayed ossification at the highest dose level (Glomot
& Vannier, 1977).
Deltamethrin was dissolved in corn oil and administered by
gastric intubation at doses of 0, 1.25, 2.5, or 5.0 mg/kg body
weight on days 7 - 20 of gestation. Rats were sacrificed on day 21
of gestation. There was a dose-related reduction ( P <0.01) in
maternal weight gain during pregnancy, and dams in the high-dose
group gained only 80% of the control value. Treatment did not
affect the number of implantation sites, fetal mortality, fetal
weight, or the number of sternal and caudal ossification centres
(Kavlock et al., 1979).
7.6.1.3 Rabbit
Groups of 15 pregnant New Zealand White rabbits received
deltamethrin dissolved in sesame oil at levels of 0, 1, 4, or
16 mg/kg body weight per day during days 6 - 19 of pregnancy.
Examination was carried out on day 28 of gestation. The mean fetal
loss was not dose-related. The mean fetal weight in the highest-
dose group was decreased. Some malformations (hydrocephaly,
exencephaly, and thoracogastroschisis) were observed in 2 fetuses
of animals at the highest dose level. In a supplementary study,
pregnant rabbits were similarly dosed with 16 mg/kg body weight per
day; one fetus with spina bifida and shortened tail was detected
among 69 apparently normal fetuses. Malformations were within the
normal limits of the strain used and were not considered to be
related to the treatment, despite the occurrence at the highest
dose level only (Glomot & Vannier, 1977, 1978).
7.6.2 Reproduction studies
Groups of 10 male and 20 female Charles River rats were fed
deltamethrin in the diet at 0, 2, 20, or 50 mg/kg and mated to
begin a 3-generation, 2-litter (first generation, 3 litter)
standard reproduction study. Parental body weights and food
consumption were recorded during the study. After weaning of the
second litter, the surviving parent rats were sacrificed and
necropsied. Five male and 5 female pups of the F3b generation were
necropsied. No changes relevant to treatment were observed in
general behaviour or survival of parent rats or pups. The body
weight of F0 males of the 50 mg/kg group was decreased from week 11
onwards. There were some slight decreases in mean food consumption
of F1 male parent rats in the 50 mg/kg group. The basic
reproduction indices (fertility, gestation, lactation, viability,
and litter size) were not affected by the treatment. However, the
mean pup weight in some litters, especially in the 50 mg/kg group,
was slightly decreased in comparison to the controls on day 21 of
lactation. Gross external examination did not reveal any
abnormalities. No gross or microscopic lesions of treatment-
related significance or significant effects on the organ weights of
the F3b generation were observed (Wrenn et al., 1980).
Deltamethrin was dissolved in corn oil and administered by
gastric intubation at doses of 0, 2.5, or 5.0 mg/kg body weight to
Sprague-Dawley rats from day 7 of gestation to day 15 of lactation.
The dams were allowed to litter and rear their young: litters were
reduced at birth to 4 males and 4 females per litter. The pups
were weighed weekly and examined for the development of eye-
opening, startle reflex, and air-righting. The litters were weaned
on day 22 post-partum and the males discarded. Weekly weighing of
the females continued and at 6 weeks of age they were tested in a
circular open-field. There were no effects on parturition, litter
size, or pup viability. Weights at birth were similar for all
groups, but a dose-related depression in growth was observed during
the pre-weaning period. This early diminution in pre-weaning
weight appeared to have little effect on the morphological and
behavioural parameters measured (Kavlock et al., 1979).
7.7 Neurotoxicity and Behavioural Effects
Adult hens (10 per group) were gavaged with a single dose of 0,
500, 1250, or 5000 mg deltamethrin/kg body weight suspended in corn
oil or 0 or 100 mg/kg body weight dissolved in sesame oil. During
21 days, observations were made on mortality, health, neurotoxic
signs, and body weight. Deltamethrin did not induce any clinical,
macroscopic, or histological signs of delayed neurotoxicity (Ross
et al., 1978).
Groups of 5 male and 5 female Wistar rats were administered
25 mg deltamethrin/kg body weight in 10 mg corn oil/kg on 2
consecutive days. Controls received 10 mg corn oil/kg body weight.
A tilting plane test was performed every second day from day 4 to
day 16 of the study. Two male rats died at 25 mg/kg. No effect
was found on the slip-angle (Davies et al., 1983).
The effects of deltamethrin were studied in a rat performance
test that arranged for milk delivery after every fortieth lever
press. Deltamethrin (1 - 8 mg/kg body weight, given orally, 2 h
before the test) produced both dose-related increases in pause
duration and decreases in response rate. Deltamethrin was also
studied using a conditional flavour-aversion test. Deltamethrin-
treated, trained rats displayed an aversion to saccharin that was
greatest at 2 mg/kg (Macphail, 1981).
The neurological effects of the 4 synthetic pyrethroids,
resmethrin, permethrin, cypermethrin, and deltamethrin, have been
investigated in the rat to establish whether there is a correlation
between the clinical-functional status of the animal and peripheral
nerve damage, as measured biochemically (Rose & Dewar, 1983).
Neuromuscular dysfunction was assessed by means of the inclined
plane test and peripheral nerve damage by reference to
beta-glucuronidase and beta-galactosidase activity increases in
nerve tissue homogenates from treated and control animals. A
transient functional impairment was found in animals treated with
any one of the 4 pyrethroids tested and in all cases this was
greatest at the end of the 7-day dosing regimen (deltamethrin doses
of 5 - 20 mg/kg per day in arachis oil). Significant increases in
beta-glucuronidase and beta-galactosidase activities were found
3 - 4 weeks after the start of dosing, in the distal portion of the
sciatic/posterior tibial nerves from permethrin-, cypermethrin-,
and deltamethrin-treated animals, but no changes were found in
resmethrin-treated animals. It is concluded, therefore, that there
is no direct correlation between the time-course of the
neuromuscular dysfunction and the neurobiochemical changes. This
suggests that these pyrethroids have at least two distinct actions-
a short-term pharmacological effect at near-lethal dose levels and
a more long-term neurotoxic effect that results in sparse axonal
nerve damage.
To better characterize the behavioural toxicity of pyrethroid
insecticides, comparisons were made of the effects of cismethrin
and deltamethrin exposure on motor activity and the acoustic
startle response in male Long-Evans rats (Crofton & Reiter, 1984).
Acute dose-effect, acute time-course, and 30-day repeated-exposure
determinations of 1-h motor activity were made using figure-eight
mazes. The acoustic startle response was measured to a 13-kHz,
120-dB(A), 40-millisecond tone at each of 3 background white noise
levels (50, 65, and 80 dB). Deltamethrin (0, 2, 4, 6, or 8 mg/kg
body weight) or cismethrin (0, 6, 12, 18, or 24 mg/kg) were
administered orally in 0.2 ml/kg corn oil. Cismethrin and
deltamethrin produced similar dose-dependent decreases in motor
activity. The time course of onset and recovery for this decreased
activity was rapid (1 - 4 h). No cumulative effects on motor
activity of a 30-day exposure to 2 mg deltamethrin/kg per day or
6 mg cismethrin/kg per day were found. The effects of cismethrin
and deltamethrin on the acoustic startle response were dissimilar:
deltamethrin produced a dose-dependent decrease in amplitude and an
increase in latency, and cismethrin produced an increase in
amplitude and no change in latency. The differential effects of
cismethrin (Type I pyrethroids) and deltamethrin (Type II
pyrethroids) on the acoustic startle response may be related to the
contrasting effects previously shown with neurophysiological and/or
neurochemical techniques (see Appendix I).
7.8 Miscellaneous Effects
Analgesic effects of deltamethrin for thermic (hot plate test,
60 °C) and mechanical stimuli were investigated in mice and rats,
respectively. Deltamethrin prolonged the response-time to these
tests. Although this action was not significant at 500 mg
deltamethrin/kg body weight given orally, the reaction time was
increased at 1000 and 1500 mg/kg given orally in aqueous suspension
with 10% gum arabic (Chanh et al., 1981).
In rats, treatment with deltamethrin increased mean arterial
pressure and aortic output (Forshaw & Bradbury, 1983). The
cardiovascular effects of deltamethrin were due to both increased
catecholamine release in peripheral vascular beds, and to a direct
positive inotropic effect on the heart.
Krasnjih & Pavlova (1985) demonstrated induction of microsomal
oxygenases in rats 20 h after a single administration of 1/2 LD50.
Daily administration of 1/10th LD50 for 2 months reduced
acetylcholinesterase activity in the serum, erythrocytes, liver,
and cerebrum. It also led to some changes in the aspartate
aminotransferase activity and the urea and protein contents of
serum.
When rats were dosed orally with a single dose of 1/2 LD50 or 3
daily doses of 1/5 LD50 deltamethrin, the activities of transferrin
and ceruloplasmin in plasma, 20 h after dosing, were unchanged.
After the single dose, microsomal monooxygenase activity was
increased by 87%, and after the 3 doses, it was increased by 290%
(Kagan et al., 1986).
7.9 Potentiation
Deltamethrin was hydrolysed in vitro by esterases in blood,
brain, kidney, liver, and stomach preparations of mice.
Pretreatment of mice with the oxidase inhibitor, piperonyl butoxide
(PBO), or the esterase inhibitor, S,S,S-tributylphosphorotrithioate
(DEF), delayed metabolism of intraperitoneally administered
deltamethrin. PBO or DEF made mice more sensitive to deltamethrin
(Ruzo et al., 1979).
Plasma esterases, in addition to hepatic esterases, play a role
in the metabolism of deltamethrin in mammals and cause its rapid
detoxification by the oral route. In a potentiation study, a range
of esterase inhibitors, consisting mainly of organophosphorus
insecticides, was given to male rats in oral doses that inhibited
50% of the plasma cholinesterase. After 15 min, or 2 or 24 h, an
oral LD50 dose of deltamethrin EC formulation was given, which
showed potentiation with azinphos ethyl, omethoate, and dichlorvos.
It appears that users must handle deltamethrin in these
combinations very carefully because of their high toxicity.
Acephate, monocrotophos, phosphamidon, parathion methyl, and the 2
controls did not act as potentiators (Audegond et al., 1988).
7.10 Mechanism of Toxicity (Mode of Action)
Deltamethrin is classified as a Type II pyrethroid. For the
mode of action of pyrethroids in general see Appendix I.
The lowest concentration of deltamethrin to have an effect in
crayfish stretch receptor neurones on sodium channels was 10-12
mol/litre, but the response of the preparation to gamma-
aminobutyric acid (GABA) appeared to be unaffected by concentrations
of deltamethrin up to 10-7 mol/litre. Although 10-6 mol/litre
deltamethrin had a slight effect on the GABA response of the dactyl
abductor muscle, it appears that the majority of the effects of
cyano-pyrethroids in invertebrates could be accounted for solely by
their action on sodium channels (Chalmers et al., 1987).
Pyrethroid-induced motor symptoms, i.e., deltamethrin-induced
writhing and cismethrin-induced tremor, were studied, using a
number of pharmacological agents, in intact conscious rats and
spinal rats. The results suggest that pyrethroid-induced motor
symptoms, i.e., writhing and tremor, are mediated via a spinal site
of action, probably involving interneurones. Deltamethrin-induced
"non-motor" symptoms, i.e., increase in brain blood flow and blood
glucose may result from a supraspinal component of deltamethrin
activity. In contrast, the cardiovascular effects of deltamethrin
are mediated via a peripheral site of action (Bradbury et al.,
1983).
Tissue culture experiments have shown that the dorsal root
ganglion is more sensitive to deltamethrin than the spinal cord or
peripheral nerve fibres. The morphological alterations observed in
the neuronal bodies of the ganglia may reflect some perturbation of
the ionic equilibrium (Na+ and Ca+) (Souyri, 1985).
The results of several other, sometimes very detailed and
specialized, studies on the mode of action of deltamethrin have
been reported. Because these results do not basically influence
the present evaluation of deltamethrin, they are not reviewed in
detail. The interested reader is referred to the following
publications and a review by Bidet et al. (1988): Aldridge et al.
(1978), Duclohier & Georgescauld (1979), Gray et al. (1980),
Jacques et al. (1980), Miller & Adams (1980), Gammon et al. (1981),
Pichon (1981), Brodie & Aldridge (1982), Dyball (1982), Parkin &
LeQuesne (1982), Ray (1982), Staatz et al. (1982), Brodie (1983),
Takahashi & LeQuesne (1983), Berlin et al. (1984), Prasada Rao et
al. (1984), Bloomquist & Soderlund (1985), Brodie (1985), Brodie &
Opacka (1985), Bloomquist et al. (1986), Chinn & Narahashi (1986),
Doherty et al. (1986), Forshaw & Ray (1986), Staatz-Benson & Hosko
(1986), Brooks & Clark (1987), Forshaw et al. (1987), Leibowitz et
al. (1987), Lummis et al. (1987), Stein et al. (1987).
7.11 Experimental Studies on Antidotes
Treatments capable of counteracting acute deltamethrin
poisoning have been investigated in experimental animals. Ray &
Cremer (1979) have proposed atropine, Gammon et al. (1982),
phenobarbital and diazepam, and Bradbury et al. (1981, 1983),
mephenesin.
In order to find a therapeutically usable antagonist,
pharmacological screening was carried out by Dumont (1978), Dumont
& Chifflot (1978), and Dumont & Laurent (1979). The outcome was
that barbiturates are therapeutically active, but that the most
efficient product is ethyl carbamate. Cotonat et al. (1987) and
Fournier (1988) have confirmed that ethyl carbamate is an effective
treatment for severe deltamethrin poisoning. However, a serious
drawback is its antimitotic activity. Leclercq et al. (1986) began
by evaluating the activity of common anticonvulsants, such as
diazepam and clomethiazole. These products exhibited satisfactory
activity in rats and dogs (Thiebault et al., 1985, 1988).
Phenoprobamate and mephenesin carbamate have been shown to be
effective in the experimental treatment of deltamethrin poisoning
(Cotonat et al., 1987; Leclercq et al., 1986). A summary of the
results of the antidote studies can be found in Bleys et al.
(1986). Clinical trials based on these studies will be undertaken
(unpublished information given to the IPCS by Roussel Uclaf).
The therapeutic effects of methocarbamol have also been shown
by Hiromori et al. (1986). However, the mechanism underlying this
activity is not clear as the actual anticonvulsant activity is
weak.
It appears that for the time being barbiturates, and especially
diazepam, offer the safest symptomatic treatment in case of
deltamethrin poisoning. Advice on treatment for deltamethrin
poisoning is given in the IPCS Deltamethrin health and safety guide
(WHO, 1989).
8. EFFECTS ON MAN
8.1 General Population-Poisoning Incidents
A few cases of attempted suicides with deltamethrin
formulations (mainly EC), all non-fatal, have been reported in
anti-poison centres. Two typical cases are described below.
The first poisoning case in a 13-year-old girl who ingested
voluntarily 200 ml of a 2.5% EC formulation, (5 grams of
deltamethrin) was described by Rousselin (1983). After an unknown
time, she lost consciousness and developed generalized muscle
cramps, myosis, and tachycardia. Treatment in hospital was as
follows: gastric lavage, PAM 0.5 mg; atropine 2 mg; sodium
nitrite, 3% sodium thiosulfate; and, lastly, high doses of
diazepam. She completely recovered in 48 h.
A second poisoning case, concerning another attempted suicide
by a 23-year-old man, was reported by Foulhoux (1988). After oral
absorption of 70 cc of a 2.5% EC formulation (1.75 g pure
deltamethrin), there were no neurological signs in this patient.
Digestive and hepatic signs occurred, probably due to absorption of
the solvent, since determination of xylene in plasma was positive.
The patient was treated with haemodialysis, phenobarbital,
lidocaine, and provoked alkaline diuresis. Recovery followed
within 48 h.
8.2 Occupational Exposure
8.2.1 Acute toxicity-poisoning incidents
Rousselin (1983) described a case of poisoning in an
agricultural worker as a result of skin contamination with a liquid
containing 5 g deltamethrin/litre. He developed paraesthesia in
the legs, mouth, and tongue, and diarrhoea. Following washing of
the skin and administration of antihistamines, he still had
tingling sensations in his toes after 24 h, but was fully recovered
after 48 h.
Outbreaks of acute deltamethrin and fenvalerate poisoning
occurred in cotton growers in China in 1982 - 84. The farmers
handled the pyrethroid insecticides without taking any precautions.
Skin sensations occurred in more than 90% of the exposed workers.
After repeated spraying in the cotton fields, the mild cases
presented severe headaches, dizziness, fatigue, nausea, and
anorexia, with transient changes in the EEG. A severe case
developed muscular fasciculation, repetitive discharges in the EMG,
and frequent convulsions, which were treated with diazepam and
phenobarbital. However, in follow-up studies, all workers were
found to have made complete recovery, and the prognosis of acute
pyrethroid poisoning was found to be good (He, 1987; Tong Ying,
1988).
More recently He et al. (1989) reviewed 573 cases of acute
pyrethroid poisoning reported in the Chinese medical literature
during 1983 - 88. Among these there were 325 cases of acute
deltamethrin poisoning: 158 occupational, due to inappropriate
handling, and 167 accidental, mostly due to ingestion. Two
patients died of convulsions. All others recovered with
symptomatic and supportive treatment within 1 - 6 days. Clinical
manifestations are well reviewed (He, 1987).
8.2.2 Effects of short- and long-term exposure
Among plant workers dermally exposed to technical deltamethrin
or its formulations, cutaneous and mucuous manifestations were
observed. Initial lesions were tenacious and painful pruritus,
especially observed after exposure to hot water or perspiration,
followed by a blotchy local burning sensation with blotchy erythema
for about 2 days. Thereafter, slight and regular desquamation,
restricted to the contaminated area, occurred. Cutaneous signs
were sometimes accompanied by itching of the face (mainly around
the mouth) and/or rhinorrhoea or lachrymation (Husson, 1978).
Apart from the above-mentioned effects, no long-term or
persistent effect, or allergic diseases were reported in 70
workers, who had been exposed from 1977 - 87 in a deltamethrin-
manufacturing and -formulating plant in France (unpublished
Roussel Uclaf information supplied to the IPCS, 1988).
A field study was carried out in the United Kingdom with three
unprotected operators and one operator wearing hood, gloves, and
respirator, all of whom were involved in orchard spraying with
deltamethrin according to normal field practice. The exposure time
was 3.5 h. No changes were found in blood cell counts, total
protein, urea, alkaline phosphates, gamma-GT and SGOT in blood.
Little deltamethrin was found in the respirator pad and no residues
were found in the urine. There was no decrease in nerve conduction
velocity, but a slight tendency to the opposite reaction. None of
the operators experienced facial sensations (Hewson & Burgess, 1981).
Four operators were evaluated by the same authors during normal
field applications of deltamethrin lasting one day. Three of the
operators did not wear any protection on their heads or hands,
while one wore hood, gloves, and a respirator. Motor and sensory
nerve conduction velocities were determined as well as
haematological and biochemical parameters and urinalysis. No
changes were observed in the blood parameters measured and no
residues were found in the urine samples. Furthermore, nerve
conduction velocity did not decrease. Residues were primarily
confined to gloves and legs. None of the operators experienced
facial sensations (Hewson & Burgess, 1981).
Persons exposed to deltamethrin for 7 - 8 years in production
and formulation were subjected to clinical and haematological
examinations. Evaluations were conducted at several plants. There
were no measurable effects other than transient irritation of
cutaneous and mucous membranes, which was without sequelae.
Adequate precautionary measures, such as the wearing of gloves and
face masks, provided protection from exposure (Foulhoux, 1981).
A medical survey of agricultural workers involved in the use
and application of EC and WP formulations of deltamethrin in
Yugoslavia revealed no untoward symptoms of exposure, other than
itching and burning of the face, and nasal hypersecretion. Medical
examinations included chest X-ray, ECG, liver function tests,
neurological examinations (eye tonometry, Goldman perimetry, dark
adaptation ability), kidney function tests, and whole blood and
plasma cholinesterase activity. No adverse effects were noted.
The need for the proper use of masks and gloves, as well as good
personal hygiene (e.g., washing), was emphasized (Plestina, 1981).
Five healthy volunteers, 16 - 40 years of age, were exposed to
deltamethrin during 5 days of spraying in a cotton field in India
in 1981. A sixth volunteer was engaged in mixing and loading the
emulsion during the same period. Spraymen were exposed for 7 h
daily. No one complained about any symptoms. No clinical
abnormalities were detected, particularly with respect to
neurological examination (muscle power, coordination, tremors,
reflexes, and both light and deep sensations). No cardiovascular,
respiratory, or abdominal abnormalities were detected, and no skin,
mucous membrane, or eye lesions were observed during, and after
cessation of, exposure (Trivedi, 1981).
A health survey was carried out among spraymen exposed to 2.5%
deltamethrin emulsifiable concentrate in cotton fields in China.
The subjects were exposed to deltamethrin at concentrations of
0.022 - 24.070 µg/m3 in the air of the respiratory zone and
0.013 - 0.347 µg/cm2 of skin contact. One half of the 44 sprayers
complained of itching and burning sensations on their faces. A few
miliary red papules also appeared on the face of one of them, but
no signs of acute deltamethrin poisoning were noticed during
physical examination. There were no significant differences in the
sodium, potassium, and urea contents of the serum, the sodium,
potassium, ATPase, and serotonin contents of whole blood, and the
levels of 3-methyl-4-hydroxymandelic acid and 5-hydroxy-
indoleacetic acid in the urine between the subjects examined and
the controls. Deltamethrin in the urine of spraymen was below the
detection limit of 0.10 µg/litre (Wang et al., 1988).
Mestres et al. (1985) measured the dermal and inhalation
exposure of mixer/applicators who applied deltamethrin to vegetable
crops in greenhouses in southern France and of workers who picked
fruit from treated trees in the same area. This appeared to be
less than 0.0065% of the toxic dose per hour with a mask and
0.0017%, without a mask.
In a department producing an aerosol of the domestic Bulgarian
insecticide "Dekazol" containing 0.02, 0.04, or 0.08% deltamethrin,
severe subjective complaints of sensory irritation were found
because of the high levels of contamination of the workplace air
with deltamethrin and also dermal contamination. Skin irritation
with conjunctivitis and irritation of the respiratory system were
discovered in all 25 workers. Two of them had contact urticaria.
Patch testing with 0.03% deltamethrin showed a positive reaction in
5 out of 23 workers tested (Bainova et al., 1986).
8.3 Clinical Studies
Three formulations of deltamethrin in petroleum solvent were
patch tested on 37 human volunteers (double blind trial against
solvent control). A dose of 20 µl of a 1% suspension in water, of
a 25 g/litre emulsifiable concentrate was put on the facial skin of
each volunteer, with a randomized distribution of control and
active dilution. The duration of the irritation was short (from
some minutes to 1 h) and the severity was described as slight by
most of the volunteers. No skin damage was reported (Foulhoux et
al., 1981).
9. PREVIOUS EVALUATIONS BY INTERNATIONAL BODIES
The Joint FAO/WHO Meeting on Pesticide Residues (JMPR)
discussed and evaluated deltamethrin at its meetings in 1980, 1981,
1982, 1984, 1985, 1986, 1987, and 1988 (FAO/WHO, 1981, 1982, 1983,
1985a, 1986a, 1986b, 1988a,b,c). In 1982, an acceptable daily
intake (ADI) of 0 - 0.01 mg/kg body weight was established.
The following Maximum Residue Limits (MRLs), in mg/kg, resulted
from these evaluations:
tea 10.0
hops dry, wheat bran unprocesseda 5.0
coffee beans (post-harvest) 2.0
wheat wholemeal,a cereal grains,a 1.0
(ph) lentil (dry),a beans (dry),a
field pea (dry)a
straw and fodder (dry) of cereal 0.5
grains, legume animal feeds (dry
weight), leafy vegetables
brassica leafy vegetables,a edible 0.2
peel of fruiting vegetablesa
bulb vegetables, edible peel of 0.1
assorted fruits, legume vegetables,
oilseeds, pome fruits, wheat floura
artichokes, bananas, clementines, 0.05
coco beans, grapes, kiwi fruit,
oranges (sweet, sour), stone
fruits/strawberries
legume oilseeds, melons, 0.01
mushrooms, pineapples, root and
tuber vegetables, milksa
WHO has classified deltamethrin as a moderately hazardous
technical product in normal use (WHO, 1988). A Data Sheet on
deltamethrin (No. 50) has been issued (WHO/FAO, 1984).
-------------------------------------------------------------------
a Not yet confirmed by Codex Alimentarius Commission (FAO/WHO,
1986c, and 1988c).
REFERENCES
ADENEY, R.J., GRZYWACZ, D., & MATTHIESSEN P. (1980) The acute
toxicities of pyrethrin analogues to Sarotherodon mossambicus
(Peters), Centre for Overseas Pest Research, 6 pp. (Unpublished
proprietary data submitted to WHO by Roussel Uclaf).
AKHTAR, M.H. (1984) Metabolism of deltamethrin by cow and chicken
liver enzyme preparations. J. agric. food Chem., 32: 258-262.
AKHTAR, M.H., HAMILTON, R.M.G., & TRENHOLM, H.L. (1985)
Metabolism, distribution and excretion of deltamethrin by leghorn
hens. J. agric. food Chem., 33: 610-617.
AKHTAR, M.H., MARTIN, K.E., & TRENHOLM, H.L. (1986) Fate of
14C-deltamethrin in lactating dairy cows. J. agric. food Chem.,
34: 753-758.
AKHTAR, M.H., DANIS, C., TRENHOLM, H.L., & MARTIN, K.E. (1987)
Residues in milk and tissues of lactating dairy cows fed
deltamethrin for 28 consecutive days. J. agric. food Chem.,
ALDRIDGE, W.N., CLOTHIER, B., FORSHAW, P., & JOHNSON, M.K., PARKER,
V.H., PRICE, R.J., SKILLETER, D.N., VERSCHOYLE, R.D., STEVENS,
C. (1978) The effect of DDT and the pyrethroids cismethrin and
decamethrin on the acetyl choline and cyclic nucleotide content of
rat brain. Biochem. Pharmacol., 27: 1703-1706.
ALINIAZEE, M.T. & CRANHAM, J.E. (1980) Effects of four synthetic
pyrethroids on a predatory mite, Typhlodroums pyri, and its prey,
Panonychus ulmi, on apples in southeast England. Environ. Entomol.,
9: 436-439.
ARZONE, A. & VIDANO, C. (1978) [Azione sull'ape di etiofencaus,
decamethrin e ciexatin.] Apic. mod., 69: 157-162, (in Italian).
ATKINS, E.L., KELLUM D., & NEUMAN, K.J. (1976) Effect of
pesticides on apiculture project No. 1449: The annual report,
University of California Riverside, pp. 536-567.
AUDEGOND, L., CATEZ, D., FOULHOUX, P., FOURNEX, R., LE RUMEUR, C.,
L'HOTELLIER, M., & STEPNIEWSKI, J.P. (1988) Potentialisation de
la toxicité de la deltaméthrine par les insecticides
organophosphorés (Unpublished proprietary data submitted to WHO by
Roussel Uclaf).
AUDEGOND L., COLLAS E., & GLOMOT R. (1981) RU 22974
(deltamethrin) single administration study by oral route in the rat
(the compound is given as a suspension) (Unpublished report RU-
81239/A, submitted to WHO by Roussel Uclaf).
BAINOVA, A. & KALOYANOVA, F. (1985) [Study of allergenic and
irritating effect of synthetic pyrethroids on skin.] Hig. Zdrav.,
28(2): 19-21 (in Bulgarian).
BAINOVA, A., MIHAVSKI, M., ISMIROVA, N., & BENCHEV, V. (1986)
Specific skin irritation after contact with specific pyrethroids.
6th Congress of Bulgarian Dermatologists, Varna, 2-5 October, 1986,
(Abstract No. 74) (in Bulgarian).
BAKER, P.G. & BOTTOMLEY, P. (1982) Determination of residues of
synthetic pyrethroids in fruit and vegetables by gas-liquid and
high-performance liquid chromatography. Analyst, 107: 206-212.
BALDRY, D.A.T., EVERTS, J., ROMAN, B., BOON VON OCHSSEE, G.A., &
LAVEISSIERE, C. (1981) The experimental application of
insecticides from a helicopter for the control of riverine
populations of Glossina tachinoides in West Africa. Part VIII: The
effects of two spray applications of OMS-570 (endosulfan) and of
OMS 1998 (decamethrin) on G. tachinoides and non-target organisms
in Upper Volta. Trop. Pest Manage., 27(1): 83-110.
BEAVERS, J.B. & FINK, R. (1977a) Actue oral LD50 Mallard duck
technical DECIS final report, (Unpublished report WI77.06.06/A,
submitted to WHO by Roussel Uclaf). Wildlife International.
BERLIN, J.R., AKERA, T., BRODY, T.M., & MATSUMURA, F. (1984) The
inotropic effects of a synthetic pyrethroid decamethrin on isolated
guinea pig atrial muscle. Eur. J. Pharmacol., 98: 313-322.
BIDET, D., DUMONT, C., FOULHOUX, P., FOURNEX, R., & ROUGERON, C.
(1988) Mechanisms responsible for the toxicity of deltamethrin and
other pyrethroids. (Unpublished document, submitted to WHO by
Roussel Uclaf).
BLEYS, M., COTONAT, J., FOULHOUX, P. (1986) Lettre à l'éditeur,
J. Toxicol. clin. exp., 6(3): 211-212.
BLOOMQUIST, J.R., SODERLUND, D.M. (1985) Neurotoxic insecticides
inhibit GABA-dependent chloride uptake by mouse brain vesicles.
Biochem. biophys. Res. Commun., 133(1): 37-43.
BLOOMQUIST, J.R., ADAMS, P.M., & SODERLUND, D.M. (1986)
Inhibition of gamma-aminobutyric acid-stimulated chloride flux in
mouse brain vesicles by polychlorocycloalkane and pyrethroid
insecticides. Neurotoxicology, 7(3): 11-20.
BOCQUET, J.C., PASTRE, P., ROA, L., & BAUMEISTER, R. (1980) Etude
de l'action de la deltaméthrine sur Apis mellifera en conditions de
plein-champ. Phytiatr. Phytopharm., 29: 83-92.
BOCQUET, J.C., PASTRE, P., & BAUMEISTER, R. (1983) Bilan de cinq
années d'études de l'effet de la deltaméthrine sur abeilles en
conditions naturelles. 6ème Congrès International du Colza, Paris,
mai 1983.
BOUCHE, M.B. & FAYOLLE, L. (1979) Tests carried out with Procida
compound Decis EC 2.5 on earthworms lethal effect in relation with
time (Unpublished proprietary report No. INRA-VT-79.05.22/78.10.
10/A, submitted to WHO by Roussel Uclaf).
BOWMAN, H. & CARPENTER, M. (1987) Determination of
photodegradation of 14C-deltamethrin in aqueous solution
(Unpublished proprietary report ABC LABS 35491, submitted to WHO by
Roussel Uclaf).
BRADBURY, J.E., GRAY, A.J., & FORSHAW, P. (1981) Protection
against pyrethroid toxicity in rats with mephenesin. Toxicol.
appl. Pharmacol., 60: 382.
BRADBURY, J.E., FORSHAW, P.J., GRAY, A.J., & RAY, D.E. (1983) The
action of mephenesin and other agents on the effects produced by
two neurotoxic pyrethroids in the intact and spinal rat.
Neuropharmacology, 22(7): 907-914.
BRODIE, M.E. (1983) Correlations between cerebellar cyclic GMP
and motor effects induced by deltamethrin: independence of olivo-
cerebellar tract. Neurotoxicology, 4(4): 1-11.
BRODIE, M.E. (1985) Deltamethrin infusion into different sites in
the neuraxis of freely moving rats. Neurobehav. Toxicol. Teratol.,
7(1): 51-55.
BRODIE, M.E. & ALDRIDGE, W.N. (1982) Elevated cerebellar cyclic
GMP levels during the deltamethrin-induced motor syndrome.
Neurobehav. Toxicol. Teratol., 4: 109-113.
BRODIE, M.E. & OPACKA, J. (1985) Dissociation between circling
behaviour and striatal dopamine activity following unilateral
deltamethrin administration to rats. Naunyn-Schmiedeberg's Arch.
Pharmacol., 331: 341-346.
BROOKS, M.W. & CLARK, J.M. (1987) Enhancement of norepinephrine
release from rat brain synaptosomes by alpha cyano pyrethroids.
Pestic. Biochem. Physiol., 28: 127-139.
BUCCAFUSCO, R.J., ELLS, S.J., & CARY, G.A. (1977a) Acute toxicity
of NRDC 161 to bluegill (Lepomis macrochirus) under dynamic test
conditions, E.G. & G. Bionomics Aquatic Toxicology Laboratory, 12 pp.
(Unpublished proprietary data submitted to WHO by Roussel Uclaf).
BUCCAFUSCO R.J., ELLS, S.J., & CARY, G.A. (1977b) Acute toxicity
of NRDC 161 (Decamethrine) to Channel Catfish (Ictalurus
punctatus), E.G. & G. Bionomics Aquatic Toxicology Laboratory,
7 pp. (Unpublished proprietary data supplied to WHO by Roussel
Uclaf).
CABRAL, J.R.P., GALENDO, D., LAVAL, M., & LYANDRUT, N. (1986)
Carcinogenicity study of the pesticide deltamethrin in mice and
rats. Summary Report in Poster Session at IUPAC Meeting, Ottawa,
August 1986.
CARY, G.A. (1978) Kinetics of 14C-NRDC-161 in a model aquatic
ecosystem, E.G. & G. Bionomics Aquatic Toxicology Laboratory.
(Unpublished proprietary report BW-78-2-075, submitted to WHO by
Roussel Uclaf).
CHALMERS, A.E., MILLER, T.A, & OLSEN, R.W. (1987) Deltamethrin:
a neurophysiological study of the sites of action. Pestic.
Biochem. Physiol., 27: 36-41.
CHAMBON, A. & LEPAILLEUR, H. (1984) Etude de l'effet toxique de
la deltamethrine (produit technique > 98%) et de la formulation CE
25 g/l Decis vis-à-vis de l'espece de vers (Eisenia fetida andrei)
(Unpublished proprietary report IRCHA-84.30.07/F, submitted to WHO
by Roussel Uclaf).
CHANH, P.H., NAVARRO-DELMASURE, C., CHANH, A.P.H., LEAN, C.S.,
ZIADE, F., & SAMAHA, F. (1981) Analgesic effects of decamethrin,
Surg. Transplant., 9: 503-504.
CHAPMAN, R.A. & HARRIS, C.R. (1981) Persistence of four
pyrethroid insecticides in a mineral and organic soil. J. environ.
Sci. Health, B16: 605-615.
CHAPMAN, R.A., TU, C.M., HARRIS, C.R., & COLE, C. (1981)
Persistence of five pyrethroid insecticides in sterile and natural,
mineral and organic soil. Bull. Environ. Contam. Toxicol, 26:
513-519.
CHESTERMAN, H., HEYWOOD, R., PERKIN, C.J., BEARD, D., STREET, E., &
PRENTICE, D.E. (1977) RU 22974. Oral toxicity study in Beagle
dogs, Huntingdon, Huntingdon Research Centre, (Unpublished report
RSL 253/7751/A3, submitted to WHO by Roussel Uclaf).
CHINN, K. & NARAHASHI, T. (1986) Stabilization of sodium channel
states by deltamethrin in mouse neuroblastoma cells. J. Physiol.,
380: 191-207.
CLAIR, M. (1977) RU 22974 DECIS. Acute toxicity in the rabbit by
percutaneous administration. Joinville-le-Pont, Institut Français
de Recherches et Essais Biologique (Unpublished report IFREB-R
770257.1/A, submitted to WHO by Roussel Uclaf).
CLARK, G.C., JACKSON, G.C., & ALEXANDER, D.J. (1980) Acute
inhalation toxicity in rats, 4-hour exposure (Decis PM 2.5
percent), Huntingdon Research Centre, (Unpublished report RSL
437/80568, submitted to WHO by Roussel Uclaf).
COOMBS, D.W. & CLARK, G.C. (1978) RU 22974: Acute inhalation
toxicity in rats. 6 Hour LC50. Huntingdon, Huntingdon Research
Centre, (Unpublished report RSL 310/78453/A, submitted to WHO by
Roussel Uclaf).
COOMBS, D.W., CLARK, G.C., STREET, A.E., & GIBSON, W.A. (1978) RU
22974 inhalation toxicity study in rats 14 x 6 hour exposures over
a period of 3 weeks (Unpublished proprietary report
RSL/318/78638/A, submitted to WHO by Roussel Uclaf).
COQUET, B. (1976a) RU 22974. Test to determine primary cutaneous
irritation in the rabbit. Joinville-le-Pont, Institut Francais de
Recherches et Essais Biologiques (Unpublished report IFREB-R
761157/A, submitted to WHO by Roussel Uclaf).
COQUET, B. (1976b) RU 22974. Test to evaluate ocular irritation
in the rabbit, Joinville-le-Pont, Institut Français de Recherches
et Essais Biologiques (Unpublished report IFREB- R 761158/A,
submitted to WHO by Roussel Uclaf).
COQUET, B. (1977) RU 22974. Decis formulations - Determination of
the LD50 in the rat by oral administration, Joinville-le-Pont,
Institut Français de Recherches et Essais Biologiques (Unpublished
report 770257-A, submitted to WHO by Roussel Uclaf).
COTONAT, J., BLEYS, M., & FOULHOUX, P. (1987) Effets antagonistes
du phenprobamate et du carbamate de mephenesine sur l'intoxication
à la deltaméthrine. J. Toxicol. clin. exp., 7: 5-19.
CROFTON, K.M. & REITER, L.W. (1984) Effects of two pyrethroid
insecticides on motor activity and the acoustic startle response in
the rat. Toxicol. appl. Pharmacol., 75: 318-328
DAVID, D. (1981) Laboratory evaluation of repellent properties
against birds of the synthetic pyrethroid decamethrin. Poult. Sci.,
60: 1149-1151.
DAVIES, J.E., MUNT, P.L., & GOOR, J.L. (1983) RU 22974.
Investigation of possible neurological effects using the tilting
plane test. Huntingdon, Huntingdon Research Centre (Unpublished
proprietary report RSL603/83232/A, submitted to WHO by Roussel
Uclaf).
DE LAVAUR, E., LE SECH, J., & GROLLEAU, G. (1985) Accumulation et
élimination de la deltaméthrine chez la caille japonaise. Ann.
Fals. Exp. Chim., 835: 73-78.
DOHERTY, J.D., LAUTER, C.J., & SALEM, N., Jr (1986) Synaptic
effects of the synthetic pyrethroid resmethrin in rat brain in
vitro. Comp. Biochem. Physiol., C84(2): 373-9.
DUCLOHIER, H. & GEORGESCAULD, D. (1979) The effects of the
insecticide decamethrin on action potential and voltage-clamp
currents of myxicola giant axon. Comp. Biochem. Physiol., C62(2):
217-223.
DUMONT, C. & CHIFFLOT, L. (1978) Recherche d'un antidote contre
les effets toxiques aigus (Unpublished report RU-AE-72, submitted
to WHO by Roussel Uclaf).
DUMONT, C. & LAURENT, J. (1979) Recherche d'antagonistes des
effets neurotoxiques aigus du RU 22 974 (Unpublished report RU-AG-
35, submitted to WHO by Roussel Uclaf).
DUNNING, R.A., COOPER, J.M., WARDMAN, J.M., & WINDER, G. (1981)
Susceptibility of the carabid Pterostichus melanarius (Illiger) to
aphicide sprays applied to the sugar-beet crop. (Unpublished
proprietary report UK-SOIL-F.81/A, submitted to WHO by Roussel
Uclaf).
DYBALL, R.E.J. (1982) Inhibition by decamethrin and resmethrin of
hormone release from the isolated rat neurohypophysis - a model
mammalian neurosecretory system. Pestic. Biochem. Physiol., 17:
42-47.
ELLIOTT, M. (1977) Synthetic pyrethroids, Washington, DC,
American Chemical Society, p. 229 (ACS Symposium Series 42).
ELLIOTT, M., FARNHAN, A.W., JANES, N.F., NEEDHAM, P.H., & PULMAN,
D.A. (1974) Synthetic insecticide with a new order of activity.
Nature, 248: 710-711.
ESTESEN, B.J., BUCH, N.A., & WARE, G.W. (1979) Dislodgeable
insecticide residue on cotton foliage; permethrin, curacron,
fenvalerate, sulprofos, decis and endosulfan. Bull. environ.
Contam. Toxicol., 22: 245-248.
EVANS, M.H. (1976) End-plate potentials in frog muscle exposed to
a synthetic pyrethroid. Pestic. Biochem. Physiol., 6: 547-550.
EVERTS, J.W., VAN FRANKENHUYZEN, K., ROMAN, B., & KOEMAN, J.H.
(1983) Side effects of experimental pyrethroid applications for
the control of tsetse flies in a riverine forest habitat in Africa.
Arch. environ. Contam. Toxicol., 12: 91-97.
EVERTS, J.W., KORTENHOFF, B.A., HOOGLAND, H., VLUG, H.J., JACQUE,
R., & KOEMAN. J.H. (1985) Effects on man-target terrestrial
arthropods of synthetic pyrethroids asked for the control of the
Tsetse Fly ( Glossina spp.) in settlement areas of the Southern
Ivory Coast, Africa. Arch. environ. Contam. Toxicol., 14: 647-650.
FAO (1982) Second Government Consultation on International
Harmonization of Pesticide Registration Requirements, Rome, 11-15
October, 1982, Rome, Food and Agriculture Organization of the
United Nations.
FAO/WHO (1981) 1980 Evaluations of some pesticide residues in
food, Rome, Food and Agriculture Organization of the United Nations
(FAO Plant Production and Protection Paper No. 26 Sup).
FAO/WHO (1982) 198l Evaluations of some pesticide residues in
food, Rome, Food and Agriculture Organization of the United Nations
(FAO Plant Production and Protection Paper No. 42).
FAO/WHO (1983) 1982 Evaluations of some pesticide residues in
food, Rome, Food and Agriculture Organization of the United Nations
(FAO Plant Production and Protection Paper No. 49).
FAO/WHO (1985a) 1984 Evaluations of some pesticide residues in
food, Rome, Food and Agriculture Organization of the United Nations
(FAO Plant Production and Protection Paper No. 67).
FAO/WHO (1985b) Guide to Codex recommendations concerning
pesticide residues. Part 8. Recommendations for methods of
analysis of pesticide residues, 3rd ed., Rome, Food and Agriculture
Organization of the United Nations, Codex Committee on Pesticide
Residues.
FAO/WHO (1986a) 1985 Evaluations of some pesticide residues in
food. Part I - Residues, Rome, Food and Agriculture Organization
of the United Nations (FAO Plant Production and Protection Paper
No. 72/1).
FAO/WHO (1986b) 1986 Evaluations of some pesticide residues in
food. Part I - Residues, Rome, Food and Agriculture Organization
of the United Nations (FAO Plant Production and Protection Paper
No. 78).
FAO/WHO (1986c) Codex maximum limits for pesticide residues, 2nd
ed., Rome, Food and Agriculture Organization of the United Nations,
Codex Alimentarius Commission, CAC XIII.
FAO/WHO, (1988a) 1987 Evaluations of some pesticide residues in
food. Part I - Residues, Rome, Food and Agriculture Organization
of the United Nations (FAO Plant Production and Protection Paper
No. 86/1).
FAO/WHO, (1988b) 1988 Evaluations of some pesticide residues in
food. Part 1 - residues, Rome, Food and Agriculture Organization
of the United Nations. (FAO Plant Production and Protection Paper
93/1.
FAO/WHO, (1988c) Supplement 1 to Codex maximum limits for pesticide
residues, Rome, Food and Agriculture Organization of the United
Nations.
FISCHER, L. & CHAMBON, J.P. (1987) Faunistical inventory of
cereal arthropods after flowering and incidence of insecticide
treatments with deltamethrin, dimethoate and phosalone on the
apigeal fauna. Meded. Fac. Landbouwwet. Rijksuniv. Gent, 52(2a):
FLANNIGAN, S.A. & TUCKER, S.B. (1985) Variation in cutaneous
sensation between synthetic pyrethroid insecticides. Contact
Dermatitis, 13: 140-147.
FLORELLI, F., GARNIER, P., & ROA, L. (1987a) Bilan de 8 années
d'expérimentation sur la selectivite du Decis vis à vis des
abeilles - La défense des végétaux, 243: 8
FLORELLI, F., HELLER, J.J., GARNIER, P., & BAUMEISTER, R. (1987b)
Incidence sur les abeilles, les ruches et leur production, de
traitements aériens du colza avec la deltaméthrine - Annales ANPP -
Conférence Internationale sur les Ravageurs en Agriculture, Paris,
1-3 décembre 1987, 189-203.
FORSHAW, P.J. & BRADBURY, J.E. (1983) Pharmacological effects of
pyrethroids on the cardiovascular system of the rat. Eur. J.
Pharmacol., 91: 207-213.
FORSHAW, P.J. & RAY, D.E. (1986) The effects of two pyrethroids,
cismethrin and deltamethrin, on skeletal muscle and the trigeminal
reflex system in the rat. Pestic. Biochem. Physiol., 25: 143-151.
FORSHAW, P.J., LISTER, T., & RAV, D.E. (1987) The effects of two
types of pyrethroid on rat skeletal muscle. Eur. J. Pharmacol.,
134(1): 89-96.
FOUILLET, X. (1976) RU 22974 Mutagenicity study of various
preparations. Salmonella microsome test, Institut Français de
Recherches et Essais Biologiques (Unpublished report no. IFREB-R
761153/A, submitted to WHO by Roussel Uclaf).
FOULHOUX, P. (1981) Medical observations of personnel working on
synthesis or formulation of deltamethrin (Unpublished report RU-
81.18.06/A, submitted to WHO by Roussel Uclaf).
FOULHOUX, P. (1988) Assessment of potential adverse effects of
deltamethrin on man, Département Central de Toxicovigilance Roussel
Uclaf, 16 pp. (Unpublished report submitted to WHO by Roussel
Uclaf).
FOULHOUX, P., BOGGIN, CORNILLIET, POTTIER, RAYNAUD, STERN-VEYRIN, &
SUTTET (1981) Cutaneous irritation patch test on human volunteers
with Decis EC25, Decis flowable 25, Decis + mineral oil 25
(Unpublished report RU-81.03.02/DM/A, submitted to WHO by Roussel
Uclaf).
FOURNIER, P.E. (1988) Etude de l'activité antidote du carbamate
d'éthyl dans l'intoxication aiguë par la deltaméthrine chez le rat,
Paris, Unité de Pharmacologie Clinique, Hospital Fernand Widal
(Unpublished report submitted to WHO by Roussel Uclaf).
GAINES, T.B. & LINDER, R.E. (1986) Acute toxicity of pesticides
in adult and weanling rats, Fundam. appl. Toxicol., 7: 299-308.
GAMMON, D.W. & CASIDA, J.E. (1983) Pyrethroids of the most potent
class antagonize GABA action at the crayfish neuromuscular
junction. Neurosci. Lett., 40: 163-168.
GAMMON, D.W., BROWN, M.A., & CASIDA, J.E. (1981) Two classes of
pyrethroid action in the cockroach. Pestic. Biochem. Physiol., 15:
181-191.
GAMMON, D.W., LAWRENCE, L.J., & CASIDA, J.E. (1982) Pyrethroid
toxicology: Protective effects of diazepam and phenobarbital in
the mouse and the cockroach. Toxicol. appl. Pharmacol., 66:
290-296.
GLICKMAN, A.H. & CASIDA, J.E. (1982) Species and structural
variations affecting pyrethroid neurotoxicity. Neurobehav.
Toxicol. Teratol., 4(6): 793-799.
GLOMOT, R. (1979) Acute toxicity study by oral route in the rat
(Proprietary report RU-79803-54/A2, submitted to WHO by Roussel
Uclaf).
GLOMOT, R. & CHEVALIER, B. (1976a) RU 22974. Acute toxicity
study mouse and rat by oral route (Unpublished report TOX 76810/A,
submitted to WHO by Roussel Uclaf).
GLOMOT, R. & CHEVALIER, B. (1976b) RU 22974. Acute toxicity
study mouse and rat by intraperitoneal route (Unpublished report
TOX 76811/A, submitted to WHO by Roussel Uclaf).
GLOMOT, R. & CHEVALIER, B. (1976c) RU 22974. Acute toxicity
study mouse and rat by intravenous route (Unpublished report TOX
76812/A, submitted to WHO by Roussel Uclaf).
GLOMOT, R. & VANNIER, B. (1977) RU 22974. Teratological study in
mouse, rat and rabbit (Unpublished report TOX 76534-76536/2/A,
submitted to WHO by Roussel Uclaf).
GLOMOT, R. & VANNIER, B. (1978) RU 22974. Teratological study in
rabbit. Complementary information (Unpublished report TOX 76534-
76536/A3, submitted to WHO by Roussel Uclaf).
GLOMOT, R., AUDEGOND, L., & COLLAS, E. (1979) Acute oral toxicity
study in the rat (Decis 25 G/L Mixofluid) (Proprietary report RU
79824/A, submitted to WHO by Roussel Uclaf).
GLOMOT, R., AUDEGOND, L., & COLLAS, E. (1980a) Single
administration by oral route in the mouse (Decis Wettable Powder,
2.5%) (Proprietary report RU-80817/A, submitted to WHO by Roussel
Uclaf).
GLOMOT, R., AUDEGOND, L., & COLLAS, E. (1980b) Single
administration study by oral route in the dog (Decis Wettable
Powder, 2.5%) (Proprietary report RU 80194/A, submitted to WHO by
Roussel Uclaf).
GLOMOT, R., AUDEGOND, L., & COLLAS, E. (1981a) Single
administration study by oral route in the rat (Proprietary report
RU-81239/A, submitted WHO by Roussel Uclaf).
GLOMOT, R., AUDEGOND, L., & COLLAS, E. (1981b) Primary dermal
irritation study in the rabbit. Deltamethrin 25 G/L Mixofluid
(Proprietary report RU-79193/A, submitted to WHO by Roussel Uclaf).
GLOMOT, R., AUDEGOND, L., & COLLAS, E. (1981c) Primary dermal
irritation study in the rabbit (Deltamethrin wettable powder, 2.5%)
(Proprietary report RU-80200/A, submitted to WHO by Roussel Uclaf).
GLOMOT, R., AUDEGOND, L., & COLLAS, E. (1981d) Primary eye
irritation study in the rabbit (Deltamethrin in 25 G/L Mixofluid)
(Proprietary report RU-79192/A, submitted to WHO by Roussel Uclaf).
GLOMOT, R., AUDEGOND, L., & COLLAS., E. (1981e) Primary eye
irritation study in the rabbit (Decis Wettable Powder, 2.5%)
(Proprietary report RU-79192/A, submitted WHO by Roussel Uclaf).
GLOMOT, R., CHEVALIER, B., COLLAS, E., & AUDEGOND, L. (1977) RU
22974. Acute toxicity study by oral route in male and female
Beagle dogs (Unpublished report TOX 77804/JL-5, submitted to WHO
by Roussel Uclaf).
GOLDENTHAL, E.I., BLAIR, M., JEFFERSON, N.D., SPICER, E.J.F.,
ARCEO, R.J., & KAHN, A., III (1980a) RU 22974. Two year toxicity
and carcinogenicity study in mice, Mattawan, Michigan,
International Research and Development Corporation (Unpublished
report IRDC 406-001/A-4, submitted to WHO by Roussel Uclaf).
GOLDENTHAL, E.I., JEFFERSON, N.D., BLAIR, M., THORSTENSON, J.H.,
SPICER, E.J.F., ARCEO, R.J., & KAHN, A., III (1980b) RU 22974.
Two year oral toxicity and carcinogenicity study in rats,
Mattawan, Michigan, International Research and Development
Corporation (Unpublished report IRDC 406-002/A.4, submitted to WHO
by Roussel Uclaf).
GRANDADAM, A. (1976) Test to determine the toxicity in the
chicken by oral route (Unpublished report RU76.05.05/A, submitted
to WHO by Roussel Uclaf).
GRAY, A.J. & RICKARD, J. (1982) The toxicokinetics of
deltamethrin in rats after intravenous administration of a toxic
dose. Pestic. Biochem. Physiol.., 18: 205-215.
GRAY, A.J., CONNORS, T.A., & RICKARD, J. (1980) Mechanism of
mammalian toxicity of the pyrethroids. In: Littaner, U.Z. et al.,
ed. Neurotransmitters and their receptors, New York, John Wiley
and Sons, pp. 565-568.
GROLLEAU G. & GRIBAN, J. (1976a) Toxicity of deltamethrin or
DECIS in single ingestion in game duck Anas platyrhnchos L.,
JoyenJossas, France, Institut National de la Recherche Agronomique
(Unpublished report INRA-76.21 12/A, submitted to WHO by Roussel
Uclaf).
GROLLEAU, G. & GRIBAN, J. (1976b) Toxicity of deltamethrin or
DECIS by single ingestion in grey partridge, Perdix Perdix L. and
red partridge, Alectotis rufa L., JoyenJossas, France, Institut
National de la Recherche Agronomique (Unpublished report INRA76.28
09/A, submitted to WHO by Roussel Uclaf).
GUILLOT, J.P. & GUILAINE, J. (1977) RU 22974. Decaméthrine.
Decis technical Roussel Uclaf. Sensitization test in the guinea
pig. Joinville-le-Pont, Institut Français de Recherches et Essais
Biologique (Unpublished report no. IFREB-R 709241/A, submitted to
WHO by Roussel Uclaf).
GULYAS P. & CSANYI B. (undated) Acute fish toxicological
investigation of diverse insecticides, Hungary, Scientific Research
Centre of Economy of Water Supplies, 21 pp. (Unpublished
proprietary data supplied to WHO by Roussel Uclaf).
HALLS, G.R.H. & PERIAM, A.W. (1980) The fate of residues of NRDC
161 on wheat during storage and after milling and baking - Report
after 9 months storage, November 1980, Wellcome Research
Laboratories Report (Unpublished report HEFH 80-4, submitted to
WHO by Roussel Uclaf).
HARGREAVES, J.R. & COOPER, L.P. (1979) Phytotoxicity tests with
pyrethroid insecticides on glasshouse grown tomato seedlings.
Queensland J. Agric. anim. Sci., 36: 151-154.
HASCOET, M. (1977) Laboratory leaching soil study with
decamethrin (RU 22974), Versailles, Institut National de la
Recherche Agronomique (Unpublished proprietary report INRA-
77.20.09/A, submitted to WHO by Roussel Uclaf).
HE, F. (1987) Occupational neurotoxicology: Current problems and
trends. Paper presented at the XXII International Congress on
Occupational Health, Sydney, 27 September-2 October, 1987.
HE, F., WANG, S., LIU, L., CHEN, S., ZHANG, Z., & SUN, J. (1989)
Clinical manifestations and diagnosis of acute pyrethroid
poisoning. Arch. Toxicol., 63: 54-58.
HEHEITMULLER, T., SHUBA, P.J., & PARRISH, R. (1978) Acute
toxicity of Decis to sheepshead minnows (Cyprinodon variegatus),
E.G. & G. Bionomics Marine Laboratory, 7 pp. (Unpublished
proprietary report BR-78-12219, supplied to WHO by Roussel Uclaf).
HEWSON, R.T. & BURGESS, J.E. (1981) An operator study with
deltamethrin including measurements of nerve conduction times
(Unpublished proprietary report GB-NT-11.81A, submitted to WHO by
Roussel Uclaf).
HILL, B.D. (1982) Determination of Deltamethrin residues in
soils, Alberta, Agriculture Canada Research Station, 7 pp
(Unpublished proprietary report CAN-82.12.10/A, submitted to WHO by
Roussel Uclaf).
HILL, B.D. (1983) Persistence of deltamethrin in a Lethbridge
sandy clay loam. J. environ. Sci. Health, B18(6): 691-703.
HILL, B.D. & JOHNSON, D.L. (1987) Persistence of deltamethrin and
its isomers on pasture forage and litter. J. agric. Food Chem.,
35: 373-378.
HILL, B.D. & SCHAALJE, G.B. (1985) A two-compartment model for
the dissipation of deltamethrin on soil. J. agric. food Chem., 33:
1001-1006.
HIROMORI, T., NAKANISHI, T., KAWAGUCHI, S., SAKO, H., SUZUKI, T., &
KIYAMOTO, J. (1986) Therapeutic effects of methocarbamol on acute
intoxication by pyrethroids in rats. J. pestic. Sci., 11: 9-14.
HUNTER, B., JORDAN, J., HEYWOOD, R., HEPWORTH, P., STREET, A.E., &
PRENTICE, D.E. (1977) RU 22974. Assessment of toxicity to rats
by oral administration for 13 weeks (followed by a 4-week
withdrawal period) (Unpublished report RSL-254/76938/A3, submitted
to WHO by Roussel Uclaf).
HUSSON, J.M. (1978) Medical observations made on people working
on the manufacture and formulation of the pyrethroid insecticide
decamethrin (Unpublished report RU 78.25.08/A, submitted to WHO by
Roussel Uclaf).
IRDC (1980) Two year chronic dog feeding study, Matawan,
Michigan, International Research and Development Corporation
(Unpublished proprietary report IRDC-406-004/AI, submitted to WHO
by Roussel Uclaf).
JACKSON, G. & HARDY, C. (1986) RU 22974. Acute inhalation
toxicity in rats 1-hour exposure (Unpublished report RSL-DTM-
851457/A, submitted to WHO by Roussel Uclaf).
JACQUES, Y., ROMEY, G., CAVEY, M.T., KARTAALOVSKI, B., & LAZDUNSKI,
M. (1980) Interaction of pyrethroids with the sodium channel in
mammalian neuronal cells in culture. Biochem. Biophys. Acta, 600:
882-897.
KAGAN, YU. S., PANSHINA, T.N., & SASINOVICH, L.M. (1986)
Biochemical effects of toxic action of synthetic pyrethroids. Gig.
i Sanit., 1: 7-9.
KAUFMAN, D.D. & KAYSER, A.J. (1979a) Degradation of 14C-phenoxy-
and 14C-cyano-decamethrin in soil (Unpublished proprietary report
USDA-I-23.04.79/A2, submitted to WHO by Roussel Uclaf).
KAUFMAN, D.D. & KAYSER, A.J. (1979b) The effect of soil
temperature on the degradation of 14C-cyano-decamethrin in soil
(Unpublished proprietary report USDA-II-24.04.79/A2, submitted to
WHO by Roussel Uclaf).
KAUFMAN, D.D. & KAYSER, A.J. (1980) Degradation of 14C-cyano-,
14C-phenoxy-, and 14C-vinyl-decamethrin in flooded soil
(Unpublished proprietary report USDA-III-12.05.80/A, submitted to
WHO by Roussel Uclaf).
KAUFMAN, D.D., RUSSEL, B.A., HELLING, C.S., & KAYSER, A.J. (1981)
Movement of cypermethrin, decamethrin, permethrin and their
degradation products in soil. J. agric. food Chem., 29: 239-245.
KAVLOCK, R., CHERNOFF, N., BARON, R., LINDER, R., ROGERS, E.,
CARVER, B., DILLEY, J., & SIMMON, V. (1979) Toxicity studies with
decamethrin, a synthetic pyrethroid insecticide. J. environ.
Pathol. Toxicol., 2: 751-765.
KNAUF, W. & HORLEIN, G. (1979 The acute toxicity of decamethrine
to the Rainbow trout Salmo gairdneri, Richardson, Frankfurt,
Hoechst AG, 19 pp (Unpublished proprietary report 19/79, submitted
to WHO by Roussel Uclaf).
KNAUF, W. & SCHULZE, E.F. (1977a) Effect of decamethrine on
Cyprinus carpio, Frankfurt, Hoechst AG, 9 pp (Unpublished
proprietary report 15/77, submitted to WHO by Roussel Uclaf).
KNAUF, W. & SCHULZE, E.F. (1977b) The effect of decamethrin
6E0660 on Catfish (Ictalurus nebulosus) in a static test,
Frankfurt, Hoechst Ag, 18 pp (Unpublished proprietary report
32/79, submitted to WHO by Roussel Uclaf).
KRASNJIH, A. & PAVLOVA, L. (1985) Harmful effects of
deltamethrin. In: Levina, E., ed. Harmful substances in industry,
Moscow, Khimija Publishing House, p. 154.
KYNOCH, S.R., LLOYD, G.K., & ANDREWS, C.D. (1979) Acute
percutaneous toxicity to rats of decamethrin, Huntingdon,
Huntingdon Research Centre (Unpublished report RSL-1009 8/D,
submitted to WHO by Roussel Uclaf).
LAWRENCE, L.J. & CASIDA, J.E. (1982) Pyrethroid toxicology: Mouse
intracerebral structure-toxicity relationship. Pestic. Biochem.
Physiol., 18: 9-14.
LAWRENCE, L.J. & CASIDA, J.E. (1983) Stereospecific action of
pyrethroid insecticides on the gamma-aminobutyric acid receptor-
ionophore complex. Science, 221: 1399-1401.
LAWRENCE, L.J., GEE, K.W., & YAMAMURA, H.I. (1985) Interactions
of pyrethroid insecticides with chloride ionophore-associated
binding sites. Neurotoxicology, 6: 87-98.
LEAHEY, J.P. (1985) The pyrethroid insecticide, London, Taylor &
Francis Ltd., p. 440.
LECLERCQ, M., COTONAT, J., & FOULHOUX, P. (1986) Recherche d'un
antagoniste à l'intoxication par la deltaméthrine. J. Toxicol.
clin. exp., 6, 83-85.
LEIBOWITZ, M.D., SCHWARZ, J.R., HOLAN, G., & HILLE, B. (1987)
Electrophysiological comparison of insecticide and alkaloid
agonists of Na channels. J. gen. Physiol., 90(1): 75-93.
LEPAILLEUR, H. & CHAMBON, A. (1984) Etude de la toxicité létale
du produit technique deltaméthrine vis à vis du poisson-zebré, 5 pp
(Unpublished proprietary dated report IRCHA B.7827, submitted to
WHO by Roussel Uclaf).
LHOSTE, J., FRANCOIS, Y., & RUPAUD, Y. (1979) Ichtyotoxicité de
la decaméthrine vis à vis de Salmo trutta L. (poisson téléostéen)
en fonction de l'age et des conditions expérimentales - Congrès sur
la lutte contre les insectes en milieu tropical, Marseilles - March
13-16 1979, pp. 885-902.
L'HOTELLIER, M. & VINCENT, P. (1986) Assessment of the impact of
deltamethrin on aquatic species. In British Crop Protection
Conference - Pests and Disease: 1109-1116.
LOUVEAUX, J., MISSONNIER, J., & MESQUIDA, J. (1977) Tests de
toxicité sur abeille domestique avec le Decis CE (Unpublished
proprietary report submitted to WHO by Roussel Uclaf).
LUMMIS, S.C., CHOW, S.C., HOLAN, G., & JOHNSTON, G.A. (1987)
Gamma-aminobutyric acid receptor ionophore complexes: differential
effects of deltamethrin, dichlorodiphenyltrichloroethane, and some
novel insecticides in a rat brain membrane preparation. J.
Neurochem., 48(3): 689-94.
LUND, A.E. & NARAHASHI, T. (1983) Kinetics of sodium channel
modification as the basis for the variation in the nerve membrane
effects of pyrethroids and DDT analogs. Pestic. Biochem. Physiol.,
20: 203-216.
MACPHAIL, R.C. (1981) Behavioral effects of a synthetic
insecticide (decamethrin). Fed. Proc. Fed. Am. Soc. Exp. Biol.,
40: 678.
MEISTER, R.T., BERG, G.L., SINE, C., MEISTER, S., & POPLYK, J.
(1983) Farm chemicals handbook. Section C. Pesticide dictionary,
Willoughby, Ohio, Meister Publishing Co.
MESTRES, R., CHEVALLIER, C., ESPNOZA, C., & CORNET, R. (1978a)
Dosage des résidues de decaméthrine dans les produits végétaux.
Trav. Soc. Pharm. Montpellier, 38: 183-192.
MESTRES R., CHEVALLIER C., & ESPINOZA, C. (1978b) Analytical
method for decamethrine residue analysis, Montpellier, Faculte de
Pharmacie, 2 pp. (Proprietary report FP-78.08.07/A, dated 7/8/1978
submitted to WHO by Roussel Uclaf).
MESTRES, R., FRANCOIS, C., CAUSSE C., VIAN L., & WINNETT, G.
(1985) Survey of exposure to pesticides in greenhouses. Bull.
environ. Contam. Toxicol., 35: 750-756.
MESTRES, R., ESPINOZA, C., & CHEVALLIER, C. (1986) Effets sur les
résidus de deltaméthrine de la transformation des produits
agricoles en vue de leur consommation. Med. Nutr., XXII (3):
181-184.
MILLER, T.A. & ADAMS, M.E. (1980) Neural and behavioral
correlates of pyrethroid and DDT-type poisoning in the housefly,
Musca domestica. Pestic. Biochem. Physiol., 13: 137-147.
MIYAMOTO, J. (1976) Degradation, metabolism and toxicity of
synthetic pyrethroids. Environ. Health Perspect., 14: 15-28.
MIYAMOTO, J. (1981) The chemistry, metabolism and residue
analysis of synthetic pyrethroids. Pure appl. Chem., 53: 1967-2022.
MIYAMOTO, J. & KEARNEY, P.C. (1983) Pesticide chemistry - Human
welfare and the environment. Proceedings of the Fifth International
Congress of Pesticide Chemistry, Kyoto, Japan, 29 August - 4
September, 1982, Oxford, Pergamon Press, Vol. 1-4.
MOHSEN, Z.H. & MULLA, M.S. (1981) Toxicity of blackfly larvicidal
formulations to some aquatic insects in the laboratory. Bull.
environ. Contam. Toxicol., 26: 696-703.
MOUROT, D., DELEPINE, B., BOISSEAU, J., & GAYOT, G. (1979) High-
performance liquid chromatography of decamethrin. J. Chromatogr.,
173: 412-414.
MUIR, D.C.G., RAWN, G.P., & GRIFT, N.P., (1985) Fate of the
pyrethroid insecticide deltamethrin in small ponds: A mass balance
study. J. agric. food Chem., 33: 603-609.
MULLA, M.S., NAVVAB GOJRATI, H.A., & DARWAZEH, H.A. (1978)
Toxicity of mosquito larvicidal pyrethroids to four species of
freshwater fishes. Environ. Entomol.., 7(3): 428-430.
NETO, P.X.R., RODRIGUES, I.C.F., & FILHO, A.M (1983) Etude sur
les effets secondaires de la deltaméthrine sur l'ichtyofaune de la
region du projet "Rio Formoso" (Unpublished report submitted to WHO
by Roussel Uclaf).
NOBLE, R.M., HAMILTON, D.J., & OSBORNE, W.J. (1982) Stability of
pyrethroids on wheat in storage. Pestic. Sci., 13: 246-252.
PANSHINA T.N. & SASINOVICH, L.M. (1983) Toxicology of synthetic
pyrethroids. Khim. sel'sk. khoz., 12: 51-53.
PANSU, M., DHOUIBI, M.H., & PINTA, M. (1981) Determination of
traces of biopermethrine and decamethrine pyrethrinoids in
biological substrates by gas chromatography. Analysis, 9: 55-59.
PAPALEXIOU, Ph., BITAR, N., & STOCKIS, A. (1984) Absorption of
radiocarbon labelled deltamethrin given orally in healthy
volunteers, Charleroi, Belgium, Institut de Pharmacologie et
d'Investigations Biomédicales (Unpublished proprietary report
50/22, submitted to WHO by Roussel Uclaf).
PARKIN, P.J. & LEQUESNE, P.M. (1982) Effects of a synthetic
pyrethroid deltamethrin on excitability changes following a nerve
impulse. J. Neurol. Neurosurg. Psychiatry., 45: 337-342.
PEYRE, M., CHANTOT, J.F., GLOMOT, R., & PENASSE, L. (1980)
Detection of a mutagenic potency of decamethrin (RU 22974), in
bacterial tests (Unpublished report RU/TOX/80-2101/A, submitted to
WHO by Roussel Uclaf).
PHAM, H.C., NAVARRO-DALMASURE, C., PHAM, H.C., CLAVEL, P., VAN
HAVERBEKE, G., CHEAV, S.L. (1984) Toxicological studies of
deltamethrin. Int. J. Tissue React., 6(2): 127-33.
PICHON, Y. (1981) Pharmacological characterization of ionic
channels in unmyelinated axons. J. Physiol., 17(9): 1119-1128.
PLAPP, F.W., Jr & BULL, D.L. (1978) Toxicity and selectivity of
some insecticides to Chrysopa carnea, a predator of the tobacco
budworm. Environ. Entomol., 7: 431-434.
PLESTINA, R. (1981) An evaluation of the use of deltamethrin in
public health (Unpublished report ZAG. KOT. 81/A, submitted to WHO
by Roussel Uclaf).
PLUIJMEN, M., DREVON, C., MONTESANO, R., MALAVEILLE C.,
HAUTEFEUILLE, A., & BARTSCH, H. (1984) Lack of mutagenicity of
synthetic pyrethroids in Salmonella typhimurium strains and in V79
Chinese hamster cells. Mutat. Res., 137: 7-15.
POLAKOVA, H. & VARGOVA, M. (1983) Evaluation of the mutagenic
effects of decamethrin: cytogenetic analysis of bone marrow. Mutat.
Res., 120: 167-171.
POTTIER J., CHATELET, P., & JOUQUEY, S. (1982) Resorption
percutanée de la deltaméthrine - Etude d'un modèle animal, 10 pp.
(Unpublished proprietary report AN.34, submitted to WHO by Roussel
Uclaf).
PRASADA RAO, K.S., CHETTY, C.S., & DESAIAH, D. (1984) In vitro
effects of pyrethroids on rat brain and liver ATPase activities. J
Toxicol. environ. Health, 14(2-3): 257-65.
RAWN, G.P., MUIR, D.O.G., & GRIFT, P.G. (1985) Fate of the
pyrethroid insecticide deltamethrin in small ponds, a mass balance
study. J. agr. food Chem., 33: 603-609.
RAY, D.E. (1980) An EEG investigation of decamethrin-induced
choreathetosis in the rat. Exp. Brain Res., 38: 221-227.
RAY, D.E. (1982) Changes in brain blood flow associated with
deltamethrin-induced choreoathetosis in the rat. Exp. Brain Res.,
45: 269-276.
RAY, D.E. & CREMER, J.E. (1979) The action of decamethrin (a
synthetic pyrethroid) on the rat. Pestic. Biochem. Physiol., 10:
333-340.
RICHTER, W.R. & GOLDENTHAL, E.I. (1983) Two-year oral toxicity
and carcinogenicity study in rats. Amendment to the final report.
Matawan, Michinga, International Research and Development
Corporation (Unpublished proprietary report IRDC 406.002/A5,
submitted to WHO by Roussel Uclaf).
RICKARD, J. & BRODIE, M.E. (1985) Correlation of blood and brain
levels of the neurotoxic pyrethroid deltamethrin with the onset of
symptoms in rats. Pestic. Biochem. Physiol., 23: 143-156.
RICOU (1978) Assessment of side effects of S 276-B (Decis EC
25 g/l) towards grey slugs (Agriolimax spp.) (Unpublished
proprietary report INRA-LIM.78.10/A, submitted to WHO by Roussel
Uclaf).
ROSE, G.P. & DEWAR, A.J. (1983) Intoxication with four synthetic
pyrethroids fail to show any correlation between neuromuscular
dysfunction and neurobiochemical abnormalities in rats. Arch.
Toxicol., 53: 297-316.
ROSE, D.B., ROBERTS, N.L., CAMERON, M.McD., PRENTICE, D.E., COOKE,
L., & GIBSON, W.A. (1978) RU 22974. (Decamethrine) LD50
determination and assessment of neurotoxicity in the domestic hen,
Huntingdon, Huntingdon Research Centre (Unpublished report RSL 293-
NT/7830/A, submitted to WHO by Roussel Uclaf).
ROUSSELIN, X. (1983) Toxicité des dérivés du pyrèthre, Paris,
Faculté de Médecine Lariboisiere-Saint-Louis (Thèse de l'Université
Paris VII).
RUIGT, G.S.F. & VAN DEN BERCKEN, J. (1986) Action of pyrethroids
on a nerve-muscle preparation of the clawed frog, Xenopus laevis.
Pestic. Biochem. Physiol., 25: 176-187.
RUZO, L.O. & CASIDA, J.E. (1979) Degradation of decamethrin on
cotton plants. J. agric. food Chem., 27: 572-575.
RUZO, L.O., HOLMSTEAD, R.L., & CASIDA, J.E. (1976) Solution
photochemistry of the potent pyrethroid insecticide alpha-cyano-3-
phenoxy-benzyl cis-2,2-dimethyl-3-(2,2-dibromovinyl)
cyclopropanecarboxylate. Tetrahedron Lett., 3045-3048.
RUZO, L.O., HOLMSTEAD, R.L., & CASIDA, J.E. (1977) Pyrethroid
photochemistry: decamethrin. J. agric. food Chem., 25: 1385-1394.
RUZO, L.O., UNAI, T., & CASIDA, J.E. (1978) Decamethrin metabolism
in rats. J. agric. food Chem., 26: 918-925.
RUZO, L.O., ENGEL, J.L., & CASIDA, J.E. (1979) Decamethrin
metabolites from oxidative, hydrolytic and conjugative reactions in
mice. J. agric. food Chem., 27: 725-731.
SANTOSA, K. (1983) Toxicity of Decis 2.5 EC (Decamethrin) to
fish, Jakarta, Department of Agriculture Research and Development
Inland Fisheries Institute, 4 pp. (Unpublished proprietary data
submitted to WHO by Roussel Uclaf).
SANTOSA, K. & HADI, H. (1980) Trial results pesticide lethal
toxicity to fish, Department of Agriculture Research and
Development Inland Fisheries Institute, Jakarta, Indonesia -
Report no. 174/UI/80 dated 5 December 1980, 3 pp. (Unpublished
proprietary data submitted to WHO by Roussel Uclaf).
SMIES, M., EVERS, R.H.J., REIJNENBURG, P.H.M., & KOEMAN, J.H.
(1980) Environmental aspects of field trials with pyrethroids to
eradicate tsetse fly in Nigeria. Ecotoxicol. environ. Saf., 4:
114-128.
SOBELS, F.H., TATES, A.D., & VANNIER, B. (1978) Cytogenetic study
with RU 22974. Detection of a mutagenic potency in mammalian
cells, Leiden, The Netherlands, State University of Leiden
(Unpublished report ULN-782211/A, submitted to WHO by Roussel
Uclaf).
SODERLUND, D.M. & CASIDA, J.E. (1977) Effects of pyrethroid
structure on rates of hydrolysis and oxidation by mouse liver
microsomal enzymes. Pestic. Biochem. Physiol., 7: 391-401.
SOUYRI, F. (1985) Autoradiographic studies of (3H)-deltamethrin
binding with nerve tissue cultures. Pestic. Sci., 6(6): 701-703.
STAATZ, C.G., BLOOM, A.S., & LECH, J.J. (1982) Effects of
pyrethroids on [3H]-kainic acid binding to mouse forebrain
membranes. Toxicol. appl. Pharmacol., 64: 566-569.
STAATZ-BENSON, C.G. & HOSKO, M.J. (1986) Interaction of
pyrethroids with mammalian spinal neurons. Pestic. Biochem.
Physiol., 25: 19-30.
STEIN, E.A., WASHBURN, M., WALCZAK, C., & BLOOM, A.S. (1987)
Effects of pyrethroid insecticides on operant responding maintained
by food. Neurotoxicol. Teratol., 9(1): 27-31.
STEVENSON, J.H., NEEDHAM, P.H., & WALKER, J. (1978) Poisoning of
honey bee by pesticides: investigations of the changing pattern in
Britain over 20 years. Rothamsted Exp. Stn Rep., Part 2: 55-72.
TAKAHASHI, M. & LEQUESNE, P. ((1982) The effects of the
pyrethroids deltamethrin and cismethrin on nerve excitability in
rats. J. Neurol. Neurosurg. Psychiatry, 45: 1005-1011.
TAKKEN, W., BALK, F., JANSEN, R.C., & KOEMAN, J.H. (1978) The
experimental application of insecticides from a helicopter for the
control of riverine populations of Glossina tachinoides in West
Africa. VI. Observations on side effects. P.A.N.S., 24: 455-466.
THIEBAULT, J.J., BOST, J., & FOULHOUX, P. (1985) Intoxication
expérimentale par la deltaméthrine chez le chien et son traitement.
Toxicol. vét. Collect. Méd. lég. Toxicol. méd., 131, 47-62.
THIEBAULT, J.J., BOST, J., FOULHOUX, P., DEVAUX, P., & TILLIER, C.
(in press) Intravenous deltamethrin intoxication of dogs.
Therapeutic attempts. Veterinary and Human Toxicology.
THIER, W. & SCHMIDT, D. (1976) Laboratory leaching studies of
Decis EC 25 in three different German soils (Unpublished
proprietary report RL-77.25.05/A, submitted to WHO by Roussel
Uclaf).
THIER, W. & SCHMIDT, D. (1977) The behaviour of the pesticide in
the soil: Decamethrin, (Unpublished proprietary report A09232,
submitted to WHO by Roussel Uclaf) (in German).
TONG YING (1988) Clinical manifestations of 211 cases intoxicated
by deltamethrin and fenvalerate (Unpublished report submitted to
WHO by Roussel Uclaf).
TOOBY, T.E., THOMSON, A.N., RYCROFT, R.J., BLACK, I.A., & HEWSON,
R.T. (1981) A pond study to investigate the effects on fish and
aquatic invertebrates of deltamethrin applied directly onto water,
Fisheries Laboratory, Ministry of Agriculture, Fisheries and Food
(Unpublished report AEP-81.30.09A, submitted to WHO by Roussel
Uclaf).
TRIVEDI, K. (1981) Spray worker exposure and health survey during
decamethrin (decis) spray in the field, Sevagram, India, Mahatma
Gandhi Institute of Medical Sciences (Unpublished information
submitted to WHO by Roussel Uclaf).
TU, C.M. (1980) Influence of five pyrethroid insecticides on
microbial populations and activities in soil. Microb. Ecol., 5:
321-327.
VAN DEN BERCKEN, J. (l977) The action of allethrin on the
peripheral nervous system of the frog. Pestic. Sci., 8: 692-699.
VAN DEN BERCKEN, J. & VIJVERBERG, H.P.M. (1980) Voltage clamp
studies on the effects of allethrin and DDT on the sodium channels
in frog myelinated nerve membrane. In: Insect neurobiology and
pesticide action (Neutox 79), London, Society of Chemical Industry,
Vol. 2, pp. 79-85.
VAN DEN BERCKEN, J., AKKERMANS, L.M.A., & VAN DER ZALM, J.M.
(1973) DDT-like action of allethrin in the sensory nervous system
of Xenopus laevis. Eur. J. Pharmacol., 21: 95-106.
VAN DEN BERCKEN, J., KROESE, A.B.A., & AKKERMANS, L.M.A. (1979)
Effect of insecticides on the sensory nervous system. In:
Narahashi, T., ed. Neurotoxicology of insecticides and pheromones,
New York, London, Plenum Press, pp. 183-210.
VANNIER, B. & FOURNEX, R. (1983) Deltamethrin detection of a
mutagenic potency/micronucleus test in the mouse (Proprietary
unpublished report RU-83607/A, submitted to WHO by Roussel Uclaf).
VANNIER, B. & GLOMOT, R. (1977) RU 22974. Mutagenic study.
Dominant lethal assay in the male mouse (Unpublished report TOX
76533/DB9/A, submitted to WHO by Roussel Uclaf).
VANNIER, B. & GLOMOT, R. (1982) RU 22974 complementary
teratological study in the mouse (Unpublished report RU-82506-12/A,
submitted to WHO by Roussel Uclaf).
VARANKA, I. (1987) Effect of mosquito killer insecticides on
freshwater mussels. Comp. Biochem. Physiol. C. Comp. Pharmacol.,
86: 157-162.
VAYSSE, M., GIUDICELLI, J.C., DEVAUX, P., & L'HOTELLIER, M. (1984)
Decis, in Zweig analytical methods for pesticides and plant growth
regulators. Vol XIII, 3: 53-68, New York, Academic Press.
VERSCHOYLE, R.D. & ALDRIDGE, W.N. (1980) Structure-activity
relationship of some pyrethroids in rats. Arch. Toxicol., 45:
325-329.
VICKERMAN, G.P., COOMBES D.S., TURNER, G., MEAD-BRIGGS, M.A., &
EDWARDS, J. (1987a) The effects of pirimicarb, dimethoate and
deltamethrin on carabidae and staphylinidae in winter wheat. Meded.
Fac. Landbouwwet. Rijksuniv. Gent, 52(2a):
VICKERMAN, G.P., COOMBES, D.S., TURNER, G., MEAD-BRIGGS, M.A., &
EDWARDS, J. (1987b) The effects of pirimicarb, dimethoate and
deltamethrin on non-target arthropods in winter wheat.
International Conference on Pests in Agriculture, Paris, 1-3
December 1987, Annales ANPP, pp. 67-74.
VIJVERBERG, H.P.M., VAN DEN BECKEN, J. (1982) Action of
pyrethroid insecticides on the vertebrate nervous system.
Neuropathol. appl. Neurobiol., 8: 421-440.
VIJVERBERG, H.P.M. & VAN DEN BERCKEN, J. (1979) Frequency
dependent effects of the pyrethroid insecticide decamethrin in frog
myelinated nerve fibres. Eur. J. Pharmacol., 58: 501-504.
VIJVERBERG, H.P.M., RUIGT, G.S.F., & VAN DEN BERCKEN, J. (1982a)
Structure-related effects of pyrethroid insecticides on the
lateral-line sense organ and on peripheral nerves of the clawed
frog, Xenopus laevis. Pestic. Biochem. Physiol., 18: 315-324.
VIJVERBERG, H.P.M., VAN DER ZALM, J.M., & VAN DEN BERCKEN, J.
(1982b) Similar mode of action of pyrethroids and DDT on sodium
channel gating in myelinated nerves. Nature, (Lond.) 295: 601-603.
VIJVERBERG, H.P.M., VAN DER ZALM, J.M., VAN KLEEF, R.G.D.M., & VAN
DEN BERCKEN, J. (1983) Temperature and structure-dependent
interaction of pyrethroids with the sodium channels in frog node of
Ranvier. Biochim. Biophys. Acta, 728: 73-82.
WALTERSDORFER & SCHULZE E.F. (1976a) Decis 2.5. Effect on
Lebistes reticulatus, Francfort, Hoechst Ag, 8 pp. (Unpublished
proprietary report 42/76, submitted to WHO by Roussel Uclaf).
WALTERSDORFER & SCHULZE E.F. (1976b) Decis 2.5. Effect on Idus
melanotus, Frankfurt, Hoechst AG, 5 pp (Unpublished proprietary
report 33/76, submitted to WHO by Roussel Uclaf).
WALTERSDORFER & SCHULZE E.F. (1976c) Decis 2.5. Effect on Salmo
gairdneri (rainbow trout), Frankfurt, Hoechst AG, 7 pp (Unpublished
proprietary report 35/76, submitted to WHO by Roussel Uclaf).
WALTERSDORFER & SCHULZE E.F. (1976d) Decamethrine effect on
Lepomis gibbosus (Bluegill sunfish), Frankfurt, Hoechst AG, 6 pp.
(Unpublished proprietary report 16/77, submitted to WHO by Roussel
Uclaf).
WANG, S., ZHENG, Q., YU, L., XU, B., & LIX., Y. (1988) Health
survey among farmers exposed to deltamethrin in the cotton fields.
Ecotoxicol. environ. Saf., 15: 1-6.
WELLCOME FOUNDATION (1979) Residues in cow's milk resulting from
intrarumenal, and later, dermal administration of 14C-labelled
decamethrin (Unpublished report submitted to WHO by Roussel Uclaf).
WHO (1979) Safe use of pesticides. Third Report of the WHO
Expert Committee on Vector Biology and Control, Geneva, World
Health Organization, pp. 18-23. (WHO Technical Report Series,
No. 634).
WHO (1988) The WHO recommended classification of pesticides by
hazard and guidelines to classification 1988-1989, Geneva, World
Health Organization (Unpublished document WHO/VBC/88.953).
WHO (1989) Health and Safety Guide No. 30: Deltamethrin, Geneva,
World Health Organization.
WHO (1984) Data sheet on pesticides, No. 50: Deltamethrin, Geneva,
World Health Organization, 9 pp (Unpublished document
VBC/DS/84.50).
WOOD MACKENZIE (1980) Agrochem. Monit., 9: 12.
WOOD MACKENZIE (1981) Agrochem. Monit., 15: 12.
WOOD MACKENZIE (1982) Agrochem. Monit., 21: 13.
WOOD MACKENZIE (1983) Agrochem. Monit., 27: 3-12.
WORTHING, C.R. & WALKER, S.B. (1983) Pesticide manual, 7th ed.,
Croydon, British Crop Protection Council, p. 161.
WOUTERS, W. & VAN DEN BERCKEN, J. (1978) Action of pyrethroids.
Gen. Pharmacol., 9: 387-398.
WRENN, J.M., RODWELL, D.E., GOLDENTHAL, E.I., SPIDER, E.J.C., &
RAJASEKARAN, D. (1980) Three-generation reproduction study in
rats, Matawan, Michigan, International Research and Development
Corporation (Unpublished report IDC-406 003/A4. submitted to WHO by
Roussel Uclaf).
ZHANG, L.Z., KHAN, S.U., AKHTAR, M.H., & IVARSON, K.C. (1984)
Persistence, degradation, and distribution of Deltamethrin in an
organic soil under laboratory conditions, J. agric. food Chem., 32:
1207-1211
ZITKO, V., MCLEESE, D.W., METCALFE, C.D., & CARSON, W.G. (1979)
Toxicity of permethrin, decamethrin, and related pyrethroids to
salmon and lobster. Bull. environ. Contam. Toxicol., 21: 338-343.
APPENDIX I
On the basis of electrophysiological studies with peripheral
nerve preparations of frogs (Xenopus laevis; Rana temporaria; and
Rana esculenta), it is possible to distinguish between 2 classes of
pyrethroid insecticides: (Type I and Type II). A similar
distinction between these 2 classes of pyrethroids has been made on
the basis of the symptoms of toxicity in mammals and insects (Van
den Bercken et al., 1979; WHO, 1979; Verschoyle & Aldridge, 1980;
Glickman & Casida, 1982; Lawrence & Casida, 1982). The same
distinction was found in studies on cockroaches (Gammon et al.,
1981).
Based on the binding assay on the gamma-aminobutyric acid
(GABA) receptor-ionophore complex, synthetic pyrethroids can also
be classified into two types: the alpha-cyano-3-phenoxybenzyl
pyrethroids and the non-cyano pyrethroids (Gammon et al., 1982;
Gammon & Casida, 1983; Lawrence & Casida, 1983; Lawrence et al.,
1985).
Pyrethroids that do not contain an alpha-cyano group
(allethrin, d-phenothrin, permethrin, tetramethrin, cismethrin, and
bioresmethrin) (Type I: T-syndrome)
The pyrethroids that do not contain an alpha-cyano group give
rise to pronounced repetitive activity in sense organs and in
sensory nerve fibres (Van den Bercken et al., 1973). At room
temperature, this repetitive activity usually consists of trains of
3 - 10 impulses and occasionally up to 25 impulses. Train duration
is between 10 and 5 milliseconds.
These compounds also induce pronounced repetitive firing of the
presynaptic motor nerve terminal in the neuromuscular junction (Van
den Bercken, 1977). There was no significant effect of the
insecticide on neurotransmitter release or on the sensitivity of
the subsynaptic membrane, nor on the muscle fibre membrane.
Presynaptic repetitive firing was also observed in the sympathetic
ganglion treated with these pyrethroids.
In the lateral-line sense organ and in the motor nerve
terminal, but not in the cutaneous touch receptor or in sensory
nerve fibres, the pyrethroid-induced repetitive activity increases
dramatically as the temperature is lowered, and a decrease of 5 °C
in temperature may cause a more than 3-fold increase in the number
of repetitive impulses per train. This effect is easily reversed
by raising the temperature. The origin of this "negative
temperature coefficient" is not clear (Vijverberg et al., 1983).
Synthetic pyrethroids act directly on the axon through
interference with the sodium channel gating mechanism that
underlies the generation and conduction of each nerve impulse. The
transitional state of the sodium channel is controlled by 2
separately acting gating mechanisms, referred to as the activation
gate and the inactivation gate. Since pyrethroids only appear to
affect the sodium current during depolarization, the rapid opening
of the activation gate and the slow closing of the inactivation
gate proceed normally. However, once the sodium channel is open,
the activation gate is restrained in the open position by the
pyrethroid molecule. While all pyrethroids have essentially the
same basic mechanism of action, however, the rate of relaxation
differs substantially for the various pyrethroids (Flannigan &
Tucker, 1985).
In the isolated node of Ranvier, allethrin causes prolongation
of the transient increase in sodium permeability of the nerve
membrane during excitation (Van den Bercken & Vijverberg, 1980).
Evidence so far available indicates that allethrin selectively
slows down the closing of the activation gate of a fraction of the
sodium channels that open during depolarization of the membrane.
The time constant of closing of the activation gate in the
allethrin-affected channels is about 100 milliseconds compared with
less than 100 microseconds in the normal sodium channel, i.e., it
is slowed down by a factor of more than 100. This results in a
marked prolongation of the sodium current across the nerve membrane
during excitation, and this prolonged sodium current is directly
responsible for the repetitive activity induced by allethrin
(Vijverberg et al., 1983).
The effects of cismethrin on synaptic transmission in the frog
neuromuscular junction, as reported by Evans (1976), are almost
identical to those of allethrin, i.e., presynaptic repetitive
firing, and no significant effects on transmitter release or on the
subsynaptic membrane.
Interestingly, the action of these pyrethroids closely
resembles that of the insecticide DDT in the peripheral nervous
system of the frog. DDT also causes pronounced repetitive activity
in sense organs, in sensory nerve fibres, and in motor nerve
terminals, due to a prolongation of the transient increase in
sodium permeability of the nerve membrane during excitation.
Recently was demonstrated that allethrin and DDT have essentially
the same effect on sodium channels in frog myelinated nerve
membrane. Both compounds slow down the rate of closing of a
fraction of the sodium channels that open on depolarization of the
membrane (Van den Bercken et al., 1973, 1979; Vijverberg et al.,
1982b).
In the electrophysiological experiments using giant axons of
crayfish, the type I pyrethroids and DDT analogues retain sodium
channels in a modified open state only intermittantly, cause large
depolarizing afterpotentials, and evoke repetitive firing with
minimal effect on the resting potential (Lund & Narahashi, 1983).
These results strongly suggest that permethrin and cismethrin,
like allethrin, primarily affect the sodium channels in the nerve
membrane and cause a prolongation of the transient increase in
sodium permeability of the membrane during excitation.
The effects of pyrethroids on end-plate and muscle action
potentials were studied in the pectoralis nerve-muscle preparation
of the clawed frog (Xenopus laevis). Type I pyrethroids
(allethrin, cismethrin, bioresmethrin, and 1R, cis-phenothrin)
caused moderate presynaptic repetitive activity, resulting in the
occurrence of multiple end-plate potentials (Ruigt & Van den
Bercken, 1986).
Pyrethroids with an alpha-cyano group on the 3-phenoxybenzyl
alcohol (deltamethrin, cypermethrin, fenvalerate, and fenpropanate)
(Type II: CS-syndrome)
The pyrethroids with an alpha-cyano group cause an intense
repetitive activity in the lateral line organ in the form of long-
lasting trains of impulses (Vijverberg et al., 1982a). Such a
train may last for up to 1 min and contains thousands of impulses.
The duration of the trains and the number of impulses per train
increase markedly on lowering the temperature. Cypermethrin does
not cause repetitive activity in myelinated nerve fibres. Instead,
this pyrethroid causes a frequency-dependent depression of the
nervous impulse, brought about by a progressive depolarization of
the nerve membrane as a result of the summation of depolarizing
after-potentials during train stimulation (Vijverberg & Van den
Bercken, 1979; Vijverberg et al., 1983).
In the isolated node of Ranvier, cypermethrin, like allethrin,
specifically affects the sodium channels of the nerve membrane and
causes a long-lasting prolongation of the transient increase in
sodium permeability during excitation, presumably by slowing down
the closing of the activation gate of the sodium channel
(Vijverberg & Van den Bercken, 1979; Vijverberg et al., 1983). The
time constant of closing of the activation gate in the
cypermethrin-affected channels is prolonged to more than 100
milliseconds. Apparently, the amplitude of the prolonged sodium
current after cypermethrin is too small to induce repetitive
activity in nerve fibres, but is sufficient to cause the long-
lasting repetitive firing in the lateral-line sense organ.
These results suggest that alpha-cyano pyrethroids primarily
affect the sodium channels in the nerve membrane and cause a long-
lasting prolongation of the transient increase in sodium
permeability of the membrane during excitation.
In the electrophysiological experiments using giant axons of
cray-fish, the Type II pyrethroids retain sodium channels in a
modified continuous open state persistently, depolarize the
membrane, and block the action potential without causing repetitive
firing (Lund & Narahashi, 1983).
Diazepam, which facilitates GABA reaction, delayed the onset of
action of deltamethrin and fenvalerate, but not permethrin and
allethrin, in both the mouse and cockroach. Possible mechanisms of
the Type II pyrethroid syndrome include action at the GABA receptor
complex or a closely linked class of neuroreceptor (Gammon et al.,
1982).
The Type II syndrome of intracerebrally administered pyrethroids
closely approximates that of the convulsant picrotoxin (PTX).
Deltamethrin inhibits the binding of [3H]-dihydropicrotoxin to rat
brain synaptic membranes, whereas the non-toxic R epimer of
deltamethrin is inactive. These findings suggest a possible
relation between the Type II pyrethroid action and the GABA receptor
complex. The stereospecific correlation between the toxicity of
Type II pyrethroids and their potency to inhibit the [35S]-TBPS
binding was established using a radioligand,
[35S]- t-butyl-bicyclophosphorothionate [35S]-TBPS. Studies
with 37 pyrethroids revealed an absolute correlation, without any
false positive or negative, between mouse intracerebral toxicity and
in vitro inhibition: all toxic cyano compounds including
deltamethrin, 1R, cis-cypermethrin, 1R, trans-cypermethrin, and
[2S, alpha S]-fenvalerate were inhibitors, but their non-toxic
stereoisomers were not; non-cyano pyrethroids were much less potent
or were inactive (Lawrence & Casida, 1983).
In the [35S]-TBPS and [3H]-Ro 5-4864 (a convulsant benzo-
diazepine radioligand) binding assay, the inhibitory potencies of
pyrethroids were closely related to their mammalian toxicities.
The most toxic pyrethroids of Type II were the most potent
inhibitors of [3H]-Ro 5-4864 specific binding to rat brain
membranes. The [3H]-dihydropicrotoxin and [35S]-TBPS binding
studies with pyrethroids strongly indicated that Type II effects of
pyrethroids are mediated, at least in part, through an interaction
with a GABA-regulated chloride ionophore-associated binding site.
Moreover, studies with [3H]-Ro 5-4864 support this hypothesis and,
in addition, indicate that the pyrethroid-binding site may be very
closely related to the convulsant benzodiazepine site of action
(Lawrence et al., 1985).
The Type II pyrethroids (deltamethrin, 1R, cis-cypermethrin and
[2S, alphaS]-fenvalerate) increased the input resistance of
crayfish claw opener muscle fibres bathed in GABA. In contrast,
two non-insecticidal stereoisomers and Type I pyrethroids
(permethrin, resmethrin, allethrin) were inactive. Therefore,
cyanophenoxybenzyl pyrethroids appear to act on the GABA receptor-
ionophore complex (Gammon & Casida, 1983).
The effects of pyrethroids on end-plate and muscle action
potentials were studied in the pectoralis nerve-muscle preparation
of the clawed frog (Xenopus laevis). Type II pyrethroids
(cypermethrin and deltamethrin) induced trains of repetitive muscle
action potentials without presynaptic repetitive activity.
However, an intermediate group of pyrethroids (1R-permethrin,
cyphenothrin, and fenvalerate) caused both types of effect. Thus,
in muscle or nerve membrane the pyrethroid induced repetitive
activities due to a prolongation of the sodium current. But no
clear distinction was observed between non-cyano and alpha-cyano
pyrethroids (Ruigt & Van den Bercken, 1986).
Appraisal
In summary, the results strongly suggest that the primary
target site of pyrethroid insecticides in the vertebrate nervous
system is the sodium channel in the nerve membrane. Pyrethroids
without an alpha-cyano group (allethrin, d-phenothrin, permethrin,
and cismethrin) cause a moderate prolongation of the transient
increase in sodium permeability of the nerve membrane during
excitation. This results in relatively short trains of repetitive
nerve impulses in sense organs, sensory (afferent) nerve fibres,
and, in effect, nerve terminals. On the other hand, the alpha-
cyano pyrethroids cause a long-lasting prolongation of the
transient increase in sodium permeability of the nerve membrane
during excitation. This results in long-lasting trains of
repetitive impulses in sense organs and a frequency-dependent
depression of the nerve impulse in nerve fibres. The difference in
effects between permethrin and cypermethrin, which have identical
molecular structures except for the presence of an alpha-cyano
group on the phenoxybenzyl alcohol, indicates that it is this
alpha-cyano group that is responsible for the long-lasting
prolongation of the sodium permeability.
Since the mechanisms responsible for nerve impulse generation
and conduction are basically the same throughout the entire nervous
system, pyrethroids may also induce repetitive activity in various
parts of the brain. The difference in symptoms of poisoning by
alpha-cyano pyrethroids, compared with the classical pyrethroids,
is not necessarily due to an exclusive central site of action.
It may be related to the long-lasting repetitive activity in
sense organs and possibly in other parts of the nervous system,
which, in a more advance state of poisoning, may be accompanied by
a frequency-dependent depression of the nervous impulse.
Pyrethroids also cause pronounced repetitive activity and a
prolongation of the transient increase in sodium permeability of
the nerve membrane in insects and other invertebrates. Available
information indicates that the sodium channel in the nerve membrane
is also the most important target site of pyrethroids in the
invertebrate nervous system (Wouters & Van den Bercken, 1978; WHO,
1979).
Because of the universal character of the processes underlying
nerve excitability, the action of pyrethroids should not be
considered restricted to particular animal species, or to a certain
region of the nervous system. Although it has been established
that sense organs and nerve endings are the most vulnerable to the
action of pyrethroids, the ultimate lesion that causes death will
depend on the animal species, environmental conditions, and on the
chemical structure and physical characteristics of the pyrethroid
molecule (Vijverberg & Van den Bercken, 1982).
1. RESUME ET EVALUATION, CONCLUSIONS ET RECOMMANDATIONS
1.1 Résumé et évaluation
1.1.1 Identité, propriétés physiques et chimiques, méthodes
d'analyse
La deltaméthrine a été synthétisée en 1974 et commercialisée
pour la première fois en 1977. Sur le plan chimique, c'est un
ester de l'analogue dibromé de l'acide chrysanthémique (acide
diméthyl-2,2(dimbromo-2,2 vinyl)-3 cyclopropanecarboxylique)
(Br2CA) et de l'alcol alpha-cyano-phénoxy-3 benzylique; plus
précisément, c'est l'isomère [1R, cis;alpha S] parmi les huit
stéréoisomères que compte cet ester.
La deltaméthrine de qualité technique se présente sous la forme
d'une poudre blanche inodore dont le point de fusion est de 98 -
101 °C et qui contient plus de 98% de deltaméthrine. Sa tension de
vapeur est de 2,0 x 10-6 Pa à 25 °C; autrement dit, elle n'est
pratiquement pas volatile. Insoluble dans l'eau, elle est en
revanche soluble dans des solvants organiques tels que l'acétone, la
cyclohexanone et le xylène. Elle est stable à la lumière, à la
chaleur et à l'air, mais instable en milieu alcalin.
Pour doser les résidus et analyser des échantillons prélevés
dans l'environnement, on procède à une extraction par solvant au
moyen d'un mélange n-hexane/acétone, à un partage entre le
n-hexane, l'acétone et l'eau, suivi d'une purification par
chromatographie sur colonne de gel de silice, le dosage final
s'effectuant par chromatographie en phase gazeuse avec détection
par capture d'électrons. La concentration minimale décelable par
cette méthode est de 0,01 mg/kg, voire moins. L'analyse des
produits s'effectue par chromatographie en phase liquide à haute
performance avec détecteur UV.
1.1.2 Production et usage
En 1987, la consommation mondiale de deltaméthrine était
d'environ 250 tonnes. On l'utilise essentiellement pour traiter le
coton (45% de la consommation) ainsi que pour les cultures telles
que le café, le maïs, les céréales, les fruits, les légumes, le
houblon et les produits stockés. On l'utilise aussi pour l'hygiène
des animaux, la lutte contre les vecteurs ainsi qu'à des fins de
santé publique. On l'applique soit seule, soit associée à d'autres
pesticides en formulations telles que concentrés émulsionnables,
concentrés pour pulvérisation à très bas volume, poudres
mouillables, concentrés pour suspension, poudres pour poudrage, etc.
1.1.3 Exposition humaine
L'exposition de la population dans son ensemble provient
principalement de la présence de résidus alimentaires mais peut
également résulter de son utilisation en santé publique. Sur les
récoltes correctement traitées, le taux de résidus est généralement
très faible sauf lorsque le traitement a eu lieu après la récolte.
La FAO et l'OMS ont passé en revue de nombreuses données sur ces
questions.
L'exposition de la population dans son ensemble est vrai-
semblablement très faible, mais on manque de données effectives
découlant d'études sur la ration alimentaire totale.
1.1.4 Exposition et destinée dans l'environnement
En exposant à la lumière solaire une fine pellicule de
deltaméthrine [1R, cis; alpha S] marquée au C14 au niveau de la
fonction acide, de la fonction alcool ou du groupe cyano, pendant 4
à 8 heures, on a constaté que 70 % du produit était transformé en
isomères [1R, trans alpha S] et [1S, trans alpha S] par
isomérisation cis/trans, accompagnés des produits de clivage de
l'ester, en particulier l'acide diméthyl-2,2(dibromovinyl-2,2)-3
cyclo-propane-carboxylique et l'alcool alpha-cyano-phénoxy-3
benzylique.
La deltaméthrine présente sur des plants de coton en serre se
dégrade avec une demi-vie initiale de 1,1 semaine, le taux de
dégradation étant de 90 % au bout de 4,5 semaines.
Les principaux métabolites consistent en Br2CA libre et
conjugué, en trans-hydroxyméthyl-Br2CA et en acide (hydroxy-4
phénoxy)-3 benzoïque résultant du clivage de l'ester, d'une
oxydation et d'une conjugaison.
Après avoir fait incuber de deltaméthrine dans du sable et du
terreau à 28 °C dans les conditions du laboratoire, on en a
récupéré respectivement 52 % et 74 %, huit semaines après ce
traitement.
La deltaméthrine ne se déplace pas dans l'environnement du fait
qu'elle est fortement adsorbée sur les particules, qu'elle est
insoluble dans l'eau et qu'elle est appliquée à très faibles doses.
On ne dispose pas de données sur les concentrations effectives
dans l'environnement, mais compte tenu des modalités actuelles
d'utilisation et pour peu que l'insecticide soit utilisé
normalement, l'exposition environnementale devrait être très
faible. La deltaméthrine se dégrade rapidement en produits moins
toxiques.
1.1.5 Absorption, métabolisme et excrétion
Administrée par voie orale, la deltraméthrine est facilement
absorbée; l'absorption est moindre par la voie percutanée encore
que sa vitesse dépende fortement du véhicule ou du solvant utilisé.
Une fois absorbée, la deltaméthrine est rapidement métabolisée et
excrétée.
On a administré à des rats par voie orale, à raison de 0,64 à
1,60 mg/kg, de la deltaméthrine marquée au carbone-14 au niveau des
fonctions acide, alcool et nitrile; le radiocarbone provenant de
la fraction acide et de la fraction alcoolique a été complètement
éliminé en l'espace de 2 à 4 jours. Les taux de résidus
tissulaires étaient généralement faibles sauf dans les graisses où
ils étaient un peu plus élevés. Cependant, le reste nitrile a été
excrété plus lentement, le taux global de récupération étant de
79 % en huit jours. Les principales réactions métaboliques étaient
l'oxydation (au niveau du méthyl- trans, du cycle cyclopropane et
des positions 2', 4' et 5' du reste alcoolique), le clivage de
l'ester et la conversion du nitrile en thiocyanate. Les acides
carboxyliques et les phénols résultants étaient conjugués à l'acide
sulfurique, à la glycine et à l'acide glucuronique.
Après avoir administré par voie orale à des souris de la
deltaméthrine marquée au 14C au niveau des fonctions acide, alcool
et nitrile à raison de 1,7 et 4,4 mg/kg, on a constaté que
l'excrétion du radiocarbone s'effectuait rapidement sauf dans le
cas du reste nitrile. Les principales réactions métaboliques
étaient en général analogues chez les souris et chez les rats.
Chez les vaches et la volaille, les voies de dégradation sont
tout à fait analogues à celles des rongeurs.
1.1.6 Effets sur les êtres vivants dans leur milieu naturel
La deltaméthrine est extrêmement toxique pour les poissons, la
CL50 à 96 heures allant de 0,4 à 2,0 µg/litre. Elle est également
très toxique pour les invertébrés aquatiques; pour la daphnie, la
CL50 à 48 heures est de 5 µg/litre. Toutefois des études
approfondies sur des étangs expérimentaux ainsi que les résultats
de l'utilisation en plein champ ont montré que cette forte toxicité
potentielle était inopérante. On a observé sur le terrain une
certaine mortalité parmi les invertébrés aquatiques mais les
populations se reconstituent généralement assez vite.
pour les oiseaux, la toxicité de la deltaméthrine est très
faible, les valeurs de la DL50 pour une dose unique par voie orale
étant supérieure à 1000 mg/kg. Au laboratoire, la deltaméthrine
est très toxique pour les abeilles, avec une DL50 de contact de
0,051 µg/abeille. Les essais en situation réelle et l'expérience
acquise dans l'utilisation effective du produit ont montré que les
formulations de deltaméthrine exerçaient une action répulsive, ce
qui signifie qu'en pratique le danger pour les abeilles est très
réduit.
1.1.7 Effets sur les animaux d'expérience et systemes d'epreuve
in vitro
Dans un véhicule non aqueux, la deltaméthrine présente une
toxicité aiguë par voie orale forte à modérée avec des DL50 de 19 à
34mg/kg chez la souris et de 39 à 139 mg/kg chez le rat.
Toutefois, en suspension dans l'eau, la toxicité est bien moidre,
les valeurs de la DL50 dépassant 5000 mg/kg chez le rat. La
deltaméthrine est un pyréthroide du Type II. Les signes cliniques
d'intoxication consistent en tremblements, salivation et
convulsions. L'intoxication est rapide et chez les survivants, les
signes disparaissent en quelques jours. L'électro-encéphalogramme
présente des décharges pointes-ondes généralisées qui précèdent
la choréo-athétose.
Une seule application de deltaméthrine technique n'a pas
produit d'effets irritants sur la peau intacte ou abrasée du lapin.
Toutefois elle a produit de effets irritants passagers au niveau de
l'oeil avec ou sans rinçage. La deltaméthrine n'a pas d'effet
sensibilisateur cutané chez le cobaye.
En administrant à des rats par gavage de la deltaméthrine à des
doses quotidiennes allant jusqu'à 10 mg/kg de poids corporel
pendant 13 semaines, on a provoqué chez ces animaux une
hyperexcitabilité qui se manifestait au bout de six semaines chez
les mâles recevant la dose la plus forte. Aux doses de 2,5 et
10 mg/kg, le gain de poids a été plus faible chez les mâles.
Chez des chiens beagle qui avaient reçu par voie orale de la
deltaméthrine en doses quotidiennes allant jusqu'à 10 mg/kg de
poids corporel pendant 13 semaines, on a observé divers symptômes
liés à cette substance, tels que des vomissements, des
tremblements, de la salivation et un affaiblissement du réflexe
pharyngé, du réflexe rotulien et du réflexe de flexion. Lors d'une
étude d'alimentation de deux ans sur des chiens, on a constaté que
la dose sans effet observable se situait à 1 mg/kg de poids
corporel par jour (dose la plus forte expérimentée).
L'administration de deltraméthrine à des souris à des doses
atteingnant 100 mg/kg de nourriture pendant 24 mois n'a pas modifié
l'incidence des tumeurs. La dose sans effet observé relative à la
toxicité générale était de 100 mg/kg de nourriture.
Chez des rats à qui l'on avait administré de la deltaméthrine
en doses allant jusqu'à 50 mg/kg de nourriture pendant deux ans, on
n'a observé aucune tumeur attribuable à cette substance. La dose
sans effet observé relative à la toxicité générale était de 5 mg/kg
de nourriture.
La deltaméthrine ne s'est pas révélée mutagène dans divers
systèmes d'épreuve in vivo et in vitro; notamment la réparation de
l'ADN, la mutation génique, les aberrations chromosomiques,
l'échange entre chromatides-soeurs, la formation de micronoyaux et
la létalité dominante.
Des études de tératogénicité ont été effectuées chez des rates
et des souris gravides à qui l'on administrait par voie orale des
doses quotidiennes de deltaméthrine allant jusqu'à 1000 mg/kg
pendant la phase principale de l'organogénèse. On n'a constaté
aucun effet tératogène ni altération de la fonction de reproduction
chez ces rates et ces souris, si ce n'est une réduction, liée à la
dose, dans le poids moyen des foetus chez les souris et un léger
retard d'ossification chez les rats.
Des lapines gravides ont reçu au sixième et dix-neuvième jours
de leur grossesse des doses quotidiennes de deltaméthrine allant
jusqu'à 16 mg/kg. A la dose la plus forte, on a noté une réduction
du poids moyen des foetus. Aucun effet tératogène n'a été observé
chez les lapins.
Après administration de deltaméthrine à des rats à des doses
allant jusqu'à 5 mg/kg de nourriture, dans le but d'effectuer une
étude de reproduction sur trois générations et deux portées, on a
constaté l'absence totale de tout effet.
Il existe certains indices selon lesquels l'association de
delta- méthrine à certains composés organophosphorés pourrai
conduire à une potentialisation de la toxicité de ces produits.
1.1.8 Effets sur l'homme
La deltaméthrine peut provoquer certaines sensations cutanées
chez des travailleurs exposés. Plusieurs cas d'intoxication
professionnelle non mortelle ont été signalés qui étaient dus à
l'inobservation des mesures de sécurité. Un engourdissement, un
prurit, des fourmillements et une sensation de brûlure de la peau
ainsi que des vertiges sont des symptômes fréquemment signalés. On
a décrit occasionnellement un érythème papulaire ou une couperose à
caractère passager. La plupart de ces symptômes sont passagers et
disparaissent en cinq à sept jours. Aucun effet indésirable à long
terme n'a été signalé. On a décrit trois cas d'empoisonnement non
mortel par la deltamétrine à la suite de l'ingestion de plusieurs
grammes de ce produit.
1.2 Conclusions
Population générale: l'exposition de la population générale à la
deltaméthrine est vraisemblablement très faible et ce produit,
lorsqu'il est utilisé conformément aux recommandations, ne présente
probablement aucun risque.
Exposition professionnelle: moyennant de bonnes méthodes de
travail et l'application de mesures d'hygiène et de sécurité, la
deltaméthrine ne devrait pas présenter de danger pour les personnes
qui y sont exposées de par leur profession.
Environnement: Utilisée aux doses recommandées, il est improbable
que la deltaméthrine ou ses produits de dégradation s'accumulent au
point d'avoir des effets nocifs sur l'environnement. Au
laboratoire, la deltaméthrine est très toxique pour les poissons,
les arthropodes aquatiques et les abeilles. Toutefois, sur le
terrain, il est peu probable que le produit ait des effets nocifs
s'il est utilisé conformément aux recommandations.
1.3 Recommandations
Bien que les teneurs dans les aliments soient très faibles
lorsque la deltaméthrine est utilisée conformément aux
recommandations, il est souhaitable de s'en assurer en soumettant
la deltaméthrine à une surveillance.
La deltaméthrine est utilisée depuis de nombreuses années et
l'on signale un certain nombre de cas d'intoxications non mortelles
ainsi que des effets passagers par suite d'exposition
professionnelle. Il est souhaitable que l'exposition humaine
continue d'être surveillée.
1. RESUMEN Y EVALUACION, CONCLUSIONES Y RECOMENDACIONES
1.1 Resumen y evaluación
1.1.1 Identidad, propiedades fíisicas y químicas, métodos de
análisis
La deltametrina fue sintetizada en 1974 y se comercializó por
primera vez en 1977. Químicamente, es el isómero [1R, cis; S] de 8
ésteres estereoisoméricos del análogo dibromo del ácido
crisantémico, ácido 2,2-dimetil-3-(2,2-dibromovinil) ciclopro-
panocarboxílico (Br2CA) con alcohol alpha-ciano-3-fenoxibencílico.
La deltametrina de calidad técnica es un polvo blanco inodoro
con un punto de fusión de 98 - 101 °C y contiene más del 98% del
material. La presión del vapor es 2,0 x 10-6 Pa a 25 °C y es
prácticamente no volátil. Es insoluble en agua, pero soluble en
disolventes orgánicos como la acetona, la ciclohexanona y el
xileno. Es estable a la luz, el calor y el aire, pero inestable en
medios alcalinos.
La determinación de la presencia de residuos y el análisis de
muestras ambientales se efectuaron por extracción con los
disolventes n-hexano/acetona, partición con n-hexano/acetona/agua,
absorción con un cromatógrafo de columna de gel de sílice y
determinación con un cromatógrafo de fase gaseosa equipado con un
detector de captura de electrones con una concentración mínima
detectable de 0,01 mg/kg o menos. Para el análisis de productos se
utiliza la cromatografía de fase líquida de alto rendimiento con un
detector de rayos UV.
1.1.2 Producción y empleo
El consumo mundial de deltametrina era de 250 toneladas
aproximadamente en 1987. La deltametrina se utiliza sobre todo en
el algodón (45% del consumo), en cultivos como el café, el maíz,
los cereales, la fruta, las hortalizas y el lúpulo, y en productos
almacenados. Se emplea también en higiene animal, en la lucha
contra los vectores y en salud pública. Se fabrica como solución
concentrada emulsionable, solución concentrada de pequeñísimo
volumen, polvo humectable, solución concentrada en suspensión y
polvo para utilización en seco, sola o en combinación con otros
plaguicidas.
1.1.3 Exposición humana
La exposición de la población en general a la deltametrina
procede principalmente de residuos en la dieta, pero puede resultar
también de su empleo en salud pública. En los cultivos tratados de
acuerdo con buenas prácticas agrícolas, las concentraciones
residuales son por lo general muy bajas, excepto en caso de
tratamiento después de la recolección. La FAO y la OMS han
examinado abundantes datos al respecto.
Se cree que la exposición de la población en general es muy
baja, pero no se dispone de datos en forma de estudios de dietas
totales.
1.1.4 Exposición ambiental y evolución
Cuando la deltametrina-[1R, cis; alpha S] marcada con
C14-(compuesto irradiado ácido, alcohol o ciano) se expuso a la
luz del sol en forma de una película fina durante 4 a 8 horas, el
70% se transformó por isomerización- cis/trans produciendo los
isómeros [1R, trans; alpha S] y [1S, trans; alpha S], junto con
productos de la escisión de los ésteres, entre ellos Br2CA y
alcohol alpha-ciano-3-fenoxibencílico.
La deltametrina se degradó en las plantas de algodón, en
condiciones de invernadero, con una vida media inicial de 1,1
semanas, y el tiempo necesario para una pérdida del 90% fue de 4,6
semanas.
Los principales metabolitos fueron Br2CA libre y combinado,
trans-hidroximetil-Br2CA y ácido 3-(4-hidroxifenoxi)benzoico,
formados por la escisión, la oxidación y la combinación de los
ésteres.
La deltametrina se incubó en arena y mantillo orgánico a 28 °C
en condiciones de laboratorio. Ocho semanas después del
tratamiento, se recuperó de la arena y del mantillo orgánico
alrededor del 52% y el 74%, respectivamente, de la deltametrina
aplicada.
La deltametrina no es móvil en el medio debido a su fuerte
adsorción en las partículas, su insolubilidad en el agua y sus
tasas muy bajas de aplicación.
No se dispone de datos sobre las concentraciones existentes en
el medio pero, dadas las modalidades actuales de uso y en
condiciones normales, es de esperar que la exposición ambiental sea
muy baja. Se degrada rápidamente, convirtiéndose en productos
menos tóxicos.
1.1.5 Ingestion, metabolismo y excreción
La deltametrina se absorbe fácilmente por vía oral y con menor
facilidad por la piel, pero la tasa de absorción depende
considerablemente del portador o disolvente. La deltametrina
absorbida se metaboliza y se excreta fácilmente.
En la administración por vía oral a ratas de deltametrina
marcada con C14-(compuesto irradiado ácido, alcohol o ciano), a
tasas de 0,64 - 1,60 mg/kg, el radiocarbono de las partes ácido y
alcohol se eliminó casi por completo en 2 - 4 días. Las
concentraciones residuales en los tejidos fueron por lo general muy
bajas, excepto en la grasa, en donde fueron ligeramente más altas.
Sin embargo, la parte ciano se excretó más lentamente, con una
recuperación total del 79% en 8 días. Las principales reacciones
metabólicas fueron oxidación (en el trans-metilo del anillo de
ciclopropano y en las posiciones 2'-, 4'-, y 5 de la parte
alcohol), escisión de ésteres y conversión de la parte ciano en
tiocianato. Los ácidos carboxílicos y fenoles resultantes se
combinaron con ácido sulfúrico, glicocola y ácido glucurónico.
Cuando se administró a ratones C14-(compuesto irradiado ácido,
alcohol o ciano)-deltametrina por vía oral, a tasas de 1,7 - 4,4
mg/kg, la excreción del radiocarbono fue rápida y casi total,
excepto en la parte ciano. Las principales reacciones metabólicas
observadas en los ratones fueron en general semejantes a las
observadas en las ratas.
En el ganado vacuno y las aves de corral, las vías de
degradación son muy similares a las observadas en los roedores.
1.1.6 Efectos en los organismos presentes en el medio
La deltametrina es muy tóxica para los peces, ya que la CL50 en
96 horas oscila entre 0,4 y 2,0 µg/litro. Es también muy tóxica
para los invertebrados acuáticos: la CL50 en 48 horas para Daphnia
es de 5 µg/litro. Sin embargo, extensos estudios realizados en
estanques experimentales y el empleo sobre el terreno han
demostrado que esa elevada toxicidad potencial no llega a
concretarse. En la práctica hay algunas muertes de invertebrados
acuáticos que, por lo general, se compensan con rapidez.
La toxicidad de la deltametrina en las aves es muy baja, con
valores de la DL50 superiores a 1000 mg/kg cuando se administra una
sola dosis por vía oral. En condiciones de laboratorio, la
deltametrina es muy tóxica para las abejas, con una DL50 por
contacto de 0,051 µg/abeja. Los ensayos sobre el terreno y el
empleo no experimental han demostrado que los preparados de
deltametrina tienen una acción repelente, lo cual quiere decir que,
en la práctica, el riesgo para las abejas es escaso.
1.1.7 Efectos en animales experimentales y en sistemas de pruebas
in vitro
En un vehículo no acuoso, la toxicidad de la deltametrina
administrada por vía oral en forma aguda va de alta a moderada, con
DL50 de 19 a 34 mg/kg (ratones) y de 31 a 139 mg/kg (ratas). Sin
embargo, cuando la deltametrina está en suspensión en agua, su
toxicidad es mucho menor con DL50 superiores a 5000 mg/kg (ratas).
La deltametrina es un piretroide de tipo II; los signos clínicos
comprenden temblor, salivación y convulsiones. La intoxicación se
presenta rápidamente y, en los supervivientes, los signos
desaparecen en unos días. El electroencefalograma muestra
descargas generalizadas en picos y ondas, seguidas de
coreoatetosis.
Aplicaciones únicas de deltametrina técnica no tuvieron ningún
efecto irritante en la piel, intacta o con abrasiones, de conejos.
Sin embargo, se observaron efectos irritantes transitorios en los
ojos de conejos, con y sin lavado. La deltametrina no actuó como
sensibilizante dérmico en cobayas.
En ratas a las que se administró deltametrina con sonda, en
dosis de hasta 10,0 mg/kg de peso corporal diarios durante 13
semanas, a las 6 semanas se observó hiperexcitabilidad en los
machos que habían recibido la dosis más alta. El aumento del peso
corporal en los machos fue menor con dosis de 2,5 y 10 mg/kg.
En perros de la raza beagle a los que se administró por vía
oral deltametrina en dosis de hasta 10 mg/kg de peso corporal
diarios durante 13 semanas, se observaron varios síntomas
relacionados con el compuesto, como vómitos, temblor, salivación y
reflejos faríngeo, patelar y flexor deprimidos. En un estudio de
alimentación en perros, de dos años de duración, la dosis
desprovista de efectos fue de 1 mg/kg de peso corporal diario
(dosis más alta estudiada).
En ratones a los que se administró deltametrina en dosis de
hasta 100 mg/kg de alimentos durante 24 meses, no resultó afectada
la incidencia de tumores. En cuanto a la toxicidad general, la
dosis desprovista de efectos fue de 100 mg/kg de alimentos.
En ratas a las que se administró deltametrina a niveles de
hasta 50 mg/kg de alimentos durante dos años, no se observaron
tumores relacionados con el compuesto. La concentración
desprovista de efectos de la toxicidad general fue de 50 mg/kg de
alimentos.
No se observaron efectos mutagénicos de la deltametrina en una
multitud de sistemas de pruebas in vivo e in vitro, incluidos:
reparación del ADN, mutación génica, aberración cromosómica,
intercambio de cromátidas hermanas, formación de micronúcleos y
genes letales dominantes.
Se realizaron estudios teratológicos con ratas y ratonas
preñadas, administrando deltametrina por vía oral en dosis de hasta
10 mg/kg diarias durante el periodo de mayor organogénesis. No se
observaron efectos teratogénicos ni reproductivos en las ratas ni
en las ratonas, excepto una disminución, relacionada con la dosis,
del peso fetal medio en el estudio con ratonas y una osificación
ligeramente retrasada en el estudio con ratas.
Se administró a conejas deltametrina en dosis de hasta 16 mg/kg
diarios entre los días 6 y 19 del embarazo. Con la dosis más alta,
se registró una disminución del peso fetal medio. No se observaron
efectos teratogénicos.
Se administró a ratas deltametrina en dosis de hasta 50 mg/kg
de alimentos en un estudio sobre reproducción con tres generaciones
y dos camadas, sin que se observaran efectos sobre la reproducción.
Hay indicios de que la toxicidad puede potenciarse cuando la
deltametrina se combina con algunos compuestos organofosforados.
1.1.8 Efectos en los seres humanos
La deltametrina puede provocar sensaciones cutáneas en los
trabajadores expuestos. Se han notificado varios casos de
intoxicación no mortal debidos a exposición ocupacional por no
respetar las precauciones de seguridad. Son síntomas
frecuentemente mencionados adormecimiento, picor, hormigueo y
quemazón de la piel, y vértigo. En ocasiones se ha descrito un
eritema papular o maculoso. La mayor parte de esos síntomas son
temporales y desaparecen en 5 ó 7 días. No se han comunicado
efectos negativos a largo plazo. Se han descrito tres casos no
mortales de intoxicación por deltametrina tras la ingestión de
varios gramos del producto.
1.2 Conclusiones
Población en general: No es probable que la exposición de la
población en general a la deltametrina, que se cree muy baja,
represente un riesgo en las condiciones de empleo recomendadas.
Exposición ocupacional: Si se aplican buenas prácticas laborales,
medidas de higiene y precauciones de seguridad, no es probable que
la deltametrina represente un riesgo para las personas
ocupacionalmente expuestas a ella.
Medio ambiente: Es improbable que la deltametrina o los productos
de su degradación alcancen niveles que puedan tener efectos
negativos en el medio con las tasas de aplicación recomendadas. En
condiciones de laboratorio, la deltametrina es muy tóxica para los
peces, los artrópodos acuáticos y las abejas. No obstante, en la
práctica, no es probable que haya efectos perjudiciales duraderos
con las condiciones de empleo recomendadas.
1.3 Recomendaciones
Aunque se cree que con el uso recomendado las concentraciones
en la dieta son muy bajas, debe considerarse la conveniencia de
confirmarlo incluyendo la deltametrina en estudios de vigilancia.
La deltametrina se utiliza desde hace muchos años y se han
notificado varios casos de intoxicación no mortal y efectos
transitorios debidos a exposición ocupacional. Debe mantenerse la
observación de la exposición humana.
After oral administration to male rats at 0.64 - 1.60 mg/kg,
the acid and alcohol moieties of deltamethrin were almost
completely eliminated from the body within 2 - 4 days (Ruzo et al.,
1978). On the other hand, the cyano group was eliminated more
slowly, the total recovery during 8 days being 79% of the
radiocarbon dose (43% and 36% in the urine and faeces,
respectively). Tissue residues of deltamethrin labelled with 14C
at the dibromovinyl carbon in the acid moiety and the benzylic
carbon in the alcohol moiety were generally very low, whereas
residue levels in the fat were somewhat higher (0.1 - 0.2 mg/kg).
Residue levels of the radiocarbon derived from the cyano group were
relatively high, especially in the skin and stomach. Essentially,
all the radiocarbon in the stomach was thiocyanate. No noticeable
14CO2 was evolved from any of the radioactive preparations,
including the CN-labelled group, in contrast to the CN group from
fenvalerate, which yielded 14CO2 in considerable amounts.
The major metabolic reactions of deltamethrin were oxidation
(at the trans methyl relative to carbonyl group of the acid moiety
and at the 2'-, 4'-, and 5-positions of the alcohol moiety),
cleavage of the ester linkage, and conversion of the cyano portion
to thiocyanate and 2-iminothiazolidine-4-carboxylic acid (31)
(ITCA) (see Fig. 3). These carboxylic acid and phenol derivatives
were conjugated with sulfuric acid, glycine, and/or glucuronic
acid.
The major faecal metabolites were unchanged deltamethrin (9),
accounting for 13 - 21% of the dose, followed by 4'-OH- (10) and
5-OH-deltamethrin (28), and a trace amount of 2'-OH-deltamethrin
(29). Intact deltamethrin and the 4'-OH-derivative appeared not
only as the administered S-epimer, but also in parts as the
R-epimer, probably due to artefactural racemization on exchange of
the alpha-position hydrogen in methanol solution. The metabolites
from the acid moiety were mostly 3-(2,2-dibromovinyl)-2,2-
dimethylcyclopropanecarboxylic acid (18) (Br2CA) in free form
(10% of the dose), glucuronide (51%) and glycine (trace level)
conjugates, and OH-Br2CA (20) in free form and glucuronide
conjugate (<1%).
The major metabolites of the aromatic portion of the alcohol
moiety were 3-phenoxybenzoic acid (25) (PBacid) in free form (5%),
and glucuronide (13%) and glycine (4%) conjugates and its
4'-hydroxy derivative (26) (4'-OH-PBacid).
Sulfate of 4'-OH-PBacid accounted for about 50% of the dose,
together with small amounts of free (4%) and glucuronide forms
(2%). The CN group was converted mainly to thiocyanate (30) and,
in small amounts, to ITCA (31) (Ruzo et al., 1978). The trans-
isomer of deltamethrin was also rapidly metabolized and yielded
almost the same metabolites as deltamethrin, though 5-OH-derivative
was found in the cis-isomer, but not in the trans-isomer (Ruzo et
al., 1978).
When a single oral dose of 14C-(acid-, alcohol-, or cyano-
labelled) deltamethrin was administered to male mice at 1.7 - 4.4
mg/kg, the acid moiety and the aromatic portion of the alcohol
moiety were rapidly and almost completely excreted, whereas the CN
group was excreted relatively slowly (Ruzo et al., 1979).
Gray & Rickard (1982) followed the distribution of 14C-acid-,
14C-alcohol-, and 14C-cyano-labelled deltamethrin and selected
metabolites in the liver, blood, cerebrum, cerebellum, and spinal
cord after iv administration of a toxic, but non-lethal, dose
(1.75 mg/kg) to rats. Approximately 50% of the dose was cleared
from the blood within 0.7 - 0.8 min, after which the rate of
clearance decreased. 3-Phenoxybenzoic acid (PBacid) was isolated
from the blood in vivo, and was also the major metabolite when
14C-alcohol-labelled deltamethrin was incubated with blood in
vitro. Deltamethrin levels in the liver peaked at 7 - 10 nmol/g at
5 min and then decreased to 1 nmol/g by 30 min. In contrast, peak
central nervous system levels of deltamethrin were achieved within
1 min (0.5 nmol/g), decreasing to 0.2 nmol/g at 15 min, and
remaining stable until 60 min. Peak levels of deltamethrin were
not related to the severity of toxicity, though the levels of
unextractable pentane radiolabel did appear to be correlated with
signs of motor toxicity. Experiments with brain homogenates from
animals injected iv with deltamethrin failed to reproduce the
pentane-unextractable radioactivity in vitro and metabolism of the
compound was not demonstrated.
The major metabolic pathways of deltamethrin in mice were
similar to those in rats, though there were some differences. These
included the presence of more unchanged deltamethrin in mouse faeces
than in rat faeces. In mouse faeces, there were 4 monohydroxy ester
metabolites (2'-OH-, 4'-OH-, 5-OH-, and trans-OH- deltamethrin
(11)) and one dihydroxy metabolite (12) (4'-OH- trans-
OH-deltamethrin) that were not found in mouse urine. Major
metabolites from the acid moiety in mice were Br2CA,
trans-OH-Br2CA (20), and their glucuronide and sulfate
conjugates. Among them, trans-OH-Br2CA-sulfate was detected
only in mice, but not in rats. Compared with rats, much larger
amounts of trans-OH-Br2CA and its conjugates were formed in
mice. A major metabolite of the alcohol moiety in mice was the
taurine conjugate of PBacid in the urine, which was not detected in
rats. Generally, mice produced smaller amounts of phenolic
compounds compared with rats. Also, 3-phenoxybenzaldehyde (24)
(PBald), 3-phenoxybenzyl alcohol (32) (PBalc), and its glucuronide,
and glucuronides of 3-(4- hydroxyphenoxy)benzyl alcohol (33)
(4'-OH-PBalc) and 5-hydroxy-3- phenoxybenzoic acid (34)
(5-OH-PBacid) were found in mice, but not in rats. When mice were
given an ip dose of 14C-deltamethrin, with or without piperonyl
butoxide (PBO) and/or S,S,S-tributyl- phosphorotrithioate (DEF),
the same metabolites were obtained as with oral administration.
However, DEF decreased the hydrolytic products relative to the
controls, while PBO decreased the oxidation products (Ruzo et al.,
1979).
The comparison between the excreted radioactivity of
14C-deltamethrin in rats treated by the percutaneous route and iv
(controls) showed that only 3.6% of the dosage applied on the skin
was absorbed and excreted in 24 h with 1.1% excreted during the
first 6 h. Since the rat skin is more permeable than human skin,
the uptake of deltamethrin through the human skin should be
relatively weak (Pottier et al., 1982).
5.2 Metabolism and Fate in Farm Animals
In a metabolic study, 14C-deltamethrin was administered orally
to lactating dairy cows at the rate of 10 mg/kg body weight per day
for 3 consecutive days. It was poorly absorbed and mainly
eliminated in the faeces as unchanged deltamethrin. Only 4 - 6% of
the administered 14C was eliminated in the urine, and 0.42 - 1.62%
was secreted in the milk. The radiocarbon contents of various
tissues were generally very low with the exception of those of the
liver, kidney, and fat, which were higher (Akhtar et al. 1986).
Deltamethrin degradation occurred by cleavage of the ester bond, as
already reported in rats and mice (Ruzo et al. 1978, 1979). The
enzymes responsible for the ester bond cleavage were located in cow
liver homogenate, mainly in the microsomal fraction, as seen in an
in vitro study (Akhtar, 1984). Metabolites resulting from ester
bond cleavage were further metabolized and/or conjugated, resulting
in a large number of compounds excreted in the urine (see Fig. 3).
In milk, the major identifiable radiolabelled compound was
deltamethrin.
In a feeding study by Akhtar et al. (1987), deltamethrin was
administered twice daily to lactating dairy cows in portions of
their daily feed at the rate of 2 or 10 mg/kg diet for 28
consecutive days. The level of 2 mg/kg diet was the residue level
found in a recently treated pasture (Hill & Johnson, 1987), whereas
10 mg/kg diet was five times this level. Deltamethrin residues in
the milk were dose-dependent and appeared to reach a plateau
between 7 and 9 days after the start of treatment. At the high
deltamethrin intake of 10 mg/kg diet, the deltamethrin residue in
milk was about 0.025 mg/litre. Deltamethrin residues in tissues
were measured 1, 4, and 9 days after the last dose. At the
10 mg/kg diet intake, very small amounts of deltamethrin residues
were found in the liver (<0.005 mg/kg), kidney (<0.002 mg/kg),
and muscle (0.002 - 0.014 mg/kg). Residues in fat were about
0.04 mg/kg and 0.2 mg/kg for the 2 and 10 mg/kg intake,
respectively. Depletion of deltamethrin residues in milk was very
rapid (estimated half-life was about 1 day); while in fat (renal
and subcutaneous) the half-life was 7 - 9 days. Br2CA (3-(2,2-
dibromovinyl)-2,2-dimethylcyclopropanecarboxylic acid) and PBacid
(3-phenoxybenzoic acid) were the only metabolites detected in the
milk and tissues of treated cows. In all cases, they were found at
trace levels of < 0.0235 mg/litre and < 0.034 mg/litre,
respectively. These two metabolites were also previously
identified in rats and mice as the major degradation products of
deltamethrin (Ruzo et al., 1978, 1979).
The fate of 14C-deltamethrin was examined in Leghorn hens
(Akhtar et al., 1985). When laying hens were administered 7.5 mg
of 14C-labelled deltamethrin/hen per day orally for 3 consecutive
days, about 83% and 90% of the administered 14C was eliminated
during the first 24 h and 48 h after dosing, respectively. Tissue
residues were generally very low with the exception of those in the
liver and kidney. Very low levels of residues were found in eggs
obtained within the first 24 h after dosing, but levels increased
reaching a peak within 48 h of the last dose. Residue levels were
higher in the yolk (up to 0.6 mg/kg) than in the albumen (up to
0.2 mg/kg), which is probably related to the lipid content of
yolks. Metabolites were the same as those found in rats and mice.
These studies showed that feeding domestic animals on
deltamethrin-treated feed resulted in very low levels of residues
(if any) in products of animal origin and is unlikely to present a
hazard for the consumer.
5.3 Enzymatic Systems for Biotransformation
Deltamethrin (1 µg) was incubated at 37 °C for 30 min with each
of the following mouse microsome preparations; a) tetraethyl
pyrophosphate (TEPP)-treated microsomes (no esterase and oxidase
activity); b) normal microsomes (esterase activity); c) TEPP-
treated microsomes plus NADPH (oxidase activity); and d) normal
microsomes plus NADPH (esterase plus oxidase activity) (Shono et
al., 1979). Deltamethrin was more rapidly metabolized under the
oxidase system than under the esterase system. The major site of
ring hydroxylation was the 4'-position and the secondary site was
the 5-position. The trans methyl group was an important site of
hydroxylation of the esters and cis methyl oxidation was evident in
the metabolites of the cleaved acid moiety. The preferred sites of
hydroxylation were as follows; trans of dimethyl group,
4'-position in the phenol group, and cis of the dimethyl group,
which was equal to the 5-position in the phenoxy group. Cleavage
of deltamethrin to cyanohydrin may result from both esterase and
oxidase enzyme activities, since larger amounts of the cleaved
products were evident in the oxidase system.
However, at a much higher (approximately 35-fold) concentration
of deltamethrin than that in the above study, it was not detectably
hydrolysed (Miyamoto, 1976; Soderlund & Casida, 1977).
Deltamethrin was hydrolysed by esterases in the blood, brain,
kidney, and stomach of mice yielding PBald and PBacid (Ruzo et al.,
1979).
5.4 Metabolism in Human Beings
Three young male human volunteers underwent a complete medical
check-up one week prior to the morning of the study. Each of them
received a single dose of 3 mg of 14C-deltamethrin mixed in 1 g
glucose and diluted first in 10 ml PEG 300 and again in 150 ml
water. Total radioactivity was 1.8 ± 09 mBq. Samples of blood,
urine, saliva, and faeces were taken at intervals over 5 days.
Clinical and biological examinations were performed every 12 h
during the trial and one week after its termination. Radioactivity
in the biological samples was measured with a liquid scintillation
spectrometer. The clinical and biological checks did not detect
any abnormal findings. There were no signs of side effects or
intolerance reactions, either during or after the trial period.
The maximum plasma radioactivity appeared between 1 and 2 h after
administration of the product, and remained over the detection
limit (0.2 KBq/litre) during the 48 h. The apparent elimination
half-life was between 10.0 and 11.5 h. The radioactivity of blood
cells, as well as the saliva, was extremely low. Urinary excretion
was 51 - 59% of the initial radioactivity; 90% of this
radioactivity was excreted during the 24 h following absorption.
The apparent half-life of urinary excretion was 10.0 - 13.5 h,
which is consistent with the plasma data. Faecal elimination at
the end of the observation period represented 10 - 26% of the dose.
The total faecal plus urine elimination was around 64 - 77% of the
initial dose after 96 h (Papalexiou et al., 1984).
6. EFFECTS ON ORGANISMS IN THE ENVIRONMENT
6.1 Aquatic Organisms
6.1.1 Acute toxicity for fish
Acute toxicity data for deltamethrin in fish have been
summarized by L'Hotellier & Vincent (1986) (Table 6). From this,
it appears that deltamethrin is highly toxic for fish, though the
toxicity varies with the formulation tested.
Table 6. Acute toxicity of deltamethrin tested as the technical or formulated product on
fish; lethal concentrations all expressed as µg active ingredient (a.i.)/litre (96-h)
------------------------------------------------------------------------------------------
Species Systema LC50 (µg/litre) Ref. LC50 (µg/litre) Ref.
(Common name) tested as No. tested as No.
technical formulated
product productb
------------------------------------------------------------------------------------------
Alburnus alburnus S 0.69 4 82 (ULV) 4
(Bleak)
Brachydanio rerio F,S 2.0 10 -
(Zebra fish)
Cyprinodon macularius S - 0.6c (EC) 13
(Desert pupfish)
Cyprinodon S - 0.9 (EC) 19
variegatusd
(Sheepshead minnow)
Cyrpinus carpio F, S 1.84 4 0.65 (EC) 4
(Common carp) 0.86 3 210.0 (ULV)
Gambusia affinis F, S - 1.0c (EC) 13
(Mosquito fish)
Ictalurus nebulosus F, S 1.2 7 2.3 (EC) 15
(Brown bullhead)
Ictalurus punctatus F, S 0.63 8 -
(Hannel catfish)
Idus idus melanotus S - 1.2 (EC) 16
(Golden orfe)
Lebistes reticulatus F, S - 1.8 (EC) 17
(Guppy)
Lepomis gibbosus F, S 0.58 5 0.87 (EC) 14
(Pumpkinseed sunfish)
Lepomis machrochirus F 1.2 6 -
(Bluegill sunfish)
Osteochilus hasseltie S - 1.2 (EC) 20
(Nilem carp)
Puntius gonionotuse F,S - 0.87 (EC) 18
(Jawa carp)
Rhodeus sericeus S 1.12 4 140 (ULV) 4
amarus
Salmo gairdneri F, S 0.39 1 2.2 (EC) 12
(Rainbow trout)
Salmo salar 1.97 2 0.59 (EC) 2
------------------------------------------------------------------------------------------
Table 6. (contd.)
------------------------------------------------------------------------------------------
Species Systema LC50 (µg/litre) Ref. LC50 (µg/litre) Ref.
(Common name) tested as No. tested as No.
technical formulated
product productb
------------------------------------------------------------------------------------------
Salmo trutta F, S - 4.7c (EC) 11
(Brown trout)
Sarotherodon F, S 3.5 9 2.0 (EC) 9
mossambicuse
Tilapia mossambicae F, S - 0.8c (EC) 13
------------------------------------------------------------------------------------------
a F: Flow system, S: Static condition.
b EC: 25 g a.i./litre; ULV: 1 g a.i./litre; values in a.i. equivalent obtained by
calculation.
c LC50 (48-h)
d Marine fish.
e River or pond fish from tropical areas (water temperature > 24 °C).
References
(1) Knauf & Horlein (1979); (2) Zitko et al. (1979); (3) Knauf & Schulze (1977a);
(4) Gulyas & Csanyi (undated); (5) Waltersdorfer & Schulze (1976a); (6) Buccafusco et al.
(1977a); (7) Knauf & Schulze (1977b); (8) Buccafusco et al. (1977b); (9) Adeney et al.
(1980); (10) Lepailleur & Chambon (1984); (11) Lhoste et al. (1979); (12) Waltersdorfer &
Schulze (1976c); (13) Mulla et al. (1978); (14) Waltersdorfer & Schulze (1976d); (15) Knauf
& Schulze (1977b); (16) Waltersdorfer & Schulze (1976b); (17) Waltersdorfer & Schulze
(1976a); (18) Santosa & Hadi (1980); (19) Heitmuller et al. (1978); (20) Santosa (1983)
Zitko et al. (1979) established a 96-h lethal threshold for
Atlantic salmon (Salmo salar) of 1.97 µg/litre.
6.1.2 Acute toxicity for other aquatic organisms
Data on aquatic organisms other than fish are presented in
Table 7 and are of the same order as those for fish, although the
oyster (Crassostrea virginica) is somewhat more tolerant and the
Northern lobster (Homarus americanus) (96-h lethal threshold
0.0014 µg/litre) is far more sensitive (Zitko et al., 1979).
Mohsen & Mulla (1981) exposed aquatic insect larvae to
deltamethrin (as a 2.5% emulsifiable concentrate) for 1 h under
flow-through conditions, and calculated the LC50 after a 24-h
holding period. For the target species blackfly (Simulium
virgatum) an LC50 of 0.9 µg/litre was calculated. Non-target
species tested, mayfly (Baetis parvus) and caddisfly (Hydropsyche
californica), were found to be more susceptible, with LC50 values
of 0.4 µg/litre.
Varanka, (1987) investigated the effects of deltamethrin on
three species of freshwater mussels. Results presented in Table 8
show that the mussels are very insensitive to the pyrethroid.
Table 7. Acute toxicity of deltamethrin tested as technical or
formulated product on other aquatic organisms-lethal concentrations
expressed as µg active ingredient (a.i.)/litre (96-h)a
-------------------------------------------------------------------
Species LC50 (µg/litre) LC50 (µg/litre)
tested as technical tested as formulated
product productb
-------------------------------------------------------------------
Crassostrea virginica - 12.0
(Eastern oyster)
Daphnia magna 5c -
(Water flea)
Gammarus pulex - 0.03c
(Scud)
Penaeus duorarum - 0.35
(Pink shrimp)
Uca pugilatorulosus - 1.1
(Fiddler crab)
Bufo bufo (larvae) - 0.93
(Common toad)
-------------------------------------------------------------------
a Adapted from: L'Hotellier & Vincent (1986).
b EC: 25 g a.i./litre; ULV: 1 g a.i./litre; values in a.i.
equivalent obtained by calculation.
c LC50 (48-h).
Table 8. Acute toxicitya of deltamethrin formulationb in
freshwater mussels, under static conditions at 21 - 23 °Cc
-------------------------------------------------------------------
Species 24-h 48-h 72-h 96-h 7-day
-------------------------------------------------------------------
Anodonta cygnea nd nd ~24.6 12.0 7.6
Anodonta anatina nd nd nd ~23.4 10.3
Unio pictorum nd ~31.8 9.7 7.0 6.0
-------------------------------------------------------------------
a LC50 µg active ingredient (a.i.)/litre): values in a.i.
equivalent obtained by calculation.
b ULV 0.12%.
c From: Varanka (1987).
6.1.3 Field studies and community effects
Two experimental pond studies have been performed. Tooby et
al. (1981) reported that application of deltamethrin to static
water at 10 g a.i./ha did not have any lethal effects on two fish
species (Canassius auratus, Rutilus rutilus) or on molluscs.
Aquatic insects and crustaceans present were killed. Rawn et al.
(1985) applied deltamethrin at a similar rate and also reported
that no fish were killed. The half-life of deltamethrin in the
pond was 2 - 4 h for water and 2 - 14 days for bottom sediment.
Neto et al. (1983) sprayed-flooded fields in Brazil, at
intervals of 2 days, with rates of deltamethrin progressively
increased at 5, 10, 12, and 13 g a.i./ha. The expected
concentrations in water from these applications were between 3 and
7 µg/litre. No mortality was recorded in fish placed in the
sprayed area in experimental cages. Slight "agitation" was
reported after exposure to the highest dose.
Impact assessments on the use of deltamethrin on paddy fields
have been made in the field in various countries throughout the
world. The maximum normal usage rate of the compound was 6.5 g
a.i./ha. In these studies, fish ( Tilapia spp., Cyprinus carpio,
Gambusia spp.) tolerated deltamethrin up to 18.75 g a.i./ha without
any adverse effects. The compound is known to be toxic for aquatic
organisms and is not recommended for use over water under any but
exceptional circumstances. However, it has been used to control
vectors of major human diseases, i.e., mosquitos and blackfly
( Elossina spp.), where benefit outweighed potential risk. In these
cases, extensive field evaluations of the environmental impact have
been made. While there have not been any instances of fish kills
from these applications, there are reports of large numbers of
deaths of aquatic invertebrates. The populations usually recovered
rapidly and all studies have shown numbers back to normal before
the compound was applied again in the following season. It is
suggested that relatively resistant parts of the population soon
recolonize the area; immigration also occurs (Takken et al., 1978;
Smies et al., 1980; Baldry et al., 1981; Everts et al., 1983).
6.1.4 Appraisal
Notwithstanding its high toxicity for fish and crustacea, the
results of many studies, as well as the wide use of deltamethrin
for several years, have confirmed that its normal use does not
cause significant mortality in fish populations. This difference
is due to its strong adsorption on soil and its rapid breakdown,
decreasing its bioavailability under field conditions.
6.2 Terrestrial Organisms
6.2.1 Plants
Hargreaves & Cooper (1979) sprayed glasshouse-grown tomato
seedlings with 50 mg deltamethrin/litre (2.5% emulsifiable
concentrate) 3 weeks after emergence and again 7 days later. Three
days after the second application, plants were examined for damage.
No damage was found and, at this rate of use, deltamethrin was not
phytotoxic.
6.2.2 Soil microorganisms
In a study by Tu (1980) on the effects of 5 pyrethroids on
microbial populations and their activity in soil, 0.5 mg
deltamethrin/kg incorporated into sandy loams (residues under
normal use conditions would be of the order of < 0.001 mg/kg)
produced only a few transient effects. No effects were noted on
the nitrifying microorganisms and their capacity to produce nitrate
and there were no inhibitory effects on deshydrogenase or urease
activity. Deltamethrin induced an increase in oxygen consumption
because of an increase in microbial respiration (probably linked
with the microbial degradation of deltamethrin). It also
stimulated the growth of soil fungi and inhibited the development
of bacteria. Four weeks after treatment, deltamethrin-treated soil
recovered completely and microorganism activity was equal to that
in untreated soil.
6.2.3 Soil fauna
6.2.3.1 Earthworms
When deltamethrin at 12.5 g a.i./ha (high agricultural dose)
was incorporated into the soil to a depth of 1 cm, there were no
toxic effects on earthworms (Lumbricus terrestris) during an
observation period of 28 days (Bouche & Fayolle, 1979). However,
significant toxic effects on earthworms were observed at levels of
60 - 125 g a.i./ha (5 - 10 times the highest rates applied in
agriculture).
In another study with Eisenia foetida andrei, deltamethrin
incorporated in artificial soil at concentrations of 1.7 mg/kg and
10 mg/kg did not produce any lethal effects (Chambon & Lepailleur,
1984).
6.2.3.2 Slugs
Lettuce leaves treated with 4 times normal dosage rates, were
fed to slugs ( Agrolimax sp.). Leaves were quickly consumed but no
toxic effects (mortality or activity) were observed (Ricou, 1978).
6.2.3.3 Soil arthropods
Under laboratory conditions, deltamethrin, applied topically
and by immersion, was very toxic for the carabid beetle
Pterostichus melanarius (Illiger). Under natural conditions in the
field, deltamethrin applied at normal dose rates was not toxic for
these organisms (Dunning et al., 1981).
Everts et al. (1985) monitored the effects, on non-target
organisms, of various compounds when used for the control of tsetse
fly in the Ivory Coast in Africa. Deltamethrin was the most
effective compound against the tsetse and also killed non-target
musca flies. After deltamethrin spraying, Orthoptera and
Proctotrupoidea were also significantly decreased while Nematocera
increased in number. The results of this study suggest that ground
spraying of the pyrethroid had greater effects on terrestrial
arthropods than aerial applications.
Concurrent laboratory and field studies were conducted on the
effects of deltamethrin on beneficial predatory spiders in a polder
area of the Netherlands (Everts et al., 1988). During two growing
seasons, 2800 samples were taken over an area of 17 different
fields. The authors found that effects on spiders were eliminated
when it rained soon after application, since the effect of the
pyrethroid appeared to be indirect, causing the dehydration of
spiders. This different response under dry and damp conditions was
confirmed in the laboratory. However, reduction of spider numbers
in the field was much greater than predicted from laboratory tests
and recovery was more rapid in laboratory populations than in field
populations. The uptake and effects of deltamethrin were greater
through exposure to residues than through contact or oral exposure.
There was a positive correlation between temperature and the
toxicity of deltamethrin for spiders in the field. This contrasted
with reports of a negative correlation for target insects reported
in the literature. Laboratory studies showed that the negative
temperature effect only occurred when spiders could not drink. It
appeared that qualitative prediction from laboratory to field was
possible but that quantitative prediction was not.
6.2.4 Beneficial insects
6.2.4.1 Honey-bees
Single applications of deltamethrin are highly toxic for honey-
bees (Apis mellifera). Stevenson et al (1978) found a contact LD50
of 0.051 µg/bee and an oral LD50 of 0.079 µg/bee.
Arzone & Vidano (1978) did not find any difference in mortality
between controls and bees fed on sugar solutions containing 0.2 µg
deltamethrin/litre. Increased mortality was recorded at all higher
exposures reaching 100% within 1 h at a concentration of 12.5
µg/litre.
In the field, direct treatment of caged bees caused a high
mortality rate with doses of from 11.2 g/ha upwards (Atkins et al.,
1976). Rape flowers were treated at a rate of 0.75 g a.i./100
litre and 1.5 g a.i./100 litre with an emulsifiable concentrate
formulation, 25 g/litre; control plots were treated with water.
Cages (3 x 2 x 2 m) containing a small hive (2 frames + open brood)
were put over the treated flowers once the spray had dried. The
mortality of the bees was then assessed over 7 days. The average
mortalities were not significantly higher in the treated plots than
in water-sprayed control plots (Louveaux et al., 1977).
However, Bocquet et al. (1980, 1983) demonstrated, after 3
years of field experiments, that deltamethrin under field
conditions was innocuous at doses up to 12.5 g/ha. They also noted
a repellant effect by the formulating materials, which lasted for
2 - 3 h. Further studies have been reported by Florelli et al.
(1987a,b).
6.2.4.2 Foliar insects
Deltamethrin was 70 times more toxic to the tobacco budworm
(Heliothis virescens) than to its predator, green lacewing
(Chrysopa carnea), but it was only 1.25 times more toxic to the
tobacco budworm than to its parasite (Campoletis sonorensis) (Plapp
& Bull, 1978).
In an apple orchard, where deltamethrin was applied at
12.5 mg/kg, no predatory mites (Typhlodromus pyri) were found
during 10 weeks of observation, but spider mites (Paponychus ulmi)
were not affected. The elimination of the predatory mite led to a
marked increase in spider mite populations, later in the same
season (Aliniazee & Cranham, 1980).
The impact of deltamethrin used against the English grain aphid
(Sitobion avenae) was studied in 1983, 1984, and 1985 in the Paris
basin. This study was carried out on wheat with pitfall traps,
yellow water traps, suction sampling (D-vac), and sampling of ears.
Effects were noted on: S. avenae, phytophagous Diptera (Opimyza
florum, Phytomyza nigra, and Oscinella frit), Homoptera (Zyginidia
scutellaris, Metopolophium dirhodum), Thysanoptera (Limothrips
cerealium, Acolothrips intermedius), predatory Diptera (Empididae,
Dolichopodidae), and on spiders (Erigonidae, Lycosidae,
Linyphiidae, Theridiidae). The detritiphagous insects (Sciaridae,
Chironomidae), the Carabidae and Staphylinidae and most
microhymenoptera showed little or no difference after treatment.
During the 3 years, no differences were observed from year to year
as a result of field treatment, populations appearing homogeneous
at the beginning of each trial (Fischer & Chambon, 1987).
A large-scale field trial was carried out in 1984 in southern
England to investigate the side-effects of deltamethrin on non-
target arthropods in winter wheat. The insecticides were applied
in June and two methods, suction sampling (D-vac) and quadrats,
were used to sample the arthropods for up to 75 days after
treatment. During the post-treatment period, the numbers of
Carabidae and Staphylinidae adults found in D-vac samples were
reduced by 22% and 20%, respectively, compared with the controls
(Vickerman et al., 1987a).
In the same field trial, arthropods were sampled with a D-vac
for 11 weeks. Total numbers were similar in the control and
deltamethrin-treated plots. The numbers of Empididae were reduced
by deltamethrin, but Dolichopodidae were more numerous in treated
than in control plots. The numbers of Aphidius spp. were higher in
the deltamethrin-treated plots than in the control plots. The
numbers of Coccinellidae larvae were reduced (Vickerman et al.,
1987b).
6.2.5 Birds
6.2.5.1 Laboratory studies
Data on the acute toxicity of deltamethrin for birds are given
in Table 9.
Table 9. Acute toxicity of deltamethrin for birds
---------------------------------------------------------------------------
Species Sex Application LD50 (mg/kg) Reference
---------------------------------------------------------------------------
Red partridge male & oral >3000 Grolleau & Griban,
(Alectonis tufa) female 1976b
Grey partridge male & oral >1800 Grolleau & Griban,
(Perdix perdix) female 1976b
Chicken oral >1000 Grandadam, 1976
(Gallus domestica)
Hen adult oral >2500 Ross et al., 1978
female
Mallard duck oral >4640 Beavers & Fink,
(Anas platyrhynchos) 1977a
Game duck oral >4000 Grolleau & Griban,
1976a
---------------------------------------------------------------------------
The toxicity of deltamethrin for birds is very low. Both
technical grade and commercially formulated deltamethrin
administered in feed at 100 mg/kg diet was not palatable to
Japanese quail (Coturnix coturnix japonica), with strong individual
variations. Unpalatability diminished after repeated exposure and
even became reversed in the case of the purified deltamethrin,
which attracted quail already suffering from toxic effects (David,
1981).
Groups of 39 female Japanese quail (Coturnix coturnix japonica)
were given daily doses of 0, 0.2, or 1 mg technical deltamethrin
per animal, by gavage, over 34 days. No significant effects were
observed on reproduction (De Lavaur et al., 1985).
6.2.5.2 Field studies on birds
The low toxicity of deltamethrin for birds, indicated by
laboratory studies, has been confirmed in the field. In studies on
the ecological consequences of the use of the compound to control
tsetse fly (Takken et al., 1978) and blackfly (Smies et al., 1980)
in West Africa, populations of various species of insectivorous,
granivorous, and piscivorous birds were examined before and after
spraying. There were no indications of any effects on either
numbers or species diversity.
7. EFFECTS ON EXPERIMENTAL ANIMALS AND IN VITRO TEST SYSTEMS
7.1 Single Exposures
Tables 10 and 11 show the results of acute toxicity studies on
various animal species. From these tables, it is clear that the
vehicle has a great influence on the LD50, probably by influencing
absorption. Powder formulations and aqueous suspensions are
significantly less toxic than formulations in oils or organic
solvents (Pham Huu Chanh et al., 1984).
The acute oral toxicity of deltamethrin for rats produced such
symptoms as: staining of the fur, excessive grooming, salivation,
diarrhoea, drowsiness, weakness, dyspnoea, piloerection, ptosis,
difficulty in walking, general motor incoordination, hypotonia,
choreoathetosis, clonic seizures, and death (Glomot, 1979; Glomot
et al., 1979, 1981a; Kavlock et al., 1979; Ray & Cremer, 1979; Pham
Huu Chanh et al., 1984). Electroencephalogram (EEG) records showed
generalized spike discharges prior to choreoathetosis (Ray &
Cremer, 1979; Ray, 1980).
Mice presented far fewer symptoms than rats after oral dosing
at comparable levels, diarrhoea being the only reportable
observation (Glomot et al., 1980a).
Rats were injected intraperitoneally with 14C-labelled
deltamethrin at the threshold doses required to produce the motor
symptoms of toxicity of tremor and choreoathetosis. Blood and brain
samples were analysed for their total radiolabel content, and were
also extracted with ethyl acetate to determine the levels of
extractable parent deltamethrin and 3-phenoxybenzyl-derived acid and
the residual radiolabel after this extraction. There was a clear
correlation between onset of symptoms and blood and brain levels of
deltamethrin. It was found that certain threshold levels of parent
deltamethrin in the blood and brain were required for symptoms
development, and that the symptoms persisted for as long as this
threshold was maintained (Rickard & Brodie, 1985).
7.1.1 Mouse
Mice intravenously injected with deltamethrin showed intense
tremors, convulsions, and ataxia, immediately after administration.
Tachycardia and respiratory defects were also observed at higher
dosages. Surviving animals appeared normal after 4 - 5 h.
Immediately after intraperitoneal injection, jumping movements,
slight convulsions and prostration, ptosis, tail hypertonicity, and
cyanosis were observed. These toxic signs disappeared after 72 h
in surviving animals.
Animals administered deltamethrin by gavage showed muscular
stiffening and convulsions, 1 h after dosing. After 24 h,
hypermotility, stereotype movements of the head, tachycardia,
hypertonicity of the tail, and a few convulsions were observed.
Behaviour and appearance were normal again after 48 h (Glomot &
Chevalier, 1976a,c).
Table 10. Acute toxicity of technical grade deltamethrin
--------------------------------------------------------------------------------------------------------------
Species Sex Route Vehicle LD50 (mg/kg Reference
body weight)
--------------------------------------------------------------------------------------------------------------
Rat male oral sesame oil 128 Glomot & Chevalier (1976a)
female 139
male PEG 200 67
female 86
Rat male adult peanut oil 52 Kavlock et al. (1979)
female adult 31
female weanling 50
Rat male adult peanut oil 53 Gaines & Linder (1986)
female adult 30
female weanling 48
Rat male + female aqueous suspension > 5000 Audegond et al. (1981)
with carboxy- (no mortality)
methylcellulose
Rat dermal - 700 Panshina & Sasinovich (1983)
Rat male methylcellulose (1%) > 2940 Kynoch et al. (1979)
female
Rat female adult xylene > 800 Kavlock et al. (1979)
Rat male + female inhalation (6 h) dust 600 mg/m3 Coombs & Clark (1978)
Rat male adult (2 h) DMSO 10% aerosol 940 mg/m3 Kavlock et al. (1979)
female adult > 785 mg/m3
Rat male + female (1 h) micronized powder > 4620 mg/m3 Jackson & Hardy (1986)
Rat intraperitoneal - 58.8 Panshina & Sasinovich (1983)
Rat male intraperitoneal sesame oil 209 Glomot & Chevalier (1976b)
female 186
male PEG 200 24 Glomot & Chevalier (1976b)
female 25
--------------------------------------------------------------------------------------------------------------
Table 10. (contd.)
--------------------------------------------------------------------------------------------------------------
Species Sex Route Vehicle LD50 (mg/kg Reference
body weight)
--------------------------------------------------------------------------------------------------------------
Rat male intravenous PEG 200 3.3 Glomot & Chevalier, (1976c)
female 3.3
Rat female adult acetone 4 Kavlock et al. (1979)
female weanling 1.8
Mouse male oral sesame oil 33 Glomot & Chevalier (1976a)
female 34
male PEG 200 21 Glomot & Chevalier (1976a)
female 19
Mouse intraperitoneal - 33 Panshina & Sasinovich (1983)
Mouse male intraperitoneal sesame oil 171 Glomot & Chevalier (1976b)
female 166
Mouse male PEG 200 18 Glomot & Chevalier (1976b)
female 12
Mouse male PEG 200 4.1 Glomot & Chevalier (1976c)
female 4.0
Mouse male glycerol formal 5 Glomot & Chevalier (1976c)
female 5.8
Dog male + female oral in capsules >300 Glomot et al. (1977)
no mortality
Dog male + female PEG 200 2 Glomot & Chevalier (1976c)
Rabbit male dermal PEG 400 > 2000 Clair (1977)
female > 2000
--------------------------------------------------------------------------------------------------------------
Table 11. Acute toxicity of some formulations
--------------------------------------------------------------------------------------------------------------
Species Sex Route Formulation LD50 (mg/kg Reference
body weight)
--------------------------------------------------------------------------------------------------------------
Rat male, female oral 2.5% flowable formulation 22 000 Glomot et al. (1979)
Rat male, female oral 2.5% wettable powder >15 000 Glomot (1979)
Mouse male, female oral 2.5% wettable powder >15 000 Glomot et al. (1980a)
Dog male, female oral 2.5% wettable powder >10 000 Glomot et al. (1980b)
Rat male, female oral 2.5% emulsifiable concentrate 535 Coquet (1977)
Rat male, female oral 10 g/litre ULV >6 470 Coquet (1977)
Rat male, female inhalation (4 h) aerosol-2.5% wettable powder >2 800 mg/m3 Clark et al. (1980)
--------------------------------------------------------------------------------------------------------------
7.1.2 Rats
Rats intravenously injected with deltamethrin showed muscular
contractions, piloerection, respiratory defects, convulsions, and
paresis of the hind quarters, immediately following treatment.
Surviving animals showed normal behaviour after 48 h. Immediately
after intraperitoneal injection, tremor, convulsions, prostration,
and cyanosis were observed. These toxic signs disappeared after
48 h in surviving animals. Animals administered deltamethrin by
gavage showed motor incoordination, convulsions, respiratory
defects, and hypomotility, shortly after dosing. Normal behaviour
was observed after 3 days (Glomot & Chevalier, 1976c).
In an inhalation study (whole body exposure for 6 h),
hyperactivity, grooming, and irritation were observed during
exposure. The animals were hypersensitive to touch and noise and
showed uncoordinated movements. Gross pathological investigation
showed a gas-filled stomach and small intestine, and massive
haemorrhage and degeneration in the lung (Coombs & Clark, 1978).
Rats were exposed (whole body exposure) for 4 h to an aerosol
concentration of deltamethrin equal to 2.8 g/m3, the highest
attainable airborne concentration of a 2.5% wettable powder
formulation. Approximately 80% of the total aerosol had a mean
aerodynamic diameter of less than 5.5 µm. Dyspnoea and gasping
were observed in exposed rats. Relative lung weights and
macroscopic pathology were normal. There was no mortality (Clark
et al., 1980).
7.1.3 Rabbit
Rabbits (10 males and 10 females) were treated with 2 g
deltamethrin in 2 ml PEG 400 per kg body weight on 80 cm2 of
occluded shaved skin for 24 h. The animals were observed for 14
days. Two animals showed obvious erythema. No body weight changes
or abnormal behaviour were observed. On histological observation
of the liver, kidneys, and skin, small changes were observed, but
these were common for this strain of rabbit and not related to
treatment (Clair, 1977).
7.1.4 Dog
Dogs given oral doses of 100 mg deltamethrin/kg body weight or
more showed transient hyperexcitability, akinesia, vomiting, and
stiffness of the hind legs (Glomot et al., 1977).
Dogs orally dosed with 10.0 mg deltamethrin/kg body weight did
not display any clinical signs related to treatment (Glomot et al.,
1980b).
7.2 Irritation and Sensitization
7.2.1 Skin irritation
Male albino rabbits (12 per group) weighing 2.5 - 3.5 kg were
administered 0.5 g deltamethrin on either shaved intact or abraded
skin. The occlusive patch was fixed on the skin for 23 h.
Technical deltamethrin (98% purity) did not produce any irritant
effects (Coquet, 1976a).
Male albino rabbits (6) weighing 2.5 - 2.9 kg were administered
0.5 ml of formulated deltamethrin (25 g/litre flowable suspension
concentrate) to both shaved intact and abraded skin. The Primary
Irritation Index after 24 h exposure of occluded sites was 1.2,
i.e., slightly irritating (Glomot et al., 1981b).
An evaluation similar to the one described above was carried out
for a 2.5% wettable powder concentrate deltamethrin formulation.
Rabbits had a Primary Irritation Index of 2.41, i.e., moderately
irritating. Moderate erythema continued for 72 h, while the oedema
generally diminished, with the exception of scarified skin sites
(Glomot et al., 1981c).
The skin irritation potentials of Decis emulsifiable
concentrate 2.5% and Decis Flowable 2.5% were studied on rabbits
and guinea-pigs with 0.05, 0.10, 0.5, 1, and 2.5% deltamethrin.
The threshold irritative levels were 0.05% for Decis emulsifiable
concentrate and 2.5% for Decis Flowable. The intensity of
irritation depended on the relative content of organic solvents and
emulsifiers in the trade products. The water-soluble concentrate
of Decis 2.5% caused negligible risk of contact irritative
dermatitis (Bainova & Kaloyanova, 1985).
7.2.2 Eye irritation
Deltamethrin (0.1 g/animal) was administered into the
conjunctival sac of the eyes of 6 male albino rabbits, weighing
2.5 kg, with or without rinsing 60 seconds after instillation.
Deltamethrin produced transient irritating effects, both with and
without rinsing (Coquet, 1976b).
Male albino rats (9) weighing between 2 and 3 kg were
administered 0.1 ml of formulated deltamethrin (25 g/litre flowable
suspension concentrate) in the conjunctival sac. Six of the
treated eyes remained unwashed, while the remaining three were
rinsed with lukewarm water 20 - 30 seconds after instillation.
There was only transient clouding of the cornea in 2 animals 1 h
after dosing (1 washed, 1 unwashed), which cleared by day 2. Low
grade conjunctival irritation was noted among all animals
initially, which disappeared following day 2 of observations
(Glomot et al., 1981d).
A 2.5% deltamethrin formulation diluted 1/10 in distilled water
(0.1 ml per rabbit) elicited a similar pattern of initial transient
corneal clouding in 3 out of 9 rabbits examined, which cleared by
day 4. The undiluted formulation (100 mg) administered in the
conjunctival sac of rabbits produced increased involvement of the
conjunctiva, iris, and cornea in all animals, generally moderate in
severity, with low grade corneal opacity persisting in 2 rabbits
until day 7 (1 washed, 1 unwashed) (Glomot et al., 1981e).
7.2.3 Sensitization
Deltamethrin (0.5 g/animal) was applied topically to the skin
of albino guinea-pigs (10 male and female) 3 times per week, with a
2-day interval for 3 weeks, and once at the start of the fourth
week. The preparation was covered with an occlusive patch for
48 h. On days 1 and 10, the guinea-pigs received an intradermal
injection of 0.1 ml of Freund's adjuvant. The animals were
challenged 12 days after the last application with 0.5 g
deltamethrin. No sensitization was found (Guillot & Guilaine,
1977).
7.3 Short-Term Exposure
7.3.1 Rat
Male and female weanling Sprague-Dawley rats (20 of each sex
per group) were dosed (by gavage) with 0, 0.1, 1, 2.5, or 10 mg
deltamethrin in PEG 200/kg body weight per day for 13 weeks. No
treatment-related effects were observed on food and water
consumption, mortality, urinalysis, and haematology. Neurological
examinations and ophthalmoscopy did not reveal any abnormalities.
At the highest dose level, a slight hyperexcitibility was observed
among some rats in week 6. Lower body weight gain was noted in
males at 2.5 and 10 mg/kg. No clear treatment-related effects were
noted in the results of laboratory investigations or on the weights
of the organs. Gross and microscopic examination of a variety of
tissues and organs did not show any treatment-related findings.
Following the 13-week dosage period, 5 males and 5 females per
group were allowed to recover for 4 weeks. No evidence of
hyperexcitability was observed among the rats; body weight gain was
slightly higher in the treated groups than in the controls. The
no-observed-effect level was 1 mg/kg body weight (Hunter et al.,
1977).
Four groups of CD rats (8 of each sex per group) were exposed to
aerosolized deltamethrin (technical grade powder) for 6 h per day, 5
days a week, for 2 weeks, and for 4 days during a third week. Mean
aerosol concentrations were 3, 9.6, and 56.3 mg a.i./m3 with
about 87% of respirable particles (diameter lower than 5.5 µm). No
rats died as a result of exposure. Signs of irritation (agitated
grooming and ptyalism due to the powder were noted in all groups
during exposure, with more pronounced toxic signs (ataxia and
walking with arched backs) in the group receiving the highest dose
tested. Male rats also showed a reduced body weight (-5%) in all
groups. An elevation of the serum sodium ion content was noted at
the two highest doses. No increased incidence of any particular
lesion was observed in the high-dose group compared with the control
group. Irritation and weight loss were only slight at 3 mg/m3 and
this can be considered as a no-effect level (Coombs et al., 1978).
7.3.2 Dog
Male and female beagle dogs (3 - 5/sex per group), 25 weeks of
age, received a daily oral dose of 0, 0.1, 1, 2.5, or 10 mg
deltamethrin/kg body weight in PEG 200 in gelatin capsules over 13
weeks. All treated groups showed reduced body weight gain, but
this was not dose-related. Liquid faeces were associated with all
groups of treated dogs throughout the dosing period. Dilatation of
the pupils was seen in dogs receiving 2.5 and 10 mg/kg per day.
The sign was first seen 4 - 7 h after dosing and persisted
throughout the day. The incidence of vomiting increased dose-
dependently in all treated groups, except the group receiving
0.1 mg/kg. In the highest dose group, unsteadiness, body tremors,
and jerking movements were seen, particularly in males, in weeks 2,
3, and 4. Excessive salivation was seen initially and diminished
during the dosing period. After 5 and 12 weeks, depression of the
gag reflex was noted in a proportion of animals in all treated
groups. However, this was not considered to be of toxicological
significance. Exaggeration or depression of the patellar reflex
was observed in some animals in all treated groups after 5 and 12
weeks, mainly at 1, 2.5, or 10 mg/kg per day. Some animals in all
treated groups showed depression of the flexor reflex. Dose levels
of 2.5 and 10 mg deltamethrin/kg per day caused modification of the
EEG pattern in some animals, 12 weeks following administration.
Histopathological evaluations of tissues and organs, including the
nervous system and muscle tissue, did not reveal any abnormalities
that could be related to the administration of the compound.
During recovery, the gag reflex continued to be depressed, whereas
exaggeration of the patellar reflex was still seen in some dogs
that had previously received 1 mg/kg per day (Chesterman et al.,
1977).
7.4 Long-Term Exposure and Carcinogenicity
7.4.1 Mouse and rat
Male and female Charles River CD-1 mice (80 of each sex per
group) were fed dietary levels of deltamethrin of 0, 1, 5, 25, or
100 mg/kg, daily, for 24 months. There were no clear effects
related to the administration of deltamethrin on general behaviour,
mortality, body weight, and food consumption. Blood chemistry,
haematology, and urine analysis parameters were normal after 12, 18,
and 24 months (at the times of interim and terminal sacrifice).
Microscopic examination of tissues did not reveal any lesions
indicative of a compound-related effect. The tumour incidence was
unaffected by deltamethrin administration. The no-observed-effect
level was 100 mg/kg diet (Goldenthal et al., 1980a).
Deltamethrin was administered, by gavage, to C57BL/6 mice at 4
dose levels (0, 1, 4, and 8 mg/kg body weight) and to BDVI rats at
3 dose levels (0, 3, and 6 mg/kg body weight) on 5 days a week for
104 weeks. After completion of the treatment, the animals were
observed until 120 weeks of age, when all survivors were killed.
The treatment had a slight effect on body growth and survival
rates, especially in the groups of mice and rats treated with the
highest dose. In C57BL/6 mice, various types of tumours were
observed in all treated groups. An increased incidence of
lymphomas was observed in mice receiving deltamethrin at levels of
1 and 4 mg/kg body weight, but not in the group treated with
8 mg/kg body weight. No significant difference in the incidences
of lung adenomas, liver-cell tumours, or other tumours was observed
in treated groups compared with controls. In BDVI rats, an
increased incidence of pituitary, thyroid, and mammary tumours was
noted; however, no clear dose-response relationship was shown
(Cabral et al., 1986). The details of this study were not
available for evaluation.
Male and female Charles River CD rats (90 of each sex per
group) were fed with 0, 2, 20, or 50 mg deltamethrin/kg diet for
2 years. A second control group (60 of each sex) was also used.
Interim sacrifices (10 of each sex per group excluding control
group 2) were made after 6, 12, and 18 months. No changes in
general behaviour and appearance were observed in relation to
treatment. Survival rate was similar for control and treated rats
(50 - 67%). Rats in the 50 mg/kg group gained slightly less weight
than control rats, whereas the food consumption was essentially the
same. Ophthalmoscopic findings were generally similar for control
and treated rats. No haematological and biochemical parameters
were changed in a biologically significant way in relation to
treatment at any time, except for a decrease in SGPT (serum
glutamic pyruvic transaminase) activity at 6 months in the mid- and
high-dose groups. Organ weights were not affected. The
macroscopic and microscopic findings were common for the species
and the strain, except for a slightly increased incidence of axonal
degeneration in sciatic, tibial, and/or plantar nerves in the 20
and 50 mg/kg groups at 18 months, but not at termination. Thus,
this was not considered to be indicative of a compound-related
effect. The incidence of benign testicular tumours (interstitial
cell adenomas) at terminal sacrifice in this study was: control
group 1, 0/37; control group 2, 4/35; low-dose group 1/38; mid-dose
group 1/30; high-dose group 6/38. The incidence seen in the high-
dose group was considered to be spontaneous, because it was not
significantly higher than in the second control group or in
historical control groups (Goldenthal et al., 1980b; Richter &
Goldenthal, 1983).
7.4.2 Dog
Deltamethrin dissolved in maize oil was administered in the diet
to 64 beagle dogs (8 of each sex per group) at levels of 0, 1, 10,
and 40 mg/kg for 24 months. This corresponds to 0, 0.025, 0.25, and
1 mg/kg body weight, respectively. Individual body weights and food
consumption values were determined weekly. Ophthalmoscopic,
haematological, biochemical, and urinalysis examinations were
conducted during the pre-test period and at 6, 12, 18, and 24 months
of the study. Neurological examinations were conducted at
approximately 1 year and before termination. No signs of overt
toxicity were observed in any of the dogs. Body weight and food
consumption values were similar for control and treated dogs. No
compound-related effects were observed during the ophthalmoscopic
and physical examinations. Although there were some random
statistically significant differences between the control and
other dose groups in the haematological and biochemical tests,
physiologically significant changes were not observed at any
interval in the study. Two treated and two control animals died
during the study. No compound-related gross or microscopic changes
were observed in the surviving dogs that were sacrificed and
necropsied. Inflammatory, degenerative, and proliferative changes
described were spontaneous in nature, or related to the estrous
phase of the menstrual cycle, and unrelated to compound
administration. On the basis of this study, it has been concluded
that the no-observed-effect level is 40 mg/kg diet (equivalent to
1 mg/kg body weight per day) (IRDC, 1980).
7.5 Mutagenicity
7.5.1 Microorganisms
DNA repair tests in Escherichia coli were conducted at dose
levels of 1250, 2500, or 5000 µg deltamethrin/ml. Deltamethrin was
dissolved in dimethyl sulfoxide (DMSO) and 0.1 ml of the solution
was spread on a plate. Growth inhibition was compared between DNA
repair deficient mutants (p3478 and CM611) and wild types (W3110
and WP2). Partial precipitation of deltamethrin from the solution
occurred when it came into contact with the aqueous bacterial
growth medium. Deltamethrin did not have any damaging effects on
DNA (Peyre et al., 1980).
Deltamethrin was examined for its mutagenic potential in the
Ames test with 5 strains of Salmonella typhimurium (TA 1535, TA
1537, TA 1538, TA 98, and TA100) at doses of 2, 10, 50, 200, 500,
1000, or 5000 µg/plate, with and without S-9 mix (metabolic enzyme
system). It was dissolved in DMSO and precipitated out of solution
at concentrations of 200 µg/plate or more. Deltamethrin did not
have any effect on the mutation rate in any of the strains at any
of the concentrations tested (Peyre et al., 1980).
A similar Ames test was carried out at 0.2, 2, 20, 200, or
400 µg deltamethrin/plate with microsome enzymes. The compounds
did not influence the number of revertants of the 5 strains (same
as above) of S. typhimurium. Again, deltamethrin was dissolved in
DMSO and precipitated out of solution at 200 µg/plate or more
(Fouillet, 1976).
Kavlock et al. (1979) found deltamethrin not to be mutagenic in
2 assays with S. typhimurium at doses of 0 - 1000 µg/plate in DMSO,
with or without metabolic activation. They also obtained negative
results with E. coli at 10 - 1000 µg/plate as well as with
Saccharomyces cerevisiae at concentrations of 1 - 5%, in both cases
with and without metabolic activation. Deltamethrin was found not
to be mutagenic in S. typhimurium strains TA100 and TA98, in the
presence or absence of a rat liver activation system, using the
plate incorporation assay and fluctuation tests. The compound,
dissolved in DMSO, precipitated out of solution at 600 µg/plate
(Pluijmen et al., 1984).
7.5.2 Cultured cells
Deltamethrin, dissolved in a mixture of cremaphor oil and
ethanol (1:1), was applied to a culture of Chinese hamster ovary
cells (CHO) at levels of 0.04, 0.2, 1.0, or 5.0 mmol/litre, with or
without metabolic activation, and examined for chromosomal
aberrations and sister chromatid exchanges (SCE). Because of the
cytotoxic effect of cremaphor oil when combined with S-9 mix or
deltamethrin, no cells would grow in the control dish with
activation, or in the 5 mmol/litre deltamethrin dishes, either with
or without metabolic activation. A high incidence of chromosomal
aberrations and SCEs was observed in the dishes containing 1 mmol
deltamethrin/litre, with activation. However, the absence of
control values (both with and without activation) because of a
broken test tube, made the interpretation equivocal. A second
study was conducted in which deltamethrin was dissolved in DMSO and
applied to the cells at levels of 0.001, 0.01, 0.1, or 0.2
mmol/litre, with or without metabolic activation. In this study,
deltamethrin did not produce any cytotoxic effects and did not
induce either chromosomal aberrations or SCEs in CHO cells.
However, no positive controls were tested and only single plates
were prepared per dose level (Sobels et al., 1978).
Deltamethrin was found not to be mutagenic in V79 Chinese
hamster cells, in the presence or absence of hepatocytes. It is
not known which solvent was used (Pluijmen et al., 1984).
7.5.3 Mouse
An in vivo cytogenetic test was conducted on mice (3 males and
3 females per group). Mice were treated orally with deltamethrin
in sesame oil for 2 consecutive days at 5 or 10 mg/kg body weight.
The incidence of chromosomal aberrations in bone marrow cells or
micronuclei in the polychromatic erythrocytes of treated groups
was, however, comparable to that of the control groups. No
positive controls were tested (Sobels et al., 1978).
Deltamethrin was applied orally, once, at 15 mg/kg body weight
to Swiss mice. A time-related effect on the chromosomes in bone
marrow cells was observed by killing 2 animals every 3 h during
24 h. The report stated that the incidences of chromated
aberrations were low and that there were no consistent time-related
trends in the distribution of the aberrations. However, the time-
related trend of aberrations was not reported. Again, no positive
controls were tested (Sobels et al., 1978).
A dominant-lethal assay with deltamethrin was performed.
Groups of 9 - 13 male mice were dosed orally at 3 mg/kg body weight
in sesame oil for 7 days or at a single dose of 6 or 15 mg/kg body
weight in sesame oil, and mated with 6 - 18 non-treated females.
There were no effects on the rates of pre- and post-implantation
losses, while the positive control, triethylene triphosphoramide
(10 mg/kg body weight), reduced pregnancies in the second and third
weeks after treatment and increased embryonal losses (Vannier &
Glomot, 1977).
Deltamethrin in olive oil was administered orally to female
Swiss mice at single or repeated (5 times at daily intervals) doses
of 1.36, 3.4, or 6.8 mg/kg per day. Bone marrow smears were
prepared 6, 24, or 48 h after treatment. No mutagenic activity was
observed with deltamethrin, whereas the positive control,
cyclophosphamide, induced a positive response (Polàkovà & Vargovà,
1983).
In a micronucleus test, a single dose of deltamethrin in corn
oil was administered orally at 16 mg/kg body weight to Swiss CD-1
mice (5 of each sex per group). No mutagenic activity was observed
with deltamethrin, whereas the positive controls,
triethylenemelamine and dimethylbenzanthracene, both induced
positive responses (Vannier & Fournex, 1983).
7.5.4 Appraisal
Deltamethrin is not mutagenic or clastogenic in a variety of in
vitro and in vivo test systems.
7.6 Teratological and Reproductive Effects
7.6.1 Teratology
7.6.1.1 Mouse
Deltamethrin was dissolved in corn oil and administered by
gastric intubation at doses of 0, 3.0, 6.0, or 12.0 mg/kg body
weight on days 7 - 16 of gestation to groups of CD-1 mice. Mice
were sacrificed on day 18 of gestation. There was a dose-related
( P <0.001) reduction in maternal weight gain during pregnancy and
high-dose females gained 58% less weight than the controls. There
was no dose-related mortality but dams in the high- and mid-dose
groups became convulsive after dosing. Treatment did not affect
the number of implantation sites, fetal mortality, fetal weights,
or the number of sternal and caudal ossification centres.
A significant ( P <0.01) dose-related increase in the
occurrence of supernumerary ribs was observed. No other dose-
related skeletal or visceral anomalies were observed (Kavlock et
al., 1979).
Pregnant female Swiss CD-1 SPF mice (24 per group) were given
deltamethrin dissolved in sesame oil by oral intubation at dose-
levels of 0, 0.1, 1, or 10 mg/kg body weight per day on days
6 - 17 of pregnancy. The animals were necropsied on day 18 of
pregnancy. The numbers of implantation sites, fetal losses, and
viable fetuses were not affected by treatment. There was a dose-
related decrease in mean fetal weight. Apart from delayed
ossification at all dose levels, skeletal examination revealed no
abnormalities. A teratogenic effect was not observed (Glomot &
Vannier, 1977).
In a complementary teratology study, pregnant female Swiss CD-1
mice were given deltamethrin dissolved in sesame oil by oral
intubation at 0, 0.1, 1, or 10 mg/kg body weight per day from day 6
to day 17 of gestation. Females were either sacrificed on day 18
of gestation or allowed to litter for subsequent examination of
pups on days 1 or 21 of lactation. The compound caused a moderate
and transient retardation of development of the fetus at the 1 and
10 mg/kg body weight dose rate, but these effects were not observed
on days 1 or 21 post-partum. There were no teratogenic effects
related to treatment (Vannier & Glomot, 1982).
7.6.1.2 Rat
Pregnant female Sprague-Dawley rats (24 per group) received 0,
0.1, 1, or 10 mg deltamethrin/kg body weight per day by oral
intubation on days 6 - 18 of pregnancy. Apart from 12 females in
the control and 10 mg/kg groups, which were allowed to deliver, the
dams were sacrificed and examined on day 21. There were no effects
on reproduction or on the teratogenic parameters examined, except
for slightly delayed ossification at the highest dose level (Glomot
& Vannier, 1977).
Deltamethrin was dissolved in corn oil and administered by
gastric intubation at doses of 0, 1.25, 2.5, or 5.0 mg/kg body
weight on days 7 - 20 of gestation. Rats were sacrificed on day 21
of gestation. There was a dose-related reduction ( P <0.01) in
maternal weight gain during pregnancy, and dams in the high-dose
group gained only 80% of the control value. Treatment did not
affect the number of implantation sites, fetal mortality, fetal
weight, or the number of sternal and caudal ossification centres
(Kavlock et al., 1979).
7.6.1.3 Rabbit
Groups of 15 pregnant New Zealand White rabbits received
deltamethrin dissolved in sesame oil at levels of 0, 1, 4, or
16 mg/kg body weight per day during days 6 - 19 of pregnancy.
Examination was carried out on day 28 of gestation. The mean fetal
loss was not dose-related. The mean fetal weight in the highest-
dose group was decreased. Some malformations (hydrocephaly,
exencephaly, and thoracogastroschisis) were observed in 2 fetuses
of animals at the highest dose level. In a supplementary study,
pregnant rabbits were similarly dosed with 16 mg/kg body weight per
day; one fetus with spina bifida and shortened tail was detected
among 69 apparently normal fetuses. Malformations were within the
normal limits of the strain used and were not considered to be
related to the treatment, despite the occurrence at the highest
dose level only (Glomot & Vannier, 1977, 1978).
7.6.2 Reproduction studies
Groups of 10 male and 20 female Charles River rats were fed
deltamethrin in the diet at 0, 2, 20, or 50 mg/kg and mated to
begin a 3-generation, 2-litter (first generation, 3 litter)
standard reproduction study. Parental body weights and food
consumption were recorded during the study. After weaning of the
second litter, the surviving parent rats were sacrificed and
necropsied. Five male and 5 female pups of the F3b generation were
necropsied. No changes relevant to treatment were observed in
general behaviour or survival of parent rats or pups. The body
weight of F0 males of the 50 mg/kg group was decreased from week 11
onwards. There were some slight decreases in mean food consumption
of F1 male parent rats in the 50 mg/kg group. The basic
reproduction indices (fertility, gestation, lactation, viability,
and litter size) were not affected by the treatment. However, the
mean pup weight in some litters, especially in the 50 mg/kg group,
was slightly decreased in comparison to the controls on day 21 of
lactation. Gross external examination did not reveal any
abnormalities. No gross or microscopic lesions of treatment-
related significance or significant effects on the organ weights of
the F3b generation were observed (Wrenn et al., 1980).
Deltamethrin was dissolved in corn oil and administered by
gastric intubation at doses of 0, 2.5, or 5.0 mg/kg body weight to
Sprague-Dawley rats from day 7 of gestation to day 15 of lactation.
The dams were allowed to litter and rear their young: litters were
reduced at birth to 4 males and 4 females per litter. The pups
were weighed weekly and examined for the development of eye-
opening, startle reflex, and air-righting. The litters were weaned
on day 22 post-partum and the males discarded. Weekly weighing of
the females continued and at 6 weeks of age they were tested in a
circular open-field. There were no effects on parturition, litter
size, or pup viability. Weights at birth were similar for all
groups, but a dose-related depression in growth was observed during
the pre-weaning period. This early diminution in pre-weaning
weight appeared to have little effect on the morphological and
behavioural parameters measured (Kavlock et al., 1979).
7.7 Neurotoxicity and Behavioural Effects
Adult hens (10 per group) were gavaged with a single dose of 0,
500, 1250, or 5000 mg deltamethrin/kg body weight suspended in corn
oil or 0 or 100 mg/kg body weight dissolved in sesame oil. During
21 days, observations were made on mortality, health, neurotoxic
signs, and body weight. Deltamethrin did not induce any clinical,
macroscopic, or histological signs of delayed neurotoxicity (Ross
et al., 1978).
Groups of 5 male and 5 female Wistar rats were administered
25 mg deltamethrin/kg body weight in 10 mg corn oil/kg on 2
consecutive days. Controls received 10 mg corn oil/kg body weight.
A tilting plane test was performed every second day from day 4 to
day 16 of the study. Two male rats died at 25 mg/kg. No effect
was found on the slip-angle (Davies et al., 1983).
The effects of deltamethrin were studied in a rat performance
test that arranged for milk delivery after every fortieth lever
press. Deltamethrin (1 - 8 mg/kg body weight, given orally, 2 h
before the test) produced both dose-related increases in pause
duration and decreases in response rate. Deltamethrin was also
studied using a conditional flavour-aversion test. Deltamethrin-
treated, trained rats displayed an aversion to saccharin that was
greatest at 2 mg/kg (Macphail, 1981).
The neurological effects of the 4 synthetic pyrethroids,
resmethrin, permethrin, cypermethrin, and deltamethrin, have been
investigated in the rat to establish whether there is a correlation
between the clinical-functional status of the animal and peripheral
nerve damage, as measured biochemically (Rose & Dewar, 1983).
Neuromuscular dysfunction was assessed by means of the inclined
plane test and peripheral nerve damage by reference to
beta-glucuronidase and beta-galactosidase activity increases in
nerve tissue homogenates from treated and control animals. A
transient functional impairment was found in animals treated with
any one of the 4 pyrethroids tested and in all cases this was
greatest at the end of the 7-day dosing regimen (deltamethrin doses
of 5 - 20 mg/kg per day in arachis oil). Significant increases in
beta-glucuronidase and beta-galactosidase activities were found
3 - 4 weeks after the start of dosing, in the distal portion of the
sciatic/posterior tibial nerves from permethrin-, cypermethrin-,
and deltamethrin-treated animals, but no changes were found in
resmethrin-treated animals. It is concluded, therefore, that there
is no direct correlation between the time-course of the
neuromuscular dysfunction and the neurobiochemical changes. This
suggests that these pyrethroids have at least two distinct actions-
a short-term pharmacological effect at near-lethal dose levels and
a more long-term neurotoxic effect that results in sparse axonal
nerve damage.
To better characterize the behavioural toxicity of pyrethroid
insecticides, comparisons were made of the effects of cismethrin
and deltamethrin exposure on motor activity and the acoustic
startle response in male Long-Evans rats (Crofton & Reiter, 1984).
Acute dose-effect, acute time-course, and 30-day repeated-exposure
determinations of 1-h motor activity were made using figure-eight
mazes. The acoustic startle response was measured to a 13-kHz,
120-dB(A), 40-millisecond tone at each of 3 background white noise
levels (50, 65, and 80 dB). Deltamethrin (0, 2, 4, 6, or 8 mg/kg
body weight) or cismethrin (0, 6, 12, 18, or 24 mg/kg) were
administered orally in 0.2 ml/kg corn oil. Cismethrin and
deltamethrin produced similar dose-dependent decreases in motor
activity. The time course of onset and recovery for this decreased
activity was rapid (1 - 4 h). No cumulative effects on motor
activity of a 30-day exposure to 2 mg deltamethrin/kg per day or
6 mg cismethrin/kg per day were found. The effects of cismethrin
and deltamethrin on the acoustic startle response were dissimilar:
deltamethrin produced a dose-dependent decrease in amplitude and an
increase in latency, and cismethrin produced an increase in
amplitude and no change in latency. The differential effects of
cismethrin (Type I pyrethroids) and deltamethrin (Type II
pyrethroids) on the acoustic startle response may be related to the
contrasting effects previously shown with neurophysiological and/or
neurochemical techniques (see Appendix I).
7.8 Miscellaneous Effects
Analgesic effects of deltamethrin for thermic (hot plate test,
60 °C) and mechanical stimuli were investigated in mice and rats,
respectively. Deltamethrin prolonged the response-time to these
tests. Although this action was not significant at 500 mg
deltamethrin/kg body weight given orally, the reaction time was
increased at 1000 and 1500 mg/kg given orally in aqueous suspension
with 10% gum arabic (Chanh et al., 1981).
In rats, treatment with deltamethrin increased mean arterial
pressure and aortic output (Forshaw & Bradbury, 1983). The
cardiovascular effects of deltamethrin were due to both increased
catecholamine release in peripheral vascular beds, and to a direct
positive inotropic effect on the heart.
Krasnjih & Pavlova (1985) demonstrated induction of microsomal
oxygenases in rats 20 h after a single administration of 1/2 LD50.
Daily administration of 1/10th LD50 for 2 months reduced
acetylcholinesterase activity in the serum, erythrocytes, liver,
and cerebrum. It also led to some changes in the aspartate
aminotransferase activity and the urea and protein contents of
serum.
When rats were dosed orally with a single dose of 1/2 LD50 or 3
daily doses of 1/5 LD50 deltamethrin, the activities of transferrin
and ceruloplasmin in plasma, 20 h after dosing, were unchanged.
After the single dose, microsomal monooxygenase activity was
increased by 87%, and after the 3 doses, it was increased by 290%
(Kagan et al., 1986).
7.9 Potentiation
Deltamethrin was hydrolysed in vitro by esterases in blood,
brain, kidney, liver, and stomach preparations of mice.
Pretreatment of mice with the oxidase inhibitor, piperonyl butoxide
(PBO), or the esterase inhibitor, S,S,S-tributylphosphorotrithioate
(DEF), delayed metabolism of intraperitoneally administered
deltamethrin. PBO or DEF made mice more sensitive to deltamethrin
(Ruzo et al., 1979).
Plasma esterases, in addition to hepatic esterases, play a role
in the metabolism of deltamethrin in mammals and cause its rapid
detoxification by the oral route. In a potentiation study, a range
of esterase inhibitors, consisting mainly of organophosphorus
insecticides, was given to male rats in oral doses that inhibited
50% of the plasma cholinesterase. After 15 min, or 2 or 24 h, an
oral LD50 dose of deltamethrin EC formulation was given, which
showed potentiation with azinphos ethyl, omethoate, and dichlorvos.
It appears that users must handle deltamethrin in these
combinations very carefully because of their high toxicity.
Acephate, monocrotophos, phosphamidon, parathion methyl, and the 2
controls did not act as potentiators (Audegond et al., 1988).
7.10 Mechanism of Toxicity (Mode of Action)
Deltamethrin is classified as a Type II pyrethroid. For the
mode of action of pyrethroids in general see Appendix I.
The lowest concentration of deltamethrin to have an effect in
crayfish stretch receptor neurones on sodium channels was 10-12
mol/litre, but the response of the preparation to gamma-
aminobutyric acid (GABA) appeared to be unaffected by concentrations
of deltamethrin up to 10-7 mol/litre. Although 10-6 mol/litre
deltamethrin had a slight effect on the GABA response of the dactyl
abductor muscle, it appears that the majority of the effects of
cyano-pyrethroids in invertebrates could be accounted for solely by
their action on sodium channels (Chalmers et al., 1987).
Pyrethroid-induced motor symptoms, i.e., deltamethrin-induced
writhing and cismethrin-induced tremor, were studied, using a
number of pharmacological agents, in intact conscious rats and
spinal rats. The results suggest that pyrethroid-induced motor
symptoms, i.e., writhing and tremor, are mediated via a spinal site
of action, probably involving interneurones. Deltamethrin-induced
"non-motor" symptoms, i.e., increase in brain blood flow and blood
glucose may result from a supraspinal component of deltamethrin
activity. In contrast, the cardiovascular effects of deltamethrin
are mediated via a peripheral site of action (Bradbury et al.,
1983).
Tissue culture experiments have shown that the dorsal root
ganglion is more sensitive to deltamethrin than the spinal cord or
peripheral nerve fibres. The morphological alterations observed in
the neuronal bodies of the ganglia may reflect some perturbation of
the ionic equilibrium (Na+ and Ca+) (Souyri, 1985).
The results of several other, sometimes very detailed and
specialized, studies on the mode of action of deltamethrin have
been reported. Because these results do not basically influence
the present evaluation of deltamethrin, they are not reviewed in
detail. The interested reader is referred to the following
publications and a review by Bidet et al. (1988): Aldridge et al.
(1978), Duclohier & Georgescauld (1979), Gray et al. (1980),
Jacques et al. (1980), Miller & Adams (1980), Gammon et al. (1981),
Pichon (1981), Brodie & Aldridge (1982), Dyball (1982), Parkin &
LeQuesne (1982), Ray (1982), Staatz et al. (1982), Brodie (1983),
Takahashi & LeQuesne (1983), Berlin et al. (1984), Prasada Rao et
al. (1984), Bloomquist & Soderlund (1985), Brodie (1985), Brodie &
Opacka (1985), Bloomquist et al. (1986), Chinn & Narahashi (1986),
Doherty et al. (1986), Forshaw & Ray (1986), Staatz-Benson & Hosko
(1986), Brooks & Clark (1987), Forshaw et al. (1987), Leibowitz et
al. (1987), Lummis et al. (1987), Stein et al. (1987).
7.11 Experimental Studies on Antidotes
Treatments capable of counteracting acute deltamethrin
poisoning have been investigated in experimental animals. Ray &
Cremer (1979) have proposed atropine, Gammon et al. (1982),
phenobarbital and diazepam, and Bradbury et al. (1981, 1983),
mephenesin.
In order to find a therapeutically usable antagonist,
pharmacological screening was carried out by Dumont (1978), Dumont
& Chifflot (1978), and Dumont & Laurent (1979). The outcome was
that barbiturates are therapeutically active, but that the most
efficient product is ethyl carbamate. Cotonat et al. (1987) and
Fournier (1988) have confirmed that ethyl carbamate is an effective
treatment for severe deltamethrin poisoning. However, a serious
drawback is its antimitotic activity. Leclercq et al. (1986) began
by evaluating the activity of common anticonvulsants, such as
diazepam and clomethiazole. These products exhibited satisfactory
activity in rats and dogs (Thiebault et al., 1985, 1988).
Phenoprobamate and mephenesin carbamate have been shown to be
effective in the experimental treatment of deltamethrin poisoning
(Cotonat et al., 1987; Leclercq et al., 1986). A summary of the
results of the antidote studies can be found in Bleys et al.
(1986). Clinical trials based on these studies will be undertaken
(unpublished information given to the IPCS by Roussel Uclaf).
The therapeutic effects of methocarbamol have also been shown
by Hiromori et al. (1986). However, the mechanism underlying this
activity is not clear as the actual anticonvulsant activity is
weak.
It appears that for the time being barbiturates, and especially
diazepam, offer the safest symptomatic treatment in case of
deltamethrin poisoning. Advice on treatment for deltamethrin
poisoning is given in the IPCS Deltamethrin health and safety guide
(WHO, 1989).
8. EFFECTS ON MAN
8.1 General Population-Poisoning Incidents
A few cases of attempted suicides with deltamethrin
formulations (mainly EC), all non-fatal, have been reported in
anti-poison centres. Two typical cases are described below.
The first poisoning case in a 13-year-old girl who ingested
voluntarily 200 ml of a 2.5% EC formulation, (5 grams of
deltamethrin) was described by Rousselin (1983). After an unknown
time, she lost consciousness and developed generalized muscle
cramps, myosis, and tachycardia. Treatment in hospital was as
follows: gastric lavage, PAM 0.5 mg; atropine 2 mg; sodium
nitrite, 3% sodium thiosulfate; and, lastly, high doses of
diazepam. She completely recovered in 48 h.
A second poisoning case, concerning another attempted suicide
by a 23-year-old man, was reported by Foulhoux (1988). After oral
absorption of 70 cc of a 2.5% EC formulation (1.75 g pure
deltamethrin), there were no neurological signs in this patient.
Digestive and hepatic signs occurred, probably due to absorption of
the solvent, since determination of xylene in plasma was positive.
The patient was treated with haemodialysis, phenobarbital,
lidocaine, and provoked alkaline diuresis. Recovery followed
within 48 h.
8.2 Occupational Exposure
8.2.1 Acute toxicity-poisoning incidents
Rousselin (1983) described a case of poisoning in an
agricultural worker as a result of skin contamination with a liquid
containing 5 g deltamethrin/litre. He developed paraesthesia in
the legs, mouth, and tongue, and diarrhoea. Following washing of
the skin and administration of antihistamines, he still had
tingling sensations in his toes after 24 h, but was fully recovered
after 48 h.
Outbreaks of acute deltamethrin and fenvalerate poisoning
occurred in cotton growers in China in 1982 - 84. The farmers
handled the pyrethroid insecticides without taking any precautions.
Skin sensations occurred in more than 90% of the exposed workers.
After repeated spraying in the cotton fields, the mild cases
presented severe headaches, dizziness, fatigue, nausea, and
anorexia, with transient changes in the EEG. A severe case
developed muscular fasciculation, repetitive discharges in the EMG,
and frequent convulsions, which were treated with diazepam and
phenobarbital. However, in follow-up studies, all workers were
found to have made complete recovery, and the prognosis of acute
pyrethroid poisoning was found to be good (He, 1987; Tong Ying,
1988).
More recently He et al. (1989) reviewed 573 cases of acute
pyrethroid poisoning reported in the Chinese medical literature
during 1983 - 88. Among these there were 325 cases of acute
deltamethrin poisoning: 158 occupational, due to inappropriate
handling, and 167 accidental, mostly due to ingestion. Two
patients died of convulsions. All others recovered with
symptomatic and supportive treatment within 1 - 6 days. Clinical
manifestations are well reviewed (He, 1987).
8.2.2 Effects of short- and long-term exposure
Among plant workers dermally exposed to technical deltamethrin
or its formulations, cutaneous and mucuous manifestations were
observed. Initial lesions were tenacious and painful pruritus,
especially observed after exposure to hot water or perspiration,
followed by a blotchy local burning sensation with blotchy erythema
for about 2 days. Thereafter, slight and regular desquamation,
restricted to the contaminated area, occurred. Cutaneous signs
were sometimes accompanied by itching of the face (mainly around
the mouth) and/or rhinorrhoea or lachrymation (Husson, 1978).
Apart from the above-mentioned effects, no long-term or
persistent effect, or allergic diseases were reported in 70
workers, who had been exposed from 1977 - 87 in a deltamethrin-
manufacturing and -formulating plant in France (unpublished
Roussel Uclaf information supplied to the IPCS, 1988).
A field study was carried out in the United Kingdom with three
unprotected operators and one operator wearing hood, gloves, and
respirator, all of whom were involved in orchard spraying with
deltamethrin according to normal field practice. The exposure time
was 3.5 h. No changes were found in blood cell counts, total
protein, urea, alkaline phosphates, gamma-GT and SGOT in blood.
Little deltamethrin was found in the respirator pad and no residues
were found in the urine. There was no decrease in nerve conduction
velocity, but a slight tendency to the opposite reaction. None of
the operators experienced facial sensations (Hewson & Burgess, 1981).
Four operators were evaluated by the same authors during normal
field applications of deltamethrin lasting one day. Three of the
operators did not wear any protection on their heads or hands,
while one wore hood, gloves, and a respirator. Motor and sensory
nerve conduction velocities were determined as well as
haematological and biochemical parameters and urinalysis. No
changes were observed in the blood parameters measured and no
residues were found in the urine samples. Furthermore, nerve
conduction velocity did not decrease. Residues were primarily
confined to gloves and legs. None of the operators experienced
facial sensations (Hewson & Burgess, 1981).
Persons exposed to deltamethrin for 7 - 8 years in production
and formulation were subjected to clinical and haematological
examinations. Evaluations were conducted at several plants. There
were no measurable effects other than transient irritation of
cutaneous and mucous membranes, which was without sequelae.
Adequate precautionary measures, such as the wearing of gloves and
face masks, provided protection from exposure (Foulhoux, 1981).
A medical survey of agricultural workers involved in the use
and application of EC and WP formulations of deltamethrin in
Yugoslavia revealed no untoward symptoms of exposure, other than
itching and burning of the face, and nasal hypersecretion. Medical
examinations included chest X-ray, ECG, liver function tests,
neurological examinations (eye tonometry, Goldman perimetry, dark
adaptation ability), kidney function tests, and whole blood and
plasma cholinesterase activity. No adverse effects were noted.
The need for the proper use of masks and gloves, as well as good
personal hygiene (e.g., washing), was emphasized (Plestina, 1981).
Five healthy volunteers, 16 - 40 years of age, were exposed to
deltamethrin during 5 days of spraying in a cotton field in India
in 1981. A sixth volunteer was engaged in mixing and loading the
emulsion during the same period. Spraymen were exposed for 7 h
daily. No one complained about any symptoms. No clinical
abnormalities were detected, particularly with respect to
neurological examination (muscle power, coordination, tremors,
reflexes, and both light and deep sensations). No cardiovascular,
respiratory, or abdominal abnormalities were detected, and no skin,
mucous membrane, or eye lesions were observed during, and after
cessation of, exposure (Trivedi, 1981).
A health survey was carried out among spraymen exposed to 2.5%
deltamethrin emulsifiable concentrate in cotton fields in China.
The subjects were exposed to deltamethrin at concentrations of
0.022 - 24.070 µg/m3 in the air of the respiratory zone and
0.013 - 0.347 µg/cm2 of skin contact. One half of the 44 sprayers
complained of itching and burning sensations on their faces. A few
miliary red papules also appeared on the face of one of them, but
no signs of acute deltamethrin poisoning were noticed during
physical examination. There were no significant differences in the
sodium, potassium, and urea contents of the serum, the sodium,
potassium, ATPase, and serotonin contents of whole blood, and the
levels of 3-methyl-4-hydroxymandelic acid and 5-hydroxy-
indoleacetic acid in the urine between the subjects examined and
the controls. Deltamethrin in the urine of spraymen was below the
detection limit of 0.10 µg/litre (Wang et al., 1988).
Mestres et al. (1985) measured the dermal and inhalation
exposure of mixer/applicators who applied deltamethrin to vegetable
crops in greenhouses in southern France and of workers who picked
fruit from treated trees in the same area. This appeared to be
less than 0.0065% of the toxic dose per hour with a mask and
0.0017%, without a mask.
In a department producing an aerosol of the domestic Bulgarian
insecticide "Dekazol" containing 0.02, 0.04, or 0.08% deltamethrin,
severe subjective complaints of sensory irritation were found
because of the high levels of contamination of the workplace air
with deltamethrin and also dermal contamination. Skin irritation
with conjunctivitis and irritation of the respiratory system were
discovered in all 25 workers. Two of them had contact urticaria.
Patch testing with 0.03% deltamethrin showed a positive reaction in
5 out of 23 workers tested (Bainova et al., 1986).
8.3 Clinical Studies
Three formulations of deltamethrin in petroleum solvent were
patch tested on 37 human volunteers (double blind trial against
solvent control). A dose of 20 µl of a 1% suspension in water, of
a 25 g/litre emulsifiable concentrate was put on the facial skin of
each volunteer, with a randomized distribution of control and
active dilution. The duration of the irritation was short (from
some minutes to 1 h) and the severity was described as slight by
most of the volunteers. No skin damage was reported (Foulhoux et
al., 1981).
9. PREVIOUS EVALUATIONS BY INTERNATIONAL BODIES
The Joint FAO/WHO Meeting on Pesticide Residues (JMPR)
discussed and evaluated deltamethrin at its meetings in 1980, 1981,
1982, 1984, 1985, 1986, 1987, and 1988 (FAO/WHO, 1981, 1982, 1983,
1985a, 1986a, 1986b, 1988a,b,c). In 1982, an acceptable daily
intake (ADI) of 0 - 0.01 mg/kg body weight was established.
The following Maximum Residue Limits (MRLs), in mg/kg, resulted
from these evaluations:
tea 10.0
hops dry, wheat bran unprocesseda 5.0
coffee beans (post-harvest) 2.0
wheat wholemeal,a cereal grains,a 1.0
(ph) lentil (dry),a beans (dry),a
field pea (dry)a
straw and fodder (dry) of cereal 0.5
grains, legume animal feeds (dry
weight), leafy vegetables
brassica leafy vegetables,a edible 0.2
peel of fruiting vegetablesa
bulb vegetables, edible peel of 0.1
assorted fruits, legume vegetables,
oilseeds, pome fruits, wheat floura
artichokes, bananas, clementines, 0.05
coco beans, grapes, kiwi fruit,
oranges (sweet, sour), stone
fruits/strawberries
legume oilseeds, melons, 0.01
mushrooms, pineapples, root and
tuber vegetables, milksa
WHO has classified deltamethrin as a moderately hazardous
technical product in normal use (WHO, 1988). A Data Sheet on
deltamethrin (No. 50) has been issued (WHO/FAO, 1984).
-------------------------------------------------------------------
a Not yet confirmed by Codex Alimentarius Commission (FAO/WHO,
1986c, and 1988c).
REFERENCES
ADENEY, R.J., GRZYWACZ, D., & MATTHIESSEN P. (1980) The acute
toxicities of pyrethrin analogues to Sarotherodon mossambicus
(Peters), Centre for Overseas Pest Research, 6 pp. (Unpublished
proprietary data submitted to WHO by Roussel Uclaf).
AKHTAR, M.H. (1984) Metabolism of deltamethrin by cow and chicken
liver enzyme preparations. J. agric. food Chem., 32: 258-262.
AKHTAR, M.H., HAMILTON, R.M.G., & TRENHOLM, H.L. (1985)
Metabolism, distribution and excretion of deltamethrin by leghorn
hens. J. agric. food Chem., 33: 610-617.
AKHTAR, M.H., MARTIN, K.E., & TRENHOLM, H.L. (1986) Fate of
14C-deltamethrin in lactating dairy cows. J. agric. food Chem.,
34: 753-758.
AKHTAR, M.H., DANIS, C., TRENHOLM, H.L., & MARTIN, K.E. (1987)
Residues in milk and tissues of lactating dairy cows fed
deltamethrin for 28 consecutive days. J. agric. food Chem.,
ALDRIDGE, W.N., CLOTHIER, B., FORSHAW, P., & JOHNSON, M.K., PARKER,
V.H., PRICE, R.J., SKILLETER, D.N., VERSCHOYLE, R.D., STEVENS,
C. (1978) The effect of DDT and the pyrethroids cismethrin and
decamethrin on the acetyl choline and cyclic nucleotide content of
rat brain. Biochem. Pharmacol., 27: 1703-1706.
ALINIAZEE, M.T. & CRANHAM, J.E. (1980) Effects of four synthetic
pyrethroids on a predatory mite, Typhlodroums pyri, and its prey,
Panonychus ulmi, on apples in southeast England. Environ. Entomol.,
9: 436-439.
ARZONE, A. & VIDANO, C. (1978) [Azione sull'ape di etiofencaus,
decamethrin e ciexatin.] Apic. mod., 69: 157-162, (in Italian).
ATKINS, E.L., KELLUM D., & NEUMAN, K.J. (1976) Effect of
pesticides on apiculture project No. 1449: The annual report,
University of California Riverside, pp. 536-567.
AUDEGOND, L., CATEZ, D., FOULHOUX, P., FOURNEX, R., LE RUMEUR, C.,
L'HOTELLIER, M., & STEPNIEWSKI, J.P. (1988) Potentialisation de
la toxicité de la deltaméthrine par les insecticides
organophosphorés (Unpublished proprietary data submitted to WHO by
Roussel Uclaf).
AUDEGOND L., COLLAS E., & GLOMOT R. (1981) RU 22974
(deltamethrin) single administration study by oral route in the rat
(the compound is given as a suspension) (Unpublished report RU-
81239/A, submitted to WHO by Roussel Uclaf).
BAINOVA, A. & KALOYANOVA, F. (1985) [Study of allergenic and
irritating effect of synthetic pyrethroids on skin.] Hig. Zdrav.,
28(2): 19-21 (in Bulgarian).
BAINOVA, A., MIHAVSKI, M., ISMIROVA, N., & BENCHEV, V. (1986)
Specific skin irritation after contact with specific pyrethroids.
6th Congress of Bulgarian Dermatologists, Varna, 2-5 October, 1986,
(Abstract No. 74) (in Bulgarian).
BAKER, P.G. & BOTTOMLEY, P. (1982) Determination of residues of
synthetic pyrethroids in fruit and vegetables by gas-liquid and
high-performance liquid chromatography. Analyst, 107: 206-212.
BALDRY, D.A.T., EVERTS, J., ROMAN, B., BOON VON OCHSSEE, G.A., &
LAVEISSIERE, C. (1981) The experimental application of
insecticides from a helicopter for the control of riverine
populations of Glossina tachinoides in West Africa. Part VIII: The
effects of two spray applications of OMS-570 (endosulfan) and of
OMS 1998 (decamethrin) on G. tachinoides and non-target organisms
in Upper Volta. Trop. Pest Manage., 27(1): 83-110.
BEAVERS, J.B. & FINK, R. (1977a) Actue oral LD50 Mallard duck
technical DECIS final report, (Unpublished report WI77.06.06/A,
submitted to WHO by Roussel Uclaf). Wildlife International.
BERLIN, J.R., AKERA, T., BRODY, T.M., & MATSUMURA, F. (1984) The
inotropic effects of a synthetic pyrethroid decamethrin on isolated
guinea pig atrial muscle. Eur. J. Pharmacol., 98: 313-322.
BIDET, D., DUMONT, C., FOULHOUX, P., FOURNEX, R., & ROUGERON, C.
(1988) Mechanisms responsible for the toxicity of deltamethrin and
other pyrethroids. (Unpublished document, submitted to WHO by
Roussel Uclaf).
BLEYS, M., COTONAT, J., FOULHOUX, P. (1986) Lettre à l'éditeur,
J. Toxicol. clin. exp., 6(3): 211-212.
BLOOMQUIST, J.R., SODERLUND, D.M. (1985) Neurotoxic insecticides
inhibit GABA-dependent chloride uptake by mouse brain vesicles.
Biochem. biophys. Res. Commun., 133(1): 37-43.
BLOOMQUIST, J.R., ADAMS, P.M., & SODERLUND, D.M. (1986)
Inhibition of gamma-aminobutyric acid-stimulated chloride flux in
mouse brain vesicles by polychlorocycloalkane and pyrethroid
insecticides. Neurotoxicology, 7(3): 11-20.
BOCQUET, J.C., PASTRE, P., ROA, L., & BAUMEISTER, R. (1980) Etude
de l'action de la deltaméthrine sur Apis mellifera en conditions de
plein-champ. Phytiatr. Phytopharm., 29: 83-92.
BOCQUET, J.C., PASTRE, P., & BAUMEISTER, R. (1983) Bilan de cinq
années d'études de l'effet de la deltaméthrine sur abeilles en
conditions naturelles. 6ème Congrès International du Colza, Paris,
mai 1983.
BOUCHE, M.B. & FAYOLLE, L. (1979) Tests carried out with Procida
compound Decis EC 2.5 on earthworms lethal effect in relation with
time (Unpublished proprietary report No. INRA-VT-79.05.22/78.10.
10/A, submitted to WHO by Roussel Uclaf).
BOWMAN, H. & CARPENTER, M. (1987) Determination of
photodegradation of 14C-deltamethrin in aqueous solution
(Unpublished proprietary report ABC LABS 35491, submitted to WHO by
Roussel Uclaf).
BRADBURY, J.E., GRAY, A.J., & FORSHAW, P. (1981) Protection
against pyrethroid toxicity in rats with mephenesin. Toxicol.
appl. Pharmacol., 60: 382.
BRADBURY, J.E., FORSHAW, P.J., GRAY, A.J., & RAY, D.E. (1983) The
action of mephenesin and other agents on the effects produced by
two neurotoxic pyrethroids in the intact and spinal rat.
Neuropharmacology, 22(7): 907-914.
BRODIE, M.E. (1983) Correlations between cerebellar cyclic GMP
and motor effects induced by deltamethrin: independence of olivo-
cerebellar tract. Neurotoxicology, 4(4): 1-11.
BRODIE, M.E. (1985) Deltamethrin infusion into different sites in
the neuraxis of freely moving rats. Neurobehav. Toxicol. Teratol.,
7(1): 51-55.
BRODIE, M.E. & ALDRIDGE, W.N. (1982) Elevated cerebellar cyclic
GMP levels during the deltamethrin-induced motor syndrome.
Neurobehav. Toxicol. Teratol., 4: 109-113.
BRODIE, M.E. & OPACKA, J. (1985) Dissociation between circling
behaviour and striatal dopamine activity following unilateral
deltamethrin administration to rats. Naunyn-Schmiedeberg's Arch.
Pharmacol., 331: 341-346.
BROOKS, M.W. & CLARK, J.M. (1987) Enhancement of norepinephrine
release from rat brain synaptosomes by alpha cyano pyrethroids.
Pestic. Biochem. Physiol., 28: 127-139.
BUCCAFUSCO, R.J., ELLS, S.J., & CARY, G.A. (1977a) Acute toxicity
of NRDC 161 to bluegill (Lepomis macrochirus) under dynamic test
conditions, E.G. & G. Bionomics Aquatic Toxicology Laboratory, 12 pp.
(Unpublished proprietary data submitted to WHO by Roussel Uclaf).
BUCCAFUSCO R.J., ELLS, S.J., & CARY, G.A. (1977b) Acute toxicity
of NRDC 161 (Decamethrine) to Channel Catfish (Ictalurus
punctatus), E.G. & G. Bionomics Aquatic Toxicology Laboratory,
7 pp. (Unpublished proprietary data supplied to WHO by Roussel
Uclaf).
CABRAL, J.R.P., GALENDO, D., LAVAL, M., & LYANDRUT, N. (1986)
Carcinogenicity study of the pesticide deltamethrin in mice and
rats. Summary Report in Poster Session at IUPAC Meeting, Ottawa,
August 1986.
CARY, G.A. (1978) Kinetics of 14C-NRDC-161 in a model aquatic
ecosystem, E.G. & G. Bionomics Aquatic Toxicology Laboratory.
(Unpublished proprietary report BW-78-2-075, submitted to WHO by
Roussel Uclaf).
CHALMERS, A.E., MILLER, T.A, & OLSEN, R.W. (1987) Deltamethrin:
a neurophysiological study of the sites of action. Pestic.
Biochem. Physiol., 27: 36-41.
CHAMBON, A. & LEPAILLEUR, H. (1984) Etude de l'effet toxique de
la deltamethrine (produit technique > 98%) et de la formulation CE
25 g/l Decis vis-à-vis de l'espece de vers (Eisenia fetida andrei)
(Unpublished proprietary report IRCHA-84.30.07/F, submitted to WHO
by Roussel Uclaf).
CHANH, P.H., NAVARRO-DELMASURE, C., CHANH, A.P.H., LEAN, C.S.,
ZIADE, F., & SAMAHA, F. (1981) Analgesic effects of decamethrin,
Surg. Transplant., 9: 503-504.
CHAPMAN, R.A. & HARRIS, C.R. (1981) Persistence of four
pyrethroid insecticides in a mineral and organic soil. J. environ.
Sci. Health, B16: 605-615.
CHAPMAN, R.A., TU, C.M., HARRIS, C.R., & COLE, C. (1981)
Persistence of five pyrethroid insecticides in sterile and natural,
mineral and organic soil. Bull. Environ. Contam. Toxicol, 26:
513-519.
CHESTERMAN, H., HEYWOOD, R., PERKIN, C.J., BEARD, D., STREET, E., &
PRENTICE, D.E. (1977) RU 22974. Oral toxicity study in Beagle
dogs, Huntingdon, Huntingdon Research Centre, (Unpublished report
RSL 253/7751/A3, submitted to WHO by Roussel Uclaf).
CHINN, K. & NARAHASHI, T. (1986) Stabilization of sodium channel
states by deltamethrin in mouse neuroblastoma cells. J. Physiol.,
380: 191-207.
CLAIR, M. (1977) RU 22974 DECIS. Acute toxicity in the rabbit by
percutaneous administration. Joinville-le-Pont, Institut Français
de Recherches et Essais Biologique (Unpublished report IFREB-R
770257.1/A, submitted to WHO by Roussel Uclaf).
CLARK, G.C., JACKSON, G.C., & ALEXANDER, D.J. (1980) Acute
inhalation toxicity in rats, 4-hour exposure (Decis PM 2.5
percent), Huntingdon Research Centre, (Unpublished report RSL
437/80568, submitted to WHO by Roussel Uclaf).
COOMBS, D.W. & CLARK, G.C. (1978) RU 22974: Acute inhalation
toxicity in rats. 6 Hour LC50. Huntingdon, Huntingdon Research
Centre, (Unpublished report RSL 310/78453/A, submitted to WHO by
Roussel Uclaf).
COOMBS, D.W., CLARK, G.C., STREET, A.E., & GIBSON, W.A. (1978) RU
22974 inhalation toxicity study in rats 14 x 6 hour exposures over
a period of 3 weeks (Unpublished proprietary report
RSL/318/78638/A, submitted to WHO by Roussel Uclaf).
COQUET, B. (1976a) RU 22974. Test to determine primary cutaneous
irritation in the rabbit. Joinville-le-Pont, Institut Francais de
Recherches et Essais Biologiques (Unpublished report IFREB-R
761157/A, submitted to WHO by Roussel Uclaf).
COQUET, B. (1976b) RU 22974. Test to evaluate ocular irritation
in the rabbit, Joinville-le-Pont, Institut Français de Recherches
et Essais Biologiques (Unpublished report IFREB- R 761158/A,
submitted to WHO by Roussel Uclaf).
COQUET, B. (1977) RU 22974. Decis formulations - Determination of
the LD50 in the rat by oral administration, Joinville-le-Pont,
Institut Français de Recherches et Essais Biologiques (Unpublished
report 770257-A, submitted to WHO by Roussel Uclaf).
COTONAT, J., BLEYS, M., & FOULHOUX, P. (1987) Effets antagonistes
du phenprobamate et du carbamate de mephenesine sur l'intoxication
à la deltaméthrine. J. Toxicol. clin. exp., 7: 5-19.
CROFTON, K.M. & REITER, L.W. (1984) Effects of two pyrethroid
insecticides on motor activity and the acoustic startle response in
the rat. Toxicol. appl. Pharmacol., 75: 318-328
DAVID, D. (1981) Laboratory evaluation of repellent properties
against birds of the synthetic pyrethroid decamethrin. Poult. Sci.,
60: 1149-1151.
DAVIES, J.E., MUNT, P.L., & GOOR, J.L. (1983) RU 22974.
Investigation of possible neurological effects using the tilting
plane test. Huntingdon, Huntingdon Research Centre (Unpublished
proprietary report RSL603/83232/A, submitted to WHO by Roussel
Uclaf).
DE LAVAUR, E., LE SECH, J., & GROLLEAU, G. (1985) Accumulation et
élimination de la deltaméthrine chez la caille japonaise. Ann.
Fals. Exp. Chim., 835: 73-78.
DOHERTY, J.D., LAUTER, C.J., & SALEM, N., Jr (1986) Synaptic
effects of the synthetic pyrethroid resmethrin in rat brain in
vitro. Comp. Biochem. Physiol., C84(2): 373-9.
DUCLOHIER, H. & GEORGESCAULD, D. (1979) The effects of the
insecticide decamethrin on action potential and voltage-clamp
currents of myxicola giant axon. Comp. Biochem. Physiol., C62(2):
217-223.
DUMONT, C. & CHIFFLOT, L. (1978) Recherche d'un antidote contre
les effets toxiques aigus (Unpublished report RU-AE-72, submitted
to WHO by Roussel Uclaf).
DUMONT, C. & LAURENT, J. (1979) Recherche d'antagonistes des
effets neurotoxiques aigus du RU 22 974 (Unpublished report RU-AG-
35, submitted to WHO by Roussel Uclaf).
DUNNING, R.A., COOPER, J.M., WARDMAN, J.M., & WINDER, G. (1981)
Susceptibility of the carabid Pterostichus melanarius (Illiger) to
aphicide sprays applied to the sugar-beet crop. (Unpublished
proprietary report UK-SOIL-F.81/A, submitted to WHO by Roussel
Uclaf).
DYBALL, R.E.J. (1982) Inhibition by decamethrin and resmethrin of
hormone release from the isolated rat neurohypophysis - a model
mammalian neurosecretory system. Pestic. Biochem. Physiol., 17:
42-47.
ELLIOTT, M. (1977) Synthetic pyrethroids, Washington, DC,
American Chemical Society, p. 229 (ACS Symposium Series 42).
ELLIOTT, M., FARNHAN, A.W., JANES, N.F., NEEDHAM, P.H., & PULMAN,
D.A. (1974) Synthetic insecticide with a new order of activity.
Nature, 248: 710-711.
ESTESEN, B.J., BUCH, N.A., & WARE, G.W. (1979) Dislodgeable
insecticide residue on cotton foliage; permethrin, curacron,
fenvalerate, sulprofos, decis and endosulfan. Bull. environ.
Contam. Toxicol., 22: 245-248.
EVANS, M.H. (1976) End-plate potentials in frog muscle exposed to
a synthetic pyrethroid. Pestic. Biochem. Physiol., 6: 547-550.
EVERTS, J.W., VAN FRANKENHUYZEN, K., ROMAN, B., & KOEMAN, J.H.
(1983) Side effects of experimental pyrethroid applications for
the control of tsetse flies in a riverine forest habitat in Africa.
Arch. environ. Contam. Toxicol., 12: 91-97.
EVERTS, J.W., KORTENHOFF, B.A., HOOGLAND, H., VLUG, H.J., JACQUE,
R., & KOEMAN. J.H. (1985) Effects on man-target terrestrial
arthropods of synthetic pyrethroids asked for the control of the
Tsetse Fly ( Glossina spp.) in settlement areas of the Southern
Ivory Coast, Africa. Arch. environ. Contam. Toxicol., 14: 647-650.
FAO (1982) Second Government Consultation on International
Harmonization of Pesticide Registration Requirements, Rome, 11-15
October, 1982, Rome, Food and Agriculture Organization of the
United Nations.
FAO/WHO (1981) 1980 Evaluations of some pesticide residues in
food, Rome, Food and Agriculture Organization of the United Nations
(FAO Plant Production and Protection Paper No. 26 Sup).
FAO/WHO (1982) 198l Evaluations of some pesticide residues in
food, Rome, Food and Agriculture Organization of the United Nations
(FAO Plant Production and Protection Paper No. 42).
FAO/WHO (1983) 1982 Evaluations of some pesticide residues in
food, Rome, Food and Agriculture Organization of the United Nations
(FAO Plant Production and Protection Paper No. 49).
FAO/WHO (1985a) 1984 Evaluations of some pesticide residues in
food, Rome, Food and Agriculture Organization of the United Nations
(FAO Plant Production and Protection Paper No. 67).
FAO/WHO (1985b) Guide to Codex recommendations concerning
pesticide residues. Part 8. Recommendations for methods of
analysis of pesticide residues, 3rd ed., Rome, Food and Agriculture
Organization of the United Nations, Codex Committee on Pesticide
Residues.
FAO/WHO (1986a) 1985 Evaluations of some pesticide residues in
food. Part I - Residues, Rome, Food and Agriculture Organization
of the United Nations (FAO Plant Production and Protection Paper
No. 72/1).
FAO/WHO (1986b) 1986 Evaluations of some pesticide residues in
food. Part I - Residues, Rome, Food and Agriculture Organization
of the United Nations (FAO Plant Production and Protection Paper
No. 78).
FAO/WHO (1986c) Codex maximum limits for pesticide residues, 2nd
ed., Rome, Food and Agriculture Organization of the United Nations,
Codex Alimentarius Commission, CAC XIII.
FAO/WHO, (1988a) 1987 Evaluations of some pesticide residues in
food. Part I - Residues, Rome, Food and Agriculture Organization
of the United Nations (FAO Plant Production and Protection Paper
No. 86/1).
FAO/WHO, (1988b) 1988 Evaluations of some pesticide residues in
food. Part 1 - residues, Rome, Food and Agriculture Organization
of the United Nations. (FAO Plant Production and Protection Paper
93/1.
FAO/WHO, (1988c) Supplement 1 to Codex maximum limits for pesticide
residues, Rome, Food and Agriculture Organization of the United
Nations.
FISCHER, L. & CHAMBON, J.P. (1987) Faunistical inventory of
cereal arthropods after flowering and incidence of insecticide
treatments with deltamethrin, dimethoate and phosalone on the
apigeal fauna. Meded. Fac. Landbouwwet. Rijksuniv. Gent, 52(2a):
FLANNIGAN, S.A. & TUCKER, S.B. (1985) Variation in cutaneous
sensation between synthetic pyrethroid insecticides. Contact
Dermatitis, 13: 140-147.
FLORELLI, F., GARNIER, P., & ROA, L. (1987a) Bilan de 8 années
d'expérimentation sur la selectivite du Decis vis à vis des
abeilles - La défense des végétaux, 243: 8
FLORELLI, F., HELLER, J.J., GARNIER, P., & BAUMEISTER, R. (1987b)
Incidence sur les abeilles, les ruches et leur production, de
traitements aériens du colza avec la deltaméthrine - Annales ANPP -
Conférence Internationale sur les Ravageurs en Agriculture, Paris,
1-3 décembre 1987, 189-203.
FORSHAW, P.J. & BRADBURY, J.E. (1983) Pharmacological effects of
pyrethroids on the cardiovascular system of the rat. Eur. J.
Pharmacol., 91: 207-213.
FORSHAW, P.J. & RAY, D.E. (1986) The effects of two pyrethroids,
cismethrin and deltamethrin, on skeletal muscle and the trigeminal
reflex system in the rat. Pestic. Biochem. Physiol., 25: 143-151.
FORSHAW, P.J., LISTER, T., & RAV, D.E. (1987) The effects of two
types of pyrethroid on rat skeletal muscle. Eur. J. Pharmacol.,
134(1): 89-96.
FOUILLET, X. (1976) RU 22974 Mutagenicity study of various
preparations. Salmonella microsome test, Institut Français de
Recherches et Essais Biologiques (Unpublished report no. IFREB-R
761153/A, submitted to WHO by Roussel Uclaf).
FOULHOUX, P. (1981) Medical observations of personnel working on
synthesis or formulation of deltamethrin (Unpublished report RU-
81.18.06/A, submitted to WHO by Roussel Uclaf).
FOULHOUX, P. (1988) Assessment of potential adverse effects of
deltamethrin on man, Département Central de Toxicovigilance Roussel
Uclaf, 16 pp. (Unpublished report submitted to WHO by Roussel
Uclaf).
FOULHOUX, P., BOGGIN, CORNILLIET, POTTIER, RAYNAUD, STERN-VEYRIN, &
SUTTET (1981) Cutaneous irritation patch test on human volunteers
with Decis EC25, Decis flowable 25, Decis + mineral oil 25
(Unpublished report RU-81.03.02/DM/A, submitted to WHO by Roussel
Uclaf).
FOURNIER, P.E. (1988) Etude de l'activité antidote du carbamate
d'éthyl dans l'intoxication aiguë par la deltaméthrine chez le rat,
Paris, Unité de Pharmacologie Clinique, Hospital Fernand Widal
(Unpublished report submitted to WHO by Roussel Uclaf).
GAINES, T.B. & LINDER, R.E. (1986) Acute toxicity of pesticides
in adult and weanling rats, Fundam. appl. Toxicol., 7: 299-308.
GAMMON, D.W. & CASIDA, J.E. (1983) Pyrethroids of the most potent
class antagonize GABA action at the crayfish neuromuscular
junction. Neurosci. Lett., 40: 163-168.
GAMMON, D.W., BROWN, M.A., & CASIDA, J.E. (1981) Two classes of
pyrethroid action in the cockroach. Pestic. Biochem. Physiol., 15:
181-191.
GAMMON, D.W., LAWRENCE, L.J., & CASIDA, J.E. (1982) Pyrethroid
toxicology: Protective effects of diazepam and phenobarbital in
the mouse and the cockroach. Toxicol. appl. Pharmacol., 66:
290-296.
GLICKMAN, A.H. & CASIDA, J.E. (1982) Species and structural
variations affecting pyrethroid neurotoxicity. Neurobehav.
Toxicol. Teratol., 4(6): 793-799.
GLOMOT, R. (1979) Acute toxicity study by oral route in the rat
(Proprietary report RU-79803-54/A2, submitted to WHO by Roussel
Uclaf).
GLOMOT, R. & CHEVALIER, B. (1976a) RU 22974. Acute toxicity
study mouse and rat by oral route (Unpublished report TOX 76810/A,
submitted to WHO by Roussel Uclaf).
GLOMOT, R. & CHEVALIER, B. (1976b) RU 22974. Acute toxicity
study mouse and rat by intraperitoneal route (Unpublished report
TOX 76811/A, submitted to WHO by Roussel Uclaf).
GLOMOT, R. & CHEVALIER, B. (1976c) RU 22974. Acute toxicity
study mouse and rat by intravenous route (Unpublished report TOX
76812/A, submitted to WHO by Roussel Uclaf).
GLOMOT, R. & VANNIER, B. (1977) RU 22974. Teratological study in
mouse, rat and rabbit (Unpublished report TOX 76534-76536/2/A,
submitted to WHO by Roussel Uclaf).
GLOMOT, R. & VANNIER, B. (1978) RU 22974. Teratological study in
rabbit. Complementary information (Unpublished report TOX 76534-
76536/A3, submitted to WHO by Roussel Uclaf).
GLOMOT, R., AUDEGOND, L., & COLLAS, E. (1979) Acute oral toxicity
study in the rat (Decis 25 G/L Mixofluid) (Proprietary report RU
79824/A, submitted to WHO by Roussel Uclaf).
GLOMOT, R., AUDEGOND, L., & COLLAS, E. (1980a) Single
administration by oral route in the mouse (Decis Wettable Powder,
2.5%) (Proprietary report RU-80817/A, submitted to WHO by Roussel
Uclaf).
GLOMOT, R., AUDEGOND, L., & COLLAS, E. (1980b) Single
administration study by oral route in the dog (Decis Wettable
Powder, 2.5%) (Proprietary report RU 80194/A, submitted to WHO by
Roussel Uclaf).
GLOMOT, R., AUDEGOND, L., & COLLAS, E. (1981a) Single
administration study by oral route in the rat (Proprietary report
RU-81239/A, submitted WHO by Roussel Uclaf).
GLOMOT, R., AUDEGOND, L., & COLLAS, E. (1981b) Primary dermal
irritation study in the rabbit. Deltamethrin 25 G/L Mixofluid
(Proprietary report RU-79193/A, submitted to WHO by Roussel Uclaf).
GLOMOT, R., AUDEGOND, L., & COLLAS, E. (1981c) Primary dermal
irritation study in the rabbit (Deltamethrin wettable powder, 2.5%)
(Proprietary report RU-80200/A, submitted to WHO by Roussel Uclaf).
GLOMOT, R., AUDEGOND, L., & COLLAS, E. (1981d) Primary eye
irritation study in the rabbit (Deltamethrin in 25 G/L Mixofluid)
(Proprietary report RU-79192/A, submitted to WHO by Roussel Uclaf).
GLOMOT, R., AUDEGOND, L., & COLLAS., E. (1981e) Primary eye
irritation study in the rabbit (Decis Wettable Powder, 2.5%)
(Proprietary report RU-79192/A, submitted WHO by Roussel Uclaf).
GLOMOT, R., CHEVALIER, B., COLLAS, E., & AUDEGOND, L. (1977) RU
22974. Acute toxicity study by oral route in male and female
Beagle dogs (Unpublished report TOX 77804/JL-5, submitted to WHO
by Roussel Uclaf).
GOLDENTHAL, E.I., BLAIR, M., JEFFERSON, N.D., SPICER, E.J.F.,
ARCEO, R.J., & KAHN, A., III (1980a) RU 22974. Two year toxicity
and carcinogenicity study in mice, Mattawan, Michigan,
International Research and Development Corporation (Unpublished
report IRDC 406-001/A-4, submitted to WHO by Roussel Uclaf).
GOLDENTHAL, E.I., JEFFERSON, N.D., BLAIR, M., THORSTENSON, J.H.,
SPICER, E.J.F., ARCEO, R.J., & KAHN, A., III (1980b) RU 22974.
Two year oral toxicity and carcinogenicity study in rats,
Mattawan, Michigan, International Research and Development
Corporation (Unpublished report IRDC 406-002/A.4, submitted to WHO
by Roussel Uclaf).
GRANDADAM, A. (1976) Test to determine the toxicity in the
chicken by oral route (Unpublished report RU76.05.05/A, submitted
to WHO by Roussel Uclaf).
GRAY, A.J. & RICKARD, J. (1982) The toxicokinetics of
deltamethrin in rats after intravenous administration of a toxic
dose. Pestic. Biochem. Physiol.., 18: 205-215.
GRAY, A.J., CONNORS, T.A., & RICKARD, J. (1980) Mechanism of
mammalian toxicity of the pyrethroids. In: Littaner, U.Z. et al.,
ed. Neurotransmitters and their receptors, New York, John Wiley
and Sons, pp. 565-568.
GROLLEAU G. & GRIBAN, J. (1976a) Toxicity of deltamethrin or
DECIS in single ingestion in game duck Anas platyrhnchos L.,
JoyenJossas, France, Institut National de la Recherche Agronomique
(Unpublished report INRA-76.21 12/A, submitted to WHO by Roussel
Uclaf).
GROLLEAU, G. & GRIBAN, J. (1976b) Toxicity of deltamethrin or
DECIS by single ingestion in grey partridge, Perdix Perdix L. and
red partridge, Alectotis rufa L., JoyenJossas, France, Institut
National de la Recherche Agronomique (Unpublished report INRA76.28
09/A, submitted to WHO by Roussel Uclaf).
GUILLOT, J.P. & GUILAINE, J. (1977) RU 22974. Decaméthrine.
Decis technical Roussel Uclaf. Sensitization test in the guinea
pig. Joinville-le-Pont, Institut Français de Recherches et Essais
Biologique (Unpublished report no. IFREB-R 709241/A, submitted to
WHO by Roussel Uclaf).
GULYAS P. & CSANYI B. (undated) Acute fish toxicological
investigation of diverse insecticides, Hungary, Scientific Research
Centre of Economy of Water Supplies, 21 pp. (Unpublished
proprietary data supplied to WHO by Roussel Uclaf).
HALLS, G.R.H. & PERIAM, A.W. (1980) The fate of residues of NRDC
161 on wheat during storage and after milling and baking - Report
after 9 months storage, November 1980, Wellcome Research
Laboratories Report (Unpublished report HEFH 80-4, submitted to
WHO by Roussel Uclaf).
HARGREAVES, J.R. & COOPER, L.P. (1979) Phytotoxicity tests with
pyrethroid insecticides on glasshouse grown tomato seedlings.
Queensland J. Agric. anim. Sci., 36: 151-154.
HASCOET, M. (1977) Laboratory leaching soil study with
decamethrin (RU 22974), Versailles, Institut National de la
Recherche Agronomique (Unpublished proprietary report INRA-
77.20.09/A, submitted to WHO by Roussel Uclaf).
HE, F. (1987) Occupational neurotoxicology: Current problems and
trends. Paper presented at the XXII International Congress on
Occupational Health, Sydney, 27 September-2 October, 1987.
HE, F., WANG, S., LIU, L., CHEN, S., ZHANG, Z., & SUN, J. (1989)
Clinical manifestations and diagnosis of acute pyrethroid
poisoning. Arch. Toxicol., 63: 54-58.
HEHEITMULLER, T., SHUBA, P.J., & PARRISH, R. (1978) Acute
toxicity of Decis to sheepshead minnows (Cyprinodon variegatus),
E.G. & G. Bionomics Marine Laboratory, 7 pp. (Unpublished
proprietary report BR-78-12219, supplied to WHO by Roussel Uclaf).
HEWSON, R.T. & BURGESS, J.E. (1981) An operator study with
deltamethrin including measurements of nerve conduction times
(Unpublished proprietary report GB-NT-11.81A, submitted to WHO by
Roussel Uclaf).
HILL, B.D. (1982) Determination of Deltamethrin residues in
soils, Alberta, Agriculture Canada Research Station, 7 pp
(Unpublished proprietary report CAN-82.12.10/A, submitted to WHO by
Roussel Uclaf).
HILL, B.D. (1983) Persistence of deltamethrin in a Lethbridge
sandy clay loam. J. environ. Sci. Health, B18(6): 691-703.
HILL, B.D. & JOHNSON, D.L. (1987) Persistence of deltamethrin and
its isomers on pasture forage and litter. J. agric. Food Chem.,
35: 373-378.
HILL, B.D. & SCHAALJE, G.B. (1985) A two-compartment model for
the dissipation of deltamethrin on soil. J. agric. food Chem., 33:
1001-1006.
HIROMORI, T., NAKANISHI, T., KAWAGUCHI, S., SAKO, H., SUZUKI, T., &
KIYAMOTO, J. (1986) Therapeutic effects of methocarbamol on acute
intoxication by pyrethroids in rats. J. pestic. Sci., 11: 9-14.
HUNTER, B., JORDAN, J., HEYWOOD, R., HEPWORTH, P., STREET, A.E., &
PRENTICE, D.E. (1977) RU 22974. Assessment of toxicity to rats
by oral administration for 13 weeks (followed by a 4-week
withdrawal period) (Unpublished report RSL-254/76938/A3, submitted
to WHO by Roussel Uclaf).
HUSSON, J.M. (1978) Medical observations made on people working
on the manufacture and formulation of the pyrethroid insecticide
decamethrin (Unpublished report RU 78.25.08/A, submitted to WHO by
Roussel Uclaf).
IRDC (1980) Two year chronic dog feeding study, Matawan,
Michigan, International Research and Development Corporation
(Unpublished proprietary report IRDC-406-004/AI, submitted to WHO
by Roussel Uclaf).
JACKSON, G. & HARDY, C. (1986) RU 22974. Acute inhalation
toxicity in rats 1-hour exposure (Unpublished report RSL-DTM-
851457/A, submitted to WHO by Roussel Uclaf).
JACQUES, Y., ROMEY, G., CAVEY, M.T., KARTAALOVSKI, B., & LAZDUNSKI,
M. (1980) Interaction of pyrethroids with the sodium channel in
mammalian neuronal cells in culture. Biochem. Biophys. Acta, 600:
882-897.
KAGAN, YU. S., PANSHINA, T.N., & SASINOVICH, L.M. (1986)
Biochemical effects of toxic action of synthetic pyrethroids. Gig.
i Sanit., 1: 7-9.
KAUFMAN, D.D. & KAYSER, A.J. (1979a) Degradation of 14C-phenoxy-
and 14C-cyano-decamethrin in soil (Unpublished proprietary report
USDA-I-23.04.79/A2, submitted to WHO by Roussel Uclaf).
KAUFMAN, D.D. & KAYSER, A.J. (1979b) The effect of soil
temperature on the degradation of 14C-cyano-decamethrin in soil
(Unpublished proprietary report USDA-II-24.04.79/A2, submitted to
WHO by Roussel Uclaf).
KAUFMAN, D.D. & KAYSER, A.J. (1980) Degradation of 14C-cyano-,
14C-phenoxy-, and 14C-vinyl-decamethrin in flooded soil
(Unpublished proprietary report USDA-III-12.05.80/A, submitted to
WHO by Roussel Uclaf).
KAUFMAN, D.D., RUSSEL, B.A., HELLING, C.S., & KAYSER, A.J. (1981)
Movement of cypermethrin, decamethrin, permethrin and their
degradation products in soil. J. agric. food Chem., 29: 239-245.
KAVLOCK, R., CHERNOFF, N., BARON, R., LINDER, R., ROGERS, E.,
CARVER, B., DILLEY, J., & SIMMON, V. (1979) Toxicity studies with
decamethrin, a synthetic pyrethroid insecticide. J. environ.
Pathol. Toxicol., 2: 751-765.
KNAUF, W. & HORLEIN, G. (1979 The acute toxicity of decamethrine
to the Rainbow trout Salmo gairdneri, Richardson, Frankfurt,
Hoechst AG, 19 pp (Unpublished proprietary report 19/79, submitted
to WHO by Roussel Uclaf).
KNAUF, W. & SCHULZE, E.F. (1977a) Effect of decamethrine on
Cyprinus carpio, Frankfurt, Hoechst AG, 9 pp (Unpublished
proprietary report 15/77, submitted to WHO by Roussel Uclaf).
KNAUF, W. & SCHULZE, E.F. (1977b) The effect of decamethrin
6E0660 on Catfish (Ictalurus nebulosus) in a static test,
Frankfurt, Hoechst Ag, 18 pp (Unpublished proprietary report
32/79, submitted to WHO by Roussel Uclaf).
KRASNJIH, A. & PAVLOVA, L. (1985) Harmful effects of
deltamethrin. In: Levina, E., ed. Harmful substances in industry,
Moscow, Khimija Publishing House, p. 154.
KYNOCH, S.R., LLOYD, G.K., & ANDREWS, C.D. (1979) Acute
percutaneous toxicity to rats of decamethrin, Huntingdon,
Huntingdon Research Centre (Unpublished report RSL-1009 8/D,
submitted to WHO by Roussel Uclaf).
LAWRENCE, L.J. & CASIDA, J.E. (1982) Pyrethroid toxicology: Mouse
intracerebral structure-toxicity relationship. Pestic. Biochem.
Physiol., 18: 9-14.
LAWRENCE, L.J. & CASIDA, J.E. (1983) Stereospecific action of
pyrethroid insecticides on the gamma-aminobutyric acid receptor-
ionophore complex. Science, 221: 1399-1401.
LAWRENCE, L.J., GEE, K.W., & YAMAMURA, H.I. (1985) Interactions
of pyrethroid insecticides with chloride ionophore-associated
binding sites. Neurotoxicology, 6: 87-98.
LEAHEY, J.P. (1985) The pyrethroid insecticide, London, Taylor &
Francis Ltd., p. 440.
LECLERCQ, M., COTONAT, J., & FOULHOUX, P. (1986) Recherche d'un
antagoniste à l'intoxication par la deltaméthrine. J. Toxicol.
clin. exp., 6, 83-85.
LEIBOWITZ, M.D., SCHWARZ, J.R., HOLAN, G., & HILLE, B. (1987)
Electrophysiological comparison of insecticide and alkaloid
agonists of Na channels. J. gen. Physiol., 90(1): 75-93.
LEPAILLEUR, H. & CHAMBON, A. (1984) Etude de la toxicité létale
du produit technique deltaméthrine vis à vis du poisson-zebré, 5 pp
(Unpublished proprietary dated report IRCHA B.7827, submitted to
WHO by Roussel Uclaf).
LHOSTE, J., FRANCOIS, Y., & RUPAUD, Y. (1979) Ichtyotoxicité de
la decaméthrine vis à vis de Salmo trutta L. (poisson téléostéen)
en fonction de l'age et des conditions expérimentales - Congrès sur
la lutte contre les insectes en milieu tropical, Marseilles - March
13-16 1979, pp. 885-902.
L'HOTELLIER, M. & VINCENT, P. (1986) Assessment of the impact of
deltamethrin on aquatic species. In British Crop Protection
Conference - Pests and Disease: 1109-1116.
LOUVEAUX, J., MISSONNIER, J., & MESQUIDA, J. (1977) Tests de
toxicité sur abeille domestique avec le Decis CE (Unpublished
proprietary report submitted to WHO by Roussel Uclaf).
LUMMIS, S.C., CHOW, S.C., HOLAN, G., & JOHNSTON, G.A. (1987)
Gamma-aminobutyric acid receptor ionophore complexes: differential
effects of deltamethrin, dichlorodiphenyltrichloroethane, and some
novel insecticides in a rat brain membrane preparation. J.
Neurochem., 48(3): 689-94.
LUND, A.E. & NARAHASHI, T. (1983) Kinetics of sodium channel
modification as the basis for the variation in the nerve membrane
effects of pyrethroids and DDT analogs. Pestic. Biochem. Physiol.,
20: 203-216.
MACPHAIL, R.C. (1981) Behavioral effects of a synthetic
insecticide (decamethrin). Fed. Proc. Fed. Am. Soc. Exp. Biol.,
40: 678.
MEISTER, R.T., BERG, G.L., SINE, C., MEISTER, S., & POPLYK, J.
(1983) Farm chemicals handbook. Section C. Pesticide dictionary,
Willoughby, Ohio, Meister Publishing Co.
MESTRES, R., CHEVALLIER, C., ESPNOZA, C., & CORNET, R. (1978a)
Dosage des résidues de decaméthrine dans les produits végétaux.
Trav. Soc. Pharm. Montpellier, 38: 183-192.
MESTRES R., CHEVALLIER C., & ESPINOZA, C. (1978b) Analytical
method for decamethrine residue analysis, Montpellier, Faculte de
Pharmacie, 2 pp. (Proprietary report FP-78.08.07/A, dated 7/8/1978
submitted to WHO by Roussel Uclaf).
MESTRES, R., FRANCOIS, C., CAUSSE C., VIAN L., & WINNETT, G.
(1985) Survey of exposure to pesticides in greenhouses. Bull.
environ. Contam. Toxicol., 35: 750-756.
MESTRES, R., ESPINOZA, C., & CHEVALLIER, C. (1986) Effets sur les
résidus de deltaméthrine de la transformation des produits
agricoles en vue de leur consommation. Med. Nutr., XXII (3):
181-184.
MILLER, T.A. & ADAMS, M.E. (1980) Neural and behavioral
correlates of pyrethroid and DDT-type poisoning in the housefly,
Musca domestica. Pestic. Biochem. Physiol., 13: 137-147.
MIYAMOTO, J. (1976) Degradation, metabolism and toxicity of
synthetic pyrethroids. Environ. Health Perspect., 14: 15-28.
MIYAMOTO, J. (1981) The chemistry, metabolism and residue
analysis of synthetic pyrethroids. Pure appl. Chem., 53: 1967-2022.
MIYAMOTO, J. & KEARNEY, P.C. (1983) Pesticide chemistry - Human
welfare and the environment. Proceedings of the Fifth International
Congress of Pesticide Chemistry, Kyoto, Japan, 29 August - 4
September, 1982, Oxford, Pergamon Press, Vol. 1-4.
MOHSEN, Z.H. & MULLA, M.S. (1981) Toxicity of blackfly larvicidal
formulations to some aquatic insects in the laboratory. Bull.
environ. Contam. Toxicol., 26: 696-703.
MOUROT, D., DELEPINE, B., BOISSEAU, J., & GAYOT, G. (1979) High-
performance liquid chromatography of decamethrin. J. Chromatogr.,
173: 412-414.
MUIR, D.C.G., RAWN, G.P., & GRIFT, N.P., (1985) Fate of the
pyrethroid insecticide deltamethrin in small ponds: A mass balance
study. J. agric. food Chem., 33: 603-609.
MULLA, M.S., NAVVAB GOJRATI, H.A., & DARWAZEH, H.A. (1978)
Toxicity of mosquito larvicidal pyrethroids to four species of
freshwater fishes. Environ. Entomol.., 7(3): 428-430.
NETO, P.X.R., RODRIGUES, I.C.F., & FILHO, A.M (1983) Etude sur
les effets secondaires de la deltaméthrine sur l'ichtyofaune de la
region du projet "Rio Formoso" (Unpublished report submitted to WHO
by Roussel Uclaf).
NOBLE, R.M., HAMILTON, D.J., & OSBORNE, W.J. (1982) Stability of
pyrethroids on wheat in storage. Pestic. Sci., 13: 246-252.
PANSHINA T.N. & SASINOVICH, L.M. (1983) Toxicology of synthetic
pyrethroids. Khim. sel'sk. khoz., 12: 51-53.
PANSU, M., DHOUIBI, M.H., & PINTA, M. (1981) Determination of
traces of biopermethrine and decamethrine pyrethrinoids in
biological substrates by gas chromatography. Analysis, 9: 55-59.
PAPALEXIOU, Ph., BITAR, N., & STOCKIS, A. (1984) Absorption of
radiocarbon labelled deltamethrin given orally in healthy
volunteers, Charleroi, Belgium, Institut de Pharmacologie et
d'Investigations Biomédicales (Unpublished proprietary report
50/22, submitted to WHO by Roussel Uclaf).
PARKIN, P.J. & LEQUESNE, P.M. (1982) Effects of a synthetic
pyrethroid deltamethrin on excitability changes following a nerve
impulse. J. Neurol. Neurosurg. Psychiatry., 45: 337-342.
PEYRE, M., CHANTOT, J.F., GLOMOT, R., & PENASSE, L. (1980)
Detection of a mutagenic potency of decamethrin (RU 22974), in
bacterial tests (Unpublished report RU/TOX/80-2101/A, submitted to
WHO by Roussel Uclaf).
PHAM, H.C., NAVARRO-DALMASURE, C., PHAM, H.C., CLAVEL, P., VAN
HAVERBEKE, G., CHEAV, S.L. (1984) Toxicological studies of
deltamethrin. Int. J. Tissue React., 6(2): 127-33.
PICHON, Y. (1981) Pharmacological characterization of ionic
channels in unmyelinated axons. J. Physiol., 17(9): 1119-1128.
PLAPP, F.W., Jr & BULL, D.L. (1978) Toxicity and selectivity of
some insecticides to Chrysopa carnea, a predator of the tobacco
budworm. Environ. Entomol., 7: 431-434.
PLESTINA, R. (1981) An evaluation of the use of deltamethrin in
public health (Unpublished report ZAG. KOT. 81/A, submitted to WHO
by Roussel Uclaf).
PLUIJMEN, M., DREVON, C., MONTESANO, R., MALAVEILLE C.,
HAUTEFEUILLE, A., & BARTSCH, H. (1984) Lack of mutagenicity of
synthetic pyrethroids in Salmonella typhimurium strains and in V79
Chinese hamster cells. Mutat. Res., 137: 7-15.
POLAKOVA, H. & VARGOVA, M. (1983) Evaluation of the mutagenic
effects of decamethrin: cytogenetic analysis of bone marrow. Mutat.
Res., 120: 167-171.
POTTIER J., CHATELET, P., & JOUQUEY, S. (1982) Resorption
percutanée de la deltaméthrine - Etude d'un modèle animal, 10 pp.
(Unpublished proprietary report AN.34, submitted to WHO by Roussel
Uclaf).
PRASADA RAO, K.S., CHETTY, C.S., & DESAIAH, D. (1984) In vitro
effects of pyrethroids on rat brain and liver ATPase activities. J
Toxicol. environ. Health, 14(2-3): 257-65.
RAWN, G.P., MUIR, D.O.G., & GRIFT, P.G. (1985) Fate of the
pyrethroid insecticide deltamethrin in small ponds, a mass balance
study. J. agr. food Chem., 33: 603-609.
RAY, D.E. (1980) An EEG investigation of decamethrin-induced
choreathetosis in the rat. Exp. Brain Res., 38: 221-227.
RAY, D.E. (1982) Changes in brain blood flow associated with
deltamethrin-induced choreoathetosis in the rat. Exp. Brain Res.,
45: 269-276.
RAY, D.E. & CREMER, J.E. (1979) The action of decamethrin (a
synthetic pyrethroid) on the rat. Pestic. Biochem. Physiol., 10:
333-340.
RICHTER, W.R. & GOLDENTHAL, E.I. (1983) Two-year oral toxicity
and carcinogenicity study in rats. Amendment to the final report.
Matawan, Michinga, International Research and Development
Corporation (Unpublished proprietary report IRDC 406.002/A5,
submitted to WHO by Roussel Uclaf).
RICKARD, J. & BRODIE, M.E. (1985) Correlation of blood and brain
levels of the neurotoxic pyrethroid deltamethrin with the onset of
symptoms in rats. Pestic. Biochem. Physiol., 23: 143-156.
RICOU (1978) Assessment of side effects of S 276-B (Decis EC
25 g/l) towards grey slugs (Agriolimax spp.) (Unpublished
proprietary report INRA-LIM.78.10/A, submitted to WHO by Roussel
Uclaf).
ROSE, G.P. & DEWAR, A.J. (1983) Intoxication with four synthetic
pyrethroids fail to show any correlation between neuromuscular
dysfunction and neurobiochemical abnormalities in rats. Arch.
Toxicol., 53: 297-316.
ROSE, D.B., ROBERTS, N.L., CAMERON, M.McD., PRENTICE, D.E., COOKE,
L., & GIBSON, W.A. (1978) RU 22974. (Decamethrine) LD50
determination and assessment of neurotoxicity in the domestic hen,
Huntingdon, Huntingdon Research Centre (Unpublished report RSL 293-
NT/7830/A, submitted to WHO by Roussel Uclaf).
ROUSSELIN, X. (1983) Toxicité des dérivés du pyrèthre, Paris,
Faculté de Médecine Lariboisiere-Saint-Louis (Thèse de l'Université
Paris VII).
RUIGT, G.S.F. & VAN DEN BERCKEN, J. (1986) Action of pyrethroids
on a nerve-muscle preparation of the clawed frog, Xenopus laevis.
Pestic. Biochem. Physiol., 25: 176-187.
RUZO, L.O. & CASIDA, J.E. (1979) Degradation of decamethrin on
cotton plants. J. agric. food Chem., 27: 572-575.
RUZO, L.O., HOLMSTEAD, R.L., & CASIDA, J.E. (1976) Solution
photochemistry of the potent pyrethroid insecticide alpha-cyano-3-
phenoxy-benzyl cis-2,2-dimethyl-3-(2,2-dibromovinyl)
cyclopropanecarboxylate. Tetrahedron Lett., 3045-3048.
RUZO, L.O., HOLMSTEAD, R.L., & CASIDA, J.E. (1977) Pyrethroid
photochemistry: decamethrin. J. agric. food Chem., 25: 1385-1394.
RUZO, L.O., UNAI, T., & CASIDA, J.E. (1978) Decamethrin metabolism
in rats. J. agric. food Chem., 26: 918-925.
RUZO, L.O., ENGEL, J.L., & CASIDA, J.E. (1979) Decamethrin
metabolites from oxidative, hydrolytic and conjugative reactions in
mice. J. agric. food Chem., 27: 725-731.
SANTOSA, K. (1983) Toxicity of Decis 2.5 EC (Decamethrin) to
fish, Jakarta, Department of Agriculture Research and Development
Inland Fisheries Institute, 4 pp. (Unpublished proprietary data
submitted to WHO by Roussel Uclaf).
SANTOSA, K. & HADI, H. (1980) Trial results pesticide lethal
toxicity to fish, Department of Agriculture Research and
Development Inland Fisheries Institute, Jakarta, Indonesia -
Report no. 174/UI/80 dated 5 December 1980, 3 pp. (Unpublished
proprietary data submitted to WHO by Roussel Uclaf).
SMIES, M., EVERS, R.H.J., REIJNENBURG, P.H.M., & KOEMAN, J.H.
(1980) Environmental aspects of field trials with pyrethroids to
eradicate tsetse fly in Nigeria. Ecotoxicol. environ. Saf., 4:
114-128.
SOBELS, F.H., TATES, A.D., & VANNIER, B. (1978) Cytogenetic study
with RU 22974. Detection of a mutagenic potency in mammalian
cells, Leiden, The Netherlands, State University of Leiden
(Unpublished report ULN-782211/A, submitted to WHO by Roussel
Uclaf).
SODERLUND, D.M. & CASIDA, J.E. (1977) Effects of pyrethroid
structure on rates of hydrolysis and oxidation by mouse liver
microsomal enzymes. Pestic. Biochem. Physiol., 7: 391-401.
SOUYRI, F. (1985) Autoradiographic studies of (3H)-deltamethrin
binding with nerve tissue cultures. Pestic. Sci., 6(6): 701-703.
STAATZ, C.G., BLOOM, A.S., & LECH, J.J. (1982) Effects of
pyrethroids on [3H]-kainic acid binding to mouse forebrain
membranes. Toxicol. appl. Pharmacol., 64: 566-569.
STAATZ-BENSON, C.G. & HOSKO, M.J. (1986) Interaction of
pyrethroids with mammalian spinal neurons. Pestic. Biochem.
Physiol., 25: 19-30.
STEIN, E.A., WASHBURN, M., WALCZAK, C., & BLOOM, A.S. (1987)
Effects of pyrethroid insecticides on operant responding maintained
by food. Neurotoxicol. Teratol., 9(1): 27-31.
STEVENSON, J.H., NEEDHAM, P.H., & WALKER, J. (1978) Poisoning of
honey bee by pesticides: investigations of the changing pattern in
Britain over 20 years. Rothamsted Exp. Stn Rep., Part 2: 55-72.
TAKAHASHI, M. & LEQUESNE, P. ((1982) The effects of the
pyrethroids deltamethrin and cismethrin on nerve excitability in
rats. J. Neurol. Neurosurg. Psychiatry, 45: 1005-1011.
TAKKEN, W., BALK, F., JANSEN, R.C., & KOEMAN, J.H. (1978) The
experimental application of insecticides from a helicopter for the
control of riverine populations of Glossina tachinoides in West
Africa. VI. Observations on side effects. P.A.N.S., 24: 455-466.
THIEBAULT, J.J., BOST, J., & FOULHOUX, P. (1985) Intoxication
expérimentale par la deltaméthrine chez le chien et son traitement.
Toxicol. vét. Collect. Méd. lég. Toxicol. méd., 131, 47-62.
THIEBAULT, J.J., BOST, J., FOULHOUX, P., DEVAUX, P., & TILLIER, C.
(in press) Intravenous deltamethrin intoxication of dogs.
Therapeutic attempts. Veterinary and Human Toxicology.
THIER, W. & SCHMIDT, D. (1976) Laboratory leaching studies of
Decis EC 25 in three different German soils (Unpublished
proprietary report RL-77.25.05/A, submitted to WHO by Roussel
Uclaf).
THIER, W. & SCHMIDT, D. (1977) The behaviour of the pesticide in
the soil: Decamethrin, (Unpublished proprietary report A09232,
submitted to WHO by Roussel Uclaf) (in German).
TONG YING (1988) Clinical manifestations of 211 cases intoxicated
by deltamethrin and fenvalerate (Unpublished report submitted to
WHO by Roussel Uclaf).
TOOBY, T.E., THOMSON, A.N., RYCROFT, R.J., BLACK, I.A., & HEWSON,
R.T. (1981) A pond study to investigate the effects on fish and
aquatic invertebrates of deltamethrin applied directly onto water,
Fisheries Laboratory, Ministry of Agriculture, Fisheries and Food
(Unpublished report AEP-81.30.09A, submitted to WHO by Roussel
Uclaf).
TRIVEDI, K. (1981) Spray worker exposure and health survey during
decamethrin (decis) spray in the field, Sevagram, India, Mahatma
Gandhi Institute of Medical Sciences (Unpublished information
submitted to WHO by Roussel Uclaf).
TU, C.M. (1980) Influence of five pyrethroid insecticides on
microbial populations and activities in soil. Microb. Ecol., 5:
321-327.
VAN DEN BERCKEN, J. (l977) The action of allethrin on the
peripheral nervous system of the frog. Pestic. Sci., 8: 692-699.
VAN DEN BERCKEN, J. & VIJVERBERG, H.P.M. (1980) Voltage clamp
studies on the effects of allethrin and DDT on the sodium channels
in frog myelinated nerve membrane. In: Insect neurobiology and
pesticide action (Neutox 79), London, Society of Chemical Industry,
Vol. 2, pp. 79-85.
VAN DEN BERCKEN, J., AKKERMANS, L.M.A., & VAN DER ZALM, J.M.
(1973) DDT-like action of allethrin in the sensory nervous system
of Xenopus laevis. Eur. J. Pharmacol., 21: 95-106.
VAN DEN BERCKEN, J., KROESE, A.B.A., & AKKERMANS, L.M.A. (1979)
Effect of insecticides on the sensory nervous system. In:
Narahashi, T., ed. Neurotoxicology of insecticides and pheromones,
New York, London, Plenum Press, pp. 183-210.
VANNIER, B. & FOURNEX, R. (1983) Deltamethrin detection of a
mutagenic potency/micronucleus test in the mouse (Proprietary
unpublished report RU-83607/A, submitted to WHO by Roussel Uclaf).
VANNIER, B. & GLOMOT, R. (1977) RU 22974. Mutagenic study.
Dominant lethal assay in the male mouse (Unpublished report TOX
76533/DB9/A, submitted to WHO by Roussel Uclaf).
VANNIER, B. & GLOMOT, R. (1982) RU 22974 complementary
teratological study in the mouse (Unpublished report RU-82506-12/A,
submitted to WHO by Roussel Uclaf).
VARANKA, I. (1987) Effect of mosquito killer insecticides on
freshwater mussels. Comp. Biochem. Physiol. C. Comp. Pharmacol.,
86: 157-162.
VAYSSE, M., GIUDICELLI, J.C., DEVAUX, P., & L'HOTELLIER, M. (1984)
Decis, in Zweig analytical methods for pesticides and plant growth
regulators. Vol XIII, 3: 53-68, New York, Academic Press.
VERSCHOYLE, R.D. & ALDRIDGE, W.N. (1980) Structure-activity
relationship of some pyrethroids in rats. Arch. Toxicol., 45:
325-329.
VICKERMAN, G.P., COOMBES D.S., TURNER, G., MEAD-BRIGGS, M.A., &
EDWARDS, J. (1987a) The effects of pirimicarb, dimethoate and
deltamethrin on carabidae and staphylinidae in winter wheat. Meded.
Fac. Landbouwwet. Rijksuniv. Gent, 52(2a):
VICKERMAN, G.P., COOMBES, D.S., TURNER, G., MEAD-BRIGGS, M.A., &
EDWARDS, J. (1987b) The effects of pirimicarb, dimethoate and
deltamethrin on non-target arthropods in winter wheat.
International Conference on Pests in Agriculture, Paris, 1-3
December 1987, Annales ANPP, pp. 67-74.
VIJVERBERG, H.P.M., VAN DEN BECKEN, J. (1982) Action of
pyrethroid insecticides on the vertebrate nervous system.
Neuropathol. appl. Neurobiol., 8: 421-440.
VIJVERBERG, H.P.M. & VAN DEN BERCKEN, J. (1979) Frequency
dependent effects of the pyrethroid insecticide decamethrin in frog
myelinated nerve fibres. Eur. J. Pharmacol., 58: 501-504.
VIJVERBERG, H.P.M., RUIGT, G.S.F., & VAN DEN BERCKEN, J. (1982a)
Structure-related effects of pyrethroid insecticides on the
lateral-line sense organ and on peripheral nerves of the clawed
frog, Xenopus laevis. Pestic. Biochem. Physiol., 18: 315-324.
VIJVERBERG, H.P.M., VAN DER ZALM, J.M., & VAN DEN BERCKEN, J.
(1982b) Similar mode of action of pyrethroids and DDT on sodium
channel gating in myelinated nerves. Nature, (Lond.) 295: 601-603.
VIJVERBERG, H.P.M., VAN DER ZALM, J.M., VAN KLEEF, R.G.D.M., & VAN
DEN BERCKEN, J. (1983) Temperature and structure-dependent
interaction of pyrethroids with the sodium channels in frog node of
Ranvier. Biochim. Biophys. Acta, 728: 73-82.
WALTERSDORFER & SCHULZE E.F. (1976a) Decis 2.5. Effect on
Lebistes reticulatus, Francfort, Hoechst Ag, 8 pp. (Unpublished
proprietary report 42/76, submitted to WHO by Roussel Uclaf).
WALTERSDORFER & SCHULZE E.F. (1976b) Decis 2.5. Effect on Idus
melanotus, Frankfurt, Hoechst AG, 5 pp (Unpublished proprietary
report 33/76, submitted to WHO by Roussel Uclaf).
WALTERSDORFER & SCHULZE E.F. (1976c) Decis 2.5. Effect on Salmo
gairdneri (rainbow trout), Frankfurt, Hoechst AG, 7 pp (Unpublished
proprietary report 35/76, submitted to WHO by Roussel Uclaf).
WALTERSDORFER & SCHULZE E.F. (1976d) Decamethrine effect on
Lepomis gibbosus (Bluegill sunfish), Frankfurt, Hoechst AG, 6 pp.
(Unpublished proprietary report 16/77, submitted to WHO by Roussel
Uclaf).
WANG, S., ZHENG, Q., YU, L., XU, B., & LIX., Y. (1988) Health
survey among farmers exposed to deltamethrin in the cotton fields.
Ecotoxicol. environ. Saf., 15: 1-6.
WELLCOME FOUNDATION (1979) Residues in cow's milk resulting from
intrarumenal, and later, dermal administration of 14C-labelled
decamethrin (Unpublished report submitted to WHO by Roussel Uclaf).
WHO (1979) Safe use of pesticides. Third Report of the WHO
Expert Committee on Vector Biology and Control, Geneva, World
Health Organization, pp. 18-23. (WHO Technical Report Series,
No. 634).
WHO (1988) The WHO recommended classification of pesticides by
hazard and guidelines to classification 1988-1989, Geneva, World
Health Organization (Unpublished document WHO/VBC/88.953).
WHO (1989) Health and Safety Guide No. 30: Deltamethrin, Geneva,
World Health Organization.
WHO (1984) Data sheet on pesticides, No. 50: Deltamethrin, Geneva,
World Health Organization, 9 pp (Unpublished document
VBC/DS/84.50).
WOOD MACKENZIE (1980) Agrochem. Monit., 9: 12.
WOOD MACKENZIE (1981) Agrochem. Monit., 15: 12.
WOOD MACKENZIE (1982) Agrochem. Monit., 21: 13.
WOOD MACKENZIE (1983) Agrochem. Monit., 27: 3-12.
WORTHING, C.R. & WALKER, S.B. (1983) Pesticide manual, 7th ed.,
Croydon, British Crop Protection Council, p. 161.
WOUTERS, W. & VAN DEN BERCKEN, J. (1978) Action of pyrethroids.
Gen. Pharmacol., 9: 387-398.
WRENN, J.M., RODWELL, D.E., GOLDENTHAL, E.I., SPIDER, E.J.C., &
RAJASEKARAN, D. (1980) Three-generation reproduction study in
rats, Matawan, Michigan, International Research and Development
Corporation (Unpublished report IDC-406 003/A4. submitted to WHO by
Roussel Uclaf).
ZHANG, L.Z., KHAN, S.U., AKHTAR, M.H., & IVARSON, K.C. (1984)
Persistence, degradation, and distribution of Deltamethrin in an
organic soil under laboratory conditions, J. agric. food Chem., 32:
1207-1211
ZITKO, V., MCLEESE, D.W., METCALFE, C.D., & CARSON, W.G. (1979)
Toxicity of permethrin, decamethrin, and related pyrethroids to
salmon and lobster. Bull. environ. Contam. Toxicol., 21: 338-343.
APPENDIX I
On the basis of electrophysiological studies with peripheral
nerve preparations of frogs (Xenopus laevis; Rana temporaria; and
Rana esculenta), it is possible to distinguish between 2 classes of
pyrethroid insecticides: (Type I and Type II). A similar
distinction between these 2 classes of pyrethroids has been made on
the basis of the symptoms of toxicity in mammals and insects (Van
den Bercken et al., 1979; WHO, 1979; Verschoyle & Aldridge, 1980;
Glickman & Casida, 1982; Lawrence & Casida, 1982). The same
distinction was found in studies on cockroaches (Gammon et al.,
1981).
Based on the binding assay on the gamma-aminobutyric acid
(GABA) receptor-ionophore complex, synthetic pyrethroids can also
be classified into two types: the alpha-cyano-3-phenoxybenzyl
pyrethroids and the non-cyano pyrethroids (Gammon et al., 1982;
Gammon & Casida, 1983; Lawrence & Casida, 1983; Lawrence et al.,
1985).
Pyrethroids that do not contain an alpha-cyano group
(allethrin, d-phenothrin, permethrin, tetramethrin, cismethrin, and
bioresmethrin) (Type I: T-syndrome)
The pyrethroids that do not contain an alpha-cyano group give
rise to pronounced repetitive activity in sense organs and in
sensory nerve fibres (Van den Bercken et al., 1973). At room
temperature, this repetitive activity usually consists of trains of
3 - 10 impulses and occasionally up to 25 impulses. Train duration
is between 10 and 5 milliseconds.
These compounds also induce pronounced repetitive firing of the
presynaptic motor nerve terminal in the neuromuscular junction (Van
den Bercken, 1977). There was no significant effect of the
insecticide on neurotransmitter release or on the sensitivity of
the subsynaptic membrane, nor on the muscle fibre membrane.
Presynaptic repetitive firing was also observed in the sympathetic
ganglion treated with these pyrethroids.
In the lateral-line sense organ and in the motor nerve
terminal, but not in the cutaneous touch receptor or in sensory
nerve fibres, the pyrethroid-induced repetitive activity increases
dramatically as the temperature is lowered, and a decrease of 5 °C
in temperature may cause a more than 3-fold increase in the number
of repetitive impulses per train. This effect is easily reversed
by raising the temperature. The origin of this "negative
temperature coefficient" is not clear (Vijverberg et al., 1983).
Synthetic pyrethroids act directly on the axon through
interference with the sodium channel gating mechanism that
underlies the generation and conduction of each nerve impulse. The
transitional state of the sodium channel is controlled by 2
separately acting gating mechanisms, referred to as the activation
gate and the inactivation gate. Since pyrethroids only appear to
affect the sodium current during depolarization, the rapid opening
of the activation gate and the slow closing of the inactivation
gate proceed normally. However, once the sodium channel is open,
the activation gate is restrained in the open position by the
pyrethroid molecule. While all pyrethroids have essentially the
same basic mechanism of action, however, the rate of relaxation
differs substantially for the various pyrethroids (Flannigan &
Tucker, 1985).
In the isolated node of Ranvier, allethrin causes prolongation
of the transient increase in sodium permeability of the nerve
membrane during excitation (Van den Bercken & Vijverberg, 1980).
Evidence so far available indicates that allethrin selectively
slows down the closing of the activation gate of a fraction of the
sodium channels that open during depolarization of the membrane.
The time constant of closing of the activation gate in the
allethrin-affected channels is about 100 milliseconds compared with
less than 100 microseconds in the normal sodium channel, i.e., it
is slowed down by a factor of more than 100. This results in a
marked prolongation of the sodium current across the nerve membrane
during excitation, and this prolonged sodium current is directly
responsible for the repetitive activity induced by allethrin
(Vijverberg et al., 1983).
The effects of cismethrin on synaptic transmission in the frog
neuromuscular junction, as reported by Evans (1976), are almost
identical to those of allethrin, i.e., presynaptic repetitive
firing, and no significant effects on transmitter release or on the
subsynaptic membrane.
Interestingly, the action of these pyrethroids closely
resembles that of the insecticide DDT in the peripheral nervous
system of the frog. DDT also causes pronounced repetitive activity
in sense organs, in sensory nerve fibres, and in motor nerve
terminals, due to a prolongation of the transient increase in
sodium permeability of the nerve membrane during excitation.
Recently was demonstrated that allethrin and DDT have essentially
the same effect on sodium channels in frog myelinated nerve
membrane. Both compounds slow down the rate of closing of a
fraction of the sodium channels that open on depolarization of the
membrane (Van den Bercken et al., 1973, 1979; Vijverberg et al.,
1982b).
In the electrophysiological experiments using giant axons of
crayfish, the type I pyrethroids and DDT analogues retain sodium
channels in a modified open state only intermittantly, cause large
depolarizing afterpotentials, and evoke repetitive firing with
minimal effect on the resting potential (Lund & Narahashi, 1983).
These results strongly suggest that permethrin and cismethrin,
like allethrin, primarily affect the sodium channels in the nerve
membrane and cause a prolongation of the transient increase in
sodium permeability of the membrane during excitation.
The effects of pyrethroids on end-plate and muscle action
potentials were studied in the pectoralis nerve-muscle preparation
of the clawed frog (Xenopus laevis). Type I pyrethroids
(allethrin, cismethrin, bioresmethrin, and 1R, cis-phenothrin)
caused moderate presynaptic repetitive activity, resulting in the
occurrence of multiple end-plate potentials (Ruigt & Van den
Bercken, 1986).
Pyrethroids with an alpha-cyano group on the 3-phenoxybenzyl
alcohol (deltamethrin, cypermethrin, fenvalerate, and fenpropanate)
(Type II: CS-syndrome)
The pyrethroids with an alpha-cyano group cause an intense
repetitive activity in the lateral line organ in the form of long-
lasting trains of impulses (Vijverberg et al., 1982a). Such a
train may last for up to 1 min and contains thousands of impulses.
The duration of the trains and the number of impulses per train
increase markedly on lowering the temperature. Cypermethrin does
not cause repetitive activity in myelinated nerve fibres. Instead,
this pyrethroid causes a frequency-dependent depression of the
nervous impulse, brought about by a progressive depolarization of
the nerve membrane as a result of the summation of depolarizing
after-potentials during train stimulation (Vijverberg & Van den
Bercken, 1979; Vijverberg et al., 1983).
In the isolated node of Ranvier, cypermethrin, like allethrin,
specifically affects the sodium channels of the nerve membrane and
causes a long-lasting prolongation of the transient increase in
sodium permeability during excitation, presumably by slowing down
the closing of the activation gate of the sodium channel
(Vijverberg & Van den Bercken, 1979; Vijverberg et al., 1983). The
time constant of closing of the activation gate in the
cypermethrin-affected channels is prolonged to more than 100
milliseconds. Apparently, the amplitude of the prolonged sodium
current after cypermethrin is too small to induce repetitive
activity in nerve fibres, but is sufficient to cause the long-
lasting repetitive firing in the lateral-line sense organ.
These results suggest that alpha-cyano pyrethroids primarily
affect the sodium channels in the nerve membrane and cause a long-
lasting prolongation of the transient increase in sodium
permeability of the membrane during excitation.
In the electrophysiological experiments using giant axons of
cray-fish, the Type II pyrethroids retain sodium channels in a
modified continuous open state persistently, depolarize the
membrane, and block the action potential without causing repetitive
firing (Lund & Narahashi, 1983).
Diazepam, which facilitates GABA reaction, delayed the onset of
action of deltamethrin and fenvalerate, but not permethrin and
allethrin, in both the mouse and cockroach. Possible mechanisms of
the Type II pyrethroid syndrome include action at the GABA receptor
complex or a closely linked class of neuroreceptor (Gammon et al.,
1982).
The Type II syndrome of intracerebrally administered pyrethroids
closely approximates that of the convulsant picrotoxin (PTX).
Deltamethrin inhibits the binding of [3H]-dihydropicrotoxin to rat
brain synaptic membranes, whereas the non-toxic R epimer of
deltamethrin is inactive. These findings suggest a possible
relation between the Type II pyrethroid action and the GABA receptor
complex. The stereospecific correlation between the toxicity of
Type II pyrethroids and their potency to inhibit the [35S]-TBPS
binding was established using a radioligand,
[35S]- t-butyl-bicyclophosphorothionate [35S]-TBPS. Studies
with 37 pyrethroids revealed an absolute correlation, without any
false positive or negative, between mouse intracerebral toxicity and
in vitro inhibition: all toxic cyano compounds including
deltamethrin, 1R, cis-cypermethrin, 1R, trans-cypermethrin, and
[2S, alpha S]-fenvalerate were inhibitors, but their non-toxic
stereoisomers were not; non-cyano pyrethroids were much less potent
or were inactive (Lawrence & Casida, 1983).
In the [35S]-TBPS and [3H]-Ro 5-4864 (a convulsant benzo-
diazepine radioligand) binding assay, the inhibitory potencies of
pyrethroids were closely related to their mammalian toxicities.
The most toxic pyrethroids of Type II were the most potent
inhibitors of [3H]-Ro 5-4864 specific binding to rat brain
membranes. The [3H]-dihydropicrotoxin and [35S]-TBPS binding
studies with pyrethroids strongly indicated that Type II effects of
pyrethroids are mediated, at least in part, through an interaction
with a GABA-regulated chloride ionophore-associated binding site.
Moreover, studies with [3H]-Ro 5-4864 support this hypothesis and,
in addition, indicate that the pyrethroid-binding site may be very
closely related to the convulsant benzodiazepine site of action
(Lawrence et al., 1985).
The Type II pyrethroids (deltamethrin, 1R, cis-cypermethrin and
[2S, alphaS]-fenvalerate) increased the input resistance of
crayfish claw opener muscle fibres bathed in GABA. In contrast,
two non-insecticidal stereoisomers and Type I pyrethroids
(permethrin, resmethrin, allethrin) were inactive. Therefore,
cyanophenoxybenzyl pyrethroids appear to act on the GABA receptor-
ionophore complex (Gammon & Casida, 1983).
The effects of pyrethroids on end-plate and muscle action
potentials were studied in the pectoralis nerve-muscle preparation
of the clawed frog (Xenopus laevis). Type II pyrethroids
(cypermethrin and deltamethrin) induced trains of repetitive muscle
action potentials without presynaptic repetitive activity.
However, an intermediate group of pyrethroids (1R-permethrin,
cyphenothrin, and fenvalerate) caused both types of effect. Thus,
in muscle or nerve membrane the pyrethroid induced repetitive
activities due to a prolongation of the sodium current. But no
clear distinction was observed between non-cyano and alpha-cyano
pyrethroids (Ruigt & Van den Bercken, 1986).
Appraisal
In summary, the results strongly suggest that the primary
target site of pyrethroid insecticides in the vertebrate nervous
system is the sodium channel in the nerve membrane. Pyrethroids
without an alpha-cyano group (allethrin, d-phenothrin, permethrin,
and cismethrin) cause a moderate prolongation of the transient
increase in sodium permeability of the nerve membrane during
excitation. This results in relatively short trains of repetitive
nerve impulses in sense organs, sensory (afferent) nerve fibres,
and, in effect, nerve terminals. On the other hand, the alpha-
cyano pyrethroids cause a long-lasting prolongation of the
transient increase in sodium permeability of the nerve membrane
during excitation. This results in long-lasting trains of
repetitive impulses in sense organs and a frequency-dependent
depression of the nerve impulse in nerve fibres. The difference in
effects between permethrin and cypermethrin, which have identical
molecular structures except for the presence of an alpha-cyano
group on the phenoxybenzyl alcohol, indicates that it is this
alpha-cyano group that is responsible for the long-lasting
prolongation of the sodium permeability.
Since the mechanisms responsible for nerve impulse generation
and conduction are basically the same throughout the entire nervous
system, pyrethroids may also induce repetitive activity in various
parts of the brain. The difference in symptoms of poisoning by
alpha-cyano pyrethroids, compared with the classical pyrethroids,
is not necessarily due to an exclusive central site of action.
It may be related to the long-lasting repetitive activity in
sense organs and possibly in other parts of the nervous system,
which, in a more advance state of poisoning, may be accompanied by
a frequency-dependent depression of the nervous impulse.
Pyrethroids also cause pronounced repetitive activity and a
prolongation of the transient increase in sodium permeability of
the nerve membrane in insects and other invertebrates. Available
information indicates that the sodium channel in the nerve membrane
is also the most important target site of pyrethroids in the
invertebrate nervous system (Wouters & Van den Bercken, 1978; WHO,
1979).
Because of the universal character of the processes underlying
nerve excitability, the action of pyrethroids should not be
considered restricted to particular animal species, or to a certain
region of the nervous system. Although it has been established
that sense organs and nerve endings are the most vulnerable to the
action of pyrethroids, the ultimate lesion that causes death will
depend on the animal species, environmental conditions, and on the
chemical structure and physical characteristics of the pyrethroid
molecule (Vijverberg & Van den Bercken, 1982).
1. RESUME ET EVALUATION, CONCLUSIONS ET RECOMMANDATIONS
1.1 Résumé et évaluation
1.1.1 Identité, propriétés physiques et chimiques, méthodes
d'analyse
La deltaméthrine a été synthétisée en 1974 et commercialisée
pour la première fois en 1977. Sur le plan chimique, c'est un
ester de l'analogue dibromé de l'acide chrysanthémique (acide
diméthyl-2,2(dimbromo-2,2 vinyl)-3 cyclopropanecarboxylique)
(Br2CA) et de l'alcol alpha-cyano-phénoxy-3 benzylique; plus
précisément, c'est l'isomère [1R, cis;alpha S] parmi les huit
stéréoisomères que compte cet ester.
La deltaméthrine de qualité technique se présente sous la forme
d'une poudre blanche inodore dont le point de fusion est de 98 -
101 °C et qui contient plus de 98% de deltaméthrine. Sa tension de
vapeur est de 2,0 x 10-6 Pa à 25 °C; autrement dit, elle n'est
pratiquement pas volatile. Insoluble dans l'eau, elle est en
revanche soluble dans des solvants organiques tels que l'acétone, la
cyclohexanone et le xylène. Elle est stable à la lumière, à la
chaleur et à l'air, mais instable en milieu alcalin.
Pour doser les résidus et analyser des échantillons prélevés
dans l'environnement, on procède à une extraction par solvant au
moyen d'un mélange n-hexane/acétone, à un partage entre le
n-hexane, l'acétone et l'eau, suivi d'une purification par
chromatographie sur colonne de gel de silice, le dosage final
s'effectuant par chromatographie en phase gazeuse avec détection
par capture d'électrons. La concentration minimale décelable par
cette méthode est de 0,01 mg/kg, voire moins. L'analyse des
produits s'effectue par chromatographie en phase liquide à haute
performance avec détecteur UV.
1.1.2 Production et usage
En 1987, la consommation mondiale de deltaméthrine était
d'environ 250 tonnes. On l'utilise essentiellement pour traiter le
coton (45% de la consommation) ainsi que pour les cultures telles
que le café, le maïs, les céréales, les fruits, les légumes, le
houblon et les produits stockés. On l'utilise aussi pour l'hygiène
des animaux, la lutte contre les vecteurs ainsi qu'à des fins de
santé publique. On l'applique soit seule, soit associée à d'autres
pesticides en formulations telles que concentrés émulsionnables,
concentrés pour pulvérisation à très bas volume, poudres
mouillables, concentrés pour suspension, poudres pour poudrage, etc.
1.1.3 Exposition humaine
L'exposition de la population dans son ensemble provient
principalement de la présence de résidus alimentaires mais peut
également résulter de son utilisation en santé publique. Sur les
récoltes correctement traitées, le taux de résidus est généralement
très faible sauf lorsque le traitement a eu lieu après la récolte.
La FAO et l'OMS ont passé en revue de nombreuses données sur ces
questions.
L'exposition de la population dans son ensemble est vrai-
semblablement très faible, mais on manque de données effectives
découlant d'études sur la ration alimentaire totale.
1.1.4 Exposition et destinée dans l'environnement
En exposant à la lumière solaire une fine pellicule de
deltaméthrine [1R, cis; alpha S] marquée au C14 au niveau de la
fonction acide, de la fonction alcool ou du groupe cyano, pendant 4
à 8 heures, on a constaté que 70 % du produit était transformé en
isomères [1R, trans alpha S] et [1S, trans alpha S] par
isomérisation cis/trans, accompagnés des produits de clivage de
l'ester, en particulier l'acide diméthyl-2,2(dibromovinyl-2,2)-3
cyclo-propane-carboxylique et l'alcool alpha-cyano-phénoxy-3
benzylique.
La deltaméthrine présente sur des plants de coton en serre se
dégrade avec une demi-vie initiale de 1,1 semaine, le taux de
dégradation étant de 90 % au bout de 4,5 semaines.
Les principaux métabolites consistent en Br2CA libre et
conjugué, en trans-hydroxyméthyl-Br2CA et en acide (hydroxy-4
phénoxy)-3 benzoïque résultant du clivage de l'ester, d'une
oxydation et d'une conjugaison.
Après avoir fait incuber de deltaméthrine dans du sable et du
terreau à 28 °C dans les conditions du laboratoire, on en a
récupéré respectivement 52 % et 74 %, huit semaines après ce
traitement.
La deltaméthrine ne se déplace pas dans l'environnement du fait
qu'elle est fortement adsorbée sur les particules, qu'elle est
insoluble dans l'eau et qu'elle est appliquée à très faibles doses.
On ne dispose pas de données sur les concentrations effectives
dans l'environnement, mais compte tenu des modalités actuelles
d'utilisation et pour peu que l'insecticide soit utilisé
normalement, l'exposition environnementale devrait être très
faible. La deltaméthrine se dégrade rapidement en produits moins
toxiques.
1.1.5 Absorption, métabolisme et excrétion
Administrée par voie orale, la deltraméthrine est facilement
absorbée; l'absorption est moindre par la voie percutanée encore
que sa vitesse dépende fortement du véhicule ou du solvant utilisé.
Une fois absorbée, la deltaméthrine est rapidement métabolisée et
excrétée.
On a administré à des rats par voie orale, à raison de 0,64 à
1,60 mg/kg, de la deltaméthrine marquée au carbone-14 au niveau des
fonctions acide, alcool et nitrile; le radiocarbone provenant de
la fraction acide et de la fraction alcoolique a été complètement
éliminé en l'espace de 2 à 4 jours. Les taux de résidus
tissulaires étaient généralement faibles sauf dans les graisses où
ils étaient un peu plus élevés. Cependant, le reste nitrile a été
excrété plus lentement, le taux global de récupération étant de
79 % en huit jours. Les principales réactions métaboliques étaient
l'oxydation (au niveau du méthyl- trans, du cycle cyclopropane et
des positions 2', 4' et 5' du reste alcoolique), le clivage de
l'ester et la conversion du nitrile en thiocyanate. Les acides
carboxyliques et les phénols résultants étaient conjugués à l'acide
sulfurique, à la glycine et à l'acide glucuronique.
Après avoir administré par voie orale à des souris de la
deltaméthrine marquée au 14C au niveau des fonctions acide, alcool
et nitrile à raison de 1,7 et 4,4 mg/kg, on a constaté que
l'excrétion du radiocarbone s'effectuait rapidement sauf dans le
cas du reste nitrile. Les principales réactions métaboliques
étaient en général analogues chez les souris et chez les rats.
Chez les vaches et la volaille, les voies de dégradation sont
tout à fait analogues à celles des rongeurs.
1.1.6 Effets sur les êtres vivants dans leur milieu naturel
La deltaméthrine est extrêmement toxique pour les poissons, la
CL50 à 96 heures allant de 0,4 à 2,0 µg/litre. Elle est également
très toxique pour les invertébrés aquatiques; pour la daphnie, la
CL50 à 48 heures est de 5 µg/litre. Toutefois des études
approfondies sur des étangs expérimentaux ainsi que les résultats
de l'utilisation en plein champ ont montré que cette forte toxicité
potentielle était inopérante. On a observé sur le terrain une
certaine mortalité parmi les invertébrés aquatiques mais les
populations se reconstituent généralement assez vite.
pour les oiseaux, la toxicité de la deltaméthrine est très
faible, les valeurs de la DL50 pour une dose unique par voie orale
étant supérieure à 1000 mg/kg. Au laboratoire, la deltaméthrine
est très toxique pour les abeilles, avec une DL50 de contact de
0,051 µg/abeille. Les essais en situation réelle et l'expérience
acquise dans l'utilisation effective du produit ont montré que les
formulations de deltaméthrine exerçaient une action répulsive, ce
qui signifie qu'en pratique le danger pour les abeilles est très
réduit.
1.1.7 Effets sur les animaux d'expérience et systemes d'epreuve
in vitro
Dans un véhicule non aqueux, la deltaméthrine présente une
toxicité aiguë par voie orale forte à modérée avec des DL50 de 19 à
34mg/kg chez la souris et de 39 à 139 mg/kg chez le rat.
Toutefois, en suspension dans l'eau, la toxicité est bien moidre,
les valeurs de la DL50 dépassant 5000 mg/kg chez le rat. La
deltaméthrine est un pyréthroide du Type II. Les signes cliniques
d'intoxication consistent en tremblements, salivation et
convulsions. L'intoxication est rapide et chez les survivants, les
signes disparaissent en quelques jours. L'électro-encéphalogramme
présente des décharges pointes-ondes généralisées qui précèdent
la choréo-athétose.
Une seule application de deltaméthrine technique n'a pas
produit d'effets irritants sur la peau intacte ou abrasée du lapin.
Toutefois elle a produit de effets irritants passagers au niveau de
l'oeil avec ou sans rinçage. La deltaméthrine n'a pas d'effet
sensibilisateur cutané chez le cobaye.
En administrant à des rats par gavage de la deltaméthrine à des
doses quotidiennes allant jusqu'à 10 mg/kg de poids corporel
pendant 13 semaines, on a provoqué chez ces animaux une
hyperexcitabilité qui se manifestait au bout de six semaines chez
les mâles recevant la dose la plus forte. Aux doses de 2,5 et
10 mg/kg, le gain de poids a été plus faible chez les mâles.
Chez des chiens beagle qui avaient reçu par voie orale de la
deltaméthrine en doses quotidiennes allant jusqu'à 10 mg/kg de
poids corporel pendant 13 semaines, on a observé divers symptômes
liés à cette substance, tels que des vomissements, des
tremblements, de la salivation et un affaiblissement du réflexe
pharyngé, du réflexe rotulien et du réflexe de flexion. Lors d'une
étude d'alimentation de deux ans sur des chiens, on a constaté que
la dose sans effet observable se situait à 1 mg/kg de poids
corporel par jour (dose la plus forte expérimentée).
L'administration de deltraméthrine à des souris à des doses
atteingnant 100 mg/kg de nourriture pendant 24 mois n'a pas modifié
l'incidence des tumeurs. La dose sans effet observé relative à la
toxicité générale était de 100 mg/kg de nourriture.
Chez des rats à qui l'on avait administré de la deltaméthrine
en doses allant jusqu'à 50 mg/kg de nourriture pendant deux ans, on
n'a observé aucune tumeur attribuable à cette substance. La dose
sans effet observé relative à la toxicité générale était de 5 mg/kg
de nourriture.
La deltaméthrine ne s'est pas révélée mutagène dans divers
systèmes d'épreuve in vivo et in vitro; notamment la réparation de
l'ADN, la mutation génique, les aberrations chromosomiques,
l'échange entre chromatides-soeurs, la formation de micronoyaux et
la létalité dominante.
Des études de tératogénicité ont été effectuées chez des rates
et des souris gravides à qui l'on administrait par voie orale des
doses quotidiennes de deltaméthrine allant jusqu'à 1000 mg/kg
pendant la phase principale de l'organogénèse. On n'a constaté
aucun effet tératogène ni altération de la fonction de reproduction
chez ces rates et ces souris, si ce n'est une réduction, liée à la
dose, dans le poids moyen des foetus chez les souris et un léger
retard d'ossification chez les rats.
Des lapines gravides ont reçu au sixième et dix-neuvième jours
de leur grossesse des doses quotidiennes de deltaméthrine allant
jusqu'à 16 mg/kg. A la dose la plus forte, on a noté une réduction
du poids moyen des foetus. Aucun effet tératogène n'a été observé
chez les lapins.
Après administration de deltaméthrine à des rats à des doses
allant jusqu'à 5 mg/kg de nourriture, dans le but d'effectuer une
étude de reproduction sur trois générations et deux portées, on a
constaté l'absence totale de tout effet.
Il existe certains indices selon lesquels l'association de
delta- méthrine à certains composés organophosphorés pourrai
conduire à une potentialisation de la toxicité de ces produits.
1.1.8 Effets sur l'homme
La deltaméthrine peut provoquer certaines sensations cutanées
chez des travailleurs exposés. Plusieurs cas d'intoxication
professionnelle non mortelle ont été signalés qui étaient dus à
l'inobservation des mesures de sécurité. Un engourdissement, un
prurit, des fourmillements et une sensation de brûlure de la peau
ainsi que des vertiges sont des symptômes fréquemment signalés. On
a décrit occasionnellement un érythème papulaire ou une couperose à
caractère passager. La plupart de ces symptômes sont passagers et
disparaissent en cinq à sept jours. Aucun effet indésirable à long
terme n'a été signalé. On a décrit trois cas d'empoisonnement non
mortel par la deltamétrine à la suite de l'ingestion de plusieurs
grammes de ce produit.
1.2 Conclusions
Population générale: l'exposition de la population générale à la
deltaméthrine est vraisemblablement très faible et ce produit,
lorsqu'il est utilisé conformément aux recommandations, ne présente
probablement aucun risque.
Exposition professionnelle: moyennant de bonnes méthodes de
travail et l'application de mesures d'hygiène et de sécurité, la
deltaméthrine ne devrait pas présenter de danger pour les personnes
qui y sont exposées de par leur profession.
Environnement: Utilisée aux doses recommandées, il est improbable
que la deltaméthrine ou ses produits de dégradation s'accumulent au
point d'avoir des effets nocifs sur l'environnement. Au
laboratoire, la deltaméthrine est très toxique pour les poissons,
les arthropodes aquatiques et les abeilles. Toutefois, sur le
terrain, il est peu probable que le produit ait des effets nocifs
s'il est utilisé conformément aux recommandations.
1.3 Recommandations
Bien que les teneurs dans les aliments soient très faibles
lorsque la deltaméthrine est utilisée conformément aux
recommandations, il est souhaitable de s'en assurer en soumettant
la deltaméthrine à une surveillance.
La deltaméthrine est utilisée depuis de nombreuses années et
l'on signale un certain nombre de cas d'intoxications non mortelles
ainsi que des effets passagers par suite d'exposition
professionnelle. Il est souhaitable que l'exposition humaine
continue d'être surveillée.
1. RESUMEN Y EVALUACION, CONCLUSIONES Y RECOMENDACIONES
1.1 Resumen y evaluación
1.1.1 Identidad, propiedades fíisicas y químicas, métodos de
análisis
La deltametrina fue sintetizada en 1974 y se comercializó por
primera vez en 1977. Químicamente, es el isómero [1R, cis; S] de 8
ésteres estereoisoméricos del análogo dibromo del ácido
crisantémico, ácido 2,2-dimetil-3-(2,2-dibromovinil) ciclopro-
panocarboxílico (Br2CA) con alcohol alpha-ciano-3-fenoxibencílico.
La deltametrina de calidad técnica es un polvo blanco inodoro
con un punto de fusión de 98 - 101 °C y contiene más del 98% del
material. La presión del vapor es 2,0 x 10-6 Pa a 25 °C y es
prácticamente no volátil. Es insoluble en agua, pero soluble en
disolventes orgánicos como la acetona, la ciclohexanona y el
xileno. Es estable a la luz, el calor y el aire, pero inestable en
medios alcalinos.
La determinación de la presencia de residuos y el análisis de
muestras ambientales se efectuaron por extracción con los
disolventes n-hexano/acetona, partición con n-hexano/acetona/agua,
absorción con un cromatógrafo de columna de gel de sílice y
determinación con un cromatógrafo de fase gaseosa equipado con un
detector de captura de electrones con una concentración mínima
detectable de 0,01 mg/kg o menos. Para el análisis de productos se
utiliza la cromatografía de fase líquida de alto rendimiento con un
detector de rayos UV.
1.1.2 Producción y empleo
El consumo mundial de deltametrina era de 250 toneladas
aproximadamente en 1987. La deltametrina se utiliza sobre todo en
el algodón (45% del consumo), en cultivos como el café, el maíz,
los cereales, la fruta, las hortalizas y el lúpulo, y en productos
almacenados. Se emplea también en higiene animal, en la lucha
contra los vectores y en salud pública. Se fabrica como solución
concentrada emulsionable, solución concentrada de pequeñísimo
volumen, polvo humectable, solución concentrada en suspensión y
polvo para utilización en seco, sola o en combinación con otros
plaguicidas.
1.1.3 Exposición humana
La exposición de la población en general a la deltametrina
procede principalmente de residuos en la dieta, pero puede resultar
también de su empleo en salud pública. En los cultivos tratados de
acuerdo con buenas prácticas agrícolas, las concentraciones
residuales son por lo general muy bajas, excepto en caso de
tratamiento después de la recolección. La FAO y la OMS han
examinado abundantes datos al respecto.
Se cree que la exposición de la población en general es muy
baja, pero no se dispone de datos en forma de estudios de dietas
totales.
1.1.4 Exposición ambiental y evolución
Cuando la deltametrina-[1R, cis; alpha S] marcada con
C14-(compuesto irradiado ácido, alcohol o ciano) se expuso a la
luz del sol en forma de una película fina durante 4 a 8 horas, el
70% se transformó por isomerización- cis/trans produciendo los
isómeros [1R, trans; alpha S] y [1S, trans; alpha S], junto con
productos de la escisión de los ésteres, entre ellos Br2CA y
alcohol alpha-ciano-3-fenoxibencílico.
La deltametrina se degradó en las plantas de algodón, en
condiciones de invernadero, con una vida media inicial de 1,1
semanas, y el tiempo necesario para una pérdida del 90% fue de 4,6
semanas.
Los principales metabolitos fueron Br2CA libre y combinado,
trans-hidroximetil-Br2CA y ácido 3-(4-hidroxifenoxi)benzoico,
formados por la escisión, la oxidación y la combinación de los
ésteres.
La deltametrina se incubó en arena y mantillo orgánico a 28 °C
en condiciones de laboratorio. Ocho semanas después del
tratamiento, se recuperó de la arena y del mantillo orgánico
alrededor del 52% y el 74%, respectivamente, de la deltametrina
aplicada.
La deltametrina no es móvil en el medio debido a su fuerte
adsorción en las partículas, su insolubilidad en el agua y sus
tasas muy bajas de aplicación.
No se dispone de datos sobre las concentraciones existentes en
el medio pero, dadas las modalidades actuales de uso y en
condiciones normales, es de esperar que la exposición ambiental sea
muy baja. Se degrada rápidamente, convirtiéndose en productos
menos tóxicos.
1.1.5 Ingestion, metabolismo y excreción
La deltametrina se absorbe fácilmente por vía oral y con menor
facilidad por la piel, pero la tasa de absorción depende
considerablemente del portador o disolvente. La deltametrina
absorbida se metaboliza y se excreta fácilmente.
En la administración por vía oral a ratas de deltametrina
marcada con C14-(compuesto irradiado ácido, alcohol o ciano), a
tasas de 0,64 - 1,60 mg/kg, el radiocarbono de las partes ácido y
alcohol se eliminó casi por completo en 2 - 4 días. Las
concentraciones residuales en los tejidos fueron por lo general muy
bajas, excepto en la grasa, en donde fueron ligeramente más altas.
Sin embargo, la parte ciano se excretó más lentamente, con una
recuperación total del 79% en 8 días. Las principales reacciones
metabólicas fueron oxidación (en el trans-metilo del anillo de
ciclopropano y en las posiciones 2'-, 4'-, y 5 de la parte
alcohol), escisión de ésteres y conversión de la parte ciano en
tiocianato. Los ácidos carboxílicos y fenoles resultantes se
combinaron con ácido sulfúrico, glicocola y ácido glucurónico.
Cuando se administró a ratones C14-(compuesto irradiado ácido,
alcohol o ciano)-deltametrina por vía oral, a tasas de 1,7 - 4,4
mg/kg, la excreción del radiocarbono fue rápida y casi total,
excepto en la parte ciano. Las principales reacciones metabólicas
observadas en los ratones fueron en general semejantes a las
observadas en las ratas.
En el ganado vacuno y las aves de corral, las vías de
degradación son muy similares a las observadas en los roedores.
1.1.6 Efectos en los organismos presentes en el medio
La deltametrina es muy tóxica para los peces, ya que la CL50 en
96 horas oscila entre 0,4 y 2,0 µg/litro. Es también muy tóxica
para los invertebrados acuáticos: la CL50 en 48 horas para Daphnia
es de 5 µg/litro. Sin embargo, extensos estudios realizados en
estanques experimentales y el empleo sobre el terreno han
demostrado que esa elevada toxicidad potencial no llega a
concretarse. En la práctica hay algunas muertes de invertebrados
acuáticos que, por lo general, se compensan con rapidez.
La toxicidad de la deltametrina en las aves es muy baja, con
valores de la DL50 superiores a 1000 mg/kg cuando se administra una
sola dosis por vía oral. En condiciones de laboratorio, la
deltametrina es muy tóxica para las abejas, con una DL50 por
contacto de 0,051 µg/abeja. Los ensayos sobre el terreno y el
empleo no experimental han demostrado que los preparados de
deltametrina tienen una acción repelente, lo cual quiere decir que,
en la práctica, el riesgo para las abejas es escaso.
1.1.7 Efectos en animales experimentales y en sistemas de pruebas
in vitro
En un vehículo no acuoso, la toxicidad de la deltametrina
administrada por vía oral en forma aguda va de alta a moderada, con
DL50 de 19 a 34 mg/kg (ratones) y de 31 a 139 mg/kg (ratas). Sin
embargo, cuando la deltametrina está en suspensión en agua, su
toxicidad es mucho menor con DL50 superiores a 5000 mg/kg (ratas).
La deltametrina es un piretroide de tipo II; los signos clínicos
comprenden temblor, salivación y convulsiones. La intoxicación se
presenta rápidamente y, en los supervivientes, los signos
desaparecen en unos días. El electroencefalograma muestra
descargas generalizadas en picos y ondas, seguidas de
coreoatetosis.
Aplicaciones únicas de deltametrina técnica no tuvieron ningún
efecto irritante en la piel, intacta o con abrasiones, de conejos.
Sin embargo, se observaron efectos irritantes transitorios en los
ojos de conejos, con y sin lavado. La deltametrina no actuó como
sensibilizante dérmico en cobayas.
En ratas a las que se administró deltametrina con sonda, en
dosis de hasta 10,0 mg/kg de peso corporal diarios durante 13
semanas, a las 6 semanas se observó hiperexcitabilidad en los
machos que habían recibido la dosis más alta. El aumento del peso
corporal en los machos fue menor con dosis de 2,5 y 10 mg/kg.
En perros de la raza beagle a los que se administró por vía
oral deltametrina en dosis de hasta 10 mg/kg de peso corporal
diarios durante 13 semanas, se observaron varios síntomas
relacionados con el compuesto, como vómitos, temblor, salivación y
reflejos faríngeo, patelar y flexor deprimidos. En un estudio de
alimentación en perros, de dos años de duración, la dosis
desprovista de efectos fue de 1 mg/kg de peso corporal diario
(dosis más alta estudiada).
En ratones a los que se administró deltametrina en dosis de
hasta 100 mg/kg de alimentos durante 24 meses, no resultó afectada
la incidencia de tumores. En cuanto a la toxicidad general, la
dosis desprovista de efectos fue de 100 mg/kg de alimentos.
En ratas a las que se administró deltametrina a niveles de
hasta 50 mg/kg de alimentos durante dos años, no se observaron
tumores relacionados con el compuesto. La concentración
desprovista de efectos de la toxicidad general fue de 50 mg/kg de
alimentos.
No se observaron efectos mutagénicos de la deltametrina en una
multitud de sistemas de pruebas in vivo e in vitro, incluidos:
reparación del ADN, mutación génica, aberración cromosómica,
intercambio de cromátidas hermanas, formación de micronúcleos y
genes letales dominantes.
Se realizaron estudios teratológicos con ratas y ratonas
preñadas, administrando deltametrina por vía oral en dosis de hasta
10 mg/kg diarias durante el periodo de mayor organogénesis. No se
observaron efectos teratogénicos ni reproductivos en las ratas ni
en las ratonas, excepto una disminución, relacionada con la dosis,
del peso fetal medio en el estudio con ratonas y una osificación
ligeramente retrasada en el estudio con ratas.
Se administró a conejas deltametrina en dosis de hasta 16 mg/kg
diarios entre los días 6 y 19 del embarazo. Con la dosis más alta,
se registró una disminución del peso fetal medio. No se observaron
efectos teratogénicos.
Se administró a ratas deltametrina en dosis de hasta 50 mg/kg
de alimentos en un estudio sobre reproducción con tres generaciones
y dos camadas, sin que se observaran efectos sobre la reproducción.
Hay indicios de que la toxicidad puede potenciarse cuando la
deltametrina se combina con algunos compuestos organofosforados.
1.1.8 Efectos en los seres humanos
La deltametrina puede provocar sensaciones cutáneas en los
trabajadores expuestos. Se han notificado varios casos de
intoxicación no mortal debidos a exposición ocupacional por no
respetar las precauciones de seguridad. Son síntomas
frecuentemente mencionados adormecimiento, picor, hormigueo y
quemazón de la piel, y vértigo. En ocasiones se ha descrito un
eritema papular o maculoso. La mayor parte de esos síntomas son
temporales y desaparecen en 5 ó 7 días. No se han comunicado
efectos negativos a largo plazo. Se han descrito tres casos no
mortales de intoxicación por deltametrina tras la ingestión de
varios gramos del producto.
1.2 Conclusiones
Población en general: No es probable que la exposición de la
población en general a la deltametrina, que se cree muy baja,
represente un riesgo en las condiciones de empleo recomendadas.
Exposición ocupacional: Si se aplican buenas prácticas laborales,
medidas de higiene y precauciones de seguridad, no es probable que
la deltametrina represente un riesgo para las personas
ocupacionalmente expuestas a ella.
Medio ambiente: Es improbable que la deltametrina o los productos
de su degradación alcancen niveles que puedan tener efectos
negativos en el medio con las tasas de aplicación recomendadas. En
condiciones de laboratorio, la deltametrina es muy tóxica para los
peces, los artrópodos acuáticos y las abejas. No obstante, en la
práctica, no es probable que haya efectos perjudiciales duraderos
con las condiciones de empleo recomendadas.
1.3 Recomendaciones
Aunque se cree que con el uso recomendado las concentraciones
en la dieta son muy bajas, debe considerarse la conveniencia de
confirmarlo incluyendo la deltametrina en estudios de vigilancia.
La deltametrina se utiliza desde hace muchos años y se han
notificado varios casos de intoxicación no mortal y efectos
transitorios debidos a exposición ocupacional. Debe mantenerse la
observación de la exposición humana.
